Data expands upon previously reported results,
including individual patient data, additional liver biomarkers, and
comparisons to matching arms from a contemporaneous study
Company on track to report data from ongoing
Phase 2b AHFIRM trial in 2H
2023
CUPERTINO, Calif., April 10,
2023 /PRNewswire/ -- DURECT Corporation (Nasdaq:
DRRX), a biopharmaceutical company focused on developing epigenetic
modulator programs for the treatment of acute organ injury and
chronic liver diseases, today announced that additional data from
the company's previously completed Phase 2a trial evaluating
larsucosterol in alcohol-associated hepatitis (AH) has been
accepted by the peer-reviewed journal American Journal of
Gastroenterology. In addition to previously reported
safety and efficacy data from the 19-patient, open label Phase 2a
trial, the publication includes cross-study comparisons of severe
AH patients from the Phase 2a trial with two matching comparison
arms from a contemporaneous study conducted by the DASH (Defeat
Alcoholic Steatohepatitis) Consortium. The two well-matched
comparison arms consist of an 8-patient observational arm
(Observational Arm) and a separate 16-patient steroid-treated arm
(Study-Steroid Arm). Both comparison arms received
standard-of-care including corticosteroids. The full article,
entitled "Safety, Pharmacokinetics, & Efficacy Signals of
Larsucosterol (DUR-928) in Alcohol-associated Hepatitis," can be
accessed here. Supplemental tables and figures referred to in
the article will be available in the final print publication.
"The Phase 2a study showed very promising efficacy signals of
larsucosterol in AH patients, with all patients surviving the
28-day follow-up period," said Tarek
Hassanein, M.D., Professor of Medicine at University of California San Diego Health, Medical
Director of Southern California Research Center, and the lead
author of the study. "This is an extremely encouraging result
considering that approximately a quarter of AH patients entering
hospitals don't survive longer than a month, an outcome that has
not improved in the past 50 years."
Craig McClain, M.D., Professor of
Departments of Medicine and Pharmacology & Toxicology,
Distinguished University Scholar at University
of Louisville, and co-corresponding author of the article,
commented, "The efficacy signals from this Phase 2a study compared
favorably with two matched arms of AH subjects treated with
standard of care, including steroids, from a contemporaneous study
of the DASH Consortium. We hope that this novel drug,
larsucosterol, will provide much needed treatment for a disease
process having very high morbidity and mortality for which there is
currently no FDA-approved therapy."
James E. Brown, D.V.M., President
and CEO of DURECT, added "We are excited to announce the acceptance
of this peer-reviewed publication of our Phase 2a AH trial which
provides further expert support of the data and larsucosterol's
potential for the treatment of AH patients. We look forward to
building upon this positive safety and efficacy data as we complete
our ongoing Phase 2b AHFIRM
trial. We are approaching our target enrollment of 300
patients and anticipate topline data in 2H 2023. Larsucosterol's
mechanism of action as an epigenetic modulator is different from
any other treatment previously evaluated for AH, tying directly
into the biology of the disease. We believe AHFIRM, if it is
successful, has the potential to support an NDA filing. With
FDA Fast Track Designation, our goal is to advance larsucosterol to
approval as quickly as possible in the U.S. and other regions."
Key results from the article are highlighted below:
- Survival: 100% of patients (n=19) treated with
larsucosterol, including 12 patients with severe AH, survived the
28-day follow-up period compared to a historical 28-day mortality
rate of 26%.
- Safety: Larsucosterol was well-tolerated and safe at the
three doses studied (30, 90, and 150 mg) when administered as one
or two intravenous infusions in subjects with moderate or severe
AH. The drug exposure was not affected by the disease severity and
was dose proportional.
- Time to discharge: 74% of patients treated with
larsucosterol were discharged in under 4 days after a single
dose.
- Serum bilirubin: Rapid reductions from baseline of serum
total bilirubin levels were observed at both Day 7 and Day 28 after
larsucosterol dosing, including significant reductions from
baseline in moderate AH patients at day 7 and in severe AH patients
at day 28.
- Model of End Stage Liver Disease (MELD) Score:
Reductions of MELD scores from baseline were observed at Day 7 and
Day 28. At Day 28, patients with moderate AH had statistically
significantly lower MELD scores at Day 28 and those with severe AH
had MELD scores that decreased from baseline at Day 28 but did not
achieve statistical significance.
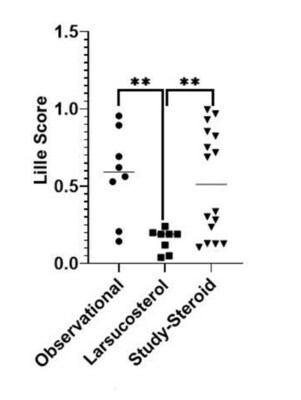
- Lille score: All 8
severe AH patients in the 30 or 90 mg dose cohorts were treatment
responders (Lille score <0.45)
and their Lille scores were
statistically lower than those of well-matched patients from the
Observational Arm and Study-Steroid Arm of the DASH Consortium
trial in a cross-study comparison (see figure).
- Liver biomarkers: Both AST and ALT enzymes decreased
rapidly in severe AH patients in the 30 or 90mg dose cohorts, with
ALT being statistically significantly lower than those in the DASH
patients in a cross-study comparison.
About the Larsucosterol AH Phase 2a Trial
Patients with moderate and severe AH were treated with
larsucosterol intravenously in this open label, dose escalation,
U.S. multi-center, Phase 2a clinical trial. Final enrollment
was 19 patients and comprised of: 8 patients (4 moderate and 4
severe) who received 30 mg, 7 patients (3 moderate and 4 severe)
who received 90 mg, and 4 patients (4 severe) who received 150 mg
of larsucosterol. The study objectives included assessment of
safety, pharmacokinetics (PK) and efficacy signals, including liver
chemistry and biomarkers.
About the AHFIRM Trial
Enrollment is ongoing in our Phase 2b randomized,
double-blind, placebo-controlled, international, multi-center study
in subjects with severe acute alcohol-associated hepatitis
(AH) to evaluate saFety and effIcacy of
laRsucosterol (DUR-928) treatMent (AHFIRM). The study
is comprised of three arms targeting enrollment of 300 total
patients, with approximately 100 patients in each arm: (1) Placebo
plus supportive care, with or without methylprednisolone capsules
at the investigators' discretion; (2) larsucosterol (30 mg); and
(3) larsucosterol (90 mg). Patients in the larsucosterol arms
receive the same supportive care without steroids. In order
to maintain blinding, patients in the two active arms receive
matching placebo capsules if the investigator prescribes steroids.
The primary outcome measure will be the 90-Day incidence of
mortality or liver transplantation for patients treated with
larsucosterol compared to those treated with placebo. DURECT is
enrolling patients at more than 60 clinical trial sites across the
U.S., EU, U.K., and Australia. Reflecting the life-threatening
nature of AH and the lack of therapeutic options, the U.S. Food and
Drug Administration (FDA) has granted larsucosterol Fast Track
Designation for the treatment of AH. We believe a positive outcome
in the AHFIRM trial could support a New Drug Application filing.
For more information, refer to ClinicalTrials.gov Identifier:
NCT04563026.
About Alcohol-associated Hepatitis (AH)
AH is an acute
form of alcohol-associated liver disease (ALD), associated with
long-term heavy intake of alcohol and often occurs after a recent
period of increased alcohol consumption (i.e., a binge). AH is
typically characterized by severe inflammation and destruction of
liver tissue (i.e., necrosis), potentially leading to
life-threatening complications including liver failure, acute
kidney injury and multi-organ failure. There are no FDA approved
therapies for AH and a retrospective analysis of 77 studies
published between 1971 and 2016, which included data from a total
of 8,184 patients, showed the overall mortality from AH was 26% at
28 days, 29% at 90 days and 44% at 180 days. A subsequent global
study published in December 2021, which included 85 tertiary
centers in 11 countries across 3 continents, prospectively enrolled
2,581 AH patients with a median Model of End-Stage Liver Disease
(MELD) score of 23.5, reported mortality at 28 and 90 days of
approximately 20% and 31%, respectively. Stopping alcohol
consumption is frequently not sufficient for recovery in many AH
patients and treatments that reduce liver inflammation, such as
corticosteroids, are limited by contraindications and have not been
shown to improve survival at 90 days or one year, and have
demonstrated an increased risk of infection. While liver
transplantation is becoming more common for ALD patients, including
AH patients, the procedure is available to a minority of AH
patients, costs exceed $875,000 on average, and patients
require lifelong immunosuppressive therapy to prevent organ
rejection.
About Larsucosterol (DUR-928)
Larsucosterol is an
endogenous sulfated oxysterol and an epigenetic modulator.
Epigenetic regulators are compounds that regulate patterns of gene
expression without modifying the DNA sequence. DNA
hypermethylation, an example of epigenetic dysregulation, results
in transcriptomic reprogramming and cellular dysfunction, and is
reportedly associated with many acute (e.g., AH) or chronic
diseases (e.g., NASH). As an inhibitor of DNA methyltransferases
(DNMT1, DNMT3a and 3b), larsucosterol inhibits DNA
hypermethylation, which subsequently modulates expression of genes
that are involved in cell signaling pathways associated with stress
responses, cell death and survival, liver regeneration, and lipid
metabolism. This may ultimately lead to improved cell survival,
reduced inflammation, and decreased lipotoxicity. As an epigenetic
modulator, the proposed mechanism of action provides further
scientific rationale for developing larsucosterol for the treatment
of certain acute organ injuries and chronic diseases.
About DURECT Corporation
DURECT is a
biopharmaceutical company committed to transforming the treatment
of acute organ injury and chronic liver diseases by advancing novel
and potentially lifesaving therapies based on its endogenous
epigenetic regulator program. Larsucosterol (also known as
DUR-928), DURECT's lead drug candidate, binds to and inhibits the
activity of DNA methyltransferases (DNMTs), DNA-modifying enzymes
that are elevated and associated with the hypermethylation found in
patients with alcohol-associated hepatitis (AH). Larsucosterol is
in clinical development for the potential treatment of AH, for
which FDA has granted a Fast Track Designation. Non-alcoholic
steatohepatitis (NASH) is also being explored. In addition,
POSIMIR® (bupivacaine solution) for infiltration use, a
non-opioid analgesic utilizing the innovative SABER®
platform technology, is FDA-approved and has been exclusively
licensed to Innocoll Pharmaceuticals for commercialization
in the United States. For
more information about DURECT, please
visit www.durect.com and follow us on
Twitter https://twitter.com/DURECTCorp.
DURECT Forward-Looking Statements
This press release contains forward-looking statements, including
statements made pursuant to the safe harbor provisions of the
Private Securities Litigation Reform Act of 1995, relating to: our
plans to complete enrollment of the AHFIRM trial in the second
quarter of 2023 and report topline data in the second half of 2023,
the potential FDA approval of larsucosterol for the treatment of
AH, the ability of a positive outcome in the AHFIRM trial to
support a New Drug Application filing, the commercialization of
POSIMIR by Innocoll, the potential to develop larsucosterol for AH,
NASH or other indications, and the potential benefits, if any, of
our product candidates. Actual results may differ materially from
those contained in the forward-looking statements contained in this
press release, and reported results should not be considered as an
indication of future performance. The potential risks and
uncertainties that could cause actual results to differ from those
projected include, among other things, the risks that the AHFIRM
trial takes longer to conduct than anticipated due to COVID-19 or
other factors, the risk that ongoing and future clinical trials of
larsucosterol do not confirm the results from earlier clinical
or pre-clinical trials, or do not demonstrate the safety or
efficacy of larsucosterol in a statistically significant
manner, the risk that the FDA or other government agencies may
require additional clinical trials for larsucosterol before
approving it for the treatment of AH even if the results of the
AHFIRM trial are successful, risks that Innocoll may not
commercialize POSIMIR successfully, and risks related to the
sufficiency of our cash resources, our anticipated capital
requirements, our need or desire for additional financing, our
ability to obtain capital to fund our operations and expenses and
our ability to continue to operate as a going concern. Further
information regarding these and other risks is included in DURECT's
most recent Securities and Exchange Commission (SEC) filings,
including its annual report on Form 10-K for the year
ended December 31, 2022 under the heading "Risk
Factors." These reports are available on our
website www.durect.com under the "Investors" tab and on
the SEC's website at www.sec.gov. All information provided in
this press release and in the attachments is based on information
available to DURECT as of the date hereof, and DURECT assumes no
obligation to update this information as a result of future events
or developments, except as required by law.
NOTE: POSIMIR® is a trademark of Innocoll
Pharmaceuticals, Ltd. in the U.S. and a trademark of DURECT
Corporation outside of the U.S. SABER® is a trademark
of DURECT Corporation. Other referenced trademarks belong
to their respective owners. Larsucosterol (DUR-928) is an
investigational drug candidate under development and has not been
approved for commercialization by the U.S. Food and Drug
Administration or other health authorities for any
indication.
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/durect-announces-peer-reviewed-article-accepted-for-publication-with-additional-data-from-previously-completed-phase-2a-study-of-larsucosterol-in-alcohol-associated-hepatitis-301792963.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/durect-announces-peer-reviewed-article-accepted-for-publication-with-additional-data-from-previously-completed-phase-2a-study-of-larsucosterol-in-alcohol-associated-hepatitis-301792963.html
SOURCE DURECT Corporation