FDA: Quidel Recalls Some Lyra Covid-19 Tests Due to Risk of False Negatives
July 07 2021 - 5:03PM
Dow Jones News
By Josh Beckerman
Quidel Corp. is recalling certain Covid-19 testing products due
to a "significant risk of false negative results" for some
patients, according to the U.S. Food and Drug Administration.
Quidel initiated the recall of the Lyra SARS-CoV-2 Assay (M120)
on April 26, the FDA said. At that time, Quidel sent all affected
customers an "Urgent: Field Corrective Action" email, calling for
various steps including reviewing revised instructions and
re-testing certain samples.
Write to Josh Beckerman at josh.beckerman@wsj.com
(END) Dow Jones Newswires
July 07, 2021 16:49 ET (20:49 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
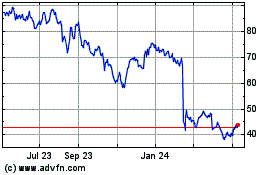
QuidelOrtho (NASDAQ:QDEL)
Historical Stock Chart
From May 2024 to Jun 2024
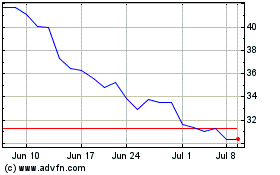
QuidelOrtho (NASDAQ:QDEL)
Historical Stock Chart
From Jun 2023 to Jun 2024