Anika Announces Plans to Showcase HYALOFAST at the International Cartilage Regeneration & Joint Preservation Society (ICRS) S...
January 16 2019 - 4:05PM
Business Wire
Anika Therapeutics, Inc. (NASDAQ: ANIK), a global, integrated
orthopedic and regenerative medicines company specializing in
therapeutics based on its proprietary hyaluronic acid ("HA")
technology, today announced plans to showcase HYALOFAST, a
biodegradable, HA-based cartilage repair scaffold, at the 2019
International Cartilage Repair Society (ICRS) Summit. The 2019 ICRS
Summit, which will be focused on Bio-Orthopaedics in Sports
Medicine, will be held in San Diego, CA at the Hyatt Regency
Mission Bay on January 17-18, 2019.
At the 2019 ICRS Summit, Dr. Alberto Gobbi, world-renowned
specialist in arthroscopic surgery, cartilage repair and
regenerative medicine, will present findings from a 10-year
follow-up study of patients treated with HYALOFAST HA scaffold with
Bone Marrow Aspirate Concentrate (BMAC) in a one-step surgery. The
session will also provide a comparative review of HYALOFAST in
comparison to ACI (Autologous Chondrocyte Implantation) and MACI
(Matrix Autologous Chondrocyte Implantation), two-step surgeries
requiring cell cultivation.
“The ICRS Summit in San Diego is a great opportunity to showcase
the significant advantages of HYALOFAST before an assembly of
world-renowned specialists in the field of cartilage repair and
regeneration,” said Joseph Darling, President and CEO, Anika
Therapeutics. “We believe that HYALOFAST is a truly remarkable
advancement in the treatment of cartilage injuries and defects due
to its flexible, minimally-invasive means of application and
adaptive fit to individual patient needs.”
HYALOFAST is a non-woven, single-step, off-the-shelf,
biodegradable HA-based scaffold for hyaline-like cartilage
regeneration to treat cartilage injuries and defects. HYALOFAST is
commercially available in approximately 15 countries and has been
used to successfully treat more than 15,000 patients
internationally, with strong ten-year patient follow-up data.
HYALOFAST is pending regulatory submission in the United States
following its FastTRACK Phase III trial which is currently
enrolling patients across the U.S. and Europe.
Technical Session 3: Cartilage
Title: “One or Two Steps: Long Term Results”Presenter: Dr.
Alberto Gobbi, OASI Bioresearch Foundation, Milano –
ItalyDate/Time: January 17, 2019 at 10:45 – 11:40am
Company-Sponsored Booth
Anika will be a Gold Sponsor and will host a booth (#6), where
it will showcase HYALOFAST.
About ICRS
The ICRS (International Cartilage Regeneration and Joint
Preservation Society) is the main forum for international
collaboration in cartilaginous tissue research that brings together
basic scientists, clinical researchers, physicians and members of
industry, engaged or interested in the field of articular biology,
its genetic basis and regenerative medicine. It provides continuing
education and training to physicians and scientists with an active
interest in the prevention and treatment of joint disease to
improve patient care through regenerative medicine approaches.
2018 HYALOFAST Publications
- Gobbi A., et al. Use of bone marrow
aspirate concentrate combined with hyaluronan-based scaffold for
early osteoarthritis in athletes. Football Medicine Outcomes
Abstract book XXVII Isokinetic Medical Group International
Conference – Camp Nou, Barcelona 2-4 June 2018. Ed Calzetti &
Mariucci. Page 442.
- Sofu H. et al. Clinical and
radiographic outcomes of chitosan-glycerol phosphate/blood implant
are similar with hyaluronic acid-based cell-free scaffold in the
treatment of focal osteochondral lesions of the knee joint. Knee
Surg Sports Traumatol Arthrosc. 2018 Aug 1. (Epub ahead of
print).
- Gollwitzer H. Operative Therapie -
Knorpelschäden der Hϋfte Einzeitiges arthroskopisches Verfahren zur
Regeneration. Sportärztezeitung 03/2018 (Cartilage Damage to the
Hip. One-Stage Arthroscopic Method of Regeneration).
About Anika Therapeutics, Inc.
Anika Therapeutics, Inc. (NASDAQ: ANIK) is a global,
integrated orthopedic and regenerative medicines company based
in Bedford, Massachusetts. Anika is committed to improving the
lives of patients with degenerative orthopedic diseases and
traumatic conditions with clinically meaningful therapies along the
continuum of care, from palliative pain management to regenerative
tissue repair. The Company has over two decades of global expertise
developing, manufacturing, and commercializing more than 20
products based on its proprietary hyaluronic acid (HA)
technology. Anika's orthopedic medicine portfolio
includes ORTHOVISC®, MONOVISC®,
and CINGAL®, which alleviate pain and restore
joint function by replenishing depleted HA,
and HYALOFAST, a solid HA-based scaffold to aid
cartilage repair and regeneration. For more information about
Anika, please visit www.anikatherapeutics.com.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20190116005053/en/
For Investor Inquiries:Anika Therapeutics, Inc.Sylvia Cheung,
781-457-9000Chief Financial OfficerFor Media Inquiries:W2O
GroupSonal Vasudev, 917-523-1418sonal@w2ogroup.com
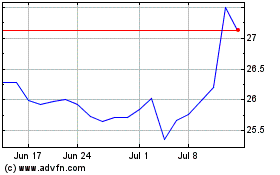
Anika Therapeutics (NASDAQ:ANIK)
Historical Stock Chart
From Mar 2024 to Apr 2024
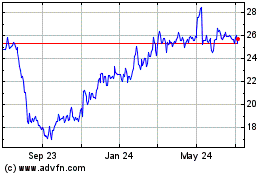
Anika Therapeutics (NASDAQ:ANIK)
Historical Stock Chart
From Apr 2023 to Apr 2024