Graphite Bio Gets Fast Track Designation for GPH101 Sickle Cell Treatment
May 03 2022 - 8:50AM
Dow Jones News
By Chris Wack
Graphite Bio Inc. said Tuesday that the U.S. Food and Drug
Administration has granted fast track designation to GPH101 for the
treatment of sickle cell disease.
The company said that GPH101 is an investigational
next-generation gene-edited autologous hematopoietic stem cell
therapy designed to directly correct the genetic mutation that
causes SCD.
Graphite Bio said it is continuing to enroll patients in its
Phase 1/2 trial, and expects to dose its first patient later this
year, with initial proof-of-concept data anticipated next year.
The FDA's fast track program speeds up the development and
review of new drugs or biologics that are intended to treat serious
or life-threatening conditions and demonstrate the potential to
address unmet medical needs. GPH101 was previously granted orphan
drug designation by the FDA.
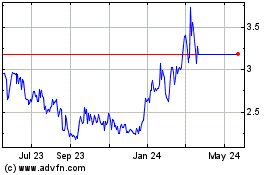
Graphite Bio shares were up 9% to $4.36 in premarket
trading.
Write to Chris Wack at chris.wack@wsj.com
(END) Dow Jones Newswires
May 03, 2022 08:35 ET (12:35 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
Graphite Bio (NASDAQ:GRPH)
Historical Stock Chart
From May 2024 to Jun 2024
Graphite Bio (NASDAQ:GRPH)
Historical Stock Chart
From Jun 2023 to Jun 2024