Unite Cellular Immunotherapy Expertise to
Accelerate Clinical Translation of Off-the-Shelf Products
Offering Broad Patient Access
Fate Therapeutics, Inc. (NASDAQ:FATE), a biopharmaceutical company
dedicated to the development of programmed cellular immunotherapies
for cancer and immune disorders, announced today a partnership with
Memorial Sloan Kettering Cancer Center for the development of
off-the-shelf T-cell product candidates using engineered
pluripotent cell lines. Research and development activities under
the multi-year collaboration will be led by Michel Sadelain, M.D.,
Ph.D., Director of the Center for Cell Engineering and the Stephen
and Barbara Friedman Chair at Memorial Sloan Kettering Cancer
Center.
“This partnership brings together Memorial Sloan Kettering’s
excellence in the manufacture and delivery of cell-based
immunotherapies, and our established expertise in pluripotent cell
generation, engineering and differentiation,” said Scott Wolchko,
President and Chief Executive Officer of Fate Therapeutics.
“Together, we are at the forefront of an off-the-shelf paradigm
shift, seeking to broaden patient access to revolutionary T-cell
immunotherapies through a renewable, robust and standardized
product approach.”
“Engineering therapeutic attributes into pluripotent cell lines,
such as antigen specificity, lack of alloreactivity, enhanced
persistence and histocompatibility, is a breakthrough approach to
renewably generate potent T-cell immunotherapies,” said Dr.
Sadelain. “This unique approach offers the prospect for
off-the-shelf delivery of T-cell immunotherapies with enhanced
safety and therapeutic potential at the scale necessary to serve
significant numbers of patients.”
The collaboration unites research, preclinical development and
manufacturing work currently being conducted independently at Fate
Therapeutics and Memorial Sloan Kettering to accelerate the
clinical translation of T-cell product candidates derived from
engineered pluripotent cells. Collectively, the groups have amassed
significant and complementary expertise necessary to deliver
off-the-shelf T-cell immunotherapies, including the engineering,
maintenance and expansion of induced pluripotent cell lines and the
scalable generation of T cells with enhanced safety profiles and
effector functions.
In connection with the partnership, Fate Therapeutics has
exclusively licensed from Memorial Sloan Kettering foundational
intellectual property covering induced pluripotent cell-derived
immune cells, including T cells and NK cells derived from
pluripotent cells engineered with chimeric antigen receptors, for
human therapeutic use. Additionally, Fate Therapeutics maintains an
option to exclusively license intellectual property arising from
all research and development activities under the
collaboration.
Off-the-Shelf Immunotherapy OpportunityCellular
immunotherapies are poised to transform the treatment of cancer and
immunological conditions. However, cellular immunotherapies
currently undergoing clinical investigation are patient-specific
and their delivery requires the extraction, engineering, expansion
and re-introduction of each individual patient’s T cells. This
multi-step manufacturing process is logistically challenging and
complex, and significant hurdles remain to ensure that
patient-specific T-cell immunotherapies can be efficiently and
consistently manufactured, and safely and reliably delivered, at
the scale necessary to support broad patient access and wide-spread
commercialization.
Induced pluripotent cells possess the unique dual properties of
self-renewal and differentiation potential into all cell types of
the body including T cells. Similar to master cell lines used for
the manufacture of monoclonal antibodies, engineered pluripotent
cell lines can repeatedly deliver clonal populations of T cells
with broad histocompatibility and enhanced effector functions.
These highly-stable pluripotent cell lines have the potential to
serve as a renewable cell source for the consistent manufacture of
homogeneous populations of effector cells for the treatment of many
thousands of patients.
Exclusive License & Development PlanThrough
the three-year collaboration, the group aims to leapfrog the
field’s current patient-specific approach to T-cell immunotherapy.
Over the last decade, Fate Therapeutics has developed a
proprietary, patent-protected platform to efficiently generate,
genetically engineer, isolate and bank pluripotent cell lines.
Memorial Sloan Kettering is leading the field in generating
pluripotent cell-derived, tumor-targeting T cells that are capable
of profound tumor clearance in vivo. The scientific teams will
combine forces to create pluripotent cell lines that have been
engineered for enhanced antigen specificity and functionality,
optimize T-cell differentiation protocols, and clinically translate
off-the-shelf engineered T-cell product candidates.
New Subsidiary FormedFate Therapeutics has also
launched a new venture company, Tfinity Therapeutics, Inc., which
will focus exclusively on the advancement of off-the-shelf T-cell
immunotherapies across a wide range of diseases using Fate’s
proprietary, patent-protected pluripotent cell platform. Fate
Therapeutics has an intellectual property portfolio consisting of
over 60 issued patents and 90 pending patent applications, which
are owned or exclusively licensed by Fate Therapeutics, that cover
compositions and methods critical for deriving, engineering,
maintaining and differentiating induced pluripotent cells. Tfinity
Therapeutics is a majority-owned subsidiary of Fate Therapeutics,
and holds an option to license from Fate Therapeutics intellectual
property covering pluripotent cell-derived T-cell
immunotherapies.
About Fate Therapeutics, Inc.Fate Therapeutics
is a biopharmaceutical company dedicated to the development of
programmed cellular immunotherapies for cancer and immune
disorders. The Company’s cell therapy pipeline is comprised of
immuno-oncology programs, including off-the-shelf NK- and T-cell
cancer immunotherapies derived from engineered induced pluripotent
cells, and immuno-regulatory programs, including hematopoietic cell
immunotherapies for protecting the immune system of patients
undergoing hematopoietic cell transplantation and for regulating
autoimmunity. Its adoptive cell therapy programs are based on the
Company’s novel ex vivo cell programming approach, which it applies
to modulate the therapeutic function and direct the fate of immune
cells. Fate Therapeutics is headquartered in San Diego, CA. For
more information, please visit www.fatetherapeutics.com.
Forward-Looking StatementsThis release contains
"forward-looking statements" within the meaning of the Private
Securities Litigation Reform Act of 1995, including statements
regarding the impact, benefits, timing, and conduct of the
partnership between Memorial Sloan Kettering and Fate Therapeutics,
as well as the capabilities, expertise, and responsibilities of
each, and the therapeutic potential of any programmed cellular
immunotherapies derived from induced pluripotent cells developed
under the partnership. These and any other forward-looking
statements in this release are based on management's current
expectations of future events and are subject to a number of risks
and uncertainties that could cause actual results to differ
materially and adversely from those set forth in or implied by such
forward-looking statements. These risks and uncertainties include,
but are not limited to, risks associated with the success, cost,
and timing of research and product development activities under the
collaboration, the risk of cessation or delay of any development
activities under the collaboration for a variety of reasons,
including any inability to develop or manufacture off-the-shelf
T-cell products, and the risk that any off-the-shelf T-cell
therapies developed under the collaboration may not be suitable for
therapeutic applications and may not provide the anticipated
therapeutic benefits. For a discussion of other risks and
uncertainties, and other important factors, any of which could
cause our actual results to differ from those contained in the
forward-looking statements, see the risks and uncertainties
detailed in the Company’s periodic filings with the Securities and
Exchange Commission, including but not limited to the Company’s
most recently filed periodic report, and from time to time the
Company’s other investor communications. Fate Therapeutics is
providing the information in this release as of this date and does
not undertake any obligation to update any forward-looking
statements contained in this release as a result of new
information, future events or otherwise.
Contact:
Jesse Baumgartner
Stern Investor Relations, Inc.
212.362.1200
jesse@sternir.com
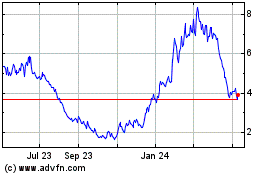
Fate Therapeutics (NASDAQ:FATE)
Historical Stock Chart
From Mar 2024 to Apr 2024
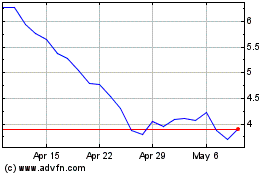
Fate Therapeutics (NASDAQ:FATE)
Historical Stock Chart
From Apr 2023 to Apr 2024