As filed with the Securities and Exchange
Commission on March 31,
2017
Registration No. 333-212384
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
________________________
Form S-1/A
Amendment No.
3
REGISTRATION
STATEMENT UNDER THE SECURITIES ACT OF 1933
________________________
Guided Therapeutics, Inc.
(Exact name of registrant as specified in its
charter)
________________________
|
Delaware
|
|
3845
|
|
58-2029543
|
|
(State or other
jurisdiction of incorporation or organization)
|
|
(Primary Standard
Industrial Classification Code Number)
|
|
(I.R.S.
Employer
Identification Number)
|
5835 Peachtree Corners East, Suite
D
Norcross, Georgia
30092
(770) 242-8723
(Address, including zip code, and telephone
number, including area code, of registrant’s principal
executive offices)
________________________
Gene S. Cartwright, Ph.D
President and Chief Executive
Officer
Guided Therapeutics,
Inc.
5835 Peachtree Corners East, Suite
D
Norcross, Georgia
30092
(770) 242-8723
(Name, address, including zip code, and
telephone number, including area code, of agent for
service)
________________________
Copy
to:
Heith D. Rodman, Esq.
Jones Day
1420 Peachtree Street,
N.E.
Suite 800
Atlanta, Georgia
30309-3053
(404) 581-3939
________________________
Approximate date of commencement of proposed
sale to the public
: From time to time following the
effective date of this registration statement.
If any of the
securities being registered on this Form are to be offered on a
delayed or continuous basis pursuant to Rule 415 under the
Securities Act of 1933, check the following box.
☑
If this Form is
filed to register additional securities for an offering pursuant to
Rule 462(b) under the Securities Act, please check the following
box and list the Securities Act registration statement number of
the earlier effective registration statement for the same offering.
◻
If this Form is a
post-effective amendment filed pursuant to Rule 462(c) under the
Securities Act, check the following box and list the Securities Act
registration statement number of the earlier effective registration
statement for the same offering. ◻
If this Form is a
post-effective amendment filed pursuant to Rule 462(d) under the
Securities Act, check the following box and list the Securities Act
registration statement number of the earlier effective registration
statement for the same offering. ◻
Indicate by check
mark whether the registrant is a large accelerated filer, an
accelerated filer, a non-accelerated filer, or a smaller reporting
company. See the definitions of “large accelerated
filer,” “accelerated filer,” and “smaller
reporting company” in Rule 12b-2 of the Exchange
Act.
|
Large accelerated
filer
|
◻
|
Accelerated
filer
|
◻
|
Non-accelerated
filer
|
◻
|
Smaller reporting
company
|
☑
|
________________________
CALCULATION OF REGISTRATION
FEE
|
Title of Each Class of
Securities to be Registered
(1)
|
Amount to be Registered
|
Proposed Maximum Offering Price Per
Unit
|
Proposed Maximum Aggregate
Offering
Price
|
Amount of Registration
Fee
|
|
Series D preferred
stock, par value $0.001
|
5,000
|
$1,000
|
$5,000,000
|
(2)
|
|
Warrants to purchase
shares of common stock
|
5,000
|
--
|
--
|
(3)
|
|
Shares of common stock
issuable upon conversion of Series D preferred stock
|
20,000,000
|
--
|
--
|
-
|
|
Shares of common stock
issuable upon exercise of the Warrants
|
20,000,000
|
$0.33 (4)
|
$6,600,000
(4)
|
(2)
|
|
|
(1)
|
In the event of a stock split, stock dividend
or other similar transaction involving the registrant’s
common stock, the number of shares of common stock registered
hereby shall be automatically increased to cover the additional
common shares in accordance with Rule 416(a) under the Securities
Act of 1933.
|
|
|
(2)
|
The registration fee for these securities was
previously paid.
|
|
|
(3)
|
No separate registration fee is payable
pursuant to Rule 457(g) under the Securities Act.
|
|
|
(4)
|
Estimated solely for the purpose of
calculating the registration fee pursuant to Rules 457(g) and (c)
under the Securities Act.
|
The registrant hereby amends this registration
statement on such date or dates as may be necessary to delay its
effective date until the registrant shall file a further amendment
which specifically states that this registration statement shall
thereafter become effective in accordance with Section 8(a) of the
Securities Act of 1933 or until this registration statement shall
become effective on such date as the Securities and Exchange
Commission, acting pursuant to said Section 8(a), may
determine.
The information in this prospectus is
not complete and may be changed. We may not sell or offer these
securities until the registration statement filed with the
Securities and Exchange Commission is declared effective. This
prospectus is not an offer to sell these securities and it is not
soliciting an offer to buy these securities in any state where the
offer or sale is not permitted.
PRELIMINARY PROSPECTUS
Subject to completion, dated March
31, 2017
$5,000,000
5,000 Shares of Series D Convertible Preferred
Stock
Warrants to Purchase up to
16,670,000 Shares of Common Stock
Shares of
Common Stock Underlying the Series D Preferred
Stock
Shares of
Common Stock Underlying the Warrants
________________________
We are offering up
to 5,000 shares of our Series D convertible preferred stock,
together with warrants to purchase an aggregate of up to
16,670,000
shares of common stock (and the shares issuable from time to time
upon conversion of the Series D preferred stock and the exercise of
the warrants), at a purchase price of $1,000 per share of Series D
preferred stock and warrant, pursuant to this prospectus. The
shares of Series D preferred stock and warrants are immediately
separable and will be separately issued.
Subject
to certain ownership limitations, each shares of Series D preferred
stock will be convertible at any time at the holder’s option
into shares of our common stock at an initial conversion price of
$__ per share of common stock. Subject to similar ownership
limitations, each warrant will be immediately exercisable for
up to 3,334 shares of our common stock (based on
warrant coverage of 100% of the shares issued upon conversion
of a share of Series D preferred stock), have an exercise
price of $__ per share, and expire five years from the date of
issuance.
Our
common stock is quoted on the OTCQB marketplace under the symbol
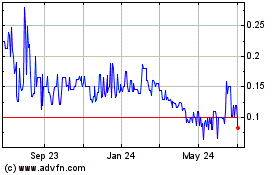
“GTHP.” The last reported sale price of our common
stock on the OTCQB on March 27, 2017 was
$0.40 per share.
|
|
|
|
|
Offering
Price
|
$
1,000
|
$
5,000,000
|
|
Placement
Agent’s Fees (1)
|
$
100
|
$
500,000
|
|
Offering Proceeds,
Before Expenses
|
$
900
|
$
4,500,000
|
_____________________________
|
|
(1)
|
We have agreed to reimburse to the placement
agent for its reasonable, out-of-pocket expenses. See “Plan
of Distribution” on page 20 for more
information.
|
We have retained
Moody Capital Solutions, Inc. to act as our exclusive placement
agent in connection with this offering and to use its “best
efforts” to solicit offers to purchase the securities. The
placement agent is not required to sell any specific number or
dollar amount of securities but will use its best efforts to sell
the securities offered. This best-efforts offering does not have a
minimum purchase requirement and therefore is not certain to raise
any specific amount.
Investing in our common stock involves a high
degree of risk. These risks are described under the caption
“Risk Factors” that begins on page 7 of this
prospectus.
Neither the
Securities and Exchange Commission, or SEC, nor any state
securities commission has approved or disapproved of the securities
that may be offered under this prospectus, nor have any of these
organizations determined if this prospectus is truthful or
complete. Any representation to the contrary is a criminal
offense.
Moody Capital Solutions,
Inc.
The date of this prospectus is ,
2017.

TABLE OF CONTENTS
|
FORWARD-LOOKING STATEMENTS
|
ii
|
|
SUMMARY
|
1
|
|
RISK FACTORS
|
6
|
|
USE OF PROCEEDS
|
18
|
|
DILUTION
|
19
|
|
CAPITALIZATION
|
20
|
|
PLAN OF DISTRIBUTION
|
21
|
|
DESCRIPTION OF SECURITIES WE ARE
OFFERING
|
23
|
|
DESCRIPTION OF SECURITIES
|
26
|
|
OUR BUSINESS
|
30
|
|
PROPERTIES
|
39
|
|
LEGAL PROCEEDINGS
|
39
|
|
MARKET FOR OUR COMMON STOCK AND RELATED
STOCKHOLDER MATTERS
|
39
|
|
MANAGEMENT’S DISCUSSION AND ANALYSIS OF
FINANCIAL CONDITION AND RESULTS OF OPERATIONS
|
40
|
|
DIRECTORS AND EXECUTIVE OFFICERS
|
46
|
|
CORPORATE GOVERNANCE
|
47
|
|
EXECUTIVE COMPENSATION
|
49
|
|
SHARE OWNERSHIP OF DIRECTORS, OFFICERS AND
CERTAIN BENEFICIAL OWNERS
|
51
|
|
CERTAIN RELATIONSHIPS AND RELATED
TRANSACTIONS AND DIRECTOR INDEPENDENCE
|
52
|
|
LEGAL MATTERS
|
53
|
|
EXPERTS
|
53
|
|
WHERE YOU CAN GET MORE INFORMATION
|
55
|
i
ABOUT THIS PROSPECTUS
You should rely
only on the information contained in this prospectus or a
prospectus supplement. We have not authorized any other person to
provide you with different information. If anyone provides you with
different or inconsistent information, you should not rely on it.
This prospectus is an offer to sell only the common stock offered
hereby, but only under circumstances and in jurisdictions where it
is lawful to do so. You should assume that the information
appearing in this prospectus is accurate only as of the
date hereof. Our business, financial condition, results of
operations and prospects may have changed.
The terms
“Guided Therapeutics,” “Company,”
“our,” “we,” and “us,” as used
in this prospectus, refer to Guided Therapeutics, Inc. and its
wholly owned subsidiary.
FORWARD-LOOKING
STATEMENTS
Statements in this
prospectus, which express “belief,”
“anticipation” or “expectation,” as well as
other statements that are not historical facts, are forward-looking
statements. These forward-looking statements are subject to risks
and uncertainties that could cause actual results to differ
materially from historical results or anticipated results,
including those identified in the foregoing “Risk
Factors” and elsewhere in this prospectus. Examples of these
uncertainties and risks include, but are not limited
to:
|
|
●
|
access to sufficient debt or equity capital to
meet our operating and financial needs;
|
|
|
●
|
the extent of dilution of the holdings of our
existing stockholders upon the issuance, conversion or exercise of
securities issued as part of our capital raising
efforts;
|
|
|
●
|
whether and when we or any potential strategic
partners will obtain required regulatory approvals in the markets
in which we plan to operate;
|
|
|
●
|
the effectiveness and ultimate market acceptance
of our products and our ability to generate sufficient sales
revenues to sustain our growth and strategy plans;
|
|
|
●
|
whether our products in development will prove
safe, feasible and effective;
|
|
|
●
|
our need to achieve manufacturing scale-up in a
timely manner, and our need to provide for the efficient
manufacturing of sufficient quantities of our
products;
|
|
|
●
|
the lack of immediate alternate sources of
supply for some critical components of our products;
|
|
|
●
|
our ability to establish and protect the
proprietary information on which we base our products, including
our patent and intellectual property position;
|
|
|
●
|
the need to fully develop the marketing,
distribution, customer service and technical support and other
functions critical to the success of our product
lines;
|
|
|
●
|
the dependence on potential strategic partners
or outside investors for funding, development assistance, clinical
trials, distribution and marketing of some of our products;
and
|
|
|
●
|
other risks and uncertainties described from
time to time in our reports filed with the SEC.
|
Forward-looking
statements should not be read as a guarantee of future performance
or results, and will not necessarily be accurate indications of the
times at, or by which, such performance or results will be
achieved. Forward-looking information is based on information
available at the time and/or management’s good faith belief
with respect to future events, and is subject to risks and
uncertainties that could cause actual performance or results to
differ materially from those expressed in the
statements.
Forward-looking
statements speak only as of the date the statements are made. We
assume no obligation to update forward-looking statements to
reflect actual results, changes in assumptions or changes in other
factors affecting forward-looking information except to the extent
required by applicable securities laws. If we update one or more
forward-looking statements, no inference should be drawn that we
will make additional updates with respect thereto or with respect
to other forward-looking statements.
ii
SUMMARY
This summary highlights information contained
elsewhere in this prospectus. This summary is not complete and may
not contain all of the information that may be important to you. We
urge you to read the entire prospectus carefully, including the
‘‘Risk Factors’’ section, before making an
investment decision. Except for the financial statements included
with this prospectus and unless otherwise noted, all share and per
share data in this prospectus give effect to the 1:800 reverse
stock split of our common stock that was implemented on November 7,
2016. For more information about our reverse stock split, see
“Recent Developments.”
Our Company
We are a medical
technology company focused on developing innovative medical devices
that have the potential to improve healthcare. Our primary focus is
the sales and marketing of our LuViva® Advanced Cervical Scan
non-invasive cervical cancer detection device. The underlying
technology of LuViva primarily relates to the use of biophotonics
for the non-invasive detection of cancers. LuViva is designed to
identify cervical cancers and precancers painlessly, non-invasively
and at the point of care by scanning the cervix with light, then
analyzing the reflected and fluorescent light.
LuViva provides a
less invasive and painless alternative to conventional tests for
cervical cancer screening and detection. Additionally, LuViva
improves patient well-being not only because it eliminates pain,
but also because it is convenient to use and provides rapid results
at the point of care. We focus on two primary applications for
LuViva: first, as a cancer screening tool in the developing world,
where infrastructure to support traditional cancer-screening
methods is limited or non-existent, and second, as a triage
following traditional screening in the developed world, where a
high number of false positive results cause a high rate of
unnecessary and ultimately costly follow-up tests.
Screening for
cervical cancer represents one of the most significant demands on
the practice of diagnostic medicine. As cervical cancer is linked
to a sexually transmitted disease—the human papillomavirus
(HPV)—every woman essentially becomes “at risk”
for cervical cancer simply after becoming sexually active. In the
developing world, there are approximately 2.0 billion women aged 15
and older who are potentially eligible for screening with LuViva.
Guidelines for screening intervals vary across the world, but U.S.
guidelines call for screening every three years. Traditionally, the
Pap smear screening test, or Pap test, is the primary cervical
cancer screening methodology in the developed world. However, in
developing countries, cancer screening using Pap tests is expensive
and requires infrastructure and skill not currently existing, and
not likely to be developed in the near future, in these
countries.
We
believe LuViva is the answer to the developing world’s
cervical cancer screening needs. Screening for cervical cancer in
the developing world often requires working directly with foreign
governments or non-governmental agencies (NGOs). By partnering with
governments or NGOs, we can provide immediate access
to cervical cancer detection to large segments of a nation’s
population as part of national or regional governmental healthcare
programs, eliminating the need to develop expensive and
resource-intensive infrastructures.
Manufacturing,
Sales Marketing and Distribution
We
manufacture LuViva at our Norcross, Georgia facility. Most of the
components of LuViva are custom made for us by third-party
manufacturers. We adhere to ISO 13485:2003 quality standards in our
manufacturing processes.
Research,
Development and Engineering
We have
been engaged primarily in the research, development and testing of
our LuViva non-invasive cervical cancer detection product and our
core biophotonic technology. Since 2013, we have incurred about
$7.2 million in research and development expenses, net
of about $927,000 reimbursed through collaborative arrangements and
government grants. Research and development costs were
approximately $0.7 million and $1.5 million in
2016 and 2015, respectively
Patents
We have
pursued a course of developing and acquiring patents and patent
rights and licensing technology. Our success depends in large part
on our ability to establish and maintain the proprietary nature of
our technology through the patent process and to license from
others patents and patent applications necessary to develop our
products. As of March 27, 2017, we have
25 granted U.S. patents relating to our biophotonic
cancer detection technology and 5 pending U.S. patent
applications. We also have three granted patents that apply to our
interstitial fluid analysis system.
We are a Delaware
corporation, originally incorporated in 1992 under the name
“SpectRx, Inc.,” and, on February 22, 2008, changed our
name to Guided Therapeutics, Inc. At the same time, we renamed our
wholly owned subsidiary, InterScan, which originally had been
incorporated as “Guided Therapeutics.”
Since our
inception, we have raised capital through the public and private
sale of debt and equity, funding from collaborative arrangements,
and grants.
Our
prospects must be considered in light of the substantial risks,
expenses and difficulties encountered by entrants into the medical
device industry. This industry is characterized by an increasing
number of participants, intense competition and a high failure
rate. We have experienced operating losses since our inception and,
as of December 31, 2016 we have an accumulated deficit
of approximately $127.6 million. To date, we have
engaged primarily in research and development efforts and the early
stages of marketing our products. We do not have significant
experience in manufacturing, marketing or selling our products. We
may not be successful in growing sales for our products. Moreover,
required regulatory clearances or approvals may not be obtained in
a timely manner, or at all. Our products may not ever gain market
acceptance and we may not ever generate significant revenues or
achieve profitability. The development and commercialization of our
products requires substantial development, regulatory, sales and
marketing, manufacturing and other expenditures. We expect our
operating losses to continue through at least the end of
2017 as we continue to expend substantial resources to
complete commercialization of our products, obtain regulatory
clearances or approvals, build our marketing, sales,
manufacturing and finance capabilities, and conduct further
research and development.
Our
product revenues to date have been limited. In 2016,
the majority of our revenues were from the sale of LuViva devices
and disposables, as well as some revenue from grants from the NIH
and licensing agreement fees received. We expect that the majority
of our revenue in 2017 will be derived from revenue
from the sale of LuViva devices and disposables.
Our principal
executive and operations facility is located at 5835 Peachtree
Corners East, Suite D, Norcross, Georgia 30092, and our telephone
number is (770) 242-8723.
Recent Developments
Convertible
Promissory Note.
On February 13, 2017, we entered into a
securities purchase agreement with Auctus Fund, LLC for the
issuance and sale to Auctus of $170,000 in aggregate principal
amount of a 12% convertible promissory note for an aggregate
purchase price of $156,400 (representing a $13,600 original issue
discount). On February 13, 2017, we issued the note to Auctus.
Pursuant to the purchase agreement, we also issued to Auctus a
warrant exercisable to purchase an aggregate of 200,000 shares of
our common stock. The warrant is exercisable at any time, at an
exercise price per share equal to $0.77 (110% of the closing price
of the common stock on the day prior to issuance), subject to
certain customary adjustments and price-protection provisions
contained in the warrant. The warrant has a five-year term. The
note matures nine months from the date of issuance and, in addition
to the original issue discount, accrues interest at a rate of 12%
per year. We may prepay the note, in whole or in part, for 115% of
outstanding principal and interest until 30 days from issuance, for
125% of outstanding principal and interest at any time from 31 to
60 days from issuance, and for 130% of outstanding principal and
interest at any time from 61 days from issuance to 180 days from
issuance. After six months from the date of issuance, Auctus may
convert the note, at any time, in whole or in part, into shares of
our common stock, at a conversion price equal to the lower of the
price offered in our next public offering or a 40% discount to the
average of the two lowest trading prices of the common stock during
the 20 trading days prior to the conversion, subject to certain
customary adjustments and price-protection provisions contained in
the note. The note includes customary events of default provisions
and a default interest rate of 24% per year. Upon the occurrence of
an event of default, Auctus may require us to redeem the note (or
convert it into shares of common stock) at 150% of the outstanding
principal balance plus accrued and unpaid interest. In connection
with the transaction, we agreed to reimburse Auctus for $30,000 in
legal and diligence fees, of which we paid $10,000 in cash and
$20,000 in restricted shares of common stock, valued at $0.40 per
share (a 42.86% discount to the closing price of the common stock
on the day prior to issuance).
SMI License
Agreement
. On January 22, 2017, we entered into a license
agreement with Shandong Yaohua Medical Instrument Corporation, or
SMI, pursuant to which we granted SMI an exclusive global license
to manufacture the LuViva device and related disposables (subject
to a carve-out for manufacture in Turkey) and exclusive
distributionrights in the Peoples Republic of China, Macau, Hong
Kong and Taiwan. In exchange for the license, SMI will pay a $1.0
million licensing fee, payable in five installments through October
2017, underwrite the cost of securing approval of LuViva with the
Chinese Food and Drug Administration, or CFDA, and, once it obtains
CFDA approval, pay $1.90 royalty on each disposable sold in the
territories by purchasing directly from us a Controlled
Programmable Chip (CPC), necessary for the operation of disposable
unit. Pursuant to the SMI agreement, SMI must become capable of
manufacturing LuViva in accordance with ISO 13485 for medical
devices by the second anniversary of the SMI agreement, and achieve
CFDA approval by July 22, 2019, or else forfeit the license. During
2017, SMI must purchase no fewer than ten devices at $13,000 each
(with up to four devices pushed to 2018 if there is a delay in
obtaining approval from the CFDA). In the three full calendar years
following CFDA approval, SMI must sell a minimum of 3,500 devices
(500 in the first year, 1,000 in the second, and 2,000 in the
third), and purchase a minimum of 25,200,000 CPCs from us. If SMI
is able to meet these minimums or otherwise agrees to make up the
difference between payments for CPC orders and the minimum payments
to GTI (i.e., “pay to play”), this would result in
revenues of $47,880,000 to us over the three-year period.
Otherwise, SMI would forfeit the license. In any event, we cannot
be assured that SMI will be able to secure CFDA approval or meet
its minimum sales targets, or agree to make up the difference to
preserve their license, and therefore we cannot be assured of any
such revenue. As manufacturer of the devices and disposables, SMI
will be obligated to sell each to us at costs no higher than our
current costs. As partial consideration for, and as a condition to,
the license, and to further align the strategic interests of the
parties, we agreed to issue $1.0 million in shares of our common
stock to SMI, in five installments through October 2017, at a price
per share equal to the lesser of the average closing price for the
five days prior to issuance and $1.25. On March 28, 2017, we
amended the license agreement via a letter agreement to limit
SMI’s beneficial ownership of our common stock on any given
date to no more than 4.99% of our then-outstanding common stock. In
connection with the letter agreement, we agreed to issue to SMI
three warrants, each exercisable for 15,000 shares of our common
stock, to be issued in conjunction with the next three cash
payments by SMI under the license agreement. Each warrant will be
immediately exercisable (subject to the beneficial ownership
limitation), have an exercise price equal to the lesser of the
closing price per share of our common stock for the average of five
consecutive days preceding the payment by SMI and $1.25 per share,
and have a term of five years.
In order to
facilitate the SMI agreement, immediately prior to its execution we
entered into an agreement with Shenghuo Medical, LLC, regarding our
previous license to Shenghuo (see “—Shenghuo
License”). Under the terms of the new agreement, Shenghuo
agreed to relinquish its manufacturing license and its distribution
rights in SMI’s territories, and to waive its rights under
the original Shenghuo agreement, all for as long as SMI performs
under the SMI agreement. As consideration, we have agreed to split
with Shenghuo the licensing fees and net royalties from SMI that we
will receive under the SMI agreement. Should the SMI agreement be
terminated, we have agreed to re-issue the original license to
Shenghuo under the original terms. Our COO and director, Mark
Faupel, is a shareholder of Shenghuo, and another director, Richard
Blumberg, is a managing member of Shenghuo.
OID
Note.
On December 28, 2016, we entered
into a securities purchase agreement with RedDiamond Partners LLC
for the issuance and sale to RedDiamond of up to $330,000 in
aggregate principal amount of 10% original issuance discount
convertible promissory notes, for an aggregate purchase price of
$300,000. On that date, we issued to RedDiamond a note in the
principal amount of $222,000, for a purchase price of $200,000. At
our option, at any time within 60 days from that issuance, we may
issue another note in the principal amount of $110,000, for a
purchase price of $100,000, subject to customary conditions
contained in the purchase agreement. The notes mature six months
from their date of issuance and, in addition to the 10% original
issue discount, accrue interest at a rate of 10% per year. We may
prepay the notes, in whole or in part, for 115% of outstanding
principal and interest until 30 days from issuance, for 125% of
outstanding principal and interest at any time from 31 to 60 days
from issuance, and for 130% of outstanding principal and interest
at any time from 61 days from issuance until immediately prior to
the maturity date. After six months from the date of issuance
(i.e., if we fail to repay all principal and interest due under the
notes at the maturity date), RedDiamond may convert the notes, at
any time, in whole or in part, into shares of our common stock, at
a conversion price equal to 60% of the lowest volume weighted
average price of our common stock during the 20 trading days prior
to conversion, subject to certain customary adjustments and
anti-dilution provisions contained in the notes.
New Directors
and Chief Operating Officer.
On November 10, 2016,
the board of directors increased its size from three members to
four and appointed Richard P. Blumberg to fill the newly created
vacancy. On December 8, 2016, the board of directors appointed Mark
Faupel, our co-founder and former President and CEO, as our new COO
and, after again increasing the size of the board to five,
appointed Dr. Faupel to fill the newly created vacancy. Dr. Faupel
and Mr. Blumberg serves as a Managing Member of
Shenghuo, holder of a license to manufacture our products in Asia.
See “—Shenghuo License Agreement.”
Debt
Restructuring.
On December 7, 2016, we entered into
an exchange agreement with GPB Debt Holdings II LLC with regard to
the $1,525,000 in outstanding principal amount of senior secured
convertible note originally issued to GPB on February 11, 2016 (see
“—Senior Secured Convertible Note”), and the
$306,863 in outstanding principal amount of our secured promissory
note that GPB holds (see “—Secured Promissory
Note”). Pursuant to the exchange agreement, upon completion
of the next financing resulting in at least $1 million in cash
proceeds, GPB will exchange both securities for a new convertible
note in principal amount of $1,831,863. The new convertible note
will mature on February 12, 2018 and will accrue
interest at a rate of 19% per year. We will pay monthly interest
coupons and, beginning one year after issuance, will pay amortized
quarterly principal payments. Subject to resale restrictions under
Federal securities laws and the availability of sufficient
authorized but unissued shares of our common stock, the new
convertible note will be convertible at any time, in whole or in
part, at the holder’s option, into shares of our common
stock, at a conversion price equal to the price offered in the
qualifying financing that triggers the exchange, subject to certain
customary adjustments and anti-dilution provisions contained in the
new convertible note. The new convertible note will include
customary event of default provisions and a default interest rate
of the lesser of 21% or the maximum amount permitted by law. Upon
the occurrence of an event of default, GPB will be entitled to
require us to redeem the new convertible note at 120% of the
outstanding principal balance. The new convertible note will be
secured by a lien on all of our assets, including our intellectual
property, pursuant to the security agreement entered into by us and
GPB in connection with the issuance of the original senior secured
convertible note. As an inducement to GPB to enter into these
transactions, we agreed to increase the royalty payable to GPB
pursuant to its consulting agreement with us from 3.5% to 3.85% of
revenues from the sales of our products. We expect this offering to
trigger GPB’s obligation to exchange.
Reverse Stock
Split.
A 1:800 reverse stock split of all of our
issued and outstanding common stock was implemented on November 7,
2016. As a result of the reverse stock split, every 800 shares of
issued and outstanding common stock was converted into 1 share of
common stock. All fractional shares created by the reverse stock
split were rounded to the nearest whole share. The number of
authorized shares of common stock did not change.
Lockup and
Exchange Agreement.
On November 2, 2016, we entered
into a lockup and exchange agreement with GHS Investments, LLC,
holder of approximately $221,000 in outstanding principal amount of
our secured promissory note and all of the outstanding shares of
our Series C preferred stock (see “—Series C
Exchanges”). Pursuant to the agreement, upon the
effectiveness of the 1:800 reverse stock split and continuing for
45 days after, GHS and its affiliates were prohibited from
converting any portion of the secured promissory note or any of the
shares of Series C preferred stock or selling any of our securities
that they beneficially owned. We agreed that, upon consummation of
our next financing, we would use $260,000 of net cash proceeds
first, to repay GHS’s portion of the secured promissory note
and second, with any remaining amount from the $260,000, to
repurchase a portion of GHS’s shares of Series C preferred
stock. In addition, GHS has agreed to exchange the stated value per
share (plus any accrued but unpaid dividends) of its remaining
shares of Series C preferred stock for new securities of the same
type that we separately issue in the next qualifying financing we
undertake, on a dollar-for-dollar basis in a private placement
exchange. We expect this offering will be a qualifying financing
and, therefore, GHS will be contractually obligated to exchange
those shares of Series C preferred stock for shares of Series D
preferred stock and warrants immediately after we consummate this
offering, in a private placement exchange.
Royalty
Agreement.
On September 6, 2016, we entered into a
royalty agreement with one of our directors, John Imhoff, and
another stockholder, Dolores Maloof, pursuant to which we sold to
them a royalty of future sales of single-use cervical guides for
LuViva. Under the terms of the royalty agreement, and for
consideration of $50,000, we will pay them an aggregate perpetual
royalty initially equal to $0.10, and from and after October 2,
2016, equal to $0.20, for each disposable that we sell (or that is
sold by a third party pursuant to a licensing arrangement with
us).
Warrant
Restructuring.
Between June 13, 2016 and June 14,
2016, we entered into various agreements with holders of certain
warrants (including John Imhoff, one of our directors) originally
issued in May 2013, and with GPB Debt Holdings II LLC, holder of a
warrant issued February 12, 2016, pursuant to which each holder
separately agreed to exchange warrants for either (1) shares of
common stock equal to 166% of the number of shares of common stock
underlying the surrendered warrants, or (2) new warrants
exercisable for 200% of the number of shares underlying the
surrendered warrants, but without certain anti-dilution protections
included with the surrendered warrants. As a result of the
exchanges, we effectively eliminate any potential exponential
increase in the number of underlying shares issuable upon exercise
of our outstanding warrants. In total, for surrendered warrants
then-exercisable for an aggregate of 1,814,598 shares of common
stock (but subject to exponential increase upon operation of
certain anti-dilution provisions), we issued or are obligated to
issue 16,898 shares of common stock and new warrants that, if
exercised as of June 14, 2016, would have been exercisable for an
aggregate of 3,608,835 shares of common stock. In certain
circumstances, in lieu of presently issuing all of the shares (for
each holder that opted for shares of common stock), we and the
holder further agreed that we will, subject to the terms and
conditions set forth in the applicable warrant exchange agreement,
from time to time, be obligated to issue the remaining shares to
the holder. No additional consideration will be payable in
connection with the issuance of the remaining shares. The holders
that elected to receive shares for their surrendered warrants have
agreed that they will not sell shares on any trading day in an
amount, in the aggregate, exceeding 20% of the composite aggregate
trading volume of the common stock for that trading day. The
holders that elected to receive new warrants will be required to
surrender their old warrants upon consummation of this offering.
The new warrants will have an initial exercise price equal to the
exercise price of the surrendered warrants as of immediately prior
to consummation of this offering, subject to customary
“downside price protection” for as long as our common
stock is not listed on a national securities exchange, and will
expire five years from the date of issuance.
Shenghuo
License Agreement.
On June 5, 2016, we entered into a license
agreement with Shenghuo Medical, LLC pursuant to which we granted
Shenghuo an exclusive license to manufacture, sell and distribute
LuViva in Taiwan, Brunei Darussalam, Cambodia, Laos, Myanmar,
Philippines, Singapore, Thailand, and Vietnam. Shenghuo was already
our exclusive distributor in China, Macau and Hong Kong, and the
license extended to manufacturing in those countries
as well. Under the terms of the license agreement, once Shenghuo
was capable of manufacturing LuViva in accordance with
ISO 13485 for medical devices, Shenghuo would pay us a
royalty equal to $2.00 or 20% of the distributor price (subject to
a discount under certain circumstances), whichever is higher, per
disposable distributed within Shenghuo’s exclusive
territories. In connection with the license grant, Shenghuo would
underwrite the cost of securing approval of LuViva with Chinese
Food and Drug Administration. At its option, Shenghuo also
would provide up to $1.0 million in furtherance of our
efforts to secure regulatory approval for LuViva from the U.S. Food
and Drug Administration, or U.S. FDA, in exchange for
the right to receive payments equal to 2% of our future sales in
the United States, up to an aggregate of $4.0 million. Pursuant to
the license agreement, Shenghuo had the option to have
a designee appointed to our board of directors (and Richard
Blumberg is that designee). As partial consideration for,
and as a condition to, the license, and to further align the
strategic interests of the parties, on March 24, 2017
we issued a $300,000 convertible note to Shenghuo, in
exchange for Shenghuo’s June 2016 aggregate cash
investment of $200,000. The unsecured note
accrues interest at 20% per year, compounded
annually, from January 1, 2017 until repayment, and is
convertible into shares of our common stock at a conversion price
per share of $13.92, subject to customary anti-dilution adjustment.
We also issued Shenghuo a five-year warrant
exercisable immediately for 17,239 shares of common stock at an
exercise price equal to theconversion price of the note, subject to
customary anti-dilution adjustment.
On January 22, 2017, we entered into a license agreement with SMI,
pursuant to which we granted SMI an exclusive global license to
manufacture the LuViva device and related disposables (subject to a
carve-out for manufacture in Turkey) and exclusive distribution
rights in the Peoples Republic of China, Macau, Hong Kong and
Taiwan. In order to facilitate the SMI agreement, immediately prior
to its execution we entered into an agreement with Shenghuo,
regarding the previous license to Shenghuo. Under the terms of the
new agreement, Shenghuo agreed to relinquish its manufacturing
license and its distribution rights in SMI’s territories, and
to waive its rights under the original Shenghuo agreement, all for
as long as SMI performs under the SMI agreement
(see
“–SMI License Agreement”).
Cash
Advances.
On May 4, 2016, May 26, 2016, and June 22,
2016 Aquarius Opportunity Fund advanced us a total of $107,500 for
2% simple interest notes due the earlier of December 31, 2016 or
consummation of this offering. Also on May 26, 2016, GPB Debt
Holdings II LLC, holder of our outstanding senior secured
convertible note, advanced as an additional $87,500, on the same
terms as its note. We intend to offer Aquarius the
opportunity to participate in the offering at least up to the
extent of the outstanding principal and interest on its cash
advance, by extinguishing all or a portion of the debt on a
dollar-for-dollar basis.
Series C
Exchanges.
Between April 27, 2016 and May 3, 2016, we
entered into various agreements with certain holders of our Series
C preferred stock, including John Imhoff and Mark Faupel, two of
our directors, pursuant to which those holders separately agreed to
exchange each share of Series C preferred stock held for 2.25
shares of our newly created Series C1 preferred stock and 12 shares
of our common stock. In connection with these exchanges, each
holder also agreed to exchange the $1,000 stated value per share of
the holder’s shares of Series C1 preferred stock for new
securities of the same type that we separately issue in the next
qualifying financing we undertake, on a dollar-for-dollar basis in
private placement exchanges. This obligation is contingent on all
holders of the then-outstanding Series C preferred stock exchanging
their shares of Series C preferred stock on the same terms. We do
not expect the remaining holder of our Series C preferred stock
to exchange its shares on the same terms, and
therefore the holders of our Series C1 preferred stock will not be
so obligated. Finally, each holder agreed, except in the event of
an additional $50,000 cash investment by the holder in our next
public financing of at least $1 million, to execute a customary
“lockup” agreement in connection with such financing.
In total, for 1,916 shares of Series C preferred stock surrendered,
we issued 4,311 shares of Series C1 preferred stock and 22,996
shares of common stock. The Series C1 preferred stock has terms
that are substantially the same as the Series C preferred stock,
except that the Series C1 preferred stock does not pay dividends
(unless and to the extent declared on the common stock) or
at-the-market “make-whole payments.” See
“Description of Securities” for a description of the
material terms of the Series C1 preferred stock. Separately, on
April 27, 2016, we entered into a similar agreement with a prior
holder of Series C preferred stock, Aquarius Opportunity Fund, but
Aquarius subsequently sold all of its shares of Series C preferred
stock (and its portion of our secured promissory note, also subject
to the agreement) to GHS. Pursuant to the agreement, Aquarius also
agreed to return to us for cancelation warrants exercisable for 903
shares of our common stock that it held. Finally, Aquarius, as the
holder of a majority of the outstanding Series C preferred stock,
agreed to amend the Series C stock purchase agreement to eliminate
any participation rights held by the Series C shareholders and to
waive operation of certain anti-dilution provisions of the Series C
that would otherwise be triggered by the transactions described in
“—Recent Developments.”
February 2016
Reverse Stock Split.
On February 24, 2016, we
implemented a 1:100 reverse stock split of all of our issued and
outstanding common stock. As a result of the reverse stock split,
every 100 shares of issued and outstanding common stock was
converted into 1 share of common stock. All fractional shares
created by the reverse stock split were rounded to the nearest
whole share. The number of authorized shares of common stock did
not change.
Secured
Promissory Note.
On February 11, 2016, we consented
to an assignment of our outstanding secured promissory note to two
accredited investors, Aquarius Opportunity Fund and GPB Debt
Holdings II LLC. In connection with the assignment, the holders
waived an ongoing event of default under the notes related to our
minimum market capitalization, and agreed to eliminate the
requirement going forward. On March 7, 2016, we further amended the
notes to eliminate the volume limitations on sales of common stock
issued or issuable upon conversion of the notes. GPB subsequently
sold the note to GHS.
Senior
Secured Convertible Note.
On
February 11, 2016, we entered into a securities purchase agreement
with GPB Debt Holdings II LLC for the issuance and sale on February
12, 2016 of $1.4375 million in aggregate principal amount of a
senior secured convertible note for an aggregate purchase price of
$1.15 million (a 20% original issue discount). The note matures on
the second anniversary of issuance and, in addition to the 20%
original issue discount, accrues interest at a rate of 17% per
year. Subject to certain restrictions, it is convertible at any
time, in whole or in part, at the holder’s option, into
shares of our common stock, at an initial conversion price equal to
$640.00 per share, subject to certain customary adjustments and
anti-dilution provisions. In addition, the investor received a
five-year warrant exercisable to purchase an aggregate of
approximately 2,247 shares of our common stock with an initial
exercise price of $640.00 per share, subject to certain customary
adjustments and anti-dilution provisions. As of March
27, 2017, as a result of the operation of the anti-dilution
provisions, the conversion price of the note and the exercise price
of the warrants are $.2639 per share and
5,447,817 shares were issuable upon exercise of
the warrant. On March 18, 2016, our placement agent received
a warrant with similar terms exercisable for 197,807
shares.
Increase in
Authorized Shares.
On September 15, 2015, we amended
our certificate of incorporation to increase the number of shares
of common stock authorized for issuance to 500.0 million, and on
November 12, 2015, we further amended our certificate of
incorporation to increase the number of shares of common stock
authorized for issuance to 1.0 billion.
Series C
Financing.
On June 29, 2015, we entered into
a securities purchase agreement with certain accredited investors
(including John Imhoff and Mark Faupel, two of our directors),
amended on September 3, 2015, for the issuance and sale of shares
of an aggregate of 7,903 shares of our Series C preferred stock, at
a purchase price of $750 per share and an initial conversion price
of $76.00 per share, and five-year warrants exercisable to purchase
an aggregate of approximately 156,000 shares of our common stock at
an initial exercise price of $76.00 per share, subject to certain
customary adjustments and anti-dilution provisions.
The Offering
|
Issuer
|
Guided Therapeutics, Inc.
|
|
Securities
offered
|
Up to 5,000 shares of Series D preferred
stock and warrants exercisable for an aggregate of up to
16,670,000 shares of our common stock. The shares of
Series D preferred stock and the warrants are immediately separable
and will be issued separately, but will be purchased together in
this offering. This prospectus also covers the shares of common
stock issuable upon conversion of the Series D preferred stock and
upon the exercise of the warrants.
|
|
Offering
price
|
$1,000 combined price for each share of Series D
preferred stock and accompanying warrant
|
|
Description of
Series D preferred stock
|
Series D preferred stock pays a two-year
cumulative dividend of 12% per year, has a liquidation
preference senior to our common stock but junior to our
Series C preferred stock, is convertible at the option of the
holder (subject to certain ownership limitations) into shares of
our common stock at a conversion price of $__ per share (and is
mandatorily convertible upon the election of two-thirds of the
then-outstanding holders), subject to anti-dilution adjustment, and
is redeemable under certain circumstances at our option or the
option of two-thirds of the then-outstanding holders. See
“Description of Securities We Are Offering” on page
22.
|
|
Shares of common
stock underlying the shares of Series D preferred
stock
|
Up to 16,670,000
shares, based on an assumed conversion price of $0.30. The actual
number of shares will depend on the actual conversion price, which
has not been determined, but is expected to be a discount to the
then-current market price of our common
stock
|
|
Description of
warrants
|
Each warrant will
be immediately exercisable for up to 3,334 shares of
our common stock (based on warrant coverage of 100% of the
shares issued upon conversion of a share of Series D
preferred stock), have an exercise price assumed to be
$0.36 per share, and expire five years after the date of
issuance. The actual exercise price has not been determined,
but is expected to be 120% of the conversion price of the Series D
preferred stock. See “Description of Securities We Are
Offering” on page
1.
|
|
Shares of common
stock underlying the warrants
|
Up to 16,670,000
shares (based on warrant coverage of 100% of the shares issued upon
conversion of all 5,000 shares of Series D preferred
stock).
|
|
Shares of common
stock outstanding before and after this
offering
|
1,494,741 shares before this
offering and 18,164,741 shares after this offering,
assuming all shares of Series D preferred stock are sold and
converted to common stock.* See “Dilution” on page
17.
|
|
Use of
proceeds
|
We intend to apply net cash proceeds received in
connection with the offering to increase inventory of LuViva to
meet current demand, repay our outstanding secured promissory note
and repurchase approximately $43,000 in shares of
Series C preferred stock, support our efforts to re-file for
U.S. FDA approval of LuViva, and support general working
capital and operations. However, we will retain broad discretion
over the use of proceeds and may use the money for other corporate
purposes. See “Use of Proceeds” on page
17.
|
|
Market for the
common stock
|
Our common stock is quoted on the OTCQB
marketplace under the symbol “GTHP.” See “Market
for Our Common Stock and Related Stockholder Matters” on page
37.
|
|
Risk
factors
|
You should read “Risk Factors” on
page 7 for an explanation of the risks of investing in this
offering.
|
|
|
*
|
Share numbers are
based on shares outstanding as of March 27, 2017 and
exclude:
|
|
|
●
|
125 shares
of common stock reserved for future issuance under our 1995 Stock
Plan;
|
|
|
●
|
16,864,630
shares of common stock reserved for issuance upon conversion of, or
as dividends on, our Series C and Series C1 convertible preferred
stock;
|
|
|
●
|
7,743,293
shares reserved for issuance upon conversion of our outstanding
convertible debt;
|
|
|
●
|
5,738,112
shares of common stock issuable upon the exercise of outstanding
warrants or warrants to be exchanged for outstanding
warrants;
|
|
|
●
|
up to 16,670,000
shares of common stock issuable upon exercise of warrants sold as
part of this offering.
|
RISK
FACTORS
Your investment in shares of our common stock
involves substantial risks. In consultation with your own advisers,
you should carefully consider, among other matters, the factors set
forth below before deciding whether an investment in shares of our
common stock is suitable for you. If any of the risks contained in
this prospectus develop into actual events, our business, financial
condition, liquidity, results of operations and prospects could be
materially and adversely affected, the market price of our common
stock could decline and you may lose all or part of your
investment. Some statements in this prospectus, including
statements in the following risk factors, constitute
forward-looking statements. See “Forward-Looking
Statements” in this prospectus.
Risks
Related to this Offering
As a new investor, you will incur substantial dilution as a result
of this offering and future equity issuances.
Our net tangible
book value deficiency as of December 31, 2016 was
approximately $10.6 million, or $15.79
per share of common stock. Net tangible book value per share
represents total tangible assets less total liabilities, divided by
the number of shares of common stock outstanding. On a pro forma
basis after giving effect to the assumed sale of 5,000 shares of
Series D preferred stock in this offering at a public offering
price of $1,000 per share, and assuming the conversion of all the
shares of Series D preferred stock sold in the offering at an
assumed conversion price of $0.30 per share, which
represents a 25% discount off the last reported sale price for our
common stock on March 27, 2017 (and excluding shares
of common stock issuable upon exercise of the warrants), and after
deducting estimated placement agent fees and estimated offering
expenses payable by us, if you purchase securities in this
offering, you will suffer immediate and substantial dilution of
approximately $0.53 per share in the net tangible book
value of the common stock underlying the Series D preferred stock
that you acquire. See “Dilution” for a more detailed
discussion of the dilution you will incur if you participate in
this offering. In addition to this offering, subject to market
conditions and other factors, it is likely that we will pursue
additional capital to finance our operations. Accordingly, we may
conduct substantial future offerings of equity securities. The
exercise of outstanding options and warrants and future equity
issuances, including future public offerings or future private
placements of equity securities, would result in further dilution
to investors.
The actual offering amount, the offering price and the net proceeds
to us, if any, in this offering may be substantially less than the
amounts set forth above.
Because there
is no minimum offering amount or offering price required as a
condition to closing in this offering, the actual public offering
amount, offering price and net proceeds to us, if any, in this
offering are not presently determinable and may be substantially
less than the amounts set forth above. We are not required to sell
any specific number or dollar amount of the securities offered in
this offering, but the placement agent will use its best efforts to
sell the securities offered.
Sales of a significant number of shares of our common stock in the
public markets, or the perception that such sales could occur,
could depress the market price of our common stock.
If our
stockholders (including those persons who may become stockholders
upon conversion of outstanding convertible securities or exercise
of outstanding warrants or options) sell substantial amounts of our
common stock, or the public market perceives that stockholders
might sell substantial amounts of our common stock, the market
price of our common stock could decline significantly. Such sales
also might make it more difficult for us to sell equity or
equity-related securities in the future at a time and price that
our management deems appropriate.
We will have broad discretion over the use of the proceeds of this
offering and may not realize a return.
We will have
considerable discretion in the application of the net cash proceeds
of this offering. We intend to use the net cash proceeds to
increase inventory of LuViva to meet current demand for the
product, and to support general working capital and operations.
However, we will retain broad discretion over the use of the net
proceeds and may use the money for other corporate purposes. We may
use the net cash proceeds for purposes that do not yield a
significant return, if any, for our stockholders.
There is no public market for the Series D preferred stock or
warrants being offered in this offering.
There is no
established public trading market for the Series D preferred stock
or warrants being offered in this offering and we do
not expect a market to develop for the securities. In
addition, we do not intend to apply for listing of the
securities on any securities exchange or expect the
securities to trade on the OTCQB marketplace. Without
an active market, the liquidity of the securities will
be limited.
The Series D preferred stock provides for the payment of dividends
in cash, but we are not currently permitted to pay such
dividends.
Each share of
Series D preferred stock will be entitled to receive cumulative
dividends until the second anniversary of the date of issuance. The
dividends are payable in cash, out of any funds legally available
for such purpose. We will not be permitted to pay the dividend in
cash unless we are legally permitted to do so under Delaware law,
which requires cash to be available from surplus or net profits,
neither of which we currently have available. Additionally, we are
also subject to certain restrictions pursuant to the terms of our
outstanding indebtedness, which prohibit us from paying cash
dividends or distributions on our capital stock. As such, we do not
expect to have cash available to pay the dividends on the Series D
preferred stock or to be permitted to make such payments under the
terms of our indebtedness. In such a case, the only way you will
realize the benefits of accrued but unpaid dividends will be upon
conversion, liquidation, or redemption of your shares of Series D
preferred stock.
Risks Related to Our
Business
Although we
are required to raise funds in this offering in order to continue
as a going concern, there is no assurance that such funds can be
raised on terms that we would find acceptable, on a timely basis,
or at all.
Additional debt or
equity financing is required for us to continue as a going concern.
If this offering is unsuccessful or insufficient, we may seek to
obtain additional funds for the financing of our cervical cancer
detection business through additional debt or equity financings
and/or new collaborative arrangements. Any required additional
funding may not be available on terms attractive to us, on a timely
basis, or at all.
If we cannot
obtain additional funds or achieve profitability, we may not be
able to continue as a going concern.
Because we must
obtain additional funds through financing transactions or through
new collaborative arrangements in order to grow the revenues of our
cervical cancer detection product line, there exists substantial
doubt about our ability to continue as a going concern. Therefore,
it will be necessary to raise additional funds. There can be no
assurance that we will be able to raise these additional funds. If
we do not secure additional funding when needed, we will be unable
to conduct all of our product development efforts as planned, which
may cause us to alter our business plan in relation to the
development of our products. Even if we obtain additional funding,
we will need to achieve profitability thereafter.
Our independent
registered public accountants’ report on our consolidated
financial statements as of and for the year ended December 31,
2016, indicated that there was substantial doubt about
our ability to continue as a going concern because we had suffered
recurring losses from operations and had an accumulated deficit of
$127.6 million at December 31, 2016
summarized as follows:
|
Accumulated deficit,
from inception to 12/31/2014
|
$
|
113.1 million
|
|
Preferred dividends and deemed
dividends
|
$
|
2.6 million
|
|
Net Loss for fiscal year 2015, ended
12/31/2015
|
$
|
6.9 million
|
|
Accumulated deficit, from inception to
12/31/2015
|
$
|
122.6 million
|
|
Preferred
dividends
|
$
|
1.0 million
|
|
Net Loss for year to date ended
12/31/2016
|
$
|
4.0 million
|
|
Accumulated deficit, from inception to
12/31/2016
|
$
|
127.6
million
|
Our management has
implemented reductions in operating expenditures and reductions in
some development activities. We have determined to make cervical
cancer detection the focus of our business. We are managing the
development of our other programs only when funds are made
available to us via grants or contracts with government entities or
strategic partners. However, there can be no assurance that we will
be able to successfully implement or continue these
plans.
If we cannot
obtain additional funds when needed, we will not be able to
implement our business plan.
We require
substantial additional capital to develop our products, including
completing product testing and clinical trials, obtaining all
required regulatory approvals and clearances, beginning and scaling
up manufacturing, and marketing our products. We have historically
financed our operations though the public and private sale of debt
and equity, funding from collaborative arrangements, and grants.
Any failure to achieve adequate funding in a timely fashion would
delay our development programs and could lead to abandonment of our
business plan. To the extent we cannot obtain additional funding,
our ability to continue to manufacture and sell our current
products, or develop and introduce new products to market, will be
limited. Further, financing our operations through the public or
private sale of debt or equity may involve restrictive covenants or
other provisions that could limit how we conduct our business or
finance our operations. Financing our operations through
collaborative arrangements generally means that the obligations of
the collaborative partner to fund our expenditures are largely
discretionary and depend on a number of factors, including our
ability to meet specified milestones in the development and testing
of the relevant product. We may not be able to obtain an acceptable
collaboration partner, and even if we do, we may not be able to
meet these milestones, or the collaborative partner may not
continue to fund our expenditures.
We do not have
a long operating history, especially in the cancer detection field,
which makes it difficult to evaluate our
business.
Although we have
been in existence since 1992, we have only recently begun to
commercialize our cervical cancer detection technology. Because
limited historical information is available on our revenue trends
and manufacturing costs, it is difficult to evaluate our
business. Our prospects must be considered in light of the
substantial risks, expenses, uncertainties and difficulties
encountered by entrants into the medical device industry, which is
characterized by increasing intense competition and a high failure
rate.
We have a
history of losses, and we expect losses to
continue.
We have never been
profitable and we have had operating losses since our inception. We
expect our operating losses to continue as we continue to expend
substantial resources to complete commercialization of our
products, obtain regulatory clearances or approvals; build our
marketing, sales, manufacturing and finance capabilities, and
conduct further research and development. The further development
and commercialization of our products will require substantial
development, regulatory, sales and marketing, manufacturing and
other expenditures. We have only generated limited revenues from
product sales. Our accumulated deficit was approximately
$127.6 million at December 31,
2016.
Our ability to
sell our products is controlled by government regulations, and we
may not be able to obtain any necessary clearances or
approvals.
The design,
manufacturing, labeling, distribution and marketing of medical
device products are subject to extensive and rigorous government
regulation in most of the markets in which we sell, or plan to
sell, our products, which can be expensive and uncertain and can
cause lengthy delays before we can begin selling our products in
those markets.
In foreign
countries, including European countries, we are subject to
government regulation, which could delay or prevent our ability to
sell our products in those
jurisdictions.
In order for us to
market our products in Europe and some other international
jurisdictions, we and our distributors and agents must obtain
required regulatory registrations or approvals. We must also comply
with extensive regulations regarding safety, efficacy and quality
in those jurisdictions. We may not be able to obtain the required
regulatory registrations or approvals, or we may be required to
incur significant costs in obtaining or maintaining any regulatory
registrations or approvals we receive. Delays in obtaining any
registrations or approvals required for marketing our products,
failure to receive these registrations or approvals, or future loss
of previously obtained registrations or approvals would limit our
ability to sell our products internationally. For example,
international regulatory bodies have adopted various regulations
governing product standards, packaging requirements, labeling
requirements, import restrictions, tariff regulations, duties and
tax requirements. These regulations vary from country to country.
In order to sell our products in Europe, in 2017 we
must undergo an inspection and re-file for ISO
13485:2003 and the CE MARK, which is an
international symbol of quality and compliance with applicable
European medical device directives. Failure to maintain ISO
13485:2003 certification or CE mark certification or other
international regulatory approvals would prevent us from selling in
some countries in the European Union.
As part of our
agreement with SMI, SMI will underwrite the cost of securing
approval of LuViva with the CFDA and is responsible for securing
CFDA approval by July 2019, although we cannot be assured that SMI
will successfully secure such approval without delays or conditions
unacceptable to us, or at all.
In the United
States, we are subject to regulation by the U.S. FDA,
which could prevent us from selling our products
domestically.
In order for us to
market our products in the United States, we must obtain clearance
or approval from the U.S. FDA. We cannot be sure
that:
|
|
●
|
we, or any collaborative partner, will make
timely filings with the
U.S.
FDA;
|
|
|
●
|
the
U.S.
FDA will act favorably or quickly on these
submissions;
|
|
|
●
|
we will not be required to submit additional
information or perform additional clinical studies; or
|
|
|
●
|
we will not face other significant difficulties
and costs necessary to obtain
U.S.
FDA clearance or approval.
|
It can take several
years from initial filing of a PMA application and require the
submission of extensive supporting data and clinical
information. The
U.S.
FDA may impose strict labeling or other requirements as a condition
of its clearance or approval, any of which could limit our ability
to market our products domestically. Further, if we wish to modify
a product after
U.S.
FDA approval of a PMA application, including changes in indications
or other modifications that could affect safety and efficacy,
additional clearances or approvals will be required from
the
U.S.
FDA. Any request by the
U.S.
FDA for additional data, or any requirement by
the
U.S.
FDA that we conduct additional clinical studies, could result in a
significant delay in bringing our products to market domestically
and require substantial additional research and other expenditures.
Similarly, any labeling or other conditions or restrictions imposed
by the
U.S.
FDA could hinder our ability to effectively market our products
domestically. Further, there may be new
U.S.
FDA policies or changes in
U.S.
FDA policies that could be adverse to us.
Even if we
obtain clearance or approval to sell our products, we are subject
to ongoing requirements and inspections that could lead to the
restriction, suspension or revocation of our
clearance.
We, as well as any
potential collaborative partners, will be required to adhere to
applicable regulations in the markets in which we operate and sell
our products, regarding good manufacturing practice, which include
testing, control, and documentation requirements. Ongoing
compliance with good manufacturing practice and other applicable
regulatory requirements will be strictly enforced applicable
regulatory agencies. Failure to comply with these regulatory
requirements could result in, among other things, warning letters,
fines, injunctions, civil penalties, recall or seizure of products,
total or partial suspension of production, failure to obtain
premarket clearance or premarket approval for devices, withdrawal
of approvals previously obtained, and criminal prosecution. The
restriction, suspension or revocation of regulatory approvals or
any other failure to comply with regulatory requirements would
limit our ability to operate and could increase our
costs.
We depend on a
limited number of customers and any reduction, delay or
cancellation of an order from these customers or the loss of any of
these customers could cause our revenue to
decline.
Each year we have
had one or a few customers that have accounted for substantially
all of our limited revenues. As a result, the termination of a
purchase order with any one of these customers may result in the
loss of substantially all of our revenues. We are constantly
working to develop new relationships with existing or new
customers, but despite these efforts we may not be successful at
generating new orders to maintain similar revenues as current
purchase orders are filled. In addition, since a significant
portion of our revenues is derived from a relatively few customers,
any financial difficulties experienced by any one of these
customers, or any delay in receiving payments from any one of these
customers, could have a material adverse effect on our business,
results of operations, financial condition and cash
flows.
To
successfully market and sell our products internationally, we must
address many issues with which we have limited
experience.
All of our sales of
LuViva to date have been to customers outside of the United States.
We expect that substantially all of our business will continue to
come from sales in foreign markets, through increased penetration
in countries where we currently sell LuViva, combined with
expansion into new international markets. However, international
sales are subject to a number of risks, including:
●
difficulties in staffing and managing international
operations;
●
difficulties in penetrating markets in which our competitors’
products may be more established;
● reduced or
no protection for intellectual property rights in some
countries;
● export
restrictions, trade regulations and foreign tax laws;
● fluctuating
foreign currency exchange rates;
● foreign
certification and regulatory clearance or approval
requirements;
●
difficulties in developing effective marketing campaigns for
unfamiliar, foreign countries;
● customs
clearance and shipping delays;
● political
and economic instability; and
● preference
for locally produced products.
If one or more of
these risks were realized, it could require us to dedicate
significant resources to remedy the situation, and even if we are
able to find a solution, our revenues may still
decline.
To market and
sell LuViva internationally, we depend on distributors and they may
not be successful.
We currently depend
almost exclusively on third-party distributors to sell and service
LuViva internationally and to train our international customers,
and if these distributors terminate their relationships with us or
under-perform, we may be unable to maintain or increase our level
of international revenue. We will also need to engage additional
international distributors to grow our business and expand the
territories in which we sell LuViva. Distributors may not commit
the necessary resources to market, sell and service LuViva to the
level of our expectations. If current or future distributors do not
perform adequately, or if we are unable to engage distributors in
particular geographic areas, our revenue from international
operations will be adversely affected
Our success
largely depends on our ability to maintain and protect the
proprietary information on which we base our
products.
Our success depends
in large part upon our ability to maintain and protect the
proprietary nature of our technology through the patent process, as
well as our ability to license from others patents and patent
applications necessary to develop our products. If any of our
patents are successfully challenged, invalidated or circumvented,
or our right or ability to manufacture our products was to be
limited, our ability to continue to manufacture and market our
products could be adversely affected. In addition to patents, we
rely on trade secrets and proprietary know-how, which we seek to
protect, in part, through confidentiality and proprietary
information agreements. The other parties to these agreements may
breach these provisions, and we may not have adequate remedies for
any breach. Additionally, our trade secrets could otherwise become
known to or be independently developed by competitors.
As of March
27, 2017, we have been issued, or have rights
to, 25 U.S. patents (including those under license).
In addition, we have filed for, or have rights to,
five U.S. patents (including those under license) that
are still pending. There are additional international patents and
pending applications. One or more of the patents we hold directly
or license from third parties, including those for our cervical
cancer detection products, may be successfully challenged,
invalidated or circumvented, or we may otherwise be unable to rely
on these patents. These risks are also present for the process we
use or will use for manufacturing our products. In addition, our
competitors, many of whom have substantial resources and have made
substantial investments in competing technologies, may apply for
and obtain patents that prevent, limit or interfere with our
ability to make, use and sell our products, either in the United
States or in international markets.
The medical device
industry has been characterized by extensive litigation regarding
patents and other intellectual property rights. In addition, the
U.S. Patent and Trademark Office, or USPTO, may institute
interference proceedings. The defense and prosecution of
intellectual property suits, USPTO proceedings and related legal
and administrative proceedings are both costly and time consuming.
Moreover, we may need to litigate to enforce our patents, to
protect our trade secrets or know-how, or to determine the
enforceability, scope and validity of the proprietary rights of
others. Any litigation or interference proceedings involving us may
require us to incur substantial legal and other fees and expenses
and may require some of our employees to devote all or a
substantial portion of their time to the proceedings. An adverse
determination in the proceedings could subject us to significant
liabilities to third parties, require us to seek licenses from
third parties or prevent us from selling our products in some or
all markets. We may not be able to reach a satisfactory settlement
of any dispute by licensing necessary patents or other intellectual
property. Even if we reached a settlement, the settlement process
may be expensive and time consuming, and the terms of the
settlement may require us to pay substantial royalties. An adverse
determination in a judicial or administrative proceeding or the
failure to obtain a necessary license could prevent us from
manufacturing and selling our products.
We may not be
able to generate sufficient sales revenues to sustain our growth
and strategy plans.
Our cervical cancer
diagnostic activities have been financed to date through a
combination of government grants, strategic partners and direct
investment. Growing revenues for this product is the main focus of
our business. In order to effectively market the cervical cancer
detection product, additional capital will be needed.
Additional product
lines involve the modification of the cervical cancer detection
technology for use in other cancers. These product lines are only
in the earliest stages of research and development and are
currently not projected to reach market for several years. Our goal
is to receive enough funding from government grants and contracts,
as well as payments from strategic partners, to fund development of
these product lines without diverting funds or other necessary
resources from the cervical cancer program.
Because our
products, which use different technology or apply technology in
different ways than other medical devices, are or will be new to
the market, we may not be successful in launching our products and
our operations and growth would be adversely
affected.
Our products are
based on new methods of cancer detection. If our products do not
achieve significant market acceptance, our sales will be limited
and our financial condition may suffer. Physicians and individuals
may not recommend or use our products unless they determine that
these products are an attractive alternative to current tests that
have a long history of safe and effective use. To date, our
products have been used by only a limited number of people, and few
independent studies regarding our products have been published. The
lack of independent studies limits the ability of doctors or
consumers to compare our products to conventional
products.
If we are
unable to compete effectively in the highly competitive medical
device industry, our future growth and operating results will
suffer.
The medical device
industry in general and the markets in which we expect to offer
products in particular, are intensely competitive. Many of our
competitors have substantially greater financial, research,
technical, manufacturing, marketing and distribution resources than
we do and have greater name recognition and lengthier operating
histories in the health care industry. We may not be able to
effectively compete against these and other competitors. A number
of competitors are currently marketing traditional laboratory-based
tests for cervical cancer screening and diagnosis. These tests are
widely accepted in the health care industry and have a long history
of accurate and effective use. Further, if our products are not
available at competitive prices, health care administrators who are
subject to increasing pressures to reduce costs may not elect to
purchase them. Also, a number of companies have announced that they
are developing, or have introduced, products that permit
non-invasive and less invasive cancer detection. Accordingly,
competition in this area is expected to increase.
Furthermore, our
competitors may succeed in developing, either before or after the
development and commercialization of our products, devices and
technologies that permit more efficient, less expensive
non-invasive and less invasive cancer detection. It is also
possible that one or more pharmaceutical or other health care
companies will develop therapeutic drugs, treatments or other
products that will substantially reduce the prevalence of cancers
or otherwise render our products obsolete.
We have
limited manufacturing experience, which could limit our
growth.
We do not have
manufacturing experience that would enable us to make products in
the volumes that would be necessary for us to achieve significant
commercial sales, and we rely upon our suppliers. In addition, we
may not be able to establish and maintain reliable, efficient, full
scale manufacturing at commercially reasonable costs in a timely
fashion. Difficulties we encounter in manufacturing scale-up, or
our failure to implement and maintain our manufacturing facilities
in accordance with good manufacturing practice regulations,
international quality standards or other regulatory requirements,
could result in a delay or termination of production. In the past,
we have had substantial difficulties in establishing and
maintaining manufacturing for our products and those difficulties
impacted our ability to increase sales. Companies often encounter
difficulties in scaling up production, including problems involving
production yield, quality control and assurance, and shortages of
qualified personnel.
Since we rely
on sole source suppliers for several of the components used in our
products, any failure of those suppliers to perform would hurt our
operations.
Several of the
components used in our products or planned products, are available
from only one supplier, and substitutes for these components could
not be obtained easily or would require substantial modifications
to our products. Any significant problem experienced by one of our
sole source suppliers may result in a delay or interruption in the
supply of components to us until that supplier cures the problem or
an alternative source of the component is located and qualified.
Any delay or interruption would likely lead to a delay or
interruption in our manufacturing operations. For our products that
require premarket approval, the inclusion of substitute components
could require us to qualify the new supplier with the appropriate
government regulatory authorities. Alternatively, for our products
that qualify for premarket notification, the substitute components
must meet our product specifications.
Because we
operate in an industry with significant product liability risk, and
we have not specifically insured against this risk, we may be
subject to substantial claims against our
products.
The development,
manufacture and sale of medical products entail significant risks
of product liability claims. We currently have no product liability
insurance coverage beyond that provided by our general liability
insurance. Accordingly, we may not be adequately protected from any
liabilities, including any adverse judgments or settlements, we
might incur in connection with the development, clinical testing,
manufacture and sale of our products. A successful product
liability claim or series of claims brought against us that result
in an adverse judgment against or settlement by us in excess of any
insurance coverage could seriously harm our financial condition or
reputation. In addition, product liability insurance is expensive
and may not be available to us on acceptable terms, if at
all.
The
availability of third party reimbursement for our products is
uncertain, which may limit consumer use and the market for our
products.
In the United
States and elsewhere, sales of medical products are dependent, in
part, on the ability of consumers of these products to obtain
reimbursement for all or a portion of their cost from third-party
payors, such as government and private insurance plans. Any
inability of patients, hospitals, physicians and other users of our
products to obtain sufficient reimbursement from third-party payors
for our products, or adverse changes in relevant governmental
policies or the policies of private third-party payors regarding
reimbursement for these products, could limit our ability to sell
our products on a competitive basis. We are unable to predict what
changes will be made in the reimbursement methods used by
third-party health care payors. Moreover, third-party payors are
increasingly challenging the prices charged for medical products
and services, and some health care providers are gradually adopting
a managed care system in which the providers contract to provide
comprehensive health care services for a fixed cost per person.
Patients, hospitals and physicians may not be able to justify the
use of our products by the attendant cost savings and clinical
benefits that we believe will be derived from the use of our
products, and therefore may not be able to obtain third-party
reimbursement.
Reimbursement and
health care payment systems in international markets vary
significantly by country and include both government-sponsored
health care and private insurance. We may not be able to obtain
approvals for reimbursement from these international third-party
payors in a timely manner, if at all. Any failure to receive
international reimbursement approvals could have an adverse effect
on market acceptance of our products in the international markets
in which approvals are sought.
We have a
substantial amount of indebtedness, which may adversely affect our
cash flow and our ability to operate our
business.
Our outstanding
indebtedness, including ordinary course accounts payable and
accrued payroll liabilities, was $5.3 million at
December 31, 2016.
The terms of our
indebtedness could have negative consequences to us, such
as:
|
|
●
|
we may be unable to obtain additional financing
to fund working capital, operating losses, capital expenditures or
acquisitions on terms acceptable to us, or at all;
|
|
|
●
|
the amount of our interest expense may increase
if we are unable to make payments when due;
|
|
|
●
|
our assets might be subject to foreclosure if we
default on our secured debt (see “—
We have outstanding debt that is
collateralized by a general security interest in all of our assets,
including our intellectual property. If we were to fail to repay
the debt when due, the holders would have the right to foreclose on
these assets.
”);
|
|
|
●
|
our vendors or employees may, and some have,
instituted proceedings to collect on amounts owed
them;
|
|
|
●
|
we have to use a substantial portion of our cash
flows from operations to repay our indebtedness, including ordinary
course accounts payable and accrued payroll liabilities, which
reduces the amount of money we have for future operations, working
capital, inventory, expansion, or general corporate or other
business activities; and
|
|
|
●
|
we may be unable to refinance our indebtedness
on terms acceptable to us, or at all.
|
Our ability to meet
our expenses and debt obligations will depend on our future
performance, which will be affected by financial, business,
economic, regulatory and other factors. We will be unable to
control many of these factors, such as economic conditions. We
cannot be certain that our earnings will be sufficient to allow us
to pay the principal and interest on our debt and meet any other
obligations. If we do not have enough money to service our debt, we
may be required, but unable, to refinance all or part of our
existing debt, sell assets, borrow money or raise equity on terms
acceptable to us, if at all.
We have
outstanding debt that is collateralized by a general security
interest in all of our assets, including our intellectual property.
If we were to fail to repay the debt when due, the holders would
have the right to foreclose on these
assets.
We
have notes outstanding that are collateralized by a
security interest in our current and future inventory and accounts
receivable. We also have a note outstanding that is
collateralized by a security interest in all of our assets,
including our intellectual property. When the debt is repaid, the
holders’ security interests on our assets will be
extinguished. However, if an event of default occurs under the
notes prior to their repayment, the holders may exercise their
rights to foreclose on these secured assets for the payment of
these obligations. Under “cross-default” provisions in
each of the notes, an event of default under one note is
automatically an event of default under the other notes. Any such
default and resulting foreclosure would have a material adverse
effect on our business, financial condition and results of
operations.
We are subject
to restrictive covenants under the terms of our outstanding secured
debt. If we were to default under the terms of these covenants, the
holders would have the right to foreclose on the assets that secure
the debt.
The instruments
governing our outstanding secured debt contain restrictive
covenants. For example, our senior secured convertible note
prohibits us from incurring additional indebtedness for borrowed
money, repurchasing any outstanding shares of our common stock, or
paying any dividends on our capital stock, in each case without the
note holder's prior written consent, If we were to breach any of
these covenants, the holder could declare an event of default on
the note, and exercise its rights to foreclose on the assets
securing the note.
Our success
depends on our ability to attract and retain scientific, technical,
managerial and finance personnel.
Our ability to
operate successfully and manage our future growth depends in
significant part upon the continued service of key scientific,
technical, managerial and finance personnel, as well as our ability
to attract and retain additional highly qualified personnel in
these fields. We may not be able to attract and retain key
employees when necessary, which would limit our operations and
growth. Only our Chief Executive Officer and our Senior Vice
President of Engineering have employment contracts with us, and
none of our employees are covered by key person or similar
insurance. In addition, if we are able to successfully develop and
commercialize our products, we will need to hire additional
scientific, technical, marketing, managerial and finance personnel.
We face intense competition for qualified personnel in these areas,
many of whom are often subject to competing employment
offers.
We are
significantly influenced by our directors, executive officers and
their affiliated entities.
Our directors,
executive officers and entities affiliated with them controlled
approximately 1% of the voting power of our
outstanding common stock as of March 27, 2017. These
stockholders, acting together, would be able to exert significant
influence on substantially all matters requiring approval by our
stockholders, including the election of directors and the approval
of mergers and other business combination
transactions.
Certain
provisions of our certificate of incorporation that authorize the
issuance of additional shares of preferred stock may make it more
difficult for a third party to effect a change in
control.
Our certificate of
incorporation authorizes our board of directors to issue up to 5.0
million shares of preferred stock. Our undesignated shares of
preferred stock may be issued in one or more series, the terms of
which may be determined by the board without further stockholder
action. These terms may include, among other terms, voting rights,
including the right to vote as a series on particular matters,
preferences as to liquidation and dividends, repurchase rights,
conversion rights, redemption rights and sinking fund provisions.
The issuance of any preferred stock could diminish the rights of
holders of our common stock, and therefore could reduce the value
of our common stock. In addition, specific rights granted to future
holders of preferred stock could be used to restrict our ability to
merge with or sell assets to a third party. The ability of our
board to issue preferred stock could make it more difficult, delay,
discourage, prevent or make it more costly to acquire or effect a
change in control, which in turn could prevent our stockholders
from recognizing a gain in the event that a favorable offer is
extended and could materially and negatively affect the market
price of our common stock.
Risks
Related to Our Common Stock
On November 7,
2016, a 1:800 reverse stock split of all of our issued and
outstanding common stock was implemented. There are risks
associated with a reverse stock split.
On November 7,
2016, a 1:800 reverse stock split of all of our issued and
outstanding common stock was implemented. As a result of the
reverse stock split, every 800 shares of issued and outstanding
common stock were converted into 1 share of common
stock. All fractional shares created by the reverse stock split
were rounded to the nearest whole share. The number of authorized
shares of common stock did not change.
There are certain
risks associated with the reverse stock split, including the
following:
|
|
●
|
We have additional authorized shares of common
stock that the board could issue in future without stockholder
approval, and such additional shares could be issued, among other
purposes, in financing transactions or to resist or frustrate a
third-party transaction that is favored by a majority of the
independent stockholders. This could have an anti-takeover effect,
in that additional shares could be issued, within the limits
imposed by applicable law, in one or more transactions that could
make a change in control or takeover of us more
difficult.
|
|
●
|
There can be no assurance that the reverse stock
split will achieve the benefits that we hope it will achieve. The
total market capitalization of our common stock after the reverse
stock split may be lower than the total market capitalization
before the reverse stock split.
|
The reverse
stock split may decrease the liquidity of the shares of our common
stock.
The liquidity of
the shares of our common stock may be affected adversely by the
reverse stock split given the reduced number of shares that were
outstanding immediately following the reverse stock split,
especially if the market price of our common stock does not
increase as a result of the reverse stock split. In addition, the
reverse stock split may have increased the number of stockholders
who own odd lots of our common stock, creating the potential for
such stockholders to experience an increase in the cost of selling
their shares and greater difficulty effecting such
sales.
Following the
reverse stock split, the resulting market price of our common stock
may not attract new investors, including institutional investors,
and may not satisfy the investing requirements of those investors.
Consequently, the trading liquidity of our common stock may not
improve.
Although we believe
that a higher market price of our common stock may help generate
greater or broader investor interest, there can be no assurance
that the reverse stock split will result in a share price that will
attract new investors, including institutional investors. In
addition, there can be no assurance that the market price of our
common stock will satisfy the investing requirements of those
investors. As a result, the trading liquidity of our common stock
may not necessarily improve.
The number of
shares of our common stock issuable upon the conversion of our
outstanding convertible debt and preferred stock or exercise of
outstanding warrants and options (excluding the convertible stock
or warrants in this offering) is
substantial.
As
ofMarch 27, 2017, our outstanding convertible debt was
convertible into an aggregate of 7,743,293 shares of
our common stock, and the outstanding shares of our Series C and
Series C1 preferred stock were convertible into an aggregate of
16,864,630 shares of common stock. Also, as of that
date we had warrants outstanding that were exercisable for an
aggregate of 5,738,112 shares and
outstanding options to purchase 125 shares. The shares
of common stock issuable upon conversion or exercise of these
securities would have constituted approximately 96.0%
of the total number of shares of common stock then issued and
outstanding.
Further, under the
terms of our convertible debt and preferred stock, as well as
certain of our outstanding warrants, the conversion price or
exercise price, as the case may be, could be adjusted downward,
causing substantial dilution. See “—
Adjustments to the conversion price
of our convertible debt and preferred stock, and the
exercise price for certain of our warrants (including the
convertible stock and warrants in this offering), will dilute the
ownership interests of our existing
stockholders
.”
Adjustments to the conversion price of our convertible debt
and preferred stock, and the exercise price for
certain of our warrants (including the convertible stock and
warrants in this offering), will dilute the ownership
interests of our existing stockholders.
Under
the terms of a portion of our convertible debt and preferred
stock, the conversion price fluctuates with the market price
of our common stock. Additionally, under the terms of our Series C
preferred stock and the preferred stock part of this offering,any
dividends we choose to pay in shares of our common stock will be
calculated based on the then-current market price of our common
stock. Accordingly, if the market price of our common stock
decreases, the number of shares of our common stock issuable upon
conversion of the convertible debt or upon payment of dividends on
our outstanding preferred stock and preferred stock part of this
offering will increase, and may result in the issuance of a
significant number of additional shares of our common
stock.
Under
the terms of our preferred stock (including the preferred stock
part of this offering) and certain of our convertible notes
and outstanding warrants, the conversion price
or exercise price will be lowered if we issue common stock
at a per share price below the then- conversion
price or then-exercise price for those securities.
Reductions in the conversion price or exercise price
would result in the issuance of a significant number of additional
shares of our common stock upon conversion or
exercise, which would result in dilution in the value of the
shares of our outstanding common stock and the voting power
represented thereby.
Our stock is
thinly traded, so you may be unable to sell at or near ask prices
or at all.
The shares of our
common stock are dually quoted on the OTCBB and the OTCQB. Shares
of our common stock are thinly traded, meaning that the number of
persons interested in purchasing our common shares at or near ask
prices at any given time may be relatively small or non-existent.
This situation is attributable to a number of factors,
including:
|
|
●
|
we are a small company that is relatively
unknown to stock analysts, stock brokers, institutional investors
and others in the investment community that generate or influence
sales volume; and
|
|
|
●
|
stock analysts, stock brokers and institutional
investors may be risk-averse and be reluctant to follow a company
such as ours that faces substantial doubt about its ability to
continue as a going concern or to purchase or recommend the
purchase of our shares until such time as we became more
viable.
|
As a consequence,
our stock price may not reflect an actual or perceived value. Also,
there may be periods of several days or more when trading activity
in our shares is minimal or non-existent, as compared to a seasoned
issuer that has a large and steady volume of trading activity that
will generally support continuous sales without an adverse effect
on share price. A broader or more active public trading market for
our common shares may not develop or if developed, may not be
sustained. Due to these conditions, you may not be able to sell
your shares at or near ask prices or at all if you need money or
otherwise desire to liquidate your shares.
Trading in our
common stock is subject to special sales practices and may be
difficult to sell.
Our common stock is
subject to the SEC’s “penny stock”
rule, which imposes special sales practice requirements upon
broker-dealers who sell such securities to persons other than
established customers or accredited investors. Penny stocks are
generally defined to be an equity security that has a market price
of less than $5.00 per share. For transactions covered by the rule,
the broker-dealer must make a special suitability determination for
the purchaser and receive the purchaser’s written agreement
to the transaction prior to the sale. Consequently, the rule may
affect the ability of broker-dealers to sell our securities and
also may affect the ability of our stockholders to sell their
securities in any market that might develop.
Stockholders should
be aware that, according to SEC, the market for penny
stocks has suffered from patterns of fraud and abuse. Such patterns
include:
|
|
●
|
control of the market for the security by one or
a few broker-dealers that are often related to the promoter or
issuer;
|
|
|
●
|
manipulation of prices through prearranged
matching of purchases and sales and false and misleading press
releases;
|
|
|
●
|
“boiler room” practices involving
high-pressure sales tactics and unrealistic price projections by
inexperienced sales persons;
|
|
|
●
|
excessive and undisclosed bid-ask differentials
and markups by selling broker-dealers; and
|
|
|
●
|
the wholesale dumping of the same securities by
promoters and broker-dealers after prices have been manipulated to
a desired level, along with the resulting inevitable collapse of
those prices and with consequent investor losses.
|
Our management is
aware of the abuses that have occurred historically in the penny
stock market. Although we do not expect to be in a position to
dictate the behavior of the market or of broker-dealers who
participate in the market, management will strive within the
confines of practical limitations to prevent the described patterns
from being established with respect to our common
stock.
Our need to
raise additional capital in the near future or to use our equity
securities for payments could have a dilutive effect on your
investment.
In order to
continue operations, we will need to raise additional capital. We
may attempt to raise capital through the public or private sale of
our common stock or securities convertible into or exercisable for
our common stock. In addition, from time to time we have issued our
common stock or warrants in lieu of cash payments. If we sell
additional shares of our common stock or other equity securities,
or issue such securities in respect of other claims or
indebtedness, such sales or issuances will further dilute the
percentage of our equity that you own. Depending upon the price per
share of securities that we sell or issue in the future, if any,
your interest in us could be further diluted by any adjustments to
the number of shares and the applicable exercise price required
pursuant to the terms of the agreements under which we previously
issued convertible securities.
USE
OF PROCEEDS
We estimate that
the net cash proceeds to us from the sale of the securities offered
by this prospectus, excluding the proceeds, if any, from the
exercise of the warrants included in the offering, will be
approximately $4.16 million, assuming the sale by us of 5,000
shares of Series D preferred stock and warrants at an assumed
public offering price of $1,000 per share and after deducting the
estimated placement agent fees and estimated offering expenses
payable by us. However, this is a best efforts offering with no
minimum, and we may not sell all or any of the securities; as a
result, we may receive significantly less in net cash proceeds, and
the net cash proceeds received may not be sufficient to continue to
operate our business. In addition, expected net proceeds include a
non-cash benefit for the extinguishment of up to $107,500 in
outstanding principal and interest on simple interest notes from
one of our investors. See “Summary—Recent
Developments—Cash Advances.”
We intend to apply
any proceeds received in connection with the offering to increase
inventory of LuViva to meet current demand for the product, to
repay our secured promissory note in full and repurchase
approximately $43,000 in shares of Series C preferred stock,
support our efforts to re-file for U.S. FDA approval of
LuViva, and to support general working capital and
operations. However, we will retain broad discretion over the use
of the net proceeds and may use the money for other corporate
purposes.
The following table
summarizes our currently estimated and intended use of net cash
proceeds if we receive the maximum amount of proceeds to be
potentially obtained in this offering, which we expect would
provide funding for our operations for approximately 24 months. The
data in the table set forth below excludes any proceeds we could
receive from the exercise of the warrants to be issued in this
offering.
|
Expected Use for
Net Cash Proceeds
|
Expected Amount
of Net Cash Proceeds($ in
000s)
|
Expected %
of
Net Cash
Proceeds
|
|
Increase inventory
of LuViva advanced cervical device
|
$
1,750,000
|
42.1
%
|
|
Repayment of
Secured Promissory Note and repurchase approximately $43,000 of
shares of Series C preferred stock (approximately 30
shares)
|
260,000
|
6.3
%
|
|
Support Efforts to
Re-File for U.S. FDA approval of LuViva advanced cervical
device
|
1,000,000
|
24.0
%
|
|
General working
capital and operations
|
1,150,000
|
27.6
%
|
|
Total
|
$
4,160,000
|
100
%
|
If the net cash
proceeds of this offering are less than the maximum, we expect to
spend the net cash proceeds in the following order of priority:
first, to use $260,000 to repay the secured promissory note in full
and repurchase approximately $43,000 of shares of Series C
preferred stock, second, to increase inventory, third, to use
approximately $200,000 to support our efforts to re-file for U.S.
FDA approval of LuViva (as further described below), and fourth for
general working capital and operations.
Based on our recent
communication with the U.S. FDA, and should the maximum $5 million
be raised in this offering, we intend to use up to $1.0 million of
the net proceeds over the next 18-24 months to accomplish the
following:
(1)
Write and submit to
the U.S. FDA a new protocol for the confirmatory
study
(2)
Engage regulatory
counsel to assist with the approval process
(3)
Meet with the U.S.
FDA if needed to finalize the protocol and identify the least
burdensome regulatory pathway
(4)
Identify clinical
sites for the study
(5)
Execute clinical
trial agreements for qualified clinical sites
(6)
Conduct the study
and analyze the results
(7)
Submit the study
results and supporting information to the U.S.
FDA
We estimate that
approximately $200,000 of the projected $1 million will be needed
to accomplish goals (1) through (4), with the majority of the costs
(estimated at $800,000) occurring when we engage clinical sites,
enroll patients, analyze results, and submit them to the U.S. FDA.
If the amount raised in this offering results in net proceeds
insufficient to conduct the actual study, we intend to use a
portion of the proceeds to accomplish goals (1) through (4) and
will seek additional funds later in 2017 or in 2018 to complete the
study, analyze the results and submit them to the U.S. FDA,
although there can be no assurance that such funds will be readily
available when needed.
See
“Description of Securities—Secured Promissory
Note” for information on the interest rate and maturity of
our secured promissory note.
If a warrant holder
elects to exercise the warrants issued in this offering, we may
also receive proceeds from the exercise of the warrants. We cannot
predict when or if the warrants will be exercised. It is possible
that the warrants may expire and may never be
exercised.
DILUTION
Our net
tangible book value deficiency as of December 31, 2016
was approximately $10.6 million, or
$15.79 per share of common stock. “Net tangible
book value” represents total tangible assets less total
liabilities. “Net tangible book value per share”
represents net tangible book value divided by the total number of
shares of common stock outstanding.
After
giving effect to the assumed sale of 5,000 shares of Series D
preferred stock and accompanying warrants in this offering at a
public offering price of $1,000 per share, and assuming the
conversion of all the shares of Series D preferred stock sold in
the offering at an assumed conversion price of $0.30
per share, which represents a 25% discount off the last reported
sale price for our common stock on March 27, 2017 (and
excluding shares of common stock issuable upon exercise of
warrants), and after deducting estimated placement agent fees and
estimated offering expenses payable by us, our pro forma net
tangible book value deficiency as of December 31, 2016
would have been approximately $10.6 million, or
$(0.53) per share of common stock. This represents an
immediate increase in net tangible book value of
$15.79 per share to our existing stockholders and an
immediate dilution in net tangible book value of $0.83
per share to investors participating in this offering. The
following table illustrates this dilution per share to investors
participating in this offering:
|
Assumed Series D Conversion
Price
|
|
$
|
0.30
|
|
|
Net tangible book value per share as of
December 31, 2016
|
|
$
|
(15.79
|
)
|
|
Increase in net tangible book value per share
attributable to this offering
|
|
$
|
15.26
|
|
|
Pro forma net tangible book value per share
after giving effect to the offering
|
|
$
|
(0.53
|
)
|
|
Dilution per share to new investors in this
offering
|
|
$
|
0.83
|
|
The above
discussion and table are based on 668,651 shares of
common stock outstanding as of December 31, 2016,
which does not include the following, all as of March
27, 2017:
|
|
●
|
125 shares of common stock reserved
for future issuance under our 1995 Stock Plan;
|
|
|
●
|
16,864,630 shares of common stock
issued or reserved for issuance upon conversion of, or issuance as
dividends on, our Series C and Series C1 convertible preferred
stock;
|
|
|
●
|
7,743,293 shares issued or reserved
for issuance upon conversion of our outstanding convertible
debt;
|
|
|
●
|
5,738,112 shares of common stock
issuable upon the exercise of outstanding warrants or warrants to
be exchanged for outstanding warrants;and
|
|
|
●
|
up to 16,670,000 shares of common
stock issuable upon exercise of warrants sold as part of this
offering.
|
A $0.01 increase
(decrease) in the assumed Series D preferred stock conversion price
of $0.30 per share would not change the pro forma as
adjusted net tangible book value deficiency resulting from the
offering ($10.6 million) or our as adjusted net
tangible book value deficiency per share, but would increase
(decrease) the dilution per share to new investors by approximately
$0.10, assuming the assumed number of shares of Series
D preferred stock sold remains the same. A 10% increase (decrease)
in the number of shares sold would not change the pro forma as
adjusted net tangible book value deficiency resulting from the
offering ($10.6 million) or our as adjusted net
tangible book value deficiency per share, but would increase
(decrease) the dilution per share to new investors by approximately
$0.06, assuming the assumed number of shares of Series
D preferred stock sold remains the same.
Certain of our
outstanding indebtedness and our outstanding warrants are subject
to customary “downside price protection” provisions in
the event we sell any common stock at a price lower than the
then-conversion price of these securities. The Series D preferred
stock offered pursuant to this prospectus will also be subject to
similar adjustment. These provisions are further described under
the captions “Description of Securities” and
“Description of Securities We Are
Offering.”
CAPITALIZATION
The following table
shows our cash and cash equivalents and our capitalization as of
December 31, 2016:
|
|
●
|
as adjusted to give effect to applicable
transactions occurring after December 31, 2016
described in “Summary—Recent Developments”; and
as further adjusted to give effect to this offering and certain
other transactions that are expected to occur as a result of this
offering..
|
You should read
this table together with the information under the heading
“Management’s Discussion and Analysis of Financial
Condition and Results of Operations” and our audited annual
consolidated financial statements and the related notes and other
financial information incorporated by reference into this
prospectus from our annual report on Form 10-K for the fiscal year
ended December 31, 2016.
|
|
|
|
|
|
|
|
Cash and cash
equivalents
|
$
14
|
3,834
|
|
|
|
|
|
Short-term
debt
|
1,205
|
1,097
|
|
Short-term
convertible notes
|
468
|
201
|
|
Convertible notes
payable
|
2,361
|
2,361
|
|
Outstanding
warrants, at fair value
|
1,420
|
114
|
|
|
|
|
|
Total
liabilities
|
5,454
|
3,774
|
|
|
|
|
|
Series C preferred
stock
|
601
|
-
|
|
Series C1 preferred
stock
|
701
|
701
|
|
Series D preferred
stock
|
-
|
4,806
|
|
Common
stock
|
742
|
743
|
|
Additional paid-in
capital
|
116,380
|
117,836
|
|
Treasury stock, at
cost
|
(132
)
|
(132
)
|
|
Accumulated
deficit
|
(127,558
)
|
(127,638
)
|
|
|
|
|
|
Total
stockholders’ surplus (deficit)
|
(9,266
)
|
(3,685
)
|
|
|
|
|
|
Total
capitalization
|
$
(14,720
)
|
(7,459
)
|
___________________________________
|
|
*
|
Assumes sale of all of the securities offered,
except shares of common stock that could be issued upon conversion
of the Series D preferred stock or upon exercise of the warrants
sold as part of this offering. Reflects receipt of the net cash
proceeds from this offering prior to the application thereof,
except for the repayment in full of our secured promissory note.
Also reflects the following, each of which is contingent upon
consummation of the offering, as further described under
“Summary—Recent Developments”:
|
|
|
●
|
the extinguishment of
indebtedness represented by the May 2016 short-term cash advances
as a result of the participation by Aquarius Opportunity Fund in
this offering at least to the extent of the principal and interest
on the cash advances;
|
|
|
●
|
the exchange of our
senior secured convertible note and the remaining portion of our
secured promissory note for a new convertible note;
|
|
|
●
|
the exchange, on a
dollar-for-dollar basis, of all outstanding shares Series C
preferred stock for shares of Series D preferred stock and
warrants; and
|
|
|
●
|
the exchange of certain
warrants, exercisable for 1,814,598 shares of common stock, for new
warrants, exercisable for 3,608,835 shares of common
stock.
|
PLAN
OF DISTRIBUTION
We are
offering up to 5,000 shares of our Series D convertible preferred
stock, together with warrants to purchase an aggregate of up
to 16,670,000 shares of common stock (and the shares
issuable from time to time upon conversion of the Series D
preferred stock and the exercise of the warrants), at a purchase
price of $1,000 per share of Series D preferred stock and warrant,
pursuant to this prospectus. The shares of Series D preferred stock
and warrants are immediately separable and will be separately
issued. There is no minimum offering amount required as a condition
to closing and we may sell significantly fewer securities in the
offering. We expect that the offering will be
open for no more than four weeks, unless the offering is
fully subscribed sooner or we decide to terminate the
offering sooner.
In
determining the offering price (
i.e.
, the conversion price and exercise
price) of the securities offered hereby, we will consider a
number of factors including, but not limited to, the current market
price of our common stock, trading prices of our common stock over
time, the illiquidity and volatility of our common stock, our
current financial condition and the prospects for our future cash
flows and earnings, and market and economic conditions at the time
of the offering. Once the offering price is determined, the
offering price for the securities will remain fixed for the
duration of the offering.
Moody
Capital Solutions. Inc. has agreed to act as our exclusive
placement agent in connection with this offering, subject to the
terms and conditions of a placement agent agreement
to be entered into with Moody. Moody is not purchasing
or selling any securities offered by this prospectus, nor is it
required to arrange the purchase or sale of any specific number or
dollar amount of securities, but has agreed to use its best efforts
to arrange for the sale of all of the shares of Series D preferred
stock and warrants offered hereby. Therefore, we will enter into
purchase agreements directly with investors in connection with this
offering. Moody may retain other brokers or dealers to act as
sub-agents or selected-dealers on its behalf in connection with the
offering. Offers will only be made to, and subscriptions accepted
from, institutional investors within the meaning of state
securities laws of the state in which such investor is
domiciled.
All cash funds
received in payment for securities sold in this offering will be
required to be submitted by subscribers to a non-interest bearing
escrow account, and will be held by the escrow agent for such
account until we and the placement agent notify the escrow agent
that the offering has closed. The closing will occur, as to all
subscriptions duly received and accepted by us, in one closing; we
do not intend to hold multiple closings in the offering. In the
event we do not accept the subscriptions and do not close the
offering, escrowed funds will be promptly returned to subscribers
without interest or offset.
We have agreed to
pay Moody a placement agent’s fee of 10% of the aggregate
cash purchase price of the securities sold in this offering. We
will also reimburse Moody up to $50,000 for its reasonable
out-of-pocket expenses, including, without limitation, fees and
expenses of its counsel.
The following table
shows the per share and total placement agent’s fees that we
will pay to Moody in connection with the sale of the securities
offered pursuant to this prospectus, assuming the purchase of all
of the shares of Series D preferred stock and accompanying warrants
offered hereby.
|
Per share placement agent’s
fees
|
$
100
|
|
Maximum offering total placement agent’s
fees
|
$
500,000
|
Because there is no
minimum amount required as a condition to the closing in this
offering, the actual total offering commissions, if any, are not
presently determinable and may be substantially less than the
maximum amount set forth above.
In addition, we
have granted a right of first refusal to the placement agent
pursuant to which it has the right to act as the lead manager or
underwriter or agent, as applicable, if we or our subsidiary raise
capital through a public or private offering of equity or debt
securities at any time prior to eighteen months from the date of
closing of this offering.
Our obligations to
issue and sell securities to the purchasers is subject to the
conditions set forth in the securities purchase agreement, which
may be waived by us at our discretion. A purchaser’s
obligation to purchase securities is subject to the conditions set
forth in the securities purchase agreement as well, which may also
be waived by the purchaser in its discretion.
We estimate the
total offering expenses in this offering that will be payable by
us, excluding the placement agent’s fees, will be
approximately $340,000, which includes legal, accounting and
printing costs, various other fees and reimbursement of the
placement agent’s expenses.
We have also agreed
to indemnify the placement agent for certain liabilities under the
Securities Act.
The foregoing does
not purport to be a complete statement of the terms and conditions
of the placement agent agreement or the securities purchase
agreement. A copy of the placement agent agreement and the form of
securities purchase agreement with investors are included as
exhibits to the registration statement of which this prospectus
forms a part.
Moody is an
underwriter within the meaning of Section 2(a)(11) of the
Securities Act, and any commissions received by it and any profit
realized on the resale of the securities sold by it while acting as
principal might be deemed to be underwriting discounts or
commissions under the Securities Act. As an underwriter, Moody is
required to comply with the Securities Act and the Securities
Exchange Act of 1934, including Rule 10b-5 and Regulation M under
the Exchange Act. These rules and regulations may limit the timing
of purchases and sales of our securities by Moody acting as
principal. Under these rules and regulations, Moody:
|
|
●
|
may not engage in
any stabilization activity in connection with our securities;
and
|
|
|
●
|
may not bid for or
purchase any of our securities or attempt to induce any person to
purchase any of our securities, other than as permitted under the
Exchange Act, until it has completed its participation in the
distribution.
|
Pursuant to the
purchase agreement to be entered into in connection with this
offering, we will be prohibited from issuing, entering into any
agreement to issue or announcing the issuance or proposed issuance
of any shares of common stock or common stock equivalents (subject
to certain exceptions) for a period of 90 days from the closing of
this offering.
DESCRIPTION
OF SECURITIES WE ARE OFFERING
We are offering up
to 5,000 shares of our Series D convertible preferred stock,
together with warrants to purchase an aggregate of up to
16,670,000 shares of common stock (and the shares issuable
from time to time upon conversion of the Series D preferred stock
and the exercise of the warrants), at a purchase price of $1,000
per share of Series D preferred stock and warrant, pursuant to this
prospectus. The shares of Series D preferred stock and warrants are
immediately separable and will be separately issued.
This prospectus
also covers the shares of common stock issuable upon conversion of
the Series D preferred stock and upon exercise of the warrants. The
material terms and provisions of our common stock, which underlies
the Series D preferred stock and the warrants, and each other class
of securities which qualifies or limits the foregoing, are
described under the caption “Description of
Securities.”
Series D Preferred Stock
The material terms
and provisions of the Series D preferred stock being offered
pursuant to this prospectus are described below. The following
description is qualified in its entirety by reference to the form
of certificate of designation filed as an exhibit to the
registration statement of which this prospectus is a part.
Prospective investors should carefully review the terms and
provisions of the form of the certificate of designation for a
complete description of the terms and conditions of the Series D
preferred stock.
Our Board has
designated 5,000 shares of preferred stock as Series D Convertible
Preferred Stock, par value $0.001 per share. There are no shares of
Series D preferred stock outstanding.
Voting
Rights.
Except as required by law, holders of the
Series D preferred stock will not have rights to vote on any
matters, questions or proceedings, including the election of
directors. However, as long as any shares of Series D preferred
stock are outstanding, we may not, without the affirmative vote of
the holders of a majority of the then-outstanding shares of Series
D preferred stock, (1) alter or change adversely the powers,
preferences or special rights given to the Series D preferred stock
or (2) alter or amend the certificate of designation governing
the Series D preferred stock.
Liquidation
Preference.
The Series D preferred stock ranks, with
respect to rights upon liquidation, winding-up or dissolution, (1)
senior to common stock, (2) senior to any series of preferred stock
ranked junior to the Series D preferred stock, (3) junior to our
Series C preferred stock, and (4) junior to all of our existing and
future indebtedness.
Redemption.
We have the right to
redeem the Series D preferred stock for a cash payment equal to
120% of the stated value of the Series D preferred stock, plus all
accrued but unpaid dividends and any liquidated damages or other
amounts due thereon. At any time after the second anniversary of
the consummation of this offering, the holders of two-thirds of the
then-outstanding shares of Series D preferred stock can, upon
notice to us, force us to redeem the Series D preferred stock, for
a cash payment equal to 100% of the stated value of the Series D
preferred stock, plus all accrued but unpaid dividends and any
liquidated damages or other amounts due thereon. Holders of Series
D preferred stock will receive 20 trading days prior notice of any
redemption and will have the ability to convert the Series D
preferred stock into common stock during this notice
period.
Conversion;
Beneficial Ownership Limitation.
Subject to
certain ownership limitations as described below, the Series D
preferred stock is convertible at any time at the option of the
holder into shares of our common stock at a conversion ratio
determined by dividing the sum of the $1,000 stated value of the
Series D preferred stock plus all accrued but unpaid dividends and
any liquidated damages or other amounts due thereon, by an
initial conversion price estimated at this time to be
$0.30 per share of common stock. The
actual conversion price has not been determined, but is expected to
be a discount to the then-current market price of our common
stock. The conversion price is subject to adjustment in the
case of stock splits, stock dividends, combinations of shares and
similar recapitalization transactions. In addition, until our
common stock is listed on a national securities exchange, if we
sell or grant any right to purchase or sell any common stock or
common stock equivalents entitling any person to acquire shares of
common stock (subject to exceptions for certain exempt issuances,
including issuances pursuant to equity compensation plans, certain
issuances to consultants, issuances in connection with exercise or
exchange of common stock equivalents already outstanding and
issuances pursuant to acquisitions or strategic transactions or
arrangements) at an effective price per share that is lower than
the then-conversion price of the Series D preferred stock, then the
conversion price will be further reduced to equal such lower price
per share.
The holders of
two-thirds of the then-outstanding shares of Series D preferred
stock can, upon notice to us, force all holders to convert their
shares of Series D preferred stock pursuant to the terms of
conversion described above. In addition, if certain conditions
relating to our stock price and trading volume are met, we can
similarly force all holders to convert.
Subject to limited
exceptions, a holder of shares of Series D preferred stock will not
have the right to, and cannot be forced to, convert any portion of
its Series D preferred stock if the holder, together with its
affiliates, would beneficially own in excess of 4.99% of the number
of shares of our common stock outstanding immediately after giving
effect to its conversion; provided, however, that upon 61
days’ prior notice to us, the holder may increase such
percentage to 9.99%. We have the right to force the conversion of
the Series D preferred stock if our common stock meets certain
minimum price and trading volume thresholds.
Dividends.
From the original
issue date until the second anniversary thereafter, the holders of
Series D preferred stock will be entitled to receive quarterly
cumulative dividends at a rate per share (as a percentage of stated
value per share) of __% per year, payable quarterly in cash on each
March 31, June 30, September 30, and December 31 during such
period. In addition, the Series D preferred stock is entitled to
receive dividends (on an “as converted to common stock”
basis) upon and in the same form as dividends actually paid on
shares of our common stock. We are currently not permitted to pay
cash dividends under the terms of the Delaware General Corporation
Law and we will so advise the holders of Series D preferred stock
at the closing of this offering. If we do not have funds legally
available to pay cash dividends, such dividends accrete to and
effectively increase the outstanding stated value of the Series D
preferred stock. There are no sinking fund provisions applicable to
the Series D preferred stock.
Liquidation.
Upon our
liquidation, dissolution or winding up after payment or provision
for payment of our debts and other liabilities, but before any
distribution or payment is made to the holders of any junior
securities, the holders of Series D preferred stock will be
entitled to be paid out of our assets available for distribution to
our stockholders in an amount equal to $1,000 per share, plus any
accrued and unpaid dividends thereon and any other fees or
liquidated damages then due and owing thereon, after which any of
our remaining assets will be distributed among the holders of
junior classes of our capital stock in accordance with our
certificate of incorporation.
Warrants
The material terms
and provisions of the warrants being offered pursuant to this
prospectus are described below. The following description is
qualified in its entirety by reference to the form of warrant filed
as an exhibit to the registration statement of which this
prospectus is a part. Prospective investors should carefully review
the terms and provisions of the form of the warrant for a complete
description of the terms and conditions of the
warrants.
Underlying
Shares, Exercise Price, and
Duration.
Each warrant will be
immediately exercisable for up to 3,334 shares of our common
stock (based on warrant coverage of 100% of the shares issued
upon conversion of a share of Series D preferred stock), at
a exercise price estimated at this time to be $0.36
per share of common stock. The actual exercise price has not
been determined, but is expected to be 120% of the conversion price
of the Series D preferred stock. The warrants will be
exercisable immediately on the issuance date and will
expire five years following the issuance date. The warrants
will be issued separately from the Series D preferred stock
offered, and may be transferred separately immediately thereafter.
No warrants exercisable for a fractional amount of shares of common
stock will be issued. If an investor would otherwise be entitled to
receive a fractional warrant, the number of shares issuable upon
exercise of the warrant will be rounded up to the nearest whole
share. The warrant holders must pay the exercise price
in cash upon exercise of the warrants. After the close of business
on the expiration date, unexercised warrants will become
void.
Anti-Dilution
Protection.
The exercise price of the warrants is
subject to appropriate adjustment in the event of stock dividends,
stock splits, stock combinations, reclassifications,
reorganizations or similar events affecting our common stock, and
also upon any distributions of assets, including cash, stock or
other property to our stockholders.
Fundamental
Transactions.
In the event of any fundamental
transaction, as described in the warrants and generally including
any merger with another entity, the sale, transfer or other
disposition of all or substantially all of our assets to another
entity, or the acquisition by a person of more than 50% of our
common stock, then, subject to the agreement of the counterparty to
the fundamental transaction, the holders of the warrants will
thereafter have the right to receive upon exercise of the warrants
such shares of stock, securities or assets as would have been
issuable or payable with respect to or in exchange for a number of
shares of our common stock equal to the number of shares of our
common stock issuable upon exercise of the warrants immediately
prior to the fundamental transaction, had the fundamental
transaction not taken place, and appropriate provision will be made
so that the provisions of the warrants (including, for example,
provisions relating to the adjustment of the exercise price) will
thereafter be applicable, as nearly equivalent as may be
practicable in relation to any share of stock, securities or assets
deliverable upon the exercise of the warrants after the fundamental
transaction.
Transferabilit
y.
The warrants
may be transferred at the option of the holder upon surrender of
the warrants with the appropriate instruments of
transfer.
Right as a
Stockholder.
Except by virtue of a holder’s
ownership of shares of our common stock, the holders of the
warrants do not have the rights or privileges of holders of our
common stock, including any voting rights, until they exercise
their warrants.
Exercisability.
The warrants
will be exercisable, at the option of each holder, in whole or in
part, by delivering to us a duly executed exercise notice
accompanied by payment in full for the number of shares of our
common stock purchased upon such exercise. Subject to limited
exceptions, a holder of will not have the right to exercise any
portion of its warrant if the holder, together with its affiliates,
would beneficially own in excess of 4.99% of the number of shares
of our common stock outstanding immediately after giving effect to
its exercise; provided, however, that upon 61 days’ prior
notice to us, the holder may increase such percentage to
9.99%.
Waivers and
Amendments.
Subject to certain exceptions, any term
of the warrants may be amended or waived with our written consent
and the written consent of each holder of the then-outstanding
warrants.
DESCRIPTION
OF SECURITIES
We are
authorized to issue 1,005,000,000 shares of stock, in two classes:
1,000,000,000 shares of common stock, par value $.001 per share,
and 5,000,000 shares of preferred stock. As of March
27, 2017, there were 1,494,741 shares of common
stock outstanding, which were held of record by 210
stockholders, and 5,763.5 shares of preferred stock
outstanding, consisting of 1,422 shares of Series C
preferred stock, which were held of record by 1
stockholder, and 4312.5 shares of Series C1 preferred stock,
which were held of record by 6
stockholders.
Common Stock
The holders of
common stock are entitled to one vote for each share held of record
on all matters submitted to a vote of stockholders. Subject to
preferences that may be applicable to any outstanding preferred
stock, holders of common stock are entitled to receive ratably such
dividends as may be declared by the board out of funds legally
available therefor and in liquidation proceedings. Holders of
common stock have no preemptive or subscription rights and there
are no redemption rights with respect to such shares.
Preferred Stock
Our board is
authorized, without further stockholder action, to issue preferred
stock in one or more series and to fix the voting rights,
liquidation preferences, dividend rights, repurchase rights,
conversion rights, redemption rights and terms, including sinking
fund provisions, and certain other rights and preferences, of the
preferred stock. Our board designated 525,000 shares of preferred
stock as redeemable convertible preferred stock, none of which
remain outstanding; 3,000 shares of preferred stock as Series B
Convertible Preferred Stock, none of which remain outstanding;
9,000 shares of preferred stock as Series C Convertible Preferred
Stock, of which 1,422 were outstanding as of
March 27, 2017; 20,250 shares of preferred stock as
Series C1 Convertible Preferred Stock, of which 4,312.50 were
outstanding as of March 27, 2017; and 5,000 shares of
preferred stock as Series D Convertible Preferred Stock, none of
which were outstanding as of March 27, 2017. The
material terms and provisions of our Series D preferred stock are
described under the caption “Description of Securities We Are
Offering.”
Although there is
no current intention to do so, our board may, without stockholder
approval, issue additional shares of preferred stock with voting
and conversion rights that could adversely affect the voting power
or dividend rights of the holders of common stock and may have the
effect of delaying, deferring or preventing a change in
control.
Series C
Convertible Preferred Stock
Dividends
. From the original
issue date until 42 months thereafter, the holders of Series C
preferred stock are entitled to receive quarterly cumulative
dividends at a rate per share (as a percentage of stated value per
share) of 12% per year per share payable quarterly on each January
1, April 1, July 1 and October 1 during such period (beginning
October 1, 2015) in shares of our common stock (subject to certain
conditions) or, at our option, in cash. In addition, upon the
conversion of the Series C preferred stock (other than a forced
conversion, described below) during the such period, we must pay
the converting holder a “make-whole payment” in cash
or, at our option (subject to certain conditions), shares of our
common stock with respect to the converted shares of Series C
preferred stock in an amount equal to $420 per $1,000 of stated
value, less the amount of any dividends already paid on such shares
of Series C preferred stock. To the extent we choose to pay any
dividends or make-whole payments in shares of our common stock,
such shares will be valued at 80% of then-current market price
(calculated as the average daily volume weighted average price of
our common stock for the five consecutive trading days prior to
payment). After the dividend payment period, holders of Series C
preferred stock are only entitled to receive dividends on an
as-if-converted basis) with holder of our common stock (other than
dividends paid in additional shares of common stock).
Voting
. Except as otherwise
proved by law or in the Series C designations, the Series C
preferred stock has no voting rights. We may not, without the
consent of the holders of a majority of the shares of Series C
preferred stock then outstanding, alter or change adversely the
powers, preferences or rights given to the Series C preferred stock
or alter or amend the Series C designations, create any class of
stock with a liquidation preference equal or senior to the Series C
preferred stock, amend our charter in any manner that adversely
affects any rights of the holders of Series C preferred stock,
increase the number of authorized shares of Series C preferred
stock, or enter into any agreement with respect to any of the
foregoing.
Liquidation
. In the event of our
liquidation, dissolution or winding-up, whether voluntary or
involuntary, the holders of our Series C preferred stock will be
entitled to receive out of our assets an amount equal to the stated
value of their shares, plus any other fees, liquidated damages or
dividends then due and owing on their shares, before any
distribution or payment to the holders of any junior
securities.
Conversion
. The Series C
preferred stock is convertible at any time, at the option of the
holder. In addition, if the market price of our common stock for
each trading day for 20 of any 30 consecutive trading-day period
exceeds 250% of the highest conversion price in effect during such
period, we may force each holder to convert all or part of such
holder’s shares into shares of common stock.
Each share of
Series C preferred stock is convertible into the number of shares
of common stock equal to the quotient obtained by dividing (1) the
sum of the stated value plus all accrued but unpaid dividends on
such share, by (2) the per share conversion price. The initial per
share conversion price was $76.00, but has been reset pursuant to
the Series C designations to $0.4016 as of March
27, 2017. The conversion price will automatically adjust
downward to 80% of the then-current market price of our common
stock 15 trading days after any subsequent reverse stock split of
the common stock, and 5 trading days after any conversions of our
outstanding convertible debt. The conversion price is subject to
further adjustment under certain circumstances to protect the
holders of Series C preferred stock from dilution relative to
certain issuances of common stock, or securities convertible into
or exercisable for shares of common stock. Subject to certain
exceptions, if we issue shares of common stock, or such other
securities, at a price per share less than the then-effective
conversion price, the conversion price will be adjusted to equal
such lower per share consideration.
We expect to
exchange all outstanding shares of Series C preferred stock for
shares of Series D preferred stock and warrants as soon as
practicable following the consummation of this
offering.
Series C1
Convertible Preferred Stock
The Series C1
preferred stock has terms that are substantially the same as the
Series C preferred stock, except that the Series C1 preferred stock
does not pay dividends (unless and to the extent declared on the
common stock) or at-the-market “make-whole payments and,
while it has the same anti-dilution protections afforded the Series
C preferred stock, it does not automatically reset in connection
with a reverse stock split or conversion of our outstanding
convertible debt.
Secured Promissory Note
As of March
27, 2017, we have issued and outstanding
$515,077 in aggregate principal and interest amount of
secured promissory notes that, as amended, provide the holders the
right to convert the outstanding balance into shares of our common
stock at a conversion price per share equal to the lower of (1)
$20,000 and (2) 75% of the lowest daily volume weighted
average price per share of our common stock during the five
business days prior to conversion. If the conversion price at the
time of any conversion request would be lower than $12,000 per
share, we have the option of delivering the conversion amount in
cash in lieu of shares of our common stock. The note is secured by
a lien on our inventory and accounts receivables. We expect to
repay this the GHS portion of this note in full using proceeds from
this offering (see “Summary—Recent
Developments—Secured Promissory Note”), and GPB will
exchange its portion for a new convertible note (see
“Summary—Recent Developments—Debt
Restructuring”).
Senior Secured Convertible
Notes
As of March
27, 2017, we have issued and outstanding $1,525,000 million
in aggregate principal amount of a senior secured convertible notes
that are convertible at any time, in whole or in part, at the
holder’s option, into shares of our common stock, at a
conversion price equal to $0.2639 per share, subject
to certain customary adjustments and anti-dilution provisions. The
notes are secured by a lien on all of our assets. Upon consummation
of this offering, we expect that the principal amount will be
increased to $1,831,863 in connection with the
exchange of the original note and the holder’s portion of the
secured promissory note (see “Summary—Recent
Developments—Debt Restructuring”).
Convertible Note
As of March 24,
2017, we have issued and outstanding
$300,000 in aggregate principal amount of a
convertible note convertible into shares of our common stock at a
conversion price per share of $13.92, subject to customary
anti-dilution adjustment (see “Summary—Recent
Developments—Shenghuo License
Agreement”.
OID
Note
As of March 27,
2017, we have issued and outstanding $220,000 in aggregate
principal amount of 10% original issuance discount convertible
promissory note. The note matures six months from its date of
issuance and, in addition to the 10% original issue discount,
accrues interest at a rate of 10% per year. We may prepay the note,
in whole or in part, for 115% of outstanding principal and interest
until 30 days from issuance, for 125% of outstanding principal and
interest at any time from 31 to 60 days from issuance, and for 130%
of outstanding principal and interest at any time from 61 days from
issuance until immediately prior to the maturity date. After six
months from the date of issuance (i.e., if we fail torepay all
principal and interest due under the notes at the maturity date),
the holder may convert the note, at any time, in whole or in part,
into shares of our common stock, at a conversion price equal to 60%
of the lowest volume weighted average price of our common stock
during the 20 trading days prior to conversion, subject to certain
customary adjustments and anti-dilution provisions contained in the
note.
Convertible
Promissory Note
As of March 27,
2017, we have issued and outstanding $170,000 in aggregate
principal amount of a 12% convertible promissory note. The note
matures nine months from the date of issuance and, in addition to
the original issue discount, accrues interest at a rate of 12% per
year. We may prepay the note, in whole or in part, for 115% of
outstanding principal and interest until 30 days from issuance, for
125% of outstanding principal and interest at any time from 31 to
60 days from issuance, and for 130% of outstanding principal and
interest at any time from 61 days from issuance to 180 days from
issuance. After six months from the date of issuance, the holder
may convert the note, at any time, in whole or in part, into shares
of our common stock, at a conversion price equal to the lower of
the price offered in our next public offering or a 40% discount to
the average of the two lowest trading prices of the common stock
during the 20 trading days prior to the conversion, subject to
certain customary adjustments and price-protection provisions
contained in the note. The note includes customary events of
default provisions and a default interest rate of 24% per year.
Upon the occurrence of an event of default, the holder may require
us to redeem the note (or convert it into shares of common stock)
at 150% of the outstanding principal balance plus accrued and
unpaid interest.
Warrants and Options
We have
issued warrants to purchase our common stock from time to time in
connection with certain financing arrangements. As of March
27, 2017, there are warrants exercisable for an aggregate of
5,735,980 shares of common stock outstanding, as
follows:
|
Warrants
(Underlying
Shares)
|
Exercise Price Per Share
|
Expiration Date
|
|
24
|
(1)
|
$8,368.00
|
May 23, 2018
|
|
7,542
|
(2)
|
$75.00
|
June 14,
2021
|
|
3
|
(3)
|
$40,000.00
|
April 23, 2019
|
|
8
|
(4)
|
$36,000.00
|
May 22, 2019
|
|
3
|
(5)
|
$30,400.00
|
September 10, 2019
|
|
5
|
(6)
|
$36,864.80
|
September 27, 2019
|
|
10
|
(7)
|
$22,504.00
|
December 2, 2019
|
|
105
|
(8)
|
$7,200.00
|
December 2, 2020
|
|
105
|
(9)
|
$8,800.00
|
December 2, 2020
|
|
25
|
(11)
|
$20,400.00
|
March 30, 2018
|
|
22
|
(12)
|
$9,504.00
|
June 29, 2020
|
|
659
|
(10)
|
$640.00
|
June 29, 2020
|
|
343
|
(11)
|
$640.00
|
September 4, 2020
|
|
363
|
(12)
|
$640.00
|
September 21, 2020
|
|
7
|
(13)
|
$9,504.00
|
September 4, 2020
|
|
198
|
(14)
|
$640.00
|
October 23, 2020
|
|
7
|
(15)
|
$9,504.00
|
October 23, 2020
|
|
5,447,817
|
(16)
|
$0.3488
|
June 14,
2021
|
|
261,495
|
(17)
|
$0.3488
|
February 21, 2021
|
|
17,239
|
(18)
|
$13.92
|
June 6, 2021
|
|
5,735,980
|
|
|
|
|
(1)
|
Issued in June 2015 in
exchange for warrants originally issued as part of a
May 2013 private placement.
|
|
(2)
|
Issued in June 2015 in
exchange for warrants originally issued as part of a
May 2013 private placement.
|
|
(3)
|
Issued to a placement
agent in conjunction with an April 2014 private
placement.
|
|
(4)
|
Issued to a placement
agent in conjunction with a September 2014 private
placement.
|
|
(5)
|
Issued as part of a
September 2014 Regulation S offering.
|
|
(6)
|
Issued to a placement
agent in conjunction with a 2014 public offering.
|
|
(7)
|
Issued in June 2015 in
exchange for warrants originally issued as part of a 2014 public
offering.
|
|
(8)
|
Issued as part of a
March 2015 private placement.
|
|
(9)
|
Issued to a placement
agent in conjunction with a June 2015 private
placement.
|
|
(10)
|
Issued as part of a June
2015 private placement.
|
|
(11)
|
Issued as part of a June
2015 private placement.
|
|
(12)
|
Issued as part of a June
2015 private placement.
|
|
(13)
|
Issued to a placement
agent in conjunction with a June 2015 private
placement.
|
|
(14)
|
Issued as part of a June
2015 private placement.
|
|
(15)
|
Issued to a placement
agent in conjunction with a June 2015 private
placement.
|
|
(16)
|
Issued as part of a
February 2016 private placement.
|
|
(17)
|
Issued to a placement
agent in conjunction with a February 2016 private
placement.
|
|
(18)
|
Issued
pursuant to a strategic license agreement.
|
All outstanding
warrant agreements provide for anti-dilution adjustments in the
event of certain mergers, consolidations, reorganizations,
recapitalizations, stock dividends, stock splits or other changes
in our corporate structure; except for (9). In
addition, warrants subject to footnotes (2) and
(10)-(12), (14), and
(16)-(18) in the table above are subject to “lower
price issuance” anti-dilution provisions that automatically
reduce the exercise price of the warrants (and, in the cases of
warrants subject to footnote (2), (16) and
(17) in the table above, increase the number of shares of
common stock issuable upon exercise), to the offering price in a
subsequent issuance of our common stock, unless such subsequent
issuance is exempt under the terms of the warrants.
The warrants
subject to footnote (2) are subject to a mandatory
exercise provision. This provision permits us, subject to certain
limitations, to require exercise of such warrants at any time
following (a) the date that is the 30th day after the later of our
receipt of an approvable letter from the FDA for LuViva and the
date on which the common stock achieves an average market price for
20 consecutive trading days of at least $1,040.00 with
an average daily trading volume during such 20 consecutive trading
days of at least 250 shares, or (b) the
date on which the average market price of the common stock for 20
consecutive trading days immediately prior to the date we deliver a
notice demanding exercise is at least $129,600.00 and
the average daily trading volume of the common stock exceeds
250 shares for such 20 consecutive
trading days. If these warrants are not timely exercised upon
demand, they will expire. Upon the occurrence of certain events, we
may be required to repurchase these warrants, as well as the
warrants subject to footnote (2) in the table above.
The warrants
subject to footnote (5) in the table above are also
subject to a mandatory exercise provision. This provision permits
us, subject to certain limitations, to require the exercise of such
warrants should the average trading price of our common stock over
any 30 consecutive day trading period exceed
$92.16.
The warrants
subject to footnote (7) in the table
above are also subject to a mandatory exercise provision. This
provision permits us, subject to certain limitations, to require
exercise of 50% of the then-outstanding warrants if the trading
price of our common stock is at least two times the initial warrant
exercise price for any 20-day trading period. Further, in the event
that the trading price of our common stock is at least 2.5 times
the initial warrant exercise price for any 20-day trading period,
we will have the right to require the immediate exercise of 50% of
the then-outstanding warrants. Any warrants not exercised within
the prescribed time periods will be canceled to the extent of the
number of shares subject to mandatory exercise.
The holders of the
warrants subject to footnote (2) in the table above
have agreed to surrender the warrants upon consummation of this
offering for new warrants exercisable for 200% of the number of
shares underlying the surrendered warrants, but without certain
anti-dilution protections included with the surrendered
warrants.
As of March
27, 2017, we have outstanding options to purchase a total of
125 shares of our common stock pursuant to our equity
incentive plan, at a weighted average exercise price of
$37,920 per share. Recommendations for option grants
under our equity incentive plans are made by the compensation
committee of our board, subject to ratification by the full board.
The compensation committee may issue options with varying vesting
schedules, but all options granted pursuant to our equity incentive
plans must be exercised within ten years from the date of
grant.
OUR
BUSINESS
We are a medical
technology company focused on developing innovative medical devices
that have the potential to improve healthcare. Our primary focus is
the sales and marketing of our LuViva® Advanced Cervical Scan
non-invasive cervical cancer detection device. The underlying
technology of LuViva primarily relates to the use of biophotonics
for the non-invasive detection of cancers. LuViva is designed to
identify cervical cancers and precancers painlessly, non-invasively
and at the point of care by scanning the cervix with light, then
analyzing the reflected and fluorescent light.

LuViva provides a
less invasive and painless alternative to conventional tests for
cervical cancer screening and detection. Additionally, LuViva
improves patient well-being not only because it eliminates pain,
but also because it is convenient to use and provides rapid results
at the point of care. We focus on two primary applications for
LuViva: first, as a cancer screening tool in the developing world,
where infrastructure to support traditional cancer-screening
methods is limited or non-existent, and second, as a triage
following traditional screening in the developed world, where a
high number of false positive results cause a high rate of
unnecessary and ultimately costly follow-up tests.
Screening for
cervical cancer represents one of the most significant demands on
the practice of diagnostic medicine. As cervical cancer is linked
to a sexually transmitted disease—the human
papillomavirus (HPV)—every woman essentially
becomes “at risk” for cervical cancer simply after
becoming sexually active. In the developing world, there are
approximately 2.0 billion women aged 15 and older who are
potentially eligible for screening with LuViva. Guidelines for
screening intervals vary across the world, but U.S. guidelines call
for screening every three years. Traditionally, the Pap smear
screening test, or Pap test, is the primary cervical cancer
screening methodology in the developed world. However, in
developing countries, cancer screening using Pap tests is expensive
and requires infrastructure and skill not currently existing, and
not likely to be developed in the near future, in these
countries.
The
Need for LuViva
We believe LuViva
is the answer to the developing world’s cervical cancer
screening needs. Screening for cervical cancer in the developing
world often requires working directly with foreign governments or
non-governmental agencies (NGOs). By partnering with governments or
NGOs, we are able to provide immediate access to cervical cancer
detection to large segments of a nation’s population as part
of national or regional governmental healthcare programs,
eliminating the need to develop expensive and resource-intensive
infrastructures.
In the developed
world, we believe LuViva offers a more accurate and ultimately
cost-effective triage medical device, to be used once a traditional
Pap test indicates the possibility of cervical cancer. Due to the
high number of false positive results from Pap tests, traditional
follow-on tests entail increased medical treatment costs. We
believe these costs can be minimized by utilizing LuViva as a
triage to determine whether follow-on tests are
warranted.
We believe our
non-invasive cervical cancer detection technology can be applied to
the early detection of other cancers as well. In 2013, we announced
a license agreement with Konica Minolta, Inc. allowing us to
manufacture and develop a non-invasive esophageal cancer detection
product from Konica Minolta based on our biophotonic technology
platform. Early market analyses of our biophotonic technology
indicated that skin cancer detection was also promising, but
currently we are focused primarily on the large-scale
commercialization of LuViva.
Cancer
Cancer is a group
of many related diseases. All forms of cancer involve the
out-of-control growth and spread of abnormal cells. Normal cells
grow, divide, and die in an orderly fashion. Cancer cells, however,
continue to grow and divide and can spread to other parts of the
body. In America, half of all men and one-third of all women will
develop some form of cancer during their lifetimes. According to
the American Cancer Society, the sooner a cancer is found and
treatment begins, the better a patient’s chances are of being
cured. We began investigating the applications of our biophotonic
technology to cancer detection before 1997, when we initiated a
preliminary market analysis. We concluded that our biophotonic
technology had applications for the detection of a variety of
cancers through the exposure of tissue to light. We selected
detection of cervical cancer and skin cancer from a list of the ten
most promising applications to pursue initially, and ultimately
focused primarily on our LuViva cervical cancer detection
device.
Cervical cancer is
a cancer that begins in the lining of the cervix (which is located
in the lower part of the uterus). Cervical cancer forms over time
and may spread to other parts of the body if left untreated. There
is generally a gradual change from a normal cervix to a cervix with
precancerous cells to cervical cancer. For some women, precancerous
changes may go away without any treatment. While the majority of
precancerous changes in the cervix do not advance to cancer, if
precancers are treated, the risk that they will become cancers can
be greatly reduced.
The Developing
World
According to the
most recent data published by the World Health Organization (WHO),
cervical cancer is the fourth most frequent cancer in women
worldwide, with an estimated 530,000 new cases in 2012. For women
living in less developed regions, however, cervical cancer is the
second most common cancer, with an estimated 445,000 new cases in
2012 (84% of the new cases worldwide). In 2012, approximately 270
000 women died from cervical cancer; more than 85% of these deaths
occurring in low- and middle-income countries.

As noted by the
WHO, in developed countries, programs are in places that enable
women to get screened, making most pre-cancerous lesions
identifiable at stages when they can easily be treated. Early
treatment prevents up to 80% of cervical cancers in these
countries. In developing countries, however, limited access to
effective screening means that the disease is often not identified
until it is further advanced and symptoms develop. In addition,
prospects for treatment of such late-stage disease may be poor,
resulting in a higher rate of death from cervical cancer in these
countries.
We believe that the
greatest need and market opportunity for LuViva lies in screening
for cervical cancer in developing countries where the
infrastructure for traditional screening may be limited or
non-existent.
We are actively
working with distributors in the following countries to implement
government-sponsored screening programs: Turkey, Bangladesh,
Indonesia, Kenya and Nigeria. The number of screening candidates in
those countries is approximately 246 million and represents 3 of
the 10 most populous countries in the world.
The Developed
World

The Pap test, which
involves a sample of cervical tissue being placed on a slide and
observed in a laboratory, is currently the most common form of
cervical cancer screening. Since the introduction of screening and
diagnostic methods, the number of cervical cancer deaths in the
developed world has declined dramatically, due mainly to the
increased use of the Pap test. However, the Pap test has a wide
variation in sensitivity, which is the ability to detect the
disease, and specificity, which is the ability to exclude false
positives. A study by Duke University for the U.S. Agency for
Health Care Policy and Research published in 1999 showed Pap test
performance ranging from a 22%-95% sensitivity and 78%-10%
specificity, although new technologies improving the sensitivity
and specificity of the Pap test have recently been introduced and
are finding acceptance in the marketplace. About 60 million Pap
tests are given annually in the United States, at an average price
of approximately $26 per test.
After a Pap test
returns a positive result for cervical cancer, accepted protocol
calls for a visual examination of the cervix using a colposcope,
usually followed by a biopsy, or tissue sampling, at one or more
locations on the cervix. This method looks for visual
changes attributable to cancer. There are about two million
colposcope examinations annually in the United States and Europe.
In 2003, the average cost of a stand-alone colposcope examination
in the United States was $185 and the average cost of a colposcopy
with biopsy was $277.
Given this
landscape, we believe that there is a material need and market
opportunity for LuViva as a triage device in the developed world
where LuViva represents a more cost-effective method of verifying a
positive Pap test than the alternatives.
The LuViva Advanced Cervical
Scan
LuViva is designed
to identify cervical cancers and precancers painlessly,
non-invasively and at the point of care by scanning the cervix with
light, then analyzing the light reflected from the cervix. The
information presented by the light would be used to indicate the
likelihood of cervical cancer or precancers. Our product, in
addition to detecting the structural changes attributed to cervical
cancer, is also designed to detect the biochemical changes that
precede the development of visual lesions. In this way, cervical
cancer may be detected earlier in its development, which should
increase the chances of effective treatment. In addition to the
device itself, operation of LuViva requires employment of our
single-use, disposable calibration and alignment cervical
guide.
To date, thousands
of women in multiple international clinical settings have been
tested with LuViva. As a result, more than 25 papers and
presentations have been published regarding LuViva in a clinical
setting, including at the International Federation of
Gynaecology and Obstetrics Congress in London in 2015
and at the Indonesian National Obstetrics and Gynecology
(POGI) Meeting in Solo in 2016.
Internationally, we
contract with country-specific or regional distributors. We believe
that the international market will be significantly larger than the
U.S. market due to the international demand for cervical cancer
screening. We have formal distribution agreements in place covering
54 countries and plan on adding additional countries in
2017.
We have
previously obtained regulatory approval to sell LuViva
in Europe under our Edition 3 CE Mark. Additionally, LuViva has
also obtained marketing approval from Health Canada,
COFEPRIS in Mexico, Ministry of Health in Kenya and the Singapore
Health Sciences Authority. We currently are seeking regulatory
approval to market LuViva in the United States, but have not yet
received approval from the U.S. FDA. As of March 27,
2017, we have sold 121 LuViva devices and
approximately 64,000 single-use-disposable cervical
guides to international distributors.
We believe our
non-invasive cervical cancer detection technology can be applied to
the early detection of other cancers as well. From 2008 to early
2013, we worked with Konica Minolta to explore the feasibility of
adapting our microporation and biophotonic cancer detection
technology to other areas of medicine and to determine potential
markets for these products in anticipation of a development
agreement. In February 2013, we replaced our existing agreements
with Konica Minolta with a new agreement, pursuant to which,
subject to the payment of a nominal license fee due upon FDA
approval, Konica Minolta has granted us a five-year, world-wide,
non-transferable and non-exclusive right and license to manufacture
and to develop a non-invasive esophageal cancer detection product
from Konica Minolta and based on our biophotonic technology
platform. The license permits us to use certain related
intellectual property of Konica Minolta. In return for the license,
we have agreed to pay Konica Minolta a royalty for each licensed
product we sell. We continue to seek new collaborative partners to
further develop our biophotonic technology.
Manufacturing, Sales Marketing and
Distribution
We manufacture
LuViva at our Norcross, Georgia facility. Most of the components of
LuViva are custom made for us by third-party manufacturers. We
adhere to ISO 13485:2003 quality standards in our manufacturing
processes. Our single-use cervical guides are manufactured by a
vendor that specializes in injection molding of plastic medical
products.
On January 22,
2017, we entered into a license agreement with SMI pursuant to
which we granted SMI an exclusive global license to manufacture the
LuViva device and related disposables (subject to a carve-out for
manufacture in Turkey).
We rely on
distributors to sell our products. Distributors can be country
exclusive or cover multiple countries in a region. We manage these
distributors, provide them marketing materials and train them to
demonstrate and operate LuViva. We seek distributors that have
experience in gynecology and in introducing new technology into
their assigned territories.
We have only
limited experience in the production planning, quality system
management, facility development, and production scaling that will
be needed to bring production to increased sustained commercial
levels. We will likely need to develop additional expertise in
order to successfully manufacture, market, and distribute any
future products.
Research, Development and
Engineering
We have been
engaged primarily in the research, development and testing of our
LuViva non-invasive cervical cancer detection product and our core
biophotonic technology. Since 2013, we have incurred about
$7.2 million in research and development expenses, net
of about $927,000 reimbursed through collaborative arrangements and
government grants. Research and development costs were
approximately $0.7 and $1.5 million in
2016 and 2015, respectively.
Since 2013, we have
focused our research and development and our engineering resources
almost exclusively on development of our biophotonic technology,
with only limited support of other programs funded through
government contracts or third party funding. Because our research
and clinical development programs for other cancers are at a very
early stage, substantial additional research and development and
clinical trials will be necessary before we can produce commercial
prototypes of other cancer detection products.
Several of the
components used in LuViva currently are available from
only one supplier, and substitutes for these components could not
be obtained easily or would require substantial modifications to
our products.
Patents
We have pursued a
course of developing and acquiring patents and patent rights and
licensing technology. Our success depends in large part on our
ability to establish and maintain the proprietary nature of our
technology through the patent process and to license from others
patents and patent applications necessary to develop our products.
As of December 31, 2016, we have 25
granted U.S. patents relating to our biophotonic cancer detection
technology and 5 pending U.S. patent applications. We
also have three granted patents that apply to our interstitial
fluid analysis system.
Competition
The medical device
industry in general and the markets for cervical cancer detection
in particular, are intensely competitive. If successful in our
product development, we will compete with other providers of
cervical cancer detection and prevention products.
Current cervical
cancer screening and diagnostic tests, primarily the Pap test, HPV
test, and colposcopy, are well established and pervasive.
Improvements and new technologies for cervical cancer detection and
prevention, such as Thin-Prep from Hologic and HPV testing from
Qiagen, have led to other new competitors. In addition, there are
other companies attempting to develop products using forms of
biophotonic technologies in cervical cancer detection, such as
Spectrascience, which has a very limited U.S. FDA
approval to market its device for detection of cervical cancers,
but has not yet entered the market. The approval limits use of the
Spectrascience device only after a colposcopy, as an adjunct. In
addition to the Spectrascience device, there are other technologies
that are seeking to enter the market as adjuncts to colposcopy,
including devices from Dysis and Zedco. While these technologies
are not direct competitors to LuViva, modifications to them or
other new technologies will require us to develop devices that are
more accurate, easier to use or less costly to administer so that
our products have a competitive advantage.
In April 2014, the
U.S.
FDA approved the use of the Roche cobas HPV test as a primary
screener for cervical cancer. Using a sample of cervical cells, the
cobas HPV test detects DNA from 14 high-risk HPV types. The test
specifically identifies HPV 16 and HPV 18, while concurrently
detecting 12 other types of high-risk HPVs. This could make
HPV testing a competitor to the Pap test. However, due to its lower
specificity, we believe that screening with HPV will increase the
number of false positive results if widely adopted.
In June 2006, the
U.S.
FDA approved the HPV vaccine Gardasil from drug maker Merck.
Gardasil is a prophylactic HPV vaccine, meaning that it is designed
to prevent the initial establishment of HPV infections. For maximum
efficacy, it is recommended that girls receive the vaccine prior to
becoming sexually active. Since Gardasil will not block infection
with all of the HPV types that can cause cervical cancer, the
vaccine should not be considered a substitute for routine Pap
tests. On October 16, 2009, GlaxoSmithKline PLC was granted
approval in the United States for a similar preventive HPV vaccine,
known as Cervarix. Due to the limited availability and lack of 100%
protection against all potentially cancer-causing strains of HPV,
we believe that the vaccines will have a limited impact on the
cervical cancer screening and diagnostic market for many
years.
Government Regulation
The medical devices
that we manufacture are subject to regulation by numerous
regulatory bodies, including the CFDA and the U.S. FDA
and comparable international regulatory agencies. These agencies
require manufacturers of medical devices to comply with applicable
laws and regulations governing the development, testing,
manufacturing, labeling, marketing and distribution of medical
devices. Devices are generally subject to varying levels of
regulatory control, the most comprehensive of which requires that a
clinical evaluation program be conducted before a device receives
approval for commercial distribution.
In the European
Union, medical devices are required to comply with the Medical
Devices Directive and obtain CE Mark certification in order to
market medical devices. The CE Mark certification, granted
following approval from an independent “Notified Body,”
is an international symbol of adherence to quality assurance
standards and compliance with applicable European Medical Devices
Directives. We have maintained ISO 13485:2003 certification since
2014 and until the end of 2016, which has allowed us to issue our
Edition 3 CE Mark and sell LuViva in the European Union and other
markets. In order to continue marketing in those territories, we
must undergo an inspection and re-file for ISO 13485:2003 and the
CE Mark in 2017.
China has a
regulatory regime similar to that of the European Union, but due to
interaction with the U.S. regulatory regime, the CFDA also shares
some similarities with its U.S. counterpart. Devices are classified
by the CFDA’s Center for Medical Device Evaluation (CMDE)
into three categories based on medical risk, with the level of
regulatory oversight determined by degree of risk and invasiveness.
CMDE’s device classifications and definitions are as
follows:
●
Class I device: The
safety and effectiveness of the device can be ensured through
routine administration.
●
Class II device:
Further control is required to ensure the safety and effectiveness
of the device.
●
Class III device:
The device is implanted into the human body; used for life support
or sustenance; or poses potential risk to the human body, and thus
must be strictly controlled in respect to safety and
effectiveness.
Based on the above
definitions and several discussions with regulatory consultants and
potential partners, we believe that LuViva is most likely to be
classified as a Class II device, however, this is not certain and
the CFDA may determine that LuViva requires a Class III
registration. Class III registrations are granted by the national
CFDA office while Class I and II registrations occur at the
provincial level. Typically, registration granted at the provincial
level allows a medical device to be marketed in all of
China’s provinces.
While Class I
devices usually do not require clinical trial data from Chinese
patients and Class III devices almost always do, Class II medical
devices sometimes do and sometimes do not require Chinese clinical
trials, and this determination may depend on the claim for the
device and quality of clinical trials conducted outside of China.
If clinical trials conducted in China are required, they usually
are less burdensome for Class II devices than Class III
devices.
CFDA labs also
conduct electrical, mechanical and electromagnetic emission safety
testing for medical devices similar to those required for the CE
Mark. As is the case with the U.S. FDA, manufacturers in China
undergo periodic inspections and must comply with international
quality standards such as ISO 13485 for medical devices. As part of
our agreement with SMI, SMI will underwrite the cost of securing
approval of LuViva with the CFDA and is responsible for securing
CFDA approval by July 2019, although we cannot be assured that SMI
will successfully secure such approval without delays or conditions
unacceptable to us, or at all.
In the
United States, permission to distribute a new device generally can
be met in one of two ways. The first process requires that a
pre-market notification (510(k) Submission) be made to the
U.S.
FDA to demonstrate that the device is as safe and effective as, or
substantially equivalent to, a legally marketed device that is not
subject to premarket approval (PMA). A legally marketed device is a
device that (1) was legally marketed prior to May 28, 1976, (2) has
been reclassified from Class III to Class II or I, or (3) has been
found to be substantially equivalent to another legally marketed
device following a 510(k) Submission. The legally marketed device
to which equivalence is drawn is known as the
“predicate” device. Applicants must submit descriptive
data and, when necessary, performance data to establish that the
device is substantially equivalent to a predicate device. In some
instances, data from human clinical studies must also be submitted
in support of a 510(k) Submission. If so, these data must be
collected in a manner that conforms with specific requirements in
accordance with federal regulations. The
U.S.
FDA must issue an order finding substantial equivalence before
commercial distribution can occur. Changes to existing devices
covered by a 510(k) Submission which do not significantly affect
safety or effectiveness can generally be made by us without
additional 510(k) Submissions.
The second process
requires that an application for premarket approval (PMA) be made
to the
U.S.
FDA to demonstrate that the device is safe and effective for its
intended use as manufactured. This approval process applies to most
Class III devices, including LuViva. In this case, two steps of
U.S.
FDA approval are generally required before marketing in the United
States can begin. First, investigational device exemption (IDE)
regulations must be complied with in connection with any human
clinical investigation of the device in the United States. Second,
the
U.S.
FDA must review the PMA application, which contains, among other
things, clinical information acquired under the IDE. The
U.S.
FDA will approve the PMA application if it finds that there is a
reasonable assurance that the device is safe and effective for its
intended purpose.
We completed
enrollment in our
U.S.
FDA pivotal trial of LuViva in 2008 and after the
U.S.
FDA reqiested two-years of follow-up-data for patients enrolled in
the study, the
U.S.
FDA accepted our completed PMA application on November 18,
2010, effective September 23, 2010, for substantive review.
On March 7, 2011, we announced that the
U.S.
FDA had inspected two clinical trial sites and audited our
clinical trial data base systems as part of its review
process and raised no formal compliance issues. On January 20,
2012, we announced our intent to seek an independent panel review
of our PMA application after receiving a
“not-approvable” letter from the
U.S.
FDA. On November 14, 2012 we filed an amended PMA with the
U.S.
FDA. On September 6, 2013, we received a letter from the
U.S.
FDA with additional questions and met with the
U.S.
FDA on May 8, 2014 to discuss our response. On July 25, 2014, we
announced that we had responded to the
U.S.
FDA’s most recent questions.
We received a
“not-approvable” letter from the
U.S.
FDA on May 15, 2015. We had a follow up meeting with the
U.S.
FDA to discuss a path forward on November 30, 2015, at which we
agreed to submit a detailed clinical protocol for
U.S.
FDA review so that additional studies can be completed. These
studies will not be completed in 2017, although we intend to
pursue U.S. FDA approval and start studies in 2017 once funds are
available, including from this offering. We remain committed
to obtaining
U.S.
FDA approval, but we are focused on international sales growth,
where we believe the commercial opportunities are larger and the
clinical need is more significant.
Based on our recent
communication with the U.S. FDA, and should the maximum $5 million
be raised in this offering, we intend to use up to $1.0 million of
the net proceeds over the next 18-24 months to accomplish the
following:
(1)
Write and submit to
the U.S. FDA a new protocol for the confirmatory
study
(2)
Engage regulatory
counsel to assist with the approval process
(3)
Meet with the U.S.
FDA if needed to finalize the protocol and identify the least
burdensome regulatory pathway
(4)
Identify clinical
sites for the study
(5)
Execute clinical
trial agreements for qualified clinical sites
(6)
Conduct the study
and analyze the results
(7)
Submit the study
results and supporting information to the U.S.
FDA
We estimate that
approximately $200,000 of the projected $1 million will be needed
to accomplish goals (1) through (4), with the majority of the costs
(estimated at $800,000) occurring when we engage clinical sites,
enroll patients, analyze results, and submit them to the U.S. FDA.
If the amount raised in this offering results in net proceeds
insufficient to conduct the actual study, we intend to use a
portion of the proceeds to accomplish goals (1) through (4) and
will seek additional funds later in 2017 or in 2018 to complete the
study, analyze the results and submit them to the U.S. FDA,
although there can be no assurance that such funds will be readily
available when needed.
The process of
obtaining clearance to market products is costly and time-consuming
in virtually all of the major markets in which we sell, or expect
to sell, our products and may delay the marketing and sale of our
products. Countries around the world have recently adopted more
stringent regulatory requirements, which are expected to add to the
delays and uncertainties associated with new product releases, as
well as the clinical and regulatory costs of supporting those
releases. No assurance can be given that our products will be
approved on a timely basis in any particular jurisdiction, if at
all. In addition, regulations regarding the development,
manufacture and sale of medical devices are subject to future
change. We cannot predict what impact, if any, those changes might
have on our business. Failure to comply with regulatory
requirements could have a material adverse effect on our business,
financial condition and results of operations.
Noncompliance with
applicable requirements can result in import detentions, fines,
civil penalties, injunctions, suspensions or losses of regulatory
approvals or clearances, recall or seizure of products, operating
restrictions, denial of export applications, governmental
prohibitions on entering into supply contracts, and criminal
prosecution. Failure to obtain regulatory approvals or the
restriction, suspension or revocation of regulatory approvals or
clearances, as well as any other failure to comply with regulatory
requirements, would have a material adverse effect on our business,
financial condition and results of operations.
Regulatory
approvals and clearances, if granted, may include significant
labeling limitations and limitations on the indicated uses for
which the product may be marketed. In addition, to obtain
regulatory approvals and clearances, the
U.S.
FDA and some foreign regulatory authorities impose numerous other
requirements with which medical device manufacturers must comply.
U.S.
FDA enforcement policy strictly prohibits the marketing of approved
medical devices for unapproved uses. Any products we manufacture or
distribute under
U.S.
FDA clearances or approvals are subject to pervasive and continuing
regulation by the
U.S.
FDA. The
U.S.
FDA also requires us to provide it with information on death and
serious injuries alleged to have been associated with the use of
our products, as well as any malfunctions that would likely cause
or contribute to death or serious injury.
The
U.S.
FDA requires us to register as a medical device manufacturer and
list our products. We are also subject to inspections by the
U.S.
FDA and state agencies acting under contract with the
U.S.
FDA to confirm compliance with good manufacturing practice. These
regulations require that we manufacture our products and maintain
documents in a prescribed manner with respect to manufacturing,
testing, quality assurance and quality control activities. The
U.S.
FDA also has promulgated final regulatory changes to these
regulations that require, among other things, design controls and
maintenance of service records. These changes will increase the
cost of complying with good manufacturing practice
requirements.
Distributors of
medical devices may also be required to comply with other foreign
regulatory agencies, and we or our distributors currently have
marketing approval for LuViva from Health Canada, COFEPRIS in
Mexico, the Ministry of Health in Kenya, and the Singapore Health
Sciences Authority. The time required to obtain these foreign
approvals to market our products may be longer or shorter than that
required in China or the United States, and requirements for those
approvals may differ from those required by the CFDA or the U.S.
FDA.
We are also subject
to a variety of other controls that affect our business. Labeling
and promotional activities are subject to scrutiny by the
U.S.
FDA and, in some instances, by the U.S. Federal Trade Commission.
The
U.S.
FDA actively enforces regulations prohibiting marketing of products
for unapproved users. We are also subject, as are our products, to
a variety of state and local laws and regulations in those states
and localities where our products are or will be marketed. Any
applicable state or local regulations may hinder our ability to
market our products in those regions. Manufacturers are also
subject to numerous federal, state and local laws relating to
matters such as safe working conditions, manufacturing practices,
environmental protection, fire hazard control and disposal of
hazardous or potentially hazardous substances. We may be required
to incur significant costs to comply with these laws and
regulations now or in the future. These laws or regulations may
have a material adverse effect on our ability to do
business.
Although our
marketing and distribution partners around the world assist in the
regulatory approval process, ultimately we are responsible
for obtaining and maintaining regulatory approvals for our
products. The inability or failure to comply with the varying
regulations or the imposition of new regulations would materially
adversely affect our business, financial condition and results of
operations.
Employees and Consultants
As of
December 31, 2016, we had ten regular employees and
three consultants to provide services to us on a full- or part-time
basis. Of the 13 people employed or engaged by us, 3 are engaged in
engineering, manufacturing and development, 3 are engaged in sales
and marketing activities, 3 are engaged in clinical
testing and regulatory affairs, 1 is engaged in research and
development activities, and 3 are engaged in administration and
accounting. No employees are covered by collective bargaining
agreements, and we believe we maintain good relations with our
employees.
Our ability to
operate successfully and manage our potential future growth depends
in significant part upon the continued service of key scientific,
technical, managerial and finance personnel, and our ability to
attract and retain additional highly qualified personnel in these
fields. Two of these key employees have an employment contract with
us; none are covered by key person or similar insurance. In
addition, if we are able to successfully develop and commercialize
our products, we likely will need to hire additional scientific,
technical, marketing, managerial and finance personnel. We face
intense competition for qualified personnel in these areas, many of
whom are often subject to competing employment offers. The loss of
key personnel or our inability to hire and retain additional
qualified personnel in the future could have a material adverse
effect on our business, financial condition and results of
operations.
Corporate History
We are a Delaware
corporation, originally incorporated in 1992 under the name
“SpectRx, Inc.,” and, on February 22, 2008, changed our
name to Guided Therapeutics, Inc. At the same time, we renamed our
wholly owned subsidiary, InterScan, which originally had been
incorporated as “Guided Therapeutics.”
PROPERTIES
Our corporate
offices, which also comprise our administrative, research and
development, marketing and production facilities, are located at
5835 Peachtree Corners East, Suite D, Norcross, Georgia 30092,
where we lease approximately 23,000 square feet under a lease that
expires in June 2017.
LEGAL
PROCEEDINGS
We are subject to
claims and legal actions that arise in the ordinary course of
business. However, we are not currently subject to any claims or
actions that we believe would have a material adverse effect on our
financial position or results of operations.
MARKET
FOR OUR COMMON STOCK AND RELATED STOCKHOLDER MATTERS
Our common stock is
dually quoted on the OTC Bulletin Board (OTCBB) and the OTCQB
quotation systems under the ticker symbol “GTHP.” The
number of record holders of our common stock at March
27, 2017 was 210.
A
1:800 reverse stock split of all of our issued and outstanding
common stock was implemented on November 7, 2016. As a result of
the reverse stock split, every 800 shares of issued and outstanding
common stock was converted into 1 share of common stock. All
fractional shares created by the reverse stock split were rounded
to the nearest whole share. The number of authorized shares of
common stock did not change.
The high and low
sales prices for the first quarter of 2017 and calendar years 2016
and 2015, as reported by the OTCBB, are as set forth in the
following table. All share prices reflect the 1:800 reverse stock
split.
|
|
|
2017
|
|
2016
|
|
2015
|
|
|
|
High
|
|
Low
|
|
High
|
|
Low
|
|
High
|
|
Low
|
|
First Quarter*
|
|
$
|
2.13
|
|
|
$
|
0.31
|
|
|
$
|
1,352.00
|
|
|
$
|
85.60
|
|
|
$
|
18,400.00
|
|
|
$
|
11,200.00
|
|
|
Second Quarter
|
|
|
|
|
|
|
|
|
|
$
|
140.00
|
|
|
$
|
3.28
|
|
|
$
|
20,000.00
|
|
|
$
|
6,400.00
|
|
|
Third Quarter
|
|
|
|
|
|
|
|
|
|
$
|
7.84
|
|
|
$
|
0.80
|
|
|
$
|
9,600.00
|
|
|
$
|
4,000.00
|
|
|
Fourth Quarter
|
|
|
|
|
|
|
|
|
|
$
|
1.35
|
|
|
$
|
0.02
|
|
|
$
|
4,800.00
|
|
|
$
|
800.00
|
|
*Through
March 27, 2017.
Dividend Policy
We have not paid
any common-stock dividends since our inception and do
not intend to pay any dividends in the foreseeable
future.
Securities Authorized for Issuance Under Equity
Compensation Plans
All the securities
we have provided our employees, directors and consultants have been
issued under our stock option plans, which are approved by our
stockholders. We have issued common stock to other individuals that
are not employees or directors, in lieu of cash payments, that are
not part of any plan approved by our stockholders.
Securities
authorized for issuance under equity compensation plans, as of
March 27, 2017:
|
|
Number of securities to be issued upon
exercise of outstanding options, warrants and
rights
|
Weighted-average exercise price of outstanding
options, warrants and rights
|
Number of securities
remaining available for future issuance under
equity compensation plans (excluding securities reflected in column
(a))
|
|
|
|
|
|
|
Equity compensation plans approved by security
holders
|
125
|
$
37,920
|
–
|
|
Equity compensation plans not approved by
security holders
|
–
|
–
|
–
|
|
TOTAL
|
125
|
$
37,920
|
–
|
MANAGEMENT’S
DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION
AND RESULTS OF OPERATIONS
The following discussion should be read in
conjunction with our financial statements and notes thereto
accompanying this prospectus.
Overview
We are a medical
technology company focused on developing innovative medical devices
that have the potential to improve healthcare. Our primary focus is
the sales and marketing of our LuViva® Advanced Cervical Scan
non-invasive cervical cancer detection device. The underlying
technology of LuViva primarily relates to the use of biophotonics
for the non-invasive detection of cancers. LuViva is designed to
identify cervical cancers and precancers painlessly, non-invasively
and at the point of care by scanning the cervix with light, then
analyzing the reflected and fluorescent light.
LuViva provides a
less invasive and painless alternative to conventional tests for
cervical cancer screening and detection. Additionally, LuViva
improves patient well-being not only because it eliminates pain,
but also because it is convenient to use and provides rapid results
at the point of care. We focus on two primary applications for
LuViva: first, as a cancer screening tool in the developing world,
where infrastructure to support traditional cancer-screening
methods is limited or non-existent, and second, as a triage
following traditional screening in the developed world, where a
high number of false positive results cause a high rate of
unnecessary and ultimately costly follow-up tests.
We are a Delaware
corporation, originally incorporated in 1992 under the name
“SpectRx, Inc.,” and, on February 22, 2008, changed our
name to Guided Therapeutics, Inc. At the same time, we renamed our
wholly owned subsidiary, InterScan, which originally had been
incorporated as “Guided Therapeutics.”
Since our
inception, we have raised capital through the public and private
sale of debt and equity, funding from collaborative arrangements,
and grants.
Our prospects must
be considered in light of the substantial risks, expenses and
difficulties encountered by entrants into the medical device
industry. This industry is characterized by an increasing number of
participants, intense competition and a high failure rate. We have
experienced operating losses since our inception and, as of
December 31, 2016 we have an accumulated deficit of
approximately $127.6 million. To date, we have engaged
primarily in research and development efforts and the early stages
of marketing our products. We do not have significant experience in
manufacturing, marketing or selling our products. We may not be
successful in growing sales for our products. Moreover, required
regulatory clearances or approvals may not be obtained in a timely
manner, or at all. Our products may not ever gain market acceptance
and we may not ever generate significant revenues or achieve
profitability. The development and commercialization of our
products requires substantial development, regulatory, sales and
marketing, manufacturing and other expenditures. We expect our
operating losses to continue through at least the end of
2017 as we continue to expend substantial resources to
complete commercialization of our products, obtain regulatory
clearances or approvals, build our marketing, sales, manufacturing
and finance capabilities, and conduct further research and
development.
Our product
revenues to date have been limited. In 2016, the
majority of our revenues were from the sale of LuViva devices and
disposables, as well as some revenue from grants from the NIH and
licensing agreement fees received. We expect that the majority of
our revenue in 2017 will be derived from revenue from
the sale of LuViva devices and disposables.
Current Demand for LuViva
Our view of
current demand for our products is based on information provided
by customers, and leads us to anticipate
purchase orders for approximately $3 million to $4 million in
LuViva devices and disposables in 2017. Based on those
anticipated purchase orders, we expect actual sales of $750,000 to
$1 million in 2017. We cannot be assured that we will
generate all or any of these additional purchase orders, or that
existing orders will not be canceled by the customers, or
that parts to build product will be available in a timely manner to
meet demand, such that existing orders will result in actual
sales. Because we have a short history of sales of our
products, we cannot confidently predict future sales of our
products beyond this time frame, and cannot be assured of any
particular amount of sales. Accordingly, we have not identified any
particular trends with regard to sales of our
products.
We currently have
several upcoming milestones on which we are working actively and
that we believe we can execute on in the near term. We are shipping
five units to our Chinese distributor so that it can initiate the
work on generating clinical data for CFDA approval. Domestically,
we plan to initiate work on generating additional data for our U.S.
FDA submission upon completion of this financing. We are also
working with our distributor for Russia to aid it in the regulatory
process there. We also expect to finalize negotiations with an
experienced health care distributor for India in the near
term.
Recent Developments
Convertible Promissory
Note.
On February 13, 2017, we entered into a securities
purchase agreement with Auctus Fund, LLC for the issuance and sale
to Auctus of $170,000 in aggregate principal amount of a 12%
convertible promissory note for an aggregate purchase price of
$156,400 (representing a $13,600 original issue discount). On
February 13, 2017, we issued the note to Auctus. Pursuant to the
purchase agreement, we also issued to Auctus a warrant exercisable
to purchase an aggregate of 200,000 shares of our common stock. The
warrant is exercisable at any time, at an exercise price per share
equal to $0.77 (110% of the closing price of the common stock on
the day prior to issuance), subject to certain customary
adjustments and price-protection provisions contained in the
warrant. The warrant has a five-year term. The note matures nine
months from the date of issuance and, in addition to the original
issue discount, accrues interest at a rate of 12% per year. We may
prepay the note, in whole or in part, for 115% of outstanding
principal and interest until 30 days from issuance, for 125% of
outstanding principal and interest at any time from 31 to 60 days
from issuance, and for 130% of outstanding principal and interest
at any time from 61 days from issuance to 180 days from issuance.
After six months from the date of issuance, Auctus may convert the
note, at any time, in whole or in part, into shares of our common
stock, at a conversion price equal to the lower of the price
offered in our next public offering or a 40% discount to the
average of the two lowest trading prices of the common stock during
the 20 trading days prior to the conversion, subject to certain
customary adjustments and price-protection provisions contained in
the note. The note includes customary events of default provisions
and a default interest rate of 24% per year. Upon the occurrence of
an event of default, Auctus may require us to redeem the note (or
convert it into shares of common stock) at 150% of the outstanding
principal balance plus accrued and unpaid interest. In connection
with the transaction, we agreed to reimburse Auctus for $30,000 in
legal and diligence fees, of which we paid $10,000 in cash and
$20,000 in restricted shares of common stock, valued at $0.40 per
share (a 42.86% discount to the closing price of the common stock
on the day prior to issuance).
SMI License
Agreement.
On January 22, 2017, we entered into a license
agreement with SMI, pursuant to which we granted SMI an exclusive
global license to manufacture the LuViva device and related
disposables (subject to a carve-out for manufacture in Turkey) and
exclusive distribution rights in the Peoples Republic of China,
Macau, Hong Kong and Taiwan. In exchange for the license, SMI will
pay a $1.0 million licensing fee, payable in five installments
through October 2017, underwrite the cost of securing approval of
LuViva with the Chinese Food and Drug Administration, or CFDA, and,
once it obtains CFDA approval, pay $1.90 royalty on each disposable
sold in the territories by purchasing directly from us a Controlled
Programmable Chip (CPC), necessary for the operation of disposable
unit. Pursuant to the SMI agreement, SMI must become capable of
manufacturing LuViva in accordance with ISO 13485 for medical
devices by the second anniversary of the SMI agreement, and achieve
CFDA approval by July 22, 2019, or else forfeit the license. During
2017, SMI must purchase no fewer than ten devices at $13,000 each
(with up to four devices pushed to 2018 if there is a delay in
obtaining approval from the CFDA). In the three full calendar
yearsfollowing CFDA approval, SMI must sell a minimum of 3,500
devices (500 in the first year, 1,000 in the second, and 2,000 in
the third), and purchase a minimum of 25,200,000 CPCs from us. If
SMI is able to meet these minimums or otherwise agrees to make up
the difference between payments for CPC orders and the minimum
payments to GTI (i.e., “pay to play”), this would
result in revenues of $47,880,000 to us over the three-year period.
Otherwise, SMI would forfeit the license. In any event, we cannot
be assured that SMI will be able to secure CFDA approval or meet
its minimum sales targets, or agree to make up the difference to
preserve their license, and therefore we cannot be assured of any
such revenue. As manufacturer of the devices and disposables, SMI
will be obligated to sell each to us at costs no higher than our
current costs. As partial consideration for, and as a condition to,
the license, and to further align the strategic interests of the
parties, we agreed to issue $1.0 million in shares of our common
stock to SMI, in five installments through October 2017, at a price
per share equal to the lesser of the average closing price for the
five days prior to issuance and $1.25. On March 28, 2017, we
amended the license agreement via a letter agreement to limit
SMI’s beneficial ownership of our common stock on any given
date to no more than 4.99% of our then-outstanding common stock. In
connection with the letter agreement, we agreed to issue to SMI
three warrants, each exercisable for 15,000 shares of our common
stock, to be issued in conjunction with the next three cash
payments by SMI under the license agreement. Each warrant will be
immediately exercisable (subject to the beneficial ownership
limitation), have an exercise price equal to the lesser of the
closing price per share of our common stock for the average of five
consecutive days preceding the payment by SMI and $1.25 per share,
and have a term of five years.
In order to
facilitate the SMI agreement, immediately prior to its execution we
entered into an agreement with Shenghuo Medical, LLC, regarding our
previous license to Shenghuo (see “—Liquidity and
Capital Resources”). Under the terms of the new agreement,
Shenghuo agreed to relinquish its manufacturing license and its
distribution rights in SMI’s territories, and to waive its
rights under the original Shenghuo agreement, all for as long as
SMI performs under the SMI agreement. As consideration, we have
agreed to split with Shenghuo the licensing fees and net royalties
from SMI that we will receive under the SMI agreement. Should the
SMI agreement be terminated, we have agreed to re-issue the
original license to Shenghuo under the original terms. Our COO and
director, Mark Faupel, is a shareholder of Shenghuo, and another
director, Richard Blumberg, is a managing member of
Shenghuo.
Critical Accounting
Policies
Our material
accounting policies, which we believe are the most critical to an
investors understanding of our financial results and condition, are
discussed below. Because we are still early in our enterprise
development, the number of these policies requiring explanation is
limited. As we begin to generate increased revenue from different
sources, we expect that the number of applicable policies and
complexity of the judgments required will increase.
Revenue Recognition:
We recognize
revenue from contracts on a straight line basis, over the terms of
the contract. We recognize revenue from grants based on the grant
agreement, at the time the expenses are incurred. Revenue from the
sale of the Company’s products is recognized upon shipment of
such products to its customers.
Valuation of Deferred Taxes:
We
account for income taxes in accordance with the liability method.
Under the liability method, we recognize deferred assets and
liabilities based upon anticipated future tax consequences
attributable to differences between financial statement carrying
amounts of assets and liabilities and their respective tax bases.
We establish a valuation allowance to the extent that it is more
likely than not that deferred tax assets will not be utilized
against future taxable income.
Valuation of Equity Instruments Granted to
Employee, Service Providers and Investors:
On the date
of issuance, the instruments are recorded at their fair value as
determined using either the Black-Scholes valuation model or Monte
Carlo Simulation model. See Note 4 to the consolidated financial
statements accompanying this report for the assumptions used in the
Black-Scholes valuation.
Allowance for Accounts
Receivable:
We estimate losses from the inability of
our customers to make required payments and periodically review the
payment history of each of our customers, as well as their
financial condition, and revise our reserves as a
result.
Inventory Valuation:
All
inventories are stated at lower of cost or market, with cost
determined substantially on a “first-in, first-out”
basis. Selling, general, and administrative expenses are not
inventoried, but are charged to expense when
purchased.
Reverse Stock Split:
A 1:800 reverse stock split of all of our issued and
outstanding common stock was implemented on November 7,
2016. As a result of the reverse stock split, every 800
shares of issued and outstanding common stock was converted into 1
share of common stock. All fractional shares created by the reverse
stock split were rounded to the nearest whole share. The number of
authorized shares of common stock did not change.
The reverse stock split decreased our
issued and outstanding shares of common stock from 453,694,400
shares of common stock to 570,707 shares as of that
date. Unless otherwise specified, all per share amounts are
reported on a post-stock split basis, as of December
31, 2016. On February 24, 2016, the Company had also
implemented a 1:100 reverse stock split of its issued and
outstanding common stock.
Results of Operations
Comparison of 2016 and 2015
Sales Revenue, Cost of
Sales and Gross Loss from Devices and Disposables:
Revenues from the sale of LuViva devices for
2016 and 2015 were approximately $605,000 and $564,000,
respectively. Revenues in 2016 were approximately, $41,000 or 7%
higher when compared to the same period in 2015, due
to increased sales in new territories. Related costs of
sales and valuation allowances on the net
realizable values were approximately $493,000 and $537,000 in
2016 and 2015, respectively. Costs of sales in 2016, were
approximately, $44,000 or 8% lower when compared to the same
period in 2015, due to reduction in material costs. This
resulted in a gross profit of approximately $112,000 on the
sales of devices and disposables for 2016 compared
with a gross profit of approximately $27,000 for
2015.
Research and Development
Expenses:
Research and development expenses for 2016,
decreased to approximately $733,000, from approximately $1,477,000
in 2015. The decrease of $744,000, or 50%, was primarily due to
reduction in research and development payroll
expenses.
Sales and Marketing
Expenses:
Sales and marketing expenses for 2016, decreased
to approximately $393,000, compared to $718,000 in 2015. The
decrease, of approximately $325,000, or 45% was primarily due to
Company-wide expense reduction and cost savings
efforts.
General and Administrative
Expense:
General and administrative expenses for 2016,
decreased to approximately $2,806,000, compared to $4,101,000 for
the same period in 2015. The decrease of approximately $1,295,000,
or 32%, was primarily related to lower compensation and option
expenses incurred during the same period.
Other Income:
Other
income was approximately $68,000 in 2016, compared to $74,000 in
2015.
Interest Expense:
Interest expense for 2016 increased to approximately $1,895,000,
compared to $1,317,000 for the same period in 2015. The increase of
approximately $578,000, or 44%, was primarily related to
amortization expense of debt issuance cost and penalty on event
default of convertible debt that were higher than the same period
in 2015.
Fair Value of Warrants
Expense:
Fair value of warrants recovery for 2016, increased
to approximately $1,677,000 compared to $568,000 for the same
period in 2015. The increase of approximately $1,109,000, or 195%
was primarily due to the significant changes in warrant conversion
prices, in the fiscal year ended December 31,
2015.
Net Loss:
Net loss
attributable to common stockholders deceased to approximately
$4,995,000, or $24.62 per share, in 2016, from $9,503,000, or
$5,939.38 per share, in 2015. The decrease in the net loss of
$4,508,000, or 47% was for reasons outlined
above.
There
was no income tax benefit recorded for 2016 or 2015,
due to recurring net operating losses.
Liquidity and Capital Resources
Since
our inception, we have raised capital through the public and
private sale of debt and equity, funding from collaborative
arrangements, and grants. At December 31, 2016, we had cash of
approximately $14,000 and a negative working capital of
approximately $8.3 million.
Our
major cash flows for the year ended December 31, 2016 consisted of
cash out-flows of $1.8 million from operations, including
approximately $4.0 million of net loss, and a net change from
financing activities of $1.8 million, which primarily represented
the proceeds received from issuance of common stock and warrants,
proceeds from debt financing, as well as exercise of outstanding
warrants and options.
On
March 16, 2015 and March 19, 2015, we entered into subscription
agreements with certain accredited investors, pursuant to which we
agreed to sell an aggregate of 50 shares of our common stock and
three-year warrants to purchase an additional 25 shares with an
exercise price per share of $20,400, for an aggregate purchase
price of $720,000.
On June
29, 2015, we entered into a securities purchase agreement with
certain accredited investors (including John Imhoff and Mark
Faupel, two of our directors), amended on September 3, 2015,
for the issuance and sale of shares of an aggregate of 7,903 shares
of our Series C preferred stock, at a purchase price of $750per
share and an initial conversion price of $76.00 per share, and
five-year warrants exercisable to purchase an aggregate of
approximately 156,000 shares of our common stock at an initial
exercise price of $76.00 per share, subject to certain customary
adjustments and anti-dilution provisions.
On
February 11, 2016, we consented to an assignment of our outstanding
secured promissory note to two accredited investors, Aquarius
Opportunity Fund and GPB Debt Holdings II LLC. In connection with
the assignment, the holders waived an ongoing event of default
under the notes related to our minimum market capitalization, and
agreed to eliminate the requirement going forward. On March 7,
2016, we further amended the notes to eliminate the volume
limitations on sales of common stock issued or issuable upon
conversion of the notes. GPB subsequently sold the note to
GHS.
On
February 11, 2016, we entered into a securities purchase agreement
with GPB Debt Holdings II LLC for the issuance and sale on February
12, 2016 of $1.4375 million in aggregate principal amount of a
senior secured convertible note for an aggregate purchase price of
$1.15 million (a 20% original issue discount). The note matures on
the second anniversary of issuance and, in addition to the 20%
original issue discount, accrues interest at a rate of 17% per
year. Subject to certain restrictions, it is convertible at any
time, in whole or in part, at the holderís option, into shares
of our common stock, at an initial conversion price equal to
$640.00 per share, subject to certain customary adjustments and
anti-dilution provisions. In addition, the investor received a
five-year warrant exercisable to purchase an aggregate of
approximately 2,247 shares of our common stock with an initial
exercise price of $640.00 per share, subject to certain customary
adjustments and anti-dilution provisions. As of March 27, 2017, as
a result of the operation of the anti-dilution provisions, the
conversion price of the note and the exercise price of the warrants
were $0.2639 per share. On March 18, 2016, our placement agent
received a warrant with similar terms exercisable for 108 shares.
In connection with the transaction, on February 12, 2016, we and
the investor entered into a four-year consulting agreement,
pursuant to which the investor will provide management consulting
services to us in exchange for a royalty payment, payable
quarterly, equal to 3.5% of our revenues from the sale of
products.
On
February 24, 2016, we implemented a 1:100 reverse stock split of
all of our issued and outstanding common stock. As a result of the
reverse stock split, every 100 shares of issued and outstanding
common stock was converted into 1 share of common stock. All
fractional shares created by the reverse stock split were rounded
to the nearest whole share. The number of authorized shares of
common stock did not change.
Between
April 27, 2016 and May 3, 2016, we entered into various agreements
with certain holders of our Series C preferred stock, including
John Imhoff and Mark Faupel, two of our directors, pursuant to
which those holders separately agreed to exchange each share of
Series C preferred stock held for 2.25 shares of our newly created
Series C1 preferred stock and 12 shares of our common stock. In
connection with these exchanges, each holder also agreed to
exchange the $1,000 stated value per share of the holderís
shares of Series C1 preferred stock for new securities that we
separately issue in the next qualifying financing we undertake on a
dollar-for-dollar basis. In total, for 1,916 shares of Series C
preferred stock surrendered, we issued 4,311 shares of Series C1
preferred stock and 22,996 shares of common stock. The Series C1
preferred stock has terms that are substantially the same as the
Series C preferred stock, except that the Series C1 preferred stock
does not pay dividends (unless and to the extent declared on the
common stock) or at-the-market “make-whole payments.”
Separately, on April 27, 2016, we entered into a rollover and
amendment agreement with a prior holder of Series C preferred
stock, Aquarius Opportunity Fund, pursuant to which Aquarius agreed
to exchange any shares of Series C preferred stock (stated value
plus make-whole dividend), as well as any remaining principal and
accrued interest on our secured promissory note Aquarius holds, for
new securities that we issue in our next financing, all on a
dollar-for-dollar basis, as long as the next financing involves at
least $1 million in cash from investors unaffiliated with Aquarius.
Aquarius sold all of its sharesof Series C preferred stock and its
portion of our secured promissory note to GHS. Aquarius also agreed
to return to us for cancelation warrants exercisable for 903 shares
of our common stock that it held. Except in the event of an
additional $50,000 cash investment by Aquarius in the qualifying
financing, Aquarius has agreed to execute a customary
“lockup” agreement in connection with the financing.
Finally, Aquarius, as the holder of a majority of the outstanding
Series C preferred stock, agreed to amend the Series C stock
purchase agreement to eliminate any participation rights held by
the Series C shareholders and to waive operation of certain
anti-dilution provisions of the Series C that would otherwise
have been triggered.
On May
4, 2016, May 26, 2016, and June 22, 2016, Aquarius Opportunity Fund
advanced us a total of $107,500 for 2% simple interest notes due
the earlier of December 31, 2016 or consummation of our next
public offering. Also on May 26, 2016, GPB Debt Holdings II
LLC, holder of our outstanding senior secured convertible note,
advanced as an additional $87,500, on the same terms as their note.
We intend to offer Aquarius the opportunity to participate in
our next public offering at least up to the extent of
the outstanding principal and interest on its cash advances, by
extinguishing all or a portion of the debt on a dollar-for-dollar
basis.
On June 5, 2016, we entered into a license
agreement with Shenghuo Medical, LLC pursuant to which we granted
Shenghuo an exclusive license to manufacture, sell and distribute
LuViva in Taiwan, Brunei Darussalam, Cambodia, Laos, Myanmar,
Philippines, Singapore, Thailand, and Vietnam. Shenghuo was already
our exclusive distributor in China, Macau and Hong Kong, and the
license extended to manufacturing in those countries as well. Under
the terms of the license agreement, once Shenghuo was capable of
manufacturing LuViva in accordance with ISO 13485 for medical
devices, Shenghuo would pay us a royalty equal to $2.00 or 20% of
the distributor price (subject to a discount under certain
circumstances), whichever is higher, per disposable distributed
within Shenghuoís exclusive territories. In connection with
the license grant, Shenghuo would underwrite the cost of securing
approval of LuViva with Chinese Food and Drug Administration. At
its option, Shenghuo also would provide up to $1.0 million in
furtherance of our efforts to secure regulatory approval for LuViva
from the U.S. Food and Drug Administration, or U.S. FDA, in
exchange for the right to receive payments equal to 2% of our
future sales in the United States, up to an aggregate of $4.0
million. Pursuant to the license agreement, Shenghuo had the option
to have a designee appointed to our board of directors (and Richard
Blumberg is that designee). As partial consideration for, and as a
condition to, the license, and to further align the strategic
interests of the parties, on March 24, 2017 we issued a $300,000
convertible note to Shenghuo, in exchange for Shenghuoís June
2016 aggregate cash investment of $200,000. The unsecured note
accrues interest at 20% per year, compounded annually, from January
1, 2017 until repayment, and is convertible into shares of our
common stock at a conversion price per share of $13.92, subject to
customary anti-dilution adjustment. We also issued Shenghuo a
five-year warrant exercisable immediately for 17,239 shares of
common stock at an exercise price equal to the conversion price of
the note, subject to customary anti-dilution
adjustment.
On January 22, 2017, we entered intoa license
agreement with SMI, pursuant to which we granted SMI an exclusive
global license to manufacture the LuViva device and related
disposables (subject to a carve-out for manufacture in Turkey) and
exclusive distribution rights in the Peoples Republic of China,
Macau, Hong Kong and Taiwan. In order to facilitate the SMI
agreement, immediately prior to its execution we entered into an
agreement with Shenghuo, regarding the previous license to
Shenghuo. Under the terms of the new agreement, Shenghuo agreed to
relinquish its manufacturing license and its distribution rights in
SMIís territories, and to waive its rights under the original
Shenghuo agreement, all for as long as SMI performs under the SMI
agreement
. See “óRecent
Developments.”
Between
June 13, 2016 and June 14, 2016, we entered into various agreements
with holders of certain warrants (including John Imhoff, one of our
directors) originally issued in May 2013, and with GPB Debt
Holdings II LLC, holder of a warrant issued February 12, 2016,
pursuant to which each holder separately agreed to exchange
warrants for either (1) shares of common stock equal to 166% of the
number of shares of common stock underlying the surrendered
warrants, or (2) new warrants exercisable for 200% of the number of
shares underlying the surrendered warrants, but without certain
anti-dilution protections included with the surrendered warrants.
As a result of the exchanges, we effectively eliminate any
potential exponential increase in the number of underlying shares
issuable upon exercise of our outstanding warrants. In total, for
surrendered warrants then-exercisable for an aggregate of 1,814,598
shares of common stock (but subject to exponential increase upon
operation of certain anti-dilution provisions), we issued or are
obligated to issue 16,898 shares of common stock and new warrants
that, if exercised as of June 14, 2016, would have been exercisable
for an aggregate of 3,608,835 shares of common stock. In certain
circumstances, in lieu of presently issuing all of the shares (for
each holder that opted for shares of common stock), we and the
holder further agreed that we will, subject to the terms and
conditions set forth in the applicable warrant exchange agreement,
from time to time, be obligated to issue the remaining shares to
the holder. No additional consideration will be payable in
connection with the issuance of the remaining shares. The holders
that elected to receive shares for their surrendered warrants have
agreed that they will not sell shares on any trading day in an
amount, in the aggregate, exceeding 20% of the composite aggregate
trading volume of the common stock for that trading day. The
holders that elected to receive new warrants will be required to
surrender their old warrants upon consummation of our next public
offering. The new warrants will have an initial exercise price
equal to the exercise price of the surrendered warrants as of
immediately prior to consummation of the offering, subject to
customary “downside price protection” for as long as
our common stock is not listed on a national securities exchange,
and will expire five years from the date of issuance.
On
September 6, 2016, we entered into a royalty agreement with one of
our directors, John Imhoff, and another stockholder, Dolores
Maloof, pursuant to which we sold to them a royalty of future sales
of single-use cervical guides for LuViva. Under the terms of the
royalty agreement, and for consideration of $50,000, we will pay
them an aggregate perpetual royalty initially equal to $0.10, and
from and after October 2, 2016, equal to $0.20, for each disposable
that we sell (or that is sold by a third party pursuant to a
licensing arrangement with us).
On
November 2, 2016, we entered into a lockup and exchange agreement
with GHS Investments, LLC, holder of approximately $221,000 in
outstanding principal amount of our secured promissory note and all
of the outstanding shares of our Series C preferred stock. Pursuant
to the agreement, upon the effectiveness of the 1:800 reverse stock
split and continuing for 45 days after, GHS and its affiliates were
prohibited from converting any portion of the secured promissory
note or any of the shares of Series C preferred stock or selling
any of our securities that they beneficially owned. We agreed that,
upon consummation of our next financing, we would use $260,000 of
net cash proceeds first, to repay GHSís portion of the secured
promissory note and second, with any remaining amount from the
$260,000, to repurchase a portion of GHSís shares of Series C
preferred stock. In addition, GHS has agreed to exchange the stated
value per share (plus any accrued but unpaid dividends) of its
remaining shares of Series C preferred stock for new securities of
the same type that we separately issue in the next qualifying
financing we undertake, on a dollar-for-dollar basis in a private
placement exchange.
A
1:800 reverse stock split of all of our issued and outstanding
common stock was implemented on November 7, 2016. As a result of
the reverse stock split, every 800 shares of issued and outstanding
common stock was converted into 1 share of common stock. All
fractional shares created by the reverse stock split were rounded
to the nearest whole share. The number of authorized shares of
common stock did not change.
On December 7,
2016, we entered into an exchange agreement with GPB Debt Holdings
II LLC with regard to the $1,525,000 in outstanding principal
amount of senior secured convertible note originally issued to GPB
on February 11, 2016, and the $306,863 in outstanding principal
amount of our secured promissory note that GPB holds. Pursuant to
the exchange agreement, upon completion of the next financing
resulting in at least $1 million in cash proceeds, GPB will
exchange both securities for a new convertible note in principal
amount of $1,831,863. The new convertible note will mature on
February 12, 2018 and will accrue interest at a rate of 19% per
year. We will pay monthly interest coupons and, beginning one year
after issuance, will pay amortized quarterly principal payments.
Subject to resale restrictions under Federal securities laws and
the availability of sufficient authorized but unissued shares of
our common stock, the new convertible note will be convertible at
any time, in whole or in part, at the holderís option, into
shares of our common stock, at a conversion price equal to the
price offered in the qualifying financing that triggers the
exchange, subject to certain customary adjustments and
anti-dilution provisions contained in the new convertible note. The
new convertible note will include customary event of default
provisions and a default interest rate of the lesser of 21% or the
maximum amount permitted by law. Upon the occurrence of an event of
default, GPB will be entitled to require us to redeem the
newconvertible note at 120% of the outstanding principal balance.
The new convertible note will be secured by a lien on all of our
assets, including our intellectual property, pursuant to the
security agreement entered into by us and GPB in connection with
the issuance of the original senior secured convertible note. As an
inducement to GPB to enter into these transactions, we agreed to
increase the royalty payable to GPB pursuant to its consulting
agreement with us from 3.5% to 3.85% of revenues from the sales of
our products.
On December 28,
2016, we entered into a securities purchase agreement with
RedDiamond Partners LLC for the issuance and sale to RedDiamond of
up to $330,000 in aggregate principal amount of 10% original
issuance discount convertible promissory notes, for an aggregate
purchase price of $300,000. On that date, we issued to RedDiamond a
note in the principal amount of $222,000, for a purchase price of
$200,000. At our option, at any time within 60 days from that
issuance, we may issue another note in the principal amount of
$110,000, for a purchase price of $100,000, subject to customary
conditions contained in the purchase agreement. The notes mature
six months from their date of issuance and, in addition to the 10%
original issue discount,accrue interest at a rate of 10% per year.
We may prepay the notes, in whole or in part, for 115% of
outstanding principal and interest until 30 days from issuance, for
125% of outstanding principal and interest at any time from 31 to
60 days from issuance, and for 130% of outstanding principal and
interest at any time from 61 days from issuance until immediately
prior to the maturity date. After six months from the date of
issuance (i.e., if we fail to repay all principal and interest due
under the notes at the maturity date), RedDiamond may convert the
notes, at any time, in whole or in part, into shares of our common
stock, at a conversion price equal to 60% of the lowest volume
weighted average price of our common stock during the 20 trading
days prior to conversion, subject to certain customary adjustments
and anti-dilution provisions contained in the
notes.
See
“óRecent Developments” for information regarding
capital-raising activities since December 31,
2016.
We will
be required to raise additional funds through public or private
financing, additional collaborative relationships or other
arrangements, as soon as possible. We cannot be certain that our
existing and available capital resources will be sufficient to
satisfy our funding requirements through the second quarter of
2017. We are evaluating various options to further reduce our cash
requirements to operate at a reduced rate, as well as options to
raise additional funds, including loans.
Generally,
substantial capital will be required to develop our products,
including completing product testing and clinical trials, obtaining
all required U.S. and foreign regulatory approvals and clearances,
and commencing and scaling up manufacturing and marketing our
products. Any failure to obtain capital would have a material
adverse effect on our business, financial condition and results of
operations. Our view of current demand for our products is
based on information provided by customers, and leads us to
anticipate purchase orders for approximately $3 million to $4
million in LuViva devices and disposables in 2017. Based on those
anticipated purchase orders, we expect actual sales of $750,000 to
$1 million in 2017. We cannot be assured that we will generate all
or any of these additional purchase orders, orthat existing orders
will not be canceled by the customers, or that parts to build
product will be available in a timely manner to meet demand, such
that existing orders will result in actual sales. Because we have a
short history of sales of our products, we cannot confidently
predict future sales of our products beyond this time frame, and
cannot be assured of any particular amount of sales. Accordingly,
we have not identified any particular trends with regard to sales
of our products. We currently do not have cash on hand
sufficient to build the inventory required to fill these orders,
and material delays in product deliveries could result in canceled
orders.
Our
financial statements have been prepared and presented on a basis
assuming we will continue as a going concern. The above factors
raise substantial doubt about our ability to continue as a going
concern, as more fully discussed in Note 1 to the consolidated
financial statements contained herein and in the report of our
independent registered public accounting firm accompanying our
financial statements contained in our annual report on Form 10-K
for the year ended December 31, 2016.
Off-Balance Sheet Arrangements
We have
no material off-balance sheet arrangements, no special purpose
entities, and no activities that include non-exchange-traded
contracts accounted for at fair value.
DIRECTORS
AND EXECUTIVE OFFICERS
Our executive
officers are elected by and serve at the discretion of our board of
directors. The following table lists information about our
directors and executive officers as of M
arch 27
, 2017:
|
Name
|
Age
|
Position with
Guided Therapeutics
|
|
Gene S. Cartwright, Ph.D.
|
62
|
Chief Executive Officer, President, Acting
Chief Financial Officer and Director
|
|
Mark Faupel, Ph.D.
|
61
|
Chief Operating Officer and
Director
|
|
Richard L. Fowler
|
60
|
Senior Vice President of
Engineering
|
|
Richard P. Blumberg
|
60
|
Director
|
|
John E. Imhoff, M.D.
|
67
|
Director
|
|
Michael C. James
|
58
|
Chairman and Director
|
Except as set forth
below, all of the executive officers have been associated with us
in their present or other capacities for more than the past five
years. Officers are elected annually by the board of directors and
serve at the discretion of the board. There are no family
relationships among any of our executive officers and
directors.
Gene S. Cartwright, Ph.D.
joined us in
January 2014 as the President, Chief Executive Officer and Acting
Chief Financial Officer. He was elected as a director on January
31, 2014. His most recent position was with Omnyx, LLC, a Joint
Venture between GE Healthcare and the University of Pittsburgh
Medical Center, where, as CEO for over four years he founded and
managed the successful development of products for the field of
Digital Pathology. Prior to his work with Omnyx, LLC, he was
President of Molecular Diagnostics for GE Healthcare. Prior to GE,
Dr. Cartwright was Divisional Vice President/General Manager for
Abbott Diagnostics’ Molecular Diagnostics business. In his 24
year career at Abbott, he also served as Divisional Vice President
for U.S. Marketing for five years. He received a Masters of
Management degree from Northwestern’s Kellogg School of
Management and also holds a Ph.D. in chemistry from Stanford
University and an AB from Dartmouth College.
Dr. Cartwright
brings over 30 years of experience working in the IVD diagnostics
industry. He has great experience in the diagnostics market both in
the development and introduction of new diagnostics technologies,
as well as extensive successful commercial experience with global
businesses. With his background and experience, Dr. Cartwright, as
President and Chief Executive Officer, as well as Acting Chief
Financial Officer, works with and advises the board as to how we
can successfully market and build LuViva international
sales.
Mark Faupel, Ph.D.
, rejoined us as
Chief Operating Officer and director on December 8, 2016. He
previously served on our board of directors through 2013 and has
more than 30 years of experience in developing non-invasive
alternatives to surgical biopsies and blood tests, especially in
the area of cancer screening and diagnostics. Dr. Faupel was one of
our co-founders and also served as our Chief Executive Officer from
May 2007 through 2013. Prior thereto was our Chief Technical
Officer from April 2001 to May 2007. Dr. Faupel has served as a
National Institutes of Health reviewer, is the inventor on 26 U.S.
patents and has authored numerous scientific publications and
presentations, appearing in such peer-reviewed journals as The
Lancet. Dr. Faupel earned his Ph.D. in neuroanatomy and physiology
from the University of Georgia. Dr. Faupel is also a
shareholder of Shenghuo Medical, LLC. See Item 13, Certain
Relationships and Related Transactions and Director
Independence.
Rick Fowler
, Senior Vice President of
Engineering is an accomplished Executive with significant
experience in the management of businesses that sell, market,
produce and develop sophisticated medical devices and
instrumentation. Mr. Fowler’s 25 plus years of experience
includes assembling and managing teams, leading businesses and
negotiating contracts, conducting litigation, and developing ISO,
CE, FDA QSR, GMP and GCP compliant processes and products. He is
adept at providing product life cycle management through effective
process definition and communication - from requirements gathering,
R&D feasibility, product development, product launch,
production startup and support. Mr. Fowler combines outstanding
analytical, out-of-the-box, and strategic thinking with strong
leadership, technical, and communication skills and he excels in
dynamic, demanding environments while remaining pragmatic and
focused. He is able to deliver high risk projects on time and under
budget as well as enhance operational effectiveness through
outstanding cross-functional team leadership (R&D, marketing,
product development, operations, quality assurance, sales, service,
and finance). In addition, Mr. Fowler is well versed in global
medical device regulatory and product compliance
requirements.
Richard P. Blumberg
was appointed to
the Board of Directors on November 10, 2016. Mr. Blumberg has been
a long-time investor in the Company. Since 1978, Mr. Blumberg has
been a Principal at Webster, Mrak & Blumberg, a medical-legal
and class action labor litigation firm. He is also currently the
Managing Member of Elysian Medical, LLC,a company with world-wide
rights for certain breast cancer detection technology. He served
from 2004 to 2007 as Chief Executive Officer of Energy Logics, a
wind power company that developed projects in Alberta, Canada and
Montana. Mr. Blumberg holds a B.S. in Electrical Engineering and
Computer Science from the University of Illinois and received a J.
D. from Stanford University. He also brings extensive experience as
a venture capitalist specializing in high-tech and life science
companies. Mr. Blumberg is also a Managing Member of Shenghuo
Medical, LLC. See Item 13, Certain Relationships and Related
Transactions and Director Independence.
John E. Imhoff, M.D.
has served as a
member of our Board of Directors since April 2006. Dr. Imhoff is an
ophthalmic surgeon who specializes in cataract and refractive
surgery. He is one of our principal stockholders and invests in
many other private and public companies. He has a B.S. in
Industrial Engineering from Oklahoma State University, an M.D. from
the University of Oklahoma and completed his ophthalmic residency
at the Dean A. McGee Eye Institute. He has worked as an ophthalmic
surgeon and owner of Southeast Eye Center since 1983.
Dr. Imhoff has
experience in clinical trials and in other technical aspects of a
medical device company. His background in industrial engineering is
especially helpful to us, especially as Dr. Imhoff can combine this
knowledge with clinical applications. His experience in the
investment community is invaluable to a public company often
undertaking capital raising efforts.
Michael C. James
has served as a member of
our Board of Directors since March 2007 and as Chairman of the
Board since October 15, 2013. Mr. James is also the Managing
Partner of Kuekenhof Capital Management, LLC, a private investment
management company, Chief Executive Officer and the Chief Financial
Officer of Inergetics, Inc., a nutraceutical supplements company
and also the Chief Financial Officer of Terra Tech Corporation,
which is a hydroponic and agricultural company. He also holds the
position of Managing Director of Kuekenhof Equity Fund, L.P. and
Kuekenhof Partners, L.P. Mr. James currently sits on the Board of
Directors of Inergetics; Inc. Mr. James was Chief Executive
Officer of Nestor, Inc. from January 2009 to September 2009
and served on their Board of Directors from July 2006 to June 2009.
He was employed by Moore Capital Management, Inc., a private
investment management company from 1995 to 1999 and held position
of Partner. He was employed by Buffalo Partners, L.P., a private
investment management company from 1991 to 1994 and held the
position of Chief Financial and Administrative Officer. He began
his career in 1980 as a staff accountant with Eisner LLP. Mr. James
received a B.S. degree in Accounting from Farleigh Dickinson
University in 1980.
Mr. James has
experience both in the areas of company finance and accounting,
which is invaluable to us during financial audits and offerings.
Mr. James has extensive experience in the management of both small
and large companies and his entrepreneurial background is relevant
as we develop as a company.
CORPORATE
GOVERNANCE
Board Meetings and
Committees
Our board of
directors held 12 meetings in 2016. No
director attended fewer than 75% of the meetings of the board of
directors or the committees on which he served during
2016. We encourage our directors to attend the annual
meeting of stockholders. In 2016, all of our directors
attended our annual meeting. Dr. Imhoff and Mr. James
serve on the audit, compensation and nomination committees.
Although we are not subject to the listing standards of any
national securities exchange or inter-dealer quotation system,
based on the definition of independence in the NASDAQ listing
standards, Dr. Imhoff and Mr. James are independent directors. The
board works with its members and management to identify new board
members, and will consider nominees recommended by stockholders.
Any recommendation should be addressed in writing to the Board of
Directors, c/o Corporate Secretary, 5835 Peachtree Corners East,
Suite D, Norcross, Georgia 30092.
The audit committee
selects and engages the independent registered public accounting
firm to audit our annual financial statements and pre-approves all
allowable audit services and any special assignments given to the
accountants. The audit committee also determines the planned scope
of the annual audit, any changes in accounting principles, the
effectiveness and efficiency of our internal accounting staff and
the independence of our external auditors. The audit committee met
12 times in 2016. The audit committee
currently consists of Dr. Imhoff and Mr. James. Each member of the
audit committee is independent in accordance with the NASDAQ
listing standards for audit committee independence and applicable
SEC regulations. None of the members of the audit committee has
participated in the preparation of our financial statements at any
time during the past three years. The board has also determined
that Mr. James and Dr. Imhoff meet the criteria specified under
applicable SEC regulations for an “audit committee financial
expert” and that the committee members are financially
sophisticated.
The board of
directors, in consultation with our Chief Executive Officer, sets
the compensation for our officers, reviews management organization
and development, reviews significant employee benefit programs and
establishes and administers executive compensation programs. The
compensation committee currently consists of Dr. Imhoff and Mr.
James, each of whom is independent under NASDAQ listing standards.
The compensation committee met 12 times in
2016.
The board of
directors, in consultation with our Chief Executive Officer,
reviews and recommends individuals to be nominated as directors.
Our board has historically evaluated all candidates based upon,
among other factors, a candidate’s financial literacy,
knowledge of our industry or other background relevant to our
needs, status as a stakeholder, independence, and willingness,
ability and availability for service. Other than the foregoing,
there have been no stated minimum criteria for director nominees,
although our board has considered such other factors as it has
deemed to be in the best interests of us and our stockholders. The
board has considered diversity as it has deemed appropriate in this
context (without having a formal diversity policy), given current
needs and the current needs of the board to maintain a balance of
knowledge, experience and capability. When considering diversity,
the board has considered diversity as one factor, of no greater or
lesser importance than other factors and has considered diversity
in a broad context of race, gender, age, business experience,
skills, international experience, education, other board experience
and other relevant factors.
The audit committee
and the compensation committee have each adopted charters, which
are available on our web site, at www.guidedinc.com/Investors.htm.
The nomination committee currently operates without a
charter.
Board Leadership Structure and Role in Risk
Oversight
Dr. Cartwright, our
President and Chief Executive Officer, also serves as a director;
our board is led by the Chairman, Mr. James, one of our independent
directors. Our board, as a whole, has responsibility for risk
oversight, with reviews of certain areas being conducted by the
relevant board committees that report on their deliberations to the
full board, as further described below. In addition, our management
regularly communicates with the board to discuss important risks
for their review and oversight, including regulatory risk and risks
stemming from periodic litigation or other legal matters in which
we are involved. Given the small size of the board, the board feels
that this structure for risk oversight is appropriate (except for
those risks that require risk oversight by independent directors
only).
The board of
directors is specifically charged with discussing risk management
(primarily financial and internal control risk), and receives
regular reports from management, independent auditors, internal
audit and outside legal counsel on risks related to, among others,
our financial controls and reporting. The board of directors
reviews risks related to compensation and makes recommendations to
the board with respect to whether our compensation policies are
properly aligned to discourage inappropriate risk-taking, and is
regularly advised by management and, as deemed appropriate, outside
legal counsel.
Communication with
Directors
Any stockholder is
welcome to communicate with any director or the board of directors
by writing to a director or the board as a whole, c/o Corporate
Secretary, 5835 Peachtree Corners East, Suite D, Norcross, Georgia
30092.
Director Compensation
None of our
directors received any compensation or reimbursement in cash in
2016; however, they did receive stock options or
shares, in lieu of cash, for 2016 and
2015, respectively, in connection with their services
as members of the board of directors and their service on board
committees.
Director Compensation Table, for year ended
December 31, 2016
|
Name and Principal
Position
|
|
|
Michael C. James, Chairman and
Director
|
8
|
|
John E. Imhoff, M.D., Director
|
8
|
|
Jonathan M. Niloff, M.D., former
Director
|
8
|
|
Linda Rosenstock, M.D., former
Director
|
8
|
EXECUTIVE
COMPENSATION
The purpose of our
compensation philosophy, policies and practices is to attract and
retain experienced, highly qualified executives critical to our
long-term success and enhancement of stockholder value. We rely on
a combination of base salary and customary benefits along with
long-term equity incentive compensation to achieve these
objectives. In establishing compensation packages for our executive
officers, the compensation committee believes that we provide our
executive officers with the opportunity to earn a competitive
annual base salary, and that options grants reward our executive
officers for meaningful performance that contributes to enhanced
long-term stockholder value and our general long-term financial
health, and helps to align the interests of our executive officers
with those of our stockholders.
Summary Compensation Table
The following table
lists specified compensation we paid or accrued during each of the
fiscal years ended December 31, 2015 and 2014 to the Chief
Executive Officer and our two other most highly compensated
executive officers, collectively referred to as the “named
executive officers,” in 2015:
2016 and 2015
Summa
ry Compensation Table
|
Name and Principal Position
|
Year
|
Salary
($)
|
Bonus
($)
|
Option Awards
($)(1)
|
Total
($)
|
|
Gene S.
Cartwright, Ph.D.
President,
CEO, Acting CFO and Director (2)
|
2016
2015
|
104,990
300,000
|
150,000
150,000
|
-
-
|
254,990
450,000
|
|
Mark
Faupel, Ph.D.
COO and Director
(3)
|
2016
2015
|
132,557
198,073
|
-
-
|
-
30,400
|
132,557
228,473
|
|
Richard
Fowler,
Senior
Vice President of Engineering
|
2016
2015
|
129,995
243,000
|
-
-
|
-
30,880
|
129,995
273,880
|
|
|
(1)
|
See Note 4 to the
audited consolidated financial statements that accompany this
prospectus.
|
|
|
(2)
|
All amounts reported as
accrued. Dr. Cartwright has elected to get paid partial salary, due
to our cash position.
|
|
|
(3)
|
In 2015, Dr. Faupel was
not employed by us, but instead provided consulting services to us
on an as-needed basis. On December 8, 2016, the board of directors
appointed Dr. Faupel as our new COO and director.
|
On March 2016, Dr.
Cartwright agreed to reduce his base salary compensation to $75,000
from $300,000. The board-granted performance bonus remained the
same at $150,000 for all both years, and he received usual
customary company benefits. During 2015, he also received 20,000
performance-based restricted shares of common stock, which will
vest as follows: (1) seven shares will vest if the stock price
closes at or above $1,200 for 30 consecutive trading days, and an
additional seven will vest on the first anniversary of such vesting
date, in each case subject to continuous employment through the
applicable vesting date; and (2) seven shares will vest if the
stock price closes at or above $200,000 for 30 consecutive trading
days, and an additional seven will vest on the first anniversary of
such vesting date, in each case subject to continuous employment
through the applicable vesting date. As of December 31, 2016, Dr.
Cartwrightís deferred salary plus interest was $361,526 and
his deferred bonus was $450,000.
Dr. Faupelís
2016 and 2015 compensation consisted of a base salary of $132,557
and $198,073, respectively, plus usual and customary company
benefits. He received no bonus in the years ended December 31, 2016
and 2015. In 2015, he received options to purchase 1,900 shares of
common stock, which vest over 48 months. As of December 31, 2016,
Dr. Faupelís remaining deferred salary plus interest and bonus
was $166,426. He also holds a promissory note of $290,704 for past
unpaid salary.
On March 2016, Mr.
Fowler began working half-time and agreed to reduce his base salary
compensation to $107,500 from $243,000 in 2015. For both years he
received the usual and customary company benefits. He received no
bonus in the years ended December 31, 2016 and 2015. In 2015, he
received options to purchase 1,930 shares of common stock, which
vest over 48 months. As of December 31, 2016, Mr. Fowlerís
total deferred salary plus interest was approximately
$320,042.
Outstanding Equity Awards to Officers at
December 31, 2016
|
|
Option
Awards
|
|
Name and Principal
Position
|
Number of
Securities
Underlying
Options
Exercisable (#)(1)
|
Number of Securities
Underlying
Options Un-exercisable
(#)
|
Equity Incentive Plan Awards: Number of
Securities Under-
lying Unexercised
Unearned Options (#)
|
Option
Exercise
Price
($)(2)
|
Option
Expiration
Date
|
|
Gene S. Cartwright,
Ph.D.
President, CEO,
Acting CFO and Director
|
2
|
-
|
3
|
21,600.00
|
12/31/2024
|
|
Mark Faupel,
Ph.D.
C
OO
and Director
|
32
|
-
|
3
|
57,600.00
|
12/31/2024
|
|
Richard
Fowler
Senior Vice
President of Engineering
|
11
|
-
|
3
|
47,200.00
|
12/31/2024
|
|
|
(1)
|
Represents fully vested
options.
|
|
|
(2)
|
Based on all outstanding
options.
|
Outstanding Equity Awards to Directors at
December 31, 2016
|
|
Option
Awards
|
|
Name and
Principal Position
|
Option Awards
(#)
|
Exercise Price
($)
|
|
John E. Imhoff, M.D., Director
|
16
|
26,400.00
|
|
Michael C. James, Chairman and
Director
|
13
|
16,000.00
|
|
Jonathan Niloff, M.D., former
Director
|
14
|
17,600.00
|
|
Linda Rosenstock, M.D., former
Director
|
14
|
16,800.00
|
SHARE
OWNERSHIP OF DIRECTORS, OFFICERS AND CERTAIN BENEFICIAL
OWNERS
The following table
lists information regarding the beneficial ownership of our equity
securities as of March 27, 2017 by (1) each
person whom we know to beneficially own more than 5% of the
outstanding shares of our common stock, (2) each director, (3) each
officer named in the summary compensation table below, and (4) all
directors and executive officers as a group. Unless otherwise
indicated, the address of each officer and director is 5835
Peachtree Corners East, Suite D, Norcross, Georgia
30092.
|
|
|
Series C
Preferred Stock (3)
|
Series C1
Preferred Stock (4)
|
|
Name
and Address of Beneficial Owner (1)
|
|
|
|
|
|
|
|
John E. Imhoff
(5)
|
5,995,843
|
80.18
%
|
-
|
-
|
2,400.75
|
55.67
%
|
|
Lynne Imhoff
(6)
|
1,685,362
|
53.06
%
|
-
|
-
|
675.00
|
15.65
%
|
|
Michael C.
James/Kuekenhof Equity Fund, LLP (7)
|
28
|
*
|
-
|
-
|
-
|
-
|
|
Gene Cartwright
(8)
|
38
|
*
|
-
|
-
|
-
|
-
|
|
Richard L. Fowler
(9)
|
16
|
*
|
-
|
-
|
-
|
-
|
|
Richard P. Blumberg
(10)
|
37
|
*
|
-
|
-
|
-
|
-
|
|
Mark Faupel
(11)
|
748,050
|
33.38
%
|
|
|
300.00
|
6.96
%
|
|
All directors and
executive officers as a group (6 persons) (12)
|
6,744,012
|
82.00
%
|
-
|
-
|
2,700.75
|
62.63
%
|
|
(*)
|
Less
than 1%.
|
|
|
(1)
|
Except
as otherwise indicated in the footnotes to this table and pursuant
to applicable community property laws, the persons named in the
table have sole voting and investment power with respect to all
shares of common stock.
|
|
|
(2)
|
Percentage
ownership is based on 1,494,741 shares of common stock outstanding
as of March 27, 2017. Beneficial ownership is determined in
accordance with the rules of the SEC, based on factors that include
voting and investment power with respect to shares. Shares of
common stock subject to convertible securities convertible or
exercisable within 60 days after therecord date, are deemed
outstanding for purposes of computing the percentage ownership of
the person holding those securities, but are not deemed outstanding
for purposes of computing the percentage ownership of any other
person. Note that certain of our outstanding securities, including
certain warrants and the shares of Series C1 preferred stock held
by the persons listed in this table, have anti-dilution
“ratchet” or “price-protection” provisions
that, when triggered, will increase the number of shares of common
stock underlying such securities. Subject to customary exceptions,
these provisions are triggered anytime we issue shares of common
stock to third parties at a price lower than the then-current
conversion price or exercise price of the subject securities. As a
result, the beneficial ownership reported in this table is only as
of the date presented, and the beneficial ownership amounts of the
persons in this table may increase on a future date, even though
such persons have not actually acquired any additional shares of
common stock.
|
|
|
(3)
|
As of
March 27, 2017, there were 1,422 shares of Series C
preferred stock outstanding, and each such share was convertible
into approximately 1,250 shares of common stock.
|
|
|
(4)
|
As of
March 27, 2017, there were 4,312.5 shares of Series C1
preferred stock outstanding, and each such share was convertible
into approximately 1,250 shares of common stock.
|
|
|
(5)
|
Shares
of common stock consist of 12,952 shares of common stock directly
held, 4,912 shares issuable upon exercise of warrants, 16 shares
subject to options, and 5,977,963 shares issuable upon
conversion of 2,400.75 shares of Series C1 preferred stock. Dr.
Imhoff is on the board of directors.
|
|
|
(6)
|
Shares
of common stock consist of 3,612 shares of common stock directly
held, 973 shares issuable upon exercise of warrants, and
1,680,777 shares issuable upon conversion of 675.00
shares of Series C1 preferred stock.
|
|
|
(7)
|
Shares
of commons stock consist of 10 shares of common stock directly
held, 4 shares issuable upon exercise of warrants, and 14 shares
subject to options. Mr. James is on the board of
directors.
|
|
|
(8)
|
Shares of commons stock consist of 29 shares of common stock
directly held, 4 shares issuable upon exercise of warrants, and 5
shares subject to options.
Dr. Cartwright is the CEO and on
the board of directors.
|
|
|
(9)
|
Shares
of commons stock consist of 2 shares of common stock directly held
and 14
shares subject
to options.
|
|
|
(10)
|
Shares
of common stock consist of 23 shares of common stock directly held
and 14 shares issuable upon exercise of warrants.
|
|
|
(11)
|
Shares
of common stock consist of 1,600 shares of common stock directly
held, 46 shares issuable upon exercise of warrants, 27 shares
subject to options, and 746,377 shares issuable upon
conversion of 300.00 shares of Series C1 preferred stock. Dr.
Faupel is the COO and on the board of directors.
|
|
|
(12)
|
Shares of commons stock consists of 12,993 shares of common stock
directly held, 4,920 shares issuable upon exercise of
warrants, 49 shares subject to options, and 6,724,340
shares issuable upon conversion of 2,700.75 shares of Series C1
preferred stock.
|
|
CERTAIN
RELATIONSHIPS AND RELATED TRANSACTIONS
AND DIRECTOR INDEPENDENCE
Our board
recognizes that related person transactions present a heightened
risk of conflicts of interest. The audit committee has the
authority to review and approve all related party transactions
involving our directors or executive officers.
Under the policy,
when management becomes aware of a related person transaction,
management reports the transaction to the audit committee and
requests approval or ratification of the transaction. Generally,
the audit committee will approve only related party transactions
that are on terms comparable to those that could be obtained in
arm’s length dealings with an unrelated third person. The
audit committee will report to the full board all related person
transactions presented to it.
Based on the
definition of independence of the NASDAQ Stock Market, the board
has determined that Mr. James and Dr. Imhoff are independent
directors.
John E. Imhoff is
one of our directors. In June 2015, Dr. Imhoff agreed to exchange
certain of his warrants, originally issued in December 2014 and
exercisable for 1 share of our common stock, for two new warrants
that, unlike the original warrant, do not contain any price or
share reset provisions. Each new warrant is exercisable for the
same number of shares of our common stock as the original warrant,
at any time until December 2, 2020. The exercise price of the first
new warrant is $72 per share and the second new warrant is $88 per
share but, aside from the exercise price, the new warrants are
identical in terms to each other. As additional consideration, we
issued Dr. Imhoff an additional 1 share of common stock. Dr. Imhoff
participated on terms equal to those of other holders of the
December 2014 warrants. As a result of these transactions, Dr.
Imhoff’s beneficial ownership of our common stock increased
from approximately 11.7% immediately prior to the exchange, to
approximately 11.8% immediately afterward.
In September 2015,
Dr. Imhoff participated in our Series C preferred stock issuance by
exchanging all of his shares of Series B preferred stock and
investing $300,000 in cash, for a total of 1,067 shares of Series C
preferred stock and warrants to purchase 211 shares of common
stock. Dr. Imhoff participated on terms equal to those of other
Series C investors. As a result of these transactions, Dr.
Imhoff’s beneficial ownership of our common stock increased
from approximately 14% immediately prior to his first acquisition
of shares of Series C preferred stock, to 25% immediately
afterward.
On March 11, 2016,
Dr. Imhoff received 24 shares of common stock as a dividend on his
Series B preferred stock (previously accrued but unpaid), in
accordance with the terms of the Series B preferred
stock.
In April 2016, Dr.
Imhoff exchanged his shares of Series C preferred stock for a total
of 2,400.75 shares of Series C1 preferred stock and 12,804 shares
of common stock. See “Summary—Recent
Developments—Series C Exchanges”. Dr. Imhoff
participated on terms equal to those of other Series C1 investors.
As a result of this transaction, Dr. Imhoff’s beneficial
ownership of our common stock increased from approximately 25%
immediately prior to the transaction, to 77% immediately
afterward.
In June 2016, Dr.
Imhoff agreed to exchange certain of his warrants, exercisable for
4,560 shares of our common stock and subject to certain
anti-dilution provisions, in exchange for new warrants, exercisable
for 9,120 shares of our common stock, but without those
anti-dilution provisions. Dr. Imhoff will be required to surrender
his old warrants upon consummation of our next financing resulting
in net cash proceeds to us of at least $1 million. The new warrants
will have an initial exercise price equal to the exercise price of
the surrendered warrants as of immediately prior to consummation of
the financing, subject to customary “downside price
protection” for as long as our common stock is not listed on
a national securities exchange, and will expire five years from the
date of issuance.
On September 6,
2016, we entered into a royalty agreement with Dr. Imhoff and
another party. Pursuant to the royalty agreement, in exchange for a
payment of $50,000 by Dr. Imhoff and the other party, we granted
them a royalty on future sales of our single-use cervical guides.
The royalty rate was initially $0.10 per disposable, until October
2, 2016, at which point the royalty rate increased to $0.20 per
disposable. Any royalty payments will be split evenly between Dr.
Imhoff and the other party.
Lynne Imhoff (no
relation) currently beneficially owns in excess of 10% of our
outstanding common stock. In September 2015, Ms. Imhoff
participated in our Series C preferred stock issuance by exchanging
all of her shares of Series B preferred stock and investing
$125,000 in cash, for a total of 300 shares of Series C preferred
stock and warrants to purchase 592 shares of common stock. Ms.
Imhoff participated on terms equal to those of other Series C
investors. As a result of these transactions, Ms. Imhoff’s
beneficial ownership of our common stock increased from
approximately 2% immediately prior to her first acquisition of
shares of Series C preferred stock, to 4% immediately
afterward.
In April 2016, Ms.
Imhoff exchanged her shares of Series C preferred stock for a total
of 675 shares of Series C1 preferred stock and 3,600 shares of
common stock. See “Summary—Recent
Developments—Series C Exchanges”. Ms. Imhoff
participated on terms equal to those of other Series C1 investors.
As a result of this transaction, Ms. Imhoff’s beneficial
ownership of our common stock increased from approximately 4%
immediately prior to the transaction, to 45% immediately
afterward.
In June 2016, Ms.
Imhoff agreed to exchange certain of her warrants, exercisable for
912 shares of our common stock and subject to certain anti-dilution
provisions, in exchange for new warrants, exercisable for 1,824
shares of our common stock, but without those anti-dilution
provisions. Ms. Imhoff will be required to surrender her old
warrants upon consummation of our next financing resulting in net
cash proceeds to us of at least $1 million. The new warrants will
have an initial exercise price equal to the exercise price of the
surrendered warrants as of immediately prior to consummation of the
financing, subject to customary “downside price
protection” for as long as our common stock is not listed on
a national securities exchange, and will expire five years from the
date of issuance.
Mark Faupel is one
of our directors and our Chief Operating Officer, and Richard
Blumberg is another one of our directors. Dr. Faupel is a
shareholder of Shenghuo, and Mr. Blumberg, is a managing member of
Shenghuo. We entered into a license agreement with Shenghuo
pursuant to which we granted Shenghuo an exclusive license to
manufacture, sell and distribute our LuViva Advanced Cervical
Cancer device and related disposables in Taiwan, Brunei Darussalam,
Cambodia, Laos, Myanmar, Philippines, Singapore, Thailand, and
Vietnam. Shenghuo has been our exclusive distributor in China,
Macau and Hong Kong, and the license extends to manufacturing in
those countries as well. Pursuant to the license agreement,
Shenghuo had the option to have a designee appointed to our board
of directors.
As partial consideration
for, and as a condition to, the license, and to further align the
strategic interests of the parties, on March 24, 2017 we issued a
$300,000 convertible note to Shenghuo, in exchange for
Shenghuoís June 2016 aggregate cash investment of $200,000.
The unsecured note accrues interest at 20% per year, compounded
annually, from January 1, 2017 until repayment, and is convertible
into shares of our common stock at a conversion price per share of
$13.92, subject to customary anti-dilution adjustment. We also
issued Shenghuo a five-year warrant exercisable immediately for
17,239 shares of common stock at an exercise price equal to the
conversion price of the note, subject to customary anti-dilution
adjustment.
In
September 2015, Dr. Faupel participated in our Series C preferred
stock issuance by investing $100,000 in cash, for a total of 133
shares of Series C preferred stock and warrants to purchase 46
shares of common stock. Dr. Faupel participated on terms equal to
those of other Series C investors. In April 2016, Dr. Faupel
exchanged his shares of Series C preferred stock for a total of 300
shares of Series C1 preferred stock and 1,600 shares of common
stock. See “Summary—Recent Developments—Series C
Exchanges”. Dr. Faupel participated on terms equal to those
of other Series C1 investors.
LEGAL
MATTERS
Jones Day, Atlanta,
Georgia, passed upon the validity of the shares of common stock
that may be offered by this prospectus. Ellenoff Grossman &
Schole LLP, New York, New York, acted as counsel to the placement
agent.
EXPERTS
Our consolidated
financial statements as of December 31, 2
016
and 2015, and
for the years then ended have been audited by UHY LLP, an
independent registered public accounting firm, as set forth in its
report, included in this prospectus. Our financial statements and
the related independent registered public accounting firm report
thereon have been included herein in reliance upon such report
given on the authority of such firm as experts in accounting and
auditing.
WHERE
YOU CAN GET MORE INFORMATION
We have filed with
the SEC under the Securities Act a registration statement on Form
S-1 of which this prospectus forms a part. This prospectus does not
contain all of the information contained in the registration
statement and its exhibits. We strongly encourage you to read
carefully the registration statement and its exhibits.
Any statement made
in this prospectus concerning the contents of any contract,
agreement or other document is only a summary of the actual
contract, agreement or other document. If we have filed any
contract, agreement or other document as an exhibit to the
registration statement, you should read the exhibit for a more
complete understanding of the document or matter
involved.
We file annual,
quarterly and current reports; proxy statements and other
information with the SEC. You may read and copy any of this
information at the SEC’s Public Reference Room at 100 F
Street, N.E., Washington, D.C. 20549. Please call the SEC at
1-800-SEC-0330 for information on the operation of the Public
Reference Room. The SEC also maintains an Internet website that
contains reports, proxy statements and other information regarding
issuers, including us, who file electronically with the SEC. The
address of that site is http://www.sec.gov. The information
contained on the SEC’s website is expressly not incorporated
by reference into this prospectus.
INDEX
TO FINANCIAL STATEMENTS
Annual Consolidated Financial
Statements
|
Report of
Independent Registered Public Accounting Firm
|
F-2
|
|
Consolidated
Balance Sheets as of December 31, 2016 and 2015
|
F-3
|
|
Consolidated
Statements of Operations for the Years Ended December 31, 2016 and
2015
|
F-4
|
|
Consolidated
Statements of Stockholdersí Deficit for the Years Ended
December 31, 2016 and 2015
|
F-5
|
|
Consolidated
Statements of Cash Flows for the Years Ended December 31, 2016 and
2015
|
F-6
|
|
Notes to
Consolidated Financial Statements
|
F-8
|
|
|
|
Report
of Independent Registered Public Accounting
Firm
To
the Board of Directors and
Stockholders
of Guided Therapeutics, Inc.
We have audited the
accompanying consolidated balance sheets of Guided Therapeutics,
Inc. and Subsidiary (the “Company”) as of December 31,
2016 and 2015, and the related consolidated statements of
operations, stockholdersí deficit, and cash flows for the
years then ended. The Companyís management is responsible for
these consolidated financial statements. Our responsibility is to
express an opinion on these consolidated financial statements based
on our audits.
We conducted our
audits in accordance with the standards of the Public Company
Accounting Oversight Board (United States). Those standards require
that we plan and perform the audit to obtain reasonable assurance
about whether the consolidated financial statements are free of
material misstatement. The Company is not required to have, nor
were we engaged to perform, an audit of its internal control over
financial reporting. Our audit included consideration of internal
control over financial reporting as a basis for designing audit
procedures that are appropriate in the circumstances, but not for
the purpose of expressing an opinion on the effectiveness of the
Companyís internal control over financial reporting.
Accordingly, we express no such opinion. An audit also includes
examining, on a test basis, evidence supporting the amounts and
disclosures in the consolidated financial statements, assessing the
accounting principles used and significant estimates made by
management, as well as evaluating the overall consolidated
financial statement presentation. We believe that our audits
provide a reasonable basis for our opinion.
In our opinion, the
consolidated financial statements referred to above present fairly,
in all material respects, the financial position of Guided
Therapeutics, Inc. and Subsidiary as of December 31, 2016 and 2015,
and the results of their operations and their cash flows for the
years then ended in conformity with accounting principles generally
accepted in the United States of America.
The accompanying
consolidated financial statements have been prepared assuming that
the Company will continue as a going concern. As discussed in Note
1 to the consolidated financial statements, the Companyís
significant operating losses raise substantial doubt about its
ability to continue as a going concern. Managementís plans
regarding those matters are also discussed in Note 1 to the
consolidated financial statements. The consolidated financial
statements do not include any adjustments that might result from
the outcome of this uncertainty.
/s/ UHY LLP
UHY
LLP
Sterling
Heights, Michigan
March
21, 2017
|
GUIDED
THERAPEUTICS, INC. AND SUBSIDIARY
|
|
CONDENSED
CONSOLIDATED BALANCE SHEETS (in thousands)
|
|
AS
OF DECEMBER 31,
|
|
|
|
|
|
ASSETS
|
|
|
|
CURRENT
ASSETS:
|
|
|
|
Cash
and cash equivalents
|
$
14
|
$
35
|
|
Accounts
receivable, net of allowance for doubtful accounts of $279 and $95
at December 31, 2016 and 2015, respectively
|
-
|
190
|
|
Inventory,
net of reserves of $278 and $118 at December 31, 2016 and 2015,
respectively
|
773
|
1,119
|
|
Other
current assets
|
259
|
780
|
|
Total
current assets
|
1,046
|
2,124
|
|
|
|
|
|
Property
and equipment, net
|
126
|
318
|
|
Other
assets
|
320
|
73
|
|
Total
noncurrent assets
|
446
|
391
|
|
|
|
|
|
TOTAL
ASSETS
|
1,492
|
2,515
|
|
|
|
|
|
LIABILITIES
AND STOCKHOLDERS’ DEFICIT
|
|
|
|
CURRENT
LIABILITIES:
|
|
|
|
Notes
payable in default, including related parties
|
1,008
|
133
|
|
Short-term
note payable
|
197
|
704
|
|
Convertible
note in default
|
2,361
|
-
|
|
Short-term
convertible notes payable, net
|
468
|
686
|
|
Accounts
payable
|
2,600
|
1,824
|
|
Accrued
liabilities
|
2,670
|
1,907
|
|
Deferred
revenue
|
34
|
217
|
|
Total
current liabilities
|
9,338
|
5,471
|
|
|
|
|
|
Warrants,
at fair value
|
1,420
|
2,606
|
|
|
|
|
|
TOTAL
LIABILITIES
|
10,758
|
8,077
|
|
|
|
|
|
COMMITMENTS & CONTINGENCIES (Note 8)
|
|
|
|
|
|
|
|
STOCKHOLDERS’
DEFICIT:
|
|
|
|
Series
C convertible preferred stock, $.001 par value; 9.0 shares
authorized, 1.6 and 5.6 shares issued and outstanding as of
December 31, 2016 and 2015, respectively. (Liquidation preference
of $1,643 and $5,555 at December 31, 2016 and 2015,
respectively).
|
601
|
2,052
|
|
Series
C1 convertible preferred stock, $.001 par value; 20.3 shares
authorized, 4.3 shares and none issued and outstanding as of
December 31, 2016 and 2015, respectively. (Liquidation preference
of $4,312 and none at December 31, 2016 and 2015,
respectively).
|
701
|
-
|
|
Common
stock, $.001 par value; 1,000,000 shares authorized, 669 and 3
shares issued and outstanding as of December 31, 2016 and 2015,
respectively
|
742
|
236
|
|
Additional
paid-in capital
|
116,380
|
114,845
|
|
Treasury
stock, at cost
|
(132
)
|
(132
)
|
|
Accumulated
deficit
|
(127,558
)
|
(122,563
)
|
|
|
|
|
|
TOTAL
STOCKHOLDERS’ DEFICIT
|
(9,266
)
|
(5,562
)
|
|
|
|
|
|
TOTAL
LIABILITIES AND STOCKHOLDERS’ DEFICIT
|
$
1,492
|
$
2,515
|
The accompanying
notes are an integral part of these consolidated
statements.
|
GUIDED
THERAPEUTICS, INC. AND SUBSIDIARY
|
|
CONSOLIDATED
STATEMENTS OF OPERATIONS (in thousands)
|
|
FOR
THE YEARS ENDED DECEMBER 31,
|
|
|
|
|
|
|
|
|
|
REVENUE:
|
|
|
|
Sales
– devices and disposables, net
|
$
605
|
$
564
|
|
Cost
of goods sold
|
493
|
537
|
|
Gross
profit
|
112
|
27
|
|
|
|
|
|
Contract
and grant revenue
|
-
|
42
|
|
|
|
|
|
OPERATING
EXPENSES:
|
|
|
|
|
|
|
|
Research
and development
|
733
|
1,477
|
|
Sales
and marketing
|
393
|
718
|
|
General
and administrative
|
2,806
|
4,101
|
|
Total
operating expenses
|
3,932
|
6,296
|
|
|
|
|
|
Operating
loss
|
(3,820
)
|
(6,227
)
|
|
|
|
|
|
OTHER
INCOME (EXPENSES):
|
|
|
|
Other
income
|
68
|
74
|
|
Interest
expense
|
(1,895
)
|
(1,317
)
|
|
Change
in fair value of warrants
|
1,677
|
568
|
|
Total
other income (expenses)
|
(150
)
|
(675
)
|
|
|
|
|
|
LOSS
FROM OPERATIONS
|
(3,970
)
|
(6,902
)
|
|
|
|
|
|
PROVISION
FOR INCOME TAXES
|
-
|
-
|
|
|
|
|
|
NET
LOSS
|
(3,970
)
|
(6,902
)
|
|
|
|
|
|
DEEMED
DIVIDENDS
|
-
|
(1,263
)
|
|
|
|
|
|
PREFERRED
STOCK DIVIDENDS
|
(1,025
)
|
(1,338
)
|
|
|
|
|
|
NET
LOSS ATTRIBUTABLE TO COMMON STOCKHOLDERS
|
$
(4,995
)
|
$
(9,503
)
|
|
|
|
|
|
BASIC
AND DILUTED NET LOSS PER SHARE ATTRIBUTABLE
TO
COMMON STOCKHOLDERS
|
$
(24.62
)
|
$
(5,939.38
)
|
|
|
|
|
|
WEIGHTED
AVERAGE SHARES OUTSTANDING
|
203
|
2
|
|
The accompanying
notes are an integral part of these consolidated
statements.
|
|
|
|
|
GUIDED
THERAPEUTICS, INC. AND SUBSIDIARY
CONSOLIDATED
STATEMENTS OF STOCKHOLDERS’ DEFICIT
FOR
THE YEARS ENDED DECEMBER 31, 2016 AND 2015 (In
Thousands)
|
|
|
Preferred Stock
Series
C1
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
BALANCE, January 1,
2015
|
-
|
$
-
|
-
|
$
- -
|
1
|
$
97
|
$
107,952
|
$
(132
)
|
$
(113,060
)
|
$
(4,465
)
|
|
Preferred
dividends
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
(352
)
|
(352
)
|
|
Conversion of
Series C preferred stock to common stock
|
(2
)
|
(840
)
|
-
|
-
|
1
|
99
|
1,727
|
-
|
(986
)
|
-
|
|
Issuance of common
stock and warrants
|
-
|
-
|
-
|
-
|
-
|
11
|
1,327
|
-
|
-
|
1,338
|
|
Exercise of
warrants and options for common stock
|
-
|
-
|
-
|
-
|
-
|
11
|
132
|
-
|
-
|
143
|
|
Conversion of debt
into common stock
|
-
|
-
|
-
|
-
|
-
|
15
|
999
|
-
|
-
|
1,014
|
|
December 2014
public offering warrants exchange and common shares
issuance
|
-
|
-
|
-
|
-
|
-
|
3
|
1,368
|
-
|
(1,049
)
|
322
|
|
Series B, Tranche
A, warrant price adjustment
|
-
|
-
|
-
|
-
|
-
|
-
|
64
|
-
|
(64
)
|
-
|
|
Series B preferred
stock exchange
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
(1
)
|
(678
)
|
|
Series C preferred
stock and warrant issuance
|
8
|
2,892
|
-
|
-
|
-
|
-
|
268
|
-
|
(150
)
|
3,010
|
|
Stock-based
compensation
|
-
|
-
|
-
|
-
|
-
|
-
|
1,008
|
-
|
-
|
1,008
|
|
Net
Loss
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
(6,902
)
|
(6,902
)
|
|
BALANCE, December
31, 2015
|
6
|
$
2,052
|
-
|
$
-
|
3
|
$
236
|
$
114,845
|
$
(132
)
|
$
(122,563
)
|
$
(5,562
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Preferred
dividends
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
(191
)
|
(191
)
|
|
Issuance of common
stock from accrued dividends
|
-
|
-
|
-
|
-
|
-
|
-
|
54
|
-
|
-
|
54
|
|
Conversion of
Series C preferred stock to common stock
|
(2
)
|
(750
)
|
-
|
-
|
531
|
456
|
1,128
|
-
|
(834
)
|
-
|
|
Conversion of debt
into common stock
|
-
|
-
|
-
|
-
|
53
|
20
|
238
|
-
|
-
|
258
|
|
Issuance of common
stock due to Series B, Tranche B warrants exchanged for shares and
rights to shares
|
-
|
-
|
-
|
-
|
19
|
12
|
(12
)
|
-
|
-
|
-
|
|
Series C preferred
stock exchanged for Series C1 preferred stock
|
(2
)
|
(751
)
|
4
|
701
|
23
|
18
|
(18
)
|
-
|
-
|
-
|
|
Issuance of common
stock for cash
|
-
|
-
|
-
|
-
|
40
|
-
|
50
|
-
|
-
|
50
|
|
Stock-based
compensation
|
|
|
|
|
-
|
-
|
95
|
-
|
-
|
95
|
|
Net
Loss
|
|
|
|
|
-
|
-
|
-
|
-
|
(3,970
)
|
(3,970
)
|
|
BALANCE,
December 31, 2016
|
2
|
$
601
|
4
|
$
701
|
669
|
$
742
|
$
116,380
|
$
(132
)
|
$
(127,558
)
|
$
(9,266
)
|
The accompanying notes are an
integral part of these consolidated
statements.
|
GUIDED
THERAPEUTICS, INC. AND SUBSIDIARY
|
|
CONSOLIDATED
STATEMENTS OF CASH FLOWS
|
|
FOR
THE YEARS ENDED DECEMBER 31,
|
|
(In
Thousands)
|
|
|
|
|
|
|
|
CASH
FLOWS FROM OPERATING ACTIVITIES:
|
|
|
|
Net
loss
|
$
(3,970
)
|
$
(6,902
)
|
|
Adjustments
to reconcile net loss to net cash used in operating
activities:
|
|
|
|
Bad
debt expense
|
221
|
19
|
|
Depreciation
and Amortization
|
1,223
|
1,054
|
|
Stock-based
compensation
|
95
|
1,008
|
|
Non-employee
stock based compensation
|
-
|
400
|
|
Change
in fair value of warrants
|
(1,677
)
|
(568
)
|
|
Changes
in operating assets and liabilities:
|
|
|
|
Accounts
receivable
|
(31
)
|
61
|
|
Inventory
|
345
|
129
|
|
Other
current assets
|
519
|
(681
)
|
|
Other
assets
|
(247
)
|
28
|
|
Accounts
payable
|
775
|
91
|
|
Deferred
revenue
|
(183
)
|
193
|
|
Accrued
liabilities
|
1,128
|
1,125
|
|
Total
adjustments
|
2,168
|
2,859
|
|
|
|
|
|
Net
cash used in operating activities
|
(1,802
)
|
(4,043
)
|
|
|
|
|
|
CASH
FLOWS FROM INVESTING ACTIVITIES:
|
|
|
|
Additions
to fixed assets
|
-
|
(8
)
|
|
|
|
|
|
Net
cash used in investing activities
|
-
|
(8
)
|
|
|
|
|
|
CASH
FLOWS FROM FINANCING ACTIVITIES:
|
|
|
|
Net
proceeds from issuance of preferred stock and warrants,
net
|
-
|
3,698
|
|
Net
proceed from issuance of common stock and
warrants
|
50
|
720
|
|
Proceeds
from debt financing, net of discount and debt issuance
costs
|
1,958
|
377
|
|
Payments
on notes
|
(227
)
|
(1,014
)
|
|
Proceeds
from options and warrants exercised
|
-
|
143
|
|
|
|
|
|
Net
cash provided by financing activities
|
1,781
|
3,924
|
|
|
|
|
|
NET
CHANGE IN CASH AND CASH EQUIVALENTS
|
(21
)
|
(127
)
|
|
|
|
|
|
CASH
AND CASH EQUIVALENTS, beginning of year
|
35
|
162
|
|
|
|
|
|
CASH
AND CASH EQUIVALENTS, end of year
|
$
14
|
$
35
|
|
|
|
|
|
SUPPLEMENTAL
SCHEDULE OF:
|
|
|
|
Cash paid
for:
|
|
|
|
Interest
|
$
-
|
$
76
|
|
NONCASH
INVESTING AND FINANCING ACTIVITIES:
|
|
|
|
Issuance
of common stock as debt repayment
|
$
258
|
$
1,014
|
|
Dividends
on preferred stock
|
$
1,025
|
$
1,338
|
|
Deemed
dividend on December 2014 public offering
warrants
|
$
-
|
$
1,049
|
|
Term
changes on Series B preferred stock and December 2014 public
offering warrants resulting in transfer to
equity
|
$
-
|
$
324
|
|
Repayment
of deferred compensation via issuance of preferred
stock
|
$
-
|
$
100
|
|
Deemed
dividend on beneficial conversion features of Series C Preferred
stock
|
$
-
|
$
150
|
|
Deemed
dividend on price changes for Series B preferred stock
warrants
|
$
-
|
$
64
|
The accompanying
notes are an integral part of these consolidated
statements.
|
GUIDED
THERAPEUTICS, INC. AND SUBSIDIARY
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31,
2016 AND 2015
1.
ORGANIZATION, BACKGROUND, AND BASIS OF
PRESENTATION
Guided
Therapeutics, Inc. (formerly SpectRx, Inc.), together with its
wholly owned subsidiary, InterScan, Inc. (formerly Guided
Therapeutics, Inc.), collectively referred to herein as the
“Company”, is
a medical
technology company focused on developing innovative medical devices
that have the potential to improve healthcare. The Company’s
primary focus is the continued commercialization of its LuViva
non-invasive cervical cancer detection device and extension of its
cancer detection technology into other cancers, including
esophageal. The Company’s technology, including products in
research and development, primarily relates to biophotonics
technology for the non-invasive detection of
cancers.
Basis
of Presentation
All information and
footnote disclosures included in the consolidated financial
statements have been prepared in accordance with accounting
principles generally accepted in the United
States.
A 1:800 reverse
stock split of all of the Company’s issued and outstanding
common stock was implemented on November 7, 2016. As a result of
the reverse stock split, every 800 shares of issued and outstanding
common stock was converted into 1 share of common stock. All
fractional shares created by the reverse stock split were rounded
to the nearest whole share. The number of authorized shares of
common stock did not change. The reverse stock split decreased the
Company’s issued and outstanding shares of common stock from
453,694,400 shares of Common Stock to 570,707 shares as of that
date. See Note 4, Stockholders’ Deficit. Unless otherwise
specified, all per share amounts are reported on a post-stock split
basis, as of December 31, 2016. On February 24, 2016, the Company
had also implemented a 1:100 reverse stock split of its issued and
outstanding common stock.
The Company’s
prospects must be considered in light of the substantial risks,
expenses and difficulties encountered by entrants into the medical
device industry. This industry is characterized by an increasing
number of participants, intense competition and a high failure
rate. The Company has experienced net losses since its inception
and, as of December 31, 2016, it had an accumulated deficit of
approximately $127.6 million. Through December 31, 2016, the
Company has devoted substantial resources to research and
development efforts. The Company first generated revenue from
product sales in 1998, but does not have significant experience in
manufacturing, marketing or selling its products. The
Company’s development efforts may not result in commercially
viable products and it may not be successful in growing sales for
its products. Moreover, required regulatory clearances or approvals
may not be obtained. The Company’s products may not ever gain
market acceptance and the Company may not ever achieve levels of
revenue to sustain further development costs and support ongoing
operations or achieve profitability. The development and continued
commercialization of the Company’s products will require
substantial development, regulatory, sales and marketing,
manufacturing and other expenditures. The Company expects operating
losses to continue through the foreseeable future as it continues
to expend substantial resources to complete development of its
products, obtain regulatory clearances or approvals and conduct
further research and development.
Going
Concern
The Company’s
consolidated financial statements have been prepared and presented
on a basis assuming it will continue as a going concern. The
factors below raise substantial doubt about the Company’s
ability to continue as a going concern. The financial statements do
not include any adjustments that might be necessary from the
outcome of this uncertainty.
At December 31,
2016, the Company had a negative working capital of approximately
$8.3 million, accumulated deficit of $127.6 million, and incurred a
net loss of $4.0 million for the year then ended.
Stockholders’ deficit totaled approximately $9.3 million at
December 31, 2016, primarily due to recurring net losses from
operations, deemed dividends on warrants and preferred stock,
offset by proceeds from the exercise of options and warrants and
proceeds from sales of stock.
The Company’s
capital-raising efforts are ongoing. If sufficient capital cannot
be raised during the second quarter of 2017, the Company will
continue its plans of curtailing operations by reducing
discretionary spending and staffing levels, and attempting to
operate by only pursuing activities for which it has external
financial support. However, there can be no assurance that such
external financial support will be sufficient to maintain even
limited operations or that the Company will be able to raise
additional funds on acceptable terms, or at all. In such a case,
the Company might be required to enter into unfavorable agreements
or, if that is not possible, be unable to continue operations, and
to the extent practicable, liquidate and/or file for bankruptcy
protection.
The Company had
warrants exercisable for approximately 4.3 million shares of its
common stock outstanding at December 31, 2016, with exercise prices
ranging between $0.35 and $84,000 per share. Exercises of these
warrants would generate a total of approximately $6.2 million in
cash, assuming full exercise, although the Company cannot be
assured that holders will exercise any warrants. Management may
obtain additional funds through the public or private sale of debt
or equity, and grants, if available.
2.
SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Use
of Estimates
The preparation of
financial statements in conformity with accounting principles
generally accepted in the United States requires management to make
estimates and assumptions that affect the reported amounts of
assets and liabilities and disclosure of contingent assets and
liabilities at the date of the financial statements and the
reported amounts of revenues and expenses during the reporting
period. Actual results could differ from those estimates.
Significant areas where estimates are used include the allowance
for doubtful accounts, inventory valuation and input variables for
Black-Scholes, Monte Carlo simulations and binomial
calculations.
Principles
of Consolidation
The accompanying
consolidated financial statements include the accounts of Guided
Therapeutics, Inc. and its wholly owned
subsidiary.
Accounting
Standard Updates
In May 2014, the
Financial Accounting Standards Board (“FASB”) issued
ASU 2014-09, “Revenue from Contracts with Customers (Topic
606),” (“ASU 2014-09”). ASU 2014-09 outlines a
new, single comprehensive model for entities to use in accounting
for revenue arising from contracts with customers and supersedes
most current revenue recognition guidance, including
industry-specific guidance. This new revenue recognition model
provides a five-step analysis in determining when and how revenue
is recognized. The new model requires revenue recognition to depict
the transfer of promised goods or services to customers in an
amount that reflects the consideration a company expects to
receive. ASU 2014-09 also requires additional disclosure about the
nature, amount, timing and uncertainty of revenue and cash flows
arising from customer contracts, including significant judgments
and changes in judgments and assets recognized from costs incurred
to obtain or fulfill a contract. In August 2015, the FASB issued
ASU 2015-14, “Deferral of the Effective Date”, which
amends ASU 2014-09. As a result, the effective date will be the
first quarter of fiscal year 2018 with early adoption permitted in
the first quarter of fiscal year 2017. Subsequently, the FASB has
issued the following standards related to ASU 2014-09: ASU 2016-08,
“Revenue from Contracts with Customers (Topic 606), Principal
versus Agent Considerations (Reporting Revenue Gross versus
Net),” (“ASU 2016-08”); ASU 2016-10,
“Revenue from Contracts with Customers (Topic 606),
Identifying Performance Obligations and Licensing,”
(“ASU 2016-10”); ASU 2016-12, “Revenue from
Contracts with Customers (Topic 606) Narrow-Scope Improvements and
Practical Expedients,” (“ASU 2016-12”); and ASU
2016-20, “Technical Corrections and Improvements to Topic
606, Revenue from Contracts with Customers,” (“ASU
2016-20”), which are intended to provide additional guidance
and clarity to ASU 2014-09. The Company must adopt ASU 2016-08, ASU
2016-10, ASU 2016-12 and ASU 2016-20 along with ASU 2014-09
(collectively, the “New Revenue Standards”). The New
Revenue Standards may be applied using one of two retrospective
application methods: (1) a full retrospective approach for all
periods presented, or (2) a modified retrospective approach that
presents a cumulative effect as of the adoption date and additional
required disclosures. The Company is evaluating the impact that
adoption of this guidance will have on the determination or
reporting of its financial results.
In June 2014, the
FASB issued ASU 2014-12, “Accounting for Share-Based Payments
When the Terms of an Award Provide that a Performance Target Could
be Achieved after the Requisite Service Period,” (“ASU
2014-12”). ASU 2014-12 requires that a performance target
that affects vesting, and that could be achieved after the
requisite service period, be treated as a performance condition. As
such, the performance target should not be reflected in estimating
the grant date fair value of the award. ASU 2014-12 is effective
for the reporting periods beginning after December 15, 2015. Early
adoption is permitted. The effective date of this policy was the
first quarter of fiscal year 2016.
In August 2014, the
FASB issued ASU 2014-15, “Disclosure of Uncertainties about
an Entity’s Ability to Continue as a Going Concern,”
(“ASU 2014-15”). ASU 2014-15 requires management to
perform interim and annual assessments of an entity’s ability
to continue as a going concern for a one year period subsequent to
the date of issuance of its financial statements. An entity must
provide certain disclosures if conditions or events raise
substantial doubt about the entity’s ability to continue as a
going concern. The Company is evaluating the impact that adoption
of this guidance will have on the determination or reporting of its
financial results.
In July 2015, the
FASB issued ASU 2015-11, “Simplifying the Measurement of
Inventory,” (“ASU 2015-11”). ASU 2015-11 requires
inventory be measured at the lower of cost and net realizable value
and options that currently exist for market value be eliminated.
ASU 2015-11 defines net realizable value as estimated selling
prices in the ordinary course of business, less reasonably
predictable costs of completion, disposal, and transportation. The
guidance is effective for reporting periods beginning after
December 15, 2016 and interim periods within those fiscal
years with early adoption permitted. ASU 2015-11 should be applied
prospectively. The Company is evaluating the impact adoption of
this guidance will have on determination or reporting of its
financial results.
In April 2015, the
FASB issued ASU 2015-03, “Imputation of Interest: Simplifying
the Presentation of Debt Issuance Costs,” (“ASU
2015-03”). ASU 2015-03 requires that debt issuance costs
related to a recognized debt liability be presented in the balance
sheet as a direct deduction from the carrying amount of that debt
liability, consistent with debt discounts. The guidance is
effective for reporting periods beginning after December 15,
2015 and interim periods within those fiscal years with early
adoption permitted. ASU 2015-03 should be applied on a
retrospective basis, wherein the balance sheet of each period
presented should be adjusted to reflect the effects of adoption.
The effective date of this policy was the first quarter of fiscal
year 2016.
In November 2015,
the FASB issued ASU 2015-17, “Income Taxes (Topic 740)
Balance Sheet Classification of Deferred Taxes.” The
amendments in ASU 2015-17 seek to simplify the presentation of
deferred income taxes and require that deferred tax liabilities and
assets be classified as noncurrent in a classified statement of
financial position. ASU 2015-17 is effective for financial
statements issued for annual periods beginning after December 15,
2016, and interim periods within those annual periods, with early
application permitted for all entities as of the beginning of an
interim or annual reporting period. The Company believes that
adoption of this new standard will not be material to its
consolidated financial statements.
In February 2016,
the FASB issued ASU 2016-02, “Leases (Topic 842)” that
requires lessees to recognize on the balance sheet the assets and
liabilities associated with the rights and obligations created by
those leases. Under the new guidance, a lessee will be required to
recognize assets and liabilities for leases with lease terms of
more than 12 months. Consistent with current U.S. GAAP, the
recognition, measurement, and presentation of expenses and cash
flows arising from a lease by a lessee primarily will depend on its
classification as finance or operating lease. The update is
effective for reporting periods beginning after December 15, 2018.
Early adoption is permitted. The Company is evaluating the impact
adoption of this guidance will have on determination or reporting
of its financial results.
In March 2016, the
FASB issued ASU 2016-05, “Derivatives and Hedging (Topic
815),” (“ASU 2016-05”). ASU 2016-05 provides
guidance clarifying that novation of a derivative contract (i.e., a
change in counterparty) in a hedge accounting relationship does
not, in and of itself, require dedesignation of that hedge
accounting relationship. The effective date will be the first
quarter of fiscal year 2017, with early adoption permitted.
Adoption is not expected to have a material effect on the
Company’s consolidated financial
statements.
In March 2016, the
FASB issued ASU 2016-06, “Derivatives and Hedging (Topic
815),” (“ASU 2016-06”). ASU 2016-06 simplifies
the embedded derivative analysis for debt instruments containing
contingent call or put options by clarifying that an exercise
contingency does not need to be evaluated to determine whether it
relates to interest rates and credit risk in an embedded derivative
analysis. The effective date will be the first quarter of fiscal
year 2017, with early adoption permitted. Adoption is not expected
to have a material effect on the Company’s consolidated
financial statements.
In March 2016, the
FASB issued ASU 2016-09, “Compensation-Stock Compensation
(Topic 718), Improvements to Employee Share-Based Payment
Accounting,” (“ASU 2016-09”). ASU 2016-09 is
intended to simplify several aspects related to how share-based
payments are accounted for and presented in the financial
statements, such as requiring all income tax effects of awards to
be recognized in the income statement when the awards vest or are
settled and allowing a policy election to account for forfeitures
as they occur. In addition, all related cash flows resulting from
share-based payments will be reported as operating activities on
the statement of cash flows. ASU 2016-09 could result in increased
volatility of the Company’s provision for income taxes and
earnings per share, depending on the Company’s share price at
exercise or vesting of share-based awards compared to grant date.
The effective date will be the first quarter of fiscal year 2017,
with early adoption permitted. The Company believes that adoption
of this new standard will not be material to its consolidated
financial statements; however, the impact on future effective tax
rates could be significant.
In June 2016, the
FASB issued ASU 2016-13, “Financial Instruments - Credit
Losses,” (“ASU 2016-13”). ASU 2016-13 sets forth
a “current expected credit loss” model which requires
the Company to measure all expected credit losses for financial
instruments held at the reporting date based on historical
experience, current conditions and reasonable supportable
forecasts. The guidance in this new standard replaces the existing
incurred loss model and is applicable to the measurement of credit
losses on financial assets measured at amortized cost and applies
to some off-balance sheet credit exposures. The effective date will
be the first quarter of fiscal year 2020. The Company is evaluating
the impact that adoption of this new standard will have on its
consolidated financial statements.
In August 2016, the
FASB issued ASU 2016-15, “Statement of Cash Flows (Topic
230), Classification of Certain Cash Receipts and Cash
Payments,” (“ASU 2016-15”). ASU 2016-15 reduces
the existing diversity in practice in financial reporting by
clarifying existing principles in ASC 230, “Statement of Cash
Flows,” and provides specific guidance on certain cash flow
classification issues. The effective date for ASU 2016-15 will be
the first quarter of fiscal year 2018, with early adoption
permitted. The Company is evaluating the impact adoption of this
guidance will have on determination or reporting of its financial
results.
In November 2016,
the FASB issued ASU 2016-18, “Statement of Cash Flows (Topic
230) - Restricted Cash,” (“ASU 2016-18”). ASU
2016-18 requires a statement of cash flows to explain the change
during the period in the total of cash, cash equivalents, and
amounts generally described as restricted cash or restricted cash
equivalents. Amounts generally described as restricted cash and
restricted cash equivalents should be included with cash and cash
equivalents when reconciling the beginning-of-period and
end-of-period total amounts shown on the statement of cash flows.
The Company is evaluating the impact adoption of this guidance will
have on determination or reporting of its financial
results.
In January 2017,
the FASB issued ASU 2017-04, “Intangibles - Goodwill and
Other (Topic 350): Simplifying the Test for Goodwill
Impairment,” (“ASU 2017-04”). ASU 2017-04
eliminates Step 2 from the goodwill impairment test. Instead, an
entity should perform its annual, or interim, goodwill impairment
test by comparing the fair value of a reporting unit with its
carrying amount. An entity should recognize an impairment charge
for the amount by which the carrying amount exceeds the reporting
unit’s fair value, if any. The loss recognized should not
exceed the total amount of goodwill allocated to that reporting
unit. Additionally, an entity should consider income tax effects
from any tax deductible goodwill on the carrying amount of the
reporting unit when measuring the goodwill impairment. The
effective date will be the first quarter of fiscal year 2020, with
early adoption permitted in 2017. Adoption is not expected to have
a material effect on the Company’s consolidated financial
statements.
Except as noted
above, the guidance issued by the FASB during the current year is
not expected to have a material effect on the Company’s
consolidated financial statements.
Cash
Equivalents
The Company
considers all highly liquid investments with an original maturity
of three months or less when purchased to be a cash
equivalent.
Accounts
Receivable
The Company
performs periodic credit evaluations of its customers’
financial conditions and generally does not require collateral. The
Company reviews all outstanding accounts receivable for
collectability on a quarterly basis. An allowance for doubtful
accounts is recorded for any amounts deemed uncollectable. The
Company does not accrue interest receivable on past due accounts
receivable.
Concentrations
of Credit Risk
The Company, from
time to time during the years covered by these consolidated
financial statements, may have bank balances in excess of its
insured limits. Management has deemed this a normal business
risk.
The Company
performs periodic credit evaluations of its customers’
financial conditions and generally does not require collateral. The
Company reviews all outstanding accounts receivable for
collectability on a quarterly basis. An allowance for doubtful
accounts is recorded for any amounts deemed uncollectable. The
Company does not accrue interest receivable on past due accounts
receivable.
Inventory
Valuation
All inventories are
stated at lower of cost or market, with cost determined
substantially on a “first-in, first-out” basis.
Selling, general, and administrative expenses are not inventoried,
but are charged to expense when purchased. At December 31, 2016 and
December 31, 2015, our inventories were as follows (in
thousands):
|
|
|
|
|
|
|
|
Raw
materials
|
$
795
|
$
686
|
|
Work in
process
|
115
|
186
|
|
Finished
goods
|
141
|
365
|
|
Inventory
reserve
|
(278
)
|
(118
)
|
|
Total
|
$
773
|
$
1,119
|
Property
and Equipment
Property and
equipment are recorded at cost. Depreciation is computed using the
straight-line method over estimated useful lives of three to seven
years. Leasehold improvements are depreciated at the shorter of the
useful life of the asset or the remaining lease term. Depreciation
expense is included in general and administrative expense on the
statement of operations. Expenditures for repairs and maintenance
are expensed as incurred. Property and equipment are summarized as
follows at December 31, 2016 and 2015 (in
thousands):
|
|
|
|
|
|
|
|
Equipment
|
$
1,378
|
$
1,377
|
|
Software
|
740
|
740
|
|
Furniture and
fixtures
|
124
|
124
|
|
Leasehold
Improvement
|
199
|
199
|
|
|
2,441
|
2,440
|
|
Less accumulated
depreciation
|
(2,315
)
|
(2,122
)
|
|
Total
|
$
126
|
$
318
|
Debt
Issuance Costs
Debt issuance costs
are capitalized as described in ASU 2015-03 and adopted
retrospectively.
Other
Assets
Other assets
primarily consist of short, and long-term deposits for various
tooling inventory that are being constructed for the Company and
deferred financing costs.
Patent
Costs (Principally Legal Fees)
Costs incurred in
filing, prosecuting, and maintaining patents are recurring, and
expensed as incurred. Maintaining patents are expensed as incurred
as the Company has not yet received FDA approval and recovery of
these costs is uncertain. Such costs aggregated approximately
$23,000 and $47,000 in 2016 and 2015,
respectively.
Accrued
Liabilities
Accrued liabilities
are summarized as follows at December 31, 2016 and 2015 (in
thousands):
|
|
|
|
|
|
|
|
Accrued
compensation
|
$
1,656
|
$
1,235
|
|
Accrued
professional fees
|
161
|
154
|
|
Deferred
rent
|
13
|
36
|
|
Accrued
warranty
|
58
|
82
|
|
Accrued
vacation
|
175
|
177
|
|
Accrued
dividends
|
296
|
167
|
|
Other accrued
expenses
|
311
|
56
|
|
Total
|
$
2,670
|
$
1,907
|
Revenue
Recognition
Revenue from the
sale of the Company’s products is recognized upon shipment of
such products to its customers. The Company recognizes revenue from
contracts on a straight-line basis, over the terms of the
contracts. The Company recognizes revenue from grants based on the
grant agreements, at the time the expenses are
incurred.
Significant
Customers
In 2016 and 2015,
the majority of the Company’s revenues were from three and
four customers, respectively. Revenue from these customers totaled
approximately $534,000 or 73% and approximately $280,000 or 73% of
gross revenue for the year ended December 31, 2016 and 2015,
respectively. Accounts receivable due from those customers
represents 43% and 62% of unreserved accounts receivable as of
December 31, 2016 and 2015, respectively.
Deferred
Revenue
The Company defers
payments received as revenue until earned based on the related
contracts on a straight line basis, over the terms of the
contract.
Research
and Development
Research and
development expenses consist of expenditures for research conducted
by the Company and payments made under contracts with consultants
or other outside parties and costs associated with internal and
contracted clinical trials. All research and development costs are
expensed as incurred.
Income
Taxes
The Company uses
the liability method of accounting for income taxes. Under this
method, deferred tax assets and liabilities are determined based on
differences between the financial reporting and tax bases of assets
and liabilities and are measured using the enacted tax rates and
laws that will be in effect when the differences are expected to
reverse. Management provides valuation allowances against the
deferred tax assets for amounts that are not considered more likely
than not to be realized.
The Company is
current with its federal and applicable state tax returns filings.
Although we have been experiencing recurring losses, its is
obligated to file tax returns for compliance with Internal Revenue
Service (“IRS”) regulations and that of applicable
state jurisdictions. At December 31, 2016, the Company has
approximately $33 million of net operating loss as compared to $28
million for the same period in 2015. This net operating loss will
be eligible to be carried forward for tax purposes at federal and
applicable states level. A full valuation allowance has been
recorded related the deferred tax assets generated from the net
operating losses.
None of the
Company’s federal or state income tax returns are currently
under examination by the IRS or state
authorities.
Uncertain
Tax Positions
The Company
assesses each income tax position is assessed using a two-step
process. A determination is first made as to whether it is more
likely than not that the income tax position will be sustained,
based upon technical merits, upon examination by the taxing
authorities. If the income tax position is expected to meet the
more likely than not criteria, the benefit recorded in the
financial statements equals the largest amount that is greater than
50% likely to be realized upon its ultimate settlement. At December
31, 2016 and 2015 there were no uncertain tax
positions.
Warrants
The Company has
issued warrants, which allow the warrant holder to purchase one
share of stock at a specified price for a specified period of time.
The Company records equity instruments including warrants issued to
non-employees based on the fair value at the date of issue. The
fair value of warrants classified as equity instruments at the date
of issuance is estimated using the Black-Scholes Model. The fair
value of warrants classified as liabilities at the date of issuance
is estimated using the Monte Carlo Simulation or Binomial
model.
Stock
Based Compensation
The Company records
compensation expense related to options granted to non-employees
based on the fair value of the award.
Compensation cost
is recorded as earned for all unvested stock options outstanding at
the beginning of the first year based upon the grant date fair
value estimates, and for compensation cost for all share-based
payments granted or modified subsequently based on fair value
estimates.
For the years ended
December 31, 2016 and 2015, share-based compensation for options
attributable to employees, officers and Board members were
approximately $95,000 and $1,008,000, respectively. These amounts
have been included in the Company’s statements of operations.
Compensation costs for stock options which vest over time are
recognized over the vesting period. As of December 31, 2016, the
Company had $132,667 of unrecognized compensation costs related to
granted stock options to be recognized over the remaining vesting
period of approximately three years.
3.
FAIR VALUE OF FINANCIAL INSTRUMENTS
The guidance for
fair value measurements, ASC820, Fair Value Measurements and
Disclosures, establishes the authoritative definition of fair
value, sets out a framework for measuring fair value, and outlines
the required disclosures regarding fair value measurements. Fair
value is the price that would be received to sell an asset or paid
to transfer a liability (an exit price) in the principal or most
advantageous market for the asset or liability in an orderly
transaction between market participants at the measurement date.
The Company uses a three-tier fair value hierarchy based upon
observable and non-observable inputs as follow:
●
Level 1 –
Quoted market prices in active markets for identical assets and
liabilities;
●
Level 2 –
Inputs, other than level 1 inputs, either directly or indirectly
observable; and
●
Level 3 –
Unobservable inputs developed using internal estimates and
assumptions (there is little or no market date) which reflect those
that market participants would use.
The Company records
its derivative activities at fair value, which consisted of
warrants as of December 31, 2016. The fair value of the warrants
was estimated using the Binomial Simulation model. Gains and losses
from derivative contracts are included in net gain (loss) from
derivative contracts in the statement of operations. The fair value
of the Company’s derivative warrants is classified as a Level
3 measurement, since unobservable inputs are used in the
valuation.
The following table
presents the fair value for those liabilities measured on a
recurring basis as of December 31, 2016 and
2015:
FAIR
VALUE MEASUREMENTS (In Thousands)
The following is
summary of items that the Company measures at fair value on a
recurring basis:
|
|
Fair Value at
December 31, 2016
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Warrants issued in
connection with Distributor Debt
|
-
|
-
|
(114
)
|
(114
)
|
|
Warrants issued in
connection with Senior Secured Debt
|
-
|
-
|
(1,306
)
|
(1,306
)
|
|
Total
long-term liabilities at fair value
|
$
-
|
$
-
|
$
(1,420
)
|
$
(1,420
)
|
|
|
Fair Value at
December 31, 2015
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Warrants issued in
connection with the issuance of Series C preferred
stock
|
$
—
|
$
—
|
$
(1,145
)
|
$
(1,145
)
|
|
Warrants issued in
connection with the issuance of Series B preferred
stock
|
—
|
—
|
(1,461
)
|
(1,461
)
|
|
Total
long-term liabilities at fair value
|
$
—
|
$
—
|
$
(2,606
)
|
$
(2,606
)
|
The
following is a summary of changes to Level 3 instruments during the
year ended December 31, 2016:
|
|
Fair Value
Measurements Using Significant Unobservable
Inputs (Level
3)
|
|
|
|
|
|
|
|
|
|
Series
C Warrants
|
Series
B Warrants
|
Senior
Secured Debt
|
Distributor
Debt
|
Total
|
|
Balance,
December 31, 2015
|
$
(1,145
)
|
$
(1,461
)
|
$
-
|
$
-
|
$
(2,606
)
|
|
Warrants
issued during the period
|
-
|
-
|
(377
)
|
(114
)
|
(491
)
|
|
Change
in fair value during the period
|
1,145
|
1,461
|
(929
)
|
-
|
1,677
|
|
Balance, December
31, 2016
|
$
-
|
$
-
|
$
1,306
|
$
(114
)
|
$
(1,420
)
|
As of December 31,
2016, the fair value of warrants was approximately $1.4 million. A
net change of approximately $1.7 million has been recorded to the
accompanying statement of operations for the year
ended.
4.
STOCKHOLDER’S DEFICIT
Common
Stock
The Company has
authorized 1,000,000,000 shares of common stock with $0.001 par
value, of which 668,651 were issued and outstanding as of December
31, 2016. For the year ended December 31, 2015, there were
1,000,000,000 authorized shares of common stock, of which 2,964
were issued and outstanding.
A 1:800 reverse
stock split of all of our issued and outstanding common stock was
implemented on November 7, 2016. As a result of the reverse stock
split, every 800 shares of issued and outstanding common stock was
converted into 1 share of common stock. All fractional shares
created by the reverse stock split were rounded to the nearest
whole share. The number of authorized shares of common stock did
not change. On February 24, 2016, the Company had also implemented
a 1:100 reverse stock split of its issued and outstanding common
stock. The number of the authorized shares did not
change.
For the year ended
December 31, 2016, the Company issued 665,687 shares of common
stock as listed below:
|
Series C Preferred
Stock Conversions
|
341,110
|
|
Series C Preferred
Stock Dividends
|
190,107
|
|
Common Stock Issued
as Payment for Accrued Dividends
|
38
|
|
Convertible Debt
Conversions
|
53,080
|
|
Series C
Exchanges
|
22,996
|
|
Series B Tranche B
Warrants Exchanges
|
14,766
|
|
Issuance of shares
due to rounding
|
3,590
|
|
Issuance of shares
in Transit due to Shandong agreement
|
40,000
|
|
Total
|
665,687
|
Preferred
Stock
The Company has
authorized 5,000,000 shares of preferred stock with a $.001 par
value. The board of directors has the authority to issue these
shares and to set dividends, voting and conversion rights,
redemption provisions, liquidation preferences, and other rights
and restrictions. The board of directors designated 525,000 shares
of preferred stock as redeemable convertible preferred stock, none
of which remain outstanding; 33,000 shares of preferred stock as
Series B Preferred Stock, none of which remained outstanding, 9,000
shares of preferred stock as Series C Convertible Preferred Stock,
of which 1,643 and 5,555 were issued and outstanding at December
31, 2016 and 2015, respectively, and 20,250 shares of Series C1
Convertible Preferred Stock, of which 4,312 and none were issued
and outstanding at December 31, 2016 and 2015,
respectively.
Series B Convertible Preferred Stock
Pursuant to the
terms of the Series B Preferred Stock set forth in the Series B
designations, shares of Series B Preferred Stock were convertible
into common stock by their holder at any time, and were mandatorily
convertible upon the achievement of certain conditions, including
the receipt of certain approvals from the U.S. Food and Drug
Administration and the achievement by the Company of specified
average trading prices and volumes for the common
stock.
Holders of the
Series B Preferred Stock were entitled to quarterly dividends at an
annual rate of 10.0%, payable in cash or, subject to certain
conditions, common stock, at the Company’s option. Preferred
dividends totaled approximately none and $352,000 for 2016 and
2015, respectively. Dividends were paid via issuance of common
stock.
The Series B
Preferred Stock were issued with Tranche A warrants to purchase 24
shares of common stock and Tranche B warrants purchasing 7,539
shares of common stock, at an exercise price of $8,364 and $75 per
share, respectively.
At December 31,
2015, as a result of the operation of certain anti-dilution
provisions, the Tranche B warrants were convertible into 1 shares
of common stock. These warrants are re-measured based upon their
fair value each reporting period and classified as a liability on
the Balance Sheet.
Series C Convertible Preferred Stock
On June 29, 2015,
the Company entered into a securities purchase agreement with
certain accredited investors, including John Imhoff, a member of
the Board, for the issuance and sale of an aggregate of 6,737
shares of Series C convertible preferred stock, at a purchase price
of $750 per share and a stated value of $1,000 per share. On
September 3, 2015 the Company entered into an interim agreement
amending the securities purchase agreement to provide for certain
of the investors to purchase an additional aggregate of 1,166
shares. Total cash and non-cash expenses were valued at $853,000,
resulting in net proceeds of $3,698,000.
Pursuant to the
Series C certificate of designations, shares of Series C preferred
stock are convertible into common stock by their holder at any
time, and may be mandatorily convertible upon the achievement of
specified average trading prices for the Company’s common
stock. At December 31, 2016, there were 1,643 shares outstanding
with a conversion price of $1.119 per share, such that each share
of Series C preferred stock would convert into approximately 893
shares of the Company’s common stock, subject to customary
adjustments, including for any accrued but unpaid dividends and
pursuant to certain anti-dilution provisions, as set forth in the
Series C certificate of designations. The conversion price will
automatically adjust downward to 80% of the then-current market
price of the Company’s common stock 15 trading days after any
reverse stock split of the Company’s common stock, and 5
trading days after any conversions of the Company’s
outstanding convertible debt.
Holders of the
Series C preferred stock are entitled to quarterly cumulative
dividends at an annual rate of 12.0% until 42 months after the
original issuance date (the “Dividend End Date”),
payable in cash or, subject to certain conditions, the
Company’s common stock. In addition, upon conversion of the
Series C preferred stock prior to the Dividend End Date, the
Company will also pay to the converting holder a “make-whole
payment” equal to the amount of unpaid dividends through the
Dividend End Date on the converted shares. At December 31, 2016,
the “make-whole payment” for a converted share of
Series C preferred stock would convert to 747 shares of the
Company’s common stock. The Series C preferred stock
generally has no voting rights except as required by Delaware law.
Upon the Company’s liquidation or sale to or merger with
another corporation, each share will be entitled to a liquidation
preference of $1,000, plus any accrued but unpaid
dividends.
In addition, the
purchasers of the Series C preferred stock received, on a pro rata
basis, warrants exercisable to purchase an aggregate of
approximately 150 shares of Company’s common stock. The
warrants contain anti-dilution adjustments in the event that the
Company issues shares of common stock, or securities exercisable or
convertible into shares of common stock, at prices below the
exercise price of such warrants. As a result of the anti-dilution
protection, the Company is required to account for the warrants as
a liability recorded at fair value each reporting period. At
December 31, 2016, the exercise price per share was
$640.
On May 23, 2016, an
investor canceled certain of these warrants, exercisable into 903
shares of common stock. The same investor also transferred certain
of these warrants, exercisable for 150 shares of common stock, to
two investors who also had participated in the 2015 Series C
financing.
Series C1 Convertible Preferred Stock
Between April 27,
2016 and May 3, 2016, the Company entered into various agreements
with certain holders of Series C preferred stock, including
directors John Imhoff and Mark Faupel, pursuant to which those
holders separately agreed to exchange each share of Series C
preferred stock held for 2.25 shares of the Company’s newly
created Series C1 preferred stock and 12 (9,600 pre-split) shares
of the Company’s common stock (the “Series C
Exchanges”). In connection with the Series C Exchanges, each
holder also agreed to roll over the $1,000 stated value per share
of the holder’s shares of Series C1 preferred stock into the
next qualifying financing undertaken by the Company on a
dollar-for-dollar basis and, except in the event of an additional
$50,000 cash investment in the Company by the holder, to execute a
customary “lockup” agreement in connection with the
financing. In total, for 1,916 shares of Series C preferred stock
surrendered, the Company issued 4,312 shares of Series C1 preferred
stock and 22,996 shares of common stock. At December 31, 2016,
there were 4,312 shares outstanding with a conversion price of
$1.119 per share, such that each share of Series C preferred stock
would convert into approximately 893 shares of the Company’s
common stock
The Series C1
preferred stock has terms that are substantially the same as the
Series C preferred stock, except that the Series C1 preferred stock
does not pay dividends (unless and to the extent declared on the
common stock) or at-the-market “make-whole payments”
and, while it has the same anti-dilution protections afforded the
Series C preferred stock, it does not automatically reset in
connection with a reverse stock split or conversion of our
outstanding convertible debt.
Warrants
The following table
summarizes transactions involving the Company’s outstanding
warrants to purchase common stock for the year ended December 31,
2016:
|
|
Warrants
(Underlying
Shares)
|
|
Outstanding,
January 1, 2016
|
3,503
|
|
Issuances
|
4,334,898
|
|
To be
issued
|
21,549
|
|
Canceled /
Expired
|
(10,188
)
|
|
Outstanding,
December 31, 2016
|
4,349,762
|
The Company had the
following shares reserved for the warrants as of December 31,
2016:
Warrants
(Underlying
Shares)
|
|
|
|
|
24
|
(1)
|
$8,368.00 per
share
|
May 23,
2018
|
|
7,542
|
(2)
|
$75.00 per
share
|
June 14,
2021
|
|
3
|
(3)
|
$40,000.00 per
share
|
April 23,
2019
|
|
8
|
(4)
|
$36,000.00 per
share
|
May 22,
2019
|
|
3
|
(5)
|
$30,400.00 per
share
|
September 10,
2019
|
|
5
|
(6)
|
$36,864.80 per
share
|
September 27,
2019
|
|
10
|
(7)
|
$22,504.00 per
share
|
December 2,
2019
|
|
105
|
(8)
|
$7,200.00 per
share
|
December 2,
2020
|
|
105
|
(9)
|
$8,800.00 per
share
|
December 2,
2020
|
|
25
|
(11)
|
$20,400.00 per
share
|
March 30,
2018
|
|
22
|
(12)
|
$9,504.00 per
share
|
June 29,
2020
|
|
659
|
(10)
|
$640.00 per
share
|
June 29,
2020
|
|
343
|
(11)
|
$640.00 per
share
|
September 4,
2020
|
|
363
|
(12)
|
$640.00 per
share
|
September 21,
2020
|
|
7
|
(13)
|
$9,504.00 per
share
|
September 4,
2020
|
|
198
|
(14)
|
$640.00 per
share
|
October 23,
2020
|
|
7
|
(15)
|
$9,504.00 per
share
|
October 23,
2020
|
|
4,120,977
|
(16)
|
$0.3488 per
share
|
June 14,
2021
|
|
197,807
|
(17)
|
$0.3488 per
share
|
February 21,
2021
|
|
21,549
|
(18)
|
$13.92 per
share
|
June 6,
2021
|
|
4,349,762
|
|
|
|
|
|
|
|
(1)
|
Issued in June 2015
in exchange for warrants originally issued as part of a May 2013
private placement.
|
|
(2)
|
Issued in June 2015
in exchange for warrants originally issued as part of a May 2013
private placement.
|
|
(3)
|
Issued to a
placement agent in conjunction with an April 2014 private
placement.
|
|
(4)
|
Issued to a
placement agent in conjunction with a September 2014 private
placement.
|
|
(5)
|
Issued as part of a
September 2014 Regulation S offering.
|
|
(6)
|
Issued to a
placement agent in conjunction with a 2014 public
offering.
|
|
(7)
|
Issued in June 2015
in exchange for warrants originally issued as part of a 2014 public
offering.
|
|
(8)
|
Issued as part of a
March 2015 private placement.
|
|
(9)
|
Issued to a
placement agent in conjunction with a June 2015 private
placement.
|
|
(10)
|
Issued as part of a
June 2015 private placement.
|
|
(11)
|
Issued as part of a
June 2015 private placement.
|
|
(12)
|
Issued as part of a
June 2015 private placement.
|
|
(13)
|
Issued to a
placement agent in conjunction with a June 2015 private
placement.
|
|
(14)
|
Issued as part of a
June 2015 private placement.
|
|
(15)
|
Issued to a
placement agent in conjunction with a June 2015 private
placement.
|
|
(16)
|
Issued as part of a
February 2016 private placement.
|
|
(17)
|
Issued to a
placement agent in conjunction with a February 2016 private
placement.
|
|
(18)
|
Contractually
obligated to be issued pursuant to a strategic license
agreement.
|
All outstanding
warrant agreements provide for anti-dilution adjustments in the
event of certain mergers, consolidations, reorganizations,
recapitalizations, stock dividends, stock splits or other changes
in the Company’s corporate structure; except for (9). In
addition, warrants subject to footnotes (2) and (10)-(12), (14),
and (16) – (18) in the table above are subject to
“lower price issuance” anti-dilution provisions that
automatically reduce the exercise price of the warrants (and, in
the cases of warrants subject to footnote (2), (16) and (17) in the
table above, increase the number of shares of common stock issuable
upon exercise), to the offering price in a subsequent issuance of
the Company’s common stock, unless such subsequent issuance
is exempt under the terms of the warrants.
The warrants
subject to footnote (2) are subject to a mandatory exercise
provision. This provision permits the Company, subject to certain
limitations, to require exercise of such warrants at any time
following (a) the date that is the 30th day after the later of the
Company’s receipt of an approvable letter from the FDA for
LuViva and the date on which the common stock achieves an average
market price for 20 consecutive trading days of at least $1,040.00
with an average daily trading volume during such 20 consecutive
trading days of at least 250 shares, or (b) the date on which the
average market price of the common stock for 20 consecutive trading
days immediately prior to the date the Company delivers a notice
demanding exercise is at least $129,600.00 and the average daily
trading volume of the common stock exceeds 250 shares for such 20
consecutive trading days. If these warrants are not timely
exercised upon demand, they will expire. Upon the occurrence of
certain events, the Company may be required to repurchase these
warrants, as well as the warrants subject to footnote (2) in the
table above.
The warrants
subject to footnote (5) in the table above are also subject to a
mandatory exercise provision. This provision permits the Company,
subject to certain limitations; to require the exercise of such
warrants should the average trading price of its common stock over
any 30 consecutive day trading period exceed
$92.16.
The warrants
subject to footnote (7) in the table above are also subject to a
mandatory exercise provision. This provision permits the Company,
subject to certain limitations, to require exercise of 50% of the
then-outstanding warrants if the trading price of its common stock
is at least two times the initial warrant exercise price for any
20-day trading period. Further, in the event that the trading price
of the Company’s common stock is at least 2.5 times the
initial warrant exercise price for any 20-day trading period, the
Company will have the right to require the immediate exercise of
50% of the then-outstanding warrants. Any warrants not exercised
within the prescribed time periods will be canceled to the extent
of the number of shares subject to mandatory
exercise.
The holders of the
warrants subject to footnote (2) in the table above have agreed to
surrender the warrants, upon consummation of a qualified public
financing, for new warrants exercisable for 200% of the number of
shares underlying the surrendered warrants, but without certain
anti-dilution protections included with the surrendered
warrants.
Series
B Tranche B Warrants
As discussed in
Note 3, Fair Value Measurements, between June 13, 2016 and June 14,
2016, the Company entered into various agreements with holders of
the Company’s “Series B Tranche B” warrants,
pursuant to which each holder separately agreed to exchange the
warrants for either (1) shares of common stock equal to 166% of the
number of shares of common stock underlying the surrendered
warrants, or (2) new warrants exercisable for 200% of the number of
shares underlying the surrendered warrants, but without certain
anti-dilution protections included with the surrendered warrants.
In total, for surrendered warrants then-exercisable for an
aggregate of 1,185,357 shares of common stock (but subject to
exponential increase upon operation of certain anti-dilution
provisions), the Company issued or is obligated to issue 16,897
shares of common stock and new warrants that, if exercised as of
the date hereof, would be exercisable for an aggregate of 216,707
shares of common stock. As of December 31, 2016, the Company had
issued 14,766 shares of common stock and rights to common stock
shares for 2,131. In certain circumstances, in lieu of presently
issuing all of the shares (for each holder that opted for shares of
common stock), the Company and the holder further agreed that the
Company will, subject to the terms and conditions set forth in the
applicable warrant exchange agreement, from time to time, be
obligated to issue the remaining shares to the holder. No
additional consideration will be payable in connection with the
issuance of the remaining shares. The holders that elected to
receive shares for their surrendered warrants have agreed that they
will not sell shares on any trading day in an amount, in the
aggregate, exceeding 20% of the composite aggregate trading volume
of the common stock for that trading day. The holders that elected
to receive new warrants will be required to surrender their old
warrants upon consummation of the Company’s next financing
resulting in net cash proceeds to the Company of at least $1
million. The new warrants will have an initial exercise price equal
to the exercise price of the surrendered warrants as of immediately
prior to consummation of the financing, subject to customary
“downside price protection” for as long as the
Company’s common stock is not listed on a national securities
exchange, and will expire five years from the date of
issuance.
5.
INCOME TAXES
The Company has
incurred net operating losses ("NOLs") since inception. As of
December 31, 2016, the company had NOL carryforwards available
through 2035 of approximately $79.3 million to offset its future
income tax liability. The NOL carryforwards began to expire in
2008. The company has not recorded deferred tax assets due to
uncertainties related to utilization of NOLs as well as calculation
of effective tax rate. Utilization of existing NOL carryforwards
may be limited in future years based on significant ownership
changes. The company is in the process of analyzing their NOL and
has not determined if the company has had any change of control
issues that could limit the future use of NOL.
Components of
deferred taxes are as follow at December 31 (in
thousands):
|
|
|
|
|
Deferred tax
assets
|
$
795
|
$
626
|
|
Net operating loss
carry forwards
|
27,958
|
28,201
|
|
Deferred tax
liabilities: intangible assets and other
|
-
|
-
|
|
|
28,753
|
28,827
|
|
Valuation
allowance
|
(28,753
)
|
(28,827
)
|
|
|
$
0
|
$
0
|
The following is a
summary of the items that caused recorded income taxes to differ
from taxes computed using the statutory federal income tax rate for
the years ended December 31:
|
|
|
|
|
Statutory federal
tax rate
|
34
%
|
34
%
|
|
State taxes, net of
federal benefit
|
4
|
4
|
|
Nondeductible
expenses
|
-
|
-
|
|
Valuation
allowance
|
(38
)
|
(38
)
|
|
|
0
%
|
0
%
|
6.
STOCK OPTIONS
The Company’s
1995 Stock Plan (the “Plan”) has expired pursuant to
its terms, so zero shares remained available for issuance at
December 31, 2016 and 2015. The Plan allowed for the issuance of
incentive stock options, nonqualified stock options, and stock
purchase rights. The exercise price of options was determined by
the Company’s board of directors, but incentive stock options
were granted at an exercise price equal to the fair market value of
the Company’s common stock as of the grant date. Options
historically granted have generally become exercisable over four
years and expire ten years from the date of
grant.
As of December 31,
2016, the Company has issued and outstanding options to purchase a
total of 125 shares of common stock pursuant to the Plan, at a
weighted average exercise price of $37,920 per
share.
The fair value of
stock options granted in the period ended December 31, 2015 were
estimated using the Black-Scholes option pricing model. No options
were issued during the period ended December 31,
2016.
Stock option
activity for December 31, 2016 and 2015 as
follows:
|
|
|
|
|
|
Weighted
Average
Exercise
Price
|
|
Outstanding at
beginning of year
|
87
|
$
36,000
|
|
Options
granted
|
52
|
$
9,600
|
|
Options
exercised
|
(2
)
|
$
38,400
|
|
Options
expired/forfeited
|
(5
)
|
$
34,400
|
|
Outstanding at end
of year
|
132
|
$
36,000
|
|
Options available
for issue
|
-
|
|
|
|
2016
|
|
|
|
Weighted
Average
Exercise
Price
|
|
Outstanding at
beginning of year
|
132
|
$
36,000
|
|
Options
granted
|
-
|
$
-
|
|
Options
exercised
|
-
|
$
-
|
|
Options
expired/forfeited
|
(7
)
|
$
74,160
|
|
Outstanding at end
of year
|
125
|
$
37,920
|
|
Options available
for issue
|
-
|
|
|
|
|
|
|
|
|
|
|
Options Vested as
of December 31, 2015
|
113
|
$
39,200
|
|
Options
vested in 2016
|
4
|
$
22,860
|
|
Options vested as
of December 31, 2016
|
117
|
$
38,640
|
|
|
|
|
|
|
|
|
|
Options Unvested as
of December 31, 2015
|
19
|
$
39,200
|
|
Options
vested in 2016
|
(4
)
|
$
22,860
|
|
Options
expired/forfeited in 2016
|
(7
)
|
$
74,160
|
|
Options Unvested as
of December 31, 2016
|
8
|
|
7.
LITIGATION AND CLAIMS
From time to time,
the Company may be involved in various legal proceedings and claims
arising in the ordinary course of business. Management believes
that the dispositions of these matters, individually or in the
aggregate, are not expected to have a material adverse effect on
the Company’s financial condition. However, depending on the
amount and timing of such disposition, an unfavorable resolution of
some or all of these matters could materially affect the future
results of operations or cash flows in a particular
period.
As of December 31,
2016 and 2015, there was no accrual recorded for any potential
losses related to pending litigation.
8.
COMMITMENTS AND CONTINGENCIES
Operating
Leases
In December 2009,
the Company moved its offices, which comprise its administrative,
research and development, marketing and production facilities to
5835 Peachtree Corners East, Suite D, Norcross, Georgia 30092. The
Company leases approximately 23,000 square feet under a lease that
expires in June 2017. The fixed monthly lease expense is
approximately $15,000 plus common charges. The Company also leases
office and equipment under operating lease agreements with monthly
payments of approximately $2,000. These leases expire at various
dates through April 2016. Future minimum rental payments at
December 31, 2016 under non-cancellable operating leases for office
space and equipment are as follows (in
thousands):
Related
Party Contracts
On June 5, 2016,
the Company entered into a license agreement with Shenghuo Medical,
LLC pursuant to which the Company granted Shenghuo an exclusive
license to manufacture, sell and distribute LuViva in Taiwan,
Brunei Darussalam, Cambodia, Laos, Myanmar, Philippines, Singapore,
Thailand, and Vietnam. Shenghuo was already the Company’s
exclusive distributor in China, Macau and Hong Kong, and the
license extended to manufacturing in those countries as well. Under
the terms of the license agreement, once Shenghuo was capable of
manufacturing LuViva in accordance with ISO 13485 for medical
devices, Shenghuo would pay the Company a royalty equal to $2.00 or
20% of the distributor price (subject to a discount under certain
circumstances), whichever is higher, per disposable distributed
within Shenghuo’s exclusive territories. In connection with
the license grant, Shenghuo was to underwrite the cost of securing
approval of LuViva with Chinese Food and Drug Administration. At
its option, Shenghuo also would provide up to $1.0 million in
furtherance of the Company’s efforts to secure regulatory
approval for LuViva from the U.S. Food and Drug Administration, in
exchange for the right to receive payments equal to 2% of the
Company’s future sales in the United States, up to an
aggregate of $4.0 million. Pursuant to the license agreement,
Shenghuo had the option to have a designee appointed to the
Company’s board of directors (director Richard Blumberg is
that designee). As partial consideration for, and as a condition
to, the license, and to further align the strategic interests of
the parties, the Company agreed to issue a convertible note to
Shenghuo, in exchange for an aggregate cash investment of $200,000.
The note will provide for a payment to Shenghuo of $300,000,
expected to be due the earlier of 90 days from issuance and
consummation of any capital raising transaction by the Company with
net cash proceeds of at least $1.0 million. The note will accrue
interest at 20% per year on any unpaid amounts due after that date.
The note will be convertible into shares of the Company’s
common stock at a conversion price per share of $13.92, subject to
customary anti-dilution adjustment. The note will be unsecured, and
is expected to provide for customary events of default. The Company
will also issue Shenghuo a five-year warrant exercisable
immediately for approximately 21,549 shares of common stock at an
exercise price equal to the conversion price of the note, subject
to customary anti-dilution adjustment. On January 22, 2017, the
Company entered into a license agreement with Shandong Yaohua
Medical Instrument Corporation, or SMI, pursuant to which the
Company granted SMI an exclusive global license to manufacture the
LuViva device and related disposables (subject to a carve-out for
manufacture in Turkey) and exclusive distribution rights in the
Peoples Republic of China, Macau, Hong Kong and Taiwan. In order to
facilitate the SMI agreement, immediately prior to its execution
the Company entered into an agreement with Shenghuo Medical, LLC,
regarding its previous license to Shenghuo. Under the terms of the
new agreement, Shenghuo agreed to relinquish its manufacturing
license and its distribution rights in SMI’s territories, and
to waive its rights under the original Shenghuo agreement, all for
as long as SMI performs under the SMI agreement. See Note 13,
Subsequent Events.
On September 6,
2016, the Company entered into a royalty agreement with one of its
directors, John Imhoff, and another stockholder, Dolores Maloof,
pursuant to which the Company sold to them a royalty of future
sales of single-use cervical guides for LuViva. Under the terms of
the royalty agreement, and for consideration of $50,000, the
Company will pay them an aggregate perpetual royalty initially
equal to $0.10, and from and after October 2, 2016, equal to $0.20,
for each disposable that the Company sells (or that is sold by a
third party pursuant to a licensing arrangement with the
Company).
9.
NOTES PAYABLE
Notes
Payable in Default
At December 31,
2016 and 2015, the Company maintained notes payable and accrued
interest to both related and non-related parties totaling
$1,539,000 and $133,000, respectively. These notes are short term,
straight-line amortizing notes. The notes carry annual interest
rates between 5% and 10% and have default rates as high a
16.5%.
Short
Term Notes Payable
At December 31,
2016 and 2015, the Company maintained short term notes payable and
accrued interest to both related and non-related parties totaling
$127,000 and $634,000, respectively. These notes are short term,
straight-line amortizing notes. The notes carry annual interest
rates between 5% and 10%.
In June 2016, the
Company entered into a premium finance agreement to finance its
insurance policies totaling $193,862. The note requires monthly
payments of $17,622, including interest at 4.87% and matures in
April 2017. The balance due on this note totaled $70,000 at
December 31, 2016.
In June 2015, the
Company entered into a similar short-term note payable for the
financing of its insurance policies. This note required monthly
payments of $17,614, including interest at 5.2% and matured in
April 2016. The balance due on this note totaled $70,000 at
December 31, 2015.
10.
SHORT-TERM CONVERTIBLE DEBT
Related
Party Convertible Note Payable –
Short-Term
On June 5, 2016,
the Company entered into a license agreement with a distributor
pursuant to which the Company granted the distributor an exclusive
license to manufacture, sell and distribute the Company’s
LuViva Advanced Cervical Cancer device and related disposables in
Taiwan, Brunei Darussalam, Cambodia, Laos, Myanmar, Philippines,
Singapore, Thailand, and Vietnam. The distributor was already the
Company’s exclusive distributor in China, Macau and Hong
Kong, and the license will extend to manufacturing in those
countries as well.
As
partial consideration for, and as a condition to, the license, and
to further align the strategic interests of the parties, the
Company agreed to issue a convertible note to the distributor, in
exchange for an aggregate cash investment of $200,000. The note
will provide for a payment to the distributor of $240,000, due upon
consummation of any capital raising transaction by the Company
within 90 days and with net cash proceeds of at least $1.0 million.
As of December 31, 2016 the Company had a note due of $300,000. The
note will accrue interest at 20% per year on any unpaid amounts due
after that date. The note will be convertible into shares of the
Company’s common stock at a conversion price per share of
$13.92, subject to customary anti-dilution adjustment. The note
will be unsecured, and is expected to provide for customary events
of default. The Company will also issue the distributor a five-year
warrant exercisable immediately for 17,239 shares of common stock
at an exercise price equal to the conversion price of the note,
subject to customary anti-dilution adjustment.
Convertible Note Payable – Short-Term
On
December 28, 2016, the Company entered into a securities purchase
agreement with an investor for the issuance and sale to investor of
up to $330,000 in aggregate principal amount of 10% original
issuance discount convertible promissory notes, for an aggregate
purchase price of $300,000. On that date, the Company issued to the
investor a note in the principal amount of $222,000, for a purchase
price of $200,000. The note matures six months from their date of
issuance and, in addition to the 10% original issue discount,
accrue interest at a rate of 10% per year. The Company may prepay
the notes, in whole or in part, for 115% of outstanding principal
and interest until 30 days from issuance, for 125% of outstanding
principal and interest at any time from 31 to 60 days from
issuance, and for 130% of outstanding principal and interest at any
time from 61 days from issuance until immediately prior to the
maturity date. After six months from the date of issuance (i.e., if
the Company fails to repay all principal and interest due under the
notes at the maturity date), the investor may convert the notes, at
any time, in whole or in part, into shares of the Company’s
common stock, at a conversion price equal to 60% of the lowest
volume weighted average price of our common stock during the 20
trading days prior to conversion, subject to certain customary
adjustments and anti-dilution provisions contained in the
note.
As
of December 31, 2016, the Company had debt issue costs of $30,000,
net of debt of $168,000.
11.
CONVERTIBLE DEBT IN DEFAULT
Secured Promissory Note.
On
September 10, 2014, the Company sold a secured promissory note to
an accredited investor with an initial principal amount of
$1,275,000, for a purchase price of $700,000 (an original issue
discount of $560,000). The Company may prepay the note at any time.
The note is secured by the Company’s current and future
accounts receivable and inventory, pursuant to a security agreement
entered into in connection with the sale. On March 10, 2015, May 4,
2015, June 1, 2015, June 16, 2015, June 29, 2015, January 21, 2016,
January 29, 2016, and February 12, 2016 the Company amended the
terms of the note to extend the maturity ultimately until August
31, 2016. During the extension, interest accrues on the note at a
rate of the lesser of 18% per year or the maximum rate permitted by
applicable law. On February 11, 2016, the Company consented to an
assignment of the note to two accredited investors. In connection
with the assignment, the holders waived an ongoing event of default
under the notes related to the Company’s minimum market
capitalization, and agreed to eliminate the requirement going
forward. Pursuant to the terms of the amended note, the holder may
convert the outstanding balance into shares of common stock at a
conversion price per share equal to the lower of (1) $25.0 or (2)
75% of the lowest daily volume weighted average price of the common
stock during the five days prior to conversion. If the conversion
price at the time of any conversion is lower than $15.00, the
Company has the option of delivering the conversion amount in cash
in lieu of shares of common stock. On March 7, 2016, the Company
further amended the note to eliminate the volume limitations on
sales of common stock issued or issuable upon conversion. On July
13, 2016, the Company consented to the assignment by one of the
accredited investors of its portion of the note of to a third
accredited investor.
The
balance due on the note was $530,691 and $685,864 at December 31,
2016 and 2015, respectively. The balance was reduced by $306,863 as
part of a debt restructuring completed with an accredited investor
on December 7, 2016 (see Note 11).
Total
debt issuance costs as originally capitalized were approximately
$130,000. This amount was amortized over nine months and was fully
amortized as of December 31, 2015. Total amortized expense for the
years ended December 31, 2015 was approximately $49,000. For the
year ended December 31, 2015, the Company recorded amortization of
approximately $213,000 on the discount. The original issue discount
of $560,000 was fully amortized as of December 31,
2015.
On
November 2, 2016, the Company entered into a lockup and exchange
agreement with GHS Investments, LLC, holder of approximately
$221,000 in outstanding principal amount of the Company’s
secured promissory note and all of the outstanding shares of the
its Series C preferred stock. Pursuant to the agreement, upon the
effectiveness of the 1:800 reverse stock split and continuing for
45 days after, GHS and its affiliates were prohibited from
converting any portion of the secured promissory note or any of the
shares of Series C preferred stock or selling any of the
Company’s securities that they beneficially owned. The
Company agreed that, upon consummation of its next financing, the
Company would use $260,000 of net cash proceeds first, to repay
GHS’s portion of the secured promissory note and second, with
any remaining amount from the $260,000, to repurchase a portion of
GHS’s shares of Series C preferred stock. In addition, GHS
has agreed to exchange the stated value per share (plus any accrued
but unpaid dividends) of its remaining shares of Series C preferred
stock for new securities of the same type that the Company
separately issue in the next qualifying financing it undertakes, on
a dollar-for-dollar basis in a private placement
exchange.
Senior Secured Promissory Note
On
February 11, 2016, the Company entered into a securities purchase
agreement with an accredited investor for the issuance and sale on
February 12, 2016 of $1.4375 million in aggregate principal amount
of a senior secured convertible note for an aggregate purchase
price of $1.15 million (a 20% original issue discount of $287,500)
and a discount for debt issuance costs paid at closing of $121,000
for a total of $408,500. In addition, the investor received a
warrant exercisable to purchase an aggregate of approximately 2,246
shares of the Company’s common stock. The Company allocated
proceeds totaling $359,555 to the fair value of the warrants at
issuance. This was recorded as an additional discount on the debt.
The convertible note matures on the second anniversary of issuance
and, in addition to the 20% original issue discount, accrues
interest at a rate of 17% per year. The Company is required to pay
monthly interest coupons and beginning nine months after issuance,
the Company is required to pay amortized quarterly principal
payments. If the Company does not receive, on or before the first
anniversary after issuance, an aggregate of at least $3.0 million
from future equity or debt financings or non-dilutive grants, then
the holder will have the option of accelerating the maturity date
to the first anniversary of issuance. The Company may prepay the
convertible note, in whole or in part, without penalty, upon 20
days’ prior written notice. Subject to resale restrictions
under Federal securities laws and the availability of sufficient
authorized but unissued shares of the Company’s common stock,
the convertible note is convertible at any time, in whole or in
part, at the holder’s option, into shares of the
Company’s common stock, at a conversion price equal to the
lesser of $0.80 per share or 70% of the average closing price per
share for the five trading days prior to issuance, subject to
certain customary adjustments and anti-dilution provisions
contained in the convertible note. On May 28, 2016, in exchange for
an additional $87,500 in cash from the holder to the Company, the
principal balance was increased by the same amount. The Company is
currently in default as they are past due on the required monthly
interest payments. In the event of default, the Company shall
accrue interest at a rate the lesser of 22% or the maximum
permitted by law. The Company has accrued $78,500 for past due
interest payments at December 31, 2016. Upon the occurrence of an
event of default, the holder may require the Company to redeem the
convertible note at 120% of the outstanding principal balance (but
as of December 31, 2016, had not done so). As of December 31, 2016,
the balance due on the convertible debt was $1,830,000 as the
Company has fully amortized debt issuance costs of $47,675 and the
debt discount of $768,055 and recorded a 20% penalty totaling
$305,000. The convertible note is secured by a lien on all of the
Company’s assets, including its intellectual property,
pursuant to a security agreement entered into by the Company and
the accredited investor with the transaction
holder.
The
warrant is exercisable at any time, pending availability of
sufficient authorized but unissued shares of the Company’s
common stock, at an exercise price per share equal to the
conversion price of the convertible note, subject to certain
customary adjustments and anti-dilution provisions contained in the
warrant. The warrant has a five-year term. As of December 31, 2016,
the exercise price had been adjusted to $0.35 and the number of
common stock shares exchangeable for was 4,120,978. As of December
31, 2016, the effective interest rate considering debt costs was
29%.
The
Company used a placement agent in connection with the transaction.
For its services, the placement agent received a cash placement fee
equal to 4% of the aggregate gross proceeds from the transaction
and a warrant to purchase shares of common stock equal to an
aggregate of 6% of the total number of shares underlying the
securities sold in the transaction, at an exercise price equal to,
and terms otherwise identical to, the warrant issued to the
investor. Finally, the Company agreed to reimburse the placement
agent for its reasonable out-of-pocket
expenses.
In
connection with the transaction, on February 12, 2016, the Company
and the investor entered into a four-year consulting agreement,
pursuant to which the investor will provide management consulting
services to the Company in exchange for a royalty payment, payable
quarterly, equal to 3.5% of the Company’s revenues from the
sale of products. As of December 31, 2016 the investor had earned
approximately $23,000 of royalties.
Debt
Restructuring
.
On
December 7, 2016, the Company entered into an exchange agreement
with GPB Debt Holdings II LLC with regard to the $1,525,000 in
outstanding principal amount of senior secured convertible note
originally issued to GPB on February 11, 2016, and the $306,863 in
outstanding principal amount of the Company’s secured
promissory note that GPB holds (see “—Secured
Promissory Note”). Pursuant to the exchange agreement, upon
completion of the next financing resulting in at least $1 million
in cash proceeds, GPB will exchange both securities for a new
convertible note in principal amount of $1,831,863. The new
convertible note will mature on the second anniversary of issuance
and will accrue interest at a rate of 19% per year. The Company
will pay monthly interest coupons and, beginning one year after
issuance, will pay amortized quarterly principal payments. Subject
to resale restrictions under Federal securities laws and the
availability of sufficient authorized but unissued shares of the
Company’s common stock, the new convertible note will be
convertible at any time, in whole or in part, at the holder’s
option, into shares of common stock, at a conversion price equal to
the price offered in the qualifying financing that triggers the
exchange, subject to certain customary adjustments and
anti-dilution provisions contained in the new convertible note. The
new convertible note will include customary event of default
provisions and a default interest rate of the lesser of 21% or the
maximum amount permitted by law. Upon the occurrence of an event of
default, GPB will be entitled to require the Company to redeem the
new convertible note at 120% of the outstanding principal balance.
The new convertible note will be secured by a lien on all of the
Company’s assets, including its intellectual property,
pursuant to the security agreement entered into by the Company and
GPB in connection with the issuance of the original senior secured
convertible note. As an inducement to GPB to enter into these
transactions, the Company agreed to increase the royalty payable to
GPB pursuant to its consulting agreement with us from 3.5% to 3.85%
of revenues from the sales of the Company’s
products.
12.
INCOME (LOSS) PER COMMON SHARE
Basic net income
(loss) per share attributable to common stockholders amounts are
computed by dividing the net income (loss) plus preferred stock
dividends and deemed dividends on preferred stock by the weighted
average number of shares outstanding during the
period.
Diluted net income
(loss) per share attributable to common stockholders amounts are
computed by dividing the net income (loss) plus preferred stock
dividends, deemed dividends on preferred stock, after-tax interest
on convertible debt and convertible dividends by the weighted
average number of shares outstanding during the period, plus Series
C convertible preferred stock, convertible debt, convertible
preferred dividends and warrants convertible into common stock
shares.
Diluted net loss
per common share is the same as basic net loss per common share
since the Company was operating in a loss position for 2016 and
2015.
13. SUBSEQUENT
EVENTS
On January 22,
2017, the Company entered into a license agreement with Shandong
Yaohua Medical Instrument Corporation, or SMI, pursuant to which
the Company granted SMI an exclusive global license to manufacture
the LuViva device and related disposables (subject to a carve-out
for manufacture in Turkey) and exclusive distribution rights in the
Peoples Republic of China, Macau, Hong Kong and Taiwan. In exchange
for the license, SMI will pay a $1.0 million licensing fee, payable
in five installments through October 2017, as well as a royalty on
each disposable sold in the territories. SMI will also underwrite
the cost of securing approval of LuViva with the Chinese Food and
Drug Administration, or CFDA. Pursuant to the SMI agreement, SMI
must become capable of manufacturing LuViva in accordance with ISO
13485 for medical devices by the second anniversary of the SMI
agreement, or else forfeit the license. During 2017, SMI must
purchase no fewer than ten devices (with up to two devices pushed
to 2018 if there is a delay in obtaining approval from the CFDA).
In the three years following CFDA approval, SMI must purchase a
minimum of 3,500 devices (500 in the first year, 1,000 in the
second, and 2,000 in the third) or else forfeit the license. As
manufacturer of the devices and disposables, SMI will be obligated
to sell each to us at costs no higher than our current costs. As
partial consideration for, and as a condition to, the license, and
to further align the strategic interests of the parties, the
Company agreed to issue $1.0 million in shares of its common stock
to SMI, in five installments through October 2017, at a price per
share equal to the lesser of the average closing price for the five
days prior to issuance and $1.25.
In order to
facilitate the SMI agreement, immediately prior to its execution
the Company entered into an agreement with Shenghuo Medical, LLC,
regarding its previous license to Shenghuo (see Note 8, Commitments
and Contingencies). Under the terms of the new agreement, Shenghuo
agreed to relinquish its manufacturing license and its distribution
rights in SMI’s territories, and to waive its rights under
the original Shenghuo agreement, all for as long as SMI performs
under the SMI agreement. As consideration, the Company agreed to
split with Shenghuo the licensing fees and net royalties from SMI
that the Company will receive under the SMI agreement. Should the
SMI agreement be terminated, the Company have agreed to re-issue
the original license to Shenghuo under the original terms. The
Company’s COO and director, Mark Faupel, is a shareholder of
Shenghuo, and another director, Richard Blumberg, is a managing
member of Shenghuo.
On February 13,
2017, the Company entered into a securities purchase agreement with
Auctus Fund, LLC for the issuance and sale to Auctus of $170,000 in
aggregate principal amount of a 12% convertible promissory note for
an aggregate purchase price of $156,400 (representing a $13,600
original issue discount). On February 13, 2017, the Company issued
the note to Auctus. Pursuant to the purchase agreement, the Company
also issued to Auctus a warrant exercisable to purchase an
aggregate of 200,000 shares of the Company’s common stock.
The warrant is exercisable at any time, at an exercise price per
share equal to $0.77 (110% of the closing price of the common stock
on the day prior to issuance), subject to certain customary
adjustments and price-protection provisions contained in the
warrant. The warrant has a five-year term. The note matures nine
months from the date of issuance and, in addition to the original
issue discount, accrues interest at a rate of 12% per year. The
Company may prepay the note, in whole or in part, for 115% of
outstanding principal and interest until 30 days from issuance, for
125% of outstanding principal and interest at any time from 31 to
60 days from issuance, and for 130% of outstanding principal and
interest at any time from 61 days from issuance to 180 days from
issuance. After six months from the date of issuance, Auctus may
convert the note, at any time, in whole or in part, into shares of
the Company’s common stock, at a conversion price equal to
the lower of the price offered in the Company’s next public
offering or a 40% discount to the average of the two lowest trading
prices of the common stock during the 20 trading days prior to the
conversion, subject to certain customary adjustments and
price-protection provisions contained in the note. The note
includes customary events of default provisions and a default
interest rate of 24% per year. Upon the occurrence of an event of
default, Auctus may require the Company to redeem the note (or
convert it into shares of common stock) at 150% of the outstanding
principal balance plus accrued and unpaid interest. In connection
with the transaction, the Company agreed to reimburse Auctus for
$30,000 in legal and diligence fees, of which we paid $10,000 in
cash and $20,000 in restricted shares of common stock, valued at
$0.40 per share (a 42.86% discount to the closing price of the
common stock on the day prior to issuance).
PART II
INFORMATION NOT REQUIRED IN
PROSPECTUS
ITEM 13.
OTHER EXPENSES OF ISSUANCE AND
DISTRIBUTION
The following table
sets forth the various expenses and costs incurred or to be
incurred by us in connection with the sale of the shares of common
stock offered hereby, other than selling commissions, which will be
borne by the selling stockholders. All the amounts shown are
estimated except the SEC registration fee.
|
Expense
|
|
|
Dollar
Amount
|
|
|
|
SEC filing fee
|
|
$
|
1
,269
|
|
|
Legal fees and expenses
|
|
|
250,000
|
|
|
Accounting fees and expenses
|
|
|
75,000
|
|
|
Blue sky and related expenses
|
|
|
5,800
|
|
|
Miscellaneous
|
|
|
7
,931
|
|
|
Total
|
|
$
|
340,000
|
|
ITEM 14.
INDEMNIFICATION OF DIRECTORS AND
OFFICERS
Section 145 of the
Delaware General Corporation Law permits a corporation to include
in its charter documents, and in agreements between the corporation
and its directors and officers, provisions expanding the scope of
indemnification beyond that specifically provided by the current
law. Article VII of our certificate of incorporation provides for
the indemnification of directors to the fullest extent permissible
under Delaware law. Article VII of our bylaws provides for the
indemnification of officers, directors and third parties acting on
our behalf if such person acted in good faith and in a manner
reasonably believed to be in and not opposed to our best interest
and, with respect to any criminal action or proceeding, if the
indemnified party had no reason to believe his conduct was
unlawful.
ITEM 15. RECENT SALES OF UNREGISTERED
SECURITIES
On February 24, 2016, we implemented a 1:100
reverse stock split of all of our issued and outstanding common
stock. As a result of the reverse stock split, every 100 shares of
issued and outstanding common stock was converted into 1 share of
common stock
A
1:800 reverse
stock split of all of our issued and outstanding common stock
was implemented on November 7, 2016. As a result of
the reverse stock split, every 800 shares of issued and outstanding
common stock was converted into 1 share of common stock. In this
Item 15, all share and per share amounts for transactions occurring
prior to these dates are reported on a pre-stock split basis with
regard to the applicable reverse stock split.
Simple Interest Notes.
On February
20, 2014, our President and CEO, Gene Cartwright, advanced us
$50,000, in cash, for a 10% simple interest note. On October 23,
2014, August 4, 2014, and May 21, 2014, he advanced us $30,000,
$200,000, and $100,000, respectively, in cash for 5% simple
interest notes. On December 30, 2015, he advanced us $10,000 in
cash for a 6% simple interest note. On October 24, 2014, October 7,
2014, and August 26, 2014, our Senior Vice President of
Engineering, Richard Fowler, advanced us $6,100, $20,000 and
$75,000, respectively, in cash for 6% simple interest notes. On
October 7, 2014, our Director of Marketing advanced us $10,000 in
cash for a 6% simple interest note. On November 4, 2014, Richard
Blumberg, one of our stockholders, advanced us $100,000 in cash for
a note for $106,500 in aggregate principal and interest due
November 30, 2014. On February 20, 2014, directors Michael James,
Linda Rosenstock (since resigned), and John Imhoff advanced us
$50,000, $50,000, $50,000, and $25,000 in cash, respectively, for
10% simple interest notes.
April 2014 Senior Convertible Note
Financing.
On April 23, 2014, we entered into a
securities purchase agreement with Magna Equities II, LLC (formerly
known as Hanover Holdings I, LLC). Pursuant to the purchase
agreement, we sold Magna a senior convertible note with a principal
amount of $1.5 million, for a purchase price of $1.0 million (an
approximately 33.3% original issue discount). As part of the sale,
we also issued Magna 321,820 shares of common stock. Additionally,
pursuant to the purchase agreement, Magna purchased on May 23, 2014
an additional senior convertible note with a principal amount of
$2.0 million and an 18-month term, for a fixed purchase price of
$2.0 million. We had the right at any time to redeem all or a
portion of the total outstanding amount then remaining on the
notes, at a 25% premium. As of June 30, 2015, the notes are no
longer outstanding. The notes accrued interest at an annual rate of
6.0% and were convertible into shares of our common stock, at a
conversion price equal to the lesser of $0.55 per share and a
discount from the lowest daily volume-weighted average price of our
common stock in the five trading days prior to
conversion.
Regulation S Offering
. On September 2,
2014, we accepted a subscription agreement with ITEM Medikal
Teknolojileri LTD STI, a Turkish corporation, referred to as ITEM,
pursuant to we sold 651,042 shares of our common stock and a
warrant to purchase an additional 325,521 shares, for an aggregate
purchase price of $200,000. The warrant is immediately exercisable,
has an exercise price per share of $0.4608, and expires five years
from the date of issuance.
Secured Promissory Note
. On September
10, 2014, we entered into a note purchase agreement with Tonaquint,
Inc., pursuant to which we sold a secured promissory note with an
initial principal amount of $1,275,000, for a purchase price of
$700,000 (an original issue discount of $560,000). We may prepay
the note at any time. The note is secured by our current and future
accounts receivable and inventory, pursuant to a security agreement
entered into in connection with the note purchase agreement. As a
result of a series of amendments to the note (since assigned to two
accredited investors), the maturity date currently is August 31,
2016, and has been accruing interest at a rate of the lesser of 18%
per year or the maximum rate permitted by applicable law. Pursuant
to the amendments, the holders may convert the outstanding balance
into shares of our common stock, at a conversion price per share
equal to the lower of (1) $0.25 and (2) 75% of the lowest daily
volume weighted average price per share of our common stock during
the five business days prior to conversion. If the conversion price
at the time of any conversion request would be lower than $0.15 per
share, we have the option of delivering the conversion amount in
cash in lieu of shares of our common stock.
March 2015 Private Placement
. In March
2015, we entered into subscription agreements with certain
accredited investors, pursuant to which we sold an aggregate of 4.0
million shares of our common stock and warrants to purchase an
additional 2.0 million shares, for an aggregate purchase price of
$720,000.
2015 Series B Warrant Exchanges
. In
order to secure the consent to an amendment to the terms of our
Series B preferred stock, effective June 19, 2015 we agreed with
each Series B holder to reduce the exercise price on certain
“Tranche A” and “Tranche B” warrants,
originally issued to the Series B holders on May 21, 2013. We
reduced the “Tranche A” warrant exercise price per
share from $1.08 to $0.10455, and the “Tranche B”
warrant exercise price per share from $0.10455 to $0.09. We further
agreed to grant the Series B holders the right to participate in
our next capital-raising transaction by exchanging their shares of
Series B preferred stock for the securities to be offered in any
such transaction. Separately, we informed all other holders of
outstanding “Tranche A” and “Tranche B”
warrants that we had lowered the exercise prices per share for
those warrants to $0.10455 and $0.09, respectively.
Public Offering Warrant
Exchanges.
On June 25 and 26, 2015, we entered into
various agreements with holders of the warrants originally issued
in December 2014, pursuant to which each holder separately agreed
to exchange its warrant for two new warrants that, unlike the
original warrant, do not contain any price- or share-reset
provisions. Each new warrant is exercisable for the same number of
shares of our common stock as the original warrant, at any time
beginning December 2, 2015 and ending December 2, 2020. The
exercise price of the first new warrant is $0.09 per share and the
second new warrant is $0.11 per share but, aside from the exercise
price, the new warrants are identical in terms to each other. As
additional consideration, we agreed to issue an aggregate of
approximately 3.1 million shares of our common stock to the
holders, pro rata based on the amount of shares underlying their
original warrants. The holders further agreed to amend to the
securities purchase agreement, dated December 2, 2014 that governed
(among other things) the issuance of the original
warrants.
In connection with
the agreement with one of the holders, Magna Equities II, LLC, on
June 25, 2015 we also agreed to exchange an outstanding senior
convertible note held by Magna for a new note, in stated principal
amount equal to the then-outstanding principal on the original
note. The new note had the same terms as the original note, except
that, among other things, Magna was temporarily prohibited from
converting any of the balance into shares of our common stock. On
June 30, 2015, we paid in full the entire balance of the new
note.
Series C Financing.
On June 29,
2015, we entered into a securities purchase agreement with certain
accredited investors, amended on September 3, 2015, for the
issuance and sale of shares of an aggregate of 7,903 shares of our
Series C preferred stock, at a purchase price of $750 per share and
an initial conversion price of $0.095 per share, and five-year
warrants exercisable to purchase an aggregate of approximately
124.8 million shares of our common stock at an initial exercise
price of $0.095 per share, subject to certain customary adjustments
and anti-dilution provisions.
Senior Secured Convertible Note.
On
February 11, 2016, we entered into a securities purchase agreement
with GPB Debt Holdings II LLC for the issuance and sale on February
12, 2016 of approximately $1.4 million in aggregate principal
amount of a senior secured convertible note for an aggregate
purchase price of $1.15 million (a 20% original issue discount).
The note matures on the second anniversary of issuance and, in
addition to the 20% original issue discount, accrues interest at a
rate of 17% per year. Subject to certain restrictions, it is
convertible at any time, in whole or in part, at the holder’s
option, into shares of our common stock, at a conversion price
equal to $0.80 per share, subject to certain customary adjustments
and anti-dilution provisions. In addition, the investor received a
five-year warrant exercisable to purchase an aggregate of
approximately 1.79 million shares of our common stock with an
exercise price of $0.80 per share, subject to certain customary
adjustments and anti-dilution provisions. On March 17, 2016, our
placement agent for the transaction received a warrant with similar
terms exercisable for 86,250 shares. In connection with the
transaction, on February 12, 2016, we and the investor entered into
a four-year consulting agreement, pursuant to which the investor
will provide management consulting services to us in exchange for a
royalty payment, payable quarterly, equal to 3.5% of our revenues
from the sale of products.
Series C Exchanges.
Between April 27,
2016 and May 3, 2016, we entered into various agreements with
certain holders of our Series C preferred stock, including John
Imhoff, one of our directors, pursuant to which those holders
separately agreed to exchange each share of Series C preferred
stock held for 2.25 shares of our newly created Series C1 preferred
stock and 9,600 shares of our common stock. In connection with
these exchanges, each holder also agreed to exchange the $1,000
stated value per share of the holder’s shares of Series C1
preferred stock for new securities that we separately issue in the
next qualifying financing we undertake on a dollar-for-dollar
basis. This obligation is contingent on all holders of the
then-outstanding Series C preferred stock similarly exchanging
their shares of Series C preferred stock. We do not expect the
remaining holder of our Series C preferred stock to agree to
exchange its shares for shares of Series D preferred stock and
warrants upon consummation of this offering, and therefore the
holders of our Series C1 preferred stock will not be so obligated.
Finally, each holder agreed, except in the event of an additional
$50,000 cash investment by the holder in our next public financing
of at least $1 million, to execute a customary “lockup”
agreement in connection with such financing. In total, for 1,916
shares of Series C preferred stock surrendered, we issued 4,311
shares of Series C1 preferred stock and 22,996 shares of common
stock. The Series C1 preferred stock has terms that are
substantially the same as the Series C preferred stock, except that
the Series C1 preferred stock does not pay dividends (unless and to
the extent declared on the common stock) or at-the-market
“make-whole payments and, while it has the same anti-dilution
protections afforded the Series C preferred stock, it does not
automatically reset in connection with a reverse stock split or
conversion of our outstanding convertible debt.
Separately, on
April 27, 2016, we entered into a rollover and amendment agreement
with another holder of Series C preferred stock, Aquarius
Opportunity Fund, pursuant to which Aquarius agreed to exchange any
shares of Series C preferred stock (stated value plus make-whole
dividend), as well as any remaining principal and accrued interest
on our secured promissory note Aquarius holds, for new securities
that we issue in our next financing, all on a dollar-for-dollar
basis, as long as the next financing involves at least $1 million
in cash from investors unaffiliated with Aquarius. We expect this
offering will be a qualifying financing and, therefore, Aquarius
will be contractually obligated to exchange its shares of Series C
preferred stock and any remaining portion of our convertible note
it holds for shares of Series D preferred stock and warrants
immediately after we consummate this offering. Aquarius also agreed
to return to us for cancelation warrants exercisable for 7,148,813
shares of our common stock that it held. Except in the event of an
additional $50,000 cash investment by Aquarius in the qualifying
financing, Aquarius has agreed to execute a customary
“lockup” agreement in connection with the financing.
Finally, Aquarius, as the holder of a majority of the outstanding
Series C preferred stock, agreed to amend the Series C stock
purchase agreement to eliminate any participation rights held by
the Series C shareholders and to waive operation of certain
anti-dilution provisions of the Series C that would otherwise be
triggered by the transactions described above.
Shenghuo License
Agreement.
On June 5, 2016, we
entered into a license agreement with Shenghuo Medical, LLC
pursuant to which we granted Shenghuo an exclusive license to
manufacture, sell and distribute LuViva in Taiwan, Brunei
Darussalam, Cambodia, Laos, Myanmar, Philippines, Singapore,
Thailand, and Vietnam. Shenghuo was already our exclusive
distributor in China, Macau and Hong Kong, and the license extended
to manufacturing in those countries as well. As partial
consideration for, and as a condition to, the license, and to
further align the strategic interests of the parties, on March 24,
2017 we issued a $300,000 convertible note to Shenghuo, in exchange
for Shenghuoís June 2016 aggregate cash investment of
$200,000. The unsecured note accrues interest at 20% per year,
compounded annually, from January 1, 2017 until repayment, and is
convertible into shares of our common stock at a conversion price
per share of $13.92, subject to customary anti-dilution adjustment.
We also issued Shenghuo a five-year warrant exercisable immediately
for 17,239 shares of common stock at an exercise price equal to the
conversion price of the note, subject to customary anti-dilution
adjustment.
Warrant Restructuring.
Between June 13,
2016 and June 14, 2016, we entered into various agreements with
holders of certain warrants (including John Imhoff, one of our
directors) originally issued in May 2013, and with GPB Debt
Holdings II LLC, holder of a warrant issued February 12, 2016,
pursuant to which each holder separately agreed to exchange
warrants for either (1) shares of common stock equal to 166% of the
number of shares of common stock underlying the surrendered
warrants, or (2) new warrants exercisable for 200% of the number of
shares underlying the surrendered warrants, but without certain
anti-dilution protections included with the surrendered warrants.
As a result of the exchanges, we effectively eliminate any
potential exponential increase in the number of underlying shares
issuable upon exercise of our outstanding warrants. In total, for
surrendered warrants then-exercisable for an aggregate of
94,828,888 shares of common stock (but subject to exponential
increase upon operation of certain anti-dilution provisions), we
issued or are obligated to issue 13,517,342 shares of common stock
and new warrants that, if exercised as of June 14, 2016, would have
been exercisable for an aggregate of 214,189,622 shares of common
stock.
In certain
circumstances, in lieu of presently issuing all of the shares (for
each holder that opted for shares of common stock), we and the
holder further agreed that we will, subject to the terms and
conditions set forth in the applicable warrant exchange agreement,
from time to time, be obligated to issue the remaining shares to
the holder. No additional consideration will be payable in
connection with the issuance of the remaining shares.
The holders that
elected to receive shares for their surrendered warrants have
agreed that they will not sell shares on any trading day in an
amount, in the aggregate, exceeding 20% of the composite aggregate
trading volume of the common stock for that trading
day.
The holders that
elected to receive new warrants will be required to surrender their
old warrants upon consummation of this offering. The new warrants
will have an initial exercise price equal to the exercise price of
the surrendered warrants as of immediately prior to consummation of
this offering, subject to customary “downside price
protection” for as long as our common stock is not listed on
a national securities exchange, and will expire five years from the
date of issuance.
Debt Restructuring.
On December 7, 2016, we entered into an exchange agreement with GPB
Debt Holdings II LLC with regard to the $1,525,000 in outstanding
principal amount of senior secured convertible note originally
issued to GPB on February 11, 2016 (see “—Senior
Secured Convertible Note”), and the $306,863 in outstanding
principal amount of our secured promissory note that GPB holds (see
“—Secured Promissory Note”). Pursuant to the
exchange agreement, upon completion of the next financing resulting
in at least $1 million in cash proceeds, GPB will exchange both
securities for a new convertible note in principal amount of
$1,831,862.50. The new convertible note will mature on the second
anniversary of issuance and will accrue interest at a rate of 19%
per year. We will pay monthly interest coupons and, beginning one
year after issuance, will pay amortized quarterly principal
payments. Subject to resale restrictions under Federal securities
laws and the availability of sufficient authorized but unissued
shares of our common stock, the new convertible note will be
convertible at any time, in whole or in part, at the holder’s
option, into shares of our common stock, at a conversion price
equal to the price offered in the qualifying financing that
triggers the exchange, subject to certain customary adjustments and
anti-dilution provisions contained in the new convertible note. The
new convertible note will include customary event of default
provisions and a default interest rate of the lesser of 21% or the
maximum amount permitted by law. Upon the occurrence of an event of
default, GPB will be entitled to require us to redeem the new
convertible note at 120% of the outstanding principal balance. The
new convertible note will be secured by a lien on all of our
assets, including our intellectual property, pursuant to the
security agreement entered into by us and GPB in connection with
the issuance of the original senior secured convertible note. As an
inducement to GPB to enter into these transactions, we agreed to
increase the royalty payable to GPB pursuant to its consulting
agreement with us from 3.5% to 3.85% of revenues from the sales of
our products. We expect this offering to trigger GPB’s
obligation to exchange.
OID Note.
On December 28, 2016, we
entered into a securities purchase agreement with RedDiamond
Partners LLC for the issuance and sale to RedDiamond of up to
$330,000 in aggregate principal amount of 10% original issuance
discount convertible promissory notes, for an aggregate purchase
price of $300,000. On that date, we issued to RedDiamond a note in
the principal amount of $222,000, for a purchase price of $200,000.
At our option, at any time within 60 days from that issuance, we
may issue another note in the principal amount of $110,000, for a
purchase price of $100,000, subject to customary conditions
contained in the purchase agreement. The notes mature six months
from their date of issuance and, in addition to the 10% original
issue discount, accrue interest at a rateof 10% per year. We may
prepay the notes, in whole or in part, for 115% of outstanding
principal and interest until 30 days from issuance, for 125% of
outstanding principal and interest at any time from 31 to 60 days
from issuance, and for 130% of outstanding principal and interest
at any time from 61 days from issuance until immediately prior to
the maturity date. After six months from the date of issuance
(i.e., if we fail to repay all principal and interest due under the
notes at the maturity date), RedDiamond may convert the notes, at
any time, in whole or in part, into shares of our common stock, at
a conversion price equal to 60% of the lowest volume weighted
average price of our common stock during the 20 trading days prior
to conversion, subject to certain customary adjustments and
anti-dilution provisions contained in the
notes.
SMI License Agreement.
On January 22,
2017, we entered into a license agreement with Shandong Yaohua
Medical Instrument Corporation, or SMI, pursuant to which we
granted SMI an exclusive global license to manufacture the LuViva
device and related disposables (subject to a carve-out for
manufacture in Turkey) and exclusive distribution rights in the
Peoples Republic of China, Macau, Hong Kong and Taiwan. As partial
consideration for, and as a condition to, the license, and to
further align the strategic interests of the parties, we agreed to
issue $1.0 million in shares of our common stock to SMI, in five
installments through October 2017, at a price per share equal to
the lesser of the average closing price for the five days prior to
issuance and $1.25. On March 28, 2017, we amended the license
agreement via a letter agreement to limit SMIís beneficial
ownership of our common stock on any given date to no more than
4.99% of our then-outstanding common stock. In connection with the
letter agreement, we agreed to issue to SMI three warrants, each
exercisable for 15,000 shares of our common stock, to be issued in
conjunction with the next three cash payments by SMI under the
license agreement. Each warrant will be immediately exercisable
(subject to the beneficial ownership limitation), have an exercise
price equal to the lesser of the closing price per share of our
common stock for the average of five consecutive days preceding the
payment by SMI and $1.25 per share, and have a term of five
years.
Convertible Promissory Note.
On
February 13, 2017, we entered into a securities purchase agreement
with Auctus Fund, LLC for the issuance and sale to Auctus of
$170,000 in aggregate principal amount of a 12% convertible
promissory note for an aggregate purchase price of $156,400
(representing a $13,600 original issue discount). On February 13,
2017, we issued the note to Auctus. Pursuant to the purchase
agreement, we also issued to Auctus awarrant exercisable to
purchase an aggregate of 200,000 shares of our common stock. The
warrant is exercisable at any time, at an exercise price per share
equal to $0.77 (110% of the closing price of the common stock on
the day prior to issuance), subjectto certain customary adjustments
and price-protection provisions contained in the warrant. The
warrant has a five-year term. The note matures nine months from the
date of issuance and, in addition to the original issue discount,
accrues interest at a rateof 12% per year. We may prepay the note,
in whole or in part, for 115% of outstanding principal and interest
until 30 days from issuance, for 125% of outstanding principal and
interest at any time from 31 to 60 days from issuance, and for 130%
of outstanding principal and interest at any time from 61 days from
issuance to 180 days from issuance. After six months from the date
of issuance, Auctus may convert the note, at any time, in whole or
in part, into shares of our common stock, at a conversion price
equal to the lower of the price offered in our next public offering
or a 40% discount to the average of the two lowest trading prices
of the common stock during the 20 trading days prior to the
conversion, subject to certain customary adjustments and
price-protection provisions contained in the note. The note
includes customary events of default provisions and a default
interest rate of 24% per year. Upon the occurrence of an event of
default, Auctus may require us to redeem the note (or convert it
into shares of common stock) at 150% of the outstanding principal
balance plus accrued and unpaid interest. In connection with the
transaction, we agreed to reimburse Auctus for $30,000 in legal and
diligence fees, of which we paid $10,000 in cash and $20,000 in
restricted shares of common stock, valued at $0.40 per share (a
42.86% discount to the closing price of the common stock on the day
prior to issuance).
The
sale of securities to ITEM was made to a non-U.S. person in an
offshore offering in accordance with Regulation S under the
Securities Act. The issuances of securities pursuant to the
November 2013 warrant exchange program, the Series B warrant
exchanges, the public offering warrant exchanges, the Series C
exchanges, the warrant restructuring, and the debt restructuring
described above were exempt from registration under the Securities
Act in reliance upon Section 3(a)(9) of the Securities Act as
exchanges with existing securities holders exclusively where no
commission or other remuneration was paid or given directly or
indirectly for soliciting such exchanges. The remaining issuances
of securities described above (as well as the issuances of common
stock upon exercise of warrants received in the warrant exchange
programs) were pursuant to private placements to accredited
investors, and therefore were exempt from registration under the
Securities Act in reliance upon Section 4(2) of the Securities Act
as transactions by an issuer not involving a public offering. The
securities described above are restricted securities for the
purpose of the Securities Act. Any certificates representing the
securities bear a restrictive legend providing that the securities
have not been registered under the Securities Act and cannot be
sold or otherwise transferred without an effective registration or
an exemption therefrom. All cash proceeds from these issuances were
used in product development, working capital and other general
corporate purposes.
ITEM 16. EXHIBITS
(a)
Exhibits
|
EXHIBIT NO.
|
DESCRIPTION
|
|
3.1
|
Restated Certificate of Incorporation, as
amended through November 3, 2016
|
|
3.2
|
Amended and Restated Bylaws (incorporated by
reference to Exhibit 3.1 to the current report on Form 8-K, filed
March 23, 2012)
|
|
3.3**
|
Form of Certificate of Designations (Series
D)
|
|
4.1
|
Specimen Common Stock Certificate (incorporated
by reference to Exhibit 4.1 to the amended registration statement
on Form S-1/A (No. 333-22429) filed April 24, 1997)
|
|
4.2
|
Secured Promissory Note, dated September 10,
2014 (incorporated by reference to Exhibit 4.1 to the current
report on Form 8-K filed September 10, 2014)
|
|
4.3
|
Amendment #1 to Secured Promissory Note, dated
March 10, 2015 (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K filed March 19, 2015)
|
|
4.4
|
Amendment #2 to Secured Promissory Note, dated
May 4, 2015 (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K filed May 7, 2015)
|
|
4.5
|
Amendment #3 to Secured Promissory Note, dated
June 1, 2015 (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K filed June 5, 2015)
|
|
4.6
|
Amendment #4 to Secured Promissory Note, dated
June 16, 2015 (incorporated by reference to Exhibit 10.4 to the
current report on Form 8-K filed June 30, 2015)
|
|
4.7
|
Amendment #5 to Secured Promissory Note, dated
June 29, 2015 (incorporated by reference to Exhibit 10.5 to the
current report on Form 8-K filed June 30, 2015)
|
|
4.8
|
Amendment #6 to Secured Promissory Note, dated
January 20, 2016 (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K filed February 16, 2016)
|
|
4.9
|
Amendment #7 to Secured Promissory Note, dated
February 11, 2016 (incorporated by reference to Exhibit 10.2 to the
current report on Form 8-K filed February 16, 2016)
|
|
4.10
|
Amendment #8 to Secured Promissory Note, dated
March 7, 2016 (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K filed March 7, 2016)
|
|
4.11
|
Senior Secured Convertible Note, dated February
12, 2016 (incorporated by reference to Exhibit 4.1 to the current
report on Form 8-K filed February 12, 2016)
|
|
4.12
|
Form of Exchange Note (GPB) (incorporated by
reference to Exhibit 4.1 to the current report on Form 8-K filed
December 7, 2016)
|
|
4.13
|
10% OID Convertible Promissory Note
(incorporated by reference to Exhibit 4.1 to the current report on
Form 8-K filed December 30, 2016)
|
|
4
.14
|
Convertible
Promissory Note (incorporated by reference to Exhibit 4.1 to the
current report on Form 8-K filed February 16,
2017)
|
|
4.15
|
Form of Warrant (Standard Form) (incorporated by
reference to Exhibit 4.1 to the current report on Form 8-K, filed
September 14, 2010)
|
|
4.16
|
Form of Warrant (InterScan) (incorporated by
reference to Exhibit 4.13 to the annual report on Form 10-K for the
year ended December 31, 2013, filed March 27, 2014)
|
|
4.17
|
Form of Warrant (November 2011 Private
Placement) (incorporated by reference to Exhibit 4.1 to the current
report on Form 8-K/A, filed November 28, 2011)
|
|
4.18
|
Form of Warrant (Series B-Tranche A)
(incorporated by reference to Exhibit 10.2 to amendment no. 1 to
the current report on Form 8-K, filed May 23, 2013)
|
|
4.19
|
Form of Warrant (Series B-Tranche B)
(incorporated by reference to Exhibit 10.3 to amendment no. 1 to
the current report on Form 8-K, filed May 23, 2013)
|
|
4.20
|
Form of Warrant (Regulation S) (incorporated by
reference to Exhibit 4.1 to the current report on Form 8-K, filed
September 8, 2014)
|
|
4.21
|
Form of Warrant (2014 Public Offering Placement
Agent) (incorporated by reference to Exhibit 4.2 to the current
report on Form 8-K filed December 4, 2014)
|
|
4.22
|
Form of Warrant (2014 Public Offering Warrant
Exchanges) (incorporated by reference to Exhibit 4.1 to the current
report on Form 8-K filed June 30, 2015)
|
|
4.23
|
Form of Warrant (Series C) (incorporated by
reference to Exhibit 4.3 to the current report on Form 8-K filed
June 30, 2015)
|
|
4.24
|
Form of Warrant (Senior Secured Convertible
Note) (incorporated by reference to Exhibit 10.5 to the current
report on Form 8-K filed February 12, 2016)
|
|
4.25
|
Form of Warrant (Series B-Tranche B Exchanges;
GPB Exchange) (incorporated by reference to Exhibit 4.1 to the
current report on Form 8-K filed June 14, 2016)
|
|
4
.26
|
Common Stock
Purchase Warrant (Convertible Promissory Note) (incorporated by
reference to Exhibit 4.2 to the current report on Form 8-K filed
February 16, 2017)
|
|
4.27**
|
Form of Warrant (Series D)
|
|
5.1**
|
Opinion of Jones Day regarding
validity
|
|
10.1
|
1995 Stock Plan and form of Stock Option
Agreement (incorporated by reference to Exhibit 10.2 to the
registration statement on Form S-1 (No. 333-22429) filed February
27, 1997)
|
|
|
10.2
|
2005 Amendment to 1995 Stock Plan (incorporated
by reference to Appendix 1 to the proxy statement on Schedule 14A,
filed May 10, 2005)
|
|
10.3
|
2010 Amendment to 1995 Stock Plan (incorporated
by reference to Exhibit 10.3 to the registration statement on Form
S-8 (File No. 333-178261), filed December 1, 2011)
|
|
10.4
|
2012 Amendment to 1995 Stock Plan (incorporated
by reference to Annex 1 to the proxy statement on Schedule 14A,
filed April 30, 2012)
|
|
10.5
|
Agreement and Release, dated August 30, 2011
(incorporated by reference to 10.2 to the current report on Form
8-K, filed September 2, 2011)
|
|
10.6
|
Employment Agreement between the Company and
Mark Faupel dated March 24, 2013 (incorporated by reference to
Exhibit 10.10 to the annual report on Form 10-K for the year ended
December 31, 2013, filed March 27, 2014)
|
|
10.7
|
Employment Agreement between the Company and
Gene Cartwright, dated January 6, 2014 (incorporated by reference
to Exhibit 10.11 to the annual report on Form 10-K for the year
ended December 31, 2013, filed March 27, 2014).
|
|
10.8
|
Employment Agreement between the Company and
Rick L. Fowler, automatically renewed on May 9, 2013 (incorporated
by reference to Exhibit 10.12 to the annual report on Form 10-K for
the year ended December 31, 2013, filed March 27,
2014)
|
|
10.9
|
Consulting Agreement between the Company and GPB
Debt Holdings II LLC, dated February 12, 2016 (incorporated by
reference to Exhibit 10.6 to the current report on Form 8-K filed
February 12, 2016)
|
|
10.10
|
Securities Purchase Agreement (Magna Note),
dated April 23, 2014, by and between the Company and Hanover
Holdings I, LLC (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K, filed April 24, 2014).
|
|
10.11
|
Registration Rights Agreement (Magna Note),
dated April 23, 2014, by and between the Company and Hanover
Holdings I, LLC (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K, filed April 24, 2014)
|
|
10.12
|
Standstill Agreement (Magna Note), dated as of
November 6, 2014, by and between the Company and Magna Equities II,
LLC (incorporated by reference to Exhibit 19 to the registration
statement on Form S-1 (No. 333-198733) filed November 10,
2014)
|
|
10.13
|
Exchange Agreement (Magna Note), dated as of
June 25, 2015 (incorporated by reference to Exhibit 10.3 to the
current report on Form 8-K filed June 30, 2015)
|
|
10.14
|
Subscription Agreement (Regulation S), accepted
September 2, 2014 (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K, filed September 8, 2014)
|
|
10.15
|
Form of Registration Rights Agreement
(Regulation S), dated September 8, 2014 by and between the Company
and the investor party thereto (incorporated by reference to
Exhibit 10.2 to the current report on Form 8-K, filed September 8,
2014)
|
|
10.16
|
Note Purchase Agreement (Secured Promissory
Note), dated as of September 10, 2014, by and between the Company
and Tonaquint, Inc. (incorporated by reference to Exhibit 10.1 to
the current report on Form 8-K, filed September 10,
2014)
|
|
10.17
|
Security Agreement (Secured Promissory Note),
dated as of September 10, 2014, by the Company and Tonaquint, Inc.
(incorporated by reference to Exhibit 10.2 to the current report on
Form 8-K, filed September 10, 2014)
|
|
10.18
|
Form of Securities Purchase Agreement (2014
Public Offering) (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K filed December 4, 2014)
|
|
10.19
|
Placement Agent Agreement (2014 Public
Offering), by and between the Company and Olympus Securities, LLC
(incorporated by reference to Exhibit 10.2 to the current report on
Form 8-K filed December 4, 2014)
|
|
10.20
|
Amendment to Securities Purchase Agreement (2014
Public Offering), dated as of June 26, 2015 (incorporated by
reference to Exhibit 10.2 to the current report on Form 8-K filed
June 30, 2015)
|
|
10.21
|
Form of Letter Agreement (2014 Public Offering
Warrant Exchanges) (incorporated by reference to Exhibit 10.1 to
the current report on Form 8-K filed June 30, 2015)
|
|
10.22
|
Securities Purchase Agreement (Series C), dated
June 29, 2015 (incorporated by reference to Exhibit 10.6 to the
current report on Form 8-K filed June 30, 2015)
|
|
10.23
|
Registration Rights Agreement (Series C), dated
June 29, 2015 (incorporated by reference to Exhibit 10.7 to the
current report on Form 8-K filed June 30, 2015)
|
|
10.24
|
Form of Joinder Agreement (Series C)
(incorporated by reference to Exhibit 10.1 to the current report on
Form 8-K filed July 13, 2015)
|
|
10.25
|
Interim Securities Purchase Agreement (Series
C), dated September 3, 2015 (incorporated by reference to Exhibit
10.1 to the current report on Form 8-K filed September 3,
2015)
|
|
10.26
|
Securities Purchase Agreement (Senior Secured
Convertible Note), dated February 11, 2016 (incorporated by
reference to Exhibit 10.3 to the current report on Form 8-K filed
February 12, 2016)
|
|
10.27
|
Security Agreement (Senior Secured Convertible
Note), dated February 11, 2016 (incorporated by reference to
Exhibit 10.4 to the current report on Form 8-K filed February 12,
2016)
|
|
10.28
|
Rollover and Amendment Agreement, dated April
27, 2016, by and between the Company and Aquarius Opportunity Fund
(incorporated by reference to Exhibit 10.2 to the current report on
Form 8-K filed May 3, 2016)
|
|
10.29
|
Form of Letter Agreement (Series C Exchanges)
(incorporated by reference to Exhibit 10.1 to the current report on
Form 8-K filed May 3, 2016)
|
|
10.30
|
License Agreement, dated June 5, 2016
(incorporated by reference to Exhibit 10.1 to the current report on
Form 8-K filed June 8, 2016)
|
|
10.31
|
Form of Warrant Exchange Agreement
(Warrant-for-Shares) (incorporated by reference to Exhibit 10.1 to
the current report on Form 8-K filed June 14, 2016)
|
|
10.32
|
Form of Warrant Exchange Agreement
(Warrant-for-Warrant) (incorporated by reference to Exhibit 10.2 to
the current report on Form 8-K filed June 14, 2016)
|
|
10.33
|
Royalty Agreement, dated September 6, 2016,
between the Company and Imhoff and Maloof (incorporated by
reference to Exhibit 10.1 to the current report on Form 8-K filed
September 8, 2016)
|
|
10.34
|
Lockup and Exchange Agreement, dated November 2,
2016, by the Company and GHS Investments, LLC (incorporated by
reference to Exhibit 10.1 to the current report on Form 8-K filed
November 2, 2016)
|
|
10.35
|
Exchange Agreement, dated December 7, 2016,
between the Company and GPB Debt Holdings II LLC (incorporated by
reference to Exhibit 10.1 to the current report on Form 8-K filed
December 7, 2016)
|
|
10.36
|
Amendment to Consulting Agreement, dated
December 7, 2016, between the Company and GPB Debt Holdings II LLC
(incorporated by reference to Exhibit 10.2 to the current report on
Form 8-K filed December 7, 2016)
|
|
10.37
|
Securities Purchase Agreement, dated December
28, 2016, between the Company and RedDiamond (incorporated by
reference to Exhibit 10.1 to the current report on Form 8-K filed
December 30, 2016)
|
|
10.38
|
Agreement between
Shandong Yaohua Medical Instrument Corporation and Guided
Therapeutics, Inc., Confidential, Final 22 January 2017
(incorporated by reference to Exhibit 10.1 to the amended current
report on Form 8-K/A filed March 24,
2017)
|
|
10.39
|
Guided
Therapeutics-Shenghuo Medical Agreement, 22 Jan 2017 (incorporated
by reference to Exhibit 10.2 to the current report on Form 8-K
filed January 26, 2017)
|
|
10.40
|
Letter Agreement,
dated March 28, 2017, between Shandong Yaohua Medical Instrument
Corporation and Guided Therapeutics, Inc. (incorporated by
reference to Exhibit 10.1 to the current report on Form 8-K filed
March 28, 2017)
|
|
10.41**
|
Securities Purchase
Agreement, dated as of February 13, 2017, by and between Guided
Therapeutics, Inc. and Auctus Fund, LLC (incorporated by reference
to Exhibit 10.2 to the current report on Form 8-K filed February
16, 2017)
|
|
1
0.42**
|
Form of Securities
Purchase Agreement
|
|
21.1
|
Subsidiaries (incorporated by reference to
Exhibit 21.1 to the registration statement on Form S-1 (No.
333-169755) filed October 5, 2010)
|
|
23.1*
|
Consent of UHY LLP.
|
|
23.2**
|
Consent of Jones Day (included in Exhibit
5.1).
|
|
24.1
|
Powers of Attorney (included at signature
page).
|
|
101.1*
|
Interactive Data File
|
|
*Filed herewith
|
|
**To be filed via pre-effective
amendment
|
ITEM
17. UNDERTAKINGS
(a)
The undersigned registrant hereby
undertakes:
(1)
To file, during any period in which
offers or sales are being made, a post-effective amendment to this
registration statement:
(i)
To include any prospectus required by
section 10(a)(3) of the Securities Act of 1933;
(ii)
To reflect in the prospectus any
facts or events arising after the effective date of the
registration statement (or the most recent post-effective amendment
thereof) which, individually or in the aggregate, represent a
fundamental change in the information set forth in the registration
statement. Notwithstanding the foregoing, any increase or decrease
in volume of securities offered (if the total dollar value of
securities offered would not exceed that which was registered) and
any deviation from the low or high end of the estimated maximum
offering range may be reflected in the form of prospectus filed
with the Commission pursuant to Rule 424(b) if, in the aggregate,
the changes in volume and price represent no more than 20% change
in the maximum aggregate offering price set forth in the
“Calculation of Registration Fee” table in the
effective registration statement;
(iii)
To include any material information
with respect to the plan of distribution not previously disclosed
in the registration statement or any material change to such
information in the registration statement.
(2)
That, for the purpose of determining
any liability under the Securities Act of 1933, each such
post-effective amendment shall be deemed to be a new registration
statement relating to the securities offered therein, and the
offering of such securities at that time shall be deemed to be the
initial
bona fide
offering
thereof.
(3) To remove from
registration by means of a post-effective amendment any of the
securities being registered which remain unsold at the termination
of the offering.
(4)
That, for the purpose of determining
liability of the registrant under the Securities Act of 1933 to any
purchaser in the initial distribution of the securities: The
undersigned registrant undertakes that in a primary offering of
securities of the undersigned registrant pursuant to this
registration statement, regardless of the underwriting method used
to sell the securities to the purchaser, if the securities are
offered or sold to such purchaser by means of any of the following
communications, the undersigned registrant will be a seller to the
purchaser and will be considered to offer or sell such securities
to such purchaser:
(i)
Any preliminary prospectus or
prospectus of the undersigned registrant relating to the offering
required to be filed pursuant to Rule 424;
(ii)
Any free writing prospectus relating
to the offering prepared by or on behalf of the undersigned
registrant or used or referred to by the undersigned
registrant;
(iii)
The portion of any other free writing
prospectus relating to the offering containing material information
about the undersigned registrant or its securities provided by or
on behalf of the undersigned registrant; and
(iv)
Any other communication that is an
offer in the offering made by the undersigned registrant to the
purchaser.
(5)
That, for purposes of determining any
liability under the Securities Act of 1933, the information omitted
from the form of prospectus filed as part of this registration
statement in reliance upon Rule 430A and contained in a form of
prospectus filed by the registrant pursuant to Rule 424(b) (1) or
(4) or 497(h) under the Securities Act shall be deemed to be part
of this registration statement as of the time it was declared
effective.
(6) That, for the
purpose of determining any liability under the Securities Act of
1933, each post-effective amendment that contains a form of
prospectus shall be deemed to be a new registration statement
relating to the securities offered therein, and the offering of
such securities at that time shall be deemed to be the initial bona
fide offering thereof.
(b)
Insofar as indemnification for
liabilities arising under the Securities Act of 1933 may be
permitted to directors, officers and controlling persons of the
registrant pursuant to the foregoing provisions, or otherwise, the
registrant has been advised that in the opinion of the Securities
and Exchange Commission such indemnification is against public
policy as expressed in the Act and is, therefore, unenforceable. In
the event that a claim for indemnification against such liabilities
(other than the payment by the registrant of expenses incurred or
paid by a director, officer or controlling person of the registrant
in the successful defense of any action, suit or proceeding) is
asserted by such director, officer or controlling person in
connection with the securities being registered, the registrant
will, unless in the opinion of its counsel the matter has been
settled by controlling precedent, submit to a court of appropriate
jurisdiction the question whether such indemnification by it is
against public policy as expressed in the Act and will be governed
by the final adjudication of such issue.
SIGNATURES
Pursuant to the
requirements of the Securities Act of 1933, the registrant has duly
caused this registration statement to be signed on its behalf by
the undersigned, thereunto duly authorized in the City of Norcross,
State of Georgia, on
March 30
,
2017.
|
|
GUIDED THERAPEUTICS, INC.
|
|
|
|
|
|
By:
|
/s/
Gene S. Cartwright
|
|
|
President, Chief Executive Officer and
Acting
Chief Financial Officer
|
Pursuant to the
requirements of the Securities Act of 1933, this registration
statement has been signed by the following persons in the
capacities and on the dates indicated:
|
DATE
|
|
SIGNATURE
|
|
TITLE
|
|
|
|
|
|
|
|
March 30, 2017
|
|
/s/ Gene S.
Cartwright
|
|
President, Chief Executive Officer, Acting Chief
Financial Officer (Principal Executive Officer and Principal
Financial and Accounting Officer)
|
|
Gene S. Cartwright
|
|
|
|
|
|
|
|
March
30
, 2017
|
|
*
|
|
Chairman of the Board and Director
|
|
|
|
Michael C. James
|
|
|
|
|
|
|
|
|
|
March
30
, 2017
|
|
*
|
|
Director
|
|
|
|
John E. Imhoff
|
|
|
|
|
|
|
|
|
|
March
30
, 2017
|
|
*
|
|
Director
|
|
|
|
Richard P. Blumberg
|
|
|
|
|
|
|
|
|
|
March
30
, 2017
|
|
*
|
|
Chief Operating Officer
and Director
|
|
|
|
Mark Faupel
|
|
|
*By
/s/ Gene S. Cartwright
Gene S. Cartwright,
Attorney-in-Fact
March
30
, 2017
EXHIBIT INDEX
|
EXHIBIT NO.
|
DESCRIPTION
|
|
3.1
|
Restated Certificate of Incorporation, as
amended through November 3, 2016
|
|
3.2
|
Amended and Restated Bylaws (incorporated by
reference to Exhibit 3.1 to the current report on Form 8-K, filed
March 23, 2012)
|
|
3.3**
|
Form of Certificate of Designations (Series
D)
|
|
4.1
|
Specimen Common Stock Certificate (incorporated
by reference to Exhibit 4.1 to the amended registration statement
on Form S-1/A (No. 333-22429) filed April 24, 1997)
|
|
4.2
|
Secured Promissory Note, dated September 10,
2014 (incorporated by reference to Exhibit 4.1 to the current
report on Form 8-K filed September 10, 2014)
|
|
4.3
|
Amendment #1 to Secured Promissory Note, dated
March 10, 2015 (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K filed March 19, 2015)
|
|
4.4
|
Amendment #2 to Secured Promissory Note, dated
May 4, 2015 (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K filed May 7, 2015)
|
|
4.5
|
Amendment #3 to Secured Promissory Note, dated
June 1, 2015 (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K filed June 5, 2015)
|
|
4.6
|
Amendment #4 to Secured Promissory Note, dated
June 16, 2015 (incorporated by reference to Exhibit 10.4 to the
current report on Form 8-K filed June 30, 2015)
|
|
4.7
|
Amendment #5 to Secured Promissory Note, dated
June 29, 2015 (incorporated by reference to Exhibit 10.5 to the
current report on Form 8-K filed June 30, 2015)
|
|
4.8
|
Amendment #6 to Secured Promissory Note, dated
January 20, 2016 (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K filed February 16, 2016)
|
|
4.9
|
Amendment #7 to Secured Promissory Note, dated
February 11, 2016 (incorporated by reference to Exhibit 10.2 to the
current report on Form 8-K filed February 16, 2016)
|
|
4.10
|
Amendment #8 to Secured Promissory Note, dated
March 7, 2016 (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K filed March 7, 2016)
|
|
4.11
|
Senior Secured Convertible Note, dated February
12, 2016 (incorporated by reference to Exhibit 4.1 to the current
report on Form 8-K filed February 12, 2016)
|
|
4.12
|
Form of Exchange Note (GPB) (incorporated by
reference to Exhibit 4.1 to the current report on Form 8-K filed
December 7, 2016)
|
|
4.13
|
10% OID Convertible Promissory Note
(incorporated by reference to Exhibit 4.1 to the current report on
Form 8-K filed December 30, 2016)
|
|
4
.14
|
Convertible
Promissory Note (incorporated by reference to Exhibit 4.1 to the
current report on Form 8-K filed February 16,
2017)
|
|
4.15
|
Form of Warrant (Standard Form) (incorporated by
reference to Exhibit 4.1 to the current report on Form 8-K, filed
September 14, 2010)
|
|
4.16
|
Form of Warrant (InterScan) (incorporated by
reference to Exhibit 4.13 to the annual report on Form 10-K for the
year ended December 31, 2013, filed March 27, 2014)
|
|
4.17
|
Form of Warrant (November 2011 Private
Placement) (incorporated by reference to Exhibit 4.1 to the current
report on Form 8-K/A, filed November 28, 2011)
|
|
4.18
|
Form of Warrant (Series B-Tranche A)
(incorporated by reference to Exhibit 10.2 to amendment no. 1 to
the current report on Form 8-K, filed May 23, 2013)
|
|
4.19
|
Form of Warrant (Series B-Tranche B)
(incorporated by reference to Exhibit 10.3 to amendment no. 1 to
the current report on Form 8-K, filed May 23, 2013)
|
|
4.20
|
Form of Warrant (Regulation S) (incorporated by
reference to Exhibit 4.1 to the current report on Form 8-K, filed
September 8, 2014)
|
|
4.21
|
Form of Warrant (2014 Public Offering Placement
Agent) (incorporated by reference to Exhibit 4.2 to the current
report on Form 8-K filed December 4, 2014)
|
|
4.22
|
Form of Warrant (2014 Public Offering Warrant
Exchanges) (incorporated by reference to Exhibit 4.1 to the current
report on Form 8-K filed June 30, 2015)
|
|
4.23
|
Form of Warrant (Series C) (incorporated by
reference to Exhibit 4.3 to the current report on Form 8-K filed
June 30, 2015)
|
|
4.24
|
Form of Warrant (Senior Secured Convertible
Note) (incorporated by reference to Exhibit 10.5 to the current
report on Form 8-K filed February 12, 2016)
|
|
4.25
|
Form of Warrant (Series B-Tranche B Exchanges;
GPB Exchange) (incorporated by reference to Exhibit 4.1 to the
current report on Form 8-K filed June 14, 2016)
|
|
4
.26
|
Common Stock
Purchase Warrant (Convertible Promissory Note) (incorporated by
reference to Exhibit 4.2 to the current report on Form 8-K filed
February 16, 2017)
|
|
4.27**
|
Form of Warrant (Series D)
|
|
5.1**
|
Opinion of Jones Day regarding
validity
|
|
10.1
|
1995 Stock Plan and form of Stock Option
Agreement (incorporated by reference to Exhibit 10.2 to the
registration statement on Form S-1 (No. 333-22429) filed February
27, 1997)
|
|
|
10.2
|
2005 Amendment to 1995 Stock Plan (incorporated
by reference to Appendix 1 to the proxy statement on Schedule 14A,
filed May 10, 2005)
|
|
10.3
|
2010 Amendment to 1995 Stock Plan (incorporated
by reference to Exhibit 10.3 to the registration statement on Form
S-8 (File No. 333-178261), filed December 1, 2011)
|
|
10.4
|
2012 Amendment to 1995 Stock Plan (incorporated
by reference to Annex 1 to the proxy statement on Schedule 14A,
filed April 30, 2012)
|
|
10.5
|
Agreement and Release, dated August 30, 2011
(incorporated by reference to 10.2 to the current report on Form
8-K, filed September 2, 2011)
|
|
10.6
|
Employment Agreement between the Company and
Mark Faupel dated March 24, 2013 (incorporated by reference to
Exhibit 10.10 to the annual report on Form 10-K for the year ended
December 31, 2013, filed March 27, 2014)
|
|
10.7
|
Employment Agreement between the Company and
Gene Cartwright, dated January 6, 2014 (incorporated by reference
to Exhibit 10.11 to the annual report on Form 10-K for the year
ended December 31, 2013, filed March 27, 2014).
|
|
10.8
|
Employment Agreement between the Company and
Rick L. Fowler, automatically renewed on May 9, 2013 (incorporated
by reference to Exhibit 10.12 to the annual report on Form 10-K for
the year ended December 31, 2013, filed March 27,
2014)
|
|
10.9
|
Consulting Agreement between the Company and GPB
Debt Holdings II LLC, dated February 12, 2016 (incorporated by
reference to Exhibit 10.6 to the current report on Form 8-K filed
February 12, 2016)
|
|
10.10
|
Securities Purchase Agreement (Magna Note),
dated April 23, 2014, by and between the Company and Hanover
Holdings I, LLC (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K, filed April 24, 2014).
|
|
10.11
|
Registration Rights Agreement (Magna Note),
dated April 23, 2014, by and between the Company and Hanover
Holdings I, LLC (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K, filed April 24, 2014)
|
|
10.12
|
Standstill Agreement (Magna Note), dated as of
November 6, 2014, by and between the Company and Magna Equities II,
LLC (incorporated by reference to Exhibit 19 to the registration
statement on Form S-1 (No. 333-198733) filed November 10,
2014)
|
|
10.13
|
Exchange Agreement (Magna Note), dated as of
June 25, 2015 (incorporated by reference to Exhibit 10.3 to the
current report on Form 8-K filed June 30, 2015)
|
|
10.14
|
Subscription Agreement (Regulation S), accepted
September 2, 2014 (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K, filed September 8, 2014)
|
|
10.15
|
Form of Registration Rights Agreement
(Regulation S), dated September 8, 2014 by and between the Company
and the investor party thereto (incorporated by reference to
Exhibit 10.2 to the current report on Form 8-K, filed September 8,
2014)
|
|
10.16
|
Note Purchase Agreement (Secured Promissory
Note), dated as of September 10, 2014, by and between the Company
and Tonaquint, Inc. (incorporated by reference to Exhibit 10.1 to
the current report on Form 8-K, filed September 10,
2014)
|
|
10.17
|
Security Agreement (Secured Promissory Note),
dated as of September 10, 2014, by the Company and Tonaquint, Inc.
(incorporated by reference to Exhibit 10.2 to the current report on
Form 8-K, filed September 10, 2014)
|
|
10.18
|
Form of Securities Purchase Agreement (2014
Public Offering) (incorporated by reference to Exhibit 10.1 to the
current report on Form 8-K filed December 4, 2014)
|
|
10.19
|
Placement Agent Agreement (2014 Public
Offering), by and between the Company and Olympus Securities, LLC
(incorporated by reference to Exhibit 10.2 to the current report on
Form 8-K filed December 4, 2014)
|
|
10.20
|
Amendment to Securities Purchase Agreement (2014
Public Offering), dated as of June 26, 2015 (incorporated by
reference to Exhibit 10.2 to the current report on Form 8-K filed
June 30, 2015)
|
|
10.21
|
Form of Letter Agreement (2014 Public Offering
Warrant Exchanges) (incorporated by reference to Exhibit 10.1 to
the current report on Form 8-K filed June 30, 2015)
|
|
10.22
|
Securities Purchase Agreement (Series C), dated
June 29, 2015 (incorporated by reference to Exhibit 10.6 to the
current report on Form 8-K filed June 30, 2015)
|
|
10.23
|
Registration Rights Agreement (Series C), dated
June 29, 2015 (incorporated by reference to Exhibit 10.7 to the
current report on Form 8-K filed June 30, 2015)
|
|
10.24
|
Form of Joinder Agreement (Series C)
(incorporated by reference to Exhibit 10.1 to the current report on
Form 8-K filed July 13, 2015)
|
|
10.25
|
Interim Securities Purchase Agreement (Series
C), dated September 3, 2015 (incorporated by reference to Exhibit
10.1 to the current report on Form 8-K filed September 3,
2015)
|
|
10.26
|
Securities Purchase Agreement (Senior Secured
Convertible Note), dated February 11, 2016 (incorporated by
reference to Exhibit 10.3 to the current report on Form 8-K filed
February 12, 2016)
|
|
10.27
|
Security Agreement (Senior Secured Convertible
Note), dated February 11, 2016 (incorporated by reference to
Exhibit 10.4 to the current report on Form 8-K filed February 12,
2016)
|
|
10.28
|
Rollover and Amendment Agreement, dated April
27, 2016, by and between the Company and Aquarius Opportunity Fund
(incorporated by reference to Exhibit 10.2 to the current report on
Form 8-K filed May 3, 2016)
|
|
10.29
|
Form of Letter Agreement (Series C Exchanges)
(incorporated by reference to Exhibit 10.1 to the current report on
Form 8-K filed May 3, 2016)
|
|
10.30
|
License Agreement, dated June 5, 2016
(incorporated by reference to Exhibit 10.1 to the current report on
Form 8-K filed June 8, 2016)
|
|
10.31
|
Form of Warrant Exchange Agreement
(Warrant-for-Shares) (incorporated by reference to Exhibit 10.1 to
the current report on Form 8-K filed June 14, 2016)
|
|
10.32
|
Form of Warrant Exchange Agreement
(Warrant-for-Warrant) (incorporated by reference to Exhibit 10.2 to
the current report on Form 8-K filed June 14, 2016)
|
|
10.33
|
Royalty Agreement, dated September 6, 2016,
between the Company and Imhoff and Maloof (incorporated by
reference to Exhibit 10.1 to the current report on Form 8-K filed
September 8, 2016)
|
|
10.34
|
Lockup and Exchange Agreement, dated November 2,
2016, by the Company and GHS Investments, LLC (incorporated by
reference to Exhibit 10.1 to the current report on Form 8-K filed
November 2, 2016)
|
|
10.35
|
Exchange Agreement, dated December 7, 2016,
between the Company and GPB Debt Holdings II LLC (incorporated by
reference to Exhibit 10.1 to the current report on Form 8-K filed
December 7, 2016)
|
|
10.36
|
Amendment to Consulting Agreement, dated
December 7, 2016, between the Company and GPB Debt Holdings II LLC
(incorporated by reference to Exhibit 10.2 to the current report on
Form 8-K filed December 7, 2016)
|
|
10.37
|
Securities Purchase Agreement, dated December
28, 2016, between the Company and RedDiamond (incorporated by
reference to Exhibit 10.1 to the current report on Form 8-K filed
December 30, 2016)
|
|
10.38
|
Agreement between
Shandong Yaohua Medical Instrument Corporation and Guided
Therapeutics, Inc., Confidential, Final 22 January 2017
(incorporated by reference to Exhibit 10.1 to the amended current
report on Form 8-K/A filed March 24,
2017)
|
|
10.39
|
Guided
Therapeutics-Shenghuo Medical Agreement, 22 Jan 2017 (incorporated
by reference to Exhibit 10.2 to the current report on Form 8-K
filed January 26, 2017)
|
|
10.40
|
Letter Agreement,
dated March 28, 2017, between Shandong Yaohua Medical Instrument
Corporation and Guided Therapeutics, Inc. (incorporated by
reference to Exhibit 10.1 to the current report on Form 8-K filed
March 28, 2017)
|
|
10.41**
|
Securities Purchase
Agreement, dated as of February 13, 2017, by and between Guided
Therapeutics, Inc. and Auctus Fund, LLC (incorporated by reference
to Exhibit 10.2 to the current report on Form 8-K filed February
16, 2017)
|
|
1
0.42**
|
Form of Securities
Purchase Agreement
|
|
21.1
|
Subsidiaries (incorporated by reference to
Exhibit 21.1 to the registration statement on Form S-1 (No.
333-169755) filed October 5, 2010)
|
|
23.1*
|
Consent of UHY LLP.
|
|
23.2**
|
Consent of Jones Day (included in Exhibit
5.1).
|
|
24.1
|
Powers of Attorney (included at signature
page).
|
|
101.1*
|
Interactive Data File
|
|
*Filed herewith
|
|
**To be filed via pre-effective
amendment
|
Guided Therapeutics (QB) (USOTC:GTHP)
Historical Stock Chart
From Aug 2024 to Sep 2024
Guided Therapeutics (QB) (USOTC:GTHP)
Historical Stock Chart
From Sep 2023 to Sep 2024