Bayer Seeks FDA OK to Extend Mirena Duration to Six Years
October 21 2019 - 11:13AM
Dow Jones News
By Colin Kellaher
Bayer AG (BAYN.XE) on Monday said it filed a supplemental
new-drug application with the U.S. Food and Drug Administration
seeking to extend the approved duration of its Mirena intrauterine
device to six years.
The Leverkusen, Germany, chemical-and-pharmaceutical company
said the request is based on results from a phase 3 extension trial
evaluating the efficacy and safety of the contraceptive.
Mirena, which in 2000 became the first hormonal intrauterine
device cleared for use in the U.S., is currently approved to
prevent pregnancy for up to five years.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
October 21, 2019 10:58 ET (14:58 GMT)
Copyright (c) 2019 Dow Jones & Company, Inc.
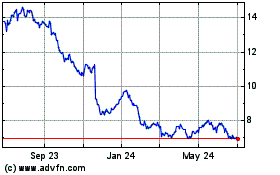
Bayer Aktiengesellschaft (PK) (USOTC:BAYRY)
Historical Stock Chart
From Aug 2024 to Sep 2024
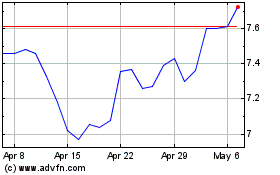
Bayer Aktiengesellschaft (PK) (USOTC:BAYRY)
Historical Stock Chart
From Sep 2023 to Sep 2024