Eloxx Pharmaceuticals Gets FDA Fast Track Designation for ELX-02 in Cystic Fibrosis
September 09 2021 - 9:21AM
Dow Jones News
By Michael Dabaie
Eloxx Pharmaceuticals Inc. Thursday said the U.S. Food and Drug
Administration gave fast track designation for ELX-02.
ELX-02 is a drug candidate intended to treat cystic fibrosis
patients with nonsense mutations. ELX-02 is currently in Phase 2
clinical trials in CF patients affected by nonsense mutations in
the CF transmembrane conductance regulator gene for whom there are
no effective disease modifying therapies.
Shares were up 6% to $1.47 in premarket trading.
FDA grants fast track designation to drugs being developed for
the treatment of serious or life-threatening diseases or conditions
where there is an unmet medical need. Sponsors of drugs that
receive fast track designation have the opportunity for more
frequent interactions with the FDA review team throughout the
development program.
Write to Michael Dabaie at michael.dabaie@wsj.com
(END) Dow Jones Newswires
September 09, 2021 09:06 ET (13:06 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
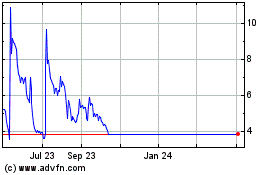
Eloxx Pharmaceuticals (NASDAQ:ELOX)
Historical Stock Chart
From Aug 2024 to Sep 2024
Eloxx Pharmaceuticals (NASDAQ:ELOX)
Historical Stock Chart
From Sep 2023 to Sep 2024