Philips launches pre-hospital wireless monitoring solution for emergency medical response in U.S.
July 29 2020 - 9:00AM

Philips launches pre-hospital wireless monitoring solution for
emergency medical response in U.S.
July 29, 2020
- Philips receives 510(k) clearance from the FDA for its
professional defibrillator (Tempus LS- Manual), now offering its
remote monitor and defibrillator solution (Tempus ALS) to EMS
customers in the U.S.
- Solution delivers real-time bidirectional data transfer for
remote patient monitoring, giving EMS responders a new approach to
pre-hospital care
Amsterdam, the Netherlands – Royal Philips (NYSE: PHG,
AEX: PHIA), a global leader in health technology, today announced
the launch of its remote monitoring and defibrillator solution
(Tempus ALS) for pre-hospital settings in the U.S. The solution is
a complete end-to-end system that combines innovative hardware and
advanced software to expand the pre-hospital scope of care for
first responders. The professional defibrillator (Tempus LS-Manual)
is the final element of the overall solution to receive 510(k)
clearance from the U.S. Food and Drug Administration (FDA) and is
now available for sale in the U.S. market.
As a unique modular platform, the remote monitoring and
defibrillator solution (Tempus ALS) consists of a remote portable
vital signs patient monitor (Tempus Pro), and remote professional
defibrillator (Tempus LS-Manual). While the monitor and
defibrillator can be used separately, the devices also connect
wirelessly to share data and transfer vitals, waveforms and images
into Philips web-based software platform (IntelliSpace Corsium).
The software platform provides robust, real-time transfer of
clinical data and events, interactive ECG measurement, two-way
communication and more, enabling rapid clinical and transport
decision support and seamless electronic patient care recording
(ePCR) integration outside the hospital in emergency settings.
“In emergency situations, where seconds count, having access to
advanced patient data collection and sharing and real-time secure
data streaming, can help inform confident treatment and transport
decisions outside the hospital,” said Arman Voskerchyan, General
Manager of Therapeutic Care at Philips. “The integrated remote
monitoring and defibrillator solution combined with our web-based
software platform will help front line responders provide
emergency care, diagnosis and treatment - including defibrillation
therapy, data management and clinical and operational
efficiency features – in a fully integrated solution.”
Emergencies and care events outside the hospital continue to
rise, with an estimated 240 million calls made to 9-1-1 in the U.S.
each year [1]. In addition to the stress of the unknown and what to
expect at the scene of the call, emergency medical providers must
deal with manual handling issues. Equipment carried is heavy, often
damaged due to use in unpredictable conditions and has limited data
connectivity – inhibiting the ability for on-scene support. In an
effort to address these challenges, both elements of the Philips
remote monitoring and defibrillator (Tempus ALS) solution are
designed with a small, rugged exterior and long-lasting battery to
allow emergency medical providers to focus on caring for the
patient without the hassle or distraction of bulky equipment.
Philips leadership in Emergency Care solutions Earlier
this year, Philips launched its new emergency care informatics
suite in the U.S. market, previously in use in Europe, helping care
teams spot life-threatening conditions remotely, improve accuracy
of support from on-scene crews, and enhance tailoring of
in-hospital care based on pre-hospital physiology. In October 2019,
Philips announced a first-of-its-kind collaboration with Air
Ambulance Kent Surrey Sussex (AAKSS) where helicopter
Emergency Service (HEMS) teams were able to live stream patient
medical information from the scene to the hospital through the
Philips pre-hospital solution. Philips offers a wide range of
emergency care offerings, including automated external
defibrillators (AEDs), advanced life support monitors, and more.
Visit Philips Emergency Care and Resuscitation for more information
on the remote monitoring and defibrillator solution and Philips
broad portfolio of Emergency Care solutions. [1]
https://www.nena.org/page/911Statistics
For further information, please contact:
Kathy O'Reilly Philips Global Press OfficeTel: +1
978-221-8919E-mail : Kathy.OReilly@philips.com
About Royal PhilipsRoyal Philips (NYSE: PHG,
AEX: PHIA) is a leading health technology company focused on
improving people's health and enabling better outcomes across the
health continuum from healthy living and prevention, to diagnosis,
treatment and home care. Philips leverages advanced technology and
deep clinical and consumer insights to deliver integrated
solutions. Headquartered in the Netherlands, the company is a
leader in diagnostic imaging, image-guided therapy, patient
monitoring and health informatics, as well as in consumer health
and home care. Philips generated 2019 sales of EUR 19.5 billion and
employs approximately 81,000 employees with sales and services in
more than 100 countries. News about Philips can be found at
www.philips.com/newscenter.
- Tempus ALS Gurney AirLift
- Tempus Grocery Store
- Tempus ALS Woman at Home
- Tempus LS Manual in Bag
- Tempus_ALS_US
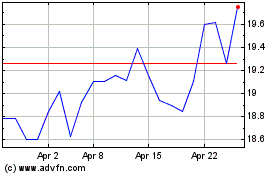
Koninklijke Philips NV (EU:PHIA)
Historical Stock Chart
From Aug 2024 to Sep 2024
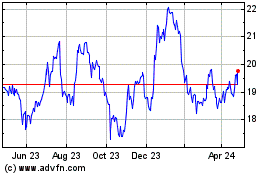
Koninklijke Philips NV (EU:PHIA)
Historical Stock Chart
From Sep 2023 to Sep 2024