Bruker Launches FluoroType® SARS-CoV-2/Flu/RSV PCR Panel in Europe with CE-IVD Mark
December 18 2020 - 7:00AM
Business Wire
- Winter four-plex panel detects clinically most important
respiratory viruses SARS-CoV-2, Influenza A, Influenza B and
Respiratory Syncytial Virus (RSV)
- Validation with nasopharyngeal swabs and oropharyngeal swabs
for highest sensitivity
- Saliva sampling option convenient for patients, while also
achieving high PCR sensitivity; saliva samples do not require
extraction and enable a faster, resource-effective
workflow
Bruker Corporation (Nasdaq: BRKR) today announced the launch of
the FluoroType SARS-CoV-2/Flu/RSV winter four-plex PCR
panel, which enables the sensitive and simultaneous detection of
the SARS-CoV-2 virus, of both influenza A and B viruses, as well as
of the respiratory syncytial virus (RSV). In the winter season,
multiple viruses need to be differentiated in one assay to
facilitate diagnosis and patient management.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20201218005104/en/
Figure 1: FluoroType SARS-CoV-2/Flu/RSV
Test Kit (Photo: Business Wire)
The FluoroType SARS-CoV-2/Flu/RSV panel is CE-IVD
labelled according to European IVD Directive (98/79/EC). It has
been validated for nasopharyngeal swabs, oropharyngeal swabs and
saliva samples. The kit includes all reagents to generate up to 96
PCR result panels in under two hours after nucleic acid extraction.
In evaluations, the sensitivity and specificity from 112
nasopharyngeal or oropharyngeal swabs have achieved 100% for all
four viruses with very low limits of detection. In addition, the
winter four-plex panel has also been validated with an RNA
extraction-free protocol on 60 saliva samples with 100% sensitivity
and 97.5% specificity. In large scale patient sampling and
diagnostic use, sensitivity and specificity near 100% may not
always be achievable, but swab and saliva PCR protocols remain the
most sensitive assays.
Saliva samples are convenient for patients, e.g. during repeat
surveillance or longitudinal testing. Saliva PCR testing is also
resource-effective for clinical laboratories in case of extraction
kit shortages, and time-to-result is shortened. For optimal
performance from saliva, it is recommended not to eat, drink, brush
teeth or rinse prior to sampling.
The FluoroType SARS-CoV-2/Flu/RSV panel can be used with
Bruker´s high-precision FluoroCycler® XT real-time PCR
system, or with other common real-time thermocyclers. It is
validated for use on Bruker´s GenoXtract® (GXT) automated
nucleic acid extraction devices with associated Bruker RNA
extraction kits, and for Qiagen extraction chemistry, as well as
for the saliva workflow without RNA extraction
Dr. Steffi Czieschnek, the Head of PCR Diagnostics at the
Medical Healthcare Center (MVZ) for Clinical Chemistry and
Microbiology in Suhl, Germany, explained: “For many months,
Bruker´s high-quality FluoroType SARS-CoV-2 plus assay was
our standard for COVID-19 testing. For the winter season, with an
increased incidence of influenza infections, we also needed
differentiation of multiple respiratory viruses in one test to
avoid additional workload or costs. In an early access program, we
have internally validated the FluoroType SARS-CoV-2/Flu/RSV
panel, and we are very pleased with the performance of this winter
four-plex panel, which we now use broadly for our respiratory
disease samples.”
Dr. Wolfgang Pusch, Executive Vice President Microbiology &
Diagnostics at Bruker Daltonics, commented: “We appreciate the
positive customer feedback on the performance of our new
FluoroType SARS-CoV-2/Flu/RSV panel. Compared to PCR assays
that detect only the SARS-CoV-2 virus, our winter four-plex panel
offers added value in a cost-effective workflow. Our clinical
laboratory customers now also have the option of offering the
saliva workflow for patient convenience, faster time-to-result, and
reduced resource requirements without RNA extraction.”
About Bruker-Hain Diagnostics
Bruker-Hain Diagnostics is focused on Molecular Diagnostics
(MDx) products within Bruker´s Microbiology & Diagnostics
business. Hain Lifescience GmbH is also the manufacturer of the
FluoroCycler® XT, MTBDR 2.0 assay, GXT nucleic acid preparation
kits and of the FluoroType SARS-CoV-2 plus assays. For more
information, please visit, www.hain-lifescience.de.
About Bruker Corporation (Nasdaq: BRKR)
Bruker is enabling scientists to make breakthrough discoveries
and develop new applications that improve the quality of human
life. Bruker’s high-performance scientific instruments and
high-value analytical and diagnostic solutions enable scientists to
explore life and materials at molecular, cellular and microscopic
levels. In close cooperation with our customers, Bruker is enabling
innovation, improved productivity and customer success in life
science molecular research, in applied and pharma applications, in
microscopy and nanoanalysis, and in industrial applications, as
well as in cell biology, preclinical imaging, clinical phenomics
and proteomics research and clinical microbiology. For more
information, please visit: www.bruker.com.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20201218005104/en/
Investor Contact: Miroslava Minkova Director of Investor
Relations and Corporate Development T: +1 (978) 663–3660, ext. 1479
E: Investor.Relations@bruker.com Contact for Media and
Customers: Philip Perry Bruker Daltonics T: +49-172-313-7216 E:
Philip.Perry@bruker.com
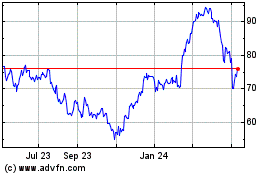
Bruker (NASDAQ:BRKR)
Historical Stock Chart
From Mar 2024 to Apr 2024
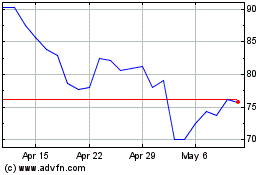
Bruker (NASDAQ:BRKR)
Historical Stock Chart
From Apr 2023 to Apr 2024