Registration No. 333-216155
PROSPECTUS
SUMMARY
This summary does not contain all of the information
you should consider before investing in our securities. You should read this summary together with the more detailed information
appearing in this prospectus, including “Risk Factors,” “Selected Historical Financial Data,” “Management’s
Discussion and Analysis of Financial Condition and Results of Operations,” “Business” and our financial statements
and the related notes included at the end of this prospectus, before making an investment in our ordinary shares. All references
to “Medigus,” “we,” “us,” “our,” the “Company” and similar designations
refer to Medigus Ltd. The terms “shekels,” “Israeli shekels” and “NIS” refer to New Israeli
Shekels, the lawful currency of the State of Israel, the terms “dollar,” “US$” or “$” refer
to U.S. dollars, the lawful currency of the United States. Unless derived from our financial statements or otherwise indicated,
U.S. dollar translations of NIS amounts presented in this prospectus are translated using the rate of NIS 3.658 to $1.00, based
on the exchange rates reported by the Bank of Israel on March 15, 2017.
Our
Company
Overview
We
are a medical device company dedicated to the development, manufacturing and marketing of surgical endostaplers and direct vision
systems for minimally invasive medical procedures. Our expertise is in the development, production and marketing of innovative
surgical devices with direct visualization capabilities for the treatment of Gastroesophageal Reflux Disease, or GERD, a common
ailment, which is predominantly treated by medical therapy (e.g. proton pump inhibitors) or in chronic cases, conventional open
or laparoscopic surgery. Our FDA-cleared and CE-marked endosurgical system, known as the MUSE
TM
(Medigus Ultrasonic Surgical
Endostapler) system, enables minimally-invasive and incisionless procedures for the treatment of GERD by reconstruction of the
esophageal valve via the mouth and esophagus, eliminating the need for surgery in eligible patients. We believe that this procedure
offers a safe, effective and economical alternative to the current modes of GERD treatment for certain GERD patients, and has
the ability to provide results which are equivalent to those of standard surgical procedures while reducing pain and trauma, minimizing
hospital stays, and delivering economic value to hospitals and payors.
The
key elements of the MUSE
TM
system include a single-use endostapler containing several sophisticated innovative technologies
such as flexible stapling technology, a miniature camera and ultrasound sensor, as well as a control console offering a video
image transmitted from the tip of the endostapler.
In
addition to the MUSE
TM
system for the treatment of GERD, we have developed miniaturized video cameras for use in various
medical procedures as well as specialized industrial applications.
Prevalence
of GERD
GERD, is a worldwide disorder, with evidence suggesting
an increase in GERD disease prevalence since 1995. The sample size weighted mean for the GERD population in the United States
and Europe is 19.8% and 15.2% respectively. In the United States alone, over 49 million adults are affected by the disease, with
over 29 million adults suffering daily GERD symptoms. Proton pump inhibitors, or PPIs, are a class of effective and generally
safe medication to treat GERD, but not everyone who experiences heartburn needs a PPI. Several PPIs have been widely advertised
to consumers and heavily promoted by physicians. This has led to an overuse of the drug. PPIs are the third highest selling class
of drugs in the U.S. and Nexium has the second highest retail sales among all drugs at $4.8 billion in 2008. This figure does
not include sales of other brands of PPIs.
After
being swallowed, food descends through the esophagus to the stomach, which contains acids and enzymes intended to digest and break
down food. GERD is caused by the defective operation of the lower esophageal sphincter, or LES, a valve, which controls the flow
of ingested food from the esophagus into the stomach. While eating and between eating periods, a properly operating LES prevents
stomach contents from entering the esophagus. Among GERD sufferers, the valve opens spontaneously or is unable to close properly.
This results in acidic stomach contents rising into the esophagus, causing irritation, acid reflux and heartburn, as well as other
potentially dangerous conditions.
Beyond painful symptoms, GERD may also increase sufferers’
susceptibility to cancer. Whereas the stomach is lined by the “gastric mucosal barrier” which allows acidic material
to be contained harmlessly, the surface of the esophagus consists of flat, thin cells called squamous cells, which are
not resistant to acid. Repeated episodes of acid reflux can cause inflammation of the esophagus, a condition called esophagitis.
The flat cells lining the esophagus can also undergo genetic changes due to exposure to acid, causing these cells to resemble those
found in the stomach lining, a condition known as Barrett’s Esophagus.
Barrett
Esophagus is a complication of GERD and predisposes patients to esophageal adenocarcinoma, a tumor that has increased in incidence
more than 7-fold over the past several decades
.
Studies have shown that
people exhibiting Barrett’s Esophagus have a higher risk of developing cancer of the esophagus. Studies have also shown,
that compared to patients not exhibiting GERD symptoms, patients exhibiting weekly symptoms of GERD have a five times higher probability
for developing esophageal cancer while patients exhibiting daily symptoms of GERD have a seven times higher probability for developing
esophageal cancer. The most common risk factors for cancer in Barrett esophagus patients include chronic GERD, hiatal hernia,
advanced age, male sex, white race, cigarette smoking and obesity.
Treatment of GERD
Treatment of GERD involves a stepwise
approach. The goals are to control symptoms, to heal esophagitis and to prevent recurrent esophagitis. The treatment is based on
lifestyle modification and control of gastric acid through medical treatment (antacids, PPI’s, H2 blockers or other reflux
inhibitors) or antireflux surgery. Mild GERD may be defined as intermittent reflux symptoms that can be managed with lifestyle
changes or over-the-counter medications. Moderate to severe GERD represents more chronic symptoms that may require stronger drugs,
long term medication or surgical intervention.
For moderate to severe GERD, physicians
usually prescribe proton pump inhibiting drugs, or PPI. This class of drugs reduces acid production by the stomach, and thereby
relieves the patients of their symptoms. Drugs of this class are among the most commonly prescribed medications in the world. There
are several brands on the market, best known are Prilosec (omeprazole), Prevacid (lansoprazole) and Nexium (esomeprazole). Certain
PPI drugs are available over the counter in the United States and in other countries, but the over the counter dosage may be inadequate
to control GERD symptoms, except in mild cases.
While PPI drugs effectively reduce the
severity and frequency of GERD symptoms, they have a number of drawbacks:
a) In approximately 30% of patients, symptom control
is incomplete;
b) The drugs do not treat the disease,
they only control its manifestations, therefore must be taken for life at a dosage which requires prescription. Accumulated costs
may be substantial; and
c) Long term use is associated with a number
of serious adverse effects. In particular, they increase the risk of osteoporosis and fractures of the hip, wrist and spine. The
FDA had issued a warning on this effect as well as warnings against other untoward effects on absorption of other essential minerals,
which may lead to chronic kidney disease, irregular heartbeat, diarrhea and increase flatulence.
Interventional treatment
The most common operation for GERD is
called a surgical fundoplication, a procedure that prevents reflux by wrapping or attaching the upper part of the stomach around
the lower esophagus and securing it with sutures. Due to the presence of the wrap or attachment, increasing pressure in the stomach
compresses the portion of the esophagus which is wrapped or attached by the stomach, and prevents acidic gastric content from flowing
up into the esophagus. Today, the operation is usually performed laparoscopically: instead of a single large incision into the
chest or abdomen, four or five smaller incisions are made in the abdomen, and the operator uses a number of specially designed
tools to operate under video control.
The operation does not completely eliminate the use
of PPI, and up to approximately 60% still use some in long term follow up. Nevertheless, the dose is usually lower – in
the over the counter range - and the response rate is excellent. Since the majority of patients referred to surgeons are incomplete
responders, or require a high dose of PPI, the patients are generally satisfied with the operation, and the overall costs of treatment
are lower in the long run.
In spite of the clinical outcome of
surgery, relatively few patients undergo surgery. We estimate that large numbers of patients who are candidates for operative treatment
are either not referred by their treating physician or decline it. We believe that many patients decline to undergo operations
to avoid even minute scars or violation of the abdominal cavity.
Given the current environment
in which the vast majority of GERD sufferers in North America and Europe must choose between long-term pharmaceutical therapy and
surgery, leading to what is known in our industry as the “treatment gap”, we believe there is a demand for a minimally-invasive,
incision-less procedure which treats the root cause of the disease. We believe that the MUSE
TM
system is positioned
to fill this need.
Our system achieves the general physiological result
of surgical fundoplication, by inserting the MUSE
TM
endostapler through the mouth and the esophagus, and stapling
the top of the stomach to the side of the esophagus. The endostapler contains a video camera and stapling system. Staples have
long been used in surgical procedures in place of sutures, and we believe that they are at least as reliable and potentially more
durable. Our endostapler uses standard surgical staples.
First line therapy for GERD includes a combination
of lifestyle modifications and medical therapy, or PPIs. Unfortunately, 25% to 42% of patients with GERD do not respond to an
initial 4-8-week treatment of PPI. In those who do respond to therapy, the effectiveness of PPI treatment decreases over time.
Antireflux surgery controls acid reflux and treats an incompetent lower esophageal sphincter, while also improving patient quality
of life in the long term. Thus, PPI therapy and lifestyle modifications are frequently eliminated.
Despite the effectiveness of surgery, it is invasive,
requires hospitalization, and carries the risk of short and long-term complications, including dysphagia, diarrhea, and gas bloat
syndrome Thus, endoscopic therapies that mimic the mechanism through which surgery works and can reduce surgical morbidity have
gained popularity for the treatment of GERD.
The market for medical devices, including the market
for endoscopic therapies, is very broad, with an increasing demand for new less invasive alternatives to the existing surgical
procedures for the treatment of various diseases. This increasing need for minimally invasive and incision-less treatments, such
as endoscopy-based procedures, are also augmented by the increase in the average age of global population. In 2000, the worldwide
population of persons aged more than 65 years was an estimated 420 million. During 2000-2030, the worldwide population aged more
than 65 years is projected to increase by approximately 550 million to 973 million. This increase in age will potentially lead
to increased health-care costs and may have dramatic consequences for public health and the health care financing and delivery
systems significant patient benefits and cost savings.
Endoscopy is a minimally invasive method
of performing investigative, diagnostic and therapeutic medical procedures, employing an endoscope, which allows real-time visual
observation of the patient’s internal organs during the procedure. Endoscopic procedures are most commonly performed through
natural orifices, including via the mouth, to avoid incisions. Because of the accessibility of the digestive tract through the
mouth, the endoscopy field is largely focused on disorders of the gastrointestinal tract such as disorders of the colon, esophagus,
stomach and duodenum.
Endoscopes are commonly composed
of a flexible tube with a camera installed at its tip. Endoscopes often include “working channels” through which catheters
or other endoscopic tools or devices may be inserted directly into the patient’s digestive system. The primary advantage
of endoscopy is the elimination of incisions to the patient’s body during a medical procedure. We believe that this is safer,
prevents most post-operative pain and facilitates faster recuperation. Patient perception or preference is important as well. The
perception of endoscopy procedures as being safer, and less painful than, corresponding surgical procedures may have the effect
of minimizing patient fears.
Endoscopic procedures generally involve less
recovery time and patient discomfort than conventional open or laparoscopic surgery. These procedures are also typically performed
in the outpatient hospital setting as opposed to an inpatient setting. Typically, outpatient procedures cost the hospital or the
insurer less money since there is no overnight stay in the hospital.
Our Solution and
Products
The MUSE
TM
system
Our primary product, the MUSE
TM
system for transoral fundoplication, is a single use innovative device for the incisionless treatment of GERD, which is based on
our proprietary platform technology and know-how. While at present substantially all of our revenue is derived from the miniature
video camera and related equipment, our strategy is focused on the development and promotion of its MUSE
TM
System, which
we therefore refer to as our ‘primary product’.
Transoral means the procedure is performed
through the mouth, rather than through incisions in the abdomen. The MUSE
TM
system for transoral fundoplication was
previously known as the SRS
TM
Endoscopic Stapling System. We rebranded to the MUSE
TM
system following the launch
of the most recent generation product. The MUSE
TM
system is used to perform a procedure as an alternative to a surgical
fundoplication. The MUSE
TM
offers an endoscopic, incisionless alternative to surgery. A single surgeon or gastroenterologist
can perform the MUSE
TM
procedure, unlike in a laparoscopic fundoplication which requires incisions.
The system consists of the MUSE
TM
controller
console, the MUSE
TM
endostapler and several accessories, including an overtube, irrigation bottle, tubing supplies
and staple cartridges. The endostapler incorporates a video camera, a flexible surgical stapler and an ultrasonic sight, which
is used to measure the distance between the anvil and the cartridge of the stapler, to ensure their proper alignment and tissue
thickness. The device also contains an alignment pin, which is used for initial positioning of the anvil against the cartridge,
two anvil screws, which are used to reduce the thickness of the tissue that needs to be stapled and to fix the position of the
anvil and the MUSE
TM
endostapler during stapling. The system allows the operator to staple the fundus of the stomach
to the esophagus, in two or more locations, typically around the circumference, thereby creating a fundoplication, without any
incisions.
The clearance by the FDA, or ‘Indications
for Use’, of the MUSE
TM
System is "for endoscopic placement of surgical staples in the soft tissue of
the esophagus and stomach in order to create anterior partial fundoplication for treatment of symptomatic
chronic Gastro-Esophageal Reflux Disease in patients who require and respond to pharmacological
therapy". As such, the FDA clearance covers the use by an operator of
the MUSE
TM
endoscopic stapler as described in the above paragraph. In
addition,
in the pivotal study that was presented to the
FDA in order to gain clearance, only patients who were currently taking
GERD medications (i.e. pharmacological therapy) were allowed in the study. In addition, all patients had to
have a significant decrease in their symptoms when they were taking medication
compared to when they were off the medication. As such, the
FDA clearance included the indication that MUSE
TM
is intended for patients who require
and respond to pharmacological therapy. The MUSE
TM
System indication does not restrict its use with
respect to GERD severity from a regulatory point of view. However, clinicians typically only consider interventional
treatment options for moderate to severe GERD. Therefore, it is reasonable to expect the MUSE
TM
System would
be primarily used to treat moderate and severe GERD in practice. The system has received 510(k) marketing clearance from
the FDA in the United States, as well as a CE mark in Europe and a license from Health Canada. It is also cleared for use in
Turkey and in Israel.
Clinical studies
The original FDA submission for the
MUSE
TM
System included short-term (6 month) results from a multi-center clinical trial. The trial was conducted in support
of the 510(k) marketing clearance submission for the system and pursuant to an FDA-issued Investigational Device Exemption (IDE).
Enrollment was completed in November 2010. A total
of 72 patients were enrolled and 69 were treated with the MUSE
TM
system during the study. A manuscript detailing the
results of this study was published in Surgical Endoscopy and is currently available online. Publication in the hardcopy of the
journal was in the January 2015 issue.
The primary objective of the study was
to assess the safety and efficacy of the system in the treatment of subjects with GERD. The primary efficacy endpoint
was at least a 50% improvement in the GERD-HRQL (Health Related Quality of Life) scores in 53% of the subjects. HRQL is the standard
assessment of how an individual’s well-being may be affected over time by a disease. Secondary efficacy assessments
included PPI intake, esophageal acid exposure during a 24-hour period and anatomical changes. The follow-up period was set at six
months following each procedure.
The primary endpoint was met in that
73% of subjects exhibited at least a 50% reduction in HRQL at six months. In addition, 85% of subjects reduced their PPI intake
by at least 50%, with 65% of subjects eliminating PPI use completely at six months.
FDA marketing clearance for our system
was granted in May 2012 following the original FDA submission. Subsequent improvements to the system included improvements to the
camera, illumination and alignment mechanisms, the addition of an electronic stapling motor, and condensing two control consoles
into a single unit. FDA clearance for the modified system was obtained in March 2014. The modified system has also obtained a CE
mark in Europe and a license from Health Canada and was approved in Turkey and Israel.
In May 2013, we received five years of follow-up results
for a precursor IRB (Institutional Review Board) approved pilot study of the system conducted in 2007 at Deenanath Mangeshkar Hospital
and Research Center in the city of Pune, India. The results of this follow-up study were published in the peer review journal Surgical
Endoscopy in March 2015. As noted in the journal article, the five-year results are similar to the results obtained from subjects
who received-laparoscopic procedures for GERD in the same period. Each year, eleven of the thirteen patients were reached (although
not always the same eleven). All thirteen patients had at least a four year follow-up. Throughout the follow up period, GERD-HRQL
scores were normal in all but one patient. All patients indicated that they would agree to do the procedure again. Out of the initial
thirteen patients, seven (54%) had eliminated PPI and another three (23%) reduced PPI use by 50% or more. It should be emphasized
that for this trial patients were selected with GERD severity at a higher than average level (moderate to severe), a fact which
may indicate an even greater outcome of the effect of the system in an average GERD level patient population.
In November 2015 a follow-up study conducted
in the United States looked at evaluating the long-term clinical outcome of 37 patients who received GERD treatment with the MUSE
TM
device in the multi-center study mentioned above was concluded. Efficacy and safety data were analyzed up to four years post-procedure.
No new complications have been reported in such long-term analysis. The proportions of patients who remained off daily PPI were
83.8% (31/37) at six months, and 69.4% (25/36) at 4 years post-procedure. GERD-HRQL scores off PPI were significantly decreased
following six months and four years post-procedure. The authors concluded that the MUSE
TM
stapling device appeared to
be safe and effective in improving symptom scores as well as reducing PPI use in patients with GERD. These results appeared to
be equal to or better than those of the other devices for endoluminal GERD therapy.
In February 2017, we received an approval to start
a multi-center MUSE
TM
clinical study in China after the China Food and Drug Administration reviewed the ethics committees'
approval and agreements were in place with each study location. Under Principal Investigator, Yunsheng Yang, Director of Gastroenterology
Department Clinical center at 301 Hospital and Chairman of Chinese Society of Gastroenterology, The General Hospital of People's
Liberation Army in Beijing, the clinical study will include approximately 62 patients, will take place at 5 centers across China:
The General Hospital of People’s Liberation Army, Renji Hospital of Shanghai, Shanghai General Hospital, Peking University
Third Hospital and Navy General Hospital.
Procedures started in March 2017 and are expected
to carry on through 2017, with results being reported to the CFDA in 2018 as part of the CFDA submission for clearance to sell
MUSE
TM
in China.
Miniature Video Cameras
By definition all endoscopes must include
vision apparatus to facilitate the operator’s view of the internal organs of the patient. In the past, fiber optics were
utilized for this purpose, and have been gradually replaced with electronic video systems offering higher resolution and higher-quality
images. We have developed several models of miniaturized digital video cameras and video processing equipment, for use in medical
endoscopy products as well as industrial uses. Our cameras range between 3.45mm to 0.99 mm in diameter, and are based on single-use
Complementary Metal Oxide Semiconductor, or CMOS, image sensors. In some cases, our cameras are relatively inexpensive, allowing
them to be used in single-use devices.
Our miniature cameras are intended for
use in medical applications in which it has not yet been feasible to use miniature video cameras, and may be integrated into devices
developed by the company, or by third parties who source the camera from us. We expect that the growing demand for single-use medical
devices will increase demand for the CMOS cameras in particular, in fields such as gastroenterology, orthopedics, gynecology, ears
nose throat, urology, cardio-vascular, and other fields in which diagnostic and surgical procedures may be performed endoscopically.
Small-diameter video cameras permit not only smaller camera-based endoscopes which are able to penetrate previously inaccessible
organs or visualize them in improved image quality, but also allows for the addition of working channels and other features in
the valuable space freed by the reduction in camera size.
Our most advanced camera is a prototype
CMOS-based camera measuring only 0.99 mm in diameter transmitting 45,000 pixels in HDMI format, which we believe to be the smallest
video camera ever produced. This camera is based on “through-silicon-via” technology whereby the electronics pass vertically
through the sensor, permitting smaller diameter devices. This prototype camera will not be commercially available in the foreseeable
future.
Other products
We have utilized the MUSE
TM
system
technological platform for the development of prototypes for other endoscopy and direct vision products, including a device aiding
colonoscopy, a device used in dental surgery and others. To date, we have not yet applied for regulatory approvals for these devices,
nor have we entered into agreements for the commercialization of these devices.
Our strategy
Our primary goal is to generate recurring
revenues by driving sales of our MUSE
TM
system and establishing it as the standard-of-care procedure and device
for the treatment of moderate to severe GERD. We believe that we can achieve this goal by continuing to accumulate clinical data
and promote reimbursement for the procedure in the principal markets of North America, Europe and Asia. Our strategy includes the
following key elements:
Driving MUSE
TM
sales
We intend to continue to focus on commercializing
the MUSE
TM
system in key geographies and markets. We have begun expanding our distribution network for further commercialization
in key European markets including Italy and Germany. During 2017, we anticipate additional European distribution agreements to
be implemented. In addition, we have successfully completed the technical testing for CFDA approval in China and have already
begun the necessary clinical trial in March 2017. In the US, we will continue marketing the MUSE
TM
through a direct effort
at key U.S. institutions.
Collaborating and co-developing with established
companies
We seek to initiate co-development or
licensing collaborations with leading companies which have existing marketing channels or significant marketing power in critical
geographies and sales channels.
Out-licensing products
We may consider plans to issue a license
for various endoscopic systems which are based on owned and patent-protected technology which has been developed by us. We continue
to work to engage in agreements with companies which produce and market medical devices, to include the production of systems for
the foregoing companies which will be integrated by them in the endoscopic systems which they produce or that we will develop or
produce for them.
Developing additional products
Additionally, we intend to develop other
products which will be based on the integrated and platform technology which we have developed to date, including our miniaturized
visualization imaging products, combined with our flexible stapling platform, similar to the MUSE
TM
system. Additional products
could include a fully integrated, endoscopic platform designed for endoscopic surgical tissue dissection or for endoscopic sleeve
gastrectomy.
Recent Developments
China
Multi-Center Clinical Study
We have previously entered into an exclusive
distribution agreement with Sinopharm (China National Pharmaceutical Group Corporation) which granted the Chinese pharmaceutical
company exclusive rights to distribute the MUSE
TM
system in China following its approval by the CFDA. Under the agreement,
Sinopharm has also committed to purchasing a minimum of $17.6 million worth of MUSE
TM
and related products over the course
of four years, from the date of CFDA approval. The agreement also provides Sinopharm with the discretionary right to appoint Shanghai
Golden Grand-Medical Instruments Ltd., a Chinese company specializing in the distribution of medical devices, as its exclusive
sub-distributor in China. Under the agreement, Golden will manage sales and customer service, marketing and training, and recording
of clinical trials.
Recently, we
have received the approval of the China Food and Drug Administration, or the CFDA, for the commencement of the first multi-center
MUSE
TM
system clinical study in China. As part of our efforts to obtain CFDA approval for the MUSE
TM
system, the company
entered into an addendum to its current agreement with Golden, under which we paid Golden an aggregate sum of US$175,000 to be
used in financing of the efforts of the study.
On March 4, 2017, we completed our first human MUSE
TM
procedure in China.
Italian
Distribution Agreement
On November 22, 2016, we entered into
an exclusive distribution agreement with Innovamedica S.p.A., or Innovamedica. Under the terms of the agreement, Innovamedica,
a privately owned distributor of minimally invasive medical devices, will act as our exclusive distributor in Italy for a period
of four years commencing as of January 1, 2017. The agreement requires Innovamedica to purchase minimum annual quantities of our
products, which are expected to amount to at least EUR 1 million during the term of the Agreement. Innovamedica’s exclusivity
is conditioned upon ordering the minimum annual quantities.
Capital
Raise
On November 30, 2016, we entered into a securities
purchase agreements as part of an offering of ADSs pursuant to our shelf registration statement in the United States. As part
of the offering we issued a total of 113,917 ADSs representing a total of 5,695,850 ordinary shares, at a purchase price of US$6.70
per ADS, and warrants to purchase up to a total of 39,871 ADSs representing 1,993,550 ordinary shares, at an initial exercise
price of US$9.00 per ADS, in a concurrent private placement. The offering resulted in gross proceeds of $763,244. For their services
in the offering, we issued to Rodman & Renshaw, a unit of H.C. Wainwright & Co., who acted as placement agent in the offering,
warrants to purchase up to
an aggregate of 3,987
ADSs representing 199,355 ordinary
shares. Pursuant to the engagement letter executed with the placement agent, Rodman & Renshaw, relating to the offering, we
agreed to provide the placement agent with the right of first refusal, expiring on the twelve month anniversary following the
closing of the offering, if we or our subsidiaries decide to raise funds by means of a public offering or a private placement
of equity or debt securities using an underwriter or placement agent in the U.S. In addition, we issued to Roth Capital Partners
and Maxim Group LLC warrants to purchase up to
an aggregate of 1,993
ADSs representing
99,675 ordinary shares.
On September 8, 2016, we entered into a securities
purchase agreements as part of an offering of ADSs pursuant to our shelf registration statement in the United States. As part
of the offering, we sold a total of 128,000 ADSs representing a total of 6,400,000 ordinary shares, at a purchase price of US$11.50
per ADS. The offering resulted in gross proceeds of approximately $1.47 million. For their services in the offering, we issued
to Roth Capital Partners and Maxim Group LLC, who acted as co-placement agents in the offering, warrants to purchase up to
an
aggregate of 3,955
ADSs representing 197,750 ordinary shares.
Increase
of Authorized Share Capital
On
March 20, 2017, our shareholders approved an increase of our authorized share capital to 800,000,000 ordinary shares, par value
NIS 0.10 per share.
ADS
Ratio Change
On
January 12, 2017, we received a letter from NASDAQ indicating that we are not in compliance with the minimum bid price requirement
for continued listing set forth in Listing Rule 5550(a)(2) which requires listed securities to maintain a minimum bid price of
$1.00 per share. On March 15, 2017, we effected a change in the
ratio of ordinary shares
per ADS
from five ordinary shares per ADS to 50 ordinary shares per ADS. The change in the ordinary shares ratio for the
ADSs had the same effect as a 1-for-10 reverse stock split of the ADSs.
Preliminary
Results of Operations for the Three Months Ended December 31, 2016 and the Year Ended December 31, 2016
Our
consolidated financial statements for the three months ended December 31, 2016 and for the year ended December 31, 2016 are not
yet available. Accordingly, the information presented below reflects our preliminary financial data subject to the completion
of our financial closing procedures and any adjustments that may result from the completion of the annual audit of our consolidated
financial statements. As a result, this preliminary financial data may differ from the actual results that will be reflected in
our consolidated financial statements for the year when they are completed and publicly disclosed. This preliminary financial
data may change and those changes may be material. Accordingly, you should not place undue reliance upon these preliminary estimates.
Please see “Special Note Regarding Forward-Looking Statements.”
Our
expectations with respect to our unaudited financial data for the period discussed below are based upon management estimates and
are the responsibility of management. Our independent registered public accounting firm, Kesselman& Kesselman, has not audited,
reviewed, compiled or performed any procedures with respect to this preliminary financial data. Accordingly, Kesselman& Kesselman
does not express an opinion or any other form of assurance with respect thereto. We believe that the following information about
our revenues, operating expenses and cash and cash equivalents is helpful to an investor’s understanding of our operating
performance.
Revenues
|
|
|
Year
Ended December 31,
|
|
|
Three
Months Ended December 31,
|
|
|
|
|
2015
|
|
|
2016
|
|
|
2015
|
|
|
2016
|
|
|
|
|
Actual
|
|
|
Estimated
|
|
|
Actual
|
|
|
Estimated
|
|
|
|
|
(in
thousands)
|
|
|
Revenues
|
|
$
|
624
|
|
|
$
|
549
|
|
|
$
|
225
|
|
|
$
|
53
|
|
Our
revenue to date has been primarily based on the sale of imaging equipment, which we develop and manufacture. In
the year ended December 31, 2016, we estimate that we generated approximately $ 0.55 million of total revenues, compared to
$0.62 million in the year ended December 31, 2015, a decrease of approximately $75,000, or 12%. This decrease was primarily
due to the changes in the quantity of products sold.
We
estimate our revenues for the three months ended December 31, 2016, are $53,000 compared to approximately $225,000 in the three
months ended December 31, 2015, a decrease of approximately $172,000, or 76%. The changes are primarily due to revenues we recorded
during the three months ended December 31, 2015, for development services provided to a customer in the amount of approximately
$144,000 (see ‘Customer A’ in note 18e to our financial statements for the year ended December 31, 2015). We did not
receive any revenue from this customer during the three months ended December 31, 2016.
Operating
Expenses
|
|
|
Year
Ended December 31,
|
|
|
Three
Months Ended December 31,
|
|
|
|
|
2015
|
|
|
2016
|
|
|
2015
|
|
|
2016
|
|
|
|
|
Actual
|
|
|
Estimated
|
|
|
Actual
|
|
|
Estimated
|
|
|
|
|
(in
thousands)
|
|
|
Operating
expenses, net
|
|
$
|
9,903
|
|
|
$
|
9,500
|
|
|
$
|
3,009
|
|
|
$
|
1,618
|
|
We
estimate that our operating expenses are approximately $9.5 million for the year ended December 31, 2016, compared to $9.9 million
for the year ended December 31, 2015. The decrease of approximately $0.4 million, or 4% was primarily due to the decrease in research
and development activities and a decrease in selling and marketing expenses, which is reflected primarily by a cost reduction
program implemented by the Company since the third quarter of 2016, offset by an increase on general and administrative expenses
resulted primarily from an increase in professional expenses in connection with an intellectual property litigation.
Estimated
operating expenses for the three months ended December 31, 2016 are approximately $1.6 million, compared to $3.0 million for the
three month ended December 31, 2015. The decrease of approximately $1.4 million, or 86%, was primarily due to a cost reduction
program implemented by the Company since the third quarter of 2016.
Cash and Cash Equivalents
|
|
|
Year
Ended December 31,
|
|
|
|
|
2015
|
|
|
2016
|
|
|
|
|
Actual
|
|
|
Estimated
|
|
|
|
|
(in thousands)
|
|
|
Cash
and cash equivalents
|
|
$
|
10,312
|
|
|
$
|
3,001
|
|
We estimate our cash and cash equivalents are $3
million as of December 31, 2016 compared to $10.3 million as of December 31, 2015. The decrease in cash and cash equivalents resulted
from operating loss offset proceeds from issuance of shares and warrants, net.
Risk Factors
Investing in our securities involves risks. You should
carefully consider the risks described in “Risk Factors” section beginning on page 14 before making a decision to
invest in these securities. If any of these risks actually occur, our business, financial condition or results of operations would
likely be materially adversely affected. In each case, the trading price of our securities would likely decline, and you may lose
all or part of your investment. The following is a summary of some of the principal risks we face:
|
|
●
|
Our auditors have expressed substantial doubt about our ability to continue as a going concern, which may hinder our ability to obtain further financing.
|
|
|
●
|
Even if this offering is successful, we will need additional funding. If we are unable to raise capital, we will be forced to reduce or eliminate our operations.
|
|
|
●
|
We are currently implementing a cost reduction program which may be unsuccessful in its execution, and, even if successful, may lead to undesirable outcomes.
|
|
|
●
|
We have a history of operating losses and expect to incur additional losses in the future.
|
|
|
●
|
Insufficient coverage or reimbursement from medical insurers to users of our products could harm our ability to market and commercialize our current and future product.
|
|
|
●
|
We depend on the success of a limited portfolio of products for our revenue, which could impair our ability to achieve profitability.
|
|
|
●
|
We may encounter failure in the operation of our products, which may adversely harm patients operated by using our products.
|
|
|
●
|
We have only limited clinical data to support the value of the MUSE
TM
system, as well as our other products, which may make patients, physicians and hospitals reluctant to accept or purchase our products.
|
Corporate Information
Our registered office and principal
place of business are located at Omer Industrial Park, No. 7A, P.O. Box 3030, Omer 8496500, Israel and our telephone number in
Israel is + 972 72 260 2200. Our website address is http://www.medigus.com. The information contained on our website
or available through our website does not constitute part of this prospectus. Our registered agent in the United States is Medigus
USA LLC. The address of Medigus USA LLC is 140 Town & Country Dr., Suite C, Danville, CA 94526, USA.
Implications of Being an Emerging
Growth Company
We are an “emerging
growth company,” as defined in Section 2(a) of the Securities Act of 1933, as amended, or the Securities Act, as modified
by the JOBS Act. As such, we are eligible to, and intend to, take advantage of certain exemptions from various reporting requirements
applicable to other public companies that are not “emerging growth companies” such as not being required to comply
with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act of 2002. We could remain an “emerging
growth company” for up to five years, or until the earliest of (a) the last day of the first fiscal year in which our
annual gross revenue exceeds $1 billion, (b) the date that we become a “large accelerated filer” as defined
in Rule 12b-2 under the U.S. Securities Exchange Act of 1934, as amended, or the Exchange Act, which would occur if the market
value of the securities that is held by non-affiliates exceeds $700 million as of the last business day of our most recently
completed second fiscal quarter, or (c) the date on which we have issued more than $1 billion in nonconvertible debt
during the preceding three-year period.
Implications of being a Foreign
Private Issuer
We are subject
to the information reporting requirements of the Exchange Act that are applicable to “foreign private issuers,” and
under those requirements we will file reports with the SEC. As a foreign private issuer, we are not subject to the same requirements
that are imposed upon U.S. domestic issuers by the SEC. Under the Exchange Act, we are subject to reporting obligations that,
in certain respects, are less detailed and less frequent than those of U.S. domestic reporting companies. For example, although
we report our financial results on a quarterly basis, we will not be required to issue quarterly reports, proxy statements that
comply with the requirements applicable to U.S. domestic reporting companies, or individual executive compensation information
that is as detailed as that required of U.S. domestic reporting companies. We also have four months after the end of each fiscal
year to file our annual reports with the SEC and are not required to file current reports as frequently or promptly as U.S. domestic
reporting companies. We may also present financial statements pursuant to IFRS instead of pursuant to U.S. generally accepted
accounting principles. Furthermore, although the members of our management and supervisory boards will be required to notify the
Israeli Securities Authority, of certain transactions they may undertake, including with respect to our ordinary shares, our officers,
directors and principal shareholders will be exempt from the requirements to report transactions in our equity securities and
from the short-swing profit liability provisions contained in Section 16 of the Exchange Act. As a foreign private issuer, we
are also not subject to the requirements of Regulation FD (Fair Disclosure) promulgated under the Exchange Act. In addition, as
a foreign private issuer, we are permitted, and follow certain home country corporate governance practices instead of those otherwise
required under the listing rules of NASDAQ for domestic U.S. issuers (See “Risk Factors — Risks Related
to this Offering and the Ownership of the ADSs and Warrants.”) These exemptions and leniencies reduce the frequency and
scope of information and protections available to you in comparison to those applicable to a U.S. domestic reporting companies.
The
Offering
|
Class
A Units offered by us
|
|
We are offering up to 929,752 Class A Units. Each Class A Unit will
consist of (i) one ADSs, and (ii) a warrant to purchase one ADS, or a Series A warrant. The Class A Units will not be certificated
and the ADSs and Series A warrants part of such unit are immediately separable and will be issued separately in this offering.
This prospectus also relates to the offering of ADSs issuable
upon the exercise of the Series A warrants part of the Class A Units.
|
|
|
|
|
|
Class
B units offered by us
|
|
We
are also offering to those purchasers, if any, whose purchase of Class A Units in this
offering would result in the purchaser, together with its affiliates and certain related
parties, beneficially owning more than 4.99%
(or
at the election of the purchaser, 9.99%)
of our outstanding ordinary shares immediately
following the consummation of this offering, the opportunity to purchase, if they so
choose, up to 309,917 Class B units, in lieu of Class A Units that would otherwise result
in ownership in excess of 4.99%
(or 9.99%, as applicable)
of our outstanding ordinary shares.
Each Class B unit will consist of (i) a pre-funded warrant to
purchase one ADS, or a Series B warrant and (ii) and one Series A warrant. The Class B units will not be certificated
and the pre-funded Series B warrants and the Series A warrants part of such unit are immediately separable and will be
issued separately in this offering.
This prospectus also relates to the offering of ADSs issuable
upon exercise of the pre-funded Series B warrants and the Series A warrants part of the Class B Units.
|
|
|
|
|
|
Series
A Warrants
|
|
Each
Series A warrant will have an exercise price of $ per
full ADS, and will be exercisable at any time after the date of issuance and will expire
. To better understand
the terms of the Series A warrants, you should carefully read the “Description
of the Offered Securities” section of this prospectus. You should also
read the form of Series A Warrant, which is filed as an exhibit to the registration statement
that is included in this prospectus.
|
|
|
|
|
|
Pre-Funded
Series B Warrants
|
|
Each
Series B warrant will have an exercise price of $0.01 per full ADS and will be exercisable until it is exercised in full.
To better understand the terms of the Series B warrants, you should carefully read the “Description of the Offered Securities”
section of this prospectus. You should also read the form of Pre-Funded Series B Warrant, which is filed as an exhibit to
the registration statement that is included in this prospectus.
|
|
|
|
|
|
Total
ordinary shares outstanding immediately before this offering
|
|
44,142,884
ordinary shares.
|
|
|
|
|
|
Total
ordinary shares outstanding immediately after this offering
|
|
90,630,484
ordinary shares or 168,109,784 ordinary shares if the warrants offered in this offering are exercised in full.
|
|
The ADSs
|
|
Each ADS represents 50 ordinary shares. The ADSs
will be evidenced by American Depositary Receipts, or ADRs, executed and delivered by The Bank of New York Mellon, as
Depositary.
The Depositary will be the holder of the ordinary shares underlying
your ADSs and you will have rights as provided in the Deposit Agreement, among us, The Bank of New York Mellon, as Depositary,
and all owners and holders from time to time of ADSs issued thereunder, or the Deposit Agreement, a form of which has been filed
as Exhibit 1 to the Registration Statement on Form F-6 filed by The Bank of New York Mellon with the Securities and Exchange Commission
on May 7, 2015.
Subject to compliance with the relevant requirements set out in
the prospectus, you may turn in your ADSs to the Depositary in exchange for ordinary shares underlying your ADSs.
The Depositary will charge you fees for such exchanges pursuant
to the Deposit Agreement.
You should carefully read the “Description of our American
Depositary Shares” section of the accompanying prospectus and the Deposit Agreement to better understand the terms of the
ADSs.
|
|
|
|
|
|
Offering Price
|
|
The
offering price is $ per Class A unit and $ per Class
B unit. The actual offering price per Class A and Class B unit will be determined between us and the placement agent at
the time of pricing, and may be at a discount to the current market price.
|
|
|
|
|
|
Use of proceeds
|
|
We currently intend to use the net proceeds from the sale of our securities for general corporate purposes, including marketing, production and research and development related purposes. However, we have no present binding commitments or agreements to enter into any acquisitions. See “Use of Proceeds” for additional information.
|
|
|
|
|
|
Risk factors
|
|
Before deciding to invest in our securities, you should carefully consider the risks related to our business, the offering and our securities, and our location in Israel. See “Risk Factors.”
|
|
|
|
|
|
Dividend Policy
|
|
We have never declared or paid any cash dividends to our shareholders, and we currently do not expect to declare or pay any cash dividends in the foreseeable future. See “Dividend Policy.”
|
|
|
|
|
|
Listing
|
|
Our
ADSs are listed on NASDAQ under the symbol “MDGS” and our ordinary shares currently trade on the TASE in Israel under
the symbol “MDGS”. The Series A warrants and Series B warrants are not and will not be listed for trading on any national
securities exchange.
|
|
|
|
|
|
Depositary
|
|
The Bank of New York Mellon.
|
The number of ordinary shares to be
outstanding immediately after the offering as shown above is based on 44,142,884 ordinary shares outstanding as of March 15, 2017.
This number does not include, as of such date (i) 1,346,964 ordinary shares issuable upon the exercise of outstanding options
at a weighted average exercise price of NIS 5.20 per share or $1.42 per share (based on the exchange rate reported by the Bank
of Israel on such date), equivalent to 26,939 ADSs at a weighted average exercise price of $71.11 per ADS, (ii) 9,427,443 ordinary
shares issuable upon the exercise of outstanding warrants at a weighted average exercise price of NIS 4.44 per share or $1.21
per share (based on the exchange rate reported by the Bank of Israel on such date), equivalent to 188,549 ADSs at a weighted average
exercise price of $60.66 per ADS, (iii) 77,479,300 ordinary shares issuable upon the exercise of Series A warrants and Series
B warrants to purchase 1,549,586 ADSs to be issued in this offering, and (iv) 4,338,850 ordinary shares issuable upon the exercise
of warrants to purchase 86,777 ADSs, to be issued to the placement agent in connection with the offering.
Unless otherwise stated, all information in this
prospectus (i) assumes no exercise of the outstanding options and warrants and offered warrants into ordinary shares or ADSs as
described above, (ii) is adjusted retroactively to reflect the
change in the ratio of ordinary
shares per ADS from five deposited ordinary shares per ADS to 50 deposited ordinary shares per ADS effected on March 15, 2017
and (iii) is adjusted
retroactively to reflect
the 10:1 reverse share split and the
change in the ratio of ordinary shares per ADS to five deposited ordinary shares per ADS effected on November 6, 2015.
Summary
Financial Data
We
derived the summary financial statement data for the years ended December 31, 2013, 2014 and 2015 set forth below from our
audited consolidated financial statements and related notes included elsewhere in this prospectus. We derived the summary financial
statement data for the nine months ended September 30, 2015 and 2016 from our unaudited condensed interim financial statements
and related notes included elsewhere in this prospectus. Our results for interim periods are not necessarily indicative of the
results that may be expected for the entire year. You should read the information presented below together with our financial
statements, the notes to those statements and the other financial information included elsewhere in this prospectus.
The
December 31, 2015 consolidated financial statements included in this registration statement are presented in New Israeli Shekels,
which was the Company’s functional and presentation currency as of such date. Effective January 1, 2016, the Company changed
its functional currency to the U.S. dollar from the New Israeli Shekel. The December 31, 2015 financial data presented in this
prospectus was translated from NIS to USD as follows: (1) all assets and liabilities of the Company were translated using the
dollar exchange rate as of December 31 2015; (2) equity items were translated using historical exchange rates at the relevant
transaction dates; (3) the statement of comprehensive loss items has been translated at the average exchange rates for the year
2015; and (4) the resulting translation differences have been reported as “currency translation differences” within
other comprehensive loss.
|
|
|
Year Ended December 31,
|
|
|
Nine Months ended
September 30,
|
|
|
|
|
2013
|
|
|
2014
|
|
|
2015
|
|
|
2015
|
|
|
2016
|
|
|
|
|
|
|
|
(unaudited)
|
|
|
|
|
(U.S. Dollars, in thousands, except per share and weighted average shares data)
|
|
|
Consolidated Statements of Loss and Other Comprehensive Loss
|
|
|
|
|
Revenues
|
|
$
|
691
|
|
|
$
|
744
|
|
|
$
|
624
|
|
|
$
|
399
|
|
|
$
|
496
|
|
|
Cost of revenues
|
|
|
311
|
|
|
|
351
|
|
|
|
277
|
|
|
|
169
|
|
|
|
154
|
|
|
Gross profit
|
|
|
380
|
|
|
|
393
|
|
|
|
347
|
|
|
|
230
|
|
|
|
342
|
|
|
Operating Expenses
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Research and development expenses, net
|
|
|
2,275
|
|
|
|
4,025
|
|
|
|
4,384
|
|
|
|
3,043
|
|
|
|
3,021
|
|
|
Selling and marketing expenses
|
|
|
900
|
|
|
|
2,341
|
|
|
|
2,680
|
|
|
|
1,943
|
|
|
|
1,845
|
|
|
General and administrative expenses
|
|
|
1,908
|
|
|
|
2,280
|
|
|
|
2,842
|
|
|
|
1,911
|
|
|
|
3,016
|
|
|
Other income, net
|
|
|
181
|
|
|
|
269
|
|
|
|
3
|
|
|
|
3
|
|
|
|
|
|
|
Total operating expenses
|
|
|
4,902
|
|
|
|
8,377
|
|
|
|
9,903
|
|
|
|
6,894
|
|
|
|
7,882
|
|
|
Operating loss
|
|
|
(4,522
|
)
|
|
|
(7,984
|
)
|
|
|
(9,556
|
)
|
|
|
(6,664
|
)
|
|
|
(7,540
|
)
|
|
Profit from changes in fair value of warrants issued to investors
|
|
|
3,228
|
|
|
|
980
|
|
|
|
106
|
|
|
|
15
|
|
|
|
17
|
|
|
Financial income (expenses), net
|
|
|
(111
|
)
|
|
|
650
|
|
|
|
(14
|
)
|
|
|
28
|
|
|
|
118
|
|
|
Loss before taxes on income
|
|
|
(1,405
|
)
|
|
|
(6,354
|
)
|
|
|
(9,464
|
)
|
|
|
(6,621
|
)
|
|
|
(7,405
|
)
|
|
Taxes on income
|
|
|
(23
|
)
|
|
|
(4
|
)
|
|
|
(68
|
)
|
|
|
(50
|
)
|
|
|
(24
|
)
|
|
Loss for the period
|
|
$
|
(1,428
|
)
|
|
$
|
(6,358
|
)
|
|
$
|
(9,532
|
)
|
|
$
|
(6,671
|
)
|
|
$
|
(7,429
|
)
|
|
Other comprehensive income (loss) for the period, net of tax
|
|
|
408
|
|
|
|
(1,573
|
)
|
|
|
(211
|
)
|
|
|
(298
|
)
|
|
|
|
|
|
Total comprehensive loss for the period
|
|
$
|
(1,020
|
)
|
|
$
|
(7,931
|
)
|
|
$
|
(9,743
|
)
|
|
$
|
(6,969
|
)
|
|
$
|
(7,429
|
)
|
|
Basic and diluted loss per share
|
|
$
|
(0.11
|
)
|
|
$
|
(0.33
|
)
|
|
$
|
(0.34
|
)
|
|
$
|
(0.25
|
)
|
|
$
|
(0.22
|
)
|
|
Weighted average of ordinary shares (in thousands)
|
|
|
13,020
|
|
|
|
19,500
|
|
|
|
28,415
|
|
|
|
27,190
|
|
|
|
33,369
|
|
|
|
|
As
of September 30, 2016
|
|
|
|
|
Actual
|
|
|
Pro
forma
(1)
|
|
|
Pro
forma as
adjusted
(2)(3)
|
|
|
|
|
(in
thousands)
|
|
|
Consolidated
Balance Sheet Data:
|
|
|
|
|
|
|
|
|
|
|
Cash
and cash equivalents
|
|
$
|
3,868
|
|
|
$
|
4,475
|
|
|
$
|
10,989
|
|
|
Working
capital
(4)
|
|
|
3,155
|
|
|
|
3,762
|
|
|
|
10,276
|
|
|
Total
assets
|
|
|
6,040
|
|
|
|
6,647
|
|
|
|
13,161
|
|
|
Total
long-term liabilities
|
|
|
133
|
|
|
|
386
|
|
|
|
6,029
|
|
|
Total
shareholders’ equity
|
|
|
4,069
|
|
|
|
4,423
|
|
|
|
5,294
|
|
(1) Pro forma gives effect
to the issuance and sale of 113,917 ADSs representing 5,695,850 ordinary shares in the offering we closed on December 6,
2016.
(2) Pro forma as adjusted
gives further effect to the issuance and sale in this offering of an aggregate of 1,239,669 units at the assumed public
offering price of $6.05 per Class A unit, the last reported sales price of our ADSs on the NASDAQ on March 22, 2017, and of $6.04
per Class B unit, after deducting the estimated placement agent’s fees and estimated offering expenses payable by us.
|
(3)
|
A
$1.00 increase (decrease) in the assumed public offering price per unit, would increase (decrease) the as pro forma adjusted amount
of each of cash and cash equivalents by approximately $1.14 million and total shareholders’ equity by approximately $208,000,
assuming that the number of units offered by us, as set forth on the cover page of this prospectus, remains the same and after
deducting estimated placement agent’s fees and estimated offering expenses payable by us. A 10,000 units increase in the
number of units offered by us together with a concomitant $1.00 increase in the assumed public offering price per unit would increase
our pro forma as adjusted cash and cash equivalents by approximately $1.21 million and total shareholders’ equity by approximately
$219,000, after deducting estimated placement agent’s fees and estimated offering expenses payable by us. Conversely, a
10,000 unit decrease in the number of units offered by us together with a concomitant $1.00 decrease in the assumed public offering
price per unit would decrease our pro forma as adjusted cash and cash equivalents by approximately $1.19 million and total shareholders’
equity by approximately $216,000, after deducting estimated placement agent’s fees and estimated offering expenses payable
by us.
|
|
(4)
|
Working capital is defined as total current assets minus total current liabilities.
|
RISK
FACTORS
You
should carefully consider the risks described below, as well as the other information included in this prospectus, including our
financial statements and the related notes, before you decide to buy our securities. The risks and uncertainties described below
are not the only risks facing us. We may face additional risks and uncertainties not currently known to us or that we currently
deem to be immaterial. Any of the risks described below, and any such additional risks, could materially adversely affect our
business, financial condition or results of operations. In such case, you may lose all or part of your original investment.
Risks
Related to Our Business
Our
auditors have expressed substantial doubt about our ability to continue as a going concern, which may hinder our ability to obtain
further financing.
Our
audited financial statements for the year ended December 31, 2015, were prepared under the assumption that we would continue
our operations as a going concern. Our independent registered public accounting firm has included a “going concern”
explanatory paragraph in its report on our financial statements for the year ended December 31, 2015, indicating that
we
have suffered recurring losses from operations and have a net capital deficiency that raises substantial doubt about our ability
to continue as a going concern
. Uncertainty concerning our ability to continue as a going concern may hinder our ability
to obtain future financing. Continued operations and our ability to continue as a going concern are dependent on our ability to
obtain additional funding in the near future and thereafter, and there are no assurances that such funding will be available to
us at all or will be available in sufficient amounts or on reasonable terms. Our financial statements do not include any adjustments
that may result from the outcome of this uncertainty. Without additional funds from private and/or public offerings of debt or
equity securities, sales of assets, sales or out-licenses of intellectual property or technologies, or other transactions, we
will exhaust our resources and will be unable to continue operations. If we cannot continue as a viable entity, our shareholders
would likely lose most or all of their investment in us.
Even
if this offering is successful, we will need additional funding. If we are unable to raise capital, we will be forced to reduce
or eliminate our operations.
As of March 15, 2017, we had a total cash balance
of approximately $1.7 million. Based on our projected cash flows and our cash balances as of the date of this prospectus,
our management is of the opinion that without further fund raising we will not have sufficient resources to enable us to continue
advancing our activities and as a result, there is substantial doubt about our ability to continue as a going concern. At our
current burn rate and without taking into account the proceeds from this offering, our current cash balance will be sufficient
until approximately April 2017, taking into account shut down costs. Assuming the proceeds from this offering will be approximately
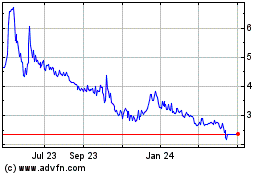
$6.5 million, based upon an assumed public offering price of $6.05 per Class A unit, the last reported sales price of our ADSs
on the NASDAQ on March 22, 2017, and of $6.04 per Class B unit, after deducting the estimated placement agent fee and estimated
offering expenses payable by us, at our expected burn rate following this offering, the proceeds from this offering will be sufficient
until approximately January 2018.
Even
if this offering is successful, if we are unable to obtain additional sufficient financing, we will be forced to reduce the scope
of, or eliminate our operations. We will also have to reduce marketing, customer service or other resources devoted to our products.
Any of these factors will materially harm our business and results of operations.
Our
management’s plans include the continued commercialization of our products, taking cost reduction steps and securing
sufficient financing through the sale of additional equity securities, debt or capital inflows from strategic partnerships.
There are no assurances however, that we will be successful in obtaining the level of financing needed for our operations.
Even
if we are able to continue to finance our business, the sale of additional equity or debt securities could result in dilution
to our current shareholders and could require us to grant a security interest in our assets. If we raise additional funds through
the issuance of debt securities, these securities may have rights senior to those of our ordinary shares and could contain covenants
that could restrict our operations. In addition, we may require additional capital beyond our currently forecasted amounts to
achieve profitability. Any such required additional capital may not be available on reasonable terms, or at all.
We
are currently implementing a cost reduction program which may be unsuccessful in its execution, and, even if successful, may lead
to undesirable outcomes.
We
are currently implementing a cost reduction program that will affect the structure and operation of our business. Such plan reflects
assumptions and analyses based on our experience and perception of historical trends, current conditions and expected future developments
as well as other factors that we consider appropriate under the circumstances. Whether our cost reduction program will prove successful
depends on a number of factors, including but not limited to (i) our ability to substantially raise additional funding and to
obtain adequate liquidity; (ii) our ability to maintain suppliers’, hospitals’, medical facilities’ and practitioners’
confidence; (iii) our ability to efficiently reduce our operational expenditures, while retaining key employees; and (iv) the
overall success of our business. In addition, as long as these cost reduction measurements last, and for a substantial time afterwards,
our employees may face considerable distraction and uncertainty and we may experience increased levels of employee attrition.
A loss of key personnel could have a material adverse effect on our ability to meet operational and financial expectations. The
pursuit of additional funding and the application of the cost reduction program has occupied and will continue to occupy a substantial
portion of the time and attention of our management and will impact how our business is conducted.
We
have a history of operating losses and expect to incur additional losses in the future.
We
have sustained losses in recent years, which as of September 30, 2016, accumulated to $52 million, including an operating net
loss of $9.6 million and $7.5 million for the year ended December 31, 2015 and the nine months ended September 30, 2016, respectively.
We anticipate that we are likely to continue to incur significant net losses for at least the next several years as we continue
the development of the MUSE
TM
system and potentially other products, expand our sales and marketing capabilities in the endoscopy-based
products market, continue our commercialization of our MUSE
TM
system, expand our adoption and clinical implementation, and
continue to develop the corporate infrastructure required to sell and market our products. Our losses have had, and will continue
to have, an adverse effect on our shareholders’ equity and working capital. Any failure to achieve and maintain profitability
would continue to have an adverse effect on our shareholders’ equity and working capital and could result in a decline in
our share price or cause us to cease operations.
The
future success of our business depends on our ability to continue to develop and obtain regulatory clearances or approvals for
innovative and commercially successful products in our field, which we may be unable to do in a timely manner, or at all. Our
success and ability to generate revenue or be profitable also depends on our ability to establish our sales and marketing force,
generate product sales and control costs, all of which we may be unable to do.
The commercial success of the MUSE
TM
system or any
future product, if approved, depends upon the degree of market acceptance by physicians, patients, third-party payors, and others
in the medical community.
The commercial success of the MUSE
TM
system and any future product, if approved, depends in part on the medical community, patients, and third-party payors accepting
our products as medically useful, cost-effective, and safe. Any product that we bring to the market may or may not gain market
acceptance by physicians, patients, third-party payors, and others in the medical community. In addition, since the MUSE
TM
system is a therapeutic device being used for a quality of life, benign disease, market penetration may be more difficult.
To date, we have experienced slower than expected market penetration. If the MUSE
TM
system or any future product, if
approved, does not achieve an adequate level of acceptance, we may not generate significant product revenue and may not become
profitable. The degree of market acceptance of these products, if approved for commercial sale, will depend on a number of factors,
including:
|
|
●
|
the cost, safety, efficacy, and convenience
of the MUSE
TM
system and any future product in relation to alternative treatments and products;
|
|
|
●
|
the ability of third parties to enter
into relationships with us without violating their existing agreements;
|
|
|
●
|
the effectiveness of our sales and marketing
efforts;
|
|
|
●
|
the prevalence and severity of any side
effects resulting from the procedure;
|
|
|
●
|
the willingness of the target patient
population to try new procedures and of physicians to perform new procedures;
|
|
|
●
|
the strength of marketing and distribution
support for, and timing of market introduction of, competing products;
|
|
|
●
|
publicity concerning our products or
competing products and treatments; and
|
|
|
●
|
sufficient third-party insurance coverage
or reimbursement.
|
Even if the MUSE
TM
system and
any future product, if approved, displays a favorable safety and efficacy profile in clinical trials, market acceptance of the
product will not be known until after it is launched. Our efforts to educate the medical community and third-party payors on the
benefits of the products may require significant resources and may never be successful. Such efforts to educate the marketplace
may require more resources than are required by conventional technologies.
Insufficient
coverage or reimbursement from medical insurers to users of our products could harm our ability to market and commercialize our
current and future products.
Our
ability to successfully commercialize our products, mainly the MUSE
TM
system, depends significantly on the
availability of coverage and reimbursement for endoscopic procedures from third-party insurers, including governmental
programs, as well as private insurance and private health plans. Reimbursement is a significant factor considered by
hospitals, medical facilities and practitioners in determining whether to acquire and utilize new capital equipment or to
implement new procedures such as our technology.
In January 2016, the American Medical Association’s (AMA) Current
Procedural Terminology (CPT) published a new Category I CPT Code for transoral esophagogastric fundoplasty procedures, which
describes procedures conducted with the MUSE
TM
system. In the U.S., the CPT Editorial Panel assigns specific
billing codes for physician services and outpatient hospital procedures, which are used by providers, who are our customers,
to bill for procedures. Once a CPT code is established, the Centers for Medicare and Medicaid Services (CMS) in
turn establishes payment levels and coverage rules under Medicare, and private payors establish rates and coverage rules.
Notwithstanding the issuance of a CPT to report the MUSE procedure and the establishment of payment rates for the code, we
cannot guarantee that the MUSE
TM
system is or will be covered and, if covered, that reimbursement will be
sufficient, and furthermore, we cannot guarantee that the MUSE
TM
system or any future product will be approved for
coverage or reimbursement by Medicare, Medicaid or any third-party payor. Reimbursement decisions in the European Union
and in other jurisdictions outside of the United States vary by country and region and there can be no assurance that we will
be successful in obtaining adequate reimbursement.
We
depend on the success of a limited portfolio of products for our revenue, which could impair our ability to achieve
profitability.
Though
we have plans for the development of additional natural orifice surgical products based on our technology, including miniature
cameras, flexible stapling and ultrasound, and although we currently derive most of our revenue from the sale of miniature
cameras and related imaging equipment, we plan to derive most of our future revenue from product sales of our imaging equipment
and our flagship MUSE
TM
system and its future applications, as well as recurring sales of associated
products required to use the MUSE
TM
system. Our future growth and success is dependent on the successful commercialization
of the MUSE
TM
system. If we are unable to achieve increased commercial acceptance of the MUSE
TM
system,
obtain regulatory clearances or approvals for future products, or experience a decrease in the utilization of our product line
or procedure volume, our revenue would be adversely affected.
We
may encounter manufacturing issues during the assembly process of our flagship product.
Due
to the characteristics of the technologies on which the main parts of the MUSE
TM
system are manufactured, which include plastic
and metal injection, sheet metals, laser welding and rubber vulcanization, using production tools such as molds, templates and
jigs, in the event that parts are found which are inaccurate and/or which have been rendered defective and/or which have failed
preliminary tests, we will be forced to repair the manufacturing tools and re-manufacture and/or re-order the parts, a process
which will delay the production timetable. Furthermore, in the event that certain parts are not suitable, due to a situation whereby
the manufacturing tools have not produced the part in the appropriate manner, it may be necessary to redesign and re-manufacture
the manufacturing tool and to manufacture the parts rapidly and at additional cost.
Furthermore,
if we are unable to satisfy commercial demand for our MUSE
TM
system due to our inability to assemble, test
and deliver the system in compliance with applicable regulations, our business and financial results, including our ability to
generate revenue, would be impaired, market acceptance of our products could be materially adversely affected and customers may
instead purchase or use competing products.
We
may encounter failure in the operation of our products, which may adversely harm patients operated by using our products.
Users
of our products may encounter failures in mechanical components, which could result in difficulties in operation, or opening or
releasing the products, leading to the need for surgical procedures to correct the mechanical failure, in which case, a patients’
medical condition may worsen.
Additionally,
in the event that users of our products do not follow the instructions for use or the available product training or instructions
(which appear on the screen during the performance of the procedure) the foregoing may cause injury and in certain cases, could
even cause death. A result of this kind could reduce the rate of progress of, or even prevent, the marketing for the MUSE
TM
product and our other products.
Furthermore,
users of our products may encounter failure in electronic components of our products used in the system software, which could
lead to incorrect interpretation by the users or to failure in the operation of the endoscope, and to injury to the patient’s
critical internal organs.
We
have only limited clinical data to support the value of the MUSE
TM
system, as well as our other products, which may make
patients, physicians and hospitals reluctant to accept or purchase our products.
Physicians,
hospitals and patients will only accept or purchase our products if they believe them to be safe and effective, with advantages
over competing products or procedures. To date, we have collected only limited clinical data with which to assess our products’
(mainly the MUSE
TM
system) clinical and economic value. The collection of clinical and economic data and the process of generating
peer review publications in support of our product and procedure is an ongoing focus for us.
If
future publications of clinical studies indicate that medical procedures using the MUSE
TM
system are less safe or less effective
than competing products or procedures, patients may choose not to undergo our procedure, and physicians or hospitals may choose
not to purchase or use our system. Furthermore, unsatisfactory patient outcomes or patient injury could cause negative publicity
for our products, particularly in the early phases of product introduction.
Current
economic conditions could delay or prevent our customers from obtaining budgetary approval to purchase a MUSE
TM
system or
other products, which would adversely affect our business, financial condition and results of operations.
As
a result of the concerns relating to the current economic situation or related to ongoing healthcare reimbursement changes, customers
and distributors may be delayed in obtaining, or may not be able to obtain, budgetary approval or financing for their purchases
or leases of medical equipment including our products. These delays may in some instances lead to our customers or distributors
postponing the shipment and use of previously ordered systems and products, cancelling their orders, or cancelling their agreements
with us. An increase in delays and order cancellations of this nature could adversely affect our products sales and revenues and,
therefore, harm our business and results of operations.
In
addition, negative worldwide economic conditions and market instability may make it increasingly difficult for us, our customers,
our distributors and our suppliers to accurately forecast future product demand trends, which could cause us to order or produce
excess products that can increase our inventory carrying costs and result in obsolete inventory. Alternatively, this forecasting
difficulty could cause a shortage of products, or materials used in our products, that could result in an inability to satisfy
demand for our products and a resulting material loss of potential revenue.
Our reliance on third-party
suppliers for most of the components of our products could harm our ability to meet demand for our products
in a timely and cost effective manner.
Though
we attempt to ensure the availability of more than one supplier for each important component in our products, the number
of suppliers engaged in the provision of miniature video sensors which are suitable for our CMOS technology products is very limited,
and therefore in some cases we engage with a single supplier, which may result in dependency on such supplier. This is the case
regarding sensors for the CMOS type technology that is produced by a single supplier in the United States. As we do not have a
contract in place with this supplier, there is no contractual commitment on the part of such supplier for any set quantity of
such sensors. The loss of our sole supplier in providing us with miniature sensors for our CMOS technology products, and our inability
or delay in finding a suitable replacement supplier, could significantly affect our business, financial condition, results of
operations and reputation.
Modifications
to our current regulator-cleared products or the introduction of new products may require new regulatory clearances or approvals
or require us to recall or cease marketing our current products until clearances or approvals are obtained.
Our
MUSE
TM
system has received marketing clearance from the U.S. Food and Drug Administration, or FDA, based on several 510(k)
applications, bears the CE Mark (a mark assigned to a product certifying its fulfillment of the Medical Devices Directive of the
European Union), as required in order to market the system in European Union countries and has obtained the necessary license
to market the product in Canada, Turkey and Israel.
Ongoing
modifications to our products may require new regulatory approvals, as with prior 510k clearances, or require us to recall or
cease marketing the modified products until these clearances or approvals are obtained. Any modification to one of our cleared
devices that would constitute a major change in its intended use, or any change that could significantly affect the safety or
effectiveness of the device would require us to obtain a new 510(k) marketing clearance and may even, in some circumstances, require
the submission of a premarket approval, or PMA, track application if the change raises complex or novel scientific issues or the
product has a new intended use. The FDA requires every manufacturer to make the determination regarding the need for a new 510(k)
submission in the first instance, but the FDA may review any manufacturer’s decision. We may make modifications in the future
to the MUSE
TM
system without seeking additional clearances or approvals if we believe such clearances or approvals are not
necessary. However, it is possible that the FDA could change existing policy and practices regarding the assessment of whether
a new 510(k) clearance is required for changes or modifications to existing devices. Under these changed circumstances, the FDA
may disagree with our past or future decisions not to seek a new 510(k) for changes or modifications to existing devices and require
new clearances or approvals. In that case, we may be required to recall and stop marketing our products as modified, which could
require us to redesign our products, conduct clinical trials to support any modifications, and pay significant regulatory fines
or penalties. In addition, the FDA may not approve or clear our products for the indications that are necessary or desirable for
successful commercialization or could require additional clinical trials to support any modifications.
Significant
changes that could be reasonably expected to affect the safety or effectiveness of one of our devices may require us to obtain
a license amendment or possibly a new license from Health Canada, Turkey, or Israel. In addition, we started the process for receiving
a regulatory clearance in China by the China Food and Drug Administration, which could be significantly affected by such changes.
Substantial changes to the quality system or changes to the CE marked device which could affect compliance with the essential
requirements of the device or its intended use must be reported to the Notified Body (an independent and neutral institution appointed
to conduct conformity assessment). This may result in a decision that an existing certificate is valid, an addendum to the certificate
is needed or a new certificate must be obtained. Any failure to maintain our existing clearances or approvals, or delay or failure
in obtaining required clearances or approvals would adversely affect our ability to introduce new or enhanced products in a timely
manner, which in turn would harm our future growth. Any of these actions would harm our operating results. Further, we may also
be required to seek regulatory clearance in additional countries as we expand our marketing efforts.
Moreover,
clearances and approvals by the applicable regulator are subject to continual review, and the later discovery of previously unknown
problems can result in product labeling restrictions or withdrawal of the product from the market. The loss of previously received
approvals or clearances, or the failure to comply with existing or future regulatory requirements could reduce our sales, profitability
and future growth prospects.
We
are currently required by the FDA to refrain from using certain terms to label and market our products, which could harm our ability
to market and commercialize our current or future products.
The
FDA’s 510(k) clearances include a specification of a product’s indication for use, and also authorize specific labeling
and marketing claims and language in promotional materials for the U.S. market. Failure to conform with the specific cleared labeling
of our products or corporate promotional material would be considered mislabeling or off-label promotion which might lead to:
|
|
●
|
untitled
letters, warning letters, fines, injunctions, consent decrees and civil penalties;
|
|
|
|
|
|
|
●
|
customer
notifications, refunds, detention or seizure of our products;
|
|
|
|
|
|
|
●
|
refusing
or delaying requests for 510(k) marketing clearance or PMA approvals of new products or modified products;
|
|
|
|
|
|
|
●
|
withdrawing
510(k) marketing clearances or PMA approvals that have already been granted;
|
|
|
|
|
|
|
●
|
refusing
to provide Certificates for Foreign Government;
|
|
|
|
|
|
|
●
|
refusing
to grant export approval for our products; or
|
|
|
|
|
|
|
●
|
pursuing
criminal prosecution.
|
Any
of these sanctions could impair our ability to produce our products in a cost-effective and timely manner in order to meet our
customers’ demands. We may also be required to bear other costs or take other actions that may have a negative impact on
our future sales and financial condition.
If
we or our third-party manufacturers or suppliers fail to comply with the FDA’s Quality System Regulation or other regulatory
authorities, our manufacturing operations could be interrupted and our product sales and operating results could suffer
.
We
and some of our third-party manufacturers and suppliers are required to comply with the FDA’s Quality System Regulation,
or QSR, which covers the methods and documentation of the design, testing, production, control, quality assurance, labeling, packaging,
sterilization, storage and shipping of our products. We and our manufacturers and suppliers are also subject to the regulations
of foreign jurisdictions regarding the manufacturing process if we or our distributors market our products abroad. We monitor
our quality management in order to improve our overall level of compliance. Our facilities are subject to periodic and unannounced
inspection by U.S. and foreign regulatory agencies, including notified bodies, to audit compliance with the QSR and comparable
foreign regulations. If our facilities or those of our third-party manufacturers or suppliers are found to be in violation of
applicable laws and regulations, or if we or our manufacturers or suppliers fail to take satisfactory corrective action in response
to an adverse inspection, the regulatory authority could take enforcement action, including any of the following sanctions:
|
|
●
|
untitled
letters, warning letters, fines, injunctions, consent decrees and civil penalties;
|
|
|
●
|
customer
notifications or repair, replacement, refunds, detention or seizure of our products;
|
|
|
●
|
operating
restrictions or partial suspension or total shutdown of production;
|
|
|
●
|
refusing
or delaying requests for 510(k) marketing clearance or PMA approvals of new products or modified products;
|
|
|
●
|
withdrawing
510(k) marketing clearances that have already been granted, or PMA approvals that we may receive in the future;
|
|
|
●
|
refusing
to provide Certificates for foreign government;
|
|
|
●
|
refusing
to grant export approval for our products; or
|
|
|
●
|
pursuing
criminal prosecution.
|
Any
of these sanctions could impair our ability to produce our products in a cost-effective and timely manner in order to meet our
customers’ demands, and could have a material adverse effect on our reputation, business, results of operations and financial
condition. We may also be required to bear other costs or take other actions that may have a negative impact on our future sales
and our ability to generate profits.
We
face possible competition from the pharmaceutical sector, which could harm our ability to market and commercialize our current
and future products.
The
development of more powerful drug treatments to assist in the suppression of
Gastroesophageal
Reflux Disease
, or GERD, or other medical problems which compete with our products, may
reduce the size of our target markets and may reduce the need for the use of our systems and products, either available now, or
which will be developed in the future, thus adversely affecting our ability to market and commercialize our current and future
products. While we are unaware of any current pharmaceutical product that could directly compete with the MUSE
TM
system at
this time, there may be new pharmaceutical entrants in the future.
There
can be no assurance that we will be able to compete successfully against current or future competitors or that competition will
not have a material adverse effect on our future revenues and, consequently, on our business, operating results and financial
condition.
We
face competition from medical device companies that develop and market similar related products and systems, or may launch
products in the future, as well as new techniques and devices for treatments performed by our products.
Several
medical device companies have commercial products which compete with the MUSE
TM
system for the treatment of GERD
using an endoscopic method. While we believe that the MUSE
TM
system has several advantages over competing devices, such as
the requirement of one operator, inclusion of visualization and ultrasound apparatuses, use of standard titanium staples, and
reduced risk of harm to adjacent organs, there can be no assurance that we will be able to compete successfully against current
or future competitors or that competition will not have a material adverse effect on our future revenues and, consequently, on
our business, operating results and financial condition.
Reporting
requirements on payments to physicians in the United States may deter doctors from providing advice to the Company.
The
implementation of the reporting and disclosure obligations of the Physician Payment Sunshine Act, which is part of the Affordable
Care Act of 2010, or the Sunshine Act, could adversely affect our business.
The
Sunshine Act has imposed new reporting and disclosure requirements for drug and device manufacturers with regard to payments or
other transfers of value made to certain practitioners (including physicians, dentists and teaching hospitals), and for such manufacturers
and for group purchasing organizations with regard to certain ownership interests held by physicians in the reporting entity.
On February 1, 2013, Centers for Medicare & Medicaid Services, or CMS, released the final rule to implement the
Sunshine Act. Under this rule, data collection activities began on August 1, 2013, and first disclosure reports were due by March
31, 2014, for the period August 1, 2013, through December 31, 2013. As required under the Sunshine Act, CMS publishes information
from these reports on a publicly available website, including amounts transferred and physician, dentist and teaching hospital
identities.
The final rule
implementing the Sunshine Act is complex, ambiguous, and broad in scope. Accordingly, we are required to collect and report detailed
information regarding certain financial relationships we have with U.S. licensed physicians, dentists (if any) and teaching hospitals
in the United States. It is difficult to predict how the new requirements may impact existing relationships among manufacturers,
distributors, physicians, dentists and teaching hospitals. The Sunshine Act preempts similar state reporting laws, although we,
or our subsidiaries, may be required to continue to report under certain of such state laws. While we expect to have substantially
compliant programs and controls in place to comply with the Sunshine Act requirements, and we have completed our initial registration
with CMS and our 2015 report with respect to Sunshine Act reporting, our continued compliance with the Sunshine Act imposes
continuing additional costs on us.
Medical
device development is costly and involves continual technological change which may render our current or future products obsolete.
Innovation
is rapid and continuous in the medical device industry, and our competitors in the medical device industry make significant investments
in research and development. If new products or technologies emerge that provide the same or superior benefits as our products
at equal or lower cost, they could render our products obsolete or unmarketable. We must anticipate changes in the marketplace
and the direction of technological innovation and customer demands. In addition, we face increasing competition from well-financed
medical device companies to develop new technologies and may face competition should we attempt to acquire new technologies, products
and businesses. As a result, we cannot be certain that our products will be competitive with current or future products and technologies.
We
may be subject to product liability claims, product actions, including product recalls, and other field or regulatory actions
that could be expensive, divert management’s attention and harm our business.
Our
business exposes us to potential liability risks, product actions and other field or regulatory actions that are inherent in the
manufacturing, marketing and sale of medical device products. We may be held liable if our products cause injury or death or is
found otherwise unsuitable or defective during usage. The MUSE
TM
system incorporates mechanical and electrical parts, complex
computer software and other sophisticated components, any of which can contain errors or failures. Complex computer software is
particularly vulnerable to errors and failures, especially when first introduced. In addition, new products or enhancements to
our existing products may contain undetected errors or performance problems that, despite testing, are discovered only after installation.
If
any of our products are defective, whether due to design or manufacturing defects, improper use of the product, or other reasons,
we may voluntarily or involuntarily undertake an action to remove, repair, or replace the product at our expense. In some circumstances
we will be required to notify regulatory authorities of an action pursuant to a product failure.
The
medical device industry has historically been subject to extensive litigation over product liability claims. We anticipate that
as part of our ordinary course of business we will be subject to product liability claims alleging defects in the design, manufacture
or labeling of our products. A product liability claim, regardless of its merit or eventual outcome, could result in significant
legal defense costs and high punitive damage payments. Although we maintain product liability insurance, the coverage may not
be adequate to cover future claims. Additionally, we may be unable to maintain our existing product liability insurance in the
future at satisfactory rates or adequate amounts.
Broad-based
domestic and international government initiatives to reduce spending, particularly those related to healthcare costs, may reduce
reimbursement rates for endoscopic procedures, which will reduce the cost-effectiveness of our products.
Healthcare
reforms, changes in healthcare policies and changes to third-party coverage and reimbursements, including legislation enacted
reforming the U.S. healthcare system, and any future changes to such legislation, may affect demand for our products and may have
a material adverse effect on our financial condition and results of operations. There can be no assurance that current levels
of reimbursement will not be decreased in the future, or that future legislation, regulation, or reimbursement policies of third-parties
will not adversely affect the demand for our products or our ability to sell products on a profitable basis. The adoption of significant
changes to the healthcare system in the United States, Europe or other jurisdictions in which we may market our products, could
limit the prices we are able to charge for our products or the amounts of reimbursement available for our products, could limit
the acceptance and availability of our products, reduce medical procedure volumes and increase operational and other costs. For
example, U.S. President Donald Trump has recently publicly indicated an intent to lower healthcare costs through various potential
initiatives. In addition, President Trump and other U.S. lawmakers have made statements about potentially repealing or replacing
the Affordable Care Act, although specific legislation for such a repeal or replacement is still in its early stages. While we
are unable to predict what changes may ultimately be enacted, to the extent that future changes affect how our products are paid
for and reimbursed by government and private payers our business could be adversely impacted.
We
cannot predict whether future healthcare initiatives will be implemented at the federal or state level or internationally, or
the effect that any future legislation or regulation will have on us. The expansion of government’s role in any country’s
healthcare industry may result in decreased profits to us, lower reimbursements by third-parties for procedures in which our products
are used, and reduced medical procedure volumes, all of which may adversely affect our business, financial condition and results
of operations.
We
depend on key employees, and if we fail to attract and retain employees with the expertise required for our business and provide
for the succession of senior management, we cannot grow or achieve profitability.
We
are dependent on the continued service and performance of members of our senior management and other key personnel, for example
our Chief Executive Officer, Chris Rowland. We do not maintain key-man life insurance. Our future success will depend
in part on our ability to retain our management and scientific teams, to identify, hire and retain additional qualified personnel
with expertise in research and development and sales and marketing, and to effectively provide for the succession of
senior management. Competition for qualified personnel in the medical device industry is intense. We may be unable to replace
key persons if they leave or to fill new positions requiring key persons with appropriate experience.
The
loss of key employees, the failure of any key employee to perform or our inability to attract and retain skilled employees, as
needed, or an inability to effectively plan for and implement a succession plan for key employees could harm our business.
If
we or our contractors or service providers fail to comply with regulatory laws and regulations, we or they could be subject to
regulatory actions, which could affect our ability to develop, market and sell our products and any other or future products that
we may develop and may harm our reputation.
If
we or our manufacturers or other third-party contractors fail to comply with applicable federal, state or foreign laws or regulations,
we could be subject to regulatory actions, which could affect our ability to develop, market and sell our current products or
any future products which we may develop in the future and could harm our reputation and lead to reduced demand for or non-acceptance
of our proposed products by the market.
If
our employees commit fraud or other misconduct, including noncompliance with regulatory standards and requirements and insider
trading, our business may experience serious adverse consequences.
We
are exposed to the risk of employee fraud or other misconduct. Misconduct by employees could include intentional failures to comply
with FDA regulations, to provide accurate information to the FDA, to comply with manufacturing standards we have established,
to comply with federal and state health-care fraud and abuse laws and regulations, to report financial information or data accurately
or to disclose unauthorized activities to us. In particular, sales, marketing and business arrangements in the healthcare industry
are subject to extensive laws and regulations intended to prevent fraud, kickbacks, self-dealing and other abusive practices.
These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, sales commission,
customer incentive programs and other business arrangements. Employee misconduct could also involve the improper use of information
obtained in the course of clinical trials, which could result in regulatory sanctions and serious harm to our reputation.
Our
board of directors adopted a Code of Ethics in March 2016. However, it is not always possible to identify and deter employee misconduct,
and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks
or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to be in compliance
with such laws or regulations. If any such actions are instituted against us, and we are not successful in defending ourselves
or asserting our rights, those actions could have a significant impact on our business, including the imposition of significant
fines or other sanctions.
In
addition, during the course of our operations, our directors, executives and employees may have access to material, nonpublic
information regarding our business, our results of operations or potential transactions we are considering. If a director, executive
or employee was to be investigated, or an action was to be brought against a director, executive or employee for insider trading,
it could have a negative impact on our reputation and the market price of our securities. Such a claim, with or without merit,
could also result in substantial expenditures of time and money, and divert attention of our management team from other tasks
important to the success of our business.
If
we fail to withhold our position against the Israeli tax authorities in connection with tax withholding, we may be required to
pay additional taxes.
Following a
tax deduction assessment conducted by the Israeli Tax Authorities, or the ITA, in October 2016, we are deemed to be in debt of
approximately $1.45 million of additional withholding taxes
, including penalties and interest
.
It
is our management’s opinion, based on the assessment of our legal counsel, that the chances of the claims of the ITA being
dismissed are more likely than not. Therefore, no allowance regarding this assessment was recorded in our financial statements.
However, if our position is not accepted in the event this case is litigated, our business can be materially adversely affected.
Risks
Related to Our Intellectual Property
If
we are unable to secure and maintain patent or other intellectual property protection for the intellectual property used in our
products, our ability to compete will be harmed.
Our
commercial success depends, in part, on obtaining and maintaining patent and other intellectual property protection for the technologies
used in our products. The patent positions of medical device companies, including ours, can be highly uncertain and involve complex
and evolving legal and factual questions. Furthermore, we might in the future opt to license intellectual property from other
parties. If we, or the other parties from whom we may license intellectual property, fail to obtain and maintain adequate patent
or other intellectual property protection for intellectual property used in our products, or if any protection is reduced or eliminated,
others could use the intellectual property used in our products, resulting in harm to our competitive business position. In addition,
patent and other intellectual property protection may not provide us with a competitive advantage against competitors that devise
ways of making competitive products without infringing any patents that we own or have rights to.
U.S.
patents and patent applications may be subject to interference proceedings, and U.S. patents may be subject to re-examination
proceedings in the U.S. Patent and Trademark Office. Foreign patents may be subject to opposition or comparable proceedings in
the corresponding foreign patent offices. Any of these proceedings could result in loss of the patent or denial of the patent
application, or loss or reduction in the scope of one or more of the claims of the patent or patent application. Changes in either
patent laws or in interpretations of patent laws may also diminish the value of our intellectual property or narrow the scope
of our protection. Interference, re-examination and opposition proceedings may be costly and time consuming, and we, or the other
parties from whom we might potentially license intellectual property, may be unsuccessful in defending against such proceedings.
Thus, any patents that we own or might license may provide limited or no protection against competitors. In addition, our pending
patent applications and those we may file in the future may have claims narrowed during prosecution or may not result in patents
being issued. Even if any of our pending or future applications are issued, they may not provide us with adequate protection or
any competitive advantages. Our ability to develop additional patentable technology is also uncertain.
Non-payment
or delay in payment of patent fees or annuities, whether intentional or unintentional, may also result in the loss of patents
or patent rights important to our business. Many countries, including certain countries in Europe, have compulsory licensing laws
under which a patent owner may be compelled to grant licenses to other parties. In addition, many countries limit the enforceability
of patents against other parties, including government agencies or government contractors. In these countries, the patent owner
may have limited remedies, which could materially diminish the value of the patent. In addition, the laws of some foreign countries
do not protect intellectual property rights to the same extent as do the laws of the United States, particularly in the field
of medical products and procedures.
If we are unable
to prevent unauthorized use or disclosure of our proprietary trade secrets and unpatented know-how, our ability to compete will
be harmed.
Proprietary
trade secrets, copyrights, trademarks and unpatented know-how are also very important to our business. We rely on a combination
of trade secrets, copyrights, trademarks, confidentiality agreements and other contractual provisions and technical security measures
to protect certain aspects of our technology, especially where we do not believe that patent protection is appropriate or obtainable.
We require our office holders, employees, consultants and distributers of our products and most third parties (such as contractors
or clinical collaborators) to execute confidentiality agreements in connection with their relationships with us. However, these
measures may not be adequate to safeguard our proprietary intellectual property and conflicts may, nonetheless, arise regarding
ownership of inventions. Such conflicts may lead to the loss or impairment of our intellectual property or to expensive litigation
to defend our rights against competitors who may be better funded and have superior resources. Our office holders, employees,
consultants and other advisors may unintentionally or willfully disclose our confidential information to competitors. In addition,
confidentiality agreements may be unenforceable or may not provide an adequate remedy in the event of unauthorized disclosure.
Enforcing a claim that a third party illegally obtained and is using our trade secrets is expensive and time consuming, and the
outcome is unpredictable. Moreover, our competitors may independently develop equivalent knowledge, methods and know-how. Unauthorized
parties may also attempt to copy or reverse engineer certain aspects of our products that we consider proprietary. As a result,
other parties may be able to use our proprietary technology or information, and our ability to compete in the market would be
harmed.
We
could become subject to patent and other intellectual property litigation that could be costly, result in the diversion of management’s
attention, require us to pay damages and force us to discontinue selling our products.
Our
industry is characterized by competing intellectual property and a substantial amount of litigation over patent and other intellectual
property rights. Determining whether a product infringes a patent involves complex legal and factual issues, and the outcome of
a patent litigation action is often uncertain. No assurance can be given that patents containing claims covering our products,
parts of our products, technology or methods do not exist, have not been filed or could not be filed or issued. Furthermore, our
competitors or other parties may assert that our products and the methods we employ in the use of our products are covered by
U.S. or foreign patents held by them. In addition, because patent applications can take many years to issue and because publication
schedules for pending applications vary by jurisdiction, there may be applications now pending of which we are unaware and which
may result in issued patents which our current or future products infringe. Also, because the claims of published patent applications
can change between publication and patent grant, there may be published patent applications with claims that we infringe. There
could also be existing patents that one or more of our products or parts may infringe and of which we are unaware. As the number
of competitors in the endoscopic procedure market grows, and as the number of patents issued in this area grows, the possibility
of patent infringement claims against us increases.
Infringement
actions and other intellectual property claims and proceedings brought against or by us, whether with or without merit, may cause
us to incur substantial costs and could place a significant strain on our financial resources, divert the attention of management
from our business and harm our reputation. Some of our competitors may be able to sustain the costs of complex patent or intellectual
property litigation more effectively than we can because they have substantially greater resources
.
We
cannot be certain that we will successfully defend against allegations of infringement of patents and intellectual property rights
of others. In the event that we become subject to a patent infringement or other intellectual property lawsuit and if the other
party’s patents or other intellectual property were upheld as valid and enforceable and we were found to infringe the other
party’s patents or violate the terms of a license to which we are a party, we could be required to pay damages. We could
also be prevented from selling our products unless we could obtain a license to use technology or processes covered by such patents
or will be able to redesign the product to avoid infringement. A license may not be available at all or on commercially reasonable
terms or we may not be able to redesign our products to avoid infringement. Modification of our products or development of new
products could require us to conduct clinical trials and to revise our filings with the applicable regulatory bodies, which would
be time consuming and expensive. In these circumstances, we may be unable to sell our products at competitive prices or at all,
our business and operating results could be harmed.
For example,
on October 28, 2016, we settled all litigation and administrative proceedings with EndoChoice, Inc., or EndoChoice, including
those actions pending in the U.S. District Court for the District of Delaware C.A. Nos. 15-505-LPS-CJB and C.A. No. 15-1215-LPS-CJB
and the trademark opposition proceedings in the State of Israel involving Trademark Application Nos. 257172, 260433 and 262423.
Under the terms of the confidential settlement, we were granted a covenant not to sue with respect to EndoChoice FUSE-related
trademarks and EndoChoice was granted a non-exclusive license to our U.S. Patent No. 6,997,871 and related patents. Each party
has agreed to bear its own costs and fees associated with the litigation
.
We
may be subject to claims that our employees, consultants, or independent contractors have wrongfully used or disclosed confidential
information of third parties or, that our employees have wrongfully used or disclosed alleged trade secrets of their former employers.
Certain
of our employees and personnel were previously employed at universities, medical institutions, or other biotechnology or pharmaceutical
companies. Although we try to ensure that our employees, consultants, and independent contractors do not use the proprietary information
or know-how of others in their work for us, we may be subject to claims that we or our employees, consultants, or independent
contractors have inadvertently or otherwise used or disclosed intellectual property, including trade secrets or other proprietary
information, of any of our employee's former employer or other third parties. Litigation may be necessary to defend against these
claims. If we fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property
rights or personnel. Even if we are successful in defending against such claims, litigation could result in substantial costs
and be a distraction to management and other employees. Furthermore, universities or medical institutions who employ some of our
key employees and personnel in parallel to their engagement by us may claim that intellectual property developed by such person
is owned by the respective academic or medical institution under the respective institution intellectual property policy or applicable
law.
We
may be subject to claims challenging the inventorship or ownership of our patents and other intellectual property.
We
may be subject to claims that former employees, collaborators, or other third parties have an ownership interest in our patents
or other intellectual property. Ownership disputes may arise in the future, for example, from conflicting obligations of consultants
or others who are involved in developing our product candidates. Litigation may be necessary to defend against these and other
claims challenging inventorship or ownership. If we fail in defending any such claims, in addition to paying monetary damages,
we may lose valuable intellectual property rights, such as exclusive ownership of, or right to use, valuable intellectual property.
Such an outcome could have a material adverse effect on our business. Even if we are successful in defending against such claims,
litigation could result in substantial costs and be a distraction to management and other employees.
Risks
Related to Regulatory Compliance
If
we fail to comply with the extensive government regulations relating to our business, we may be subject to fines, injunctions
and other penalties that could harm our business.
Our
medical device products and operations are subject to extensive regulation by the FDA, pursuant to the Federal Food, Drug, and
Cosmetic Act, or FDCA, and various other federal, state and foreign governmental authorities. Government regulations and requirements
specific to medical devices are wide ranging and govern, among other things:
|
|
●
|
design, development and manufacturing;
|
|
|
|
|
|
|
●
|
testing, labeling and storage;
|
|
|
|
|
|
|
●
|
clinical trials;
|
|
|
|
|
|
|
●
|
product safety;
|
|
|
|
|
|
|
●
|
marketing, sales and distribution;
|
|
|
|
|
|
|
●
|
premarket clearance or approval;
|
|
|
|
|
|
|
●
|
record keeping procedures;
|
|
|
|
|
|
|
●
|
advertising and promotions; and
|
|
|
|
|
|
|
●
|
product recalls and field corrective actions.
|
For
the purpose of receiving FDA clearance through the 510(k) track, the applicant must prove, inter alia, that the device subject
to the application is substantially equivalent to one or more products which have already been approved by the FDA (predicate
device). Additionally, the applicant is required to provide a detailed description of the device, including specifications and
technical information, labeling, instructions for use, and the relevant indications for use of the device which is the subject
of the application.
Clinical
trials are usually not required under the 510(k) track, unless the FDA suspects the device subject to application contains new
technical characteristics requiring clinical results regarding safety and efficacy. Clinical trials whose results are attached
to the application for marketing approval are subject to advance approval by the FDA regarding the protocol of the trial of the
Investigative Device Exemption (IDE) type.
Approval
for marketing of medical devices in the United States can be submitted through a PMA, which is required when the device subject
to approval is not substantially equivalent to a previously approved device, particularly high risk life-saving devices.
Though
the PMA track consists of more stringent requirements than the 510(k) track and can be expensive and lengthy and entail significant
fees, unless exempt. The FDA’s 510(k) marketing clearance process usually takes from three to 12 months, but it can last
longer. The process of obtaining PMA approval is more expensive and uncertain than the 510(k) marketing clearance process. It
generally takes from one to three years, or even longer, from the time the PMA application is submitted to the FDA, until an approval
is obtained. There is no assurance that we will be able to obtain FDA clearance or approval for any new products on a timely basis,
or at all.
In
addition, we are subject to annual regulatory audits in order to maintain our quality system certifications, CE mark permissions,
FDA Clearance and Canadian medical device license. We do not know whether we will be able to continue to affix the CE mark for
new or modified products or that we will continue to meet the quality and safety standards required to maintain the permissions
and license we have already received. If we are unable to maintain our quality system certifications and permission to affix the
CE mark to our products, we will no longer be able to sell our products in member countries of the European Union or other areas
of the world that require CE’s or FDA’s approval of medical devices. If we are unable to maintain our quality system
certifications and Canadian medical device license, we will not be able to sell our products in Canada.
Our
medical device products and operations are also subject to regulation by the Medical Devices and Accessories Division in the Israeli
Ministry of Health, or AMAR, which is responsible for the registration of medical devices in Israel, issuance of import licenses
and monitoring marketing of medical equipment. We have received an AMAR approval in Israel.
Failure
to obtain regulatory approval in additional foreign jurisdictions will prevent us from expanding the commercialization of our
products.
To
be able to market and sell our products in most other countries, we must obtain regulatory approvals and comply with the regulations
of those countries. These regulations, including the requirements for approvals and the time required for regulatory review, vary
from country to country. Obtaining and maintaining foreign regulatory approvals are expensive and time consuming, and we
cannot be certain that we will receive regulatory approvals in the various countries in which we plan to market our products.
Failure to obtain or maintain regulatory approval in such countries could have an adverse effect on our financial condition and
results of operations.
Our
products may be subject to product actions in the future that could harm our reputation, business operations and financial results.
The
FDA and similar foreign health or governmental authorities have the authority to require an involuntary recall of commercialized
products in the event of material deficiencies or defects in design, or manufacturing or labeling. In the case of the FDA, the
authority to require a recall must be based on an FDA finding. In addition, foreign governmental bodies have the authority to
require a recall of our products in the event of material deficiencies or defects in design or manufacture. Product actions involving
any of our products would divert managerial and financial resources and have an adverse effect on our financial condition and
results of operations.
If
our products, or malfunction of our products, cause or contribute to adverse medical events such as death or a serious injury,
we are required to report to the FDA, and if we fail to do so, we would be subject to sanctions that would materially harm our
business.
Our
marketed products are subject to Medical Device Reporting, or MDR, obligations, which require that we report to the FDA any incident
in which our products may have caused or contributed to a death or serious injury, or in which our products malfunctioned and,
if the malfunction were to recur, it could likely cause or contribute to a death or serious injury. The timing of our obligation
to report under the MDR regulations is triggered by the date we become aware of the adverse event as well as the nature of the
event. We may fail to report adverse events of which we become aware within the prescribed timeframe. We may also fail to recognize
that we have become aware of a reportable adverse event, especially if it is not reported to us as an adverse event or if it is
an adverse event that is unexpected.
In addition, all manufacturers placing medical devices
in the European Union, Israel and Canada markets are legally bound to report any serious or potentially serious incidents involving
devices they produce or sell to the relevant authority in whose jurisdiction the incident occurred.
If
we fail to comply with our reporting obligations, the FDA or other agencies in whose jurisdiction the incident occurred, could
take action including warning letters, untitled letters, administrative actions, criminal prosecution, imposition of civil monetary
penalties, revocation of our device clearances, seizure of our products, or delay in clearance of future products.
Any
corrective action, whether voluntary or involuntary, as well as defending ourselves in a lawsuit, will require the dedication
of our time and capital, distract management from operating our business, and may materially harm our reputation and financial
results.
We
may be subject to fines, penalties or injunctions if we promote the use of our products for unapproved uses, resulting in damage
to our reputation and business.
Our
promotional materials and training methods must comply with FDA and other applicable laws and regulations, including the prohibition
of the promotion of a medical device for a use that has not been cleared or approved by FDA. Use of a device outside its cleared
or approved indications is known as “off-label” use. We are not allowed to promote the MUSE
TM
for off label
use. If the FDA determines that we promote an off-label use, it could request that we modify our promotional materials or subject
us to regulatory or enforcement actions, which could have an adverse impact on our reputation and financial results. Similarly,
a CE mark and an AMAR approval is invalidated if any part of the device is modified or used in a manner that is outside of its
intended use.
Regulatory reforms may adversely
affect our ability to sell our products profitably.
From
time to time, legislation is drafted and introduced in the United States, European Union or other countries in which we operate,
that could significantly change the statutory provisions governing the clearance or approval, manufacture and marketing of medical
devices. In addition, regulations and guidance may often be revised or reinterpreted by the regulatory authorities in ways that
may significantly affect our business and our products. It is impossible to predict whether legislative changes will be enacted
or interpretations changed, and what the impact of such changes, if any, may be.
On
September 24, 2013, the FDA published a final rule establishing a unique device identification system, or the UDI Rule. The UDI
Rule mandates new labeling requirements that will impact our medical products. We will be required to meet compliance dates as
early as September 24, 2015 for implantable devices (such as staples and cartridges), and additional compliance dates of September
24, 2016 and September 24, 2018 for all other Class II (such as staplers) and reusable components (such as consoles), respectively.
Compliance may involve increases costs and require new equipment, quality systems and manufacturing processes. As of the date
of this prospectus, we are on schedule with the UDI Rule compliance.
If
we fail to comply with federal or state fraud and abuse laws, we could be subject to criminal and civil penalties, loss of licenses
and exclusion from Medicare, Medicaid and other federal and state healthcare programs which could have a material adverse effect
on our business, financial condition and results of operations.
There
are numerous United States federal and state laws pertaining to healthcare fraud and abuse, including anti-kickback laws, false
claims, and physician transparency laws. Section 1128B(b) of the Social Security Act, or the SSA, commonly referred to as the
“Anti-Kickback Statute”, prohibits the offer, payment, solicitation or receipt of any form of remuneration in return
for referring, ordering, leasing, purchasing or arranging for or recommending the ordering, purchasing or leasing of items or
services payable by the Medicare and Medicaid programs or any other federally funded healthcare program. The Anti-Kickback Statute
is very broad in scope, and many of its provisions have not been uniformly or definitively interpreted by courts or regulations.
We have consulting or fee for services arrangements with physicians, hospitals and other entities, which may be subject to scrutiny.
To the extent we are found to not be in compliance, we could face potentially significant fines and penalties in addition to other
more significant sanctions and we may be required to restructure our operations.
Another
development affecting the healthcare industry is the increased use of the federal Civil False Claims Act and, in particular, actions
brought pursuant to the False Claims Act’s “whistleblower” or “qui tam” provisions. The False Claims
Act imposes liability on any person or entity that, among other things, knowingly presents, or causes to be presented, a false
or fraudulent claim for payment by a federal healthcare program. The qui tam provisions of the False Claims Act allow a private
individual to bring actions on behalf of the federal government alleging that the defendant has submitted a false claim to the
federal government, and to share in any monetary recovery. In recent years, the number of suits brought against healthcare providers
by private individuals has increased dramatically. In addition, various states have enacted false claim laws analogous to the
Civil False Claims Act.
The
federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, as amended, created two new federal crimes: healthcare
fraud and false statements relating to healthcare matters. Violations can result in criminal and civil liabilities.
Compliance
with complex foreign and U.S. laws and regulations that apply to our international operations increases our cost of doing business
in international jurisdictions and could expose us or our employees to fines and penalties in the U.S. and abroad. These numerous
and sometimes conflicting laws and regulations include the Foreign Corrupt Practices Act. Many foreign countries have enacted
similar laws addressing fraud and abuse in the healthcare sector. The shifting commercial compliance environment and the need
to build and maintain robust and expandable systems to comply with different compliance requirements in multiple jurisdictions
increases the possibility that a healthcare company may run afoul of one or more of the requirements.
Violations
of any fraud and abuse may result in significant fines, imprisonment and exclusion from the Medicare, Medicaid and other federal
or state healthcare programs which could have a material adverse effect on our business, financial condition and results of operations.
If our operations are found to be in violation of any of these laws or any other governmental regulations that may apply to us,
we may be subject to significant civil, criminal and administrative penalties, damages, fines, exclusion from federal healthcare
programs, such as Medicare and Medicaid, and the curtailment or restructuring of our operations. Dealing with investigations can
be time and resource consuming and can divert management’s attention from the business. In addition, settlements with law
enforcement agencies have forced healthcare providers to agree to additional onerous compliance and reporting requirements as
part of a consent decree or corporate integrity agreement. Any violations of these laws, or any action against us for violation
of these laws, even if we successfully defend against it, could have a material adverse effect on our reputation, business and
financial condition. See “Business - Health Care Laws and Regulations - Fraud and Abuse Laws”.
The new disclosure rules
regarding the use of conflict minerals may affect our relationships with suppliers and customers.
The
Securities and Exchange Commission adopted disclosure rules in August 2012 for companies that use conflict minerals in their products,
with substantial supply chain verification requirements in the event that the materials come from, or could have come from, the
Democratic Republic of the Congo or adjoining countries. These new rules and verification requirements may impose additional costs
on us and on our suppliers, and limit the sources or increase the prices of materials used in our products. Among other things,
this new rule could affect sourcing at competitive prices and availability in sufficient quantities of certain minerals used in
the manufacture of components that are incorporated into our products. In addition, the number of suppliers who provide conflict-free
minerals may be limited, and there may be material costs associated with complying with the disclosure requirements, such as costs
related to the process of determining the source of certain minerals used in our products, as well as costs of possible changes
to products, processes, or sources of supply as a consequence of such verification activities. We may not be able to sufficiently
verify the origins of the relevant minerals used in components manufactured by third parties through the procedures that we implement,
and we may encounter challenges to satisfy those customers who require that all of the components of our products be certified
as conflict-free, which could place us at a competitive disadvantage if we are unable to do so. If we are unable to certify that
our products are conflict free, we may face challenges with our customers, which could place us at a competitive disadvantage,
and our reputation may be harmed.
Risks
Related to Our Operations in Israel
Our
headquarters, manufacturing facilities, and most of our administrative offices are located in Israel and, therefore, our results
may be adversely affected by military instability in Israel.
Our
offices are located in Israel. In addition, the majority of our officers and directors are residents of Israel. Accordingly, geopolitical
or military conditions in Israel and its region may directly or indirectly affect our business. Since the establishment of the
State of Israel in 1948, a number of armed conflicts have taken place between Israel and its neighboring countries. Any hostilities
involving Israel or the interruption or curtailment of trade between Israel and its trading partners could adversely affect our
operations and results of operations. During July and August 2014, Hamas and Israel were engaged in a military conflict that
caused damage and disrupted economic activities in Israel. During November 2012, Hamas and Israel were engaged in an armed
conflict and during the summer of 2006, Israel was engaged in an armed conflict with Hezbollah, a Lebanese Islamist Shiite militia
group and political party. These conflicts involved missile strikes against civilian targets in various parts of Israel, including
areas in which our employees and consultants are located, and negatively affected business conditions in Israel. Any armed conflicts,
terrorist activities or political instability in the region could adversely affect business conditions and could harm our results
of operations and could make it more difficult for us to raise capital. The conflict situation in Israel could cause situations
where medical product certifying or auditing bodies could not be able to visit our manufacturing facilities in order to review
our certifications or clearances, thus possibly leading to temporary suspensions or even cancellations of our clearances or manufacturing
certifications. The conflict situation in Israel could also result in parties with whom we have agreements involving performance
in Israel claiming that they are not obligated to perform their commitments under those agreements pursuant to force majeure provisions
in such agreements. Furthermore, several countries, principally in the Middle East, restrict doing business with Israel and Israeli
companies, and additional countries may impose restrictions on doing business with Israel and Israeli companies if hostilities
in the region continue or intensify. Such restrictions may seriously limit our ability to sell our products to customers in those
countries.
Although
the Israeli government is currently committed to covering the reinstatement value of direct damages that are caused by terrorist
attacks or acts of war, we cannot assure that this government coverage will be maintained, or if maintained, will be sufficient
to compensate us fully for damages incurred. Any losses or damages incurred by us could have a material adverse effect on our
business. Any armed conflicts would likely negatively affect business conditions generally and could harm our results of operations.
Our
operations may be disrupted as a result of the obligation of management or key personnel to perform military service.
Many
of our male employees in Israel are obligated to perform one month, and in some cases more, of annual military reserve duty until
they reach the age of 40 (or older, for officers or reservists with certain occupations) and, in the event of a military conflict,
may be called to active duty. In response to increases in terrorist activity, there have been periods of significant call-ups
of military reservists, and recently some of our employees have been called up in connection with armed conflicts. It is possible
that there will be military reserve duty call-ups in the future. Our operations could be disrupted by the absence of a significant
number of our employees or of one or more of our key employees. Such disruption could materially adversely affect our business,
financial condition and results of operations.
Exchange
rate fluctuations between the foreign currencies and the NIS may negatively affect our earnings.
Our
reporting and functional currency is the U.S. dollar. Our revenues are currently primarily payable in U.S. dollars and Euros and
we expect our future revenues to be denominated primarily in U.S. dollars and Euros. However, certain amount of our expenses are
in NIS and as a result, we are exposed to the currency fluctuation risks relating to the recording of our expenses in U.S. dollars.
We may, in the future, decide to enter into currency hedging transactions. These measures, however, may not adequately protect
us from material adverse effects.
The
government tax benefits that we currently are entitled to receive require us to meet several conditions and may be terminated
or reduced in the future.
Some
of our operations in Israel may entitle us to certain tax benefits under the Law for the Encouragement of Capital Investments,
5719-1959, or the Investments Law, once we begin to produce revenues. From time to time, the government of Israel has considered
reducing or eliminating the tax benefits available to Benefitted Enterprise programs such as ours. If we do not meet the requirements
for maintaining these benefits, they may be reduced or cancelled and the relevant operations would be subject to Israeli corporate
tax at the standard rate, which was set at 25% for 2016
(to
be reduced to 24% in 2017 and 23% in 2018
and thereafter). In addition to being subject
to the standard corporate tax rate, we could be required to refund any tax benefits that we have already received, plus interest
and penalties thereon. Even if we continue to meet the relevant requirements, the tax benefits that our current “Benefitted
Enterprise” is entitled to may not be continued in the future at their current levels, or at all. If these tax benefits
were reduced or eliminated, the amount of taxes that we would have to pay if we produce revenues would likely increase, as all
of our operations would consequently be subject to corporate tax at the standard rate, which could adversely affect our results
of operations. Additionally, if we increase our activities outside of Israel, for example, by way of acquisitions, our increased
activities may not be eligible for inclusion in Israeli tax benefits programs. See “Material Tax Considerations.”
In
the past, we received Israeli government grants for certain of our research and development activities. The terms of those grants
may require us, in addition to payment of royalties, to satisfy specified conditions in order to manufacture products and transfer
technologies outside of Israel. If we fail to comply with the requirements of the Innovation Law (as defined below), we may be
required to pay penalties in addition to repayment of the grants, and may impair our ability to sell our technology outside of
Israel.
Some
of our research and development efforts were financed in part through royalty-bearing grants, in an amount of $0.2 million
that we received from the Israeli National Authority for Technological Innovation of the Israeli Ministry of Economy and Industry
(formerly known as the Office of the Chief Scientist, or the OCS), or NATI. When know-how is developed using OCS grants, the Encouragement
of Research, Development and Technological Innovation in the Industry Law 5744-1984 (formerly known as the Law for the Encouragement
of Research and Development in Industry 5744-1984), or the Innovation Law and the regulations thereunder, restricts our ability
to manufacture products and transfer technology and know-how, developed as a result of OCS funding, outside of Israel.
Under
the Innovation Law and the regulations thereunder, a recipient of OCS grants is required to return the grants by the payment of
royalties of 3% to 6% on the revenues generated from the sale of products (and related services) developed (in whole or in
part) under the OCS program up to the total amount of the grants received from the OCS, linked to the U.S. dollar and bearing
interest at an annual rate of LIBOR applicable to U.S. dollar deposits, as published on the first business day of each calendar
year.
Transfer
of OCS funded know-how and related intellectual property rights outside of Israel, including by way of license for
research
and development
purpose, where the transferring company remains an operating Israeli entity
or where the transferring company ceases to exist as an Israeli entity, requires pre-approval by the OCS and imposes certain conditions,
including, in certain circumstances, requirement of payment of a redemption fee calculated according to the formula provided in
the Innovation Law which takes into account, among others, the consideration for such know-how paid to us in the transaction in
which the technology is transferred, research and development expenses, the amount of OCS support, the time of completion of the
OCS supported research project and other factors, while the redemption fee will not exceed 600% of the grants amount plus interest.
Approval
of the transfer of OCS funded know-how and related intellectual property rights to an Israeli company is required, and may be
granted if the recipient abides by the provisions of Innovation Law, including the restrictions on the transfer of know-how and
the manufacturing rights outside of Israel and the obligation to pay royalties
(note
that there will be an obligation to pay royalties to the OCS from the income of such sale transaction as part of the royalty payment
obligation)
. No assurance can be given that approval to any such transfer, if requested,
will be granted.
In
addition, the products may be manufactured outside Israel by us or by another entity only if prior approval is received from the
OCS (such approval is not required for the transfer of less than 10% of the manufacturing capacity in the aggregate, and in such
event a notice to the OCS is required). As a condition for obtaining approval to manufacture outside Israel, we would be required
to pay increased royalties, which
usually amount
to 1% in addition to the standard rate, up to an increased royalties cap of between 120% and 300% of the grants we received from
the OCS, depending on the manufacturing volume that is performed outside Israel
(less royalties
already paid to the OCS). This restriction may impair our ability to outsource manufacturing rights abroad, however, does not
restrict export of our products that incorporate OCS funded know-how.
A
company also has the option of declaring in its OCS grant application its intention to exercise a portion of the manufacturing
capacity abroad, thus avoiding the need to obtain additional approval
and
pay the increased royalties cap
.
The
restrictions under the Innovation Law (such as with respect to transfer of manufacturing rights abroad or the transfer of OCS
funded know-how and related intellectual property rights abroad) will continue to apply even after we repay the full amount of
royalties payable pursuant to the grants.
These
restrictions may impair our ability to sell our technology assets or to perform or outsource manufacturing outside of Israel,
or otherwise transfer our know-how outside of Israel and require us to obtain the approval of the OCS for certain actions
and transactions and pay additional royalties and other amounts to the OCS. We cannot be certain that any approval of the OCS
will be obtained. Furthermore, in the event that we undertake a transaction involving the transfer to a non-Israeli entity of
technology developed with OCS funding pursuant to a merger or similar transaction, the consideration available to our shareholders
may be reduced by the amounts we are required to pay to the OCS. Any approval, if given, will generally be subject to additional
financial obligations. Failure to comply with the requirements under the Innovation Law may subject us to mandatory repayment
of grants received by us (together with interest and penalties), as well as expose us to criminal proceedings.
The
Innovation Law was amended as of July 29, 2015, or the 2015 Amendment. On January 1, 2016, pursuant to the 2015 Amendment the
National Authority for Technological Innovation, or NATI, was established and in June 2016, NATI was fully constituted. Pursuant
to the 2015 Amendment, NATI is authorized to change the current restrictions imposed on the recipients of grants under the Innovation
Law with a new set of arrangements in connection with ownership obligations of know-how (including with respect to restrictions
on transfer of know-how and manufacturing activities outside of Israel), as well as royalties obligations associated with approved
programs. The Innovation Law as existed prior to the 2015 Amendment will continue to be in effect with respect to
research
and development
programs which were in effect prior to January 1, 2016 until: (i) June 2017,
which is one year following the date of appointment of all members of the NATI council; or (ii) earlier, if otherwise resolved
by the NATI council. The Innovation Law in its form following the 2015 Amendment also includes new provisions with respect to
sanctions imposed for violations of the Innovation Law. As of the date of this
prospectus
,
we are unable to determine whether NATI will promulgate a new set of arrangements or adopt the arrangements which were stipulated
under the Innovation Law as existed prior to the 2015 Amendment, therefore, as of the date of this
prospectus
,
we are unable as assess the effect, if any, of the promulgation of such arrangements on us.
We
were members of an OCS-related consortium, in which certain of our technologies were developed. We are required to provide licenses
to the other members of the consortium to use such technologies for no consideration, which could reduce our profitability.
Certain
of our miniaturized imaging equipment may be based on technological models developed as part of the Bio Medical Photonic Consortium
in the framework of Magnet program of the OCS. The property rights in and to “new information” (as such term is defined
therein) which has been developed by a member of the Consortium, in the framework of a research and development program conducted
as part of the Consortium, belongs solely to the Consortium member that developed it. The developing member is obligated to provide
the other members in the Consortium a non-sublicensable license to use of the “new information” developed by such
member, without consideration, provided that the other members do not transfer such “new information” to any entity
which is not a member of the Consortium, without the consent of such member.
Provisions
of Israeli law and our articles of association may delay, prevent or otherwise impede a merger with, or an acquisition of, our
company, which could prevent a change of control, even when the terms of such a transaction are favorable to us and our shareholders.
Israeli
corporate law regulates mergers, requires tender offers for acquisitions of shares above specified thresholds, requires special
approvals for transactions involving directors, officers or significant shareholders and regulates other matters that may be relevant
to such types of transactions. For example, a merger may not be consummated unless at least 50 days have passed from the
date on which a merger proposal is filed by each merging company with the Israel Registrar of Companies and at least 30 days
have passed from the date on which the shareholders of both merging companies have approved the merger. In addition, a majority
of each class of securities of the target company must approve a merger. Moreover, a tender offer for all of a company’s
issued and outstanding shares can only be completed if the acquirer receives positive responses from the holders of at least 95%
of the issued share capital. Completion of the tender offer also requires approval of a majority of the offerees that do not have
a personal interest in the tender offer, unless, following consummation of the tender offer, the acquirer would hold at least
98% of the Company’s outstanding shares. Furthermore, the shareholders, including those who indicated their acceptance of
the tender offer, may, at any time within six months following the completion of the tender offer, claim that the consideration
for the acquisition of the shares does not reflect their fair market value, and petition an Israeli court to alter the consideration
for the acquisition accordingly, unless the acquirer stipulated in its tender offer that a shareholder that accepts the offer
may not seek such appraisal rights, and the acquirer or the company published all required information with respect to the tender
offer prior to the tender offer’s response date.
Furthermore,
Israeli tax considerations may make potential transactions unappealing to us or to our shareholders whose country of residence
does not have a tax treaty with Israel exempting such shareholders from Israeli tax.
These
and other similar provisions could delay, prevent or impede an acquisition of us or our merger with another company, even if such
an acquisition or merger would be beneficial to us or to our shareholders
.
It
may be difficult to enforce a judgment of a U.S. court against us and our officers and directors and the Israeli experts named
in this prospectus in Israel or the U.S., to assert United States securities laws claims in Israel or to serve process on our
officers and directors and these experts.
We
are incorporated in Israel. Certain of our executive officers and directors reside in Israel and most of our assets and most of
the assets of these persons are located outside of the United States. Therefore, a judgment obtained against us, or any of these
persons in the United States, including one based on the civil liability provisions of the U.S. federal securities laws, may not
be collectible in the United States and may not necessarily be enforced by an Israeli court. It may also be difficult to affect
service of process on these persons in the United States or to assert United States securities law claims in original actions
instituted in Israel.
Even
if an Israeli court agrees to hear such claim, it may determine that Israeli law, and not U.S. law is applicable to the claim.
Under Israeli law, if U.S. law is found to be applicable to such claim, the content of applicable U.S. law must be proven as a
fact by expert witnesses, which can be a time consuming and costly process, and certain matters of procedure would also be governed
by Israeli law. There is little binding case law in Israel that addresses the matters. See “Enforceability of Civil Liabilities”
on page 120 of this prospectus for additional information on your ability to enforce civil claim against us and our executive
officers and directors.
The
rights and responsibilities of a shareholder will be governed by Israeli law which differs in some material respects from the
rights and responsibilities of shareholders of U.S. companies.
The
rights and responsibilities of the holders of our ordinary shares are governed by our articles of association and by Israeli law.
These rights and responsibilities differ in some material respects from the rights and responsibilities of shareholders in typical
U.S.-registered corporations. In particular, a shareholder of an Israeli company has certain duties to act in good faith and fairness
towards the company and other shareholders, and to refrain from abusing its power in the company. There is limited case law available
to assist us in understanding the nature of this duty or the implications of these provisions. These provisions may be interpreted
to impose additional obligations and liabilities on holders of our ordinary shares that are not typically imposed on shareholders
of U.S. corporations.
The
ability of any Israeli company to pay dividends is subject to Israeli law and the amount of cash dividends payable may be subject
to devaluation in the Israeli currency.
The
ability of an Israeli company to pay dividends is governed by Israeli law, which provides that cash dividends may be paid only
out of retained earnings or earnings derived over the two most recent fiscal years, whichever is higher, as determined for statutory
purposes in Israeli currency, provided that there is no reasonable concern that payment of a dividend will prevent a company from
satisfying its existing and foreseeable obligations as they become due. In the event of a devaluation of the Israeli currency
against the U.S. dollar, the amount in U.S. dollars available for payment of cash dividends out of prior years’ earnings
will decrease.
The
termination or reduction of tax and other incentives that the Israeli Government provides to domestic companies may increase the
costs involved in operating a company in Israel.
The
Israeli government currently provides major tax and capital investment incentives to domestic companies, as well as grant and
loan programs relating to research and development and marketing and export activities. In recent years, the Israeli Government
has reduced the benefits available under these programs and the Israeli Governmental authorities have indicated that the government
may in the future further reduce or eliminate the benefits of those programs. We currently take advantage of these programs. There
is no assurance that such benefits and programs would continue to be available in the future to us. If such benefits and programs
were terminated or further reduced, it could have an adverse effect on our business, operating results and financial condition.
We
may become subject to claims for remuneration or royalties for assigned service invention rights by our employees, which could
result in litigation and adversely affect our business.
A
significant portion of our intellectual property has been developed by our employees in the course of their employment for us.
Under the Israeli Patent Law, 5727-1967, or the Patent Law, inventions conceived by an employee in the course and as a result
of or arising from his or her employment with a company are regarded as “service inventions,” which belong to the
employer, absent a specific agreement between the employee and employer giving the employee service invention rights. The Patent
Law also provides that if there is no such agreement between an employer and an employee, the Israeli Compensation and Royalties
Committee, or the Committee, a body constituted under the Patent Law, shall determine whether the employee is entitled to remuneration
for his inventions. Decisions by the Committee (which have been upheld by the Israeli Supreme Court on appeal) have created uncertainty
in this area, as it held that employees may be entitled to remuneration for their service inventions despite having specifically
waived any such rights. However, a later decision by the Committee held that such right can be waived by the employee. The Committee
further held that an explicit reference to the waived right is not necessary in every circumstance in order for the employee’s
waiver of such right to be valid. Such waiver can be formalized in writing or orally or be implied by the actions of the parties
in accordance with the rules of interpretation of Israeli contract law. We generally enter into assignment-of-invention agreements
with our employees pursuant to which such individuals assign to us all rights to any inventions created in the scope of their
employment or engagement with us. Although our employees have agreed to assign to us service invention rights and have specifically
waived their right to receive any special remuneration for such assignment beyond their regular salary and benefits, we may face
claims demanding remuneration in consideration for assigned inventions.
Risks
Related to an Investment in the Securities and this Offering
Our
management team will have immediate and broad discretion over the use of the net proceeds from this offering and may not use them
effectively.
We
currently intend to use the net proceeds of this offering for general corporate purposes, including marketing, production, and
research and development related purposes. See “Use of Proceeds.” However, our management will have broad discretion
in the application of the net proceeds. Our shareholders may not agree with the manner in which our management chooses to allocate
the net proceeds from this offering. The failure by our management to apply these funds effectively could have a material adverse
effect on our business, financial condition and results of operation. Pending their use, we may invest the net proceeds from this
offering in a manner that does not produce income. The decisions made by our management may not result in positive returns on
your investment and you will not have an opportunity to evaluate the economic, financial or other information upon which our management
bases its decisions.
The
offering may not be fully subscribed, and, even if the offering is fully subscribed, we will need additional capital in the future.
If additional capital is not available, we may not be able to continue to operate our business pursuant to our business plan or
we may have to discontinue our operations entirely.
The
placement agent in this offering will offer the securities on a “best-efforts” basis, meaning that we may raise substantially
less than the total maximum offering amount. We will not provide any refund to investors if less than all of the securities are
sold. We have incurred losses in each year since our inception. If we continue to use cash at our historical rates of use we will
need significant additional financing, which we may seek through a combination of private and public equity offerings, debt financings
and collaborations and strategic and licensing arrangements. To the extent that we raise additional capital through the sale of
equity or convertible debt securities, the ownership interest will be diluted, and the terms of any such offerings may include
liquidation or other preferences that may adversely affect the then existing shareholders rights. Debt financing, if available,
would result in increased fixed payment obligations and may involve agreements that include covenants limiting or restricting
our ability to take specific actions such as incurring debt or making capital expenditures. If we raise additional funds through
collaboration, strategic alliance or licensing arrangements with third parties, we may have to relinquish valuable rights to our
technologies, future revenue streams or product candidates, or grant licenses on terms that are not favorable to us.
You
will experience immediate dilution in the net tangible book value of any ADSs you purchase.
Because the effective
price per ADS being offered is substantially higher than our net tangible book value per ADS, you will suffer substantial dilution
in the net tangible book value of any ADSs you purchase in this offering. Therefore, if you purchase ADSs in this offering, you
will suffer immediate and substantial dilution of our pro forma as adjusted net tangible book value.
To
the extent outstanding options, warrants or offered warrants are exercised, you will incur further dilution.
See “Dilution”
on page 43 for a more detailed discussion of the dilution you will incur in connection with this offering.
ADSs
and warrants representing a substantial percentage of our outstanding shares may be sold in this offering, which could cause
the price of ADSs and
ordinary
shares to decline.
Pursuant
to this offering, we may sell up to 929,752 ADSs representing 46,487,600 ordinary shares, or approximately 105%,
of our outstanding ordinary shares as of March 15, 2017. In addition, the investors in this offering will be issued warrants to
purchase up to 1,549,586 ADSs representing 77,479,300 ordinary shares, and the placement agents will receive unregistered
warrants to purchase up to 86,777 ADSs representing 4,338,850 ordinary shares. This sale and any future sales of a substantial
number of ADSs in the public market, or the perception that such sales may occur, could materially adversely affect the price
of the ADSs and ordinary shares. We cannot predict the effect, if any, that market sales of those ADSs or the availability of
those ADSs for sale will have on the market price of the ADSs and ordinary shares.
We
may be a passive foreign investment company, or PFIC, for U.S. federal income tax purposes in 2016 or in any subsequent year.
This may result in adverse U.S. federal income tax consequences for U.S. taxpayers that are holders of our securities.
We will be treated as a PFIC for U.S. federal income tax purposes in any taxable year
in which either (1) at least 75% of our gross income is “passive income” or (2) on average at least 50% of our
assets by value produce passive income or are held for the production of passive income. Passive income for this purpose generally
includes, among other things, certain dividends, interest, royalties, rents and gains from commodities and securities transactions
and from the sale or exchange of property that gives rise to passive income. Passive income also includes amounts derived by reason
of the temporary investment of funds, including those raised in a public offering. In determining whether a non-U.S. corporation
is a PFIC, a proportionate share of the income and assets of each corporation in which it owns, directly or indirectly, at least
a 25% interest (by value) is taken into account. We do not believe we were a PFIC for 2015 or 2016 but there can be no assurance
that we were not a PFIC in 2015 or 2016 and will not be a PFIC in subsequent years, as our operating results for any such years
may cause us to be a PFIC. If we are a PFIC in 2016, or any subsequent year, and a U.S. shareholder does not make an election
to treat us as a “qualified electing fund,” or QEF, or make a “mark-to-market” election, then “excess
distributions” to a U.S. shareholder, and any gain realized on the sale or other disposition of our securities will be subject
to special rules. Under these rules: (1) the excess distribution or gain would be allocated ratably over the U.S. shareholder’s
holding period for the securities; (2) the amount allocated to the current taxable year and any period prior to the first day
of the first taxable year in which we were a PFIC would be taxed as ordinary income; and (3) the amount allocated to each of the
other taxable years would be subject to tax at the highest rate of tax in effect for the applicable class of taxpayer for that
year, and an interest charge for the deemed deferral benefit would be imposed with respect to the resulting tax attributable to
each such other taxable year. In addition, if the IRS determines that we are a PFIC for a year with respect to which we have determined
that we were not a PFIC, it may be too late for a U.S. shareholder to make a timely QEF or mark-to-market election. U.S. shareholders
who hold or have held our securities during a period when we were or are a PFIC will be subject to the foregoing rules, even if
we cease to be a PFIC in subsequent years, subject to exceptions for U.S. shareholders who made a timely QEF or mark-to-market
election. A U.S. shareholder can make a QEF election by completing the relevant portions of and filing IRS Form 8621 in accordance
with the instructions thereto. If applicable, upon request, we will annually furnish U.S. shareholders with information needed
in order to complete IRS Form 8621 (which form would be required to be filed with the IRS on an annual basis by the U.S. shareholder)
and to make and maintain a valid QEF election for any year in which we or any of our subsidiaries are a PFIC.
The
market prices of our securities are subject to fluctuation, which could result in substantial losses by our investors.
The stock market in general and the market
prices of our ordinary shares on the TASE and the ADSs and warrants, if listed, on the NASDAQ, in particular, are or will be subject
to fluctuation, and changes in these prices may be unrelated to our operating performance. We anticipate that the market prices
of our securities will continue to be subject to wide fluctuations. The market price of our securities are, and will
be, subject to a number of factors, including:
|
|
●
|
announcements of technological innovations or new products by us or others;
|
|
|
|
|
|
|
●
|
announcements by us of significant acquisitions, strategic partnerships, in-licensing, out-licensing, joint ventures or capital commitments;
|
|
|
|
|
|
|
●
|
expiration or terminations of licenses, research contracts or other collaboration agreements;
|
|
|
|
|
|
|
●
|
public concern as to the safety of our equipment we sell;
|
|
|
|
|
|
|
●
|
general market conditions;
|
|
|
|
|
|
|
●
|
the volatility of market prices for shares of medical devices companies generally;
|
|
|
|
|
|
|
●
|
developments concerning intellectual property rights or regulatory approvals;
|
|
|
|
|
|
|
●
|
developments concerning standard-of-care in endoscopic procedures;
|
|
|
|
|
|
|
●
|
variations in our and our competitors’ results of operations;
|
|
|
|
|
|
|
●
|
changes in revenues, gross profits and earnings announced by the company;
|
|
|
|
|
|
|
●
|
changes in estimates or recommendations by securities analysts, if our ordinary shares or the ADSs are covered by analysts;
|
|
|
|
|
|
|
●
|
changes in government regulations or patent decisions; and
|
|
|
|
|
|
|
●
|
general market conditions and other factors, including factors unrelated to our operating performance.
|
These factors may materially and
adversely affect the market price of our securities and result in substantial losses by our investors.
We
do not know whether a market for the ADSs will be sustained or what the trading price of the ADSs will be and as a result it may
be difficult for you to sell your ADSs.
Although
the ADSs now trade on NASDAQ, an active trading market for the ADSs may not be sustained. It may be difficult for you to sell
your ADSs without depressing the market price for the ADSs or at all. As a result of these and other factors, you may not be able
to sell your ADSs. Further, an inactive market may also impair our ability to raise capital by selling ADSs and ordinary Shares
and may impair our ability to enter into strategic partnerships or acquire companies or products by using our ordinary shares
as consideration.
There is no public
market for the Series A warrants and Series B warrants being offered by us in this offering.
There is no established public trading market for the Series A warrants and Series
B warrants being offered in this offering, and we do not expect a market to develop. In addition, we do not intend to apply to
list the Series A warrants and Series B warrants on any national securities exchange or other nationally recognized trading system,
including the NASDAQ Capital Market. Without an active market, the liquidity of the Series A warrants and Series B warrants will
be limited.
Future
sales of our securities could reduce their market price.
Substantial
sales of our securities, either on the TASE or on NASDAQ, may cause the market price of our securities to decline. All of our
outstanding ordinary shares are registered and available for sale in Israel. Sales by us or our security holders of substantial
amounts of our securities, or the perception that these sales may occur in the future, could cause a reduction in the market price
of our securities.
The issuance of any additional ordinary shares, ADSs,
warrants or any securities that are exercisable for or convertible into our ordinary shares or ADSs, may have an adverse effect
on the market price of our securities and will have a dilutive effect on our existing shareholders and holders of ADSs.
The
warrants are speculative in nature.
The Series A warrants offered by us in this
offering do not confer any rights of ownership of ordinary shares or ADSs on their holders, such as voting rights or the right
to receive dividends, but only represent the right to acquire ADSs at a fixed price for a limited period of time. Specifically,
commencing on the date of issuance, holders of the Series A warrants may exercise their right to acquire ADSs and pay an assumed
exercise price per share of $ , subject to adjustment upon certain events, prior to years from the date
of issuance, after which date any unexercised warrants will expire and have no further value.
Holders
of the ADSs may not receive the same distributions or dividends as those we make to the holders of our ordinary shares,
and, in some limited circumstances, you may not receive dividends or other distributions on our ordinary shares and you may not
receive any value for them, if it is illegal or impractical to make them available to you.
The
Depositary for the ADSs has agreed to pay to you the cash dividends or other distributions it or the custodian receives on ordinary
shares or other deposited securities underlying the ADSs, after deducting its fees and expenses. You will receive these distributions
in proportion to the number of ordinary shares your ADSs represent. However, the Depositary is not responsible if it decides that
it is unlawful or impractical to make a distribution available to any holders of ADSs. For example, it would be unlawful to make
a distribution to a holder of ADSs if it consists of securities that require registration under the Securities Act of 1933, as
amended, or the Securities Act, but that are not properly registered or distributed under an applicable exemption from registration.
In addition, conversion into U.S. dollars from foreign currency that was part of a dividend made in respect of deposited ordinary
shares may require the approval or license of, or a filing with, any government or agency thereof, which may be unobtainable.
In these cases, the Depositary may determine not to distribute such property and hold it as “deposited securities”
or may seek to effect a substitute dividend or distribution, including net cash proceeds from the sale of the dividends that the
Depositary deems an equitable and practicable substitute. We have no obligation to register under U.S. securities laws any ADSs,
ordinary shares, rights or other securities received through such distributions. We also have no obligation to take any other
action to permit the distribution of ADSs, ordinary shares, rights or anything else to holders of ADSs. In addition, the Depositary
may withhold from such dividends or distributions its fees and an amount on account of taxes or other governmental charges to
the extent the Depositary believes it is required to make such withholding. This means that you may not receive the same distributions
or dividends as those we make to the holders of our ordinary shares, and, in some limited circumstances, you may not receive any
value for such distributions or dividends if it is illegal or impractical for us to make them available to you. These restrictions
may cause a material decline in the value of the ADSs.
Holders
of ADSs must act through the Depositary to exercise their rights as shareholders of our company.
Holders of our ADSs do not have the same
rights of our shareholders and may only exercise the voting rights with respect to the underlying ordinary shares in accordance
with the provisions of the Deposit Agreement. Under Israeli law and our articles of association, the minimum notice period required
to convene a shareholders meeting is no less than 21 or 35 calendar days, depending on the proposals on the agenda for the shareholders
meeting. When a shareholder meeting is convened, holders of ADSs may not receive sufficient notice of a shareholders’ meeting
to permit them to withdraw their ordinary shares to allow them to cast their vote with respect to any specific matter. In addition,
the Depositary and its agents may not be able to send voting instructions to holders of ADSs or carry out their voting instructions
in a timely manner. We will make all reasonable efforts to cause the Depositary to extend voting rights to holders of the ADSs
in a timely manner, but we cannot assure holders that they will receive the voting materials in time to ensure that they can instruct
the Depositary to vote their ordinary shares underlying the ADSs. Furthermore, the Depositary and its agents will not be responsible
for any failure to carry out any instructions to vote, for the manner in which any vote is cast or for the effect of any such
vote. As a result, holders of ADSs may not be able to exercise their right to vote and they may lack recourse if their ordinary
shares underlying the ADSs are not voted as they requested. In addition, in the capacity as a holder of ADSs, they will not be
able to call a shareholders’ meeting.
We
do not intend to pay any cash dividends on our ordinary shares in the foreseeable future and, therefore, any return on your investment
in our securities must come from increases in the value and trading price of our securities.
We
have never declared or paid cash dividends on our securities and do not anticipate that we will pay any cash dividends on our
securities in the foreseeable future, therefore, any return on your investment in our securities must come from increases in the
value and trading price of our securities.
We
intend to retain our earnings to finance the development and expenses of our business. Any future determination relating to our
dividend policy will be at the discretion of our board of directors and will depend on a number of factors, including future earnings,
our financial condition, operating results, contractual restrictions, capital requirements, business prospects, applicable Israeli
law and other factors our board of directors may deem relevant.
We
are an “emerging growth company,” and the reduced disclosure requirements applicable to emerging growth companies
may make our securities less attractive to investors.
We
are an “emerging growth company,” as defined in the Jumpstart Our Business Startups Act of 2012, or the JOBS Act,
and may remain an emerging growth company for up to five years. For so long as we remain an emerging growth company, we are permitted
and intend to rely on exemptions from certain disclosure requirements that are applicable to other public companies that are not
emerging growth companies. These exemptions include not being required to comply with the auditor attestation requirements of
Section 404 of the Sarbanes-Oxley Act of 2002 and not being required to comply with any requirement that may be adopted by the
Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report
providing additional information about the audit and the financial statements. We cannot predict whether investors will find our
securities less attractive if we rely on these exemptions. If some investors find our securities less attractive as a result,
there may be a less active trading market for our securities and the price of our securities may be more volatile.
In
addition, the JOBS Act provides that an emerging growth company can take advantage of an extended transition period for complying
with new or revised accounting standards. This allows an emerging growth company to delay the adoption of certain accounting standards
until those standards would otherwise apply to private companies. We are choosing to “opt out” of this provision and,
as a result, we will comply with new or revised accounting standards as required when they are adopted. This decision to opt out
of the extended transition period is irrevocable.
If
securities or industry analysts do not publish research or publish inaccurate or unfavorable research about our business,
our share price and trading volume could decline.
The trading market for our securities will
depend on the research and reports that securities or industry analysts publish about us or our business. We do not have any control
over these analysts. There can be no assurance that analysts will cover us, or provide favorable coverage. If one or more analysts
downgrade our share or change their opinion of our securities, the price of our securities would likely decline. In addition,
if one or more analysts cease coverage of our company or fail to regularly publish reports on us, we could lose visibility in
the financial markets, which could cause our share price or trading volume to decline.
Our
securities are traded on different markets and this may result in price variations.
Our
ordinary shares have been traded on the TASE since February 2006, the ADSs have been traded on the NASDAQ since August
5, 2015. Trading in these securities on these markets takes place in different currencies (dollars on the NASDAQ and NIS
on the TASE), and at different times (resulting from different time zones, different trading days and different public holidays
in the United States and Israel). The trading prices of these securities on these two markets may differ due to these and other
factors. Any decrease in the price of these securities on one of these markets could cause a decrease in the trading price of
these securities on the other market.
We incur additional
costs as a result of the listing of the ADSs for trading on the NASDAQ, and our management is required to devote substantial time
to new compliance initiatives and reporting requirements.
As a public company in the United States, we
incur significant accounting, legal and other expenses as a result of the listing of the ADSs on the NASDAQ. These include
costs associated with corporate governance requirements of the SEC and the Marketplace Rules of the NASDAQ Stock Market, as well
as requirements under Section 404 and other provisions of the Sarbanes-Oxley Act of 2002, or the Sarbanes-Oxley Act. These rules
and regulations will increase our legal and financial compliance costs, introduced new costs such as investor relations, stock
exchange listing fees and shareholder reporting, and made some activities more time consuming and costly. Any future
changes in the laws and regulations affecting public companies in the United States and Israel, including Section 404 and other
provisions of the Sarbanes-Oxley Act, the rules and regulations adopted by the SEC and the rules of the NASDAQ Stock Market, as
well as compliance with the applicable full Israeli reporting requirements which currently apply to us as a company listed on the
TASE (for so long as they apply to us, pending shareholder approval by special majority of a change to our TASE reporting requirements
to allow us to report to the TASE in the same manner in which we report to the SEC), will result in increased costs to us as we
respond to such changes. These laws, rules and regulations could make it more difficult or more costly for us to obtain certain
types of insurance, including director and officer liability insurance, and we may be forced to accept reduced policy limits and
coverage or incur substantially higher costs to obtain the same or similar coverage. The impact of these requirements could also
make it more difficult for us to attract and retain qualified persons to serve on our board of directors, our board committees
or as executive officers.
As
a foreign private issuer, we are permitted to follow certain home country corporate governance practices instead of
applicable SEC and NASDAQ requirements, which may result in less protection than is accorded to investors under rules applicable
to domestic issuers.
As
a foreign private issuer, we are permitted to follow certain home country corporate governance practices instead of those otherwise
required under the rules of the NASDAQ Stock Market for domestic issuers. For instance, we may follow home country practice in
Israel with regard to: distribution of annual and quarterly reports to shareholders, director independence requirements,
director nomination procedures, approval of compensation of officers, approval of related party transactions, shareholder approval
requirements, equity compensation plans and quorum requirements at shareholders’ meetings. In addition, we follow our home
country law, instead of the rules of the NASDAQ Stock Market, which require that we obtain shareholder approval for certain dilutive
events, such as for the establishment or amendment of certain equity based compensation plans, an issuance that will result in
a change of control of the company, certain transactions other than a public offering involving issuances of a 20% or more interest
in the company and certain acquisitions of the stock or assets of another company. Following our home country governance
practices as opposed to the requirements that would otherwise apply to a U.S. company listed on the NASDAQ Stock Market, may provide
less protection than is accorded to investors under the rules of the NASDAQ Stock Market applicable to domestic issuers.
In
addition, as a foreign private issuer, we are exempt from the rules and regulations under the Securities Exchange Act of 1934,
as amended, or the Exchange Act, related to the furnishing and content of proxy statements, and our officers, directors
and principal shareholders are exempt from the reporting and short-swing profit recovery provisions contained in Section 16 of
the Exchange Act. In addition, we are not required under the Exchange Act to file annual, quarterly and current reports and financial
statements with the SEC as frequently or as promptly as domestic companies whose securities are registered under the Exchange
Act.
We
may lose our foreign private issuer status in the future, which could result in significant additional costs and expenses.
We
are a foreign private issuer, as such term is defined in Rule 405 under the Securities Act, and therefore, we are not required
to comply with all the periodic disclosure and current reporting requirements of the Exchange Act and related rules and regulations.
Under Rule 405, the determination of foreign private issuer status is made annually on the last business day of an issuer’s
most recently completed second fiscal quarter and, accordingly, the next determination will be made with respect to us on June
30, 2017.
In
the future, we would lose our foreign private issuer status if a majority of our shareholders, directors or management are U.S.
citizens or residents and we fail to meet additional requirements necessary to avoid loss of foreign private issuer status. Although
we have elected to comply with certain U.S. regulatory provisions, our loss of foreign private issuer status would make such provisions
mandatory. The regulatory and compliance costs to us under U.S. securities laws as a U.S. domestic issuer may be significantly
higher. If we are not a foreign private issuer, we will be required to file periodic reports and registration statements on U.S.
domestic issuer forms with the U.S. Securities and Exchange Commission, or the SEC, which are more detailed and extensive than
the forms available to a foreign private issuer. For example, the annual report on Form 10-K requires domestic issuers to disclose
executive compensation information on an individual basis with specific disclosure regarding the domestic compensation philosophy,
objectives, annual total compensation (base salary, bonus, equity compensation) and potential payments in connection with change
in control, retirement, death or disability, while the SEC forms applicable to foreign private issuers permit them to disclose
compensation information on an aggregate basis if executive compensation disclosure on an individual basis is not required or
otherwise has not been provided in the issuer’s home jurisdiction. We disclose individual compensation information, but
this disclosure is not as comprehensive as that required of U.S. domestic issuers since we are not required to disclose more detailed
information in Israel. We intend to continue this practice as long as it is permitted under the SEC’s rules and Israel’s
rules do not require more detailed disclosure. We will also have to mandatorily comply with U.S. federal proxy requirements, and
our officers, directors and principal shareholders will become subject to the short-swing profit disclosure and recovery provisions
of Section 16 of the Exchange Act. We may also be required to modify certain of our policies to comply with good governance practices
associated with U.S. domestic issuers. Such conversion and modifications will involve additional costs. In addition, we may lose
our ability to rely upon exemptions from certain corporate governance requirements on U.S. stock exchanges that are available
to foreign private issuers.
If we are unable to satisfy the
requirements of Section 404 of the Sarbanes-Oxley Act of 2002 as they apply to a foreign private issuer that is listing on a U.S.
exchange for the first time, or our internal control over financial reporting is not effective, the reliability of our financial
statements may be questioned and our securities price may suffer.
Section
404 of the Sarbanes-Oxley Act requires a company subject to the reporting requirements of the U.S. securities laws to do a comprehensive evaluation of
its and its subsidiaries’ internal control over financial reporting. When applicable, to comply with this statute, we will
be required to document and test our internal control procedures; our management will be required to assess and issue a report
concerning our internal control over financial reporting. In addition, our independent registered public accounting firm may be
required to issue an opinion on the effectiveness of our internal control over financial reporting at a later date.
The continuous process of strengthening
our internal controls and complying with Section 404 is complicated and time-consuming. Furthermore, as our business continues
to grow both domestically and internationally, our internal controls will become more complex and will require significantly more
resources and attention to ensure our internal controls remain effective overall. During the course of its testing, our management
may identify weaknesses or deficiencies, which may not be remedied in a timely manner. If our management cannot favorably assess
the effectiveness of our internal controls over financial reporting, or if our independent registered public accounting firm identifies
material weaknesses in our internal control, investor confidence in our financial results may weaken, and the market price of
our securities may suffer.
We
may not satisfy NASDAQ’s requirements for continued listing. If we cannot satisfy these requirements, NASDAQ
could delist our securities.
Our
ADSs are listed on the NASDAQ under the symbol “MDGS”. To continue to be listed on NASDAQ, we are required to satisfy
a number of conditions, including a minimum bid price of at least $1.00 per share, a market value of our publicly held shares
of at least $1 million and shareholders’ equity of at least $2.5 million.
On January 12, 2017, we received
a letter from NASDAQ indicating that we are not in compliance with the minimum bid price requirement for continued listing set
forth in Listing Rule 5550(a)(2) which requires listed securities to maintain a minimum bid price of $1.00 per share. This notice
has no immediate effect on the listing or trading of our ADSs on NASDAQ. According to the letter, we have a grace period of 180
calendar days, commencing on January 12, 2017, to regain compliance with the minimum bid price requirement. We can regain
compliance if, at any time before the grace period ends, the bid price of our ADSs closes at or above $1.00 per share for a minimum
of ten consecutive business days. If we cannot demonstrate bid price compliance by the end of the 180 day grace period,
we may become eligible for an additional 180 day grace period if we meet the continued listing requirement for market value of
publicly held shares and all other initial listing standards for The NASDAQ Capital Market, with the exception of the bid price
requirement. On March 15, 2017, we effected a change in the
ratio of ordinary shares per
ADS
from five ordinary shares per ADS to 50 ordinary shares per ADS. The change in the ordinary shares ratio for the ADSs
had the same effect as a 1-for-10 reverse stock split of the ADSs.
If
we are delisted from NASDAQ, trading in our securities may be conducted, if available, on the OTC Markets or, if available, via
another market. In the event of such delisting, our shareholders would likely find it significantly more difficult
to dispose of, or to obtain accurate quotations as to the value of our securities, and our ability to raise future capital through
the sale of our securities could be severely limited. In addition, if our securities were delisted from NASDAQ, our ADSs could
be considered a “penny stock” under the U.S. federal securities laws. Additional regulatory requirements
apply to trading by broker-dealers of penny stocks that could result in the loss of an effective trading market for our securities.
Moreover, if our securities were delisted from NASDAQ, we will no longer be exempt from certain provision of the Israeli Securities
Law, and therefore will have increased disclosure requirements.
Special
Note Regarding Forward-Looking Statements
We
make forward-looking statements in this prospectus that are subject to risks and uncertainties. These forward-looking statements
include information about possible or assumed future results of our business, financial condition, results of operations, liquidity,
plans and objectives. In some cases, you can identify forward-looking statements by terminology such as “believe,”
“may,” “estimate,” “continue,” “anticipate,” “intend,” “should,”
“plan,” “expect,” “predict,” “potential,” or the negative of these terms or other
similar expressions. Forward-looking statements are based on information we have when those statements are made or our management’s
good faith belief as of that time with respect to future events, and are subject to risks and uncertainties that could cause actual
performance or results to differ materially from those expressed in or suggested by the forward-looking statements. Important
factors that could cause such differences include, but are not limited to:
|
|
●
|
the
overall global economic environment;
|
|
|
●
|
insufficient
coverage or reimbursement from medical insurers;
|
|
|
●
|
the
impact of competition and new technologies;
|
|
|
●
|
general
market, political, reimbursement and economic conditions in the countries in which we
operate;
|
|
|
●
|
our
ability to continue as a going concern;
|
|
|
●
|
projected
capital expenditures and liquidity;
|
|
|
●
|
changes
in our strategy;
|
|
|
●
|
government
regulations and approvals;
|
|
|
●
|
changes
in customers’ budgeting priorities;
|
|
|
●
|
litigation
and regulatory proceedings; and
|
You
should review carefully the risks and uncertainties described under the heading “Risk Factors” in this prospectus
for a discussion of these and other risks that relate to our business and investing in our ordinary shares. The forward-looking
statements contained in this prospectus are expressly qualified in their entirety by this cautionary statement. Except as required
by law, we undertake no obligation to update publicly any forward-looking statements after the date of this prospectus to conform
these statements to actual results or to changes in our expectations.
Trademarks,
Service Marks and Trade Names
Solely
for convenience, some of the trademarks, service marks, and trade names referred to in this prospectus are without the ® and
TM
symbols, but such references are not intended to indicate, in any way, that we will not assert, to the fullest extent
under applicable law, our rights or the rights of the applicable licensors to these trademarks, service marks and trade names.
This prospectus contains additional trademarks, service marks and trade names of others, which are the property of their respective
owners. All trademarks, service marks and trade names appearing in this prospectus are, to our knowledge, the property of their
respective owners. We do not intend our use or display of other companies’ trademarks, service marks or trade names to imply
a relationship with, or endorsement or sponsorship of us by, any other companies.
Exchange
Rate Information
The
following table sets forth information regarding the exchange rates of NIS per U.S. dollar for the periods indicated. Average
rates are calculated by using the daily representative rates as reported by the Bank of Israel on the last day of each month during
the periods presented.
|
|
|
NIS
per U.S. dollars
|
|
|
Annual
|
|
High
|
|
|
Low
|
|
|
Average
|
|
|
Period
End
|
|
|
2016
|
|
|
3.983
|
|
|
|
3.746
|
|
|
|
3.841
|
|
|
|
3.845
|
|
|
2015
|
|
|
4.053
|
|
|
|
3.761
|
|
|
|
3.884
|
|
|
|
3.902
|
|
|
2014
|
|
|
3.994
|
|
|
|
3.402
|
|
|
|
3.577
|
|
|
|
3.889
|
|
|
2013
|
|
|
3.791
|
|
|
|
3.471
|
|
|
|
3.609
|
|
|
|
3.471
|
|
|
2012
|
|
|
4.084
|
|
|
|
3.700
|
|
|
|
3.856
|
|
|
|
3.733
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Monthly
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
February 2017
|
|
|
3.768
|
|
|
|
3.659
|
|
|
|
3.729
|
|
|
|
3.659
|
|
|
January 2017
|
|
|
3.860
|
|
|
|
3.769
|
|
|
|
3.818
|
|
|
|
3.769
|
|
|
December 2016
|
|
|
3.867
|
|
|
|
3.787
|
|
|
|
3.829
|
|
|
|
3.845
|
|
|
November 2016
|
|
|
3.876
|
|
|
|
3.799
|
|
|
|
3.843
|
|
|
|
3.839
|
|
|
October 2016
|
|
|
3.856
|
|
|
|
3.778
|
|
|
|
3.822
|
|
|
|
3.849
|
|
|
September 2016
|
|
|
3.786
|
|
|
|
3.746
|
|
|
|
3.766
|
|
|
|
3.758
|
|
|
August 2016
|
|
|
3.829
|
|
|
|
3.754
|
|
|
|
3.795
|
|
|
|
3.786
|
|
On March 15, 2017, the daily representative
rate was $1.00 to NIS 3.658, as reported by the Bank of Israel.
Price
Range of our Ordinary Shares
Our
ordinary shares have been trading on the TASE under the symbol “MDGS” since February 2006.
The
following table sets forth, for the periods indicated, the reported high and low sale prices of our ordinary shares on the TASE
in NIS and U.S. dollars. U.S. dollar per ordinary share amounts are calculated using the U.S. dollar representative rate of exchange
on the date for which the high or low market price is applicable, as reported by the Bank of Israel.
|
|
|
NIS
Price
Per Ordinary Share*
|
|
|
US$
Price
Per Ordinary Share*
|
|
|
|
|
High
|
|
|
Low
|
|
|
High
|
|
|
Low
|
|
|
Annual:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2017
(until March 22, 2017)
|
|
|
0.70
|
|
|
|
0.40
|
|
|
|
0.19
|
|
|
|
0.11
|
|
|
2016
|
|
|
2.23
|
|
|
|
0.47
|
|
|
|
0.58
|
|
|
|
0.12
|
|
|
2015
|
|
|
5.89
|
|
|
|
0.26
|
|
|
|
0.71
|
|
|
|
1.48
|
|
|
2014
|
|
|
6.73
|
|
|
|
1.50
|
|
|
|
1.93
|
|
|
|
0.39
|
|
|
2013
|
|
|
11.30
|
|
|
|
5.61
|
|
|
|
2.99
|
|
|
|
1.59
|
|
|
2012
|
|
|
13.60
|
|
|
|
5.66
|
|
|
|
3.55
|
|
|
|
1.41
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Quarterly:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
First
Quarter 2017 (until March 22, 2017)
|
|
|
0.70
|
|
|
|
0.40
|
|
|
|
0.19
|
|
|
|
0.11
|
|
|
Fourth
Quarter 2016
|
|
|
1.19
|
|
|
|
0.47
|
|
|
|
0.31
|
|
|
|
0.12
|
|
|
Third
Quarter 2016
|
|
|
2.23
|
|
|
|
0.65
|
|
|
|
0.58
|
|
|
|
0.17
|
|
|
Second
Quarter 2016
|
|
|
1.42
|
|
|
|
0.74
|
|
|
|
0.38
|
|
|
|
0.19
|
|
|
First
Quarter 2016
|
|
|
1.87
|
|
|
|
1.26
|
|
|
|
0.48
|
|
|
|
0.32
|
|
|
Fourth
Quarter 2015
|
|
|
3.05
|
|
|
|
1.50
|
|
|
|
0.79
|
|
|
|
0.39
|
|
|
Third
Quarter 2015
|
|
|
4.43
|
|
|
|
2.72
|
|
|
|
1.17
|
|
|
|
0.70
|
|
|
Second
Quarter 2015
|
|
|
5.89
|
|
|
|
3.65
|
|
|
|
1.48
|
|
|
|
0.92
|
|
|
First
Quarter 2015
|
|
|
3.90
|
|
|
|
2.59
|
|
|
|
0.98
|
|
|
|
0.67
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Most
Recent Six Months:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March
2017 (until March 22, 2017)
|
|
|
0.70
|
|
|
|
0.48
|
|
|
|
0.19
|
|
|
|
0.13
|
|
|
February
2017
|
|
|
0.55
|
|
|
|
0.40
|
|
|
|
0.15
|
|
|
|
0.11
|
|
|
January
2017
|
|
|
0.58
|
|
|
|
0.40
|
|
|
|
0.15
|
|
|
|
0.11
|
|
|
December
2016
|
|
|
0.70
|
|
|
|
0.47
|
|
|
|
0.18
|
|
|
|
0.12
|
|
|
November
2016
|
|
|
1.19
|
|
|
|
0.70
|
|
|
|
0.31
|
|
|
|
0.18
|
|
|
October
2016
|
|
|
1.07
|
|
|
|
0.93
|
|
|
|
0.28
|
|
|
|
0.24
|
|
|
September
2016
|
|
|
1.41
|
|
|
|
0.97
|
|
|
|
0.37
|
|
|
|
0.26
|
|
*
price per ordinary share adjusted to reflect the 10:1 reverse share split effected on November 6, 2015.
On March 22, 2017, the last reported
sale price of our ordinary shares on the TASE was NIS 0.48 per share, or approximately $0.13 per share (based on the exchange
rate reported by the Bank of Israel for such date). On March 22, 2017, the exchange rate of the NIS to the dollar was $1.00 =
NIS 3.656, as reported by the Bank of Israel.
Price
Range of our
ADSs
Our
ADSs commenced trading on the NASDAQ under the symbol “MDGS” on August 5, 2015
.
The
following table sets forth, for the periods indicated, the reported high and low sale prices of our ADSs on the Nasdaq Capital
Market in U.S. dollars.
|
|
|
US$
Price Per ADR*
|
|
|
|
|
High
|
|
|
Low
|
|
|
Yearly:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2017
(until March 22, 2017)
|
|
|
10.7
|
|
|
|
4.1
|
|
|
2016
|
|
|
32.5
|
|
|
|
5.23
|
|
|
2015
(commencing August 5, 2015)
|
|
|
51.7
|
|
|
|
25.1
|
|
|
|
|
|
|
|
|
|
|
|
|
Quarterly:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
First
Quarter 2017 (until March 22, 2017)
|
|
|
10.7
|
|
|
|
4.1
|
|
|
Fourth
Quarter 2016
|
|
|
17.8
|
|
|
|
5.23
|
|
|
Third
Quarter 2016
|
|
|
32.5
|
|
|
|
7.63
|
|
|
Second
Quarter 2016
|
|
|
19.0
|
|
|
|
10.1
|
|
|
First
Quarter 2016
|
|
|
26.80
|
|
|
|
15.0
|
|
|
Fourth
Quarter 2015
|
|
|
44.8
|
|
|
|
25.7
|
|
|
Third
Quarter 2015 (commencing August 5, 2015)
|
|
|
51.7
|
|
|
|
35.2
|
|
|
|
|
|
|
|
|
|
|
|
|
Most
Recent Six Months:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
March
2017 (until March 22, 2017)
|
|
|
10.7
|
|
|
|
5.82
|
|
|
February
2017
|
|
|
8.3
|
|
|
|
4.15
|
|
|
January
2017
|
|
|
7.5
|
|
|
|
4.1
|
|
|
December
2016
|
|
|
8.80
|
|
|
|
5.2
|
|
|
November
2016
|
|
|
17.8
|
|
|
|
7.7
|
|
|
October
2016
|
|
|
13.4
|
|
|
|
11.6
|
|
|
September
2016
|
|
|
19.7
|
|
|
|
12.1
|
|
*
price per ADS adjusted to reflect
retroactively
(i)
the 10:1 reverse share split and the change in the ratio of ordinary shares per ADS to five deposited ordinary shares per ADS
effected on November 6, 2015, and (ii)
the
change in the ratio of ordinary shares
per ADS from five deposited ordinary shares per ADS to 50 deposited ordinary shares per ADS effected on March 15, 2017.
On March 22, 2017, the last reported sale
price of our ADS on the NASDAQ Capital Market was $6.05 per share.
Use of Proceeds
We estimate that the net proceeds from this offering
will be approximately $6.5 million, based upon an assumed public offering price of $6.05 per Class A unit, the last reported sales
price of our ADSs on the NASDAQ on March 22, 2017, and of $6.04 per Class B unit, after deducting the estimated placement
agent’s fees and estimated offering expenses payable by us, and excluding the proceeds, if any, from the exercise of the
warrants issued pursuant to this offering.
A $1.00 increase (decrease) in the
assumed public offering price per unit, would increase the net proceeds we receive from this offering by $1.14 million, assuming
that the number of units offered by us, as set forth on the cover page of this prospectus, remains the same and after deducting
estimated placement agent’s fees and estimated offering expenses payable by us.
A 10,000 units increase in the number of
units offered by us together with a concomitant $1.00 increase in the assumed public offering price per unit would increase the
net proceeds we receive from this offering by $1.21 million, after deducting estimated placement agent’s fees and estimated
offering expenses payable by us. Conversely, a 10,000 units decrease in the number of units offered by us together with a concomitant
$1.00 decrease in the assumed public offering price per unit would decrease the net proceeds we receive from this offering by
$1.19 million, after deducting estimated placement agent’s fees and estimated offering expenses payable by us.
The
net proceeds from the sale of our securities will be used for general corporate purposes, including marketing, production and
research and development related purposes. However, we have no present binding commitments or agreements to enter into any acquisitions.
The amounts and timing of our actual expenditures will depend upon numerous factors, including the progress of our development
and commercialization efforts, whether or not we enter into strategic collaborations or partnerships, and our operating costs
and expenditures. Accordingly, our management will have significant flexibility in applying the net proceeds of this offering.
In addition, while we have not entered into any binding agreements or commitments relating to any significant transaction as of
the date of this prospectus, we may use a portion of the net proceeds to pursue acquisitions, joint ventures and other strategic
transactions.
Dividend
Policy
We
have never declared or paid any cash dividends to our shareholders. We currently anticipate that we will retain future earnings
for the development, operation and expansion of our business and do not anticipate declaring or paying any cash dividends for
the foreseeable future.
The
distribution of dividends may also be limited by the Companies Law, which permits the distribution of dividends only out of retained
earnings or earnings derived over the two most recent fiscal years, whichever is higher, provided that there is no reasonable
concern that payment of a dividend will prevent a company from satisfying its existing and foreseeable obligations as they become
due.
Payment
of dividends may be subject to Israeli withholding taxes. See “Material Tax Considerations—Israeli Tax Considerations
and Government Programs” for additional information.
Capitalization
The
following table sets forth our capitalization as of September 30, 2016, on:
|
|
●
|
on
pro forma basis, to give effect to the issuance and sale of 113,917 ADSs representing
5,695,850 ordinary shares in the offering we closed on December 6, 2016;
|
|
|
●
|
on pro-forma
as adjusted basis, to give further effect to the issuance and sale in this offering of 1,239,669 units at the assumed
public offering price of $6.05 per Class A unit, the last reported sales price of our ADSs on the NASDAQ on March 22, 2017,
and of $6.04 per Class B unit, after deducting the estimated placement agent’s fees and estimated offering expenses
payable by us.
|
The
financial data in the following table should be read in conjunction with our unaudited condensed consolidated financial statements
for the period ended September 30, 2016, which are included elsewhere in this prospectus.
|
|
|
As
of September 30, 2016
|
|
|
|
|
Actual
|
|
|
Pro
forma
|
|
|
Pro
forma
as adjusted
|
|
|
|
|
(unaudited,
in thousands, except share
and per share data)
|
|
|
Total
debt
(1)
|
|
|
1,971
|
|
|
|
2,224
|
|
|
|
7,867
|
|
|
Shareholders’
equity:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Ordinary
shares, par value NIS 0.10 per share: 150,000,000 shares authorized (actual, pro forma and pro forma as adjusted); 38,447,034
shares outstanding (actual), 44,142,884 shares outstanding (pro forma) and 90,630,484 shares outstanding (pro forma
as adjusted)
|
|
$
|
1,040
|
|
|
|
1,189
|
|
|
|
2,461
|
|
|
Share premium
|
|
|
53,108
|
|
|
|
53,343
|
|
|
|
53,621
|
|
|
Other
reserves
|
|
|
147
|
|
|
|
180
|
|
|
|
427
|
|
|
Receipts
on account of warrants
|
|
|
1,532
|
|
|
|
1,532
|
|
|
|
1,532
|
|
|
Accumulated
deficit
|
|
|
(51,758
|
)
|
|
|
(51,821
|
)
|
|
|
(52,747
|
)
|
|
Total
shareholders’ equity
|
|
|
4,069
|
|
|
|
4,423
|
|
|
|
5,294
|
|
|
Total
capitalization and indebtedness
|
|
$
|
6,040
|
|
|
|
6,647
|
|
|
|
13,161
|
|
|
(1)
|
Includes
$1.84 million which are classified as current liabilities.
|
A $1.00 increase (decrease) in the
assumed public offering price per unit, would increase (decrease) the pro forma as adjusted amount of the total shareholders’
equity by approximately $208,000, assuming that the number of units offered by us, as set forth on the cover page of this prospectus,
remains the same and after deducting estimated placement agent’s fees and estimated offering expenses payable by us. A 10,000
units increase in the number of units offered by us together with a concomitant $1.00 increase in the assumed public offering
price per unit would increase our pro forma as adjusted total shareholders’ equity by approximately $219,000, after deducting
estimated placement agent’s fees and estimated offering expenses payable by us. Conversely, a 10,000 units decrease in the
number of units offered by us together with a concomitant $1.00 decrease in the assumed public offering price per unit would decrease
our pro forma as adjusted total shareholders’ equity by approximately $216,000, after deducting estimated placement agent’s
fees and estimated offering expenses payable by us.
The preceding table excludes as of
September 30, 2016 (i) 1,773,500 ordinary shares issuable upon the exercise of outstanding options at a weighted average exercise
price of NIS 4.51 per share or $1.20 per share (based on the exchange rate reported by the Bank of Israel on such date), equivalent
to 35,470 ADSs at a weighted average exercise price of $60.1 per ADS, (ii) 8,859,541 ordinary shares issuable upon the exercise
of outstanding warrants at a weighted average exercise price of NIS 6.66 per share or $1.77 per share (based on the exchange rate
reported by the Bank of Israel on such date), equivalent to 177,190 ADSs at a weighted average exercise price of $88.6 per ADS,
(iii) 77,479,300 ordinary shares issuable upon the exercise of Series A warrants and Series B warrants to purchase 1,549,586
ADSs to be issued in this offering, and (iv) 4,338,850 ordinary shares issuable upon the exercise of warrants to purchase 86,777
ADSs, to be issued to the placement agent in connection with the offering.
Dilution
If you invest in our securities in this offering,
your ownership interest will be immediately diluted to the extent of the difference between the public offering price per ADS
and the as adjusted net tangible book value per ADS after this offering.
Our net tangible book value (deficit) as of September 30,
2016, was approximately $4.1 million, or approximately $5.29 per ADS. Net tangible book value per ADS represents the amount of
our total tangible assets less total liabilities divided by the total number of our ordinary shares outstanding as of September
30, 2016, and multiplying such amount by 50 (one ADS represents 50 ordinary shares).
After giving effect to the issuance and sale of 113,917
ADSs representing 5,695,850 ordinary shares in the offering we closed on December 6, 2016, our pro forma net tangible book value
as of September 30, 2016 was $4.4 million, or $5.01 per ADS. Pro forma net tangible book value per ADS represents the
amount of our total tangible assets less total liabilities divided by the total number of our ordinary shares outstanding as of
September 30, 2016 on a pro forma basis, and multiplying such amount by fifty (one ADS represents fifty ordinary shares).
After giving effect to the issuance and sale in this
offering of 1,239,669 units at an assumed public offering price of $6.05 per Class A unit, the last reported sales price
of our ADSs on the NASDAQ on March 22, 2017, and of $6.04 per Class B unit, after deducting the estimated placement agent’s
fees and estimated offering expenses payable by us, our pro forma as adjusted net tangible book value on September 30, 2016, would
have been approximately $5.29 million, or approximately $2.92 per ADS. This represents an immediate dilution in the pro forma
as adjusted net tangible book value of approximately $2.09 per ADS to investors purchasing our ADSs and warrants in this
offering.
The following table illustrates the immediate dilution
to new investors:
|
Assumed
public offering price per Class A unit
|
|
$
|
6.05
|
|
|
|
|
|
|
|
|
Pro forma net
tangible book value per ADS at September 30, 2016
|
|
$
|
5.01
|
|
|
|
|
|
|
|
|
Decrease
in pro forma net tangible book value per ADS attributable to investors purchasing our securities in this offering
|
|
$
|
(2.09
|
)
|
|
|
|
|
|
|
|
Pro
forma as adjusted net tangible book value per ADS as of September 30, 2016, after giving effect to this offering
|
|
$
|
2.92
|
|
|
|
|
|
|
|
|
Dilution
per ADS to investors purchasing our securities in this offering
|
|
$
|
3.13
|
|
The preceding table excludes as of
September 30, 2016 (i) 1,773,500 ordinary shares issuable upon the exercise of outstanding options at a weighted average exercise
price of NIS 4.51 per share or $1.20 per share (based on the exchange rate reported by the Bank of Israel on such date), equivalent
to 35,470 ADSs at a weighted average exercise price of $60.05 per ADS, (ii) 8,859,541 ordinary shares issuable upon the exercise
of outstanding warrants at a weighted average exercise price of NIS 6.66 per share or $1.77 per share (based on the exchange rate
reported by the Bank of Israel on such date), equivalent to 177,190 ADSs at a weighted average exercise price of $88.58 per ADS,
(iii) 77,479,300 ordinary shares issuable upon the exercise of Series A warrants and Series B warrants to purchase 1,549,586 ADSs
to be issued in this offering, and (iv) 4,338,850 ordinary shares issuable upon the exercise of warrants to purchase 86,777 ADSs,
to be issued to the placement agent in connection with the offering.
To
the extent outstanding options or warrants or offered warrants are exercised, you will incur further dilution.
A $1.00 increase in the assumed public
offering price per unit would increase our pro forma net tangible book value per ADS after this offering by approximately $0.11
and the dilution per ADS to new investors by approximately $0.89, assuming the number of units offered by us, as set forth on
the cover page of this prospectus, remains the same, after deducting the estimated placement agent’s fees and estimated
offering expenses payable by us. A $1.00 decrease in the assumed public offering price per unit would decrease our pro forma net
tangible book value per ADS after this offering by approximately $0.11 and the dilution per ADS to new investors by approximately
$0.89, assuming the number of units offered by us, as set forth on the cover page of this prospectus, remains the same, after
deducting the estimated placement agent’s fees and estimated offering expenses payable by us. We may also increase or decrease
the number of units we are offering. A 10,000 units increase in the number of units offered by us together with a concomitant
$1.00 increase in the assumed public offering price per unit would increase our pro forma net tangible book value per ADS after
this offering by approximately $0.11 and the dilution per ADS to new investors by approximately $0.89, after deducting estimated
placement agent’s fees and estimated offering expenses payable by us. Conversely, a 10,000 units decrease in the number
of units offered by us together with a concomitant $1.00 decrease in the assumed aggregate public offering price per unit would
decrease our pro forma net tangible book value per ADS after this offering by approximately $0.11 and the dilution per ADS to
new investors by approximately $0.89, after deducting estimated placement agent’s fees and estimated offering expenses payable
by us.
Selected
Financial Data
We
derived the selected financial statement data for the years ended December 31, 2013, 2014 and 2015 set forth below from our
audited consolidated financial statements and related notes included elsewhere in this prospectus. We derived the selected financial
statement data for the nine months ended September 30, 2015 and 2016 from our unaudited condensed interim financial statements
and related notes included elsewhere in this prospectus. Our results for interim periods are not necessarily indicative of the
results that may be expected for the entire year. You should read the information presented below together with our financial
statements, the notes to those statements and the other financial information included elsewhere in this prospectus.
The
December 31, 2015 consolidated financial statements included in this registration statement are presented in New Israeli Shekels,
which was the Company’s functional and presentation currency as of such date. Effective January 1, 2016, the Company changed
its functional currency to the U.S. dollar from the New Israeli Shekel. The December 31, 2015 financial data presented in this
prospectus was translated from NIS to USD as follows: (1) all assets and liabilities of the Company were translated using the
dollar exchange rate as of December 31 2015; (2) equity items were translated using historical exchange rates at the relevant
transaction dates; (3) the statement of comprehensive loss items has been translated at the average exchange rates for the year
2015; and (4) the resulting translation differences have been reported as “currency translation differences” within
other comprehensive loss.
|
|
|
Year Ended December 31,
|
|
|
Nine Months ended
September 30,
|
|
|
|
|
2013
|
|
|
2014
|
|
|
2015
|
|
|
2015
|
|
|
2016
|
|
|
|
|
|
|
|
(unaudited)
|
|
|
|
|
(U.S. Dollars, in thousands, except per share and
weighted average shares data)
|
|
|
Consolidated Statements of Loss and Other Comprehensive Loss
|
|
|
|
|
Revenues
|
|
$
|
691
|
|
|
$
|
744
|
|
|
$
|
624
|
|
|
$
|
399
|
|
|
$
|
496
|
|
|
Cost of revenues
|
|
|
311
|
|
|
|
351
|
|
|
|
277
|
|
|
|
169
|
|
|
|
154
|
|
|
Gross profit
|
|
|
380
|
|
|
|
393
|
|
|
|
347
|
|
|
|
230
|
|
|
|
342
|
|
|
Operating Expenses
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Research and development expenses, net
|
|
|
2,275
|
|
|
|
4,025
|
|
|
|
4,384
|
|
|
|
3,043
|
|
|
|
3,021
|
|
|
Selling and marketing expenses
|
|
|
900
|
|
|
|
2,341
|
|
|
|
2,680
|
|
|
|
1,943
|
|
|
|
1,845
|
|
|
General and administrative expenses
|
|
|
1,908
|
|
|
|
2,280
|
|
|
|
2,842
|
|
|
|
1,911
|
|
|
|
3,016
|
|
|
Other income, net
|
|
|
181
|
|
|
|
269
|
|
|
|
3
|
|
|
|
3
|
|
|
|
|
|
|
Total operating expenses
|
|
|
4,902
|
|
|
|
8,377
|
|
|
|
9,903
|
|
|
|
6,894
|
|
|
|
7,882
|
|
|
Operating loss
|
|
|
(4,522
|
)
|
|
|
(7,984
|
)
|
|
|
(9,556
|
)
|
|
|
(6,664
|
)
|
|
|
(7,540
|
)
|
|
Profit from changes in fair value of warrants issued to investors
|
|
|
3,228
|
|
|
|
980
|
|
|
|
106
|
|
|
|
15
|
|
|
|
17
|
|
|
Financial income (expenses), net
|
|
|
(111
|
)
|
|
|
650
|
|
|
|
(14
|
)
|
|
|
28
|
|
|
|
118
|
|
|
Loss before taxes on income
|
|
|
(1,405
|
)
|
|
|
(6,354
|
)
|
|
|
(9,464
|
)
|
|
|
(6,621
|
)
|
|
|
(7,405
|
)
|
|
Taxes on income
|
|
|
(23
|
)
|
|
|
(4
|
)
|
|
|
(68
|
)
|
|
|
(50
|
)
|
|
|
(24
|
)
|
|
Loss for the period
|
|
$
|
(1,428
|
)
|
|
$
|
(6,358
|
)
|
|
$
|
(9,532
|
)
|
|
$
|
(6,671
|
)
|
|
$
|
(7,429
|
)
|
|
Other comprehensive income (loss) for the period, net of tax
|
|
|
408
|
|
|
|
(1,573
|
)
|
|
|
(211
|
)
|
|
|
(298
|
)
|
|
|
|
|
|
Total comprehensive loss for the period
|
|
$
|
(1,020
|
)
|
|
$
|
(7,931
|
)
|
|
$
|
(9,743
|
)
|
|
$
|
(6,969
|
)
|
|
$
|
(7,429
|
)
|
|
Basic and diluted loss per share
|
|
$
|
(0.11
|
)
|
|
$
|
(0.33
|
)
|
|
$
|
(0.34
|
)
|
|
$
|
(0.25
|
)
|
|
$
|
(0.22
|
)
|
|
Weighted average of ordinary shares (in thousands)
|
|
|
13,020
|
|
|
|
19,500
|
|
|
|
28,415
|
|
|
|
27,190
|
|
|
|
33,369
|
|
|
(1)
|
Basic
loss per ordinary share and diluted loss per ordinary share are the same because outstanding
options and warrants would be anti-dilutive due to our net losses in these periods.
|
|
|
|
As
of September 30, 2016
|
|
|
|
|
Actual
|
|
|
Pro
forma
(1)
|
|
|
Pro
forma as
adjusted
(2)(3)
|
|
|
|
|
(in
thousands)
|
|
|
Consolidated
Balance Sheet Data:
|
|
|
|
|
|
|
|
|
|
|
Cash
and cash equivalents
|
|
$
|
3,868
|
|
|
$
|
4,475
|
|
|
$
|
10,989
|
|
|
Working
capital
(4)
|
|
|
3,155
|
|
|
|
3,762
|
|
|
|
10,276
|
|
|
Total
assets
|
|
|
6,040
|
|
|
|
6,647
|
|
|
|
13,161
|
|
|
Total
long-term liabilities
|
|
|
133
|
|
|
|
386
|
|
|
|
6,029
|
|
|
Total
shareholders’ equity
|
|
|
4,069
|
|
|
|
4,423
|
|
|
|
5,294
|
|
|
(1)
|
Pro forma gives
effect to the issuance and sale of 113,917 ADSs representing 5,695,850 ordinary shares in the offering we closed on
December 6, 2016.
|
|
(2)
|
Pro
forma as adjusted gives further effect to the issuance and sale in this offering of an aggregate of 1,239,669 units
at the assumed public offering price of $6.05 per Class A unit, the last reported sales price of our ADSs on the NASDAQ on
March 22, 2017, and of $6.04 per Class B unit, after deducting the estimated placement agent’s fees and estimated offering
expenses payable by us.
|
|
(3)
|
A
$1.00 increase (decrease) in the assumed public offering price per unit, would increase (decrease) the as pro forma adjusted
amount of each of cash and cash equivalents by approximately $1.14 million and total shareholders’ equity by approximately
$208,000, assuming that the number of units offered by us, as set forth on the cover page of this prospectus, remains the
same and after deducting estimated placement agent’s fees and estimated offering expenses payable by us. A 10,000 units
increase in the number of units offered by us together with a concomitant $1.00 increase in the assumed public offering price
per unit would increase our pro forma as adjusted cash and cash equivalents by approximately $1.21 million and total shareholders’
equity by approximately $219,000, after deducting estimated placement agent’s fees and estimated offering expenses
payable by us. Conversely, a 10,000 unit decrease in the number of units offered by us together with a concomitant $1.00 decrease
in the assumed public offering price per unit would decrease our pro forma as adjusted cash and cash equivalents by approximately
$1.19 million and total shareholders’ equity by approximately $216,000, after deducting estimated placement agent’s
fees and estimated offering expenses payable by us.
|
|
(4)
|
Working capital is defined as total current assets minus total current liabilities.
|
Management’s
Discussion and Analysis of Financial Condition
and
Results of Operations
The
following discussion and analysis should be read in conjunction with our financial statements and related notes included elsewhere
in this prospectus. This discussion and other parts of the prospectus contain forward-looking statements based upon current expectations
that involve risks and uncertainties. Our actual results and the timing of selected events could differ materially from those
anticipated in these forward-looking statements as a result of several factors, including those set forth under “Risk Factors”
and elsewhere in this prospectus. See
“
Special Note Regarding Forward-Looking Statements.”
Overview
We
are a medical device company specializing in developing innovative endoscopic procedures and devices. We are a developer of a
unique proprietary endoscopic system for the treatment of GERD, a common chronic disease. We have an advanced technology platform
for performing a wide range of endoscopic procedures. The platform includes various types of rigid, semi-flexible and flexible
video endoscopes, as well as respective endoscopy suites.
From
our date of incorporation, we have invested the majority of our research and development efforts on the MUSE
TM
system, intended
for the minimally invasive and endoscopic treatment of GERD, one of the most widespread chronic diseases in the western world.
The MUSE
TM
system has also been given marketing approval by the FDA through the 510(k) track and bears a CE mark, as required
in order to market the system in European Union countries. In addition, we have an approval from Health Canada and AMAR for marketing
in Canada and Israel, respectively, and we are also in the process of obtaining regulatory approvals in China.
For
the nine months ended September 30, 2016 we derived approximately $0.4 million of revenues from miniature camera and related equipment
(82%), and $91,000 of revenues from MUSE
TM
and related equipment (18%). For the year ended December 31, 2015 we derived
approximately $0.56 million of revenues from miniature camera and related equipment (which represents approximately 89% of our
revenue), and $66,000 from MUSE
TM
and related equipment (which represents approximately 11% of our revenue). For the
year ended December 31, 2014, we derived approximately $0.65 million of revenues from miniature camera and related equipment (88%),
and $91,000 of revenues from MUSE
TM
and related equipment (12%).
While
at present substantially all of our revenue is derived from the miniature camera and related equipment, our strategy is focused
on the development and promotion of our MUSE
TM
System.
We
have incurred net losses in all years since our inception, and, as of September 30, 2016, we had an accumulated deficit of approximately
$52 million.
We anticipate that we are likely to continue to incur significant net losses for at least the next several
years as we continue development of the MUSE
TM
system and potentially other products, expand our sales and marketing capabilities
in the endoscopy-based products market, continue the commercialization of our MUSE
TM
system and work to expand its adoption
and clinical implementation, and continue to develop the corporate infrastructure required to sell and market our products.
Recent
business events and key milestones in the development of our business, include the following:
|
|
●
|
In
December 2016, pursuant to a shelf prospectus in the United States, we raised approximately
$0.76 million through the issuance of ADSs and warrants. See “Certain Relationships
and Related Party Transactions – Material Contracts.”
|
|
|
●
|
In
November 2016, we entered into an exclusive distribution agreement with Innovamedica
S.p.A pursuant to which Innovamedica S.p.A will act as our exclusive distributor in Italy
for a period of four years commencing as of January 1, 2017. See “Business.”
|
|
|
●
|
In
September 2016, pursuant to a shelf prospectus in the United States, we raised approximately
$1.47 million through the issuance of ADSs. See “Certain Relationships and Related
Party Transactions – Material Contracts.”
|
Critical
Accounting Policies and Significant Judgments and Estimates
The
preparation of financial statements in conformity with IFRS requires management to make judgments, estimates and assumptions that
affect the application of accounting policies and the reported amounts of assets, liabilities, income and expenses. Actual results
may differ from these estimates.
The
accounting estimates used in the preparation of our financial statements require management to make assumptions regarding circumstances
and events that involve considerable uncertainty. Management prepares the estimates on the basis of past experience, various facts,
external circumstances, and reasonable assumptions according to the pertinent circumstances of each estimate.
Estimates
and underlying assumptions are reviewed on an ongoing basis. Revisions to accounting estimates are recognized in the period in
which the estimates are revised and in any affected future periods.
Our
significant accounting policies are more fully described in Note 2 to our consolidated financial statements as of December 31,
2015. However, certain of our accounting policies are particularly important to the description of our financial position and
results of operations. In applying these critical accounting policies, our management uses its judgment to determine the appropriate
assumptions to be used in making certain estimates. Those estimates are based on our historical experience, the terms of existing
contracts, our observation of trends in the industry, information provided by our customers and information available from other
outside sources, as appropriate. These estimates are subject to an inherent degree of uncertainty. Our critical accounting policies
include:
Functional
Currency
The
December 31, 2015 consolidated financial statements included in this registration statement are presented in New Israeli Shekels,
which was the Company’s functional and presentation currency as of such date. Effective January 1, 2016, the Company changed
its functional currency to the U.S. dollar from the New Israeli Shekel. The December 31, 2015 financial data presented in this
prospectus was translated from NIS to USD as follows: (1) all assets and liabilities of the Company were translated using the
dollar exchange rate as of December 31 2015; (2) equity items were translated using historical exchange rates at the relevant
transaction dates; (3) the statement of comprehensive loss items has been translated at the average exchange rates for the year
2015; and (4) the resulting translation differences have been reported as “currency translation differences” within
other comprehensive loss.
Revenue
Recognition
Our
revenue in recent years is primarily derived from the sale of imaging equipment which we develop and manufacture. We also derive
revenue from the sale of the MUSE
TM
system. Our revenues from the imaging equipment originate from medical
device companies and research institutions which are engaged in the research and development of medical products in the United
States, Japan and Europe. However, these video cameras are also sold to other customers.
We
recognize revenues in accordance with International Accounting Standard No. 18, or IAS 18. Under IAS 18, revenues from the sale
of goods are recognized when all of the following criteria have been met as of the applicable balance sheet date:
|
|
●
|
The
significant risks and rewards that are derived from the ownership of the goods have been
transferred to the purchaser;
|
|
|
●
|
We
do not retain continuing managerial involvement at a level that generally typifies ownership
and we do not retain effective control over the goods being sold;
|
|
|
●
|
The
amount of the revenues can be measured reliably;
|
|
|
●
|
It
is expected that the economic benefits that are connected to the transaction will flow
to us; and
|
|
|
●
|
The
costs that have been incurred or that will be incurred in respect of the transaction
can be measured reliably.
|
When
multiple-element arrangements exist, the amount of revenue allocated to each element is based upon the relative fair values of
the various elements. The fair values of each element are determined based on the current market price of each of the elements
when sold separately.
We
also recognize revenues in accordance with International Accounting Standard No. 11, or IAS 11.
A
construction contract is defined by IAS 11, “Construction contracts”, as a contract specifically negotiated for the
construction of an asset.
When
the outcome of a construction contract can be estimated reliably and it is probable that the contract will be profitable, contract
revenue is recognized over the period of the contract by reference to the stage of completion. Contract costs are recognized as
expenses by reference to the stage of completion of the contract activity at the end of the reporting period. When it is probable
that total contract costs will exceed total contract revenue, we recognize the expected loss as an expense immediately.
When
the outcome of a construction contract cannot be estimated reliably, then we recognize revenue only to the extent of contract
costs incurred that it is probable will be recoverable.
Variations
in contract work, claims and incentive payments are included in contract revenue to the extent that may have been agreed with
the customer and are capable of being reliably measured.
We
use the “percentage-of-completion method” to determine the appropriate amount to recognize in a given period. The
stage of completion is measured by reference to the contract costs incurred up to the end of the reporting period as a percentage
of total estimated costs for each contract. Costs incurred in the year in connection with the future activities on a contract
are excluded from contract costs in determining the stage of completion.
On
the balance sheet, we report the net contract position for each contract as either an asset or a liability. A contract represents
an asset where costs incurred plus recognized profits (less recognized losses) exceed progress billings; a contract represents
a liability when the opposite in the case.
Share-
Based Compensation
We
account for share-based compensation arrangements in accordance with the provisions of IFRS2. IFRS2 requires us to recognize share-based
compensation expense for awards of equity instruments based on the grant-date fair value of those awards. The cost is recognized
as compensation expense, based upon the grant-date fair value of the equity or liability instruments issued. The fair value of
our option grants is computed as of the grant date based on the Black-Scholes model, using standard parameters established in
that model. The value of the transactions, measured as described above, is recognized as an expense over the vesting period.
Government
Participation in Research and Development Expenses
We
received research and development grants from the State of Israel through the OCS. In accordance with the OCS programs, we were
entitled to a specific grant with respect to a development project only after we incurred development costs related to the project.
Such grants are accounted for as forgivable loans according to International Accounting Standards No. 20, “Accounting for
Government Grants and Disclosure of Government Assistance,” or IAS 20, since they are repayable only if we generate revenues
related to the underlying project.
In
accordance with IAS 20, we account for grants received from the OCS as a liability according to their fair value on the date of
their receipt, unless on that date it is reasonably certain that the amount we received will not be refunded, in which case the
grants are carried to income as a reduction of the research and development expenses.
Upon
the initiation of any project for which we have received a grant, we consider if it is reasonably certain that the project will
reach the revenue-generating stage during the entire development phase of the project when determining the accounting treatment
of the related grant. Our determination is based on various factors including our past experience. We reexamine the liability
to the OCS each reporting period by reviewing the estimate of our future payments to the OCS based on our future sales forecasts.
Capitalization of Development Costs
We
capitalize development expenditure in accordance with International Accounting Standard No. 38, or IAS 38, only if development
costs can be measured reliably, the product or process is technically and commercially feasible, future economic benefits are
probable and we intend to and have sufficient resources to complete development and to use or sell the asset.
We
capitalize development costs based on our judgment regarding technological and economic feasibility, which generally exists when
a product development project reaches a defined milestone, or when entering into a transaction to sell the know-how that was derived
from the development.
Accounting
for Income Taxes
As
part of the process of preparing our consolidated financial statements we are required to estimate our income taxes in each of
the jurisdictions in which we operate. This process requires us to estimate our actual current tax exposures and make an assessment
of temporary differences resulting from differing treatment of items for tax and accounting purposes. These differences result
in deferred tax assets and liabilities, which are included within our consolidated balance sheet. Significant management judgment
is required in determining our provision for income taxes, deferred tax assets and liabilities. Changes to these estimates may
result in a significant increase or decrease to our tax provision in the current or subsequent period.
We
recognize deferred tax assets for unused tax losses, tax benefits, and deductible temporary differences to the extent that it
is probable that future taxable profits will be available against which that can be utilized. Deferred tax assets are reviewed
at each reporting date and are reduced to the extent that it is no longer probable that the related tax benefit will be realized.
The
calculation of our tax liabilities or reduction in deferred tax asset involves dealing with uncertainties in the application of
complex tax regulations and estimates of future taxable income in different geographical jurisdictions. We recognize liabilities
for uncertain tax positions if it is probable to be realized. It is inherently difficult and subjective to estimate such amounts,
as we have to determine the probability of various possible outcomes. We reevaluate these uncertain tax positions on a quarterly
basis. This evaluation is based on factors including, but not limited to, changes in facts or circumstances, changes in tax law,
effective settlement of audit issues, and new audit activity. Such a change in recognition or measurement would result in the
recognition of a tax benefit or an additional charge to the tax provision.
Warrants
On December 6, 2016, we issued warrants to various
institutional investors to purchase up to an aggregate of 39,871 ADSs representing 1,993,550 ordinary shares, with an exercise
price of $9.00 per ADS. The warrants will be exercisable beginning on the six month anniversary of their issuance date and have
a term of five and a half years. The warrants can also be exercised using a cashless exercise mechanism, in which the number of
shares issued would be decreased in accordance with the reduced cash realization price. The warrant exercise price is adjustable
upon certain events (e.g. dividend, distribution of bonus shares, etc.).
On December 6, 2016, we issued to Rodman & Renshaw,
a unit of H.C. Wainwright & Co., acting as placement agent in our offering closed of such date, warrants to purchase up to
an aggregate of 3,987
ADSs representing 199,355 ordinary shares, with an exercise
price of $7.37 per ADS. The warrants will be exercisable beginning on the six month anniversary of their issuance date and have
a term of five years. The warrants can also be exercised by a cashless exercise mechanism, in which the number of shares issued
would be decreased in accordance with the reduced cash realization price. The warrant exercise price is adjustable upon certain
events (e.g. dividend, distribution of bonus shares, etc.).
On December 6, 2016, we issued to Roth Capital Partners
and Maxim Group LLC warrants to purchase up to
an aggregate of 1,993
ADSs representing
99,675 ordinary shares, with an exercise price of $9.00 per ADS. The warrants will be exercisable beginning on the six month anniversary
of their issuance date and have a term of five and a half years. The warrants can also be exercised by a cashless exercise mechanism,
in which the number of shares issued would be decreased in accordance with the reduced cash realization price. The warrant exercise
price is adjustable upon certain events (e.g. dividend, distribution of bonus shares, etc.).
On September 12, 2016, we issued to Roth Capital
Partners and Maxim Group LLC, acting as co-placement agents in our offering closed on such date, warrants to purchase up to an
aggregate of 3,955 ADSs representing 197,750 ordinary shares, with an exercise price of $14.375 per ADS. The warrants have a term
of five years. The warrants can also
be exercised by a cashless
exercise mechanism,
in which the number of shares issued would be decreased in accordance with the reduced cash realization price. The warrant exercise
price is adjustable upon certain events (e.g. dividend, distribution of bonus shares, etc.).
On August 21, 2014 and August 26, 2014, the Company allotted in a private placement warrants to purchase
up to an aggregate of 3,410,861 ordinary shares, with an exercise price of NIS 6.27 per share. The warrants are exercisable during
the 36 months following their issuance. 21,847,610 of the warrants which were issued may, under certain circumstances, also be
exercised via a cashless exercise mechanism. In addition, the number of warrants outstanding will be adjusted to certain events
specified in the warrant agreement (such as: dividends, distribution of bonus shares, etc.)
In
accordance with International Accounting Standard 32: “Financial Instruments: Presentation,” these warrants are a
“financial liability”, classified in the statement of financial position as a non-current liability on the line “warrants
at fair value”. As the aforementioned liability is a non-equity derivative financial instrument, it is classified in accordance
with IAS 39 to the category of financial liability at fair value through the statement of income, which is measured at its fair
value at each date of the statement of financial position, with changes in the fair value currently reflected in the statement
of income.
In
calculating the value of warrants a Black & Scholes model was used. The expected volatility is based on fluctuations in the
price of the Company’s share.
Comparison
of the Nine Months ended September 30, 2016 and September 30, 2015
Revenues
The
following tables present our total revenues, in thousands of U.S. dollars, by geographic area and by products for the fiscal years
indicated:
|
|
|
For the Nine Months Ended September 30,
|
|
|
|
|
2015
|
|
|
2016
|
|
|
United States
|
|
|
277
|
|
|
|
70
|
%
|
|
|
337
|
|
|
|
68
|
%
|
|
Europe
|
|
|
37
|
|
|
|
9
|
%
|
|
|
10
|
|
|
|
2
|
%
|
|
Asia
|
|
|
47
|
|
|
|
12
|
%
|
|
|
5
|
|
|
|
1
|
%
|
|
Other
|
|
|
38
|
|
|
|
9
|
%
|
|
|
144
|
|
|
|
29
|
%
|
|
Total
|
|
|
399
|
|
|
|
100
|
%
|
|
|
496
|
|
|
|
100
|
%
|
|
|
|
2015
|
|
|
2016
|
|
|
MUSE
TM
and related equipment
|
|
|
27
|
|
|
|
7
|
%
|
|
|
91
|
|
|
|
18
|
%
|
|
Miniature camera and related equipment
|
|
|
372
|
|
|
|
93
|
%
|
|
|
405
|
|
|
|
82
|
%
|
|
Total
|
|
|
399
|
|
|
|
100
|
%
|
|
|
496
|
|
|
|
100
|
%
|
Our
revenue to date has been primarily based on the sale of imaging equipment, which we develop and manufacture. In the
nine months ended September 30, 2016, we generated approximately $0.5 million of total revenues, compared to approximately $0.4
million in the nine months ended September 30, 2015, an increase of $97,000, or 24%. The increase was primarily due to development
services provided to National Aeronautics and Space Administration, or NASA
.
Gross
Profit
Gross
profit was $0.34 million for the nine months ended September 30, 2016, or 69% of revenues, compared to approximately $0.23 million,
or 58% of revenues for the nine months ended September 30, 2015. The increase in gross profit of $112,000, or 49% and increase
in gross profit margin was primarily due to development services provided to NASA and to one of Israel's leading industrial companies.
Operating
Expenses
The
following table presents operating expenses for the periods indicated (in thousands of U.S. dollars):
|
|
|
For the Nine Months Ended September 30,
|
|
|
|
|
2015
|
|
|
2016
|
|
|
Increase (Decrease)
|
|
|
|
|
(in thousands, U.S. dollars)
|
|
|
%
|
|
|
Research and development expenses
|
|
|
3,043
|
|
|
|
3,021
|
|
|
|
(22
|
)
|
|
|
(1
|
)
|
|
Selling and marketing expenses
|
|
|
1,943
|
|
|
|
1,845
|
|
|
|
(98
|
)
|
|
|
(5
|
)
|
|
General and Administrative expenses
|
|
|
1,911
|
|
|
|
3,016
|
|
|
|
1,105
|
|
|
|
58
|
|
|
Other income, net
|
|
|
3
|
|
|
|
|
|
|
|
(3
|
)
|
|
|
(100
|
)
|
|
Total operating expenses, net
|
|
|
6,894
|
|
|
|
7,882
|
|
|
|
988
|
|
|
|
14
|
|
Research
and Development Expenses
Research
and development cost was approximately $3 million for the nine month period ended September 30, 2016, compared to approximately
$3 million for the nine month period ended September 30, 2015.
Sales
and Marketing Expenses
Sales
and marketing expenses were approximately $1.85 million for the nine month period ended September 30, 2016, compared to approximately
$1.94 million for the nine month period ended September 30, 2015.
General
and Administrative Expenses
General
and administrative expenses were approximately $3 million for the nine month period ended September 30, 2016, compared to approximately
$1.9 million for the nine month period ended September 30, 2015. The increase of approximately $1.1 million, or 58%, resulted
primarily from an increase in professional expenses in connection with an intellectual property litigation (for more information
see “Business – Intellectual Property”).
Loss
and Loss per Share
For
the nine month period ended September 30, 2016, the loss was approximately $7.4 million or $0.22 per share, compared to loss of
approximately $6.67 million or $0.25 per share, for the nine months ended September 30, 2015. The increase in loss was mainly
due to an increase in operating expenses, resulting primarily from an increase in professional expenses in connection with an
intellectual property litigation.
Effective
Corporate Tax Rate
Our
effective consolidated tax rate for the nine months ended September 30, 2016, and September 30, 2015, was close to zero percent,
primarily due to the tax losses we accrued in Israel in those periods.
Comparison
of the fiscal years ended December 31, 2015 and December 31, 2014
Revenues
The
following tables present our total revenues, in thousands of U.S. dollars, by geographic area and by products for the fiscal years
indicated:
|
|
|
For the Year Ended December 31,
|
|
|
|
|
2014
|
|
|
2015
|
|
|
United States
|
|
|
460
|
|
|
|
62
|
%
|
|
|
446
|
|
|
|
72
|
%
|
|
Europe
|
|
|
113
|
|
|
|
15
|
%
|
|
|
84
|
|
|
|
13
|
%
|
|
Asia
|
|
|
134
|
|
|
|
18
|
%
|
|
|
51
|
|
|
|
8
|
%
|
|
Other
|
|
|
37
|
|
|
|
5
|
%
|
|
|
43
|
|
|
|
7
|
%
|
|
Total
|
|
|
744
|
|
|
|
100
|
%
|
|
|
624
|
|
|
|
100
|
%
|
|
|
|
2014
|
|
|
2015
|
|
|
MUSE
TM
and related equipment
|
|
|
91
|
|
|
|
12
|
%
|
|
|
66
|
|
|
|
11
|
%
|
|
Miniature camera and related equipment
|
|
|
653
|
|
|
|
88
|
%
|
|
|
558
|
|
|
|
89
|
%
|
|
Total
|
|
|
744
|
|
|
|
100
|
%
|
|
|
624
|
|
|
|
100
|
%
|
In
the year ended December 31, 2015, we generated approximately $0.62 million of total revenues, compared to approximately $0.74
million in the year ended December 31, 2014, a decrease of approximately $120,000, or 16%. This decrease was primarily due to
a $95,000 decrease in imaging equipment revenues and a $25,000 decrease in our MUSE
TM
system revenues. The decrease
in revenues was primarily due to the decrease in the quantity of products sold.
Gross
Profit
Gross
profit was approximately $0.35 million for the year ended December 31, 2015, or 56% of revenues, compared to approximately $0.39
million, or 53% of revenues for the year ended December 31, 2014. The decrease in gross profit of $46,000, or 12% was primarily
due the decrease in revenues.
Operating
Expenses
The
following table presents operating expenses for the periods indicated (in thousands of U.S. dollars):
|
|
|
For the Year Ended December 31,
|
|
|
|
|
2014
|
|
|
2015
|
|
|
Increase (Decrease)
|
|
|
|
|
(in thousands, U.S. dollars)
|
|
|
%
|
|
|
Research and development expenses
|
|
|
4,025
|
|
|
|
4,384
|
|
|
|
359
|
|
|
|
9
|
|
|
Selling and marketing expenses
|
|
|
2,341
|
|
|
|
2,680
|
|
|
|
339
|
|
|
|
14
|
|
|
General and Administrative expenses
|
|
|
2,280
|
|
|
|
2,842
|
|
|
|
562
|
|
|
|
25
|
|
|
Other income, net
|
|
|
269
|
|
|
|
3
|
|
|
|
(266
|
)
|
|
|
(99
|
)
|
|
Total operating expenses, net
|
|
|
8,377
|
|
|
|
9,903
|
|
|
|
1,526
|
|
|
|
18
|
|
Research
and Development Expenses
Research
and development cost was approximately $4.4 million for the year ended December 31, 2015, compared to approximately $4 million
for the year ended December 31, 2014. The increase of approximately $0.36 million, or 9% was primarily due to the increase in
the acquisition of materials by the Company and services rendered to the Company for trainings and registry activities and the
recruitment of additional human resources during 2015.
Sales
and Marketing Expenses
Sales
and marketing expenses were approximately $2.7 million for the year ended December 31, 2015, compared to approximately $2.34 million
for the year ended December 31, 2014. The increase of approximately $0.34 million, or 14% resulted primarily from the Company’s
preparations towards the marketing of its products, which is reflected primarily by recruitment of additional human resources
in the U.S.
General
and Administrative Expenses
General
and administrative expenses were approximately $2.8 million for the year ended December 31, 2015, compared to approximately $2.3
million for the year ended December 31, 2014. The increase of approximately $0.5 million, or 25%, resulted primarily from an increase
in professional expenses of approximately $0.25 million in connection with compliance requirements of the SEC and NASDAQ regulations
which we are required to comply with following our listing on NASDAQ and to the IP litigation (see “Business –
Intellectual Property”) and increase in salary costs of approximately $98,000 attributed primarily to the strengthening
of management.
Other
Income, Net
Other
income, net, in the year ended December 31, 2015 amounted to a total of approximately $3,000, compared to a total of approximately
$0.27 million for the year ended December 31, 2014. The decrease of approximately $0.27 million, or 99%, resulted primarily from
the composition of other income, net. In the year ended December 31, 2015, other income, net, included the Company’s income
from its investment portfolio in the amount of approximately $3,000. For the year ended December 31, 2014, other income, net,
included the Company’s income from its investment portfolio in the amount of approximately $19,000, and income in the amount
of approximately $0.25 million in respect of the termination of the Company’s agreement with a customer (see note 14 to
our financial statements). The decrease in income in respect of our investments resulted from a change in our risk management
made in 2013.
Profit
from Change in Fair Value of Warrants Issued to Investors
Profit
from change in the fair value of warrants issued to investors in the financial statements resulted from the revaluation of the
warrants which included a cashless exercise mechanism. During the year ended December 31, 2015, a profit of approximately $106,000
was recorded, compared to a profit of approximately $0.98 million for the year ended December 31, 2014. This profit representing
the decrease in the value of these warrants during the reported period. The decrease in value primarily resulted from the decrease
in our share price and the reduction in the time remaining for exercising the warrants due to the passage of time.
Finance
income (expenses), net
Financial
expenses, net were $14,000 for the year ended December 31, 2015, compared to financial income of approximately $0.65 million for
the year ended December 31, 2014. The decrease in finance income, net of approximately $0.66 million, was primarily from a decrease
in the financing income in respect of currency exchange differences. The increase in the average exchange rate of the U.S.
dollar in relation to the NIS in 2014 was approximately 12%, while in 2015 the increase was only 0.33%. As a result, the financing
income in respect of currency exchange differences in 2014 amounted to approximately $0.65 million (as a result of the positive
effect on our net assets (primarily cash and cash equivalents) denominated in U.S. dollars), compared to financial expenses of
$8,000 in 2015.
Loss
and Loss per Share
For
the year ended December 31, 2015, the loss was approximately $9.53 million or $0.34 per share, compared to loss of approximately
$6.34 million or $0.33 per share, for the year ended December 31, 2014. The increase in loss and loss per share in the year ended
December 31, 2015, compared to the year ended December 31, 2014, was mainly due to an increase in operating expenses, a decrease
in profits due to the change in the fair value of warrants issued to investors and increase in finance expenses as described above.
Effective
Corporate Tax Rate
Our effective consolidated tax rate for
the years ended December 31, 2015, and December 31, 2014, was close to zero percent, primarily due to the tax losses we accrued
in Israel in those periods.
Comparison of the fiscal years ended December 31, 2014 and
December 31, 2013
Revenues
The following tables present our total revenues,
in thousands of U.S. dollars, by geographic area and by products for the fiscal years indicated:
|
|
|
For
the Year Ended December 31,
|
|
|
|
|
2013
|
|
|
2014
|
|
|
United States
|
|
|
451
|
|
|
|
65
|
%
|
|
|
460
|
|
|
|
62
|
%
|
|
Europe
|
|
|
66
|
|
|
|
10
|
%
|
|
|
113
|
|
|
|
15
|
%
|
|
Asia
|
|
|
137
|
|
|
|
20
|
%
|
|
|
134
|
|
|
|
18
|
%
|
|
Other
|
|
|
37
|
|
|
|
5
|
%
|
|
|
37
|
|
|
|
5
|
%
|
|
Total
|
|
|
691
|
|
|
|
100
|
%
|
|
|
744
|
|
|
|
100
|
%
|
|
|
|
2013
|
|
|
2014
|
|
|
MUSE
TM
and related equipment
|
|
|
13
|
|
|
|
2
|
%
|
|
|
91
|
|
|
|
12
|
%
|
|
Miniature camera and related equipment
|
|
|
678
|
|
|
|
98
|
%
|
|
|
653
|
|
|
|
88
|
%
|
|
Total
|
|
|
691
|
|
|
|
100
|
%
|
|
|
744
|
|
|
|
100
|
%
|
In the year ended December 31, 2014, we generated
approximately $0.74 million of total revenues, compared to approximately $0.70 million in the year ended December 31, 2013, an
increase of approximately $53,000, or 7.7%. This increase was primarily due to an approximately $78,000 increase in MUSE
TM
system revenues, offset by a decrease of approximately $25,000 in imaging equipment revenues. The increase in the MUSE
TM
system revenues was primarily due to the increase in the quantity of products sold. The decrease in revenues in the imaging
equipment revenues was primarily due to the following:
|
|
(i)
|
during
the year ended December 31, 2013 we recorded revenues for development services provided to a customer in the amount of approximately
$86,000 (see ‘Customer A’ in note 18e to our financial statements for the year ended December 31, 2015).We did
not receive any revenue from this customer during the year ended December 31, 2014;
|
|
|
(ii)
|
during
the year ended December 31, 2013 we recorded revenues from equipment sales to a customer in the amount of approximately $150,000
(see ‘Customer B’ in note 18e to our financial statements for the year ended December 31, 2015) compared to revenues
from this client of approximately $46,000 recorded for the year ended December 31,2014. The decrease in revenues of approximately
$104,000 was primarily due to decrease in the quantity of products sold ;
|
|
|
(iii)
|
during
the year ended December 31, 2013 we recorded revenues from equipment sales to a customer in the amount of approximately $26,000.
We did not receive any revenue from this customer during the year ended December 31, 2014;
|
|
|
(iv)
|
during
the year ended December 31, 2014, we recorded
revenues of approximately $100,000 from the termination of
an agreement with a customer (see note 14 to our financial statements for the year ended December 31, 2015) compared to revenues
under this agreement of approximately $50,000 recorded for the year ended December 31, 2013;
|
|
|
(v)
|
during
the year ended December 31, 2014 we recorded revenues for development services provided to a customer in the amount of approximately
$114,000 (see ‘Customer D’ in note 18d to our financial statements for the year ended December 31, 2015) . We
did not receive any revenue from this customer during the year ended December 31, 2013.
|
Gross Profit
Gross profit was $0.39 million for the year
ended December 31, 2014, or 53% of revenues, compared to $0.38 million, or 55% of revenues for the year ended December 31, 2013.
The increase in gross profit of approximately $13,000, or 3%, was primarily due to the increase in revenues.
Operating Expenses
The following table presents operating expenses
for the periods indicated (in thousands of U.S. dollars):
|
|
|
For
the Year Ended December 31,
|
|
|
|
|
2013
|
|
|
2014
|
|
|
Increase (Decrease)
|
|
|
|
|
(in
thousands, U.S. dollars)
|
|
|
%
|
|
|
Research and development expenses, net
|
|
|
2,275
|
|
|
|
4,025
|
|
|
|
1,750
|
|
|
|
77
|
|
|
Selling and marketing expenses
|
|
|
900
|
|
|
|
2,341
|
|
|
|
1,441
|
|
|
|
160
|
|
|
General and Administrative expenses
|
|
|
1,908
|
|
|
|
2,280
|
|
|
|
372
|
|
|
|
19
|
|
|
Other income, net
|
|
|
181
|
|
|
|
269
|
|
|
|
88
|
|
|
|
49
|
|
|
Total operating expenses, net
|
|
|
4,902
|
|
|
|
8,377
|
|
|
|
3,475
|
|
|
|
71
|
|
Research and Development Expenses
Research and development cost, net was $4
million for the year ended December 31, 2014, compared to $2.3 million for the year ended December 31, 2013. The increase of $1.7
million, or 77%, was primarily due to the increase in the acquisition of materials by the Company and services rendered to the
Company by sub-contractors of approximately NIS $0.9 million, the increase in compensation expenses of approximately $0.6 million,
the increase in travel expenses and related expenses of approximately $0.2 million as a result of the cancellation of the reduction
of expenses which took place in 2013 until the completion of the investment by OrbiMed, the recruitment of additional employees
and consultants by the Company in Israel and our U.S. Subsidiary and decrease of approximately $126,000 in grants received from
the OCS due to the conclusion of OCS funding of approved programs.
Sales and Marketing Expenses
Sales and marketing expenses were approximately
$2.3 million for the year ended December 31, 2014, compared to approximately $0.9 million for the year ended December 31, 2013.
The increase of approximately $1.4 million, or 160%, resulted from an increase in salaries, wages and related expenses, marketing
expenses and travel expenses, attributed primarily to our increased efforts to penetrate the U.S. and European markets. Such efforts
included, among other things, the recruitment of additional sales and marketing personnel in Israel, Europe and the United States
and the expansion of our marketing activities including participating in exhibitions and re-branding.
General and Administrative Expenses
General and administrative expenses were
approximately $2.3 million for the year ended December 31, 2014, compared to approximately $1.9 million for the year ended December
31, 2013. The increase of approximately $0.4 million, or 19%, resulted primarily from an increase in salary costs of approximately
$0.2 million attributed primarily to the hiring of Chris Rowland as our chief executive officer, an increase in professional expenses
of approximately $0.5 million in connection with the preparation of a shelf offering and from activities required for implementing
the registration of an ADR facility offset partially by expenses in connection with the 2013 shelf prospectus offering to the
public in Israel and a decrease in management fees of approximately $0.4 million attributed to the termination of employment of
Dr. Elazar Sonnenschein as the our chief executive officer.
Other Income, Net
Other income, net, was approximately $0.3
million for the year ended December 31, 2014, compared to approximately $0.2 million for the year ended December 31, 2013. The
increase of approximately $0.1 million, or 49%, resulted primarily from the composition of other income, net, in those two periods.
For the year ended December 31, 2014, other income, net, included the Company’s income from its investment portfolio in
the amount of approximately $19,000, and income in the amount of approximately $0.25 million in respect of the termination of
the Company’s agreement with a customer (see note 14 to our financial statements for the year ended December 31, 2015).
In the year ended December 31, 2013, other income, net, included the Company’s income from its investment portfolio in the
amount of approximately $91,000, and income in the amount of approximately $91,000 in connection with the realization of a financial
asset available for sale. The decrease in income in respect of our investments portfolio as compared with the same period in 2013
resulted from a decision by our Investment Committee, as part of our risk management and a more conservative investment management
approach, pursuant to which, during the second quarter of 2013 we disposed of the bonds held at that time, and invested the cash
balances solely in deposits and short-term government loan notes, which have a lower return than the rates on bonds.
Profit from Change in Fair Value of Warrants Issued
to Investors
Gains recorded in the financial statements
on the change in the fair value of warrants issued to investors for the year ended December 31, 2014, resulted from the revaluation
of the warrants which included a cashless exercise mechanism. The revaluation was performed in accordance with the fair value
of the warrants as of the December 31, 2014. During the year ended December 31, 2014, a gain of approximately $1 million was recorded,
representing the decrease in the value of the warrants during this reporting period. The primary reasons for the decrease in value
was due to the decrease in our share price and the reduction in the time remaining for exercising the warrants due to the passage
of time.
Finance income (expenses), net
Financing income, net was approximately $0.7
million for the year ended December 31, 2014, compared to financial expense, net of approximately $100,000 for the year ended
December 31, 2013. The increase in financing income, net of approximately $0.8 million, was primarily due to the increase in the
average exchange rate of the U.S. dollar in relation to the NIS, which had a positive effect on our net assets (primarily cash
and cash equivalents) denominated in U.S. dollars.
Loss and Loss per Share
For the year ended December 31, 2014, the loss was $6.4 million
or $0.33 per share, compared to loss of $1.4 million or $0.11 per share, for the year ended December 31, 2013. The increase in
loss and loss per share in the year ended December 31, 2014, compared to the year ended December 31, 2013, is mainly due to an
increase in operating expenses, net of approximately $3.5 million, a decrease in profits due to the change in the fair value of
warrants issued to investors of approximately $2.2 million, offset by an increase in finance income of approximately $0.8 million.
Effective Corporate Tax Rate
Our effective consolidated tax rate for the years ended December
31, 2014, and December 31, 2013, was close to zero percent, primarily due to the tax losses we accrued in Israel in those periods.
Impact
of Inflation, Devaluation and Fluctuation in Currencies on Results of Operations, Liabilities and Assets
We
generate part of our revenues in different currencies than our functional currency, such as NIS and Euros. As a result, some of
our financial assets are denominated in these currencies, and fluctuations in these currencies could adversely affect our financial
results. A considerable amount of our expenses are generated in U.S. dollars, but a significant portion of our expenses such as
salaries is generated in other currencies such as NIS. In addition to our operations in Israel, we are expanding our international
operations
in the European Union. Accordingly, we incur and expect to continue to incur additional expenses in non
U.S. dollar currencies, such as the Euro.
As a result, some of our financial liabilities are denominated in non-U.S.
dollar currencies.
Due to the foregoing and the fact that our financial results are currently measured in U.S. dollar,
our results could be adversely affected as a result of a strengthening or weakening of the U.S. Dollar compared to these other
currencies.
We
believe that inflation in Israel has not had a material effect on our results of operations.
Effective
January 1, 2016, the Company changed its functional currency to the U.S. dollar from the NIS. This change was based on an assessment
by Company management that the dollar is the primary currency of the economic environment in which the Company operates.
Liquidity
and Capital Resources
Since inception,
we have funded our operations primarily through public and private offerings of our securities, sales of our products and grants
from the OCS. Since inception, we have raised approximately $58 million in aggregate net proceeds from issuing our equity securities.
As of September 30, 2016, we held approximately $3.9 million in cash and cash equivalents.
As
of
March 15
, 2017, we had a total cash balance of approximately $1.7 million.
Net
cash used in operating activities primarily reflects the operating loss for those periods, sales (acquisitions) of financial assets
at fair value through profit or loss and changes in operating assets and liabilities. Net cash used in operating activities was
approximately $7.8 million for the nine months period ended September 30, 2016, compared with net cash used in operating activities
of approximately $4.2 million for the nine months period ended September 30, 2015. The $3.6 million increase in net cash used
in operating activities during the nine-month period ended September 30, 2016, compared to the nine-month period ended September
30, 2015 was primarily the result of a sales of tradeable securities (financial assets at fair value through profit or loss) of
approximately $2.1 million in the first quarter of 2015 compared to no tradeable securities in the nine months period ended September
30, 2016. Net cash used in operating activities was approximately $7 million for the year ended December 31, 2015, compared with
net cash used in operating activities of approximately $7.8 million for the year ended December 31, 2014. The decrease from 2015
to 2014 of approximately $0.8 million was primarily the result of a sales of tradeable securities (financial assets at fair value
through profit or loss) of approximately $2.1 million in the first quarter of 2015 offset the increase of an operating loss of
approximately $1.6 million.
Net
cash used in investing activities for the nine months period ended September 30, 2016, was approximately $35,000, compared to
net cash used in investing activities of approximately $78,000 for the nine months period ended September 30, 2015. Net cash used
in investing activities for the year ended December 31, 2015, was approximately $0.1 million, compared to net cash generated from
investing activities of approximately $2.2 million for the year ended December 31, 2014. The decrease for the year ended December
31, 2015, compared to the year ended December 31, 2014, in the amount of $2.3 million was the result of a withdrawal of short-term
deposits in 2014.
Operating
Capital and Capital Expenditure Requirements
To
date, we have not achieved profitability and have sustained net losses in every fiscal year since our inception, including a net
loss of $7.4 million for the nine months period ended September 30, 2016, and $9.5 million for the year ended December 31, 2015.
Based
on the projected cash flows and our cash balance as of September 30, 2016, the Company’s management is of the opinion that
without further fund raising it will not have sufficient resources to enable it to continue advancing its activities including
the development, manufacturing and marketing of its products for a period of at least 12 months from the date of approval of the
financial statements. As a result, there is substantial doubt about our ability to continue as a going concern.
At
our current burn rate, our current cash balance will be sufficient until approximately April 2017, taking into account shut down
costs.
We
will need to seek additional sources of funds, including selling additional equity, debt or other securities or entering into
a credit facility, take costs reduction steps or modify our current business plan to achieve profitability. If we raise additional
funds through the issuance of debt securities, these securities may have rights senior to those of our ordinary shares and could
contain covenants that could restrict our operations and ability to issue dividends. We may also require additional capital beyond
our currently forecasted amounts. Any required additional capital, whether forecasted or not, may not be available on reasonable
terms, or at all. If we are unable to obtain additional financing, we may be required to reduce the scope of, delay or eliminate
some or all of our planned research, development and commercialization activities, which could materially harm our business and
results of operations.
Because
of the numerous risks and uncertainties associated with the development of medical devices and the current economic situation,
we are unable to estimate the exact amounts of capital outlays and operating expenditures necessary to complete the development
of our products and successfully deliver commercial products to the market. Our future capital requirements will depend on many
factors, including but not limited to the following:
|
|
●
|
the revenue generated by sales of our current and future products;
|
|
|
|
|
|
|
●
|
the expenses we incur in selling and marketing our products and supporting our growth;
|
|
|
|
|
|
|
●
|
the costs and timing of regulatory clearance or approvals for new products or upgrades or changes to our products;
|
|
|
|
|
|
|
●
|
the expenses we incur in complying with domestic or foreign regulatory requirements imposed on medical device companies;
|
|
|
|
|
|
|
●
|
the expenses associated with achieving a reimbursement code for our MUSE
TM
procedure;
|
|
|
|
|
|
|
●
|
the rate of progress, cost and success or failure of on-going development activities;
|
|
|
|
|
|
|
●
|
the emergence of competing or complementary technological developments;
|
|
|
|
|
|
|
●
|
the costs of filing, prosecuting, defending and enforcing any patent or license claims and other intellectual property rights;
|
|
|
|
|
|
|
●
|
the terms and timing of any collaborative, licensing, or other arrangements that we may establish;
|
|
|
|
|
|
|
●
|
the future unknown impact of recently enacted healthcare legislation;
|
|
|
|
|
|
|
●
|
the acquisition of businesses, products and technologies; and
|
|
|
|
|
|
|
●
|
general economic conditions and interest rates.
|
Research
and Development, Patents and Licenses, Etc.
Our
research and development efforts are focused on continuous improvement of the MUSE
TM
system, as well as investment in future
products. We conduct all of our research activity in Israel. As of September 30, 2016, our research and development team, including
regulatory and quality team members, consisted of 12 employees and consultants. In addition, we work with subcontractors for the
development of our products when needed. We have assembled an experienced team with recognized expertise in mechanical and electrical
engineering, software, control algorithms and systems integration, as well as significant medical and clinical knowledge and expertise.
We
finance our research and development activities mainly through sale of our products, capital raising and grants received from
the OCS. As of September 30, 2016, we had received total grants from the OCS of $2.5 million. For further information see “Business.”
For a description
of the amount spent during each of the last three fiscal years on company-sponsored research and development activities, see “Management’s
Discussion and Analysis of Financial Condition and Results of Operations.”
We
also invest resources in the protection of our intellectual property. For this purpose, we file from time to time applications
for patent registration in the certain countries in which we are active and in other countries, which we consider potential markets.
Trend
Information
The
following is a description of factors that may influence our future results of operations, including significant trends and challenges
that we believe are important to an understanding of our business and results of operations:
To
date, substantially all of our revenues have been generated from the sale of imaging equipment, with the balance being generated
from the
sale of the
MUSE
TM
System. The level of our future revenues is hard to predict and depends
on many factors, which are outside of our control. For instance, future revenues from the sale of our products may
be adversely affected by current general economic conditions and the resulting tightening of credit markets, which may cause purchasing
decisions to be delayed, our customers to have difficulty securing adequate funding to buy our products or, in an extraordinary
event, may cause our customers to experience difficulties in complying with their engagements with us. In addition, revenue growth
depends on the acceptance of our technology in the market.
The
healthcare industry in the United States has experienced a trend toward cost containment as government and private insurers seek
to manage healthcare costs by imposing lower payment rates and negotiating reduced contract rates with service providers. This
trend may result in inadequate coverage for procedures, especially those utilizing new technology, or result in new technology
not receiving reimbursement coverage, which may negatively impact utilization of our products. In addition, medical
malpractice carriers are withdrawing coverage in certain states or substantially increasing premiums. If this trend continues
or worsens, physicians and surgeons may discontinue using our system or may choose to not purchase our system in the future due
to the cost or inability to procure insurance coverage. However, we believe there is also a trend for hospitals and physicians
to use devices which enable less invasive procedures and to replace older more invasive devices which may result in longer hospital
stays and therefore higher medical costs. We believe this trend provides a positive market outlook for our products.
We
sell our products using a direct sales force and in certain markets via third-party distributors. Since January 2013, we have
entered into certain distribution agreements to sell our MUSE
TM
system in Italy and China (the latter pending achievement
of regulatory clearance for our MUSE
TM
system in China). We sell our products in the United States and certain parts
of Europe
using our direct sales force.
Off-Balance
Sheet Arrangements
We
do not currently have any off-balance sheet arrangements that have had, or are reasonably likely to have, a current or future
effect on our financial condition, changes in financial condition, revenues or expenses, results of operations, liquidity, capital
expenditures or capital resources that are material to investors.
Tabular
Disclosure of Contractual Obligations
The
following table summarizes our known contractual obligations and commitments as of December 31, 2015:
|
|
|
Total
|
|
|
Less than
1 year
|
|
|
1 – 3 years
|
|
|
|
|
(U.S. dollar, in thousands)
|
|
|
Car lease obligations
|
|
|
184
|
|
|
|
93
|
|
|
|
91
|
|
|
Premises leasing obligations
|
|
|
232
|
|
|
|
118
|
|
|
|
114
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
416
|
|
|
|
211
|
|
|
|
205
|
|
Other
Long-Term Liabilities Reflected on the Company’s Balance Sheet
We
have a financial liability in respect of warrants convertible into a variable number of our shares, which is a derivative
instrument in the amount $32,000 as of September 30, 2016.
As
of September 30, 2016, the total amount set aside as an actuarial estimate by the Company to provide post-employment benefits
for certain employees and office holders was in the aggregate amount of approximately $101,000. The Company has not set aside
amounts to provide post-employment benefits for the remaining employees and office holders. The liability for employees’
severance benefits is calculated on the basis of the latest monthly salary paid to each employee multiplied by the number of years
of employment. The liability is covered by the amounts deposited by us into employees’ managers’ insurance or pension
fund accounts in respect of severance obligations to such employees, including accumulated income thereon as well as by the unfunded
provision reflected on the balance sheet. While the timing of such obligations cannot be pre-determined (and as such
were not included in the above table), such liability will be removed, either by termination of employment or retirement.
Business
Overview
We
are a medical device company dedicated to the development, manufacturing and marketing of surgical endostaplers and direct vision
systems for minimally invasive medical procedures. To date, substantially all of our revenues have derived from
our
miniaturized
imaging equipment for use in medical and industrial applications. Our expertise is in the development, production and marketing
of innovative surgical devices with direct visualization capabilities for the treatment of Gastroesophageal Reflux Disease, or
GERD, a common ailment, which is predominantly treated by medical therapy (e.g. proton pump inhibitors) or in chronic cases, conventional
open or laparoscopic surgery. Our FDA-cleared and CE-marked endosurgical system, known as the MUSE
TM
(Medigus Ultrasonic
Surgical Endostapler) system, enables minimally-invasive and incisionless procedures for the treatment of GERD by reconstruction
of the esophageal valve via the mouth and esophagus, eliminating the need for surgery in eligible patients. We believe that this
procedure offers a safe, effective and economical alternative to the current modes of GERD treatment for certain GERD patients,
and has the ability to provide results which are equivalent to those of standard surgical procedures while reducing pain and trauma,
minimizing hospital stays, and delivering economic value to hospitals and payors.
The
key elements of the MUSE
TM
system include a single-use endostapler containing several sophisticated innovative technologies
such as flexible stapling technology, a miniature camera and ultrasound sensor, as well as a control console offering a video
image transmitted from the tip of the endostapler.
In
addition to the MUSE
TM
system for the treatment of GERD, we have developed miniaturized video cameras for use in various
medical
procedures
as well as specialized industrial applications.
Prevalence
of GERD
GERD, is a worldwide disorder, with evidence
suggesting an increase in GERD disease prevalence since 1995. The sample size weighted mean for the GERD population in the United
States and Europe is 19.8% and 15.2% respectively. In the United States alone, over 49 million adults are affected by the disease,
with over 29 million adults suffering daily GERD symptoms. Proton pump inhibitors, or PPIs, are a class of effective and generally
safe medication to treat GERD, but not everyone who experiences heartburn needs a PPI. Several PPIs have been widely advertised
to consumers and heavily promoted by physicians. This has led to an overuse of the drug. PPIs are the third highest selling class
of drugs in the U.S. and Nexium has the second highest retail sales among all drugs at $4.8 billion in 2008. This figure does
not include sales of other brands of PPIs.
After
being swallowed, food descends through the esophagus to the stomach, which contains acids and enzymes intended to digest and break
down food. GERD is caused by the defective operation of the lower esophageal sphincter, or LES, a valve, which controls the flow
of ingested food from the esophagus into the stomach. While eating and between eating periods, a properly operating LES prevents
stomach contents from entering the esophagus. Among GERD sufferers, the valve opens spontaneously or is unable to close properly.
This results in acidic stomach contents rising into the esophagus, causing irritation, acid reflux and heartburn, as well as other
potentially dangerous conditions.
Beyond painful symptoms, GERD may also increase
sufferers’ susceptibility to cancer. Whereas the stomach is lined by the “gastric mucosal barrier” which allows
acidic material to be contained harmlessly, the surface of the esophagus consists of flat, thin cells called squamous cells,
which are not resistant to acid. Repeated episodes of acid reflux can cause inflammation of the esophagus, a condition called esophagitis.
The flat cells lining the esophagus can also undergo genetic changes due to exposure to acid, causing these cells to resemble those
found in the stomach lining, a condition known as Barrett’s Esophagus. Studies have shown that people exhibiting Barrett’s
Esophagus have a higher risk of developing cancer of the esophagus.
Barrett Esophagus is a complication of GERD
and predisposes patients to esophageal adenocarcinoma, a tumor that has increased in incidence more than 7-fold over the past
several decades. Studies have also shown, that compared to patients not exhibiting GERD symptoms, patients exhibiting weekly symptoms
of GERD have a five times higher probability for developing esophageal cancer while patients exhibiting daily symptoms of GERD
have a seven times higher probability for developing esophageal cancer. The most common risk factors for cancer in Barrett esophagus
patients include chronic GERD, hiatal hernia, advanced age, male sex, white race, cigarette smoking and obesity.
Treatment
of GERD
Treatment
of GERD involves a stepwise approach. The goals are to control symptoms and to heal esophagitis and to prevent recurrent esophagitis.
The treatment is based on lifestyle modification and control of gastric acid through medical treatment (antacids, PPI’s,
H2 blockers or other reflux inhibitors) or antireflux surgery. Mild GERD may be defined as intermittent reflux symptoms that can
be managed with lifestyle changes or over-the-counter medications. Moderate to severe GERD represents more chronic symptoms that
may require stronger drugs, long term medication or surgical intervention.
1.
Drug treatment - Proton pump inhibitors (PPI)
For
moderate to severe GERD, physicians usually prescribe proton pump inhibiting drugs, or PPI. This class of drugs reduces acid production
by the stomach, and thereby relieves the patients of their symptoms. Drugs of this class are among the most commonly prescribed
medications in the world. There are several brands on the market, best known are Prilosec (omeprazole), Prevacid (
lansoprazole)
and Nexium (esomeprazole
). Certain PPI drugs are available over the counter in the United States and in other countries,
but the over the counter dosage may be inadequate to control GERD symptoms, except in mild cases.
While
PPI drugs effectively reduce the severity and frequency of GERD symptoms, they have a number of drawbacks:
a)
In approximately 30% of patients, symptom control is incomplete;
b)
The drugs do not treat the disease, they only control its manifestations, therefore must be taken for life at a dosage which requires
prescription. Accumulated costs may be substantial; and
c)
Long term use is associated with a number of serious adverse effects. In particular, they increase the risk of osteoporosis and
fractures of the hip, wrist and spine. The FDA had issued a warning on this effect as well as warnings against other untoward
effects on absorption of other essential minerals, which may lead to chronic kidney disease, irregular heartbeat, diarrhea and
increase flatulence.
2.
Interventional treatment
The
most common operation for GERD is called a surgical fundoplication, a procedure that prevents reflux by wrapping or attaching
the upper part of the stomach around the lower esophagus and securing it with sutures. Due to the presence of the wrap or attachment,
increasing pressure in the stomach compresses the portion of the esophagus which is wrapped or attached by the stomach, and prevents
acidic gastric content from flowing up into the esophagus. Today, the operation is usually performed laparoscopically: instead
of a single large incision into the chest or abdomen, four or five smaller incisions are made in the abdomen, and the operator
uses a number of specially designed tools to operate under video control.
The
operation does not completely eliminate the use of PPI, and up to approximately 60% still use some in long term follow up. Nevertheless,
the dose is usually lower – in the over the counter range - and the response rate is excellent. Since the majority of patients
referred to surgeons are incomplete responders, or require a high dose of PPI, the patients are generally satisfied with the operation,
and the overall costs of treatment are lower in the long run.
In
spite of the clinical outcome of surgery, relatively few patients undergo surgery. We estimate that large numbers of patients
who are candidates for operative treatment are either not referred by their treating physician or decline it. We believe that
many patients decline to undergo operations to avoid even minute scars or violation of the abdominal cavity.
Given
the current environment in which the vast majority of GERD sufferers in North America and Europe must choose between long-term
pharmaceutical therapy and surgery, leading to what is known in our industry as the “treatment gap”, we believe there
is a demand for a minimally-invasive, incision-less procedure which treats the root cause of the disease. We believe that the
MUSE
TM
system is positioned to fill this need.
Our
system achieves the general physiological result of surgical fundoplication, by inserting the MUSE
TM
endostapler
through the mouth and the esophagus, and stapling the top of the stomach to the side of the esophagus. The endostapler contains
a video camera and stapling system. Staples have long been used in surgical procedures in place of sutures, and we believe that
they are at least as reliable and potentially more durable. Our endostapler uses standard surgical staples.
First line therapy for GERD includes a combination
of lifestyle modifications and medical therapy, or PPIs. Unfortunately, 25% to 42% of patients with GERD do not respond to an
initial 4-8-week treatment of PPI. In those who do respond to therapy, the effectiveness of PPI treatment decreases over time.
Antireflux surgery controls acid reflux and treats an incompetent lower esophageal sphincter, while also improving patient quality
of life in the long term. Thus, PPI therapy and lifestyle modifications are frequently eliminated.
Despite the effectiveness of surgery, it
is invasive, requires hospitalization, and carries the risk of short and long-term complications, including dysphagia, diarrhea,
and gas bloat syndrome Thus, endoscopic therapies that mimic the mechanism through which surgery works and can reduce surgical
morbidity have gained popularity for the treatment of GERD.
The market for medical devices, including
the market for endoscopic therapies, is very broad, with an increasing demand for new less invasive alternatives to the existing
surgical procedures for the treatment of various diseases. This increasing need for minimally invasive and incision-less treatments,
such as endoscopy-based procedures, are also augmented by the increase in the average age of global population. In 2000, the worldwide
population of persons aged more than 65 years was an estimated 420 million. During 2000-2030, the worldwide population aged more
than 65 years is projected to increase by approximately 550 million to 973 million. This increase in age will potentially lead
to increased health-care costs and may have dramatic consequences for public health and the health care financing and delivery
systems significant patient benefits and cost savings.
Endoscopy is a minimally invasive method of performing
investigative, diagnostic and therapeutic medical procedures, employing an endoscope, which allows real-time visual observation
of the patient’s internal organs during the procedure. Endoscopic procedures are most commonly performed through natural
orifices, including via the mouth, to avoid incisions. Because of the accessibility of the digestive tract through the mouth, the
endoscopy field is largely focused on disorders of the gastrointestinal tract such as disorders of the colon, esophagus, stomach
and duodenum.
Endoscopes are commonly composed of a flexible
tube with a camera installed at its tip. Endoscopes often include “working channels” through which catheters or other
endoscopic tools or devices may be inserted directly into the patient’s digestive system. The primary advantage of endoscopy
is the elimination of incisions to the patient’s body during a medical procedure. We believe that this is safer, prevents
most post-operative pain and facilitates faster recuperation. Additionally, the patient perception or preference is important
as well. The perception of endoscopy procedures as being safer, and less painful than, corresponding surgical procedures may have
the effect of minimizing patient fears.
Endoscopic procedures generally involve less
recovery time and patient discomfort than conventional open or laparoscopic surgery. These procedures are also typically performed
in the outpatient hospital setting as opposed to an inpatient setting. Typically, outpatient procedures cost the hospital or the
insurer less money since there is no overnight stay in the hospital.
Our
Solution and Products
The
MUSE
TM
system
Our
primary product, the MUSE
TM
system for transoral fundoplication, is a single use innovative device for the
incisionless treatment of GERD, which is based on our proprietary platform technology and know-how. While at present substantially
all of our revenue is derived from the miniature video camera and related equipment, our strategy is focused on the development
and promotion of its MUSE
TM
System, which we therefore refer to as our ‘primary product’.
Transoral means the procedure is
performed through the mouth, rather than through incisions in the abdomen. The MUSE
TM
system for transoral fundoplication
was previously known as the SRS
TM
Endoscopic Stapling System. We rebranded to the MUSE
TM
system following the
launch of the most recent generation product. The MUSE
TM
system is used to perform a procedure as an alternative to
a surgical fundoplication. The MUSE
TM
offers an endoscopic, incisionless alternative to surgery. A single surgeon or
gastroenterologist can perform the MUSE
TM
procedure, unlike in a laparoscopic fundoplication which requires incisions.
The system consists of the MUSE
TM
controller console, the MUSE
TM
endostapler
and several accessories, including an overtube, irrigation bottle, tubing supplies and staple cartridges. The endostapler incorporates
a video camera, a flexible surgical stapler and an ultrasonic sight, which is used to measure the distance between the anvil and
the cartridge of the stapler, to ensure their proper alignment and tissue thickness. The device also contains an alignment pin,
which is used for initial positioning of the anvil against the cartridge, two anvil screws, which are used to reduce the thickness
of the tissue that needs to be stapled and to fix the position of the anvil and the MUSE
TM
endostapler during stapling.
The system allows the operator to staple the fundus of the stomach to the esophagus, in two or more locations, typically around
the circumference, thereby creating a fundoplication, without any incisions.
The
clearance by the FDA, or ‘Indications for Use’, of the MUSE
TM
System is "for endoscopic
placement of surgical staples in the soft tissue of the esophagus and stomach in order to create anterior partial
fundoplication for treatment of symptomatic chronic Gastro-Esophageal Reflux Disease in patients who
require and respond to pharmacological therapy". As such, the FDA clearance covers the use by an operator of
the MUSE
TM
endoscopic stapler as described in the above paragraph. In addition,
in the pivotal study that was presented to the
FDA in order to gain clearance, only patients who were currently taking GERD
medications (i.e. pharmacological therapy) were allowed in the study. In addition, all patients had to have
a significant decrease in their symptoms when they were taking medication compared
to when they were off the medication.
As such, the FDA clearance included the indication that MUSE
TM
is intended for patients
who require and respond to pharmacological therapy. The MUSE
TM
System indication does not restrict its
use with respect to GERD severity from a regulatory point of view. However, clinicians typically only consider
interventional treatment options for moderate to severe GERD. Therefore, it is reasonable to expect the MUSE
TM
System would be primarily used to treat moderate and severe GERD in practice. The system has received 510(k)
marketing clearance from the FDA in the United States, as well as a CE mark in Europe and a license from Health Canada. It is
also cleared for use in Turkey and in Israel.
Clinical
studies
The
original FDA submission for the MUSE
TM
System included short-term (6 month) results from a multi-center clinical trial.
The trial was conducted in support of the 510(k) marketing clearance submission for the system and pursuant to an FDA-issued Investigational
Device Exemption (IDE).
Enrollment was completed in November 2010.
A total of 72 patients were enrolled and 69 were treated with the MUSE
TM
system during the study. A manuscript detailing
the results of this study was published in Surgical Endoscopy and is currently available online. Publication in the hardcopy of
the journal was in the January 2015 issue.
The
primary objective of the study was to assess the safety and efficacy of the system in the treatment of subjects
with GERD. The primary efficacy endpoint was at least a 50% improvement in the GERD-HRQL (Health Related Quality of Life) scores
in 53% of the subjects. HRQL is the standard assessment of how an individual’s well-being may be affected over time by a disease.
Secondary efficacy assessments included PPI intake, esophageal acid exposure during a 24-hour period and anatomical changes. The
follow-up period was set at six months following each procedure.
The
primary endpoint was met in that 73% of subjects exhibited at least a 50% reduction in HRQL at six months. In addition, 85% of
subjects reduced their PPI intake by at least 50%, with 65% of subjects eliminating PPI use completely at six months.
FDA
marketing clearance for our system was granted in May 2012 following the original FDA submission. Subsequent improvements to the
system included improvements to the camera, illumination and alignment mechanisms, the addition of an electronic stapling motor,
and condensing two control consoles into a single unit. FDA clearance for the modified system was obtained in June 2015. The modified
system has also obtained a CE mark in Europe and a license from Health Canada and was approved in Turkey and Israel.
In
May 2013, we received five years of follow-up results for a precursor IRB (Institutional Review Board) approved pilot study of
the system conducted in 2007 at Deenanath Mangeshkar Hospital and Research Center in the city of Pune, India. The results of this
follow-up study were published in the peer review journal Surgical Endoscopy in
March 2015. As noted in the journal
article, the five-year results are similar to the results obtained from subjects who received-laparoscopic procedures for GERD
in the same period. Each year, eleven of the thirteen patients were reached (although not always the same eleven). All thirteen
patients had at least a four year follow-up. Throughout the follow up period, GERD-HRQL scores were normal in all but one patient.
All patients indicated that they would agree to do the procedure again. Out of the initial thirteen patients, seven (54%) had
eliminated PPI and another three (23%) reduced PPI use by 50% or more. It should be emphasized that for this trial patients were
selected with GERD severity at a higher than average level (moderate to severe), a fact which may indicate an even greater outcome
of the effect of the system in an average GERD level patient population.
In
November 2015, a follow-up study conducted in the United States looked at evaluating the long-term clinical outcome of 37 patients
who received GERD treatment with the MUSE
TM
system in the multi-center study mentioned above was concluded. Efficacy
and safety data were analyzed up to four years post-procedure. No new complications have been reported in such long-term analysis.
The proportions of patients who remained off daily PPI were 83.8% (31/37) at six months, and 69.4% (25/36) at 4 years post-procedure.
GERD-HRQL scores off PPI were significantly decreased following six months and four years post-procedure. The authors concluded
that the MUSE
TM
stapling device appeared to be safe and effective in improving symptom scores as well as reducing PPI
use in patients with GERD. These results appeared to be equal to or better than those of the other devices for endoluminal GERD
therapy.
In
February 2017, we received an approval to start a multi-center MUSE
TM
clinical study in China after the CFDA reviewed the
ethics committees' approval and agreements were in place with each study location. Under Principal Investigator, Yunsheng Yang,
Director of Gastroenterology Department Clinical center at 301 Hospital and Chairman of Chinese Society of Gastroenterology, The
General Hospital of People's Liberation Army in Beijing, the clinical study will include approximately 62 patients, will take
place at 5 centers across China: The General Hospital of People’s Liberation Army, Renji Hospital of Shanghai, Shanghai
General Hospital, Peking University Third Hospital and Navy General Hospital.
Procedures started in March 2017 and are
expected to carry on through 2017, with results being reported to the CFDA in 2018 as part of the CFDA submission for clearance
to sell MUSE
TM
in China.
Miniature
Video Cameras
By
definition all endoscopes must include vision apparatus to facilitate the operator’s view of the internal organs of the
patient. In the past, fiber optics were utilized for this purpose, and have been gradually replaced with electronic video systems
offering higher resolution and higher-quality images. We have developed several models of miniaturized digital video cameras and
video processing equipment, for use in medical endoscopy products as well as industrial uses. Our cameras range between 3.45mm
to 0.99 mm in diameter, and are based on single-use CMOS image sensors. In some cases, our cameras are relatively inexpensive,
allowing them to be used in single-use devices.
Our
miniature cameras are intended for use in medical applications in which it has not yet been feasible to use miniature video cameras,
and may be integrated into devices developed by the company, or by third parties who source the camera from us. We expect that
the growing demand for single-use medical devices will increase demand for the CMOS cameras in particular, in fields such as gastroenterology,
orthopedics, gynecology, ears nose throat, urology, cardio-vascular, and other fields in which diagnostic and surgical procedures
may be performed endoscopically. Small-diameter video cameras permit not only smaller camera-based endoscopes which are able to
penetrate previously inaccessible organs or visualize them in improved image quality, but also allows for the addition of working
channels and other features in the valuable space freed by the reduction in camera size.
Our
most advanced camera is a prototype CMOS-based camera measuring only 0.99 mm in diameter transmitting 45,000 pixels in HDMI format,
which we believe to be the smallest video camera ever produced. This camera is based on “through-silicon-via” technology
whereby the electronics pass vertically through the sensor, permitting smaller diameter devices. This prototype camera will not
be commercially available in the foreseeable future.
Other
products
We
have utilized the MUSE
TM
system technological platform for the development of prototypes for other endoscopy and direct vision
products, including a device aiding colonoscopy, a device used in dental surgery and others. To date, we have not yet applied
for regulatory approvals for these devices, nor have we entered into agreements for the commercialization of these devices.
Our
strategy
Our
primary goal is to generate recurring revenues by driving sales of our MUSE
TM
system and establishing it as
the standard-of-care procedure and device for the treatment of moderate to severe GERD. We believe that we can achieve this goal
by continuing to accumulate clinical data and promote reimbursement for the procedure in the principal markets of North America,
Europe and Asia. Our strategy includes the following key elements:
Driving
MUSE
TM
sales
We intend to continue to focus on commercializing
the MUSE
TM
system in key geographies and markets. We have begun expanding our distribution network for further commercialization
in key European markets including Italy and Germany. During 2017, we anticipate additional European distribution agreements to
be implemented. In addition, we have successfully completed the technical testing for CFDA approval in China and have already
begun the necessary clinical trial in March 2017. In the US, we will continue marketing the MUSE
TM
through a direct effort
at key U.S. institutions.
Collaborating
and co-developing with established companies
We
seek to initiate co-development or licensing collaborations with leading companies which have existing marketing channels or significant
marketing power in critical geographies and sales channels.
Out-licensing
products
We
may consider plans to issue a license for various endoscopic systems which are based on owned and patent-protected technology
which has been developed by us. We continue to work to engage in agreements with companies which produce and market medical devices,
to include the production of systems for the foregoing companies which will be integrated by them in the endoscopic systems which
they produce or that we will develop or produce for them.
Developing
additional products
Additionally,
we intend to develop other products which will be based on the integrated and platform technology which we have developed to date,
including our miniaturized visualization imaging products, combined with our flexible stapling platform, similar to the MUSE
TM
system. Additional products could include a fully integrated, endoscopic platform designed for endoscopic surgical tissue dissection
or for endoscopic sleeve gastrectomy.
Substantially
all of our revenues in recent years are based on the sale of miniature cameras which we develop and manufacture. The following
data reflects our total revenue arising from the following services:
|
|
|
Revenues
|
|
|
|
|
Year Ended December 31,
|
|
|
Nine Months ended September
|
|
|
|
|
2014
|
|
|
2015
|
|
|
2016
|
|
|
|
|
(Thousands of U.S. dollars)
|
|
|
Sales of Miniature Cameras and related equipment
|
|
|
653
|
|
|
|
558
|
|
|
|
405
|
|
|
Sales of the MUSE
TM
System
|
|
|
91
|
|
|
|
66
|
|
|
|
91
|
|
|
Total
|
|
|
744
|
|
|
|
624
|
|
|
|
496
|
|
The
following data reflects our total revenue broken down by geographic region:
|
|
|
Revenues
|
|
|
|
|
Year Ended December 31,
|
|
|
Nine Months ended September
|
|
|
|
|
2014
|
|
|
2015
|
|
|
2016
|
|
|
|
|
(Thousands of U.S. dollars)
|
|
|
United States
|
|
|
460
|
|
|
|
446
|
|
|
|
337
|
|
|
Europe
|
|
|
113
|
|
|
|
84
|
|
|
|
10
|
|
|
Asia
|
|
|
134
|
|
|
|
51
|
|
|
|
5
|
|
|
Other
|
|
|
37
|
|
|
|
43
|
|
|
|
144
|
|
|
Total
|
|
|
744
|
|
|
|
624
|
|
|
|
496
|
|
Seasonality
of Business
During
the last few years we have not seen any seasonality in our sales.
Raw
Materials and Suppliers
The
main raw materials required for the assembly and production of our various products mainly include electronic components, mechanical
components, lighting components, tubes, lenses, sensors and cables, which we purchase from various suppliers and subcontractors
in Israel and around the world.
We
generally engage with our suppliers and subcontractors in routine purchase orders for the performance of specific orders of goods,
and not via long-term contracts. We are not required to provide collateral of any kind with respect to our orders, though occasionally
we have to pay some, or all, of the purchase order amount up front. The payment is usually made in various currencies as agreed
by the parties.
Though
we attempt to ensure the availability of more than one supplier for each important component in our products, the number
of suppliers engaged in the provision of miniature video sensors which are suitable for our CMOS technology products is very limited,
and therefore in some cases we engage with a single supplier, which may result in dependency on such supplier. This is the case
regarding sensors for the CMOS type technology that is produced by a single supplier in the United States. As we do not have a
contract in place with this supplier, there is no contractual commitment on the part of such supplier for any set quantity of
such sensors. The loss of our sole supplier in providing us with miniature sensors for our CMOS technology products, and our inability
or delay in finding a suitable replacement supplier, could significantly affect our business, financial condition, results of
operations and reputation.
In
general, alternative suppliers can be trained within a short period. However, we do have a small number of suppliers who the replacement
of which could be longer, due to the adjustment of their products to our needs.
Marketing
and Distribution
Company
Sales and Marketing Efforts
In
the United States, Europe and China, our commercial goals in the short term are to initiate highly selective sites for procedural
experience. We will support these physicians and hospitals through a number of activities, including: marketing materials to help
drive GERD patient awareness, hands-on training, animations, procedure simulator, attendance at key physician society meetings
and trade shows, training courses, reimbursement analysis, and procedural assistance.
Engagement
in External Distribution Agreements for the MUSE
TM
System
We
currently have distribution agreements with local distributors in Italy and China
for the distribution of the MUSE
TM
system, (in China the distribution arrangement is pending achievement of regulatory clearance for the MUSE
TM
system which
is to be sought by the distributor). To date, a few commercial procedures have been performed for treatment of GERD using the
MUSE
TM
system, which were provided in accordance with distribution agreements.
In
general, the distribution agreements with respect to the MUSE
TM
system are uniform and provide the following:
|
|
●
|
The
distributor serves as the exclusive distributor in the territory relevant to the agreement;
|
|
|
●
|
The
distributor must obtain all local approvals required to import and market the systems
in the relevant country;
|
|
|
●
|
A
distribution period of one to several years is determined;
|
|
|
●
|
The
distributor undertakes to market the systems in accordance with an annual plan coordinated
with us, and serves as a service center for the systems in that country; and
|
|
|
●
|
The
distributor undertakes to purchase a minimum quantity of systems throughout the preliminary
period of the agreement.
|
In
China, we have entered into an exclusive distribution agreement with Sinopharm (China National Pharmaceutical Group Corporation)
which granted the Chinese pharmaceutical company exclusive rights to distribute the MUSE
TM
system in China following its
approval by the CFDA. Under the agreement, Sinopharm has also committed to purchasing a minimum of $17.6 million worth of MUSE
TM
and related products over the course of four years, from the date of CFDA approval. The agreement also provides Sinopharm with
the discretionary right to appoint Shanghai Golden Grand-Medical Instruments Ltd., or Golden, a Chinese company specializing in
the distribution of medical devices, as its exclusive sub-distributor in China. Under the agreement, Golden will manage sales
and customer service, marketing and training, and recording of clinical trials.
Recently, we have received the approval of
the CFDA for the commencement of the first multi-center MUSE
TM
system clinical study in China. As we work to obtain CFDA
approval for MUSE
TM
, the company entered into an addendum to its current agreement with Golden, under which we paid Golden
an aggregate sum of US$175,000 to be used in financing of the efforts of the study. On March 4, 2017, we completed our first human
MUSE
TM
procedure in China.
In
the U.S., we are serving our customers directly and not through a distributor. We are currently working with key opinion leaders
at leading institutions including The Johns Hopkins Hospital, Mayo Clinic and others. In the fourth quarter of 2016, we completed
a post market study of over 70 patients designed to promote usage of the MUSE
TM
in patients with severe reflux disease. The
first publication titled ‘Interim Results from a Multi-Center Post-Marketing Surveillance Registry Study for Endoscopic
Anterior Fundoplication’ will be presented at the 2017 DDW on May 9, 2017 by Dr. Ali Lankarani from Borland Groover Clinic
in Jacksonville FL.
With
respect to our distribution agreement in Italy, on November 22, 2016, we entered into an exclusive distribution agreement with
Innovamedica S.p.A., or Innovamedica. Under the terms of the agreement, Innovamedica, a privately owned distributor of minimally
invasive medical devices, will act as our exclusive distributor in Italy for a period of four years commencing as of January 1,
2017. The agreement requires Innovamedica to purchase minimum annual quantities of our products, which are expected to amount
to at least EUR 1 million during the term of the Agreement. Innovamedica’s exclusivity is conditioned upon ordering the
minimum annual quantities.
Marketing
of Imaging Equipment
With
respect to our visualization technology, we sell and market our off-the-shelf and customized products globally and also engage
in co-development efforts when possible. We also maintain a dedicated web-site for our imaging products.
Intellectual
Property
Our
commercial success depends, in part, on obtaining and maintaining patent and other intellectual property protection, in the United
States and internationally, for the technologies used in our products. We cannot be sure that any of our patents will be commercially
useful in protecting our technology. We also rely on trade secrets to protect our product candidates. Our commercial success also
depends in part on our non-infringement of the patents or proprietary rights of third parties. The patent positions of medical
device companies, including ours, can be highly uncertain and involve complex and evolving legal and factual questions. For additional
information see “Risk Factors – Risks Related to Our Intellectual Property.”
We own 19 U.S. patents and have filed 3 additional
patent applications. In addition, we own 50 patents that were granted in other countries. We also have 12 pending patent applications
outside of U.S. and one patent application with the Patent Cooperation Treaty. Our patents, and any patents which may be granted
under our pending patent applications, expire between the years 2021 and 2036.
We
cannot be sure that any patents will be granted with respect to any of our pending patent applications or with respect to any
patent applications filed by us in the future. There is also a significant risk that any issued patents will have substantially
narrower claims than those that are currently sought.
We
also protect our proprietary technology and processes, in part, by confidentiality and invention assignment agreements with our
employees, consultants, scientific advisors and other contractors. These agreements may be breached, and we may not have adequate
remedies for any breach. We also rely on trade secrets to protect our product candidates. However, our trade secrets may otherwise
become known or be independently discovered by competitors. To the extent that our employees, consultants, scientific advisors
or other contractors use intellectual property owned by others in their work for us, disputes may arise as to the rights in related
or resulting know-how and inventions.
On
October 28, 2016, we settled all litigation and administrative proceedings with EndoChoice, Inc., or EndoChoice, including those
actions pending in the U.S. District Court for the District of Delaware C.A. Nos. 15-505-LPS-CJB and C.A. No. 15-1215-LPS-CJB
and the trademark opposition proceedings in the State of Israel involving Trademark Application Nos. 257172, 260433 and 262423.
Under the terms of the confidential settlement, we were granted a covenant not to sue with respect to EndoChoice FUSE-related
trademarks and EndoChoice was granted a non-exclusive license to our U.S. Patent No. 6,997,871 and related patents. Each party
has agreed to bear its own costs and fees associated with the litigation.
Competition
The
rapidly changing market for the treatment of GERD, which is comprised of pharmaceutical products, surgical procedures, medical
devices and potential other treatments, can be significantly affected by new product introductions and other market activities
of industry participants. We believe that the principal competitive factors in our market include:
|
|
●
|
safety,
efficacy and clinically effective performance of products
;
|
|
|
●
|
product
benefits, including the ability to offer users (both physicians and patients) a solution
for treatment of GERD using
endoscopic
-based
methods or other;
|
|
|
●
|
ease
of use and comfort for the physician and patient;
|
|
|
●
|
the
cost of product offerings and the availability of product coverage and reimbursement
from third-party payors, insurance companies and other parties;
|
|
|
●
|
the
strength
of acceptance and adoption by physicians
and hospitals;
|
|
|
●
|
the
ability to deliver new product offerings and enhanced technology to expand or improve
upon existing applications
through
continued
research and development;
|
|
|
●
|
the
quality
of training, services and clinical
support provided to physicians and hospitals;
|
|
|
●
|
effective
sales, marketing and distribution;
|
|
|
●
|
the
ability
to provide proprietary products
protected by strong intellectual property rights; and
|
|
|
●
|
the
ability
to offer products that are intuitive
and easy to learn and use.
|
Competition
to the MUSE
TM
System
We
have several competitors in the medical device and pharmaceutical industries. Patients and physicians may opt for more established
existing therapies to treat GERD, including PPI pharmaceutical treatment or laparoscopic fundoplication surgery. PPIs are currently
being offered by several large pharmaceutical manufacturers, most of whom have significantly greater financial, clinical, manufacturing,
marketing, distribution and technical resources and experience than we have.
Over
the last few years a number of different medical devices and treatments have been introduced to address the “treatment gap”
in GERD treatments and therapies which is found between long-term pharmaceutical therapy on one hand and surgery on the other.
These devices and treatments seek to treat GERD less invasively than fundoplication and without the need for long-term use of
drug therapy, and include the following options that, to our knowledge, are currently commercialized:
|
|
●
|
EsophyX
TM
Transoral
Incisionless Fundoplication (EndoGastric Solutions) — a device to endoscopically
replicate a partial fundoplication.
|
|
|
●
|
LINX
TM
Reflux
Management System (Torax Medical) — an implantable magnetic mechanical collar around
the
LES
.
|
|
|
●
|
Stretta
TM
(Mederi Therapeutics) — a catheter to deliver radiofrequency energy to the
lower esophageal sphincter to stimulate collagen deposition (scarring) in the LES.
|
|
|
●
|
GERDX
(G-Surg) – An endoscopic plication device for the LES.
|
Due
to the fact that the market is broad, it is also possible that there are additional companies who are working on the development
of endoscopic devices for the treatment of GERD. However, we do not have any indications concerning any commercial product or
product approaching commercialization, beyond information published publicly, from time to time, in medical journals and databases
of the FDA.
In
addition, new companies have been, and are likely to continue to be, formed to pursue opportunities in our market. For example,
EndoStim Inc. is a medical device company focused on the development and commercialization of a neurostimulation system for the
treatment of GERD, via an implant which includes electrodes which stimulates the LES through a permanent electrical current, and
is intended for the treatment of GERD. The Endostim device is implanted in the body by means of a laparoscopic surgery.
There
is also a relatively new surgical technology without the use of a dedicated device, called Anti-Reflux Mucosectomy to treat GERD.
Competition
to Miniature Video Cameras
The
main devices that compete with our miniature cameras are manufactured by Awaiba, Fujikura, MicroCam (Sanovas), Precision Optics,
and Opcom. The miniature cameras of each of these vendors differ in various factors include image quality and resolution, camera
shape and dimensions, sensor technology, optic characteristics, and user flexibility/customization.
Property
and Infrastructure
Our
offices and main research and development facility are located at Omer Industrial Park, No. 7A, P.O. Box 3030, Omer 8496500 Israel,
where we occupy approximately 902 square meters. We lease our facilities and our lease ends on December 31, 2017.
Legal Proceedings
From time to time we may assert or be subject
to various asserted or unasserted legal proceedings and claims. Any such claims, regardless of merit, could be time-consuming
and expensive to defend and could divert management’s attention and resources from our operations. While management
believes we have adequate insurance coverage and we accrue loss contingencies for all known matters that are probable and can
be reasonably estimated, we cannot assure that the outcome of all current or future litigation will not have a material adverse
effect on us and our results of operations.
On March 30, 2016, a former secretary and
internal legal advisor filed a lawsuit against us and our chief executive officer. The lawsuit notes several claims, among which
is that the employee was wrongfully terminated. Based on the assessment of our legal counsel we are of the opinion that
the lawsuit should not have a material unfavorable effect on our results of operations.
On October 6 2016, following a withholding
tax audit in Israel, we are deemed to be in debt of approximately $1.45 million of additional withholding taxes, including penalties
and interest. It is our management’s opinion, based on the assessment of our legal counsel, that the chances for the claims
being dismissed are more likely than not.
Government
Regulation
The
healthcare industry, and thus our business, is subject to extensive federal, state, local and foreign regulation. Some of the
pertinent laws have not been definitively interpreted by the regulatory authorities or the courts, and their provisions are open
to a variety of interpretations. In addition, these laws and their interpretations are subject to change.
Both
federal and state governmental agencies continue to subject the healthcare industry to intense regulatory scrutiny, including
heightened civil and criminal enforcement efforts. As indicated by work plans and reports issued by these agencies, the federal
government will continue to scrutinize, among other things, the billing practices of healthcare providers and the marketing of
healthcare products.
We
believe that we have structured our business operations and relationships with our customers to comply with all applicable legal
requirements. However, it is possible that governmental entities or other third parties could interpret these laws differently
and assert otherwise. In addition, because there is a risk that our products are used off label, we believe we are subject to
increased risk of prosecution under these laws and by these entities even if we believe we are acting appropriately. We discuss
below the statutes and regulations that are most relevant to our business and most frequently cited in enforcement actions.
U.S.
Food and Drug Administration
All
of our products sold in the U.S. are subject to regulation as medical devices under the FDA, as implemented and enforced by the
FDA. The FDA governs the following activities that we perform or that are performed on our behalf, to ensure that medical products
we manufacture, promote and distribute domestically or export internationally are safe and effective for their intended uses:
|
|
●
|
product
design, preclinical and clinical development and manufacture;
|
|
|
●
|
product
premarket clearance and approval;
|
|
|
●
|
product
safety, testing, labeling and storage;
|
|
|
●
|
record
keeping procedures;
|
|
|
●
|
product
marketing, sales and distribution; and
|
|
|
●
|
post-marketing
surveillance, complaint handling, medical device reporting, reporting of deaths, serious
injuries or device malfunctions and repair or recall of products.
|
FDA
Premarket Clearance and Approval Requirements
Unless
an exemption applies, each medical device we wish to commercially distribute in the United States will require either premarket
notification, or 510(k) marketing clearance or approval of a premarket approval application, or PMA, from the FDA. The FDA
classifies medical devices into one of three classes. Class I devices, considered to have the lowest risk, are those for which
safety and effectiveness can be assured by adherence to the FDA’s general regulatory controls for medical devices, which
include compliance with the applicable portions of the Quality System Regulation, facility registration and product listing, reporting
of adverse medical events, and appropriate, truthful and non-misleading labeling, advertising, and promotional materials (General
Controls). Class II devices are subject to the FDA’s General Controls, and any other special controls as deemed necessary
by the FDA to ensure the safety and effectiveness of the device (Special Controls). Manufacturers of most class II and some class
I devices are required to submit to the FDA a premarket notification under Section 510(k) of the FDCA requesting permission to
commercially distribute the device. This process is generally known as 510(k) marketing clearance. Devices deemed by the FDA to
pose the greatest risks, such as life sustaining, life supporting or some implantable devices, or devices that have a new intended
use, or use advanced technology that is not substantially equivalent to that of a legally marketed device, are placed in class
III, requiring approval of a PMA.
510(k)
Marketing Clearance Pathway
To
obtain 510(k) marketing clearance, we must submit a premarket notification demonstrating that the proposed device is “substantially
equivalent” to a legally marketed “predicate device” that is either in class I or class II, or to a class III
device that was in commercial distribution before May 28, 1976 for which the FDA has not yet called for the submission of a PMA.
A Special 510(k) is an abbreviated 510(k) application which can be used to obtain clearance for certain types of device modification
such as modifications that do not affect the intended use of the device or alter the device’s fundamental scientific technology.
A Special 510(k) generally requires less information and data than a complete, or Traditional 510(k). In addition, a Special 510(k)
application often takes a shorter period of time, which could be as short as 30 days, than a Traditional 510(k) marketing clearance
application, which can be used for any type of 510(k) device. The FDA’s 510(k) marketing clearance pathway usually takes
from three to twelve months, but may take significantly longer. The FDA may require additional information, including clinical
data, to make a determination regarding substantial equivalence. There is no guarantee that the FDA will grant 510(k) marketing
clearance for our future products and failure to obtain necessary clearances for our future products would adversely affect our
ability to grow our business.
The
FDA is currently considering proposals to reform its 510(k) marketing clearance process and such proposals could include increased
requirements for clinical data and a longer review period. In response to industry and healthcare provider concerns regarding
the predictability, consistency and rigor of the 510(k) regulatory pathway, the FDA initiated an evaluation of the 510(k) program,
and in January 2011, announced several proposed actions intended to reform the review process governing the clearance of medical
devices. The FDA intends these reform actions to improve the efficiency and transparency of the clearance process, as well as
bolster patient safety. For example, in July 2011, the FDA issued a draft guidance document entitled “510(k) Device Modifications:
Deciding When to Submit a 510(k) for a Change to an Existing Device,” which was intended to assist manufacturers in deciding
whether to submit a new 510(k) for changes or modifications made to the manufacturer’s previously cleared device. While
this draft guidance was subsequently withdrawn, the FDA is expected to replace the 1997 guidance document on the same topic. As
part of FDASIA, Congress reauthorized the Medical Device User Fee Amendments with various FDA performance goal commitments and
enacted several “Medical Device Regulatory Improvements” and miscellaneous reforms which are further intended to clarify
and improve medical device regulation both pre- and post-approval. One of these provisions obligates the FDA to prepare a report
for Congress on the FDA’s approach for determining when a new 510(k) will be required for modifications or changes to a
previously cleared device. After submitting this report, the FDA is expected to issue revised guidance to assist device manufacturers
in making this determination. Until then, manufacturers may continue to adhere to the FDA’s 1997 guidance on this topic
when making a determination as to whether or not a new 510(k) is required for a change or modification to a device, but the practical
impact of the FDA’s continuing scrutiny of these issues remains unclear. It is possible that any new guidance will make
substantive changes to existing policy and practice regarding the assessment of whether a new 510(k) is required for changes or
modifications to existing devices. Specifically, industry has interpreted the withdrawn draft guidance to take a more conservative
approach in requiring a new 510(k) for certain changes or modifications to existing, cleared devices that might not have triggered
a new 510(k) under the 1997 guidance. As of July 28, 2014, the FDA released final guidance entitled “The 510(k) Program:
Evaluating Substantial Equivalence in Premarket Notifications” which is intended to identify, explain, and clarify each
of the critical decision points in the decision-making process FDA uses to determine substantial equivalence. We cannot predict
which of the 510(k) marketing clearance reforms currently being discussed or proposed might be enacted, finalized or implemented
by the FDA and whether the FDA will propose additional modifications to the regulations governing medical devices in the future.
Any such modification could have a material adverse effect on our ability to commercialize our products.
Medical
devices can be marketed only for the indications for which they are cleared or approved. After a device receives 510(k) marketing
clearance, any modification that could significantly affect its safety or effectiveness, or that would constitute a new or major
change in its intended use, will require a new 510(k) marketing clearance or, depending on the modification, PMA approval. The
FDA requires each manufacturer to determine whether the proposed changes requires submission of a 510(k) or a PMA, but the FDA
can review any such decision and can disagree with a manufacturer’s determination. If the FDA disagrees with a manufacturer’s
determination, the FDA can require the manufacturer to cease marketing or recall the modified device until 510(k) marketing clearance
or PMA approval is obtained. Also, in these circumstances, we may be subject to significant regulatory fines or penalties. We
have made, and plan to continue to make, additional product enhancements to MUSE
TM
system and other products that we believe
do not require new 510(k) marketing clearances. We cannot be assured that the FDA would agree with any of our decisions not to
seek 510(k) marketing clearance or PMA approval. For risks related to 510(k) marketing clearance, see “Risk Factors –
Risks Related to Regulatory Compliance.”
PMA
Approval Pathway
A
PMA must be submitted to the FDA if the device cannot be cleared through the 510(k) process or is not otherwise exempt from the
FDA’s premarket clearance and approval requirements. A PMA must generally be supported by extensive data, including, but
not limited to, technical, preclinical, clinical trials, manufacturing and labeling, to demonstrate to the FDA’s satisfaction
the safety and effectiveness of the device for its intended use. Also, an advisory panel of experts from outside the FDA may be
convened to review and evaluate the application and provide recommendations to the FDA as to the approvability of the device.
The FDA may or may not accept the panel’s recommendation. In addition, the FDA will generally conduct a pre-approval inspection
of the applicant or its third-party manufacturers’ or suppliers’ manufacturing facility or facilities to ensure compliance
with the QSR.
New
PMAs or PMA supplements are required for modifications that affect the safety or effectiveness of the device, including, for example,
certain types of modifications to the device’s indication for use, manufacturing process, labeling and design. PMA supplements
often require submission of the same type of information as a PMA, except that the supplement is limited to information needed
to support any changes from the device covered by the original PMA and may not require as extensive clinical data or the convening
of an advisory panel. None of our products are currently approved under a PMA approval. However, we may in the future develop
devices which will require the approval of a PMA. There is no guarantee that the FDA will grant PMA approval of our future products
and failure to obtain necessary approvals for our future products would adversely affect our ability to grow our business.
C
linical
Trials
Clinical
trials are generally required to support a PMA application and are sometimes required for 510(k) marketing clearance. Such trials
generally require an Investigational Device Exemption application, or IDE, approved in advance by the FDA for a specified number
of patients and study sites, unless the product is deemed a non-significant risk device eligible for more abbreviated IDE requirements.
A significant risk device is one that presents a potential for serious risk to the health, safety or welfare of a patient and
either is implanted, used in supporting or sustaining human life, substantially important in diagnosing, curing, mitigating or
treating disease or otherwise preventing impairment of human health, or otherwise presents a potential for serious risk to a subject.
Clinical trials are subject to extensive monitoring, recordkeeping and reporting requirements. Clinical trials must be conducted
under the oversight of an Institutional Review Board, or IRB, for the relevant clinical trial sites and must comply with FDA regulations,
including but not limited to those relating to good clinical practices. To conduct a clinical trial, we are also required to obtain
the patient’s informed consent in form and substance that complies with both FDA requirements and state and federal privacy
and human subject protection regulations. We, the FDA or the IRB could suspend a clinical trial at any time for various reasons,
including a belief that the risks to study subjects outweigh the anticipated benefits. Even if a trial is completed, the results
of clinical testing may not adequately demonstrate the safety and efficacy of the device or may otherwise not be sufficient to
obtain FDA clearance or approval to market the product in the United States. Similarly, in Europe the clinical study must
be approved by a local ethics committee and in some cases, including studies with high-risk devices, by the ministry of health
in the applicable country.
Pervasive
and Continuing Regulation
After
a device is placed on the market, numerous regulatory requirements continue to apply. In addition to the requirements below, the
Medical Device Reporting, or MDR, regulations require that we report to the FDA any incident in which our products may have caused
or contributed to a death or serious injury or in which our product malfunctioned and, if the malfunction were to recur, would
likely cause or contribute to death or serious injury. See “Risk Factors – Risks Related to Regulatory Compliance,”
for further information regarding our reporting obligations under MDR regulations. Additional regulatory requirements include:
|
|
●
|
product listing and establishment registration, which helps facilitate FDA inspections and other regulatory action;
|
|
|
|
|
|
|
●
|
QSR, which requires manufacturers, including third-party manufacturers, to follow stringent design, testing, control, documentation and other quality assurance procedures during all phases of the design and manufacturing process;
|
|
|
|
|
|
|
●
|
labeling regulations and FDA prohibitions against the promotion of products for uncleared, unapproved or off-label use or indication;
|
|
|
|
|
|
|
●
|
clearance of product modifications that could significantly affect safety or efficacy or that would constitute a major change in intended use of one of our cleared devices;
|
|
|
|
|
|
|
●
|
approval of product modifications that affect the safety or effectiveness of one of our approved devices;
|
|
|
|
|
|
|
●
|
post-approval restrictions or conditions, including post-approval study commitments;
|
|
|
|
|
|
|
●
|
post-market surveillance regulations, which apply, when necessary, to protect the public health or to provide additional safety and effectiveness data for the device;
|
|
|
|
|
|
|
●
|
the FDA’s recall authority, whereby it can ask, or under certain conditions order, device manufacturers to recall from the market a product that is in violation of governing laws and regulations; and
|
|
|
|
|
|
|
●
|
notices of corrections or removals.
|
We
must also register with the FDA as a medical device manufacturer and must obtain all necessary state permits or licenses to operate
our business.
Failure
to comply with applicable regulatory requirements, including delays in or failures to report incidents to the FDA as required
under the MDR regulations, can result in enforcement action by the FDA, which may include any of the following sanctions:
|
|
●
|
warning letters, fines, injunctions, consent decrees and civil penalties;
|
|
|
|
|
|
|
●
|
customer notifications or repair, replacement, refunds, recall, detention or seizure of our products;
|
|
|
|
|
|
|
●
|
operating restrictions or partial suspension or total shutdown of production;
|
|
|
|
|
|
|
●
|
refusing or delaying requests for 510(k) marketing clearance or PMA approvals of new products or modified products;
|
|
|
|
|
|
|
●
|
withdrawing 510(k) marketing clearances or PMA approvals that have already been granted;
|
|
|
|
|
|
|
●
|
refusal to grant export approval for our products; or
|
|
|
|
|
|
|
●
|
criminal prosecution.
|
In
January 2016, we performed an FDA mock audit by an FDA veteran specialist, following which we implemented improvements in our
quality management system. We cannot be assured that we have adequately complied with all regulatory requirements or that one
or more of the referenced sanctions will not be applied to us as a result of a failure to comply.
Marketing
Approvals Outside the United States
Sales
of medical devices outside the United States are subject to foreign government regulations, which vary substantially from country
to country. The time required to obtain approval by a foreign country may be longer or shorter than that required for FDA clearance
or approval, and the requirements may differ.
The
European Union has adopted numerous directives and standards regulating the design, manufacture, clinical trials, labeling and
adverse event reporting for medical devices. Each European Union member state has implemented legislation applying these directives
and standards at the national level. Other countries, such as Switzerland, have voluntarily adopted laws and regulations that
mirror those of the European Union with respect to medical devices. Devices that comply with the requirements of the laws of the
relevant member state applying the applicable European Union directive are entitled to bear CE conformity marking and, accordingly,
can be commercially distributed throughout the member states of the European Union and other countries that comply with or mirror
these directives. The method of assessing conformity varies depending on the type and class of the product, but normally involves
a combination of self-assessment by the manufacturer and a third-party assessment by a “Notified Body,” an independent
and neutral institution appointed to conduct conformity assessment. This third-party assessment consists of an audit of the manufacturer’s
quality system and clinical information, as well as technical review of the manufacturer’s product. An assessment by a Notified
Body in one country within the European Union is required in order for a manufacturer to commercially distribute the product throughout
the European Union. In addition, compliance with ISO 13845 on quality systems issued by the International Organization for Standards,
among other standards, establishes the presumption of conformity with the essential requirements for a CE marking. In addition,
many countries apply requirements in their reimbursement, pricing or health care systems that affect companies’ ability
to market products.
We
have been authorized by Health Canada and have received AMAR approval in Israel. In addition, we received approval form the MedCert
Zertifizierungs und Prufungsgsesellschaft fur die Medizin GmbH of Germany, and are entitled to print the CE Mark on our products,
after having examined the EU Technical File for each new product.
Health
Care Laws and Regulations
Reimbursement
In
the United States and elsewhere, health care providers that perform surgical procedures using medical devices such as ours
generally rely on third-party payors, including governmental payors such as Medicare and Medicaid and private payors, to cover
and reimburse the associated medical and surgical costs. Consequently, sales of medical devices are dependent in part on the availability
of reimbursement to the customer from third-party payors. The manner in which reimbursement is sought and obtained varies based
upon the type of payor involved and the setting in which the product is furnished and utilized. In general, third-party payors
will provide coverage and reimbursement for medically reasonable and necessary procedures and tests that utilize medical devices
and may provide separate payments for the implanted or disposable devices themselves. Most payors, however, will not pay separately
for capital equipment. Instead, payment for the cost of using the capital equipment is considered to be covered as part of payments
received for performing the procedure. In determining payment rates, third-party payors are increasingly scrutinizing the prices
charged for medical products and services in comparison to other therapies. The procedures in which our products are used may
not be reimbursed by these third-party payors at rates sufficient to allow us to sell our products on a competitive and profitable
basis.
In
addition, in many foreign markets, including the countries in the European Union, pricing of medical devices is subject to governmental
control. In the United States, there have been, and we expect that there will continue to be, a number of federal and state proposals
to limit payments by governmental payors for medical devices, and the procedures in which medical devices are used.
In
March 2010, comprehensive health care reform legislation was enacted through the passage of PPACA, also known as the Affordable
Care Act. Significant measures contained in the health care reform legislation include initiatives to revise Medicare payment
methodologies, initiatives to promote quality indicators in payment methodologies (including the bundling of hospital and physician
payments), initiatives related to the coordination and promotion of research on comparative clinical effectiveness of different
technologies and procedures, and annual reporting requirements related to payments to physicians and teaching hospitals. At this
time it is not possible to predict whether these initiatives will have a positive or negative impact on us. The health care reform
legislation also includes new taxes impacting certain health-related industries, including medical device manufacturers. As of
2013, each medical device manufacturer or importer has to pay an excise tax (or sales tax) in an amount equal to 2.3% of the price
for which such manufacturer sells its medical devices. In addition to the health care reform legislation, various healthcare reform
proposals have also emerged at the state level. We cannot predict whether future healthcare initiatives will be implemented at
the federal or state level or internationally, or the effect any future legislation or regulation will have on us. The taxes imposed
by the health care reform legislation and the expansion in government’s role in the U.S. healthcare industry may result
in decreased profits to us, lower reimbursements by payors for our products, and reduced medical procedure volumes, all of which
may adversely affect our business, financial condition and results of operations, possibly materially.
Medicare
and Medicaid
The Medicare program is a federal health
benefit program administered by the CMS that covers and pays for certain medical care items and services for eligible elderly
persons. The Medicaid program is a federal-state partnership under which states receive matching federal payments to fund healthcare
services for the poor.
In January 2016, the American Medical Association’s (AMA) Current
Procedural Terminology (CPT) published a new Category I CPT Code for transoral esophagogastric fundoplasty procedures, which describes
procedures conducted with the MUSE
TM
system. In the U.S., the CPT Editorial Panel assigns specific billing codes for physician
services and outpatient hospital procedures, which are used by providers, who are our customers, to bill for procedures. Once
a CPT code is established, the Centers for Medicare and Medicaid Services (CMS) in turn establishes payment levels and
coverage rules under Medicare, and private payors establish rates and coverage rules. Notwithstanding the issuance of a CPT to
report the MUSE procedure and the establishment of payment rates for the code, we cannot guarantee that the MUSE
TM
system
is or will be covered and, if covered, that reimbursement will be sufficient, and furthermore, we cannot guarantee that the MUSE
TM
system or any future product will be approved for coverage or reimbursement by Medicare, Medicaid or any third-party payor.
Reimbursement decisions in the European Union and in other jurisdictions outside of the United States vary by country and region
and there can be no assurance that we will be successful in obtaining adequate reimbursement
.
Commercial
Insurers
Many
private payors look to CMS policies as a guideline in setting their coverage policies and payment amounts. A decrease of, or limitation
on, reimbursement payments for physicians and hospitals by CMS or other agencies may affect coverage and reimbursement determinations
by many private payors. Additionally, some private payors may reimburse only a portion of the costs associated with the use of
our products, or not at all.
Fraud
and Abuse Laws
Because
of the significant federal funding involved in Medicare and Medicaid, Congress and the states have enacted, and actively enforce,
a number of laws whose purpose is to eliminate fraud and abuse in federal health care programs. Our business is subject to compliance
with these laws.
Anti-Kickback
Statutes and Federal False Claims Act
The
federal healthcare programs’ Anti-Kickback Statute prohibits persons from soliciting, offering, receiving or providing remuneration,
directly or indirectly, in exchange for or to induce either the referral of an individual, or the furnishing or arranging for
a good or service, for which payment may be made under a federal healthcare program such as Medicare or Medicaid. Penalties for
violations include criminal penalties and civil sanctions such as fines, imprisonment and possible exclusion from Medicare, Medicaid
and other federal healthcare programs. The Anti-Kickback Statute is broad and prohibits many arrangements and practices that are
lawful in businesses outside of the healthcare industry. There are a number of statutory exceptions as well as regulatory safe
harbors protecting certain common activities from prosecution or other regulatory sanctions, however, the exceptions and safe
harbors are drawn narrowly, and practices that do not fit squarely within an exception or safe harbor may be subject to scrutiny.
Many states have adopted laws similar to the Anti-Kickback Statute. Some of these state prohibitions apply to referral of patients
for healthcare items or services reimbursed by any source, not only the Medicare and Medicaid programs.
Government
officials have focused their enforcement efforts on marketing of healthcare services and products, among other activities, and
recently have brought cases against companies, and certain sales, marketing and executive personnel, for allegedly offering unlawful
inducements to potential or existing customers in an attempt to procure their business. Another development affecting the healthcare
industry is the increased use of the federal Civil False Claims Act and, in particular, actions brought pursuant to the False
Claims Act’s “whistleblower” or “qui tam” provisions. The federal civil False Claims Act prohibits,
among other things, individuals or entities from knowingly presenting, or causing to be presented, a false or fraudulent claim
for payment of government funds or knowingly making, using or causing to be made or used, a false record or statement material
to an obligation to pay money to the government or knowingly concealing or knowing and improperly avoiding, decreasing or concealing
an obligation to pay money to the federal government. Violation of the False Claims Act can result in significant civil and administrative
penalties, up to treble damages and exclusion from participation in federal health care programs like Medicare and Medicaid. The
False Claims Act also allows a private individual or entity to sue on behalf of the government. Medical device manufacturers and
other health care companies have been investigated by the U.S. Department of Justice and have reached substantial financial settlements
with the federal government under the civil False Claims Act for a variety of alleged improper marketing activities, including
providing free product, providing consulting fees, grants, free travel and other benefits to physicians to induce them to prescribe
the company’s products, and for causing false claims to be submitted as a result of the marketing of their products for
unapproved, and thus non-reimbursable, uses. Resolution of such investigations has often included manufacturers entering into
corporate integrity agreements with the Office of Inspector General for the U.S. Department of Health and Human Services that
require, among other things, substantial reporting and remedial actions.
Additionally,
several bills have been passed or are pending, at both the state and federal levels that expand the anti-kickback laws to require,
among other things, extensive tracking and maintenance of databases regarding relationships to physicians and healthcare providers.
The PPACA imposes new reporting and disclosure requirements on device manufacturers for any “transfer of value” made
or distributed to physicians and teaching hospitals, otherwise known as the Physician Payment Sunshine Act. Device manufacturers
were required to begin collecting data on August 1, 2013 and were be required to submit reports to CMS by March 31, 2014 (and
the 90th day of each subsequent calendar year). In addition, there has been a recent trend of increased federal and state regulation
of payments made to physicians. Some states, such as California, Massachusetts and Nevada, mandate implementation of commercial
compliance programs, while certain states, such as Massachusetts and Vermont, impose restrictions on device manufacturer marketing
practices and tracking and reporting of gifts, compensation and other remuneration to physicians. The implementation of the infrastructure
to comply with these bills and regulations could be costly and any failure to provide the required information may result in civil
monetary penalties.
We
believe our current consulting agreements with physicians represent legitimate compensation for needed documented services actually
furnished to us
.
However, engagement of physician consultants by medical device manufacturers has recently been subject
to heightened scrutiny. In this environment, our engagement of physician consultants in product development or clinical testing
could subject us to similar scrutiny. We are unable to predict whether we would be subject to actions under the Anti-Kickback
Statute or False Claims Act or any similar state law, or the impact of such actions.
HIPAA
and Other Fraud and Privacy Regulations
Among
other things, HIPAA created two new federal crimes: healthcare fraud and false statements relating to healthcare matters. The
HIPAA health care fraud statute prohibits, among other things, knowingly and willfully executing, or attempting to execute, a
scheme to defraud any healthcare benefit program, including private payors. A violation of this statute is a felony and may result
in fines, imprisonment and exclusion from government sponsored programs. The HIPAA false statements statute prohibits, among other
things, knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious
or fraudulent statement or representation in connection with the delivery of or payment for healthcare benefits, items or services.
A violation of this statute is a felony and may result in fines and imprisonment.
In
addition to federal regulations issued under HIPAA, some states have enacted privacy and security statutes or regulations that,
in some cases, are more stringent than those issued under HIPAA. In those cases, it may be necessary to modify our planned operations
and procedures to comply with the more stringent state laws. If we fail to comply with applicable state laws and regulations,
we could be subject to additional sanctions.
Anti-Bribery
Laws
Compliance
with complex foreign and U.S. laws and regulations that apply to our international operations increases our cost of doing business
in international jurisdictions and could expose us or our employees to fines and penalties in the U.S. and abroad. These numerous
and sometimes conflicting laws and regulations include the FCPA. The FCPA prohibits U.S. companies, companies whose securities
are listed for trading in the United States and other entities, and their officers, directors, employees, shareholders acting
on their behalf and agents from offering, promising, authorizing or making payments to foreign officials for the purpose of obtaining
or retaining business abroad or otherwise obtaining favorable treatment. The FCPA also requires companies to maintain records
that fairly and accurately reflect transactions and maintain internal accounting controls. In many countries, hospitals are government-owned
and healthcare professionals employed by such hospitals, with whom we regularly interact, may meet the definition of a foreign
official for purposes of the FCPA. Additionally, recently enacted U.S. legislation increases the monetary reward available to
whistleblowers who report violations of federal securities laws, including the FCPA, which may result in increased scrutiny and
allegations of violations of these laws and regulations. Violations of these laws and regulations could result in fines, criminal
sanctions against us, our officers, or our employees, prohibitions on the conduct of our business, and damage to our reputation.
Israeli
Government Programs
Under the
Encouragement
of Research, Development and Technological Innovation in the Industry Law
, 5744-1984, or the Innovation Law, research and
development programs which meet specified criteria and are approved by a committee of the Office of Chief Scientist of the Israeli
Ministry of Economy (formerly named the Ministry of Industry, Trade and Labor, or the OCS, are eligible for grants from the OCS.
The grant amounts are determined by the research committee, and are typically a percentage of the project’s expenditures.
Under most programs, the grantee is required to pay royalties to the State of Israel from the sale of products developed under
the program. Regulations under the Innovation Law generally provide for the payment of royalties of 3% to 6% on sales of products
and services based on or incorporating technology developed using grants or know-how deriving therefrom, up to 100% of the grant,
linked to the dollar and bearing interest at the LIBOR rate, is repaid. The royalty rates and the aggregate repayment amount may
be higher if manufacturing rights are transferred outside of Israel, as further detailed below. The manufacturing rights of products
incorporating technology developed thereunder may not be transferred outside of Israel, unless approval is received from the OCS
and additional royalty payments are made to the State of Israel, as further detailed below. However, this does not restrict the
export of products that incorporate the funded technology.
The pertinent
obligations under the Innovation Law are as follows:
|
|
●
|
Local
Manufacturing Obligation
. The terms of the grants under the Innovation Law require
that we manufacture the products developed with these grants in Israel. Under the regulations
promulgated under the Innovation Law, the products may be manufactured outside Israel
by us or by another entity only if prior approval is received from the OCS (such approval
is not required for the transfer of less than 10% of the manufacturing capacity in the
aggregate, in which case a notice should be provided to the OCS). As a condition to obtaining
approval to manufacture outside Israel, we would be required to pay royalties at an increased
rate (usually 1% in addition to the standard rate and increased royalties cap (between
120% and 300% of the grants, depending on the manufacturing volume that is performed
outside Israel). We note that a company also has the option of declaring in its OCS grant
application an intention to exercise a portion of the manufacturing capacity abroad,
thus avoiding the need to obtain additional approvals and pay the increased royalties
cap.
|
|
|
●
|
Know-How
transfer limitation
. The Innovation Law restricts the ability to transfer know-how
funded by the OCS outside of Israel. Transfer of OCS funded know-how outside of Israel
requires prior OCS approval and in certain circumstances is subject to certain payment
to the OCS calculated according to formulae provided under the Innovation Law. If we
wish to transfer OCS funded know-how, the terms for approval will be determined according
to the character of the transaction and the consideration paid to us for such transfer.
The OCS approval to transfer know-how created, in whole or in part, in connection with
an OCS-funded project to third party outside Israel where the transferring company remains
an operating Israeli entity is subject to payment of a redemption fee to the OCS calculated
according to a formula provided under the Innovation Law that is based, in general, on
the ratio between the aggregate OCS grants to the company’s aggregate investments
in the project that was funded by these OCS grants, multiplied by the transaction consideration,
considering depreciation mechanism and less royalties already paid to the OCS. The transfer
of such know-how to a party outside Israel where the transferring company ceases to exist
as an Israeli entity is subject to a redemption fee formula that is based, in general,
on the ratio between aggregate OCS grants received by the company and the company’s
aggregate research and development expenses, multiplied by the transaction consideration
considering depreciation mechanism and less royalties already paid to the OCS. The regulations
promulgated under the Innovation Law establish a maximum payment of the redemption fee
paid to the OCS under the above mentioned formulas and differentiates between two situations:
(i) in the event that the company sells its OCS funded know-how, in whole or in part,
or is sold as part of an M&A transaction, and subsequently ceases to conduct business
in Israel, the maximum redemption fee under the above mentioned formulas will be no more
than six times the amount received (plus annual interest) for the applicable know-how
being transferred, or the entire amount received from the OCS, as applicable; (ii) in
the event that following the transactions described above (i.e. asset sale of OCS funded
know-how or transfer as part of an M&A transaction) the company continues to conduct
its research and development activity in Israel (for at least three years following such
transfer and maintain staff of at least 75% of the number of research and development
employees it had for the six months before the know-how was transferred and keeps the
same scope of employment for such research and development staff), then the company is
eligible for a reduced cap of the redemption fee of no more than three times the amounts
received (plus annual interest) for the applicable know-how being transferred, or the
entire amount received from the OCS, as applicable.
|
|
|
|
|
|
|
●
|
Approval
of the transfer of OCS funded technology to another Israeli company may be granted only
if the recipient abides by the provisions of the Innovation law and related regulations,
including the restrictions on the transfer of know-how and manufacturing rights outside
of Israel (note that there will be an obligation to pay royalties to the OCS from the
income of such sale transaction as part of the royalty payment obligation).
|
Approval
to manufacture products outside of Israel or consent to the transfer of technology, if requested, might not be granted. Furthermore,
the OCS may impose certain conditions on any arrangement under which it permits us to transfer technology or development out of
Israel.
The
Innovation Law was amended as of July 29, 2015, or the 2015 Amendment. On January 1, 2016, pursuant to the 2015 Amendment, the
National Authority for Technological Innovation, or NATI, was established and in June 2016, NATI was fully constituted. Pursuant
to the 2015 Amendment, NATI is authorized to change the current restrictions imposed on the recipients of grants under the Innovation
Law with a new set of arrangements in connection with ownership obligations of know-how (including with respect to restrictions
on transfer of know-how and manufacturing activities outside of Israel), as well as royalties obligations associated with approved
programs. The Innovation Law as existed prior to the 2015 Amendment will continue to be in effect with respect to
research
and development
programs which were in affect prior to January 1, 2016 until: (i) June 2017,
which is one year following the date of appointment of all members of the NATI council; or (ii) earlier, if otherwise resolved
by the NATI council. The Innovation Law in its form following the 2015 Amendment also includes new provisions with respect to
sanctions imposed for violations of the Innovation Law. As of the date of this
prospectus
,
we are unable to determine whether NATI will promulgate a new set of arrangements or adopt the arrangements which were stipulated
under the Innovation Law as existed prior to the 2015 Amendment, therefore, as of the date of this
prospectus
,
we are unable as assess the effect, if any, of the promulgation of such arrangements on us.
Grants
Received from the Chief Scientist
We
have received grants from the OCS as part of our participation in two programs as described below:
Membership
in the Activities of the Bio Medical Photonic Consortium
The
Bio Medical Photonic Consortium, or the Consortium, commenced its activities in June 2007, and concluded its activities on December
31, 2012. The purpose of the Consortium was to develop generic photonic technologies in the field of diagnostics and therapeutics
in the biomedical industry in Israel, and specifically on the subject of the digestive system. The activities of the Consortium
were performed under our management and the management of Given Imaging Ltd., where each would develop technological models which
are based on their internal developments and on developments of the members of the Consortium.
Within
the framework of the activities of the Consortium, the Company worked to develop the next generation technology of miniature cameras.
The cameras were integrated, within the framework of the Consortium, in technological models for minimally invasive procedures
which were developed by the members of the Consortium. The various combinations of surgical tools and advanced visual capabilities
with miniature endoscopes are innovative, and we predict that the Consortium framework will continue serving as a fruitful basis
for the development of innovative medical procedures through the creation of intellectual property. Additionally, we will cooperate
with research groups which develop indicators for early detection of colorectal cancer, with the aim of integrating the visualization
techniques and key products in this field.
The
following are details regarding the rights and obligations within the framework of our activity in the Consortium, which continue
to apply notwithstanding the conclusion of the program:
|
|
(i)
|
The property rights to information
which has been developed belongs to the Consortium member that developed it. However, the developing entity is obligated to
provide the other members in the Consortium a license for the use of the new information, without consideration, provided
that the other members do not transfer such information to any entity which is not a member of the Consortium. The provision
of a license or of the right to use the new information to a third party is subject to approval by the administration of the
MAGNET Program at the OCS;
|
|
|
(ii)
|
We are is entitled to register
a patent for the new information which has been developed by it within the framework of its activity in the Consortium. The
foregoing registration does not require approval from the administration; and
|
|
|
(iii)
|
The know-how and technology
developed under the program is subject to the restrictions set forth under the Innovation Law, including restrictions on the
transfer of such know-how and any manufacturing rights with respect thereto, without first obtaining the approval of the OCS.
Such approval may entail additional payments to the OCS, as determined under the Innovation Law and regulations, and as further
detailed above.
|
Collaboration
Grant for the Development of a Miniature Diameter Endoscope for Use in Dental Implants
In
July 2011, the OCS approved our application for support for a joint project regarding the development of an innovative, miniature
diameter endoscopic product in the field of dental surgery, or the Dental Project. In October 2012, the Company received
a notice according to which approval was given for continued support for the Dental Project for a second year. The OCS support
for the Dental Project concluded on July 31, 2013.
The
Dental Project was performed in collaboration with Qioptiq GmbH, a German corporation, or Qioptiq, in the field of sophisticated
medical micro-optics, including in the medical and life sciences sector. The collaboration between the Company and Qioptiq was
performed within the framework of the Eureka organization, a Pan-European organization which includes approximately 40 member
states, including the State of Israel, and which acts to coordinate and to finance research and development enterprises in and
outside of Europe.
In
accordance with the outline of the Dental Project, we and Qioptiq collaborated on the development of an innovative miniature-diameter
endoscope, with side viewing capabilities, intended for use in various dental implant procedures, the Dental Endoscope. During
the Dental Project, each of the parties developed different parts of the Dental Endoscope. In accordance with the terms of the
collaboration, the intellectual property which originated from the development of the Dental Endoscope remained the exclusive
property of the party which developed it. Subject to the completion of the project, the parties agreed to conduct negotiations
regarding the method used to produce and market the Product (the foregoing negotiations have not yet been conducted and we have
notified the OCS that there are no revenues from this project).
Implantation
procedures are complex, and in many cases, damage is caused to the tissue of the mouth and the jaw due to the dentist’s
inability to see the entire operating area. The Dental Endoscope is intended to allow improved visual monitoring of the surgical
procedure using a miniature video camera which has been developed by us, which is installed on the edge of the endoscope, thereby
significantly reducing the risk to the patient. It is estimated that hundreds of thousands of procedures of the kind for which
the product is intended are performed each year in Europe. Construction of a prototype for the product concluded in November 2012.
The prototype is intended for use in pre-clinical trials and in human clinical trials.
As
of the date hereof, there is no certainty that it will be possible to produce and market the product, which may be developed or
that the regulatory approvals required for the product’s marketing will be received. At present, we and Qioptiq are not
acting to commercialize the Dental Project.
Grants
and Royalty Obligations
We
received various grants from the OCS in connection with our participation in its programs. We received a grant of approximately
$2.3 million in connection with our participation in the Bio Medical Photonics Consortium in the production of generic technology
related to the partial development of miniature or the Consortium Grant. Under the terms of the Consortium Grant we are not required
to pay royalties. In addition, we received a grant of approximately $0.2 million in connection with a collaboration within the
framework of the Eureka organization related to miniature endoscope for dental implants, or the Eureka Grant. Under the terms
of the Eureka Grant, we would have to pay royalties at a rate of 3%-5% from the actual sales of the relevant device, up to the
repayment of the grant, with the addition of interest and linkage. As of December 31, 2016, there have been no sales that requires
us to pay royalties under the Eureka Grant and we believe that the probability we could generate income from the miniature endoscope
for dental implants is low.
Management
Executive
officers, directors and director nominees
The
following table sets forth information concerning our executive officers and directors:
|
Name
|
|
Age
|
|
Position(s)
|
|
Dr.
Nissim Darvish
|
|
52
|
|
Chairman
of the Board of Directors
|
|
Christopher
(Chris) Rowland
|
|
54
|
|
Chief
Executive Officer, Director
|
|
Eitan
Machover
(1)
|
|
55
|
|
External
Director
|
|
Efrat
Venkert
(1)(2)
|
|
50
|
|
External
Director
|
|
Doron
Birger
(1)(2)
|
|
65
|
|
Director
|
|
Anat
Naschitz
(2)
|
|
49
|
|
Director
|
|
Oded
Yatzkan
|
|
50
|
|
Chief
Financial Officer
|
|
Minelu
(Menashe) Sonnenschein
|
|
51
|
|
VP
Israel Operations
|
|
Yaron
Silberman
|
|
46
|
|
VP
Sales and Marketing
|
|
Dr.
Aviel Roy Shapira
|
|
66
|
|
Medical
Director
|
|
(1)
|
Member
of audit committee and compensation committee.
|
|
(2)
|
Member
of the investment committee.
|
Dr.
Nissim Darvish
has been serving as the chairman of our Board since March 2013, and on September 29, 2016, was re-elected
for service as a director and chairman of our Board until our next annual general meeting. Dr. Darvish serves as a Senior Managing
Director in OrbiMed Advisors LLC. Dr. Darvish currently serves as a director of Ornim Medical Inc., RDD Pharma Ltd., Otic Pharma
Ltd., Tyto Care., Keystone Heart Ltd., OrbiMed Israel Partner Ltd., OrbiMed Israel Limited Partnership, Asdanit Medical Ltd.,
Asdan Medical Ltd. and Ramot the Tel Aviv University’s (TAU) Technology Transfer Company. Dr. Darvish has served as a General
Partner in Pitango VC, which focuses on life sciences investments. Dr. Darvish holds an MD and a D.Sc., Doctor of Medical Science
in Biophysics, from The Technion Institute of Technology, Israel.
Christopher
(Chris) Rowland
has been serving as a member of our Board since March 2013 and as Chief Executive Officer since October
2013. On September 29, 2016, Mr. Rowland was re-elected for service as a director until our next annual general meeting. Between
2011 and 2013, Mr. Rowland served as President of IntraPace Inc., a company that develops medical equipment in the field of obesity.
Mr. Rowland has served as President and CEO of Neo Tract Inc., a medical equipment company in the field of urology, and as President
of Americas Given Imaging Inc. Mr. Rowland holds a B.Sc. in marketing from the University of Southern Illinois. Mr. Rowland completed
the Executive Management Program of Columbia Business School, and the Executive Leadership Program of Harvard Business School.
Eitan
Machover
has been serving as an external director on our Board since September 29, 2016, and was elected to serve for a three
years term. Mr. Machover currently serves as an external director at Electra Real Estate Ltd. and Related Commercial Portfolio
Ltd., both Israeli public companies traded on the Tel-Aviv Stock Exchange Ltd. Mr. Machover also serves on the board of directors
of Epsilon Underwriting Ltd., Trackimo Ltd., Medtek Inc., VVT Medical Ltd. and MedyMatch Technology Ltd. From 2013 to 2015, Mr.
Machover served as the chief executive officer and director of Enhanced Surface Dynamics, Inc. From 2011 to 2012, Mr. Machover
served as an interim chief executive officer at Ovalum Vascular, Ltd. From 2002 to 2012, Mr. Machover served as general partner
at MediTech Partners, LLC, an advisory and investment firm specializing in medical device technologies. From 1989 to 2010, Mr.
Machover served in several positions in General Electric Company including national executive for Israel. Mr. Machover holds a
B.A. in business communications from Emerson College, Boston, Massachusetts, and an MBA in finance and marketing from Boston College.
Efrat
Venkert
has been serving as an external director on our Board since September 2013, and on September 29, 2016, was reelected
to serve for an additional three years term. Ms. Venkert is the owner of a law firm specializing in legal advice to companies
and in business law. From 2009 to 2010, Ms. Venkert served as a member of an advisory group advising to the Government Investigation
Committee for the water sector. From 2007 to 2009, Ms. Venkert served as the chairperson secretary of the Steering Committee,
and Chairperson Manager of the Ministry of Justice and Ministry of Welfare pilot project on the subject of changes in the legislation
of minors’ participation in legal proceedings and from 2004 to 2008 Ms. Venkert served as a lecturer in the Law Faculty
of the University of Haifa. Ms. Venkert holds an LL.B. from Hebrew University, Jerusalem, Israel and an MBA from Haifa University,
Haifa, Israel. Ms. Venkert is a Certified Mediator of ICNM, a graduate of the Companies Secretary Course of the Israel Management
Center, and a graduate of Directors’ Course, Lahav, Tel-Aviv University, Israel.
Doron
Birger
has been serving as a member of our Board since May 2015, and on September 29, 2016, was elected by the Board
for service as an independent director until our next annual general meeting. Mr. Birger currently serves as chairman of the board
of Insuline and as a director of MCS Medical Compression Ltd, Hadasit Bio-Holdings Ltd. and Icecure Medical Ltd, four life
sciences companies publicly traded in Israel, and as chairman and director of several private companies in Israel in the hi-tech
sector mainly in the medical device field. From 2002 to 2007, Mr. Birger served as the chairman of the board of directors of Given
Imaging Ltd and later on as board member until February 2014. Mr. Birger served as chief executive officer of Elron Electronic
Industries, Ltd., or Elron, from August 2002 to April 2009. Prior to that, he held other executive positions at Elron, including
President since 2001, Chief Financial Officer from 1994 to August 2002, and Corporate Secretary from 1994 to 2001. Mr. Birger
is a director of variety of none profit organizations in Israel Mr. Birger holds a B.A. and an M.A. in economics from the Hebrew
University Jerusalem.
Anat Naschitz
has been serving
as a member of our Board since March 2013, and on September 29, 2016, was re-elected for service as a director until our next
annual general meeting. Ms. Naschitz currently serves as a director of Treato Ltd., Tyto Care Ltd., Axiom One Ltd., Nutrinia Ltd.
and MDClonce Ltd., privately held companies. Ms. Naschitz currently serves as a Managing Partner at OrbiMed Advisors LLC. Ms.
Naschitz has served as a Principal at Apax Partners, specializing in investments in medical equipment companies, and as an Associate
Principal at McKinsey and Company, managing international teams that worked with senior management of large pharmaceutical companies
on strategy, mergers and acquisitions, establishing new companies, research and development and marketing. Ms. Naschitz holds
an MBA from INSEAD and an LLB from Tel-Aviv University, Israel.
Oded
Yatzkan
has served as our Chief Financial Officer since February 2017 and from September 2003 to August 2015. From
August 2015 to February 2017, Mr. Yatzkan served as our VP Director of finance and accounting. Since August 2015, Mr. Yatzkan
is also serving as an external director at Oron Group Holdings and Investments. Mr. Yatzkan previously served as Controller and
Administrative Director of the Be’er Sheva Municipal Theatre from 1997 to 2003, as an Internal Auditor of Pandor Ltd., a
TASE-listed Israeli company, from 1999 to 2001, as Assistant to Chief Financial Manager of Gold & Honey Ltd. Mr. Yatzkan is
a licensed CPA, and holds a BA in Economics with a specialization in Accounting and an MBA with specialization in finance, both
from Ben-Gurion University of Be’er Sheva, Israel.
Minelu
(Menashe) Sonnenschein
is a founding member and officer of our company who has been serving as our VP Israel Operations
since June 2014. Among other roles, Mr. Sonnenschein previously served as our Director of Research and Development and has been
directly responsible for the development of the MUSE
TM
System since the founding of the Company. Mr. Sonnenschein holds
an M.Sc. in Electrical and Electronics Engineering from Ben-Gurion University of Be’er Sheva, Israel.
Yaron
Silberman
has been serving as VP Sales and Marketing since January 2011. Dr. Silberman has served as Marketing Director
of Niti Surgical Solutions Ltd., and as Product Manager of Given Imaging Ltd. Dr. Silberman holds a PhD in Computational Neuroscience
and Data Processing from Hebrew University of Jerusalem, Israel, an MBA from the College of Management Academic Studies of Rishon
Le’Zion, Israel, and a B.A. in Theoretical Mathematics from The Technion Institute of Technology, Israel.
Dr.
Aviel Roy Shapira
has served as our Medical Director since January 2000. Dr. Shapira has been serving as a Senior Head
Surgeon at Soroka Hospital in Be’er Sheva, Israel since 1993. Dr. Shapira holds an MD from Hebrew University of Jerusalem,
Israel. He was trained in surgery at Creighton University in Omaha, Nebraska and underwent fellowships at University of Chicago
(1986-1988), and Baylor University in Houston, Texas (1993). He was certified by the American Board of Surgery in 1988, and is
registered in the Israeli registry of medical specialists as a specialist in surgery (1989) and critical care (1993). He serves
on the faculty of health sciences at Ben-Gurion University in Be’er Sheva, Israel. He is the author of a number of publications
on GERD and its treatment.
Family
Relationships
There
are no family relationships between any members of our executive management and our directors.
Arrangements
for Election of Directions and Members of Management
There
are no arrangements or understandings with major shareholders, customers, suppliers or others pursuant to which any of our executive
management or our directors were appointed.
Compensation
of Executive Officers and Directors
In accordance with the provisions of the
Companies Law, the compensation of our directors and officer holders must generally comply with the terms and conditions of our
compensation policy, as approved by our compensation committee, board of directors and general meeting of our shareholders, subject
to certain exceptions under the Companies Law. Our current compensation policy was approved by our general meeting on December
29, 2015
.
The table below reflects the compensation
granted to our five most highly compensated office holders (as defined in the Companies Law) during or with respect to the year
ended December 31, 2016:
|
|
|
Annual Compensation
|
|
|
Long-Term Compensation
|
|
|
|
|
|
|
|
U.S. Dollars in thousands
|
|
|
Name and Position
|
|
Salary and Related Benefits*
|
|
|
Shares Underlying Options**
|
|
|
Total
|
|
Christopher (Chris) Rowland
Chief Executive Officer
|
|
|
330
|
|
|
|
19
|
|
|
|
349
|
|
Gilad Mamlok
former Chief Financial Officer
|
|
|
281
|
|
|
|
17
|
|
|
|
298
|
|
Jeremy Starkweather,
former Vice President, U.S. Sales & Marketing
|
|
|
221
|
|
|
|
0
|
|
|
|
221
|
|
Minelu (Menashe) Sonnenschein
Vice President, Israel Operations
|
|
|
159
|
|
|
|
0
|
|
|
|
165
|
|
Yaron Silberman
Vice President, Sales and Marketing
|
|
|
149
|
|
|
|
8
|
|
|
|
157
|
|
* Includes car expenses. We did not pay any bonuses
to our five most highly compensated office holders during 2016.
** Represents the equity-based compensation expenses recorded
in the Company’s consolidated financial statements for the year ended December 31, 2016, based on the option’s fair
value, calculated in accordance with accounting guidance for equity-based compensation.
The
aggregate compensation paid by us to our executive officers for the year ended December 31, 2016 was approximately $1.3 million.
This amount includes set aside or accrued to provide pension, severance, retirement or similar benefits or expenses, car expenses
and
value of the ordinary shares underlying the options representing accounting expenses
,
but does not include business travel, relocation, professional and business association dues and expenses reimbursed to officers,
and other benefits commonly reimbursed or paid by companies in Israel. The aggregate amount paid by us to our directors for the
year ended December 31, 2016, was approximately $66,000.
Under
the Companies Law and the rules and regulations promulgated thereunder, external directors are entitled to fixed annual compensation
and to an additional payment for each meeting attended. We currently pay our external directors an annual fee of NIS 37,115 and
a per meeting fee of NIS 1,860, in accordance with the external director fees allowed pursuant to applicable regulations under
the Companies Law, as applicable to the Company. We currently do not pay Nissim Darvish (our current chairman), Anat Naschitz
and Chris Rowland any cash fees for their service as directors. We have not granted any options to directors since 2010. The compensation
of our external directors is determined at the time of their election.
Employment
Agreements
We
have entered into written employment agreements with each of our executive officers. All of these agreements contain customary
provisions regarding noncompetition, confidentiality of information and assignment of inventions. However, the enforceability
of the non-competition provisions may be limited under applicable law. In addition, we have entered into agreements with each
executive officer and director pursuant to which we have agreed to indemnify each of them to the fullest extent permitted by law
to the extent that these liabilities are not covered by directors and officers insurance.
Our
office holders are generally eligible for bonuses each year. The bonuses are established and granted in accordance with our compensation
policy and, and are generally payable upon meeting objectives and targets that are approved by our compensation committee and
board of directors (and if required by our shareholders).
Equity Based Compensation of our Executive Officers and Directors
As of March 15, 2017, options to purchase
763,500 of our ordinary shares were outstanding and held by certain current executive officers and directors (consisting of 10
persons) with an average exercise price of NIS6.07 per ordinary share, of which options to purchase 552,000 of our ordinary shares
are currently exercisable or exercisable within 60 days as of March 15, 2017.
Employment
Agreement with Mr. Rowland
On September 29, 2013, our shareholders approved
that as of October 1, 2013, our U.S. Subsidiary, Medigus USA LLC, would enter into an employment agreement with Mr. Rowland, who
serves as our Chief Executive Officer and currently carries out his work from our U.S. Subsidiary’s office in California,
USA. The agreement has a term of 3 years, and may be automatically renewed for additional periods of one year unless either party
gives 60 days advance notice of non-renewal of the agreement. The agreement may be terminated by either party by giving 60 days
advance notice, or shorter periods in some cases.
In accordance with the employment agreement
with Mr. Rowland, he is entitled to an annual base salary of $315,000. In addition, by meeting certain pre-determined milestones
or goals, set by our board of directors, Mr. Rowland may be entitled to an annual target bonus, which may not exceed 35% of Mr.
Rowland’s annual base salary. The annual target bonus may be reduced by our board of directors according to our financial
position and Mr. Rowland’s performance, and must be returned by Mr. Rowland if later shown to be granted in error which
shall be restated in our financial statements. We have not granted Mr. Rowland an annual bonus for the year 2016.
Mr. Rowland is also entitled to various social
benefits, such as medical and dental insurance, for himself and his immediate family, participation in a “401(k)”
plan (similar to provident fund in Israel), and a manager’s insurance plan, which may not exceed total inclusive costs of
25% of his annual salary (approximately $79,000).
In the event that we terminate Mr. Rowland’s
employment without cause or if we elect not to renew his employment, Mr. Rowland will be entitled to (i) receive the amounts owed
to him up to the termination of his employment, (ii) receive a severance payment in an amount equal to six monthly base salaries
(total inclusive amount of approximately $158,000), (iii) continue his group health and dental insurance following the date of
termination until the earlier of the six months anniversary of the date of termination or until an alternate employer assumes
his group health and dental insurance (subject to certain conditions), (iv) a pro-rated incentive payment for the quarter or a
year in which his employment is terminated subject to the attainment of established targets, and (v) acceleration of option vesting.
On September 29, 2013, our shareholders approved
a grant of 4,500,000 options under the 2013 Share Option and Incentive Plan exercisable into 450,000 of our ordinary shares to
Mr. Rowland, for an exercise price of NIS 8.3 per share, of which 3,200,000 options vest over a period of 24 months, commencing
one year following the start of his employment, 650,000 options vested on June 30, 2014, following achievement of goals set by
the board of directors, and the remaining 650,000 options were to vest on February 1, 2015, subject to achieving the goals set
by the board of directors, and the latter 650,000 options expired on March 31, 2015 following a decision by the board of directors.
The options granted will expire at the earliest of: (a) following 5 years from the start date of the agreement; (b) 180 days from
the date Mr. Rowland’s employment shall end or he shall resign; or (c) immediately following the dismissal of Mr. Rowland,
for various causes. In addition, options not vested by the termination of Mr. Rowland’s employment agreement shall expire
at such date.
On
December 29, 2015, our shareholders approved a grant of 100,000 options under the 2013 Share Option and Incentive Plan to purchase
100,000 of our ordinary shares to Mr. Rowland, for an exercise price of NIS 2.05, which vest annually in four equal parts over
four years. The options granted will expire at the earliest of: (a) 6 years following the grant date; or (b) at the time at which
the options expire pursuant to the terms of 2013 Share Option and Incentive Plan. In addition, options not vested by the termination
of Mr. Rowland’s employment agreement shall expire at such date.
Introduction
Under
the Companies Law and our articles of association, the management of our business is vested in our board of directors. Our board
of directors may exercise all powers and may take all actions that are not specifically granted to our shareholders or to management.
Our executive officers are responsible for our day-to-day management and have individual responsibilities established by our board
of directors. Our chief executive officer is appointed by the general meeting of our shareholders, subject to his personal contract
with the Company.
Under
our articles of association, our board of directors must consist of at least three and not more than 12 directors, not including
at least two external directors, which are required to be appointed under the Companies Law. Our board of directors currently
consists of six members, including our non-executive chairman of the board of directors, which is also appointed by the general
meeting of our shareholders. Other than our two external directors, our directors are elected at the annual general meeting of
our shareholders by a simple majority. Because our ordinary shares do not have cumulative voting rights in the election of directors,
the holders of a majority of the voting power represented at a shareholders meeting have the power to elect all of our directors,
subject to the special approval requirements for external directors (See “— External Directors”). The general
meeting of the our shareholders may resolve, at any time, by an ordinary majority resolution, to remove from office any director
other than external director (for whom a special majority for appointment and removal is required as described below) prior to
the termination of his respective term of service and it may appoint another director in his place, provided that the director
was given a reasonable opportunity to state his case before the general meeting.
In
addition, if a director's office becomes vacant, the remaining serving directors may continue to act in any manner, provided that
their number is of the minimal number specified in our articles of association. If the number of serving directors is lower than
three, then our board of directors shall not be permitted to act, other than for the purpose of convening a general meeting of
the Company's shareholders for the purpose of appointing additional directors. In addition, the directors may appoint, immediately
or of a future date, additional director(s) to serve until the subsequent annual general meeting of our shareholders, provided
that the total number of directors in office shall not exceed twelve directors (not including external directors).
Pursuant
to the Companies Law and our articles of association, a resolution proposed at any meeting of our board of directors at which
a quorum is present is adopted if approved by a vote of a majority of the directors present and eligible to vote. A quorum of
the board of directors requires at least a majority of the directors then in office who are lawfully entitled to participate in
the meeting.
According
to the Companies Law, the board of directors of a public company must determine the minimum number of board members that should
have financial and accounting expertise while considering, among other, the nature of the company, its size, the scope and
complexity of its operations and the number of directors stated in the articles of association. Our board of directors resolved
that the minimum number of board members that need to have financial and accounting expertise, including the external director
with financial and accounting expertise, is one and that Mr. Eitan Machover has accounting and financial expertise as required
under the Companies Law.
External
Directors
Under
the Companies Law, we are required to appoint at least two external directors to serve on our board of directors. Our external
directors are Mr. Machover and Ms. Venkert, both elected by the general meeting of our shareholders to serve for a three years
term as of September 29, 2016.
Election
and removal of external directors
Under
Israeli law, external directors are elected by a special majority vote of the shares present and voting at a shareholders’
meeting, provided that either:
|
|
●
|
such
majority includes at least a majority of the shares held by all shareholders who are
non-controlling shareholders and do not have a personal interest in the election of the
external director (other than a personal interest not deriving from a relationship with
a controlling shareholder) that are voted at the meeting, excluding abstentions, to which
we refer as a disinterested majority; or
|
|
|
●
|
the
total number of shares voted by non-controlling shareholders and by shareholders who
do not have a personal interest in the election of the external director, against the
election of the external director, does not exceed 2% of the aggregate voting rights
in the company.
|
The
initial term of an external director of an Israeli public company is three years. The external director may be re-elected, subject
to certain circumstances and conditions, for up to two additional terms of three years each, and thereafter, subject to conditions
set out in the regulations promulgated under the Companies Law, to further three year terms. Each re-election is subject to one
of the following:
|
|
(i)
|
his
or her service for each such additional term is recommended by one or more shareholders
holding at least 1% of the company’s voting rights and is approved at a shareholders’
meeting by a disinterested majority (other than a personal interest not deriving from
a relationship with a controlling shareholder), where the total number of shares held
by non-controlling, disinterested shareholders voting for such reelection exceeds 2%
of the aggregate voting rights in the company. In such event, the external director so
reappointed may not be a Related or Competing Shareholder, or a relative of such shareholder,
at the time of the appointment, and is not and has not had any affiliation with a Related
or Competing Shareholder, at such time or during the two years preceding such person’s
reappointment to serve an additional term as external director. The term “Related
or Competing Shareholder” means a shareholder proposing the reappointment or a
shareholder holding 5% or more of the outstanding shares or voting rights of the company,
provided, that at the time of the reappointment, such shareholder, the controlling shareholder
of such shareholder, or a company controlled by such shareholder, have a business relationship
with the company or are competitors of the company. Additionally, the Israeli Minister
of Justice, in consultation with the Israeli Securities Authority, may determine matters
that under certain conditions will not constitute a business relationship or competition
with the company;
|
|
|
(ii)
|
his
or her service for each such additional term is recommended by the board of directors
and is approved at a shareholders meeting by the same majority required for the initial
election of an external director (as described above); or
|
|
|
(iii)
|
the
external director proposed his or her own nomination, and such nomination was approved
in accordance to the requirements described in the paragraph (i) above.
|
The
term of office for external directors for Israeli companies traded on certain foreign stock exchanges, including the NASDAQ Capital
Market, may be extended indefinitely in increments of additional three-year terms, in each case provided that the audit committee
and the board of directors of the company determined that in light of the external director’s expertise and special contribution
to the work of the board of directors and its committees, the reelection for such additional period(s) is in the best interest
of the company, and provided that the external director is reelected subject to the same shareholder vote requirements as if elected
for the first time (as described above). Prior to the approval of the reelection of the external director at a general shareholders
meeting, the company’s shareholders must be informed of the term previously served by him or her and of the reasons, which
led the board of directors and audit committee to recommend the extension of his or her tenure.
External
directors may be removed from office by a special general meeting of shareholders called by the board of directors, which approves
such dismissal by the same majority vote required for their election or by a court, in each case, only under limited circumstances,
including ceasing to meet the statutory qualifications for appointment, or violating their duty of loyalty towards the company.
If an external directorship becomes vacant and there are fewer than two external directors on the board of directors at the time,
then the board of directors is required under the Companies Law to call a special shareholders’ meeting as soon as practicable
to appoint a replacement external director.
If
the vacancy of an external directorship causes a company to have fewer than two external directors, the company’s board
of directors is required under the Companies Law to call a special general meeting of the company’s shareholders as soon
as practicable to appoint such number of new external directors so that the company thereafter has two external directors.
Qualifications
of External Directors
A
person may not be appointed as an external director if such person is a relative of a controlling shareholder or if on the date
of the person’s appointment or within the preceding two years the person or his or her relatives, partners, employers or
anyone to whom that person is subordinate, whether directly or indirectly, or entities under the person’s control have or
had any affiliation with any of (each an “Affiliated Party”): (1) us; (2) any person or entity controlling us on the
date of such appointment; (3) any relative of a controlling shareholder; or (4) any entity controlled, on the date of such appointment
or within the preceding two years, by us or by a controlling shareholder. If there is no controlling shareholder or any shareholder
holding 25% or more of voting rights in the company, a person may not be appointed as an external director if the person has any
affiliation to the chairman of the board of directors, the general manager (chief executive officer), any shareholder holding
5% or more of the company’s shares or voting rights or the senior financial officer as of the date of the person’s
appointment.
The
term “controlling shareholder” means a shareholder with the ability to direct the activities of the company, excluding
such ability deriving solely from his or her position as a director of the company or from any other position with the company.
A shareholder is presumed to have “control” of the company and thus to be a controlling shareholder of the company
if the shareholder holds 50% or more of the “means of control” of the company. “Means of control” is defined
as (1) the right to vote at a general meeting of a company or a corresponding body of another corporation; or (2) the right to
appoint directors of the corporation or its general manager. For the purpose of approving transactions with controlling shareholders,
the term also includes any shareholder that holds 25% or more of the voting rights of the company if the company has no shareholder
that owns more than 50% of its voting rights. For the purpose of determining the holding percentage stated above, two or more
shareholders who have a personal interest in a transaction that is brought for the company’s approval are deemed as joint
holders.
Under
the Companies Law, “affiliation” includes:
|
|
●
|
an
employment relationship;
|
|
|
●
|
a
business or professional relationship even if not maintained on a regular basis (excluding
insignificant relationships);
|
|
|
●
|
service
as an office holder, excluding service as a director of a private company prior to the
first offering of its shares to the public if such director was appointed as a director
of the private company in order to serve as an external director following the initial
public offering.
|
The
term “relative” is defined as a spouse, sibling, parent, grandparent, descendant, spouse’s sibling, parent or
descendant, and the spouse of each of the foregoing.
The
term “office holder” is defined under the Companies Law as a general manager, chief business manager, deputy general
manager, vice general manager any other person assuming the responsibilities of any of these positions regardless of that person’s
title, a director and any other manager directly subordinate to the general manager. Each person listed in the table under “Management”
is an office holder.
Under
the Companies Law, a person cannot serve as an external director if such person, his or her relative, partner, employer, another
person to whom he or she is subordinate, directly or indirectly, or any entity under such person’s control, has or had,
any professional relationship with an Affiliated Party, except for insignificant relationships, even if such professional relationship
are extraordinary or intermediate. In addition, a person who received compensation other than in accordance with the applicable
provisions for the compensation of external directors under the Companies Law may not serve as an external director.
In
addition, a person cannot serve as an external director if: (i) the person’s position or professional or other affairs create,
or may create, a conflict of interest with such person’s responsibilities as a director or may otherwise interfere with
the person’s ability to serve as an external director, (ii) if such a person is an employee of the Israeli Securities Authority
or of an Israeli stock exchange, (iii) at the time appointment, such person serves as a director of another company and an external
director of the other company is also a director of the company, or (iv) such person received direct or indirect compensation
from the company in connection with such person’s services as an external director, other than as permitted by the Companies
Law and the regulations promulgated thereunder. If at the time an external director is appointed, all members of the board of
directors who are not controlling shareholders or relatives of controlling shareholders are of the same gender, then the external
director to be appointed must be of the other gender.
The
Companies Law provides that an external director must meet certain professional qualifications or have financial and accounting
expertise and that at least one external director must have financial and accounting expertise. However, if at least one of our
other directors (1) meets the independence requirements of the Exchange Act, (2) meets the standards of the NASDAQ Marketplace
Rules for membership on the audit committee and (3) has financial and accounting expertise as defined in the Companies Law and
applicable regulations, then neither of our external directors is required to possess financial and accounting expertise as long
as both possess other requisite professional qualifications. The determination of whether a director possesses financial and accounting
expertise is made by the board of directors. is a director who, due to his or her education, experience and skills, possesses
an expertise in, and an understanding of, financial and accounting matters and financial statements, such that he or she is able
to understand the financial statements of the company and initiate a discussion about the presentation of financial data. In determining
whether the director has financial and accounting expertise the board of directors shall consider education, experience and the
knowledge in the following subjects: (i) accounting issues and internal auditing issues typical to the company’s industry
and to companies of the same size and complexity as the company; (ii) the nature of the Internal Auditor’s position in the
company and his or her duties; and (iii) the preparation of financial statements and their approval subject to the Companies Law
and the Israeli Securities Law 5728-1968, or the Israeli Securities Law.
The
regulations promulgated under the Companies Law define an external director with requisite “professional qualifications”
as a director who satisfies one of the following requirements: (1) the director holds an academic degree in either economics,
business administration, accounting, law or public administration, (2) the director either holds an academic degree in any other
field or has completed another form of higher education in the company’s primary field of business or in an area which is
relevant to his or her office as an external director in the company, or (3) the director has at least five years of experience
serving in any one of the following, or at least five years of cumulative experience serving in two or more of the following capacities:
(a) a senior business management position in a company with a substantial scope of business, (b) a senior position in the company’s
primary field of business or (c) a senior position in public administration.
Additional
provisions
Under
the Companies Law, external directors of a company are prohibited from receiving, directly or indirectly, any compensation from
the company other than compensation and reimbursement of expenses amounts for their services as external directors except for
certain exculpation, indemnification and insurance provided by the company, as specifically allowed by the Companies Law. Compensation
of an external director is determined prior to his or her appointment and may not be changed during his or her term (subject to
certain exceptions).
In
addition, until the lapse of a two-year period from the date that an external director of a company ceases to act in such capacity,
the company in which such external director served, and its controlling shareholder or any entity under control of such controlling
shareholder may not, directly or indirectly, grant such former external director, or his or her spouse or child, any benefit,
including via (i) the appointment of such former director or his or her spouse or his child as an officer in the company
or in an entity controlled by the company’s controlling shareholder, (ii) the employment of such former director, and (iii)
the engagement, directly or indirectly, of such former director as a provider of professional services for compensation, directly
or indirectly, including via an entity under his or her control. With respect to a relative who is not a spouse or a child, such
limitations shall only apply for one year from the date such external director ceased to be engaged in such capacity.
Alternate
directors
Our
articles of association provide, as allowed by the Companies Law, that any director may, by written notice to us, appoint another
person who is qualified to serve as a director to serve as an alternate director and to terminate such appointment. Under the
Companies Law, a person who is not qualified to be appointed as a director, a person who is already serving as a director or a
person who is already serving as an alternate director for another director, may not be appointed as an alternate director. Nevertheless,
a director who is already serving as a director may be appointed as an alternate director for a member of a committee of the board
of directors as long as he or she is not already serving as a member of such committee, and if the alternate director is to replace
an external director he must have the relevant qualifications of such external director.
Board
committees
The
board of directors may, subject to the provisions of the Companies Law, delegate any or all of its powers to committees, each
consisting of one or more directors (except the audit and compensation committees, as described below), and it may, from time
to time, revoke such delegation or alter the composition of any such committees. Unless otherwise expressly provided by the board
of directors, the committees shall not be empowered to further delegate such powers. Each committee of the board of directors
that is authorized to exercise the powers of the board of directors must include at least one external director, except that the
audit committee and the compensation committee must include all external directors then serving on the board of directors. The
composition and duties of our audit committee and compensation committee are established under the Israeli Companies Law and described
below.
Audit
committee
Our
audit committee is currently comprised of Ms. Venkert, Mr. Machover and Mr. Birger. Ms. Venkert acts as the Chairperson of our
audit committee.
Companies
Law requirements
Under
the Companies Law, the board of directors of any public company must also appoint an audit committee comprised of at least three
directors, including all of the external directors, one of whom must serve as chairman of the committee. The audit committee may
not include:
|
|
●
|
the
chairman of the board of directors;
|
|
|
●
|
a
controlling shareholder or a relative of a controlling shareholder; or
|
|
|
●
|
a
director employed by or providing services on a regular basis to the company, to a controlling
shareholder or to an entity controlled by a controlling shareholder or a director most
of whose livelihood depends on a controlling shareholder
|
In
addition, the majority of the members of the audit committee are required to be independent directors. The term “independent
director” is generally defined under the Companies Law as an external director or a director who meets the following criteria:
|
|
●
|
he
or she meets the qualifications for being appointed as an external director, except for
the requirement that the director be an Israeli resident (which does not apply to companies
such as ours whose securities have been offered outside of Israel or are listed outside
of Israel); and
|
|
|
●
|
he
or she has not served as a director of the company for a period exceeding nine consecutive
years, provided that, for this purpose, a break of less than two years in service shall
not be deemed to interrupt the continuation of the service.
|
Any
person disqualified from serving as a member of the audit committee may not be present at the audit committee meetings, unless
the chairman of the audit committee has determined that such person is required to be present at the meeting or if such person
qualifies under one of the exemptions of the Companies Law.
The
quorum required for the convening of meetings of the audit committee and for adopting resolutions by the audit committee is a
majority of the members of the audit committee, provided such majority is comprised of a majority of independent directors, and
at least one of those present is an external director.
Listing
requirements
Under
the NASDAQ Capital Market corporate governance rules, we are required to maintain an audit committee consisting of at least three
independent directors, all of whom are financially literate and one of whom has accounting or related financial management expertise.
All members of our audit committee meet the requirements for financial literacy under the applicable rules and regulations of
the SEC and the NASDAQ Capital Market corporate governance rules. Our board of directors has determined that each of Mr. Birger
and Mr. Machover is an audit committee financial expert as defined by the SEC rules and has the requisite financial experience
as defined by the NASDAQ Marketplace Rules.
Each
of the members of the audit committee is required to be “independent” as such term is defined in Rule 10A-3(b)(1)
under the Exchange Act, which is different from the general test for independence of board and committee members.
The
approval of the audit committee is required to effect specified actions and transactions with office holders and controlling shareholders
and their relatives, or in which they have a personal interest. See “Fiduciary duties and approval of specified related
party transactions and compensation under Israeli law.” The audit committee may not approve an action or a transaction with
a controlling shareholder or with an office holder unless at the time of approval the audit committee meets the composition requirements
under the Companies Law.
Audit
committee role
Our
audit committee provides assistance to our board of directors in fulfilling its legal and fiduciary obligations in matters involving
our accounting, auditing, financial reporting, internal control and legal compliance functions by pre-approving the services performed
by our independent accountants and reviewing their reports regarding our accounting practices and systems of internal control
over financial reporting. Our audit committee also oversees the audit efforts of our independent accountants and takes those actions
that it deems necessary to satisfy itself that the accountants are independent of management.
Under
the Companies Law, the audit committee is responsible, among others, for:
|
|
(i)
|
determining
whether there are deficiencies in the business management practices of our Company, including
in consultation with our internal auditor or the independent auditor, and making recommendations
to our board of directors to improve such practices;
|
|
|
(ii)
|
determining
the approval process for transactions that are ‘non-negligible’ (i.e., transactions
with a controlling shareholder that are classified by the audit committee as non-negligible,
even though they are not deemed extraordinary transactions), as well as determining which
types of transactions would require the approval of the audit committee, optionally based
on criteria which may be determined annually in advance by the audit committee;
|
|
|
(iii)
|
determining
whether to approve certain related party transactions (including transactions in which
an office holder has a personal interest and whether such transaction is extraordinary
or material under Companies Law;
|
|
|
(iv)
|
where
the board of directors approves the working plan of the internal auditor, to examine
such working plan before its submission to our board of directors and proposing amendments
thereto;
|
|
|
(v)
|
examining
our internal controls and internal auditor’s performance, including whether the
internal auditor has sufficient resources and tools to dispose of its responsibilities;
|
|
|
(vi)
|
examining
the scope of our auditor’s work and compensation and submitting a recommendation
with respect thereto to our board of directors or shareholders, depending on which of
them is considering the appointment of our auditor; and
|
|
|
(vii)
|
establishing
procedures for the handling of employees’ complaints as to the management of our
business and the protection to be provided to such employees.
|
Compensation
Committee
Our
compensation committee is currently comprised of Ms. Venkert, Mr. Machover and Mr. Birger. Mr. Machover acts as the Chairman of
our compensation committee.
Companies
Law requirements
Under
the Companies Law, the board of directors of a public company must appoint a compensation committee and adopt a compensation policy.
The compensation committee must consist of at least three members. All of the external directors must serve on the committee and
constitute a majority of its members. The chairman of the compensation committee must be an external director. However, subject
to certain exceptions, Israeli companies whose securities are traded on stock exchanges such as the NASDAQ Capital Market, and
who do not have a controlling shareholder, do not have to meet this majority requirement; provided, however, that the compensation
committee meets other Companies Law composition requirements, as well as the requirements of the jurisdiction where the company’s
securities are listed. Each compensation committee member that is not an external director must be a director whose compensation
does not exceed an amount that may be paid to an external director (under the Companies Law and applicable regulations). The compensation
committee is subject to the same Companies Law restrictions as the audit committee as to who may not serve as a member of the
committee.
Role
of the Compensation Committee
Under
the Companies Law, the roles of the compensation committee are, among others, as follows:
|
|
●
|
to
recommend to the board of directors the approval of compensation policy for directors
and officers in accordance with the requirements of the Companies Law;
|
|
|
●
|
to
oversee the development and implementation of such compensation policy and recommending
to the board of directors regarding any amendments or modifications that the compensation
committee deems appropriate;
|
|
|
●
|
to
determine whether to approve transactions concerning the terms of engagement and employment
of office holders that require approval of the compensation committee; and
|
|
|
●
|
to
resolve whether to exempt a transaction with a candidate for chief executive officer
from shareholder’s approval.
|
In
addition, any amendment of existing terms of office and employment of office holders (other than directors or controlling shareholders
and their relatives, who serve as office holders) requires the sole approval of the compensation committee, if the committee determines
that the amendment is not material in relation to its existing terms and if such amendment is in accordance with the then approved
in effect compensation policy of the company.
The
compensation policy
The
compensation policy must be based on certain considerations, must include certain provisions and needs to reference certain matters
as set forth in the Companies Law. The compensation policy must be approved by the company’s board of directors after considering
the recommendations of the compensation committee. In addition, the compensation policy needs to be approved by the company’s
shareholders by a simple majority, provided that (i) such majority includes a majority of the votes cast by the shareholders who
are not controlling shareholders and who do not have a personal interest in the matter, present and voting (abstentions are disregarded)
or (ii) the votes cast by shareholders who are not controlling shareholders and who do not have a personal interest in the matter
who were present and voted against the compensation policy, constitute two percent or less of the voting power of the company.
Such majority determined in accordance with clause (i) or (ii) is hereinafter referred to as the Compensation Special Majority
Requirement.
To
the extent a compensation policy is not approved by shareholders at a duly convened shareholders meeting or by the Compensation
Special Majority Requirement, the board of directors of a company may override the resolution of the shareholders following a
re-discussion of the matter by the board of directors and the compensation committee and for specified reasons, and after determining
that despite the rejection by the shareholders, the adoption of the compensation policy is in the best interest of the company.
A compensation policy that is for a period of more than three years must be approved in accordance with the above procedure once
every three years.
Notwithstanding
the above, the amendment of existing terms of office and employment of office holders (other than directors or controlling shareholders
and their relatives, who serve as office holders) requires the sole approval of the compensation committee, if such committee
determines that the amendment is not material in relation to its existing terms.
The
compensation policy must serve as the basis for decisions concerning the financial terms of employment or engagement of office
holders, including exculpation, insurance, indemnification or any monetary payment or obligation of payment in respect of employment
or engagement. The compensation policy must relate to certain factors, including advancement of the company’s objectives,
the company’s business plan and its long-term strategy, and creation of appropriate incentives for office holders. It must
also consider, among other things, the company’s risk management, size and the nature of its operations. The compensation
policy must furthermore consider the following additional factors:
|
|
●
|
the
knowledge, skills, expertise and accomplishments of the relevant office holder;
|
|
|
●
|
the
office holder’s roles and responsibilities and prior compensation agreements with
him or her;
|
|
|
●
|
the
ratio between the cost of the terms of employment of an office holder and the cost of
the compensation of the other employees of the company, including those employed through
manpower companies, in particular the ratio between such cost and the average and median
compensation of the other employees of the company, as well as the impact such disparities
may have on the work relationships in the company;
|
|
|
●
|
the
possibility of reducing variable compensation, if any, at the discretion of the board
of directors; and the possibility of setting a limit on the exercise value of non-cash
variable equity-based compensation; and
|
|
|
●
|
as
to severance compensation, if any, the period of service of the office holder, the terms
of his or her compensation during such service period, the company’s performance
during that period of service, the person’s contribution towards the company’s
achievement of its goals and the maximization of its profits, and the circumstances under
which the person is leaving the company.
|
The
compensation policy must also include the following principles:
|
|
●
|
the
link between variable compensation and long-term performance and measurable criteria;
|
|
|
●
|
the
relationship between variable and fixed compensation, and the ceiling for the value of
variable compensation;
|
|
|
●
|
the
conditions under which an office holder would be required to repay compensation paid
to him or her if it was later shown that the data upon which such compensation was based
was inaccurate and was required to be restated in the company’s financial statements;
|
|
|
●
|
the
minimum holding or vesting period for variable, equity-based compensation; and
|
|
|
●
|
maximum
limits for severance compensation.
|
In
accordance with the Companies Law, and following the recommendation of our compensation committee, our board of directors approved
our compensation policy, and our shareholders, in turn, approved the compensation policy at our annual general meeting of shareholders
that was held in December 2015.
Investment Committee
Our
investment committee consists of three directors, Mr. Birger, Ms. Venkert and Ms. Naschitz. The investment committee’s
duties include reviewing and making recommendations to the board of directors regarding the company’s investment policies,
hedging policy and other banking related matters.
Internal
auditor
Under
the Companies Law, the board of directors of a public company must appoint an internal auditor based on the recommendation of
the audit committee. Under the Companies Law, Each of the following may not be appointed as internal auditor:
|
|
●
|
a
person (or a relative of a person) who holds more than 5% of the company’s outstanding
shares or voting rights;
|
|
|
●
|
a
person (or a relative of a person) who has the power to appoint a director or the general
manager of the company;
|
|
|
●
|
an
office holder (including a director) of the company (or a relative thereof); or
|
|
|
●
|
a
member of the company’s independent accounting firm, or anyone on his or her behalf.
|
The
role of the internal auditor is, among other things, to examine whether a company’s actions comply with applicable law and
orderly business procedure. The audit committee is required to oversee the activities and to assess the performance of the
internal auditor as well as to review the internal auditor’s work plan. Our internal auditor is Hila Bar of Brightman Almagor
Zohar & Co., a member of Deloitte Touche Tohmatsu.
Fiduciary duties and approval of specified related party transactions and compensation under Israeli law
Fiduciary
duties of office holders
The
Companies Law imposes a fiduciary duties on all office holders of a company comprised of a duty of care and a duty of loyalty.
The
duty of care requires an office holder to act in the same degree of proficiency with which a reasonable office holder in the same
position would have acted under the same circumstances. The duty of care includes, among other things, a duty to use reasonable
means, in light of the circumstances, to obtain:
|
|
●
|
information
on the business advisability of a given action brought for his or her approval or performed
by virtue of his or her position; and
|
|
|
●
|
all
other important information pertaining to such action.
|
The
duty of loyalty requires an office holder to act in good faith and for the benefit of the company, and includes, among other things,
the duty to:
|
|
●
|
refrain
from any act involving a conflict of interest between the performance of his or her duties
in the company and his or her other duties or personal affairs;
|
|
|
●
|
refrain
from any activity that is competitive with the business of the company;
|
|
|
●
|
refrain
from exploiting any business opportunity of the company for the purpose of gaining a
personal advantage for himself or herself or others; and
|
|
|
●
|
disclose
to the company any information or documents relating to the company’s affairs which
the office holder received as a result of his or her position as an office holder.
|
We
may approve an act specified above, provided that the office holder acted in good faith, the act or its approval does not harm
the company’s best interest, and the office holder discloses his or her personal interest a sufficient time before the approval
of such act, including any relevant document.
Disclosure of personal interests of an office holder and approval of transactions
The
Companies Law requires that an office holder promptly disclose to the company any personal interest that he or she may have and
all related material information or documents relating to any existing or proposed transaction by the company. An interested office
holder’s disclosure must be made promptly and in any event no later than the first meeting of the board of directors at
which the transaction is considered. Under the Companies Law, once an office holder complies with the above disclosure requirement,
the board of directors at which the transaction is considered. An office holder is not obliged to disclose such information if
the personal interest of the office holder derives solely from the personal interest of his or her relative in a transaction that
is not considered an extraordinary transaction.
Under
the Companies Law, a company may approve a transaction between the company and the office holder or a third party in which the
office holder has a personal interest only if the office holder has complied with the above disclosure requirement, provided,
however, that a company may not approve a transaction or action that is not to the company’s benefit.
Under
the Companies Law, unless the articles of association of a company provide otherwise, a transaction with an office holder or with
a third party in which the office holder has a personal interest, which is not an extraordinary transaction, requires approval
by the board of directors. Our articles of association do not state otherwise. If the transaction considered with an office holder
or third party in which the office holder has a personal interest is an extraordinary transaction, then the audit committee’s
approval is required prior to approval by the board of directors. For the approval of compensation arrangements with directors
and executive officers, see “—Compensation of directors and executive officers.”
Any
person who has a personal interest in the approval of a transaction that is brought before a meeting of the board of directors
or the audit committee may not be present at the meeting or vote on the matter. However, if the chairperson of the board of directors
or the chairperson of the audit committee has determined that the presence of an office holder with a personal interest is required,
such office holder may be present at the meeting for the purpose of presenting the matter. Notwithstanding the foregoing, a director
who has a personal interest may be present at the meeting and vote on the matter if a majority of the directors or members of
the audit committee have a personal interest in the approval of such transaction’ provided, however, that if a majority
of the directors at a board of directors meeting have a personal interest in the approval of the transaction, such transaction
also requires the approval of the shareholders of the company.
A
“personal interest” is defined under the Companies Law as the personal interest of a person in an action or in a transaction
of the company, including the personal interest of such person’s relative or the interest of any other corporate body in
which such person and/or such person’s relative is a director or general manager, a 5% shareholder or holds 5% or more of
the voting rights, or has the right to appoint at least one director or the general manager, but excluding a personal interest
stemming solely from the fact of holding shares in the company. A personal interest also includes (1) a personal interest of a
person who votes according to a proxy of another person, including in the event that the other person has no personal interest,
and (2) a personal interest of a person who gave a proxy to another person to vote on his or her behalf regardless of whether
the discretion of how to vote lies with the person voting or not.
An
“extraordinary transaction” is defined under the Companies Law as any of the following:
|
|
●
|
a
transaction other than in the ordinary course of business;
|
|
|
●
|
a
transaction that is not on market terms; or
|
|
|
●
|
a
transaction that may have a material impact on the company’s profitability, assets
or liabilities.
|
An
extraordinary transaction in which an office holder has a personal interest requires approval of the company’s audit committee
followed by the approval of the board of directors.
Disclosure
of personal interests of a controlling shareholder and approval of transactions
The
Companies Law also requires that a controlling shareholder promptly disclose to the company any personal interest that he or she
may have and all related material information or documents relating to any existing or proposed transaction by the company. A
controlling shareholder’s disclosure must be made promptly and in any event no later than the first meeting of the board
of directors at which the transaction is considered. The following require the approval of each of (i) the audit committee (or
the compensation committee with respect to the terms of engagement of the controlling shareholder or relative thereof with the
company related for the provision of service, including among others as an office holder or employee of the company), (ii) the
board of directors and (iii) the shareholders (in that order): (a) extraordinary transactions with a controlling shareholder or
in which a controlling shareholder has a personal interest (including a private placement in which a controlling shareholder has
a personal interest), (b) the engagement with a controlling shareholder or his or her relative, directly or indirectly, for the
provision of services to the company, (c) the terms of engagement and compensation of a controlling shareholder or his or her
relative as an office holder, and (d) the employment of a controlling shareholder or his or her relative by the company, other
than as an office holder (collectively referred as Transaction with a Controlling Shareholder). In addition, the shareholder approval
must fulfill one of the following requirements:
|
|
●
|
at
least a majority of the shares held by shareholders who have no personal interest in
the transaction and are voting at the meeting must be voted in favor of approving the
transaction, excluding abstentions; or
|
|
|
●
|
the
shares voted by shareholders who have no personal interest in the transaction who vote
against the transaction represent no more than two percent (2%) of the voting rights
in the company.
|
In
addition, any extraordinary transaction with a controlling shareholder or in which a controlling shareholder has a personal interest
with a term of more than three years requires the abovementioned approval every three years, however, unless, with respect to
certain transactions the audit committee determines that such longer term is reasonable under the circumstances.
Pursuant
to regulations promulgated under the Companies Law, certain transactions with a controlling shareholder, a relative thereof, or
with a director, that would otherwise require approval of a company’s shareholders may be exempt from shareholder approval
upon certain determinations of the audit committee and board of directors.
The
Companies Law requires that every shareholder that participates, in person, by proxy or by voting instrument, in a vote regarding
a transaction with a controlling shareholder, must indicate in advance or in the ballot whether or not that shareholder has a
personal interest in the vote in question. Failure to so indicate will result in the invalidation of that shareholder’s
vote.
Disclosure
of Compensation of Executive Officers
For
so long as we qualify as a foreign private issuer, we are not required to comply with the proxy rules applicable to U.S. domestic
companies, including the requirement applicable to emerging growth companies to disclose the compensation of our chief executive
officer and other two most highly compensated executive officers on an individual, rather than an aggregate, basis. Under the
Companies Law and the regulations promulgated thereunder we are required to disclose the annual compensation of our five most
highly compensated office holders on an individual basis, rather than on an aggregate basis. This disclosure will not be as extensive
as that required of a U.S. domestic issuer.
Approval
of the compensation of directors and executive officers
The
compensation of, or an undertaking to indemnify, insure or exculpate, an office holder who is not a director requires the approval
of the company’s compensation committee, followed by the approval of the company’s board of directors, and, if such
compensation arrangement or an undertaking to indemnify or insure is inconsistent with the company’s stated compensation
policy, or if the said office holder is the chief executive officer of the company (apart from a number of specific exceptions),
then such arrangement is subject to the approval of our shareholders, subject to the Compensation Special Majority Requirement.
Directors
.
Under the Companies Law, the compensation of our directors requires the approval of our compensation committee, the subsequent
approval of the board of directors and, unless exempted under the regulations promulgated under the Companies Law, the approval
of the general meeting of our shareholders. If the compensation of our directors is inconsistent with our stated compensation
policy, then, provided that those provisions that must be included in the compensation policy according to the Companies Law have
been considered by the compensation committee and board of directors, shareholder approval will also be required to be approved
by the Compensation Special Majority Requirement.
Executive
officers other than the chief executive officer.
The Companies Law requires the approval of the compensation of a public
company’s executive officers (other than the chief executive officer) in the following order: (i) the compensation committee,
(ii) the company’s board of directors, and (iii) if such compensation arrangement is inconsistent with the company’s
stated compensation policy, the company’s shareholders by the Compensation Special Majority Requirement. However, if the
shareholders of the company do not approve a compensation arrangement with an executive officer that is inconsistent with the
company’s stated compensation policy, the compensation committee and board of directors may override the shareholders’
decision if each of the compensation committee and the board of directors provide detailed reasons for their decision.
Chief
executive officer.
Under the Companies Law, the compensation of a public company’s chief executive officer is required
to be approved by: (i) the company’s compensation committee; (ii) the company’s board of directors, and (iii) the
company’s shareholders by the Compensation Special Majority Requirement. However, if the shareholders of the company do
not approve the compensation arrangement with the chief executive officer, the compensation committee and board of directors may
override the shareholders’ decision if each of the compensation committee and the board of directors provide a detailed
reasoning for their decision. The approval of each of the compensation committee and the board of directors must be in accordance
with the company’s stated compensation policy; however, under special circumstances, the compensation committee and the
board of directors may approve compensation terms of a chief executive officer that are inconsistent with the company’s
compensation policy provided that they have considered those provisions that must be included in the compensation policy according
to the Companies Law and that shareholder approval was obtained by the Compensation Special Majority Requirement. In addition,
the compensation committee may resolve that the shareholder approval is not required for the approval of the engagement terms
of a candidate to serve as the chief executive officer, if the compensation committee determines that the compensation arrangement
is consistent with the company’s stated compensation policy, that the chief executive officer did not had a prior business
relationship with the company or a controlling shareholder of the company, and that subjecting the approval to a shareholder vote
would impede the company’s ability to attain the candidate to serve as the company’s chief executive officer.
Duties
of shareholders
Under
the Companies Law, a shareholder has a duty to refrain from abusing its power in the company and to act in good faith and in an
acceptable manner in exercising its rights and performing its obligations to the company and other shareholders, including, among
other things, when voting at meetings of shareholders on the following matters:
|
|
●
|
an
amendment to the articles of association;
|
|
|
●
|
an
increase in the company’s authorized share capital;
|
|
|
●
|
the
approval of related party transactions and acts of office holders that require shareholder
approval.
|
A
shareholder also has a general duty to refrain from discriminating against other shareholders.
The
remedies generally available upon a breach of contract will also apply to a breach of the shareholder duties mentioned above,
and in the event of discrimination against other shareholders, additional remedies are available to the injured shareholder.
In
addition, any controlling shareholder, any shareholder that knows that its vote can determine the outcome of a shareholder vote
and any shareholder that, under a company’s articles of association, has the power to appoint or prevent the appointment
of an office holder, or any other power with respect to a company, is under a duty to act with fairness towards the company. The
Companies Law does not describe the substance of this duty except to state that the remedies generally available upon a breach
of contract will also apply in the event of a breach of the duty to act with fairness, taking each shareholder’s position
in the company into account.
Approval
of private placements
Under
the Companies Law and the regulations promulgated thereunder, a private placement of securities does not require approval at a
general meeting of the shareholders of a company; provided however, that in special circumstances, such as a private placement
completed in lieu of a special tender offer (See “Description of Share Capital – Acquisitions under Israeli law”)
or a private placement which qualifies as a related party transaction (See “– Fiduciary duties and approval of specified
related party transactions and compensation under Israeli law”), approval at a general meeting of the shareholders of a
company is required.
Exemption,
Insurance and Indemnification of Directors and Officers
Under
the Companies Law, a company may not exculpate an office holder from liability for a breach of a fiduciary duty. An Israeli company
may exculpate an office holder in advance from liability to the company, in whole or in part, for damages caused to the company
as a result of a breach of duty of care but only if a provision authorizing such exculpation is included in its articles of association.
Our articles of association include such a provision. The company may not exculpate in advance a director from liability arising
out of a prohibited dividend or distribution to shareholders.
Under the Companies Law and the Securities
Law, 5738-1968, or the Securities Law, a company may indemnify an office holder in respect of the following liabilities, payments
and expenses incurred for acts performed by him or her as an office holder, either in advance of an event or following an event,
provided its articles of association include a provision authorizing such indemnification:
|
|
●
|
a
monetary liability incurred by or imposed on the office holder in favor of another person
pursuant to a court judgment, including pursuant to a settlement confirmed as judgment
or arbitrator’s decision approved by a competent court. However, if an undertaking
to indemnify an office holder with respect to such liability is provided in advance,
then such an undertaking must be limited to events which, in the opinion of the board
of directors, can be foreseen based on the company’s activities when the undertaking
to indemnify is given, and to an amount or according to criteria determined by the board
of directors as reasonable under the circumstances, and such undertaking shall detail
the abovementioned foreseen events and amount or criteria;
|
|
|
●
|
reasonable
litigation expenses, including reasonable attorneys’ fees, which were incurred
by the office holder as a result of an investigation or proceeding filed against the
office holder by an authority authorized to conduct such investigation or proceeding,
provided that such investigation or proceeding was either (i) concluded without the filing
of an indictment against such office holder and without the imposition on him of any
monetary obligation in lieu of a criminal proceeding; (ii) concluded without the filing
of an indictment against the office holder but with the imposition of a monetary obligation
on the office holder in lieu of criminal proceedings for an offense that does not require
proof of criminal intent; or (iii) in connection with a monetary sanction;
|
|
|
●
|
reasonable
litigation expenses, including attorneys’ fees, incurred by the office holder or
which were imposed on the office holder by a court (i) in a proceeding instituted against
him or her by the company, on its behalf, or by a third party, (ii) in connection with
criminal indictment of which the office holder was acquitted, or (iii) in a criminal
indictment which the office holder was convicted of an offense that does not require
proof of criminal intent;
|
|
|
●
|
a
monetary liability imposed on the office holder in favor of a payment for a breach offended
at an Administrative Procedure (as defined below) as set forth in Section 52(54)(a)(1)(a)
to the Securities Law;
|
|
|
●
|
expenses
expended by the office holder with respect to an Administrative Procedure under the Securities
Law, including reasonable litigation expenses and reasonable attorneys’ fees; and
|
|
|
●
|
any
other obligation or expense in respect of which it is permitted or will be permitted
under applicable law to indemnify an office holder, including, without limitation, matters
referenced in Section 56H(b)(1) of the Securities Law.
|
An
“Administrative Procedure” is defined as a procedure pursuant to chapters H3 (Monetary Sanction by the Israeli Securities
Authority), H4 (Administrative Enforcement Procedures of the Administrative Enforcement Committee) or I1 (Arrangement to prevent
Procedures or Interruption of procedures subject to conditions) to the Securities Law.
Under
the Companies Law and the Securities Law, a company may insure an office holder against the following liabilities incurred for
acts performed by him or her as an office holder if and to the extent provided in the company’s articles of association:
|
|
●
|
a
breach of a fiduciary duty to the company, provided that the office holder acted in good
faith and had a reasonable basis to believe that the act would not harm the company;
|
|
|
●
|
a
breach of duty of care to the company or to a third party, to the extent such a breach
arises out of the negligent conduct of the office holder;
|
|
|
●
|
a
monetary liability imposed on the office holder in favor of a third party;
|
|
|
●
|
a
monetary liability imposed on the office holder in favor of an injured party at an Administrative
Procedure pursuant to Section 52(54)(a)(1)(a) of the Securities Law; and
|
|
|
●
|
expenses
incurred by an office holder in connection with an Administrative Procedure, including
reasonable litigation expenses and reasonable attorneys’ fees.
|
Under
the Companies Law, a company may not indemnify, exculpate or insure an office holder against any of the following:
|
|
●
|
a
breach of fiduciary duty, except for indemnification and insurance for a breach of the
fiduciary duty to the company to the extent that the office holder acted in good faith
and had a reasonable basis to believe that the act would not prejudice the company;
|
|
|
●
|
a
breach of duty of care committed intentionally or recklessly, excluding a breach arising
out of the negligent conduct of the office holder;
|
|
|
●
|
an
act or omission committed with intent to derive illegal personal benefit; or
|
|
|
●
|
a
civil or administrative fine or forfeit levied against the office holder.
|
Under
the Companies Law, exculpation, indemnification and insurance of office holders must be approved by the compensation committee
and the board of directors and, with respect to directors or controlling shareholders, their relatives and third parties in which
such controlling shareholders have a personal interest, also by the shareholders.
Our
articles of association permit us to exculpate, indemnify and insure our office holders to the fullest extent permitted or to
be permitted by law. Our office holders are currently covered by a directors’ and officers’ liability insurance policy.
Employment
and consulting agreements with executive officers
We
have entered into written employment or service agreements with each of our executive officers. See “Certain Relationships
and Related Party Transactions – Employment agreements” for additional information.
Directors’
service contracts
There
are no arrangements or understandings between us, on the one hand, and any of our directors, on the other hand, providing for
benefits upon termination of their employment or service as directors of our company.
Employees
Number
of Employees
As
of December 31, 2016, we employed 35 employees: 34 in Israel, and one, our Chief Executive Officer, in the United States by U.S.
Subsidiary, Medigus USA LLC.
Distribution
of Employees
The
following is the distribution of our employees (including those employed by our subsidiary) by areas of engagement and geographic
location:
|
|
|
As of December 31,
|
|
|
|
|
2014
|
|
|
2015
|
|
|
2016
|
|
|
Numbers of employees by category of activity
|
|
|
|
|
|
|
|
|
|
|
Management and administrative
|
|
|
5
|
|
|
|
9
|
|
|
|
9
|
|
|
Research and development
|
|
|
10
|
|
|
|
12
|
|
|
|
8
|
|
|
Operations
|
|
|
8
|
|
|
|
13
|
|
|
|
8
|
|
|
Sales and marketing
|
|
|
6
|
|
|
|
9
|
|
|
|
2
|
|
|
Production
|
|
|
11
|
|
|
|
12
|
|
|
|
8
|
|
|
Total workforce
|
|
|
40
|
|
|
|
55
|
|
|
|
35
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Numbers of employees by geographic location
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Israel
|
|
|
36
|
|
|
|
47
|
|
|
|
34
|
|
|
Europe
|
|
|
1
|
|
|
|
1
|
|
|
|
-
|
|
|
United States
|
|
|
3
|
|
|
|
7
|
|
|
|
1
|
|
|
Total workforce
|
|
|
40
|
|
|
|
55
|
|
|
|
35
|
|
During
the years covered by the above table, we did not employ a significant number of temporary employees. We consider our relations
with our employees excellent and have never experienced a labor dispute, strike or work stoppage. None of our employees is represented
by a labor union.
In
Israel we are subject to certain labor statutes and national labor court precedent rulings, as well as to certain provisions of
the collective bargaining agreements between the Histadrut (General Federation of Labor in Israel) and the Coordination Bureau
of Economic Organizations including the Industrialists’ Associations. These provisions of collective bargaining agreements
are applicable to our Israeli employees by virtue of extension orders issued in accordance with relevant labor laws by the Israeli
Ministry of Economy and Industry, and which apply such agreement provisions to our employees even though they are not directly
part of a union that has signed a collective bargaining agreement. The laws and labor court rulings that apply to our employees
principally concern the minimum wage laws, length of the work day and workweek, overtime payment, procedures for dismissing employees,
determination of severance pay, leaves of absence (such as annual vacation or maternity leave), sick pay and other conditions
for employment. The extension orders which apply to our employees principally concern mandatory contributions to a pension fund
or managers' insurance, annual recreation allowance, travel expenses payment and other conditions of employment. We generally
provide our employees with benefits and working conditions beyond the required minimums.
Israeli
law generally requires severance pay, which may be funded by allocating payments to a managers’ insurance and/or a pension
fund described below, upon the retirement or death of an employee or termination of employment without cause (as defined in the
law). The payments to the managers’ insurance and/or pension fund in respect of severance pay amount to approximately 8.33%
of an employee’s wages, in the aggregate. Furthermore, Israeli employees and employers are required to pay predetermined
sums to the National Insurance Institute, which is similar to the United States Social Security Administration. Such amounts also
include payments for national health insurance. The payments to the National Insurance Institute are equal to approximately 16%
of an employee’s wages, of which the employee contributes approximately 62.5% and the employer contributes approximately
37.5%.
The
employees of U.S. Subsidiary are subject to local labor laws and regulations in the United States.
Share
Ownership
Share
ownership by Directors and Executive Officers
For
information regarding ownership of our ordinary shares by our directors and executive officers, see “Principal Shareholders.”
Share
Option Plans
All
of our option plans are administered by our board of directors. Upon the expiration of the plans, no further grants may be made
there under, although any existing awards will continue in full force in accordance with the terms under which they were granted.
Options granted under any of the plans which are currently outstanding generally may not expire later than six years from the
date of grant, unless otherwise specified. Unvested awards that are cancelled and/or forfeited go back into the respective plan.
2013
Share Option and Incentive Plan
In August 2013, our board of directors
approved and adopted our 2013 Share Option and Incentive Plan, or the 2013 Plan, which expires in August 2023. The 2013 Plan provides
for the issuance of shares and the granting of options, restricted shares, restricted share units and other share-based awards
to employees, directors, officers, consultants, advisors, and service providers of us and our U.S. Subsidiary. The Plan provides
for awards to be issued at the determination of our board of directors in accordance with applicable law. As of March 15,
2017, there were 4,700,000 ordinary shares reserved under the 2013 Plan and 1,148,964 ordinary shares issuable upon the exercise
of awards issued under the 2013 Plan:
|
Plan
|
|
Number
of options outstanding – March 15, 2017
|
|
exercise
price per one ordinary share (NIS)
|
|
Number
of shares issuable upon the exercise
|
|
Expiration
date
|
|
Series
C
|
|
3,850,000
|
|
8.3
|
|
385,000
|
|
September
30, 2018
|
|
Series
D
|
|
1,905,000
|
|
5.37
|
|
190,500
|
|
July
17, 2020
|
|
Series
F
|
|
447,075
|
|
2.05
|
|
447,075
|
|
December
29, 2021
|
|
Series
G
|
|
126,389
|
|
2.05
|
|
126,389
|
|
December
29, 2021
|
The
2013 Plan provides for the grant to residents of Israel of options that qualify under the provisions of Section 102 of the Israeli
Income Tax Ordinance (New Version) 1961, as well as for the grant of options that do not qualify under such provisions. The 2013
Plan was submitted to the ITA, as required by applicable law. The 2013 Plan also provides for the grant of options to U.S. resident
employees that are “qualified”, i.e., incentive stock options, under the U.S. Internal Revenue Code of 1986, as amended,
or the Code, and options that are not qualified. In addition to the grant of awards under the relevant tax regimes of the United
States and Israel, the 2013 Plan allows for the grant of awards to grantees in other jurisdictions, with respect to which our
board of directors is empowered to make the requisite adjustments in the plan.
Series
B Option Plan
In
February 2012, we adopted our Series B Option Plan. Our directors, officers, employees and certain consultants and dealers were
eligible to participate in this plan. As of March 15, 2017, there were 82,000 ordinary shares issuable upon the exercise of outstanding
options under the plan. These options have an expiration date of April 23, 2018. No additional options will be issued under this
plan. Israeli grantees who were directors, officers and employees could be granted options under the plan that would qualify for
special tax treatment under the Capital Gains Route. This plan was submitted to the ITA as required by applicable law. The exercise
price for the Series B Options is NIS 6.8 per share, linked to the consumer price index as of February 6 of each calendar year
following the grant of the Series B Options, compared to the known consumer price index as of February 6 of the previous calendar
year or as of the date of grant. If the rate of increase of the consumer price index is higher than 3%, the exercise price is
increased by the difference between 3% and the rate of increase of the consumer price index.
Series
A Option Plan
In
August 2011, we adopted our Series A Option Plan. Our directors, officers, employees and certain consultants and dealers were
eligible to participate in this plan. As of March 15, 2017, there were 116,000 ordinary shares issuable upon the exercise of outstanding
options under the plan. These options have an expiration date of between November 2, 2017 and April 23, 2018. No additional options
will be issued under this plan. Israeli grantees who were directors, officers and employees could be granted options under the
plan that would qualify for special tax treatment in Israel. This plan was submitted to the ITA, as required by applicable law.
The exercise price for the Series A Options is NIS 9.1 per share, linked to the consumer price index as of August 1 of each calendar
year following the grant of the Series A Options, compared to the known consumer price index as of August 1 of the previous calendar
year or as of the date of grant. If the rate of increase of the consumer price index is higher than 3%, the exercise price is
increased by the difference between 3% and the rate of increase of the consumer price index.
Principal
Shareholders
Beneficial
Ownership
The
following table sets forth information with respect to the beneficial ownership of our ordinary shares as of March 15, 2017, by:
|
|
●
|
each
person or entity known by us to own beneficially more than 5% of our outstanding ordinary
shares;
|
|
|
●
|
each
of our directors and executive officers individually; and
|
|
|
●
|
all
of our executive officers and directors as a group.
|
The
beneficial ownership of our ordinary shares is determined in accordance with the rules of the SEC. Under these rules, a person
is deemed to be a beneficial owner of a security if that person has or shares voting power, which includes the power to vote or
to direct the voting of the security, or investment power, which includes the power to dispose of or to direct the disposition
of the security. For purposes of the table below, we deem ordinary shares issuable pursuant to options that are currently exercisable
or exercisable within 60 days as of March 15, 2017, if any, to be outstanding and to be beneficially owned by the person holding
the options or warrants for the purposes of computing the percentage ownership of that person, but we do not treat them as outstanding
for the purpose of computing the percentage ownership of any other person. The percentage of ordinary shares beneficially owned
prior to the offering is based on
44,142,884
ordinary shares outstanding as of March
15, 2017. The percentage of ordinary shares beneficially owned after the offering is based on the number of shares outstanding
prior to the offering plus the ordinary shares that we are selling in this offering.
Except
where otherwise indicated, we believe, based on information furnished to us by such owners, that the beneficial owners of the
ordinary shares listed below have sole investment and voting power with respect to such shares.
Upon
the closing of this offering, none of our shareholders will have different voting rights from other shareholders. To the best
of our knowledge, we are not owned or controlled, directly or indirectly, by another corporation or by any foreign government.
We are not aware of any arrangement that may, at a subsequent date, result in a change of control of our company.
As
of March 15, 2017, there was one shareholder of record of our ordinary shares, which was located in Israel. The number of record
holders is not representative of the number of beneficial holders of our ordinary shares, as the shares of all shareholders for
a publicly traded company such as ours which is listed on the Tel Aviv Stock Exchange are recorded in the name of our Israeli
share registrar, Bank Hapoalim Registration Company Ltd.
Unless
otherwise noted below, each beneficial owner’s address is Medigus Ltd., Omer Industrial Park, No. 7A, P.O. Box 3030, Omer
8496500, Israel.
|
Name of Beneficial
Owner
|
|
Number
of Shares Beneficially Owned
|
|
|
Percentage
of Shares Beneficially Owned Before This Offering
|
|
|
Percentage of
Shares Beneficially
Owned
After this
Offering
|
|
|
5% or greater shareholders
|
|
|
|
|
|
|
|
|
|
|
Orbimed
Israel GP Ltd.
(1)
|
|
|
7,407,282
|
|
|
|
16.39
|
%
|
|
|
8.08
|
%
|
|
Migdal
Insurance & Financial Holdings Ltd.
(2)
|
|
|
5,316,509
|
|
|
|
11.72
|
%
|
|
|
5.79
|
%
|
|
Senvest
Management LLC
(3)
|
|
|
6,545,670
|
|
|
|
14.38
|
%
|
|
|
7.12
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Directors and executive
officers
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Christopher (Chris) Rowland
|
|
|
*
|
|
|
|
*
|
|
|
|
*
|
|
|
Minelu
(Menashe) Sonnenschein
(4)
|
|
|
631,243
|
|
|
|
1.43
|
%
|
|
|
*
|
|
|
Aviel
Roy-Shapira
(5)
|
|
|
672,204
|
|
|
|
1.52
|
%
|
|
|
*
|
|
|
Oded Yatzkan
|
|
|
*
|
|
|
|
*
|
|
|
|
*
|
|
|
Yaron Silberman
|
|
|
*
|
|
|
|
*
|
|
|
|
*
|
|
|
Dr. Nissim Darvish
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Anat Naschitz
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Efrat Venkert
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Doron Birger
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Eitan Machover
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
All
directors and executive officers as a group (10 persons)
(6)
|
|
|
1,813,072
|
|
|
|
4.06
|
%
|
|
|
1.99
|
%
|
*
less than 1%.
|
(1)
|
Consists
of 6,352,758 ordinary shares and warrants to purchase 1,054,524 ordinary shares exercisable within 60 days as of March 15,
2017. Based on a Schedule 13D/A filed with the SEC on December 8, 2016, by OrbiMed Israel GP Ltd., an Israeli limited liability
company, or OrbiMed Israel, and OrbiMed Israel BioFund GP Limited Partnership, an Israeli limited partnership, or OrbiMed
BioFund
,
OrbiMed Israel is the general partner of OrbiMed BioFund, which is the
general partner of OrbiMed Israel Partners Limited Partnership, an Israeli limited partnership, which holds the securities.
The address of the principal office of each OrbiMed Israel and OrbiMed BioFund is 89 Medinat HaYehudim St., Build E, 11
th
Floor,
Herzliya 46766 Israel.
|
|
(2)
|
Based
on information provided to us, consists of 4,090,412 ordinary shares and warrants to purchase 1,226,097 ordinary shares exercisable
within 60 days as of March 15, 2017. The principal business address of Migdal Insurance & Financial Holdings Ltd. is 4
Efal Street, Petach Tikva 49512, Israel, P.O. Box 3063.
|
|
(3)
|
Based
on a Schedule 13G/A filed with the SEC on February 13, 2017, as of December 31, 2016, Senvest Management LLC held shared voting
power and shared dispositive power over 6,545,670 shares, including 1,363,049 Ordinary Shares issuable upon exercise of warrants.
The address of each Senvest Management, LLC and Richard Mashaal is 540 Madison Avenue, 32
nd
Floor, New York,
New York 10022.
|
|
(4)
|
Consists
of 598,243 ordinary shares and options to purchase 33,000 ordinary shares exercisable within 60 days as of March 15, 2017.
|
|
(5)
|
Consists
of 662,829 ordinary shares and options to purchase 9,375 ordinary shares exercisable within 60 days as of March 15, 2017.
|
|
(6)
|
Consists
of 1,261,072 ordinary shares and options to purchase 552,000 ordinary shares exercisable within 60 days as of March 15, 2017.
|
Significant
Changes
in Percentage Ownership by Major Shareholders
To
our knowledge, the significant changes in the percentage of ownership held by our major shareholders during the past three
years have been:
(i)
On November 30, 2016, we entered into a securities purchase agreements as part of an offering of our ADSs pursuant to our
shelf registration statement in the United States. In connection with the offering, CVI Investments, Inc., purchased 56,958
ADSs (representing 2,847,925 of our ordinary shares) and warrants to purchase up to 19,935 ADSs (representing 996,775 of our ordinary
shares) and Empery Asset Master, Ltd., Empery Tax Efficient, LP, and Empery Tax Efficient, II LP, collectively
Empery
Asset Management LP
, purchased and aggregate of 56,958 of our ADSs representing 2,847,925 of our ordinary shares and warrants
to purchase up to 19,935 ADSs.
(ii) On September 8, 2016, we entered into
a securities purchase agreements as part of an offering of our ADSs pursuant to our shelf registration statement in the United
States. As part of the offering, Senvest Management LLC purchased 15,000 ADSs (representing 750,000 of our ordinary shares) and
Sabby Healthcare Master Fund Ltd. purchased 113,000 of our ADSs representing 5,650,000 of our ordinary shares.
(iii)
On July 8, 2015, we completed a public offering of ordinary shares and warrants under a shelf offering report issued pursuant
to our shelf prospectus in Israel. Subsequent to the offering, we were informed that OrbiMed Israel GP Ltd. purchased a total
of 1,496,000 of our ordinary shares at a price of NIS 3.8 per share and 7,480,000 of our warrants, exercisable into additional
748,000 of our ordinary shares, affiliates of the Senvest Management LLC purchased an aggregate of 1,500,000 of our ordinary
shares at a price of NIS 3.8 per share and 7,500,000 of our warrants, exercisable into additional 750,000 of our ordinary shares
and affiliates of Migdal Insurance & Financial Holdings Ltd. purchased 842,000 of our ordinary shares at a price of NIS
3.8 per share and 4,210,000 of our warrants, exercisable into additional 421,000 of our ordinary shares.
(iv)
In June 2014 we entered into securities purchase agreements with affiliates of OrbiMed Israel GP Ltd., Sabby Healthcare Master
Fund Ltd., Senvest Management LLC and Migdal Insurance & Financial Holdings Ltd. Under these securities purchase agreements,
we issued affiliates of Orbimed Israel GP Ltd. an aggregate of 766,310 of our ordinary shares and a total of 3,065,244 warrants
exercisable for 306,524 of our ordinary shares, Sabby Healthcare Master Fund Ltd. an aggregate of 1,247,200 of our ordinary shares
and a total of 4,988,800 warrants exercisable for 498,880 of our ordinary shares, Senvest Management LLC an aggregate of 1,532,622
of our ordinary shares and a total of 6,130,487 warrants exercisable for 613,048 of our ordinary shares and Migdal Insurance &
Financial Holdings Ltd. an aggregate of 3,065,244 of our ordinary shares and a total of 12,260,974 warrants exercisable for 1,226,097
of our ordinary shares.
(v) We have been informed that Sabby Healthcare
Master Fund Ltd., CVI Investments, Inc.
, Empery Asset Management LP, Armistice Capital
Master Fund Ltd. and Oren Dan have sold their holdings in the Company and are no longer beneficial owners of 5% or more of the
share capital of the Company.
Certain
Relationships and Related Party Transactions
Employment
Agreements
We
have entered into written employment agreements with each of our executive officers. All of these agreements contain customary
provisions regarding noncompetition, confidentiality of information and assignment of inventions. However, the enforceability
of the noncompetition provisions may be limited under applicable law. In addition, we have entered into agreements with each executive
officer and director pursuant to which we have agreed to indemnify each of them to the fullest extent permitted by law to the
extent that these liabilities are not covered by directors and officers insurance.
Our
office holders are generally eligible for bonuses each year. The bonuses are established and granted in accordance with our compensation
policy and, and are generally payable upon meeting objectives and targets that are approved by our compensation committee and
board of directors (and if required by our shareholders).
For
further information see
“Management—Employment and consulting agreements with
executive officers.”
Directors
and Officers Insurance Policy and Indemnification Agreements
Our
articles of association permit us to exculpate, indemnify and insure our directors and officeholders to the fullest extent permitted
by the Companies Law.
We
have entered into agreements with each of our current director and officers exculpating them from a breach of their duty of care
to us to the fullest extent permitted by law, subject to limited exceptions, and undertaking to indemnify them to the fullest
extent permitted by law, to the extent that these liabilities are not covered by insurance. This indemnification is limited, with
respect to any monetary liability imposed in favor of a third party, to events determined as foreseeable by the board of directors
based on our activities. The maximum aggregate amount of indemnification that we may pay to our directors and officers based on
such indemnification agreement is equal to 25% of our shareholders’ equity pursuant to our latest audited or unaudited consolidated
financial statements, as applicable, as of the date of the indemnification payment. Such indemnification amounts are in addition
to any insurance amounts. Each director or officer who agrees to receive this letter of indemnification also gives his approval
to the termination of all previous letters of indemnification that we have provided to him or her in the past, if any.
We
currently have directors’ and officers’ liability insurance providing total coverage of $15 million for the benefit
of all of our directors and officers, in respect of which we are charged a twelve-month premium of $ 63,745, and which includes
a deductible of up to $50,000 per claim, other than securities related claims filed in the United States or Canada, for which
the deductible shall not exceed $200,000.
In
addition, at general meeting of our shareholders held on December 29, 2015, our shareholders approved our compensation policy,
which determines, among others, that we may provide our directors and officers, including those serving in any of our subsidiaries
from time or time and those who are controlling shareholders, with liability insurance policies provided that the engagement is
in the ordinary course of business, in market terms and is not expected to materially influence our profits, properties and undertakings.
The coverage limit shall be of up to $20 million per occurrence and for the insurance period (additional coverage for legal expenses
not included), provided that the annual premium shall not exceed $80,000 and that the deductible (except for extraordinary matters
as prescribed in the insurance policy, such as lawsuits against the Company pursuant to securities laws and/or lawsuits to be
filed in the US/Canada) shall not exceed $50,000 per occurrence.
For
further information see
“Management—Exemption, Insurance and Indemnification
of Directors and Officers.”
Material
Contracts
Securities Purchase Agreement, dated November 30, 2016
On November 30, 2016, we entered into a securities
purchase agreements as part of an offering of our ADSs pursuant to our shelf registration statement in the United States. As part
of the offering we issued a total of 113,917 of our ADSs representing a total of 5,695,850 ordinary shares, at a purchase price
of US$6.70 per ADS, and warrants to purchase up to a total of 39,871 ADSs representing 1,993,550 ordinary shares, at an initial
exercise price of $9.00 per ADS, in a concurrent private placement. Pursuant to the engagement letter executed with the placement
agent, Rodman & Renshaw, relating to the offering, we agreed to provide the placement agent with the right of first refusal,
expiring on the twelve month anniversary following the closing of the offering, if we or our subsidiaries decide to raise funds
by means of a public offering or a private placement of equity or debt securities using an underwriter or placement agent in the
U.S.
Securities
Purchase Agreement, dated September 8, 2016
On September 8, 2016, we entered into a securities
purchase agreements as part of an offering of our ADSs pursuant to our shelf registration statement in the United States. As part
of the offering we issued a total of 128,000 of our ADSs representing a total of 6,400,000 ordinary shares, at a purchase price
of $11.50 per ADS.
Securities
Purchase Agreements, dated June 2014
In June 2014, we entered into securities
purchase agreements with certain investors, including Sabby Healthcare Volatility Master Fund Ltd., Sabby Volatility Warrant Master
Fund Ltd., Senvest Israel Partners LP, Senvest International LLC, Migdal Insurance Company Ltd., Capital Point Ltd. and OrbiMed.
Under these securities purchase agreements, the investors invested an aggregate amount of approximately $11.1 million (approximately
NIS 39 million). In connection with the securities purchase agreements, we issued an aggregate amount of 8,527,154 ordinary shares
for a price per ordinary share of NIS 4.4786, and warrants, exercisable into an additional 3,410,861 ordinary shares for a period
of 36 months from their date of issuance. Each ten warrants are exercisable into on ordinary share at a price per share of
NIS 6.27. If at any time after the six-month anniversary of the closing date of the securities purchase agreements, the ordinary
shares held by the investors have not been released from the applicable Israeli or U.S. transfer restrictions (i.e. via the filing
of an Israeli prospectus or the registration of the securities in the United States), and subject to certain other conditions
set forth in the warrants, then the warrants (other than those held by Migdal Insurance Company Ltd.) may also be exercised, in
whole or in part, by means of a cashless exercise.
Shareholders’
Undertaking to Orbimed
In
January 2013, we entered into a share purchase agreement with OrbiMed pursuant to which we issued OrbiMed an aggregate amount
of 3,994,547 ordinary shares and warrants to purchase 3,994,547 additional ordinary shares, for a total consideration of $8 million.
The warrants have expired on March 3, 2016
. In
connection with the share purchase agreement, several of the Company’s shareholders, including Menashe Sonnenschein and
Aviel Shapira signed a letter of undertaking to OrbiMed, or the Shareholders’ Undertaking, according to which each undertook,
until the earlier of: (i) the three year anniversary of the closing date; or (ii) the date on which OrbiMed’s voting rights
exceed 45% of the aggregate voting rights of the Company, to vote their shares in accordance with OrbiMed’s instructions
in connection with certain matters such as the size of the board, to elect four directors nominated by OrbiMed (including the
Chairman) and for any other matter in accordance with OrbiMed’s instructions provided that such matter is not ‘unreasonable’.
The shareholders subject to the undertaking also agreed to certain transfer restrictions on their shares during the three year
period. On March 3, 2016, the Shareholders’ Undertaking has expired.
Description
of Share Capital
The
following description of our share capital and provisions of our amended and restated articles of association are summaries and
do not purport to be complete.
General
Our
legal and commercial name is Medigus Ltd. We were incorporated in the State of Israel on December 9, 1999, as a private company
pursuant to the Israeli Companies Ordinance (New Version), 1983. In February 2006, we completed our initial public offering in
Israel, and our ordinary shares have since traded on the TASE, under the symbol “MDGS”. In May 2015, we listed our
ADSs on NASDAQ, and since August 2015 our ADSs have been traded on the NASDAQ under the symbol “MDGS”. Each ADS
represents 50 ordinary shares.
Our authorized share capital consists of
800,000,000 ordinary shares, par value NIS 0.10 per share. As of March 15, 2017, 44,142,884 ordinary shares were issued and outstanding.
All of our outstanding ordinary shares have been fully paid and non-assessable. Holders of paid-up ordinary shares are entitled
to participate equally in the payment of dividends and other distributions and, in the event of liquidation, in all distributions
after the discharge of liabilities to creditors.
Our ordinary shares are not redeemable.
Pursuant to the terms of the securities purchase
agreements relating to an offering of our ADSs and warrants which closed on December 6, 2016, we granted the purchasers in such
offering an eighteen month contractual preemptive right to participate in future financings, subject to certain exceptions, in
an amount equal to up to 50% of the amount raised in such future financings. Other than the preemptive rights described herein,
our ordinary shares are not subject to any other preemptive rights.
Options
As of March 15, 2017, options to purchase
an aggregate of 1,346,964 ordinary shares have been granted under our share option plans. See “Business – Share ownership.”
Warrants
As
of March 15, 2017, the following warrants are outstanding:
|
|
●
|
Warrants
publicly traded on the TASE to purchase an aggregate of 3,526,250 ordinary shares at an exercise price per ordinary share
of NIS
5.32
, approximately $1.45. These warrants expire on
July
8, 2018
;
|
|
|
●
|
Unregistered
warrants to purchase an aggregate of
3,410,861
ordinary shares at an exercise
price per ordinary share of NIS 6
.27
, approximately $1.71. These warrants expire
in August 2017.
|
|
|
●
|
Unregistered
warrants to purchase an aggregate of 3,955 ADSs at an exercise price per ADS of $
14.375
.
These warrants expire on
September 8, 2021
.
|
|
|
●
|
Unregistered
warrants to purchase an aggregate of
41,864
ADSs at an exercise price per ADS
of $9.00. These warrants expire on June 6, 2022.
|
|
|
●
|
Unregistered
warrants to purchase an aggregate of 3,987 ADSs at an exercise price per ADS of $
7.37
.
These warrants expire on December 6, 2021.
|
Registration
Number and Purposes of the Company
Our
registration number with the Israeli Registrar of Companies is 51-286697-1. Our purpose as set forth in our articles of association
is to engage in any lawful activity.
Transfer
of shares
Our
fully paid ordinary shares are issued in registered form and may be freely transferred under our articles of association, unless
the transfer is restricted or prohibited by another instrument, applicable law or the rules of a stock exchange on which the shares
are listed for trade. The ownership or voting of our ordinary shares by non-residents of Israel is not restricted in any way by
our articles of association or the laws of the State of Israel, except for ownership by nationals of certain countries that are,
or have been, in a state of war with Israel.
Liability
to further capital calls
Our
board of directors may make, from time to time, such calls as it may deem fit upon shareholders with respect to any sum unpaid
with respect to shares held by such shareholders which is not payable at a fixed time. Such shareholder shall pay the amount of
every call so made upon him or her.
Election
of Directors
Our
ordinary shares do not have cumulative voting rights for the election of directors. As a result, the holders of a majority of
the voting power represented at a shareholders meeting have the power to elect all of our directors, subject to the special approval
requirements for external directors under the Israeli Companies Law, 5759-1999, or the Companies Law, under “Management
– External directors.”
Under
our articles of association, our board of directors must consist of not less than three but no more than twelve directors, including
two external directors as required by the Companies Law. Pursuant to our articles of association, other than the external directors,
for whom special election requirements apply under the Companies Law, the vote required to appoint a director is a simple majority
vote of holders of our voting shares participating and voting at the relevant meeting. In addition, our articles of association
allow our board of directors to appoint new directors to fill vacancies on the board of directors to serve until the subsequent
annual general meeting of our shareholders, provided, that the number of directors shall not exceed twelve directors. External
directors are elected for an initial term of three years, may be elected for additional terms of three years each under certain
circumstances, and may be removed from office pursuant to the terms of the Companies Law. For further information on the election
and removal of external directors see “Management – External directors.”
Dividend
and liquidation rights
We
may declare a dividend to be paid to the holders of our ordinary shares in proportion to their respective shareholdings. Under
the Companies Law, dividend distributions are determined by the board of directors and do not require the approval of the shareholders
of a company unless the company’s articles of association provide otherwise. Our articles of association do not require
shareholder approval of a dividend distribution and provide that dividend distributions may be determined by our board of directors.
Pursuant
to the Companies Law, the distribution amount is limited to the greater of retained earnings or earnings generated over the previous
two years, according to our then last reviewed or audited consolidated financial statements, provided that the date of the financial
statements is not more than six months prior to the date of the distribution, or we may distribute dividends that do not meet
such criteria only with court approval. In each case, we are only permitted to distribute a dividend if our board of directors
and the court, if applicable, determines that there is no reasonable concern that payment of the dividend will prevent us from
satisfying our existing and foreseeable obligations as they become due.
In
the event of our liquidation, after satisfaction of liabilities to creditors, our assets will be distributed to the holders of
our ordinary shares in proportion to their shareholdings. This right, as well as the right to receive dividends, may be affected
by the grant of preferential dividend or distribution rights to the holders of a class of shares with preferential rights that
may be authorized in the future.
Exchange
controls
There
are currently no Israeli currency control restrictions on remittances of dividends on our ordinary shares, proceeds from the sale
of the shares or interest or other payments to non-residents of Israel, except for shareholders who are subjects of certain countries
that are, or have been, in a state of war with Israel.
Shareholder
Meetings
Under
the Companies Law, we are required to hold an annual general meeting of our shareholders once every calendar year that must be
held no later than 15 months after the date of the previous annual general meeting. All general meetings other than the annual
meeting of shareholders are referred to in our articles of association as extraordinary meetings. Our board of directors may call
extraordinary meetings whenever it sees fit, at such time and place, within or outside of Israel, as it may determine. In addition,
the Companies Law provides that our board of directors is required to convene a special meeting upon the written request of (i)
any two of our directors or one-quarter of the members of our board of directors or (ii) one or more shareholders holding, in
the aggregate, either (a) 5% or more of our outstanding issued shares and 1% or more of our outstanding voting power or (b) 5%
or more of our outstanding voting power.
Under
the Companies Law, one or more shareholders holding at least 1% of the voting rights at the general meeting may request that the
board of directors include a matter in the agenda of a general meeting to be convened in the future, provided that it is appropriate
to discuss such a matter at the general meeting.
Subject
to the provisions of the Companies Law and the regulations promulgated thereunder, shareholders entitled to participate and vote
at general meetings are the shareholders of record on a date to be decided by the board of directors, which may be between four
and 40 days prior to the date of the meeting. Furthermore, the Companies Law requires that resolutions regarding the following
matters must be passed at a general meeting of our shareholders:
|
|
●
|
amendments
to our articles of association;
|
|
|
|
|
|
|
●
|
appointment
or termination of our auditors;
|
|
|
●
|
appointment
of external directors;
|
|
|
|
|
|
|
●
|
approval
of certain related party transactions;
|
|
|
|
|
|
|
●
|
increases
or reductions of our authorized share capital;
|
|
|
●
|
mergers;
and
|
|
|
|
|
|
|
●
|
the
exercise of our board of directors powers by a general meeting, if our board of directors is unable to exercise its powers
and the exercise of any of its powers is required for our proper management.
|
Under
our articles of association, we are not required to give notice to our registered shareholders pursuant to the Companies Law,
unless otherwise required by law. The Companies Law requires that a notice of any annual general meeting or extraordinary general
meeting be provided to shareholders at least 21 days prior to the meeting and if the agenda of the meeting includes the appointment
or removal of directors, the approval of transactions with office holders or interested or related parties, or an approval of
a merger, or as otherwise required under applicable law, notice must be provided at least 35 days prior to the meeting.
Voting
rights
Voting
rights
All
our ordinary shares have identical voting and other rights in all respects.
Quorum
requirements
Pursuant
to our articles of association, holders of our ordinary shares have one vote for each ordinary share held on all matters submitted
to a vote before the shareholders at a general meeting. The quorum required for our general meetings of shareholders consists
of at least two shareholders, present in person or by proxy, holding at least ten percent (10%) of the voting rights of the Company.
A meeting adjourned for lack of a quorum will be adjourned to the same day of the following week at the same time and place, or
to such other day, time or place if such is stated in the notice of the meeting. At the reconvened meeting, if a quorum is not
present within an half an hour, any number of shareholders present in person or by proxy shall constitute a lawful quorum.
Vote
requirements
Our
articles of association provide that all resolutions of our shareholders require a simple majority vote, unless otherwise required
by the Companies Law or by our articles of association. Under the Companies Law, each of (i) the approval of an extraordinary
transaction with a controlling shareholder and (ii) the terms of employment or other engagement of the controlling shareholder
of the company or such controlling shareholder’s relative (even if not extraordinary) requires the approval described under
“Management – Fiduciary duties and approval of specified related party transactions and compensation under Israeli
law – Disclosure of personal interests of a controlling shareholder and approval of transactions.” Certain transactions
with respect to remuneration of our office holders and directors require further approvals described under “Management –
Fiduciary duties and approval of specified related party transactions and compensation under Israeli law – Approval of compensation
of directors and executive officers.” Another exception to the simple majority vote requirement is a resolution for the
voluntary winding up, or an approval of a scheme of arrangement or reorganization, of the company pursuant to Section 350 of the
Companies Law, which requires the approval of the majority of the shareholders voting their shares, other than abstainees, holding
at least 75% of the voting rights represented at the meeting, in person, by proxy or by voting deed and voting on the resolution.
Access
to corporate records
Under
the Companies Law, shareholders are provided access to minutes of our general meetings, our shareholders register and principal
shareholders register, our articles of association, our financial statements and any document that we are required by law to file
publicly with the Israeli Companies Registrar or the Israel Securities Authority. In addition, shareholders may request to be
provided with any document related to an action or transaction requiring shareholder approval under the related party transaction
provisions of the Companies Law. We may deny this request if we believe it has not been made in good faith or if such denial is
necessary to protect our interest or protect a trade secret or patent.
Modification
of class rights
Under
the Companies Law and our articles of association, the rights attached to any class of shares, such as voting, liquidation and
dividend rights, may be amended by adoption of a resolution by the holders of a majority of the shares of that class present at
a separate class meeting, or otherwise in accordance with the rights attached to such class of shares, as set forth in our articles
of association.
Acquisitions
under Israeli law
Full
tender offer
A
person wishing to acquire shares of an Israeli public company and who would as a result hold over 90% of the target company’s
issued and outstanding share capital is required by the Companies Law to make a tender offer to all of the company’s shareholders
for the purchase of all of the issued and outstanding shares of the company. A person wishing to acquire shares of a public Israeli
company and who would as a result hold over 90% of the issued and outstanding share capital of a certain class of shares is required
to make a tender offer to all of the shareholders who hold shares of the relevant class for the purchase of all of the issued
and outstanding shares of that class. If the shareholders who do not accept the offer hold less than 5% of the issued and outstanding
share capital of the company or of the applicable class, and more than half of the shareholders who do not have a personal interest
in the offer accept the offer, all of the shares that the acquirer offered to purchase will be transferred to the acquirer by
operation of law. However, a tender offer will also be accepted if the shareholders who do not accept the offer hold less than
2% of the issued and outstanding share capital of the company or of the applicable class of shares.
Upon
a successful completion of such a full tender offer, any shareholder that was an offeree in such tender offer, whether such shareholder
accepted the tender offer or not, may, within six months from the date of acceptance of the tender offer, petition an Israeli
court to determine whether the tender offer was for less than fair value and that the fair value should be paid as determined
by the court. However, under certain conditions, the offeror may include in the terms of the tender offer that an offeree who
accepted the offer will not be entitled to petition the Israeli court as described above.
If
a tender offer is not accepted in accordance with the requirements set forth above, the acquirer may not acquire shares of the
company that will increase its holdings to more than 90% of the company’s issued and outstanding share capital or of the
applicable class from shareholders who accepted the tender offer.
Special
tender offer
The
Companies Law provides that an acquisition of shares of an Israeli public company must be made by means of a special tender offer
if as a result of the acquisition the purchaser would become a holder of 25% or more of the voting rights in the company. This
requirement does not apply if there is already another holder of at least 25% of the voting rights in the company. Similarly,
the Companies Law provides that an acquisition of shares in a public company must be made by means of a special tender offer if
as a result of the acquisition the purchaser would become a holder of more than 45% of the voting rights in the company, if there
is no other shareholder of the company who holds more than 45% of the voting rights in the company, subject to certain exceptions.
A
special tender offer must be extended to all shareholders of a company but the offeror is not required to purchase shares representing
more than 5% of the voting power attached to the company’s outstanding shares, regardless of how many shares are tendered
by shareholders. A special tender offer may be consummated only if (i) at least 5% of the voting power attached to the company’s
outstanding shares will be acquired by the offeror and (ii) the number of shares tendered in the offer exceeds the number of shares
whose holders objected to the offer (excluding the purchaser and its controlling shareholder, holders of 25% or more of the voting
rights in the company or any person having a personal interest in the acceptance of the tender offer or any other person acting
on their behalf, including relatives and entities under such person’s control). If a special tender offer is accepted, then
the purchaser or any person or entity controlling it or under common control with the purchaser or such controlling person or
entity may not make a subsequent tender offer for the purchase of shares of the target company and may not enter into a merger
with the target company for a period of one year from the date of the offer, unless the purchaser or such person or entity undertook
to effect such an offer or merger in the initial special tender offer.
Merger
The
Companies Law permits merger transactions if approved by each party’s board of directors and, unless certain requirements
described under the Companies Law are met, by a majority vote of each party’s shares, and, in the case of the target company,
a majority vote of each class of its shares voted on the proposed merger at a shareholders meeting.
For
purposes of the shareholder vote, unless a court rules otherwise, the merger will not be deemed approved if a majority of the
votes of the shares represented at the shareholders meeting that are held by parties other than the other party to the merger,
or by any person (or group of persons acting in concert) who holds (or hold, as the case may be) 25% or more of the voting rights
or the right to appoint 25% or more of the directors of the other party, vote against the merger. If, however, the merger involves
a merger with a company’s own controlling shareholder or if the controlling shareholder has a personal interest in the merger,
then the merger is instead subject to the same special majority approval that governs all extraordinary transactions with controlling
shareholders (as described under “Management – Fiduciary duties and approval of specified related party transactions
and compensation under Israeli law – Disclosure of personal interests of a controlling shareholder and approval of transactions”).
If
the transaction would have been approved by the shareholders of a merging company but for the separate approval of each class
or the exclusion of the votes of certain shareholders as provided above, a court may still approve the merger upon the request
of holders of at least 25% of the voting rights of a company, if the court holds that the merger is fair and reasonable, taking
into account the value to the parties to the merger and the consideration offered to the shareholders of the company.
Upon
the request of a creditor of either party to the proposed merger, the court may delay or prevent the merger if it concludes that
there exists a reasonable concern that, as a result of the merger, the surviving company will be unable to satisfy the obligations
of the merging entities, and may further give instructions to secure the rights of creditors.
In
addition, a merger may not be consummated unless at least 50 days have passed from the date on which a proposal for approval of
the merger was filed by each party with the Israeli Registrar of Companies and at least 30 days have passed from the date on which
the merger was approved by the shareholders of each party.
Borrowing
powers
Pursuant
to the Companies Law and our articles of association, our board of directors may exercise all powers and take all actions that
are not required under law or under our articles of association to be exercised or taken by a certain organ of the Company, including
the power to borrow money for company purposes.
Changes
in capital
Our
articles of association enable us to increase or reduce our share capital. Any such changes are subject to the provisions of the
Companies Law and must be approved by a resolution duly adopted by our shareholders at a general meeting. In addition, transactions
that have the effect of reducing capital, such as the declaration and payment of dividends in the absence of sufficient retained
earnings or profits, require the approval of both our board of directors and an Israeli court.
Transfer
agent and registrar
Our
transfer agent and registrar is the Depositary for our ADSs, Bank of New York Mellon, and its address is 101 Barclay Street, New
York, NY.
Listing
Our ordinary shares currently trade on the
TASE in Israel under the symbol “MDGS,” and our ADSs are listed on NASDAQ under the symbol “MDGS.” The
Series A warrants and Series B warrants are not and will not be listed for trading on any national securities exchange.
DESCRIPTION
OF THE OFFERED SECURITIES
AMERICAN
DEPOSITARY SHARES
General
The following is a summary description of
the ADSs and does not purport to be complete. Each ADS represents 50 ordinary shares (or a right to receive 50 ordinary shares)
deposited with the principal Tel Aviv office of either of Bank Hapoalim or Bank Leumi, as custodian for the Bank of New York Mellon
as the Depositary. Each ADS also represents any other securities, cash or other property which may be held by the Depositary.
The Depositary’s office at which the ADSs will be administered is located at 101 Barclay Street, New York, New York 10286.
The Bank of New York Mellon’s principal executive office is located at 225 Liberty Street, New York, New York 10286.
You
may hold ADSs either (A) directly (i) by having an American Depositary Receipt, or ADR, which is a certificate evidencing a specific
number of ADSs, registered in your name, or (ii) by having uncertificated ADSs registered in your name, or (B) indirectly by holding
a security entitlement in ADSs through your broker or other financial institution. If you hold ADSs directly, you are a registered
ADS holder, also referred to as an ADS holder. This description assumes you are an ADS holder. If you hold the ADSs indirectly,
you must rely on the procedures of your broker or other financial institution to assert the rights of ADS holders described in
this section. You should consult with your broker or financial institution to find out what those procedures are.
Registered
holders of uncertificated ADSs will receive statements from the Depositary confirming their holdings. As an ADS holder, we will
not treat you as one of our shareholders and you will not have shareholder rights. Israeli law governs shareholder rights. The
Depositary will be the holder of the shares underlying your ADSs. As a registered holder of ADSs, you will have ADS holder rights.
A deposit agreement among us, the Depositary, ADS holders and all other persons indirectly or beneficially holding ADSs sets out
ADS holder rights as well as the rights and obligations of the Depositary. New York law governs the deposit agreement and the
ADSs.
The
following is a summary of the material provisions of the deposit agreement. For more complete information, you should read the
entire deposit agreement and the form of ADR.
Dividends
and Other Distributions
How
will you receive dividends and other distributions on the shares?
The
Depositary has agreed to pay to ADS holders the cash dividends or other distributions it or the custodian receives on shares or
other deposited securities, after deducting its fees and expenses. You will receive these distributions in proportion to the number
of shares your ADSs represent.
Cash
.
The Depositary will convert any cash dividend or other cash distribution we pay on the shares into U.S. dollars, if it can do
so on a reasonable basis and can transfer the U.S. dollars to the United States. If that is not possible or if any government
approval is needed and cannot be obtained, the deposit agreement allows the Depositary to distribute the NIS only to those ADS
holders to whom it is possible to do so. It will hold the NIS it cannot convert for the account of the ADS holders who have not
been paid. It will not invest the NIS and it will not be liable for any interest.
Before
making a distribution, any withholding taxes, or other governmental charges that must be paid will be deducted. For more information
see “
Material Tax Considerations
.” The Depositary will distribute only
whole U.S. dollars and cents and will round fractional cents to the nearest whole cent.
If the exchange rates fluctuate during
a time when the Depositary cannot convert the NIS, you may lose some or all of the value of the distribution.
Shares.
The Depositary may distribute additional ADSs representing any shares we distribute as a dividend or free distribution. The
Depositary will only distribute whole ADSs. It will sell shares which would require it to deliver a fraction of an ADS (or ADSs
representing those shares) and distribute the net proceeds in the same way as it does with cash. If the Depositary does not distribute
additional ADSs, the outstanding ADSs will also represent the new shares. The Depositary may sell a portion of the distributed
shares sufficient to pay its fees and expenses in connection with that distribution (or ADSs representing those shares).
Rights
to purchase additional shares.
If we offer holders of our securities any rights to subscribe for additional ordinary shares
or any other rights, the Depositary may make these rights available to ADS holders. If the Depositary decides it is not legal
and practical to make the rights available but that it is practical to sell the rights, the Depositary will use reasonable efforts
to sell the rights and distribute the proceeds in the same way as it does with cash. The Depositary will allow rights that are
not distributed or sold to lapse.
In that case, you will receive no value for them.
If
the Depositary makes rights available to ADS holders, it will exercise the rights and purchase the shares on your behalf. The
Depositary will then deposit the shares and deliver ADSs to the persons entitled to them. It will only exercise rights if you
pay it the exercise price and any other charges the rights require you to pay.
U.S.
securities laws may restrict transfers and cancellation of the ADSs represented by shares purchased upon exercise of rights. For
example, you may not be able to trade these ADSs freely in the United States. In this case, the Depositary may deliver restricted
depositary shares that have the same terms as the ADSs described in this section except for changes needed to put the necessary
restrictions in place.
Other
Distributions.
The Depositary will send to ADS holders anything else we distribute on deposited securities by any means it
thinks is legal, fair and practical. If it cannot make the distribution in that way, the Depositary has a choice. It may decide
to sell what we distributed and distribute the net proceeds, in the same way as it does with cash. Or, it may decide to hold what
we distributed, in which case ADSs will also represent the newly distributed property. However, the Depositary is not required
to distribute any securities (other than ADSs) to ADS holders unless it receives satisfactory evidence from us that it is legal
to make that distribution. The Depositary may sell a portion of the distributed securities or property sufficient to pay its fees
and expenses in connection with that distribution.
The
Depositary is not responsible if it decides that it is unlawful or impractical to make a distribution available to any ADS holders.
We have no obligation to register ADSs, shares, rights or other securities under the Securities Act. We also have no obligation
to take any other action to permit the distribution of ADSs, shares, rights or anything else to ADS holders.
This means
that you may not receive the distributions we make on our shares or any value for them if it is illegal or impractical for us
to make them available to you
.
Deposit,
Withdrawal and Cancellation
How
are ADSs issued?
The
Depositary will deliver ADSs if you or your broker deposits shares or evidence of rights to receive shares with the custodian.
Upon payment of its fees and expenses and of any taxes or charges, such as stamp taxes or stock transfer taxes or fees, the Depositary
will register the appropriate number of ADSs in the names you request and will deliver the ADSs to or upon the order of the person
or persons that made the deposit.
How
can ADS holders withdraw the deposited securities?
You
may surrender your ADSs at the Depositary’s office. Upon payment of its fees and expenses and of any taxes or charges, such
as stamp taxes or stock transfer taxes or fees, the Depositary will deliver the shares and any other deposited securities
underlying the ADSs to the ADS holder or a person the ADS holder designates at the office of the custodian. Or, at your request,
risk and expense, the Depositary will deliver the deposited securities at its office, if feasible.
How
do ADS holders interchange between certificated ADSs and uncertificated ADSs?
You
may surrender your ADR to the Depositary for the purpose of exchanging your ADR for uncertificated ADSs. The Depositary will cancel
that ADR and will send to the ADS holder a statement confirming that the ADS holder is the registered holder of uncertificated
ADSs. Alternatively, upon receipt by the Depositary of a proper instruction from a registered holder of uncertificated ADSs requesting
the exchange of uncertificated ADSs for certificated ADSs, the Depositary will execute and deliver to the ADS holder an ADR evidencing
those ADSs.
Voting
Rights
How
do you vote?
ADS
holders may instruct the Depositary how to vote the number of deposited shares their ADSs represent. The Depositary will notify
ADS holders of shareholders’ meetings and arrange to deliver our voting materials to them if we ask it to. Those materials
will describe the matters to be voted on and explain how ADS holders may instruct the Depositary how to vote. For instructions
to be valid, they much reach the Depositary by a date set by the Depositary.
Otherwise, you won’t be able to exercise
your right to vote unless you withdraw the shares. However, you may not know about the meeting enough in advance to withdraw the
shares.
The
Depositary will try, as far as practical, subject to the laws of Israel, and of our articles of association or similar documents,
to vote or to have its agents vote the shares or other deposited securities as instructed by ADS holders. The Depositary will
only vote or attempt to vote as instructed.
We
cannot assure you that you will receive the voting materials in time to ensure that you can instruct the Depositary to vote your
shares. In addition, the Depositary and its agents are not responsible for failing to carry out voting instructions or for the
manner of carrying out voting instructions.
This means that you may not be able to exercise your right to vote and there may
be nothing you can do if your shares are not voted as you requested.
In
order to give you a reasonable opportunity to instruct the Depositary as to the exercise of voting rights relating to deposited
securities, if we request the Depositary to act, we agree to give the Depositary notice of any such meeting and details concerning
the matters to be voted upon at least 30 days in advance of the meeting date.
Fees
and Expenses
|
Persons
depositing or withdrawing shares or ADS holders must pay
:
|
|
|
For
:
|
|
$5.00
(or less) per 100 ADSs (or portion of 100 ADSs)
|
|
●
|
Issuance
of ADSs, including issuances resulting from a distribution of shares or rights or other property
|
|
|
|
●
|
Cancellation
of ADSs for the purpose of withdrawal, including if the deposit agreement terminates
|
|
$.05
(or less) per ADS
|
|
●
|
Any
cash distribution to ADS holders
|
|
A
fee equivalent to the fee that would be payable if securities distributed to you had been shares and the shares had been deposited
for issuance of ADSs
|
|
●
|
Distribution
of securities distributed to holders of deposited securities which are distributed by the Depositary to ADS holders
|
|
$.05
(or less) per ADS per calendar year
|
|
●
|
Depositary
services
|
|
Registration
or transfer fees
|
|
●
|
Transfer
and registration of shares on our share register to or from the name of the Depositary or its agent when you deposit or withdraw
shares
|
|
Expenses
of the Depositary
|
|
●
|
Cable,
telex and facsimile transmissions (when expressly provided in the deposit agreement)
|
|
|
|
●
|
converting
foreign currency to U.S. dollars
|
|
Taxes
and other governmental charges the Depositary or the custodian has to pay on any ADSs or shares underlying ADSs, such as stock
transfer taxes, stamp duty or withholding taxes
|
|
●
|
As
necessary
|
|
Any
charges incurred by the depositary or its agents for servicing the deposited securities
|
|
●
|
As
necessary
|
The
Depositary collects its fees for delivery and surrender of ADSs directly from investors depositing shares or surrendering ADSs
for the purpose of withdrawal or from intermediaries acting for them. The Depositary collects fees for making distributions to
investors by deducting those fees from the amounts distributed or by selling a portion of distributable property to pay the fees.
The Depositary may collect its annual fee for depositary services by deduction from cash distributions or by directly billing
investors or by charging the book-entry system accounts of participants acting for them. The Depositary may collect any of its
fees by deduction from any cash distribution payable to ADS holders that are obligated to pay those fees. The Depositary may generally
refuse to provide fee-attracting services until its fees for those services are paid.
From
time to time, the Depositary may make payments to us to reimburse and/or share revenue from the fees collected from ADS holders,
or waive fees and expenses for services provided, generally relating to costs and expenses arising out of establishment and maintenance
of the ADS program. In performing its duties under the deposit agreement, the Depositary may use brokers, dealers or other service
providers that are affiliates of the Depositary and that may earn or share fees or commissions.
Payment
of Taxes
You
will be responsible for any taxes or other governmental charges payable on your ADSs or on the deposited securities represented
by any of your ADSs. The Depositary may refuse to register any transfer of your ADSs or allow you to withdraw the deposited securities
represented by your ADSs until such taxes or other charges are paid. It may apply payments owed to you or sell deposited securities
represented by your ADSs to pay any taxes owed and you will remain liable for any deficiency. If the Depositary sells deposited
securities, it will, if appropriate, reduce the number of ADSs to reflect the sale and pay to ADS holders any proceeds, or send
to ADS holders any property, remaining after it has paid the taxes.
Reclassifications,
Recapitalizations and Mergers
|
If
we:
|
|
Then:
|
|
●
Change the nominal or par value of our shares
●
Reclassify, split up or consolidate any of the deposited securities
●
Distribute securities on the shares that are not distributed to you
●
Recapitalize, reorganize, merge, liquidate, sell all or
substantially all of our assets, or take any similar action
|
|
The
cash, shares or other securities received by the Depositary will become deposited securities. Each ADS will automatically
represent its equal share of the new deposited securities.
The
Depositary may distribute new ADSs representing the new deposited securities or ask you to surrender your outstanding
ADRs in exchange for new ADRs identifying the new deposited securities.
|
|
|
|
|
Amendment
and Termination
How
may the deposit agreement be amended?
We
may agree with the Depositary to amend the deposit agreement and the ADRs without your consent for any reason. If an amendment
adds or increases fees or charges, except for taxes and other governmental charges or expenses of the Depositary for registration
fees, facsimile costs, delivery charges or similar items, or prejudices a substantial right of ADS holders, it will not become
effective for outstanding ADSs until 30 days after the Depositary notifies ADS holders of the amendment.
At the time an amendment
becomes effective, you are considered, by continuing to hold your ADSs, to agree to the amendment and to be bound by the ADRs
and the deposit agreement as amended
.
How
may the deposit agreement be terminated?
The
Depositary will terminate the deposit agreement at our direction by mailing notice of termination to the ADS holders then outstanding
at least 30 days prior to the date fixed in such notice for such termination. The Depositary may also terminate the deposit agreement
by mailing notice of termination to us and the ADS holders if 60 days have passed since the Depositary told us it wants to resign
but a successor depositary has not been appointed and accepted its appointment.
After
termination, the Depositary and its agents will do the following under the deposit agreement but nothing else: collect distributions
on the deposited securities, sell rights and other property, and deliver shares and other deposited securities upon cancellation
of ADSs. Four months after termination, the Depositary may sell any remaining deposited securities by public or private sale.
After that, the Depositary will hold the money it received on the sale, as well as any other cash it is holding under the deposit
agreement for the pro rata benefit of the ADS holders that have not surrendered their ADSs. It will not invest the money and has
no liability for interest. The Depositary’s only obligations will be to account for the money and other cash. After termination
our only obligations will be to indemnify the Depositary and to pay fees and expenses of the Depositary that we agreed to pay.
Limitations
on Obligations and Liability
Limits
on our Obligations and the Obligations of the Depositary; Limits on Liability to Holders of ADSs
The
deposit agreement expressly limits our obligations and the obligations of the Depositary. It also limits our liability and the
liability of the Depositary. We and the Depositary:
|
|
●
|
are
only obligated to take the actions specifically set forth in the deposit agreement without negligence or bad faith;
|
|
|
|
|
|
|
●
|
are
not liable if we are or it is prevented or delayed by law or circumstances beyond our or its control from performing our or
its obligations under the deposit agreement;
|
|
|
|
|
|
|
●
|
are
not liable if we or it exercises discretion permitted under the deposit agreement;
|
|
|
|
|
|
|
●
|
are
not liable for the inability of any holder of ADSs to benefit from any distribution on deposited securities that is not made
available to holders of ADSs under the terms of the deposit agreement, or for any special, consequential or punitive damages
for any breach of the terms of the deposit agreement;
|
|
|
|
|
|
|
●
|
have
no obligation to become involved in a lawsuit or other proceeding related to the ADSs or the deposit agreement on your behalf
or on behalf of any other person;
|
|
|
|
|
|
|
●
|
are
not liable for the acts or omissions of any securities depository, clearing agency or settlement system; and
|
|
|
|
|
|
|
●
|
may
rely upon any documents we believe or it believes in good faith to be genuine and to have been signed or presented by the
proper person.
|
In
the deposit agreement, we and the Depositary agree to indemnify each other under certain circumstances.
Requirements
for Depositary Actions
Before
the Depositary will deliver or register a transfer of ADSs, make a distribution on ADSs, or permit withdrawal of shares, the Depositary
may require:
|
|
●
|
payment
of stock transfer or other taxes or other governmental charges and transfer or registration fees charged by third parties
for the transfer of any shares or other deposited securities;
|
|
|
|
|
|
|
●
|
satisfactory
proof of the identity and genuineness of any signature or other information it deems necessary; and
|
|
|
|
|
|
|
●
|
compliance
with regulations it may establish, from time to time, consistent with the deposit agreement, including presentation of transfer
documents.
|
The
Depositary may refuse to deliver ADSs or register transfers of ADSs when the transfer books of the Depositary or our transfer
books are closed or at any time if the Depositary or we think it advisable to do so.
Right
to Receive the Shares Underlying your ADSs
ADS
holders have the right to cancel their ADSs and withdraw the underlying shares at any time except:
|
|
●
|
when
temporary delays arise because: (i) the Depositary has closed its transfer books or we have closed our transfer books; (ii)
the transfer of shares is blocked to permit voting at a shareholders' meeting; or (iii) we are paying a dividend on our shares;
|
|
|
|
|
|
|
●
|
when
you owe money to pay fees, taxes and similar charges; or
|
|
|
|
|
|
|
●
|
when
it is necessary to prohibit withdrawals in order to comply with any laws or governmental regulations that apply to ADSs or
to the withdrawal of shares or other deposited securities.
|
This
right of withdrawal may not be limited by any other provision of the deposit agreement.
Pre-release
of ADSs
The
deposit agreement permits the Depositary to deliver ADSs before deposit of the underlying shares. This is called a pre-release
of the ADSs. The Depositary may also deliver shares upon cancellation of pre-released ADSs (even if the ADSs are canceled before
the pre-release transaction has been closed out). A pre-release is closed out as soon as the underlying shares are delivered to
the Depositary. The Depositary may receive ADSs instead of shares to close out a pre-release. The Depositary may pre-release ADSs
only under the following conditions: (1) before or at the time of the pre-release, the person to whom the pre-release is being
made represents to the Depositary in writing that it or its customer owns the shares or ADSs to be deposited; (2) the pre-release
is fully collateralized with cash or other collateral that the Depositary considers appropriate; and (3) the Depositary must be
able to close out the pre-release on not more than five business days' notice. In addition, the Depositary will limit the number
of ADSs that may be outstanding at any time as a result of pre-release, although the Depositary may disregard the limit from time
to time if it thinks it is appropriate to do so.
Direct
Registration System
In
the deposit agreement, all parties to the deposit agreement acknowledge that the Direct Registration System, or DRS, and Profile
Modification System, or Profile, will apply to uncertificated ADSs upon acceptance thereof to DRS by the Depository Trust Company,
or DTC. DRS is the system administered by DTC that facilitates interchange between registered holding of uncertificated ADSs and
holding of security entitlements in ADSs through DTC and a DTC participant. Profile is a required feature of DRS that allows a
DTC participant, claiming to act on behalf of a registered holder of ADSs, to direct the Depositary to register a transfer of
those ADSs to DTC or its nominee and to deliver those ADSs to the DTC account of that DTC participant without receipt by the Depositary
of prior authorization from the ADS holder to register that transfer.
In
connection with and in accordance with the arrangements and procedures relating to DRS/Profile, the parties to the deposit agreement
understand that the Depositary will not determine whether the DTC participant that is claiming to be acting on behalf of an ADS
holder in requesting registration of transfer and delivery described in the paragraph above has the actual authority to act on
behalf of the ADS holder (notwithstanding any requirements under the Uniform Commercial Code). In the deposit agreement, the parties
agree that the Depositary’s reliance on and compliance with instructions received by the Depositary through the DRS/Profile
System and in accordance with the deposit agreement will not constitute negligence or bad faith on the part of the Depositary.
Shareholder
communications; inspection of register of holders of ADSs
The
Depositary will make available for your inspection at its office all communications that it receives from us as a holder of deposited
securities that we make generally available to holders of deposited securities. The Depositary will send you copies of those communications
if we ask it to. You have a right to inspect the register of holders of ADSs, but not for the purpose of contacting those holders
about a matter unrelated to our business or the ADSs.
WARRANTS
TO BE ISSUED AS PART OF THIS OFFERING
Series A Warrants
The following summary of certain terms
and provisions of the Series A warrants offered hereby is not complete and is subject to, and qualified in its entirety by the
provisions of the Series A Warrant which is filed as an exhibit to the registration statement of which this prospectus is a part.
Prospective investors should carefully review the terms and provisions set forth in the form of Series A Warrant agreement.
Exercisability
The Series A warrants are exercisable immediately
upon issuance and at any time up to the date that is years from the date of issuance.
The Series A warrants will be exercisable, at the option of each holder, in whole or in part by delivering to us a duly executed
exercise notice accompanied by payment in full for the number of ADSs purchased upon such exercise (except in the case of a cashless
exercise as discussed below), and other applicable charges and taxes. Unless otherwise specified in the Series A warrant, the
holder will not have the right to exercise the Series A warrants, in whole or in part, if the holder (together with its affiliates)
would beneficially own in excess of 4.99% of the number of our ordinary shares outstanding immediately after giving effect to
the exercise, as such percentage is determined in accordance with the terms of the Series A warrants. However, any holder
may increase or decrease such percentage to any other percentage not in excess of 9.99% upon at least 61 days’ prior notice
from the holder to us.
Cashless
Exercise
In the event that a registration statement
covering the ordinary shares to be represented by ADSs underlying the Series A warrants is not effective, or no current prospectus
is available for the issuance of the ordinary shares to be represented by ADSs underlying the Series A warrants, the holder may,
in its sole discretion, exercise the Series A warrants and, in lieu of making the cash payment otherwise contemplated to be made
to us upon such exercise in payment of the aggregate exercise price, elect instead to receive upon such exercise the net number
of ADSs determined according to the formula set forth in the Series A warrant agreement.
Exercise Price
The initial exercise price per ADS purchasable
upon exercise of the Series A warrants is equal to $ per ADS and is subject to adjustments for stock splits, reclassifications,
subdivisions, and other similar transactions.
Fundamental Transaction
If, at any time while the Series A warrants
are outstanding, (1) we consolidate or merge with or into another corporation and we are not the surviving corporation, (2) we
sell, lease, license, assign, transfer, convey or otherwise dispose of all or substantially all of our assets, (3) any purchase
offer, tender offer or exchange offer (whether by us or another individual or entity) is completed pursuant to which holders of
the ordinary shares are permitted to sell, tender or exchange their ordinary shares for other securities, cash or property and
has been accepted by the holders of 50% or more of the ordinary shares, (4) we effect any reclassification or recapitalization
of the ordinary shares or any compulsory exchange pursuant to which the ordinary shares are converted into or exchanged for other
securities, cash or property, or (5) we consummate a securities purchase agreement or other business combination with another
person or entity whereby such other person or entity acquires more than 50% of the outstanding ordinary shares, each, a “Fundamental
Transaction”, then upon any subsequent exercise of Series A warrants, the holders thereof will have the right to receive
the same amount and kind of securities, cash or property as it would have been entitled to receive upon the occurrence of such
Fundamental Transaction if it had been, immediately prior to such Fundamental Transaction, the holder of the number of ordinary
shares then issuable upon exercise of those Series A warrants, and any additional consideration payable as part of the Fundamental
Transaction.
In the event of an Fundamental Transaction
(other than a Fundamental Transaction not approved by the Company’s Board of Directors), we or any successor entity shall,
subject to any applicable law, at the option of a registered holder of a Series A warrant, exercisable at any time concurrently
with, or within 30 days after, the consummation of the Fundamental Transaction, purchase the Series A warrants of that holder
from that holder by paying to that holder an amount of cash equal to the Black Scholes value of the remaining unexercised Series
A warrants of that holder on the date of the consummation of such Fundamental Transaction.
Transferability
Subject to applicable laws, the Series A
warrants may be transferred at the option of the holders upon surrender of the Series A warrants to us, together with the appropriate
instruments of transfer.
Listing
The Series A warrants will be issued in physical
form. We do not plan on applying to list the Series A warrants on The NASDAQ Capital Market, any other national securities exchange
or any other nationally recognized trading system.
Rights as a Shareholder
Except as otherwise provided in the Series
A warrant or by virtue of such holder’s ownership of ADSs or ordinary shares, the holder of Series A warrants does not have
rights or privileges of a holder of ADSs or ordinary shares, including any voting rights, until the holder exercises the warrants.
Pre-Funded Series B Warrants
The following summary of certain terms and provisions
of the Series B warrants offered hereby is not complete and is subject to, and qualified in its entirety by the provisions of
the form of Pre-Funded Series B warrant, which is filed as an exhibit to the registration statement of which this prospectus is
a part. Prospective investors should carefully review the terms and provisions set forth in the Series B warrant agreement.
The purpose of the pre-funded Series B warrants
is to enable investors that may have restrictions on their ability to beneficially own more than 4.99% (or at the election of
the investor, 9,99%) of our outstanding ordinary shares following the consummation of this offering the opportunity to invest
capital into the Company without triggering such ownership restrictions. By receiving pre-funded Series B warrants in lieu of
the ADSs contained in the Class A units which would result in such holders’ ownership exceeding 4.99% (or at the election
of the investor, 9,99%), such holders will have the ability to exercise their options to purchase the ADSs underlying the pre-funded
Series B warrants for nominal consideration of $0.01 per ADS at a later date.
Exercisability
The Series B warrants are exercisable until fully
exercised. The Series B warrants will be exercisable, at the option of each holder, in whole or in part by delivering to us a
duly executed exercise notice accompanied by payment in full for the number of ADSs purchased upon such exercise (except in the
case of a cashless exercise as discussed below). Unless otherwise specified in the Series B warrant, a holder (together with its
affiliates) may not exercise any portion of the Series B warrants to the extent that the holder would own more than 4.99% of the
outstanding ordinary shares after exercise, except that upon at least 61 days’ prior notice from the holder to us, the holder
may increase or decrease the amount of ownership of outstanding shares after exercising the holder’s warrants, as applicable,
up to 9.99% of the number of our ordinary shares outstanding immediately after giving effect to the exercise, as such percentage
ownership is determined in accordance with the terms of the warrants.
Cashless
Exercise
In the event that a registration statement covering
the ordinary shares to be represented by ADSs underlying the Series B warrants is not effective, or no current prospectus is available
for the issuance of the ordinary shares to be represented by ADSs underlying the Series B warrants, the holder may, in its sole
discretion, exercise Series B warrants and, in lieu of making the cash payment otherwise contemplated to be made to us upon such
exercise in payment of the aggregate exercise price, elect instead to receive upon such exercise the net number of ADSs determined
according to the formula set forth in the Series B warrant.
Exercise
Price
The
initial exercise price per ADS purchasable upon exercise of the Series B warrants is equal to $0.01.
Listing
The Series B warrants will be issued in physical
form. We do not plan on applying to list the pre-funded Series B warrants on The NASDAQ Capital Market, any other national securities
exchange or any other nationally recognized trading system.
Fundamental Transaction
In the event of a fundamental
transaction, as described in the Series B warrants and generally including any reorganization, recapitalization or reclassification
of our share capital, the sale, transfer or other disposition of all or substantially all of our properties or assets, our consolidation
or merger with or into another person, the acquisition of more than 50% of our outstanding ordinary shares, or any person or group
becoming the beneficial owner of 50% of the voting power represented by our outstanding ordinary shares, the holders of the Series
B Warrants will be entitled to receive upon exercise of the Series B warrants the kind and amount of securities, cash or other
property that the holders would have received had they exercised the Series B warrants immediately prior to such fundamental transaction.
Material
Tax Considerations
The
following description is not intended to constitute a complete analysis of all tax consequences relating to the acquisition, ownership
and disposition of our ordinary shares. You should consult your own tax advisor concerning the tax consequences of your particular
situation, as well as any tax consequences that may arise under the laws of any state, local, foreign or other taxing jurisdiction.
Israeli
Tax Considerations and Government Programs
The
following is a summary of the material Israeli tax laws applicable to us, and some Israeli Government programs benefiting us.
This section also contains a discussion of some Israeli tax consequences to persons owning our ordinary shares. This summary does
not discuss all the aspects of Israeli tax law that may be relevant to a particular investor in light of his or her personal investment
circumstances or to some types of investors subject to special treatment under Israeli law. Examples of this kind of investor
include traders in securities or persons that own, directly or indirectly, 10% or more of our outstanding voting capital, all
of whom are subject to special tax regimes not covered in this discussion. Some parts of this discussion are based on a new tax
legislation which has not been subject to judicial or administrative interpretation. The discussion should not be construed as
legal or professional tax advice and does not cover all possible tax considerations.
SHAREHOLDERS
ARE URGED TO CONSULT THEIR OWN TAX ADVISORS AS TO THE ISRAELI OR OTHER TAX CONSEQUENCES OF THE PURCHASE, OWNERSHIP AND DISPOSITION
OF OUR ORDINARY SHARES, INCLUDING, IN PARTICULAR, THE EFFECT OF ANY FOREIGN, STATE OR LOCAL TAXES.
General
Corporate Tax Structure in Israel
Israeli
resident companies are generally subject to corporate tax on their taxable income at the rate of 25% of a company’s taxable
income for 2016 tax year (to be reduced to 24% in 2017 and 23% in 2018. However, the effective tax rate payable by a company that
derives income from a Benefited Enterprise or a Preferred Enterprise (as discussed below) may be considerably less. Capital gains
derived by an Israeli resident company are subject to tax at the prevailing corporate tax rate.
Under
Israeli tax legislation, a corporation will be considered as an “Israeli resident company” if it meets one of the
following: (i) it was incorporated in Israel; or (ii) the control and management of its business are exercised in Israel.
Law
for the Encouragement of Industry (Taxes), 5729-1969
The
Law for the Encouragement of Industry (Taxes), 5729-1969, generally referred to as the Industry Encouragement Law, provides several
tax benefits for “Industrial Companies.”
The
Industry Encouragement Law defines an “Industrial Company” as a company resident in Israel, of which 90% or more of
its income in any tax year, other than income from defense loans, is derived from an “Industrial Enterprise” owned
by it. An “Industrial Enterprise” is defined as an enterprise whose principal activity in a given tax year is industrial
production.
The
following corporate tax benefits, among others, are available to Industrial Companies:
|
|
●
|
amortization
over an eight-year period of the cost of purchased know-how and patents and rights to
use a patent and know-how which are used for the development or advancement of the Industrial
Enterprise;
|
|
|
●
|
under
limited conditions, an election to file consolidated tax returns with related Israeli
Industrial Companies; and
|
|
|
●
|
expenses
related to a public offering are deductible in equal amounts over three years.
|
Although
at the date of this prospectus, we still have no industrial production activities, we may qualify as an Industrial Company in
the future and may be eligible for the benefits described above.
Tax
Benefits and Grants for Research and Development
Israeli
tax law allows, under certain conditions, a tax deduction for expenditures, including capital expenditures, for the year in which
they are incurred. Expenditures are deemed related to scientific research and development projects, if:
|
|
●
|
The
expenditures are approved by the relevant Israeli government ministry, determined by
the field of research;
|
|
|
●
|
The
research and development must be for the promotion of the company; and
|
|
|
●
|
The
research and development is carried out by or on behalf of the company seeking such tax
deduction.
|
The
amount of such deductible expenses is reduced by the sum of any funds received through government grants for the finance of such
scientific research and development projects. No deduction under these research and development deduction rules is allowed if
such deduction is related to an expense invested in an asset depreciable under the general depreciation rules of the income Tax
Ordinance, 1961. Expenditures not so approved are deductible in equal amounts over three years.
From
time to time we may apply the Office of the Chief Scientist for approval to allow a tax deduction for all research and development
expenses during the year incurred. There can be no assurance that such application will be accepted.
Law
for the Encouragement of Capital Investments, 5719-1959
The
Law for the Encouragement of Capital Investments, 5719-1959, generally referred to as the Investment Law, provides certain incentives
for capital investments in production facilities (or other eligible assets) by “Industrial Enterprises” (as defined
under the Investment Law).
Tax
Benefits Prior to the 2005 Amendment
An
investment program that is implemented in accordance with the provisions of the Investment Law prior to the 2005 Amendment, referred
to as an “Approved Enterprise,” is entitled to certain benefits. A company that wished to receive benefits as an Approved
Enterprise must have received approval from the Investment Center of the Israeli Ministry of Economy, or the Investment Center.
Each certificate of approval for an Approved Enterprise relates to a specific investment program in the Approved Enterprise, delineated
both by the financial scope of the investment and by the physical characteristics of the facility or the asset.
In
general, an Approved Enterprise is entitled to receive a grant from the Government of Israel or an alternative package of tax
benefits, known as the alternative benefits track. The tax benefits from any certificate of approval relate only to taxable profits
attributable to the specific Approved Enterprise. Income derived from activity that is not integral to the activity of the Approved
Enterprise does not enjoy tax benefits.
In
addition, a company that has an Approved Enterprise program is eligible for further tax benefits if it qualifies as a Foreign
Investors’ Company, or FIC, which is a company with a level of foreign investment, as defined in the Investment Law, of
more than 25%. The level of foreign investment is measured as the percentage of rights in the company (in terms of shares, rights
to profits, voting and appointment of directors), and of combined share and loan capital, that are owned, directly or indirectly,
by persons who are not residents of Israel. The determination as to whether a company qualifies as an FIC is made on an annual
basis.
We
are currently not entitled to tax benefits for Approved Enterprise.
Tax
Benefits Subsequent to the 2005 Amendment
The
2005 Amendment applies to new investment programs and investment programs commencing after 2004, but does not apply to investment
programs approved prior to April 1, 2005. The 2005 Amendment provides that terms and benefits included in any certificate
of approval that was granted before the 2005 Amendment became effective (April 1, 2005) will remain subject to the provisions
of the Investment Law as in effect on the date of such approval. Pursuant to the 2005 Amendment, the Investment Center will continue
to grant Approved Enterprise status to qualifying investments. The 2005 Amendment, however, limits the scope of enterprises that
may be approved by the Investment Center by setting criteria for the approval of a facility as an Approved Enterprise, such as
provisions generally requiring that at least 25% of the Approved Enterprise’s income be derived from exports.
The
2005 Amendment provides that Approved Enterprise status will only be necessary for receiving cash grants. As a result, it was
no longer necessary for a company to obtain Approved Enterprise status in order to receive the tax benefits previously available
under the alternative benefits track. Rather, a company may claim the tax benefits offered by the Investment Law directly in its
tax returns, provided that its facilities meet the criteria for tax benefits set forth in the amendment. Companies are entitled
to approach the Israeli Tax Authority for a pre-ruling regarding their eligibility for benefits under the Investment Law, as amended
In
order to receive the tax benefits, the 2005 Amendment states that a company must make an investment which meets all of the conditions,
including exceeding a minimum investment amount specified in the Investment Law. Such investment allows a company to receive “Benefited
Enterprise” status, and may be made over a period of no more than three years from the end of the year in which the company
requested to have the tax benefits apply to its Benefited Enterprise. Where the company requests to apply the tax benefits to
an expansion of existing facilities, only the expansion will be considered to be a Benefited Enterprise and the company’s
effective tax rate will be the weighted average of the applicable rates. In this case, the minimum investment required in order
to qualify as a Benefited Enterprise is required to exceed a certain percentage of the value of the company’s production
assets before the expansion.
The
extent of the tax benefits available under the 2005 Amendment to qualifying income of a Benefited Enterprise depend on, among
other things, the geographic location in Israel of the Benefited Enterprise. The location will also determine the period for which
tax benefits are available. Such tax benefits include an exemption from corporate tax on undistributed income for a period of
between two to ten years, depending on the geographic location of the Benefited Enterprise in Israel, and a reduced corporate
tax rate of between 10% to the applicable corporate tax for the remainder of the benefits period, depending on the level of foreign
investment in the company in each year. A company qualifying for tax benefits under the 2005 Amendment which pays a dividend out
of income derived by its Benefited Enterprise during the tax exemption period will be subject to corporate tax in respect of the
gross amount of the dividend at the otherwise applicable corporate tax rate, or a lower rate in the case of a qualified FIC which
is at least 49% owned by non-Israeli residents. Dividends paid out of income attributed to a Benefited Enterprise are generally
subject to withholding tax at source at the rate of 15% or such lower rate as may be provided in an applicable tax treaty.
The
benefits available to a Benefited Enterprise are subject to the fulfillment of conditions stipulated in the Investment Law and
its regulations. If a company does not meet these conditions, it may be required to refund the amount of tax benefits, as adjusted
by the Israeli consumer price index, and interest, or other monetary penalties.
We
applied for tax benefits as a “Benefited Enterprise” with 2012 as a “Year of Election.” We may be entitled
to tax benefits under this regime once we are profitable for tax purposes and subject to the fulfillment of all the relevant conditions.
If we do not meet these conditions, the tax benefits may not be applicable which would result in adverse tax consequences to us.
Alternatively, and subject to the fulfillment of all the relevant conditions, we may elect in the future to irrevocably waive
the tax benefits available for Benefited Enterprise and claim the tax benefits available to Preferred Enterprise under the 2011
Amendment (as detailed below).
Tax
Benefits under the 2011 Amendment
The
Investment Law was significantly amended as of January 1, 2011 (the “2011 Amendment”). The 2011 Amendment introduced
new benefits to replace those granted in accordance with the provisions of the Investment Law in effect prior to the 2011 Amendment.
The
2011 Amendment introduced new tax benefits for income generated by a “Preferred Company” through its “Preferred
Enterprise,” in accordance with the definition of such term in the Investments Law, which generally means that a “Preferred
Company” is an industrial company meeting certain conditions (including a minimum threshold of 25% export).
A
Preferred Company is entitled to a reduced flat tax rate with respect to the income attributed to the Preferred Enterprise, at
the following rates:
|
Tax Year
|
|
Development Region “A”
|
|
|
Other Areas within Israel
|
|
2011 – 2012
|
|
|
10
|
%
|
|
15%
|
|
2013
|
|
|
7
|
%
|
|
12.5%
|
|
2014
|
|
|
9
|
%
|
|
16%
|
|
2017 onwards
(1)
|
|
|
7.5
|
%
|
|
16%
|
|
(1)
|
In
December 2016, the Israeli Parliament (the Knesset) approved an amendment to the Investment
Law pursuant to which the tax rate applicable to Preferred Enterprises in Development
Region “A” would be reduced to 7.5% as of 2017.
|
Dividends
distributed from income which is attributed to a “Preferred Enterprise” will be subject to withholding tax at source
at the following rates: (i) Israeli resident corporations — 0%, (ii) Israeli resident individuals — 20% in 2016 (iii)
non-Israeli residents — 20% in 2016, subject to a reduced tax rate under the provisions of an applicable double tax treaty.
Under
the 2011 Amendment, a company located in Development Region “A” may be entitled to cash grants and the provision of
loans under certain conditions, if approved. The rates for grants and loans shall not be fixed, but up to 20% of the amount of
the approved investment (may be increased with additional 4%). In addition, a company owning a Preferred Enterprise under the
Grant Track may be entitled also to the tax benefits which are prescribed for a Preferred Company.
The
termination or substantial reduction of any of the benefits available under the Investment Law could materially increase our tax
liabilities.
We
are currently not entitled to tax benefits for Preferred Enterprise.
Taxation
of Our Shareholders
Capital
Gains
Capital
gain tax is imposed on the disposition of capital assets by an Israeli resident, and on the disposition of such assets by a non-Israeli
resident if those assets are either (i) located in Israel; (ii) are shares or a right to a share in an Israeli resident corporation,
or (iii) represent, directly or indirectly, rights to assets located in Israel. The Israeli Income Tax Ordinance of 1961 (New
Version) (the “Ordinance”) distinguishes between “Real Gain” and the “Inflationary Surplus.”
Real Gain is the excess of the total capital gain over Inflationary Surplus computed generally on the basis of the increase in
the Israeli CPI between the date of purchase and the date of disposition. Inflationary Surplus is not subject to tax in Israel.
Real
Gain accrued by individuals on the sale of our ordinary shares will be taxed at the rate of 25%. However, if the individual shareholder
is a “Controlling Shareholder” (i.e., a person who holds, directly or indirectly, alone or together with another,
10% or more of one of the Israeli resident company’s means of control) at the time of sale or at any time during the preceding
12 months period, such gain will be taxed at the rate of 30%.
Real
Gain derived by corporations will be generally subject to a corporate tax rate of 25% in 2016 (to be reduced to 24% in 2017 and
23% in 2018 and thereafter).
Individual
and corporate shareholder dealing in securities in Israel are taxed at the tax rates applicable to business income — 25%
for corporations in 2016 (to be reduced to 24% in 2017 and 23% in 2018 and thereafter) and a marginal tax rate of up to 50% in
2016 for individuals, including an excess tax which is levied on individuals whose taxable income in Israel exceeds certain
threshold (NIS 810,720 in 2016 and NIS 640,000 in 2017 and thereafter, as discussed below).
Notwithstanding
the foregoing, capital gain derived from the sale of our ordinary shares by a non-Israeli shareholder may be exempt under the
Ordinance from Israeli taxation provided that the following cumulative conditions are met: (i) the shares were purchased upon
or after the registration of the securities on the stock exchange (this condition will not apply to shares purchased on or after
January 1, 2009), (ii) the seller does not have a permanent establishment in Israel to which the derived capital gain is attributed,
(iii) if the seller is a corporation, no more than 25% of its means of control are held, directly and indirectly, by an Israeli
resident shareholders, and (iv) if the seller is a corporation, there is no Israeli Resident that is entitled to 25% or more of
the revenues or profits of the corporation directly or indirectly. In addition, such exemption would not be available to a person
whose gains from selling or otherwise disposing of the securities are deemed to be business income.
In
addition, the sale of shares may be exempt from Israeli capital gain tax under the provisions of an applicable tax treaty. For
example, the U.S.-Israel Double Tax Treaty exempts U.S. resident from Israeli capital gain tax in connection with such sale, provided
(i) the U.S. resident owned, directly or indirectly, less than 10% of an Israeli resident company’s voting power at any
time within the 12 month period preceding such sale; (ii) the seller, being an individual, is present in Israel for a period or
periods of less than 183 days at the taxable year; and (iii) the capital gain from the sale was not derived through a permanent
establishment of the U.S. resident in Israel.
Either
the purchaser, the Israeli stockbrokers or financial institution through which the shares are held is obliged, subject to the
above mentioned exemptions, to withhold tax upon the sale of securities from the Real Gain at the rate of 25%.
At
the sale of securities traded on a stock exchange a detailed return, including a computation of the tax due, must be filed and
an advanced payment must be paid on January 31 and June 30 of every tax year in respect of sales of securities made
within the previous six months. However, if all tax due was withheld at source according to applicable provisions of the Ordinance
and regulations promulgated thereunder the aforementioned return need not be filed and no advance payment must be paid. Capital
gain is also reportable on the annual income tax return.
Dividends
We
have never paid cash dividends. A distribution of dividend by our company from income attributed to a Benefited Enterprise will
generally be subject to withholding tax in Israel at a rate of 15% unless a reduced tax rate is provided under an applicable tax
treaty. A distribution of dividend by our company from income attributed to a Preferred Enterprise will generally be subject to
withholding tax in Israel at the following tax rates: Israeli resident individuals — 20% with respect to dividends to be
distributed as of 2016; Israeli resident companies — 0% for a Preferred Enterprise; Non-Israeli residents — 20% with
respect to dividends to be distributed as of 2016, subject to a reduced rate under the provisions of any applicable double tax
treaty. A distribution of dividends from income, which is not attributed to a Preferred Enterprise to an Israeli resident individual,
will generally be subject to income tax at a rate of 25%. However, a 30% tax rate will apply if the dividend recipient is a “Controlling
Shareholder” (as defined above) at the time of distribution or at any time during the preceding 12 months period. If the
recipient of the dividend is an Israeli resident corporation, such dividend will be exempt from income tax provided the income
from which such dividend is distributed was derived or accrued within Israel.
The
Ordinance provides that a non-Israeli resident (either individual or corporation) is generally subject to an Israeli income tax
on the receipt of dividends at the rate of 25% (30% if the dividends recipient is a “Controlling Shareholder” (as
defined above), at the time of distribution or at any time during the preceding 12 months period); those rates are subject to
a reduced tax rate under the provisions of an applicable double tax treaty. Thus, under the U.S.-Israel Double Tax Treaty the
following rates will apply in respect of dividends distributed by an Israeli resident company to a U.S. resident: (i) if the U.S.
resident is a corporation which holds during that portion of the taxable year which precedes the date of payment of the dividend
and during the whole of its prior taxable year (if any), at least 10% of the outstanding shares of the voting share capital of
the Israeli resident paying corporation and not more than 25% of the gross income of the Israeli resident paying corporation for
such prior taxable year (if any) consists of certain type of interest or dividends — the tax rate is 12.5%, (ii) if both
the conditions mentioned in section (i) above are met and the dividend is paid from an Israeli resident company’s income
which was entitled to a reduced tax rate applicable to an Approved Enterprise — the tax rate is 15% and (iii) in all other
cases, the tax rate is 25%. The aforementioned rates under the Israel U.S. Double Tax Treaty will not apply if the dividend income
was derived through a permanent establishment of the U.S. resident in Israel.
A
non-Israeli resident who receives dividends from which tax was withheld is generally exempt from the obligation to file tax returns
in Israel with respect to such income, provided that (i) such income was not generated from business conducted in Israel by the
taxpayer, and (ii) the taxpayer has no other taxable sources of income in Israel with respect to which a tax return is required
to be filed.
Payers
of dividends on our ordinary shares, including the Israeli stockbroker effectuating the transaction, or the financial institution
through which the securities are held, are generally required, subject to any of the foregoing exemptions, reduced tax rates and
the demonstration of a shareholder regarding his, her or its foreign residency, to withhold tax upon the distribution of dividend
at the rate of 25%, so long as the shares are registered with a nominee company.
Excess
Tax
Individuals
who are subject to tax in Israel are also subject to an additional tax at a rate of 2% in 2016 (to be increased to 3% in 2017
and thereafter) on annual income exceeding a certain threshold (NIS 810,720 for 2016 and NIS 640,000 for 2017 and thereafter,
linked to the annual change in the Israeli Consumer Price Index), including, but not limited to income derived from, dividends,
interest and capital gains.
Foreign
Exchange Regulations
Non-residents
of Israel who hold our ordinary shares are able to receive any dividends, and any amounts payable upon the dissolution, liquidation
and winding up of our affairs, repayable in non-Israeli currency at the rate of exchange prevailing at the time of conversion.
However, Israeli income tax is generally required to have been paid or withheld on these amounts. In addition, the statutory framework
for the potential imposition of currency exchange control has not been eliminated, and may be restored at any time by administrative
action.
Estate
and Gift Tax
Israeli
law presently does not impose estate or gift taxes.
U.S.
Federal Income Tax Consequences
The
following discussion describes certain material U.S. federal income tax consequences to U.S. Holders (as defined below) under
present law of an investment in our ordinary shares. This discussion applies only to U.S. Holders that hold our ordinary shares
as capital assets within the meaning of Section 1221 of the Internal Revenue Code of 1986, as amended (the “Code”),
that have acquired their ordinary shares in this offering and that have the U.S. dollar as their functional currency.
This
discussion is based on the tax laws of the United States, including the Code, as in effect on the date hereof and on U.S. Treasury
regulations as in effect or, in some cases, as proposed, on the date hereof, as well as judicial and administrative interpretations
thereof available on or before such date. All of the foregoing authorities are subject to change, which change could apply retroactively
and could affect the tax consequences described below. This summary does not address any estate or gift tax consequences, the
alternative minimum tax, the Medicare tax on net investment income or any state, local, or non-U.S. tax consequences.
The
following discussion neither deals with the tax consequences to any particular investor nor describes all of the tax consequences
applicable to persons in special tax situations such as:
|
|
●
|
certain
financial institutions;
|
|
|
●
|
regulated
investment companies;
|
|
|
●
|
real
estate investment trusts;
|
|
|
●
|
traders
that elect to mark to market;
|
|
|
●
|
persons
holding our ordinary shares as part of a straddle, hedging, constructive sale, conversion
or integrated transaction;
|
|
|
●
|
persons
that actually or constructively own 10% or more of the total combined voting power of
all classes of our voting share capital;
|
|
|
●
|
persons
that are resident or ordinarily resident in or have a permanent establishment in a jurisdiction
outside the United States;
|
|
|
●
|
persons
who acquired our ordinary shares pursuant to the exercise of any employee share option
or otherwise as compensation; or
|
|
|
●
|
pass-through
entities, or persons holding our ordinary shares through pass-through entities.
|
INVESTORS
ARE URGED TO CONSULT THEIR TAX ADVISORS ABOUT THE APPLICATION OF THE U.S. FEDERAL TAX RULES TO THEIR PARTICULAR CIRCUMSTANCES
AS WELL AS THE STATE, LOCAL, NON-U.S. AND OTHER TAX CONSEQUENCES TO THEM OF THE PURCHASE, OWNERSHIP AND DISPOSITION OF OUR ORDINARY
SHARES.
The
discussion below of the U.S. federal income tax consequences to “U.S. Holders” will apply to you if you are the beneficial
owner of our ordinary shares and you are, for U.S. federal income tax purposes,
|
|
●
|
an
individual who is a citizen or resident of the United States;
|
|
|
●
|
a
corporation (or other entity taxable as a corporation for U.S. federal income tax purposes)
created or organized in the United States or under the laws of the United States, any
state thereof or the District of Columbia;
|
|
|
●
|
an
estate, the income of which is subject to U.S. federal income taxation regardless of
its source; or
|
|
|
●
|
a
trust that (1) is subject to the primary supervision of a court within the United States
and the control of one or more U.S. persons for all substantial decisions or (2) has
a valid election in effect under applicable U.S. Treasury regulations to be treated as
a U.S. person.
|
If
an entity or other arrangement treated as a partnership for U.S. federal income tax purposes holds our ordinary shares, the tax
treatment of a partner will generally depend upon the status of the partner and the activities of the partnership. A person that
would be a U.S. Holder if it held our ordinary shares directly and that is a partner of a partnership holding our ordinary shares
is urged to consult its own tax advisor.
Passive
Foreign Investment Company
Based
on our anticipated income and the composition of our income and assets, there is a significant risk that we will be a passive
foreign investment company (“PFIC”) for U.S. federal income tax purposes at least until we start generating a substantial
amount of active revenue. However, because PFIC status is a factual determination based on actual results for the entire taxable
year, our U.S. counsel expresses no opinion with respect to our PFIC status. A non-U.S. entity treated as a corporation for U.S.
federal income tax purposes will generally be a PFIC for U.S. federal income tax purposes for any taxable year if either:
|
|
●
|
at
least 75% of its gross income for such year is passive income (such as interest income);
or
|
|
|
●
|
at
least 50% of the value of its assets (based on an average of the quarterly values of
the assets) during such year is attributable to assets that produce passive income or
are held for the production of passive income.
|
For
this purpose, we will be treated as owning our proportionate share of the assets and earning our proportionate share of the income
of any other entity treated as a corporation for U.S. federal income tax purposes in which we own, directly or indirectly, 25%
or more (by value) of the stock.
A
separate determination must be made after the close of each taxable year as to whether we were a PFIC for that year. Because the
value of our assets for purposes of the PFIC test will generally be determined by reference to the market price of our ordinary
shares, our PFIC status may depend in part on the market price of our ordinary shares, which may fluctuate significantly. In addition,
there may be certain ambiguities in applying the PFIC test to us. No rulings from the U.S. Internal Revenue Service (the “IRS”),
however, have been or will be sought with respect to our status as a PFIC.
If
we are a PFIC for any taxable year during which you hold our ordinary shares, we generally will continue to be treated as a PFIC
with respect to your investment in our ordinary shares for all succeeding years during which you hold our ordinary shares, unless
we cease to be a PFIC and you make a “deemed sale” election with respect to our ordinary shares. If such election
is made, you will be deemed to have sold our ordinary shares you hold at their fair market value on the last day of the last taxable
year in which we were a PFIC, and any gain from such deemed sale would be subject to the consequences described below. After the
deemed sale election, your ordinary shares with respect to which the deemed sale election was made will not be treated as shares
in a PFIC unless we subsequently become a PFIC.
For
each taxable year that we are treated as a PFIC with respect to you, you will be subject to special tax rules with respect to
any “excess distribution” (as defined below) you receive and any gain you realize from a sale or other disposition
(including a pledge) of our ordinary shares, unless you make a valid “mark-to-market” election as discussed below.
Distributions you receive in a taxable year that are greater than 125% of the average annual distributions you received during
the shorter of the three preceding taxable years or your holding period for our ordinary shares will be treated as an excess distribution.
Under these special tax rules:
|
|
●
|
the
excess distribution or gain will be allocated ratably over your holding period for our
ordinary shares;
|
|
|
●
|
the
amount allocated to the current taxable year, and any taxable years in your holding period
prior to the first taxable year in which we were a PFIC, will be treated as ordinary
income; and
|
|
|
●
|
the
amount allocated to each other taxable year will be subject to the highest tax rate in
effect for individuals or corporations, as applicable, for each such year and the interest
charge generally applicable to underpayments of tax will be imposed on the resulting
tax attributable to each such year.
|
The
tax liability for amounts allocated to taxable years prior to the year of disposition or excess distribution cannot be offset
by any net operating losses, and gains (but not losses) realized on the sale of our ordinary shares cannot be treated as capital
gains, even if you hold our ordinary shares as capital assets.
If
we are treated as a PFIC with respect to you for any taxable year, to the extent any of our subsidiaries are also PFICs, you may
be deemed to own shares in such lower-tier PFICs that are directly or indirectly owned by us in that proportion which the value
of our ordinary shares you own bears to the value of all of our ordinary shares, and you may be subject to the adverse tax consequences
described above with respect to the shares of such lower-tier PFICs you would be deemed to own. As a result, you may incur liability
for any excess distribution described above if we receive a distribution from our lower-tier PFICs or if any shares in such lower-tier
PFICs are disposed of (or deemed disposed of). You should consult your tax advisor regarding the application of the PFIC
rules to any of our subsidiaries.
A
U.S. Holder of “marketable stock” (as defined below) in a PFIC may make a mark-to-market election for such
stock to elect out of the tax treatment discussed above. If you make a valid mark-to-market election for our ordinary shares,
you will include in income for each year that we are treated as a PFIC with respect to you an amount equal to the excess, if any,
of the fair market value of our ordinary shares as of the close of your taxable year over your adjusted basis in such ordinary
shares. You will be allowed a deduction for the excess, if any, of the adjusted basis of our ordinary shares over their fair market
value as of the close of the taxable year. However, deductions will be allowable only to the extent of any net mark-to-market
gains on our ordinary shares included in your income for prior taxable years. Amounts included in your income under a mark-to-market
election, as well as gain on the actual sale or other disposition of our ordinary shares, will be treated as ordinary income.
Ordinary loss treatment will also apply to the deductible portion of any mark-to-market loss on our ordinary shares, as well as
to any loss realized on the actual sale or disposition of our ordinary shares, to the extent the amount of such loss does not
exceed the net mark-to-market gains for such ordinary shares previously included in income. Your basis in our ordinary shares
will be adjusted to reflect any such income or loss amounts. If you make a mark-to-market election, any distributions we make
would generally be subject to the rules discussed below under “— Taxation of dividends and other distributions on
our ordinary shares,” except the lower rates applicable to qualified dividend income would not apply.
The
mark-to-market election is available only for “marketable stock,” which is stock that is regularly traded on a qualified
exchange or other market, as defined in applicable U.S. Treasury regulations. We expect our ordinary shares will be listed on
NASDAQ. Because a mark-to-market election cannot be made for equity interests in any lower-tier PFICs we own, you generally will
continue to be subject to the PFIC rules with respect to your indirect interest in any investments held by us that are treated
as an equity interest in a PFIC for U.S. federal income tax purposes. NASDAQ is a qualified exchange, but there can be no assurance
that the trading in our ordinary shares will be sufficiently regular to qualify our ordinary shares as marketable stock. You should
consult your tax advisor as to the availability and desirability of a mark-to-market election, as well as the impact of such election
on interests in any lower-tier PFICs. Alternatively, if a non-U.S. entity treated as a corporation is a PFIC, a holder of shares
in that entity may avoid taxation under the PFIC rules described above regarding excess distributions and recognized gains by
making a “qualified electing fund” election to include in income its share of the entity’s income on a current
basis. However, you may make a qualified electing fund election with respect to your ordinary shares only if we furnish you annually
with certain tax information, and we currently do not intend to prepare or provide such information.
A
U.S. Holder of a PFIC may be required to file an IRS Form 8621. If we are a PFIC, you should consult your tax advisor regarding
any reporting requirements that may apply to you. You are urged to consult your tax advisor regarding the application of the PFIC
rules to the acquisition, ownership and disposition of our ordinary shares.
YOU
ARE STRONGLY URGED TO CONSULT YOUR TAX ADVISOR REGARDING THE IMPACT OF OUR BEING A PFIC ON YOUR INVESTMENT IN OUR ORDINARY SHARES
AS WELL AS THE APPLICATION OF THE PFIC RULES AND THE POSSIBILITY OF MAKING A MARK-TO-MARKET ELECTION.
Taxation
of Dividends and Other Distributions on our Ordinary Shares
Subject
to the PFIC rules discussed above, the gross amount of any distributions we make to you (including the amount of any tax withheld)
with respect to our ordinary shares generally will be includible in your gross income as dividend income on the date of receipt
by the holder, but only to the extent the distribution is paid out of our current or accumulated earnings and profits (as determined
under U.S. federal income tax principles). The dividends will not be eligible for the dividends-received deduction allowed to
corporations in respect of dividends received from other U.S. corporations. To the extent the amount of the distribution exceeds
our current and accumulated earnings and profits (as determined under U.S. federal income tax principles), such excess amount
will be treated first as a tax-free return of your tax basis in your ordinary shares, and then, to the extent such excess amount
exceeds your tax basis in your ordinary shares, as capital gain. We currently do not, and we do not intend to, calculate our earnings
and profits under U.S. federal income tax principles. Therefore, you should expect that a distribution will generally be reported
as a dividend even if that distribution would otherwise be treated as a non-taxable return of capital or as capital gain under
the rules described above.
With
respect to certain non-corporate U.S. Holders, including individual U.S. Holders, dividends may be taxed at the lower capital
gain rates applicable to “qualified dividend income,” provided (1) our ordinary shares are readily tradable on an
established securities market in the United States (such as NASDAQ), (2) we are neither a PFIC nor treated as such with respect
to you (as discussed above) for either the taxable year in which the dividend was paid or the preceding taxable year, (3) certain
holding period requirements are met and (4) you are not under an obligation to make related payments with respect to positions
in substantially similar or related property. As discussed above under “Passive foreign investment company,” there
is a significant risk that we will be a PFIC for U.S. federal income tax purposes, and, as a result, the qualified dividend rate
may be unavailable with respect to dividends we pay.
The
amount of any distribution paid in a currency other than U.S. dollars will be equal to the U.S. dollar value of such currency
on the date such distribution is includible in your income, regardless of whether the payment is in fact converted into U.S. dollars
at that time. The amount of any distribution of property other than cash will be the fair market value of such property on the
date of distribution.
Any
dividends will constitute foreign source income for foreign tax credit limitation purposes. If the dividends are taxed as qualified
dividend income (as discussed above), the amount of the dividend taken into account for purposes of calculating the foreign tax
credit limitation will in general be limited to the gross amount of the dividend, multiplied by the reduced tax rate applicable
to qualified dividend income and divided by the highest tax rate normally applicable to dividends. The limitation on foreign taxes
eligible for credit is calculated separately with respect to specific classes of income. For this purpose, dividends distributed
by us with respect to our ordinary shares will generally constitute “passive category income” but could, in the case
of certain U.S. Holders, constitute “general category income.”
If
Israeli withholding taxes apply to any dividends paid to you with respect to our ordinary shares, subject to certain conditions
and limitations, such withholding taxes may be treated as foreign taxes eligible for credit against your U.S. federal income tax
liability. Instead of claiming a credit, you may elect to deduct such taxes in computing taxable income, subject to applicable
limitations. If a refund of the tax withheld is available under the applicable laws of Israel or under the Israel-U.S. income
tax treaty (the “Treaty”), the amount of tax withheld that is refundable will not be eligible for such credit against
your U.S. federal income tax liability (and will not be eligible for the deduction against your U.S. federal taxable income).
The rules relating to the determination of the foreign tax credit are complex, and you should consult your tax advisor regarding
the availability of a foreign tax credit in your particular circumstances, including the effects of the Treaty.
Taxation
of Disposition of Ordinary Shares
Subject
to the PFIC rules discussed above, upon a sale or other disposition of ordinary shares, you will generally recognize capital gain
or loss for U.S. federal income tax purposes in an amount equal to the difference between the amount realized (including the amount
of any tax withheld) and your tax basis in such ordinary shares. If the consideration you receive for our ordinary shares is not
paid in U.S. dollars, the amount realized will be the U.S. dollar value of the payment received determined by reference to the
spot rate of exchange on the date of the sale or other disposition. However, if our ordinary shares are treated as traded on an
“established securities market” and you are either a cash basis taxpayer or an accrual basis taxpayer that has made
a special election (which must be applied consistently from year to year and cannot be changed without the consent of the IRS),
you will determine the U.S. dollar value of the amount realized in a non-U.S. dollar currency by translating the amount received
at the spot rate of exchange on the settlement date of the sale. If you are an accrual basis taxpayer that is not eligible to
or does not elect to determine the amount realized using the spot rate on the settlement date, you will recognize foreign currency
gain or loss to the extent of any difference between the U.S. dollar amount realized on the date of sale or disposition and the
U.S. dollar value of the currency received at the spot rate on the settlement date.
Your
tax basis in our ordinary shares generally will equal the cost of such ordinary shares. Any gain or loss on the sale or other
disposition of our ordinary shares will generally be treated as U.S. source income or loss, and treated as long-term capital gain
or loss if your holding period in our ordinary shares at the time of the disposition exceeds one year. Accordingly, in the event
any Israeli tax (including withholding tax) is imposed upon the sale or other disposition, you may not be able to utilize foreign
tax credit unless you have foreign source income or gain in the same category from other sources. Long-term capital gain of non-corporate
U.S. Holders generally will be subject to U.S. federal income tax at reduced tax rates. The deductibility of capital losses is
subject to significant limitations.
Information
Reporting and Backup Withholding
Dividend
payments with respect to ordinary shares and proceeds from the sale, exchange or redemption of ordinary shares may be subject
to information reporting to the IRS and possible U.S. backup withholding. Backup withholding will not apply, however, to a U.S.
Holder that furnishes a correct taxpayer identification number and makes any other required certification or that is otherwise
exempt from backup withholding. U.S. Holders that are required to establish their exempt status generally must provide such certification
on IRS Form W-9. You should consult your tax advisor regarding the application of the U.S. information reporting and backup withholding
rules.
Backup
withholding is not an additional tax. Amounts withheld as backup withholding may be credited against your U.S. federal income
tax liability, and you may obtain a refund of any excess amounts withheld under the backup withholding rules by filing the appropriate
claim for refund with the IRS and furnishing any required information in a timely manner.
Information
with respect to Foreign Financial Assets
Certain
U.S. Holders may be required to report information relating to an interest in our ordinary shares, subject to certain exceptions
(including an exception for ordinary shares held in accounts maintained by certain U.S. financial institutions). Penalties can
apply if U.S. Holders fail to satisfy such reporting requirements. You should consult your tax advisor regarding the effect, if
any, of this requirement on your ownership and disposition of our ordinary shares.
THE
SUMMARY OF U.S. FEDERAL INCOME TAX CONSEQUENCES SET OUT ABOVE IS FOR GENERAL INFORMATIONAL PURPOSES ONLY. INVESTORS ARE URGED
TO CONSULT THEIR TAX ADVISORS ABOUT THE APPLICATION OF THE U.S. FEDERAL TAX RULES TO THEIR PARTICULAR CIRCUMSTANCES AS WELL AS
THE STATE, LOCAL, NON-U.S. AND OTHER TAX CONSEQUENCES TO THEM OF THE PURCHASE, OWNERSHIP AND DISPOSITION OF OUR ORDINARY SHARES.
Plan
of
DISTRIBUTION
Pursuant
to an engagement agreement, we have engaged Rodman & Renshaw, a unit of H.C. Wainwright & Co., LLC, or the placement agent,
to act as our exclusive placement agent in connection with this offering of our securities pursuant to this prospectus on a reasonable
best efforts basis. The terms of this offering are subject to market conditions and negotiations between us, the placement agent
and prospective investors. The engagement agreement does not give rise to any commitment by the placement agent to purchase any
of our securities, and the placement agent will have no authority to bind us by virtue of the engagement agreement. The placement
agent may engage sub-agents or selected dealers to assist with the offering.
Only
certain institutional investors purchasing the securities offered hereby will execute a securities purchase agreement with us,
providing such investors with certain representations, warranties and covenants from us, which representations, warranties and
covenants will not be available to other investors who will not execute a securities purchase agreement in connection with the
purchase of the securities offered pursuant to this prospectus. Therefore, those investors shall rely solely on this prospectus
in connection with the purchase of securities in the offering.
The
placement agent is not purchasing or selling any of the securities offered by us under this prospectus, nor is it required to
arrange the purchase or sale of any specific number or dollar amount of securities. The placement agent has agreed to use reasonable
best efforts to arrange for the sale of the securities. There is no required minimum number of securities that must be sold as
a condition to completion of this offering. Further, the placement agent does not guarantee that it will be able to raise new
capital in any prospective offering.
The
securities purchase agreement we entered with the investors provides that the obligations of the investors of the securities are
subject to certain conditions precedent, including, among other things, the absence of any material adverse change in our business
and receipt of customary legal opinions, letters and certificates. We will deliver the securities being issued to the investors
upon receipt of investor funds for the purchase of the securities offered pursuant to this prospectus. We expect to deliver the
securities being offered pursuant to this prospectus on or about March , 2017.
Upon
closing, we will deliver to each investor delivering funds the number of ADSs and warrants purchased by such investor through
the facilities of The Bank of New York Mellon.
The
placement agent may be deemed to be an underwriter within the meaning of Section 2(a)(11) of the Securities Act, and any commissions
received by the placement agent and any profit realized by it while acting as principal might be deemed to be underwriting discounts
or commissions under the Securities Act. As an underwriter, the placement agent would be required to comply with the requirements
of the Securities Act and the Exchange Act, including, without limitation, Rule 415(a)(4) under the Securities Act and Rule 10b-5
and Regulation M under the Exchange Act. These rules and regulations may limit the timing of purchases and sales of ordinary shares
by the placement agent acting as principal. Under these rules and regulations, the placement agent:
|
|
●
|
may
not engage in any stabilization activity in connection with our securities; and
|
|
|
●
|
may
not bid for or purchase any of our securities or attempt to induce any person to purchase
any of our securities, other than as permitted under the Exchange Act, until it has completed
its participation in the distribution.
|
Commissions
and Expenses
We
have agreed to pay the placement agent a total cash fee equal to 7% of the gross proceeds of this offering, except with respect
to cash consideration paid to us in this offering by certain existing shareholders, in which case we will pay the placement agent
a cash fee equal to 4% of the gross proceeds received from such investors. We will also pay the placement agent a management fee
equal to 1% of the gross proceeds of this offering, a reimbursement for non-accountable expenses of $40,000 and a reimbursement
for the placement agent’s legal fees and expenses in the amount of up to $100,000. This fee will be distributed among the
placement agent and any selected-dealers that it has retained to act on their behalf in connection with this offering. We estimate
the total offering expenses of this offering that will be payable by us, excluding the placement agent’s fees and expenses,
will be approximately $243,000.
Placement
Agent Warrants
In addition, we have agreed to issue to
the placement agent warrants to purchase up to ADSs, which represents 7% of the aggregate number of ADSs sold in this
offering (including the number of ADSs issuable upon exercise of the Series B warrants issued in this offering) at an
exercise price of $ per ADS (representing 125% of the public offering
price for a Class A unit to be sold in this offering), exercisable for five years from the date of issuance. The placement
agent warrants will otherwise have substantially the same terms as the Series A warrants being sold to the investors in this
offering. Pursuant to FINRA Rule 5110(g), the placement agent warrants and any ordinary shares issued upon exercise of the
placement agent warrants shall not be sold, transferred, assigned, pledged, or hypothecated, or be the subject of any
hedging, short sale, derivative, put or call transaction that would result in the effective economic disposition of the
securities by any person for a period of 180 days immediately following the date of effectiveness or commencement of sales of
this offering, except the transfer of any security: (i) by operation of law or by reason of our reorganization; (ii) to any
FINRA member firm participating in the offering and the officers or partners thereof, if all securities so transferred remain
subject to the lock-up restriction set forth above for the remainder of the time period; (iii) if the aggregate amount of our
securities held by the placement agent or related persons do not exceed 1% of the securities being offered; (iv) that is
beneficially owned on a pro-rata basis by all equity owners of an investment fund, provided that no participating member
manages or otherwise directs investments by the fund and the participating members in the aggregate do not own more than 10%
of the equity in the fund; or (v) the exercise or conversion of any security, if all securities remain subject to the lock-up
restriction set forth above for the remainder of the time period.
Preemptive Rights
Pursuant to the terms of the securities purchase
agreements relating to an offering of our ADSs and warrants which closed on December 6, 2016, we granted the purchasers in such
offering an eighteen month contractual preemptive right to participate in future financings, subject to certain exceptions, in
an amount equal to up to 50% of the amount raised in such future financings. Other than the preemptive rights described herein,
our ordinary shares and ADSs are not subject to any other preemptive rights.
Right
of First Refusal
We have also agreed to give the placement agent,
subject to the completion of this offering, a right of first refusal, for twelve months from the commencement of sales in the
offering, to act as our lead underwriter or placement agent for any further capital raising transactions undertaken by us and
a twelve-month tail fee equal to the cash and warrant compensation in this offering, if any investor who was contacted by the
placement agent provides us with further capital during such twelve-month period following the expiration or termination of our
engagement with the placement agent.
Lock-up
Agreements
We and our executive officers and
directors have agreed, subject to certain exceptions, not to offer, sell, agree to sell, directly or indirectly, or otherwise
dispose of any ordinary shares, ADSs or warrants or any other securities convertible into or exchangeable for ordinary shares
except for the ordinary shares offered in this offering without the prior written consent of the representative for a period of
90 days after the consummation of this offering.
Indemnification
We
have agreed to indemnify the placement agent and specified other persons against certain liabilities relating to or arising out
of the placement agent’s activities under the placement agency agreement and to contribute to payments that the placement
agent may be required to make in respect of such liabilities.
Determination
of offering price
The
offering price of the securities we are offering was negotiated between us and the investors, in consultation with the placement
agent based on the trading of our ADSs prior to the offering, among other things. Other factors considered in determining the
offering price of the ADSs we are offering include the history and prospects of the company, the stage of development of our business,
our business plans for the future and the extent to which they have been implemented, an assessment of our management, general
conditions of the securities markets at the time of the offering and such other factors as were deemed relevant.
Listing
Our ADSs are listed on The NASDAQ Capital Market
under the trading symbol “MDGS.” The Series A warrants and Series B warrants are not and will not be listed for trading
on any national securities exchange.
Other
Relationships
From
time to time, the placement agent has provided, and may provide in the future, various advisory, investment and commercial banking
and other services to us in the ordinary course of business, for which they have received and may continue to receive customary
fees and commissions. However, except as disclosed in this prospectus, we have no present arrangements with the placement agent
for any further services.
The
placement agent in this offering served as our exclusive placement agent in a securities offering we consummated in December 2016,
pursuant to which it received compensation, including warrants to purchase our ADSs.
Selling
Restrictions outside the United States
This
prospectus does not constitute an offer to sell to, or a solicitation of an offer to buy from, anyone in any country or jurisdiction
(i) in which such an offer or solicitation is not authorized, (ii) in which any person making such offer or solicitation is not
qualified to do so or (iii) in which any such offer or solicitation would otherwise be unlawful. No action has been taken that
would, or is intended to, permit a public offer of the securities or possession or distribution of this prospectus or any other
offering or publicity material relating to the securities in any country or jurisdiction (other than the United States) where
any such action for that purpose is required. Accordingly, the placement agent has undertaken that it will not, directly or indirectly,
offer or sell any ordinary shares or have in its possession, distribute or publish any prospectus, form of application, advertisement
or other document or information in any country or jurisdiction except under circumstances that will, to the best of its knowledge
and belief, result in compliance with any applicable laws and regulations and all offers and sales of securities by it will be
made on the same terms.
Israel
This document does not constitute a prospectus
under the Israeli Securities Law, 5728-1968, and has not been filed with or approved by the Israel Securities Authority. In Israel,
this prospectus may be distributed only to, and is directed only at, investors listed in the first addendum, or the Addendum,
to the Israeli Securities Law, consisting primarily of joint investment in trust funds; provident funds; insurance companies;
banks; portfolio managers, investment advisors, members of the Tel Aviv Stock Exchange Ltd., underwriters, each purchasing for
their own account; venture capital funds; entities with equity in excess of NIS 50 million and “qualified individuals,”
each as defined in the Addendum (as it may be amended from time to time), collectively referred to as qualified investors and
up to additional 35 non-qualified investors in a 12 month period. Qualified investors shall be required to submit written confirmation
that they fall within the scope of the Addendum.
Expenses
Related to Offering
The
following table sets forth the costs and expenses, other than placement agent’s fees, payable by us in connection with the
offer and sale of ordinary shares in this offering. All amounts listed below are estimates except the SEC registration fee and
the Financial Industry Regulatory Authority, Inc., or FINRA, filing fee.
|
Itemized expense
|
|
Amount
|
|
|
SEC registration fee
|
|
$
|
1,970
|
|
|
FINRA filing fee
|
|
|
3,075
|
|
|
Legal fees and expenses
|
|
|
158,000
|
|
|
Transfer agent and registrar fees
|
|
|
22,000
|
|
|
Accounting fees and expenses
|
|
|
50,000
|
|
|
Miscellaneous
|
|
|
7,955
|
|
|
Total
|
|
$
|
243,000
|
|
Legal
Matters
The validity of the securities offered hereby
and certain matters of Israeli law will be passed upon for us by
Meitar Liquornik Geva Leshem
Tal
, Ramat Gan, Israel. Certain matters of United States federal securities law relating to this offering will be passed
upon for us by Zysman, Aharoni, Gayer and Sullivan & Worcester, LLP, New York, New York. Certain matters of United States
federal securities law relating to this offering will be passed upon for the placement agent by
McDermott,
Will & Emery LLP, New York, New York
. Certain matters of Israeli law relating to this offering will be passed upon
for the placement agent by
Barnea & Co.
, Tel-Aviv, Israel.
Experts
The
financial statements as of December 31, 2015 and 2014 and for each of the three years in the period ended December 31, 2015, included
in this prospectus (which contains an explanatory paragraph relating to the Company’s ability to continue as a going concern
as described in Note 1d to the financial statements) have been so included in reliance on the report of Kesselman & Kesselman,
Certified Public Accountants (Isr.), a member firm of PricewaterhouseCoopers International Limited, an independent registered
public accounting firm, given on the authority of said firm as experts in auditing and accounting.
Enforceability
of Civil Liabilities
We
are incorporated under the laws of the State of Israel. Service of process upon us and upon our directors and officers and the
Israeli experts named in this prospectus, substantially all of whom reside outside the United States, may be difficult to obtain
within the United States. Furthermore, because substantially all of our assets and substantially all of our directors and officers
are located outside the United States, any judgment obtained in the United States against us or any of our directors and officers
may not be collectible within the United States
We
have irrevocably appointed Medigus USA LLC as our agent to receive service of process in any action against us in any U.S. federal
or state court arising out of this offering or any purchase or sale of securities in connection with this offering. The address
of our agent is 140 Town & Country Dr., Suite C Danville, CA 94526, USA.
We
have been informed by our legal counsel in Israel, Meitar Liquornik Geva Leshem Tal, that it may be difficult to initiate an action
with respect to U.S. securities law in Israel. Israeli courts may refuse to hear a claim based on an alleged violation of U.S.
securities laws reasoning that Israel is not the most appropriate forum to hear such a claim. In addition, even if an Israeli
court agrees to hear a claim, it may determine that Israeli law and not U.S. law is applicable to the claim. If U.S. law is found
to be applicable, the content of applicable U.S. law must be proved as a fact by expert witnesses which can be a time-consuming
and costly process. Certain matters of procedure may also be governed by Israeli law.
Subject
to certain time limitations and legal procedures, Israeli courts may enforce a U.S. judgment in a civil matter which, subject
to certain exceptions, is non-appealable, including judgments based upon the civil liability provisions of the Securities Act
and the Exchange Act and including a monetary or compensatory judgment in a non-civil matter, provided that:
|
|
●
|
the
judgment was rendered by a court which was, according to the laws of the state of the
court, competent to render the judgment;
|
|
|
●
|
the
judgment may no longer be appealed;
|
|
|
●
|
the
obligation imposed by the judgment is enforceable according to the rules relating to
the enforceability of judgments in Israel and the substance of the judgment is not contrary
to public policy; and
|
|
|
●
|
the
judgment is executory in the state in which it was given.
|
Even
if these conditions are met, an Israeli court will not declare a foreign civil judgment enforceable if:
|
|
●
|
the
judgment was given in a state whose laws do not provide for the enforcement of judgments
of Israeli courts (subject to exceptional cases);
|
|
|
●
|
the
enforcement of the judgment is likely to prejudice the sovereignty or security of the
State of Israel;
|
|
|
●
|
the
judgment was obtained by fraud;
|
|
|
●
|
the
opportunity given to the defendant to bring its arguments and evidence before the court
was not reasonable in the opinion of the Israeli court;
|
|
|
●
|
the
judgment was rendered by a court not competent to render it according to the laws of
private international law as they apply in Israel;
|
|
|
●
|
the
judgment is contradictory to another judgment that was given in the same matter between
the same parties and that is still valid; or
|
|
|
●
|
at
the time the action was brought in the foreign court, a lawsuit in the same matter and
between the same parties was pending before a court or tribunal in Israel.
|
If
a foreign judgment is enforced by an Israeli court, it generally will be payable in Israeli currency, which can then be converted
into non-Israeli currency and transferred out of Israel. The usual practice in an action before an Israeli court to recover an
amount in a non-Israeli currency is for the Israeli court to issue a judgment for the equivalent amount in Israeli currency at
the rate of exchange in force on the date of the judgment, but the judgment debtor may make payment in foreign currency. Pending
collection, the amount of the judgment of an Israeli court stated in Israeli currency ordinarily will be linked to the Israeli
consumer price index plus interest at the annual statutory rate set by Israeli regulations prevailing at the time. Judgment creditors
must bear the risk of unfavorable exchange rates.
Where
You Can Find Additional Information
We
are subject to the informational requirements of the United States Securities Exchange Act of 1934, as amended, and in accordance
therewith file annual and special reports with, and furnish other information to, the SEC. You may read and copy the registration
statement and any other documents we have filed at the SEC, including any exhibits and schedules, at the SEC’s public reference
room at 100 F Street N.E., Washington, D.C. 20549. You may call the SEC at 1-800-SEC-0330 for further information on this public
reference room. In addition, the SEC maintains a web site that contains reports and other information regarding issuers that file
electronically with the SEC. You may access the SEC’s website at http://www.sec.gov. These SEC filings are also available
to the public on the Israel Securities Authority’s Magna website at www.magna.isa.gov.il and from commercial document retrieval
services.
This
prospectus is part of the registration statement on Form F-1 filed with the SEC in connection with this offering and does not
contain all of the information included in the registration statement. Whenever a reference is made in this prospectus to any
of our contracts or other documents, the reference may not be complete and, for a copy of the contract or document, you should
refer to the exhibits that are a part of the registration statement.
MEDIGUS
LTD.
CONSOLIDATED
FINANCIAL STATEMENTS
AS
OF DECEMBER 31, 2015
MEDIGUS
LTD.
CONSOLIDATED
INTERIM FINANCIAL STATEMENTS
AS
OF DECEMBER 31, 2015
TABLE
OF CONTENTS

Report
of Independent Registered Public Accounting Firm
To
the shareholders of Medigus Ltd.,
In
our opinion, the accompanying consolidated statements of financial position and the related consolidated statements of loss and
other comprehensive loss, of changes in equity and of cash flows present fairly, in all material respects, the financial position
of Medigus Ltd. and its subsidiary at December 31, 2015 and 2014, and the results of their operations and their cash flows for
each of the three years in the period ended December 31, 2015, in conformity with International Financial Reporting Standards
as issued by the International Accounting Standards Board. These financial statements are the responsibility of the Company’s
Board of Directors and management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audits of these statements in accordance with the standards of the Public Company Accounting Oversight Board
(United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial
statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and
disclosures in the financial statements, assessing the accounting principles used and significant estimates made by the Board
of Directors and management, and evaluating the overall financial statement presentation. We believe that our audits provide a
reasonable basis for our opinion.
As
discussed in Note 1c to the financial statements, the Company has effected a reverse split of its ordinary shares and retrospectively
applied this split to all periods presented.
The
accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed
in Note 1d to the financial statements, the Company has suffered recurring losses from operations and has a net capital deficiency
that raises substantial doubt about its ability to continue as a going concern. Management’s plans in regard to these matters
are also described in Note 1d. The financial statements do not include any adjustments that might result from the outcome of this
uncertainty.
|
Tel-Aviv,
Israel
|
Kesselman
& Kesselman
|
|
March
30, 2016
|
Certified
Public Accountants (Isr.)
|
|
|
A
member firm of PricewaterhouseCoopers International Limited
|
Kesselman
& Kesselman, Trade Tower, 25 Hamered Street, Tel-Aviv 6812508, Israel,
P.O
Box 50005 Tel-Aviv 6150001 Telephone: +972 -3-
MEDIGUS LTD.
CONSOLIDATED
STATEMENTS OF FINANCIAL POSITION
|
|
|
|
|
|
December 31
|
|
|
|
|
Note
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
|
|
NIS in thousands
|
|
|
Assets
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CURRENT ASSETS:
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
|
5
|
|
|
|
40,239
|
|
|
|
42,067
|
|
|
Financial assets at fair value through profit or loss
|
|
|
4
|
|
|
|
|
|
|
|
8,187
|
|
|
Accounts receivable:
|
|
|
6
|
|
|
|
|
|
|
|
|
|
|
Trade
|
|
|
|
|
|
|
992
|
|
|
|
513
|
|
|
Other
|
|
|
|
|
|
|
3,924
|
|
|
|
1,738
|
|
|
Inventory
|
|
|
2 (i), 7
|
|
|
|
1,187
|
|
|
|
1,403
|
|
|
|
|
|
|
|
|
|
46,342
|
|
|
|
53,908
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
NON-CURRENT ASSETS:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Inventory
|
|
|
2 (i), 7
|
|
|
|
|
|
|
|
541
|
|
|
Property and equipment
|
|
|
8
|
|
|
|
880
|
|
|
|
945
|
|
|
Intangible assets
|
|
|
|
|
|
|
152
|
|
|
|
185
|
|
|
|
|
|
|
|
|
|
1,032
|
|
|
|
1,671
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL ASSETS
|
|
|
|
|
|
|
47,374
|
|
|
|
55,579
|
|
The
accompanying notes are an integral part of these consolidated financial statements.
MEDIGUS LTD.
CONSOLIDATED
STATEMENTS OF FINANCIAL POSITION
|
|
|
|
|
|
December 31
|
|
|
|
|
Note
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
|
|
NIS in thousands
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities and equity
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CURRENT LIABILITIES -
|
|
|
|
|
|
|
|
|
|
|
Accounts payable and accruals:
|
|
|
10
|
|
|
|
|
|
|
|
|
|
|
Trade
|
|
|
|
|
|
|
1,395
|
|
|
|
791
|
|
|
Other
|
|
|
|
|
|
|
5,836
|
|
|
|
3,223
|
|
|
|
|
|
|
|
|
|
7,231
|
|
|
|
4,014
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
NON-CURRENT LIABILITIES:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Warrants at fair value
|
|
|
4
|
|
|
|
34
|
|
|
|
428
|
|
|
Retirement benefit obligation, net
|
|
|
|
|
|
|
381
|
|
|
|
381
|
|
|
|
|
|
|
|
|
|
415
|
|
|
|
809
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
COMMITMENTS
|
|
|
11
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL LIABILITIES
|
|
|
|
|
|
|
7,646
|
|
|
|
4,823
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
EQUITY:
|
|
|
12
|
|
|
|
|
|
|
|
|
|
|
Ordinary share capital
|
|
|
|
|
|
|
3,204
|
|
|
|
2,499
|
|
|
Share premium
|
|
|
|
|
|
|
193,113
|
|
|
|
170,741
|
|
|
Other capital reserves
|
|
|
|
|
|
|
4,640
|
|
|
|
4,498
|
|
|
Warrants
|
|
|
|
|
|
|
5,586
|
|
|
|
2,828
|
|
|
Accumulated deficit
|
|
|
|
|
|
|
(166,815
|
)
|
|
|
(129,810
|
)
|
|
TOTAL EQUITY
|
|
|
|
|
|
|
39,728
|
|
|
|
50,756
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL LIABILITIES AND EQUITY
|
|
|
|
|
|
|
47,374
|
|
|
|
55,579
|
|
The
accompanying notes are an integral part of these consolidated financial statements.
MEDIGUS LTD.
CONSOLIDATED
STATEMENTS OF LOSS AND OTHER COMPREHENSIVE LOSS
|
|
|
|
|
|
For the Year Ended December 31
|
|
|
|
|
Note
|
|
|
2015
|
|
|
2014
|
|
|
2013
|
|
|
|
|
|
|
|
NIS in thousands
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
REVENUES
|
|
|
18
|
|
|
|
2,416
|
|
|
|
2,664
|
|
|
|
2,498
|
|
|
COST OF REVENUES
|
|
|
13
|
|
|
|
1,073
|
|
|
|
1,252
|
|
|
|
1,126
|
|
|
GROSS PROFIT
|
|
|
|
|
|
|
1,343
|
|
|
|
1,412
|
|
|
|
1,372
|
|
|
RESEARCH AND DEVELOPMENT EXPENSES, NET
|
|
|
13
|
|
|
|
17,020
|
|
|
|
14,401
|
|
|
|
8,180
|
|
|
SELLING AND MARKETING EXPENSES
|
|
|
13
|
|
|
|
10,391
|
|
|
|
8,353
|
|
|
|
3,234
|
|
|
ADMINISTRATIVE AND GENERAL
EXPENSES
|
|
|
13
|
|
|
|
11,028
|
|
|
|
8,206
|
|
|
|
6,877
|
|
|
OTHER INCOME, NET
|
|
|
14
|
|
|
|
10
|
|
|
|
941
|
|
|
|
666
|
|
|
OPERATING LOSS
|
|
|
|
|
|
|
(37,086
|
)
|
|
|
(28,607
|
)
|
|
|
(16,253
|
)
|
|
PROFIT FROM CHANGES IN FAIR VALUE OF WARRANTS ISSUED TO INVESTORS
|
|
|
4
|
|
|
|
394
|
|
|
|
3,605
|
|
|
|
11,544
|
|
|
FINANCING INCOME (EXPENSES) IN RESPECT OF DEPOSITS AND EXCHANGE DIFFERENCES
|
|
|
|
|
|
|
78
|
|
|
|
2,513
|
|
|
|
(182
|
)
|
|
FINANCING EXPENSES IN RESPECT OF BANK COMMISSIONS
|
|
|
|
|
|
|
(127
|
)
|
|
|
(127
|
)
|
|
|
(213
|
)
|
|
FINANCING INCOME (EXPENSES), NET
|
|
|
15
|
|
|
|
(49
|
)
|
|
|
2,386
|
|
|
|
(395
|
)
|
|
LOSS BEFORE TAXES ON INCOME
|
|
|
|
|
|
|
(36,741
|
)
|
|
|
(22,616
|
)
|
|
|
(5,104
|
)
|
|
TAXES ON INCOME
|
|
|
9
|
|
|
|
(264
|
)
|
|
|
(13
|
)
|
|
|
(85
|
)
|
|
LOSS FOR THE YEAR
|
|
|
|
|
|
|
(37,005
|
)
|
|
|
(22,629
|
)
|
|
|
(5,189
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
OTHER COMPREHENSIVE INCOME (LOSS):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Amounts which will not be reclassified to
profit or loss - re-measurement of net liabilities for employee benefits
|
|
|
|
|
|
|
|
|
|
|
(105
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Amounts which may be subsequently reclassified to profit or loss:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Currency translation differences
|
|
|
|
|
|
|
2
|
|
|
|
14
|
|
|
|
|
|
|
Transfer of capital reserve in respect of financial asset available for sale to statement of loss, net of tax
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(254
|
)
|
|
OTHER COMPREHENSIVE INCOME (LOSS) FOR THE YEAR, NET OF TAX
|
|
|
|
|
|
|
2
|
|
|
|
(91
|
)
|
|
|
(254
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL
COMPREHENSIVE LOSS FOR THE YEAR
|
|
|
|
|
|
|
(37,003
|
)
|
|
|
(22,720
|
)
|
|
|
(5,443
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
NIS
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
BASIC AND DILUTED LOSS PER SHARE
|
|
|
16
|
|
|
|
(1.3
|
)
|
|
|
(1.2
|
)
|
|
|
(0.4
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
WEIGHTED AVERAGE OF ORDINARY SHARES (IN THOUSANDS)
|
|
|
|
|
|
|
28,415
|
|
|
|
19,500
|
|
|
|
13,020
|
|
The
accompanying notes are an integral part of these consolidated financial statements.
MEDIGUS
LTD.
CONSOLIDATED
STATEMENTS OF CHANGES IN EQUITY
(Continued)
- 1
|
|
|
|
|
|
|
|
Equity
attributed to the owners of the company
|
|
|
|
|
|
|
|
Note
|
|
|
|
Ordinary
shares
|
|
|
|
Share
premium
|
|
|
|
Capital
reserves from options granted
|
|
|
|
Capital
reserves from transactions with controlling
shareholders
|
|
|
|
Capital
reserve
from financial asset
available
for sale
|
|
|
|
Warrants
|
|
|
|
Accumulated
deficit
|
|
|
Total
equity
|
|
|
|
|
|
|
|
|
NIS in thousands
|
|
BALANCE
AS OF JANUARY 1, 2013
|
|
|
|
|
|
|
902
|
|
|
|
93,925
|
|
|
|
3,922
|
|
|
|
1,912
|
|
|
|
254
|
|
|
|
3,910
|
|
|
|
(101,887
|
)
|
|
2,938
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL
COMPREHENSIVE LOSS FOR THE YEAR
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(254
|
)
|
|
|
|
|
|
|
(5,189
|
)
|
|
(5,443)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TRANSACTIONS
WITH SHAREHOLDERS:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Proceeds
from issuance of shares and warrants
|
|
|
12B
|
|
|
|
744
|
|
|
|
38,191
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,671
|
|
|
|
|
|
|
40,606
|
|
Options
granted to employees and service providers
|
|
|
12C
|
|
|
|
|
|
|
|
|
|
|
|
511
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
511
|
|
Forfeiture
and expiration of options and warrants
|
|
|
12C
|
|
|
|
|
|
|
|
6,262
|
|
|
|
(2,352
|
)
|
|
|
|
|
|
|
|
|
|
|
(3,910
|
)
|
|
|
|
|
|
|
|
Amount
carried to capital reserve as a result of transaction with controlling shareholder
|
|
|
17
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
138
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
138
|
|
TOTAL
TRANSACTIONS WITH SHAREHOLDERS
|
|
|
|
|
|
|
744
|
|
|
|
44,453
|
|
|
|
(1,841
|
)
|
|
|
138
|
|
|
|
|
|
|
|
(2,239
|
)
|
|
|
|
|
|
41,255
|
|
BALANCE
AS OF DECEMBER 31, 2013
|
|
|
|
|
|
|
1,646
|
|
|
|
138,378
|
|
|
|
2,081
|
|
|
|
2,050
|
|
|
|
-
|
|
|
|
1,671
|
|
|
|
(107,076
|
)
|
|
38,750
|
MEDIGUS
LTD.
CONSOLIDATED
STATEMENTS OF CHANGES IN EQUITY
(Continued)
- 2
|
|
|
|
|
|
|
|
Equity attributed to the owners of the company
|
|
|
|
|
|
|
|
Note
|
|
|
|
Ordinary
shares
|
|
|
|
Share
premium
|
|
|
|
Capital
reserves from options granted
|
|
|
|
Capital
reserves
from transactions with controlling
shareholders
|
|
|
|
Currency
translation
differences
|
|
|
|
Warrants
|
|
|
|
Accumulated
deficit
|
|
|
Total
equity
|
|
|
|
|
|
|
|
|
NIS in thousands
|
|
|
BALANCE
AS OF JANUARY 1, 2014
|
|
|
|
|
|
|
1,646
|
|
|
|
138,378
|
|
|
|
2,081
|
|
|
|
2,050
|
|
|
|
|
|
|
|
1,671
|
|
|
|
(107,076
|
)
|
|
38,750
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL
COMPREHENSIVE INCOME (LOSS) FOR THE YEAR
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
14
|
|
|
|
|
|
|
|
(22,734
|
)
|
|
(22,720)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TRANSACTIONS
WITH SHAREHOLDERS:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Proceeds
from issuance of shares and warrants
|
|
|
12B
|
|
|
|
853
|
|
|
|
31,605
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,157
|
|
|
|
|
|
|
33,615
|
|
Options
granted to employees and service providers
|
|
|
12C
|
|
|
|
|
|
|
|
|
|
|
|
1,111
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,111
|
|
Forfeiture
and expiration of options and warrants
|
|
|
12C
|
|
|
|
|
|
|
|
758
|
|
|
|
(758
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL
TRANSACTIONS WITH SHAREHOLDERS
|
|
|
|
|
|
|
853
|
|
|
|
32,363
|
|
|
|
353
|
|
|
|
|
|
|
|
|
|
|
|
1,157
|
|
|
|
|
|
|
34,726
|
|
BALANCE
AS OF DECEMBER 31, 2014
|
|
|
|
|
|
|
2,499
|
|
|
|
170,741
|
|
|
|
2,434
|
|
|
|
2,050
|
|
|
|
14
|
|
|
|
2,828
|
|
|
|
(129,810
|
)
|
|
50,756
|
MEDIGUS
LTD.
CONSOLIDATED
STATEMENTS OF CHANGES IN EQUITY
(Concluded)
- 3
|
|
|
|
|
|
|
|
Equity attributed to the owners of the company
|
|
|
|
|
|
|
|
Note
|
|
|
|
Ordinary
shares
|
|
|
|
Share
premium
|
|
|
|
Capital
reserves from options granted
|
|
|
|
Capital
reserves
from transactions with controlling
shareholders
|
|
|
|
Currency
translation
differences
|
|
|
|
Warrants
|
|
|
|
Accumulated
deficit
|
|
|
Total
equity
|
|
|
|
|
|
|
|
|
NIS in thousands
|
|
BALANCE
AS OF JANUARY 1, 2015
|
|
|
|
|
|
|
2,499
|
|
|
|
170,741
|
|
|
|
2,434
|
|
|
|
2,050
|
|
|
|
14
|
|
|
|
2,828
|
|
|
|
(129,810
|
)
|
|
50,756
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL
COMPREHENSIVE INCOME (LOSS) FOR THE YEAR
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2
|
|
|
|
|
|
|
|
(37,005
|
)
|
|
(37,003)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TRANSACTIONS
WITH SHAREHOLDERS:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Proceeds
from issuance of shares and warrants
|
|
|
12B
|
|
|
|
705
|
|
|
|
22,062
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2,758
|
|
|
|
|
|
|
25,525
|
|
Options
granted to employees and service providers
|
|
|
12C
|
|
|
|
|
|
|
|
|
|
|
|
450
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
450
|
|
Forfeiture
of options
|
|
|
12C
|
|
|
|
|
|
|
|
310
|
|
|
|
(310
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL
TRANSACTIONS WITH SHAREHOLDERS
|
|
|
|
|
|
|
705
|
|
|
|
22,372
|
|
|
|
140
|
|
|
|
|
|
|
|
|
|
|
|
2,758
|
|
|
|
|
|
|
25,975
|
|
BALANCE
AS OF DECEMBER 31, 2015
|
|
|
|
|
|
|
3,204
|
|
|
|
193,113
|
|
|
|
2,574
|
|
|
|
2,050
|
|
|
|
16
|
|
|
|
5,586
|
|
|
|
(166,815
|
)
|
|
39,728
|
The
accompanying notes are an integral part of these consolidated financial statements.
(Continued)
- 1
MEDIGUS
LTD.
CONSOLIDATED
STATEMENTS OF CASH FLOWS
|
|
|
For the year ended December 31
|
|
|
|
|
2015
|
|
|
2014
|
|
|
2013
|
|
|
|
|
NIS in thousands
|
|
|
CASH FLOWS FROM OPERATING ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CASH FLOWS USED IN OPERATIONS (see Appendix)
|
|
|
(26,919
|
)
|
|
|
(28,015
|
)
|
|
|
(22,606
|
)
|
|
Income tax paid
|
|
|
(175
|
)
|
|
|
(13
|
)
|
|
|
|
|
|
Interest received
|
|
|
127
|
|
|
|
96
|
|
|
|
59
|
|
|
NET CASH USED IN OPERATING ACTIVITIES
|
|
|
(26,967
|
)
|
|
|
(27,932
|
)
|
|
|
(22,547
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CASH FLOWS FROM INVESTING ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Purchase of property and equipment
|
|
|
(327
|
)
|
|
|
(240
|
)
|
|
|
(298
|
)
|
|
Purchase of intangible assets
|
|
|
(48
|
)
|
|
|
(149
|
)
|
|
|
(77
|
)
|
|
Withdrawal of short-term deposits
|
|
|
|
|
|
|
8,086
|
|
|
|
881
|
|
|
Investment in short-term deposits
|
|
|
|
|
|
|
|
|
|
|
(9,303
|
)
|
|
Net cash generated from (used in) investing activities
|
|
|
(375
|
)
|
|
|
7,697
|
|
|
|
(8,797
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CASH FLOWS FROM FINANCING ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Proceeds from issuance of shares and warrants, net; note 12b
|
|
|
25,525
|
|
|
|
35,970
|
|
|
|
53,828
|
|
|
Net cash flows generated from financing activities
|
|
|
25,525
|
|
|
|
35,970
|
|
|
|
53,828
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
INCREASE (DECREASE) IN CASH AND CASH EQUIVALENTS
|
|
|
(1,817
|
)
|
|
|
15,735
|
|
|
|
22,484
|
|
|
BALANCE OF CASH AND CASH EQUIVALENTS AT BEGINNING OF YEAR
|
|
|
42,067
|
|
|
|
23,926
|
|
|
|
1,312
|
|
|
GAINS (LOSSES) FROM EXCHANGE DIFFERENCES ON CASH AND CASH EQUIVALENTS
|
|
|
(11
|
)
|
|
|
2,406
|
|
|
|
130
|
|
|
BALANCE OF CASH AND CASH EQUIVALENTS AT END OF YEAR
|
|
|
40,239
|
|
|
|
42,067
|
|
|
|
23,926
|
|
(Concluded-2)
MEDIGUS
LTD.
CONSOLIDATED
STATEMENTS OF CASH FLOWS
APPENDIX
TO THE STATEMENTS OF CASH FLOWS:
|
|
|
For the year ended December 31
|
|
|
|
|
2015
|
|
|
2014
|
|
|
2013
|
|
|
|
|
NIS in thousands
|
|
|
NET CASH USED IN OPERATIONS:
|
|
|
|
|
|
|
|
|
|
|
Loss for the year before taxes on income
|
|
|
(36,741
|
)
|
|
|
(22,616
|
)
|
|
|
(5,104
|
)
|
|
Adjustment in respect of:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Profit on change in the fair value of warrants issued to investors
|
|
|
(394
|
)
|
|
|
(3,605
|
)
|
|
|
(11,544
|
)
|
|
Losses (gains) from exchange differences on cash and cash equivalents
|
|
|
13
|
|
|
|
(2,392
|
)
|
|
|
(130
|
)
|
|
Retirement benefit obligation, net
|
|
|
|
|
|
|
51
|
|
|
|
|
|
|
Gains on change in the fair value of financial instruments at fair value through profit or loss
|
|
|
(10
|
)
|
|
|
(66
|
)
|
|
|
(278
|
)
|
|
Revaluation of and exchange differences on short-term deposits
|
|
|
|
|
|
|
(13
|
)
|
|
|
349
|
|
|
Interest received
|
|
|
(127
|
)
|
|
|
(96
|
)
|
|
|
(59
|
)
|
|
Depreciation
|
|
|
392
|
|
|
|
448
|
|
|
|
315
|
|
|
Amortization of intangible assets
|
|
|
81
|
|
|
|
294
|
|
|
|
105
|
|
|
Amounts charged in respect of options granted to employees and service providers
|
|
|
450
|
|
|
|
1,111
|
|
|
|
511
|
|
|
Amounts carried to capital reserves as a result of transactions with controlling shareholders
|
|
|
|
|
|
|
|
|
|
|
138
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CHANGES IN OPERATING ASSET AND LIABILITY ITEMS:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Decrease (increase) in accounts receivable :
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Trade
|
|
|
(479
|
)
|
|
|
(265
|
)
|
|
|
71
|
|
|
Other
|
|
|
(1,645
|
)
|
|
|
(134
|
)
|
|
|
(873
|
)
|
|
Increase (decrease) in accounts payable and accruals :
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Trade
|
|
|
604
|
|
|
|
263
|
|
|
|
298
|
|
|
Other
|
|
|
2,524
|
|
|
|
(796
|
)
|
|
|
733
|
|
|
Decrease (increase) in inventory
|
|
|
216
|
|
|
|
(36
|
)
|
|
|
(538
|
)
|
|
Net sales (purchases) of financial assets at fair value through profit or loss
|
|
|
8,197
|
|
|
|
(163
|
)
|
|
|
(6,600
|
)
|
|
NET CASH USED IN OPERATIONS
|
|
|
(26,919
|
)
|
|
|
(28,015
|
)
|
|
|
(22,606
|
)
|
The
accompanying notes are an integral part of these consolidated financial statements.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
NOTE
1 - GENERAL:
|
|
a.
|
Medigus
Ltd. (hereinafter – the “Company”) together with its subsidiary (hereinafter
– the “Group”) is a medical device group specializing in research and
development of innovative endoscopic procedures and devices. To date most of the Group’s
research and development activities have been focused in the development of the MUSE
endoscopy system (hereinafter - “MUSE”) for the treatment of gastroesophageal
reflux disease (GERD), which is one of the most common chronic diseases in the western
world. In addition, the Group uses the technological platform it developed for the purpose
of additional special endoscopy-based systems and products and endeavors to enter into
agreements and/or joint ventures with companies in the medical device industry in order
to integrate the systems and products it has developed. To date, the MUSE product has
not generated significant revenues and most of the Group’s revenues arise from sales
of miniature cameras and related equipment, which it developed and manufactures and which
are used in endoscopic procedures.
|
In
addition, the Company has FDA approval to market the MUSE endoscopy system in the USA, and it continues negotiations to market
the main product and sell miniature cameras for endoscopic devices and other endoscopy instruments, which can serve as a source
of future revenues.
The
Company is a limited liability corporation whose shares are listed for trade on the Tel Aviv Stock Exchange Ltd. (“TASE”)
and as of May 20, 2015, the Company’s American Depository Shares (ADSs) evidenced by American Depositary Receipts (ADRs)
are listed for trade on the NASDAQ Capital Market. As of the report date, each ADR represents five ordinary shares of the Company.
The Company’s depositary agent for the ADR program is The Bank of New York Mellon.
The
Company was incorporated in Israel on December 9, 1999 and is resident in Israel. The address of its registered office is P.O.
Box 3030, Omer, 84965.
|
|
b.
|
On
July 22, 2007 the Company established a wholly owned subsidiary, MEDIGUS USA LLC, in
the USA (hereinafter - the “Subsidiary”). The Subsidiary did not engage in
any business activities until October 2013.
|
On
October 1, 2013, the Company and the Subsidiary entered into an agreement whereby the Subsidiary provides services to the Company
in consideration for reimbursement of direct costs plus a reasonable premium.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
1 - GENERAL
(continued)
:
|
|
c.
|
On
May 6, 2015 a special general meeting of the Company’s shareholders approved 1:10
reverse share split such that every 10 ordinary shares par value NIS 0.01 of the Company
shall be converted into one ordinary share par value NIS 0.10, without changing the rights
attached to each share.
|
In
November 2015, the Company effected the 1:10 reverse split of its ordinary shares. Concurrently with the reverse share split,
the Company effected a change in the ratio of ordinary shares to each of its ADRs, such that after the reverse share split each
ADR represents five ordinary shares (instead of fifty ordinary shares).
The
reverse share split reduced the Company’s registered share capital to 150 million ordinary shares and the number of outstanding
ordinary shares to approximately 32.05 million ordinary shares.
Following
the reverse share split, the Company’s share option plans and outstanding options and warrants to purchase its ordinary
shares were also adjusted to reflect the reverse share split.
|
|
d.
|
During
the year ended December 31, 2015, the Group had a total comprehensive loss of NIS 37
million. As of December 31, 2015 the Group had accumulated losses of NIS 167 million
and a positive working capital of NIS 39 million.
|
Based
on the projected cash flows and its cash balances as of December 31, 2015, the Group’s Management is of the opinion that without
further fund raising it will not have sufficient resources to enable it to continue advancing its activities including the development,
manufacturing and marketing of its products for a period of at least 12 months from the date of approval of these financial statements.
As a result, there is substantial doubt about the Group’s ability to continue as a going concern.
Management’s
plans include the continued commercialization of their products, taking cost reduction steps and securing sufficient financing
through the sale of additional equity securities, debt or capital inflows from strategic partnerships. There are no assurances
however, that the Group will be successful in obtaining the level of financing needed for its operations. If the Group is unsuccessful
in commercializing its products and securing sufficient financing, it may need to reduce activities, curtail or cease operations.
The
financial statements do not include any adjustments relating to the recoverability and classification of recorded assets and the
amounts and classification of liabilities that might be necessary should the Group be unable to continue as a going concern.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
2 - SIGNIFICANT ACCOUNTING POLICIES:
|
|
a.
|
Basis
for preparation of the financial statements:
|
The
Group’s financial statements as of December 31, 2015 and 2014 and for each of the three years in the period ended December 31,
2015, are in compliance with International Financial Reporting Standards, which are standards and interpretations thereto issued
by the International Accounting Standard Board (hereinafter “IFRS”).
In
connection with the presentation of these financial statements it is noted as follows:
|
|
1)
|
The
significant accounting policies applied in the preparation of these financial statements
are set out below. These policies have been consistently applied to all years presented,
unless otherwise stated.
|
|
|
2)
|
These
financial statements have been prepared under the historical cost convention, as modified
by the revaluation of plan assets related to the retirement benefit obligation, and financial
assets and financial liabilities (including derivative instruments) measured at fair
value through profit or loss.
|
|
|
3)
|
The
preparation of financial statements in conformity with IFRS requires the use of certain
critical accounting estimates. It also requires the Group’s management to exercise its
judgment in the process of applying the Group’s accounting policies. Areas involving
a higher degree of judgment or complexity, or areas where assumptions and estimates are
significant to the consolidated financial statements are disclosed in note 3. Actual
results may differ materially from estimates and assumptions used by the Group’s management.
|
|
|
4)
|
The
Group analyzes the expenses recognized in the consolidated statement of loss and other
comprehensive loss using a classification method based on the expenses’ function.
|
The
Subsidiary is an entity over which the Company has control. The Company controls an entity when the Company is exposed to, or
has rights to, variable returns from its involvement with the entity and has the ability to affect those returns through its power
over the entity. Subsidiaries are fully consolidated from the date on which control is transferred to the Company. They are deconsolidated
from the date that control ceases. The Subsidiary is fully consolidated.
Inter-company
transactions and balances as well as revenues and expenses relating to intercompany transactions have been eliminated.
Accounting
policies of the Subsidiary are those of the Group and have been consistently applied.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
2 - SIGNIFICANT ACCOUNTING POLICIES
(continued):
|
|
c.
|
Translation of foreign currency balances and transactions
:
|
|
|
1)
|
The
functional currency and the presentation currency
|
Items
included in the financial statements of each of the companies in the Group have been prepared in the currency of the principal
economic environment in which it operates (hereinafter – “the functional currency”). The consolidated financial
statements are presented in New Israel Shekels, which is the Company’s functional and presentation currency, and rounded to the
nearest thousand. The Subsidiary’s functional currency is the US Dollar.
|
|
2)
|
Transactions
and balances
|
Transactions
made in a currency which is different from the functional currency (“foreign currency”) are translated into the functional
currency using the exchange rates prevailing at the dates of the transactions. Foreign exchange gains and losses resulting from
the settlement of such transactions and from the translation at the end-of-year exchange rates of monetary assets and liabilities
denominated in foreign currencies are recognized in income or loss.
Gains
and losses from changes in exchange rates are presented in the statement of comprehensive loss among “Financing income (expenses)
in respect of deposits and exchange differences.”
|
|
3)
|
Translation
of financial statements of the Subsidiary
|
The
results and financial position of the Subsidiary are translated into the presentation currency as follows:
|
|
(a)
|
Assets
and liabilities for each statement of financial position presented are translated at
the closing rate at the date of that statement of financial position, resulting exchange
differences are recognized in other comprehensive income (loss).
|
|
|
(b)
|
Income
and expenses for each income statement are translated at average rates on the period;
|
|
|
d.
|
Property and equipment
|
Property
and equipment are initially recognized at purchased cost. Subsequent costs are included in the asset’s carrying amount or
recognized as a separate asset, as appropriate, only when it is probable that future economic benefits associated with the item
will flow to the Group and the cost of the item can be measured reliably. The carrying amount of replaced items is derecognized.
All other repairs and maintenance are charged to income or loss during the financial period in which they are incurred.
Property
and equipment is recognized at cost less accumulated depreciation.
Depreciation
on assets is calculated using the straight line method of depreciation, in order to depreciate their cost to residual value over
their estimated useful life as follows:
|
|
Machinery and equipment
|
|
6 – 10 years (primarily 10)
|
|
|
Furniture
|
|
7 – 14 years
|
|
|
Computers
|
|
3 years
|
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
2 - SIGNIFICANT ACCOUNTING POLICIES
(continued):
Leasehold
improvements are depreciated using the straight line method over the shorter of the term of the lease or the estimated useful
lives of the improvements.
The
assets’ residual values, their useful lives and the depreciation method are reviewed, and adjusted if appropriate, at the
end of each year.
An
asset’s carrying amount is written down immediately to its recoverable amount if the asset’s carrying amount is greater
than its estimated recoverable amount (see f. below).
Gains
and losses on disposals are determined by comparing the net proceeds with the carrying amount and are recognized within ‘Other
income – net’ in the statement of comprehensive loss.
Licenses
to use purchased computer programs are capitalized on the basis of the costs incurred in their purchase and preparation for use
of the specific program. These costs are depreciated using the straight line method over the estimated useful life of these licenses
(three years).
Costs
related to the maintenance of computer programs are recognized as expenses when incurred.
|
|
2)
|
Research
and development
|
Research
costs are recognized as an expense when incurred. Development costs incurred in respect of design and testing of new or improved
products are recognized as intangible assets only when the following criteria are met:
|
|
●
|
It
is technically feasible to complete the intangible asset so that it will be available
for use;
|
|
|
●
|
Management
intends to complete the intangible asset and use or sell it;
|
|
|
●
|
There
is an ability to use or sell the intangible asset;
|
|
|
●
|
It
can be demonstrated how the intangible asset will generate probable future economic benefits;
|
|
|
●
|
Adequate
technical, financial and other resources to complete the development and to use or sell
the intangible asset are available; and
|
|
|
●
|
The
expenditure attributable to the intangible asset during its development can be reliably
measured.
|
Other
development costs that do not meet these criteria are recognized as an expense as incurred. Development costs previously recognized
as an expense are not recognized as an asset in a subsequent period. Capitalized development costs are presented as intangible
assets, and are amortized from the time when the asset is available for use, meaning when it is in the place and condition intended
by management, using the straight line method, over its useful life.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
2 - SIGNIFICANT ACCOUNTING POLICIES
(continued):
|
|
f.
|
Impairment
of non-monetary assets
|
Non-monetary
assets are tested for impairment whenever events or changes in circumstances indicate that the carrying amount may not be recoverable.
An
impairment loss is recognized for the amount by which the asset’s carrying amount exceeds its recoverable amount. The recoverable
amount is the higher of an asset’s fair value less selling costs and value in use. For the purposes of assessing impairment,
assets are grouped at the lowest levels of identifiable cash flows (cash-generating units). Non-monetary assets that were impaired
are reviewed for possible reversal of the impairment recognized at each statement of financial position date.
Government
grants are recognized at their fair value when there is reasonable assurance that they will be received and the Group will comply
with all the attached conditions.
A
forgivable loan received from the government is accounted for as a government grant when there is reasonable assurance that the
Group will comply with the conditions for forgiveness of the loan.
Government
grants relating to costs are deferred and recognized in the income statement on a systematic basis over the period necessary to
match them with the costs that they are intended to compensate.
Grants
received from the Office of the Chief Scientist in the Ministry of Industry, Trade and Labor (hereinafter - the “Chief Scientist”),
as participation in research and development performed by the Company (hereinafter - “Scientist Grants”) are classified
as “forgivable loans” as set out in International Accounting Standard 20 “Accounting for Government Grants and
Disclosing the Government’s Assistance” (hereinafter -“IAS 20”).
The
Chief Scientist grants received are recognized and measured in accordance with IAS 39. If on the date on which the right for the
Chief Scientist grant is established (hereafter – “the entitlement date”) the Group’s management concludes
that it is not reasonably assured that the Chief Scientist grant to which entitlement has been established, will not be repaid,
the Group recognizes a financial liability on that date, which is accounted for under the provisions of IAS 39 regarding financial
liabilities measured at amortized cost. The difference between the received grant and the fair value of the said financial liability
at date of initial recognition is treated as a government grant recognized in profit or loss as a reduction of research and development
expenses.
In
the event that on entitlement date the Group’s management concludes that there is reasonable assurance that the Chief Scientist
grant which was received will not be repaid, the grant is recorded to income at that date as a reduction of research and development
expenses. If in subsequent periods Group’s management concludes for the first time that there is no reasonable assurance
that the Chief Scientist grant received will not be repaid, the Group recognizes on that date a financial liability against profit
or loss. The aforementioned financial liability is accounted for in accordance with the provisions set out in IAS 39 regarding
financial liabilities measured at amortized cost.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
2 - SIGNIFICANT ACCOUNTING POLICIES
(continued):
Over
all of the reporting periods presented, the Company has concluded that, with respect to all of the grants received from the Chief
Scientist, there is reasonable assurance that the grants received will not be repaid, therefore the grants were carried to income
as reduction of research and development expenses.
|
|
h.
|
Financial instruments:
|
The
Group classifies its financial assets to the following categories: financial assets at fair value through profit or loss, loans
and receivables and financial assets available for sale. The classification depends, among other things, on the purpose for which
the financial assets were purchased. The Group’s management determines the classification of financial assets upon initial
recognition.
|
|
a)
|
Financial
assets at fair value through profit or loss.
|
This
category includes financial assets held for trading. A financial asset is classified to this category if it is purchased primarily
for the purpose of selling in the short-term.
Instruments
included in this category are marketable securities which are denominated in NIS.
Changes
in financial assets at fair value through profit or loss are presented among “operating activities” as part of the
changes in working capital in the statement of cash flows.
Changes
in the fair values of financial assets at fair value through profit or loss are recorded to “other income, net” in
the statement of comprehensive loss (see Note 14).
The
fair value of marketable securities is based on their quoted price in an active market.
Loans
and receivables are non-derivative financial assets with fixed or determinable payments that are not quoted on an active market.
These assets are classified as current assets, except for maturities longer than 12 months after the date of the statement of
financial position which are classified as non-current assets. The Group’s loans and receivables are included in “accounts
receivable”, “short-term deposits” and “cash and cash equivalents” in the statement of financial position
(see also sections j and k below).
|
|
c)
|
Available for sale financial assets
|
Available
for sale financial assets are non-derivatives which are not classified in any of the other categories. They are classified as
non-current assets, unless management intends to dispose of them within a period of up to 12 months from the date of the statement
of financial position, in which case they are classified as current assets.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
2 - SIGNIFICANT ACCOUNTING POLICIES
(continued):
The
Group classifies its financial liabilities to the following categories: financial liabilities at fair value through profit or
loss and financial liabilities at amortized cost. The Group’s management determines the classification of financial liabilities
upon initial recognition.
|
|
a)
|
Financial liabilities at fair value through profit or
loss.
|
Warrants
allotted to investors with a cashless exercise mechanism. In accordance with International Accounting Standard 32: “Financial
Instruments: Presentation”, these warrants are a “financial liability”. As the aforementioned liability is a
non-equity derivative financial instrument, it is classified in accordance with IAS 39 as a financial liability at fair value
through profit or loss, which is measured at its fair value at each date of the statement of financial position, with changes
in the fair value carried to “profit from changes in fair value of warrants issued to investors” in the statement of
comprehensive loss.
|
|
b)
|
Financial liabilities at amortized cost
|
Trade
payables and financial liabilities included in “other liabilities” are recognized initially at fair value and subsequently
measured at amortized cost using the effective interest method.
|
|
2)
|
Recognition
and measurement
|
Regular
purchases and sales of financial assets are recorded at the date of the settlement which is the date on which the asset was delivered
to the Group or delivered from the Group.
Investments
are initially recognized at fair value plus transaction costs for all financial assets not carried at fair value through profit
or loss. Financial assets measured at fair value through profit or loss are initially recognized at fair value and transaction
costs are charged to income or loss. Financial assets are derecognized when the rights to receive cash flows from the investments
have expired or have been transferred and the Group has transferred substantially all risks and rewards of ownership associated
with these assets. Available for sale financial assets and financial assets at fair value through profit or loss are measured
in subsequent periods at fair value. Loans and receivables are subsequently carried at amortized cost using the effective interest
method.
Gains
or losses arising from the changes in the fair value of financial assets at fair value through profit or loss are presented in
the statement of comprehensive loss among “other income - net” in the period in which they were incurred.
Gains
or losses from the changes in fair value of available for sale financial assets are recognized in other comprehensive income,
except for impairment losses, and exchange rate gains and losses on available for sale financial assets which constitute monetary
assets, until the asset is written off.
When
a financial asset available for sale is sold or impaired, the accumulated gain or loss, which was previously recognized in other
comprehensive loss, is reclassified from equity to income or loss under “other income, net”.
As
to methods for measurement of the Company’s financial instruments, see note 4.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
2 - SIGNIFICANT ACCOUNTING POLICIES
(continued):
|
|
3)
|
Impairment of financial assets
|
The
Group assesses at each date of the statement of financial position whether there is objective evidence that a financial asset
or group of financial assets measured at depreciated cost or available for sale is impaired. A financial asset or a group of financial
assets is impaired and impairment losses are incurred only if there is objective evidence of impairment as a result of one or
more events that occurred after the initial recognition of the asset (a ‘loss event’) and that loss event (or events)
has an impact on the estimated future cash flows of the financial asset or group of financial assets that can be reliably estimated.
Inventory
is measured at the lower of cost or net realizable value.
The
cost is determined on the basis of “first in-first out” basis. Cost of purchased products and inventory in process
includes costs of design, raw materials, direct labor, other direct costs and fixed production overheads.
Net
realizable value is an estimated selling price in the ordinary course of business less applicable variable selling expenses.
The
balance of trade receivables includes amounts due from customers for products sold or services rendered in the ordinary course
of business. If collection is expected in one year or less, they are classified as current assets. If not, they are presented
as noncurrent assets.
Trade
receivables are recognized initially at fair value and subsequently measured at amortized cost using the effective interest method,
less provision for doubtful accounts.
|
|
k.
|
Cash and cash equivalents
|
The
consolidated statements of financial position and the consolidated statements of cash flows include cash and cash equivalents,
which are short-term bank deposits with maturities of three months or less.
|
|
l.
|
Current and deferred taxes
|
Tax
expenses for the reported years include current and deferred taxes. The taxes are recognized in the Statements of Loss and Other
Comprehensive Loss, except for taxes relating to items carried to other comprehensive income, which are recognized in other comprehensive
income.
The
amount that was recorded as current taxes, is calculated based on the tax laws that have been enacted or substantively enacted
at the date of the report, in countries in which the Company and its subsidiaries operate and generate taxable income. The Group’s
management periodically evaluates the tax implications applicable to the taxable income, in accordance with the relevant tax laws,
and creates provisions in accordance with the amounts expected to be paid to the tax authorities.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
2 - SIGNIFICANT ACCOUNTING POLICIES
(continued):
The
Group recognizes deferred taxes using the liability method, for temporary differences between the amounts of assets and liabilities
included in the financial statements, and the amounts for tax purposes. Deferred taxes are not recognized, if the temporary differences
arise at the initial recognition of the asset or liability which at the time of the transaction has no effect on profit or loss,
whether for accounting or tax reporting. The amount of deferred taxes is determined using the tax rates (and laws) that have been
enacted or substantially enacted by the date of the statement of financial position and are expected to apply when the related
deferred tax assets is realized or the deferred tax liabilities will be settled.
Deferred
tax assets are recognized for temporary differences that are tax deductible, up to the amount of the differences that are expected
to be utilized in the future, against taxable income.
No
deferred tax assets have been recorded in the Group’s books of accounts for current losses carried forward since it is not
probable that the Group will be able to utilize those losses in the foreseeable future against taxable income.
Deferred
tax assets and liabilities are offset only if:
|
|
-
|
There is a legally enforceable right to offset current
tax assets against current tax liabilities; and
|
|
|
|
|
|
|
-
|
Deferred income tax assets and liabilities relate to
income taxes imposed by the same taxation authority on the same taxable entity.
|
In
the event of a dividend distribution originating from tax exempted “benefited enterprises”, tax will be levied on
the amount distributed using the tax rate that would have been applicable to Company had it not been exempted from tax. In the
event of such a distribution, the amount of tax will be recognized as an expense in the statement of comprehensive loss.
|
|
1)
|
Retirement benefit obligation
|
A
defined contribution plan is a post-employment employee benefit plan, to which Group companies pay fixed deposits to a separate
and independent entity, so that the Group has no legal or constructive obligation to pay further contributions if the fund does
not hold sufficient assets to pay all employees the benefits relating to employee service in the current and prior periods.
According
to Israeli labor laws and work agreements, and as customary in the Group’s companies, the Group’s companies are obligated
to pay severance pay to employees who are fired or leave their employment in certain circumstances.
The
obligation of Group companies to the employees which is in the framework of a defined contribution plan, is to make fixed deposits
to a separate and independent entity, so that the Group has no obligation, legal or otherwise, to make additional deposits, when
assets of this fund are insufficient to pay to all of the employees their benefits for their current and past employment services.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
2 - SIGNIFICANT ACCOUNTING POLICIES
(continued):
|
|
2)
|
Vacation
and recreation pay
|
Under
the law each employee is legally entitled to vacation and recreation benefits, both calculated on an annual basis. The entitlement
is based on term of employment. The Group records the obligation and expense for vacation and recreation pay based on the benefits
that have been accumulated for each employee.
The
Company recognizes the obligation and expense for bonuses when a contractual or constructive obligation exists. The obligation
is recognized in the amount expected to be paid, to the extent that the Company can reliably estimate the amount expected to be
paid.
The
Group operates several equity-settled share based compensation plans to employees and other service providers, under which the
Group receives services from employees and service providers in return for equity instruments (options) of the Company. The fair
value of the services received from employees and service providers in return for granting the options is recognized as an expense
in the statement of comprehensive loss. The total amount charged as an expense is determined taking into consideration the fair
value of the options granted:
Without
considering service and performance conditions, which are non-market vesting conditions (e.g. meeting profit and sales targets
and continued employment in the Company for a certain period).
Non-market
vesting conditions are included among the assumptions used to estimate the number of options expected to vest. The total expense
is recognized during the vesting period, which is the period over which all of the specified vesting conditions of the share-based
payment are to be satisfied.
At
each date of the statement of financial position, the Group revises the estimates of the number of options that are expected to
vest, based on non-market vesting conditions. It recognizes the impact of the revision to original estimates, if any, in profit
or loss, with a corresponding adjustment to equity.
When
the options are exercised, the Company issues new shares. The proceeds, less directly related transaction costs, are reflected
in the share capital (at par value) and in share premium.
Group
revenues are measured in accordance with the fair value of the consideration received or receivable in respect of sales supplied
in the ordinary course of business. Revenues are presented net of V.A.T. returns, rebates and discounts.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
2 - SIGNIFICANT ACCOUNTING POLICIES
(continued):
Revenue
from the sale of goods is recognized when all of the following conditions are met:
|
|
●
|
The
Group transferred the significant risks and rewards of ownership of the goods to the
purchaser;
|
|
|
|
|
|
|
●
|
The
Group does not retain continuing managerial involvement to the degree usually associated
with ownership nor effective control over the goods sold;
|
|
|
|
|
|
|
●
|
The
amount of the revenues can be measured reliably. The amount of the revenue is not considered
as being reliably measured until all the conditions relating to the transaction are met.
The Group bases its estimates on past experience, considering the type of customer, type
of transaction and special details of each arrangement.
|
|
|
|
|
|
|
●
|
It
is probable that the economic benefits that are associated with the transaction will
flow to the Group; and
|
|
|
|
|
|
|
●
|
The
costs incurred or to be incurred in respect of the transaction can be measured reliably.
|
When
multiple-element arrangements exist, the amount of revenue allocated to each element is based upon the relative fair values of
the various elements. The fair value of each element is determined based on the current market price of each of the elements when
sold separately.
|
|
2.
|
Construction
contracts:
|
A
construction contract is defined by IAS 11, “Construction contracts”, as a contract specifically negotiated for the
construction of an asset.
When
the outcome of a construction contract can be estimated reliably and it is probable that the contract will be profitable, contract
revenue is recognized over the period of the contract by reference to the stage of completion. Contract costs are recognised as
expenses by reference to the stage of completion of the contract activity at the end of the reporting period. When it is probable
that total contract costs will exceed total contract revenue, the Group recognizes the expected loss as an expense immediately.
When
the outcome of a construction contract cannot be estimated reliably, the Group recognizes revenue only to the extent of contract
costs incurred that it is probable will be recoverable.
Variations
in contract work, claims and incentive payments are included in contract revenue to the extent that may have been agreed with
the customer and are capable of being reliably measured.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
2 - SIGNIFICANT ACCOUNTING POLICIES
(continued):
The
Group uses the “percentage-of-completion method” to determine the appropriate amount to recognize in a given period.
The stage of completion is measured by reference to the contract costs incurred up to the end of the reporting period as a percentage
of total estimated costs for each contract. Costs incurred in the year in connection with the future activities on a contract
are excluded from contract costs in determining the stage of completion.
On
the balance sheet, the Group reports the net contract position for each contract as either an asset or a liability. A contract
represents an asset where costs incurred plus recognized profits (less recognized losses) exceed progress billings; a contract
represents a liability when the opposite is the case.
Lease
agreements in which a significant portion of the risks and rewards of ownership are retained by the lessor are classified as operating
leases. Payments made in connection with operating leases are recognized in profit or loss using the straight-line basis over
the term of the lease.
As
a general rule, calculation of the basic loss per share is based on the loss that is attributed to the shareholders holding ordinary
shares, divided by the weighted average number of ordinary shares in issue during the period.
For
purposes of the calculation of the diluted loss per share, the Group adjusts the loss that is attributed to the holders of the
Company’s ordinary shares, and the weighted average number of ordinary shares in issue, to assume conversion of all of the
dilutive potential shares.
The
potential shares are taken into account only if their effect is dilutive (increases loss per share).
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
2 - SIGNIFICANT ACCOUNTING POLICIES
(continued):
|
|
r.
|
New
international financial reporting standards, amendments to standards and new interpretations:
|
Standards,
amendments and interpretations to existing standards which are not yet effective and have not been early adopted by the Group:
|
|
a)
|
International
Financial Reporting Standard 9 “Financial Instruments” (hereinafter –
“IFRS 9” or “the Standard”):
|
IFRS
9 deals with the classification, measurement and recognition of financial assets and financial liabilities. The full version of
IFRS 9 was published in July 2014. This Standard replaces the present existing directives in International Accounting Standard
39 “Financial Instruments: Recognition and Measurement” (hereinafter IAS 39) regarding the classification and measurement
of financial instruments. IFRS 9 leaves the measurement model connected with measuring financial assets, but simplifies it and
sets forth three main categories: amortized cost, fair value through other comprehensive income, and fair value through profit
or loss. The classification is based on the business model of the entity and on characteristics of the contractual cash flows
of the financial asset. Investments in capital instruments will be measured at fair value through profit or loss. Nevertheless,
the entity’s management can choose, on the date of initial recognition, irrevocably, to present the changes in fair value of an
investment in a capital instrument in other comprehensive income, without recycling them to profit or loss.
The
Standard presents a new model for an impairment of financial instruments, based on the Expected Credit Loss Model. This model
replaces the existing model in IAS 39, which is based on the Incurred Loss Model.
Regarding
classification and measurement of financial liabilities, there were no changes compared to existing model in IAS 39, excluding
the recognition of changes in the fair value of liabilities designated to the fair value through “profit or loss” category,
resulting from the entity’s own credit risk, in other comprehensive income.
The
Standard will be implemented retrospectively as from annual periods starting January 1, 2018. According to the provisions of IFRS
9 it may be implemented earlier. The Group is examining the expected effect of IFRS 9 on its financial statements.
|
|
b)
|
International
Financial Reporting Standard 15 “Revenues from Contracts with Customers” (hereinafter
– IFRS 15).
|
IFRS
15 will replace, on its first implementation, the directives on the subject of recognizing revenues existing today under International
Financial Reporting Standards.
The
core principle of IFRS 15 is that revenues from contracts with customers must be recognized in a way that reflects the transfer
of control of goods or services supplied to customers in the framework of the contracts by amounts which reflect the proceeds
that the entity expects that it will be entitled to receive for those goods or services.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
2 - SIGNIFICANT ACCOUNTING POLICIES
(continued):
IFRS
15 sets forth a single model for recognizing revenues, according to which the entity will recognize revenues according to the
said core principle by implementing five stages:
(1) Identifying
the contract(s) with the customer.
(2) Identifying
the separate performance obligations in the contract.
(3) Determining
the transaction price.
(4) Allocating
the transaction price to separate performance obligations in the contract.
(5) Recognizing
revenue when (or as) each of the performance obligations is satisfied.
The
Company is still assessing the impact of IFRS 15.
|
|
c)
|
International Financial Reporting Standard 16 “Leases”
(hereafter - IFRS 16)
|
IFRS16,
Leases
, which replaces the current guidance in IAS 17. IFRS 16 requires lessees, with certain exceptions, to recognize
a lease liability reflecting future lease payments and a ‘right-of-use asset’ for lease contracts. The standard is
effective for annual periods beginning on or after January 1, 2019, with earlier application permitted if IFRS 15,
Revenue
from Contracts with Customer
, is also applied. The Group is yet to assess IFRS 16’s full impact.
NOTE
3 - SIGNIFICANT JUDGMENTS:
Judgments
are continually evaluated and are based on historical experience and other factors, including expectations of future events that
are believed to be reasonable under the circumstances.
Development
costs are capitalized according to the accounting policies specified in Note 2e(2). Capitalization of those costs is based on
management’s judgment that technological and economic feasibility exist, a situation which exists for the most part when
a product development project reaches a milestone, or when the Company enters into agreement for the sale of know-how arising
from the development. In determining amounts to be capitalized, management makes assumptions in relation to the future cash flows
expected from the assets, discount rate to be used and expected benefit period.
To
date, the Group did not capitalize significant of development costs.
As
of December 31, 2015 Chief Scientist grants received by the Group and for which there is an obligation to pay royalties totaled
approximately – NIS 794 thousand (see also Note 11. Over all of the reporting periods presented, the Company has concluded
that, with respect to all of the grants received from the Chief Scientist, there is reasonable assurance that the grants received
will not be repaid, therefore the grants were recorded to income as reduction of research and development expenses (see also Note
2g).
Based
on management’s judgment, no deferred tax assets have been recorded in the Group’s books of accounts for current losses carried
forward for tax purposes since it is not probable that the Group will be able to utilize those losses in the foreseeable future
against taxable income as of December 31, 2015. Had the Group recorded a deferred tax asset for all of the accumulated losses
carried forward for tax purposes the accumulated deficit of the Group would have been decreased by approximately NIS 42 million.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
4 - FINANCIAL INSTRUMENTS AND FINANCIAL RISK MANAGEMENT:
Financial
risk management
:
|
|
1)
|
Financial
risk factors
|
The
Group’s activities expose it to a variety of financial risks: market risks (including currency risks, fair value interest
rate risk, cash flow interest rate risk and price risk), credit risks and liquidity risks. The Group’s overall risk management
plan focuses on the unpredictability of financial markets and seeks to minimize the potential adverse effects on the Group’s
financial performance.
Risk
management is performed by the finance department according to the policy authorized by the board of directors.
|
|
(1)
|
Foreign
exchange risk
|
The
Company operates internationally and is exposed to foreign exchange risks due to exposure to foreign currencies, primarily the
U.S. Dollar. Foreign exchange risk arises from future commercial transactions, assets or liabilities denominated in foreign currency.
The
Group’s policy to reduce the exposure to changes in exchange rates is based on maintaining, where possible, the balances
of current monetary assets, according to the currency of the current liabilities.
As
of December 31, 2015, if the functional currency of the Company had weakened/strengthened by 10% against the Dollar, with all
other variables held constant, the loss for the year would have been lower/higher by NIS 1,201 thousand (2014- NIS 2,184 thousand,
2013- NIS 749 thousand), primarily as a result of foreign exchange gains/losses on cash and cash equivalents.
The
Group had investments in financial instruments which were traded on the stock exchange, and were classified as financial assets
at fair value through profit or loss; the Company was exposed to fluctuations in the price of the security based on stock exchange
market prices.
At
December 31, 2015 the Group does not hold any of the said investments in financial instruments.
The
following table summarizes the changes in fair value of financial instruments which were sensitive to changes in the price of
traded securities as of December 31, 2014:
|
|
|
|
Profit
from changes
|
|
|
|
|
|
Loss
from changes
|
|
|
|
|
|
10%
increase in value
|
|
5%
increase in value
|
|
|
Fair
value December 31,
2014
|
|
|
5%
decrease in value
|
|
|
10%
decrease in value
|
|
|
|
|
|
NIS
in thousands
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Government
bonds
|
|
819
|
|
|
409
|
|
|
|
8,187
|
|
|
|
(409
|
)
|
|
|
(819
|
)
|
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
4 - FINANCIAL INSTRUMENTS AND FINANCIAL RISK MANAGEMENT
(continued):
Credit
risks are treated at the Group level. Credit risks arise from cash and cash equivalents, bank deposits and from credit exposures
in connection with outstanding receivables and committed transactions.
The
Group’s cash and cash equivalents and short-term deposits as of December 31, 2015 and 2014 are held with large established banks
with, at least, AA rating. The Group’s marketable securities were managed by a portfolio management company. As of December 31,
2014 the Group’s securities were comprised solely of short-term government debentures.
Most
of the Company’s sales are made to a limited number of customers. To reduce the credit risk, the Company generally receives as
advance payment a substantial portion of the consideration receivable from the relevant parties.
No
credit limits were exceeded during the reported periods and Group’s management does not expect any losses from non-performance
of these parties.
Cash
flow forecasting is performed by the Group’s finance department. The finance department monitors rolling forecasts of the Company’s
liquidity requirements to ensure that it has sufficient cash to meet operational needs, while maintaining sufficient headroom
on its undrawn committed borrowing facilities, so that the Group does not breach any of its credit facilities.
The
Group invests cash surpluses in interest bearing investments such as time deposits and short-term government debentures, choosing
instruments with appropriate maturities or sufficient liquidity to provide sufficient headroom as determined by the above-mentioned
forecasts.
As
of December 31, 2015 the Group had unutilized credit facilities of NIS 50 thousand.
Liquidity
risk arises from financial liabilities due to payable balances (except for institutions and advanced payments) and amounted to
NIS 4,980 thousand on December 31, 2015 (2014 - NIS 3,377 thousand).
These
liabilities are classified as current liabilities, and are expected to mature within 12 months from the date of the statement
of financial position.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
4 - FINANCIAL INSTRUMENTS AND FINANCIAL RISK MANAGEMENT
(continued):
|
|
2)
|
Estimates of fair value
|
The
following is an analysis of the financial instruments measured at fair value, according to valuation methods. The levels are defined
as follows:
|
|
●
|
Quoted
prices (unadjusted) in active markets for identical assets or liabilities (Level 1).
|
|
|
|
|
|
|
●
|
Inputs
for the asset or liability that are not based on observable market data (unobservable
inputs) (Level 3).
|
The
following table presents the group’s financial assets and liabilities that are measured at fair value December 31, 2015 and December
31, 2014:
|
|
|
|
|
December
31,
|
|
|
|
|
|
2015
|
|
|
|
2014
|
|
|
|
|
|
Level
3
|
|
|
|
Total
|
|
|
|
Level
1
|
|
|
|
Level
3
|
|
|
Total
|
|
|
|
|
|
NIS in thousands
|
|
|
Financial
assets at fair value through profit or loss -
financial
assets held for trading
|
|
|
|
|
|
|
|
|
|
|
8,187
|
|
|
|
|
|
|
8,187
|
|
|
Financial
liabilities at fair value through profit or loss -
Warrants
at fair value
|
|
|
34
|
|
|
|
34
|
|
|
|
|
|
|
|
428
|
|
|
428
|
|
|
a)
|
Financial instruments in level 1
|
The
fair value of financial instruments traded in active markets is based on quoted market prices at the date of the statement of
financial position. A market is considered to be active if the quoted prices are readily and regularly available from an exchange,
dealer, broker, industry group, pricing service or regulatory agency and those prices represent actual and regularly occurring
market transactions on an arm’s length basis.
The
quoted market price used for financial assets at fair value through profit or loss held by the Group is the current bid price
at the time of closing of trade.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
4 - FINANCIAL INSTRUMENTS AND FINANCIAL RISK MANAGEMENT
(continued):
|
|
b)
|
Financial instruments in level 3
|
The
Company’s financial liability at fair value through profit or loss is the obligation for warrants (Orbimed) (see Note 12(b)(1))
and warrants (series E) (see Note 12(b)(3)).
|
|
|
|
December
31,
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
standard
deviation
|
|
|
risk-free
interest
|
|
|
fair
value (NIS thousands)
|
|
|
standard
deviation
|
|
|
risk-free
interest
|
|
|
fair
value (NIS thousands)
|
|
|
|
Warrants
(Orbimed) (*)
|
|
|
45
|
%
|
|
|
0.001
|
%
|
|
|
-
|
|
|
|
39
|
%
|
|
|
0.35
|
%
|
|
|
1
|
|
|
|
Warrants
(Series E) (**)
|
|
|
48
|
%
|
|
|
0.4
|
%
|
|
|
34
|
|
|
|
47
|
%
|
|
|
0.79
|
%
|
|
|
427
|
|
(*)
In calculating the value of warrants (Orbimed) a binomial model was used. The risk-free interest rate between the periods is derived
from a curve of “Shachar” type Israel government bond for a period which corresponds to the term of the warrants at
the time of the calculation was made. The expected volatility is based on fluctuations in the price of the Company’s share.
On
March 3, 2016 the warrants (Orbimed) expired as the exercise period was ended.
(**)
In calculating the value of warrants (Series E) a Black and Scholes model was used. If the change in standard deviation for that
warrants shifted +/- 5%, the impact on profit or loss would be NIS 10 thousand. The higher the standard deviation, the higher
the fair value. The risk-free interest rate between the periods is derived from a curve of “Shachar” type Israel government
bond for a period which corresponds to the term of the warrants at the time of the calculation was made. The expected volatility
is based on fluctuations in the price of the Company’s share.
The
following table presents the changes in Level 3 instruments for the year ended December 31, 2015:
|
|
|
|
Warrants
|
|
|
|
|
|
NIS
thousands
|
|
|
|
Opening
balance at January 1
|
|
|
428
|
|
|
|
Gains
recognized in profit or loss
|
|
|
(394
|
)
|
|
|
Closing
balance at December 31
|
|
|
34
|
|
|
|
|
|
|
|
|
|
|
Total
unrealized profits for the period included in profit or loss for liabilities held at the end of the reporting period
|
|
|
394
|
|
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
4 - FINANCIAL INSTRUMENTS AND FINANCIAL RISK MANAGEMENT
(continued):
The
following table presents the changes in Level 3 instruments for the year ended December 31, 2014:
|
|
|
|
Warrants
|
|
|
|
Opening balance at
January 1
|
|
|
--
|
|
|
|
|
|
|
|
|
|
|
Issue of
warrants (see note 12(b)(1))
|
|
|
2,355
|
|
|
|
Transfer to Level 3
|
|
|
2,317
|
|
|
|
Gains
and losses recognized in profit or loss
|
|
|
(4,244
|
)
|
|
|
Closing
balance at December 31
|
|
|
428
|
|
|
|
|
|
|
|
|
|
|
Total
unrealized profits for the period included in profit or loss for liabilities held at the end of the reporting period
|
|
|
4,244
|
|
In
2014, the Group transferred warrants from level 2 to level 3 due to the fact that during the second quarter of 2014 the Company
commenced using a standard deviation parameter that was calculated based on historical share prices instead of current standard
deviation embedded in the market price of traded warrants.
NOTE
5 - CASH AND CASH EQUIVALENTS:
|
|
|
|
As of December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
NIS in thousands
|
|
|
|
Cash in banks
|
|
|
7,073
|
|
|
|
7,442
|
|
|
|
Short-term bank deposits
|
|
|
33,166
|
|
|
|
34,625
|
|
|
|
|
|
|
40,239
|
|
|
|
42,067
|
|
The
currencies in which the cash and cash equivalents are denominated or to which they are linked are as follows:
|
|
|
|
As of December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
NIS in thousands
|
|
|
|
NIS
|
|
|
26,625
|
|
|
|
18,809
|
|
|
|
US Dollar
|
|
|
13,372
|
|
|
|
22,965
|
|
|
|
Other currencies
|
|
|
242
|
|
|
|
293
|
|
|
|
|
|
|
40,239
|
|
|
|
42,067
|
|
The
carrying amount of the cash and cash equivalents approximates their fair value.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
6 - ACCOUNTS RECEIVABLE:
|
|
|
|
|
As of December 31, 2015
|
|
|
|
|
|
|
|
NIS
unlinked
|
|
|
|
Denominated
in US Dollars
|
|
|
|
Denominated
in Euro
|
|
|
Total
|
|
|
|
|
|
|
NIS in thousands
|
|
|
|
Open
accounts
|
|
|
|
|
|
|
754
|
|
|
|
206
|
|
|
960
|
|
|
|
Credit
cards
|
|
|
32
|
|
|
|
|
|
|
|
|
|
|
32
|
|
|
|
|
|
|
32
|
|
|
|
754
|
|
|
|
206
|
|
|
992
|
|
|
|
|
|
|
As of December 31, 2014
|
|
|
|
|
|
|
NIS
unlinked
|
|
|
|
Denominated
in US Dollars
|
|
|
|
Denominated
in Euro
|
|
|
Total
|
|
|
|
|
|
|
NIS in thousands
|
|
|
|
Open
accounts
|
|
|
|
|
|
|
301
|
|
|
|
180
|
|
|
481
|
|
|
|
Credit
cards
|
|
|
32
|
|
|
|
|
|
|
|
|
|
|
32
|
|
|
|
|
|
|
32
|
|
|
|
301
|
|
|
|
180
|
|
|
513
|
|
|
|
|
|
As of December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
NIS in thousands
|
|
|
|
|
|
|
|
|
|
|
|
|
Institutions
|
|
|
284
|
|
|
|
329
|
|
|
|
Prepaid expenses
|
|
|
3,163
|
*
|
|
|
1,276
|
*
|
|
|
Advances to suppliers
|
|
|
372
|
|
|
|
127
|
|
|
|
Receivables
|
|
|
105
|
|
|
|
|
|
|
|
Other
|
|
|
|
|
|
|
6
|
|
|
|
|
|
|
3,924
|
|
|
|
1,738
|
|
*
Including NIS 2,546 thousand (December 31, 2014: NIS 716 thousand) materials used to manufacture MUSE systems which will be used,
for testing, training, demonstrations and promotional activities. The cost of these materials will be recorded to research and
development expenses or selling and marketing expenses depending on actual use.
|
|
c
.
|
Balances included in respect of monetary items among
“trade and other receivables” do not include doubtful accounts.
|
The
carrying amounts of other receivables (except for the non-monetary balances from institutions, prepaid expenses, and advances
to suppliers) approximate their fair value.
The
maximum exposure to credit risks as of the date of the statement of financial position in respect of accounts receivable is the
carrying amount of all the aforementioned Group of receivables net of the non-monetary balances (institutions, prepaid expenses
and advances to suppliers), amounting to NIS 1,097 thousand (December 31, 2014 - NIS 519 thousand). The Group does not hold any
collateral in respect of these balances.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
7 - INVENTORY:
Composed
as follows:
|
|
|
|
As of December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
NIS in thousands
|
|
|
|
Raw materials and supplies
|
|
|
775
|
|
|
|
1,453
|
|
|
|
Work in progress
|
|
|
77
|
|
|
|
68
|
|
|
|
Finished products
|
|
|
335
|
|
|
|
423
|
|
|
|
|
|
|
1,187
|
|
|
|
1,944
|
|
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
8 - PROPERTY AND EQUIPMENT:
|
|
a.
|
Composition
of property and equipment and accumulated depreciation thereon, grouped by major classifications
and changes therein, and their movements during 2015:
|
|
|
|
|
|
Cost
|
|
|
|
Accumulated
Depreciation
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at
|
|
|
|
Additions
|
|
|
|
Balance
|
|
|
|
Balance
at
|
|
|
|
Additions
|
|
|
|
Balance
|
|
|
|
Depreciated
balance
|
|
|
|
|
|
|
beginning
|
|
|
|
during
|
|
|
|
at end of
|
|
|
|
beginning
|
|
|
|
during
|
|
|
|
at end of
|
|
|
|
December
31
|
|
|
|
|
|
|
of
year
|
|
|
|
the
year
|
|
|
|
year
|
|
|
|
of
year
|
|
|
|
the
year
|
|
|
|
year
|
|
|
|
2015
|
|
|
|
2014
|
|
|
|
Property
and equipment:
|
|
|
NIS in thousands
|
|
|
|
NIS in thousands
|
|
|
|
NIS in thousands
|
|
|
|
Machinery
and equipment
|
|
|
2,635
|
|
|
|
144
|
|
|
|
2,779
|
|
|
|
1,876
|
|
|
|
299
|
|
|
|
2,175
|
|
|
|
604
|
|
|
|
759
|
|
|
|
Leasehold
improvements
|
|
|
129
|
|
|
|
53
|
|
|
|
182
|
|
|
|
129
|
|
|
|
11
|
|
|
|
140
|
|
|
|
42
|
|
|
|
|
|
|
|
Office
furniture and equipment (including computers)
|
|
|
1,365
|
|
|
|
130
|
|
|
|
1,495
|
|
|
|
1,179
|
|
|
|
82
|
|
|
|
1,261
|
|
|
|
234
|
|
|
|
186
|
|
|
|
|
|
|
4,129
|
|
|
|
327
|
|
|
|
4,456
|
|
|
|
3,184
|
|
|
|
392
|
|
|
|
3,576
|
|
|
|
880
|
|
|
|
945
|
|
|
|
b.
|
Composition
of property and equipment and accumulated depreciation thereon, grouped by major classifications
and changes therein, and their movements during 2014:
|
|
|
|
|
Cost
|
|
|
Accumulated
Depreciation
|
|
|
Depreciated
balance
|
|
|
|
|
|
Balance at
beginning
of year
|
|
|
Additions
during the year
|
|
|
Balance at
end of
year
|
|
|
Balance at
beginning of year
|
|
|
Additions
during the year
|
|
|
Balance at
end of
year
|
|
|
|
|
|
|
Property
and equipment:
|
|
NIS
in thousands
|
|
|
NIS
in thousands
|
|
|
NIS
in thousands
|
|
|
|
Machinery
and equipment
|
|
|
2,492
|
|
|
|
143
|
|
|
|
2,635
|
|
|
|
1,530
|
|
|
|
346
|
|
|
|
1,876
|
|
|
|
759
|
|
|
|
Leasehold
improvements
|
|
|
92
|
|
|
|
37
|
|
|
|
129
|
|
|
|
92
|
|
|
|
37
|
|
|
|
129
|
|
|
|
|
|
|
|
Office
furniture and equipment (including computers)
|
|
|
1,305
|
|
|
|
60
|
|
|
|
1,365
|
|
|
|
1,114
|
|
|
|
65
|
|
|
|
1,179
|
|
|
|
186
|
|
|
|
|
|
|
3,889
|
|
|
|
240
|
|
|
|
4,129
|
|
|
|
2,736
|
|
|
|
448
|
|
|
|
3,184
|
|
|
|
945
|
|
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
9 - TAXES ON INCOME:
|
|
a.
|
Corporate taxation in Israel:
|
|
|
1)
|
Measurement
of results for tax purposes:
|
Commencing
with tax year 2008 the results of the Company for tax purposes are measured in nominal values. Through the end of tax year
2007 the results of the Company for tax purposes were measured having regard to the changes in the Israeli consumer price
index (“CPI”), in accordance with the Income Tax Law (Inflationary Adjustments), 1985 (here in after-the the
“Inflationary Adjustments Law”). The transitional provisions regarding the discontinuation of the application of
the Inflationary Adjustments Law stipulate that losses carried forward for tax purposes, deduction for depreciation, and real
loss from sale of a depreciable asset or security will be linked to the CPI until the end of tax year 2007 and linkage shall
be discontinued as of this date.
The
income of the Company (other than income which is eligible for reduced tax rates in accordance with encouragement laws in Israel,
see C below) is subject to corporate tax at the regular corporate tax rates.
On
December 6, 2011, the Law for the Amendment of the Tax Burden (Legislative Amendments), 2011 was published in the official gazette.
Under this law, a previously approved gradual decrease in corporate tax was discontinued and the corporate tax rate was increased
to 25% beginning with 2012.
On
August 5, 2013, the Law for the Amendment of National Priorities (Legislative Amendments for Achieving the Budgetary Goals for
2013-2014), 2013 (hereinafter - the Law) was published in the official gazette. The Law stipulated, among other things, that the
corporate tax rate should be increased to 26.5% (instead of 25%) for 2014 and thereafter. (As to the increase of tax rates on
the income of a Preferred Enterprise as set out in the Law for the Encouragement of Capital Investments-1959, see C below).
On
January 4, 2016 the Israeli Parliament approved an amendment for the Israeli tax Ordinance (Number 216), 2016 according to which
corporate tax rate will be updated to 25% commencing the tax year 2016.
The
Company’s capital gains are subject to tax at the regular corporate tax rates.
|
|
b.
|
Taxation of a subsidiary outside Israel
|
The
Subsidiary incorporated in the U.S. is assessed according to U.S. tax law.
|
|
c.
|
Encouragement laws in Israel
:
|
Tax
benefits under the Law for the Encouragement of Capital Investments-1959 (hereinafter- the “Law for the Encouragement of
Capital Investments”):
Under
the Law for the Encouragement of Capital Investments, companies are entitled to various tax benefits by virtue of their “approved
enterprise” or “benefited enterprise” status subject to the fulfillment of certain conditions. In addition, companies
may be entitled to additional tax benefits as “foreign investors’ companies,” as defined by the Law for the Encouragement
of Capital Investments.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
9 - TAXES ON INCOME
(continued):
The
Law for Encouragement of Capital Investments was amended as part of the Economic Policy Law for the years 2011 and 2012 (Legislative
Amendments), 2011, which was passed in the Knesset on December 29, 2010 (hereinafter – the amendment). The amendment became
effective as from January 1, 2011.
The
amendement sets alternative benefit tracks to the ones that were in place under the provisions of the Law for the Encouragement
of Capital Investments, as follows: investment grants track designed for enterprises located in national development zone A and
two new tax benefits tracks (preferred enterprise and a special preferred enterprise), which provide for application of a unified
tax rate to all preferred income of the company, as defined in the law.
Under
the amended law, a company which qualifies for benefits under the encouragement law prior to the amendment thereof may opt for
application of the amendment on each year, commencing with the first year in which the amendment became effective (2011) thereby
making available to itself the tax benefits in accordance with the tracks set in the amendment subject to the fulfillment of certain
conditions. A company’s election for application of the amendment is irrevocable and once it opts for application thereof, it
will no longer be entitled to the tax benefits available to it under the pre-amendment regime of the Law for the Encouragement
of Capital Investments. A company will be allowed to continue and enjoy the tax benefits available under the law prior to its
amendment until the end of the period of benefits, as defined in the law.
The
Company has not decided at this stage whether and when to elect the application of the amendment of the law. Once the Company
generates taxable income, it is currently scheduled to be eligible for tax benefits available under the Law for the Encouragement
of Capital Investments before it was amended in accordance with the provisions of the benefited enterprise regime, as follows:
Reduced
tax rates
During
the period of benefits - 10 years commencing in the first year in which the Company earns taxable income from the benefited enterprises
(provided the maximum period to which it is restricted by law has not elapsed) - the income from the benefited enterprises owned
by the Company is tax exempt so long as it is not distributed or deemed to be distributed. The portion of income which qualifies
for tax exemption as above is based on the ratio between the turnover relating to the “benefited enterprise” and the
total turnover of the Company.
In
the event of a dividend distribution or deemed dividend distribution from income which was previously exempt, the Company will
be subject to tax on the grossed-up amount of the (deemed) dividend, according to the tax rate which would have applied to the
income were it not eligible for the exemption.
The
Company has not yet utilized the tax benefits for the main plant, nor for the expansion of the plant.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
9 - TAXES ON INCOME
(continued):
|
|
c)
|
Conditions to receive the benefits
|
The
entitlement to the above benefits is conditional upon the Company’s fulfillment of the conditions stipulated by the Law for the
Encouragement of Capital Investments, and the regulations promulgated thereunder. In the event of failure to comply with these
conditions, the benefits may be cancelled and the Company may be required to refund the amount of the benefits, in whole or in
part, with the addition of interest. As of the date of approval of these financial statements, the Company has met the aforementioned
conditions.
|
|
d.
|
Tax losses carried forward to future years
|
Carry
forward losses aggregate NIS 168 million and NIS 133 million as of December 31, 2015 and 2014, respectively. The Company did not
record deferred taxes for these losses, as the utilization thereof is not expected in the foreseeable future.
|
|
e.
|
Tax advances for certain employee benefits expenses
|
Tax
advances for certain employee benefits expenses that are not deductable for tax purposes paid to the tax authorities are charged
to income on a current basis; the Company does not record these advances as a tax asset in its books of accounts, as utilization
thereof is not expected in the foreseeable future.
The
total said tax advances paid which have not yet been offset is approximately NIS 163 thousand as of December 31, 2015 (December
31, 2014- NIS 137 thousand).
|
|
f.
|
Taxes
on income included in the Statements of Loss and Other Comprehensive Loss for the periods
presented:
|
The
following is reconciliation between the “theoretical” tax, which would apply to the Company if all of its income were
taxed at the regular rate applicable to the Company in Israel (see a2 above) and the amount of tax reflected in the Statements
of Loss for the reported year:
|
|
|
|
2015
|
|
|
2014
|
|
|
2013
|
|
|
|
|
|
NIS
in thousands
|
|
|
|
Loss
before taxes on income as reported in the Statements of Loss and Other Comprehensive Loss
|
|
|
(36,741
|
)
|
|
|
(22,616
|
)
|
|
|
(5,104
|
)
|
|
|
Theoretical
tax savings
|
|
|
(9,736
|
)
|
|
|
(5,993
|
)
|
|
|
(1,276
|
)
|
|
|
Increase
in taxes arising from tax losses created in the reported year for which deferred taxes were not recorded
|
|
|
10,000
|
|
|
|
6,006
|
|
|
|
1,361
|
|
|
|
Taxes
on income reported in the Statement of Loss and Other Comprehensive Loss
|
|
|
264
|
|
|
|
13
|
|
|
|
85
|
|
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
9 - TAXES ON INCOME
(continued):
The
Company has not received final tax assessments from the date it commenced its operations (January 1, 2000).
On
November 19, 2015, the Knesset approved the draft Law for Economic Efficiency (Legislative Amendments to Achieve Budget change
Budget 2015 and 2016), nine years -2015, which is an amendment to the Income Tax Ordinance, extending the period of limitation
of tax assessments submitted by taxpayers, regarding tax assessments in respect of the tax year 2013 onwards, for a period of
four years from the end of the tax year in which the assessment was submitted, instead of three years.
Accordingly,
by law, tax assessments filed by the Company in 2011 are considered final (subject to the submission date of reports and period
of limitation under the law).
The
Company is registered as an Authorized Dealer for Value Added Tax purposes.
NOTE
10 - ACCOUNTS PAYABLE AND ACCRUALS:
|
|
a
.
|
Trade payables are denominated in the following currencies:
|
|
|
|
|
As of December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
NIS in thousands
|
|
|
|
NIS unlinked
|
|
|
716
|
|
|
|
387
|
|
|
|
US Dollar
|
|
|
679
|
|
|
|
184
|
|
|
|
Euro
|
|
|
|
|
|
|
220
|
|
|
|
|
|
|
1,395
|
|
|
|
791
|
|
|
|
|
|
As of December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
NIS in thousands
|
|
|
|
Payroll and related expenses
|
|
|
1,431
|
|
|
|
619
|
|
|
|
Wages and fees of related parties and related expenses
|
|
|
639
|
|
|
|
722
|
|
|
|
Institutions
|
|
|
716
|
|
|
|
362
|
|
|
|
Provision for vacations and recreation pay
|
|
|
710
|
|
|
|
551
|
|
|
|
Advanced payments from customers
|
|
|
1,535
|
|
|
|
275
|
|
|
|
Accrued expenses
|
|
|
805
|
|
|
|
694
|
|
|
|
|
|
|
5,836
|
|
|
|
3,223
|
|
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
10 - ACCOUNTS PAYABLE AND ACCRUALS
(continued)
:
Other
payable balances (except for institutions and advances from customers) are denominated in the following currencies:
|
|
|
|
As of December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
NIS in thousands
|
|
|
|
|
|
|
|
|
|
|
|
|
NIS unlinked
|
|
|
2,017
|
|
|
|
1,335
|
|
|
|
US Dollar
|
|
|
1,439
|
|
|
|
1,241
|
|
|
|
Euro
|
|
|
129
|
|
|
|
10
|
|
|
|
|
|
|
3,585
|
|
|
|
2,586
|
|
The
balances of the financial instruments included within the trade payables and other payables approximate their fair value as the
effect of the discounting is immaterial.
NOTE
11 - COMMITMENTS:
|
|
a.
|
Development
program supported by the Chief Scientist:
|
|
|
1)
|
In
2007 the management of the Generic Technological Research and Development Department
in the Israeli Ministry of Industry and Commerce (hereinafter “MAGNET”) approved
the activities of the consortium of bio-medical photonics (hereinafter- the “Consortium”).
The Company was one of the participants of the Consortium.
|
The
Consortium was active from June 2007 until the end of 2012. The consortium was dissolved at the end of 2013.
There
is no requirement to repay the grants or pay royalties thereof.
MAGNET
grants recognized by the Company during 2013 that were offset against research and development expenses amounted to NIS 181 thousand
(see Note 13).
|
|
2)
|
On July 12, 2011 the Office of the Chief Scientist of
the Ministry of Industry, Trade and Labor informed the Company that it resolved to approve the Company’s application for
support in a joint project for the development of an innovative small-diameter endoscope used in dental surgery (hereinafter-
the “Project”).
|
The
Project was carried out during two years from August 2011 until the end of July 2013.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
11 - COMMITMENTS
(continued):
Chief
Scientist grants that the Company recognized during 2013 that were offset against research and development expenses amounted to
NIS 273 thousand (see Note 13).
In
accordance with provisions of the Encouragement of Industrial Research and Development Law - 1984, the Company is required to
pay royalties at the rate of 3% to 5% on all of its revenues from the product, up to 100% of the amount of the grant received
(approximately NIS 794 thousand) by the Company with the addition of annual interest and linkage (see also note 3 regarding of
Government Grants).
Since
the commencement of the Project and as of the date of approval of these financial statements, no royalties’ payments were required.
In
the event of failure to comply with the terms attached to the receipt of the grants, the Company may be required to refund the
amount of the grants, in whole or in part, with interest and linkage differences from the date of receipt. In the opinion of Company’s
management, as of the date of approval of these financial statements, the Company meets all of the said terms.
The
Group has lease agreements for buildings it uses. These agreements expire during 2017.
The
annual lease fees are linked to the CPI and total approximately NIS 346 thousand as of December 31, 2015.
MEDIGUS LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
12 - EQUITY:
|
|
|
|
Number of shares
|
|
|
Amount
|
|
|
|
|
|
Authorized
|
|
|
Issued and paid
|
|
|
Authorized
|
|
|
Issued and paid
|
|
|
|
|
|
December 31
|
|
|
December 31
|
|
|
December 31
|
|
|
December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
2015
|
|
|
2014
|
|
|
2015
|
|
|
2014
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
In thousands
|
|
|
NIS in thousands
|
|
|
|
Ordinary shares of NIS 0.1 par value*
|
|
|
150,000
|
|
|
|
50,000
|
|
|
|
32,047
|
|
|
|
24,995
|
|
|
|
15,000
|
|
|
|
5,000
|
|
|
|
3,204
|
|
|
|
2,499
|
|
*
Traded on the Tel Aviv Stock exchange at NIS 1.753 per ordinary share of NIS 0.1 par value on December 31, 2015.
|
|
2)
|
The
ordinary shares confer upon their holders voting rights and the right to participate
in shareholders’ meetings, the right to receive dividends and the right to participate
in surplus assets in the event of liquidation of the Company.
|
|
|
b.
|
Share
offering
to the public and existing shareholders:
|
|
|
1)
|
On March 3, 2013 the Company allotted to Orbimed Israel Partners Limited
Partnership (hereinafter-“Orbimed”) 3,994,547 ordinary shares (39,945,474 ordinary shares before reverse split) of
the Company and 39,945,474 warrants to purchase 3,994,547 additional ordinary shares (hereinafter- the “Warrants”).
Each 10 warrants is exercisable into 1 share. The Warrants are exercisable in return for payment of the exercise price of NIS
9.17 per share during the 18 months following the allotment of the Warrants, and NIS 11.004 per share as from the end of the said
18 months until the end of 36 months following the allotment of the Warrants. The Warrants can also be exercised using a cashless
exercise mechanism, in which the number of shares arising from the exercise of the warrant would be reduced by a number of shares,
the value of which equals the cash exercise price. The Warrants would be adjusted in respect of certain events as set out in the
agreement (e.g. dividend, distribution of bonus shares, etc.).
|
In
accordance with International Accounting Standard 32: “Financial Instruments: Presentation”, these Warrants are a
“financial liability”, which is classified in the statement of financial position as a non-current liability among
“warrants at fair value”.
The
immediate proceeds (gross) from the allotment of all the securities offered amounted to NIS 29,664 thousand (net 29,502 thousand
NIS). A total of NIS 13,222 thousand was attributed to the Warrants (Orbimed) representing the fair value thereof on that date
and a total of NIS 16,280 thousand (net of issuance expenses), was allocated to the allotted shares.
As
to the fair value of the said warrants as of December 31, 2015 and 2014 see note 4(2)(b).
On
March 3
, 2016 warrants (Orbimed)
expired as the exercise period was ended.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
12 – EQUITY
(continued)
:
|
|
2)
|
On October 17, 2013 the Company issued a shelf offering report in accordance
with a shelf prospectus of the Company dated August 28, 2013.
|
In
accordance with results of the offering to the public the Company allotted 344,936 units at the price of NIS 72 per unit. Each
unit was comprised of 10 ordinary shares (100 ordinary shares before reverse split) and 50 warrants (Series 8).
Each
10 warrants (Series 8) is exercisable into 1 share in consideration for an exercise price of NIS 10.8 until October 17, 2016.
The
immediate proceeds (gross) from the allotment of all securities offered amounted to NIS 24,835 thousand. Net proceeds from the
issuance, net of cash issuance expenses, amounted to NIS 24,326 thousand and were attributed to shares (a total of NIS 22,655
thousand) and warrants (a total of NIS 1,671 thousand) in accordance with their relative fair values.
|
|
3)
|
On August 21, 2014 and August 26, 2014, the Company allotted in a private
issue, a total of 8,527,154 ordinary shares (85,271,536 ordinary shares after reverse split) of the Company, and also a total
of 34,108,614 warrants (Series E) for the purchase of an additional 3,410,861 shares for total cash consideration of approximately
NIS 39 million. Each 10 warrants (Series E) is exercisable into 1 ordinary share of the Company at an exercise price of NIS 6.27
per share during the 36 months following the allotment.
|
21,847,610
warrants (Series E) of the warrants which were issued may, under certain circumstances, also be exercised via a cashless exercise
mechanism, whereby the number of shares the value of which equals the exercise premium in cash will be deducted from the number
of shares to be issued upon exercise of the warrant. In addition, the number of warrants outstanding will be adjusted to certain
events specified in the warrant agreement (such as: dividends, distribution bonus shares, etc.)
In
accordance with International Accounting Standard 32: “Financial Instruments: Presentation”, these warrants are a
“financial liability”, which was classified in the statement of financial position as a non-current liability among
“warrants at fair value”.
The
remainder of the warrants which do not have a cashless exercise mechanism were classified as an equity instrument.
Of
the securities issued 766,311 shares (7,663,109 shares before reverse split) and 3,065,244 warrants (Series E) were issued to
OrbiMed Israel Partners Limited Partnership, which is one of the controlling shareholders of the Company.
Net
proceeds from the issuance, net of cash issuance expenses, amounted to approximately NIS 36 million.
The
fair value of 12,260,974 warrants which were allotted on August 21, 2014 was approximately NIS 1,289 thousand. Calculation of
fair value was made based on the following assumptions: quoted share price on August 21, 2014 of NIS 4.34, standard deviation
of 51.8%, risk-free interest rate of 1.06%, no dividend expectation, and expected period to exercise of 3 years.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
12 – EQUITY
(continued)
:
The
fair value of 21,847,640 warrants which were allotted on August 26, 2014 was approximately NIS 2,429 thousand. Calculation of
fair value was made based on the following assumptions: quoted share price on August 26, 2014 of NIS 4.46, standard deviation
of 51.8%, risk-free interest rate of 0.96%, no dividend expectation, and expected period to exercise of 3 years.
As
of December 31, 2015 a liability in the amount of NIS 34 thousand (December 31, 2014 - NIS 427 thousand) was presented in respect
of the aforementioned warrants, representing their fair value on that date. The decrease in the fair value of the warrants over
the period from the allotment date until the date of the statement of financial position was recorded in the Statements of Loss
and Other Comprehensive Loss among “Profit from change in fair value of warrants issued to investors”. For further
details of the measurement and classification of warrants, see Note 4.
The
gross proceeds were attributed as follows: first to the fair value of the warrants classified as financial liabilities at fair
value through profit or loss and the remainder was attributed between shares and warrants classified as equity in accordance with
their relative fair values.
Issuance
expenses were attributed to equity and liability in proportion with the allocation of the proceeds, as detailed below:
|
|
|
|
EQUITY
|
|
|
LIABILITIES
|
|
|
|
|
|
|
|
|
Ordinary share capital and share premium
|
|
|
Warrants
|
|
|
Warrants
|
|
|
Total
|
|
|
|
|
|
NIS in thousands
|
|
|
|
Proceeds gross
|
|
|
35,288
|
|
|
|
1,258
|
|
|
|
2,355
|
|
|
|
38,901
|
|
|
|
Issuance expenses
|
|
|
(2,830
|
)
|
|
|
(101
|
)
|
|
|
*(189
|
)
|
|
|
(3,120
|
)
|
|
|
Proceeds net
|
|
|
32,458
|
|
|
|
1,157
|
|
|
|
2,166
|
|
|
|
35,781
|
|
*
The issuance expenses in the amount of NIS 189 thousand which were attributed to the warrants classified as a financial liability
were charged directly to profit or loss.
|
|
4)
|
On July 8, 2015, the Company issued a shelf offering report in Israel
in accordance with a shelf prospectus of the Company dated August 28, 2013.
|
In
accordance with results of the offering to the public the Company issued 70,525 units at the price of NIS 380 per unit. Each unit
was comprised of 100 ordinary shares (1,000 ordinary shares before reverse split) and 500 warrants (Series 9).
Each
10 warrants (Series 9) is exercisable into 1 share in consideration for an exercise price of NIS 5.32 until July 8, 2018.
The
immediate proceeds (gross) from the issuance of all securities offered amounted to approximately NIS 26.8 million.
Net
proceeds from the issuance, net of issuance expenses, amounted to approximately NIS 25.5 million of which a total of NIS 22.7
million were allocated to shares, and a total of NIS 2.8 million to warrants (Series 9) in accordance to the fair value ratios
of the instruments.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
12 – EQUITY
(continued)
:
|
|
1)
|
The
following are the grants of options to employees and other service providers:
|
|
|
Date
of grant
|
|
Plan
|
|
Number
of options granted
|
|
|
exercise
price per option (NIS)
|
|
|
Fair
value on grant date-NIS in thousands
|
|
|
Number
of options outstanding- December 31,
2015
|
|
|
Number
of options exercisable at
31 December 2015
|
|
|
Expiration
date
|
|
|
February
2010
|
|
Series
6
|
|
|
(*)
790,000
|
|
|
|
(**)2.26
|
|
|
|
491
|
|
|
|
90,000
|
|
|
|
90,000
|
|
|
February
8, 2016
|
|
|
February
2010
|
|
Series
6
|
|
|
970,000
|
|
|
|
(**)2.26
|
|
|
|
538
|
|
|
|
480,000
|
|
|
|
480,000
|
|
|
February
8, 2016
|
|
|
September
2011
|
|
Series
A
|
|
|
(*)
585,000
|
|
|
|
(**)0.91
|
|
|
|
279
|
|
|
|
125,000
|
|
|
|
125,000
|
|
|
November
2, 2017
|
|
|
November
2011
|
|
Series
A
|
|
|
1,420,000
|
|
|
|
(**)0.91
|
|
|
|
536
|
|
|
|
935,000
|
|
|
|
935,000
|
|
|
November
2, 2017
|
|
|
April
2012
|
|
Series
A
|
|
|
230,000
|
|
|
|
(**)0.91
|
|
|
|
117
|
|
|
|
200,000
|
|
|
|
150,000
|
|
|
April
23, 2018
|
|
|
April
2012
|
|
Series
B
|
|
|
1,150,000
|
|
|
|
(**)0.68
|
|
|
|
659
|
|
|
|
820,000
|
|
|
|
820,000
|
|
|
April
23, 2018
|
|
|
October
2013
|
|
Series
C
|
|
|
(***)4,500,000
|
|
|
|
(**)0.83
|
|
|
|
1,357
|
|
|
|
3,850,000
|
|
|
|
2,649,999
|
|
|
September
30, 2018
|
|
|
July
2014
|
|
Series
D
|
|
|
(*)310,000
|
|
|
|
(**)0.537
|
|
|
|
56
|
|
|
|
310,000
|
|
|
|
77,500
|
|
|
July
17, 2020
|
|
|
July
2014
|
|
Series
D
|
|
|
3,070,000
|
|
|
|
(**)0.537
|
|
|
|
554
|
|
|
|
2,477,500
|
|
|
|
707,500
|
|
|
July
17, 2020
|
|
|
December
2015
|
|
Series
F
|
|
|
(*)157,500
|
|
|
|
2.05
|
|
|
|
116
|
|
|
|
157,500
|
|
|
|
0
|
|
|
December
29, 2021
|
|
|
December
2015
|
|
Series
F
|
|
|
664,800
|
|
|
|
2.05
|
|
|
|
491
|
|
|
|
664,800
|
|
|
|
0
|
|
|
December
29, 2021
|
|
|
December
2015
|
|
Series
G
|
|
|
350,000
|
|
|
|
2.05
|
|
|
|
259
|
|
|
|
350,000
|
|
|
|
0
|
|
|
December
29, 2021
|
|
|
Total
|
|
|
|
|
14,197,300
|
|
|
|
|
|
|
|
|
|
|
|
10,459,800
|
|
|
|
6,034,999
|
|
|
|
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
12 - EQUITY
(continued)
:
|
|
c.
|
Share
based payments:
(Continued)
|
|
|
(*)
|
Granted to related parties.
|
|
|
|
|
|
|
(**)
|
Linked to the CPI as set out in the option allotment plan.
|
|
|
|
|
|
|
(***)
|
The board of directors meeting dated August 12, 2013, and the general
meeting of the Company's shareholders dated September 29, 2013 authorized the allotment of 4,500,000 options (Series C), to the
CEO of the Company. The options (Series C) are subject to the conditions of the option allotment plan, for the allotment of non-marketable
options to officers, employees and advisors of the Group. Each 10 options is exercisable into one ordinary Company share of NIS
0.1 par value at the exercise price of NIS 8.3.
|
The
right to exercise options is vested as follows:
|
|
(1)
|
3,200,000
options will vest in 24 equal monthly batches, on the first day of each month, for a
period of 24 months, commencing one year following the date employment started according
to the agreement, i.e., commencing October 1, 2014.
|
|
|
(2)
|
650,000
options will vest on June 30, 2014, provided that a target is met which was set by the
Company’s board of directors and which is related to clinical activities of the
MUSE system in the USA through this date. These options were vested as of June 30, 2014.
|
|
|
(3)
|
650,000
options will vest on February 1, 2015 provided that a sales revenues target is met in
2014, as set by the Company’s board of directors. In accordance with the decision
of the Company's board, these options expired on March 31, 2015 due to failure to meet
the target.
|
Vesting
conditions of all of the above options, except for the 1,300,000 options (Series C) as detailed in (2) and (3) above are service
conditions.
Each
10 options (Series 6, Series A, Series B, Series C and Series D) is exercisable into 1 ordinary share.
Each
1 option (Series F and Series G) is exercisable into 1 ordinary share.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
12 - EQUITY
(continued)
:
|
|
c.
|
Share
based payments:
(continued):
|
The
fair value of all of the options was calculated using the Black and Scholes options pricing model, and based on the following
assumptions:
|
|
Date
of grant
|
|
|
Fair
value on grant date-NIS in thousands
|
|
|
Share
price on date of grant
|
|
|
Expected
dividend
|
|
Expected
volatility
|
|
|
Risk
free interest
|
|
|
Vesting
conditions
|
|
Expected
term
|
|
|
October
2013
|
|
|
|
1,357
|
|
|
|
0.748
|
|
|
None
|
|
|
46
|
%
|
|
|
2.53
|
%
|
|
See
above
|
|
5
years
|
|
|
July
2014
|
|
|
|
610
|
|
|
|
0.482
|
|
|
None
|
|
|
40
|
%
|
|
|
1.90
|
%
|
|
four
equal batches, following one, two, three and four years from their grant date
|
|
6
years
|
|
|
December
2015
|
|
|
|
607
|
|
|
|
1.627
|
|
|
None
|
|
|
54
|
%
|
|
|
1.39
|
%
|
|
four
equal batches, following one, two, three and four years from their grant date
|
|
6
years
|
|
|
December
2015
|
|
|
|
259
|
|
|
|
1.627
|
|
|
None
|
|
|
54
|
%
|
|
|
1.39
|
%
|
|
will
vest in 36 equal monthly equal batches commencing January 29, 2016.
|
|
6
years
|
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
12 - EQUITY
(continued)
:
|
|
c.
|
Share
based payments:
(continued):
|
|
|
1
)
|
The
changes in the number of share options and the weighted averages of their exercise prices
are as follows:
|
|
|
|
|
For the year ended December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
2013
|
|
|
|
|
|
Number of options
|
|
|
Weighted average of exercise price per 1 ordinary share-(NIS)
|
|
|
Number of options
|
|
|
Weighted average of exercise price per 1 ordinary share-(NIS)
|
|
|
Number of options
|
|
|
Weighted average of exercise price per 1 ordinary share-(NIS)
|
|
|
|
Outstanding at the beginning of year
|
|
|
11,110,000
|
|
|
|
8.4
|
|
|
|
9,150,000
|
|
|
|
10.7
|
|
|
|
6,683,000
|
|
|
|
14.6
|
|
|
|
Granted
|
|
|
1,172,300
|
|
|
|
2.05
|
|
|
|
3,380,000
|
|
|
|
5.37
|
|
|
|
4,500,000
|
|
|
|
8.3
|
|
|
|
Forfeited
|
|
|
(485,833
|
)
|
|
|
15.6
|
|
|
|
(644,667
|
)
|
|
|
16.1
|
|
|
|
(622,083
|
)
|
|
|
17.0
|
|
|
|
Expired
|
|
|
(1,336,667
|
)
|
|
|
6.98
|
|
|
|
(775,333
|
)
|
|
|
15.9
|
|
|
|
(1,410,917
|
)
|
|
|
18.3
|
|
|
|
Outstanding at year end
|
|
|
10,459,800
|
|
|
|
7.58
|
|
|
|
11,110,000
|
|
|
|
8.4
|
|
|
|
9,150,000
|
|
|
|
10.7
|
|
|
|
Exercisable at year end
|
|
|
6,034,999
|
|
|
|
9.23
|
|
|
|
3,547,498
|
|
|
|
11.6
|
|
|
|
2,683,334
|
|
|
|
15.3
|
|
|
|
2)
|
The
amounts of expenses that were recorded for options to employees and other service providers
in the reported years are NIS 450 thousand, NIS 1,111 thousand and NIS 511 thousand for
the years ended December 31, 2015, 2014 and 2013, respectively.
|
|
|
3)
|
The
plans are intended to be governed by the terms stipulated by Section 102 to the Israeli
Income Tax Ordinance (except for the options to controlling shareholders).
|
In
accordance with these general rules and the track chosen by the Company pursuant to the terms thereof, in respect of options granted
to employees under the option allotment plan, the Company is not allowed to claim as an expense for tax purposes the amounts credited
to employees as a benefit, including amounts recorded as salary benefits in the Company's books, with the exception of the salary-benefit
component, if exists, determined on the grant date.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
13 -EXPENSES BY NATURE:
|
|
|
For the year ended
December 31
|
|
|
|
|
2015
|
|
|
2014
|
|
|
2013
|
|
|
|
|
NIS in thousands
|
|
|
Payroll and related expenses
|
|
|
17,749
|
|
|
|
14,429
|
|
|
|
8,321
|
|
|
Materials used and subcontracted work
|
|
|
8,226
|
|
|
|
5,459
|
|
|
|
3,282
|
|
|
Preparation of patents
|
|
|
442
|
|
|
|
522
|
|
|
|
427
|
|
|
Depreciation and amortization
|
|
|
473
|
|
|
|
637
|
|
|
|
417
|
|
|
Vehicle maintenance
|
|
|
686
|
|
|
|
677
|
|
|
|
764
|
|
|
Travel
|
|
|
2,755
|
|
|
|
2,173
|
|
|
|
869
|
|
|
Insurance
|
|
|
267
|
|
|
|
94
|
|
|
|
131
|
|
|
Advertising and participation in exhibitions
|
|
|
2,513
|
|
|
|
2,333
|
|
|
|
1,347
|
|
|
Management fees (see Note 17d. )
|
|
|
|
|
|
|
888
|
|
|
|
1,352
|
|
|
Office supplies and printing
|
|
|
283
|
|
|
|
126
|
|
|
|
190
|
|
|
Rent and office maintenance
|
|
|
925
|
|
|
|
1,063
|
|
|
|
741
|
|
|
Professional fees
|
|
|
4,743
|
|
|
|
3,343
|
|
|
|
1,626
|
|
|
Others
|
|
|
450
|
|
|
|
468
|
|
|
|
404
|
|
|
|
|
|
39,512
|
|
|
|
32,212
|
|
|
|
19,871
|
|
Less- grants and participations from the Chief Scientist, see
Note 11
|
|
|
|
|
|
|
|
|
|
|
454
|
|
|
TOTAL COST OF REVENUES, ADMINISTRATIVE AND GENERAL, SELLING AND MARKETING AND RESEARCH AND DEVELOPMENT EXPENSES, NET
|
|
|
39,512
|
|
|
|
32,212
|
|
|
|
19,417
|
|
NOTE
14 - OTHER INCOME, NET:
|
|
|
For the Year Ended
December 31
|
|
|
|
|
2015
|
|
|
2014
|
|
|
2013
|
|
|
|
|
NIS in thousands
|
|
|
|
|
|
|
|
|
|
|
|
|
|
In respect of financial assets at fair value through profit or loss:
|
|
|
|
|
|
|
|
|
|
|
Interest income
|
|
|
|
|
|
|
|
49
|
|
|
Profit from changes in fair value
|
|
|
10
|
|
|
|
66
|
|
|
|
278
|
|
|
|
|
|
10
|
|
|
|
66
|
|
|
|
327
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
In respect of financial asset available for sale
|
|
|
|
|
|
|
|
|
|
|
339
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
In respect of cancellation of an agreement with a customer (*)
|
|
|
|
|
|
|
875
|
|
|
|
|
|
|
Total
|
|
|
10
|
|
|
|
941
|
|
|
|
666
|
|
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
14 - OTHER INCOME, NET
(continued)
:
(*) Supply
agreement
On
January 10, 2010 the Company and Voyage Medical Inc. (a US medical device company, hereinafter – the “Customer”)
entered into a multi-year supply agreement (hereinafter – the "Agreement") for the supply of disposable miniature
video cameras and control systems which were developed by the Company.
On
February 12, 2014, after suitable assurances were not received from the Customer, in accordance with the terms of the Agreement,
the Company terminated the said agreement. As a result of the termination of the Agreement the Company recognized the remaining
deferred revenue balance (of the initial NIS 1,111 thousands) amounting to NIS 370 thousand. In addition, since almost all of
the products relating to the remaining additional advance payments received in 2010 and 2011 totaling NIS 875 thousand were never
delivered to the Customer (excluding a few units relating to an immaterial advance payment), the said advance payments were recorded
to Other Income, Net.
NOTE
15 - FINANCING INCOME (EXPENSES), NET:
|
|
|
|
For the Year Ended
December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
2013
|
|
|
|
|
|
NIS in thousands
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Financing income (expenses) in respect of exchange differences
|
|
|
(25
|
)
|
|
|
2,393
|
|
|
|
(236
|
)
|
|
|
Financing income in respect of deposits
|
|
|
103
|
|
|
|
120
|
|
|
|
54
|
|
|
|
Financing expenses in respect of bank commissions
|
|
|
(127
|
)
|
|
|
(127
|
)
|
|
|
(213
|
)
|
|
|
|
|
|
(49
|
)
|
|
|
2,386
|
|
|
|
(395
|
)
|
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
16 - LOSS PER SHARE:
The
basic loss per share is calculated by dividing the loss attributable to equity holders of the Company by the weighted average
number of shares in issue.
|
|
|
|
For the year ended
December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
2013
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss attributed to shareholders (NIS in thousands)
|
|
|
(37,005
|
)
|
|
|
(22,629
|
)
|
|
|
(5,189
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average of the number of ordinary shares issued (in thousands, adjusted to reverse split
)
|
|
|
28,415
|
|
|
|
19,500
|
|
|
|
13,020
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic loss per share (NIS)
|
|
|
(1.3
|
)
|
|
|
(1.2
|
)
|
|
|
(0. 4)
|
|
|
|
b.
|
Diluted loss per share
|
The
diluted loss per share is calculated by adjusting the weighted average number of ordinary shares outstanding to assume conversion
of all dilutive potential ordinary shares. The Company has one category of dilutive potential ordinary shares - share options
and warrants. A calculation is done to determine the number of shares which could have been acquired at fair value (determined
as the average annual market share price of the Company’s shares) based on the monetary value and according to the conditions
attached to the outstanding options and warrants. The number of shares calculated as above is compared to the number of shares
which would have been issued assuming the exercise of the share options and warrants.
However,
in calculating the diluted loss per share for the years reported, share options granted to employees and others and warrants,
were not taken into account – as their impact in the event of full dilution is anti-dilutive. Therefore, the diluted loss
per share is equal to the basic loss per share.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
17 -TRANSACTIONS AND BALANCES WITH RELATED PARTIES
On
March 3, 2013, as a result of the completion of the transaction with Orbimed (see Note 12 B), Elazar Sonnenschein, Menashe Sonnenschein,
Aviel Roy Shapira, Esther and Kfir Luzzatto and Yair Rabinowitch have signed a letter of undertaking to OrbiMed, or the Shareholders’
Undertaking, according to which each undertook, until the earlier of: (i) the three year anniversary of the closing date; or (ii)
the date on which OrbiMed’s voting rights exceed 45% of the aggregate voting rights of the Company, to vote their shares
in accordance with OrbiMed’s instructions in connection with certain matters such as the size of the board, to elect four
directors nominated by OrbiMed (including the chairman of the board of directors) and for any other matter in accordance with
OrbiMed’s instructions provided that such matter is not ‘unreasonable’. On March 3, 2016, the Shareholders’
Undertaking has expired.
"Related
Parties" – As defined in IAS 24 – 'Related Party Disclosures" (hereinafter- “IAS 24”)
Key
management personnel of the Company - included together with other entities, in the said definition of “Related Parties”
mentioned in IAS 24, include some members of senior management.
|
|
a.
|
Transactions
with related parties:
|
1)
|
|
|
|
For the year ended
December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
2013
|
|
|
|
|
|
NIS in thousands
|
|
|
|
Preparation of patents
|
|
|
442
|
|
|
|
522
|
|
|
|
427
|
|
|
|
Benefits to related parties:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Payroll and related expenses to related parties employed by the Company* (3 recipients)
|
|
|
2,778
|
|
|
|
3,338
|
|
|
|
1,120
|
|
|
|
Compensation to directors not employed by the Company (2015: 3 recipients, 2014: 4 recipients, 2013: 9 recipients,)
|
|
|
226
|
|
|
|
219
|
|
|
|
270
|
|
|
|
Management fees to a related party (see B, D and E following)
|
|
|
|
|
|
|
1,017
|
|
|
|
1,459
|
|
|
|
Compensation to a director employed by the Company
|
|
|
52
|
|
|
|
43
|
|
|
|
46
|
|
|
|
Directors’ insurance
|
|
|
186
|
|
|
|
74
|
|
|
|
47
|
|
|
|
*
|
Includes an amount of NIS 274 thousand (2014: NIS 725 thousand, 2013:
NIS 210 thousand) representing the value of the benefit in respect of options granted. As for the method used to determine the
said value and the assumptions used in calculation thereof, see Note 12 C. The said amount does not include amounts charged in
2013 to the capital reserve as described in section d below.
|
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
17 -TRANSACTIONS AND BALANCES WITH RELATED PARTIES
(continued):
|
|
2)
|
Compensation
to key management personnel
|
The
compensation to key management personnel for employment services they provide to the Company is as follows:
|
|
|
|
For the year ended
December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
2013
|
|
|
|
|
|
NIS in thousands
|
|
|
|
For employment services:
|
|
|
|
|
|
|
|
|
|
|
|
Payroll and other short-term benefits
|
|
*1,768
|
|
|
1,871
|
|
|
813
|
|
|
|
Management fees (see B, D and E following)
|
|
|
|
|
|
|
|
|
|
|
1,439
|
|
|
|
Post-retirement benefits
|
|
|
|
|
|
|
|
|
|
|
51
|
|
|
|
Share based payments
|
|
|
241
|
|
|
|
685
|
|
|
|
195
|
|
|
|
|
|
|
2,009
|
|
|
|
2,556
|
|
|
|
2,498
|
|
*
Including provision for bonus of NIS 337 thousand.
|
|
3)
|
Indemnification,
exemption and insurance for directors and officers of the Company
|
|
|
a.
|
The
Company provides its directors and officers with an obligation for indemnification and
exemption.
|
|
|
b.
|
The
Company has a directors and officers' liability insurance policy covering all Company’s
directors and officers. We currently have directors’ and officers’ liability
insurance providing total coverage of $15 million for the benefit of all of our directors
and officers, in respect of which we are charged a twelve-month premium of $ 52,855,
and which includes a deductible of up to $35,000 per claim, other than such claims related
to extraordinary events such as securities related claims (other than claims filed in
the United States or Canada), for which the deductible shall not exceed $100,000, claims
filed in the United States or Canada, for which the deductible shall not exceed $35,000,
or securities related claims filed in the United States or Canada, for which the deductible
shall not exceed $100,000.
|
|
|
b.
|
Balances
with related parties
:
|
|
|
|
|
As of December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
NIS in thousands
|
|
|
|
Current liabilities-presented in the statements of financial position among “trade and other payables”)
|
|
|
639
|
|
|
|
722
|
|
|
|
c.
|
As
to options granted to related parties, see Note 12 C.
|
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
17 -TRANSACTIONS AND BALANCES WITH RELATED PARTIES
(continued):
|
|
d.
|
Change
in the employment terms of related parties
|
On
March 3, 2013 the Company received notices from DLLD Consulting Ltd. (through Dr. Elazar Sonnenschein, the Company’s CEO
at that time and one of its controlling shareholders), from the Company’s R&D director and one of its controlling shareholders,
and from the Company’s medical director and one of its controlling shareholders, requesting that the Company cancel the
reduction of fees and salaries (where relevant) paid to the above controlling shareholders, with effect from the date of issuance
of the notice. In light of the above, the fees and the salaries paid to the above controlling shareholders were increased.
Amounts
relating to waiver of salaries by controlling shareholders during the reporting periods were credited to capital reserve from
transactions with controlling shareholders against payroll expenses.
|
|
e.
|
Kfir
Luzzatto and Esther Luzzatto provide patents and trademarks preparation and registration
services to the Company through Luzzatto and Luzzatto Patent Attorneys (General Partnership),
whose partners are Kfir Luzzatto and Esther Luzzatto, directly and/or indirectly, and
through Alandal Ltd., that to the best knowledge of the Company is under the ownership
and control of Kfir Luzzatto and/or Esther Luzzatto. In accordance with an agreement
between the Company and Luzzatto and Luzzatto Patent Attorneys of February 27, 2007,
the fee paid by the Company to Luzzatto and Luzzatto Patent Attorneys amounts to USD
125 (plus V.A.T.) per hour invested by a patent attorney partner, patent writer, attorney,
engineer, or technical professional, and USD 60 (plus VAT) per hour invested by a technical
draftsman. The fee is paid in NIS and is linked to the representative exchange rate of
the USD with trade credit terms of Net 60. In addition, Luzzatto and Luzzatto Patent
Attorneys is to be reimbursed for its expenses against invoices or actual payment vouchers,
including payment to patent attorneys abroad and fees to patent registration offices.
It should be noted that the above agreement does not specify the engagement period or
the conditions for termination thereof.
|
On
March 24, 2014 and March 27, 2014 the audit committee and the board of directors, respectively, re-approved the terms of the agreement
described above, for an additional period of three years in accordance with Amendment No. 16 of the Israeli Companies Law.
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
18 – Entity level disclosures:
|
|
a.
|
The Chief Operation Decision Maker ("CODM") decided to no longer
evaluate separately the performance of the Visual segment and the MUSE segment, but rather evaluates the performance of all operations
together. The CODM allocates resources and evaluates activities on a Group-wide basis and no longer receives information on a
disaggregated basis. As such, the Group has only one operating segment.
|
|
|
|
|
For the year ended
December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
2013
|
|
|
|
|
|
NIS in thousands
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Miniature camera and related equipment
|
|
|
2,160
|
|
|
|
2,336
|
|
|
|
2,451
|
|
|
|
MUSE and related equipment
|
|
|
256
|
|
|
|
328
|
|
|
|
47
|
|
|
|
|
|
|
2,416
|
|
|
|
2,664
|
|
|
|
2,498
|
|
|
|
c.
|
Revenues by geographical area (based on the location of customers):
|
|
|
|
|
For the year ended
December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
2013
|
|
|
|
|
|
NIS in thousands
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
USA
|
|
|
1,730
|
|
|
|
1,645
|
|
|
|
1,631
|
|
|
|
Japan
|
|
|
126
|
|
|
|
358
|
|
|
|
362
|
|
|
|
Others
|
|
|
560
|
|
|
|
661
|
|
|
|
505
|
|
|
|
|
|
|
2,416
|
|
|
|
2,664
|
|
|
|
2,498
|
|
|
|
d.
|
All of the Group's long-lived assets are located in Israel
.
|
Set
forth below is a breakdown of Company's revenue by major customers (major customer –revenues from this customer constitute
at least 10% of total revenues in a certain year):
|
|
|
|
For the year ended
December 31
|
|
|
|
|
|
2015
|
|
|
2014
|
|
|
2013
|
|
|
|
|
|
NIS in thousands
|
|
|
|
Customer A
|
|
|
1,020
|
|
|
|
|
|
|
|
323
|
|
|
|
Customer B
|
|
|
|
|
|
|
169
|
|
|
|
536
|
|
|
|
Customer C (see also Note 14)
|
|
|
|
|
|
|
370
|
|
|
|
185
|
|
|
|
Customer D
|
|
|
|
|
|
|
443
|
|
|
|
|
|
MEDIGUS
LTD.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS (Continued)
NOTE
19 - POST BALANCE SHEET EVENTS:
|
|
a.
|
On
March 3, 2016 the Shareholders’ Undertaking with Orbimed and warrants (Orbimed)
were expired, see note 12(b)(1) and note 17.
|
|
|
|
|
|
|
b.
|
On
February 9, 2016 570,000 options (Series 6) expired as the exercise period was ended.
|
|
|
|
|
|
|
c.
|
During
January-March 2016, 62,500 options (Series D) forfeited and 37,500 options (Series D)
and 24,800 options (Series F) expired, due to the termination of employment of several
employees.
|
|
|
|
|
|
|
d.
|
On March 16, 2016, a former Company secretary and internal legal advisor (the "Employee") issued a notice letter to the Company. The letter notes a number of claims, among which is that the Employee was wrongfully terminated. At this early stage and based on the advice of its legal counsels, the Company's management cannot assess the outcome of these proceedings. Accordingly, no provision has been recorded in the financial statements.
|
MEDIGUS
LTD.
CONDENSED
CONSOLIDATED INTERIM FINANCIAL STATEMENTS
(UNAUDITED)
AS
OF SEPTEMBER 30, 2016
MEDIGUS
LTD.
CONDENSED
CONSOLIDATED INTERIM FINANCIAL STATEMENTS
(UNAUDITED)
AS
OF SEPTEMBER 30, 2016
TABLE
OF CONTENTS
MEDIGUS
LTD.
CONDENSED
CONSOLIDATED STATEMENTS OF FINANCIAL POSITION
|
|
|
September 30,
|
|
|
December 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
|
|
USD in thousands
|
|
|
Assets
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CURRENT ASSETS:
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
|
3,868
|
|
|
|
10,312
|
|
|
Other receivables:
|
|
|
|
|
|
|
|
|
|
Trade receivables
|
|
|
258
|
|
|
|
254
|
|
|
Other
|
|
|
630
|
|
|
|
1,006
|
|
|
Inventory
|
|
|
237
|
|
|
|
304
|
|
|
|
|
|
4,993
|
|
|
|
11,876
|
|
|
|
|
|
|
|
|
|
|
|
|
NON-CURRENT ASSETS:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Inventory
|
|
|
826
|
|
|
|
-
|
|
|
Property and equipment, net
|
|
|
194
|
|
|
|
226
|
|
|
Intangible assets, net
|
|
|
27
|
|
|
|
39
|
|
|
|
|
|
1,047
|
|
|
|
265
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL ASSETS
|
|
|
6,040
|
|
|
|
12,141
|
|
MEDIGUS
LTD.
CONDENSED
CONSOLIDATED STATEMENTS OF FINANCIAL POSITION
|
|
|
September 30,
|
|
|
December 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
|
|
USD in thousands
|
|
|
Liabilities and equity
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CURRENT LIABILITIES:
|
|
|
|
|
|
|
|
Accounts payable:
|
|
|
|
|
|
|
|
Trade payables
|
|
|
311
|
|
|
|
358
|
|
|
Other
|
|
|
1,527
|
|
|
|
1,495
|
|
|
|
|
|
1,838
|
|
|
|
1,853
|
|
|
NON-CURRENT LIABILITIES:
|
|
|
|
|
|
|
|
|
|
Warrants at fair value
|
|
|
32
|
|
|
|
9
|
|
|
Retirement benefit obligation , net
|
|
|
101
|
|
|
|
98
|
|
|
|
|
|
133
|
|
|
|
107
|
|
|
TOTAL LIABILITIES
|
|
|
1,971
|
|
|
|
1,960
|
|
|
|
|
|
|
|
|
|
|
|
|
EQUITY:
|
|
|
|
|
|
|
|
|
|
Ordinary share capital
|
|
|
1,040
|
|
|
|
870
|
|
|
Share premium
|
|
|
53,108
|
|
|
|
51,990
|
|
|
Other capital reserves
|
|
|
147
|
|
|
|
118
|
|
|
Warrants
|
|
|
1,532
|
|
|
|
1,532
|
|
|
Accumulated deficit
|
|
|
(51,758
|
)
|
|
|
(44,329
|
)
|
|
TOTAL EQUITY
|
|
|
4,069
|
|
|
|
10,181
|
|
|
TOTAL LIABILITIES AND EQUITY
|
|
|
6,040
|
|
|
|
12,141
|
|
The
accompanying notes are an integral part of these condensed consolidated financial statements.
MEDIGUS
LTD.
CONDENSED
CONSOLIDATED STATEMENTS OF LOSS AND
OTHER COMPREHENSIVE LOSS
|
|
|
Nine
months ended
|
|
|
Three
months ended
|
|
|
Year
ended
|
|
|
|
|
September 30
|
|
|
September 30
|
|
|
December 31
|
|
|
|
|
2016
|
|
|
2015
|
|
|
2016
|
|
|
2015
|
|
|
2015
|
|
|
|
|
USD
in thousands
|
|
|
Revenues
|
|
|
496
|
|
|
|
399
|
|
|
|
136
|
|
|
|
239
|
|
|
|
624
|
|
|
Cost
of revenues
|
|
|
154
|
|
|
|
169
|
|
|
|
33
|
|
|
|
112
|
|
|
|
277
|
|
|
Gross
profit
|
|
|
342
|
|
|
|
230
|
|
|
|
103
|
|
|
|
127
|
|
|
|
347
|
|
|
Research and development
expenses
|
|
|
3,021
|
|
|
|
3,043
|
|
|
|
708
|
|
|
|
1,032
|
|
|
|
4,384
|
|
|
Selling and marketing
expenses
|
|
|
1,845
|
|
|
|
1,943
|
|
|
|
349
|
|
|
|
716
|
|
|
|
2,680
|
|
|
General and administrative
expenses
|
|
|
3,016
|
|
|
|
1,911
|
|
|
|
993
|
|
|
|
715
|
|
|
|
2,842
|
|
|
Other
income, net
|
|
|
|
|
|
|
3
|
|
|
|
|
|
|
|
|
|
|
|
3
|
|
|
Operating
loss
|
|
|
(7,540
|
)
|
|
|
(6,664
|
)
|
|
|
(1,947
|
)
|
|
|
(2,336
|
)
|
|
|
(9,556
|
)
|
|
Profit from changes
in fair value of warrants issued to investors
|
|
|
17
|
|
|
|
15
|
|
|
|
8
|
|
|
|
210
|
|
|
|
106
|
|
|
Financial income in
respect of deposits and exchange differences
|
|
|
130
|
|
|
|
54
|
|
|
|
43
|
|
|
|
191
|
|
|
|
19
|
|
|
Financial
expenses in respect of bank commissions
|
|
|
(12
|
)
|
|
|
(26
|
)
|
|
|
(3
|
)
|
|
|
(9
|
)
|
|
|
(33
|
)
|
|
Financial
income (expenses), net
|
|
|
118
|
|
|
|
28
|
|
|
|
40
|
|
|
|
182
|
|
|
|
(14
|
)
|
|
Loss before taxes on
income
|
|
|
(7,405
|
)
|
|
|
(6,621
|
)
|
|
|
(1,899
|
)
|
|
|
(1,944
|
)
|
|
|
(9,464
|
)
|
|
Taxes
on income
|
|
|
-24
|
|
|
|
(50
|
)
|
|
|
-
|
|
|
|
(26
|
)
|
|
|
(68
|
)
|
|
Loss
for the period
|
|
|
(7,429
|
)
|
|
|
(6,671
|
)
|
|
|
(1,899
|
)
|
|
|
(1,970
|
)
|
|
|
(9,532
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other
comprehensive loss:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Currency
translation differences
|
|
|
|
|
|
|
(298
|
)
|
|
|
|
|
|
|
(549
|
)
|
|
|
(211
|
)
|
|
Total
comprehensive loss for the period
|
|
|
(7,429
|
)
|
|
|
(6,969
|
)
|
|
|
(1,899
|
)
|
|
|
(2,519
|
)
|
|
|
(9,743
|
)
|
|
Basic
and diluted loss per share
|
|
|
(0.22
|
)
|
|
|
(0.25
|
)
|
|
|
(0.05
|
)
|
|
|
(0.06
|
)
|
|
|
(0.34
|
)
|
The
accompanying notes are an integral part of these condensed consolidated financial statements.
MEDIGUS
LTD.
CONDENSED
CONSOLIDATED STATEMENTS OF CHANGES IN EQUITY
|
|
|
Equity
attributed to the owners of the Company
|
|
|
|
|
|
|
|
Ordinary
shares
|
|
|
Share
premium
|
|
|
Capital
reserves from options
granted
|
|
|
Capital
reserves
from transactions
with controlling
shareholders
|
|
|
Currency
translation differences
|
|
|
Warrants
|
|
|
Accumulated
deficit
|
|
|
Total
equity
|
|
|
|
|
USD
in thousands
|
|
|
BALANCE
AS OF JANUARY 1, 2016
|
|
|
870
|
|
|
|
51,990
|
|
|
|
697
|
|
|
|
538
|
|
|
|
(1,117
|
)
|
|
|
1,532
|
|
|
|
(44,329
|
)
|
|
|
10,181
|
|
|
CHANGES
DURING THE 9-MONTH PERIOD ENDED SEPTEMBER 30, 2016 -
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL
COMPREHENSIVE LOSS FOR THE PERIOD
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(7,429
|
)
|
|
|
(7,429
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TRANSACTIONS
WITH SHAREHOLDERS:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Proceeds
from issuance of shares
|
|
|
170
|
|
|
|
1,025
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,195
|
|
|
Options
granted to employees and service providers
|
|
|
|
|
|
|
|
|
|
|
122
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
122
|
|
|
Forfeiture
of options
|
|
|
|
|
|
|
93
|
|
|
|
(93
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL
TRANSACTIONS WITH SHAREHOLDERS
|
|
|
170
|
|
|
|
1,118
|
|
|
|
29
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,317
|
|
|
BALANCE
AS OF SEPTEMBER 30, 2016
|
|
|
1,040
|
|
|
|
53,108
|
|
|
|
726
|
|
|
|
538
|
|
|
|
(1,117
|
)
|
|
|
1,532
|
|
|
|
(51,758
|
)
|
|
|
4,069
|
|
MEDIGUS
LTD.
CONDENSED
CONSOLIDATED STATEMENTS OF CHANGES IN EQUITY
|
|
|
Equity
attributed to the owners of the Company
|
|
|
|
|
|
|
|
Ordinary
shares
|
|
|
Share
premium
|
|
|
Capital
reserves from
options
granted
|
|
|
Capital
reserves
from transactions
with
controlling
shareholders
|
|
|
Currency
translation differences
|
|
|
Warrants
|
|
|
Accumulated
deficit
|
|
|
Total
equity
|
|
|
|
|
USD
in thousands
|
|
|
BALANCE
AS OF JULY 1, 2016
|
|
|
870
|
|
|
|
52,080
|
|
|
|
703
|
|
|
|
538
|
|
|
|
(1,117
|
)
|
|
|
1,532
|
|
|
|
(49,859
|
)
|
|
|
4,747
|
|
|
CHANGES
DURING THE 3-MONTH PERIOD ENDED SEPTEMBER 30, 2016 -
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL
COMPREHENSIVE LOSS FOR THE PERIOD
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1,899
|
)
|
|
|
(1,899
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TRANSACTIONS
WITH SHAREHOLDERS:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Proceeds
from issuance of shares
|
|
|
170
|
|
|
|
1,025
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,195
|
|
|
Options
granted to employees and service providers
|
|
|
|
|
|
|
|
|
|
|
26
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
26
|
|
|
Forfeiture
of options
|
|
|
|
|
|
|
3
|
|
|
|
(3
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL
TRANSACTIONS WITH SHAREHOLDERS
|
|
|
170
|
|
|
|
1,028
|
|
|
|
23
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,221
|
|
|
BALANCE
AS OF SEPTEMBER 30, 2016
|
|
|
1,040
|
|
|
|
53,108
|
|
|
|
726
|
|
|
|
538
|
|
|
|
(1,117
|
)
|
|
|
1,532
|
|
|
|
(51,758
|
)
|
|
|
4,069
|
|
MEDIGUS
LTD.
CONDENSED
CONSOLIDATED STATEMENTS OF CHANGES IN EQUITY
|
|
|
Equity
attributed to the owners of the Company
|
|
|
|
|
|
|
Ordinary
shares
|
|
|
Share
premium
|
|
|
Capital
reserves from options granted
|
|
|
Capital
reserves for transactions with controlling shareholders
|
|
|
Currency
translation differences
|
|
|
Warrants
|
|
|
Accumulated
deficit
|
|
|
Total
equity
|
|
|
|
|
USD
in thousands
|
|
|
BALANCE
AS OF JANUARY 1, 2015
|
|
|
683
|
|
|
|
46,065
|
|
|
|
665
|
|
|
|
538
|
|
|
|
(905
|
)
|
|
|
801
|
|
|
|
(34,796
|
)
|
|
|
13,051
|
|
|
CHANGES
DURING THE 9-MONTH PERIOD ENDED SEPTEMBER 30, 2015 -
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL
COMPREHENSIVE LOSS FOR THE PERIOD
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(298
|
)
|
|
|
|
|
|
|
(6,671
|
)
|
|
|
(6,969
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TRANSACTIONS
WITH SHAREHOLDERS:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Proceeds
from issuance of shares and warrants
|
|
|
187
|
|
|
|
5,840
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
731
|
|
|
|
|
|
|
|
6,758
|
|
|
Options
granted to employees and service providers
|
|
|
|
|
|
|
|
|
|
|
99
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
99
|
|
|
Forfeiture
of options
|
|
|
|
|
|
|
83
|
|
|
|
(83
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL
TRANSACTIONS WITH SHAREHOLDERS
|
|
|
187
|
|
|
|
5,923
|
|
|
|
16
|
|
|
|
|
|
|
|
|
|
|
|
731
|
|
|
|
|
|
|
|
6,857
|
|
|
BALANCE
AS OF SEPTEMBER 30, 2015
|
|
|
870
|
|
|
|
51,988
|
|
|
|
681
|
|
|
|
538
|
|
|
|
(1,203
|
)
|
|
|
1,532
|
|
|
|
(41,467
|
)
|
|
|
12,939
|
|
MEDIGUS
LTD.
CONDENSED
CONSOLIDATED STATEMENTS OF CHANGES IN EQUITY
|
|
|
Equity
attributed to the owners of the Company
|
|
|
|
|
|
|
Ordinary
shares
|
|
|
Share
premium
|
|
|
Capital
reserves from options granted
|
|
|
Capital
reserves for transactions with controlling shareholders
|
|
|
Currency
translation differences
|
|
|
Warrants
|
|
|
Accumulated
deficit
|
|
|
Total
equity
|
|
|
|
|
USD
in thousands
|
|
|
BALANCE
AS OF JULY 1, 2015
|
|
|
683
|
|
|
|
46,148
|
|
|
|
656
|
|
|
|
538
|
|
|
|
(654
|
)
|
|
|
801
|
|
|
|
(39,497
|
)
|
|
|
8,675
|
|
|
CHANGES
DURING THE 3-MONTH PERIOD ENDED JUNE SEPTEMBER, 2015 -
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TOTAL
COMPREHENSIVE LOSS FOR THE PERIOD
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(549
|
)
|
|
|
|
|
|
|
(1,970
|
)
|
|
|
(2,519
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TRANSACTIONS
WITH SHAREHOLDERS:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Proceeds
from issuance of shares and warrants
|
|
|
187
|
|
|
|
5,840
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
731
|
|
|
|
|
|
|
|
6,758
|
|
|
Options
granted to employees and service providers
|
|
|
|
|
|
|
|
|
|
|
25
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
25
|
|
|
TOTAL
TRANSACTIONS WITH SHAREHOLDERS
|
|
|
187
|
|
|
|
5,840
|
|
|
|
25
|
|
|
|
|
|
|
|
|
|
|
|
731
|
|
|
|
|
|
|
|
6,783
|
|
|
BALANCE
AS OF SEPTEMBER 30, 2015
|
|
|
870
|
|
|
|
51,988
|
|
|
|
681
|
|
|
|
538
|
|
|
|
(1,203
|
)
|
|
|
1,532
|
|
|
|
(41,467
|
)
|
|
|
12,939
|
|
The
accompanying notes are an integral part of these condensed consolidated financial statements.
MEDIGUS
LTD.
CONDENSED
CONSOLIDATED STATEMENTS OF CASH FLOWS
|
|
|
Three months ended
|
|
|
Nine months ended
|
|
|
|
|
September 30
|
|
|
|
|
2015
|
|
|
2016
|
|
|
2015
|
|
|
2016
|
|
|
|
|
USD in thousands
|
|
|
CASH FLOWS FROM OPERATING ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash used in operations (see Appendix)
|
|
|
(7,764
|
)
|
|
|
(4,149
|
)
|
|
|
(1,906
|
)
|
|
|
(1,982
|
)
|
|
Income tax paid
|
|
|
(10
|
)
|
|
|
(35
|
)
|
|
|
|
|
|
|
(11
|
)
|
|
Interest received
|
|
|
|
|
|
|
33
|
|
|
|
|
|
|
|
29
|
|
|
Net cash flow used in operating activities
|
|
|
(7,774
|
)
|
|
|
(4,151
|
)
|
|
|
(1,906
|
)
|
|
|
(1,964
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CASH FLOWS FROM INVESTING ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Acquisition of property and equipment
|
|
|
(32
|
)
|
|
|
(67
|
)
|
|
|
|
|
|
|
(35
|
)
|
|
Acquisition of intangible assets
|
|
|
(3
|
)
|
|
|
(11
|
)
|
|
|
(1
|
)
|
|
|
(2
|
)
|
|
Net cash flow used in investing activities
|
|
|
(35
|
)
|
|
|
(78
|
)
|
|
|
(1
|
)
|
|
|
(37
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CASH FLOWS FROM FINANCING ACTIVITIES:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Proceeds from issuance of shares and warrants, note 5
|
|
|
1,235
|
|
|
|
6,758
|
|
|
|
1,235
|
|
|
|
6,758
|
|
|
Net cash flows generated from financing activities
|
|
|
1,235
|
|
|
|
6,758
|
|
|
|
1,235
|
|
|
|
6,758
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
INCREASE (DECREASE) IN CASH AND CASH EQUIVALENTS
|
|
|
(6,574
|
)
|
|
|
2,529
|
|
|
|
(672
|
)
|
|
|
4,757
|
|
|
BALANCE OF CASH AND CASH EQUIVALENTS AT BEGINNING OF PERIOD
|
|
|
10,312
|
|
|
|
10,817
|
|
|
|
4,488
|
|
|
|
8,729
|
|
|
EXCHANGE DIFFERENCES ON CASH AND CASH EQUIVALENTS
|
|
|
130
|
|
|
|
(236
|
)
|
|
|
52
|
|
|
|
(376
|
)
|
|
BALANCE OF CASH AND CASH EQUIVALENTS AT END OF PERIOD
|
|
|
3,868
|
|
|
|
13,110
|
|
|
|
3,868
|
|
|
|
13,110
|
|
MEDIGUS
LTD.
CONDENSED
CONSOLIDATED STATEMENTS OF CASH FLOWS
Appendix
to the condensed consolidated statements of cash flows
|
|
|
Nine Months ended
|
|
|
Three months ended
|
|
|
|
|
September 30
|
|
|
|
|
2016
|
|
|
2015
|
|
|
2016
|
|
|
2015
|
|
|
|
|
USD in thousands
|
|
|
Net cash used in operations:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss for the period before taxes on income
|
|
|
(7,405
|
)
|
|
|
(6,621
|
)
|
|
|
(1,899
|
)
|
|
|
(1,944
|
)
|
|
Adjustment in respect of:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest received
|
|
|
|
|
|
|
(33
|
)
|
|
|
|
|
|
|
(29
|
)
|
|
Liability for employee benefits, net
|
|
|
3
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Depreciation
|
|
|
64
|
|
|
|
76
|
|
|
|
23
|
|
|
|
25
|
|
|
Amortization of intangible assets
|
|
|
15
|
|
|
|
15
|
|
|
|
4
|
|
|
|
5
|
|
|
Exchange differences on cash and cash equivalents
|
|
|
(130
|
)
|
|
|
(33
|
)
|
|
|
(52
|
)
|
|
|
(176
|
)
|
|
Amounts charged in respect of options granted to employees and service providers
|
|
|
122
|
|
|
|
99
|
|
|
|
26
|
|
|
|
25
|
|
|
Gains on change in the fair value of financial instruments at fair value through profit or loss
|
|
|
|
|
|
|
(3
|
)
|
|
|
|
|
|
|
|
|
|
Profit on change in the fair value of warrants issued to investors
|
|
|
(17
|
)
|
|
|
(15
|
)
|
|
|
(8
|
)
|
|
|
(210
|
)
|
|
Changes in operating asset and liability items:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Decrease (increase) in accounts receivables :
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Trade
|
|
|
(4
|
)
|
|
|
11
|
|
|
|
30
|
|
|
|
(92
|
)
|
|
Other
|
|
|
376
|
|
|
|
(413
|
)
|
|
|
159
|
|
|
|
(178
|
)
|
|
Increase (decrease) in accounts payable :
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Trade
|
|
|
(47
|
)
|
|
|
59
|
|
|
|
45
|
|
|
|
(5
|
)
|
|
Other
|
|
|
18
|
|
|
|
623
|
|
|
|
(204
|
)
|
|
|
614
|
|
|
Decrease (increase) in inventory
|
|
|
(759
|
)
|
|
|
8
|
|
|
|
(30
|
)
|
|
|
(17
|
)
|
|
Net sales of financial assets at fair value through profit or loss
|
|
|
|
|
|
|
2,078
|
|
|
|
|
|
|
|
|
|
|
Net cash used in operations
|
|
|
(7,764
|
)
|
|
|
(4,149
|
)
|
|
|
(1,906
|
)
|
|
|
(1,982
|
)
|
The
accompanying notes are an integral part of these condensed consolidated financial statements.
MEDIGUS
LTD.
NOTES
TO THE CONDENSED FINANCIAL STATEMENTS
SEPTEMBER
30, 2016
NOTE
1 - GENERAL:
|
|
a.
|
General
Medigus Ltd. (hereinafter – the “Company") together with its subsidiary
(hereinafter – the “Group") is a medical device group specializing in
developing innovative endoscopic procedures and devices. To date most of the Group's
research and development activities have been focused in developing and manufacturing
of the MUSE endoscopy system (hereinafter - “MUSE”), a FDA approved system,
for the treatment of gastroesophageal reflux disease (GERD), which is one of the most
common chronic diseases in the western world. In addition, the Group uses the technological
platform it developed for the purpose of additional special systems and products that
are suitable for both medical and industrial applications.
|
To
date, in order to increase its revenues, the company continues negotiations to market the MUSE endoscopy system, together with
marketing and selling miniature cameras and related equipment, which it developed and manufactures.
The
Company's shares are listed for trade on the Tel Aviv Stock Exchange Ltd. (“TASE”) and as of May 20, 2015, the Company’s
American Depository Shares (ADSs) evidenced by American Depositary Receipts (ADRs) are listed for trade on the NASDAQ Capital
Market. As of the report date, each ADR represents five ordinary shares of the Company. The Company’s depositary agent for
the ADR program is The Bank of New York Mellon.
The
Company was incorporated in Israel on December 9, 1999 and is resident in Israel. The address of its registered office is P.O.
Box 3030, Omer, 8496500.
MEDIGUS
LTD.
NOTES
TO THE CONDENSED FINANCIAL STATEMENTS
SEPTEMBER
30, 2016
NOTE
1 – GENERAL
(continued)
:
|
|
b.
|
Change
in the functional currency
|
Effective
January 1, 2016, the Company changed its functional currency to the U.S. dollar (“dollar” or “USD”) from
the New Israeli Shekel (“NIS”). This change was based on an assessment by Company management that the dollar is the
primary currency of the economic environment in which the Company operates. Accordingly, the functional and reporting currency
of the Company in these financial statements is the USD.
In
determining the appropriate functional currency to be used, the Company followed the guidance in International Accounting Standard
(IAS) 21, which states that economic factors relating to sales, costs and expenses, financing activities and cash flows, as well
as other potential factors, should be considered both individually and collectively. The Company recently incurred a significant
increase in expenses denominated in dollars relating to the marketing of its products in the US, which is reflected primarily
by recruitment of additional human resources in the US. The Company expects an additional increase in expenses denominated in
dollars related to these activities. These changes, as well as the fact that all of the Company’s budgeting and planning
is conducted solely in dollars, led to the decision to make the change in functional currency as of January 1, 2016, as indicated
above.
The
effect of the change in the functional currency is accounted for prospectively. All items were translated into the new functional
currency using the exchange rate at the date of the change. The resulting translated amounts for non-monetary items are treated
as their historical cost.
Due
to the change in its functional currency as above and concurrently with it, the Company decided to change its presentation currency
from NIS to USD.
The
change in presentation currency was applied retrospectively to all comparative figures presented.
In
effecting the change in presentation currency to the dollar, with respect to comparative figures: (1) all assets and liabilities
of the Company were translated using the dollar exchange rate as of each balance sheet presented; (2) equity items were translated
using historical exchange rates at the relevant transaction dates; (3) the statements of comprehensive loss items have been translated
at the average exchange rates for the relevant reporting periods; and (4) the resulting translation differences have been reported
as "currency translation differences" within other comprehensive loss.
On
July 22, 2007 the Company established a wholly owned subsidiary, MEDIGUS USA LLC, in the USA (hereinafter - the “Subsidiary”).
The
Subsidiary did not engage in any business activities until October 2013.
On October 1, 2013, the Company and the Subsidiary
entered into an agreement whereby the Subsidiary provides services to the Company in consideration for reimbursement of direct
costs plus a reasonable premium.
MEDIGUS
LTD.
NOTES
TO THE CONDENSED FINANCIAL STATEMENTS
SEPTEMBER
30, 2016
NOTE
1 – GENERAL
(continued)
:
During
the nine-month period ended September 30, 2016, the Group had a total comprehensive loss of USD 7.4 million. As of September 30,
2016 the Group had accumulated losses of USD 52 million and a positive working capital of USD 3.2 million. As at the approval
date of these financial statements the Group had a total cash balance of approximately USD 3.1 million. Based on the projected
cash flows and its cash balances as of September 30, 2016, the Group's Management is of the opinion that without further fund
raising it will not have sufficient resources to enable it to continue advancing its activities including the development, manufacturing
and marketing of its products for a period of at least 12 months from the date of approval of these financial statements. As a
result, there is substantial doubt about the Group’s ability to continue as a going concern.
Management’s
plans include the continued commercialization of their products, continue taking cost reduction steps and securing sufficient
financing through the sale of additional equity securities, debt or capital inflows from strategic partnerships. There are no
assurances however, that the Group will be successful in obtaining the level of financing needed for its operations. If the Group
is unsuccessful in commercializing its products and securing sufficient financing, it may need to reduce activities, curtail or
cease operations.
The
financial statements do not include any adjustments relating to the recoverability and classification of recorded assets and the
amounts and classification of liabilities that might be necessary should the Group be unable to continue as a going concern.
NOTE
2 - BASIS FOR PREPARATION OF THE CONDENSED FINANCIAL STATEMENTS:
|
|
a.
|
The
Group’s condensed consolidated financial information as of September 30, 2016,
and for the nine-month and three-month interim periods ended on that date (hereinafter-“the
interim financial information”) has been prepared in accordance with the guidance
of IAS 34 ‘Interim Financial Reporting’ (hereafter – “IAS 34”).
The interim financial information does not include all of the information and disclosures
required in annual financial statements. The interim financial information should be
read in conjunction with the 2015 annual financial statements and its accompanying notes,
which are in compliance with International Financial Reporting Standards (hereinafter
– “IFRS”), which are standards and interpretations issued by the International
Accounting Standards Board. Interim results are not indicative of future or full year
results. The financial statements were approved on November 28, 2016.
|
The
preparation of interim financial statements requires the Group’s management to exercise judgment and also requires the use
of accounting estimates and assumptions that affect the application of the Group’s accounting policy and the reported amounts
of assets, liabilities, revenues and expenses. Actual results could differ from those estimates.
In
the preparation of these interim financial statements, the significant judgments exercised by management in the application of
the Group’s accounting policy and the uncertainty involved in the key sources of those estimates were identical to the ones
used in the Group’s annual financial statements for the year ended December 31, 2015.
MEDIGUS
LTD.
NOTES
TO THE CONDENSED FINANCIAL STATEMENTS
SEPTEMBER
30, 2016
NOTE
3 - SIGNIFICANT ACCOUNTING POLICIES
The
accounting policies adopted in the preparation of the interim financial information are consistent with those followed in the
preparation of the Group's annual financial statements for the year ended December 31, 2015.
NOTE
4 - SUPPLIES AND PRODUCTS
The
Company's supplies and products comprises of:
|
|
|
|
September 30,
|
|
|
December 31,
|
|
|
|
|
|
2016
|
|
|
2015
|
|
|
|
|
|
USD in thousands
|
|
|
|
CURRENT ASSETS:
|
|
|
|
|
|
|
|
|
Inventory
|
|
|
237
|
|
|
|
304
|
|
|
|
Pre-paid expenses
|
|
|
385
|
|
|
|
652
|
|
|
|
NON-CURRENT ASSETS:
|
|
|
|
|
|
|
|
|
|
|
Inventory
|
|
|
826
|
|
|
|
-
|
|
|
|
|
|
|
1,448
|
|
|
|
956
|
|
Supplies
and products recorded as pre-paid expenses represent non-commercial activities such as training and clinical and registry trials.
MEDIGUS
LTD.
NOTES
TO THE CONDENSED FINANCIAL STATEMENTS
SEPTEMBER
30, 2016
NOTE
5 - EQUITY:
On
September 12, 2016, the Company closed a registered direct offering, pursuant to which the Company issued a total of 1.28 million
American depositary shares (ADS) at a purchase price per ADS of $1.15.
The
immediate proceeds (gross) from the issuance of securities offered amounted to approximately USD 1.47 million.
Net
proceeds from the issuance, net of issuance expenses, amounted to approximately USD 1.2 million.
NOTE
6 - TRANSACTIONS WITH RELATED PARTIES:
Compensation
to key management personnel for employment services which were provided to the Group (including stock based compensation) in the
nine-month and three-month periods ended September 30, 2016 was USD 282 thousand and USD 55 thousand, respectively (in the nine-month
and three-month periods ended September 30, 2015, USD 400 thousand and USD 113 thousand, respectively, and in the year ended December
31, 2015 – USD 517 thousand).
MEDIGUS
LTD.
NOTES
TO THE CONDENSED FINANCIAL STATEMENTS
SEPTEMBER
30, 2016
NOTE
7 – LEGAL PROCEEDINGS
:
|
|
a.
|
On
June 16, 2015, the Company filed a claim in the District Court of Delaware against EndoChoice,
Inc., or EndoChoice, for its patent infringement, seeking an injunctive order and damages.
On October 6, 2015, the Company filed claims against EndoChoice’s trademark applications
with the Israeli Patent Office. In response, on December 29, 2015, EndoChoice filed a
separate trademark and unfair competition action against the Company and its U.S. Subsidiary.
In addition, on January 27, 2016, EndoChoice commenced an opposition procedure with the
Israeli Patent Office against the Company's trademark application in Israel.
|
On
October 28, 2016, the Group and EndoChoice, Inc. (“EndoChoice”) settled all litigation and administrative proceedings
between themselves, including those actions pending in the U.S. District Court for the District of Delaware C.A. Nos. 15-505-LPS-CJB
and C.A. No. 15-1525-LPS-CJB and the trademark opposition proceedings in the State of Israel involving Trademark Application Nos.
257172, 260433 and 262423. Under the terms of the confidential settlement, Medigus was granted a covenant not to sue with respect
to EndoChoice FUSE-related trademarks and EndoChoice was granted a non-exclusive license to Medigus’ U.S. Patent No. 6,997,871
and related patents. Each party has agreed to bear its own costs and fees associated with the Litigations.
|
|
b.
|
On
March 30, 2016, a former Company secretary and internal legal advisor (the "Employee")
filed a lawsuit against the Company. The lawsuit notes several claims, among which is
that the Employee was wrongfully terminated. According to the Company's management
and based on the advice of its legal advisors, the lawsuit should not have a material
unfavorable effect on the Company's financial statements. Therefore, no allowance regarding
this lawsuit was recorded in these financial statements.
|
|
|
c.
|
On October
6 2016, following a withholding tax audit in Israel, the company is deemed to be in debt
of approximately USD 900 thousand additional withholding taxes. It is the management's
opinion, based on the opinion of its legal advisors, that the chances for the claims
being dismissed are more likely than not. Therefore, no allowance regarding this assessment
was recorded in these financial statements.
|
NOTE
8 - SUBSEQUENT EVENTS
:
On
November 17, 2016, the Company, took additional steps in its cost reduction program (hereinafter – the program). As part
of the program, Mr. Gilad Mamlok, the CFO of the Company, will leave the Company at the end of January 2017. Mr. Jeremy Starkweather,
the Company’s VP U.S. Sales and Marketing will leave the Company at the end of December 2016. Mr. Oded Yatzkan will be appointed
as the Company’s CFO upon Mr. Mamlok’s termination. Mr. Yatzkan is the Company’s Director of Finance and accounting,
who also has served as the Company’s CFO from September 2003 until August 2015. Mr. Chris Rowland, the CEO, will assume
the responsibilities of the VP U.S. Sales and Marketing.

MEDIGUS LTD.
Up to 929,752
of
Class A Units consisting of American Depositary Shares and Warrants
and
309,917
of
Class B Units consisting of Pre-Funded Warrants and Warrants
(1,549,586
American Depositary Shares underlying the Warrants)
|
|
|
|
|
|
Rodman
& Renshaw
a
unit of H.C. Wainwright & Co.
|
|
|
|
PRELIMINARY
PROSPECTUS
|
|
Part
II
Information
Not Required in Prospectus
Item
6. Indemnification of Office Holders (including Directors).
Under
the Companies Law, a company may not exculpate an office holder from liability for a breach of a fiduciary duty. An Israeli company
may exculpate an office holder in advance from liability to the company, in whole or in part, for damages caused to the company
as a result of a breach of duty of care but only if a provision authorizing such exculpation is included in its articles of association.
Our articles of association include such a provision. The company may not exculpate in advance a director from liability arising
out of a prohibited dividend or distribution to shareholders.
Under
the Companies Law and the Securities Law, 5738-1968 (the “Securities Law”) a company may indemnify an office holder
in respect of the following liabilities, payments and expenses incurred for acts performed by him or her as an office holder,
either in advance of an event or following an event, provided its articles of association include a provision authorizing such
indemnification:
|
|
●
|
a
monetary liability incurred by or imposed on the office holder in favor of another person
pursuant to a court judgment, including pursuant to a settlement confirmed as judgment
or arbitrator’s decision approved by a competent court. However, if an undertaking
to indemnify an office holder with respect to such liability is provided in advance,
then such an undertaking must be limited to events which, in the opinion of the board
of directors, can be foreseen based on the company’s activities when the undertaking
to indemnify is given, and to an amount or according to criteria determined by the board
of directors as reasonable under the circumstances, and such undertaking shall detail
the abovementioned foreseen events and amount or criteria;
|
|
|
●
|
reasonable
litigation expenses, including reasonable attorneys’ fees, which were incurred
by the office holder as a result of an investigation or proceeding filed against the
office holder by an authority authorized to conduct such investigation or proceeding,
provided that such investigation or proceeding was either (i) concluded without the filing
of an indictment against such office holder and without the imposition on him of any
monetary obligation in lieu of a criminal proceeding; (ii) concluded without the filing
of an indictment against the office holder but with the imposition of a monetary obligation
on the office holder in lieu of criminal proceedings for an offense that does not require
proof of criminal intent; or (iii) in connection with a monetary sanction;
|
|
|
●
|
reasonable
litigation expenses, including attorneys’ fees, incurred by the office holder or
which were imposed on the office holder by a court (i) in a proceeding instituted against
him or her by the company, on its behalf, or by a third party, (ii) in connection with
criminal indictment of which the office holder was acquitted, or (iii) in a criminal
indictment which the office holder was convicted of an offense that does not require
proof of criminal intent;
|
|
|
●
|
a
monetary liability imposed on the office holder in favor of a payment for a breach offended
at an Administrative Procedure (as defined below) as set forth in Section 52(54)(a)(1)(a)
to the Securities Law;
|
|
|
●
|
expenses
expended by the office holder with respect to an Administrative Procedure under the Securities
Law, including reasonable litigation expenses and reasonable attorneys’ fees; and
|
|
|
●
|
any
other obligation or expense in respect of which it is permitted or will be permitted
under applicable law to indemnify an office holder, including, without limitation, matters
referenced in Section 56H(b)(1) of the Securities Law.
|
An
“Administrative Procedure” is defined as a procedure pursuant to chapters H3 (Monetary Sanction by the Israeli Securities
Authority), H4 (Administrative Enforcement Procedures of the Administrative Enforcement Committee) or I1 (Arrangement to prevent
Procedures or Interruption of procedures subject to conditions) to the Securities Law.
Under
the Companies Law and the Securities Law, a company may insure an office holder against the following liabilities incurred for
acts performed by him or her as an office holder if and to the extent provided in the company’s articles of association:
|
|
●
|
a
breach of a fiduciary duty to the company, provided that the office holder acted in good
faith and had a reasonable basis to believe that the act would not harm the company;
|
|
|
●
|
a
breach of duty of care to the company or to a third party, to the extent such a breach
arises out of the negligent conduct of the office holder;
|
|
|
●
|
a
monetary liability imposed on the office holder in favor of a third party;
|
|
|
●
|
a
monetary liability imposed on the office holder in favor of an injured party at an Administrative
Procedure pursuant to Section 52(54)(a)(1)(a) of the Securities Law; and
|
|
|
●
|
expenses
incurred by an office holder in connection with an Administrative Procedure, including
reasonable litigation expenses and reasonable attorneys’ fees.
|
Under
the Companies Law, a company may not indemnify, exculpate or insure an office holder against any of the following:
|
|
●
|
a
breach of fiduciary duty, except for indemnification and insurance for a breach of the
fiduciary duty to the company to the extent that the office holder acted in good faith
and had a reasonable basis to believe that the act would not prejudice the company;
|
|
|
●
|
a
breach of duty of care committed intentionally or recklessly, excluding a breach arising
out of the negligent conduct of the office holder;
|
|
|
●
|
an
act or omission committed with intent to derive illegal personal benefit; or
|
|
|
●
|
a
civil or administrative fine or forfeit levied against the office holder.
|
Under
the Companies Law, exculpation, indemnification and insurance of office holders must be approved by the compensation committee
and the board of directors and, with respect to directors or controlling shareholders, their relatives and third parties in which
such controlling shareholders have a personal interest, also by the shareholders.
Our articles of association permit us to
exculpate, indemnify and insure our office holders to the fullest extent permitted or to be permitted by law. Our office holders
are currently covered by a directors’ and officers’ liability insurance policy.
We
have entered into agreements with each of our current director and officers exculpating them from a breach of their duty of care
to us to the fullest extent permitted by law, subject to limited exceptions, and undertaking to indemnify them to the fullest
extent permitted by law, to the extent that these liabilities are not covered by insurance. This indemnification is limited, with
respect to any monetary liability imposed in favor of a third party, to events determined as foreseeable by the board of directors
based on our activities. The maximum aggregate amount of indemnification that we may pay to our directors and officers based on
such indemnification agreement is equal to 25% of our shareholders’ equity pursuant to our latest audited or unaudited consolidated
financial statements, as applicable, as of the date of the indemnification payment. Such indemnification amounts are in addition
to any insurance amounts. Each director or officer who agrees to receive this letter of indemnification also gives his approval
to the termination of all previous letters of indemnification that we have provided to him or her in the past, if any. However,
in the opinion of the SEC, indemnification of office holders for liabilities arising under the Securities Act is against public
policy and therefore unenforceable.
Item
7. Recent Sales of Unregistered Securities.
Set
forth below are the sales of all securities by the Company during the three years preceding this offering, which were not registered
under the Securities Act.
In December 2016, we issued investors warrants
to purchase up to a total of 39,871 ADSs representing 1,993,550 ordinary shares, at an initial exercise price of $9.00 per ADS
in a
concurrent private placement
to an offering pursuant to our shelf registration
statement in the United States. See “Certain Relationships and Related Party Transactions – Material Contracts.”
In December 2016, we issued Rodman &
Renshaw a unit of H.C. Wainwright & Co. warrants to purchase up to a total of
3,987 ADSs
representing 199,355 ordinary shares at an exercise price of $7.37 per ADS as part of the consideration for its services as placement
agent. For a description of the warrants’ terms see “Management’s Discussion and Analysis of Financial Condition
and Results of Operations—Overview—Warrants.”
In December 2016, we issued Roth Capital
Partners and Maxim Group LLC warrants to purchase up to a total of 1,993
ADSs representing
99,675 ordinary shares at an exercise price of $9.00 per ADS as part of a “tail” fee consideration for its services
as placement agent. For a description of the warrants’ terms see “Management’s Discussion and Analysis of Financial
Condition and Results of Operations—Overview—Warrants.”
In
September 2016, we issued to Roth Capital Partners and Maxim Group LLC warrants purchase up to 3,955 ADSs representing 197,750
ordinary share at an exercise price of $14.375 per ADS as part of the consideration for its services as placement agent. For a
description of the warrants’ terms see “Management’s Discussion and Analysis of Financial Condition and Results
of Operations—Overview—Warrants.”
In
July 2015, pursuant to a public offering under our shelf prospectus in Israel, we raised $7 million through the issuance of a
total of 7,052,500 of our ordinary shares at a price of NIS 3.8 per share and warrants exercisable into additional 3,526,250 of
our ordinary shares. See “Certain Relationships and Related Party Transactions – Material Contracts.”
In
June and August 2014, following entering into private equity placement agreements, we raised an aggregate amount of approximately
$11.1 million through the issuance of 8,527,154 of our ordinary shares at a price per share of NIS 4.56 and warrants to purchase
up to 3,410,861 of our ordinary shares at an exercise price of NIS 6.27 per share. See “Certain Relationships and Related
Party Transactions – Material Contracts.”
The
foregoing issuances of warrants to purchase ADSs in 2016 were offered pursuant to Rule 506 of Regulation D and Section 4(a)(2)
of the Securities Act.
The
foregoing issuances in 2014 and in 2015 were all made outside of the United States pursuant to Regulation S or to U.S. entities
pursuant to Section 4(a)(2) of the Securities Act.
The foregoing issuances were (i) adjusted
retroactively to reflect the
change in the ratio of ordinary shares per ADS from five deposited
ordinary shares per ADS to 50 deposited ordinary shares per ADS effected on March 15, 2017
,
and
(ii) adjusted
retroactively to reflect
the 10:1 reverse share split and the change
in the ratio of ordinary shares per ADS to five deposited ordinary shares per ADS effected on November 6, 2015
.
Item
8. Exhibits and Financial Statement Schedules.
|
|
(a)
|
The Exhibit Index is hereby incorporated herein by reference.
|
|
|
|
|
|
|
(b)
|
Financial Statement Schedules.
|
All
Financial Statement Schedules have been omitted because either they are not required, are not applicable or the information required
therein is otherwise set forth in the Registrant’s consolidated financial statements and related notes thereto.
Item
9. Undertakings.
|
|
(a)
|
The undersigned Registrant hereby undertakes:
|
|
|
(1)
|
To
file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement:
|
|
|
i.
|
To
include any prospectus required by section 10(a)(3) of the Securities Act of 1933;
|
|
|
ii.
|
To
reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most
recent post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information
set forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered
(if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low
or high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the Commission pursuant
to Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than 20% change in the maximum aggregate
offering price set forth in the “Calculation of Registration Fee” table in the effective registration statement;
|
|
|
iii.
|
To
include any material information with respect to the plan of distribution not previously disclosed in the registration statement
or any material change to such information in the registration statement.
|
|
|
(2)
|
That,
for the purpose of determining any liability under the Securities Act of 1933, each such post-effective amendment shall be
deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities
at that time shall be deemed to be the initial bona fide offering thereof.
|
|
|
(3)
|
To
remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold
at the termination of the offering.
|
|
|
(4)
|
To
file a post-effective amendment to the registration statement to include any financial statements required by Item 8.A. of
Form 20-F at the start of any delayed offering or throughout a continuous offering. Financial statements and information otherwise
required by Section 10(a)(3) of the Act need not be furnished, provided that the registrant includes in the prospectus, by
means of a post-effective amendment, financial statements required pursuant to this paragraph (a)(4) and other information
necessary to ensure that all other information in the prospectus is at least as current as the date of those financial statements.
Notwithstanding the foregoing, with respect to registration statements on Form F-3, a post-effective amendment need not be
filed to include financial statements and information required by Section 10(a)(3) of the Act or Rule 3-19 of this chapter
if such financial statements and information are contained in periodic reports filed with or furnished to the Commission by
the registrant pursuant to section 13 or section 15(d) of the Securities Exchange Act of 1934 that are incorporated by reference
in the Form F-3.
|
|
|
(5)
|
That,
for the purpose of determining liability under the Securities Act of 1933 to any purchaser:
|
|
|
i.
|
If
the registrant is relying on Rule 430B:
|
|
|
A.
|
Each
prospectus filed by the registrant pursuant to Rule 424(b)(3) shall be deemed to be part of the registration statement as
of the date the filed prospectus was deemed part of and included in the registration statement; and
|
|
|
|
|
|
|
B.
|
Each
prospectus required to be filed pursuant to Rule 424(b)(2), (b)(5), or (b)(7) as part of a registration statement in reliance
on Rule 430B relating to an offering made pursuant to Rule 415(a)(1)(i), (vii), or (x) for the purpose of providing the information
required by section 10(a) of the Securities Act of 1933 shall be deemed to be part of and included in the registration statement
as of the earlier of the date such form of prospectus is first used after effectiveness of the date of the first contract
or sale of securities in the offering described in the prospectus. As provided in Rule 430B, for liability purposes of the
issuer and any person that is at that date an underwriter, such date shall be deemed to be a new effective date of the registration
statement relating to the securities in the registration statement to which that prospectus relates, and the offering of such
securities at that time shall be deemed to be the initial bona fide offering thereof. Provided, however, that no statement
made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated
or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement
will, as to a purchaser with a time of contract sale prior to such effective date, supersede or modify any statement that
was made in the registration statement or prospectus that was part of the registration statement or made in any such document
immediately prior to such effective date; or
|
|
|
ii.
|
If
the registrant is subject to Rule 430C, each prospectus filed pursuant to Rule 424(b) as part of a registration statement
relating to an offering, other than registration statements relying on Rule 430B or other prospectuses filed in reliance on
Rule 430A, shall be deemed to be part of and included in the registration statement as of the date it is first used after
effectiveness. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration
statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus
that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such first use,
supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration
statement or made in any such document immediately prior to such date of first use.
|
|
|
(6)
|
That,
for the purpose of determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial
distribution of the securities, the undersigned registrant undertakes that in a primary offering of securities of the undersigned
registrant pursuant to this registration statement, regardless of the underwriting method used to sell the securities to the
purchaser, if the securities are offered or sold to such purchaser by means of any of the following communications, the undersigned
registrant will be a seller to the purchaser and will be considered to offer or sell securities to such purchaser:
|
|
|
i.
|
Any
preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant
to Rule 424;
|
|
|
|
|
|
|
ii.
|
Any
free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred
to by the undersigned registrant;
|
|
|
|
|
|
|
iii.
|
The
portion of any other free writing prospectus relating to the offering containing material information about the undersigned
registrant or its securities provided by or on behalf of the undersigned registrant; and
|
|
|
|
|
|
|
iv.
|
Any
other communication that is an offer in the offering made by the undersigned registrant to the purchaser.
|
(b)
Insofar as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers and
controlling persons of the registrant pursuant to the provisions described in Item 6 hereof, or otherwise, the registrant
has been advised that in the opinion of the SEC such indemnification is against public policy as expressed in the Act and is,
therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the
registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense
of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities
being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent,
submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed
in the Act and will be governed by the final adjudication of such issue.
(c)
The undersigned registrant hereby undertakes that:
(1)
That for purposes of determining any liability under the Securities Act of 1933, the information omitted from the form of prospectus
filed as part of this registration statement in reliance upon Rule 430A and contained in a form of prospectus filed by the
registrant pursuant to Rule 424(b)(1) or (4), or 497(h) under the Securities Act shall be deemed to be part of this registration
statement as of the time it was declared effective.
(2)
That for the purpose of determining any liability under the Securities Act of 1933, each post-effective amendment that contains
a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering
of such securities at that time shall be deemed to be the initial
bona fide
offering thereof.
SIGNATURES
Pursuant to the requirements of the Securities
Act of 1933, the registrant certifies that it has reasonable grounds to believe that it meets all of the requirements for filing
on Form F-1 and has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized,
in the City of Omer, State of Israel on March 23, 2017.
|
|
Medigus
Ltd.
|
|
|
|
|
|
By:
|
/s/
Christopher Rowland
|
|
|
Name:
|
Christopher Rowland
|
|
|
Title:
|
Chief Executive Officer
|
Pursuant
to the requirements of the Securities Act of 1933, this registration statement has been signed by the following persons in the
capacities and on the dates indicated.
|
Signatures
|
|
Title
|
|
Date
|
|
|
|
|
|
|
|
/s/
Christopher Rowland
|
|
Chief Executive
Officer and Director
|
|
March
23, 2017
|
|
Christopher Rowland
|
|
|
|
|
|
|
|
|
|
|
|
/s/
Oded Yatzkan
|
|
Chief Financial
Officer
|
|
March
23, 2017
|
|
Oded Yatzkan
|
|
|
|
|
|
|
|
|
|
|
|
*
|
|
Chairman of the
Board of Directors
|
|
March
23, 2017
|
|
Dr. Nissim Darvish
|
|
|
|
|
|
|
|
|
|
|
|
*
|
|
Director
|
|
March
23, 2017
|
|
Eitan Machover
|
|
|
|
|
|
|
|
|
|
|
|
*
|
|
Director
|
|
March
23, 2017
|
|
Efrat Venkert
|
|
|
|
|
|
|
|
|
|
|
|
*
|
|
Director
|
|
March
23, 2017
|
|
Doron Birger
|
|
|
|
|
|
|
|
|
|
|
|
*
|
|
Director
|
|
March
23, 2017
|
|
Anat Naschitz
|
|
|
|
|
|
*
|
/s/
Christopher Rowland
|
|
|
|
Christopher Rowland
Attorney-in-fact
|
|
Signature
of authorized representative in the United States
Pursuant to the requirements of the Securities
Act of 1933, as amended, the Registrant’s duly authorized representative has signed this registration statement on Form
F-1 in on this 23
rd
day of March 2017.
|
|
Medigus
USA LLC
|
|
|
|
|
|
By:
|
/s/
Christopher Rowland
|
|
|
Name:
|
Christopher Rowland
|
|
|
Title:
|
Chief Executive Officer
|
EXHIBIT
INDEX
|
Exhibit No.
|
|
Description
|
|
1.1
|
|
Form of Engagement Letter with Rodman &
Renshaw a unit of H.C. Wainwright & Co.
(1)
|
|
3.1
|
|
Articles of Association of Medigus Ltd., as amended
(2)
|
|
4.1
|
|
Form of American Depositary Receipt
(2)
|
|
4.2
|
|
Form of Series A Warrant
(1)
|
|
4.3
|
|
Form of Pre-Funded Series B Warrant
(1)
|
|
4.4
|
|
Form of Placement Agent Warrant
(1)
|
|
4.5
|
|
Form of Warrant to purchase Ordinary Shares Represented by American Depositary Shares issued in connection with the December 2016 Securities Purchase Agreements
(6)
|
|
5.1
|
|
Form of Opinion of Meitar Liquornik Geva Leshem Tal, Israeli counsel to the Registrant, as to the validity of the ordinary shares
(1)
|
|
5.2
|
|
Form of Opinion of Zysman, Aharoni, Gayer and Sullivan & Worcester, U.S. counsel to the Registrant
(1)
|
|
10.1
|
|
Form of Deposit Agreement between Medigus Ltd., The Bank of New York Mellon as Depositary, and owners and holders from time to time of ADSs issued thereunder, including the Form of American Depositary Shares.
(2)
|
|
10.2
|
|
2013 Share Option and Incentive Plan
(2)
|
|
10.3
|
|
Series A Option Plan
(2)
¥
|
|
10.4
|
|
Series B Option Plan
(2)
¥
|
|
10.5
|
|
Compensation Policy of Medigus Ltd.
(3)
|
|
10.6
|
|
Lease Agreement between Medigus USA LLC and Regus Plc dated December 4, 2013
(2)
|
|
10.7
|
|
Lease Agreement between Medigus Ltd. and Sky-City Office Center for HiTech Industries Ltd. dated June 15, 2014
(2)
¥
|
|
10.8
|
|
Lease Agreement Tefen Yazamut Ltd. regarding main offices in Omer Industrial Park dated December 10, 2013
(2)
¥
|
|
10.9
|
|
Form of Indemnification and Exculpation Undertaking
(2)
|
|
10.10
|
|
Securities Purchase Agreement by and Securities Purchase Agreement by and between the Registrant and the purchasers named therein, dated September 8, 2016
(4)
|
|
10.11
|
|
Securities Purchase Agreement by and between the Registrant and the purchasers named therein, dated November 30, 2016.
(5)
|
|
10.12
|
|
Form of Securities Purchase Agreement
(1)
|
|
21.1
|
|
List of Subsidiaries
(2)
|
|
23.1
|
|
Consent of Kesselman and Kesselman, Member Firm of PricewaterhouseCoopers International Limited
(1)
|
|
23.2
|
|
Consent of Meitar Liquornik Geva Leshem Tal, Israeli counsel to the Registrant (included in Exhibit 5.1)
(1)
|
|
23.3
|
|
Consent of Zysman, Aharoni, Gayer and Sullivan & Worcester, U.S. counsel to the Registrant (included in Exhibit 5.2)
(1)
|
|
24.1
|
|
Power of Attorney (included in the signature
page of the Registration Statement)
(7)
|
|
(1)
|
Furnished
herewith.
|
|
(2)
|
Previously filed with the Securities and Exchange Commission on May 7, 2015,
as an exhibit to the Registrant’s annual report
on Form 20-F (File No 001-37381) and incorporated by reference herein.
|
|
(3)
|
Previously filed with the Securities and Exchange Commission on March 30, 2016, as an exhibit to the Registrant’s annual report on Form 20-F (File No 001-37381) and incorporated by reference herein.
|
|
(4)
|
Previously filed with the Securities and Exchange Commission on September 13, 2016, as Exhibit 10.1 to the Registrant’s report on Form 6-K (File No 001-37381) and incorporated by reference herein.
|
|
(5)
|
Previously filed with the Securities and Exchange Commission on December 1, 2016, as Exhibit 10.1 to the Registrant’s report on Form 6-K (File No 001-37381) and incorporated by reference herein.
|
|
(6)
|
Previously filed with the Securities and Exchange Commission on December 1, 2016, as Exhibit 4.1 to the Registrant’s report on Form 6-K (File No 001-37381) and incorporated by reference herein.
|
|
(7)
|
Previously filed.
|
|
¥
|
English
translation of original Hebrew document.
|
II-9
Medigus (NASDAQ:MDGS)
Historical Stock Chart
From Aug 2024 to Sep 2024
Medigus (NASDAQ:MDGS)
Historical Stock Chart
From Sep 2023 to Sep 2024