NeuroMetrix Announces Quell Fibromyalgia Pilot with Synovation Medical Group
September 22 2016 - 7:00AM
Business Wire
NeuroMetrix, Inc. (Nasdaq: NURO, NUROW) announced today
that Synovation Medical Group will conduct an evaluation of
Quell® Wearable Pain Relief Technology™ in fibromyalgia
patients.
"We have seen both clinical data and positive reviews that
suggest that Quell may be beneficial in reducing pain and improving
the quality of life in people who suffer from fibromyalgia,"
said Shai N. Gozani, M.D., Ph.D., President and CEO
of NeuroMetrix. "This clinical evaluation will provide
additional evidence for fibromyalgia patients and their healthcare
providers who are considering Quell as a pain relief option."
"Clinical research on innovative therapies is an integral part
of what we do," said Kunal Gogna, M.D., of Synovation Medical
Group. "We are excited to test the Quell technology and potentially
add it to our multidisciplinary approach to pain management.” Led
by Mauro Zappaterra, M.D., Ph.D., Director of Clinical Research,
and by Dr. Gogna, Synovation Medical Group supports the use of
medical devices as viable alternatives to medication for the
treatment of various chronic pain ailments.
About Synovation Medical Group
Synovation Medical Group offers top-quality care through an
extensive network of doctors, surgeons and specialists in areas
including pain management, physical medicine and rehabilitation,
physical therapy and spine care. In 1990, Synovation Medical Group
was founded by Dr. Clayton Varga who wanted to share his vision to
serve patients and community as leaders in musculoskeletal
medicine. Visit Synovation Medical Group for more information.
About Quell
Quell is designed for millions of people suffering from chronic
pain. The advanced wearable device is lightweight and can be worn
during the day while active, and at night while sleeping. It has
been cleared by the FDA for treatment of chronic pain
without a prescription. In a recent study, 81% of Quell users
reported an improvement in their chronic pain. Quell users can
start, stop, and adjust therapy discreetly via the optional Quell
Relief app. Quell also offers advanced sleep tracking that provides
feedback on eight dimensions of sleep including sleep duration,
quality, body position, time out of bed, and repetitive leg
movements throughout the night. Quell was the winner of the
2016 SXSW (South by Southwest) Innovation Award for Best
Wearable Technology. Quell is available at select healthcare
professionals and retailers. Visit QuellRelief.com for more
information.
About NeuroMetrix
NeuroMetrix is a commercial stage, innovation driven
healthcare company combining bioelectrical and digital medicine to
address chronic health conditions including chronic pain, sleep
disorders, and diabetes. The company's lead product is Quell, an
over-the-counter wearable therapeutic device for chronic pain.
Quell is integrated into a digital health platform that helps
patients optimize their therapy and decrease the impact of chronic
pain on their quality of life. The company also markets DPNCheck®,
a rapid point-of-care test for diabetic neuropathy, which is the
most common long-term complication of Type 2 diabetes. The company
maintains an active research effort and has several pipeline
programs, including a therapeutic device for restless leg syndrome.
The company is located in Waltham, Massachusetts and was
founded as a spinoff from the Harvard-MIT Division of Health
Sciences and Technology in 1996. For more information, please
visit www.NeuroMetrix.com.
View source
version on businesswire.com: http://www.businesswire.com/news/home/20160922005191/en/
NeuroMetrix, Inc.Thomas T. Higgins, 781-314-2761SVP and Chief
Financial Officerneurometrix.ir@neurometrix.com
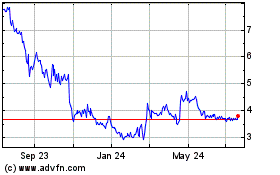
NeuroMetrix (NASDAQ:NURO)
Historical Stock Chart
From Mar 2024 to Apr 2024
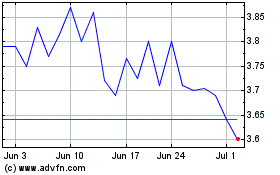
NeuroMetrix (NASDAQ:NURO)
Historical Stock Chart
From Apr 2023 to Apr 2024