PROCESSA PHARMACEUTICALS ENTERS INTO A LICENSING AGREEMENT WITH OCUPHIRE PHARMA, INC., FOR THE DEVELOPMENT OF RX-3117
June 17 2021 - 9:15AM

Processa Pharmaceuticals, Inc. (NASDAQ: PCSA) announced today that
it has entered into a licensing agreement with Ocuphire Pharma,
Inc. (NASDAQ: OCUP) to license in RX-3117. RX-3117 is an oral,
anticancer agent with an improved pharmacological profile relative
to gemcitabine and other nucleoside analogs. Rx-3117 has a family
of patents extending into 2036 as well as U.S. Food and Drug
Administration (FDA) Orphan Designation for the treatment of
Pancreatic Cancer. Processa will evaluate the potential benefit of
RX-3117 for patients with such cancers as pancreatic or non-small
cell lung cancer.
Under the terms of the agreement, Processa has
an exclusive worldwide license (excluding China), to develop,
manufacture, use, commercialize and sublicense RX-3117.
Processa will be developing biomarker assays to
identify those patients who will most likely benefit from this
targeted therapy. Prior to conducting a pivotal trial, Processa
will first conduct a Phase 2b trial in 2022 to assess the
correlation of the biomarker measurements with the clinical
benefit-risk of RX-3117 in patients with pancreatic cancer or
non-small cell lung cancer.
“We are excited to expand our oncology
portfolio, while providing an important solution for patients with
pancreatic and non-small cell lung cancer,” said Dr. David Young,
Chief Executive Officer of Processa Pharmaceuticals. “The asset
aligns with our mission to identify and bring to market better and
safer drugs for patients who need treatment options to improve
their survival and/or quality of life. From our Phase 2b trial, we
expect to obtain biomarker data that will identify patients who
will benefit the most from this drug while significantly increasing
the probability of a successful Phase 3 trial.”
“The RX-3117 program is a legacy asset from our
merger with Rexahn Pharmaceuticals last year, and outside our core
ophthalmology competency. We are very pleased to establish this
partnership with Processa which has the expertise needed to further
develop RX-3117. The economic terms of the license will be 75%
attributed to the holders of the Rexahn Contingent Value Rights and
25% attributed to Ocuphire,” said Mina Sooch, Chief Executive
Officer for Ocuphire Pharma.
Additional information and updates are available on the
company’s website: http://www.processapharma.com
About Processa Pharmaceuticals, Inc.
The mission of Processa has been to develop
products where existing clinical evidence of efficacy already
exists in unmet medical need conditions, medical conditions where
patients need treatment options that will improve survival and/or
quality of life. The Company has assembled a proven regulatory
science development team, management team, and Board of Directors.
The Processa development team has been involved with more than 30
drug approvals by the FDA (including drug products targeted to
orphan disease conditions) and 100 FDA meetings. For more
information, please visit http://www.processapharma.com.
About Ocuphire Pharma
Ocuphire is a publicly traded (NASDAQ: OCUP),
clinical-stage ophthalmic biopharmaceutical company focused on
developing and commercializing therapies for the treatment of
several eye disorders. Ocuphire’s pipeline currently includes two
small-molecule product candidates – Nyxol and APX3330 – targeting
front and back of the eye indications in late-stage trials. As part
of its strategy, Ocuphire will continue to explore opportunities to
acquire additional ophthalmic assets and to seek strategic partners
for late-stage development, regulatory preparation, and
commercialization in key global markets. For more information,
please visit www.ocuphire.com.
Forward-Looking Statements
This release contains forward-looking
statements. The statements in this press release that are not
purely historical are forward-looking statements which involve
risks and uncertainties. Actual future performance outcomes and
results may differ materially from those expressed in
forward-looking statements. Please refer to the documents filed by
Processa Pharmaceuticals with the SEC, specifically the most recent
reports on Forms 10-K and 10-Q, which identify important risk
factors which could cause actual results to differ from those
contained in the forward-looking statements.
# # #
For More Information: Michael
Floyd mfloyd@processapharma.com301-651-4256
James Carbonara Hayden IR (646)
755-7412 james@haydenir.com
Mina Sooch, CEOOcuphire Pharma,
Inc.ir@ocuphire.comwww.ocuphire.com
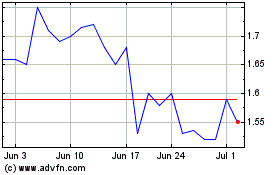
Ocuphire Pharma (NASDAQ:OCUP)
Historical Stock Chart
From Mar 2024 to Apr 2024
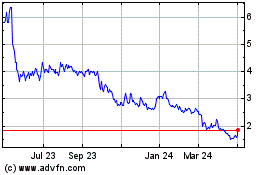
Ocuphire Pharma (NASDAQ:OCUP)
Historical Stock Chart
From Apr 2023 to Apr 2024