SAN DIEGO, June 12, 2019 /PRNewswire/ -- NuVasive, Inc.
(NASDAQ: NUVA), the leader in spine technology innovation, focused
on transforming spine surgery with minimally disruptive,
procedurally integrated solutions, today announced its LessRay®
radiation reduction and workflow enhancement platform was voted a
gold winner in the Radiological, Imaging and Electromechanical
Devices category at the 2019 Medical Design Excellence Awards
(MDEA) held in New York City on
June 11, 2019.
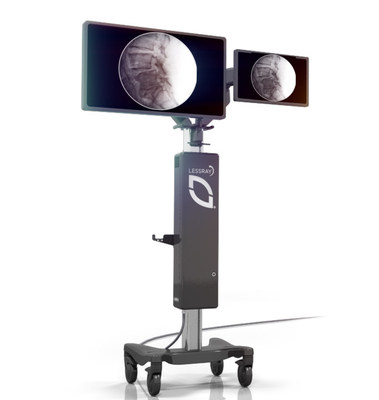
The LessRay platform is built on a proprietary algorithm
that captures and enhances low-dose, low-quality images
to produce images with similar diagnostic
capabilities as conventional full-dose images. This
technology has the ability to reduce radiation emissions to
the operating room (OR) staff and patient by 75 percent
compared to standard fluoroscopy.1 Additionally,
LessRay contains a number of surgical efficiency features designed
to save time during spine surgery, which can decrease time under
anesthesia for the patient. In the newest software upgrade, LessRay
showcases Integrated Global Alignment® (iGA®) measurements on a
stitched image which furthers workflow efficiencies for surgeons
and OR staff. Also included, compatibility with picture archiving
and communications system (PACS) enables seamless information
transfer between LessRay and a hospital's network.
Increased radiation exposure and limited imaging capabilities
are some of the many barriers faced in adopting minimally invasive
surgery (MIS) in spine. LessRay is designed to maintain the
diagnostic image quality and anatomical visualization necessary to
perform MIS procedures during spine surgery. This platform further
accelerates the adoption of MIS to enable potential patient
benefits including decreased blood loss, lower risk of infection
and faster recovery time.
"We are honored to be recognized by the MDEA for the LessRay
platform and our ongoing commitment to delivering better clinical
outcomes in spine surgery," said Matt
Link, president of NuVasive. "Studies show surgeons can
receive their lifetime occupational radiation limit within the
first decade of their career, and NuVasive is proud to play an
integral role in building a safer OR experience for patients,
surgeons and staff."
The Medical Design Excellence Awards is the premier awards
program for the medtech industry. Since its inception in 1998, the
mission of the MDEAs has been to recognize significant achievements
in medical product design and engineering that improve the quality
of healthcare delivery and accessibility. The 2019 MDEA Juror
Panel—composed of practicing doctors, nurses, technicians,
industrial designers, engineers, manufacturers and human factors
experts—selected 46 finalists in nine medical technology product
categories. Products and technologies were judged based on patient
benefits, market differentiation, benefits to overall healthcare,
design, and engineering innovation and functional innovation.
The MDEA award is the second honor the LessRay platform has
achieved in 2019; the platform was recently voted a gold winner in
the Advanced Surgical Instruments category at the 2019 Edison
Awards™.
About NuVasive
NuVasive, Inc. (NASDAQ:
NUVA) is the leader in spine technology innovation, focused on
transforming spine surgery and beyond with minimally disruptive,
procedurally integrated solutions designed to deliver reproducible
and clinically-proven surgical outcomes. The Company's portfolio
includes access instruments, implantable hardware, biologics,
software systems for surgical planning, navigation and imaging
solutions, magnetically adjustable implant systems for spine and
orthopedics, and intraoperative monitoring service offerings. With
more than $1 billion in revenues,
NuVasive has approximately 2,600 employees and operates in more
than 50 countries serving surgeons, hospitals and patients. For
more information, please visit www.nuvasive.com.
Forward-Looking Statements
NuVasive cautions
you that statements included in this news release that are not a
description of historical facts are forward-looking statements that
involve risks, uncertainties, assumptions and other factors which,
if they do not materialize or prove correct, could cause NuVasive's
results to differ materially from historical results or those
expressed or implied by such forward-looking statements. The
potential risks and uncertainties which contribute to the uncertain
nature of these statements include, among others, risks associated
with acceptance of the Company's surgical products and procedures
by spine surgeons, development and acceptance of new products or
product enhancements, clinical and statistical verification of the
benefits achieved via the use of NuVasive's products (including the
iGA platform), the Company's ability to effectually manage
inventory as it continues to release new products, its ability to
recruit and retain management and key personnel, and the other
risks and uncertainties described in NuVasive's news releases and
periodic filings with the Securities and Exchange Commission.
NuVasive's public filings with the Securities and Exchange
Commission are available at www.sec.gov. NuVasive assumes no
obligation to update any forward-looking statement to reflect
events or circumstances arising after the date on which it was
made.
1Wang TY, Farber SH, Perkins SS, et al. An
internally randomized control trial of radiation exposure using
ultra-low radiation imaging (ULRI) versus traditional C-arm
fluoroscopy for patients undergoing single-level minimally invasive
transforaminal lumbar interbody fusion.
Spine 2017;42(4);217-23.
 View original content to download
multimedia:http://www.prnewswire.com/news-releases/nuvasive-lessray-platform-wins-gold-at-2019-medical-design-excellence-awards-300866022.html
View original content to download
multimedia:http://www.prnewswire.com/news-releases/nuvasive-lessray-platform-wins-gold-at-2019-medical-design-excellence-awards-300866022.html
SOURCE NuVasive, Inc.