Filed Pursuant to Rule 424(b)(5)
Registration No. 333-199093
PROSPECTUS SUPPLEMENT
(To Prospectus Dated October 14, 2014)

Up to $20,000,000
Common Stock
We have entered into a Controlled Equity Offering
SM
Sales Agreement (the “Sales Agreement”) with Cantor Fitzgerald & Co. (“Cantor Fitzgerald”) relating
to shares of our common stock offered by this prospectus supplement. In accordance with the terms of the Sales Agreement, we may
offer and sell shares of our common stock having an aggregate offering price of up to $20,000,000 from time to time through Cantor
Fitzgerald, acting as sales agent.
Our common stock is listed on the NASDAQ Capital Market under the
symbol “LPCN.” On March 3, 2017, the last reported sale price of our common stock on the NASDAQ Capital Market was
$4.00 per share.
Sales of our common stock, if any, under this prospectus supplement
may be made in sales deemed to be an “at the market offering” as defined in Rule 415(a)(4) promulgated under the Securities
Act of 1933, as amended (the “Securities Act”), including sales made directly on or through the NASDAQ Capital Market
or on any other existing trading market for our common stock. Cantor Fitzgerald has agreed to act as sales agent on a best efforts
basis and use commercially reasonable efforts to sell on our behalf all of the shares of common stock requested to be sold by us,
consistent with its normal trading and sales practices, on mutually agreed terms between Cantor Fitzgerald and us. There is no
arrangement for funds to be received in any escrow, trust or similar arrangement.
The compensation to Cantor Fitzgerald for sales of common stock
sold pursuant to the Sales Agreement will be at a fixed commission rate of 3.0% of the gross proceeds of any shares of common stock
sold under the Sales Agreement. In connection with the sale of the common stock on our behalf, Cantor Fitzgerald will be deemed
to be an “underwriter” within the meaning of the Securities Act and the compensation of Cantor Fitzgerald will be deemed
to be underwriting commissions or discounts.
As of the date of this prospectus supplement, the aggregate market
value of our common stock held by non-affiliates was approximately $70,812,827, based on 18,594,889 shares of common stock outstanding,
of which 17,398,729 shares were held by non-affiliates, and a per share price of $4.07 based on the closing sale price of our common
stock on March 2, 2017. We have not sold any securities pursuant to General Instruction I.B.6. of Form S-3 during the prior 12
calendar month period that ends on, and includes, the date of this prospectus supplement.
Investing in our securities involves a high degree of risk. See
“Risk Factors” beginning on page S-11 of this prospectus supplement, “Risk Factors” beginning on page 7
of the accompanying prospectus, the “Risk Factors” section beginning on page 17 of our Annual Report on Form 10-K for
the year ended December 31, 2016, and under similar headings in the other documents that are filed after the date hereof and incorporated
by reference into this prospectus supplement and accompanying prospectus.
Neither the Securities and Exchange Commission nor any state
securities commission has approved or disapproved of these securities or passed upon the adequacy or accuracy of this prospectus
supplement or the accompanying prospectus. Any representation to the contrary is a criminal offense.

The date of this prospectus supplement
is March 6, 2017.
TABLE OF CONTENTS
Prospectus Supplement
Prospectus
ABOUT THIS PROSPECTUS SUPPLEMENT
References in this prospectus supplement, the
accompanying prospectus and the documents incorporated by reference to the “Company,” “Lipocine,” “our,”
“us” or “we” refer to Lipocine Inc. and its subsidiaries.
This document is in two parts. The first part
is this prospectus supplement, which describes the specific terms of this offering and certain other matters. The second part,
the accompanying prospectus, provides more general information about us and our common stock. Generally, when we refer to the prospectus,
we are referring to both parts of this document combined. To the extent information in this prospectus supplement conflicts with
information in the accompanying prospectus, you should rely on the information in this prospectus supplement. You should rely only
on the information contained in, or incorporated by reference in, this prospectus supplement and the accompanying prospectus and
any related “free writing prospectus.” None of the Company or the underwriters has authorized anyone to provide information
different from that contained in, incorporated or deemed incorporated by reference into this prospectus supplement or the accompanying
prospectus.
We note
that the representations, warranties and covenants made by us in any agreement that is filed as an exhibit to any document that
is incorporated by reference herein were made solely for the benefit of the parties to such agreement, including, in some cases,
for the purpose of allocating risk among the parties to such agreement, and should not be deemed to be a representation, warranty
or covenant to you. Moreover, such representations, warranties or covenants were accurate only as of the date when made. Accordingly,
such representations, warranties and covenants should not be relied on as accurately representing the current state of our affairs.
Before you invest, you should read the registration
statement of which this document forms a part, this document, the accompanying prospectus and the documents incorporated by reference
herein that are described under the headings “Where You Can Find More Information” and “Incorporation by Reference.”
The information in this document may only be
accurate on the date of the document. You should assume that the information appearing in this prospectus supplement is accurate
only as of the date on the front cover of this prospectus supplement. Our business, financial condition, results of operations
and prospects may have changed since that date.
We are not making an offer of these securities
in any jurisdiction where the offer is not permitted.
This prospectus supplement contains summaries
of certain provisions contained in some of the documents described herein, but reference is made to the actual documents for complete
information. All of the summaries are qualified in their entirety by the actual documents. Copies of some of the documents referred
to herein have been filed, will be filed or will be incorporated by reference as exhibits to the registration statement of which
this prospectus supplement is a part, and you may obtain copies of those documents as described below under the heading “Where
You Can Find More Information.”
SPECIAL NOTE REGARDING FORWARD-LOOKING
STATEMENTS
This prospectus supplement, the accompanying
prospectus, and the documents incorporated by reference contain forward-looking statements within the meaning of Section 27A of
the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended, that involve risks
and uncertainties. Forward-looking statements provide current expectations of future events based on certain assumptions and include
any statement that does not directly relate to any historical or current fact. Forward-looking statements may refer to such matters
as products, product benefits, pre-clinical and clinical development timelines, clinical and regulatory expectations and plans,
regulatory developments and requirements, the receipt of regulatory approvals, the results of clinical trials, patient acceptance
of Lipocine’s products, manufacturing and commercialization of Lipocine’s products, anticipated financial performance,
future revenues or earnings, business prospects, projected ventures, new products and services, anticipated market performance,
future expectations for liquidity and capital resources needs and similar matters. These are based on our management’s current
beliefs, expectations and assumptions about future events, conditions and results and on information currently available to us.
Discussions containing these forward-looking statements may be found, among other places, in the Sections of this prospectus supplement
entitled “Prospectus Summary” and “Risk Factors.” See also “Special Note Regarding Forward-Looking
Statements” in the accompanying prospectus.
All statements, other than statements of historical
fact, included or incorporated herein regarding our strategy, future operations, financial position, future revenues, projected
costs, plans, prospects and objectives are forward-looking statements. In some cases, you can identify forward-looking statements
by terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,”
“intend,” “may,” “plan,” “potential,” “predict,” “project,”
“should,” “will,” “would” and similar expressions. These statements involve risks, uncertainties
and other factors that may cause our actual results, performance, time frames or achievements to be materially different from any
future results, performance, time frames or achievements expressed or implied by the forward-looking statements. Risks, uncertainties
and other factors that might cause or contribute to such differences include, but are not limited to, those discussed in the Section
entitled “Risk Factors” in our most recent Annual Report on Form 10-K. Given these risks, uncertainties and other factors,
many of which are beyond our control, you should not place undue reliance on these forward-looking statements.
In addition, past financial and/or operating
performance is not necessarily a reliable indicator of future performance and you should not use our historical performance to
anticipate results or future period trends. We can give no assurances that any of the events anticipated by the forward-looking
statements will occur or, if any of them do, what impact they will have on our results of operations and financial condition. Except
as required by law, we assume no obligation to update these forward-looking statements publicly, or to revise any forward-looking
statements to reflect events or developments occurring after the date of this prospectus supplement, even if new information becomes
available in the future.
PROSPECTUS SUPPLEMENT SUMMARY
This summary is not complete and does
not contain all of the information that you should consider before investing in the securities offered by this prospectus supplement
and the accompanying prospectus. You should carefully read this prospectus supplement and the accompanying prospectus, including
the documents we incorporate by reference, to understand fully our common stock as well as other considerations that are important
to you in making a decision to invest in our common stock. You should pay special attention to the “Risk Factors” section
beginning on page S-11 of this prospectus supplement, the “Risk Factors” section beginning on page 7 of the accompanying
prospectus, and the “Risk Factors” section beginning on page 17 of our Annual Report on Form 10-K for the year ended
December 31, 2016, which is incorporated by reference in this prospectus supplement, along with our consolidated financial statements
and notes to those consolidated financial statements and the other information incorporated by reference in this prospectus supplement
and the accompanying prospectus to determine whether an investment in our common stock is appropriate for you. This prospectus
supplement and the accompanying prospectus include forward-looking statements that involve risks and uncertainties.
Summary of Our Business
We are a specialty pharmaceutical company focused
on applying our oral drug delivery technology for the development of pharmaceutical products in the area of men’s and women’s
health. Our proprietary delivery technologies are designed to improve patient compliance, efficacy and safety through orally available
treatment options. Our primary development programs are based on oral delivery solutions for poorly bioavailable drugs. We have
a portfolio of proprietary product candidates designed to produce favorable pharmacokinetic (“PK”) characteristics
and facilitate lower dosing requirements, bypass first-pass metabolism in certain cases, reduce side effects, and eliminate gastrointestinal
interactions that limit bioavailability. Our lead product candidate, LPCN 1021, is an oral testosterone replacement therapy (“TRT”)
that received a Complete Response Letter (“CRL”) from the U.S. Food and Drug Administration (“FDA”) on
June 28, 2016, after filing a New Drug Application (“NDA”). A CRL indicates that the FDA will not approve an NDA in
its current form. We completed a Post Action meeting with the FDA relating to the CRL for LPCN 1021 and announced the on-going
conduct of the Dosing Validation (“DV”) clinical study and the Dosing Flexibility (“DF”) clinical study.
Additional pipeline candidates include LPCN 1111, a next generation oral testosterone therapy product with the potential for once
daily dosing, that is currently in Phase 2 testing, and LPCN 1107, which has the potential to become the first oral hydroxyprogesterone
caproate product indicated for the prevention of recurrent preterm birth, and has completed an End of Phase 2 meeting with the
FDA.
Industry
Testosterone Background
Testosterone, or T, is the primary circulating
sex hormone in males and is critical to the development and maturation of reproductive tissues as well as other secondary male
characteristics such as muscle growth and bone density. Synthesized in the gonads of both males (testis) and females (ovaries),
testosterone circulates bound to sex hormone binding globulin (“SHBG”, ~60%), loosely bound to albumin, a protein in
the blood that binds to testosterone (~40%), or as a free molecule (~1%). Once circulating, testosterone enters cells directly
and activates a network of proteins that ultimately result in metabolic conversions, which in turn produce observable effects.
The concentration of circulating testosterone can vary drastically over time or between individuals and can be dependent on genetic
factors, other medical conditions, lifestyle behaviors, and/or concurrent medication administration. Although large variability
exists, the effects of testosterone are also determined by a number of factors including the amount of steroid penetration, sensitivity
of enzymes and cellular proteins to the hormone, and the action of genomic receptors at the cellular level. As a result, assessing
clinically low, or potentially high, levels of naturally occurring testosterone often requires a number of quantitative tests in
conjunction with clinical evaluations.
Hypogonadism Overview
Low serum testosterone causes significant clinical
impact and can result in erectile dysfunction, low libido, decreased muscle mass and strength, increased body fat, decreased bone
density, decreased vitality and depressed mood. Furthermore, low serum testosterone concentrations have been found to be an independent
predictor of a number of cardiovascular risk factors including obesity, abnormal lipid levels, hypertension, type 2 diabetes, and
systemic inflammation. Well-designed, prospective clinical trials have determined that low testosterone levels are also independently
associated with mortality risk. These findings have generated interest amongst the medical community and general public regarding
the importance of maintaining appropriate serum testosterone levels, which has stimulated growth of the testosterone replacement
therapy market which peaked in 2013. The testosterone therapy market has contracted since 2013 due to a number of factors including
the withdrawal of direct to consumer advertising mid-2014.
Hypogonadism typically refers to a permanent
deficiency of sex hormones rather than a temporary deficiency that may be related to acute/chronic illnesses or other medical,
personal, or environmental factors. Primary hypogonadism describes disease states that intrinsically affect the gonads. Examples
of these include the genetic disorders, Turner syndrome and Kleinfelter syndrome. Secondary hypogonadism refers to disease states
that affect gonadal-related structures such as the hypothalamus and pituitary gland that directly impact the development of gonads
and as such the release of testosterone and other sexual hormones. Kallmann syndrome, in which patients fail to undergo all of
the changes associated with puberty, is a type of secondary hypogonadism. Although a number of inherited diseases are known to
affect the gonads either directly or indirectly, it is generally believed that the majority of individuals with hypogonadism develop
the condition as a result of age-related declines in testosterone or other acquired conditions.
Diagnosis and Treatment of Hypogonadism
Epidemiological studies have determined that
total testosterone follows an age-related decline with mean serum concentration at the age of 75 years approximately two thirds
that at 25 years. Because naturally occurring testosterone exists at low concentrations, with normal testosterone levels in the
range of 300 to 1080 ng/dL automated platform-based assays have been found to lack specificity and are prone to inter-lab variability.
The lack of reliable laboratory tests is complicated further by the inter-individual variability seen in an unaffected population.
Thus, in order to accurately diagnose hypogonadism in a male, at least two morning serum testosterone levels are performed in conjunction
with a clinical assessment of patient symptoms. Patients can only be diagnosed when they present with symptoms that are directly
related to low morning serum testosterone level.
Treatment for male hypogonadism (both primary
and secondary) is testosterone replacement therapy, or TRT. Some of the reported benefits of TRT include improved libido and sexual
function; increased bone density, muscle development, and cognition; as well as a reduction in other risk factors caused by low
testosterone.
Testosterone Replacement Market
Due to the wide variability in therapeutic range
and other medical conditions that may confound an accurate diagnosis, there is a consensus that male hypogonadism is significantly
undertreated. A large study of 2,162 men over the age of 45 visiting primary care practices in the United States revealed that
the prevalence of hypogonadism is about 39%. Based on this prevalence rate and the U.S. Census Bureau’s 2010 estimate that
there are 50 million men between 45 and 75 years old, approximately 19 million men in the U.S. may have low testosterone. In the
study, fewer than 4% of patients were receiving treatment for hypogonadism.
Testosterone replacement therapies have been
commercially available in the United States for over 70 years and have followed a progression of delivery systems that included
subcutaneous, or under-the-skin, intramuscular, transdermal patch, and finally topical gels, which initially surfaced in 2000,
and creams. In 2014, a long acting intramuscular injection and an intranasal delivery system for testosterone were approved. The
difficulty in creating an easy to use/administer and clinically effective testosterone therapy is related to the molecule’s
complex pharmacokinetics. Pharmacokinetics, or PK, describe how the body affects a specific drug after administration through the
mechanism of absorption and distribution, as well as the chemical changes of the substance in the body. For example, oral therapies,
which would ideally be the most popular route of delivery, require multiple, high daily doses due to low bioavailability. Bioavailability
is the fraction of a drug dose that is actually absorbed into the bloodstream. Additionally, the few oral therapies that have been
used in the United States previously quickly went out of favor after significant side effects were revealed, most notably liver
toxicity.
Currently, the U.S. TRT market consists of therapies
that exist in four forms:
|
|
•
|
buccal tablet, which is a tablet shaped patch applied to
the upper gums.
|
Although transdermal patches were previously
the most desirable application type, gel-based TRT has gained increasing popularity due to improved skin tolerability. Despite
becoming a popular approach to male hypogonadism treatment, topical gels are not without limitations. Topical gels place women
and children at risk of testosterone transference (secondary exposure to gels), which has prompted the FDA to add black box warnings
relating to testosterone transference in the label of approved topical products. Despite these limitations, gels have continued
to demonstrate significant market penetration.
The male testosterone market was $2 billion
in 2015 according to IMS Health data. Additionally testosterone replacement prescriptions were approximately 6.2 million in 2015
according to IMS Health data. Injectables are the predominant dosage form in this market in terms of annual prescriptions written
although topical gels also have a significant share of total annual prescriptions. The historical growth in the market was driven
by increasing recognition by both patients and providers of the prevalence of hypogonadism and its far-reaching medical consequences.
Top treatments are marketed by AbbVie, Eli Lilly, and Endo.
Product Candidates
Our current portfolio, shown below, includes
our lead product candidate, LPCN 1021, an oral testosterone replacement therapy, that received a CRL from the FDA on June 28, 2016
and recently announced the on-going conduct of the DV clinical study and the DF clinical study. Additionally, we are in the process
of establishing our pipeline of other clinical candidates including a next generation potential once daily oral testosterone replacement
therapy, LPCN 1111, and an oral therapy for the prevention of preterm birth, LPCN 1107.
Our Development Pipeline
|
Product Candidate
|
|
Indication
|
|
Status
|
|
Next Expected Milestone(s)
|
|
Men’s Health
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
LPCN 1021
|
|
Testosterone Replacement
|
|
Phase 3
|
|
Top-line efficacy results from the DV Study
and DF Study (2Q 2017)
|
|
|
|
|
|
|
|
|
|
LPCN 1111
|
|
Testosterone Replacement
|
|
Phase 2
|
|
End of Phase 2 meeting with FDA (2H 2017)
|
|
|
|
|
|
|
|
|
|
Women’s Health
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
LPCN 1107
|
|
Prevention of Preterm Birth
|
|
Phase 2
|
|
Submit Phase 3 protocol to FDA via SPA (1H 2017)
|
These products are based on our proprietary
Lip’ral promicellar drug delivery technology platform. Lip’ral promicellar technology is a patented technology based
on lipidic compositions which form an optimal dispersed phase in the gastrointestinal environment for improved absorption of insoluble
drugs. The drug loaded dispersed phase presents the solubilized drug efficiently at the absorption site (gastrointestinal tract
membrane) thus improving the absorption process and making the drug less dependent on physiological variables such as dilution,
gastro-intestinal pH and food effects for absorption. Lip’ral based formulation enables improved solubilization and higher
drug-loading capacity, which can lead to improved bioavailability, reduced dose, faster and more consistent absorption, reduced
variability, reduced sensitivity to food effects, improved patient compliance, and targeted lymphatic delivery where appropriate.
LPCN 1021: An Oral Product Candidate for Testosterone Replacement
Therapy
Our lead product, LPCN 1021, is an oral formulation
of the chemical, testosterone undecanoate (“TU”), an eleven carbon side chain attached to testosterone. TU is an ester
prodrug of testosterone. An ester is chemically formed by bonding an acid and an alcohol. Upon the cleavage, or breaking, of the
ester bond, testosterone is formed. TU has been approved for use outside the United States for many years for delivery via intra-muscular
injection and in oral dosage form and recently TU has received regulatory approval in the United States for delivery via intra-muscular
injection. We are using our Lip’ral technology to facilitate steady gastrointestinal solubilization and absorption of TU.
Proof of concept was initially established in 2006, and subsequently LPCN 1021 was licensed in 2009 to Solvay Pharmaceuticals,
Inc. which was then acquired by Abbott Products, Inc. (“Abbott”). Following a portfolio review associated with the
spin-off of AbbVie by Abbott in 2011, the rights to LPCN 1021 were reacquired by us. All obligations under the prior license agreement
have been completed except that Lipocine will owe Abbott a perpetual 1% royalty on net sales. Such royalties are limited to $1
million in the first two calendar years following product launch, after which period there is not a cap on royalties and no maximum
aggregate amount. If generic versions of any such product are introduced, then royalties are reduced by 50%.
We are currently conducting two on-going Phase
3 clinical studies with LPCN 1021, the DV Study and the DF Study. The DV study will assess the impact of LPCN 1021 in hypogonadal
males (testosterone < 300 ng/dL) on a fixed daily dose of 450 mg divided into two equal doses whereas the DF study will assess
LPCN 1021 in hypogonadal males on a fixed daily dose of 450 mg divided into three equal doses. Both of the DV and DF studies are
an open-label, fixed dose, no titration, single treatment arm study of LPCN 1021 and are expected to enroll 100 hypogonadal males
in each study. Efficacy will be assessed via responder analysis at the end of the dosing period which is 24 days. The pre-specified
primary endpoint is the percentage of subjects with an average 24-hour serum testosterone concentration (“Cavg”) within
the normal range, with secondary endpoints based on maximum serum testosterone concentrations (“Cmax”). We currently
expect top-line results from the DV study and DF study in the second quarter of 2017.
Prior to conducting the DV study and the DF
study, we completed our Study of Oral Androgen Replacement (“SOAR”) pivotal Phase 3 clinical study evaluating efficacy
and safety of LPCN 1021 and have received efficacy results and 52-week safety results.
Results from SOAR
SOAR was a randomized, open-label, parallel-group,
active-controlled, Phase 3 clinical study of LPCN 1021 in hypogonadal males with low testosterone (< 300 ng/dL). In total, 315
subjects at 40 active sites were assigned, such that 210 were randomized to LPCN 1021 and 105 were randomized to the active control,
Androgel 1.62%®, for 52 weeks of treatment. The active control is included for safety assessment. LPCN 1021 subjects were started
at 225 mg TU (equivalent to ~ 142 mg of T) twice daily (“BID”) with a standard meal and then dose titrated, if needed,
based on Cavg and Cmax up to 300 mg TU BID or down to 150 mg TU BID based on serum testosterone measured at weeks 3 and 7 based
on PK profiling with multiple blood samples drawn at each time period. The mean age of the subjects in the trial was ~53 years
with ~91% of the patients < 65 years of age.
Primary statistical analysis was conducted using
the Efficacy Population Set (“EPS”). The EPS is defined as subjects randomized into the study with at least one PK
profile and no significant protocol deviations and includes imputed missing data by last observation carried forward, N=151. Further
analysis was performed using the full analysis set (“FAS”) (any subject randomized into the study with at least one
post-baseline efficacy variable response, N=193) and the safety set (“SS”) (any subject that was randomized into the
study and took at least one dose, N=210).
Efficacy
The primary efficacy end point is the percentage
of subjects with Cavg within the normal range, which was defined as 300-1140 ng/dL, after 13 weeks of treatment. The FDA guidelines
for primary efficacy success is that at least 75% of the subjects on active treatment achieve a testosterone Cavg within the normal
range; and the lower bound of the 95% confidence interval (“CI”) must be greater than or equal to 65%.
LPCN 1021 met the FDA primary efficacy guideline.
In the EPS analysis, 87% of the subjects on active treatment achieved testosterone Cavg within the normal range with lower bound
CI of 82%. Additionally, sensitivity analysis using the FAS and SS reaffirmed the finding that LPCN 1021 met the FDA primary efficacy
guideline as 87% and 80%, respectively, of the subjects on active treatment achieved testosterone Cavg within the normal range
with lower bound CI of 82% and 74%, respectively.
Secondary efficacy endpoints are based on subjects
Cmax levels. The FDA guidelines for secondary efficacy success is that at least 85% of the subjects achieve Cmax less than 1500
ng/dL; no greater than 5% of the subjects have Cmax between 1800 ng/dl and 2500 ng/dL; and zero percent of the subjects have Cmax
greater than 2500 ng/dL.
In the EPS analysis, Cmax ≤1500 ng/dL was
83%, Cmax between 1800 and 2500 ng/dL was 4.6% and Cmax > 2500 ng/dL was 2%. Three patients had a Cmax >2500 ng/dL which
were transient, isolated and sporadic. Moreover, none of these subjects reported any adverse events (“AEs”) through
the efficacy readout at week 13. Results were generally consistent with those of approved TRT products.
Safety
The safety component of the SOAR trial was completed
the last week of April 2015. The safety extension phase was designed to assess safety based on information such as metabolites,
biomarkers, laboratory values, serious adverse events (“SAEs”) and AEs, with subjects on their stable dose regimen
in both the treatment arm and the active control arm. LPCN 1021 treatment was well tolerated in that there were no hepatic, cardiac
or drug related SAEs.
LPCN 1021 safety highlights include:
|
|
·
|
LPCN 1021 was well tolerated during 52 weeks of dosing;
|
|
|
·
|
Overall AE profile for LPCN 1021 was comparable to the active control;
|
|
|
·
|
Cardiac AE profiles were consistent between treatment groups and none of the observed cardiac AEs occurred in greater than
1.0% of the subjects in the LPCN 1021 arm and none were classified as severe; and
|
|
|
·
|
All observed adverse drug reactions (“ADRs”) were classified as mild or moderate in severity and no serious ADRs
occurred during the 52-week treatment period.
|
Food Effect Study
We also completed our labeling “food effect”
study in May 2015. Results from the labeling “food effect” study indicate that bioavailability of testosterone from
LPCN 1021 is not affected by changes in meal fat content. The results demonstrate comparable testosterone levels between the standard
fat meal (similar to the meal instruction provided in the Phase 3 clinical study) and both the low and high fat meals. The labeling
“food effect” study was conducted per the FDA requirement and we submitted preliminary results from this study to the
FDA in the second quarter of 2015 prior to submitting the NDA.
Other Safety Requirements
Based on our meetings with the FDA, we do not
expect to be required to conduct a heart attack and stroke risk study or any additional safety studies prior to the potential approval
of our NDA for LPCN 1021. We may, however, be required to conduct a heart attack and stroke risk study on our own or with a consortium
of sponsors that have an approved TRT product subsequent to the potential approval of LPCN 1021.
NDA Submission
We submitted our NDA to the FDA in September
2015 based on data from our SOAR trial. The FDA accepted our NDA in October 2015 and assigned a Prescription Drug User Fee Act
(“PDUFA”) goal date of June 28, 2016 for completion of the review. On June 28, 2016, we received a CRL from the FDA.
A CRL is a communication from the FDA that informs companies that an application cannot be approved in its present form. The CRL
identified deficiencies related to the dosing algorithm for the label. Specifically, the proposed titration scheme for clinical
practice was significantly different from the titration scheme used in the Phase 3 trial leading to discordance in titration decisions
between the Phase 3 trial and real-world clinical practice. In response to the CRL, we met with the FDA in a Post Action meeting,
and proposed a dosing regimen to the FDA based on analyses of existing data. The FDA noted that while the proposed dosing regimen
might be acceptable, validation in a clinical trial would be needed prior to resubmission. As a result, we initiated the DV study
in response to the FDA’s request in December 2016. The DV study is assessing LPCN 1021 in 100 hypogonadal males on a fixed
daily dose (no titration) of 450 mg divided into two equal doses. We also initiated the DF study to assess LPCN 1021 in 100 hypogonadal
males on a fixed daily dose (no titration) of 450 mg divided into three equal doses. This additional clinical trial for LPCN 1021
will require capital and may result in delays in approving LPCN 1021 and commercializing LPCN 1021 if it is approved. There is
no guarantee of approval of LPCN 1021 even if these additional activities are performed.
LPCN 1111: A Next-Generation Oral Product
Candidate for TRT
LPCN 1111 is a next-generation, novel ester
prodrug of testosterone which uses the Lip’ral technology to enhance solubility and improve systemic absorption. We completed
a Phase 2b dose finding study in hypogonadal men in the third quarter of 2016. The primary objectives of the Phase 2b clinical
study were to determine the starting Phase 3 dose of LPCN 1111 along with safety and tolerability of LPCN 1111 and its metabolites
following oral administration of single and multiple doses in hypogonadal men. The Phase 2b clinical trial was a randomized, open
label, two-period, multi-dose PK study that enrolled hypogonadal males into five treatment groups. Each of the 12 subjects in a
group received treatment for 14 days. Results of the Phase 2b study suggest that the primary objectives were met, including identifying
the dose expected to be tested in a Phase 3 study. Good dose-response relationship was observed over the tested dose range in the
Phase 2b study. Additionally, the target Phase 3 dose met primary and secondary end points. Overall, LPCN 1111 was well tolerated
with no drug-related severe or serious adverse events reported in the Phase 2b study.
Additionally in October 2014, we completed a
Phase 2a proof-of-concept study in hypogonadal men. The Phase 2a open-label, dose-escalating single and multiple dose study enrolled
12 males. Results from the Phase 2a clinical study demonstrated the feasibility of a once daily dosing with LPCN 1111 in hypogonadal
men and a good dose response. Additionally, the study confirmed that steady state is achieved by day 14 with consistent inter-day
performance observed on day 14, 21 and 28. No subjects exceeded Cmax of 1500 ng/dL at any time during the 28-day dosing period
on multi-dose exposure. Overall, LPCN 1111 was well tolerated with no serious AE’s reported.
The next step will be to conduct a preclinical
toxicology study with LPCN 1111 and subsequently meet with the FDA for an End of Phase 2 meeting. We anticipate the End of Phase
2 meeting will occur in the second half of 2017.
LPCN 1107: An Oral Product Candidate for the Prevention of
Preterm Birth
We believe LPCN 1107
has the potential to become the first oral hydroxyprogesterone caproate (“HPC”) product indicated for the reduction
of risk of preterm birth (“PTB”) in women with singleton pregnancy who have a history of singleton spontaneous PTB.
Prevention of PTB is a significant unmet need as ~11.7% of all U.S. pregnancies result in PTB (delivery less than 37 weeks), a
leading cause of neonatal mortality and morbidity.
A traditional pharmacokinetics/pharmacodynamics
(“PK/PD”) based Phase 2 clinical study in the intended patient population is not expected to be required prior to entering
into Phase 3. Therefore; based on the results of our multi-dose PK study we had an End-of-Phase 2 meeting with the FDA as well
as other guidance meetings with the FDA to define a Phase 3 development plan for LPCN 1107. During the End-of-Phase 2 meeting and
subsequent guidance meetings, the FDA agreed to a randomized, open-label, two-arm clinical study to include a LPCN 1107 arm and
a comparator IM arm with treatment up to 23 weeks. The FDA also provided feedback on other critical Phase 3 study design considerations
including: positive feedback on the proposed 800 mg BID Phase 3 dose and dosing regimen; confirmation of the use of a surrogate
primary endpoint focusing on rate of delivery less than 37 weeks gestation rather on clinical infant outcomes; acknowledged that
the use of a gestational age endpoint would likely lead to any FDA approval, if granted, being a Subpart H approval as opposed
to a full approval; and, recommended a non-inferiority (“NI”) study margin of 7% with interim analyses. A standard
statistical design for a NI study based on the FDA suggested NI margin of 7% for the primary endpoint may require ~1,100 subjects
per treatment arm with a 90% power. However, based on the FDA’s feedback of including an interim analysis in the NI design,
an adaptive study design is under consideration that may allow for fewer subjects. Lipocine plans to submit the LPCN 1107 Phase
3 protocol to the FDA via a SPA in the first half of 2017. Additionally, manufacturing scale-up work for LPCN 1107 is being planned
and needs to occur before the start of the Phase 3 clinical study for LPCN 1107. A planned food-effect study will also need to
be conducted either before or in parallel with the Phase 3 clinical study.
The FDA has granted
orphan drug designation to LPCN 1107 based on a major contribution to patient care. Orphan designation qualifies Lipocine for various
development incentives, including tax credits for qualified clinical testing, and a waiver of the prescription drug user fee when
we file our NDA.
Corporate Information
Marathon
Bar Corp. (“Marathon Bar”) was incorporated on October 13, 2011, in the State of Delaware. On July 24, 2013, Marathon
Bar and MBAR Acquisition Corp. (“Merger Sub”), a wholly owned subsidiary of Marathon Bar, and Lipocine Operating Inc.
(“Lipocine Operating”), a privately held company incorporated in Delaware, executed an Agreement and Plan of Merger
(“Merger Agreement”). Pursuant to the Merger Agreement, Merger Sub merged with and into Lipocine Operating and Lipocine
Operating was the surviving entity. Additionally, pursuant to the Merger Agreement, Marathon Bar changed its name to Lipocine Inc.
Our
principal executive offices are located at 675 Arapeen Drive, Suite 202, Salt Lake City, Utah 84108 and our telephone number is
(801) 994-7383. We maintain a website at www.lipocine.com. Information contained in or accessible through our website does not
constitute a part of this prospectus supplement.
THE OFFERING
|
Common stock offered by us
|
|
Shares of our common stock having an aggregate offering price of up to $20 million.
|
|
|
|
|
|
Common stock outstanding after this offering
|
|
Up to 5,000,000 shares, assuming sales at a price of $4.00 per share, which was the closing price of our common stock on The NASDAQ Capital Market on March 3, 2017. The actual number of shares issued will vary depending on the sales price under this offering.
|
|
|
|
|
|
Manner of offering
|
|
“At the market offering” that may be made from time to time through our sales agent, Cantor Fitzgerald. See “Plan of Distribution” on page S-15.
|
|
|
|
|
|
Use of Proceeds
|
|
We currently intend to use the net proceeds from this offering primarily for general corporate purposes. See “Use of Proceeds” on page S-12 of this prospectus supplement.
|
|
|
|
|
|
Risk Factors
|
|
Investing in our common stock involves a high degree of risk. You should consider carefully all the information included or incorporated by reference in this prospectus supplement and the sections entitled “Risk Factors” beginning on page S-11 of this prospectus supplement, page 7 of the accompanying prospectus, page 17 of our Annual Report on Form 10-K for the year ended December 31, 2016 and under similar headings in the other documents that are filed after the date hereof and incorporated by reference in this prospectus supplement before deciding whether to purchase our common stock in this offering.
|
|
|
|
|
|
NASDAQ Capital Market symbol
|
|
LPCN
|
The number of shares of common stock to be outstanding
after this offering as reflected in the table above is based on the actual number of shares outstanding as of March 3, 2017, which
was 18,594,889, and does not include, as of that date 2,132,094 options to purchase shares of our common stock issued under our
2014 Stock and Incentive Plan and our 2011 Equity Incentive Plan and 287,000 restricted stock units of our common stock issued
under our 2014 Stock and Incentive Plan.
RISK FACTORS
Before you make a decision to invest
in our securities, you should consider carefully the risks described below, together with other information in this prospectus
supplement, the accompanying prospectus and the information incorporated by reference herein and therein, including any risk factors
contained in our annual and other reports filed with the SEC. If any of the following events actually occur, our business, operating
results, prospects or financial condition could be materially and adversely affected. This could cause the trading price of our
common stock to decline and you may lose all or part of your investment. The risks described below are not the only ones that we
face. Additional risks not presently known to us or that we currently deem immaterial may also significantly impair our business
operations and could result in a complete loss of your investment.
RISKS RELATED TO THIS OFFERING
Since we have broad discretion in how we use the proceeds
from this offering, we may use the proceeds in ways with which you disagree.
We have not allocated specific amounts of the
net proceeds from this offering for any specific purpose. Accordingly, our management will have some flexibility in applying the
net proceeds of this offering. You will be relying on the judgment of our management with regard to the use of these net proceeds,
and you will not have the opportunity, as part of your investment decision, to assess whether the proceeds are being used appropriately.
It is possible that the net proceeds will be invested in a way that does not yield a favorable, or any, return for us. The failure
of our management to use such funds effectively could have a material adverse effect on our business, financial condition, operating
results and cash flow.
You will experience immediate dilution in the book value
per share of the common stock you purchase.
The offering price per share in this offering
may exceed the net tangible book value per share of our common stock outstanding prior to this offering. Assuming that an aggregate
of 5,000,000 shares of our common stock are sold at a price of $4.00 per share, the last reported sale price of our common stock
on the NASDAQ Capital Market on March 3, 2017, for aggregate gross proceeds of $20.0 million, and after deducting commissions and
estimated offering expenses payable by us, you will experience immediate dilution of $2.07 per share, representing the difference
between our as adjusted net tangible book value per share as of December 31, 2016 after giving effect to this offering and the
assumed offering price. The exercise of outstanding stock options and the vesting of outstanding restricted stock units will result
in further dilution of your investment. See “Dilution” on page S-14 for a more detailed discussion of the dilution
you will incur in connection with this offering.
You may experience future dilution as a result of future equity
offerings.
In order to raise additional capital, we may
at any time, including during the pendency of this offering, offer additional shares of our common stock or other securities convertible
into or exchangeable for our common stock at prices that may not be the same as the price per share in this offering. We may sell
shares or other securities in any other offering at a price per share that is less than the price per share paid by investors in
this offering, and investors purchasing shares or other securities in the future could have rights superior to existing stockholders.
The price per share at which we sell additional shares of our common stock, or securities convertible or exchangeable into common
stock, in future transactions may be higher or lower than the price per share paid by investors in this offering.
USE OF PROCEEDS
We may issue and sell shares of our common stock
having aggregate sales proceeds of up to $20.0 million from time to time. Because there is no minimum offering amount required
as a condition to close this offering, the actual total public offering amount, commissions and proceeds to us, if any, are not
determinable at this time. There can be no assurance that we will sell any shares under or fully utilize the Sales Agreement with
Cantor Fitzgerald as a source of financing.
We will retain broad discretion over the use
of the net proceeds from the sale of the securities offered hereby. Accordingly, we reserve the right to use these proceeds for
different purposes or uses which we have not listed above. We currently intend to use the net proceeds from this offering primarily
for general corporate purposes. See “Risk Factors –
Since we have broad discretion in how we use the proceeds from
this offering, we may use the proceeds in ways with which you disagree
.”
Until we use the net proceeds of this offering,
we intend to invest the funds in investment grade, interest-bearing securities.
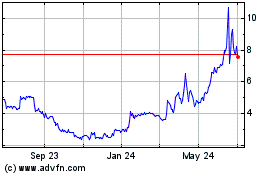
MARKET INFORMATION
Our common stock is quoted on The NASDAQ Capital
Market. The following table sets forth, for the periods indicated, the high and low closing sales prices for our common stock,
as reported on the NASDAQ Capital Market.
|
|
|
High
|
|
|
Low
|
|
|
2015
|
|
|
|
|
|
|
|
|
|
First Quarter
|
|
$
|
8.20
|
|
|
$
|
5.07
|
|
|
Second Quarter
|
|
|
8.58
|
|
|
|
6.50
|
|
|
Third Quarter
|
|
|
18.54
|
|
|
|
7.89
|
|
|
Fourth Quarter
|
|
|
14.38
|
|
|
|
10.22
|
|
|
|
|
|
|
|
|
|
|
|
|
2016
|
|
|
|
|
|
|
|
|
|
First Quarter
|
|
$
|
12.92
|
|
|
$
|
8.14
|
|
|
Second Quarter
|
|
|
12.48
|
|
|
|
3.04
|
|
|
Third Quarter
|
|
|
4.48
|
|
|
|
3.04
|
|
|
Fourth Quarter
|
|
|
5.54
|
|
|
|
3.06
|
|
|
|
|
|
|
|
|
|
|
|
|
2017
|
|
|
|
|
|
|
|
|
|
First Quarter*
|
|
$
|
4.07
|
|
|
$
|
3.28
|
|
*Through March 3, 2017
As of March 3, 2017, there were approximately
136 holders of record of our common stock. This number does not include an undetermined number of stockholders whose stock is held
in “street” or “nominee” name.
DILUTION
Net tangible book value per share is total tangible
assets, reduced by total liabilities, divided by the total number of outstanding shares of common stock. Our net tangible book
value as of December 31, 2016 was approximately $26.0 million, or approximately $1.41 per outstanding share of common stock.
After giving effect to the assumed sale of 5,000,000
shares of our common stock in this offering at an assumed offering price of $4.00 per share, the last reported sale price of our
common stock on the NASDAQ Capital Market on March 3, 2017, and after deducting estimated offering commissions and estimated offering
expenses payable by us, our as adjusted net tangible book value as of December 31, 2016 would have been approximately $45.3 million,
or $1.93 per share. This represents an immediate increase in net tangible book value of $0.52 per share to existing stockholders
and immediate dilution of $2.07 per share to investors purchasing our common stock in this offering at the public offering price.
The following table illustrates this calculation on a per share basis, assuming that we sell all of the securities we are offering:
|
Assumed public offering price per share
|
|
|
|
|
|
$
|
4.00
|
|
|
Net tangible book value per share as of December 31, 2016
|
|
$
|
1.41
|
|
|
|
|
|
|
Increase in net tangible book value per share after this offering
|
|
$
|
0.52
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
As adjusted net tangible book value per share as of December 31, 2016, after this offering
|
|
|
|
|
|
$
|
1.93
|
|
|
|
|
|
|
|
|
|
|
|
|
Dilution in as adjusted net tangible book value per share to new investors
|
|
|
|
|
|
$
|
2.07
|
|
The table above assumes for illustrative purposes
that an aggregate of 5,000,000 shares of our common stock are sold during the term of the Sales Agreement with Cantor Fitzgerald
at a price of $4.00 per share, the last reported sale price of our common stock on The NASDAQ Capital Market on March 3, 2017,
for aggregate gross proceeds of $20.0 million. The shares subject to the Sales Agreement with Cantor Fitzgerald are being sold
from time to time at various prices. An increase of $0.50 per share in the price at which the shares are sold from the assumed
offering price of $4.00 per share shown in the table above, assuming all of our common stock in the aggregate amount of $20.0 million
during the term of the Sales Agreement with Cantor Fitzgerald is sold at that price, would increase our adjusted net tangible book
value per share after the offering to $1.98 per share and would increase the dilution in net tangible book value per share to new
investors in this offering to $2.52 per share, after deducting commissions and estimated aggregate offering expenses payable by
us. A decrease of $0.50 per share in the price at which the shares are sold from the assumed offering price of $4.00 per share
shown in the table above, assuming all of our common stock in the aggregate amount of $20.0 million during the term of the Sales
Agreement with Cantor Fitzgerald is sold at that price, would increase our adjusted net tangible book value per share after the
offering to $1.87 per share and would decrease the dilution in net tangible book value per share to new investors in this offering
to $1.63 per share, after deducting commissions and estimated offering expenses payable by us. This information is supplied for
illustrative purposes only and may differ based on the actual offering price and the actual number of shares offered.
The above discussion is based on
18,456,615
shares of common stock outstanding as of December 31, 2016, and does not include 2,225,850 options to purchase shares of
our common stock issued under our 2014 Stock and Incentive Plan and our 2011 Equity Incentive Plan.
PLAN OF DISTRIBUTION
We have entered into a Controlled Equity Offering
SM
Sales Agreement with Cantor Fitzgerald under which we may issue and sell shares of our common stock having an aggregate gross sales
price of up to $20.0 million from time to time through Cantor Fitzgerald acting as agent. A copy of the Sales Agreement has been
filed as an exhibit to our Annual Report on Form 10-K for the year ended December 31, 2016 and is incorporated by reference into
the registration statement of which this prospectus supplement is a part. See “Where You Can Find More Information”
below.
Upon delivery of a placement notice and subject
to the terms and conditions of the Sales Agreement, Cantor Fitzgerald may sell our common stock by any method permitted by law
deemed to be an “at the market offering” as defined in Rule 415(a)(4) promulgated under the Securities Act, including
sales made directly on or through the NASDAQ Capital Market or on any other existing trading market for our common stock. We may
instruct Cantor Fitzgerald not to sell common stock if the sales cannot be effected at or above the price designated by us from
time to time. We or Cantor Fitzgerald may suspend the offering of common stock upon notice and subject to other conditions.
We will pay Cantor Fitzgerald commissions, in
cash, for its services in acting as agent in the sale of our common stock. Cantor Fitzgerald will be entitled to compensation at
a fixed commission rate of 3.0% of the gross sales price per share sold. Because there is no minimum offering amount required as
a condition to close this offering, the actual total public offering amount, commissions and proceeds to us, if any, are not determinable
at this time. We have also agreed to reimburse Cantor Fitzgerald for certain specified expenses, including the fees and disbursements
of its legal counsel, in an amount up to $50,000. We estimate that the total expenses for the offering, excluding compensation
and reimbursement payable to Cantor Fitzgerald under the terms of the Sales Agreement, will be approximately $80,000.
Settlement for sales of common stock will occur
on the third business day following the date on which any sales are made, or on some other date that is agreed upon by us and Cantor
Fitzgerald in connection with a particular transaction, in return for payment of the net proceeds to us. Sales of our common stock
as contemplated in this prospectus supplement will be settled through the facilities of The Depository Trust Company or by such
other means as we and Cantor Fitzgerald may agree upon. There is no arrangement for funds to be received in an escrow, trust or
similar arrangement.
Cantor Fitzgerald will use its commercially
reasonable efforts, consistent with its sales and trading practices, to solicit offers to purchase the common stock shares under
the terms and subject to the conditions set forth in the Sales Agreement. In connection with the sale of the common stock on our
behalf, Cantor Fitzgerald will be deemed to be an “underwriter” within the meaning of the Securities Act and the compensation
of Cantor Fitzgerald will be deemed to be underwriting commissions or discounts. We have agreed to provide indemnification and
contribution to Cantor Fitzgerald against certain civil liabilities, including liabilities under the Securities Act.
The offering of our common stock pursuant to
the Sales Agreement will terminate upon the termination of the Sales Agreement as permitted therein. We and Cantor Fitzgerald may
each terminate the Sales Agreement at any time upon ten days’ prior notice.
Cantor Fitzgerald and its affiliates may provide
various investment banking, commercial banking and other financial services for us and our affiliates, for which services they
may in the future receive customary fees. To the extent required by Regulation M, Cantor Fitzgerald will not engage in any market
making activities involving our common stock while the offering is ongoing under this prospectus supplement in violation of Regulation
M.
This prospectus supplement in electronic format
may be made available on a website maintained by Cantor Fitzgerald and Cantor Fitzgerald may distribute this prospectus supplement
electronically.
LEGAL MATTERS
Certain legal matters in connection with the
offering and the validity of the securities offered by this prospectus supplement will be passed upon for us by Dorsey &
Whitney LLP, Salt Lake City, Utah. Cantor Fitzgerald & Co. is being represented in connection with this offering by Cooley
LLP, New York, New York.
EXPERTS
The consolidated financial statements of Lipocine
Inc. as of December 31, 2016 and 2015, and for each of the years in the three-year period ended December 31, 2016, have been incorporated
by reference herein and in the registration statement in reliance upon the reports of KPMG LLP, independent registered public accounting
firm, incorporated by reference herein, and upon the authority of said firm as experts in accounting and auditing.
WHERE YOU CAN FIND MORE INFORMATION
We are a reporting company and file annual,
quarterly and current reports, proxy statements and other information with the SEC. We have filed with the SEC a registration statement
on Form S-3 under the Securities Act with respect to the securities we are offering under this prospectus supplement and the accompanying
prospectus. This prospectus supplement and the accompanying prospectus do not contain all of the information set forth in the registration
statement and the exhibits to the registration statement. For further information with respect to us and the securities we are
offering under this prospectus supplement and the accompanying prospectus, we refer you to the registration statement and the exhibits
and schedules filed as a part of the registration statement. You may read and copy the registration statement, as well as our reports,
proxy statements and other information, at the SEC’s Public Reference Room at 100 F Street, N.E., Washington, D.C. 20549.
You can request copies of these documents by writing to the SEC and paying a fee for the copying cost. Please call the SEC at 1-800-SEC-0330
for more information about the operation of the Public Reference Room. The SEC maintains an internet site that contains reports,
proxy and information statements, and other information regarding issuers that file electronically with the SEC, where our SEC
filings are also available. The address of the SEC’s web site is http://www.sec.gov. We maintain a website at www.lipocine.com.
Information contained in or accessible through our website does not constitute a part of this prospectus supplement.
INCORPORATION BY REFERENCE
The SEC allows us to “incorporate by reference”
information that we file with it into this prospectus supplement and the accompanying prospectus, which means that we can disclose
important information to you by referring you to those documents. The information incorporated by reference is an important part
of this prospectus supplement and the accompanying prospectus. The information incorporated by reference is considered to be a
part of this prospectus supplement and the accompanying prospectus, and information that we file later with the Commission will
automatically update and supersede information contained in this prospectus supplement and the accompanying prospectus. We incorporate
by reference the documents listed below that we have previously filed with the Commission:
|
|
·
|
our Annual Report on Form 10-K for the year ended December 31, 2016;
|
|
|
·
|
our current reports on Form 8-K filed with the SEC on January 31, 2017 and March 6, 2017; and
|
|
|
·
|
the description of our common stock contained on our Registration Statement on Form 8-A filed with the SEC on March 18, 2014,
including any amendments or reports filed for the purpose of updating such description.
|
We also incorporate by reference into this prospectus
supplement additional documents that we may file with the SEC under Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act, excluding,
in each case, information deemed furnished and not filed. Any statements contained in a previously filed document incorporated
by reference into this prospectus supplement is deemed to be modified or superseded for purposes of this prospectus supplement
to the extent that a statement contained in this prospectus supplement, or in a subsequently filed document also incorporated by
reference herein, modifies or supersedes that statement.
We will provide to each person, including any
beneficial owner, to whom a prospectus supplement is delivered, without charge upon written or oral request, a copy of any or all
of the information that has been incorporated by reference into this prospectus supplement but not delivered with the prospectus
supplement, including exhibits that are specifically incorporated by reference into such documents. Requests should be directed
to: Lipocine Inc., Attention: Investor Relations, 675 Arapeen Drive, Suite 202, Salt Lake City, Utah 84108, telephone: (801) 994-7383.
PROSPECTUS
LIPOCINE INC.
$100,000,000
Common Stock, Preferred Stock,
Debt Securities,
Warrants and Units
From time to time, we may offer and sell up
to $100,000,000 of any combination of the securities described in this prospectus, either individually or in combination. We may
also offer common stock or preferred stock upon conversion of debt securities, common stock upon conversion of preferred stock,
or common stock, preferred stock or debt securities upon the exercise of warrants.
This prospectus provides a general description
of the securities we may offer. Each time we sell securities, we will provide specific terms of the securities offered in a supplement
to this prospectus. We may also authorize one or more free writing prospectuses to be provided to you in connection with these
offerings. The prospectus supplement and any related free writing prospectus may also add, update or change information contained
in this prospectus. You should carefully read this prospectus, the applicable prospectus supplement and any related free writing
prospectus, as well as any documents incorporated by reference in this prospectus before you invest in any securities. This prospectus
may not be used to consummate a sale of securities unless accompanied by the applicable prospectus supplement.
Our common stock is listed on The NASDAQ Capital
Market under the symbol “LPCN”. On September 29, 2014, the last reported sale price for our common stock was $6.29
per share. The applicable prospectus supplement will contain information, where applicable, as to any other listing on The NASDAQ
Capital Market or any securities market or other exchange of the securities, if any, covered by the prospectus supplement.
INVESTING IN OUR SECURITIES INVOLVES RISKS.
YOU SHOULD REVIEW CAREFULLY THE RISKS AND UNCERTAINTIES DESCRIBED UNDER THE HEADING “RISK FACTORS” ON PAGE 7 AND CONTAINED
IN THE APPLICABLE PROSPECTUS SUPPLEMENT AND ANY RELATED FREE WRITING PROSPECTUS AND UNDER SIMILAR HEADINGS IN THE OTHER DOCUMENTS
THAT ARE INCORPORATED BY REFERENCE INTO THIS PROSPECTUS.
The securities may be sold directly to investors,
through agents designated from time to time or to or through underwriters or dealers. For additional information on the methods
of sale, you should refer to the section entitled “Plan of Distribution” in this prospectus. If any underwriters or
agents are involved in the sale of any securities with respect to which this prospectus is being delivered, the names of such underwriters
or agents and any applicable commissions or discounts and over-allotment options will be set forth in a prospectus supplement.
The price to the public of such securities and the net proceeds we expect to receive from such sale will also be set forth in a
prospectus supplement.
NEITHER THE SECURITIES AND EXCHANGE COMMISSION
NOR ANY STATE SECURITIES COMMISSION HAS APPROVED OR DISAPPROVED OF THESE SECURITIES OR DETERMINED IF THIS PROSPECTUS IS TRUTHFUL
OR COMPLETE. ANY REPRESENTATION TO THE CONTRARY IS A CRIMINAL OFFENSE.
The date of this prospectus is October 14, 2014
TABLE
OF CONTENTS
ABOUT
THIS PROSPECTUS
This prospectus is a part of a registration
statement that we filed with the Securities and Exchange Commission, or SEC, utilizing a “shelf” registration process.
Under this shelf registration process, we may sell any combination of the securities described in this prospectus in one or more
offerings up to a total dollar amount of $100,000,000. This prospectus provides you with a general description of the securities
we may offer. Each time we sell securities under this shelf registration, we will provide a prospectus supplement that will contain
specific information about the terms of that offering. We may also authorize one or more free writing prospectuses to be provided
to you that may contain material information relating to these offerings. The prospectus supplement and any related free writing
prospectus that we may authorize to be provided to you may also add, update or change information contained in this prospectus
or in any documents that we have incorporated by reference into this prospectus. You should read this prospectus, any applicable
prospectus supplement and any related free writing prospectus, together with the information incorporated herein by reference as
described under the heading “Incorporation by Reference.”
You should rely only on the information that
we have provided or incorporated by reference in this prospectus, any applicable prospectus supplement and any related free writing
prospectus that we may authorize to be provided to you. We have not authorized any dealer, salesman or other person to give any
information or to make any representation other than those contained or incorporated by reference in this prospectus, any applicable
prospectus supplement or any related free writing prospectus that we may authorize to be provided to you. You must not rely upon
any information or representation not contained or incorporated by reference in this prospectus or the accompanying prospectus
supplement. This prospectus and the accompanying supplement to this prospectus do not constitute an offer to sell or the solicitation
of an offer to buy any securities other than the registered securities to which they relate, nor do this prospectus and the accompanying
supplement to this prospectus constitute an offer to sell or the solicitation of an offer to buy securities in any jurisdiction
to any person to whom it is unlawful to make such offer or solicitation in such jurisdiction. You should not assume that the information
contained in this prospectus, any applicable prospectus supplement or any related free writing prospectus is accurate on any date
subsequent to the date set forth on the front of the document or that any information we have incorporated by reference is correct
on any date subsequent to the date of the document incorporated by reference, even though this prospectus, any applicable prospectus
supplement or any related free writing prospectus is delivered or securities sold on a later date.
SUMMARY
Prospectus Summary
This summary highlights selected information
from this prospectus and does not contain all of the information that you need to consider in making your investment decision.
You should carefully read the entire prospectus, including the risks of investing discussed under “Risk Factors” beginning
on page 7, the information incorporated by reference, including our financial statements, and the exhibits to the registration
statement of which this prospectus is a part.
Throughout this prospectus, references to “Lipocine,”
the “Company,” “we,” “us,” and “our” refer to Lipocine Inc.
Our Company
We are a specialty pharmaceutical company focused
on applying our oral drug delivery technology for the development of pharmaceutical products in the area of men’s and women’s
health. Our proprietary delivery technology is designed to improve patient compliance and safety through orally available treatment
options. Our primary development programs are based on oral delivery solutions for poorly bioavailable drugs. We have a portfolio
of proprietary product candidates designed to produce favorable pharmacokinetic characteristics and facilitate lower dosing requirements,
bypass first-pass metabolism, reduce side effects, and eliminate gastrointestinal interactions that limit bioavailability.
Overview of Our Product Candidates
Our current portfolio, includes our lead product
candidate LPCN 1021, an oral testosterone replacement therapy, which is currently in a pivotal Phase 3 clinical study. Additionally,
we are currently in the process of establishing our pipeline of early clinical treatments including a next generation testosterone
replacement therapy, LPCN 1111, and an oral therapy for the prevention of preterm birth, LPCN 1107.
LPCN 1021: An Oral Product Candidate for Testosterone Replacement
Therapy
Our lead product, LPCN 1021, is an oral formulation
of the chemical testosterone undecanoate, or TU, an eleven carbon side chain attached to testosterone. It is an ester prodrug of
testosterone, which is an inactive form of testosterone. Upon the cleavage, or breaking, of the ester bond, the pharmacologically
active drug, testosterone is formed. An ester is a chemical between an acid and alcohol. TU has been approved for use outside the
United States for many years for delivery via intra-muscular injection and in oral dosage form and TU recently received approval
in the United States for delivery via intra-muscular. However, this oral dosage form which is approved outside the United States
provides sub-therapeutic serum testosterone levels at the approved dose. We are using our Lip’ral technology to facilitate
steady gastrointestinal solubilization and absorption of TU for convenient twice daily dosing of TU. Proof of concept was initially
established in 2006, and subsequently LPCN 1021 was licensed to Solvay Pharmaceuticals, Inc., or Solvay, which was then acquired
by Abbott Products, Inc., or Abbott, in 2009. Following a portfolio review associated with the spin-off of AbbVie by Abbott in
2011, the rights to LPCN 1021 were reacquired by us.
We have received top-line efficacy results from
our ongoing Study of Oral Androgen Replacement, or SOAR, pivotal Phase 3 clinical study evaluating efficacy and safety of LPCN
1021. SOAR is a randomized, open-label, parallel-group, active-controlled, Phase 3 clinical study of oral TRT in hypogonadal males
with low testosterone (< 300 ng/dL). In total, 315 subjects at 40 active sites were assigned, such that 210 were randomized
to LPCN 1021 and 105 were randomized to the active control, for 52 weeks of treatment. The active control is included for safety
assessment. LPCN 1021 subjects were started at 225 mg Testosterone Undecanoate (“TU”) (equivalent to ~ 142 mg of T)
twice daily (“BID”) with a standard meal and then dose titrated, if needed, up to 300 mg TU BID or down to 150 mg TU
BID based on serum testosterone measured during weeks 3 and 7. The mean age of the subjects in the trial is ~53 yrs with ~91% of
the patients < 65 yrs of age.
Top Line Results From SOAR
Primary statistical analysis was conducted using
the Efficacy Population Set ("EPS"). The EPS is defined as subjects randomized into the study with at least one PK profile
and no significant protocol deviations and includes imputed missing data by last observation carried forward, N=152. Further analysis
was performed using the safety set (“SS”) (any subject that was randomized into the study and took at least one dose,
N=210).
Efficacy.
The primary efficacy
end point is the percentage of subjects with an average 24 hour serum testosterone concentration (“Cavg”) within the
normal range, which is defined as 300-1140 ng/dL, after 13 weeks of treatment. The FDA guidelines for primary efficacy success
is that at least 75% of the subjects on active treatment achieve a testosterone Cavg within the normal range; and the lower bound
of the 95% confidence interval (“CI”) must be greater than 65%.
LPCN 1021 successfully met the FDA primary efficacy
guideline. In the EPS analysis, 88% of the subjects on active treatment achieved testosterone Cavg within the normal range with
lower bound CI of 82%. Additionally, sensitivity analysis using the SS reaffirmed the finding that LPCN 1021 successfully met the
FDA primary efficacy guideline as 80% of the subjects on active treatment achieved testosterone Cavg within the normal range with
lower bound CI of 74%.
Other highlights from the efficacy results include:
|
|
·
|
Mean Cavg was 447 ng/dL with coefficient of variance of 37%;
|
|
|
·
|
Less than 12% of the subjects were outside the testosterone Cavg normal range at final dose;
|
|
|
·
|
85% of subjects arrived at final dose with no more than one titration; and
|
|
|
·
|
51% of subjects were on final dose of 225 mg BID.
|
Safety
.
Although the safety component
of the SOAR trial is on-going, LPCN 1021 treatment has been well tolerated.
LPCN 1021 safety highlights include:
|
|
·
|
3% of the subjects reported a serious adverse events ("SAE"), with none of the SAE's being drug related;
|
|
|
·
|
All the drug related adverse events were either mild or moderate in intensity (none were severe); and
|
|
|
·
|
Hematocrit (“Hct”) and prostate specific antigen (“PSA”) increases were noted and consistent with other TRT products with one subject discontinued for elevated Hct exceeding pre-specified limits and one subject discontinued for elevated PSA exceeding pre-specified limits.
|
In the EPS analysis, Cmax ≤1500 ng/dL was
83%, Cmax between 1800 and 2500 ng/dL was 4.6% and Cmax > 2500 ng/dL was 2%. Three patients had a Cmax >2500 ng/dL which
were transient, isolated and sporadic. Moreover, none of these subjects reported any AE’s. Results were generally consistent
with those of approved TRT products.
The safety extension phase of the SOAR trial
is on-going. The safety extension phase is designed to assess safety information such as metabolites, biomarkers, laboratory values,
SAEs and AEs, with subjects on their stable dose regimen in both the treatment arm and the active control arm.
LPCN 1111: A Next-Generation Oral Product Candidate for TRT
LPCN 1111 is a next-generation, novel ester
prodrug of testosterone which uses the Lip’ral technology to enhance solubility and improve systemic absorption. A Phase
1 single dose, randomized, open label, crossover study in 8 postmenopausal women has been completed and the pharmacokinetics suggested
feasibility of either once-daily dosing or twice daily dosing with relatively high Cavg. This study was conducted ex-U.S. after
obtaining the necessary regulatory approvals for conducting the study in the foreign country so no IND application was required
in the United States. We have recently completed a pre-investigational new drug, or pre-IND, meeting with FDA. In May 2014, we
initiated a Phase 2a proof-of-concept study in hypogonadal men and expect efficacy results in the fourth quarter of 2014. We expect
to initiate a Phase 2b dose finding study in hypogonadal men in either the fourth quarter of 2014 or the first quarter of 2015.
LPCN 1107: An Oral Product Candidate for the Prevention of
Preterm Birth
We believe LPCN 1107
has the potential to become the first oral hydroxyprogesterone caproate (“HPC”) product indicated for the prevention
of preterm birth in women with a prior history of at least one preterm birth. We successfully completed a proof-of-concept Phase
1 clinical study of LPCN 1107 in healthy women in May 2014. The study was designed to determine the pharmacokinetics and bioavailability
of LPCN 1107 relative to an intramuscular ("IM") HPC, as well as safety and tolerability, in healthy non-pregnant female
volunteers. Results of this study confirmed our pre-clinical data and suggest meaningful drug levels of HPC can be obtained after
oral administration. The product has also completed a 28-day repeat dose toxicity study in dogs. We initiated a second Phase 1
pharmacokinetic study in healthy pregnant women and expect efficiency results in the first quarter of 2015.
Corporate Information
On July 24, 2013, Marathon Bar Corp., a Delaware
corporation, MBAR Acquisition Corp., a Delaware corporation and a wholly-owned subsidiary of Marathon Bar, or Merger Sub, and Lipocine
Operating Inc., a Delaware corporation, entered into an Agreement and Plan of Merger and Reorganization, or the Merger Agreement.
Pursuant to the Merger Agreement, Merger Sub merged with and into Lipocine Operating, and Lipocine Operating was the surviving
corporation of the transaction, or the Merger. Following the closing of the Merger, Lipocine Operating became a wholly-owned subsidiary
of Marathon Bar, with the former stockholders of Lipocine Operating owning 99.9% of the outstanding shares of common stock of the
combined company.
On July 24, 2013, immediately prior to the execution
and delivery of the Merger Agreement, Marathon Bar amended its certificate of incorporation to change the name of Marathon Bar
to “Lipocine Inc.” Prior to the execution and delivery of the Merger Agreement, Lipocine had changed its name to “Lipocine
Operating Inc.”
The Merger closed concurrently with the execution
and delivery of the Merger Agreement. As used in this Prospectus (1) all references to the “Combined Company” refer
to Marathon Bar (renamed Lipocine Inc.) and its subsidiaries, including Lipocine Inc. (renamed Lipocine Operating Inc.), following
the closing of the Merger, and (2) unless the context otherwise indicates or requires, all references to “we,” “our”
and “us” refer to the Combined Company from and after the closing of the Merger. Marathon Bar was incorporated on October
13, 2011, in the State of Delaware.
Our principal executive offices are located
at 675 Arapeen Drive, Suite 202, Salt Lake City, Utah 84108 and our telephone number is (801) 994-7383.
The Securities We May Offer
We may offer shares of our common stock and
preferred stock, various series of debt securities and warrants to purchase any of such securities, either individually or in units,
with a total value of up to $100,000,000 million from time to time under this prospectus, together with any applicable prospectus
supplement and related free writing prospectus, at prices and on terms to be determined by market conditions at the time of offering.
This prospectus provides you with a general description of the securities we may offer. Each time we offer a type or series of
securities, we will provide a prospectus supplement that will describe the specific amounts, prices and other important terms of
the securities, including, to the extent applicable:
|
|
·
|
designation or classification;
|
|
|
·
|
aggregate principal amount or aggregate offering price;
|
|
|
·
|
maturity, if applicable;
|
|
|
·
|
original issue discount, if any;
|
|
|
·
|
rates and times of payment of interest or dividends, if any;
|
|
|
·
|
redemption, conversion, exchange or sinking fund terms, if any;
|
|
|
·
|
conversion or exchange prices or rates, if any, and, if applicable, any provisions for changes to or adjustments in the conversion or exchange prices or rates and in the securities or other property receivable upon conversion or exchange;
|
|
|
·
|
restrictive covenants, if any;
|
|
|
·
|
voting or other rights, if any; and
|
|
|
·
|
important United States federal income tax considerations.
|
A prospectus supplement and any related free
writing prospectus that we may authorize to be provided to you may also add, update or change information contained in this prospectus
or in documents we have incorporated by reference. However, no prospectus supplement or free writing prospectus will offer a security
that is not registered and described in this prospectus at the time of the effectiveness of the registration statement of which
this prospectus is a part.
We may sell the securities directly to or through
underwriters, dealers or agents. We, and our underwriters or agents, reserve the right to accept or reject all or part of any proposed
purchase of securities. If we do offer securities through underwriters or agents, we will include in the applicable prospectus
supplement:
|
|
·
|
the names of those underwriters or agents;
|
|
|
·
|
applicable fees, discounts and commissions to be paid to them;
|
|
|
·
|
details regarding over-allotment options, if any; and
|
|
|
·
|
the net proceeds to us.
|
Common Stock
. We may offer shares
of our common stock from time to time. Each outstanding share of common stock entitles the holder thereof to one vote per share
on all matters. Our bylaws provide that any vacancy occurring in the board of directors may be filled by the affirmative vote of
a majority of the remaining directors. Stockholders do not have preemptive rights to purchase shares in any future issuance of
our common stock. In the event of our liquidation, dissolution or winding up, holders of our common stock are entitled to receive,
ratably, the net assets available to stockholders after payment of all creditors. All of the issued and outstanding shares of our
common stock are duly authorized, validly issued, fully paid and non-assessable. Our common stock is described in greater detail
in this prospectus under “Description of Capital Stock — Common Stock.”
Preferred Stock
. Our Board of
directors has the authority under our amended and restated certificate of incorporation, without further action by our stockholders,
to issue up to 10,000,000 shares of preferred stock in one or more series, to establish from time to time the number of shares
to be included in each such series, to fix the rights, preferences, privileges and restrictions of the shares of each wholly unissued
series, including dividend rights, conversion rights, voting rights, terms of redemption, liquidation preference and sinking fund
terms, and to increase or decrease the number of shares of any such series (but not below the number of shares of such series then
outstanding). Our Preferred Stock is described in greater detail in this prospectus under “Description of Capital Stock —
Preferred Stock.”
We will fix the rights, preferences, privileges,
qualifications and restrictions of the preferred stock of each series that we sell under this prospectus and applicable prospectus
supplements in the certificate of designation relating to that series. We will incorporate by reference into the registration statement
of which this prospectus is a part the form of any certificate of designation that describes the terms of the series of preferred
stock we are offering before the issuance of the related series of preferred stock. We urge you to read the prospectus supplements
and any free writing prospectus that we may authorize to be provided to you related to the series of preferred stock being offered,
as well as the complete certificate of designation that contains the terms of the applicable series of preferred stock.
Debt Securities
. We may offer
debt securities from time to time, in one or more series, as either senior or subordinated debt or as senior or subordinated convertible
debt. The debt securities will be issued under one or more documents called indentures, which are contracts between us and a trustee
for the holders of the debt securities. In this prospectus, we have summarized certain general features of the debt securities
under “Description of Debt Securities.” We urge you, however, to read the prospectus supplements and any free writing
prospectus that we may authorize to be provided to you related to the series of debt securities being offered, as well as the complete
indentures that contain the terms of the debt securities. Forms of indentures have been filed as exhibits to the registration statement
of which this prospectus is a part, and supplemental indentures and forms of debt securities containing the terms of debt securities
being offered will be filed as exhibits to the registration statement of which this prospectus is a part, or incorporated by reference
from a current report on Form 8-K that we file with the SEC.
Warrants
. We may offer warrants
for the purchase of our common stock, preferred stock and/or debt securities in one or more series, from time to time. We may issue
warrants independently or together with common stock, preferred stock and/or debt securities, and the warrants may be attached
to or separate from those securities.
In this prospectus, we have summarized certain
general features of the warrants under “Description of Warrants.” We urge you, however, to read the prospectus supplements
and any free writing prospectus that we may authorize to be provided to you related to the particular warrants being offered, as
well as the complete warrant document or agreement that contain the terms of the warrants. Specific warrant documents or agreements
will contain additional important terms and provisions and will be filed as exhibits to the registration statement of which this
prospectus is a part, or incorporated by reference from a current report on Form 8-K that we file with the SEC.
Units
. We may offer units consisting
of common stock, preferred stock, debt securities and/or warrants to purchase any of such securities in one or more series. In
this prospectus, we have summarized certain general features of the units under “Description of Units.” We urge you,
however, to read the prospectus supplements and any free writing prospectus that we may authorize to be provided to you related
to the particular units being offered, as well as the unit agreements that contain the terms of the units. We will file as exhibits
to the registration statement of which this prospectus is a part, or will incorporate by reference from a current report on Form 8-K
that we file with the SEC, the form of unit agreement and any supplemental agreements that describe the terms of the units we are
offering before the issuance of the related units.
THIS PROSPECTUS MAY NOT BE USED TO OFFER
OR SELL ANY SECURITIES UNLESS ACCOMPANIED BY A PROSPECTUS SUPPLEMENT.
NASDAQ Capital Market Listing
Our common stock is listed on The NASDAQ Capital
Market under the symbol “LPCN”. The applicable prospectus supplement will contain information, where applicable, as
to other listings, if any, on the NASDAQ Capital Market or other securities exchange of the securities covered by the applicable
prospectus supplement.
RISK
FACTORS
An investment in our securities involves a
high degree of risk. The prospectus supplement applicable to each offering of our securities will contain a discussion of the risks
applicable to an investment in our securities. Prior to making a decision about investing in our securities, you should carefully
consider the specific factors discussed under the heading “Risk Factors” in the applicable prospectus supplement, together
with all of the other information contained or incorporated by reference in the prospectus supplement or appearing or incorporated
by reference in this prospectus. You should also consider the risks, uncertainties and assumptions discussed under Item 1A,
“Risk Factors,” in our Annual Report on Form 10-K for the fiscal year ended December 31, 2013, and our Quarterly
Reports on Form 10-Q for the quarterly periods ended March 31, 2014 and June 30, 2014, both of which are incorporated herein by
reference, and may be amended, supplemented or superseded from time to time by other reports we file with the Securities and Exchange
Commission in the future. The risks and uncertainties we have described are not the only ones we face. Additional
risks and uncertainties not presently known to us or that we currently deem immaterial may also affect our operations.
SPECIAL
NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus and the documents incorporated
by reference contain forward-looking statements. These are based on our management’s current beliefs, expectations and assumptions
about future events, conditions and results and on information currently available to us. Discussions containing these forward-looking
statements may be found, among other places, in the Sections entitled “Business,” “Risk Factors” and “Management’s
Discussion and Analysis of Financial Condition and Results of Operations” incorporated by reference from our most recent
Annual Report on Form 10-K and in our Quarterly Reports on Form 10-Q, as well as any amendments thereto, filed with the
SEC. Within the meaning of Section 27A of the Securities Act of 1933, as amended, or the Securities Act, and Section 21E
of the Securities Exchange Act of 1934, as amended, or the Exchange Act, these forward-looking statements include, but are not
limited to, statements that are not historical facts relating to expectations, the receipt of regulatory approvals, the results
of clinical trials, patient acceptance of Lipcoine’s products, the potential uses and benefits of our product candidates,
product development efforts, the manufacturing and commercialization of Lipcoine’s products, and customer acceptance of our
products.
All statements, other than statements of historical
fact, included or incorporated herein regarding our products, product benefits, pre-clinical and clinical development timelines,
clinical and regulatory expectations and plans, anticipated financial performance, future revenues or earnings, business prospects,
projected ventures, new products and services, anticipated market performance, future expectations for liquidity and capital resources
needs and similar matters are forward-looking statements. In some cases, you can identify forward-looking statements by terms such
as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,”
“may,” “plan,” “potential,” “predict,” “project,” “should,”
“will,” “would” and similar expressions. These statements involve risks, uncertainties and other factors
that may cause our actual results, performance, time frames or achievements to be materially different from any future results,
performance, time frames or achievements expressed or implied by the forward-looking statements. Risks, uncertainties and other
factors that might cause or contribute to such differences include, but are not limited to, those discussed in the Section entitled
“Risk Factors” in our most recent Annual Report on Form 10-K and in our Quarterly Report on Form 10-Q, as
well as any amendments thereto filed with the SEC. Given these risks, uncertainties and other factors, many of which are beyond
our control, you should not place undue reliance on these forward-looking statements.
Except as required by law, we assume no obligation
to update these forward-looking statements publicly, or to revise any forward-looking statements to reflect events or developments
occurring after the date of this prospectus, even if new information becomes available in the future.
USE
OF PROCEEDS
Except as described in any applicable prospectus
supplement and in any free writing prospectuses in connection with a specific offering, we currently intend to use the net proceeds
from the sale of the securities offered hereby for operating costs, capital expenditures and for general corporate purposes, including
working capital. We may also use a portion of the net proceeds to invest in or acquire businesses, or assets that we believe are
complementary to our own, although we have no current plans, commitments or agreements with respect to any acquisitions as of the
date of this prospectus.
DESCRIPTION
OF CAPITAL STOCK
As of the date of this prospectus, our certificate
of incorporation authorizes us to issue 100,000,000 shares of common stock, par value $0.0001 per share, and 10,000,000 shares
of preferred stock, par value $0.0001 per share. The following is a summary of the rights of our common and preferred stock and
some of the provisions of our amended and restated certificate of incorporation and amended and restated bylaws, our outstanding
warrants, our registration rights agreements and the Delaware General Corporation Law. Because it is only a summary, it does not
contain all the information that may be important to you and is subject to and qualified in its entirety by our amended and restated
certificate of incorporation and our amended and restated bylaws, a copy of each of which has been incorporated as an exhibit to
the registration statement of which this prospectus forms a part.
Our amended and restated certificate of incorporation
and our amended and restated bylaws contain certain provisions that are intended to enhance the likelihood of continuity and stability
in the composition of the board of directors, which may have the effect of delaying, deferring or preventing a future takeover
or change in control of Lipocine unless such takeover or change in control is approved by our board of directors.
Common Stock
As of August 31, 2014 there were 12,792,382
shares of common stock outstanding. In addition, as of August 31, 2014 there were: (i) 1,557,096 shares of common stock subject
to outstanding options; (ii) 15,000 shares of common stock subject to outstanding restricted stock units; (iii) 991,717 shares
of common stock reserved for future issuance under our Amended and Restated 2014 Stock and Incentive Plan; and (iv) 20,467 shares
of common stock subject to an outstanding warrant. Each outstanding share of common stock entitles the holder thereof to one vote
per share on all matters. Our bylaws provide that any vacancy occurring in the board of directors may be filled by the affirmative
vote of a majority of the remaining directors. Stockholders do not have preemptive rights to purchase shares in any future issuance
of our common stock. In the event of our liquidation, dissolution or winding up, holders of our common stock are entitled to receive,
ratably, the net assets available to stockholders after payment of all creditors.
All outstanding shares of common stock are,
and all shares of common stock to be outstanding upon the closing of this offering will be, fully paid and nonassessable.
Additional shares of authorized common stock
may be issued, as authorized by our board of directors from time to time, without stockholder approval, except as may be required
by applicable stock exchange requirements.
The transfer agent and registrar for our common
stock is American Stock Transfer & Trust Company LLC. Their telephone number is 1-800-937-5449. Our common stock is listed
on The NASDAQ Capital Market under the symbol “LPCN”.
Preferred Stock
Our Board of directors has the authority under
our amended and restated certificate of incorporation, without further action by our stockholders, to issue up to 10,000,000 shares
of preferred stock in one or more series, to establish from time to time the number of shares to be included in each such series,
to fix the rights, preferences, privileges and restrictions of the shares of each wholly unissued series, including dividend rights,
conversion rights, voting rights, terms of redemption, liquidation preference and sinking fund terms, and to increase or decrease
the number of shares of any such series (but not below the number of shares of such series then outstanding).
Our board of directors may authorize the issuance
of preferred stock with voting or conversion rights that could have the effect of restricting dividends on our common stock, diluting
the voting power of our common stock, impairing the liquidation rights of our common stock or otherwise adversely affecting the
rights of holders of our common stock. The issuance of preferred stock, while providing flexibility in connection with possible
acquisitions and other corporate purposes, could, among other things, have the effect of delaying, deferring or preventing a change
of control and may adversely affect the market price of our common stock. As of August 31, 2014, no shares of preferred stock were
outstanding, and we have no current plans to issue any shares of preferred stock.
Future Preferred Stock
. Our board
of directors will fix the rights, preferences, privileges, qualifications and restrictions of the preferred stock of each series
that we sell under this prospectus and applicable prospectus supplements in the certificate of designation relating to that series.
We will file as an exhibit to the registration statement of which this prospectus is a part, or incorporate by reference into the
registration statement of which this prospectus is a part the form of any certificate of designation that describes the terms of
the series of preferred stock we are offering before the issuance of the related series of preferred stock. This description will
include:
|
|
·
|
the title and stated value;
|
|
|
·
|
the number of shares we are offering;
|
|
|
·
|
the liquidation preference per share;
|
|
|
·
|
the purchase price per share;
|
|
|
·
|
the dividend rate per share, dividend period and payment dates and method of calculation for dividends;
|
|
|
·
|
whether dividends will be cumulative or non-cumulative and, if cumulative, the date from which dividends will accumulate;
|
|
|
·
|
our right, if any, to defer payment of dividends and the maximum length of any such deferral period;
|
|
|
·
|
the procedures for any auction and remarketing, if any;
|
|
|
·
|
the provisions for a sinking fund, if any;
|
|
|
·
|
the provisions for redemption or repurchase, if applicable, and any restrictions on our ability to exercise those redemption and repurchase rights;
|
|
|
·
|
any listing of the preferred stock on any securities exchange or market;
|
|
|
·
|
whether the preferred stock will be convertible into our common stock or other securities of ours, including warrants, and, if applicable, the conversion period, the conversion price, or how it will be calculated, and under what circumstances it may be adjusted;
|
|
|
·
|
whether the preferred stock will be exchangeable into debt securities, and, if applicable, the exchange period, the exchange price, or how it will be calculated, and under what circumstances it may be adjusted;
|
|
|
·
|
voting rights, if any, of the preferred stock;
|
|
|
·
|
preemption rights, if any;
|
|
|
·
|
restrictions on transfer, sale or other assignment, if any;
|
|
|
·
|
a discussion of any material or special United States federal income tax considerations applicable to the preferred stock;
|
|
|
·
|
the relative ranking and preferences of the preferred stock as to dividend rights and rights if we liquidate, dissolve or wind up our affairs;
|
|
|
·
|
any limitations on issuances of any class or series of preferred stock ranking senior to or on a parity with the series of preferred stock being issued as to dividend rights and rights if we liquidate, dissolve or wind up our affairs; and
|
|
|
·
|
any other specific terms, rights, preferences, privileges, qualifications or restrictions of the preferred stock.
|
When we issue shares of preferred stock under this prospectus,
the shares will be fully paid and nonassessable and will not have, or be subject to, any preemptive or similar rights.
The General Corporation Law of the State of
Delaware, the state of our incorporation, provides that the holders of preferred stock will have the right to vote separately as
a class on any proposal involving fundamental changes in the rights of holders of that preferred stock. This right is in addition
to any voting rights that may be provided for in the applicable certificate of designation.
Outstanding Warrants
As of August 31, 2014, there was an outstanding
warrant to purchase 20,467 shares of our common stock at an exercise price of $12.21 per share (the “Outstanding Warrant”).
The warrant has a net exercise provision and contains provisions for the adjustment of the exercise price and the number of shares
issuable upon exercise in the event of certain stock dividends, stock splits, recapitalizations, reclassifications and consolidations.
The warrant expires on December 31, 2015
.
Registration Rights
Pursuant to two separate registration rights
agreements, the holders of 1,380,796 shares of our common stock or their permitted transferees, are entitled to rights with respect
to the registration of their shares under the Securities Act. These registration rights will expire when such security holder is
able to sell all of its shares pursuant to Rule 144 of the Securities Act, without any volume or timing restrictions. In an underwritten
offering, the underwriter has the right, subject to specified conditions, to limit the number of shares such holders may include
in an offering.
Demand Registration Rights
The holders of 1,071,835 shares of our common
stock or permitted transferees are entitled to certain demand registration rights. The holders of at least 290,201 shares under
the first registration rights agreement and 24,572 shares under the second registration rights agreement may each once request
that we register all or a portion of their shares of our common stock. The request must result in an effective registration statement
or the holders retain the right to request registration. The anticipated net proceeds must be at least $2.5 million, if the registration
is pursuant to Form S-1, or $1.0 million, if the registration is pursuant to Form S-3. If we determine that it would in certain
circumstances reasonably be expected to have a material adverse effect on us to effect such a demand registration, we have the
right to defer such registration, not more than once in any 12-month period, for a period of up to 90 days
.
Piggyback Registration Rights
If we propose to register to offer and sale
of any of our securities under the Securities Act, in connection with the public offering of such securities the holders of 1,380,796
shares of our common stock or permitted transferees, will be entitled to certain “piggyback” registration rights allowing
the holders to include their shares in such registration, subject to certain marketing and other limitations that may be imposed
by the underwriters, if any, in such a registration. As a result, whenever we propose to file a registration statement under the
Securities Act, other than with respect to (1) a registration related to an employee benefit plan or (2) a registration related
to a corporate reorganizations or certain other transactions under Rule 145 of the Securities Act, the holders of these shares
are entitled to notice of the registration and have the right, subject to limitations that the underwriters may impose on the number
of shares included in the registration, to include their shares in the registration.
S-3 Registration Rights
The holders of 1,071,835 shares of our common
stock or permitted transferees may make a written request that we register the offer and sale of these shares on Form S-3, provided
we are eligible to file a registration statement on Form S-3 and the anticipated aggregate offering price of the shares to be sold
is at least $1.0 million. These stockholders may make an unlimited number of requests for registration on Form S-3, which requests
shall not be counted as “demand registrations.” If we determine that it would in certain circumstances reasonably be
expected to have a material adverse effect on us to effect such registration, we have the right to defer such registration, not
more than once in any 12-month period, for a period of up to 90 days
.
Expenses Generally
Generally, we are required to bear all registration
expenses, other than selling expenses such as underwriting discounts and selling commissions, incurred in connection with the demand,
piggyback and Form S-3 registrations described above
.
Anti-Takeover Effects of Our Certificate of Incorporation and
Bylaws
Our amended and restated certificate of incorporation
and amended and restated bylaws contain certain provisions that may have anti-takeover effects, making it more difficult for or
preventing a third party from acquiring control of Lipocine or changing our board of directors and management. According to our
amended and restated certificate of incorporation and amended and restated bylaws, the holders of our common stock do not have
cumulative voting rights in the election of our directors. The combination of the present ownership and control of 14.3% of our
issued and outstanding common stock by our executive officers and directors as a group as of August 31, 2014 and the lack of cumulative
voting, may make it more difficult for other stockholders to replace our board of directors or for a third party to obtain control
of the company by replacing our board of directors.
Delaware Anti-Takeover Law
We are subject to Section 203 of the Delaware
General Corporation Law, or Section 203. Section 203 generally prohibits a public Delaware corporation from engaging in a “business
combination” with an “interested stockholder” for a period of three years after the date of the transaction in
which the person became an interested stockholder, unless:
|
|
·
|
prior to the date of the transaction, the board of directors of the corporation approved either the business combination or the transaction that resulted in the stockholder becoming an interested stockholder;
|
|
|
·
|
upon consummation of the transaction which resulted in the stockholder becoming an interested stockholder, the interested stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced; or
|
|
|
·
|
at or subsequent to the time of the transaction, the business combination is approved by the board of directors of the corporation and authorized at an annual or special meeting of its stockholders, and not by written consent, by the affirmative vote of at least 66 2/3% of our outstanding voting stock that is not owned by the interested stockholder.
|
In general, Section 203 defines a “business
combination” to include mergers, asset sales and other transactions resulting in financial benefit to a stockholder and an
“interested stockholder” as a person who, together with affiliates and associates, owns (or within three years, did
own) 15% or more of a corporation’s voting stock.
S
ection 203 could prohibit or delay mergers
or other takeover or change in control attempts not approved in advance by our Board of directors and, accordingly, may discourage
attempts to acquire us even though such a transaction may offer our stockholders the opportunity to sell their stock at a price
above the prevailing market price.
Amended and Restated Certificate of Incorporation and Amended
and Restated Bylaws
Provisions of our amended and restated certificate
of incorporation bylaws, which became effective following the closing of the Merger, may delay or discourage transactions involving
an actual or potential change of control or change in our Board of directors or our management, including transactions in which
stockholders might otherwise receive a premium for their shares, or transactions that our stockholders might otherwise deem to
be in their best interests. Therefore, these provisions could adversely affect the price of our common stock. Among other things,
our amended and restated certificate of incorporation and bylaws:
|
|
·
|
permit our Board of directors to issue up to 10,000,000 shares of preferred stock, with any rights, preferences and privileges as they may designate (including the right to approve an acquisition or other change of control);
|
|
|
·
|
provide that the authorized number of directors may be changed only by resolution of our Board of directors;
|
|
|
·
|
provide that directors may only be removed, subject to any limitation imposed by law, by the holders of at least a majority of all of our then-outstanding shares of the capital stock entitled to vote generally at an election of directors;
|
|
|
·
|
provide that all vacancies, including newly created directorships, may, except as otherwise required by law, be filled by the affirmative vote of a majority of directors then in office, even if less than a quorum;
|
|
|
·
|
require that any action to be taken by our stockholders must be effected at a duly called annual or special meeting of stockholders and not be taken by written consent or electronic transmission;
|
|
|
·
|
provide that stockholders seeking to present proposals before a meeting of stockholders or to nominate candidates for election as directors at a meeting of stockholders must provide advance notice in writing, and also specify requirements as to the form and content of a stockholder’s notice;
|
|
|
·
|
provide that special meetings of our stockholders may be called only by the chairman of our Board of directors, our chief executive officer or by our Board of directors pursuant to a resolution adopted by a majority of the total number of authorized directors; and
|
|
|
·
|
do not provide for cumulative voting rights (therefore allowing the holders of a majority of the shares of common stock entitled to vote in any election of directors to elect all of the directors standing for election, if they should so choose).
|
The amendment of any of these provisions would
require approval by the holders of at least 66 2/3% of the voting power of all of our then-outstanding common stock entitled to
vote generally in the election of directors, voting together as a single class.
DESCRIPTION
OF DEBT SECURITIES
This section describes the general terms and
provisions of the debt securities that we may offer using this prospectus and the related indentures. This section is only a summary
and does not purport to be complete. You must look to the relevant form of debt security and the related indenture for a full understanding
of all terms of any series of debt securities. The form of debt security and the related indenture have been or will be filed or
incorporated by reference as exhibits to the registration statement of which this prospectus is a part. See “Where You Can
Find More Information” for information on how to obtain copies.
We may issue senior or subordinated debt securities
from time to time in one or more series under one of two separate indentures, which may be supplemented or amended from time to
time. Senior debt securities will be issued under a senior indenture and subordinated debt securities will be issued under a subordinated
indenture. The senior debt indenture and the subordinated debt indenture are referred to individually in this prospectus as the
“indenture” and collectively as the “indentures.” This prospectus outlines briefly the provisions of the
indentures. The particular terms of a series of debt securities and the extent, if any, to which the particular terms of the issue
modify the terms of the indenture will be described in the accompanying prospectus supplement relating to such series of debt securities.
In some instances, certain of the precise terms of debt securities you are offered may be described in a further prospectus supplement,
known as a pricing supplement. The indentures are subject to and governed by the Trust Indenture Act of 1939, as amended, and may
be supplemented or amended from time to time following their execution.
The debt securities may be denominated and payable
in U.S. dollars or foreign currencies. We may also issue debt securities with the principal amount, interest or other amounts payable
to be determined by reference to one or more currency exchange rates, securities or baskets of securities, commodity prices, indices
or any other financial, economic or other measure or instrument, including the occurrence or non-occurrence of any event or circumstance.
Debt securities may bear interest at a fixed rate, which may be zero, or a floating rate.
Some of the debt securities may be issued as
original issue discount debt securities. Original issue discount securities bear no interest or bear interest at below-market rates
and will be sold at a discount from their stated principal amount. The prospectus supplement relating to an issue of original issue
discount securities will contain information relating to United States federal income tax, accounting, and other special considerations
applicable to original issue discount securities.
Holders may present debt securities for exchange
or transfer, in the manner, at the places and subject to the restrictions stated in the debt securities and described in the applicable
prospectus supplement and other offering material we will provide. We will provide these services without charge except for any
tax or other governmental charge payable in connection with these services and subject to any limitations provided in the applicable
indenture pursuant to which such debt securities are issued.
Holders may transfer debt securities in definitive
bearer form and the related coupons, if any, by delivery to the transferee. If any of the securities are held in global form, the
procedures for transfer of interests in those securities will depend upon the procedures of the depositary for those global securities.
We will generally have no obligation to repurchase,
redeem, or change the terms of debt securities upon any event (including a change in control) that might have an adverse effect
on our credit quality.
DESCRIPTION
OF WARRANTS
The following description, together with the
additional information we may include in any applicable prospectus supplements and free writing prospectuses, summarizes the material
terms and provisions of the warrants that we may offer under this prospectus, which may consist of warrants to purchase common
stock, preferred stock or debt securities and may be issued in one or more series. This information does not relate to the Outstanding
Warrant. Warrants may be offered independently or together with common stock, preferred stock or debt securities offered by any
prospectus supplement, and may be attached to or separate from those securities. While the terms we have summarized below will
apply generally to any warrants that we may offer under this prospectus, we will describe the particular terms of any warrants
that we may offer in more detail in the applicable prospectus supplement and any applicable free writing prospectus. The terms
of any warrants offered under a prospectus supplement may differ from the terms described below. However, no prospectus supplement
will fundamentally change the terms that are set forth in this prospectus or offer a security that is not registered and described
in this prospectus at the time of its effectiveness.
We will file as exhibits to the registration
statement of which this prospectus is a part, or will incorporate by reference from a current report on Form 8-K that we file
with the SEC, the form of warrant document or agreement that describes the terms of the particular warrants we are offering before
the issuance of the related warrants. The following summaries of material provisions of the warrants are subject to, and qualified
in their entirety by reference to, all the provisions of the warrant document or agreement applicable to particular warrants. We
urge you to read the applicable prospectus supplement and any applicable free writing prospectus related to the particular warrants
that we sell under this prospectus, as well as the complete warrant document or agreement that contain the terms of the warrants.
General
We will describe in the applicable prospectus
supplement the terms relating to the warrants, including, if applicable:
|
|
·
|
the offering price and aggregate number of warrants offered;
|
|
|
·
|
the currency for which the warrants may be purchased;
|
|
|
·
|
if applicable, the designation and terms of the securities with which the warrants are issued and the number of warrants issued with each such security or each principal amount of such security;
|
|
|
·
|
if applicable, the date on and after which the warrants and the related securities will be separately transferable;
|
|
|
·
|
in the case of warrants to purchase debt securities, the principal amount of debt securities purchasable upon exercise of one warrant and the price at, and currency in which, this principal amount of debt securities may be purchased upon such exercise;
|
|
|
·
|
in the case of warrants to purchase common stock or preferred stock, the number of shares of common stock or preferred stock, as the case may be, purchasable upon the exercise of one warrant and the price at which these shares may be purchased upon such exercise;
|
|
|
·
|
the effect of any merger, consolidation, sale or other disposition of our business on the warrant agreements and the warrants;
|
|
|
·
|
the terms of any rights to redeem or call the warrants;
|
|
|
·
|
any provisions for changes to or adjustments in the exercise price or number of securities issuable upon exercise of the warrants;
|
|
|
·
|
the dates on which the right to exercise the warrants will commence and expire;
|
|
|
·
|
the manner in which the warrant agreements and warrants may be modified;
|
|
|
·
|
material United States federal income tax consequences of holding or exercising the warrants;
|
|
|
·
|
the terms of the securities issuable upon exercise of the warrants; and
|
|
|
·
|
any other specific terms, preferences, rights or limitations of or restrictions on the warrants.
|
Before exercising their warrants, holders of
warrants will not have any of the rights of holders of the securities purchasable upon such exercise, including:
|
|
·
|
in the case of warrants to purchase debt securities, the right to receive payments of principal of, or premium, if any, or interest on, the debt securities purchasable upon exercise or to enforce covenants in the applicable indenture; or
|
|
|
·
|
in the case of warrants to purchase common stock or preferred stock, the right to receive dividends, if any, or, payments upon our liquidation, dissolution or winding up or to exercise voting rights, if any.
|
Exercise of Warrants
Each warrant will entitle the holder to purchase
the securities that we specify in the applicable prospectus supplement at the exercise price that we describe in the applicable
prospectus supplement. Unless we otherwise specify in the applicable prospectus supplement, holders of the warrants may exercise
the warrants at any time up to the specified time on the expiration date that we set forth in the applicable prospectus supplement.
After the close of business on the expiration date, unexercised warrants will become void.
Holders of the warrants may exercise the warrants
by delivering the warrant certificate representing the warrants to be exercised together with specified information, and paying
the required amount to the warrant agent in immediately available funds, as provided in the applicable prospectus supplement. We
will set forth in the warrant agreement or documents and in the applicable prospectus supplement the information that the holder
of the warrant will be required to deliver to the warrant agent.
Upon receipt of the required payment and the
warrant documents properly completed and duly executed at the office of the warrant agent or any other office indicated in the
applicable prospectus supplement, we will issue and deliver the securities purchasable upon such exercise. If fewer than all of
the warrants represented by the warrant certificate are exercised, then we will issue a new warrant certificate for the remaining
amount of warrants. If we so indicate in the applicable prospectus supplement, holders of the warrants may surrender securities
as all or part of the exercise price for warrants.
DESCRIPTION
OF UNITS
The following description, together with the
additional information we may include in any applicable prospectus supplements, summarizes the material terms and provisions of
the units that we may offer under this prospectus. While the terms we have summarized below will apply generally to any units that
we may offer under this prospectus, we will describe the particular terms of any series of units in more detail in the applicable
prospectus supplement. The terms of any units offered under a prospectus supplement may differ from the terms described below.
However, no prospectus supplement will fundamentally change the terms that are set forth in this prospectus or offer a security
that is not registered and described in this prospectus at the time of its effectiveness.
We will file as exhibits to the registration
statement of which this prospectus is a part, or will incorporate by reference from a current report on Form 8-K that we file
with the SEC, the form of unit agreement that describes the terms of the units we are offering, and any supplemental agreements,
before the issuance of the related units. The following summaries of material terms and provisions of the units are subject to,
and qualified in their entirety by reference to, all the provisions of the unit agreement and any supplemental agreements applicable
to particular units. We urge you to read the applicable prospectus supplements related to the particular units that we sell under
this prospectus, as well as the complete unit agreement and any supplemental agreements that contain the terms of the units.
General
We may issue units comprised of one or more
debt securities, shares of common stock, shares of preferred stock and warrants in any combination. Each unit will be issued so
that the holder of the unit is also the holder of each security included in the unit. Thus, the holder of a unit will have the
rights and obligations of a holder of each included security. The unit agreement under which a unit is issued may provide that
the securities included in the unit may not be held or transferred separately, at any time or at any time before a specified date.
We will describe in the applicable prospectus
supplement the terms of the units, including:
|
|
·
|
the designation and terms of the units and of the securities comprising the units, including whether and under what circumstances those securities may be held or transferred separately;
|
|
|
·
|
any provisions of the governing unit agreement that differ from those described below; and
|
|
|
·
|
any provisions for the issuance, payment, settlement, transfer or exchange of the units or of the securities comprising the units.
|
The provisions described in this section, as
well as those described under “Description of Capital Stock,” “Description of Debt Securities” and “Description
of Warrants” will apply to each unit and to any common stock, preferred stock, debt security or warrant included in each
unit, respectively.
Issuance in Series
We may issue units in such amounts and in numerous
distinct series as we determine.
Enforceability of Rights by Holders of Units
Each unit agent will act solely as our agent
under the applicable unit agreement and will not assume any obligation or relationship of agency or trust with any holder of any
unit. A single bank or trust company may act as unit agent for more than one series of units. A unit agent will have no duty or
responsibility in case of any default by us under the applicable unit agreement or unit, including any duty or responsibility to
initiate any proceedings at law or otherwise, or to make any demand upon us. Any holder of a unit may, without the consent of the
related unit agent or the holder of any other unit, enforce by appropriate legal action its rights as holder under any security
included in the unit.
We, the unit agents, and any of their agents
may treat the registered holder of any unit certificate as an absolute owner of the units evidenced by that certificate for any
purpose and as the person entitled to exercise the rights attaching to the units so requested, despite any notice to the contrary.
PLAN
OF DISTRIBUTION
We may sell the securities from time to time
pursuant to underwritten public offerings, direct sales to the public, negotiated transactions, block trades or a combination of
these methods. We may sell the securities to or through underwriters or dealers, through agents, directly to one or more purchasers,
or through any combination of these methods. The distribution of the securities may be effected from time to time in one or more
transactions at a fixed price or prices, which may be changed, at market prices prevailing at the time of sale, at prices related
to the prevailing market prices or at negotiated prices.
A prospectus supplement or supplements (and
any related free writing prospectus that we may authorize to be provided to you) will describe the terms of the offering of the
securities, including, to the extent applicable:
|
|
·
|
the name or names of any underwriters or dealers, if any;
|
|
|
·
|
the purchase price of the securities and the proceeds we will receive from the sale;
|
|
|
·
|
any over-allotment options under which underwriters may purchase additional securities from us;
|
|
|
·
|
any agency fees or underwriting discounts and other items constituting agents’ or underwriters’ compensation;
|
|
|
·
|
any public offering price;
|
|
|
·
|
any discounts or concessions allowed or reallowed or paid to dealers; and
|
|
|
·
|
any securities exchange or market on which the securities may be listed.
|
Only underwriters named in the prospectus supplement
are underwriters of the securities offered by the prospectus supplement.
By Underwriters
If underwriters are used in the sale, they will
acquire the securities for their own account and may resell the securities from time to time in one or more transactions at a fixed
public offering price or at varying prices determined at the time of sale. The obligations of the underwriters to purchase the
securities will be subject to the conditions set forth in the applicable underwriting agreement. We may offer the securities to
the public through underwriting syndicates represented by managing underwriters or by underwriters without a syndicate. Subject
to certain conditions, the underwriters will be obligated to purchase all of the securities offered by the prospectus supplement.
Any public offering price and any discounts or concessions allowed or reallowed may change from time to time. We may use underwriters
with whom we have a material relationship. We will describe in the prospectus supplement, naming the underwriter, the nature of
any such relationship.
By Dealers
If a dealer is utilized in the sale of any securities
offered by this prospectus, we will sell those securities to the dealer, as principal. The dealer may then resell the securities
to the public at varying prices to be determined by the dealer at the time of resale. We will set forth the names of the dealers
and the terms of the transaction in the applicable prospectus supplement.
By Agents
We may sell securities directly or through agents
we designate from time to time. We will name any agent involved in the offering and sale of securities and we will describe any
commissions we will pay the agent in the prospectus supplement. Unless the prospectus supplement states otherwise, our agent will
act on a best-efforts basis for the period of its appointment.
By Direct Sales
We may also directly sell securities offered
by this prospectus. In this case, no underwriters or agents would be involved. We will describe the terms of those sales in the
applicable prospectus supplement.
General Information
Underwriters, dealers and agents that participate
in the distribution of the securities offered by this prospectus may be deemed underwriters under the Securities Act, and any discounts
or commissions they receive from us and any profit on their resale of the securities may be treated as underwriting discounts and
commissions under the Securities Act.
We may authorize agents, dealers or underwriters
to solicit offers by certain types of institutional investors to purchase securities from us at the public offering price set forth
in the prospectus supplement pursuant to delayed delivery contracts providing for payment and delivery on a specified date in the
future. We will describe the conditions to these contracts and the commissions we must pay for solicitation of these contracts
in the prospectus supplement.
We may provide agents and underwriters with
indemnification against civil liabilities related to this offering, including liabilities under the Securities Act, or contribution
with respect to payments that the agents or underwriters may make with respect to these liabilities. Agents and underwriters may
engage in transactions with, or perform services for, us in the ordinary course of business.
Some or all of the securities we offer, other
than common stock, will be new issues of securities with no established trading market. Any underwriters may make a market in these
securities, but will not be obligated to do so and may discontinue any market making at any time without notice. We cannot guarantee
the liquidity of the trading markets for any securities.
We may enter into derivative transactions with
third parties, or sell securities not covered by this prospectus to third parties in privately negotiated transactions. If the
applicable prospectus supplement indicates, in connection with those derivatives, the third parties may sell securities covered
by this prospectus and the applicable prospectus supplement, including in short sale transactions. If so, the third parties may
use securities pledged by us or borrowed from us or others to settle those sales or to close out any related open borrowings of
stock, and may use securities received from us in settlement of those derivatives to close out any related open borrowings of stock.
The third parties in such sale transactions will be identified in the applicable prospectus supplement.
One or more firms, referred to as “remarketing
firms,” may also offer or sell the securities, if the prospectus supplement so indicates, in connection with a remarketing
arrangement upon their purchase. Remarketing firms will act as principals for their own accounts or as agents for us. These remarketing
firms will offer or sell the securities in accordance with the terms of the securities. The prospectus supplement will identify
any remarketing firm and the terms of its agreement, if any, with us and will describe the remarketing firm’s compensation.
Remarketing firms may be deemed to be underwriters in connection with the securities they remarket.
Any underwriter may engage in overallotment,
stabilizing transactions, short covering transactions and penalty bids in accordance with Regulation M under the Exchange Act.
Overallotment involves sales in excess of the offering size, which create a short position. This short sales position may involve
either “covered” short sales or “naked” short sales. Covered short sales are short sales made in an amount
not greater than the underwriters’ over-allotment option to purchase additional securities in this offering described above.
The underwriters may close out any covered short position either by exercising their over-allotment option or by purchasing securities
in the open market. To determine how they will close the covered short position, the underwriters will consider, among other things,
the price of securities available for purchase in the open market, as compared to the price at which they may purchase securities
through the over-allotment option. Naked short sales are short sales in excess of the over-allotment option. The underwriters must
close out any naked short position by purchasing securities in the open market. A naked short position is more likely to be created
if the underwriters are concerned that, in the open market after pricing, there may be downward pressure on the price of the securities
that could adversely affect investors who purchase securities in this offering. Stabilizing transactions permit bids to purchase
the underlying security for the purpose of fixing the price of the security so long as the stabilizing bids do not exceed a specified
maximum. Penalty bids permit the underwriters to reclaim a selling concession from a dealer when the securities originally sold
by the dealer are purchased in a covering transaction to cover short positions.
Any underwriters who are qualified market makers
on the NASDAQ Capital Market may engage in passive market making transactions in our common stock, preferred stock, warrants and
debt securities, as applicable, on the NASDAQ Capital Market in accordance with Rule 103 of Regulation M, during the
business day prior to the pricing of the offering, before the commencement of offers or sales of the securities. Passive market
makers must comply with applicable volume and price limitations and must be identified as passive market makers. In general, a
passive market maker must display its bid at a price not in excess of the highest independent bid for such security; if all independent
bids are lowered below the passive market maker’s bid, however, the passive market maker’s bid must then be lowered
when certain purchase limits are exceeded.
Similar to other purchase transactions, an
underwriter’s purchase to cover the syndicate short sales or to stabilize the market price of our securities may have the
effect of raising or maintaining the market price of our securities or preventing or mitigating a decline in the market price of
our securities. As a result, the price of our securities may be higher than the price that might otherwise exist in the open market.
The imposition of a penalty bid might also have an effect on the price of the securities if it discourages resales of the securities.
Neither we nor the underwriters make any representation
or prediction as to the effect that the transactions described above may have on the price of the securities. If such transactions
are commenced, they may be discontinued without notice at any time.
Our common stock is listed on The NASDAQ Capital
Market under the symbol “LPCN”.
LEGAL
MATTERS
Dorsey & Whitney LLP, Salt Lake City, Utah
will pass for us upon the validity of the securities being offered by this prospectus and applicable prospectus supplement, and
counsel named in the applicable prospectus supplement will pass upon legal matters for any underwriters, dealers or agents.
EXPERTS
The consolidated financial statements of Lipocine
Inc. as of December 31, 2013 and 2012, and for each of the years in the two-year period ended December 31, 2013, have been incorporated
by reference herein in reliance on the reports of KPMG LLP, independent registered public accounting firm, incorporated by reference
herein, and upon the authority of said firm as experts in accounting and auditing.
WHERE
YOU CAN FIND MORE INFORMATION
We are a reporting company and file annual,
quarterly and current reports, proxy statements and other information with the SEC. We have filed with the SEC a registration statement
on Form S-3 under the Securities Act with respect to the securities we are offering under this prospectus. This prospectus does
not contain all of the information set forth in the registration statement and the exhibits to the registration statement. For
further information with respect to us and the securities we are offering under this prospectus, we refer you to the registration
statement and the exhibits and schedules filed as a part of the registration statement. You may read and copy the registration
statement, as well as our reports, proxy statements and other information, at the SEC’s Public Reference Room at 100 F Street,
N.E., Washington, D.C. 20549. You can request copies of these documents by writing to the SEC and paying a fee for the copying
cost. Please call the SEC at 1-800-SEC-0330 for more information about the operation of the Public Reference Room. The SEC maintains
an internet site that contains reports, proxy and information statements, and other information regarding issuers that file electronically
with the SEC, where our SEC filings are also available. The address of the SEC’s web site is “http://www.sec.gov.”
We maintain a website at www.bsdmc.com. Information contained in or accessible through our website does not constitute a part of
this prospectus.
INCORPORATION
BY REFERENCE
The SEC allows us to “incorporate by reference”
information that we file with it into this prospectus, which means that we can disclose important information to you by referring
you to those documents. The information incorporated by reference is an important part of this prospectus. The information incorporated
by reference is considered to be a part of this prospectus, and information that we file later with the Commission will automatically
update and supersede information contained in this prospectus and any accompanying prospectus supplement. We incorporate
by reference the documents listed below that we have previously filed with the Commission:
|
|
·
|
Our Annual Report on Form 10-K for the year ended December 31, 2013;
|
|
|
·
|
Our Definitive Proxy Statement filed on Form 14A on April 29, 2014;
|
|
|
·
|
Our Quarterly Reports on Form 10-Q for the quarters ended March 31, 2014 and June 30, 2014;
|
|
|
·
|
Our Current Reports on Form 8-K filed on January 7, 2014 (only with respect to item 5.02), January 24, 2014, February 10, 2014 (only with respect to item 8.01), March 18, 2014, April 16, 2014, April 29, 2014, May 15, 2014, June 11, 2014, July 15, 2014, September 8, 2014 and September 24, 2014; and
|
|
|
·
|
The description of our common stock, which is contained in the Registration Statement on Form 8-A, as filed with the SEC on March 18, 2014, including any amendment or report filed for the purpose of updating such description.
|
We also incorporate by reference into this prospectus
additional documents that we may file with the SEC under Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act, excluding,
in each case, information deemed furnished and not filed until we sell all of the securities we are offering or the termination
of the offering. Any statements contained in a previously filed document incorporated by reference into this prospectus is deemed
to be modified or superseded for purposes of this prospectus to the extent that a statement contained in this prospectus, or in
a subsequently filed document also incorporated by reference herein, modifies or supersedes that statement.
We will provide to each person, including any
beneficial owner, to whom a prospectus is delivered, without charge upon written or oral request, a copy of any or all of the information
that has been incorporated by reference into this prospectus but not delivered with the prospectus, including exhibits that are
specifically incorporated by reference into such documents. Requests should be directed to: Lipocine Inc., Attention: Investor
Relations, 675 Arapeen Drive, Suite 202, Salt Lake City, Utah 84108 and our telephone number is (801) 994-7383.
Up to $20,000,000
Common Stock


March 6, 2017
Lipocine (NASDAQ:LPCN)
Historical Stock Chart
From Aug 2024 to Sep 2024
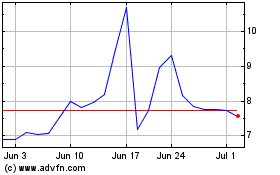
Lipocine (NASDAQ:LPCN)
Historical Stock Chart
From Sep 2023 to Sep 2024