Lightning 12 and Separator 12 Now Indicated for
Treatment of PE
Penumbra, Inc. (NYSE: PEN), a global healthcare company focused
on innovative therapies, today announced U.S. Food and Drug
Administration 510(k) clearance for expanded indication of the
latest iteration of the Indigo® Aspiration System, Lightning™ 12.
As part of the Indigo Aspiration System, Lightning 12 (Indigo
System CAT™ 12 Aspiration Catheter with Lightning Intelligent
Aspiration) and Separator 12 are now indicated for the removal of
fresh, soft emboli and thrombi from vessels of the peripheral
arterial and venous systems as well as for the treatment of
pulmonary embolism.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20201120005262/en/
The Indigo® Aspiration System Lightning™
12 received FDA clearance for the treatment of pulmonary embolism.
(Photo: Business Wire)
Pulmonary embolism, or PE, is a deadly condition causing blood
clots within a patient’s lungs that has grown in prevalence during
the COVID-19 pandemic. The Indigo Aspiration System with Lightning
12 removes these difficult-to-access, life-threatening clots,
potentially reducing the need for thrombolytics and their
associated ICU stays.
“Penumbra continues to strive to bring innovative therapies to
the medical community, and this is another important milestone that
will help advance pulmonary embolism intervention,” said Adam
Elsesser, president and chief executive officer, Penumbra. “COVID
has increased the awareness of the need for therapies that remove
blood clots, and we are thrilled with the FDA clearance of the PE
treatment indication for Lightning 12.”
The Indigo System Lightning 12 is the company’s newest
generation aspiration system for peripheral thrombectomy. Lightning
12 combines the Indigo System CAT™12 Aspiration Catheter with
Lightning Intelligent Aspiration, enabling physicians to focus on
optimizing thrombus removal using the system’s unique clot
detection mechanism. CAT12 is a large-lumen aspiration catheter
that incorporates novel laser-cut hypotube-based catheter
engineering to provide advanced deliverability and torqueability
within the body. This combination of intelligent aspiration and
large-lumen catheter engineering makes Lightning 12 Penumbra’s most
advanced clot removal technology for the treatment of pulmonary
embolism.
The Indigo System Lightning 12 was launched in July and is
available immediately for patients needing interventional treatment
for PE. For more information, visit
https://www.penumbrainc.com/indigo-lightning/.
About Pulmonary Embolism
Pulmonary embolism (PE) is a condition that occurs when blood
clots, typically traveling from the veins in the legs, get caught
in the arteries of the lungs. Blood clots in the lungs can block
the lungs from absorbing oxygen, causing strain on the heart and
other organs. According to the American Heart Association, PE
affects roughly 367,000 Americans per year.1
About Indigo System
The latest generation of Penumbra’s continuous aspiration
thrombectomy system features Indigo System Lightning 12 which
combines the new Indigo System CAT 12 Aspiration Catheter with
Lightning Intelligent Aspiration powered by Penumbra ENGINE,
enabling physicians to focus on optimizing thrombus removal using
the system’s unique clot detection mechanism. The Indigo System’s
proprietary Separator technology, Separator 12 is also available
with Lightning 12 and is designed to enable unobstructed aspiration
for the duration of the procedure. Lightning 12 and Separator 12
are designed for the removal of fresh, soft emboli and thrombi from
the peripheral arterial and venous systems, and for the treatment
of pulmonary embolism.
In addition to Lightning 12, the Indigo System also now includes
Lightning 8 which combines the Indigo System CAT 8 Aspiration
Catheter with Lightning Intelligent Aspiration for the removal of
fresh, soft emboli and thrombi from the peripheral arterial and
venous systems, and for the treatment of pulmonary embolism.
Lightning 12 and Lightning 8 expand the already broad offering of
CAT8, CATD, CAT6, CAT5, and CAT3 and are paired with Penumbra
ENGINE, the company’s proprietary continuous, mechanical vacuum
aspiration pump.
Important Safety Information
Additional information about Penumbra’s products can be located
on Penumbra’s website at
http://www.penumbrainc.com/healthcare-professionals. Prior to use,
please refer to Instructions for Use for complete product
indications, contraindications, warnings, precautions, potential
adverse events and detailed instructions for use.
About Penumbra
Penumbra, Inc., headquartered in Alameda, California, is a
global healthcare company focused on innovative therapies. Penumbra
designs, develops, manufactures and markets novel products and has
a broad portfolio that addresses challenging medical conditions in
markets with significant unmet need. Penumbra sells its products to
hospitals and healthcare providers primarily through its direct
sales organization in the U.S., most of Europe, Canada and
Australia, and through distributors in select international
markets. Penumbra, the Penumbra P logo, Indigo, CAT, Separator,
Lightning, and Penumbra ENGINE are trademarks of Penumbra, Inc. For
more information, visit www.penumbrainc.com and connect on Twitter
and LinkedIn.
Forward-Looking Statements
Except for historical information, certain statements in this
press release are forward-looking in nature and are subject to
risks, uncertainties and assumptions about us. Our business and
operations are subject to a variety of risks and uncertainties and,
consequently, actual results may differ materially from those
projected by any forward-looking statements. Factors that could
cause actual results to differ from those projected include, but
are not limited to: the impact of the COVID-19 pandemic on our
business, results of operations and financial condition; failure to
sustain or grow profitability or generate positive cash flows;
failure to effectively introduce and market new products; delays in
product introductions; significant competition; inability to
further penetrate our current customer base, expand our user base
and increase the frequency of use of our products by our customers;
inability to achieve or maintain satisfactory pricing and margins;
manufacturing difficulties; permanent write-downs or write-offs of
our inventory; product defects or failures; unfavorable outcomes in
clinical trials; inability to maintain our culture as we grow;
fluctuations in foreign currency exchange rates; and potential
adverse regulatory actions. These risks and uncertainties, as well
as others, are discussed in greater detail in our filings with the
Securities and Exchange Commission, including our Annual Report on
Form 10-K for the year ended December 31, 2019 filed with the SEC
on February 26, 2020, our Quarterly Report on Form 10-Q for the
quarter ended March 31, 2020 filed with the SEC on May 7, 2020, our
Quarterly Report on Form 10-Q for the quarter ended June 30, 2020
filed with the SEC on August 3, 2020, and our Quarterly Report on
Form 10-Q for the quarter ended September 30, 2020 filed with the
SEC on November 2, 2020. There may be additional risks of which we
are not presently aware or that we currently believe are immaterial
which could have an adverse impact on our business. Any
forward-looking statements are based on our current expectations,
estimates and assumptions regarding future events and are
applicable only as of the dates of such statements. We make no
commitment to revise or update any forward-looking statements in
order to reflect events or circumstances that may change.
1Virani, S.S. et al. on behalf of the American Heart Association
Council on Epidemiology and Prevention Statistics Committee and
Stroke Statistics Subcommittee. Heart disease and stroke
statistics—2020 update: a report from the American Heart
Association. Circulation. 2020;141(9):e139–e596.
Source: Penumbra, Inc.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20201120005262/en/
Investor Relations: Penumbra, Inc. investors@penumbrainc.com
510-995-2461 Media Relations: Betsy Merryman Merryman
Communications betsy@merrymancommunications.com 310-560-8176
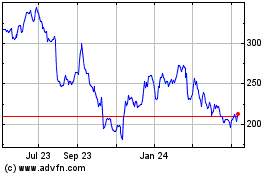
Penumbra (NYSE:PEN)
Historical Stock Chart
From Mar 2024 to Apr 2024
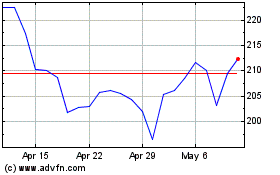
Penumbra (NYSE:PEN)
Historical Stock Chart
From Apr 2023 to Apr 2024