Seasoned Biotech Financial Executive, Jay M. Moyes, Joins Sera Prognostics
April 02 2020 - 8:00AM

Sera Prognostics, Inc., The Pregnancy Company™, focused on
improving maternal and neonatal health through innovative precision
biomarker approaches, today announced the appointment of Jay M.
Moyes as Chief Financial Officer. Mr. Moyes joins Sera Prognostics
as it transitions from rigorous scientific and groundbreaking
clinical stage company into a rapidly growing commercial
organization. For more than two decades, Jay Moyes has served in
C-level executive and board positions of both private and public
companies. He currently serves as Director on the public boards of
Achieve Life Sciences (NASDAQ: ACHV), BioCardia (NASDAQ: BCDA)
and Puma Biotechnologies (NASDAQ: PBYI). Previous roles include
board director service with Predictive Technology Group (PRED);
Osiris Therapeutics (OSIR); and Integrated Diagnostics (acquired by
Biodesix). Jay also served as Chief Financial Officer and Director
of the publicly traded biomaterials company, Amedica Corporation
(now SINTX Technologies), managing the company’s IPO and listing on
NASDAQ; and as Chief Financial Officer at XDx (now CareDx –
CDNA).
At Myriad Genetics (MYGN) , Jay led the finance function for 14
years from the company’s early private stage development beginning
with 7 employees, through its successful IPO in 1996, and serving
until 2007 as Chief Financial Officer during Myriad’s rapid growth
trajectory to become one of the world’s most highly profitable
molecular diagnostics companies, with over 900 employees.
During that time, Moyes spearheaded the efforts in raising more
than $500 million in public and private financings. “I am thrilled
to be working with Jay Moyes again, as we build Sera Prognostics
into a highly successful and profitable enterprise,” said Gregory
C. Critchfield, MD., MS, chairman and CEO. “Jay’s deep financing
connections and expertise in leading biopharmaceutical, medical
device and diagnostics companies, both in private and public
company settings, will help Sera to achieve its vision of improving
the health of mothers and babies and reducing the costs of
healthcare delivery.”
|
“I am excited to join the talented Sera team and help accelerate
the company’s progress, as Greg and I successfully did together at
Myriad.” -Jay MoyesClick to tweet |
“Sera has achieved remarkable progress in building a world-class
approach to address the immense burden of premature birth,” said
Jay Moyes. “The early data from outcome study reports show
that Sera’s PreTRM® strategy works, and that by improving the
health of women and babies, the enormous personal, societal and
economic consequences of prematurity can be mitigated. I am
excited to join the talented Sera team and help accelerate the
company’s progress, as Greg and I successfully did together at
Myriad.”
A photo accompanying this announcement is available
at: https://www.globenewswire.com/NewsRoom/AttachmentNg/b1b82cc8-0a89-413b-bf84-1354b59a87da
About Sera Prognostics, Inc. Sera Prognostics is
the leading health diagnostics company dedicated to improving the
lives of women and babies through precision pregnancy care. Sera
delivers pivotal information in early pregnancy to physicians,
enabling them to improve health of their patients, resulting in
reductions in the costs healthcare delivery. Sera has a
robust pipeline of innovative diagnostic tests focused on the early
prediction of preterm birth risk and other complications of
pregnancy. Sera’s precision medicine PreTRM® test reports to a
physician the individualized risk of premature delivery in a
pregnancy, enabling earlier proactive interventions in women with
higher risk. Sera Prognostics is located in Salt Lake City, Utah.
For more information, please visit the company’s website at
www.seraprognostics.com.
About Preterm Birth Preterm birth is defined as
any birth before 37 weeks gestation and is the leading cause of
illness and death in newborns. The 2019 March of Dimes Report
Card shows that of nearly 4 million babies born annually in the
U.S., more than one in ten is born prematurely.1 Prematurity is
associated with a significantly increased risk of major long-term
medical complications, including learning disabilities, cerebral
palsy, chronic respiratory illness, intellectual disability,
seizures, and vision and hearing loss, and can generate significant
costs throughout the lives of affected children. The annual US
health care costs to manage complications of prematurity were
estimated at $31.5B for 2015.2
About the PreTRM® Test The PreTRM® test is the
only clinically validated commercially available blood test that
provides an early individual risk prediction for spontaneous
preterm birth in asymptomatic, singleton pregnancies. The PreTRM®
test measures and analyzes proteins in the blood that are highly
predictive of preterm birth. The PreTRM® test permits physicians to
identify, as early as 19 weeks of pregnancy, which women are at
increased risk for premature delivery, enabling more informed
clinical decisions based on each woman’s individual risk, so that
her care can be personalized to address her risk. The PreTRM® test
is ordered by a medical professional. For more information about
the PreTRM® test, please visit www.PreTRM.com and the
PreTRM® test YouTube Channel. You can also join the conversation on
Facebook and @PreTRM.
About Sera’s Science Using its advanced mass
spectrometry platform, Sera detects biologically important protein
expression changes to build high performing predictions of risk for
adverse pregnancy outcomes (including preterm birth, preeclampsia,
gestational diabetes, growth restriction, and others).
Rigorous clinical validation of PreTRM® test performance (accuracy
of predicting premature delivery) was reported in the American
Journal of Obstetrics & Gynecology in 2016 in a US cohort of
5,501 patients across 11 centers. Subsequent scientific work
has confirmed performance of Sera’s biomarker predictions in
cohorts of patients across the globe: US, Europe, Asia and Africa,
with over 10,500 patients tested in studies.
About Clinical Outcomes DataSera’s validated
PreTRM® prediction has been taken into prospective intervention
studies, where populations of pregnant women have been screened by
the Sera test. Higher PreTRM®-risk pregnancies are stratified
to receive earlier more proactive interventions, and important
outcomes are examined in comparison to pregnancies in control
groups where the test is not available.
Topline results showing the benefit of the PreTRM® strategy in a
recently completed randomized controlled prospective intervention
study (PREVENT PTB Study, NCT03530332) became available online for
the Late Breaking Poster Session of the Annual Meeting of the
Society of Reproductive Investigation, scheduled for March 14 in
2020 in Vancouver, Canada.
1
http://www.marchofdimes.org/mission/prematurity-reportcard.aspx 2
Caughey et al., Am J Perinatol Rep 2016;6:e407-e416
Contacts:Fern Lazar, Lazar FINN
fern.lazar@finnpartners.com 212 867
1765 Chantal
Beaudry, Lazar FINN chantal.beaudry@finnpartners.com 646 871
8480
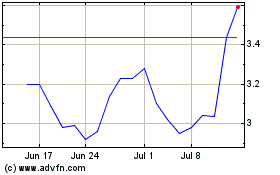
Puma Biotechnology (NASDAQ:PBYI)
Historical Stock Chart
From Aug 2024 to Sep 2024
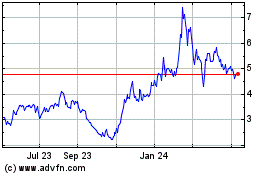
Puma Biotechnology (NASDAQ:PBYI)
Historical Stock Chart
From Sep 2023 to Sep 2024