Company recently supported “World Contraception
Day” on September 26th
Company celebrates “World Menopause Month”
TherapeuticsMD, Inc. (NASDAQ:TXMD), an innovative women’s
healthcare company, is proud to reinforce the importance of
empowering women to make informed choices about their sexual and
reproductive health, in consultation with their healthcare
provider.
"It's important for a woman to have a discussion with her
healthcare provider to learn about effective treatment options
available to navigate changes and make informed decisions about her
sexual and reproductive health,” said Dawn Halkuff, Chief
Commercial Officer of TherapeuticsMD. “TherapeuticsMD continues to
generate awareness of treatment options available to women through
our consumer campaigns ‘Unapologetically ANNOVERA®’ and IMVEXXY®’s
‘Sex Care is Self-Care,’ both designed to empower a woman to
self-advocate, start the conversation with her healthcare provider,
and ask about ANNOVERA (segesterone acetate and ethinyl estradiol
vaginal system) and IMVEXXY (estradiol vaginal inserts).”
TherapeuticsMD celebrated “World Contraception Day” with
healthcare professional communication outreach, social posts on the
Company’s brand channels, and media stories featuring leading
medical experts, about innovative new contraceptive options, such
as ANNOVERA. ANNOVERA, which was developed to address the needs of
women who desire a long-lasting form of birth control without
requiring a procedure for insertion and removal coupled with the
patient control and immediate reversibility aspects of short-acting
contraceptive products, was highlighted in the October issue of
Contemporary OBGYN.
ANNOVERA continues to experience growing demand in orders from
wholesalers, retail pharmacies and online distributors as well as
the Company’s expansion into secondary channels, such as public
health and the military. The Company believes that industry
prescription tracking databases do not fully capture the diversity
of prescriptions being filled in these multiple channels.
In honor of Menopause Awareness Month, TherapeuticsMD kicked off
a video series called “Menopause Below the Belt.” The video series
seeks to improve dialogue about menopause and vulvar and vaginal
atrophy (VVA) and encourage women to discuss their menopause
symptoms with their healthcare providers. TherapeuticsMD also
recently sponsored the Oprah Video on the
Myths & Facts of Menopause and the supporting article on
OprahMag.com.
IMPORTANT RISK INFORMATION FOR ANNOVERA (segesterone acetate
and ethinyl estradiol vaginal system)
- Do not use ANNOVERA if you smoke cigarettes and are over 35
years old. Smoking increases your risk of serious heart and blood
vessel (cardiovascular) side effects from hormonal birth control
methods, including death from heart attack, blood clots, or stroke.
This risk increases with age and the number of cigarettes you
smoke.
- ANNOVERA does not protect against HIV infection (AIDS) and
other sexually transmitted infections (STIs)
WARNINGS
ANNOVERA can cause serious side effects, including: blood
clots; toxic shock syndrome (TSS); liver problems, including liver
tumors; high blood pressure; gallbladder problems; changes in the
sugar and fat (cholesterol and triglycerides) levels in your blood;
headache; irregular or unusual vaginal bleeding and spotting
between your menstrual periods; depression; possible cancer in your
cervix; swelling of your skin especially around your mouth, eyes,
and in your throat (angioedema); dark patches of skin on your
forehead, cheeks, upper lip, and chin (chloasma). Call your
healthcare provider or get emergency medical care right away if any
of these serious side effects occur.
The most common side effects of ANNOVERA include:
- headache, including migraine
- nausea/vomiting
- vaginal yeast infection (candidiasis)
- lower/upper abdomen pain
- painful periods
- vaginal discharge
- urinary tract infection
- breast pain/tenderness
- irregular vaginal bleeding
- diarrhea
- genital itching
USE
ANNOVERA is a ring-shaped vaginal system with hormones used by
females to prevent pregnancy.
ANNOVERA has not been adequately studied in females with a body
mass index >29 kg/m2.
Please note that this information is not comprehensive. For
Full Prescribing Information, including BOXED WARNING, please visit
annovera.com/pi.pdf.
IMPORTANT RISK INFORMATION FOR
IMVEXXY (estradiol vaginal inserts)
MOST IMPORTANT INFORMATION YOU
SHOULD KNOW ABOUT IMVEXXY (an estrogen hormone)
- Using estrogen-alone may increase your chance of getting
cancer of the uterus (womb).
- Report any unusual vaginal bleeding right away while you are
using IMVEXXY. Vaginal bleeding after menopause may be a warning
sign of cancer of the uterus (womb). Your healthcare provider
should check any unusual vaginal bleeding to find out the
cause.
- Do not use estrogen-alone or with progestins to prevent
heart disease, heart attacks, strokes, or dementia (decline of
brain function).
- Using estrogen-alone or with progestin may increase your
chances of getting strokes or blood clots.
- Using estrogen-alone or with progestins may increase your
chance of getting dementia, based on a study of women 65 years of
age or older.
- Using estrogens with progestins may increase your chances of
getting heart attacks or breast cancer.
- Talk to your doctor regularly about whether you still need
treatment with IMVEXXY.
DO NOT USE IF YOU
- have unusual vaginal bleeding.
- currently have or have had certain cancers.
- currently have or have had blood clots.
- had a stroke or heart attack.
- currently have or have had liver problems.
- have been diagnosed with a bleeding disorder.
- are allergic to IMVEXXY or any of its ingredients.
- think you may be pregnant.
WARNINGS
Tell your healthcare provider about all of your medical
conditions, including if you:
- have any unusual vaginal bleeding.
- have asthma (wheezing), epilepsy (seizures), diabetes,
migraine, endometriosis, lupus, angioedema (swelling of face and
tongue), problems with your heart, liver, thyroid, kidneys, or have
high calcium levels in your blood.
- are going to have surgery or will be on bed rest. You may need
to stop using IMVEXXY.
- are breast feeding.
Tell your healthcare provider about all the medicines you
take. IMVEXXY may affect the way other medicines work, and
other medicines may affect how IMVEXXY works.
WHEN USING IMVEXXY YOU MAY
HAVE
- headache
- breast tenderness or pain
- nausea and vomiting
USE
IMVEXXY is a prescription medicine that contains an estrogen
hormone in a vaginal insert. It is used after menopause to treat
moderate to severe painful intercourse, a symptom of changes in and
around your vagina, due to menopause.
Please note that this information is not comprehensive. For
Full Prescribing Information, including BOXED WARNING, please visit
imvexxy.com/pi.pdf.
You are encouraged to report negative side effects of
prescription drugs to the FDA. Visit MedWatch, or call
1-800-FDA-1088. You may also report side effects to TherapeuticsMD
at 1-888-228-0150.
About TherapeuticsMD, Inc.
TherapeuticsMD, Inc. is an innovative, leading healthcare
company, focused on developing and commercializing novel products
exclusively for women. Our products are designed to address the
unique changes and challenges women experience through the various
stages of their lives with a therapeutic focus in family planning,
reproductive health, and menopause management. The company is
committed to advancing the health of women and championing
awareness of their healthcare issues. To learn more about
TherapeuticsMD, please visit www.therapeuticsmd.com or follow us on
Twitter: @TherapeuticsMD and on Facebook: TherapeuticsMD.
Forward-Looking Statements for TherapeuticsMD Inc.
This press release by TherapeuticsMD, Inc. may contain
forward-looking statements. Forward-looking statements may include,
but are not limited to, statements relating to TherapeuticsMD’s
objectives, plans and strategies as well as statements, other than
historical facts, that address activities, events or developments
that the company intends, expects, projects, believes or
anticipates will or may occur in the future. These statements are
often characterized by terminology such as "believes," "hopes,"
"may," "anticipates," "should," "intends," "plans," "will,"
"expects," "estimates," "projects," "positioned," "strategy" and
similar expressions and are based on assumptions and assessments
made in light of management’s experience and perception of
historical trends, current conditions, expected future developments
and other factors believed to be appropriate. Forward-looking
statements in this press release are made as of the date of this
press release, and the company undertakes no duty to update or
revise any such statements, whether as a result of new information,
future events or otherwise. Forward-looking statements are not
guarantees of future performance and are subject to risks and
uncertainties, many of which are outside of the company’s control.
Important factors that could cause actual results, developments and
business decisions to differ materially from forward-looking
statements are described in the sections titled "Risk Factors" in
the company’s filings with the Securities and Exchange Commission,
including its most recent Annual Report on Form 10-K and Quarterly
Reports on Form 10-Q, as well as reports on Form 8-K, and include
the following: the effects of the COVID-19 pandemic; the company’s
ability to maintain or increase sales of its products; the
company’s ability to develop and commercialize IMVEXXY®, ANNOVERA®,
and BIJUVA® and obtain additional financing necessary therefor;
whether the company will be able to comply with the covenants and
conditions under its term loan facility; the potential of adverse
side effects or other safety risks that could adversely affect the
commercialization of the company’s current or future approved
products or preclude the approval of the company’s future drug
candidates; whether the FDA will approve the efficacy supplement
for the lower dose of BIJUVA; the company’s ability to protect its
intellectual property, including with respect to the Paragraph IV
notice letters the company received regarding IMVEXXY and BIJUVA;
the length, cost and uncertain results of future clinical trials;
the company’s reliance on third parties to conduct its
manufacturing, research and development and clinical trials; the
ability of the company’s licensees to commercialize and distribute
the company’s products; the ability of the company’s marketing
contractors to market ANNOVERA; the availability of reimbursement
from government authorities and health insurance companies for the
company’s products; the impact of product liability lawsuits; the
influence of extensive and costly government regulation; the
volatility of the trading price of the company’s common stock and
the concentration of power in its stock ownership. PDF copies of
the company’s historical press releases and financial tables can be
viewed and downloaded at its website:
https://ir.therapeuticsmd.com/press-releases.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20201009005281/en/
Nichol Ochsner Vice President, Investor Relations 561-961-1900,
ext. 2088 Nochsner@TherapeuticsMD.com
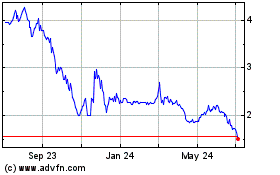
TherapeuticsMD (NASDAQ:TXMD)
Historical Stock Chart
From Mar 2024 to Apr 2024
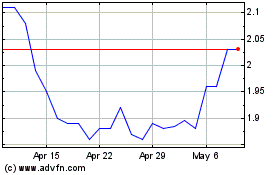
TherapeuticsMD (NASDAQ:TXMD)
Historical Stock Chart
From Apr 2023 to Apr 2024