Simulations Plus Releases DDDPlus™ Version 6
January 15 2019 - 8:30AM
Business Wire
New models for in vitro systems lead to
improved inputs for GastroPlus® simulations
Simulations Plus, Inc. (Nasdaq:SLP), the leading provider of
modeling and simulation solutions for the pharmaceutical,
biotechnology, and chemicals industries, today announced that it
has released version 6 of its in vitro dissolution modeling
software, DDDPlus™.
Key improvements include:
- New formulation models for advanced
technologies, including long-acting injectable options developed
through the funded collaboration with the U.S. FDA
- New models for in vitro biorelevant
dissolution experiments, including Artificial Stomach and Duodenum
(ASD), Biphasic, and Membrane systems
- Enhanced multistage dissolution
capabilities to guide improved in vitro-in vivo correlation (IVIVC)
development with GastroPlus®
- Improved options for modeling
precipitation kinetics
- And more …
Dr. Viera Lukacova, director of simulation sciences, said: “Our
users asked us to accelerate development of new approaches to help
them in their ongoing quest to extrapolate key insights from in
vitro experiments. With the release of version 6, our talented team
of scientists and engineers developed unique mathematical models
that no other in silico tool offers and validated them for
different applications, especially related to the interplay between
dissolution, precipitation, and absorption kinetics.”
John DiBella, president of the Lancaster division, added: “The
DDDPlus client base has steadily increased over the years, with
several regulatory agencies now using the program to assess
dissolution method and product specification questions. We are
especially excited about the improved functionality and synergies
between this new release of DDDPlus and GastroPlus. Version 6 will
offer users one-of-a-kind approaches that better inform their in
vivo predictions and allow them to make formulation development
decisions with confidence. I am proud of the advances we have made
and look forward to having users apply this to their day-to-day
research.”
An in-depth webinar showcasing the new features in DDDPlus can
be streamed on the company website.
About Simulations Plus, Inc.
Simulations Plus Inc. is a premier developer of drug discovery
and development software as well as a leading provider of both
preclinical and clinical pharmacometric consulting services for
regulatory submissions and quantitative systems pharmacology models
for drug-induced liver injury, drug-induced kidney injury, and
nonalcoholic fatty liver disease. The company is a global leader
focused on improving the ways scientists use knowledge and data to
predict the properties and outcomes of pharmaceutical,
biotechnology, and chemical agents. Our software is licensed to and
used in the conduct of drug research by major pharmaceutical,
biotechnology, chemical, and consumer goods companies and
regulatory agencies worldwide. Our innovations in integrating new
and existing science in medicinal chemistry, computational
chemistry, pharmaceutical science, biology, and physiology into our
software have made us the leading software provider for
physiologically based pharmacokinetic modeling and simulation. For
more information, visit our website at
www.simulations-plus.com.
Safe Harbor Statement Under the Private Securities Litigation
Reform Act of 1995 – With the exception of historical
information, the matters discussed in this press release are
forward-looking statements that involve a number of risks and
uncertainties. Words like “believe,” “expect” and “anticipate” mean
that these are our best estimates as of this writing, but that
there can be no assurances that expected or anticipated results or
events will actually take place, so our actual future results could
differ significantly from those statements. Factors that could
cause or contribute to such differences include, but are not
limited to: our ability to maintain our competitive advantages,
acceptance of new software and improved versions of our existing
software by our customers, the general economics of the
pharmaceutical industry, our ability to finance growth, our ability
to continue to attract and retain highly qualified technical staff,
our ability to identify and close acquisitions on terms favorable
to the Company, and a sustainable market. Further information on
our risk factors is contained in our quarterly and annual reports
and filed with the U.S. Securities and Exchange Commission.
Follow us on Twitter | LinkedIn
View source
version on businesswire.com: https://www.businesswire.com/news/home/20190115005320/en/
Simulations Plus Investor
RelationsMs. Renee
Bouche661-723-7723renee@simulations-plus.com
Hayden IRMr. Cameron
Donahue651-653-1854cameron@haydenir.com
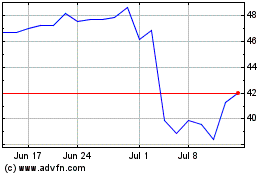
Simulations Plus (NASDAQ:SLP)
Historical Stock Chart
From Mar 2024 to Apr 2024
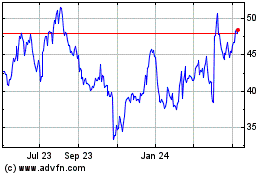
Simulations Plus (NASDAQ:SLP)
Historical Stock Chart
From Apr 2023 to Apr 2024