Filed Pursuant to Rule
424(b)(3)
Registration No. 333-200461
$6,250,000

OCULUS INNOVATIVE
SCIENCES, INC.
1,250,000 Shares
of Common Stock and
Warrants to Purchase
937,500 Shares of Common Stock
We previously offered 1,250,000 shares of our
common stock, $0.0001 par value per share, together with warrants to purchase 937,500 shares of our common stock, as adjusted for
the reverse split of our common stock effected on June 24, 2016. Five shares of common stock were sold together with 3.75 of a
warrant. Five warrants are exercisable for one share of common stock at an exercise price of $6.50 per share commencing upon
consummation of this offering and terminating on the fifth anniversary of the date of issuance.
All costs
associated with this registration will be borne by us. Our common stock is traded on The NASDAQ Capital Market under the trading
symbol “OCLS.”
The warrants issued in this offering began
trading on The NASDAQ Capital Market on January 21, 2015.
None
of our other warrants are listed or traded on a national securities exchange or market. On
September 8,
2016, the last reported sale price of our common stock on The NASDAQ Capital Market was $4.34 per share.
|
|
|
Per 5 Shares*
|
|
|
|
Per 3.75 of a Warrant (1)*
|
|
|
|
Total*
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Public offering price
|
|
$
|
4.95
|
|
|
|
$
|
0.05
|
|
|
|
$
|
5.00
|
|
|
|
Underwriter discounts and commissions (2)
|
|
$
|
0.396
|
(4)
|
|
|
$
|
0.004
|
(4)
|
|
|
$
|
0.40
|
(4)
|
|
|
Proceeds, before expenses, to us (3)
|
|
$
|
4.554
|
(4)
|
|
|
$
|
0.046
|
(4)
|
|
|
$
|
4.60
|
(4)
|
|
*All share numbers and share prices have
been adjusted to reflect the reverse stock split effective June 24, 2016.
(1) Five shares of common stock were
sold together with 3.75 of a warrant, with each five warrants being exercisable for the purchase of one share of common stock.
(2) We issued warrants to the underwriters
to reimburse the underwriters for certain expenses. See “Underwriting - Other Terms” on page 68 of this prospectus
for a description of these arrangements.
(3) We estimate the total expenses
of this offering were approximately $485,000. See “Underwriting - Commissions” on page 69 of this prospectus.
(4) The underwriter discounts and
commissions for each five shares together with 3.75 of a warrant were $0.40, or $500,000 in the aggregate. The total proceeds,
before expenses, to us from the sale of the shares together with the warrants were $5,750,000.
The underwriters delivered the securities,
against payment, on or about January 26, 2015.
We granted the underwriters a 45-day
option to purchase 187,500 additional shares of common stock and/or additional warrants to purchase 140,625 additional shares of
common stock from us at the offering price for each security, less underwriting discounts and commissions, to cover over-allotments,
if any.
THIS INVESTMENT
INVOLVES A HIGH DEGREE OF RISK. YOU SHOULD PURCHASE
SECURITIES ONLY
IF YOU CAN AFFORD A COMPLETE LOSS.
SEE
“RISK FACTORS” BEGINNING ON PAGE 4.
We
will not sell any more shares of the Common Stock or Warrants described in this Prospectus Supplement.
Neither the Securities
and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this
prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
Sole Book-Running Manager
Maxim Group LLC
Co-Manager
Dawson James Securities, Inc.
The date of this prospectus
supplement is September 9, 2016.
TABLE OF CONTENTS
|
|
Page
|
|
Prospectus Summary
|
1
|
|
Risk Factors
|
4
|
|
Cautionary Note Regarding Forward-looking Statements
|
20
|
|
Use of Proceeds
|
20
|
|
Dilution
|
20
|
|
Price Range of our Common Stock
|
21
|
|
Dividend Policy
|
21
|
|
Capitalization
|
21
|
|
Management’s Discussion and Analysis of Financial Condition and Results of Operations
|
22
|
|
Our Business
|
33
|
|
Management
|
50
|
|
Certain Relationships and Related Transactions
|
65
|
|
Security Ownership of Certain Beneficial Owners and Management
|
65
|
|
Description of Securities
|
67
|
|
Underwriting
|
68
|
|
Legal Matters
|
72
|
|
Experts
|
72
|
|
Interests of Named Experts and Counsel
|
72
|
|
Where You Can Find More Information
|
72
|
|
Disclosure of Commission Position on Indemnification for Securities Act Liabilities
|
73
|
|
Financial Statements
|
F-1
|
OCULUS INNOVATIVE
SCIENCES, INC.
PROSPECTUS SUMMARY
The following information
is a summary of the prospectus and it does not contain all of the information you should consider before making an investment decision.
You should read the entire prospectus carefully, including the financial statements and the notes relating to the financial statements.
OUR BUSINESS
We are a specialty pharmaceutical company dedicated
to identifying, developing and commercializing unique, affordable differentiated therapies to improve the lives of patients with
dermatologic diseases or conditions. Our products, which are sold throughout the United States and internationally, have improved
patient outcomes for more than five million patients globally by treating and reducing certain topical skin diseases including
acne, atopic dermatitis, scarring, infections, itch, pain and harmful inflammatory responses.
We currently focus on the development and commercialization
of therapeutic solutions in medical dermatology to treat or reduce skin conditions, such as acne, atopic dermatitis and scarring.
These diseases impact millions of patients worldwide and can have significant, multi-dimensional effects on patients’ quality
of life, including their physical, functional and emotional well-being.
Since our founding in 1999, we built our business
by developing and promoting products via partnerships for multiple therapeutic indications, with a primary focus on advanced tissue
care. Starting in 2013, with a new Board of Directors and new management team, we pivoted to focus on one specialty pharmaceutical
area, medical dermatology, and created our own sales force in the United States to promote our unique, affordable, differentiated
prescription dermatology products.
Some of our key products in the United States
are:
|
|
·
|
Celacyn®, a prescription hypochlorous acid based scar management gel clinically proven to soften and flatten raised scars while reducing redness and discoloration.
|
|
|
·
|
Ceramax™ Skin Barrier Cream helps manage dry itchy skin, minor skin irritations, rashes, and inflammation caused by various skin conditions.
|
|
|
·
|
Alevicyn™, a prescription hypochlorous acid based atopic dermatitis product line clinically proven to reduce pruritus (itch) and pain associated with various dermatoses.
|
|
|
·
|
Mondoxyne™, a prescription oral tetracycline antibiotic used for the treatment of certain bacterial infections, including acne.
|
|
|
·
|
Microcyn® or Microdacyn60® (sold under a variety of brand names), a line of products based on electrically charged oxychlorine small molecules designed to target a wide range of pathogens including viruses, fungi, spores and bacteria, including antibiotic-resistant strains.
|
Our key product outside the United States is:
|
|
·
|
Microcyn® or Microdacyn60® (sold under a variety of brand names), a line of products based on electrically charged oxychlorine small molecules designed to target a wide range of pathogens including viruses, fungi, spores and bacteria, including antibiotic-resistant strains.
|
To date, we have obtained 15 clearances from
the U.S. Food and Drug Administration, or FDA, that permit us to sell our products as medical devices for Section 510(k) of the
Federal Food, Drug and Cosmetic Act in the United States.
Outside the United Sates, we sell products
for dermatological and advanced tissue care with a European Conformity marking (known as Conformité Européenne or
CE) covering 25 of our products, 14 approvals from the Mexican Ministry of Health, and various approvals in Central America, China,
Southeast Asia, and the Middle East.
Our Strategy
Our strategy is to in-license, acquire, develop
and commercialize unique, affordable and differentiated therapies that we believe advance the standard of care for patients with
dermatological diseases. The key components of our strategy are to:
|
|
·
|
Expand our Internal U.S. Sales Force:
We continue to
hire additional experienced sales people who have established relationships with dermatologists in their territories. As
of March 31, 2016, we had a U.S. direct sales force team of 19 dedicated sales people.
|
|
|
·
|
Develop and Launch New Dermatology Products:
We currently sell six prescription dermatology products in the United States, and have a strong product pipeline of new products, including our new product, Lasercyn, intended for the management of post-non-ablative laser therapy procedures, post-microdermabrasion therapy and following superficial chemical peels, that we intend to launch over the next nine months.
|
|
|
·
|
Create a Competitive Pricing Strategy:
We have and will continue to develop a unique product pricing strategy, which we believe solves many of the challenges associated with the prescription dermatology market’s current pricing and rebate programs.
|
|
|
·
|
Develop a Pharmaceutical Line:
We plan to acquire or develop pharmaceutical products with affordable clinical trials to increase our market presence and create innovator patent protection.
|
Our plan is to evolve into a leading dermatology
company, providing innovative and cost-effective solutions to patients, while generating strong, consistent revenue growth
and maximizing long-term shareholder value.
THE OFFERING
|
Common stock outstanding as of January 13, 2015 (1)
|
|
1,732,116 shares
|
|
|
|
|
|
Common Stock issued on January 26, 2015
|
|
1,250,000 shares
|
|
|
|
|
Common Stock issued pursuant to the
over-allotment option on March 6, 2015
|
|
26,900 shares
|
|
|
|
|
|
Warrants issued on January 26, 2015
|
|
4,687,500 warrants to purchase an aggregate of 937,500 shares
|
|
|
|
|
Warrants issued pursuant to the
over-allotment option on January 26, 2015
|
|
703,125 warrants to purchase an aggregate of 140,625 shares
|
|
|
|
|
|
Description of Warrants
|
|
Each five warrants entitle the holder to purchase one share of common stock at a purchase price equal to $6.50 per share, at any time commencing upon consummation of this offering and terminating on the fifth anniversary of the date of issuance. See “Description of Securities – Warrants.”
|
|
|
|
|
|
Underwriters’ Over Allotment Option
|
|
The underwriting agreement provided that we grant to the underwriters an option, exercisable within 45 days after the closing of this offering, to acquire an additional 15% of the total number of shares of common stock and/or warrants to be offered by us pursuant to this offering, solely for the purpose of covering over-allotments.
|
|
|
|
|
|
Use of Proceeds
|
|
We intend to use the proceeds from the sale of the shares and from the exercise of warrants, if any, to increase our direct sales force, to develop and launch new products and for general working capital. See “Use of Proceeds.”
|
|
|
|
|
|
Stock Symbol
|
|
OCLS
|
|
|
|
|
|
Stock Symbol for the Warrants
|
|
OCLSW
|
|
|
|
|
|
Risk Factors
|
|
Investing in our securities involves substantial risks. You should carefully review and consider the “Risk Factors” section of this prospectus beginning on page 4 and the other information in this prospectus for a discussion of the factors you should consider before you decide to invest in this offering.
|
|
(1)
|
Excludes shares of common stock issuable upon exercise
of 571,933 outstanding options and 406,333 warrants as of January 13, 2015.
|
SUMMARY FINANCIAL INFORMATION
Because this is only a summary of our financial
information, it does not contain all of the financial information that may be important to you. Therefore, you should carefully
read all of the information in this prospectus and any prospectus supplement, including the financial statements and their explanatory
notes and the section entitled “Management’s Discussion and Analysis of Financial Condition and Results of Operations,”
before making a decision to invest in our common stock. The information contained in the following summary is derived from our
audited, consolidated financial statements for the fiscal years ended March 31, 2016 and 2015 (in thousands, except share and per
share amounts).
|
|
|
Year ended
|
|
|
|
|
March 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
|
|
|
|
|
|
|
|
Total revenues
|
|
$
|
15,084
|
|
|
$
|
13,854
|
|
|
Total cost of revenues
|
|
|
7,874
|
|
|
|
6,566
|
|
|
Gross profit
|
|
|
7,210
|
|
|
|
7,288
|
|
|
Operating expenses
|
|
|
|
|
|
|
|
|
|
Research and development
|
|
|
1,806
|
|
|
|
1,533
|
|
|
Selling, general and administrative
|
|
|
15,556
|
|
|
|
12,414
|
|
|
Total operating expenses
|
|
|
17,362
|
|
|
|
13,947
|
|
|
Loss from operations
|
|
|
(10,152
|
)
|
|
|
(6,659
|
)
|
|
Net (loss) income
|
|
$
|
(10,162
|
)
|
|
$
|
(8,203
|
)
|
|
(Loss) earnings per common share: basic and diluted
|
|
$
|
(0.62
|
)
|
|
$
|
(0.85
|
)
|
|
|
|
March 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
|
|
|
|
|
Balance Sheet Data:
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
7,469
|
|
|
$
|
6,136
|
|
|
Working capital
|
|
|
9,337
|
|
|
|
7,066
|
|
|
Total assets
|
|
|
13,803
|
|
|
|
15,048
|
|
|
Accumulated deficit
|
|
|
(152,375
|
)
|
|
|
(142,213
|
)
|
|
Total stockholders’ equity
|
|
$
|
10,140
|
|
|
$
|
12,054
|
|
RISK FACTORS
Investing in our securities involves a high
degree of risk. This prospectus contains a discussion of risks applicable to an investment in the securities offered. Prior to
making a decision about investing in our securities, you should carefully consider the specific factors discussed below together
with all of the other information contained in this prospectus or appearing or incorporated by reference in this prospectus.
Risks Related to Our Business
We have a history of losses, we expect
to continue to incur losses and we may never
achieve profitability and our March 31, 2016 audited consolidated financial
statements included disclosure that casts substantial doubt regarding our ability to continue as a going concern.
We reported a net loss of $10,162,000 and $8,203,000
and for the year ended March 31, 2016 and 2015, respectively. At March 31, 2016 and 2015, our accumulated deficit amounted to $152,375,000
and $142,213,000, respectively. We had working capital of $9,337,000 and $7,066,000 as of March 31, 2016 and 2015, respectively.
During the year ended March 31, 2016 and 2015, net cash used in operating activities amounted to $8,746,000 and $6,694,000, respectively.
We incurred a net loss of $2,568,000 for the three months ended June 30, 2016. At June 30, 2016, our accumulated deficit amounted
to $154,943,000. At June 30, 2016, our working capital amounted to $6,965,000. We expect to continue incurring losses for the foreseeable
future and may never achieve or sustain profitability. We must raise additional capital to pursue our product development initiatives,
penetrate markets for the sale of our products and continue as a going concern. We cannot provide any assurance that we will raise
additional capital. We believe that we have access to capital resources through possible public or private equity offerings, debt
financings, corporate collaborations or other means. We may not raise enough capital in this offering to meet our needs and we
may have to raise additional capital in the future. If we are unable to secure additional capital, we may be required to curtail
our research and development initiatives and take additional measures to reduce costs in order to conserve our cash in amounts
sufficient to sustain operations and meet our obligations. These measures could cause significant delays in our efforts to further
commercialize our products, which are critical to the realization of our business plan and to our future operations. These matters
raise substantial doubt about our ability to continue as a going concern or become profitable.
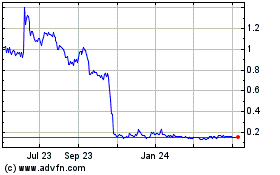
Our Board of Directors elected to effect
a reverse stock split of our outstanding common stock, which may affect our overall market capitalization and may decrease the
liquidity of the common stock.
On June 29, 2015, our stockholders approved
an amendment to our Restated Certificate of Incorporation, as amended, and authorized our Board of Directors, if in their judgment
it is necessary, to effect a reverse stock split of our outstanding common stock, $0.0001 par value per share, at a whole number
ratio in the range of 1-for-5 to 1-for-9, such ratio to be determined in the discretion of our Board of Directors, and to proportionally
decrease the total number of shares that we are authorized to issue by a factor of 1-for-5 to 1-for-9, such ratio to be determined
in the sole discretion of our Board of Directors, in conjunction with the proposed reverse split, and authorized our Board of Directors
to file such amendment, if in their judgment it is necessary, that would effect the foregoing in order to regain compliance with
the minimum bid requirement of NASDAQ. On June 2, 2016, our Board of Directors approved the reverse stock split with a ratio of
1-for-5 and authorized the Corporation to take all steps necessary to effect the reverse stock split which became effective June
24, 2016 after the market closed. Should the market price of our common stock decline after the reverse stock split the percentage
decline may be greater, due to the smaller number of shares outstanding, than it would have been prior to the reverse stock split.
A reverse stock split is often viewed negatively by the market and, consequently, can lead to a decrease in our overall market
capitalization. If the per share market price does not increase in proportion to the reverse stock split ratio, then the value
of our Company, as measured by our stock capitalization, will be reduced. In some cases, the per-share stock price of companies
that have effected reverse stock splits subsequently declined back to pre-reverse split levels, and accordingly, we cannot assure
you that the total market value of your shares will remain the same after the reverse stock split is effected, or that the reverse
stock split will not have an adverse effect on our stock price due to the reduced number of shares outstanding after the reverse
stock split. The reverse stock split may decrease the liquidity of the common stock. Although our Board of Directors believes that
the anticipated increase in the market price of our common stock could encourage interest in our common stock and possibly promote
greater liquidity for our stockholders, such liquidity could also be adversely affected by the reduced number of shares outstanding
after the reverse stock split. Our authorized common share capital was also reduced by a factor of 1-for-5.
If we are unable to expand our direct
domestic sales force, we may not be able to successfully sell our products in the United States.
We currently use a direct sales force to sell
our products in the dermatology markets. Expanding our sales force is expensive and time consuming, and the lack of qualified sales
personnel could delay or limit the success of our product launch in the United States. Our domestic sales force competes with the
sales operations of our competitors, which are better funded and more experienced. We may not be able to expand our domestic sales
capacity on a timely basis, or in the markets that we desire, or at all.
Our inability to raise additional capital
on acceptable terms in the future may cause
us to curtail certain operational activities, including regulatory trials,
sales and
marketing, and international operations, in order to reduce costs and sustain the
business,
and such inability would have a material adverse effect on our business and financial
condition.
We expect capital outlays and operating expenditures
to increase over the next several years as we work to expand our sales force, conduct regulatory trials, commercialize our products
and expand our infrastructure. We may need to raise additional capital in order to, among other things:
|
|
·
|
fund our clinical trials and preclinical studies;
|
|
|
·
|
sustain commercialization of our current products or new products;
|
|
|
·
|
expand our manufacturing capabilities;
|
|
|
·
|
increase our sales and marketing efforts to drive market adoption and address competitive developments;
|
|
|
·
|
acquire or license technologies;
|
|
|
·
|
finance capital expenditures and our general and administrative expenses; and
|
Our present and future funding requirements
will depend on many factors, including:
|
|
·
|
the progress and timing of our clinical trials;
|
|
|
·
|
the level of research and development investment required to maintain and improve our technology position;
|
|
|
·
|
cost of filing, prosecuting, defending and enforcing patent claims and other intellectual property rights;
|
|
|
·
|
our efforts to acquire or license complementary technologies or acquire complementary businesses;
|
|
|
·
|
changes in product development plans needed to address any difficulties in commercialization;
|
|
|
·
|
competing technological and market developments; and
|
|
|
·
|
changes in regulatory policies or laws that affect our operations.
|
If we raise additional funds by issuing equity
securities, dilution to our stockholders will result. Any equity securities issued also may provide for rights, preferences or
privileges senior to those of holders of our common stock. If we raise additional funds by issuing debt securities, these debt
securities would have rights, preferences and privileges senior to those of holders of our common stock, and the terms of the debt
securities issued could impose significant restrictions on our operations. If we raise additional funds through collaborations
or licensing arrangements, we might be required to relinquish significant rights to our technologies or products, or grant licenses
on terms that are not favorable to us. A failure to obtain adequate funds may cause us to curtail certain operational activities,
including regulatory trials, sales and marketing, and international operations, in order to reduce costs and sustain our business,
and would have a material adverse effect on our business and financial condition.
We do not have the necessary regulatory
approvals to market Microcyn
®
as a drug in the
United States.
We have obtained 14 510(k) clearances in the
United States that permit us to sell Microcyn®-based and other products as medical devices. However, before we are permitted
to sell Microcyn® as a drug in the United States, we must, among other things, successfully complete additional preclinical
studies and well-controlled clinical trials, submit a new drug application to the FDA and obtain FDA approval.
The FDA approval process is expensive and uncertain,
requires detailed and comprehensive scientific and other data and generally takes several years. Despite the time and expense exerted,
approval is never guaranteed. Even if we obtain FDA approval to sell Microcyn® as a drug, we may not be able to successfully
commercialize Microcyn® as a drug in the United States and may never recover the substantial costs we have invested in the
development of our Microcyn®-based products.
Delays or adverse results in clinical
trials could result in increased costs to us
and could delay our ability to generate revenue.
Clinical trials can be long and expensive,
and the outcome of clinical trials is uncertain and subject to delays. It may take several years to complete clinical trials, if
at all, and a product candidate may fail at any stage of the clinical trial process. The length of time required varies substantially
according to the type, complexity, novelty and intended use of the product candidate. Interim results of a preclinical study or
clinical trial do not necessarily predict final results, and acceptable results in preclinical studies or early clinical trials
may not be repeatable in later subsequent clinical trials. The commencement or completion of any of our clinical trials may be
delayed or halted for a variety of reasons, including the following:
|
|
·
|
insufficient funds to continue our clinical trials;
|
|
|
·
|
changes in the FDA requirements for approval, including requirements for testing efficacy and safety;
|
|
|
·
|
delay in obtaining or failure to obtain FDA or other regulatory authority approval of a clinical trial protocol;
|
|
|
·
|
patients not enrolling in clinical trials at the rate we expect;
|
|
|
·
|
delays in reaching agreement on acceptable clinical trial agreement terms with prospective sites;
|
|
|
·
|
delays in obtaining institutional review board approval to conduct a study at a prospective site;
|
|
|
·
|
third party clinical investigators not performing our clinical trials on our anticipated schedule or performance is not consistent with the clinical trial protocol and good clinical practices, or the third party organizations not performing data collection and analysis in a timely or accurate manner; and
|
|
|
·
|
changes in governmental regulations or administrative actions.
|
We do not know whether future clinical trials
will demonstrate safety and efficacy sufficiently to result in additional FDA approvals. While a number of physicians have conducted
clinical studies assessing the safety and efficacy of Microcyn® for various indications, the data from these studies are not
sufficient to support approval of Microcyn® as a drug in the United States.
Clinical trials involve a lengthy and
expensive process with an uncertain outcome, and results of earlier studies and trials may not be predictive of future trial results.
The results of preclinical studies and early
clinical trials of new drugs do not necessarily predict the results of later-stage clinical trials. The design of our clinical
trials is based on many assumptions about the expected effects of our product candidates, and if those assumptions are incorrect,
the trials may not produce statistically significant results. Preliminary results may not be confirmed upon full analysis of the
detailed results of an early clinical trial. Product candidates in later stages of clinical trials may fail to show safety and
efficacy sufficient to support intended use claims despite having progressed through initial clinical testing. The data collected
from clinical trials of our product candidates may not be sufficient to obtain regulatory approval in the United States or elsewhere.
Because of the uncertainties associated with drug development and regulatory approval, we cannot determine if or when we will have
an approved product for commercialization or achieve sales or profits.
If we fail to obtain, or experience significant
delays in obtaining, additional
regulatory clearances or approvals to market our current or future products, we may
be unable to commercialize these products.
The developing, testing, manufacturing, marketing
and selling of medical technology products is subject to extensive regulation by numerous governmental authorities in the United
States and other countries. The process of obtaining regulatory clearance and approval of medical technology products is costly
and time consuming. Even though their underlying product formulations may be the same or similar, our products are subject to different
regulations and approval processes depending upon their intended use.
To obtain regulatory approval of our products
as drugs in the United States, we must first show that our products are safe and effective for target indications through preclinical
studies consisting of laboratory and animal testing and clinical trials consisting of human testing. The FDA generally clears marketing
of a medical device through the 510(k) pre-market clearance process if it is demonstrated the new product has the same intended
use and the same or similar technological characteristics as another legally marketed Class II device, such as a device already
cleared by the FDA through the 510(k) premarket notification process, and otherwise meets the FDA’s requirements. Product
modifications, including labeling the product for a new intended use, may require the submission of a new 510(k) clearance and
FDA approval before the modified product can be marketed.
The outcomes of clinical trials are inherently
uncertain. In addition, we do not know whether the necessary approvals or clearances will be granted or delayed for future products.
The FDA could request additional information, changes to product formulation(s) or clinical testing that could adversely affect
the time to market and sale of products as drugs. If we do not obtain the requisite regulatory clearances and approvals, we will
be unable to commercialize our products as drugs or devices and may never recover any of the substantial costs we have invested
in the development of Microcyn®.
Distribution of our products outside the United
States is subject to extensive government regulation. These regulations, including the requirements for approvals or clearance
to market; the time required for regulatory review and the sanctions imposed for violations, vary from country to country. We do
not know whether we will obtain regulatory approvals in such countries or that we will not be required to incur significant costs
in obtaining or maintaining these regulatory approvals. In addition, the export by us of certain of our products that have not
yet been cleared for domestic commercial distribution may be subject to FDA export restrictions. Failure to obtain necessary regulatory
approvals, the restriction, suspension or revocation of existing approvals or any other failure to comply with regulatory requirements
would have a material adverse effect on our future business, financial condition, and results of operations.
If our products do not gain market acceptance,
our business will suffer because we might not be able to fund future operations.
A number of factors may affect the market acceptance
of our products or any other products we develop or acquire, including, among others:
|
|
·
|
the price of our products relative to other products for the same or similar treatments;
|
|
|
·
|
the perception by patients, physicians and other members of the healthcare community of the effectiveness and safety of our products for their indicated applications and treatments;
|
|
|
·
|
changes in practice guidelines and the standard of care for the targeted indication;
|
|
|
·
|
our ability to fund our sales and marketing efforts; and
|
|
|
·
|
the effectiveness of our sales and marketing efforts or our partners’ sales and marketing efforts.
|
Our ability to effectively promote and sell
any approved products will also depend on pricing and cost-effectiveness, including our ability to produce a product at a competitive
price and our ability to obtain sufficient third-party coverage or reimbursement, if any. In addition, our efforts to educate the
medical community on the benefits of our product candidates may require significant resources, may be constrained by FDA rules
and policies on product promotion, and may never be successful. If our products do not gain market acceptance, we may not be able
to fund future operations, including developing, testing and obtaining regulatory approval for new product candidates and expanding
our sales and marketing efforts for our approved products, which would cause our business to suffer.
If our competitors develop products similar
to Microcyn
®
, we may need to modify or
alter our business strategy, which may delay the achievement
of our goals.
Competitors have and may continue to develop
products with similar characteristics to Microcyn®. Such similar products marketed by larger competitors can hinder our efforts
to penetrate the market. As a result, we may be forced to modify or alter our business and regulatory strategy and sales and marketing
plans, as a response to changes in the market, competition and technology limitations, among others. Such modifications may pose
additional delays in achieving our goals.
We depend on third parties and intend
to continue to license or collaborate with third parties in various potential markets,
and events involving these
strategic partners or any future collaboration could
delay or prevent us from developing or commercializing products.
Our business strategy and our short- and long-term
operating results depend in part on our ability to execute on existing strategic collaborations and to license or partner with
new strategic partners. We believe collaborations allow us to leverage our resources and technologies and to access markets that
are compatible with our own core areas of expertise while avoiding the cost of establishing or maintaining a direct sales force
in each market. We may incur significant costs in the use of third parties to identify and assist in establishing relationships
with potential collaborators. We currently have a small direct sales force, which sells our products in the tissue care, dermatology,
and women’s health markets, and we intend to slowly expand the geographical coverage of our direct sales force.
To penetrate our target markets, we may need
to enter into additional collaborative agreements to assist in the development and commercialization of products. For example,
depending upon our analysis of the time and expense involved in obtaining FDA approval to sell a product to treat open wounds,
we may choose to license our technology to a third party as opposed to pursuing commercialization ourselves. Establishing strategic
collaborations is difficult and time-consuming. Potential collaborators may reject collaborations based upon their assessment of
our financial, regulatory or intellectual property position and our internal capabilities. Our discussions with potential collaborators
may not lead to the establishment of new collaborations on favorable terms and may have the potential to provide collaborators
with access to our key intellectual property filings and next generation formations. We have limited control over the amount and
timing of resources that our current collaborators or any future collaborators devote to our collaborations or potential products.
These collaborators may breach or terminate their agreements with us or otherwise fail to conduct their collaborative activities
successfully and in a timely manner. Further, our collaborators may not develop or commercialize products that arise out of our
collaborative arrangements or devote sufficient resources to the development, manufacture, marketing or sale of these products.
By entering into collaboration, we may preclude opportunities to collaborate with other third parties who do not wish to associate
with our existing third party strategic partners. Moreover, in the event of termination of a collaboration agreement, termination
negotiations may result in less favorable terms.
Our dependence on a commission-based
sales force and distributors for sales could limit or prevent us from selling our
products in certain markets.
We currently depend on a commission-based sales
force and distributors to sell Microcyn® in the United States, Europe and other countries, and intend to continue to sell our
products primarily through a commission-based sales force and distributors in Europe and the United States for the foreseeable
future. If we are unable to expand our direct sales force, we will continue to rely on a commission-based sales force and distributors
to sell Microcyn®. Our existing commission-based sales force and distribution agreements are generally short-term in duration,
and we may need to pursue alternate partners if the other parties to these agreements terminate or elect not to renew their agreements.
If we are unable to retain our current commission-based sales force and distributors for any reason, we must replace them with
alternate salespeople and distributors experienced in supplying the tissue care market, which could be time-consuming and divert
management’s attention from other operational matters. In addition, we will need to attract additional distributors to expand
the geographic areas in which we sell Microcyn®. Distributors may not commit the necessary resources to market and sell our
products to the level of our expectations, which could harm our ability to generate revenues. In addition, some of our distributors
may also sell products that compete with ours. In some countries, regulatory licenses must be held by residents of the country.
For example, the regulatory approval for one of our products in India is owned and held by our Indian distributor. If the licenses
are not in our name or under our control, we might not have the power to ensure their ongoing effectiveness and use by us. If current
or future distributors do not perform adequately, or we are unable to locate distributors in particular geographic areas, we may
not realize long-term revenue growth in certain markets.
If we fail to comply with ongoing regulatory
requirements, or if we experience
unanticipated problems with our products, these products could be subject to
restrictions or withdrawal from the market.
Regulatory approvals or clearances that we
currently have and that we may receive in the future are subject to limitations on the indicated uses for which the products may
be marketed, and any future approvals could contain requirements for potentially costly post-marketing follow-up studies. If the
FDA determines that our promotional materials or activities constitute promotion of an unapproved use or we otherwise fail to comply
with FDA regulations, we may be subject to regulatory enforcement actions, including warning letters, injunctions, seizures, civil
fines or criminal penalties. In addition, the manufacturing, labeling, packaging, adverse event reporting, storing, advertising,
promoting, distributing and record-keeping for approved products are subject to extensive regulation. We are subject to continued
supervision by European regulatory agencies relating to our CE markings and are required to report any serious adverse incidents
to the appropriate authorities. Our manufacturing facilities, processes and specifications are subject to periodic inspection by
the FDA, Mexican and other regulatory authorities and from time to time, we may receive notices of deficiencies from these agencies
as a result of such inspections. Our failure to continue to meet regulatory standards or to remedy any deficiencies could result
in restrictions being imposed on our products or manufacturing processes, fines, suspension or loss of regulatory approvals or
clearances, product recalls, termination of distribution, product seizures or the need to invest substantial resources to comply
with various existing and new requirements. In the more egregious cases, criminal sanctions, civil penalties, disgorgement of profits
or closure of our manufacturing facilities are possible. The subsequent discovery of previously unknown problems with Microcyn®,
including adverse events of unanticipated severity or frequency, may result in restrictions on the marketing of our products, and
could include voluntary or mandatory recall or withdrawal of products from the market.
New government regulations may be enacted and
changes in FDA policies and regulations and, their interpretation and enforcement, could prevent or delay regulatory approval of
our products. We cannot predict the likelihood, nature or extent of adverse government regulation that may arise from future legislation
or administrative action, either in the United States or abroad. Therefore, we do not know whether we will be able to continue
to comply with any regulations or that the costs of such compliance will not have a material adverse effect on our future business,
financial condition, and results of operations. If we are not able to maintain regulatory compliance, we will not be permitted
to market our products and our business would suffer.
We may experience difficulties in manufacturing
Microcyn
®
, which could prevent us from
commercializing one or more of our products.
The machines used to manufacture our Microcyn®-based
products are complex, use complicated software and must be monitored by highly trained engineers. Slight deviations anywhere in
our manufacturing process, including quality control, labeling and packaging, could lead to a failure to meet the specifications
required by the FDA, the Environmental Protection Agency, European notified bodies, Mexican regulatory agencies and other foreign
regulatory bodies, which may result in lot failures or product recalls. If we are unable to obtain quality internal and external
components, mechanical and electrical parts, if our software contains defects or is corrupted, or if we are unable to attract and
retain qualified technicians to manufacture our products, our manufacturing output of Microcyn®, or any other product candidate
based on our platform that we may develop, could fail to meet required standards, our regulatory approvals could be delayed, denied
or revoked, and commercialization of one or more of our Microcyn®-based products may be delayed or foregone. Manufacturing
processes that are used to produce the smaller quantities of Microcyn® needed for clinical tests and current commercial sales
may not be successfully scaled up to allow production of significant commercial quantities. Any failure to manufacture our products
to required standards on a commercial scale could result in reduced revenues, delays in generating revenue and increased costs.
Our competitive position depends on our
ability to protect our intellectual property
and our proprietary technologies.
Our ability to compete and to achieve and maintain
profitability depends on our ability to protect our intellectual property and proprietary technologies. We currently rely on a
combination of patents, patent applications, trademarks, trade secret laws, confidentiality agreements, license agreements and
invention assignment agreements to protect our intellectual property rights. We also rely upon unpatented know-how and continuing
technological innovation to develop and maintain our competitive position. These measures may not be adequate to safeguard our
Microcyn® Technology. If we do not protect our rights adequately, third parties could use our technology, and our ability to
compete in the market would be reduced.
We also have agreed to certain prohibitions
on our intellectual property. Pursuant to the License and Supply Agreement we entered into with Pulmatrix, Inc., we agreed to exclusively
license certain of our proprietary technology to Pulmatrix (formerly, Ruthigen, Inc.) to enable Pulmatrix’ research and development
and commercialization of RUT58-60, and any improvements to it, in the United States, Canada, European Union and Japan for certain
invasive procedures in human treatment as defined in the License and Supply Agreement. Under the terms of the agreement, we are
also prohibited from using the licensed proprietary technology to sell products that compete with Pulmatrix’ products within
the defined territory.
Although we have filed several U.S. and
foreign patent applications related to our Microcyn®-based products, the manufacturing technology for making the products,
and their uses, only 10 U.S. patents have been issued from these applications to date.
Our pending patent applications and any patent
applications we may file in the future may not result in issued patents, and we do not know whether any of our in-licensed patents
or any additional patents that might ultimately be issued by the U.S. Patent and Trademark Office or foreign regulatory body
will protect our Microcyn® Technology. Any claims that are issued may not be sufficiently broad to prevent third parties from
producing competing substitutes and may be infringed, designed around, or invalidated by third parties. Even issued patents may
later be found to be invalid, or may be modified or revoked in proceedings instituted by third parties before various patent offices
or in courts. For example, our European patent that was initially issued on May 30, 2007 was revoked by the Opposition Division
of the European Patent Office in December 2009 following opposition proceedings instituted by a competitor.
The degree of future protection for our proprietary
rights is more uncertain in part because legal means afford only limited protection and may not adequately protect our rights,
and we will not be able to ensure that:
|
|
·
|
we were the first to invent the inventions described in patent applications;
|
|
|
·
|
we were the first to file patent applications for inventions;
|
|
|
·
|
others will not independently develop similar or alternative technologies or duplicate our products without infringing our intellectual property rights;
|
|
|
·
|
any patents licensed or issued to us will provide us with any competitive advantages;
|
|
|
·
|
we will develop proprietary technologies that are patentable; or
|
|
|
·
|
the patents of others will not have an adverse effect on our ability to do business.
|
The policies we use to protect our trade secrets
may not be effective in preventing misappropriation of our trade secrets by others. In addition, confidentiality and invention
assignment agreements executed by our employees, consultants and advisors may not be enforceable or may not provide meaningful
protection for our trade secrets or other proprietary information in the event of unauthorized use or disclosures.
We operate in the State of California. The
laws of California prevent us from imposing a delay before an employee who may have access to trade secret and propriety know-how
can commence employment with a competing company. Although we may be able to pursue legal action against competitive companies
improperly using our proprietary information, we may not be aware of any use of our trade secrets and proprietary know-how until
after significant damages has been done to our Company.
We cannot be certain that the steps we have
taken will prevent the misappropriation and use of our intellectual property in the United States, or in foreign countries where
the laws may not protect our proprietary rights as fully as in the United States.
We may face intellectual property infringement
claims that could be time-consuming,
costly to defend and could result in our loss of significant rights and, in
the case
of patent infringement claims, the assessment of treble damages.
On occasion, we may receive notices of claims
of infringement, misappropriation or misuse of other parties’ proprietary rights. We may have disputes regarding intellectual
property rights with the parties that have licensed those rights to us. We may also initiate claims to defend our intellectual
property. Intellectual property litigation, regardless of its outcome, is expensive and time-consuming, and could divert management’s
attention from our business and have a material negative effect on our business, operating results or financial condition. In addition,
the outcome of such litigation may be unpredictable. If there is a successful claim of infringement against us, we may be required
to pay substantial damages, including treble damages if we were to be found to have willfully infringed a third party’s patent,
to the party claiming infringement, develop non-infringing technology, stop selling our products or using technology that contains
the allegedly infringing intellectual property or enter into royalty or license agreements that may not be available on acceptable
or commercially practical terms, if at all. Our failure to develop non-infringing technologies or license the proprietary rights
on a timely basis could harm our business. In addition, modifying our products to exclude infringing technologies could require
us to seek re-approval or clearance from various regulatory bodies for our products, which would be costly and time consuming.
Also, we may be unaware of pending patent applications that relate to our technology. Parties making infringement claims on future
issued patents may be able to obtain an injunction that would prevent us from selling our products or using technology that contains
the allegedly infringing intellectual property, which could harm our business.
Our ability to generate revenue will
be diminished if we are unable to obtain
acceptable prices or an adequate level of reimbursement from third-party
payors of
health care costs.
The continuing efforts of governmental and
other third-party payors, including managed care organizations such as health maintenance organizations, or HMOs, to contain or
reduce costs of health care may affect our future revenue and profitability, and the future revenue and profitability of our potential
customers, suppliers and collaborative or license partners and the availability of capital. For example, in certain foreign markets,
pricing or profitability of prescription pharmaceuticals is subject to government control. In the United States, governmental and
private payors have limited the growth of health care costs through price regulation or controls, competitive pricing programs
and drug rebate programs. Our ability to commercialize our products successfully will depend in part on the extent to which appropriate
coverage and reimbursement levels for the cost of our Microcyn® products and related treatment are obtained from governmental
authorities, private health insurers and other organizations, such as HMOs.
There is significant uncertainty concerning
third-party coverage and reimbursement of newly approved medical products and drugs. Third-party payors are increasingly challenging
the prices charged for medical products and services. Also, the trend toward managed healthcare in the United States and the concurrent
growth of organizations such as HMOs, as well as the recently enacted “Affordable Care Act,” may result in lower prices
for or rejection of our products. The cost containment measures that health care payors and providers are instituting and the effect
of any healthcare reform could materially and adversely affect our ability to generate revenues.
In both the United States and some foreign
jurisdictions, there have been a number of legislative and regulatory proposals to change the health care system in ways that could
affect our ability to sell our products profitably. In the United States, the Medicare Prescription Drug, Improvement, and Modernization
Act of 2003, also called the Medicare Modernization Act, or MMA, changed the way Medicare covers and pays for pharmaceutical products.
The legislation expanded Medicare coverage for drug purchases by the elderly and introduced a new reimbursement methodology based
on average sales prices for physician-administered drugs. In addition, this legislation provided authority for limiting the number
of drugs that will be covered in any therapeutic class. As a result of this legislation and the expansion of federal coverage of
drug products, we expect that there will be additional pressure to contain and reduce costs. These cost reduction initiatives and
other provisions of this legislation could decrease the coverage and price that we receive for any approved products and could
seriously harm our business. While the MMA applies only to drug benefits for Medicare beneficiaries, private payors often follow
Medicare coverage policies and payment limitations in setting their own reimbursement rates, and therefore any reduction in reimbursement
that results from the MMA may result in a similar reduction in payments from private payors.
In March 2010, the Patient Protection and Affordable
Care Act, as amended by the Health Care and Education Affordability Reconciliation Act, or collectively, the PPACA, became law
in the United States. The goal of PPACA is to reduce the cost of health care and substantially change the way health care is financed
by both governmental and private insurers. While we cannot predict what impact on federal reimbursement policies this legislation
will have in general or on our business specifically, the PPACA may result in downward pressure on pharmaceutical reimbursement,
which could negatively affect market acceptance of our Microcyn® products.
We expect to experience pricing pressures in
connection with the sale of our Microcyn® products, due to the trend toward managed health care, the increasing influence of
health maintenance organizations and additional legislative proposals. If we fail to successfully secure and maintain reimbursement
coverage for our products or are significantly delayed in doing so, we will have difficulty achieving market acceptance of our
products and our business will be harmed.
We could be required to indemnify third
parties for alleged intellectual property infringement, which could
cause us to incur significant costs.
Some of our distribution agreements contain
commitments to indemnify our distributors against liability arising from infringement of third party intellectual property such
as patents. We may be required to indemnify our customers for claims made against them or contribute to license fees they are required
to pay. If we are forced to indemnify for claims or to pay license fees, our business and financial condition could be substantially
harmed.
A significant part of our business is
conducted outside of the United States,
exposing us to additional risks that may not exist in the United States,
which in
turn could cause our business and operating results to suffer.
We have material international operations in
Mexico and Europe. During the year ended March 31, 2016 and 2015, approximately 67% and 73% of our total product related revenue
(including product license fees and royalties), respectively, were generated from sales outside of the United States. Our business
is highly regulated for the use, marketing and manufacturing of our Microcyn®-based products both domestically and internationally.
Our international operations are subject to risks, including:
|
|
·
|
local political or economic instability;
|
|
|
·
|
changes in governmental regulation;
|
|
|
·
|
changes in import/export duties;
|
|
|
·
|
lack of experience in foreign markets;
|
|
|
·
|
difficulties and costs of staffing and managing operations in certain foreign countries;
|
|
|
·
|
work stoppages or other changes in labor conditions;
|
|
|
·
|
difficulties in collecting accounts receivables on a timely basis or at all; and
|
|
|
·
|
adverse tax consequences or overlapping tax structures.
|
We plan to continue to market and sell our
products internationally to respond to customer requirements and market opportunities. We currently have manufacturing facilities
in Mexico and the United States. Establishing operations in any foreign country or region presents risks such as those described
above as well as risks specific to the particular country or region. In addition, until a payment history is established over time
with customers in a new geographic area or region, the likelihood of collecting receivables generated by such operations could
be less than our expectations. As a result, there is a greater risk that the reserves set with respect to the collection of such
receivables may be inadequate. If our operations in any foreign country are unsuccessful, we could incur significant losses and
we may not achieve profitability.
In addition, changes in policies or laws of
the United States or foreign governments resulting in, among other things, changes in regulations and the approval process, higher
taxation, currency conversion limitations, restrictions on fund transfers or the expropriation of private enterprises, could reduce
the anticipated benefits of our international expansion. If we fail to realize the anticipated revenue growth of our future international
operations, our business and operating results could suffer.
Our sales in international markets subject
us to foreign currency exchange and other
risks and costs which could harm our business.
A substantial portion of our revenues are derived
from outside the United States; primarily from Mexico and Europe. We anticipate that revenues from international customers will
continue to represent a substantial portion of our revenues for the foreseeable future. Because we generate revenues in foreign
currencies, we are subject to the effects of exchange rate fluctuations. The functional currency of our Mexican subsidiary is the
Mexican Peso and the functional currency of our Netherlands subsidiary is the Euro. For the preparation of our consolidated financial
statements, the financial results of our foreign subsidiaries are translated into U.S. dollars using average exchange rates
during the applicable period. If the U.S. dollar appreciates against the Mexican Peso or the Euro, as applicable, the revenues
we recognize from sales by our subsidiaries will be adversely impacted. Foreign exchange gains or losses as a result of exchange
rate fluctuations in any given period could harm our operating results and negatively impact our revenues. Additionally, if the
effective price of our products were to increase as a result of fluctuations in foreign currency exchange rates, demand for our
products could decline and adversely affect our results of operations and financial condition.
We rely on a number of key customers
who may not consistently purchase our products in the future and if we lose any one of these customers, our revenues may decline.
Although we have a significant number of customers
in each of the geographic markets that we operate in, we rely on certain key customers for a significant portion of our revenues.
We rely on certain key customers for a significant portion of our revenues. At March 31, 2016, one customer represented 40%, one
customer represented 15%, one customer represented 14% and two customers each represented 12% of net revenues. For the three months
ended June 30, 2016, one customer represented 29% of net revenue. For the three months ended June 30, 2015, one customer represented
53% of net revenue. In the future, a small number of customers may continue to represent a significant portion of our total revenues
in any given period. These customers may not consistently purchase our products at a particular rate over any subsequent period.
The loss of any of these customers could adversely affect our revenues.
Negative economic conditions increase
the risk that we could suffer unrecoverable losses on our customers’ accounts receivable which would adversely affect our
financial results.
We grant credit to our business customers,
which are primarily located in Mexico, Europe and the United States. Collateral is generally not required for trade receivables.
We maintain allowances for potential credit losses. At March 31, 2016, one customer represented 33% of the net accounts receivable
balance. At March 31, 2015, one customer represented 56%, and one customer represented 14% of the net accounts receivable balance.
At June 30, 2016, one customer represented 41%, and three customers each represented 11% of the net accounts receivable balance.
At March 31, 2016, one customer represented 33% of the net accounts receivable balance. While we believe we have a varied customer
base and have experienced strong collections in the past, if current economic conditions disproportionately impact any one of our
key customers, including reductions in their purchasing commitments to us or their ability to pay their obligations, it could have
a material adverse effect on our revenues and liquidity. We have not purchased insurance on our accounts receivable balances.
The loss of key members of our senior
management team, any of our directors, or our highly skilled scientists, technicians and salespeople could
adversely
affect our business.
Our success depends largely on the skills,
experience and performance of key members of our executive management team, including Jim Schutz, our Chief Executive Officer,
Robert Miller, our Chief Financial Officer, Robert Northey, our Executive Vice President of Research and Development, and Jeffrey
Day, head of our IntraDerm™ Pharmaceuticals division. The efforts of these people will be critical to us as we continue to
develop our products and attempt to commercialize products in the tissue and dermatology markets. If we were to lose one or more
of these individuals, we might experience difficulties in competing effectively, developing our technologies and implementing our
business strategies.
Our research and development programs depend
on our ability to attract and retain highly skilled scientists and technicians. We may not be able to attract or retain qualified
scientists and technicians in the future due to the intense competition for qualified personnel among medical technology businesses,
particularly in the San Francisco Bay Area. We also face competition from universities and public and private research institutions
in recruiting and retaining highly qualified personnel. In addition, our success depends on our ability to attract and retain salespeople
with extensive experience in dermatology or in the markets we seek, and who have close relationships with the medical community,
including physicians and other medical staff. We may have difficulties locating, recruiting or retaining qualified salespeople,
which could cause a delay or decline in the rate of adoption of our products. If we are not able to attract and retain the necessary
personnel to accomplish our business objectives, we may experience constraints that will adversely affect our ability to support
our research, development and sales programs.
The dermatology, tissue and animal healthcare
industries are highly competitive and subject to rapid technological change. If our competitors are better able to develop and
market products that are
less expensive or more effective than any products that we may develop, our
commercial
opportunity will be reduced or eliminated.
Our success depends, in part, upon our ability
to stay at the forefront of technological change and maintain a competitive position. We compete with large healthcare, pharmaceutical
and biotechnology companies, along with smaller or early-stage companies that have collaborative arrangements with larger pharmaceutical
companies, academic institutions, government agencies and other public and private research organizations. Many of our competitors
have significantly greater financial resources and expertise in research and development, manufacturing, pre-clinical testing,
conducting clinical trials, obtaining regulatory approvals and marketing approved products than we do. Our competitors may:
|
|
·
|
develop and patent processes or products earlier than we will;
|
|
|
·
|
develop and commercialize products that are less expensive or more efficient than any products that we may develop;
|
|
|
·
|
obtain regulatory approvals for competing products more rapidly than we will; and
|
|
|
·
|
improve upon existing technological approaches or develop new or different approaches that render our technology or products obsolete or non-competitive.
|
As a result, we may not be able to successfully
commercialize any future products.
The success of our research and development
efforts may depend on our ability to find
suitable collaborators to fully exploit our capabilities. If we are unable
to
establish collaborations or if these future collaborations are unsuccessful, our
research and development
efforts may be unsuccessful, which could adversely affect
our results of operations and financial condition.
An important element of our business strategy
is to enter into collaborative or license arrangements under which we license our Microcyn® Technology to other parties for
development and commercialization. We expect to seek collaborators for our drug candidates and for a number of our potential products
because of the expense, effort and expertise required to conduct additional clinical trials and further develop those potential
product candidates. Because collaboration arrangements are complex to negotiate, we may not be successful in our attempts to establish
these arrangements. If we need third party assistance in identifying and negotiating one or more acceptable arrangements, it might
be costly. Also, we may not have products that are desirable to other parties, or we may be unwilling to license a potential product
because the party interested in it is a competitor. The terms of any arrangements that we establish may not be favorable to us.
Alternatively, potential collaborators may decide against entering into an agreement with us because of our financial, regulatory
or intellectual property position or for scientific, commercial or other reasons. If we are not able to establish collaborative
agreements, we may not be able to develop and commercialize new products, which would adversely affect our business and our revenues.
In order for any of these collaboration or
license arrangements to be successful, we must first identify potential collaborators or licensees whose capabilities complement
and integrate well with ours. We may rely on these arrangements for not only financial resources, but also for expertise or economies
of scale that we expect to need in the future relating to clinical trials, manufacturing, sales and marketing, and for licenses
to technology rights. However, it is likely that we will not be able to control the amount and timing or resources that our collaborators
or licensees devote to our programs or potential products. If our collaborators or licensees prove difficult to work with, are
less skilled than we originally expected, or do not devote adequate resources to the program, the relationship will not be successful.
If a business combination involving a collaborator or licensee and a third party were to occur, the effect could be to diminish,
terminate or cause delays in development of a potential product.
If we are unable to comply with broad
and complex federal and state fraud and abuse
laws, including state and federal anti-kickback laws, we could face
substantial
penalties and our products could be excluded from government healthcare programs.
We are subject to various federal and state
laws pertaining to healthcare fraud and abuse, which include, among other things, “anti-kickback” laws that prohibit
payments to induce the referral of products and services, and “false claims” statutes that prohibit the fraudulent
billing of federal healthcare programs. Our operations are subject to the Federal Anti-Kickback Statute, a criminal statute that,
subject to certain statutory exceptions, prohibits any person from knowingly and willfully offering, paying, soliciting or receiving
remuneration, directly or indirectly, to induce or reward a person either (i) for referring an individual for the furnishing
of items or services for which payment may be made in whole or in part by a government healthcare program such as Medicare or Medicaid,
or (ii) for purchasing, leasing, ordering or arranging for or recommending the purchasing, leasing or ordering of an item
or service for which payment may be made under a government healthcare program. Because of the breadth of the Federal Anti-Kickback
Statute, the Office of Inspector General of the U.S. Department of Health and Human Services, was authorized to adopt regulations
setting forth additional exceptions to the prohibitions of the statute commonly known as “safe harbors.” If all of
the elements of an applicable safe harbor are fully satisfied, an arrangement will not be subject to prosecution under the Federal
Anti-Kickback Statute.
In addition, if there is a change in law, regulation
or administrative or judicial interpretations of these laws, we may have to change our business practices or our existing business
practices could be challenged as unlawful, which could have a negative effect on our business, financial condition and results
of operations.
Healthcare fraud and abuse laws are complex,
and even minor, inadvertent irregularities can potentially give rise to claims that a statute or regulation has been violated.
The frequency of suits to enforce these laws has increased significantly in recent years and has increased the risk that a healthcare
company will have to defend a false claim action, pay fines or be excluded from the Medicare, Medicaid or other federal and state
healthcare programs as a result of an investigation arising out of such action. We cannot assure you that we will not become subject
to such litigation. Any violations of these laws, or any action against us for violation of these laws, even if we successfully
defend against it, could harm our reputation, be costly to defend and divert management’s attention from other aspects of
our business. Similarly, if the physicians or other providers or entities with which we do business are found to have violated
abuse laws, they may be subject to sanctions, which could also have a negative impact on us.
Our efforts to discover and develop potential
products may not lead to the discovery,
development, commercialization or marketing of actual drug products.
We are currently engaged in a number of different
approaches to discover and develop new product applications and product candidates. Discovery and development of potential drug
candidates are expensive and time-consuming, and we do not know if our efforts will lead to discovery of any drug candidates that
can be successfully developed and marketed. If our efforts do not lead to the discovery of a suitable drug candidate, we may be
unable to grow our clinical pipeline or we may be unable to enter into agreements with collaborators who are willing to develop
our drug candidates.
We may not be able to maintain sufficient
product liability insurance to cover claims
against us.
Product liability insurance for the healthcare
industry is generally expensive to the extent it is available at all. We may not be able to maintain such insurance on acceptable
terms or be able to secure increased coverage if the commercialization of our products progresses, nor can we be sure that existing
or future claims against us will be covered by our product liability insurance. Moreover, the existing coverage of our insurance
policy or any rights of indemnification and contribution that we may have may not be sufficient to offset existing or future claims.
A successful claim against us with respect to uninsured liabilities or in excess of insurance coverage and not subject to any indemnification
or contribution could have a material adverse effect on our future business, financial condition, and results of operations.
If any of our
third-party contractors fail to perform their responsibilities to comply with FDA rules and regulations, the manufacture, marketing
and sales of our products could be delayed, which could decrease our revenues.
Supplying the market
with our Microcyn® Technology products requires us to manage relationships with an increasing number of collaborative partners,
suppliers and third-party contractors. As a result, our success depends partially on the success of these third parties in performing
their responsibilities to comply with FDA rules and regulations. Although we pre-qualify our contractors and we believe that they
are fully capable of performing their contractual obligations, we cannot directly control the adequacy and timeliness of the resources
and expertise that they apply to these activities. For example, we and our suppliers are required to comply with the FDA’s
quality system regulations, which cover the methods and documentation of the design, testing, production, control, quality assurance,
labeling, packaging, storage and shipping of our products. The FDA enforces the quality system regulation through inspections.
If any of our partners
or contractors fail to perform their obligations in an adequate and timely manner, or fail to comply with the FDA’s rules
and regulations, including failure to comply with quality systems regulations or a corrective action submitted to the FDA after
notification by the FDA of a deficiency is deemed insufficient, then the manufacture, marketing and sales of our products could
be delayed. Our products could be detained or seized, the FDA could order a recall, or require our partner to replace or offer
refunds for our products. The FDA could also require our partner, and, depending on our agreement with our partner, us, to notify
health professionals and others that the products present unreasonable risks of substantial harm to the public health. If any of
these events occur, the manufacture, marketing and sales of our products could be delayed which could decrease our revenues.
If we fail to
comply with the FDA’s rules and regulations and are subject to an FDA recall as part of an FDA enforcement action, the associated
costs could have a material adverse effect on our business, financial position, results of operations and cash flows.
Our Company, our products,
the manufacturing facilities for our products, the distribution of our products, and our promotion and marketing materials are
subject to strict and continual review and periodic inspection by the FDA and other regulatory agencies for compliance with pre-approval
and post-approval regulatory requirements.
If we fail to comply
with the FDA’s rules and regulations, we could be subject to an enforcement action by the FDA. The FDA could undertake regulatory
actions, including seeking a consent decree, recalling or seizing our products, ordering a total or partial shutdown of production,
delaying future marketing clearances or approvals, and withdrawing or suspending certain of our current products from the market.
A product recall, restriction, or withdrawal could result in substantial and unexpected expenditures, destruction of product inventory,
and lost revenues due to the unavailability of one or more of our products for a period of time, which could reduce profitability
and cash flow. In addition, a product recall or withdrawal could divert significant management attention and financial resources.
If any of our products are subject to an FDA recall, we could incur significant costs and suffer economic losses. Production of
our products could be suspended and we could be required to establish inventory reserves to cover estimated inventory losses for
all work-in-process and finished goods related to products we, or our third-party contractors, manufacture. A recall of a material
amount of our products could have a significant, unfavorable impact on our future gross margins.
If our products
fail to comply with FDA and other governmental regulations, or our products are deemed defective, we may be required to recall
our products and we could suffer adverse public relations that could adversely impact our sales, operating results, and reputation
which would adversely affect our business operations.
We may be exposed
to product recalls, including voluntary recalls or withdrawals, and adverse public relations if our products are alleged to cause
injury or illness, or if we are alleged to have mislabeled or misbranded our products or otherwise violated governmental regulations.
Governmental authorities can also require product recalls or impose restrictions for product design, manufacturing, labeling, clearance,
or other issues. For the same reasons, we may also voluntarily elect to recall, restrict the use of a product or withdraw products
that we consider below our standards, whether for quality, packaging, appearance or otherwise, in order to protect our brand reputation.
Product recalls, product
liability claims, even if unmerited or unsuccessful, or any other events that cause consumers to no longer associate our brand
with high quality and safe products may also result in adverse publicity, hurt the value of our brand, harm our reputation among
our customers and other healthcare professionals who use or recommend the products, lead to a decline in consumer confidence in
and demand for our products, and lead to increased scrutiny by federal and state regulatory agencies of our operations, any of
which could have a material adverse effect on our brand, business, performance, prospects, value, results of operations and financial
condition.
Risks Related to Our Common Stock
The market price of our common stock
may be volatile, and the value of your investment could decline significantly.
The trading price for our common stock has
been, and we expect it to continue to be, volatile. The price at which our common stock trades depends upon a number of factors,
including our historical and anticipated operating results, our financial situation, announcements of new products by us or our
competitors, our ability or inability to raise the additional capital we may need and the terms on which we raise it, and general
market and economic conditions. Some of these factors are beyond our control. Broad market fluctuations may lower the market price
of our common stock and affect the volume of trading in our stock, regardless of our financial condition, results of operations,
business or prospects. It is impossible to assure you that the market price of our shares of common stock will not fall in the
future.
Our operating results may fluctuate,
which could cause our stock price to decrease.
Fluctuations in our operating results may lead
to fluctuations, including declines, in our share price. Our operating results and our share price may fluctuate from period to
period due to a variety of factors, including:
|
|
·
|
demand by physicians, other medical staff and patients for our Microcyn®-based products;
|
|
|
·
|
reimbursement decisions by third-party payors and announcements of those decisions;
|
|
|
·
|
clinical trial results published by others in our industry and publication of results in peer-reviewed journals or the presentation at medical conferences;
|
|
|
·
|
the inclusion or exclusion of our Microcyn®-based products in large clinical trials conducted by others;
|
|
|
·
|
actual and anticipated fluctuations in our quarterly financial and operating results;
|
|
|
·
|
developments or disputes concerning our intellectual property or other proprietary rights;
|
|
|
·
|
issues in manufacturing our product candidates or products;
|
|
|
·
|
new or less expensive products and services or new technology introduced or offered by our competitors or by us;
|
|
|
·
|
the development and commercialization of product enhancements;
|
|
|
·
|
changes in the regulatory environment;
|
|
|
·
|
delays in establishing our sales force or new strategic relationships;
|
|
|
·
|
costs associated with collaborations and new product candidates;
|
|
|
·
|
introduction of technological innovations or new commercial products by us or our competitors;
|
|
|
·
|
litigation or public concern about the safety of our product candidates or products;
|
|
|
·
|
changes in recommendations of securities analysts or lack of analyst coverage;
|
|
|
·
|
failure to meet analyst expectations regarding our operating results;
|
|
|
·
|
additions or departures of key personnel; and
|
|
|
·
|
general market conditions.
|
Variations in the timing of our future revenues
and expenses could also cause significant fluctuations in our operating results from period to period and may result in unanticipated
earning shortfalls or losses. In addition, The NASDAQ Capital Market, in general, and the market for life sciences companies, in
particular, have experienced significant price and volume fluctuations that have often been unrelated or disproportionate to the
operating performance of those companies.
Anti-takeover provisions in our certificate
of incorporation and bylaws and under Delaware law may make
it more difficult for stockholders to change our management
and may also make a
takeover difficult.
Our corporate documents and Delaware law contain
provisions that limit the ability of stockholders to change our management and may also enable our management to resist a takeover.
These provisions include:
|
|
·
|
the ability of our Board of Directors to issue and designate, without stockholder approval, the rights of up to 714,286 shares of convertible preferred stock, which rights could be senior to those of common stock;
|
|
|
·
|
limitations on persons authorized to call a special meeting of stockholders; and
|
|
|
·
|
advance notice procedures required for stockholders to make nominations of candidates for election as directors or to bring matters before meetings of stockholders.
|
We are subject to Section 203 of the Delaware
General Corporation Law, which, subject to certain exceptions, prohibits “business combinations” between a publicly-held
Delaware corporation and an “interested stockholder,” which is generally defined as a stockholder who became a beneficial
owner of 15% or more of a Delaware corporation’s voting stock for a three-year period following the date that such stockholder
became an interested stockholder.
These provisions might discourage, delay or
prevent a change of control in our management. These provisions could also discourage proxy contests and make it more difficult
for you and other stockholders to elect directors and cause us to take other corporate actions. In addition, the existence of these
provisions, together with Delaware law, might hinder or delay an attempted takeover other than through negotiations with our Board
of Directors.
We currently have significant “equity
overhang” which could adversely affect the market price of our common stock and impair our ability to raise additional capital
through the sale of equity securities in the future.
We currently have
significant “equity overhang.” The possibility that substantial amounts of our common stock may be issued to and then
sold by investors, or the perception that such issuances and sales could occur, often called “equity overhang,” could
adversely affect the market price of our common stock and could impair our ability to raise additional capital through the sale
of equity securities in the future. The consummation of the exercise of warrants for common stock would significantly increase
the number of issued and outstanding shares of our common stock.
Our stockholders may experience substantial
dilution in the value of their investment
if we issue additional shares of our capital stock or other securities
convertible into common stock.
Our Restated Certificate of Incorporation,
as amended, allows us to issue up to 12,000,000 shares of our common stock and to issue and designate, without stockholder approval,
the rights of up to 714,286 shares of preferred stock. In the event we issue additional shares of our capital stock, dilution to
our stockholders could result. In addition, if we issue and designate a class of convertible preferred stock, these securities
may provide for rights, preferences or privileges senior to those of holders of our common stock. Additionally, if we issue preferred
stock, it may convert into common stock at a ratio of 1:1 or greater because our Restated Certificate of Incorporation, as amended,
allows us to designate a conversion ratio without limitations.
Shares issuable upon the conversion of
warrants or the exercise of outstanding options may substantially increase the number of shares available for sale in the public
market and depress the price of our common stock.
As of June 30, 2016, we had outstanding warrants
exercisable for an aggregate of 1,468,000 shares of our common stock at a weighted average exercise price of approximately $10.00
per share. In addition, as of June 30, 2016, options to purchase an aggregate of 754,000 shares of our common stock were outstanding
at a weighted average exercise price of approximately $21.18 per share and a weighted average contractual term of 7.69 years. In
addition, 578,000 shares of our common stock were available on June 30, 2016 for future option grants under our Amended and Restated
2006 Stock Incentive Plan, and our 2011 Stock Incentive Plan. To the extent any of these warrants or options are exercised and
any additional options are granted and exercised, there will be further dilution to stockholders and investors. Until the options
and warrants expire, these holders will have an opportunity to profit from any increase in the market price of our common stock
without assuming the risks of ownership. Holders of options and warrants may convert or exercise these securities at a time when
we could obtain additional capital on terms more favorable than those provided by the options or warrants. The exercise of the
options and warrants will dilute the voting interest of the owners of presently outstanding shares by adding a substantial number
of additional shares of our common stock.
We have filed several registration statements
with the SEC, so that substantially all of the shares of our common stock which are issuable upon the exercise of outstanding warrants
and options may be sold in the public market. The sale of our common stock issued or issuable upon the exercise of the warrants
and options described above, or the perception that such sales could occur, may adversely affect the market price of our common
stock.
CAUTIONARY NOTE REGARDING FORWARD-LOOKING
STATEMENTS
This prospectus supplement, the accompanying
prospectus and the documents incorporated by reference in this prospectus supplement contain forward looking statements. When used
in this prospectus supplement, the words “anticipate,” “intend,” “estimate,” “plan,”
“project,” “continue,” “ongoing,” “potential,” “expect,” “predict,”
“believe,” “intend,” “may,” “can,” “will,” “should,” “could,”
“would,” “proposal,” and similar expressions are intended to identify forward-looking statements.
You should not place undue reliance on these
forward-looking statements. Our actual results could differ materially from those anticipated in the forward-looking statements
for many reasons, including the reasons described in our “Risk Factors” section. Although we believe the expectations
reflected in the forward-looking statements are reasonable, they relate only to events as of the date on which the statements
are made. These forward-looking statements speak only as of the date of this prospectus supplement. We expressly disclaim any
obligation or undertaking to update or revise any forward-looking statements contained herein to reflect any change in our expectations
with regard thereto or any change in events, conditions or circumstances on which any such statement is based, except as required
by law.
USE OF
PROCEEDS
We
received $5,444,000 in net proceeds from the sale of shares and warrants in this offering after deducting underwriting discounts
and commissions and estimated offering expenses payable by us. If a warrant holder exercises their warrants for cash, we will also
receive proceeds from such exercise at the time of such exercise. We cannot predict when or if the warrants will be exercised.
It is possible that the warrants may expire and may never be exercised, in which case we will not receive any additional proceeds.
We intend to use the net proceeds received from this offering to increase our direct sales force, to develop and launch new products
and for general working capital.
DILUTION
Our
net tangible book value as of June 30, 2016 was approximately $7,749,000, or approximately $1.84 per share. Net tangible book value
per share represents our total tangible assets less total liabilities, divided by the number of shares of common stock outstanding
as of June 30, 2016.
After
giving effect to the exercise of 5,379,525 warrants sold in the offering that remain outstanding at June 30, 2016 which would have
resulted in 1,075,905 shares of common stock being issued
at an exercise
price of $6.50 per share, our as adjusted net tangible book value as of June 30, 2016 would have been $14,743,000, or $2.79 per
share. This represents an immediate increase in net tangible book value of $0.94 per share to existing stockholders of our Company
and an immediate decrease in the net tangible book value of $3.71 per share to warrants exercised from this offering, as illustrated
in the following table:
|
Exercise price per warrant
|
|
$
|
6.50
|
|
|
Net tangible book value per share as of June 30, 2016
|
|
$
|
1.84
|
|
|
Increase in net tangible book value per share attributable to existing stockholders
|
|
$
|
0.95
|
|
|
Adjusted net tangible book value per share as of June 30, 2016, after giving effect to the exercise of 5,379,525 warrants
|
|
$
|
2.79
|
|
|
Decrease in net tangible book value per share to holders of warrants purchased in this offering
|
|
$
|
3.71
|
|
The
above discussion and tables do not include the following (as of June 30, 2016):
|
|
·
|
754,000
shares of common stock issuable upon exercise of outstanding stock options, at a weighted average exercise price of $21.18
per share, under our equity incentive plans;
|
|
|
·
|
578,000 additional shares of common stock reserved for future issuance under our equity incentive plans;
|
|
|
·
|
1,468,000 shares of common stock issuable upon exercise of outstanding warrants, with current exercise prices ranging from $5.00 per share to $630.00 per share.
|
PRICE RANGE OF OUR
COMMON STOCK
Market Information
Our common
stock is quoted on The NASDAQ Capital Market under the symbol “OCLS.” and has been trading since our initial
public offering on January 25, 2007. The following table sets forth the high and low sales prices for our common stock
for each quarter during the last two fiscal years and the current fiscal year as reported on Bloomberg. The prices have been
adjusted to reflect a 1-for-5 reverse split, effective June 24, 2016.
|
|
|
High
|
|
|
Low
|
|
|
For the Fiscal Year Ending March 31, 2017
|
|
|
|
|
|
|
|
|
|
Second Quarter ending September 30, 2016*
|
|
$
|
4.98
|
|
|
$
|
3.57
|
|
|
First Quarter ended June 30, 2016
|
|
$
|
6.65
|
|
|
$
|
3.62
|
|
|
For the Fiscal Year Ended March 31, 2016
|
|
|
|
|
|
|
|
|
|
Fourth Quarter ended March 31, 2016
|
|
$
|
7.15
|
|
|
$
|
4.00
|
|
|
Third Quarter ended December 31, 2015
|
|
$
|
7.00
|
|
|
$
|
5.40
|
|
|
Second Quarter ended September 30, 2015
|
|
$
|
9.65
|
|
|
$
|
5.00
|
|
|
First Quarter ended June 30, 2015
|
|
$
|
9.85
|
|
|
$
|
3.25
|
|
|
For the Fiscal Year Ended March 31, 2015
|
|
|
|
|
|
|
|
|
|
Fourth Quarter ended March 31, 2015
|
|
$
|
7.90
|
|
|
$
|
3.80
|
|
|
Third Quarter ended December 31, 2014
|
|
$
|
11.75
|
|
|
$
|
6.65
|
|
|
Second Quarter ended September 30, 2014
|
|
$
|
15.40
|
|
|
$
|
10.00
|
|
|
First Quarter ended June 30, 2014
|
|
$
|
21.08
|
|
|
$
|
14.25
|
|
* Through August
30, 2016
The warrants from
this offering began trading on The NASDAQ Capital Market on January 21, 2015 under the symbol “OCLSW.”
Holders
As of August 1, 2016, we had approximately
345 holders of record of our common stock. Holders of record include nominees who may hold shares on behalf of multiple owners.
DIVIDEND
POLICY
We have never declared
or paid any cash dividends on our common stock. We currently anticipate that we will retain all future earnings for the operation
of our business and we do not currently intend to pay any cash dividends on our common stock in the foreseeable future.
CAPITALIZATION
The
following table sets forth our capitalization as of June 30, 2016 on an actual basis and on a pro forma basis, based upon the exercise
of 5,379,525 warrants sold in the offering that remain outstanding at June 30, 2016 which would have resulted in 1,075,905 shares
of common stock being issued
at an exercise price of $6.50 per share.
Based on the exercise
price of $6.50 per share, we allocated the gross proceeds of $6,993,000 aggregate consideration to common stock. The pro forma
information below is for illustrative purposes and our capitalization following the exercise of all warrants may be adjusted. You
should read this table in conjunction with “Use of Proceeds” above as well as our “Management’s Discussion
and Analysis of Financial Condition and Results of Operations” and financial statements and the related notes appearing elsewhere
in this prospectus.
|
|
|
June 30, 2016
|
|
|
|
|
Actual
|
|
|
Pro Forma (1)
|
|
|
|
|
(in thousands)
(unaudited)
|
|
|
Total assets
|
|
$
|
11,320
|
|
|
$
|
18,313
|
|
|
Current portion of long-term debt
|
|
|
66
|
|
|
|
66
|
|
|
Stockholders’ equity:
|
|
|
|
|
|
|
|
|
|
Convertible preferred stock, $0.0001 par value; 714,286 shares authorized, none issued and outstanding actual
|
|
|
–
|
|
|
|
–
|
|
|
Common stock, $0.0001 par value; 12,000,000 shares authorized, 4,200,756 shares issued and outstanding actual; and 12,000,000 shares authorized, 5,276,661 shares issued and outstanding pro forma (1)
|
|
|
1
|
|
|
|
1
|
|
|
Additional paid-in capital
|
|
|
166,779
|
|
|
|
173,771
|
|
|
Accumulated deficit
|
|
|
(154,943
|
)
|
|
|
(154,943
|
)
|
|
Accumulated other comprehensive loss
|
|
|
(4,087
|
)
|
|
|
(4,087
|
)
|
|
Total stockholders’ equity
|
|
$
|
7,750
|
|
|
$
|
14,741
|
|
The above discussion
and table does not include the following (as of June 30, 2016):
|
|
·
|
754,000
shares of common stock issuable upon exercise of outstanding stock options, at a weighted average exercise price of $21.18 per share, under our equity incentive plans;
|
|
|
·
|
578,000 additional shares of common stock reserved for future issuance under our equity incentive plans;
|
|
|
·
|
1,468,000
shares of common stock issuable upon exercise of outstanding warrants, with current exercise prices ranging from $5.00
per share to $
630.00
per share.
|
Management’s
Discussion and Analysis of Financial Condition and Results of Operations
Critical Accounting Policies
The preparation of our consolidated financial
statements in conformity with accounting principles generally accepted in the United States requires management to exercise its
judgment. We exercise considerable judgment with respect to establishing sound accounting policies and in making estimates and
assumptions that affect the reported amounts of our assets and liabilities, our recognition of revenues and expenses, and disclosure
of commitments and contingencies at the date of the consolidated financial statements.
On an ongoing basis, we evaluate our estimates
and judgments. Areas in which we exercise significant judgment include, but are not necessarily limited to, our valuation of accounts
receivable, inventory, income taxes, equity transactions (compensatory and financing) and contingencies. We have also adopted certain
polices with respect to our recognition of revenue that we believe are consistent with the guidance provided under Securities and
Exchange Commission Staff Accounting Bulletin No. 104.
We base our estimates and judgments on a variety
of factors including our historical experience, knowledge of our business and industry, current and expected economic conditions,
the attributes of our products, the regulatory environment, and in certain cases, the results of outside appraisals. We periodically
re-evaluate our estimates and assumptions with respect to these judgments and modify our approach when circumstances indicate that
modifications are necessary.
While we believe that the factors we evaluate
provide us with a meaningful basis for establishing and applying sound accounting policies, we cannot guarantee that the results
will always be accurate. Since the determination of these estimates requires the exercise of judgment, actual results could differ
from such estimates.
A description of significant accounting policies
that require us to make estimates and assumptions in the preparation of our consolidated financial statements is as follows:
Long-Term Investments
Our long-term investments consisted of the
shares we owned in Ruthigen Inc., now Pulmatrix Inc., at March 31, 2015. We carry securities that do not have a readily determinable
fair value at cost. During the year ended March 31, 2015, we recorded an impairment loss in the amount of $4,650,000 which represents
the difference between cost and the amount we agreed to sell our shares of Ruthigen. During the year ended March 31, 2016, we sold
all our shares in Pulmatrix.
Stock-based Compensation
We account for share-based awards exchanged
for employee services based on the estimated fair value of the award on the grant date. We estimate the fair value of employee
stock awards using the Black-Scholes option pricing model. We amortize the fair value of employee stock options on a straight-line
basis over the requisite service period of the awards. Compensation expense includes the impact of an estimate for forfeitures
for all stock options.
We account for equity instruments issued to
non-employees based on the estimated fair value of the instrument on the measurement date. The measurement of stock-based compensation
is subject to periodic adjustment as the underlying equity instrument vests or becomes non-forfeitable. Non-employee stock-based
compensation charges are amortized over the vesting period or as earned.
Revenue Recognition and Accounts Receivable
We generate revenue from sales of our products
to a customer base including hospitals, medical centers, doctors, pharmacies, distributors and wholesalers. The Company sells products
directly to end users and to distributors. We also entered into agreements to license its technology and products.
We also provide regulatory compliance testing
and quality assurance services to medical device and pharmaceutical companies.
We record revenue when (i) persuasive
evidence of an arrangement exists, (ii) delivery has occurred, (iii) the fee is fixed or determinable, and (iv) collectability
of the sale is reasonably assured.
We require all product sales to be supported
by evidence of a sale transaction that clearly indicates the selling price to the customer, shipping terms and payment terms. Evidence
of an arrangement generally consists of a contract or purchase order approved by the customer. We have ongoing relationships with
certain customers from which we customarily accept orders by telephone in lieu of purchase orders.
We recognize revenue at the time we receive
confirmation that the goods were either tendered at their destination, when shipped “FOB destination,” or transferred
to a shipping agent, when shipped “FOB shipping point.” Delivery to the customer is deemed to have occurred when the
customer takes title to the product. Generally, title passes to the customer upon shipment, but could occur when the customer receives
the product based on the terms of the agreement with the customer.
The selling prices of all goods are fixed,
and agreed to with the customer, prior to shipment. Selling prices are generally based on established list prices. The right to
return product is customarily based on the terms of the agreement with the customer. We estimate and accrue for potential returns
and record this as a reduction of revenue in the same period the related revenue is recognized. Additionally, distribution fees
are paid to certain wholesale distributors based on contractually determined rates. We estimate and accrue the fee on shipment
to the respective wholesale distributors and recognize the fee as a reduction of revenue in the same period the related revenue
is recognized. We also offer cash discounts to certain customers, generally 2% of the sales price, as an incentive for prompt payment.
We account for cash discounts by reducing accounts receivable by the prompt pay discount amount and recognize the discount as a
reduction of revenue in the same period the related revenue is recognized. Additionally, we participate in certain rebate programs
which provide discounted prescriptions to qualified patients. We contract a third-party to administer the program. We estimate
and accrue for future rebates based on historical data for rebate redemption rates and the historical value of redemptions. Rebates
are recognized as a reduction of revenue in the same period the related revenue is recognized.
We evaluate the creditworthiness of new customers
and monitors the creditworthiness of its existing customers to determine whether an event or changes in their financial circumstances
would raise doubt as to the collectability of a sale at the time in which a sale is made. Payment terms on sales made in the United
States are generally 30 days and are extended up to 90 days for initial product launches, payment terms internationally generally
range from prepaid prior to shipment to 90 days.
In the event a sale is made to a customer under
circumstances in which collectability is not reasonably assured, we either require the customer to remit payment prior to shipment
or defer recognition of the revenue until payment is received. We maintain a reserve for amounts which may not be collectible due
to risk of credit losses.
Product license revenue is generated through
agreements with strategic partners for the commercialization of Microcyn® products. The terms of the agreements sometimes include
non-refundable upfront fees. We analyze multiple element arrangements to determine whether the elements can be separated. Analysis
is performed at the inception of the arrangement and as each product is delivered. If a product or service is not separable, the
combined deliverables are accounted for as a single unit of accounting and recognized over the performance obligation period.
When appropriate, we defer recognition of non-refundable
upfront fees. If we have continuing performance obligations then such up-front fees are deferred and recognized over the period
of continuing involvement.
We recognize royalty revenues from licensed
products upon the sale of the related products.
Revenue from consulting contracts is recognized
as services are provided. Revenue from testing contracts is recognized as tests are completed and a final report is sent to the
customer.
Inventory
Inventories are stated at the lower of cost,
cost being determined on a standard cost basis (which approximates actual cost on a first-in, first-out basis), or market. Due
to changing market conditions, estimated future requirements, age of the inventories on hand and production of new products, we
regularly review inventory quantities on hand and record a provision to write down excess and obsolete inventory to its estimated
market value.
Income Taxes
We are required to determine the aggregate
amount of income tax expense or loss based upon tax statutes in jurisdictions in which we conduct business. In making these estimates,
we adjust our results determined in accordance with generally accepted accounting principles for items that are treated differently
by the applicable taxing authorities. Deferred tax assets and liabilities resulting from these differences are reflected on our
balance sheet for temporary differences in loss and credit carryforwards that will reverse in subsequent years. We also establish
a valuation allowance against deferred tax assets when it is more likely than not that some or all of the deferred tax assets will
not be realized. Valuation allowances are based, in part, on predictions that management must make as to our results in future
periods. The outcome of events could differ over time which would require that we make changes in our valuation allowance.
Use of Estimates
The preparation of consolidated financial statements
in conformity with accounting principles generally accepted in the United States requires management to make estimates and assumptions
that affect the reported amounts of assets and liabilities and disclosures of contingent liabilities at the dates of the consolidated
financial statements and the reported amounts of revenues and expenses during the reporting periods. Actual results could differ
from these estimates. Significant estimates and assumptions include reserves and write-downs related to receivables and inventories,
the recoverability of long-lived assets, the valuation allowance related to our deferred tax assets, valuation of equity and derivative
instruments, debt discounts, valuation of investments and the estimated amortization periods of upfront product licensing fees
received from customers.
Recent Accounting Pronouncements
In February 2016, the Financial Accounting
Standards Board, or FASB, issued ASU 2016-02,
Leases
(“ASU 2016-02”). This standard amends the existing accounting
standards for lease accounting, including requiring lessees to recognize most leases on their balance sheets and making targeted
changes to lessor accounting. ASU 2016-02 will be effective beginning in the first quarter of 2019. Early adoption of ASU 2016-02
is permitted. The new leases standard requires a modified retrospective transition approach for all leases existing at, or entered
into after, the date of initial application, with an option to use certain transition relief. We are currently evaluating the impact
of adopting ASU 2016-02 on our consolidated financial statements.
In March 2016, the FASB issued ASU No. 2016-08,
Revenue from Contracts with Customers (Topic 606): Principal versus Agent Considerations (Reporting Revenue Gross versus Net).
This ASU amends the principal versus agent guidance in ASU No. 2014-09,
Revenue from Contracts with Customers (Topic 606)
which was issued in May 2014 (“ASU 2014-09”). Further, in April 2016, the FASB issued ASU No. 2016-10,
Revenue from
Contracts with Customers (Topic 606): Identifying Performance Obligations and Licensing
. This ASU also amends ASU 2014-09 and
is related to the identification of performance obligations and accounting for licenses. The effective date and transition requirements
for both of these amendments to ASU 2014-09 are the same as those of ASU 2014-09, which was deferred for one year by ASU No. 2015-14,
Revenue from Contracts with Customers (Topic 606): Deferral of the Effective Date
. That is, the guidance under these standards
is to be applied using a full retrospective method or a modified retrospective method, as outlined in the guidance, and is effective
for annual periods, and interim periods within those annual periods, beginning after December 15, 2017. Early adoption is permitted
only for annual periods, and interim period within those annual periods, beginning after December 15, 2016. We are currently evaluating
the provisions of each of these standards and assessing their impact on our consolidated financial statements and disclosures.
In March 2016, the FASB issued ASU No. 2016-09,
Compensation-Stock Compensation (Topic 718): Improvements to Employee Share-Based Payment Accounting.
This ASU makes targeted
amendments to the accounting for employee share-based payments. This guidance is to be applied using various
transition
methods such as full retrospective, modified retrospective, and prospective based on the criteria for the specific amendments as
outlined in the
guidance. The guidance is effective for annual periods, and interim periods within those annual periods,
beginning after December 15, 2016. Early adoption
is permitted, as long as all of the amendments are adopted in the same
period. We are currently evaluating the provisions of this guidance and
assessing its impact on the Company’s consolidated
financial statements and disclosures.
Accounting standards that have been issued
or proposed by the FASB, SEC and/or other standards-setting bodies that do not require adoption until a future date are not expected
to have a material impact on the consolidated financial statements upon adoption.
Results of Operations
Comparison of the Years Ended March 31,
2016 and 2015
Revenues
Total revenues for the year ended March 31,
2016 of $15,084,000 increased by $1,230,000 or 9%, as compared to $13,854,000 for the year ended March 31, 2015. Product revenues
for the year ended March 31, 2016 of $13,042,000 increased by $3,103,000 or 31% when compared to the same period in fiscal 2015.
This increase was the result of strong growth in the United States and Rest of World, partially offset by a slight decline in Latin
America due to the decline in the peso. Product licensing fees and royalties of $981,000 decreased $2,075,000, largely related
to the termination of our contract with our former partner Innovacyn and a reduction of amortization of upfront payments from Laboratorios
Sanfer, S.A. de C.V.
Product revenues in the United States for the
year ended March 31, 2016 of $4,371,000, increased by $2,393,000, or 121%, when compared to the same period in the prior year.
This increase was mostly the result of higher sales of our dermatology products. We currently have a direct sales force team of
19 focused on dermatology and through March 31, 2016, we have launched six new dermatology products.
Product revenue in Europe and the Rest of the
World for the year ended March 31, 2016 of $3,706,000, increased by $798,000, or 27%, as compared to the same period in the prior
year, with the largest increases in Asia, partially offset by a slight decline in the Middle East. The revenue in Europe for the
year ended March 31, 2016, increased 21% in Euro and 6% in U.S. dollars, when compared to the same period last year.
Product revenue in Latin America for the year
ended March 31, 2016 was $4,965,000, down $88,000 or 2%, when compared to the same period in the prior year. This decrease was
caused by a 19% decline in the peso from the same period in the prior year. The sales growth in local currency, when compared to
the prior year, was 17%.
The following table shows our product revenues
by geographic region:
|
|
|
Year Ended March 31,
|
|
|
|
|
|
|
|
|
|
|
2016
|
|
|
2015
|
|
|
$ Change
|
|
|
% Change
|
|
|
United States
|
|
$
|
4,371,000
|
|
|
$
|
1,978,000
|
|
|
$
|
2,393,000
|
|
|
|
121%
|
|
|
Latin America
|
|
|
4,965,000
|
|
|
|
5,053,000
|
|
|
|
(88,000
|
)
|
|
|
(-2%
|
)
|
|
Europe and Rest of the World
|
|
|
3,706,000
|
|
|
|
2,908,000
|
|
|
|
798,000
|
|
|
|
27%
|
|
|
|
|
|
13,042,000
|
|
|
|
9,939,000
|
|
|
|
3,103,000
|
|
|
|
31%
|
|
|
Product license fees and royalties
|
|
|
981,000
|
|
|
|
3,056,000
|
|
|
|
(2,075,000
|
)
|
|
|
(68%
|
)
|
|
Total
|
|
$
|
14,023,000
|
|
|
$
|
12,995,000
|
|
|
$
|
1,028,000
|
|
|
|
8%
|
|
In the year ended March 31, 2016, product license
fees and royalties revenue declined primarily as a result of the termination of our agreement with our former partner Innovacyn,
and a decrease in the amortization of upfront payments from Laboratorios Sanfer, S.A. de C.V.
The following table shows our product license
fees and royalties revenue by partner:
|
|
|
Year Ended March 31,
|
|
|
|
|
|
|
|
|
Product license fees and royalties
|
|
2016
|
|
|
2015
|
|
|
$ Change
|
|
|
% Change
|
|
|
Exeltis (formerly Quinnova)
|
|
$
|
201,000
|
|
|
$
|
437,000
|
|
|
$
|
(236,000
|
)
|
|
|
(54%
|
)
|
|
Innovacyn
|
|
|
29,000
|
|
|
|
1,120,000
|
|
|
|
(1,091,000
|
)
|
|
|
(97%
|
)
|
|
Laboratorios Sanfer (formerly More Pharma)
|
|
|
751,000
|
|
|
|
1,499,000
|
|
|
|
(748,000
|
)
|
|
|
(50%
|
)
|
|
Total product license fees and royalties
|
|
$
|
981,000
|
|
|
$
|
3,056,000
|
|
|
$
|
(2,075,000
|
)
|
|
|
(68%
|
)
|
Service revenues for the year ended March 31,
2016 of $1,061,000 increased by $202,000 when compared to $859,000 in the prior period. This increase was due to an increase in
the number of tests and services provided by our lab services business.
Gross Profit
For the year ended March 31, 2016, we reported
gross profit of $7,210,000 or 48% of revenues, compared to a gross profit of $7,288,000, or 53% of revenues, for the same period
in the prior year. The decline in gross profit was primarily due to the decline in our license fees and royalties revenue of $2,075,000.
For the year ended March 31, 2016, we reported
product gross profit of $6,049,000, or 46%, compared to product gross profit of $4,031,000, or 41%, for the same period in the
prior year. The increase in product gross profit was primarily related to improved margins in the United States as a result of
the launch of higher margin dermatology products.
For the year ended March 31, 2016, we reported
service gross profit of $180,000, or 17%, compared to service gross profit of $201,000, or 23%, for the same period in the prior
year. The decline in service gross profit was primarily related to the mix of tests and services performed.
Research and Development Expense
We reported research and development expense
of $1,806,000 for the year ended March 31, 2016, an increase of $273,000, or 18%, when compared to the same period in the prior
year. The increase is largely due to higher costs related to clinical studies.
Selling, General and Administrative Expense
We reported selling, general and administrative
expenses of $15,556,000 for the year ended March 31, 2016, an increase of $3,142,000, or 25%, when compared to the same period
in the prior year. The increase for the year ended March 31, 2016 was primarily due to higher sales and marketing expenses of $2,212,000
primarily related to the addition of our direct dermatology sales force in the United States, higher costs related to the launch
of new dermatology products and higher stock compensation charges of $505,000.
We expect selling, general and administrative
expenses to increase as we add people to our direct sales force.
Gain due to Change in Fair Value of Derivative
Liabilities
In connection with our December 9, 2013 and
February 26, 2014 registered direct offerings we issued a series of common stock purchase warrants, which contain cash settlement
provisions. During the year ended March 31, 2016, we recorded a decrease in the fair value of our derivative liabilities of $11,000,
primarily due to a decrease in our common stock price and the decreasing contractual term of the warrants. During the year ended
March 31, 2015, we recorded a decrease in the fair value of our derivative liabilities of $3,164,000, primarily due to a decrease
in our common stock price and the decreasing contractual term of the warrants.
Other Expense, Net
Other expense, net of $20,000 for the year
ended March 31, 2016, decreased $36,000, from $56,000 for the same period in the prior year. The decrease in other expense, net
for the year ended March 31, 2016 was primarily related to foreign exchange gains and losses.
Net Loss
Net loss for the year ended March 31, 2016
was $10,162,000 compared to $8,203,000, for the same period in the prior year. The increase in net loss is primarily due to an
increase of $3,415,000 in our operating expenses mostly related to our dermatology division and $570,000 of additional stock compensation
expenses in the current period. Additionally, for the year ended March 31, 2016 our product licensing and royalties’ revenues
decreased $2,075,000 due to a combined decrease in amortization of upfront fees related to Laboratorios Sanfer and the loss of
our animal health partner Innovacyn. This was offset by an increase in our product related gross profit of $2,018,000. During the
year ended March 31, 2015, we recorded an impairment loss in the amount of $4,650,000 related to our investment in Ruthigen, offset
by $3,164,000 gain due to a change in fair value of our derivative liabilities. These transactions did not recur during the year
ended March 31, 2016.
Comparison of the Three Months Ended June
30, 2016 and 2015
Revenues
Total revenues for the three months ended June
30, 2016 of $3,811,000 increased by $131,000 or 4%, as compared to $3,680,000 for the three months ended June 30, 2015. Product
revenues for the three months ended June 30, 2016 of $3,509,000 increased by $593,000 or 20% when compared to the same period in
the prior year. This increase was the result of strong growth in the United States, Europe and Rest of World, partially offset
by a decline in Latin America due to the 19% decline in the peso and a robust quarter last year. Product licensing fees and royalties
of $75,000 decreased $372,000, largely related to a reduction in the amortization of upfront payments from Laboratorios Sanfer,
S.A. de C.V.
Product revenues in the United States for the
three months ended June 30, 2016 of $1,373,000, increased by $586,000, or 74%, when compared to the same period in the prior year.
This increase was mostly the result of higher sales of our dermatology and animal health products. We currently have a direct sales
force team of 20 focused on dermatology and a strong product portfolio of seven products, for the treatment of atopic dermatitis,
scar management, surgical procedures, an oral anti-infective for severe acne and Ceramax, which utilizes a “state of the
art” skin repair technology. In addition, sales to a new animal health care partner increased during the period compared
to the prior year.
Product revenue in Europe and the Rest of the
World for the three months ended June 30, 2016 of $1,038,000, increased by $467,000, or 82%, as compared to the same period in
the prior year, with increases in Europe, Asia, Middle East and India. Revenue in Europe for the three months ended June 30, 2016,
increased 56% in U.S. dollars, when compared to the same period last year primarily due to the launch of new products and expansion
of the customer base.
Product revenue in Latin America for the three
months ended June 30, 2016 was $1,098,000, down $460,000 or 30%, when compared to the same period in the prior year. This decrease
was caused by a 19% decline in the peso from the same period in the prior year, as well as higher sales during the three months
ended June 30, 2015 due to stocking by our new Latin American partner, Sanfer, to fill their expansive pharmacy store network.
The following table shows our product revenues
by geographic region:
|
|
|
Three months ended June 30,
|
|
|
|
|
|
|
|
|
|
|
2016
|
|
|
2015
|
|
|
$ Change
|
|
|
% Change
|
|
|
United States
|
|
$
|
1,373,000
|
|
|
$
|
787,000
|
|
|
$
|
586,000
|
|
|
|
74%
|
|
|
Latin America
|
|
|
1,098,000
|
|
|
|
1,558,000
|
|
|
|
(460,000
|
)
|
|
|
(30%
|
)
|
|
Europe and Rest of the World
|
|
|
1,038,000
|
|
|
|
571,000
|
|
|
|
467,000
|
|
|
|
82%
|
|
|
|
|
|
3,509,000
|
|
|
|
2,916,000
|
|
|
|
593,000
|
|
|
|
20%
|
|
|
Product license fees and royalties
|
|
|
75,000
|
|
|
|
447,000
|
|
|
|
(372,000
|
)
|
|
|
(83%
|
)
|
|
Total
|
|
$
|
3,584,000
|
|
|
$
|
3,363,000
|
|
|
$
|
221,000
|
|
|
|
7%
|
|
In the three months ended June 30, 2016, product
license fees and royalties revenue declined primarily as a result of a decrease in the amortization of upfront payments from Laboratorios
Sanfer, S.A. de C.V.
The following table shows our product license
fees and royalties revenue by partner:
|
|
|
Three Months Ended June 30,
|
|
|
|
|
|
|
|
|
Product license fees and royalties
|
|
2016
|
|
|
2015
|
|
|
$ Change
|
|
|
% Change
|
|
|
Exeltis
|
|
$
|
–
|
|
|
$
|
54,000
|
|
|
$
|
(54,000
|
)
|
|
|
(100%
|
)
|
|
Innovacyn
|
|
|
–
|
|
|
|
20,000
|
|
|
|
(20,000
|
)
|
|
|
(100%
|
)
|
|
Laboratorios Sanfer, S.A. de C.V.
|
|
|
75,000
|
|
|
|
373,000
|
|
|
|
(298,000
|
)
|
|
|
(80%
|
)
|
|
Total product license fees and royalties
|
|
$
|
75,000
|
|
|
$
|
447,000
|
|
|
$
|
(372,000
|
)
|
|
|
(83%
|
)
|
Service revenues for the three months ended
June 30, 2016 of $227,000 decreased by $90,000 when compared to $317,000 in the prior period. This decrease was due to a decrease
in the number of tests and services provided by our lab services business.
Gross Profit
For the three months ended June 30, 2016, we
reported total gross profit of $1,919,000 or 50% of revenues, compared to a gross profit of $1,873,000, or 51% of revenues, for
the same period in the prior year. The decline in gross profit was primarily due to the decline in our license fees and royalties
revenue of $372,000.
For the three months ended June 30, 2016, we
reported product gross profit of $1,802,000, or 51%, compared to product gross profit of $1,400,000, or 48%, for the same period
in the prior year. The increase in product gross profit was primarily related to product mix from higher sales of dermatology products
which have higher margins.
For the three months ended June 30, 2016, we
reported service gross profit of $42,000, or 19%, compared to service gross profit of $26,000, or 8%, for the same period in the
prior year. The increase in service gross profit was primarily related to the mix of tests and services performed.
Research and Development Expense
We reported research and development expense
of $360,000 for the three months ended June 30, 2016, a decrease of $107,000, or 23%, when compared to the same period in the prior
year. The decrease is largely due to a decrease in development milestone payments and license fees related to a dermatology product.
Selling, General and Administrative Expense
We reported selling, general and administrative
expenses of $4,130,000 for the three months ended June 30, 2016, an increase of $413,000, or 11%, when compared to the same period
in the prior year. The increase for the three months ended June 30, 2016 was primarily due to higher sales and marketing expenses
of $532,000 primarily related to the addition of our direct dermatology sales force in the United States and the launch of new
dermatology products, partly offset by lower expenses in Mexico and Europe and lower stock compensation charges.
We expect selling, general and administrative
expenses to increase as we add people to our direct sales force.
Interest Expense
Interest expense was negligible for the three
months ended June 30, 2016 and 2015.
Interest Income
Interest income was negligible for the three
months ended June 30, 2016 and 2015.
Loss due to Change in Fair Value of Derivative
Liabilities
In connection with our December 9, 2013 and
February 26, 2014 registered direct offerings we issued a series of common stock purchase warrants, which contain cash settlement
provisions. During the quarter ended June 30, 2015, we recorded a loss due to an increase in the fair value of our derivative liabilities
of $59,000, primarily due to an increase in our common stock price, partially offset by the decreasing contractual term of the
warrants.
Other Income, Net
Other income, net of $3,000 for the three months
ended June 30, 2016, decreased $27,000, from $30,000 of other income, net for the same period in the prior year. The decrease in
other income, net for the three months ended June 30, 2016 was primarily related to foreign exchange gains and losses.
Net Loss
Net loss for the three months ended June 30,
2016 was $2,568,000 compared to $2,340,000, for the same period in the prior year. The increase in net loss of $228,000 is primarily
due to an increase of $306,000 in our operating expenses mostly related to our dermatology division, partly offset by an increase
in gross profit.
Liquidity and Capital Resources
We incurred a net loss of $2,568,000 for the
three months ended June 30, 2016. At June 30, 2016 and March 31, 2016, our accumulated deficit amounted to $154,943,000 and $152,375,000,
respectively. At June 30, 2016 and March 31, 2016, our working capital amounted to $6,965,000 and $9,337,000, respectively. We
may raise additional capital from external sources in order to fund working capital and continue the longer term efforts contemplated
under our business plan. We expect to continue incurring losses for the foreseeable future and may need to raise additional capital
to pursue our product development initiatives, to penetrate markets for the sale of our products and continue as a going concern.
We cannot provide any assurances that we will be able to raise additional capital.
Management believes that we have access to
capital resources through possible public or private equity offerings, debt financings, corporate collaborations or other means;
however, we cannot provide any assurance that new financing will be available on commercially acceptable terms, if at all. If the
economic climate in the U.S. deteriorates, our ability to raise additional capital could be negatively impacted. If we are unable
to secure additional capital, we may be required to curtail our research and development initiatives and take additional measures
to reduce costs in order to conserve our cash in amounts sufficient to sustain operations and meet our obligations. These measures
could cause significant delays in our efforts to commercialize our products, which is critical to the realization of our business
plan and our future operations. These matters raise substantial doubt about our ability to continue as a going concern.
Sources of Liquidity
As of June 30, 2016 we had cash and cash equivalents
of $4,970,000. Since our inception, substantially all of our operations have been financed through sales of equity securities.
Other sources of financing that we have used to date include our revenues, as well as various loans.
Since July 1, 2014, substantially all of our
operations have been financed through the following transactions:
|
|
·
|
proceeds of $14,000 received from the exercise of common stock purchase warrants and options;
|
|
|
·
|
net proceeds of $5,444,000 received from an underwritten public offering on January 26, 2015;
|
|
|
·
|
net proceeds of $5,335,000 received from the sale of Ruthigen common stock;
|
|
|
·
|
net proceeds of $2,994,000 received from an underwritten public offering on March 18, 2016; and
|
|
|
·
|
net proceeds of $4,491,000 received from the sale of common stock through our At the Market Issuance Sales Agreement as of June 30, 2016.
|
Cash Flows for the year ended March 31, 2016, and 2015
As of March 31, 2016, we had cash and cash
equivalents of $7,469,000, compared to $6,136,000 as of March 31, 2015.
Net cash used in operating activities during
the year ended March 31, 2016 was $8,746,000, primarily due to our net loss of $10,162,000, offset by non-cash transactions during
the year ended March 31, 2016, including $2,341,000 of stock-based compensation expenses.
Net cash used in operating activities during
the year ended March 31, 2015 was $6,694,000, primarily due to our net loss of $8,203,000, offset by non-cash transactions during
the year ended March 31, 2015, including: $4,650,000 of impairment loss related to our investment in Ruthigen; a $3,164,000 gain
on the fair value adjustment of our derivative liabilities; and $1,771,000 of stock-based compensation expenses and $1,499,000
of upfront fees amortized related to More Pharma.
Net cash provided by investing activities was
$4,191,000 for the year ended March 31, 2016, consisting of $345,000 related to equipment purchases offset by $4,538,000 received
from the sale of 1,650,000 of our shares of Ruthigen common stock.
Net cash provided by investing activities was
$875,000 for the year ended March 31, 2015, consisting of $139,000 related to equipment purchases offset by $963,000 received from
the sale of 350,000 of our shares of Ruthigen common stock and $51,000 related to changes in long-term assets.
Net cash provided by financing activities was
$6,039,000 for the year ended March 31, 2016. During the period ended March 31, 2016, we received net proceeds from the March 18,
2016 underwritten offering of common stock and common stock purchase warrants of $2,994,000 and net proceeds of $3,150,000 from
an At the Market Issuance of common stock. The offering proceeds were offset by principal payments on the debt in the amount of
$119,000.
Net cash provided by financing activities was
$6,609,000 for the year ended March 31, 2015. During the period ended March 31, 2015, we received net proceeds from the January
26, 2015 underwritten offering of common stock and common stock purchase warrants of $5,444,000 and net proceeds of $1,341,000
from an At the Market Issuance of common stock. The offering proceeds were offset by principal payments on the debt in the amount
of $176,000.
Cash Flows for the three months ended June
30, 2016 and 2015
As of June 30 2016, we had cash and cash equivalents
of $4,970,000, compared to $7,469,000 as of March 31, 2016.
Net cash used in operating activities during
the three months ended June 30, 2016 was $2,377,000, primarily due to our net loss of $2,568,000 offset by stock compensation of
$411,000 in the period.
Net cash used in operating activities during
the three months ended June 30, 2015 was $2,585,000, primarily due to our net loss of $2,340,000 for the period and an increase
in accounts receivable of $1,052,000 due to increased sales on payment terms, offset by an increase of $549,000 in accounts payable
and accrued liabilities
Net cash used in investing activities was $11,000
for three months ended June 30, 2016, primarily related to the purchase of equipment.
Net cash provided by investing activities was
$4,378,000 for the three months ended June 30, 2015, consisting of $4,538,000 received from the sale of 1,650,000 of our shares
of Ruthigen common stock, offset by $148,000 related to equipment purchases and $12,000 related to changes in long-term assets.
Net cash used in financing activities was $48,000
for the three months ended June 30, 2016 related to principal payments on debt.
Net cash provided by financing activities was
$827,000 for the three months ended June 30, 2015 was primarily related to $879,000 of net proceeds received from At-the-Market
Issuances of common stock which was offset by principal payments on debt in the amount of $52,000.
Contractual Obligations
As of March 31, 2016, we had contractual obligations
as follows (long-term debt amounts include principal payments only):
|
|
|
Payments Due by Period
|
|
|
|
|
Total
|
|
|
Less Than
1 Year
|
|
|
1-3
Years
|
|
|
After
3 Years
|
|
|
Long-term debt
|
|
$
|
114,000
|
|
|
$
|
114,000
|
|
|
$
|
–
|
|
|
$
|
–
|
|
|
Operating leases
|
|
|
691,000
|
|
|
|
370,000
|
|
|
|
312,000
|
|
|
|
–
|
|
|
Total
|
|
$
|
805,000
|
|
|
$
|
484,000
|
|
|
$
|
312,000
|
|
|
$
|
–
|
|
Operating Capital and Capital Expenditure Requirements
We reported a net loss of $10,162,000 for the
year ended March 31, 2016. At March 31, 2016, our accumulated deficit amounted to $152,375,000. We had working capital of $9,337,000
as of March 31, 2016.
We may need to raise additional capital from
external sources in order to continue the longer term efforts contemplated under our business plan. We expect to continue incurring
losses for the foreseeable future and may need to raise additional capital to pursue our product development initiatives and to
penetrate markets for the sale of our products.
Our future funding requirements will depend
on many factors, including:
|
|
·
|
Our current and future revenues;
|
|
|
·
|
the scope, rate of progress and cost of our research and development activities;
|
|
|
·
|
future clinical trial results;
|
|
|
·
|
the terms and timing of any collaborative, licensing and other arrangements that we may establish;
|
|
|
·
|
the cost and timing of regulatory approvals;
|
|
|
·
|
the cost and delays in product development as a result of any changes in regulatory oversight applicable to our products;
|
|
|
·
|
the cost and timing of establishing sales, marketing and distribution capabilities;
|
|
|
·
|
the effect of competing technological and market developments;
|
|
|
·
|
the cost of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights; and
|
|
|
·
|
the extent to which we acquire or invest in businesses, products and technologies.
|
Off-Balance Sheet Transactions
We currently have no off-balance sheet arrangements
that have or are reasonably likely to have a current or future material effect on our financial condition, changes in financial
condition, revenues or expenses, results of operations, liquidity, capital expenditures or capital resources.
Changes
In and Disagreements with Accountants on Accounting and Financial Disclosure
There have been no
disagreements with our independent public accountant in regards to accounting and financial disclosure.
Quantitative
and Qualitative Disclosures about Market Risk
As a smaller reporting company as defined by
Rule 12b-2 of the Exchange Act and in Item 10(f)(1) of Regulation S-K, we are electing scaled disclosure reporting
obligations and therefore are not required to provide the information requested by this Item.
OUR BUSINESS
Corporate Information
We incorporated under the laws of the State
of California in April 1999 as Micromed Laboratories, Inc. In August 2001, we changed our name to Oculus Innovative Sciences, Inc.
In December 2006, we reincorporated under the laws of the State of Delaware. Our principal executive offices are located at 1129 N. McDowell
Blvd., Petaluma, California, 94954, and our telephone number is (707) 283-0550. We have two principal wholly-owned subsidiaries:
Oculus Technologies of Mexico, S.A. de C.V., organized in Mexico; and Oculus Innovative Sciences Netherlands, B.V., organized in
the Netherlands. Our fiscal year end is March 31. Our website is www.oculusis.com. We do not intend for information on our
website to be incorporated into this annual report.
Overview
We are a specialty pharmaceutical company dedicated
to identifying, developing and commercializing unique, affordable differentiated therapies to improve the lives of patients with
dermatologic diseases or conditions. Our products, which are sold throughout the United States and internationally, have improved
patient outcomes for more than five million patients globally by treating and reducing certain topical skin diseases including
acne, atopic dermatitis, scarring, infections, itch, pain and harmful inflammatory responses.
We currently focus on the development and commercialization
of therapeutic solutions in medical dermatology to treat or reduce skin conditions, such as acne, atopic dermatitis and scarring.
These diseases impact millions of patients worldwide and can have significant, multi-dimensional effects on patients’ quality
of life, including their physical, functional and emotional well-being.
Since our founding in 1999, we built our business
by developing and promoting products via partnerships for multiple therapeutic indications, with a primary focus on advanced tissue
care. Starting in 2013, with a new Board of Directors and new management team, we pivoted to focus on one specialty pharmaceutical
area, medical dermatology, and created our own sales force in the United States to promote our unique, affordable, differentiated
prescription dermatology products.
Some of our key products in the United States
are:
|
|
·
|
Celacyn®, a prescription hypochlorous acid based scar management gel clinically proven to soften and flatten raised scars while reducing redness and discoloration.
|
|
|
·
|
Ceramax™ Skin Barrier Cream helps manage dry itchy skin, minor skin irritations, rashes, and inflammation caused by various skin conditions.
|
|
|
·
|
Alevicyn™, a prescription hypochlorous acid based atopic dermatitis product line clinically proven to reduce pruritus (itch) and pain associated with various dermatoses.
|
|
|
·
|
Mondoxyne™, a prescription oral tetracycline antibiotic used for the treatment of certain bacterial infections, including acne.
|
|
|
·
|
Microcyn® or Microdacyn60® (sold under a variety of brand names), a line of products based on electrically charged oxychlorine small molecules designed to target a wide range of pathogens including viruses, fungi, spores and bacteria, including antibiotic-resistant strains.
|
Our key product outside the United States is:
|
|
·
|
Microcyn® or Microdacyn60® (sold under a variety of brand names), a line of products based on electrically charged oxychlorine small molecules designed to target a wide range of pathogens including viruses, fungi, spores and bacteria, including antibiotic-resistant strains.
|
To date, we have obtained 15 clearances from
the U.S. Food and Drug Administration, or FDA, that permit us to sell our products as medical devices for Section 510(k) of the
Federal Food, Drug and Cosmetic Act in the United States.
Outside the United Sates, we sell products
for dermatological and advanced tissue care with a European Conformity marking (known as Conformité Européenne or
CE) covering 25 of our products, 14 approvals from the Mexican Ministry of Health, and various approvals in Central America, China,
Southeast Asia, and the Middle East.
Our Strategy
Our strategy is to in-license, acquire, develop
and commercialize unique, affordable and differentiated therapies that we believe advance the standard of care for patients with
dermatological diseases. The key components of our strategy are to:
|
|
·
|
Expand our Internal U.S. Sales Force:
We continue to hire additional experienced sales people who have established relationships with dermatologists in their territories. As of March 31, 2016, we had a U.S. direct sales force team of 19 dedicated sales people.
|
|
|
·
|
Develop and Launch New Dermatology Products:
We currently sell six prescription dermatology products in the United States, and have a strong product pipeline of new products, including our new product, Lasercyn, intended for the management of post-non-ablative laser therapy procedures, post-microdermabrasion therapy and following superficial chemical peels, that we intend to launch over the next nine months.
|
|
|
·
|
Create a Competitive Pricing Strategy:
We have and will continue to develop a unique product pricing strategy, which we believe solves many of the challenges associated with the prescription dermatology market’s current pricing and rebate programs.
|
|
|
·
|
Develop a Pharmaceutical Line:
We plan to acquire or develop pharmaceutical products with affordable clinical trials to increase our market presence and create innovator patent protection.
|
Our plan is to evolve into a leading dermatology
company, providing innovative and cost-effective solutions to patients, while generating strong, consistent revenue growth
and maximizing long-term shareholder value.
OUR PRODUCTS
In the United States some of our key dermatology
products are:
Celacyn® – Prescription Scar
Management Gel

|
Celacyn®, is a prescription hypochlorous
acid based scar management gel designed to soften and flatten raised scars while reducing redness and discoloration. In our studies,
Celacyn® has been shown to reduce scar itch pain and performed better than the market-leading comparable gel brand. In the
United States, topical prescription scar treatment products are usually sold over the counter. By contrast, we actively market
Celacyn® to clinicians.
Scars are a natural part of the healing process
and a reaction to skin injury. Scars form when the dermis, or the lower level of the skin, is damaged and then repaired by a process
called granulation, where the body produces collagen fibers to repair the damage. Celacyn® works on keloid and hypertrophic
scars. Keloid scars continue to grow after the skin has healed which causes the scars to grow beyond the originally damaged area.
Hypertrophic scars are marked by excessive scar tissue in a local area and appear thick, red and lumpy.
Celacyn® scar gel is intended for the management
of old and new scars resulting from burns, general surgical procedures and trauma wounds.
|
Ceramax™ – Skin Barrier Cream

|
Ceramax™ Skin Barrier Cream helps manage
dry itchy skin, minor skin irritations, rashes, and inflammation caused by various skin conditions based on patented Lipogrid®
Technology. Ceramax™ Skin Barrier Cream can be used to treat a variety of disease states with skin barrier disruption, including
eczema and atopic dermatitis.
According to the National Eczema Association,
eczema or atopic dermatitis affects over 10% of the children in the United States and one out of every three children with eczema
or atopic dermatitis have moderate to severe symptoms. Additionally, 31.6 million people have some form of eczema with approximately
17.8 million of those having moderate to severe eczema or atopic dermatitis.
Ceramax™ Skin Barrier Cream is intended
to be used as a topical skincare preparation to relieve and manage the burning and itching associated with various skin conditions,
including atopic dermatitis, and other dry skin conditions, by maintaining a moist wound and skin environment. Lipogrid® Technology
contains lipids that blend in with the skin’s natural lipid building blocks to hydrate and restore the natural skin barrier
and penetrate the skin.
|
Alevicyn™ SG Antipruritic Spray
Gel, Dermal Spray and Antipruritic Gel

|
Alevicyn™ is indicated to manage and
relieve the burning, itching and pain experienced with various types of skin conditions, including radiation dermatitis and atopic
dermatitis. It may be also used to relieve the pain of first- and second-degree burns, and helps to relieve dry waxy skin by maintaining
a moist wound and skin environment, which is beneficial to the healing process. Alevicyn™ Antipruritic Gel is intended for
management of itch and pain associated with dermal irritations and wounds, such as sores, injuries and ulcers of dermal tissue.
Alevicyn™ Antipruritic Spray gel’s
unique formulation is a “spray-on” that does not run or drip after application and no “rubbing-in” is required
on sensitive or difficult-to-access areas of the body. Alevicyn™ dermal spray is intended for the cleansing, irrigation,
moistening, debridement and removal of foreign material including microorganisms and debris from wounds, among others, first- and
second-degree bums, abrasions, minor irritations of the skin, diabetic foot ulcers, and ingrown toe nails.
|
Mondoxyne™ – Prescription
Oral Antibiotic

|
Mondoxyne™ is a prescription oral tetracycline
antibiotic that contains doxycycline, a broad spectrum antibacterial synthetically derived from oxytertracycline, used as a treatment
for acne vulgaris.
According to the British Association of Dermatologists,
acne vulgaris is estimated to affect 660 million people, or 9.4% of the global population, and it is the eighth most common disease
worldwide. Acne is thought to have multiple contributing factors, including, among other things, excess sebum, or oil, production,
which creates an optimal environment for the proliferation of the bacterium
Propionibacterium acnes
. The
Propionibacterium
acnes
bacteria feed on the sebum and secrete enzymes and other byproducts that irritate the skin and result in the inflammation
commonly known as acne.
Mondoxyne™ is an oral antibiotic that
can be effective against acne because of its antimicrobial and anti-inflammatory activity. It is usually prescribed as adjunct
therapy for severe inflammatory acne. Mondoxyne™ treats acne by targeting the bacterium
Propionibacterium acnes
. Patients
have rated doxycycline, the active ingredient in Mondoxyne™, as effective or very effective in 85% of cases, as reported
by a 1989 double-blind study published in the
Journal of Dermatological Treatment
comparing the effectiveness of doxycycline
and minocycline in the treatment of moderate to moderately severe acne.
|
Microcyn® -Advanced Tissue Care Management

|
Microcyn® is based on electrically charged oxychlorine small molecules designed to target a wide range of pathogens including viruses, fungi, spores and bacteria, including antibiotic-resistant strains. Several Microcyn® Technology advanced tissue care products are designed to treat infections and enhance healing while reducing the need for antibiotics. When a wound is slow to heal or becomes hard to heal, the costs to treat increase and the quality of life for the patient also suffers as infected, malodorous wounds prevent them from participating in daily life activities. As a result of our patented manufacturing process, Microcyn® is a proprietary solution of oxychlorine compounds that, among other things, interacts with and inactivates surface proteins on cell walls and membranes of microorganisms. The functions of these proteins are varied and play significant roles in cell communication, nutrient and waste transport and other required functions for cell viability.
|
Once Microcyn® surrounds single cell microorganisms,
it damages these proteins, causing the cell membrane to rupture, leading to cell death, which we believe is caused by increased
membrane permeability and induced osmotic pressure imbalance. This destruction of the cell appears to occur through a fundamentally
different process than that which occurs as a result of contact with a bleach-based solution because experiments have demonstrated
that Microcyn® kills bleach-resistant bacteria. However, we believe the solution remains non-irritating to human tissues because
human cells have unique protective mechanisms, are interlocked, and prevent Microcyn® from targeting and surrounding single
cells topically on the body. Laboratory tests suggest that our solution does not penetrate and kill multi-cellular organisms, and
does not damage or affect human DNA.
In laboratory tests, Microcyn® has been
shown to destroy certain biofilms. A biofilm is a complex cluster of microorganisms or bacteria marked by the formation of a protective
shell, allowing the bacteria to collect and proliferate. It is estimated that over 65% of microbial infections in the body involve
bacteria growing as a biofilm. Bacteria living in a biofilm typically have significantly different properties from free-floating
bacteria of the same species. One result of this film environment is increased resistance to antibiotics and to the body’s
immune system. In chronic wounds, biofilms interfere with the normal healing process and halt or slow wound closure. Bacteria growing
in biofilms can become up to 1000-fold more resistant to antibiotics and other biocides as compared to their planktonic, or free
floating, counterparts. As a result, biofilm infections cannot be effectively treated with conventional antibiotic therapy. In
our laboratory studies, Microcyn® was shown to destroy two common biofilms after five minutes of exposure.
In published studies, Microcyn® has been
shown to significantly increase the dilation of capillaries in wounds as indicated by higher levels of oxygen at a wound site after
the application of our product and also to reduce inflammation by inhibiting certain inflammatory responses from allergy-producing
mast cells. It is widely accepted that reducing chronic inflammation surrounding an injury or wound is beneficial to wound healing.
Our laboratory research suggests that Microcyn®’s interference with these cells is selective to only the inflammatory
response and does not interfere with other functions of these cells. Microcyn® Technology has demonstrated antimicrobial activity
against numerous bacterial, viral and fungal pathogens, including antibiotic-resistant strains, as evidenced by passing results
in numerous standardized laboratory microbiology tests conducted on our 510(k) approved technology by a variety of certified independent
testing laboratories.
Regulatory Approvals and Clearances
To date, we have obtained 15 clearances from
the U.S. Food and Drug Administration, or FDA, that permit us to sell our products as medical devices for Section 510(k) of the
Federal Food, Drug and Cosmetic Act in the United States.
Outside the United Sates, we sell products
for dermatological and advanced tissue care with a European Conformity marking (known as Conformité Européenne or
CE) covering 25 of our products, 14 approvals from the Mexican Ministry of Health, and various approvals in Central America, China,
Southeast Asia, and the Middle East.
The following table
summarizes our material current regulatory approvals and clearances by brand:
|
Brand
|
|
Approval Type
|
|
Year of Approval
|
|
Summary Indication
|
|
|
|
|
|
|
|
|
|
Lasercyn™
|
|
U.S. 510(k)
|
|
2016
|
|
Indicated for the management of post non ablative laser therapy procedures, post microdermabrasion therapy and following superficial chemical peels; and to relieve itch and pain from minor skin irritations, lacerations, abrasions and minor burns.
|
|
|
|
|
|
|
|
|
|
|
EU CE Mark
|
|
2016
|
|
|
|
|
|
|
|
|
|
|
MucoClyns™
|
|
EU CE Mark
|
|
2016
|
|
Indicated for the use in emergencies, safe to use on mucous membranes, cuts, abrasions, burns and body surfaces for the treatment immediately after an unexpected exposure to infection risk, and professional medical attention.
|
|
|
|
|
|
|
|
|
|
Sinudox™
|
|
EU CE Mark
|
|
2016
|
|
Solution intended for nasal irrigation, including the moistening of cuts, abrasions and lacerations located in the nasal cavity.
|
|
|
|
|
|
|
|
|
|
|
Mex. Medical Device
|
|
2014
|
|
|
|
|
|
|
|
|
|
|
SebDerm Gel
|
|
U.S. 510(k)
|
|
2015
|
|
Manages and relieves the burning, itching, erythema, scaling, and pain experienced with seborrhea and seborrheic dermatitis. It also helps to relieve dry, waxy skin by maintaining a moist wound and skin environment, which is beneficial to the healing process.
|
|
|
|
|
|
|
|
|
|
Celacyn®
|
|
U.S. 510(k)
|
|
2013
|
|
As hydrogel for the management of old and new hypertrophic and keloid scarring resulting from burns, general surgical procedures and trauma wounds.
|
|
|
|
|
|
|
|
|
|
Alevicyn™
|
|
U.S. 510(k)
|
|
2011
|
|
As a hydrogel, for management and relief of burning, itching and pain experienced with various types of dermatoses, including atopic dermatitis and radiation dermatitis.
|
|
|
|
|
|
|
|
|
|
|
EU CE Mark
|
|
2013
|
|
|
|
|
|
|
|
|
|
|
|
Mex. Medical Device
|
|
2013
|
|
|
|
|
|
|
|
|
|
|
Epicyn™
|
|
U.S. 510(k)
|
|
2011
|
|
Manages and relieves itching, burning and pain experienced with various types of dermatoses, including atopic dermatitis, first and second degree burns. Indicated as an adjuvant in the wound healing process with wounds that can only heal by secondary intention in maturation phase. Epicyn™ is effective for the management and reduction of new and existing hypertrophic and keloid scars.
|
|
|
|
|
|
|
|
|
|
|
EU CE Mark
|
|
2013
|
|
|
|
|
|
|
|
|
|
|
|
Mex. Medical Device
|
|
2013
|
|
|
|
|
|
|
|
|
|
|
Microcyn™ Skin and Wound Care or HydroGel
|
|
U.S. 510(k)
|
|
2010
|
|
As a solution or hydrogel, for debridement and moistening of acute and chronic wounds, ulcers, cuts, abrasions and burns, including those located in any human cavity such as the oral, nasal or ear. .
|
|
|
|
|
|
|
|
|
EU CE Mark
|
|
2013
|
|
|
|
|
|
|
|
|
|
|
Mex. Medical Device
|
|
2014
|
|
|
|
|
|
|
|
|
|
|
Gramaderm®
|
|
EU CE Mark
|
|
2013
|
|
As a dermatological solution or hydrogel for the topical treatment of mild to moderate acne.
|
|
|
|
|
|
|
|
|
|
|
Mex. Medical Device
|
|
2010 & 2012
|
|
|
|
|
|
|
|
|
|
|
Microcyn™ Skin and Wound Cleanser
|
|
U.S. 510(k)
|
|
2009
|
|
Debridement of wounds, such as stage I-IV pressure ulcers, diabetic foot ulcers, post-surgical wounds, first- and second-degree burns, grafted and donor sites as preservative, which can kill listed bacteria such as MRSA & VRE and required as a prescription.
|
|
|
|
|
|
|
|
|
|
Microcyn™ Wound Gel
|
|
U.S. 510(k)
|
|
2009
|
|
Manages exuding wounds such as leg ulcers, pressure ulcers, diabetic ulcers and mechanical or surgical debridement of wounds in a gel form.
|
|
|
|
|
|
|
|
|
|
Alevicyn™ SG Antipruritic Gel
|
|
U.S. 510(k)
|
|
2015
|
|
As a thin hydrogel, for the management and relief of burning, itching and pain experienced with various types of dermatoses, including atopic dermatitis and radiation dermatitis.
|
|
|
|
|
|
|
|
|
|
Ceramax™ Skin Barrier Cream
|
|
U.S. 510(k)
|
|
2015
|
|
Management of dry itchy skin, minor skin irritations, rashes, and inflammation caused by various skin conditions based on patented Lipogrid® Technology.
|
Clinical Trials
We have completed a proof-of-concept Phase II
trial in the United States, which demonstrated the effectiveness of Microcyn® Technology in mildly infected diabetic foot ulcers
with the primary endpoint of clinical cure and improvement of infection. We used 15 clinical sites and enrolled 48 evaluable patients
in three arms, using Microcyn® alone, Microcyn® plus an oral antibiotic and saline plus an oral antibiotic. We announced
the results of our Phase II trial in March 2008. In the clinically evaluable population of the study, the clinical success
rate at visit four, or test of cure, for Microcyn®-alone-treated patients was 93.3% compared to 56.3% for the oral antibiotic
levofloxacin plus saline-treated patients. This study was not statistically powered, but the high clinical success rate (93.3%)
and the p-value (0.033) suggest the difference is meaningfully positive for the Microcyn®-treated patients. Also, for this
set of data, the 95.0% confidence interval for the Microcyn®-only arm ranged from 80.7% to 100% while the 95.0% confidence
interval for the oral antibiotic levofloxacin and saline arm ranged from 31.9% to 80.6%; the confidence intervals do not overlap,
indicating a favorable clinical success for Microcyn® compared to the oral antibiotic levofloxacin. At visit three (end of
treatment), the clinical success rate for patients treated with Microcyn®-alone was 77.8% compared to 61.1% for the oral antibiotic
levofloxacin plus saline-treated patients. We have not done any subsequent clinical trials in the drug process for tissue care.
Domestic Sales and Marketing
In the United States, we sell into dermatology
markets through our IntraDerm™ Pharmaceuticals division staffed with a seasoned management and sales team. We sell into the
advanced tissue care markets with our dedicated contract sales force, and the animal health market through our business partners.
Our dermatology products are primarily purchased by distributors and wholesalers, pharmacies and dermatologist. Our tissue care
products are primarily purchased by hospitals, physicians, nurses, and other healthcare practitioners, who are the primary caregivers
to patients, both human and animal, being treated for acute or chronic wounds or undergoing surgical procedures, as well as to
dermatologists for treatment of various skin afflictions.
Although specific customer requirements can
vary depending on applications, customers generally demand quality, innovation, affordability and clinically-supported efficacy.
We have responded to these customer demands by introducing new products focused on these requirements in the markets we serve.
Specifically, we believe that we introduce new products and applications that are innovative, address the specific dermatological
procedures in demand, and supported by human clinical data. In addition, we provide attractive product line extensions and pricing
to new product families. In the future to increase market penetration, in addition to marketing to our core dermatologists, we
may also market our products to aesthetic dermatologists and plastic surgeons.
We seek to establish strong ongoing relationships
with our customers through the new products, sales of existing products, ongoing training and support, and distributing skincare
products. We primarily target our marketing efforts to practitioners through office visits, workshops, trade shows, webinars and
trade journals. We also market to potential patients through brochures, workshops and our website. In addition, we offer clinical
forums with recognized expert panelists to promote advanced treatment techniques using our products to further enhance customer
loyalty and uncover new sales opportunities.
International Sales and Marketing by Our
Strategic Business Partners
We sell our products
through a worldwide distributor network in over 40 countries.
In the international markets, we work with a network of partners,
ranging from country specific distributors to a large pharmaceutical company to a full services sales and marketing company. International
sales are generally made through a worldwide distributor network in over 40 countries. Our international revenue as a percentage
of total revenue represented 67% in FY 2016 and 73% in FY 2015.
Europe
We currently rely on exclusive agreements with
country-specific distributors for the sale of Microcyn®-based products in Europe, including Austria, Belgium, Italy, Luxemburg,
the Netherlands, Germany, Greece, the Czech Republic, Sweden, Spain, Norway, Switzerland, Poland, Finland, Denmark and Serbia.
Mexico
In Mexico, we partner with Laboratorios Sanfer
S.A. de C.V. Laboratorios Sanfer is one of the largest independent pharmaceutical companies in Mexico, operating in nine countries
across Latin America. Laboratorios Sanfer manufactures, markets and sells prescription and over the counter branded medications
across five therapeutic areas including gastroenterology, cardiology, anti-infective and dermatology. Pursuant to our agreement
with Laboratorios Sanfer, we granted Laboratorios Sanfer an exclusive license, with the right to sublicense, under certain conditions
and with our consent, to all of our proprietary rights related to certain of our pharmaceutical products for human application
that utilize our Microcyn® technology within Mexico. We also agreed to appoint Laboratorios Sanfer as the exclusive distributor
of certain of our products in Mexico for the term of the agreement, and an exclusive license to certain of our then-held trademarks.
The term of the agreement is twenty-five years from the effective date of August 15, 2012. The term of the license agreement will
automatically renew after the twenty-five year term for successive two year terms, as long as Laboratorios Sanfer has materially
complied with any and all of the obligations under the license agreement, including but not limited to, meeting the minimum purchase
requirements set forth therein.
“Rest of World”
Through our partner Laboratorios Sanfer, we
market and sell certain of our products within the following countries: Antigua & Barbuda, Argentina, Aruba & Curacao,
Bahamas, Barbados, Belize, Bolivia, Bonaire, Brazil, British Guyana, British Islands, Cayman Islands, Chile, Colombia, Cuba, Dominica,
Dominican Republic, Ecuador, El Salvador, French Guyana, Grenada, Guadalupe, Guatemala, Haiti, Honduras, Jamaica, Martinique, Nicaragua,
Panama, Paraguay, Peru, St. Bartolome, St. Vincent & Grenades, Surinam, Trinidad & Tobago, Turks & Caicos Islands,
Uruguay, Venezuela and Virgin Islands.
Throughout the rest of the world, we use strategic
partners and distributors for the sale of Microcyn®-based products, including Bangladesh, Pakistan, India, the People’s
Republic of China, South Korea, United Arab Emirates, Saudi Arabia, Dubai, Kuwait, Iraq, New Zealand, Singapore, Indonesia and
Malaysia.
Contract Testing
We also operate a microbiology contract testing
laboratory division that provides consulting and laboratory services to medical companies that design and manufacture biomedical
devices and drugs, as well as testing of our products and potential products. Our testing laboratory complies with U.S. Current
Good Manufacturing Practices and Quality Systems Regulations.
Manufacturing and Packaging
We manufacture our products at our facilities
in Petaluma, California and Zapopan, Mexico. We have developed an automated manufacturing process and conduct quality
assurance testing on each production batch in accordance with current U.S., Mexican and international Current Good Manufacturing
Practices. Our facilities are required to meet and maintain regulatory standards applicable to the manufacture of pharmaceutical
and medical device products. Our United States facilities are certified and comply with U.S. Current Good Manufacturing Practices,
Quality Systems Regulations for medical devices, and International Organization for Standardization, or ISO, guidelines. Our Mexico
facility has been approved by the Ministry of Health and is also ISO certified.
Our machines are subjected to a series of tests,
which is part of a validation protocol mandated by U.S., Mexican and international Current Good Manufacturing Practices, Quality
Systems Regulation, and ISO requirements. This validation is designed to ensure that the final product is consistently manufactured
in accordance with product specifications at all manufacturing sites. Certain materials and components used in manufacturing our
machines are proprietary to us.
We believe we have a sufficient number of machines
to produce an adequate amount of Microcyn® to meet anticipated future requirements for at least the next two years. As we expand
into new geographic markets, we may establish additional manufacturing facilities to better serve those new markets.
Intellectual Property
Our success depends in part on our ability
to obtain and maintain proprietary protection for our product technology and know-how, to operate without infringing proprietary
rights of others, and to prevent others from infringing our proprietary rights. We seek to protect our proprietary position by,
among other methods, filing, when possible, U.S. and foreign patent applications relating to our technology, inventions and
improvements that are important to our business. We also rely on trade secrets, know-how, continuing technological innovation,
and in-licensing opportunities to develop and maintain our proprietary position.
As of June 6, 2016, we own a total of 52 issued
patents, consisting of 10 issued U.S. patents and 42 issued foreign patents. We also have 59 pending U.S. and foreign patent
applications. One of the U.S. applications is directed to chlorogenic acid. The remaining patent applications as well as the issued
patents are directed at our Microcyn® Technology. The issued U.S. and foreign patents expire in 2022-2027.
In addition to our own patents and applications,
we have licensed technology developed in Japan relating to an electrolyzed water solution, methods of manufacture and electrolytic
cell designs. This license includes four issued Japanese patents.
Although we work diligently to protect our
technology, we can make no assurances that any patent will be issued from our currently pending patent applications or from future
patent applications. The scope of any patent protection may not exclude competitors or provide competitive advantages to us, and
any of our patents may not be held valid if subsequently challenged, and others may claim rights in or ownership of our patents
and proprietary rights. Furthermore, others may develop products similar to our products and may duplicate any of our products
or design around our patents.
We have also filed for trademark protection
for marks used with our Microcyn® products in each of the following regions: United States, Europe, Canada, certain countries
in Central and South America, including Mexico and Brazil, certain countries in the Middle East and certain countries in Asia,
including Japan, China, Hong Kong, the Republic of Korea, India and Australia. In addition to patents and trademarks, we rely on
trade secret and other intellectual property laws, nondisclosure agreements and other measures to protect our intellectual property
rights. We believe that in order to have a competitive advantage, we must develop and maintain the proprietary aspects of our technologies.
We require our employees, consultants and advisors to execute confidentiality agreements in connection with their employment, consulting
or advisory relationship with us. We also require our employees, consultants and advisors with whom we expect to work on our products
to agree to disclose and assign to us all inventions made in the course of our working relationship with them, while using our
property or which relate to our business. Despite any measures taken to protect our intellectual property, unauthorized parties
may attempt to copy aspects of our products or to wrongfully obtain or use information that we regard as proprietary.
Competition
Dermatology
The dermatology market is highly competitive.
Our dermatology products face competition in the United States from several prescription products, including Novartis’ Elidel®
Cream, a prescription medicine used topically on the skin to treat eczema, which is also called atopic dermatitis, and Astellas’
Protopic®, a prescription ointment used to treat moderate to severe eczema. In addition, corticosteroids are commonly used
to treat inflammation and itch on atopic dermatitis patients as the standard of care. Many doctors and patients will tend to use
steroids for a limited time period to manage flare-ups due to their side effects. Since atopic dermatitis is a chronic disease,
a safe product such as Alevicyn™ , which reduces the itching, can be used as a maintenance product.
Advanced Tissue Care Markets
Competition in the markets for advanced tissue
care is intense. We compete with a number of large, well-established and well-funded companies that sell a broad range of wound
and tissue care products, including topical anti-infectives and antibiotics, as well as some advanced wound technologies, such
as skin substitutes, growth factors and sophisticated delayed release silver-based dressings. We believe the principal competitive
factors in our target market are related to improved patient outcomes, such as shortened time in the hospital, accelerated healing
time, lack of adverse events, safety of products, ease-of-use, stability, pathogen, or disease causing micro-organism, killing
and cost effectiveness.
Our products compete with a variety of products
used for wound cleaning, debriding and moistening, including sterile saline and chlorhexidine-based products. They also compete
with a large number of prescription and over-the-counter products for the prevention and treatment of infections, including topical
anti-infectives, such as Betadine, silver sulfadiazine, hydrogen peroxide, Dakin’s solution and hypochlorous acid, and topical
antibiotics, such as Neosporin, Mupirocin and Bacitracin. Currently, no single anti-infective product dominates the chronic or
acute wound markets because many of the products have serious limitations or tend to inhibit the wound healing process.
Our products can replace the use of saline
for debriding and moistening a dressing and can be used as a complementary product with many advanced tissue care technologies,
such as the Vacuum-Assisted Closure, or V.A.C. Therapy System from Kinetic Concepts Inc., skin substitute products from Smith &
Nephew, Advanced BioHealing, now called Shire Regenerative Medicine, Integra Life Sciences, Life Cell, Organogenesis and Ortec
International, and ultrasound products from Celleration. We believe that Microcyn® Technology can enhance the effectiveness
of many of these advanced tissue care technologies. Because Microcyn® is competitive with some of the large wound care companies’
products and complementary to others, we may compete with such companies in some product lines and complement such companies in
other product lines.
Factors Affecting Our Competitive Position
While many companies are able to produce oxychlorine
formulations, their products, unlike ours, typically become unstable after a relatively short period of time or use very large
ranges of effectiveness to improve their shelf lives. We believe Microcyn® is a stable anti-infective therapeutic available,
or soon to be available, throughout many parts of the world that treats infection while also enhancing wound healing through increased
blood flow to the wound bed and reduction of inflammation.
Some of our competitors in the dermatology,
advanced tissue care markets and animal healthcare enjoy several competitive advantages, including:
|
|
·
|
significantly greater name recognition;
|
|
|
·
|
established relationships with healthcare professionals, patients and third-party payors;
|
|
|
·
|
established distribution networks;
|
|
|
·
|
additional product lines and the ability to offer rebates or bundle products to offer discounts or incentives;
|
|
|
·
|
greater experience in conducting research and development, manufacturing, obtaining regulatory approval for products and marketing; and
|
|
|
·
|
greater financial and human resources for product development, sales and marketing and patient support.
|
Government Regulation
Government authorities in the United States
at the federal, state and local levels and foreign countries extensively regulate, among other things, the research, development,
testing, manufacture, labeling, promotion, advertising, distribution, sampling, marketing, and import and export of pharmaceutical
products, biologics and medical devices. All of our products in development will require regulatory approval or clearance by government
agencies prior to commercialization. In particular, human therapeutic products are subject to rigorous pre-clinical and clinical
trials and other approval procedures of the FDA and similar regulatory authorities in foreign countries. Various federal, state,
local and foreign statutes and regulations also govern testing, manufacturing, safety, labeling, storage, distribution and record-keeping
related to such products and their marketing. The process of obtaining these approvals and clearances, and the subsequent process
of maintaining substantial compliance with appropriate federal, state, local, and foreign statutes and regulations, require the
expenditure of substantial time and financial resources. In addition, statutes, rules, regulations and policies may change and
new legislation or regulations may be issued that could delay such approvals.
Medical Device Regulation
To date, we have received 15 510(k) clearances
for use of our Microcyn® technology products as medical devices in tissue care management, such as cleaning, debridement, lubricating,
moistening and dressing, including for acute and chronic wounds, and in dermatology applications. Any future product candidates
or new applications using Microcyn® that are classified as medical devices will require clearance by the FDA.
Medical devices, such as Microcyn® Wound
Care, are subject to FDA clearance and extensive regulation under the Federal Food Drug and Cosmetic Act. Under the Federal Food
Drug and Cosmetic Act, medical devices are classified into one of three classes: Class I, Class II or Class III. The classification
of a device into one of these three classes generally depends on the degree of risk associated with the medical device and the
extent of control needed to ensure safety and effectiveness. Devices may also be designated unclassified. Unclassified devices
are legally marketed pre-amendment devices for which a classification regulation has yet to be finalized and for which a pre-market
approval is not required.
Class I devices are devices for which safety
and effectiveness can be assured by adherence to a set of general controls. These general controls include compliance with the
applicable portions of the FDA’s Quality System Regulation, which sets forth good manufacturing practice requirements; facility
registration, device listing and product reporting of adverse medical events; truthful and non-misleading labeling; and promotion
of the device only for its cleared or approved intended uses. Class II devices are also subject to these general controls,
and any other special controls as deemed necessary by the FDA to ensure the safety and effectiveness of the device. Review and
clearance by the FDA for these devices is typically accomplished through the 510(k) pre-market notification procedure. When 510(k)
clearance is sought, a sponsor must submit a pre-market notification demonstrating that the proposed device is substantially equivalent
to a legally marketed device. If the FDA agrees that the proposed device is substantially equivalent to the predicate device, then
510(k) clearance to market will be granted. After a device receives 510(k) clearance, any modification that could significantly
affect its safety or effectiveness, or that would constitute a major change in its intended use, requires a new 510(k) clearance
or could require a pre-market approval.
Clinical trials are almost always required
to support a pre-market approval application and are sometimes required for a 510(k) pre-market notification. These trials generally
require submission of an application for an investigational device exemption. An investigational device exemption must be supported
by pre-clinical data, such as animal and laboratory testing results, which show that the device is safe to test in humans and that
the study protocols are scientifically sound. The FDA must approve an investigational device exemption, in advance, for a specified
number of patients, unless the product is deemed a non-significant risk device and is eligible for more abbreviated investigational
device exemption requirements.
Both before and after a medical device is commercially
distributed, manufacturers and marketers of the device have ongoing responsibilities under FDA regulations. The FDA reviews design
and manufacturing practices, labeling and record keeping, and manufacturers’ required reports of adverse experiences and
other information to identify potential problems with marketed medical devices. Device manufacturers are subject to periodic and
unannounced inspection by the FDA for compliance with the Quality System Regulation, which sets forth the Current Good Manufacturing
Practice requirements that govern the methods used in, and the facilities and controls used for, the design, manufacture, packaging,
servicing, labeling, storage, installation and distribution of all finished medical devices intended for human use.
FDA regulations prohibit the advertising and
promotion of a medical device for any use outside the scope of a 510(k) clearance or pre-market approval or for unsupported safety
or effectiveness claims. Although the FDA does not regulate physicians’ practice of medicine, the FDA does regulate manufacturer
communications with respect to off-label use.
If the FDA finds that a manufacturer has failed
to comply with FDA laws and regulations or that a medical device is ineffective or poses an unreasonable health risk, it can institute
or seek a wide variety of enforcement actions and remedies, ranging from a public warning letter to more severe actions such as:
|
|
·
|
imposing fines, injunctions and civil penalties;
|
|
|
·
|
requiring a recall or seizure of products;
|
|
|
·
|
implementing operating restrictions, which can include a partial suspension or total shutdown of production;
|
|
|
·
|
refusing requests for 510(k) clearance or pre-market approval of new products;
|
|
|
·
|
withdrawing 510(k) clearance or pre-market approval approvals already granted; and
|
The FDA also has the authority to require a
company to repair, replace, or refund the cost of any medical device.
The FDA also administers certain controls over
the export of medical devices from the United States, as international sales of medical devices that have not received FDA clearance
are subject to FDA export requirements. Additionally, each foreign country subjects such medical devices to its own regulatory
requirements. In the European Union, there is a single regulatory approval process and approval is represented by the presence
of a CE Mark.
Other Regulation in the United States
The Physician Payments Sunshine Act
The Physician Payments Sunshine Act signed
into law in 2010 as part of the Affordable Care Act requires manufacturers of medical devices, drugs, biologicals, and medical
supplies to track and report certain payments made to and transfers of value provided to physicians and teaching hospitals as well
as to report certain ownership and investment interests held by physicians and their immediate family members. These manufacturers
must report annually to the Center for Medicare & Medicaid Services any direct or indirect payments and transfers of value
of $10 or more, or annual aggregate of $100 or more, made to physicians or to a third party at the request of or on behalf of a
physician, including dentists. Payment includes: consulting fees, compensation for services other than consulting, honoraria, gifts,
entertainment, food, travel (including the specified destinations), education, research, charitable contribution, royalty or license,
current or prospective ownership or investment interest, direct compensation for serving as faculty or as a speaker for a medical
education program, grants, any other nature of the payment, or other transfer of value. Manufacturers face monetary penalties for
non-compliance. Certain payments related to research must be reported separately. Product samples intended for patient use need
not be reported.
Health Care Coverage and Reimbursement by
Third-Party Payors
Commercial success in marketing and selling
our products depends, in part, on the availability of adequate coverage and reimbursement from third-party health care payors,
such as government and private health insurers and managed care organizations. Third-party payors are increasingly challenging
the pricing of medical products and services. Government and private sector initiatives to limit the growth of health care costs,
including price regulation, competitive pricing, and managed-care arrangements, are continuing in many countries where we do business,
including the United States. These changes are causing the marketplace to be more cost-conscious and focused on the delivery of
more cost-effective medical products. Government programs, including Medicare and Medicaid, private health care insurance companies,
and managed-care plans control costs by limiting coverage and the amount of reimbursement for particular procedures or treatments.
This has created an increasing level of price sensitivity among customers for our products. Some third-party payors also require
that a favorable coverage determination be made for new or innovative medical devices or therapies before they will provide reimbursement
of those medical devices or therapies. Even though a new medical product may have been cleared or approved for commercial distribution,
we may find limited demand for the product until adequate coverage and reimbursement have been obtained from governmental and other
third-party payors.
Fraud and Abuse Laws
In the United States, we are subject to various
federal and state laws pertaining to healthcare fraud and abuse, which, among other things, prohibit the offer or acceptance of
remuneration intended to induce or in exchange for the purchase of products or services reimbursed under a federal healthcare program
and the submission of false or fraudulent claims with the government. These laws include the federal Anti-Kickback Statute, the
False Claims Act and comparable state laws. These laws regulate the activities of entities involved in the healthcare industry,
such as us, by limiting the kinds of financial arrangements such entities may have with healthcare providers who use or recommend
the use of medical products, including, for example, sales and marketing programs, advisory boards and research and educational
grants. In addition, in order to ensure that healthcare entities comply with healthcare laws, the Office of Inspector General of
the U.S. Department of Health and Human Services recommends that healthcare entities institute effective compliance programs.
To assist in the development of effective compliance programs, the Office of Inspector General has issued model Compliance Program
Guidance, materials for a variety of healthcare entities which, among other things, identify practices to avoid that may implicate
the federal Anti-Kickback Statute and other relevant laws and describes elements of an effective compliance program. While compliance
with the Compliance Program Guidance materials is voluntary, a California law requires pharmaceutical and devices manufacturers
to initiate compliance programs that incorporate the Compliance Program Guidance and the July 2002 Pharmaceuticals Research and
Manufacturers of America Code on Interactions with Healthcare Professionals.
Due to the scope and breadth of the provisions
of some of these laws, it is possible that some of our practices might be challenged by the government under one or more of these
laws in the future. Violations of these laws, which are discussed more fully below, can lead to civil and criminal penalties, damages,
imprisonment, fines, exclusion from participation in Medicare, Medicaid and other federal health care programs, and the curtailment
or restructuring of our operations. Any such violations could have a material adverse effect on our business, financial condition,
results of operations or cash flows.
Anti-Kickback Laws
Our operations are subject to federal and state
anti-kickback laws. The federal Anti-Kickback Statute prohibits persons from knowingly and willfully soliciting, receiving, offering
or providing remuneration directly or indirectly to induce either the referral of an individual for a good or service reimbursed
under a federal healthcare program, or the furnishing, recommending, or arranging of a good or service, for which payment may be
made under a federal healthcare program, such as Medicare or Medicaid. The definition of “remuneration” has been broadly
interpreted to include anything of value, including such items as gifts, discounts, the furnishing of supplies or equipment, waiver
of co-payments, and providing anything at less than its fair market value. Because the Anti-Kickback Statute makes illegal a wide
variety of common, even beneficial, business arrangements, the Office of Inspector General was tasked with issuing regulations,
commonly known as “safe harbors,” that describe arrangements where the risk of illegal remuneration is minimal. As
long as all of the requirements of a particular safe harbor are strictly met, the entity engaging in that activity will not be
prosecuted under the federal Anti-Kickback Statute. The failure of a transaction or arrangement to fit precisely within one or
more safe harbors does not necessarily mean that it is illegal or that prosecution will be pursued. However, business arrangements
that do not fully satisfy an applicable safe harbor may result in increased scrutiny by government enforcement authorities, such
as the Office of Inspector General. Our agreements to pay compensation to our advisory board members and physicians who provide
other services for us may be subject to challenge to the extent they do not fall within relevant safe harbors under state and federal
anti-kickback laws. In addition, many states have adopted laws similar to the federal Anti-Kickback Statute, which apply to the
referral of patients for health care services reimbursed by Medicaid, and some have adopted such laws with respect to private insurance.
Violations of the Anti-Kickback Statute are subject to significant fines and penalties and may lead to a company being excluded
from participating in federal health care programs.
False Claims Laws
The federal False Claims Act prohibits knowingly
filing a false claim, knowingly causing the filing of a false claim, or knowingly using false statements to obtain payment from
the federal government. Under the False Claims Act, such suits are known as “qui tam” actions. Individuals may file
suit on behalf of the government and share in any amounts received by the government pursuant to a settlement. In addition, certain
states have enacted laws modeled after the federal False Claims Act under the Deficit Reduction Act of 2005, where the federal
government created financial incentives for states to enact false claims laws consistent with the federal False Claims Act. As
more states enact such laws, we expect the number of qui tam lawsuits to increase. Qui tam actions have increased significantly
in recent years, causing greater numbers of healthcare companies to have to defend false claims actions, pay fines or be excluded
from Medicare, Medicaid or other federal or state government healthcare programs as a result of investigations arising out of such
actions.
HIPAA
Two federal crimes were created under the Health
Insurance Portability and Accountability Act of 1996, or HIPAA: healthcare fraud and false statements relating to healthcare matters.
The healthcare fraud statute prohibits knowingly and willfully executing a scheme to defraud any healthcare benefit program, including
private payors. The false statements statute prohibits knowingly and willfully falsifying, concealing or covering up a material
fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for healthcare
benefits, items or services.
Health Information Privacy and Security
Individually, identifiable health information
is subject to an array of federal and state regulation. Federal rules promulgated pursuant to HIPAA regulate the use and disclosure
of health information by “covered entities.” Covered entities include individual and institutional health care providers
from which we may receive individually identifiable health information. These regulations govern, among other things, the use and
disclosure of health information for research purposes, and require the covered entity to obtain the written authorization of the
individual before using or disclosing health information for research. Failure of the covered entity to obtain such authorization
could subject the covered entity to civil and criminal penalties. We may experience delays and complex negotiations as we deal
with each entity’s differing interpretation of the regulations and what is required for compliance. Also, where our customers
or contractors are covered entities, including hospitals, universities, physicians or clinics, we may be required by the HIPAA
regulations to enter into “business associate” agreements that subject us to certain privacy and security requirements.
In addition, many states have laws that apply to the use and disclosure of health information, and these laws could also affect
the manner in which we conduct our research and other aspects of our business. Such state laws are not preempted by the federal
privacy law when such laws afford greater privacy protection to the individual than the federal law. While activities to assure
compliance with health information privacy laws are a routine business practice, we are unable to predict the extent to which our
resources may be diverted in the event of an investigation or enforcement action with respect to such laws.
Foreign Regulation
Whether or not we obtain FDA approval for a
product, we must obtain approval of a product by the applicable regulatory authorities of foreign countries before we can commence
clinical trials or marketing of the product in those countries. The approval process varies from country to country, and the time
may be longer or shorter than that required for FDA approval. The requirements governing the conduct of clinical trials, product
licensing, pricing and reimbursement also vary greatly from country to country. Although governed by the applicable country, clinical
trials conducted outside of the United States typically are administered under a three-phase sequential process similar to that
discussed above for medical devices.
European Union Regulation
Medical Device Regulation
Our products are classified as medical devices
in the European Union. In order to sell our medical device products within the European Union, we are required to comply with the
requirements of the Medical Devices Directive, and its national implementations, including affixing CE markings on our products.
The CE marking indicates a product’s compliance with EU legislation and so enables the sale of products throughout the European
Economic Area (EEA, the 28 Member States of the EU and European Free Trade Association (EFTA) countries Iceland, Norway, Liechtenstein).
In order to comply with the Medical Devices Directive, we must meet certain requirements relating to the safety and performance
of our products and, prior to marketing our products, we must successfully undergo verification of our products’ regulatory
compliance, or conformity assessment.
Medical devices are divided into three regulatory
classes: Class I, Class IIB and Class III. The nature of the conformity assessment procedures depends on the regulatory
class of the product. In order to comply with the examination, we completed, among other things, a risk analysis and presented
clinical data, which demonstrated that our products met the performance specifications claimed by us, provided sufficient evidence
of adequate assessment of unwanted side effects and demonstrated that the benefits to the patient outweigh the risks associated
with the device. We are subject to continued supervision and are required to report any serious adverse incidents to the appropriate
authorities. We are also required to comply with additional national requirements that are beyond the scope of the Medical Devices
Directive.
We received a CE certificate for 25 of our
Class IIB medical devices, which allows us to affix CE markings on these products and sell them in Europe. The CE certificate is
valid through December 2018. Currently, the European Commission and the European Parliament are discussing changes to the Medical
Devices Directive which could include stricter requirements for obtaining CE markings or continued compliance. We may not be able
to maintain the requirements established for CE markings for any or all of our products or be able to produce these products in
a timely and profitable manner while complying with the requirements of the Medical Devices Directive and other regulatory requirements.
Marketing Authorizations for Drugs
In order to obtain marketing approval of any
of our drug products in Europe, we must submit for review an application similar to a U.S. new drug application to the relevant
authority. In contrast to the United States, where the FDA is the only authority that administers and approves new drug applications,
in Europe there are multiple authorities that administer and approve these applications. Marketing Authorizations in Europe expire
after five years but may be renewed.
We believe that any drug candidate will be
reviewed by the Committee for Medicinal Products for Human Use, on behalf of the European Medicines Agency. Based upon the review
of the Committee for Medicinal Products for Human Use, the European Medicines Agency provides an opinion to the European Commission
on the safety, quality and efficacy of the drug. The decision to grant or refuse an authorization is made by the European Commission.
Approval of Marketing Applications can take
several months to several years, or may be denied. This approval process can be affected by many of the same factors relating to
safety, quality and efficacy as in the approval process for new drug applications in the United States. As in the United States,
European drug regulatory authorities can require us to perform additional non-clinical studies and clinical trials. The need for
such studies or trials, if imposed, may delay marketing approval and involve unanticipated costs. Inspection of clinical investigation
sites by a competent authority may also be required as part of the regulatory approval procedure. In addition, as a condition of
marketing approval, regulatory agencies in Europe may require post-marketing surveillance to monitor for adverse effects, or other
additional studies may be required as deemed appropriate. The terms of any approval, including labeling content, may be more restrictive
than expected and could affect the marketability of a product. In addition, after approval for the initial indication, further
clinical studies are usually necessary to gain approval for any additional indications.
European Good Manufacturing Process
In the European Union, the manufacture of pharmaceutical
products and clinical trial supplies is subject to good manufacturing practice as set forth in the relevant laws and guidelines.
Compliance with good manufacturing practice is generally assessed by the competent regulatory authorities. They may conduct inspections
of relevant facilities, and review manufacturing procedures, operating systems and personnel qualifications. In addition to obtaining
approval for each product, in many cases each drug manufacturing facility must be approved. Further inspections may occur over
the life of the product.
Mexican Regulation
The Ministry of Health is the authority in
charge of sanitary controls in Mexico. Sanitary controls are a group of practices related to the orientation, education, testing,
verification and application of security measures and sanctions exercised by the Ministry of Health. The Ministry of Health is
responsible for the issuance of Official Mexican Standards and specifications for drugs subject to the provisions of the General
Health Law, which govern the process and specifications of drugs, including the obtaining, preparing, manufacturing, maintaining,
mixing, conditioning, packaging, handling, transporting, distributing, storing and supplying of products to the public at large.
In addition, a medical device is defined as a device that may contain antiseptics or germicides used in surgical practice or in
the treatment of continuity solutions, skin injuries or its attachments.
Regulations applicable to medical devices and
drugs are divided into two sections: the business that manufactures the medical device or drug and the product itself.
Manufacturing a Medical Device or Drug
Under the General Health Law, a business that
manufactures drugs is either required to obtain a “Sanitary Authorization” or to file an “Operating Notice.”
Our Mexico subsidiary, Oculus Technologies of Mexico, S.A. de C.V., is considered a business that manufactures medical devices
and therefore is not subject to a Sanitary Authorization, but rather only to file an Operating Notice.
In addition to its Operating Notice, our Mexico
subsidiary has obtained a “Good Processing Practices Certificate” issued by Mexican Federal Commission for the Protection
against Sanitary Risks, which demonstrates that the manufacturing of Microcyn® at the facility located in Zapopan, Mexico,
operates in accordance with the applicable official standards.
Commercialization of Drugs and Medical Devices
Drugs and medical devices should be commercialized
in appropriate packaging containing labels printed in accordance with specific official standards. For medical devices, there are
no specific standards or regulations related to the labeling of the product, but rather only a general standard related to the
labeling for all types of products to be commercialized in Mexico. Advertising of medical devices is regulated in the General Health
Law and in the specific regulations of the General Health Law related to advertising. Generally, the advertising of medical devices
is subject to a permit only in the case that such advertising is directed to the general public.
Medical Devices and Drugs as a Product
To produce, sell or distribute medical devices,
a Sanitary Registry is required in accordance with the General Health Law and the Regulation for Drugs. Such registry is granted
for a term of five years, and this term may be extended. The Sanitary Registry may be revoked if the interested party does not
request the extension in the term or the product or the manufacturer or the raw material is changed without the permission of the
Ministry of Health.
The Ministry of Health classifies the medical
devices in three classes:
|
|
·
|
Class I.
Devices for which safety and effectiveness have been duly proved and are generally not used inside the body;
|
|
|
·
|
Class II.
Devices that may vary with respect to the material used for its fabrication or in its concentration and generally used inside of the body for a period no greater than 30 days; and
|
|
|
·
|
Class III.
New devices or recently approved devices in the medical practice or those used inside the body and which shall remain inside the body for a period greater than 30 days.
|
Currently, we have 14 approvals from the Mexican
Ministry of Health to market and distribute our products in Mexico.
Violation of these regulations may result in
the revocation of the registrations or approvals, and economic fines. In some cases, such violations may constitute criminal actions.
In addition, regulatory approval of prices
is required in most countries other than the United States, which could result in lengthy negotiations delaying our ability to
commercialize our products. We face the risk that the prices which result from the regulatory approval process would be insufficient
to generate an acceptable return.
Research and Development
Research and development expense consists primarily
of personnel expenses, clinical and regulatory services and supplies. For the years ended March 31, 2016 and 2015, research and
development expense amounted to $1,806,000 and $1,533,000, respectively. None of these expenses were borne by our customers.
Significant Customers
We rely on certain key customers for a significant
portion of our revenues. At March 31, 2016, one customer represented 33% of the net accounts receivable balance. At March 31, 2016,
one customer represented 40%, one customer represented 15%, one customer represented 14% and two customers each represented 12%
of net revenues. At March 31, 2015, one customer represented 56%, and one customer represented 14% of the net accounts receivable
balance. During the year ended March 31, 2015, one customer represented 47% of net revenues.
Our Employees
As of March 31, 2016, we employed a total
of 38 employees in the United States and the Netherlands, 37 of which were full-time. Additionally, we had 168 employees in Mexico,
all of which were contracted through an employment agency. As of March 31, 2016, we had a U.S. direct sales force of 19 employees.
We are not a party to any collective bargaining agreements. We believe our relations with our employees are good.
Description
of Property
We currently lease the following material properties:
|
Location
|
|
Rent per month
|
|
Purpose
|
|
1129 N. McDowell Blvd., Petaluma, CA 94954, USA
|
|
USD 11,072
|
|
Principal executive office, also used for research and manufacturing
|
|
454 North 34th Street, Seattle, Wash. 98103, USA
|
|
USD 2,700
|
|
Shared office and laboratory space
|
|
Suite 130, First Floor, 2500 York Road, Jamison, PA 18929
|
|
USD 2,126
|
|
Office
|
|
Industria Vidriera 81, Zapopan Industrial Norte, Zapopan, Jalisco, 45132, Mexico
|
|
MXN 121,395
|
|
Office, manufacturing, storage
|
|
Industria Maderera 124 & 106 & 815 Zapopan Industrial Norte, Zapopan, Jalisco, 45132, Mexico
|
|
MXN 124,500
|
|
Storage
|
|
Nusterweg 123, 6136 ST Sittard (gemeente Sittard, sectie K, nummers 2765 en 2778) Herten, the Netherlands
|
|
Euro 6,250
|
|
Office
|
As we expand, we may need to establish manufacturing
facilities in other countries. We believe that our properties will be adequate to meet our needs for at least the next 12 months.
Legal Proceedings
On occasion, we may be involved in legal matters
arising in the ordinary course of our business including matters involving proprietary technology. While management believes that
such matters are currently insignificant, matters arising in the ordinary course of business for which we are or could become involved
in litigation may have a material adverse effect on our business, financial condition or results of comprehensive loss.
MANAGEMENT
Directors
At our 2008 Annual Meeting of Stockholders,
our stockholders approved an amendment to our Restated Certificate of Incorporation, as amended, which provided that directors
are classified into three classes, as nearly equal in number as possible, with each class serving for a staggered three-year term.
Our Board currently consists of five directors:
|
|
Name
|
Age
|
Position with the Company
|
Director Since
|
Term Expires
|
|
Class I
|
|
|
|
|
|
|
|
Sharon Barbari
|
62
|
Class I Director
|
03/2014
|
2018
|
|
|
Russell Harrison
|
71
|
Class I Director
|
02/2014
|
2018
|
|
Class II
|
|
|
|
|
|
|
|
Jay Birnbaum
|
71
|
Class II Director
|
04/2007
|
2016
|
|
|
Jim Schutz
|
53
|
Chief Executive Officer and Class II Director
|
05/2004
|
2016
|
|
Class III
|
|
|
|
|
|
|
|
Jerry McLaughlin
|
68
|
Class III Director
|
03/2013
|
2017
|
The biographies of our directors and certain
information regarding each director’s experience, attributes, skills and/or qualifications that led to the conclusion that
the director should be serving as a director of Oculus are stated below.
Sharon Barbari
: Ms. Barbari has served
as Executive Vice President of Finance and Chief Financial Officer of Cytokinetics Inc. since July 2009. She served as Senior Vice
President of Finance and Chief Financial Officer from September 2004 through June 2009. From September 2002 to August 2004, Ms.
Barbari served as Chief Financial Officer and Senior Vice President of Finance and Administration of InterMune, Inc., a biopharmaceutical
company. From January 1998 to June 2002, she served at Gilead Sciences, Inc., a biopharmaceutical company, and held several positions
of increasing responsibility including most recently as its Vice President and Chief Financial Officer. From 1996 to 1998, Ms.
Barbari served as Vice President of Strategic Planning at Foote, Cone & Belding Healthcare in San Francisco, an international
advertising and marketing firm. From 1972 to 1995, she was employed by Syntex Corporation where she held various management positions
in corporate finance, financial planning, marketing and commercial planning.
Russell Harrison
: Mr. Harrison has served
on our Board of Directors since February 2014. Mr. Harrison is the founding principal of The Leadership Group, LLC, a firm specializing
in strategic change and executive coaching for U.S. and international companies, a position he has held since January 2012. From
2006 to 2012, he was employed by CoolSystems, Inc. d/b/a Game Ready, Inc. and was appointed as its President and Chief Executive
Officer in 2007. Mr. Harrison has also served in the role of Chief Executive Officer for a number of publicly traded and private
technology businesses in both the information technology and medical device technology sectors. From 1995 to 1997, he served as
Chief Information Officer at SITA Telecommunications Holdings in Paris, where he led a team responsible for technology spanning
more than 200 countries and territories. From 1991 to 1993, Mr. Harrison served as the first Chief Information Officer for McKesson
Corporation, responsible for all computer technology-related activities. He also served as a Captain in the United States Marine
Corps.
Jay Birnbaum
: Dr. Birnbaum is a pharmacologist
and since 1999, has been a consultant to pharmaceutical companies in his area of expertise. He previously served as Vice President
of Global Project Management at Novartis/Sandoz Pharmaceuticals Corporation, where he had responsibility for strategic planning
and development of the company’s dermatology portfolio. Dr. Birnbaum is also a co-founder and former Chief Medical Officer
of Kythera Biopharmaceuticals, and has served on the board of directors of Excaliard Pharmaceuticals (a company recently acquired
by Pfizer) and on the scientific advisory boards of several companies. During 2015, Dr. Birnbaum co-founded Hallux, Inc. and serves
as Hallux’ Chief Scientific Officer and member of the Board of Directors.
Jim Schutz
: Mr. Schutz was appointed
our President and Chief Executive Officer on February 4, 2013. Prior to this appointment, he most recently held the position of
our Chief Operating Officer and General Counsel, and has served in various other capacities as an executive officer of our Company
since August 2003. From August 2001 to August 2003, Mr. Schutz served as General Counsel at Jomed (formerly EndoSonic Corp.), an
international medical device company. From 1999 to July 2001, Mr. Schutz served as in-house counsel at Urban Media Communications
Corporation, an internet/telecom company based in Palo Alto, California.
Jerry McLaughlin
: Mr. McLaughlin served
as Interim Chief Executive Officer of Applied BioCode, Inc. from November 2011 to April 2013. In April 2011, he also founded and
currently serves as Chairman and Chief Executive Officer, of DataStream Medical Imaging Systems, Inc., a start-up to develop diagnostic
imaging software applications that work in conjunction with existing digital radiology platforms. He previously served as President
of DataFlow Information Systems, from July 2007 to December 2011, and President and Chief Executive Officer of CompuMed, Inc. from
May 2002 to June 2007. Mr. McLaughlin also serves on the board of directors of DataStream Medical Imaging Systems, Inc., a private
company in the medical imaging software industry.
Committees of the Board of Directors
Our
Board of Directors has appointed an Audit Committee, a Compensation Committee, and a Nominating and Corporate Governance Committee.
The Board of Directors has determined that each director who serves on these committees is “independent,” as that term
is defined by the NASDAQ Listing Rules and rules of the SEC. The Board of Directors has adopted written charters for its Audit
Committee, its Compensation Committee and its Nominating and Corporate Governance Committee. Copies of these charters are available
on our website at http://ir.oculusis.com/governance.cfm. In addition to the number of meetings referenced below, the Committees
also took actions by unanimous written consent.
Information
about each of our committees is stated below
|
Name of Committee Member
|
Audit
|
Compensation
|
Nominating and Corporate Governance
|
|
Sharon Barbari
|
C
|
|
M
|
|
Jerry McLaughlin
|
|
C
|
|
|
Russell Harrison
|
M
|
M
|
C
|
|
Jay Birnbaum
|
M
|
|
|
C =
Committee Chair
M =
Member
Director Independence
We determine independence using the definitions
set forth in the NASDAQ Listing Rules and the rules under the Securities Exchange Act of 1934. The Board has determined that the
following directors are independent:
It is our policy that all employees, officers
and directors must avoid any activity that is, or has the appearance of, conflicting with the interests of our Company. This policy
is included in our Code of Business Conduct, and our Board formally adopted a Related Party Transaction Policy and Procedures in
July 2007 for the approval of interested transactions with persons who are Board members or nominees, executive officers, holders
of 5% of our common stock, or family members of any of the foregoing. The Related Party Transaction Policy and Procedures are administered
by our Audit Committee. We conduct a review of all related party transactions for potential conflict of interest situations on
an ongoing basis and all such transactions relating to executive officers and directors must be approved by the Audit Committee.
Director Compensation
The following table sets forth the amounts
and the value of compensation earned or paid to our directors for their service in fiscal year 2016. All share numbers have been
adjusted for the 1-for-5 reverse stock split effective June 24, 2016.
|
Name of Director
|
Fees Earned or
Paid in Cash
($) (1)
|
Stock Awards
($) (2) (3) (4)
|
Option Awards
($) (4) (5)
|
All Other
Compensation
($) (6)
|
Total
($)
|
|
Jim Schutz (7)
|
0
|
0
|
0
|
0
|
0
|
|
Russell Harrison
(8)
|
34,063
|
21,010
|
14,942
|
8,405
|
78,420
|
|
Sharon Barbari (9)
|
34,063
|
19,761
|
13,053
|
7,905
|
74,782
|
|
Jay Birnbaum (10)
|
29,688
|
19,256
|
12,297
|
7,705
|
68,946
|
|
Jerry McLaughlin (11)
|
32,813
|
24,258
|
19,863
|
9,705
|
86,639
|
|
|
(1)
|
The table omits the columns, non-equity incentive plan compensation, change
in pension value and nonqualified deferred compensation earnings because our directors did not receive any of these compensation
items in fiscal year 2016.
|
|
|
(2)
|
Represents the aggregate grant date fair value of stock awards granted
during the fiscal year ended March 31, 2016, as computed in accordance with FASB ASC Topic 718, Compensation – Stock Compensation.
The fair value of each stock award is determined on the date of grant by taking the number of shares of common stock granted multiplied
by the closing price of the common stock on the NASDAQ Capital Market on the day of grant.
|
|
|
(3)
|
Pursuant to our non-employee director compensation plan, on the last business
day of the month of February, each non-employee director is automatically granted 2,250 shares of our common stock, provided that
no annual grant is granted to a non-employee director in the same calendar year that such person received his or her initial grant.
The annual stock grants are subject to a 40% tax gross up granted as an additional cash payment equal to 0.40 multiplied by the
number of shares of common stock granted multiplied by the closing price of the common stock on the NASDAQ Capital Market on the
day of the grant.
|
|
|
(4)
|
Pursuant to our non-employee director compensation plan effective March
26, 2014, as amended, non-employee directors may elect to receive any fees, except for Audit Committee fees, in one of three ways
at the director’s election: (1) in cash; (2) in options, or (3) as a stock grant. Options to purchase shares of our common
stock vest immediately and the exercise price for such options shall be the closing price of our stock on the date such options
are granted. Stock grants vest immediately and the actual number of shares underlying the stock grant is determined by dividing
the amount of the award due in cash by the closing price of our stock on the NASDAQ Capital Market on the day of the grant. All
of our directors elected to receive a portion of their retainer in stock or stock options for the year ended March 31, 2016, as
indicated in the table below. The table includes options granted on June 1, 2015, for services as a director during the period
of January 1 to March 31, 2015.
|
|
Name of Director
|
Amount in $
|
Number of
Shares
received in
Lieu of
Cash
|
Number of
Options
received in
Lieu of
Cash
|
|
Russell Harrison
|
24,688
|
2,015
|
3,891
|
|
Sharon Barbari
|
21,563
|
1,760
|
3,399
|
|
Jay Birnbaum
|
20,313
|
1,657
|
3,202
|
|
Jerry McLaughlin
|
32,813
|
2,678
|
5,173
|
|
|
|
As of March 31, 2016, our directors had the following aggregate numbers
of granted and outstanding options, respectively: Mr. Schutz – 48,481, Mr. Harrison – 17,893, Ms. Barbari – 16,192,
Mr. Birnbaum – 34,798, and Mr. McLaughlin – 29,224.
|
|
|
(5)
|
Represents the aggregate grant date fair value of stock option awards granted
during the fiscal year ended March 31, 2016, as computed in accordance with FASB ASC Topic 718, Compensation — Stock Compensation.
The fair value of each stock option award is estimated for the fiscal year ended March 31, 2016, on the date of grant using the
Black-Scholes option valuation model. A discussion of the assumptions used in calculating the amounts in this column may be found
in Note 14 to our audited consolidated financial statements for the year ended March 31, 2016, included in our Annual Report on
Form 10-K filed with the SEC on June 21, 2016. These amounts do not represent the actual amounts paid to or realized by the directors
during the fiscal year ended March 31, 2016.
|
|
|
(6)
|
Pursuant to our non-employee director compensation program, all elective
and automatic stock grants are subject to a 40% tax gross up granted as an additional cash payment equal to 0.40 multiplied by
the number of shares of common stock granted multiplied by the closing price of the common stock on the NASDAQ Capital Market on
the day of the grant.
|
|
|
(7)
|
As a Company employee, Mr. Schutz did not receive compensation for his
service as a director during fiscal year ended March 31, 2016.
|
|
|
(8)
|
For the fiscal year ended in March 31, 2016, Mr. Harrison earned $47,000
for his services as a director. Of this aggregate amount of $47,000, Mr. Harrison received $32,500 as his annual retainer for serving
on the Board, $7,500 for his services as a member of the Audit Committee, $2,000, for his services as a member of the Compensation
Committee and $5,000, for his services as the chairperson of the Nominations and Corporate Governance Committee. Other than the
retainer for his services as a member of the Audit Committee, which must be paid in cash, Mr. Harrison elected to receive 50% of
the aggregate retainer owed for his services as a director in stock options in lieu of cash for the quarters ended June 30, 2015,
and September 31, 2015. For the quarters ended December 31, 2015, and March 31, 2016, Mr. Harrison elected to receive 50% of the
aggregate retainer owed for his services as a director, other than the retainer for his services as a member of the Audit Committee,
in common stock in lieu of cash. Pursuant to the Director Compensation Program, we paid an additional $3,950 in cash representing
the 40% tax gross up owed Mr. Harrison for all fees paid in stock. Additionally, pursuant to the non-employee director compensation
plan, Mr. Harrison received an annual grant in February 2016 consisting of 2,250 shares of common stock and $4,455 in cash, representing
the 40% tax gross up. The table reflects a payment of $6,813 in cash and $4,938 in common stock made in June 2016 for service during
the period of January 1, 2016 through March 31, 2016, and a payment of $6,813 in cash and $4,938 in options made in June 2015 for
services during the period January 1 to March 31, 2015.
|
|
|
(9)
|
For the fiscal year ended in March 31, 2016, Ms. Barbari earned $44,500
for her services as a director. Of this aggregate amount of $44,500, Ms. Barbari received $32,500 as her annual retainer for serving
on the Board, $10,000 for her services as the chairperson of the Audit Committee, $2,000, for her services as a member of the Nominating
and Corporate Governance Committee. Other than the retainer for her services as the chairperson of the Audit Committee, which must
be paid in cash, Ms. Barbari elected to receive 50% of the aggregate retainer owed for her services as a director in stock options
in lieu of cash for the quarters ended June 30, 2015, and September 31, 2015. For the quarters ended December 31, 2015, and March
31, 2016, Ms. Barbari elected to receive 50% of the aggregate retainer owed for her services as a director, other than the retainer
for her services as the chairperson of the Audit Committee, in common stock in lieu of cash. Pursuant to the Director Compensation
Program we paid an additional $3,450 in cash representing the 40% tax gross up owed Ms. Barbari for all fees paid in stock. Additionally,
pursuant to the non-employee director compensation plan, Ms. Barbari received an annual grant in February 2016 consisting of 2,250
shares of common stock and $4,455 in cash, representing the 40% tax gross up. The retainer fee reflects a payment of $6,813 in
cash and $4,313 in common stock made in June 2016 for service during the period of January 1, 2016 through March 31, 2016, and
a payment of $6,813 in cash and $4,313 in options made in June 2015 for services during the period January 1 to March 31, 2015.
|
|
|
(10)
|
For the fiscal year ended in March 31, 2016, Mr. Birnbaum earned $40,000
for his services as a director. Of this aggregate amount of $40,000, Mr. Birnbaum received $32,500 as his annual retainer for serving
on the Board, $7,500 for his services as a member of the Audit Committee. Other than the retainer for his services as a member
of the Audit Committee, which must be paid in cash, Mr. Birnbaum elected to receive 50% of the aggregate retainer owed for his
services as a director in stock options in lieu of cash for the quarters ended June 30, 2015, and September 31, 2015. For the quarters
ended December 31, 2015, and March 31, 2016, Mr. Birnbaum elected to receive 50% of the aggregate retainer owed for his services
as a director, other than the retainer for his services as a member of the Audit Committee, in common stock in lieu of cash. Pursuant
to the Director Compensation Program we paid an additional $3,250 in cash representing the 40% tax gross up owed Mr. Birnbaum for
all fees paid in stock. Additionally, pursuant to the non-employee director compensation plan, Mr. Birnbaum received an annual
grant in February 2016 consisting of 2,250 shares of common stock and $4,455 in cash, representing the 40% tax gross up. The retainer
fee reflects a payment of $5,938 in cash and $4,063 in common stock made in June 2016 for service during the period of January
1, 2016 through March 31, 2016, and a payment of $5,938 in cash and $4,063 in options made in June 2015 for services during the
period January 1 to March 31, 2015.
|
|
|
(11)
|
For the fiscal year ended in March 31, 2016, Mr. McLaughlin earned $52,500
for his services as a director. Of this aggregate amount of $52,500, Mr. McLaughlin received $32,500 as his annual retainer for
serving on the Board, $15,000 for his annual retainer as the Lead Independent Director and $5,000, for his services as the chairperson
of the Compensation Committee. Mr. McLaughlin elected to receive 50% of the aggregate retainer owed for his services as a director
in stock options in lieu of cash for the quarters ended June 30, 2015, and September 31, 2015. For the quarters ended December
31, 2015, and March 31, 2016, Mr. McLaughlin elected to receive 50% of the aggregate retainer owed for his services as a director
in common stock in lieu of cash. Pursuant to the Director Compensation Program we paid an additional $5,249 in cash representing
the 40% tax gross up owed to Mr. McLaughlin for all fees paid in stock. Additionally, pursuant to the non-employee director compensation
plan, Mr. McLaughlin received an annual grant in February 2016 consisting of 2,250 shares of common stock and $4,455 in cash, representing
the 40% tax gross up. The retainer fee reflects a payment of $6,563 in cash and $6,563 in common stock made in June 2016 for service
during the period of January 1, 2016 through March 31, 2016, and a payment of $6,563 in cash and $6,563 in options made in June
2015 for services during the period January 1 to March 31, 2015.
|
Narrative to Director Compensation Table
All share numbers have been adjusted for the 1–for–5
reverse stock split effective June 24, 2016.
Non-Employee
Director Compensation Plan Adopted February 24, 2016
Pursuant to our restated and amended
non-employee director compensation plan adopted February 24, 2016, as of April 1, 2016, each non-employee director is entitled
to the following annual retainers:
|
·
Board Member
|
$32,500
|
|
·
Lead Independent Director
|
$15,000
|
|
·
Chair of the Audit Committee
|
$10,000
|
|
·
Chair of the Compensation Committee
|
$7,500
|
|
·
Chair of the Nominating and Corporate Governance Committee
|
$7,500
|
|
·
Audit Committee member (other than Chair)
|
$7,500
|
|
·
Compensation Committee Member (other than Chair)
|
$7,500
|
|
·
Nominating and Corporate Governance Committee Member (other than the Chair)
|
$7,500
|
All Audit
Committee retainers must be paid in cash. All other retainers may be paid in (i) cash, (ii) options or (iii) as a stock grant with
an additional cash payment equal to 0.40 multiplied by the number of shares of common stock granted multiplied by the closing price
of the common stock on the NASDAQ Capital Market on the day of the grant, at the election of each director. We also reimburse our
non-employee directors for reasonable expenses in connection with attendance at board of director and committee meetings.
In addition
to cash compensation for services as a member of the board, non-employee directors are also eligible to receive nondiscretionary,
automatic grants of stock options under the Non-Employee Director Compensation Plan. An outside, non-employee director who joins
our board is automatically granted an initial option to purchase 10,000 shares upon first becoming a member of our board. The initial
option vests and becomes exercisable over three years, with 3,334 shares vesting on the first anniversary of the date of grant
and the remainder vesting monthly thereafter. Each non-employee director will receive an annual grant of 2,250 shares of common
stock on the last business day in February. Such shares of common stock will vest immediately on the day of grant. Additionally,
each non-employee director receiving an annual grant will receive an additional cash payment equal to 0.40 multiplied by the number
of shares of common stock granted multiplied by the closing price of the common stock on the Trading Market on the last business
day in February. No annual grant shall be granted to any non-employee director in the same calendar year that such person received
his or her initial grant.
In the
interest of good corporate governance and to further align the interests of members of the Board of Directors with the Company’s
stockholders, the Nominating and Corporate Governance Committee of the Board of Directors has adopted stock ownership guidelines
for directors. Under these guidelines, if a Director exercises a stock option, it is expected that such director would, from such
date of option exercise, maintain ownership of at least a number of shares equal to twenty percent of the net value of the shares
acquired (after deducting the exercise price and taxes). In the case of shares acquired upon the exercise of a stock option, each
director is expected to hold such shares for nine months after termination of his or her service on the Board of Directors.
Non-Employee
Director Compensation Plan Prior to February 24, 2016
Pursuant
to our non-employee director compensation plan prior to February 24, 2016, each non-employee director was entitled to the following
annual retainers:
|
·
Board Member
|
$32,500
|
|
·
Lead Independent Director
|
$15,000
|
|
·
Chair of the Audit Committee
|
$10,000
|
|
·
Chair of the Compensation Committee
|
$5,000
|
|
·
Chair of the Nominating and Corporate Governance Committee
|
$5,000
|
|
·
Audit Committee member (other than Chair)
|
$7,500
|
|
·
Compensation Committee Member (other than Chair)
|
$2,000
|
|
·
Nominating and Corporate Governance Committee Member (other than the Chair)
|
$2,000
|
All Audit Committee
retainers must be paid in cash. All other retainers may be paid in cash, options or as a stock grant, at the election of each director.
We also reimburse our non-employee directors for reasonable expenses in connection with attendance at board of director and committee
meetings.
In addition
to cash compensation for services as a member of the board, non-employee directors are also eligible to receive nondiscretionary,
automatic grants of stock options under the Non-Employee Director Compensation Plan. An outside, non-employee director who joins
our board is automatically granted an initial option to purchase 10,000 shares upon first becoming a member of our board. The initial
option vests and becomes exercisable over three years, with the first one-third of the shares vesting on the first anniversary
of the date of grant and the remainder vesting in equal monthly increments thereafter. After each of our regularly scheduled Annual
Meetings of Stockholders, each non-employee director is automatically granted an option to purchase 3,000 shares of our common
stock, provided that no annual grant shall be granted to a non-employee director in the same calendar year that such person received
his or her initial grant. These options vest in equal monthly increments over the period of one year.
In July 2014,
the Board eliminated the provisions in the 2006 Plan that provided for automatic option grants to non-employee directors to eliminate
redundancy in our non-employee director compensation.
Executive
Officers
The
Company has the following executive officers:
|
Name
|
Age
|
Position with the Company
|
|
Jim Schutz
|
53
|
Chief Executive Officer
|
|
Robert Miller
|
73
|
Secretary, Chief Financial Officer, Chief Operating Officer
|
|
Bruce Thornton
|
52
|
Executive Vice President
|
|
Robert Northey
|
59
|
Executive Vice President of Research and Development
|
The biographies of our executive
officers and certain information regarding each officer’s experience, attributes, skills and/or qualifications that led to
the conclusion that the officer should be serving as an officer of the Company are stated below.
Jim Schutz
: For Mr. Schutz’s full biography, please
refer to page 51 in the section entitled “Directors.”
Robert Miller
: Mr. Miller has served
as our Chief Financial Officer since June 2004 and Secretary and Chief Operating Officer since February 2013. He was a consultant
to us from March 2003 to May 2004. Mr. Miller has served as a director of Scanis, Inc., a technology company, since 1998 and served
as the acting Chief Financial Officer of Scanis from 1998 to June 2006. He was a Chief Financial Officer consultant to Evit Labs
from June 2003 to December 2004, Wildlife International Network from October 2002 to December 2005, Endoscopic Technologies from
November 2002 to March 2004, Biolog from January 2000 to December 2002 and Webware from August 2000 to August 2002. Prior to this,
Mr. Miller was the Chief Financial Officer for GAF Corporation, Penwest Ltd. and Bugle Boy, and the Treasurer of Mead Corporation.
Bruce Thornton
: Mr. Thornton has been
working for our Company for a decade. Since June 2005, he served as Executive Vice President of International Operations and Sales.
He has an extensive knowledge of our operations, the market for our products and our vision and goals for the future. Mr. Thornton
served as our General Manager for U.S. operations from March 2004 to July 2005. He served as Vice President of Operations for Jomed
(formerly EndoSonic Corp.) from January 1999 to September 2003, and as Vice President of Manufacturing for Volcano Therapeutics,
an international medical device company, following its acquisition of Jomed, until March 2004.
Robert Northey, Ph.D.
: Dr. Northey has
served as our Executive Vice President of Research and Development since July 2005. Dr. Northey served as a consultant to us from
May 2001 to June 2005. From August 1998 until June 2005, he was an assistant professor in the paper science and engineering department
at the University of Washington. Dr. Northey received a B.S. in wood and fiber science and a Ph.D. in wood chemistry, each from
the University of Washington.
Executive Compensation
This prospectus contains information about
the compensation paid to our Named Executive Officers, as defined by Item 402(m)(2) of Regulation S-K, during our fiscal year
ended March 31, 2016, or fiscal year 2016. For fiscal year 2016, in accordance with the rules and regulations of the Securities
and Exchange Commission for smaller reporting companies, we determined that the following officers were our Named Executive Officers:
|
|
·
|
Jim Schutz, Chief Executive Officer,
|
|
|
·
|
Bob Miller, Chief Financial Officer,
|
|
|
·
|
Bruce Thornton, Executive Vice President of International Sales, and
|
|
|
·
|
Robert Northey, Executive Vice President of Research and Development
|
Summary Compensation Table
The
following table sets forth, for the fiscal years ended March 31, 2016 and 2015, all compensation paid or earned by (i) all individuals
serving as our Principal Executive Officer; (ii) our two most highly compensated executive officers, other than our Principal Executive
Officer, who were serving as executive officers at the end of our fiscal year ended March 31, 2016; and (iii) up to two individuals
for whom disclosure would have been provided but for the fact that the individual was not serving as an executive officer. These
executive officers are referred to herein as our “Named Executive Officers.”
|
Name and Principal Position
|
Fiscal Year Ended March 31, 2016
|
Salary ($)
|
Bonus
($)
|
Option Awards ($) (1) (2)
|
All Other Compensation ($)
|
Total
($)
|
|
Jim Schutz
Chief Executive Officer
|
2016
|
250,000
|
–
|
–
|
50,601 (3)
|
300,601
|
|
2015
|
250,000
|
–
|
–
|
46,215 (4)
|
296,215
|
|
Robert Miller
Secretary, Chief Financial Officer and Chief Operating
Officer
|
2016
|
250,000
|
–
|
30,402
|
45,319 (5)
|
325,721
|
|
2015
|
250,000
|
–
|
–
|
71,053 (6)
|
321,053
|
|
Bruce Thornton
Executive Vice President of International Operations and Sales
|
2016
|
250,000
|
–
|
40,862
|
53.615 (7)
|
344,477
|
|
2015
|
250,000
|
20,000 (8)
|
–
|
55,860 (9)
|
325,860
|
|
Robert Northey
Executive Vice President of Research and Development
|
2016
|
194,000
|
20,000 (10)
|
38,002
|
44,886 (11)
|
296,888
|
|
2015
|
185,000
|
30,000 (12)
|
–
|
40,828 (13)
|
255,828
|
|
|
(1)
|
Represents the aggregate grant date fair value of stock option awards granted
in the covered fiscal year as computed in accordance with FASB ASC Topic 718, Compensation — Stock Compensation. The fair
value of each stock option award is estimated for the covered fiscal year on the date of grant using the Black-Scholes option valuation
model. A discussion of the assumptions used in calculating the amounts in this column may be found in Note 14 to our audited consolidated
financial statements for the year ended March 31, 2016, included in our Annual Report on Form 10-K filed with the SEC on June 21,
2016. The amounts in this column do not represent the actual amounts paid to or realized by our Named Executive Officers during
the fiscal years ended March 31, 2016 and 2015.
|
|
|
(2)
|
No stock options were awarded during the fiscal year ended March 31, 2015.
On August 21, 2015, we granted stock options to the executive officers pursuant to our 2016 Bonus Plan. The 2016 options were awarded
pursuant to our Stock Incentive Plans in effect for the applicable fiscal year. All bonus options granted in 2016 vested based
on the achievement of target goals and milestones, with specific milestones and a bonus potential range for each executive officer.
The bonus did not entitle executive officers to payment of a bonus but rather allowed for potential bonuses for executive officers
if they meet their respective target goals and milestones, subject to the determination of the Compensation Committee. Options
only vested, in whole or in part, in the event the executive achieved a minimum of 80% of each of his target milestones in the
applicable fiscal year, as determined by the Compensation Committee by June 30, 2016. If the executive achieved at least 80% of
the target milestones by March 31, 2016, then 80% of the executive’s options vested on June 30, 2016, as determined by the
Compensation Committee. The exercise price of the options granted in 2016 is the adjusted closing price of the Company’s
common stock on the date of grant. The stock options if vested have a term of ten years.
|
|
|
(3)
|
The 2016 perquisites and personal benefits for Mr. Schutz include (a) personal
use of a Company automobile in the amount of $2,055, (b) matching 401k contribution in the amount of $10,000, and (c) payment of
$38,546 to cover premium for life, health, dental and vision insurance policy for the benefit of Mr. Schutz.
|
|
|
(4)
|
The 2015 perquisites and personal benefits for Mr. Schutz include (a) personal
use of a Company automobile in the amount of $2,055, (b) matching 401k contribution in the amount of $10,000, and (c) payment of
$34,160 to cover premium for life, health, dental and vision insurance policy for the benefit of Mr. Schutz.
|
|
|
(5)
|
The 2016 perquisites and personal benefits for Mr. Miller include (a) matching
401k contribution in the amount of $10,000, and (b) payment of $35,319 to cover premium for life, health, dental and vision insurance
policy for the benefit of Mr. Miller.
|
|
|
(6)
|
The 2015 perquisites and personal benefits for Mr. Miller include (a) matching
401k contribution in the amount of $10,000, and (b) payment of $61,053 to cover premium for life, health, dental and vision insurance
policy for the benefit of Mr. Miller.
|
|
|
(7)
|
The 2016 perquisites and personal benefits for Mr. Thornton include (a)
a car allowance in the amount of $11,700, (b) matching 401k contribution in the amount of $10,000, and (c) payment of $31,915 to
cover premiums for life, health, dental and vision insurance policies for the benefit of Mr. Thornton.
|
|
|
(8)
|
Mr. Thornton’s 2015 bonus consisted of a one-time cash bonus of $20,000.
|
|
|
(9)
|
The 2015 perquisites and personal benefits for Mr. Thornton include (a)
a car allowance in the amount of $11,700, (b) matching 401k contribution in the amount of $10,000, and (c) payment of $34,160 to
cover premiums for life, health, dental and vision insurance policies for the benefit of Mr. Thornton.
|
|
|
(10)
|
Mr. Northey’s 2016 bonus compensation consisted of a one-time cash
bonus of $20,000.
|
|
|
(11)
|
The 2016 perquisites and personal benefits for Mr. Northey include (a)
matching 401k contribution in the amount of $7,656, and (b) payment of $37,230 to cover premiums for life, health, dental and vision
insurance policies for the benefit of Mr. Northey.
|
|
|
(12)
|
Mr. Northey’s 2015 bonus compensation consisted of a one-time cash
bonus of $30,000.
|
|
|
(13)
|
The 2015 perquisites and personal benefits for Mr. Northey include (a)
matching 401k contribution in the amount of $7,400, and (b) payment of $33,428 to cover premiums for life, health, dental and vision
insurance policies for the benefit of Mr. Northey.
|
Employment Agreements and Potential
Payments upon Termination
Employment Agreement with Mr.
Jim Schutz
On July 26, 2016, we entered into a new employment
agreement with Jim Schutz, our President and Chief Executive Officer to update his agreements and responsibilities.
The terms of the new employment agreement provide
for a continued annual base salary of $250,000 or such other amount as the Board of Directors may set. In addition, Mr. Schutz
is eligible to receive an annual bonus, the payment, type and amount of which is in the sole discretion of the Compensation Committee.
Mr. Schutz also receives certain benefits, such as participation in our health and welfare plans, vacation and reimbursement of
expenses.
The employment agreement provides Mr. Schutz
with certain separation benefits in the event of termination without cause, upon change of control or resignation by the executive
for good reason; as such terms are defined in the employment agreement. In the event Mr. Schutz is terminated without cause, or
upon change of control, or resigns for good reason, he is entitled to:
|
|
·
|
a lump severance payment equal to one-and-a-half times the executive’s
base salary;
|
|
|
·
|
a bonus, upon determination by the Corporation’s Board of Directors
or Compensation Committee, as appropriate, to be made in its sole discretion as to whether to grant a bonus, and if such bonus
is granted, the amount, form and payment schedule. For the avoidance of doubt, executive shall not be entitled to any bonus solely
for reason of termination, unless the Board of Directors or the Compensation Committee, as appropriate, in its sole discretion
awards a bonus to executive;
|
|
|
·
|
automatic vesting of all unvested time-based options and equity awards
and exercisability of awards for the remainder of their respective terms;
|
|
|
·
|
vesting of performance-based equity compensation awards in accordance with
the terms of the awards, if the performance goals are satisfied; and
|
|
|
·
|
up to 18 months (the lesser of one year following the date of termination
or until such executive becomes eligible for medical insurance coverage provided by another employer) reimbursement for health
care premiums under COBRA.
|
Mr. Schutz may terminate his employment for
any reason upon at least 30 days prior written notice. Receipt of the termination benefits described above is contingent on each
executive executing a general release of claims against our Company, his resignation from any and all directorships and every other
position held by him with our Company or any of our subsidiaries, and his return to our Company of all Company property received
from or on account of our Company or any of our affiliates by such executive. In addition, the executive is not entitled to such
benefits if he did not comply with the non-competition and invention assignment provisions of his employment agreement during the
term of his employment or the confidentiality provisions of his employment agreement, whether during or after the term of his employment.
Furthermore, we are under no obligation to pay the above-mentioned benefits if the executive does not comply with the non-solicitation
provisions of his employment agreement, which prohibit a terminated executive from interfering with the business relations of our
Company or any of our affiliates and from soliciting employees of our Company. These provisions apply during the term of employment
and for two years following termination.
In connection with the entry into the new agreement,
the Compensation Committee intended to eliminate certain outdated pay practices, including providing a full tax gross up upon termination,
automatic vesting of all performance-based equity awards under certain circumstances, and single-trigger change of control payments.
Additionally, the Compensation Committee wanted to bring the employment agreement in line with current law by adding provisions.
Employment Agreement with Mr.
Robert Miller
The terms of the June 20, 2013 employment agreement
with Robert Miller, our Chief Financial Officer, provides for an annual salary of $250,000 or such other amount as the Board of
Directors may set. Mr. Miller also receives certain benefits, such as participation in our health and welfare plans, vacation and
reimbursement of expenses.
The employment agreement provides Mr. Miller
with certain separation benefits in the event of termination without cause or resignation for good reason; as such terms are defined
in the employment agreement. In the event Mr. Miller is terminated without cause or resigns for good reason, he is entitled to:
|
|
·
|
a lump severance payment equal to 18 times the average monthly base salary
paid to the executive over the preceding 12 months (or for the term of the executive’s employment if less than 12 months);
|
|
|
·
|
automatic vesting of all unvested options and other equity awards;
|
|
|
·
|
the extension of exercisability of all options and other equity awards
to at least 12 months following the date the executive terminates employment or, if earlier, until the option expires;
|
|
|
·
|
up to one year (the lesser of one year following the date of termination
or until such executive becomes eligible for medical insurance coverage provided by another employer) reimbursement for health
care premiums under COBRA; and
|
|
|
·
|
a full gross up of any excise taxes payable by the executive under Section
4999 of the Internal Revenue Code because of the foregoing payments and acceleration (including the reimbursement of any additional
federal, state and local taxes payable as a result of the gross up).
|
Mr. Miller may terminate his employment for
any reason upon at least 30 days prior written notice. Receipt of the termination benefits described above is contingent on each
executive executing a general release of claims against our Company, his resignation from any and all directorships and every other
position held by him with our Company or any of our subsidiaries, and his return to our Company of all Company property received
from or on account of our Company or any of our affiliates by such executive. In addition, the executive is not entitled to such
benefits if he did not comply with the non-competition and invention assignment provisions of his employment agreement during the
term of his employment or the confidentiality provisions of his employment agreement, whether during or after the term of his employment.
Furthermore, we are under no obligation to pay the above-mentioned benefits if he does not comply with the non-solicitation provisions
of his employment agreement, which prohibit a terminated executive from interfering with the business relations of our Company
or any of our affiliates and from soliciting employees of our Company. These provisions apply during the term of employment and
for two years following termination.
Employment Agreements with Mr. Bruce
Thornton and Mr. Robert Northey
We entered into an employment agreement, dated
as of June 1, 2005, as amended on August 5, 2008, with Bruce Thornton, our Executive Vice President of International Operations
and Sales. We also entered into an employment agreement with Mr. Robert Northey, our Executive Vice President of Research and Development
on April 1, 2008. The terms of the employment agreements provide for an annual salary of $250,000 for Mr. Thornton and $185,000
for Mr. Northey, respectively, or such other amount as the Chief Executive Officer may set. Mr. Northey is entitled to receive
an annual bonus of $50,000 upon meeting mutually agreed upon annual milestones. Mr. Thornton and Mr. Northey also receive certain
benefits, such as participation in our health and welfare plans, vacation and reimbursement of expenses. Additionally, Mr. Thornton
receives a car allowance.
The employment agreements provide each executive
with certain separation benefits in the event of termination without cause or resignation by Messrs. Thornton or Northey for good
reason; as such terms are defined in the employment agreement. In the event Messrs. Thornton or Northey are terminated without
cause or resigns for good reason, the respective executive is entitled to:
|
|
·
|
a lump severance payment equal to 12 times the average monthly base salary
paid to Mr. Thornton over the preceding 12 months (or for the term of the executive’s employment if less than 12 months)
or equal to 6 times the monthly base salary paid to Mr. Northey in the calendar month immediately preceding the month of termination;
|
|
|
·
|
automatic vesting of all unvested options and other equity awards;
|
|
|
·
|
the extension of exercisability of all options and other equity awards
to at least 12 months following the date the executive terminates employment or, if earlier, until the option expires;
|
|
|
·
|
up to one year (the lesser of one year following the date of termination
or until such executive becomes eligible for medical insurance coverage provided by another employer) reimbursement for health
care premiums under COBRA; and
|
|
|
·
|
a full gross up of any excise taxes payable by the executive under Section
4999 of the Internal Revenue Code because of the foregoing payments and acceleration (including the reimbursement of any additional
federal, state and local taxes payable as a result of the gross up).
|
Messrs. Thornton or Northey may terminate their
respective employment for any reason upon at least 30 days prior written notice. Receipt of the termination benefits described
above is contingent on each executive executing a general release of claims against our Company, his resignation from any and all
directorships and every other position held by him with our Company or any of our subsidiaries, and his return to our Company of
all Company property received from or on account of our Company or any of our affiliates by such executive. In addition, the executive
is not entitled to such benefits if he did not comply with the non-competition and invention assignment provisions of his employment
agreement during the term of his employment or the confidentiality provisions of his employment agreement, whether during or after
the term of his employment. Furthermore, we are under no obligation to pay the above-mentioned benefits if the executive does not
comply with the non-solicitation provisions of his employment agreement, which prohibit a terminated executive from interfering
with the business relations of our Company or any of our affiliates and from soliciting employees of our Company. These provisions
apply during the term of employment and for two years following termination.
Potential Payments upon Termination
The
table below was prepared as though each of Messrs. Schutz, Miller, Thornton, and Northey had been terminated on March 31, 2016,
the last day of our last completed fiscal year, without cause, or upon change of control, or resigned for good reason, as these
terms are defined in the agreements with our Company. More detailed information about the payment of benefits, including duration,
is contained in the discussion above. All such payments and benefits would be provided by our Company. The assumptions and valuations
are noted in the footnotes.
|
Name
|
Salary
Continuation
($)
|
Health and Welfare
Benefits
Continuation
($)
(1)
|
Excise and Tax
Gross-up
($)
(2)
|
|
Jim Schutz
|
375,000
|
38,546
|
0
|
|
Robert Miller
|
375,000
|
35,319
|
191,824
|
|
Bruce Thornton
|
250,000
|
31,915
|
131,795
|
|
Robert Northey
|
194,000
|
37,230
|
108,100
|
|
(1)
|
Amount assumes our cost of providing life, health, dental and vision insurance
at the same rate for 12 months.
|
|
|
|
|
(2)
|
In calculating these amounts we assumed a termination date on March 31, 2016, and the maximum Federal and California income
and other payroll taxes, aggregating an effective tax rate of 46.75%.
|
Annual Incentive Plans
Pursuant to
our annual Bonus Plan, executive officers of our Company, including Messrs. Schutz, Miller, Thornton, and Northey have the potential
to earn an annual bonus based on the individual’s contribution to the Company’s target goals and milestones. Specific
goals and milestones and a bonus potential range for each executive officer are set forth in the bonus plan. The Compensation Committee
determines whether a bonus pool for executive officers will be established within a specified time period after the end of each
fiscal year. If a bonus pool is established, the Compensation Committee has discretion to set appropriate bonus amounts within
an executive officer’s bonus range, based on the Compensation Committee’s assessment of corporate and individual achievements.
The Compensation
Committee may decide that bonuses awarded to executive officers under the bonus plan will be paid in cash, stock options, stock
or a combination of cash, options, and/or stock depending on our year-end cash position, cash needs and projected cash receipts.
The Compensation Committee will not declare any bonus pool or grant any cash awards that will endanger our ability to finance our
operations and strategic objectives or place us in a negative cash flow position, in light of our anticipated cash needs.
2015 Bonus Awards for Named Executive
Officers
We awarded
Mr. Northey, our Vice President of Research and Development a bonus of $20,000 for his efforts related to the research and development
and launch of new products in fiscal year 2015. The bonus was paid in fiscal year 2016.
We awarded
Mr. Thornton, our Executive Vice President of International Operations and Sales, a bonus of $20,000 for meeting milestones established
in his compensation and bonus plan in fiscal year 2015. The bonus was paid in fiscal year 2016.
2016 Bonus Awards for Named Executive
Officers
For
fiscal year 2016, each executive officer, including our Named Executive Officers, had the potential to earn an annual bonus based
on the Compensation Committee’s assessment of the individual’s and our Company’s contribution to target goals
and milestones. Specific goals and milestones and a bonus potential range for employees and executive officers, including our Named
Executive Officers, was set forth in the bonus plan.
For
fiscal year 2016, the Compensation Committee granted stock options to executive officers. Such stock options vested in whole or
in part in the event that each executive officer meets target milestones established in the Fiscal Year 2016 Bonus Plan. On July
26, 2016, the Compensation Committee determined that target milestones have been met in part and determined that part of the options
vested. Employees were eligible for a similar bonus program.
The table below shows the grant
and vesting of FY 2016 bonus options for our Named Executive Officers. Share numbers have been adjusted for the 1-for-5 reverse
stock split effective June 24, 2016.
|
Name
|
Number
of options granted
(1)
|
Number of options vested (2)
|
|
Jim Schutz
|
0
|
0
|
|
Robert Miller
|
8,000
|
7,912
|
|
Bruce Thornton
|
10,000
|
9,870
|
|
Robert Northey
|
10,000
|
9,890
|
|
(1)
|
The exercise price of $5.80 equals the adjusted closing price of our common stock on the day of the grant, August 21, 2015.
|
|
|
|
|
(2)
|
Such options vested in whole or in part in the event the executive achieved a minimum of 80% of his target milestones. If
such executive did not achieve 80% of their target milestones, then 100% of the options expired. If the executive achieved at least
80% of his target milestones, then 80% of the executive’s options vested. The vesting of the remaining of the remaining 20%
was in the discretion of the Compensation Committee.
|
2017 Bonus Plan Structure for Executive
Officers
:
For fiscal year 2017 ending on March 31, 2017,
we anticipate that the Compensation Committee will determine target milestones specific to each executive officer’s job and
a pool of options, stock and cash as a bonus payment. Specific goals and milestones and a bonus potential range for our executive
officers, including our Named Executive Officers, will be set forth in the 2017 bonus plan.
Equity Compensation Plan Information
Pursuant to Item 201(d) of Regulation S-K,
“Securities Authorized for Issuance Under Equity Compensation Plans,” we are providing the following information summarizing
information about our equity compensation plans as of March 31, 2016. All share numbers have been updated for the 1-for-5 reverse
stock split of the Company’s common stock effective as of June 24, 2016.
|
Plan Category
|
Number of Securities
to be issued upon
exercise of outstanding
options
and rights
|
Weighted average
exercise price of
outstanding
options and rights
|
Number of Securities
remaining available
for
future issuance under
equity compensation plans
(excluding securities
reflected
in column (a))
|
|
Equity compensation plans approved by security holders
|
752,000
|
$21.47
|
583,000
|
|
Equity compensation plans not approved by security holders
|
–
|
–
|
–
|
|
Total
|
752,000
|
$21.47
|
583,000
|
Our Oculus Innovative
Sciences, Inc. Amended and Restated 2006 Stock Incentive Plan and our Oculus Innovative Sciences, Inc. 2011 Stock Incentive Plan
were adopted with the approval of our stockholders, and we have previously provided the material terms of such plans.
Outstanding Equity Awards
The following table shows grants of options
outstanding on March 31, 2016, the last day of our last completed fiscal year, to each of the Named Executive Officers named in
the Summary Compensation Table. All shares and per share data have been adjusted to reflect a 1-for-7 reverse stock split, effective
April 1, 2013 and a 1-for-5 reverse stock split, effective June 24, 2016.
|
Name
|
Grant Date
|
Initial
Number
of
Securities
Granted
|
Equity
Incentive
Plan Awards:
Number of
Securities
Underlying
Unexercised
Unearned
Options (#)
|
Option
Exercise
Price ($)
|
Number of Securities
Underlying
Unexercised
Options (#)
Exercisable
|
Number of
Securities
Underlying
Unexercised
Options (#)
Unexercisable
|
Option
Expiration
Date
|
|
Jim Schutz (1)
|
6/15/2007
|
2,856
|
0
|
$254.45
|
2,856
|
0
|
6/15/2017
|
|
3/10/2009
|
5,271
|
0
|
$38.15
|
5,271
|
0
|
3/10/2019
|
|
2/10/2010
|
3,571
|
0
|
$66.85
|
3,571
|
0
|
2/10/2020
|
|
6/7/2010
|
1,785
|
0
|
$68.95
|
1,785
|
0
|
6/7/2020
|
|
3/31/2011
|
3,928
|
0
|
$70.35
|
3,928
|
0
|
3/31/2021
|
|
6/16/2011
|
1,428
|
0
|
$56.00
|
1,428
|
0
|
6/16/2021
|
|
3/7/2012
|
5,357
|
0
|
$43.75
|
5,357
|
0
|
3/7/2022
|
|
8/24/2012
|
4,285
|
0
|
$32.55
|
4,285
|
0
|
8/24/2022
|
|
9/19/2013
|
20,000
|
0
|
$30.00
|
16,666
|
3,334
|
9/19/2023
|
|
Robert Miller (2)
|
3/10/2009
|
5,271
|
0
|
$38.15
|
5,271
|
0
|
3/10/2019
|
|
6/7/2010
|
5,357
|
0
|
$68.95
|
5,357
|
0
|
6/7/2020
|
|
3/31/2011
|
357
|
0
|
$70.35
|
357
|
0
|
3/31/2021
|
|
6/16/2011
|
5,000
|
0
|
$56.00
|
5,000
|
0
|
6/16/2021
|
|
3/7/2012
|
1,785
|
0
|
$105.00
|
1,785
|
0
|
3/7/2022
|
|
8/24/2012
|
4,285
|
0
|
$32.55
|
4,285
|
0
|
8/24/2022
|
|
9/19/2013
|
5,351
|
0
|
$14.85
|
4,459
|
892
|
9/19/2023
|
|
3/4/2014
|
26,020
|
0
|
$19.50
|
17,346
|
8,674
|
3/4/2024
|
|
8/21/2015
|
8,000
|
88
|
$5.80
|
7,912
|
0
|
8/21/2025
|
|
Bruce Thornton (3)
|
6/15/2007
|
272
|
0
|
$254.45
|
272
|
0
|
6/15/2017
|
|
6/15/2007
|
441
|
0
|
$35.00
|
441
|
0
|
6/15/2017
|
|
12/9/2008
|
5,428
|
0
|
$14.00
|
1,395*
|
0
|
12/9/2018
|
|
6/7/2010
|
1,428
|
0
|
$68.95
|
1,428
|
0
|
6/7/2020
|
|
6/7/2010
|
1,428
|
0
|
$105.00
|
1,428
|
0
|
6/7/2020
|
|
6/16/2011
|
5,357
|
0
|
$56.00
|
5,357
|
0
|
6/16/2021
|
|
3/7/2012
|
1,785
|
0
|
$43.75
|
1,785
|
0
|
3/7/2022
|
|
9/19/2013
|
3,745
|
0
|
$14.85
|
3,121
|
624
|
9/19/2023
|
|
3/4/2014
|
21,602
|
0
|
$19.50
|
14,285
|
7,143
|
3/4/2024
|
|
8/21/2015
|
10,000
|
130
|
$5.80
|
9,870
|
0
|
8/21/2025
|
|
Robert Northey (4)
|
6/15/2007
|
964
|
0
|
$254.45
|
964
|
0
|
6/15/2017
|
|
12/9/2008
|
2,857
|
0
|
$29.75
|
2,857
|
0
|
12/9/2018
|
|
2/25/2010
|
2,571
|
0
|
$64.05
|
2,571
|
0
|
2/25/2020
|
|
5/17/2011
|
3,142
|
0
|
$66.15
|
3,142
|
0
|
5/17/2021
|
|
9/19/2013
|
3,745
|
0
|
$14.85
|
3,121
|
624
|
9/19/2023
|
|
3/4/2014
|
20,140
|
0
|
$19.50
|
13,426
|
6,714
|
3/4/2024
|
|
8/21/2015
|
10,000
|
110
|
$5.80
|
9,890
|
0
|
8/21/2025
|
|
|
*
|
4,033 shares were exercised.
|
|
|
(1)
|
Options with an expiration date of September 19, 2023, will vest quarterly
over a three-year vesting schedule commencing on the date of grant. The options expiring on September 19, 2023, represent 1/3 of
the equity grant awarded to Mr. Schutz pursuant to our employment agreement with him effective June 20, 2013.
|
|
|
(2)
|
Options with an expiration date of March 4, 2024, will vest 1/36th per month
over a three-year vesting schedule commencing on the date of grant. Options with an expiration date of September 19, 2023, will
vest quarterly over a three-year vesting schedule commencing on the date of grant.
|
|
|
(3)
|
Options with an expiration date of September 19, 2023 or March 4, 2024, will
vest 1/36th per month over a three-year vesting schedule commencing on the date of grant.
|
|
|
(4)
|
Options with an expiration date of September 19, 2023 or March 4, 2024, will
vest 1/36th per month over a three-year vesting schedule commencing on the date of grant.
|
Retirement Benefits
On January 1,
2011, we established a qualified 401(k) employee savings and retirement plan for all regular full-time U.S. employees. Eligible
employees may elect to defer a percentage of their eligible compensation in the 401(k) plan, subject to the statutorily prescribed
annual limit. We may make matching contributions on behalf of all participants in the 401(k) plan in the amount equal to 4% of
an employee’s contributions. All contributions are immediately fully vested. We intend the 401(k) plan to qualify under Sections
401(k) and 501 of the Internal Revenue Code of 1986, as amended, so that contributions by employees or us to the 401(k) plan and
income earned, if any, on plan contributions are not taxable to employees until withdrawn from the 401(k) plan (except as regards
Roth contributions), and so that we will be able to deduct our contributions when made. The trustee of the 401(k) plan, at the
direction of each participant, invests the assets of the 401(k) plan in any of a number of investment options. Company contributions
to the 401(k) plan amounted to an aggregate of $158,000 for the year ended March 31, 2016.
Code of Business Conduct and Senior Financial
Officers’ Code of Ethics
We have adopted a Code of Business Conduct
that applies to all of our officers and employees, including our Chief Executive Officer, Chief Financial Officer, and other employees
who perform financial or accounting functions. The Code of Business Conduct sets forth the basic principles that guide the business
conduct of our employees. We have also adopted a Senior Financial Officers’ Code of Ethics that specifically applies to our
Chief Executive Officer, Chief Financial Officer, and other key management employees.
To date, there have been no waivers under our
Code of Business Conduct or Senior Financial Officers’ Code of Ethics. We intend to disclose future amendments to certain
provisions of our Code of Business Conduct or Senior Officers’ Code of Ethics or any waivers, if and when granted, of our
Code of Business Conduct or Senior Officers’ Code of Ethics on our website at http://www.oculusis.com within four business
days following the date of such amendment or waiver.
Certain
Relationships AND Related Transactions
It is our policy that all employees, officers
and directors must avoid any activity that is, or has the appearance of, conflicting with the interests of our Company. This policy
is included in our Code of Business Conduct, and our Board formally adopted a Related Party Transaction Policy and Procedures in
July 2007 for the approval of interested transactions with persons who are Board members or nominees, executive officers, holders
of 5% of our common stock, or family members of any of the foregoing. The Related Party Transaction Policy and Procedures are administered
by our Audit Committee. We conduct a review of all related party transactions for potential conflict of interest situations on
an ongoing basis and all such transactions relating to executive officers and directors must be approved by the Audit Committee.
There have been no relevant related party transactions meeting the disclosure requirements in this period.
Arrangements or Understandings between our
Executive Officers or Directors and Others
There are no arrangements or understandings
between our executive officers or directors and any other person pursuant to which he or she was or is to be selected as a director
or officer.
Security
Ownership of Certain Beneficial Owners and Management
The following
table sets forth certain information as of July 5, 2016, as to shares of our common stock beneficially owned by: (1) each of our
Named Executive Officers listed in the Summary Compensation Table, (2) each of our current directors and (3) all of our directors
and executive officers as a group. We currently do not know of any shareholder who beneficially owns 5% or more of our common stock.
We have
determined beneficial ownership in accordance with the rules of the SEC. Except as indicated by the footnotes below, we believe,
based on the information furnished to us, that the persons and entities named in the table below have sole voting and investment
power with respect to all shares of common stock that they beneficially own, subject to applicable community property laws.
In computing
the number of shares of common stock beneficially owned by a person and the percentage ownership of that person, we deemed outstanding
shares of common stock subject to options held by that person that are currently exercisable or exercisable upon vesting. We did
not deem these shares outstanding, however, for the purpose of computing the percentage ownership of any other person.
Share
numbers have been adjusted for the 1-for-5 reverse stock split effective June 24, 2016.
Officers and Directors
|
Name and address of
beneficial owner (1)
|
Nature of beneficial ownership
|
Amount of Beneficial Ownership
|
Percent of Shares Beneficially Owned (3)
|
|
Shares Owned
|
Shares – Includes all Rights to Acquire (2)
|
Total
|
|
Jim Schutz (4)
|
Chief Executive Officer and Director
|
5,177
|
48,481
|
53,658
|
1.3%
|
|
Robert Miller (5)
|
Secretary, Chief Financial Officer and Chief Operating Officer
|
2,085
|
60,866
|
62,951
|
1.5%
|
|
Bruce Thornton (6)
|
Executive Vice President of International Operations and Sales
|
0
|
47,453
|
47,453
|
1.1%
|
|
Robert Northey (7)
|
Executive Vice President of Research and Development
|
0
|
42,719
|
42,719
|
1.0%
|
|
Jerry McLaughlin (8)
|
Lead Independent Director
|
4,928
|
29,224
|
34,152
|
*
|
|
Sharon Barbari (9)
|
Director
|
4,010
|
16,192
|
20,202
|
*
|
|
Russell Harrison (10)
|
Director
|
4,265
|
17,893
|
22,158
|
*
|
|
Jay Birnbaum (11)
|
Director
|
3,907
|
34,798
|
38,705
|
*
|
|
All directors and executive officers as a group (8 persons)
|
24,372
|
297,626
|
321,998
|
7.7%
|
*Indicates
ownership of less than 1.0%
|
|
(1)
|
Unless otherwise stated, the address of each beneficial owner listed in
the table is c/o Oculus Innovative Sciences, Inc. 1129 North McDowell Blvd. Petaluma, CA 94954.
|
|
|
(2)
|
Represents shares subject to outstanding stock options and warrants currently
exercisable or exercisable upon vesting.
|
|
|
(3)
|
We had a total of 4,200,756 shares of common stock issued and outstanding on July 5, 2016.
|
|
|
(4)
|
Mr. Schutz is our President and Chief Executive Officer. He is also a member of our Board of Directors.
Mr. Schutz beneficially owns 5,177 shares of common stock and 48,481 shares of common stock issuable upon the exercise of options.
|
|
|
(5)
|
Mr. Miller is our Chief Financial Officer. Mr. Miller beneficially owns 2,085 shares of common
stock, which includes 1,714 shares held by The Miller 2005 Grandchildren’s Trust, for which Mr. Miller and his wife, Margaret
I. Miller, are the trustees. Mr. Miller and Ms. Miller share voting and dispositive control over the shares held by The Miller
2005 Grandchildren’s Trust. Mr. Miller also beneficially owns 60,866 shares of common stock issuable upon the exercise of
options.
|
|
|
(6)
|
Mr. Thornton is our Executive Vice President of International Operations and Sales. Mr. Thornton
beneficially owns 0 shares of common stock and 47,453 shares of common stock issuable upon the exercise of options.
|
|
|
(7)
|
Mr. Northey is our Executive Vice President of Research and Development. He beneficially owns 0
shares of common stock and 42,719 shares of common stock issuable upon the exercise of options.
|
|
|
(8)
|
Mr. McLaughlin is a member of our Board of Directors and was appointed as Lead Independent Director
on March 26, 2014. He beneficially owns 4,928 shares of common stock and 29,224 shares of common stock issuable upon the exercise
of options.
|
|
|
(9)
|
Ms. Barbari is a member of our Board of Directors. She beneficially owns 4,010 shares of common
stock held by The Barbari Family Trust – Sharon Ann Barbari and Edward Paul Barbari Trustees, and 16,192 shares of common
stock issuable upon the exercise of options.
|
|
|
(10)
|
Mr. Harrison is a member of our Board of Directors. He beneficially owns 4,265 shares of common
stock and 17,893 shares of common stock issuable upon the exercise of options.
|
|
|
(11)
|
Dr. Birnbaum is a member of our Board of Directors. He beneficially owns 3,907 shares of common
stock and 34,798 shares of common stock issuable upon the exercise of options.
|
DESCRIPTION
OF SECURITIES
The following description
of our capital stock and provisions of our Restated Certificate of Incorporation and our Amended and Restated Bylaws, is only a
summary. You should also refer to our Restated Certificate of Incorporation, and our Amended and Restated Bylaws, copies of which
are incorporated by reference as exhibits to the registration statement of which this prospectus is a part. All shares have been
adjusted for a 1-for-5 reverse stock split effective June 24, 2016.
Preferred Stock
Our Board of Directors
is authorized to issue 714,286 shares of preferred stock in one or more series and to fix the rights, preferences, privileges,
qualifications, limitations and restrictions thereof, including dividend rights and rates, conversion rights, voting rights, terms
of redemption, redemption prices, liquidation preferences and the number of shares constituting any series or the designation of
such series, without any vote or action by our shareholders. Any preferred stock to be issued could rank prior to our common stock
with respect to dividend rights and rights on liquidation. Our Board of Directors, without shareholder approval, may issue preferred
stock with voting and conversion rights which could adversely affect the voting power of holders of our common stock and discourage,
delay or prevent a change in control of the Company. As of the date of this prospectus, no shares of preferred stock are outstanding.
Common Stock
We are authorized
to issue up to a total of 12,000,000 shares of common stock, $0.0001 par value per share. Each holder of common stock is entitled
to one vote for each share of common stock held on all matters submitted to a vote of stockholders. We have not provided for cumulative
voting for the election of directors in our Restated Certificate of Incorporation, as amended. This means that the holders of a
majority of the shares voted can elect all of the directors then standing for election. Subject to preferences that may apply to
shares of preferred stock outstanding at the time, the holders of outstanding shares of our common stock are entitled to receive
dividends out of assets legally available at the times and in the amounts that our Board of Directors may determine from time to
time.
Holders of common
stock have no preemptive subscription, redemption or conversion rights or other subscription rights. Upon our liquidation, dissolution
or winding-up, the holders of common stock are entitled to share in all assets remaining after payment of all liabilities
and the liquidation preferences of any outstanding preferred stock. Each outstanding share of common stock is, and all shares of
common stock to be issued in this offering, when they are paid for will be, fully paid and nonassessable.
Warrants
In connection with
this offering, we issued warrants to purchase 937,500 shares of our common stock. For every five shares of common stock issued,
we issued 3.75 of a warrant. Each five full warrants are exercisable for one share of our common stock at an initial exercise price
of $6.50 per share. The warrants are exercisable commencing upon consummation of this offering and terminating on the fifth
anniversary of the date of issuance.
The warrants were
issued in registered form under a warrant agreement between us and our warrant agent. The material provisions of the warrants are
set forth herein but are only a summary and are qualified in their entirety by the provisions of the warrant agreement that has
been filed as an exhibit to the registration statement of which this prospectus forms a part.
The warrants may be exercised upon surrender
of the warrant certificate on or prior to the expiration date at the offices of the warrant agent, with the exercise form on the
reverse side of the public warrant certificate completed and executed as indicated, accompanied by full payment of the exercise
price, by certified or official bank check payable to us, for the number of warrants being exercised. Under the terms of the warrant
agreement, we have agreed to use our best efforts to maintain the effectiveness of the registration statement and current prospectus
relating to common stock issuable upon exercise of the warrants until the expiration of the warrants. During any period we fail
to have maintained an effective registration statement covering the shares underlying the warrants, the warrant holder may exercise
the warrants on a cashless basis. The warrant holders do not have the rights or privileges of holders of common stock and any voting
rights until they exercise their warrants and receive shares of common stock. After the issuance of shares of common stock upon
exercise of the warrants, each holder will be entitled to one vote for each share held of record on all matters to be voted on
by stockholders.
No fractional shares of common stock will be
issued upon exercise of the warrants. If, upon exercise of the warrants, a holder would be entitled to receive a fractional interest
in a share, we, at our sole discretion may, upon exercise, either round up to the nearest whole number of shares of common stock
to be issued to the warrant holder or pay a cash adjustment in respect of such final fraction in an amount equal to such fraction
multiplied by the price per share at which shares of common stock may be purchased at the time a warrant is exercised. If multiple
warrants are exercised by the holder at the same time, we will aggregate the number of whole shares issuable upon exercise of all
the warrants.
The exercise price and number of shares of
common stock issuable upon exercise of the warrants may be adjusted in certain circumstances, including in the event of a stock
dividend, extraordinary dividend on or recapitalization, reorganization, merger or consolidation. However, the warrants will not
be adjusted for issuances of common stock at a price below their respective exercise prices.
Representative’s Warrants
We granted to Maxim Group LLC, the representative
of the underwriters, warrants to purchase a number of shares of common stock equal to 5.0% of the total number of shares of common
stock sold in this offering at a price equal to 110% of the price per share of the common stock sold in this offering. The warrants
are not redeemable and have substantially similar terms as those warrants sold to purchasers in the offering.
UNDERWRITING
We entered into an
underwriting agreement with Maxim Group LLC acting as the sole book-running manager and sole representative for the underwriters
named below. Subject to the terms and conditions of the underwriting agreement, the underwriters named below agreed to purchase,
and we agreed to sell to them, the number of shares of common stock and warrants to purchase common stock at the public offering
price, less the underwriting discounts and commissions, as set forth on the cover page of this prospectus and as indicated below
(adjusted for the 1-for-5 reverse stock split effective June 24, 2016):
|
Underwriter
|
|
|
Number of Shares
|
|
|
Maxim Group LLC
|
|
|
625,000
|
|
|
Dawson James Securities, Inc.
|
|
|
625,000
|
|
|
Total
|
|
|
1,250,000
|
|
The underwriting agreement
provides that the obligations of the underwriters to pay for and accept delivery of the shares and warrants that were offered by
this prospectus are subject to the approval of certain legal matters by their counsel and to other conditions. The underwriters
are obligated to take and pay for all of the shares and warrants offered by this prospectus if any such shares and warrants are
taken, other than those shares and warrants covered by the over-allotment option described below.
Over-Allotment
Option
We granted to the
underwriters an option, exercisable no later than 45 calendar days after the date of the underwriting agreement to purchase 187,500
additional shares of common stock at a price, after the underwriting discount, of $4.554
per five shares, and/or
703,125
warrants to purchase 140,625 shares of common
stock at a price, after the underwriting discount, of $0.046 per 3.75 warrants from us to cover over-allotments. On January 21,
2015, the underwriters exercised their over-allotment option with respect to 703,125 warrants. On March 4, 2015, the underwriters
exercised their over-allotment option with respect to 26,900 shares of common stock.
Commissions
We agreed to pay the
underwriters (i) a cash fee equal to 8% of the aggregate gross proceeds raised in this offering and (ii) warrants to purchase that
number of shares of our common stock equal to an aggregate of 5% of the shares of common stock sold in the offering (or 71,875
shares, assuming the over-allotment option is fully exercised). Such underwriters’ warrants shall have an exercise price
equal to $5.50 per share, which is 110% of the public offering price, terminate five years after the effective date of the
registration statement of which this prospectus forms a part, and otherwise have the same terms as the warrants sold in this offering
except that (1) they will not be subject to redemption by the Company and (2) they will provide for limited “piggyback”
registration rights with respect to the underlying shares during the three year period commencing six months after the effective
date of this offering in the event we fail to keep the registration statement, of which this prospectus forms a part, current.
Such underwriters’ warrants will be subject to FINRA Rule 5110(g)(1) in that, except as otherwise permitted by FINRA rules,
for a period of 180 days following the effective date of the registration statement, of which this prospectus forms a part, the
underwriters’ warrants shall not be (A) sold, transferred, assigned, pledged, or hypothecated, or (B) the subject of any
hedging, short sale, derivative, put, or call transaction that would result in the effective economic disposition of the securities
by any person.
The representative
has advised us that the underwriters propose to offer the shares and warrants directly to the public at the public offering price
set forth on the cover of this prospectus. In addition, the representative may offer some of the shares and warrants to other securities
dealers at such price less a concession of up to $0.1875 per share. After the offering to the public, the offering price and other
selling terms may be changed by the representative without changing the Company’s proceeds from the underwriters’ purchase
of the shares and warrants.
The following table
summarizes the public offering price per share and per warrant, underwriting commissions and proceeds before expenses to us assuming
both no exercise and full exercise of the underwriters’ option to purchase additional shares and warrants. The underwriting
commissions are equal to the public offering price per share less the amount per share the underwriters pay us for the shares and
warrants. All numbers have been adjusted for a 1 – for – 5 reverse stock split effective June 24, 2016.
|
|
|
Per
5 Shares
|
|
|
Per 3.75
of a
Warrant (1)
|
|
|
Total Without
Over-Allotment
|
|
|
Total Assuming
Full
Over-Allotment
|
|
|
Public Offering price
|
|
$
|
4.95
|
|
|
$
|
0.05
|
|
|
$
|
6,250,000
|
|
|
$
|
7,187,500
|
|
|
Underwriting discounts and commissions (2)
|
|
$
|
0.0396
|
|
|
$
|
0.004
|
|
|
$
|
500,000
|
|
|
$
|
575,000
|
|
|
Proceeds, before expenses, to us
|
|
$
|
4.554
|
|
|
$
|
0.046
|
|
|
$
|
5,750,000
|
|
|
$
|
6,612,500
|
|
|
(1)
|
Five shares of common stock were sold together with 3.75 of a warrant, with each five full warrants being exercisable for the purchase of one share of common stock.
|
|
(2)
|
The fees shown do not include the warrant to purchase shares of common stock issuable to the underwriters at closing.
|
We estimate that the
total expenses of the offering, including registration, filing and listing fees, printing fees and legal and accounting expenses,
but excluding underwriting discounts and commissions, were approximately $485,000, all of which are payable by us.
Lock-Up Agreements
We and each of our
officers and directors aggregating approximately 0.4% of our outstanding shares have agreed, subject to certain exceptions, not
to offer, issue, sell, contract to sell, encumber, grant any option for the sale of or otherwise dispose of any shares of our common
stock or other securities convertible into or exercisable or exchangeable for shares of our common stock for a period of six months
after the effective date of the registration statement of which this prospectus is a part without the prior written consent of
Maxim Group LLC.
Maxim may in its sole
discretion and at any time without notice release some or all of the shares subject to lock-up agreements prior to the expiration
of the lock-up period. When determining whether or not to release shares from the lock-up agreements, the representative will consider,
among other factors, the security holder’s reasons for requesting the release, the number of shares for which the release
is being requested and market conditions at the time.
Price Stabilization,
Short Positions and Penalty Bids
In connection with
this offering, the underwriters may engage in transactions that stabilize, maintain or otherwise affect the price of our common
stock. Specifically, the underwriters may over-allot in connection with this offering by selling more shares and warrants than
are set forth on the cover page of this prospectus. This creates a short position in our common stock for its own account. The
short position may be either a covered short position or a naked short position. In a covered short position, the number of shares
common stock or warrants over-allotted by the underwriters is not greater than the number of shares of common stock or warrants
that they may purchase in the over-allotment option. In a naked short position, the number of shares of common stock or warrants
involved is greater than the number of shares common stock or warrants in the over-allotment option. To close out a short position,
the underwriters may elect to exercise all or part of the over-allotment option. The underwriters may also elect to stabilize the
price of our common stock and/or warrants, or reduce any short position by bidding for, and purchasing, common stock and/or warrants in
the open market.
The underwriters may
also impose a penalty bid. This occurs when a particular underwriter or dealer repays selling concessions allowed to it for distributing
a security in this offering because the underwriter repurchases that security in stabilizing or short covering transactions.
Finally, the underwriters
may bid for, and purchase, shares of our common stock in market making transactions, including “passive” market making
transactions as described below.
These activities may
stabilize or maintain the market price of our common stock at a price that is higher than the price that might otherwise exist
in the absence of these activities. The underwriters are not required to engage in these activities, and may discontinue any of
these activities at any time without notice. These transactions may be effected on NASDAQ, in the over-the-counter market, or otherwise.
In connection with
this offering, the underwriters and selling group members, if any, or their affiliates may engage in passive market making transactions
in our common stock immediately prior to the commencement of sales in this offering, in accordance with Rule 103 of Regulation
M under the Exchange Act. Rule 103 generally provides that:
|
|
·
|
a passive market maker may not effect transactions or display bids for our common stock in excess of the highest independent bid price by persons who are not passive market makers;
|
|
|
·
|
net purchases by a passive market maker on each day are generally limited to 30% of the passive market maker’s average daily trading volume in our common stock during a specified two-month prior period or 200 shares, whichever is greater, and must be discontinued when that limit is reached; and
|
|
|
·
|
passive market making bids must be identified as such.
|
Other Terms
We agreed to bear
the cost of all actual expenses related to the offering, including, without limitation, all filing fees and communication expenses
relating to the registration of the shares to be sold in the offering. We provided Maxim Group LLC an advance of $25,000 for its
anticipated out-of-pocket accountable expenses and will provide an additional advance of $25,000 upon written request by Maxim
Group LLC which shall also be applied to its anticipated out-of-pocket expenses, for aggregate advances of $50,000. Maxim Group
LLC will reimburse us for any remaining portion of the advance to the extent such monies were not used for out-of-pocket accountable
expenses actually incurred if this offering is not completed. If this offering is completed, we will reimburse Maxim Group LLC
for certain out-of-pocket actual expenses related to the offering, including legal fees and expenses incurred to clear the offering
with FINRA, including background searches of our officers and directors and roadshow expenses, up to a maximum aggregate reimbursable
amount of $125,000 (of which $100,000 can be allocated to legal expenses and $25,000 for non-legal expenses). In addition, we will
be responsible for the costs and expenses of “tombstone” advertisements not to exceed $5,000 and commemorative lucite
memorabilia valued up to $1,500.
We granted Maxim Group LLC a right of first
refusal for future public and private financings (excluding (i) at-the-market offerings, (ii) funding from a strategic investor,
or (iii) equity issued to purchase business assets or to acquire a strategic company) for a period of 12 months from January 20,
2015.
Pulmatrix/Ruthigen Transaction
On March 23, June 16 and 17, 2015, we closed
the sale of our shares in Pulmatrix (formerly Ruthigen) to several investors for an aggregate purchase price of $5.5 million upon
the merger of Ruthigen with Pulmatrix, Inc. We agreed to pay Dawson James Securities, Inc. a cash fee in the amount of $200,000.
This fee will not be deemed an Item of Value under FINRA Rule 5110(c) and will not be included in the aggregate underwriting compensation
for this offering.
Indemnification
We have agreed to
indemnify the underwriters against liabilities relating to the offering arising under the Securities Act and the Exchange Act,
liabilities arising from breaches of some or all of the representations and warranties contained in the underwriting agreement,
and to contribute to payments that the underwriters may be required to make for these liabilities.
Electronic Distribution
A prospectus in electronic
format may be made available on a website maintained by the representatives of the underwriters and may also be made available
on a website maintained by other underwriters. The underwriters may agree to allocate a number of shares to underwriters for sale
to their online brokerage account holders. Internet distributions will be allocated by the representatives of the underwriters
to underwriters that may make Internet distributions on the same basis as other allocations. In connection with the offering, the
underwriters or syndicate members may distribute prospectuses electronically. No forms of electronic prospectus other than prospectuses
that are printable as Adobe® PDF will be used in connection with this offering.
The underwriters have
informed us that they do not expect to confirm sales of shares and warrants offered by this prospectus to accounts over which they
exercise discretionary authority.
Other than the prospectus
in electronic format, the information on any underwriter’s website and any information contained in any other website maintained
by an underwriter is not part of the prospectus or the registration statement of which this prospectus forms a part, has not been
approved and/or endorsed by us or any underwriter in its capacity as underwriter and should not be relied upon by investors.
LEGAL
MATTERS
Trombly
Business Law, PC passed upon the validity of the securities offered hereby. Certain legal matters in connection with this offering
were passed upon for the underwriters by Ellenoff Grossman & Schole LLP.
EXPERTS
The consolidated
financial statements as of and for the years ended March 31, 2016 and 2015 included in this Registration Statement have been so
included in reliance on the report, which includes an explanatory paragraph as to the Company’s ability to continue as a
going concern, of Marcum LLP, an independent registered public accounting firm, appearing elsewhere herein and in the Registration
Statement, given on the authority of said firm as experts in auditing and accounting.
INTERESTS
OF NAMED EXPERTS AND COUNSEL
No expert or counsel
named in this registration statement as having prepared or certified any part of this registration statement or having given an
opinion upon the validity of the securities being registered or upon other legal matters in connection with the registration or
offering of the common stock was employed for such purpose on a contingency basis, or had, or is to receive, in connection with
this offering, a substantial interest, direct or indirect, in us or any of our parents or subsidiaries, nor was any such person
connected with us or any of our parents or subsidiaries as a promoter, managing or principal underwriter, voting trustee, director,
officer, or employee.
WHERE
YOU CAN FIND MORE INFORMATION
We have filed with the Securities
and Exchange Commission, Washington, D.C., 20549, under the Securities Act of 1933, a registration statement on Form S-1 relating
to the securities offered hereby. This prospectus does not contain all of the information set forth in the registration statement
and the exhibits and schedules thereto. For further information with respect to our company and the securities we are offering
by this prospectus you should refer to the registration statement, including the exhibits and schedules thereto. You may inspect
a copy of the registration statement without charge at the Public Reference Section of the Securities and Exchange Commission at
Room 1024, 450 Fifth Street, N.W., Washington, D.C. 20549. The public may obtain information on the operation of the Public Reference
Room by calling the Securities and Exchange Commission at 1-800-SEC-0330. The Securities and Exchange Commission also maintains
an Internet site that contains reports, proxy and information statements and other information regarding registrants that file
electronically with the Securities and Exchange Commission. The Securities and Exchange Commission's World Wide Web address is
http://www.sec.gov.
We file periodic reports, proxy statements
and other information with the Securities and Exchange Commission in accordance with requirements of the Exchange Act. These periodic
reports, proxy statements and other information are available for inspection and copying at the regional offices, public reference
facilities and Internet site of the Securities and Exchange Commission referred to above. In addition, you may request a copy of
any of our periodic reports filed with the Securities and Exchange Commission at no cost, by writing or telephoning us at the following
address:
Investor Relations
Oculus Innovative Sciences, Inc.
1129 N. McDowell Blvd.
Petaluma, CA 94954
(707) 283-0550
Investors and others should note that we announce
material financial information using our company website: www.oculusis.com, our investor relations website: ir.oculusis.com, SEC
filings, press releases, public conference calls and webcasts. Information about Oculus, our business, and our results of operations
may also be announced by posts on the following social media channels:
|
|
·
|
Oculus corporate blog: http://oculusis.com/dialogue/
|
|
|
·
|
Oculus Facebook page :www.facebook.com/oculusinnovativesciences
|
|
|
·
|
Dan McFadden’s Twitter feed: http://twitter.com/dmcfaddenocls. Mr. McFadden is the Vice President of Public and Investor Relations of our Company.
|
The information that we post on these social
media channels could be deemed to be material information. Therefore, we encourage investors, the media, and others interested
in Oculus to review the information that we post on these social media channels. These social media channels may be updated from
time to time on Oculus’ investor relations website.
The information on or accessible through our
websites and social media channels is not incorporated by reference in this prospectus.
You should rely only on the information
contained in or incorporated by reference or provided in this prospectus or any supplement to this prospectus. We have not authorized
anyone else to provide you with different information. We are not making an offer of these securities in any state where the offer
is not permitted. You should not assume the information in this prospectus is accurate as of any date other than the date on the
front of this prospectus.
DISCLOSURE
OF COMMISSION POSITION ON INDEMNIFICATION FOR SECURITIES ACT LIABILITIES
Insofar as indemnification for liabilities
arising under the Securities Act may be permitted to our directors, officers, or persons controlling our Company pursuant to the
foregoing provisions, we have been informed that in the opinion of the Securities and Exchange Commission, such indemnification
is against public policy as expressed in the Securities Act and is therefore unenforceable.
Oculus Innovative Sciences, Inc.
Index to Consolidated Financial Statements
|
|
|
Page
|
|
|
|
|
|
Report of Independent Registered Public Accounting Firm
|
|
F-2
|
|
|
|
|
|
Consolidated Balance Sheets as of March 31, 2016 and 2015
|
|
F-3
|
|
|
|
|
|
Consolidated Statements of Comprehensive (Loss) Income for the years ended March 31, 2016 and 2015
|
|
F-4
|
|
|
|
|
|
Consolidated Statements of Changes in Stockholders’ Equity for the years ended March 31, 2016 and 2015
|
|
F-5
|
|
|
|
|
|
Consolidated Statements of Cash Flows for the years ended March 31, 2016 and 2015
|
|
F-6
|
|
|
|
|
|
Notes to Consolidated Financial Statements
|
|
F-7
|
|
|
|
|
|
Consolidated Balance Sheets as of June 30, 2016 and March 31, 2016
|
|
F-26
|
|
|
|
|
|
Consolidated Statements of Comprehensive (Loss) Income for the quarters ended June 30, 2016 and 2015
|
|
F-
27
|
|
|
|
|
|
Consolidated Statements of Cash Flows for the quarters ended June 30, 2016 and 2015
|
|
F-
28
|
|
|
|
|
|
Notes to Consolidated Financial Statements
|
|
F-
29
|
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING
FIRM
To the Audit Committee of the
Board of Directors and Shareholders
of Oculus Innovative Sciences, Inc.
We have audited the accompanying consolidated
balance sheets of Oculus Innovative Sciences, Inc. and Subsidiaries (the “Company”) as of March 31, 2016 and 2015,
and the related consolidated statements of comprehensive loss, changes in stockholders’ equity and cash flows for the years
then ended. These financial statements are the responsibility of the Company’s management. Our responsibility is to express
an opinion on these financial statements based on our audits.
We conducted our audits in accordance with
the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform
the audits to obtain reasonable assurance about whether the financial statements are free of material misstatement. The Company
is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audits
included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate
in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control
over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence
supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates
made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide
a reasonable basis for our opinion.
In our opinion, the financial statements referred
to above present fairly, in all material respects, the consolidated financial position of Oculus Innovative Sciences, Inc. and
Subsidiaries, as of March 31, 2016 and 2015, and the consolidated results of its operations and its cash flows for the years then
ended in conformity with accounting principles generally accepted in the United States of America.
The accompanying financial statements have
been prepared assuming that the Company will continue as a going concern. As more fully discussed in Note 2, the Company has incurred
significant net losses and needs to raise additional funds to meet its obligations and sustain its operations. These conditions
raise substantial doubt about the Company’s ability to continue as a going concern. Management’s plans with regard
to these matters are also described in Note 2. The financial statements do not include any adjustments that might result from the
outcome of this uncertainty.
/s/ Marcum LLP
Marcum LLP
New York, NY
June 21, 2016
OCULUS INNOVATIVE SCIENCES, INC. AND SUBSIDIARIES
CONSOLIDATED BALANCE
SHEETS
|
|
|
March 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
|
|
(In thousands, except share
and per share amounts)
|
|
|
ASSETS
|
|
|
|
|
|
|
|
|
|
Current assets:
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
7,469
|
|
|
$
|
6,136
|
|
|
Accounts receivable, net
|
|
|
2,274
|
|
|
|
1,517
|
|
|
Inventories, net
|
|
|
1,640
|
|
|
|
1,402
|
|
|
Prepaid expenses and other current assets
|
|
|
1,505
|
|
|
|
592
|
|
|
Total current assets
|
|
|
12,888
|
|
|
|
9,647
|
|
|
Property and equipment, net
|
|
|
850
|
|
|
|
795
|
|
|
Long-term investment
|
|
|
–
|
|
|
|
4,538
|
|
|
Other assets
|
|
|
65
|
|
|
|
68
|
|
|
Total assets
|
|
$
|
13,803
|
|
|
$
|
15,048
|
|
|
|
|
|
|
|
|
|
|
|
|
LIABILITIES AND STOCKHOLDERS’ EQUITY
|
|
|
|
|
|
|
|
|
|
Current liabilities:
|
|
|
|
|
|
|
|
|
|
Accounts payable
|
|
$
|
1,337
|
|
|
$
|
932
|
|
|
Accrued expenses and other current liabilities
|
|
|
1,526
|
|
|
|
782
|
|
|
Deferred revenue
|
|
|
574
|
|
|
|
769
|
|
|
Current portion of long-term debt
|
|
|
114
|
|
|
|
87
|
|
|
Derivative liabilities
|
|
|
–
|
|
|
|
11
|
|
|
Total current liabilities
|
|
|
3,551
|
|
|
|
2,581
|
|
|
Deferred revenue, less current portion
|
|
|
112
|
|
|
|
413
|
|
|
Total liabilities
|
|
|
3,663
|
|
|
|
2,994
|
|
|
Commitments and Contingencies (Note 12)
|
|
|
|
|
|
|
|
|
|
Stockholders’ Equity
|
|
|
|
|
|
|
|
|
|
Convertible preferred stock, $0.0001 par value; 714,286 shares authorized, none issued and outstanding at March 31, 2016 and March 31, 2015, respectively
|
|
|
–
|
|
|
|
–
|
|
|
Common stock, $0.0001 par value; 60,000,000 shares authorized at March 31, 2016 and March 31, 2015, 20,984,369 and 15,045,080 shares issued and outstanding at March 31, 2016 and March 31, 2015, respectively (Note 13)
|
|
|
2
|
|
|
|
2
|
|
|
Additional paid-in capital
|
|
|
166,367
|
|
|
|
157,772
|
|
|
Accumulated deficit
|
|
|
(152,375
|
)
|
|
|
(142,213
|
)
|
|
Accumulated other comprehensive loss
|
|
|
(3,854
|
)
|
|
|
(3,507
|
)
|
|
Total stockholders’ equity
|
|
|
10,140
|
|
|
|
12,054
|
|
|
Total liabilities and stockholders’ equity
|
|
$
|
13,803
|
|
|
$
|
15,048
|
|
The accompanying footnotes are an integral part
of these consolidated financial statements.
OCULUS INNOVATIVE SCIENCES, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF COMPREHENSIVE
LOSS
|
|
|
Year Ended March 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
|
|
(In thousands, except
per share amounts)
|
|
|
Revenues
|
|
|
|
|
|
|
|
|
|
Product
|
|
$
|
13,042
|
|
|
$
|
9,939
|
|
|
Product licensing fees and royalties
|
|
|
981
|
|
|
|
3,056
|
|
|
Service
|
|
|
1,061
|
|
|
|
859
|
|
|
Total revenues
|
|
|
15,084
|
|
|
|
13,854
|
|
|
Cost of revenues
|
|
|
|
|
|
|
|
|
|
Product
|
|
|
6,993
|
|
|
|
5,908
|
|
|
Service
|
|
|
881
|
|
|
|
658
|
|
|
Total cost of revenues
|
|
|
7,874
|
|
|
|
6,566
|
|
|
Gross profit
|
|
|
7,210
|
|
|
|
7,288
|
|
|
Operating expenses
|
|
|
|
|
|
|
|
|
|
Research and development
|
|
|
1,806
|
|
|
|
1,533
|
|
|
Selling, general and administrative
|
|
|
15,556
|
|
|
|
12,414
|
|
|
Total operating expenses
|
|
|
17,362
|
|
|
|
13,947
|
|
|
Loss from operations
|
|
|
(10,152
|
)
|
|
|
(6,659
|
)
|
|
Interest expense
|
|
|
(3
|
)
|
|
|
(3
|
)
|
|
Interest income
|
|
|
2
|
|
|
|
1
|
|
|
Gain due to change in fair value of derivative liabilities
|
|
|
11
|
|
|
|
3,164
|
|
|
Impairment loss on long-term investment (See Note 3)
|
|
|
–
|
|
|
|
(4,650
|
)
|
|
Other expense, net
|
|
|
(20
|
)
|
|
|
(56
|
)
|
|
Net loss
|
|
|
(10,162
|
)
|
|
|
(8,203
|
)
|
|
Net loss per common share: basic and diluted
|
|
$
|
(0.62
|
)
|
|
$
|
(0.85
|
)
|
|
Weighted-average number of shares used in per common share calculations:
|
|
|
|
|
|
|
|
|
|
Basic and diluted
|
|
|
16,444
|
|
|
|
9,657
|
|
|
Other comprehensive loss
|
|
|
|
|
|
|
|
|
|
Net loss
|
|
$
|
(10,162
|
)
|
|
$
|
(8,203
|
)
|
|
Foreign currency translation adjustments
|
|
|
(347
|
)
|
|
|
(438
|
)
|
|
Comprehensive loss
|
|
$
|
(10,509
|
)
|
|
$
|
(8,641
|
)
|
The accompanying footnotes are an integral part
of these consolidated financial statements.
OCULUS INNOVATIVE SCIENCES, INC.
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’
EQUITY
For the Years Ended March 31, 2016 and 2015
(In thousands, except share amounts)
|
|
|
Common Stock
($0.0001 par Value)
|
|
|
Additional
Paid in
|
|
|
Accumulated
|
|
|
Accumulated Other Comprehensive
|
|
|
|
|
|
|
|
Shares
|
|
|
Amount
|
|
|
Capital
|
|
|
Deficit
|
|
|
Loss
|
|
|
Total
|
|
|
Balance, March 31, 2014
|
|
|
8,160,145
|
|
|
$
|
1
|
|
|
$
|
149,141
|
|
|
$
|
(134,010
|
)
|
|
$
|
(3,069
|
)
|
|
$
|
12,063
|
|
|
Issuance of common stock in connection with At-the-Market issuances of common stock, net of commissions, expenses and other offering costs
|
|
|
467,934
|
|
|
|
–
|
|
|
|
1,341
|
|
|
|
–
|
|
|
|
–
|
|
|
|
1,341
|
|
|
Issuance of common stock and common stock purchase warrants in connection with January 26, 2015 closing of offering, net of commissions, expenses and other offering costs
|
|
|
6,384,500
|
|
|
|
1
|
|
|
|
5,443
|
|
|
|
–
|
|
|
|
–
|
|
|
|
5,444
|
|
|
Issuance of common stock for settlement of service fees
|
|
|
32,501
|
|
|
|
–
|
|
|
|
76
|
|
|
|
–
|
|
|
|
–
|
|
|
|
76
|
|
|
Stock based compensation expense, net of forfeitures
|
|
|
–
|
|
|
|
–
|
|
|
|
1,771
|
|
|
|
–
|
|
|
|
–
|
|
|
|
1,771
|
|
|
Foreign currency translation adjustment
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
(438
|
)
|
|
|
(438
|
)
|
|
Net loss
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
(8,203
|
)
|
|
|
–
|
|
|
|
(8,203
|
)
|
|
Balance, March 31, 2015
|
|
|
15,045,080
|
|
|
|
2
|
|
|
|
157,772
|
|
|
|
(142,213
|
)
|
|
|
(3,507
|
)
|
|
|
12,054
|
|
|
Issuance of common stock in connection with At-the-Market issuances of common stock, net of commissions, expenses and other offering costs
|
|
|
2,254,596
|
|
|
|
–
|
|
|
|
3,150
|
|
|
|
–
|
|
|
|
–
|
|
|
|
3,150
|
|
|
Issuance of common stock and common stock purchase warrants in connection with March 23, 2016 closing of offering, net of commissions, expenses and other offering costs
|
|
|
3,400,000
|
|
|
|
–
|
|
|
|
2,994
|
|
|
|
–
|
|
|
|
–
|
|
|
|
2,994
|
|
|
Issuance of common stock upon exercise of common stock purchase warrants
|
|
|
11,100
|
|
|
|
–
|
|
|
|
14
|
|
|
|
–
|
|
|
|
–
|
|
|
|
14
|
|
|
Issuance of common stock for settlement of service fees
|
|
|
208,519
|
|
|
|
–
|
|
|
|
286
|
|
|
|
–
|
|
|
|
–
|
|
|
|
286
|
|
|
Issuance of common stock in connection with Board Compensation
|
|
|
65,074
|
|
|
|
–
|
|
|
|
64
|
|
|
|
|
|
|
|
|
|
|
|
64
|
|
|
Issuance of common stock purchase warrants for payment of service fees
|
|
|
–
|
|
|
|
–
|
|
|
|
128
|
|
|
|
–
|
|
|
|
–
|
|
|
|
128
|
|
|
Stock based compensation, net of forfeitures
|
|
|
–
|
|
|
|
–
|
|
|
|
1,959
|
|
|
|
–
|
|
|
|
|
|
|
|
1,959
|
|
|
Foreign currency translation adjustment
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
(347
|
)
|
|
|
(347
|
)
|
|
Net loss
|
|
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
|
(10,162
|
)
|
|
|
–
|
|
|
|
(10,162
|
)
|
|
Balance, March 31, 2016
|
|
|
20,984,369
|
|
|
$
|
2
|
|
|
$
|
166,367
|
|
|
$
|
(152,375
|
)
|
|
$
|
(3,854
|
)
|
|
$
|
10,140
|
|
The accompanying footnotes are an integral part
of these consolidated financial statements.
OCULUS INNOVATIVE
SCIENCES, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CASH FLOWS
|
|
|
Year Ended March 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
|
|
(In thousands)
|
|
|
Cash flows from operating activities
|
|
|
|
|
|
|
|
|
|
Net loss
|
|
$
|
(10,162
|
)
|
|
$
|
(8,203
|
)
|
|
Adjustments to reconcile net loss to net cash used in operating activities:
|
|
|
|
|
|
|
|
|
|
Depreciation and amortization
|
|
|
244
|
|
|
|
253
|
|
|
Change in provision for doubtful accounts
|
|
|
(5
|
)
|
|
|
29
|
|
|
Change in provision for discounts, rebates, distributor fees and returns
|
|
|
470
|
|
|
|
183
|
|
|
Change in provision for obsolete inventory
|
|
|
77
|
|
|
|
141
|
|
|
Stock-based compensation
|
|
|
2,341
|
|
|
|
1,771
|
|
|
Change in fair value of derivative liabilities
|
|
|
(11
|
)
|
|
|
(3,164
|
)
|
|
Impairment loss on long-term investment
|
|
|
–
|
|
|
|
4,650
|
|
|
Foreign currency transaction (gain) loss
|
|
|
(38
|
)
|
|
|
24
|
|
|
Gain on disposal of property and equipment
|
|
|
–
|
|
|
|
(13
|
)
|
|
Changes in operating assets and liabilities:
|
|
|
|
|
|
|
|
|
|
Accounts receivable
|
|
|
(1,282
|
)
|
|
|
(143
|
)
|
|
Due from affiliate
|
|
|
–
|
|
|
|
537
|
|
|
Inventories
|
|
|
(382
|
)
|
|
|
(627
|
)
|
|
Prepaid expenses and other current assets
|
|
|
(751
|
)
|
|
|
162
|
|
|
Accounts payable
|
|
|
429
|
|
|
|
222
|
|
|
Accrued expenses and other current liabilities
|
|
|
919
|
|
|
|
(1
|
)
|
|
Deferred revenue and other liabilities
|
|
|
(595
|
)
|
|
|
(2,515
|
)
|
|
Net cash used in operating activities
|
|
|
(8,746
|
)
|
|
|
(6,694
|
)
|
|
Cash flows from investing activities:
|
|
|
|
|
|
|
|
|
|
Purchases of property and equipment
|
|
|
(345
|
)
|
|
|
(139
|
)
|
|
Proceeds from sale of long-term investment
|
|
|
4,538
|
|
|
|
963
|
|
|
Long-term deposits
|
|
|
(2
|
)
|
|
|
51
|
|
|
Net cash provided by investing activities
|
|
|
4,191
|
|
|
|
875
|
|
|
Cash flows from financing activities:
|
|
|
|
|
|
|
|
|
|
Proceeds from issuance of common stock and common stock purchase warrants in offerings, net of offering costs
|
|
|
6,144
|
|
|
|
6,785
|
|
|
Proceeds from issuance of common stock upon exercise of stock options and warrants
|
|
|
14
|
|
|
|
–
|
|
|
Principal payments on long-term debt
|
|
|
(119
|
)
|
|
|
(176
|
)
|
|
Net cash provided by financing activities
|
|
|
6,039
|
|
|
|
6,609
|
|
|
Effect of exchange rate on cash and cash equivalents
|
|
|
(151
|
)
|
|
|
(134
|
)
|
|
Net increase in cash and cash equivalents
|
|
|
1,333
|
|
|
|
656
|
|
|
Cash and cash equivalents, beginning of year
|
|
|
6,136
|
|
|
|
5,480
|
|
|
Cash and cash equivalents, end of year
|
|
$
|
7,469
|
|
|
$
|
6,136
|
|
|
Supplemental disclosure of cash flow information:
|
|
|
|
|
|
|
|
|
|
Cash paid for interest
|
|
$
|
3
|
|
|
$
|
3
|
|
|
Non-cash operating and financing activities:
|
|
|
|
|
|
|
|
|
|
Insurance premiums financed
|
|
$
|
146
|
|
|
$
|
116
|
|
|
Issuance of common stock to settle obligations
|
|
$
|
96
|
|
|
$
|
76
|
|
The accompanying footnotes are an integral part
of these consolidated financial statements.
OCULUS INNOVATIVE SCIENCES, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
NOTE 1 – Organization and
Recent Developments
Organization
Oculus Innovative Sciences, Inc. (the “Company”)
was incorporated under the laws of the State of California in April 1999 and was reincorporated under the laws of the State of
Delaware in December 2006. The Company’s principal office is located in Petaluma, California. The Company is a specialty
pharmaceutical company that develops and markets solutions for the treatment of dermatological conditions and advanced tissue care.
The Company’s products, which are sold throughout the United States and 39 countries around the world, have improved patient
outcomes for more than five million patients globally by reducing infections, itch, pain, scarring, odor and harmful inflammatory
responses.
NOTE 2 – Liquidity and Financial
Condition
The Company reported a net loss of $10,162,000
for the year ended March 31, 2016. At March 31, 2016 and March 31, 2015, the Company’s accumulated deficit amounted to $152,375,000
and $142,213,000, respectively. The Company had working capital of $9,337,000 and $7,066,000 as of March 31, 2016 and March 31,
2015, respectively. The Company expects to continue incurring losses for the foreseeable future and may need to raise additional
capital to pursue its product development initiatives, penetrate markets for the sale of its products and continue as a going concern.
On March 18, 2016, the Company entered
into an underwriting agreement with Dawson James Securities, Inc. with respect to the issuance and sale of an aggregate of
3,400,000 units, each unit consisting of one share of common stock, par value $0.0001 per share, together with one quarter or
25% of one warrant to purchase one share of common stock at an exercise price equal to $1.00 per share, in an underwritten
public offering. The public offering price for each unit, consisting of one share of common stock together with one quarter
or 25% of one warrant, was $1.00. Because the Company is prohibited from issuing fractional shares, the warrants can only be
exercised in lots of four, which means that each holder must exercise four March 2016 Warrants to receive one share of common
stock, or a total of 850,000 shares. The warrants have an initial exercise price of $1.00 per share and have a term of three
years. Pursuant to the underwriting agreement, the Company paid Dawson James Securities, Inc. a cash fee equal to 8% of the
aggregate gross proceeds raised in this offering and also paid $50,000 in legal fees and expenses of the underwriter’s
legal counsel. The gross proceeds from the sale of the shares of common stock and the warrants were $3,400,000, and net
proceeds were $2,994,000 after deducting underwriting commissions and other estimated offering expenses. We also issued
warrants to purchase up to 250,000 shares of our common stock to Dawson James Securities, Inc. related to a service
agreement.
Pursuant to an At-the-Market Issuance
Sales Agreement with MLV & Co. LLC dated April 2, 2014, the Company may issue and sell shares of common stock having an
aggregate offering price of up to $9,159,000 from time to time through MLV acting as the Company’s sales agent. To
date, the Company has raised an aggregate $4,706,000 in connection with this agreement. During the year ended March 31, 2016,
the Company sold 2,254,596 shares of common stock for gross proceeds of $3,263,000 and net proceeds of $3,150,000 after
deducting commissions and other offering expenses.
Management believes that the Company has access
to additional capital resources through possible public or private equity offerings, debt financings, corporate collaborations
or other means; however, the Company cannot provide any assurance that other new financings will be available on commercially acceptable
terms, if needed. If the economic climate in the U.S. deteriorates, the Company’s ability to raise additional capital could
be negatively impacted. If the Company is unable to secure additional capital, it may be required to curtail its research and development
initiatives and take additional measures to reduce costs in order to conserve its cash in amounts sufficient to sustain operations
and meet its obligations. These measures could cause significant delays in the Company’s efforts to commercialize its products,
which is critical to the realization of its business plan and the future operations of the Company. These matters raise substantial
doubt about the Company’s ability to continue as a going concern. The accompanying consolidated financial statements
do not include any adjustments that may be necessary should the Company be unable to continue as a going concern.
NOTE 3 – Summary of Significant Accounting Policies
Principles of Consolidation
The accompanying consolidated financial statements
include the accounts of the Company and its wholly-owned subsidiaries, Aquamed Technologies, Inc. (“Aquamed”), Oculus
Technologies of Mexico S.A. de C.V. (“OTM”), and Oculus Innovative Sciences Netherlands, B.V. (“OIS Europe”).
Aquamed has no current operations. All significant intercompany accounts and transactions have been eliminated in consolidation.
Use of Estimates
The preparation of consolidated financial statements
in conformity with accounting principles generally accepted in the United States of America requires management to make estimates
and assumptions that affect the reported amounts of assets and liabilities and disclosures of contingent liabilities at the dates
of the consolidated financial statements and the reported amounts of revenues and expenses during the reporting periods.
Actual results could differ from these estimates. Significant estimates and assumptions include reserves and write-downs related
to receivables and inventories, the recoverability of long-lived assets, the valuation allowance relating to the Company’s
deferred tax assets, valuation of equity and derivative instruments, and the estimated amortization periods of upfront product
licensing fees received from customers. Periodically, the Company evaluates and adjusts estimates accordingly.
Reclassifications
Certain prior period amounts have been reclassified
for comparative purposes to conform to the fiscal 2016 presentation. These reclassifications have no impact on the Company’s
previously reported net loss.
Revenue Recognition and Accounts Receivable
The Company generates revenue from sales of
its products to a customer base including hospitals, medical centers, doctors, pharmacies, distributors and wholesalers. The Company
sells products directly to end users and to distributors. The Company also entered into agreements to license its technology and
products.
The Company also provides regulatory compliance
testing and quality assurance services to medical device and pharmaceutical companies.
The Company records revenue when (i) persuasive
evidence of an arrangement exists, (ii) delivery has occurred, (iii) the fee is fixed or determinable, and (iv) collectability
of the sale is reasonably assured.
The Company requires all product sales to be
supported by evidence of a sale transaction that clearly indicates the selling price to the customer, shipping terms and payment
terms. Evidence of an arrangement generally consists of a contract or purchase order approved by the customer. The Company has
ongoing relationships with certain customers from which it customarily accepts orders by telephone in lieu of purchase orders.
The Company recognizes revenue at the time
it receives confirmation that the goods were either tendered at their destination, when shipped “FOB destination,”
or transferred to a shipping agent, when shipped “FOB shipping point.” Delivery to the customer is deemed to have occurred
when the customer takes title to the product. Generally, title passes to the customer upon shipment, but could occur when the customer
receives the product based on the terms of the agreement with the customer.
The selling prices of all goods are fixed,
and agreed to with the customer, prior to shipment. Selling prices are generally based on established list prices. The right to
return product is customarily based on the terms of the agreement with the customer. The Company estimates and accrues for potential
returns and records this as a reduction of revenue in the same period the related revenue is recognized. Additionally, distribution
fees are paid to certain wholesale distributors based on contractually determined rates. The Company estimates and accrues the
fee on shipment to the respective wholesale distributors and recognizes the fee as a reduction of revenue in the same period the
related revenue is recognized. The Company also offers cash discounts to certain customers, generally 2% of the sales price, as
an incentive for prompt payment. The Company accounts for cash discounts by reducing accounts receivable by the prompt pay discount
amount and recognizes the discount as a reduction of revenue in the same period the related revenue is recognized. Additionally,
the Company participates in certain rebate programs which provide discounted prescriptions to qualified patients. The Company contracts
a third-party to administer the program. The Company estimates and accrues for future rebates based on historical data for rebate
redemption rates and the historical value of redemptions. Rebates are recognized as a reduction of revenue in the same period the
related revenue is recognized.
The Company evaluates the creditworthiness
of new customers and monitors the creditworthiness of its existing customers to determine whether an event or changes in their
financial circumstances would raise doubt as to the collectability of a sale at the time in which a sale is made. Payment terms
on sales made in the United States are generally 30 days and are extended up to 90 days for initial product launches, payment
terms internationally generally range from prepaid prior to shipment to 90 days.
In the event a sale is made to a customer under
circumstances in which collectability is not reasonably assured, the Company either requires the customer to remit payment prior
to shipment or defers recognition of the revenue until payment is received. The Company maintains a reserve for amounts which may
not be collectible due to risk of credit losses.
Product license revenue is generated through
agreements with strategic partners for the commercialization of Microcyn® products. The terms of the agreements sometimes include
non-refundable upfront fees. The Company analyzes multiple element arrangements to determine whether the elements can be separated.
Analysis is performed at the inception of the arrangement and as each product is delivered. If a product or service is not separable,
the combined deliverables are accounted for as a single unit of accounting and recognized over the performance obligation period.
When appropriate, the Company defers recognition
of non-refundable upfront fees. If the Company has continuing performance obligations then such up-front fees are deferred and
recognized over the period of continuing involvement.
The Company recognizes royalty revenues from
licensed products upon the sale of the related products.
Revenue from consulting contracts is recognized
as services are provided. Revenue from testing contracts is recognized as tests are completed and a final report is sent to the
customer.
Sales Tax and Value Added Taxes
The Company accounts for sales taxes and value
added taxes imposed on its goods and services on a net basis.
Cash and Cash Equivalents
The Company considers all highly liquid investments
with an original maturity of three months or less when purchased to be cash equivalents. Cash equivalents may be invested in money
market funds, commercial paper, variable rate demand instruments, and certificates of deposits.
Long-Term Investments
The Company accounted for its ownership of
shares of Ruthigen, Inc. (“Ruthigen”) common stock at cost in accordance with Accounting Standards Codification (“ASC”)
325-20 as a result of (a) the restrictions on voting the shares held, (b) the Company having no representation on the Ruthigen
Board of Directors, (c) the Company’s inability to set policy at Ruthigen (d) the Company having no further commitments for
funding the operations of Ruthigen and (e) the restrictions on transferability of its shares.
The Company’s long-term investments consisted
of the Company’s ownership of 1,650,000 shares of Ruthigen common stock at March 31, 2015. During the year ended March 31,
2016, the Company sold its remaining 1,650,000 shares of Ruthigen common stock for proceeds of $4,537,500 pursuant to a securities
purchase agreement with several investors. Additionally, during the year ended March 31, 2016, the Company paid a $165,000 banker
fee related to the sale transaction.
Concentration of Credit Risk and Major
Customers
Financial instruments that potentially subject
the Company to concentration of credit risk consist principally of cash, cash equivalents and accounts receivable. Cash and cash
equivalents are maintained in financial institutions in the United States, Mexico and the Netherlands. The Company is exposed to
credit risk in the event of default by these financial institutions for amounts in excess of the Federal Deposit Insurance Corporation
insured limits. Cash and cash equivalents held in foreign banks are intentionally kept at minimal levels, and therefore have minimal
credit risk associated with them.
The Company grants credit to its business customers,
which are primarily located in Mexico, Europe and the United States. Collateral is generally not required for trade receivables.
The Company maintains allowances for potential credit losses. At March 31, 2016, one customer represented 33% of the net accounts
receivable balance. At March 31, 2016, one customer represented 40%, one customer represented 15%, one customer represented 14%
and two customers each represented 12% of net revenues. At March 31, 2015, one customer represented 56%, and one customer represented
14% of the net accounts receivable balance. During the year ended March 31, 2015, one customer represented 47% of net revenues.
Accounts Receivable
Trade accounts receivable are recorded net
of allowances for cash discounts for prompt payment, doubtful accounts, and sales returns. Estimates for cash discounts and sales
returns are based on analysis of contractual terms and historical trends.
The Company’s policy is to reserve for
uncollectible accounts based on its best estimate of the amount of probable credit losses in its existing accounts receivable.
The Company periodically reviews its accounts receivable to determine whether an allowance for doubtful accounts is necessary based
on an analysis of past due accounts and other factors that may indicate that the realization of an account may be in doubt. Other
factors that the Company considers include its existing contractual obligations, historical payment patterns of its customers and
individual customer circumstances, an analysis of days sales outstanding by customer and geographic region, and a review of the
local economic environment and its potential impact on government funding and reimbursement practices. Account balances deemed
to be uncollectible are charged to the allowance after all means of collection have been exhausted and the potential for recovery
is considered remote. The allowance for doubtful accounts represents probable credit losses at March 31, 2016 and March 31, 2015
in the amounts of $15,000 and $20,000, respectively. Additionally at March 31, 2016 and March 31, 2015 the Company has allowances
of $653,000 and $183,000, respectively, related to potential discounts, returns, distributor fees and rebates. The allowances are
included in Accounts Receivable, net in the accompanying consolidated balance sheets.
Inventories
Inventories are stated at the lower of cost,
cost being determined on a standard cost basis (which approximates actual cost on a first-in, first-out basis), or market.
Due to changing market conditions, estimated
future requirements, age of the inventories on hand and production of new products, the Company regularly reviews inventory quantities
on hand and records a provision to write down excess and obsolete inventory to its estimated net realizable value. The Company
recorded reserves to reduce the carrying amounts of inventories to their net realizable value in the amounts of $164,000 and $87,000
at March 31, 2016 and 2015, respectively, which is included in cost of product revenues on the Company’s accompanying consolidated
statements of comprehensive loss.
Fair Value of Financial Assets and Liabilities
Financial instruments, including cash and cash
equivalents, accounts receivable, accounts payable and accrued expenses and other liabilities are carried at cost, which management
believes approximates fair value due to the short-term nature of these instruments. The fair value of capital lease obligations
and equipment loans approximates their carrying amounts as a market rate of interest is attached to their repayment. The Company
measures the fair value of financial assets and liabilities based on the exchange price that would be received for an asset or
paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly
transaction between market participants on the measurement date. The Company maximizes the use of observable inputs and minimizes
the use of unobservable inputs when measuring fair value. The Company uses three levels of inputs that may be used to measure fair
value:
Level 1 – quoted
prices in active markets for identical assets or liabilities
Level 2 – quoted
prices for similar assets and liabilities in active markets; quoted prices for identical or similar instruments in markets that
are not active; and model-derived valuations in which all significant inputs and significant value drivers are observable in active
markets.
Level 3 – inputs
that are unobservable (for example cash flow modeling inputs based on assumptions)
Financial liabilities measured at fair value on a recurring basis
are summarized below:
|
|
|
Fair Value Measurements at March 31, 2016 Using
|
|
|
|
|
Total
March 31,
2016
|
|
|
Quoted prices in active markets for identical assets
(Level 1)
|
|
|
Significant other observable inputs
(Level 2)
|
|
|
Significant other unobservable inputs
(Level 3)
|
|
|
Liabilities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Derivative liabilities – warrants
|
|
$
|
–
|
|
|
|
–
|
|
|
|
–
|
|
|
$
|
–
|
|
|
|
|
Fair Value Measurements at March 31, 2015 Using
|
|
|
|
|
Total
March 31,
2015
|
|
|
Quoted prices in active markets for identical assets
(Level 1)
|
|
|
Significant other observable inputs
(Level 2)
|
|
|
Significant other unobservable inputs
(Level 3)
|
|
|
Liabilities:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Derivative liabilities – warrants
|
|
$
|
11,000
|
|
|
|
–
|
|
|
|
–
|
|
|
$
|
11,000
|
|
Level 3 liabilities are valued using unobservable
inputs to the valuation methodology that are significant to the measurement of the fair value of the liabilities. For fair value
measurements categorized within Level 3 of the fair value hierarchy, the Company’s accounting and finance department, who
report to the Chief Financial Officer, determine its valuation policies and procedures. The development and determination of the
unobservable inputs for Level 3 fair value measurements and fair value calculations are the responsibility of the Company’s
accounting and finance department and are approved by the Chief Financial Officer.
Level 3 Valuation Techniques
:
Level 3 financial liabilities consist of the
derivative liabilities for which there is no current market for these securities such that the determination of fair value requires
significant judgment or estimation. Changes in fair value measurements categorized within Level 3 of the fair value hierarchy are
analyzed each period based on changes in estimates or assumptions and recorded as appropriate.
The Company uses the Black-Scholes option valuation
model to value Level 3 derivatives at inception and on subsequent valuation dates. This model incorporates transaction details
such as the Company’s stock price, contractual terms, maturity, risk free rates, as well as volatility. A significant decrease
in the volatility or a significant decrease in the Company’s stock price, in isolation, would result in a significantly lower
fair value measurement. Changes in the values of the derivative liabilities are recorded in “Gain (loss) due to change in
fair value of derivative liabilities” in the Company’s consolidated statements of comprehensive (loss) income.
As of March 31, 2016 and 2015, there were no
transfers in or out of Level 3 from other levels in the fair value hierarchy.
Property and Equipment
Property and equipment are stated at cost less
accumulated depreciation and amortization. Depreciation of property and equipment is computed using the straight-line method over
the estimated useful lives of the respective assets. Depreciation of leasehold improvements is computed using the straight-line
method over the lesser of the estimated useful life of the improvement or the remaining term of the lease. Estimated useful asset
life by classification is as follows:
|
|
|
Years
|
|
|
Office equipment
|
|
3
|
|
|
Manufacturing, lab and other equipment
|
|
5
|
|
|
Furniture and fixtures
|
|
7
|
|
Upon retirement or sale, the cost and related
accumulated depreciation are removed from the consolidated balance sheet and the resulting gain or loss is reflected in operations.
Maintenance and repairs are charged to operations as incurred.
Impairment of Long-Lived Assets
The Company periodically reviews the carrying
values of its long-lived assets when events or changes in circumstances would indicate that it is more likely than not that their
carrying values may exceed their realizable values, and records impairment charges when considered necessary. Specific potential
indicators of impairment include, but are not necessarily limited to:
|
|
·
|
a significant decrease in the fair value
of an asset;
|
|
|
·
|
a significant change in the extent or
manner in which an asset is used or a significant physical change in an asset;
|
|
|
·
|
a significant adverse change in legal
factors or in the business climate that affects the value of an asset;
|
|
|
·
|
an adverse action or assessment by the
U.S. Food and Drug Administration or another regulator; and
|
|
|
·
|
an accumulation of costs
significantly in excess of the amount originally expected to acquire or construct an asset; and operating or cash flow losses combined
with a history of operating or cash flow losses or a projection or forecast that demonstrates continuing losses associated with
an income-producing asset.
|
When circumstances indicate that an impairment
may have occurred, the Company tests such assets for recoverability by comparing the estimated undiscounted future cash flows expected
to result from the use of such assets and their eventual disposition to their carrying amounts. In estimating these future cash
flows, assets and liabilities are grouped at the lowest level for which there are identifiable cash flows that are largely independent
of the cash flows generated by other such groups. If the undiscounted future cash flows are less than the carrying amount of the
asset, an impairment loss, measured as the excess of the carrying value of the asset over its estimated fair value, will be recognized.
The cash flow estimates used in such calculations are based on estimates and assumptions, using all available information that
management believes is reasonable.
In connection with an agreement with certain
investors to sell its interest in Ruthigen at a fixed price of $2.75 per share, the Company determined that the carrying value
of the shares held in Ruthigen was impaired. As a result, during the year ended March 31, 2015, the Company recorded an impairment
loss in the amount of $4,650,000 which represents the difference between the cost and aggregate purchase price of $2.75 per share
the Company agreed to sell its interest in Ruthigen. At March 31, 2015, the Company’s interest in Ruthigen was reported as
a long-term asset on its consolidated financial statements rather than a consolidated subsidiary given that the Company no longer
controlled Ruthigen, the Company and had very little means to control the value of the asset.
During the year ended March 31, 2016, the Company
had noted no indicators of impairment.
Research
and Development
Research and development expense is charged
to operations as incurred and consists primarily of personnel expenses, clinical and regulatory services and supplies. For the
years ended March 31, 2016 and 2015, research and development expense amounted to $1,806,000 and $1,533,000, respectively.
Advertising Costs
Advertising costs are charged to operations
as incurred. Advertising costs amounted to $175,000 and $231,000, for the years ended March 31, 2016 and 2015, respectively. Advertising
costs are included in selling, general and administrative expenses in the accompanying consolidated statements of comprehensive
loss.
Shipping and Handling Costs
The Company classifies amounts billed to customers
related to shipping and handling in sale transactions as product revenues. Shipping and handling costs incurred are recorded in
cost of product revenues. For the years ended March 31, 2016 and 2015, the Company recorded revenue related to shipping and handling
costs of $59,000 and $114,000, respectively.
Foreign Currency Reporting
The Company’s subsidiary, OTM, uses the
local currency (Mexican Pesos) as its functional currency and its subsidiary, OIS Europe, uses the local currency (Euro) as its
functional currency. Assets and liabilities are translated at exchange rates in effect at the balance sheet date, and revenue and
expense accounts are translated at average exchange rates during the period. Resulting translation adjustments amounted to $347,000
and $438,000 for the years ended March 31, 2016 and 2015, respectively, and were recorded in other comprehensive loss in the accompanying
consolidated statements of comprehensive loss.
Foreign currency transaction gains (losses)
relate primarily to trade payables and receivables between subsidiaries OTM and OIS Europe. These transactions are expected to
be settled in the foreseeable future. The Company recorded foreign currency transaction gains of $38,000 and losses of $24,000
for the years ended March 31, 2016 and 2015, respectively. The related gains and losses were recorded in other expense, net, in
the accompanying consolidated statements of comprehensive loss.
Stock-Based Compensation
The Company accounts for share-based awards
exchanged for employee services at the estimated grant date fair value of the award. The Company estimates the fair value of employee
stock option awards using the Black-Scholes option pricing model. The Company amortizes the fair value of employee stock options
on a straight-line basis over the requisite service period of the awards. Compensation expense includes the impact of an
estimate for forfeitures for all stock options.
The Company accounts for equity instruments
issued to non-employees at their fair value on the measurement date. The measurement of stock-based compensation is subject to
periodic adjustment as the underlying equity instrument vests or becomes non-forfeitable. Non-employee stock-based compensation
charges are amortized over the vesting period or as earned.
Income Taxes
Deferred tax assets and liabilities are determined
based on the differences between the financial reporting and tax bases of assets and liabilities and net operating loss and credit
carryforwards using enacted tax rates in effect for the year in which the differences are expected to impact taxable income. Valuation
allowances are established when necessary to reduce deferred tax assets to the amounts expected to be realized.
Tax benefits claimed or expected to be claimed
on a tax return are recorded in the Company’s consolidated financial statements. A tax benefit from an uncertain tax position
is only recognized if it is more likely than not that the tax position will be sustained on examination by the taxing authorities,
based on the technical merits of the position. The tax benefits recognized in the consolidated financial statements from such a
position are measured based on the largest benefit that has a greater than fifty percent likelihood of being realized upon ultimate
resolution. Uncertain tax positions have had no impact on the Company’s consolidated financial condition, results of comprehensive
loss or cash flows.
Comprehensive Loss
Other comprehensive loss includes all changes
in stockholders’ equity during a period from non-owner sources and is reported in the consolidated statement of changes in
stockholders’ equity. To date, other comprehensive loss consists of changes in accumulated foreign currency translation adjustments.
Accumulated other comprehensive losses at March 31, 2016 and 2015 were $3,854,000 and $3,507,000, respectively.
Net Loss per Share
The Company computes basic net loss per share
by dividing net loss per share available to common stockholders by the weighted average number of common shares outstanding for
the period and excludes the effects of any potentially dilutive securities. Diluted earnings per share, if presented, would include
the dilution that would occur upon the exercise or conversion of all potentially dilutive securities into common stock using the
“treasury stock” and/or “if converted” methods as applicable. The computation of basic loss per share for
the years ended March 31, 2016 and 2015 excludes the potentially dilutive securities summarized in the table below because their
inclusion would be anti-dilutive.
|
|
|
March 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
Options to purchase common stock
|
|
|
3,769,000
|
|
|
|
2,877,000
|
|
|
Warrants to purchase common stock
|
|
|
7,424,000
|
|
|
|
7,741,000
|
|
|
|
|
|
11,193,000
|
|
|
|
10,618,000
|
|
Common Stock Purchase Warrants and Other
Derivative Financial Instruments
The Company classifies common stock purchase
warrants and other free standing derivative financial instruments as equity if the contracts (i) require physical settlement or
net-share settlement or (ii) give the Company a choice of net-cash settlement or settlement in its own shares (physical settlement
or net-share settlement). The Company classifies any contracts that (i) require net-cash settlement (including a requirement to
net cash settle the contract if an event occurs and if that event is outside the control of the Company), (ii) give the counterparty
a choice of net cash settlement or settlement in shares (physical settlement or net-share settlement), or (iii) contain reset provisions
as either an asset or a liability. The Company assesses classification of its freestanding derivatives at each reporting date to
determine whether a change in classification between assets and liabilities is required. The Company determined that its freestanding
derivatives, which principally consist of warrants to purchase common stock, satisfied the criteria for classification as equity
instruments, other than certain warrants that contained reset provisions and certain warrants that required net-cash settlement
that the Company classified as derivative liabilities as more fully described in Note 11.
Preferred Stock
The Company applies the accounting standards
for distinguishing liabilities from equity when determining the classification and measurement of its preferred stock. Shares that
are subject to mandatory redemption (if any) are classified as liability instruments and are measured at fair value. The Company
classifies conditionally redeemable preferred shares, which includes preferred shares that feature redemption rights that are either
within the control of the holder or subject to redemption upon the occurrence of uncertain events not solely within the Company’s
control, as temporary equity. At all other times, preferred shares are classified as stockholders' equity.
Convertible Instruments
The Company evaluates and bifurcates conversion
options from their host instruments and accounts for them as free standing derivative financial instruments according to certain
criteria. The criteria include circumstances in which (a) the economic characteristics and risks of the embedded derivative instrument
are not clearly and closely related to the economic characteristics and risks of the host contract, (b) the hybrid instrument that
embodies both the embedded derivative instrument and the host contract is not re-measured at fair value under otherwise applicable
generally accepted accounting principles with changes in fair value reported in earnings as they occur and (c) a separate instrument
with the same terms as the embedded derivative instrument would be considered a derivative instrument. An exception to this rule
is when the host instrument is deemed to be conventional as that term is described under applicable Generally Accepted Accounting
Principles (“GAAP”).
Subsequent Events
Management has evaluated subsequent events
or transactions occurring through the date these consolidated financial statements were issued.
Recent Accounting Pronouncements
In February 2016, the FASB issued ASU 2016-02,
Leases
("ASU 2016-02"). The standard amends the existing accounting standards for lease accounting, including
requiring lessees to recognize most leases on their balance sheets and making targeted changes to lessor accounting. ASU 2016-02
will be effective beginning in the first quarter of 2019. Early adoption of ASU 2016-02 is permitted. The new leases standard requires
a modified retrospective transition approach for all leases existing at, or entered into after, the date of initial application,
with an option to use certain transition relief. The Company is currently evaluating the impact of adopting ASU 2016-02 on our
consolidated financial statements.
In March 2016, the FASB issued ASU No. 2016-08,
Revenue from Contracts with Customers (Topic 606): Principal versus Agent Considerations (Reporting Revenue Gross versus Net).
This ASU amends the principal versus agent guidance in ASU No. 2014-09,
Revenue from Contracts with Customers (Topic 606)
which was issued in May 2014 (“ASU 2014-09”). Further, in April 2016, the FASB issued ASU No. 2016-10,
Revenue from
Contracts with Customers (Topic 606): Identifying Performance Obligations and Licensing
. This ASU also amends ASU 2014-09 and
is related to the identification of performance obligations and accounting for licenses. The effective date and transition requirements
for both of these amendments to ASU 2014-09 are the same as those of ASU 2014-09, which was deferred for one year by ASU No. 2015-14,
Revenue from Contracts with Customers (Topic 606): Deferral of the Effective Date
. That is, the guidance under these standards
is to be applied using a full retrospective method or a modified retrospective method, as outlined in the guidance, and is effective
for annual periods, and interim periods within those annual periods, beginning after December 15, 2017. Early adoption is permitted
only for annual periods, and interim period within those annual periods, beginning after December 15, 2016. The Company is currently
evaluating the provisions of each of these standards and assessing their impact on the Company’s consolidated financial statements
and disclosures.
In March 2016, the FASB issued ASU No. 2016-09,
Compensation-Stock Compensation (Topic 718): Improvements to Employee Share-Based Payment Accounting.
This ASU makes targeted
amendments to the accounting for employee share-based payments. This guidance is to be applied using various
transition
methods such as full retrospective, modified retrospective, and prospective based on the criteria for the specific amendments as
outlined in the
guidance. The guidance is effective for annual periods, and interim periods within those annual periods,
beginning after December 15, 2016. Early adoption
is permitted, as long as all of the amendments are adopted in the same
period. The Company is currently evaluating the provisions of this guidance and
assessing its impact on the Company’s
consolidated financial statements and disclosures.
Accounting standards that have been issued
or proposed by the FASB, SEC and/or other standards-setting bodies that do not require adoption until a future date are not expected
to have a material impact on the condensed consolidated financial statements upon adoption.
NOTE 4 – Accounts Receivable
Accounts receivable consists of the following:
|
|
|
March 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
Accounts receivable
|
|
$
|
2,942,000
|
|
|
$
|
1,720,000
|
|
|
Less: allowance for doubtful accounts
|
|
|
(15,000
|
)
|
|
|
(20,000
|
)
|
|
Less: discounts, rebates, distributor fees and returns
|
|
|
(653,000
|
)
|
|
|
(183,000
|
)
|
|
|
|
$
|
2,274,000
|
|
|
$
|
1,517,000
|
|
NOTE 5 – Inventories
Inventories consist of the following:
|
|
|
March 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
Raw materials
|
|
$
|
1,104,000
|
|
|
$
|
865,000
|
|
|
Finished goods
|
|
|
536,000
|
|
|
|
537,000
|
|
|
|
|
$
|
1,640,000
|
|
|
$
|
1,402,000
|
|
NOTE 6 – Prepaid Expenses
and Other Current Assets
Prepaid expenses and other current assets consist
of the following:
|
|
|
March 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
Prepaid insurance
|
|
$
|
405,000
|
|
|
$
|
367,000
|
|
|
Prepaid rebates
|
|
|
378,000
|
|
|
|
14,000
|
|
|
Other prepaid expenses and other current assets
|
|
|
722,000
|
|
|
|
211,000
|
|
|
|
|
$
|
1,505,000
|
|
|
$
|
592,000
|
|
NOTE 7 – Property and Equipment
Property and equipment consists of the following:
|
|
|
March 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
Manufacturing, lab, and other equipment
|
|
$
|
3,075,000
|
|
|
$
|
2,937,000
|
|
|
Office equipment
|
|
|
298,000
|
|
|
|
278,000
|
|
|
Furniture and fixtures
|
|
|
83,000
|
|
|
|
82,000
|
|
|
Leasehold improvements
|
|
|
307,000
|
|
|
|
249,000
|
|
|
|
|
|
3,763,000
|
|
|
|
3,546,000
|
|
|
Less: accumulated depreciation and amortization
|
|
|
(2,913,000
|
)
|
|
|
(2,751,000
|
)
|
|
|
|
$
|
850,000
|
|
|
$
|
795,000
|
|
Depreciation and amortization expense amounted
to $244,000 and $253,000 for the years ended March 31, 2016 and 2015, respectively.
During the year ended March 31, 2015, the
Company realized a gain of $13,000 on the disposal of property and equipment. This amount was recorded
within operating expenses in the accompanying consolidated statements of comprehensive loss.
NOTE 8 – Investment in Ruthigen, Inc.
The Company’s long-term investments consisted
of the Company’s ownership of 1,650,000 shares of Ruthigen common stock at March 31, 2015. During the year ended March 31,
2016, the Company sold its remaining 1,650,000 shares of Ruthigen common stock for proceeds of $4,537,500 pursuant to a securities
purchase agreement with two investors. Additionally, in the year ended March 31, 2016, the Company paid a $165,000 banker fee related
to the sale transaction.
On January 8, 2015, the Company entered into a securities
purchase agreement pursuant to which the Company agreed to sell its 2,000,000 shares in Ruthigen to two accredited investors for
an aggregate purchase price of $5,000,000 upon the occurrence of a trigger event during a standstill period of 60 calendar days.
The securities purchase agreement lapsed according to its terms, however, as a result of the Company entering into the agreement,
the Company determined that the carrying value of the shares held in Ruthigen were impaired. As a result, the Company recorded
an impairment loss in the amount of $4,650,000 which represents the difference between cost and aggregate purchase price of $2.75
per share the Company agreed to sell its interest in Ruthigen.
NOTE 9 – Accrued Expenses
and Other Current Liabilities
Accrued expenses and other current liabilities
consist of the following:
|
|
|
March 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
Salaries and related costs
|
|
$
|
693,000
|
|
|
$
|
545,000
|
|
|
Professional fees
|
|
|
557,000
|
|
|
|
137,000
|
|
|
Other
|
|
|
276,000
|
|
|
|
100,000
|
|
|
|
|
$
|
1,526,000
|
|
|
$
|
782,000
|
|
NOTE 10 – Long-Term Debt
Financing of Insurance Premiums
On January 25, 2015, the Company entered into
a note agreement for $116,000 with an interest rate of 5.50% per annum. This instrument was issued in connection with financing
insurance premiums. The note was payable in monthly installments of $17,000 with the final payment on August 25, 2015. During the
year ended March 31, 2015, the Company made principal and interest payments of $33,000 and $1,000, respectively. During the year
ended March 31, 2016, the Company made principal and interest payments of $83,000 and $1,000, respectively. The note was paid in
full during the year ended March 31, 2016.
On January 25, 2016, the Company entered into
a note agreement for $146,000 with an interest rate of 6.25% per annum. This instrument was issued in connection with financing
insurance premiums. The note is payable in monthly installments of $17,000 with the final payment on August 25, 2016. During the
year ended March 31, 2016, the Company made principal and interest payments of $32,000 and $1,000, respectively. The remaining
balance of this note amounted to $114,000 at March 31, 2016 which is included in the current portion of long-term debt in the accompanying
consolidated balance sheet.
NOTE 11 – Derivative Liability
Warrants Issued in Conjunction with the
Company’s December 9, 2013 and February 26, 2014 Registered Direct Offerings
The Company deems financial instruments which
require net-cash settlement as either an asset or a liability. The common stock purchase warrants issued in conjunction with the
Company’s December 9, 2013 and February 26, 2014 registered direct offerings contain a net-cash settlement feature which
give the warrant holder the right to net-cash settlement in the event certain transactions occur. Pursuant to the terms of the
warrants, if such a transaction occurs the warrant holder will be entitled to a net-cash settlement value calculated using the
Black-Scholes valuation model using an expected volatility equal to the greater of 100% and the 30 day volatility obtained from
the HVT function on Bloomberg, an expected term equal to the remaining term of the warrants, and applicable risk-free interest
rate corresponding to the U.S. Treasury.
The derivative liabilities relating to the
warrants with net-cash settlement provisions were valued using the Black-Scholes option valuation model and the following assumptions
on the following dates:
|
|
|
Measurement
Date
|
|
Warrants
|
|
|
Remaining
Contract
Term in
Years
|
|
|
Exercise
Price
|
|
|
Volatility
|
|
|
Risk-free
Interest
Rate
|
|
|
Fair
Value
|
|
|
Warrant
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Placement Agent Warrants
|
|
March 31, 2015
|
|
|
16,500
|
|
|
|
1.09
|
|
|
|
5.00
|
|
|
|
100%
|
|
|
|
0.26%
|
|
|
$
|
1,000
|
|
|
Investor - Series A Warrants
|
|
March 31, 2015
|
|
|
1,000
|
|
|
|
0.41
|
|
|
|
3.00
|
|
|
|
100%
|
|
|
|
0.14%
|
|
|
|
–
|
|
|
Investor - Series B Warrants
|
|
March 31, 2015
|
|
|
1,400,000
|
|
|
|
0.41
|
|
|
|
3.63
|
|
|
|
100%
|
|
|
|
0.14%
|
|
|
|
5,000
|
|
|
Placement Agent Warrants
|
|
March 31, 2015
|
|
|
69,037
|
|
|
|
1.09
|
|
|
|
3.00
|
|
|
|
100%
|
|
|
|
0.26%
|
|
|
|
5,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
11,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Warrant
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Placement Agent Warrants
|
|
March 31, 2016
|
|
|
16,500
|
|
|
|
0.09
|
|
|
|
5.00
|
|
|
|
100%
|
|
|
|
0.21%
|
|
|
$
|
–
|
|
|
Placement Agent Warrants
|
|
March 31, 2016
|
|
|
69,037
|
|
|
|
0.09
|
|
|
|
3.00
|
|
|
|
100%
|
|
|
|
0.21%
|
|
|
|
–
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
–
|
|
The following table sets forth a summary of
the changes in the fair value of our Level 3 financial liabilities that are measured at fair value on a recurring basis:
|
|
|
Year Ended March 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
Beginning balance
|
|
$
|
11,000
|
|
|
$
|
3,175,000
|
|
|
Fair value of warrants issued
|
|
|
–
|
|
|
|
–
|
|
|
Mark to market net unrealized gain
|
|
|
(11,000
|
)
|
|
|
(3,164,000
|
)
|
|
Reclassification to additional paid in capital
|
|
|
–
|
|
|
|
–
|
|
|
Ending balance
|
|
$
|
–
|
|
|
$
|
11,000
|
|
NOTE 12 – Commitments and
Contingencies
Lease Commitments
On June 15, 2013, the Company leased
office space in Mexico with an address of: Av De Las Americas, 1592 Piso 7, en la Colonia Country Club en Guadalajara
Jalisco, CP 44637 for 23,400 Mexican Pesos (approximately $1,800 USD) per month. One month’s rent was required as a
deposit. The lease was terminated in June 2015.
The Company also shares certain office and
laboratory space, as well as certain laboratory equipment, in a building located at 454 North 34th Street, Seattle, Washington.
The space is rented for $2,700 per month and requires a ninety day notice for cancellation.
The Company currently rents approximately 800
square feet of sales office space in Herten, the Netherlands. The office space is rented on a month to month basis at $1,700
per month and requires a sixty day notice for cancellation.
On October 10, 2012, the Company entered into
Amendment No. 7 to its property lease agreement, extending the lease on its Petaluma, California facility to September 30, 2017.
Pursuant to the amendment, in exchange for certain improvements on the building, the Company agreed to increase the lease payment
from $10,380 to $11,072 per month.
Minimum lease payments for non-cancelable operating
leases are as follows:
|
For Years Ending March 31,
|
|
|
|
|
2017
|
|
$
|
370,000
|
|
|
2018
|
|
|
222,000
|
|
|
2019
|
|
|
91,000
|
|
|
2020
|
|
|
8,000
|
|
|
Total minimum lease payments
|
|
$
|
691,000
|
|
Rental expense amounted to $442,000 and $446,000
for the years ended March 31, 2016 and 2015, respectively and is recorded in the accompanying consolidated statement of comprehensive
loss.
Legal Matters
The Company, on occasion, may be involved
in legal matters arising in the ordinary course of business including matters involving proprietary technology. While management
believes that such matters are currently insignificant, matters arising in the ordinary course of business for which the Company
is or could become involved in litigation may have a material adverse effect on its business, financial condition of comprehensive
loss.
Employment Agreements
As of March 31, 2016, the Company had employment
agreements in place with four of its key executives. The agreements provide, among other things, for the payment of nine to twenty-four
months of severance compensation for terminations under certain circumstances. With respect to these agreements, at March 31, 2016,
potential severance amounted to $1,097,000 and aggregated annual salaries amounted to $944,000.
Commercial Agreements
On August 9, 2012, the Company, along with
its Mexican subsidiary and manufacturer Oculus Technologies of Mexico S.A. de C.V. (“Manufacturer”), entered into a
license, exclusive distribution and supply agreement with More Pharma Corporation, S. de R.L. de C.V. (“More Pharma”)
(the “License Agreement”). For a one-time payment of $500,000, the Company granted More Pharma an exclusive license,
with the right to sublicense, under certain conditions and with the Company’s consent, to all of the Company’s proprietary
rights related to certain of its pharmaceutical products for human application that utilize the Company’s Microcyn® Technology
within Mexico. For an additional one-time payment of $3,000,000, the Company also agreed to appoint More Pharma as the exclusive
distributor of certain of its products in Mexico for the term of the agreement. Additionally, Manufacturer granted More Pharma
an exclusive license to certain of Manufacturer’s then-held trademarks in exchange for a payment of $100,000 to Manufacturer.
The Company has the ability to terminate the agreement if certain annual purchase minimums are not met. The term of the agreement
is twenty-five years from the effective date of August 15, 2012. The term of the License Agreement will automatically renew after
the twenty-five year term for successive two year terms as long as More Pharma has materially complied with any and all of the
obligations under the License Agreement, including but not limited to, meeting the minimum purchase requirements set forth therein.
On January 6, 2015, the Company was notified that More Pharma had been acquired by Laboratorios Sanfer S.A. de C.V (“Sanfer”).
Additionally, on August 9, 2012, the Company,
along with Manufacturer, entered into an exclusive distribution and supply agreement with More Pharma (the “Distribution
Agreement”). For a one-time payment of $1,500,000, the Company granted More Pharma the exclusive ability to market and sell
certain of its pharmaceutical products for human application that utilize the Company’s Microcyn® Technology. The Company
also appointed More Pharma as its exclusive distributor, with the right to execute sub-distribution agreements, under certain conditions,
and with the Company’s consent, within a number of Central and South American countries.
The Company will recognize the $5,100,000 related
to the License Agreement and the Distribution Agreement as revenue on the straight line basis consistent with the Company’s
historical experience with contracts having similar terms, which is typically over three to five years of the contract. During
years ended March 31, 2016 and 2015, the Company recognized $751,000 and $1,499,000, respectively, related to the amortization
of the upfront fees received in the transaction. At March 31, 2016, the Company had outstanding accounts receivable of $912,000
due from Laboratorios Sanfer. At March 31, 2015, the Company had outstanding accounts receivable of $843,000 due from Laboratorios
Sanfer.
NOTE 13 – Stockholders’
Equity
Authorized Capital
On June 29, 2015, the stockholders of the Company
approved a reverse stock split of the Company’s outstanding common stock and to proportionally decrease the total number
of shares that the Company is authorized to issue at a whole number ratio in the range of 1-for-5 to 1-for-9, such ratio to be
determined in the discretion of the Company’s Board of Directors, and authorized the Company’s Board of Directors to
effect the reverse stock split, if in their judgment it is necessary, at any time until June 29, 2016, upon which date the resolution
lapses. To date, no reverse stock split has been authorized by the Board of Directors.
Effective October 22, 2015, the Company increased
its authorized share capital from 30,000,000 shares of common stock to 60,000,000 shares of common stock, $0.0001 par value per
share by filing a certificate of amendment with the Secretary of State of Delaware. The share increase was approved by the Company’s
stockholders at the 2015 Annual Meeting of Stockholders on October 9, 2015. Additionally, the Company is authorized to issue
up to 714,286 shares of convertible preferred stock with a par value of $0.0001 per share.
Description of Common Stock
Each share of common stock has the right to
one vote. The holders of common stock are entitled to dividends when funds are legally available and when declared by the board
of directors.
April 2014 At-the-Market Offering
On April 2, 2014, the Company entered into
an At-the-Market Issuance Sales Agreement with MLV & Co. LLC under which the Company can issue and sell shares of its common
stock having an aggregate offering price of up to $9,159,000 from time to time through MLV acting as its sales agent. To date,
the Company has raised an aggregate $4,706,000 in connection with this agreement. The Company will pay MLV a commission rate equal
to 3.0% of the gross proceeds from the sale of any shares of common stock sold through MLV as agent under the Sales Agreement.
For the year ended March 31, 2016 the Company sold 2,254,596 shares for gross proceeds of $3,263,000 and net proceeds of $3,150,000
after deducting commissions and other offering expenses. For the year ended March 31, 2015, the Company sold 467,934 shares for
gross proceeds of $1,443,000 and net proceeds of $1,341,000 after deducting commissions and other offering expenses.
March 2016 Underwritten Public Offering
On March 18, 2016, the Company entered into
an underwriting agreement with Dawson James Securities, Inc. with respect to the issuance and sale of an aggregate of 3,400,000
units, each unit consisting of one share of common stock, par value $0.0001 per share, together with one quarter (0.25) of one
warrant to purchase one share of common stock at an exercise price equal to $1.00 per share, in an underwritten public offering.
The public offering price for each unit, consisting of one share of common stock together with one quarter (0.25) of one warrant,
was $1.00. Because the Company is prohibited from issuing fractional shares, the warrants can only be exercised in lots of four,
which means that each holder must exercise four March 2016 Warrants to receive one share of common stock, or a total of 850,000
shares. The warrants have an initial exercise price of $1.00 per share and have a term of three years. Pursuant to the underwriting
agreement, the Company paid Dawson James Securities, Inc. a cash fee equal to 8% of the aggregate gross proceeds raised in this
offering and also paid $50,000 in legal fees and expenses of the underwriter’s legal counsel. The gross proceeds from the
sale of the shares of common stock and the warrants was $3,400,000, and net proceeds of 2,994,000 after deducting underwriting
commissions and other estimated offering expenses.
January 2015 Underwritten Public Offering
On January 20, 2015, the Company entered into
an underwriting agreement with Maxim Group LLC with respect to the issuance and sale of an aggregate of 6,250,000 shares of common
stock, par value $0.0001 per share, together with warrants to purchase an aggregate of 4,687,500 shares of its common stock at
an exercise price equal to $1.30 per share in an underwritten public offering. The public offering price for each share of common
stock together with 0.75 of a warrant was $1.00. Pursuant to the underwriting agreement, the Company also granted Maxim Group LLC
a 45-day option to purchase an additional 937,500 shares of common stock and/or 703,125 warrants to purchase an additional 703,125
shares of common stock to cover any over-allotments made by the underwriters in the sale and distribution of the shares and warrants.
On January 21, 2015, Maxim Group LLC exercised the over-allotment option with respect to 703,125 warrants. The offering, including
the partial exercise of the over-allotment option, closed on January 26, 2015. On March 3, 2015, Maxim Group LLC exercised the
over-allotment option with respect to 134,500 shares of common stock, which closed on March 6, 2015. The registration statement
for the sale of the shares of common stock and warrants sold in the public offering became effective January 20, 2015, file number
333-200461. The gross proceeds from the sale of the shares of common stock and the warrants, including the partial exercise of
the over-allotment option was $6,392,000, and net proceeds of $5,444,000 after deducting underwriting discounts and commissions
and other offering expenses.
Common Stock Issued to Settle Fees for Services Provided
On April 24, 2009, the Company entered into
an agreement with Advocos LLC, a contract sales organization that serves as part of the Company’s sales force, for the sale
of the Company’s tissue care products in the United States. Pursuant to the agreement, the Company agreed to pay the contract
sales organization a monthly fee and potential bonuses that will be based on achievement of certain levels of sales. The Company
agreed to issue the contract sales organization cash or shares of common stock to settle fees for its services. The Company has
determined that the fair value of the common stock was more readily determinable than the fair value of the services rendered.
During the year ended March 31, 2016, the Company issued 208,519 shares of common stock related to this agreement. The fair market
value of the common stock was $286,000 at issuance. During the year ended March 31, 2016, the Company recorded $190,000 of expense
related to stock issued pursuant to this agreement and settled $96,000 of fees accrued in prior periods. During the year ended
March 31, 2015 the Company issued 32,501 shares of common stock in connection with this agreement. During the year ended March
31, 2015 the Company recorded $76,000 expense related to stock issued in connection with this agreement. The expense was recorded
as selling, general and administrative expense in the accompanying consolidated statements of comprehensive loss.
Common Stock Purchase Warrants
On March 31, 2016,
the Company issued Dawson James Securities, Inc. a warrant to purchase 250,000 shares of the Company’s common stock
at an exercise price of $1.00 per share in connection with a service agreement. The warrants were non-forfeitable at date of issuance.
The warrants were valued using the Black-Scholes option pricing model. Assumptions used were as follows: Fair value of the underlying
stock $0.95; risk-free interest rate 0.01%; contractual life of 3 years; dividend yield of 0%; and volatility of 87%. The
fair value of the warrants amounted to $128,000 and was recorded as selling, general and administrative expense in the accompanying
consolidated statement of comprehensive loss for the year ended March 31, 2016.
NOTE 14 – Stock-Based Compensation
2006 Stock Plan
The board initially adopted the 2006 Stock
Incentive Plan on August 25, 2006. On December 14, 2006, the stockholders approved the 2006 Stock Incentive Plan which became
effective at the close of the Company’s initial public offering. The 2006 Stock Incentive Plan was later amended and restated
by a unanimous board resolution on April 26, 2007, and such amendments were subsequently approved by the stockholders. On
September 10, 2009, the Company’s shareholders approved a subsequent amendment to the 2006 Stock Incentive Plan. The 2006
Stock Incentive Plan, as amended and restated, is hereafter referred to as the “2006 Plan.”
The 2006 Plan provides for the granting of
incentive stock options to employees and the granting of nonstatutory stock options to employees, non-employee directors, advisors
and consultants. The 2006 Plan also provides for grants of restricted stock, stock appreciation rights and stock unit awards to
employees, non-employee directors, advisors and consultants.
In accordance with the 2006 Plan the stated
exercise price may not be less than 100% and 85% of the estimated fair market value of common stock on the date of grant for ISOs
and NSOs, respectively, as determined by the board of directors at the date of grant. With respect to any 10% stockholder, the
exercise price of an ISO or NSO shall not be less than 110% of the estimated fair market value per share on the date of grant.
Options issued under the 2006 Plan generally
have a ten-year term.
Shares subject to awards that expire unexercised
or are forfeited or terminated will again become available for issuance under the 2006 Plan. No participant in the 2006 Plan can
receive option grants, restricted shares, stock appreciation rights or stock units for more than 26,786 shares in the aggregate
in any calendar year.
On November 7, 2006, the board initially authorized
a total of 178,571 of the Company’s common stock shares (adjusted for the reverse stock split effective April 1, 2013) for
issuance under the 2006 Plan in addition to increases provided for in the 2006 Plan through August 25, 2016. On September 10,
2009, the Company’s shareholders approved an amendment of the 2006 Plan which authorized and reserved an additional 142,858
shares (adjusted for the reverse stock split effective April 1, 2013) for issuance under the 2006 Plan. The number of shares of
the Company’s common stock reserved for issuance under the 2006 Plan may increase if such increase is approved by the board,
with no further action by the stockholders, at the beginning of each fiscal year by an amount equal to the lesser of (i) 250,000
shares (adjusted for the reverse stock split effective April 1, 2013); (ii) 5% of the outstanding shares of common stock of the
Company on the last day of the immediately preceding year, or (iii) an amount determined by the Company’s board of the directors.
As provided under the 2006 Plan, the aggregate
number of shares authorized for issuance as awards under the 2006 Plan increased on April 1, 2012, by 207,199 shares
(which number constitutes 5% of the outstanding shares on the last day of the year ended March 31, 2012). During
the year ended March 31, 2015, the board of directors approved an increase of 250,000 shares authorized for issuance. During the
year ended March 31, 2016, the board of directors approved an increase of 250,000 shares authorized for issuance.
2011 Stock Plan
On September 12, 2011, upon recommendation
of the board, the stock holders approved the Company’s 2011 Stock Incentive Plan (the “2011 Plan”). The 2011
Plan is effective as of June 21, 2012.
The 2011 Plan provides for the grant of incentive
stock options as defined in Section 422 of the Internal Revenue Code to employees, and the grant of non-statutory stock options
and stock purchase rights to employees, non-employee directors, advisors and consultants. The 2011 Plan also permits the grant
of stock appreciation rights, stock units and restricted stock.
The board has authorized 428,572 of the Company’s
common stock for issuance under the 2011 Plan, in addition to automatic increases provided for in the 2011 Plan through April 1,
2021. The number of shares of the Company’s common stock reserved for issuance under the 2011 Plan will automatically increase,
with no further action by the stockholders, at the beginning of each fiscal year by an amount equal to the lesser of (i) 15% of
the outstanding shares of the Company’s common stock on the last day of the immediately preceding year, or (ii) an amount
approved by the Company’s board of directors.
Options issued under the 2011 Plan will generally
have a ten-year term.
In accordance with the 2011 Plan, the stated
exercise price of an employee incentive stock option shall not be less than 100% of the estimated fair market value of a share
of common stock on the date of grant, and the stated exercise price of an nonstatutory option shall not be less 85% of the estimated
fair market value of a share of common stock on the date of grant, as determined by the board of directors. An employee who owns
more than 10% of the total combined voting power of all classes of outstanding stock of the Company shall not be eligible for the
grant of an employee incentive stock option unless such grant satisfies the requirements of Section 422(c)(5) of the Internal Revenue
Code.
Shares subject to awards that expire unexercised
or are forfeited or terminated for any other reason will again become available for issuance under the 2011 Plan. No participant
in the 2011 Plan can receive option grants, stock appreciation rights, restricted shares, or stock units for more than 107,143
shares in the aggregate in any calendar year. As provided under the 2011 Plan, the aggregate number of shares authorized for issuance
as awards under the 2011 Plan automatically increases on April 1 of each year by in an amount equal to the lesser of (i) 15% of
the outstanding shares on the last day of the immediately preceding year, or (ii) an amount determined by the board. During the
year ended March 31, 2015, the board of directors approved an increase of 1,120,021 shares authorized for issuance. During the
year ended March 31, 2016, the board of directors approved an increase of 2,256,762 shares authorized for issuance.
Performance Based Awards Program
The Company’s Compensation Committee
approved a short-term performance based bonus program for fiscal 2016 with predetermined objectives related to revenue and expense
targets. In the event the fiscal 2016 objectives are met, eighty-percent of the options will vest on June 30, 2016. On August 21,
2015, certain executives and senior managers were granted an aggregate of 377,500 stock options in connection with this program.
At March 31, 2016, it was determined targets were met related to 252,000 stock options which will vest on June 30, 2016. Additionally,
at March 31, 2016, 50,000 stock options expired due to targets that were not met. The remaining 75,500 stock options will vest
at the discretion of the Company’s Compensation Committee on June 30, 2016. In the event the objectives are not met the stock
options will expire unvested. The stock options have an exercise price of $1.16 and if they vest will expire ten years from the
date of grant. At March 31, 2016, it was determined by the Company that it’s probable the discretionary options would vest.
The Company approved a long-term market-based
stock option bonus program for senior managers. Vesting of the stock options granted as part of this program is contingent upon
the achievement of four separate target stock prices. The market-based options vest based on the 30 trading day trailing average
of the stock price of the Company’s common stock with options vesting in 25% increments at each of the target stock prices.
On the last day of each quarter, the chief executive officer and/or chief financial officer will determine if any of the target
stock prices have been met by evaluating the period between the quarter end date and the grant date of the option. In the event
that a target stock price has been met, the senior manager will be notified that such options have vested. At the end of five
years from the date of the grant, if the stock target prices have not been met, then the unvested portion of the option will expire.
On August 21, 2015, certain senior managers were granted an aggregate of 118,750 stock options in connection with this program.
The stock options have an exercise price of $1.16 and if they vest will expire ten years from the date of grant.
Stock-Based Compensation
The Company issues service, performance and
market-based stock options to employees and non-employees. The Company estimates the fair value of service and performance stock
option awards using the Black-Scholes option pricing model. The Company estimates the fair value of market-based stock option awards
using a Monte-Carlo simulation. Compensation expense for stock option awards is amortized on a straight-line basis over the awards’
vesting period. Compensation expense includes the impact of an estimate for forfeitures for all stock options.
The expected term of the stock options represents
the average period the stock options are expected to remain outstanding and is based on the expected term calculated using the
approach prescribed by the Securities and Exchange Commission's Staff Accounting Bulletin No. 110 for “plain
vanilla” options. The expected stock price volatility for the Company’s stock options was determined by using an average
of the historical volatilities of the Company and its industry peers. The Company will continue to analyze the stock price volatility
and expected term assumptions as more data for the Company’s common stock and exercise patterns become available. The risk-free
interest rate assumption is based on the U.S. Treasury instruments whose term was consistent with the expected term of the Company’s
stock options. The expected dividend assumption is based on the Company’s history and expectation of dividend payouts. The
Company estimates forfeitures based on historical experience and reduces compensation expense accordingly. The estimated forfeiture
rates used during the year ended March 31, 2016 ranged from 1.18% to 4.71%. The estimated forfeiture rates used during the year
ended March 31, 2015 ranged from 0.21% to 0.85%.
The Company estimated the fair value of employee
and non-employee stock options using the Black-Scholes option pricing model. The fair value of employee stock options is being
amortized on a straight-line basis over the requisite service periods of the respective awards. The fair value of employee stock
options was estimated using the following weighted-average assumptions:
|
|
|
Year Ended March 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
Fair value of the Company’s common stock on date of grant
|
|
$
|
1.18
|
|
|
$
|
2.28
|
|
|
Expected Term
|
|
|
6.39 yrs
|
|
|
|
6.67 yrs
|
|
|
Risk-free interest rate
|
|
|
1.63%
|
|
|
|
1.70%
|
|
|
Dividend yield
|
|
|
0.00%
|
|
|
|
0.00%
|
|
|
Volatility
|
|
|
93.0%
|
|
|
|
93.0%
|
|
The weighted-average fair values of options
granted during the years ended March 31, 2016 and 2015 were $0.89 and $1.92, respectively.
Share-based awards compensation expense is as follows:
|
|
|
Stock-based
Compensation
for the Year Ended
March 31, 2016
|
|
|
Stock-based
Compensation
for the Year Ended
March 31, 2015
|
|
|
Cost of revenues
|
|
$
|
364,000
|
|
|
$
|
235,000
|
|
|
Research and development
|
|
|
339,000
|
|
|
|
339,000
|
|
|
Selling, general and administrative
|
|
|
1,320,000
|
|
|
|
1,197,000
|
|
|
Total stock-based compensation
|
|
$
|
2,023,000
|
|
|
$
|
1,771,000
|
|
At March 31, 2016, there were unrecognized
compensation costs of $1,958,000 related to stock options which are expected to be recognized over a weighted-average amortization
period of 1.42 years.
No income tax benefit has been recognized relating to stock-based
compensation expense and no tax benefits have been realized from exercised stock options.
Stock-Based Award Activity
Stock-based awards outstanding at March 31,
2016 under the various plans are as follows:
|
Plan
|
|
|
Awards Outstanding
|
|
|
2004 Plan
|
|
|
|
1,000
|
|
|
2006 Plan
|
|
|
|
859,000
|
|
|
2011 Plan
|
|
|
|
2,909,000
|
|
|
|
|
|
|
|
3,769,000
|
|
|
Awards available for grant as of March 31, 2016
|
|
|
|
2,917,000
|
|
Stock options award activity is as follows:
|
|
|
Number of
Shares
|
|
|
Weighted-
Average
Exercise Price
|
|
|
Weighted-
Average
Contractual Term
|
|
|
Aggregate
Intrinsic
Value
|
|
|
Outstanding at April 1, 2015
|
|
|
2,877,000
|
|
|
$
|
6.96
|
|
|
|
|
|
|
|
|
|
|
Options granted
|
|
|
1,421,000
|
|
|
|
1.18
|
|
|
|
|
|
|
|
|
|
|
Options exercised
|
|
|
–
|
|
|
|
–
|
|
|
|
|
|
|
|
|
|
|
Options forfeited
|
|
|
(190,000
|
)
|
|
|
2.23
|
|
|
|
|
|
|
|
|
|
|
Options expired
|
|
|
(339,000
|
)
|
|
|
16.25
|
|
|
|
|
|
|
|
|
|
|
Outstanding at March 31, 2016
|
|
|
3,769,000
|
|
|
$
|
4.18
|
|
|
|
7.80
|
|
|
$
|
5,000
|
|
|
Exercisable at March 31, 2016
|
|
|
2,075,000
|
|
|
$
|
5.97
|
|
|
|
6.84
|
|
|
$
|
5,000
|
|
The aggregate intrinsic value of stock options is calculated as
the difference between the exercise price of the underlying stock options and the fair value of the Company’s common stock,
or $0.95 per share at March 31, 2016.
Restricted stock award activity is as follows:
|
|
|
Number of
Shares
|
|
|
Weighted
Average Award
Date Fair Value
per Share
|
|
|
Unvested restricted stock awards outstanding at April 1, 2015
|
|
|
–
|
|
|
$
|
–
|
|
|
Restricted stock awards granted
|
|
|
65,000
|
|
|
|
0.99
|
|
|
Restricted stock awards vested
|
|
|
(65,000
|
)
|
|
|
0.99
|
|
|
Restricted stock awards forfeited
|
|
|
–
|
|
|
|
–
|
|
|
Unvested restricted stock awards outstanding at March 31, 2016
|
|
|
–
|
|
|
$
|
–
|
|
Restricted stock activities were
issued to non-employee directors for year ended March 31, 2016.
The Company did not capitalize any cost associated with stock-based
compensation.
The Company issues new shares of common stock
upon exercise of stock based awards.
NOTE 15 – Income Taxes
The Company has the following net deferred
tax assets:
|
|
|
March 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
Deferred tax assets:
|
|
|
|
|
|
|
|
|
|
Net operating loss carryforwards
|
|
$
|
36,454,000
|
|
|
$
|
36,661,000
|
|
|
Research and development tax credit carryforwards
|
|
|
1,710,000
|
|
|
|
1,667,000
|
|
|
Stock-based compensation
|
|
|
5,083,000
|
|
|
|
4,880,000
|
|
|
Reserves and accruals
|
|
|
1,111,000
|
|
|
|
1,731,000
|
|
|
Other deferred tax assets
|
|
|
241,000
|
|
|
|
49,000
|
|
|
State income taxes
|
|
|
(1,000
|
)
|
|
|
(1,000
|
)
|
|
Basis difference in assets
|
|
|
8,000
|
|
|
|
5,000
|
|
|
Total deferred tax assets
|
|
$
|
44,606,000
|
|
|
$
|
44,992,000
|
|
|
Deferred tax liabilities:
|
|
|
|
|
|
|
|
|
|
Unrealized gain on Ruthigen
|
|
|
–
|
|
|
|
(1,105,000
|
)
|
|
Net deferred tax asset
|
|
|
44,606,000
|
|
|
|
43,887,000
|
|
|
Valuation allowance
|
|
|
(44,606,000
|
)
|
|
|
(43,887,000
|
)
|
|
Net deferred tax asset
|
|
$
|
–
|
|
|
$
|
–
|
|
The Company’s recorded income tax expense,
net of the change in the valuation allowance, for each of the periods presented is as follows:
|
|
|
Years Ended March 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
Income tax (benefit)
|
|
$
|
(719,000
|
)
|
|
$
|
(2,613,000
|
)
|
|
Change in valuation allowance
|
|
|
719,000
|
|
|
|
2,613,000
|
|
|
Net income tax expense
|
|
$
|
–
|
|
|
$
|
–
|
|
A reconciliation of the statutory federal income
tax rate to the Company’s effective tax rate is as follows:
|
|
|
Years Ended March 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
Expected federal statutory rate
|
|
|
(34.0%
|
)
|
|
|
(34.0%
|
)
|
|
State income taxes, net of federal benefit
|
|
|
(1.8%
|
)
|
|
|
(1.8%
|
)
|
|
Research and development credit
|
|
|
(0.4%
|
)
|
|
|
(0.2%
|
)
|
|
Foreign earnings taxed at different rates
|
|
|
0.7%
|
|
|
|
0.5%
|
|
|
Effect of state net operating loss expiration
|
|
|
5.5%
|
|
|
|
–
|
|
|
Effect of permanent differences
|
|
|
(3.3%
|
)
|
|
|
(20.6%
|
)
|
|
Impact of foreign exchange rate fluctuations on foreign deferred income taxes
|
|
|
8.5%
|
|
|
|
25.0%
|
|
|
Impact of change in foreign net operating loss
|
|
|
6.3%
|
|
|
|
(9.6%
|
)
|
|
Cancellation of stock options and other true-ups
|
|
|
0.0%
|
|
|
|
(2.7%
|
)
|
|
Adjustment of NOL due to Ruthigen deconsolidation
|
|
|
0.0%
|
|
|
|
8.6%
|
|
|
True-up of state deferred assets
|
|
|
11.4%
|
|
|
|
3.0%
|
|
|
|
|
|
(7.1%
|
)
|
|
|
(31.8%
|
)
|
|
Change in valuation allowance
|
|
|
7.1%
|
|
|
|
31.8%
|
|
|
Totals
|
|
|
0.0%
|
|
|
|
0.0%
|
|
At March 31, 2016, the Company had net operating loss carryforwards
for federal, state and foreign income tax purposes of approximately $92,883,000, $42,588,000 and $11,666,000, respectively. The
federal net operating loss carryforwards will expire at various dates, if not utilized, beginning in the fiscal year ending March
31, 2021. The state net operating loss carryforwards will expire at various dates, if not utilized, beginning in the fiscal year
ending March 31, 2018. The foreign net operating loss carryforwards will expire at various dates, if not utilized, beginning in
the fiscal year ending March 31, 2018. The Company also had, at March 31, 2016, federal and state research credit carryforwards
of approximately $870,000 and $790,000, respectively. The federal credits will expire, if not utilized at various dates, beginning in the fiscal year ending March 31, 2026, and the state credits do not expire. The Company also had, at March
31, 2016 foreign tax credits carryforwards of approximately $50,000. The foreign credits will expire, if not utilized at various
dates, beginning in the fiscal year ending March 31, 2024.
The Company has completed a study to
assess whether a change in control has occurred or whether there have been multiple changes of control since the
Company’s formation through March 31, 2016. The Company determined, based on the results of the study, no change in
control occurred for purposes of Internal Revenue Code section 382. The Company, after considering all available evidence,
fully reserved for these and its other deferred tax assets since it is more likely than not such benefits will not be
realized in future periods. The Company has incurred losses for both financial reporting and income tax purposes for the year
ended March 31, 2016. Accordingly, the Company is continuing to fully reserve for its deferred tax assets. The Company will
continue to evaluate its deferred tax assets to determine whether any changes in circumstances could affect the realization
of their future benefit. If it is determined in future periods that portions of the Company’s deferred income tax
assets satisfy the realization standards, the valuation allowance will be reduced accordingly.
As a result of certain realization requirements
of Accounting Standards Codification Topic 718, the table of deferred tax assets and liabilities shown above does not include certain
deferred tax assets at March 31, 2016 that arose directly from tax deductions related to equity compensation in excess of compensation
recognized for financial reporting purposes. Equity will be increased by approximately $533,000 if and when such deferred tax assets
are ultimately realized.
The Company only recognizes tax benefits from
an uncertain tax position if it is more likely than not that the tax position will be sustained on examination by the taxing authorities,
based on the technical merits of the position. The tax benefits recognized in the financial statements from such a position are
measured based on the largest benefit that has a greater than fifty percent likelihood of being realized upon ultimate resolution.
To date, the Company has not recognized such tax benefits in its consolidated financial statements.
The Company has identified its federal tax
return and its state tax return in California as major tax jurisdictions. The Company also filed tax returns in foreign jurisdictions,
principally Mexico and The Netherlands. The Company’s evaluation of uncertain tax matters was performed for tax years ended
through March 31, 2016. Generally, the Company is subject to audit for the years ended March 31, 2015, 2014 and 2013 and may be
subject to audit for amounts relating to net operating loss carryforwards generated in periods prior to March 31, 2015. The Company
has elected to retain its existing accounting policy with respect to the treatment of interest and penalties attributable to income
taxes, and continues to reflect interest and penalties attributable to income taxes, to the extent they arise, as a component of
its income tax provision or benefit as well as its outstanding income tax assets and liabilities. The Company believes that its
income tax positions and deductions would be sustained on audit and does not anticipate any adjustments.
The Company does not have any tax positions
for which it is reasonably possible the total amount of gross unrecognized tax benefits will increase or decrease within 12 months
of March 31, 2016. The unrecognized tax benefits may increase or change during the next year for items that arise in the ordinary
course of business.
NOTE 16 – Employee Benefit
Plan
The Company has a program to contribute and
administer a qualified 401(k) plan. Under the 401(k) plan, the Company matches employee contributions to the plan up to 4% of the
employee’s salary. Company contributions to the plans amounted to an aggregate of $158,000 and $137,000 for the years ended
March 31, 2016 and 2015, respectively.
NOTE 17 – Geographic Information
The Company generates product revenues from
products which are sold into the human and animal healthcare markets, and the Company generates service revenues from laboratory
testing services which are provided to medical device manufacturers.
The following table shows the Company’s
product revenues by geographic region:
|
|
|
Year Ended March 31,
|
|
|
|
|
|
|
|
|
|
|
2016
|
|
|
2015
|
|
|
$ Change
|
|
|
% Change
|
|
|
United States
|
|
$
|
4,371,000
|
|
|
$
|
1,978,000
|
|
|
$
|
2,393,000
|
|
|
|
121%
|
|
|
Latin America
|
|
|
4,965,000
|
|
|
|
5,053,000
|
|
|
|
(88,000
|
)
|
|
|
(2%
|
)
|
|
Europe and Rest of the World
|
|
|
3,706,000
|
|
|
|
2,908,000
|
|
|
|
798,000
|
|
|
|
27%
|
|
|
|
|
|
13,042,000
|
|
|
|
9,939,000
|
|
|
|
3,103,000
|
|
|
|
31%
|
|
|
Product license fees and royalties
|
|
|
981,000
|
|
|
|
3,056,000
|
|
|
|
(2,075,000
|
)
|
|
|
(68%
|
)
|
|
Total
|
|
$
|
14,023,000
|
|
|
$
|
12,995,000
|
|
|
$
|
1,028,000
|
|
|
|
8%
|
|
In the year ended March 31, 2016, product license
fees and royalties revenue declined primarily as a result of the termination of the Company’s agreement with its former partner
Innovacyn and a decrease in the amortization of upfront fees paid by Laboratorios Sanfer.
The following table shows the Company’s
product license fees and royalties revenue by partner:
|
|
|
Year Ended March 31,
|
|
|
|
|
|
|
|
|
|
|
2016
|
|
|
2015
|
|
|
$ Change
|
|
|
% Change
|
|
|
Exeltis/Quinnova
|
|
$
|
201,000
|
|
|
$
|
437,000
|
|
|
$
|
(236,000
|
)
|
|
|
(54%
|
)
|
|
Innovacyn
|
|
|
29,000
|
|
|
|
1,120,000
|
|
|
|
(1,091,000
|
)
|
|
|
(97%
|
)
|
|
Laboratorios Sanfer
|
|
|
751,000
|
|
|
|
1,499,000
|
|
|
|
(748,000
|
)
|
|
|
(50%
|
)
|
|
Total
|
|
$
|
981,000
|
|
|
$
|
3,056,000
|
|
|
$
|
(2,075,000
|
)
|
|
|
(68%
|
)
|
The Company’s service revenues amounted
to $1,061,000 and $859,000 for the years ended March 31, 2016 and 2015.
NOTE 18 – Subsequent Events
On June 29, 2015, the Company’s stockholders approved
an amendment to the Restated Certificate of Incorporation, as amended, and authorized the Company’s Board of Directors, if
in their judgment it is necessary, to effect a reverse stock split of our outstanding common stock, $0.0001 par value per share,
at a whole number ratio in the range of 1-for-5 to 1-for-9, such ratio to be determined in the discretion of the Board of Directors,
and to proportionally decrease the total number of shares that the Company is authorized to issue by a factor of 1-for-5 to 1-for-9,
such ratio to be determined in the sole discretion of the Board of Directors. On June 2, 2016, the Company’s Board of Directors
approved the reverse stock split with a ratio of 1-for-5 and authorized the Corporation to take all steps necessary to effect the
reverse stock split to become effective June 24, 2016.
OCULUS INNOVATIVE SCIENCES, INC. AND SUBSIDIARIES
Condensed Consolidated Balance Sheets
(In thousands, except share and per share amounts)
|
|
|
June 30,
|
|
|
March 31,
|
|
|
|
|
2016
|
|
|
2016
|
|
|
|
|
(Unaudited)
|
|
|
|
|
|
ASSETS
|
|
|
|
|
|
|
|
|
|
Current assets:
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
4,970
|
|
|
$
|
7,469
|
|
|
Accounts receivable, net
|
|
|
2,593
|
|
|
|
2,274
|
|
|
Inventories, net
|
|
|
1,992
|
|
|
|
1,640
|
|
|
Prepaid expenses and other current assets
|
|
|
942
|
|
|
|
1,505
|
|
|
Total current assets
|
|
|
10,497
|
|
|
|
12,888
|
|
|
Property and equipment, net
|
|
|
764
|
|
|
|
850
|
|
|
Other assets
|
|
|
59
|
|
|
|
65
|
|
|
Total assets
|
|
$
|
11,320
|
|
|
$
|
13,803
|
|
|
|
|
|
|
|
|
|
|
|
|
LIABILITIES AND STOCKHOLDERS’ EQUITY
|
|
|
|
|
|
|
|
|
|
Current liabilities:
|
|
|
|
|
|
|
|
|
|
Accounts payable
|
|
$
|
1,368
|
|
|
$
|
1,337
|
|
|
Accrued expenses and other current liabilities
|
|
|
1,442
|
|
|
|
1,526
|
|
|
Deferred revenue
|
|
|
656
|
|
|
|
574
|
|
|
Current portion of long-term debt
|
|
|
66
|
|
|
|
114
|
|
|
Total current liabilities
|
|
|
3,532
|
|
|
|
3,551
|
|
|
Deferred revenue, less current portion
|
|
|
38
|
|
|
|
112
|
|
|
Total liabilities
|
|
|
3,570
|
|
|
|
3,663
|
|
|
Commitments and Contingencies (Note 5)
|
|
|
|
|
|
|
|
|
|
Stockholders’ Equity
|
|
|
|
|
|
|
|
|
|
Convertible preferred stock, $0.0001 par value; 714,286 shares authorized, none issued and outstanding at June 30, 2016 and March 31, 2016, respectively
|
|
|
–
|
|
|
|
–
|
|
|
Common stock, $0.0001 par value; 12,000,000 shares authorized at June 30, 2016 and March 31, 2016, 4,200,756 (unaudited) and 4,196,873 shares issued and outstanding at June 30, 2016 and March 31, 2016, respectively (Note 6)
|
|
|
1
|
|
|
|
1
|
|
|
Additional paid-in capital
|
|
|
166,779
|
|
|
|
166,368
|
|
|
Accumulated deficit
|
|
|
(154,943
|
)
|
|
|
(152,375
|
)
|
|
Accumulated other comprehensive loss
|
|
|
(4,087
|
)
|
|
|
(3,854
|
)
|
|
Total stockholders’ equity
|
|
|
7,750
|
|
|
|
10,140
|
|
|
Total liabilities and stockholders’ equity
|
|
$
|
11,320
|
|
|
$
|
13,803
|
|
The accompanying footnotes are an integral part
of these condensed consolidated financial statements.
OCULUS INNOVATIVE SCIENCES, INC. AND SUBSIDIARIES
Condensed Consolidated Statements of Comprehensive
Loss
(In thousands, except per share amounts)
(Unaudited)
|
|
|
Three Months Ended June 30,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
Revenues
|
|
|
|
|
|
|
|
|
|
Product
|
|
$
|
3,509
|
|
|
$
|
2,916
|
|
|
Product licensing fees and royalties
|
|
|
75
|
|
|
|
447
|
|
|
Service
|
|
|
227
|
|
|
|
317
|
|
|
Total revenues
|
|
|
3,811
|
|
|
|
3,680
|
|
|
Cost of revenues
|
|
|
|
|
|
|
|
|
|
Product
|
|
|
1,707
|
|
|
|
1,516
|
|
|
Service
|
|
|
185
|
|
|
|
291
|
|
|
Total cost of revenues
|
|
|
1,892
|
|
|
|
1,807
|
|
|
Gross profit
|
|
|
1,919
|
|
|
|
1,873
|
|
|
Operating expenses
|
|
|
|
|
|
|
|
|
|
Research and development
|
|
|
360
|
|
|
|
467
|
|
|
Selling, general and administrative
|
|
|
4,130
|
|
|
|
3,717
|
|
|
Total operating expenses
|
|
|
4,490
|
|
|
|
4,184
|
|
|
Loss from operations
|
|
|
(2,571
|
)
|
|
|
(2,311
|
)
|
|
Interest expense
|
|
|
(1
|
)
|
|
|
–
|
|
|
Interest income
|
|
|
1
|
|
|
|
–
|
|
|
Loss due to change in fair value of derivative liabilities
|
|
|
–
|
|
|
|
(59
|
)
|
|
Other income, net
|
|
|
3
|
|
|
|
30
|
|
|
Net loss
|
|
|
(2,568
|
)
|
|
|
(2,340
|
)
|
|
Net loss per common share: basic and diluted
|
|
$
|
(0.61
|
)
|
|
$
|
(0.77
|
)
|
|
Weighted-average number of shares used in common share calculations:
|
|
|
|
|
|
|
|
|
|
Basic and diluted
|
|
|
4,198
|
|
|
|
3,034
|
|
|
Other comprehensive loss
|
|
|
|
|
|
|
|
|
|
Net loss
|
|
$
|
(2,568
|
)
|
|
$
|
(2,340
|
)
|
|
Foreign currency translation adjustments
|
|
|
(233
|
)
|
|
|
(71
|
)
|
|
Comprehensive loss
|
|
$
|
(2,801
|
)
|
|
$
|
(2,411
|
)
|
The accompanying footnotes are an integral part
of these condensed consolidated financial statements.
OCULUS INNOVATIVE SCIENCES, INC. AND SUBSIDIARIES
Condensed Consolidated Statements of Cash
Flows
(In thousands)
(Unaudited)
|
|
|
Three Months Ended June 30,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
|
|
(In thousands)
|
|
|
Cash flows from operating activities
|
|
|
|
|
|
|
|
|
|
Net loss
|
|
$
|
(2,568
|
)
|
|
$
|
(2,340
|
)
|
|
Adjustments to reconcile net loss to net cash used in operating activities:
|
|
|
|
|
|
|
|
|
|
Depreciation and amortization
|
|
|
61
|
|
|
|
62
|
|
|
Stock-based compensation
|
|
|
411
|
|
|
|
412
|
|
|
Service provider expenses settled with common stock
|
|
|
–
|
|
|
|
107
|
|
|
Loss due to change in fair value of derivative liabilities
|
|
|
–
|
|
|
|
59
|
|
|
Foreign currency transaction gain
|
|
|
(12
|
)
|
|
|
(9
|
)
|
|
Changes in operating assets and liabilities:
|
|
|
|
|
|
|
|
|
|
Accounts receivable, net
|
|
|
(384
|
)
|
|
|
(1,052
|
)
|
|
Inventories, net
|
|
|
(410
|
)
|
|
|
(82
|
)
|
|
Prepaid expenses and other current assets
|
|
|
538
|
|
|
|
84
|
|
|
Accounts payable
|
|
|
55
|
|
|
|
284
|
|
|
Accrued expenses and other current liabilities
|
|
|
(126
|
)
|
|
|
265
|
|
|
Deferred revenue
|
|
|
58
|
|
|
|
(375
|
)
|
|
Net cash used in operating activities
|
|
|
(2,377
|
)
|
|
|
(2,585
|
)
|
|
Cash flows from investing activities:
|
|
|
|
|
|
|
|
|
|
Purchases of property and equipment
|
|
|
(14
|
)
|
|
|
(148
|
)
|
|
Proceeds from sale of long-term investment
|
|
|
–
|
|
|
|
4,538
|
|
|
Long-term deposits
|
|
|
3
|
|
|
|
(12
|
)
|
|
Net cash (used in) provided by investing activities
|
|
|
(11
|
)
|
|
|
4,378
|
|
|
Cash flows from financing activities:
|
|
|
|
|
|
|
|
|
|
Proceeds from issuance of common stock, net of offering costs
|
|
|
–
|
|
|
|
879
|
|
|
Principal payments on long-term debt
|
|
|
(48
|
)
|
|
|
(52
|
)
|
|
Net cash (used in) provided by financing activities
|
|
|
(48
|
)
|
|
|
827
|
|
|
Effect of exchange rate on cash and cash equivalents
|
|
|
(63
|
)
|
|
|
(4
|
)
|
|
Net (decrease) increase in cash and cash equivalents
|
|
|
(2,499
|
)
|
|
|
2,616
|
|
|
Cash and cash equivalents, beginning of period
|
|
|
7,469
|
|
|
|
6,136
|
|
|
Cash and cash equivalents, end of period
|
|
$
|
4,970
|
|
|
$
|
8,752
|
|
|
Supplemental disclosure of cash flow information:
|
|
|
|
|
|
|
|
|
|
Cash paid for interest
|
|
$
|
1
|
|
|
$
|
–
|
|
|
Non-cash operating and financing activities:
|
|
|
|
|
|
|
|
|
|
Issuance of common stock to settle obligation
|
|
$
|
–
|
|
|
$
|
96
|
|
The accompanying footnotes are an integral part
of these condensed consolidated financial statements.
OCULUS INNOVATIVE SCIENCES, INC. AND SUBSIDIARIES
Notes to Condensed Consolidated Financial
Statements
(Unaudited)
|
Note 1.
|
Organization and Recent Developments
|
Organization
Oculus Innovative Sciences, Inc. (the “Company”)
was incorporated under the laws of the State of California in April 1999 and was reincorporated under the laws of the State of
Delaware in December 2006. The Company’s principal office is located in Petaluma, California. The Company is a specialty
pharmaceutical company that develops and markets solutions for the treatment of dermatological conditions and advanced tissue care.
The Company’s products, which are sold throughout the United States and 39 countries around the world, have improved patient
outcomes for more than five million patients globally by reducing infections, itch, pain, scarring, odor and harmful inflammatory
responses.
Basis of Presentation
The accompanying unaudited condensed
consolidated financial statements as of June 30, 2016 and for the three months then ended have been prepared in accordance
with the accounting principles generally accepted in the United States of America for interim financial information and
pursuant to the instructions to Form 10-Q and Article 8 of Regulation S-X of the Securities and Exchange Commission
(“SEC”) and on the same basis as the Company prepares its annual audited consolidated financial statements. The
condensed consolidated balance sheet as of June 30, 2016 and the condensed consolidated statements of comprehensive loss and
cash flows for the three months ended June 30, 2016 and 2015 are unaudited, but include all adjustments, consisting only of
normal recurring adjustments, which the Company considers necessary for a fair presentation of the consolidated financial
position, operating results and cash flows for the periods presented. The results for the three months ended June 30, 2016
are not necessarily indicative of results to be expected for the year ending March 31, 2017 or for any future interim period.
The condensed consolidated balance sheet at March 31, 2016 has been derived from audited consolidated financial statements.
These unaudited condensed consolidated financial statements of the Company have been prepared in accordance with U.S.
Generally Accepted Accounting Principles (“GAAP”) for interim financial information. Accordingly, they do not
include all of the information and notes required by GAAP for complete financial statements. The accompanying
condensed consolidated financial statements should be read in conjunction with the consolidated financial statements for the
year ended March 31, 2016, and notes thereto included in the Company’s annual report on Form 10-K, which was filed with
the SEC on June 21, 2016.
Reverse Stock Split
Effective as of the open of business on June
24, 2016, the Company effected a reverse stock split of its common stock, par value $0.0001 per share. Every 5 shares of common
stock were reclassified and combined into one share of common stock. No fractional shares were issued as a result of the reverse
stock split. Instead, stockholders entitled to receive fractional shares received
cash in the amount equal to the closing price per share of the Company’s common stock as reported on the NASDAQ Capital Market
as of 5:00 p.m. Eastern Time on June 24, 2016, multiplied by the fraction of one share owned by the stockholder. The reverse stock
split reduced the number of shares of the Company’s common stock outstanding from 21,004,857 to 4,200,756. The total number
of authorized shares of common stock was also proportionally decreased by a ratio of 1:5 and the par value per share of the common
stock continued to be $0.0001.
All common shares and per share amounts contained
in the condensed consolidated financial statements have been retroactively adjusted to reflect a 1 for 5 reverse stock split.
|
Note 2.
|
Liquidity
and Financial Condition
|
The Company reported a net loss of $2,568,000
for the three months ended June 30, 2016. At June 30, 2016 and March 31, 2016, the Company’s accumulated deficit amounted
to $154,943,000 and $152,375,000, respectively. The Company had working capital of $6,965,000 and $9,337,000 as of June 30, 2016
and March 31, 2016, respectively. The Company expects to continue incurring losses for the foreseeable future and may need to raise
additional capital to pursue its product development initiatives, penetrate markets for the sale of its products and continue as
a going concern.
Management believes that the Company has access
to additional capital resources through possible public or private equity offerings, debt financings, corporate collaborations
or other means; however, the Company cannot provide any assurance that other new financings will be available on commercially acceptable
terms, if needed. If the economic climate in the U.S. deteriorates, the Company’s ability to raise additional capital could
be negatively impacted. If the Company is unable to secure additional capital, it may be required to curtail its research and development
initiatives and take additional measures to reduce costs in order to conserve its cash in amounts sufficient to sustain operations
and meet its obligations. These measures could cause significant delays in the Company’s efforts to commercialize its products,
which is critical to the realization of its business plan and the future operations of the Company. These matters raise substantial
doubt about the Company’s ability to continue as a going concern. The accompanying condensed consolidated financial statements
do not include any adjustments that may be necessary should the Company be unable to continue as a going concern.
|
Note 3.
|
Summary of Significant Accounting Policies
|
Use of Estimates
The preparation of condensed consolidated financial
statements in conformity with accounting principles generally accepted in the United States of America requires management to make
estimates and assumptions that affect the reported amounts of assets and liabilities and disclosures of contingent liabilities
at the dates of the condensed consolidated financial statements and the reported amounts of revenues and expenses during the reporting
periods. Actual results could differ from these estimates. Significant estimates and assumptions include reserves and write-downs
related to receivables and inventories, the recoverability of long-lived assets, the valuation allowance relating to the Company’s
deferred tax assets, valuation of equity and derivative instruments, debt discounts, valuation of investments, and the estimated
amortization periods of upfront product licensing fees received from customers. Periodically, the Company evaluates and adjusts
estimates accordingly. The allowance for doubtful accounts represents probable credit losses of $15,000 at June 30, 2016 and March
31, 2016. Additionally at June 30, 2016 and March 31, 2016 the Company has allowances of $715,000 and $653,000, respectively, related
to potential discounts, returns, distributor fees and rebates. The allowances are included in Accounts Receivable, net in the accompanying
condensed consolidated balance sheets.
Net Loss per Share
The Company computes basic net loss per share
by dividing net loss per share available to common stockholders by the weighted average number of common shares outstanding for
the period and excludes the effects of any potentially dilutive securities. Diluted earnings per share, if presented, would include
the dilution that would occur upon the exercise or conversion of all potentially dilutive securities into common stock using the
“treasury stock” and/or “if converted” methods as applicable. The following were excluded from the computation
of diluted shares outstanding due to the losses for the three months ended June 30, 2016 and 2015, as they would have had an anti-dilutive
impact on the Company’s net loss (all amounts are rounded to the nearest thousand).
|
|
|
June 30,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
Options to purchase common stock
|
|
|
754,000
|
|
|
|
596,000
|
|
|
Warrants to purchase common stock
|
|
|
1,468,000
|
|
|
|
1,548,000
|
|
|
|
|
|
2,222,000
|
|
|
|
2,144,000
|
|
Common Stock Purchase Warrants and Other
Derivative Financial Instruments
The Company classifies common stock purchase
warrants and other free standing derivative financial instruments as equity if the contracts (i) require physical settlement or
net-share settlement or (ii) give the Company a choice of net-cash settlement or settlement in its own shares (physical settlement
or net-share settlement). The Company classifies any contracts that (i) require net-cash settlement (including a requirement to
net cash settle the contract if an event occurs and if that event is outside the control of the Company), (ii) give the counterparty
a choice of net cash settlement or settlement in shares (physical settlement or net-share settlement), or (iii) contain reset provisions
as either an asset or a liability. The Company assesses classification of its freestanding derivatives at each reporting date to
determine whether a change in classification between assets and liabilities is required. The Company determined that its freestanding
derivatives, which principally consist of warrants to purchase common stock, satisfied the criteria for classification as equity
instruments, other than certain warrants that contained reset provisions and certain warrants that required net-cash settlement
that the Company classified as derivative liabilities.
Revenue Recognition and Accounts Receivable
The Company generates revenue from sales of
its products to a customer base including hospitals, medical centers, doctors, pharmacies, distributors and wholesalers. The Company
sells products directly to end users and to distributors. The Company also entered into agreements to license its technology and
products.
The Company also provides regulatory compliance
testing and quality assurance services to medical device and pharmaceutical companies.
The Company records revenue when (i) persuasive
evidence of an arrangement exists, (ii) delivery has occurred, (iii) the fee is fixed or determinable, and (iv) collectability
of the sale is reasonably assured.
The Company requires all product sales to be
supported by evidence of a sale transaction that clearly indicates the selling price to the customer, shipping terms and payment
terms. Evidence of an arrangement generally consists of a contract or purchase order approved by the customer. The Company has
ongoing relationships with certain customers from which it customarily accepts orders by telephone in lieu of purchase orders.
The Company recognizes revenue at the time
it receives confirmation that the goods were either tendered at their destination, when shipped “FOB destination,”
or transferred to a shipping agent, when shipped “FOB shipping point.” Delivery to the customer is deemed to have occurred
when the customer takes title to the product. Generally, title passes to the customer upon shipment, but could occur when the customer
receives the product based on the terms of the agreement with the customer.
The selling prices of all goods are fixed,
and agreed to with the customer, prior to shipment. Selling prices are generally based on established list prices. The right to
return product is customarily based on the terms of the agreement with the customer. The Company estimates and accrues for potential
returns and records this as a reduction of revenue in the same period the related revenue is recognized. Additionally, distribution
fees are paid to certain wholesale distributors based on contractually determined rates. The Company estimates and accrues the
fee on shipment to the respective wholesale distributors and recognizes the fee as a reduction of revenue in the same period the
related revenue is recognized. The Company also offers cash discounts to certain customers, generally 2% of the sales price, as
an incentive for prompt payment. The Company accounts for cash discounts by reducing accounts receivable by the prompt pay discount
amount and recognizes the discount as a reduction of revenue in the same period the related revenue is recognized. Additionally,
the Company participates in certain rebate programs which provide discounted prescriptions to qualified patients. The Company contracts
a third-party to administer the program. The Company estimates and accrues for future rebates based on historical data for rebate
redemption rates and the historical value of redemptions. Rebates are recognized as a reduction of revenue in the same period the
related revenue is recognized.
The Company evaluates the creditworthiness
of new customers and monitors the creditworthiness of its existing customers to determine whether an event or changes in their
financial circumstances would raise doubt as to the collectability of a sale at the time in which a sale is made. Payment terms
on sales made in the United States are generally 30 days and are extended up to 90 days for initial product launches, payment
terms internationally generally range from prepaid prior to shipment to 90 days.
In the event a sale is made to a customer under
circumstances in which collectability is not reasonably assured, the Company either requires the customer to remit payment prior
to shipment or defers recognition of the revenue until payment is received. The Company maintains a reserve for amounts which may
not be collectible due to risk of credit losses.
Product license revenue is generated through
agreements with strategic partners for the commercialization of Microcyn® products. The terms of the agreements sometimes include
non-refundable upfront fees. The Company analyzes multiple element arrangements to determine whether the elements can be separated.
Analysis is performed at the inception of the arrangement and as each product is delivered. If a product or service is not separable,
the combined deliverables are accounted for as a single unit of accounting and recognized over the performance obligation period.
When appropriate, the Company defers recognition
of non-refundable upfront fees. If the Company has continuing performance obligations then such up-front fees are deferred and
recognized over the period of continuing involvement.
The Company recognizes royalty revenues from
licensed products upon the sale of the related products.
Revenue from consulting contracts is recognized
as services are provided. Revenue from testing contracts is recognized as tests are completed and a final report is sent to the
customer.
Inventories
Inventories are stated at the lower of cost,
cost being determined on a standard cost basis (which approximates actual cost on a first-in, first-out basis), or market.
Due to changing market conditions, estimated
future requirements, age of the inventories on hand and production of new products, the Company regularly reviews inventory quantities
on hand and records a provision to write down excess and obsolete inventory to its estimated net realizable value. The Company
recorded reserves to reduce the carrying amounts of inventories to their net realizable value in the amounts of $167,000 and $164,000
at June 30, 2016 and March 31, 2016.
Income Taxes
The Company is required to determine the aggregate
amount of income tax expense or loss based upon tax statutes in jurisdictions in which it conducts business. In making these estimates,
the Company adjusts its results determined in accordance with generally accepted accounting principles for items that are treated
differently by the applicable taxing authorities. Deferred tax assets and liabilities resulting from these differences are reflected
on its balance sheet for temporary differences in loss and credit carryforwards that will reverse in subsequent years. The Company
also establishes a valuation allowance against deferred tax assets when it is more likely than not that some or all of the deferred
tax assets will not be realized. Valuation allowances are based, in part, on predictions that management must make as to the results
in future periods. The outcome of events could differ over time which would require that the Company makes changes in its valuation
allowance.
Financial Assets and Liabilities
Financial instruments, including cash and cash
equivalents, accounts receivable, accounts payable and accrued expenses and other liabilities are carried at cost, which management
believes approximates fair value due to the short-term nature of these instruments. The fair value of capital lease obligations
and equipment loans approximates their carrying amounts as a market rate of interest is attached to their repayment. The Company
measures the fair value of financial assets and liabilities based on the exchange price that would be received for an asset or
paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly
transaction between market participants on the measurement date. The Company maximizes the use of observable inputs and minimizes
the use of unobservable inputs when measuring fair value. The Company uses three levels of inputs that may be used to measure fair
value:
Level 1 – quoted prices in
active markets for identical assets or liabilities
Level 2 – quoted prices for
similar assets and liabilities in active markets; quoted prices for identical or similar instruments in markets that are not active;
and model-derived valuations in which all significant inputs and significant value drivers are observable in active markets.
Level 3 – inputs that are
unobservable (for example cash flow modeling inputs based on assumptions)
Level 3 liabilities are valued using unobservable inputs to the
valuation methodology that are significant to the measurement of the fair value of the liabilities. For fair value measurements
categorized within Level 3 of the fair value hierarchy, the Company’s accounting and finance department, who report to the
Chief Financial Officer, determine its valuation policies and procedures. The development and determination of the unobservable
inputs for Level 3 fair value measurements and fair value calculations are the responsibility of the Company’s accounting
and finance department and are approved by the Chief Financial Officer.
Level 3 Valuation Techniques
:
Level 3 financial liabilities consist of the
derivative liabilities for which there is no current market for these securities such that the determination of fair value requires
significant judgment or estimation. Changes in fair value measurements categorized within Level 3 of the fair value hierarchy are
analyzed each period based on changes in estimates or assumptions and recorded as appropriate.
As of June 30, 2016 and March 31, 2016, there
were no transfers in or out of Level 3 from other levels in the fair value hierarchy.
Subsequent Events
Management has evaluated subsequent events
or transactions occurring through the date the condensed consolidated financial statements were issued (Note 11).
Recent Accounting Pronouncements
Accounting standards that have been issued
or proposed by the Financial Accounting Standards Board (“FASB”), SEC and/or other standards-setting bodies that do
not require adoption until a future date are not expected to have a material impact on the condensed consolidated financial statements
upon adoption.
|
Note 4.
|
Condensed Consolidated Balance Sheets
|
Inventories, net
Inventories, net consist of the following:
|
|
|
June 30,
|
|
|
March 31,
|
|
|
|
|
2016
|
|
|
2016
|
|
|
Raw materials
|
|
$
|
1,218,000
|
|
|
$
|
1,104,000
|
|
|
Finished goods
|
|
|
774,000
|
|
|
|
536,000
|
|
|
|
|
$
|
1,992,000
|
|
|
$
|
1,640,000
|
|
|
Note 5.
|
Commitments and Contingencies
|
Legal Matters
The Company, on occasion, may be involved in
legal matters arising in the ordinary course of its business including matters involving proprietary technology. While management
believes that such matters are currently insignificant, matters arising in the ordinary course of business for which the Company
is or could become involved in litigation may have a material adverse effect on its business, financial condition or results of
operations.
Employment Agreements
As of June 30, 2016 the Company had employment
agreements in place with four of its key executives. The agreements provide, among other things, for the payment of nine to twenty-four
months of severance compensation for terminations under certain circumstances. With respect to these agreements, at June 30, 2016,
the potential severance payments to these key executives would be $1,194,000 and aggregated annual salaries would be $944,000 if
triggered.
|
Note 6.
|
Stockholders’ Equity
|
Authorized Capital
The Company is authorized to issue up to 12,000,000
shares of common stock with a par value of $0.0001 per share and 714,286 shares of convertible preferred stock with a par value
of $0.0001 per share.
|
Note 7.
|
Stock-Based Compensation
|
Performance Based Awards Program
The Company’s Compensation Committee
approved a short-term performance based bonus program for fiscal 2016 with predetermined objectives related to revenue and expense
targets. In the event the fiscal 2016 objectives were met, eighty-percent of the options would have vested on June 30, 2016. On
August 21, 2015, certain executives and senior managers were granted an aggregate of 75,500 stock options in connection with this
program. The stock options have an exercise price of $5.80 and expire ten years from the date of grant. At March 31, 2016, it was
determined targets were met related to 50,400 stock options which vested on June 30, 2016. At March 31, 2016, 10,000 stock options
expired due to targets that were not met. The vesting of the remaining 15,100 stock options was at the discretion of the Company’s
Compensation Committee to be determined during the three months ended June 30, 2016. The Company’s Compensation Committee
determined 14,772 of the 15,100 discretionary stock options would vest at June 30, 2016.
The Company also approved a long-term market-based
stock option bonus program for senior managers. Vesting of the stock options granted as part of this program is contingent upon
the achievement of four separate target stock prices. The market-based options vest based on the 30 trading day trailing average
of the stock price of the Company’s common stock with options vesting in 25% increments at each of the target stock prices.
On the last day of each quarter, the chief executive officer and/or chief financial officer will determine if any of the target
stock prices have been met by evaluating the period between the quarter end date and the grant date of the option. In the event
that a target stock price has been met, the senior manager will be notified that such options have vested. At the end of five years
from the date of the grant, if the stock target prices have not been met, then the unvested portion of the option will expire.
On August 21, 2015, certain senior managers were granted an aggregate of 23,750 stock options in connection with this program.
The stock options have an exercise price of $5.80 and if they vest will expire ten years from the date of grant. None of these
options vested as of June 30, 2016.
Stock-Based Compensation
The Company issues service, performance and
market-based stock options to employees and non-employees. The Company estimates the fair value of service and performance stock
option awards using the Black-Scholes option pricing model. The Company estimates the fair value of market-based stock option awards
using a Monte-Carlo simulation. Compensation expense for stock option awards is amortized on a straight-line basis over the awards’
vesting period. Compensation expense includes the impact of an estimate for forfeitures for all stock options.
The expected term of the stock options represents
the average period the stock options are expected to remain outstanding and is based on the expected term calculated using the
approach prescribed by the Securities and Exchange Commission's Staff Accounting Bulletin No. 110 for “plain
vanilla” options. The expected stock price volatility for the Company’s stock options was determined by using an average
of the historical volatilities of the Company and its industry peers. The Company will continue to analyze the stock price volatility
and expected term assumptions as more data for the Company’s common stock and exercise patterns become available. The risk-free
interest rate assumption is based on the U.S. Treasury instruments whose term was consistent with the expected term of the Company’s
stock options. The expected dividend assumption is based on the Company’s history and expectation of dividend payouts. The
Company estimates forfeitures based on historical experience and reduces compensation expense accordingly. The estimated forfeiture
rates used during the three months ended June 30, 2016 ranged from 4.65% to 4.71%. The estimated forfeiture rates used during the
three months ended June 30, 2015 ranged from 0.85% to 1.81%.
The Company estimated the fair value of employee
and non-employee stock options using the Black-Scholes option pricing model. The fair value of employee stock options is being
amortized on a straight-line basis over the requisite service periods of the respective awards. The fair value of employee stock
options was estimated using the following weighted-average assumptions:
|
|
|
Three Months Ended June 30,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
Expected Term
|
|
|
7.45 yrs
|
|
|
|
8.96 yrs
|
|
|
Risk-free interest rate
|
|
|
1.40%
|
|
|
|
2.18%
|
|
|
Dividend yield
|
|
|
0.00%
|
|
|
|
0.00%
|
|
|
Volatility
|
|
|
92.4%
|
|
|
|
86.0%
|
|
|
Fair value of options granted
|
|
$
|
3.46
|
|
|
$
|
5.50
|
|
Share-based awards compensation expense is as follows:
|
|
|
Three Months Ended June 30,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
Cost of revenues
|
|
$
|
68,000
|
|
|
$
|
55,000
|
|
|
Research and development
|
|
|
64,000
|
|
|
|
73,000
|
|
|
Selling, general and administrative
|
|
|
279,000
|
|
|
|
284,000
|
|
|
Total stock-based compensation
|
|
$
|
411,000
|
|
|
$
|
412,000
|
|
At June 30, 2016, there were unrecognized compensation
costs of $1,377,000 related to stock options which are expected to be recognized over a weighted-average amortization period of
1.30 years.
Stock-Based Award Activity
Stock-based awards outstanding at June 30,
2016 under the various plans are as follows:
|
Plan
|
|
Awards Outstanding
|
|
2006 Plan
|
|
172,000
|
|
2011 Plan
|
|
582,000
|
|
|
|
754,000
|
|
Awards available for grant as of June 30, 2016
|
|
578,000
|
Stock options award activity is as follows:
|
|
|
Number of
Shares
|
|
|
Weighted-
Average
Exercise Price
|
|
|
Weighted-
Average
Contractual Term
|
|
|
Aggregate
Intrinsic
Value
|
|
|
Outstanding at April 1, 2016
|
|
|
753,000
|
|
|
$
|
21.47
|
|
|
|
|
|
|
|
|
|
|
Options granted
|
|
|
16,000
|
|
|
|
4.65
|
|
|
|
|
|
|
|
|
|
|
Options exercised
|
|
|
–
|
|
|
|
–
|
|
|
|
|
|
|
|
|
|
|
Options forfeited
|
|
|
(1,000
|
)
|
|
|
19.14
|
|
|
|
|
|
|
|
|
|
|
Options expired
|
|
|
(14,000
|
)
|
|
|
17.81
|
|
|
|
|
|
|
|
|
|
|
Outstanding at June 30, 2016
|
|
|
754,000
|
|
|
$
|
21.18
|
|
|
|
7.69
|
|
|
$
|
–
|
|
|
Exercisable at June 30, 2016
|
|
|
504,000
|
|
|
$
|
26.81
|
|
|
|
7.15
|
|
|
$
|
–
|
|
The aggregate intrinsic value of stock options is calculated as
the difference between the exercise price of the underlying stock options and the fair value of the Company’s common stock,
or $4.01 per share at June 30, 2016.
Restricted stock award activity is as follows:
|
|
|
Number
of
Shares
|
|
|
Weighted
Average Award
Date Fair Value
per Share
|
|
|
Unvested restricted stock awards outstanding at April 1, 2016
|
|
|
–
|
|
|
$
|
–
|
|
|
Restricted stock awards granted
|
|
|
4,000
|
|
|
|
4.85
|
|
|
Restricted stock awards vested
|
|
|
(4,000
|
)
|
|
|
4.85
|
|
|
Restricted stock awards forfeited
|
|
|
–
|
|
|
|
–
|
|
|
Unvested restricted stock awards outstanding at June 30, 2016
|
|
|
–
|
|
|
$
|
–
|
|
Restricted stock awards were issued to non-employee
directors for services in the year ended June 30, 2016.
No income tax benefit has been recognized relating to stock-based
compensation expense and no tax benefits have been realized from exercised stock options.
The Company did not capitalize any cost associated with stock-based
compensation.
The Company issues new shares of common stock
upon exercise of stock based awards.
The Company has completed a study to assess
whether a change in control has occurred or whether there have been multiple changes of control since the Company’s formation.
The Company determined, based on the results of the study, no change in control occurred for purposes of Internal Revenue Code
section 382. The Company, after considering all available evidence, fully reserved its deferred tax assets since it is more likely
than not, such benefits, will not be realized in future periods. The Company incurred losses for both financial reporting and income
tax purposes for the year ended March 31, 2016. Accordingly, the Company is continuing to fully reserve for its deferred tax assets.
The Company will continue to evaluate its deferred tax assets to determine whether any changes in circumstances could affect the
realization of their future benefit. If it is determined in future periods that portions of the Company’s deferred income
tax assets satisfy the realization standards, the valuation allowance will be reduced accordingly.
As a result of certain realization requirements
of Accounting Standards Codification Topic 718, the Company’s deferred tax assets and liabilities do not include certain
deferred tax assets at June 30, 2016 that arose directly from tax deductions related to equity compensation in excess of compensation
recognized for financial reporting purposes. Equity will be increased by approximately $533,000 if and when such deferred tax assets
are ultimately realized.
|
Note 9.
|
Segment and Geographic Information
|
The Company generates product revenues from
wound care products that are sold into the human and animal healthcare markets, and the Company generates service revenues from
laboratory testing services, which are provided to medical device manufacturers.
The Company operates a single segment business
for product revenues, which consists of three geographical sales territories as follows:
|
|
|
Three Months Ended June 30,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
United States
|
|
$
|
1,373,000
|
|
|
$
|
787,000
|
|
|
Latin America
|
|
|
1,098,000
|
|
|
|
1,558,000
|
|
|
Europe and Rest of the World
|
|
|
1,038,000
|
|
|
|
571,000
|
|
|
|
|
|
3,509,000
|
|
|
|
2,916,000
|
|
|
Product license fees and royalties
|
|
|
75,000
|
|
|
|
447,000
|
|
|
Total
|
|
$
|
3,584,000
|
|
|
$
|
3,363,000
|
|
The following table shows the Company’s product license fees
and royalties revenues by partner:
|
|
|
Three Months Ended June 30,
|
|
|
Product license fees and royalties
|
|
2016
|
|
|
2015
|
|
|
Exeltis
|
|
$
|
–
|
|
|
$
|
54,000
|
|
|
Innovacyn
|
|
|
–
|
|
|
|
20,000
|
|
|
Laboratorios Sanfer, S.A. de C.V.
|
|
|
75,000
|
|
|
|
373,000
|
|
|
Total product license fees and royalties
|
|
$
|
75,000
|
|
|
$
|
447,000
|
|
The Company’s service revenues amounted
to $227,000 and $317,000 for the three months ended June 30, 2016 and 2015, respectively.
|
Note 10.
|
Significant Customer Concentrations
|
For the three months ended June 30, 2016, one
customer represented 29% of net revenue. For the three months ended June 30, 2015, one customer represented 53% of net revenue.
At June 30, 2016, one customer represented
41%, and three customers each represented 11% of the net accounts receivable balance. At March 31, 2016, one customer represented
33% of the net accounts receivable balance.
|
Note 11.
|
Subsequent Events
|
On July 26,
2016, the Company entered into a new employment agreement with Jim Schutz, its President and Chief Executive Officer to update his
agreements and responsibilities. The terms of the new employment agreement provide for a continued annual base salary of
$250,000 or such other amount as the Board of Directors may set. In addition, Mr. Schutz is eligible to receive an annual
bonus, the payment, type and amount of which is in the sole discretion of the Compensation Committee. Mr. Schutz also
receives certain benefits, such as participation in our health and welfare plans, vacation and reimbursement of expenses.
1,250,000 Shares
of Common Stock and
Warrants to Purchase
937,500 Shares of Common Stock

OCULUS INNOVATIVE
SCIENCES, INC.
_______________________
PROSPECTUS
_______________________
S
ole Book-Running Manager
Maxim Group LLC
Co-Manager
Dawson James Securities, Inc.
Sonoma Pharmaceuticals (NASDAQ:SNOA)
Historical Stock Chart
From Aug 2024 to Sep 2024
Sonoma Pharmaceuticals (NASDAQ:SNOA)
Historical Stock Chart
From Sep 2023 to Sep 2024