Cyclacel Pharmaceuticals Announces First Patient Treated With Oral CYC065 in a Phase 1 Study in Patients With Advanced Solid...
September 09 2019 - 7:00AM

Cyclacel Pharmaceuticals, Inc. (Nasdaq:CYCC, Nasdaq:CYCCP)
("Cyclacel" or the "Company"), a biopharmaceutical company
developing innovative medicines based on cancer biology, announced
treatment of the first patient with an oral formulation of CYC065,
a CDK2/9 inhibitor, in part 3 of a Phase 1 study (CYC065-01) which
is evaluating single agent CYC065 in patients with advanced solid
cancers. Parts 1 and 2 of the study are evaluating CYC065
administered as an intravenous infusion.
“We are excited to report that the first patient
has been treated with oral CYC065 in part 3 of our Phase 1 study,”
said Spiro Rombotis, President and Chief Executive Officer of
Cyclacel. “We recently reported that in part 2 of the same study, a
patient with endometrial cancer with MCL1 amplification achieved
tumor shrinkage after single agent CYC065 administered by
intravenous infusion. The study is being conducted at the
Dana-Farber Cancer Institute. We look forward to reporting data
from Part 2 and 3 of this study in 2020.”
Study CYC065-01 (NCT02552953) is intended to enroll
up to approximately 70 patients with advanced cancers with the
objective of determining safety, pharmacokinetics, pharmacodynamics
and identifying the recommended Phase 2 dose (RP2D) of both
intravenously and orally administered CYC065.
Data from 26 patients enrolled in part 1 were
reported at the 2018 AACR Annual Meeting and RP2D was established
for a 4-hour intravenous infusion of CYC065 once every 3 weeks.
Consistent suppression of MCL1 over 24 hours after a single dose of
CYC065 at RP2D has been observed in the majority of patients. Part
2 is testing a more frequent dosing schedule of one-hour infusions
on Day 1, 2, 8 and 9 of a three week cycle. Part 3 is evaluating
the oral formulation administered by the same dosing schedule as
part 2.
About CYC065CYC065, a second
generation CDK2/9 inhibitor, is being evaluated in Phase 1 studies
as a single agent in patients with advanced solid tumors and in
combination with venetoclax in relapsed or refractory CLL, and in
relapsed or refractory AML or MDS. It is mechanistically similar
but has higher dose potency, in vitro and in vivo, and improved
properties compared to seliciclib, a first generation CDK
inhibitor. Similarly to FDA approved CDK4/6 inhibitors, CYC065 may
be most useful in combination with other anticancer drugs,
including BCL2 inhibitors, such as venetoclax, or HER2 inhibitors,
such as trastuzumab. Preclinical data show that CYC065 may benefit
patients with adult and pediatric hematological malignancies,
including acute myeloid leukemias (AML), acute lymphocytic
leukemias (ALL), and in particular those with MLL rearrangements,
chronic lymphocytic leukemias (CLL), B-cell lymphomas, multiple
myelomas, and certain solid tumors, including breast and uterine
cancers, and neuroblastomas.
About Cyclacel Pharmaceuticals,
Inc.Cyclacel Pharmaceuticals is a clinical-stage
biopharmaceutical company developing innovative cancer medicines
based on cell cycle, transcriptional regulation and DNA damage
response biology. The transcriptional regulation program is
evaluating CYC065 in combination with venetoclax in patients with
relapsed or refractory CLL and AML/MDS. The DNA damage response
program is evaluating an oral combination regimen of sapacitabine
and venetoclax in patients with relapsed or refractory AML/MDS. An
IST is evaluating an oral combination regimen of sapacitabine and
olaparib in patients with BRCA mutant breast cancer. The
anti-mitotic program is evaluating CYC140, a PLK1 inhibitor, in
AML/MDS patients. Cyclacel's strategy is to build a diversified
biopharmaceutical business focused in hematology and oncology based
on a pipeline of novel drug candidates. For additional information,
please visit www.cyclacel.com.
Forward-looking StatementsThis
news release contains certain forward-looking statements that
involve risks and uncertainties that could cause actual results to
be materially different from historical results or from any future
results expressed or implied by such forward-looking statements.
Such forward-looking statements include statements regarding, among
other things, the efficacy, safety and intended utilization of
Cyclacel's product candidates, the conduct and results of future
clinical trials, plans regarding regulatory filings, future
research and clinical trials and plans regarding partnering
activities. Factors that may cause actual results to differ
materially include the risk that product candidates that appeared
promising in early research and clinical trials do not demonstrate
safety and/or efficacy in larger-scale or later clinical trials,
trials may have difficulty enrolling, Cyclacel may not obtain
approval to market its product candidates, the risks associated
with reliance on outside financing to meet capital requirements,
and the risks associated with reliance on collaborative partners
for further clinical trials, development and commercialization of
product candidates. You are urged to consider statements that
include the words "may," "will," "would," "could," "should,"
"believes," "estimates," "projects," "potential," "expects,"
"plans," "anticipates," "intends," "continues," "forecast,"
"designed," "goal," or the negative of those words or other
comparable words to be uncertain and forward-looking. For a further
list and description of the risks and uncertainties the Company
faces, please refer to our most recent Annual Report on Form 10-K
and other periodic filings we file with the Securities and Exchange
Commission, which are available at www.sec.gov. Such
forward-looking statements are current only as of the date they are
made, and we assume no obligation to update any forward-looking
statements, whether as a result of new information, future events
or otherwise.
|
Contacts |
|
| Company: |
Paul McBarron, (908) 517-7330, pmcbarron@cyclacel.com |
| Investor Relations: |
Russo partners LLC, Tony Russo, (212)
845-4251tony.russo@russopartnersllc.com |
© Copyright 2019 Cyclacel Pharmaceuticals, Inc. All Rights
Reserved. The Cyclacel logo and Cyclacel® are trademarks of
Cyclacel Pharmaceuticals, Inc.
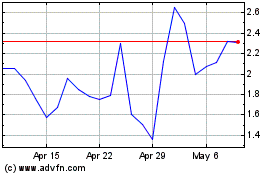
Cyclacel Pharmaceuticals (NASDAQ:CYCC)
Historical Stock Chart
From Aug 2024 to Sep 2024
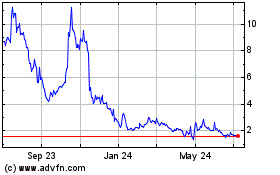
Cyclacel Pharmaceuticals (NASDAQ:CYCC)
Historical Stock Chart
From Sep 2023 to Sep 2024