Quarterly Report (10-q)
May 12 2022 - 7:43AM
Edgar (US Regulatory)
503 Service Unavailable
No server is available to handle this request.
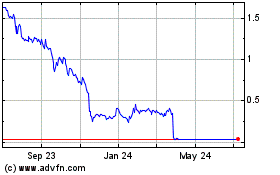
Gamida Cell (NASDAQ:GMDA)
Historical Stock Chart
From Aug 2024 to Sep 2024
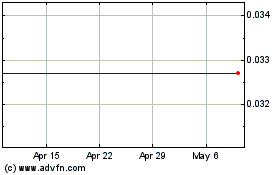
Gamida Cell (NASDAQ:GMDA)
Historical Stock Chart
From Sep 2023 to Sep 2024