WASHINGTON, D.C. 20549
[X] ANNUAL
REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
[_] TRANSITION
REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934
Indicate by check mark if the registrant is a well-known seasoned
issuer, as defined in Rule 405 of the Securities Act. Yes [_] No [X]
Indicate by check mark if the registrant is not required to
file reports pursuant to Section 13 or 15(d) of the Act. Yes [_] No [X]
Indicate by check mark whether the registrant
(1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding
12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such
filing requirements for the past 90 days. Yes [X] No [_]
Indicate by check mark whether the registrant
has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§
232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit
such files). Yes [X] No [_]
Indicate by check mark whether the registrant
is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth
company. See the definitions of “large accelerated filer,” “accelerated filer”, “smaller reporting
company”, and “emerging growth company” in Rule 12b-2 of the Exchange Act. (Check one)
If an emerging growth company, indicate
by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial
accounting standards provided pursuant to Section 13(a) of the Exchange Act. [_]
Indicate by check mark whether the registrant
has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial
reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared
or issued its audit report. [_]
Indicate by check mark whether the registrant is a shell company
(as defined in Rule 12b-2 of the Act). Yes [_] No [X]
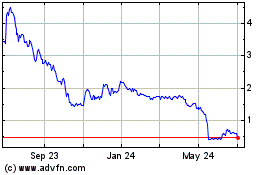
The aggregate market value of the common
stock held by non-affiliates of the registrant as of September 30, 2019 was approximately $4.5 million, computed by reference to
the closing sale price of the common stock of $3.45 per share on the Nasdaq Capital Market on September 30, 2019. Shares of common
stock held by each executive officer and director and by each person who owns 10% or more of the outstanding common stock have
been excluded in that such persons may be deemed to be affiliates. The determination of affiliate status is not necessarily a conclusive
determination for other purposes.
The number of shares of the common stock of the registrant outstanding
as of June 23, 2020 was 12,052,771.
Portions of the registrant’s proxy
statement to be filed with the Securities and Exchange Commission, or SEC, pursuant to Regulation 14A in connection with the registrant’s
2020 Annual Meeting of Stockholders, which will be filed subsequent to the date hereof, are incorporated by reference into Part
III of this annual report on Form 10-K. Such proxy statement will be filed with the SEC not later than 120 days following the end
of the registrant’s fiscal year ended March 31, 2020.
PART I
CAUTIONARY NOTICE REGARDING FORWARD LOOKING STATEMENTS
This Annual Report
on Form 10-K (the “Form 10-K”) contains “forward-looking statements” within the meaning of Section 27A
of the Securities Act of 1933, as amended (the “Securities Act”), and Section 21E of the Securities Exchange Act
of 1934, as amended, which are subject to the safe harbor created by those sections.
We may, in some cases,
use words such as “anticipate,” “believe,” “could,” “estimate,” “expect,”
“intend,” “may,” “plan,” “potential,” “predict,” “project,”
“should,” “will,” “would” or the negative of these terms, and similar expressions that convey
uncertainty of future events or outcomes to identify these forward-looking statements. Any statements contained herein that are
not statements of historical facts may be deemed to be forward-looking statements and are based upon our current expectations,
beliefs, estimates and projections, and various assumptions, many of which, by their nature, are inherently uncertain and beyond
our control. Such statements, include, but are not limited to, statements contained in this Form 10-K relating to our business,
business strategy, products and services we may offer in the future, the timing and results of future regulatory filings, the timing
and results of future clinical trials, and capital outlook. Forward-looking statements are based on our current expectations and
assumptions regarding our business, the economy and other future conditions. Because forward looking statements relate to the future,
they are subject to inherent uncertainties, risks and changes in circumstances that are difficult to predict. Our actual results
may differ materially from those contemplated by the forward-looking statements. They are neither statement of historical fact
nor guarantees of assurance of future performance. We caution you therefore against relying on any of these forward-looking statements.
Important factors that could cause actual results to differ materially from those in the forward looking statements include, but
are not limited to, a decline in general economic conditions nationally and internationally; the ability to protect our intellectual
property rights; competition from other providers and products; risks in product development; inability to raise capital to fund
continuing operations; changes in government regulation; the ability to complete capital raising transactions, and other factors
(including the risks contained in Item 1A of this Form 10-K under the heading “Risk Factors”) relating to our industry,
our operations and results of operations and any businesses that may be acquired by us. Should one or more of these risks or uncertainties
materialize, or should the underlying assumptions prove incorrect, actual results may differ significantly from those anticipated,
believed, estimated, expected, intended or planned.
Factors or events that
could cause our actual results to differ may emerge from time to time, and it is not possible for us to predict all of them, nor
can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause
actual results to differ materially from those contained in any forward-looking statements we may make. Given these uncertainties,
you should not place undue reliance on these forward-looking statements. We cannot guarantee future results, levels of activity,
performance or achievements. Except as required by applicable law, we undertake no obligation to and do not intend to update any
of the forward-looking statements to conform these statements to actual results.
ITEM 1. DESCRIPTION OF BUSINESS
Unless otherwise indicated
or the context otherwise requires, references to the “Company”, “Aethlon”, “we”, “us”
and “our” refer to Aethlon Medical, Inc., combined with its majority-owned subsidiary, Exosome Sciences, Inc.
Overview and Corporate History
We are a medical technology
company focused on developing products to diagnose and treat life and organ threatening diseases. The Aethlon Hemopurifier®
is a clinical-stage immunotherapeutic device designed to combat cancer and life-threatening viral infections. In cancer, the Hemopurifier
is designed to deplete the presence of circulating tumor-derived exosomes that promote immune suppression, seed the spread of metastasis
and inhibit the benefit of leading cancer therapies. The U.S. Food and Drug Administration, or FDA, has designated the Hemopurifier
as a “Breakthrough Device” for two independent indications:
|
|
·
|
the treatment of individuals with advanced or metastatic cancer who are either unresponsive to or intolerant of standard of care therapy, and with cancer types in which exosomes have been shown to participate in the development or severity of the disease; and
|
|
|
·
|
the treatment of life-threatening viruses that are not addressed with approved therapies.
|
We believe the Hemopurifier
can be a substantial advance in the treatment of patients with advanced and metastatic cancer through the clearance of exosomes
that promote the growth and spread of tumors through multiple mechanisms. We are currently preparing for the initiation of clinical
trials in patients with advanced and metastatic cancers. We are initially focused on the treatment of solid tumors, including head
and neck cancer, gastrointestinal cancers and other cancers. As we advance our clinical trials, we are in close contact with our
clinical sites to navigate and assess the impact of COVID-19 on our clinical trials and current timelines.
On October 4, 2019,
the FDA approved our Investigational Device Exemption, or IDE, application to initiate an Early Feasibility Study, or EFS, of
the Hemopurifier in patients with head and neck cancer in combination with standard of care pembrolizumab (Keytruda). The primary
endpoint for the EFS, which will enroll 10 to 12 subjects at a single center, will be safety, with secondary endpoints including
measures of exosome clearance and characterization, as well as response and survival rates. This study, which will be conducted
at the UPMC Hillman Cancer Center in Pittsburgh, PA, has been approved by the Institutional Review Board, or IRB and is in the
process of starting up.
We also believe the
Hemopurifier can be part of the broad-spectrum treatment of life-threatening highly glycosylated, or carbohydrate coated, viruses
that are not addressed with an already approved treatment. In small-scale or early feasibility human studies, the Hemopurifier
has been used to treat individuals infected with HIV, hepatitis-C, and Ebola.
Additionally, in-vitro,
the Hemopurifier has been demonstrated to capture Zika virus, Lassa virus, MERS-CoV, cytomegalovirus, Epstein-Barr virus, Herpes
simplex virus, Chikungunya virus, Dengue virus, West Nile virus, smallpox-related viruses, H1N1 swine flu virus, H5N1 bird flu
virus, and the reconstructed Spanish flu virus of 1918. In several cases, these validations were conducted in collaboration with
leading government or non-government research institutes.
We are also the majority
owner of Exosome Sciences, Inc., or ESI, a company focused on the discovery of exosomal biomarkers to diagnose and monitor life-threatening
diseases. Included among ESI’s activities is the advancement of a TauSomeTM biomarker candidate to diagnose chronic
traumatic encephalopathy, or CTE, in the living. ESI previously documented TauSome levels in former NFL players to be nine times
higher than same age-group control subjects. Through ESI, we are also developing exosome based biomarkers in patients with, or
at risk for, a number of cancers. We consolidate ESI’s activities in our consolidated financial statements.
Successful outcomes
of human trials will also be required by the regulatory agencies of certain foreign countries where we plan to sell the Hemopurifier.
Some of our patents may expire before FDA approval or approval in a foreign country, if any, is obtained. However, we believe that
certain patent applications and/or other patents issued more recently will help protect the proprietary nature of the Hemopurifier
treatment technology.
In addition to the
foregoing, we are monitoring closely the impact of the COVID-19 global pandemic on our business and have taken steps designed to
protect the health and safety of our employees while continuing our operations. Given the level of uncertainty regarding the duration
and impact of the COVID-19 pandemic on capital markets and the U.S. economy, we are unable to assess the impact of the worldwide
spread of SARS-CoV-2 and the resulting COVID-19 pandemic on our timelines and future access to capital. We are continuing to monitor
the spread of COVID-19 and its potential impact on our operations. The full extent to which the COVID-19 pandemic will impact our
business, results of operations, financial condition, clinical trials, and preclinical research will depend on future developments
that are highly uncertain, including actions taken to contain or treat COVID-19 and their effectiveness, as well as the economic
impact on national and international markets.
We were formed on March
10, 1999. Our executive offices are located at 9635 Granite Ridge Drive, Suite 100, San Diego, California 92123. Our telephone
number is (858) 459-7800. Our website address is www.aethlonmedical.com. All references
to “us” or “we” are references to Aethlon Medical, Inc.
The Mechanism of the Hemopurifier
The Hemopurifier is
an affinity hemofiltration device designed for the single-use removal of exosomes and life-threatening viruses from the human circulatory
system. In the United States, the Hemopurifier is classified as a combination product whose regulatory jurisdiction is The Center
for Devices and Radiological Health, or CDRH, the branch of FDA responsible for the premarket approval of all medical devices.
In application, our
Hemopurifier can be used on the established infrastructure of continuous renal replacement therapy, or CRRT, and dialysis instruments
located in hospitals and clinics worldwide. It could also potentially be developed as part of a proprietary closed system with
its own pump and tubing set, negating the requirement for dialysis infrastructure. Incorporated within the Hemopurifier is a protein
called a lectin that binds to a glycosylated, or sugar substituted, membrane, which exosomes and most infectious viruses share.
The Hemopurifier - Clinical Trials In Viral Infections
The initial development
of the Hemopurifier was focused on viral infections. In non-clinical bench experiments using a laboratory version of the Hemopurifier,
performed in Company labs as well as multiple other outside labs including the Centers for Disease Control, or CDC, the United
States Army Medical Research Institute of Infectious Diseases, or USAMRIID, Battelle Memorial Research Institute and others, we
have demonstrated that the mini-Hemopurifier can bind and clear multiple different glycosylated, or containing sugar molecules
on their membranes, viruses. These viruses include HIV, hepatitis C, or HCV, Dengue, West Nile, multiple strains of influenza,
Ebola, Chikungunya, multiple herpes viruses, a MERS-CoV related pseudovirus and others.
Initial clinical trials
on the Hemopurifier were conducted overseas on dialysis patients with HCV, with a subsequent Early Feasibility Study conducted
in the U.S. under an FDA approved Investigational Device Exemption, or IDE.
On March 13, 2017,
we concluded an FDA-approved early feasibility study under an IDE in end stage renal disease patients on dialysis who were infected
with HCV. The study was conducted at DaVita MedCenter Dialysis in Houston, Texas. We reported that there were no device-related
adverse events in enrolled subjects who met the study inclusion-exclusion criteria. We also reported that an average capture of
154 million copies of HCV (in International Units, I.U.) within the Hemopurifier during four-hour treatments. Prior to this approval,
we collected supporting Hemopurifier data through investigational human studies conducted overseas.
SARS-CoV-2/COVID-19
SARS-COV-2, the causative agent of COVID-19
is a member of the coronavirus family which includes the original SARS virus, SARS-CoV, and the MERS virus. SARS-CoV-2 like all
coronaviruses is glycosylated which suggests that the Hemopurifier could potentially clear it from biologic fluids including blood.
While we have not tested the ability of the laboratory version of the Hemopurifier to clear live SARS-CoV-2 virus for safety reasons,
we have demonstrated in our labs that the laboratory version of the Hemopurifier clears substantial quantities of the protein that
binds to cells allowing viral entry. Based on this observation, along with our in vitro data with a MERS related virus we
believe the Hemopurifier could potentially clear SARS-CoV-2 from the bloodstream. While COVID-19 initially targets the respiratory
tract, accumulating evidence suggests that virus circulating in the bloodstream may be associated with more severe disease. These
observations suggest that the Hemopurifier could have a role in treating severely affected patients during the current pandemic.
On June 17, 2020,
the FDA approved a supplement to the Company’s open IDE for the Company’s Hemopurifier in viral disease to allow for
the testing of the Hemopurifier in patients with SARS-CoV-2/COVID-19 in a New Feasibility Study. That
study’s plan is to enroll up to 40 subjects at up to 20 centers in the U.S. Subjects will have established laboratory
diagnosis of COVID-19, be admitted to an intensive care unit, orICU, and will have acute lung injury and/or severe or life threatening
disease among other criteria. Endpoints for this study, in addition to safety, will include reduction in circulating virus as
well as clinical outcomes.
The Hemopurifier – Clinical Trials Conducted Overseas
in Viral Infections
EBOLA Virus
In December of 2014,
Time Magazine named the Hemopurifier a “Top 25 Invention” as the result of treating an Ebola-infected physician at
Frankfurt University Hospital in Germany. The physician was comatose with multiple organ failure at the time of treatment with
the Hemopurifier. At the American Society of Nephrology Annual Meeting, Dr. Helmut Geiger, Chief of Nephrology at Frankfurt University
Hospital reported that the patient received a single 6.5 hour Hemopurifier treatment. Prior to treatment, viral load was measured
at 400,000 copies/ml. Post-treatment viral load reported to be at 1,000 copies/ml. Dr. Geiger also reported that 242 million copies
of Ebola virus were captured within the Hemopurifier during treatment. The patient ultimately made a full recovery. Based on this
experience, the Company filed an Expanded Access protocol with the FDA to treat Ebola virus infected patients in up to ten centers
in the U.S. and a corresponding protocol was approved by HealthCanada. These protocols remain open allowing Hemopurifier treatment
to be offered to patients presenting for care in both countries. In 2018, we applied for and were granted a Breakthrough Designation
by the FDA “… for the treatment of life-threatening viruses that are not addressed with approved therapies.”
Hepatitis C Virus (HCV)
Prior to FDA approval
of the IDE feasibility study, we conducted investigational HCV treatment studies at the Apollo Hospital, Fortis Hospital and the
Medanta Medicity Institute in India. In the Medanta Medicity Institute study, twelve HCV-infected individuals were enrolled to
receive three six-hour Hemopurifier treatments during the first three days of a 48-week peginterferon+ribavirin treatment regimen.
The study was conducted under the leadership of Dr. Vijay Kher. Dr. Kher’s staff reported that Hemopurifier therapy was well
tolerated and without device-related adverse events in the twelve treated patients.
Of these twelve patients,
ten completed the Hemopurifier-peginterferon+ribavirin treatment protocol, including eight genotype-1 patients and two genotype-3
patients. Eight of the ten patients achieved a sustained virologic response, which is the clinical definition of treatment cure
and is defined as undetectable HCV in the blood 24 weeks after the completion of the 48-week peginterferon+ribavirin drug regimen.
Both genotype-3 patients achieved a sustained virologic response, while six of the eight genotype-1 patients achieved a sustained
virologic response, which defines a cure of the infection.
Hemopurifier - Human Immunodeficiency Virus (HIV)
In addition to treating
Ebola and HCV-infected individuals, we also conducted a single proof-of-principle treatment study at the Sigma New Life Hospital
in an AIDS patient who was not being administered HIV antiviral drugs. In the study, viral load was reduced by 93% as the result
of 12 Hemopurifier treatments (each four hours in duration) that were administered over the course of one month.
The Hemopurifier in Cancer
While hepatitis C is
no longer a major commercial opportunity in developed markets due to the wide availability of curative, oral direct acting anti-viral
agents, or DAAs, we continue to investigate potential viral targets for the Hemopurifier. Recently, however, our primary focus
has been on the evaluation of the Hemopurifier in cancer, where we have shown in non-clinical studies that it is capable of clearing
exosomes, which are subcellular particles that are secreted by both normal and malignant cells. Tumor derived exosomes, have been
shown in multiple laboratories to be critical components in the progression of cancers. They can mediate resistance to chemotherapy,
resistance to targeted agents such as trastuzumab (Herceptin), metastasis and resistance to the newer immuno-oncology agents, such
as pembrolizumab (Keytruda). Based on these observations and data, in November 2019 the FDA granted us a second Breakthrough Designation
“…for the treatment of individuals with advanced or metastatic cancer who are either unresponsive to or intolerant
of standard of care therapy, and with cancer types in which exosomes have been shown to participate in the development or severity
of the disease.”
In June 2019, we met
with the FDA in Bethesda, Maryland to discuss the development program for the Hemopurifier in cancer. Following this meeting,
in September 2019, we filed an IDE to support initiating an Early Feasibility Study, or EFS, to investigate the Hemopurifier in
patients with advanced and/or metastatic squamous cell carcinoma of the head and neck in combination with pembrolizumab (Keytruda)
which was recently approved in the front line setting. The IDE was approved on October 4, 2019. We are now preparing to initiate
the trial, which will enroll 10 to 12 subjects at the UPMC Hillman Cancer Center in Pittsburgh. The trial has received IRB approval
and we expect it to open for enrollment in the September quarter. Endpoints for the trial will include safety, clearance and characterization
of cleared exosomes and clinical tumor response and survival. We are in contact with the UPMC Hillman Cancer Center to navigate
and assess the impact, if any, of COVID-19 on our trial and current timelines.
Exosome Sciences, Inc. – Majority Owned Biomarker Discovery
Company
We are the majority
owner of Exosome Sciences, Inc., or ESI, a company focused on the discovery of exosomal biomarkers to diagnose and monitor life-threatening
disease conditions that may be current or future therapeutic targets for Aethlon Medical. At present, the priority of ESI is directed
toward exosomal biomarkers to diagnose and monitor cancer and neurological disorders.
Since it began operations
in 2013, ESI researchers disclosed the discovery of an exosomal biomarker that may be associated with neurodegenerative diseases
that involve the abnormal accumulation of tau protein in the brain. These diseases, known as tauopathies, are a family of 21 different
neurological disorders that include Alzheimer’s disease and Chronic Traumatic Encephalopathy, or CTE. Related to CTE,
the ESI team was invited to participate in an NIH-funded research study with The Boston University CTE Center. In the study, ESI
researchers investigated an exosomal tau biomarker, or TauSome, as a candidate to diagnose and monitor CTE in living individuals.
At the present time, CTE can only be diagnosed through post-mortem brain autopsy.
The results of the
study indicated that TauSome levels in the blood of former professional American football players, a high CTE risk group, were
significantly higher as compared to same-age group control subjects who did not participate in activities that involved repetitive
head trauma. Additionally, high TauSome levels also correlated with poor performance in cognitive decline testing. These results
were published in an article entitled “Preliminary Study of Plasma Exosomal Tau as a Potential Biomarker for Chronic Traumatic
Encephalopathy” in the Journal of Alzheimer’s Disease on April 12, 2016.
To further validate
these observations, ESI has initiated a follow-on study to evaluate TauSome levels in up to 200 former professional football players
and control subjects. If fully enrolled, the study would be the largest study to date related to the advancement of a candidate
biomarker to diagnose and monitor CTE in the living. Enrollment of study participants began in March 2018 at the Translational
Genomics Research Institute, or TGEN, in Phoenix, AZ. Kendall Van Keuren-Jensen, Ph.D., Co-Director of TGEN’s Center for
Noninvasive Diagnostics is the principal investigator at this site location. Dr. Van Keuren-Jensen is neurodegenerative disease
thought leader whose research includes discovery and detection of biomarkers for central nervous system disorders. Additional site
locations are anticipated.
In September 2019,
we announced that ESI had entered into a collaboration with the Hoag Hospital Presbyterian in Newport Beach, California to identify
and characterize potential early disease markers for cancer diagnostics, cancer progression and treatment resistance. The Principal
Investigator on this study is Michael Demeure, M.D., program director of Precision Medicine at Hoag. Samples from patients at Hoag
will be analyzed by ESI scientists to identify and characterize exosomal “liquid biopsy” markers of cancer incidence
and progression. We believe that our recently announced NCI-SBIR Phase II contract to develop a benchtop instrument to isolate
and characterize exosomes could substantially expand the capabilities of the ESI programs.
U.S. GOVERNMENT CONTRACTS
We have entered into the following three contracts/grants with
the National Cancer Institute, or NCI, part of the NIH over the past two years:
Phase 2 Melanoma Cancer Contract
On September 12, 2019,
the NCI awarded to us a Small Business Innovation Research, or SBIR, Phase II Award Contract, for NIH/NCI Topic 359, entitled “A
Device Prototype for Isolation of Melanoma Exosomes for Diagnostics and Treatment Monitoring”, referred to as the Award Contract.
The Award Contract amount is $1,860,561 and runs for the period from September 16, 2019 through September 15, 2021.
The work to be performed
pursuant to this Award Contract will focus on melanoma exosomes. This work follows from our completion of a Phase I contract for
the Topic 359 solicitation that ran from September 2017 through June 2018 (see Phase 1 Melanoma Cancer Contract below). Following
on the Phase I work, the deliverables in the Phase II program involve the design and testing of a pre-commercial prototype of a
more advanced version of the exosome isolation platform.
During the fiscal year
ended March 31, 2020, we recognized $620,187 in government contract revenue under this contract as a result of the work involved
completing the first three milestones in the project as reported in the kick-off presentation to the NCI and the first and second
quarterly reports. The kick-off presentation covered the Company's organization and project status, recent achievements, the status
of the field, the status of commercial and academic competitors, where the proposed project was positioned against the state of
the art, the IP landscape, a refresher on the proposed technology, the detailed plan for the first budget period of the contract
and technical risks and alternative approaches. The first and second quarterly reports covered a summary of technical objectives,
a description of activities accomplished in the quarter, an analysis of experimental data, comments regarding the timeliness of
performance, and a brief explanation of activities to be pursued in the following quarter.
Phase 1 Melanoma Cancer Contract
We entered into a contract
with the NCI in September 2017. This award was under the NIH’s SBIR program. The title of the award was “SBIR Topic
359 Phase 1 Device Strategy for Selective Isolation of Oncosomes and Non-Malignant Exosomes.” The award from NIH was a firm,
fixed-price contract with potential total payments to us of $299,250 over the course of nine months.
Fixed price contracts
require the achievement of multiple, incremental milestones to receive the full award during each period of the contract. The NIH
also had the unilateral right to require us to perform additional work under an option period for an additional fixed amount of
$49,800. Under the terms of the contract, we were required to perform certain incremental work toward the achievement of specific
milestones against which we invoiced the government for fixed payment amounts. The Phase 1 Melanoma Cancer Contract was completed
in June 2018.
In the fiscal year
ended March 31, 2019, we performed work under the contract covering the remainder of the technical objectives of the contract (Aim
1: To validate the Hemopurifier as a device for capture and recovery of melanoma exosomes from plasma, and Aim 2: To validate a
method of melanoma exosome isolation consisting of the Hemopurifier followed by mab-based immunocapture to select out the tumor-derived
exosomes from non-malignant exosomes, and Aim 3: To evaluate the functional integrity of melanoma exosomes purified by the Hemopurifier
and immunocapture isolation steps). As a result we invoiced NIH for $149,625 during the fiscal year ended March 31, 2019. The Melanoma
Cancer Contract is now completed.
Breast Cancer Grant
In September 2018,
the NCI awarded us a government grant (number 1R43CA232977-01). The title of this SBIR Phase I grant is “The Hemopurifier
Device for Targeted Removal of Breast Cancer Exosomes from the Blood Circulation.”
This NCI Phase I grant
period originally ran from September 14, 2018 through August 31, 2019. In August 2019, we applied for and received a no cost, twelve
month extension on this grant, so the expiration date was extended to August 31, 2020. The total amount of the firm grant is $298,444.
The grant calls for two subcontractors to work with us. Those subcontractors are University of Pittsburgh and Massachusetts General
Hospital.
During the fiscal year ended March 31, 2020,
we recognized $30,000 in government contract revenue under this grant as a result of the work involved in one of the three technical
objectives of the contract (Aim 2. “Elution of a population of breast cancer exosomes from Hemopurifier cartridges that bear
the signatures of malignancy based on expression of CSPG4 and HER2, for triple-negative or HER2-overexpressing cancers, respectively”).
We also invoiced the NCI for an additional $100,000 during the fiscal year ended March 31, 2020 in order to pay our subcontractors
under the contract. As we did not complete any additional technical objectives beyond Aim 2 noted above during the period, we recorded
this $100,000 as deferred revenue as of March 31, 2020.
During the fiscal year
ended March 31, 2019, we recognized $80,000 in government contract revenue under this grant as a result of the work involved in
completing one of the three technical objectives of the contract (Aim 1. “To evaluate Hemopurifier-mediated capture of breast
cancer exosomes”).
Research and Development Costs
A substantial portion
of our operating budget is used for research and development activities. The cost of research and development, all of which has
been charged to operations, amounted to approximately $927,000 and $896,000 in the fiscal years ended March 31, 2020 and 2019,
respectively.
Intellectual Property
We currently own or
have license rights to a number of U.S. and foreign patents and patent applications and endeavor to continually improve our intellectual
property position. We consider the protection of our technology, whether owned or licensed, to the exclusion of use by others,
to be vital to our business. While we intend to focus primarily on patented or patentable technology, we may also rely on trade
secrets, unpatented property, know-how, regulatory exclusivity, patent extensions and continuing technological innovation to develop
our competitive position. We also own certain trademarks.
Our success depends
in large part on our ability to protect our proprietary technology, including the Hemopurifier product platform, and to operate
without infringing the proprietary rights of third parties. We rely on a combination of patent, trade secret, copyright and trademark
laws, as well as confidentiality agreements, licensing agreements and other agreements, to establish and protect our proprietary
rights. Our success also depends, in part, on our ability to avoid infringing patents issued to others. If we were judicially determined
to be infringing on any third-party patent, we could be required to pay damages, alter our products or processes, obtain licenses
or cease sales of products or certain activities.
To protect our proprietary
medical technologies, including the Hemopurifier product platform and other scientific discoveries, we have a portfolio of over
50 issued patents and pending applications worldwide. We currently have five issued U.S. patents and 33 issued patents in countries
outside of the United States. In addition, we have 17 patent applications worldwide related to our Hemopurifier product platform
and other technologies. We are seeking additional patents on our scientific discoveries. In 2020, we filed several provisional
patent applications related to our products and technologies.
It is possible that
our pending patent applications may not result in issued patents, that we will not develop additional proprietary products that
are patentable, that any patents issued to us may not provide us with competitive advantages or will be challenged by third parties
and that the patents of others may prevent the commercialization of products incorporating our technology. Furthermore, others
may independently develop similar products, duplicate our products or design around our patents. U.S. patent applications are not
immediately made public, so it is possible that a third party may obtain a patent on a technology we are actively using.
There
is a risk that any patent applications that we file and any patents that we hold or later obtain could be challenged by third parties
and declared invalid or unenforceable. For many of our pending applications, patent interference proceedings may be instituted
with the U.S. Patent and Trademark Office, or the USPTO when more than one person files a patent application covering the same
technology, or if someone wishes to challenge the validity of an issued patent. At the completion of the interference proceeding,
the USPTO will determine which competing applicant is entitled to the patent, or whether an issued patent is valid. Patent interference
proceedings are complex, highly contested legal proceedings, and the USPTO’s decision is subject to appeal. This means that
if an interference proceeding arises with respect to any of our patent applications, we may experience significant expenses and
delay in obtaining a patent, and if the outcome of the proceeding is unfavorable to us, the patent could be issued to a competitor
rather than to us. Third parties can file post-grant proceedings in the USPTO, seeking to have issued patent invalidated,
within nine months of issuance. This means that patents undergoing post-grant proceedings may be lost, or some or all claims may
require amendment or cancellation, if the outcome of the proceedings is unfavorable to us. Post-grant proceedings are complex and
could result in a reduction or loss of patent rights. The institution of post-grant proceedings against our patents could also
result in significant expenses.
Patent law outside
the United States is uncertain and in many countries, is currently undergoing review and revisions. The laws of some countries
may not protect our proprietary rights to the same extent as the laws of the United States. Third parties may attempt to oppose
the issuance of patents to us in foreign countries by initiating opposition proceedings. Opposition proceedings against any of
our patent filings in a foreign country could have an adverse effect on our corresponding patents that are issued or pending in
the United States. It may be necessary or useful for us to participate in proceedings to determine the validity of our patents
or our competitors’ patents that have been issued in countries other than the United States. This could result in substantial
costs, divert our efforts and attention from other aspects of our business, and could have a material adverse effect on our results
of operations and financial condition. Outside of the United States, we currently have pending patent applications or issued patents
in Europe, India, Russia, Australia, Canada and Hong Kong.
In addition to patent
protection, we rely on unpatented trade secrets and proprietary technological expertise. It is possible that others could independently
develop or otherwise acquire substantially equivalent technology, somehow gain access to our trade secrets and proprietary technological
expertise or disclose such trade secrets, or that we may not successfully ultimately protect our rights to such unpatented trade
secrets and proprietary technological expertise. We rely, in part, on confidentiality agreements with our marketing partners, employees,
advisors, vendors and consultants to protect our trade secrets and proprietary technological expertise. We cannot assure you that
these agreements will not be breached, that we will have adequate remedies for any breach or that our unpatented trade secrets
and proprietary technological expertise will not otherwise become known or be independently discovered by competitors.
Patents
The following table lists our issued patents
and patent applications, including their ownership status:
Patents Issued in the United States
|
PATENT #
|
PATENT NAME
|
ISSUANCE
DATE
|
OWNED OR
LICENSED
|
EXPIRATION
DATE
|
|
9,707,333
|
Extracorporeal removal of microvesicular particles
|
7/18/17
|
Owned
|
1/6/29
|
|
9,364,601
|
Extracorporeal removal of microvesicular particles
|
6/14/16
|
Owned
|
10/2/29
|
|
8,288,172
|
Extracorporeal removal of microvesicular particles
|
10/16/12
|
Owned
|
3/30/29
|
|
7,226,429
|
Method for removal of viruses from blood by lectin affinity hemodialysis
|
6/5/07
|
Owned
|
1/20/24
|
|
10,022,483
|
Method for removal of viruses from blood by lectin affinity hemodialysis
|
7/17/18
|
Owned
|
1/20/24
|
Patent Applications Pending in the United
States
|
APPLICATION #
|
APPLICATION NAME
|
FILING
DATE
|
OWNED OR
LICENSED
|
|
16/415,713
|
Affinity capture of circulating biomarkers
|
5/17/19
|
Owned
|
|
16/506,864
|
Brain specific exosome based diagnostics and extracorporeal therapies
|
7/09/19
|
Owned
|
|
16/036,608
|
Method for removal of viruses from blood by lectin affinity hemodialysis
|
1/16/18
|
Owned
|
|
16/459,220
|
Methods and compositions for quantifying exosomes
|
7/01/19
|
Owned
|
|
15/777,168
|
Plasma exosomal tau as a biomarker for chronic traumatic encephalopathy
|
5/17/18
|
Owned
|
|
16/636,297
|
Multiplex cerebrospinal fluid processing system
|
2/03/20
|
Owned
|
|
Provisional
|
Devices and Methods for Mediating SARS-COV-2 (COVID-19)
|
Filed
|
Owned
|
|
Provisional
|
Methods of improving the efficacy of immune checkpoint inhibitors
|
Filed
|
Owned
|
|
Provisional
|
Methods of improving the efficacy of monoclonal antibodies
|
Filed
|
Owned
|
|
Provisional
|
Systems and methods for capturing circulating exosomes
|
Filed
|
Owned
|
Foreign Patents
|
PATENT #
|
PATENT NAME
|
ISSUANCE
DATE
|
OWNED OR
LICENSED
|
EXPIRATION
DATE
|
|
3110977
|
Brain specific exosome based diagnostics and extracorporeal therapies (Denmark)
|
5/16/18
|
Owned
|
9/12/36
|
|
3110977
|
Brain specific exosome based diagnostics and extracorporeal therapies (France)
|
5/16/18
|
Owned
|
9/12/36
|
|
3110977
|
Brain specific exosome based diagnostics and extracorporeal therapies (Germany)
|
5/16/18
|
Owned
|
9/12/36
|
|
3110977
|
Brain specific exosome based diagnostics and extracorporeal therapies (Ireland)
|
5/16/18
|
Owned
|
9/12/36
|
|
3110977
|
Brain specific exosome based diagnostics and extracorporeal therapies (Great Britain)
|
5/16/18
|
Owned
|
9/12/36
|
|
3110977
|
Brain specific exosome based diagnostics and extracorporeal therapies (Sweden)
|
5/16/18
|
Owned
|
9/12/36
|
|
3110977
|
Brain specific exosome based diagnostics and extracorporeal therapies (Netherlands)
|
5/16/18
|
Owned
|
9/12/36
|
|
3110977
|
Brain specific exosome based diagnostics and extracorporeal therapies (Switzerland)
|
5/16/18
|
Owned
|
9/12/36
|
|
2353399
|
Method for removal of viruses from blood by lectin affinity hemodialysis (Russia)
|
4/27/09
|
Owned
|
1/20/24
|
|
1624785
|
Method for removal of viruses from blood by lectin affinity hemodialysis (Belgium)
|
7/17/13
|
Owned
|
1/20/24
|
|
1624785
|
Method for removal of viruses from blood by lectin affinity hemodialysis (Ireland)
|
7/17/13
|
Owned
|
1/20/24
|
|
1624785
|
Method for removal of viruses from blood by lectin affinity hemodialysis (Italy)
|
7/17/13
|
Owned
|
1/20/24
|
|
1624785
|
Method for removal of viruses from blood by lectin affinity hemodialysis (Great Britain)
|
7/17/13
|
Owned
|
1/20/24
|
|
1624785
|
Method for removal of viruses from blood by lectin affinity hemodialysis (France)
|
7/17/13
|
Owned
|
1/20/24
|
|
1624785
|
Method for removal of viruses from blood by lectin affinity hemodialysis (Germany)
|
7/17/13
|
Owned
|
1/20/24
|
|
2516403
|
Method for removal of viruses from blood by lectin affinity hemodialysis (Canada)
|
8/12/14
|
Owned
|
1/20/24
|
|
2591359
|
Methods for quantifying exosomes (Germany)
|
3/01/17
|
Owned
|
7/07/31
|
|
2591359
|
Methods for quantifying exosomes (France)
|
3/01/17
|
Owned
|
7/07/31
|
|
2591359
|
Methods for quantifying exosomes (Great Britain)
|
3/01/17
|
Owned
|
7/07/31
|
|
2591359
|
Methods for quantifying exosomes (Spain)
|
3/01/17
|
Owned
|
7/07/31
|
|
2644855
|
Extracorporeal removal of microvesicular particles (Canada)
|
11/19/19
|
Owned
|
1/20/24
|
|
1993600
|
Extracorporeal removal of microvesicular particles (Germany)
|
4/24/19
|
Owned
|
1/20/24
|
|
1993600
|
Extracorporeal removal of microvesicular particles (Switzerland)
|
4/24/19
|
Owned
|
1/20/24
|
|
1993600
|
Extracorporeal removal of microvesicular particles (Spain)
|
4/24/19
|
Owned
|
1/20/24
|
|
1993600
|
Extracorporeal removal of microvesicular particles (France)
|
4/24/19
|
Owned
|
1/20/24
|
|
1993600
|
Extracorporeal removal of microvesicular particles (Great Britain)
|
4/24/19
|
Owned
|
1/20/24
|
|
1993600
|
Extracorporeal removal of microvesicular particles (Italy)
|
4/24/19
|
Owned
|
1/20/24
|
|
1993600
|
Extracorporeal removal of microvesicular particles (Netherlands)
|
4/24/19
|
Owned
|
1/20/24
|
|
1993600
|
Extracorporeal removal of microvesicular particles (Sweden)
|
4/24/19
|
Owned
|
1/20/24
|
|
3366784
|
Brain specific exosome based diagnostics and extracorporeal therapies (Denmark)
|
11/13/19
|
Owned
|
9/12/36
|
|
3366784
|
Brain specific exosome based diagnostics and extracorporeal therapies (France)
|
11/13/19
|
Owned
|
9/12/36
|
|
3366784
|
Brain specific exosome based diagnostics and extracorporeal therapies (Germany)
|
11/13/19
|
Owned
|
9/12/36
|
|
3366784
|
Brain specific exosome based diagnostics and extracorporeal therapies (Ireland)
|
11/13/19
|
Owned
|
9/12/36
|
Foreign Patent Applications
|
APPLICATION #
|
APPLICATION NAME
|
FILING DATE
|
OWNED OR LICENSED
|
|
DE 112016001400.7
|
Methods of delivering regional citrate anticoagulation (RCA) during extracorporeal blood treatments
|
10/23/17
|
Owned
|
|
EP19161598.8
|
Extracorporeal removal of microvesicular particles (exosomes) (Europe)
|
3/8/19
|
Owned
|
|
9104740.6
|
Extracorporeal removal of microvesicular particles (exosomes) (Hong Kong)
|
3/9/07
|
Owned
|
|
8139/DELNP/2008
|
Extracorporeal removal of microvesicular particles (exosomes) (India)
|
3/9/07
|
Owned
|
|
3061952
|
Extracorporeal removal of microvesicular particles (Canada)
|
11/18/19
|
Owned
|
|
2939652
|
Brain specific exosome based diagnostics and extracorporeal therapies (Canada)
|
8/12/06
|
Owned
|
|
18166085.3
|
Brain specific exosome based diagnostics and extracorporeal therapies (Europe)
|
4/6/18
|
Owned
|
|
16867003.2
|
Plasma exosomal tau as a biomarker for chronic traumatic encephalopathy
|
11/16/16
|
Owned
|
International Patent Applications
|
APPLICATION #
|
APPLICATION NAME
|
FILING
DATE
|
OWNED OR
LICENSED
|
|
PCT/US2018/
044576
|
Multiplex cerebrospinal fluid processing system
|
7/31/18
|
Owned
|
Licensing and Assignment Agreements
On November 7, 2006,
we executed an assignment agreement with the London Health Science Center Research, Inc. under which an invention and related patent
rights for a method to treat cancer were assigned to us. The invention provides for the "Extracorporeal removal of microvesicular
particles" for which the U.S. Patent and Trademark Office allowed a patent (Patent NO.8,288,172) in the U.S. as of October
2012. The agreement provided for an upfront payment of 800 shares of unregistered common stock and a 2% royalty on any future net
sales of all products or services, the sale of which would infringe in the absence of the assignment granted under this agreement.
We are also responsible for paying certain patent application and filing costs. Under the assignment agreement, we own the patents
until their expiration in May 2029. Under certain circumstances, ownership of the patents may revert to the London Health Science
Center Research, Inc. if there is an uncured substantial breach of the assignment agreement.
Industry & Competition
The industry for treating
infectious disease and cancer is extremely competitive, and companies developing new treatment procedures face significant capital
and regulatory challenges. As our Hemopurifier is a clinical-stage device, we have the additional challenge of establishing medical
industry support, which will be driven by treatment data resulting from human clinical studies. Should our device become market
cleared by FDA or the regulatory body of another country, we may face significant competition from well-funded pharmaceutical organizations.
Additionally, we would likely need to establish large-scale production of our device in order to be competitive. We believe that
our Hemopurifier is a first-in-class therapeutic candidate and we are not aware of any affinity hemofiltration device being market
cleared in any country for the single-use removal of circulating viruses or tumor-derived exosomes.
Government Regulation
The Hemopurifier is
subject to regulation by numerous regulatory bodies, primarily the FDA, and comparable international regulatory agencies. These
agencies require manufacturers of medical devices to comply with applicable laws and regulations governing the development, testing,
manufacturing, labeling, marketing, storage, distribution, advertising and promotion, and post-marketing surveillance reporting
of medical devices. As the primary mode of action of the Hemopurifier is attributable to the device component of this combination
product, the FDA’s Center for Devices and Radiological Health, or the CDRH, has primary jurisdiction over its premarket development,
review and approval. Failure to comply with applicable requirements may subject a device and/or its manufacturer to a variety of
administrative sanctions, such as issuance of warning letters, import detentions, civil monetary penalties and/or judicial sanctions,
such as product seizures, injunctions and criminal prosecution.
FDA’s Pre-market Clearance
and Approval Requirements
Each medical device
we seek to commercially distribute in the United States will require either a prior 510(k) clearance, unless it is exempt, or a
pre-market approval from the FDA. Generally, if a new device has a predicate that is already on the market under a 510(k) clearance,
the FDA will allow that new device to be marketed under a 510(k) clearance; otherwise, a premarket approval, or PMA, is required.
Medical devices are classified into one of three classes—Class I, Class II or Class III—depending on
the degree of risk associated with each medical device and the extent of control needed to provide reasonable assurance of safety
and effectiveness. Class I devices are deemed to be low risk and are subject to the general controls of the Federal Food,
Drug and Cosmetic Act, such as provisions that relate to: adulteration; misbranding; registration and listing; notification, including
repair, replacement, or refund; records and reports; and good manufacturing practices. Most Class I devices are classified
as exempt from pre-market notification under section 510(k) of the FD&C Act, and therefore may be commercially distributed
without obtaining 510(k) clearance from the FDA. Class II devices are subject to both general controls and special controls
to provide reasonable assurance of safety and effectiveness. Special controls include performance standards, post market surveillance,
patient registries and guidance documents. A manufacturer may be required to submit to the FDA a pre-market notification requesting
permission to commercially distribute some Class II devices. Devices deemed by the FDA to pose the greatest risk, such as
life-sustaining, life-supporting or implantable devices, or devices deemed not substantially equivalent to a previously cleared
510(k) device, are placed in Class III. A Class III device cannot be marketed in the United States unless the FDA approves
the device after submission of a PMA. However, there are some Class III devices for which FDA has not yet called for a PMA.
For these devices, the manufacturer must submit a pre-market notification and obtain 510(k) clearance in orders to commercially
distribute these devices. The FDA can also impose sales, marketing or other restrictions on devices in order to assure that they
are used in a safe and effective manner. We believe that the Hemopurifier will be classified as a Class III device and as such
will be subject to PMA submission and approval.
Pre-market Approval Pathway
A pre-market approval application must be
submitted to the FDA for Class III devices for which the FDA has required a PMA. The pre-market approval application process
is much more demanding than the 510(k) pre-market notification process. A pre-market approval application must be supported by
extensive data, including but not limited to technical, preclinical, clinical trials, manufacturing and labeling to demonstrate
to the FDA’s satisfaction reasonable evidence of safety and effectiveness of the device.
After a pre-market approval application
is submitted, the FDA has 45 days to determine whether the application is sufficiently complete to permit a substantive review
and thus whether the FDA will file the application for review. The FDA has 180 days to review a filed pre-market approval application,
although the review of an application generally occurs over a significantly longer period of time and can take up to several years.
During this review period, the FDA may request additional information or clarification of the information already provided. Also,
an advisory panel of experts from outside the FDA may be convened to review and evaluate the application and provide recommendations
to the FDA as to the approvability of the device.
Although the FDA is not bound by the advisory
panel decision, the panel’s recommendations are important to the FDA’s overall decision making process. In addition,
the FDA may conduct a preapproval inspection of the manufacturing facility to ensure compliance with the Quality System Regulation,
or QSR. The agency also may inspect one or more clinical sites to assure compliance with FDA’s regulations.
Upon completion of the PMA review, the FDA
may: (i) approve the PMA which authorizes commercial marketing with specific prescribing information for one or more indications,
which can be more limited than those originally sought; (ii) issue an approvable letter which indicates the FDA’s belief
that the PMA is approvable and states what additional information the FDA requires, or the post-approval commitments that must
be agreed to prior to approval; (iii) issue a not approvable letter which outlines steps required for approval, but which
are typically more onerous than those in an approvable letter, and may require additional clinical trials that are often expensive
and time consuming and can delay approval for months or even years; or (iv) deny the application. If the FDA issues an approvable
or not approvable letter, the applicant has 180 days to respond, after which the FDA’s review clock is reset.
Emergency Use Authorizations,
or EUAs, are granted by FDA in public health emergencies but allow use of the authorized device only during the period of the
respective public health emergency, and do not change the requirement to ultimately seek PMA approval after the authorization
period has ended.
Clinical Trials
Clinical trials are
almost always required to support pre-market approval and are sometimes required for 510(k) clearance. In the United States, for
significant risk devices, these trials require submission of an application for an IDE to the FDA. The IDE application must be
supported by appropriate data, such as animal and laboratory testing results, showing it is safe to test the device in humans and
that the testing protocol is scientifically sound. The IDE must be approved in advance by the FDA for a specific number of patients
at specified study sites. During the trial, the sponsor must comply with the FDA’s IDE requirements for investigator selection,
trial monitoring, reporting and recordkeeping. The investigators must obtain patient informed consent, rigorously follow the investigational
plan and study protocol, control the disposition of investigational devices and comply with all reporting and recordkeeping requirements.
Clinical trials for significant risk devices may not begin until the IDE application is approved by the FDA and the appropriate
institutional review boards, or IRBs, at the clinical trial sites. An IRB is an appropriately constituted group that has been formally
designated to review and monitor medical research involving subjects and which has the authority to approve, require modifications
in, or disapprove research to protect the rights, safety and welfare of human research subjects. The FDA or the IRB at each site
at which a clinical trial is being performed may withdraw approval of a clinical trial at any time for various reasons, including
a belief that the risks to study subjects outweigh the benefits or a failure to comply with FDA or IRB requirements. Even if a
trial is completed, the results of clinical testing may not demonstrate the safety and effectiveness of the device, may be equivocal
or may otherwise not be sufficient to obtain approval or clearance of the product. As we advance our clinical trials, we are in
close contact with our clinical sites to navigate and assess the impact, if any, of COVID-19 on our clinical trials and current
timelines, including delays or difficulties in enrolling patients and/or delays or difficulties in clinical site initiation.
Ongoing Regulation by the FDA
Even after a device receives clearance
or approval and is placed on the market, numerous regulatory requirements apply. These include:
|
|
·
|
establishment registration and device listing;
|
|
|
·
|
the QSR, which requires manufacturers, including third-party manufacturers, to follow stringent design, testing, control, documentation and other quality assurance procedures during all aspects of the manufacturing process;
|
|
|
·
|
labeling regulations and the FDA prohibitions against the promotion of products for uncleared, unapproved or “off-label” uses and other requirements related to promotional activities;
|
|
|
·
|
medical device reporting regulations, which require that manufactures report to the FDA if their device may have caused or contributed to a death or serious injury, or if their device malfunctioned and the device or a similar device marketed by the manufacturer would be likely to cause or contribute to a death or serious injury if the malfunction were to recur;
|
|
|
·
|
corrections and removal reporting regulations, which require that manufactures report to the FDA field corrections or removals if undertaken to reduce a risk to health posed by a device or to remedy a violation of the FDCA that may present a risk to health; and
|
|
|
·
|
post market surveillance regulations, which apply to certain Class II or III devices when necessary to protect the public health or to provide additional safety and effectiveness data for the device.
|
Some changes to an
approved PMA device, including changes in indications, labeling or manufacturing processes or facilities, require submission and
FDA approval of a new PMA or PMA supplement, as appropriate, before the change can be implemented. Supplements to a PMA often require
the submission of the same type of information required for an original PMA, except that the supplement is generally limited to
that information needed to support the proposed change from the device covered by the original PMA. The FDA uses the same procedures
and actions in reviewing PMA supplements as it does in reviewing original PMAs.
Failure by us or by
our suppliers to comply with applicable regulatory requirements can result in enforcement action by the FDA or state authorities,
which may include any of the following sanctions:
|
|
·
|
warning or untitled letters, fines, injunctions, consent decrees and civil penalties;
|
|
|
·
|
customer notifications, voluntary or mandatory recall or seizure of our products;
|
|
|
·
|
operating restrictions, partial suspension or total shutdown of production;
|
|
|
·
|
delay in processing submissions or applications for new products or modifications to existing products;
|
|
|
·
|
withdrawing approvals that have already been granted; and
|
The Medical Device
Reporting laws and regulations require us to provide information to the FDA when we receive or otherwise become aware of information
that reasonably suggests our device may have caused or contributed to a death or serious injury as well as a device malfunction
that likely would cause or contribute to death or serious injury if the malfunction were to recur. In addition, the FDA prohibits
an approved device from being marketed for off-label use. The FDA and other agencies actively enforce the laws and regulations
prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject
to significant liability, including substantial monetary penalties and criminal prosecution.
Newly discovered or
developed safety or effectiveness data may require changes to a product’s labeling, including the addition of new warnings
and contraindications, and also may require the implementation of other risk management measures. Also, new government requirements,
including those resulting from new legislation, may be established, or the FDA’s policies may change, which could delay or
prevent regulatory clearance or approval of our products under development.
Healthcare Regulation
In addition to the
FDA’s restrictions on marketing of pharmaceutical products, the U.S. healthcare laws and regulations that may affect our
ability to operate include: the federal fraud and abuse laws, including the federal anti-kickback and false claims laws; federal
data privacy and security laws; and federal transparency laws related to payments and/or other transfers of value made to physicians
and other healthcare professionals and teaching hospitals. Many states have similar laws and regulations that may differ from each
other and federal law in significant ways, thus complicating compliance efforts. For example, states have anti-kickback and false
claims laws that may be broader in scope than analogous federal laws and may apply regardless of payor. In addition, state data
privacy laws that protect the security of health information may differ from each other and may not be preempted by federal law.
Moreover, several states have enacted legislation requiring pharmaceutical manufacturers to, among other things, establish marketing
compliance programs, file periodic reports with the state, make periodic public disclosures on sales and marketing activities,
report information related to drug pricing, require the registration of sales representatives, and prohibit certain other sales
and marketing practices. These laws may adversely affect our sales, marketing and other activities with respect to any product
candidate for which we receive approval to market in the United States by imposing administrative and compliance burdens on us.
Because of the breadth
of these laws and the narrowness of available statutory exceptions and regulatory safe harbors, it is possible that some of our
business activities, particularly any sales and marketing activities after a product candidate has been approved for marketing
in the United States, could be subject to legal challenge and enforcement actions. If our operations are found to be in violation
of any of the federal and state laws described above or any other governmental regulations that apply to us, we may be subject
to significant civil, criminal, and administrative penalties, including, without limitation, damages, fines, imprisonment, exclusion
from participation in government healthcare programs, additional reporting obligations and oversight if we become subject to a
corporate integrity agreement or other agreement to resolve allegations of non-compliance with these laws, and the curtailment
or restructuring of our operations, any of which could adversely affect our ability to operate our business and our results of
operations.
From time to time,
legislation is drafted and introduced in Congress that could significantly change the statutory provisions governing the regulatory
approval, manufacture and marketing of regulated products or the reimbursement thereof. For example, in the U.S., the Patient Protection
and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act of 2010, or collectively, PPACA, among
other things, reduced and/or limited Medicare reimbursement to certain providers and imposed an annual excise tax of 2.3% on any
entity that manufactures or imports medical devices offered for sale in the United States, with limited exceptions. However, the
2020 federal spending package permanently eliminated, effective January 1, 2020, this PPACA-mandated medical device tax. On December
14, 2018, a Texas U.S. District Court Judge ruled that the PPACA is unconstitutional in its entirety because the “individual
mandate” was repealed by Congress as part of the legislation enacted in 2017, informally titled the Tax Cuts and Jobs Act
of 2017. Additionally, on December 18, 2019, the U.S. Court of Appeals for the 5th Circuit upheld the District Court ruling that
the individual mandate was unconstitutional and remanded the case back to the District Court to determine whether the remaining
provisions of the PPACA are invalid as well. On March 2, 2020, the United States Supreme Court granted the petitions for writs
of certiorari to review this case, and has allotted one hour for oral arguments, which are expected to occur in the fall of 2020.
In addition, the Budget Control Act of 2011, as amended by subsequent legislation, further reduces Medicare’s payments to
providers by two percent through fiscal year 2030. The Coronavirus Aid, Relief and Economic Security Act, or CARES Act, which was
signed into law in March 2020 and is designed to provide financial support and resources to individuals and businesses affected
by the COVID-19 pandemic, suspended the two percent Medicare sequester from May 1, 2020 through December 31, 2020, and extended
the sequester by one year, through 2030. These reductions may reduce providers’ revenues or profits, which could affect their
ability to purchase new technologies. Furthermore, the healthcare industry in the U.S. has experienced a trend toward cost containment
as government and private insurers seek to control healthcare costs by imposing lower payment rates and negotiating reduced contract
rates with service providers. Legislation could be adopted in the future that limits payments for our products from governmental
payors. It is possible that additional governmental action will be taken to address the COVID-19 pandemic.
Coverage and Reimbursement
In both the U.S. and
international markets, the use of medical devices is dependent in part on the availability of reimbursement from third-party payors,
such as government and private insurance plans. Healthcare providers that use medical devices generally rely on third-party payors
to pay for all or part of the costs and fees associated with the medical procedures being performed or to compensate them for their
patient care services. Should our products under development be approved for commercialization by the FDA, any such products may
not be considered cost-effective, reimbursement may not be available in the U.S. or other countries, if approved, and reimbursement
may not be sufficient to allow sales of our future products on a profitable basis. The coverage decisions of third-party payors
will be significantly influenced by the assessment of our future products by health technology assessment bodies. If approved for
use in the U.S., we expect that any products that we develop will be purchased primarily by medical institutions, which will in
turn bill various third-party payors for the health care services provided to patients at their facility. Payors may include the
Centers for Medicare & Medicaid Services, or CMS, which administers the Medicare program and works in partnership with state
governments to administer Medicaid, other government programs and private insurance plans. The process involved in applying for
coverage and reimbursement from CMS is lengthy and expensive. Further, Medicare coverage is based on our ability to demonstrate
that the treatment is “reasonable and necessary” for Medicare beneficiaries. Even if products utilizing our Aethlon
Hemopurifier® technology receive FDA and other regulatory clearance or approval, they may not be granted coverage and reimbursement
by any payor, including by CMS. Many private payors use coverage decisions and payment amounts determined by CMS as guidelines
in setting their coverage and reimbursement policies and amounts. However, no uniform policy for coverage and reimbursement for
medical devices exists among third-party payors in the United States. Therefore, coverage and reimbursement can differ significantly
from payor to payor.
Manufacturing
Manufacturing of our
Hemopurifier occurs in collaboration with two contract manufacturers based in California that are compliant with the Good Manufacturing
Practice regulations promulgated by the FDA. Our contract manufacturers are registered with the FDA. Previously, we did receive
an export license from FDA that allows for the export of our Hemopurifier to support clinical studies in India. To date, our manufacture
of the Hemopurifier has been limited to quantities necessary to support our clinical studies. Although we have not yet been impacted,
we are continuing to assess the interruption of, or delays in receiving, our Hemopurifier from our contract manufacturers due to
staffing shortages, production slowdowns or stoppages and disruptions in delivery systems contributable to COVID-19.
Our costs of compliance
with federal, state and local environmental laws have been immaterial to date.
Sources and Availability of Raw Materials and the
Names of Principal Suppliers
Our Hemopurifiers are
assembled by Aethlon personnel in a manufacturing facility provided and maintained following current Good Manufacturing Practices
by Life Science Outsourcing, Inc, or LSO. Aethlon personnel assemble the various components of the Hemopurifier with materials
from our various suppliers, which are purchased and released by Aethlon and stored at LSO prior to use in manufacturing. Specifically,
the Hemopurifier contains three critical components with limited available suppliers. The base cartridge on which the Hemopurifier
is constructed is sourced from Medica S.p.A and we are dependent on the continued availability of these cartridges. Although there
are other suppliers, the process of qualifying a new supplier takes time and regulatory approvals must be obtained. We currently
purchase the diatomaceous earth from Janus Scientific, Inc., as the distributor; however, the product is manufactured by Imerys
Minerals Ltd. There potentially are other suppliers of this product, but as with the cartridges, qualifying and obtaining required
regulatory approvals takes time and resources. The GNA lectin is sourced from Vector Laboratories Inc. and also is available from
other suppliers; however, Sigma Aldrich is the only approved back up supplier at this time. A business interruption at any of these
sources could have a material impact on our ability to manufacture the Hemopurifier.
Sales and Marketing
We do not currently
have any sales and marketing capability. With respect to commercialization efforts in the future, we intend to build or contract
for distribution, sales and marketing capabilities for any product candidate that is approved. From time to time, we have had and
are having strategic discussions with potential collaboration partners for our product candidates, although no assurance can be
given that we will be able to enter into one or more collaboration agreements for our product candidates on acceptable terms, if
at all.
Product Liability
The risk of product
liability claims, product recalls and associated adverse publicity is inherent in the testing, manufacturing, marketing and sale
of medical products. We have limited clinical trial liability insurance coverage. It is possible that future insurance coverage
may not be adequate or available. We may not be able to secure product liability insurance coverage on acceptable terms or at reasonable
costs when needed. Any liability for mandatory damages could exceed the amount of our coverage. A successful product liability
claim against us could require us to pay a substantial monetary award. Moreover, a product recall could generate substantial negative
publicity about our products and business and inhibit or prevent commercialization of other future product candidates.
Employees
We have eight full-time
employees. We utilize, whenever appropriate, consultants in order to conserve cash and resources.
We believe our employee
relations are good. None of our employees are represented by a labor union or are subject to collective-bargaining agreements.
ITEM 1A. RISK FACTORS
An investment in
our securities involves a high degree of risk. You should carefully consider the risks described below as well as the other information
in this Annual Report before deciding to invest in or maintain your investment in our company. The risks described below are not
intended to be an all-inclusive list of all of the potential risks relating to an investment in our securities. Any of the risk
factors described below could significantly and adversely affect our business, prospects, financial condition and results of operations.
Additional risks and uncertainties not currently known or that are currently considered to be immaterial may also materially and
adversely affect our business. As a result, the trading price or value of our securities could be materially adversely affected
and you may lose all or part of your investment.
Risks Relating to Our Financial Position and Need for Additional
Capital
We have incurred significant losses and expect to continue
to incur losses for the foreseeable future.
We have never been
profitable. We have generated revenues during the fiscal years ended March 31, 2020 and March 31, 2019, in the amounts of $650,187,
and $229,625, respectively, primarily from our contracts with the NIH. Our revenues, from research grants, continue to be insufficient
to cover our cost of operations. It is possible that we may not be able to enter into future government contracts beyond our current
contract with the NIH. Future profitability, if any, will require the successful commercialization of our Hemopurifier technology,
other products that may emerge from our potential diagnostic products or from additional government contract or grant income.
We may not be able to successfully commercialize one or more of our products, and even if commercialization is successful, we
may never be profitable.
We will require additional financing to sustain our operations.
We will require significant additional financing
for our operations and for expected additional future clinical trials in the U.S., as well as to fund all of our continued research
and development activities for the Hemopurifier and other future products. In addition, as we expand our activities, our overhead
costs to support personnel, laboratory materials and infrastructure will increase. If the financing we may require to sustain our
working capital needs be unavailable to us on reasonable terms, or at all, we may be unable to support our research and FDA development
activities, including our planned clinical trials. The failure to implement our research and clearance activities would have a
material adverse effect on our ability to commercialize our products or continue our business.
We also will need to raise additional funds through debt
or equity financings to achieve our business objectives and to satisfy our cash obligations, which may dilute the ownership of
our existing stockholders.
We will need to raise
additional funds through debt and/or equity financings in order to complete our ultimate business objectives, including funding
working capital to support development and regulatory clearance of our potential products. We also may choose to raise additional
funds in debt or equity financings if they are available to us on reasonable terms to increase our working capital and to strengthen
our financial position. Any sales of additional equity or convertible debt securities could result in dilution of the equity interests
of our existing stockholders, which could be substantial. Also, new investors may require that we and certain of our stockholders
enter into voting arrangements that give them additional voting control or representation on our Board of Directors.
Risks Related to Our Business Operations
We face intense competition in the medical device industry.
We compete with numerous
U.S. and foreign companies in the medical device industry, and many of our competitors have greater financial, personnel, operational
and research and development resources than we do. We believe that because the field of exosome research is burgeoning, multiple
competitors are or will be developing competing technologies to address exosomes in cancer. Progress is constant in the treatment
and prevention of viral diseases, so the opportunities for the Hemopurifier may be reduced there as well. Diagnostic technology
may be developed that can supplant diagnostics we are developing for neurodegenerative diseases and cancer. Our commercial opportunities
will be reduced or eliminated if our competitors develop and market products for any of the diseases we target that:
|
|
·
|
have fewer or less severe adverse side effects;
|
|
|
·
|
are more adaptable to various modes of dosing;
|
|
|
·
|
are easier to administer; or
|
|
|
·
|
are less expensive than the products or product candidates we are developing.
|
Even if we are successful
in developing the Hemopurifier and potential diagnostic products, and obtain FDA and other regulatory approvals necessary for commercializing
them, our products may not compete effectively with other successful products. Researchers are continually learning more about
diseases, which may lead to new technologies for treatment. Our competitors may succeed in developing and marketing products that
are either more effective than those that we may develop, alone or with our collaborators, or that are marketed before any products
we develop are marketed. Our competitors include fully integrated pharmaceutical companies and biotechnology companies as well
as universities and public and private research institutions. Many of the organizations competing with us have substantially greater
capital resources, larger research and development staffs and facilities, greater experience in product development and in obtaining
regulatory approvals, and greater marketing capabilities than we do. If our competitors develop more effective pharmaceutical treatments
for infectious disease or cancer, or bring those treatments to market before we can commercialize the Hemopurifier for such uses,
we may be unable to obtain any market traction for our products, or the diseases we seek to treat may be substantially addressed
by competing treatments. If we are unable to successfully compete against larger companies in the pharmaceutical industry, we may
never generate significant revenue or be profitable.
We have limited experience in identifying and working
with large-scale contracts with medical device manufacturers; manufacture of our devices must comply with good manufacturing practices
in the U.S.
To achieve the levels
of production necessary to commercialize our Hemopurifier and any other future products, we will need to secure large-scale manufacturing
agreements with contract manufacturers which comply with good manufacturing practice standards and other standards prescribed by
various federal, state and local regulatory agencies in the U.S. and any other country of use. We have limited experience coordinating
and overseeing the manufacture of medical device products on a large-scale. It is possible that manufacturing and control problems
will arise as we attempt to commercialize our products and that manufacturing may not be completed in a timely manner or at a commercially
reasonable cost. In addition, we may not be able to adequately finance the manufacture and distribution of our products on terms
acceptable to us, if at all. If we cannot successfully oversee and finance the manufacture of our products if they obtain regulatory
clearances, we may never generate revenue from product sales and we may never be profitable.
Our Hemopurifier technology may become obsolete.
Our Hemopurifier product
may be made unmarketable prior to commercialization by us by new scientific or technological developments by others with new treatment
modalities that are more efficacious and/or more economical than our products. The homeland security industry is growing rapidly
with many competitors that are trying to develop products or vaccines to protect against infectious disease. Any one of our competitors
could develop a more effective product which would render our technology obsolete. Further, our ability to achieve significant
and sustained penetration of our key target markets will depend upon our success in developing or acquiring technologies developed
by other companies, either independently, through joint ventures or through acquisitions. If we fail to develop or acquire, and
manufacture and sell, products that satisfy our customers’ demands, or we fail to respond effectively to new product announcements
by our competitors by quickly introducing competitive products, then market acceptance of our products could be reduced and our
business could be adversely affected. Our products may not remain competitive with products based on new technologies.
Our success is dependent in part on our executive officers.
Our success depends
to a critical extent on the continued services of our Chief Executive Officer, Timothy Rodell, MD, and our Chief Financial Officer,
James B. Frakes. If one or both of these key executive officers were to leave us, we would be forced to expend significant time
and money in the pursuit of a replacement, which would result in both a delay in the implementation of our business plan and the
diversion of limited working capital. The unique knowledge and expertise of these individuals would be difficult to replace within
the biotechnology field. We do not currently carry key man life insurance policies on any of our key executive officers which would
assist us in recouping our costs in the event of the loss of those officers. If either of our key officers were to leave us, it
could make it impossible, if not cause substantial delays and costs, to implement our long-term business objectives and growth.
Our inability to attract and retain qualified personnel
could impede our ability to achieve our business objectives.
We have eight full-time
employees, consisting of our Chief Executive Officer, our Chief Financial Officer, a recently hired Vice President, Manufacturing
and Product Development, four research scientists and an executive assistant. We utilize, whenever appropriate, consultants in
order to conserve cash and resources.
Although we believe
that these employees and consultants will be able to handle most of our additional administrative, research and development and
business development in the near term, we will nevertheless be required over the longer-term to hire highly skilled managerial,
scientific and administrative personnel to fully implement our business plan and growth strategies, including to mitigate the material
weakness in our internal control over financial reporting described above. Due to the specialized scientific nature of our business,
we are highly dependent upon our ability to attract and retain qualified scientific, technical and managerial personnel. Competition
for these individuals, especially in San Diego, California, where many biotechnology companies are located, is intense and we may
not be able to attract, assimilate or retain additional highly qualified personnel in the future. We may not be able to engage
the services of qualified personnel at competitive prices or at all, particularly given the risks of employment attributable to
our limited financial resources and lack of an established track record. Also, if we are required to attract personnel from other
parts of the U.S. or abroad, we may have significant difficulty doing so due to the high cost of living in the Southern California
area and due to the costs incurred with transferring personnel to the area. If we cannot attract and retain qualified staff and
executives, we will be unable to develop our products and achieve regulatory clearance, and our business could fail.
We plan to grow rapidly which will strain our resources;
our inability to manage our growth could delay or derail implementation of our business objectives.
We will need to significantly
expand our operations to implement our longer-term business plan and growth strategies. We will also be required to manage multiple
relationships with various strategic partners, technology licensors, customers, manufacturers and suppliers, consultants and other
third parties. This expansion and these expanded relationships will require us to significantly improve or replace our existing
managerial, operational and financial systems, procedures and controls; to improve the coordination between our various corporate
functions; and to manage, train, motivate and maintain a growing employee base. The time and costs to effectuate these steps may
place a significant strain on our management personnel, systems and resources, particularly given the limited amount of financial
resources and skilled employees that may be available at the time. We cannot assure you that we will institute, in a timely manner
or at all, the improvements to our managerial, operational and financial systems, procedures and controls necessary to support
our anticipated increased levels of operations and to coordinate our various corporate functions, or that we will be able to properly
manage, train, motivate and retain our anticipated increased employee base. If we cannot manage our growth initiatives, we will
be unable to commercialize our products on a large-scale in a timely manner, if at all, and our business could fail.
As a public company with limited financial resources undertaking
the launch of new medical technologies, we may have difficulty attracting and retaining executive management and directors.
The directors and management
of publicly traded corporations are increasingly concerned with the extent of their personal exposure to lawsuits and stockholder
claims, as well as governmental and creditor claims which may be made against them, particularly in view of recent changes in securities
laws imposing additional duties, obligations and liabilities on management and directors. Due to these perceived risks, directors
and management are also becoming increasingly concerned with the availability of directors’ and officers’ liability
insurance to pay on a timely basis the costs incurred in defending such claims. While we currently carry directors’ and officers’
liability insurance, such insurance is expensive and difficult to obtain. If we are unable to continue or provide directors’
and officers’ liability insurance at affordable rates or at all, it may become increasingly more difficult to attract and
retain qualified outside directors to serve on our Board of Directors. We may lose potential independent board members and management
candidates to other companies in the biotechnology field that have greater directors’ and officers’ liability insurance
to insure them from liability or to biotechnology companies that have revenues or have received greater funding to date which can
offer greater compensation packages. The fees of directors are also rising in response to their increased duties, obligations and
liabilities. In addition, our products could potentially be harmful to users, and we are exposed to claims of product liability
including for injury or death. We have limited insurance and may not be able to afford robust coverage even as our products are
introduced into the market. As a company with limited resources and potential exposures to management, we will have a more difficult
time attracting and retaining management and outside independent directors than a more established public or private company due
to these enhanced duties, obligations and potential liabilities.
If we fail to comply with extensive regulations of U.S.
and foreign regulatory agencies, the commercialization of our products could be delayed or prevented entirely.
Our Hemopurifier product
is subject to extensive government regulations related to development, testing, manufacturing and commercialization in the U.S.
and other countries. The determination of when and whether a product is ready for large-scale purchase and potential use will be
made by the U.S. Government through consultation with a number of governmental agencies, including the FDA, the National Institutes
of Health, the Centers for Disease Control and Prevention and the Department of Homeland Security. Our product candidates are in
the pre-clinical and clinical stages of development and have not received required regulatory approval from the FDA, or any foreign
regulatory agencies, to be commercially marketed and sold. The process of obtaining and complying with FDA and other governmental
regulatory approvals and regulations in the U.S. and in foreign countries is costly, time consuming, uncertain and subject to unanticipated
delays. Obtaining such regulatory approvals, if any, can take several years. Despite the time and expense exerted, regulatory approval
is never guaranteed. We also are subject to the following risks and obligations, among others:
|
|
·
|
the FDA may refuse to approve an application if they believe that applicable regulatory criteria are not satisfied;
|
|
|
·
|
the FDA may require additional testing for safety and effectiveness;
|
|
|
·
|
the FDA may interpret data from pre-clinical testing and clinical trials in different ways than we interpret them;
|
|
|
·
|
if regulatory approval of a product is granted, the approval may be limited to specific indications or limited with respect to its distribution; and
|
|
|
·
|
the FDA may change their approval policies and/or adopt new regulations.
|
Failure to comply with
these or other regulatory requirements of the FDA may subject us to administrative or judicially imposed sanctions, including:
|
|
·
|
product seizure or detention;
|
|
|
·
|
total or partial suspension of productions.
|
Delays in successfully completing
our planned clinical trials could jeopardize our ability to obtain regulatory approval.
Our business prospects
will depend on our ability to complete studies, clinical trials, including our currently planned EFS trial in 10 to 12 patients,
obtain satisfactory results, obtain required regulatory approvals and successfully commercialize our Hemopurifier product candidate.
Completion of our clinical trials, announcement of results of the trials and our ability to obtain regulatory approvals could
be delayed for a variety of reasons, including:
|
|
·
|
slow patient enrollment;
|
|
|
·
|
serious adverse events related to our medical device candidates;
|
|
|
·
|
unsatisfactory results of any clinical trial;
|
|
|
·
|
the failure of our principal third-party investigators to perform our clinical trials on our anticipated schedules;
|
|
|
·
|
different interpretations of our pre-clinical and clinical data, which could initially lead to inconclusive results; and
|
|
|
|
|
|
|
·
|
delays resulting from the coronavirus pandemic.
|
Our development costs
will increase if we have material delays in any clinical trial or if we need to perform more or larger clinical trials than planned.
If the delays are significant, or if any of our product candidates do not prove to be safe or effective or do not receive required
regulatory approvals, our financial results and the commercial prospects for our product candidates will be harmed. Furthermore,
our inability to complete our clinical trials in a timely manner could jeopardize our ability to obtain regulatory approval.
If we or our suppliers fail to comply with ongoing FDA
or foreign regulatory authority requirements, or if we experience unanticipated problems with our products, these products could
be subject to restrictions or withdrawal from the market.
Any product for which
we obtain clearance or approval, and the manufacturing processes, reporting requirements, post-approval clinical data and promotional
activities for such product, will be subject to continued regulatory review, oversight and periodic inspections by the FDA and
other domestic and foreign regulatory bodies. In particular, we and our third-party suppliers may be required to comply with the
FDA’s Quality System Regulation, or QSR. These FDA regulations cover the methods and documentation of the design, testing,
production, control, quality assurance, labeling, packaging, sterilization, storage and shipping of our products. Compliance with
applicable regulatory requirements is subject to continual review and is monitored rigorously through periodic inspections by the
FDA. If we, or our manufacturers, fail to adhere to QSR requirements in the U.S., this could delay production of our products and
lead to fines, difficulties in obtaining regulatory clearances, recalls, enforcement actions, including injunctive relief or consent
decrees, or other consequences, which could, in turn, have a material adverse effect on our financial condition or results of operations.
In addition, the FDA
assesses compliance with the QSR through periodic announced and unannounced inspections of manufacturing and other facilities.
The failure by us or one of our suppliers to comply with applicable statutes and regulations administered by the FDA, or the failure
to timely and adequately respond to any adverse inspectional observations or product safety issues, could result in any of the
following enforcement actions:
|
|
·
|
untitled letters, warning letters, fines, injunctions, consent decrees and civil penalties;
|
|
|
·
|
unanticipated expenditures to address or defend such actions;
|
|
|
·
|
customer notifications or repair, replacement, refunds, recall, detention or seizure of our products;
|
|
|
·
|
operating restrictions or partial suspension or total shutdown of production;
|
|
|
·
|
refusing or delaying our requests for 510(k) clearance or premarket approval of new products or modified products;
|
|
|
·
|
withdrawing 510(k) clearances or premarket approvals that have already been granted;
|
|
|
·
|
refusal to grant export approval for our products; or
|
|
|
·
|
criminal prosecution.
|
Moreover, the FDA strictly
regulates the promotional claims that may be made about approved products. In particular, a product may not be promoted for uses
that are not approved by the FDA as reflected in the product’s approved labeling. However, companies may share truthful and
not misleading information that is otherwise consistent with a product’s FDA approved labeling. The FDA and other agencies
actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly
promoted off-label uses may be subject to significant civil, criminal and administrative penalties. The COVID-19 pandemic could
also potentially affect the business of the FDA and comparable authorities in other countries, which could result in delays in
meetings related to planned clinical trials and ultimately of reviews and approvals of our product candidates.
Any of these sanctions
could have a material adverse effect on our reputation, business, results of operations and financial condition. Furthermore, our
key component suppliers may not currently be or may not continue to be in compliance with all applicable regulatory requirements,
which could result in our failure to produce our products on a timely basis and in the required quantities, if at all.
If our products, or malfunction of our products, cause
or contribute to a death or a serious injury, we will be subject to medical device reporting regulations, which can result in voluntary
corrective actions or agency enforcement actions.
Under the FDA medical
device reporting regulations, medical device manufacturers are required to report to the FDA information that a device has or may
have caused or contributed to a death or serious injury or has malfunctioned in a way that would likely cause or contribute to
death or serious injury if the malfunction of the device or one of our similar devices were to recur. If we fail to report these
events to the FDA within the required timeframes, or at all, FDA could take enforcement action against us. Any such adverse event
involving our products also could result in future voluntary corrective actions, such as recalls or customer notifications, or
agency action, such as inspection or enforcement action. Any corrective action, whether voluntary or involuntary, as well as defending
ourselves in a lawsuit, will require the dedication of our time and capital, distract management from operating our business, and
may harm our reputation and financial results.
We outsource almost all of our operational and development
activities, and if any party to which we have outsourced certain essential functions fails to perform its obligations under agreements
with us, the development and commercialization of our lead product candidate and any future product candidates that we may develop
could be delayed or terminated.
We generally rely on
third-party consultants or other vendors to manage and implement the day-to-day conduct of our operations, including conducting
clinical trials and manufacturing our current product candidates and any future product candidates that we may develop. Accordingly,
we are and will continue to be dependent on the timeliness and effectiveness of their efforts. Our dependence on third parties
includes key suppliers and third-party service providers supporting the development, manufacture and regulatory approval of our
products as well as support for our information technology systems and other infrastructure. While our management team oversees
these vendors, failure of any of these third parties to meet their contractual, regulatory and other obligations or the development
of factors that materially disrupt the performance of these third parties could have a material adverse effect on our business.
For example, all of the key oversight responsibilities for the development and manufacture of our lead product candidate are conducted
by our management team, but all other activities are the responsibility of third-party vendors. It is possible that the current
COVID-19 epidemic might constrain the ability of needed third-party vendors to provide services that we require.
If a clinical research
organization that we utilize is unable to allocate sufficient qualified personnel to our studies in a timely manner or if the work
performed by it does not fully satisfy the requirements of the FDA or other regulatory agencies, we may encounter substantial delays
and increased costs in completing our development efforts. Any manufacturer that we select may encounter difficulties in the manufacture
of new products in commercial quantities, including problems involving product yields, product stability or shelf life, quality
control, adequacy of control procedures and policies, compliance with FDA regulations and the need for further FDA approval of
any new manufacturing processes and facilities. If any of these occur, the development and commercialization of our product candidates
could be delayed, curtailed or terminated because we may not have sufficient financial resources or capabilities to continue such
development and commercialization on our own. If we rely on only one source for the manufacture of the clinical or commercial supplies
of any of our product candidates or products, any production problems or supply constraints with that manufacturer could adversely
impact the development or commercialization of that product candidate or product.
If we or our contractors or service providers fail to
comply with regulatory laws and regulations, we or they could be subject to regulatory actions, which could affect our ability
to develop, market and sell our product candidates and any other or future product candidates that we may develop and may harm
our reputation.
If we or our manufacturers
or other third-party contractors fail to comply with applicable federal, state or foreign laws or regulations, we could be subject
to regulatory actions, which could affect our ability to develop, market and sell our current product candidates or any future
product candidates under development successfully and could harm our reputation and lead to reduced or non-acceptance of our proposed
product candidates by the market. Even technical recommendations or evidence by the FDA through letters, site visits, and overall
recommendations to academia or biotechnology companies may make the manufacturing of a clinical product extremely labor intensive
or expensive, making the product candidate no longer viable to manufacture in a cost-efficient manner. The mode of administration
may make the product candidate not commercially viable. The required testing of the product candidate may make that candidate no
longer commercially viable. The conduct of clinical trials may be critiqued by the FDA, or a clinical trial site’s Institutional
Review Board or Institutional Biosafety Committee, which may delay or make impossible clinical testing of a product candidate.
The Institutional Review Board for a clinical trial may stop a trial or deem a product candidate unsafe to continue testing. This
would have a material adverse effect on the value of the product candidate and our business prospects.
We will need to outsource and rely on third parties for
the clinical development and manufacture, sales and marketing of our current product candidates or any future product candidates
that we may develop, and our future success will be dependent on the timeliness and effectiveness of the efforts of these third
parties.
We do not have the
required financial and human resources to carry out on our own all the pre-clinical and clinical development for our current product
candidates or any other or future product candidates that we may develop, and do not have the capability and resources to manufacture,
market or sell our current product candidates or any future product candidates that we may develop. Our business model calls for
the partial or full outsourcing of the clinical and other development and manufacturing, sales and marketing of our product candidates
in order to reduce our capital and infrastructure costs as a means of potentially improving our financial position. Our success
will depend on the performance of these outsourced providers. If these providers fail to perform adequately, our development of
product candidates may be delayed and any delay in the development of our product candidates would have a material and adverse
effect on our business prospects.
We are and will be exposed to product liability risks,
and clinical and preclinical liability risks, which could place a substantial financial burden upon us should we be sued.
Our business exposes
us to potential product liability and other liability risks that are inherent in the testing, manufacturing and marketing of medical
devices. Claims may be asserted against us. A successful liability claim or series of claims brought against us could have a material
adverse effect on our business, financial condition and results of operations. We may not be able to continue to obtain or maintain
adequate product liability insurance on acceptable terms, if at all, and such insurance may not provide adequate coverage against
potential liabilities. Claims or losses in excess of any product liability insurance coverage that we may obtain could have a material
adverse effect on our business, financial condition and results of operations.
Our Hemopurifier product
may be used in connection with medical procedures in which it is important that those products function with precision and accuracy.
If our products do not function as designed, or are designed improperly, we may be forced by regulatory agencies to withdraw such
products from the market. In addition, if medical personnel or their patients suffer injury as a result of any failure of our products
to function as designed, or our products are designed inappropriately, we may be subject to lawsuits seeking significant compensatory
and punitive damages. The risk of product liability claims, product recalls and associated adverse publicity is inherent in the
testing, manufacturing, marketing and sale of medical products. We have recently obtained general clinical trial liability insurance
coverage. However, our insurance coverage may not be adequate or available. We may not be able to secure product liability insurance
coverage on acceptable terms or at reasonable costs when needed. Any product recall or lawsuit seeking significant monetary damages
may have a material effect on our business and financial condition. Any liability for mandatory damages could exceed the amount
of our coverage. Moreover, a product recall could generate substantial negative publicity about our products and business and inhibit
or prevent commercialization of other future product candidates.
We have not received, and may never receive, approval
from the FDA to market a medical device in the United States.
Before a new medical
device can be marketed in the U.S., it must first receive a PMA or 510(k) clearance from the FDA, unless an exemption applies.
A PMA submission, which is a higher standard than a 510(k) clearance, is used to demonstrate to the FDA that a new or modified
device is safe and effective. The 510(k) is used to demonstrate that a device is “substantially equivalent” to a predicate
device (one that has been cleared by the FDA). We expect that any product we seek regulatory approval for, including the Hemopurifier,
will require a PMA. The FDA approval process involves, among other things, successfully completing clinical trials and filing for
and obtaining a PMA. The PMA process requires us to prove the safety and effectiveness of our products to the FDA’s satisfaction.
This process, which includes preclinical studies and clinical trials, can take many years and requires the expenditure of substantial
resources and may include post-marketing surveillance to establish the safety and efficacy of the product. Notwithstanding the
effort and expense incurred, the process may never result in the FDA granting a PMA. Data obtained from preclinical studies and
clinical trials are subject to varying interpretations that could delay, limit or prevent regulatory approval. Delays or rejections
may also be encountered based upon changes in governmental policies for medical devices during the period of product development.
The FDA can delay, limit or deny approval of a PMA application for many reasons, including:
|
|
·
|
our inability to demonstrate safety or effectiveness of the Hemopurifier or any other product we develop to the FDA’s satisfaction;
|
|
|
·
|
insufficient data from our preclinical studies and clinical trials, including for our Hemopurifier, to support approval;
|
|
|
·
|
failure of the facilities of our third-party manufacturer or suppliers to meet applicable requirements;
|
|
|
·
|
inadequate compliance with preclinical, clinical or other regulations;
|
|
|
·
|
our failure to meet the FDA’s statistical requirements for approval; and
|
|
|
·
|
changes in the FDA’s approval policies, or the adoption of new regulations that require additional data or additional clinical studies.
|
Modifications to products
that are approved through a PMA application generally need FDA approval. Similarly, some modifications made to products cleared
through a 510(k) may require a new 510(k). The FDA’s 510(k) clearance process usually takes from three to 12 months, but
may last longer. The process of obtaining a PMA is much costlier and more uncertain than the 510(k) clearance process and generally
takes from one to three years, or even longer, from the time the application is submitted to the FDA until an approval is obtained.
Any of our products considered to be a class III device, which are considered to pose the greatest risk and the approval of which
is governed by the strictest guidelines, will require the submission and approval of a PMA in order for us to market it in the
U.S. We also may design new products in the future that could require the clearance of a 510(k).
Although we have received
approval to proceed with clinical trials of the Hemopurifier in the U.S. under the investigational device exemption, the current
approval from the FDA to proceed could be revoked, the study could be unsuccessful, or the FDA PMA approval may not be obtained
or could be revoked. Even if we obtain approval, the FDA or other regulatory authorities may require expensive or burdensome post-market
testing or controls. Any delay in, or failure to receive or maintain, clearance or approval for our future products could prevent
us from generating revenue from these products or achieving profitability. Additionally, the FDA and other regulatory authorities
have broad enforcement powers. Regulatory enforcement or inquiries, or other increased scrutiny on us, could dissuade some physicians
from using our products and adversely affect our reputation and the perceived safety and efficacy of our products.
The approval requirements for medical products used to
fight bioterrorism are still evolving, and any products we develop for such uses may not meet these requirements.
We are advancing product
candidates under governmental policies that regulate the development and commercialization of medical treatment countermeasures
against bioterror and pandemic threats. While we intend to pursue FDA market clearance to treat infectious bioterror and
pandemic threats, it is often not feasible to conduct human studies against these deadly high threat pathogens. For example, the
Hemopurifier is an investigational device that has not yet received FDA approval for any indication. We continue to investigate
the potential for the use of the Hemopurifier in viral diseases under an open IDE and our FDA Breakthrough Designation for “…the
treatment of life-threatening glycosylated viruses that are not addressed with an approved therapy.” We currently have an
open FDA approved Expanded Access Protocol for the treatment of Ebola infected patients in the U.S. and a corresponding HealthCanada
approval in Canada. Based on our studies to date, the Hemopurifier can potentially clear many viruses that are pathogenic in humans,
including HCV, HIV and Ebola. We do have preclinical data suggesting that it could clear a closely related coronavirus (MERS).
We have tested the ability of a lab version of the Hemopurifier to clear a component of SARS-CoV-2, the causative agent in COVID-19,
but cannot test the whole virus in our labs for safety reasons. Furthermore, we also do not know whether clearing SARS-CoV-2 from
the blood, which is what the Hemopurifier is designed to do, would have a positive effect on this disease, which primarily targets
the lung; and even if effective, the Hemopurifier would only potentially be indicated in those patients with the most severe and
life-threatening manifestations. Additionally, we have a very limited supply of Hemopurifiers and therefore any use in this pandemic
will be only investigational in a very small number of patients, even if it appears that the device can help those patients.
Thus, we may not be
able to demonstrate the effectiveness of our treatment countermeasures through controlled human efficacy studies. Additionally,
a change in government policies could impair our ability to obtain regulatory approval and the FDA may not approve any of our product
candidates.
The results of our clinical trials may not support our
product candidate claims or may result in the discovery of adverse side effects.
Any research and development,
pre-clinical testing and clinical trial activities involving any products that we are developing or may develop will be subject
to extensive regulation and review by numerous governmental authorities both in the U.S. and abroad. In the future, we may conduct
clinical trials to support approval of new products. Clinical studies must be conducted in compliance with FDA regulations or the
FDA may take enforcement action. The data collected from these clinical studies may ultimately be used to support market clearance
for these products. Even if our clinical trials are completed as planned, the results of these trials may not support our product
candidate claims and the FDA may not agree with our conclusions regarding the trial results. Success in pre-clinical studies and
early clinical trials does not ensure that later clinical trials will be successful, and the later trials may not replicate the
results of prior trials and pre-clinical studies. The clinical trial process may fail to demonstrate that our product candidates
are safe and effective for the proposed indicated uses, which could cause us to abandon a product candidate and may delay development
of others. Any delay or termination of our clinical trials will delay the filing of our product submissions and, ultimately, our
ability to commercialize our product candidates and generate revenues. It is also possible that patients enrolled in clinical trials
will experience adverse side effects that are not currently part of the product candidate’s profile.
U.S. legislative or FDA regulatory reforms may make it
more difficult and costly for us to obtain regulatory approval of our product candidates and to manufacture, market and distribute
our products after approval is obtained.
From time to time,
legislation is drafted and introduced in Congress that could significantly change the statutory provisions governing the regulatory
approval, manufacture and marketing of regulated products or the reimbursement thereof. In addition, FDA regulations and guidance
are often revised or reinterpreted by the FDA in ways that may significantly affect our business and our products. Any new regulations
or revisions or reinterpretations of existing regulations may impose additional costs or lengthen review times of future products.
It is impossible to predict whether legislative changes will be enacted or FDA regulations, guidance or interpretations changed,
and what the impact of such changes, if any, may be or new product development efforts.
Our current and future business activities may be subject,
directly or indirectly, to applicable anti-kickback, fraud and abuse, false claims, physician payment transparency, health information
privacy and security and other healthcare laws and regulations, which could expose us to significant penalties.
We are currently or
will in the future be subject to healthcare regulation and enforcement by the U.S. federal government and the states in which we
will conduct our business once our product candidates are approved by the FDA and commercialized in the United States. In addition
to the FDA’s restrictions on marketing of approved products, the U.S. healthcare laws and regulations that may affect our
ability to operate include: the federal fraud and abuse laws, including the federal anti-kickback and false claims laws; federal
data privacy and security laws; and federal transparency laws related to payments and/or other transfers of value made to physicians
and other healthcare professionals and teaching hospitals. Many states have similar laws and regulations that may differ from each
other and federal law in significant ways, thus complicating compliance efforts. These laws may adversely affect our sales, marketing
and other activities with respect to any product candidate for which we receive approval to market in the United States by imposing
administrative and compliance burdens on us.
Because of the breadth
of these laws and the narrowness of available statutory exceptions and regulatory safe harbors, it is possible that some of our
business activities, particularly any sales and marketing activities after a product candidate has been approved for marketing
in the United States, could be subject to legal challenge and enforcement actions. If our operations are found to be in violation
of any of the federal and state laws described above or any other governmental regulations that apply to us, we may be subject
to significant civil, criminal, and administrative penalties, including, without limitation, damages, fines, imprisonment, exclusion
from participation in government healthcare programs, additional reporting obligations and oversight if we become subject to a
corporate integrity agreement or other agreement to resolve allegations of non-compliance with these laws, and the curtailment
or restructuring of our operations, any of which could adversely affect our ability to operate our business and our results of
operations.
Should our products be approved for commercialization,
lack of third-party coverage and reimbursement for our devices could delay or limit their adoption.
In both the U.S. and
international markets, the use of medical devices is dependent in part on the availability of reimbursement from third-party payors,
such as government and private insurance plans. Healthcare providers that use medical devices generally rely on third-party payors
to pay for all or part of the costs and fees associated with the medical procedures being performed or to compensate them for their
patient care services. Should our products under development be approved for commercialization by the FDA, any such products may
not be considered cost-effective, reimbursement may not be available in the U.S. or other countries, if approved, and reimbursement
may not be sufficient to allow sales of our future products, including the Hemopurifier, on a profitable basis. The coverage decisions
of third-party payors will be significantly influenced by the assessment of our future products by health technology assessment
bodies. These assessments are outside our control and any such evaluations may not be conducted or have a favorable outcome.
If approved for use
in the U.S., we expect that any products that we develop, including the Hemopurifier, will be purchased primarily by medical institutions,
which will in turn bill various third-party payors for the health care services provided to patients at their facility. Payors
may include the Centers for Medicare & Medicaid Services, or CMS, which administers the Medicare program and works in partnership
with state governments to administer Medicaid, other government programs and private insurance plans. The process involved in applying
for coverage and incremental reimbursement from CMS is lengthy and expensive. Further, Medicare coverage is based on our ability
to demonstrate that the treatment is “reasonable and necessary” for Medicare beneficiaries. Even if products utilizing
our Aethlon Hemopurifier technology receive FDA and other regulatory clearance or approval, they may not be granted coverage and
reimbursement by any payor, including by CMS. For some governmental programs, such as Medicaid, coverage and adequate reimbursement
differ from state to state and some state Medicaid programs may not pay adequate amounts for the procedure necessary to utilize
products utilizing our technology system, or any payment at all. Moreover, many private payors use coverage decisions and payment
amounts determined by CMS as guidelines in setting their coverage and reimbursement policies and amounts. However, no uniform policy
requirement for coverage and reimbursement for medical devices exists among third-party payors in the United States. Therefore,
coverage and reimbursement can differ significantly from payor to payor. If CMS or other agencies limit coverage or decrease or
limit reimbursement payments for doctors and hospitals, this may affect coverage and reimbursement determinations by many private
payors for any products that we develop.
Should any of our potential products, including the Hemopurifier,
be approved for commercialization, adverse changes in reimbursement policies and procedures by payors may impact our ability to
market and sell our products.
Healthcare costs have
risen significantly over the past decade, and there have been and continue to be proposals by legislators, regulators and third-party
payors to decrease costs. Third-party payors are increasingly challenging the prices charged for medical products and services
and instituting cost containment measures to control or significantly influence the purchase of medical products and services.
For example, in the
U.S., the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act of 2010, or
collectively, PPACA, among other things, reduced and/or limited Medicare reimbursement to certain providers. However, on December
14, 2018, a Texas U.S. District Court Judge ruled that the Affordable Care Act is unconstitutional in its entirety because the
“individual mandate” was repealed by Congress as part of the Tax Cuts and Jobs Act of 2017. Additionally, on December
18, 2019, the U.S. Court of Appeals for the 5th Circuit upheld the District Court ruling that the individual mandate was unconstitutional
and remanded the case back to the District Court to determine whether the remaining provisions of the PPACA are invalid as well.
On March 2, 2020, the United States Supreme Court granted the petitions for writs of certiorari to review this case, and has allotted
one hour for oral arguments, which are expected to occur in the fall. The Budget Control Act of 2011, as amended by subsequent
legislation, further reduces Medicare’s payments to providers by two percent through fiscal year 2030. The Coronavirus Aid,
Relief and Economic Security Act, or CARES Act, which was signed into law in March 2020 and is designed to provide financial support
and resources to individuals and businesses affected by the COVID-19 pandemic, suspended the two percent Medicare sequester from
May 1, 2020 through December 31, 2020, and extended the sequester by one year, through 2030. These reductions may reduce providers’
revenues or profits, which could affect their ability to purchase new technologies. Furthermore, the healthcare industry in the
U.S. has experienced a trend toward cost containment as government and private insurers seek to control healthcare costs by imposing
lower payment rates and negotiating reduced contract rates with service providers. Legislation could be adopted in the future that
limits payments for our products from governmental payors. It is possible that additional governmental action is taken to address
the COVID-19 pandemic. In addition, commercial payors such as insurance companies, could adopt similar policies that limit reimbursement
for medical device manufacturers’ products. Therefore, it is possible that our product or the procedures or patient care
performed using our product will not be reimbursed at a cost-effective level. We face similar risks relating to adverse changes
in reimbursement procedures and policies in other countries where we may market our products. Reimbursement and healthcare payment
systems vary significantly among international markets. Our inability to obtain international reimbursement approval, or any adverse
changes in the reimbursement policies of foreign payors, could negatively affect our ability to sell our products and have a material
adverse effect on our business and financial condition.
Our ability to use
net operating loss carryforwards and certain other tax attributes to offset future taxable income or taxes may be limited.
Under
the Tax Cuts and Jobs Act of 2017, as modified by the Coronavirus Aid, Relief, and Economic Security Act, or the CARES Act, federal
net operating losses incurred in tax years beginning after December 31, 2017, may be carried forward indefinitely, but the deductibility
of such federal net operating losses in tax years beginning after December 31, 2020, is limited to 80% of taxable income. It is
uncertain if and to what extent various states will conform to the Tax Cuts and Jobs Act of 2017 or the CARES Act. In addition,
under Sections 382 and 383 of the Internal Revenue Code of 1986, as amended, and corresponding provisions of state law, if a corporation
undergoes an “ownership change,” which is generally defined as a greater than 50% change in its equity ownership value
over a three-year period, the corporation’s ability to use its pre-change net operating loss carryforwards and other pre-change
tax attributes to offset its post-change income or taxes may be limited. We believe we have not experienced an ownership change
in the past three years, however, we could experience ownership changes in the future as a result of subsequent shifts in our stock
ownership, some of which may be outside of our control. If we achieve profitability and an ownership change occurs and our ability
to use our net operating loss carryforwards is materially limited, it would harm our future operating results by effectively increasing
our future tax obligations. In addition, at the state level, there may be periods during which the use of net operating loss carryforwards
is suspended or otherwise limited, which could accelerate or permanently increase state taxes owed.
Our use of hazardous materials, chemicals and viruses
exposes us to potential liabilities for which we may not have adequate insurance.
Our research and development
involves the controlled use of hazardous materials, chemicals and viruses. The primary hazardous materials include chemicals needed
to construct the Hemopurifier cartridges and the infected plasma samples used in preclinical testing of the Hemopurifier. All other
chemicals are fully inventoried and reported to the appropriate authorities, such as the fire department, who inspect the facility
on a regular basis. We are subject to federal, state, local and foreign laws governing the use, manufacture, storage, handling
and disposal of such materials. Although we believe that our safety procedures for the use, manufacture, storage, handling and
disposal of such materials comply with the standards prescribed by federal, state, local and foreign regulations, we cannot completely
eliminate the risk of accidental contamination or injury from these materials. We have had no incidents or problems involving hazardous
chemicals or biological samples. In the event of such an accident, we could be held liable for significant damages or fines.
We currently carry
a limited amount of insurance to protect us from damages arising from hazardous materials. Our product liability policy has a $3,000,000
limit of liability that would cover certain releases of hazardous substances away from our facilities. For our facilities, our
property policy provides $25,000 in coverage for contaminant clean-up or removal and $50,000 in coverage for damages to the premises
resulting from contamination. Should we violate any regulations concerning the handling or use of hazardous materials, or should
any injuries or death result from our use or handling of hazardous materials, we could be the subject of substantial lawsuits by
governmental agencies or individuals. We may not have adequate insurance to cover all or any of such claims, if any. If we were
responsible to pay significant damages for violations or injuries, if any, we might be forced to cease operations since such payments
could deplete our available resources.
Our products may in the future be subject to product recalls.
A recall of our products, either voluntarily or at the direction of the FDA or another governmental authority, including a third-country
authority, or the discovery of serious safety issues with our products, could have a significant adverse impact on us.
The FDA and similar
foreign governmental authorities have the authority to require the recall of commercialized products in the event of material deficiencies
or defects in design or manufacture. For the FDA, the authority to require a recall must be based on a finding that there is reasonable
probability that the device would cause serious injury or death. In addition, foreign governmental bodies have the authority to
require the recall of our products in the event of material deficiencies or defects in design or manufacture. Manufacturers may,
under their own initiative, recall a product if any material deficiency in a device is found. The FDA requires that certain classifications
of recalls be reported to the FDA within 10 working days after the recall is initiated. A government-mandated or voluntary recall
by us or one of our international distributors could occur as a result of an unacceptable risk to health, component failures, malfunctions,
manufacturing errors, design or labeling defects or other deficiencies and issues. Recalls of any of our products would divert
managerial and financial resources and have an adverse effect on our reputation, results of operations and financial condition,
which could impair our ability to produce our products in a cost-effective and timely manner in order to meet our customers’
demands. We may also be subject to liability claims, be required to bear other costs, or take other actions that may have a negative
impact on our future sales and our ability to generate profits. Companies are required to maintain certain records of recalls,
even if they are not reportable to the FDA or another third-country competent authority. We may initiate voluntary recalls involving
our products in the future that we determine do not require notification of the FDA or another third-country competent authority.
If the FDA disagrees with our determinations, they could require us to report those actions as recalls. A future recall announcement
could harm our reputation with customers and negatively affect our sales. In addition, the FDA could take enforcement action for
failing to report recalls. We are also required to follow detailed recordkeeping requirements for all firm-initiated medical device
corrections and removals.
Our business is subject to risks
arising from the recent COVID-19 pandemic.
The current COVID-19 worldwide pandemic
has presented substantial public health and economic challenges and is affecting our employees, patients, communities and business
operations, as well as the U.S. and global economy and financial markets.
International and U.S. governmental authorities
in impacted regions are taking actions in an effort to slow the spread of COVID-19, including issuing varying forms of “stay-at-home”
orders, and restricting business functions outside of one’s home. In response, we have implemented a work from home policy
for all non-laboratory employees, following the guidelines or directives issued by federal, state and local government agencies
in the U.S.
To date, we do not currently anticipate
any interruptions in supply. In addition, while we are continuing the process of getting our clinical trial underway, we expect
that COVID-19 precautions may directly or indirectly impact the timeline for the trial. As the COVID-19 pandemic continues to spread
around the globe, we may experience disruptions that could severely impact our business, clinical trials and manufacturing and
supply chains, including:
|
|
•
|
further delays or difficulties in enrolling patients in our clinical trials;
|
|
|
|
|
|
|
•
|
delays or difficulties in clinical site initiation, including difficulties in recruiting clinical site investigators and clinical
site staff;
|
|
|
|
|
|
|
•
|
diversion of healthcare resources away from the conduct of clinical trials, including the diversion of hospitals serving as our
clinical trial sites and hospital staff supporting the conduct of our clinical trials;
|
|
|
|
|
|
|
•
|
interruption of key clinical trial activities, such as clinical trial site monitoring, due to limitations on travel imposed or
recommended by federal or state governments, employers and others or interruption of clinical trial subject visits and study procedures,
which may impact the integrity of subject data and clinical study endpoints;
|
|
|
|
|
|
|
•
|
interruption of, or delays in receiving, supplies of our product candidates from our contract manufacturing organizations due
to staffing shortages, production slowdowns or stoppages and disruptions in delivery systems;
|
|
|
|
|
|
|
•
|
delays in clinical sites receiving the supplies and materials needed to conduct our clinical trials and interruption in global
shipping that may affect the transport of clinical trial materials;
|
|
|
|
|
|
|
•
|
limitations on employee resources that would otherwise be focused on the conduct of our clinical trials, including because of
sickness of employees or their families or the desire of employees to avoid contact with large groups of people;
|
|
|
|
|
|
|
•
|
delays in receiving feedback or approvals from the FDA or other regulatory authorities with respect to future clinical trials
or regulatory submissions;
|
|
|
|
|
|
|
•
|
changes in local regulations as part of a response to COVID-19 which may require us to change the ways in which our clinical trials
are conducted, which may result in unexpected costs, or to discontinue the clinical trials altogether;
|
|
|
|
|
|
|
•
|
delays in necessary interactions with local regulators, ethics committees and other important agencies and contractors due to
limitations in employee resources or forced furlough of government employees;
|
|
|
•
|
refusal of the FDA to accept data from clinical trials in affected geographies; and
|
|
|
|
|
|
|
•
|
difficulties launching or commercializing products, including due to reduced access to doctors as a result of social distancing
protocols.
|
In addition, the spread of COVID-19 has
had and may continue to impact the trading price of shares of our common stock and could further negatively impact our ability
to raise additional capital on a timely basis or at all.
The COVID-19 pandemic continues to rapidly
evolve. The extent to which COVID-19 may impact our business, including our clinical trials, manufacturing and supply chains and
financial condition will depend on future developments, which are highly uncertain and cannot be predicted with confidence, such
as the continued geographic spread of the disease, the duration of the pandemic, travel restrictions and social distancing in the
United States and other countries, continued business closures or business disruptions and the effectiveness of actions taken in
the United States and other countries to contain and treat the disease.
Our products are manufactured with
raw materials that are sourced from specialty suppliers with limited competitors and we may therefore be unable to access the materials
we need to manufacture our products.
Specifically, the Hemopurifier
contains three critical components with limited supplier numbers. The base cartridge on which the Hemopurifier is constructed is
sourced from Medica S.p.A and we are dependent on the continued availability of these cartridges. We currently purchase the diatomaceous
earth from Janus Scientific Inc., our distributor; however, the product is manufactured by Imerys Minerals Ltd., which is the only
supplier of this product. The Galanthus nivalis agglutinin, or GNA, is sourced from Vector Laboratories, Inc. and also is available
from other suppliers; however, Sigma Aldrich is the only approved back up supplier at this time. A business interruption at any
of these sources, including interruption resulting from the coronavirus pandemic, could have a material impact on our ability to
manufacture the Hemopurifier.
Even though we have received breakthrough
device designation for the Hemopurifier for two independent indications, this designation may not expedite the development or review
of the Hemopurifier and does not provide assurance ultimately of PMA submission or approval by the FDA.
The Breakthrough Devices
Program is a voluntary program intended to expedite the review, development, assessment and review of certain medical devices that
provide for more effective treatment or diagnosis of life-threatening or irreversibly debilitating human diseases or conditions
for which no approved or cleared treatment exists or that offer significant advantages over existing approved or cleared alternatives.
All submissions for devices designated as Breakthrough Devices will receive priority review, meaning that the review of the submission
is placed at the top of the appropriate review queue and receives additional review resources, as needed.
Although breakthrough
designation or access to any other expedited program may expedite the development or approval process, it does not change the standards
for approval. Although we obtained breakthrough device designation for the Hemopurifier for two indications, we may not experience
faster development timelines or achieve faster review or approval compared to conventional FDA procedures. For example, the time
required to identify and resolve issues relating to manufacturing and controls, the acquisition of a sufficient supply of our product
for clinical trial purposes or the need to conduct additional nonclinical or clinical studies may delay approval by the FDA, even
if the product qualifies for breakthrough designation or access to any other expedited program. Access to an expedited program
may also be withdrawn by the FDA if it believes that the designation is no longer supported by data from our clinical development
program. Additionally, qualification for any expedited review procedure does not ensure that we will ultimately obtain regulatory
approval for the product.
Compliance with laws, regulations,
and related interpretations and related legal claims or other regulatory enforcement actions could impact our business, and we
face additional risks and uncertainties related to any potential actions resulting from the Securities and Exchange Commission’s,
or the SEC, ongoing investigation, or any other investigation or action.
On February 7, 2020,
the SEC issued an Order of Suspension of Trading, or SEC Order, temporarily suspending trading in our stock for a period of ten
days. The SEC Order stated that the suspension was due to concerns regarding the accuracy and adequacy of information in the marketplace
that appeared to be disseminated by third party promotors and recent and unusual market activity since at least January 22, 2020.
Although our stock resumed trading upon expiration of the SEC Order, we are unable to predict the outcome of the SEC investigation
or any other actions the SEC may take in connection therewith. Furthermore, the Company’s reputation may be negatively impacted.
As a result, the potential impact to the Company’s business, if any, cannot be determined.
Our bylaws designate the Eighth Judicial
District Court of Clark County, Nevada, as the sole and exclusive forum for certain types of actions and proceedings that may be
initiated by our stockholders, which could limit our stockholders’ ability to obtain a favorable judicial forum for disputes
with us or our directors, officers, employees or agents.
Our bylaws require
that, to the fullest extent permitted by law, and unless the Company consents in writing to the selection of an alternative forum,
the Eighth Judicial District Court of Clark County, Nevada, will, to the fullest extent permitted by law, be the sole and exclusive
forum for each of the following:
|
|
·
|
any derivative action or proceeding brought in the name or right of the Company or on its behalf,
|
|
|
|
|
|
|
·
|
any action asserting a claim for breach of any fiduciary duty owed by any director, officer, employee or agent of the Company to the Company or the Company’s stockholders,
|
|
|
|
|
|
|
·
|
any action arising or asserting a claim arising pursuant to any provision of NRS Chapters 78 or 92A or any provision of our articles of incorporation or bylaws, or
|
|
|
|
|
|
|
·
|
any action asserting a claim governed by the internal affairs doctrine, including, without limitation, any action to interpret, apply, enforce or determine the validity of our articles of incorporation or bylaws.
|
However, our bylaws
provide that the exclusive forum provisions do not apply to suits brought to enforce any liability or duty created by the Securities
Exchange Act of 1934, as amended, or any other claim for which the federal courts have exclusive jurisdiction. We note that there
is uncertainty as to whether a court would enforce the provision and that investors cannot waive compliance with the federal securities
laws and the rules and regulations thereunder. Although we believe this provision benefits us by providing increased consistency
in the application of Nevada law in the types of lawsuits to which it applies, the provision may have the effect of discouraging
lawsuits against our directors and officers.
Risks Related to Our Intellectual Property and Related
Litigation
We rely upon licenses and patent rights from third parties
which are subject to termination or expiration.
We rely in part upon
third-party licenses and ownership rights assigned from third parties for the development of specific uses for our Hemopurifier
devices. For example, we are researching, developing and testing cancer-related applications for our devices under patents assigned
from the London Health Science Center Research, Inc. Should any of our licenses be prematurely terminated for any reason, or if
the patents and intellectual property assigned to us or owned by such entities that we have licensed are challenged or defeated
by third parties, our research efforts could be materially and adversely affected. Our licenses and patents assigned to us may
not continue in force for as long as we require for our research, development and testing of cancer treatments. It is possible
that, if our licenses terminate or the underlying patents and intellectual property is challenged or defeated or the patents and
intellectual property assigned to us is challenged or defeated, suitable replacements may not be obtained or developed on terms
acceptable to us, if at all. There is also the related risk that we may not be able to make the required payments under any patent
license or assignment agreement, in which case we may lose to ability to use one or more of the licensed or assigned patents.
We could become subject to intellectual property litigation
that could be costly, result in the diversion of management’s time and efforts, require us to pay damages, prevent us from
selling our commercially available products and/or reduce the margins we may realize from our products.
The medical devices
industry is characterized by extensive litigation and administrative proceedings over patent and other intellectual property rights.
Whether a product infringes a patent involves complex legal and factual issues, and the determination is often uncertain. There
may be existing patents of which we are unaware that our products under development may inadvertently infringe. The likelihood
that patent infringement claims may be brought against us increases as the number of participants in the infectious market increases
and as we achieve more visibility in the market place and introduce products to market.
Any infringement claim
against us, even if without merit, may cause us to incur substantial costs, and would place a significant strain on our financial
resources, divert the attention of management from our core business, and harm our reputation. In some cases, litigation may be
threatened or brought by a patent holding company or other adverse patent owner who has no relevant product revenues and against
whom our patents may provide little or no deterrence. If we are found to infringe any patents, we could be required to pay substantial
damages, including triple damages if an infringement is found to be willful. We also could be required to pay royalties and could
be prevented from selling our products unless we obtain a license or are able to redesign our products to avoid infringement. We
may not be able to obtain a license enabling us to sell our products on reasonable terms, or at all. If we fail to obtain any required
licenses or make any necessary changes to our technologies or the products, we may be unable to commercialize one or more of our
products or may have to withdraw products from the market, all of which would have a material adverse effect on our business, financial
condition and results of operations.
If the combination of patents, trade secrets and contractual
provisions upon which we rely to protect our intellectual property is inadequate, our ability to commercialize our products successfully
will be harmed.
Our success depends
significantly on our ability to protect our proprietary rights to the technologies incorporated in our products. We currently have
five issued U.S. patents and nine pending U.S. patent applications. We also have 33 issued foreign patents and have applied for
nine additional international patents. Our issued patents begin to expire in 2024, with the last of these patents expiring in 2036,
although terminal disclaimers, patent term extension or patent term adjustment can shorten or lengthen the patent term. We rely
on a combination of patent protection, trade secret laws and nondisclosure, confidentiality and other contractual restrictions
to protect our proprietary technology. However, these may not adequately protect our rights or permit us to gain or keep any competitive
advantage.
The issuance of a patent
is not conclusive as to its scope, validity or enforceability. The scope, validity or enforceability of our issued patents can
be challenged in litigation or proceedings before the U.S. Patent and Trademark Office or foreign patent offices where our applications
are pending. The U.S. Patent and Trademark Office or foreign offices may deny or require significant narrowing of claims in our
pending patent applications. Patents issued as a result of the pending patent applications, if any, may not provide us with significant
commercial protection or be issued in a form that is advantageous to us. Proceedings before the U.S. Patent and Trademark Office
or foreign offices could result in adverse decisions as to the priority of our inventions and the narrowing or invalidation of
claims in issued patents. The laws of some foreign countries may not protect our intellectual property rights to the same extent
as the laws of the U.S., if at all. Some of our patents may expire before we receive FDA approval to market our products in the
U.S. or we receive approval to market our products in a foreign country. Although we believe that certain patent applications and/or
other patents issued more recently will help protect the proprietary nature of the Hemopurifier treatment technology, this protection
may not be sufficient to protect us during the development of that technology.
Our competitors may
successfully challenge and invalidate or render unenforceable our issued patents, including any patents that may issue in the future,
which could prevent or limit our ability to market our products and could limit our ability to stop competitors from marketing
products that are substantially equivalent to ours. In addition, competitors may be able to design around our patents or develop
products that provide outcomes that are comparable to our products but that are not covered by our patents.
We have also entered
into confidentiality and assignment of intellectual property agreements with all of our employees, consultants and advisors directly
involved in the development of our technology as one of the ways we seek to protect our intellectual property and other proprietary
technology. However, these agreements may not be enforceable or may not provide meaningful protection for our trade secrets or
other proprietary information in the event of unauthorized use or disclosure or other breaches of the agreements.
In the event a competitor
infringes upon any of our patents or other intellectual property rights, enforcing our rights may be difficult, time consuming
and expensive, and would divert management’s attention from managing our business. We may not be successful on the merits
in any enforcement effort. In addition, we may not have sufficient resources to litigate, enforce or defend our intellectual property
rights.
We may rely on licenses for new technology, which may
affect our continued operations with respect thereto.
As we develop our technology,
we may need to license additional technologies to optimize the performance of our products. We may not be able to license these
technologies on commercially reasonable terms or at all. In addition, we may fail to successfully integrate any licensed technology
into our proposed products. Our inability to obtain any necessary licenses could delay our product development and testing until
alternative technologies can be identified, licensed and integrated. The inability to obtain any necessary third-party licenses
could cause us to abandon a particular development path, which could seriously harm our business, financial position and results
of our operations.
New technology may lead to our competitors developing
superior products which would reduce demand for our products.
Research into technologies
similar to ours is proceeding at a rapid pace, and many private and public companies and research institutions are actively engaged
in the development of products similar to ours. These new technologies may, if successfully developed, offer significant performance
or price advantages when compared with our technologies. Our existing patents or our pending and proposed patent applications may
not offer meaningful protection if a competitor develops a novel product based on a new technology.
If we are unable to protect our proprietary technology
and preserve our trade secrets, we will increase our vulnerability to competitors which could materially adversely impact our ability
to remain in business.
Our ability to successfully
commercialize our products will depend on our ability to protect those products and our technology with domestic and foreign patents.
We will also need to continue to preserve our trade secrets. The issuance of a patent is not conclusive as to its validity or as
to the enforceable scope of the claims of the patent. The patent positions of technology companies, including us, are uncertain
and involve complex legal and factual issues. Our patents may not prevent other companies from developing similar products or products
which produce benefits substantially the same as our products, and other companies may be issued patents that may prevent the sale
of our products or require us to pay significant licensing fees in order to market our products.
From time to time,
we may need to obtain licenses to patents and other proprietary rights held by third parties in order to develop, manufacture and
market our products. If we are unable to timely obtain these licenses on commercially reasonable terms, our ability to commercially
exploit such products may be inhibited or prevented. Our pending patent applications may not result in issued patents, patent protection
may not be secured for any particular technology, and our issued patents may not be valid or enforceable or provide us with meaningful
protection.
If we are required to engage in expensive and lengthy
litigation to enforce our intellectual property rights, such litigation could be very costly and the results of such litigation
may not be satisfactory.
Although we have entered
into invention assignment agreements with our employees and with certain advisors, and we routinely enter into confidentiality
agreements with our contract partners, if those employees, advisors or contract partners develop inventions or processes independently
that may relate to products or technology under development by us, disputes may arise about the ownership of those inventions or
processes. Time-consuming and costly litigation could be necessary to enforce and determine the scope of our rights under these
agreements. In addition, we may be required to commence litigation to enforce such agreements if they are violated, and it is certainly
possible that we will not have adequate remedies for breaches of our confidentiality agreements as monetary damages may not be
sufficient to compensate us. We may be unable to fund the costs of any such litigation to a satisfactory conclusion, which could
leave us without recourse to enforce contracts that protect our intellectual property rights.
Other companies may claim that our technology infringes
on their intellectual property or proprietary rights and commence legal proceedings against us which could be time-consuming and
expensive and could result in our being prohibited from developing, marketing, selling or distributing our products.
Because of the complex
and difficult legal and factual questions that relate to patent positions in our industry, it is possible that our products or
technology could be found to infringe upon the intellectual property or proprietary rights of others. Third parties may claim that
our products or technology infringe on their patents, copyrights, trademarks or other proprietary rights and demand that we cease
development or marketing of those products or technology or pay license fees. We may not be able to avoid costly patent infringement
litigation, which will divert the attention of management away from the development of new products and the operation of our business.
We may not prevail in any such litigation. If we are found to have infringed on a third-party’s intellectual property rights,
we may be liable for money damages, encounter significant delays in bringing products to market or be precluded from manufacturing
particular products or using particular technology.
Other parties may challenge
certain of our foreign patent applications. If any such parties are successful in opposing our foreign patent applications, we
may not gain the protection afforded by those patent applications in particular jurisdictions and may face additional proceedings
with respect to similar patents in other jurisdictions, as well as related patents. The loss of patent protection in one jurisdiction
may influence our ability to maintain patent protection for the same technology in other jurisdictions.
Risks Related to U.S. Government Contracts
We may not obtain additional U.S. Government contracts
to further develop our technology.
We may not be successful
in obtaining additional government grants or contracts. The process of obtaining government contracts is lengthy with the uncertainty
that we will be successful in obtaining announced grants or contracts for therapeutics as a medical device technology. Accordingly,
we may not be awarded any additional U.S. Government grants or contracts utilizing our Hemopurifier platform technology.
U.S. Government agencies have special contracting requirements,
including a right to audit us which create additional risks; a negative audit would be detrimental to us.
Our business plan to
utilize the Aethlon Hemopurifier technology is likely to continue to involve contracts with the U.S. Government, such as our contract
with the National Institute of Health and the National Cancer Institute, effective September 12, 2019. Contracts such as this one,
typically contain unfavorable termination provisions and are subject to audit and modification by the government at its sole discretion,
which subjects us to additional risks. These risks include the ability of the U.S. Government to unilaterally:
|
|
·
|
suspend or prevent us for a period of time from receiving new contracts or extending existing contracts based on violations or suspected violations of laws or regulations;
|
|
|
·
|
audit and object to our contract-related costs and fees, including allocated indirect costs;
|
|
|
·
|
control and potentially prohibit the export of our products; and
|
|
|
·
|
change certain terms and conditions in our contracts.
|
As a U.S. Government
contractor, we are required to comply with applicable laws, regulations and standards relating to our accounting practices and
would be subject to periodic audits and reviews. As part of any such audit or review, the U.S. Government may review the adequacy
of, and our compliance with, our internal control systems and policies, including those relating to our purchasing, property, estimating,
compensation and management information systems. Based on the results of its audits, the U.S. Government may adjust our contract-related
costs and fees, including allocated indirect costs. In addition, if an audit or review uncovers any improper or illegal activity,
we would possibly be subject to civil and criminal penalties and administrative sanctions, including termination of our contracts,
forfeiture of profits, suspension of payments, fines and suspension or prohibition from doing business with the U.S. Government.
We could also suffer serious harm to our reputation if allegations of impropriety were made against us. Although we have not had
any government audits and reviews to date, future audits and reviews could cause adverse effects. In addition, under U.S. Government
purchasing regulations, some of our costs, including most financing costs, amortization of intangible assets, portions of our research
and development costs, and some marketing expenses, would possibly not be reimbursable or allowed under such contracts. Further,
as a U.S. Government contractor, we would be subject to an increased risk of investigations, criminal prosecution, civil fraud,
whistleblower lawsuits and other legal actions and liabilities.
As a U.S. Government contractor, we are subject to a number
of procurement rules and regulations.
Government contractors
must comply with specific procurement regulations and other requirements. These requirements, although customary in government
contracts, impact our performance and compliance costs. In addition, current U.S. Government budgetary constraints could lead to
changes in the procurement environment, including the Department of Defense’s recent initiative focused on efficiencies,
affordability and cost growth and other changes to its procurement practices. If and to the extent such changes occur, they could
impact our results of operations and liquidity, and could affect whether and, if so, how we pursue certain opportunities and the
terms under which we are able to do so.
In addition, failure
to comply with these regulations and requirements could result in reductions of the value of contracts, contract modifications
or termination, and the assessment of penalties and fines, which could negatively impact our results of operations and financial
condition. Our failure to comply with these regulations and requirements could also lead to suspension or debarment, for cause,
from government contracting or subcontracting for a period of time. Among the causes for debarment are violations of various statutes,
including those related to procurement integrity, export control, government security regulations, employment practices, protection
of the environment, accuracy of records and the recording of costs, and foreign corruption. The termination of our government contract
as a result of any of these acts could have a negative impact on our results of operations and financial condition and could have
a negative impact on our reputation and ability to procure other government contracts in the future.
Risks Relating to Our Common Stock and Our Corporate Governance
Our failure to meet the continued
listing requirements of The Nasdaq Capital Market could result in a de-listing of our common stock.
If we fail to satisfy
the continued listing requirements of The Nasdaq Capital Market, or Nasdaq, such as the minimum stockholders’ equity requirement
or the minimum closing bid price requirement, Nasdaq may take steps to de-list our common stock. For example, in May 2019 we received
a letter from Nasdaq indicating that Nasdaq has determined that we have failed to comply with the minimum bid price requirement
of Nasdaq Listing Rule 5550(a)(2). Nasdaq Listing Rule 5550(a)(2) requires that companies listed on the Nasdaq Capital Market maintain
a minimum closing bid price of at least $1.00 per share. In July 2019, we received another letter from Nasdaq indicating that
Nasdaq has determined that we have failed to comply with the minimum stockholder’s equity requirement of Nasdaq Listing Rule
5550(b)(1). Nasdaq Listing Rule 5550(b)(1) requires that companies listed on the Nasdaq Capital Market maintain a minimum of $2,500,000
in stockholder’s equity. If we fail to maintain compliance with these, or any other of the continued listing requirements
of The Nasdaq Capital Market, Nasdaq may take steps to de-list our common stock. Such a de-listing would likely have a negative
effect on the price of our common stock and would impair your ability to sell or purchase our common stock when you wish to do
so. In the event of a de-listing, we would take actions to restore our compliance with Nasdaq’s listing requirements, but
any such action taken by us may not be successful.
Historically we have not paid dividends
on our common stock, and we do not anticipate paying any cash dividends in the foreseeable future.
We have never paid
cash dividends on our common stock. We intend to retain our future earnings, if any, to fund operational and capital expenditure
needs of our business, and do not anticipate paying any cash dividends in the foreseeable future. As a result, capital appreciation,
if any, of our common stock will be the sole source of gain for our common stockholders in the foreseeable future.
Our stock price is speculative, and there is a risk of
litigation.
The trading price of
our common stock has in the past and may in the future be subject to wide fluctuations in response to factors such as the following:
|
|
·
|
failure to raise additional funds when needed;
|
|
|
|
|
|
|
·
|
announcements regarding our ongoing development of the Hemopurifier;
|
|
|
|
|
|
|
·
|
results from our clinical trials with the Hemopurifier;
|
|
|
·
|
failure to maintain our listing on Nasdaq;
|
|
|
·
|
results of operations or revenue in any quarter failing to meet the expectations, published or otherwise, of the investment community;
|
|
|
·
|
reduced investor confidence in equity markets;
|
|
|
·
|
speculation in the press or analyst community;
|
|
|
·
|
wide fluctuations in stock prices, particularly with respect to the stock prices for other medical device companies;
|
|
|
·
|
announcements of technological innovations by us or our competitors;
|
|
|
·
|
new products or the acquisition of significant customers by us or our competitors;
|
|
|
·
|
changes in interest rates;
|
|
|
·
|
changes in investors’ beliefs as to the appropriate price-earnings ratios for us and our competitors;
|
|
|
·
|
changes in recommendations or financial estimates by securities analysts who track our common stock or the stock of other medical device companies;
|
|
|
·
|
sales of common stock by directors and executive officers;
|
|
|
·
|
rumors or dissemination of false or misleading information, particularly through Internet chat rooms, instant messaging, and other rapid-dissemination methods;
|
|
|
·
|
conditions and trends in the medical device industry generally;
|
|
|
·
|
the announcement of acquisitions or other significant transactions by us or our competitors;
|
|
|
·
|
adoption of new accounting standards affecting our industry;
|
|
|
|
|
|
|
·
|
changes in the structure of healthcare payment systems;
|
|
|
·
|
general market conditions;
|
|
|
·
|
domestic or international terrorism and other factors, including the effects of the ongoing pandemic; and
|
|
|
·
|
the other factors described in this section.
|
Fluctuations in the
price of our common stock may expose us to the risk of securities class action lawsuits. Although no such lawsuits are currently
pending against us and we are not aware that any such lawsuit is threatened to be filed in the future, future lawsuits are possible
as a result of fluctuations in the price of our common stock. Defending against any such suits could result in substantial cost
and divert management’s attention and resources. In addition, any settlement or adverse determination of such lawsuits could
subject us to significant liability.
If at any time our common stock
is subject to the SEC’s penny stock rules, broker-dealers may experience difficulty in completing customer transactions
and trading activity in our securities may be adversely affected.
If at any time our
common stock is not listed on a national securities exchange or we have net tangible assets of $2,000,000 or less, or we have
an average revenue of less than $6,000,000 for the last three years, and our common stock has a market price per share of less
than $5.00, transactions in our common stock will be subject to the SEC’s “penny stock” rules. If our common
stock is subject to the “penny stock” rules promulgated under the Exchange Act, broker-dealers may find it difficult
to effectuate customer transactions and trading activity in our securities may be adversely affected. For any transaction involving
a penny stock, unless exempt, the rules require:
|
|
·
|
that a broker or dealer approve a person’s account for transactions in penny stocks;
|
|
|
·
|
furnish the investor a disclosure document describing the risks of investing in penny stocks;
|
|
|
·
|
disclose to the investor the current market quotation, if any, for the penny stock;
|
|
|
·
|
disclose to the investor the amount of compensation the firm and its broker will receive for the trade; and
|
|
|
·
|
The broker or dealer receive from the investor a written agreement to the transaction, setting forth the identity and quantity of the penny stock to be purchased.
|
In order to approve
a person’s account for transactions in penny stocks, the broker or dealer must:
|
|
·
|
obtain financial information and investment experience objectives of the person; and
|
|
|
·
|
make a reasonable determination that the transactions in penny stocks are suitable for that person and the person has sufficient knowledge and experience in financial matters to be capable of evaluating the risks of transactions in penny stocks.
|
The broker or dealer
must also deliver, prior to any transaction in a penny stock, a disclosure schedule prescribed by the SEC relating to the penny
stock market, which, in highlight form:
|
|
·
|
sets forth the basis on which the broker or dealer made the suitability determination; and
|
|
|
·
|
that the broker or dealer received a signed, written agreement from the investor prior to the transaction.
|
Generally, brokers
may be less willing to execute transactions in securities subject to the “penny stock” rules. This may make it more
difficult for investors to dispose of our common stock and cause a decline in the market value of our stock.
Disclosure also has
to be made about the risks of investing in penny stocks in both public offerings and in secondary trading and about the commissions
payable to both the broker-dealer and the registered representative, current quotations for the securities and the rights and remedies
available to an investor in cases of fraud in penny stock transactions. Finally, monthly statements have to be sent disclosing
recent price information for the penny stock held in the account and information on the limited market in penny stocks.
Our common stock has had an unpredictable trading volume
which means you may not be able to sell our shares at or near trading prices or at all.
Trading in our common
shares historically has been volatile and often has been thin, meaning that the number of persons interested in purchasing our
common shares at or near trading prices at any given time may be relatively small or non-existent. This situation is attributable
to a number of factors, including the fact that we are a small company which is relatively unknown to stock analysts, stock brokers,
institutional investors and others in the investment community that generate or influence sales volume, and that even if we came
to the attention of such persons, they tend to be risk-averse and would be reluctant to follow an unproven company such as ours
or purchase or recommend the purchase of our shares until such time as we became more seasoned and viable. As a consequence, there
may be periods of several days or more when trading activity in our shares is minimal, as compared to a seasoned issuer which has
a large and steady volume of trading activity that will generally support continuous sales without an adverse effect on share price.
A broader or more active public trading market for our common shares may not develop or be sustained, and current trading levels
may decrease.
The market price for our common stock is volatile; you
may not be able to sell our common stock at or above the price you have paid for them, which may result in losses to you.
The market for our
common stock is characterized by significant price volatility when compared to seasoned issuers, and we expect that our share price
will continue to be more volatile than a seasoned issuer for the indefinite future. During the 52-week period ended March 31, 2020,
the high and low closing sale prices for a share of our common stock were $13.74 and $0.84, respectively. The volatility in our
share price is attributable to a number of factors. First, as noted above, trading in our common stock often has been thin. As
a consequence of this lack of liquidity, the trading of relatively small quantities of shares by our stockholders may disproportionately
influence the price of those shares in either direction. The price for our shares could, for example, decline precipitously in
the event that a large number of our common shares are sold on the market without commensurate demand, as compared to a seasoned
issuer which could better absorb those sales without adverse impact on its share price. Secondly, we are a speculative investment
due to our limited operating history, limited amount of cash and revenue, lack of profit to date, and the uncertainty of future
market acceptance for our potential products. As a consequence of this enhanced risk, more risk-adverse investors may, under the
fear of losing all or most of their investment in the event of negative news or lack of progress, be more inclined to sell their
shares on the market more quickly and at greater discounts than would be the case with the stock of a seasoned issuer.
The following factors
also may add to the volatility in the price of our common stock: actual or anticipated variations in our quarterly or annual operating
results; announcements regarding our clinical trials and the development of the Hemopurifier; acceptance of our proprietary technology
as a viable method of augmenting the immune response of clearing viruses and toxins from human blood; government regulations, announcements
of significant acquisitions, strategic partnerships or joint ventures; our capital commitments and additions or departures of our
key personnel. Many of these factors are beyond our control and may decrease the market price of our common shares regardless of
our operating performance. We cannot make any predictions or projections as to what the prevailing market price for our common
shares will be at any time, including as to whether our common shares will sustain their current market prices, or as to what effect
the sale of shares or the availability of common shares for sale at any time will have on the prevailing market price.
Our issuance of additional shares of common stock or convertible
securities, could be dilutive.
We are entitled under
our articles of incorporation to issue up to 30,000,000 shares of common stock. We have reserved for issuance 2,072,492 of those
shares of common stock for outstanding restricted stock units, stock options and warrants. As of March 31, 2020, we had issued
and outstanding 9,367,171 shares of common stock. As a result, as of March 31, 2020 we had 18,560,337 shares of common stock available
for issuance to new investors or for use to satisfy indebtedness or pay service providers.
Our Board of Directors
may generally issue shares of common stock, restricted stock units or stock options or warrants to purchase those shares, without
further approval by our stockholders, based upon such factors as our Board of Directors may deem relevant at that time. It is likely
that we will be required to issue a large amount of additional securities to raise capital to further our development. It is also
likely that we will be required to issue a large amount of additional securities to directors, officers, employees and consultants
as compensatory grants in connection with their services, both in the form of stand-alone grants or under our stock plans.
Our officers and directors are entitled to indemnification
from us for liabilities under our articles of incorporation, which could be costly to us and may discourage the exercise of stockholder
rights.
Our articles of incorporation
provide that we possess and may exercise all powers of indemnification of our officers, directors, employees, agents and other
persons and our bylaws also require us to indemnify our officers and directors as permitted under the provisions of the Nevada
Revised Statutes, or NRS. We may also have contractual indemnification obligations under our agreements with our directors, officers
and employees. The foregoing indemnification obligations could result in our company incurring substantial expenditures to cover
the cost of settlement or damage awards against directors and officers. These provisions and resultant costs may also discourage
our company from bringing a lawsuit against directors, officers and employees for breaches of their fiduciary duties, and may similarly
discourage the filing of derivative litigation by our stockholders against our directors, officers and employees even though such
actions, if successful, might otherwise benefit our company and stockholders.
Our bylaws and Nevada law may discourage, delay or prevent
a change of control of our company or changes in our management, would have the result of depressing the trading price of our common
stock.
Certain anti-takeover
provisions of Nevada law could have the effect of delaying or preventing a third-party from acquiring us, even if the acquisition
arguably could benefit our stockholders.
Nevada’s “combinations
with interested stockholders” statutes (NRS 78.411 through 78.444, inclusive) prohibit specified types of business “combinations”
between certain Nevada corporations and any person deemed to be an “interested stockholder” for two years after such
person first becomes an “interested stockholder” unless the corporation’s board of directors approves the combination
(or the transaction by which such person becomes an “interested stockholder”) in advance, or unless the combination
is approved by the board of directors and sixty percent of the corporation’s voting power not beneficially owned by the interested
stockholder, its affiliates and associates. Further, in the absence of prior approval certain restrictions may apply even after
such two year period. However, these statutes do not apply to any combination of a corporation and an interested stockholder after
the expiration of four years after the person first became an interested stockholder. For purposes of these statutes, an “interested
stockholder” is any person who is (1) the beneficial owner, directly or indirectly, of ten percent or more of the voting
power of the outstanding voting shares of the corporation, or (2) an affiliate or associate of the corporation and at any time
within the two previous years was the beneficial owner, directly or indirectly, of ten percent or more of the voting power of the
then outstanding shares of the corporation. The definition of the term “combination” is sufficiently broad to cover
most significant transactions between a corporation and an “interested stockholder.” A Nevada corporation may elect
in its articles of incorporation not to be governed by these particular laws, but if such election is not made in the corporation’s
original articles of incorporation, the amendment (1) must be approved by the affirmative vote of the holders of stock representing
a majority of the outstanding voting power of the corporation not beneficially owned by interested stockholders or their affiliates
and associates, and (2) is not effective until 18 months after the vote approving the amendment and does not apply to any combination
with a person who first became an interested stockholder on or before the effective date of the amendment. We did not make such
an election in our original articles of incorporation and have not amended our articles of incorporation to so elect.
Nevada’s “acquisition
of controlling interest” statutes (NRS 78.378 through 78.3793, inclusive) contain provisions governing the acquisition of
a controlling interest in certain Nevada corporations. These “control share” laws provide generally that any person
that acquires a “controlling interest” in certain Nevada corporations may be denied voting rights, unless a majority
of the disinterested stockholders of the corporation elects to restore such voting rights. These laws would apply to us if we were
to have 200 or more stockholders of record (at least 100 of whom have addresses in Nevada appearing on our stock ledger) and do
business in the State of Nevada directly or through an affiliated corporation, unless our articles of incorporation or bylaws in
effect on the tenth day after the acquisition of a controlling interest provide otherwise. These laws provide that a person acquires
a “controlling interest” whenever a person acquires shares of a subject corporation that, but for the application of
these provisions of the NRS, would enable that person to exercise (1) one fifth or more, but less than one third, (2) one third
or more, but less than a majority or (3) a majority or more, of all of the voting power of the corporation in the election of directors.
Once an acquirer crosses one of these thresholds, shares which it acquired in the transaction taking it over the threshold and
within the 90 days immediately preceding the date when the acquiring person acquired or offered to acquire a controlling interest
become “control shares” to which the voting restrictions described above apply. These laws may have a chilling effect
on certain transactions if our articles of incorporation or bylaws are not amended to provide that these provisions do not apply
to us or to an acquisition of a controlling interest, or if our disinterested stockholders do not confer voting rights in the control
shares.
Various provisions
of our bylaws may delay, defer or prevent a tender offer or takeover attempt of us that a stockholder might consider in his or
her best interest. Our bylaws may be adopted, amended or repealed by the affirmative vote of the holders of at least a majority
of our outstanding shares of capital stock entitled to vote for the election of directors, and except as provided by Nevada law,
our Board of Directors shall have the power to adopt, amend or repeal the bylaws by a vote of not less than a majority of our directors.
The interests of these stockholders and directors may not be consistent with your interests, and they may make changes to the bylaws
that are not in line with your concerns.
Nevada law also provides
that directors may resist a change or potential change in control if the directors determine that the change is opposed to, or
not in the best interests of, the corporation. The existence of the foregoing provisions and other potential anti-takeover measures
could limit the price that investors might be willing to pay in the future for shares of our common stock. They could also deter
potential acquirers of our company, thereby reducing the likelihood that you could receive a premium for your common stock in an
acquisition.
We incur substantial costs as a result of being a public
company and our management expects to devote substantial time to public company compliance programs.
As a public company,
we incur significant legal, insurance, accounting and other expenses, including costs associated with public company reporting.
We intend to invest resources to comply with evolving laws, regulations and standards, and this investment will result in increased
general and administrative expenses and may divert management’s time and attention from product development and commercialization
activities. If our efforts to comply with new laws, regulations and standards differ from the activities intended by regulatory
or governing bodies due to ambiguities related to practice, regulatory authorities may initiate legal proceedings against us, and
our business may be harmed. These laws and regulations could make it more difficult and costly for us to obtain director and officer
liability insurance for our directors and officers, and we may be required to accept reduced coverage or incur substantially higher
costs to obtain coverage. These factors could also make it more difficult for us to attract and retain qualified executive officers
and qualified members of our Board of Directors, particularly to serve on our audit and compensation committees. In addition, if
we are unable to continue to meet the legal, regulatory and other requirements related to being a public company, we may not be
able to maintain the quotation of our common stock on the Nasdaq Capital Market or on any other senior market to which we may apply
for listing, which would likely have a material adverse effect on the trading price of our common stock.
If securities or industry analysts do not publish research
or reports about our business, or if they change their recommendations regarding our stock adversely, our stock price and trading
volume could decline.
The trading market
for our common stock will be influenced by the research and reports that industry or securities analysts publish about us or our
business. Our research coverage by industry and financial analysts is currently limited. Even if our analyst coverage increases,
if one or more of the analysts who cover us downgrade our stock, our stock price would likely decline. If one or more of these
analysts cease coverage of our company or fail to regularly publish reports on us, we could lose visibility in the financial markets,
which in turn could cause our stock price or trading volume to decline.
ITEM 1B. UNRESOLVED STAFF COMMENTS
None.
ITEM 2. PROPERTIES
We currently lease approximately 2,600 square
feet of executive office space at 9635 Granite Ridge Drive, Suite 100, San Diego, California 92123 under a 39-month gross
plus utilities lease that commenced on December 1, 2014 and expires on August 31, 2021. The current rental rate under the lease
extension is $8,265 per month. We believe this leased facility will be satisfactory for our office needs over the term of the lease.
We also rent approximately 1,700 square
feet of laboratory space at 11585 Sorrento Valley Road, Suite 109, San Diego, California 92121 at the rate of $4,700 per month
on a one-year lease that originally was to expire on November 30, 2019. In October 2019, we entered into a lease extension for
an additional twelve months running from December 1, 2019 through November 30, 2020, at the rate of $5,961 per month.
ITEM 3. LEGAL PROCEEDINGS
We may be involved
from time to time in various claims, lawsuits, and/or disputes with third parties or breach of contract actions incidental to the
normal course of our business operations. We are currently not involved in any litigation or any pending legal proceedings.
ITEM 4. MINE SAFETY DISCLOSURES
We have no disclosure applicable to this
item.
AETHLON MEDICAL, INC. AND SUBSIDIARY
CONSOLIDATED BALANCE
SHEETS
|
|
|
March 31, 2020
|
|
|
March 31, 2019
|
|
|
ASSETS
|
|
|
|
|
|
|
|
|
|
CURRENT ASSETS
|
|
|
|
|
|
|
|
|
|
Cash
|
|
$
|
9,604,780
|
|
|
$
|
3,828,074
|
|
|
Accounts receivable
|
|
|
206,729
|
|
|
|
–
|
|
|
Prepaid expenses and other current assets
|
|
|
229,604
|
|
|
|
210,042
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL CURRENT ASSETS
|
|
|
10,041,113
|
|
|
|
4,038,116
|
|
|
|
|
|
|
|
|
|
|
|
|
Property and equipment, net
|
|
|
140,484
|
|
|
|
6,021
|
|
|
Right-of-use lease asset
|
|
|
136,426
|
|
|
|
–
|
|
|
Patents, net
|
|
|
57,504
|
|
|
|
66,668
|
|
|
Deposits
|
|
|
12,159
|
|
|
|
12,159
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL ASSETS
|
|
$
|
10,387,686
|
|
|
$
|
4,122,964
|
|
|
|
|
|
|
|
|
|
|
|
|
LIABILITIES AND STOCKHOLDERS’ EQUITY
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CURRENT LIABILITIES
|
|
|
|
|
|
|
|
|
|
Accounts payable
|
|
$
|
285,036
|
|
|
$
|
131,931
|
|
|
Due to related parties
|
|
|
111,707
|
|
|
|
83,654
|
|
|
Convertible notes payable, net
|
|
|
–
|
|
|
|
962,301
|
|
|
Deferred revenue
|
|
|
100,000
|
|
|
|
–
|
|
|
Lease liability, current portion
|
|
|
98,557
|
|
|
|
–
|
|
|
Other current liabilities
|
|
|
472,420
|
|
|
|
646,000
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL CURRENT LIABILITIES
|
|
|
1,067,720
|
|
|
|
1,823,886
|
|
|
|
|
|
|
|
|
|
|
|
|
Lease liability, less current portion
|
|
|
42,540
|
|
|
|
–
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL LIABILITIES
|
|
|
1,110,260
|
|
|
|
1,823,886
|
|
|
|
|
|
|
|
|
|
|
|
|
COMMITMENTS AND CONTINGENCIES (Note 12)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
STOCKHOLDERS’ EQUITY
|
|
|
|
|
|
|
|
|
|
Common stock, $0.001 par value, 30,000,000 shares authorized at March 31, 2020 and 2019; 9,367,171 and 1,266,979 issued and outstanding at March 31, 2020 and 2019, respectively
|
|
|
9,368
|
|
|
|
1,267
|
|
|
Additional paid-in capital
|
|
|
121,426,563
|
|
|
|
108,076,275
|
|
|
Accumulated deficit
|
|
|
(112,026,381
|
)
|
|
|
(105,652,433
|
)
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL AETHLON MEDICAL, INC. STOCKHOLDERS’ EQUITY BEFORE NONCONTROLLING INTERESTS
|
|
|
9,409,550
|
|
|
|
2,425,109
|
|
|
|
|
|
|
|
|
|
|
|
|
NONCONTROLLING INTERESTS
|
|
|
(132,124
|
)
|
|
|
(126,031
|
)
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL STOCKHOLDERS’ EQUITY
|
|
|
9,277,426
|
|
|
|
2,299,078
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL LIABILITIES AND STOCKHOLDERS’ EQUITY
|
|
$
|
10,387,686
|
|
|
$
|
4,122,964
|
|
See accompanying notes to the consolidated
financial statements.
AETHLON MEDICAL, INC. AND SUBSIDIARY
CONSOLIDATED STATEMENTS
OF OPERATIONS
|
|
|
Years Ended March 31,
|
|
|
|
|
2020
|
|
|
2019
|
|
|
REVENUES:
|
|
|
|
|
|
|
|
Government contract and grant revenue
|
|
$
|
650,187
|
|
|
$
|
229,625
|
|
|
Total revenues
|
|
|
650,187
|
|
|
|
229,625
|
|
|
|
|
|
|
|
|
|
|
|
|
OPERATING COSTS AND EXPENSES
|
|
|
|
|
|
|
|
|
|
Professional fees
|
|
|
2,729,025
|
|
|
|
2,192,048
|
|
|
Payroll and related expenses
|
|
|
2,302,599
|
|
|
|
3,083,116
|
|
|
General and administrative
|
|
|
1,548,551
|
|
|
|
953,478
|
|
|
Total operating expenses
|
|
|
6,580,175
|
|
|
|
6,228,642
|
|
|
|
|
|
|
|
|
|
|
|
|
OPERATING LOSS
|
|
|
(5,929,988
|
)
|
|
|
(5,999,017
|
)
|
|
|
|
|
|
|
|
|
|
|
|
OTHER EXPENSE
|
|
|
|
|
|
|
|
|
|
Loss on debt extinguishment
|
|
|
447,011
|
|
|
|
–
|
|
|
(Gain) on share for warrant exchanges
|
|
|
(51,190
|
)
|
|
|
–
|
|
|
Interest and other expenses
|
|
|
54,232
|
|
|
|
220,487
|
|
|
Total other expense
|
|
|
450,053
|
|
|
|
220,487
|
|
|
|
|
|
|
|
|
|
|
|
|
NET LOSS BEFORE NONCONTROLLING INTERESTS
|
|
|
(6,380,041
|
)
|
|
|
(6,219,504
|
)
|
|
|
|
|
|
|
|
|
|
|
|
LOSS ATTRIBUTABLE TO NONCONTROLLING INTERESTS
|
|
|
(6,093
|
)
|
|
|
(24,785
|
)
|
|
|
|
|
|
|
|
|
|
|
|
NET LOSS ATTRIBUTABLE TO COMMON STOCKHOLDERS
|
|
$
|
(6,373,948
|
)
|
|
$
|
(6,194,719
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Basic and diluted net loss per share attributable to common stockholders
|
|
$
|
(1.87
|
)
|
|
$
|
(5.13
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average number of common shares outstanding - basic and diluted
|
|
|
3,414,840
|
|
|
|
1,208,314
|
|
See accompanying notes to the consolidated
financial statements.
AETHLON MEDICAL, INC. AND SUBSIDIARY
CONSOLIDATED STATEMENTS
OF EQUITY
FOR THE YEARS ENDED MARCH 31, 2020 AND 2019
|
|
|
ATTRIBUTABLE
TO AETHLON MEDICAL, INC.
|
|
|
|
|
|
|
|
|
|
|
COMMON
STOCK
|
|
|
ADDITIONAL
PAID IN
|
|
|
ACCUMULATED
|
|
|
NON-
CONTROLLING
|
|
|
TOTAL
|
|
|
|
|
SHARES
|
|
|
AMOUNT
|
|
|
CAPITAL
|
|
|
DEFICIT
|
|
|
INTERESTS
|
|
|
EQUITY
|
|
|
BALANCE - MARCH 31, 2018
|
|
|
1,182,630
|
|
|
$
|
1,182
|
|
|
$
|
105,590,572
|
|
|
$
|
(99,457,714
|
)
|
|
$
|
(101,246
|
)
|
|
$
|
6,032,794
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Issuances of common stock for cash under at the
market program
|
|
|
51,581
|
|
|
|
52
|
|
|
|
1,048,319
|
|
|
|
–
|
|
|
|
–
|
|
|
|
1,048,371
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Issuances of common stock for cash under warrant
exercises
|
|
|
18,887
|
|
|
|
19
|
|
|
|
292,913
|
|
|
|
–
|
|
|
|
–
|
|
|
|
292,932
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Issuance of common shares upon vesting of restricted
stock units.
|
|
|
12,881
|
|
|
|
13
|
|
|
|
(193,879
|
)
|
|
|
–
|
|
|
|
–
|
|
|
|
(193,866
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Common stock issued for services
|
|
|
1,000
|
|
|
|
1
|
|
|
|
19,349
|
|
|
|
–
|
|
|
|
–
|
|
|
|
19,350
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stock-based compensation expense
|
|
|
–
|
|
|
|
–
|
|
|
|
1,319,001
|
|
|
|
–
|
|
|
|
–
|
|
|
|
1,319,001
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
(6,194,719
|
)
|
|
|
(24,785
|
)
|
|
|
(6,219,504
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
BALANCE - MARCH 31, 2019
|
|
|
1,266,979
|
|
|
$
|
1,267
|
|
|
$
|
108,076,275
|
|
|
$
|
(105,652,433
|
)
|
|
$
|
(126,031
|
)
|
|
$
|
2,299,078
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Issuances of common stock for cash under at the
market program
|
|
|
161,149
|
|
|
|
162
|
|
|
|
895,869
|
|
|
|
–
|
|
|
|
–
|
|
|
|
896,031
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss on debt extinguishment
|
|
|
–
|
|
|
|
–
|
|
|
|
447,011
|
|
|
|
–
|
|
|
|
–
|
|
|
|
447,011
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Issuances of common stock for cash under warrant
exercises
|
|
|
2,700,000
|
|
|
|
2,700
|
|
|
|
3,804,462
|
|
|
|
–
|
|
|
|
–
|
|
|
|
3,807,162
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Issuance of common shares upon vesting of restricted
stock units.
|
|
|
12,393
|
|
|
|
12
|
|
|
|
(40,950
|
)
|
|
|
–
|
|
|
|
–
|
|
|
|
(40,938
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Proceeds from the issuance of common stock in public
offerings, net
|
|
|
5,218,712
|
|
|
|
5,219
|
|
|
|
7,451,096
|
|
|
|
–
|
|
|
|
–
|
|
|
|
7,456,315
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Issuances of common stock upon warrant exchanges
|
|
|
3,992
|
|
|
|
4
|
|
|
|
(51,194
|
)
|
|
|
–
|
|
|
|
–
|
|
|
|
(51,190
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Par value of DTC roundup of shares following reverse split
|
|
|
3,946
|
|
|
|
4
|
|
|
|
(4
|
)
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stock-based compensation expense
|
|
|
–
|
|
|
|
–
|
|
|
|
843,998
|
|
|
|
–
|
|
|
|
–
|
|
|
|
843,998
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
(6,373,948
|
)
|
|
|
(6,093
|
)
|
|
|
(6,380,041
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
BALANCE - MARCH 31, 2020
|
|
|
9,367,171
|
|
|
$
|
9,368
|
|
|
$
|
121,426,563
|
|
|
$
|
(112,026,381
|
)
|
|
$
|
(132,124
|
)
|
|
$
|
9,277,426
|
|
See accompanying notes to the consolidated
financial statements.
AETHLON MEDICAL, INC. AND SUBSIDIARY
CONSOLIDATED STATEMENTS
OF CASH FLOWS
FOR THE YEARS ENDED MARCH 31, 2020 AND 2019
|
|
|
2020
|
|
|
2019
|
|
|
Cash flows from operating activities:
|
|
|
|
|
|
|
|
|
|
Net loss
|
|
$
|
(6,380,041
|
)
|
|
$
|
(6,219,504
|
)
|
|
Adjustments to reconcile net loss to net cash used in operating activities:
|
|
|
|
|
|
|
|
|
|
Depreciation and amortization
|
|
|
26,366
|
|
|
|
30,695
|
|
|
Gain on share for warrant exchanges
|
|
|
(51,190
|
)
|
|
|
–
|
|
|
Loss on debt extinguishment
|
|
|
447,011
|
|
|
|
–
|
|
|
Stock based compensation
|
|
|
843,998
|
|
|
|
1,319,001
|
|
|
Amortization of debt discount and deferred financing costs
|
|
|
30,287
|
|
|
|
121,148
|
|
|
Non-cash rent expense
|
|
|
964
|
|
|
|
–
|
|
|
Fair market value of common stock issued for services
|
|
|
–
|
|
|
|
19,350
|
|
|
|
|
|
|
|
|
|
|
|
|
Changes in operating assets and liabilities:
|
|
|
|
|
|
|
|
|
|
Accounts receivable
|
|
|
(206,729
|
)
|
|
|
74,813
|
|
|
Prepaid expenses and other current assets
|
|
|
(19,562
|
)
|
|
|
(28,675
|
)
|
|
Other assets
|
|
|
–
|
|
|
|
6,111
|
|
|
Accounts payable and other current liabilities
|
|
|
(16,765
|
)
|
|
|
390,340
|
|
|
Deferred revenue
|
|
|
100,000
|
|
|
|
–
|
|
|
Due to related parties
|
|
|
28,053
|
|
|
|
(6,712
|
)
|
|
Net cash used in operating activities
|
|
|
(5,197,608
|
)
|
|
|
(4,293,433
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows from investing activities:
|
|
|
|
|
|
|
|
|
|
Purchases of property and equipment
|
|
|
(151,665
|
)
|
|
|
–
|
|
|
Net cash used in investing activities
|
|
|
(151,665
|
)
|
|
|
–
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows from financing activities:
|
|
|
|
|
|
|
|
|
|
Tax withholding payments or tax equivalent payments for net share settlement of restricted stock units
|
|
|
(40,938
|
)
|
|
|
(193,866
|
)
|
|
Principal repayments of notes payable
|
|
|
(992,591
|
)
|
|
|
–
|
|
|
Net proceeds from the issuance of common stock and exercise of warrants
|
|
|
12,159,508
|
|
|
|
1,341,303
|
|
|
Net cash provided by financing activities
|
|
|
11,125,979
|
|
|
|
1,147,437
|
|
|
|
|
|
|
|
|
|
|
|
|
Net increase (decrease) in cash
|
|
|
5,776,706
|
|
|
|
(3,145,996
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Cash at beginning of year
|
|
|
3,828,074
|
|
|
|
6,974,070
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash at end of year
|
|
$
|
9,604,780
|
|
|
$
|
3,828,074
|
|
|
|
|
|
|
|
|
|
|
|
|
Supplemental disclosures of cash flow information:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash paid during the period for:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest
|
|
$
|
–
|
|
|
$
|
95,388
|
|
|
|
|
|
|
|
|
|
|
|
|
Supplemental information of non-cash investing and financing activities:
|
|
|
|
|
|
|
|
|
|
Issuance of shares under vested restricted stock units
|
|
$
|
12
|
|
|
$
|
13
|
|
|
Issuance of common stock upon warrant exchanges
|
|
$
|
51,190
|
|
|
$
|
–
|
|
|
Initial recognition of right-of-use lease asset and lease liability
|
|
$
|
228,694
|
|
|
$
|
–
|
|
See accompanying notes to the consolidated
financial statements.
Aethlon Medical, Inc. and Subsidiary
Notes to Consolidated
Financial Statements
1. ORGANIZATION, LIQUIDITY AND SUMMARY OF SIGNIFICANT ACCOUNTING
POLICIES
ORGANIZATION
Aethlon Medical, Inc. and its subsidiary
(collectively, “Aethlon”, the “Company”, “we” or “us”), is a medical technology
company focused on developing products to diagnose and treat life and organ threatening diseases. The Aethlon Hemopurifier®
is a clinical-stage immunotherapeutic device designed to combat cancer and life-threatening viral infections. In cancer, the Hemopurifier
is designed to deplete the presence of circulating tumor-derived exosomes that promote immune suppression, seed the spread of
metastasis and inhibit the benefit of leading cancer therapies. The U.S. Food and Drug Administration, or FDA, has designated
the Hemopurifier as a “Breakthrough Device” for two independent indications:
|
|
·
|
the treatment of individuals with advanced or metastatic cancer who are either unresponsive to or intolerant of standard of care therapy, and with cancer types in which exosomes have been shown to participate in the development or severity of the disease; and
|
|
|
·
|
the treatment of life-threatening viruses that are not addressed with approved therapies.
|
We believe the Hemopurifier can be a substantial
advance in the treatment of patients with advanced and metastatic cancer through the clearance of exosomes that promote the growth
and spread of tumors through multiple mechanisms. We are currently preparing for the initiation of clinical trials in patients
with advanced and metastatic cancers. We are initially focused on the treatment of solid tumors, including head and neck cancer,
gastrointestinal cancers and other cancers. As we advance our clinical trials, we are in close contact with our clinical sites
to navigate and assess the impact of COVID-19 on our clinical trials and current timelines.
On October 4, 2019, the FDA approved our Investigational Device
Exemption, or IDE, application to initiate an Early Feasibility Study, or EFS, of the Hemopurifier in patients with head and neck
cancer in combination with standard of care pembrolizumab (Keytruda). The primary endpoint for the EFS, which will enroll
10-12 subjects at a single center, will be safety, with secondary endpoints including measures of exosome clearance and characterization,
as well as response and survival rates. This study, which will be conducted at the UPMC Hillman Cancer Center in Pittsburgh, PA,
has been approved by the Institutional Review Board, or IRB, and is in the process of starting up.
We also believe the Hemopurifier can be
a part of the broad-spectrum treatment of life-threatening highly glycosylated, or carbohydrate coated, viruses that are not addressed
with an already approved treatment. In small-scale or early feasibility human studies, the Hemopurifier has been used to treat
individuals infected with HIV, hepatitis-C, and Ebola.
Additionally, in vitro, the Hemopurifier has been demonstrated to
capture Zika virus, Lassa virus, MERS-CoV, cytomegalovirus, Epstein-Barr virus, Herpes simplex virus, Chikungunya virus, Dengue
virus, West Nile virus, smallpox-related viruses, H1N1 swine flu virus, H5N1 bird flu virus, and the reconstructed Spanish flu
virus of 1918. In several cases, these studies were conducted in collaboration with leading government or non-government research
institutes.
We are also the majority owner of Exosome
Sciences, Inc., or ESI, a company focused on the discovery of exosomal biomarkers to diagnose and monitor life-threatening diseases.
Included among ESI’s activities is the advancement of a TauSome™ biomarker candidate to diagnose chronic traumatic
encephalopathy, or CTE, in the living. ESI previously documented TauSome levels in former NFL players to be nine times higher than
same age-group control subjects. Through ESI, we are also developing exosome based biomarkers in patients with, or at risk for,
a number of cancers. We consolidate ESI’s activities in our consolidated financial statements.
Successful outcomes of human trials will
also be required by the regulatory agencies of certain foreign countries where we plan to sell the Hemopurifier. Some of our patents
may expire before FDA approval or approval in a foreign country, if any, is obtained. However, we believe that certain patent applications
and/or other patents issued more recently will help protect the proprietary nature of the Hemopurifier treatment technology.
In addition to the foregoing, we are monitoring closely the
impact of the COVID-19 global pandemic on our business and have taken steps designed to protect the health and safety of our employees
while continuing our operations. Given the level of uncertainty regarding the duration and impact of the COVID-19 pandemic on capital
markets and the U.S. economy, we are unable to assess the impact of the worldwide spread of SARS-CoV-2 and the resulting COVID-19
pandemic on our timelines and future access to capital. We are continuing to monitor the spread of COVID-19 and its potential impact
on our operations. The full extent to which the COVID-19 pandemic will impact our business, results of operations, financial condition,
clinical trials, and preclinical research will depend on future developments that are highly uncertain, including actions taken
to contain or treat COVID-19 and their effectiveness, as well as the economic impact on national and international markets.
Our executive offices are located at 9635
Granite Ridge Drive, Suite 100, San Diego, California 92123. Our telephone number is (858) 459-7800. Our website address is www.aethlonmedical.com.
Our common stock is listed on the Nasdaq Capital Market under
the symbol “AEMD.”
REVERSE STOCK SPLIT
Effective October 14, 2019, the Company
completed a 1-for-15 reverse stock split. Accordingly, 15 shares of outstanding common stock then held by stockholders were combined
into one share of common stock. Any fractional shares resulting from the reverse split were rounded up to the next whole share.
Authorized common stock remained at 30,000,000 shares (see Note 14). The accompanying consolidated financial statements and accompanying
notes have been retroactively revised to reflect such reverse stock split as if it had occurred on April 1, 2018. All shares and
per share amounts have been revised accordingly.
LIQUIDITY AND GOING CONCERN
Management expects existing cash as of
March 31, 2020 to be sufficient to fund the Company’s operations for at least twelve months from the issuance date of these
consolidated financial statements.
PRINCIPLES OF CONSOLIDATION
The accompanying consolidated financial
statements include the accounts of Aethlon Medical, Inc. and its majority-owned (80% ownership) and controlled subsidiary, Exosome
Sciences, Inc., or ESI. All significant intercompany balances and transactions have been eliminated in consolidation. The Company
has classified the (20% ownership) noncontrolling interests in ESI as part of consolidated net loss in the fiscal years ended
March 31, 2020 and 2019 and includes the accumulated amount of noncontrolling interests as part of equity.
The losses at ESI during the fiscal year
ended March 31, 2020 reduced the noncontrolling interests on our consolidated balance sheet by $6,093 from $(126,031) at March
31, 2019 to $(132,124) at March 31, 2020.
RISKS AND UNCERTAINTIES
We operate in an industry that is subject
to intense competition, government regulation and rapid technological change. Our operations are subject to significant risk and
uncertainties including financial, operational, technological, regulatory, and including the potential risk of business failure.
USE OF ESTIMATES
We prepare our consolidated financial statements
in conformity with accounting principles generally accepted in the United States of America, or GAAP, which requires us to make
a number of estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets
and liabilities at the date of the financial statements. Such estimates and assumptions affect the reported amounts of expenses
during the reporting period. On an ongoing basis, we evaluate estimates and assumptions based upon historical experience and various
other factors and circumstances. We believe our estimates and assumptions are reasonable in the circumstances; however, actual
results may differ from these estimates under different future conditions. We believe that the estimates and assumptions that are
most important to the portrayal of our financial condition and results of operations, in that they require the most difficult,
subjective or complex judgments, form the basis for the accounting policies deemed to be most critical to us. These critical accounting
estimates relate to revenue recognition, stock purchase warrants issued with notes payable, beneficial conversion feature of convertible
notes payable, impairment of intangible assets and long lived assets, stock compensation, deferred tax asset valuation allowance,
and contingencies.
CASH AND CASH EQUIVALENTS
Accounting standards define “cash
and cash equivalents” as any short-term, highly liquid investment that is both readily convertible to known amounts of cash
and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates. For the
purpose of financial statement presentation, we consider all highly liquid investment instruments with original maturities of three
months or less when purchased, or any investment redeemable without penalty or loss of interest to be cash equivalents. As of March
31, 2020 and 2019, we had no assets that were classified as cash equivalents.
CONCENTRATIONS OF CREDIT RISKS
Cash is maintained at one financial institution
in checking accounts. Accounts at this institution are secured by the Federal Deposit Insurance Corporation up to $250,000. Our
March 31, 2020 cash balances were approximately $9,415,000 over such insured amount. We do
not believe that the Company is exposed to any significant risk with respect to its cash.
All of our accounts receivable at March 31, 2020 and all of
our revenue in the fiscal years ended March 31, 2020 and 2019 were directly from the National Cancer Institute.
PROPERTY AND EQUIPMENT
Property and equipment are stated at cost.
Depreciation is computed using the straight-line method over the estimated useful lives of the related assets, which range from
two to five years. Repairs and maintenance are charged to expense as incurred while improvements are capitalized. Upon the sale
or retirement of property and equipment, the accounts are relieved of the cost and the related accumulated depreciation with any
gain or loss included in the consolidated statements of operations.
INCOME TAXES
Deferred tax assets and liabilities are
recognized for the future tax consequences attributable to the difference between the consolidated financial statements and their
respective tax basis. Deferred income taxes reflect the net tax effects of (a) temporary differences between the carrying amounts
of assets and liabilities for financial reporting purposes and the amounts reported for income tax purposes, and (b) tax credit
carryforwards. We record a valuation allowance for deferred tax assets when, based on our best estimate of taxable income (if any)
in the foreseeable future, it is more likely than not that some portion of the deferred tax assets may not be realized.
LONG-LIVED ASSETS
Long-lived assets are reviewed for impairment
whenever events or changes in circumstances indicate that their carrying amounts may not be recoverable. If the cost basis of a
long-lived asset is greater than the projected future undiscounted net cash flows from such asset, an impairment loss is recognized.
We believe no impairment charges were necessary during the fiscal years ended March 31, 2020 and 2019.
LOSS PER SHARE
Basic loss per share is computed by dividing
net income available to common stockholders by the weighted average number of common shares outstanding during the period of computation.
Diluted loss per share is computed similar to basic loss per share except that the denominator is increased to include the number
of additional common shares that would have been outstanding if potential common shares had been issued, if such additional common
shares were dilutive. Since we had net losses for all periods presented, basic and diluted loss per share are the same, and additional
potential common shares have been excluded as their effect would be antidilutive.
As of March 31, 2020 and 2019, a total
of 2,072,492 and 437,784 potential common shares, consisting of shares underlying outstanding stock options, restricted stock units,
warrants and convertible notes payable were excluded as their inclusion would be antidilutive.
SEGMENTS
Historically, we operated in one segment
that was based on our development of therapeutic devices. However, in the December 2013 quarter, we initiated the operations of
ESI to develop diagnostic tests. As a result, we now operate in two segments, Aethlon for therapeutic applications and ESI for
diagnostic applications (See Note 10).
We record discrete financial information
for ESI and our chief operating decision maker reviews ESI’s operating results in order to make decisions about resources
to be allocated to the ESI segment and to assess its performance.
DEFERRED FINANCING COSTS
Costs related to the issuance of debt are
capitalized as a deduction to our convertible notes based on the new accounting standard on imputation of interest, and amortized
to interest expense over the life of the related debt using the effective interest method. There was no amortization related to
our deferred financing costs in the fiscal years ended March 31, 2020 and 2019.
REVENUE RECOGNITION
Our revenues consist entirely of amounts
earned under contracts and grants with the National Institutes of Health, or NIH. During the fiscal years ended March 31, 2020
and 2019, we recognized revenues totaling $650,187 and $229,625, respectively, under such contracts. We have concluded that these
agreements are not within the scope of ASC Topic, 606, Revenue from Contracts with Customers, or Topic 606, as the NIH grants
and contracts do not meet the definition of a “customer” as defined by Topic 606. Prior to the effective date of ASC
Topic 606, which for the Company was April 1, 2018, we accounted for our grant/contract revenues under the Milestone Method as
prescribed by the legacy guidance of ASC 605-28, Revenue Recognition – Milestone Method, or Milestone Method. In the absence
of other applicable guidance under US GAAP, effective April 1, 2018, we elected to continue to use the Milestone Method by analogy
to recognize revenue under these grants/contracts.
We identify the deliverables included within
these agreements and evaluate which deliverables represent separate units of accounting based on if certain criteria are met, including
whether the delivered element has standalone value to the collaborator. The consideration received is allocated among the separate
units of accounting, and the applicable revenue recognition criteria are applied to each of the separate units.
A milestone is an event having all of the
following characteristics:
(1) There is substantive uncertainty at
the date the arrangement is entered into that the event will be achieved. A vendor’s assessment that it expects to achieve
a milestone does not necessarily mean that there is not substantive uncertainty associated with achieving the milestone.
(2) The event can only be achieved based
in whole or in part on either: (a) the vendor’s performance; or (b) a specific outcome resulting from the vendor’s
performance.
(3) If achieved, the event would result
in additional payments being due to the vendor.
A milestone does not include events for
which the occurrence is either: (a) contingent solely upon the passage of time; or (b) the result of a counterparty’s performance.
The policy for recognizing deliverable
consideration contingent upon achievement of a milestone must be applied consistently to similar deliverables.
The assessment of whether a milestone is
substantive is performed at the inception of the arrangement. The consideration earned from the achievement of a milestone must
meet all of the following for the milestone to be considered substantive:
(1) The consideration is commensurate with
either: (a) the vendor’s performance to achieve the milestone; or (b) the enhancement of the value of the delivered item
or items as a result of a specific outcome resulting from the vendor’s performance to achieve the milestone;
(2) The consideration relates solely to
past performance; and
(3) The consideration is reasonable relative
to all of the deliverables and payment terms (including other potential milestone consideration) within the arrangement.
A milestone is not considered substantive
if any portion of the associated milestone consideration relates to the remaining deliverables in the unit of accounting (i.e.,
it does not relate solely to past performance). To recognize the milestone consideration in its entirety as revenue in the period
in which the milestone is achieved, the milestone must be substantive in its entirety. Milestone consideration cannot be bifurcated
into substantive and nonsubstantive components. In addition, if a portion of the consideration earned from achieving a milestone
may be refunded or adjusted based on future performance, the related milestone is not considered substantive.
We have entered into the following three
contracts/grants with the NCI, part of the NIH over the past two years:
Phase 2 Melanoma Cancer Contract
On September 12, 2019, the NCI awarded
to us an SBIR Phase II Award Contract, for NIH/NCI Topic 359, entitled “A Device Prototype for Isolation of Melanoma Exosomes
for Diagnostics and Treatment Monitoring”, or the Award Contract. The Award Contract amount is $1,860,561 and runs for the
period from September 16, 2019 through September 15, 2021.
The work to be performed pursuant to this
Award Contract will focus on melanoma exosomes. This work follows from our completion of a Phase I contract for the Topic 359 solicitation
that ran from September 2017 through June 2018 (see Phase 1 Melanoma Cancer Contract below). Following on the Phase I work, the
deliverables in the Phase II program involve the design and testing of a pre-commercial prototype of a more advanced version of
the exosome isolation platform.
During the fiscal year ended March 31,
2020, we recognized $620,187 in government contract revenue under this contract as a result of the work involved completing the
first three milestones in the project as reported in the kick-off presentation to the NCI and the first and second quarterly reports.
The kick-off presentation covered the Company's organization and project status, recent achievements, the status of the field,
the status of commercial and academic competitors, where the proposed project was positioned against the state of the art, the
IP landscape, a refresher on the proposed technology, the detailed plan for the first budget period of the contract and technical
risks and alternative approaches. The first and second quarterly reports covered a summary of technical objectives, a description
of activities accomplished in the quarter, an analysis of experimental data, comments regarding the timeliness of performance,
and a brief explanation of activities to be pursued in the following quarter.
Phase 1 Melanoma Cancer Contract
We entered into a contract with the NCI
in September 2017. This award was under the NIH’s SBIR program. The title of the award was “SBIR Topic 359 Phase 1
Device Strategy for Selective Isolation of Oncosomes and Non-Malignant Exosomes.” The award from NIH was a firm, fixed-price
contract with potential total payments to us of $299,250 over the course of nine months.
Fixed price contracts require the achievement
of multiple, incremental milestones to receive the full award during each period of the contract. The NIH also had the unilateral
right to require us to perform additional work under an option period for an additional fixed amount of $49,800. Under the terms
of the contract, we were required to perform certain incremental work toward the achievement of specific milestones against which
we invoiced the government for fixed payment amounts. The Phase 1 Melanoma Cancer Contract was completed in June 2018.
In the fiscal year ended March 31, 2019,
we performed work under the contract covering the remainder of the technical objectives of the contract (Aim 1: To validate the
Hemopurifier as a device for capture and recovery of melanoma exosomes from plasma, and Aim 2: To validate a method of melanoma
exosome isolation consisting of the Hemopurifier followed by mab-based immunocapture to select out the tumor-derived exosomes from
non-malignant exosomes, and Aim 3: To evaluate the functional integrity of melanoma exosomes purified by the Hemopurifier and immunocapture
isolation steps). As a result we invoiced NIH for $149,625 during the fiscal year ended March 31, 2019. The Melanoma Cancer Contract
is now completed.
Breast Cancer Grant
In September 2018, the NCI awarded us
a government grant (number 1R43CA232977-01). The title of this Small Business Innovation Research, or SBIR Phase I grant is “The
Hemopurifier Device for Targeted Removal of Breast Cancer Exosomes from the Blood Circulation.”
This NCI Phase I grant period originally
ran from September 14, 2018 through August 31, 2019. In August 2019, we applied for and received a no cost, twelve month extension
on this grant, so the expiration date was extended to August 31, 2020. The total amount of the firm grant is $298,444. The grant
calls for two subcontractors to work with us. Those subcontractors are University of Pittsburgh and Massachusetts General Hospital.
During the fiscal year ended March 31,
2020, we recognized $30,000 in government contract revenue under this grant as a result of the work involved in one of the three
technical objectives of the contract (Aim 2. “Elution of a population of breast cancer exosomes from Hemopurifier cartridges
that bear the signatures of malignancy based on expression of CSPG4 and HER2, for triple-negative or HER2-overexpressing cancers,
respectively”). We also invoiced the NCI for an additional $100,000 during the fiscal year ended March 31, 2020 in order
to pay our subcontractors under the contract. As we did not complete any additional technical objectives beyond Aim 2 noted above
during the period, we recorded this $100,000 as deferred revenue as of March 31, 2020.
During the fiscal year ended March 31,
2019, we recognized $80,000 in government contract revenue under this grant as a result of the work involved in completing one
of the three technical objectives of the contract (Aim 1. “To evaluate Hemopurifier-mediated capture of breast cancer exosomes”).
STOCK-BASED COMPENSATION
Employee stock options and rights to purchase
shares under stock participation plans are accounted for under the fair value method. Accordingly, share-based compensation is
measured when all granting activities have been completed, generally the grant date, based on the fair value of the award. The
exercise price of options is generally equal to the market price of the Company’s common stock (defined as the closing price
as quoted on the Nasdaq Capital Market or OTCBB on the date of grant). Compensation cost recognized by the Company includes (a)
compensation cost for all equity incentive awards granted prior to April 1, 2006, but not yet vested, based on the grant-date fair
value estimated in accordance with the original provisions of the then current accounting standards, and (b) compensation cost
for all equity incentive awards granted subsequent to March 31, 2006, based on the grant-date fair value estimated in accordance
with the provisions of subsequent accounting standards. We use a Binomial Lattice option pricing model for estimating fair value
of options granted (see Note 5).
The following table summarizes share-based
compensation expenses relating to shares and options granted and the effect on loss per common share during the years ended March
31, 2020 and 2019:
Our total stock-based compensation for fiscal years ended March
31, 2020 and 2019 included the following:
|
|
|
Fiscal Years Ended
|
|
|
|
|
March 31, 2020
|
|
|
March 31, 2019
|
|
|
Vesting of Stock Options and Restricted Stock Units
|
|
$
|
843,998
|
|
|
$
|
1,319,001
|
|
|
Total Stock-Based Compensation Expense
|
|
$
|
843,998
|
|
|
$
|
1,319,001
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average number of common shares outstanding – basic and diluted
|
|
|
3,414,840
|
|
|
|
1,208,314
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic and diluted loss per common share
|
|
$
|
(0.25
|
)
|
|
$
|
(1.09
|
)
|
We record share-based compensation expenses
for awards of stock options and restricted stock units, or RSUs, under ASC 718, Share-based compensation, or ASC 718. For awards
to non-employees for periods prior to the adoption of ASU 2018-07, Compensation-Stock Compensation: Improvements to Non-employee
Share-Based Payment Accounting, on April 1, 2019, the Company had applied ASC 505-50, Equity – Equity-based payments to
non-employees, or ASC 505-50. ASC 718 establishes guidance for the recognition of expenses arising from the issuance of share-based
compensation awards at their fair value at the grant date.
We recognize share-based compensation expense
related to stock options and SARs granted to employees, directors and consultants based on the estimated fair value of the awards
on the date of grant. We estimate the grant date fair value, and the resulting share-based compensation expense, for stock options
that only have service vesting requirements or performance-based vesting requirements without market conditions using the binomial
lattice option-pricing model. The grant date fair value of the share-based awards with service vesting requirements is generally
recognized on a straight-line basis over the requisite service period, which is generally the vesting period of the respective
awards. Determining the appropriate amount to expense for performance-based awards based on the achievement of stated goals requires
judgment. The estimate of expense is revised periodically based on the probability of achieving the required performance targets
and adjustments are made as appropriate. The cumulative impact of any revisions is reflected in the period of change. If any applicable
financial performance goals are not met, no compensation cost is recognized and any previously recognized compensation cost is
reversed. For performance-based awards with market conditions, we determine the fair value of awards as of the grant date using
a Monte Carlo simulation model.
We review share-based compensation on a
quarterly basis for changes to the estimate of expected award forfeitures based on actual forfeiture experience. The effect of
adjusting the forfeiture rate for all expense amortization after March 31, 2007 is recognized in the period the forfeiture estimate
is changed. The effect of forfeiture adjustments for the fiscal year ended March 31, 2020 was insignificant.
PATENTS
Patents include both foreign and domestic
patents. We capitalize the cost of patents, some of which were acquired, and amortize such costs over the shorter of the remaining
legal life or their estimated economic life, upon issuance of the patent. The unamortized costs of patents are subject to our review
for impairment under our long-lived asset policy above.
STOCK PURCHASE WARRANTS
We grant warrants in connection with the
issuance of common stock for cash. Warrants issued in connection with common stock for cash, if classified as equity, are considered
issued in connection with equity transactions and the warrant fair value is recorded to additional paid-in-capital.
BENEFICIAL CONVERSION FEATURE OF CONVERTIBLE
NOTES PAYABLE
The convertible feature of certain notes
payable provides for a rate of conversion that is below market value. Such feature is normally characterized as a Beneficial Conversion
Feature, or BCF. We measure the estimated fair value of the BCF in circumstances in which the conversion feature is not required
to be separated from the host instrument and accounted for separately, and record that value in the consolidated financial statements
as a discount from the face amount of the notes. Such discounts are amortized to interest expense over the term of the notes.
RESEARCH AND DEVELOPMENT EXPENSES
Our research and development costs are
expensed as incurred. We incurred approximately $927,000 and $896,000 of research and development expenses for the years ended
March 31, 2020 and 2019, respectively, which are included in various operating expenses in the accompanying consolidated statements
of operations.
OFF-BALANCE SHEET ARRANGEMENTS
We have not entered into any off-balance
sheet arrangements that have or are reasonably likely to have a current or future material effect on our consolidated financial
statements.
SIGNIFICANT RECENT ACCOUNTING PRONOUNCEMENTS
In June 2018, the FASB issued ASU No.
2018-07, Compensation-Stock Compensation (Topic 718), Improvements to Nonemployee Share-Based Payment Accounting, or ASU No. 2018-07.
ASU No. 2018-07 expands the scope of Topic 718 to include share-based payment transactions for acquiring goods and services from
nonemployees. ASU No. 2018-07 is effective for interim and annual reporting periods beginning after December 15, 2018 and early
adoption is permitted. Entities must apply the guidance retrospectively with a cumulative effect adjustment to retained earnings
as of the beginning of the period of adoption. The adoption of ASU No. 2018-07 on April 1, 2019 did not have a material impact
on the Company's consolidated financial position, results of operations and related disclosures.
During the fiscal year ended March 31,
2020, the Company adopted ASU Topic 842 on April 1, 2019 utilizing the alternative transition method allowed for under this guidance.
As a result, the Company recorded lease liabilities and right-of-use lease assets of $228,694 on its balance sheet as of April
1, 2019. The lease liabilities represent the present value of the remaining lease payments of the Company’s corporate headquarters
lease (see Note 13), discounted using the Company’s incremental borrowing rate as of April 1, 2019. The corresponding right-of-use
lease assets are recorded based on the lease liabilities and the cumulative difference between rent expense and amounts paid under
its corporate headquarters lease. The Company also elected the short-term lease recognition exemption for its laboratory lease.
For the laboratory lease that qualified as short-term, the Company did not recognize ROU assets or lease liabilities at adoption.
Topic 842 also allows lessees and lessors
to elect certain practical expedients. The Company elected the following practical expedients:
|
|
·
|
Transitional practical expedients, which must be elected as a package and applied consistently to all of the Company’s leases:
|
|
|
°
|
The Company need not reassess whether any expired or existing contracts are or contain leases.
|
|
|
|
|
|
|
°
|
The Company need not reassess the lease classification for any expired or existing leases (that is, all existing leases that were classified as operating leases in accordance with the previous guidance will be classified as operating leases, and all existing leases that were classified as capital leases in accordance with the previous guidance will be classified as finance leases).
|
|
|
|
|
|
|
°
|
The Company need not reassess initial direct costs for any existing leases.
|
|
|
·
|
Hindsight practical expedient. The Company elected the hindsight practical expedient in determining the lease term (that is, when considering lessee options to extend or terminate the lease and to purchase the underlying asset) and in assessing impairment of the Company’s right-of-use assets.
|
2. PROPERTY AND EQUIPMENT, NET
Property and equipment, net, consist of the following:
|
|
|
March 31, 2020
|
|
|
March 31, 2019
|
|
|
Furniture and office equipment, at cost
|
|
$
|
526,029
|
|
|
$
|
374,364
|
|
|
Accumulated depreciation
|
|
|
(385,545
|
)
|
|
|
(368,343
|
)
|
|
|
|
$
|
140,484
|
|
|
$
|
6,021
|
|
Depreciation expense for the fiscal years ended March 31, 2020
and 2019 was $17,202 and $21,531, respectively.
3. PATENTS, NET
Patents, net consist of the following:
|
|
|
March 31, 2020
|
|
|
March 31, 2019
|
|
|
Patents
|
|
$
|
211,645
|
|
|
$
|
211,645
|
|
|
Accumulated amortization
|
|
|
(154,141
|
)
|
|
|
(144,977
|
)
|
|
|
|
$
|
57,504
|
|
|
$
|
66,668
|
|
Amortization expense for capitalized patents
for each of the fiscal years ended March 31, 2020 and 2019 was $9,164. As several patents expired during the fiscal year ended
March 31, 2020 and only one capitalized patent remains to be amortized, future amortization expense on patents is estimated to
be approximately $550 per year based on the estimated life of the patent. The weighted average remaining life of our remaining
capitalized patent is approximately 6 years.
4. CONVERTIBLE NOTES PAYABLE
We paid off our convertible notes in full
in July 2019. The following table shows the activity related to our convertible notes during the fiscal year ended March 31, 2020:
|
Total Convertible Notes Payable at March 31, 2019
|
|
$
|
992,591
|
|
|
Less Principal Payments in Cash on Convertible Notes During the Fiscal Year Ended March 31, 2020
|
|
|
(992,591
|
)
|
|
Total Convertible Notes Payable at March 31, 2020
|
|
$
|
–
|
|
During the fiscal year ended March 31,
2020, we recorded interest expense of $23,759 related to the contractual interest rates of our convertible notes and interest expense
of $30,287 related to the amortization of the note discount, for a total interest expense of $54,046 related to our convertible
notes in the fiscal year ended March 31, 2020.
Convertible Notes Payable, Net consisted of the following at
March 31, 2019:
|
|
|
Principal
|
|
|
Unamortized
Discount
|
|
|
Net
Amount
|
|
|
Accrued
Interest
|
|
|
Convertible Notes Payable, Net:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
November 2014 10% Convertible Notes (due July 1, 2019)
|
|
$
|
612,811
|
|
|
$
|
(18,701
|
)
|
|
$
|
594,110
|
|
|
$
|
37,309
|
|
|
December 2016 10% Convertible Notes (due July 1, 2019)
|
|
|
379,780
|
|
|
|
(11,589
|
)
|
|
|
368,191
|
|
|
|
22,264
|
|
|
Total Convertible Notes Payable, Net
|
|
$
|
992,591
|
|
|
$
|
(30,290
|
)
|
|
$
|
962,301
|
|
|
$
|
59,573
|
|
During the fiscal year ended March 31,
2019, we recorded interest expense of $99,260 related to the contractual interest rates of our convertible notes and interest expense
of $121,148 related to the amortization of the note discount for a total interest expense of $220,408 related to our convertible
notes in the fiscal year ended March 31, 2019.
5. EQUITY TRANSACTIONS
ISSUANCES OF COMMON STOCK AND WARRANTS
Equity Transactions in the Fiscal Year Ended March 31, 2020.
December 2019 Public Offering
On December 13, 2019, we entered into an
underwriting agreement with H.C. Wainwright and Co., or Wainwright, as representative of the several underwriters named therein,
relating to the public offering, issuance and sale of 3,333,334 shares of common stock (which includes pre-funded warrants to purchase
shares of common stock in lieu thereof), and common warrants to purchase up to an aggregate of 3,333,334 shares of common stock,
at a public offering price of $1.50 per share and common warrant. Each share of common stock (or pre-funded warrant in lieu thereof)
was sold together with a common warrant to purchase one share of common stock. The common warrants have an exercise price of $1.50
per share, were immediately exercisable, and will expire five years from the date of issuance. The offering closed on December
17, 2019.
The gross proceeds of the December 2019
Public Offering were approximately $5 million, prior to deducting underwriting discounts and commissions and estimated offering
expenses and excluding the exercise of any common warrants and the underwriter's option to purchase additional securities. The
net proceeds from the December 2019 Public Offering were $4,091,437.
Subsequent to the completion of the December
2019 Public Offering and prior to March 31, 2020, all of the holders of pre-funded warrants exercised their pre-funded warrants
in full.
In the event of a Fundamental Transaction
(a transfer of ownership of the Company as defined in the common warrants issued in the December 2019 Public Offering) within our
control, the holders of the unexercised common stock warrants exercisable for $1.50 per share, are entitled to receive cash consideration
equal to a Black-Scholes valuation, as defined in the warrant. If such Fundamental Transaction is not within our control,
the warrant holders would only be entitled to receive the same form of consideration (and in the same proportion) as the holders
of our common stock, hence these warrants are classified as a component of permanent equity.
January 2020 Registered Direct Offering
and Private Placement
On January 16, 2020, we engaged Wainwright
to act as our exclusive placement agent in connection with the private placement and a concurrent registered direct offering,
or together, the Offering, of an aggregate of 1,885,378 shares of our common stock at a purchase price per share of $2.00, or
the Shares, for aggregate gross proceeds to us of approximately $3.77 million, before deducting fees payable to Wainwright and
other estimated offering expenses payable by us. We also entered into a securities purchase agreement, or the Purchase Agreement
with certain institutional investors, or the Purchasers, pursuant to which we agreed to sell and issue to the Purchasers warrants,
or the Purchase Warrants, to purchase up to an aggregate of 942,689 shares of our common stock, or the Purchase Warrant Shares.
We agreed to pay Wainwright a cash fee of 6.0% of the aggregate gross proceeds in the Offering, excluding the proceeds, if any,
from the exercise of the Purchase Warrants. We paid Wainwright an additional 1.0% of the aggregate gross proceeds in the Offering
as a management fee and also paid Wainwright an additional $70,000 for certain expenses in connection with the Offering. In addition,
Wainwright received placement agent warrants on substantially the same terms as the Purchase Warrants in an amount equal to 3.0%
of the aggregate number of Shares sold in the offering, or 56,561 shares of Common Stock, at an exercise price of $2.50 per share
and a term expiring on January 17, 2025, or the Placement Agent Warrants, and the shares of common stock issuable thereunder,
or the Placement Agent Warrant Shares.
On January 22, 2020, the Company closed
the Offering and issued the Purchase Warrants to the Purchasers. The Purchase Warrants are exercisable immediately at an exercise
price of $2.75 per share and will expire five and one-half years from the issuance date.
The net proceeds from the January 2020
Registered Direct Offering and Private Placement were $3,364,878.
Common Stock Sales Agreement with H.C.
Wainwright
On June 28, 2016, we entered into a Common
Stock Sales Agreement, or the Agreement, with H.C. Wainwright & Co., LLC, or H.C. Wainwright, which established an at-the-market
equity program pursuant to which we may offer and sell shares of our common stock from time to time as set forth in the Agreement.
The Agreement provides for the sale of shares, or the Shares, of our common stock having an aggregate offering price of up to $12,500,000.
On March 30, 2020, we executed Amendment No. 2 to the Agreement
with Wainwright, effective as of the same date. The amendment provides that references in the Agreement to the registration statement
shall refer to the registration statement on Form S-3 (File No. 333-237269), originally filed with the SEC on March 19, 2020, declared
effective by the SEC on March 30, 2020.
Subject to the terms and conditions set
forth in the Agreement, H.C. Wainwright agreed to use its commercially reasonable efforts consistent with its normal trading and
sales practices to sell the Shares from time to time, based upon our instructions. We have provided H.C. Wainwright with customary
indemnification rights, and H.C. Wainwright is entitled to a commission at a fixed rate equal to three percent (3.0%) of the gross
proceeds per Share sold. In addition, we agreed to pay certain expenses incurred by H.C. Wainwright in connection with the Agreement,
including up to $50,000 of the fees and disbursements of their counsel. The Agreement will terminate upon the sale of all of the
Shares under the Agreement, unless terminated earlier by either party as permitted under the Agreement.
Sales of the Shares, if any, under the
Agreement will be made in transactions that are deemed to be “at the market offerings” as defined in Rule 415 under
the Securities Act, including sales made by means of ordinary brokers’ transactions, including on the Nasdaq Capital Market,
at market prices or as otherwise agreed with H.C. Wainwright. We have no obligation to sell any of the Shares, and, at any time,
we may suspend offers under the Agreement or terminate the Agreement.
In the fiscal year ended March 31, 2020,
we raised aggregate net proceeds of $896,031 (net of $27,896 in commissions to H.C. Wainwright and $5,929 in other offering expenses)
under this Agreement through the sale of 161,149 shares at an average price of $5.56 per share of net proceeds.
Warrant Exercises
In fiscal year ended March 31, 2020, investors
that participated in the December 2019 public offering exercised 2,700,000 warrants for aggregate cash proceeds to us of $3,807,162.
Restricted Stock Unit Grants to Non-Employee
Directors
In 2012, as amended on August 9, 2016,
our Board of Directors established the Non-Employee Directors Compensation Program, to provide for cash and equity compensation
for persons serving as non-employee directors of the Company. Under this program, each new director receives either stock options
or a grant of restricted stock unites, or RSUs, as well as an annual grant of RSUs at the beginning of each fiscal year. The RSUs
are subject to vesting and represent the right to be issued on a future date shares of our common stock for upon vesting.
In April 2019, pursuant to the Non-Employee
Directors Compensation Program, we issued RSUs with a value of $35,000, in accordance with the terms of the plan, to each of our
non-employee directors, as the stock-based compensation element of their overall directors’ compensation, for the fiscal
year ending March 31, 2020. Those grants were based on the closing price of our common stock on the grant date, or $14.25 per share,
resulting in 2,456 RSUs being issued to each of our five non-employee directors, for a total of 12,280 RSUs. All of the RSUs were
subject to vesting in equal quarterly installments on June 30, 2019, September 30, 2019, December 31, 2019 and March 31, 2020.
During the fiscal year ended March 31,
2020, 12,280 vested RSUs held by our outside directors were exchanged into the same number of shares of our common stock. As four
of our five independent directors elected to return 40% of their RSUs in exchange for cash in order to pay their withholding taxes
on the share issuances, 3,926 of the RSUs were cancelled and we paid $11,230 in cash to those independent directors.
In addition, during the fiscal year ended
March 31, 2020, 8,793 vested RSUs then held by our executive officers were exchanged into the same number of shares of our common
stock. As our executives elected to net settle a portion of their RSU’s in exchange for the Company paying the related withholding
taxes on the share issuance, 4,657 of the RSUs were cancelled and we issued a net 4,136 shares to our executives.
There were no vested RSUs outstanding as
of March 31, 2020.
Common Stock for Warrant Cancellation
During the fiscal year ended March 31,
2020, we agreed with seven accredited investors to issue an aggregate of 3,992 shares of our common stock to these investors in
exchange for the cancellation of outstanding warrants then held by the investors to purchase an aggregate of 39,900 shares of our
common stock. We measured the fair value of the shares issued and the fair value of the warrants exchanged for those shares and
recorded a gain of $51,190 on those exchanges based on the changes in fair value between the instruments exchanged.
Equity Transactions in the Fiscal Year
Ended March 31, 2019.
Common Stock Sales Agreement with H.C.
Wainwright
On June 28, 2016, we entered into a Common
Stock Sales Agreement, or the Agreement, with H.C. Wainwright & Co., LLC, or Wainwright, which establishes an at-the-market
equity program pursuant to which we may offer and sell shares of our common stock from time to time as set forth in the Agreement.
The Agreement provides for the sale of shares of our common stock having an aggregate offering price of up to $12,500,000, or
the Shares.
Subject to the terms and conditions set
forth in the Agreement, Wainwright will use its commercially reasonable efforts consistent with its normal trading and sales practices
to sell the Shares from time to time, based upon our instructions. We have provided Wainwright with customary indemnification rights,
and Wainwright will be entitled to a commission at a fixed rate equal to three percent (3.0%) of the gross proceeds per Share sold.
In addition, we have agreed to pay certain expenses incurred by Wainwright in connection with the Agreement, including up to $50,000
of the fees and disbursements of their counsel. The Agreement will terminate upon the sale of all of the Shares under the Agreement
unless terminated earlier by either party as permitted under the Agreement.
Sales of the Shares, if any, under the
Agreement shall be made in transactions that are deemed to be “at the market offerings” as defined in Rule 415 under
the Securities Act, including sales made by means of ordinary brokers’ transactions, including on the Nasdaq Capital Market,
at market prices or as otherwise agreed with Wainwright. We have no obligation to sell any of the Shares, and, at any time, we
may suspend offers under the Agreement or terminate the Agreement.
In the fiscal year ended March 31, 2019,
we raised aggregate net proceeds of $1,048,371 (net of $32,471 in commissions to Wainwright and $5,447 in other offering expenses)
under the Agreement through the sale of 51,548 shares at an average price of $20.34 per share of net proceeds.
Warrant Exercises
In the fiscal year ended March 31, 2019,
investors that participated in our October 2017 public offering exercised 18,887 warrants for aggregate cash proceeds to us of
$292,932.
Restricted Shares Issued for Services
During the fiscal year ended March 31,
2019, we issued 1,000 shares of restricted common stock at a price of $19.35 per share, the market price at time of issuance, in
payment for investor relations consulting services. The aggregate value of this share issuance was $19,350.
Stock Option Issuances
During the fiscal year ended March 31,
2019, we issued an option to our new CEO under the Company’s 2010 Stock Incentive Plan to purchase 36,842 shares of our common
stock at an exercise price of $18.75 per share, the closing price on the date of the option grant.
Restricted Stock Unit Grants to Non-Employee
Directors
During the fiscal year ended March
31, 2019, 13,831 vested RSUs held by our executives were exchanged into the same number of shares of our common stock. As our executives
elected to net settle a portion of their RSU’s in exchange for the Company paying the related withholding taxes on the share
issuance, 7,484 of the RSUs were cancelled and we issued a net 6,347 shares to our executives.
During the fiscal year ended March 31, 2019, 9,699 RSUs held
by our outside directors were exchanged into the same number of shares of our common stock. As four of our five independent directors
elected to return 40% of their RSUs in exchange for cash in order to pay their withholding taxes on the share issuances, 3,165
of the RSUs were cancelled and we paid $54,278 in cash to those independent directors.
WARRANTS:
During the fiscal year ended March 31,
2020, we issued 4,432,585 warrants in association with our December 2019 Public Offering and our January 2020 Registered Direct
Financing and associated private placement (see Note 6). All of those warrants had a five year term and had exercise prices as
follows:
|
Financing
|
|
Warrants Issued
|
|
Exercise Price
|
|
December 2019 Public Offering – Investors’ Warrants
|
|
3,333,334
|
|
$1.50
|
|
December 2019 Public Offering – Placement Agents’ Warrants
|
|
100,000
|
|
$1.875
|
|
January 2020 Registered Direct – Investors’ Warrants
|
|
942,689
|
|
$2.75
|
|
January 2020 Registered Direct – Placement Agents’ Warrants
|
|
56,562
|
|
$2.50
|
Based on the above assumptions, we valued the warrants issued
during the fiscal year ended March 31, 2020 as follows:
|
|
·
|
The 999,251 warrants
issued in our January 2020 Registered Direct Financing were valued at $2,388,776 and we classified that fair value as equity.
|
|
|
|
|
|
|
·
|
The 3,433,334 warrants issued in our December 2019 public offering were valued at $3,021,334 and we classified that fair value
as equity.
|
We did not issue any warrants in the fiscal year ended March
31, 2019.
A summary of the aggregate warrant activity for the years ended
March 31, 2020 and 2019 is presented below:
|
|
|
Fiscal Year Ended March 31,
|
|
|
|
|
2020
|
|
|
2019
|
|
|
|
|
Warrants
|
|
|
Weighted
Average
Exercise Price
|
|
|
Warrants
|
|
|
Weighted
Average
Exercise Price
|
|
|
Outstanding, beginning of year
|
|
|
342,992
|
|
|
$
|
27.00
|
|
|
|
394,839
|
|
|
$
|
27.00
|
|
|
Granted
|
|
|
4,432,585
|
|
|
|
1.79
|
|
|
|
–
|
|
|
$
|
N/A
|
|
|
Adjustment for reverse split
|
|
|
73
|
|
|
|
N/A
|
|
|
|
--
|
|
|
|
N/A
|
|
|
Exercised
|
|
|
(2,700,000
|
)
|
|
$
|
1.50
|
|
|
|
(18,887
|
)
|
|
$
|
16.50
|
|
|
Cancelled/Forfeited
|
|
|
(54,282
|
)
|
|
$
|
91.23
|
|
|
|
(32,960
|
)
|
|
$
|
42.60
|
|
|
Outstanding, end of year
|
|
|
2,021,368
|
|
|
$
|
5.21
|
|
|
|
342,992
|
|
|
$
|
27.00
|
|
|
Exercisable, end of year
|
|
|
2,021,368
|
|
|
$
|
5.21
|
|
|
|
342,992
|
|
|
$
|
27.00
|
|
|
Weighted average estimated fair value of warrants granted
|
|
|
|
|
|
$
|
1.22
|
|
|
|
|
|
|
|
N/A
|
|
The following outlines the significant weighted average assumptions
used to estimate the fair value of warrants granted in the fiscal year ended March 31, 2020 utilizing the Binomial Lattice option
pricing model:
|
|
|
|
Fiscal Year Ended
|
|
|
|
|
|
March 31, 2020
|
|
|
Risk free interest rate
|
|
|
1.57% - 1.71%
|
|
|
Average expected life
|
|
|
5 years
|
|
|
Expected volatility
|
|
|
148.6% - 233.0%
|
|
|
Expected dividends
|
|
|
None
|
|
The expected volatility was based on the historic volatility.
The expected life of options granted was based on the “simplified method” as described in the SEC’s guidance
due to changes in the vesting terms and contractual life of current option grants compared to our historical grants.
The detail of the warrants outstanding and exercisable as of
March 31, 2020 is as follows:
|
|
|
|
Warrants Outstanding
|
|
|
|
|
|
|
|
Warrants Exercisable
|
|
Range of
Exercise Prices
|
|
|
Number
Outstanding
|
|
|
|
Weighted
Average
Remaining
Life (Years)
|
|
|
|
Weighted
Average
Exercise Price
|
|
|
|
Number
Outstanding
|
|
|
|
Weighted
Average
Exercise Price
|
|
|
$2.75 or Below
|
|
|
1,732,585
|
|
|
|
4.80
|
|
|
$
|
2.23
|
|
|
|
1,732,585
|
|
|
$
|
4.80
|
|
|
$16.50 - $59.25
|
|
|
249,985
|
|
|
|
2.07
|
|
|
$
|
23.24
|
|
|
|
249,985
|
|
|
$
|
2.07
|
|
|
$64.50 - $135.00
|
|
|
38,798
|
|
|
|
0.58
|
|
|
$
|
92.48
|
|
|
|
38,798
|
|
|
$
|
0.58
|
|
|
|
|
|
2,021,368
|
|
|
|
|
|
|
|
|
|
|
|
2,021,368
|
|
|
|
|
|
STOCK-BASED COMPENSATION:
2010 STOCK INCENTIVE PLAN
In August 2010, we adopted the 2010 Stock
Incentive Plan, to provide incentives to attract, retain and motivate employees, directors and consultants, whose present and potential
contributions are important to our success, by offering them an opportunity to participate in our future performance through awards
of options, the right to purchase common stock, stock bonuses and stock appreciation rights and other awards. We initially authorized
a total of 4,667 common shares for issuance under the 2010 Stock Incentive Plan.
On January 26, 2016, our Board of Directors
approved an amendment to the 2010 Stock Incentive Plan to increase the total number of shares of common stock authorized for issuance
under the plan to 211,333 shares, subject to amendment of our Articles of Incorporation to increase our authorized common stock.
On March 29, 2016, at which our stockholders approved the Amended 2010 Stock Incentive Plan and an amendment of our Articles of
Incorporation to increase our authorized common stock to 30,000,000 shares. On March 31, 2016, we filed a Certificate of Amendment
to our Articles of Incorporation to effect the increase in our authorized common stock. As a result of such amendment, the Amended
2010 Stock Incentive Plan became effective on March 31, 2016.
Effective October 14, 2019, we completed
a 1-for-15 reverse stock split. Accordingly, 15 shares of outstanding common stock then held by stockholders were combined into
one share of common stock. Any fractional shares resulting from the reverse split were rounded up to the next whole share. Authorized
common stock remained at 30,000,000 shares.
At March 31, 2020, we had 119,343 shares
available for issuance under this plan See Note 11 to cross reference April 2020 RSU issuance to independent directors.
2012 NON-EMPLOYEE DIRECTORS COMPENSATION PROGRAM
In July 2012, our Board of Directors approved
a board compensation program that modified and superseded the Company’s 2005 Directors Compensation Program, or the Non-Employee
Director Plan, which was previously in effect for our non-employee Directors. Under the Non-Employee Director Plan, an eligible
director will receive initial and annual equity grants and cash compensation.
In June 2014 and August 2016, our Board
of Directors approved further amendments to the Non-Employee Director Plan. Under this modified program, a new eligible director
will receive an initial grant of $50,000 worth of RSUs or, at the discretion of our Board of Directors, options to acquire shares
of common stock. RSUs granted under this provision will be valued based on the average of the closing prices of the common stock
for the five trading days preceding and including the date of grant and will vest at a rate determined by our Board of Directors
in its discretion, typically over one year, partially on the date of grant and in equal quarterly installments thereafter. Options
granted under this plan will have an exercise price equal to the fair market value on the date of grant. Such options will have
a term of ten years and will vest at a rate determined by our Board of Directors in its discretion.
In addition, under the Non-Employee Director
Plan, at the beginning of each fiscal year, each existing director eligible to participate will receive a grant of $35,000 worth
of RSUs or, at the discretion of our Board of Directors, options to acquire shares of common stock. RSUs granted under this provision
will be valued based on the average of the closing prices of the common stock for the five trading days preceding and including
the first day of the fiscal year (or preceding and including the date of grant, if such grant is not made on the first day of the
fiscal year) and will vest at a rate determined by our Board of Directors in its discretion, typically in equal quarterly installments
over one year. Options granted under this plan will have an exercise price equal to the Fair Market value on the date of grant.
Such options will have a term of ten years and will vest at a rate determined by our Board of Directors in its discretion.
In lieu of per meeting fees, eligible directors
receive an annual board retainer fee of $30,000. The Non-Employee Director Plan also provides for the following annual retainer
fees: Audit Committee Chair - $5,000, Compensation Committee chair - $5,000, Nominating Committee chair - $5,000, Audit Committee
member - $4,000, Compensation Committee member - $4,000, Nominating Committee member - $4,000 and lead independent director - $15,000.
The RSU grants and the changes to the Non-Employee
Director Plan were approved and recommended by our Compensation Committee prior to approval by our Board of Directors.
Dr. Fisher additionally is compensated
$90,000 per year for his services as Chairman of our Board, which our Board of Directors considers to be fees payable as a member
of our Board of Directors or a Committee of our Board for purposes of Section 10A-3 of the rules promulgated under the Securities
Exchange Act of 1934, as amended. To the extent payment of such fees are construed to not be fees payable as a member of our Board
of Directors or a Committee of our Board, then our Board of Directors considers that Dr. Fisher may act as a member of its Audit
Committee under Nasdaq Rule 5605(c)(2)(B) as our Board of Directors has determined that it is in the best interests of our Company
and its stockholders for Dr. Fisher to continue to serve on its Audit Committee. The Board has awarded compensation to non-employee
directors in the past outside of the Non-Employee Director Plan.
In April 2019, pursuant to the Non-Employee
Directors Compensation Program, we issued RSUs with a value of $35,000, in accordance with the terms of the plan, to each of our
non-employee directors, as the stock-based compensation element of their overall directors’ compensation, for the fiscal
year ending March 31, 2020. Those grants were based on the closing price of our common stock on the grant date, or $14.25 per share,
resulting in 2,456 RSUs being issued to each of our five non-employee directors, for a total of 12,280 RSUs. All of the RSUs were
subject to vesting in equal quarterly installments on June 30, 2019, September 30, 2019, December 31, 2019 and March 31, 2020.
During the fiscal year ended March 31,
2020, 12,280 vested RSUs held by our outside directors were exchanged into the same number of shares of our common stock. As four
of our five independent directors elected to return 40% of their RSUs in exchange for cash in order to pay their withholding taxes
on the share issuances, 3,926 of the RSUs were cancelled and we paid $11,230 in cash to those independent directors.
In addition, during the fiscal year ended
March 31, 2020, 8,793 vested RSUs then held by our executive officers were exchanged into the same number of shares of our common
stock. As our executives elected to net settle a portion of their RSU’s in exchange for the Company paying the related withholding
taxes on the share issuance, 4,657 of the RSUs were cancelled and we issued a net 4,136 shares to our executives.
There were no vested RSUs outstanding as of March 31, 2020.
STAND-ALONE GRANTS
From time to time our Board of Directors
grants common stock or options to purchase common stock or warrants exercisable to common stock to selected directors, officers,
employees and consultants as equity compensation to such persons on a stand-alone basis outside of any of our formal stock plans.
The terms of these grants are individually negotiated.
STOCK OPTION ACTIVITY
During the fiscal year ended March 31,
2019, we issued an option to our new CEO to purchase 36,842 shares of common stock at a price of $18.75 per share, the closing
price on the date of the option grant.
The following is a summary of the stock
options outstanding at March 31, 2020 and 2019 and the changes during the years then ended:
|
|
|
Fiscal Year Ended March 31,
|
|
|
|
|
2020
|
|
|
2019
|
|
|
|
|
Options
|
|
|
Weighted
Average
Exercise Price
|
|
|
Options
|
|
|
Weighted
Average
Exercise Price
|
|
|
Outstanding, beginning of year
|
|
|
59,111
|
|
|
$
|
56.85
|
|
|
|
27,670
|
|
|
$
|
145.05
|
|
|
Granted
|
|
|
--
|
|
|
$
|
N/A
|
|
|
|
36,842
|
|
|
$
|
18.75
|
|
|
Exercised
|
|
|
–
|
|
|
|
N/A
|
|
|
|
–
|
|
|
|
N/A
|
|
|
Cancelled/Forfeited
|
|
|
(7,987
|
)
|
|
$
|
138.75
|
|
|
|
(5,400
|
)
|
|
$
|
248.25
|
|
|
Outstanding, end of year
|
|
|
51,124
|
|
|
$
|
44.12
|
|
|
|
59,111
|
|
|
$
|
56.85
|
|
|
Exercisable, end of year
|
|
|
25,197
|
|
|
$
|
70.08
|
|
|
|
21,070
|
|
|
$
|
125.40
|
|
|
Weighted average estimated fair value of options granted
|
|
|
|
|
|
$
|
N/A
|
|
|
|
|
|
|
$
|
16.50
|
|
The detail of the options outstanding and exercisable as of
March 31, 2020 is as follows:
|
|
|
|
|
|
Options Outstanding
|
|
|
|
Options Exercisable
|
|
|
|
Exercise Prices
|
|
|
|
Number
Outstanding
|
|
|
|
Weighted
Average
Remaining
Life (Years)
|
|
|
|
Weighted
Average
Exercise
Price
|
|
|
|
Number
Outstanding
|
|
|
|
Weighted
Average
Exercise
Price
|
|
|
|
$18.75 - $25.20
|
|
|
|
38,642
|
|
|
|
7.89 years
|
|
|
$
|
19.05
|
|
|
|
12,715
|
|
|
$
|
19.36
|
|
|
|
$57.00 - $75.00
|
|
|
|
6,113
|
|
|
|
3.17 years
|
|
|
$
|
64.28
|
|
|
|
6,113
|
|
|
$
|
64.28
|
|
|
|
$142.50 - $187.50
|
|
|
|
6,369
|
|
|
|
1.38 years
|
|
|
$
|
176.90
|
|
|
|
6,369
|
|
|
$
|
176.90
|
|
|
|
|
|
|
|
51,124
|
|
|
|
|
|
|
|
|
|
|
|
25,197
|
|
|
|
|
|
We recorded stock-based compensation expense
related to restricted stock unit issuances and to options granted totaling $843,998 and $1,319,001 for the fiscal years ended March
31, 2020 and 2019, respectively. These expenses were recorded as stock compensation included in payroll and related expenses in
the accompanying consolidated statement of operations for the years ended March 31, 2019 and 2018.
Our total stock-based compensation for
fiscal years ended March 31, 2020 and 2019 included the following:
|
|
|
Fiscal Year Ended
|
|
|
|
|
March 31, 2020
|
|
|
March 31, 2019
|
|
|
Vesting of restricted stock units
|
|
$
|
678,028
|
|
|
$
|
1,262,794
|
|
|
Vesting of stock options
|
|
|
165,970
|
|
|
|
56,207
|
|
|
Total Stock-Based Compensation
|
|
$
|
843,998
|
|
|
$
|
1,319,001
|
|
We review share-based compensation on a
quarterly basis for changes to the estimate of expected award forfeitures based on actual forfeiture experience. The cumulative
effect of adjusting the forfeiture rate for all expense amortization is recognized in the period the forfeiture estimate is changed.
The effect of forfeiture adjustments for the fiscal year ended March 31, 2020 was insignificant.
As of March 31, 2020, we had $1,528,684
of remaining unrecognized stock-based compensation expense, which is expected to be recognized over a weighted average remaining
vesting period of 4.33 years.
On March 31, 2020, our stock options had
a negative intrinsic value since the closing price on that date of $1.52 per share was below the weighted average exercise price
of our stock options.
6. RELATED PARTY TRANSACTIONS
DUE TO RELATED PARTIES
Historically, certain of our officers and
other related parties have advanced us funds, agreed to defer compensation and/or paid expenses on our behalf to cover working
capital deficiencies. There were no such related party transactions during the fiscal year ended March 31, 2020, except that we
had accrued unpaid Board fees of $69,750 owed to our outside directors as of March 31, 2020.
Due to related parties were comprised of the following items:
|
|
|
March 31, 2020
|
|
|
March 31, 2019
|
|
|
Accrued board fees
|
|
$
|
69,750
|
|
|
$
|
69,750
|
|
|
Accrued vacation
|
|
|
41,957
|
|
|
|
13,904
|
|
|
Total due to related parties
|
|
$
|
111,707
|
|
|
$
|
83,654
|
|
7. OTHER CURRENT LIABILITIES
Other current liabilities were comprised of the following items:
|
|
|
March 31, 2020
|
|
|
March 31, 2019
|
|
|
Accrued interest
|
|
$
|
–
|
|
|
$
|
59,573
|
|
|
Accrued separation expenses for former executives (see Note 6 and Note 12)
|
|
|
–
|
|
|
|
355,189
|
|
|
Accrued professional fees
|
|
|
472,420
|
|
|
|
231,238
|
|
|
Total other current liabilities
|
|
$
|
472,420
|
|
|
$
|
646,000
|
|
8. INCOME TAXES
For the years ended March 31, 2020 and
2019, we had no income tax expense due to our net operating losses and 100% deferred tax asset valuation allowance.
At March 31, 2020 and 2019, we had net
deferred tax assets as detailed below. These deferred tax assets are primarily composed of capitalized research and development
costs and tax net operating loss carryforwards. Due to uncertainties surrounding our ability to generate future taxable income
to realize these assets, a 100% valuation has been established to offset the net deferred tax assets.
Significant components of our net deferred
tax assets at March 31, 2020 and 2019 are shown below:
|
|
|
YEAR ENDED MARCH 31,
|
|
|
|
|
2020
|
|
|
2019
|
|
|
Deferred tax assets:
|
|
|
|
|
|
|
|
|
|
Capitalized research and development
|
|
$
|
3,442,000
|
|
|
$
|
3,442,000
|
|
|
Net operating loss carryforwards
|
|
|
18,384,000
|
|
|
|
17,221,000
|
|
|
Stock compensation
|
|
|
1,181,000
|
|
|
|
944,000
|
|
|
Total deferred tax assets
|
|
|
23,007,000
|
|
|
|
21,607,000
|
|
|
|
|
|
|
|
|
|
|
|
|
Total deferred tax liabilities
|
|
|
–
|
|
|
|
–
|
|
|
|
|
|
|
|
|
|
|
|
|
Net deferred tax assets
|
|
|
23,007,000
|
|
|
|
21,607,000
|
|
|
Valuation allowance for deferred tax assets
|
|
|
(23,007,000
|
)
|
|
|
(21,607,000
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net deferred tax assets
|
|
$
|
–
|
|
|
$
|
–
|
|
At March 31, 2020, we had tax net operating loss carryforwards
for federal and state purposes approximating $71 million and $53 million, which begin to expire in the year 2021.
The provision for income taxes on earnings subject to income
taxes differs from the statutory federal rate for the years ended March 31, 2020 and 2019 due to the following:
|
|
|
2020
|
|
|
2019
|
|
|
|
|
|
|
|
|
|
|
Income taxes (benefit) at federal statutory rate of 21.00%
|
|
$
|
(1,340,000
|
)
|
|
$
|
(1,306,000
|
)
|
|
State income tax, net of federal benefit
|
|
|
(446,000
|
)
|
|
|
(434,000
|
)
|
|
Tax effect on non-deductible expenses and credits
|
|
|
122,000
|
|
|
|
41,000
|
|
|
True up items
|
|
|
42,000
|
|
|
|
(41,000
|
)
|
|
Expiration of net operating loss carryforwards
|
|
|
222,000
|
|
|
|
407,000
|
|
|
Change in valuation allowance1
|
|
|
1,400,000
|
|
|
|
1,333,000
|
|
|
|
|
$
|
–
|
|
|
$
|
–
|
|
______________
(1) Pursuant to Internal Revenue Code Sections 382, use of our
tax net operating loss carryforwards may be limited.
ASC 740, “Income Taxes”, clarifies
the accounting for uncertainty in income taxes recognized in an entity's financial statements, and prescribes recognition thresholds
and measurement attributes for financial statement disclosure of tax positions taken or expected to be taken on a tax return. Under
ASC 740, the impact of an uncertain income tax position on the income tax return must be recognized at the largest amount that
is more-likely-than-not to be sustained upon audit by the relevant taxing authority. An uncertain income tax position will not
be recognized if it has less than a 50% likelihood of being sustained. Additionally, ASC 740 provides guidance on derecognition,
classification, interest and penalties, accounting in interim periods, disclosure and transition. Our practice is to recognize
interest and/or penalties related to income tax matters in income tax expense. During the years ended March 31, 2020 and 2019,
we did not recognize any interest or penalties relating to tax matters.
At and for the years ended March 31, 2020
and 2019, management does not believe the Company has any uncertain tax positions. Accordingly, there are no unrecognized tax benefits
at March 31, 2020 or March 31, 2019.
Our tax returns remain open for examination
by the applicable authorities, generally 3 years for federal and 4 years for state. We are currently not under examination by
any taxing authorities.
9. SEGMENTS
We operate our businesses principally through
two reportable segments: Aethlon, which represents our therapeutic business activities, and ESI, which represents our diagnostic
business activities. Our reportable segments have been determined based on the nature of the potential products being developed.
We record discrete financial information for ESI and our chief operating decision maker reviews ESI’s operating results in
order to make decisions about resources to be allocated to the ESI segment and to assess its performance.
Aethlon’s revenue is generated primarily
from government contracts to date and ESI does not yet have any revenues. We have not included any allocation of corporate overhead
to the ESI segment.
The following tables set forth certain information regarding
our segments:
|
|
|
Fiscal Years Ended March 31,
|
|
|
|
|
2020
|
|
|
2019
|
|
|
Revenues:
|
|
|
|
|
|
|
|
Aethlon
|
|
$
|
650,187
|
|
|
$
|
229,625
|
|
|
ESI
|
|
|
–
|
|
|
|
–
|
|
|
Total Revenues
|
|
$
|
650,187
|
|
|
$
|
229,625
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating Losses:
|
|
|
|
|
|
|
|
|
|
Aethlon
|
|
$
|
(5,899,523
|
)
|
|
$
|
(5,875,094
|
)
|
|
ESI
|
|
|
(30,465
|
)
|
|
|
(123,923
|
)
|
|
Total Operating Loss
|
|
$
|
(5,929,988
|
)
|
|
$
|
(5,999,017
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Net Losses:
|
|
|
|
|
|
|
|
|
|
Aethlon
|
|
$
|
(6,349,576
|
)
|
|
$
|
(6,095,581
|
)
|
|
ESI
|
|
|
(30,465
|
)
|
|
|
(123,923
|
)
|
|
Net Loss Before Non-Controlling Interests
|
|
$
|
(6,380,041
|
)
|
|
$
|
(6,219,504
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Cash:
|
|
|
|
|
|
|
|
|
|
Aethlon
|
|
$
|
9,604,583
|
|
|
$
|
3,827,555
|
|
|
ESI
|
|
|
197
|
|
|
|
519
|
|
|
Total Cash
|
|
$
|
9,604,780
|
|
|
$
|
3,828,074
|
|
|
|
|
|
|
|
|
|
|
|
|
Total Assets:
|
|
|
|
|
|
|
|
|
|
Aethlon
|
|
$
|
10,387,489
|
|
|
$
|
4,122,445
|
|
|
ESI
|
|
|
197
|
|
|
|
519
|
|
|
Total Assets
|
|
$
|
10,387,686
|
|
|
$
|
4,122,964
|
|
|
|
|
|
|
|
|
|
|
|
|
Capital Expenditures:
|
|
|
|
|
|
|
|
|
|
Aethlon
|
|
$
|
151,665
|
|
|
$
|
–
|
|
|
ESI
|
|
|
–
|
|
|
|
–
|
|
|
Capital Expenditures
|
|
$
|
–
|
|
|
$
|
–
|
|
|
|
|
|
|
|
|
|
|
|
|
Depreciation and Amortization:
|
|
|
|
|
|
|
|
|
|
Aethlon
|
|
$
|
26,366
|
|
|
$
|
30,695
|
|
|
ESI
|
|
|
–
|
|
|
|
–
|
|
|
Total Depreciation and Amortization
|
|
$
|
26,366
|
|
|
$
|
30,695
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest Expense:
|
|
|
|
|
|
|
|
|
|
Aethlon
|
|
$
|
54,232
|
|
|
$
|
220,487
|
|
|
ESI
|
|
|
–
|
|
|
|
–
|
|
|
Total Interest Expense
|
|
$
|
54,232
|
|
|
$
|
220,487
|
|
10. SUBSEQUENT EVENTS
Management has evaluated events subsequent to March 31, 2020
through the date that the accompanying consolidated financial statements were filed with the Securities and Exchange Commission
for transactions and other events which may require adjustment of and/or disclosure in such financial statements.
Management Update
In June 2020, Thomas L. Taccini joined
our management team as Vice President, Manufacturing and Product Development. Mr. Taccini has over 35 years of experience in leading
teams in engineering, product development, project management, quality systems and regulatory affairs for multiple different classes
of medical devices.
IDE Supplement
On June 17, 2020, the FDA approved a supplement
to the Company’s open IDE for the Company’s Hemopurifier in viral disease to allow for the testing of the Hemopurifier
in patients with SARS-CoV-2/COVID-19 in a new feasibility study. That study’s plan is to enroll up to 40 subjects at up to
20 centers in the U.S. Subjects will have established laboratory diagnosis of COVID-19, be admitted to an intensive care unit,
or ICU and will have acute lung injury and/or severe or life threatening disease among other criteria. Endpoints for this study,
in addition to safety, will include reduction in circulating virus as well as clinical outcomes.
Sales Under ATM Facility
In June 2020, we raised aggregate net
proceeds of $7,260,869, net of $224,825 in commissions to Wainwright and $8,472 in other offering expenses, under the Agreement
described above through the sale of 2,685,600 shares at an average price of $2.70 per share of net proceeds.
COVID-19 Update
In March 2020, the World Health Organization declared COVID-19
a pandemic. The COVID-19 pandemic has negatively impacted the global economy, disrupted global supply chains and created significant
volatility and disruption of financial markets.
We are monitoring closely the impact of the COVID-19 global
pandemic on our business and have taken steps designed to protect the health and safety of our employees while continuing our operations,
including clinical trials. Given the level of uncertainty regarding the duration and impact of the COVID-19 pandemic on capital
markets and the U.S. economy, we are unable to assess the impact of the worldwide spread of SARS-CoV-2 and the resulting COVID-19
pandemic on our future access to capital. Further, while we have not experienced significant disruptions to our manufacturing supply
chain, business, results of operations, financial condition, clinical trials, or preclinical research to date, we are unable to
assess the potential impact this pandemic could have on our manufacturing supply chain, business, results of operations, financial
condition, clinical trials, or preclinical research in the future.
As we continue to actively advance our clinical trials, we remain
in close contact with our clinical sites and are assessing the impact of COVID-19 on our trials, expected timelines and costs on
an ongoing basis. We will assess any potential delays in our ability to timely ship clinical trial materials, including internationally,
due to transportation interruptions. The extent of the impact of COVID-19 on our operational and financial performance will depend
on certain developments, including the duration and spread of the outbreak, impact on our clinical trials, employees and vendors,
all of which are uncertain and cannot be predicted. Given these uncertainties, we cannot reasonably estimate the related impact
to our business, operating results and financial condition, if any.
RSU Grants
On April 3, 2020, pursuant to the terms
of the Company’s 2012 Non-Employee Directors Compensation Program, as modified on August 9, 2016, or the Directors Plan,
the Compensation Committee of the Board granted RSUs under the Company’s 2010 Stock Incentive Plan, or the 2010 Plan, to
each non-employee director of the Company. The Director’s Plan provides for a grant of $35,000 worth of RSUs at the beginning
of each fiscal year, priced at the average for the closing prices for the five days preceding and including the date of grant,
or $1.41 per share as of April 3, 2020. Each eligible director was granted an RSU in the amount of 23,893 shares under the 2010
Plan, as the number of available shares under the 2010 Plan was not sufficient for each director’s full grant. The RSU’s
are subject to vesting in four equal quarterly installments on June 30, September 30, December 31, 2020, and March 31, 2021, subject
to the recipient's continued service with the Company on each such vesting date. Each eligible director will receive the remaining
portion of the annual RSU grants, or 929 RSU’s, contingent upon stockholder approval at the 2020 annual meeting of the Company’s
stockholders, of a new 2020 Equity Incentive Plan.
11. COMMITMENTS AND CONTINGENCIES
CONTRACTUAL OBLIGATIONS AND COMMITMENTS
We have had the following material changes to our contractual
obligations and commitments outside the ordinary course of business during the fiscal year ended March 31, 2020:
On March 17, 2020, we entered into an Amended
and Restated Executive Employment Agreement, or the Amended Agreement, for Timothy C. Rodell, M.D., FCCP, or Executive, dated March
17, 2020, by and between the Company and Executive. This Amended Agreement supersedes and replaces Executive’s current Executive
Employment Agreement with the Company, dated December 10, 2018, or Prior Agreement.
The Amended Agreement updated the Prior
Agreement to set forth (i) Executive’s base salary for calendar year 2020, previously approved by the Company, of $430,000
per year; (ii) Executive’s title of Chief Executive Officer; (iii) Executive’s eligibility to participate in and receive
additional stock option or equity award grants under the Company’s equity incentive plans from time to time, in the discretion
of the Board or the Compensation Committee, and in accordance with the terms and conditions of such plans; and (iv) severance payments
in the event that Executive’s employment is terminated by the Company for any reason other than Cause (as defined in the
Amended Agreement) or if it is terminated by Executive for Good Reason (as defined in the Amended Agreement).
LEASE COMMITMENTS
We currently lease approximately 2,600
square feet of executive office space at 9635 Granite Ridge Drive, Suite 100, San Diego, California 92123 under a 39-month
gross plus utilities lease that commenced on December 1, 2014 and expires on August 31, 2021 the “Granite Ridge Lease.”
The current rental rate under the lease extension is $8,265 per month. We believe this leased facility will be satisfactory for
our office needs over the term of the lease.
We also rent approximately 1,700 square
feet of laboratory space at 11585 Sorrento Valley Road, Suite 109, San Diego, California 92121 at the rate of $4,700 per month
on a one-year lease that originally was to expire on November 30, 2019. In October 2019, we entered into a lease extension for
an additional twelve months running from December 1, 2019 through November 30, 2020, at the rate of $5,961 per month.
Rent expense, which is included in general
and administrative expenses, approximated $178,000 and $169,000 for the fiscal years ended March 31, 2020 and 2019, respectively.
As of March 31, 2020, our commitments under the lease agreements
are as follows:
|
|
|
2021
|
|
|
2022
|
|
|
Totals
|
|
|
9635 Granite Ridge Drive, Suite 100, San Diego, CA 92123 office lease
|
|
$
|
102,074
|
|
|
$
|
43,670
|
|
|
$
|
145,744
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
11585 Sorrento Valley Road, Suite 109, San Diego, CA 92121 office lease
|
|
|
47,684
|
|
|
|
–
|
|
|
|
47,684
|
|
|
Total Lease Commitments
|
|
$
|
149,758
|
|
|
$
|
43,670
|
|
|
$
|
193,428
|
|
LEGAL MATTERS
From time to time, claims are made against
us in the ordinary course of business, which could result in litigation. Claims and associated litigation are subject to inherent
uncertainties and unfavorable outcomes could occur, such as monetary damages, fines, penalties or injunctions prohibiting us from
selling one or more products or engaging in other activities.
The occurrence of an unfavorable outcome
in any specific period could have a material adverse effect on our results of operations for that period or future periods. We
are not presently a party to any pending or threatened legal proceedings.
Aethlon Medical (NASDAQ:AEMD)
Historical Stock Chart
From Aug 2024 to Sep 2024
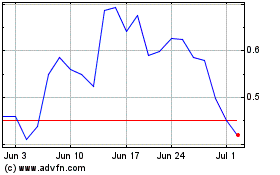
Aethlon Medical (NASDAQ:AEMD)
Historical Stock Chart
From Sep 2023 to Sep 2024