Bacterial Biofilms, Responsible for Up to 75% of All Infectious Diseases in America, Explored in New Webinar From Quidel Corporation
August 24 2022 - 9:00AM
Business Wire
The National Institutes of Health reports that bacterial
biofilms are responsible for up to 75% of all infectious diseases
in humans, including infections of wounds, the lungs and the
urinary tract. This important medical reality is now being
addressed in a new webinar made available by Quidel Corporation,
the California-based diagnostic health care manufacturer known for
successfully developing rapid diagnostic health solutions.
The webinar, available at
https://education.quidel.com/educational-categories, discusses the
basics of bacterial biofilms including when and how they develop,
its composition and factors that contribute to antibiotic
resistance and virulence. The session also explores the biofilms in
the context of Lyme disease and the potential link between chronic
Lyme and biofilm formation. Sponsored in collaboration with Global
Lyme Alliance, the webinar is conducted by Cezar M. Khursigara,
Ph.D., professor of molecular and cellular biology at the
University of Guelph in Ontario, Canada.
“Biofilms are everywhere in our environment and in many common
industries,” says Dr. Khursigara. “It is important to understand
how these biofilms are formed and their maturation as well as their
dispersal. This is especially true for people with Cystic Fibrosis
who are prone to the buildup of mucus in the lungs, creating
perfect conditions for bacteria to colonize and form biofilms,
which can lead to chronic inflammation and increased morbidity and
mortality.”
During the webinar, Dr. Khursigara also discusses the
relationship between bacterial biofilms and Lyme disease, noting
that Lyme borreliosis is increasing worldwide with approximately
476,000 new cases annually in the United States and 85,000 cases in
Europe each year. “Lyme borreliosis is the most common tick-borne
disease and has the potential to affect multiple organ systems,
including the skin, joints, heart, endocrine glands,
gastrointestinal tract and the nervous system. It may even develop
into chronic infections,” he says.
Once Lyme disease is suspected, early detection is critical. For
rapid point-of-care testing Dr. Khursigara points to the Sofia® 2
Lyme FIA test by Quidel, the only FDA-cleared rapid-point-of-care
test on the market, which provides the patient and physician with
indicative results within 15 minutes, as opposed to days, which has
historically been the norm. Performed in the privacy of a doctor’s
office or local clinic, it is also the only test that can get
results from a simple finger prick of blood. “Not having to wait
days for test results allows clinicians to more rapidly treat those
patients with positive results while more quickly pursuing other
diagnosis and treatment for those who test negative,” says Dr.
Khursigara.
The “Bacterial Biofilms” webinar is the latest in an ongoing
series sponsored by Quidel that has featured noted scientists,
researchers and physicians discussing issues surrounding the
prevalence of Lyme Disease in America, including ways to reduce the
chances of infection, early detection and treatment options. The
webinars are directed toward physicians, allied health
professionals, health researchers, representatives of physician
offices, laboratories and urgent care centers, and others
interested in the subject.
About Quidel Corporation
Quidel Corporation (Nasdaq: QDEL) is a leading
manufacturer of diagnostic solutions at the point of care,
delivering a continuum of rapid-testing technologies that further
improve the quality of health care throughout the globe. An
innovator for over 40 years in the medical device industry, Quidel
pioneered the first FDA-cleared point-of-care test for influenza in
1999 and was the first to market a rapid SARS-CoV-2 antigen test in
the U.S. Under trusted brand names, Sofia®, Solana®, Lyra®, Triage®
and QuickVue®, Quidel’s comprehensive product portfolio includes
tests for a wide range of infectious diseases, cardiac and
autoimmune biomarkers as well as a host of products to detect
COVID-19. With products made in America, Quidel’s mission is to
provide patients with immediate and frequent access to highly
accurate, affordable testing for the good of our families, our
communities and the world. For more information about Quidel, visit
quidel.com.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220824005166/en/
Jim Yeager breakwhitelight (for Quidel
Corporation) jim@breakwhitelight.com Mobile:
818-264-6812
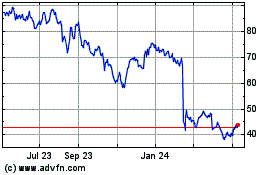
QuidelOrtho (NASDAQ:QDEL)
Historical Stock Chart
From Jun 2024 to Jul 2024
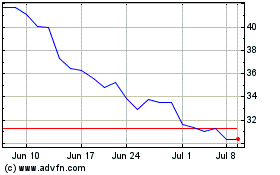
QuidelOrtho (NASDAQ:QDEL)
Historical Stock Chart
From Jul 2023 to Jul 2024