Filed Pursuant to Rule 424(b)(5)
Registration No. 333-254711
PROSPECTUS SUPPLEMENT
(To prospectus dated May 3, 2021)
Up to $20,000,000 of
Common Stock
ENDRA
Life Sciences Inc. has entered into an At-The-Market Issuance Sales
Agreement (the “Sales Agreement”) with Ascendiant
Capital Markets, LLC (the “Sales Agent”) relating to
the offer and sale of up to $20,000,000 of our common stock, par
value $0.0001 per share, offered by this prospectus supplement and
the accompanying prospectus. Effective upon entering into the Sales
Agreement, we and the Sales Agent terminated our previous
At-The-Market Issuance Sales Agreement, dated February 19,
2021.
Sales
of our common stock, if any, under this prospectus supplement and
the accompanying prospectus may be made in transactions that are
deemed to be “at-the-market” offerings as defined in
Rule 415 under the Securities Act of 1933, as amended (the
“Securities Act”), including sales made directly on or
through the Nasdaq Capital Market (“Nasdaq”), the
trading market for our common stock, or any other trading market in
the United States for our common stock, sales made to or through a
market maker other than on an exchange, directly to the Sales Agent
as principal for its own account in negotiated transactions at
market prices prevailing at the time of sale or at prices related
to such prevailing market prices, in privately negotiated
transactions, in block trades, or through a combination of any such
methods of sale. The Sales Agent will act as sales agent on a
commercially reasonable efforts basis consistent with its normal
trading and sales practices. There is no arrangement for funds to
be received in any escrow, trust or similar
arrangement.
We will pay the Sales Agent a commission equal to 3.0% of the gross
sales price per share of common stock issued by us and sold through
the Sales Agent as our sales agent under the Sales Agreement. In
connection with the sale of the common stock on our behalf, the
Sales Agent will be deemed to be an “underwriter”
within the meaning of the Securities Act and the compensation of
the Sales Agent will be deemed to be underwriting commissions or
discounts.
We are
an “emerging growth company” as that term is defined in
the Jumpstart Our Business Startups Act of 2012 and, as such, are
subject to certain reduced public company reporting requirements.
See “The Company—Implications of Being an Emerging
Growth Company” in the accompanying prospectus.
Our
common stock is listed on the Nasdaq Capital Market under the
symbol “NDRA.” The last reported sale price of our
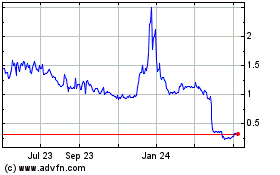
common stock on the Nasdaq Capital Market on June 18, 2021 was
$2.11 per share. Our common stock has
recently experienced extreme volatility in price and trading
volume. From January 1, 2021 to
June 21, 2021, the closing price of our common stock on the Nasdaq
Capital Market has ranged from $0.80 to $2.85 per share and daily
trading volume has ranged from approximately 300,000 to 33 million
shares. This volatility reflects a risk of investing in our common
stock; please see the section of this prospectus supplement titled
“Risk Factors.”
Investing in our common stock involves risks. See “Risk
Factors” beginning on page S-8.
Neither the Securities and Exchange Commission (the
“SEC”), nor any state securities commission, or any
other regulatory body has approved or disapproved of these
securities or determined if this prospectus supplement is truthful
or complete. Any representation to the contrary is a criminal
offense.
___________________
___________________
The
date of this prospectus supplement is June 21,
2021.
TABLE OF CONTENTS
|
Prospectus Supplement
|
|
|
|
Page
|
|
ABOUT THIS PROSPECTUS SUPPLEMENT
|
S-1
|
|
FORWARD-LOOKING
STATEMENTS AND OTHER INFORMATION CONTAINED IN THIS PROSPECTUS
SUPPLEMENT
|
S-2
|
|
SUMMARY
|
S-4
|
|
THE
OFFERING
|
S-7
|
|
RISK
FACTORS
|
S-8
|
|
USE OF
PROCEEDS
|
S-10
|
|
DIVIDEND
POLICY
|
S-11
|
|
DILUTION
|
S-11
|
|
PLAN OF
DISTRIBUTION
|
S-12
|
|
LEGAL
MATTERS
|
S-13
|
|
EXPERTS
|
S-13
|
|
WHERE YOU CAN FIND
MORE INFORMATION
|
S-13
|
|
INCORPORATION OF
CERTAIN INFORMATION BY REFERENCE
|
S-13
|
Base Prospectus
|
|
Page
|
|
|
1
|
|
|
1
|
|
|
3
|
|
|
4
|
|
|
4
|
|
|
4
|
|
|
5
|
|
|
5
|
|
|
6
|
|
|
14
|
|
|
15
|
|
|
16
|
|
|
17
|
|
|
17
|
|
|
17
|
|
|
17
|
ABOUT THIS PROSPECTUS SUPPLEMENT
This prospectus supplement and the accompanying prospectus form
part of a registration statement on Form S-3 (File No.
333-254711) that we filed with the SEC utilizing a
“shelf” registration process. This document contains
two parts. The first part consists of this prospectus supplement,
which provides you with specific information about this offering.
The second part, the accompanying prospectus, provides more general
information, some of which may not apply to this offering.
Generally, when we refer only to the “prospectus,” we
are referring to both parts combined together with all documents
incorporated by reference.
In this
prospectus supplement, the terms “ENDRA,”
“we,” “us,” “our” and the
“Company” refer to ENDRA Life Sciences Inc. unless
otherwise stated or the context otherwise requires.
This
prospectus supplement, and the information incorporated herein by
reference, may add, update or change information in the
accompanying prospectus and in any free writing prospectuses we may
provide to you in connection with this offering. You should read
both this prospectus supplement and the accompanying prospectus
together with additional information described under the headings
“Where You Can Find More Information” and
“Incorporation of Certain Information by Reference.” If
there is any inconsistency between the information in this
prospectus supplement and the accompanying prospectus, you should
rely on the information in this prospectus supplement.
You may rely only on the information contained in or incorporated
by reference in this prospectus supplement and the accompanying
prospectus. Neither we nor the Sales Agent has authorized anyone to
provide information different from that contained in this
prospectus supplement, the accompanying prospectus and the
documents incorporated by reference herein and therein. If anyone
provides you with different or inconsistent information, you should
not rely on it. The information in this prospectus supplement, the
accompanying prospectus and in any free writing prospectuses we may
provide to you in connection with this offering is accurate only as
of their respective dates, regardless of time of delivery. Our
business, financial condition, results of operations and prospects
may have changed since those dates.
We are
offering to sell, and seeking offers to buy, our securities only in
jurisdictions where offers and sales are permitted. The
distribution of this prospectus supplement and the offering of the
securities in certain jurisdictions may be restricted by law.
Persons outside the United States who come into possession of this
prospectus supplement must inform themselves about, and observe any
restrictions relating to, the offering of the securities and the
distribution of this prospectus supplement outside the United
States. This prospectus supplement does not constitute, and may not
be used in connection with, an offer to sell, or a solicitation of
an offer to buy, any securities offered by this prospectus
supplement by any person in any jurisdiction in which it is
unlawful for such person to make such an offer or
solicitation.
All
references in this prospectus to our consolidated financial
statements include, unless the context indicates otherwise, the
related notes.
We further note that the representations, warranties and covenants
made by us in any agreement that is filed as an exhibit to any
document that is incorporated by reference herein were made solely
for the benefit of the parties to such agreement, including, in
some cases, for the purpose of allocating risk among the parties to
such agreements, and should not be deemed to be a representation,
warranty or covenant to you. Moreover, such representations,
warranties or covenants were accurate only as of the date when
made. Accordingly, such representations, warranties and covenants
should not be relied on as accurately representing the current
state of our affairs.
This prospectus and the information incorporated by reference
herein include trademarks, service marks and trade names owned by
us or other companies. All trademarks, service marks and trade
names included or incorporated by reference into this prospectus
are the property of their respective owners.
The
industry and market data and other statistical information
contained in this prospectus supplement, the accompanying
prospectus and the documents we incorporate by reference are based
on management’s own estimates, independent publications,
government publications, reports by market research firms or other
published independent sources, and, in each case, are believed by
management to be reasonable estimates. Although we believe these
sources are reliable, we have not independently verified the
information. None of the independent industry publications used in
this prospectus supplement, the accompanying prospectus or the
documents we incorporate by reference were prepared on our or our
affiliates’ behalf and none of the sources cited by us
consented to the inclusion of any data from its reports, nor have
we sought their consent.
FORWARD-LOOKING STATEMENTS AND OTHER INFORMATION
CONTAINED IN THIS PROSPECTUS SUPPLEMENT
Certain information set forth in this prospectus supplement, set
forth in the accompanying prospectus or incorporated by reference
herein or therein may contain
“forward-looking statements” within the meaning of
Section 27A of the Securities Act and Section 21E of the Exchange
Act, that are intended to be covered by the “safe
harbor” created by those sections. Forward-looking
statements, which are based on certain assumptions and describe our
future plans, strategies and expectations, can generally be
identified by the use of forward-looking terms such as
“believe,” “expect,” “may,”
“will,” “should,” “would,”
“could,” “seek,” “intend,”
“plan,” “goal,” “project,”
“estimate,” “anticipate,”
“strategy”, “future”, “likely”
or other comparable terms and references to future periods. All
statements other than statements of historical facts included in
this prospectus supplement and the documents incorporated by
reference regarding our strategies, prospects, financial condition,
operations, costs, plans and objectives are forward-looking
statements. Examples of forward-looking statements include, among
others, statements we make regarding expectations for revenues,
cash flows and financial performance, the anticipated results of
our development efforts and the timing for receipt of required
regulatory approvals and product launches.
Forward-looking statements are neither historical facts nor
assurances of future performance. Instead, they are based only on
our current beliefs, expectations and assumptions regarding the
future of our business, future plans and strategies, projections,
anticipated events and trends, the economy and other future
conditions. Because forward-looking statements relate to the
future, they are subject to inherent uncertainties, risks and
changes in circumstances that are difficult to predict and many of
which are outside of our control. Our actual results and financial
condition may differ materially from those indicated in the
forward-looking statements. Therefore, you should not rely on any
of these forward-looking statements. Important factors that could
cause our actual results and financial condition to differ
materially from those indicated in the forward-looking statements
include, among others, the following:
●
our limited
commercial experience, limited cash and history of
losses;
●
our ability to
obtain adequate financing to fund our business operations in the
future;
●
our ability to
achieve profitability;
●
our ability to
develop a commercially feasible application based on our
Thermo-Acoustic Enhanced Ultrasound (“TAEUS”)
technology;
●
market acceptance
of our technology;
●
uncertainties
associated with COVID-19, or coronavirus, including its possible
effects on our operations;
●
results of our
human studies, which may be negative or inconclusive;
●
our ability to find
and maintain development partners;
●
our reliance on
third parties, collaborations, strategic alliances and licensing
arrangements to complete our business strategy;
●
the amount and
nature of competition in our industry;
●
our
ability to protect our intellectual property;
●
potential
changes in the healthcare industry or third-party reimbursement
practices;
●
our ability to
maintain CE mark certification and secure required U.S. Food and
Drug Administration (“FDA”) and other governmental
approvals for our TAEUS applications;
●
delays and changes
in regulatory requirements, policy and guidelines including
potential delays in obtaining required regulatory
approvals;
●
our
ability to regain compliance with Nasdaq listing
standards;
●
our
ability to comply with regulation by various federal, state, local
and foreign governmental agencies and to maintain necessary
regulatory clearances or approvals;
●
our
dependence on our senior management team;
●
the volatility of our stock price;
and
●
the other risks and
uncertainties described in “Risk Factors” beginning on
page S-8 of this prospectus supplement and in Part I, Item 1A, Risk
Factors of our Annual Report on Form 10-K for the fiscal year ended
December 31, 2020 and in Part II, Item 1A, Risk Factors of
subsequently filed Quarterly Reports on Form 10-Q, which are
incorporated by reference in this prospectus supplement and the
accompanying prospectus.
Any forward-looking statement made by us in this prospectus
supplement is based only on information currently available to us
and speaks only as of the date on which it is made. We undertake no
obligation to publicly update any forward-looking statement,
whether written or oral, that may be made from time to time,
whether as a result of new information, future developments or
otherwise. We anticipate that subsequent events and developments
will cause our views to change. You should read this prospectus
supplement, the accompanying prospectus and the documents
referenced herein or
therein and filed as exhibits
to the registration statement, of which this prospectus supplement
is a part, completely and with the understanding that our actual
future results may be materially different from what we expect. Our
forward-looking statements do not reflect the potential impact of
any future acquisitions, merger, dispositions, joint ventures or
investments we may undertake. We qualify all of our forward-looking
statements by these cautionary statements.
|
|
|
|
|
|
This
summary highlights selected information contained elsewhere in or
incorporated by reference in this prospectus supplement and the
accompanying prospectus. The summary may not contain all of the
information that is important to you or that you should consider
before making an investment decision. You should carefully read
this entire prospectus supplement and the accompanying prospectus,
as well as the information to which we refer you, before deciding
whether to invest in our common stock. You should pay special
attention to the “Risk Factors” section of this
prospectus supplement to determine whether an investment in our
common stock is appropriate for you.
About ENDRA Life Sciences Inc.
We are leveraging experience with pre-clinical enhanced ultrasound
devices to develop technology for increasing the capabilities of
clinical diagnostic ultrasound in order to broaden patient access
to the safe diagnosis and treatment of a number of significant
medical conditions in circumstances where expensive X-ray computed
tomography (“CT”) and magnetic resonance imaging
(“MRI”) technology, or other diagnostic technologies
such as surgical biopsy, are unavailable or
impractical.
In
2010, we began marketing and selling our Nexus 128 system, which
combined light-based thermoacoustics and ultrasound to address the
imaging needs of researchers studying disease models in
pre-clinical applications. Building on this expertise in
thermoacoustics, we have developed a next-generation technology
platform — Thermo Acoustic Enhanced Ultrasound, or TAEUS
— which is intended to enhance the capability of clinical
ultrasound technology and support the diagnosis and treatment of a
number of significant medical conditions that currently require the
use of expensive CT or MRI imaging or where imaging is not
practical using existing technology. We ceased production, service
support and party for our Nexus 128 system in July 2019 in order to
focus our resources exclusively on the development of our TAEUS
technology.
Unlike
the near-infrared light pulses used in our legacy Nexus 128 system,
our TAEUS technology uses radio frequency (“RF”) pulses
to stimulate tissues, using a small fraction (less than 1%) of the
energy that would be transmitted into the body during an MRI scan.
The use of RF energy allows our TAEUS technology to penetrate deep
into tissue, enabling the imaging of human anatomy at depths
equivalent to those of conventional ultrasound. The RF pulses are
absorbed by tissue and converted into ultrasound signals, which are
detected by an external ultrasound receiver and a digital
acquisition system that is part of the TAEUS system. The detected
ultrasound is processed into images and other forms of data using
our proprietary algorithms and displayed to complement conventional
gray-scale ultrasound images. The TAEUS imaging concept is
illustrated below:
|
|
|
|
|
|
|
|
|
|
|
|
We believe that our TAEUS technology has the potential to add a
number of new capabilities to conventional ultrasound and thereby
enhance the utility of both existing and new ultrasound systems and
extend the use of ultrasound technology to circumstances that
either currently require the use of expensive CT or MRI imaging
systems, where imaging is not practical using existing technology,
or where other assessment tools such as surgical biopsy are
required.
Our TAEUS platform is not intended to replace CT or MRI systems,
both of which are versatile imaging technologies with capabilities
and uses beyond the focus of our business. However, they are also
expensive, with a CT system costing approximately $1 million and an
MRI system costing up to $3 million. In addition, and in contrast
to ultrasound systems, due to their limited number and the fact
that they are usually fixed-in-place at major medical facilities,
CT or MRI systems are frequently inaccessible to patients.
Ultrasound systems are more broadly available to patients than
either CT or MRI systems. There are an estimated one million
ultrasound systems globally in use generating over 400 million
annual diagnostic ultrasound procedures globally. Sales of
ultrasound diagnostic equipment were approximately $4.4 billion
globally in 2017 and an estimated 30,000 to 50,000 new and
replacement ultrasound systems are sold into the market annually.
Ultrasound systems are relatively inexpensive compared to CT and
MRI systems, with smaller portable ultrasound systems costing as
little as $10,000 and new cart-based ultrasound systems costing
between $75,000 and $200,000. These numbers cover all types of
diagnostic ultrasound procedures, including systems intended for
cardiology, prenatal and abdominal use. However, we do not
currently intend to address ultrasound systems focused on
applications in prenatal care, where we believe our TAEUS
technology will not substantially impact patient care. Accordingly,
we define our addressable market for one or more of our TAEUS
applications at approximately 365,000 cart-based ultrasound systems
currently in use throughout the world.
CT and MRI systems are stationary systems, requiring the patient to
travel to a medical center. As a result, they are less accessible
to primary care and rural clinics, economically developing markets,
and patient bedsides. As of 2018, there were only approximately
63,000 CT systems and 50,000 MRI systems in the world,
approximately 50% of which were located in the U.S. and Japan. Many
ultrasound systems are designed to be moved by an operator from
room to room, or closer to patients. Ultrasound technology does not
present the same safety concerns as CT and MRI technology, since
ultrasound does not emit ionizing radiation and ultrasound contrast
agents are considered to be generally safe. The ultrasound’s
imaging capabilities, however, are more limited compared to CT and
MRI technologies, which are able to measure tissue temperature
during thermal ablation surgery or quantify fat to diagnose early
stage liver disease.
To
demonstrate the capabilities of our TAEUS platform, we have
conducted various internal ex-vivo laboratory experiments and
limited internal in-vivo large animal studies. In our ex-vivo and
in-vivo testing, we have demonstrated that the TAEUS platform has
the following capabilities and potential clinical
applications:
●
Tissue
Composition: Our TAEUS technology enables ultrasound to distinguish
fat from lean tissue. This capability would enable the use of
TAEUS-enhanced ultrasound for the early identification, staging and
monitoring of nonalcoholic fatty liver disease
(“NAFLD”), a precursor to nonalcoholic steatohepatitis
(“NASH”), liver fibrosis, cirrhosis and liver
cancer.
●
Temperature
Monitoring: Our TAEUS technology enables traditional ultrasound to
visualize changes in tissue temperature, in real time. This
capability would enable the use of TAEUS-enhanced ultrasound to
guide thermoablative therapy, which uses heat or cold to remove
tissue, such as in the treatment of cardiac atrial fibrillation, or
removal of cancerous liver and kidney lesions, with greater
accuracy.
●
Vascular
Imaging: Our TAEUS technology enables ultrasound to view blood
vessels from any angle, using only a saline solution contrasting
agent, unlike Doppler ultrasound, which requires precise viewing
angles. This capability would enable the use of TAEUS-enhanced
ultrasound to easily identify arterial plaque or malformed
vessels.
●
Tissue
Perfusion: Our TAEUS technology enables ultrasound to image blood
flow at the capillary level in a region, organ or tissue. This
capability could be used to assist physicians in characterizing
microvasculature fluid flows symptomatic of damaged tissue, such as
internal bleeding from trauma, or diseased tissue, such as certain
cancers.
The
first TAEUS application we intend to commercialize is our NAFLD
TAEUS application addressing liver tissue composition. Our initial
target market for this application is the European Union. For
commercial reasons and to support our application for CE marking,
which is required before we can sell the application in the
European Union, we engaged the Centre for Imaging Technology
Commercialization, a contract research organization, to initiate
human studies through Canada-based Robarts Research Institute to
demonstrate our NAFLD TAEUS application’s ability to
distinguish fat from lean tissue. In December 2018, Robarts
Research Institute completed its initial healthy subject enrollment
and data collection of 25 subjects and received authorization from
Health Canada to expand the study to 50 subjects. In September
2019, we announced the completion and reported top-level findings
of the expanded study, which was included in our TAEUS liver device
technical file submission for device CE mark. We received CE mark
approval for our TAEUS FLIP (Fatty Liver Imaging Probe) NAFLD
application in March 2020. In June 2020, we submitted to the FDA
our application under the Food, Drug and Cosmetic Act (the
“FD&C Act”) to sell our NAFLD TAEUS application in
the U.S. The application was submitted for approval under Section
510(k) of the FD&C Act and we anticipate that any other TAEUS
applications that we develop will similarly be submitted under
Section 510(k). We expect that our initial FDA clearance will allow
us to sell the NAFLD TAEUS application in the U.S. with general
imaging claims. However, we will need to obtain additional FDA
clearances to be able to make diagnostic claims for fatty tissue
content determination. Accordingly, to support our
commercialization efforts we expect that, following receipt of our
initial FDA clearance, we would submit one or more additional
applications to the FDA, each of which would need to include
additional clinical trial data, so that following receipt of the
necessary clearances we may make those diagnostic
claims.
After required regulatory approvals, our TAEUS technology can be
added as an accessory to existing ultrasound systems, helping to
improve clinical decision-making on the front lines of patient
care, without requiring substantially new clinical workflows or
large capital investments. We also intend to develop TAEUS for
incorporation into new ultrasound systems manufactured by companies
such as GE Healthcare.
Because of the large number of traditional ultrasound systems
currently in global use, we are first developing our TAEUS
technology for sale as an aftermarket accessory that works with
existing ultrasound systems. Because our TAEUS technology is
designed to enhance the utility of, not replace, conventional
ultrasound, we believe healthcare providers will be able to
increase the utilization of, and generate new revenue from, their
existing ultrasound systems once we obtain required regulatory
approval for specific applications. Based on our design work and
our understanding of the ultrasound accessory market, we intend to
price our initial NAFLD TAEUS application at a price point
approximating $35,000 to $50,000, which should enable purchasers to
recoup their investment by performing a relatively small number of
additional ultrasound procedures. We further believe that
clinicians will be attracted to our technology because it will
enable them to perform more procedures with existing ultrasound
equipment, thereby retaining more imaging patients in their clinics
rather than referring patients out to a regional medical center for
a CT or MRI scan.
Each of our TAEUS platform applications will require regulatory
approvals before we are able to sell or license the application.
Based on certain factors, such as the installed base of ultrasound
systems, availability of other imaging technologies, such as CT and
MRI, economic strength and applicable regulatory requirements, we
are seeking initial approval of our applications for sale in the
European Union, followed by the United States, and plan to seek
additional approval in China.
|
|
|
|
|
|
|
|
|
|
|
|
Recent Developments
Termination of February 2021 At-The-Market Offering
In February 2021, the Company entered into an At-The-Market
Issuance Sales Agreement (the “Prior ATM Agreement”)
with the Sales Agent to sell shares of the Company’s common
stock for aggregate gross proceeds of up to $12.6 million, from
time to time, through an at-the-market equity offering program. The
Company sold an aggregate of approximately 4.5 million
shares of common stock for gross
proceeds of approximately $10.6 million million under the Prior ATM Agreement. The Prior ATM
Agreement was terminated effective June 21, 2021.
General Company Information
We were incorporated in Delaware in July 2007. Our corporate
headquarters is located at 3600 Green Court, Suite 350, Ann Arbor,
Michigan 48105-1570. The telephone number of our principal
executive office is (734) 335-0468. Our website can be accessed at
www.endrainc.com. The information contained on, or that may
be obtained from, our website is not, and shall not be deemed to
be, a part of this prospectus supplement or the accompanying
prospectus.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Common
stock offered by us
|
|
Shares of our
common stock having an aggregate offering price of up to
$20,000,000.
|
|
|
|
|
|
|
|
|
|
Common stock to be outstanding after the
offering
|
|
Up to 9,478,673
shares, after giving effect to the sale of $20.0 million of shares
of our common stock at an assumed price of $2.11 per share, which
was the closing price of our common stock on the Nasdaq Capital
Market on June 18, 2021. The maximum number of shares that may be
issued will vary depending on the price at which shares may be sold
from time to time during this offering. (1)
|
|
|
|
|
|
|
|
|
|
Manner of
offering
|
|
“At the market
offering,” as defined in Rule 415(a)(4) of the Securities
Act, that may be made from time to time by the Sales Agent. We may
also sell the shares of our common stock to the Sales Agent as
principal for its own accounts, at a price per share agreed upon at
the time of sale. If we sell shares to a Sales Agent as principal,
we will enter into a separate terms agreement setting forth the
terms of such transaction, and we will describe the agreement in a
pricing supplement. See “Plan of Distribution” on page
S-12 of this prospectus supplement.
|
|
|
|
|
|
|
|
|
|
Use
of proceeds
|
|
We intend to use
the net proceeds from this offering for working capital and other
general corporate purposes. See “Use of Proceeds” for
additional information.
|
|
|
|
|
|
|
|
|
|
|
|
Investing in our
common stock involves significant risks. See the section entitled
“Risk Factors” beginning on page S-8 and the other
information included or incorporated by reference in this
prospectus supplement and the accompanying prospectus for a
discussion of factors you should carefully consider before deciding
to invest in shares of our common stock.
|
|
|
|
|
|
|
|
|
|
The
Nasdaq Capital Market symbol
|
|
Our common stock is
currently listed on the Nasdaq Capital Market under the symbol
“NDRA.”
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1)
The number of shares of our common stock to be
outstanding after this offering is based on 41,672,695
shares of common stock outstanding as
of June 14, 2021 and excludes the following:
●
1,932,000 shares of
common stock issuable upon the exercise of outstanding warrants
issued in our initial public offering listed on the Nasdaq Capital
Market under the symbol “NDRAW,” at an exercise price
of $6.25 per share;
●
668,284 shares of
common stock issuable upon the exercise of outstanding unregistered
warrants, at a weighted average exercise price of $2.83 per
share;
●
177,272 shares of
common stock issuable upon the conversion of outstanding shares of
Series A Convertible Preferred Stock;
●
4,540,417 shares of
common stock issuable upon the exercise of outstanding stock
options issued pursuant to our 2016 Omnibus Incentive Plan (the
“Incentive Plan”) at a weighted average exercise price
of $2.16 per share; and
●
2,910,811 shares of
common stock reserved for future issuance under our Incentive
Plan.
|
|
|
|
|
|
|
|
You
should carefully consider the following risk factors and the risk
factors discussed under “Item 1A - Risk Factors”
contained in our Annual Report on Form 10-K for the fiscal year
ended December 31, 2020 and in Part
II, Item 1A, Risk Factors of subsequently filed Quarterly Reports
on Form 10-Q, which are incorporated by
reference in this prospectus supplement in their entirety, together
with all of the other information contained in this prospectus
supplement and the accompanying prospectus or incorporated by
reference in this prospectus supplement and the accompanying
prospectus. The risks described in any document incorporated by
reference are not the only ones we have, but are considered to be
the most material. Additional risks of which we are not presently
aware or that we currently believe are immaterial may also harm our
business and results of operations. If any of these risks actually
occur, our business, financial condition and results of operations
would likely suffer. In that case, the market price of the common
stock could decline, and you may lose part or all of your
investment in our company.
Risks Related to this Offering of Our Common Stock
Sales of our common stock in this offering, or the perception that
such sales may occur, could cause the market price of our common
stock to fall.
We may
issue and sell shares of our common stock for aggregate gross
proceeds of up to $20.0 million from time to time in connection
with this offering. The issuance and sale from time to time of
these new shares of common stock, or our ability to issue these new
shares of common stock in this offering, could have the effect of
depressing the market price of our common stock.
You will suffer immediate and substantial dilution in the net
tangible book value per share of the common stock that you purchase
in this offering.
The shares sold in this
offering, if any, will be sold from time to time at various prices;
however, the assumed public offering price of our common
stock is substantially higher than the as-adjusted net tangible
book value per share of our common stock. Therefore,
investors purchasing shares of our common stock in this offering
will pay a price per share that substantially exceeds the
as-adjusted net tangible book value per share after this offering.
Assuming that an aggregate of 9,478,673 shares of our common stock
are sold at a public offering price of $2.11 per share, the last
reported sale price of our common stock on the Nasdaq Capital
Market on June 18, 2021, for aggregate gross proceeds of $20.0
million, and after deducting commissions and estimated offering
expenses payable by us, new investors in this offering will
experience immediate dilution of $1.38 per share, representing the
difference between the assumed public offering price and our as
adjusted net tangible book value per share after giving effect to
this offering. See “Dilution” for a more detailed
discussion of the dilution you would incur if you purchase common
stock in this offering.
You may experience future dilution as a result of future equity
offerings.
In
order to raise additional capital, we may in the future offer
additional shares of our common stock or other securities
convertible into or exchangeable for our common stock at prices
that may not be the same as the price per share in this offering.
The price per share at which we sell additional shares of our
common stock, or securities convertible or exchangeable into common
stock, in future transactions may be lower than the price per share
paid by investors in this offering.
Our management will have broad discretion in the use of the net
proceeds from this offering and may not use them
effectively.
Our
management will have broad discretion in the application of the net
proceeds from this offering and our stockholders will not have the
opportunity as part of their investment decisions to assess whether
the net proceeds are being used appropriately. You may not agree
with our decisions, and our use of the proceeds may not yield any
return on your investment. Because of the number and variability of
factors that will determine our use of the net proceeds from this
offering, their ultimate use may vary substantially from their
currently intended use. Our failure to apply the net proceeds of
this offering effectively could compromise our ability to pursue
our growth strategy and we might not be able to yield a significant
return, if any, in our investment of these net proceeds. You will
not have the opportunity to influence our decisions on how to use
our net proceeds from this offering.
The common stock offered hereby will be sold in
“at-the-market” offerings, and investors who buy shares
at different times will likely pay different prices.
Investors
who purchase shares in this offering at different times will likely
pay different prices, and so may experience different outcomes in
their investment results. We will have discretion, subject to
market demand, to vary the timing, prices, and numbers of shares
sold, and there is no minimum or maximum sales price. Investors may
experience a decline in the value of their shares as a result of
share sales made at prices lower than the prices they
paid.
The actual number of shares we will issue under the Sales
Agreement, at any one time or in total, is uncertain.
Subject to certain limitations in the Sales Agreement and
compliance with applicable law, we have the discretion to deliver a
sales notice to the Sales Agent at any time throughout the term of
the Sales Agreement. The number of shares that are sold by the
Sales Agent after we deliver a sales notice will fluctuate based on
the market price of the common stock during the sales period and
limits we set with the Sales Agent. Because the price per share of
each share sold will fluctuate based on the market price of our
common stock during the sales period, it is not possible at this
stage to predict the number of shares, if any, that will ultimately
be issued.
Our stock price and trading volume has fluctuated in the past, has
recently been volatile and may be volatile in the future for
reasons unrelated to our operating performance or prospects, and as
a result, investors in our common stock could incur substantial
losses.
Our stock price has fluctuated in the past, has recently been
volatile and may be volatile in the future. By way of example,
during 2021 to date, the market price of our common stock on the
Nasdaq has fluctuated from an intra-day low of $0.76 on January 4,
2021 to an intra-day high of $3.10 on March 18, 2021 and the last
reported sale price of our common stock on the Nasdaq on June 18,
2021 was $2.11 per share. During 2021 to date, daily trading volume
ranged from approximately 284,000 to approximately 33.3 million
shares. We may incur rapid and substantial decreases in our stock
price in the foreseeable future that are unrelated to our operating
performance or prospects. In addition, the COVID-19 pandemic has
caused broad stock market and industry fluctuations. The stock
market in general and the market for healthcare companies in
particular have experienced extreme volatility that has often been
unrelated to the operating performance of particular companies. As
a result of this volatility, investors may experience losses on
their investment in our common stock.
These broad market and industry factors may seriously harm the
market price of our common stock, regardless of our operating
performance. In the past, following periods of volatility in the
market, securities class-action litigation has often been
instituted against companies. Such litigation, if instituted
against us, could result in substantial costs and diversion of
management’s attention and resources, which could materially
and adversely affect our business, financial condition, results of
operations and growth prospects. There can be no guarantee that our
stock price will remain at current prices or that future sales of
our common stock will not be at prices lower than those sold to
investors.
Additionally,
recently, securities of certain companies have experienced
significant and extreme volatility in stock price due to a sudden
increase in demand for stock resulting in aggregate short positions
in the stock exceeding the number of shares available for purchase,
forcing investors with short exposure to pay a premium to
repurchase shares for delivery to share lenders. This is known as a
“short squeeze.” These short squeezes have led to the
price per share of those companies to trade at a significantly
inflated rate that is disconnected from the underlying value of the
company. Many investors who have purchased shares in those
companies at an inflated rate face the risk of losing a significant
portion of their original investment as the price per share
declines steadily as interest in those stocks abates. While we have
no reason to believe our shares would be the target of a short
squeeze, there can be no assurance that they will not be in the
future, and you may lose a significant portion or all of your
investment if you purchase our shares at a rate that is
significantly disconnected from their underlying value.
We may
issue and sell shares of our common stock having aggregate gross
proceeds of up to $20.0 million from time to time. Because there is
no minimum offering amount required as a condition to close this
offering, the actual total public offering amount, commissions paid
to the Sales Agent and proceeds to us, if any, are not determinable
at this time. We estimate that the net proceeds from the sale of
the shares of common stock that we are offering may be up to
approximately $19.3 million, after deducting commissions payable to
the Sales Agent and estimated offering expenses payable by
us.
We
intend to use the net proceeds of this offering for working capital
and other general corporate purposes. We have not yet determined
the amount of net proceeds to be used specifically for any
particular purposes or the timing of these expenditures.
Accordingly, our management will have significant discretion and
flexibility in applying the net proceeds from the sale of these
securities, and investors will be relying on our judgment regarding
the application of the net proceeds from this
offering.
Pending
our use of the net proceeds from this offering, we intend to
maintain the net proceeds as cash deposits or cash management
instruments, such as U.S. government securities or money market
mutual funds.
We have
not declared or paid any cash dividends on our capital stock. We
currently intend to retain any future earnings for use in the
operation of our business and do not anticipate paying any
dividends on our common stock in the foreseeable future. Any future
determination to declare dividends will be made at the discretion
of our board of directors and will depend on our financial
condition, operating results, capital requirements, general
business conditions and other factors that our board of directors
may deem relevant.
If you
purchase shares of common stock in this offering, you will
experience dilution to the extent of the difference between the
public offering price per share in this offering and our as
adjusted net tangible book value per share after this
offering.
Net
tangible book value per share represents total tangible assets less
total liabilities, divided by the number of shares of common stock
outstanding. Our historical net tangible book value as of March 31,
2021 was approximately $17.9 million, or $0.43 per share of common
stock. After giving effect to the sale of our common stock in the
aggregate amount of $20.0 million at an assumed public offering
price of $2.11 per share, the last reported sale price of our
common stock on the Nasdaq Capital Market on June 18, 2021, and after deducting
commissions and estimated offering expenses payable by us, our as
adjusted net tangible book value as of March 31, 2021 would have been
approximately $37.2 million, or approximately $0.73 per share. This
represents an immediate increase in net tangible book value of
approximately $0.30 per share to existing stockholders and an
immediate dilution in as adjusted net tangible book value of
approximately $1.38 per share to new investors in this
offering.
The
following table illustrates this dilution on a per share basis. The
as adjusted information is illustrative only and will adjust based
on the actual public offering price, the actual number of shares
sold and other terms of the offering determined at the time shares
of our common stock are sold pursuant to this prospectus
supplement. The as adjusted information assumes that all of our
common stock in the aggregate amount of $20.0 million is sold at
the assumed public offering price of $2.11 per share. The shares
sold in this offering, if any, will be sold from time to time at
various prices.
|
Assumed public
offering price per share
|
|
$2.11
|
|
Net tangible book
value per share as of March 31, 2021
|
$ 0.43
|
|
|
Increase in net
tangible book value per share attributable to this
offering
|
$ 0.30
|
|
|
As adjusted net
tangible book value per share, after giving effect to this
offering
|
|
$0.73
|
|
Dilution per share
to new investors in this offering
|
|
$1.38
|
The shares sold in this offering, if any, will be sold from time to
time at various prices. An increase of $0.25 per share in the price
at which the shares are sold from the assumed offering price of
$2.11 per share shown in the
table above, assuming all of our common stock in the aggregate
amount of $20.0 million is sold at that price,
would increase our adjusted net tangible book value per share after
the offering to $0.74 per share and would
increase the dilution in net tangible book value per share to new
investors in this offering to $1.62 per share, after
deducting commissions and estimated aggregate offering expenses
payable by us. A decrease of $0.25 per share in the price
at which the shares are sold from the assumed offering price of
$2.11 per share shown in the
table above, assuming all of our common stock in the aggregate
amount of $20.0 million is sold at that
price, would decrease our adjusted net tangible book value per
share after the offering to $0.71 per share and would
decrease the dilution in net tangible book value per share to new
investors in this offering to $1.15 per share, after
deducting commissions and estimated aggregate offering expenses
payable by us. This information is supplied for illustrative
purposes only, and will adjust based on the actual offering prices,
the actual number of shares that we offer and sell in this offering
and other terms of each sale of shares in this
offering.
The
above discussion and tables are based on 41,614,653 shares of
common stock outstanding as of March 31, 2021 and excludes the
following:
|
●
|
An
aggregate of 4,497,850 shares of common stock issued subsequent to
March 31, 2021 pursuant to the Prior Sales Agreement with the Sales
Agent;
|
|
●
|
1,932,000
shares of common stock issuable upon the exercise of outstanding
warrants issued in our initial public offering listed on the Nasdaq
Capital Market under the symbol “NDRAW,” at an exercise
price of $6.25 per share;
|
|
●
|
730,204
shares of common stock issuable upon the exercise of outstanding
unregistered warrants, at a weighted average exercise price of
$2.72 per share;
|
|
●
|
175,269
shares of common stock issuable upon the conversion of outstanding
shares of Series A Convertible Preferred Stock;
|
|
●
|
3,759,417
shares of common stock issuable upon the exercise of outstanding
stock options issued pursuant to our 2016 Omnibus Incentive Plan
(the “Incentive Plan”) at a weighted average exercise
price of $2.07 per share; and
|
|
●
|
3,691,811
shares of common stock reserved for future issuance under our
Incentive Plan.
|
We have
entered into an At-The-Market Issuance Sales Agreement with
Ascendiant Capital Markets, LLC, as Sales Agent, under which we may
issue and sell over a period of time, and from time to time, shares
of our common stock having an aggregate offering price of up to
$20.0 million through the Sales Agent acting as sales agent or
directly to a Sales Agent acting as principal. This prospectus
supplement relates to our ability to issue and sell over a period
of time, and from time to time, shares of our common stock to or
through the Sales Agent pursuant to the Sales Agreement. Sales of
the shares to which this prospectus supplement and the accompanying
prospectus relate, if any, may be made in transactions that are
deemed to be “at-the-market” offerings as defined in
Rule 415 under the Securities Act, including sales made directly on
or through the Nasdaq Capital Market (“Nasdaq”), the
trading market for our common stock, or any other trading market in
the Unites States for our common stock, sales made to or through a
market maker other than on an exchange, directly to the Sales Agent
as principal for its account in negotiated transactions at market
prices prevailing at the time of sale or at prices related to such
prevailing market prices, in privately negotiated transactions, in
block trades, or through a combination of any such methods of sale.
To the extent required by Regulation M, the Sales Agent acting as
our sales agent will not engage in any transactions that stabilize
our common stock while the offering is ongoing under this
prospectus supplement.
Upon
written instructions from us, the Sales Agent will offer the shares
of our common stock, subject to the terms and conditions of the
Sales Agreement, on a daily basis or as otherwise agreed upon by us
and the Sales Agent. We will designate the maximum amount of shares
of our common stock to be sold through the Sales Agent on a daily
basis or otherwise determine such maximum amount together with the
Sales Agent, subject to certain limitations set forth by the SEC.
Subject to the terms and conditions of the Sales Agreement, the
Sales Agent will use commercially reasonable efforts to sell on our
behalf all of the shares of our common stock so designated or
determined. We may instruct the Sales Agent not to sell shares of
our common stock if the sales cannot be effected at or above the
price designated by us in any such instruction. The Sales Agent may
also sell our common stock in negotiated transactions with our
prior approval. We or the Sales Agent may suspend the offering of
shares of our common stock being made under the Sales Agreement
upon proper notice to the other party.
For
their services as sales agent in connection with the sale of shares
of our common stock that may be offered hereby, we will pay the
Sales Agent an aggregate fee of 3.0% of the gross sales price per
share for any shares sold through it acting as our sales agent. The
remaining sales proceeds, after deducting any expenses payable by
us and any transaction fees imposed by any governmental, regulatory
or self-regulatory organization in connection with the sales, will
equal our net proceeds for the sale of such shares. We have agreed
to reimburse the Sales Agent for certain of its expenses in an
amount not to exceed $20,000, and, thereafter, reasonable fees and
expenses of the Sales Agent’s incurred in conjunction of
performing legal services related to the Sales Agreement for the
Company.
The
Sales Agent will provide written confirmation to us no later than
the opening of the trading day immediately following the day in
which shares of common stock are sold by it on our behalf under the
Sales Agreement. Each confirmation will include the number of
shares sold on that day, the compensation payable by us to the
Sales Agent and the proceeds to us net of such
compensation.
Settlement
for sales of our common stock will occur, unless the parties agree
otherwise, on the second business day following the date on which
any sales were made in return for payment of the proceeds to us net
of compensation paid by us to the Sales Agent. There is no
arrangement for funds to be received in an escrow, trust or similar
arrangement.
Unless
otherwise required, we will report at least quarterly the number of
shares of common stock sold through the Sales Agent under the Sales
Agreement, the net proceeds to us and the compensation paid by us
to the Sales Agent in connection with the sales of common
stock.
In
connection with the sale of common stock on our behalf, the Sales
Agent will be deemed to be an “underwriter” within the
meaning of the Securities Act, and the compensation paid to it will
be deemed to be underwriting commissions or discounts. We have
agreed, under the Sales Agreement, to provide indemnification and
contribution to the Sales Agent against certain civil liabilities,
including liabilities under the Securities Act.
In the
ordinary course of its business, the Sales Agent and/or its
affiliates may perform investment banking, broker-dealer, financial
advisory or other services for us for which it may receive separate
fees.
We
estimate that the total expenses from this offering payable by us,
excluding compensation payable to the Sales Agent under the Sales
Agreement, will be approximately $75,000. Additionally, pursuant to
the terms of the Sales Agreement, we agreed to reimburse the Sales
Agent for the reasonable fees and expenses of its legal counsel
incurred in connection with quarterly and annual bring-downs
required under the Sales Agreement in an amount not to exceed
$2,500 in the aggregate for each such bring-down.
The
offering of common stock pursuant to the Sales Agreement will
terminate upon the earlier of (1) the sale of shares of our common
stock with an aggregate offering price of $20.0 million subject to
the Sales Agreement, (2) June 21, 2024 and (3) the termination of
the Sales Agreement, pursuant to its terms, by either the Sales
Agent or us.
The
Company and the Sales Agent may in the future agree to add one or
more additional sales agents to the offering, in which case the
Company will file a further prospectus supplement providing the
name of such additional sales agents and any other required
information.
K&L
Gates LLP, Charlotte, North Carolina, will pass upon the validity
of the shares of common stock offered by this prospectus
supplement.
The financial
statements of ENDRA Life Sciences Inc. as of December 31, 2020 and
December 31, 2019 included in the Annual Report on Form 10-K for
the year ended December 31, 2020 have been audited by RBSM LLP,
independent registered public accounting firm, as stated in their
report, which is incorporated herein by reference. We have
incorporated these financial statements by reference in reliance
upon the report of RBSM LLP, given upon their authority as experts
in accounting and auditing.
WHERE YOU CAN FIND MORE
INFORMATION
We file
annual reports, quarterly reports, current reports, proxy
statements and other information with the SEC. Our SEC filings are
and will become available to the public over the Internet at the
SEC’s website at www.sec.gov. You can also find our public
filings on our website at www.cuebiopharma.com. Our website and the
information contained therein or connected thereto are not part of
this prospectus supplement or the accompanying
prospectus.
We have
filed with the SEC a registration statement on Form S-3 under the
Securities Act with respect to the shares of common stock being
offered by this prospectus supplement. This prospectus supplement
and the accompanying prospectus are a part of that registration
statement but do not contain all of the information set forth in
the registration statement or the exhibits to the registration
statement. For further information with respect to us and the
shares we are offering pursuant to this prospectus supplement, you
should refer to the registration statement and its exhibits.
Statements contained in this prospectus supplement as to the
contents of any contract, agreement or other document referred to
are not necessarily complete, and you should refer to the copy of
that contract or other documents filed as an exhibit to the
registration statement. You may read or obtain a copy of the
registration statement at the SEC’s website referred to
above.
INCORPORATION OF CERTAIN INFORMATION BY
REFERENCE
The SEC
allows us to “incorporate by reference” information
from other documents that we file with it, which means that we can
disclose important information to you by referring you to those
documents. The information incorporated by reference is considered
to be part of this prospectus supplement and the accompanying
prospectus. Information in this prospectus supplement supersedes
information incorporated by reference that we filed with the SEC
prior to the date of this prospectus supplement.
We
incorporate by reference the information or documents listed below
that we have filed with the SEC:
|
●
|
our annual report on Form 10-K for the fiscal year ended December
31, 2020 filed with the SEC on March
25, 2021;
|
|
●
|
our quarterly report on Form 10-Q for the quarterly period ended
March 31, 2021, which was filed with the SEC
on
May
17, 2021;
|
|
●
|
the information specifically incorporated by reference into our
Annual Report on Form 10-K for the fiscal year ended December 31,
2020 from our definitive proxy statement on Schedule 14A (other
than information furnished rather than filed), filed with the SEC
on
April 29,
2021;
and
|
|
●
|
the description of our common stock contained in our Registration
Statement on Form 8-A (File No. 001-37969) filed with the SEC
on December
16, 2016,
including any amendment or reports filed for the purpose of
updating such description.
|
All
documents we file with the SEC pursuant to Sections 13(a), 13(c),
14 or 15(d) of the Exchange Act, except as to any portion of any
report or documents that is not deemed filed under such provisions,
on or after the date of this prospectus supplement until the
termination of this offering shall be deemed incorporated by
reference in this prospectus supplement and the accompanying
prospectus and to be a part of this prospectus supplement from the
date of filing of those documents.
We will
furnish without charge to you, on written or oral request, a copy
of any or all of the documents incorporated by reference, including
exhibits to these documents. You should direct any requests for
documents to ENDRA Life Sciences Inc.,
3600 Green Court, Suite 350, Ann Arbor, Michigan 48105; Telephone:
(734) 335-0468. Copies of the above reports may also be accessed
from our website at www.endrainc.com. The information
contained on, or that may be obtained from, our website is not, and
shall not be deemed to be, a part of this prospectus supplement or
the accompanying prospectus.
We have
authorized no one to provide you with any information that differs
from that contained in this prospectus supplement, the accompanying
prospectus or incorporated by reference herein or therein.
Accordingly, you should not rely on any information that is not
contained in this prospectus supplement or the accompanying
prospectus or incorporated by reference herein or therein. You
should not assume that the information in this prospectus
supplement is accurate as of any date other than the date of the
front cover of this prospectus supplement.
Any
statement contained in a document incorporated or deemed to be
incorporated by reference in this prospectus supplement will be
deemed modified, superseded or replaced for purposes of this
prospectus supplement to the extent that a statement contained in
this prospectus supplement or any other subsequently filed document
that is deemed to be incorporated by reference into this prospectus
supplement modifies, supersedes or replaces such
statement.
ENDRA
Life Sciences Inc.
Prospectus
$50,000,000
Common
Stock
Preferred
Stock
Debt
Securities
Warrants
Units
This prospectus
relates to common stock, preferred stock, debt securities and
warrants that ENDRA Life Sciences Inc. may sell from time to time
in one or more offerings on terms to be determined at the time of
sale. We will provide specific terms of these securities in
supplements to this prospectus. You should read this prospectus and
any supplement carefully before you invest. This prospectus may not
be used to offer and sell securities unless accompanied by a
prospectus supplement for those securities.
These securities
may be sold directly by us, through dealers or agents designated
from time to time, to or through underwriters or through a
combination of these methods. See “Plan of
Distribution” in this prospectus. We may also describe the
plan of distribution for any particular offering of these
securities in any applicable prospectus supplement. If any agents,
underwriters or dealers are involved in the sale of any securities
in respect of which this prospectus is being delivered, we will
disclose their names and the nature of our arrangements with them
in a prospectus supplement. The net proceeds we expect to receive
from any such sale will also be included in a prospectus
supplement.
Our common stock
trades on the Nasdaq Capital Market under the symbol
“NDRA.” On March 24, 2021, the last reported sale price
for our Common Stock was $2.31 per share. Warrants issued in our
May 2017 initial public offering are traded on the Nasdaq Capital
Market under the symbol “NDRAW.”
We are an
“Emerging Growth Company” as defined in the Jumpstart
Our Business Startups Act of 2012 and, as such, have elected to
comply with certain reduced public company reporting requirements
for this prospectus and future filings. See “Prospectus
Summary − Implications of Being an Emerging Growth
Company.”
INVESTING
IN OUR SECURITIES INVOLVES RISKS. YOU SHOULD REVIEW CAREFULLY THE
RISKS AND UNCERTAINTIES DESCRIBED UNDER THE HEADING “RISK
FACTORS” CONTAINED IN THE APPLICABLE PROSPECTUS SUPPLEMENT
AND ANY RELATED FREE WRITING PROSPECTUS, AND UNDER SIMILAR HEADINGS
IN OTHER DOCUMENTS THAT ARE INCORPORATED BY REFERENCE INTO THIS
PROSPECTUS OR ANY SUCH PROSPECTUS SUPPLEMENT. SEE “RISK
FACTORS” ON PAGE 3 OF THIS PROSPECTUS.
Neither
the Securities and Exchange Commission nor any state securities
commission has approved or disapproved of these securities or
passed upon the adequacy or accuracy of this prospectus. Any
representation to the contrary is a criminal offense.
The date of this
prospectus is May 3, 2021.
Table
of Contents
|
|
Page
|
|
|
1
|
|
|
1
|
|
|
3
|
|
|
4
|
|
|
4
|
|
|
4
|
|
|
5
|
|
|
5
|
|
|
6
|
|
|
14
|
|
|
15
|
|
|
16
|
|
|
17
|
|
|
17
|
|
|
17
|
|
|
17
|
ABOUT
THIS PROSPECTUS
This prospectus is
part of a registration statement on Form S-3 that we filed with the
Securities and Exchange Commission, or the SEC, utilizing a
“shelf” registration process. Under this shelf process,
we may from time to time sell any combination of securities
described in this prospectus in one or more offerings.
This prospectus
provides you with a general description of the securities we may
offer. Each time we sell securities under this shelf registration
process, we will provide a prospectus supplement that will contain
specific information about the terms of the securities being
offered. That prospectus supplement may include a discussion of any
risk factors or other special consideration that apply to those
securities. The prospectus supplement may also add, update or
change information contained in this prospectus. If there is any
inconsistency between the information in this prospectus and a
prospectus supplement, you should rely on the information in that
prospectus supplement. You should read both this prospectus and any
applicable prospectus supplement together with additional
information described below under the headings “Where You Can
Find More Information” and “Incorporation by
Reference.”
When acquiring any
securities discussed in this prospectus, you should rely on the
information provided in this prospectus and the prospectus
supplement, including the information incorporated by reference.
Neither we, nor any underwriters or agents, have authorized anyone
to provide you with different information. We are not offering the
securities in any state where such an offer is prohibited. You
should not assume that the information in this prospectus, any
prospectus supplement, or any document incorporated by reference,
is truthful or complete at any date other than the date mentioned
on the cover page of those documents. You should also carefully
review the section entitled “Risk Factors”, which
highlights certain risks associated with an investment in our
securities, to determine whether an investment in our securities is
appropriate for you.
Unless otherwise
stated or the context requires otherwise, references to
“ENDRA”, the “Company,” “we,”
“us” or “our” are to ENDRA Life Sciences
Inc and its subsidiaries.
FORWARD-LOOKING STATEMENTS
Certain information
set forth in this prospectus or incorporated by reference in this
prospectus may contain forward-looking statements within the
meaning of Section 27A of the Securities Act of 1933, as amended
(the “Securities Act”), and Section 21E of the Exchange
Act, that are intended to be covered by the “safe
harbor” created by those sections. Forward-looking
statements, which are based on certain assumptions and describe our
future plans, strategies and expectations, can generally be
identified by the use of forward-looking terms such as
“believe,” “expect,” “may,”
“will,” “should,” “would,”
“could,” “seek,” “intend,”
“plan,” “estimate,” “goal,”
“anticipate,” “project” or other comparable
terms. All statements other than statements of historical facts
included in this prospectus regarding our strategies, prospects,
financial condition, operations, costs, plans and objectives are
forward-looking statements. Examples of forward-looking statements
include, among others, statements we make regarding: expectations
for revenues, cash flows and financial performance, the anticipated
results of our development efforts and the timing for receipt of
required regulatory approvals and product launches.
Forward-looking
statements are neither historical facts nor assurances of future
performance. Instead, they are based only on our current beliefs,
expectations and assumptions regarding the future of our business,
future plans and strategies, projections, anticipated events and
trends, the economy and other future conditions. Because
forward-looking statements relate to the future, they are subject
to inherent uncertainties, risks and changes in circumstances that
are difficult to predict and many of which are outside our control.
Our actual results and financial condition may differ materially
from those in the forward-looking statements. Therefore, you should
not rely on any of these forward-looking statements. Important
factors that could cause our actual results and financial condition
to differ materially from those indicated in the forward-looking
statements include, among others, the following:
● our limited
commercial experience, limited cash and history of
losses;
● our ability
to obtain adequate financing to fund our business operations in the
future;
● our ability
to achieve profitability;
● our ability
to develop a commercially feasible application based on our
Thermo-Acoustic Enhanced Ultrasound
(“TAEUS”) technology;
● market
acceptance of our technology;
●
uncertainties associated with COVID-19
or coronavirus, including its possible effects on our
operations;
● results of
our human studies, which may be negative or
inconclusive;
● our ability
to find and maintain development partners;
●
our reliance on third parties,
collaborations, strategic alliances and licensing arrangements to
complete our business strategy;
● the amount
and nature of competition in our industry;
● our ability
to protect our intellectual property;
● potential
changes in the healthcare industry or third-party reimbursement
practices;
● delays and
changes in regulatory requirements, policy and guidelines,
including potential delays in submitting required regulatory
applications or other submissions with respect to U.S. Food and
Drug Administration (“FDA”) or other regulatory agency
approval;
● our ability
to obtain CE mark certification and secure required FDA and other
governmental approvals for our TAEUS applications;
● our ability
to comply with regulation by various federal, state, local and
foreign governmental agencies and to maintain necessary regulatory
clearances or approvals;
● our ability
to maintain compliance with Nasdaq listing standards;
● our
dependence on our senior management team; and
● the other
risks and uncertainties described in the Risk Factors section of
this prospectus, any prospectus supplement and our most recent
Annual Form 10-K and subsequently filed Quarterly Reports on Form
10-Q, which filings are incorporated herein by
reference.
We urge you to
consider those risks and uncertainties in evaluating our
forward-looking statements. All subsequent written and oral
forward-looking statements attributable to us or to persons acting
on our behalf are expressly qualified in their entirety by the
applicable cautionary statements. We further caution readers not to
place undue reliance upon any such forward-looking statements,
which speak only as of the date made. Except as otherwise required
by the federal securities laws, we undertake no obligation to
publicly update any forward-looking statement, whether written or
oral, that may be made from time to time, whether as a result of
new information, future developments or otherwise.
We were
incorporated as a Delaware corporation in 2007. We are leveraging
experience with pre-clinical enhanced ultrasound devices to develop
technology for increasing the capabilities of clinical diagnostic
ultrasound in order to broaden patient access to the safe diagnosis
and treatment of a number of significant medical conditions in
circumstances where expensive X-ray computed tomography
(“CT”) and magnetic resonance imaging
(“MRI”) technology, or other diagnostic technologies
such as surgical biopsy, are unavailable or
impractical.
In 2010, we began
marketing and selling our Nexus 128 system, which combined
light-based thermoacoustics and ultrasound to address the imaging
needs of researchers studying disease models in pre-clinical
applications. Building on this expertise in thermoacoustics, we
have developed a next-generation technology platform — Thermo
Acoustic Enhanced Ultrasound, or TAEUS — which is intended to
enhance the capability of clinical ultrasound technology and
support the diagnosis and treatment of a number of significant
medical conditions that currently require the use of expensive CT
or MRI imaging or where imaging is not practical using existing
technology. We ceased production, service support and party for our
Nexus 128 system in July 2019 in order to focus our resources on
the development of our TAEUS technology.
Implications
of Being an Emerging Growth Company
We are an
“emerging growth company,” as defined in the Jumpstart
Our Business Startups Act of 2012, or the JOBS Act, and, for as
long as we continue to be an “emerging growth company,”
we may choose to take advantage of exemptions from various
reporting requirements applicable to other public companies but not
to “emerging growth companies,” including, but not
limited to, not being required to comply with the auditor
attestation requirements of Section 404 of the Sarbanes-Oxley Act
of 2002, reduced disclosure obligations regarding executive
compensation in our periodic reports and proxy statements, and
exemptions from the requirements of holding a nonbinding advisory
vote on executive compensation and stockholder approval of any
golden parachute payments not previously approved. We will remain
an “emerging growth company” until December 31, 2022
or, if earlier, (i) the last day of the first fiscal year in which
our annual gross revenues exceed $1.07 billion, (ii) the date that
we become a “large accelerated filer” as defined in
Rule 12b-2 under the Securities Exchange Act of 1934, as amended,
which would occur if the market value of our common stock that is
held by non-affiliates exceeds $700 million as of the last business
day of our most recently completed second fiscal quarter, or (iii)
the date on which we have issued more than $1 billion in
non-convertible debt during the preceding three-year period. We are
choosing to “opt out” of the extended transition
periods available under the JOBS Act for complying with new or
revised accounting standards, but intend to take advantage of the
other exemptions discussed above.
We are also
currently considered a “smaller reporting company,”
which generally means that we have a public float of less than $250
million or that our annual revenues are $100 million or less and we
have a public float of $700 million or less. If we are still
considered a “smaller reporting company” at such time
as we cease to be an “emerging growth company,” we will
be subject to increased disclosure requirements. However, the
disclosure requirements will still be less than they would be if we
were not considered either an “emerging growth company”
or a “smaller reporting company.” Specifically, similar
to “emerging growth companies,” “smaller
reporting companies” are able to provide simplified executive
compensation disclosures in their filings; are exempt, as
non-accelerated filers, from the provisions of Section 404(b) of
the Sarbanes-Oxley Act requiring that independent registered public
accounting firms provide an attestation report on the effectiveness
of internal control over financial reporting; and have certain
other decreased disclosure obligations in their Securities and
Exchange Commission, or SEC, filings, including, among other
things, being required to provide only two years of audited
financial statements in annual reports.
Corporate
Information
We were
incorporated in Delaware in July 2007. Our corporate headquarters
is located at 3600 Green Court, Suite 350, Ann Arbor, Michigan
48105-1570. Our website can be accessed at
www.endrainc.com. The telephone number of our principal
executive office is (734)
335-0468. The information contained on, or that may be
obtained from, our website is not, and shall not be deemed to be, a
part of this prospectus.
Investing in our
securities involves a high degree of risk. You should carefully
consider the risk factors described in our Annual Report on Form
10-K for our most recent fiscal year (together with any material
changes thereto contained in subsequent filed Quarterly Reports on
Form 10-Q) and those contained in our other filings with the SEC,
which are incorporated by reference in this prospectus and any
accompanying prospectus supplement.
The prospectus
supplement applicable to each type or series of securities we offer
may contain a discussion of risks applicable to the particular
types of securities that we are offering under that prospectus
supplement. Prior to making a decision about investing in our
securities, you should carefully consider the specific factors
discussed under the caption “Risk Factors” in the
applicable prospectus supplement, together with all of the other
information contained in the prospectus supplement or appearing or
incorporated by reference in this prospectus. These risks could
materially affect our business, results of operations or financial
condition and cause the value of our securities to decline. You
could lose all or part of your investment.
We currently intend
to use the estimated net proceeds from the sale of these securities
for working capital and general corporate purposes, including to
fund our product development efforts and to fund our
commercialization activities for our TAEUS technology. Accordingly,
our management will have significant discretion and flexibility in
applying the net proceeds from the sale of these securities. Our
plans to use the estimated net proceeds from the sale of these
securities may change, and if they do, we will update this
information in a prospectus supplement.
DESCRIPTION OF SECURITIES WE MAY
OFFER
We may issue from
time to time, in one or more offerings, the following
securities:
● shares of
common stock;
● shares of
preferred stock;
● debt
securities, which may include senior debt securities, subordinated
debt securities and senior subordinated debt
securities;
● warrants
for the purchase of debt securities, preferred stock or common
stock; and
● units
consisting of two or more of the foregoing.
Set forth below is
a description of the common stock and preferred stock that may be
offered under this prospectus. We will set forth in the applicable
prospectus supplement and/or free writing prospectus a description
of the debt securities and warrants that may be offered under this
prospectus. The terms of the offering of our common stock,
preferred stock or any such other securities, the initial offering
price and the net proceeds to us will be contained in the
prospectus supplement, and other offering material, relating to
such offer.
We may sell the
securities being offered pursuant to this prospectus directly to
purchasers, to or through underwriters, through dealers or agents,
or through a combination of such methods. The prospectus supplement
with respect to the securities being offered will set forth the
terms of the offering of those securities, including the names of
any such underwriters, dealers or agents, the purchase price, the
net proceeds to us, any underwriting discounts and other items
constituting underwriters’ compensation, the initial public
offering price, any discounts or concessions allowed or reallowed
or paid to dealers and any securities exchanges on which such
securities may be listed.
DESCRIPTION
OF COMMON STOCK WE MAY
OFFER
The following
summary description of our common stock is based on the provisions
of our certificate of incorporation and bylaws, and the applicable
provisions of the General Corporation Law of the State of Delaware.
This information may not be complete in all respects and is
qualified entirely by reference to the provisions of our Fourth
Amended and Restated Certificate of Incorporation (the
“Certificate of Incorporation”), and our amended and
restated bylaws (the “Bylaws”) and the General
Corporation Law of the State of Delaware. For information on how to
obtain copies of our Certificate of Incorporation and Bylaws, see
the discussion below under the heading “Where You Can Find
More Information.”
We may offer our
common stock issuable upon the conversion of debt securities or
preferred stock and upon the exercise of warrants.
Authorized
Capital
We currently have
authority to issue 80,000,000 shares of our common stock, par value
of $0.0001 per share.
As of March 24, 2021, 41,599,709 shares of our common stock were
issued and outstanding, held of record by 25 stockholders. Our
authorized but unissued shares of common stock are available for
issuance without further action by our stockholders, unless such
action is required by applicable law or the rules of any stock
exchange or automated quotation system on which our securities may
be listed or traded.
Voting
Rights
Each holder of our
common stock is entitled to one vote for each such share
outstanding in the holder’s name. No holder of
common stock is entitled to cumulate votes in voting for
directors.
Dividend
and Liquidation Rights
The holders of
outstanding shares of our common stock are entitled to such
dividends as may be declared by our board of directors out of funds
legally available for such purpose. The shares of our common stock
are neither redeemable nor convertible. Holders of our common stock
have no preemptive or subscription rights to purchase any of our
securities. In the event of our liquidation, dissolution or winding
up, the holders of our common stock are entitled to receive pro
rata our assets, which are legally available for distribution,
after payments of all debts and other liabilities. All
of the outstanding shares of our common stock are fully paid and
non-assessable.
We have never paid
any cash dividends on our common stock.
Our shares of
common stock are listed on the Nasdaq Capital Market under the
symbol “NDRA.”
DESCRIPTION
OF PREFERRED STOCK WE MAY
OFFER
This section
describes the general terms and provisions of the preferred stock
we may offer. This information may not be complete in all respects
and is qualified entirely by reference to our Certificate of
Incorporation, with respect to each series of preferred stock. The
specific terms of any series will be described in a prospectus
supplement. Those terms may differ from the terms discussed below.
Any series of preferred stock we issue will be governed by our
Certificate of Incorporation and by the certificate of designations
relating to that series. We will file the certificate of
designations with the SEC and incorporate it by reference as an
exhibit to our registration statement at or before the time we
issue any preferred stock of that series.
Authorized
Preferred Stock
Our Certificate of
Incorporation authorizes us to issue 10,000,000 shares of preferred
stock, par value $0.0001 per share. We have designated
10,000 shares of preferred stock as Series A Convertible Preferred
Stock (“Series A Preferred Stock”) and 1,000 shares of
its preferred stock as Series B Convertible Preferred Stock
(“Series B Preferred Stock”), and the remainder of
9,989,000 shares remain authorized but undesignated. As of March
24, 2021, we had 141.397 shares of Series A Preferred Stock and no
shares of Series B Preferred Stock issued and outstanding. Our
authorized but unissued shares of preferred stock are available for
issuance without further action by our stockholders, unless such
action is required by applicable law or the rules of any stock
exchange or automated quotation system on which our securities may
be listed or traded.
Our board of
directors has the authority to issue preferred stock in one or more
series and to fix the designations, powers, rights, preferences,
qualifications, limitations and restrictions thereof. These
designations, powers, rights and preferences could include voting
rights, dividend rights, dissolution rights, conversion rights,
exchange rights, redemption rights, liquidation preferences, and
the number of shares constituting any series or the designation of
such series, any or all of which may be greater than the rights of
common stock. The issuance of preferred stock could adversely
affect the voting power of holders of common stock and the
likelihood that such holders will receive dividend payments and
payments upon liquidation. In addition, the issuance of preferred
stock could have the effect of delaying, deferring or preventing
change in our control or other corporate action.
Specific
Terms of a Series of Preferred Stock
The preferred stock
we may offer under this prospectus will be issued in one or more
series. A prospectus supplement will discuss the following features
of the series of preferred stock to which it relates:
● the
designations and stated value per share;
● the number
of shares offered;
● the amount
of liquidation preference per share;
● the public
offering price at which the preferred stock will be
issued;
● the
dividend rate, the method of its calculation, the dates on which
dividends would be paid and the dates, if any, from which dividends
would cumulate;
● any
redemption or sinking fund provisions;
● any
conversion or exchange rights; and
● any
additional voting, dividend, liquidation, redemption, sinking fund
and other rights, preferences, privileges, limitations and
restrictions.
DESCRIPTION
OF DEBT SECURITIES WE MAY
OFFER
General
The debt securities
that we may issue will constitute debentures, notes, bonds or other
evidences of indebtedness of ENDRA, to be issued in one or more
series, which may include senior debt securities, subordinated debt
securities and senior subordinated debt securities. The particular
terms of any series of debt securities we offer, including the
extent to which the general terms set forth below may be applicable
to a particular series, will be described in a prospectus
supplement relating to such series.
Debt securities
that we may issue will be issued under an indenture between us and
a trustee qualified to act as such under the Trust Indenture Act of
1939. We have filed the form of the indenture as an exhibit to the
registration statement of which this prospectus is a part. When we
refer to the “indenture” in this prospectus, we are
referring to the indenture under which the debt securities are
issued as supplemented by any supplemental indenture applicable to
the debt securities. We will provide the name of the trustee in any
prospectus supplement related to the issuance of debt securities,
and we will also provide certain other information related to the
trustee, including describing any relationship we have with the
trustee, in such prospectus supplement.
THE FOLLOWING
DESCRIPTION IS A SUMMARY OF THE MATERIAL PROVISIONS OF THE
INDENTURE. IT DOES NOT RESTATE THE INDENTURE IN ITS ENTIRETY. THE
INDENTURE IS GOVERNED BY THE TRUST INDENTURE ACT OF 1939. THE TERMS
OF THE DEBT SECURITIES INCLUDE THOSE STATED IN THE INDENTURE AND
THOSE MADE PART OF THE INDENTURE BY REFERENCE TO THE TRUST
INDENTURE ACT. WE URGE YOU TO READ THE INDENTURE BECAUSE IT, AND
NOT THIS DESCRIPTION, DEFINES YOUR RIGHTS AS A HOLDER OF THE DEBT
SECURITIES.
Information
You Will Find in the Prospectus Supplement
The indenture
provides that we may issue debt securities from time to time in one
or more series and that we may denominate the debt securities and
make them payable in foreign currencies. The indenture does not
limit the aggregate principal amount of debt securities that can be
issued thereunder. The prospectus supplement for a series of debt
securities will provide information relating to the terms of the
series of debt securities being offered, which may
include:
● the title
and denominations of the debt securities of the
series;
● any limit
on the aggregate principal amount of the debt securities of the
series;
● the date or
dates on which the principal and premium, if any, with respect to
the debt securities of the series are payable or the method of
determination thereof;
● the rate or
rates, which may be fixed or variable, at which the debt securities
of the series shall bear interest, if any, or the method of
calculating and/or resetting such rate or rates of
interest;
● the dates
from which such interest shall accrue or the method by which such
dates shall be determined and the duration of the extensions and
the basis upon which interest shall be calculated;
● the
interest payment dates for the series of debt securities or the
method by which such dates will be determined, the terms of any
deferral of interest and any right of ours to extend the interest
payment periods;
● the place
or places where the principal and interest on the series of debt
securities will be payable;
● the terms
and conditions upon which debt securities of the series may be
redeemed, in whole or in part, at our option or
otherwise;
● our
obligation, if any, to redeem, purchase, or repay debt securities
of the series pursuant to any sinking fund or other specified event
or at the option of the holders and the terms of any such
redemption, purchase, or repayment;
● the terms,
if any, upon which the debt securities of the series may be
convertible into or exchanged for other securities, including,
among other things, the initial conversion or exchange price or
rate and the conversion or exchange period;
● if the
amount of principal, premium, if any, or interest with respect to
the debt securities of the series may be determined with reference
to an index or formula, the manner in which such amounts will be
determined;
● if any
payments on the debt securities of the series are to be made in a
currency or currencies (or by reference to an index or formula)
other than that in which such securities are denominated or
designated to be payable, the currency or currencies (or index or
formula) in which such payments are to be made and the terms and
conditions of such payments;
● any changes
or additions to the provisions of the indenture dealing with
defeasance, including any additional covenants that may be subject
to our covenant defeasance option;
● the
currency or currencies in which payment of the principal and
premium, if any, and interest with respect to debt securities of
the series will be payable, or in which the debt securities of the
series shall be denominated, and the particular provisions
applicable thereto in accordance with the indenture;
● the portion
of the principal amount of debt securities of the series which will
be payable upon declaration of acceleration or provable in
bankruptcy or the method by which such portion or amount shall be
determined;
● whether the
debt securities of the series will be secured or guaranteed and, if
so, on what terms;
● any
addition to or change in the events of default with respect to the
debt securities of the series;
● the
identity of any trustees, authenticating or paying agents, transfer
agents or registrars;
● the
applicability of, and any addition to or change in, the covenants
currently set forth in the indenture;
● the
subordination, ranking or priority, if any, of the debt securities
of the series and terms of the subordination;
● any other
terms of the debt securities of the series which are not prohibited
by the indenture; and
Holders of debt
securities may present debt securities for exchange in the manner,
at the places, and subject to the restrictions set forth in the
debt securities, the indenture, and the prospectus supplement. We
will provide these services without charge, other than any tax or
other governmental charge payable in connection therewith, but
subject to the limitations provided in the indenture, any board
resolution establishing such debt securities and any applicable
indenture supplement.
Senior
Debt
We may issue senior
debt securities under the indenture. Unless otherwise set forth in
the applicable indenture supplement and described in a prospectus
supplement, the senior debt securities will be senior unsecured
obligations, ranking equally with all of our existing and future
senior unsecured debt. The senior debt securities will be senior to
all of our subordinated debt and junior to any secured debt we may
incur as to the assets securing such debt.
Subordinated
Debt
We may issue
subordinated debt securities under the indenture. These
subordinated debt securities will be subordinate and junior in
right of payment, to the extent and in the manner set forth in the
indenture and any applicable indenture supplement, to all of our
senior indebtedness.
If this prospectus
is being delivered in connection with a series of subordinated debt
securities, the accompanying prospectus supplement or the
information incorporated by reference will set forth the
approximate amount of senior indebtedness outstanding as of the end
of the most recent fiscal quarter.
Senior
Subordinated Debt
We may issue senior
subordinated debt securities under the indenture. These senior
subordinated debt securities will be, to the extent and in the
manner set forth in the applicable indenture supplement,
subordinate and junior in right of payment to all of our
“senior indebtedness” and senior to our other
subordinated debt. See the discussions above under
“—Senior Debt” and “—Subordinated
Debt” for a more detailed explanation of our senior and
subordinated indebtedness.
Interest
Rate
Debt securities
that bear interest will do so at a fixed rate or a variable rate.
We may sell, at a discount below the stated principal amount, any
debt securities which bear no interest or which bear interest at a
rate that at the time of issuance is below the prevailing market
rate. The relevant prospectus supplement will describe the special
United States federal income tax considerations applicable
to:
● any
discounted debt securities; and
● any debt
securities issued at par which are treated as having been issued at
a discount for United States federal income tax
purposes.
Registered
Global Securities
We may issue
registered debt securities of a series in the form of one or more
fully registered global securities. We will deposit the registered
global security with a depository or with a nominee for a
depository identified in the prospectus supplement relating to such
series. The global security or global securities will represent and
will be in a denomination or aggregate denominations equal to the
portion of the aggregate principal amount of outstanding registered
debt securities of the series to be represented by the registered
global security or securities. Unless it is exchanged in whole or
in part for debt securities in definitive registered form, a
registered global security may not be transferred, except as a
whole in three cases:
● by the
depository for the registered global security to a nominee of the
depository;
● by a
nominee of the depository to the depository or another nominee of
the depository; and
● by the
depository or any nominee to a successor of the depository or a
nominee of the successor.
The prospectus
supplement relating to a series of debt securities will describe
the specific terms of the depository arrangement concerning any
portion of that series of debt securities to be represented by a
registered global security. We anticipate that the following
provisions will generally apply to all depository
arrangements.
Upon the issuance
of a registered global security, the depository will credit, on its
book-entry registration and transfer system, the principal amounts
of the debt securities represented by the registered global
security to the accounts of persons that have accounts with the
depository. These persons are referred to as
“participants.” Any underwriters, agents or dealers
participating in the distribution of debt securities represented by
the registered global security will designate the accounts to be
credited. Only participants or persons that hold interests through
participants will be able to beneficially own interests in a
registered global security. The depository for a global security
will maintain records of beneficial ownership interests in a
registered global security for participants. Participants or
persons that hold through participants will maintain records of
beneficial ownership interests in a global security for persons
other than participants. These records will be the only means to
transfer beneficial ownership in a registered global
security.
The laws of some
states may require that specified purchasers of securities take
physical delivery of the securities in definitive form. These laws
may limit the ability of those persons to own, transfer or pledge
beneficial interests in global securities.
So long as the
depository, or its nominee, is the registered owner of a registered
global security, the depository or its nominee will be considered
the sole owner or holder of the debt securities represented by the
registered global security for all purposes under the indenture.
Except as set forth below, owners of beneficial interests in a
registered global security:
● may not
have the debt securities represented by a registered global
security registered in their names;
● will not
receive or be entitled to receive physical delivery of debt
securities represented by a registered global security in
definitive form; and
● will not be
considered the owners or holders of debt securities represented by
a registered global security under the indenture.
Accordingly, each
person owning a beneficial interest in a registered global security
must rely on the procedures of the depository for the registered
global security and, if the person is not a participant, on the
procedures of the participant through which the person owns its
interests, to exercise any rights of a holder under the indenture
applicable to the registered global security.
We understand that,
under existing industry practices, if we request any action of
holders, or if an owner of a beneficial interest in a registered
global security desires to give or take any action which a holder
is entitled to give or take under the indenture, the depository for
the registered global security would authorize the participants
holding the relevant beneficial interests to give or take the
action, and the participants would authorize beneficial owners
owning through the participants to give or take the action or would
otherwise act upon the instructions of beneficial owners holding
through them.
Payment
of Interest on and Principal of Registered Global
Securities
We will make
principal, premium, if any, and interest payments on debt
securities represented by a registered global security registered
in the name of a depository or its nominee to the depository or its
nominee as the registered owner of the registered global security.
None of ENDRA, the trustee, or any paying agent for debt securities
represented by a registered global security will have any
responsibility or liability for:
● any aspect
of the records relating to, or payments made on account of,
beneficial ownership interests in such registered global
security;
●
maintaining, supervising, or reviewing any records relating to
beneficial ownership interests;
● the
payments to beneficial owners of the global security of amounts
paid to the depository or its nominee; or
● any other
matter relating to the actions and practices of the depository, its
nominee or any of its participants.
We expect that the
depository, upon receipt of any payment of principal, premium or
interest in respect of the global security, will immediately credit
participants’ accounts with payments in amounts proportionate
to their beneficial interests in the principal amount of a
registered global security as shown on the depository’s
records. We also expect that payments by participants to owners of
beneficial interests in a registered global security held through
participants will be governed by standing instructions and
customary practices. This is currently the case with the securities
held for the accounts of customers registered in “street
name.” Such payments will be the responsibility of
participants.
Exchange
of Registered Global Securities
We may issue debt
securities in definitive form in exchange for the registered global
security if both of the following occur:
● the
depository for any debt securities represented by a registered
global security is at any time unwilling or unable to continue as
depository or ceases to be a clearing agency registered under the
Exchange Act; and
● we do not
appoint a successor depository within 90 days.
In addition, we
may, at any time, determine not to have any of the debt securities
of a series represented by one or more registered global
securities. In this event, we will issue debt securities of that
series in definitive form in exchange for all of the registered
global security or securities representing those debt
securities.
Covenants
by ENDRA
The indenture
includes covenants by us, including among other things that we will
make all payments of principal and interest at the times and places
required. The supplemental indenture establishing each series of
debt securities may contain additional covenants, including
covenants which could restrict our right to incur additional
indebtedness or liens and to take certain actions with respect to
our businesses and assets.
Events
of Default
Unless otherwise
indicated in the applicable prospectus supplement, the following
will be events of default under the indenture with respect to each
series of debt securities issued under the indenture:
● failure to
pay when due any interest on any debt security of that series,
continued for 30 days;
● failure to
pay when due the principal of, or premium, if any, on, any debt
security of that series;
● failure to
perform any other covenant or agreement of ours under the indenture
or the supplemental indenture with respect to that series or the
debt securities of that series, continued for 90 days after
written notice to us by the trustee or holders of at least 25% in
aggregate principal amount of the outstanding debt securities of
the series to which the covenant or agreement relates;
● certain
events of bankruptcy, insolvency or similar proceedings affecting
us; and
● any other
event of default specified in any supplemental indenture under
which such series of debt securities is issued.
Except as to
certain events of bankruptcy, insolvency or similar proceedings
affecting us and except as provided in the applicable prospectus
supplement, if any event of default shall occur and be continuing
with respect to any series of debt securities under the indenture,
either the trustee or the holders of at least 25% in aggregate
principal amount of outstanding debt securities of such series may
accelerate the maturity of all debt securities of such series. Upon
certain events of bankruptcy, insolvency or similar proceedings
affecting us, the principal, premium, if any, and interest on all
debt securities of each series shall be immediately due and
payable.
After any such
acceleration, but before a judgment or decree based on acceleration
has been obtained by the trustee, the holders of a majority in
aggregate principal amount of each affected series of debt
securities may waive all defaults with respect to such series and
rescind and annul such acceleration if all events of default, other
than the non-payment of accelerated principal, have been cured,
waived or otherwise remedied.
No holder of any
debt securities will have any right to institute any proceeding
with respect to the indenture or for any remedy under the
indenture, unless such holder shall have previously given to the
trustee written notice of a continuing event of default and the
holders of at least 25% in aggregate principal amount of the
outstanding debt securities of the relevant series shall have made
written request and offered indemnity satisfactory to the trustee
to institute such proceeding as trustee, and the trustee shall not
have received from the holders of a majority in aggregate principal
amount of the outstanding debt securities of such series a
direction inconsistent with such request and shall have failed to
institute such proceeding within 60 days. However, such
limitations do not apply to a suit instituted by a holder of a debt
security for enforcement of payment of the principal of and
premium, if any, or interest on such debt security on or after the
respective due dates expressed in such debt security.
Supplemental
Indentures
We and the trustee
may, at any time and from time to time, without prior notice to or
consent of any holders of debt securities, enter into one or more
indentures supplemental to the indenture, among other
things:
● to add
guarantees to or secure any series of debt securities;
● to provide
for the succession of another person pursuant to the provisions of
the indenture relating to consolidations, mergers and sales of
assets and the assumption by such successor of our covenants,
agreements, and obligations, or to otherwise comply with the
provisions of the indenture relating to consolidations, mergers,
and sales of assets;
● to
surrender any right or power conferred upon us under the indenture
or to add to our covenants further covenants, restrictions,
conditions or provisions for the protection of the holders of all
or any series of debt securities;
● to cure any
ambiguity or to correct or supplement any provision contained in
the indenture, in any supplemental indenture or in any debt
securities that may be defective or inconsistent with any other
provision contained therein;
● to modify
or amend the indenture in such a manner as to permit the
qualification of the indenture or any supplemental indenture under
the Trust Indenture Act;
● to add to
or change any of the provisions of the indenture to supplement any
of the provisions of the indenture in order to permit the
defeasance and discharge of any series of debt securities pursuant
to the indenture, so long as any such action does not adversely
affect the interests of the holders of debt securities of any
series in any material respect;
● to add to,
change, or eliminate any of the provisions of the indenture with
respect to one or more series of debt securities, so long as any
such addition, change or elimination shall not apply to any debt
securities of any series created prior to the execution of such
supplemental indenture and entitled to the benefit of such
provision;
● to evidence
and provide for the acceptance of appointment by a successor or
separate trustee; and
● to
establish the form or terms of debt securities of any series and to
make any change that does not adversely affect the interests of the
holders of debt securities.
With the consent of
the holders of at least a majority in principal amount of debt
securities of each series affected by such supplemental indenture
(each series voting as one class), we and the trustee may enter
into one or more supplemental indentures for the purpose of adding
any provisions to or changing in any manner or eliminating any of
the provisions of the indenture or modifying in any manner the
rights of the holders of debt securities of each such
series.
Notwithstanding our
rights and the rights of the trustee to enter into one or more
supplemental indentures with the consent of the holders of debt
securities of the affected series as described above, no such
supplemental indenture shall, without the consent of the holder of
each outstanding debt security of the affected series, among other
things:
● change the
final maturity of the principal of, or any installment of interest
on, any debt securities;
● reduce the
principal amount of any debt securities or the rate of interest on
any debt securities;
● change the
currency in which any debt securities are payable;
● impair the
right of the holders to conduct a proceeding for any remedy
available to the trustee;
● reduce the
percentage in principal amount of any series of debt securities
whose holders must consent to an amendment or supplemental
indenture;
● modify the
ranking or priority of the securities; or
● reduce any
premium payable upon the redemption of any debt
securities.
Satisfaction
and Discharge of the Indenture; Defeasance
Except to the
extent set forth in a supplemental indenture with respect to any
series of debt securities, we, at our election, may discharge the
indenture and the indenture shall generally cease to be of any
further effect with respect to that series of debt securities if
(a) we have delivered to the trustee for cancellation all debt
securities of that series (with certain limited exceptions) or
(b) all debt securities of that series not previously
delivered to the trustee for cancellation shall have become due and
payable, or are by their terms to become due and payable within one
year or are to be called for redemption within one year, and we
have deposited with the trustee the entire amount sufficient to pay
at maturity or upon redemption all such debt
securities.
In addition, we
have a “legal defeasance option” (pursuant to which we
may terminate, with respect to the debt securities of a particular
series, all of our obligations under such debt securities and the
indenture with respect to such debt securities) and a
“covenant defeasance option” (pursuant to which we may
terminate, with respect to the debt securities of a particular
series, our obligations with respect to such debt securities under
certain specified covenants contained in the indenture). If we
exercise our legal defeasance option with respect to a series of
debt securities, payment of such debt securities may not be
accelerated because of an event of default. If we exercise our
covenant defeasance option with respect to a series of debt
securities, payment of such debt securities may not be accelerated
because of an event of default related to the specified
covenants.
We may exercise our
legal defeasance option or our covenant defeasance option with
respect to the debt securities of a series only if we irrevocably
deposit in trust with the trustee cash or U.S. government
obligations (as defined in the indenture) for the payment of
principal, premium, if any, and interest with respect to such debt
securities to maturity or redemption, as the case may be. In
addition, to exercise either of our defeasance options, we must
comply with certain other conditions, including the delivery to the
trustee of an opinion of counsel to the effect that the holders of
debt securities of such series will not recognize income, gain or
loss for Federal income tax purposes as a result of such defeasance
and will be subject to Federal income tax on the same amounts, in
the same manner and at the same times as would have been the case
if such defeasance had not occurred (and, in the case of legal
defeasance only, such opinion of counsel must be based on a ruling
from the Internal Revenue Service or other change in applicable
Federal income tax law).
The trustee will
hold in trust the cash or U.S. government obligations deposited
with it as described above and will apply the deposited cash and
the proceeds from deposited U.S. government obligations to the
payment of principal, premium, if any, and interest with respect to
the debt securities of the defeased series.
Mergers,
Consolidations and Certain Sales of Assets
We may
not:
● consolidate
with or merge into any other person or entity or permit any other
person or entity to consolidate with or merge into us in a
transaction in which we are not the surviving entity,
or
● transfer,
lease or dispose of all or substantially all of our assets to any
other person or entity,
unless:
o the resulting,
surviving or transferee entity shall be a corporation organized and
existing under the laws of the United States or any state thereof
and such resulting, surviving or transferee entity shall expressly
assume, by supplemental indenture, executed and delivered in form
satisfactory to the trustee, all of our obligations under the debt
securities and the indenture;
o immediately after
giving effect to such transaction (and treating any indebtedness
which becomes an obligation of the resulting, surviving or
transferee entity as a result of such transaction as having been
incurred by such entity at the time of such transaction), no
default or event of default would occur or be continuing;
and
o we shall have
delivered to the trustee an officers’ certificate and an
opinion of counsel, each stating that such consolidation, merger or
transfer and such supplemental indenture (if any) comply with the
indenture.
The phrase
“substantially all” of our assets will likely be
interpreted under applicable state law and will be dependent upon
particular facts and circumstances. As a result, there may be a
degree of uncertainty in ascertaining whether a sale or transfer of
“substantially all” of our assets has
occurred.
Governing
Law
The indenture and
the debt securities will be governed by the laws of the State of
New York.
No
Personal Liability of Directors, Officers, Employees and
Stockholders
No director,
officer, incorporator or stockholder of ENDRA, as such, shall have
any liability for any obligations of ENDRA under the debt
securities or the indenture or for any claim based on, in respect
of, or by reason of, such obligations or their creation, solely by
reason of his, her, or its status as director, officer,
incorporator or stockholder of ENDRA. By accepting a debt security,
each holder waives and releases all such liability, but only such
liability. The waiver and release are part of the consideration for
issuance of the debt securities. Nevertheless, such waiver may not
be effective to waive liabilities under the federal securities laws
and it has been the view of the SEC that such a waiver is against
public policy.
Conversion
or Exchange Rights
Any debt securities
offered hereby may be convertible into or exchangeable for shares
of our equity or other securities. The terms and conditions of such
conversion or exchange will be set forth in the applicable
prospectus supplement. Such terms may include, among others, the
following:
● the
conversion or exchange price;
● the
conversion or exchange period;
● provisions
regarding our ability or that of the holder to convert or exchange
the debt securities;
● events
requiring adjustment to the conversion or exchange price;
and
● provisions
affecting conversion or exchange in the event of our redemption of
such debt securities.
Concerning
the Trustee
The indenture
provides that there may be more than one trustee with respect to
one or more series of debt securities. If there are different
trustees for different series of debt securities, each trustee will
be a trustee of a trust under a supplemental indenture separate and
apart from the trust administered by any other trustee under such
indenture. Except as otherwise indicated in this prospectus or any
prospectus supplement, any action permitted to be taken by a
trustee may be taken by the trustee only with respect to the one or
more series of debt securities for which it is the trustee under an
indenture. Any trustee under the indenture or a supplemental
indenture may resign or be removed with respect to one or more
series of debt securities. All payments of principal of, premium,
if any, and interest on, and all registration, transfer, exchange
authentication and delivery (including authentication and delivery
on original issuance of the debt securities) of, the debt
securities of a series will be effected by the trustee with respect
to such series at an office designated by the trustee.
The indenture
contains limitations on the right of the trustee, should it become
a creditor of ENDRA, to obtain payment of claims in certain cases
or to realize on certain property received in respect of any such
claim as security or otherwise. If the trustee acquires an interest
that conflicts with any duties with respect to the debt securities,
the trustee is required to either resign or eliminate such
conflicting interest to the extent and in the manner provided by
the indenture.
DESCRIPTION
OF WARRANTS WE MAY
OFFER
We may issue
warrants for the purchase of debt securities, preferred stock or
common stock. Warrants may be issued independently or together with
debt securities, preferred stock or common stock and may be
attached to or separate from any offered securities. Any issue of
warrants will be governed by the terms of the applicable form of
warrant and any related warrant agreement which we will file with
the SEC and they will be incorporated by reference to the
registration statement of which this prospectus is a part on or
before the time we issue any warrants.
The particular
terms of any issue of warrants will be described in the prospectus
supplement relating to the issue. Those terms may
include:
● the title
of such warrants;
● the
aggregate number of such warrants;
● the price
or prices at which such warrants will be issued;
● the
currency or currencies (including composite currencies) in which
the price of such warrants may be payable;
● the terms
of the securities purchasable upon exercise of such warrants and
the procedures and conditions relating to the exercise of such
warrants;
● the price
at which the securities purchasable upon exercise of such warrants
may be purchased;
● the date on
which the right to exercise such warrants will commence and the
date on which such right shall expire;
● any
provisions for adjustment of the number or amount of securities
receivable upon exercise of the warrants or the exercise price of
the warrants;
● if
applicable, the minimum or maximum amount of such warrants that may
be exercised at any one time;
● if
applicable, the designation and terms of the securities with which
such warrants are issued and the number of such warrants issued
with each such security;
● if
applicable, the date on and after which such warrants and the
related securities will be separately transferable;
● information
with respect to book-entry procedures, if any; and
● any other
terms of such warrants, including terms, procedures and limitations
relating to the exchange or exercise of such warrants.
The prospectus
supplement relating to any warrants to purchase equity securities
may also include, if applicable, a discussion of certain U.S.
federal income tax and ERISA considerations.
Warrants for the
purchase of preferred stock and common stock will be offered and
exercisable for U.S. dollars only.
Each warrant will
entitle its holder to purchase the principal amount of debt
securities or the number of shares of preferred stock or common
stock at the exercise price set forth in, or calculable as set
forth in, the applicable prospectus supplement.
After the close of
business on the expiration date, unexercised warrants will become
void. We will specify the place or places where, and the manner in
which, warrants may be exercised in the applicable prospectus
supplement.
Prior to the
exercise of any warrants to purchase debt securities, preferred
stock or common stock, holders of the warrants will not have any of
the rights of holders of the debt securities, preferred stock or
common stock purchasable upon exercise.
As
of March 24, 2021, warrants to purchase a total of 575,641 shares
of common stock at a weighted average exercise price of $1.77 per
share were outstanding. Warrants to purchase total of 1,932,000
shares of common stock that were issued as part of the units issued
in our initial public offering, which expire in May 2022, have an
exercise price of $6.25 per share. Warrants to purchase a total of
154,560 shares of common stock issued to the underwriters in our
initial public offering also expire in May 2022 and have an
exercise price of $6.25 per share. Warrants to purchase a total of
190,234 shares of common stock expire in June 2021 and have an
exercise price of $2.52 per share. Warrants to purchase a total of
44,333 shares of common stock expire in October 2023 and have an
exercise price of $2.625 per share. Warrants to purchase a total of
205,768 shares of common stock expire in July 2022 and have an
exercise price of $1.49 per share. Warrants to purchase a total of
115,499 shares of common stock expire in December 2024 and have an
exercise price of $0.87 per share. Warrants to purchase a total of
11,948 shares of common stock expire in December 2024 and have an
exercise price of $0.99 per share. Warrants to purchase a total of
7,859 shares of common stock have varying exercise prices and
expiration dates.
DESCRIPTION
OF UNITS WE MAY OFFER
We may issue units
consisting of any combination of the other types of securities
offered under this prospectus in one or more series. We may
evidence each series of units by unit certificates that we will
issue under a separate agreement. We may enter into unit agreements
with a unit agent. Each unit agent will be a bank or trust company
that we select. We will indicate the name and address of the unit
agent in the applicable prospectus supplement relating to a
particular series of units.
The following
description, together with the additional information included in
any applicable prospectus supplement, summarizes the general
features of the units that we may offer under this prospectus. You
should read any prospectus supplement and any free writing
prospectus that we may authorize to be provided to you related to
the series of units being offered, as well as the complete unit
agreements that contain the terms of the units. Specific unit
agreements will contain additional important terms and provisions
and we will file as an exhibit to the registration statement of
which this prospectus is a part, or will incorporate by reference
from another report that we file with the SEC, the form of each
unit agreement relating to units offered under this
prospectus.
If we offer any
units, certain terms of that series of units will be described in
the applicable prospectus supplement, including, without
limitation, the following, as applicable:
● the title
of the series of units;
●
identification and description of the separate constituent
securities comprising the units;
● the price
or prices at which the units will be issued;
● the date,
if any, on and after which the constituent securities comprising
the units will be separately transferable;
● a
discussion of certain United States federal income tax
considerations applicable to the units; and
● any other
terms of the units and their constituent securities.
We may sell the
securities offered by this prospectus to one or more underwriters
or dealers for public offering, through agents, directly to
purchasers or through a combination of any such methods of sale.
The name of any such underwriters, dealers or agents involved
in the offer and sale of the securities, the amounts underwritten
and the nature of its obligation to take the securities will be
specified in the applicable prospectus supplement. We have
reserved the right to sell the securities directly to investors on
our own behalf in those jurisdictions where we are authorized to do
so. The sale of the securities may be effected in
transactions (a) on any national or international securities
exchange or quotation service on which the securities may be listed
or quoted at the time of sale, (b) in the over-the-counter market,
(c) in transactions otherwise than on such exchanges or in the
over-the-counter market or (d) through the writing of
options.
We and our agents
and underwriters may offer and sell the securities at a fixed price
or prices that may be changed, at market prices prevailing at the
time of sale, at prices related to such prevailing market prices or
at negotiated prices. The securities may be offered on an
exchange, which will be disclosed in the applicable prospectus
supplement. We may, from time to time, authorize dealers,
acting as our agents, to offer and sell the securities upon such
terms and conditions as set forth in the applicable prospectus
supplement. We may also
sell the securities offered by any applicable prospectus supplement
in “at-the-market offerings” within the meaning of Rule
415 of the Securities Act, to or through a market maker or into an
existing trading market, on an exchange or otherwise.
If we use
underwriters to sell securities, we will enter into an underwriting
agreement with them at the time of the sale to them. In
connection with the sale of the securities, underwriters may
receive compensation from us in the form of underwriting discounts
or commissions and may also receive commissions from purchasers of
the securities for whom they may act as agent. Any
underwriting compensation paid by us to underwriters or agents in
connection with the offering of the securities, and any discounts,
concessions or commissions allowed by underwriters to participating
dealers, will be set forth in the applicable prospectus supplement
to the extent required by applicable law. Underwriters may
sell the securities to or through dealers, and such dealers may
receive compensation in the form of discounts, concessions or
commissions from the underwriters or commissions (which may be
changed from time to time) from the purchasers for whom they may
act as agents.
Dealers and agents
participating in the distribution of the securities may be deemed
to be underwriters, and any discounts and commissions received by
them and any profit realized by them on resale of the securities
may be deemed to be underwriting discounts and commissions under
the Securities Act. Unless otherwise indicated in the
applicable prospectus supplement, an agent will be acting on a best
efforts basis and a dealer will purchase debt securities as a
principal, and may then resell the debt securities at varying
prices to be determined by the dealer.
If so indicated in
the prospectus supplement, we will authorize underwriters, dealers
or agents to solicit offers by certain specified institutions to
purchase offered securities from us at the public offering price
set forth in the prospectus supplement pursuant to delayed delivery
contracts providing for payment and delivery on a specified date in
the future. Such contracts will be subject to any conditions
set forth in the applicable prospectus supplement and the
prospectus supplement will set forth the commission payable for
solicitation of such contracts. The underwriters and other
persons soliciting such contracts will have no responsibility for
the validity or performance of any such contracts.
Underwriters,
dealers and agents may be entitled, under agreements entered into
with us, to indemnification against and contribution towards
certain civil liabilities, including any liabilities under the
Securities Act.
To facilitate the
offering of securities, certain persons participating in the
offering may engage in transactions that stabilize, maintain, or
otherwise affect the price of the securities. These may
include over-allotment, stabilization, syndicate short covering
transactions and penalty bids. Over-allotment involves sales
in excess of the offering size, which creates a short position.
Stabilizing transactions involve bids to purchase the
underlying security so long as the stabilizing bids do not exceed a
specified maximum. Syndicate short covering transactions
involve purchases of securities in the open market after the
distribution has been completed in order to cover syndicate short
positions. Penalty bids permit the underwriters to reclaim
selling concessions from dealers when the securities originally
sold by the dealers are purchased in covering transactions to cover
syndicate short positions. These transactions may cause the
price of the securities sold in an offering to be higher than it
would otherwise be. These transactions, if commenced, may be
discontinued by the underwriters at any time.
Any securities
other than our common stock issued hereunder may be new issues of
securities with no established trading market. Any
underwriters or agents to or through whom such securities are sold
for public offering and sale may make a market in such securities,
but such underwriters or agents will not be obligated to do so and
may discontinue any market making at any time without notice.
No assurance can be given as to the liquidity of the trading
market for any such securities. The amount of expenses
expected to be incurred by us in connection with any issuance of
securities will be set forth in the applicable prospectus
supplement. Certain of the underwriters, dealers or agents
and their associates may engage in transactions with, and perform
services for, us and certain of our affiliates in the ordinary
course of business.
During such time as
we may be engaged in a distribution of the securities covered by
this prospectus we are required to comply with Regulation M
promulgated under the Exchange Act. With certain exceptions,
Regulation M precludes us, any affiliated purchasers, and any
broker-dealer or other person who participates in such distribution
from bidding for or purchasing, or attempting to induce any person
to bid for or purchase, any security which is the subject of the
distribution until the entire distribution is complete.
Regulation M also restricts bids or purchases made in order
to stabilize the price of a security in connection with the
distribution of that security. All of the foregoing may
affect the marketability of our shares of common
stock.
The validity and
legality of the securities offered hereby and certain other legal
matters will be passed upon for the Company by K&L Gates LLP,
Charlotte, North Carolina.
EXPERTS
RBSM LLP,
independent registered public accounting firm, has audited our
financial statements included in our Annual Report on Form 10-K for
the year ended December 31, 2020, which is incorporated by
reference into this prospectus and elsewhere in the registration
statement of which this prospectus is a part. Our financial
statements are incorporated by reference in reliance on RBSM
LLP’s report, given on their authority as experts in
accounting and auditing.
WHERE
YOU CAN FIND MORE INFORMATION
We file annual,
quarterly and special reports, proxy statements and other
information with the Securities and Exchange Commission (the
“SEC”). You can inspect and copy these reports, proxy
statements and other information at the SEC’s Public
Reference Room at 100 F Street, N.E., Washington, D. C. 20549.
Please call the SEC at 1-800-SEC-0330 for further information on
the Public Reference Room. The SEC also maintains a web site that
contains reports, proxy and information statements and other
information regarding issuers, such as ENDRA Life Sciences Inc.
(www.sec.gov). Our web site is located at www.endrainc.com. The
information contained on, or that may be obtained from, our website
is not, and shall not be deemed to be, a part of this
prospectus.
We will provide,
upon written or oral request, without charge to you, including any
beneficial owner to whom this prospectus is delivered, a copy of
any or all of the documents incorporated herein by reference other
than the exhibits to those documents, unless the exhibits are
specifically incorporated by reference into the information that
this prospectus incorporates. You should direct a request for
copies to ENDRA Life Sciences Inc., 3600 Green Court, Suite 350,
Ann Arbor, Michigan 48105; Telephone: (734) 335-0468.
INCORPORATION
BY REFERENCE
The SEC allows us
to “incorporate by reference” information from other
documents that we file with it, which means that we can disclose
important information to you by referring you to those documents.
The information incorporated by reference is considered to be part
of this prospectus. Information in this prospectus supersedes
information incorporated by reference that we filed with the SEC
prior to the date of this prospectus.
We incorporate by
reference into this prospectus and the registration statement of
which this prospectus is a part the information or documents listed
below that we have filed with the SEC:
● Annual
Report on Form 10-K for the fiscal year ended December 31, 2020
filed with the SEC on March 25, 2021;
● Current
Reports on Form 8-K filed with the SEC on February 3, 2021 and
February 19, 2021;
● The
information incorporated by reference into our Annual Report on
Form 10-K for the fiscal year ended December 31, 2019 from our
Proxy Statement on Schedule 14A related to our 2020 Annual Meeting
of Stockholders, filed with the SEC on April 29, 2020;
and
● The
description of the Company’s Common Stock contained in the
Company’s Registration Statement on Form 8-A (File No.
001-37969) initially filed with the SEC on December 16, 2016
pursuant to Section 12(g) of the Exchange Act, including any
amendment or reports filed for the purpose of updating such
description.
We also incorporate
by reference any future filings (other than current reports
furnished under Item 2.02 or Item 7.01 of Form 8-K and exhibits
filed on such form that are related to such items unless such Form
8-K expressly provides to the contrary) made with the SEC pursuant
to Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act,
including those made on or after the date of the initial filing of
the registration statement of which this prospectus is a part and
prior to effectiveness of such registration statement, until we
file a post-effective amendment that indicates the termination of
the offering of the Securities made by this prospectus and will
become a part of this prospectus from the date that such documents
are filed with the SEC. Information in such future filings updates
and supplements the information provided in this prospectus. Any
statements in any such future filings will automatically be deemed
to modify and supersede any information in any document we
previously filed with the SEC that is incorporated or deemed to be
incorporated herein by reference to the extent that statements in
the later filed document modify or replace such earlier
statements.
We will furnish
without charge to you, on written or oral request, a copy of any or
all of the documents incorporated by reference, including exhibits
to these documents. You should direct any requests for documents to
ENDRA Life Sciences Inc., 3600 Green Court, Suite 350, Ann Arbor,
Michigan 48105; Telephone: (734) 335-0468. Copies of the above
reports may also be accessed from our web site at www.endrainc.com. We have authorized no
one to provide you with any information that differs from that
contained in this prospectus. Accordingly, you should not rely on
any information that is not contained in this prospectus. You
should not assume that the information in this prospectus is
accurate as of any date other than the date of the front cover of
this prospectus.
Any statement
contained in a document incorporated or deemed to be incorporated
by reference in this prospectus will be deemed to be modified or
superseded for purposes of this prospectus to the extent that a
statement contained in this prospectus or in any other subsequently
filed document which also is or is deemed to be incorporated by
reference in this prospectus modifies or supersedes that statement.
Any statement that is modified or superseded will not constitute a
part of this prospectus, except as modified or
superseded.
Up to $20,000,000 of
Common Stock
PROSPECTUS
SUPPLEMENT
June 21,
2021
ENDRA Life Sciences (NASDAQ:NDRA)
Historical Stock Chart
From Aug 2024 to Sep 2024
ENDRA Life Sciences (NASDAQ:NDRA)
Historical Stock Chart
From Sep 2023 to Sep 2024