ACADIA Pharmaceuticals Appoints James (Randy) R. Owen, M.D., as Senior Vice President, Clinical Development & Chief Medical O...
January 25 2016 - 9:00AM
Business Wire
ACADIA Pharmaceuticals Inc. (NASDAQ: ACAD), a biopharmaceutical
company focused on the development and commercialization of
innovative medicines to address unmet medical needs in central
nervous system (CNS) disorders, today announced the appointment of
James (Randy) R. Owen, M.D., as Senior Vice President, Clinical
Development and Chief Medical Officer. Dr. Owen will be responsible
for evolving and executing the clinical development programs for
ACADIA’s product candidates and report to Serge Stankovic, M.D.,
M.S.P.H., ACADIA’s Executive Vice President, Head of Research and
Development.
“Randy brings significant expertise in clinical research and
development along with a deep knowledge of CNS science,” said Steve
Davis, ACADIA’s President and Chief Executive Officer. “He has led
the development and approval of important therapeutics in CNS and
will be a valued addition to the ACADIA team as we seek to help
patients who suffer from serious neurological and psychiatric
disorders.”
Dr. Owen most recently served as Vice President, U.S. Clinical
Affairs at Lundbeck LLC from 2010 to 2016. During his tenure, his
team of medical experts and scientists made significant
contributions in drug development across a wide range of
neurological and psychiatric disorders, including innovative
development programs in schizophrenia, Alzheimer’s disease and
stroke, as well as pediatric and adult orphan indications. Prior to
joining Lundbeck, Dr. Owen served as Group Director, Global
Clinical Research at Bristol-Myers Squibb Company where he co-led
the life-cycle management of Abilify® during a period of product
extensions in psychiatric indications in the United States and
Europe. Dr. Owen received his B.A. at Haverford College and his
M.D. at East Tennessee State University, Quillen College of
Medicine. He completed his residency at Emory University and is
board certified in psychiatry.
About ACADIA Pharmaceuticals
ACADIA is a biopharmaceutical company focused on the development
and commercialization of innovative medicines to address unmet
medical needs in central nervous system disorders. ACADIA has a
pipeline of product candidates led by NUPLAZID™ (pimavanserin), for
which we have submitted a New Drug Application (NDA) in Parkinson’s
disease psychosis to the FDA and which has the potential to be the
first drug approved in the United States for this condition. The
FDA has classified the NUPLAZID NDA as having Priority Review
status. Pimavanserin is also in Phase II development for
Alzheimer’s disease psychosis and has successfully completed a
Phase II trial in schizophrenia. ACADIA also has clinical-stage
programs for glaucoma and, in collaboration with Allergan, Inc.,
for chronic pain. ACADIA maintains a website at
www.acadia-pharm.com to which we regularly post copies of our press
releases as well as additional information and through which
interested parties can subscribe to receive e-mail alerts.
Forward-Looking Statements
Statements in this press release that are not strictly
historical in nature are forward-looking statements. These
statements include but are not limited to statements related to the
potential for NUPLAZID (pimavanserin) to be the first drug approved
in the United States for Parkinson’s disease psychosis (PDP) and
the potential timing of such approval, if approved at all, by the
FDA; the potential contributions of Dr. Owen to ACADIA; the
progress, timing and results of ACADIA’s drug discovery and
development programs, either alone or with a partner, including the
progress and expected timing of clinical trials; and the benefits
to be derived from ACADIA’s product candidates, including
pimavanserin. These statements are only predictions based on
current information and expectations and involve a number of risks
and uncertainties. Actual events or results may differ materially
from those projected in any of such statements due to various
factors, including the risks and uncertainties inherent in drug
discovery, development, approval and commercialization, and
collaborations with others, and the fact that past results of
clinical trials and past regulatory decisions may not be indicative
of future trial results or future regulatory decisions,
respectively. For a discussion of these and other factors, please
refer to ACADIA’s annual report on Form 10-K for the year ended
December 31, 2014 as well as ACADIA’s subsequent filings with the
Securities and Exchange Commission. You are cautioned not to place
undue reliance on these forward-looking statements, which speak
only as of the date hereof. This caution is made under the safe
harbor provisions of the Private Securities Litigation Reform Act
of 1995. All forward-looking statements are qualified in their
entirety by this cautionary statement and ACADIA undertakes no
obligation to revise or update this press release to reflect events
or circumstances after the date hereof, except as required by
law.
View source
version on businesswire.com: http://www.businesswire.com/news/home/20160125005593/en/
ACADIA Pharmaceuticals Inc.Lisa Barthelemy, Director of Investor
Relations(858) 558-2871
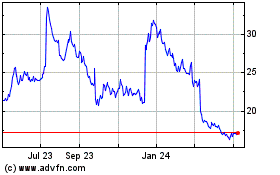
Acadia Pharmaceuticals (NASDAQ:ACAD)
Historical Stock Chart
From Aug 2024 to Sep 2024
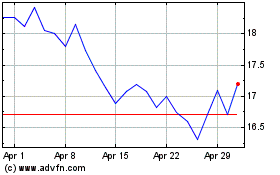
Acadia Pharmaceuticals (NASDAQ:ACAD)
Historical Stock Chart
From Sep 2023 to Sep 2024