Exact Sciences to Provide Mayo Clinic Patients with Individualized Cancer Tumor Information to Inform Treatment
April 04 2024 - 6:05AM
Business Wire
Initiative advances precision cancer
medicine by bringing comprehensive genomic profiling and hereditary
cancer tests to patients at the Mayo Clinic Comprehensive Cancer
Center
Exact Sciences (NASDAQ: EXAS), a leading provider of cancer
screening and diagnostic tests, today announced the launch of an
initiative with Mayo Clinic to provide comprehensive genomic
profiling and hereditary cancer tests to patients. The initiative
expands the collection of data accessible to Mayo Clinic
researchers and clinicians to advance cancer research and patient
care.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20240404198461/en/
Initiative advances precision cancer
medicine by bringing comprehensive genomic profiling and hereditary
cancer tests to patients at the Mayo Clinic Comprehensive Cancer
Center (Graphic: Business Wire)
“This collaboration supports our mission to help eradicate
cancer by preventing it, detecting it earlier, and guiding
personalized treatment,” said Kevin Conroy, chairman and chief
executive officer, Exact Sciences. “By combining the strength of
Exact Sciences' portfolio of tests with Mayo Clinic's
world-renowned medical and scientific expertise, we'll help put
cancer patients on a path to potentially more effective, targeted
therapies and better outcomes.”
Specialists at Mayo Clinic Comprehensive Cancer Center care for
more than 130,000 unique cancer patients each year at three Mayo
Clinic Comprehensive Cancer Center sites across the United States:
Rochester, Minnesota and the Upper Midwest: Phoenix and Scottsdale,
Arizona; and Jacksonville, Florida. Having access to the results
from Exact Sciences' comprehensive genomic profiling test,
OncoExTra®, and hereditary cancer test, Riskguard™, will help guide
therapeutic decisions, advance cancer research, and support the
development of new diagnostic tests and therapies for cancer
treatment.
The OncoExTra test—a comprehensive DNA and RNA-based genomic
test—provides doctors and their patients with a comprehensive
molecular picture of the patient’s cancer. The test’s clinical
report includes actionable results personalized to each patient.
The Riskguard hereditary cancer test helps patients and their
physicians understand the patients' hereditary risk of cancer.
“Having access to comprehensive genomic sequencing, using whole
exome and transcriptome sequencing for the patients we serve across
the United States and globally, allows us to precisely diagnose the
underlying mutations driving a patient's cancer and to develop an
individualized treatment plan uniquely suited for their disease,”
said Cheryl Willman, M.D., The Stephen and Barbara Slaggie
Executive Director, Mayo Clinic Comprehensive Cancer Center and the
David A. Ahlquist, M.D. Professor of Cancer Research. “Germline
genetic sequencing allows us to determine an individual's risk of
developing cancer and whether this risk has been inherited in their
family members.”
“This comprehensive sequencing data will also be integrated into
the development of large language models (LLMs) and multi-modal
artificial intelligence (AI) algorithms to predict cancer risk,
detect cancers earlier, and guide intervention. Integration of this
genomic data into our longitudinal patient healthcare records in
Mayo Clinic Cloud and Mayo Clinic Platform, will facilitate not
only the care of each individual patient, but the care of all
cancer patients now and in the future,” Dr. Willman added. “It will
also lead to new scientific and clinical discoveries by Mayo Clinic
Comprehensive Cancer Center and Mayo Clinic's Center for
Individualized Medicine.”
Patients will consent to have the tests performed and to have
the data shared with Mayo Clinic. They will also receive copies of
the results for their personal record keeping.
Exact Sciences offers patients and their health care providers
access to molecular and genomic data that may provide new insights
into the causes of cancers and help researchers develop more
effective, personalized treatments for cancer patients.
About Exact Sciences’ Precision Oncology portfolio
Exact Sciences’ Precision Oncology portfolio delivers actionable
genomic insights to inform prognosis and cancer treatment after a
diagnosis. In breast cancer, the Oncotype DX Breast Recurrence
Score® test is the only test shown to predict the likelihood of
chemotherapy benefit as well as recurrence in invasive breast
cancer. The Oncotype DX Breast Recurrence Score test is recognized
as a standard of care and is included in all major breast cancer
treatment guidelines. The OncoExTra® test applies comprehensive
tumor profiling, utilizing whole exome and whole transcriptome
sequencing, to aid in therapy selection for patients with advanced,
metastatic, refractory, relapsed, or recurrent cancer. With an
extensive test of approximately 20,000 genes and 169 introns, the
OncoExTra test is one of the most comprehensive molecular tests
available to patients today. Exact Sciences enables patients to
take a more active role in their cancer care and makes it easy for
providers to order tests, interpret results, and personalize
medicine by applying real-world evidence and guideline
recommendations. The Oncotype DX Breast Recurrence Score test and
OncoExTra test were developed, and the performance characteristics
validated by Genomic Health, Inc., a wholly-owned subsidiary of
Exact Sciences Corporation following College of American
Pathologists (CAP) and Clinical Laboratory Improvement Amendments
(CLIA) regulations. The Oncotype DX Breast Recurrence Score test is
performed at the Genomic Health Redwood City clinical laboratory
and the OncoExTra test is performed at the Genomic Health Phoenix
clinical laboratory. Exact Sciences clinical laboratories are
accredited by CAP, certified under CLIA regulations, and qualified
to perform high-complexity clinical laboratory testing. These tests
have not been cleared or approved by the US Food and Drug
Administration or other notified regulatory authority. To learn
more, visit precisiononcology.exactsciences.com.
About Exact Sciences Corp.
A leading provider of cancer screening and diagnostic tests,
Exact Sciences gives patients and health care professionals the
clarity needed to take life-changing action earlier. Building on
the success of the Cologuard® and Oncotype® tests, Exact Sciences
is investing in its pipeline to develop innovative solutions for
use before, during, and after a cancer diagnosis. For more
information, visit ExactSciences.com, follow Exact Sciences on X
(formerly known as Twitter) @ExactSciences, or find Exact Sciences
on LinkedIn and Facebook.
NOTE: Oncotype, Oncotype DX Breast Recurrence Score, and
OncoExTra are trademarks of Genomic Health, Inc., a wholly owned
subsidiary of Exact Sciences. Exact Sciences, Cologuard and
Riskguard are trademarks of Exact Sciences Corporation.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20240404198461/en/
Media (U.S.): Gisela Pedroza +1 949-468-7854
gpedroza@exactsciences.com
Media (OUS): Federico Maiardi +41 79-138-1326
fmaiardi@exactsciences.com
Investors: Nathan Harrill +1 608-535-8659
investorrelations@exactsciences.com
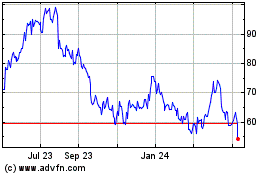
EXACT Sciences (NASDAQ:EXAS)
Historical Stock Chart
From Apr 2024 to May 2024
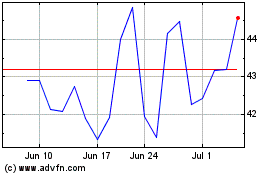
EXACT Sciences (NASDAQ:EXAS)
Historical Stock Chart
From May 2023 to May 2024