HERTFORDSHIRE, England, and
PITTSBURGH, Jan. 8, 2020 /PRNewswire/ -- Mylan
N.V. (NASDAQ: MYL) today announced that its U.S. based Mylan
Pharmaceuticals business is conducting a voluntary nationwide
recall, to the consumer level, of three lots of Nizatidine
Capsules, USP (including the 150mg and 300mg strengths). While
Mylan has not received any reports of adverse events related to
these batches to date, this product is being voluntarily recalled
due to detected trace amounts of an impurity N-nitrosodimethylamine
(NDMA) contained in the API Nizatidine, USP, manufactured by Solara
Active Pharma Sciences Limited.
NDMA is a known environmental contaminant and found in water and
foods, including meats, dairy products and vegetables. NDMA has
been classified as a probable human carcinogen (a substance that
could cause cancer) according to the International Agency for
Research on Cancer (IARC).
The finished products are manufactured by Mylan Pharmaceuticals
Inc. These batches were distributed nationwide to wholesalers, mail
order pharmacies, retail pharmacies, and a distributor between
June 2017 and August 2018. The recalled batches are as
follows:
|
NDC
|
Product
Description
|
Strength
|
Size
|
Lot Number
|
Expiry
|
|
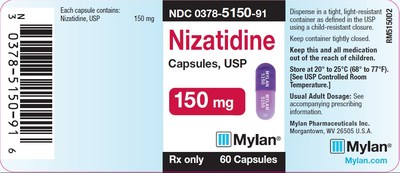
0378-5150-91
|
Nizatidine Capsules,
USP
|
150mg
|
Bottles of
60
|
3086746
|
May 2020
|
|
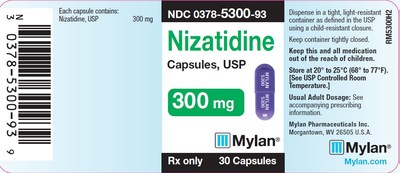
0378-5300-93
|
Nizatidine Capsules,
USP
|
300mg
|
Bottles of
30
|
3082876
|
Jan 2020
|
|
0378-5300-93
|
Nizatidine Capsules,
USP
|
300mg
|
Bottles of
30
|
3082877
|
Jan 2020
|
Nizatidine is indicated for the short-term treatment (up to 8
weeks) of active duodenal ulcers and active benign gastric ulcers,
as maintenance therapy for duodenal ulcer patients for up to one
year, and for up to 12 weeks for the treatment of endoscopically
diagnosed esophagitis and associated heartburn due to
gastroesophageal reflux disease (GERD).
Mylan is notifying its distributors and customers by letter and
is arranging for return of all recalled products. Wholesalers,
retailers and consumers that are in possession of recalled product
should contact Stericycle at 888-628-0727 for the return of the
recalled product. Normal business hours are Monday through Friday
8 a.m. to 5 p.m. EST.
Consumers should contact their physician or healthcare provider
if they have experienced any problems that may be related to using
these drug products.
- Adverse reactions or quality problems experienced with the use
of this product may be reported to the FDA's MedWatch Adverse Event
Reporting program either online, by regular mail or by fax.
Complete and submit the report Online:
www.fda.gov/medwatch/report.htm
- Regular Mail or Fax: Download form
www.fda.gov/MedWatch/getforms.htm or call 1-800-332-1088 to request
a reporting form, then complete and return to the address on the
pre-addressed form or submit by fax to 1-800-FDA-0178.
This recall is being conducted with the knowledge of the U.S.
Food and Drug Administration.
About Mylan
Mylan is a global pharmaceutical company
committed to setting new standards in healthcare. Working together
around the world to provide 7 billion people access to high quality
medicine, we innovate to satisfy unmet needs; make reliability and
service excellence a habit; do what's right, not what's easy; and
impact the future through passionate global leadership. We offer a
growing portfolio of more than 7,500 marketed products around the
world, including antiretroviral therapies on which approximately
40% of people being treated for HIV/AIDS globally depend. We market
our products in more than 165 countries and territories. We are one
of the world's largest producers of active pharmaceutical
ingredients. Every member of our approximately 35,000-strong
workforce is dedicated to creating better health for a better
world, one person at a time. Learn more at Mylan.com. We routinely
post information that may be important to investors on our website
at investor.mylan.com.
 View original content to download
multimedia:http://www.prnewswire.com/news-releases/mylan-initiates-voluntary-nationwide-recall-of-three-lots-of-nizatidine-capsules-usp-due-to-the-detection-of-trace-amounts-of-ndma-n-nitrosodimethylamine-impurity-found-in-the-active-pharmaceutical-ingredient-manufactured-by-s-300983578.html
View original content to download
multimedia:http://www.prnewswire.com/news-releases/mylan-initiates-voluntary-nationwide-recall-of-three-lots-of-nizatidine-capsules-usp-due-to-the-detection-of-trace-amounts-of-ndma-n-nitrosodimethylamine-impurity-found-in-the-active-pharmaceutical-ingredient-manufactured-by-s-300983578.html
SOURCE Mylan N.V.