Prospectus
Filed Pursuant to Rule 424(b)(3)
File No. 333-211862
PROSPECTUS
OPTIMIZERX
CORP.
982,472
Shares of Common Stock
This
prospectus relates to the resale by the Selling Shareholders of up to 982,472 shares of our Common Stock, par value $0.001 per
share (the “Common Stock”). Of the Common Stock offered herein, 804,139 shares are issuable upon the exercise of warrants
held by placement agents that were issued in connection with our March 2014 Private Placement (described below), 45,000 shares
were issued to Merriman Capital, Inc. under an Advisory Agreement dated February 23, 2015 and 133,333 shares were issued to Mr.
Shadron Stastney under a Separation Agreement dated September 20, 2013.
The Selling Shareholders may offer
and sell or otherwise dispose of the shares described in this prospectus from time to time through public or private transaction
at prevailing market prices, at prices related to such prevailing market prices, at varying prices determined at the time of sale,
at negotiated prices, or at fixed prices. See “Plan of Distribution” beginning on page 11 for more information.
We
will not receive any of the proceeds from the Common Stock sold by the Selling Shareholders, unless the warrants described herein
are issued for cash and not on a cashless basis.
We
have agreed to pay certain expenses in connection with this registration statement. The Selling Shareholders will pay all underwriting
discounts and selling commissions, if any, in connection with the sale of the shares of Common Stock.
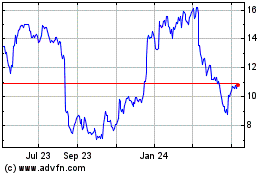
Our Common Stock is traded on the OTCQB
under the symbol “OPRX.” On June 13, 2016, the last reported sale price of our Common Stock was $1.05 per share.
Investing in our common stock involves
risks. See "Risk Factors" beginning on page 4.
Neither
the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or
determined if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
The date of this prospectus June
17, 2016
Table
of Contents
ABOUT
THIS PROSPECTUS
This
prospectus is part of a registration statement we filed with the Securities and Exchange Commission, or the SEC. Under this registration
process, the selling shareholders may, from time to time, offer and sell up to 982,472 shares of our common stock, as described
in this prospectus, in one or more offerings. This prospectus provides you with a general description of the securities the selling
shareholders may offer. You should read this prospectus carefully before making an investment decision.
You
may only rely on the information contained in this prospectus or that we have referred you to. We have not authorized anyone to
provide you with additional or different information. This prospectus does not constitute an offer to sell or a solicitation of
an offer to buy any securities other than the shares of our common stock offered by this prospectus. This prospectus does not
constitute an offer to sell or a solicitation of an offer to buy any common stock in any circumstances or any jurisdiction in
which such offer or solicitation is not permitted. You should not assume that the information contained in this prospectus is
accurate as of any date other than the date on the front cover of this prospectus regardless of the time of delivery of this prospectus
or any sale of our common stock. The rules of the SEC may require us to update this prospectus in the future.
As
used in this prospectus, unless the context requires otherwise, the terms “we”, “us”, “our”,
or “the Company” refer to OptimizeRx Corp. and its subsidiaries on a consolidated basis. References to “Selling
Shareholders” refer to those shareholders listed herein under “Selling Shareholders” and their successors, assignees
and permitted transferees.
ABOUT
FORWARD-LOOKING STATEMENTS
This
prospectus contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended (the
“Securities Act”), and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”),
about the Company and its subsidiaries. These forward-looking statements are intended to be covered by the safe harbor for forward-looking
statements provided by the Private Securities Litigation Reform Act of 1995. Forward-looking statements are not statements of
historical fact, and can be identified by the use of forward-looking terminology such as “believes”, “expects”,
“may”, “will”, “could”, “should”, “projects”, “plans”,
“goal”, “targets”, “potential”, “estimates”, “pro forma”, “seeks”,
“intends”, or “anticipates” or the negative thereof or comparable terminology. Forward-looking statements
include discussions of strategy, financial projections, guidance and estimates (including their underlying assumptions), statements
regarding plans, objectives, expectations or consequences of various transactions, and statements about the future performance,
operations, products and services of the Company and its subsidiaries. We caution our shareholders and other readers not to place
undue reliance on such statements.
Our businesses and operations are and
will be subject to a variety of risks, uncertainties and other factors. Consequently, actual results and experience may materially
differ from those contained in any forward-looking statements. Such risks, uncertainties and other factors that could cause actual
results and experience to differ from those projected include, but are not limited to, the risk factors set forth in the section
entitled “Risk Factors” beginning on page 4 of this prospectus.
All
written or oral forward-looking statements attributable to us or any person acting on our behalf made after the date of this prospectus
are expressly qualified in their entirety by the risk factors and cautionary statements contained in and incorporated by reference
into this prospectus. Unless legally required, we do not undertake any obligation to release publicly any revisions to such forward-looking
statements to reflect events or circumstances after the date of this prospectus or to reflect the occurrence of unanticipated
events.
SUMMARY
The
following summary highlights selected information contained elsewhere in this prospectus and in the documents incorporated by
reference in this prospectus and does not contain all the information you will need in making your investment decision. You should
read carefully this entire prospectus and the documents incorporated by reference in this prospectus before making an investment
decision, especially the information presented under the heading “Risk Factors.”
Business
Summary
We
provide unique consumer and physician platforms and strategies to help patients better afford and comply with their medicines
and healthcare products, while offering pharmaceutical and healthcare companies more effective ways to expand physician and patient
awareness, access and adherence to their medications and available product support.
Our
core product offering is our SampleMD™ software application which delivers an automated system that replaces traditional
physical drug samples by enabling doctors or other healthcare providers to automatically print and electronically distribute sample
vouchers or co-pay discount coupons within their ePrescribing/ EHR (electronic health record) system or from their computer desktops.
Through SampleMD, pharmaceutical and biotech manufacturers have a digital paperless platform to directly offer patients critical
cost savings, patient education and adherence tools-- right at point of prescribing.
The
Shares Underlying Warrants Issued in Connection With the March 2014 Private Placement Financing
On
March 17, 2014 (the “March 2014 Private Placement”), we entered into a Stock Purchase Agreement (the “Purchase
Agreement”) with certain accredited investors (the “Purchasers”) pursuant to which we: (i) sold to the Purchasers
an aggregate of 8,333,333 shares of our Common Stock at a price per share of $1.20 (the “Common Shares”) for aggregate
gross proceeds of $10 million and (ii) issued warrants to purchase up to 804,139 shares of the our Common Stock to our placement
agents (the “Agent Warrants”). Placement Agents also received commissions equal to approximately 9.7% of the gross
proceeds, for an aggregate commission of approximately $970,000, including reimbursements for their reasonable out of pocket expenses.
We
filed a Form S-1 registration statement on April 11, 2014 to register the Common Shares and Agent Warrants. That registration
statement went effective on May 28, 2015. We are filing this Form S-1 registration statement to maintain the registration of the
shares of common stock underlying the Agent Warrants. The Common Shares are not a part of this registration statement.
The
Shares Issued in Connection with the Merriman Capital, Inc. Advisory Agreement
On
February 23, 2015, we entered into an advisory agreement (the “Advisory Agreement”) with Merriman Capital, Inc. (“Merriman”).
In exchange for advisory services pertaining to capital market interactions, we agreed to issue to Merriman 45,000 shares of common
stock upon execution of the Advisory Agreement, with 45,000 additional shares of common stock due on or before August 15, 2015
and 60,000 shares of common stock as a bonus if we successfully up-list to a national exchange.
In
August 2015, we and Merriman agreed to terminate the Advisory Agreement as it pertains to the issuance of any additional shares.
We have issued and delivered to Merriman the 45,000 initial shares, which are being registered herein as a condition of the Advisory
Agreement (the “Advisory Shares”). The sale of these shares has been completed and Merriman bears the market risk
in connection with these shares.
The
Shares Issued in Connection with the Separation Agreement with Mr. Shadron Stastney
On
September 20, 2013, we entered into a Separation Agreement with Mr. Shadron Stastney regarding the terms and conditions of his
departure from our company (the “Separation Agreement”). Pursuant to the provisions of the Separation Agreement, we
agreed to issue to Mr. Stastney 500,000 shares of our common stock.
On
May 27, 2016, we entered into a Settlement Agreement with Mr. Stastney to register the remaining 133,333 shares of his original
500,000 shares of common stock (the “Separation Shares”).
The
Offering
|
Common
stock offered by the Selling Shareholders
|
982,472
shares of common stock, consisting of Agent Warrants to purchase up to 804,139 shares of our common stock, 45,000 Advisory
Shares of our common stock and 133,333 Separation Shares of our common stock.
|
|
|
|
|
Selling
Shareholders
|
See
“
Selling Shareholders
” beginning on page 9.
|
|
|
|
|
Common
stock outstanding
|
29,077,660
common shares as of June 2, 2016.
|
|
|
|
|
Use
of proceeds
|
We
will not receive any proceeds from the sale or other disposition of the shares of common stock covered by this prospectus.
However, upon any exercise of the Agent Warrants for cash, such Selling Shareholders would pay us the exercise price of the
warrants. Cash received from exercise of Agent Warrants will be used for general corporate purposes. Additionally, the Warrants
are exercisable on a cashless basis. If any Agent Warrants are exercised on a cashless basis, we would not receive any cash
payment from such Selling Shareholders upon any exercise of such Agent Warrants. See “Use of Proceeds” on page
9.
|
|
|
|
|
OTCQB
Symbol
|
Our
Common Stock is quoted on the OTCQB under the ticker symbol “OPRX”.
|
|
|
|
|
Risk
Factors
|
You
should consider the matters set forth under “
Risk Factors
” beginning on page 4, as well as other cautionary
statements throughout or incorporated by reference in this prospectus, before deciding to invest in shares of our common stock.
|
Summary
Financial Information
|
Balance
Sheet Data
|
|
December 31,
2015
|
|
|
December 31,
2014
|
|
|
March
31, 2016
|
|
|
Cash
|
|
$
|
8,207,565
|
|
|
$
|
3,446,973
|
|
|
$
|
7,548,261
|
|
|
Total
Assets
|
|
$
|
12,314,280
|
|
|
$
|
7,028,806
|
|
|
$
|
11,097,757
|
|
|
Liabilities
|
|
$
|
3,371,784
|
|
|
$
|
2,418,722
|
|
|
$
|
2,551,208
|
|
|
Total
Stockholders’ Equity
|
|
$
|
8,942,496
|
|
|
$
|
4,610,084
|
|
|
$
|
8,546,549
|
|
|
Statement
of Operations
|
|
Year
Ended December 31, 2015
|
|
|
Year
Ended December 31, 2014
|
|
|
Three
Months Ended
March 31,
2016
|
|
|
Three
Months Ended
March 31,
2015
|
|
|
Revenue
|
|
$
|
7,220,678
|
|
|
$
|
6,502,962
|
|
|
$
|
1,759,528
|
|
|
$
|
1,487,533
|
|
|
Income
(Loss) for the Period
|
|
$
|
(595,140
|
)
|
|
$
|
(1,025,393
|
)
|
|
$
|
(351,916
|
)
|
|
$
|
(111,201
|
)
|
RISK
FACTORS
Readers
and prospective investors in our common stock should carefully consider the following risk factors as well as the other information
contained or incorporated by reference in this prospectus.
If
any of the following risks actually occurs, our financial condition, results of operations and liquidity could be materially adversely
affected. If this were to happen, the value of our common stock could decline, and if you invest in our common stock, you could
lose all or part of your investment.
The
discussion below highlights some important risks we have identified related to our business and operations and an investment in
shares of our common stock, but these should not be assumed to be the only factors that could affect our future performance and
condition, financial and otherwise. We do not have a policy of updating or revising forward-looking statements except as otherwise
required by law, and silence by management over time should not be construed to mean that actual events are occurring as estimated
in such forward-looking statements.
Risks
Relating to Business and Financial Condition
Because
we have historically experienced losses, if we are unable to achieve profitability, our financial condition and company could
suffer.
Since
the inception of our business we have historically incurred losses. While we have increased revenues significantly, we have not
yet been able to achieve profitability due to significant investments in our growth. Our ability to achieve consistent profitability
depends on our ability to generate sales through our technology platform and advertising model, while maintaining reasonable expense
levels. If we do not achieve sustainable profitability, it may impact our ability to continue our operations.
Our
business and growth may suffer if we are unable to attract and retain key employees.
Our
success depends on the expertise of our executive officers and certain other key technical personnel. It may be difficult to find
sufficiently qualified individuals to replace management or other key technical personnel in the event of death, disability or
resignation, thus frustrating our ability to implement our business plan, which could negatively affect our operating results.
Furthermore,
our ability to expand operations to accommodate our anticipated growth will also depend on our ability to attract and retain qualified
media, management, finance, marketing, sales and technical personnel. However, competition for these types of employees is intense
due to the limited number of qualified professionals. Our ability to meet our business development objectives will depend in part
on our ability to recruit, train and retain top quality people with advanced skills who understand our technology and business.
No assurance can be given that we will be successful in this regard. If we are unable to engage and retain the necessary personnel,
our business may be materially and adversely affected.
Our
failure to obtain retain or attract additional customers could prevent us from successfully executing our business plan.
We
currently work with many leading pharmaceutical companies, including Pfizer, Eli Lilly, Actavis, AstraZeneca, Alcon, Daiichi Sankyo,
Novartis, Novo Nordisk, Valeant, Shire, and others. Our failure to retain existing customers or expand with new customers could
negatively impact our business.
We
are dependent on a concentrated group of customers
Our
revenues are concentrated in approximately 25 customers, primarily large pharmaceutical manufacturers and large advertising agencies.
Approximately 52% of our revenue came from our largest five customers. Loss of one or more of these customers could have a significant
negative impact on our operating results.
We
may be unable to support our technology to further scale our operations successfully.
Our
plan is to grow rapidly through further integration of our technology in electronic platforms. Our growth will place significant
demands on our management and technology development, as well as our financial, administrative and other resources. We cannot
guarantee that any of the systems, procedures and controls we put in place will be adequate to support the commercialization of
our operations. Our operating results will depend substantially on the ability of our officers and key employees to manage changing
business conditions and to implement and improve our financial, administrative and other resources. If we are unable to respond
to and manage changing business conditions, or the scale of our products, services and operations, then the quality of our services,
our ability to retain key personnel and our business could be harmed.
If
we are unable to maintain our contracts with electronic prescription platforms, our business will suffer.
We
are reliant upon our contracts with leading electronic prescribing platforms, including Allscripts, Dr. First, Quest Diagnostics,
and others. We will need to maintain these relationships as well as diversify them. The inability to do so could adversely impact
our business.
Our
agreements with electronic prescription platforms are subject to audit, which could subject us to additional costs that might
affect our results of operations.
Our
agreements with our electronic prescription platform partners provide for revenue sharing payments to the platform partners based
on the revenue we generate through the platform. These payments are subject to audit by our partners, at their cost, and if there
is a dispute as to the calculation, we may be liable for additional payments. If an underpayment is determined to be in excess
of a certain amount, for example 10%, some agreements would require us to pay for the cost of the audit, as well.
Developing
and implementing new and updated applications, features and services for our portals may be more difficult than expected, may
take longer and cost more than expected and may not result in sufficient increases in revenue to justify the costs.
We
have completed the development and migration of SampleMD 2.0’s on-demand, rule based content delivery platform. The system
can now manage up to 1 million rules and return the appropriate content within 1 second. This allows unsurpassed response time
to avoid delays, and the ability to meet the upcoming dramatic scale we expect. Despite the launch of Sample MD 2.0, attracting
and retaining users of our portals requires us to continue to improve the technology underlying those portals and to continue
to develop new and updated applications, features and services for those portals. If we are unable to do so on a timely basis
or if we are unable to implement new applications, features and services without disruption to our existing ones, we may lose
potential users and clients. The costs of development of these enhancements may negatively impact our ability to achieve profitability.
We
rely on a combination of internal development, strategic relationships, licensing and acquisitions to develop our portals and
related applications, features and services. Our development and/or implementation of new technologies, applications, features
and services may cost more than expected, may take longer than originally expected, may require more testing than originally anticipated
and may require the acquisition of additional personnel and other resources. There can be no assurance that the revenue opportunities
from any new or updated technologies, applications, features or services will justify the amounts spent.
If
we are unable to adhere to the regulatory and competitive climate in which we operate, we could be materially and negatively impacted.
Do
to the labyrinth of regulations in the healthcare space, state and federal, as well as political sensitivity of healthcare delivery,
our business model could be negatively impacted or fail.
The
markets in which we operate are competitive, continually evolving and, in some cases, subject to rapid change.
|
|
●
|
Our
portals face competition from numerous other companies, both in attracting users and
in generating revenue from advertisers and sponsors. We compete for users with online
services and websites that provide savings on medications and healthcare products, including
both commercial sites and not-for-profit sites. We compete for advertisers and sponsors
with: health-related web sites; general purpose consumer web sites that offer specialized
health sub-channels; other high-traffic web sites that include both healthcare-related
and non-healthcare-related content and services; search engines that provide specialized
health search; and advertising networks that aggregate traffic from multiple sites.
|
|
|
●
|
Our
healthcare provider portals compete with: providers of healthcare decision-support tools
and online health management applications; wellness and disease management vendors; and
health information services and health management offerings of healthcare benefits companies
and their affiliates.
|
Many
of our competitors have greater financial, technical, product development, marketing and other resources than we do. These organizations
may be better known than we are and have more customers or users than we do. We cannot provide assurance that we will be able
to compete successfully against these organizations or any alliances they have formed or may form. Since there are no substantial
barriers to entry into the markets in which our public portals participate, we expect that competitors will continue to enter
these markets.
Developments
in the healthcare industry could adversely affect our business
Most
of our revenue is derived from the healthcare industry and could be affected by changes affecting healthcare spending. We are
particularly dependent on pharmaceutical, biotechnology and medical device companies for our advertising and sponsorship revenue.
General
reductions in expenditures by healthcare industry participants could result from, among other things:
|
|
●
|
government
regulation or private initiatives that affect the manner in which healthcare providers
interact with patients, payers or other healthcare industry participants, including changes
in pricing or means of delivery of healthcare products and services;
|
|
|
●
|
government
regulation prohibiting the use of coupons by patients covered by federally funded health
insurance programs;
|
|
|
●
|
consolidation
of healthcare industry participants;
|
|
|
●
|
reductions
in governmental funding for healthcare; and
|
|
|
●
|
adverse
changes in business or economic conditions affecting healthcare payers or providers,
pharmaceutical, biotechnology or medical device companies or other healthcare industry
participants.
|
Even
if general expenditures by industry participants remain the same or increase, developments in the healthcare industry may result
in reduced spending in some or all of the specific market segments that we serve or are planning to serve. For example, use of
our products and services could be affected by:
|
|
●
|
changes
in the design of health insurance plans;
|
|
|
●
|
a
decrease in the number of new drugs or medical devices coming to market;
|
|
|
●
|
a
decrease in marketing expenditures by pharmaceutical or medical device companies, including
as a result of governmental regulation or private initiatives that discourage or prohibit
advertising or sponsorship activities by pharmaceutical or medical device companies;
and
|
|
|
●
|
payor
pressure to move to generic brands.
|
In
addition, our customers’ expectations regarding pending or potential industry developments may also affect their budgeting
processes and spending plans with respect to products and services of the types we provide.
The
healthcare industry has changed significantly in recent years and we expect that significant changes will continue to occur. However,
the timing and impact of developments in the healthcare industry are difficult to predict. We cannot assure you that the markets
for our products and services will continue to exist at current levels or that we will have adequate technical, financial and
marketing resources to react to changes in those markets.
Because
we are embroiled in various lawsuits from time to time with uncertain consequences, the outcome of potential judgments may negatively
affect our financial condition and results of operations.
We
are currently involved in litigation and other disputes, as described in this Prospectus. As we continue to grow, we can expect
to have to deal with lawsuits that affect our business. Lawsuits are uncertain and involve a substantial degree of risk. If we
are unable to successfully prosecute or defend these actions, our financial condition and results of operations could suffer.
Our
success is dependent in part on obtaining, maintaining and enforcing our proprietary rights and our ability to avoid infringing
on the proprietary rights of others.
We
seek patent protection for those inventions and technologies for which we believe such protection is suitable and is likely to
provide a competitive advantage to us. Because patent applications in the United States are maintained in secrecy until either
the patent application is published or a patent is issued, we may not be aware of third-party patents, patent applications and
other intellectual property relevant to our products that may block our use of our intellectual property or may be used in third-party
products that compete with our products and processes. In the event a competitor or other party successfully challenges our products,
processes, patents or licenses or claims that we have infringed upon their intellectual property, we could incur substantial litigation
costs defending against such claims, be required to pay royalties, license fees or other damages or be barred from using the intellectual
property at issue, any of which could have a material adverse effect on our business, operating results and financial condition.
We
also rely substantially on trade secrets, proprietary technology, nondisclosure and other contractual agreements, and technical
measures to protect our technology, application, design, and manufacturing know-how, and work actively to foster continuing technological
innovation to maintain and protect our competitive position. We cannot assure you that steps taken by us to protect our intellectual
property and other contractual agreements for our business will be adequate, that our competitors will not independently develop
or patent substantially equivalent or superior technologies or be able to design around patents that we may receive, or that our
intellectual property will not be misappropriated.
Our
business will suffer if our network systems fail or become unavailable.
A
reduction in the performance, reliability and availability of our network infrastructure would harm our ability to distribute
our products to our users, as well as our reputation and ability to attract and retain customers. Our systems and operations could
be damaged or interrupted by fire, flood, power loss, telecommunications failure, Internet breakdown, earthquake and similar events.
Our systems could also be subject to viruses, break-ins, sabotage, acts of terrorism, acts of vandalism, hacking, cyber-terrorism
and similar misconduct. We might not carry adequate business interruption insurance to compensate us for losses that may occur
from a system outage. Any system error or failure that causes interruption in availability of our product or an increase in response
time could result in a loss of potential customers, which could have a material adverse effect on our business, financial condition
and results of operations. If we suffer sustained or repeated interruptions, then our products and services could be less attractive
to our users and our business would be materially harmed.
If
we are unable to manage growth, our operations could be adversely affected.
Our
progress is expected to require the full utilization of our management, financial and other resources. Our ability to manage growth
effectively will depend on our ability to improve and expand operations, including our financial and management information systems,
and to recruit, train and manage personnel. There can be no absolute assurance that management will be able to manage growth effectively.
If
we do not properly manage the growth of our business, we may experience significant strains on our management and operations and
disruptions in our business. Various risks arise when companies and industries grow quickly. If our business or industry grows
too quickly, our ability to meet customer demand in a timely and efficient manner could be challenged. We may also experience
development delays as we seek to meet increased demand for our products. Our failure to properly manage the growth that we or
our industry might experience could negatively impact our ability to execute on our operating plan and, accordingly, could have
an adverse impact on our business, our cash flow and results of operations, and our reputation with our current or potential customers.
Our
business is subject to changing regulation of corporate governance and public disclosure.
Because
our common stock is publicly traded, we are subject to certain rules and regulations of federal and state entities charged with
the protection of investors and the oversight of companies whose securities are publicly traded. These entities have continued
to develop additional regulations and requirements in response to laws enacted by Congress, most notably the Sarbanes-Oxley Act
of 2002. Complying with these new regulations has resulted in, and is likely to continue to result in, increased general and administrative
costs and a diversion of management time and attention from revenue generating and other business activities to compliance activities.
Risks
Relating to Our Securities
If
a market for our common stock does not develop, shareholders may be unable to sell their shares.
Our
common stock is quoted under the symbol “OPRX” on the OTCQB operated by OTC Markets Group, Inc., an electronic inter-dealer
quotation medium for equity securities. We do not currently have an active trading market. There can be no assurance that an active
and liquid trading market will develop or, if developed, that it will be sustained.
Our
securities are very thinly traded. Accordingly, it may be difficult to sell shares of our common stock without significantly depressing
the value of the stock. Unless we are successful in developing continued investor interest in our stock, sales of our stock could
continue to result in major fluctuations in the price of the stock.
Because
we are subject to the “Penny Stock” rules, the level of trading activity in our stock may be reduced.
The
Securities and Exchange Commission has adopted regulations which generally define "penny stock" to be any listed, trading
equity security that has a market price less than $5.00 per share or an exercise price of less than $5.00 per share, subject to
certain exemptions. The penny stock rules require a broker-dealer, prior to a transaction in a penny stock not otherwise exempt
from the rules, to deliver a standardized risk disclosure document that provides information about penny stocks and the risks
in the penny stock market. The broker-dealer must also provide the customer with current bid and offer quotations for the penny
stock, the compensation of the broker-dealer and its salesperson in the transaction, and monthly account statements showing the
market value of each penny stock held in the customer’s account. In addition, the penny stock rules generally require that
prior to a transaction in a penny stock, the broker-dealer make a special written determination that the penny stock is a suitable
investment for the purchaser and receive the purchaser’s written agreement to the transaction. These disclosure requirements
may have the effect of reducing the level of trading activity in the secondary market for a stock that becomes subject to the
penny stock rules which may increase the difficulty Purchasers may experience in attempting to liquidate such securities.
We
do not expect to pay dividends in the foreseeable future. Any return on investment may be limited to the value of our common stock.
We
do not anticipate paying cash dividends on our common stock in the foreseeable future. The payment of dividends on our common
stock will depend on earnings, financial condition and other business and economic factors affecting it at such time as the board
of directors may consider relevant. If we do not pay dividends, our common stock may be less valuable because a return on your
investment will occur only if our stock price appreciates.
Provisions
in the Nevada Revised Statutes and our Bylaws could make it very difficult for an investor to bring any legal actions against
our directors or officers for violations of their fiduciary duties or could require us to pay any amounts incurred by our directors
or officers in any such actions.
Members
of our board of directors and our officers will have no liability for breaches of their fiduciary duty of care as a director or
officer, except in limited circumstances, pursuant to provisions in the Nevada Revised Statutes and our Bylaws as authorized by
the Nevada Revised Statutes. Specifically, Section 78.138 of the Nevada Revised Statutes provides that a director or officer is
not individually liable to the company or its shareholders or creditors for any damages as a result of any act or failure to act
in his or her capacity as a director or officer unless it is proven that (1) the director’s or officer’s act or failure
to act constituted a breach of his or her fiduciary duties as a director or officer and (2) his or her breach of those duties
involved intentional misconduct, fraud or a knowing violation of law. This provision is intended to afford directors and officers
protection against and to limit their potential liability for monetary damages resulting from suits alleging a breach of the duty
of care by a director or officer. Accordingly, you may be unable to prevail in a legal action against our directors or officers
even if they have breached their fiduciary duty of care. In addition, our Bylaws allow us to indemnify our directors and officers
from and against any and all costs, charges and expenses resulting from their acting in such capacities with us. This means that
if you were able to enforce an action against our directors or officers, in all likelihood, we would be required to pay any expenses
they incurred in defending the lawsuit and any judgment or settlement they otherwise would be required to pay. Accordingly, our
indemnification obligations could divert needed financial resources and may adversely affect our business, financial condition,
results of operations and cash flows, and adversely affect prevailing market prices for our common stock.
USE
OF PROCEEDS
All
of the shares of common stock covered by this prospectus are being sold by the Selling Shareholders. See “Selling Shareholders”
below. We will not receive any proceeds from these sales of shares of our common stock. A portion of the shares covered by this
prospectus are issuable upon exercise of the Agent Warrants to purchase our common stock. Upon any exercise of the Agent Warrants
for cash, such Selling Shareholders would pay us the exercise price of the warrants. Cash received from exercise of Agent Warrants
will be used for general corporate purposes. Additionally, the Warrants are exercisable on a cashless basis. If any Agent Warrants
are exercised on a cashless basis, we would not receive any cash payment from such Selling Shareholders upon any exercise of such
Agent Warrants.
The
Selling Shareholders will pay any underwriting discounts and commissions and expenses incurred by the Selling Shareholders for
brokerage, accounting, tax, or legal services or any other expenses incurred by the Selling Shareholders in disposing of the shares.
We will bear all other costs, fees, and expenses incurred in effecting the registration of the shares covered by this prospectus,
including, without limitation, all registration and filing fees, and fees and expenses of our counsel and our accountants.
DETERMINATION
OF OFFERING PRICE
The
Selling Shareholders will determine at what price they may sell the offered shares, and such sales may be made at prevailing market
prices, or at privately negotiated prices.
SELLING
SHAREHOLDERS
We
have prepared this prospectus to allow the Selling Shareholders or their successors, assignees or other permitted transferees
to sell or otherwise dispose of, from time to time, up to 982,472 shares of our Common Stock. Of the shares of common stock being
offered under this prospectus 804,139 shares are able to be acquired pursuant to the Agent Warrants we issued in connection with
our March 2014 Private Placement, which Agent Warrants are currently exercisable. There are also 45,000 Advisory Shares that were
issued in connection with an Advisory Agreement with Merriman. We have issued and delivered to Merriman the 45,000 shares of common
stock that were earned under the Advisory Agreement as of the date of execution. The remaining 133,333 Separation Shares were
issued in connection with a Separation Agreement with Mr. Shadron Stastney. We have issued and delivered to Mr. Stastney these
shares of common stock.
The
Agent Warrants, Advisory Shares and Separation Shares were sold pursuant to an exemption from registration provided by Section
4(a)(2) of the Securities Act and/or Regulation D under the Securities Act. In connection therewith, the investors made to us
certain representations, warranties, covenants, and conditions customary for private placement investments.
The
table below presents information regarding the Selling Shareholders and the shares of our Common Stock that they may sell or otherwise
dispose of from time to time under this prospectus. Percentages of beneficial ownership are based upon 29,077,660 shares of Common
Stock issued and outstanding as of June 2, 2016. Beneficial ownership is determined under Section 13(d) of the Exchange Act and
generally includes voting or investment power with respect to securities and including any securities that grant the Selling Shareholders
the right to acquire Common Stock within 60 days of June 2, 2016. Unless otherwise noted, each person or group identified possesses
sole voting and investment power with respect to the shares, subject to community property laws where applicable.
We
do not know when or in what amounts the Selling Shareholders may sell or otherwise dispose of the shares covered hereby. We currently
have no agreements, arrangements or understandings with the Selling Shareholders regarding the sale of any of the shares by them
other than the registration rights agreement described below. The Selling Shareholders might not sell any or all of the shares
covered by this prospectus or may sell or dispose of some or all of the shares other than pursuant to this prospectus. Because
the Selling Shareholders may not sell or otherwise dispose of some or all of the shares covered by this prospectus and because
there are currently no agreements, arrangements or understandings with respect to the sale or other disposition of any of the
shares, we cannot estimate the number of the shares that will be held by the Selling Shareholders after completion of the offering.
Each
Selling Shareholder has indicated to us that neither it nor any of its affiliates has held any position or office or had any other
material relationship with us in the past three years except as described in the footnotes to the table.
The
shares of common stock being offered under this prospectus may be offered for sale from time to time during the period the registration
statement of which this prospectus is a part remains effective, by or for the accounts of the Selling Shareholders named below.
|
|
|
Shares
of Common Stock
|
|
|
Name
of Selling Shareholder
|
|
Beneficially
Owned Prior
to the Sale
of all Shares
covered by
this
Prospectus
|
|
|
Covered by
this
Prospectus
|
|
|
Beneficially
Owned After
the Sale
of all Shares
covered by
this
Prospectus
|
|
|
As
a
Percent of
Total
Outstanding
After the
Sale of
Shares covered by
this
Prospectus
|
|
|
Merriman Capital, Inc.(1)
|
|
|
45,000
|
|
|
|
45,000
|
|
|
|
0
|
|
|
|
0
|
|
|
Merriman Capital, Inc.(2)
|
|
|
591,613
|
|
|
|
591,613
|
|
|
|
0
|
|
|
|
0
|
|
|
Taglich Brothers, Inc.(3)
|
|
|
212,526
|
|
|
|
212,526
|
|
|
|
0
|
|
|
|
0
|
|
|
Shadron Stastney
|
|
|
133,333
|
|
|
|
133,333
|
|
|
|
0
|
|
|
|
0
|
|
|
(1)
|
Consists
of 45,000 shares of common stock issued under an Advisory Agreement with Merriman Capital,
Inc. Merriman is a registered broker-dealer that served as the placement agent in connection
with our March 2014 Private Placement. Jon Merriman is the CEO of Merriman and an affiliate
of a registered-broker-dealer. Mr. Merriman has voting and dispositive power with respect
to the shares.
|
|
(2)
|
Consists
of warrants received as our placement agent in our March 2014 Private Placement.
|
|
(3)
|
Consists
of warrants received as our placement agent in our March 2014 Private Placement. Taglich
Brothers, Inc. is a registered broker-dealer that served as the placement agent in connection
with our March 2014 Private Placement. Michael N. Taglich is a stockholder and executive
officer of Taglich Brothers, Inc. and an affiliate of a registered-broker-dealer. Mr.
Taglich has voting and dispositive power with respect to the warrants.
|
As
of the time of the purchase of the securities to be resold under this Prospectus, the Selling Shareholders either received their
securities as compensation in connection with investment banking services provided in connection with the March 2014 Private Placement,
or purchased the securities in the ordinary course of business and represented to us that they did not have any agreements or
understandings, directly or indirectly, with any person to distribute the securities.
The
Selling Shareholders, or their partners, pledgees, donees, transferees or other successors that receive the shares and their corresponding
registration in accordance with the registration rights agreement to which the Selling Shareholder is party (each also a selling
shareholder for purposes of this prospectus), may sell up to all of the shares of our common stock shown in the table above pursuant
to this Prospectus in one or more transactions from time to time as described below under “Plan of Distribution.”
However, the Selling Shareholders are not obligated to sell any of the shares of our common stock offered by this prospectus.
Information
about the Selling Shareholders may change from time to time. Any changed information with respect to which we are given notice
will be included in prospectus supplements.
PLAN
OF DISTRIBUTION
We
are registering the shares of Common Stock to permit the resale of these shares of Common Stock by the Selling Shareholders and
any of their transferees, pledgees, assignees, donees, and successors-in-interest from time to time after the date of this prospectus.
We will not receive any of the proceeds from the sale by the Selling Shareholders of the shares of Common Stock. Upon any exercise
of the Agent Warrants by payment of cash, however, we will receive the exercise price of the Agent Warrants. We will bear all
fees and expenses incident to our obligation to register the shares of Common Stock.
Each
Selling Shareholder (the “Selling Shareholders”) of the securities and any of their pledgees, assignees and successors-in-interest
may, from time to time, sell any or all of their securities covered hereby on the OTC Bulletin Board, OTCQB or any other stock
exchange, market or trading facility on which the securities are traded or in private transactions. These sales may be at fixed
or negotiated prices. A Selling Shareholder may use any one or more of the following methods when selling securities:
|
|
●
|
ordinary
brokerage transactions and transactions in which the broker-dealer solicits purchasers;
|
|
|
●
|
block
trades in which the broker-dealer will attempt to sell the securities as agent but may
position and resell a portion of the block as principal to facilitate the transaction;
|
|
|
●
|
purchases
by a broker-dealer as principal and resale by the broker-dealer for its account;
|
|
|
●
|
an
exchange distribution in accordance with the rules of the applicable exchange;
|
|
|
●
|
privately
negotiated transactions;
|
|
|
●
|
settlement
of short sales entered into after the effective date of the registration statement of
which this prospectus is a part;
|
|
|
●
|
in
transactions through broker-dealers that agree with the Selling Shareholders to sell
a specified number of such securities at a stipulated price per security;
|
|
|
●
|
through
the writing or settlement of options or other hedging transactions, whether through an
options exchange or otherwise;
|
|
|
●
|
a
combination of any such methods of sale; or
|
|
|
●
|
any
other method permitted pursuant to applicable law.
|
The
Selling Shareholders may also sell securities under Rule 144 under the Securities Act of 1933, as amended (the “Securities
Act”), if available, rather than under this prospectus.
Broker-dealers
engaged by the Selling Shareholders may arrange for other brokers-dealers to participate in sales. Broker-dealers may receive
commissions or discounts from the Selling Shareholders (or, if any broker-dealer acts as agent for the purchaser of securities,
from the purchaser) in amounts to be negotiated, but, except as set forth in a supplement to this Prospectus, in the case of an
agency transaction not in excess of a customary brokerage commission in compliance with FINRA Rule 2440; and in the case of a
principal transaction a markup or markdown in compliance with FINRA IM-2440.
In
connection with the sale of the securities or interests therein, the Selling Shareholders may enter into hedging transactions
with broker-dealers or other financial institutions, which may in turn engage in short sales of the securities in the course of
hedging the positions they assume. The Selling Shareholders may also sell securities short and deliver these securities to close
out their short positions, or loan or pledge the securities to broker-dealers that in turn may sell these securities. The Selling
Shareholders may also enter into option or other transactions with broker-dealers or other financial institutions or create one
or more derivative securities which require the delivery to such broker-dealer or other financial institution of securities offered
by this prospectus, which securities such broker-dealer or other financial institution may resell pursuant to this prospectus
(as supplemented or amended to reflect such transaction).
The
Selling Shareholders and any broker-dealers or agents that are involved in selling the securities may be deemed to be “underwriters”
within the meaning of the Securities Act in connection with such sales. In such event, any commissions received by such broker-dealers
or agents and any profit on the resale of the securities purchased by them may be deemed to be underwriting commissions or discounts
under the Securities Act. Each Selling Shareholder has informed the Company that it does not have any written or oral agreement
or understanding, directly or indirectly, with any person to distribute the securities. In no event shall any broker-dealer receive
fees, commissions and markups which, in the aggregate, would exceed eight percent (8%).
The
Company is required to pay certain fees and expenses incurred by the Company incident to the registration of the securities. The
Company has agreed to indemnify the Selling Shareholders against certain losses, claims, damages and liabilities, including liabilities
under the Securities Act.
Because
Selling Shareholders may be deemed to be “underwriters” within the meaning of the Securities Act, they will be subject
to the prospectus delivery requirements of the Securities Act including Rule 172 thereunder. In addition, any securities covered
by this prospectus which qualify for sale pursuant to Rule 144 under the Securities Act may be sold under Rule 144 rather than
under this prospectus. The Selling Shareholders have advised us that there is no underwriter or coordinating broker acting in
connection with the proposed sale of the resale securities by the Selling Shareholders.
We
agreed to keep this prospectus effective until the earlier of (i) the date on which the securities may be resold by the Selling
Shareholders without registration and without regard to any volume or manner-of-sale limitations by reason of Rule 144, without
the requirement for the Company to be in compliance with the current public information under Rule 144 under the Securities Act
or any other rule of similar effect or (ii) all of the securities have been sold pursuant to this prospectus or Rule 144 under
the Securities Act or any other rule of similar effect. The resale securities will be sold only through registered or licensed
brokers or dealers if required under applicable state securities laws. In addition, in certain states, the resale securities covered
hereby may not be sold unless they have been registered or qualified for sale in the applicable state or an exemption from the
registration or qualification requirement is available and is complied with.
Under
applicable rules and regulations under the Exchange Act, any person engaged in the distribution of the resale securities may not
simultaneously engage in market making activities with respect to the common stock for the applicable restricted period, as defined
in Regulation M, prior to the commencement of the distribution. In addition, the Selling Shareholders will be subject to applicable
provisions of the Exchange Act and the rules and regulations thereunder, including Regulation M, which may limit the timing of
purchases and sales of securities of the common stock by the Selling Shareholders or any other person. We will make copies of
this prospectus available to the Selling Shareholders and have informed them of the need to deliver a copy of this prospectus
to each purchaser at or prior to the time of the sale (including by compliance with Rule 172 under the Securities Act).
LEGAL
PROCEEDINGS
In
September, 2014, we initiated litigation against Shadron Stastney, our CEO from January 2013 to September 2013, in the U.S. District
Court in the Eastern District of Michigan as a result of a dispute related to his Separation Agreement. On May 27, 2016, we settled
the action. For a complete release of claims and dismissal of the action, we agreed to pay Mr. Stastney $50,000 and to issue him
100,000 shares of our common stock. We further agreed to register 133,333 of his existing shares with the Securities and Exchange
Commission on Form S-1 by June 30, 2016.
In
March, 2015, we initiated litigation against LDM Group, LLC and PDR Network, LLC in the U.S. District Court in the Eastern District
of Missouri related to the breach by LDM, and PDR as successor, of the settlement agreement signed February 28, 2014 related to
previous litigation with LDM. LDM has failed to live up to its obligations under the settlement agreement including, but not limited
to, not allowing us to distribute our eCoupon programs in the LDM network, not allowing us to distribute the LDM patient education
programs, and not providing other information required under the settlement agreement. We are seeking enforcement of the settlement
agreement and we are seeking damages in an amount at least equal to the amounts paid to date to LDM under the settlement agreement,
which is in excess of $1.0 million, as well as damages for lost income and business value as a result of LDM’s breach of
the agreement.
In
March, 2015, we also initiated litigation against PDR Network, LLC in the U.S. District Court in the District of New Jersey as
a result of PDR’s breach of the Master Services Agreement between the parties requiring PDR to exclusively use our eCoupon
solution. We assert that PDR’s acquisition of LDM and the use of the LDM network to distribute coupons by PDR violates the
agreement between the parties and we are seeking damages in an amount at least equal the amounts paid to date by us to LDM under
the settlement agreement, which is in excess of $1.0 million, as well as damages for lost income and business value as a result
of PDR’s actions.
In
May, 2015, we filed an amended complaint in the Missouri case to consolidate the two cases and withdrew the case against PDR Networks
in the U.S. District Court in the District of New Jersey, without prejudice. In July, 2015, the U.S. District Court for the Eastern
District of Missouri dismissed the case, citing lack of Federal jurisdiction in the matter. We refiled the consolidated case against
PDR Network and LDM group in State court in Missouri. The defendants have filed a motion to dismiss two of the four counts in
the consolidated complaint. In January, 2016, the Court dismissed one of our four claims, but allowed the other three to continue
forward. The parties are currently in the discovery process.
DIRECTORS,
EXECUTIVE OFFICERS, PROMOTERS AND CONTROL PERSONS
The
following information sets forth the names, ages, and positions of our current directors and executive officers.
|
Name
|
|
|
Age
|
|
|
Positions
and Offices Held
|
|
David
A. Harrell
|
|
|
49
|
|
|
Chairman
and Director
|
|
William
J. Febbo
|
|
|
47
|
|
|
Chief
Executive Officer and Director
|
|
Terence
J. Hamilton
|
|
|
50
|
|
|
S
VP
of Sales
|
|
Douglas
Baker
|
|
|
59
|
|
|
Chief
Financial Officer
|
|
James
Brooks
|
|
|
47
|
|
|
Senior
Vice President of Business Development
|
|
Gus
D. Halas
|
|
|
65
|
|
|
Director
|
|
Jack
Pinney
|
|
|
59
|
|
|
Director
|
|
Lynn
Vos
|
|
|
60
|
|
|
Director
|
Set
forth below is a brief description of the background and business experience of each of our current executive officers and directors.
David
A. Harrell
Mr.
Harrell founded the Company in January of 2006. He became a director when the Company changed from a limited liability to a corporation
in 2007. He has served as our Chairman since September 20, 2013, and our Chief Executive Officer from September 20, 2013 through
February 21, 2016. Mr. Harrell was the Vice President of Development for Meridian Incorporated from 2003-2005 and, prior to that,
had been Vice President of Sales and Marketing since 1999 at Advance Graphic Systems. Mr. Harrell has spent two decades leading
sales, marketing and business development units within the pharmaceutical and national retail industries. Prior to his work at
Advance Graphic Systems, Mr. Harrell served for ten years at SmithKline Beecham, specializing in the managed markets healthcare
segment. As part of the Integrated Health Division, Mr. Harrell was responsible for contracting and achieving regional revenue
growth for SmithKline Beecham's four business units: Pharmaceuticals, Consumer Health, Clinical Labs and Diversified Pharmaceutical
Services (PBM). During his tenure with SmithKline Beecham, he was a recipient of numerous national awards and served as a member
of the Division's Strategic Planning Committee. Mr. Harrell graduated from Oakland University with a Bachelor of Science in Business
Administration.
Aside
from that provided above, Mr. Harrell does not hold and has not held over the past five years any other directorships in any company
with a class of securities registered pursuant to Section 12 of the Exchange Act or subject to the requirements of Section 15(d)
of the Exchange Act or any company registered as an investment company under the Investment Company Act of 1940.
Mr.
Harrell is qualified to serve on our Board of Directors because of his sales, marketing and business development experience in
the pharmaceutical sector.
William
J. Febbo
Mr.
Febbo joined the company as Chief Executive Officer on February 22, 2016. Mr. Febbo brings more than 18 years of experience in
building and managing health services and financial businesses. Before joining our company, Mr. Febbo served as Chairman and Founder
of Plexus, LLC, a payment processing business for medical professionals. From 2007 to 2015, he worked with Merriman Holdings,
Inc., an investment banking firm. There he served as Chief Operating Officer and assisted with capital raises in the tech, biotech,
cleantech, consumer and resources industries. From 2013 to 2015, he also worked with Digital Capital Network, Inc., which operated
a transaction platform for institutional and accredited investors. There he served as Chief Executive Officer and Co-Founder and
managed the day-to-day operations of the digital portal for institutional level investments. Prior to Merriman, Mr Febbo was CEO
and co-founder of MedPanel, a provider of market intelligence and communications for the pharmaceutical, biomedical, and medical
device industries, which was eventually acquired by MCF Corporation.
Aside
from that provided above, Mr. Febbo does not hold and has not held over the past five years any other directorships in any company
with a class of securities registered pursuant to Section 12 of the Exchange Act or subject to the requirements of Section 15(d)
of the Exchange Act or any company registered as an investment company under the Investment Company Act of 1940.
Mr.
Febbo is qualified to serve on our Board of Directors because of his leadership and sales experience in health services and financial
businesses.
Terence
J. Hamilton
Mr.
Hamilton joined the Company as a Director and VP of Sales in February 2008. Prior to that, Mr. Hamilton was Manager at MedImmune
since 2005 and was Senior National Account Manager for Glaxo SmithKline pharmaceuticals for 13 years prior to that. Mr. Hamilton
has spent the last 19 years working in the pharmaceutical and biotech arenas within various sales, marketing and managed markets
management positions. He also has held many positions within the pharmaceutical and biotech industries, including District Manager,
Brand Manager, Managed Market Specialist, Contract Manager, and Government Account Manager.
Aside
from that provided above, Mr. Hamilton does not hold and has not held over the past five years any other directorships in any
company with a class of securities registered pursuant to Section 12 of the Exchange Act or subject to the requirements of Section
15(d) of the Exchange Act or any company registered as an investment company under the Investment Company Act of 1940.
Douglas
Baker
Mr.
Baker has served as our CFO since May 19, 2014. Mr. Baker is a Certified Public Account with a Master’s Degree in Business
Administration. He has extensive business experience including 9 years in public accounting with Plante Moran, 4 years as CFO
of a privately held printing company, 5 years in a variety of divisional financial roles at MascoTech, Inc., a Fortune 500 automotive
supplier, and from 1996 to 2014 as Chief Financial Officer of Applied Nanotech Holdings, Inc., (“APNT”) a publicly
held nanotechnology research and licensing company. Mr. Baker was also a member of the Board of Directors of APNT from 2006 through
2014. He is also currently Chairman of the Board of Total Health Care, Inc., a Detroit based Health Maintenance Organization and
has been a member of that Board since 1987.
Aside
from that provided above, Mr. Baker does not hold and has not held over the past five years any other directorships in any company
with a class of securities registered pursuant to Section 12 of the Exchange Act or subject to the requirements of Section 15(d)
of the Exchange Act or any company registered as an investment company under the Investment Company Act of 1940.
James
Brooks
Mr.
Brooks brings more than 20 years of experience in sales & marketing, including sales force development, sales management,
and organizational leadership processes within the Electronic Health Record (EHR) and Healthcare Technology sector. Prior to joining
our company, he served as chief revenue officer at iCare, an enterprise cloud EHR and revenue cycle management system vendor.
Prior to iCare, Mr. Brooks served as executive vice president at Modernizing Medicine, a leading provider of EHR systems focused
on the dermatology, ophthalmology, plastic, and orthopedic marketplace. While at Modernizing Medicine, he was responsible for
developing and managing a world-class sales and marketing team and grew the business into one of the largest specialty EHRs. Mr.
Brooks additionally served as vice president of the enterprise sales and radiology division at Sage North America, a provider
of information systems with a focus on healthcare with $300 million in annual revenue and 1,200 staff members. Earlier in his
career, he served as senior sales executive and enterprise vice president of clinical sales at McKesson, a FORTUNE 500 company
delivering healthcare services and information technology. Mr. Brooks holds a Bachelor of Arts in Economics and Speech Communication
from St. Cloud State University in Minnesota.
Aside
from that provided above, Mr. Brooks does not hold and has not held over the past five years any other directorships in any company
with a class of securities registered pursuant to Section 12 of the Exchange Act or subject to the requirements of Section 15(d)
of the Exchange Act or any company registered as an investment company under the Investment Company Act of 1940.
Gus
D. Halas
Mr.
Halas has served as CEO of several companies. He was Chief Executive Officer and President of the Central Operating Companies
at Central Garden & Pet Company from April 2011 through May 2013 and currently serves as a consultant to that Company. Mr.
Halas was President and Chief Executive Officer of T-3 Energy Services, Inc. from May 2003 to March 2009 and also served as Chairman
of the Board of Directors from March 2004 to March 2009. From August 2001 to April 2003, Mr. Halas served as President and Chief
Executive Officer of Clore Automotive, Inc. He also serves as a director for Triangle Petroleum Corp. and Hooper Holmes, Inc.
Aside
from that provided above, Mr. Halas does not hold and has not held over the past five years any other directorships in any company
with a class of securities registered pursuant to Section 12 of the Exchange Act or subject to the requirements of Section 15(d)
of the Exchange Act or any company registered as an investment company under the Investment Company Act of 1940.
Mr.
Halas is qualified to serve on our Board of Directors because of his experience and expertise as an executive and a director with
companies implementing “turnaround” strategies.
Jack
Pinney
From
2007 to the present, Dr. Pinney has served as Team Physician to the Great Lakes Loons baseball team in the LA Dodgers organization.
From 2011 to the present, he has served as Medical Director for WellSport MidMichigan Medical Center. From 1992 to the present,
he has served as Assistant Clinical Professor of Family Medicine for the Department of Family Medicine at Michigan State University
College of Human Medicine. From 1992 to 2012, he served as Assistant Director for the Midland Family Practice Residency Program
at MidMichigan Medical Center.
Aside
from that provided above, Dr. Pinney does not hold and has not held over the past five years any other directorships in any company
with a class of securities registered pursuant to Section 12 of the Exchange Act or subject to the requirements of Section 15(d)
of the Exchange Act or any company registered as an investment company under the Investment Company Act of 1940.
Dr.
Pinney is qualified to serve on our Board of Directors because of his expertise medicine and prescription practices of physicians.
Lynn
Vos
Ms.
Vos has been chief executive officer of ghg | greyhealth group since 1994 and is a champion of using digital capabilities to improve
the public health. Ms. Vos also serves on the board of nTelos Wireless, a NASDAQ listed company, the Jed Foundation, a leading
nonprofit dedicated to protecting the emotional health of college students, and was a founding board member of MMRF, a pioneering
cancer research foundation.
Aside
from that provided above, Ms. Vos does not hold and has not held over the past five years any other directorships in any company
with a class of securities registered pursuant to Section 12 of the Exchange Act or subject to the requirements of Section 15(d)
of the Exchange Act or any company registered as an investment company under the Investment Company Act of 1940.
Ms.
Vos is qualified to serve on our Board of Directors because of her experience and expertise in digital marketing and communications
in the healthcare industry.
Directors
Our
bylaws authorize no less than three (3) and no more than Seven (7) Directors unless changed by the Board of Directors. The Investor
Rights Agreement we signed with WPP Luxembourg Gamma Three Sarl states that our Board of Directors shall consist of five (5) Directors.
We currently have five (5) Directors.
Term
of Office
Our
Directors are appointed for a one-year term to hold office until the next annual general meeting of our shareholders or until
removed from office in accordance with our bylaws. Our officers are appointed by our board of directors and hold office until
removed by the board, subject to their respective employment agreements.
Significant
Employees
We
have no significant employees other than our officers and directors.
Family
Relationships
There
are no family relationships between or among the directors, executive officers or persons nominated or chosen by us to become
directors or executive officers.
Involvement
in Certain Legal Proceedings
During
the past 10 years, none of our current directors, nominees for directors or current executive officers has been involved in any
legal proceeding identified in Item 401(f) of Regulation S-K, including:
|
|
1.
|
Any
petition under the Federal bankruptcy laws or any state insolvency law filed by or against,
or a receiver, fiscal agent or similar officer was appointed by a court for the business
or property of such person, or any partnership in which he or she was a general partner
at or within two years before the time of such filing, or any corporation or business
association of which he or she was an executive officer at or within two years before
the time of such filing;
|
|
|
2.
|
Any
conviction in a criminal proceeding or being named a subject of a pending criminal proceeding
(excluding traffic violations and other minor offenses);
|
|
|
3.
|
Being
subject to any order, judgment, or decree, not subsequently reversed, suspended or vacated,
of any court of competent jurisdiction, permanently or temporarily enjoining him or her
from, or otherwise limiting, the following activities: i. Acting as a futures commission
merchant, introducing broker, commodity trading advisor, commodity pool operator, floor
broker, leverage transaction merchant, any other person regulated by the Commodity Futures
Trading Commission, or an associated person of any of the foregoing, or as an investment
adviser, underwriter, broker or dealer in securities, or as an affiliated person, director
or employee of any investment company, bank, savings and loan association or insurance
company, or engaging in or continuing any conduct or practice in connection with such
activity; ii. Engaging in any type of business practice; or iii. Engaging in any activity
in connection with the purchase or sale of any security or commodity or in connection
with any violation of Federal or State securities laws or Federal commodities laws;
|
|
|
4.
|
Being
subject to any order, judgment or decree, not subsequently reversed, suspended or vacated,
of any Federal or State authority barring, suspending or otherwise limiting for more
than 60 days the right of such person to engage in any type of business regulated by
the Commodity Futures Trading Commission, securities, investment, insurance or banking
activities, or to be associated with persons engaged in any such activity;
|
|
|
5.
|
Being
found by a court of competent jurisdiction in a civil action or by the SEC to have violated
any Federal or State securities law, and the judgment in such civil action or finding
by the Commission has not been subsequently reversed, suspended, or vacated;
|
|
|
6.
|
Being
found by a court of competent jurisdiction in a civil action or by the Commodity Futures
Trading Commission to have violated any Federal commodities law, and the judgment in
such civil action or finding by the Commodity Futures Trading Commission has not been
subsequently reversed, suspended or vacated;
|
|
|
7.
|
Being
subject to, or a party to, any Federal or State judicial or administrative order, judgment,
decree, or finding, not subsequently reversed, suspended or vacated, relating to an alleged
violation of: i. Any Federal or State securities or commodities law or regulation; or
ii. Any law or regulation respecting financial institutions or insurance companies including,
but not limited to, a temporary or permanent injunction, order of disgorgement or restitution,
civil money penalty or temporary or permanent cease-and-desist order, or removal or prohibition
order; or iii. Any law or regulation prohibiting mail or wire fraud or fraud in connection
with any business entity; or
|
|
|
8.
|
Being
subject to, or a party to, any sanction or order, not subsequently reversed, suspended
or vacated, of any self-regulatory organization (as defined in Section 3(a)(26) of the
Exchange Act (15 U.S.C. 78c(a)(26))), any registered entity (as defined in Section 1(a)(29)
of the Commodity Exchange Act (7 U.S.C. 1(a)(29))), or any equivalent exchange, association,
entity or organization that has disciplinary authority over its members or persons associated
with a member.
|
Audit
Committee
We
do not have a separately-designated standing audit committee. The entire board of directors performs the functions of an audit
committee, but no written charter governs the actions of the board of directors when performing the functions of that would generally
be performed by an audit committee. The board of directors approves the selection of our independent accountants and meets and
interacts with the independent accountants to discuss issues related to financial reporting. In addition, the board of directors
reviews the scope and results of the audit with the independent accountants, reviews with management and the independent accountants
our annual operating results, considers the adequacy of our internal accounting procedures and considers other auditing and accounting
matters including fees to be paid to the independent auditor and the performance of the independent auditor.
For
the fiscal year ending December 31, 2015, the board of directors:
|
|
1.
|
Reviewed
and discussed the audited financial statements with management, and
|
|
|
2.
|
Reviewed
and discussed the written disclosures and the letter from our independent auditors on
the matters relating to the auditor's independence.
|
Based
upon the board of directors’ review and discussion of the matters above, the board of directors authorized inclusion of
the audited financial statements for the year ended December 31, 2015 to be included in this Prospectus filed with the Securities
and Exchange Commission.
Section
16(a) Beneficial Ownership Reporting Compliance
Section
16(a) of the Exchange Act requires our directors and executive officers and persons who beneficially own more than ten percent
of a registered class of the Company’s equity securities to file with the SEC initial reports of ownership and reports of
changes in ownership of common stock and other equity securities of the Company. Officers, directors and greater than ten percent
beneficial shareholders are required by SEC regulations to furnish us with copies of all Section 16(a) forms they file. To the
best of our knowledge based solely on a review of Forms 3, 4, and 5 (and any amendments thereof) received by us, no persons have
failed to file, on a timely basis, the identified reports required by Section 16(a) of the Exchange Act during fiscal year ended
December 31, 2015, other than the Form 3 for Director Vos which was filed late.
Code
of Ethics
As
of December 31, 2015, we had not adopted a Code of Ethics. We felt, until recently, the small number of individuals comprising
our board and management did not warrant the adoption of a Code of Ethics. Now that we have expanded our board and our increasing
the size of our organization, we intend to adopt a Code of Ethics in the near future.
SECURITY
OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The
following table sets forth the beneficial ownership by each person, other than executive officers and directors, known to us to
beneficially own 5% or more of our outstanding common stock as of June 2, 2016. This information is based on public filings as
of June 2, 2016. For the purposes of this Prospectus, beneficial ownership of securities is defined in accordance with the rules
of the SEC to mean generally the power to vote or dispose of securities, regardless of any economic interest therein, including
any such security that the person has the right to acquire within 60 days after such date.
|
More
Than 5% Beneficial Owners:
|
|
Name
and Address
|
|
Common
Shares Owned
|
|
|
Percentage
of Class
|
|
|
Common
|
|
WPP PLC
27
Farm Street
London, United Kingdom
W1J
5RJ
|
|
|
6,011,106
|
|
|
|
20.7
|
%
|
|
Common
|
|
Wolverine Flagship Fund
Trading
Limited 175 W Jackson Blvd,
3rd Flr Chicago, IL 60604
|
|
|
2,154,500
|
|
|
|
7.4
|
%
|
|
Common
|
|
Harvey L. Poppel
110
El Mirasol
Palm Beach, FL 33480
|
|
|
2,129,028
|
|
|
|
7.3
|
%
|
|
Common
|
|
Goldman
Capital Management, Inc.
767
Third Ave., 25
th
Floor
New
York, NY 10017
|
|
|
1,532,394
|
|
|
|
5.3
|
%
|
|
Common
|
|
Ronald L. Chez
55
East Monroe Street, Suite 3700
Chicago, IL 60603
|
|
|
2,721,976
|
|
|
|
9.4
|
%
|
Set
forth below is certain information with respect to beneficial ownership of our common stock as of June 2, 2016, by each director,
each executive officer, and by the directors and executive officers as a group. Unless otherwise indicated, each person or member
of the group listed has sole voting and investment power with respect to the shares of common stock listed.
|
Name(1)
|
|
Options
Included
in
Beneficial
Ownership
(2)
|
|
|
Shares
Related to Stock Awards and Note Payable (3)
|
|
|
Common
Shares
Owned
|
|
|
Common
Stock
Beneficial
Ownership
|
|
|
Percentage
of
Class
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
David A. Harrell
|
|
|
0
|
|
|
|
0
|
|
|
|
2,590,897
|
|
|
|
2,590,897
|
|
|
|
8.9
|
%
|
|
William J. Febbo
|
|
|
0
|
|
|
|
0
|
|
|
|
9,433
|
|
|
|
9,433
|
|
|
|
*
|
|
|
Terence J. Hamilton
|
|
|
0
|
|
|
|
384,188
|
|
|
|
380,413
|
|
|
|
764,601
|
|
|
|
2.6
|
%
|
|
Lynn Vos
|
|
|
0
|
|
|
|
0
|
|
|
|
0
|
|
|
|
0
|
|
|
|
*
|
|
|
James Brooks
|
|
|
|
|
|
|
0
|
|
|
|
0
|
|
|
|
0
|
|
|
|
*
|
|
|
Douglas P. Baker
|
|
|
100,000
|
|
|
|
0
|
|
|
|
20,000
|
|
|
|
120,000
|
|
|
|
*
|
|
|
Gus D. Halas
|
|
|
0
|
|
|
|
0
|
|
|
|
45,019
|
|
|
|
45,019
|
|
|
|
*
|
|
|
Jack Pinney
|
|
|
0
|
|
|
|
0
|
|
|
|
777,079
|
|
|
|
777,079
|
|
|
|
2.7
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
All Executive Officers and
Directors as a
group (6 persons)
|
|
|
100,000
|
|
|
|
384,188
|
|
|
|
3,822,841
|
|
|
|
4,307,029
|
|
|
|
14.8
|
%
|
|
*
|
Less
than 1%
|
|
(1)
|
The
address of each person named in this table is c/o OptimizeRx Corp., 400 Water Street,
Suite 200, Rochester, MI 48307.
|
|
(2)
|
This
column lists shares that are subject to options exercisable within sixty (60) days of
June 2, 2016, and are included in common stock beneficial ownership pursuant to Rule
13d-3(d)(1) of the Exchange Act.
|
|
(3)
|
This
column lists shares that are obtainable as result of stock awards for shares not yet
issued or notes that are payable in stock as of June 2, 2016.
|
DESCRIPTION
OF SECURITIES
The
following descriptions are summaries of the material terms that are included in our amended and restated articles of incorporation
(as amended) and our bylaws (as amended) as well as the specific agreements such descriptions relate to. This summary is qualified
in its entirety by the specific terms and provisions contained in our restated articles of incorporation, bylaws and the specific
agreements described herein, copies of which we have filed as exhibits to the registration statement of which this prospectus
is a part, and by the provisions of applicable law.
Overview
Authorized
Capital Stock
Our
authorized capital stock consists of 500,000,000 shares of common stock and 10,000,000 shares of preferred stock, par value $0.001
per share. As of June 2, 2016, there were 29,077,660 shares of our common stock issued and outstanding and 0 shares of our preferred
stock issued and outstanding.
Common
Stock
Our
common stock is entitled to one vote per share on all matters submitted to a vote of the stockholders, including the election
of directors. Except as otherwise required by law or provided in any resolution adopted by our board of directors with respect
to any series of preferred stock, the holders of our common stock will possess all voting power. Generally, all matters to be
voted on by stockholders must be approved by a majority (or, in the case of election of directors, by a plurality) of the votes
entitled to be cast by all shares of our common stock that are present in person or represented by proxy, subject to any voting
rights granted to holders of any preferred stock. Holders of our common stock representing fifty percent (50%) of our capital
stock issued, outstanding and entitled to vote, represented in person or by proxy, are necessary to constitute a quorum at any
meeting of our stockholders. A vote by the holders of a majority of our outstanding shares is required to effectuate certain fundamental
corporate changes such as liquidation, merger or an amendment to our Articles of Incorporation. Our Articles of Incorporation
do not provide for cumulative voting in the election of directors.
Subject
to any preferential rights of any outstanding series of preferred stock created by our board of directors from time to time, the
holders of shares of our common stock will be entitled to such cash dividends as may be declared from time to time by our board
of directors from funds available therefore.
Subject
to any preferential rights of any outstanding series of preferred stock created from time to time by our board of directors, upon
liquidation, dissolution or winding up, the holders of shares of our common stock will be entitled to receive pro rata all assets
available for distribution to such holders.
In
the event of any merger or consolidation with or into another company in connection with which shares of our common stock are
converted into or exchangeable for shares of stock, other securities or property (including cash), all holders of our common stock
will be entitled to receive the same kind and amount of shares of stock and other securities and property (including cash). Holders
of our common stock have no pre-emptive rights, no conversion rights and there are no redemption provisions applicable to our
common stock.
Preferred
Stock
Our
board of directors may become authorized to authorize preferred shares of stock and to divide the authorized shares of our preferred
stock into one or more series, each of which must be so designated as to distinguish the shares of each series of preferred stock
from the shares of all other series and classes. Our board of directors is authorized, within any limitations prescribed by law
and our articles of incorporation, to fix and determine the designations, rights, qualifications, preferences, limitations and
terms of the shares of any series of preferred stock including, but not limited to, the following:
|
|
(1)
|
The
number of shares constituting that series and the distinctive designation of that series, which may be by distinguishing number,
letter or title;
|
|
|
(2)
|
The
dividend rate on the shares of that series, whether dividends will be cumulative, and if so, from which date(s), and the relative
rights of priority, if any, of payment of dividends on shares of that series;
|
|
|
(3)
|
Whether
that series will have voting rights, in addition to the voting rights provided by law, and, if so, the terms of such voting
rights;
|
|
|
(4)
|
Whether
that series will have conversion privileges, and, if so, the terms and conditions of such conversion, including provision
for adjustment of the conversion rate in such events as the Board of Directors determines;
|
|
|
(5)
|
Whether
or not the shares of that series will be redeemable, and, if so, the terms and conditions of such redemption, including the
date or date upon or after which they are redeemable, and the amount per share payable in case of redemption, which amount
may vary under different conditions and at different redemption dates;
|
|
|
(6)
|
Whether
that series will have a sinking fund for the redemption or purchase of shares of that series, and, if so, the terms and amount
of such sinking fund;
|
|
|
(7)
|
The
rights of the shares of that series in the event of voluntary or involuntary liquidation, dissolution or winding up of the
corporation, and the relative rights of priority, if any, of payment of shares of that series; and
|
|
|
(8)
|
Any
other relative rights, preferences and limitations of that series.
|
Registration
Rights
The
Shares Underlying Warrants Issued in Connection With the March 2014 Private Placement Financing
In
connection with the March 2014 Private Placement, we entered into a Registration Rights Agreement. The Registration Rights Agreement
required us to file a registration statement with the Securities and Exchange Commission in connection with shares sold in the
March 2014 Private Placement. We filed a registration statement that went effective on May 28, 2014 to comply with the terms of
the Registration Rights Agreement. In addition, we granted piggyback registration rights to the Placement Agents in the March
2014 Private Placement, which comprise the shares of common stock underlying the Agent Warrants that are being registered herein.
We are filing this registration statement to maintain the registered status of those shares underlying the Agent Warrants.
The
Shares Issued in Connection with the Merriman Capital, Inc. Advisory Agreement
On
February 23, 2015, we entered into the Advisory Agreement with Merriman. We granted registration rights to Merriman concerning
the 150,000 shares in the Advisory Agreement.
The
Shares Issued in Connection with the Settlement Agreement
On
May 27, 2016, we entered into a Settlement Agreement with Mr. Shadron Stastney whereby we granted registration rights to his 133,333
shares issued under a Separation Agreement.
Investor
Rights Agreement
On
September 24, 2015, we entered into an Investor Rights Agreement with WPP Luxembourg (the “Rights Agreement”), pursuant
to which we agreed to the following:
|
|
●
|
Demand
Registration Rights. We granted the Investor registration rights for the 6,011,106 acquired
from us and any securities acquired in connection with an Amended and Restated Co-Marketing
Agreement after a period of two years.
|
|
|
●
|
Inspection
Rights. So long as the Investor owns not less than 25% of the Shares, we granted the
Investor an annual right to inspect our books and records.
|
|
|
●
|
Observer
Rights. So long as the Investor owns not less than 25% of the Shares, we will allow the
Investor to choose a representative to attend our board meetings as a nonvoting observer.
|
|
|
●
|
Board
Seat. So long as the Investor owns not less than 25% of the Shares, we agreed to appoint
a nominee of the Investor as a member of our board of directors. We also agreed to a
five member Board of Directors provided that it is not prohibited by the rules and regulations
of an exchange that we trade on. We also agreed to enter into an Indemnity Agreement
with the nominee.
|
|
|
●
|
Budget
Review. So long as the Investor owns not less than 25% of the Shares, we agreed to review
our budget plans with the Investor’s nominee prior to submission to the Board of
Directors, at the request of the Investor.
|
|
|
●
|
Right
of First Refusal. We agreed that, in the event that it proposes to sell new securities,
we will first offer such new securities to the Investor.
|
|
|
●
|
Special
Approval Matters. So long as the Investor owns not less than 25% of the Shares, and provided
that it is not prohibited by the rules and regulations of an exchange that we trades
on, we agreed that 80% Board approval will be required for certain decisions, including:
|
|
|
●
|
the
incurrence of any indebtedness in excess of $1.5 million in the aggregate during any
fiscal year
|
|
|
●
|
the
sale, transfer or other disposition of all or substantially all of our assets;
|
|
|
●
|
the
acquisition of any assets or properties (in one or more related transactions) for cash
or otherwise for an amount in excess of $1.5 million in the aggregate during any fiscal
year;
|
|
|
●
|
capital
expenditures in excess of $1.5 million individually (or in the aggregate if related to
an integrated program of activities) or in excess of $1.5 million in the aggregate during
any fiscal year;
|
|
|
●
|
making,
or permitting any subsidiary to make, loans to, investments in, or purchasing, or permitting
any subsidiary to purchase, any stock or other securities in another corporation, joint
venture, partnership or other entity;
|
|
|
●
|
the
commencement or settlement of any lawsuit, arbitration or other legal proceeding related
to our intellectual property or involving an amount in controversy greater than $1.5
million; and
|
|
|
●
|
the
issuance of new securities, except for securities issued under an equity incentive plan
and any issuance of common stock to vendors, advisors, financial institutions, suppliers
or joint venturers that do not exceed, individually or in the aggregate 5% of then issued
and outstanding capital stock of the Company.
|
Provisions
in Our Articles of Incorporation and By-Laws That Would Delay, Defer or Prevent a Change in Control
Our
articles of incorporation authorize our board of directors to issue a class of preferred stock commonly known as a "blank
check" preferred stock. Specifically, the preferred stock may be issued from time to time by the board of directors as shares
of one (1) or more classes or series. Our board of directors, subject to the provisions of our Articles of Incorporation and limitations
imposed by law, is authorized to adopt resolutions; to issue the shares; to fix the number of shares; to change the number of
shares constituting any series; and to provide for or change the following: the voting powers; designations; preferences; and
relative, participating, optional or other special rights, qualifications, limitations or restrictions, including the following:
dividend rights, including whether dividends are cumulative; dividend rates; terms of redemption, including sinking fund provisions;
redemption prices; conversion rights and liquidation preferences of the shares constituting any class or series of the preferred
stock.
In
each such case, we will not need any further action or vote by our shareholders. One of the effects of undesignated preferred
stock may be to enable the board of directors to render more difficult or to discourage an attempt to obtain control of us by
means of a tender offer, proxy contest, merger or otherwise, and thereby to protect the continuity of our management. The issuance
of shares of preferred stock pursuant to the board of director's authority described above may adversely affect the rights of
holders of common stock. For example, preferred stock issued by us may rank prior to the common stock as to dividend rights, liquidation
preference or both, may have full or limited voting rights and may be convertible into shares of common stock. Accordingly, the
issuance of shares of preferred stock may discourage bids for the common stock at a premium or may otherwise adversely affect
the market price of the common stock.
Dividend
Policy
We
have never declared or paid any cash dividends on our common stock. We currently intend to retain future earnings, if any, to
finance the expansion of our business. As a result, we do not anticipate paying any cash dividends in the foreseeable future.
Share
Purchase Warrants
We had warrants outstanding to purchase
2,044,583 shares of our common stock at a weighted average exercise price of $ 1.67 as of March 31, 2016.
Options
We had options outstanding to purchase
2,780,000 shares of our common stock at a weighted average exercise price of $1.10 as of March 31, 2016.
Convertible
Securities
We
have not issued and do not have outstanding any securities convertible into shares of our common stock or any rights convertible
or exchangeable into shares of our common stock.
Certain
Anti-Takeover Provisions
Nevada
Anti-Takeover Laws
Nevada
Revised Statutes sections 78.378 to 78.379 provide state regulation over the acquisition of a controlling interest in certain
Nevada corporations unless the articles of incorporation or bylaws of the corporation provide that the provisions of these sections
do not apply. Our articles of incorporation and bylaws do not state that these provisions do not apply. The statute creates a
number of restrictions on the ability of a person or entity to acquire control of a Nevada company by setting down certain rules
of conduct and voting restrictions in any acquisition attempt, among other things. The statute is limited to corporations that
are organized in the state of Nevada and that have 200 or more stockholders, at least 100 of whom are stockholders of record and
residents of the State of Nevada; and does business in the State of Nevada directly or through an affiliated corporation. Because
of these conditions, the statute currently does not apply to our company.
Listing
of Common Stock
Our
common stock is currently traded on the OTCQB under the trading symbol “OPRX.”
Transfer
Agent and Registrar
The
transfer agent and registrar of our common stock is Empire Stock Transfer, 1859 Whitney Mesa Dr, Henderson, NV 89014, telephone:
(702) 974-1444.
INTERESTS
OF NAMED EXPERTS
No
expert or counsel named in this prospectus as having prepared or certified any part of this prospectus or having given an opinion
upon the validity of the securities being registered or upon other legal matters in connection with the registration or offering
of the common stock was employed on a contingency basis, or had, or is to receive, in connection with the offering, a substantial
interest, direct or indirect, in the registrant or any of its parents or subsidiaries. Nor was any such person connected with
the registrant or any of its parents or subsidiaries as a promoter, managing or principal underwriter, voting trustee, director,
officer, or employee.
The
Doney Law Firm, our independent legal counsel, has provided an opinion on the validity of our common stock.
KLJ
& Associates, LLP has audited our financial statements included in this prospectus and registration statement to the extent
and for the periods set forth in their audit report. KLJ & Associates, LLP has presented their report with respect to our
audited financial statements. The report of KLJ & Associates, LLP is included in reliance upon their authority as experts
in accounting and auditing.
DISCLOSURE
OF COMMISSION POSITION ON INDEMNIFICATION
FOR
SECURITIES ACT LIABILITIES
.
Insofar
as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to our directors, officers and controlling
persons pursuant to the following provisions, or otherwise, we have been advised that in the opinion of the Securities and Exchange
Commission such indemnification is against public policy as expressed in the Act and is, therefore, unenforceable. In the event
that a claim for indemnification against such liabilities (other than the payment by us of expenses incurred or paid by a director,
officer or controlling person in the successful defense of any action, suit or proceeding) is asserted by such director, officer
or controlling person in connection with the shares being registered, we will, unless in the opinion of its counsel the matter
has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification
by it is against public policy as expressed in the Act and will be governed by the final adjudication of such issue.
DESCRIPTION
OF BUSINESS
Company
Highlights
Through
April 2016
|
|
1)
|
Our
sales for the first three months of 2016 were approximately $1.76 million, an 18% increase
over the same period in 2015.
|
|
|
2)
|
We
hired an experienced Senior Vice President of Business Development to lead the expansion
of our EHR network, as well as increase utilization of our existing network.
|
|
|
3)
|
We
hired a new CEO to lead the Company in its next stage of growth.
|
|
|
4)
|
We
sponsored the ePrescribe/EHR conference held in Philadelphia in March 2016, which generated
significant leads for our sales force.
|
|
|
5)
|
We
completed an agreement with TrialCard to co-market our joint capabilities.
|
|
|
6)
|
We
implemented a CRM for our expanding direct sales team, multiple partnerships as well
as EHR growth opportunities.
|
|
|
7)
|
Our
partnerships with WPP is starting to show promise with the addition of XX new brands
in our pipeline.
|
|
|
8)
|
We
initiated a re-branding of our company to demonstrate our ability to bring financial
and clinical messaging as well as brand support services, such as drug file integration
and sale force training.
|
|
|
9)
|
We
completed a full technology review and have kicked off several efforts to drive revenue
growth with additional services for our existing clients. We expect those to be completed
by Q3 2016.
|
|
|
10)
|
We
announced our first partnership in the independent pharmacy space with RxWiki.
|
Our
success in acquiring, integrating and expanding into new promotional EHR/eRx platforms continues to grow as well. We are actively
engaged in discussions with several EHRs to integrate our technology into their platforms. We are also working extensively with
our existing platforms to expand the reach of our eCoupon product to all of their providers, as well as increasing the utilization
of the eCoupon functionality by their existing users.
With
the growth of both our pharmaceutical products and our distribution network, we expect that our distribution of e-coupons will
continue to increase over last year.
Year
Ended 2015
|
|
1)
|
Our
sales for 2015 were $7.2 million, an 11% increase over 2014.
|
|
|
2)
|
In
2015, our promotional transactions, primarily from our core eCoupon distributions, increased
approximately 25% over those in 2014.
|
|
|
3)
|
In
2015, we distributed eCoupons related to approximately 85 different brands.
|
|
|
4)
|
Excluding
non-cash expenses, in 2015 we generated operating income of approximately $350,000.
|
|
|
5)
|
In
2015, we successfully launched our e-Coupon solution within the Practice Fusion network.
|
|
|
6)
|
We
continued to acquire new pharmaceutical manufacturers and brands promoting through our
platforms.
|
|
|
7)
|
We
hired an experienced sales professional in November, 2015 to lead our relationship with
media agencies and in December, an experienced executive to lead the expansion of our
network.
|
|
|
8)
|
We
secured an investment of approximately $4.7 million from WPP, a global provider of advertising,
marketing, and branding.
|
|
|
9)
|
We
completed an upgrade of our technology platform to Oracle database software to further
improve system and reporting capacity.
|
|
|
10)
|
We
proved an outstanding Return on Investment associated with our pharmaceutical promotions
through an independent analytics firm with multiple pharmaceutical brands.
|
|
|
11)
|
We
unveiled VoucherDVM and engaged leading platforms to offer automated vet product savings.
We initiated a beta launch in January, 2016 with the National Veterinary Associates.
|
|
|
12)
|
We
began a search for a new CEO to help lead us through our next stage of growth, which
culminated in the hiring of Will Febbo, who became CEO on February 22, 2016.
|
We
generated positive cash flow from operations in 2015, and we expect to continue to do so in 2016, as well as to be profitable
during the upcoming year based on the expected escalation of revenues.
Our
success in acquiring, integrating and expanding into new promotional EHR/eRx platforms continues to grow as well. We are discussing
2016 rollout dates with potential additional networks.
Pharmaceutical
Sales and Marketing Updates
Our
sales team continues to expand opportunities within existing and new clients. We are focused on adding additional brands for existing
clients, expanding the utilization of our network for existing brands, and obtaining new clients.
Additionally,
we are expanding our non e-Coupon services as follows:
|
|
●
|
New
Drug File Integration – we are designing a service to better insure that manufacturers’
drugs are present in every ePrescribing platform available.
|
|
|
●
|
We
are actively expanding our messaging capabilities with the goal of rapidly increasing
revenue through this complementary service.
|
|
|
●
|
ePrescribe
Training – we are pursuing opportunities to leverage our partnership with WPP/Grey
to train representatives on understanding and leveraging EHR sales opportunities.
|
|
|
●
|
VoucherDVM
- we continue advancing our negotiations with each of the leading veterinarian technology
platforms and have signed an agreement with National Veterinary Associates, one of the
largest veterinary groups in the U.S., to initiate a beta launch in January, 2016.
|
We
are also continuing to ramp up our marketing efforts as follows:
|
|
●
|
Held
multiple meetings initiated and arranged by Pharmaceutical companies to bring on new
Health Systems/ePrescribe Platforms.
|
|
|
●
|
Spoke
at Coupon and Co-Pay Off-set Strategies Conference.
|
|
|
●
|
Spoke
at multiple conferences in 2015, including the Marcum 2015 MicroCap conference, the Liolios
4th annual Gateway conference and the LD Micro investor conference.
|
|
|
●
|
Sponsored
the 3rd Annual ePrescribe/EHR Conference.
|
With
the growth of both our pharmaceutical products and our distribution network, we expect that our distribution of eCoupons will
continue to increase substantially over last year.
Operational
Update
In
2016, in addition to expanding our network, we plan to intensively focus on increasing physician utilization of our partner networks.
We intend to working individually with each of our partners based on their particular situation to improve workflow to increase
coupon utilization by those providers that have access, obtain access for those prescribers that currently do not have e-coupon
access, and increase overall revenue derived from each channel. We believe there is significant revenue growth available within
our existing brands by better utilizing our existing partner networks, in addition to the revenue growth provided by new brands
and new network partners.
In
2015, we began the search for an experienced EHR executive to lead our expansion efforts. In January, 2016, we hired James Brooks
as Senior Vice President of Business Development to champion our EHR business through the development of new relationships with
other EHR providers and patient platforms.
Technology
Updates
To
support our growth, we have migrated our platform to Oracle database software. The system can now manage up to 1 million rules
and return the appropriate content within 1 second. This allows unsurpassed response time to avoid delays, and the ability to
meet the upcoming dramatic scale we expect.
We
have launched downloadable “wrapper” code, which streamlines the integration requirements for our solution from a
few weeks to a few days, if EHR channel partners choose to utilize this method. This addresses one of the biggest hurdles we face
in getting health systems and EHRs to implement our system, given the extensive demand on their available technical resources.
We
have developed a stand-alone desktop eCoupon application that can be used by prescribers that either are not using an eprescribing
application, or whose EHR vendor does not offer e-Coupon functionality. We have sold one such application to a major pharmaceutical
manufacturer for use as a tool by their sales force and to distribute to physicians. We expect this to be a source of revenue
growth in 2016.
Managerial
Updates
We
recently added one executive officer to our team.
James
Brooks
Effective
January 4, 2016, the board of directors appointed Mr. James Brooks to act as our Senior Vice President of Business Development.
Mr. Brooks brings more than 20 years of experience in sales & marketing, including sales force development, sales management,
and organizational leadership processes within the Electronic Health Record (EHR) and Healthcare Technology sector.
Effective
January 4, 2016, we entered into an employment agreement with Mr. Brooks. The agreement is at will and may be terminated by either
party at any time. Under the agreement, we agreed to compensate Mr. Brooks $185,000 annually and, subject to meeting certain assigned
goals, we agreed to pay him a bonus of up to $100,000 per year, along with a maximum of 300,000 stock options during the first
two years of employment.
Mr.
Brooks is entitled to participate in any existing employee benefit plans. He has paid vacation and sick days, and other benefits
included in the agreement.
Mr.
Brooks agreed to covenants not to solicit customers or recruit employees both during employment and for a period of one year following
termination. The agreement also contains a confidentiality provision.
David
Harrell
As
of March 31, 2016, Mr. David Harrell, ended his employment as our Chief Executive Officer.
On
May 9, 2016, we entered into a Separation Agreement and Release with Mr. Harrell that grants us a standard release of employment
claims in consideration for, among other things, a stock payout of $720,415 to Mr. Harrell.
On
the same date, we entered into a Corporate Consulting Agreement with Mr. Harrell that sets forth the terms his continued relationship
with our company. He will remain our employee through May 31, 2016 and the Corporate Consulting Agreement is effective as of June
1, 2016. Under the terms of this agreement, Mr. Harrell will consult for our company for a period of 16 months and he will receive
a monthly payment of $15,000, with the potential for up to $54,000 in additional bonus payments during the term of the agreement.
This agreement also calls for insurance benefits for seven months. Finally, the agreement contains a Consultant Confidentiality,
Invention Assignment and Non-Compete Agreement that contains restrictive covenants that include a one year non-compete following
the completion of Mr. Harrell’s 18 months of consulting, and an inventions assignment clause during the term of his consulting
relationship.
Other
Key Events in 2015
We
secured a strategic investment from WPP of approximately $4.7 million. The completion of this transaction represents a significant
expansion of our relationship with WPP, the largest marketing services company in the world, and Lynn Vos, CEO of WPP’s
subsidiary, Grey Healthcare Group, has joined our Board. We have hired a top executive to focus on overall client services, including
the WPP relationship, and to focus on expanding opportunities available to us.
In
addition to the strategic value, we expect the WPP relationship to provide promotional support, help add pharmaceutical brands,
aid in expansion of our EHR network, expand our service offerings, and help us expand our management and infrastructure. We anticipate
that that the WPP relationship will have a significant and positive impact on our business.
We
also signed an agreement with Allscripts to become its exclusive provider of eCoupons throughout all of its platforms as well
as to integrate our e-Coupons into its Touchworks network. The Touchworks platform is used by large health systems throughout
the country and is expected to represent a significant expansion of our network. ECoupon functionality within Touchworks is expected
to launch on a test basis in late 2016 and on a wide-scale basis in early 2017.
Summary
Despite
the lengthy sales cycle involved in creating this new eCoupon market, we remain very excited about our core eCoupon business and
expect acceleration to continue with the launch of additional channels and our joint pursuit of leading health systems with our
pharmaceutical partners. We expect our active network to grow substantially in 2016.
Principal
Products and Applications
Our
principal products and applications can be summarized as follows:
|
|
●
|
SAMPLEMD
- Our platform, which we refer to as SampleMD, is a revolutionary virtual "Patient
Support Center" that allows doctors and staff to access a universe of sample vouchers,
co-pay coupons and other patient support through their EMR and/or e-Prescribe systems
to search, print or electronically dispense directly to patients and a national network
of pharmacies. SampleMD eliminates the need for physicians to manage and store physical
drug samples by offering a more convenient and efficient way to allocate, administer
and track samples and co-pay savings provided to their patients. Today, almost 60% of
doctors’ offices ban or limit drug representatives and the samples they offer.
Although samples are still valuable, many healthcare systems and doctors are looking
for an easier, more effective way to increase affordable access and adherence to their
prescribed branded medications. Over 90% of our revenue comes through activities related
to our SampleMD platform.
|
|
|
●
|
OPTIMIZEHR
– Our consulting practice is focused on educating and working with pharmaceutical
manufacturers on identifying, formulating, and implementing new eRx media strategies
for promoting their products. Our consulting services include: 1) Drug File Integration
- a service designed to better insure that manufacturers’ drugs are present in
every ePrescribing platform available; 2) Sales Force Training – a service to educate
the extended field sales force on this new integrated solution and what to look for within
their client base to insure maximum exposure of their brands; and 3) Strategy Development
– a service that assists manufactures in identifying and building a competitive
strategy to take advantage of this new digital frontier. Currently, this activity results
in less than 5% of our revenue, but represents a significant growth opportunity for us.
|
|
|
●
|
OPTIMIZERx.com
– Our Direct to Consumer Website is a portal to healthcare savings for patients
to centrally review and participate in prescription and healthcare savings and support
programs. To date, we have over 2.4 million members who have registered. We strive to
provide all the information and guidance that patients undergoing long-term pharmaceutical
treatments may require. Patients can search by their medication or their condition in
order to access educational information regarding their condition, information regarding
their medication, coupons for instant savings when they purchase their medications, information
on free drug trials, and guidance to any other savings programs available to them. At
the present time, we generate no revenue through this site, but we believe it represents
a significant potential future revenue source.
|
Marketing
and Sales
We
continue to extend our marketing efforts to build both brand and capabilities awareness in the market. As previously discussed,
we continue to actively participate in industry and partner events such as exlPharma and the ACE – Allscripts Users Conference,
as well as taking a lead sponsor position in the CBInet eRx and EHR conferences in March and October of 2015. We are also the
named sponsor of the March 2016 conference. During the course of the year, we also initiated and delivered successful email marketing
campaigns, which generated viable leads for our sales force.
In
2015, we also announced the expansion of our strategic partnership with WPP/Grey Health Group, a leading agency within the healthcare
marketplace, which included a significant investment by an affiliated entity of WPP. We plan to continue to increase our marketing
efforts with all of our strategic partners, as we intend to continue to promote our platform primarily through the following:
|
|
●
|
Industry
and Partner Events;
|
|
|
|
|
|
|
●
|
Public
Relations Campaigns;
|
|
|
|
|
|
|
●
|
Direct
to Consumer Marketing;
|
|
|
|
|
|
|
●
|
Trade
Media Advertising;
|
|
|
|
|
|
|
●
|
Physician
Organizations and Associations; and
|
|
|
|
|
|
|
●
|
Strategic
Relationships.
|
Competition
Our
platform competes in the highly competitive pharmaceutical and healthcare advertising industry that is dominated by large well-known
companies with established names, solid market niches, wide arrays of product offerings and marketing networks. Coupon offerings
compete for pharmaceutical budgets with a variety of other forms of advertising and promotion.
Despite
these overall competitors, we do not have major competition in our specific portion of the market. We have been experiencing a
growing list of potential partners whom either have content that they want to deliver through our distribution engine and network,
or whom have complementary technology and want to integrate our solution as a channel partner, expanding our reach to clinicians.
The primary competitors in our space of the market are PDR Network, LLC and Physicians Interactive Holdings, Inc. However, we
believe our breadth of brands offered, extensive list of pharmaceutical clients, and the vast reach of our network give us a substantial
advantage and allow us to achieve a dominant position in the marketplace.
Intellectual
Property
In
2012, we were awarded a patent for our innovative solution (US Patent No. 8,341,015). This award was a result of our extensive
research and development efforts. The awarded claims cover our ability to electronically process, display and distribute eligible
prescription savings on the medications and therapies healthcare providers wish to prescribe for their patients.
We
have hired Harness, Dickey & Pierce, a nationally ranked IP firm, to further expand and protect our intellectual property.
Through them, we have filed two additional patents on our technology. We believe our current and expanding IP will allow us to
continue being the leader in this rapidly growing space. We stand ready to prepare additional filings, as necessary, to protect
our intellectual property on any forthcoming solutions that will further assist and support physicians, pharmacists and patients.
OPTIMIZERx
and SampleMD are our licensed trademarks.
Government
Regulation
Fraud
and Abuse Laws
Anti-Kickback
Statutes
The
federal healthcare program Anti-Kickback Statute prohibits persons from knowingly and willfully soliciting, offering, receiving
or providing remuneration, directly or indirectly, in exchange for or to induce either the referral of an individual for, or the
furnishing, arranging for or recommending a good or service for which payment may be made in whole or part under a federal healthcare
program such as Medicare or Medicaid. The definition of remuneration has been broadly interpreted to include anything of value,
including for example gifts, discounts, the furnishing of supplies or equipment, credit arrangements, payments of cash and waivers
of payments. Several courts have interpreted the statute's intent requirement to mean that if any one purpose of an arrangement
involving remuneration is to induce referrals or otherwise generate business involving goods or services reimbursed in whole or
in part under federal healthcare programs, the statute has been violated. The law contains a few statutory exceptions, including
payments to bona fide employees, certain discounts and certain payments to group purchasing organizations. Violations can result
in significant penalties, imprisonment and exclusion from Medicare, Medicaid and other federal healthcare programs. Exclusion
of a manufacturer would preclude any federal healthcare program from paying for its products. In addition, kickback arrangements
can provide the basis for an action under the Federal False Claims Act, which is discussed in more detail below. The Anti-Kickback
Statute is broad and potentially prohibits many arrangements and practices that are lawful in businesses outside of the healthcare
industry. Recognizing that the Anti-Kickback Statute is broad and may technically prohibit many innocuous or beneficial arrangements,
the Office of Inspector General of Health and Human Services, or OIG, issued a series of regulations, known as the safe harbors,
beginning in July 1991. These safe harbors set forth provisions that, if all the applicable requirements are met, will assure
healthcare providers and other parties that they will not be prosecuted under the Anti-Kickback Statute. The failure of a transaction
or arrangement to fit precisely within one or more safe harbors does not necessarily mean that it is illegal or that prosecution
will be pursued. However, conduct and business arrangements that do not fully satisfy each applicable safe harbor may result in
increased scrutiny by government enforcement authorities such as the OIG. Arrangements that implicate the Anti-Kickback Law, and
that do not fall within a safe harbor, are analyzed by the OIG on a case-by-case basis. Government officials have focused recent
enforcement efforts on, among other things, the sales and marketing activities of healthcare companies, and recently have brought
cases against individuals or entities with personnel who allegedly offered unlawful inducements to potential or existing customers
in an attempt to procure their business. Settlements of these cases by healthcare companies have involved significant fines and/or
penalties and in some instances criminal pleas. In addition to the Federal Anti-Kickback Statute, many states have their own kickback
laws. Often, these laws closely follow the language of the federal law, although they do not always have the same exceptions or
safe harbors. In some states, these anti-kickback laws apply with respect to all payors, including commercial health insurance
companies.
False
Claims Laws
Federal
false claims laws prohibit any person from knowingly presenting, or causing to be presented, a false claim for payment to the
federal government or knowingly making, or causing to be made, a false statement to get a false claim paid. Manufacturers can
be held liable under false claims laws, even if they do not submit claims to the government, if they are found to have caused
submission of false claims. The Federal Civil False Claims Act also includes whistle blower provisions that allow private citizens
to bring suit against an entity or individual on behalf of the United States and to recover a portion of any monetary recovery.
Many of the recent highly publicized settlements in the healthcare industry related to sales and marketing practices have been
cases brought under the False Claims Act. The majority of states also have statutes or regulations similar to the federal false
claims laws, which apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply
regardless of the payor. Sanctions under these federal and state laws may include civil monetary penalties, exclusion of a manufacturer's
products from reimbursement under government programs, criminal fines and imprisonment.
Privacy
and Security
The
Health Insurance Portability and Accountability Act of 1996, or HIPAA, and the rules promulgated there under require certain entities,
referred to as covered entities, to comply with established standards, including standards regarding the privacy and security
of protected health information, or PHI. HIPAA further requires that covered entities enter into agreements meeting certain regulatory
requirements with their business associates, as such term is defined by HIPAA, which, among other things, obligate the business
associates to safeguard the covered entity's PHI against improper use and disclosure. While not directly regulated by HIPAA, our
customers or distributors might face significant contractual liability pursuant to such an agreement if the business associate
breaches the agreement or causes the covered entity to fail to comply with HIPAA. It is possible that HIPPA compliance could become
a substantial regulatory burden and expense to our operations, although we do not believe that this will occur as a general website
publisher.
Employees
As
of the date of this Prospectus, we had 14 full-time employees and 4 part-time employees, in addition to contracted programmers,
as needed, through our established relationship with Simple eSolutions, a technical and programming resources partner.
Subsidiaries
We
conduct our operations through our wholly-owned subsidiary, OptimizeRx Corporation, a Michigan corporation.
Description
of Property
Currently,
we do not own any real estate. Our principal executive offices are located at 400 Water Street, Suite 200, Rochester, Michigan,
48307. We initially entered into a 3 year lease for this 3,648 square foot facility, with a cost of $5,049.25 per month. We renewed
that lease for a two year period on December 1, 2014 for a monthly rental rate of $5,201.50. We believe that our properties are
adequate for our current needs, but growth potential may require larger facilities due to anticipated addition of personnel. We
do not have any policies regarding investments in real estate, securities or other forms of property.
Management’s
Discussion and Analysis of Financial Condition and Results of Operations
Results
of Operations for the Three Months Ended March 31, 2016 and 2015
Revenues
Our
total revenue reported for the three months ended March 31, 2016 was approximately $1.76 million, an increase of 18% over the
approximately $1.49 million from the same period in 2015. These increased revenues result from both increased pharmaceutical brands
being promoted and expanded distribution channels. We expect quarter over quarter revenue increases for the balance of 2016.
Cost
of Sales
Our
cost of sales, composed of revenue share expense, increased over the same period in 2015 as a result of the revenue increases.
Our revenue share expense as a percentage of revenue remained relatively constant at approximately 51% in both periods. We expect
this percentage to gradually decrease in future quarters as we implement new channels with lower revenue share percentages and
as we update our existing agreements to share third party costs, with a goal of a decrease of at least 5%.
Operating
Expenses
Operating
expenses increased from approximately $850,000 for the three month period ended March 31, 2015 to approximately $1.23 million
for the same period in 2016, an increase of approximately 46%. The detail by major category is reflected in the table below.
|
|
|
Three
months ended March 31
|
|
|
|
|
2016
|
|
|
2015
|
|
|
|
|
|
|
|
|
|
|
Salaries, Wages, & Benefits
|
|
$
|
557,206
|
|
|
$
|
395,199
|
|
|
Stock-based compensation
|
|
|
106,384
|
|
|
|
79,591
|
|
|
Professional Fees
|
|
|
179,278
|
|
|
|
73,420
|
|
|
Board Compensation
|
|
|
12,500
|
|
|
|
12,500
|
|
|
Investor Relations
|
|
|
28,019
|
|
|
|
25,962
|
|
|
Consultants
|
|
|
31,323
|
|
|
|
21,115
|
|
|
Advertising and Promotion
|
|
|
70,765
|
|
|
|
43,150
|
|
|
Depreciation and Amortization
|
|
|
50,268
|
|
|
|
79,668
|
|
|
Development and Maintenance
|
|
|
74,616
|
|
|
|
50,414
|
|
|
Office, Facility, and other
|
|
|
43,440
|
|
|
|
33,058
|
|
|
Travel
|
|
|
74,765
|
|
|
|
28,532
|
|
|
Total Operating
Expense
|
|
$
|
1,228,564
|
|
|
$
|
842,610
|
|
The
largest increases in operating expenses related to human resource costs and professional fees. Since the first quarter of 2015,
we have hired a Vice President of Client Services, a Senior Vice President of Business Development, an additional Vice President
of Sales, and a new CEO. The first quarter of 2016 includes an overlap period in the CEO position where our new CEO started in
February and our previous CEO remained on the payroll until March 31. These new hires also resulted in increases in benefits,
payroll taxes, and travel. Our professional fees increased significantly as a result of the litigation described in the footnotes
to our financial statements. Both of our legal cases are in active periods and we expect to continue to incur significant costs
related to the litigation.
We
expect our overall operating expenses to continue to increase as we further implement our business plan and expand our operations.
Net
Loss
Our
net loss for the three months ended March 31, 2016 was approximately $350,000 as compared to a loss of approximately $110,000
during the same period in 2015. The reasons for specific components are discussed above. Overall, the increased margin resulting
from the increased sales was offset by the increased operating expenses described above.
Results
of Operations for the Years Ended December 31, 2015 and 2014
Revenues
Our
total revenue reported for the year ended December 31, 2015 was approximately $7.2 million, an increase of 11% from the year ended
December 31, 2014. This increased revenue resulted from increases in all major revenue categories including setup fees, reporting
fees, and e-Coupon distributions and resulted from both increased pharmaceutical brands being promoted and expanded distribution
channels. We expect continued revenue increases in 2016.
Because
the pharmaceutical industry is dominated by large companies with multiple brands, our revenue is concentrated in a relatively
small number of companies. We have approximately 25 pharmaceutical companies as customers and we received approximately 52% of
our revenue in the year ended December 31, 2015 from our largest 5 customers, with approximately 20% of revenue from our largest
customer. During the year ended December 31, 2014, approximately 64% of revenue came from our 5 largest customers, with 20% from
our largest customer. While we are still dependent on major customers, as our customer base expands, we are becoming less dependent
on any one customer.
Cost
of Sales
Our
total cost of sales, composed of revenue share expense, increased in the year ended December 31, 2015, over the year ended December
31, 2014 as a result of the revenue increases. In addition, revenue share expense as a percentage of revenue in 2015 increased
over 2014 from approximately 49.5% in the year ended December 31, 2014 to approximately 50.1% in the year ended December 31, 2015.
These
increases in revenue share expense as a percentage of revenue result from a combination of factors, including product mix whereby
a larger percentage of overall revenues are subject to revenue share. We expect revenue share expense as a percentage of revenue
in 2016 to continue at levels similar to, or greater than, that of 2015 as revenues subject to revenue share expense continues
to increase as a percentage of our overall revenues. In addition, our comarketing agreement with WPP requires us to pay revenue
share to WPP agencies on new brands secured for us by those agencies. If we successfully expand revenue through the WPP relationship,
our revenue share percentage will increase.
Operating
Expenses
Operating
expenses decreased to approximately $4.2 million for the year ended December 31, 2015 from approximately $4.3 million for the
year ended December 31, 2014, a decrease of approximately 3%. The detail by major category is reflected in the table below.
|
|
|
Years
Ended December 31
|
|
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
|
|
|
|
|
Salaries, Wages, & Benefits
|
|
$
|
1,788,471
|
|
|
$
|
1,477,450
|
|
|
Professional Fees
|
|
|
397,566
|
|
|
|
242,169
|
|
|
Board Compensation
|
|
|
50,000
|
|
|
|
19,565
|
|
|
Investor Relations
|
|
|
80,618
|
|
|
|
110,998
|
|
|
Consultants
|
|
|
66,985
|
|
|
|
72,487
|
|
|
Advertising and Promotion
|
|
|
47,927
|
|
|
|
87,201
|
|
|
Depreciation and Amortization
|
|
|
333,950
|
|
|
|
264,340
|
|
|
Development and Maintenance
|
|
|
290,371
|
|
|
|
189,566
|
|
|
Exclusivity Fee
|
|
|
250,000
|
|
|
|
-
|
|
|
Office, Facility, and other
|
|
|
151,826
|
|
|
|
140,101
|
|
|
Travel
|
|
|
157,062
|
|
|
|
131,637
|
|
|
|
|
|
|
|
|
|
|
|
|
Subtotal
|
|
|
3,614,776
|
|
|
|
2,735,514
|
|
|
|
|
|
|
|
|
|
|
|
|
Stock-based compensation
|
|
|
581,106
|
|
|
|
1,172,242
|
|
|
Lawsuit settlement
|
|
|
-
|
|
|
|
400,000
|
|
|
|
|
|
|
|
|
|
|
|
|
Total Operating
Expense
|
|
$
|
4,195,882
|
|
|
$
|
4,307,756
|
|
The
main reasons for the overall decrease in operating expenses in 2015 are the substantial decrease in stock based compensation from
2014 to 2015 and the absence of a lawsuit settlement in 2015. Ignoring those items, operating expenses increased approximately
32% as a result of building a solid base for future growth.
Within
the remaining operating expenses, there were a variety of increases. Salaries, wages and benefits increased as a result of additional
staff in 2015, as well as bonuses to executive officers totaling $195,000. We expect additional increases in compensation in 2016
as we hire additional high-level staff, as well as the impact of a full year of 2015 hires and the payout and severance benefits
we are required to pay to our prior CEO. Professional fees increased as a result of the litigation in process, primarily related
to the PDR/LDM lawsuit. We expect a continued increase in professional fees in 2016 as a result of increased activity on our litigation.
Advertising and promotion increased as a result of our sponsorship of e-Coupon conferences. Development and maintenance increased
as a result of expansion of our system capacity and capabilities and the move to a more robust Oracle database. Exclusivity fees
of $250,000 in 2015 resulted from the initial payment related to the Allscripts Touchworks integration. An additional $650,000
is due under the Allscripts Touchworks agreement when e-Coupon functionality becomes widely available within the Touchworks platform,
which is expected to be in early 2017.
Net
Loss
We
finished the year ended December 31, 2015 with a loss of approximately $600,000, as compared to a loss of approximately $1.0 million
during the year ended December 31, 2014. The reasons for specific components are discussed above. Overall, we had an increase
in revenue and resulting gross margin as well as decreased operating expenses. In addition, the loss in both periods included
noncash items. We had approximately $600,000 of stock based compensation and $335,000 of depreciation and amortization in 2015,
and approximately $1.2 million related to stock based compensation and approximately $265,000 of depreciation and amortization
in 2014. Excluding noncash expenses, we would have been profitable in both years. In addition, ignoring working capital changes,
we had positive cash flow from operations in both years - approximately $325,000 during the year ended December 31, 2015 and approximately
$400,000 during the year ended December 31, 2014.
Quarterly
Financial Information
Following
is our quarterly operating results for 2015 for information purposes.
|
|
|
First
|
|
|
Second
|
|
|
Third
|
|
|
Fourth
|
|
|
Total
|
|
|
|
|
Quarter
|
|
|
Quarter
|
|
|
Quarter
|
|
|
Quarter
|
|
|
Year
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Revenues
|
|
$
|
1,487,553
|
|
|
$
|
1,705,457
|
|
|
$
|
2,007,409
|
|
|
$
|
2,020,259
|
|
|
$
|
7,220,678
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Revenue Share Expense
|
|
|
756,440
|
|
|
|
882,327
|
|
|
|
1,044,415
|
|
|
|
929,020
|
|
|
|
3,622,203
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gross Profit
|
|
|
731,113
|
|
|
|
823,130
|
|
|
|
962,994
|
|
|
|
1,081,239
|
|
|
|
3,598,475
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating Expenses
|
|
|
842,610
|
|
|
|
980,659
|
|
|
|
875,425
|
|
|
|
1,497,188
|
|
|
|
4,195,882
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Income (Loss) from Operations
|
|
|
(111,497
|
)
|
|
|
(157,529
|
)
|
|
|
87,569
|
|
|
|
(415,949
|
)
|
|
|
(597,407
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other income
|
|
|
296
|
|
|
|
304
|
|
|
|
368
|
|
|
|
1,299
|
|
|
|
2,267
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss before Taxes
|
|
|
(111,201
|
)
|
|
|
(157,225
|
)
|
|
|
87,937
|
|
|
|
(414,650
|
)
|
|
|
(595,140
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Provision for Taxes
|
|
|
-0-
|
|
|
|
-0-
|
|
|
|
-0-
|
|
|
|
-0-
|
|
|
|
-0-
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net Income (Loss)
|
|
|
(111,201
|
)
|
|
|
(157,225
|
)
|
|
|
87,937
|
|
|
|
(414,650
|
)
|
|
|
(595,140
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss per share
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic and Diluted
|
|
|
(0.00
|
)
|
|
|
(0.01
|
)
|
|
|
0.00
|
|
|
|
(0.01
|
)
|
|
|
(0.02
|
)
|
Liquidity
and Capital Resources
As
of March 31, 2016, we had total current assets of approximately $9.9 million, compared with current liabilities of approximately
$2.6 million, resulting in working capital of approximately $7.3 million and a current ratio of approximately 3.8 to 1, similar
to the working capital of approximately $7.8 million and current ratio of 3.3 to 1 at December 31, 2015.
Our
cash flow for the quarter ended March 31, 2016 was negatively impacted by a one-time payment of $720,415 to our previous CEO in
lieu of issuance of approximately 595,000 common shares due to him from prior years. This payment impacts two sections of the
statement of cash flows. As discussed in more detail below, a portion of these shares were reflected in accounts payable and payment
of that portion affects cash flow from operations. The remaining portion was reflected in stock payable in the equity section
and payment affected cash used in financing activities.
Following
is a table with summary data from the consolidated statement of cash flows for the quarter ended March 31, 2016, as presented
and after removing the effect of this payment.
|
|
|
As
presented
|
|
|
Adjusted
to
remove
effect of
one-time
payment
|
|
|
Net cash provided by (used
in) operating activities
|
|
$
|
(286,087
|
)
|
|
$
|
76,913
|
|
|
Net cash used in investing activities
|
|
|
(15,802
|
)
|
|
|
(15,802
|
)
|
|
Net cash provided
by (used in) financing activities
|
|
|
(357,415
|
)
|
|
|
-
|
|
|
Net increase
(decrease) in cash and cash equivalents
|
|
$
|
(659,304
|
)
|
|
$
|
61,111
|
|
Our
operating activities used approximately $286,000 in cash flow during the three months ended March 31, 2016, compared with cash
generated of approximately $86,000 in the same period in 2015. This decrease resulted from the payment of $363,000 to our previous
CEO to extinguish the accounts payable related to the purchase of a patent from the CEO in 2010. Excluding that one-time payment,
we would have had positive cash flow from operations of approximately $76,000.
We
used approximately $16,000 in investing activities in the three months ended March 31, 2016 compared with approximately $23,000
in the same period in 2015. These investment activities relate to improvements implemented in our SampleMD website, equipment
purchases, and expansion of our patent portfolio.
We
used approximately $357,000 in financing activities by retiring stock payable due to our previous CEO. These shares were due as
a result of previously granted stock awards in 2014 and 2015, for which shares had not yet been issued. These shares were recorded
as stock payable on the balance sheet at December 31, 2015.
As
of December 31, 2015, we had total current assets of approximately $11.1 million, compared with current liabilities of approximately
$3.4 million, resulting in working capital of approximately $6.7 million and a current ratio of approximately 3.3 to 1. This significant
improvement over the working capital balance of approximately $3.2 million and the current ratio of 2.3 to 1 at December 31, 2014
is largely a result of our financing transaction with WPP. We are currently generating positive cash flow from operations and
we expect our working capital balance to continue to improve in future quarters.
Our
operating activities generated approximately $500,000 in the year ended December 31, 2015 as comparted with approximately $50,000
used by operating activities in the year ended December 31, 2014. The positive cash flow from operations in the year ended December
31, 2015 resulted from positive cash flow from operations prior to working capital items of approximately $350,000 and approximately
$250,000 of working capital improvements. We expect to have positive cash flow from operations in future quarters in 2016, however
severance payments due at the end of March 2016, may make that unlikely in Q1.
We
used approximately $400,000 in investing activities in the year ended December 31, 2014 compared with about $100,000 in the year
ended December 31, 2015. These investment activities relate to improvements being implemented in our SampleMD website, as well
as protection and expansion of our patent portfolio. These items both represent important components of our business strategy
moving forward. The 2014 was significantly higher as a result of the launch of our upgraded SampleMD 2.0 platform in early 2014,
as well as the initial work on our migration to Oracle Software started in 2014 and completed in 2015.
Financing
activities provided approximately $4.3 million in the year ended December 31, 2015, resulting from the strategic investment of
approximately $4.7 million by WPP, net of costs associated with the investment. Financing activities provided approximately $2.8
million during the year ended December 31, 2014. This results from a $10 million equity raise in March 2014, partially offset
by costs of the raise and redemption of all the common stock, preferred stock, and warrants held by a major shareholder that significantly
reduced the potential fully diluted shares count, even when considering the new equity issued. With the financing and cash on
hand, we have sufficient cash to operate our business for more than the next twelve months and we do not anticipate the need to
raise additional equity for operating purposes.
Off
Balance Sheet Arrangements
As
of March 31, 2016, there were no off balance sheet arrangements.
Critical
Accounting Policies
In
December 2001, the SEC requested that all registrants list their most “critical accounting polices” in the Management
Discussion and Analysis. The SEC indicated that a “critical accounting policy” is one which is both important to the
portrayal of a company’s financial condition and results, and requires management’s most difficult, subjective or
complex judgments, often as a result of the need to make estimates about the effect of matters that are inherently uncertain.
Our accounting policies are discussed in the footnotes to our financial statements included in our annual report on Form 10-K
for the year ended December 31, 2015, however we consider our critical accounting policies to be those related to the amount of
revenue to be billed, the timing of revenue recognition, calculation of revenue share expense, stock-based compensation, capitalization
and related amortization of intangible assets, and impairment of assets.
Recently
Issued Accounting Pronouncements
The
Company does not expect the adoption of recently issued accounting pronouncements to have a significant impact on the Company’s
results of operation, financial position or cash flow.
CERTAIN
RELATIONSHIPS AND RELATED TRANSACTIONS
Other
than described below or the transactions described under the heading “Executive Compensation” (or with respect to
which such information is omitted in accordance with SEC regulations), there have not been, and there is not currently proposed,
any transaction or series of similar transactions to which we were or will be a participant in which the amount involved exceeded
or will exceed the lesser of $120,000 or one percent of the average of our total assets at year-end for the last three completed
fiscal years, and in which any director, executive officer, holder of 5% or more of any class of our capital stock or any member
of the immediate family of any of the foregoing persons had or will have a direct or indirect material interest.
During
the year ended December 31, 2015, WPP made a strategic investment in the Company and is a shareholder that owns approximately
20% of the shares of the Company. During 2015, we had billings of $420,503 to agencies that are part of the WPP group and recognized
revenue of $178,855 related to those billings. As of December 31, 2015, we have receivables included in trade receivables on the
balance sheet of $381,125 from WPP agencies and amounts due to WPP agencies included in revenue share payable of $37,803 as of
December 31, 2015.
On
September 24, 2015, we entered into an Investor Rights Agreement with WPP Luxembourg (the “Rights Agreement”), pursuant
to which we agreed to the following:
|
|
●
|
Demand
Registration Rights. We granted the Investor registration rights for the 6,011,106 acquired
from us and any securities acquired in connection with an Amended and Restated Co-Marketing
Agreement after a period of two years.
|
|
|
●
|
Inspection
Rights. So long as the Investor owns not less than 25% of the Shares, we granted the
Investor an annual right to inspect our books and records.
|
|
|
●
|
Observer
Rights. So long as the Investor owns not less than 25% of the Shares, we will allow the
Investor to choose a representative to attend our board meetings as a nonvoting observer.
|
|
|
●
|
Board
Seat. So long as the Investor owns not less than 25% of the Shares, we agreed to appoint
a nominee of the Investor as a member of our board of directors. We also agreed to a
five member Board of Directors provided that it is not prohibited by the rules and regulations
of an exchange that we trade on. We also agreed to enter into an Indemnity Agreement
with the nominee.
|
|
|
●
|
Budget
Review. So long as the Investor owns not less than 25% of the Shares, we agreed to review
our budget plans with the Investor’s nominee prior to submission to the Board of
Directors, at the request of the Investor.
|
|
|
●
|
Right
of First Refusal. We agreed that, in the event that it proposes to sell new securities,
we will first offer such new securities to the Investor.
|
|
|
●
|
Special
Approval Matters. So long as the Investor owns not less than 25% of the Shares, and provided
that it is not prohibited by the rules and regulations of an exchange that we trades
on, we agreed that 80% Board approval will be required for certain decisions, including:
|
|
|
●
|
the
incurrence of any indebtedness in excess of $1.5 million in the aggregate during any
fiscal year
|
|
|
●
|
the
sale, transfer or other disposition of all or substantially all of our assets;
|
|
|
●
|
the
acquisition of any assets or properties (in one or more related transactions) for cash
or otherwise for an amount in excess of $1.5 million in the aggregate during any fiscal
year;
|
|
|
●
|
capital
expenditures in excess of $1.5 million individually (or in the aggregate if related to
an integrated program of activities) or in excess of $1.5 million in the aggregate during
any fiscal year;
|
|
|
●
|
making,
or permitting any subsidiary to make, loans to, investments in, or purchasing, or permitting
any subsidiary to purchase, any stock or other securities in another corporation, joint
venture, partnership or other entity;
|
|
|
●
|
the
commencement or settlement of any lawsuit, arbitration or other legal proceeding related
to our intellectual property or involving an amount in controversy greater than $1.5
million; and
|
|
|
●
|
the
issuance of new securities, except for securities issued under an equity incentive plan
and any issuance of common stock to vendors, advisors, financial institutions, suppliers
or joint venturers that do not exceed, individually or in the aggregate 5% of then issued
and outstanding capital stock of the Company.
|
On
September 24, 2015, we amended and restated an existing Co-Marketing Agreement with Grey Healthcare Group, LLC (“GHG”)
an affiliate of WPP (the “Amended and Restated Co-Marketing Agreement”). The Amended and Restated Co-Marketing Agreement
was amended to give the GHG the option to receive all or part of the compensation due under the agreement in shares of our common
stock. Shares issuable under the Amended and Restated Co-Marketing Agreement will be issued to WPP or any other affiliate of GHG
designated in writing by GHG at the following rates:
|
|
●
|
Until
June 30, 2016, we will issue the number of shares of common stock equal to GHG’s
share of net revenues received for sales of new services to GHG or Company clients (“GHG
Net Revenues”) divided by $0.7875.
|
|
|
●
|
After
June 30, 2016, we will issue the number of shares of common stock equal to the GHG Net
Revenues divided by a price equal to 80% multiplied by the average trading price of one
share of common stock during the 30 trading day period immediately prior to the date
of the most recent statement of GHG Net Revenues set forth by the Company.
|
On
March 17, 2014, we raised gross proceeds of $10,000,000 in an unregistered offering (the “Offering”) with certain
accredited investors. We used a portion of the net proceeds of the Offering to exercise the Securities Redemption Option Agreement,
as amended, with Vicis Capital Master Fund (“Vicis”) that provides us with an option to purchase all of the outstanding
shares and derivative securities held by Vicis for total payment of six million dollars ($6,000,000). The shares and derivative
securities include the Series A Convertible Preferred Stock, Series B Convertible Preferred Stock, Common Stock, and warrants
to purchase shares of common stock held by Vicis in our company. In connection with this fundraising, we agreed to grant 200,000
shares of fully vested common stock to officers David, Harrell, David Lester, and Terry Hamilton.
In
February 2014, we agreed to grant 337,500 shares of common stock, half of which vested immediately and half of which vested in
August 2014, to both David Harrell and Terry Hamilton as bonuses based on their efforts to recapitalize the company to secure
approximately $3 million in working capital while reducing potential fully diluted shares by approximately 7 million shares. Stock-based
compensation related to these bonuses was $570,375 during the year ended December 31, 2014. These shares have not yet been issued
and are recorded as stock payable, but can be requested by the officers at any time.
MARKET
FOR COMMON EQUITY AND RELATED STOCKHOLDER MATTERS
Market
Information
Our
common stock is quoted under the symbol “OPRX” on the OTCQB operated by OTC Markets Group, Inc. Only a limited
market exists for our securities. There is no assurance that a regular trading market will develop, or if developed, that it will
be sustained. Therefore, a shareholder may be unable to resell his securities in our company.
The
following tables set forth the range of high and low prices for our common stock for the each of the periods indicated as reported
by the OTCQB. These quotations reflect inter-dealer prices, without retail mark-up, mark-down or commission and may not necessarily
represent actual transactions.
|
Fiscal
Year Ending December 31, 2014
|
|
Quarter
Ended
|
|
High
$
|
|
|
Low
$
|
|
|
December
31, 2014
|
|
|
1.15
|
|
|
|
0.80
|
|
|
September
30, 2014
|
|
|
1.54
|
|
|
|
1.10
|
|
|
June
30, 2014
|
|
|
1.80
|
|
|
|
1.42
|
|
|
March
31, 2014
|
|
|
1.95
|
|
|
|
1.41
|
|
|
Fiscal
Year Ending December 31, 2015
|
|
Quarter
Ended
|
|
High
$
|
|
|
Low
$
|
|
|
December
31, 2015
|
|
|
1.34
|
|
|
|
1.10
|
|
|
September
30, 2015
|
|
|
1.28
|
|
|
|
0.76
|
|
|
June
30, 2015
|
|
|
1.73
|
|
|
|
0.81
|
|
|
March
31, 2015
|
|
|
1.59
|
|
|
|
0.98
|
|
|
|
|
|
|
|
|
|
|
|
|
Quarter
Ended March 31, 2016
|
|
|
1.24
|
|
|
$
|
0.89
|
|
On June 13, 2016, the last sales price per
share of our common stock was $1.05.
Penny
Stock
The
SEC has adopted rules that regulate broker-dealer practices in connection with transactions in penny stocks. Penny stocks are
generally equity securities with a market price of less than $5.00, other than securities registered on certain national securities
exchanges or quoted on the NASDAQ system, provided that current price and volume information with respect to transactions in such
securities is provided by the exchange or system. The penny stock rules require a broker-dealer, prior to a transaction in a penny
stock, to deliver a standardized risk disclosure document prepared by the SEC, that: (a) contains a description of the nature
and level of risk in the market for penny stocks in both public offerings and secondary trading; (b) contains a description of
the broker's or dealer's duties to the customer and of the rights and remedies available to the customer with respect to a violation
of such duties or other requirements of the securities laws; (c) contains a brief, clear, narrative description of a dealer market,
including bid and ask prices for penny stocks and the significance of the spread between the bid and ask price; (d) contains a
toll-free telephone number for inquiries on disciplinary actions; (e) defines significant terms in the disclosure document or
in the conduct of trading in penny stocks; and (f) contains such other information and is in such form, including language, type
size and format, as the SEC shall require by rule or regulation.
The
broker-dealer also must provide, prior to effecting any transaction in a penny stock, the customer with (a) bid and offer quotations
for the penny stock; (b) the compensation of the broker-dealer and its salesperson in the transaction; (c) the number of shares
to which such bid and ask prices apply, or other comparable information relating to the depth and liquidity of the market for
such stock; and (d) a monthly account statement showing the market value of each penny stock held in the customer's account.
In
addition, the penny stock rules require that prior to a transaction in a penny stock not otherwise exempt from those rules, the
broker-dealer must make a special written determination that the penny stock is a suitable investment for the purchaser and receive
the purchaser's written acknowledgment of the receipt of a risk disclosure statement, a written agreement as to transactions involving
penny stocks, and a signed and dated copy of a written suitability statement.
These
disclosure requirements may have the effect of reducing the trading activity for our common stock. Therefore, stockholders may
have difficulty selling our securities.
Holders
of Our Common Stock
As
of June 2, 2016, we had 29,077,660 shares of our common stock issued and outstanding, held by 320 shareholders of record at our
transfer agent, with holding our shares in street name.
Dividends
We
currently intend to retain future earnings for the operation of our business. We have never declared or paid cash dividends on
our common stock, and we do not anticipate paying any cash dividends in the foreseeable future.
In
the event that a dividend is declared, common stockholders on the record date are entitled to share ratably in any dividends that
may be declared from time to time on the common stock by our board of directors from funds legally available.
There
are no restrictions in our articles of incorporation or bylaws that restrict us from declaring dividends. The Nevada Revised Statutes,
however, do prohibit us from declaring dividends where, after giving effect to the distribution of the dividend:
|
|
1.
|
We
would not be able to pay our debts as they become due in the usual course of business; or
|
|
|
2.
|
Our
total assets would be less than the sum of our total liabilities, plus the amount that would be needed to satisfy the rights
of shareholders who have preferential rights superior to those receiving the distribution.
|
Securities
Authorized for Issuance under Equity Compensation Plans
On
June 13, 2013, our Board of Directors adopted the 2013 Equity Incentive Plan (the “Plan”). The purpose of the Plan
is to attract and retain the best available personnel for positions of substantial responsibility with us, to provide additional
incentive to employees, directors and consultants, and to promote our success. Under the initial Plan, we were able to issue up
to an aggregate total of 1,500,000 incentive or non-qualified options to purchase our common stock, or stock awards. In March,
2016, the Board expanded the number of shares issuable under the plan to 4,000,000.
Equity
Compensation Plans as of December 31, 2015
Equity
Compensation
Plans Not Approved by
the Shareholders
|
|
Number of
Securities to be issued
upon
exercise of
outstanding
options
|
|
|
Weighted-
average
exercise
price of
outstanding
options
|
|
|
Number of
Securities
remaining
available
for future
issuance
under
equity compensation
plans
|
|
|
|
|
(a)
|
|
|
(b)
|
|
|
(c)
|
|
|
2013
Equity Compensation Plan
|
|
|
1,085,000
|
|
|
$
|
1.17
|
|
|
|
–
|
|
|
Other
Equity Compensation (includes options and warrants)
|
|
|
2,609,139
|
|
|
$
|
1.54
|
|
|
|
–
|
|
|
Total
|
|
|
3,709,583
|
|
|
$
|
1.41
|
|
|
|
410,000
|
|
Recent
Sales of Unregistered Securities
In
April 2016, we issued 34,235 shares of common stock in connection with the cashless exercise of an expiring option to purchase
100,000 shares of common stock previously granted to a consultant.
In
March 2016, we issued 12,500 shares of common stock to our independent directors in connection with our Director Compensation
Plan.
In
December, 2015, we issued 12,500 shares of restricted common stock to our outside Directors as part of our director compensation
package for services rendered in the fourth quarter of 2015.
On
September 24, 2015, we entered into a Stock Purchase Agreement with WPP Luxembourg Gamma Three S.à r.l., a private limited
liability company (société à responsabilité limitée) incorporated under the laws of the Grand
Duchy of Luxembourg, pursuant to which we sold to WPP 6,011,106 shares of our common stock for $0.7875 per share, or gross proceeds
of $4,733,746. Placement agents in the offering received warrants to purchase up to 240,444 shares of our common stock with an
exercise price of $0.7875 per share and a term of 5 years.
In
September, 2015, we issued 6,250 shares of our common stock to each of our independent directors under our compensation plan.
In
September, 2015, we issued 45,000 shares of our common stock under a capital markets advisory agreement.
In
February 2015 we issued 45,000 shares of our common stock to an investment banking firm in connection with a capital markets advisory
agreement.
We
also issued 12,500 shares of common stock to our outside directors in connection with our Director Compensation Plan.
In
January 2015, we issued 12,500 shares of restricted common stock to our outside Directors as part of our director compensation
package for services rendered in the fourth quarter of 2014.
These
securities were issued pursuant to Section 4(2) of the Securities Act and/or Rule 506 promulgated thereunder. The holders represented
their intention to acquire the securities for investment only and not with a view towards distribution. The investors were given
adequate information about us to make an informed investment decision. We did not engage in any general solicitation or advertising.
We directed our transfer agent to issue the stock certificates with the appropriate restrictive legend affixed to the restricted
stock.
EXECUTIVE
COMPENSATION
The
table below summarizes all compensation awarded to, earned by, or paid to our former or current executive officers for the fiscal
years ended December 31, 2015 and 2014.
Name
and
principal
position
|
|
|
Year
|
|
|
|
Salary
($)
|
|
|
|
Bonus
($)
|
|
|
Stock
Awards
($)
|
|
Option
Awards
($)
|
|
All
Other
Compensation
($)
|
|
|
Total
($)
|
|
David
A. Harrell
Chairman, Chief Executive Officer,
Chief Strategic Officer and Director
|
|
|
2015
2014
|
|
|
|
201,508
183,750
|
|
|
|
75,000
100,000
|
|
|
73,509
394,969
|
|
|
|
|
|
|
350,017
678,719
|
|
Terence
J. Hamilton
VP of Sales and Director
|
|
|
2015
2014
|
|
|
|
165,000
163,438
|
|
|
|
60,000
70,000
|
|
|
110,264
458,906
|
|
|
|
|
|
|
335,264
692,344
|
|
|
Douglas
Baker
CFO
|
|
|
2015
2014
|
|
|
|
143,750
78,125
|
|
|
|
60,000
|
|
|
|
|
10,668
131,110
|
|
|
|
|
214,418
209,235
|
|
Narrative
Disclosure to the Summary Compensation Table
On
June 1, 2008, we entered into an employment agreement with Mr. Harrell to serve as our CEO. The agreement was amended on January
14, 2013 to account for his new positions as CSO and Vice Chairman. The terms of his compensation, was an annual salary of $144,000
with a 5% cost of living increase on each 12 month anniversary. Mr. Harrell was also eligible for additional quarterly and annual
bonus compensation, stock options, and stock grants based on performance metrics outlined by our board of directors. He was entitled
to vacation and sick days, and other benefits included in the agreement. On March 18, 2010, we entered into an addendum to the
employment agreement to increase his compensation to $152,004 annually.
On
July 28, 2010, we amended Mr. Harrell’s employment agreement to include a covenant not to compete covering the term of employment
and continuing for a period of two years thereafter. As a result of the same amendment, Mr. Harrell is entitled to severance payments
if his employment terminated, with or without cause. Such payments would be due monthly at his then current salary rate for a
period of 24 months following termination. On August 14, 2013, we further amended the employment agreement with Mr. Harrell. Pursuant
to the terms and conditions of the Amendment to Employment Agreement with David Harrell:
|
|
●
|
Mr.
Harrell will serve as Vice Chairman of the Board and Chief Strategy Officer of our company;
|
|
|
|
|
|
|
●
|
The
term of Mr. Harrell’s employment shall be for one year, and shall automatically
renew for each year thereafter unless terminated on thirty days’ notice before
the end of the term; and
|
|
|
|
|
|
|
●
|
Mr.
Harrell will earn a base salary of $183,750 per year;
|
On
August 14, 2013 we granted restricted stock awards under our 2013 Incentive Plan. Mr. Harrell was awarded 121,875 shares of our
common stock. The award vested in 2014 and was valued at $1.69 per share. Mr. Harrell was granted an additional restricted stock
award of 100,000 shares under our 2013 Incentive Plan on January 9, 2014. This award was fully vested at the time of grant and
was valued at $1.89 per share. In 2015, Mr. Harrell was awarded an additional 79,042 shares of restricted common stock, valued
at $0.93 per share. All shares described in this paragraph remain unissued and are recorded as common stock payable at December
31, 2015.
On
May 9, 2016, we entered into a Separation Agreement and Release with Mr. Harrell that grants us a standard release of employment
claims in consideration for, among other things, a stock payout of $720,415 to Mr. Harrell to retire shares due and owing to him,
awarded in prior years, but not yet issued.
On
the same date, we entered into a Corporate Consulting Agreement with Mr. Harrell that sets forth the terms his continued relationship
with our company. He remained our employee through May 31, 2016 and the Corporate Consulting Agreement went effective as of June
1, 2016. Under the terms of this agreement, Mr. Harrell will consult for our company for a period of 16 months and he will receive
a monthly payment of $15,000, with the potential for up to $54,000 in additional bonus payments during the term of the agreement.
This agreement also calls for insurance benefits for seven months. Finally, the agreement contains a Consultant Confidentiality,
Invention Assignment and Non-Compete Agreement that contains restrictive covenants that include a one year non-compete following
the completion of Mr. Harrell’s 18 months of consulting, and an inventions assignment clause during the term of his consulting
relationship.
On
August 1, 2008, we entered into an employment agreement with Mr. Hamilton to serve as our VP of Sales. Under the agreement, we
agreed to compensate Mr. Hamilton $120,000 annually and we granted him options to purchase 150,000 shares of our common stock
in 2009. Mr. Hamilton is also eligible for additional quarterly and annual bonus compensation, stock options, and stock grants
based on performance metrics outlined by our board of directors. He is entitled to vacation and sick days, and other benefits
included in the agreement. On March 18, 2010, we entered into an addendum to the employment agreement to increase his compensation
to $150,000 annually.
On
July 28, 2010, we amended Mr. Hamilton’s employment agreement to include a covenant not to compete covering the term of
employment and continuing for a period of one year thereafter. As a result of the same amendment, Mr. Hamilton is entitled to
severance payments if he is terminated with or without cause. Such payments would be due monthly at his then current salary rate
for a period of 12 months following termination. On August 14, 2013, we amended the employment agreement with Mr. Hamilton to
increase his base salary to $157,500 per year.
On
March 16, 2014, Mr. Hamilton’s salary was increased to a base salary of $165,000, and on February 1, 2016, his base salary
was increased to $181,500, but no formal contract amendment was signed, in either instance.
On
August 14, 2013 we granted restricted stock awards under our 2013 Incentive Plan. Mr. Hamilton was awarded 215,625 shares of our
common stock. The award vested in 2014 and was valued at $1.69 per share. Mr. Hamilton was granted an additional restricted stock
award of 50,000 shares under our 2013 Incentive Plan on January 9, 2014. This award was fully vested at the time of grant and
was valued at $1.89 per share. In 2015, Mr. Hamilton was awarded an additional 118,563 shares of restricted common stock, valued
at $0.93 per share. All shares described in this paragraph remain unissued and are recorded as common stock payable at December
31, 2015.
On
May 12, 2014, we entered into an employment agreement with Mr. Baker, our Chief Financial Officer. Under the agreement, we agreed
to compensate Mr. Baker $125,000 annually and we granted him options to purchase 100,000 shares of our common stock, with 50%
vesting after one year and 50% vesting after two years of hire. The options were valued at 1.3111 per share, or a total of $131,110,
for financial statement purposes using the Black-Scholes pricing model. Effective April 1, 2015, Mr. Baker’s salary was
increased to an annual rate of $150,000, and effective February 1, 2016 to an annual rate of $165,000. During 2015, Mr. Baker
was granted an additional 100,000 options that vest in 2017.
Mr.
Baker is also eligible for additional quarterly and annual bonus compensation, stock options, and stock grants based on performance
metrics outlined by our board of directors. He is entitled to vacation and sick days, and other benefits included in the agreement.
Outstanding
Equity Awards at Fiscal Year-End
The
table below summarizes all unexercised options, stock that has not vested, and equity incentive plan awards for each named executive
officers as of December 31, 2015.
|
OUTSTANDING
EQUITY AWARDS AT FISCAL YEAR-END
|
|
OPTION
AWARDS
|
|
STOCK
AWARDS
|
|
|
Name
|
|
Number
of
Securities
Underlying
Unexercised
Options
(#)
Exercisable
|
|
|
Number
of
Securities
Underlying
Unexercised
Options
(#)
Unexercisable
|
|
|
Equity
Incentive
Plan
Awards:
Number
of
Securities
Underlying
Unexercised
Unearned
Options
(#)
|
|
|
Option
Exercise
Price
($)
|
|
|
Option
Expiration
Date
|
|
Number
of
Shares
or Units
of
Stock That
Have
Not
Vested
(#)
|
|
|
Market
Value
of
Shares
or
Units
of
Stock
That
Have
Not
Vested
($)
|
|
|
Equity
Incentive
Plan
Awards:
Number
of
Unearned
Shares,
Units
or
Other
Rights
That Have
Not
Vested
(#)
|
|
|
Equity
Incentive
Plan
Awards:
Market
or
Payout
Value
of
Unearned
Shares,
Units or
Other
Rights
That
Have Not
Vested
(#)
|
|
|
David
Harrell
|
|
|
46,850
|
|
|
|
|
|
|
|
|
|
|
$
|
1.00
|
|
|
5/31/16
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Douglas
|
|
|
50,000
|
|
|
|
50,000
|
|
|
|
|
|
|
$
|
1.05
|
|
|
5/19/19
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Baker
|
|
|
0
|
|
|
|
100,000
|
|
|
|
|
|
|
$
|
1.05
|
|
|
6/24/20
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Terry Hamilton
|
|
|
122,000
|
|
|
|
|
|
|
|
|
|
|
$
|
1.00
|
|
|
5/31/16
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Director
Compensation
The
table below summarizes all compensation of our directors as of December 31, 2015.
|
Name
|
|
Fees
Earned or Paid in Cash
($)
|
|
|
Stock
Awards
($)
|
|
|
Option
Awards
($)
|
|
|
All
Other Compensation
($)
|
|
|
Total
($)
|
|
|
Gus D. Halas
|
|
|
25,000
|
|
|
|
11,827
|
|
|
|
|
|
|
|
|
|
|
|
21,813
|
|
|
Jack Pinney
|
|
|
25,000
|
|
|
|
11,315
|
|
|
|
|
|
|
|
|
|
|
|
20,894
|
|
|
Lynn Vos
|
|
|
-0-
|
|
|
|
-0-
|
|
|
|
|
|
|
|
|
|
|
|
-0-
|
|
Narrative
Disclosure to the Director Compensation Table
Pursuant
to our Director Compensation Plan, independent directors (“Outside Directors”) shall receive (a) a $25,000 annual
cash retainer, payable in equal quarterly installments, and (b) reimbursement for expenses related to Board meeting attendance
and any committee participation. Directors are expected to attend four meetings per year as well as spend an additional 10 –
20 hours per month on company matters. In addition, Outside Directors shall receive 25,000 shares of Common Stock, payable in
equal quarterly installments, which shall vest immediately. Directors that are also employees of our company shall not receive
additional compensation for serving on the Board. Both the cash retainer and stock awards are prorated for partial quarters of
service when a new Director joins the Board.
FINANCIAL
STATEMENTS
Index
to Financial Statements Required by Article 8 of Regulation S-X:
OPTIMIZERx
CORPORATION
CONDENSED
CONSOLIDATED BALANCE SHEETS (UNAUDITED)
AS
OF MARCH 31, 2016 AND DECEMBER 31, 2015
|
|
|
March
31, 2016
|
|
|
December
31, 2015
|
|
|
ASSETS
|
|
|
|
|
|
|
|
Current Assets
|
|
|
|
|
|
|
|
Cash
and cash equivalents
|
|
$
|
7,548,261
|
|
|
$
|
8,207,565
|
|
|
Accounts
receivable
|
|
|
2,340,765
|
|
|
|
2,847,450
|
|
|
Prepaid
expenses
|
|
|
54,555
|
|
|
|
70,623
|
|
|
Total
Current Assets
|
|
|
9,943,581
|
|
|
|
11,125,638
|
|
|
Property
and equipment, net
|
|
|
23,076
|
|
|
|
10,239
|
|
|
Other
Assets
|
|
|
|
|
|
|
|
|
|
Patent
rights, net
|
|
|
817,239
|
|
|
|
832,884
|
|
|
Web
development costs, net
|
|
|
308,812
|
|
|
|
340,470
|
|
|
Security
deposit
|
|
|
5,049
|
|
|
|
5,049
|
|
|
Total
Other Assets
|
|
|
1,131,100
|
|
|
|
1,178,403
|
|
|
TOTAL
ASSETS
|
|
$
|
11,097,757
|
|
|
$
|
12,314,280
|
|
|
|
|
|
|
|
|
|
|
|
|
LIABILITIES
AND STOCKHOLDERS' EQUITY
|
|
|
|
|
|
|
|
|
|
Current
Liabilities
|
|
|
|
|
|
|
|
|
|
Accounts
payable - trade
|
|
$
|
195,076
|
|
|
$
|
212,191
|
|
|
Accounts
payable - related party
|
|
|
-
|
|
|
|
570,000
|
|
|
Accrued
expenses
|
|
|
8,090
|
|
|
|
6,983
|
|
|
Revenue share payable
|
|
|
1,502,816
|
|
|
|
2,355,608
|
|
|
Deferred
revenue
|
|
|
845,226
|
|
|
|
227,002
|
|
|
Total
Liabilities
|
|
|
2,551,208
|
|
|
|
3,371,784
|
|
|
Stockholders'
Equity
|
|
|
|
|
|
|
|
|
|
Common
stock, $.001 par value, 500,000,000 shares authorized, 29,043,425 and 29,030,925 shares issued and outstanding, respectively
|
|
|
29,043
|
|
|
|
29,031
|
|
|
Preferred
stock, $.001 par value, 10,000,000 shares authorized, 0 and 65 shares issued and outstanding, respectively
|
|
|
-
|
|
|
|
-
|
|
|
Stock
warrants
|
|
|
2,294,416
|
|
|
|
2,329,508
|
|
|
Additional
paid-in-capital
|
|
|
32,631,226
|
|
|
|
32,185,499
|
|
|
Stock
payable
|
|
|
663,670
|
|
|
|
1,132,148
|
|
|
Deferred
stock compensation
|
|
|
-
|
|
|
|
(13,800
|
)
|
|
Accumulated
deficit
|
|
|
(27,071,806
|
)
|
|
|
(26,719,890
|
)
|
|
Total
Stockholders' Equity
|
|
|
8,546,549
|
|
|
|
8,942,496
|
|
|
TOTAL
LIABILITIES AND STOCKHOLDERS' EQUITY
|
|
$
|
11,097,757
|
|
|
$
|
12,314,280
|
|
The
accompanying notes are an integral part of these financial statements.
OPTIMIZERx
CORPORATION
CONDENSED
CONSOLIDATED STATEMENTS OF OPERATIONS (UNAUDITED)
FOR
THE THREE MONTHS ENDED MARCH 31, 2016 AND 2015
|
|
For
the Three Months
Ended March 31
|
|
|
|
|
2016
|
|
|
2015
|
|
|
|
|
|
|
|
|
|
|
NET
REVENUE
|
|
$
|
1,759,528
|
|
|
$
|
1,487,553
|
|
|
|
|
|
|
|
|
|
|
|
|
COST
OF SALES
|
|
|
892,793
|
|
|
|
756,440
|
|
|
|
|
|
|
|
|
|
|
|
|
GROSS
MARGIN
|
|
|
866,735
|
|
|
|
731,113
|
|
|
|
|
|
|
|
|
|
|
|
|
OPERATING
EXPENSES
|
|
|
1,228,564
|
|
|
|
842,610
|
|
|
|
|
|
|
|
|
|
|
|
|
LOSS
FROM OPERATIONS
|
|
|
(361,829
|
)
|
|
|
(111,497
|
)
|
|
|
|
|
|
|
|
|
|
|
|
OTHER
INCOME (EXPENSE)
|
|
|
|
|
|
|
|
|
|
Interest
income
|
|
|
10,076
|
|
|
|
296
|
|
|
Interest
Expense
|
|
|
(163
|
)
|
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL
OTHER INCOME (EXPENSE)
|
|
|
9,913
|
|
|
|
296
|
|
|
|
|
|
|
|
|
|
|
|
|
LOSS
BEFORE PROVISION FOR INCOME TAXES
|
|
|
(351,916
|
)
|
|
|
(111,201
|
)
|
|
|
|
|
|
|
|
|
|
|
|
PROVISION
FOR INCOME TAXES
|
|
|
-
|
|
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
NET
LOSS
|
|
$
|
(351,916
|
)
|
|
$
|
(111,201
|
)
|
|
|
|
|
|
|
|
|
|
|
|
WEIGHTED AVERAGE
SHARES OUTSTANDING
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
BASIC
AND DILUTED
|
|
|
29,030,925
|
|
|
|
22,897,819
|
|
|
|
|
|
|
|
|
|
|
|
|
NET LOSS PER SHARE
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
BASIC
AND DILUTED
|
|
$
|
(0.01
|
)
|
|
$
|
(0.00
|
)
|
The
accompanying notes are an integral part of these financial statements.
OPTIMIZERx
CORPORATION
CONDENSED
CONSOLIDATED STATEMENTS OF CASH FLOWS (UNAUDITED)
FOR
THE THREE MONTHS ENDED MARCH 31, 2016 AND 2015
|
|
|
For
the three months
Ended March 31
|
|
|
|
|
2016
|
|
|
2015
|
|
|
CASH
FLOWS FROM OPERATING ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
Net
loss for the period
|
|
$
|
(351,916
|
)
|
|
$
|
(111,201
|
)
|
|
Adjustments
to reconcile net loss to net cash provided by (used in) operating activities:
|
|
|
|
|
|
|
|
|
|
Depreciation
and amortization
|
|
|
50,268
|
|
|
|
79,668
|
|
|
Stock
and options issued for services
|
|
|
106,384
|
|
|
|
79,591
|
|
|
Changes
in:
|
|
|
|
|
|
|
|
|
|
Accounts
receivable
|
|
|
506,685
|
|
|
|
286,840
|
|
|
Prepaid
expenses
|
|
|
16,068
|
|
|
|
(10,206
|
)
|
|
Accounts
payable
|
|
|
(380,115
|
)
|
|
|
(37,241
|
)
|
|
Revenue share payable
|
|
|
(852,792
|
)
|
|
|
(339,459
|
)
|
|
Accrued
expenses
|
|
|
1,107
|
|
|
|
5,392
|
|
|
Deferred
revenue
|
|
|
618,224
|
|
|
|
133,308
|
|
|
NET
CASH PROVIDED BY (USED IN) OPERATING ACTIVITIES
|
|
|
(286,087
|
)
|
|
|
86,692
|
|
|
|
|
|
|
|
|
|
|
|
|
CASH
FLOWS FROM INVESTING ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
Patent
rights
|
|
|
(1,295
|
)
|
|
|
(3,042
|
)
|
|
Equipment
|
|
|
(14,507
|
)
|
|
|
-
|
|
|
Website
site development costs
|
|
|
-
|
|
|
|
(19,800
|
)
|
|
NET
CASH USED IN INVESTING ACTIVITIES
|
|
|
(15,802
|
)
|
|
|
(22,842
|
)
|
|
|
|
|
|
|
|
|
|
|
|
CASH
FLOWS FROM FINANCING ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
Repurchase
of common stock payable
|
|
|
(357,415
|
)
|
|
|
-
|
|
|
NET
CASH USED IN FINANCING ACTIVITIES
|
|
|
(357,415
|
)
|
|
|
-
|
|
|
NET
INCREASE (DECREASE) IN CASH AND CASH EQUIVALENTS
|
|
|
(659,304
|
)
|
|
|
63,850
|
|
|
CASH
AND CASH EQUIVALENTS - BEGINNING OF PERIOD
|
|
|
8,207,565
|
|
|
|
3,446,973
|
|
|
CASH
AND CASH EQUIVALENTS - END OF PERIOD
|
|
$
|
7,548,261
|
|
|
$
|
3,510,823
|
|
|
|
|
|
|
|
|
|
|
|
|
SUPPLEMENTAL
CASH FLOW INFORMATION:
|
|
|
|
|
|
|
|
|
|
Cash
paid for interest
|
|
$
|
163
|
|
|
$
|
-
|
|
|
Cash
paid for income taxes
|
|
$
|
-
|
|
|
$
|
-
|
|
The
accompanying notes are an integral part of these financial statements.
OPTIMIZERx
CORPORATION
NOTES
TO THE CONDENSED CONSOLIDATED FINANCIAL STATEMENTS (UNAUDITED)
MARCH
31, 2016
NOTE
1 – NATURE OF BUSINESS AND BASIS OF PRESENTATION
We
are a technology solution company focused on the health care industry. Our objective is to bring better access to better care
by leveraging our proprietary technology to provide on demand savings and clinical messaging within physicians’ and patients’
web based platforms, including Electronic Health Records, e-prescribing platforms, pharmacies and Patient Portals. Initially defined
as a marketing and advertising company through its consumer website, OptimizeRx.com, we have matured as a technology solutions
provider through our direct to physician solutions, which allows physicians to automatically display and distribute sample vouchers
and/or co-pay coupons electronically within the ePrescription platform to pharmacies on behalf of their patients. The OptimizeRx
solution is integrated into the ePrescribing or Electronic Medical Records applications, but can also be accessed on their mobile
device as well as an application on a prescriber’s desktop.
Our
solutions provide health care institutions with an alternative option to the traditional inefficiencies and issues associated
with storing and managing physical drug samples and pre-printed coupons and provides better access and affordability to patients
to improve affordability, adherence, education and outcomes. In turn, we provide pharmaceutical manufacturers with both direct
to consumer and direct to physician channels for more efficiently communicating and promoting their products and savings with
a method of transparent return on investment.
The
consolidated financial statements for the three month periods ended March 31, 2016 and 2015 have been prepared by us without audit
pursuant to the rules and regulations of the U.S. Securities and Exchange Commission. In the opinion of management, all adjustments
necessary to present fairly our financial position, results of operations, and cash flows as of March 31, 2016 and 2015, and for
the periods then ended, have been made. Those adjustments consist of normal and recurring adjustments. The consolidated balance
sheet as of December 31, 2015, has been derived from the audited consolidated balance sheet as of that date.
Certain
information and note disclosures normally included in our annual financial statements prepared in accordance with generally accepted
accounting principles have been condensed or omitted. These consolidated financial statements should be read in conjunction with
a reading of the financial statements and notes thereto included in our Annual Report on Form 10-K for the fiscal year ended December
31, 2015, as filed with the U.S. Securities and Exchange Commission.
The
results of operations for the three month period ended March 31, 2016, are not necessarily indicative of the results to be expected
for the full year. Certain reclassifications have been made in the prior period’s consolidated financial statements to conform
to the current period’s presentation.
NOTE
2 – STOCKHOLDERS EQUITY
As
described in greater detail in Note 4, related party transactions, in February 2016, we made a one-time payment of $720,415 to
our previous CEO in lieu of issuing shares owed to him from prior years. A portion of this payment, $357,415, was for 295,384
shares of common stock reflected in stock payable at December 31, 2015.
In
March 2016, we issued 12,500 shares of common stock to Independent Directors in connection with our Director Compensation plan
which calls for issuance of 6,250 shares per quarter to each Independent Director. These shares were valued at $13,125. In January
2015, we issued 12,500 shares of common stock to our Independent Directors in connection with the same compensation plan. Those
shares were recorded as stock payable at December 31, 2014. In addition, we recorded an additional 12,500 shares, valued at $16,375
as stock payable at March 31, 2015 for shares to be issued in April 2015.
In
February 2015, we entered into a capital markets advisory agreement covering a one year period, which calls for 90,000 shares
of common stock to be issued as compensation. These shares were valued at $112,500 and are being amortized to expense over the
period of service. Of these shares, 45,000 were issued in March 2015, and the balance were issued in August 2015.
OPTIMIZERx
CORPORATION
NOTES
TO THE CONDENSED CONSOLIDATED FINANCIAL STATEMENTS (UNAUDITED)
MARCH
31, 2016
NOTE
3 – SHARE BASED PAYMENTS – OPTIONS
We
use the fair value method to account for stock based compensation. We recorded $79,459 and $53,841 in compensation expense in
the periods ended March 31, 2016 and 2015, respectively, related to options issued under our stock-based incentive compensation
plan. This includes expense related to options issued in prior years for which the requisite service period for those options
includes the current year as well as options issued in the current year. The fair value of these instruments was calculated using
the Black-Scholes option pricing model. Information related to the assumptions used in this model is set forth in our Annual Report
on Form 10-K for the fiscal year ended December 31, 2015.
NOTE
4 – RELATED PARTY TRANSACTIONS
In
February 2016, after hiring a new CEO, we paid our previous CEO $720,415 in lieu of issuing him 595,384 shares of common stock
based on the 50 day average price of $1.21 per share. A total of 295,384 of these shares were due as a result of previously granted
stock awards in 2014 and 2015, for which shares had not yet been issued. These shares were recorded as stock payable on the balance
sheet at December 31, 2015. The remaining 300,000 shares were due in connection with the purchase of a patent from the previous
CEO in 2010. These shares were recorded as accounts payable – related party on the balance sheet at December 31, 2015. The
difference between the value the shares were initially recorded at in 2010 and the amount they were redeemed at in 2016 was recorded
as additional paid in capital.
Also,
in April 2016, we and the previous CEO entered into a separation agreement and an 18 month consulting agreement, both of which
we recently disclosed in a Form 8-K that we filed with the U.S. Securities and Exchange Commission. The consulting agreement set
forth the terms of the previous CEO’s continued relationship with our company. He remained our employee through March 31,
2016 and the consulting agreement began April 1, 2016. Under the terms of the consulting agreement, he will receive a monthly
payment of $15,000, with the potential for up to $54,000 in additional bonus payments during the term of the agreement. This agreement
also calls for total payments of $12,425 related to insurance benefits. The separation agreement and consulting agreement replace
and supersede all previously disclosed payments related to his severance and board fees.
NOTE
5 – CONTINGENCIES
Litigation
The
company is currently involved in the following legal proceedings.
In
September, 2014, we initiated litigation against Shadron Stastney, our CEO from January to December 2013, in the U.S. District
Court in the Eastern District of Michigan as a result of a dispute related to his separation agreement. Mr. Stastney alleged damages
related to the non-registration of shares that he was granted as part of his separation agreement signed in September 2013. Under
the terms of the contract we are not obligated to register the shares and we deny any obligation to do so. We have requested declarative
relief from the court and also requested an injunction from the court preventing Mr. Stastney from continuing to pursue his claims.
Mr. Stastney has filed a counterclaim requesting damages of $450,000 related to the non-registration of his shares. The parties
are currently in the discovery process and a dispositive motion has been filed by Mr. Stastney. We are in the process of preparing
our response to the motion.
OPTIMIZERx
CORPORATION
NOTES
TO THE CONDENSED CONSOLIDATED FINANCIAL STATEMENTS (UNAUDITED)
MARCH
31, 2016
NOTE
5 – CONTINGENCIES (continued)
In
March, 2015, we initiated litigation against LDM Group, LLC and PDR Network, LLC in the U.S. District Court in the Eastern District
of Missouri related to the breach by LDM, and PDR as successor, of the settlement agreement signed February 28, 2014 related to
previous litigation with LDM. LDM has failed to live up to its obligations under the settlement agreement including, but not limited
to, not allowing us to distribute our eCoupon programs in the LDM network, not allowing us to distribute the LDM patient education
programs, and not providing other information required under the settlement agreement. We are seeking enforcement of the settlement
agreement and we are seeking damages in an amount at least equal to the amounts paid to date to LDM under the settlement agreement,
which is in excess of $1.0 million, as well as damages for lost income and business value as a result of LDM’s breach of
the agreement.
In
March, 2015, we also initiated litigation against PDR Network, LLC in the U.S. District Court in the District of New Jersey as
a result of PDR’s breach of the Master Services Agreement between the parties requiring PDR to exclusively use our eCoupon
solution. We assert that PDR’s acquisition of LDM and the use of the LDM network to distribute coupons by PDR violates the
agreement between the parties and we are seeking damages in an amount at least equal the amounts paid to date by us to LDM under
the settlement agreement, which is in excess of $1.0 million, as well as damages for lost income and business value as a result
of PDR’s actions.
In
May, 2015, we filed an amended complaint in the Missouri case to consolidate the two cases and withdrew the case against PDR Networks
in the U.S. District Court in the District of New Jersey, without prejudice. In July, 2015, the U.S. District Court for the Eastern
District of Missouri dismissed the case, citing lack of Federal jurisdiction in the matter. We refiled the consolidated case against
PDR Network and LDM group in State court in Missouri. The defendants have filed a motion to dismiss two of the four counts in
the consolidated complaint. In January, 2016, the Court dismissed one of our four claims, but allowed the other three to continue
forward. The parties are currently in the discovery process.
NOTE
6 – SUBSEQUENT EVENTS
In
accordance with ASC 855-10, we have analyzed our operations subsequent to March 31, 2016 through the date these financial
statements were issued and have determined that we do not have any material subsequent events to disclose in these financial statements
other than the events described below.
In
April 2016, we issued 34,235 shares of common stock in connection with the cashless exercise of an expiring option to purchase
100,000 shares of common stock previously granted to a consultant.
KLJ
& Associates, LLP
5201
Eden Ave.
Suite
300
Edina,
MN 55436
Office:
630-277-2330
Fax:
763-592-8238
REPORT
OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
Board
of Directors
OptimizeRx
Corporation
Rochester,
MI
To
Whom It May Concern:
We
have audited the accompanying consolidated balance sheets of OptimizeRx Corporation as of December 31, 2015 and 2014, and the
related consolidated statements of operations, shareholders' equity (deficit), and cash flows for the years then ended. These
consolidated financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion
on these consolidated financial statements based on our audits.
We
conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those
standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements
are free of material misstatement. The Company is not required to have, nor were we engaged to perform, an audit of its internal
control over financial reporting. Our audits included consideration of internal control over financial reporting as a basis for
designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the
effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit
also includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing
the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement
presentation. We believe that our audits provide a reasonable basis for our opinion.
In
our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position
of OptimizeRx Corporation as of December 31, 2015 and 2014 and the results of their operations and their cash flows for years
ended December 31, 2015 and 2014, in conformity with U.S. generally accepted accounting principles.
Sincerely,
/s/
KLJ & Associates, LLP
KLJ
& Associates, LLP
Edina,
MN
March
15, 2016
OPTIMIZERx
CORPORATION
Consolidated
Balance Sheets as of
December
31, 2015 and 2014
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2015
|
|
|
2014
|
|
|
ASSETS
|
|
|
|
|
|
|
|
Current Assets
|
|
|
|
|
|
|
|
Cash
and cash equivalents
|
|
$
|
8,207,565
|
|
|
$
|
3,446,973
|
|
|
Accounts receivable
|
|
|
2,847,450
|
|
|
|
2,100,381
|
|
|
Prepaid
expenses
|
|
|
70,623
|
|
|
|
28,093
|
|
|
Total Current
Assets
|
|
|
11,125,638
|
|
|
|
5,575,447
|
|
|
Property and
equipment, net
|
|
|
10,239
|
|
|
|
12,813
|
|
|
Other Assets
|
|
|
|
|
|
|
|
|
|
Patent rights,
net
|
|
|
832,884
|
|
|
|
930,854
|
|
|
Web development
costs, net
|
|
|
340,470
|
|
|
|
504,643
|
|
|
Security
deposit
|
|
|
5,049
|
|
|
|
5,049
|
|
|
Total Other Assets
|
|
|
1,178,403
|
|
|
|
1,440,546
|
|
|
TOTAL ASSETS
|
|
$
|
12,314,280
|
|
|
$
|
7,028,806
|
|
|
LIABILITIES AND
STOCKHOLDERS' EQUITY
|
|
|
|
|
|
|
|
|
|
Current Liabilities
|
|
|
|
|
|
|
|
|
|
Accounts payable
- trade
|
|
$
|
212,191
|
|
|
$
|
200,372
|
|
|
Accounts payable
- related party
|
|
|
570,000
|
|
|
|
570,000
|
|
|
Accrued expenses
|
|
|
6,983
|
|
|
|
25,459
|
|
|
Revenue share payable
|
|
|
2,355,608
|
|
|
|
1,502,761
|
|
|
Deferred
revenue
|
|
|
227,002
|
|
|
|
120,130
|
|
|
Total
Liabilities
|
|
|
3,371,784
|
|
|
|
2,418,722
|
|
|
Stockholders' Equity
|
|
|
|
|
|
|
|
|
|
Preferred stock,
$.001 par value, 10,000,000 shares authorized, no issued and outstanding at December 31, 2015 and 2014,
|
|
|
-0-
|
|
|
|
-0-
|
|
|
Common stock,
$.001 par value, 500,000,000 shares authorized, 29,030,925 and 22,867,319 shares issued and outstanding at December 31, 2015
and 2014, respectively
|
|
|
29,031
|
|
|
|
22,867
|
|
|
Stock warrants
|
|
|
2,329,508
|
|
|
|
2,153,295
|
|
|
Additional paid-in-capital
|
|
|
32,185,499
|
|
|
|
27,595,609
|
|
|
Stock Payable
|
|
|
1,132,148
|
|
|
|
963,063
|
|
|
Deferred stock
compensation
|
|
|
(13,800
|
)
|
|
|
-0-
|
)
|
|
Accumulated
deficit
|
|
|
(26,719,890
|
)
|
|
|
(26,124,750
|
)
|
|
Total Stockholders'
Equity
|
|
|
8,942,496
|
|
|
|
4,610,084
|
|
|
TOTAL LIABILITIES
AND STOCKHOLDERS' EQUITY
|
|
$
|
12,314,280
|
|
|
$
|
7,028,806
|
|
The
accompanying notes are an integral part of these financial statements.
OPTIMIZERx
CORPORATION
Consolidated
Statements of Operations for the Years
Ended
December 31, 2015 and 2014
|
|
|
For the
|
|
|
For the
|
|
|
|
|
year ended
|
|
|
year ended
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
|
|
|
|
|
NET REVENUE
|
|
$
|
7,220,678
|
|
|
$
|
6,502,962
|
|
|
REVENUE SHARE
EXPENSE
|
|
|
3,622,203
|
|
|
|
3,221,534
|
|
|
GROSS
MARGIN
|
|
|
3,598,475
|
|
|
|
3,281,428
|
|
|
EXPENSES
|
|
|
|
|
|
|
|
|
|
Operating expenses
|
|
|
|
|
|
|
|
|
|
Stock-based compensation
|
|
|
581,106
|
|
|
|
1,172,242
|
|
|
Depreciation and
amortization
|
|
|
333,950
|
|
|
|
264,340
|
|
|
Lawsuit settlement
|
|
|
-0-
|
|
|
|
400,000
|
|
|
Other
operating expenses
|
|
|
3,280,826
|
|
|
|
2,471,174
|
|
|
Total
Operating expenses
|
|
|
4,195,882
|
|
|
|
4,307,756
|
|
|
LOSS
FROM OPERATIONS
|
|
|
(597,407
|
)
|
|
|
(1,026,328
|
)
|
|
OTHER INCOME
|
|
|
|
|
|
|
|
|
|
Interest income
|
|
|
2,267
|
|
|
|
935
|
|
|
Interest
expense
|
|
|
-0-
|
|
|
|
-0-
|
|
|
TOTAL
OTHER INCOME
|
|
|
2,267
|
|
|
|
935
|
|
|
LOSS BEFORE PROVISION
FOR INCOME TAXES
|
|
|
(595,140
|
)
|
|
|
(1,025,393
|
)
|
|
PROVISION FOR
INCOME TAXES
|
|
|
-0-
|
|
|
|
-0-
|
|
|
NET
LOSS
|
|
$
|
(595,140
|
)
|
|
$
|
(1,025,393
|
)
|
|
WEIGHTED AVERAGE NUMBER OF SHARES
OUTSTANDING: BASIC
|
|
|
24,562,438
|
|
|
|
22,382,415
|
|
|
NET LOSS PER SHARE:
BASIC (no separate per share amount shown for diluted because loss is antidilutive)
|
|
$
|
(0.02
|
)
|
|
$
|
(0.04
|
)
|
The
accompanying notes are an integral part of these financial statements.
OPTIMIZERx
CORPORATION
Consolidated
Statement of Stockholders’ Equity for the Year
Ended
December 31, 2014
|
|
|
Preferred
Stock
|
|
|
Preferred
Stock
|
|
|
Common
Stock
|
|
|
Common
Stock
|
|
|
Stock
|
|
|
Additional
Paid-in
|
|
|
Stock
|
|
|
Deferred
Stock
|
|
|
Accumulated
|
|
|
Total
Stockholders’
|
|
|
|
|
Shares
|
|
|
Amount
|
|
|
Shares
|
|
|
Amount
|
|
|
Warrants
|
|
|
Capital
|
|
|
Payable
|
|
|
Compensation
|
|
|
Deficit
|
|
|
Equity
|
|
|
Balance, December 31,
2013
|
|
|
65
|
|
|
$
|
-0-
|
|
|
|
14,817,496
|
|
|
$
|
14,817
|
|
|
$
|
18,148,049
|
|
|
$
|
8,875,155
|
|
|
$
|
-0-
|
|
|
$
|
(270,462
|
)
|
|
$
|
(25,099,357
|
)
|
|
$
|
1,668,202
|
|
|
Issuance
of stock options:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
to
employees
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
272,804
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
272,804
|
|
|
to
consultants
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
16,935
|
|
|
|
|
|
|
|
(16,935
|
)
|
|
|
|
|
|
|
-0-
|
|
|
Issuance
of common stock:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
for
services
|
|
|
|
|
|
|
|
|
|
|
167,065
|
|
|
|
167
|
|
|
|
|
|
|
|
26,812
|
|
|
|
14,688
|
|
|
|
|
|
|
|
|
|
|
|
41,667
|
|
|
for
cash
|
|
|
|
|
|
|
|
|
|
|
8,333,333
|
|
|
|
8,333
|
|
|
|
|
|
|
|
8,408,699
|
|
|
|
378,000
|
|
|
|
|
|
|
|
|
|
|
|
8,795,032
|
|
|
for
warrant exercise
|
|
|
|
|
|
|
|
|
|
|
445,765
|
|
|
|
446
|
|
|
|
(694,133
|
)
|
|
|
693,687
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-0-
|
|
|
Issue
warrants for equity raise
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,110,211
|
|
|
|
(1,110,211
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-0-
|
|
|
Issue
stock rights to officers
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
570,375
|
|
|
|
|
|
|
|
|
|
|
|
570,375
|
|
|
Reclassify
expired and redeemed warrants
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(16,410,832
|
)
|
|
|
16,410,832
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
-0-
|
|
|
Expense
consulting services
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
287,397
|
|
|
|
|
|
|
|
287,397
|
|
|
Redeem
shares for cash
|
|
|
(65
|
)
|
|
|
-0-
|
|
|
|
(896,340
|
)
|
|
|
(896
|
)
|
|
|
|
|
|
|
(5,999,104
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(6,000,000
|
)
|
|
Net
loss for the year
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1,025,393
|
)
|
|
|
(1,025,393
|
)
|
|
Balance,
December 31, 2014
|
|
|
-0-
|
|
|
$
|
-0-
|
|
|
|
22,867,319
|
|
|
$
|
22,867
|
|
|
$
|
2,153,295
|
|
|
$
|
27,595,609
|
|
|
$
|
963,063
|
|
|
$
|
-0-
|
|
|
$
|
(26,124,750
|
)
|
|
$
|
4,610,084
|
|
The
accompanying notes are an integral part of these financial statements.
OPTIMIZERx
CORPORATION
Consolidated
Statement of Stockholders’ Equity for the Year
Ended
December 31, 2015
|
|
|
Preferred
Stock
|
|
|
Preferred
Stock
|
|
|
Common
Stock
|
|
|
Common
Stock
|
|
|
Stock
|
|
|
Additional
Paid-in
|
|
|
Stock
|
|
|
Deferred
Stock
|
|
|
Accumulated
|
|
|
Total
Stockholders’
|
|
|
|
|
Shares
|
|
|
Amount
|
|
|
Shares
|
|
|
Amount
|
|
|
Warrants
|
|
|
Capital
|
|
|
Payable
|
|
|
Compensation
|
|
|
Deficit
|
|
|
Equity
|
|
|
Balance, December 31, 2014
|
|
|
-0-
|
|
|
$
|
-0-
|
|
|
|
22,867,319
|
|
|
$
|
22,867
|
|
|
$
|
2,153,295
|
|
|
$
|
27,595,609
|
|
|
$
|
963,083
|
|
|
$
|
-0-
|
|
|
$
|
(26,124,750
|
)
|
|
$
|
4,610,084
|
|
|
Issuance of stock options
to employees
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
253,358
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
253,358
|
|
|
Issuance of common stock:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
for
services
|
|
|
|
|
|
|
|
|
|
|
152,500
|
|
|
|
153
|
|
|
|
|
|
|
|
172,310
|
|
|
|
(14,688
|
)
|
|
|
(97,650
|
)
|
|
|
|
|
|
|
60,125
|
|
|
for
cash
|
|
|
|
|
|
|
|
|
|
|
6,011,106
|
|
|
|
6,011
|
|
|
|
176,213
|
|
|
|
4,164,222
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4,346,446
|
|
|
Issue stock rights to
officers
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
183,773
|
|
|
|
|
|
|
|
|
|
|
|
183,773
|
|
|
Expense consulting services
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
83,850
|
|
|
|
|
|
|
|
83,850
|
|
|
Net
loss for the year
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(595,140
|
)
|
|
|
(595,140
|
)
|
|
Balance, December 31,
2015
|
|
|
-0-
|
|
|
$
|
-0-
|
|
|
|
29,030,925
|
|
|
$
|
29,031
|
|
|
$
|
2,329,508
|
|
|
$
|
32,185,499
|
|
|
$
|
1,132,168
|
|
|
$
|
(13,800
|
)
|
|
$
|
(26,719,890
|
)
|
|
$
|
8,942,496
|
|
The
accompanying notes are an integral part of these financial statements.
OPTIMIZERx
CORPORATION
Consolidated
Statements of Cash Flows for the Years
Ended
December 31, 2015 and 2014
|
|
|
For the
|
|
|
For the
|
|
|
|
|
year ended
|
|
|
year ended
|
|
|
|
|
December 31,
|
|
|
December 31,
|
|
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
|
|
|
|
|
CASH FLOWS FROM OPERATING ACTIVITIES:
|
|
|
|
|
|
|
|
Net
loss for the period
|
|
$
|
(595,140
|
)
|
|
$
|
(1,025,393
|
)
|
|
Adjustments to reconcile net loss to
net cash provided by operating activities:
|
|
|
|
|
|
|
|
|
|
Depreciation and
amortization
|
|
|
333,950
|
|
|
|
264,340
|
|
|
Loss on disposal
of assets
|
|
|
31,731
|
|
|
|
3,295
|
|
|
Stock options
issued for services
|
|
|
253,358
|
|
|
|
272,804
|
|
|
Stock-based compensation
|
|
|
327,748
|
|
|
|
899,438
|
|
|
Changes in:
|
|
|
|
|
|
|
|
|
|
Accounts receivable
|
|
|
(747,069
|
)
|
|
|
(588,672
|
)
|
|
Prepaid expenses
|
|
|
(42,530
|
)
|
|
|
(16,322
|
)
|
|
Accounts payable
|
|
|
11,819
|
|
|
|
11,633
|
|
|
Revenue share payable
|
|
|
852,847
|
|
|
|
215,210
|
|
|
Accrued expenses
|
|
|
(18,476
|
)
|
|
|
13,459
|
|
|
Deferred
revenue
|
|
|
106,872
|
|
|
|
(106,142
|
)
|
|
NET
CASH PROVIDED BY (USED IN) OPERATING ACTIVITIES
|
|
|
515,110
|
|
|
|
(56,350
|
)
|
|
CASH FLOWS FROM INVESTING
ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
Purchases of property
and equipment
|
|
|
(2,046
|
)
|
|
|
(6,984
|
)
|
|
Patent rights
|
|
|
(1,519
|
)
|
|
|
(110,551
|
)
|
|
Web
development costs
|
|
|
(97,399
|
)
|
|
|
(292,417
|
)
|
|
NET
CASH USED IN INVESTING ACTIVITIES
|
|
|
(100,964
|
)
|
|
|
(409,952
|
)
|
|
CASH FLOWS FROM FINANCING
ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
Proceeds from issuance
of common stock
|
|
|
4,346,446
|
|
|
|
8,795,032
|
|
|
Redemption
of common and preferred stock
|
|
|
-0-
|
|
|
|
(6,000,000
|
)
|
|
NET
CASH PROVIDED BY FINANCING ACTIVITIES
|
|
|
4,346,446
|
|
|
|
2,795,032
|
|
|
NET INCREASE IN CASH
AND CASH EQUIVALENTS
|
|
|
4,760,592
|
|
|
|
2,328,730
|
|
|
CASH
AND CASH EQUIVALENTS - BEGINNING OF PERIOD
|
|
|
3,446,973
|
|
|
|
1,118,243
|
|
|
CASH
AND CASH EQUIVALENTS - END OF PERIOD
|
|
$
|
8,207,565
|
|
|
$
|
3,446,973
|
|
|
SUPPLEMENTAL CASH
FLOW INFORMATION:
|
|
|
|
|
|
|
|
|
|
Cash
paid for interest
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
Cash
paid for income taxes
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
|
SUPPLEMENTAL DISCLOSURE
OF NONCASH INVESTING AND FINANCING ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
Common
stock issued for future services
|
|
$
|
-0-
|
|
|
$
|
-0-
|
|
The
accompanying notes are an integral part of these financial statements.
OPTIMIZERx
CORPORATION
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31,
2015
NOTE
1 – NATURE OF BUSINESS
OptimizeRx
Corporation is a technology solutions company targeting the healthcare industry. Our objective is to bring better access to better
healthcare through connecting patients, physicians and pharmaceutical manufacturers through technology. Originally defined as
a marketing and advertising company through our consumer website, we have matured into a technology solutions provider with our
direct to physician solution, SampleMD. SampleMD allows physicians to search, print and send available sample trial vouchers and/or
co-pay coupons on behalf of their patients. The SampleMD solution is integrated into the physician’s ePrescribing or Electronic
Health Record applications, but can also be a stand-alone desktop application. OptimizeRx solutions provide pharmaceutical manufacturers
a direct to physician channel for communicating and promoting their products. It allows healthcare providers a means to provide
sampling and coupons without having to physically store samples on site, and it provides better access and affordability to patients.
The
company was originally formed as Optimizer Systems, LLC in the State of Michigan on January 31, 2006. It converted its form to
a corporation on October 22, 2007 and changed its name to OptimizeRx Corporation. On April 14, 2008, RFID, Ltd., a Colorado corporation,
consummated a reverse merger by entering into a share exchange agreement with the stockholders of OptimizeRx Corporation, pursuant
to which the stockholders of OptimizeRx Corporation exchanged all of the issued and outstanding capital stock of OptimizeRx Corporation
for 1,256,958 shares of common stock of RFID, Ltd., representing 100% of the outstanding capital stock of RFID, Ltd. As of April 30,
2008, RFID’s officers and directors resigned their positions and RFID changed its business to OptimizeRx’s business.
On April 15, 2008, RFID, Ltd.’s corporate name was changed to OptimizeRx Corporation. On September 4, 2008, a
migratory merger was completed, thereby changing the state of incorporation from Colorado to Nevada, resulting in the current
corporate structure, in which OptimizeRx Corporation, a Nevada corporation, is the parent corporation, and OptimizeRx Corporation,
a Michigan corporation, is its wholly-owned subsidiary (together, "OptimizeRx" and "the Company").
NOTE
2 – SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis
of Presentation
The
financial statements of the Company have been prepared in accordance with generally accepted accounting principles in the United
States of America and are presented in US dollars.
Accounting
Basis
The
Company uses the accrual basis of accounting and accounting principles generally accepted in the United States of America (“GAAP”
accounting). The Company has adopted a December 31 fiscal year-end.
Principles
of Consolidation
The
financial statements reflect the consolidated results of OptimizeRx Corporation (a Nevada corporation) and its wholly owned subsidiary,
OptimizeRx Corporation (a Michigan corporation). All material inter-company transactions have been eliminated in the consolidation.
Cash
and Cash Equivalents
For
purposes of the accompanying financial statements, the Company considers all highly liquid instruments with an initial maturity
of three months or less to be cash equivalents.
OPTIMIZERx
CORPORATION
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31,
2015
NOTE
2 – SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (CONTINUED)
Fair
Value of Financial Instruments
The
fair value of cash, accounts receivable, prepaid expenses, accounts payable, accounts payable – related party, accrued expenses
and deferred revenue approximates the carrying amount of these financial instruments due to their short-term nature.
Fair
value is defined as the price that would be received upon sale of an asset or paid upon transfer of a liability in an orderly
transaction between market participants at the measurement date and in the principal or most advantageous market for that asset
or liability. The fair value should be calculated based on assumptions that market participants would use in pricing the asset
or liability, not on assumptions specific to the entity. In addition, the fair value of liabilities should include consideration
of non-performance risk including our own credit risk.
In
addition to defining fair value, the disclosure requirements around fair value establish a fair value hierarchy for valuation
inputs, which is expanded. The hierarchy prioritizes the inputs into three levels based on the extent to which inputs used in
measuring fair value are observable in the market. Each fair value measurement is reported in one of the three levels, which is
determined by the lowest level input that is significant to the fair value measurement in its entirety. These levels are:
Level
1 – inputs are based upon unadjusted quoted prices for identical instruments traded in active markets.
Level
2 – inputs are based upon significant observable inputs other than quoted prices included in Level 1, such as quoted prices
for identical or similar instruments in markets that are not active, and model-based valuation techniques for which all significant
assumptions are observable in the market or can be corroborated by observable market data for substantially the full term of the
assets or liabilities.
Level
3 – inputs are generally unobservable and typically reflect management’s estimates of assumptions that market participants
would use in pricing the asset or liability. The fair values are therefore determined using model-based techniques that include
option pricing models, discounted cash flow models, and similar techniques. The Company’s stock options and warrants are
valued using level 3 inputs.
The
carrying value of the Company’s financial assets and liabilities, which consist of cash, accounts receivable, prepaid expenses,
patent rights, web development costs, accounts payable, accounts payable – related party, accrued expenses and deferred
revenue, are valued using level 1 inputs. The Company believes that the recorded values approximate their fair value due to the
short maturity of such instruments. Unless otherwise noted, it is management’s opinion that the Company is not exposed to
significant interest, exchange or credit risks arising from these financial instruments.
Accounts
Receivable and Allowance for Doubtful Accounts
Accounts
receivable are reported at realizable value, net of allowances for doubtful accounts, which is estimated and recorded in the period
the related revenue is recorded. The Company has a standardized approach to estimate and review the collectability of its receivables
based on a number of factors, including the period they have been outstanding. Historical collection and payer reimbursement experience
is an integral part of the estimation process related to allowances for doubtful accounts. In addition, the Company regularly
assesses the state of its billing operations in order to identify issues, which may impact the collectability of these receivables
or reserve estimates. Because the Company’s customers are primarily large well-capitalized companies, historically there
has been very little bad debt expense. Bad debt expense was $0 for each of the years ended December 31, 2015 and 2014. The
allowance for doubtful accounts was $0 as of both December 31, 2015 and 2014.
OPTIMIZERx
CORPORATION
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31,
2015
NOTE
2 – SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (CONTINUED)
Property
and Equipment
Capital
assets are being depreciated over their estimated useful lives of three to seven years using the straight-line method of depreciation
for book purposes.
Revenue
Recognition and Revenue Share Expense
Revenue
is recognized when it is earned. Revenues are primarily generated from our content delivery activities in which we deliver eCoupons
and eVouchers through a distribution network of ePrescribers and Electronic Health Record technology providers (channel partners),
or from reselling services that complement our business for other of our partners.
We
recognize setup fees that are required for integrating client offerings and campaigns into our rule-based content delivery system
and network upon completion of the setup when the client’s campaign is ready to launch within our system. As the eCoupons
and or eVouchers are distributed through our platform and network of channel partners (a transaction), these transactions are
recorded and revenue is recognized at the time of distribution. Revenue for transactions can be realized based on a price per
distribution, a price per redemption, or as a flat fee over a period of time, depending on the client contract. Additionally,
the Company also recognizes revenue for providing program performance reporting and maintenance, either by the Company directly
delivering reports or by providing access to its online reporting portal that the client can utilize. These fees are charged monthly
and recognized as recurring monthly revenue.
In
some instances, we also resell products and or services that are available through our channel partners, and that are complementary
to our core business and client base. In these instances, net revenue is recognized based on the commission based revenue split
that the Company receives.
Based
on the volume of transactions that are delivered through our channel partner network, we provide a revenue share to compensate
the partner for their promotion of the campaign. Revenue shares are a negotiated percentage of the transaction fees and can also
be specific to special considerations and campaigns. In addition, we pay revenue share to PDR/LDM as a result of a 2014 legal
settlement in an amount equal to the greater of 10% of eCoupon distribution revenues generated or $0.37 per eCoupon distributed.
The contractual amount due to our channel partners is recorded as an expense at the time the eCoupon is distributed.
Income
Taxes
Income
taxes are computed using the asset and liability method. Under the asset and liability method, deferred income tax assets and
liabilities are determined based on the differences between the financial reporting and tax basis of assets and liabilities and
are measured using the currently enacted tax rates and laws. A valuation allowance is provided for the amount of deferred tax
assets that, based on available evidence, are not expected to be realized.
Use
of Estimates
The
preparation of financial statements in conformity with generally accepted accounting principles requires management to make estimates
and assumptions that affect the reported amounts of assets and liabilities at the date of the financial statements and the reported
amounts of revenues and expenses during the reporting period. Estimates and assumptions have been made in determining the carrying
value of assets, depreciable and amortizable lives of tangible and intangible assets, the carrying value of liabilities, the amount
of revenue to be billed, and the timing of revenue recognition and related revenue share expenses. Actual results could differ
from these estimates.
Concentration
of Credit Risks
The
Company maintains its cash and cash equivalents in bank deposit accounts, which, at times, may exceed federally insured limits.
The Company has not experienced any losses in such accounts; however, amounts in excess of the federally insured limit may be
at risk if the bank experiences financial difficulties.
OPTIMIZERx
CORPORATION
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31,
2015
NOTE
2 – SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (CONTINUED)
Research
and Development
The
Company expenses research and development expenses as incurred. Our research efforts are focused on understanding the market dynamics
that have the potential to affect the business and increase revenue in both the short and long term. Our primary goal is to increase
revenue by helping patients better afford and access the medicines their doctors prescribe, as well as other healthcare products
and services they need. Based on this, the Company continually seeks ways to improve its technology to enhance user experiences,
and to develop new services and solutions for its customers.
Share-based
Payments
The
Company uses the fair value method to account for stock-based compensation. The fair value of the equity instrument is charged
directly to compensation expense and additional paid-in capital over the period during which services are rendered. The fair value
of each award is estimated on the date of each grant. For restricted stock, the fair market value is based on the market value
of the stock granted on the date of the grant. For options, it is estimated using the Black=Scholes option pricing model that
uses the assumptions noted in the following table. Estimated volatilities are based on the historical volatility of the Company’s
stock over the same period as the expected term of the options. The expected term of options granted represents the period of
time that options granted are expected to be outstanding. The Company uses historical data to estimate option exercise behavior
and to determine this term. The risk free rate used is based on the U.S. Treasury yield curve in effect at the time of the grant
using a time period equal to the expected option term. The Company has never paid dividends and does not expect to pay any dividends
in the future.
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
|
|
|
|
|
Expected
dividend yield
|
|
|
0
|
%
|
|
|
0
|
%
|
|
Risk
free interest rate
|
|
|
0.24%
- 0.93%
|
|
|
|
0.90%
- 1.44%
|
|
|
Expected
option term
|
|
|
2.5
- 3.5 years
|
|
|
|
3.5
years
|
|
|
Turnover/forfeiture
rate
|
|
|
0
|
%
|
|
|
0
|
%
|
|
Expected
volatility
|
|
|
67%
- 85%
|
|
|
|
117%
- 138%
|
|
The
Black-Scholes option valuation model and other existing models were developed for use in estimating the fair value of traded
options that have no vesting restrictions and are fully transferable. These option valuation models require the input of, and
are highly sensitive to, subjective assumptions including the expected stock price volatility. OptimizerRx’s stock options
have characteristics significantly different from those of traded options, and changes in the subjective input assumptions could
materially affect the fair value estimate.
Loss
Per Common and Common Equivalent Share
The
computation of basic earnings per common share is computed using the weighted average number of common shares outstanding during
the year. The computation of diluted earnings per common share is based on the weighted average number of shares outstanding during
the year plus common stock equivalents, which would arise from the exercise of warrants outstanding using the treasury stock method
and the average market price per share during the year. Options, warrants and convertible preferred stock have not been included
in the diluted earnings per share calculation for either year since their effect is anti-dilutive in all years presented.
OPTIMIZERx
CORPORATION
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31,
2015
NOTE
2 – SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (CONTINUED)
Impairment
of Long-Lived Assets
The
Company continually monitors events and changes in circumstances that could indicate carrying amounts of long-lived assets may
not be recoverable. When such events or changes in circumstances are present, the Company assesses the recoverability of long-lived
assets by determining whether the carrying value of such assets will be recovered through undiscounted expected future cash flows.
If the total of the future cash flows is less than the carrying amount of those assets, the Company recognizes an impairment loss
based on the excess of the carrying amount over the fair value of the assets. Assets to be disposed of are reported at the lower
of the carrying amount or the fair value less costs to sell.
Recently
Issued Accounting Guidance
The
Company does not expect the adoption of recently issued accounting pronouncements to have a significant impact on the Company’s
results of operations, financial position or cash flow.
NOTE
3 – PREPAID EXPENSES
Prepaid
expenses consisted of the following as of December 31, 2015 and 2014:
|
|
|
2015
|
|
|
2014
|
|
|
Insurance
|
|
$
|
30,623
|
|
|
$
|
18,093
|
|
|
Rent
|
|
|
-0-
|
|
|
|
-0-
|
|
|
Legal
|
|
|
40,000
|
|
|
|
10,000
|
|
|
Total
prepaid expenses
|
|
$
|
70,623
|
|
|
$
|
28,093
|
|
NOTE
4 – PROPERTY AND EQUIPMENT
The
Company owned equipment recorded at cost which consisted of the following as of December 31, 2015 and 2014:
|
|
|
2015
|
|
|
2014
|
|
|
Computer
equipment
|
|
$
|
21,565
|
|
|
$
|
19,519
|
|
|
Furniture
and fixtures
|
|
|
11,088
|
|
|
|
11,088
|
|
|
Subtotal
|
|
|
32,653
|
|
|
|
30,607
|
|
|
Accumulated
depreciation
|
|
|
(22,414
|
)
|
|
|
(17,794
|
)
|
|
Property
and equipment, net
|
|
$
|
10,239
|
|
|
$
|
12,813
|
|
Depreciation
expense was $4,620 and $5,933 for the years ended December 31, 2015 and 2014, respectively.
OPTIMIZERx
CORPORATION
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31,
2015
NOTE
5 – WEB-BASED TECHNOLOGY
The
Company has capitalized costs in developing its web-based technology, which consisted of the following as of December 31,
2015 and 2014:
|
|
|
2015
|
|
|
2014
|
|
|
OptimizeRx
web-based technology
|
|
$
|
154,133
|
|
|
$
|
154,133
|
|
|
SampleMD
web-based technology
|
|
|
602,517
|
|
|
|
602,517
|
|
|
SampleMD
2.0 web-based technology
|
|
|
537,877
|
|
|
|
440,477
|
|
|
Subtotal,
web-based technology
|
|
|
1,294,527
|
|
|
|
1,197,127
|
|
|
Accumulated
amortization
|
|
|
(954,057
|
)
|
|
|
(692,484
|
)
|
|
Web-based
technology, net
|
|
$
|
340,470
|
|
|
$
|
504,643
|
|
Amortization
is recorded using the straight-line method over a period of up to five years. During 2014, the Company launched its SampleMD 2.0
web-based technology. All remaining carrying value at December 31, 2015 relates to the SampleMD 2.0 web-based technology. Amortization
expense for the web-based technology costs was $261,572 and $192,760 for the years ended December 31, 2015 and 2014, respectively.
NOTE
6 – PATENT AND TRADEMARKS
On
April 26, 2010, the Company acquired the technical contributions and assignment of all exclusive rights to and for a key
patent from an officer and shareholder in exchange for 300,000 shares of common stock to be granted at the discretion of the seller
and 200,000 stock options, which expired in April 2015, that were valued at $360,000. The shares were valued on the grant date
at $570,000 and have been recorded as a payable to the related party.
The
Company has capitalized costs in purchasing and defending its patent, which consisted of the following as of December 31,
2015 and 2014:
|
|
|
2015
|
|
|
2014
|
|
|
Patent
rights and intangible assets
|
|
$
|
930,000
|
|
|
$
|
930,000
|
|
|
Patent
defense costs
|
|
|
172,457
|
|
|
|
170,937
|
|
|
New
patents and trademarks
|
|
|
58,469
|
|
|
|
90,202
|
|
|
Accumulated
amortization
|
|
|
(328,042
|
)
|
|
|
(260,285
|
)
|
|
Patent
rights and intangible assets, net
|
|
$
|
832,884
|
|
|
$
|
930,854
|
|
The
Company began amortizing the patent, using the straight-line method over the estimated useful life of 17 years, once it was put
into service in July 2010. In 2013, the Company began incurring costs related to defense of the patent. These costs have been
capitalized and will be amortized using the straight-line method over the remaining useful life of the original patent. Amortization
expense was $67,758 and $65,647 for the years ended December 31, 2015 and 2014, respectively.
OPTIMIZERx
CORPORATION
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31,
2015
NOTE
7 – DEFERRED REVENUE
The
Company has several signed contracts with customers for the distribution of coupons, or other services, which include payment
in advance. The payments are not recorded as revenue until the revenue is earned under its revenue recognition policy discussed
in Note 2. Deferred revenue was $227,002 and $120,130 as of December 31, 2015 and 2014, respectively.
NOTE
8 – RELATED PARTY TRANSACTIONS
During
the year ended December 31, 2010, the Company acquired the technical contributions and assignment of all exclusive rights
to and for the SampleMD patent in process at the time from an officer and shareholder in exchange for 300,000 shares of common
stock to be granted at the discretion of the seller and 200,000 stock options, which expired in April 2015, that were valued at
$360,000. The shares were valued on the grant date at $570,000 and have been recorded as a payable to the related party.
During
the year ended December 31, 2015, WPP made a strategic investment in the Company and is a shareholder that owns approximately
20% of the shares of the Company. During 2015, we had billings of $420,503 to agencies that are part of the WPP group and recognized
revenue of $178,855 related to those billings. As of December 31, 2015, we have receivables included in trade receivables on the
balance sheet of $381,125 from WPP agencies and amounts due to WPP agencies included in revenue share payable of $37,803 as of
December 31, 2015.
NOTE
9 – COMMON STOCK
The
Company has 500,000,000 shares of common stock, $.001 par value per share, authorized as of December 31, 2015. There were 29,030,925
and 22,867,319 shares of common stock issued and outstanding at December 31, 2015 and 2014, respectively.
In
September, 2015, we entered into a securities purchase agreement pursuant to which we sold 6,011,106 shares of our common stock
for $0.7875 per share, or gross proceeds of $4,733,746. The shares were issue to a subsidiary of WPP, the world’s largest
marketing services company, as part of a strategic investment by WPP. Placement agents in the offering received commissions and
expenses of $387,300, or approximately 8.2% of the gross proceeds. The net proceeds received were $4,346,446. Placement agents
also received warrants to purchase up to 240,444 shares of our common stock with an exercise price of $0.7875 per share and
a term of 5 years. The warrants were valued at $176,213 and have been recorded as equity issuance costs.
In
2014, the Company adopted a Director Compensation plan covering its independent non-employee Directors. A total of 50,000 shares,
valued at $60,125, were granted and issued in 2015 in connection with this compensation plan. A total of 19,565 shares were granted
in 2014 with a total value of $23,166. A total of 7,065 of these shares were issued in 2014 and the remaining 12,500 shares were
included in stock payable at December 31, 2014 and issued in January 2015.
In
February, 2015, we entered into a capital markets advisory agreement covering a one-year period, which called for 90,000 shares
of common stock to be issued as compensation. These shares were valued at $112,500 and were amortized to expense over the period
of service. 45,000 of these shares were issued in March 2015. The agreement was terminated in July 2015, effective in August,
and the remaining 45,000 shares were not issued. The total expense recognized was $56,250.
In
June, 2015, we agreed to grant 197,605 fully vested shares of our common stock to two executive officers as bonuses. These shares
have not been issued, but are recorded as stock payable and can be requested by the officers at any time.
OPTIMIZERx
CORPORATION
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31,
2015
NOTE
9 – COMMON STOCK (CONTINUED)
In
September, 2015 we entered into a new capital markets advisory agreement covering a one-year period, which called for 90,000 shares
of common stock to be issued as compensation. The first 45,000 shares were issued in September 2015 and valued at $41,400. These
shares are being amortized over a six-month period. The agreement was cancelled in February 2016 and the remaining 45,000 shares
will not be issued.
In March
2014, the Company entered into a securities purchase agreement, pursuant to which the Company sold 8,333,333
shares of the Company’s common stock for $1.20 per share, or gross proceeds of $10,000,000.
Placement
agents in the offering received commissions equal to approximately 9.7% of gross proceeds, for an aggregate commission of approximately
$970,000, including reimbursements for their reasonable out of pocket expenses. Placement agents also received warrants to purchase
up to 804,139 shares of the Company's common stock with an exercise price of $1.20 per share and a term of 5 years. The warrants
were valued at $1,110,211, have been recorded as equity issuance costs, and were registered on a registration statement dated
May 28, 2014. In addition to the warrants to placement agents, the Company also paid cash bonuses of $240,000 to three executive
officers, agreed to issue 200,000 shares to three executive officers, and issued 150,000 shares to a consultant, in connection
with the equity raise. The stock was valued based on the fair market value on the grant date, which was $630,000 in total. These
amounts have been recorded as equity issuance costs, resulting in total equity issuance costs of $2.95 million. The 200,000 shares
for the three executive officers have not been issued, but are recorded as stock payable and can be requested by the respective
officers at any time.
The
Company used the net proceeds of the 2014 offering to exercise the securities redemption option agreement, as amended, with
Vicis Capital Master Fund that provided the Company with an option to purchase all of the outstanding shares and derivative
securities held by Vicis for total payment of $6,000,000. The shares and derivative securities included the Series A Convertible
Preferred Stock, Series B Convertible Preferred Stock, Common Stock, and warrants to purchase shares of common stock held by Vicis
in the Company. The balance of the net proceeds was used for working capital purposes.
In
January 2014, an officer exercised 500,000 stock warrants using the cashless exercise feature included in the warrants. In exchange
for the 500,000 warrants, 410,348 shares of common stock were issued. In October 2014, a consultant exercised 50,000 stock warrants
using the cashless exercise feature included in the warrants. In exchange for the 50,000 warrants, 35,417 shares of common stock
were issued.
In
February 2014, the Company agreed to grant 337,500 shares of common stock, half of which vested immediately and half of which
vested in August 2014, to two executive officers as bonuses based on their efforts to recapitalize the Company to secure approximately
$3 million in working capital while reducing potential fully diluted shares by approximately 7 million shares. Stock-based compensation
related to these bonuses was $570,375 during the year ended December 31, 2014. These shares have not yet been issued and are recorded
as stock payable, but can be requested by the officers at any time.
On
June 1, 2013, the Company entered into a consulting agreement with North Coast Advisors, Inc. for various services. The Company
agreed to issue 20,000 shares of common stock as of the date of the contract. The Company also agreed to issue an additional 20,000
shares every six months in alignment with the agreement renewal up to the two years of the agreement. The first 20,000 shares
were valued at the Company’s common stock closing price as of the date of the contract, which was $1.945/share; and the
second 20,000 shares were valued at the Company’s common stock closing price of $1.50/share on the date of issuance, and
have been expensed. An additional 10,000 shares were issued in 2014 before the agreement was terminated by the Company. The 2014
shares were valued at $1.85, the closing price of the Company’s common stock on the date of issuance.
OPTIMIZERx
CORPORATION
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31,
2015
NOTE
9 – COMMON STOCK (CONTINUED)
On
June 10, 2013, the Company entered into a capital markets advisory agreement with Taglich Brothers, Inc. for various services.
The agreement covered a one-year period and the Company agreed to issue 44,000 shares of common stock to Taglich over the term
of the agreement. The shares were valued at $1.66, the closing price of the stock on the date of the agreement and were written
off over the term of the agreement. The shares were issued in June 2014 upon expiration of the contract.
On
September 20, 2013, the Company entered into a separation agreement that included post-employment consulting services with
a former CEO of the Company. The Company agreed to issue 500,000 shares of common stock, 250,000 shares immediately and 250,000
by January 1, 2014. The shares were issued and the Company recognized the entire issuance in the December 31, 2013 shares
outstanding. The shares were valued at $505,000 and $174,808 of that amount remained as deferred stock compensation as of December 31,
2013, but was fully amortized to expense in 2014.
NOTE
10 – PREFERRED STOCK
Series
A Preferred
During
the year ended December 31, 2008, 35 preferred shares were issued for $3,500,000. Issuance costs totaled $515,000 resulting
in net proceeds of $2,985,000. The 35 shares were convertible into 3,500,000 shares of common stock and bore a 10% cumulative
dividend. In addition, there was a warrant issued to purchase 6,000,000 shares of common stock at an exercise price of $2 for
a period of seven years.
The
holders of the preferred stock were entitled to semi-annual dividends payable on the stated value of the Series A preferred stock
at a rate of 10% per annum, which was cumulative, and accrued daily from the issuance date. The dividends may be paid in cash
or shares of the Company's common stock at management’s discretion. If after the conversion eligibility date, the market
price for the common stock for any ten consecutive trading days in which the stock trades for over $2 per share and trading exceeds
100,000 shares per day, the preferred shareholders can be required to convert their shares to common stock. Each share of Series
A preferred stock was convertible at the option of the holder into that number of shares of common stock of the Company at the
stated value of such share at a $1 conversion price.
The
holder could cause this conversion at the time the shares were eligible for resale by the holder. The conversion price was subject
to adjustment as hereinafter provided, at any time, or from time to time upon the terms and in the manner hereinafter set forth
in the shareholder agreement. There was no conversion expiration date, however, the holder must provide 30 days notice for the
registration of the conversion.
On
May 12, 2010, the Company’s Board declared and issued 236,598 common shares as payment for all cumulative and current
semi-annual dividends. On November 16, 2010, the Company’s Board declared and issued 173,922 common shares for its
semi-annual dividend payment. On March 25, 2011, the Company’s Board declared and issued 176,768 common shares for
its semi-annual dividend payment. On September 21, 2011, the Company's Board declared and issued 156,306 common shares for
its semi-annual dividend payment. The Company had undeclared dividends that were due in February and September 2012 totaling $350,000
and undeclared dividends of $350,000 that were due in February and September 2013 for a total undeclared amount of $700,000 as
of December 31, 2013.
As
described in greater detail in Note 9, all of the Series A Preferred shares were redeemed in 2014.
OPTIMIZERx
CORPORATION
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31,
2015
NOTE
10 – PREFERRED STOCK (CONTINUED)
Series
B Preferred
During
the year ended December 31, 2010, 15 preferred shares were issued for $1,500,000. The 15 shares were convertible into 1,000,000
shares of common stock and bore a 10% cumulative dividend. In addition, there was a warrant issued to purchase 3,000,000 shares
of common stock at an exercise price of $3 for a period of seven years.
The
preferred stock was issued for $1,500,000 less associated issuance costs of $350,000 for net proceeds of $1,150,000. Additionally,
3,000,000 common stock warrants were issued with the preferred stock. Based on the fair values of the preferred stock and common
stock warrants on the issue date, $341,100 was allocated to preferred stock and $1,158,900 was allocated to the common stock warrants.
Equity issuance costs of $350,000 were allocated to the preferred stock.
During
the quarter ended September 30, 2011, 15 preferred shares were issued to an investor for $1,500,000. The 15 shares were convertible
into 1,000,000 shares of common stock and bore a 10% cumulative dividend. In addition, there was a warrant issued to purchase
1,000,000 shares of common stock at an exercise price of $3 for a period of seven years. Based on the fair values of the preferred
stock and common stock warrants on the issue date, $855,460 was allocated to preferred stock and $644,540 was allocated to the
common stock warrants. See Note 12.
The
holders of the preferred stock were entitled to semi-annual dividends payable on the stated value of the Series B preferred stock
at a rate of 10% per annum, which was cumulative, and accrued daily from the issuance date. The dividends may be paid in cash
or shares of the Company's common stock at management’s discretion. If after the conversion eligibility date, the market
price for the common stock for any ten consecutive trading days in which the stock trades for over $2 per share and trading exceeds
100,000 shares per day, the preferred shareholders can be required to convert their shares to common stock. Each share of Series
B preferred stock was convertible at the option of the holder into that number of shares of common stock of the Company at the
stated value of such share at a $1.50 conversion price.
The
holder could cause this conversion at the time the shares were eligible for resale by the holder. The conversion price was subject
to adjustment as hereinafter provided, at any time, or from time to time upon the terms and in the manner hereinafter set forth
in the shareholder agreement.
On
March 25, 2011, the Company’s Board declared and issued 75,758 common shares for its semi-annual dividend payment.
On September 21, 2011, the Company's Board declared and issued 66,988 common shares for its semi-annual dividend payment.
The Company had undeclared dividends that were due in February and September 2012 totaling $150,000 and undeclared dividends of
$150,000 that were due in February and September 2013 for a total undeclared amount of $300,000 as of December 31, 2013.
As
described in greater detail in Note 9, all of the Series B Preferred shares were redeemed in 2014.
OPTIMIZERx
CORPORATION
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31,
2015
NOTE
11 – STOCK OPTIONS
The
Company sponsors a stock-based incentive compensation plan known as the 2013 Equity Compensation Plan (the “Plan”),
which was established by the Board of Directors of the Company in June 2013. A total of 1,500,000 shares were initially reserved
for issuance under the Plan, of which 1,085,000 options have been granted and remain outstanding and 735,105 shares have been
granted, but not issued. Of the shares granted but not issued, the Company has committed to retire 295,384 of those shares for
cash. The Company has no remaining shares available to grant under the Plan, but intends to amend the Plan to increase the number
of shares authorized.
The
Plan allows the Company to grant incentive stock options, non-qualified stock options, stock appreciation right, or restricted
stock. The incentive stock options are exercisable for up to ten years, at an option price per share not less than the fair market
value on the date the option is granted. The incentive stock options are limited to persons who are regular full-time employees
of the Company at the date of the grant of the option. Non-qualified options may be granted to any person, including, but not
limited to, employees, independent agents, consultants and attorneys, who the Company’s Board or Compensation Committee
believes have contributed, or will contribute, to the success of the Company. Non-qualified options may be issued at option prices
of less than fair market value on the date of grant and are exercisable for up to ten years from date of grant. The option vesting
schedule for options granted is determined by the Compensation Committee of the Board of Directors at the time of the grant. The
Plan provides for accelerated vesting of unvested options if there is a change in control, as defined in the Plan.
Prior
to establishment of the Plan, the Board granted options under terms similar to those described in the preceding paragraphs.
The
compensation cost that has been charged against income related to options for the years ended December 31, 2015 and 2014, was
$253,358 and $272,804, respectively. No income tax benefit was recognized in the income statement and no compensation was capitalized
in any of the years presented.
The
Company had the following option activity during the years ended December 31, 2015 and 2014:
|
|
|
Number of
Options
|
|
|
Weighted
average exercise
price
|
|
|
Outstanding,
January 1, 2014
|
|
|
1,180,000
|
|
|
$
|
.97
|
|
|
Granted - 2014
|
|
|
387,500
|
|
|
|
1.73
|
|
|
Exercised - 2014
|
|
|
0
|
|
|
|
0
|
|
|
Expired
– 2014
|
|
|
(260,000
|
)
|
|
|
(0.39
|
)
|
|
Balance, December
31, 2014
|
|
|
1,307,500
|
|
|
|
1.31
|
|
|
Granted – 2015
|
|
|
550,000
|
|
|
|
1.25
|
|
|
Exercised –
2015
|
|
|
0
|
|
|
|
0
|
|
|
Expired
– 2015
|
|
|
(242,500
|
)
|
|
|
(1.68
|
)
|
|
Balance,
December 31, 2015
|
|
|
1,615,000
|
|
|
$
|
1.09
|
|
OPTIMIZERx
CORPORATION
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31,
2015
NOTE
12 –WARRANTS
The
Company has issued warrants, primarily in connection with capital raising activities. As discussed in Note 9, in 2015, we issued
240,444 warrants, with an exercise price of $0.7875 per share in connection with the strategic investment by WPP. As also discussed
in Note 9, in 2014 we issued 804,139 warrants, with an exercise price of $1.20 per share, in connection with a $10 million equity
raise, the proceeds of which were used to retire common stock, preferred, stock, and previously existing warrants.
The
Company had the following warrants outstanding as of December 31, 2015:
|
Number
of Warrants
|
|
Exercise
Price
|
|
|
Expiration
Date
|
|
|
|
|
|
|
|
|
|
|
1,000,000
|
|
$
|
2.25
|
|
|
|
10/5/2017
|
|
|
50,000
|
|
$
|
0.89
|
|
|
|
2/17/2016
|
|
|
804,139
|
|
$
|
1.20
|
|
|
|
3/17/2019
|
|
|
240,444
|
|
$
|
0.7875
|
|
|
|
9/24/2020
|
|
The
Company had the following warrant activity during the years ended December 31, 2015 and 2014:
|
|
|
Number of
Warrants
|
|
|
Weighted
average
exercise
price
|
|
|
Outstanding,
January 1, 2014
|
|
|
11,750,000
|
|
|
$
|
2.27
|
|
|
Granted
|
|
|
804,139
|
|
|
|
1.20
|
|
|
Exercised
|
|
|
(550,000
|
|
|
|
(0.35
|
|
|
Cancelled
|
|
|
(10,000,000
|
|
|
|
(2.40
|
|
|
Expired
|
|
|
(150,000
|
)
|
|
|
(1.45
|
)
|
|
Balance, December
31, 2014
|
|
|
1,854,139
|
|
|
|
1.69
|
|
|
Granted
|
|
|
240,444
|
|
|
|
0.7875
|
|
|
Balance,
December 31, 2015
|
|
|
2,094,583
|
|
|
$
|
1.65
|
|
NOTE
13 – OPERATING LEASES
The
Company signed the lease for its current office space located in Rochester Michigan on December 1, 2011 at an approximate rent
of $5,000 per month. The initial lease term was for three years with an option to renew for an additional two years at approximately
$5,200 per month. The lease was renewed and now expires on November 30, 2016.
Minimum
annual rent payments are as follows for the remainder term of the lease:
|
Year
ended December 31, 2016
|
|
|
57,217
|
|
|
Total
lease commitment
|
|
$
|
57,217
|
|
OPTIMIZERx
CORPORATION
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31,
2015
NOTE
14 – MAJOR CUSTOMERS
The
Company had the following major customers that individually accounted for 10% or more of revenue in any one of the years presented
|
|
2015
|
|
|
Percentage
|
|
|
2014
|
|
|
Percentage
|
|
|
Eli
Lilly and Company
|
$
|
1,409,720
|
|
|
|
20
|
%
|
|
$
|
1,270,064
|
|
|
|
20
|
%
|
|
Alcon
Laboratories, Inc.
|
|
958,653
|
|
|
|
13
|
%
|
|
|
797,972
|
|
|
|
12
|
%
|
|
Astellas
Pharma US, Inc.
|
|
341,555
|
|
|
|
5
|
%
|
|
|
772,320
|
|
|
|
12
|
%
|
|
Daiichi
Sanyko, Inc.
|
|
541,201
|
|
|
|
7
|
%
|
|
|
644,702
|
|
|
|
10
|
%
|
|
Pfizer
|
|
521,317
|
|
|
|
7
|
%
|
|
|
628,741
|
|
|
|
10
|
%
|
|
All
other customers
|
|
3,448,232
|
|
|
|
48
|
%
|
|
|
2,389,163
|
|
|
|
36
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
$
|
7,220,678
|
|
|
|
100
|
%
|
|
$
|
6,502,962
|
|
|
|
100
|
%
|
NOTE
15 – INCOME TAXES
As
of December 31, 2015, the Company had net operating loss carry forwards of approximately $9.1 million that expire from 2027 through
2035 that are available to offset future taxable income. The Company was formed in 2006 as a limited liability company and changed
to a corporation in 2007. Activity prior to incorporation is not reflected in the Company’s corporate tax returns. In the
future, the cumulative net operating loss carry-forward for income tax purposes may differ from the cumulative financial statement
loss due to timing differences between book and tax reporting.
The
provision for Federal income tax consists of the following for the years ended December 31, 2015 and 2014:
|
|
|
2015
|
|
|
2014
|
|
|
Federal
income tax benefit attributable to:
|
|
|
|
|
|
|
|
|
|
Current
operations
|
|
$
|
202,000
|
|
|
$
|
349,000
|
|
|
Permanent
and Timing Differences (net)
|
|
|
(218,000
|
)
|
|
|
(160,000
|
)
|
|
Valuation
allowance
|
|
|
16,000
|
|
|
|
(189,000
|
)
|
|
Net
provision for federal income tax
|
|
$
|
0
|
|
|
$
|
0
|
|
The
cumulative tax effect at the expected rate of 34% of significant items comprising our net deferred tax amount is as follows as
of December 31, 2015 and 2014:
|
|
|
2015
|
|
|
2014
|
|
|
Deferred
tax asset attributable to:
|
|
|
|
|
|
|
|
|
|
Net
operating loss carryover
|
|
$
|
3,110,000
|
|
|
$
|
3,126,000
|
|
|
Valuation
allowance
|
|
|
(3,110,000
|
)
|
|
|
(3,126,000
|
)
|
|
Net
deferred tax asset
|
|
$
|
0
|
|
|
$
|
0
|
|
Under
certain circumstances issuance of common shares can result in an ownership change under Internal Revenue Code Section 382 which
limits the Company’s ability to utilize carry forwards from prior to the ownership change. Any such ownership change resulting
from stock issuances and redemptions could limit the Company’s ability to utilize any net operating loss carry forwards
or credits generated before this change in ownership. These limitations can limit both the timing of usage of these laws, as well
as the loss of the ability to use these net operating losses. It is likely that the fundraising activities of 2014 and 2015 have
resulted in such an ownership change.
OPTIMIZERx
CORPORATION
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31,
2015
NOTE
16 – COMMITMENTS AND CONTINGENT LIABILITIES
Legal
The
Company is currently involved in the following legal proceedings.
In
September, 2014, we initiated litigation against Shadron Stastney, our previous CEO, in the U.S. District Court in the Eastern
District of Michigan as a result of a dispute related to his separation agreement. Mr. Stastney alleged damages related to the
non-registration of shares that he was granted as part of his separation agreement signed in September 2013. Under the terms of
the contract we are not obligated to register the shares and we deny any obligation to do so. We have requested declarative relief
from the court and also requested an injunction to prevent Mr. Stastney from continuing to pursue his claims. Mr. Stastney has
filed a counterclaim requesting damages in the amount of at least $450,000 related to the nonregistration of his shares. The parties
are currently in the discovery process and a dispositive motion has been filed by Mr. Stastney. We are in the process of preparing
our response to the motion.
In
March, 2015, we initiated litigation against LDM Group, LLC and PDR Network, LLC in the U.S. District Court in the Eastern District
of Missouri related to the breach by LDM, and PDR as successor, of the settlement agreement signed February 28, 2014 to resolve
previous litigation with LDM. LDM breached its obligations under the settlement agreement including, but not limited to, not allowing
us to distribute our eCoupon programs in the LDM network, not allowing us to distribute the LDM patient education programs, and
not providing other information required under the settlement agreement. We are seeking enforcement of the settlement agreement
and damages in an amount at least equal to the amounts paid to date to LDM under the settlement agreement, which is in excess
of $1.0 million, as well as damages for lost income and business value as a result of LDM’s breach of the agreement.
In
March, 2015, we also initiated litigation against PDR Network, LLC in the U.S. District Court in the District of New Jersey as
a result of PDR’s breach of the Master Services Agreement between the parties requiring PDR to exclusively use our eCoupon
solution. We assert that PDR’s acquisition of LDM and the use of the LDM network to distribute coupons by PDR violates the
agreement between the parties. We are seeking damages in an amount at least equal the amounts paid to date by us to LDM under
the settlement agreement, which is in excess of $1.0 million, as well as damages for lost income and business value as a result
of PDR’s actions.
In
May, 2015, we filed an amended complaint in the Missouri case to consolidate the two cases and withdrew the case against PDR Networks
in the U.S. District Court in the District of New Jersey, without prejudice. In July, 2015, the U.S. District Court for the Eastern
District of Missouri dismissed the case, citing lack of Federal jurisdiction in the matter. We refiled the consolidated case against
PDR Network and LDM group in State court in Missouri. The defendants filed a motion to dismissal of our claims except those for
breach of contract. In January, the Court dismissed two of six claims asserted against LDM and PDR but allowed the remainder of
our claims to continue forward. The parties are currently engaged in in the discovery process.
Revenue-share
contracts
The
Company has contacts with various Electronic Health Records systems and ePrescribe platforms, whereby we agree to share a portion
of the revenue we generate for eCoupons distributed through their networks. These contracts grant audit rights related to the
payments to our partners, and in some cases would require us to pay for the audit if the audit determined there was an underpayment
and the underpayment meets certain thresholds, such as 10%.
OPTIMIZERx
CORPORATION
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31,
2015
NOTE
16 – COMMITMENTS AND CONTINGENT LIABILITIES (CONTINUED)
Separation
benefits
In
February 2016, the Company hired a new CEO. The previous CEO will terminate as an employee effective March 31, 2016, but remain
non-executive chairman of the Board. In connection with his separation from service, he will receive severance pay of 24 months,
totaling approximately $405,000 and the Company will redeem 595,384 unissued common shares owing to him for an additional payment
of $720,415. This includes the 300,000 shares described in Note 8, related party transactions, and 295,384 shares currently reflected
in stock payable on the balance sheet.
Allscripts
Agreement
In
2015, we signed an amendment to our Allscripts agreement whereby we became its exclusive eCoupon supplier and Allscripts agreed
to integrate our eCoupon functionality into its Touchworks platform. Under the terms of this agreement, we agreed to pay $900,000
in two installments. The first installment of $250,000 was due and paid in November 2015. The second installment of $650,000 is
due when the e-Coupon functionality is launched on a widespread basis in the Touchworks platform, which is currently expected
to be in early 2017. If e-Coupon functionality is not launched by February, 2018, the initial payment of $250,000 will be refunded.
NOTE
17 – RETIREMENT PLAN
The
Company sponsors a defined contribution 401(k) profit sharing plan which was adopted in December, 2015, effective in January 2016.
Under the terms of the plan, the Company matches 100% of the first 3% of payroll contributed by the employee and 50% of the next
2% of payroll contributed by the employee to a maximum of 4% of an employee’s payroll. There was no expense under this plan
in either 2014 or 2015, as the plan became effective in 2016.
NOTE
18 – SUBSEQUENT EVENTS
In
accordance with ASC 855-10, the Company has analyzed its operations subsequent to December 31, 2015 through the date these
financial statements were issued and has determined that it does not have any material subsequent events to disclose in these
financial statements other than the events described below.
In
February 2016, we made the payment of $720,415 described in more detail in Note 16 to redeem shares of stock due to our former
CEO.
CHANGES
IN AND DISAGREEMENTS WITH ACCOUNTANTS
None.
AVAILABLE
INFORMATION
We
file annual, quarterly and current reports, proxy statements and other documents with the SEC. These filings contain important
information which does not appear in this prospectus. You may read and copy, at prescribed rates, any documents we have filed
with the SEC at its Public Reference Room located at 100 F Street, N.E., Washington, DC 20549. You may obtain information on the
operation of the Public Reference Room by calling the SEC at 1-800-SEC-0330. We also file these documents with the SEC electronically.
You can access the electronic versions of these filings on the SEC’s website found at http://www.sec.gov.
We
have filed with the Securities and Exchange Commission (“SEC”) a registration statement for the securities on Form
S-1 under the Securities Act. This prospectus, which forms part of the registration statement, does not contain all the information
contained in the registration statement. Whenever a reference is made in this prospectus to any of our contracts or other documents,
the reference may not be complete and, for a copy of the contract or document, you should refer to the exhibits that are part
of the registration statement.
You
may inspect and copy the registration statement at the public reference facilities maintained by the SEC at 100 F Street, N.E.,
Washington, D.C. 20549 upon payment of certain prescribed fees. You may obtain information on the operation of the SEC’s
public reference facilities by calling the SEC at 1-800-SEC-0330. You may also access the registration statement electronically
through the SEC’s Electronic Data Gathering, Analysis and Retrieval, or EDGAR, system at the SEC’s website located
at http://www.sec.gov.
Until September 15, 2016 (90 days from date
of prospectus), all dealers that effect transactions in these securities whether or not participating in this offering may be
required to deliver a prospectus. This is in addition to the dealers’ obligation to deliver a prospectus when acting as
underwriters and with respect to their unsold allotments or subscriptions.
PROSPECTUS
OPTIMIZERX CORP.
982,472 Shares of Common Stock
OptimizeRx (NASDAQ:OPRX)
Historical Stock Chart
From Aug 2024 to Sep 2024
OptimizeRx (NASDAQ:OPRX)
Historical Stock Chart
From Sep 2023 to Sep 2024