Turning Point Brands: FDA Issues Marketing Denial Order For Vapor Products Application
September 17 2021 - 8:36AM
Dow Jones News
By Michael Dabaie
Turning Point Brands Inc. said the U.S. Food and Drug
Administration denied the company's application to market some of
its vapor products.
Turning Point said it will continue to engage with the FDA and
other stakeholders as it considers options, including a formal
appeal of the decision and potential legal relief.
"While we believe the FDA's current conclusion is misguided, we
will continue our dialogue with the agency in search of a path
forward," said Chief Executive Larry Wexler.
Turning Point's premarket tobacco product application included
an in-depth toxicological review, a clinical study, and studies on
patterns and likelihood of use, the company said.
Write to Michael Dabaie at michael.dabaie@wsj.com
(END) Dow Jones Newswires
September 17, 2021 08:21 ET (12:21 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
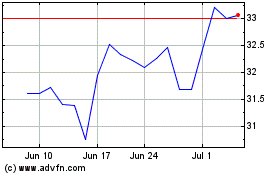
Turning Point Brands (NYSE:TPB)
Historical Stock Chart
From Mar 2024 to Apr 2024
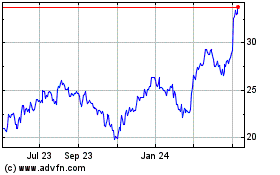
Turning Point Brands (NYSE:TPB)
Historical Stock Chart
From Apr 2023 to Apr 2024