Gamida Cell to Present at Society for Immunotherapy of Cancer's (SITC) 36th Annual Meeting
October 19 2021 - 8:00AM
Business Wire
Gamida Cell Ltd. (Nasdaq: GMDA), an advanced cell therapy
company committed to cures for cancer and other serious diseases,
today announced that data evaluating the company’s NAM-enabled NK
cell platform will be presented at the Society for Immunotherapy of
Cancer’s 36th Annual Meeting (SITC 2021) taking place in
Washington, DC, and virtually November 10-14, 2021.
Details about the SITC poster presentations are as
follows:
Title: Cytotoxicity of nicotinamide enhanced natural
killer cells GDA-201 is based on metabolic modulation as
demonstrated by AI assisted analysis of NK cell transcriptome and
metabolome Abstract number: 217 Time: Friday,
November 12, 2021, 7:00 a.m. – 8:30 p.m. EST Location: Hall
E
Title: Nicotinamide rejuvenates ex-vivo expanded NK cells
and enhances their tumor killing capacity Abstract Number:
162 Time: Saturday, November 13, 2021, 7:00 a.m. – 8:30 p.m.
EST Location: Hall E
About GDA-201
Gamida Cell applied the capabilities of its nicotinamide
(NAM)-enabled cell expansion technology to develop GDA-201, an
innate NK cell immunotherapy for the treatment of hematologic and
solid tumors in combination with standard of care antibody
therapies. GDA-201, the lead candidate in the NAM-enabled NK cell
pipeline, has demonstrated promising initial clinical trial
results, as reported at the 2020 American Society of Hematology
(ASH) Annual Meeting & Exposition1. GDA-201 addresses key
limitations of NK cells by increasing the cytotoxicity and in vivo
retention and proliferation in the bone marrow and lymphoid organs.
Furthermore, GDA-201 improves antibody-dependent cellular
cytotoxicity (ADCC) and tumor targeting of NK cells. For more
information about GDA-201, please visit
https://www.gamida-cell.com.
GDA-201 is an investigational therapy, and its safety and
efficacy have not been established by the FDA or any other health
authority.
About Gamida Cell
Gamida Cell is pioneering a diverse immunotherapy pipeline of
potentially curative cell therapies for patients with solid tumor
and blood cancers and other serious blood diseases. We apply a
proprietary expansion platform leveraging the properties of NAM to
allogeneic cell sources including umbilical cord blood-derived
cells and NK cells to create therapies with potential to redefine
standards of care. These include omidubicel, an investigational
product with potential as a life-saving alternative for patients in
need of bone marrow transplant, and a line of modified and
unmodified NAM-enabled NK cells targeted at solid tumor and
hematological malignancies. For additional information, please
visit www.gamida-cell.com or follow Gamida Cell on LinkedIn,
Twitter, Facebook or Instagram at @GamidaCellTx.
Cautionary Note Regarding Forward Looking Statements
This press release contains forward-looking statements as that
term is defined in the Private Securities Litigation Reform Act of
1995, including with respect to timing of initiation and progress
of, and data reported from, the clinical trials of Gamida Cell’s
product candidates, anticipated regulatory filings (including the
submission of the BLA for omidubicel to the FDA), commercialization
planning efforts, the potentially life-saving or curative
therapeutic and commercial potential of omidubicel, and Gamida
Cell’s expectations regarding its projected cash to be used for
operating activities and cash runway. Any statement describing
Gamida Cell’s goals, expectations, financial or other projections,
intentions or beliefs is a forward-looking statement and should be
considered an at-risk statement. Such statements are subject to a
number of risks, uncertainties and assumptions, including those
related to the impact that the COVID-19 pandemic could have on our
business, and including the scope, progress and expansion of Gamida
Cell’s clinical trials and ramifications for the cost thereof;
clinical, scientific, regulatory and technical developments; and
those inherent in the process of developing and commercializing
product candidates that are safe and effective for use as human
therapeutics, and in the endeavor of building a business around
such product candidates. In light of these risks and uncertainties,
and other risks and uncertainties that are described in the Risk
Factors section and other sections of Gamida Cell’s Annual Report
on Form 20-F, filed with the Securities and Exchange Commission
(SEC) on March 9, 2021, as amended, and other filings that Gamida
Cell makes with the SEC from time to time (which are available at
http://www.sec.gov), the events and circumstances discussed in such
forward-looking statements may not occur, and Gamida Cell’s actual
results could differ materially and adversely from those
anticipated or implied thereby. Although Gamida Cell’s
forward-looking statements reflect the good faith judgment of its
management, these statements are based only on facts and factors
currently known by Gamida Cell. As a result, you are cautioned not
to rely on these forward-looking statements.
1 Bachanova, et al. ASH 2020. Abstract #63.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20211019005369/en/
For investors: Courtney Turiano Stern Investor Relations, Inc.
Courtney.Turiano@sternir.com 1-212-362-1200
For media: Rhiannon Jeselonis Ten Bridge Communications
rhiannon@tenbridgecommunications.com 1-978-417-1946
Gamida Cell (NASDAQ:GMDA)
Historical Stock Chart
From Aug 2024 to Sep 2024
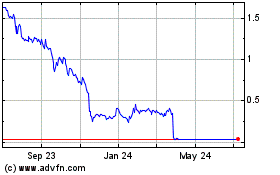
Gamida Cell (NASDAQ:GMDA)
Historical Stock Chart
From Sep 2023 to Sep 2024