Atea Gets FDA Fast-Track Designation for AT-752 in Dengue
September 26 2022 - 7:44AM
Dow Jones News
By Colin Kellaher
Atea Pharmaceuticals Inc. on Monday said the U.S. Food and Drug
Administration granted fast-track designation to AT-752, its oral
antiviral candidate for the treatment of dengue virus
infection.
The Boston clinical-stage biopharmaceutical company said AT-752
is in Phase 2 clinical development and was generally well tolerated
in a Phase 1 study.
The FDA's fast-track program is designed to facilitate the
development and expedite the review of treatments for serious or
potentially life-threatening illnesses with high unmet medical
needs.
Atea said there are no currently approved treatments for dengue,
a mosquito-borne viral infection that infects up to 400 million
people a year.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
September 26, 2022 07:29 ET (11:29 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
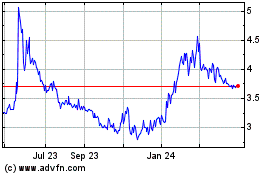
Atea Pharmaceuticals (NASDAQ:AVIR)
Historical Stock Chart
From Mar 2024 to Apr 2024
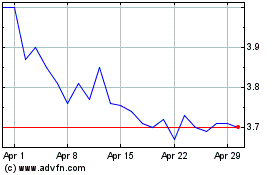
Atea Pharmaceuticals (NASDAQ:AVIR)
Historical Stock Chart
From Apr 2023 to Apr 2024