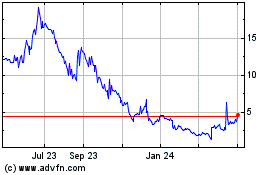
We are offering 4,700,000 shares of our common stock,
par value $0.001 per share (the “Common Stock”) and common warrants to purchase up to 3,525,000 shares of Common Stock and
the shares of Common Stock that are issuable from time to time upon exercise of the common warrants, at a purchase price of $1.70 per
share of Common Stock and 0.75 of an accompanying common warrant, pursuant to this prospectus supplement and the accompanying prospectus.
The shares of Common Stock and the accompanying common warrants can only be purchased together in this offering but will be issued separately
and will be immediately separable upon issuance. Each common warrant will have an exercise price of $1.90 per share of Common Stock, will
be exercisable upon issuance, and will expire five years from the date of issuance.
The Common Stock is listed on the Nasdaq Capital Market
under the symbol “KAVL.” The last reported sale price of the Common Stock on September 29, 2021 was $1.91 per share. There
is no established public trading market for the common warrants and we do not expect a market to develop. Without an active trading market,
the liquidity of the common warrants will be limited. In addition, we do not intend to list the common warrants on the Nasdaq Capital
Market, any other national securities exchange or any other trading system.
You should read this prospectus supplement and the
accompanying prospectus and the documents incorporated by reference in this prospectus supplement carefully before you invest.
We are an “emerging growth company”
and a “smaller reporting company” as such terms are defined in the Securities Act of 1933, as amended (the “Securities
Act”), and as such, are subject to certain reduced public company reporting requirement.
We have retained Maxim Group LLC to act as
underwriter in connection with this offering. We have agreed to pay the underwriter the fees set forth in the table below.
We have granted the underwriter a 45-day option to purchase up to an additional
705,000 shares of Common Stock at a purchase price of $1.5717 per share and/or up to an additional 528,750 common warrants
at a purchase price of $0.01239969 per common warrant from us to cover over-allotments, if any. If the underwriter exercises this option
in full, the total underwriting discounts and commissions will be $643,195, and our total proceeds, before expenses, will be $8,545,305.
We expect that delivery of the shares of the Common Stock and the
common warrants being offered pursuant to this prospectus supplement and the accompanying prospectus will be made to purchasers
through the facilities of The Depository Trust Company on or about October 4, 2021.
The date of this prospectus
supplement is September 29, 2021.
STATEMENT REGARDING FORWARD-LOOKING INFORMATION
This prospectus supplement, the accompanying
prospectus and the documents incorporated by reference herein contain forward-looking statements. All statements, other than statements
of historical facts contained in this prospectus supplement, the accompanying prospectus and in the documents incorporated by reference
herein, including statements regarding our efforts to develop and commercialize our Products, anticipated product performance,
our short-term and long-term business strategies, market and industry expectations and future results of operations and financial
position are forward-looking statements. In many cases, you can identify forward-looking statements by terms such as “may”,
“will”, “should”, “expect”, “plan”, “anticipate”, “could”,
“intend”, “target”, “project”, “contemplate”, “believe”, “estimate”,
“potential”, “continue” or other similar words.
We based these forward-looking statements largely
on our current expectations and projections about future events or trends that we believe may affect our business and financial
performance. These forward-looking statements involve known and unknown risks and uncertainties that may cause our actual results,
performance or achievements to materially differ from any future results, performance or achievements expressed or implied by these
forward-looking statements. We have described in the “Risk Factors” section and elsewhere in this prospectus supplement
and in the documents incorporated by reference herein the material risks and uncertainties that we believe could cause actual results
to differ from these forward-looking statements. Because forward-looking statements are inherently subject to risks and uncertainties,
some of which we cannot predict or quantify, you should not rely on these forward-looking statements as guarantees of future results,
performance or achievements.
The forward-looking statements in this prospectus
supplement, the accompanying prospectus and in the documents incorporated by reference herein represent our views as of the date
of the document in which the forward-looking statement appears. We undertake no obligation to update publicly, except to the extent
required by law, any forward-looking statements for any reason to conform these statements to actual results or to changes in
our expectations.
PROSPECTUS SUPPLEMENT SUMMARY
This summary highlights some information
from this prospectus supplement. It may not contain all the information important to making an investment decision. You should
read the following summary together with the more detailed information regarding our Company and the securities being sold in this
offering, including “Risk Factors” and other information incorporated by reference herein.
OUR BUSINESS
We are focused on growing and incubating innovative
and profitable products into mature, category-dominant brands. In March 2020, we commenced business operations after becoming the
exclusive distributor of certain electronic nicotine delivery systems (“ENDS”) and related components (the “Products”),
including the BIDI® Stick, manufactured by Bidi Vapor, LLC (“Bidi”), a Florida limited liability company, and a
related party company that is also owned by Nirajkumar Patel, our Founder and Chief Executive Officer.
Recent Developments
On September 14, 2021, we announced that, due
in our opinion to increased pressure from, among others, tobacco control and public health groups, members of Congress, academic
institutions, family advocacy organizations and attorneys general, the U.S. Food and Drug Administration (“FDA”) took
steps in connection with its ongoing Premarket Tobacco Application (“PMTA”) process relating to ENDS products to effectively
“ban” flavored ENDS by denying nearly all pending PMTAs for such products in connection with the September 9, 2021
PMTA review deadline. As of September 23, 2021, the FDA announced that it has taken action on over 93% of applications and issued
Marketing Denial Orders (“MDOs”) for more than 1,167,000 flavored ENDS products, while issuing zero marketing authorizations.
Unfortunately, despite submitting a comprehensive PMTA and continuing to develop robust and reliable product-specific scientific
evidence demonstrating the public health benefit of its flavored ENDS products, Bidi, along with nearly every other company in
the ENDS industry, was among the many companies that received an MDO for its non-tobacco flavored BIDI® Sticks, including its
Arctic (menthol) BIDI® Stick, which we believe the FDA mischaracterized as “flavored.” However, because its Arctic
BIDI® Stick has a menthol characterizing flavor, Bidi believes that this menthol BIDI® Stick was inadvertently included
on the MDO. This position is aligned with the FDA’s public statements and press releases stating that tobacco and menthol
ENDS are not deemed flavored products subject to the MDOs. Accordingly, along with the Classic (tobacco) BIDI® Stick, Bidi
intends to continue to manufacture and market its Arctic (menthol) BIDI® Stick for distribution by us.
Historically, substantially all of our revenues
were derived from sales of flavored BIDI® Sticks, including the Arctic (menthol) BIDI® Stick, sales of which constituted
approximately 15.2% and 18.5%, respectively, of our total sales of BIDI® Sticks for the three and nine months ended July 31,
2021. Generally, substantially all of the ENDS industry’s revenue is derived from the sales of flavored products.
Following the issuance of the MDOs, nearly
all manufacturers, not only Bidi, will be limited to manufacturing and selling only tobacco and menthol ENDS products. Accordingly,
we believe that consumers may modify their purchases to shift to products that are available, and thus that a substantial amount
of our revenue that had previously been generated from sales of flavored BIDI® Sticks (other than Arctic) may be replaced through
sales of the Classic (tobacco) and Arctic (menthol) BIDI® Sticks, given that consumers will only be able to purchase, on a
legal basis, non-flavored ENDS products. Accordingly, we believe revenues for tobacco and menthol ENDS products will restore some
of the revenue we derived from the sale of flavored products. Moreover, based on sales and consumer data, we believe that our consumers
are loyal to the Bidi brand and BIDI® Sticks and that they thus are likely to continue to purchase our Classic (tobacco) and
Arctic (menthol) BIDI® Sticks in lieu of the flavored BIDI® Sticks they may have bought in the past.
If the FDA disagrees with Bidi’s position,
issues a warning letter, or takes other action against Bidi resulting in us not being able to distribute the menthol (Arctic) BIDI®
Stick in the United States, or consumers do not purchase the tobacco (Classic) or menthol (Arctic) BIDI® Sticks, our revenues
and, thereby our financial results and condition, would be materially adversely affected. Our financial results and condition will
also be significantly impacted by our ability to continue to sell the Arctic (menthol) BIDI® Stick and the degree to which
sales of the Classic (tobacco) and Arctic (menthol) BIDI® Sticks replace sales of flavored products that are now prohibited.
Despite this setback, Bidi informed us that
it is not wavering in its commitment to demonstrating that all its BIDI® Stick products are appropriate for the protection
of the public health and proving to the FDA that these products should remain on the market as an alternative for adult cigarette
smokers. In this regard, Bidi is reviewing its legal options with respect to appealing and/or challenging the MDO and plans on
continuing to complete multiple ongoing studies, including a clinical pharmacokinetic, or PK, study, and several actual use and
perception and intention studies to support its PMTA. Preliminary results from these studies indicate that, compared to tobacco-flavored
ENDS, the flavored BIDI® Sticks do indeed provide an added benefit for adult cigarette smokers while outweighing any risks
to youth posed by flavored ENDS, particularly when considering Bidi’s stringent youth access prevention measures. We cannot
provide any assurances as to whether Bidi will be successful in its appeal of the MDO and, ultimately, the impact on our business,
which at this time is solely dependent on sales from the distribution of the BIDI® Sticks.
As has become clear, the PMTA decision process
will result in the elimination of the vast majority of all ENDS products from the marketplace, as these applications did not include
the product specific scientific evidence needed to justify marketing authorization, which Bidi is developing.
The BIDI® Stick has received marketing
and distribution approval in 11 European markets, including the United Kingdom. Bidi will now expedite the planned product launches
into foreign markets starting with the United Kingdom. We anticipate announcing soon a significant hire who will run and manage
the European business on the ground.
Overview
On March 9, 2020, we entered into an exclusive
distribution agreement with Bidi, a related party company, which Distribution Agreement was amended and restated on May 21, 2020
and again on April 20, 2021 (collectively, the “A&R Distribution Agreement”). Pursuant to the A&R Distribution
Agreement, Bidi granted to us an exclusive worldwide right to distribute the Products for sale and resale to both retail level
customers and non-retail level customers. The newly amended and restated distribution agreement extends the previous one-year,
annually renewable term to an initial term of ten years, which automatically renews for another five-year term; provided, that
we satisfy certain minimum purchase thresholds. The A&R Distribution Agreement also provides us with a right of first refusal
in the event Bidi receives an offer that would constitute a “change of control transaction,” as well as a right of
first refusal to act as the exclusive distributor of any and all future products of Bidi that arise out of or related to ENDS and
components related to ENDS, arise out of or relate to the synthetic nicotine industry, or arise out of or related to the tobacco-derived
nicotine industry.
Current Product Offerings
Pursuant to the A&R Distribution Agreement,
we sell and resell ENDS Products (also referred to as “e-cigarettes”) to both retail level customers and non-retail
level customers. The primary Product we resell is the “BIDI® Stick,” a disposable, tamper-resistant ENDS that comes
in a variety of flavor options; however, following the receipt of Bidi’s MDO from the FDA, we will only distribute the Classic
(tobacco) and Arctic (menthol) BIDI® Sticks in the United States, and all of our flavor options in the 11 European markets
we have received marketing and distribution approvals. In addition to the BIDI® Stick, we are now planning to initiate distribution
of the “BIDI® Pouch,” initially outside the United States. The initial planned February 2021 United States roll
out of BIDI® Pouch was delayed due to COVID-19 related manufacturing and supply chain constraints. Due to these complications
and in effort to prevent future bottlenecks, Bidi moved manufacturing in-house.
Recently, Bidi modified the planned formulation
of the BIDI® Pouch. The original BIDI® Pouch formulation intended to utilize a tobacco-free nicotine (synthetic) nicotine
formulation, along with natural fibers and a chew-base filler in six different flavors. However, in continuation of its long-standing
history of stressing corporate responsibility and regulatory compliance, Bidi has encouraged a potential FDA ban on the marketing
and distribution of synthetic nicotine in the United States, particularly in disposable ENDS, taking the position that synthetic
nicotine should be classified as an unapproved drug and, thus, subject to the applicable FDA drug regulations. Conversely, we believe
that many other ENDS manufacturers are utilizing synthetic nicotine as a loophole to avoid the rigorous PMTA process and that if
synthetic nicotine is deemed to be an unapproved drug, the FDA will need to regulate synthetic nicotine products as unapproved
drugs, or remove them from the market, in order to enforce and bolster compliance requirements.
Due to its position on synthetic nicotine,
which Bidi has communicated to the FDA, Bidi has formally communicated to us that the planned United States launch of the BIDI®
Pouch, which was initially delayed because of COVID-19, will now be further delayed as its tobacco-derived nicotine formula will
require PMTA authorization before it can be distributed in the United States.
While the BIDI® Stick and BIDI® Pouch
complete the PMTA process to obtain formal marketing authorization in the United States, the tobacco-derived nicotine formula for
both products is approved for distribution in several global markets, including the United Kingdom and Russia.
We do not manufacture any of the Products we
resell. The BIDI® Stick and BIDI® Pouch are manufactured by Bidi. Pursuant to the terms of the A&R Distribution Agreement,
Bidi provides us with all branding, logos, and marketing materials to be utilized by us in connection with our marketing and promotion
of our Products.
We process all sales made to retail customers
and non-retail customers, with all sales to retail customers to be made through Bidi’s age-restricted website, www.bidivapor.com.
We provide all customer service and support at our own expense. Bidi sets the minimum prices for all sales made by us. With respect
to sales to non-retail customers, we submit purchase orders to Bidi, Bidi delivers the Products to us, and we ship the Products
directly to these non-retail customers. In the case of retail customers, we maintain adequate inventory levels of the Products
in order to meet these customers’ demand, and deliver the Products sold to these retail customers.
In addition to our current product offerings,
Bidi launched its recycling program, BIDI® Cares, in the spring of 2020 that provides an opportunity for its adult (21+) customers
to recycle their BIDI® Sticks and be rewarded with a free BIDI® Stick after recycling ten used BIDI® Sticks. Each BIDI®
Stick contains UL 8139 Certified batteries, which are high-quality, recyclable batteries that are distinguishable from batteries
used in other ENDS Products. Bidi invests in recyclable batteries as a more sustainable solution to reduce electronic waste.
On July 14, 2021, we announced plans to launch
our first Kaival-branded product, a hemp CBD product. In addition to our Kaival-branded formulation, we will also be providing
white label, wholesale solutions for other product manufacturers through our subsidiary, Kaival Labs.
All CBD products will be produced and distributed
strictly in compliance under the 2018 Farm Bill, which defines hemp as the plant Cannabis sativa and any part of the plant with a delta-9
THC concentration of not more than 0.3 percent by dry weight. According to the 2018 Farm Bill, hemp-derived products can be offered for
retail sale in the many forms: smoke, pouch, tinctures, topicals, capsules, vape oil and gummies/edibles. We plan to utilize Bidi’s
best-in-class, patented BIDI® Stick delivery mechanism in order to provide a similar, premium experience in the initial CBD product
line. We expect our industrial-grade hemp CBD formula to provide greater bioavailability than many market peers, resulting in a better
consumer experience in less usage. Our use of the patented BIDI Stick delivery technology is subject to our ability to enter into a technology
license with BIDI to use their technology in our planned future CBD product.
Marketing Strategy
Currently, we market and place our Products
into national distribution channels through long-standing industry relationships in accordance with the A&R Distribution Agreement.
We process all sales made to retail customers and non-retail customers, with all sales to retail customers made through the Bidi
website.
Our long-term marketing strategy remains based
on FDA compliance and our commitment to preventing underage access to our Products. As such, we steer away from social media marketing,
and, instead, are more focused on ground-level marketing and advertising within authorized retailer locations (i.e., advertisement
on retail partners’ back-bar tobacco products area). Part of this ground-level marketing effort focuses on supporting our
authorized partner stores and distributors in spreading brand awareness of our legally marketed Products (i.e., those not subject
to MDO) to their adult (21+) consumer base by providing in-store marketing materials. Additionally, we supplement these efforts
with informational videos on Bidi’s fight against underage access to ENDS products and content on what makes the BIDI®
Stick unique, among other types of content.
Retail stores also have access to online informative
videos about the Bidi story, which can be used to educate and assist in training all staff members about the core values of Bidi.
From the recycling initiative to the commitment to preventing underage ENDS use, and stand against the illicit market of ENDS products,
we believe that together with Bidi, we are taking the necessary steps to ensure that our partners are aligned with our community
goals.
Bidi also attends trade shows at established
expos throughout the United States, such as the National Association for Convenience Stores (NACS) Show. As announced on July 8,
2021, Bidi resumed its attendance at live trade shows following an increase in COVID-19 vaccination rates and other pandemic-created
protocols. Further, Bidi has a dedicated marketing team that focuses on these marketing efforts and more.
Resellers
Building upon our significant increases in
distribution during fiscal 2020, which resulted in potential distribution reach to over 10,000 stores, including Lakshmi Distributer
Inc., doing business as C Store Master (“C Store Master”), which currently distributes to Circle K convenience stores,
as well as 7 ELEVEN locations across the country, our Products are also accessible through the digital delivery service mobile
applicable, GoPuff, in more than 50 cities across the United States.
During fiscal 2021, we have added an additional
potential reach of 44,000 stores, including, but not limited to:
|
|
●
|
Grocery Supply Warehouse (“GSW”), a 15-year-old company
with more than 26,000 stores, based in Missoula, Montana that started out distributing coffee and other beverage products to the
c-store channel;
|
|
|
●
|
Smoker Friendly International, an industry-leading tobacco-shop chain;
|
|
|
●
|
Avail Vapor LLC, a 12-state vape-store network; and
|
|
|
●
|
Hilmes Distributing, a large Midwest wholesaler.
|
Since our launch in March 2020, we have established
a distribution network with a potential reach of over 54,000 stores.
Environment and Government Regulation Related
to our Operations
Because we are only a retailer and wholesale
distributor of the BIDI® Stick, we believe that we are only subject to Federal, state, and international laws pertaining to
a distributor, not a manufacturer, of ENDS Products.
Our business is dependent entirely on the resale
of the Products provided by Bidi; thus, there is a significant risk that our business could be materially adversely affected if
Bidi, as the manufacturer, does not properly abide by any Federal, state, or international laws that regulate ENDS Products. Any
lapse in production or availability of the Products from Bidi would hamper our ability to operate as we would be limited in our
ability to supply our customers if our inventory ran low or ceased to exist entirely.
As a manufacturer of ENDS Products, Bidi is
responsible for abiding by and following various rules and regulations pertaining to the manufacturing of the ENDS Products we
sell and any lapse in abiding by any pertinent rules and regulations may negatively impact our ability to operate. As a distributor,
we are also subject to various rules and regulations. Some of the below may not directly apply to us at this time due to the nature
of our present operations. These rules and regulations include, but are not limited to, the following:
FDA and Related Regulations Relating to
ENDS Products
Effective August 8, 2016, the FDA’s regulatory
authority under The Family Smoking Prevention and Tobacco Control Act (the “Tobacco Control Act”) was extended to all
remaining tobacco products, including: (i) certain “new generation” products (such as electronic cigarettes, vaporizers
and e-liquids) and their components or parts (such as tanks, coils and batteries); (ii) cigars and their components or parts (such
as cigar tobacco); (iii) pipe tobacco; (iv) hookah products; or (v) any other tobacco product “newly deemed” by the
FDA. This “Deeming Regulation” applies to all products made or derived from tobacco intended for human consumption,
but excluding accessories of tobacco products (such as lighters).
The Deeming Regulation requires (i) U.S. manufactured
products be registered with the FDA and that products submit ingredient listings to FDA; (ii) deemed new tobacco products, including
all ENDS, be marketed only after FDA review and authorization, subject to FDA’s compliance enforcement policy; (iii) products
only make direct and implied claims of reduced risk if the FDA authorizes after finding that scientific evidence supports the claim
and that marketing the product will benefit public health as a whole; (iv) sellers of such products refrain from distributing free
samples; (v) sellers of such products implement minimum age and identification restrictions to prevent sales to individuals under
age 21; (vi) packaging of and advertisements for products include prescribed nicotine addiction warnings; and (vii) sellers refrain
from selling the products in vending machines, unless the machine is located in a facility that never admits youth under 21. We,
along with Bidi, must comply with these regulations. Any lapse in compliance by us, or Bidi, could hamper our ability to operate,
which would adversely affect our results of operations.
Newly deemed tobacco products are also subject
to the other requirements of the Tobacco Control Act, such as the products cannot be adulterated or misbranded. The FDA could in
the future promulgate good manufacturing practice regulations for these and our other Products, which could have a material adverse
impact on Bidi’s ability to, and the cost to, manufacture our Products, which would adversely affect our financial condition
and results of operations.
Failure to comply with the Tobacco Control
Act and or with any FDA regulatory requirements could result in litigation, criminal convictions or significant financial penalties
and could impair our ability to market and sell our ENDS products. Unfortunately, despite submitting a comprehensive PMTA and continuing
to develop robust and reliable product-specific scientific evidence demonstrating the public health benefit of its flavored ENDS
products, Bidi, along with nearly every other company in the ENDS industry, was among the many companies that received an MDO for
its non-tobacco flavored BIDI® Sticks, including its Arctic (menthol) BIDI® Stick, which we believe the FDA mischaracterized
as “flavored.” However, because its Arctic BIDI® Stick has a menthol characterizing flavor, Bidi Vapor believes
that this menthol BIDI® Stick was inadvertently included on the MDO. This position is aligned with the FDA’s public statements
and press releases stating that tobacco and menthol ENDS are not deemed flavored products subject to the MDOs. Accordingly, along
with the Classic (tobacco) BIDI® Stick, Bidi intends to continue to manufacture and market its Arctic (menthol) BIDI® Stick
for distribution by us.
As part of the “Consolidated Appropriations
Act, 2021,” signed into law on December 27, 2020, Congress amended the PACT Act to apply to ENDS, which includes the
BIDI® Stick. The PACT Act regulates the sale, transfer, or shipment of cigarettes, roll-your-own tobacco,
smokeless tobacco, and now ENDS, for both business-to-business transactions as well as online sales. The PACT Act imposes
substantial restrictions on sellers and shippers of ENDS products, including, but not limited to: registration with the ATF; registration
with state Tobacco Tax Administrators, and monthly reporting requirements to state and local Tobacco Tax Administrators. Delivery
sellers are subject to substantial additional restrictions, including, but not limited to, compliance with state excise tax collection
requirements, licensing requirements, shipping, and packaging requirements. Companies were required to comply with PACT Act
requirements beginning on or about March 28, 2021.
We have adopted the following compliance measures:
|
|
●
|
We have retained a team of legal, tax and accounting experts to advise
on state and local tax, licensing, and regulatory matters associated with the distribution of the BIDI® Stick;
|
|
|
|
|
|
|
●
|
We are appropriately licensed or registered in every state which requires
it;
|
|
|
|
|
|
|
●
|
We calculate and remit excise taxes where required;
|
|
|
|
|
|
|
●
|
We have made a substantial investment in excise tax reporting and
compliance software to ensure that all applicable taxes are properly calculated and remitted to the appropriate taxing authorities.
The software is now completely integrated with our systems;
|
|
|
|
|
|
|
●
|
We have registered with the ATF and the states into which we ship the
Products; and
|
|
|
|
|
|
|
●
|
We have implemented processes to ensure timely filing of all required
reporting.
|
We are committed to complying with all federal
and state laws associated with the distribution of ENDS products and ensuring that the legally marketable BIDI® Stick products
(e.g., the Classic and Arctic BIDI® Stick, which we do not believe is subject to the MDO) are marketed responsibly only to
adult consumers 21 and over. We are committed to these principles notwithstanding the current environment pursuant to which
many companies continue to distribute non-compliant ENDS products, including products that were not on the market on August
8, 2016 and for which no PMTA has been submitted with the FDA, as well as products that continue to be marketed and sold online
without collection of excise taxes.
Intellectual Property
Currently, as of the date of this prospectus
supplement, we have no intellectual property rights other than the trademarks KAIVAL BRANDS and KAIVAL LABS. We rely on certain
intellectual property rights, including logos, trademarks, and trade names, of Bidi that were granted to us pursuant to the A&R
Distribution Agreement to be used in connection with the marketing, advertisement, and sale of the Products. We also indirectly
rely on Bidi’s intellectual property rights related to the Products, such as patents. If a third-party challenged Bidi’s
patents, or infringed upon such rights, our business would be materially adversely affected.
We continue to seek, develop and evaluate new
technologies and related intellectual property that might enhance our business strategy, industry position or deployment options.
Risks Affecting Us
Our business is subject to a number of risks
and uncertainties that you should understand before making an investment decision, which are discussed more fully in the section
below entitled “Risk Factors.” These risks, any of which could have a material and adverse effect on our business,
financial condition and results of operations, include, but are not limited to, the following:
|
|
●
|
We have a limited operating history, and our historical results may
not be indicative of future performance, which makes it difficult to predict our future business prospects and financial performance.
|
|
|
|
|
|
|
●
|
The FDA has recently issued Bidi an MDO, which prevents us from marketing
Products making up a substantial majority of our revenue. If Bidi is unsuccessful in its appeal of the MDO, if our conclusion as
to our ability to sell our Arctic (menthol) BIDI® Stick is challenged or if our expectations as to how the purchasing habits
of consumers may change to offset the loss of revenue with respect to certain of our Products, our revenues, financial results
and condition would be materially adversely affected.
|
|
|
|
|
|
|
●
|
If Bidi’s planned PMTA for the tobacco-derived
nicotine formulation of the BIDI® Pouch is not authorized by the FDA, we will not be able
to sell the BIDI® Pouch in the United States.
|
|
|
|
|
|
|
●
|
If it is determined or perceived that the usage of ENDS products
poses health risks, or that ENDS products could lead to youth use of tobacco products, the use of ENDS products may decline significantly.
|
|
|
|
|
|
|
●
|
We rely exclusively
on Bidi as the supplier of the Products that we distribute. Our relationship with Bidi is subject to change, and the loss of this
relationship, or any negative impacts on Bidi’s ability to product the Products, would severely harm our business.
|
|
|
|
|
|
|
●
|
The terms of our agreements with affiliated entities including our
A&R Distribution Agreement with Bidi may not always be as favorable to us as the terms that may be obtained by arms’
length negotiation.
|
|
|
|
|
|
|
●
|
We are subject to supply shortages and interruptions, long lead times
and act-of-God events such as global pandemics, weather related catastrophes, or conflict, any of which could disrupt our supply
chain. We may be unable to identify or contract with new suppliers or producers in the event of a disruption to our supply.
|
|
|
●
|
We may not be successful in maintaining the consumer brand recognition
and loyalty of our Products and face intense competition and may fail to compete effectively.
|
|
|
|
|
|
|
●
|
Our distribution efforts rely in part on our ability to leverage
relationships with large retailers and national chains. In addition, certain large carriers, including Federal Express, UPS and
DHL, no longer carry ENDS products, and we may face additional challenges to the physical distribution of our Products.
|
|
|
|
|
|
|
●
|
Competition from illicit sources may have an adverse effect on our
overall sales volume, restricting the ability to increase selling prices and damaging brand equity.
|
|
|
|
|
|
|
●
|
Increases in tobacco and ENDS-related excise taxes have been proposed
or enacted and are likely to continue to be proposed or enacted in numerous jurisdictions.
|
|
|
|
|
|
|
●
|
The market for ENDS products is subject to a great deal of uncertainty
and is still evolving. The FDA has implemented a de facto flavor ban by denying the PMTAs for all such products to date, including
the non-tobacco flavored BIDI® Sticks. In addition, several states and localities have banned the sale of flavored tobacco
products, including ENDS. If flavors are prohibited, the use of ENDS products may decline significantly.
|
|
|
|
|
|
|
●
|
Bidi has not obtained premarket authorization from the FDA and its
pending PMTAs for our non-tobacco flavored BIDI® Sticks have been denied. Only our Classic (tobacco) BIDI® Stick is currently
marketed pursuant to a policy of FDA enforcement discretion. There could be a material adverse impact on our business development
efforts if the FDA determines that Bidi products are not subject to this compliance policy, or if our products become subject to
increased regulatory compliance burdens imposed by the FDA and other regulatory or legislative bodies.
|
|
|
|
|
|
|
●
|
Our supply to our wholesalers and retailers is dependent on the demands
of their customers who are sensitive to increased sales taxes and economic conditions affecting their disposable income.
|
|
|
|
|
|
|
●
|
We may be subject to increasing international control and regulation.
|
|
|
|
|
|
|
●
|
Our business may be damaged by events outside of our suppliers’
control, such as the impact of epidemics (e.g., COVID-19), situations such as e-cigarette use-associated lung injury (EVALI),
political upheavals, or natural disasters. The COVID-19 coronavirus could have further adverse impacts on our business, including
several key activities that are critical to our success.
|
|
|
|
|
|
|
●
|
Reliance on information technology means a significant disruption
could affect our communications and operations, and security and privacy breaches may expose us to liability and cause us to lose
customers.
|
|
|
|
|
|
|
●
|
We may fail to manage our growth.
|
|
|
|
|
|
|
●
|
We are subject to fluctuations in our results that make it difficult
to track trends and develop strategies.
|
|
|
|
|
|
|
●
|
Adverse U.S. and global economic conditions could negatively impact
us.
|
|
|
|
|
|
|
●
|
The departure of key management personnel and the failure to attract
and retain talent could adversely affect our operations.
|
|
|
|
|
|
|
●
|
Our insurance may be insufficient to cover losses that may occur
as a result of our operations.
|
|
|
|
|
|
|
●
|
Our charter and bylaws, as well as Delaware law and certain regulations,
could discourage or prohibit acquisition bids or merger proposals, which may adversely affect the market price of the Common Stock.
|
|
|
|
|
|
|
●
|
Future offerings of debt or equity securities may rank senior to
the Common Stock, and we may issue preferred stock whose terms could adversely affect the voting power or value of the Common Stock.
|
|
|
●
|
Raising additional capital may cause dilution to our existing stockholders,
restrict our operations or require us to relinquish rights to our technologies, if any, or our Products.
|
|
|
|
|
|
|
●
|
The market prices for the Common Stock are volatile and will fluctuate,
and future sales of shares of the Common Stock by officers and directors may negatively impact the market price for the Common
Stock.
|
|
|
|
|
|
|
●
|
You may experience immediate and substantial dilution.
|
|
|
|
|
|
|
●
|
There can be no assurance that we will be able to comply with the
continued listing standards of the Nasdaq Capital Market (“Nasdaq”).
|
|
|
|
|
|
|
●
|
We have broad discretion in the use of the net proceeds from this
offering and may not use them effectively.
|
|
|
|
|
|
|
●
|
Concentration of ownership among our officers, directors and principal
stockholders may prevent other stockholders from influencing significant corporate decisions and depress our stock price.
|
|
|
|
|
|
|
●
|
The Common Stock may become the target of a “short squeeze.”
|
|
|
|
|
|
|
●
|
There is no public market for the common warrants, which are speculative
in nature, and holders of the common warrants will not have rights of holders of the Common Stock until such warrants are exercised.
|
|
|
|
|
|
|
●
|
If securities or industry analysts fail to continue publishing research
about our business, if they change their recommendations adversely or if our results of operations do not meet their expectations,
our stock price and trading volume could decline.
|
|
|
|
|
|
|
●
|
We do not currently pay dividends on the Common Stock and have no
intention to pay dividends on the Common Stock for the foreseeable future.
|
|
|
|
|
|
|
●
|
For as long as we are a “emerging growth company” we
intend to take advantage of reduced disclosure and governance requirements applicable to emerging growth companies, which could
result in the Common Stock being less attractive to investors and could make it more difficult for us to raise capital as and when
we need it.
|
|
|
|
|
|
|
●
|
If we fail to achieve and subsequently maintain an effective system
of internal control over financial reporting in the future, we may not be able to accurately report our financial condition, results
of operations or cash flows, which may adversely affect investor confidence in us and, as a result, the value of the Common Stock.
|
|
|
|
|
|
|
●
|
Our disclosure controls and procedures may not prevent or detect
all errors or acts of fraud.
|
|
|
|
|
|
|
●
|
We have incurred, and will continue to incur, increased costs as
a result of operating as a public company, and our management has been required, and will continue to be required, to devote substantial
time to new compliance initiatives.
|
Corporate Information
We were incorporated on September 4, 2018 in
the State of Delaware. Effective July 12, 2019, we changed our corporate name from Quick Start Holdings, Inc. to Kaival Brands
Innovations Group, Inc. The name change was effected through a parent/subsidiary short-form merger of Kaival Brands Innovations
Group, Inc., our wholly-owned Delaware subsidiary formed solely for the purpose of the name change, with and into us. We were the
surviving entity. Our corporate headquarters are located at 4460 Old Dixie Highway, Grant, Florida 32949, and our telephone number
is +1 (833) 452-4825. Our website address is www.kaivalbrands.com. The information contained on our website or that can be accessed
through our website is not part of this prospectus and investors should not rely on any such information in deciding whether to
purchase our securities.
THE OFFERING
|
Issuer
|
Kaival Brands Innovations
Group, Inc.
|
|
|
|
|
Shares of Common Stock and
common warrants offered by us pursuant to this prospectus supplement
|
4,700,000
Shares of Common Stock and common warrants to purchase 3,525,000 shares of Common Stock.
|
|
Over-allotment
Option
|
We
have granted the underwriter a 45-day option
to purchase up to an additional 705,000 shares
of Common Stock and/or additional common warrants
to purchase up to an additional 528,750 shares
of Common Stock from us to cover over-allotments,
if any.
|
|
|
|
|
Offering
price per share and 0.75 of an accompanying common warrant
|
$1.70
|
|
|
|
|
Shares of Common Stock outstanding
prior to this offering
|
23,628,187 Shares
|
|
|
|
|
Shares
of Common Stock to be outstanding after this offering
|
28,328,187 Shares (or 29,033,187
Shares of the underwriter exercises its over-allotment option in full).
|
|
|
|
|
Description
of common warrants
|
The common warrants will be exercisable
beginning on the date of issuance and expire on the five-year anniversary of the date of issuance at an initial exercise price
per share equal to $1.90, subject to appropriate adjustment in the event of recapitalization events, stock dividends, stock splits,
stock combinations, reclassifications, reorganizations or similar events affecting the Common Stock. Notwithstanding the foregoing,
we shall not effect any exercise of common warrants to the extent that, after giving effect to an exercise, the holder of common
warrants (together with such holder’s affiliates, and any persons acting as a group together with such holder or any of
such holder’s affiliates) would beneficially own a number of shares of the Common Stock in excess of 4.99% (or, at the election
of the purchaser prior to the date of issuance, 9.99%) of the shares of the Common Stock then outstanding after giving effect
to such exercise.
|
|
|
|
|
Use
of proceeds
|
We expect to receive net proceeds
of approximately $7,031,200 from this offering after deducting the underwriter fees and discounts and estimated offering
expenses payable by us. We intend to use the net proceeds from this offering for general corporate purposes, including working
capital. See “Use of Proceeds.”
|
|
|
|
|
Transfer
agent, registrar and warrant agent
|
The transfer agent and registrar
of the Common Stock and the Warrant Agent for the common warrants is Vstock Transfer, LLC.
|
|
|
|
|
Nasdaq
Capital Market symbol
|
The
Common Stock is traded on Nasdaq under the symbol “KAVL.” We do not intend to apply to list the common warrants on
Nasdaq or on any other trading market.
|
|
|
|
|
Risk
factors
|
Investing
in our securities involves a high degree of risk. See “Risk Factors” beginning on page S-12 of this prospectus to
read about factors that you should consider carefully before buying our securities.
|
The number of shares of Common Stock that will be outstanding after
this offering is based on 23,600,597 shares outstanding as of July 31, 2021, and excludes:
|
|
●
|
150,000 shares of Common Stock issuable upon exercise of options to purchase the Common Stock outstanding as of July 31, 2021 at a weighted average exercise price of $20.49 per share;
|
|
|
|
|
|
|
●
|
555,417 shares of Common Stock reserved as of July 31, 2021 for future issuance under our 2020
Stock Option and Incentive Plan;
|
|
|
|
|
|
|
●
|
7,495,833 shares of Common Stock reserved as of July 31, 2021 for
future issuance under our 2020 Stock Option and Incentive Plan;
|
|
|
|
|
|
|
●
|
25,000,000 shares of Common Stock issuable upon exercise of convertible preferred stock outstanding as of July 31, 2021;
|
|
|
|
|
|
|
●
|
Up to 2,000,000 shares of Common Stock that may be issuable in the future to a consultant dependent upon the resolution of a disagreement between the consultant and us with respect to the terms of our arrangement; and
|
|
|
|
|
|
|
●
|
3,525,000 shares of
Common Stock issuable upon the common warrants offered hereby (or 4,053,750 shares of Common Stock issuable upon the common
warrants if the underwriter exercises its over-allotment option in full).
|
Except as otherwise noted, all information in this prospectus supplement
reflects and assumes no exercise of the underwriter’s over-allotment option.
RISK FACTORS
Our business is subject to numerous risks.
We caution you that the following important factors, among others, could cause our actual results to differ materially from those
expressed in forward-looking statements made by us or on our behalf in filings with the SEC, press releases, communications with
investors and oral statements. Any or all of our forward-looking statements contained in this prospectus supplement and in any
other public statements we make may turn out to be wrong. They can be affected by inaccurate assumptions we might make or by known
or unknown risks and uncertainties. Many factors mentioned in the discussion below will be important in determining future results.
Consequently, no forward-looking statement can be guaranteed. Actual future results may differ materially from those anticipated
in forward-looking statements. We undertake no obligation to update any forward-looking statements, whether as a result of new
information, future events or otherwise. You are advised, however, to consult any further disclosure we make in our reports filed
with the SEC.
Risks Related to Our Business and Industry
We have a limited operating history, and
our historical operating and financial results may not be indicative of future performance, which, along with the relative early
stage of the ENDS industry, makes it difficult to predict our future business prospects and financial performance.
We have had a limited operating history. Our
business and prospects may thus be difficult to evaluate, and our prospects will be dependent on our ability to meet a number of
challenges. Further, the ENDS industry is relatively new and is rapidly evolving. Changes in existing laws, regulations and policies
and the issuance of new laws, regulations, policies, and any other entry barriers in relation to the ENDS industry may materially
and adversely affect our business operations.
The FDA has recently issued Bidi an MDO,
which prevents us from marketing Products that make up a substantial majority of our revenue. If Bidi is unsuccessful in its appeal
of the MDO, if our conclusion as to our ability to sell the Arctic (menthol) BIDI® Stick is challenged or if our expectations
as to how the purchasing habits of consumers may change to offset the loss of revenue with respect to certain of our Products,
our revenues and, thereby our financial results and condition, would be materially adversely affected.
As of September 23, 2021, the FDA announced
that it has taken action on over 93% of PMTAs and issued MDOs for more than 1,167,000 flavored ENDS products, while issuing zero
marketing authorizations. Bidi was among the many companies that received a MDO for its non-tobacco flavored BIDI® Sticks.
While Bidi intends to appeal this decision on various grounds, its appeal may not be successful.
Further, the MDO received by Bidi covering
flavored ENDS products included its Arctic (menthol) BIDI® Stick, which is not consistent with the FDA’s public statements
and press releases stating that tobacco and menthol ENDS are not deemed flavored products subject to the MDOs. Because its Arctic
(menthol) BIDI® Stick has a menthol characterizing flavor, Bidi believes that this menthol BIDI® Stick may have been inadvertently
included on the MDO. Accordingly, along with the Classic (tobacco) BIDI® Stick, Bidi intends to continue to manufacture and
market its Arctic (menthol) BIDI® Stick for distribution by us, and we intend to distribute both products. Historically, substantially
all of our revenues were derived from sales of flavored BIDI® Sticks, including the Arctic (menthol) BIDI® Stick, sales
of which constituted approximately 15.2% and 18.5%, respectively, of our total sales of BIDI® Sticks for the three and nine
months ended July 31, 2021.
If the FDA disagrees with Bidi’s position,
or issues a warning letter or takes other action against Bidi resulting in us not being able to distribute the Arctic BIDI®
Stick in the United States, our revenues and, thereby our financial results and condition, would be materially adversely affected.
Following the issuance of the MDOs, nearly
all manufacturers, not only Bidi, will be limited to manufacturing and selling only tobacco and menthol ENDS products. Accordingly,
we believe that consumers may modify their purchases to shift to products that are available, and thus that a substantial amount
of our revenue that had previously been generated from sales of flavored BIDI® Sticks (other than Arctic) may be replaced through
sales of the Classic (tobacco) and Arctic (menthol) BIDI® Sticks, given that consumers will only be able to purchase, on a
legal basis, non-flavored ENDS products. Accordingly, we believe revenues for tobacco and menthol ENDS products will restore some
of the revenue we derived from the sale of flavored products. Moreover, based on sales and consumer data, we believe that our consumers
are loyal to the Bidi brand and BIDI® Sticks and that they thus are likely to continue to purchase our Classic (tobacco) and
Arctic (menthol) BIDI® Sticks in lieu of the flavored BIDI® Sticks they may have bought in the past.
However, if Bidi’s still-pending PMTA
for the Classic BIDI® Stick is ultimately unsuccessful, or if the MDO appeal is unsuccessful, or if the FDA disagrees with
Bidi’s position regarding the menthol (Arctic) BIDI® Stick, issues a warning letter, or takes other action against Bidi
resulting in us not being able to distribute the menthol (Arctic) BIDI® Stick in the United States, or consumers do not purchase
the tobacco (Classic) or menthol (Arctic) BIDI® Sticks, our revenues and, thereby our financial results and condition, would
be materially adversely affected. Our financial results and condition will also be significantly impacted by our ability to continue
to sell the Arctic (menthol) BIDI® Stick and the degree to which sales of the Classic (tobacco) and Arctic (menthol) BIDI®
Sticks replace sales of flavored products that are now prohibited.
If Bidi’s
planned PMTA for the tobacco-derived nicotine based formulation of the BIDI® Pouch is not authorized by the FDA,
we will not be able to sell the BIDI® Pouch in the U.S.
We are currently planning to initiate distribution
of the BIDI® Pouch initially outside the U.S. Bidi is also planning to submit a PMTA for a BIDI® Pouch manufactured using
a tobacco-derived nicotine formula. The BIDI® Pouch cannot be distributed in the U.S. unless the PMTA is authorized. If the
PMTA is not authorized, or if authorization is materially delayed, our revenues and, thereby our financial results and condition,
would be materially adversely.
If it is determined or perceived that the
usage of ENDS products poses long-term health risks, the use of ENDS products may decline significantly, which may materially and
adversely affect our business, financial condition and results of operations.
Negative publicity
on the health consequences of ENDS products or other similar devices may also adversely affect the usage of ENDS products. For
example, the FDA and the United States Centers for Disease Control and Prevention (“CDC”) issued a joint statement
on August 30, 2019, linking a number of cases of respiratory illnesses to ENDS product use. On November 8, 2019, the
CDC announced that it had preliminarily linked cases of severe respiratory illness to the presence of Vitamin E acetate, which
was found in certain Tetrahydrocannabinol (THC)-containing ENDS cartridges for non-electronic nicotine delivery systems (non-ENDS)
products that may have been obtained illegally. However, evidence is not sufficient to rule out the contribution of other chemicals
of concern, including chemicals in either THC or non-THC products. In January 2020, after further research, the FDA and CDC recommended
against the use of THC-containing ENDS products, especially those from unofficial sources, and that the underage, pregnant women
and adults who do not currently use tobacco products should not start using ENDS products. On February 25, 2020, the CDC issued
a final update, stating that the number of cases of severe respiratory illnesses had declined to single digits as of February 9,
2020. The CDC also reconfirmed that (i) Vitamin E acetate, which was found in some THC-containing ENDS cartridges for non-ENDS
ENDS products that were mostly obtained illegally, was strongly linked to and indicated to be the primary cause of the severe respiratory
illnesses, and (ii) THC-containing ENDS products from informal sources were linked to most cases of severe respiratory illnesses.
Furthermore, there have been recent claims that users of ENDS products may suffer a greater risk of more serious COVID-19 complications.
However, it remained unclear whether the exposure to toxic chemicals through ENDS product usage will increase the risk of COVID-19.
Research regarding
the actual causes of these illnesses is still ongoing. If ENDS product usage is determined or perceived to pose long-term health
risks or to be linked to illnesses, the usage of ENDS products may significantly decline, which would have a material adverse effect
on our business, financial condition, and results of operations. Although we currently do not offer products containing THC, any
perceived correlation between THC and Vitamin E acetate may adversely affect the public’s perception of ENDS products in
general, regardless of whether such products contain THC and/or Vitamin E.
We
rely exclusively on Bidi as the supplier of the Products that we distribute. The loss of this relationship, or any negative impacts
on Bidi’s ability to product the Products, would severely harm our business.
Pursuant to the A&R Distribution Agreement
between us and Bidi, Bidi has engaged is to act as the sole distributor of the ENDS products and related components, including
the BIDI® Stick, manufactured by Bidi. Any failure by Bidi to perform its obligation under the A&R Agreement could have
a material adverse effect on our revenue and operating results and operating cash flows; and could impair the strength of our brand.
In addition, because of our dependence on Bidi
as the exclusive supplier of Products, any loss of our relationship with Bidi, or any adverse change in the financial health of
Bidi that would affect its ability to perform its obligations under the A&R Agreement, would have a material adverse effect
on our revenue, operating results and ability to run our business.
Further, Bidi is subject to supply shortages
and interruptions, long lead times and act-of-God events such as global pandemics, weather related catastrophes, or conflict, any
of which could disrupt the operations of Bidi and have a material adverse impact on our results of operations. We may be unable
to identify or contract with new suppliers or producers in the event of a disruption to our supply, and could experience a material
adverse effect on our revenue, operating results and ability to run our business.
The terms of our agreements with affiliated
entities including our A&R Distribution Agreement with Bidi may not always be as favorable to us as the terms that may be obtained
by arms’ length negotiation.
We currently are, and we anticipate that we
will continue to be, substantially dependent on our relationships with our affiliated entities, including Bidi. We believe that
our current arrangement with Bidi provides our business with stability and transparency. Although we believe that the terms of
the A&R Distribution Agreement are as favorable to us as what we could have obtained in an arm’s length transaction,
there can be no assurance that this arrangement or any future agreements that we enter into with Bidi or any other affiliated entity
will be as favorable to us as we may be able to negotiate with unaffiliated parties.
Our relationship with Bidi is subject to
change.
We currently have no intellectual property
rights (other than the trademarks KAIVAL BRANDS and KAIVAL LABS) and rely on the intellectual property rights, including logos,
trademarks, and trade names, of Bidi that were granted to us pursuant to the A&R Distribution Agreement to be used in connection
with the marketing, advertisement, and sale of the Products. We also indirectly rely on Bidi’s intellectual property rights
related to the Products, such as patents. We have from time to time considered, and discussed with Bidi, potential alterations
to this arrangement, including a potential acquisition by us of all or a portion of the intellectual property owned by Bidi and
related to the Products. Should we pursue such a transaction, it would be a “related party transaction,” as defined
by the listing rules of The Nasdaq Stock Market, LLC, and thus subject to the review of the Audit Committee of our Board of Directors
(our “Board”). Further, should we undertake such a transaction, then we would become responsible to respond if a third-party
challenged Bidi’s patents, or infringed upon such rights, in which case our business could be materially adversely affected.
We may not be successful in maintaining
the consumer brand recognition and loyalty of our Products and face intense competition and may fail to compete effectively.
We compete in a market that relies on innovation
and the ability to react to evolving consumer preferences and, thus, are subject to significant competition in the ENDS market,
and larger tobacco industry and compete against companies in such market and industry that have access to significant resources
in terms of technology, relationships with suppliers and distributors and access to cash flow and financial markets.
Consumer perceptions of the overall safety
of tobacco and nicotine-based products is likely to continue to shift, and our success depends, in part, on our ability to anticipate
these shifting tastes and the rapidity with which the markets in which we compete will evolve in response to these changes on a
timely and affordable basis. If we are unable to respond effectively and efficiently to changing consumer preferences, the demand
for our Products may decline, which could have a material adverse effect on our business, results of operations, and financial
condition.
Regulations may be enacted in the future, particularly
in light of increasing restrictions on the form and content of marketing of tobacco products, that would make it more difficult
to appeal to our consumers or to leverage existing recognition of the Bidi brand, or other brands that we own or license in the
future. Furthermore, even if we are able to continue to distinguish our Products, there can be no assurance that the sales, marketing,
and distribution efforts of our competitors will not be successful in persuading consumers of our Products to switch to their products.
Many of our competitors have greater access to resources than we do, which better positions them to conduct market research in
relation to branding strategies or costly marketing campaigns. Any loss of consumer brand loyalty to our Products or reduction
of our ability to effectively brand our Products in a recognizable way will have a material effect on our ability to continue to
sell our Products and maintain our market share, which could have a material adverse effect on our business, results of operations,
and financial condition.
The competitive environment and our competitive
position are also significantly influenced by economic conditions, the state of consumer confidence, competitors’ introduction
of low-priced products or innovative products, higher taxes, higher absolute prices and larger gaps between price categories and
product regulation that diminishes the consumer’s ability to differentiate tobacco products. Due to the impact of these factors,
as well as higher state and local excise taxes and the market share of deep discount brands, the tobacco industry has become increasingly
price competitive. As we seek to adapt to the price competitive environment, our competitors that are better capitalized may be
able to sustain price discounts for long periods of time by spreading the loss across their expansive portfolios, with which we
are not positioned to compete.
“Big tobacco” has also established
its presence in the ENDS market and has begun to make investments in the alternative space. There can be no assurance that our
Products will be able to compete successfully against these companies or any of our other competitors, some of which have far greater
resources, capital, experience, market penetration, sales and distribution channels than do we.
Our distribution efforts rely in part on
our ability to leverage relationships with large retailers and national chains.
Our distribution efforts rely in part on our
ability to leverage relationships with large retailers and national chains to sell and promote our Products, which is dependent
upon the strength of the Bidi brand name and, in the future, any brand names that we may own or license, and our salesforce effectiveness.
To maintain these relationships, we must continue to supply products that will bring steady business to these retailers and national
chains. We may not be able to sustain these relationships or establish other relationships with such entities, which could have
a material adverse effect on our ability to execute our branding strategies, our ability to access the end-user markets with our
Products, or our ability to maintain our relationships with the manufacturer and sub-distributors of our Products. For example,
if we are unable to meet benchmarking provisions in certain of our contracts or if we are unable to maintain and leverage our retail
relationships on a scale sufficient to make us an attractive distributor, it would have a material adverse effect on our ability
to act as sole distributor for Bidi, and on our business, results of operations and financial condition.
In addition, there are factors beyond our control
that may prevent us from leveraging existing relationships, such as industry consolidation. If we are unable to develop and sustain
relationships with large retailers and national chains or are unable to leverage those relationships due to factors such as a decline
in the role of brick-and-mortar retailers in the North American economy, our capacity to maintain and grow brand and product recognition
and increase sales volume will be significantly undermined. In such an event, we may ultimately be forced to pursue and rely on
local and more fragmented sales channels, which will have a material adverse effect on our business, results of operations and
financial condition.
Competition from illicit sources may have
an adverse effect on our overall sales volume, restricting the ability to increase selling prices and damaging brand equity.
Illicit trade and tobacco trafficking in the
form of counterfeit products, smuggled genuine products and locally manufactured products on which applicable taxes or regulatory
requirements are evaded, represent a significant and growing threat to the legitimate tobacco industry. Factors such as increasing
tax regimes, regulatory restrictions, and compliance requirements are encouraging more consumers to switch to illegal, cheaper
tobacco-related products and providing greater rewards for smugglers. Illicit trade can have an adverse effect on our overall sales
volume, restrict the ability to increase selling prices, damage brand equity and may lead to commoditization of our Products.
Although we combat counterfeiting of our Products
by engaging in certain tactics, such as requiring all sales force personnel to randomly collect our Products from retailers in
order to be tested by our quality control team, maintaining a quality control group that is responsible for identifying counterfeit
products and surveillance of retailers we suspect are selling counterfeit Products through our own secret shopper force, no assurance
can be given that we will be able to detect or stop sales of all counterfeit products. In addition, we have in the past and will
continue to bring suits against retailers and distributors that sell certain counterfeit products. While we have been successful
in securing financial recoveries from and helping to obtain criminal convictions of counterfeiters in the past, no assurance can
be given that we will be successful in any such suits or that such suits will be successful in stopping other retailers or distributors
from selling counterfeit products. Even if we are successful, such suits could consume a significant amount of management’s
time and could also result in significant expenses to us. Any failure to track and prevent counterfeiting of our Products could
have a material adverse on our ability to maintain or effectively compete for our Products we distribute under the Bidi brand names,
which would have a material adverse effect on our business, results of operations and financial condition.
Our Products are regulated by the FDA, which
has broad regulatory powers. Increases in tobacco-related taxes have been proposed or enacted and are likely to continue to be
proposed or enacted in numerous jurisdictions.
Tobacco products, premium cigarette papers
and tubes have long been subject to substantial federal, state and local excise taxes. Such taxes have frequently been increased
or proposed to be increased, in some cases significantly, to fund various legislative initiatives or further disincentivize tobacco
usage. Since 1986, smokeless products have been subject to federal excise tax. Federally, smokeless products are taxed by weight
(in pounds or fractional parts thereof) manufactured or imported.
The market for ENDS products is subject
to a great deal of uncertainty and is still evolving.
ENDS products, having recently been introduced
to market over the past 10-15 years, are at a relatively early stage of development, and represent core components of a market
that is evolving rapidly, highly regulated and characterized by a number of market participants. Rapid growth in the use of, and
interest in, ENDS products is recent, and may not continue on a lasting basis. The demand and market acceptance for these products
is subject to a high level of uncertainty. Therefore, we are subject to all the business risks associated with a new enterprise
in an evolving market.
For example, ENDS products that are non-tobacco
flavored continue to face the threat of prohibition at the local level, as many state and local authorities and attorneys general
push for bans or request FDA to deny a PMTA for flavored ENDS. To date, at least four states have banned the sale of flavored ENDS
(e.g., New York, New Jersey, Rhode Island, and Massachusetts), with several more considering similar bans (e.g., Maryland, California,
and Connecticut). As the September 9, 2021 PMTA review deadline has now past, FDA has implemented a de facto ban of flavored ENDS
by denying over 93% of pending applications, while issuing zero marketing authorizations.
If flavors are prohibited, the use of ENDS
products may decline significantly, which may materially and adversely affect our business, financial condition and results of
operations. Continued evolution, uncertainty, and the resulting increased risk of failure of our new and existing product offerings
in this market could have a material adverse effect on our ability to build and maintain market share and on our business, results
of operations and financial condition.
Bidi has not obtained premarket authorization
from the FDA and its pending PMTAs for our non-tobacco flavored BIDI® Sticks have been denied. Only our Classic (tobacco) BIDI®
Stick is currently marketed pursuant to a policy of FDA enforcement discretion. There could be a material adverse impact on our
business development efforts if the FDA determines that our Products are not subject to this compliance policy, of if our Products
become subject to increased regulatory compliance burdens imposed by the FDA and other regulatory or legislative bodies.
An ENDS product that contains tobacco-derived
nicotine, or that falls within the meaning of a component or part of an ENDS product, is a “tobacco product” as defined
in the Food, Drug and Cosmetic Act (the “FDCA”). Upon effectiveness in August 2016 of the FDA’s rule pursuant
to which it regulates all tobacco products (the “Deeming Rule”), ENDS products became regulated tobacco products subject
to all the FDCA requirements, including premarket review for “new” tobacco products (i.e., products introduced
or modified after the February 15, 2007 “grandfather date”). With respect to premarket review, because there are no
confirmed grandfathered ENDS that were on the market as of February 15, 2007, all ENDS products require FDA marketing authorization
through the PMTA process. For a product to be authorized through the PMTA pathway, the applicant must demonstrate that the product
is appropriate for the protection of the public health (“APPH”). This public or population health standard is quite
high, and requires considering the product’s risks and benefits to the population as a whole, including users and nonusers
of the tobacco product, and taking into account the increased or decreased likelihood that existing users of tobacco products will
stop using such products, and the increased or decreased likelihood that those who do not use tobacco products will start using
such products. All ENDS must go through this scientifically rigorous PMTA review process to prove they are APPH before receive
marketing authorization. Moreover, all ENDS that are legally on the market today are subject to FDA’s enforcement “compliance
policy.” While the FDA could not modify the statute’s 2007 grandfather date, in the Deeming Rule, the agency established
an enforcement “compliance policy” permitting non-grandfathered deemed products (including ENDS) that were on the market
as of the effective date of the Deeming Rule (August 8, 2016) to remain on the market for a certain amount of time until premarket
applications became due. This PMTA deadline for currently marketed deemed products has shifted over the years from August 2018
to August 2022 to finally, after a court order, September 9, 2020. To be legally marketed today, ENDS must have been on the U.S.
market on August 8, 2016 and subject to a PMTA submitted to FDA on or before September 9, 2020. If they meet these criteria, then
pursuant to the 2020 district court order and FDA’s current enforcement policy, they may remain on the market during FDA’s
PMTA review for up to one year, or until September 9, 2021. FDA has further indicated that the court order continues to permit
the agency to determine at its discretion (i.e., on a case-by-case basis) whether to allow an ENDS product subject to a
timely-submitted PMTA to remain on the market after the one-year compliance period ends on September 9, 2021. Any ENDS product
marketed without authorization after that time would be subject to enforcement by FDA. If a new tobacco product is commercialized
without having the requisite marketing authorization in effect, FDA may deem the product adulterated and/or misbranded. Introduction
into interstate commerce of any such violative product is prohibited and may lead to a seizure, injunction, or other enforcement
action. On September 8, 2021, despite submitting a comprehensive PMTA and continuing to develop robust and reliable product-specific
scientific evidence demonstrating the public health benefit of its flavored ENDS products, Bidi, along with nearly every other
company in the ENDS industry, was among the many companies that received an MDO for its non-tobacco flavored BIDI® Sticks,
including its Arctic (menthol) BIDI® Stick, which we believe the FDA mischaracterized as “flavored.” However, because
its Arctic BIDI® Stick has a menthol characterizing flavor, Bidi Vapor believes that this menthol BIDI® Stick was inadvertently
included on the MDO. This position is aligned with the FDA’s public statements and press releases stating that tobacco and
menthol ENDS are not deemed flavored products subject to the MDOs. Accordingly, along with the Classic (tobacco) BIDI® Stick,
Bidi intends to continue to manufacture and market its Arctic (menthol) BIDI® Stick for distribution by us.
Some of our Product offerings through Bidi
are subject to developing and unpredictable regulation.
Our Products sold through our distribution
network may be subject to uncertain and evolving federal, state and local regulations, including hemp, non-THC cannabidiol (CBD)
and other non-tobacco consumable products. Enforcement initiatives by those authorities are therefore unpredictable and impossible
to anticipate. We anticipate that all levels of government, which have not already done so, are likely to seek in some way to regulate
these products, but the type, timing, and impact of such regulations remains uncertain. These regulations include or could include
restrictions including prohibitions on certain form factors, such as smokable hemp products, or age restrictions. Accordingly,
we cannot give any assurance that such actions would not have a material adverse effect on this emerging business.
Significant increases in state and local
regulation of our Products have been proposed or enacted and are likely to continue to be proposed or enacted in numerous jurisdictions.
The Prevent All Cigarette Trafficking (PACT)
Act (the “PACT Act”), which went into effect in June 2010, amended the Jenkins Act and initially only applied to the
sales of cigarettes, roll-your-own tobacco, and smokeless tobacco. Specifically, the PACT Act regulates the sale, transfer or shipment
of these products for both business to business transactions as well as “delivery sales” which are defined as any sale
of cigarettes, roll-your-own tobacco or smokeless tobacco where the consumer orders the product remotely, and prohibits such deliveries
through the U.S. Postal Service (“USPS”), except in certain circumstances (e.g., business-to-business deliveries).
Under the enactment of the Preventing Online
Sales of E-Cigarettes to Children Act (part of the larger 2021 Consolidated Appropriations Act), effective March 27, 2021, the
definition of “cigarettes” in the PACT Act was amended to include ENDS, which is defined as “any electronic device
that, through an aerosolized solution, delivers nicotine, flavor, or any other substance to the user inhaling from the device,”
including “an e-cigarette; an e-hookah; an e-cigar; a vape pen; an advanced refillable personal vaporizer; an electronic
pipe; and any component, liquid, part, or accessory of a device described above, without regard to whether the component, liquid,
part, or accessory is sold separately from the device.” As such, delivery sales of the BIDI® Stick are subject to the
PACT Act.
The PACT Act requires all sellers to register
with the Bureau of Alcohol, Tobacco, Firearms and Explosives (ATF), as well as the tobacco tax administrators of the states into
which a shipment is made or in which an advertisement or offer is disseminated. Delivery sellers who ship cigarettes (including
ENDS) or smokeless tobacco to consumers are further required to label packages as containing tobacco, verify the age and identity
of the customer at purchase, use a delivery method (other than through the USPS) that checks ID and obtains adult customer signature
at delivery, and maintain records of delivery sales for a period of four years after the date of sale, among other things. Delivery
sellers are also required to file a monthly report with the state tobacco tax administrator and any other local or tribal entity
that taxes the sale of the products. Such reports must include the name and address of the persons delivering and receiving the
shipment and the brand and quantity of the “cigarettes” that were shipped. These requirements apply to all sales, including
sales to consumers and sales between businesses.
In addition to the de facto FDA flavor ban
that has resulted from the denial of nearly all PMTAs for flavored ENDS, ENDS products that are non-tobacco flavored continue to
face the threat of prohibition at the local level, as many state and local authorities and attorneys general push for bans or request
FDA to deny PMTAs for flavored ENDS. To date, at least four states have banned the sale of flavored ENDS (e.g., New York,
New Jersey, Rhode Island, and Massachusetts), with several more considering similar bans (e.g., Maryland, California, and
Connecticut).
Our supply to our wholesalers and retailers
is dependent on the demands of their customers who are sensitive to increased sales taxes and economic conditions affecting their
disposable income.
Consumer purchases of tobacco products are
historically affected by economic conditions, such as changes in employment, salary and wage levels, the availability of consumer
credit, inflation, interest rates, fuel prices, sales taxes, and the level of consumer confidence in prevailing and future economic
conditions. Discretionary consumer purchases, such as the BIDI® Stick, may decline during recessionary periods or at other
times when disposable income is lower, and taxes may be higher.
We may be subject to increasing international
control and regulation.
The World Health Organization’s Framework
Convention on Tobacco Control (“FCTC”) is the first international public health treaty that establishes a global agenda
to reduce initiation of tobacco use and regulate tobacco to encourage tobacco cessation. Over 170 governments worldwide have ratified
the FCTC. The FCTC has led to increased efforts to reduce the supply and demand of tobacco products and to encourage governments
to further regulate the tobacco industry. The tobacco industry expects significant regulatory developments to take place over the
next few years, driven principally by the FCTC. Regulatory initiatives that have been proposed, introduced or enacted include:
|
|
●
|
the levying of substantial and increasing
tax and duty charges;
|
|
|
●
|
restrictions or bans on advertising, marketing
and sponsorship;
|
|
|
●
|
the display of larger health warnings,
graphic health warnings and other labeling requirements;
|
|
|
●
|
restrictions on packaging design, including
the use of colors and generic packaging;
|
|
|
●
|
restrictions or bans on the display of
tobacco product packaging at the point of sale, and restrictions or bans on cigarette vending machines;
|
|
|
●
|
requirements regarding testing, disclosure
and performance standards for tar, nicotine, carbon monoxide and other smoke constituents levels;
|
|
|
●
|
requirements regarding testing, disclosure
and use of tobacco product ingredients;
|
|
|
●
|
increased restrictions on smoking in public
and work places and, in some instances, in private places and outdoors;
|
|
|
●
|
elimination of duty-free allowances for
travelers; and
|
|
|
●
|
encouraging litigation against tobacco
companies.
|
Our business may be damaged by events outside
of our suppliers’ control, such as the impact of epidemics (e.g., COVID-19), political upheavals, or natural disasters. The
COVID-19 coronavirus could adversely impact our business, including several key activities that are critical to our success.
The global outbreak of COVID-19 continues to
rapidly evolve. As a result, businesses have closed and limits have been placed on travel. The extent to which COVID-19 may impact
our business will depend on future developments, which are highly uncertain and cannot be predicted with confidence, such as the
ultimate impact of the disease on specific geographies, the duration of the outbreak, travel restrictions and social distancing
in the United States and other countries, business closures or business disruptions and the effectiveness of actions taken in the
United States and other countries to contain and treat the disease.
The spread of COVID-19 throughout the world
has also created global economic uncertainty, which may cause partners, suppliers and potential customers to closely monitor their
costs and reduce their spending budget. Either of the foregoing could materially adversely affect our research and development
activities, clinical trials, supply chain, financial condition and cash flows.
If the COVID-19 outbreak continues to spread,
we may need to limit operations or implement other limitations on our activities. There is a risk that other countries or regions
may be less effective at containing COVID-19, in which case the risks described herein could be elevated significantly.
Reliance on information technology means
a significant disruption could affect our communications and operations.
We increasingly rely on information technology
systems for our internal communications, controls, reporting and relations with customers and suppliers and information technology
is becoming a significantly important tool for our sales staff. In addition, our reliance on information technology exposes us
to cyber-security risks, which could have a material adverse effect on our ability to compete. Security and privacy breaches may
expose us to liability and cause us to lose customers or may disrupt our relationships and ongoing transactions with other entities
with whom we contract throughout our network. The failure of our information systems to function as intended, or the penetration
by outside parties’ intent on disrupting business processes, could result in significant costs, loss of revenue, assets or
personal or other sensitive data and reputational harm.
Security and privacy breaches may expose
us to liability and cause us to lose customers.
Federal and state laws require us to safeguard
our wholesalers’, retailers’, and consumers’ financial information, including credit information. Although we
have established security procedures to protect against identity theft and the theft of our customers’ financial information,
our security and testing measures may not prevent security breaches. We cannot guarantee that a future breach would not result
in material liability or otherwise harm our business. In the event of any such breach, we may be required to notify governmental
authorities or consumers under breach disclosure laws, indemnify consumers or other third parties for losses resulting from the
breach, and expend resources investigating and remediating any vulnerabilities that contributed to the occurrence of the breach.
We rely on third-party technology to safeguard the security of sensitive information in our possession. Advances in computer capabilities,
new discoveries in the field of cryptography, inadequate facility security or other developments may result in a compromise or
breach of the technology used by us to protect customer data. Any compromise of our security, even a security breach that does
not result in a material liability could harm our reputation and therefore, our business and financial condition. In addition,
a party who is able to circumvent our security measures or exploit inadequacies in our security measures, could, among other effects,
misappropriate proprietary information, cause interruptions in our operations or expose customers and other entities with which
we interact to computer viruses or other disruptions. Actual or perceived vulnerabilities may lead to claims against us. Any insurance
coverage that we obtain to cover such risks may be insufficient to cover all claims or losses. To the extent the measures we have
taken prove to be insufficient or inadequate, we may become subject to litigation or administrative sanctions, which could result
in significant fines, penalties or damages and harm to our reputation.
We may fail to manage our growth.
We have grown significantly in a short amount
of time and intend to continue to grow in the future. However, any future growth will place additional demands on our resources,
and we cannot be sure we will be able to manage our growth effectively. If we are unable to manage our growth while expanding the
distribution of our Products and increasing profit margins, or if new systems that we implement to assist in managing our growth
do not produce the expected benefits, our business, financial position, results of operations and cash flows could be adversely
affected. We may not be able to support, financially or otherwise, future growth, or hire, train, motivate and manage the required
personnel. Our failure to manage growth effectively could also limit our ability to achieve our goals as they relate to streamlined
sales, marketing and distribution operations and the ability to achieve certain financial metrics.
We are subject to fluctuations in our results
that make it difficult to track trends and develop strategies in the short-term.
In response to competitor actions and pricing
pressures, we have engaged in significant use of promotional and sales incentives. We regularly review the results of our promotional
spending activities and adjust our promotional spending programs in an effort to maintain our competitive position as well as to
confirm compliance with our adult-focused marketing policies. Accordingly, unit sales volume and sales promotion costs in any period
are not necessarily indicative of sales and costs that may be realized in subsequent periods. Additionally, promotional activity
significantly increases net sales in the month in which it is initiated, and net sales are adversely impacted in the month after
a promotion. Accordingly, based upon the timing of our marketing and promotional initiatives, we have and may continue to experience
significant variability in our results, which could affect our ability to formulate strategies that allow us to maintain our market
presence across volatile periods. If our fluctuations obscure our ability to track important trends in our key markets, it may
have a material adverse effect on our business, results of operations and financial condition.
Adverse U.S. and global economic conditions
could negatively impact our business, prospects, results of operations, financial condition or cash flows.
Our business and operations are sensitive to
global economic conditions. These conditions include interest rates, energy costs, inflation, recession, fluctuations in debt and
equity capital markets and the general condition of the U.S. and world economy, including as a result of the effect of the COVID-19
pandemic. A material decline in the economic conditions affecting consumers, which cause a reduction in disposable income for the
average consumer, may change consumption patterns, and may result in a reduction in spending on our Product offerings or a switch
to cheaper products or products obtained through illicit channels. As such, demand for our Products may be particularly sensitive
to economic conditions such as inflation, recession, high energy costs, unemployment, changes in interest rates and money supply,
changes in the political environment, the ultimate effect on the economy of the COVID-19 pandemic and other factors beyond our
control, any combination of which could result in a material adverse effect on our business, results of operations and financial
condition.
The departure of key management personnel
and the failure to attract and retain talent could adversely affect our operations.
Our success depends upon the continued contributions
of our senior management, especially our Founder and Chief Executive Officer, Nirajkumar Patel, and our Chief Operating Officer,
Eric Mosser. If one or more of our executive officers are unable or unwilling to continue in their present positions, we may not
be able to replace them readily, if at all. Additionally, we may incur additional expenses to recruit and retain new executive
officers. If any of our executive officers join a competitor or forms a competing company, we may lose some or all of our customers.
Finally, we do not maintain “key person” life insurance on any of our executive officers. Because of these factors,
the loss of the services of any of these key persons could adversely affect our business, financial condition, and results of operations,
and thereby an investment in the Common Stock.
Our insurance may be insufficient to cover
losses that may occur as a result of our operations.
We currently maintain directors’ and
officers’ liability insurance and property and general liability insurance. This insurance or other insurance we may elect
to obtain may not be or remain available to us or be obtainable by us at commercially reasonable rates, and the amount of our coverage
may not be adequate to cover any liability we incur. Future increases in insurance costs, coupled with the increase in deductibles,
will result in higher operating costs and increased risk. If we were to incur substantial liability and such damages were not covered
by insurance or were in excess of policy limits, or if we were to incur such liability at a time when we were not able to obtain
liability insurance, our business, results of operations and financial condition could be materially adversely affected.
Risks Related to our Securities and this
Offering
Our Restated Certificate of Incorporation,
as amended (our “Certificate of Incorporation”) and our Bylaws (our “Bylaws”), as well as Delaware law
and certain regulations, could discourage or prohibit acquisition bids or merger proposals, which may adversely affect the market
price of the Common Stock.
Provisions of our Certificate of Incorporation
and Bylaws and Delaware law may discourage, delay or prevent a merger, acquisition or other change in control that stockholders
may consider favorable, including transactions in which our stockholders might otherwise receive a premium for their shares of
Common Stock. These provisions may also prevent or frustrate attempts by our stockholders to replace or remove our management.
In addition, Section 203 of the Delaware General
Corporation Law (“DGCL”) prohibits a publicly-held Delaware corporation from engaging in a business combination with
an interested stockholder, which generally refers to a person which together with its affiliates owns, or within the last three
years has owned, 15 percent or more of our voting stock, for a period of three years after the date of the transaction in which
the person became an interested stockholder, unless the business combination is approved in a prescribed manner.
The existence of the foregoing provisions and
anti-takeover measures could limit the price that investors might be willing to pay in the future for shares of Common Stock. They
could also deter potential acquirers of our company, thereby reducing the likelihood that our stockholders could receive a premium
for their Common Stock in an acquisition.
Future offerings of debt or equity securities
may rank senior to the Common Stock.
If we decide to issue debt or equity securities
in the future ranking senior to the Common Stock or otherwise incur additional indebtedness, it is possible that these securities
or indebtedness will be governed by an indenture or other instrument containing covenants restricting our operating flexibility
and limiting our ability to pay dividends to stockholders. Additionally, any convertible or exchangeable securities that we issue
in the future may have rights, preferences and privileges, including with respect to dividends, more favorable than those of the
Common Stock and may result in dilution to stockholders. Because our decision to issue debt or equity securities in any future
offering or otherwise incur indebtedness will depend on market conditions and other factors beyond our control, we cannot predict
or estimate the amount, timing or nature of our future offerings or financings, any of which could reduce the market price of the
Common Stock and dilute its value.
Raising additional capital may cause dilution
to our existing stockholders, restrict our operations or require us to relinquish rights to our technologies, if any, or Products.
We may seek additional capital through a combination
of private and public equity offerings, debt financings, strategic partnerships and alliances and licensing arrangements. To the
extent that we raise additional capital through the sale of equity or convertible debt securities, existing ownership interests
will be diluted and the terms of such financings may include liquidation or other preferences that adversely affect the rights
of existing stockholders. Debt financings may be coupled with an equity component, such as warrants to purchase shares, which could
also result in dilution of our existing stockholders’ ownership. The incurrence of indebtedness would result in increased
fixed payment obligations and could also result in certain restrictive covenants, such as limitations on our ability to incur additional
debt, limitations on our ability to acquire or license intellectual property rights and other operating restrictions that could
adversely impact our ability to conduct our business and may result in liens being placed on our assets and intellectual property.
If we were to default on such indebtedness, we could lose such assets and intellectual property. If we raise additional funds through
strategic partnerships and alliances and licensing arrangements with third parties, we may have to relinquish valuable rights to
our Products or grant licenses on terms that are not favorable to us.
We may be required to issue up to 2,000,000
shares of Common Stock in the future pursuant to a consulting agreement.
In the future, we may be required to issue
up to 2,000,000 shares of Common Stock pursuant to a consulting agreement dependent upon the resolution of a disagreement regarding
the interpretation of certain terms of our arrangements with the consultant. We are in ongoing discussions with the consultant
regarding these interpretations. We believe that once discussions are concluded the number of shares to be issued to such consultant
will be less than 2,000,000 shares; however, we cannot provide any assurances as to the ultimate resolution of this matter. If
we were required to issue 2,000,000 shares of Common Stock, our existing stockholders will be diluted. If we are unable to amicably
reach an agreement with the consultant, we or the consultant may decide to pursue litigation. Litigation is subject to significant
uncertainty and may be expensive, time-consuming, and disruptive to our operations. Any adverse resolution of this matter could
adversely affect our financial condition or the price of the Common Stock.
We may issue preferred stock whose terms
could adversely affect the voting power or value of the Common Stock.
Our Certificate of Incorporation authorizes
us to issue, without the approval of our stockholders, one or more classes or series of preferred stock having such designations,
preferences, limitations and relative rights, including preferences over the Common Stock respecting dividends and distributions,
as our Board may determine. The terms of one or more classes or series of preferred stock could adversely impact the voting power
or value of the Common Stock. For example, we might grant holders of preferred stock the right to elect some number of our directors
in all events or on the happening of specified events or the right to veto specified transactions. Similarly, the repurchase or
redemption rights or dividend or liquidation preferences we might assign to holders of preferred stock could affect the residual
value of the Common Stock.
The market prices for the Common Stock are
volatile and will fluctuate.
The market price for shares of the Common Stock
may be volatile and subject to wide fluctuations in response to numerous factors, many of which are beyond our control, including
the following: (i) actual or anticipated fluctuations in our quarterly financial results; (ii) recommendations by securities research
analysts; (iii) changes in the economic performance or market valuations of other issuers that investors deem comparable to ours;
(iv) addition or departure of our executive officers or members of our Board and other key personnel; (v) release or expiration
of lock-up or other transfer restrictions on outstanding shares of Common Stock; (vi) sales or perceived sales of additional shares
of Common Stock; (vii) liquidity of the Common Stock; (viii) significant acquisitions or business combinations, strategic partnerships,
joint ventures or capital commitments by or involving us or our competitors; and (ix) news reports relating to trends, concerns,
technological or competitive developments, regulatory changes and other related issues in our industry or target markets. Financial
markets often experience significant price and volume fluctuations that affect the market prices of equity securities of public
entities and that are, in many cases, unrelated to the operating performance, underlying asset values or prospects of such entities.
Accordingly, the market price of our shares of Common Stock may decline even if our operating results, underlying asset values
or prospects have not changed.
You may experience immediate and substantial
dilution.
If the offering price per share you pay in this offering
exceeds the net tangible book value per share of the Common Stock, you will be immediately diluted to the extent of the difference between
the amount you pay per share and the as-adjusted net tangible book value per share of the Common Stock after giving effect to this offering.
Upon the sale by us of an aggregate amount of 4,700,000 shares of Common Stock and common warrants to purchase 3,525,000 shares of Common
Stock in this offering at a public offering price of $1.70 per share, and after deducting commissions and estimated aggregate offering
expenses payable by us, you would experience immediate dilution in net tangible book value of $1.23 per share, representing the difference
between the public offering price per share and our as-adjusted net tangible book value per share after this offering. To the extent that
outstanding options are exercised, outstanding restricted stock units vest and settle, or other shares are issued, investors purchasing
the Common Stock in this offering may experience further dilution. See the section entitled “Dilution” for a more detailed
illustration of the dilution you may incur if you participate in this offering.
There can be no assurance that we will be
able to comply with the continued listing standards of Nasdaq.
We cannot assure you that we will be able to
comply with the standards that we are required to meet in order to maintain a listing of the Common Stock on Nasdaq. Nasdaq listing
rules require us to maintain certain closing bid price, stockholders’ equity and other financial metric criteria, as well
as certain corporate governance requirements, in order for the Common Stock to continue trading on Nasdaq. If we fail to comply
with the continued listing standards, the Common Stock could be delisted. A failure to maintain listing on Nasdaq could have a
material adverse effect on the liquidity and price of the Common Stock.
Future sales of shares of Common Stock by
officers and directors may negatively impact the market price for the Common Stock.
Subject to compliance with applicable securities
laws, our directors and officers and their affiliates may sell some or all of their shares of Common Stock in the future. No prediction
can be made as to the effect, if any, such future sales of shares Common Stock may have on the market price of the shares of Common
Stock prevailing from time to time. However, the future sale of a substantial number of shares of Common Stock by our directors
and officers and their affiliates, or the perception that such sales could occur, could adversely affect prevailing market prices
for our shares of Common Stock.
We have broad discretion in the use of the
net proceeds from this offering and may not use them effectively.
Our management has broad discretion in the
application of the net proceeds from this offering, and you will not have the opportunity as part of your investment decision to
assess whether the net proceeds are being used appropriately. Our management could spend the net proceeds from this offering in
ways that do not improve our results of operations or enhance the value of the Common Stock. The failure by our management to apply
these funds effectively could result in financial losses that could have a material adverse effect on our business or cause the
price of the Common Stock to decline. Pending their use, we may invest the net proceeds from this offering in a manner that does
not produce income or that loses value.
Concentration of ownership among our officers,
directors and principal stockholders may prevent other stockholders from influencing significant corporate decisions and depress
our stock price.
Based on the number of shares outstanding as
of July 31, 2021, our officers, directors and stockholders who hold at least 5% of our stock beneficially own a combined total
of approximately 77.8% percent of our outstanding Common Stock, including shares of Common Stock subject to stock options
that are currently exercisable or are exercisable and restricted stock units that vest within 60 days after July 31, 2021. If these
officers, directors, and principal stockholders or a group of our principal stockholders act together, they will be able to exert
a significant degree of influence over our management and affairs and control matters requiring stockholder approval, including
the election of directors and approval of mergers, business combinations or other significant transactions. The interests of one
or more of these stockholders may not always coincide with our interests or the interests of other stockholders. For instance,
officers, directors, and principal stockholders, acting together, could cause us to enter into transactions or agreements that
we would not otherwise consider. Similarly, this concentration of ownership may have the effect of delaying or preventing a change
in control of our company otherwise favored by our other stockholders. This, in turn, could have a negative effect on the market
price of the Common Stock. It could also prevent our stockholders from realizing a premium over the market price for their shares
of Common Stock. The concentration of ownership also may contribute to the low trading volume and volatility of the Common Stock.
The Common Stock may become the target of
a “short squeeze.”
In 2021, the securities of several companies
have increasingly experienced significant and extreme volatility in stock price due to short sellers of shares of the Common Stock
and buy-and-hold decisions of longer investors, resulting in what is sometimes described as a “short squeeze.” Short
squeezes have caused extreme volatility in those companies and in the market and have led to the price per share of those companies
to trade at a significantly inflated rate that is disconnected from the underlying value of the company. Sharp rises in a company’s
stock price may force traders in a short position to buy the stock to avoid even greater losses. Many investors who have purchased
shares in those companies at an inflated rate face the risk of losing a significant portion of their original investment as the
price per share has declined steadily as interest in those stocks have abated. We may be a target of a short squeeze, and investors
may lose a significant portion or all of their investment if they purchase our shares at a rate that is significantly disconnected
from our underlying value.
There is no public market for the common
warrants.
There is no established public trading market
for the common warrants, and we do not expect a market to develop. In addition, we do not intend to apply to list the common warrants
on any national securities exchange or other nationally recognized trading system, including Nasdaq. Without an active market,
the liquidity of the common warrants will be limited.
The common warrants are speculative in nature.
The common warrants do not confer any rights
of Common Stock ownership on its holders, such as voting rights or the right to receive dividends, but rather merely represent
the right to acquire shares of Common Stock at a fixed price, as the case maybe. In addition, following this offering, the market
value of the common warrants, if any, is uncertain and there can be no assurance that the market value of the common warrants will
equal or exceed their imputed offering price. The common warrants will not be listed or quoted for trading on any market or exchange.
Holders of the common warrants will not
have rights of holders of Common Stock until such warrants are exercised.
Until holders of common warrants acquire shares
of the Common Stock upon exercise of the common warrants, holders of common warrants will have no rights with respect to the shares
of the Common Stock underlying such securities. Upon exercise of the common warrants, the holders will be entitled to exercise
the rights of a holder of the Common Stock only as to matters for which the record date occurs after the exercise.
If securities or industry analysts fail
to continue publishing research about our business, if they change their recommendations adversely or if our results of operations
do not meet their expectations, our stock price and trading volume could decline.
The trading market for the Common Stock will
be influenced by the research and reports that industry or securities analysts publish about us or our business. If one or more
of these analysts cease coverage of our company or fail to publish reports on us regularly, we could lose visibility in the financial
markets, which in turn could cause our stock price or trading volume to decline. In addition, it is likely that in some future
period our operating results will be below the expectations of securities analysts or investors. If one or more of the analysts
who cover us downgrade our stock, or if our results of operations do not meet their expectations, our stock price could decline.
We do not currently pay dividends on our
shares of Common Stock and have no intention to pay dividends on our shares of Common Stock for the foreseeable future.
No dividends on our shares of Common Stock
have been paid by us to date. We do not intend to declare or pay any cash dividends in the foreseeable future. Payment of any future
dividends will be at the discretion of our Board, after taking into account a multitude of factors appropriate in the circumstances,
including our operating results, financial condition and current and anticipated cash needs. In addition, the terms of any future
debt or credit facility may preclude us from paying any dividends unless certain consents are obtained and certain conditions are
met. There is no assurance that future dividends will be paid, and, if dividends are paid, there is no assurance with respect to
the amount of any such dividend. Unless our Board determines to pay dividends, our stockholders will be required to look to appreciation
of the Common Stock to realize a gain on their investment. There can be no assurance that this appreciation will occur.
For as long as we are an “emerging
growth company” we intend to take advantage of reduced disclosure and governance requirements applicable to emerging growth
companies, which could result in the Common Stock being less attractive to investors and could make it more difficult for us to
raise capital as and when we need it.
We are an “emerging growth company,”
as defined in the JOBS Act, and we have taken advantage, and intend to continue to take advantage, of certain exemptions from various
reporting requirements that are applicable to other public companies that are not emerging growth companies including, but not
limited to, not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act, reduced
disclosure obligations regarding executive compensation in our periodic reports and proxy statements, and exemptions from the requirements
of holding a non-binding advisory vote on executive compensation and stockholder approval of any golden parachute payments not
previously approved.
Investors may find the Common Stock less attractive
because we rely on these exemptions, which could contribute to a less active trading market for the Common Stock or volatility
in our share price. In addition, we may be less attractive to investors and it may be difficult for us to raise additional capital
as and when we need it. Investors may be unable to compare our business with other companies in our industry if they believe that
our financial accounting is not as transparent as other companies in our industry. If we are unable to raise additional capital
as and when we need it, our financial condition and results of operations may be materially and adversely affected.
We may take advantage of these reporting exemptions
until we are no longer an emerging growth company.
We have previously identified material weaknesses
in our system of internal control over financial reporting, and if we cannot remediate these materials weaknesses, we may not be
able to accurately report our financial condition, results of operations or cash flows, which may adversely affect investor confidence
in us and, as a result, the value of the Common Stock.
A material weakness is a deficiency, or combination
of deficiencies, in internal control over financial reporting that results in more than a reasonable possibility that a material
misstatement of annual or interim financial statements will not be prevented or detected on a timely basis. Section 404 of the
Sarbanes-Oxley Act of 2002 (“Sarbanes-Oxley”) also generally requires an attestation from our independent registered
public accounting firm on the effectiveness of our internal control over financial reporting. However, for as long as we remain
an emerging growth company as defined in the JOBS Act, we intend to take advantage of the exemption permitting us not to comply
with the independent registered public accounting firm attestation requirement.
Our management has previously identified, and
we have disclosed, material weaknesses in our internal control over financial reporting. Specifically, our management has found
that our internal control over financial reporting was ineffective as of October 31, 2020 based on a determination that there was
a lack of resources to provide segregation of duties consistent with control objectives, the lack (at that time) of a formal audit
committee, and the lack of a formal review process that includes multiple levels of review over financial disclosure and reporting
processes, management has determined that material weaknesses existed as of October 31, 2020.
To address these material weaknesses, and subject
to the receipt of additional financing or cash flows, we have undertaken, and intend to continue to undertake, remediation measures
to address such material weaknesses, including implementing procedures pursuant to which we can ensure segregation of duties and
hire additional resources to ensure appropriate review and oversight.
Our compliance with Section 404 of Sarbanes-Oxley
will require that we incur substantial accounting expense and expend significant management efforts. We may not be able to complete
our evaluation, testing and any required remediation in a timely fashion. During the evaluation and testing process, if we identify
one or more material weaknesses in our internal control over financial reporting, we will be unable to assert that our internal
control over financial reporting is effective. We cannot assure you that there will not be material weaknesses or significant deficiencies
in our internal control over financial reporting in the future. Any failure to maintain internal control over financial reporting
could severely inhibit our ability to accurately report our financial condition, results of operations or cash flows. This may
expose us, including individual executives, to potential liability which could significantly affect our business.
We cannot assure you that we will, in the future,
identify areas requiring improvement in our internal control over financial reporting. We cannot assure you that the measures we
will take to remediate any areas in need of improvement will be successful or that we will implement and maintain adequate controls
over our financial process and reporting in the future as we continue to grow. If we are unable to establish appropriate internal
financial reporting controls and procedures, if we are unable to conclude that our internal control over financial reporting is
effective, or if our independent registered public accounting firm determines we have a material weakness or significant deficiency
in our internal control over financial reporting once that firm begins its audits of internal control over financial reporting,
it could cause us to fail to meet our reporting obligations, result in the restatement of our financial statements, harm our operating
results, cause investors to lose confidence in the accuracy and completeness of our financial reports, the market price of our
common shares could decline, and we could be subject to sanctions or investigations by Nasdaq, the SEC, or other regulatory authorities.
Failure to remedy any material weakness in our internal control over financial reporting, or to implement or maintain other effective
control systems required of public companies, could also restrict our future access to the capital markets.
Our disclosure controls and procedures may
not prevent or detect all errors or acts of fraud.
Our disclosure controls and procedures are
designed to reasonably assure that information required to be disclosed by us in reports we file or submit under the Securities
Exchange Act of 1934, as amended (the “Exchange Act”) is accumulated and communicated to management, recorded, processed,
summarized and reported within the time periods specified in the rules and forms of the SEC. We believe that any disclosure controls
and procedures or internal controls and procedures, no matter how well conceived and operated, can provide only reasonable, not
absolute, assurance that the objectives of the control system are met. As of October 31, 2020, our Chief Executive Officer concluded
that the disclosure controls and procedures were not effective as of such date due to material weaknesses in internal controls
identified above.
These inherent limitations include the realities
that judgments in decision-making can be faulty, and that breakdowns can occur because of simple error or mistake. Additionally,
controls can be circumvented by the individual acts of some persons, by collusion of two or more people or by an unauthorized override
of the controls. Accordingly, because of the inherent limitations in our control system, misstatements or insufficient disclosures
due to error or fraud may occur and not be detected.
We have incurred, and will continue to incur,
increased costs as a result of operating as a public company, and our management has been required, and will continue to be required,
to devote substantial time to new compliance initiatives.
As a public company, we have incurred and are
continuing to incur significant legal, accounting and other expenses and these expenses may increase even more after we are no
longer an “emerging growth company” and “smaller reporting company.” We are subject to the reporting requirements
of the Exchange Act and the rules adopted, and to be adopted, by the SEC. Our management and other personnel devote a substantial
amount of time to these compliance initiatives.
Moreover, these rules and regulations have
substantially increased our legal and financial compliance costs and made some activities more time-consuming and costly. The increased
costs can result in our reporting a net loss. These rules and regulations may make it more difficult and more expensive for us
to maintain sufficient director’s and officer’s liability insurance coverage. We cannot predict or estimate the amount
or timing of additional costs we may continue to incur to respond to these requirements. The ongoing impact of these requirements
could also make it more difficult for us to attract and retain qualified persons to serve on our Board, our Board committees, or
as executive officers.
USE OF PROCEEDS
We expect to receive net proceeds of approximately
$7,031,200 from this offering, after deducting the underwriting fees and discounts and estimated offering expenses payable by us, and
assuming no exercise of the common warrants offered hereby. In the event that the common warrants offered hereby are exercised in full,
the Company would receive an additional $6,697,500 in proceeds.
We intend to use the net proceeds from this
offering for general corporate purposes, including working capital. Our expected use of the net proceeds from this offering represents
our current intentions based upon our present plans and business condition, which could change in the future as our plans and business
conditions evolve. As of the date of this prospectus supplement, we cannot specify with certainty all of the particular uses of
the proceeds from this offering. Accordingly, we will retain broad discretion of the use of such proceeds.
CAPITALIZATION
The following table sets forth our capitalization
as of July 31, 2021 on an actual basis and as adjusted to give effect to the issuance and sale of 4,700,000 shares of Common
Stock and accompanying 3,525,000 common warrants at the offering price of $1.70 per share and 0.75 of an accompanying common warrant,
after deducting underwriting discounts and commissions and estimated offering expenses payable by us. The information set forth in the
table below is only a summary and is qualified in its entirety by, and should be read in conjunction with, the consolidated financial
statements, the notes thereto, and “Management’s Discussion and Analysis of Results of Operations and Financial Condition”
in our Annual Report on Form 10-K for the fiscal year ended October 31, 2020 and our Quarterly Reports on Form 10-Q for the periods ending
January 31, 2021, April 30, 2021 and July 31, 2021, each of which is incorporated by reference in this prospectus supplement and the accompanying
prospectus. See “Where You Can Find Additional Information” in this prospectus supplement.
|
|
|
July 31, 2021
|
|
|
|
Actual
|
|
As Adjusted
|
|
Cash
|
|
$
|
938,435
|
|
|
$
|
7,969,635
|
|
|
Stockholders’ Equity
|
|
|
|
|
|
|
|
|
|
Preferred stock, $0.001 par value, 5,000,000 shares
authorized; Series A Convertible Preferred stock, 3,000,000 shares authorized, 3,000,000 issued and outstanding, actual and
as adjusted
|
|
|
3,000
|
|
|
|
3,000
|
|
|
Common stock, $0.001 par value, 1,000,000,000 shares
authorized, 23,600,597 shares issued and outstanding, actual and 28,300,597 shares outstanding, as adjusted
|
|
|
23,600
|
|
|
|
28,300
|
|
|
Additional paid-in-capital
|
|
|
9,954,779
|
|
|
|
17,940,079
|
|
|
Retained deficit
|
|
|
(3,611,835
|
)
|
|
|
(4,570,635
|
)
|
|
Total Stockholders’ Equity
|
|
|
6,369,544
|
|
|
|
13,400,744
|
|
|
Total Capitalization
|
|
$
|
23,898,462
|
|
|
$
|
30,929,662
|
|
The table above excludes:
|
|
●
|
150,000 shares of Common Stock issuable upon exercise of options to purchase the Common Stock outstanding as of July 31, 2021 at a weighted average exercise price of $20.49 per share;
|
|
|
|
|
|
|
●
|
555,417 shares of Common Stock issuable upon settlement of restricted
stock units outstanding as of July 31, 2021;
|
|
|
|
|
|
|
●
|
7,495,833 shares of Common Stock reserved as of July 31, 2021 for future issuance under our 2020 Stock Option and Incentive Plan;
|
|
|
|
|
|
|
●
|
25,000,000 shares of Common Stock issuable upon exercise of convertible preferred stock outstanding as of July 31, 2021;
|
|
|
|
|
|
|
●
|
Up to 2,000,000 shares of Common Stock that may be issuable in the future to a consultant dependent upon the resolution of a disagreement between the consultant and us with respect to the terms of our arrangement; and
|
|
|
|
|
|
|
●
|
3,525,000 shares of Common Stock issuable upon the common warrants offered hereby (or 4,053,750
shares of Common Stock issuable upon the common warrants if the underwriter exercises its over-allotment option in full).
|
In addition, the amounts in the table above assume no exercise of
the underwriter’s over-allotment option.
DILUTION
If you invest in the Common Stock and the common
warrants, your interest will be diluted to the extent of the difference between the public offering price per share you pay in
this offering and the net tangible book value per share of the Common Stock immediately after this offering.
Net tangible book value per share is determined by
subtracting our total liabilities from our total tangible assets, which is total assets less intangible assets, and dividing this amount
by the number of shares of Common Stock outstanding. The historical net tangible book value of the Common Stock, as of July 31, 2021,
was approximately $6.37 million, or $0.27 per share, based on 23,600,597 shares of Common Stock outstanding as of July 31, 2021 as reported
in our Quarterly Report on Form 10-Q for the fiscal quarter ended July 31, 2021. Dilution in net tangible book value per share represents
the difference between the amount per share paid by purchasers of shares of Common Stock in this offering and the net tangible book value
per share of the Common Stock immediately after this offering.
After giving effect to the sale of shares
of Common Stock in this offering at an offering price of $1.70 per share, and after deducting estimated commissions and offering
expenses payable by us, our as adjusted net tangible book value at July 31, 2021 would have been approximately $13.4 million,
or $0.47 per share. This represents an immediate dilution of $0.20 per share to new investors purchasing shares of Common Stock
in this offering. The following table illustrates this dilution:
|
Offering price per share
|
|
$
|
1.70
|
|
|
|
|
|
|
|
|
Net tangible book value per share as of July 31, 2021
|
|
$
|
0.27
|
|
|
|
|
|
|
|
|
Increase in net tangible book value per share attributable
to investors in this offering
|
|
$
|
0.20
|
|
|
|
|
|
|
|
|
As adjusted net tangible book value per share after giving
effect to this offering
|
|
$
|
0.47
|
|
|
|
|
|
|
|
|
As adjusted dilution per share to investors in this offering
|
|
$
|
1.23
|
|
If the underwriter exercises its option
to purchase up to 705,000 additional shares at a purchase price of $1.5717 per share and/or up to an additional 528,750 common
warrants at a purchase price of $$0.01239969 per common warrant in full, the adjusted net tangible book value will increase to
$0.49 per share, representing an immediate increase in net tangible book value of $0.02 per share to existing stockholders and
immediate dilution of $1.21 per share to investors purchasing shares of the Common Stock in this offering.
The above illustration of dilution per share
to investors participating in this offering excludes:
|
|
●
|
150,000 shares of Common Stock issuable upon exercise of options to purchase the Common Stock outstanding as of July 31, 2021 at a weighted average exercise price of $20.49 per share;
|
|
|
|
|
|
|
●
|
555,417 shares of Common Stock issuable upon settlement of restricted
stock units outstanding as of July 31, 2021;
|
|
|
|
|
|
|
●
|
7,495,833 shares of Common Stock reserved as of July 31, 2021 for future issuance under our 2020 Stock Option and Incentive Plan;
|
|
|
|
|
|
|
●
|
25,000,000 shares of Common Stock issuable upon exercise of convertible preferred stock outstanding as of July 31, 2021;
|
|
|
|
|
|
|
●
|
Up to 2,000,000 shares of Common Stock that may be issuable in the future to a consultant dependent upon the resolution of a disagreement between the consultant and us with respect to the terms of our arrangement; and
|
|
|
|
|
|
|
●
|
3,525,000 shares of Common Stock issuable upon the common warrants offered hereby (or
4,053,750 shares of Common Stock issuable upon the common warrants if the underwriter exercises its over-allotment option
in full)
|
To the extent that any of these outstanding
options are exercised or we issue additional shares under our equity incentive plans, there will be further dilution to new investors.
In addition, we may choose to raise additional capital due to market conditions or strategic considerations even if we believe
we have sufficient funds for our current or future operating plans. To the extent that additional capital is raised through the
sale of equity or convertible debt securities, the issuance of these securities could result in further dilution to our stockholders.
DESCRIPTION OF OUR SECURITIES WE ARE OFFERING
We are offering shares of the Common Stock and common warrants pursuant
to this prospectus supplement and the accompanying prospectus. The material terms and provisions of the Common Stock are described
under the caption “Description of Capital Stock” below in this prospectus supplement and in the accompanying prospectus.
Common Stock
See “Description of Capital Stock” below in this prospectus
supplement and in the accompanying prospectus.
Common Warrants
The common warrants to be issued as a part of this offering will
be separately transferable following their issuance and through their expiration five years from the date of issuance. Each common
warrant will entitle the holder to purchase one share of the Common Stock at an exercise price of $1.90 per share from the date of
issuance through its expiration.
Each common warrant will be issued under a separate warrant agreement
to be entered into between us and Vstock Transfer, LLC, as warrant agent. The warrant agent will act solely as our agent in connection
with the common warrants and will not have any obligation or relationship of agency or trust for or with any holders or beneficial
owners of common warrants.
There is no public trading market for the common warrants and we
do not intend that they will be listed for trading on Nasdaq or any other securities exchange or market. The Common Stock underlying
the common warrants, upon issuance, will also be traded on Nasdaq under the symbol “KAVL.”
Exercisability
Each common warrant will be exercisable at any time and will expire
five years from the date of issuance. The common warrants will be exercisable, at the option of each holder, in whole or in part
by delivering to us a duly executed exercise notice and payment in full for the number of shares of the Common Stock purchased
upon such exercise, except in the case of a cashless exercise as discussed below. Each common warrant entitles the holder thereof
to purchase one share of Common Stock. Common warrants are not exercisable for a fraction of a share and may only be exercised
into whole numbers of shares. In lieu of fractional shares, we will, pay the holder an amount in cash equal to the fractional amount
multiplied by the exercise price and round down to the nearest whole share. Unless otherwise specified in the common warrant, the
holder will not have the right to exercise the common warrant, in whole or in part, if the holder (together with its affiliates)
would beneficially own in excess of 4.99% (or 9.99% at the holder’s election) of the number of our shares of Common Stock
outstanding immediately after giving effect to the exercise, as such percentage is determined in accordance with the terms of the
common warrant. However, any holder may increase or decrease such percentage to any other percentage not in excess of 9.99% upon
at least 61 days’ prior notice from the holder to us.
Cashless Exercise
If at any time there is no effective registration statement registering,
or the prospectus contained therein is not available for issuance of, the shares issuable upon exercise of the common warrants,
the holder may exercise the common warrants on a cashless basis. When exercised on a cashless basis, a portion of the common warrant
is cancelled in payment of the purchase price payable in respect of the number of shares of the Common Stock purchasable upon such
exercise.
Exercise Price
Each common warrant represents the right to purchase one share of
Common Stock at an exercise price of $1.90 per share. In addition, the exercise price per share is subject to adjustment for stock
dividends, distributions, subdivisions, combinations, or reclassifications, and for certain dilutive issuances.
Adjustments; Fundamental Transaction
The exercise price and the number of shares underlying the common
warrants are subject to appropriate adjustment in the event of stock splits, stock dividends on our shares of Common Stock, stock
combinations or similar events affecting our shares of Common Stock. In addition, in the event we consummate a merger or consolidation
with or into another person or other reorganization event in which our shares of Common Stock are converted or exchanged for securities,
cash or other property, or we sell, lease, license, assign, transfer, convey or otherwise dispose of all or substantially all of
our assets or we or another person acquire 50% or more of our outstanding shares of Common Stock (each, a Fundamental Transaction),
then following such Fundamental Transaction the holders of the common warrants will be entitled to receive upon exercise of the
common warrants the same kind and amount of securities, cash or property which the holders would have received had they exercised
the common warrants immediately prior to such Fundamental Transaction. Any successor to us or surviving entity will assume the
obligations under the common warrants.
Transferability
Subject to applicable laws and restrictions, a holder may transfer
a common warrant upon surrender of the common warrant to us with a completed and signed assignment in the form attached to the
common warrant. The transferring holder will be responsible for any tax that liability that may arise as a result of the transfer.
No Market
There is no public trading market for the common warrants and we
do not intend that they will be listed for trading on Nasdaq or any other securities exchange or market.
Rights as Stockholder
Except as set forth in the common warrant, the holder of a common
warrant, solely in such holder’s capacity as a holder of a common warrant, will not be entitled to vote, to receive dividends,
or to any of the other rights of our stockholders.
Amendments and Waivers
The provisions of each common warrant may be modified or amended
or the provisions thereof waived with the written consent of us and the holder.
Warrant Agent; Global Certificate
The common warrants will be issued pursuant to a warrant agency
agreement by and between us and Vstock Transfer, LLC, the warrant agent. The common warrants will initially be represented only
by one or more global warrants deposited with the warrant agent, as custodian on behalf of The Depository Trust Company, or DTC,
and registered in the name of Cede & Co., a nominee of DTC, or as otherwise directed by DTC.
DESCRIPTION OF CAPITAL STOCK
The following summary of our capital stock
is based on certain provisions of our Certificate of Incorporation and Bylaws and on the applicable provisions of the DGCL. This
summary does not purport to be complete and is qualified in its entirety by reference to the applicable provisions our Certificate
of Incorporation and Bylaws and the DGCL. For information on how to obtain copies of such documents, please refer to the heading
“Where You Can Find More Information” in this prospectus supplement.
General
Our authorized capital stock consists of 1,005,000,000
shares, divided into 1,000,000,000 shares of the Common Stock, and 5,000,000 shares of preferred stock, par value $0.001 per share
(“Preferred Stock”). Under our Certificate of Incorporation, our Board has the authority to issue such shares of Common
Stock and Preferred Stock in one or more classes or series, with such voting powers, designations, preferences and relative, participating,
optional or other special rights, if any, and such qualifications, limitations or restrictions thereof, if any, as shall be provided
for in a resolution or resolutions adopted by our Board and filed as designations.
Common Stock
As of July 31, 2021, 23,600,597 shares of the
Common Stock were outstanding.
Holders of the Common Stock are entitled to
one vote for each share held of record on all matters submitted to a vote of stockholders, including the election of directors,
and are entitled to receive dividends when and as declared by our Board out of funds legally available therefore for distribution
to stockholders and to share ratably in the assets legally available for distribution to stockholders in the event of the liquidation
or dissolution, whether voluntary or involuntary, of the Company. We have not paid any dividends and do not anticipate paying any
dividends on the Common Stock in the foreseeable future. It is our present policy to retain earnings, if any, for use in the development
of our business. The Common Stockholders do not have cumulative voting rights in the election of directors and have no preemptive,
subscription, or conversion rights. The Common Stock is not subject to redemption by us.
The transfer agent and registrar for the Common
Stock is Vstock Transfer, LLC
Preferred Stock
Of the 5,000,000 shares of Preferred Stock
authorized, our Board has previously designated 3,000,000 shares of Preferred Stock as Series A Convertible Preferred Stock (the
“Series A Preferred Stock”), of which 3,000,000 shares remain outstanding.
Of the 5,000,000 shares of Preferred Stock,
2,000,000 shares of our Preferred Stock remain available for designation by our Board. Accordingly, our Board is empowered, without
stockholder approval, to issue Preferred Stock with dividend, liquidation, conversion, voting or other rights that could adversely
affect the voting power or other rights of the holders of Common Stock. The issuance of Preferred Stock could have the effect of
restricting dividends on the Common Stock, diluting the voting power of the Common Stock, impairing the liquidation rights of the
Common Stock, or delaying or preventing a change in control of us, all without further action by our stockholders.
Series A Convertible Preferred Stock
The Series A Convertible Preferred Stock have
the following terms:
Liquidation Preference. If we liquidate,
dissolve, or wind up, holders of the Series A Preferred Stock will have the right to receive an amount equal to $1.00 in the aggregate
for all issued and outstanding shares of Series A Preferred Stock (as adjusted for any stock dividends, combinations, splits, recapitalizations,
and the like with respect to such shares) (the “Preference Value”). After the payment of the full applicable Preference
Value of the then issued and outstanding shares of the Series A Preferred Stock, our remaining assets legally available for distribution,
if any, will be distributed ratably to the holders of the Common Stock.
Dividends. The holders of the Series
A Preferred Stock do not have any preferential dividend rights and are entitled to receive dividends, if any, only if, when, and
as declared by our Board in its sole and absolute discretion.
Voting Rights. The holders have the
Series A Preferred Stock do not have any voting rights.
Conversion Rights. Each share of Series
A Preferred Stock was originally convertible into 100 shares of Common Stock; however, as a result of the one-for-twelve reverse
stock split effected on July 20, 2021, each share of Series A Preferred Stock is now convertible into approximately 8.33 shares
of Common Stock. The holders of the Series A Preferred Stock may convert their Series A Preferred Stock at any time on or after
November 1, 2023. Notwithstanding the foregoing, the holders of the Series A Preferred Stock may convert their shares of Series
A Preferred Stock prior to November 1, 2023 if a change of control (as provided for in the Certificate of Designation of Preferences,
Rights, and Limitations of the Series A Preferred Stock) or upon the occurrence of any other event as determined and agreed to
by us and the holders holding a majority of the issued and outstanding shares of Series A Preferred Stock. The shares of Common
Stock to be issued upon conversion will bear a restricted legend.
Ranking. All series of preferred stock,
whether now or hereafter designated, may by their respective terms have a preference over the Series A Preferred Stock in respect
of distribution upon liquidation, dividends, or any other right or matter.
Certain Provisions of our Certificate of
Incorporation, our Bylaws, and the DGCL
Certain provisions in our Certificate of Incorporation
and Bylaws, as well as certain provisions of the DGCL, may be deemed to have an anti-takeover effect and may delay, deter, or prevent
a tender offer or takeover attempt that a stockholder might consider to be in its best interests, including attempts that might
result in a premium being paid over the market price of the shares held by stockholders. These provisions contained in our Certificate
of Incorporation and Bylaws include the items described below.
|
|
●
|
Special Meetings of Stockholders.
Our Bylaws provide that special meetings of our stockholders may be called only by a majority of our Board, the President, Chief
Executive Officer, or the Secretary.
|
|
|
|
|
|
|
●
|
No Cumulative Voting. Our Certificate
of Incorporation does not include a provision for cumulative voting for directors. Under cumulative voting, a minority stockholder
holding a sufficient percentage of a class of shares could be able to ensure the election of one or more directors.
|
|
|
|
|
|
|
●
|
Undesignated Preferred Stock. Because
our Board has the power to establish the preferences and rights of the shares of any additional series of Preferred Stock, it
may afford holders of any Preferred Stock preferences, powers, and rights, including voting and dividend rights, senior to the
rights of holders of the Common Stock, which could adversely affect the holders of Common Stock and could discourage a takeover
of us even if a change of control of the Company would be beneficial to the interests of our stockholders.
|
|
|
|
|
|
|
●
|
Our Officers Beneficially Own a Majority
of Our Capital Stock. Our Chief Executive Officer and Chief Operating Officer, both of whom serve as members of the Board,
beneficially more than a majority of the Common Stock and own all of the issued and outstanding shares of Series A Preferred Stock.
Accordingly, they are able to control all matters related to the Company.
|
These and other provisions contained in our
Certificate of Incorporation and Bylaws are expected to discourage coercive takeover practices and inadequate takeover bids. These
provisions are also designed to encourage persons seeking to acquire control of us to first negotiate with our Board. However,
these provisions could delay or discourage transactions involving an actual or potential change in control of us, including transactions
in which stockholders might otherwise receive a premium for their shares over then current prices. Such provisions could also limit
the ability of stockholders to remove current management or approve transactions that stockholders may deem to be in their best
interests.
In addition, we are subject to the provisions
of Section 203 of the DGCL. Section 203 of the DGCL prohibits a publicly-held Delaware corporation from engaging in a “business
combination” with an “interested stockholder” for a period of three years after the person became an interested
stockholder, unless:
|
|
●
|
The board of directors of the corporation
approved the business combination or other transaction in which the person became an interested stockholder prior to the date
of the business combination or other transaction;
|
|
|
|
|
|
|
●
|
Upon consummation of the transaction that
resulted in the person becoming an interested stockholder, the person owned at least 85% of the voting stock of the corporation
outstanding at the time the transaction commenced, excluding for purposes of determining the number of shares outstanding, shares
owned by persons who are directors and also officers of the corporation and shares issued under which employee participants do
not have the right to determine confidentially whether shares held subject to the plan will be tendered in a tender or exchange
offer; or
|
|
|
|
|
|
|
●
|
on or subsequent to the date the person
became an interested stockholder, the board of directors of the corporation approved the business combination and the stockholders
of the corporation authorized the business combination at an annual or special meeting of stockholders by the affirmative vote
of at least 66-2/3% of the outstanding voting stock of the corporation that is not owned by the interested stockholder.
|
A “business combination” includes
mergers, asset sales, and other transactions resulting in a financial benefit to the interested stockholder. Subject to certain
exceptions, an “interested stockholder” is a person who, together with affiliates and associates, owns, or within the
prior three years did own, 15% or more of a corporation’s voting stock.
Section 203 of the DGCL could depress our stock
price and delay, discourage, or prohibit transactions not approved in advance by our Board, such as takeover attempts that might
otherwise involve the payment to our stockholders of a premium over the market price of the Common Stock.
MATERIAL UNITED STATES FEDERAL TAX CONSEQUENCES
FOR NON-U.S. HOLDERS
The following discussion is a summary of the
material U.S. federal tax consequences relating to the acquisition, ownership and disposition of the Common Stock and accompanying
common warrants by non-U.S. holders (as defined below). This discussion is based upon the provisions of the U.S. Internal Revenue
Code of 1986, as amended (the “Code”), U.S. Treasury regulations, rulings and judicial decisions, all as in effect
on the date hereof. Those authorities may be changed, perhaps retroactively, so as to result in U.S. federal income and estate
tax consequences different from those discussed below. There can be no assurance that the U.S. Internal Revenue Service (the “IRS”),
will agree with the statements herein.
A “U.S. holder” means a beneficial
owner of the Common Stock or common warrants that is for U.S. federal income tax purposes:
|
|
●
|
a citizen or individual resident of the United States;
|
|
|
|
|
|
|
●
|
a corporation (or other entity treated as a corporation for U.S. federal income tax purposes) created or organized in or under the laws of the United States, any state thereof or the District of Columbia;
|
|
|
|
|
|
|
●
|
an estate the income of which is subject to U.S. federal income taxation regardless of its source; or
|
|
|
|
|
|
|
●
|
a trust, if (1) a court within the United States is able to exercise primary supervision over the trust’s administration and one or more U.S. persons have the authority to control all of its substantial decisions, or (2) a valid election to be treated as a U.S. person is in effect under the relevant Treasury regulations with respect to such trust.
|
A “non-U.S. holder”
means a beneficial owner of the Common Stock or common warrants that is neither a U.S. holder nor a partnership (including an
entity that is treated as a partnership for U.S. federal income tax purposes).
This discussion deals only with the Common
Stock and accompanying common warrants held as a capital asset within the meaning of Section 1221 of the Code (generally, property
held for investment). This discussion does not address all of the U.S. federal income and estate tax consequences that may be relevant
to a non-U.S. holder in light of such holder’s particular circumstances, nor does it deal with special situations, such as:
|
|
●
|
tax consequences to non-U.S. holders who may be subject to special tax treatment, such as banks and other financial institutions, insurance companies, partnerships or other entities treated as pass-through entities for U.S. federal income tax purposes, certain former citizens or residents of the United States, “controlled foreign corporations”, “passive foreign investment companies”, corporations that accumulate earnings to avoid U.S. federal income tax, tax-exempt entities, common trust funds, certain trusts, hybrid entities, foreign governments, international organizations and dealers or traders in securities that elect to use a mark-to-market method of accounting for their securities holdings;
|
|
|
|
|
|
|
●
|
tax consequences to persons holding the Common Stock or common warrants as part of a hedging, integrated, constructive sale or conversion transaction or a straddle;
|
|
|
|
|
|
|
●
|
any gift tax consequences;
|
|
|
|
|
|
|
●
|
alternative minimum tax consequences, if any; or
|
|
|
|
|
|
|
●
|
any U.S. state or local or foreign tax consequences.
|
If an entity treated as a partnership
for U.S. federal income tax purposes holds the Common Stock or common warrants, the tax treatment of a partner or member in the
partnership generally will depend upon the status of the partner or member and the activities of the partnership. Prospective
investors that are entities treated as partnerships for U.S. federal income tax purposes should consult their own tax advisors
regarding the U.S. federal income and estate tax considerations to them and their partners or members of holding the Common Stock
or common warrants.
THIS DISCUSSION IS NOT A LEGAL OPINION. IF
YOU ARE CONSIDERING THE ACQUISITION OF THE COMMON STOCK AND ACCOMPANYING COMMON WARRANTS, YOU SHOULD CONSULT YOUR OWN TAX ADVISOR
CONCERNING THE U.S. FEDERAL INCOME TAX CONSEQUENCES TO YOU IN LIGHT OF YOUR OWN PARTICULAR CIRCUMSTANCES, AS WELL AS ANY TAX CONSEQUENCES
ARISING UNDER THE LAWS OF ANY OTHER TAXING JURISDICTION, THE EFFECT OF ANY CHANGES IN APPLICABLE TAX LAW, AND YOUR ENTITLEMENT
TO BENEFITS UNDER AN APPLICABLE INCOME TAX TREATY.
Allocation of Purchase Price Between Common
Stock and Accompanying Common Warrants
The acquisition of each share of Common Stock
and accompanying common warrant offered pursuant to this offering should be treated for U.S. federal income tax purposes as the
acquisition of one share of Common Stock and one common warrant to purchase one share of Common Stock. For U.S. federal income
tax purposes, each holder must allocate the purchase price paid by such holder between such one share of Common Stock and one common
warrant based on their relative fair market values at the time of issuance. Under U.S. federal income tax law, the investors must
make their own determination of value for these purposes. The price allocated to each share of Common Stock and each common warrant
should be the holder’s basis in such Common Stock or common warrant, as the case may be. Any disposition of a share of Common
Stock and accompanying common warrant should be treated for U.S. federal income tax purposes as a disposition of the one share
of Common Stock and one common warrant to purchase one share of Common Stock, and the amount realized on the disposition should
be allocated between the one share of Common Stock and accompanying common warrant based on their respective relative fair market
values as determined by the holder based on all of the relevant facts and circumstances at the time of disposition. The separation
of the Common Stock and accompanying common warrants should not be a taxable event for U.S. federal income tax purposes.
The foregoing treatment of the Common Stock
and accompanying common warrants and a holder’s purchase price allocation are not binding on the IRS or courts. Because there
are no authorities that directly address instruments that are similar to the Common Stock and accompanying common warrants, no
assurance can be given that the IRS or the courts will agree with the characterization described above or the discussion below.
Accordingly, each prospective investor is urged to consult its own tax advisors regarding the tax consequences of an investment
in the Common Stock and accompanying common warrants. The balance of this discussion assumes that the characterization of the Common
Stock and accompanying common warrants described above is respected for U.S. federal income tax purposes.
Distributions on Common Stock
We do not expect to declare or make any distributions
on the Common Stock in the foreseeable future. If we make a distribution of cash or other property (other than certain pro rata
distributions of the Common Stock) in respect of the Common Stock, the distribution will be treated as a dividend to the extent
it is paid from our current or accumulated earnings and profits (as determined under U.S. federal income tax principles). If the
amount of a distribution exceeds our current and accumulated earnings and profits, such excess first will be treated as a tax-free
return of capital to the extent of the non-U.S. holder’s adjusted tax basis in the Common Stock, and thereafter will be treated
as capital gain. Subject to the discussion of backup withholding and FATCA below, distributions treated as dividends on the Common
Stock held by a non-U.S. holder generally will be subject to U.S. federal withholding tax at a rate of 30%, or at a lower rate
if provided by an applicable income tax treaty and the non-U.S. holder has provided the documentation required to claim benefits
under such treaty. Generally, to claim the benefits of an income tax treaty, a non-U.S. holder will be required to provide a properly
executed IRS Form W-8BEN or W-8BEN-E (or appropriate substitute or successor form) certifying its entitlement to benefits under
the treaty.
If, however, a dividend is effectively connected
with the conduct of a trade or business in the United States by the non-U.S. holder (and, if an applicable tax treaty so provides,
is attributable to a permanent establishment or fixed base maintained by the non-U.S. holder in the United States), the dividend
will not be subject to U.S. federal withholding tax (so long as the non-U.S. holder has provided the appropriate documentation,
generally an IRS Form W-8ECI (or appropriate substitute or successor form), to the withholding agent), but the non-U.S. holder
generally will be subject to U.S. federal income tax in respect of the dividend on a net income basis at regular U.S. federal income
tax rates in substantially the same manner as U.S. persons. Dividends received by a non-U.S. holder that is classified as a corporation
for U.S. federal income tax purposes and which are effectively connected with the conduct of a U.S. trade or business (and which
are not eligible, under the business profits article of an applicable tax treaty, for an exemption from U.S. taxation for business
profits that are not attributable to a permanent establishment or fixed base maintained by the non-U.S. holder in the United States
(herein not “Treaty Exempt”)) may also be subject to a branch profits tax at the rate of 30% (or a lower rate if provided
by an applicable tax treaty).
A non-U.S. holder that is eligible for a reduced
rate of U.S. federal withholding tax under an income tax treaty may obtain a refund or credit of any excess amounts withheld by
timely filing an appropriate claim for a refund together with the required information with the IRS.
Constructive Distributions on Common Warrants
Pursuant to the terms of the common warrants,
the exercise price at which the Common Stock may be purchased and/or the number of shares of Common Stock that may be purchased
upon exercise of the common warrants is subject to adjustment from time to time upon the occurrence of certain events. To the extent
an adjustment, or failure to adjust, the number of shares of the Common Stock underlying the common warrants and/or the exercise
price of the common warrants results in an increase in the proportionate interest of a holder in our assets or our earnings and
profits, such holder generally will be treated as having received a distribution of property. Any such deemed distribution generally
would be treated in the same manner as cash distributions on the Common Stock, as described above under “Distributions on
Common Stock.” In the event such a deemed distribution is taxable, a non-U.S. holder’s tax basis will be increased
by an amount equal to the taxable distribution.
Not all changes in the exercise price that
result in a holder’s receiving more Common Stock on exercise, however, would be considered as increasing the proportionate
interest of a holder in our assets or our earnings and profits. For instance, a change in exercise price could simply prevent the
dilution of a holder’s interest upon a stock split or other change in capital structure. Changes of this type, if made pursuant
to a bona fide, reasonable adjustment formula, are not treated as constructive stock distributions for these purposes. Conversely,
if an event occurs that dilutes a holder’s interest and the exercise price is not adjusted, the resulting increase in the
proportionate interest of a holder in our assets or our earnings and profits could be treated as a taxable stock distribution to
our holders.
Sale, Exchange or Other Disposition of Common
Stock or Common Warrants
Subject to the discussion of backup withholding
and FATCA below, a non-U.S. holder generally will not be subject to U.S. federal income tax (including withholding tax) on gain
realized on the sale, exchange or other disposition of the Common Stock or common warrants unless:
|
|
●
|
such non-U.S. holder is an individual who
is present in the United States for 183 days or more in the taxable year of such sale, exchange or disposition, and certain other
conditions are met;
|
|
|
|
|
|
|
●
|
such gain is effectively connected with
the conduct by the non-U.S. holder of a trade or business in the United States and is not Treaty Exempt; or
|
|
|
|
|
|
|
●
|
we are or have been a “United States
real property holding corporation”, or a USRPHC, for U.S. federal income tax purposes at any time during the shorter of
the five-year testing period ending on the date of such disposition and the non-U.S. holder’s holding period of the Common
Stock or common warrants, and certain other conditions are met.
|
Gain realized by a non-U.S. holder
that is effectively connected with such non-U.S. holder’s conduct of a trade or business in the United States generally
will be subject to U.S. federal income tax on a net income basis at regular U.S. federal income tax rates in substantially the
same manner as a U.S. person (except as provided by an applicable tax treaty). In addition, if such non-U.S. holder is a corporation
for U.S. federal income tax purposes, it may also be subject to a branch profits tax at the rate of 30% (or a lower rate if provided
by an applicable tax treaty).
Generally, a corporation is a USRPHC if the
fair market value of its “United States real property interests” equals or exceeds 50% of the sum of the fair market
value of its worldwide (domestic and foreign) real property interests and its other assets used or held for use in a trade or business
(all as determined for U.S. federal income tax purposes). For this purpose, real property interests generally include land, improvements
and associated personal property. We believe that we are not currently a USRPHC for this purpose. If we were a USRPHC during the
applicable testing period, non-U.S. holders owning (directly or indirectly) more than 5% of the Common Stock generally would be
subject to U.S. federal income tax on the gain realized on the sale, exchange or disposition of the Common Stock or common warrants,
which would be treated as income effectively connected with a U.S. trade or business (and taxable as discussed above). Even if
we were a USRPHC during the testing period, U.S. federal income tax would not apply to gain realized on the sale, exchange or disposition
of the Common Stock or common warrants by a non-U.S. holder that owns (directly or indirectly) 5% or less of the Common Stock so
long as the Common Stock is “regularly traded on an established securities market” within the meaning of the applicable
U.S. Treasury regulations. Prospective investors should be aware that no assurance can be provided that the Common Stock will be
so regularly traded when a non-U.S. holder sells the Common Stock or common warrants.
Exercise, Lapse or Redemption of a Common
Warrant
A non-U.S. holder generally will not be subject
to U.S. federal income tax on the cash exercise of common warrants for shares of the Common Stock. A non-U.S. holder’s tax
basis in the share of Common Stock received upon exercise of a common warrant will generally be an amount equal to the sum of the
non-U.S. holder’s initial investment in the common warrant and the exercise price. It is unclear whether the non-U.S. holder’s
holding period for the Common Stock received upon exercise of the common warrant will begin on the date of the exercise or on the
date following the date of the exercise; in either case, the holding period will not include the period during which the non-U.S.
holder held the common warrant. If a common warrant is allowed to lapse unexercised, a non-U.S. holder will generally recognize
a capital loss equal to such holder’s tax basis in the common warrant and generally will be taxed as described above under
“Sale, Exchange or Other Disposition of Common Stock or Common Warrants.”
The U.S. federal income tax treatment of a
cashless exercise of common warrants into shares of Common Stock is unclear. A cashless exercise may not be taxable, either because
the exercise is not a realization event or because the exercise is treated as a recapitalization for U.S. federal income tax purposes.
In either situation, a non-U.S. holder’s basis in the Common Stock received would equal the holder’s basis in the common
warrants exercised therefor. If the cashless exercise were treated as not being a realization event, it is unclear whether a non-U.S.
holder’s holding period in the Common Stock received upon exercise of the common warrant will begin on the date of the exercise
or on the date following the date of the exercise; in either case, the holding period will not include the period during which
the non-U.S. holder held the common warrant. If the cashless exercise were treated as a recapitalization, the holding period of
the Common Stock would include the holding period of the common warrant exercised therefor. If a cashless exercise of the common
warrants were treated as a taxable exchange, the rules described above under “Sale, Exchange or Other Disposition of Common
Stock or Common Warrants” would apply. Non-U.S. holders should consult their tax advisors regarding the tax consequences
of a cashless exercise.
If we redeem our common warrants for cash or
if we purchase our common warrants in an open market transaction, such redemption or purchase will generally be treated as a taxable
disposition to the non-U.S. holder, taxed as described above under “Sale, Exchange or Other Disposition of Common Stock or
Common Warrants.”
Non-U.S. holders should consult their tax advisors
regarding the tax consequences of the exercise, lapse, or redemption of a common warrant.
Information Reporting and Backup Withholding
Dividends and proceeds from the sale, exchange
or other disposition of the Common Stock or common warrants are potentially subject to backup withholding at the applicable rate.
In general, backup withholding will not apply to dividends on the Common Stock or common warrants paid by us or our paying agents,
in their capacities as such, to a non-U.S. holder if the holder has provided the required certification that it is a non-U.S. holder,
such as by providing an IRS Form W-8BEN, W-8BEN-E or IRS Form W-8ECI (or appropriate substitute or successor form) and neither
we nor our paying agent has actual knowledge (or reason to know) that the holder is a U.S. holder that is not an exempt recipient.
Backup withholding is not an additional tax.
Any amounts withheld under the backup withholding rules will be allowed as a refund or a credit against a non-U.S. holder’s
U.S. federal income tax liability, provided the required information is furnished on a timely basis to the IRS.
Non-U.S. holders should consult their tax advisors
regarding the application of the information reporting and backup withholding rules to them.
Foreign Account Tax Compliance Act
The Foreign Account Tax Compliance Act (generally
referred to as “FATCA”), when applicable, will impose a U.S. federal withholding tax of 30% on certain payments to
“foreign financial institutions” (which are broadly defined for this purpose and generally include investment
vehicles) and certain other non-U.S. entities unless various U.S. information reporting and due diligence requirements (generally
relating to ownership by U.S. persons of certain interests in or accounts with those entities) have been satisfied. Payments subject
to withholding tax under FATCA include dividends on common stock of U.S. corporations (such as the Common Stock). Although under
the Code payments subject to withholding tax under FATCA also include the gross proceeds of a disposition of stock (including a
liquidating distribution from a corporation) or debt instruments, in each case with respect to any U.S. investment, proposed Treasury
Regulations published on December 18, 2018, provide that such gross proceeds are not “withholdable payments” under
FATCA and therefore not subject to withholding tax. Such proposed Treasury Regulations provide that Taxpayers generally may rely
on the proposed Treasury Regulations until final Treasury Regulations are issued. Under certain circumstances, a non-U.S. holder
might be eligible for refunds or credits of amounts withheld. An intergovernmental agreement between the United States and an applicable
foreign country may modify the requirements described in this paragraph. Non-U.S. holders should consult their own tax advisors
regarding the potential application and impact of these requirements based upon their particular circumstances.
U.S. Federal Estate Tax
Common Stock or common warrants owned or treated
as owned by an individual who is not a citizen or resident of the United States (as specifically defined for U.S. federal estate
tax purposes) at the time of death will be included in the individual’s gross estate for U.S. federal estate tax purposes
and may be subject to U.S. federal estate tax unless an applicable estate tax treaty provides otherwise.
UNDERWRITING
Maxim Group LLC (the “representative”
or “Maxim”) is acting as sole book-running manager of this offering. We have entered into an underwriting agreement
dated September 29, 2021 with the representative. Subject to the terms and conditions of the underwriting agreement, we have agreed
to sell to each underwriter named below, and each underwriter named below has severally agreed to purchase from us, at the public
offering price less the underwriting discounts set forth on the cover page of this prospectus supplement, the number of shares
of Common Stock and common warrants listed next to its name in the following table.
The underwriting agreement provides for the
purchase by the underwriter named below of the number of shares of Common Stock set forth opposite its name:
|
Name
|
|
Number of Shares
|
|
Number of Common Warrants
|
|
Maxim Group LLC
|
|
|
4,700,000
|
|
|
|
3,525,000
|
|
|
Total
|
|
|
4,700,000
|
|
|
|
3,525,000
|
|
The underwriter has agreed to purchase all
of the shares and common warrants offered by this prospectus supplement (other than those covered by the over-allotment option
described below) if any are purchased. Under the underwriting agreement, if an underwriter defaults in its commitment to purchase
shares and common warrants, the underwriting agreement may be terminated, depending on the circumstances.
The shares and common warrants should be ready
for delivery on or about October 4, 2021 against payment in immediately available funds. The underwriter is offering the shares
and common warrants subject to various conditions and may reject all or part of any order. The representative has advised us that
the underwriter proposes to offer the shares and common warrants directly to the public at the public offering price that appears
on the cover page of this prospectus supplement. In addition, the representative may offer some of the shares and common warrants
to other securities dealers at such price less a concession of $0.05525 per share and common warrant. The underwriter may also
allow, and such dealers may reallow, a concession not in excess of $0.05525 per share and common warrant to other dealers. After
the shares and common warrants are released for sale to the public, the representative may change the offering price and other
selling terms at various times.
Over-Allotment Option
We have granted the underwriter an over-allotment
option. This option, which is exercisable for up to 45 days after the date of this prospectus, permits the underwriter to purchase
a maximum of 705,000 additional shares of Common Stock and/or additional common warrants to purchase up to 528,750 shares of Common
Stock from us to cover over-allotments. If the underwriter exercises all or part of this option, it will purchase shares and/or
common warrants covered by the option at the public offering price that appears on the cover page of this prospectus supplement,
less the underwriting discount. If this option is exercised in full, the total proceeds to us in connection with this offering
will be approximately $9,188,500, before deduction of underwriting discounts and expenses and other offering expenses.
Discount
The following table shows the public offering
price, underwriting discounts and proceeds, before expenses, to us. The information assumes either no exercise or full exercise
by the underwriter of its over-allotment option.
|
|
|
Per Share and Common Warrant
|
|
Total Without Over-Allotment
Option
|
|
Total With Over-Allotment
Option
|
|
Public offering price
|
|
$
|
1.70
|
|
|
$
|
7,990,000
|
|
|
$
|
9,188,500
|
|
|
Underwriting discount (7%)
|
|
$
|
0.119
|
|
|
$
|
559,300
|
|
|
$
|
643,195
|
|
|
Proceeds, before expense, to us
|
|
$
|
1.581
|
|
|
$
|
7,430,700
|
|
|
$
|
8,545,305
|
|
We have agreed to pay Maxim’s out-of-pocket
accountable expenses, including the reasonable fees of Maxim’s legal counsel, not to exceed $125,000, if this offering is completed.
We estimate that expenses payable by us in connection with this offering, including reimbursement of the underwriter’s out-of-pocket
expenses, but excluding the underwriting discount referred to above, will be approximately $399,500.
Lock-Up Agreements
We, our officers and directors, and certain
of our other stockholders have agreed, subject to limited exceptions, for a period of ninety (90) days after the date of the underwriting
agreement, not to offer, sell, contract to sell, pledge, grant any option to purchase, make any short sale or otherwise dispose
of, directly or indirectly any shares of Common Stock or any securities convertible into or exchangeable for the Common Stock either
owned as of the date of the underwriting agreement or thereafter acquired without the prior written consent of the underwriter.
Right of First Refusal
We have agreed to grant Maxim, for the period
of eighteen (18) months following the closing of the offering, a right of first refusal to act as sole managing underwriter and
sole book runner and/or sole placement agent for any and all future public and private equity, equity-linked, convertible and debt
offerings of the Company, or any successor to or any subsidiary of the Company. Maxim will not have more than one opportunity to
waive or terminate the right of first refusal in consideration of any payment or fee.
Indemnification
We have agreed to indemnify the underwriter
against certain liabilities, including liabilities under the Securities Act.
Price Stabilization, Short Positions and
Penalty Bids
Rules of the SEC may limit the ability of the
underwriter to bid for or purchase shares before the distribution of the shares is completed. However, the underwriter may engage
in the following activities in accordance with the rules:
|
|
●
|
Stabilizing
transactions — The representative may make bids
or purchases for the purpose of pegging, fixing or maintaining the
price of the shares, so long as stabilizing bids do not exceed a
specified maximum.
|
|
|
|
|
|
|
●
|
Over-allotments
and syndicate covering transactions — The underwriter
may sell more shares of Common Stock in connection with this offering
than the number of shares that they have committed to purchase.
This over-allotment creates a short position for the underwriter.
This short sales position may involve either “covered”
short sales or “naked” short sales. Covered short sales
are short sales made in an amount not greater than the underwriter’s
over-allotment option to purchase additional shares in this offering
described above. The underwriter may close out any covered short
position either by exercising its over-allotment option or by purchasing
shares in the open market. To determine how it will close the covered
short position, the underwriter will consider, among other things,
the price of shares available for purchase in the open market, as
compared to the price at which they may purchase shares through
the over-allotment option. Naked short sales are short sales in
excess of the over-allotment option. The underwriter must close
out any naked short position by purchasing shares in the open market.
A naked short position is more likely to be created if the underwriter
is concerned that, in the open market after pricing, there may be
downward pressure on the price of the shares that could adversely
affect investors who purchase shares in this offering.
|
|
|
|
|
|
|
●
|
Penalty
bids — If the representative purchases the shares
in the open market in a stabilizing transaction or syndicate covering
transaction, it may reclaim a selling concession from the underwriter
and selling group members who sold those shares as part of this
offering.
|
|
|
|
|
|
|
●
|
Passive
market making — Market makers in the shares
who are underwriters or prospective underwriters may make bids for
or purchase the shares, subject to limitations, until the time,
if ever, at which a stabilizing bid is made.
|
Similar to other purchase transactions, the
underwriter’s purchases to cover the syndicate short sales or to stabilize the market price of the Common Stock may have
the effect of raising or maintaining the market price of the Common Stock or preventing or mitigating a decline in the market price
of the Common Stock. As a result, the price of the Common Stock may be higher than the price that might otherwise exist in the
open market. The imposition of a penalty bid might also have an effect on the price of the Common Stock if it discourages resales
of the shares.
Neither we nor the underwriter make any representation
or prediction as to the effect that the transactions described above may have on the price of the Common Stock. These transactions
may occur on Nasdaq or otherwise. If such transactions are commenced, they may be discontinued without notice at any time.
Electronic Delivery of Preliminary Supplement
and Accompanying Prospectus
A prospectus in electronic format may be delivered
to potential investors by the underwriter. The prospectus in electronic format will be identical to the paper version of such preliminary
prospectus. Other than the prospectus in electronic format, the information on any underwriter’s website and any information
contained in any other website maintained by an underwriter is not part of the prospectus or the registration statement of which
this prospectus forms a part.
Notice to Non-US Investors
Canada
The securities may be sold in Canada only to
purchasers purchasing, or deemed to be purchasing, as principal that are “accredited investors”, as defined in National
Instrument 45-106 Prospectus Exemptions or subsection 73.3(1) of the Securities Act (Ontario), and are “permitted clients”,
as defined in National Instrument 31-103 Registration Requirements, Exemptions and Ongoing Registrant Obligations. Any resale of
the securities must be made in accordance with an exemption from, or in a transaction not subject to, the prospectus requirements
of applicable securities laws. Securities legislation in certain provinces or territories of Canada may provide a purchaser with
remedies for rescission or damages if this prospectus (including any amendment thereto) contains a misrepresentation, provided
that the remedies for rescission or damages are exercised by the purchaser within the time limit prescribed by the securities legislation
of the purchaser’s province or territory. The purchaser should refer to any applicable provisions of the securities legislation
of the purchaser’s province or territory for particulars of these rights or consult with a legal advisor. Pursuant to section
3A.3 of National Instrument 33-105 Underwriting Conflicts (NI 33-105), the underwriter is not required to comply with the disclosure
requirements of NI 33-105 regarding underwriter conflicts of interest in connection with this offering.
European Economic Area
In relation to each Member State of the European
Economic Area which has implemented the Prospectus Directive, each, a Relevant Member State, with effect from and including the
date on which the European Union Prospectus Directive, or the EU Prospectus Directive, was implemented in that Relevant Member
State, or the Relevant Implementation Date, no offer of securities may be made to the public in that Relevant Member State other
than:
|
|
1.
|
to any legal entity which is a qualified
investor as defined under the EU Prospectus Directive;
|
|
|
|
|
|
|
2.
|
to fewer than 150 natural or legal persons
(other than qualified investors as defined in the EU Prospectus Directive), subject to obtaining the prior consent of the representatives;
or
|
|
|
|
|
|
|
3.
|
in any other circumstances falling within
Article 3(2) of the EU Prospectus Directive;
|
provided that no such offer of securities
shall require the Company or any underwriter to publish a prospectus pursuant to Article 3 of the Prospectus Directive and each
person who initially acquires any securities or to whom any offer is made will be deemed to have represented, acknowledged and
agreed to and with the underwriter and the Company that it is a “qualified investor” within the meaning of the law
in that Relevant Member State implementing Article 2(1)(e) of the Prospectus Directive.
In the case of any securities being offered
to a financial intermediary as that term is used in Article 3(2) of the Prospectus Directive, each such financial intermediary
will be deemed to have represented, acknowledged and agreed that the securities acquired by it in the offer have not been acquired
on a non-discretionary basis on behalf of, nor have they been acquired with a view to their offer or resale to, persons in circumstances
which may give rise to an offer of any securities to the public other than their offer or resale in a Relevant Member State to
qualified investors as so defined or in circumstances in which the prior consent of the representatives has been obtained to each
such proposed offer or resale.
For the purposes of this provision, the expression
an “offer of securities to the public” in relation to any securities in any Relevant Member State means the communication
in any form and by any means of sufficient information on the terms of the offer and the securities to be offered so as to enable
an investor to decide to purchase or subscribe for the securities, as the same may be varied in that Member State by any measure
implementing the EU Prospectus Directive in that Member State. The expression “EU Prospectus Directive” means Directive
2003/71/EC (and any amendments thereto, including the 2010 PD Amending Directive, to the extent implemented in the Relevant Member
State) and includes any relevant implementing measure in each Relevant Member State, and the expression “2010 PD Amending
Directive” means Directive 2010/73/EU.
United Kingdom
In the United Kingdom, this document is being
distributed only to, and is directed only at, and any offer subsequently made may only be directed at persons who are “qualified
investors” (as defined in the Prospectus Directive) (i) who have professional experience in matters relating to investments
falling within Article 19(5) of the Financial Services and Markets Act 2000 (Financial Promotion) Order 2005, as amended, or the
Order, and/or (ii) who are high net worth companies (or persons to whom it may otherwise be lawfully communicated) falling within
Article 49(2)(a) to (d) of the Order (all such persons together being referred to as “relevant persons”) or otherwise
in circumstances which have not resulted and will not result in an offer to the public of the securities in the United Kingdom.
Any person in the United Kingdom that is not
a relevant person should not act or rely on the information included in this document or use it as basis for taking any action.
In the United Kingdom, any investment or investment activity that this document relates to may be made or taken exclusively by
relevant persons.
WHERE YOU CAN FIND ADDITIONAL INFORMATION
We file annual, quarterly and other periodic
reports, proxy statements and other information with the SEC. You can read our SEC filings over the Internet at the SEC’s
website at www.sec.gov.
Our Internet address is www.kaivalbrands.com.
There we make available free of charge, on or through the investor relations section of our website, annual reports on Form 10-K,
quarterly reports on Form 10-Q, current reports on Form 8-K and amendments to those reports filed pursuant to Section 13(a) or
15(d) of the Exchange Act as soon as reasonably practicable after we electronically file such material with the SEC. The information
found on our website is not part of this prospectus supplement or the accompanying prospectus.
INCORPORATION OF CERTAIN INFORMATION BY REFERENCE
We are “incorporating by reference”
specific documents that we file with the SEC, which means that we can disclose important information to you by referring you to
those documents that are considered part of this prospectus supplement and the accompanying prospectus. Information that we file
subsequently with the SEC will automatically update and supersede this information. We incorporate by reference the documents listed
below, and any documents that we file with the SEC under Section 13(a), 13(c), 14 or 15(d) of the Exchange Act, after the date
of this prospectus supplement until the termination of the offering of all of the securities registered pursuant to the registration
statement of which the accompanying prospectus is a part (excluding any portions of such documents that have been “furnished”
but not “filed” for purposes of the Exchange Act):
|
|
●
|
our Annual Report on Form 10-K for the
fiscal year ended October 31, 2020 filed with the SEC on February 12, 2021;
|
|
|
|
|
|
|
●
|
our Quarterly Reports on Form 10-Q for the fiscal quarters ended January
31, 2021, April 30, 2021 and July 31, 2021, filed with the SEC on March 16, 2021, June 21, 2021 and September 14, 2021, respectively;
|
|
|
|
|
|
|
●
|
our Current Reports on Form 8-K filed with
the SEC on January 12, 2021, January 15, 2021, March 18, 2021, April 21, 2021 (Item 1.01 and Exhibit 10.1 only), May 10, 2021,
May 12, 2021, July 1, 2021 (Item 1.01 and Exhibits 10.1 and 10.2 only), July 20, 2021 (Items 3.03 and 5.03 and Exhibit 3.1 only)
and July 23, 2021; and
|
|
|
|
|
|
|
●
|
the description of the Common Stock contained
in our Registration Statement on Form 8-A filed with the SEC on July 20, 2021, to register such securities under the Exchange
Act, including any amendments filed for the purpose of updating such information.
|
You may request, and we will provide you with,
a copy of these filings, at no cost, by calling us at (833) 452-4825 or by writing to us at the following address:
Kaival Brands Innovations Group, Inc.
4460 Old Dixie Highway
Grant, Florida 32949
Attn: Eric Mosser
Any statement contained herein or in a document
incorporated or deemed to be incorporated by reference herein shall be deemed to be modified or superseded for purposes of this
prospectus supplement and the accompanying prospectus to the extent that a statement contained herein or therein, in any other
subsequently filed document that also is or is deemed to be incorporated by reference herein and in any accompanying prospectus
supplement, modifies or supersedes such statement. Any statement so modified or superseded shall not be deemed, except as so modified
and superseded, to constitute a part of this prospectus supplement.
Any statement made in this prospectus supplement
and the accompanying prospectus concerning the contents of any contract, agreement or other document is only a summary of the actual
contract, agreement or other document. If we have filed or incorporated by reference any contract, agreement or other document
as an exhibit to the registration statement, you should read the exhibit for a more complete understanding of the document or matter
involved. Each statement regarding a contract, agreement or other document is qualified by reference to the actual document.
You should not assume that the information
in this prospectus supplement or the accompanying prospectus is accurate as of any date other than the dates on the front pages
of these documents.
LEGAL MATTERS
Certain legal matters relating to the issuance
of the securities offered by this prospectus supplement will be passed upon for us by Morgan, Lewis & Bockius LLP, Philadelphia.
Loeb & Loeb, LLP, New York, New York, has acted as counsel for the underwriter.
EXPERTS
MaloneBailey, LLP, independent registered public
accounting firm, has audited our consolidated financial statements included in our Annual Report on Form 10-K for the year ended
October 31, 2020, as set forth in their report, which is incorporated by reference in this prospectus supplement. Our consolidated
financial statements are incorporated by reference in reliance on MaloneBailey, LLP’s report, given on their authority as
experts in accounting and auditing.
PROSPECTUS

$200,000,000
KAIVAL BRANDS INNOVATIONS GROUP, INC.
Common Stock
Preferred Stock
Warrants
Debt Securities
Rights to Purchase Common Stock, Preferred
Stock,
Debt Securities or Units
Units
We may offer and sell from time to time our
shares of common stock, shares of preferred stock, warrants, debt securities and rights to purchase common stock, preferred stock,
debt securities or units, as well as units that include any of these securities. We may sell any combination of these securities
in one or more offerings with an aggregate offering price of up to $200,000,000.
This prospectus provides you with a general
description of the securities we may offer. Each time we offer securities pursuant to this prospectus, we will provide a prospectus
supplement containing specific terms of the particular offering together with this prospectus. You should read this prospectus
and the applicable prospectus supplement carefully before you invest in any securities. The prospectus supplement also may add,
update or change information contained in this prospectus. This prospectus may not be used to offer and sell securities unless
accompanied by the applicable prospectus supplement.
Our common stock is listed on the Nasdaq Capital
Market under the symbol “KAVL.” On July 29, 2021, the closing price of our common stock was $8.80.
Investing in our securities involves significant
risks. We strongly recommend that you read carefully the risks we describe in this prospectus and in any accompanying prospectus
supplement, as well as the risk factors that are incorporated by reference into this prospectus from our filings made with the
Securities and Exchange Commission. See “Risk Factors” on page 3 of this prospectus.
We may sell the securities directly or to or
through underwriters or dealers, and also to other purchasers or through agents. The names of any underwriters or agents that are
included in a sale of securities to you, and any applicable commissions or discounts, will be stated in an accompanying prospectus
supplement. In addition, the underwriters, if any, may over-allot a portion of the securities.
Neither the Securities and Exchange Commission
nor any state securities commission has approved or disapproved of these securities or passed upon the adequacy or accuracy of
this prospectus. Any representation to the contrary is a criminal offense.
The date of this prospectus is , 2021.
ABOUT THIS PROSPECTUS
This prospectus is part of a registration statement
that we filed with the Securities and Exchange Commission (the “SEC”), using a “shelf” registration process.
Under this shelf registration process, we may offer and sell from time to time any combination of the securities described in this
prospectus in one or more offerings in amounts, at prices and on terms that we determine at the time of the offering, with an aggregate
offering price of up to $200,000,000. This prospectus provides you with a general description of the securities we may offer. Each
time we offer securities under this registration statement we will provide a prospectus supplement that describes the terms of
the relevant offering. The prospectus supplement also may add, update or change information contained in this prospectus. Before
making an investment decision, you should read carefully both this prospectus and any prospectus supplement together with the documents
incorporated by reference into this prospectus as described below under the heading “Information Incorporated by Reference.”
The registration statement that contains this
prospectus, including the exhibits to the registration statement and the information incorporated by reference, provides additional
information about us and our securities. That registration statement can be read at the SEC website (www.sec.gov), as discussed
below under the heading “Where You Can Find More Information.”
You should rely only on the information provided
in the registration statement, this prospectus and in any prospectus supplement, including the information incorporated by reference.
We have not authorized anyone to provide you with different information. You should not assume that the information in this prospectus
or any supplement to this prospectus is accurate at any date other than the date indicated on the cover page of these documents
or the filing date of any document incorporated by reference, regardless of its time of delivery. We are not making an offer to
sell the securities in any jurisdiction where the offer or sale is not permitted.
We may sell our securities to or through underwriters,
initial purchasers, dealers or agents, directly to purchasers or through a combination of any of these methods of sale, as designated
from time to time. We and our agents reserve the sole right to accept or reject in whole or in part any proposed purchase of our
securities. An applicable prospectus supplement, which we will provide each time we offer the securities, will set forth the names
of any underwriters, initial purchasers, dealers or agents involved in the sale of our securities, and any related fee, commission
or discount arrangements. See “Plan of Distribution.”
The terms “Kaival,” the “Company,”
“our,” “us” and “we,” as used in this prospectus, refer to Kaival Brands Innovations Group, Inc.,
unless we state otherwise or the context indicates otherwise.
KAIVAL BRANDS INNOVATIONS GROUP, INC.
We are focused on growing and incubating innovative
and profitable products into mature, dominant brands. In March 2020, we commenced business operations as a result of becoming the
exclusive distributor of certain electronic nicotine delivery systems and related components (the “Products”) manufactured
by Bidi Vapor, LLC (“Bidi”), a Florida limited liability company, and a related party company that is also owned by
Nirajkumar Patel, our President and Chief Executive Officer.
On March 9, 2020, we entered
into an exclusive distribution agreement with Bidi, which agreement was amended and restated on May 21, 2020 and again on April 20,
2021 (the “A&R Distribution Agreement”). Pursuant to the A&R Distribution Agreement, Bidi granted to us an
exclusive worldwide right to distribute the Products for sale and resale to both retail level customers and non-retail level
customers, and accordingly we sell and resell electronic nicotine delivery systems, which we may refer to
herein as “ENDS Products,” or “e-cigarettes,” to both retail level customers and non-retail level customers.
Our primary resold Product is the “Bidi Stick,” a disposable, tamper-resistant ENDS Product that comes in a variety of
flavor options. In addition to the Bidi Stick, we distribute of the “Bidi Pouch,” which provides a tobacco-free nicotine
formulation, containing natural fibers and a chew-base filler, in six different flavors. We do not manufacture any of the Products
we resell. The Bidi Stick and Bidi Pouch are manufactured by Bidi Pursuant to the terms of the A&R Distribution Agreement, Bidi
provides us with all branding, logos, and marketing materials to be utilized by us in connection with our marketing and promotion of
the Products.
Our principal executive offices are located
at 4460 Old Dixie Highway, Grant, FL 32949, and our telephone number is (833) 452-4825. Our website address is www.kaivalbrands.com.
The information contained on our website is not incorporated by reference into this prospectus, and you should not consider any
information contained on, or that can be accessed through, our website as part of this prospectus or in deciding whether to purchase
our common stock.
Our filings with the SEC are posted on our
website at www.kaivalbrands.com. The information found on our website is not part of this or any other report we file with or
furnish to the SEC. The public can also obtain copies of these filings by accessing the SEC’s website at www.sec.gov.
Recent Development
On July 20, 2021, we effected a 1-for-12 reverse
split (the “Reverse Stock Split”) of our shares of the shares of the Company’s common stock, par value $0.001
per share. No fractional shares were issued in connection with the Reverse Stock Split. Any fractional shares of common stock that
would have otherwise resulted from the Reverse Stock Split was rounded up to the nearest whole number. In connection with the Reverse
Stock Split, our board of directors approved appropriate and proportional adjustments to all outstanding securities or other rights
convertible or exercisable into shares of common stock, including, without limitation, all preferred stock, warrants, options and
other equity compensation rights. Share amounts set forth in this prospectus have been adjusted to reflect the Reverse Stock Split.
FORWARD-LOOKING STATEMENTS
From time to time, in reports filed with the
SEC (including this prospectus), in press releases and in other communications to stockholders or the investment community, we
may provide forward-looking statements concerning possible or anticipated future results of operations or business developments.
These statements are based on our management’s current expectations or predictions of future conditions, events or results
based on various assumptions and our management’s estimates of trends and economic factors in the markets in which we are
active, as well as our business plans. Words such as “expects,” “anticipates,” “intends,” “plans,”
“believes,” “seeks,” “estimates,” “projects,” “forecasts,” “may,”
“should,” and variations of such words and similar expressions are intended to identify such forward-looking statements.
The forward-looking statements may include, without limitation, statements regarding product development and commercialization,
product candidate potential, regulatory environment, sales and marketing strategies, capital resources or operating performance.
The forward-looking statements are subject to risks and uncertainties, which may cause results to differ materially from those
set forth in the statements. Forward-looking statements in this prospectus should be evaluated together with the many uncertainties
that affect our business and our market to better understand the risks and uncertainties inherent in our business and underlying
any forward-looking statements. Forward-looking statements are not guarantees of future performance, and actual results may differ
materially from those projected. The forward-looking statements are representative only as of the date of this prospectus and except
as required by law, we assume no responsibility to update any forward-looking statements, whether as a result of new information,
future events or otherwise.
You should read this prospectus and the documents
that we reference in this prospectus and have been filed as exhibits to the registration statement of which this prospectus is
a part completely and with the understanding that our actual future results may be materially different from what we expect. The
information contained in this prospectus is accurate only as of the date of this prospectus, regardless of the time of delivery
of this prospectus or any issuance or sale of our common shares. Except as required by law, we do not assume any obligation to
update any forward-looking statements.
RISK FACTORS
Investing in our securities involves risk. You
should carefully consider the specific risks discussed or incorporated by reference into the applicable prospectus supplement,
together with all the other information contained in the prospectus supplement or incorporated by reference into this prospectus
and the applicable prospectus supplement, including without limitation any that may be contained in our Annual Reports on Form
10-K and our Quarterly Reports on Form 10-Q. These risk factors may be amended, supplemented or superseded from time to time by
other reports we file with the SEC in the future or by a prospectus supplement relating to a particular offering of our securities.
These risks and uncertainties are not the only risks and uncertainties we face. Additional risks and uncertainties not presently
known to us, or that we currently view as immaterial, may also impair our business. If any of the risks or uncertainties described
in our SEC filings or any prospectus supplement or any additional risks and uncertainties actually occur, our business, financial
condition and results of operations could be materially and adversely affected. In that case, the trading price of our securities
could decline and you might lose all or part of your investment.
USE OF PROCEEDS
Unless otherwise indicated in the applicable
prospectus supplement, we will use the net proceeds from the sale of the securities offered hereby for general corporate purposes,
which include, but are not limited to, capital expenditures, additions to working capital, general and administrative
expenses or other corporate obligations. We may use a portion of the net proceeds to pay off outstanding indebtedness, if any,
and/or acquire or invest in businesses, products and technologies. We have not determined the amount of net proceeds to be used
specifically for such purposes. As a result, management will retain broad
discretion over the allocation of net proceeds.
DESCRIPTION OF CAPITAL STOCK
The following description is a general summary
of the terms of the shares of common stock or shares of preferred stock that we may issue, and gives effect to the Reverse Stock
Split. The description below and in any prospectus supplement does not include all of the terms of the shares of common stock or
shares of preferred stock and should be read together with our Amended and Restated Certificate of Incorporation and Bylaws, copies
of which have been filed previously with the SEC. For more information on how you can obtain copies of our Amended and Restated
Certificate of Incorporation and Bylaws, see “Where You Can Find More Information.”
Common Stock
General
Our Amended and Restated Certificate of
Incorporation provides the authority to issue 1,000,000,000 shares of common stock, par value $0.001 per share. At July 23, 2021,
there were 23,600,597 shares of common stock outstanding. Each share of our common stock has the same relative rights and is
identical in all respects to each other share of our common stock. The rights, preferences and privileges of holders of our common
stock are subject to the rights, preferences and privileges of the holders of shares of any series of preferred stock that we have
issued or may issue in the future.
Voting Rights
The holders of our common stock are entitled
to one vote per share on any matter to be voted upon by our stockholders. Our Amended and Restated Certificate of Incorporation
does not permit cumulative voting in connection with the election of directors.
Dividends
The holders of our common stock are entitled
to dividends, if any, when and as declared by our board of directors out of funds legally available therefore for distribution
to stockholders, subject to the holders of other classes of stock, if any, at the time outstanding having prior rights as to dividends,
if any.
Liquidation Rights
Upon any voluntary or involuntary liquidation,
dissolution, or winding up of our affairs, the holders of our common stock are entitled to share ratably in all assets remaining
after the payment of creditors, subject to any prior liquidation distribution rights of holders of other classes of stock, if any,
at the time outstanding.
Miscellaneous
Holders of our common stock have no preemptive,
conversion, redemption or sinking fund rights. The outstanding shares of our common stock are, and the shares of common stock to
be offered hereby when issued will be, validly issued, fully paid and non-assessable.
Nasdaq Listing
Our common stock is listed on the Nasdaq Capital
Market under the symbol “KAVL.”
Transfer Agent and Registrar
The transfer agent and registrar for our common
stock is Vstock Transfer, LLC whose address is 18 Lafayette Place, Woodmere, NY 11598.
Preferred Stock
General
Our Amended and Restated Certificate of Incorporation
authorizes the issuance of up to 5,000,000 shares of preferred stock, par value $0.001 per share. Of the 5,000,000 shares of preferred
stock authorized, our board of directors has previously designated 3,000,000 shares of preferred stock as Series A Convertible
Preferred Stock (the “Series A Preferred Stock”), of which 3,000,000 shares remain outstanding. The 2,000,000 shares
of preferred stock not designated as Series A Convertible Preferred Stock remain available for designation by our board of directors.
Accordingly, our board of directors is empowered, without stockholder approval, to issue preferred stock with dividend, liquidation,
conversion, voting or other rights that could adversely affect the voting power or other rights of the holders of common stock.
The issuance of preferred stock could have the effect of restricting dividends on the common stock, diluting the voting power of
the common stock, impairing the liquidation rights of the common stock, or delaying or preventing a change in control of the Company,
all without further action by the stockholders.
The description of the terms of a particular
series of preferred stock in the applicable prospectus supplement will not be complete. You should refer to the applicable certificate
of designation for complete information regarding a series of preferred stock. The prospectus supplement will also contain a description
of U.S. federal income tax consequences relating to the preferred stock, if material.
The terms of any particular series of preferred
stock will be described in the prospectus supplement relating to that particular series of preferred stock, including, where applicable:
|
|
●
|
the series designation, stated value and liquidation preference of
such preferred stock and the number of shares offered;
|
|
|
|
|
|
|
●
|
the offering price;
|
|
|
|
|
|
|
●
|
the dividend rate or rates (or method of calculation), the date or
dates from which dividends shall accrue, and whether such dividends shall be cumulative or noncumulative and, if cumulative, the
dates from which dividends shall commence to cumulate;
|
|
|
|
|
|
|
●
|
any redemption or sinking fund provisions;
|
|
|
|
|
|
|
●
|
the amount that shares of such series shall be entitled to receive
in the event of our liquidation, dissolution or winding-up;
|
|
|
●
|
the terms and conditions, if any, on which shares of such series shall be convertible or exchangeable for shares of our stock of any other class or classes, or other series of the same class;
|
|
|
|
|
|
|
●
|
the voting rights, if any, of shares of such series in addition to those set forth under the caption entitled, “Voting Rights” below;
|
|
|
|
|
|
|
●
|
the status as to reissuance or sale of shares of such series redeemed, purchased or otherwise reacquired, or surrendered to us on conversion or exchange;
|
|
|
|
|
|
|
●
|
the conditions and restrictions, if any, on the payment of dividends or on the making of other distributions on, or the purchase, redemption or other acquisition by us, of our common stock or of any other class of our stock ranking junior to the shares of such series as to dividends or upon liquidation (including, but not limited to, at such times as there are arrearages in the payment of dividends or sinking fund installments);
|
|
|
|
|
|
|
●
|
the conditions and restrictions, if any, on the creation of Company indebtedness, or on the issue of any additional stock ranking on a parity with or prior to the shares of such series as to dividends or upon liquidation; and
|
|
|
|
|
|
|
●
|
any additional dividend, liquidation, redemption, sinking or retirement fund and other rights, preferences, privileges, limitations and restrictions of such preferred stock.
|
If we issue shares of preferred stock under
this prospectus and any related prospectus supplement, the shares will be fully paid and non-assessable and will not have, or be
subject to, any preemptive or similar rights.
Series A Preferred Stock
Each share of Series A Preferred Stock is convertible
into 8.333 shares of common stock. The holders of the Series A Preferred Stock may convert their Series A Preferred Stock at
any time on or after November 1, 2023. Notwithstanding the foregoing, the holders of the Series A Preferred Stock may convert their
shares of Series A Preferred Stock prior to November 1, 2023 if a change of control (as provided for in the Certificate of Designation
of Preferences, Rights, and Limitations of the Series A Preferred Stock) or upon the occurrence of any other event as determined
and agreed to by the Company and the holders holding a majority of the issued and outstanding shares of Series A Preferred Stock.
The shares of common stock to be issued upon conversion will bear a restricted legend. The holders of the Series A Preferred Stock
do not have any voting rights nor any preferential dividend rights, and are entitled to receive dividends, if any, only if, when,
and as declared by our board of directors in its sole and absolute discretion. In the event the Company liquidates, dissolves,
or winds up, holders of the Series A Preferred Stock will have the right to receive an amount equal to $12.00 in the aggregate
for all issued and outstanding shares of Series A Preferred Stock (as adjusted for any stock dividends, combinations, splits, recapitalizations,
and the like with respect to such shares) (“Preference Value”). After the payment of the full applicable Preference
Value of the then issued and outstanding shares of the Series A Preferred Stock, the Company’s remaining assets legally available
for distribution, if any, will be distributed ratably to the holders of Common Stock.
Share and dollar amounts set forth in the foregoing paragraph have been adjusted to reflect the Reverse Stock Split.
Voting Rights
The General Corporation Law of Delaware provides
that the holders of preferred stock will have the right to vote separately as a class on any proposal involving fundamental changes
in the rights of holders of that preferred stock. This right is in addition to any voting rights that may be provided for in the
applicable certificate of designation.
Transfer Agent and Registrar
The transfer agent and registrar for any series
of preferred stock will be set forth in the applicable prospectus supplement.
Other
Our issuance of preferred stock could decrease
the amount of earnings and assets available for distribution to the holders of common stock or could adversely affect the rights
and powers, including voting rights, of the holders of common stock. The issuance of preferred stock could have the effect of decreasing
the market price of our common stock.
Delaware Law and Certain Amended and Restated Certificate
of Incorporation and Bylaws Provisions
Our Amended and Restated Certificate of Incorporation
and Bylaws contain provisions that could delay or prevent a change of control of our company or changes in our board of directors
that our stockholders might consider favorable. Some of these provisions:
|
|
●
|
Authorize the issuance of preferred stock which can be created and issued by the board of directors without prior stockholder approval, with rights senior to those of our common stock;
|
|
|
|
|
|
|
●
|
Prohibit our stockholders from filling board vacancies or calling special stockholder meetings; and
|
|
|
|
|
|
|
●
|
Require any action instituted against our officers or directors in connection with their service to the Company to be brought in the state of Delaware.
|
These and other provisions in our Amended and
Restated Certificate of Incorporation, Bylaws and Delaware law could make it more difficult for stockholders or potential acquirors
to obtain control of our board of directors or initiate actions that are opposed by our then-current board of directors, including
a merger, tender offer or proxy contest involving our company. This provision could have the effect of delaying or preventing a
change of control, whether or not it is desired by or beneficial to our stockholders. Any delay or prevention of a change of control
transaction or changes in our board of directors could cause the market price of our common stock to decline.
Indemnification
Our Amended and Restated Certificate of Incorporation
and our Bylaws contain provisions permitted under the General Corporation Law of Delaware relating to the liability of directors.
The provisions eliminate, to the extent legally permissible, a director’s liability for monetary damages for a breach of
fiduciary duty, except in circumstances involving wrongful acts, such as the breach of a director’s duty of loyalty or acts
or omissions that involve intentional misconduct or a knowing violation of law. The limitation of liability described above does
not alter the liability of our directors and officers under federal securities laws. Furthermore, our Amended and Restated Certificate
of Incorporation and our Bylaws contains provisions to indemnify our directors and officers to the fullest extent permitted by
the General Corporation Law of Delaware. These provisions do not limit or eliminate our right or the right of any stockholder of
ours to seek non-monetary relief, such as an injunction or rescission in the event of a breach by a director or an officer of his
or her duty of care to us. We believe that these provisions assist us in attracting and retaining qualified individuals to serve
as directors.
DESCRIPTION OF WARRANTS
We may issue warrants for the purchase of shares
of our common stock, shares of our preferred stock or debt securities. The following description sets forth certain general terms
and provisions of the warrants that we may offer pursuant to this prospectus. The particular terms of the warrants and the extent,
if any, to which the general terms and provisions may apply to the warrants so offered will be described in the applicable prospectus
supplement.
Warrants may be issued independently or together
with other securities and may be attached to or separate from any offered securities. Each series of warrants will be issued under
a separate warrant agreement to be entered into between us and a bank or trust company, as warrant agent. The warrant agent will
act solely as our agent in connection with the warrants and will not have any obligation or relationship of agency or trust for
or with any holders or beneficial owners of warrants.
A copy of the forms of the warrant agreement
and the warrant certificate relating to any particular issue of warrants will be filed with the SEC each time we issue warrants,
and you should read those documents for provisions that may be important to you. For more information on how you can obtain copies
of the forms of the warrant agreement and the related warrant certificate, see “Where You Can Find More Information.”
Stock Warrants
The prospectus supplement relating to a particular
offering of warrants exercisable for shares of our common stock or shares of our preferred stock will describe the terms of the
common share warrants and preferred share warrants, including the following:
|
|
●
|
the title of the warrants;
|
|
|
|
|
|
|
●
|
the offering price for the warrants, if any;
|
|
|
|
|
|
|
●
|
the aggregate number of the warrants;
|
|
|
|
|
|
|
●
|
the designation and terms of the shares of common stock or shares of
preferred stock that may be purchased upon exercise of the warrants;
|
|
|
|
|
|
|
●
|
the terms for changes or adjustments to the exercise price of the warrants;
|
|
|
|
|
|
|
●
|
if applicable, the designation and terms of the securities that the
warrants are issued with and the number of warrants issued with each security;
|
|
|
●
|
if applicable, the date from and after which the warrants and any securities issued with the warrants will be separately transferable;
|
|
|
|
|
|
|
●
|
the number of shares of common stock or shares of preferred stock that may be purchased upon exercise of a warrant and the price at which the shares may be purchased upon exercise;
|
|
|
|
|
|
|
●
|
the dates on which the right to exercise the warrants commence and expire;
|
|
|
|
|
|
|
●
|
if applicable, the minimum or maximum amount of the warrants that may be exercised at any one time;
|
|
|
|
|
|
|
●
|
the currency or currency units in which the offering price, if any, and the exercise price are payable;
|
|
|
|
|
|
|
●
|
if applicable, a discussion of material United States federal income tax considerations;
|
|
|
|
|
|
|
●
|
anti-dilution provisions of the warrants, if any;
|
|
|
|
|
|
|
●
|
redemption or call provisions, if any, applicable to the warrants;
|
|
|
|
|
|
|
●
|
any additional terms of the warrants, including terms, procedures and limitations relating to the exchange and exercise of the warrants; and
|
|
|
|
|
|
|
●
|
any other information we think is important about the warrants.
|
Debt Warrants
The prospectus supplement relating to a particular
offering of warrants exercisable for debt securities will describe the terms of those warrants, including the following:
|
|
●
|
the title of the warrants;
|
|
|
|
|
|
|
●
|
the offering price for the warrants, if any;
|
|
|
|
|
|
|
●
|
the aggregate number of the warrants;
|
|
|
|
|
|
|
●
|
the designation and terms of the debt securities purchasable upon exercise
of the warrants;
|
|
|
|
|
|
|
●
|
the terms for changes or adjustments to the exercise price of the warrants;
|
|
|
|
|
|
|
●
|
if applicable, the designation and terms of the debt securities that
the warrants are issued with and the number of warrants issued with each debt security;
|
|
|
|
|
|
|
●
|
if applicable, the date from and after which the warrants and any debt
securities issued with them will be separately transferable;
|
|
|
|
|
|
|
●
|
the principal amount of debt securities that may be purchased upon
exercise of a warrant and the price at which the debt securities may be purchased upon exercise;
|
|
|
|
|
|
|
●
|
the dates on which the right to exercise the warrants will commence
and expire;
|
|
|
|
|
|
|
●
|
if applicable, the minimum or maximum amount of the warrants that may
be exercised at any one time;
|
|
|
●
|
whether the warrants represented by the warrant certificates or debt
securities that may be issued upon exercise of the warrants will be issued in registered or bearer form;
|
|
|
|
|
|
|
●
|
information relating to book-entry procedures, if any;
|
|
|
|
|
|
|
●
|
the currency or currency units in which the offering price, if any,
and the exercise price are payable;
|
|
|
|
|
|
|
●
|
if applicable, a discussion of material United States federal income
tax considerations;
|
|
|
|
|
|
|
●
|
anti-dilution provisions of the warrants, if any;
|
|
|
|
|
|
|
●
|
redemption or call provisions, if any, applicable to the warrants;
|
|
|
|
|
|
|
●
|
any additional terms of the warrants, including terms, procedures and
limitations relating to the exchange and exercise of the warrants; and
|
|
|
|
|
|
|
●
|
any other information we think is important about the warrants.
|
Exercise of Warrants
Each warrant will entitle the holder of the
warrant to purchase at the exercise price set forth in the applicable prospectus supplement the number of shares of common stock,
shares of preferred stock or the principal amount of debt securities being offered. Holders may exercise warrants at any time up
to the close of business on the expiration date set forth in the applicable prospectus supplement. After the close of business
on the expiration date, unexercised warrants are void. Holders may exercise warrants as set forth in the prospectus supplement
relating to the warrants being offered.
Until a holder exercises the warrants to purchase
our shares of common stock, shares of preferred stock or debt securities, the holder will not have any rights as a holder of our
shares of common stock, shares of preferred stock or debt securities, as the case may be, by virtue of ownership of warrants.
DESCRIPTION OF DEBT SECURITIES
The following is a general description of the
terms of debt securities we may issue from time to time unless we provide otherwise in the applicable prospectus supplement. Particular
terms of any debt securities we offer will be described in the prospectus supplement relating to such debt securities.
As required by Federal law for all bonds and
notes of companies that are publicly offered, any debt securities we issue will be governed by a document called an “indenture,”
formed of which are filed as an exhibit to the registration statement of which this prospectus forms a part.
Because this section is a summary, it does not
describe every aspect of any debt securities we may issue or the indenture governing any such debt securities. Particular terms
of any debt securities we offer will be described in the prospectus supplement relating to such debt securities, and we urge you
to read the applicable executed indenture, which will be filed with the SEC at the time of any offering of debt securities, because
it, and not this description, will define the rights of holders of such debt securities.
A prospectus supplement will describe the particular
terms of any series of debt securities we may issue, including some or all of the following:
|
|
●
|
the designation or title of the series of debt securities;
|
|
|
|
|
|
|
●
|
the total principal amount of the series of debt securities, the denominations
in which the offered debt securities will be issued and whether the offering may be reopened for additional securities of that
series and on what terms;
|
|
|
|
|
|
|
●
|
the percentage of the principal amount at which the series of debt
securities will be offered;
|
|
|
|
|
|
|
●
|
the date or dates on which principal will be payable;
|
|
|
|
|
|
|
●
|
the rate or rates (which may be either fixed or variable) and/or the
method of determining such rate or rates of interest, if any;
|
|
|
|
|
|
|
●
|
the date or dates from which any interest will accrue, or the method
of determining such date or dates, and the date or dates on which any interest will be payable;
|
|
|
|
|
|
|
●
|
the terms for redemption, extension or early repayment, if any;
|
|
|
|
|
|
|
●
|
the currencies in which the series of debt securities are issued and
payable;
|
|
|
|
|
|
|
●
|
whether the amount of payments of principal, interest or premium, if
any, on a series of debt securities will be determined with reference to an index, formula or other method and how these amounts
will be determined;
|
|
|
|
|
|
|
●
|
the place or places of payment, transfer, conversion and/or exchange
of the debt securities;
|
|
|
|
|
|
|
●
|
the provision for any sinking fund;
|
|
|
|
|
|
|
●
|
any restrictive covenants;
|
|
|
|
|
|
|
●
|
events of default;
|
|
|
|
|
|
|
●
|
whether the series of debt securities are issuable in certificated
form;
|
|
|
|
|
|
|
●
|
any provisions for legal defeasance or covenant defeasance;
|
|
|
|
|
|
|
●
|
whether and under what circumstances we will pay additional amounts
in respect of any tax, assessment or governmental charge and, if so, whether we will have the option to redeem the debt securities
rather than pay the additional amounts (and the terms of this option);
|
|
|
|
|
|
|
●
|
any provisions for convertibility or exchangeability of the debt securities
into or for any other securities;
|
|
|
|
|
|
|
●
|
whether the debt securities are subject to subordination and the terms
of such subordination;
|
|
|
|
|
|
|
●
|
any listing of the debt securities on any securities exchange;
|
|
|
|
|
|
|
●
|
if applicable, a discussion of certain U.S. Federal income tax considerations,
including those related to original issue discount, if applicable; and
|
|
|
|
|
|
|
●
|
any other material terms.
|
The debt securities may be secured or unsecured
obligations. Unless the prospectus supplement states otherwise, principal, interest and premium, if any, will be paid by us in
immediately available funds.
DESCRIPTION OF RIGHTS
The following is a general description of the
terms of the rights we may issue from time to time unless we provide otherwise in the applicable prospectus supplement. Particular
terms of any rights we offer will be described in the prospectus supplement relating to such rights.
General
We may issue rights to purchase common stock,
preferred stock, debt securities or units. Rights may be issued independently or together with other securities and may or may
not be transferable by the person purchasing or receiving the rights. In connection with any rights offering to our stockholders,
we may enter into a standby underwriting, backstop or other arrangement with one or more underwriters or other persons pursuant
to which such underwriters or other persons would purchase any offered securities remaining unsubscribed for after such rights
offering. In connection with a rights offering to our stockholders, we would distribute certificates evidencing the rights and
a prospectus supplement to our stockholders on or about the record date that we set for receiving rights in such rights offering.
The applicable prospectus supplement will describe
the following terms of any rights we may issue, including some or all of the following:
|
|
●
|
the title and aggregate number of the rights;
|
|
|
|
|
|
|
●
|
the subscription price or a formula for the determination of the subscription
price for the rights and the currency or currencies in which the subscription price may be payable;
|
|
|
|
|
|
|
●
|
if applicable, the designation and terms of the securities with which
the rights are issued and the number of rights issued with each such security or each principal amount of such security;
|
|
|
|
|
|
|
●
|
the number or a formula for the determination of the number of the
rights issued to each stockholder;
|
|
|
|
|
|
|
●
|
the extent to which the rights are transferable;
|
|
|
|
|
|
|
●
|
in the case of rights to purchase debt securities, the principal amount
of debt securities purchasable upon exercise of one right;
|
|
|
|
|
|
|
●
|
in the case of rights to purchase common stock or preferred stock,
the type of stock and number of shares of stock purchasable upon exercise of one right;
|
|
|
|
|
|
|
●
|
the date on which the right to exercise the rights will commence, and
the date on which the rights will expire (subject to any extension);
|
|
|
|
|
|
|
●
|
if applicable, the minimum or maximum amount of the rights that may
be exercised at any one time;
|
|
|
●
|
the extent to which such rights include an over-subscription privilege with respect to unsubscribed securities;
|
|
|
|
|
|
|
●
|
if applicable, the procedures for adjusting the subscription price and number of shares of common stock or preferred stock purchasable upon the exercise of each right upon the occurrence of certain events, including stock splits, reverse stock splits, combinations, subdivisions or reclassifications of common stock or preferred stock;
|
|
|
|
|
|
|
●
|
the effect on the rights of any merger, consolidation, sale or other disposition of our business;
|
|
|
|
|
|
|
●
|
the terms of any rights to redeem or call the rights;
|
|
|
|
|
|
|
●
|
information with respect to book-entry procedures, if any;
|
|
|
●
|
the terms of the securities issuable upon exercise of the rights;
|
|
|
●
|
if applicable, the material terms of any standby underwriting, backstop or other purchase arrangement that we may enter into in connection with the rights offering;
|
|
|
|
|
|
|
●
|
if applicable, a discussion of certain U.S. Federal income tax considerations; and
|
|
|
|
|
|
|
●
|
any other terms of the rights, including terms, procedures and limitations relating to the exchange and exercise of the rights.
|
Exercise of Rights
Each right will entitle the holder to purchase
for cash or other consideration such shares of stock or principal amount of securities at the subscription price as shall in each
case be set forth in, or be determinable as set forth in, the prospectus supplement relating to the rights offered thereby. Rights
may be exercised as set forth in the applicable prospectus supplement beginning on the date specified therein and continuing until
the close of business on the expiration date set forth in the prospectus supplement relating to the rights offered thereby. After
the close of business on the expiration date, unexercised rights will become void.
Upon receipt of payment and a subscription certificate
properly completed and duly executed at the corporate trust office of the subscription agent or any other office indicated in the
prospectus supplement, we will, as soon as practicable, forward the securities purchasable upon such exercise. If less than all
of the rights represented by such subscription certificate are exercised, a new subscription certificate will be issued for the
remaining rights. If we so indicate in the applicable prospectus supplement, holders of the rights may surrender securities as
all or part of the exercise price for rights.
We may determine to offer any unsubscribed offered
securities directly to stockholders, persons other than stockholders, to or through agents, underwriters or dealers or through
a combination of such methods, including pursuant to standby underwriting, backstop or other arrangements, as set forth in the
applicable prospectus supplement.
Prior to exercising their rights, holders of
rights will not have any of the rights of holders of the securities purchasable upon subscription, including, in the case of rights
to purchase common stock or preferred stock, the right to receive dividends, if any, or payments upon our liquidation, dissolution
or winding up or to exercise any voting rights or, in the case of rights to purchase debt securities, the right to receive principal,
premium, if any, or interest payments, on the debt securities purchasable upon exercise or to enforce covenants in the applicable
indenture.
DESCRIPTION OF UNITS
We may issue units comprising one or more securities
described in this prospectus in any combination. The following description sets forth certain general terms and provisions of the
units that we may offer pursuant to this prospectus. The particular terms of the units and the extent, if any, to which the general
terms and provisions may apply to the units so offered will be described in the applicable prospectus supplement.
Each unit will be issued so that the holder
of the unit also is the holder of each security included in the unit. Thus, the unit will have the rights and obligations of a
holder of each included security. Units will be issued pursuant to the terms of a unit agreement, which may provide that the securities
included in the unit may not be held or transferred separately at any time or at any time before a specified date. A copy of the
forms of the unit agreement and the unit certificate relating to any particular issue of units will be filed with the SEC each
time we issue units, and you should read those documents for provisions that may be important to you. For more information on how
you can obtain copies of the forms of the unit agreement and the related unit certificate, see “Where You Can Find More Information.”
The prospectus supplement relating to any particular
issuance of units will describe the terms of those units, including, to the extent applicable, the following:
|
|
●
|
the designation and terms of the units and the securities comprising
the units, including whether and under what circumstances those securities may be held or transferred separately;
|
|
|
|
|
|
|
●
|
any provision for the issuance, payment, settlement, transfer or exchange
of the units or of the securities comprising the units; and
|
|
|
|
|
|
|
●
|
whether the units will be issued in fully registered or global form.
|
PLAN OF DISTRIBUTION
We may sell the securities offered by this prospectus
in any one or more of the following ways from time to time:
|
|
●
|
to or through one or more underwriters, initial purchasers, brokers or dealers;
|
|
|
|
|
|
|
●
|
through agents to investors or the public;
|
|
|
|
|
|
|
●
|
in short or long transactions;
|
|
|
|
|
|
|
●
|
through put or call option transactions relating to our common stock;
|
|
|
|
|
|
|
●
|
directly to agents or other purchasers;
|
|
|
|
|
|
|
●
|
in “at the market offerings” within the meaning of Rule 415(a)(4) of the Securities Act of 1933, as amended, or the Securities Act, to or through a market maker or into an existing trading market, on an exchange or otherwise;
|
|
|
|
|
|
|
●
|
though a combination of any such methods of sale; or
|
|
|
|
|
|
|
●
|
through any other method described in the applicable prospectus supplement.
|
The applicable prospectus supplement will set
forth the terms of the offering and the method of distribution and will identify any firms acting as underwriters, initial purchasers,
dealers or agents in connection with the offering, including:
|
|
●
|
the terms of the offering;
|
|
|
|
|
|
|
●
|
the names of any underwriters, dealers or agents;
|
|
|
|
|
|
|
●
|
the name or names of any managing underwriter or underwriters;
|
|
|
|
|
|
|
●
|
the purchase price of the securities and the proceeds to us from the
sale;
|
|
|
|
|
|
|
●
|
any over-allotment options under which the underwriters may purchase
additional shares of common stock from us;
|
|
|
|
|
|
|
●
|
any underwriting discounts, concessions, commissions or agency fees
and other items constituting compensation to underwriters, dealers or agents;
|
|
|
|
|
|
|
●
|
any delayed delivery arrangements;
|
|
|
|
|
|
|
●
|
any public offering price;
|
|
|
|
|
|
|
●
|
any discounts or concessions allowed or re-allowed or paid by underwriters
or dealers to other dealers; or
|
|
|
|
|
|
|
●
|
any securities exchange or market on which the common stock offered
in the prospectus supplement may be listed.
|
If we use underwriters for a sale of securities,
the underwriters will acquire the securities for their own account for resale to the public, either on a firm commitment basis
or a best efforts basis. The underwriters may resell the securities in one or more transactions, including negotiated transactions,
at a fixed public offering price or at varying prices determined at the time of sale. Underwriters may offer the securities to
the public either through underwriting syndicates represented by one or more managing underwriters or directly by one or more firms
acting as underwriters. If an underwriter or underwriters are used in the sale of securities hereunder, an underwriting agreement
will be executed with the underwriter or underwriters at the time an agreement for sale is reached. Unless we inform you otherwise
in the applicable prospectus supplement, the obligations of the underwriters to purchase the securities will be subject to certain
conditions. We may change from time to time any public offering price and any discounts or concessions the underwriters allow or
pay to dealers.
During and after an offering through underwriters,
the underwriters may purchase and sell the securities in the open market. These transactions may include overallotment and stabilizing
transactions and purchases to cover syndicate short positions created in connection with the offering. The underwriters may also
impose a penalty bid, which means that selling concessions allowed to syndicate members or other broker-dealers for the offered
securities sold for their account may be reclaimed by the syndicate if the offered securities are repurchased by the syndicate
in stabilizing or covering transactions. These activities may stabilize, maintain or otherwise affect the market price of the offered
securities, which may be higher than the price that might otherwise prevail in the open market. If commenced, the underwriters
may discontinue these activities at any time.
Some or all of the securities that we offer
though this prospectus may be new issues of securities with no established trading market. Any underwriters to whom we sell our
securities for public offering and sale may make a market in those securities, but they will not be obligated to do so and they
may discontinue any market making at any time without notice. Accordingly, we cannot assure you of the liquidity of, or continued
trading markets for, any securities that we offer.
If dealers are used for the sale of securities,
we, or an underwriter, will sell the securities to them as principals. The dealers may then resell those securities to the public
at varying prices determined by the dealers at the time of resale. We will include in the applicable prospectus supplement the
names of the dealers and the terms of the transaction.
We may also sell the securities through agents
designated from time to time. In the applicable prospectus supplement, we will name any agent involved in the offer or sale of
the offered securities, and we will describe any commissions payable to the agent. Unless we inform you otherwise in the applicable
prospectus supplement, any agent will agree to use its reasonable best efforts to solicit purchases for the period of its appointment.
We may sell the securities directly in transactions
not involving underwriters, dealers or agents.
We may sell the securities directly to institutional
investors or others who may be deemed to be underwriters within the meaning of the Securities Act with respect to any sale of those
securities. We will describe the terms of any such sales in the prospectus supplement.
Underwriters, dealers and agents that participate
in the distribution of the securities may be underwriters as defined in the applicable securities laws and any discounts or commissions
they receive from us and any profit on their resale of the securities may be treated as underwriting discounts and commissions
under the applicable securities laws. We will identify in the applicable prospectus supplement any underwriters, dealers or agents
and will describe their compensation. We may have agreements with the underwriters, dealers and agents to indemnify them against
specified civil liabilities, including liabilities under the applicable securities laws.
Underwriters, dealers and agents may engage
in transactions with or perform services for us in the ordinary course of their businesses for which they may receive customary
fees and reimbursement of expenses.
We may use underwriters with whom we have a
material relationship. We will describe the nature of such relationship in the applicable prospectus supplement.
Under the securities laws of some states, the
securities offered by this prospectus may be sold in those states only through registered or licensed brokers or dealers.
We may enter into hedging transactions with
broker-dealers and the broker-dealers may engage in short sales of the securities in the course of hedging the positions they assume
with us, including, without limitation, in connection with distributions of the securities by those broker-dealers. We may enter
into option or other transactions with broker-dealers that involve the delivery of the securities offered hereby to the broker-dealers,
who may then resell or otherwise transfer those securities. We may also loan or pledge the securities offered hereby to a broker-dealer
and the broker-dealer may sell the securities offered hereby so loaned or upon a default may sell or otherwise transfer the pledged
securities offered hereby.
LEGAL MATTERS
The validity of the securities being offered
hereby will be passed upon for us by Morgan, Lewis & Bockius LLP.
EXPERTS
MaloneBailey, LLP, independent registered public
accounting firm, has audited our consolidated financial statements included in our Annual Report on Form 10-K for the year
ended October 31, 2020, as set forth in their report, which is incorporated by reference in this prospectus and elsewhere in
the registration statement. Our consolidated financial statements are incorporated by reference in reliance on MaloneBailey,
LLP’s report, given on their authority as experts in accounting and auditing.
WHERE YOU CAN FIND MORE INFORMATION
We file annual, quarterly and current reports,
proxy statements and other information with the SEC. Our SEC filings are available to the public over the Internet at the SEC’s
website at http://www.sec.gov. Copies of certain information filed by us with the SEC are also available on our website at www.kaivalbrands.com.
Our website is not a part of this prospectus and is not incorporated by reference in this prospectus.
INFORMATION INCORPORATED BY REFERENCE
The SEC allows us to “incorporate by reference”
into this prospectus the information contained in documents that we file with them, which means that we can disclose important
information to you by referring you to those documents. The information incorporated by reference is considered to be part of this
prospectus. Information in this prospectus supersedes information incorporated by reference that we filed with the SEC before the
date of this prospectus, while information that we file later with the SEC will automatically update and supersede prior information.
Any information so updated and superseded shall not be deemed, except as so updated and superseded, to constitute a part of this
prospectus. We incorporate by reference the documents listed below and any future filings we will make with the SEC under Sections 13(a),
13(c), 14 or 15(d) of the Securities Exchange Act of 1934, as amended, or the Exchange Act, prior to the termination of the offering.
Notwithstanding the foregoing, unless specifically stated to the contrary, none of the information that is not deemed “filed”
with the SEC, including information furnished under Items 2.02 or 7.01 of any Current Report on Form 8-K, will be incorporated
by reference into, or otherwise included in, this prospectus:
|
|
●
|
our Annual Report on Form 10-K for the
fiscal year ended October 31, 2020 filed with the SEC on February 12, 2021;
|
|
|
|
|
|
|
●
|
our Quarterly Reports on Form 10-Q for the fiscal quarters ended January
31, 2021 and April 30, 3031, filed with the SEC on March 16, 2021 and June 21, 2021, respectively;
|
|
|
|
|
|
|
●
|
our Current Reports on Form 8-K filed with
the SEC on January 12, 2021, January 15, 2021, March 18, 2021, April 21, 2021 (Item 1.01 and Exhibit 10.1 only), May 10, 2021,
May 12, 2021, July 1, 2021 (Item 1.01 and Exhibits 10.1 and 10.2 only), July 20, 2021 (Items 3.03 and 5.03 and Exhibit 3.1
only) and July 23, 2021; and
|
|
|
|
|
|
|
●
|
the description of our common stock, par
value $0.001 per share, contained in our Registration Statement on Form 8-A filed with the SEC on July 20, 2021, to register such
securities under the Exchange Act, including any amendments filed for the purpose of updating such information.
|
We make available, free of charge, through our
website our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K and amendments
to those reports filed or furnished pursuant to Section 13(a), 13(c), 14 or 15(d) of the Exchange Act as soon as reasonably
practicable after we electronically file such material with, or furnish it to, the SEC. You may also obtain, free of charge, a
copy of any of these documents (other than exhibits to these documents unless the exhibits are specifically incorporated by reference
into these documents or referred to in this prospectus) by writing or calling us at the following address and telephone number:
Kaival Brands Innovations Group, Inc.
4460 Old Dixie Highway
Grant, Florida 32949
(833) 452-4825
DISCLOSURE OF COMMISSION’S POSITION
ON INDEMNIFICATION
FOR SECURITIES ACT LIABILITY
Insofar as indemnification for liabilities arising under the
Securities Act may be permitted to directors, officers or persons controlling the Company pursuant to the foregoing provisions,
we have been informed that in the opinion of the SEC, such indemnification is against public policy as expressed in the Securities
Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment
by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful
defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities
being registered, we will, unless in the opinion of our counsel the matter has been settled by controlling precedent, submit to
a court of appropriate jurisdiction the question whether such indemnification by us is against public policy as expressed in the
Securities Act and will be governed by the final adjudication of such issue.
4,700,000 Shares of Common Stock Shares
of Common Stock and
Common Warrants to Purchase 3,525,000
Shares of Common Stock
(and Shares of Common
Stock Issuable Upon Exercise of the Common Warrants)

KAIVAL BRANDS INNOVATIONS GROUP, INC.
PROSPECTUS SUPPLEMENT
Sole Book Running Manager
Maxim Group LLC
September 29, 2021
Kaival Brands Innovations (NASDAQ:KAVL)
Historical Stock Chart
From Mar 2024 to Apr 2024
Kaival Brands Innovations (NASDAQ:KAVL)
Historical Stock Chart
From Apr 2023 to Apr 2024