- PanOptix delivers an exceptional combination of near,
intermediate and distance vision and substantially reduces the need
for glasses1,2
- More than ninety-nine percent of PanOptix patients in the
FDA clinical study said they would choose the same lens
again1
- U.S. surgeon training and patient awareness efforts to begin
immediately with ramp up into early 2020
Alcon (SIX/NYSE:ALC), the global leader in eye care, today
announced the Food and Drug Administration (FDA) approval and its
initial commercial launch of AcrySof® IQ PanOptix® Trifocal
Intraocular Lens (IOL), the first and only trifocal lens for U.S.
patients undergoing cataract surgery. PanOptix is clinically shown
to deliver an exceptional combination of near, intermediate and
distance vision while significantly reducing the need for glasses
after surgery.1,2
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20190826005686/en/
AcrySof® IQ PanOptix® Trifocal IOL
Packaging (Photo: Business Wire)
PanOptix is already one of the leading presbyopia-correcting
IOLs in more than 70 countries. It is designed for today’s active
lifestyles, from viewing mobile devices and computer screens to
high-quality distance vision in a range of lighting conditions. The
new lens uses ENLIGHTEN® Optical Technology, a proprietary design
that optimizes intermediate vision without compromising near and
distance vision. Available in spherical and toric designs, PanOptix
is built on Alcon’s proven AcrySof IQ IOL platform that has been
implanted in more than 120 million eyes globally.
“We are proud to bring this new class of IOLs to the largest eye
care market in the world,” said David J. Endicott, Chief Executive
Officer of Alcon. “We are leveraging Alcon’s years of experience
with PanOptix in other countries to provide the best possible
training and support for U.S. surgeons. Our goal is to ensure
optimal outcomes for cataract patients looking to correct their
vision at all distances, with the vast majority of them never
needing to wear glasses post-surgery.”
The FDA approval of PanOptix was based on a pivotal study at 12
investigational sites in the U.S. With this single trifocal lens
design, PanOptix patients demonstrated exceptional, uninterrupted
vision. The results also showed high patient satisfaction with more
than ninety-nine percent of PanOptix patients saying they would
choose the same lens again.1
More than 4 million cataract surgeries are performed each year
in the U.S., which is projected to increase by more than 16 percent
by the end of 2024.3,4 The presbyopia-correcting IOL market is
expected to grow 60 percent by the end of 2024.4
“The U.S. ophthalmology community has been eagerly awaiting FDA
approval of PanOptix. While participating in the clinical trial, I
saw first-hand the impact of this lens on my patients,” said Kerry
Solomon, M.D., Carolina Eyecare Physicians, PanOptix clinical trial
investigator. “We know patients undergoing cataract surgery today
want a replacement lens that delivers the vision they need to live
full, active lives without always being dependent on glasses.
PanOptix is an excellent lens option to address these needs and is
clinically proven to deliver an outstanding range of vision.”
Alcon plans to begin training U.S. ophthalmologists and making
inventory of PanOptix and PanOptix Toric IOLs available through the
rest of 2019 into early 2020. As part of its patient education
efforts, the company will soon launch a dedicated PanOptix website
and multimedia awareness campaign. A similar effort is planned for
eye care professionals with special launch activities slated for
the annual meeting of the American Academy of Ophthalmology in
October.
About Cataracts
A cataract is a cloudy area in the natural lens of the eye that
affects vision. As a cataract develops, the eye’s lens gradually
becomes hard and cloudy allowing less light to pass through, which
makes it more difficult to see. The vast majority of cataracts
result from normal aging, but radiation exposure, taking steroids,
diabetes and eye trauma can accelerate their development.5
Cataracts are the most common age-related eye condition and the
leading cause of preventable blindness.6 Twenty million people in
the U.S. age 40 and older have cataracts.5 Cataracts are treated by
removing the eye’s cloudy natural lens and surgically replacing it
with an intraocular lens or IOL. More than 98 percent of cataract
surgeries are considered successful, and patients typically can
return to their normal routines within 24 hours.7
About Presbyopia
Presbyopia is a common, age-related vision condition in which
people have difficulty focusing on things up close. It involves the
gradual loss of the eye’s ability to focus on close objects, such
as smart phones, computers, books and menus.8 Almost everyone will
experience presbyopia to some degree as they age, with symptoms
often first appearing as an individual enters their 40s and
continues to worsen into their 60s.8 In the U.S., an estimated 112
million people experience vision issues due to presbyopia – a
number that’s expected to continue increasing.9 The condition is
not a disease, so it cannot be cured; however, there are safe and
effective ways to correct presbyopia, including eye glasses,
contact lenses or refractive surgery.
About PanOptix
The AcrySof® IQ PanOptix® Trifocal IOL is a type of multifocal
IOL used to focus images clearly onto the back of your eye (retina)
to allow clear vision after the cataract removal. In addition, the
center of the AcrySof® IQ PanOptix® Trifocal IOL allows for better
near (reading) vision and intermediate (computer work) vision
versus what a monofocal lens would provide.
Potential Side Effects: Due to the design of multifocal IOLs,
there are some side effects that can be associated with the
AcrySof® IQ PanOptix® Trifocal IOL models. These may be worse than
with a monofocal IOL, including visual disturbances such as glare,
rings around lights, starbursts (rays around light sources), and
reduced contrast sensitivity (decrease in ability to distinguish
objects from their background, especially in dim lighting). These
side effects may make it more difficult to see while driving at
night or completing tasks in low lighting conditions such as at
night or in fog, or in a dimly lit room after surgery as compared
to before surgery.
Further, a toric IOL corrects astigmatism only when it is placed
in the correct position in the eye. There is a possibility that the
toric IOL could be placed incorrectly or could move within the eye.
If the toric lens is not positioned correctly following surgery,
the change in your astigmatism correction by the IOL, along with
any necessary correction with glasses, may cause visual
distortions. If the lens rotates in your eye, you may need
additional surgery to reposition or replace the IOL.
Disclaimer
This press release contains “forward-looking statements” within
the meaning of the safe harbor provisions of the United States
Private Securities Litigation Reform Act of 1995. Forward-looking
statements can be identified by words such as: “anticipate,”
“intend,” “commitment,” “look forward,” “maintain,” “plan,” “goal,”
“seek,” “believe,” “project,” “estimate,” “expect,” “strategy,”
“future,” “likely,” “may,” “should,” “will” and similar references
to future periods.
Forward-looking statements are neither historical facts nor
assurances of future performance. Instead, they are based only on
our current beliefs, expectations and assumptions regarding the
future of our business, future plans and strategies, and other
future conditions. Because forward-looking statements relate to the
future, they are subject to inherent uncertainties and risks that
are difficult to predict. Some of these factors are discussed in
our filings with the United States Securities and Exchange
Commission, including our Form 20-F. Should one or more of these
uncertainties or risks materialize, or should underlying
assumptions prove incorrect, actual results may vary materially
from those anticipated. Therefore, you should not rely on any of
these forward-looking statements.
Forward-looking statements in this press release speak only as
of the date of its filing, and we assume no obligation to update
forward-looking statements as a result of new information, future
events or otherwise.
About Alcon
Alcon helps people see brilliantly. As the global leader in eye
care with a heritage spanning more than seven decades, we offer the
broadest portfolio of products to enhance sight and improve
people’s lives. Our Surgical and Vision Care products touch the
lives of more than 260 million people in over 140 countries each
year living with conditions like cataracts, glaucoma, retinal
diseases and refractive errors. Our more than 20,000 associates are
enhancing the quality of life through innovative products,
partnerships with eye care professionals and programs that advance
access to quality eye care. Learn more at www.alcon.com.
Connect with us on Facebook LinkedIn
1 AcrySof IQ PanOptix Directions For Use. 2 Compared to AcrySof®
monofocal IOL. 3 Market Scope. 2017 Ophthalmic Surgical Instrument
Report. 4 Market Scope. 2019 IOL Market Report. April 2019. 5
National Institutes of Health (NIH), National Eye Institute (NEI),
Facts About Cataracts. Accessed July 19, 2019. Available at:
http://www.nei.nih.gov/health/cataract/cataract_facts.asp. 6
Centers for Disease Control and Prevention, Vision Health
Initiative. Accessed July 19, 2019. Available at:
https://www.cdc.gov/visionhealth/basics/ced/index.html. 7 Cleveland
Clinic, Cataracts. Accessed August 12, 2019. Available at:
https://my.clevelandclinic.org/health/diseases/8589-cataracts. 8
Mayo Clinic, Presbyopia. Accessed August 12, 2019. Available at:
https://www.mayoclinic.org/diseases-conditions/presbyopia/symptoms-causes/syc-20363328.
9 American Optometric Association: Optometric Clinical Practice
Guideline, Care of the Patient with Presbyopia. Accessed August 12,
2019. Available at:
https://www.aoa.org/documents/optometrists/CPG-17.pdf.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20190826005686/en/
Investor Relations Christina
Cheng + 41 589 112 110 (Geneva) + 1 817 615 2789 (Fort Worth)
investor.relations@alcon.com Media
Relations Steven Smith + 41 589 112 111 (Geneva) + 1 817
551 8057 (Fort Worth) globalmedia.relations@alcon.com Blake Overby
+ 1 817 551 4328 (Office) + 1 682 321 2897 (Mobile)
blake.overby@alcon.com
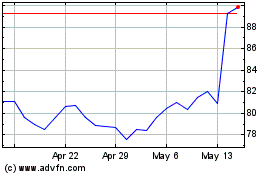
Alcon (NYSE:ALC)
Historical Stock Chart
From Jun 2024 to Jul 2024
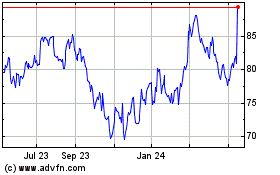
Alcon (NYSE:ALC)
Historical Stock Chart
From Jul 2023 to Jul 2024