NeuroMetrix Announces Sales Force Expansion for Quell Fibromyalgia
July 11 2023 - 9:00AM

NeuroMetrix, Inc. (Nasdaq: NURO) today announced expansion of its
sales force to drive further adoption of Quell® Fibromyalgia, which
is the first and only medical device authorized by the U.S. Food
and Drug Administration (FDA) to help reduce the symptoms of
fibromyalgia.
This expansion follows positive results from the strategic
launch of Quell Fibromyalgia in December 2022, and is designed to
intensify commercial efforts in the California, Texas and Florida
markets.
“We are encouraged by the response to Quell Fibromyalgia from
physicians and patients during our strategic launch phase,” said
Shai N. Gozani, M.D., Ph.D., Chief Executive Officer of
NeuroMetrix. “The early adoption by the physician community speaks
to the need for more effective tools to support their fibromyalgia
patients. We look forward to bringing Quell Fibromyalgia to
practices in California, Texas and Florida that seek to offer their
patients the latest innovation in fibromyalgia treatments.”
Quell Fibromyalgia Indications
Quell Fibromyalgia is a transcutaneous electrical nerve
stimulation (TENS) device indicated as an aid for reducing the
symptoms of fibromyalgia in adults with high pain sensitivity. The
device may be used during sleep.
Limitations The sale, distribution, and use of Quell
Fibromyalgia is restricted to prescription use in accordance with
21 CFR 801.109. The Product labeling should be reviewed for a
complete list of contraindications, precautions and warnings.
For more information visit QuellFibromyalgia.com.
About Quell
Quell is an advanced, non-invasive, neuromodulation platform
that is covered by 26 issued or granted U.S. utility patents. It is
the only wearable neuromodulator that is enabled by a proprietary
microchip to provide precise, high-power nerve stimulation in a
form factor the size of a credit card. Quell utilizes position and
motion sensing to automatically adjust stimulation for an optimal
user experience both day and night. The device supports Bluetooth®
low energy (BLE) to communicate with mobile apps for multiple
smartphone platforms. Quell is indicated to help reduce
fibromyalgia symptoms in patients with high pain sensitivity and to
reduce lower extremity chronic pain.
About NeuroMetrix
NeuroMetrix is a commercial stage healthcare company that
develops and commercializes neurotechnology devices to address
unmet needs in the chronic pain and diabetes markets. The Company's
products are wearable or hand-held medical devices enabled by
proprietary consumables and software solutions that include mobile
apps, enterprise software and cloud-based systems. The Company has
two commercial brands. Quell® is a wearable neuromodulation
platform. DPNCheck® is a point-of-care screening test for
peripheral neuropathy. For more information, visit
www.neurometrix.com.
Source: NeuroMetrix, Inc.
Thomas T. Higgins SVP and Chief Financial Officer
neurometrix.ir@neurometrix.com
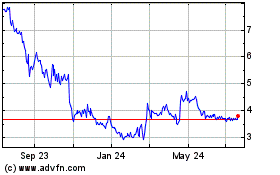
NeuroMetrix (NASDAQ:NURO)
Historical Stock Chart
From Apr 2024 to May 2024
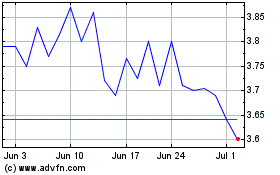
NeuroMetrix (NASDAQ:NURO)
Historical Stock Chart
From May 2023 to May 2024