Kala Pharmaceuticals to Present Phase 1b Clinical Data for KPI-012 in Patients with PCED at 2022 ARVO Annual Meeting
April 04 2022 - 4:01PM

Kala Pharmaceuticals, Inc. (NASDAQ:KALA), a commercial-stage
biopharmaceutical company focused on the discovery, development and
commercialization of innovative therapies for diseases of the eye,
today announced that it will present data from a previously
completed Phase 1b clinical trial of KPI-012 in a poster session at
the 2022 Association for Research in Vision and Ophthalmology
(ARVO) Annual Meeting, taking place in Denver, Colorado, May 1-4,
2022 and virtually, May 11-12, 2022. The data support the ongoing
development of KPI-012, a novel investigational secretome therapy,
to address the complex wound healing process in persistent corneal
epithelial defect (PCED).
The accepted abstract is listed below and is now
available on the ARVO conference website:
https://www.arvo.org/annual-meeting/.
Title: Results of a Phase 1b
clinical trial of KPI-012, a novel secretome therapy, in patients
with Persistent Corneal Epithelial Defect (PCED)Session
Title: Corneal epithelial biology and repair
mechanismsPresentation Number - Posterboard
Number: 3232 - A0267Date: Tuesday, May 3,
2022Time: 3:30 – 5:30 p.m. MDT (5:30 – 7:30 p.m.
EDT)Presenter: Valeria Sáanchez-Huerta, M.D.,
Director General, Asociación para Evitar la Ceguera en México
About Kala Pharmaceuticals,
Inc.
Kala is a commercial-stage biopharmaceutical
company focused on the discovery, development, and
commercialization of innovative therapies for diseases of the eye.
Kala has applied its AMPPLIFY® mucus-penetrating particle (MPP)
Drug Delivery Technology to two ocular therapies, EYSUVIS®
(loteprednol etabonate ophthalmic suspension) 0.25% and INVELTYS®
(loteprednol etabonate ophthalmic suspension) 1%. Kala also has a
pipeline of development programs including a clinical-stage
secretome product candidate, KPI-012, initially targeting
persistent corneal epithelial defects (PCED) and multiple
proprietary new chemical entity (NCE) preclinical development
programs targeted to address unmet medical needs, including both
front and back of the eye diseases. For more information on Kala,
please visit www.kalarx.com.
Forward Looking Statements:
This press release contains forward-looking
statements within the meaning of the Private Securities Litigation
Reform Act of 1995 that involve substantial risks and
uncertainties. Any statements in this press release about Kala’s
future expectations, plans and prospects, including but not limited
to statements about Kala’s expectations with respect to KPI-012,
the future development or commercialization of KPI-012, conduct and
timelines of clinical trials, Kala’s plans to progress its pipeline
of preclinical development programs targeted to address front and
back of the eye diseases, constitute forward-looking statements.
Actual results may differ materially from those indicated by such
forward-looking statements as a result of various important
factors, including those discussed in the “Risk Factors” section of
Kala’s Annual Report on Form 10-K, most recently filed Quarterly
Report on Form 10-Q and other filings Kala makes with the
Securities and Exchange Commission. These forward-looking
statements represent the Company’s views as of the date of this
release and should not be relied upon as representing the Kala’s
views as of any date subsequent to the date hereof. Kala does not
assume any obligation to update any forward-looking statements,
whether as a result of new information, future events or otherwise,
except as required by law.
Investor Contacts:
Jill Steierjill.steier@kalarx.com 781-996-5252
Hannah Deresiewiczhannah.deresiewicz@sternir.com
212-362-1200
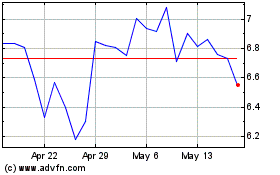
KALA BIO (NASDAQ:KALA)
Historical Stock Chart
From Jun 2024 to Jul 2024
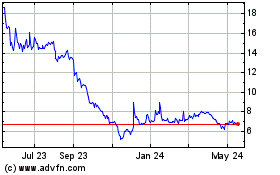
KALA BIO (NASDAQ:KALA)
Historical Stock Chart
From Jul 2023 to Jul 2024