Avita Medical to Participate in the 3rd Annual Biotech and Money / Medtech and Money London 2017 Conference
January 24 2017 - 9:02PM
Business Wire
Avita Medical Ltd (ASX: AVH) (OTCQX: AVMXY), a
regenerative medicine company specializing in new treatments for
wounds and skin defects, said today that Adam Kelliher, Avita’s
Chief Executive Officer, will be a panel member at the Biotech and
Money / Medtech and Money conference taking place in London, UK on
7-8 February, 2017.
The 3rd annual Biotech and Money / Medtech and Money London is a
two-day conference and partnering event providing the education,
strategies, solutions and contacts that life science companies need
to enable more effective funding, investment, business planning and
partnering within their businesses. The conference is a senior
level educational, best practices and networking event that
includes keynote panels with leading life science funds, discussion
groups and the opportunity to participate in 1x1 meetings.
Panel details:
Title:
Anatomy of a Medtech private financial raise
Date:
Wednesday, 8 February, 2017
Time:
11:20 GMT
Location:
etc. venues, 155 Bishopsgate, London, EC2M 4AN, United Kingdom
“With over 400 attendees including over 150 investors, we are
able to connect with industry leaders and top life science
investors to discuss Avita’s commercial, regulatory, scientific and
clinical research developments across the range of indications for
which the Company’s portfolio of products (ReCell®, ReGenerCell™
and ReNovaCell™) are in use around the world,” stated Mr.
Kelliher.
ABOUT AVITA MEDICAL LIMITED
Avita Medical develops and distributes regenerative products for
the treatment of a broad range of wounds, scars and skin defects.
Avita’s patented and proprietary collection and application
technology provides innovative treatment solutions derived from a
patient’s own skin. The Company’s lead product, ReCell®, is used in
the treatment of a wide variety of burns, plastic, reconstructive
and cosmetic procedures. ReCell® is patented, CE-marked for Europe,
TGA-registered in Australia, and CFDA-cleared in China. In the
United States, ReCell® is an investigational device limited by
federal law to investigational use, and a pivotal U.S. approval
trial is underway. To learn more, visit www.avitamedical.com.
View source
version on businesswire.com: http://www.businesswire.com/news/home/20170124006659/en/
Avita Medical LtdAdam Kelliher, +44 (0)1763 269 772Chief
Executive Officerakelliher@avitamedical.comorTim Rooney, +1 (818)
356-9400Chief Financial Officertrooney@avitamedical.comorGabriel
Chiappini, +61 (0)8 9474 7738Company
Secretarygabriel@laurus.net.auorUSAWestwicke
PartnersJamar Ismail, +1 (415)
513-1282jamar.ismail@westwicke.comorAustraliaMonsoon
CommunicationsDean FeltonPhone: +61 (0)3 9620 3333Mobile: +61
(0)411 698 499deanf@monsoon.com.au
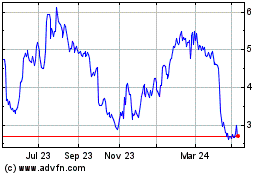
AVITA Medical (ASX:AVH)
Historical Stock Chart
From Mar 2024 to Apr 2024
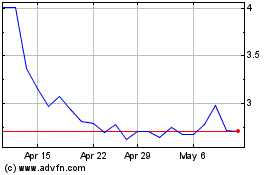
AVITA Medical (ASX:AVH)
Historical Stock Chart
From Apr 2023 to Apr 2024