As
filed with the Securities and Exchange Commission on August 3, 2015.
Registration
No. 333-____________
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
WASHINGTON,
D.C. 20549
FORM
S-1
REGISTRATION
STATEMENT
UNDER
THE SECURITIES ACT OF 1933
SOLIGENIX,
INC.
(Exact
name of registrant as specified in its charter)
| Delaware |
|
2834 |
|
41-1505029 |
(State
or other jurisdiction of
incorporation
or organization) |
|
(Primary
Standard Industrial
Classification
Code Number) |
|
(I.R.S.
Employer
Identification
No.) |
Soligenix,
Inc.
29
Emmons Drive, Suite C-10
Princeton,
New Jersey 08540
(609)
538-8200
(Address,
including zip code, and telephone number, including area code,
of
registrant's principal executive offices)
Christopher
J. Schaber, Ph.D.
President
and Chief Executive Officer
Soligenix,
Inc.
29
Emmons Drive, Suite C-10
Princeton,
New Jersey 08540
(609)
538-8200
(Name,
address, including zip code, and telephone number,
including
area code, of agent for service)
with
copies to:
Leslie
J. Croland, Esq.
Duane
Morris LLP
200
South Biscayne Boulevard
Suite
3400
Miami,
Florida 33131-2318
(305)
960-2200
Approximate
date of commencement of proposed sale to the public: As soon as practicable after the effective date hereof.
If
any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under
the Securities Act of 1933 check the following box: ☒
If
this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, please
check the following box and list the Securities Act registration statement number of the earlier effective registration statement
for the same offering. ☐
If
this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list
the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If
this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list
the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
Indicate
by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller
reporting company. See definitions of "large accelerated filer," "accelerated filer," and "smaller reporting
company" in Rule 12b-2 of the Exchange Act. (Check one):
| Large
accelerated filer |
☐ |
Accelerated
filer |
☐
|
| Non-accelerated
filer |
☐ |
Smaller reporting
company |
☒ |
| (Do
not check if a smaller reporting company) |
|
|
CALCULATION
OF REGISTRATION FEE
Title of each class of securities to be registered | |
Amount to be registered
(1) (2) | | |
Proposed maximum offering price per share
(2) (3) | | |
Proposed maximum aggregate offering price
(2) (3) | | |
Amount of registration fee
(2) (3) | |
| Common Stock, $0.001 par value per share (4) | |
| 8,661,603 | | |
$ | 1.725 | | |
$ | 14,941,265.18 | | |
$ | 1,737 | |
| (1) |
The
shares of our common stock being registered hereunder are being registered for sale by the selling stockholders, as defined
in the accompanying prospectus. |
| (2) |
Pursuant
to Rule 416, the securities being registered hereunder include such indeterminate number of additional securities as may be
issuable to prevent dilution resulting from stock splits, stock dividends, or similar transactions. |
| (3)
|
Estimated
solely for purposes of calculating the registration fee according to Rule 457(c) under the Securities Act of 1933, as amended,
on the basis of the average of the bid and asked prices of the Registrant’s common stock reported on the OTCQB on July
28, 2015. |
| (4) |
This
registration statement also covers the preferred stock purchase rights issuable in accordance with the Rights Agreement, dated
June 22, 2007, between the Registrant and American Stock Transfer & Trust Company, LLC, as Rights Agent, which are presently
attached to and trade with the Registrant’s common stock. |
The
Registrant hereby amends this Registration Statement on such date or dates as may be necessary to delay its effective date until
the Registrant shall file a further amendment which specifically states that this Registration Statement shall thereafter become
effective in accordance with Section 8(a) of the Securities Act or until the Registration Statement shall become effective on
such date as the Commission, acting pursuant to Section 8(a), may determine.
The
information in this prospectus is not complete and may be changed. The selling stockholders shall not sell these securities until
the registration statement filed with the Securities and Exchange Commission is effective. This prospectus is not an offer to
sell these securities and it is not soliciting an offer to buy these securities in any state or other jurisdiction where the offer
or sale is not permitted.
| PRELIMINARY
PROSPECTUS |
SUBJECT
TO COMPLETION, DATED AUGUST 3, 2015 |
SOLIGENIX,
INC.
8,661,603
SHARES OF COMMON STOCK
This
prospectus relates to the offer and sale, from time to time, of up to 8,661,603 shares of the common stock of Soligenix, Inc.,
a Delaware corporation (“Soligenix,” “we,” “us,” and “our,”), by the selling stockholders
named in this prospectus in the section “Selling Stockholders,” whom we refer to in this document as the “selling
stockholders.” Of the shares of common stock being offered by the selling stockholders, 7,627,120 may be issued pursuant
to the equity purchase agreements that we entered into with Kodiak Capital Group, LLC (“Kodiak Capital”), Kingsbrook
Opportunities Master Fund LP (“Kingsbrook”) and River North Equity, LLC (“River North”), which we refer
to in this prospectus as the “Purchase Agreements.” Please refer to the section of this prospectus entitled “The
Equity Purchase Transactions” for a description of the Purchase Agreements and the section entitled “Selling Stockholders”
for additional information regarding the selling stockholders. Kodiak Capital, Kingsbrook and River North are sometimes referred
to herein collectively as the “Equity Purchasers” and individually as the “Equity Purchaser.”
We
are not selling any shares of common stock in this offering. We, therefore, will not receive any proceeds from the sale of the
shares by the selling stockholders. We will, however, receive proceeds from the sale of securities pursuant to our exercise of
the put right under the Purchase Agreements.
The
Equity Purchasers are “underwriters” within the meaning of the Section 2(a)(11) of the Securities Act of 1933, as
amended. The other selling stockholder may be deemed to be "underwriters" within the meaning of the Securities Act of
1933, as amended.
The
selling stockholders may sell common stock from time to time in the principal market on which the stock will be traded at the
prevailing market price or in negotiated transactions. See “Plan of Distribution” for more information about how the
selling stockholders may sell the shares of common stock being registered pursuant to this prospectus. The selling stockholders
have informed us that they do not have any agreement or understanding, directly or indirectly, with any person to distribute the
common stock.
We
have paid and will pay the expenses incurred in registering the shares, including legal and accounting fees. See “Plan of
Distribution.”
Our
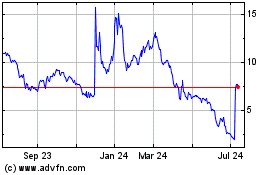
common stock is currently quoted on the OTCQB market under the symbol “SNGX”. On July 28, 2015, the last quoted sale
price of our common stock as reported on the OTCQB was $1.72 per share.
Investing
in our securities involves significant risks, including those set forth in the “Risk Factors” section of this prospectus
beginning on page 5.
Neither
the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or
passed upon the adequacy or accuracy of this prospectus. Any representation to the contrary is a criminal offense.
The
date of this prospectus is ___________________, 2015
Table
of Contents
You
should rely only on the information contained or incorporated by reference in this prospectus. We have not authorized anyone to
provide you with different information.
We
have not authorized the placement agent or any underwriters, brokers or dealers to make an offer of the securities in any jurisdiction
where the offer is not permitted.
You
should not assume that the information in this prospectus is accurate as of any date other than the date on the front of this
prospectus.
PROSPECTUS
SUMMARY
This
summary highlights certain information appearing elsewhere in this prospectus. For a more complete understanding of this offering,
you should read the entire prospectus carefully, including the risk factors and the financial statements. References in this prospectus
to “we,” “us,” “our,” and “Soligenix” refer to Soligenix, Inc. You should read
both this prospectus together with additional information described below under the heading "Where You Can Find More Information."
Business
Overview
We
are a late-stage biopharmaceutical company developing product candidates intended to address unmet medical needs in areas of inflammation,
oncology, and biodefense. We maintain two active business segments: BioTherapeutics and Vaccines/BioDefense.
Our
BioTherapeutics business segment is developing a first-in-class photo-dynamic therapy (SGX301) utilizing safe, visible light for
the treatment of cutaneous T-cell lymphoma (“CTCL”), proprietary formulations of oral beclomethasone 17,21-dipropionate
(“BDP”) for the prevention/treatment of gastrointestinal (“GI”) disorders characterized by severe inflammation,
including pediatric Crohn’s disease (SGX203) and acute radiation enteritis (SGX201), and our novel innate defense regulator
technology (SGX942) for the treatment of oral mucositis in head and neck cancer.
Our
Vaccines/BioDefense business segment includes active development programs for RiVax™, our ricin toxin vaccine candidate,
VeloThrax™, our anthrax vaccine candidate, OrbeShield™, our GI acute radiation syndrome (“GI ARS”) therapeutic
candidate and SGX943, our melioidosis therapeutic candidate. The development of our vaccine programs is supported by our heat
stabilization technology, known as ThermoVax™, under existing and on-going government contract funding. With the recently
awarded government contract from the National Institute of Allergy and Infectious Diseases (“NIAID”), we will attempt
to advance the development of RiVax™ to protect against exposure to ricin toxin. We plan to use the funds received under
our government contracts with the Biomedical Advanced Research and Development Authority (“BARDA”) and NIAID to advance
the development of OrbeShield™ for the treatment of GI ARS. Additionally, we have entered into a global and exclusive channel
collaboration with Intrexon Corporation (“Intrexon”) through which we intend to develop and commercialize a human
monoclonal antibody therapy (SGX101) to treat melioidosis.
An
outline for our business strategy follows:
| |
● |
Conduct
a Phase 3 clinical trial for SGX301 for the treatment of CTCL; |
| |
● |
Conduct
a Phase 2 clinical trial of SGX942 for the treatment of oral mucositis in head and neck cancer; |
| |
● |
Initiate
a Phase 3 clinical trial of oral BDP, known as SGX203, for the treatment of pediatric Crohn’s disease; |
| |
● |
Evaluate
the effectiveness of oral BDP in other therapeutic indications involving inflammatory conditions of the GI tract such as prevention
of acute radiation enteritis; |
| |
● |
Develop
RiVax™ and VeloThrax™ in combination with our ThermoVax™ technology, to develop new heat stable vaccines
in biodefense and infectious diseases with the potential to collaborate and/or partner with other companies in these areas; |
| |
● |
Advance
the preclinical and manufacturing development of OrbeShield™ as a biodefense medical countermeasure for the treatment
of GI ARS; |
| |
● |
Continue
to apply for and secure additional government funding for each of our BioTherapeutics and Vaccines/BioDefense programs through
grants, contracts and/or procurements; |
| |
● |
Acquire
or in-license new clinical-stage compounds for development; and |
| |
● |
Explore
other business development and merger/acquisition strategies, an example of which is our collaboration with Intrexon. |
Product
Candidates in Development
The
following tables summarize our product candidates under development:
| BioTherapeutic
Product Candidates |
| |
|
|
|
|
| Soligenix
Product Candidate |
|
Therapeutic
Indication |
|
Stage
of Development |
| SGX301 |
|
Cutaneous
T-Cell Lymphoma |
|
Phase
2 trial completed; demonstrated significantly higher response rate (p ≤ 0.04) compared
to placebo;
Phase
3 clinical trial planned for the second half of 2015, with data expected in the second half of 2016 |
| |
|
|
|
|
| SGX942 |
|
Oral
Mucositis in Head and Neck Cancer |
|
Phase
2 trial initiated in the second half of 2013, with data expected in the second half of 2015 |
| |
|
|
|
|
| SGX203** |
|
Pediatric
Crohn’s disease |
|
Phase
1/2 clinical trial completed June 2013, efficacy data, pharmacokinetic (PK)/pharmacodynamic
(PD) profile and safety confirmed;
Phase
3 clinical trial planned for the second half of 2015, with data expected in the second half of 2017 |
| |
|
|
|
|
| SGX201** |
|
Acute
Radiation Enteritis |
|
Phase
1/2 clinical trial complete;
safety
and preliminary efficacy demonstrated;
Phase
2 trial planned for the first half of 2016,
with
data expected in the first half of 2017 |
| |
|
|
|
|
| Vaccine
Thermostability Platform** |
| |
|
|
|
|
| Soligenix
Product Candidate |
|
Indication |
|
Stage
of Development |
| ThermoVax™ |
|
Thermostability
of aluminum adjuvanted vaccines |
|
Pre-clinical |
| |
|
|
|
|
| BioDefense
Product Candidates** |
| |
|
|
|
|
| Soligenix
Product Candidate |
|
Indication |
|
Stage
of Development |
| RiVax™ |
|
Vaccine
against
Ricin
Toxin Poisoning |
|
Phase
1B trial complete, safety and neutralizing antibodies for protection demonstrated;
Phase
1/2 trial planned for the second half of 2015 |
| |
|
|
|
|
| VeloThrax™ |
|
Vaccine
against Anthrax Poisoning |
|
Pre-clinical;
Phase
1 clinical trial planned for second half of 2016 |
| |
|
|
|
|
| OrbeShield™ |
|
Therapeutic
against GI ARS |
|
Pre-clinical
program initiated |
| |
|
|
|
|
| SGX943/SGX101 |
|
Melioidosis |
|
Pre-clinical
program initiated |
| ** |
Contingent upon continued government contract and grant funding. |
Corporate
Information
We
were incorporated in Delaware in 1987 under the name Biological Therapeutics, Inc. In 1987, we merged with Biological Therapeutics,
Inc., a North Dakota corporation, pursuant to which we changed our name to “Immunotherapeutics, Inc.” We changed our
name to “Endorex Corp.” in 1996, to “Endorex Corporation” in 1998, to “DOR BioPharma, Inc.”
in 2001, and finally to “Soligenix, Inc.” in 2009. Our principal executive offices are located at 29 Emmons Drive,
Suite C-10, Princeton, New Jersey 08540 and our telephone number is (609) 538-8200.
The
Offering
This
prospectus relates to the offer and sale from time to time of up to 8,661,603 shares of our common stock by the selling stockholders,
1,034,483 shares of which were issued to Intrexon Corporation in a private placement on April 27, 2013 as consideration for the
execution and delivery of a collaboration agreement.
Kodiak
Capital, Kingsbrook and River North, three of the selling stockholders under this prospectus, are offering for sale up to 3,389,831
shares, 3,389,831 shares and 847,458 shares, respectively, of our common stock. None of Kodiak Capital, Kingsbrook or River North
is an affiliate of, or has any relation to, any of the other selling stockholders named herein. On July 29, 2015, we entered into
the Purchase Agreements with Kodiak Capital, Kingsbrook and River North. Pursuant to the Purchase Agreements, Kodiak Capital,
Kingsbrook and River North have agreed to purchase from us up to an aggregate of $5 million, $4 million and $1 million, respectively,
worth of shares of our common stock from time to time, until December 31, 2016. Also on July 29, 2015, we entered into Registration
Rights Agreements (the “Registration Rights Agreements”) with the Equity Purchasers, pursuant to which we have filed
with the U.S. Securities and Exchange Commission (the “SEC”) the registration statement that includes this prospectus
to register for resale under the Securities Act of 1933, as amended (the “Securities Act”), the shares that may be
issued to the Equity Purchasers under the Purchase Agreements. In consideration for entering into the Purchase Agreements, we
issued to each of the Equity Purchasers a promissory note having a principal amount equal to 3% of the amount committed by it,
which are payable April 15, 2016.
We
do not have the right to commence any sales to the Equity Purchasers under the Purchase Agreements until the SEC has declared
effective the registration statement of which this prospectus forms a part. Thereafter, we may, from time to time and at our sole
discretion, direct the Equity Purchasers to purchase shares of our common stock, but we would be unable to sell shares to them
if such purchase would result in their respective beneficial ownership equaling more than 9.99% of the outstanding common stock.
Except as described in this prospectus, there are no trading volume requirements or restrictions under the Purchase Agreements,
and we will control the timing and amount of any sales of our common stock to the Equity Purchasers. The purchase price of the
shares that may be sold to the Equity Purchasers under the Purchase Agreements will be equal to 80% of the lowest daily volume
weighted average price of the common stock for the five consecutive trading days immediately following our request for the Equity
Purchasers to purchase the shares. We may at any time in our sole discretion terminate the Purchase Agreements without fee, penalty
or cost upon one business day notice. None of the Equity Purchasers may assign or transfer its rights and obligations under the
Purchase Agreements.
As
of July 28, 2015, there were 26,381,976 shares of our common stock outstanding, of which 18,798,079 shares were held by non-affiliates.
Although the Purchase Agreements provide that we may sell up to $5 million, $4 million and $1 million worth of shares of our common
stock to Kodiak Capital, Kingsbrook and River North, respectively, only 7,627,120 shares of our common stock are being offered
under this prospectus. If all of the 7,627,120 shares offered by the Equity Purchasers under this prospectus were issued and outstanding
as of the date hereof, such shares would represent 22.43% of the total number of shares of our common stock outstanding and 28.87%
of the total number of outstanding shares held by non-affiliates, in each case as of the date hereof. If we elect to issue and
sell more than the 7,627,120 shares offered under this prospectus to the Equity Purchasers, which we have the right, but not the
obligation, to do, we must first register for resale under the Securities Act any such additional shares, which could cause additional
substantial dilution to our stockholders. The number of shares ultimately offered for resale by the Equity Purchasers is dependent
upon the number of shares we sell to them under the Purchase Agreements.
Issuances
of our common stock in this offering will not affect the rights or privileges of our existing stockholders, except that the economic
and voting interests of each of our existing stockholders will be diluted as a result of any such issuance. Although the number
of shares of common stock that our existing stockholders own will not decrease, the shares owned by our existing stockholders
will represent a smaller percentage of our total outstanding shares after any such issuance to the Equity Purchasers.
Securities
Offered
| Common
stock offered by the selling stockholders: |
|
8,661,603
shares, including 7,627,120 that we may sell to the Equity Purchasers under the Purchase Agreements. |
| |
|
|
| Common
stock outstanding prior to the offering: |
|
26,381,976
shares. |
| |
|
|
| Common
stock to be outstanding after giving effect to the total issuance of 7,627,120 shares to the Equity Purchasers under the Purchase
Agreements registered hereunder: |
|
34,009,096
shares. |
| |
|
|
| Shares
issuable upon exercise of outstanding options and warrants: |
|
The
total number of shares of our common stock outstanding prior to the offering and to be outstanding after giving effect to the
total issuance of 7,627,120 shares to the Equity Purchasers under the Purchase Agreements registered hereunder, excludes the following:
●
329,397 shares of common stock reserved for future issuance under our equity incentive plans. As of the date of this prospectus,
there were options to purchase 2,338,237 shares of our common stock outstanding under our equity incentive plans with
a weighted average exercise price of $2.31 per share; and
●
4,941,119 shares of common stock issuable upon exercise of outstanding warrants as of the date of this prospectus with
a weighted average exercise price of $0.82 per share. |
| |
|
|
| Use
of proceeds: |
|
We
will not receive any proceeds from the sale of the shares of common stock by the selling stockholders in this offering. However,
we may receive up to $10 million from sales of shares to the Equity Purchasers under the Purchase Agreements. Any proceeds
that we receive from sales to the Equity Purchasers under the Purchase Agreements will be used to further develop our late-stage
product candidates and for general corporate purposes. See “Use of Proceeds.” |
| |
|
|
| Risk
factors: |
|
This
investment involves a high degree of risk. See “Risk Factors” for a discussion of factors you should consider
carefully before making an investment decision. |
| |
|
|
| OTC
Markets (OTCQB) symbol: |
|
SNGX |
RISK
FACTORS
An
investment in our securities involves a high degree of risk. You should carefully consider the following information about these
risks, together with the other information about these risks contained in this prospectus, as well as the other information contained
in this prospectus generally, before deciding to buy our securities. Any of the risks we describe below could adversely affect
our business, financial condition, operating results or prospects. The market prices for our securities could decline if one or
more of these risks and uncertainties develop into actual events and you could lose all or part of your investment. Additional
risks and uncertainties that we do not yet know of, or that we currently think are immaterial, may also impair our business operations.
You should also refer to the other information contained in this prospectus, including our financial statements and the related
notes.
Risks
Related to our Business
We
have had significant losses and anticipate future losses; if additional funding cannot be obtained, we may reduce or discontinue
our product development and commercialization efforts.
We
have experienced significant losses since inception and, at March 31, 2015, had an accumulated deficit of approximately $143.6
million. We expect to incur additional operating losses in the future and expect our cumulative losses to increase. As of March
31, 2015, we had approximately $5.0 million in cash available. Based on our projected budgetary needs, funding from existing contracts
and grants over the next two years and sales to Lincoln Park Capital Fund, LLC (“Lincoln Park”) under our $10.6 million
equity facility and to the Equity Purchasers under the $10 million Purchase Agreements, we expect to be able to maintain the current
level of our operations for at least the next twelve months.
We
have sufficient funds through our existing biodefense grant facilities from the NIAID, a division of the National Institutes of
Health (the “NIH”), and BARDA to finance our biodefense projects for the next six years. In September 2014, we entered
into a contract with the NIH for the development of RiVaxTM to protect against exposure to ricin toxin that would provide
up to $24.7 million of funding in the aggregate if options to extend the contract are exercised by the NIH. In September 2013,
we entered into contracts with the NIH and BARDA for the development of OrbeShield™ that would provide up to $32.7 million
of funding in the aggregate if options to extend the contracts are exercised by BARDA and the NIH. In September 2009, we received
a NIAID grant for approximately $9.4 million for the development of our biodefense programs. In July 2012, we received an additional
Small Business Innovation and Research (“SBIR”) grant from NIAID for $600,000 and in February 2014, we were awarded
a one-year NIAID SBIR grant award of approximately $300,000 to further evaluate SGX943 as a treatment for melioidosis. Our biodefense
grants have an overhead component that allows us an agency-approved percentage over our incurred costs. We estimate that the overhead
component associated with our existing contracts and grants will fund some fixed costs for direct employees working on these contracts
and grants as well as other administrative costs.
Our
product candidates are positioned for or are currently in clinical trials, and we have not yet generated any significant revenues
from sales or licensing of these product candidates. From inception through March 2015, we have expended approximately $61.9 million
developing our current product candidates for pre-clinical research and development and clinical trials, and we currently expect
to spend at least $16.1 million over the next twelve months in connection with the development of our therapeutic and vaccine
products, licenses, employment agreements, and consulting agreements of which approximately $10.4 million will be reimbursed through
our existing government contracts and grants. Unless and until we are able to generate sales or licensing revenue from one of
our product candidates, we will require additional funding to meet these commitments, sustain our research and development efforts,
provide for future clinical trials, and continue our operations. There can be no assurance we can raise such funds. If additional
funds are raised through the issuance of equity securities, stockholders may experience dilution of their ownership interests,
and the newly issued securities may have rights superior to those of the common stock. If additional funds are raised by the issuance
of debt, we may be subject to limitations on our operations. If we cannot raise such additional funds, we may have to delay or
stop some or all of our drug development programs.
If
we are unable to develop our product candidates, our ability to generate revenues and viability as a company will be significantly
impaired.
In
order to generate revenues and profits, our organization must, along with corporate partners and collaborators, positively research,
develop and commercialize our technologies or product candidates. Our current product candidates are in various stages of early
clinical and pre-clinical development and will require significant further funding, research, development, pre-clinical and/or
clinical testing, regulatory approval and commercialization, and are subject to the risks of failure inherent in the development
of products based on innovative or novel technologies. Specifically, each of the following is possible with respect to any of
our product candidates:
| |
● |
we
may not be able to maintain our current research and development schedules; |
| |
● |
we
may be unable to secure procurement contracts on beneficial economic terms or at all from the U.S. government or others for
our biodefense products; |
| |
● |
we
may encounter problems in clinical trials; or |
| |
● |
the
technology or product may be found to be ineffective or unsafe, or may fail to obtain marketing approval. |
If
any of the risks set forth above occur, or if we are unable to obtain the necessary regulatory approvals as discussed below, we
may be unable to develop our technologies and product candidates and our business will be seriously harmed. Furthermore, for reasons
including those set forth below, we may be unable to commercialize or receive royalties from the sale of any other technology
we develop, even if it is shown to be effective, if:
| |
● |
it
is not economical or the market for the product does not develop or diminishes; |
| |
● |
we
are not able to enter into arrangements or collaborations to manufacture and/or market the product; |
| |
● |
the
product is not eligible for third-party reimbursement from government or private insurers; |
| |
● |
others
hold proprietary rights that preclude us from commercializing the product; |
| |
● |
we
are not able to manufacture the product reliably; |
| |
● |
others
have brought to market similar or superior products; or |
| |
● |
the
product has undesirable or unintended side effects that prevent or limit its commercial use. |
We
expect a number of factors to cause our operating results to fluctuate on a quarterly and annual basis, which may make it difficult
to predict our future performance.
We
are a late-stage biopharmaceutical company. Our operations to date have been primarily limited to developing our technology and
undertaking pre-clinical studies and clinical trials of our product candidates in our two active business segments, BioTherapeutics
and Vaccines/BioDefense. We have not yet obtained regulatory approvals for any of our product candidates. Consequently, any predictions
made about our future success or viability may not be as accurate as they could be if we had commercialized products. Our financial
condition has varied significantly in the past and will continue to fluctuate from quarter-to-quarter or year-to-year due to a
variety of factors, many of which are beyond our control. Factors relating to our business that may contribute to these fluctuations
include other factors described elsewhere in this prospectus and also include:
| |
● |
our
ability to obtain additional funding to develop our product candidates; |
| |
● |
delays
in the commencement, enrollment and timing of clinical trials; |
| |
● |
the
success of our product candidates through all phases of clinical development; |
| |
● |
any
delays in regulatory review and approval of product candidates in clinical development; |
| |
● |
our
ability to obtain and maintain regulatory approval for our product candidates in the United States and foreign jurisdictions; |
| |
● |
potential
side effects of our product candidates that could delay or prevent commercialization, limit the indications for any approved
drug, require the establishment of risk evaluation and mitigation strategies, or cause an approved drug to be taken off the
market; |
| |
● |
our
dependence on third-party contract manufacturing organizations (“CMOs”) to supply or manufacture our products; |
| |
● |
our
dependence on contractors to conduct our clinical trials; |
| |
● |
our
ability to establish or maintain collaborations, licensing or other arrangements; |
| |
● |
market
acceptance of our product candidates; |
| |
● |
our
ability to establish and maintain an effective sales and marketing infrastructure, either through the creation of a commercial
infrastructure or through strategic collaborations; |
| |
● |
competition
from existing products or new products that may emerge; |
| |
● |
the
ability of patients or healthcare providers to obtain coverage of or sufficient reimbursement for our products; |
| |
● |
our
ability to discover and develop additional product candidates; |
| |
● |
our
ability and our licensors’ abilities to successfully obtain, maintain, defend and enforce intellectual property rights
important to our business; |
| |
● |
our
ability to attract and retain key personnel to manage our business effectively; |
| |
● |
our
ability to build our finance infrastructure and improve our accounting systems and controls; |
| |
● |
potential
product liability claims; |
| |
● |
potential
liabilities associated with hazardous materials; and |
| |
● |
our
ability to obtain and maintain adequate insurance policies. |
Accordingly,
the results of any quarterly or annual periods should not be relied upon as indications of future operating performance.
We
have no approved products on the market and therefore do not expect to generate any revenues from product sales in the foreseeable
future, if at all.
To
date, we have no approved product on the market and have not generated any significant product revenues. We have funded our operations
primarily from sales of our securities and from government grants. We have not received, and do not expect to receive for at least
the next several years, if at all, any revenues from the commercialization of our product candidates. To obtain revenues from
sales of our product candidates, we must succeed, either alone or with third parties, in developing, obtaining regulatory approval
for, manufacturing and marketing drugs with commercial potential or successfully obtain government procurement or stockpiling
agreements. We may never succeed in these activities, and we may not generate sufficient revenues to continue our business operations
or achieve profitability.
Our
business is subject to extensive governmental regulation, which can be costly, time consuming and subjects us to unanticipated
delays.
Our
business is subject to very stringent federal, foreign, state and local government laws and regulations, including the Federal
Food, Drug and Cosmetic Act, the Environmental Protection Act, the Occupational Safety and Health Act, and state and local counterparts
to these acts. These laws and regulations may be amended, additional laws and regulations may be enacted, and the policies of
the U.S. Food and Drug Administration (the “FDA”) and other regulatory agencies may change.
The
regulatory process applicable to our products requires pre-clinical and clinical testing of any product to establish its safety
and efficacy. This testing can take many years is uncertain as to outcome, and requires the expenditure of substantial capital
and other resources. We estimate that the clinical trials of our product candidates that we have planned will take at least several
years to complete. Furthermore, failure can occur at any stage of the trials, and we could encounter problems that cause us to
abandon or repeat clinical trials. Favorable results in early studies or trials, if any, may not be repeated in later studies
or trials. Even if our clinical trials are initiated and completed as planned, we cannot be certain that the results will support
our product candidate claims. Success in preclinical testing, Phase 1 and Phase 2 clinical trials does not ensure that later Phase
2 or Phase 3 clinical trials will be successful. In addition, we, the FDA or other regulatory authorities may suspend clinical
trials at any time if it appears that we are exposing participants to unacceptable health risks or the FDA or other regulatory
authorities find deficiencies in our submissions or conduct of our trials.
We
may not be able to obtain, or we may experience difficulties and delays in obtaining, necessary domestic and foreign governmental
clearances and approvals to market a product. Also, even if regulatory approval of a product is granted, that approval may entail
limitations on the indicated uses for which the product may be marketed.
Following
any regulatory approval, a marketed product and its manufacturer are subject to continual regulatory review. Later discovery of
problems with a product or manufacturer may result in restrictions on such product or manufacturer. These restrictions may include
product recalls and suspension or withdrawal of the marketing approval for the product. Furthermore, the advertising, promotion
and export, among other things, of a product are subject to extensive regulation by governmental authorities in the U.S. and other
countries. If we fail to comply with applicable regulatory requirements, we may be subject to fines, suspension or withdrawal
of regulatory approvals, product recalls, seizure of products, operating restrictions and/or criminal prosecution.
There
may be unforeseen challenges in developing our biodefense products.
For
development of biodefense vaccines and therapeutics, the FDA has instituted policies that are expected to result in accelerated
approval. This includes approval for commercial use using the results of animal efficacy trials, rather than efficacy trials in
humans, referred to as the Animal Rule. However, we will still have to establish that the vaccines we are developing are safe
in humans at doses that are correlated with the beneficial effect in animals. Such clinical trials will also have to be completed
in distinct populations that are subject to the countermeasures; for instance, the very young and the very old, and in pregnant
women, if the countermeasure is to be licensed for civilian use. Other agencies will have an influence over the risk benefit scenarios
for deploying the countermeasures and in establishing the number of doses utilized in the Strategic National Stockpile. We may
not be able to sufficiently demonstrate the animal correlation to the satisfaction of the FDA, as these correlates are difficult
to establish and are often unclear. Invocation of the Animal Rule may raise issues of confidence in the model systems even if
the models have been validated. For many of the biological threats, the animal models are not available and we may have to develop
the animal models, a time-consuming research effort. There are few historical precedents, or recent precedents, for the development
of new countermeasure for bioterrorism agents. Despite the Animal Rule, the FDA may require large clinical trials to establish
safety and immunogenicity before licensure and it may require safety and immunogenicity trials in additional populations. Approval
of biodefense products may be subject to post-marketing studies, and could be restricted in use in only certain populations. The
government’s biodefense priorities can change, which could adversely affect the commercial opportunity for the products
we are developing. Further, other countries have not, at this time, established criteria for review and approval of these types
of products outside their normal review process, i.e., there is no Animal Rule equivalent, and consequently there can be no assurance
that we will be able to make a submission for marketing approval in foreign countries based on such animal data.
Additionally,
few facilities in the United States and internationally have the capability to test animals with anthrax or ricin, or otherwise
assist us in qualifying the requisite animal models. We have to compete with other biodefense companies for access to this limited
pool of highly specialized resources. We therefore may not be able to secure contracts to conduct the testing in a predictable
timeframe or at all.
We
are dependent on government funding, which is inherently uncertain, for the success of our biodefense operations.
We
are subject to risks specifically associated with operating in the biodefense industry, which is a new and unproven business area.
We do not anticipate that a significant commercial market will develop for our biodefense products. Because we anticipate that
the principal potential purchasers of these products, as well as potential sources of research and development funds, will be
the U.S. government and governmental agencies, the success of our biodefense division will be dependent in large part upon government
spending decisions. The funding of government programs is dependent on budgetary limitations, congressional appropriations and
administrative allotment of funds, all of which are inherently uncertain and may be affected by changes in U.S. government policies
resulting from various political and military developments. Our receipt of government funding is also dependent on our ability
to adhere to the terms and provisions of the original grant documents and other regulations. We can provide no assurance that
we will receive or continue to receive funding for grants we have been awarded. The loss of government funds could have a material
adverse effect on our ability to progress our biodefense business.
If
the parties we depend on for supplying our drug substance raw materials and certain manufacturing-related services do not timely
supply these products and services, it may delay or impair our ability to develop, manufacture and market our products. We do
not have or anticipate having internal manufacturing capabilities.
We
rely on suppliers for our drug substance raw materials and third parties for certain manufacturing-related services to produce
material that meets appropriate content, quality and stability standards, which material will be used in clinical trials of our
products and, after approval, for commercial distribution. To succeed, clinical trials require adequate supplies of drug substance
and drug product, which may be difficult or uneconomical to procure or manufacture. We and our suppliers and vendors may not be
able to (i) produce our drug substance or drug product to appropriate standards for use in clinical studies, (ii) perform under
any definitive manufacturing, supply or service agreements with us or (iii) remain in business for a sufficient time to be able
to develop, produce, secure regulatory approval of and market our product candidates. If we do not maintain important manufacturing
and service relationships, we may fail to find a replacement supplier or required vendor or develop our own manufacturing capabilities
which could delay or impair our ability to obtain regulatory approval for our products and substantially increase our costs or
deplete profit margins, if any. If we do find replacement manufacturers and vendors, we may not be able to enter into agreements
with them on terms and conditions favorable to us and, there could be a substantial delay before a new facility could be qualified
and registered with the FDA and foreign regulatory authorities.
The
manufacturing of our products is a highly exacting process, and if we or one of our materials suppliers encounter problems manufacturing
our products, our business could suffer.
The
FDA and foreign regulators require manufacturers to register manufacturing facilities. The FDA and foreign regulators also inspect
these facilities to confirm compliance with current Good Manufacturing Practice (“cGMP”) or similar requirements that
the FDA or foreign regulators establish. We, or our materials suppliers, may face manufacturing or quality control problems causing
product production and shipment delays or a situation where we or the supplier may not be able to maintain compliance with the
FDA’s cGMP requirements, or those of foreign regulators, necessary to continue manufacturing our drug substance. Any failure
to comply with cGMP requirements or other FDA or foreign regulatory requirements could adversely affect our clinical research
activities and our ability to market and develop our products.
We
may use our financial and human resources to pursue a particular research program or product candidate and fail to capitalize
on programs or product candidates that may be more profitable or for which there is a greater likelihood of success.
Because
we have limited financial and human resources, we are currently focusing on the regulatory approval of certain product candidates.
As a result, we may forego or delay pursuit of opportunities with other product candidates or for other indications that later
prove to have greater commercial potential. Our resource allocation decisions may cause us to fail to capitalize on viable commercial
products or profitable market opportunities. Our spending on existing and future product candidates for specific indications may
not yield any commercially viable products. If we do not accurately evaluate the commercial potential or target market for a particular
product candidate, we may relinquish valuable rights to that product candidate through strategic alliance, licensing or other
royalty arrangements in cases in which it would have been more advantageous for us to retain sole development and commercialization
rights to such product candidate, or we may allocate internal resources to a product candidate in an area in which it would have
been more advantageous to enter into a partnering arrangement.
Even
if approved, our products will be subject to extensive post-approval regulation.
Once
a product is approved, numerous post-approval requirements apply. Among other things, the holder of an approved New Drug Application
(“NDA”) is subject to periodic and other FDA monitoring and reporting obligations, including obligations to monitor
and report adverse events and instances of the failure of a product to meet the specifications in the NDA. Application holders
must submit new or supplemental applications and obtain FDA approval for certain changes to the approved product, product labeling,
or manufacturing process. Application holders must also submit advertising and other promotional material to the FDA and report
on ongoing clinical trials.
Depending
on the circumstances, failure to meet these post-approval requirements can result in criminal prosecution, fines, injunctions,
recall or seizure of products, total or partial suspension of production, denial or withdrawal of pre-marketing product approvals,
or refusal to allow us to enter into supply contracts, including government contracts. In addition, even if we comply with FDA
and other requirements, new information regarding the safety or effectiveness of a product could lead the FDA to modify or withdraw
product approval.
Even
if we obtain regulatory approval to market our product candidates, our product candidates may not be accepted by the market.
Even
if the FDA approves one or more of our product candidates, physicians and patients may not accept it or use it. Even if physicians
and patients would like to use our products, our products may not gain market acceptance among healthcare payors such as managed
care formularies, insurance companies or government programs such as Medicare or Medicaid. Acceptance and use of our products
will depend upon a number of factors including: perceptions by members of the health care community, including physicians, about
the safety and effectiveness of our drug or device product; cost-effectiveness of our product relative to competing products;
availability of reimbursement for our product from government or other healthcare payers; and effectiveness of marketing and distribution
efforts by us and our licensees and distributors, if any.
The
degree of market acceptance of any product that we develop will depend on a number of factors, including:
| |
● |
the
safety and effectiveness of our products, including any significant potential side effects, as compared to alternative products
or treatment methods; |
| |
● |
the
timing of market entry as compared to competitive products; |
| |
● |
the
rate of adoption of our products by doctors and nurses; |
| |
● |
product
labeling or product insert required by the FDA for each of our products; |
| |
● |
reimbursement
policies of government and third-party payors; |
| |
● |
effectiveness
of our sales, marketing and distribution capabilities and the effectiveness of such capabilities of our collaborative partners,
if any; and |
| |
● |
unfavorable
publicity concerning our products or any similar products. |
Our
product candidates, if successfully developed, will compete with a number of products manufactured and marketed by major pharmaceutical
companies, biotechnology companies and manufacturers of generic drugs. Our products may also compete with new products currently
under development by others. Physicians, patients, third-party payors and the medical community may not accept and utilize any
of our product candidates. If our products do not achieve market acceptance, we will not be able to generate significant revenues
or become profitable.
Because
we expect sales of our current product candidates, if approved, to generate substantially all of our product revenues for the
foreseeable future, the failure of these products to find market acceptance would harm our business and could require us to seek
additional financing.
We
do not have extensive sales and marketing experience and our lack of experience may restrict our success in commercializing some
of our product candidates.
We
do not have extensive experience in marketing or selling pharmaceutical products whether in the U.S. or internationally. To obtain
the expertise necessary to successfully market and sell any of our products, the development of our own commercial infrastructure
and/or collaborative commercial arrangements and partnerships will be required. Our ability to make that investment and also execute
our current operating plan is dependent on numerous factors, including, the performance of third party collaborators with whom
we may contract.
Our
products, if approved, may not be commercially viable due to change in health care practice and third party reimbursement limitations.
Recent
initiatives to reduce the federal deficit and to change health care delivery are increasing cost-containment efforts. We anticipate
that Congress, state legislatures and the private sector will continue to review and assess alternative benefits, controls on
health care spending through limitations on the growth of private health insurance premiums and Medicare and Medicaid spending,
price controls on pharmaceuticals, and other fundamental changes to the health care delivery system. Any changes of this type
could negatively impact the commercial viability of our products, if approved. Our ability to successfully commercialize our product
candidates, if they are approved, will depend in part on the extent to which appropriate reimbursement codes and authorized cost
reimbursement levels of these products and related treatment are obtained from governmental authorities, private health insurers
and other organizations, such as health maintenance organizations. In the absence of national Medicare coverage determination,
local contractors that administer the Medicare program may make their own coverage decisions. Any of our product candidates, if
approved and when commercially available, may not be included within the then current Medicare coverage determination or the coverage
determination of state Medicaid programs, private insurance companies or other health care providers. In addition, third-party
payers are increasingly challenging the necessity and prices charged for medical products, treatments and services.
Our
product candidates may cause serious adverse events or undesirable side effects which may delay or prevent marketing approval,
or, if approval is received, require them to be taken off the market, require them to include safety warnings or otherwise limit
their sales.
Serious
adverse events or undesirable side effects from any of our product candidates could arise either during clinical development or,
if approved, after the approved product has been marketed. The results of future clinical trials may show that our product candidates
cause serious adverse events or undesirable side effects, which could interrupt, delay or halt clinical trials, resulting in delay
of, or failure to obtain, marketing approval from the FDA and other regulatory authorities.
If
any of our product candidates cause serious adverse events or undesirable side effects:
| |
● |
regulatory
authorities may impose a clinical hold which could result in substantial delays and adversely impact our ability to continue
development of the product; |
| |
● |
regulatory
authorities may require the addition of labeling statements, specific warnings, a contraindication or field alerts to physicians
and pharmacies; |
| |
● |
we
may be required to change the way the product is administered, conduct additional clinical trials or change the labeling of
the product; |
| |
● |
we
may be required to implement a risk minimization action plan, which could result in substantial cost increases and have a
negative impact on our ability to commercialize the product; |
| |
● |
we
may be required to limit the patients who can receive the product; |
| |
● |
we
may be subject to limitations on how we promote the product; |
| |
● |
sales
of the product may decrease significantly; |
| |
● |
regulatory
authorities may require us to take our approved product off the market; |
| |
● |
we
may be subject to litigation or product liability claims; and |
| |
● |
our
reputation may suffer. |
Any
of these events could prevent us from achieving or maintaining market acceptance of the affected product or could substantially
increase commercialization costs and expenses, which in turn could delay or prevent us from generating significant revenues from
the sale of our products.
The
technology on which our channel partnering arrangement with Intrexon is based on is early stage technology in the field of Melioidosis.
Our
exclusive channel collaboration arrangement with Intrexon contemplates the use of Intrexon’s modular genetic engineering
platform for the development of active pharmaceutical ingredients and drug products targeting the biodefense countermeasure, melioidosis.
Such technology has a limited history of use in the design and development of human therapeutic product candidates and may therefore
involve unanticipated risks or delays. Although we plan to leverage Intrexon’s technology and scientific expertise to develop
products for the treatment of melioidosis, an infectious disease caused by bacteria found in soil and water, we may not be successful
in developing and commercializing these products for a variety of reasons. The risk factors set forth herein that apply to our
other product candidates, which are in various stages of development, also apply to product candidates that we seek to develop
under our exclusive partnership with Intrexon.
We
will incur additional expenses in connection with our exclusive channel collaboration arrangement with Intrexon.
Pursuant
to our exclusive channel collaboration with Intrexon, we are responsible for future research and development expenses of product
candidates developed under such collaboration. Although it is our intent to pursue government funding to support this development,
we expect the level of our overall research and development expenses going forward will increase. Because our collaboration with
Intrexon is new, we have yet to assume development responsibility and costs associated with such program. In addition, because
development activities are determined pursuant to a joint steering committee comprised of representatives from Intrexon and the
Company, future development costs associated with this program may be difficult to anticipate and exceed our expectations. Our
actual cash requirements may vary materially from our current expectations for a number of other factors that may include, but
are not limited to, unanticipated technical challenges, changes in the focus and direction of our development activities or adjustments
necessitated by changes in the competitive landscape in which we operate. If we are unable to continue to financially support
such collaboration due to lack of sufficient government funding or our own working capital constraints, we may be forced to delay
our activities. If we are unable to obtain funding, we may be forced to seek licensing partners or discontinue development.
Federal
and/or state health care reform initiatives could negatively affect our business.
The
availability of reimbursement by governmental and other third-party payers affects the market for any pharmaceutical product.
These third-party payers continually attempt to contain or reduce the costs of healthcare. There have been a number of legislative
and regulatory proposals to change the healthcare system and further proposals are likely. Medicare's policies may decrease the
market for our products. Significant uncertainty exists with respect to the reimbursement status of newly approved healthcare
products.
In
addition, third-party payers are increasingly challenging the price and cost-effectiveness of medical products and services. Once
approved, we might not be able to sell our products profitably or recoup the value of our investment in product development if
reimbursement is unavailable or limited in scope, particularly for product candidates addressing small patient populations. On
July 15, 2008, the Medicare Improvements for Patients and Providers Act of 2008 became law with a number of Medicare and Medicaid
reforms to establish a bundled Medicare payment rate that includes services and drug/labs that were separately billed at that
time. Bundling initiatives that have been implemented in other healthcare settings have occasionally resulted in lower utilization
of services that had not previously been a part of the bundled payment.
In
addition, in some foreign countries, the proposed pricing for a drug must be approved before it may be lawfully marketed. The
requirements governing drug pricing vary widely from country to country. We expect that there will continue to be a number of
U.S. federal and state proposals to implement governmental pricing controls. While we cannot predict whether such legislative
or regulatory proposals will be adopted, the adoption of such proposals could have a material adverse effect on our business,
financial condition and profitability.
We
may not be able to retain rights licensed to us by third parties to commercialize key products or to develop the third party relationships
we need to develop, manufacture and market our products.
We
currently rely on license agreements from New York University, Yeda Research and Development Company Ltd., the University of Texas
Southwestern Medical Center, the University of British Columbia, Harvard University, the University of Colorado, and George B.
McDonald, MD for the rights to commercialize key product candidates, and we entered into an exclusive channel collaboration agreement
with Intrexon pursuant to which we acquired a license to Intrexon’s advanced human antibody discovery, isolation, and production
technologies. We may not be able to retain the rights granted under these agreements or negotiate additional agreements on reasonable
terms, if at all.
Furthermore,
we currently have very limited product development capabilities and no manufacturing, marketing or sales capabilities. For us
to research, develop and test our product candidates, we need to contract or partner with outside researchers, in most cases with
or through those parties that did the original research and from whom we have licensed the technologies. If products are successfully
developed and approved for commercialization, then we will need to enter into additional collaboration and other agreements with
third parties to manufacture and market our products. We may not be able to induce the third parties to enter into these agreements,
and, even if we are able to do so, the terms of these agreements may not be favorable to us. Our inability to enter into these
agreements could delay or preclude the development, manufacture and/or marketing of some of our product candidates or could significantly
increase the costs of doing so. In the future, we may grant to our development partners rights to license and commercialize pharmaceutical
and related products developed under the agreements with them, and these rights may limit our flexibility in considering alternatives
for the commercialization of these products. Furthermore, third-party manufacturers or suppliers may not be able to meet our needs
with respect to timing, quantity and quality for the products.
Additionally,
if we do not enter into relationships with additional third parties for the marketing of our products, if and when they are approved
and ready for commercialization, we would have to build our own sales force or enter into commercialization agreements with other
companies. Development of an effective sales force in any part of the world would require significant financial resources, time
and expertise. We may not be able to obtain the financing necessary to establish a sales force in a timely or cost effective manner,
if at all, and any sales force we are able to establish may not be capable of generating demand for our product candidates, if
they are approved.
We
may suffer product and other liability claims; we maintain only limited product liability insurance, which may not be sufficient.
The
clinical testing, manufacture and sale of our products involves an inherent risk that human subjects in clinical testing or consumers
of our products may suffer serious bodily injury or death due to side effects, allergic reactions or other unintended negative
reactions to our products. As a result, product and other liability claims may be brought against us. We currently have clinical
trial and product liability insurance with limits of liability of $10 million, which may not be sufficient to cover our potential
liabilities. Because liability insurance is expensive and difficult to obtain, we may not be able to maintain existing insurance
or obtain additional liability insurance on acceptable terms or with adequate coverage against potential liabilities. Furthermore,
if any claims are brought against us, even if we are fully covered by insurance, we may suffer harm such as adverse publicity.
We
may use hazardous chemicals in our business. Potential claims relating to improper handling, storage or disposal of these chemicals
could affect us and be time consuming and costly.
Our
research and development processes and/or those of our third party contractors may involve the controlled use of hazardous materials
and chemicals. These hazardous chemicals are reagents and solvents typically found in a chemistry laboratory. Our operations also
may produce hazardous waste products. Federal, state and local laws and regulations govern the use, manufacture, storage, handling
and disposal of hazardous materials. While we attempt to comply with all environmental laws and regulations, including those relating
to the outsourcing of the disposal of all hazardous chemicals and waste products, we cannot eliminate the risk of contamination
from or discharge of hazardous materials and any resultant injury. In the event of such an accident, we could be held liable for
any resulting damages and any liability could materially adversely affect our business, financial condition and results of operations.
Compliance
with environmental laws and regulations may be expensive. Current or future environmental regulations may impair our research,
development or production efforts. We might have to pay civil damages in the event of an improper or unauthorized release of,
or exposure of individuals to, hazardous materials. We are not insured against these environmental risks.
We
may agree to indemnify our collaborators in some circumstances against damages and other liabilities arising out of development
activities or products produced in connection with these collaborations.
In
addition, the federal, state and local laws and regulations governing the use, manufacture, storage, handling and disposal of
hazardous or radioactive materials and waste products may require us to incur substantial compliance costs that could materially
adversely affect our business, financial condition and results of operations.
We
may not be able to compete with our larger and better financed competitors in the biotechnology industry.
The
biotechnology industry is intensely competitive, subject to rapid change and sensitive to new product introductions or enhancements.
Most of our existing competitors have greater financial resources, larger technical staffs, and larger research budgets than we
have, as well as greater experience in developing products and conducting clinical trials. Our competition is particularly intense
in the gastroenterology and transplant areas and is also intense in the therapeutic area of inflammatory bowel diseases. We face
intense competition in the biodefense area from various public and private companies and universities as well as governmental
agencies, such as the U.S. Army, which may have their own proprietary technologies that may directly compete with our technologies.
In addition, there may be other companies that are currently developing competitive technologies and products or that may in the
future develop technologies and products that are comparable or superior to our technologies and products. We may not be able
to compete with our existing and future competitors, which could lead to the failure of our business.
Additionally,
if a competitor receives FDA approval before we do for a drug that is similar to one of our product candidates, FDA approval for
our product candidate may be precluded or delayed due to periods of non-patent exclusivity and/or the listing with the FDA by
the competitor of patents covering its newly-approved drug product. Periods of non-patent exclusivity for new versions of existing
drugs such as our current product candidates can extend up to three and one-half years. See “Business—The Drug Approval
Process.”
These
competitive factors could require us to conduct substantial new research and development activities to establish new product targets,
which would be costly and time consuming. These activities would adversely affect our ability to commercialize products and achieve
revenue and profits.
Competition
and technological change may make our product candidates and technologies less attractive or obsolete.
We
compete with established pharmaceutical and biotechnology companies that are pursuing other forms of treatment for the same indications
we are pursuing and that have greater financial and other resources. Other companies may succeed in developing products earlier
than us, obtaining FDA approval for products more rapidly, or developing products that are more effective than our product candidates.
Research and development by others may render our technology or product candidates obsolete or noncompetitive, or result in treatments
or cures superior to any therapy we develop. We face competition from companies that internally develop competing technology or
acquire competing technology from universities and other research institutions. As these companies develop their technologies,
they may develop competitive positions that may prevent, make futile, or limit our product commercialization efforts, which would
result in a decrease in the revenue we would be able to derive from the sale of any products.
There
can be no assurance that any of our product candidates will be accepted by the marketplace as readily as these or other competing
treatments. Furthermore, if our competitors’ products are approved before ours, it could be more difficult for us to obtain
approval from the FDA. Even if our products are successfully developed and approved for use by all governing regulatory bodies,
there can be no assurance that physicians and patients will accept our product(s) as a treatment of choice.
Furthermore,
the pharmaceutical research industry is diverse, complex, and rapidly changing. By its nature, the business risks associated therewith
are numerous and significant. The effects of competition, intellectual property disputes, market acceptance, and FDA regulations
preclude us from forecasting revenues or income with certainty or even confidence.
Our
business could be harmed if we fail to retain our current personnel or if they are unable to effectively run our business.
We
currently have 17 employees and we depend upon these employees (in particular Dr. Christopher Schaber, our President and Chief
Executive Officer) to manage the day-to-day activities of our business. Because we have such limited personnel, the loss of any
of them or our inability to attract and retain other qualified employees in a timely manner would likely have a negative impact
on our operations. We may be unable to effectively manage and operate our business, and our business may suffer, if we lose the
services of our employees.
Instability
and volatility in the financial markets could have a negative impact on our business, financial condition, results of operations,
and cash flows.
During
recent months, there has been substantial volatility in financial markets due at least in part to the uncertainty with regard
to the global economic environment and the partial government shutdown due to delays in increasing the U.S. debt limit in October
2013. In addition, there has been substantial uncertainty in the capital markets and access to additional financing is uncertain.
Moreover, customer spending habits may be adversely affected by current and future economic conditions. These conditions could
have an adverse effect on our industry and business, including our financial condition, results of operations, and cash flows.
To
the extent that we do not generate sufficient cash from operations, we may need to issue stock or incur indebtedness to finance
our plans for growth. Recent turmoil in the credit markets and the potential impact on the liquidity of major financial institutions
may have an adverse effect on our ability to fund our business strategy through borrowings, under either existing or newly created
instruments in the public or private markets on terms we believe to be reasonable, if at all.
Risks
Related to our Intellectual Property
We
may be unable to commercialize our products if we are unable to protect our proprietary rights, and we may be liable for significant
costs and damages if we face a claim of intellectual property infringement by a third party.
Our
near and long term prospects depend in part on our ability to obtain and maintain patents, protect trade secrets and operate without
infringing upon the proprietary rights of others. In the absence of patent and trade secret protection, competitors may adversely
affect our business by independently developing and marketing substantially equivalent or superior products and technology, possibly
at lower prices. We could also incur substantial costs in litigation and suffer diversion of attention of technical and management
personnel if we are required to defend ourselves in intellectual property infringement suits brought by third parties, with or
without merit, or if we are required to initiate litigation against others to protect or assert our intellectual property rights.
Moreover, any such litigation may not be resolved in our favor.
Although
we and our licensors have filed various patent applications covering the uses of our product candidates, patents may not be issued
from the patent applications already filed or from applications that we might file in the future. Moreover, the patent position
of companies in the pharmaceutical industry generally involves complex legal and factual questions, and recently has been the
subject of much litigation. Any patents we own or license, now or in the future, may be challenged, invalidated or circumvented.
To date, no consistent policy has been developed in the U.S. Patent and Trademark Office (the “PTO”) regarding the
breadth of claims allowed in biotechnology patents.
In
addition, because patent applications in the U.S. are maintained in secrecy until patent applications publish or patents issue,
and because publication of discoveries in the scientific or patent literature often lags behind actual discoveries, we cannot
be certain that we and our licensors are the first creators of inventions covered by any licensed patent applications or patents
or that we or they are the first to file. The PTO may commence interference proceedings involving patents or patent applications,
in which the question of first inventorship is contested. Accordingly, the patents owned or licensed to us may not be valid or
may not afford us protection against competitors with similar technology, and the patent applications licensed to us may not result
in the issuance of patents.
It
is also possible that our owned and licensed technologies may infringe on patents or other rights owned by others, and licenses
to which may not be available to us. We may be unable to obtain a license under such patent on terms favorable to us, if at all.
We may have to alter our products or processes, pay licensing fees or cease activities altogether because of patent rights of
third parties.
In
addition to the products for which we have patents or have filed patent applications, we rely upon unpatented proprietary technology
and may not be able to meaningfully protect our rights with regard to that unpatented proprietary technology. Furthermore, to
the extent that consultants, key employees or other third parties apply technological information developed by them or by others
to any of our proposed projects, disputes may arise as to the proprietary rights to this information, which may not be resolved
in our favor.
We
may be involved in lawsuits to protect or enforce our patents, which could be expensive and time consuming.
The
pharmaceutical industry has been characterized by extensive litigation regarding patents and other intellectual property rights,
and companies have employed intellectual property litigation to gain a competitive advantage. We may become subject to infringement
claims or litigation arising out of patents and pending applications of our competitors, or additional interference proceedings
declared by the PTO to determine the priority of inventions. The defense and prosecution of intellectual property suits, PTO proceedings,
and related legal and administrative proceedings are costly and time-consuming to pursue, and their outcome is uncertain. Litigation
may be necessary to enforce our issued patents, to protect our trade secrets and know-how, or to determine the enforceability,
scope, and validity of the proprietary rights of others. An adverse determination in litigation or interference proceedings to
which we may become a party could subject us to significant liabilities, require us to obtain licenses from third parties, or
restrict or prevent us from selling our products in certain markets. Although patent and intellectual property disputes might
be settled through licensing or similar arrangements, the costs associated with such arrangements may be substantial and could
include our paying large fixed payments and ongoing royalties. Furthermore, the necessary licenses may not be available on satisfactory
terms or at all.
Competitors
may infringe our patents, and we may file infringement claims to counter infringement or unauthorized use. This can be expensive,
particularly for a company of our size, and time-consuming. In addition, in an infringement proceeding, a court may decide that
a patent of ours is not valid or is unenforceable, or may refuse to stop the other party from using the technology at issue on
the grounds that our patents do not cover its technology. An adverse determination of any litigation or defense proceedings could
put one or more of our patents at risk of being invalidated or interpreted narrowly.
Also,
a third party may assert that our patents are invalid and/or unenforceable. There are no unresolved communications, allegations,
complaints or threats of litigation related to the possibility that our patents are invalid or unenforceable. Any litigation or
claims against us, whether or not merited, may result in substantial costs, place a significant strain on our financial resources,
divert the attention of management and harm our reputation. An adverse decision in litigation could result in inadequate protection
for our product candidates and/or reduce the value of any license agreements we have with third parties.
Interference
proceedings brought before the PTO may be necessary to determine priority of invention with respect to our patents or patent applications.
During an interference proceeding, it may be determined that we do not have priority of invention for one or more aspects in our
patents or patent applications and could result in the invalidation in part or whole of a patent or could put a patent application
at risk of not issuing. Even if successful, an interference proceeding may result in substantial costs and distraction to our
management.
Furthermore,
because of the substantial amount of discovery required in connection with intellectual property litigation or interference proceedings,
there is a risk that some of our confidential information could be compromised by disclosure. In addition, there could be public
announcements of the results of hearings, motions or other interim proceedings or developments. If investors perceive these results
to be negative, the price of our common stock could be adversely affected.
If
we infringe the rights of third parties we could be prevented from selling products, forced to pay damages, and defend against
litigation.
If
our products, methods, processes and other technologies infringe the proprietary rights of other parties, we could incur substantial
costs and we may have to: obtain licenses, which may not be available on commercially reasonable terms, if at all; abandon an
infringing product candidate; redesign our products or processes to avoid infringement; stop using the subject matter claimed
in the patents held by others; pay damages; and/or defend litigation or administrative proceedings which may be costly whether
we win or lose, and which could result in a substantial diversion of our financial and management resources.
Risks
Related to our Common Stock
Our
common stock price is highly volatile.
The
market price of our common stock, like that of many other research and development public pharmaceutical and biotechnology companies,
has been highly volatile and may continue to be so in the future due to a wide variety of factors, including:
| |
● |
announcements
by us or others of results of pre-clinical testing and clinical trials; |
| |
● |
announcements
of technological innovations, more important bio-threats or new commercial therapeutic products by us, our collaborative partners
or our present or potential competitors; |
| |
● |
our
quarterly operating results and performance; |
| |
● |
developments
or disputes concerning patents or other proprietary rights; |
| |
● |
litigation
and government proceedings; |
| |
● |
changes
in government regulations; |
| |
● |
our
available working capital; |
| |
● |
economic
and other external factors; and |
| |
● |
general
market conditions. |
Since
January 1, 2014, the closing stock price of our common stock has fluctuated between a high of $2.50 per share to a low of $0.95
per share. On July 28, 2015, the last quoted sale price of our common stock as reported on the OTCQB was $1.72 per share. The
fluctuation in the price of our common stock has sometimes been unrelated or disproportionate to our operating performance. In
addition, potential dilutive effects of future sales of shares of common stock by the Company, as well as potential sale of common
stock by the holders of warrants and options, could have an adverse effect on the market price of our shares.
Our
common stock trades on the Over-the-Counter Bulletin Board.
Our
common stock trades on the OTCQB securities market under the symbol “SNGX.” The OTCQB is a decentralized market regulated
by the Financial Industry Regulatory Authority in which securities are traded via an electronic quotation system that serves more
than 3,000 companies, but provides significantly less liquidity than national market systems such as the NYSE MKT. On the OTCQB,
securities are traded by a network of brokers or dealers who carry inventories of securities to facilitate the buy and sell orders
of investors, rather than providing the order matchmaking service seen in specialist exchanges. OTCQB securities include national,
regional, and foreign equity issues. Companies traded on the OTCQB must be current in their reports filed with the SEC and other
regulatory authorities.
Since
our common stock is not listed on a national exchange or market, the trading market for our common stock may become illiquid.
Our common stock is subject to the penny stock rules of the SEC, which generally are applicable to equity securities with a price
of less than $5.00 per share, other than securities registered on certain national securities exchanges provided that current
price and volume information with respect to transactions in such securities is provided by the exchange or system. The penny
stock rules require a broker-dealer, before a transaction in a penny stock not otherwise exempt from the rules, to deliver a standardized
risk disclosure document prepared by the SEC that provides information about penny stocks and the nature and level of risks in
the penny stock market. The broker-dealer also must provide the customer with bid and ask quotations for the penny stock, the
compensation of the broker-dealer and its salesperson in the transaction and monthly account statements showing the market value
of each penny stock held in the customer’s account. In addition, the penny stock rules require that, before a transaction
in a penny stock that is not otherwise exempt from such rules, the broker-dealer must make a special written determination that
the penny stock is a suitable investment for the purchaser and receive the purchaser’s written agreement to the transaction.
As a result of these requirements, our common stock could be priced at a lower price and our stockholders could find it more difficult
to sell their shares.
Shareholders
may suffer substantial dilution related to issued stock warrants and options.
As
of March 31, 2015, we had a number of agreements or obligations that may result in dilution to investors. These include:
| |
● |
warrants
to purchase a total of approximately 6,085,714 shares of our common stock at a current weighted average exercise price of
approximately $1.25; and |
| |
● |
options
to purchase approximately 2,272,022 shares of our common stock at a current weighted average exercise price of approximately
$2.34. |
We
also have an incentive compensation plan for our management, employees and consultants. We have granted, and expect to grant in
the future, options to purchase shares of our common stock to our directors, employees and consultants. To the extent that warrants
or options are exercised, our stockholders will experience dilution and our stock price may decrease.
Additionally,
the sale, or even the possibility of the sale, of the shares of common stock underlying these warrants and options could have
an adverse effect on the market price for our securities or on our ability to obtain future financing.
Anti-takeover
provisions in our stockholder rights plan and under Delaware law could make a third party acquisition of the Company difficult.
Our
stockholder rights plan contains provisions that could make it more difficult for a third party to acquire us, even if doing so
might be deemed beneficial by our stockholders. These provisions could limit the price that investors might be willing to pay
in the future for shares of our common stock. We are also subject to certain provisions of Delaware law that could delay, deter
or prevent a change in control of the Company. The rights issued pursuant to our stockholder rights plan will become exercisable
the tenth day after a person or group announces acquisition of 15% or more of our common stock or commences, or announces an intention
to make, a tender or exchange offer the consummation of which would result in ownership by the person or group of 15% or more
of our common stock. If the rights become exercisable, the holders of the rights (other than the person acquiring 15% or more
of our common stock) will be entitled to acquire, in exchange for the rights’ exercise price, shares of our common stock
or shares of any company in which we are merged, with a value equal to twice the rights’ exercise price.
Our
shares of common stock are thinly traded, so stockholders may be unable to sell at or near ask prices or at all if they need to
sell shares to raise money or otherwise desire to liquidate their shares.
Our
common stock has from time to time been “thinly-traded,” meaning that the number of persons interested in purchasing
our common stock at or near ask prices at any given time may be relatively small or non-existent. This situation is attributable
to a number of factors, including the fact that we are a small company that is relatively unknown to stock analysts, stock brokers,
institutional investors and others in the investment community that generate or influence sales volume, and that even if we came
to the attention of such persons, they tend to be risk-averse and would be reluctant to follow an unproven company such as ours
or purchase or recommend the purchase of our shares until such time as we become more seasoned and viable. As a consequence, there
may be periods of several days or more when trading activity in our shares is minimal or non-existent, as compared to a seasoned
issuer which has a large and steady volume of trading activity that will generally support continuous sales without an adverse
effect on share price. We cannot give stockholders any assurance that a broader or more active public trading market for our common
shares will develop or be sustained, or that current trading levels will be sustained.
Since
our common stock is not listed on a national securities exchange, U.S. holders of warrants may not be able to exercise their warrants
without compliance with applicable state securities laws and the value of your warrants may be significantly reduced.
Since
our securities are not listed for trading on a national exchange, the exercise of the warrants by U.S. holders may not be exempt
from state securities laws. As a result, depending on the state of residence of a holder of the warrants, a U.S. holder may not
be able to exercise its warrants unless we comply with any state securities law requirements necessary to permit such exercise
or an exemption applies. Although we plan to use our reasonable efforts to assure that U.S. holders will be able to exercise their
warrants under applicable state securities laws if no exemption exists, there is no assurance that we will be able to do so. As
a result, your ability to exercise your warrants may be limited. The value of the warrants may be significantly reduced if U.S.
holders are not able to exercise their warrants under applicable state securities laws.
Our
common stock is deemed to be a “penny stock,” which may make it more difficult for investors to sell their shares
due to suitability requirements.
Our
common stock is subject to Rule 15g-1 through 15g-9 under the Securities Exchange Act of 1934, as amended, which imposes certain
sales practice requirements on broker-dealers which sell our common stock to persons other than established customers and “accredited
investors” (generally, individuals with a net worth in excess of $1,000,000 or annual incomes exceeding $200,000 (or $300,000
together with their spouses)). For transactions covered by this rule, a broker-dealer must make a special suitability determination
for the purchaser and have received the purchaser’s written consent to the transaction prior to the sale. This rule adversely
affects the ability of broker-dealers to sell our common stock and the ability of our stockholders to sell their shares of common
stock.
Additionally,
our common stock is subject to the SEC regulations for “penny stock.” Penny stock includes any equity security that
is not listed on a national exchange and has a market price of less than $5.00 per share, subject to certain exceptions. The regulations
require that prior to any non-exempt buy/sell transaction in a penny stock, a disclosure schedule set forth by the SEC relating
to the penny stock market must be delivered to the purchaser of such penny stock. This disclosure must include the amount of commissions
payable to both the broker-dealer and the registered representative and current price quotations for the common stock. The regulations
also require that monthly statements be sent to holders of penny stock that disclose recent price information for the penny stock
and information of the limited market for penny stocks. These requirements adversely affect the market liquidity of our common
stock.
We
do not currently intend to pay dividends on our common stock in the foreseeable future, and consequently, our stockholders’
ability to achieve a return on their investment will depend on appreciation in the price of our common stock.
We
have never declared or paid cash dividends on our common stock and do not anticipate paying any cash dividends to holders of our
common stock in the foreseeable future. Consequently, our stockholders must rely on sales of their common stock after price appreciation,
which may never occur, as the only way to realize any future gains on their investments. There is no guarantee that shares of
our common stock will appreciate in value or even maintain the price at which our stockholders have purchased their shares.
Upon
dissolution of the Company, our stockholders may not recoup all or any portion of their investment.
In
the event of a liquidation, dissolution or winding-up of the Company, whether voluntary or involuntary, the proceeds and/or assets
of the Company remaining after giving effect to such transaction, and the payment of all of our debts and liabilities will be
distributed to the holders of common stock on a pro rata basis. There can be no assurance that we will have available assets to
pay to the holders of common stock, or any amounts, upon such a liquidation, dissolution or winding-up of the Company. In this
event, our stockholders could lose some or all of their investment.
The
sale or issuance of our common stock to Lincoln Park may cause dilution, and the sale of the shares of common stock acquired by
Lincoln Park, or the perception that such sales may occur, could cause the price of our common stock to fall.
On
November 18, 2013, we entered into a purchase agreement with Lincoln Park, pursuant to which Lincoln Park has committed to purchase
up to $10.6 million of our common stock. Concurrently with the execution of the purchase agreement, we issued 97,656 shares of
our common stock to Lincoln Park as a partial fee for its commitment to purchase shares of our common stock under the purchase
agreement and 285,714 shares of common stock for an aggregate price of $600,000. From November 18, 2013 through July 28, 2015,
we sold 825,000 additional shares to Lincoln Park and issued 19,354 additional shares to Lincoln Park as additional commitment
shares under the purchase agreement and received proceeds of approximately $1.6 million. The shares that may be sold pursuant
to the purchase agreement in the future may be sold by us to Lincoln Park at our discretion from time to time over the remaining
term of approximately 16 months from the date of this prospectus. The purchase price for the shares that we may sell to Lincoln
Park under the purchase agreement will fluctuate based on the price of our common stock. Depending on market liquidity at the
time, sales of such shares may cause the trading price of our common stock to fall.
We
generally have the right to control the timing and amount of any sales of our shares to Lincoln Park, except that, pursuant to
the terms of our agreements with Lincoln Park, we would be unable to sell shares to Lincoln Park if and when the closing sale
price of our common stock is below $1.00 per share, subject to adjustment as set forth in the purchase agreement. Additional sales
of our common stock, if any, to Lincoln Park will depend upon market conditions and other factors to be determined by us. Lincoln
Park may ultimately purchase all, some or none of the additional shares of our common stock that may be sold pursuant to the purchase
agreement and, after it has acquired shares, Lincoln Park may sell all, some or none of those shares. Therefore, sales to Lincoln
Park by us could result in substantial dilution to the interests of other holders of our common stock. Additionally, the sale
of a substantial number of shares of our common stock to Lincoln Park, or the anticipation of such sales, could make it more difficult
for us to sell equity or equity-related securities in the future at a time and at a price that we might otherwise wish to effect
sales.
The
sale of our common stock to the Equity Purchasers may cause dilution, and the sale of the shares of common stock acquired by the
Equity Purchasers, or the perception that such sales may occur, could cause the price of our common stock to fall.
On
July 29, 2015, we entered into the Purchase Agreements with the Equity Purchasers. Pursuant the Purchase Agreements, the Equity
Purchasers have committed to purchase up to an aggregate of $10 million of our common stock. The shares that may be sold pursuant
to the Purchase Agreements in the future may be sold by us to the Equity Purchasers at our discretion from time to time, commencing
after the SEC has declared effective the registration statement that includes this prospectus until December 31, 2016. The per
share purchase price for the shares that we may sell to the Equity Purchasers under the Purchase Agreements will fluctuate based
on the price of our common stock, and will be equal to 80% of the lowest daily volume weighted average price of the common stock
for the five consecutive trading days immediately following our request for the Equity Purchasers to purchase the shares. Depending
on market liquidity at the time, sales of such shares may cause the trading price of our common stock to fall.
We
generally have the right to control the timing and amount of any sales of our shares to the Equity Purchasers, except that, pursuant
to the terms of the Purchase Agreements, we would be unable to sell shares to the Equity Purchasers if such purchase would result
in an Equity Purchaser’s respective beneficial ownership equaling more than 9.99% of the outstanding common stock. The Equity
Purchasers may ultimately purchase all, some or none of the shares of our common stock that may be sold pursuant to the Purchase
Agreements and, after they have acquired shares, the Equity Purchasers may sell all, some or none of those shares. Therefore,
sales to the Equity Purchasers by us could result in substantial dilution to the interests of other holders of our common stock.
Additionally, the sale of a substantial number of shares of our common stock to the Equity Purchasers, or the anticipation of
such sales, could make it more difficult for us to sell equity or equity-related securities in the future at a time and at a price
that we might otherwise wish to effect sales.
The
Equity Purchasers will pay less than the then-prevailing market price for our common stock.
The
common stock to be issued to the Equity Purchasers pursuant to the Purchase Agreements will be purchased at an twenty percent
(20%) discount to the lowest daily volume weighted average price of the common stock for the five consecutive trading days immediately
following our request for the Equity Purchasers to purchase the shares. The Equity Purchasers have a financial incentive to sell
our common stock immediately upon receiving the shares to realize the profit equal to the difference between the discounted price
and the market price. If the Equity Purchasers sell the shares, the price of our common stock could decrease. If our stock price
decreases, the Equity Purchasers may have a further incentive to sell the shares of our common stock that they hold. These sales
may have a further impact on our stock price.
The
issuance of our common stock pursuant to the terms of the asset purchase agreement with Hy Biopharma Inc. may cause
dilution and the issuance of such shares of common stock, or the perception that such issuances may occur, could cause the price
of our common stock to fall.
On
April 1, 2014, we entered into an option agreement pursuant to which Hy Biopharma Inc. (“Hy Biopharma”) granted us
an option to purchase certain assets, properties and rights (the “Hypericin Assets”) related to the development of
Hy Biopharma’s synthetic hypericin product candidate for the treatment of CTCL, which we refer to as SGX301, from Hy Biopharma.
In exchange for the option, we paid $50,000 in cash and issued 43,067 shares of common stock in the aggregate to Hy Biopharma
and its assignees. We subsequently exercised the option, and on September 3, 2014, we entered into an asset purchase agreement
with Hy Biopharma, pursuant to which we purchased the Hypericin Assets. Pursuant to the purchase agreement, we paid $250,000 in
cash and issued 1,849,113 shares of common stock in the aggregate to Hy Biopharma and its assignees, and may issue up to an aggregate
of $10 million worth of our common stock (subject to a cap equal to 19.99% of our issued and outstanding common stock) in the
aggregate upon attainment of specified milestones. Also on September 3, 2014, we entered into the Registration Rights Agreement
with Hy Biopharma, pursuant to which we have filed a registration statement with the SEC.
The
number of shares that we may issue under the purchase agreement will fluctuate based on the market price of our common stock.
Depending on market liquidity at the time, the issuance of such shares may cause the trading price of our common stock to fall.
We
may ultimately issue all, some or none of the additional shares of our common stock that may be issued pursuant to the purchase
agreement. We are required to register any shares issued pursuant to the purchase agreement for resale under the Securities Act.
After any such shares are registered, the holders will be able to sell all, some or none of those shares. Therefore, issuances
by us under the purchase agreement could result in substantial dilution to the interests of other holders of our common stock.
Additionally, the issuance of a substantial number of shares of our common stock pursuant to the purchase agreement, or the anticipation
of such issuances, could make it more difficult for us to sell equity or equity-related securities in the future at a time and
at a price that we might otherwise wish to effect sales.
CAUTIONARY
NOTE REGARDING FORWARD-LOOKING STATEMENTS
The
information contained in this prospectus includes forward-looking statements. These forward-looking statements are often identified
by words such as “may,” “expect,” “intend,” “anticipate,” “believe,”
“estimate,” “continue,” “plan,” “potential” and similar expressions. These statements
involve estimates, assumptions and uncertainties that could cause actual results to differ materially from those expressed for
the reasons described in this prospectus. You should not place undue reliance on these forward-looking statements.
You
should be aware that our actual results could differ materially from those contained in the forward-looking statements due to
a number of factors, including:
| |
● |
our
dependence on the expertise, effort, priorities and contractual obligations of third parties in the clinical trials, manufacturing,
marketing, sales and distribution of our products; |
| |
● |
the
domestic and international regulatory process and related laws, rules and regulations governing our technologies and our proposed
products, including: (i) the timing, status and results of our or our commercial partners’ filings with the U.S. Food
and Drug Administration and its foreign equivalents, (ii) the timing, status and results of non-clinical work and clinical
studies, including regulatory review thereof and (iii) the heavily regulated industry in which we operate our business generally; |
| |
● |
uncertainty
as to whether our product candidates will be safe and effective to support regulatory approvals; |
| |
● |
significant
uncertainty inherent in developing vaccines against bioterror threats, and manufacturing and conducting preclinical and clinical
trials of vaccines; |
| |
● |
our
ability to obtain future financing or funds when needed, either through the raising of capital, the incurrence of convertible
or other indebtedness or through strategic financing or commercialization partnerships; |
| |
● |
that
product development and commercialization efforts will be reduced or discontinued due to difficulties or delays in clinical
trials or a lack of progress or positive results from research and development efforts; |
| |
● |
our
ability to obtain further grants and awards from the U.S. Government and other countries, and maintenance of our existing
grants; |
| |
● |
our
ability to enter into any biodefense procurement contracts with the U.S. Government or other countries; |
| |
● |
our
ability to patent, register and protect our technology from challenge and our products from competition; |
| |
● |
maintenance
or expansion of our license agreements with our current licensors; |
| |
● |
the
protection and control afforded by our patents or other intellectual property, and any interest in patents or other intellectual
property that we license, or our or our partners’ ability to enforce our rights under such owned or licensed patents
or other intellectual property; |
| |
● |
changes
in healthcare regulation; |
| |
● |
changes
in the needs of biodefense procurement agencies; |
| |
● |
maintenance
and progression of our business strategy; |
| |
● |
the
possibility that our products under development may not gain market acceptance; |
| |
● |
our
expectations about the potential market sizes and market participation potential for our product candidates may not be realized; |
| |
● |
our
expected revenues (including sales, milestone payments and royalty revenues) from our product candidates and any related commercial
agreements of ours may not be realized; |
| |
● |
the
ability of our manufacturing partners to supply us or our commercial partners with clinical or commercial supplies of our
products in a safe, timely and regulatory compliant manner and the ability of such partners to address any regulatory issues
that have arisen or may in the future arise; and |
| |
● |
competition
existing today or that may arise in the future, including the possibility that others may develop technologies or products
superior to our products. |
You
should also consider carefully the statements under "Risk Factors" and other sections of this prospectus, which address
additional factors that could cause our actual results to differ from those set forth in the forward-looking statements and could
materially and adversely affect our business, operating results and financial condition. All subsequent written and oral forward-looking
statements attributable to us or persons acting on our behalf are expressly qualified in their entirety by the applicable cautionary
statements.
The
forward-looking statements speak only as of the date on which they are made, and, except to the extent required by federal securities
laws, we undertake no obligation to update any forward-looking statement to reflect events or circumstances after the date on
which the statement is made or to reflect the occurrence of unanticipated events. In addition, we cannot assess the impact of
each factor on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially
from those contained in any forward-looking statements.
Industry
Data and Market Information
This
prospectus contains estimates, projections and other statistical data made by independent parties and by us relating to market
size and growth, the potential value of government procurement contracts, the incidence of certain medical conditions and other
industry data. These data, to the extent they contain estimates or projections, involve a number of subjective assumptions and
limitations, and you are cautioned not to give undue weight to such estimates or projections. Industry publications and other
reports we have obtained from independent parties generally state that the data contained in these publications or other reports
have been obtained in good faith or from sources considered to be reliable, but they do not guarantee the accuracy or completeness
of such data. While we believe that the data from these industry publications and other reports are generally reliable, we have
not independently verified the accuracy or completeness of such data. These and other factors could cause results to differ materially
from those expressed in these publications and reports.
We
have provided estimates of the potential worldwide market or value of potential government procurement contracts for certain of
our product candidates. These estimates are based on a number of factors, including our expectation as to the number of patients
with a certain medical condition that would potentially benefit from a particular product candidate, the current costs of treating
patients with the targeted medical condition, our expectation that we will be able to demonstrate to the FDA’s satisfaction
in our clinical trials that the product candidate is safe and effective, our belief that our product candidate would, if approved,
have an assumed treatment cost per patient, historic values of government procurement contracts for vaccines, and our expectation
of the dosage of the product candidate. While we have determined these estimates based on assumptions that we believe are reasonable,
there are a number of factors that could cause our expectations to change or not be realized. Among these factors are the following:
there is no assurance that the product candidate will prove to be safe and effective or will ultimately be approved for sale by
the FDA; any FDA approval of the product candidate may contain restrictions on its use or require warning labels; third party
payors may not be willing to provide reimbursement for product candidate at the assumed price per patient; the government may
not be willing to procure our vaccine candidates in amounts or at costs similar to its historic procurement activities; the dosage
that ultimately may be approved may be different from the assumed dosage; and doctors may not adopt the product candidate for
use as quickly or as broadly as we have assumed. It is possible that the ultimate market for a product candidate or value of procurement
contracts will differ significantly from our expectations due to these or other factors. As a result of these and other factors,
investors should not place undue reliance on such estimates. See “Risk Factors:”
USE
OF PROCEEDS
This
prospectus relates to shares of our common stock that may be offered and sold from time to time by the selling stockholders. We
will not receive any proceeds upon the sale of shares by the selling stockholders in this offering. However, we may receive gross
proceeds of up to $10 million under the Purchase Agreements with the Equity Purchasers, assuming that we sell the full amount
of our common stock that we have the right, but not the obligation, to sell to the Equity Purchasers under those agreements. See
“Plan of Distribution” elsewhere in this prospectus for more information.
We
currently expect to use the net proceeds from the sale of shares to the Equity Purchasers under the Purchase Agreements to further
develop our late-stage product candidates and for other general corporate purposes. We will have broad discretion in determining
how we will allocate the proceeds from any sales to the Equity Purchasers.
Even
if we sell $10 million worth of shares of our common stock to the Equity Purchasers pursuant to the Purchase Agreements, we will
need to obtain additional financing in the future in order to fully fund all of our planned research and development activities.
We may seek additional capital in the private and/or public equity markets, pursue government contracts and
grants as well as business development activities to continue our operations, respond to competitive pressures, develop new
products and services, and to support new strategic partnerships. We are evaluating additional equity financing opportunities
on an ongoing basis and may execute them when appropriate. However, there can be no assurances that we can consummate such a transaction,
or consummate a transaction at favorable pricing.
DIVIDEND
POLICY
We
have never declared nor paid any cash dividends, and currently intend to retain all our cash and any earnings for use in our business
and, therefore, do not anticipate paying any cash dividends in the foreseeable future. Any future determination to pay cash dividends
will be at the discretion of the Board of Directors and will be dependent upon our consolidated financial condition, results of
operations, capital requirements and such other factors as the Board of Directors deems relevant.
MARKET
FOR COMMON EQUITY AND RELATED STOCKHOLDER MATTERS
Market
Information
Our
common stock is quoted on the OTCQB under the symbol “SNGX.” The following table sets forth for the periods indicated,
the high and low sales prices per share of our common stock as reported by the OTCQB.
| | |
Price Range | |
| Period | |
High | | |
Low | |
| Year Ended December 31, 2013: | |
| | |
| |
| First Quarter | |
$ | 2.13 | | |
$ | 0.55 | |
| Second Quarter | |
$ | 2.05 | | |
$ | 0.86 | |
| Third Quarter | |
$ | 2.48 | | |
$ | 0.98 | |
| Fourth Quarter | |
$ | 2.36 | | |
$ | 1.65 | |
| Year Ended December 31, 2014: | |
| | | |
| | |
| First Quarter | |
$ | 2.50 | | |
$ | 1.75 | |
| Second Quarter | |
$ | 2.29 | | |
$ | 1.65 | |
| Third Quarter | |
$ | 2.25 | | |
$ | 1.67 | |
| Fourth Quarter | |
$ | 2.09 | | |
$ | 0.91 | |
| Year Ending December 31, 2015: | |
| | | |
| | |
| First Quarter | |
$ | 2.30 | | |
$ | 0.98 | |
| Second Quarter | |
$ | 2.95 | | |
$ | 1.36 | |
| Third Quarter (through July 28, 2015) | |
$ | 2.48 | | |
$ | 1.66 | |
On
July 28, 2015, the last reported price of our common stock quoted on the OTCQB was $1.72 per share. The OTCQB prices set forth
above represent inter-dealer quotations, without adjustment for retail mark-up, mark-down or commission, and may not represent
the prices of actual transactions.
Transfer
Agent
Shares
of our common stock are issued in registered form. American Stock Transfer & Trust Company, LLC, 6201 15th Avenue,
Brooklyn, NY 11219 (Telephone: (718) 921-8200; Facsimile: (718) 765-8719) is the registrar and transfer agent for shares of our
common stock.
Holders
of Common Stock
As
of July 28, 2015, there were 545 holders of record of our common stock. As of such date, 26,381,976 shares of our common stock
were issued and outstanding.
Equity
Compensation Plan Information
In
December 2005, our Board of Directors approved the 2005 Equity Incentive Plan, which was approved by stockholders on December
29, 2005. In September 2007, our stockholders approved an amendment to the 2005 Equity Incentive Plan to increase the maximum
number of shares of our common stock available for issuance under the plan by 500,000 shares, bringing the total shares reserved
for issuance under the plan to 1,000,000 shares. In September 2010, our stockholders approved a second amendment to the 2005 Equity
Incentive Plan to increase the maximum number of shares of our common stock available for issuance under the plan by 750,000 shares,
bringing the total shares reserved for issuance under the plan to 1,750,000 shares. In September 2013, our stockholders approved
a third amendment to the 2005 Equity Incentive Plan to increase the maximum number of shares of our common stock available for
issuance under the plan by 1,250,000 shares, bringing the total shares reserved for issuance under the plan to 3,000,000 shares.
The following table provides information, as of December 31, 2014 with respect to options outstanding under our 1995 Amended and
Restated Omnibus Incentive Plan and our 2005 Equity Incentive Plan.
| Plan Category | |
Number of Securities to be Issued upon Exercise of Outstanding Options, Warrants and Rights | | |
Weighted-Average Exercise Price of Outstanding Options, Warrants and Rights | | |
Number of Securities Remaining Available for Future Issuance Under Equity Compensation Plans (excluding securities reflected in the first column) | |
| Equity compensation plans approved by security holders 1 | |
| 2,488,279 | | |
$ | 2.40 | | |
| 184,045 | |
| Equity compensation plans not approved by security holders | |
| - | | |
| - | | |
| - | |
| Total | |
| 2,488,279 | | |
$ | 2.40 | | |
| 184,045 | |
| 1 |
Includes
our 1995 Amended and Restated Omnibus Incentive Plan and our 2005 Equity Incentive Plan. Our 1995 Plan expired in 2005 and
thus no securities remain available for future issuance under that plan. |
In
April 2015, our Board of Directors approved the 2015 Equity Incentive Plan, which was approved by stockholders on June 18, 2015.
A maximum of 3,000,000 shares of our common stock are available for issuance under the 2015 Equity Incentive Plan. As of July
28, 2015, no grants have been made under the 2015 Equity Incentive Plan.
DILUTION
Investors
who purchase our common stock will be diluted to the extent of the difference between the public offering price per share of our
common stock and the pro forma as adjusted net tangible book value per share of our common stock immediately after this offering.
Net tangible book value per share is determined by dividing our total tangible assets less total liabilities by the number of
outstanding shares of our common stock. As of March 31, 2015, we had a negative net tangible book value of $(2,181,751), or approximately
$(0.09) per share of common stock.
Dilution
in net tangible book value per share represents the difference between the assumed offering price per share of common
stock of $1.72 (the closing price of our common stock on July 28, 2015) and the pro forma as adjusted net tangible book value
per share of common stock immediately after the sale of $10 million to the Equity Purchasers under the Purchase Agreements. Therefore,
after giving effect to our assumed receipt of $10 million in estimated net proceeds from the issuance of 7,267,441 shares of
common stock under the Purchase Agreements (which is the maximum dollar amount that we may receive under the Purchase
Agreements) and registered in this offering (assuming a purchase price of $1.376 per share, 80% of the closing price of the
common stock on July 28, 2015, and assuming such sale was made on March 31, 2015, and after deducting estimated offering
commissions and expenses payable by us), our pro forma as adjusted net tangible book value as of March 31, 2015 would have
been approximately $7,359,749, or $0.23 per share. This would represent an immediate increase in the net tangible book value
of $0.32 per share to existing shareholders attributable to this offering. The following table illustrates this per
share dilution:
| Assumed
offering price per share of common stock |
|
|
|
|
|
$ |
1.72 |
|
| Net tangible book
value per share as of March 31, 2015 |
|
$ |
(0.09 |
) |
|
|
|
|
| Increase
in as adjusted net tangible book value per share attributable to the sale of shares under the Purchase Agreements |
|
|
0.32 |
|
|
|
|
|
| Pro
forma net tangible book value per share after the sale of shares under the Purchase Agreements |
|
|
|
|
|
|
0.23 |
|
| Dilution
per share to new investors |
|
|
|
|
|
$ |
1.49 |
|
To
the extent that we sell less than $10 million worth of shares under the Purchase Agreements, or to the extent that some or all
sales are made at prices lower than or in excess of the assumed price per share of $1.376, then the dilution reflected in the
table above will differ. The above table is based on 26,381,976 shares of our common stock outstanding as of March 31, 2015, adjusted
for the assumed sale of $10 million in shares to the Equity Purchasers under the Purchase Agreements at the assumed purchase price
described above and after deducting estimated offering commissions and expenses payable by us.
To
the extent that we issue additional shares of common stock in the future, there may be further dilution to investors participating
in this offering. In addition, we may choose to raise additional capital because of market conditions or strategic considerations,
even if we believe that we have sufficient funds for our current or future operating plans. If we raise additional capital through
the sale of equity or convertible debt securities, the issuance of these securities could result in further dilution to our shareholders.
The
number of shares of our common stock reflected in the discussion and calculations for the figures appearing in the table above
is based on 25,339,364 shares of our common stock outstanding as of March 31, 2015 and excludes, as of that date:
| |
● |
2,272,022
shares issuable upon exercise of outstanding options with a weighted average exercise price of $2.34; |
| |
● |
6,085,714
shares issuable upon exercise of outstanding warrants with a weighted average exercise price of $1.25; and |
| |
● |
400,302
shares available for future issuance under our equity incentive plans. |
MANAGEMENT'S
DISCUSSION AND ANALYSIS
OF
FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The
following discussion and analysis provides information that we believe is relevant to an assessment and understanding of our results
of operations and financial condition. You should read this analysis in conjunction with our audited consolidated financial statements
and related notes and our unaudited consolidated interim financial statements and their notes. This discussion and analysis contains
statements of a forward-looking nature relating to future events or our future financial performance. These statements are only
predictions, and actual events or results may differ materially. In evaluating such statements, you should carefully consider
the various factors identified in this prospectus, which could cause actual results to differ materially from those expressed
in, or implied by, any forward-looking statements, including those set forth in "Risk Factors" in this prospectus. See
"Cautionary Note Regarding Forward-Looking Statements."
Our
Business Overview
We
were incorporated in Delaware in 1987. We are a late-stage biopharmaceutical company developing product candidates intended to
address unmet medical needs in the areas of inflammation, oncology and biodefense. We maintain two active business segments: BioTherapeutics
and Vaccines/BioDefense.
Our
BioTherapeutics business segment is developing a first-in-class photo-dynamic therapy (SGX301) utilizing safe, visible light for
the treatment of cutaneous T-cell lymphoma (“CTCL”), proprietary formulations of oral beclomethasone 17,21-dipropionate
(“BDP”) for the prevention/treatment of gastrointestinal (“GI”) disorders characterized by severe inflammation,
including pediatric Crohn’s disease (SGX203) and acute radiation enteritis (SGX201), and our novel innate defense regulator
technology (SGX942) for the treatment of oral mucositis in head and neck cancer.
Our
Vaccines/BioDefense business segment includes active development programs for RiVax™, our ricin toxin vaccine candidate,
VeloThrax™, our anthrax vaccine candidate, OrbeShield™, our GI acute radiation syndrome (“GI ARS”) therapeutic
candidate and SGX943, our melioidosis therapeutic candidate. The development of our vaccine programs is supported by our heat
stabilization technology, known as ThermoVax™, under existing and on-going government contract funding. With the recently
awarded government contract from the National Institute of Allergy and Infectious Diseases (“NIAID”), we will attempt
to advance the development of RiVax™ to protect against exposure to ricin toxin. We plan to use the funds received under
our government contracts with the Biomedical Advanced Research and Development Authority (“BARDA”) and NIAID to advance
the development of OrbeShield™ for the treatment of GI ARS. Additionally, we have entered into a global and exclusive channel
collaboration with Intrexon Corporation (“Intrexon”) through which we intend to develop and commercialize a human
monoclonal antibody therapy (SGX101) to treat melioidosis.
An
outline of our business strategy follows:
| |
● |
Conduct
a Phase 3 clinical trial of SGX301 for the treatment of CTCL; |
| |
● |
Conduct
a Phase 2 clinical trial of SGX942 for the treatment of oral mucositis in head and neck cancer; |
| |
● |
Conduct
a Phase 3 clinical trial of oral BDP, known as SGX203 for the treatment of pediatric Crohn’s disease; |
| |
● |
Evaluate
the effectiveness of oral BDP in other therapeutic indications involving inflammatory conditions of the gastrointestinal (“GI”)
tract such as prevention of acute radiation enteritis; |
| |
● |
Develop
RiVax™ and VeloThrax™ in combination with our ThermoVax™ technology to develop new heat stable vaccines
in biodefense and infectious diseases with the potential to collaborate and/or partner with other companies in these areas; |
| |
● |
Advance
the preclinical and manufacturing development of OrbeShield™ as a biodefense medical countermeasure for the treatment
of GI ARS; |
| |
● |
Continue
to apply for and secure additional government funding for each of our BioTherapeutics and Vaccines/BioDefense programs through
grants, contracts and/or procurements; |
| |
● |
Acquire
or in-license new clinical-stage compounds for development; and |
| |
● |
Explore
other business development and merger/acquisition strategies, an example of which is our collaboration with Intrexon. |
Critical
Accounting Policies
Our
discussion and analysis of our financial condition and results of operations are based upon our consolidated financial statements,
which have been prepared in accordance with accounting principles generally accepted in the U.S. The preparation of these financial
statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities and expenses, and
related disclosure of contingent assets and liabilities. We evaluate these estimates and judgments on an on-going basis.
Intangible
Assets
One
of the most significant estimates or judgments that we make is whether to capitalize or expense patent and license costs. We make
this judgment based on whether the technology has alternative future uses, as defined in Financial Accounting Standards Board
(“FASB”) Accounting Standards Codification (“ASC”) 730, Research and Development. Based on this
consideration, we capitalized payments made to legal firms that are engaged in filing and protecting rights to intellectual property
rights for our current product candidates in both the domestic and international markets. We believe that patent rights are one
of our most valuable assets. Patents and patent applications are key components of intellectual property, especially in the early
stage of product development, as their purchase and maintenance gives us access to key product development rights from our academic
and industry partners. These rights can also be sold or sub-licensed as part of our strategy to partner our product candidates
at each stage of development as the intangible assets have alternative future use. The legal costs incurred for these patents
consist of work associated with filing new patents designed to protect, preserve, maintain and perhaps extending the lives of
the patents. We capitalize such costs and amortize intangibles over their expected useful life – generally a period of 11
to 16 years.
These
intangible assets are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount may
not be recoverable or if the underlying program is no longer being pursued. If the sum of the expected undiscounted cash flows
is less than the carrying value of the related asset or group of assets, a loss is recognized for the difference between the fair
value and carrying value of the related asset or group of assets.
Fair
Value of Financial Instruments
FASB
ASC 820 — Fair Value Measurements and Disclosures, defines fair value as the price that would be received to sell
an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. FASB ASC
820 requires disclosures about the fair value of all financial instruments, whether or not recognized, for financial statement
purposes. Disclosures about the fair value of financial instruments are based on pertinent information available to us on December
31, 2014. Accordingly, the estimates presented in these financial statements are not necessarily indicative of the amounts that
could be realized on disposition of the financial instruments.
FASB
ASC 820 specifies a hierarchy of valuation techniques based on whether the inputs to those valuation techniques are observable
or unobservable. Observable inputs reflect market data obtained from independent sources, while unobservable inputs reflect market
assumptions. The hierarchy gives the highest priority to unadjusted quoted prices in active markets for identical assets or liabilities
(Level 1 measurement) and the lowest priority to unobservable inputs (Level 3 measurement).
The
three levels of the fair value hierarchy are as follows:
| |
● |
Level
1 — Quoted prices in active markets for identical assets or liabilities that the reporting entity has the ability to
access at the measurement date. Level 1 primarily consists of financial instruments whose value is based on quoted market
prices such as exchange-traded instruments and listed equities. |
| |
● |
Level
2 — Inputs other than quoted prices included within Level 1 that are observable for the asset or liability, either directly
or indirectly. Level 2 includes financial instruments that are valued using models or other valuation methodologies. These
models consider various assumptions, including volatility factors, current market prices and contractual prices for the underlying
financial instruments. Substantially all of these assumptions are observable in the marketplace, can be derived from observable
data or are supported by observable levels at which transactions are executed in the marketplace. |
| |
● |
Level
3 — Unobservable inputs for the asset or liability. Financial instruments are considered Level 3 when their fair values
are determined using pricing models, discounted cash flows or similar techniques and at least one significant model assumption
or input is unobservable. |
The
carrying amounts reported in the consolidated balance sheet for cash and cash equivalents, accounts receivable, accounts payable
and accrued expenses approximate their fair value based on the short-term maturity of these instruments. We recognize all derivative
financial instruments as assets or liabilities in the financial statements and measure them at fair value with changes in fair
value reflected as current period income or loss unless the derivatives qualify as hedges. As a result, certain warrants issued
in connection with our June 2013 offering were accounted for as derivatives.
Research
and Development Costs
Research
and development costs are charged to expense when incurred in accordance with FASB ASC 730, Research and Development. Research
and development includes costs such as clinical trial expenses, contracted research and license agreement fees with no alternative
future use, supplies and materials, salaries, stock based compensation, employee benefits, equipment depreciation and allocation
of various corporate costs. Purchased in-process research and development expense represents the value assigned or paid for acquired
research and development for which there is no alternative future use as of the date of acquisition.
Revenue
Recognition
Principally
our revenues are generated from government contracts and grants. Recording of revenue is applied in accordance with FASB ASC 605,
Revenue Recognition and/or ASC 605-25, Revenue Recognition – Multiple Element Arrangements. The revenue from
government contracts and grants is based upon subcontractor costs and internal costs incurred that are specifically covered by
the grants, plus a facilities and administrative rate that provides funding for overhead expenses. These revenues are recognized
when expenses have been incurred by subcontractors or when we incur internal expenses that are related to the grant.
Accounting
for Warrants
We
considered FASB ASC 815, Evaluating Whether an Instrument is Considered Indexed to an Entity’s Own Stock, which provides
guidance for determining whether an equity-linked financial instrument (or embedded feature) issued by an entity is indexed to
the entity’s stock, and therefore, qualifying for the first part of the scope exception in paragraph 815-10-15. We evaluated
the provisions in our outstanding warrants and determined that warrants issued in connection with our June 2013 registered public
offering contain provisions that protect holders from a decline in the issue price of our common stock (or “down-round”
provisions) and contain net settlement provisions. Consequently, these warrants are recognized as liabilities at their fair value
on the date of grant and remeasured at fair value on each reporting date. All other warrants issued were indexed to our own stock
and therefore are accounted for as equity instruments for 2015 and 2014.
Stock-Based
Compensation
Stock
options are issued with an exercise price equal to the market price on the date of issuance. Stock options issued to directors
upon re-election vest quarterly for a period of one year (new director issuances are fully vested upon issuance). Stock options
issued to employees vest 25% immediately as of the grant date, then 25% each subsequent year for a period of three years. Stock
options vest over each three month period from the date of issuance to the end of the three year period. These options have a
ten year life for as long as the individuals remain employees or directors. In general, when an employee or director terminates
their position the options will expire within three months, unless otherwise extended by the Board.
From
time to time, we issue restricted shares of our common stock to vendors and consultants as compensation for services performed.
Stock-based compensation expense recognized during the period is based on the fair value of the portion of share-based payment
awards that is ultimately expected to vest during the period. Typically these instruments vest upon issuance and therefore the
entire stock compensation expense is recognized upon issuance to the vendors and/or consultants.
We
determine stock-based compensation expense for options, warrants and shares of common stock granted to non-employees in accordance
with FASB ASC 718, Stock Compensation, and FASB ASC 505-50, Equity-Based Payments to Non-Employees, and represents
the fair value of the consideration received, or the fair value of the equity instruments issued, whichever may be more reliably
measured. For options that vest over future periods, the fair value of options granted to non-employees is amortized as the options
vest. Stock-based compensation expense recognized during the period is based on the value of the portion of share-based payment
awards that is ultimately expected to vest during the period.
Results of Operations
Three Months Ended March 31,
2015 Compared to March 31, 2014
For the three months ended
March 31, 2015, we had a net loss of $4,569,322 as compared to a net loss of $3,331,708 for same period in the prior year, representing
an increase in the net loss of $1,237,614, or 37%. Included in the net loss for March 31, 2015 is a non-cash charge of $3,011,616
as compared to $1,742,090 for the same period in the prior year representing the change in the fair value of the warrant liability
related to warrants issued in connection with our June 2013 registered public offering.
For the three
months ended March 31, 2015, revenues relate to government contracts and grants awarded in support of our development in OrbeShield™
in GI ARS and RiVax™. We had revenues of $816,286 as compared to $910,597 for the same period in the prior year, representing
a decrease of $94,311, or 10%. The decrease in revenues was a result of our ThermoVax™ grant expiring during the fourth quarter
of 2014.
We incurred costs
related to those revenues for the three months ended March 31, 2015 and 2014 of $527,399 and $628,981, respectively, representing
a decrease of $101,582, or 16%. These costs relate to allocated employee costs and payments due to subcontractors in connection
with research performed pursuant to the contracts and grants.
Our gross profit
for the three months ended March 31, 2015 was $288,887, as compared to $281,616 for the same period in 2014, representing an increase
of $7,271, or 3%.
Research and development
spending was $1,029,884 for the three months ended March 31, 2015 as compared to $1,030,621 for the same period in 2014, representing
a nominal decrease.
General and administrative
expenses were $817,270 for the three months ended March 31, 2015, as compared to $840,904 for the same period in 2014, representing
a nominal decrease.
Other income (expense)
for the three months ended March 31, 2015 was $(3,011,055) as compared to $(1,741,799) for the same period in 2014, representing
an increase of $1,269,256, or 73%. The increase in expense is primarily attributable to a greater non-cash charge in 2015 compared
to 2014 for the change in the fair value of the warrant liability related to warrants issued in connection with our June 2013 registered
public offering.
Year Ended December 31, 2014
Compared to 2013
For the year ended December
31, 2014, we had a net loss of $6,706,972 as compared to a net loss of $10,058,996 for the prior year, representing a decreased
loss of $3,352,024 or 33%. Included in the net loss for December 31, 2014 is a non-cash gain of $3,436,195 versus a non-cash expense
of $3,654,770 for December 31, 2013 which represents the change in the fair value of the warrant liability related to warrants
issued in connection with our registered public offering in June 2013. During the third quarter of 2014, we completed the acquisition
of Hypericin, SGX301, for which we issued common stock with a value of $3,750,000 and paid cash of $250,000 which was recognized
as acquired in-process research and development expense. Additionally, we continued our progress on the Phase 2 clinical trial
with SGX942 for patients suffering from oral mucositis associated with their chemoradiation therapy, (“CRT”) for head
and neck cancer.
For the year
ended December 31, 2014 and 2013, revenues and associated costs relate to government contracts and grants awarded in support of
the development of ThermoVax™, RiVax™ GI-ARS, orBec® and OrbeShield™ in GI ARS. For the year ended
December 31, 2014, we had revenues of $7,043,016 as compared to $3,224,152 for the prior year, representing an increase of $3,818,864
or 118%. The increase in revenues was a result of research and development activities performed under our government contracts
associated with OrbeShield™ and the initiation of a research and development government contract in the fourth quarter for
RiVax™.
We incurred costs
related to contract and grant revenues in the year ended December 31, 2014 and 2013 of $5,313,855 and $2,544,285, respectively,
representing an increase of $2,769,570 or 109%. These costs primarily relate to payments made to subcontractors and allocated employee
costs in connection with research performed pursuant to contracts and grants. The fluctuations are due to the development activity
performed on the contracts and grants discussed above.
Our gross profit for the
year ended December 31, 2014 was $1,729,161 as compared to $679,867 for the prior year, representing an increase of $1,049,294
or 154%. This increase is due primarily to the OrbeShield™ and RiVax™ contracts which provide a management fee and
higher negotiated reimbursement for fixed overhead.
Research and development,
including acquired in-process research and development costs, increased by $4,015,356 or 79%, to $9,086,535 for the year ended
December 31, 2014 as compared to $5,071,179 for the prior year. This increase is primarily related to the acquisition of Hypericin,
SGX301, for which we issued common stock with a value of $3,750,000 and paid cash of $250,000 which was recognized as acquired
in-process research and development expense. Additionally, we continued our progress on the Phase 2 clinical trial with SGX942
for patients suffering from oral mucositis associated with their CRT for head and neck cancer.
General and administrative
expenses increased by $638,745 or 23%, to $3,403,975 for the year ended December 31, 2014, as compared to $2,765,230 for the prior
year. This increase is primarily related to increased headcount and an increase in outside professional services.
Other income (expense)
for the year ended December 31, 2014 was $3,437,505 as compared to $(3,652,810) for the prior year. The change is primarily related
to non-cash income of $3,436,195 which represents the change in the fair value of the warrant liability related to warrants issued
in connection with our June 2013 registered public offering for the year ended December 31, 2014 as compared to a non-cash expense
of $(3,654,770) for the year ended December 31, 2013.
During the year ended December
31, 2014, in accordance with the State of New Jersey’s Technology Business Tax Certificate Program, which allowed certain
high technology and biotechnology companies to sell unused net operating loss (“NOL”) carryforwards to other New Jersey-based
corporate taxpayers based in New Jersey, we sold New Jersey NOL carryforwards, resulting in the recognition of $616,872 of income
tax benefit, net of transaction costs as compared to $750,356 for the year ended December 31, 2013. There can be no assurance as
to the continuation or magnitude of this program in future years.
Business Segments
We maintained two active
business segments for the year ended December 31, 2014 and December 31, 2013: Vaccines/BioDefense and BioTherapeutics.
Revenues for the Vaccines/BioDefense
business segment for the year ended December 31, 2014 were $6,756,388 as compared to $3,003,822 for the year ended December 31,
2013, representing an increase of $3,752,566 or 125%. The increase in revenues were a result of our OrbeShield™ contracts
and initiating the RiVax™ contract during the fourth quarter of 2014. Revenues for the BioTherapeutics business segment for
the year ended December 31, 2014 were $286,628 as compared to $220,330 for the year ended December 31, 2013, representing an increase
of $66,298 or 30%. This increase is primarily related to work performed under our GI ARS and oral mucositis grants.
Income (loss) from operations
for the Vaccines/BioDefense business segment for the year ended December 31, 2014 was $807,164 as compared to $(1,666,130) for
the year ended December 31, 2013. Income from operations is primarily attributable to our gross margins related to our government
contracts. Loss from operations for the BioTherapeutics business segment for the year ended December 31, 2014 was $7,674,381 as
compared to $3,069,998 for the year ended December 31, 2013, representing an increase of $4,604,383. This increased loss is due
primarily to the acquisition of Hypericin, SGX301, for which we issued common stock with a value of $3,750,000 and paid cash of
$250,000 which was recognized as acquired in-process research and development expense and our continued progress in the Phase 2
clinical trial with SGX942 for patients suffering from oral mucositis associated with their CRT for head and neck cancer.
Amortization and depreciation
expense for the Vaccines/BioDefense business segment for the year ended December 31, 2014 was $39,625 as compared to $37,981 for
the year ended December 31, 2013. Amortization and depreciation expense for the BioTherapeutics business segment for the year ended
December 31, 2014 was $199,196 as compared to $190,033 for the year ended December 31, 2013.
Financial Condition and Liquidity
Cash and Working Capital
As of March 31, 2015, we had cash and cash
equivalents of $5,012,605 as compared to $5,525,094 as of December 31, 2014, representing a decrease of $512,489 or 9%. As of March
31, 2015, we had working capital of $2,913,440 as compared to working capital of $3,174,214 as of December 31, 2014, which excludes
a non-cash warrant liability of $5,152,367 and $3,789,562, respectively, representing a decrease of $260,777, or 8%. This decrease
is primarily related to expenditures to support the Phase 2 clinical trial of SGX942 for the treatment of oral mucositis in head
and neck cancer.
Based on the Company’s current rate of
cash outflows, cash on hand, proceeds from government contract and grant programs, proceeds available from the Lincoln Park equity
line and proceeds from the State of New Jersey Technology Business Tax Certificate Transfer Program, management believes that its
current cash will be sufficient to meet the anticipated cash needs for working capital and capital expenditures for at least the
next twelve months, as of March 31, 2015.
Our plans with respect to our liquidity management
include, but are not limited to, the following:
| |
● |
We have
up to $50.5 million in active contract and grant funding still available to support our associated research programs in 2015
and beyond. We plan to submit additional contract and grant applications for further support of these programs with various
funding agencies. |
| |
|
|
| |
● |
We have continued to use equity
instruments to provide a portion of the compensation due to vendors and collaboration partners and expect to continue to do
so for the foreseeable future. |
| |
|
|
| |
● |
We will
pursue NOL sales in the State of New Jersey pursuant to its Technology Business Tax Certificate Transfer Program. Based on
the receipt of $616,872 in proceeds from the sale of NJ NOL in 2014, we expect to participate in the program during 2015 and
beyond as the program is available. |
| |
|
|
| |
● |
We have
a $10.6 million equity facility, with Lincoln Park, through October 2016, of which approximately $9.3 million is available
as of March 31, 2015. |
| |
|
|
| |
● |
We may
seek additional capital in the private and/or public equity markets to continue our operations, respond to competitive pressures,
develop new products and services, and to support new strategic partnerships. We are currently evaluating additional equity
financing opportunities and may execute them when appropriate. However, there can be no assurances that we can consummate
such a transaction, or consummate a transaction at favorable pricing. |
Expenditures
Under our budget and based
upon our existing product development agreements, license agreements pursuant to letters of intent and option agreements, we expect
our total research and development expenditures for the next 12 months to be approximately $16.1 million before any grant reimbursements,
of which $5.7 million relates to the BioTherapeutics business and $10.4 million relates to the Vaccines/BioDefense business. We
anticipate contract and grant revenues in the next 12 months of approximately $10.4 million to offset research and development
expenses in the Vaccines/BioDefense business segment.
The table below details
our costs for research and development by program and amounts reimbursed for the years ended December 31, 2014 and 2013, and the
three months ended March 31, 2015 and 2014:
| | |
Years
Ended
December 31, | | |
Three Months Ended
March 31, | |
| | |
2014 | | |
2013 | | |
2015 | | |
2014 | |
| Research & Development Expenses | |
| | |
| | |
| | |
| |
| Oral BDP | |
$ | 561,655 | | |
$ | 1,467,077 | | |
$ | - | | |
$ | 271,438 | |
| RiVax™ & ThermoVax™ Vaccines | |
| 846,870 | | |
| 1,113,430 | | |
| 192,923 | | |
| 177,318 | |
| SGX94 | |
| 2,820,807 | | |
| 659,809 | | |
| 513,442 | | |
| 495,294 | |
| SGX943/101 | |
| 19,378 | | |
| 1,500,000 | | |
| 7,227 | | |
| - | |
| SGX301 | |
| 4,369,585 | | |
| - | | |
| 247,396 | | |
| - | |
| Other | |
| 468,240 | | |
| 330,863 | | |
| 68,896 | | |
| 86,571 | |
| Total | |
$ | 9,086,535 | | |
$ | 5,071,179 | | |
$ | 1,029,884 | | |
$ | 1,030,621 | |
| | |
| | | |
| | | |
| | | |
| | |
| Reimbursed under Government Contracts and Grants | |
| | | |
| | | |
| | | |
| | |
| OrbeShield™ | |
$ | 4,100,663 | | |
$ | 672,194 | | |
$ | 294,313 | | |
$ | 460,139 | |
| RiVax™ & ThermoVax™ Vaccines | |
| 930,573 | | |
| 1,872,091 | | |
| 165,814 | | |
| 168,842 | |
| Other | |
| 282,619 | | |
| - | | |
| 67,272 | | |
| - | |
| Total | |
$ | 5,313,855 | | |
$ | 2,544,285 | | |
$ | 527,399 | | |
$ | 628,981 | |
| | |
| | | |
| | | |
| | | |
| | |
| Grand Total | |
$ | 14,400,390 | | |
$ | 7,615,464 | | |
$ | 1,557,283 | | |
$ | 1,659,602 | |
Contractual Obligations
We have commitments of
approximately $500,000 as of March 31, 2015 relating to several licensing agreements with consultants and universities. Additionally,
we have collaboration and license agreements, which upon clinical or commercialization success may require the payment of milestones
of up to $7.9 million and/or royalties up to 6% of net sales of covered products, if and when achieved. However, there can be no
assurance that clinical or commercialization success will occur.
In December 2014, we entered
into a lease agreement through May 31, 2018 for existing and expanded office space. The rent for the first 12 months is approximately
$12,300 per month, or approximately $20.85 per square foot. This rent increases to approximately $12,375 per month, or approximately
$20.95 per square foot, for the next 12 months, and thereafter to approximately $12,460 per month, or approximately $21.13 per
square foot for the remainder of the lease.
On September 3, 2014, we
entered into an asset purchase agreement with Hy Biopharma, Inc. (“Hy Biopharma”) pursuant to which we acquired certain
intangible assets, properties and rights of Hy Biopharma related to the development of Hy BioPharma’s synthetic hypericin
product. As consideration for the assets acquired, we paid $250,000 in cash and issued 1,849,113 shares of common stock with a
fair value of $3,750,000. These amounts were charged to research and development expense during the third quarter of 2014 as the
assets will be used in our research and development activities and do not have alternative future use pursuant to generally accepted
accounting principles in the United States. Provided all future success-oriented milestones are attained, we will be required to
make payments of up to $10.0 million, if and when achieved. Payments will be payable in restricted securities of the Company not
to exceed 19.9% ownership of its outstanding stock.
On April 27, 2013, we entered
into an exclusive channel collaboration agreement (the “Channel Agreement”) with Intrexon to use Intrexon’s advanced
human antibody discovery, isolation and production technologies for the development of human monoclonal antibody therapies for
a new biodefense application targeting melioidosis. The Channel Agreement grants an exclusive worldwide license to use specified
patents and other intellectual property of Intrexon in connection with the research, development, use, importing, manufacture,
sale and offer for sale of products for the treatment of melioidosis through the use of exogenously produced human recombinant
monoclonal antibodies. The Channel Agreement, upon clinical or commercialization success, may require the payment of certain milestones
payments up to $7 million, if and when achieved.
In February 2007, our Board
of Directors authorized the issuance of 50,000 shares of our common stock to Dr. Schaber immediately prior to the completion of
a transaction, or series or a combination of related transactions, negotiated by our Board of Directors whereby, directly or indirectly,
a majority of our capital stock or a majority of our assets are transferred from us and/or our stockholders to a third party. Dr.
Schaber’s amended employment agreement includes our obligation to issues such shares if such event occurs.
As a result of these agreements, we have future
contractual obligations over the next five years as follows:
| Year |
|
|
Research and
Development |
|
|
Property and
Other Leases |
|
|
Total |
|
| |
April
1 through December 31, 2015 |
|
|
$ |
100,000 |
|
|
$ |
99,000 |
|
|
$ |
199,000 |
|
| |
2015 |
|
|
|
100,000 |
|
|
|
157,000 |
|
|
|
257,000 |
|
| |
2016 |
|
|
|
100,000 |
|
|
|
152,000 |
|
|
|
252,000 |
|
| |
2017 |
|
|
|
100,000 |
|
|
|
52,000 |
|
|
|
152,000 |
|
| |
2018 |
|
|
|
100,000 |
|
|
|
- |
|
|
|
100,000 |
|
| |
Total |
|
|
$ |
500,000 |
|
|
$ |
460,000 |
|
|
$ |
960,000 |
|
Off-Balance Sheet Arrangements
We do not have any off-balance
sheet arrangements, as defined in Item 303(a)(4) of Regulation S-K.
BUSINESS
Our Business Overview
We
are a late-stage biopharmaceutical company developing product candidates intended to address unmet medical needs in the areas of
inflammation, oncology, and biodefense. We maintain two active business segments: BioTherapeutics and Vaccines/BioDefense.
Our
BioTherapeutics business segment is developing a first-in-class photo-dynamic therapy (SGX301) utilizing safe, visible light for
the treatment of cutaneous T-cell lymphoma (“CTCL”), proprietary formulations of oral beclomethasone 17,21-dipropionate
(“BDP”) for the prevention/treatment of gastrointestinal (“GI”) disorders characterized by severe inflammation,
including pediatric Crohn’s disease (SGX203) and acute radiation enteritis (SGX201), and our novel innate defense regulator
(“IDR”) technology (SGX942) for the treatment of oral mucositis in head and neck cancer.
Our Vaccines/BioDefense
business segment includes active development programs for RiVax™, our ricin toxin vaccine candidate, VeloThrax™, our
anthrax vaccine candidate, OrbeShield™, our GI acute radiation syndrome (“GI ARS”) therapeutic candidate and
SGX943, our melioidosis therapeutic candidate. The development of our vaccine programs is supported by our heat stabilization technology,
known as ThermoVax™, under existing and on-going government contract funding. With the recently awarded government contract
from the National Institute of Allergy and Infectious Diseases (“NIAID”), we will attempt to advance the development
of RiVax™ to protect against exposure to ricin toxin. We plan to use the funds received under our government contracts with
the Biomedical Advanced Research and Development Authority (“BARDA”) and NIAID to advance the development of OrbeShield™
for the treatment of GI ARS. Additionally, we have entered into a global and exclusive channel collaboration with Intrexon Corporation
(“Intrexon”) through which we intend to develop and commercialize a human monoclonal antibody therapy (SGX101) to treat
melioidosis.
An outline for our business
strategy follows:
| |
● |
Conduct a Phase 3 clinical trial for SGX301 for the treatment of CTCL; |
| |
● |
Conduct a Phase 2 clinical trial of SGX942 for the treatment of oral mucositis in head and neck cancer; |
| |
● |
Initiate a Phase 3 clinical trial of oral BDP, known as SGX203, for the treatment of pediatric Crohn’s disease; |
| |
● |
Evaluate the effectiveness of oral BDP in other therapeutic indications involving inflammatory conditions of the GI tract such as prevention of acute radiation enteritis; |
| |
● |
Develop RiVax™ and VeloThrax™ in combination with our ThermoVax™ technology, to develop new heat stable vaccines in biodefense and infectious diseases with the potential to collaborate and/or partner with other companies in these areas; |
| |
● |
Advance the preclinical and manufacturing development of OrbeShield™ as a biodefense medical countermeasure for the treatment of GI ARS; |
| |
● |
Continue to apply for and secure additional government funding for each of our BioTherapeutics and Vaccines/BioDefense programs through grants, contracts and/or procurements; |
| |
● |
Acquire or in-license new clinical-stage compounds for development; and |
| |
● |
Explore other business development and merger/acquisition strategies, an example of which is our collaboration with Intrexon. |
Corporate Information
We were incorporated in
Delaware in 1987 under the name Biological Therapeutics, Inc. In 1987, we merged with Biological Therapeutics, Inc., a North Dakota
corporation, pursuant to which we changed our name to “Immunotherapeutics, Inc.” We changed our name to “Endorex
Corp.” in 1996, to “Endorex Corporation” in 1998, to “DOR BioPharma, Inc.” in 2001, and finally to
“Soligenix, Inc.” in 2009. Our principal executive offices are located at 29 Emmons Drive, Suite C-10, Princeton, New
Jersey 08540 and our telephone number is (609) 538-8200.
Our Product Candidates in Development
The following tables summarize
our product candidates under development:
| BioTherapeutic Product Candidates |
| |
|
|
|
|
Soligenix Product
Candidate |
|
Therapeutic Indication |
|
Stage of Development |
| SGX301 |
|
Cutaneous T-Cell Lymphoma |
|
Phase 2 trial completed; demonstrated significantly
higher response rate (p ≤ 0.04) compared to placebo;
Phase 3 clinical trial planned for the second
half of 2015, with data expected in the second half of 2016 |
| |
|
|
|
|
| SGX942 |
|
Oral Mucositis in Head and Neck Cancer |
|
Phase 2 trial initiated in the second half of 2013, with data expected in the second half of 2015 |
| |
|
|
|
|
| SGX203** |
|
Pediatric Crohn’s disease |
|
Phase 1/2 clinical trial completed June 2013,
efficacy data, pharmacokinetic (PK)/pharmacodynamic (PD) profile and safety confirmed;
Phase 3 clinical trial planned for the second
half of 2015, with data expected in the second half of 2017 |
| |
|
|
|
|
| SGX201** |
|
Acute Radiation Enteritis |
|
Phase 1/2 clinical trial complete;
safety and preliminary efficacy demonstrated;
Phase 2 trial planned
for the first half of 2016, with data expected in the first half of 2017 |
| |
|
|
|
|
| Vaccine Thermostability Platform** |
| |
|
|
|
|
Soligenix Product
Candidate |
|
Indication |
|
Stage of Development |
| ThermoVax™ |
|
Thermostability of aluminum adjuvanted vaccines |
|
Pre-clinical |
| |
|
|
|
|
| BioDefense Product Candidates** |
| |
|
|
|
|
Soligenix Product
Candidate |
|
Indication |
|
Stage of Development |
| RiVax™ |
|
Vaccine against
Ricin Toxin Poisoning |
|
Phase 1B trial complete, safety and neutralizing
antibodies for protection demonstrated;
Phase 1/2 trial planned for the second half
of 2015 |
| |
|
|
|
|
| VeloThrax™ |
|
Vaccine against Anthrax Poisoning |
|
Pre-clinical;
Phase 1 clinical trial planned for second half
of 2016 |
| |
|
|
|
|
| OrbeShield™ |
|
Therapeutic against GI ARS |
|
Pre-clinical program initiated |
| |
|
|
|
|
| SGX943/SGX101 |
|
Melioidosis |
|
Pre-clinical program initiated |
** Contingent
upon continued government contract and grant funding.
BioTherapeutics Overview
SGX301 – for Treating Cutaneous
T-Cell Lymphoma
SGX301 is a novel, first-in-class,
photodynamic therapy that utilizes safe visible light for activation. The active ingredient in SGX301 is synthetic hypericin, a
photosensitizer which is topically applied to skin lesions and then activated by fluorescent light 16 to 24 hours later. Hypericin
is also found in several species of Hypericum plants, although the drug used in SGX301 is chemically synthesized by a proprietary
manufacturing process and not extracted from plants. Importantly, hypericin is optimally activated with visible light thereby avoiding
the negative consequences of ultraviolet light. Other light therapies using UVA light result in serious adverse effects including
secondary skin cancers.
Combined with photoactivation,
in clinical trials hypericin has demonstrated significant anti-proliferative effects on activated normal human lymphoid cells and
inhibited growth of malignant T-cells isolated from CTCL patients. In both settings, it appears that the mode of action is an induction
of cell death in a concentration as well as a light dose-dependent fashion. These effects appear to result, in part, from the generation
of singlet oxygen during photoactivation of hypericin.
Hypericin is one of the
most efficient known generators of singlet oxygen, the key component for phototherapy. The generation of singlet oxygen induces
necrosis and apoptosis in adjacent cells. The use of topical hypericin coupled with directed visible light results in generation
of singlet oxygen only at the treated site. We believe that the use of visible light (as opposed to cancer-causing ultraviolet
light) is a major advance in photodynamic therapy. In a published Phase 2 clinical study in CTCL, after six weeks of twice-weekly
therapy, a majority of patients experienced a statistically significant (p-value ≤ 0.04) improvement with topical hypericin
treatment whereas the placebo was ineffective: 58.3% compared to 8.3%, respectively.
SGX301 has received orphan
drug designation as well as Fast Track designation from the United States Food and Drug Administration (the “FDA”).
The Orphan Drug Act is intended to assist and encourage companies to develop safe and effective therapies for the treatment of
rare diseases and disorders. In addition to providing a seven-year term of market exclusivity for SGX301 upon final FDA approval,
orphan drug designation also positions us to be able to leverage a wide range of financial and regulatory benefits, including government
grants for conducting clinical trials, waiver of FDA user fees for the potential submission of a New Drug Application (“NDA”)
for SGX301, and certain tax credits. In addition, Fast Track is a designation that the FDA reserves for a drug intended to treat
a serious or life-threatening condition and one that demonstrates the potential to address an unmet medical need for the condition.
Fast Track designation is designed to facilitate the development and expedite the review of new drugs. For instance, should events
warrant, we will be eligible to submit a NDA for SGX301 on a rolling basis, permitting the FDA to review sections of the NDA prior
to receiving the complete submission. Additionally, NDAs for Fast Track development programs ordinarily will be eligible for priority
review.
We anticipate initiating
a Phase 3 clinical study of SGX301 in the treatment of CTCL in the second half of 2015.
We estimate the potential
worldwide market for SGX301 is in excess of $250 million for all applications, including the treatment of CTCL. This potential
market information is a forward-looking statement, and investors are urged not to place undue reliance on this statement. While
we have determined this potential market size based on assumptions that we believe are reasonable, there are a number of factors
that could cause our expectations to change or not be realized. See “Risk Factors” and “Cautionary Note Regarding
Forward-Looking Statements – Industry Data and Market Information.”
Cutaneous T-Cell Lymphoma
CTCL is a class of non-Hodgkin’s
lymphoma (“NHL”), a type of cancer of the white blood cells that are an integral part of the immune system. Unlike
most NHLs, which generally involve B-cell lymphocytes (involved in producing antibodies), CTCL is caused by an expansion of malignant
T-cell lymphocytes (involved in cell-mediated immunity) normally programmed to migrate to the skin. These skin-trafficking malignant
T-cells migrate to the skin, causing various lesions to appear that may change shape as the disease progresses, typically beginning
as a rash and eventually forming plaques and tumors. Mycosis fungoides (“MF”) is the most common form of CTCL. It generally
presents with skin involvement only, manifested as scaly, erythematous patches. Advanced disease with diffuse lymph node and visceral
organ involvement is usually associated with a poorer response rate to standard therapies. A relatively uncommon sub-group of CTCL
patients present with extensive skin involvement and circulating malignant cerebriform T-cells, referred to as Sézary syndrome.
These patients have substantially graver prognoses than those with MF.
CTCL mortality is related
to stage of disease, with median survival generally ranging from about 12 years in the early stages to only 2.5 years when the
disease has advanced. There is currently no FDA-approved drug for front-line treatment of early stage CTCL. Treatment of early-stage
disease generally involves skin-directed therapies. One of the most common unapproved therapies used for early-stage disease is
oral 5 or 8-methoxypsoralen (“Psoralen”) given with ultraviolet A (“UVA”) light, referred to as PUVA, which
is approved for dermatological conditions such as disabling psoriasis not adequately responsive to other forms of therapy, idiopathic
vitiligo and skin manifestations of CTCL in persons who have not been responsive to other forms of treatment. Psoralen is a mutagenic
chemical that interferes with DNA causing mutations and other malignancies. Moreover, UVA is a carcinogenic light source that when
combined with the Psoralen, results in serious adverse effects including secondary skin cancers; therefore, the FDA requires a
Black Box warning for PUVA.
CTCL constitutes a rare
group of NHLs, occurring in about 4% of the approximate 500,000 individuals living with NHL. We estimate, based upon review of
historic published studies and reports and an interpolation of data on the incidence of CTCL, that it affects over 20,000 individuals
in the U.S., with approximately 2,800 new cases seen annually.
SGX94
SGX94 is an IDR that regulates
the innate immune system to simultaneously reduce inflammation, eliminate infection and enhance tissue healing.
SGX94 is based on a new
class of short, synthetic peptides known as innate defense regulators (“IDRs”) that have a novel mechanism of action
in that it is simultaneously anti-inflammatory and anti-infective. IDRs have no direct antibiotic activity but modulate host responses,
increasing survival after infections with a broad range of bacterial Gram-negative and Gram-positive pathogens including both antibiotic
sensitive and resistant strains, as well as accelerating resolution of tissue damage following exposure to a variety of agents
including bacterial pathogens, trauma and chemo- or radiation-therapy. IDRs represent a novel approach to the control of infection
and tissue damage via highly selective binding to an intracellular adaptor protein, sequestosome-1, also known as p62, which has
a pivotal function in signal transduction during activation and control of the innate defense system. Preclinical data indicate
that IDRs may be active in models of a wide range of therapeutic indications including life-threatening bacterial infections as
well as the severe side-effects of chemo- and radiation-therapy.
SGX94 has demonstrated
efficacy in numerous animal disease models including mucositis, colitis, skin infection and other bacterial infections and has
been evaluated in a double-blind, placebo-controlled Phase 1 clinical trial in 84 healthy volunteers with both single ascending
dose and multiple ascending dose components. SGX94 was shown to be safe and well-tolerated in all dose groups when administered
by IV over 7 days and was consistent with safety results seen in pre-clinical studies. SGX94 is the subject of an open Investigational
New Drug (“IND”) application which has been cleared by the FDA. We believe that market opportunities for SGX94 include
mucositis, acute methicillin resistant Staphylococcus aureus (MRSA) bacterial infections, acinetobacter, melioidosis, acute
radiation syndrome and as a vaccine adjuvant, with potential opportunities for non-dilutive funding to support the development.
SGX942 – for Treating Oral Mucositis in
Head and Neck Cancer
SGX942 is our product candidate
containing our IDR technology platform, SGX94, targeting the treatment of oral mucositis in head and neck cancer patients. Oral
mucositis in this patient population is an area of unmet medical need where there are currently no approved drug therapies. Accordingly,
we received “Fast Track” designation for the treatment of oral mucositis as a result of radiation and/or chemotherapy
treatment in head and neck cancer patients from the FDA in the first half of 2013.
We initiated a Phase 2
clinical study of SGX942 in the treatment of oral mucositis in head and neck cancer patients in the second half of 2013.
We estimate the potential
worldwide market for SGX942 is in excess of $500 million for all applications, including the treatment of oral mucositis. This
potential market information is a forward-looking statement, and investors are urged not to place undue reliance on this statement.
While we have determined this potential market size based on assumptions that we believe are reasonable, there are a number of
factors that could cause our expectations to change or not be realized. See “Risk Factors” and “Cautionary Note
Regarding Forward-Looking Statements – Industry Data and Market Information.”
Oral Mucositis
Mucositis is the clinical
term for damage done to the mucosa by anticancer therapies. It can occur in any mucosal region, but is most commonly associated
with the mouth, followed by the small intestine. We estimate, based upon our review of historic studies and reports, and an interpolation
of data on the incidence of mucositis, that mucositis affects approximately 500,000 people in the U.S. per year and occurs in 40%
of patients receiving chemotherapy. Mucositis can be severely debilitating and can lead to infection, sepsis, the need for parenteral
nutrition and narcotic analgesia. The GI damage causes severe diarrhea. These symptoms can limit the doses and duration of cancer
treatment, leading to sub-optimal treatment outcomes.
The mechanisms of mucositis
have been extensively studied and have been recently linked to the interaction of chemotherapy and/or radiation therapy with the
innate defense system. Bacterial infection of the ulcerative lesions is regarded as a secondary consequence of dysregulated local
inflammation triggered by therapy-induced cell death, rather than as the primary cause of the lesions.
We estimate, based upon
our review of historic studies and reports, and an interpolation of data on the incidence of oral mucositis, that oral mucositis
is a subpopulation of approximately 90,000 patients in the U.S., with a comparable number in Europe. Oral mucositis almost
always occurs in patients with head and neck cancer treated with radiation therapy (greater than 80% incidence of severe mucositis)
and is common in patients undergoing high dose chemotherapy and hematopoietic cell transplantation, where the incidence and severity
of oral mucositis depends greatly on the nature of the conditioning regimen used for myeloablation.
Oral BDP
Oral BDP (beclomethasone
17,21-dipropionate) represents a first-of-its-kind oral, locally acting therapy tailored to treat GI inflammation.
BDP has been marketed in the U.S. and worldwide since the early 1970s as the active pharmaceutical ingredient in a nasal spray
and in a metered-dose inhaler for the treatment of patients with allergic rhinitis and asthma. Oral BDP is specifically formulated
for oral administration as a single product consisting of two tablets. One tablet is intended to release BDP in the upper sections
of the GI tract and the other tablet is intended to release BDP in the lower sections of the GI tract.
Based on its pharmacological
characteristics, oral BDP may have utility in treating other conditions of the gastrointestinal tract having an inflammatory component.
We are planning to pursue development programs in the treatment of pediatric Crohn’s disease, acute radiation enteritis and
GI ARS pending further grant funding. We are also exploring the possibility of testing oral BDP for local inflammation associated
with ulcerative colitis, among other indications.
We are pursuing orphan
drug designations for relevant indications as appropriate in both the U.S. and Europe. An orphan drug designation provides seven
and ten years of market exclusivity upon approval, in the U.S. and Europe, respectively.
SGX203 –for Treating Pediatric
Crohn’s Disease
SGX203 is a two tablet
delivery system of BDP specifically designed for oral use that allows for administration of immediate and delayed release BDP throughout
the small bowel and the colon. The FDA has given SGX203 orphan drug designation as well as Fast Track designation for the treatment
of pediatric Crohn's disease.
We anticipate initiating
a Phase 3 clinical study of SGX203 in the treatment of pediatric Crohn’s disease in the second half of 2015.
We estimate the potential
worldwide market for oral BDP is in excess of $500 million for all applications, including the treatment of pediatric Crohn’s
disease. This potential market information is a forward-looking statement, and investors are urged not to place undue reliance
on this statement. While we have determined this potential market size based on assumptions that we believe are reasonable, there
are a number of factors that could cause our expectations to change or not be realized. See “Risk Factors” and “Cautionary
Note Regarding Forward-Looking Statements – Industry Data and Market Information.”
Pediatric Crohn's Disease
Crohn's disease causes
inflammation of the GI tract. Crohn's disease can affect any area of the GI tract, from the mouth to the anus, but it most commonly
affects the lower part of the small intestine, called the ileum. The swelling caused by the disease extends deep into the lining
of the affected organ. The swelling can induce pain and can make the intestines empty frequently, resulting in diarrhea. Because
the symptoms of Crohn's disease are similar to other intestinal disorders, such as irritable bowel syndrome and ulcerative colitis,
it can be difficult to diagnose. People of Ashkenazi Jewish heritage have an increased risk of developing Crohn's disease.
Crohn's disease can appear
at any age, but it is most often diagnosed in adults in their 20s and 30s. However, approximately 30% of people with Crohn's disease
develop symptoms before 20 years of age. We estimate, based upon our review of historic published studies and reports, and an interpolation
of data on the incidence of Pediatric Crohn’s disease, that Pediatric Crohn's disease is a subpopulation of approximately
80,000 patients in the U.S. with a comparable number in Europe. Crohn’s disease tends to be both severe and extensive in
the pediatric population and approximately 40% of pediatric Crohn’s patients have involvement of their upper gastrointestinal
tract.
Crohn's disease presents
special challenges for children and teens. In addition to bothersome and often painful symptoms, the disease can stunt growth,
delay puberty, and weaken bones. Crohn's disease symptoms may sometimes prevent a child from participating in enjoyable activities.
The emotional and psychological issues of living with a chronic disease can be especially difficult for young people.
SGX201 –for Preventing Acute Radiation
Enteritis
SGX201 is a delayed-release
formulation of BDP specifically designed for oral use. In 2012, we completed a Phase 1/2 clinical trial testing SGX201 in prevention
of acute radiation enteritis. Patients with rectal cancer scheduled to undergo concurrent radiation and chemotherapy prior to surgery
were randomized to one of four dose groups. The objectives of the study were to evaluate the safety and maximal tolerated dose
of escalating doses of SGX201, as well as the preliminary efficacy of SGX201 for prevention of signs and symptoms of acute radiation
enteritis. The study demonstrated that oral administration of SGX201 was safe and well tolerated across all four dose groups. There
was also evidence of a potential dose response with respect to diarrhea, nausea and vomiting and the assessment of enteritis according
to National Cancer Institute Common Terminology Criteria for Adverse Events for selected gastrointestinal events. In addition,
the incidence of diarrhea was lower than that seen in recent published historical control data in this patient population. This
program was supported in part by a $500,000 two-year Small Business Innovation and Research (“SBIR”) grant awarded
by the National Institutes of Health (“NIH”). We are currently working with our Radiation Enteritis medical advisory
board in pursuing additional funding from the NIH to support the clinical development program.
We have received Fast Track
designation from the FDA for SGX201 for acute radiation enteritis.
We estimate the potential
worldwide market for oral BDP is in excess of $500 million for all applications, including the treatment of acute radiation enteritis.
This potential market information is a forward-looking statement, and investors are urged not to place undue reliance on this statement.
While we have determined this potential market size based on assumptions that we believe are reasonable, there are a number of
factors that could cause our expectations to change or not be realized. See “Risk Factors” and “Cautionary Note
Regarding Forward-Looking Statements – Industry Data and Market Information.”
Acute Radiation Enteritis
External radiation therapy
is used to treat most types of cancer, including cancer of the bladder, uterine, cervix, rectum, prostate, and vagina. During delivery
of treatment, some level of radiation will also be delivered to healthy tissue, including the bowel, leading to acute and chronic
toxicities. The large and small bowels are very sensitive to radiation and the larger the dose of radiation the greater the damage
to normal bowel tissue. Radiation enteritis is a condition in which the lining of the bowel becomes swollen and inflamed during
or after radiation therapy to the abdomen, pelvis, or rectum. Most tumors in the abdomen and pelvis need large doses, and almost
all patients receiving radiation to the abdomen, pelvis, or rectum will show signs of acute enteritis.
Patients with acute enteritis
may have nausea, vomiting, abdominal pain and bleeding, among other symptoms. Some patients may develop dehydration and require
hospitalization. With diarrhea, the gastrointestinal tract does not function normally, and nutrients such as fat, lactose, bile
salts, and vitamin B 12 are not well absorbed.
Symptoms will usually resolve
within two to six weeks after therapy has ceased. Radiation enteritis is often not a self-limited illness, as over 80% of patients
who receive abdominal radiation therapy complain of a persistent change in bowel habits. Moreover, acute radiation injury increases
the risk of development of chronic radiation enteropathy, and overall 5% to 15% of the patients who receive abdominal or pelvic
irradiation will develop chronic radiation enteritis.
We estimate, based upon
our review of historic published studies and reports, and an interpolation of data on the treatment courses and incidence of cancers
occurring in the abdominal and pelvic regions, there to be over 100,000 patients annually in the U.S., with a comparable number
in Europe, who receive abdominal or pelvic external beam radiation treatment for cancer, and these patients are at risk of developing
acute and chronic radiation enteritis.
Vaccines/BioDefense Overview
ThermoVax™ – Thermostability Technology
Our thermostability technology,
ThermoVax™, is a novel method of rendering aluminum salt, (known colloquially as Alum), adjuvanted vaccines stable at elevated
temperatures. Alum is the most widely employed adjuvant technology in the vaccine industry. The value of ThermoVax™ lies
in its potential ability to eliminate the need for cold-chain production, transportation, and storage for Alum adjuvanted vaccines.
This would relieve companies of the high costs of producing and maintaining vaccines under refrigerated conditions. Based on historical
reports from the World Health Organization and other scientific reports, we believe that a meaningful proportion of vaccine doses
globally are wasted due to excursions from required cold chain temperature ranges. This is due to the fact that most Alum adjuvanted
vaccines need to be maintained at between 2 and 8 degrees Celsius (“C”) and even brief excursions from this temperature
range (especially below freezing) usually necessitates the destruction of the product or the initiation of costly stability programs
specific for the vaccine lots in question. We believe that the savings realized from the elimination of cold chain costs and related
product losses would significantly increase the profitability of vaccine products. We believe that elimination of the cold chain
could further facilitate the use of these vaccines in the lesser developed parts of the world. ThermoVax™ has the potential
to facilitate easier storage and distribution of strategic national stockpile vaccines in emergency settings.
ThermoVax™ development
was supported pursuant to our $9.4 million NIAID grant enabling development of thermo-stable ricin (RiVax™) and anthrax (VeloThrax™)
vaccines. Proof-of-concept preclinical studies with ThermoVax™ indicate that it is able to produce stable vaccine formulations
using adjuvants, protein immunogens, and other components that ordinarily would not withstand long temperature variations exceeding
customary refrigerated storage conditions. These studies were conducted with our aluminum-adjuvanted ricin toxin vaccine, RiVax™
and our aluminum-adjuvanted anthrax vaccine, VeloThrax™. Each vaccine was manufactured, under precise lyophilization conditions
using excipients that aid in maintaining native protein structure of the key antigen. When RiVax™ was kept at 40 degrees
C (104 degrees Fahrenheit) for up to one year, all of the animals vaccinated with the lyophilized RiVax™ vaccine developed
potent and high titer neutralizing antibodies. In contrast, animals that were vaccinated with the liquid RiVax™ vaccine kept
at 40 degrees C did not develop neutralizing antibodies and were not protected against ricin exposure. The ricin A chain is extremely
sensitive to temperature and rapidly loses the ability to induce neutralizing antibodies when exposed to temperatures higher than
8 degrees C. When VeloThrax™ was kept for up to 16 weeks at 70 degrees C, it was able to develop a potent antibody response,
unlike the liquid formulation kept at the same temperature. Moreover, we have also demonstrated the compatibility of our thermostabilization
technology with other secondary adjuvants such as TLR-4 agonists.
Additionally, the University
of Colorado conducted a study that demonstrated a heat stable vaccine formulation of a human papillomavirus (HPV) vaccine. The
work was conducted by Drs. Randolph and Garcea and demonstrated the successful conversion of a commercial virus-like particle (VLP)
based vaccine requiring cold-chain storage to a subunit, alum-adjuvanted, vaccine which is stable at ambient temperatures. This
work, funded by a University of Colorado Seed grant and the Specialized Program of Research Excellence (SPORE) in cervical cancer,
is the first demonstration of the utility of ThermoVax™ technology for the development of a subunit based commercial vaccine.
The HPV vaccine formulation was found to be stable for at least 12 weeks at 50 degrees C. In the study, mice immunized with the
ThermoVaxTM-stabilized HPV subunit vaccine were also found to achieve immune responses similar to the commercial HPV
vaccine, Cervarix®, as measured by either total antibody levels or neutralizing antibody levels. Moreover, whereas
the immune responses to Cervarix® were reduced after storage for 12 weeks at 50 degrees C, the ThermoVax™
formulated vaccine retained its efficacy. The results were published online in the European Journal of Pharmaceutics and Biopharmaceutics
(see http://www.sciencedirect.com/science/article/pii/S0939641115002416).
We intend to seek out potential
partnerships with companies marketing FDA/ex-U.S. health authority approved Alum adjuvanted vaccines that are interested in eliminating
the need for cold chain for their products. We believe that ThermoVax™ also will enable us to expand our vaccine development
expertise beyond biodefense into the infectious disease space and has the potential to allow for the development of multivalent
vaccines (e.g., combination ricin-anthrax vaccine).
RiVax™ – Ricin Toxin
Vaccine
RiVax™ is our proprietary
vaccine candidate being developed to protect against exposure to ricin toxin, and if approved would be the first ricin vaccine. The
immunogen in RiVax™ induces a protective immune response in animal models of ricin exposure and functionally active antibodies
in humans. The immunogen consists of a genetically inactivated subunit ricin A chain that is enzymatically inactive and lacks residual
toxicity of the holotoxin. RiVax™ has demonstrated statistically significant (p < 0.0001) preclinical survival results
in a lethal aerosol exposure non-human primate model (Roy et al, 2015, Thermostable ricin vaccine protects rhesus macaques against
aerosolized ricin: Epitope-specific neutralizing antibodies correlate with protection, PNAS Epub ahead of print March 9, 2015),
and has also been shown to be well tolerated and immunogenic in two Phase 1 clinical trials in healthy volunteers. Results of the
first Phase 1 human trial of RiVax™ established that the immunogen was safe and induced antibodies that we believe may protect
humans from ricin exposure. The antibodies generated from vaccination, concentrated and purified, were capable of conferring immunity
passively to recipient animals, indicating that the vaccine was capable of inducing functionally active antibodies in humans. The
outcome of this study was published in the Proceedings of the National Academy of Sciences (Vitetta et al., 2006, A Pilot Clinical
Trial of a Recombinant Ricin Vaccine in Normal Humans, PNAS, 103:2268-2273). The second trial completed in September 2012, sponsored
by University of Texas Southwestern Medical Center (“UTSW”), evaluated a more potent formulation of RiVax™ that
contained an aluminum adjuvant (Alum). The results of the Phase 1B study indicated that Alum adjuvanted RiVax™ was safe and
well tolerated, and induced greater ricin neutralizing antibody levels in humans than adjuvant-free RiVax™. The outcomes
of this second study were published in the Clinical and Vaccine Immunology (Vitetta et al., 2012, Recombinant Ricin Vaccine
Phase 1B Clinical Trial, Clin. Vaccine Immunol. 10:1697-9). We have adapted the original manufacturing process for the immunogen
contained in RiVax™ for large scale manufacturing and are further establishing correlates of the human immune response in
non-human primates.
The development of RiVax™
has been sponsored through a series of overlapping challenge grants, UC1, and cooperative grants, U01, from the NIH, granted to
Soligenix and to UTSW where the vaccine originated. The second clinical trial was supported by a grant from the FDA’s Office
of Orphan Products to UTSW. To date, we and UTSW have collectively received approximately $25 million in grant funding from the
NIH for the development of RiVax™. In September 2014, we entered into a contract with the NIH for the development of RiVax™
that would provide up to an additional $24.7 million of funding in the aggregate if options to extend the contract are exercised
by the NIH.
RiVax™ has been granted
orphan drug designation by the FDA for the prevention of ricin intoxication.
Assuming development efforts
are successful for RiVax™, we believe potential government procurement contract(s) could reach $200 million. This potential
procurement contract information is a forward-looking statement, and investors are urged not to place undue reliance on this statement.
While we have determined this potential procurement contract value based on assumptions that we believe are reasonable, there are
a number of factors that could cause our expectations to change or not be realized. See “Risk Factors” and “Cautionary
Note Regarding Forward-Looking Statements – Industry Data and Market Information.”
Ricin Toxin
Ricin toxin can be cheaply
and easily produced, is stable over long periods of time, is toxic by several routes of exposure and thus has the potential to
be used as a biological weapon against military and/or civilian targets. As a bioterrorism agent, ricin could be disseminated as
an aerosol, by injection, or as a food supply contaminant. The potential use of ricin toxin as a biological weapon of mass destruction
has been highlighted in a Federal Bureau of Investigations Bioterror report released in November 2007 titled Terrorism 2002-2005,
which states that “Ricin and the bacterial agent anthrax are emerging as the most prevalent agents involved in WMD investigations”
(http://www.fbi.gov/stats-services/publications/terrorism-2002-2005/terror02_05.pdf). In recent years, Al Qaeda in the Arabian
Peninsula has threatened the use of ricin toxin to poison food and water supplies and in connection with explosive devices. Domestically,
the threat from ricin remains a concern for security agencies. As recently as April 2013, letters addressed to the President, a
U.S. Senator and a judge tested positive for ricin.
The Centers for Disease
Control has classified ricin toxin as a Category B biological agent. Ricin works by first binding to glycoproteins found on the
exterior of a cell, and then entering the cell and inhibiting protein synthesis leading to cell death. Once exposed to ricin toxin,
there is no effective therapy available to reverse the course of the toxin. The recent ricin threat to government officials has
heightened the awareness of this toxic threat. Currently, there is no FDA approved ricin vaccine, nor is there a known antidote
for ricin toxin exposure.
VeloThrax™ – Anthrax Vaccine
VeloThrax™ is our
proprietary vaccine candidate based on a recombinant protective antigen (“rPA”) derivative intended for use against
anthrax. We have entered into an exclusive license option with Harvard College to license VeloThrax™ (also known as DNI for
dominant negative inhibitor) for a vaccine directed at the prevention of anthrax infection of humans. VeloThrax™ is a translocation-deficient
mutant of a protective antigen with double mutations of K397D and D425K that impede the conformational changes necessary for endosomal
membrane translocation into the cell cytoplasm. In the absence of that protective antigen translocation step, anthrax toxin trafficking
and function cease. We believe that VeloThrax™ is a more immunogenic candidate than native rPA. This apparent increase in
immunogenicity suggests that the DNI rPA is processed and presented to the immune system more efficiently by cellular antigen processing
pathways, which is consistent with known properties of the molecule.
DNI versions of rPA such
as VeloThrax™ are also capable of inducing antibodies that neutralize the activity of the anthrax toxin complex. Unlike fully-functional
rPA, VeloThrax™ might be given to a patient post-exposure without risk of enhancing intoxication during an infection, although
clinical tests involving intravenous administration of potentially therapeutic levels of DNI rPA resulted in serious adverse events
and so further development of this product as a therapeutic biological for blocking the effects of infection by B. anthracis was
discontinued. Our studies of VeloThrax™ will be at a dose 1,000 times lower than the dose previously tested for an intramuscular
or intradermal vaccine.
We believe that VeloThrax™’s
greater immunogenicity could lead to a vaccine that can be administered in the fewest possible doses to induce the highest level
of toxin neutralizing antibodies. Utilizing ThermoVax™, we believe that we will be able to develop VeloThrax™ into
a vaccine with an improved stability profile, an issue that has proven challenging in the development of other anthrax vaccines.
Extended stability at ambient temperatures would be a significant improvement for stockpiled vaccines and one which is not expected
from conventional vaccines. Assuming long-term stability can be met, VeloThrax™ could be stockpiled for general prophylactic
as well as a post exposure use.
The overall objective of
the VeloThrax™ program is to rapidly and efficiently develop a next generation anthrax vaccine which combines a well-established,
safe and relatively low risk vaccine development and dosing approach with targeted, proven innovative strategies. We expect that
VeloThrax™ will combine a stable, readily manufactured mutant rPA subunit antigen with next generation, clinically compatible
adjuvants which have been demonstrated to enhance potency and reduce the time and number of vaccine doses required to achieve protective
titer using a variety of vaccine antigens. We believe that VeloThrax™ has the potential to provide the Public Health Emergency
Medical Countermeasures Enterprise (“PHEMCE”) and the Department of Defense (“DoD”) with a safe and stable
alternative to the existing licensed anthrax vaccine product. We also intend to adapt newly developed glassification technology
(initially developed under an ongoing NIAID grant to stabilize exceptionally unstable ricin toxin/adjuvant formulations) to enable
a thermostable, dried, single vial, pre-formulated adjuvanted rPA vaccine which is suitable for both long term storage and field
use without typical cold chain constraints.
Assuming development efforts
are successful for VeloThrax™, we believe potential government procurement contract(s) could reach $500 million. This potential
procurement contract information is a forward-looking statement, and investors are urged not to place undue reliance on this statement.
While we have determined this potential procurement contract value based on assumptions that we believe are reasonable, there are
a number of factors that could cause our expectations to change or not be realized. See “Risk Factors” and “Cautionary
Note Regarding Forward-Looking Statements – Industry Data and Market Information.”
Anthrax
Anthrax is
an acute infectious disease that is easily transmitted to humans by environmentally durable spores that are produced by Bacillus
anthracis. Because the spores are robust and contagious, anthrax is considered a Category A bioterror threat. Anthrax infection
can occur in three forms: cutaneous (skin), inhalation, and gastrointestinal. Inhaled spores can cause a rapidly progressing form
of anthrax since the spores are transported to lymph nodes near the lungs where they germinate, releasing vegetative bacteria
into the bloodstream. Bacteria synthesize a complex series of toxin components that make up anthrax toxin, resulting in overwhelming
toxemia that causes shock and organ failure. Treatment of anthrax involves long-term antibiotic therapy, since ungerminated spores
can lie dormant in the lungs for up to 60 days. Only a few inhaled spores can cause inhalational anthrax. Once the toxin has entered
the bloodstream, antibiotics are ineffective, and only toxin-specific therapy is effective. Passively transferred antibodies can
neutralize anthrax toxins and can be used post-exposure in conjunction with antibiotics. Because of the long residence time of
spores in the lung, it is possible to vaccinate post-exposure, but the onset of neutralizing antibodies must occur during the
period of antibiotic therapy.
OrbeShield™ –for Treating
GI ARS
OrbeShield™ is an
oral immediate and delayed release formulation of the topically active corticosteroid BDP and is being developed for the treatment
of GI ARS. Corticosteroids are a widely used class of anti-inflammatory drugs. BDP is a corticosteroid with predominantly topical
activity that is approved for use in asthma, psoriasis and allergic rhinitis.
OrbeShield™ has demonstrated
positive preclinical results in a canine GI ARS model which indicate that dogs treated with OrbeShield™ demonstrated
statistically significant (p=0.04) improvement in survival with dosing at either two hours or 24 hours after exposure to lethal
doses of total body irradiation (“TBI”) when compared to control dogs. OrbeShield™ appears to significantly mitigate
the damage to the GI epithelium caused by exposure to high doses of radiation using a well-established canine model of GI ARS.
The GI tract is highly
sensitive to ionizing radiation and the destruction of epithelial tissue is one of the first effects of radiation exposure. The
rapid loss of epithelial cells leads to inflammation and infection that are often the primary cause of death in acute radiation
injury. This concept of GI damage also applies to the clinical setting of oncology, where high doses of radiation cannot be administered
effectively to the abdomen because radiation is very toxic to the intestines. We are seeking to treat the same type of toxicity
in our acute radiation enteritis clinical program with SGX201. As a result, we believe that OrbeShield™ has the potential
to be a “dual use” compound, a desirable characteristic which is a specific priority of BARDA for ARS and other medical
countermeasure indications. The FDA has cleared the IND application for OrbeShield™ for the mitigation of morbidity and mortality
associated with GI ARS.
In September 2013, we received
two government contracts from BARDA and NIAID for the advanced preclinical and manufacturing development of OrbeShield™ leading
to FDA approval to treat GI ARS. The BARDA contract contains a two year base period with two contract options, exercisable by BARDA,
for a total of five years and up to $26.3 million. The NIAID contract consists of a one year base period and two contract options,
exercisable by NIAID, for a total of three years and up to $6.4 million. Previously, development of OrbeShield™ had been
largely supported by a $1 million NIH grant to Soligenix’s academic partner, the Fred Hutchinson Cancer Research Center.
In July 2012, we received an SBIR grant from NIAID of approximately $600,000 to support further preclinical development of OrbeShield™
for the treatment of acute GI ARS. The FDA has given OrbeShield™ orphan drug designation and Fast Track designation for the
prevention of death following a potentially lethal dose of total body irradiation during or after a radiation disaster.
Assuming development efforts
are successful for OrbeShield™, we believe potential government procurement contracts could reach as much as $450 million.
This potential procurement contract information is a forward-looking statement, and investors are urged not to place undue reliance
on this statement. While we have determined this potential procurement contract value based on assumptions that we believe are
reasonable, there are a number of factors that could cause our expectations to change or not be realized. See “Risk Factors”
and “Cautionary Note Regarding Forward-Looking Statements – Industry Data and Market Information.”
GI ARS
ARS occurs after toxic
radiation exposure and involves several organ systems, notably the bone marrow, the GI tract and, later, the lungs. In the event
of a nuclear disaster or terrorist detonation of a nuclear bomb, casualties exposed to greater than 2 grays (“Gy”)
of absorbed radiation are at high risk for development of clinically significant ARS. Exposure to high doses of radiation exceeding
10-12 Gy causes acute GI injury which can result in death. The GI tract is highly sensitive due to the continuous need for crypt
stem cells and production of mucosal epithelium. The extent of injury to the bone marrow and the GI tract are the principal determinants
of survival after exposure to TBI. Although the hematopoietic syndrome can be rescued by bone marrow transplantation or growth
factor administration, there is no established treatment or preventive measure for the GI damage that occurs after high-dose radiation.
As a result, we believe there is an urgent medical need for specific medical counter measures against the lethal pathophysiological
manifestations of radiation-induced GI injury.
SGX943/SGX101– for Treating Melioidosis
SGX943 uses the same active
ingredient as SGX94 and is being developed in preclinical studies as a potential treatment for melioidosis. Because SGX943 directly
targets the innate immune system (and does not attempt to kill the bacteria directly), we believe it is particularly relevant for
antibiotic-resistant bacteria. The bacteria which causes melioidosis, Burkholderia pseudomallei, is known to be resistant
to most antibiotics and to require prolonged treatment with the few antibiotics that do work. In February 2014, we were awarded
a one-year NIAID SBIR grant award of approximately $300,000 to further evaluate SGX943 as a potential treatment for melioidosis.
Preclinical results to date have demonstrated that SGX943 treatment, in combination with standard of care antibiotics such as doxycycline,
can statistically significantly enhance survival in a lethal murine pneumonic melioidosis model (p< 0.001).
SGX101 is a human monoclonal
antibody therapy being developed in preclinical studies as a potential treatment of melioidosis using Intrexon’s advanced
human antibody discovery, isolation, and production technologies. As data becomes available from this work, we intend to pursue
grant funding to support further development of this product candidate.
Melioidosis
Melioidosis is a potentially
fatal infection caused by the Gram-negative bacillus, Burkholderia pseudomallei (“Bp”). Highly resistant to
many antibiotics, Bp can cause an acute disease characterized by a fulminant pneumonia and a chronic condition that can recrudesce.
There is no preventive vaccine or effective immunotherapy for melioidosis. We believe that there is an unmet medical need for improved
prevention and therapy.
Bp infection (melioidosis)
is a major public health concern in the endemic regions of Southeast Asia and Northern Australia. In Northeast Thailand, which
has a high incidence of melioidosis, the mortality rate associated with Bp infection is over 40 percent, making it the third most
common cause of death from infectious disease in that region after HIV/AIDS and tuberculosis. Bp activity is seen in Southeast
Asia, South America, Africa, the Middle East, India, and Australia. The highest pockets of disease activity occur in Northern Australia
and Northeast Thailand with increasing recognition of disease activity in coastal regions of India.
Beyond its public health
significance, Bp and the closely-related Burkholderia mallei (“Bm”) are considered possible biological warfare
agents by the DHHS because of the potential for widespread dissemination through aerosol. Bp like its relative Bm, the cause of
Glanders, was studied by the U.S. as a potential biological warfare agent, but was never weaponized. It has been reported that
the Soviet Union was also experimenting with Bp as a biological warfare agent. Both Bp and Bm have been designated high priority
threats by the DHHS in its PHEMCE Strategy released in 2012 and are classified as Category B Priority Pathogens by NIAID.
The Drug Approval Process
The FDA and comparable
regulatory agencies in state, local and foreign jurisdictions impose substantial requirements on the clinical development, manufacture
and marketing of new drug and biologic products. The FDA, through regulations that implement the Federal Food, Drug, and Cosmetic
Act, as amended, or FDCA, and other laws and comparable regulations for other agencies, regulate research and development activities
and the testing, manufacture, labeling, storage, shipping, approval, recordkeeping, advertising, promotion, sale, export, import
and distribution of such products. The regulatory approval process is generally lengthy, expensive and uncertain. Failure to comply
with applicable FDA and other regulatory requirements can result in sanctions being imposed on us or the manufacturers of our products,
including holds on clinical research, civil or criminal fines or other penalties, product recalls, or seizures, or total or partial
suspension of production or injunctions, refusals to permit products to be imported into or exported out of the United States,
refusals of the FDA to grant approval of drugs or to allow us to enter into government supply contracts, withdrawals of previously
approved marketing applications and criminal prosecutions.
Before human clinical testing
in the U.S. of a new drug compound or biological product can commence, an Investigational New Drug, or IND, application is required
to be submitted to the FDA. The IND application includes results of pre-clinical animal studies evaluating the safety and efficacy
of the drug and a detailed description of the clinical investigations to be undertaken.
Clinical trials are normally
done in three phases, although the phases may overlap. Phase 1 trials are smaller trials concerned primarily with metabolism and
pharmacologic actions of the drug and with the safety of the product. Phase 2 trials are designed primarily to demonstrate effectiveness
and safety in treating the disease or condition for which the product is indicated. These trials typically explore various doses
and regimens. Phase 3 trials are expanded clinical trials intended to gather additional information on safety and effectiveness
needed to clarify the product’s benefit-risk relationship and generate information for proper labeling of the drug, among
other things. The FDA receives reports on the progress of each phase of clinical testing and may require the modification, suspension
or termination of clinical trials if an unwarranted risk is presented to patients. When data is required from long-term use of
a drug following its approval and initial marketing, the FDA can require Phase 4, or post-marketing, studies to be conducted.
With certain exceptions,
once successful clinical testing is completed, the sponsor can submit a New Drug Application, or NDA, for approval of a drug, or
a Biologic License Application, or BLA, for biologics such as vaccines, which will be reviewed, and if successful, approved by
the FDA, allowing the product to be marketed. The process of completing clinical trials for a new drug is likely to take a number
of years and require the expenditure of substantial resources. Furthermore, the FDA or any foreign health authority may not grant
an approval on a timely basis, if at all. The FDA may deny the approval of an NDA or BLA, in its sole discretion, if it determines
that its regulatory criteria have not been satisfied or may require additional testing or information. Among the conditions for
marketing approval, is the requirement that the prospective manufacturer’s quality control and manufacturing procedures conform
to good manufacturing practice regulations. In complying with standards contained in these regulations, manufacturers must continue
to expend time, money and effort in the area of production, quality control and quality assurance to ensure full technical compliance.
Manufacturing facilities, both foreign and domestic, also are subject to inspections by, or under the authority of, the FDA and
by other federal, state, local or foreign agencies.
Even after initial FDA
or foreign health authority approval has been obtained, further studies, including Phase 4 post-marketing studies, may be required
to provide additional data on safety and will be required to gain approval for the marketing of a product as a treatment for clinical
indications other than those for which the product was initially tested. For certain drugs intended to treat serious, life-threatening
conditions that show great promise in earlier testing, the FDA can also grant conditional approval. However, drug developers are
required to study the drug further and verify clinical benefit as part of the conditional approval provision, and the FDA can revoke
approval if later testing does not reproduce previous findings. The FDA may also condition approval of a product on the sponsor
agreeing to certain mitigation strategies that can limit the unfettered marketing of a drug. Also, the FDA or foreign regulatory
authority will require post-marketing reporting to monitor the side effects of the drug. Results of post-marketing programs may
limit or expand the further marketing of the product. Further, if there are any modifications to the drug, including any change
in indication, manufacturing process, labeling or manufacturing facility, an application seeking approval of such changes will
likely be required to be submitted to the FDA or foreign regulatory authority.
In the U.S., the FDCA,
the Public Health Service Act, the Federal Trade Commission Act, and other federal and state statutes and regulations that govern
or influence the research, testing, manufacture, safety, labeling, storage, record keeping, approval, advertising and promotion
of drug, biological, medical device and food products. Noncompliance with applicable requirements can result in, among other things,
fines, recall or seizure of products, refusal to permit products to be imported into the U.S., refusal of the government to approve
product approval applications or to allow the Company to enter into government supply contracts, withdrawal of previously approved
applications and criminal prosecution. The FDA may also assess civil penalties for violations of the FDCA involving medical devices.
For biodefense development,
such as with RiVax™ and OrbeShield™, the FDA has instituted policies that are expected to result in shorter pathways
to market. This potentially includes approval for commercial use utilizing the results of animal efficacy trials, rather than efficacy
trials in humans. However, the Company will still have to establish that the vaccine and countermeasures it is developing are safe
in humans at doses that are correlated with the beneficial effect in animals. Such clinical trials will also have to be completed
in distinct populations that are subject to the countermeasures; for instance, the very young and the very old, and in pregnant
women, if the countermeasure is to be licensed for civilian use. Other agencies will have an influence over the benefit-risk scenarios
for deploying the countermeasures and in establishing the number of doses utilized in the Strategic National Stockpile. We may
not be able to sufficiently demonstrate the animal correlation to the satisfaction of the FDA, as these correlates are difficult
to establish and are often unclear. Invocation of the animal rule may raise issues of confidence in the model systems even if the
models have been validated. For many of the biological threats, the animal models are not available and the Company may have to
develop the animal models, a time-consuming research effort. There are few historical precedents, or recent precedents, for the
development of new countermeasure for bioterrorism agents. Despite the animal rule, the FDA may require large clinical trials to
establish safety and immunogenicity before licensure and it may require safety and immunogenicity trials in additional populations.
Approval of biodefense products may be subject to post-marketing studies, and could be restricted in use in only certain populations.
Vaccines are approved under
the BLA process that exists under the Public Health Service Act. In addition to the greater technical challenges associated with
developing biologics, the potential for generic competition is lower for a BLA product than a small molecule product subject to
an NDA under the Federal Food, Drug and Cosmetic Act. Under the Patient Protection and Affordable Care Act enacted in 2010, a “generic”
version of a biologic is known as a biosimilar and the barriers to entry – whether legal, scientific, or logistical –
for a biosimilar version of a biologic approved under a BLA are higher. Indeed, almost three years after the enactment of the Patient
Protection and Affordable Care Act, no biosimilar application has even been filed with the FDA.
Orphan Drug Designation
Under the Orphan Drug Act,
the FDA may grant orphan drug designation to drugs or biologics intended to treat a rare disease or condition—generally a
disease or condition that affects fewer than 200,000 individuals in the United States. Orphan drug designation must be requested
before submitting an NDA or BLA. After the FDA grants orphan drug designation, the generic identity of the drug or biologic and
its potential orphan use are disclosed publicly by the FDA. Orphan drug designation does not convey any advantage in, or shorten
the duration of, the regulatory review and approval process. The first NDA or BLA applicant to receive FDA approval for a particular
active ingredient to treat a particular disease with FDA orphan drug designation is entitled to a seven-year exclusive marketing
period in the United States for that product, for that indication. During the seven-year exclusivity period, the FDA may not approve
any other applications to market the same drug or biologic for the same disease, except in limited circumstances, such as a showing
of clinical superiority to the product with orphan drug exclusivity. Orphan drug exclusivity does not prevent the FDA from approving
a different drug or biologic for the same disease or condition, or the same drug or biologic for a different disease or condition.
Among the other benefits of orphan drug designation are tax credits for certain research and a waiver of the NDA or BLA application
user fee.
Fast Track Designation and Accelerated
Approval
The FDA is required to
facilitate the development, and expedite the review, of drugs or biologics that are intended for the treatment of a serious or
life-threatening disease or condition for which there is no effective treatment and which demonstrate the potential to address
unmet medical needs for the condition. Under the fast track program, the sponsor of a new drug or biologic candidate may request
that the FDA designate the candidate for a specific indication as a fast track drug or biologic concurrent with, or after, the
filing of the IND for the candidate. The FDA must determine if the drug or biologic candidate qualifies for fast track designation
within 60 days of receipt of the sponsor's request. Unique to a fast track product, the FDA may initiate review of sections of
a fast track product's NDA or BLA before the application is complete. This rolling review is available if the applicant provides,
and the FDA approves, a schedule for the submission of the remaining information and the applicant pays applicable user fees. However,
the FDA's time period goal for reviewing an application does not begin until the last section of the NDA or BLA is submitted. Additionally,
the fast track designation may be withdrawn by the FDA if the FDA believes that the designation is no longer supported by data
emerging in the clinical trial process.
Any product submitted to
the FDA for marketing, including under a fast track program, may be eligible for other types of FDA programs intended to expedite
development and review, such as accelerated approval. Drug or biological products studied for their safety and effectiveness in
treating serious or life-threatening illnesses and that provide meaningful therapeutic benefit over existing treatments may receive
accelerated approval, which means the FDA may approve the product based upon a surrogate endpoint that is reasonably likely to
predict clinical benefit, or on a clinical endpoint that can be measured earlier than irreversible morbidity or mortality, that
is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account
the severity, rarity, or prevalence of the condition and the availability or lack of alternative treatments.
In clinical trials, a surrogate
endpoint is a measurement of laboratory or clinical signs of a disease or condition that substitutes for a direct measurement of
how a patient feels, functions, or survives. Surrogate endpoints can often be measured more easily or more rapidly than clinical
endpoints. A drug or biologic candidate approved on this basis is subject to rigorous post-marketing compliance requirements, including
the completion of Phase 4 or post-approval clinical trials to confirm the effect on the clinical endpoint. Failure to conduct required
post-approval studies, or confirm a clinical benefit during post-marketing studies, will allow the FDA to withdraw the drug or
biologic from the market on an expedited basis. All promotional materials for drug candidates approved under accelerated regulations
are subject to prior review by the FDA.
Pediatric Information
Under the Pediatric Research
Equity Act, or PREA, NDAs or BLAs or supplements to NDAs or BLAs must contain data to assess the safety and effectiveness of the
drug for the claimed indications in all relevant pediatric subpopulations and to support dosing and administration for each pediatric
subpopulation for which the drug is safe and effective. The FDA may grant full or partial waivers, or deferrals, for submission
of data. Unless otherwise required by regulation, PREA does not apply to any drug for an indication for which orphan designation
has been granted.
False Claims
Laws
The federal False Claims
Act prohibits, among other things, any person or entity from knowingly presenting, or causing to be presented, a false claim for
payment to, or approval by, the federal government or knowingly making, using, or causing to be made or used a false record or
statement material to a false or fraudulent claim to the federal government. As a result of a modification made by the Fraud Enforcement
and Recovery Act of 2009, a claim includes “any request or demand” for money or property presented to the U.S. government.
Anti-Kickback
Laws
The federal Anti-Kickback
Statute prohibits, among other things, any person or entity, from knowingly and willfully offering, paying, soliciting or receiving
any remuneration, directly or indirectly, overtly or covertly, in cash or in kind, to induce or in return for purchasing, leasing,
ordering or arranging for the purchase, lease or order of any item or service reimbursable under Medicare, Medicaid or other federal
healthcare programs. The term remuneration has been interpreted broadly to include anything of value. The Anti-Kickback Statute
has been interpreted to apply to arrangements between pharmaceutical manufacturers on one hand and prescribers, purchasers, and
formulary managers on the other.
United States
Healthcare Reform
Federal Physician Payments
Sunshine Act and its implementing regulations require that certain manufacturers of drugs, devices, biological and medical supplies
for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program (with certain exceptions)
to report information related to certain payments or other transfers of value made or distributed to physicians and teaching hospitals,
or to entities or individuals at the request of, or designated on behalf of, the physicians and teaching hospitals and to report
annually certain ownership and investment interests held by physicians and their immediate family members.
In addition, we may be
subject to data privacy and security regulation by both the federal government and the states in which we conduct our business.
The Health Insurance Portability and Accountability Act, or HIPAA, as amended by the Health Information Technology for Economic
and Clinical Health Act, or HITECH, and its implementing regulations, imposes certain requirements relating to the privacy, security
and transmission of individually identifiable health information. Among other things, HITECH makes HIPAA’s privacy and security
standards directly applicable to “business associates”—independent contractors or agents of covered entities
that receive or obtain protected health information in connection with providing a service on behalf of a covered entity. HITECH
also created four new tiers of civil monetary penalties, amended HIPAA to make civil and criminal penalties directly applicable
to business associates and possibly other persons, and gave state attorneys general new authority to file civil actions for damages
or injunctions in federal courts to enforce the federal HIPAA laws and seek attorneys’ fees and costs associated with pursuing
federal civil actions. In addition, state laws govern the privacy and security of health information in certain circumstances,
many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts.
Third-Party Suppliers and Manufacturers
Drug substance and drug
product manufacturing is outsourced to qualified suppliers. We do not have manufacturing capabilities/infrastructure and do not
intend to develop the capacity to manufacture drug products substances. We have agreements with third-party manufacturers to supply
bulk drug substances for our product candidates and with third parties to formulate, package and distribute our product candidates.
Our employees include professionals with expertise in pharmaceutical manufacturing development, quality assurance and third party
supplier management who oversee work conducted by third-party companies. We believe that we have on hand or can easily obtain sufficient
amounts of product candidates to complete our currently contemplated clinical trials. All of the drug substances used in our product
candidates currently are manufactured by single suppliers. While we have not experienced any supply disruptions, the number of
manufacturers of the drug substances is limited. In the event it is necessary or advisable to acquire supplies from alternative
suppliers, assuming commercially reasonable terms could be reached, the challenge would be the efficient transfer of technology
and know-how from current manufactures to the new supplier. Formulation and distribution of our finished product candidates also
currently are conducted by single suppliers but we believe that alternative sources for these services are readily available on
commercially reasonable terms, subject to the efficient transfer of technology and know-how from current suppliers to the new supplier.
All of the current agreements
for the supply bulk drug substances for our product candidates and for the formulation or distribution of our product candidates
relate solely to the development (including preclinical and clinical) of our product candidates. Under these contracts, our product
candidates are manufactured upon our order of a specific quantity. In the event that we obtain marketing approval for a product
candidate, we will qualify secondary suppliers for all key manufacturing activities supporting the marketing application.
Marketing and Collaboration
We do not currently have
and do not intend to establish any sales and marketing capability, other than to potentially market our biodefense vaccine products
directly to government agencies. With respect to other commercialization efforts, we currently intend to seek distribution and
other collaboration arrangements for the sales and marketing of any product candidate that is approved. From time to time, we have
had and are having strategic discussions with potential collaboration partners for our biodefense vaccine product candidates, although
no assurance can be given that we will be able to enter into one or more collaboration agreements for our product candidate on
acceptable terms, if at all. We believe that both military and civilian health authorities of the U.S. and other countries will
increase their stockpiling of therapeutics and vaccines to treat and prevent diseases and conditions that could ensue following
a bioterrorism attack.
On December 20, 2012, we
re-acquired the North American and European commercial rights to oral BDP through an amendment of our collaboration and supply
agreement with Sigma-Tau Pharmaceuticals, Inc. (“Sigma-Tau”). The amendment requires us to make certain approval and
commercialization milestone payments to Sigma-Tau which could reach up to $6 million. In addition, the Company has agreed to pay
Sigma-Tau: (a) a royalty amount equal to 3% of all net sales of oral BDP made directly by the Company, and any third-party partner
and/or their respective affiliates in the U.S., Canada, Mexico and in each country in the European Territory for the later to occur
of: (i) a period of ten years from the first commercial sale of oral BDP in each country, or (ii) the expiration of the Company’s
patents and patent applications relating to oral BDP in such country (the “Payment Period”); and (b) 15% of all up-front
payments, milestone payments and any other consideration (exclusive of equity payments) received by the Company and/or a potential
partner from the Company’s and/or potential partner’s licensees, distributors and agents for oral BDP in each relevant
country in the territory, which amount will be paid on a product-by-product and a country-by-country basis for the Payment Period.
Competition
Our competitors are pharmaceutical
and biotechnology companies, most of whom have considerably greater financial, technical, and marketing resources than we do. Universities
and other research institutions, including the U.S. Army Medical Research Institute of Infectious Diseases, also compete in the
development of treatment technologies, and we face competition from other companies to acquire rights to those technologies.
SGX301 Competition
The FDA has approved several
treatments for later stages (IIB-IV) of CTCL and/or in conditions that are unresponsive to prior treatment. Two are targeted therapies
(Targretin®-caps and Ontak®), two are histone deacetylases inhibitors (Zolina® and
Istodax®) and the remaining two are topical therapies (Valchor® and Targretin®-gel).
There are currently no FDA approved therapies for the treatment of front-line, early stage (I-IIA) CTCL; however certain topical
chemotherapies and topical, radiation, photo and other therapies which are approved for indications other than CTCL are prescribed
off-label for the treatment of early stage CTCL. These include psoralen combined with ultraviolet A (UVA) light therapy (“PUVA”);
however, PUVA treatments are usually limited to three times per week and 200 times in total due to the potentially carcinogenic
side effect. There are other drugs currently in development that may have the potential to be used in early stage (I-IIA) CTCL
– one in phase 2 (vorinostat) and others in phase 1. Vorinistat has been approved by the FDA to treat CTCL patients who have
conditions that are unresponsive to other therapies. It currently is being studied in a phase 2 trial for the treatment of all
stages of CTCL, with an estimated completion date for the phase 2 trial in September 2016.
SGX94/942 Competition
Because SGX94 uses a novel
mechanism of action in combating bacterial infections, there are no direct competitors at this time. Bacterial infections are routinely
treated with antibiotics and SGX94 treatment is anticipated to be utilized primarily where antibiotics are insufficient (e.g.,
due to antibiotic resistance) or contra-indicated (e.g., in situations where the development of antibiotic resistance is a significant
concern). Many groups are working on the antibiotic resistance problem and research into the innate immune system is intensifying,
making emerging competition likely (from companies such as Celtaxsys Inc., Innaxon Therapeutics and Innate Pharma SA).
There is currently one
drug approved for the treatment of oral mucositis in hematological cancer (palifermin). There are currently no approved drugs for
treatment of oral mucositis in cancers with solid tumors (e.g., head and neck cancer). There are several drugs in clinical development
for oral mucositis – one in Phase 3 (under development by Daewoong Pharmaceutical Co., Ltd.), three in Phase 2 (under development
by Cellceutix Corporation, BioAlliance Pharma S.A. and Alder Biopharmaceuticals Inc.) and one in Phase 1 (under development by
ActoGenix N.V.). In addition, there are medical devices approved for the treatment of oral mucositis including MuGard, GelClair,
Episil and Caphosol. These devices attempt to create a protective barrier around the oral ulceration.
Oral BDP Competition
There are a number of approved
treatments for Crohn’s disease and additional compounds are in late-stage development.
Remicade (infliximab) and
Humira (adalimumab) are currently approved for the treatment of pediatric Crohn’s disease; however, both carry significant
Black Box warnings in their labeling for increased risk of serious infection and malignancy, and therefore are approved for treatment
of moderate to severe patients. There is one other marketed biologic, Tysabri (natalizumab), in a Phase 2 study for pediatric Crohn’s.
Entocort (enteric-coated budesonide) also has completed Phase 3 trials in pediatric Crohn’s disease.
ThermoVaxTM Competition
Multiple groups and companies
are working to address the unmet need of vaccine thermostability using a variety of technologies. In addition, other organizations,
such as the Bill and Melinda Gates Foundation and PATH, have programs designed to advance technologies to address this need.
Several stabilization technologies
currently being developed involve mixing vaccine antigen +/- adjuvant with various proprietary excipients or co-factors that either
serve to stabilize the vaccine or biological product in a liquid or dried (lyophilized) form. Examples of these approaches
include the use of various plant-derived sugars and macromolecules being developed by companies such as Stabilitech Ltd. Variation
Biotechnologies, Inc. (“VBI”) is developing a lipid system (resembling liposomes) to stabilize viral antigens, including
virus-like particles (VLPs), and for potential application to a conventional influenza vaccine among others.
Other approaches involve
process variations to freeze-dry live virus vaccines. For example, PaxVax, Inc., is seeking to employ a spray drying technology
in concert with enteric coating to achieve formulations for room temperature stability of live virus vaccines using adenovirus
vectors. VBI is seeking to utilize their proprietary stabilization technology for a number of vaccines (as a co-development
service, similar to the business model being developed by Stabilitech Ltd.), whereas PaxVax is applying the technology to their
own proprietary vaccine development programs. Stabilitech uses combinations of excipients, which include glassifying sugars similar
to the ThermoVax™ technology, and variations in drying cycles during lyophilization, as does the ThermoVax™ technology.
Additionally, companies
like Pharmathene, Inc., Panacea Biotec Ltd., and Compass Biotech Inc. are developing proprietary vaccines with the application
of some form of stabilization technology.
Vaccines/BioDefense Competition
We face competition in
the area of biodefense product development from various public and private companies, universities and governmental agencies, such
as the U.S. Army, some of whom may have their own proprietary technologies which may directly compete with our technologies.
BioThrax® (Anthrax
Vaccine Adsorbed or AVA) is an anthrax vaccine marketed by Emergent BioSolutions, Inc. was developed nearly 50 years ago from a
culture filtrate derived from anthrax bacteria. Consequently, it contains a number of different proteins, some of which are
believed to potentially contribute to the adverse events that have been reported in the literature (up to 7-8% serious adverse
events) and which has prompted agencies like the Institute of Medicine to recommend adoption of newer and safer anthrax vaccines.
BioThrax® is FDA approved for the prevention of anthrax infection, but requires five doses over a period of 18 months
to achieve protective immunity.
There are a number of other
companies in preclinical and clinical development of protective antigen-based vaccines and therapeutics including Emergent BioSolutions
Inc., Pharmathene, Inc., Dynavax Technologies Corporation, Panacea Biotec Ltd., Paxvax Inc., Elusys Therapeutics, Inc., and Pfenex
Inc.
Emergent is currently developing
an anthrax immune globulin therapeutic based on plasma collected from military personnel who have been vaccinated with BioThrax®,
GlaxoSmithKline plc has been approved for an antibody to Bacillus anthracis, referred to as Abthrax™ (raxibacumab),
as a post-exposure therapeutic for anthrax infection. Elusys Therapeutics is developing a monoclonal antibody to Bacillus anthracis,
known as Anthim™, as a pre-exposure and post-exposure prophylaxis against anthrax infection, as well as an active treatment
of the disease. Pharmathene and Medarex are collaborating to develop a human antibody to anthrax, known as Valortim™. Bavarian
Nordic is developing a multivalent combination vaccine against both anthrax and smallpox.
The U.S. Army Medical Research
Institute of Infectious Diseases, the DoD’s lead laboratory for medical research to counter biological threats is also developing
a ricin vaccine candidate, RVEc™. RVEc™ has been shown to be fully protective in mice exposed to lethal doses of ricin
toxin by the aerosol route. Further studies, in both rabbits and nonhuman primates, were conducted to evaluate RVEc™’s
safety as well as its immunogenicity, with positive results observed.
In the area of radiation-protective
antidotes such as OrbeShield™, various companies, such as Cleveland Biolabs, Inc., Aeolus Pharmaceuticals, Inc., Boulder
Biotechnology, Inc., RxBio, Inc., Avaxia Biologics, Inc., Exponential Biotherapies Inc., Osiris Therapeutics, Inc., ImmuneRegen
BioSciences, Inc., Neumedicines, Inc., Cellerant Therapeutics, Inc., Onconova Therapeutics, Inc., Araim Pharmaceuticals, Inc.,
EVA Pharmaceuticals, Terapio Corporation, Cangene Corporation, Humanetics Corporation and the University of Arkansas Medical Sciences
Center are developing biopharmaceutical products that may directly compete with OrbeShield™, even though their approaches
to such treatment are different.
RxBio, Avaxia Biologics
and the University of Arkansas have programs specifically for GI ARS. RxBio’s Rx100 is a stem cell protectant designed as
a single dose (oral or injection) which has shown promise in nonhuman primate studies. Avaxia is developing an orally delivered
anti-TNF antibody as a treatment agent for exposure to radiation following a nuclear accident, attack or explosion. Pasireotide,
a drug in development by Novartis for Cushing’s disease, is being developed at the University of Arkansas to protect the
intestine by reducing pancreatic secretions that exacerbate intestinal inflammation.
Patents and Other Proprietary Rights
Our goal is to obtain,
maintain and enforce patent protection for our products, formulations, processes, methods and other proprietary technologies, preserve
our trade secrets, and operate without infringing on the proprietary rights of other parties, both in the U.S. and in other countries.
Our policy is to actively seek to obtain, where appropriate, the broadest intellectual property protection possible for our product
candidates, proprietary information and proprietary technology through a combination of contractual arrangements and patents, both
in the U.S. and elsewhere in the world.
We also depend upon the
skills, knowledge and experience of our scientific and technical personnel, as well as that of our advisors, consultants and other
contractors, none of which is patentable. To help protect our proprietary knowledge and experience that is not patentable, and
for inventions for which patents may be difficult to enforce, we rely on trade secret protection and confidentiality agreements
to protect our interests. To this end, we require all employees, consultants, advisors and other contractors to enter into confidentiality
agreements, which prohibit the disclosure of confidential information and, where applicable, require disclosure and assignment
to us of the ideas, developments, discoveries and inventions important to our business.
We have issued U.S. patents
8,263,582 and 6,096,731 that cover the use of oral BDP for treating inflammatory disorders of the gastrointestinal tract and the
prevention and treatment of gastrointestinal graft-versus-host disease, respectively. U.S. patent numbers 8,263,582 and 6,096,731
are expected to expire in March 2022 and June 2018, respectively. We also have European patent EP 1392321 claiming the use of topically
active corticosteroids in orally administered dosage forms that act concurrently to treat inflammation in the upper and lower gastrointestinal
tract, as well as European patent EP 2242477 claiming the use of orally ingested BDP for treatment of interstitial lung disease.
European patents EP 1392321 and EP 2242477 are expected to expire in March 2022 and January 2029.
The subject of U.S. patent
application number 12/633,631 filed December 8, 2009 and corresponding European patent application number 09836727.9 is the use
of topically active BDP in radiation and chemotherapeutics injury. Additionally, we have numerous patent filings currently issued
or pending in foreign jurisdictions covering this subject matter, including Australia, Canada, China, Hong Kong, Israel, India,
Japan, South Korea and New Zealand.
ThermoVax™ is the
subject of U.S. patent 8,444,991 issued on May 21, 2013 titled “Method of Preparing an Immunologically-Active Adjuvant-Bound
Dried Vaccine Composition” and also U.S. patent application number 13/474,661 filed May 17, 2012 titled “Thermostable
Vaccine Compositions and Methods of Preparing Same.” The patent application and the corresponding foreign filings for both
patents are pending and licensed to us by the University of Colorado (“UC”) and they address the use of adjuvants in
conjunction with vaccines that are formulated to resist thermal inactivation. The license agreement covers thermostable vaccines
for biodefense as well as other potential vaccine indications. U.S. patent 8,444,991 is expected to expire in December 2031.
RiVax™ is the subject
of three issued U.S. patent numbers 6,566,500, 6,960,652, and 7,829,668, all titled “Compositions and methods for modifying
toxic effects of proteinaceous compounds.” This patent family includes composition of matter claims for the modified ricin
toxin A chain which is the immunogen contained in RiVax™, and issued in 2003, 2005 and 2010 respectively. The initial filing
date of these patents is March 2000 and they are expected to expire in March 2020. The issued patents contain claims that describe
alteration of sequences within the ricin A chain that affect vascular leak, one of the deadly toxicities caused by ricin toxin.
Another U.S. patent number 7,175,848 titled “Ricin A chain mutants lacking enzymatic activity as vaccines to protect against
aerosolized ricin,” was filed in October of 2000 and is expected to expire in October 2020.
In 2013, we expanded our
patent portfolio to include innate defense regulation through the acquisition of the novel drug technology, known as SGX94. By
binding to the pivotal regulatory protein p62, also known as sequestosome-1, SGX94 regulates the innate immune system to reduce
inflammation, eliminate infection and enhance healing. As part of the acquisition, we acquired all rights, including composition
of matter patents for SGX94 as well as other analogs and crystal structures of SGX94 with its protein target p62, including U.S.
patent 8,124,721 and additional pending applications, both in the U.S. and abroad. SGX94 was developed pursuant to discoveries
made by Professors B. Brett Finlay and Robert Hancock of the University of British Columbia (“UBC”). U.S. patent
8,124,721 is expected to expire in April 2028.
We recently acquired a
novel, first-in-class, photodynamic therapy that utilizes safe visible light for activation, which we refer to as SGX301. The active
ingredient in SGX301 is synthetic hypericin, a photosensitizer which is topically applied to skin lesions and then activated by
fluorescent light 16 to 24 hours later. As part of the acquisition, we acquired a license agreement relating to the use of photo-activated
hypericin, composition of matter patent for SGX301 (U.S. patent 8,629,302) and additional issued and pending applications, both
in the U.S. and abroad. U.S. patent 8,629,302 is expected to expire in June 2032.
In addition to issued and
pending patents, we also have “Orphan Drug” designations for SGX301 in the U.S. for CTCL, SGX203 in the U.S. for pediatric
Crohn’s disease, and OrbeShield™ in the U.S. for GI ARS, as well as for RiVax™ in the U.S. Our Orphan Drug designations
provide for seven years of post-approval marketing exclusivity in the U.S. and ten years exclusivity in Europe. We have pending
patent applications for this indication that, if granted, may extend our anticipated marketing exclusivity beyond the U.S. seven
year or E.U. ten year post-approval exclusivity provided by Orphan Drug legislation.
Oral BDP License Agreement
On November 24, 1998, the
Company, known at the time as Enteron Pharmaceuticals, Inc. (“Enteron”) and George B. McDonald (“Dr. McDonald”)
entered into an exclusive license agreement for the rights to intellectual property, including know-how, relating to oral BDP.
The Company has an exclusive license to commercially exploit the covered products worldwide, subject to Dr. McDonald’s
right to make and use the technology for research purposes and the U.S. Government’s right to use the technology for government
purposes. Pursuant to the license agreement, as amended, the Company is required to (i) reimburse Dr. McDonald for certain out-of-pocket
expenses incurred by Dr. McDonald in connection with the patent applications and issued patents, (ii) pay Dr. McDonald
$400,000 upon approval by the FDA of the Company’s first NDA incorporating oral BDP; (iii) pay Dr. McDonald royalty
payments equal to 3% of net sales of the covered products and (iv) pay Dr. McDonald $400,000 in cash upon an approval of oral BDP
by the European Medicines Agency.
Additionally, in the event
that the Company sublicenses its rights under the license agreement, the Company will be required to pay Dr. McDonald 10% of any
sublicense fees and royalty payments paid by the sublicense to the Company.
The term of the license
agreement expires upon the expiration of the licensed patent applications or patents. After seven years from the date of the agreement,
Dr. McDonald has the right to terminate the license agreement in its entirety or to terminate exclusivity under the agreement if
the Company or its sublicense has not commercialized or are not actively attempting to commercialize a covered product.
Additionally, the agreement
terminates: (i) automatically upon the Company becoming insolvent; (ii) upon 30 days’ notice, if the Company breaches any
obligation under the agreement without curing such breach during the notice period; and (iii) upon 90 days’ notice by the
Company. After any termination, the Company will have the right to sell its inventory for a period not to exceed three months following
the date of termination, subject to the payment of the amounts owed under the agreement.
SGX94 License Agreements
On December 18, 2012, we
announced the acquisition of a novel drug technology, known as SGX94, representing a novel approach to modulation of the innate
immune system. SGX94 is an IDR that regulates the innate immune system to reduce inflammation, eliminate infection and enhance
tissue healing by binding to the pivotal regulatory protein p62, also known as sequestosome-1. As part of the acquisition, Soligenix
acquired all rights, including composition of matter patents, preclinical and Phase 1 clinical study datasets for SGX94. We also
assumed a license agreement with UBC to advance the research and development of the SGX94 technology. The license agreement with
UBC provides us with exclusive worldwide rights to manufacture, distribute, market sell and/or license or sublicense products derived
or developed from this technology. Under the license agreement we are obligated to pay UBC (i) an annual license maintenance fee
of CAN $1,000, and (ii) milestone payments which could reach up to CAN $1.2 million.
ThermoVaxTM License Agreement
On September 1, 2009, we
executed a worldwide exclusive option to license patent applications with the UC for ThermoVax™ which is the subject of U.S.
patent number 8,444,991 issued on May 21, 2013 titled “Method of Preparing an Immunologically-Active Adjuvant-Bound Dried
Vaccine Composition.” This patent and its corresponding foreign filings are licensed to Soligenix by the UC and they address
the use of adjuvants in conjunction with vaccines that are formulated to resist thermal inactivation. U.S. Patent 8,444,991 is
expected to expire in December 2031. The license agreement also covers thermostable vaccines for biodefense as well as other potential
vaccine indications. In addition, Soligenix in conjunction with UC, filed domestic and foreign patent applications claiming
priority back to a provisional application filed on May 17, 2011 titled: “Thermostable Vaccine Compositions and Methods of
Preparing Same.”
RiVax™ License Agreement
In January 2003, we executed
a worldwide exclusive option to license patent applications from UTSW for the nasal, pulmonary and oral uses of a non-toxic ricin
vaccine. In July 2003, we entered into a license agreement with UTSW for the injectable rights to the ricin vaccine and, in October
2004, we negotiated the remaining oral rights to the ricin vaccine. Our license obligates us to pay $50,000 in annual license fees.
Through this license, we have rights to the issued patent number 7,175,848 titled “Ricin A chain mutants lacking enzymatic
activity as vaccines to protect against aerosolized ricin.” This patent includes methods of use and composition claims for
RiVax™. UTSW has initiated discussions with us with respect to the continuation of our license to the technology relating
to RiVax™.
VeloThrax™ License Option Agreement
On March 5, 2013, we optioned
a license to the VeloThrax™ patent from the President and Fellows of Harvard College. VeloThrax™ is the subject of
U.S. patent No. 7,037,503, issued on May 2, 2006 and titled, “Compounds and Methods for the Treatment and Prevention of Bacterial
Infection”, along with any reissue, renewal, reexamination, substitution or extension thereof.
Intrexon Exclusive Channel Collaboration
Agreement
On April 27, 2013, we entered
into an exclusive channel collaboration agreement with Intrexon (the “Channel Agreement”) that governs an arrangement
in which we intend to use Intrexon’s advanced human antibody discovery, isolation and production technologies for the development
of human monoclonal antibody therapies for a new biodefense application. The target of the channel collaboration will be melioidosis,
a potentially lethal disease caused by the Gram-negative bacteria Burkholderia pseudomallei, which is endemic in Southeast
Asia and Northern Australia.
The Channel Agreement grants
us an exclusive license to use specified patents and other intellectual property of Intrexon in connection with the research, development,
use, importing, manufacture, sale and offer for sale of products for the treatment of melioidosis through the use of exogenously
produced human recombinant monoclonal antibodies.
In exchange for the license,
we paid Intrexon a one-time technology access fee of $1.5 million in common stock. Additionally, the Channel Agreement requires
us to make certain milestone payments to Intrexon which could reach up to $7 million and to pay Intrexon royalty payments based
upon sales of products based upon Intrexon’s technology.
SGX301 License Agreement
In September 2014, we acquired
a worldwide exclusive license agreement with New York University and Yeda Research and Development Company Ltd. for the rights
to a novel, first-in-class, photodynamic therapy that utilizes safe visible light for activation, which we refer to as SGX301.
Our license obligates us to pay $25,000 in annual license fees. In addition, we will pay the licensors: (a) a royalty amount equal
to 3% of all net sales of SGX301 made directly by us and/or any affiliates; (b) a royalty amount equal to 2.5% of all net sales
of SGX301 made by our sublicensees, subject to stated maximums and (b) 20% of all payments, not based on net sales, received by
us from our sublicensees. The exclusive license includes rights to several issued U.S. patents, including U.S. patent numbers 6,867,235
and 7,122,518, among other domestic and foreign patent applications. U.S. Patent numbers 6,867,235 and 7,122,518 are expected to
expire in January 2020 and November 2023, respectively.
We acquired the license
agreement for SGX301 and related intangible assets, properties and rights pursuant to asset purchase agreement with Hy Biopharma
Inc. (“Hy Biopharma”). As consideration for the assets acquired, we paid $250,000 in cash and issued 1,849,113 shares
of common stock with a market value of $3,750,000. Provided all future success-orientated milestones are attained, we will be required
to make payments of up to $10.0 million, if and when achieved, payable in common stock of the Company.
Research and Development Expenditure
We spent approximately
$9.1 million and $5.1 million in the years ended December 31, 2014 and 2013, respectively, and approximately $1.0 million and $1.0
million during the three months ended March 31, 2015 and 2014, respectively on research and development. The amounts we spent on
research and development per product during the years ended December 31, 2014, and 2013, and the three months ended March 31, 2015
and 2014, are set forth in “Management’s Discussion and Analysis of Financial Condition and Results of Operations”
beginning on page 28 of this prospectus.
Employees
As of March 31, 2015, we
had 17 full-time employees, seven of whom are MDs/PhDs.
Properties
We
currently lease approximately 5,200 square feet of office space at 29 Emmons Drive, Suite C-10, Princeton, New Jersey 08540. This
office space currently serves as our corporate headquarters. In December 2014, we entered into a lease agreement through May 31,
2018 for existing and expanded office space. The rent for the first 12 months is approximately $12,300 per month, or approximately
$20.85 per square foot. This rent increases to approximately $12,375 per month, or approximately $20.95 per square foot, for the
next 12 months, and thereafter to approximately $12,460 per month, or approximately $21.13 per square foot for the remainder of
the lease. Our office space is sufficient to satisfy our current needs.
Legal Proceedings
From
time to time, we are a party to claims and legal proceedings arising in the ordinary course of business. Our management evaluates
our exposure to these claims and proceedings individually and in the aggregate and allocates additional monies for potential losses
on such litigation if it is possible to estimate the amount of loss and if the amount of the loss is probable.
MANAGEMENT
The table below contains
information regarding the current members of the Board of Directors and executive officers. The ages of individuals are provided
as of July 28, 2015:
| Name |
|
Age |
|
|
Position |
| Christopher J. Schaber, PhD |
|
|
48 |
|
|
Chairman of the Board, Chief Executive Officer and President |
| Keith L. Brownlie, CPA |
|
|
63 |
|
|
Director |
| Marco M. Brughera, DVM |
|
|
60 |
|
|
Director |
| Gregg A. Lapointe, CPA |
|
|
56 |
|
|
Director |
| Robert J. Rubin, MD |
|
|
69 |
|
|
Director |
| Jerome Zeldis, MD, PhD |
|
|
65 |
|
|
Director |
| Oreola Donini, PhD |
|
|
43 |
|
|
Chief Scientific Officer and Senior Vice President |
| Richard Straube, MD |
|
|
63 |
|
|
Chief Medical Officer and Senior Vice President |
| Joseph M. Warusz, CPA |
|
|
59 |
|
|
Vice President of Finance, Acting Chief Financial Officer and Corporate Secretary |
Christopher J. Schaber,
PhD has over 25 years of experience in the pharmaceutical and biotechnology industry. Dr. Schaber has been our President and
Chief Executive Officer and a director since August 2006. He was appointed Chairman of the Board on October 8, 2009. He also has
served on the board of directors of the Biotechnology Council of New Jersey (“BioNJ”) since January 2009 and the Alliance
for Biosecurity since October 2014, and has been a member of the corporate councils of both the National Organization for Rare
Diseases (“NORD”) and the American Society for Blood and Marrow Transplantation (“ASBMT”) since October
2009 and July 2009, respectively. Prior to joining Soligenix, Dr. Schaber served from 1998 to 2006 as Executive Vice President
and Chief Operating Officer of Discovery Laboratories, Inc., where he was responsible for overall pipeline development and key
areas of commercial operations, including regulatory affairs, quality control and assurance, manufacturing and distribution, pre-clinical
and clinical research, and medical affairs, as well as coordination of commercial launch preparation activities. From 1996 to 1998,
Dr. Schaber was a co-founder of Acute Therapeutics, Inc., and served as its Vice President of Regulatory Compliance and Drug Development.
From 1994 to 1996, Dr. Schaber was employed by Ohmeda PPD, Inc., as Worldwide Director of Regulatory Affairs and Operations. From
1989 to 1994, Dr. Schaber held a variety of regulatory, development and operations positions with The Liposome Company, Inc., and
Elkins-Sinn Inc., a division of Wyeth-Ayerst Laboratories. Dr. Schaber received his BA degree from Western Maryland College, his
MS degree in Pharmaceutics from Temple University School of Pharmacy and his PhD degree in Pharmaceutical Sciences from the Union
Graduate School. Dr. Schaber was selected to serve as a member of our Board of Directors because of his extensive experience
in drug development and pharmaceutical operations, including his experience as an executive senior officer with our Company and
Discovery Laboratories, Inc., and as a member of the board of directors of BioNJ; because of his proven ability to raise funds
and provide access to capital; and because of his advanced degrees in science and business.
Keith L. Brownlie, CPA
has been a director since June 2011. Mr. Brownlie currently serves on the Board of Directors of Rxi Pharmaceuticals Corporation,
a publicly traded biotechnology company involved in the research and development of RNAi products for the diagnosis, prevention
and treatment of human diseases, a position he has held since June 2012. From July 2013 until December 2014, Mr. Brownlie
served on the Board of Directors of Cancer Genetics, Inc., a publicly traded, early stage diagnostics company. Mr. Brownlie served
as a member of the Board of Directors of Epicept Corporation, a publicly traded, specialty pharmaceutical company focused on the
clinical development and commercialization of pharmaceutical products for the treatment of cancer and pain, from April 2011 to
August 2013 when Epicept Corporation merged with Immune Pharmaceuticals, Inc. From 1974 to 2010, Mr. Brownlie worked with the accounting
firm of Ernst & Young LLP where he served as audit partner for numerous public companies and was the Life Sciences Industry
Leader for the New York metro area. Mr. Brownlie received a BS in Accounting from Lehigh University and is a Certified Public Accountant
in the state of New Jersey. Mr. Brownlie co-founded the New Jersey Entrepreneur of the Year Program and was Vice President and
Trustee of the New Jersey Society of CPAs. In addition, he served as accounting advisor to the board of the Biotechnology Council
of New Jersey. Mr. Brownlie was selected to serve as a member of our Board of Directors because of his vast experience as an audit
partner for numerous public companies and as a director of publicly traded specialty pharmaceutical and biotechnology companies.
Marco Brughera, DVM
joined the Board of Directors in October 2013. He is the Global Head of the Rare Disease Franchise for Sigma-Tau S.p.A., a position
he has held since October 2012. From December 2011 through January 2014, Dr. Brughera served on the Board of Directors of Gentium
S.p.A., a publicly traded biopharmaceutical company. From January 2011 through October 2012, Dr. Brughera held several other positions
with the Sigma-Tau Group, including Corporate Research and Development Managing Director of Sigma-Tau S.p.A., President of Sigma-Tau
Research Switzerland S.A. and board member of Sigma-Tau Pharmaceuticals, Inc. and of Sigma Tau Rare Diseases S.A. From 2004 to
2010, Dr. Brughera served as the Vice President of Preclinical Development at Nerviano Medical Sciences S.r.l. (“NMS Group”),
a pharmaceutical oncology-focused integrated discovery and development company. He also served as the Managing Director at Accelera,
S.r.l., an independent contract research organization affiliated with the NMS Group. From 1999 to 2004, Dr. Brughera held several
senior level positions in the areas of discovery and development toxicology with Pharmacia Corporation and Pfizer, Inc. Prior to
1999, he held various positions at Pharmacia & Upjohn Company, Inc., and Farmitalia Carlo Erba S.p.A., an Italian pharmaceutical
company. Dr. Brughera earned his degree in veterinary medicine from the University of Milan and is a European Registered Toxicologist.
Pursuant to our February 11, 2009 stock purchase agreement with Sigma-Tau Pharmaceuticals, Inc., as long as Sigma-Tau beneficially
owns at least 10% of our issued and outstanding shares of common stock, we are required to use our best efforts to secure the election
of a Sigma-Tau designee to our Board of Directors. In view of Dr. Brughera’s background in the areas of drug discovery
and development and his experience as an executive officer and a director in the pharmaceutical industry, the Nominating Committee
accepted Dr. Brughera as Sigma-Tau’s designee for election to the Board of Directors.
Gregg Lapointe, CPA,
MBA has been a director since March 2009. Mr. Lapointe is currently CEO of Cerium Pharmaceuticals, Inc. and serves on the Board
of Directors of SciClone Pharmaceuticals, Inc., Cambrooke Therapeutics, Inc., Raptor Pharmaceuticals, Inc., and the Board of Trustees
of the Keck Graduate Institute of Applied Life Sciences. He has previously served on the Board of Directors of the Pharmaceuticals
Research and Manufacturers of America (PhRMA) and Questcor Pharmaceuticals, Inc., and has been a member of the Corporate Council
of NORD for several years. He previously served in varying roles for Sigma-Tau Pharmaceuticals, Inc., a private biopharmaceutical
company, from September 2001 through February 2012, including Chief Operating Officer from November 2003 to April 2008 and Chief
Executive Officer from April 2008 to February 2012. From May, 1996 to August 2001, he served as Vice President of Operations and
Vice President, Controller of AstenJohnson, Inc. (formerly JWI Inc.). Prior to that, Mr. Lapointe spent several years in the Canadian
medical products industry in both distribution and manufacturing. Mr. Lapointe began his career at Price Waterhouse World Firm.
Mr. Lapointe received his B.A. degree in Commerce from Concordia University in Montreal, Canada, a graduate diploma in Accountancy
from McGill University and his M.B.A. degree from Duke University. He is a C.P.A. in the state of Illinois. Mr. Lapointe was selected
to serve as a member of our Board of Directors because of his significant experience in the areas of global strategic planning
and implementation, business development, corporate finance, and acquisitions, and his experience as an executive officer and board
member in the pharmaceutical and medical products industries.
Robert J. Rubin, MD
has been a director since October 2009. Dr. Rubin was a clinical professor of medicine at Georgetown University from 1995 until
2012 when he was appointed a Distinguished Professor of Medicine. From 1987 to 2001, he was president of the Lewin Group (purchased
by Quintiles Transnational Corp. in 1996), an international health policy and management consulting firm. From 1994 to 1996, Dr.
Rubin served as Medical Director of ValueRx, a pharmaceutical benefits company. From 1992 to 1996, Dr. Rubin served as President
of Lewin-VHI, a health care consulting company. From 1987 to 1992, he served as President of Lewin-ICF, a health care consulting
company. From 1984 to 1987, Dr. Rubin served as a principal of ICF, Inc., a health care consulting company. From 1981 to 1984,
Dr. Rubin served as the Assistant Secretary for Planning and Evaluation at the Department of Health and Human Services and as an
Assistant Surgeon General in the United States Public Health Service. Dr. Rubin has served on the Board of BioTelemetry, Inc. (formerly
known as CardioNet, Inc.) since 2007. He is a board certified nephrologist and internist. Dr. Rubin received an undergraduate degree
in Political Science from Williams College and his medical degree from Cornell University Medical College. Dr. Rubin was selected
to serve as a member of our Board of Directors because of his vast experience in the health care industry, including his experience
as a nephrologist, internist, clinical professor of medicine and Assistant Surgeon General, and his business experience in the
pharmaceutical industry.
Jerome Zeldis, MD, PhD
has been a director since June 2011. Dr. Zeldis is currently Chief Executive Officer of Celgene Global Health and Chief Medical
Officer of Celgene Corporation, a publicly traded, fully integrated biopharmaceutical company, where he has been employed since
1997. From September 1994 to February 1997, Dr. Zeldis worked at Sandoz Research Institute and the Janssen Research Institute in
both clinical research and medical development. He has been a board member of several biotechnology companies and is currently
on the boards of the NJ Chapter of the Arthritis Foundation, the Castleman’s Disease Organization and PTC Therapeutics, Inc.
and Alliqua, Inc. Additionally, he has served as Assistant Professor of Medicine at the Harvard Medical School (from July 1987
to September 1988), Associate Professor of Medicine at University of California, Davis from (September 1988 to September 1994),
Clinical Associate Professor of Medicine at Cornell Medical School (January 1995 to December 2003) and Professor of Clinical Medicine
at the Robert Wood Johnson Medical School (July 1998 to June 2010). Dr. Zeldis received a BA and an MS from Brown University, and
an MD, and a PhD in Molecular Biophysics and Biochemistry from Yale University. Dr. Zeldis trained in Internal Medicine at the
UCLA Center for the Health Sciences and in Gastroenterology at the Massachusetts General Hospital and Harvard Medical School. Dr.
Zeldis was selected to serve as a member of our Board of Directors because of his experience as an executive officer of a publicly
traded biopharmaceutical company and in clinical research and medical development, and his experience in the health care industry,
including his experience as an internist, gastroenterologist and professor of medicine.
Oreola Donini, PhD,
has been with our company since August 15, 2013 and is currently our Chief Scientific Officer and Senior Vice President, a position
she has held since December 5, 2014. Dr. Donini served as our Vice President of Preclinical Research and Development from August
15, 2013 until December 4, 2014. She has more than 15 years’ experience in drug discovery and preclinical development with
start-up biotechnology companies. From 2012 to 2013, Dr. Donini worked with ESSA Pharma Inc. as Vice President Research and Development.
From 2004 to 2012, Dr. Donini worked with Inimex Pharmaceuticals Inc., (“Inimex”), lastly as Senior Director of Preclinical
R&D from 2007-2013. Prior to joining Inimex, she worked with Kinetek Pharmaceuticals Inc., developing therapies for infectious
disease, cancer and cancer supportive care. Dr. Donini is a co-inventor and leader of the Company’s SGX94 innate defense
regulator technology, developed by Inimex and subsequently acquired by the Company. She was responsible for overseeing the manufacturing
and preclinical testing of SGX94, which demonstrated efficacy in combating bacterial infections and mitigating the effects of tissue
damage due to trauma, infection, radiation and/or chemotherapy treatment. These preclinical studies resulted in a successful Phase
1 clinical study and clearance of Phase 2 protocols for oral mucositis in head and neck cancer and acute bacterial skin and skin
structure infections. While with ESSA Pharma Inc. as the Vice President of Research and Development, Dr. Donini led the preclinical
testing of a novel N-terminal domain inhibitor of the androgen receptor for the treatment of prostate cancer. While with Kinetek
Pharmaceuticals Inc., her work related to the discovery of novel kinase and phosphatase inhibitors for the treatment of cancer.
Dr. Donini received her PhD from Queen’s University in Kinston, Ontario, Canada and completed her post-doctoral work at the
University of California, San Francisco. Her research has spanned drug discovery, preclinical development, manufacturing and clinical
development in infectious disease, cancer and cancer supportive care.
Richard Straube, MD
has been with our company since January 2014 and is currently our Senior Vice President and Chief Medical Officer. Dr. Straube
is a board-certified pediatrician with 35 years’ experience in both academia and industry, including clinical research experience
in host-response modulation. From 2009 until joining our company, he was Chief Medical Officer of Stealth Peptides Incorporated,
a privately-held, clinical stage, biopharmaceutical company. Prior to joining the Company, Dr. Straube served from 1988 to
1993 in various capacities, including most recently as Senior Director, Infectious Diseases and Immunology, Clinical Research,
for Centocor, Inc., a privately-held biopharmaceutical company focused on developing monoclonal antibody-based diagnostics. While
at Centocor, Inc., Dr. Straube was responsible for the initial anti-cytokine and anti-endotoxin programs targeted at ameliorating
inappropriate host responses to infectious and immunologic challenges. Programs that he managed at Centocor, Inc. include assessments
of immunomodulation using monoclonal removal of inciting molecular triggers, removal of internal immune-messengers, augmentation
of normal host defenses, and maintenance of normal sub-cellular function in the face of injury. From 1993 to 1995, Dr.
Straube was Director of Medical Affairs at T-cell Sciences, Inc., a privately-held biotechnology company. From 1995
to 1997, he was Director of Clinical Investigations of the Pharmaceutical Products Division of Ohmeda Corp., a privately-held biopharmaceutical
company. He served from 1998 to 2007 as Executive Vice President of Research and Development and Chief Scientific Officer
at INO Therapeutics LLC, a privately-held biotherapeutics company, where he was responsible for the clinical trials and subsequent
approval of inhaled nitric oxide for the treatment of persistent pulmonary hypertension of the newborn. From 2007 to
2009, Dr. Straube was the Chief Medical Officer at Critical Biologics Corporation, a privately-held biotechnology company. Dr.
Straube received his medical degree and residency training at the University of Chicago, completed a joint adult and pediatrician
infectious diseases fellowship at the University of California, San Diego (“UCSD”), and as a Milbank Scholar completed
training in clinical trial design at the London School of Hygiene and Tropical Medicine. While on the faculty at the
UCSD Medical Center, his research focused on interventional studies for serious viral infections.
Joseph M. Warusz, CPA
has been with the company since June 2011 and is currently our Vice President of Finance and Acting Chief Financial Officer,
a position he has held since February 2012. He has more than 30 years of financial management experience in public and private
life science companies as well as large pharma. Prior to joining Soligenix on June 1, 2011 as Vice President of Administration
and Controller, he held senior financial positions with Amicus Therapeutics, Inc. from 2004 to 2005, Orchid Cellmark, Inc. from
2000 to 2004, and NexMed, Inc. from 1998 to 1999. From 2005 to 2011, Mr. Warusz performed consulting assignments at Ardea BioSciences,
Inc., NovaDel Pharma, Inc. and Melior Discovery, all R&D-focused companies in the biotechnology and specialty pharmaceuticals
arenas. Prior to 1998, Mr. Warusz also held management positions in financial analysis, accounting, reporting and auditing at Bristol-Myers
Squibb and Peat Marwick Main & Company. He received his BS in accounting and MBA in finance at Drexel University and is a Certified
Public Accountant.
Board Leadership Structure
Our Board of Directors
believes that Dr. Schaber’s service as both the Chairman of our Board of Directors and our Chief Executive Officer is in
the best interest of our Company and our stockholders. Dr. Schaber possesses detailed and in-depth knowledge of the issues, opportunities
and challenges facing our Company and our business and, therefore, is best positioned to develop agendas that ensure that the Board
of Directors’ time and attention are focused on the most important matters. His combined role enables decisive leadership,
ensures clear accountability, and enhances our ability to communicate our message and strategy clearly and consistently to our
stockholders, employees, and collaborative partners.
Messrs. Brownlie and Lapointe,
Dr. Rubin, and Dr. Zeldis are independent and the Board of Directors believes that the independent directors provide effective
oversight of management. Moreover, in addition to feedback provided during the course of meetings of the Board of Directors, the
independent directors hold executive sessions. Following an executive session of independent directors, the independent directors’
report back to the full Board of Directors regarding any specific feedback or issues, provide the Chairman with input regarding
agenda items for Board of Directors and Committee meetings, and coordinate with the Chairman regarding information to be provided
to the independent directors in performing their duties. The Board of Directors believes that this approach appropriately and effectively
complements the combined Chairman/Chief Executive Officer structure.
Although the Company believes
that the combination of the Chairman and Chief Executive Officer roles is appropriate under the current circumstances, our corporate
governance guidelines do not establish this approach as a policy, and the Board of Directors may determine that it is more appropriate
to separate the roles in the future.
Director Independence
The Board of Directors
has determined that Keith Brownlie, Gregg Lapointe, Dr. Robert Rubin and Dr. Jerome Zeldis are “independent” as such
term is defined by the applicable listing standards of The NASDAQ Stock Market LLC (“Nasdaq”). Our Board of Directors
based this determination primarily on a review of the responses of the Directors to questionnaires regarding their employment,
affiliations and family and other relationships.
Committees of the Board of Directors
Our Board of Directors
has the following three committees: (1) Compensation, (2) Audit and (3) Nominating and Corporate Governance. Our
Board of Directors has adopted a written charter for each of these committees, which are available on our website at www.soligenix.com
under the “Investors” section.
| Director |
|
Audit
Committee |
|
Compensation
Committee |
|
Nominating and
Corporate Governance
Committee |
| Keith L. Brownlie, CPA |
|
 |
|
|
|
 |
| Marco M. Brughera, DVM |
|
|
|
 |
|
|
| Gregg A. Lapointe, CPA |
|
 |
|
|
|
 |
| Robert J. Rubin, MD |
|
 |
|
 |
|
|
| Jerome Zeldis, MD, PhD |
|
|
|
 |
|
 |
 | | – Committee Chair |
 | | – Member |
Compensation Committee
Our Board of Directors
has a Compensation Committee, which is comprised of Dr. Rubin (Chair), Dr. Brughera and Dr. Zeldis. The Compensation Committee
is responsible for reviewing and approving the executive compensation program, assessing executive performance, setting salary,
making grants of annual incentive compensation and approving certain employment agreements. Our Board of Directors has determined
that Dr. Rubin and Dr. Zeldis are “independent” directors within the meaning of applicable listing standards of Nasdaq
and the Securities Exchange Act of 1934, as amended (the “Exchange Act”) and the rules and regulations thereunder.
Our Board of Directors reviewed Dr. Brughera’s relationship as the Global Head of the Rare Disease Franchise for Sigma-Tau
SpA., an affiliate of Sigma-Tau Pharmaceuticals, Inc., which is owns approximately 13.72% of the issued and outstanding shares
of our common stock. Our Board of Directors determined that Dr. Brughera’s position with Sigma-Tau SpA. would not impair
his ability to exercise independent judgment.
Nominating and Corporate Governance Committee
Our Board of Directors
has a Nominating and Corporate Governance Committee (“Nominating Committee”), which is comprised of Dr. Zeldis (Chair),
Mr. Brownlie and Mr. Lapointe. The Nominating Committee makes recommendations to the Board of Directors regarding the size and
composition of our Board of Directors, establishes procedures for the nomination process, identifies and recommends candidates
for election to our Board of Directors. Our Board of Directors has determined that Dr. Zeldis, Mr. Brownlie and Mr. Lapointe are
“independent” directors, as such term is defined by the applicable Nasdaq listing standards.
Audit Committee
Our Board of Directors
has an Audit Committee, which is comprised of Mr. Brownlie (Chair), Mr. Lapointe and Dr. Rubin. The Audit Committee
assists our Board of Directors in monitoring the financial reporting process, the internal control structure and the independent
registered public accountants. Its primary duties are to serve as an independent and objective party to monitor the financial reporting
process and internal control system, to review and appraise the audit effort of the independent registered public accountants and
to provide an open avenue of communication among the independent registered public accountants, financial and senior management,
and our Board of Directors. Our Board of Directors has determined that Mr. Brownlie, Mr. Lapointe and Dr. Rubin are
“independent” directors, within the meaning of applicable listing standards of Nasdaq and the Exchange Act and the
rules and regulations thereunder. Our Board of Directors has also determined that the members of the Audit Committee
are qualified to serve on the committee and have the experience and knowledge to perform the duties required of the committee and
that Mr. Brownlie qualifies as an “audit committee financial expert” as that term is defined in the applicable regulations
of the Exchange Act.
Code of Ethics
We have adopted a code
of ethics that applies to all of our executive officers and senior financial officers (including our chief executive officer, chief
financial officer, chief accounting officer and any person performing similar functions). A copy of our code of ethics is publicly
available on our website at http://www.soligenix.com under the “Investors” section. If we make any substantive
amendments to our code of ethics or grant any waiver, including any implicit waiver, from a provision of the code to our chief
executive officer, chief financial officer or chief accounting officer, we will disclose the nature of such amendment or waiver
in a Current Report on Form 8-K.
Diversity Considerations in Identifying
Director Nominees
We do not have a formal
diversity policy or set of guidelines in selecting and appointing directors that comprise our Board of Directors. However, when
making recommendations to our Board of Directors regarding the size and composition of our Board of Directors, our Nominating Committee
does consider each individual director’s qualifications, skills, business experience and capacity to serve as a director
and the diversity of these attributes for the Board of Directors as a whole.
Compensation Committee Interlocks and Insider
Participation
No member of our Compensation
Committee is or has at any time during the past year been one of our officers or employees. None of our executive officers currently
serves or in the past year has served as a member of the Board of Directors or Compensation Committee of any entity that has one
or more executive officers serving on our Board of Directors or Compensation Committee.
Stock Ownership Policy
In April 2012, our Board
of Directors adopted a stock ownership policy applicable to our non-employee directors to strengthen the link between director
and stockholder interests. Pursuant to the stock ownership policy, each non-employee director is required to hold a minimum ownership
position in the common stock equal to the annual cash compensation paid for service on the Board of Directors, exclusive of cash
compensation paid for service as a chair or member of any committees of the Board of Directors.
Stock counted toward the
ownership requirement includes common stock held by the director, unvested and vested restricted stock, and all shares of common
stock beneficially owned by the director held in a trust and by a spouse and/or minor children of the director. The policy provides
that the ownership requirement must be attained within three years after the later of June 21, 2012 and the date a director is
first elected or appointed to the Board of Directors. To monitor progress toward meeting the requirement, the Nominating Committee
will review director ownership levels at the end of March of each year. Non-employee directors are prohibited from selling any
shares of common stock unless such director is in compliance with the stock ownership policy. A copy of our director compensation
and stock ownership policy is publicly available on our website at www.soligenix.com under the “Investors” section.
EXECUTIVE
COMPENSATION
Summary
Compensation
The
following table contains information concerning the compensation paid during each of the two years ended December 31, 2014 to
our Chief Executive Officer and each of the two other most highly compensated executive officers during 2014 (collectively, the
“Named Executive Officers”).
| Name |
|
Position |
|
Year |
|
|
Salary |
|
|
Bonus |
|
|
Option
Awards |
|
|
All
Other Compensation |
|
|
Total |
|
| Christopher
J. |
|
CEO
& |
|
|
2014 |
|
|
$ |
412,000 |
|
|
$ |
115,000 |
|
|
$ |
150,000 |
|
|
$ |
29,580 |
|
|
$ |
706,580 |
|
| Schaber 1 |
|
President |
|
|
2013 |
|
|
$ |
402,000 |
|
|
$ |
239,000 |
|
|
$ |
199,000 |
|
|
$ |
33,896 |
|
|
$ |
873,896 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Joseph M. 2 |
|
VP & Acting
|
|
|
2014 |
|
|
$ |
191,000 |
|
|
$ |
41,000 |
|
|
$ |
67,500 |
|
|
$ |
21,197 |
|
|
$ |
320,697 |
|
| Warusz |
|
CFO |
|
|
2013 |
|
|
$ |
186,000 |
|
|
$ |
90,000 |
|
|
$ |
89,550 |
|
|
$ |
32,641 |
|
|
$ |
398,191 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Richard C.
Straube 3 |
|
CMO & Senior
VP |
|
|
2014 |
|
|
$ |
300,000 |
|
|
$ |
62,000 |
|
|
$ |
276,000 |
|
|
$ |
21,328 |
|
|
$ |
659,328 |
|
| 1 |
Dr.
Schaber deferred the payment of his 2014 bonus of $115,000 until January 15, 2015. Option award figures include the value
of common stock option awards at grant date as calculated under FASB ASC 718. Other compensation represents health insurance
costs paid by the Company. |
| 2 |
Mr.
Warusz deferred the payment of his 2014 bonus of $41,000 until January 15, 2015. Option award figures include the value of
common stock option awards at grant date as calculated under FASB ASC 718. Other compensation represents health insurance
costs paid by the Company. |
| 3 |
Dr.
Straube joined the Company on January 1, 2014. He deferred the payment of his 2014 bonus of $62,000 until January 15, 2015.
Option award figures include the value of common stock option awards at grant date as calculated under FASB ASC 718. Other
compensation represents health insurance costs paid by the Company. |
Employment
and Severance Agreements
In
August 2006, we entered into a three-year employment agreement with Christopher J. Schaber, PhD. Pursuant to this employment agreement
we agreed to pay Dr. Schaber a base salary of $300,000 per year and a minimum annual bonus of $100,000. Dr. Schaber’s employment
agreement was renewed in December 27, 2007 for an additional term of three years. We agreed to issue him options to purchase 125,000
shares of our common stock, with one third immediately vesting and the remainder vesting over three years. Upon termination without
“Just Cause” as defined by this agreement, we would pay Dr. Schaber nine months of severance, as well as any accrued
bonuses, accrued vacation, and we would provide health insurance and life insurance benefits for Dr. Schaber and his dependents.
No unvested options shall vest beyond the termination date. Dr. Schaber’s monetary compensation (base salary of $300,000
and bonus of $100,000) remained unchanged from 2006 with the 2007 renewal. Upon a change in control of the Company due to merger
or acquisition, all of Dr. Schaber’s options shall become fully vested, and be exercisable for a period of five years after
such change in control (unless they would have expired sooner pursuant to their terms). In the event of his death during the term
of the agreement, all of his unvested options shall immediately vest and remain exercisable for the remainder of their term and
become the property of Dr. Schaber’s immediate family. Dr. Schaber’s employment agreement automatically renewed in
December 2013 for an additional term of three years.
On
June 22, 2011, the Compensation Committee eliminated his fixed minimum annual bonus payable and revised it to an annual targeted
bonus of 40% of his annual base salary. On December 6, 2012, the Compensation Committee approved an increase in salary for Dr.
Schaber to $402,000. On December 4, 2013, the Compensation Committee approved an increase in salary for Dr. Schaber to $412,000.
On December 4, 2014, the Compensation Committee approved an increase in salary for Dr. Schaber to $424,360
In
May 2011, we entered into a one-year employment agreement with Mr. Joseph M. Warusz, our Acting Chief Financial Officer, Vice
President Finance and Chief Accounting Officer. Pursuant to the agreement, we have agreed to pay Mr. Warusz $175,000 per year
and a targeted annual bonus of 20% of base salary. We also agreed to issue him options to purchase 40,000 shares of our common
stock with one-third immediately vesting and the remainder vesting over three years. Upon termination without “Just Cause”,
as defined in Mr. Warusz’s employment agreement, we would pay Mr. Warusz three months of severance, accrued bonuses and
vacation, and health insurance benefits. No unvested options vest beyond the termination date. On December 1, 2011, the Compensation
Committee increased the salary of Mr. Warusz to $180,000. On December 6, 2012, the Compensation Committee approved an increase
in salary for Mr. Warusz to $186,000 and the targeted annual bonus to 35%. On December 4, 2013, the Compensation Committee approved
an increase in salary for Mr. Warusz to $191,000. On December 4, 2014, the Compensation Committee approved an increase in salary
for Mr. Warusz to $196,730.
In
December 2014, we entered into a one-year employment agreement with Richard C. Straube, MD, our Chief Medical Officer and Senior
Vice President. Pursuant to the agreement, we have agreed to pay Dr. Straube $300,000 per year and a targeted annual bonus of
30% of base salary. We also agreed to issue him options to purchase 100,000 shares of our common stock with one-third immediately
vesting and the remainder vesting over three years. Upon termination without “Just Cause”, as defined in Dr. Straube’s
employment agreement, we would pay Dr. Straube three months of severance, accrued bonuses and vacation, and health insurance benefits.
No unvested options vest beyond the termination date. On December 4, 2014, the Compensation Committee approved an increase in
salary for Dr. Straube to $309,000.
In
February 2007, our Board of Directors authorized the issuance of 50,000 shares to Dr. Schaber immediately prior to the completion
of a transaction, or series or a combination of related transactions negotiated by our Board of Directors whereby, directly or
indirectly, a majority of our capital stock or a majority of our assets are transferred from the Company and/or our stockholders
to a third party. The amended agreement with Dr. Schaber includes our obligation to issue such shares to him if such event occurs.
Outstanding
Equity Awards at Fiscal Year-End
The
following table contains information concerning unexercised options, stock that has not vested, and equity incentive plan awards
for the Named Executive Officers outstanding at December 31, 2014. We have never issued Stock Appreciation Rights.
| Name | |
Number of Securities Underlying Unexercised Options (#) | | |
Equity Incentive Plan Awards: Number of Securities Underlying Unexercised Unearned | | |
Option Exercise | | |
Option Expiration |
| | |
Exercisable | | |
Unexercisable | | |
Options (#) | | |
Price ($) | | |
Date |
| Christopher J. Schaber | |
| 125,000 | | |
| - | | |
| - | | |
$ | 5.40 | | |
8/28/2016 |
| | |
| 45,000 | | |
| - | | |
| - | | |
$ | 9.40 | | |
8/9/2017 |
| | |
| 140,000 | | |
| - | | |
| - | | |
$ | 1.20 | | |
12/17/2018 |
| | |
| 110,000 | | |
| - | | |
| - | | |
$ | 4.64 | | |
6/30/2020 |
| | |
| 112,185 | | |
| - | | |
| - | | |
$ | 0.64 | | |
11/30/2021 |
| | |
| 97,500 | | |
| 32,500 | | |
| 32,500 | | |
$ | 0.68 | | |
12/04/2022 |
| | |
| 50,000 | | |
| 50,000 | | |
| 50,000 | | |
$ | 2.01 | | |
12/04/2023 |
| | |
| 25,000 | | |
| 75,000 | | |
| 75,000 | | |
$ | 1.50 | | |
12/04/2024 |
| | |
| | | |
| | | |
| | | |
| | | |
|
| Richard C. Straube | |
| 43,750 | | |
| 56,250 | | |
| 56,250 | | |
$ | 2.01 | | |
1/06/2024 |
| | |
| 12,500 | | |
| 37,500 | | |
| 37,500 | | |
$ | 1.50 | | |
12/04/2024 |
| | |
| | | |
| | | |
| | | |
| | | |
|
| Joseph M. Warusz | |
| 40,000 | | |
| - | | |
| - | | |
$ | 4.10 | | |
5/30/2021 |
| | |
| 25,310 | | |
| - | | |
| - | | |
$ | 0.64 | | |
11/30/2021 |
| | |
| 41,254 | | |
| 13,746 | | |
| 13,746 | | |
$ | 0.68 | | |
12/04/2022 |
| | |
| 22,502 | | |
| 22,498 | | |
| 22,498 | | |
$ | 2.01 | | |
12/04/2023 |
| | |
| 11,250 | | |
| 33,750 | | |
| 33,750 | | |
$ | 1.50 | | |
12/04/2024 |
Compensation
of Directors
The
following table contains information concerning the compensation of the non-employee directors during the fiscal year ended December
31, 2014.
| Name | |
Fees Earned Paid in Cash 1 | | |
Option Awards 2 | | |
Total | |
| Keith Brownlie | |
$ | 57,500 | | |
$ | 30,000 | | |
$ | 87,500 | |
| Marco Brughera | |
$ | 37,500 | | |
$ | 30,000 | | |
$ | 67,500 | |
| Gregg A. Lapointe | |
$ | 47,500 | | |
$ | 30,000 | | |
$ | 77,500 | |
| Robert J. Rubin | |
$ | 52,500 | | |
$ | 30,000 | | |
$ | 82,500 | |
| Jerry Zeldis | |
$ | 50,000 | | |
$ | 30,000 | | |
$ | 80,000 | |
| 1 |
Directors
who are compensated as full-time employees receive no additional compensation for service on our Board of Directors. Each
director who is not a full-time employee is paid $35,000 annually, on a prorated basis, for his service on our Board of Directors,
the chair of our Audit Committee is paid $15,000 annually, on a prorated basis, and the chairs of our Compensation and Nominating
Committees are paid $10,000 annually, on a prorated basis. Additionally, Audit Committee members are paid $7,500 annually
and Compensation and Nominating Committee members are paid $5,000 annually. This compensation is paid quarterly. |
| 2 |
We
maintain a stock option grant program, whereby members of our Board of Directors or its committees who are not full-time employees
receive an initial grant of fully vested options to purchase 15,000 shares of common stock. Upon re-election to the Board,
each member will receive stock options to purchase up to 25,000 shares of common stock, not to exceed a value of $30,000 (based
upon the fair market value of the common stock on the date that such options are granted), which vest at the rate of 25% per
quarter, commencing with the first quarter after each annual meeting of stockholders. |
CERTAIN
RELATIONSHIPS AND RELATED TRANSACTIONS
Our
audit committee is responsible for the review, approval and ratification of related party transactions. The audit committee reviews
these transactions under our Code of Ethics, which governs conflicts of interests, among other matters, and is applicable to our
employees, officers and directors.
Other
than the employment agreements and compensation paid to our directors, we did not engage in any transactions with related parties
since January 1, 2014.
PRINCIPAL
STOCKHOLDERS
The
table below provides information regarding the beneficial ownership of the common stock as of July 28, 2015, of (1) each person
or entity who owns beneficially 5% or more of the shares of our outstanding common stock, (2) each of our directors, (3) each
of the Named Executive Officers, and (4) our directors and officers as a group. Except as otherwise indicated, and subject to
applicable community property laws, we believe the persons named in the table have sole voting and investment power with respect
to all shares of common stock held by them.
Beneficial
Ownership
| Name of Beneficial Owner | |
Shares of Common Stock Beneficially Owned | | |
Percent of Class | |
| Randall J. Kirk (1) | |
| 6,867,816 | | |
| 23.78 | % |
| NRM VII Holdings I, LLC (1) | |
| 5,833,333 | | |
| 20.20 | % |
| Paolo Cavazza (2) | |
| 3,379,950 | | |
| 12.60 | % |
| Sigma-Tau Pharmaceuticals, Inc. (2) | |
| 3,068,461 | | |
| 11.48 | % |
| Hy Biopharma, Inc. (3) | |
| 1,608,354 | | |
| 6.10 | % |
| Intrexon Corporation (1) | |
| 1,034,483 | | |
| 3.92 | % |
| Christopher J. Schaber (4) | |
| 854,423 | | |
| 3.15 | % |
| Keith Brownlie (5) | |
| 100,307 | | |
| * | |
| Marco Brughera (6) | |
| 28,699 | | |
| * | |
| Gregg A. Lapointe (7) | |
| 165,860 | | |
| * | |
| Robert J. Rubin (8) | |
| 103,574 | | |
| * | |
| Jerry Zeldis (9) | |
| 108,640 | | |
| * | |
| Joseph Warusz (10) | |
| 185,422 | | |
| * | |
| Richard Straube (11) | |
| 84,375 | | |
| * | |
| All directors and executive officers as a group (8 persons) | |
| 1,738,175 | | |
| 6.24 | % |
| (1) |
On
June 26, 2013, Randal J. Kirk, on his own behalf and on behalf of Third Security, LLC, NRM VII Holdings I, LLC and Intrexon
Corporation, filed Amendment No. 1 to Schedule 13D with the Securities and Exchange Commission (the “SEC”), which
amends the Schedule 13D filed May 9, 2013 with the SEC (as amended, “Schedule 13D”). The Schedule 13D states that
Mr. Kirk is Senior Managing Director of, and controls, Third Security, LLC, which is the Manager of an affiliate that manages
NRM VII Holdings I, LLC, and that Mr. Kirk serves as the Chairman and Chief Executive Officer of Intrexon Corporation. The
Schedule 13D indicates that (a) Mr. Kirk, Third Security, LLC and NRM VII Holdings I, LLC have sole voting and dispositive
power with respect to 3,333,333 shares of common stock and warrants to purchase 2,500,000 shares of common stock exercisable
within 60 days of July 28, 2015 held by NRM VII Holdings I, LLC, and (b) Mr. Kirk and Intrexon Corporation have shared voting
and dispositive power with respect to 1,034,483 shares of common stock held by Intrexon Corporation. The address of the principal
business office of Mr. Kirk is 2875 South Ocean Boulevard, Suite 214, Palm Beach, Florida 33480. The address of the principal
business office of NRM VII Holdings I, LLC is c/o Third Security, LLC, 1881 Grove Avenue, Redford, Virginia 24141. The address
of the principal business office of Intrexon Corporation is 20358 Seneca Meadows Parkway, Germantown, Maryland 20876. |
| (2) |
On
May 16, 2013, Paolo Cavazza, on his own behalf and on behalf of Sigma-Tau Finanziaria S.p.A., Sigma-Tau International S.A.,
Sigma-Tau America S.A. and Sigma-Tau Pharmaceuticals, Inc., filed Amendment No. 4 to Schedule 13D with the SEC, which amends
the Schedule 13D filed with the SEC on February 20, 2009 as amended by Amendment No. 1 filed with the SEC on October 2, 2009,
Amendment No. 2 filed with the SEC on June 28, 2010 and Amendment No. 3 filed with the SEC on January 2, 2013 (the “Schedule
13D”). The Schedule 13D indicates that (a) Mr. Cavazza has sole voting and dispositive power with respect to (i) 59,539
shares held by Mr. Paolo Cavazza and (ii) 164,146 shares of common stock and warrants to purchase 87,804 shares held by SINAF
SA, and (b) Mr. Cavazza, Sigma-Tau Finanziaria S.p.A., Sigma-Tau International S.A., Sigma-Tau America S.A. and Sigma-Tau
Pharmaceuticals, Inc. have shared voting and dispositive power with respect to 2,711,392 shares of common stock and warrants
to purchase 357,069 shares of common stock exercisable within 60 days of July 28, 2015 held by Sigma-Tau Pharmaceuticals,
Inc. Sigma-Tau Pharmaceuticals, Inc. is a direct wholly-owned subsidiary of Sigma-Tau America S.A., which is a direct wholly-owned
subsidiary of Sigma-Tau International S.A., which is a direct wholly-owned subsidiary of Sigma-Tau Finanziaria S.p.A. Mr.
Paolo Cavazza directly and indirectly owns 38% of Sigma-Tau Finanziaria S.p.A. SINAF SA is an indirect wholly owned subsidiary
of Aptafin S.p.A., which is owned by Mr. Paolo Cavazza and members of his family. Mr. Paolo Cavazza’s address is Via
Tesserte, 10, Lugano, Switzerland. The business address of Sigma-Tau Finanziaria S.p.A. is Via Sudafrica, 20, Rome, Italy
00144. The business address of Sigma-Tau International S.A. is 19-21 Boulevard du Prince Henri, L-1724 Luxembourg. The business
address of Sigma-Tau America S.A. is 19-21 Boulevard du Prince Henri, L-1724 Luxembourg. The business address of Sigma-Tau
Pharmaceuticals, Inc. is 9841 Washingtonian Boulevard, Suite 500, Gaithersburg, Maryland 20878. |
| |
|
| (3) |
On
October 3, 2014, Hy BioPharma, Inc. filed a Schedule 13G with the SEC (the “Schedule 13G”). The Schedule 13G indicates
that Hy Biopharma, Inc. has sole voting and dispositive power with respect to shares of common stock held by it. The address
of the principal business office of Hy BioPharma, Inc. is 2500 York Road, #100, Jamison, Pennsylvania 18929. |
| |
|
| (4) |
Includes
82,904 shares of common stock, options to purchase 766,560 shares of common stock exercisable within 60 days of July 28, 2015,
and warrants to purchase 4,959 shares of common stock exercisable within 60 days of July 28, 2015. The address of Dr. Schaber
is c/o Soligenix, 29 Emmons Drive, Suite C-10, Princeton, New Jersey 08540. |
| |
|
| (5) |
Includes
50,000 shares of common stock and options to purchase 50,307 shares of common stock exercisable within 60 days of the July
28, 2015. The address of Mr. Brownlie is c/o Soligenix, 29 Emmons Drive, Suite C-10, Princeton, New Jersey 08540. |
| |
|
| (6) |
Includes
7,500 shares of common stock, options to purchase 21,199 shares of common stock exercisable within 60 days of July 28, 2015.
The address of Dr. Brughera is c/o Soligenix, 29 Emmons Drive, Suite C-10, Princeton, New Jersey 08540. |
| |
|
| (7) |
Includes
48,781 shares of common stock, options to purchase 87,811 shares of common stock exercisable within 60 days of July 28, 2015,
and warrants to purchase 29,268 shares of common stock exercisable within 60 days of July 28, 2015. The address of Mr. Lapointe
is c/o Soligenix, 29 Emmons Drive, Suite C-10, Princeton, New Jersey 08540. |
| |
|
| (8) |
Includes
12,196 shares of common stock, options to purchase 84,061 shares of common stock exercisable within 60 days of July 28, 2015,
and warrants to purchase 7,317 shares of common stock exercisable within 60 days of July 28, 2015. The address of Dr. Rubin
is c/o Soligenix, 29 Emmons Drive, Suite C-10, Princeton, New Jersey 08540. |
| (9) |
Includes
66,666 shares of common stock and options to purchase 41,974 shares of common stock exercisable within 60 days of July 28,
2015. The address of Dr. Zeldis is c/o Soligenix, 29 Emmons Drive, Suite C-10, Princeton, New Jersey 08540. |
| |
|
| (10) |
Includes
12,955 shares of common stock, options to purchase 167,508 shares of common stock owned by Mr. Warusz exercisable within 60
days of July 28, 2015 and warrants to purchase 4,959 shares of common stock exercisable within 60 days of July 28, 2015. The
address of Mr. Warusz is c/o Soligenix, 29 Emmons Drive, Suite C-10, Princeton, New Jersey 08540. |
| |
|
| (11) |
Includes
options to purchase 84,375 shares of common stock exercisable within 60 days of July 28, 2015. The address of Dr. Straube
is c/o Soligenix, 29 Emmons Drive, Suite C-10, Princeton, New Jersey 08540. |
| |
|
| * |
Indicates
less than 1%. |
| |
|
| ** |
Beneficial
ownership is determined in accordance with the rules of the SEC. Shares of common stock subject to options or warrants currently
exercisable or exercisable within 60 days of July 28, 2015 are deemed outstanding for computing the percentage ownership of
the stockholder holding the options or warrants, but are not deemed outstanding for computing the percentage ownership of
any other stockholder. Percentage of ownership is based on 26,381,976 shares of common stock outstanding as of July 28, 2015. |
THE
EQUITY PURCHASE TRANSACTIONS
General
On
July 29, 2015, we entered into the Purchase Agreements and the Registration Rights Agreements with the Equity Purchasers. Pursuant
to the terms of the Purchase Agreements, the Equity Purchasers have agreed to purchase from us up to $10 million in the aggregate
worth of our common stock from time to time, until December 31, 2016. Pursuant to the terms of the Registration Rights Agreements,
we have filed with the SEC the registration statement that includes this prospectus to register for resale under the Securities
Act of 1933, as amended (the “Securities Act”) the shares that may be issued to the Equity Purchasers under the Purchase
Agreements.
We
do not have the right to commence any sales to the Equity Purchasers under the Purchase Agreements until the SEC has declared
effective the registration statement of which this prospectus forms a part. Thereafter, we may, from time to time and at our sole
discretion, direct the Equity Purchasers to purchase shares of our common stock. The purchase price per share will be equal to
80% of the lowest daily volume weighted average price of the common stock for the five consecutive trading days immediately following
our request for the Equity Purchasers to purchase the shares. In consideration for entering into the Purchase Agreements, we issued
to each of the Equity Purchasers a promissory note having a principal amount equal to 3% of the total amount committed by it,
which are payable on April 15, 2016.
Purchase
of Shares Under the Purchase Agreements
Under
the Purchase Agreements, we may direct Kodiak Capital, Kingsbrook and River North to purchase, on a pro rata bases, up to $5 million,
$4 million and $1 million of shares of our common stock, respectively. The closing of the sale of the shares will occur on the
sixth trading day following our request for the Equity Purchasers to purchase the shares. The purchase price per share will be
equal to 80% of the lowest daily volume weighted average price of the common stock for the five consecutive trading days immediately
following our request for the Equity Purchasers to purchase the shares. There is no minimum amount that we may require the Equity
Purchasers to purchase at any one time. We may not require the Equity Purchasers to purchase more than $3 million worth of shares
of common stock during any seven day period.
Other
than as set forth above, there are no trading volume requirements or restrictions under the Purchase Agreements, and we will control
the timing and amount of any sales of our common stock to the Equity Purchasers.
Conditions
to Sales
Under
the Purchase Agreements, the following conditions must be satisfied in order for us to sell shares of our common stock to the
Equity Purchasers:
| |
● |
The registration statement of which
this prospectus forms a part, and any amendment or supplement thereto, must be effective for the sale by the Equity Purchaser
of the shares to be purchased by the Equity Purchaser, and (i) neither we nor the Equity Purchaser have received notice that
the SEC has issued or intends to issue a stop order with respect to the registration statement or that the SEC otherwise has
suspended or withdrawn the effectiveness of the registration statement, either temporarily or permanently, or intends or has
threatened to do so and (ii) there is no other suspension of the use or withdrawal of the effectiveness of the registration
statement or this prospectus. |
| |
|
|
| |
● |
Our representations and warranties
contained in the Purchase Agreements must be true and correct in all material respects (except for representations and warranties
specifically made as of a particular date), except for any conditions that have temporarily caused any representations or
warranties to be incorrect and which have been corrected with no continuing impairment to us or the Equity Purchaser. |
| |
|
|
| |
● |
We must have performed in all material
respects all covenants, agreements and conditions required by the Purchase Agreements to be performed, satisfied or complied
with by us. |
| |
|
|
| |
● |
No statute, rule, regulation, executive
order, decree, ruling or injunction has been enacted, entered, promulgated or adopted by any court or governmental authority
of competent jurisdiction that prohibits or directly and materially adversely affects any of the transactions contemplated
by the Purchase Agreements, and no proceeding has been commenced that may have the effect of prohibiting or materially adversely
affecting any of the transactions contemplated by the Purchase Agreements. |
| |
|
|
| |
● |
The trading of our common stock has not been suspended by the SEC,
the principal trading market for our common stock or Financial Industry Regulatory Authority, Inc. and our common stock has
been approved for listing or quotation on and has not been delisted from such principal market. |
| |
|
|
| |
● |
The number of shares of our common
stock to be purchased by the Equity Purchaser at a particular closing may not exceed the number of shares that, when aggregated
with all other shares of common stock then beneficially owned by such Equity Purchaser, would result in the Equity Purchaser
owning more than 9.99% of all of our outstanding common stock. |
| |
|
|
| |
● |
We must have no knowledge of any event
more likely than not to have the effect of causing the registration statement of which this prospectus forms a part to be
suspended or otherwise ineffective. |
Our
Termination Rights
We
have the unconditional right, at any time, for any reason and without any payment or liability to us, to give notice to the Equity
Purchasers to terminate the Purchase Agreements.
No
Short-Selling by the Equity Purchasers
The
Equity Purchasers have agreed that neither they nor any of their respective affiliates shall engage in any direct or indirect
short-selling of our common stock during any time prior to the termination of the Purchase Agreements.
Effect
of Performance of the Purchase Agreements on Our Stockholders
All
shares of common stock registered in this offering are expected to be freely tradable. It is anticipated that shares registered
in this offering will be sold over a period commencing on the date that the registration statement including this prospectus becomes
effective through December 31, 2016. The sale by the Equity Purchasers of a significant amount of shares registered in this offering
at any given time could cause the market price of our common stock to decline and to be highly volatile. The Equity Purchasers
may ultimately purchase all, some or none of the shares of common stock not yet issued but registered in this offering. If we
sell these shares to the Equity Purchasers, the Equity Purchasers may sell all, some or none of such shares. Therefore, sales
to the Equity Purchasers by us under the Purchase Agreements may result in substantial dilution to the interests of other holders
of our common stock. In addition, if we sell a substantial number of shares to the Equity Purchasers under the Purchase Agreements,
or if investors expect that we will do so, the actual sales of shares or the mere existence of our arrangement with the Equity
Purchasers may make it more difficult for us to sell equity or equity-related securities in the future at a time and at a price
that we might otherwise wish to effect such sales. However, we have the right to control the timing and amount of any sales of
our shares to the Equity Purchasers and the Purchase Agreements may be terminated by us at any time at our discretion without
any cost to us.
Pursuant
to the terms of the Purchase Agreements, we have the right, but not the obligation, to direct Kodiak Capital, Kingsbrook and River
North to purchase up to $5 million, $4 million and $1 million of our common stock, respectively. Depending on the price per share
at which we sell our common stock to the Equity Purchasers, we may be authorized to issue and sell to the Equity Purchasers under
the Purchase Agreements more shares of our common stock than are offered under this prospectus. If we choose to do so, we must
first register for resale under the Securities Act any such additional shares, which could cause additional substantial dilution
to our stockholders. The number of shares ultimately offered for resale by the Equity Purchasers under this prospectus is dependent
upon the number of shares we direct the Equity Purchasers to purchase under the Purchase Agreements.
The
following table sets forth the amount of proceeds we would receive from the Equity Purchasers from the sale of shares at varying
purchase prices:
Assumed Average Purchase Price Per Share | | |
Number of Registered Shares to be Issued if Full Purchase (1) | | |
Percentage of Outstanding Shares After Giving Effect to the Issuance (2) | | |
Additional Proceeds from the Sale of Registered Shares Under the Purchase Agreement | |
| $ | 0.50 | | |
| 7,627,120 | | |
| 22.43 | % | |
$ | 3,813,560 | |
| $ | 1.00 | | |
| 7,627,120 | | |
| 22.43 | % | |
$ | 7,627,120 | |
| $ | 1.376 | (3) | |
| 7,267,441 | | |
| 21.60 | % | |
$ | 10,000,000 | |
| $ | 2.00 | | |
| 5,000,000 | | |
| 15.93 | % | |
$ | 10,000,000 | |
| $ | 2.50 | | |
| 4,000,000 | | |
| 13.17 | % | |
$ | 10,000,000 | |
| (1)
|
Although
the Purchase Agreements with the Equity Purchasers provides that we may sell up $10 million of our common stock to the Equity
Purchasers in the aggregate, we are only registering 7,627,120 shares for resale by the Equity Purchasers under this prospectus,
which may or may not cover all the shares we ultimately sell to the Equity Purchasers under the Purchase Agreements, depending
on the purchase price per share. As a result, we have included in this column only those shares that we are registering in
this offering. |
| |
|
| (2)
|
The
denominator is based on 26,381,976 shares outstanding as of July 28, 2015, and is adjusted to include the number of shares
set forth in the adjacent column which we would have sold to Kodiak Capital at the applicable assumed purchase price per share.
The numerator is based on the number of shares issuable under the Purchase Agreements at the corresponding assumed purchase
price set forth in the adjacent column. The number of shares in such column does not include shares that may be issued to
the Equity Purchasers under the Purchase Agreements which are not registered in this offering. |
| |
|
| (3)
|
$1.376
is 80% of the closing price of the common stock on July 28, 2015. |
SELLING
STOCKHOLDERS
This
prospectus relates to the possible resale by the selling stockholders, including 7,627,120 shares of common stock that may be
issued to the Equity Purchasers pursuant to the Purchase Agreements. We are filing the registration statement of which this prospectus
forms a part pursuant to the provisions of the agreements executed in connection with the selling stockholders’ agreement
to purchase the shares.
Pursuant
to the Registration Rights Agreements, which we entered into with the Equity Purchasers on July 29, 2015 concurrently with our
execution of the Purchase Agreements, we agreed to provide certain registration rights with respect to sales by the Equity Purchasers
of the shares of our common stock that may be issued to the Equity Purchasers under the Purchase Agreements. See the description
under the heading “The Equity Purchase Transactions” for more information about the Purchase Agreements.
On
April 27, 2013, we entered into the Channel Agreement with Intrexon to use Intrexon’s advanced human antibody discovery,
isolation and production technologies for the development of human monoclonal antibody therapies for a biodefense application
targeting melioidosis. The Channel Agreement granted us an exclusive worldwide license to use specified patents and other intellectual
property of Intrexon in connection with the research, development, use, importing, manufacture, sale and offer for sale of products
for the treatment of melioidosis through the use of exogenously produced human recombinant monoclonal antibodies. The Channel
Agreement, upon clinical or commercialization success, may require the payment of certain milestones payments up to $7 million,
if and when achieved. Also on April 27, 2013, we entered into a stock issuance agreement (the “Stock Issuance Agreement”)
with Intrexon, pursuant to which we issued to Intrexon 1,034,483 shares of common stock in consideration for the execution and
delivery of the Channel Agreement. Pursuant to the Stock Issuance Agreement, we agreed to provide certain registration rights
with respect to sales by Intrexon of the shares of our common stock. Additionally, as part of our June 2013 public offering, we
sold to NRM VII Holdings I, LLC, an affiliate of Intrexon, 3,333,333 shares of common stock and a warrant to purchase 2,500,000
shares of common stock for an aggregate purchase price of approximately $3.5 million.
The
selling stockholders, may, from time to time, offer and sell pursuant to this prospectus any or all of the shares that we have
sold or may sell to them. The selling stockholders may sell some, all or none of their shares. We do not know how long the selling
stockholders will hold the shares before selling them, and we currently have no agreements, arrangements or understandings with
the selling stockholders regarding the sale of any of the shares.
The
following table presents information regarding the selling stockholders and the shares that they may offer and sell from time
to time under this prospectus. The table is prepared based on information supplied to us by the selling stockholders, and reflects
their holdings as of July 28, 2015. Except as described herein, none of the selling stockholders nor any of their respective affiliates
has held a position or office, or had any other material relationship, with us or any of our predecessors or affiliates. As used
in this prospectus, the term “selling stockholders” includes the selling stockholders and any of their respective
donees, pledgees, transferees or other successors in interest selling shares received after the date of this prospectus from the
selling stockholders as a gift, pledge or other non-sale related transfer. Beneficial ownership is determined in accordance with
Rule 13d-3(d) promulgated by the SEC under the Exchange Act. The percentage of shares beneficially owned prior to the offering
is based on 26,381,976 shares of our common stock actually outstanding as of July 28, 2015.
| Selling stockholders | |
Shares Beneficially Owned Before this Offering | | |
Percentage of Outstanding Shares Beneficially Owned Before this Offering (5) | | |
Shares to be Sold in this Offering | | |
Number Of Shares Beneficially Owned After this Offering | | |
Percentage of Outstanding Shares Beneficially Owned After this Offering | |
| Kodiak Capital Group, LLC (1) | |
| -- | | |
| * | | |
| 3,389,831 | (6) | |
| — | | |
| * | |
| Kingsbrook Opportunities Master Fund LP (2) | |
| -- | | |
| * | | |
| 3,389,831 | (7) | |
| — | | |
| * | |
| River North Equity, LLC (3) | |
| -- | | |
| * | | |
| 847,458 | (8) | |
| — | | |
| * | |
| Intrexon Corporation (4) | |
| 1,034,483 | | |
| 3.92 | % | |
| 1,034,483 | | |
| — | | |
| * | |
| (1) |
Ryan
Hodson exercises voting and dispositive power with respect to the shares of our common stock being offered under this prospectus
by Kodiak Capital. |
| |
|
| (2) |
Ari
Storch, Adam J. Chill and Scott Wallace, in their capacities as managing members of the general partner of Kingsbrook, exercise
voting and dispositive power with respect to the shares of our common stock being offered under this prospectus by Kingsbrook. |
| |
|
| (3) |
Edward
M. Liceaga exercises voting and dispositive power with respect to the shares of our common stock being offered under this
prospectus by River North. |
| |
|
| (4) |
Randall
J. Kirk exercises voting and dispositive power with respect to the shares of our common stock being offered under this prospectus
by Intrexon. |
| (5)
|
Based
on 26,381,976 outstanding shares of our common stock as of July 28, 2015. Although we may at our discretion elect to issue
to the Equity Purchasers up to an aggregate amount of $10 million of our common stock under the Purchase Agreements, such
shares are not included in determining the percentage of shares beneficially owned before this offering. |
| (6)
|
Assumes
issuance of the maximum 3,389,831 shares being registered hereby. |
| (7)
|
Assumes
issuance of the maximum 3,389,831 shares being registered hereby. |
| (8)
|
Assumes
issuance of the maximum 847,458 shares being registered hereby. |
PLAN
OF DISTRIBUTION
The
Equity Purchasers are “underwriters,” and the other selling stockholders may be deemed to be an “underwriter,”
within the meaning of the Securities Act. The selling stockholders and any of their respective pledgees, donees, assignees and
successors-in-interest may, from time to time, sell any or all of their shares of common stock being offered under this prospectus
on any stock exchange, market or trading facility on which shares of our common stock are traded or in private transactions. These
sales may be at fixed or negotiated prices. The selling stockholders may use any one or more of the following methods when disposing
of shares:
| |
● |
ordinary
brokerage transactions and transactions in which the broker-dealer solicits purchasers; |
| |
|
|
| |
● |
block trades in
which the broker-dealer will attempt to sell the shares as agent but may position and resell a portion of the block as principal
to facilitate the transaction; |
| |
|
|
| |
● |
purchases by a
broker-dealer as principal and resales by the broker-dealer for its account; |
| |
|
|
| |
● |
an exchange distribution
in accordance with the rules of the applicable exchange; |
| |
|
|
| |
● |
privately negotiated
transactions; |
| |
|
|
| |
● |
broker-dealers
may agree with the selling stockholder to sell a specified number of such shares at a stipulated price per share; |
| |
|
|
| |
● |
a combination
of any of these methods of sale; and |
| |
|
|
| |
● |
any other method
permitted pursuant to applicable law. |
The
shares may also be sold under Rule 144 under the Securities Act, if available, rather than under this prospectus. The selling
stockholders have the sole and absolute discretion not to accept any purchase offer or make any sale of shares if such selling
stockholder deems the purchase price to be unsatisfactory at any particular time.
The
selling stockholders may pledge their respective shares to their brokers under the margin provisions of customer agreements. If
any selling stockholder defaults on a margin loan, the broker may, from time to time, offer and sell the pledged shares.
After
the effective date of the registration statement, the selling stockholders, other than the Equity Purchasers, may engage in short
sales against the box, puts and calls and other transactions in our securities or derivatives of our securities and may sell or
deliver shares in connection with these trades. The Equity Purchasers have agreed not to engage in any direct or indirect short
selling of our common stock during the term of the Purchase Agreements.
Broker-dealers
engaged by a selling stockholder may arrange for other broker-dealers to participate in sales. Broker-dealers may receive commissions
or discounts from a selling stockholder (or, if any broker-dealer acts as agent for the purchaser of shares, from the purchaser)
in amounts to be negotiated, which commissions as to a particular broker or dealer may be in excess of customary commissions to
the extent permitted by applicable law.
If
sales of shares offered under this prospectus are made to broker-dealers as principals, we would be required to file a post-effective
amendment to the registration statement of which this prospectus is a part. In the post-effective amendment, we would be required
to disclose the names of any participating broker-dealers and the compensation arrangements relating to such sales.
The
Equity Purchasers are “underwriters,” and the other selling stockholders and any broker-dealers or agents that are
involved in selling the shares offered under this prospectus may be deemed to be "underwriters," within the meaning
of the Securities Act in connection with these sales. Commissions received by these broker-dealers or agents and any profit on
the resale of the shares purchased by them may be deemed to be underwriting commissions or discounts under the Securities Act.
Any broker-dealers or agents that are deemed to be underwriters may not sell shares offered under this prospectus unless and until
we set forth the names of the underwriters and the material details of their underwriting arrangements in a supplement to this
prospectus or, if required, in a replacement prospectus included in a post-effective amendment to the registration statement of
which this prospectus is a part.
The
selling stockholders and any other persons participating in the sale or distribution of the shares offered under this prospectus
will be subject to applicable provisions of the Exchange Act and the rules and regulations under that act, including Regulation
M. These provisions may restrict activities of and limit the timing of purchases and sales of any of the shares by the selling
stockholders or any other person. Furthermore, under Regulation M, persons engaged in a distribution of securities are prohibited
from simultaneously engaging in market making and other activities with respect to those securities for a specified period of
time prior to the commencement of such distributions, subject to specified exceptions or exemptions. All of these limitations
may affect the marketability of the shares.
If
any of the shares of common stock offered for sale pursuant to this prospectus are transferred other than pursuant to a sale under
this prospectus, then subsequent holders could not use this prospectus until a post-effective amendment or prospectus supplement
is filed, naming such holders. We offer no assurance as to whether the selling stockholders will sell all or any portion of the
shares offered under this prospectus.
We
have agreed to pay all fees and expenses we incur incident to the registration of the shares being offered under this prospectus.
However, each selling security holder and purchaser are responsible for paying any discounts, commissions and similar selling
expenses they incur.
The
selling stockholders and the issuer have agreed to indemnify one another against certain losses, damages and liabilities arising
in connection with this prospectus, including liabilities under the Securities Act. Under the securities laws of certain states,
the shares of common stock may be sold in such states only through registered or licensed brokers or dealers. The selling stockholders
are advised to ensure that any brokers, dealers or agents effecting transactions on behalf of the selling stockholders are registered
to sell securities in all fifty states. In addition, in certain states the shares of common stock may not be sold unless the shares
have been registered or qualified for sale in such state or an exemption from registration or qualification is available and is
complied with.
We
will pay all the expenses incident to the registration, offering and sale of the shares of common stock to the public hereunder
other than commissions, fees and discounts of brokers, dealers and agents. We estimate that the expenses of the offering to be
borne by us will be approximately $78,500. The estimated offering expenses consist of: an SEC registration fee of $1,737, accounting
fees of $10,000, legal fees of $65,000 and miscellaneous expenses of $1,763. We will not receive any proceeds from the sale of
any of the shares of common stock by the selling stockholders.
The
selling stockholders should be aware that the anti-manipulation provisions of Regulation M under the Exchange Act will apply to
purchases and sales of shares of common stock by the selling stockholders, and that there are restrictions on market-making activities
by persons engaged in the distribution of the shares. Under Regulation M, the selling stockholders or its agents may not bid for,
purchase, or attempt to induce any person to bid for or purchase, shares of our common stock of while such selling stockholders
is distributing shares covered by this prospectus. The selling stockholders is advised that if a particular offer of common stock
is to be made on terms constituting a material change from the information set forth above with respect to this Plan of Distribution,
then, to the extent required, a post-effective amendment to the accompanying registration statement must be filed with the SEC.
Blue
Sky Restrictions on Resale
If
a selling stockholder wants to sell shares of our common stock under this registration statement in the United States, the selling
stockholder will also need to comply with state securities laws, also known as “Blue Sky laws,” with regard to secondary
sales. All states offer a variety of exemption from registration for secondary sales. Many states, for example, have an exemption
for secondary trading of securities registered under Section 12(g) of the Exchange Act or for securities of issuers that publish
continuous disclosure of financial and non-financial information in a recognized securities manual, such as Standard & Poor’s.
The broker for the selling stockholder will be able to advise the selling stockholder which states our common stock is exempt
from registration with that state for secondary sales.
Any
person who purchases shares of our common stock from a selling stockholder under this registration statement who then wants to
sell such shares will also have to comply with Blue Sky laws regarding secondary sales.
When
the registration statement becomes effective, and the selling stockholders indicate in which state(s) they desire to sell their
shares, we will be able to identify whether it will need to register or will rely on an exemption therefrom.
DESCRIPTION
OF CAPITAL STOCK
Our
authorized capital stock consists of 50,350,000 shares of capital stock, of which 50,000,000 shares are common stock, par value
$0.001 per share, 230,000 shares are preferred stock, 10,000 shares are Series B Convertible Preferred Stock, par value $0.05
per share (none of which are currently outstanding), 10,000 shares are Series C Convertible Preferred Stock, par value $0.05 per
share (none of which are currently outstanding) and 100,000 shares are Series A Junior Participating Preferred Stock, par value
$0.001 per share (which are available for issuance under our shareholder rights plan). As of July 28, 2015, there were issued
and outstanding 26,381,976 shares of common stock, options to purchase 2,338,237 shares of common stock and warrants to purchase
4,941,119 shares of common stock.
Common
Stock
Holders
of our common stock are entitled to one vote for each share held in the election of directors and in all other matters to be voted
on by the stockholders. There is no cumulative voting in the election of directors. Holders of common stock are entitled to receive
dividends as may be declared from time to time by our board of directors out of funds legally available therefor. In the event
of liquidation, dissolution or winding up of the corporation, holders of common stock are to share in all assets remaining after
the payment of liabilities. Holders of common stock have no pre-emptive or conversion rights and are not subject to further calls
or assessments. There are no redemption or sinking fund provisions applicable to the common stock. The rights of the holders of
the common stock are subject to any rights that may be fixed for holders of preferred stock. All of the outstanding shares of
common stock are fully paid and non-assessable.
Preferred
Stock
Our
Certificate of Incorporation authorizes the issuance of 230,000 shares of preferred stock, 10,000 shares of Series B Convertible
Preferred Stock, par value $0.05 per share (“Series B Preferred Stock”), 10,000 shares of Series C Convertible Preferred
Stock, par value $0.05 per share (“Series C Preferred Stock”), and 100,000 shares of Series A Junior Participating
Preferred Stock, par value $0.001 per share (“Junior Preferred Stock”). The board of directors is empowered, without
stockholder approval, to designate and issue additional series of preferred stock with dividend, liquidation, conversion, voting
or other rights, including the right to issue convertible securities with no limitations on conversion, which could adversely
affect the voting power or other rights of the holders of our common stock, substantially dilute a common stockholder’s
interest and depress the price of our common stock.
No
shares of the Series B Preferred Stock, the Series C Preferred Stock or the Junior Preferred Stock are outstanding. Due to the
terms of the Series C Preferred Stock, no additional shares of Series C Preferred Stock can be issued.
Series
B Preferred Stock
Our
Board of Directors has authorized the issuance of 10,000 shares of Series B Preferred Stock, 6,411 of which have been converted
to common stock and therefore are not reissuable.
Voting
Each
holder of Series B Preferred Stock is entitled to the number of votes equal to the number of whole shares of common stock into
which the shares of Series Preferred Stock held by such holder is then convertible (as adjusted from time to time pursuant to
the Certificate of Incorporation) with respect to any and all matters presented to the stockholders for their action or consideration.
Except as provided by law, holders of Series B Preferred Stock vote together with the holders of common stock as a single class.
Dividends
The
holders of the Series B Preferred Stock are entitled to a dividend of 8% per annum, payable annually in shares of Series B Preferred
Stock. In addition, when and if the Board of Directors shall declare a dividend payable with respect to the then outstanding shares
of common stock, the holders of the Series B Preferred Stock are entitled to the amount of dividends per share as would be payable
on the largest number of whole shares of common stock into which each share of Series B Preferred Stock could then be converted.
Conversion
Each
share of Series B Cumulative Convertible Preferred is convertible into 13.33 shares of common stock. The conversion ratio is subject
to an adjustment upon the issuance of additional shares of common stock for a price below the closing price of the common stock
and equitable adjustment for stock splits, dividends, combinations, reorganizations and similar events.
Liquidation
In
the event of liquidation, dissolution or winding up of the company, the holders of Series B Preferred Stock then outstanding will
be entitled to be paid an amount equal to $100 per share (subject to adjustment in the event of any stock dividend, stock split,
combination or other similar recapitalization affecting such shares pursuant to the Certificate of Incorporation), plus any dividends
declared but unpaid thereon before any payment is made to the holders of common stock, Junior Preferred Stock or any other class
or series of stock ranking on liquidation junior to the Series B Preferred Stock. After the holders of the Series B Preferred
Stock have been paid in full, the remaining assets of the company will be distributed to the holders of Junior Preferred Stock
and common stock, subject to the preferences of the Junior Preferred Stock.
Redemption
Subject
to certain conditions, after the second anniversary of the issuance of the Series B Preferred Stock, the company will have the
right, but not the obligation, to redeem the then-outstanding shares of Series B Preferred Stock for cash in an amount calculated
pursuant to the terms of the Certificate of Incorporation.
Junior
Preferred Stock
Voting
The
holders of the Junior Preferred Stock will have 1,000 votes per share of Junior Preferred Stock on all matters submitted to a
vote of our stockholders, including the election of directors.
Dividends
If
the Board of Directors declares or pays dividends on common stock, the holders of the Junior Preferred Stock would be entitled
to receive a per share dividend payment of 1,000 times the dividend declared per share of common stock. In the event we make a
distribution on the common stock, the holders of the Junior Preferred Stock will be entitled to a per share distribution, in like
kind, of 1,000 times such distribution made per share of common stock. In the event of any merger, consolidation or other transaction
in which shares of common stock are exchanged, each share of Junior Preferred Stock will be entitled to receive 1,000 times the
amount received per share of common stock. These rights are protected by customary anti-dilution provisions.
Liquidation
Upon
any liquidation, dissolution or winding up, no distribution may be made to the holders of shares of stock ranking junior to the
Junior Preferred Stock unless the holders of the Junior Preferred Stock have received the greater of (i) $3.70 per one one-thousandth
share plus an amount equal to accrued and unpaid dividends and distributions thereon, and (ii) an amount equal to 1,000 times
the aggregate amount to be distributed per share to holders of common stock. Further, no distribution may be made to the holders
of stock ranking on a parity upon liquidation, dissolution or winding up with the Junior Preferred Stock, unless distributions
are made ratably on the Junior Preferred Stock and all other shares of such parity stock in proportion to the total amounts to
which the holders of the Junior Preferred Stock are entitled above and to which the holders of such parity shares are entitled.
Shareholder
Rights Plan
On
June 22, 2007, our board of directors adopted a shareholder rights plan for our company and in connection therewith declared a
dividend of one preferred share purchase right for each outstanding share of common stock. Each Right entitles the registered
holder to purchase one one-thousandth of a share of our Junior Preferred Stock at a price of $3.70 per one one-thousandth of a
share, subject to certain adjustments. Initially, the rights are not exercisable, but become exercisable upon the earlier of (i)
10 days following a public announcement that a person or group of affiliated or associated persons, with certain exceptions, has
acquired beneficial ownership of 15% or more of the then outstanding common stock or (ii) 10 business days following the commencement
of, or announcement of an intention to make, a tender offer or exchange offer the consummation of which would result in the beneficial
ownership by a person or group of 15% or more of such outstanding common stock.
Our
board may redeem all of the rights for $0.001 per right at any time before the earlier of (i) the time the rights become exercisable
or (ii) July 1, 2017, the date the rights expire.
Anti-Takeover
Provisions
Provisions
in our Certificate of Incorporation, by-laws and stockholder rights plan may discourage certain types of transactions involving
an actual or potential change of control of our company which might be beneficial to us or our security holders.
As
noted above, our Certificate of Incorporation permits our board of directors to issue shares of any class or series of preferred
stock in the future without stockholder approval and upon such terms as our board of directors may determine. The rights of the
holders of common stock will be subject to, and may be adversely affected by, the rights of the holders of any class or series
of preferred stock that may be issued in the future.
Our
bylaws generally provide that any board vacancy, including a vacancy resulting from an increase in the authorized number of directors,
may be filled by a majority of the directors, even if less than a quorum.
Additionally,
our bylaws provide that stockholders must provide timely notice in writing to bring business before an annual meeting of shareholders
or to nominate candidates for election as directors at an annual meeting of shareholders. Notice for an annual meeting is timely
if our Secretary receives the written notice not less than 45 days and no more than 75 days prior to the anniversary of the date
that we mailed proxy materials for the preceding year’s annual meeting. However, if the date of the annual meeting is advanced
more than thirty (30) days prior to, or delayed by more than thirty (30) days after, the anniversary of the preceding year’s
annual meeting, notice by the stockholder to be timely must be delivered not later than the close of business on the later of
(i) the 90th day prior to such annual meeting or (ii) the 10th day following the day on which public announcement
of the date of such annual meeting is first made. Our bylaws also specify the form and content of a shareholder’s notice.
These provisions may prevent shareholders from bringing matters before an annual meeting of shareholders or from making nominations
for directors at an annual meeting of shareholders.
Warrants
On
June 25, 2013, we consummated a public offering of an aggregate of 6,773,995 shares of common stock, together with warrants to
purchase up to 5,080,500 shares of common stock. In connection with the offering, we also issued the placement agent a warrant
to purchase up to 336,081 shares of common stock. We refer to the warrants issued to the investors and the placement agent in
connection with the offering as the “2013 Warrants.”
As
a result of exercises, 3,036,928 shares of common stock remain issuable upon the exercise of the 2013 Warrants as of July 28,
2015. The 2013 Warrants expire in June 2018.
As
of July 28, 2015, the 2013 Warrants were exercisable to purchase shares of common stock a $0.61. per share. The 2013 Warrants
include a price protection provision pursuant to which, in the event, and on each such occasion on or before the expiration of
the 2013 Warrants, we issue any shares of common stock or convertible securities (other than shares issued or issuable in certain
transactions, including upon exercise of employee stock options, upon conversion or exercise of currently-outstanding convertible
securities, or in connection with acquisitions or strategic transactions) at a price less than the then current exercise price
(a “Dilutive Issuance”), the exercise price of the 2013 Warrants will automatically be reduced to a price equal to
the price at which such shares were issued and sold in the Dilutive Issuance. Additionally, the exercise price and the number
of shares of common stock purchasable upon the exercise of each 2013 Warrant are subject to adjustment upon the happening of certain
events, such as stock dividends, distributions, and splits.
On
December 24, 2014, we consummated a public offering of an aggregate of 1,886,530 shares of common stock, together with warrants
to purchase up to 1,131,918 shares of common stock. In connection with the offering, we also issued the underwriter a warrant
to purchase up to 37,400 shares of common stock. We refer to the warrants issued to the investors and the placement agent in connection
with the offering as the “2014 Warrants.”
As
of July 28, 2015, 1,109,318 shares of common stock remain issuable upon the exercise of the 2014 Warrants, which expire in 2019.
As
of July 28, 2015, we also had other outstanding warrants to purchase 794,873 shares of common stock, all of which are exercisable
at a weighted average exercise price of approximately $0.65 per share.
DISCLOSURE
OF COMMISSION POSITION ON INDEMNIFICATION
FOR
SECURITIES ACT LIABILITIES
Section
102(b)(7) of the Delaware General Corporation Law allows companies to limit the personal liability of its directors to the company
or its stockholders for monetary damages for breach of a fiduciary duty. Article IX of our Certificate of Incorporation, as amended,
provides for the limitation of personal liability of our directors as follows:
"A
Director of the Corporation shall have no personal liability to the Corporation or its stockholders for monetary damages for breach
of his fiduciary duty as a Director; provided, however, this Article shall not eliminate or limit the liability of a Director
(i) for any breach of the Director's duty of loyalty to the Corporation or its stockholders; (ii) for acts or omissions not in
good faith or which involve intentional misconduct or a knowing violation of law; (iii) for the unlawful payment of dividends
or unlawful stock repurchases under Section 174 of the General Corporation Law of the State of Delaware; or (iv) for any transaction
from which the Director derived an improper personal benefit. If the General Corporation Law is amended after approval by the
stockholders of this Article to authorize corporate action further eliminating or limiting the personal liability of directors,
then the liability of a director of the Corporation shall be eliminated or limited to the fullest extent permitted by the General
Corporation Law of the State of Delaware, as so amended."
Article
VIII of the our Bylaws, as amended and restated, provide for indemnification of directors and officers to the fullest extent permitted
by the Delaware General Corporation Law.
Insofar
as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers or persons
controlling the registrant pursuant to the foregoing provisions, the registrant has been informed that in the opinion of the SEC
such indemnification is against public policy as expressed in the Act and is therefore unenforceable.
LEGAL
MATTERS
The
validity of the shares of our common stock offered hereby will be passed upon by the law firm of Duane Morris LLP, Miami, Florida.
EXPERTS
The
consolidated balance sheets of Soligenix, Inc. as of December 31, 2014 and 2013 and the related consolidated statements of operations,
changes in shareholders’ deficiency, and cash flows for each of the years in the two-year period ended December 31, 2014,
have been audited by EisnerAmper LLP, independent registered public accounting firm, as stated in their report which is included
herein. Such financial statements have been included herein in reliance on the report of such firm given upon their authority
as experts in accounting and auditing.
WHERE
YOU CAN FIND MORE INFORMATION
We
have filed with the Securities and Exchange Commission, Washington, D.C. 20549, under the Securities Act of 1933, a registration
statement on Form S-1 relating to the shares offered hereby. This prospectus does not contain all of the information set forth
in the registration statement and the exhibits and schedules thereto. For further information with respect to our company and
the shares offered by this prospectus, you should refer to the registration statement, including the exhibits and schedules thereto.
You may inspect a copy of the registration statement without charge at the Public Reference Section of the Securities and Exchange
Commission at Room 1024, 450 Fifth Street, N.W., Washington, D.C. 20549. The public may obtain information on the operation of
the Public Reference Room by calling the Securities and Exchange Commission. The Securities and Exchange Commission also maintains
an Internet site that contains reports, proxy and information statements and other information regarding registrants that file
electronically with the Securities and Exchange Commission. The Securities and Exchange Commission’s World Wide Web address
is http://www.sec.gov.
Statements
contained in this prospectus as to the contents of any contract or other document that we have filed as an exhibit to the registration
statement are qualified in their entirety by reference to the exhibits for a complete statement of their terms and conditions.
The
representations, warranties and covenants made by us in any agreement that is filed as an exhibit to the registration statement
of which this prospectus is a part were made solely for the benefit of the parties to such agreement, including, in some cases,
for the purpose of allocating risk among the parties to such agreements, and should not be deemed to be a representation, warranty
or covenant to you. Moreover, such representations, warranties or covenants were made as of an earlier date. Accordingly, such
representations, warranties and covenants should not be relied on as accurately representing the current state of our affairs.
We
file periodic reports, proxy statements and other information with the Securities and Exchange Commission in accordance with requirements
of the Exchange Act. These periodic reports, proxy statements and other information are available for inspection and copying at
the regional offices, public reference facilities and Internet site of the Securities and Exchange Commission referred to above.
We make available through our website, free of charge, copies of these reports as soon as reasonably practicable after we electronically
file or furnish them to the Securities and Exchange Commission. Our website is located at http://www.soligenix.com. You can also
request copies of such documents, free of charge, by contacting the company at (609) 538-8200 or sending an email to info@soligenix.com.
Information
contained on our website is not a prospectus and does not constitute a part of this prospectus.
INDEX
TO FINANCIAL STATEMENTS
SOLIGENIX,
INC. AND SUBSIDIARIES
CONSOLIDATED
FINANCIAL STATEMENTS
Table
of Contents
| |
Page |
| Financial
Statements for the Quarter Ended March 31, 2015 |
|
| Consolidated
Balance Sheets as of March 31, 2015 (unaudited) and December 31, 2014 |
F-1 |
| Consolidated
Statements of Operations for the Three Months Ended March 31, 2015 and 2014 (unaudited) |
F-2 |
| Consolidated
Statements of Changes in Shareholders’ Deficiency for the Three Months Ended March 31, 2015 (unaudited) |
F-3 |
| Consolidated
Statements of Cash Flows for the Three Months Ended March 31, 2015 and 2014 (unaudited) |
F-4 |
| Notes
to Financial Statements |
F-5 |
| |
|
| Financial
Statements for the Year Ended December 31, 2014 |
|
| Consolidated
Balance Sheets as of December 31, 2014 and 2013 |
F-16 |
| Consolidated
Statements of Operations for the Years Ended December 31, 2014 and 2013 |
F-17 |
| Consolidated
Statements of Changes in Shareholders’ Deficiency for the Years Ended December 31, 2014 and 2013 |
F-18 |
| Consolidated
Statements of Cash Flows for the Years Ended December 31, 2014 and 2013 |
F-19 |
| Notes
to Consolidated Financial Statements |
F-20 |
| Report
of Independent Registered Public Accounting Firm |
F-35 |
Soligenix,
Inc. and Subsidiaries
Consolidated
Balance Sheets
| | |
March
31, 2015 | | |
December
31, 2014 | |
| | |
(Unaudited) | | |
| |
| Assets | |
| | |
| |
| Current
assets: | |
| | |
| |
| Cash
and cash equivalents | |
$ | 5,012,605 | | |
$ | 5,525,094 | |
| Contracts
and grants receivable | |
| 345,420 | | |
| 794,767 | |
| Prepaid
expenses | |
| 131,546 | | |
| 172,928 | |
| Total
current assets | |
| 5,489,571 | | |
| 6,492,789 | |
| | |
| | | |
| | |
| Office
furniture and equipment, net | |
| 57,176 | | |
| 51,510 | |
| Intangible
assets, net | |
| 355,910 | | |
| 409,949 | |
| Total
assets | |
$ | 5,902,657 | | |
$ | 6,954,248 | |
| | |
| | | |
| | |
| Liabilities
and shareholders’ deficiency | |
| | | |
| | |
| Current
liabilities: | |
| | | |
| | |
| Accounts
payable | |
$ | 2,543,942 | | |
$ | 3,003,545 | |
| Warrant
liability | |
| 5,152,367 | | |
| 3,789,562 | |
| Accrued
compensation | |
| 32,189 | | |
| 315,030 | |
| Total
current liabilities | |
| 7,728,498 | | |
| 7,108,137 | |
| | |
| | | |
| | |
| Commitments
and contingencies | |
| | | |
| | |
| Shareholders’
deficiency: | |
| | | |
| | |
| Preferred
stock; 350,000 shares authorized; none issued or outstanding | |
| - | | |
| - | |
| Common
stock, $.001 par value; 50,000,000 shares authorized; 25,339,364 shares and 23,936,568 shares issued and outstanding at March
31, 2015 and December 31, 2014, respectively | |
| 25,340 | | |
| 23,937 | |
| Additional
paid-in capital | |
| 141,764,490 | | |
| 138,868,523 | |
| Accumulated
deficit | |
| (143,615,671 | ) | |
| (139,046,349 | ) |
| Total
shareholders’ deficiency | |
| (1,825,841 | ) | |
| (153,889 | ) |
| Total
liabilities and shareholders’ deficiency | |
$ | 5,902,657 | | |
$ | 6,954,248 | |
The
accompanying notes are an integral part of these consolidated financial statements.
Soligenix,
Inc. and Subsidiaries
Consolidated
Statements of Operations
For
the Three Months Ended March 31, 2015 and 2014
(Unaudited)
| | |
Three
Months Ended March
31, | |
| | |
2015 | | |
2014 | |
| | |
| | |
| |
| Revenues: | |
| | |
| |
| Grant
revenue | |
$ | 103,880 | | |
$ | 244,290 | |
| Contract
revenue | |
| 712,406 | | |
| 666,307 | |
| | |
| | | |
| | |
| Total
revenues | |
| 816,286 | | |
| 910,597 | |
| Cost
of revenues | |
| (527,399 | ) | |
| (628,981 | ) |
| Gross
profit | |
| 288,887 | | |
| 281,616 | |
| | |
| | | |
| | |
| Operating
expenses: | |
| | | |
| | |
| Research
and development | |
| 1,029,884 | | |
| 1,030,621 | |
| General
and administrative | |
| 817,270 | | |
| 840,904 | |
| |
| | | |
| | |
| Total
operating expenses | |
| 1,847,154 | | |
| 1,871,525 | |
| | |
| | | |
| | |
| Loss
from operations | |
| (1,558,267 | ) | |
| (1,589,909 | ) |
| | |
| | | |
| | |
| Other
income (expense): | |
| | | |
| | |
| Change
in fair value of warrant liability | |
| (3,011,616 | ) | |
| (1,742,090 | ) |
| Interest
income | |
| 561 | | |
| 291 | |
| | |
| | | |
| | |
| Total
other income (expense) | |
| (3,011,055 | ) | |
| (1,741,799 | ) |
| | |
| | | |
| | |
| Net
loss | |
$ | (4,569,322 | ) | |
$ | (3,331,708 | ) |
| Basic
and diluted net loss per share | |
$ | (0.19 | ) | |
$ | (0.17 | ) |
| | |
| | | |
| | |
| Basic
and diluted weighted average common shares outstanding | |
| 24,405,813 | | |
| 19,739,470 | |
The
accompanying notes are an integral part of these consolidated financial statements.
Soligenix,
Inc. and Subsidiaries
Consolidated
Statement of Changes in Shareholders’ Deficiency
For
the Three Months Ended March 31, 2015
(Unaudited)
| | |
Common
Stock | | |
Additional
Paid-In | | |
Accumulated | | |
| |
| | |
Shares
| | |
Par
Value | | |
Capital
| | |
Deficit | | |
Total | |
| | |
| | |
| | |
| | |
| | |
| |
| Balance,
December 31, 2014 | |
| 23,936,568 | | |
$ | 23,937 | | |
$ | 138,868,523 | | |
$ | (139,046,349 | ) | |
$ | (153,889 | ) |
| Issuance
of common stock pursuant to Lincoln Park Equity Line | |
| 153,010 | | |
| 153 | | |
| 246,372 | | |
| - | | |
| 246,525 | |
| Issuance
of common stock to vendors | |
| 66,000 | | |
| 66 | | |
| 101,294 | | |
| - | | |
| 101,360 | |
| Reclassification
of warrant liability upon partial exercises of warrants issued in unit offering | |
| - | | |
| - | | |
| 1,648,811 | | |
| - | | |
| 1,648,811 | |
| Issuance
of common stock from exercises of warrants | |
| 1,183,786 | | |
| 1,184 | | |
| 757,465 | | |
| - | | |
| 758,649 | |
| Share-based
compensation expense | |
| - | | |
| - | | |
| 142,025 | | |
| - | | |
| 142,025 | |
| Net
loss | |
| - | | |
| - | | |
| - | | |
| (4,569,322 | ) | |
| (4,569,322 | ) |
| Balance,
March 31, 2015 | |
| 25,339,364 | | |
$ | 25,340 | | |
$ | 141,764,490 | | |
$ | (143,615,671 | ) | |
$ | (1,825,841 | ) |
The
accompanying notes are an integral part of these consolidated financial statements.
Soligenix,
Inc. and Subsidiaries
Consolidated
Statements of Cash Flows
For
the Three Months Ended March 31,
(Unaudited)
| | |
2015
| | |
2014 | |
| Operating
activities: | |
| | |
| |
| Net
loss | |
$ | (4,569,322 | ) | |
$ | (3,331,708 | ) |
| Adjustments
to reconcile net loss to net cash used in operating activities: | |
| | | |
| | |
| Amortization
and depreciation | |
| 59,926 | | |
| 62,087 | |
| Common
stock issued to vendors | |
| 101,360 | | |
| 153,540 | |
| Warrants
issued to vendors | |
| - | | |
| 4,775 | |
| Share-based
compensation | |
| 142,025 | | |
| 176,662 | |
| Change
in fair value of warrant liability | |
| 3,011,616 | | |
| 1,742,090 | |
| Change
in operating assets and liabilities: | |
| | | |
| | |
| Grants
receivable | |
| 449,347 | | |
| 172,740 | |
| Taxes
receivable | |
| - | | |
| 750,356 | |
| Prepaid
expenses | |
| 41,382 | | |
| 37,771 | |
| Accounts
payable | |
| (459,603 | ) | |
| 32,338 | |
| Accrued
compensation | |
| (282,841 | ) | |
| (188,430 | ) |
| Total
adjustments | |
| 3,063,212 | | |
| 2,943,929 | |
| Net
cash used in operating activities | |
| (1,506,110 | ) | |
| (387,779 | ) |
| | |
| | | |
| | |
| Investing
activities | |
| | | |
| | |
| Purchases
of fixed assets | |
| (11,553 | ) | |
| (19,863 | ) |
| Net
cash used in investing activities | |
| (11,553 | ) | |
| (19,863 | ) |
| | |
| | | |
| | |
| Financing
Activities: | |
| | | |
| | |
| Net
proceeds from issuance of common stock pursuant to the equity line | |
| 246,525 | | |
| 158,250 | |
| Proceeds
from exercises of warrants | |
| 758,649 | | |
| - | |
| Net
cash provided by financing activities | |
| 1,005,174 | | |
| 158,250 | |
| | |
| | | |
| | |
| Net
decrease in cash and cash equivalents | |
| (512,489 | ) | |
| (249,392 | ) |
| Cash
and cash equivalents at beginning of period | |
| 5,525,094 | | |
| 5,856,242 | |
| Cash
and cash equivalents at end of period | |
$ | 5,012,605 | | |
$ | 5,606,850 | |
| | |
| | | |
| | |
| Supplemental
disclosure of non cash investing and financing activities: | |
| | | |
| | |
| Reclassification
of warrant liability to additional paid in capital upon partial exercises of warrants issued in unit offering | |
$ | 1,648,811 | | |
$ | 502,025 | |
The
accompanying notes are an integral part of these consolidated financial statements.
Soligenix,
Inc.
Notes
to Consolidated Financial Statements
Note
1. Nature of Business
Basis
of Presentation
Soligenix,
Inc. (the “Company”) is a late-stage biopharmaceutical company developing product candidates intended to address unmet
medical needs in the areas of inflammation, oncology, and biodefense. The Company maintains two active business segments: BioTherapeutics
and Vaccines/BioDefense.
The
Company’s BioTherapeutics business segment is developing a first-in-class photo-dynamic therapy (SGX301) utilizing safe
visible light for the treatment of cutaneous T-cell lymphoma (“CTCL”), proprietary formulations of oral beclomethasone
17,21-dipropionate (“BDP”) for the prevention/treatment of gastrointestinal (“GI”) disorders characterized
by severe inflammation, including pediatric Crohn’s disease (SGX203) and acute radiation enteritis (SGX201), and its novel
innate defense regulator (“IDR”) technology (SGX942) for the treatment of oral mucositis.
The
Company’s Vaccines/BioDefense business segment includes active development programs for RiVax™, its ricin toxin vaccine
candidate, VeloThrax™, an anthrax vaccine candidate, and OrbeShield™, a GI acute radiation syndrome (“GI ARS”)
therapeutic candidate and SGX943, a melioidosis therapeutic candidate. The development of the vaccine programs is supported by
the Company’s heat stabilization technology, known as ThermoVax™, under existing and on-going government contract
funding. With the recently awarded government contracts from the National Institute of Allergy and Infectious Diseases (“NIAID”),
the Company will attempt to advance the development of RiVax™ to protect against exposure to ricin toxin. The Company plans
to use the funds received under the government contracts with the Biomedical Advanced Research and Development Authority (“BARDA”)
and NIAID to advance the development of OrbeShield™ for the treatment of GI ARS. Additionally, the Company entered into
a global and exclusive channel collaboration with Intrexon Corporation (“Intrexon”) through which it intends to develop
and commercialize a human monoclonal antibody therapy (SGX101) to treat melioidosis.
The
Company generates revenues under government grants primarily from the National Institutes of Health (the “NIH”) and
government contracts from BARDA and NIAID.
The Company is subject to risks common to companies in the biotechnology industry including,
but not limited to, development of new technological innovations, dependence on key personnel, protections of proprietary technology,
compliance with the United States Food and Drug Administration (the U.S. “FDA”) regulations, litigation, and product
liability. Results for the three months ended March 31, 2015 are not necessarily indicative of results that may be expected for
the full year.
Liquidity
As
of March 31, 2015, the Company had cash and cash equivalents of $5,012,605 as compared to $5,525,094 as of December 31, 2014,
representing a decrease of $512,489 or 9%. As of March 31, 2015, the Company had working capital of $2,913,440 as compared to
working capital of $3,174,214 as of December 31, 2014, which excludes a non-cash warrant liability of $5,152,367 and $3,789,562,
respectively, representing a decrease of $260,774 or 8%. This decrease is primarily related to expenditures to support the Phase
2 clinical trial with SGX942 for the treatment of oral mucositis in head and neck cancer.
Based
on the Company’s current rate of cash outflows, cash on hand, proceeds from its government contract and grant programs, availability
of funds from the Lincoln Park Capital Fund, LLC (“Lincoln Park”) equity line and proceeds from the state of New Jersey
Technology Business Tax Certificate Transfer Program, management believes that its current cash will be sufficient to meet the
anticipated cash needs for working capital and capital expenditures for at least the next twelve months.
Management’s
business strategy can be outlined as follows:
| ● | Conduct
a Phase 3 clinical trial of SGX301 for the treatment of CTCL; |
| ● | Conduct
a Phase 2 clinical trial of SGX942 for the treatment of oral mucositis in head and neck
cancer; |
| ● | Initiate
a Phase 3 clinical trial of oral BDP, known as SGX203, for the treatment of pediatric
Crohn’s disease; |
| ● | Evaluate
the effectiveness of oral BDP in other therapeutic indications involving inflammatory
conditions of the GI tract such as prevention of acute radiation enteritis; |
| ● | Develop
RiVax™ and VeloThrax™ in combination with the Company’s ThermoVax™
technology, to develop new heat stable vaccines in biodefense and infectious diseases
with the potential to collaborate and/or partner with other companies in these areas; |
| ● | Advance
the preclinical and manufacturing development of OrbeShield™ as a biodefense medical
countermeasure for the treatment of GI ARS; |
| ● | Continue
to apply for and secure additional government funding for each of the Company’s
BioTherapeutics and Vaccines/BioDefense programs through grants, contracts and/or procurements; |
| ● | Acquire
or in-license new clinical-stage compounds for development; and |
| ● | Explore
other business development and merger/acquisition strategies an example of which is the
collaboration with Intrexon. |
The
Company’s plans with respect to its liquidity management include, but are not limited to, the following:
| ● | The
Company has up to $50.5 million in active government contract and grant funding still
available to support its associated research programs through 2015 and beyond. The Company
plans to submit additional contract and grant applications for further support of its
programs with various funding agencies. |
| ● | The
Company has continued to use equity instruments to provide a portion of the compensation
due to vendors and collaboration partners and expects to continue to do so for the foreseeable
future. |
| ● | The
Company will pursue Net Operating Loss (“NOL”) sales in the state of New
Jersey pursuant to its Technology Business Tax Certificate Transfer Program. Based on
the receipt, in December 2014, of $616,872 in proceeds pursuant to NOL sales in 2014,
the Company expects to participate in the program during 2015 and beyond. |
| ● | The
Company has a $10.6 million equity facility, with Lincoln Park through October 2016,
of which approximately $9.3 million is available. |
| ● | The
Company may seek additional capital in the private and/or public equity markets to continue
its operations, respond to competitive pressures, develop new products and services,
and to support new strategic partnerships. The Company is currently evaluating additional
equity financing opportunities on an ongoing basis and may execute them when appropriate.
However, there can be no assurances that the Company can consummate such a transaction,
or consummate a transaction at favorable pricing. |
Note
2. Summary of Significant Accounting Policies
Principles
of Consolidation
The
consolidated financial statements include Soligenix, Inc., and its wholly and majority owned subsidiaries. All significant intercompany
accounts and transactions have been eliminated as a result of consolidation.
Operating
Segments
Operating
segments are defined as components of an enterprise about which separate financial information is available that is evaluated
on a regular basis by the chief operating decision maker, or decision making group, in deciding how to allocate resources to an
individual segment and in assessing the performance of the segment. The Company divides its operations into two operating segments:
BioTherapeutics and Vaccines/BioDefense.
Cash
and cash equivalents
The
Company considers all highly liquid investments with original maturities of three months or less to be cash equivalents.
Contracts
and Grants Receivable
Contracts
and grants receivable consist of unbilled amounts due from various grants from the NIH and contracts from BARDA and NIAID, an
institute of NIH, for costs incurred prior to the period end under reimbursement contracts. The amounts were billed to the respective
governmental agencies in the month subsequent to period end and collected shortly thereafter. Accordingly, no allowance for doubtful
amounts has been established. If amounts become uncollectible, they are charged to operations.
Intangible
Assets
One
of the most significant estimates or judgments that the Company makes is whether to capitalize or expense patent and license costs.
The Company makes this judgment based on whether the technology has alternative future uses, as defined in Financial Accounting
Standards Board (“FASB”) Accounting Standards Codification (“ASC”) 730, Research and Development.
Based on this consideration, the Company capitalizes payments made to legal firms that are engaged in filing and protecting rights
to intellectual property and rights for its current products in both the domestic and international markets. The Company believes
that patent rights are one of its most valuable assets. Patents and patent applications are a key component of intellectual property,
especially in the early stage of product development, as their purchase and maintenance gives the Company access to key product
development rights from Soligenix’s academic and industry partners. These rights can also be sold or sub-licensed as part
of its strategy to partner its products at each stage of development as the intangible assets have alternative future use. The
legal costs incurred for these patents consist of work associated with filing new patents and perhaps extending the lives of the
patents. The Company capitalizes such costs and amortizes intangibles on a straight-line basis over their expected useful life
– generally a period of 11 to 16 years.
The
Company did not capitalize any patent related costs during the three months ended March 31, 2015 and 2014.
Impairment
of Long-Lived Assets
Office
furniture and equipment and intangible assets are evaluated and reviewed for impairment whenever events or changes in circumstances
indicate that the carrying amount may not be recoverable. The Company recognizes impairment of long-lived assets in the event
the net book value of such assets exceeds the estimated future undiscounted cash flows attributable to such assets. If the sum
of the expected undiscounted cash flows is less than the carrying value of the related asset or group of assets, a loss is recognized
for the difference between the fair value and the carrying value of the related asset or group of assets. Such analyses necessarily
involve significant judgment.
The
Company did not record any impairment of long-lived assets for the three months ended March 31, 2015 and 2014.
Fair
Value of Financial Instruments
FASB
ASC 820 — Fair Value Measurements and Disclosures, defines fair value as the price that would be received to sell
an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. FASB ASC
820 requires disclosures about the fair value of all financial instruments, whether or not recognized, for financial statement
purposes. Disclosures about the fair value of financial instruments are based on pertinent information available to the Company
on March 31, 2015. Accordingly, the estimates presented in these financial statements are not necessarily indicative of the amounts
that could be realized on disposition of the financial instruments.
FASB
ASC 820 specifies a hierarchy of valuation techniques based on whether the inputs to those valuation techniques are observable
or unobservable. Observable inputs reflect market data obtained from independent sources, while unobservable inputs reflect market
assumptions. The hierarchy gives the highest priority to unadjusted quoted prices in active markets for identical assets or liabilities
(Level 1 measurement) and the lowest priority to unobservable inputs (Level 3 measurement).
The
three levels of the fair value hierarchy are as follows:
| ● | Level
1 — Quoted prices in active markets for identical assets or liabilities that the
reporting entity has the ability to access at the measurement date. Level 1 primarily
consists of financial instruments whose value is based on quoted market prices such as
exchange-traded instruments and listed equities. |
| ● | Level
2 — Inputs other than quoted prices included within Level 1 that are observable
for the asset or liability, either directly or indirectly. Level 2 includes financial
instruments that are valued using models or other valuation methodologies. These models
consider various assumptions, including volatility factors, current market prices and
contractual prices for the underlying financial instruments. Substantially all of these
assumptions are observable in the marketplace, can be derived from observable data or
are supported by observable levels at which transactions are executed in the marketplace. |
| ● | Level
3 — Unobservable inputs for the asset or liability. Financial instruments are considered
Level 3 when their fair values are determined using pricing models, discounted cash flows
or similar techniques and at least one significant model assumption or input is unobservable. |
The
carrying amounts reported in the consolidated balance sheet for cash and cash equivalents, accounts receivable, accounts payable
and accrued expenses approximate their fair value based on the short-term maturity of these instruments. The Company recognizes
all derivative financial instruments as assets or liabilities in the financial statements and measures them at fair value with
changes in fair value reflected as current period income or loss unless the derivatives qualify as hedges. As a result, certain
warrants issued in connection with the June 2013 offering were accounted for as derivatives. See Note 4, Warrant Liability.
Revenue
Recognition
The
Company’s revenues are primarily generated from government contracts and grants. Revenue is recognized in accordance with
FASB ASC 605, Revenue Recognition, and/or ASC 605-25 Revenue Recognition – Multiple Element Arrangements.
The revenue from government contracts and grants is based upon subcontractor costs and internal costs incurred that are specifically
covered by the contracts and grants, plus a facilities and administrative rate that provides funding for overhead expenses and
management fees. These revenues are recognized when expenses have been incurred by subcontractors or when the Company incurs reimbursable
internal expenses that are related to the government contracts and grants.
Research
and Development Costs
Research
and development costs are charged to expense when incurred in accordance with FASB ASC 730, Research and Development. Research
and development includes costs such as clinical trial expenses, contracted research and license agreement fees with no alternative
future use, supplies and materials, salaries, stock based compensation, employee benefits, equipment depreciation and allocation
of various corporate costs. Purchased in-process research and development expense represents the value assigned or paid for acquired
research and development for which there is no alternative future use as of the date of acquisition.
Accounting
for Warrants
The
Company considered FASB ASC 815, Evaluating Whether an Instrument is Considered Indexed to an Entity’s Own Stock,
which provides guidance for determining whether an equity-linked financial instrument (or embedded feature) issued by an entity
is indexed to the entity’s stock. The Company evaluated the warrants’ provisions and determined that warrants issued
in connection with the Company’s June 2013 registered public offering contain provisions that protect holders from a decline
in the issue price of the Company’s common stock (or “down-round” provisions) and contain net settlement provisions.
Consequently, these warrants are recognized as liabilities at their fair value on the date of grant and remeasured at fair value
on each reporting date. All other warrants issued were indexed to the Company’s stock and therefore are accounted for as
equity instruments for 2015 and 2014.
Share-Based
Compensation
Stock
options are issued with an exercise price equal to the market price on the date of grant. Stock options issued to directors upon
re-election vest quarterly for a period of one year (new director issuances are fully vested upon issuance). Stock options issued
to employees vest 25% on the grant date, then 25% each subsequent year for a period of three years. Stock options vest over each
three-month period from the date of issuance to the end of the three year period. These options have a ten year life for as long
as the individuals remain employees or directors. In general, when an employee or director terminates their position, the options
will expire within three months, unless otherwise extended by the Board.
From time to time, the Company issues
restricted shares of common stock to vendors and consultants as compensation for services performed. Typically these instruments
vest upon issuance and therefore the entire share-based compensation expense is recognized upon issuance to the vendors and/or
consultants.
Share-based compensation expense for
options, warrants and shares of common stock granted to non-employees has been determined in accordance with FASB ASC 718, Stock
Compensation, and FASB ASC 505-50, Equity-Based Payments to Non-Employees, and represents the fair value of the consideration
received, or the fair value of the equity instruments issued, whichever may be more reliably measured. For options that vest over
future periods, the fair value of options granted to non-employees is amortized as the options vest.
The
fair value of options issued during the three months ended March 31, 2015 and 2014 was estimated using the Black-Scholes option-pricing
model and the following assumptions:
| ● | an
expected life of 4 years; |
| ● | volatilities
ranging from of 139% - 141% and 133% - 135% for 2015 and 2014, respectively |
| ● | forfeitures
at a rate of 12%; and |
| ● | risk
free interest rates ranging from of 0.99% - 1.31% and 1.11% - 1.33% for 2015 and 2014
respectively. |
The
weighted average fair value of each option grant made during 2015 and 2014 was estimated on the date of each grant using the Black-Scholes
option pricing model and amortized ratably over the option vesting periods, which approximates the service period.
Income
Taxes
Deferred
tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement
carrying amounts of existing assets and liabilities and their respective tax bases. A valuation allowance is established when
it is more likely than not that all or a portion of a deferred tax asset will not be realized. A review of all available positive
and negative evidence is considered, including the Company’s current and past performance, the market environment in which
the Company operates, the utilization of past tax credits, and the length of carryback and carryforward periods. Deferred
tax assets and liabilities are measured utilizing tax rates expected to apply to taxable income in the years in which those temporary
differences are expected to be recovered or settled. No current or deferred income taxes have been provided through March 31,
2015 due to the net operating losses incurred by the Company since its inception. The Company recognizes accrued interest and
penalties associated with uncertain tax positions, if any, as part of income tax expense. There were no tax related interest and
penalties recorded for 2015 and 2014. Additionally, the Company has not recorded an asset for unrecognized tax benefits or a liability
for uncertain tax positions at March 31, 2015 and December 31, 2014. Tax years beginning in 2011 for federal purposes are generally
subject to examination by taxing authorities, although net operating losses from those years are subject to examinations and adjustments
for at least three years following the year in which the tax attributes are utilized.
Earnings
Per Share
Basic
earnings per share (“EPS”) excludes dilution and is computed by dividing income (loss) available to common stockholders
by the weighted-average number of common shares outstanding for the period. Diluted EPS reflects the potential dilution that could
occur if securities or other contracts to issue common stock were exercised or converted into common stock or resulted in the
issuance of common stock that shared in the earnings of the entity. Since there is a significant number of options and warrants
outstanding, fluctuations in the actual market price can have a variety of results for each period presented.
| | |
For the Quarter Ended | | |
For the Quarter Ended | |
| | |
March
31, 2015 | | |
March 31, 2014 | |
| Basic and diluted numerator: | |
| | |
| |
| Net loss | |
$ | (4,569,322 | ) | |
$ | (3,331,708 | ) |
| | |
| | | |
| | |
| Basic and diluted Denominator: | |
| | | |
| | |
| Weighted-average outstanding | |
| 24,405,813 | | |
| 19,739,470 | |
| | |
| | | |
| | |
| Basic and diluted net loss per share | |
$ | (0.19 | ) | |
$ | (0.17 | ) |
The
following table summarizes potentially dilutive adjustments to the weighted average number of common shares which were excluded
from the calculation because their effect would be anti-dilutive:
| | |
For the Quarter Ended | | |
For the Quarter Ended | |
| | |
March
31, 2015 | | |
March 31, 2014 | |
| Common stock purchase warrants | |
| 6,085,714 | | |
| 6,808,324 | |
| Stock options | |
| 2,272,022 | | |
| 2,127,699 | |
| Total | |
| 8,357,736 | | |
| 8,936,023 | |
The weighted average exercise price of the
Company’s stock options and warrants outstanding at March 31, 2015 were $2.34 and $1.25 per share, respectively, and at
March 31, 2014 were $2.57 and $2.08 per share, respectively.
Use
of Estimates and Assumptions
The
preparation of financial statements in conformity with accounting principles generally accepted in the U.S. requires management
to make estimates and assumptions such as the fair value of warrants, stock options and recovery of the useful life of intangibles
that affect the reported amounts in the financial statements and accompanying notes. Actual results could differ from those estimates.
Note
3. Intangible Assets
The
following is a summary of intangible assets which consists of licenses and patents:
| | |
Weighted Average
Remaining Amortization
Period (years) | |
Cost | | |
Accumulated Amortization | | |
Net Book Value | |
| March 31, 2015 | |
| |
| | |
| | |
| |
| Licenses | |
4.5 | |
$ | 462,234 | | |
$ | 313,211 | | |
$ | 149,023 | |
| Patents | |
1.7 | |
| 1,893,185 | | |
| 1,686,298 | | |
| 206,887 | |
| Total | |
2.4 | |
$ | 2,355,419 | | |
$ | 1,999,509 | | |
$ | 355,910 | |
December 31, 2014 | |
| |
| | | |
| | | |
| | |
| Licenses | |
4.7 | |
$ | 462,234 | | |
$ | 306,495 | | |
$ | 155,739 | |
| Patents | |
1.9 | |
| 1,893,185 | | |
| 1,638,975 | | |
| 254,210 | |
| Total | |
2.6 | |
$ | 2,355,419 | | |
| $ 1, 945,470 | | |
$ | 409,949 | |
Amortization
expense was $54,039 and $54,712 for the three months ended March 31, 2015 and 2014, respectively.
Based
on the balance of licenses and patents at March 31, 2015, the annual amortization expense for each of the succeeding five years
is estimated to be as follows:
| | |
Amortization
Expense | |
April 1 through
December 31,2015 | |
$ | 162,100 | |
| 2016 | |
$ | 81,900 | |
| 2017 | |
$ | 37,300 | |
| 2018 | |
$ | 37,300 | |
| 2019 | |
$ | 37,300 | |
License
fees and royalty payments are expensed as incurred as the Company does not attribute any future benefits to such payments.
Note
4. Warrant Liability
Warrants
issued in connection with the Company’s June 2013 registered public offering contain provisions that protect holders from
a decline in the issue price of its common stock (or “down-round” provision) and contain net settlement provisions.
As a result, the Company accounts for these warrants as liabilities instead of equity instruments. Down-round provisions reduce
the exercise or conversion price of a warrant if the Company issues equity shares for a price that is lower than the exercise
or conversion price of the warrants. Net settlement provisions allow the holder of the warrant to surrender shares underlying
the warrant equal to the exercise price as payment of its exercise price, instead of exercising the warrant by paying cash. The
Company evaluates whether warrants to acquire its common stock contain provisions that protect holders from declines in the stock
price or otherwise could result in modification of the exercise price and/or shares to be issued under the respective warrant
agreements based on a variable that is not an input to the fair value of a “fixed for fixed” option. As a result of
the Company’s December 2014 registered public unit offering, the exercise price of warrants outstanding in connection with
the public offering completed in June 2013 was adjusted to $0.61 per share.
The
Company recognized these warrants as liabilities at their fair value on the date of grant and remeasures them to fair value on
each reporting date.
The
Company recognized an initial warrant liability for the warrants issued in connection with the registered public offering completed
in June 2013 totaling $4,827,788, which was based on the June 25, 2013 closing price of a share of the Company’s common
stock as reported on OTC Markets of $0.96. During the three months ended March 31, 2015, 1,148,786 warrants were exercised. The
fair value of the warrants exercised, or $1,648,811, was reclassified from warrant liability to additional paid-in capital. On
March 31, 2015, the closing price of the Company’s common stock as reported on OTC Markets was $1.69. Due to the fluctuations
in the market value of the Company’s common stock from December 31, 2014 through March 31, 2015, the Company recognized
a non-cash charge of $3,011,616 for the change in the fair value of the warrant liability for the three months ended March 31,
2015.
The assumptions
used in connection with the valuation of warrants issued utilizing the binomial method were as follows:
| | |
December 31, 2014 | | |
Exercised during 2015 | | |
March 31, 2015 | |
| | |
| | |
| | |
| |
| Number of shares underlying the warrants | |
| 4,723,357 | | |
| 1,141,786 | | |
| 3,581,571 | |
| Exercise price | |
$ | 0.61 | | |
$ | 0.61 | | |
$ | 0.61 | |
| Volatility | |
| 128 | % | |
| 119 | % | |
| 119 | % |
| Risk-free interest rate | |
| 1.38 | % | |
| 0.89 | % | |
| 0.89 | % |
| Expected dividend yield | |
| 0 | | |
| 0 | | |
| 0 | |
| Expected warrant life (years) | |
| 3.5 | | |
| 3.33 | | |
| 3.24 | |
| Stock Price | |
$ | 0.98 | | |
$ | 1.69 | | |
$ | 1.69 | |
The
table below provides a reconciliation of the beginning and ending balances for the liability measured at fair value using significant
unobservable inputs (Level 3). The table reflects losses for the period ended March 31, 2015 for the financial liability categorized
as Level 3 as of March 31, 2015.
Fair Value
Measurements Using Significant Unobservable Inputs (Level 3):
| | |
December 31,
2014 | | |
Decrease
from Warrants Exercised in 2015 | | |
Increase
in Fair Value | | |
March
31, 2015 | |
| | |
| | |
| | |
| | |
| |
| Warrant liability | |
$ | 3,789,562 | | |
$ | (1,648,811 | ) | |
$ | 3,011,616 | | |
$ | 5,152,367 | |
Note
5. Income Taxes
The
Company had NOLs at December 31, 2014 of approximately $86,120,000 for federal tax purposes and approximately $5,263,000 of New
Jersey NOL carry forwards remaining after the sale of unused net operating loss carry forwards, portions of which are currently
expiring each year through 2034. In addition, the Company has $3,556,000 of various tax credits that expire from 2018 to 2034.
The Company may be able to utilize its NOLs to reduce future federal and state income tax liabilities. However, these NOLs are
subject to various limitations under Internal Revenue Code (“IRC”) Section 382. IRC Section 382 limits the use of
NOLs to the extent there has been an ownership change of more than 50 percentage points. In addition, the NOL carry forwards are
subject to examination by the taxing authority and could be adjusted or disallowed due to such exams. Although the Company has
not undergone an IRC Section 382 analysis, it is likely that the utilization of the NOLs may be substantially limited.
The
Company and one or more of its subsidiaries files income tax returns in the U.S. Federal jurisdiction, and various state and local
jurisdictions. The Company is no longer subject to Federal income tax assessment for years before 2011 for Federal and 2010 for
New Jersey income tax assessment. However, since the Company has incurred net operating losses in every tax year since inception,
all its income tax returns are subject to examination and adjustments by the Internal Revenue Service for at least three years
following the year in which the tax attributes are utilized.
The
Company has no tax provision for the three month periods ended March 31, 2015 and 2014 due to losses incurred and the recognition
of full valuation allowances recorded against net deferred tax assets.
Note
6. Shareholders’ Equity
Preferred
Stock
The
Company has 350,000 shares of preferred stock authorized, none of which are issued or outstanding.
Common
Stock
During
the three months ended March 31, 2015, the Company issued the following shares of common stock:
| ● | In
seventeen separate transactions, the Company issued 1,183,786 shares of common stock
in connection with warrant exercises; |
| ● | In
two separate transactions, the Company issued 153,010 shares of common stock pursuant
to the Lincoln Park facility; and |
| ● | In
three separate transactions, the Company issued 66,000 shares of common stock as partial
consideration for services performed. |
Note
7. Commitments and Contingencies
The
Company has commitments of approximately $500,000 as of March 31, 2015 for agreements with consultants and universities. Additionally,
the Company has collaboration and license agreements, which upon clinical or commercialization success, may require the payment
of milestones of up to $7.9 million and/or royalties up to 6% of net sales of covered products, if and when achieved. However,
there can be no assurance that clinical or commercialization success will occur.
In
December 2014, the Company entered into a lease agreement through May 31, 2018 for existing and expanded office space. The rent
for the first 12 months is approximately $12,300 per month, or approximately $20.85 per square foot. This rent increases to approximately
$12,375 per month, or approximately $20.95 per square foot, for the next 12 months and approximately $12,460 per month, or approximately
$21.13 per square foot for the remainder of the lease.
On September 3, 2014, the Company entered
into an asset purchase agreement with Hy Biopharma, Inc. (“Hy Biopharma) pursuant to which the Company acquired certain
intangible assets, properties and rights of Hy Biopharma related to the development of Hy BioPharma’s synthetic hypericin
product. As consideration for the assets acquired, the Company paid $250,000 in cash and issued 1,849,113 shares of common stock
with a fair value based on the Company’s stock price on the date of grant of $3,750,000. These amounts were charged to research
and development expense during the third quarter of 2014 as the assets will be used in the Company’s research and development
activities and do not have alternative future use pursuant to generally accepted accounting principles in the United States. Provided
all future success-oriented milestones are attained, the Company will be required to make additional payments of up to $10.0 million,
if and when achieved. Payments will be payable in restricted securities of the Company not to exceed 19.9% ownership of Company’s
outstanding stock.
On
April 27, 2013, the Company entered into an exclusive channel collaboration agreement (the “Channel Agreement”) with
Intrexon to use Intrexon’s advanced human antibody discovery, isolation and production technologies for the development
of human monoclonal antibody therapies for a new biodefense application targeting melioidosis. The Channel Agreement grants an
exclusive worldwide license to use specified patents and other intellectual property of Intrexon in connection with the research,
development, use, importing, manufacture, sale and offer for sale of products for the treatment of melioidosis through the use
of exogenously produced human recombinant monoclonal antibodies. The Channel Agreement, upon clinical or commercialization success,
may require the payment of certain milestones payments up to $7 million, if and when achieved.
In
February 2007, the Company’s Board of Directors authorized the issuance of 50,000 shares of the Company’s common stock
to Dr. Schaber immediately prior to the completion of a transaction, or series or a combination of related transactions, negotiated
by its Board of Directors whereby, directly or indirectly, a majority of its capital stock or a majority of its assets are transferred
from the Company and/or its stockholders to a third party. Dr. Schaber’s amended employment agreement includes the Company’s
obligation to issue such shares if such event occurs.
As a result
of the above agreements, the Company has future contractual obligations over the next five years as follows:
Year | |
Research and Development | | |
Property and Other Leases | | |
Total | |
| April 1 through December 31, 2015 | |
$ | 100,000 | | |
$ | 99,000 | | |
$ | 199,000 | |
| 2016 | |
| 100,000 | | |
| 157,000 | | |
| 257,000 | |
| 2017 | |
| 100,000 | | |
| 152,000 | | |
| 252,000 | |
| 2018 | |
| 100,000 | | |
| 52,000 | | |
| 152,000 | |
| 2019 | |
| 100,000 | | |
| - | | |
| 100,000 | |
| Total | |
$ | 500,000 | | |
$ | 460,000 | | |
$ | 960,000 | |
Note
8. Operating Segments
The
Company maintains two active operating segments: BioTherapeutics and Vaccines/BioDefense. Each segment includes an element of
overhead costs specifically associated with its operations, with its corporate shared services group responsible for support functions
generic to both operating segments.
| | |
Three Months Ended March 31, | |
| | |
2015 | | |
2014 | |
| Contract/Grant Revenue | |
| | |
| |
| Vaccines/BioDefense | |
$ | 802,314 | | |
$ | 877,045 | |
| BioTherapeutics | |
| 13,972 | | |
| 33,552 | |
| Total | |
$ | 816,286 | | |
$ | 910,597 | |
| | |
| | | |
| | |
| Income/(Loss) from Operations | |
| | | |
| | |
| Vaccines/BioDefense | |
$ | 84,681 | | |
$ | 139,404 | |
| BioTherapeutics | |
| (764,876 | ) | |
| (1,035,591 | ) |
| Corporate | |
| (878,072 | ) | |
| (693,722 | ) |
| Total | |
$ | (1,558,267 | ) | |
$ | (1,589,909 | ) |
Amortization and Depreciation Expense | |
| | | |
| | |
| Vaccines/BioDefense | |
$ | 9,786 | | |
$ | 9,935 | |
| BioTherapeutics | |
| 48,374 | | |
| 49,939 | |
| Corporate | |
| 1,766 | | |
| 2,213 | |
| Total | |
$ | 59,926 | | |
$ | 62,087 | |
| | |
| | | |
| | |
Interest
Income | |
| | | |
| | |
| Corporate | |
$ | 561 | | |
$ | 291 | |
| | |
| | | |
| | |
| Share-Based Compensation | |
| | | |
| | |
| Vaccines/BioDefense | |
$ | 24,592 | | |
$ | 10,450 | |
| BioTherapeutics | |
| 29,256 | | |
| 76,121 | |
| Corporate | |
| 88,177 | | |
| 90,091 | |
| Total | |
$ | 142,025 | | |
$ | 176,662 | |
| | |
As
of March
31, 2015 | | |
As of
December 31,
2014 | |
| | |
| | |
| |
| Identifiable Assets | |
| | |
| |
| Vaccines/BioDefense | |
$ | 568,565 | | |
$ | 1,025,220 | |
| BioTherapeutics | |
| 161,355 | | |
| 204,308 | |
| Corporate | |
| 5,172,737 | | |
| 5,724,720 | |
| Total | |
$ | 5,902,657 | | |
$ | 6,954,248 | |
Soligenix,
Inc. and Subsidiaries
Consolidated
Balance Sheets
As
of December 31,
| | |
2014 | | |
2013 | |
| Assets | |
| | | |
| | |
| Current
assets: | |
| | | |
| | |
| Cash
and cash equivalents | |
$ | 5,525,094 | | |
$ | 5,856,242 | |
| Contracts and grants
receivable | |
| 794,767 | | |
| 867,086 | |
| Taxes receivable | |
| - | | |
| 750,356 | |
| Prepaid
expenses | |
| 172,928 | | |
| 135,391 | |
| Total current assets | |
| 6,492,789 | | |
| 7,609,075 | |
| Office furniture and equipment, net | |
| 51,510 | | |
| 23,868 | |
| Intangible assets,
net | |
| 409,949 | | |
| 632,512 | |
| Total assets | |
$ | 6,954,248 | | |
$ | 8,265,455 | |
| | |
| | | |
| | |
| Liabilities and shareholders’
deficiency | |
| | | |
| | |
| Current liabilities: | |
| | | |
| | |
| Accounts payable | |
$ | 3,003,545 | | |
$ | 1,520,290 | |
| Warrant liability | |
| 3,789,562 | | |
| 8,281,247 | |
| Accrued
compensation | |
| 315,030 | | |
| 233,739 | |
| Total current
liabilities | |
| 7,108,137 | | |
| 10,035,276 | |
| Commitments and contingencies | |
| | | |
| | |
| Shareholders’ deficiency: | |
| | | |
| | |
| Preferred stock;
350,000 shares authorized; none issued or outstanding | |
| - | | |
| - | |
| Common
stock, $.001 par value; 50,000,000 shares authorized in 2014 and 2013, respectively; 23,936,568 shares and 19,626,439 shares
issued and outstanding in 2014 and 2013, respectively | |
| 23,937 | | |
| 19,626 | |
| Additional paid-in
capital | |
| 138,868,523 | | |
| 130,549,930 | |
| Accumulated
deficit | |
| (139,046,349 | ) | |
| (132,339,377 | ) |
| Total shareholders’
deficiency | |
| (153,889 | ) | |
| (1,769,821 | ) |
| Total liabilities
and shareholders’ deficiency | |
$ | 6,954,248 | | |
$ | 8,265,455 | |
The
accompanying notes are an integral part of these consolidated financial statements.
Soligenix,
Inc. and Subsidiaries
Consolidated
Statements of Operations
For
the Years Ended December 31,
| | |
2014 | | |
2013 | |
| Revenues: | |
| | |
| |
| Grant
revenue | |
$ | 1,497,548 | | |
$ | 2,658,836 | |
| Contract
revenue | |
| 5,545,468 | | |
| 565,316 | |
| Total revenues | |
| 7,043,016 | | |
| 3,224,152 | |
| Cost of revenues | |
| (5,313,855 | ) | |
| (2,544,285 | ) |
| Gross
profit | |
| 1,729,161 | | |
| 679,867 | |
| Operating expenses: | |
| | | |
| | |
| Research
and development | |
| 5,086,535 | | |
| 5,071,179 | |
| Acquired
in-process research and development | |
| 4,000,000 | | |
| - | |
| General
and administrative | |
| 3,403,975 | | |
| 2,765,230 | |
| Total operating
expenses | |
| 12,490,510 | | |
| 7,836,409 | |
| Loss from operations | |
| (10,761,349 | ) | |
| (7,156,542 | ) |
| Other income (expense): | |
| | | |
| | |
| Change
in fair value of warrant liability | |
| 3,436,195 | | |
| (3,654,770 | ) |
| Interest
income | |
| 1,310 | | |
| 1,960 | |
| Total other income
(expense) | |
| 3,437,505 | | |
| (3,652,810 | ) |
| Net loss before income taxes | |
| (7,323,844 | ) | |
| (10,809,352 | ) |
| Income tax benefit | |
| 616,872 | | |
| 750,356 | |
| Net loss | |
$ | (6,706,972 | ) | |
$ | (10,058,996 | ) |
| Basic net loss per share | |
$ | (0.32 | ) | |
$ | (0.65 | ) |
| Diluted net loss per share | |
$ | (0.43 | ) | |
$ | (0.65 | ) |
| Basic weighted average
common shares outstanding | |
| 20,638,421 | | |
| 15,463,256 | |
| Diluted weighted average
common shares outstanding | |
| 23,584,944 | | |
| 15,463,256 | |
The
accompanying notes are an integral part of these consolidated financial statements.
Soligenix,
Inc. and Subsidiaries
Consolidated
Statements of Changes in Shareholders’ Deficiency
For
the Years Ended December 31, 2014 and 2013
| | |
| | |
Additional | | |
| | |
| |
| | |
Common
Stock | | |
Paid–In | | |
Accumulated | | |
| |
| | |
Shares | | |
Par
Value | | |
Capital | | |
Deficit | | |
Total | |
| Balance,
December 31, 2012 | |
| 11,168,905 | | |
$ | 11,169 | | |
$ | 125,820,318 | | |
$ | (122,280,381 | ) | |
$ | 3,551,106 | |
| Common
stock issued in Unit offering, net of offering costs of $902,158 | |
| 6,773,995 | | |
| 6,774 | | |
| 6,203,763 | | |
| - | | |
| 6,210,537 | |
| Warrants
issued in Unit offering | |
| - | | |
| - | | |
| (4,827,788 | ) | |
| - | | |
| (4,827,788 | ) |
| Reclassification
of warrant liability upon partial exercise of warrants issued in unit offering | |
| - | | |
| - | | |
| 201,311 | | |
| - | | |
| 201,311 | |
| Issuance
of common stock to collaboration partner | |
| 1,034,483 | | |
| 1,034 | | |
| 1,498,966 | | |
| - | | |
| 1,500,000 | |
| Issuance
of common stock pursuant to Lincoln Park equity line, net of costs of $71,949 | |
| 383,370 | | |
| 383 | | |
| 527,668 | | |
| - | | |
| 528,051 | |
| Issuance
of shares from exercise of stock options and warrants | |
| 210,582 | | |
| 211 | | |
| 235,764 | | |
| - | | |
| 235,975 | |
| Issuance
of common stock to vendor | |
| 55,104 | | |
| 55 | | |
| 82,093 | | |
| - | | |
| 82,148 | |
| Fair
value of common stock warrants to vendors | |
| - | | |
| - | | |
| 4,775 | | |
| - | | |
| 4,775 | |
| Stock-based
compensation expense | |
| - | | |
| - | | |
| 803,060 | | |
| - | | |
| 803,060 | |
| Net
loss | |
| - | | |
| - | | |
| - | | |
| (10,058,996 | ) | |
| (10,058,996 | ) |
| Balance,
December 31, 2013 | |
| 19,626,439 | | |
$ | 19,626 | | |
$ | 130,549,930 | | |
$ | (132,339,377 | ) | |
$ | (1,769,821 | ) |
| Issuance
of common stock pursuant to Lincoln Park Equity line | |
| 230,743 | | |
| 231 | | |
| 470,244 | | |
| - | | |
| 470,475 | |
| Issuance
of common stock to vendors | |
| 121,000 | | |
| 121 | | |
| 255,919 | | |
| - | | |
| 256,040 | |
| Issuance of shares
from exercise of stock options | |
| 36,672 | | |
| 37 | | |
| 28,041 | | |
| - | | |
| 28,078 | |
| Reclassification
of warrant liability upon partial exercise of warrants issued in unit offering | |
| - | | |
| - | | |
| 1,055,490 | | |
| - | | |
| 1,055,490 | |
| Fair
value of common stock warrants issued to vendors | |
| - | | |
| - | | |
| 4,775 | | |
| - | | |
| 4,775 | |
| Issuance
of common stock to collaboration partner | |
| 43,067 | | |
| 43 | | |
| 99,959 | | |
| - | | |
| 100,002 | |
| Shares
issued in connection with acquisition of in-process research and development | |
| 1,849,113 | | |
| 1,849 | | |
| 3,748,151 | | |
| - | | |
| 3,750,000 | |
| Issuance
of common stock from cashless exercise of warrants | |
| 143,004 | | |
| 143 | | |
| (143 | ) | |
| - | | |
| - | |
| Stock-based
compensation expense | |
| - | | |
| - | | |
| 720,150 | | |
| - | | |
| 720,150 | |
| Common
stock issued in unit offering, net of offering costs of $344,808 | |
| 1,886,530 | | |
| 1,887 | | |
| 1,936,007 | | |
| - | | |
| 1,937,894 | |
| Net
loss | |
| - | | |
| - | | |
| - | | |
| (6,706,972 | ) | |
| (6,706,972 | ) |
| Balance,
December 31, 2014 | |
| 23,936,568 | | |
| 23,937 | | |
$ | 138,868,523 | | |
$ | (139,046,349 | ) | |
$ | (153,889 | ) |
The
accompanying notes are an integral part of these consolidated financial statements.
Soligenix,
Inc. and Subsidiaries
Consolidated
Statements of Cash Flows
For
the Years Ended December 31,
| | |
2014 | | |
2013 | |
| Operating activities: | |
| | |
| |
| Net
loss | |
$ | (6,706,972 | ) | |
$ | (10,058,996 | ) |
| Adjustments
to reconcile net loss to net cash used in operating activities: | |
| | | |
| | |
| Amortization
and depreciation | |
| 245,787 | | |
| 230,071 | |
| Charge
for common stock issued for collaboration agreement | |
| 100,002 | | |
| 1,500,000 | |
| Common
stock issued in exchange for services | |
| 256,040 | | |
| 82,148 | |
| Issuance
of common stock for acquisition of in-process research and development | |
| 4,000,000 | | |
| - | |
| Warrants
issued to vendor | |
| 4,775 | | |
| 4,775 | |
| Stock-based compensation | |
| 720,150 | | |
| 803,060 | |
| Change
in fair value of warrant liability | |
| (3,436,195 | ) | |
| 3,654,770 | |
| Change
in operating assets and liabilities: | |
| | | |
| | |
| Grants
and contracts receivable | |
| 72,319 | | |
| (527,778 | ) |
| Taxes
receivable | |
| 750,356 | | |
| (750,356 | ) |
| Prepaid
expenses | |
| (37,537 | ) | |
| 5,302 | |
| Accounts
payable | |
| 1,483,255 | | |
| 395,787 | |
| Accrued
compensation | |
| 81,291 | | |
| 204,244 | |
| Total
adjustments and change in operating assets and liabilities | |
| 4,240,243 | | |
| 5,602,023 | |
| Net
cash used in operating activities | |
| (2,466,729 | ) | |
| (4,456,973 | ) |
| | |
| | | |
| | |
| Investing
activities: | |
| | | |
| | |
| Payments
for acquisition of in-process research and development | |
| (250,000 | ) | |
| - | |
| Purchases
of office equipment | |
| (50,866 | ) | |
| (17,728 | ) |
| Net
cash used in investing activities | |
| (300,866 | ) | |
| (17,728 | ) |
| | |
| | | |
| | |
| Financing
activities: | |
| | | |
| | |
| Net
proceeds from sale of units containing common stock and warrants | |
| 1,937,894 | | |
| 6,210,537 | |
| Net
proceeds from issuance of common stock pursuant to the equity line | |
| 470,475 | | |
| 528,051 | |
| Proceeds
from exercise of options and warrants | |
| 28,078 | | |
| 235,975 | |
| Net
cash provided by financing activities | |
| 2,436,447 | | |
| 6,974,563 | |
| | |
| | | |
| | |
| Net
increase (decrease) in cash and cash equivalents | |
| (331,148 | ) | |
| 2,499,862 | |
| Cash
and cash equivalents at beginning of period | |
| 5,856,242 | | |
| 3,356,380 | |
| Cash
and cash equivalents at end of period | |
$ | 5,525,094 | | |
$ | 5,856,242 | |
| Supplemental
disclosure of non cash investing and financing activities: | |
| | | |
| | |
| Fair
Value of warrants issued in Unit Offering | |
$ | - | | |
$ | 4,827,788 | |
Reclassification
of warrant liability to additional paid in capital upon
partial exercise of warrants issued in unit offering | |
$ | 1,055,490 | | |
$ | 201,311 | |
| Supplemental information: | |
| | |
| |
| | |
| | | |
| | |
| Cash
paid for state income taxes | |
$ | 6,994 | | |
$ | 3,080 | |
The
accompanying notes are an integral part of these consolidated financial statements.
Soligenix,
Inc. and Subsidiaries
Notes
to Consolidated Financial Statements
Note
1. Nature of Business
Basis
of Presentation
Soligenix,
Inc. (the “Company”) is a late-stage biopharmaceutical company developing product candidates intended to address unmet
medical needs in the areas of inflammation, oncology, and biodefense. The Company maintains two active business segments: BioTherapeutics
and Vaccines/BioDefense.
The
Company’s BioTherapeutics business segment is developing a first-in-class photo-dynamic therapy (SGX301) utilizing safe
visible light for the treatment of cutaneous T-cell lymphoma (“CTCL”), proprietary formulations of oral beclomethasone
17,21-dipropionate (“BDP”) for the prevention/treatment of gastrointestinal (“GI”) disorders characterized
by severe inflammation, including pediatric Crohn’s disease (SGX203) and acute radiation enteritis (SGX201), and our novel
innate defense regulator (“IDR”) technology (SGX942) for the treatment of oral mucositis.
The
Company’s Vaccines/BioDefense business segment includes active development programs for RiVax™, its ricin toxin vaccine
candidate, VeloThrax™, an anthrax vaccine candidate, OrbeShield™, a GI acute radiation syndrome (“GI ARS”)
therapeutic candidate and SGX943, a melioidosis therapeutic candidate. The development of the vaccine programs is supported by
the heat stabilization technology, known as ThermoVax™, under existing and on-going government contract funding. With the
recently awarded government contracts from the National Institute of Allergy and Infectious Diseases (“NIAID”), the
Company will attempt to advance the development of RiVax™ to protect against exposure to ricin toxin. The Company plans
to use the funds received under the government contracts with the Biomedical Advanced Research and Development Authority (“BARDA”)
and NIAID to advance the development of OrbeShield™ for the treatment of GI ARS. Additionally, the Company entered into
a global and exclusive channel collaboration with Intrexon Corporation (“Intrexon”) through which it intends to develop
and commercialize a human monoclonal antibody therapy (SGX101) to treat melioidosis.
The
Company generates revenues under three active grants primarily from the NIH and government contracts from BARDA and NIAID.
The
Company is subject to risks common to companies in the biotechnology industry including, but not limited to, development of new
technological innovations, dependence on key personnel, protections of proprietary technology, compliance with the United States
Food and Drug Administration (the “FDA”) regulations, litigation, and product liability.
Liquidity
As
of December 31, 2014, the Company had cash and cash equivalents of $5,525,094 as compared to $5,856,242 as of December 31, 2013,
representing a decrease of $331,148 or 6%. The decrease in cash was primarily due to net cash used in operations of $2,466,729
partially offset by cash provided by financing activities of $2,436,447. As of December 31, 2014, the Company had working capital
of $3,174,214, which excludes a non-cash warrant liability of $3,789,562, as compared to working capital of $5,855,046 as of December
31, 2013, representing a decrease of $2,680,832 or 46%. The decrease in working capital was primarily the result of expenditures
related to support the Phase 2 clinical trial of SGX942 and a decrease in taxes receivable offset by the net proceeds of $1,937,894
received from our registered public offering, proceeds from our Lincoln Park equity line of $470,475 and proceeds from the exercise
of stock options of $28,078.
Based
on the Company’s current rate of cash outflows, cash on hand, proceeds from its government contract and grant programs, proceeds
expected from the Lincoln Park Capital Fund, LLC (“Lincoln Park”) equity line and proceeds from the state of New Jersey
Technology Business Tax Certificate Transfer Program, management believes that its current cash will be sufficient to meet the
anticipated cash needs for working capital and capital expenditures for at least the next twelve months.
Management’s
business strategy can be outlined as follows:
| ● | Conduct
a Phase 3 clinical trial of SGX301 for the treatment of CTCL; |
| ● | Conduct
a Phase 2 clinical trial of SGX942 for the treatment of oral mucositis in head and neck
cancer; |
| ● | Conduct
a Phase 3 clinical trial of oral BDP, known as SGX203 for the treatment of pediatric
Crohn’s disease; |
| ● | Evaluate
the effectiveness of oral BDP in other therapeutic indications involving inflammatory
conditions of the GI tract such as prevention of acute radiation enteritis, prevention
of acute radiation syndrome, and treatment of chronic GI GVHD; |
| ● | Develop
RiVax™ and VeloThrax™ in combination with its proprietary vaccine heat stabilization
technology known as ThermoVax™, to develop new heat stable vaccines in biodefense
and infectious diseases with the potential to collaborate and/or partner with other companies
in these areas; |
| ● | Advance
the preclinical and manufacturing development of OrbeShield™ as a biodefense medical
countermeasure for the treatment of GI ARS; |
| ● | Continue
to apply for and secure additional government funding for each of its BioTherapeutics
and Vaccine/BioDefense programs through grants, contracts and/or procurements; |
| ● | Acquire
or in-license new clinical-stage compounds for development; and |
| ● | Explore
other business development and merger/acquisition strategies an example of which is the
collaboration with Intrexon. |
The
Company’s plans with respect to its liquidity management include, but are not limited to the following:
| ● | The
Company has up to $51.4 million in active government contract and grant funding still
available to support its associated research programs through 2015 and beyond. The Company
plans to submit additional contract and grant applications for further support of its
programs with various funding agencies. |
| ● | The
Company has continued to use equity instruments to provide a portion of the compensation
due to vendors and collaboration partners and expects to continue to do so for the foreseeable
future. |
| ● | The
Company will pursue Net Operating Loss (“NOLs”) sales in the state of New
Jersey pursuant to its Technology Business Tax Certificate Transfer Program. Based on
the receipt, in December 2014, of $616,872 in proceeds pursuant to NOLs sales , the Company
expects to participate in the program during 2015 and beyond; |
| ● | The
Company has a $10.6 million equity facility, with Lincoln Park, through October 2016,
of which approximately $9.5 million was available at December 31, 2014; and |
| ● | The
Company may seek additional capital in the private and/or public equity markets to continue
its operations, respond to competitive pressures, develop new products and services,
and to support new strategic partnerships. The Company is currently evaluating additional
equity financing opportunities on an ongoing basis and may execute them when appropriate.
However, there can be no assurances that the Company can consummate such a transaction,
or consummate a transaction at favorable pricing. |
Note
2. Summary of Significant Accounting Policies
Principles
of Consolidation
The
consolidated financial statements include Soligenix, Inc., and its wholly and majority owned subsidiaries. All significant intercompany
accounts and transactions have been eliminated as a result of consolidation.
Operating
Segments
Operating
segments are defined as components of an enterprise about which separate financial information is available that is evaluated
on a regular basis by the chief operating decision maker, or decision making group, in deciding how to allocate resources to an
individual segment and in assessing the performance of the segment. The Company divides its operations into two operating segments:
BioTherapeutics and Vaccines/BioDefense.
Cash
and cash equivalents
The
Company considers all highly liquid investments with original maturities of three months or less to be cash equivalents.
Contracts
and Grants Receivable
Contracts
and grants receivable consist of unbilled amounts due from various grants from the National Institutes of Health (“NIH”)
and contracts from BARDA and NIAID, an institute of NIH, for costs incurred prior to the period end under reimbursement contracts.
The amounts were billed to the respective governmental agencies in the month subsequent to period end and collected shortly thereafter.
Accordingly, no allowance for doubtful amounts has been established. If amounts become uncollectible, they are charged to operations.
Intangible
Assets
One
of the most significant estimates or judgments that the Company makes is whether to capitalize or expense patent and license costs.
The Company makes this judgment based on whether the technology has alternative future uses, as defined in Financial Accounting
Standards Board (“FASB”) Accounting Standards Codification (“ASC”) 730, Research and Development.
Based on this consideration, the Company capitalizes payments made to legal firms that are engaged in filing and protecting rights
to intellectual property and rights for its current products in both the domestic and international markets. The Company believes
that patent rights are one of its most valuable assets. Patents and patent applications are a key component of intellectual property,
especially in the early stage of product development, as their purchase and maintenance gives the Company access to key product
development rights from Soligenix’s academic and industry partners. These rights can also be sold or sub-licensed as part
of its strategy to partner its products at each stage of development as the intangible assets have alternative future use. The
legal costs incurred for these patents consist of work associated with filing new patents and perhaps extending the lives of the
patents. The Company capitalizes such costs and amortizes intangibles on a straight-line basis over their expected useful life
– generally a period of 11 to 16 years.
The
Company did not capitalize any patent related costs during the years ended December 31, 2014 or 2013.
Impairment
of Long-Lived Assets
Office
furniture and equipment and intangible assets are evaluated and reviewed for impairment whenever events or changes in circumstances
indicate that the carrying amount may not be recoverable. The Company recognizes impairment of long-lived assets in the event
the net book value of such assets exceeds the estimated future undiscounted cash flows attributable to such assets. If the sum
of the expected undiscounted cash flows is less than the carrying value of the related asset or group of assets, a loss is recognized
for the difference between the fair value and the carrying value of the related asset or group of assets. Such analyses necessarily
involve significant judgment.
The
Company did not record any impairment of long-lived assets for the years ended December 31, 2014 or 2013.
Fair
Value of Financial Instruments
FASB
ASC 820 — Fair Value Measurements and Disclosures, defines fair value as the price that would be received to sell
an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. FASB ASC
820 requires disclosures about the fair value of all financial instruments, whether or not recognized, for financial statement
purposes. Disclosures about the fair value of financial instruments are based on pertinent information available to the Company
on December 31, 2014. Accordingly, the estimates presented in these financial statements are not necessarily indicative of the
amounts that could be realized on disposition of the financial instruments.
FASB
ASC 820 specifies a hierarchy of valuation techniques based on whether the inputs to those valuation techniques are observable
or unobservable. Observable inputs reflect market data obtained from independent sources, while unobservable inputs reflect market
assumptions. The hierarchy gives the highest priority to unadjusted quoted prices in active markets for identical assets or liabilities
(Level 1 measurement) and the lowest priority to unobservable inputs (Level 3 measurement).
The
three levels of the fair value hierarchy are as follows:
| |
● | Level
1 — Quoted prices in active markets for identical assets or liabilities that the
reporting entity has the ability to access at the measurement date. Level 1 primarily
consists of financial instruments whose value is based on quoted market prices such as
exchange-traded instruments and listed equities. |
| |
● | Level
2 — Inputs other than quoted prices included within Level 1 that are observable
for the asset or liability, either directly or indirectly. Level 2 includes financial
instruments that are valued using models or other valuation methodologies. These models
consider various assumptions, including volatility factors, current market prices and
contractual prices for the underlying financial instruments. Substantially all of these
assumptions are observable in the marketplace, can be derived from observable data or
are supported by observable levels at which transactions are executed in the marketplace. |
| |
● | Level
3 — Unobservable inputs for the asset or liability. Financial instruments are considered
Level 3 when their fair values are determined using pricing models, discounted cash flows
or similar techniques and at least one significant model assumption or input is unobservable. |
The
carrying amounts reported in the consolidated balance sheet for cash and cash equivalents, accounts receivable, accounts payable
and accrued expenses approximate their fair value based on the short-term maturity of these instruments. The Company recognizes
all derivative financial instruments as assets or liabilities in the financial statements and measures them at fair value with
changes in fair value reflected as current period income or loss unless the derivatives qualify as hedges. As a result, certain
warrants issued in connection with the June 2013 offering were accounted for as derivatives. See Note 4, Warrant Liabilities.
Revenue
Recognition
The
Company’s revenues are primarily generated from government contracts and grants. Revenue is recognized in accordance with
FASB ASC 605, Revenue Recognition and/or ASC 605-25, Revenue Recognition – Multiple Element Arrangements.
The revenue from government contracts and grants is based upon subcontractor costs and internal costs incurred that are specifically
covered by the grants, plus a facilities and administrative rate that provides funding for overhead expenses and management fees.
These revenues are recognized when expenses have been incurred by subcontractors or when the Company incurs reimbursable internal
expenses that are related to the government contracts and grants.
Research
and Development Costs
Research
and development costs are charged to expense when incurred in accordance with FASB ASC 730, Research and Development. Research
and development includes costs such as clinical trial expenses, contracted research and license agreement fees with no alternative
future use, supplies and materials, salaries, stock based compensation, employee benefits, equipment depreciation and allocation
of various corporate costs. Purchased in-process research and development expense represents the value assigned or paid for acquired
research and development for which there is no alternative future use as of the date of acquisition.
Accounting
for Warrants
The
Company considered FASB ASC 815, Evaluating Whether an Instrument is Considered Indexed to an Entity’s Own Stock,
which provides guidance for determining whether an equity-linked financial instrument (or embedded feature) issued by an entity
is indexed to the entity’s stock. The Company evaluated the warrants’ provisions and determined that warrants issued
in connection with the Company’s June 2013 registered public offering contain provisions that protect holders from a decline
in the issue price of the Company’s common stock (or “down-round” provisions) and contain net settlement provisions.
Consequently, these warrants are recognized as liabilities at their fair value on the date of grant and remeasured at fair value
on each reporting date. All other warrants issued were indexed to the Company’s stock and therefore are accounted for as
equity instruments for 2014 and 2013.
Stock-Based
Compensation
Stock
options are issued with an exercise price equal to the market price on the date of issuance. Stock options issued to directors
upon re-election vest quarterly for a period of one year (new director issuances are fully vested upon issuance). Stock options
issued to employees vest 25% immediately as of the grant date, then 25% each subsequent year for a period of three years. Stock
options vest over each three month period from the date of issuance to the end of the three year period. These options have a
ten year life for as long as the individuals remain employees or directors. In general, when an employee or director terminates
their position the options will expire within three months, unless otherwise extended by the Board.
From
time to time, the Company issues restricted shares of common stock to vendors and consultants as compensation for services performed.
Typically these instruments vest upon issuance and therefore the entire stock compensation expense is recognized upon issuance
to the vendors and/or consultants
Stock
compensation expense for options, warrants and shares of common stock granted to non-employees has been determined in accordance
with FASB ASC 505-50, Equity-Based Payments to Non-Employees, and represents the fair value of the consideration received,
or the fair value of the equity instruments issued, whichever may be more reliably measured. For options that vest over future
periods, the fair value of options granted to non-employees is amortized as the options vest.
The
fair value of options issued during the years ended December 31, 2014 and 2013 in accordance with FASB ASC 718, Stock Compensation,
was estimated to be $1.48 and $1.65 per share, respectively, using the Black-Scholes option-pricing model and the following assumptions:
| |
● | an
expected life of 4 years; |
| |
● | volatilities
ranging from 128% - 165% and 136% - 167% for 2014 and 2013, respectively; |
| |
● | forfeitures
at a rate of 12%; and |
| |
● | risk-free
interest rates ranging from 1.05% to 1.43% and 0.96% to 1.17% for 2014 and 2013, respectively.
|
The
weighted average fair value of each option grant made during 2014 and 2013 was estimated on the date of each grant using the Black-Scholes
option pricing model and amortized ratably over the option vesting periods, which approximates the service period.
Income
Taxes
Deferred
tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement
carrying amounts of existing assets and liabilities and their respective tax bases. A valuation allowance is established when
it is more likely than not that all or a portion of a deferred tax asset will not be realized. A review of all available positive
and negative evidence is considered, including the Company’s current and past performance, the market environment in which
the Company operates, the utilization of past tax credits, and the length of carryback and carryforward periods. Deferred
tax assets and liabilities are measured utilizing tax rates expected to apply to taxable income in the years in which those temporary
differences are expected to be recovered or settled. No current or deferred income taxes have been provided through December 31,
2014 due to the net operating losses incurred by the Company since its inception. The Company recognizes accrued interest and
penalties associated with uncertain tax positions, if any, as part of income tax expense. There were no tax related interest and
penalties recorded for 2014 and 2013. Additionally, the Company has not recorded an asset for unrecognized tax benefits or a liability
for uncertain tax positions at December 31, 2014 and 2013. Tax years beginning in 2011 for federal purposes are generally subject
to examination by the taxing authorities, although net operating losses from those years are subject to examinations and adjustments
for at least three years following the year in which the tax attributes are utilized.
Earnings
Per Share
Basic
earnings per share (“EPS”) excludes dilution and is computed by dividing income (loss) available to common stockholders
by the weighted-average number of common shares outstanding for the period. Diluted EPS reflects the potential dilution that could
occur if securities or other contracts to issue common stock were exercised or converted into common stock or resulted in the
issuance of common stock that shared in the earnings of the entity. Since there is a significant number of options and warrants
outstanding, fluctuations in the actual market price can have a variety of results for each period presented.
| | |
For
the Year Ended | | |
For
the Year Ended | |
| | |
December 31,
2014 | | |
December 31,
2013 | |
| Numerator: | |
| | |
| |
| Net loss for basic earnings
per share | |
$ | (6,706,972 | ) | |
$ | (10,058,996 | ) |
| Less
change in fair value of warrant liability | |
| 3,436,195 | | |
| - | |
| Net loss for diluted earnings per share | |
$ | (10,143,167 | ) | |
$ | (10,058,996 | ) |
| Denominator: | |
| | | |
| | |
| Weighted-average basic common shares
outstanding | |
| 20,638,421 | | |
| 15,463,256 | |
| Assumed conversion of dilutive securities: | |
| | | |
| | |
| Common
stock purchase warrants | |
| 2,946,523 | | |
| - | |
| Denominator for diluted earnings per share – adjusted weighted-average
shares | |
| 23,584,944 | | |
| 15,463,256 | |
| Basic net loss per share | |
$ | (0.32 | ) | |
$ | (0.65 | ) |
| Diluted net loss per share | |
$ | (0.43 | ) | |
$ | (0.65 | ) |
The
following table summarizes potentially dilutive adjustments to the weighted average number of common shares which were excluded
from the calculation:
| | |
For
the Year Ended | | |
For
the Year Ended | |
| | |
December 31,
2014 | | |
December 31,
2013 | |
| Common stock purchase warrants | |
| 2,546,143 | | |
| 8,156,526 | |
| Stock options | |
| 2,488,279 | | |
| 2,051,511 | |
| Total | |
| 5,034,422 | | |
| 10,208,037 | |
Shares
issuable upon the exercise of options and warrants outstanding at December 31, 2014 and 2013 were 2,488,279 and 2,051,511 shares
issuable upon the exercise of options, and 7,269,500 and 8,156,526 shares issuable upon the exercise of warrants, respectively.
The weighted average exercise price of the Company’s stock options and warrants outstanding at December 31, 2014 were $2.40
and $1.15 per share, respectively.
Use
of Estimates and Assumptions
The
preparation of financial statements in conformity with accounting principles generally accepted in the U.S. requires management
to make estimates and assumptions such as the fair value of warrants, stock options and recovery of the useful life of intangibles
that affect the reported amounts in the financial statements and accompanying notes. Actual results could differ from those estimates.
Note
3. Intangible Assets
The
following is a summary of intangible assets which consists of licenses and patents:
| | |
Weighted
Average Remaining Amortization Period (years) | |
Cost | | |
Accumulated
Amortization | | |
Net
Book Value | |
| December 31, 2014 | |
| | |
| | |
| |
| Licenses | |
4.7 | |
$ | 462,234 | | |
$ | 306,495 | | |
$ | 155,739 | |
| Patents | |
1.9 | |
| 1,893,185 | | |
| 1,638,975 | | |
| 254,210 | |
| Total | |
2.6 | |
$ | 2,355,419 | | |
$ | 1,945,470 | | |
$ | 409,949 | |
| December 31, 2013 | |
| |
| | | |
| | | |
| | |
| Licenses | |
5.7 | |
$ | 462,234 | | |
$ | 279,258 | | |
$ | 182,976 | |
| Patents | |
2.6 | |
| 1,893,185 | | |
| 1,443,649 | | |
| 449,536 | |
| Total | |
3.4 | |
$ | 2,355,419 | | |
$ | 1,722,907 | | |
$ | 632,512 | |
Amortization
expense was $222,563 and $223,216 in 2014 and 2013, respectively.
Based
on the balance of licenses and patents at December 31, 2014, the annual amortization expense for each of the succeeding five years
is estimated to be as follows:
| Year | |
Amortization
Expense | |
| 2015 | |
$ | 173,000 | |
| 2016 | |
$ | 62,000 | |
| 2017 | |
$ | 62,000 | |
| 2018 | |
$ | 62,000 | |
| 2019 | |
$ | 50,949 | |
License
fees and royalty payments are expensed annually as incurred as the Company does not attribute any future benefits other than within
that period.
Note
4. Warrant Liabilities
Warrants
issued in connection with the Company’s June 2013 registered public offering contain provisions that protect holders from
a decline in the issue price of its common stock (or “down-round” provision) and contain net settlement provisions.
As a result, the Company accounts for these warrants as liabilities instead of equity instruments. Down-round provisions reduce
the exercise or conversion price of a warrant if the Company issues equity shares for a price that is lower than the exercise
or conversion price of the warrants. Net settlement provisions allow the holder of the warrant to surrender shares underlying
the warrant equal to the exercise price as payment of its exercise price, instead of exercising the warrant by paying cash. The
Company evaluates whether warrants to acquire its common stock contain provisions that protect holders from declines in the stock
price or otherwise could result in modification of the exercise price and/or shares to be issued under the respective warrant
agreements based on a variable that is not an input to the fair value of a “fixed for fixed” option. As a result of
the Company’s December 2014 registered public unit offering, the exercise price of warrants outstanding in connection with
the public offering completed in June 2013 was adjusted to $0.61 per share.
The
Company recognizes these warrants as liabilities at their fair value on the date of grant and remeasures them to fair value on
each reporting date.
The
Company recognized an initial warrant liability for the warrants issued in connection with the registered public offering completed
in June 2013 totaling $4,827,788, which was based on the June 25, 2013 closing price of a share of the Company’s common
stock as reported on OTC Markets of $0.96. During the year ended December 31, 2014, 143,004 shares of common were issued upon
586,081 warrants exercised on a cashless basis. On January 22, 2014, 250,000 warrants were exercised and on August 19, 2014, 336,081warrants
were exercised. The fair value of the warrants exercised, or $1,055,490 was reclassified from warrant liability to additional
paid-in capital on the respective exercise date. On December 31, 2014, the closing price of the Company’s common stock as
reported on OTC Markets was $0.98. Due to the fluctuations in the market value of the Company’s common stock from December 31,
2013 through December 31, 2014, the Company recognized a non-cash gain of $3,436,195 for the change in the fair value of the warrant
liability for 2014.
The
assumptions used in connection with the valuation of warrants issued utilizing the binomial method were as follows for the year
ended December 31, 2014 and 2013:
| | |
Initial
Measurement June
25,
2013 | | |
December
31, 2013 | | |
January
22, 2014 | | |
August
19, 2014 | | |
December
31, 2014 | |
| | |
| | |
| | |
| | |
| | |
| |
| Number of shares underlying
the warrants | |
| 5,416,851 | | |
| 5,309,438 | | |
| 5,309,438 | | |
| 5,059,438 | | |
| 4,723,357 | |
| Exercise price | |
$ | 1.65 | | |
$ | 1.65 | | |
$ | 1.65 | | |
$ | 1.65 | | |
$ | 0.61 | |
| Volatility | |
| 140 | % | |
| 135 | % | |
| 135 | % | |
| 130 | % | |
| 128 | % |
| Risk-free interest rate | |
| 1.49 | % | |
| 1.75 | % | |
| 1.30 | % | |
| 1.25 | % | |
| 1.38 | % |
| Expected dividend yield | |
| 0 | | |
| 0 | | |
| 0 | | |
| 0 | | |
| 0 | |
| Expected warrant life (years) | |
| 5.0 | | |
| 4.5 | | |
| 4.4 | | |
| 3.9 | | |
| 3.5 | |
| Stock Price | |
$ | 0.96 | | |
$ | 1.80 | | |
$ | 2.29 | | |
$ | 2.05 | | |
$ | 0.98 | |
Recurring
Level 3 Activity and Reconciliation
The
table below provides a reconciliation of the beginning and ending balances for the liability measured at fair value using significant
unobservable inputs (Level 3). The table reflects gains for the year ended December 31, 2014 for the financial liability categorized
as Level 3 as of December 31, 2014.
Fair Value
Measurements Using Significant Unobservable Inputs (Level 3):
| | |
December
31, 2013 | | |
Decrease
from Warrants Exercised in 2014 | | |
Decrease
in Fair Value | | |
December
31, 2014 | |
| Warrant
liability | |
$ | 8,281,247 | | |
$ | (1,055,490 | ) | |
$ | (3,436,195 | ) | |
$ | 3,789,562 | |
Recurring
Level 3 Activity and Reconciliation
The
table below provides a reconciliation of the beginning and ending balances for the liability measured at fair value using significant
unobservable inputs (Level 3). The table reflects losses for the year ended December 31, 2013 for the financial liability categorized
as Level 3 as of December 31, 2013.
Fair Value
Measurements Using Significant Unobservable Inputs (Level 3):
| | |
Initial
Measurement June
25,
2013 | | |
Decrease
from Warrants Exercised in 2013 | | |
Increase
in Fair Value | | |
December 31,
2013 | |
| Warrant
liability | |
$ | 4,827,788 | | |
$ | (201,311 | ) | |
$ | 3,654,770 | | |
$ | 8,281,247 | |
Note
5. Income Taxes
The
income tax benefit consisted of the following for the years ended December 31, 2014 and December 31, 2013:
| | |
2014 | | |
2013 | |
| Federal | |
$ | - | | |
$ | - | |
| State | |
| (616,872 | ) | |
| (750,356 | ) |
| Income tax benefit | |
$ | (616,872 | ) | |
$ | (750,356 | ) |
The
significant components of the Company’s deferred tax assets and liability at December 31, 2014 and 2013 are as follows:
| | |
2014 | | |
2013 | |
| Net operating loss carry
forwards | |
$ | 29,594,000 | | |
$ | 27,974,000 | |
| Orphan drug and research and development
credit carry forwards | |
| 3,556,000 | | |
| 2,986,000 | |
| Equity based compensation | |
| 2,049,000 | | |
| 3,183,000 | |
| Intangibles | |
| 2,140,000 | | |
| 127,000 | |
| Total | |
| 37,339,000 | | |
| 34,270,000 | |
| Valuation allowance | |
| (37,339,000 | ) | |
| (34,270,000 | ) |
| Income tax benefit | |
$ | - | | |
$ | - | |
At
December 31, 2014, the Company had NOL carry forwards of approximately $86,120,000 for federal tax purposes and approximately
$5,263,000 of New Jersey NOL carry forwards remaining after the sale of unused net operating loss carry forwards, portions of
which are currently expiring each year through 2034. In addition, the Company has $3,556,000 of various tax credits that expire
from 2018 to 2034. The Company may be able to utilize their NOLs to reduce future federal and state income tax liabilities. However,
these NOLs are subject to various limitations under Internal Revenue Code (“IRC”) Section 382. IRC Section 382 limits
the use of NOLs to the extent there has been an ownership change of more than 50 percentage points. In addition, the NOL carry
forwards are subject to examination by the taxing authority and could be adjusted or disallowed due to such exams. Although the
Company has not undergone an IRC Section 382 analysis, it is likely that the utilization of the NOLs may be substantially limited.
The
Company and one or more of its subsidiaries files income tax returns in the U.S. Federal jurisdiction, and various state and local
jurisdictions. The Company is no longer subject to Federal income tax assessment for years before 2011 for Federal and 2010 for
New Jersey income tax assessment. However, since the Company has incurred net operating losses in every tax year since inception,
all its income tax returns are subject to examination and adjustments by the Internal Revenue Service for at least three years
following the year in which the tax attributes are utilized.
The
net change in the valuation allowance for the years ended December 31, 2014 and 2013 was an increase of approximately $3,069,000
and $1,887,000, respectively, resulting primarily from net operating losses expiring and generated. As a result of the Company’s
continuing tax losses, the Company has recorded a full valuation allowance against a net deferred tax asset.
Reconciliations
of the difference between income tax benefit computed at the federal and state statutory tax rates and the provision for income
tax benefit for the years ended December 31, 2014 and 2013 was as follows:
| | |
2014 | | |
2013 | |
| Income tax loss at federal
statutory rate | |
| (34.00 | )% | |
| (34.00 | )% |
| State tax benefits, plus sale of NJ
NOLs, net of federal benefit | |
| (6.00 | ) | |
| (6.00 | ) |
| Subtotal | |
| (40.00 | ) | |
| (40.00 | ) |
| Valuation allowance | |
| 31.58 | | |
| 33.06 | |
| Income tax benefit | |
| (8.42 | )% | |
| (6.94 | )% |
During
the years ended December 31, 2014 and 2013, in accordance with the State of New Jersey’s Technology Business Tax Certificate
Program, which allowed certain high technology and biotechnology companies to sell unused net operating loss carryforwards to
other New Jersey-based corporate taxpayers based in New Jersey, the Company sold New Jersey net operating loss carryforwards,
resulting in the recognition of $616,872 and $750,356 of income tax benefit, net of transaction costs, respectively. There can
be no assurance as to the continuation or magnitude of this program in the future.
Note
6. Shareholders’ Equity
Preferred
Stock
The
Company has 350,000 shares of preferred stock authorized, none of which are issued or outstanding.
Common
Stock
The
following items represent transactions in the Company’s common stock for the year ended December 31, 2014:
| ● | In
January 2014, the Company issued 77,889 shares of common stock in connection with the
cashless exercise of 250,000 stock warrants; |
| ● | In
March 2014, the Company issued 76,932 shares of common stock pursuant to the Lincoln
Park facility; |
| ● | In
April 2014, the Company issued 76,907 shares of common stock pursuant to the Lincoln
Park facility; |
| ● | In
May 2014, the Company issued 43,067 shares of common stock upon the execution of an agreement
to evaluate specific oncology technology; |
| ● | In
May 2014, the Company issued 29,172 shares of common stock upon the exercise of vested
stock options; |
| ● | In
July 2014, the Company issued 76,904 shares of common stock pursuant to the Lincoln Park
facility; |
| ● | In
July 2014, the Company issued 7,500 shares of common stock upon the exercise of vested
stock options; |
| ● | In
August 2014, the Company issued 65,115 shares of common stock with the cashless exercise
of 336,081 stock warrants; |
| ● | In
September 2014, the Company issued 1,849,113 shares of common stock in connection with
the Hy BioPharma Acquisition of in process research and development. |
| ● | In
December 2014, the Company issued 1,886,530 shares of common stock and 1,169,318 warrants
pursuant to a registered direct unit offering of common stock and warrants. The Company
received net proceeds of $1,937,894 from this offering. |
| ● | In
four separate transactions, the Company issued 121,000 shares of common stock as partial
consideration for services performed. |
The
following items represent transactions in the Company’s common stock for the year ended December 31, 2013:
| ● | In
April 2013, the Company issued 1,034,483 shares of common stock related to the execution
of an Exclusive Channel Collaboration agreement with Intrexon Corporation (see Note 9). |
| ● | In
June 2013, the Company issued 6,773,995 shares of common stock pursuant to a registered
direct unit offering of common stock and warrants. |
| ● | In
October 2013, the Company issued 107,143 shares of common stock for stock warrants exercised.
|
| ● | In
November, the Company issued 383,370 shares of common stock pursuant to the Lincoln Park
Capital equity facility. |
| ● | In
two separate transactions, the Company issued 103,439 shares of common stock for stock
options exercised. |
| ● | In
five separate transactions, the Company issued 55,104 shares of common stock as part
of consideration for services performed. |
Warrants
During
the year ended December 31, 2014, the Company issued warrants to purchase 1,169,318 shares of common stock pursuant to a registered
direct offering of common stock and warrants.
During
the year ended December 31, 2013, the Company issued warrants to purchase 5,416,581 shares of common stock pursuant to a registered
direct offering of common stock and warrants. Additionally, the Company issued 5,000 warrants to a consultant in exchange for
services.
A
gain of $3,436,195, related to the warrants issued in the June 2013 registered direct offering, was recognized for the change
in the fair value of the warrant liability during the year ended December 31, 2014. A charge of $3,654,770 was incurred during
the year ended December 31, 2013 for the change in the fair value of the warrant liability. Additionally, warrant expense charges
of $4,775 were recorded during the years ended December 31, 2014 and 2013.
Equity
Line
In
November 2013, the Company entered into a common stock purchase agreement with Lincoln Park Capital Fund, LLC (“Lincoln
Park”). The Lincoln Park equity facility allows the Company to require Lincoln Park to purchase up to 75,000 shares (“Regular
Purchase”) of the Company’s common stock every two business days, up to an aggregate of $10.6 million over approximately
a 36-month period depending on certain conditions, including the quoted market price of the Company’s common stock on such
date. The purchase price for the Regular Purchase shall be equal to the lesser of (i) the lowest sale price of the common shares
during the purchase date, or (ii) the average of the three lowest closing sale prices of common shares during the twelve business
days prior to the purchase date. Each Regular Purchase shall not exceed $750,000. Furthermore, for each additional purchase by
Lincoln Park, additional commitment shares in commensurate amounts up to a total of 122,070 shares will be issued based upon the
relative proportion of the aggregate amount of $10.0 million. The Regular Purchase amount may be increased up to 100,000 shares
of common stock if the closing price of the common shares is not below $2.50. In addition to the Regular Purchase and provided
that the closing price of the common shares is not below $1.50 on the purchase date, the Company in its sole discretion may direct
Lincoln Park on each purchase date to purchase on the next stock trading day (“Accelerate Purchase Date”) additional
shares of Company stock up to the lesser of (i) two times the number of shares purchased following a Regular Purchase or (ii)
30% of the trading volume of shares traded on the Accelerated Purchase Date as a price equal to the lesser of the closing sale
price on the Accelerated Purchase Date or 95% of the Accelerated Purchase Date’s volume weighted average price. During the
year ended December 31, 2013, the Company received gross proceeds of $600,000 for the issuance of 383,370 shares of common stock
to Lincoln Park. Associated costs of $71,949 were incurred resulting in net proceeds of $528,051.
During
the year ended December 31, 2014, in three separate transactions, the Company issued 230,743 shares of common stock receiving
net proceeds of $470,475.
Note
7. Stock Option Plans and Warrants to Purchase Common Stock
Stock
Option Plans
The
Amended and Restated 1995 Omnibus Plan was replaced by the 2005 Equity Incentive Plan and is divided into four separate equity
programs:
| 1) | the
Discretionary Option Grant Program, under which eligible persons may, at the discretion
of the Plan Administrator, be granted options to purchase shares of common stock, |
| 2) | the
Salary Investment Option Grant Program, under which eligible employees may elect to have
a portion of their base salary invested each year in options to purchase shares of common
stock, |
| 3) | the
Automatic Option Grant Program, under which eligible nonemployee Board members will automatically
receive options at periodic intervals to purchase shares of common stock, and |
| 4) | the
Director Fee Option Grant Program, under which non-employee Board members may elect to
have all, or any portion, of their annual retainer fee otherwise payable in cash applied
to a special option grant. |
The
2005 Equity Incentive Plan (“2005 Plan”) is divided into four separate equity programs:
| 1) | the
Discretionary Option Grant Program, under which eligible persons may, at the discretion
of the Plan Administrator, be issued common stock or granted options to purchase shares
of common stock, |
| 2) | the
Salary Investment Option Grant Program, under which eligible employees may elect to have
a portion of their base salary invested each year in options to purchase shares of common
stock, |
| 3) | the
Automatic Option Grant Program, under which eligible nonemployee Board members will automatically
receive options at periodic intervals to purchase shares of common stock, and |
| 4) | the
Director Fee Option Grant Program, under which non-employee Board members may elect to
have all, or any portion, of their annual retainer fee otherwise payable in cash applied
to a special option grant. |
In
addition, under the 2005 Plan, the Board may elect to pay certain consultants, directors, and employees in common stock. The 2005
Plan was amended in September 2007 to increase the number of options available under the plan to 1,000,000, in 2010 to increase
the number of shares under the plan to 1,750,000 and again in 2013 to increase the number shares available under the plan to 3,000,000.
The
table below only accounts for transactions occurring as part of the 2005 Plan.
| | |
December
31, | |
| | |
2014 | | |
2013 | |
| Shares
available for grant at beginning of year | |
| 672,485 | | |
| 129,711 | |
| Increase
in shares available for the plan | |
| - | | |
| 1,250,000 | |
| Options
granted | |
| (637,495 | ) | |
| (791,100 | ) |
| Options
forfeited or expired | |
| 149,055 | | |
| 83,874 | |
| | |
| | | |
| | |
| Shares
available for grant at end of year | |
| 184,045 | | |
| 672,485 | |
The
total option activity for the 1995 Omnibus Plan and the amended 2005 Plan for the years ended December 31, 2014 and 2013 was as
follows:
| | |
Options | | |
Weighted
Average Options
Exercise Price | |
| Balance
at December 31, 2012 | |
| 1,457,724 | | |
$ | 3.19 | |
| Granted | |
| 791,100 | | |
| 1.35 | |
| Exercised | |
| (103,439 | ) | |
| 0.57 | |
| Forfeited | |
| (93,874 | ) | |
| 2.84 | |
| Balance
at December 31, 2013 | |
| 2,051,511 | | |
$ | 2.63 | |
| Granted | |
| 637,495 | | |
| 1.79 | |
| Exercised | |
| (36,672 | ) | |
| 0.77 | |
| Forfeited | |
| (164,055 | ) | |
| 3.13 | |
| Balance
at December 31, 2014 | |
| 2,488,279 | | |
$ | 2.40 | |
As
of December 31, 2014, there were 1,875,609 options exercisable with a weighted average exercise price of $2.64, a weighted average
remaining contractual term of 6.9 years and an intrinsic value of $196,655. The intrinsic value of options exercised during the
years ended December 31, 2014 and 2013 was $47,241 and $56,750, respectively. As of December 31, 2014, there were 2,488,279 options
outstanding and expected to vest with a weighted average exercise price of $2.40, weighted average remaining term of 6.9 years
and an intrinsic value of $215,103. The aggregate intrinsic value represents the total pre-tax intrinsic value (the difference
between the closing price of our common stock on the last trading day on December 31, 2014 and the exercise price, multiplied
by the number of in-the-money options) what would have been received by the option holders had all option holders exercised their
options on December 31, 2014. This amount changes based on the fair market value of our common stock.
The
Company awarded 637,495 and 791,100 stock options to new employees and existing Board members during the years ended 2014
and 2013, respectively. During the year ended 2014, under the 2005 Equity Incentive plan, 569,000 option grants were issued to
employees and 68,495 option grants were issued to Board members.
The weighted-average
exercise price, by price range, for outstanding options to purchase common stock at December 31, 2014 was:
Price
Range | |
Weighted
Average Remaining
Contractual Life in Years | |
Outstanding
Options | | |
Exercisable
Options | |
| $0.30-$2.20 | |
7.7 | |
| 1,805,755 | | |
| 1,193,081 | |
| $2.26-$4.10 | |
6.9 | |
| 174,774 | | |
| 174,778 | |
| $4.64-$9.40 | |
3.7 | |
| 507,750 | | |
| 507,750 | |
| Total | |
6.9 | |
| 2,488,279 | | |
| 1,875,609 | |
The
Company’s stock-based compensation for the years ended December 31, 2014 and 2013 was $720,150 and $803,060, respectively.
At December 31, 2014, the total compensation cost for stock options not yet recognized was approximately $1,009,941 and will be
expensed over the next three years.
Warrants
to Purchase Common Stock
Warrant
activity for the years ended December 31, 2014 and 2013 was as follows:
| | |
Warrants | | |
Weighted
Average Warrant
Exercise Price | |
| Balance
at December 31, 2012 | |
| 2,843,338 | | |
$ | 3.13 | |
| Granted | |
| 5,421,581 | | |
| 1.65 | |
| Exercised | |
| (107,143 | ) | |
| 1.65 | |
| Expired/Cancelled | |
| (1,250 | ) | |
| 15.00 | |
| Balance
at December 31, 2013 | |
| 8,156,526 | | |
$ | 2.17 | |
| Granted | |
| 1,169,318 | | |
| 1.48 | |
| Exercised | |
| (586,081 | ) | |
| 1.65 | |
| Expired/Cancelled | |
| (1,470,263 | ) | |
| 3.49 | |
| Balance
at December 31, 2014 | |
| 7,269,500 | | |
$ | 1.15 | |
During
the year ended 2014, the Company issued warrants to purchase 1,169,318 shares of common stock, with an exercise price of $1.48,
pursuant to a registered direct offering of common stock and warrants. Warrants of 1,470,263 either expired or were cancelled
by the Company with an average exercise price of $3.49 and 586,081 warrants were exercised with an exercise price of $1.65.
The
weighted-average exercise price, by price range, for outstanding warrants at December 31, 2014 was:
Price
Range | |
Weighted
Average Remaining
Contractual Life in Years | |
Outstanding
Warrants | | |
Exercisable
Warrants | |
| $.53-$2.05 | |
3.7 | |
| 6,672,548 | | |
| 6,672,548 | |
| $5.12-$6.06 | |
0.5 | |
| 596,952 | | |
| 596,952 | |
| Total | |
3.4 | |
| 7,269,500 | | |
| 7,269,500 | |
During
the year ended December 31, 2015, warrants to purchase 596,952 shares of the Company’s common stock will expire.
Note
8. Concentrations
At
December 31, 2014 and 2013, the Company had deposits in major financial institutions that exceeded the amount under protection
by the Securities Investor Protection Corporation (“SIPC”). Currently, the Company is covered up to $1,000,000 by
the SIPC. The excess amount at December 31, 2014 was $4,525,094.
Note
9. Commitments and Contingencies
The
Company has commitments of approximately $375,000 at December 31, 2014 for several licensing agreements with consultants and universities.
Additionally, the Company has collaboration and license agreements, which upon clinical or commercialization success, may require
the payment of milestones of up to $7.9 million and/or royalties up to 6% of net sales of covered products, if and when achieved.
However, there can be no assurance that clinical or commercialization success will occur.
In
December 2014, the Company entered into a lease agreement through May 31, 2018 for existing and expanded office space. The rent
for the first 12 months is approximately $12,300 per month, or approximately $20.85 per square foot. This rent increases to approximately
$12,375 per month, or approximately $20.95 per square foot, for the next 12 months and approximately $12,460 per month, or approximately
$21.13 per square foot for the remainder of the lease.
On
September 3, 2014, the Company entered into an asset purchase agreement with Hy Biopharma, Inc. to which the Company acquired
certain intangible assets, properties and rights of Hy Biopharma related to the development of Hy BioPharma’s synthetic
hypericin product. As consideration for the assets acquired, the company paid $250,000 in cash and issued 1,849,113 shares of
common stock with a fair value based on the Company’s stock price on the date of grant of $3,750,000. These amounts are
charged to research and development expense as the assets will be used in the Company’s R&D activities and do not have
alternative future use pursuant to Generally Accepted Accounting Principles in the United States. Provided all future success-oriented
milestones are attained, the Company will be required to make additional payments of up to $10.0 million, if and when achieved.
Payments will be payable in restricted securities of the Company.
On
April 27, 2013, the Company entered into an exclusive channel collaboration agreement with Intrexon (the “Channel Agreement”)
to use Intrexon’s advanced human antibody discovery, isolation and production technologies for the development of human
monoclonal antibody therapies for a new biodefense application targeting melioidosis. The Channel Agreement grants an exclusive
worldwide license to use specified patents and other intellectual property of Intrexon in connection with the research, development,
use, importing, manufacture, sale and offer for sale of products for the treatment of melioidosis through the use of exogenously
produced human recombinant monoclonal antibodies. The Channel Agreement, upon clinical or commercialization success, may require
the payment of certain milestones up to $7 million, if and when achieved.
In
February 2007, the Company’s Board of Directors authorized the issuance of 50,000 shares to Dr. Schaber immediately prior
to the completion of a transaction, or series or a combination of related transactions negotiated by its Board of Directors whereby,
directly or indirectly, a majority of its capital stock or a majority of its assets are transferred from the Company and/or its
stockholders to a third party. The amended agreement with Dr. Schaber includes its obligation to issue such shares if such event
occurs.
As a result
of the above agreements, the Company has future contractual obligations over the next five years as follows:
Year | |
Research
and Development | | |
Property
and
Other Leases | | |
Total | |
| 2015 | |
$ | 75,000 | | |
$ | 130,000 | | |
$ | 205,000 | |
| 2016 | |
| 75,000 | | |
| 157,000 | | |
| 232,000 | |
| 2017 | |
| 75,000 | | |
| 152,000 | | |
| 227,000 | |
| 2018 | |
| 75,000 | | |
| 51,000 | | |
| 126,000 | |
| 2019 | |
| 75,000 | | |
| - | | |
| 75,000 | |
| Total | |
$ | 375,000 | | |
$ | 490,000 | | |
$ | 865,000 | |
Note
10. Operating Segments
The
Company maintains two active operating segments: BioTherapeutics and Vaccines/BioDefense. Each segment includes an element
of overhead costs specifically associated with its operations, with its corporate shared services group responsible for support
functions generic to both operating segments.
| | |
For
the Years Ended December 31, | |
| | |
2014 | | |
2013 | |
| Revenues | |
| | |
| |
| Vaccines/BioDefense | |
$ | 6,756,388 | | |
$ | 3,003,822 | |
| BioTherapeutics | |
| 286,628 | | |
| 220,330 | |
| Total | |
$ | 7,043,016 | | |
$ | 3,224,152 | |
| | |
| | | |
| | |
| Income (loss) from
Operations | |
| | | |
| | |
| Vaccines/BioDefense | |
$ | 807,164 | | |
$ | (1,666,130 | ) |
| BioTherapeutics | |
| (7,674,381 | ) | |
| (3,069,998 | ) |
| Corporate | |
| (3,894,132 | ) | |
| (2,420,414 | ) |
| Total | |
$ | (10,761,349 | ) | |
$ | (7,156,542 | ) |
| | |
| | | |
| | |
| Amortization and
Depreciation Expense | |
| | | |
| | |
| Vaccines/BioDefense | |
$ | 39,625 | | |
$ | 37,981 | |
| BioTherapeutics | |
| 199,196 | | |
| 190,033 | |
| Corporate | |
| 6,966 | | |
| 2,057 | |
| Total | |
$ | 245,787 | | |
$ | 230,071 | |
| | |
| | | |
| | |
| Interest Income | |
| | | |
| | |
| Corporate | |
$ | 1,310 | | |
$ | 1,960 | |
| | |
| | | |
| | |
| Stock-Based Compensation | |
| | | |
| | |
| Vaccines/BioDefense | |
$ | 114,920 | | |
$ | 80,432 | |
| BioTherapeutics | |
| 193,926 | | |
| 250,431 | |
| Corporate | |
| 411,304 | | |
| 472,197 | |
| Total | |
$ | 720,150 | | |
$ | 803,060 | |
| | |
As
of December 31, | |
| | |
2014 | | |
2013 | |
| Identifiable Assets | |
| | |
| |
| Vaccines/BioDefense | |
$ | 1,025,220 | | |
$ | 1,870,414 | |
| BioTherapeutics | |
| 204,308 | | |
| 386,721 | |
| Corporate | |
| 5,724,720 | | |
| 6,008,320 | |
| Total | |
$ | 6,954,248 | | |
$ | 8,265,455 | |
Note
11. Subsequent Events
Since January
1, 2015, the Company has received proceeds of $732,010 pursuant to 1,165,786 stock warrant exercises.
REPORT
OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
The
Board of Directors and Shareholders
Soligenix,
Inc.
We
have audited the accompanying consolidated balance sheets of Soligenix, Inc. and subsidiaries (the “Company”) as of
December 31, 2014 and 2013, and the related consolidated statements of operations, shareholders’ deficiency, and cash flows
for each of the years in the two-year period ended December 31, 2014. The financial statements are the responsibility of the Company’s
management. Our responsibility is to express an opinion on these financial statements based on our audits.
We
conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those
standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are
free of material misstatement. The Company is not required to have, nor were we engaged to perform, an audit of its internal control
over financial reporting. Our audits included consideration of internal control over financial reporting as a basis for designing
audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness
of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit includes examining,
on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing
the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement
presentation. We believe that our audits provide a reasonable basis for our opinion.
In
our opinion, the financial statements referred to above present fairly, in all material respects, the consolidated financial position
of Soligenix, Inc. and subsidiaries as of December 31, 2014 and 2015, and the consolidated results of their operations and their
cash flows for each of the years in the two-year period ended December 31, 2014, in conformity with accounting principles generally
accepted in the United States of America.
/s/
EisnerAmper LLP
Jenkintown,
PA
March
25, 2015
SOLIGENIX,
INC.
8,661,603
SHARES OF COMMON STOCK
PROSPECTUS
___________________________________,
2015
PART
II
INFORMATION
NOT REQUIRED IN PROSPECTUS
| ITEM
13. |
Other
Expenses of Issuance and Distribution. |
The
following table sets forth the estimated costs and expenses of the Registrant in connection with the offering described in the
registration statement. All of the amounts shown are estimated except for the Securities and Exchange Commission (the “SEC”)
registration fee.
| SEC registration fee | |
$ | 1,737 | |
| Legal fees and expenses | |
$ | 65,000 | |
| Accounting fees and expenses | |
$ | 10,000 | |
| Miscellaneous | |
$ | 1,763 | |
| | |
| | |
| TOTAL | |
$ | 78,500 | |
| ITEM
14. |
Indemnification
of Directors and Officers. |
Section
145(a) of the Delaware General Corporation Law provides, in general, that a corporation may indemnify any person who was or is
a party or is threatened to be made a party to any threatened, pending or completed action, suit or proceeding, whether civil,
criminal, administrative or investigative (other than an action by or in the right of the corporation), because he or she is or
was a director, officer, employee or agent of the corporation, or is or was serving at the request of the corporation as a director,
officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise, against expenses (including
attorneys’ fees), judgments, fines and amounts paid in settlement actually and reasonably incurred by the person in connection
with such action, suit or proceeding, if he or she acted in good faith and in a manner he or she reasonably believed to be in
or not opposed to the best interests of the corporation and, with respect to any criminal action or proceeding, had no reasonable
cause to believe his or her conduct was unlawful.
Section
145(b) of the Delaware General Corporation Law provides, in general, that a corporation may indemnify any person who was or is
a party or is threatened to be made a party to any threatened, pending or completed action or suit by or in the right of the corporation
to procure a judgment in its favor because the person is or was a director, officer, employee or agent of the corporation, or
is or was serving at the request of the corporation as a director, officer, employee or agent of another corporation, partnership,
joint venture, trust or other enterprise, against expenses (including attorneys’ fees) actually and reasonably incurred
by the person in connection with the defense or settlement of such action or suit if he or she acted in good faith and in a manner
he or she reasonably believed to be in or not opposed to the best interests of the corporation, except that no indemnification
shall be made with respect to any claim, issue or matter as to which he or she shall have been adjudged to be liable to the corporation
unless and only to the extent that the Court of Chancery or other adjudicating court determines that, despite the adjudication
of liability but in view of all of the circumstances of the case, he or she is fairly and reasonably entitled to indemnity for
such expenses which the Court of Chancery or other adjudicating court shall deem proper.
Section
145(g) of the Delaware General Corporation Law provides, in general, that a corporation may purchase and maintain insurance on
behalf of any person who is or was a director, officer, employee or agent of the corporation, or is or was serving at the request
of the corporation as a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other
enterprise against any liability asserted against such person and incurred by such person in any such capacity, or arising out
of his or her status as such, whether or not the corporation would have the power to indemnify the person against such liability
under Section 145 of the Delaware General Corporation Law.
Section
102(b)(7) of the Delaware General Corporation Law grants the Company the power to limit the personal liability of its directors
to the Company or its stockholders for monetary damages for breach of a fiduciary duty. Article X of the Company's Certificate
of Incorporation, as amended, provides for the limitation of personal liability of the directors of the Company as follows:
"A
Director of the Corporation shall have no personal liability to the corporation or its stockholders for monetary damages for breach
of his fiduciary duty as a Director; provided, however, this Article shall not eliminate or limit the liability of a Director
(i) for any breach of the Director's duty of loyalty to the Corporation or its stockholders; (ii) for acts or omissions not in
good faith or which involve intentional misconduct or a knowing violation of law; (iii) for the unlawful payment of dividends
or unlawful stock repurchases under Section 174 of the General Corporation Law of the State of Delaware; or (iv) for any transaction
from which the Director derived an improper personal benefit. If the General Corporation Law is amended after approval by the
stockholders of this Article to authorize corporate action further eliminating or limiting the personal liability of directors,
then the liability of a director of the Corporation shall be eliminated or limited to the fullest extent permitted by the General
Corporation Law of the State of Delaware, as so amended."
Article
VIII of the Company's Bylaws, as amended and restated, provide for indemnification of directors and officers to the fullest extent
permitted by Section 145 of the Delaware General Corporation Law.
The
Company has a directors' and officers' liability insurance policy.
The
above discussion is qualified in its entirety by reference to the Company's Certificate of Incorporation and Bylaws.
| ITEM
15. |
Recent
Sales of Unregistered Securities. |
On
January 10, 2013, the Company issued 11,063 shares of common stock to a consultant as partial consideration for services performed.
The per share closing price of the Company’s common stock on December 30, 2012 was $0.60. The issuance of the shares was
exempt from registration pursuant to Section 4(a)(2) of the Securities Act of 1933, as amended.
On
March 13, 2013, the Company issued 15,000 shares of common stock to a vendor as partial consideration for services performed.
The per share closing price of the Company’s common stock on March 13, 2013 was $1.75. The issuance of the shares was exempt
from registration pursuant to Section 4(a)(2) of the Securities Act of 1933, as amended.
On
April 12, 2013, the Company issued 2,041 shares of its common stock to a consultant as partial consideration for services performed.
The per share closing price of the Company’s common stock on March 28, 2013 was $1.47, which was the date on which the liability
was recognized. The issuance of the shares was exempt from registration pursuant to Section 4(a)(2) of the Securities Act of 1933,
as amended.
On
April 27, 2013, the Company issued 1,034,483 shares of its common stock to Intrexon as consideration for the execution and delivery
of a collaboration agreement. The closing price of the Company’s common stock on April 26, 2013 was $1.45 per share. The
issuance of the shares was exempt from registration pursuant to Section 4(a)(2) of the Securities Act of 1933, as amended, as
a transaction not involving a public offering.
On
August 22, 2013, the Company issued 15,000 shares of its common stock to a consultant as partial consideration for services performed.
The per share closing price of the Company’s common stock on August 22, 2013 was $1.58, which was the date on which the
liability was recognized. The issuance of these shares was exempt from registration pursuant to Section 4(a)(2) of the Securities
Act of 1933, as amended. The vendor is knowledgeable, sophisticated and experienced in making investment decisions of this kind
and received adequate information about the Company or had adequate access, including through the vendor’s business relationship
with the Company, to information about the Company.
On
October 15, 2013, the Company issued 12,000 shares of its common stock to a consultant as partial consideration for services performed.
The per share closing price of the Company’s common stock on October 15, 2013 was $1.58, which was the date on which the
liability was recognized. The issuance of these shares was exempt from registration pursuant to Section 4(a)(2) of the Securities
Act of 1933, as amended. The vendor is knowledgeable, sophisticated and experienced in making investment decisions of this kind
and received adequate information about the Company or had adequate access, including through the vendor’s business relationship
with the Company, to information about the Company.
On
November 18, 2013, the Company entered into a purchase agreement with Lincoln Park Capital Fund, LLC (“Lincoln Park”).
Pursuant to the terms of the agreement, the Company may require Lincoln Park to purchase between 75,000 and 100,000 shares of
common stock depending on certain conditions, up to a total of $10,600,000 over approximately a 36-month period. The purchase
price of the shares of common stock will be based on the market price of our common stock immediately preceding the time of sale
as computed under the purchase agreement without any fixed discount. The Company does not have the right to require Lincoln Park
to purchase shares of common stock in the event that the price of the common stock is less than $1.00 per share.
Pursuant
to the purchase agreement, the Company issued to Lincoln Park 97,656 shares of common stock as a partial commitment fee, and 285,714
shares of common stock for an aggregate price of $600,000. Since November 2013, the Company has sold Lincoln Park 825,000 more
shares of common stock for an aggregate price of approximately $1.6 million and issued to Lincoln Park 19,354 additional shares
of common stock as a commitment fee. Such securities were issued pursuant to an exemption provided by Section 4(a)(2) of the Securities
Act of 1933, as amended, and Rule 506 of Regulation D promulgated thereunder.
On
January 2, 2014, the Company issued 6,000 shares of its common stock to a consultant as partial consideration for services performed.
The per share closing price of the Company’s common stock on January 2, 2014 was $1.99. The issuance of these shares was
exempt from registration pursuant to Section 4(a)(2) of the Securities Act of 1933, as amended. The vendor is knowledgeable, sophisticated
and experienced in making investment decisions of this kind and received adequate information about the Company or had adequate
access, including through the vendor’s business relationship with the Company, to information about the Company.
On
February 21, 2014, the Company issued 50,000 shares of its common stock to a consultant as partial consideration for services
performed. The per share closing price of the Company’s common stock on February 21, 2014 was $2.19. The issuance of these
shares was exempt from registration pursuant to Section 4(a)(2) of the Securities Act of 1933, as amended. The vendor is knowledgeable,
sophisticated and experienced in making investment decisions of this kind and received adequate information about the Company
or had adequate access, including through the vendor’s business relationship with the Company, to information about the
Company.
On
February 24, 2014, the Company issued 15,000 shares of its common stock to a consultant as partial consideration for services
performed. The per share closing price of the Company’s common stock on February 24, 2014 was $2.14. The issuance of these
shares was exempt from registration pursuant to Section 4(a)(2) of the Securities Act of 1933, as amended. The vendor is knowledgeable,
sophisticated and experienced in making investment decisions of this kind and received adequate information about the Company
or had adequate access, including through the vendor’s business relationship with the Company, to information about the
Company.
On
May 6, 2014, the Company issued 43,067 shares of its common stock upon execution of an option agreement to purchase certain assets
related to the development of a synthetic hypericin product candidate for the treatment of cutaneous T-cell lymphoma. The per
share closing price of the Company’s common stock on May 6, 2014 was $1.98. The issuance of these shares was exempt from
registration pursuant to Section 4(a)(2) of the Securities Act of 1933, as amended. The vendor is knowledgeable, sophisticated
and experienced in making investment decisions of this kind and received adequate information about the Company or had adequate
access, including through the vendor’s business relationship with the Company, to information about the Company.
On
August 21, 2014, the Company issued 50,000 shares of its common stock to a consultant as partial consideration for services performed.
The per share closing price of the Company’s common stock on August 21, 2014 was $2.05. The issuance of these shares was
exempt from registration pursuant to Section 4(a)(2) of the Securities Act of 1933, as amended. The vendor is knowledgeable, sophisticated
and experienced in making investment decisions of this kind and received adequate information about the Company or had adequate
access, including through the vendor’s business relationship with the Company, to information about the Company.
On
September 3, 2014, the Company issued 1,849,113 shares of its common stock as partial payment for the purchase of certain assets
related to the development of a synthetic hypericin product candidate for the treatment of cutaneous T-cell lymphoma. The per
share closing price of the Company’s common stock on September 3, 2014 was $2.04. The issuance of these shares was exempt
from registration pursuant to Section 4(a)(2) of the Securities Act of 1933, as amended. The vendor is knowledgeable, sophisticated
and experienced in making investment decisions of this kind and received adequate information about the Company or had adequate
access, including through the vendor’s business relationship with the Company, to information about the Company.
On
January 7, 2015, the Company issued 6,000 shares of its common stock valued at $1.21 per share to a vendor as consideration for
services rendered. The per share closing price of the Company’s common stock on January 7, 2015 was $1.12. The issuance
of these shares was exempt from registration pursuant to Section 4(a)(2) of the Securities Act of 1933, as amended. The vendor
is knowledgeable, sophisticated and experienced in making investment decisions of this kind and received adequate information
about the Company or had adequate access, including through the vendor’s business relationship with the Company, to information
about the Company.
On
January 12, 2015, the Company issued 10,000 shares of its common stock valued at $1.21 per share to a vendor as consideration
for services rendered. The per share closing price of the Company’s common stock on January 12, 2015 was $1.15. The issuance
of these shares was exempt from registration pursuant to Section 4(a)(2) of the Securities Act of 1933, as amended. The vendor
is knowledgeable, sophisticated and experienced in making investment decisions of this kind and received adequate information
about the Company or had adequate access, including through the vendor’s business relationship with the Company, to information
about the Company.
On
February 15, 2015, the Company issued 50,000 shares of its common stock valued at $1.64 per share to a vendor as consideration
for services rendered. The per share closing price of the Company’s common stock on February 13, 2015 was $1.46. The issuance
of these shares was exempt from registration pursuant to Section 4(a)(2) of the Securities Act of 1933, as amended. The vendor
is knowledgeable, sophisticated and experienced in making investment decisions of this kind and received adequate information
about the Company or had adequate access, including through the vendor’s business relationship with the Company, to information
about the Company.
On
April 16, 2015, the Company issued 10,000 shares of its common stock valued at $1.53 per share to a consultant as consideration
for services rendered. The per share closing price of the Company’s common stock on April 15, 2015 was $1.79. The issuance
of these shares was exempt from registration pursuant to Section 4(a)(2) of the Securities Act of 1933, as amended. The consultant
is knowledgeable, sophisticated and experienced in making investment decisions of this kind and received adequate information
about the Company or had adequate access, including through the consultant’s business relationship with the Company, to
information about the Company.
On
May 12, 2015, the Company issued 1,243 shares of its common stock valued at $1.69 per share to a consultant as consideration for
services rendered. The per share closing price of the Company’s common stock on May 11, 2015 was $1.51. The issuance of
these shares was exempt from registration pursuant to Section 4(a)(2) of the Securities Act of 1933, as amended. The consultant
is knowledgeable, sophisticated and experienced in making investment decisions of this kind and received adequate information
about the Company or had adequate access, including through the consultant’s business relationship with the Company, to
information about the Company.
On
July 29, 2015, the Company entered into purchase agreements with Kodiak Capital Group, LLC (“Kodiak Capital”), Kingsbrook
Opportunities Master Fund LP (“Kingsbrook”) and River North Equity, LLC (“River North”). Pursuant to the
terms of the agreements, the Company may require Kodiak Capital, Kingsbrook and River North to purchase up to a total of $5 million,
$4 million and $1 million worth of shares of common stock of the Company, respectively, until December 31, 2016, provided certain
conditions are met. The purchase price of the shares of common stock will be equal to 80% of the lowest daily volume weighted
average price of the Company’s common stock for the five consecutive trading days immediately following the Company’s
request for the purchase of the shares. Such securities will be issued pursuant to an exemption provided by Section 4(a)(2) of
the Securities Act of 1933, as amended, and Rule 506 of Regulation D promulgated thereunder. Each of the investors represented
to the Company that it (i) is an “accredited investor” as defined in Rule 501(a) of Regulation D promulgated under
the Securities Act of 1933, as amended, (ii) is knowledgeable, sophisticated and experienced in making investment decisions of
this kind, and (iii) has had adequate access to information about the Company.
| 2.1 |
Agreement
and Plan of Merger, dated May 10, 2006 by and among the Company, Corporate Technology Development, Inc., Enteron Pharmaceuticals,
Inc. and CTD Acquisition, Inc. (incorporated by reference to Exhibit 2.1 included in our Registration Statement on Form SB-2
(File No. 333-133975) filed on May 10, 2006). |
| |
|
| 3.1 |
Second
Amended and Restated Certificate of Incorporation (incorporated by reference to Exhibit 3.1 included in our current report
on Form 8-K filed on June 22, 2012). |
| |
|
| 3.2 |
By-laws
(incorporated by reference to Exhibit 3.1 included in our Quarterly Report on Form 10-QSB, as amended, for the fiscal quarter
ended June 30, 2003). |
| |
|
| 4.1 |
Rights
Agreement dated June 22, 2007, between the Company and American Stock Transfer & Trust Company, as Rights Agent (incorporated
by reference to Exhibit 4.1 included in our current report on Form 8-K filed on June 22, 2007). |
| |
|
| 4.2 |
Form
of Right Certificate (incorporated by reference to Exhibit 4.2 included in our current report on Form 8-K filed on June 22,
2007). |
| |
|
| 4.3 |
Form
of Warrant issued to each investor in the January 2009 private placement (incorporated by reference to Exhibit 4.18 included
in our Registration Statement on Form S-1 (File No. 333-149239) filed on February 14, 2008). |
| |
|
| 4.4 |
Form
of Warrant issued to each investor in the September 2009 private placement (incorporated by reference to Exhibit 10.2 included
in our current report on Form 8-K filed on September 29, 2009). |
| |
|
| 4.5 |
Warrant
dated April 19, 2010, issued to Fusion Capital Fund II, LLC (incorporated by reference to Exhibit 4.10 included in our Post-Effective
Amendment to Registration Statement on Form S-1 filed on April 20, 2010). |
| |
|
| 4.6 |
Form
of Common Stock Purchase Warrant issued to each investor in the June 2010 private placement (incorporated by reference to
Exhibit 10.2 included in our current report on Form 8-K filed on June 18, 2010). |
| |
|
| 4.7
|
Warrant
dated December 20, 2012 and issued to Sigma-Tau to purchase 357,069 shares of the Company’s common stock (incorporated
by reference to Exhibit 10.2 of our current report on Form 8-K filed on December 27, 2012). |
| |
|
| 4.8
|
Warrant
dated December 20, 2012 and issued to SINAF S.A. to purchase 87,804 shares of the Company’s common stock (incorporated
by reference to Exhibit 10.3 of our current report on Form 8-K filed on December 27, 2012). |
| |
|
| 4.9
|
Warrant
dated December 20, 2012 and issued to McDonald to purchase 280,000 shares of the Company’s common stock (incorporated
by reference to Exhibit 10.6 of our current report on Form 8-K filed on December 27, 2012). |
| |
|
| 4.10
|
Form
of Common Stock Purchase Warrant issued to each investor in the June 2013 registered public offering (incorporated by reference
to Exhibit 10.3 included in our current report on Form 8-K filed on June 24, 2013). |
| |
|
| 4.11
|
Form
of Warrant issued to Maxim Group LLC (incorporated by reference to Exhibit 10.4 included in our current report on Form 8-K
filed on June 24, 2013). |
| |
|
| 4.12 |
Form
of Warrant to Purchase Common Stock issued to each investor in the December 2014 registered public offering (incorporated
by reference to Exhibit 4.12 included in our Registration Statement on Form S-1 (File No. 333-199761) filed on December 17,
2014). |
| 4.13 |
Form
of Warrant to Purchase Common Stock issued to Roth Capital Partners, LLC (incorporated by reference to Exhibit 4.13 included
in our Registration Statement on Form S-1 (File No. 333-199761) filed on December 17, 2014). |
| |
|
| 5.1
|
Opinion
of Duane Morris LLP. *** |
| |
|
| 10.1 |
Amended
and Restated 1995 Omnibus Incentive Plan (incorporated by reference to Exhibit 10.1 included in our Quarterly Report on Form
10-QSB, as amended, for the fiscal quarter ended September 30, 2003). ** |
| |
|
| 10.2 |
License
Agreement between the Company and the University of Texas Southwestern Medical Center (incorporated by reference to Exhibit
10.9 included in our Annual Report on Form 10-KSB filed March 30, 2004, as amended, for the fiscal year ended December 31,
2004). |
| |
|
| 10.3 |
License
Agreement between the Company and Thomas Jefferson University (incorporated by reference to Exhibit 10.9 included in our Annual
Report on Form 10-KSB, as amended, for the fiscal year ended December 31, 2004). |
| |
|
| 10.4 |
License
Agreement between the Company and the University of Texas Medical Branch (incorporated by reference to Exhibit 10.10 included
in our Annual Report on Form 10-KSB, as amended, for the fiscal year ended December 31, 2004). |
| |
|
| 10.5 |
Consulting
Agreement between the Company and Lance Simpson of Thomas Jefferson University. (incorporated by reference to Exhibit 10.43
included in our Annual Report on Form 10-KSB as amended for the fiscal year ended December 31, 2002). |
| |
|
| 10.6 |
2005
Equity Incentive Plan, as amended on September 25, 2013 (incorporated by reference to Exhibit 10.1 included in our current
report on Form 8-K filed on September 30, 2013). ** |
| |
|
| 10.7 |
Letter
of Intent dated January 3, 2007 by and between the Company and Sigma-Tau Pharmaceuticals, Inc. (incorporated by reference
to Exhibit 10.1 included in our current report on Form 8-K filed on January 4, 2007). |
| |
|
| 10.8 |
Letter
from Sigma-Tau Pharmaceuticals, Inc. dated February 21, 2007 (incorporated by reference to Exhibit 10.1 included in our current
report on Form 8-K filed on February 23, 2007). |
| |
|
| 10.9 |
Letter
dated May 3, 2007 between the Company and Sigma-Tau Pharmaceuticals, Inc. (incorporated by reference to Exhibit 10.1 included
in our current report on Form 8-K filed on May 4, 2007). |
| |
|
| 10.10 |
Employment
Agreement dated December 27, 2007, between Christopher J. Schaber, PhD and the Company (incorporated by reference to Exhibit
10.30 included in our Annual Report on Form 10-K for the fiscal year ended December 31, 2008). ** |
| |
|
| 10.11 |
Employment
Agreement dated December 27, 2007, between Evan Myrianthopoulos and the Company (incorporated by reference to Exhibit 10.31
included in our Annual Report on Form 10-K for the fiscal year ended December 31, 2008). ** |
| |
|
| 10.12 |
Common
Stock Purchase Agreement dated February 14, 2008, between the Company and Fusion Capital Fund II, LLC (incorporated by reference
to Exhibit 10.35 included in our Registration Statement on Form S-1 filed on February 14, 2008). |
| |
|
| 10.13 |
Registration
Rights Agreement dated February 14, 2008, between the Company and Fusion Capital Fund II, LLC (incorporated by reference to
Exhibit 10.35 included in our Registration Statement on Form S-1 (File No. 333-149239) filed on February 14, 2008). |
| 10.14 |
Letter
dated December 1, 2008, between the Company and Sigma-Tau Pharmaceuticals, Inc. (incorporated by reference to Exhibit 10.1
included in our current report on Form 8-K filed on December 1, 2008). |
| |
|
| 10.15 |
Exclusive
License Agreement dated November 24, 1998, between Enteron Pharmaceuticals, Inc. and George B. McDonald, MD and amendments
(incorporated by reference to Exhibit 10.42 included in our Registration Statement on Form S-1 (File No. 333-157322)
filed on February 13, 2009). |
| |
|
| 10.16
|
Collaboration
and Supply Agreement dated February 11, 2009, between the Company and Sigma-Tau Pharmaceuticals, Inc. (incorporated by reference
to Exhibit 10.43 included in our Registration Statement on Form S-1 (File No. 333-157322) filed on February 13, 2009).
† |
| |
|
| 10.17 |
First
Amendment to Common Stock Purchase Agreement dated April 19, 2010 between the Company and Fusion Capital Fund II, LLC (incorporated
by reference to Exhibit 10.34 included in our Post-Effective Amendment to Registration Statement on Form S-1 (File No. 333-149239)
filed on April 20, 2010). |
| |
|
| 10.18 |
Amendment
to Employment Agreement dated as of January 4, 2011, between the Company and Evan Myrianthopoulos (incorporated by reference
to Exhibit 10.1 included in our current report on Form 8-K filed on January 6, 2011). ** |
| |
|
| 10.19 |
Employment
Agreement dated as of January 31, 2011 between Kevin Horgan, M.D., and the Company (incorporated by reference to Exhibit 10.1
included in our current report on Form 8-K filed on February 2, 2011). ** |
| |
|
| 10.20 |
Employment
Agreement dated as of May 31, 2011, between Joseph M. Warusz and the Company (incorporated by reference to Exhibit 10.1 of
our current report on Form 8-K filed on May 31, 2011).** |
| |
|
| 10.21 |
First
Amendment to Employment Agreement dated as of July 12, 2011, between the Company and Christopher J. Schaber, PhD (incorporated
by reference to Exhibit 10.1 of our current report on Form 8-K filed on July 14, 2011).** |
| |
|
| 10.22 |
Second
Amendment to Employment Agreement dated as of July 12, 2011, between the Company and Evan Myrianthopoulos (incorporated by
reference to Exhibit 10.2 of our current report on Form 8-K filed on July 14, 2011).** |
| |
|
| 10.23 |
Amendment
to the Collaboration and Supply Agreement dated July 26, 2011, between Sigma-Tau Pharmaceuticals, Inc. and the Company (incorporated
by reference to Exhibit 10.1 of our current report on Form 8-K filed on July 28, 2011). |
| |
|
| 10.24 |
Amendment
to the Exclusive License Agreement dated as of July 26, 2011, between George McDonald, MD and the Company (incorporated by
reference to Exhibit 10.2 of our current report on Form 8-K filed on July 28, 2011). |
| |
|
| 10.25 |
Separation
Agreement dated February 15, 2012, between Evan Myrianthopoulos and the Company (incorporated by reference to Exhibit 10.28
included in our Registration Statement on Form S-1 (File No. 333-184762) filed on November 5, 2012). ** |
| |
|
| 10.26 |
First
Amendment to Separation Agreement dated July 2, 2012, between Evan Myrianthopoulos and the Company (incorporated by reference
to Exhibit 10.29 included in our Registration Statement on Form S-1 (File No. 333-184762) filed on November 5, 2012). ** |
| |
|
| 10.27 |
Amendment
No. 2 to the Collaboration and Supply Agreement between the Company, Enteron and Sigma-Tau dated as of December 20, 2012 (incorporated
by reference to Exhibit 10.1 of our current report on Form 8-K filed on December 27, 2012). † |
| 10.28 |
Amendment
to Exclusive License Agreement dated as of December 20, 2012 between Enteron and McDonald (incorporated by reference to Exhibit
10.4 of our current report on Form 8-K filed on December 27, 2012). |
| |
|
| 10.29 |
Amendment
to Consulting Agreement dated as of December 20, 2012 between Enteron and McDonald (incorporated by reference to Exhibit 10.5
of our current report on Form 8-K filed on December 27, 2012). |
| |
|
| 10.30
|
Exclusive
Channel Collaboration Agreement dated as of April 27, 2013 between the Company and Intrexon Corporation (incorporated by reference
to Exhibit 10.1 of our current report on Form 8-K filed on May 1, 2013). † |
| |
|
| 10.31 |
Stock
Issuance Agreement dated as of April 27, 2013 between the Company and Intrexon Corporation (incorporated by reference to Exhibit
10.2 of our current report on Form 8-K filed on May 1, 2013). † |
| |
|
| 10.32 |
Form
of Securities Purchase Agreement among the Company and investors in the June 2013 registered public offering (incorporated
by reference to Exhibit 10.2 included in our current report on Form 8-K filed on June 24, 2013). |
| 10.33
|
Contract
HHSO100201300023C dated September 18, 2013 between the Company and the U.S. Department of Health and Human Services Biomedical
Advanced Research and Development Authority (incorporated by reference to Exhibit 10.1 of our current report on Form 8-K filed
on September 24, 2013). † |
| |
|
| 10.34 |
Contract
HHSN272201300030C dated September 24, 2013 by and between the Company and the National Institutes of Health (incorporated
by reference to Exhibit 10.1 of our current report on Form 8-K filed on September 30, 2013). † |
| |
|
| 10.35 |
Purchase
Agreement dated as of November 18, 2013 between the Company and Lincoln Park Capital Fund, LLC (incorporated by reference
to Exhibit 10.1 of our current report on Form 8-K filed on November 21, 2013). |
| |
|
| 10.36 |
Registration
Rights Agreement dated as of November 18, 2013 between the Company and Lincoln Park Capital Fund, LLC (incorporated by reference
to Exhibit 10.2 of our current report on Form 8-K filed on November 21, 2013) |
| |
|
| 10.37 |
Employment
Agreement dated as of January 6, 2014 between the Company and Richard Straube, M.D. (incorporated by reference to Exhibit
10.1 of our current report on Form 8-K filed on January 8, 2014). ** |
| |
|
| 10.38 |
Asset
Purchase Agreement dated September 3, 2014 between the Company and Hy Biopharma, Inc. (incorporated by reference to Exhibit
10.1 of our current report on Form 8-K filed on September 5, 2014). † |
| |
|
| 10.30 |
Registration
Rights Agreement dated September 3, 2014 between the Company and Hy Biopharma, Inc. (incorporated by reference to Exhibit
10.2 of our current report on Form 8-K filed on September 5, 2014). |
| |
|
| 10.40 |
Contract
HHSN272201400039C dated September 17, 2014 by and between the Company and the National Institutes of Health (incorporated
by reference to Exhibit 10.1 of our current report on Form 8-K filed on September 23, 2014). † |
| |
|
| 10.41 |
Lease
Agreement dated November 21, 2014, between the Company and CPP II, LLC (incorporated by reference to Exhibit 10.31 included
in our Annual Report on Form 10-K for the fiscal year ended December 31, 2014). |
| 10.42 |
2015
Equity Incentive Plan, as amended on June 9, 2015 (incorporated by reference to Exhibit 10.1 of our current report on Form
8-K filed on June 19, 2015). |
| |
|
| 10.43 |
Form
of Equity Purchase Agreement dated as of July 29, 2015 between the Company and Kodiak Capital Group, LLC, Kingsbrook Opportunities
Master Fund LP and River North Equity, LLC (incorporated by reference to Exhibit 10.1 of our current report on Form 8-K filed
on July 31, 2015) |
| |
|
| 10.44 |
Form
of Registration Rights Agreement dated as of July 29, 2015 between the Company and Kodiak Capital Group, LLC, Kingsbrook Opportunities
Master Fund LP and River North Equity, LLC (incorporated by reference to Exhibit 10.2 of our current report on Form 8-K filed
on July 31, 2015) |
| |
|
| 10.45 |
Form
of Promissory Note dated as of July 29, 2015 made by the Company in favor of Kodiak Capital Group, LLC, Kingsbrook Opportunities
Master Fund LP and River North Equity, LLC (incorporated by reference to Exhibit 10.3 of our current report on Form 8-K filed
on July 31, 2015) |
| |
|
| 21.1
|
Subsidiaries
of the Company.* |
| |
|
| 23.1 |
Consent of EisnerAmper
LLP. * |
| |
|
| 23.2 |
Consent
of Duane Morris LLP (contained in the opinion filed as Exhibit 5.1 hereto). *** |
| * |
Filed
herewith. |
| ** |
Indicates
management contract or compensatory plan. |
| *** |
To
be filed by amendment. |
| † |
Portions
of this exhibit have been omitted pursuant to a request for confidential treatment. |
The
undersigned registrant hereby undertakes:
(1)
To file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement:
(i)
To include any prospectus required by Section 10(a)(3) of the Securities Act of 1933;
(ii)
To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent
post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set
forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if
the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high
end of the estimated maximum offering range may be reflected in the form of prospectus filed with the Commission pursuant to Rule
424(b) if, in the aggregate, the changes in volume and price represent no more than a 20 percent change in the maximum aggregate
offering price set forth in the “Calculation of Registration Fee” table in the effective registration statement; and
(iii)
To include any material information with respect to the plan of distribution not previously disclosed in the registration statement
or any material change to such information in the registration statement;
(2)
That, for the purpose of determining any liability under the Securities Act of 1933, each such post-effective amendment shall
be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at
that time shall be deemed to be the initial bona fide offering thereof.
(3)
To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold
at the termination of the offering.
(4)
That, for the purpose of determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial
distribution of the securities:
The
undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant to this registration
statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or
sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser
and will be considered to offer or sell such securities to such purchaser:
(i)
Any preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant
to Rule 424;
(ii)
Any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred
to by the undersigned registrant;
(iii)
The portion of any other free writing prospectus relating to the offering containing material information about the undersigned
registrant or its securities provided by or on behalf of the undersigned registrant; and
(iv)
Any other communication that is an offer in the offering made by the undersigned registrant to the purchaser.
Insofar
as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers and controlling
persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion
of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Securities Act and
is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by
the registrant of expenses incurred or paid by a director, officer or controlling person of the Registrant in the successful defense
of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities
being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent,
submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed
in the Securities Act and will be governed by the final adjudication of such issue.
SIGNATURES
Pursuant
to the requirements of the Securities Act of 1933, the registrant has duly caused this Registration Statement on Form S-1 to be
signed on its behalf by the undersigned, thereunto duly authorized, in the City of Princeton, State of New Jersey, on the 3rd
day of August, 2015.
| |
SOLIGENIX,
INC. |
| |
|
|
| |
By: |
/s/
Christopher J. Schaber |
| |
|
Christopher
J. Schaber, PhD |
| |
|
Chief Executive
Officer and President |
KNOW
ALL MEN BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints Christopher J. Schaber and
Joseph Warusz, and each of them, his or her true and lawful attorneys-in-fact and agents, with full power of substitution and
resubstitution, for him or her and in his or her name, place and stead in any and all capacities, to sign any or all amendments
to this Registration Statement on Form S-1 (including post-effective amendments), and to file the same, with all exhibits thereto,
and other documents in connection therewith with the United States Securities and Exchange Commission, granting unto said attorneys-in-fact
and agents full power and authority to do and perform each and every act and thing requisite and necessary to be done in and about
the premises, as fully and to all intents and purposes as he or she might or could do in person, hereby ratifying and confirming
that said attorneys-in-fact and agents, or their substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant
to the requirements of the Securities Act of 1933, this Registration Statement has been signed by the following persons in the
capacities and on the dates indicated.
| Signature
|
|
Title
|
|
Date
|
| |
|
|
|
|
By: |
/s/
Christopher J. Schaber |
|
|
|
|
|
Christopher
J. Schaber, PhD |
|
Chairman,
President and Chief Executive Officer |
|
August
3, 2015 |
| |
|
|
(Principal
Executive Officer) |
|
|
| |
|
|
|
|
|
| By:
|
/s/
Keith L. Brownlie |
|
|
|
|
| |
Keith
L. Brownlie, CPA |
|
Director
|
|
August
3, 2015 |
| |
|
|
|
|
|
| By:
|
/s/
Marco M. Brughera |
|
|
|
|
| |
Marco
M. Brughera, DVM |
|
Director
|
|
August
3, 2015 |
| |
|
|
|
|
|
| By:
|
/s/
Gregg A. Lapointe |
|
|
|
|
| |
Gregg
A. Lapointe, CPA |
|
Director
|
|
August
3, 2015 |
| |
|
|
|
|
|
|
By: |
/s/
Robert J. Rubin |
|
|
|
|
| |
Robert
J. Rubin, MD |
|
Director
|
|
August
3, 2015 |
| |
|
|
|
|
|
|
By: |
/s/
Jerome Zeldis |
|
|
|
|
| |
Jerome
Zeldis, MD, PhD |
|
Director
|
|
August
3, 2015 |
| |
|
|
|
|
|
By: |
/s/
Joseph M. Warusz |
|
|
|
|
| |
Joseph
M. Warusz, CPA |
|
Vice
President of Finance,
Acting Chief Financial Officer and
Corporate Secretary
(Principal Financial and Accounting Officer) |
|
August
3, 2015 |
II-11
EXHIBIT 21.1
SUBSIDIARIES OF SOLIGENIX, INC.
The following represents a list of Soligenix, Inc.’s subsidiaries:
| Name |
|
Ownership |
|
|
State of Incorporation |
|
| Enteron Pharmaceuticals, Inc. |
|
|
100.00 |
% |
|
|
Delaware |
|
| Orasomal Technologies Inc. |
|
|
75.30 |
% |
|
|
Delaware |
|
| DOR BioDefense Corp. |
|
|
100.00 |
% |
|
|
Delaware |
|
| Soligenix BioPharma Canada Incorporated |
|
|
100.00 |
% |
|
|
Canada |
|
| Soligenix UK Limited |
|
|
100.00 |
% |
|
|
United Kingdom |
|
EXHIBIT
23.1
CONSENT
OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
We
consent to the inclusion in this Registration Statement of Soligenix, Inc. on Form S-1 to be filed on or about August 3, 2015
of our report dated March 25, 2015, on our audits of the consolidated financial statements as of December 31, 2014 and 2013 and
for each of the years in the two-year period ended December 31, 2014. We
also consent to the reference to our firm under the caption “Experts” in the Registration Statement on Form S-1.
/s/ EisnerAmper LLP
Jenkintown, Pennsylvania
August 3, 2015
Soligenix (NASDAQ:SNGX)
Historical Stock Chart
From Sep 2024 to Oct 2024
Soligenix (NASDAQ:SNGX)
Historical Stock Chart
From Oct 2023 to Oct 2024