United
States Securities and Exchange Commission
Washington,
D.C. 20549
Form
10-K
| [X] |
ANNUAL
REPORT UNDER SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934:
For
the fiscal year ending August 31, 2014 |
| [
] |
TRANSITION
REPORT UNDER SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934:
For
the transition period from ___________ to ___________. |
Commission
file number: 333-154989
ENTEST
BIOMEDICAL, INC.
(Name
of small business issuer in its charter)
| Nevada |
|
26-3431263 |
| (State
or other jurisdiction of incorporation or organization) |
|
(I.R.S.
Employer Identification No.) |
4700
Spring Street, Suite 304, La Mesa, California, 91942
(Address
of Principal executive offices)
Issuer’s
telephone number: ( 619) 702-1404
_______________
Securities
registered under Section 12(b) of the “Exchange Act” None
Securities
registered under Section 12(g) of the Exchange Act: None
Indicate
by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange
Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports),
and (2) has been subject to such filing requirements for the past 90 days. Yes [X] No
[ ]
Indicate
by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K (§ 229.405 of this chapter) is not
contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements
incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K
Indicate
by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or smaller reporting
company.
| Large
accelerated filer [ ] |
Accelerated
filer [ ] |
| Non
accelerated filer [ ] |
Smaller
reporting Company [X] |
Indicate
by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes [ ] No
[X]
Indicate
by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive
Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the
preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes [X] No [ ]
State
the aggregate market value of the voting and non-voting common equity held by non-affiliates computed by reference to the price
at which the common equity was last sold, or the average bid and asked price of such common equity, as of the last business day
of the registrant’s most recently completed second fiscal quarter: $ 966,456
As
of November 21, 2014 Entest BioMedical, Inc. had 2,405,570, 642 common shares outstanding, 4, 201,397 Series B Preferred shares
outstanding , 100,000 Series AA preferred shares outstanding and 80,000 Series AAA preferred shares outstanding.
In
this annual report, the terms “Entest BioMedical, Inc.. ”, “Entest”, “Company”,
“we”, or “our”, unless the context otherwise requires, mean Entest BioMedical, Inc., a Nevada corporation,
and its subsidiary.
CAUTIONARY
STATEMENT REGARDING FORWARD-LOOKING STATEMENTS
This
annual report on Form 10-K and other reports that we file with the SEC contain statements that are considered forward-looking
statements. Forward-looking statements give the Company’s current expectations, plans, objectives, assumptions
or forecasts of future events. All statements other than statements of current or historical fact contained in this annual report,
including statements regarding the Company’s future financial position, business strategy, budgets, projected costs and
plans and objectives of management for future operations, are forward-looking statements. In some cases, you can identify forward-looking
statements by terminology such as “anticipate,” “estimate,” “plans,” “potential,”
“projects,” “ongoing,” “expects,” “management believes,” “we believe,”
“we intend,” and similar expressions. These statements are based on the Company’s current plans and are subject
to risks and uncertainties, and as such the Company’s actual future activities and results of operations may be materially
different from those set forth in the forward looking statements. Any or all of the forward-looking statements in this annual
report may turn out to be inaccurate and as such, you should not place undue reliance on these forward-looking statements. The
Company has based these forward-looking statements largely on its current expectations and projections about future events and
financial trends that it believes may affect its financial condition, results of operations, business strategy and financial needs.
The forward-looking statements can be affected by inaccurate assumptions or by known or unknown risks, uncertainties and assumptions
due to a number of factors, including:
| • |
dependence on key personnel; |
| • |
competitive factors; |
| • |
degree of success of research and development programs |
| • |
the operation of our business; and |
| • |
general economic conditions |
These
forward-looking statements speak only as of the date on which they are made, and except to the extent required by federal securities
laws, we undertake no obligation to update any forward-looking statements to reflect events or circumstances after the date on
which the statement is made or to reflect the occurrence of unanticipated events. In addition, we cannot assess the impact of
each factor on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially
from those contained in any forward-looking statements. All subsequent written and oral forward-looking statements attributable
to the Company or persons acting on its behalf are expressly qualified in their entirety by the cautionary statements contained
in this annual report.
PART
I
Item
1. Business
We
were incorporated in the State of Nevada on September 24, 2008 as JB Clothing Corporation. Until July 10, 2009, our principal
business objective was the offering of active/leisure fashion design clothing.
On
July 10, 2009 we abandoned our efforts in the field of active/leisure fashion design clothing when we acquired 100% of the share
capital of Entest BioMedical, Inc., a California corporation, (“Entest CA”) from Bio-Matrix Scientific Group, Inc.
(“BMSN”) for consideration consisting of 10,000,000 shares of the common stock of the Company and the cancellation
of 10,000,000 shares of the Company owned and held by Mr. Rick Plote.
As
a result of this transaction, the former stockholder of Entest CA held approximately 70% of the voting capital stock of the Company
immediately after the transaction. For financial accounting purposes, this acquisition was a reverse acquisition of the
Company by Entest CA under the purchase method of accounting, and was treated as a recapitalization with Entest CA as the acquirer.
As of November 1, 2011 the former stockholder of Entest CA held approximately 48% of the outstanding common shares of the Company.
Upon
acquisition of Entest CA, we abandoned our efforts in the field of active/leisure fashion design clothing. Our business
is currently the business of Entest CA, and we currently intend to develop and commercialize therapies, medical devices and medical
testing procedures. On July 12, 2009 we adopted the name of Entest CA when we changed our name to Entest BioMedical, Inc.
The
Company’s current strategy is to develop and commercialize therapies, medical devices and medical testing procedures for
the veterinary market. It is believed by the Company that any required regulatory approvals can be obtained much more rapidly
with regard to products and services developed for the veterinary market and that the achievement of successful clinical trials
and commercialization of such products and services may allow the company to enter into collaborations with larger pharmaceutical
companies for the purpose of developing and commercializing these products and services for human usage.
The
process by which a new drug is approved for use in humans within the United States generally begins prior to submission of the
IND (Investigational New Drug Application) with the FDA.
Prior
to submission of the IND, the sponsor of the drug compound under development must test the drugs on laboratory animals (preclinical
testing) in order that toxicity may be determined and efficacy may be demonstrated. The results of such preclinical testing is
crucial in determining whether or not the sponsor may proceed onto clinical trials on human beings and preclinical testing is
required to be performed on multiple species.
Drug
studies in humans can begin only after an IND is reviewed by the FDA and a local institutional review board (IRB). The board is
a panel of scientists and non-scientists in hospitals and research institutions that oversees clinical research.
IRBs
approve the clinical trial protocols, which describe the type of people who may participate in the clinical trial, the schedule
of tests and procedures, the medications and dosages to be studied, the length of the study, the study's objectives, and other
details. IRBs make sure the study is acceptable, that participants have given consent and are fully informed of their risks, and
that researchers take appropriate steps to protect patients from harm.
After
trial protocols have been approved the sponsor moves on to Phase I clinical trials (to determine safety and toxicity in a small
number of volunteers) and, if Phase 1 studies don't reveal unacceptable toxicity, Phase II and Phase III clinical trials to determine
effectiveness.
The
process by which a new drug is approved for veterinary use within the United States generally begins with the sponsor
researching and developing the new compound and conducting initial (“pilot”) studies on it for
a specific use in a specific animal species (called the “target animal” species) If the results of the pilot
studies are promising and there is a potential market for the drug, the drug sponsor contacts The US Food and Drug
Administration’s Center for Veterinary Medicine (CVM) to officially begin the drug approval process by opening an
Investigational New Animal Drug (“INAD”) file. Information is submitted regarding Chemistry, Manufacturing,
and Controls; Effectiveness; Target Animal Safety; Human Food Safety(if applicable); Environmental Impact (if applicable) and
Labeling in support of the NADA (New Animal Drug Application) which is submitted by the sponsor for approval by the FDA.
With
the exception of a biologic product which can be classified as a medical device, Biologics developed for human use generally
are undergo the same path to FDA approval as for drugs. Biologics classified as medical devices may, in most instance, be
subject to premarket approval by the FDA. Medical devices intended for veterinary use are not subject to premarket approval by
the FDA.
Veterinary
Biologics are regulated by the U.S. Department of Agriculture (USDA) which is authorized, under the 1913 Virus-Serum-Toxin
Act as amended by the 1985 Food Security Act, to ensure that all veterinary biologics produced in, or imported into, the United
States are not worthless, contaminated, dangerous, or harmful. The Veterinary Biologics Program of the USDA's Animal and Plant
Health Inspection Service (APHIS) oversees the veterinary biologics industry in the United States.
Domestic
manufacturers of veterinary biologics, for domestic use or for export, are required to possess a valid U.S. Veterinary Biologics
Establishment License and an individual U.S. Veterinary Biologics Product License for each product produced for sale. Prior
to being granted a U.S. Veterinary Biologic Establishment License, the applicant must submit detailed information regarding the
facilities and the qualifications of key personnel and must submit to an inspection of the facilities by the Center for Veterinary
Biologics, a division of the USDA . To qualify for an establishment license, an applicant also must qualify for at least one product
license.
Prior
to being granted a U.S. Veterinary Biologics Product License, the applicant must submit detailed information including test reports
and research data sufficient to establish purity, safety, potency and efficacy of the product, an Outline of Production, and information
regarding labeling and facilities that are to be used in preparation.
It
is the Company’s opinion that factors such as the lack of need for multispecies pre clinical testing, smaller subject size
in efficacy testing (subjects generally in the hundreds for veterinary equivalent of Phase III clinical trials as opposed
to generally in the thousands for Phase III clinical trials for drug compounds for use in humans), lack of the requirement
for premarket approval for medical devices intended for veterinary use should generally lead to a shorter timeframe for
approval by the appropriate regulators of drugs, biologics, and medical devices intended for veterinary use as opposed to drugs,
biologics, and medical devices intended for human use.
The
Company is currently focusing its efforts and allocating its resources towards:
(a)
The development and commercialization of ImenVax™, a therapeutic
cancer vaccine for use in canines
(b)
The acquisition of veterinary clinics
The
acquisition of existing veterinary clinics / hospitals remains an integral part of the Company’s business plan. At this
time, the company is seeking to identify and acquire veterinary practices existing and operating within the areas of San Diego,
California and Orange County, California .The Company believes that these areas are close enough in proximity the Company’s
headquarters to allow ease of interaction with the Company's management . In addition, the Company believes owning and operating
veterinary clinics within the San Diego, California and Orange County , California areas will provide greater convenience for
persons involved with the Company's research and development activities who may be required to utilitize those facilities. The
company is currently in discussions with entities it believes can expedite local clinic acquisitions.
The
Company has not undertaken any discussions with any pharmaceutical companies regarding the commercialization of any products
under development. None of the Company’s products have been approved by any regulatory body for marketing within the
United States or anywhere else. No assurance can be given that all or any of the Company’s currently planned products
will ever be commercialized. Therapies which are veterinary biologics may be administered to patients of veterinary clinics
that may be acquired prior to licensure under the exemption provided by 9 CFR 107.1, which exempts a veterinary
biologic from Federal regulation if the product was manufactured by veterinarians AND intended solely for use with their
clients' animals under a veterinarian-client-patient (VCP) relationship.
Principal
Products and Services
The
Company is currently focusing its research and development efforts toward the successful development and commercialization of
the ImenVax family of canine cancer vaccines as well as the acquisition of existing veterinary clinics / hospitals to be utilized
as potential distribution channels for its ImenVax family of canine cancer vaccines. The Company believes that, in addition to
serving as distribution channels for the Company’s immuno-therapeutic cancer vaccine for canines, these clinics will be
able to generate revenue for the Company from current operations. It is anticipated by the Company that data collected from canine
cancer treatment will provide support for eventual use of this therapy in humans and such therapy may be developed and commercialized
by the Company in collaboration with larger and better capitalized pharmaceutical companies.
ImenVax™
I
ImenVax™
I, currently under development by the Company, is a therapeutic for canine cancer which involves isolating tumor cells from the
patient and then placing the cells into a cell implant device that is inserted subcutaneously into the patient. The resulting
expression of tumor antigens from the device is intended to generate an anti-tumor immune response. The implant chamber device
provokes immune responses to the tumor cells isolated from the patient’s own tumor through a process known as indirect presentation.
Tumor cells implanted in the device are exposed to conditions that are distinct from the tumor’s environment from which
they were isolated. This altered environment allows for anti-tumor responses that are not ordinarily observed in the natural tumor
progression.
The
cells are :
1)
Isolated from the tumor and freed from the natural tumor microenvironment
2)
Subjected to an initial ischemic condition of hypoxia that induces increased antigen expression
3)
Allowed to repopulate within the device in a context that facilitates extended release of tumor antigens.
The
device utilized is comprised of a 0.4 micron inner membrane to retain the implanted cells and an
outer
5 micron membrane that allows blood vessels to form on the surface to enhance biocompatibility. The outer membrane is held in
place by a polyester mesh. The membranes are sonically sealed using a polyester mesh insert.
The
device contains a surface architecture that promotes vascularization in-vivo. There is an initial ischemic phase that may additionally
influence the tumor cell growth characteristics and genetic regulation of the tumor cells.
It
is hypothesized that shortly after implantation, the expression of immunosuppressive molecules is down regulated while the release
of antigens is maintained, thus allowing immune responses to occur that would normally be suppressed.
The
Antigens that are released from the implanted device are taken up by antigen presenting cells (APC).
It
is believed that the APCs will be trained to recognize the cancer cells and alert the body’s immune response, activating
antibodies and T cells to destroy the tumor cells.
The
Company is currently conducting a ten dog safety study to Evaluate ImenVax™ I for the Treatment of Canine Oral Melanoma
and determine adverse effects, if any. As of May 17, 2012 three dogs suffering from oral melanoma have been administered the therapy
with no dog suffering any material adverse reaction.
Inclusion
in the Safety Study is limited to ten dogs with histologically confirmed canine oral melanoma with a Studied Karnofsky
performance status of one or less. The subject are required to be over eight kg with measurable tumor lesions by
caliper or imaging, either primary or metastatic, that may or may not have had prior non-immunological-based therapy.
No concurrent NSAID therapy is allowed and previous use of immune-based therapies is not permitted. Subjects are
required to have a two month life expectancy, and, not have any disease or condition (other than the cancer) that would
preclude living for 3 to 6 months.
Toxicity
is evaluated prior to, and after, treatment and monthly for a period of 3 months. To date, subjects have been recruited solely
from patients of the McDonald Animal Hospital in order that the therapy may be administered licensure under the exemption provided
by 9 CFR 107.1, which exempts a veterinary biologic from Federal regulation if the product was manufactured by veterinarians AND
intended solely for use with their clients' animals under a veterinarian-client-patient (VCP) relationship. To date, 3 dogs have
been enrolled in the safety study with none exhibiting any adverse effects. The Company estimates that an additional $100,000
will be required to be expended to complete the safety study.
Subsequent
to completion of the safety study and pending favorable results, the Company plans to offer ImenVax™ I to its own patients
under the exemption provided by 9 CFR 107.1, which exempts a veterinary biologic from Federal regulation if the product was manufactured
by veterinarians AND intended solely for use with their clients' animals under a veterinarian-client-patient (VCP) relationship
subject to the successful acquisition of one or more veterinary clinics by the Company.
ImenVax
™ II
Also
in early stage development by the Company is a version of ImenVax ™ called ImenVax ™ II which utilizes
cell lines for sustained release of immunologically relevant cytokines for maximum anti-tumor immune responses. It is believed
by the Company that this controlled release of cytokines will act as an adjuvant to be combined with patient’s tumor cells
(antigens) within an implantable membrane encapsulation device.
ImenVax
™ II is designed to function in a manner similar to ImenVax™ I. However, In order to further potentiate the
tumor antigen specific immune responses, the Company intends to include adjuvant cytokine(s) along with tumor cells into the implantation
device. The adjuvants can be added through cytokine expressing cell line. The implantation device to be utilized for
administering ImenVax ™ II is expected to be substantially similar to that utilized in administering ImenVax™
I.
ImenVax
™ III
ImenVax
III is intended to function by harnessing the ability of placental extracts to combat canine cancers. ImenVax™III is intended
to treat existing tumors through stimulation of immune responses to:
a)
kill tumor cells directly;
b)
indirectly kill tumor cells by cutting off the tumor blood supply; and
c)
block the ability of the tumor to suppress the immune system.
Xenogeneic
(from different species) antigen induced immunity has been shown to break self tolerance and capable of engendering immune responses
against the endogenous counterpart self - antigen. The use of xenogeneic placental derived agents such as VEGF (vascular endothelial
growth factor) has demonstrated regression of soft tissue sarcomas in dogs (Kamstock D, Elmslie R, Thamm D, Dow S. 2007. Evaluation
of a xenogeneic VEGF vaccine in dogs with soft tissue sarcoma. 56(8): 1299 - 309).
ImenVax
™ III is intended to be an off the shelf formulation, manufactured under GMP, which shall harness the power of trophoblasts
(cells forming the outer layer of a blastocyst, which provide nutrients to the embryo and develop into a large part of the placenta)
derived from human placental tissue to combat canine cancers . No tissue processing is required for the administration of
the ImenVax ™ III therapy as opposed to I and II as no cellular material from the patient is utilized.
ENT-576
™
ENT-576
™ is a proprietary therapy being developed by the Company for the treatment of Chronic Obstructive Pulmonary Disease
(COPD) such therapy comprising of:
a)
extracting a therapeutic number of cells from a tissue containing in part a stem cell population;
b)
processing said population of cells derived from said tissue so as to concentrate said stem cell population;
c)
systemic re-administration of said cell population into the same patient; and
d)
exposing the patient lung to a sufficient intensity and frequency of laser irradiation necessary to augment therapeutic activity
of said cells in said patient suffering from COPD. The Company has also considered utilizing an FDA approved biochemical drug
to produce the desired augmentation of therapeutic activity.
A
therapeutic intervention in COPD would require addressing the issues of inflammation and regeneration. Although approaches such
as administration of bone marrow stem cells or fat derived cellular components have both regenerative and anti-inflammatory activity
in animal models, the Company feels that the need to enhance their potency for clinical applications can be addressed through
the usage of low level lasers which studies have demonstrated may induce growth factor production, inhibit inflammation
and stimulate angiogenesis.
There
can be no assurance that approvals required will be obtained for any of the Company’s current therapies under development,
or that if such approvals are obtained that the Company will be able to effectively market its therapies. There can be no assurance
given that actual costs and timeframes related to commercialization for any proposed product will not deviate materially from
the Company’s estimation. Currently, none of the Company’s products under development may be administered or marketed
in the United States or outside of the United states except pursuant to an exemption from relevant regulation. The Company does
not anticipate conducting further research and development related to ENT-576™ until completion of the Safety Study due
to limited resources available to the Company.
Distribution
methods of the products or services:
The
Company intends to distribute its products and services through several channels including:
| |
(a) |
utilization
of an internal sales force to market directly to veterinary professionals |
| |
(b) |
distribution
through acquired veterinary clinics if and when such clinics are acquired |
| |
(c) |
utilization
of contract sales organizations |
On
October 19, 2011 the Company entered into an agreement with RenovoCyte LLC and Medistem Inc. (“Agreement”) whereby
the Company shall provide research services to RenovoCyte LLC in connection with a ten dog pilot study to determine the
safety and effectiveness of the utilization of stem cell therapy for the treatment of arthritis in animals (“Pilot Study”).
The term of the Agreement is from October 19, 2011 until the earlier of the completion of the Pilot Study or October 19, 2015
unless terminated by RenovoCyte LLC due to an event of force majeure exceeding a period of 4 months. As consideration for providing
services pursuant to the Agreement, the Company shall enjoy joint publishing rights with regards to the results of the Pilot Study.
Canine mesenchymal multipotent stem cell injections to be utilized during the course of the Pilot Study shall be provided
to the Company by RenovoCyte LLC at no cost to the Company.
As
of November 21, 2014 there have been 8 canine patients treated through the Pilot Study.
Competitive
business conditions and Entest's competitive position in the industry and methods of competition
We
have yet to achieve revenues or profits. The animal health pharmaceutical and biologics industries in which we intend to compete
are highly competitive and characterized by rapid technological advancement. Many of our competitors have greater resources than
we do. Also, The companion animal healthcare industry (e.g. veterinary hospitals and veterinarians) although highly fragmented
is also highly competitive.
We
intend to be competitive by acquiring veterinary hospitals to serve as distribution channels for the products and services we
produce. We also intend to be competitive by utilizing the services and advice of individuals that we believe have expertise in
their field in order that we can concentrate our resources on projects in which products and services in which we have the greatest
potential to secure a competitive advantage may be developed and commercialized .
To
that effect, we have established a Scientific Advisory Board of (the Advisory Board) comprised of individuals who we believe have
a high level of expertise in their professional fields and who have agreed to provide counsel and assistance to us in (a) determining
the viability of proposed projects (b) obtaining financing for projects and (c) obtaining the resources required to initiate and
complete a project in the most cost effective and rapid manner. The members of the Advisory Board have also agreed to act as consultants
on a project by project basis in addition to other services they may provide under any other contractual obligations to us.
Members
of the Advisory Board include as follows:
Dr.
Brian Koos, MD:
Dr.
Brian Koos is Professor and Vice Chair at Obstetrics and Gynecology at the David Geffen School of Medicine at UCLA, Professor
at the Brain Research Institute at the UCLA School of Medicine, and Director of the Maternal-Fetal Medicine Fellowship (UCLA).
Dr. Koos received his MD from Loma Linda University School of Medicine. Dr. Brian Koos is the brother of David R. Koos, the Company’s
Chairman, President and CEO.
Dr.
Koos serves as a member of the Advisory Board pursuant to an agreement by and between the Company and Bio-Matrix Scientific Group,
Inc. entered into on June 19, 2009 whereby the Bio Matrix Scientific Group, Inc assigned its rights to the services of Dr. Koos
to the Company for consideration to bio matrix Scientific Group of $10,000. Those rights included the services of Dr. Koos as
a member of the Company’s Advisory Board for a period ending April 8, 2014.
Dr. Koos serves as a member of the Advisory Board at will and at the pleasure of the Board of
Directors of the Company. There is no binding agreement by and between the Company and Dr. Koos regarding membership on the Advisory
Board.
Dr.
Steven Josephs, PhD:
Dr.
Josephs is currently serving as Executive Manager and Chief Scientific Officer of TherInject LLC, a company involved in the development
of pharmaceuticals to be utilized for the treatment of cancer. Dr. Josephs has 34 years of experience in research and clinical
product development and production for biologics, gene therapy and medical devices.
Dr.
Josephs has previously served as Director of Research and Development for Therapheresis, Inc, Head of Virology and Senior Research
Scientist for Baxter Healthcare Corporation, and Director of Molecular Biology at Universal Biotechnology, Inc where Dr. Josephs
directed a group performing contract molecular biology services for government and private industry.
Dr.
Josephs has also worked for the National Cancer Institute where his duties included studies of the human T-cell leukemia virus
as well as sequence determination and functional analyses of HIV. Dr. Josephs is the co-discoverer of human herpesvirus-6, the
etiologic agent of Roseola.
Dr.
Josephs holds a B.A. in Chemistry, a Ph.D. in Chemistry and has been granted a Professional Certificate in Drug Development and
an ADMET process certificate by the University of California, San Diego. Dr. Josephs has also earned a Master of Science in Science
Teaching.
Dr.
Josephs serves as a member of the Advisory Board at will and at the pleasure of the Board of Directors of the Company. There is
no binding agreement by and between the Company and Dr. Josephs regarding membership on the Advisory Board.
Dr.
Ewa Carrier, MD:
Dr.
Carrier is Associate Professor of Clinical Medicine and Pediatrics, University of California San Diego Blood and Marrow Transplant
Program.
Dr.
Carrier has served as principal investigator for the following clinical protocols:
Protocol
For The Use of AMD3100 to Mobilize Peripheral Blood Stem Cells For Collection and Transplantation - Emergency Compassionate Use,
Single Patient IND.
Erythropoietic
Differentiation of Human ES Cells.
CTLA-4
Blockade with MDX-010 to Induce Graft-Versus-Malignancy Effects Following Allogeneic Hematopoietic Stem Cell Transplantation.
(NCI Protocol Number P-6082) (closed to accrual).
Phase
3 Randomized, Open-label Clinical Trial of Tanespimycin (KOS-953) plus Bortezomib Compared to Bortezomib Alone in Patients with
Multiple Myeloma in First Relapse [Protocol KAG-301] [Protocol Version 21-JUL-2007] Autologous Stem Cell Transplant for Myasthenia
Gravis.
Collection
of Bone Marrow from Patients with Multiple Myeloma for Study of New Therapies.
A
Pilot Study of High-Dose Immunosuppression and Autologous Stem Cell Infusion in Patients with Systemic Lupus Erythematosus Refractory
to Conventional Therapy (closed to accrual).
Autologous
Stem Cell Transplant for Myasthenia Gravis – a retrospective analysis.
Dr
Carrier has served as co investigator for the following clinical protocols:
Pilot
Study of Allogeneic Peripheral Blood Progenitor Cell Transplantation in Patients with Chemotherapy-Refractory or Poor- Prognosis
Metastatic Breast Cancer.
Pilot
Study of a Non-Myeloablative Preparative-Regimen for Allogeneic Peripheral Blood Progenitor Cell Transplantation in Patients with
Chronic Myeloid and Lymphoid Malignancies.
Phase
II Study of a Non-Myeloablative Preparative-Regimen for Allogeneic Hematopoietic Cell Transplantation From Matched Unrelated Donors
in Patients with Chronic Myeloid and Lymphoid Malignancies.
A
Phase II Study of Tumor-Specific Idiotype (Id) and Soluble GM-CSF Vaccination Following Autologous Peripheral Blood Stem Cell
Transplantation in Patients with Low-Grade Non-Hodgkin's Lymphomas.
Phase
II Study of FavId (Tumor-Specific Idiotype-KLH) and Soluble GM-CSF Immunotherapy in Patients with Stable or Progressive Grade
1 or 2 Follicular B-Cell Lymphomas [FavId01].
Phase
II Trial of Rituxan® plus FavId™ (Tumor-Specific Idiotype-KLH) and GM-CSF Immunotherapy in Patients with Grade 1 or
2 Follicular B-Cell Lymphoma [FavId-04].
Dr.
Carrier serves as a member of the Advisory Board at will and at the pleasure of the Board of Directors of the Company. There is
no binding agreement by and between the Company and Dr. Carrier regarding membership on the Advisory Board
Dr.
Feng Lin, MD:
Dr.
Lin is the Director of Research and Development of Entest BioMedical, Inc. and has previously served as Director of Research and
Development of Bio-Matrix Scientific Group, Inc., the Company’s largest shareholder.
Previously,
Dr. Lin was a Senior Research Scientist, Research & Development with Inovio BC, San Diego and Postdoctoral Fellow in Burnham
Institute for Medical Research, La Jolla.
Dr.
Lin received his M.D. from Central South University Xiangya School of Medicine, Changsha, China, and received a M.S. Biochemistry
& Molecular Biology and a Ph.D. Hematology & Physiology from the same institution.
Dr.
Lin serves as a member of the Advisory Board at will and at the pleasure of the Board of Directors of the Company. There is no
binding agreement by and between the Company and Dr. Lin regarding membership on the Advisory Board.
Brenda
S. Phillips, D.V.M.
Dr.
Phillips is a veterinary oncologist and co owner of Veterinary Specialty Hospital of San Diego. She received her Doctor of Veterinary
Medicine in 1992 from Michigan State University, College of Veterinary Medicine.
Dr.
Phillips agreed on January 6, 2011 to serve as a member of the Advisory Board for a period of 24 months. In connection with that
agreement, Dr. Phillips received 10,000 common shares of the Company.
Dr. Phillips serves as a member of the Advisory Board at will and at the pleasure of the Board
of Directors of the Company. There is no binding agreement by and between the Company and Dr. Phillips regarding membership on
the Advisory Board.
The
U.S. market for veterinary services is highly fragmented. According to the American Veterinary Medical Association, there were
more than 51,000 veterinarians practicing at the end of 2009. The principal factors in a pet owner’s decision as to which
veterinarian to use include convenient location and hours, personal recommendations, reasonable fees and quality of care. In order
to be competitive in the animal healthcare industry, we intend to direct our marketing efforts related to clinics, if and when
they may be acquired, toward increasing the number of annual visits from existing clients through customer education efforts and
toward attracting new clients through local print advertising campaigns.
Sources
and availability of raw materials and the names of principal suppliers
The
supplies and materials required to conduct our operations are available through a wide variety of sources and may be obtained
through a wide variety of sources.
Patents,
trademarks, licenses, franchises, concessions, royalty agreements or labor contracts, including duration
Entest
has not been granted any patents. Entest is not currently party to any royalty agreements. Entest is not party to any binding
labor contracts.
Need
for any government approval of principal products or services, effect of existing or probable governmental regulations on the
business
ImenVax™
I and ImenVax™ II are Veterinary Biologics. The U.S. Department of Agriculture (USDA) is authorized under the 1913 Virus-Serum-Toxin
Act to ensure that all veterinary biologics produced in, or imported into, the United States are not worthless, contaminated,
dangerous, or harmful. The Veterinary Biologics Program of the USDA's Animal and Plant Health Inspection Service (“APHIS”)
oversees the veterinary biologics industry in the United States.
Domestic
manufacturers of veterinary biologics, for domestic use or for export, are required to possess a valid U.S. Veterinary Biologics
Establishment License and an individual U.S. Veterinary Biologics Product License for each product produced for sale.
Prior
to being granted a U.S. Veterinary Biologics Product License, the applicant must submit detailed information including test reports
and research data sufficient to establish purity, safety, potency and efficacy of the product, an Outline of Production, and information
regarding labeling and facilities that are to be used in preparation.
Prior
to being granted a U.S. Veterinary Biologic Establishment License, the applicant must submit detailed information regarding the
facilities and the qualifications of key personnel and must submit to an inspection of the facilities by the Center for Veterinary
Biologics, a division of the USDA. To qualify for an establishment license, an applicant also must qualify for at least one product
license.
In
the event that a veterinary clinic or clinics can be acquired, the Company plans to attempt to distribute ImenVax™ I prior
to licensure under the exemption provided by 9 CFR 107.1, which exempts a veterinary biologic from Federal regulation if the product
was manufactured by veterinarians AND intended solely for use with their clients' animals under a veterinarian-client-patient
(VCP) relationship.
ENT-576™
can be considered a “combination product” whose primary mode of action is through animal stem cells (a veterinary
biologic) It is intended that the Company will obtain a U.S. Veterinary Biologics Establishment License and a U.S. Veterinary
Biologics Product License from the U.S. Department of Agriculture. ENT-576™ can also be administered without license if
administered in accordance with the safe harbor provided by 9 CFR 107.1.
ImenVax™
III can be considered a combination product whose primary mode of action is generated through trophoblasts derived from human
placental tissue. Entest will be required to obtain approval from the US Food and Drug Administration (FDA) in order to market
ImenVax™ III. Entest will apply for an Investigational New Animal Drug exemption (INAD) in order that the product may be
shipped for testing and trials and will submit a New Animal Drug Application for ImenVax™ III.
The
practice of veterinary medicine is primarily subject to State regulation. The Company will be required to comply with the statutes
rules and regulations of the State in which an acquired veterinary clinic is located. Within the State of California, where the
Company is focusing its acquisition efforts , the practice of veterinary medicine is primarily governed pursuant to
The California Veterinary Medicine Practice Act (CA Bus.& Prof. Code § 4800 et seq.).
Amount
spent during the last fiscal year on research and development activities
During
the fiscal year ended August 31, 2014 we expended $0 on research and development activities.
Costs
and effects of compliance with environmental laws (federal, state and local);
Entest
has not incurred any unusual or significant costs to remain in compliance with any environmental laws and does not expect
to incur any unusual or significant costs to remain in compliance with any environmental laws in the foreseeable future.
Number
of total employees and number of full-time employees
As
of November 21, 2014, Entest has 1 employee of which 1 is full time.
Item
2. Properties
On
November 1, 2011, the Company entered into an agreement to lease approximately 2,320 square feet of office space beginning December
1, 2011 for a period of five years.
Rent
to be charged to the Company pursuant to the lease is as follows:
$2,996
per month for the period beginning December 1, 2011 and ending November 30, 2012
$3,116
per month for the period beginning December 1, 2012 and ending November 30, 2013
$3,241
per month for the period beginning December 1, 2013 and ending November 30, 2014
$3,371
per month for the period beginning December 1, 2014 and ending November 30, 2015
$3,506
per month for the period beginning December 1, 2015 and ending November 30, 2016
This
property is utilized as office space. The Company believes that the foregoing property is adequate to meet its current needs.
While it is anticipated that the Company will require access to laboratory facilities in the future, the Company believes that
access to such facilities are available from a variety of sources.
Item
3. Legal Proceedings
On
May 24, 2012, a Complaint (“Complaint”) was filed in the U.S. Bankruptcy Court for the District of Oregon against
the Company by Titterington Veterinary Services Inc. (“TVS”). The Complaint is an adversary proceeding filed by TVS
arising from TVS’s bankruptcy case currently pending in U.S. Bankruptcy Court for the District of Oregon. The Complaint
alleges Breach of Contract resulting from the Company’s alleged failure to pay certain expenses the Company was required
to pay pursuant to an agreement with TVS, Dr. Ronald Titterington, DVM and Dr. Kathy Snell, DVM (“TVS Agreement”).
TVS is seeking a judgment and money award against the Company in an amount to be proven at trial which TVS estimates in the Complaint
to be up to $50,000. TVS is also seeking a judgment and order against the Company to provide an accounting of all revenues received
by the Company pursuant to the TVS Agreement, all expenses paid, unpaid, and due and owing pursuant to the TVS Agreement as well
as a revenue share which TVS claims is due them pursuant to the TVS Agreement. TVS is also seeking a judgment requiring the Company
to turn over a sum of money equal to expenses the Company was obligated to pay pursuant to the TVS Agreement. TVS is also seeking
attorney’s fees and expenses. The Company believes that the allegations in the complaint are without merit and intends to
vigorously defend its interests in this matter. At this time, it is not possible to predict the ultimate outcome of these matters
and an outcome unfavorable to the Company may have a material adverse effect on the Company. On September 19, 2012 the Plaintiff’s
Claim for Relief for turnover and an accounting under 11 U.S.C. § 542 and the Plaintiff's Claim for Relief for attorney fees
were dismissed with prejudice and , as per the claim of breach of contract, the proceeding was transferred to the United States
Bankruptcy Court for the District of Southern California for all further proceedings. On September 19, 2012 the Plaintiff’s
Claim for Relief for turnover and an accounting under 11 U.S.C. § 542 and the Plaintiff's Claim for Relief for attorney fees
were dismissed with prejudice and , as per the claim of breach of contract, the proceeding was transferred to the United States
Bankruptcy Court for the District of Southern California for all further proceedings. This Complaint was dismissed in its entirety
on June 30, 2014.
There
were no other legal proceedings against the Company with respect to matters arising in the ordinary course of business. The Company
is not involved in any other litigation either as plaintiffs or defendants, and has no knowledge of any threatened or pending
litigation against the Company.
Item
4. Submission of Matters to a Vote of Security Holders
No
matter was submitted during the fourth quarter of the fiscal year covered by this report to a vote of security holders, through
the solicitation of proxies or otherwise.
PART
II
Item
5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
The
Company’s common stock is a "penny stock," as defined in Rule 3a51-1 under the Exchange Act. The penny stock
rules require a broker-dealer, prior to a transaction in a penny stock not otherwise exempt from the rules, to deliver a standardized
risk disclosure document that provides information about penny stocks and the nature and level of risks in the penny stock market.
The broker-dealer also must provide the customer with current bid and offer quotations for the penny stock, the compensation of
the broker-dealer and its sales person in the transaction, and monthly account statements showing the market value of each penny
stock held in the customer's account. In addition, the penny stock rules require that the broker-dealer, not otherwise exempt
from such rules, must make a special written determination that the penny stock is suitable for the purchaser and receive the
purchaser's written agreement to the transaction. These disclosure rules have the effect of reducing the level of trading activity
in the secondary market for a stock that becomes subject to the penny stock rules. So long as the common stock of the Company
is subject to the penny stock rules, it may be more difficult to sell common stock of the Company.
The
Company’s authorized capital stock consists of 6,000,000,000 shares of common stock with a par value $0.0001, and 5,000,000
shares of preferred stock with a par value $0.0001 per share (of which 100,000 are designated as Series AA Preferred Stock, 4,400,000
are designated as Series B Preferred Stock and 300,000 are designated as Series AAA Preferred Stock) and 200,000 shares authorized
of Non Voting Convertible Preferred Stock, par value $1.00 As of November 21, 2014 the Company had 2,405,570, 642
common shares outstanding, 3, 201,397 Series B Preferred shares outstanding , 100,000 Series AA preferred shares outstanding and
80,000 Series AAA preferred shares outstanding .
(a)
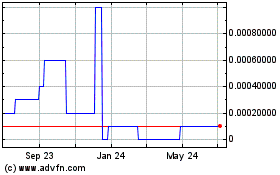
Our common stock is traded on the OTC Pink Tier of OTC Markets under the symbol "ENTB”. Below is the range of
high and low bid information for our common equity for each quarter within the last two fiscal years. These quotations reflect
inter-dealer prices, without retail mark-up, mark-down or commission and may not represent actual transactions.
| September
1, 2013 to August 31, 2014 |
|
High |
|
|
Low |
|
| First
Quarter |
|
$ |
0.0014 |
|
|
$ |
0.0002 |
|
| Second
Quarter |
|
|
0.0009 |
|
|
|
0.0003 |
|
| Third
Quarter |
|
|
0.0016 |
|
|
|
0.0004 |
|
| Fourth
Quarter |
|
$ |
0.0007 |
|
|
$ |
0.0003 |
|
| September
1, 2012 to August 31, 2013 |
|
High |
|
|
Low |
|
| First
Quarter |
|
$ |
0.0053 |
|
|
$ |
0.0007 |
|
| Second
Quarter |
|
|
0.0073 |
|
|
|
0.0005 |
|
| Third
Quarter |
|
|
0.0064 |
|
|
|
0.0016 |
|
| Fourth
Quarter |
|
$ |
0.0012 |
|
|
$ |
0.0030 |
|
Holders
As
of August 31, 2014 there were approximately 341 holders of our Common Stock.
Dividends
No
cash dividends were paid during the fiscal year ending August 31, 2014. We do not expect to declare cash dividends in the immediate
future.
Recent
Sales of Unregistered Securities
On
September 4, 2013 The Company issued 27,887,324 of its common shares (“Shares) in satisfaction of $19,800 of convertible
notes payable.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
September 16, 2013 The Company issued 28,000,000 of its common shares (“Shares”) in satisfaction of $12,600
of convertible notes payable.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
September 20, 2013 The Company issued 20,820,513 of its common shares (“Shares”) in satisfaction of $5,600 of convertible
notes payable and $2,520 of accrued interest on convertible notes payable.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
September 23, 2013 The Company issued 39,325,397 of its common shares (“Shares”) in conversion of 24,775
of the Company’s Non Voting Convertible Preferred Stock
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
September 24, 2013 The Company issued 27,9487,18 of its common shares (“Shares”) in satisfaction of $10,900
of convertible notes payable.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
September 27, 2013 The Company issued 27,9487,18 of its common shares (“Shares”) in satisfaction of $10,900 of convertible
notes payable.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
September 30, 2013 The Company issued 60,000,000 of its common shares (“Shares”) in satisfaction of $13,550 of notes
payable.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
October 2, 2013 The Company issued 24,848,485 of its common shares (“Shares”) in satisfaction of $8,200 of convertible
notes payable.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
October 8, 2013 The Company issued 27,727,273 of its common shares (“Shares”) in satisfaction of $6,100 of convertible
notes payable.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
October 9, 2013 The Company issued 28,000,000 of its common shares (“Shares”) in satisfaction of $5,600 of convertible
notes payable.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
October 10, 2013 The Company issued 27,777,778 of its common shares (“Shares”) in satisfaction of $5,000 of convertible
notes payable.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
October 14, 2013 The Company issued 60,000,000 of its common shares (“Shares”) in satisfaction of $13,550 of notes
payable.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
October 15, 2013 The Company issued 27,777,778 of its common shares (“Shares”) in satisfaction of $5,000 of convertible
notes payable.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
October 18, 2013 The Company issued 20,652,000 of its common shares (“Shares”) in satisfaction of $3,300 of convertible
notes payable.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
October 22, 2013 The Company issued 55,000,000 of its common shares (“Shares”) in satisfaction of $6,600 of convertible
notes payable.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
October 24, 2013 The Company issued 27,500,000 of its common shares (“Shares”) in satisfaction of $1,400 of convertible
notes payable and $1,900 of accrued interest on convertible notes payable.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
November 23, 2013 The Company issued 60,000,000 of its common shares (“Shares”) in satisfaction of $13,550 of notes
payable.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
January 20, 2014 the Company issued to David Koos, the Company’s Chairman and CEO, 95,000 shares of the Company’s
Series AA Preferred Stock (“Shares”) in satisfaction of $10,000 of salary accrued but unpaid owed to David Koos.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
January 24, 2014 the Company issued to David Koos, the Company’s Chairman and CEO, 1,000,000 shares of the Company’s
Series B Preferred Stock (“Shares”) in satisfaction of $1,000 of salary accrued but unpaid owed to David Koos.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
January 24, 2014 the Company issued to David Koos, the Company’s Chairman and CEO, 15,000,000 shares of the Company’s
Common Stock (“Shares”) in satisfaction of $15,000 of principal indebtedness owed to David Koos.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
January 28, 2014 the Company issued 115,000,000 shares of the Company’s Common Stock (“Shares”) in satisfaction
of $10,506 of principal indebtedness.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
March 3, 2014 the Company issued 125,000,000 shares of the Company’s Common Stock (“Shares”) in satisfaction
of $12,500 of principal indebtedness.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
March 13, 2014 the Company issued 140,000,000 shares of the Company’s Common Stock (“Shares”) in satisfaction
of $14,000 of principal indebtedness.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
March 28, 2014 the Company issued 155,000,000 shares of the Company’s Common Stock (“Shares”) in satisfaction
of $15,500 of principal indebtedness
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
April 22, 2014 the Company issued 170,000,000 shares of the Company’s Common Stock ( Shares) in satisfaction of $17,000
of principal indebtedness
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
May 16, 2014 the Company issued 125,000,000 shares of the Company’s Common Stock (“Shares”) in satisfaction
of $12,500 of principal indebtedness.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares.
On
May 22, 2014 the Company issued 80,000 shares of Series AAA Preferred Stock (“Shares”) to David R. Koos, the Company’s
Chairman, President and CEO as consideration for $10,000 of salary accrued and unpaid owed to David R. Koos by the Company.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares. A legend was placed on the certificate that evidences the Shares stating that the Shares have not
been registered under the Act and setting forth or referring to the restrictions on transferability and sale of the Shares.
On
August 7, 2014 the Company issued 195,000,000 shares of the Company’s Common Stock(“Shares”) in satisfaction
of $19,500 of principal indebtedness.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares. A legend was placed on the certificate that evidences the Shares stating that the Shares have not
been registered under the Act and setting forth or referring to the restrictions on transferability and sale of the Shares.
On
August 11, 2014 the Company issued 12,500,000 shares of the Company’s Common Stock (“Shares”) as compensation
to an employee.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares. A legend was placed on the certificate that evidences the Shares stating that the Shares have not
been registered under the Act and setting forth or referring to the restrictions on transferability and sale of the Shares.
On
October 22, 2014 the Company issued 200,000,000 shares of the Company’s Common Stock(“Shares”) in satisfaction
of $20,000 of principal indebtedness.
The
Shares were issued pursuant to Section 4(a) (2) of the Securities Act of 1933, as amended. No underwriters were retained to serve
as placement agents for the sale. The Shares were sold directly through our management. No commission or other consideration was
paid in connection with the sale of the Shares. There was no advertisement or general solicitation made in connection with this
Offer and Sale of Shares. A legend was placed on the certificate that evidences the Shares stating that the Shares have not
been registered under the Act and setting forth or referring to the restrictions on transferability and sale of the Shares.
Item
6. Selected Financial Data
As
we are a “smaller reporting company” as defined by Rule 229.10(f)(1), we are not required to provide the information
required by this Item.
Item
7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
As
of August 31, 2014, we had Cash in the amount of $734 and as of August 31, 2013 we had Cash in the amount of $9,610.
The
decrease in Cash of approximately 92% is attributable to attributable to expenses incurred by the Company in the operation of
its business and payment of its obligations partially offset by funds received by the Company as a result of net borrowings.
As
of August 31, 2014 we had Due from an Affiliate of $0 and as of August 31, 2013 we had Due from an Affiliate of $34,895.
The
decrease in Due from an Affiliate of approximately 100% is attributable to the payment of $34,895 to the Company by Bio Matrix
Scientific Group, Inc during the year ended August 31, 2014.
As
of August 31, 2014 we had Intangible Assets of $0 and as of August 31, 2013 we had Intangible Assets of $270.
The
decrease in Intangible Assets of approximately 100% is attributable to amortization expenses recognized during the year ended
August 31, 2014.
As
of August 31, 2014 we had Accounts Payable of $115,849 and as of August 31, 2013 we had Accounts Payable of $101,615.
The
increase in Accounts Payable of approximately 14% is primarily attributable to increases in outstanding obligations of the Company
incurred in the course of business.
As
of August 31, 2014 we had Notes Payable of $383, 440 and as of August 31, 2013 we had Notes Payable of $272,644
The
increase in Notes Payable of approximately 40% is primarily attributable to :
| |
(a) |
Net
Borrowings during the year ended August 31, 2014 of $5,230 from David Koos, the Company’s CEO |
(b) Borrowings during the year ended August 31, 2014 of $10,422 from Regen Biopharma, Inc., a company of which David Koos is CEO
and Chairman of the Board of Directors
| |
(c) |
Borrowings
during the during the year ended August 31, 2014 from third party lenders of $75,000 |
(d)
The reclassification of $200,000 of accrued salaries to Notes Payable during the quarter ended February 28, 2014
Offset
by:
| (a) | Satisfaction
of $128,606 of principal debt owed to third party lenders through the issuance of equity
securities of the Company during the year ended August 31, 2014 |
| (b) | Satisfaction
of $15,000 of principal debt owed to David Koos through the issuance of equity securities
of the Company during the year ended August 31, 2014 |
| (c) | Forgiveness
by the lender of $5,750 of principal debt during the quarter ended November 20, 2013 |
As
of August 31, 2014 we had Convertible Notes Payable, Net of Discount of $0 and as of August 31, 2013 we had Convertible Notes
Payable, Net of Discount of $101,000
The
decrease in Convertible Notes Payable, Net of Discount of approximately 100% is primarily attributable to conversions of principal
amounts of convertible indebtedness into the common shares of the Company.
As
of August 31, 2014 we had Accrued Expenses of $182,549 and as of August 31, 2013 we had Accrued Expenses of $258,313.
The
decrease in Accrued Expenses of approximately 29% is primarily attributable to:
| (a) | The
reclassification of $200,000 of accrued salaries to Notes Payable during the quarter
ended February 28, 2014 |
| (b) | The
satisfaction of $11,000 of accrued salaries through the issuance of equity securities
during the six months ended February 28,2014 |
Offset
primarily by accrual of salary due and payable to David Koos of $120,000 during the twelve months ended August 31, 2014, accrual
of salary due to an employee of $3,709 during the twelve months ended August 31, 2014 as well as additional accruals of interest
and payroll tax payable during the twelve months ended August 31, 2014.
Material
Changes in Results of Operations
Revenues
from continuing operations were $0 for the fiscal year ended August 31, 2014 and $0 for the fiscal year ended August 31, 2013
. Net losses from continuing operations were$1,416,943 for the fiscal year ended August 31, 2014 and $1,293,583 for the fiscal
year ended August 31, 2013.
The
increase in Net Losses from continuing operations of approximately 9% is primarily attributable to the recognition by the Company
of $600,994 of losses attributable to issuance of stock below fair value, an increase of approximately 646% of expenses attributable
to issuance of common stock below par value, a decrease in other income recognized of 91% when compared to the year ended August
31, 2013 and the recognition of $61,168 during the year ended August 31, 2013 attributable to a Gain on Derecognition of Liabilities
due to the Divestiture of the McDonald Animal Hospital offset primarily by:
| (1) | Lower
Rental , General and Administrative , Interest, and Consulting expenses recognized in
the year ended August 31, 2014 when compared to the same period ended 2013 |
| (2) | Recognition
of a $22,906 one time loss on assets disposed of in the disposition of the McDonald Animal
Hospital during the quarter ended November 30, 2012. |
| (3) | Recognition
of a Goodwill impairment charge of $405,000 attributable to the disposition as of November
30, 2012 of certain assets related to the McDonald Animal Hospital |
| (4) | $140,307
of interest attributable to amortization of Beneficial Conversion Features recognized
during the year ended August 31, 2013 |
| (5) | Gain
on issuance of stock for greater than fair value of $6,000 recognized during the year
ended August 31, 2014 |
| (6) | Penalties
of $63,000 incurred pursuant to terms and conditions of Convertible Debentures recognized
during the year ended August 31, 2013 |
As
of August 31, 2014 we had $734 cash on hand and current liabilities of $689,838 such liabilities consisting of Accounts Payable,
Notes Payable, Amounts due to Others and Accrued Expenses.
We
feel we will not be able to satisfy our cash requirements over the next twelve months and shall be required to seek additional
financing.
We
currently plan to raise additional funds primarily by offering securities for cash and acquiring existing veterinary clinics with
the ability to generate cash flow to fund operations.
There
is no guarantee that we will be able to raise any capital through any type of offerings. We can provide no assurance that we can
acquire veterinary clinics which can generate sufficient cash flow to neither fund our operations nor can any assurance be made
that we can acquire one or more additional veterinary clinics in the near future or at all. We cannot assure that we will be successful
in obtaining additional financing necessary to implement our business plan. We have not received any commitment or expression
of interest from any financing source that has given us any assurance that we will obtain the amount of additional financing in
the future that we currently anticipate. For these and other reasons, we are not able to assure that we will obtain any additional
financing or, if we are successful, that we can obtain any such financing on terms that may be reasonable in light of our current
circumstances.
As
of November 21, 2014 we are not party to any binding agreements which would commit Entest to any material capital expenditures.
Item
7A. Quantitative and Qualitative Disclosures About Market Risk
As
we are a smaller reporting company, as defined by Rule 229.10(f)(1), we are not required to provide the information required
by this Item.
Item
8. Financial Statements and Supplementary Data
SEALE AND BEERS, CPAs
PCAOB REGISTERED AUDITORS
www.sealebeers.com
REPORT
OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To
the Board of Directors and Stockholders of
Entest
Biomedical Inc.
(A
Development Stage Company)
We have audited
the accompanying consolidated balance sheets of Entest Biomedical, Inc. (A Development Stage Company) as of August 31, 2014 and
2013, and the related statements of operations, stockholders’ equity (deficit), and cash flows for each of the years in
the two-year period ended August 31, 2014 and since inception on August 22, 2008 through August 31, 2014. Entest Biomedical, Inc’s
management is responsible for these financial statements. Our responsibility is to express an opinion on these financial statements
based on our audits.
We conducted
our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards
require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material
misstatement. The company is not required to have, nor were we engaged to perform, an audit of its internal control over financial
reporting. Our audit included consideration of internal control over financial reporting as a basis for designing audit procedures
that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the company’s
internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test
basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and
significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that
our audits provide a reasonable basis for our opinion.
In our opinion,
the financial statements referred to above present fairly, in all material respects, the financial position of Entest Biomedical,
Inc. (A Development Stage Company) as of August 31, 2014 and 2013, and the related statements of operations, stockholders’
equity (deficit), and cash flows for each of the years in the two-year period ended August 31, 2014 and since inception on August
22, 2008 through August 31, 2014 in conformity with accounting principles generally accepted in the United States of America.
The accompanying
financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 4 to
the financial statements, the Company has no revenues, has negative working capital at August 31, 2014, has incurred recurring
losses and recurring negative cash flow from operating activities, and has an accumulated deficit which raises substantial doubt
about its ability to continue as a going concern. Management’s plans concerning these matters are also described in Note
4. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Seale & Beers, CPAs
Seale
and Beers, CPAs
Las
Vegas, Nevada
November
21, 2014
8250 W. Charleston Blvd., Suite 100, Las
Vegas, NV 89117 Phone: (888)727-8251 Fax: (888)782-2351
| ENTEST
BIOMEDICAL, INC. |
|
|
| (
A Development Stage Company) |
|
|
| Consolidated
Balance Sheet |
|
|
| |
|
|
| |
|
As
of |
As
of |
| |
|
August
31, 2014 |
August
31, 2013 |
| |
|
|
|
| ASSETS |
|
|
|
| Current
Assets |
|
|
| |
Cash |
734 |
9,610 |
| |
Due
from Affiliate |
0 |
34,895 |
| |
Current
Portion of Prepaid Expenses |
8,000 |
8,000 |
| Total
Current Assets |
8,734 |
52,505 |
| |
|
|
|
| Property
& Equipment (Net of Accumulated Depreciation) |
1,919 |
1,919 |
| Intangible
Assets (Net of Accumulated Amortization) |
0 |
270 |
| |
|
|
|
| |
|
|
|
| |
|
|
|
| TOTAL
ASSETS |
10,653 |
54,694 |
| |
|
|
|
| LIABILITES
AND STOCKHOLDERS' EQUITY |
|
|
| Current
Liabilities |
|
|
| |
Accounts
Payable |
115,849 |
101,615 |
| |
Notes
Payable |
383,440 |
272,644 |
| |
Convertible
notes payable, net of discount |
|
101,000 |
| |
Due
to Other |
8,000 |
8,000 |
| |
Accrued
Expenses |
182,549 |
258,313 |
| Total
Current Liabilities |
687,838 |
741,572 |
| |
|
|
|
| |
|
|
|
| TOTAL
LIABILITIES |
687,838 |
741,572 |
| |
|
|
|
| STOCKHOLDERS'
EQUITY |
|
|
| Common
Stock, authorized 6,000,000,000 shares; |
|
|
| |
issued
and outstanding 561,856,768 (par value $0.001) shares and 2,205,570,752(par value $0.0001) |
|
|
| |
as
of August 31, 2013 and 2014 respectively |
219,657 |
561,855 |
| Preferred
Stock ,par value $0.0001 5,000,000 shares authorized, |
|
|
| |
0
shares issued and outstanding as of August 31,2013 and August 31, 2014 |
|
|
| |
Series
AA Preferred Stock, 100,000 shares authorized, |
|
|
| |
5,000
shares, par value $0.001, issued and outstanding at August 31, 2013 and |
|
|
| |
and
100,000 shares (par value $0.0001) as of August 31, 2014 |
10 |
5 |
| |
Series
B Preferred |
421 |
3,201 |
| |
4,400,000
shares authorized, 3,201,397 ( par value $0.001) |
|
|
| |
issued
and outstanding as of August 31, 2013 and 4,201, 397 (par value $0.0001) |
|
|
| |
issued
and outstanding as of August 31, 2014 |
|
|
| |
Series
AAA Preferred, 300,000 shares authorized, $0.0001 par value |
8 |
0 |
| |
0
and 80,000 shares outstanding as of August 31, 2013 and August 31, 2014 respectively |
|
|
| |
NonVoting
Convertible Preferred |
0 |
24,775 |
| |
($1
par value) 200,000 shares authorized, 24,775 and 0 |
|
|
| |
issued
and outstanding as of August 31, 2013 and 2014,respectively |
|
|
| Additional
Paid in Capital |
5,239,692 |
3,441,563 |
| Contributed
Capital |
274,162 |
274,162 |
| Accumulated
Deficit |
(6,413,135) |
(4,992,439) |
| |
|
|
|
| Total
Stockholders' Equity |
(679,185) |
(686,878) |
| |
|
|
|
| TOTAL
LIABILITIES & STOCKHOLDERS' EQUITY |
10,653 |
54,694 |
The Accompanying
Notes are an Integral Part of these Financial Statements.
| ENTEST
BIOMEDICAL INC. | |
| |
| |
|
| (
A Development Stage Company) | |
| |
| |
|
| Consolidated
Statement of Operations | |
| |
| |
|
| | |
Year
ended | |
Year
ended | |
Inception
to |
| | |
August
31,
2014 | |
August
31, 2013 | |
August
31 2014 |
| | |
| |
| |
|
| TOTAL
REVENUE | |
| 0 | | |
| 0 | | |
| 0 | |
| | |
| | | |
| | | |
| | |
| | |
| | | |
| | | |
| | |
| COSTS
AND EXPENSES | |
| | | |
| | | |
| | |
| Research
and Development | |
| 0 | | |
| 0 | | |
| 145,794 | |
| Rent
Costs | |
| 38,517 | | |
| 37,032 | | |
| 247,477 | |
| General
and Administrative | |
| 265,566 | | |
| 514,310 | | |
| 2,233,043 | |
| Incorporation
Costs | |
| | | |
| | | |
| 408 | |
| Consultant's
Expenses | |
| 76,032 | | |
| 111,655 | | |
| 926,098 | |
| Miscellaneous
Expenses | |
| | | |
| | | |
| 78 | |
| Total
Costs and Expenses | |
| 380,115 | | |
| 662,887 | | |
| 3,552,898 | |
| | |
| | | |
| | | |
| | |
| OPERATING
LOSS | |
| (380,115 | ) | |
| (662,997 | ) | |
| (3,552,898 | ) |
| | |
| | | |
| | | |
| | |
| OTHER
INCOME AND EXPENSE | |
| | | |
| | | |
| | |
| Other
Income | |
| 5,700 | | |
| 70,475 | | |
| 171,223 | |
| Gain
on issuance of stock at above fair value | |
| 6,000 | | |
| | | |
| 6,000 | |
| Penalties
on Convertible Debentures | |
| | | |
| (63,000 | ) | |
| (63,000 | ) |
| Loss
on derecognition of assets due to divestiture | |
| | | |
| (22,906 | ) | |
| (22,906 | ) |
| Loss
on issuance of stock below fair value | |
| (600,994 | ) | |
| | | |
| (600,994 | ) |
| Loss
on Early Extinguishment of Debt | |
| | | |
| | | |
| (18,647 | ) |
| Income
generated from revenue | |
| | | |
| | | |
| 187,699 | |
| Share
Agreement | |
| | | |
| | | |
| | |
| Expenses
incurred from Revenue | |
| | | |
| | | |
| | |
| Share
Agreement | |
| | | |
| | | |
| (145,362 | ) |
| Loss
on Impairment of intangible | |
| | | |
| | | |
| (683,333 | ) |
| assets | |
| | | |
| | | |
| | |
| Interest
Expense | |
| (27,165 | ) | |
| (41,762 | ) | |
| (129,806 | ) |
| Interest
Expense attributable to | |
| | | |
| | | |
| | |
| amortization
of discount | |
| | | |
| (140,307 | ) | |
| (537,215 | ) |
| Loss
on Impairment of Goodwill | |
| | | |
| (405,000 | ) | |
| (405,000 | ) |
| Gain
on derecognition of Liabilities due to | |
| | | |
| | | |
| | |
| Divestiture | |
| | | |
| 61,168 | | |
| 61,168 | |
| Expense
attributable to issuance of common shares | |
| | | |
| | | |
| | |
| below
par value | |
| (420,369 | ) | |
| (56,343 | ) | |
| (476,712 | ) |
| Expense
attributable to issuance | |
| | | |
| | | |
| | |
| of
shares pursuant to contractual | |
| | | |
| | | |
| | |
| obligation | |
| | | |
| (32,911 | ) | |
| (56,779 | ) |
| Expense
attributable to issuance of | |
| | | |
| | | |
| | |
| Non
Voting Convertible Preferred | |
| | | |
| | | |
| | |
| Shares
in connection with Stock | |
| | | |
| | | |
| | |
| Purchase
Agreement | |
| | | |
| | | |
| (75,000 | ) |
| TOTAL
OTHER INCOME AND EXPENSE | |
| (1,036,828 | ) | |
| (630,586 | ) | |
| (2,788,664 | ) |
| | |
| | | |
| | | |
| | |
| LOSS
BEFORE INCOME TAXES | |
| (1,416,943 | ) | |
| (1,293,583 | ) | |
| (6,341,562 | ) |
| income
Taxes | |
| 0 | | |
| 0 | | |
| 0 | |
| | |
| | | |
| | | |
| | |
| NET
LOSS | |
| (1,416,943 | ) | |
| (1,293,583 | ) | |
| (6,341,562 | ) |
| Beneficial
conversion feature | |
| | | |
| | | |
| | |
| attributable
to issuance of NonVoting | |
| | | |
| | | |
| | |
| Preferred
Stock | |
| | | |
| | | |
| (32,142 | ) |
| | |
| | | |
| | | |
| | |
| NET
LOSS from continuing | |
| | | |
| | | |
| | |
| Operations | |
| (1,416,943 | ) | |
| (1,293,583 | ) | |
| (6,373,704 | ) |
| | |
| | | |
| | | |
| | |
| Net
Income (Loss) from discontinued operations | |
| (3,753 | ) | |
| (34,554 | ) | |
| (39,431 | ) |
| | |
| | | |
| | | |
| | |
| NET
LOSS available to common shareholders | |
| (1,420,696 | ) | |
| (1,328,137 | ) | |
| (6,413,135 | ) |
| | |
| | | |
| | | |
| | |
| BASIC
AND DILUTED EARNINGS | |
| | | |
| | | |
| | |
| (LOSS)
PER SHARE FROM CONTINUING OPERATIONS | |
| (0.001 | ) | |
| (0.003 | ) | |
| | |
| | |
| | | |
| | | |
| | |
| BASIC
AND DILUTED EARNINGS | |
| | | |
| | | |
| | |
| (LOSS)
PER SHARE FROM DISCONTINUED OPERATIONS | |
| (0.000 | ) | |
| 0.000 | | |
| | |
| | |
| | | |
| | | |
| | |
| | |
| | | |
| | | |
| | |
| WEIGHTED
AVERAGE | |
| | | |
| | | |
| | |
| NUMBER
OF COMMON | |
| | | |
| | | |
| | |
| SHARES
OUTSTANDING | |
| 1,470,725,875.0 | | |
| 458,310,240 | | |
| | |
| | |
| | | |
| | | |
| | |
| | |
| | | |
| | | |
| | |
The
Accompanying Notes are an Integral Part of these Financial Statements.
| Entest
BioMedical, Inc. |
| Consolidated
Statement of Stockholders' Equity |
| From
inception to August 31, 2014 |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
Common
|
|
Series
AA Preferred |
Series
AAA Preferred |
Series
B Preferred |
Non
Voting Convertible Preferred |
|
Additional
Paid-in |
|
Contributed
|
|
Accumulated
Deficit during the Development |
|
|
| |
Shares
|
|
Amount
|
|
Shares
|
|
Amount
|
Shares
|
|
Amount
|
Shares
|
Amount
|
Shares
|
Amount
|
|
Capital
|
|
Capital
|
|
Stage |
|
Total |
| Beginning
balances Aug. 22, 2008 |
-
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
-
|
|
-
|
|
0
|
|
0
|
| Shares
issued to parent August 23, 2008 |
1,500
|
|
408
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
408
|
| Net
Loss August 22, 2008 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| through
August 31, 2008 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(408) |
|
(408) |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Balances
August 31, 2008 |
1,500
|
|
408
|
|
|
|
|
|
|
|
|
|
|
|
|
-
|
|
-
|
|
(408) |
|
0
|
| Recapitalization
in connection with |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| reverse
acquisition July 10, 2009 |
(1,500) |
|
(408) |
|
|
|
|
|
|
|
|
|
|
|
|
408
|
|
0 |
|
|
|
0
|
| |
10,000,000
|
|
10,000
|
|
|
|
|
|
|
|
|
|
|
|
|
(10,000) |
|
0 |
|
|
|
0
|
| Common
Shares issued in Reverse |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Acquisition
July 10, 2009 |
4,000,000
|
|
4,000
|
|
|
|
|
|
|
|
|
|
|
|
|
(4,000) |
|
0 |
|
|
|
0
|
| Increases
in Contributed Capital |
-
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
-
|
|
485 |
|
|
|
485
|
| Common
Shares issued for Cash August 3, 2009 |
1,000,000
|
|
1,000
|
|
|
|
|
|
|
|
|
|
|
|
|
99,000
|
|
0 |
|
|
|
100,000
|
| Restricted
Stock Award issued to |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| employee
August 3, 2009 |
2,000,000
|
|
2,000
|
|
|
|
|
|
|
|
|
|
|
|
|
(2,000) |
|
0 |
|
|
|
0
|
| Restricted
Stock Award |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| compensation
expense for the |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| year
ended August 31, 2009 |
-
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
24,725
|
|
0 |
|
|
|
24,725
|
| Common
Stock issued to |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| consultant
August 31, 2009 |
50,000
|
|
50
|
|
|
|
|
|
|
|
|
|
|
|
|
199,950
|
|
0 |
|
|
|
200,000
|
| Net
Loss year ended Aug. 31, 2009 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(245,515) |
|
(245,515) |
| Balance
August 31 2009 |
17,050,000
|
|
17,050
|
|
|
|
|
|
|
|
|
|
|
|
|
308,083
|
|
485 |
|
(245,923) |
|
79,695
|
| Common
Shares issued for Cash September 10,2009 |
500,000
|
|
500
|
|
|
|
|
|
|
|
|
|
|
|
|
49,500
|
|
-
|
|
-
|
|
50,000
|
| Restricted
Stock Award |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| compensation
expense for the |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 3
months ended Nov. 30, 2009 |
-
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
98,916
|
|
-
|
|
-
|
|
98,916
|
| Common
Stock as Compensation October 5, 2009 |
3,040
|
|
3
|
|
|
|
|
|
|
|
|
|
|
|
|
4,557
|
|
-
|
|
-
|
|
4,560
|
| Increases
in Contributed Capital |
-
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
-
|
|
4,263
|
|
-
|
|
4,263
|
| Restricted
Stock Award |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| compensation
expense for the |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 3
months ended February 28, 2010 |
-
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
76,359
|
|
-
|
|
-
|
|
76,359
|
| Increases
in Contributed Capital |
-
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
-
|
|
10,356
|
|
-
|
|
10,356
|
| Net
Loss year ended Aug. 31, 2010 |
-
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
-
|
|
-
|
|
(544,891) |
|
(544,891) |
| Balance
August 31 2010 |
17,553,040
|
|
17,553
|
|
|
|
|
|
|
|
|
|
|
|
|
537,415
|
|
15104 |
|
(790,814) |
|
(220,742) |
| Common
Shares issued for Cash October 2 , 2010 |
2,000,000
|
|
2,000
|
|
|
|
|
|
|
|
|
|
|
|
|
98,000
|
|
-
|
|
-
|
|
100,000
|
| Common
Shares issued for Cash January 6 2011 |
200,000
|
|
200
|
|
|
|
|
|
|
|
|
|
|
|
|
99,800
|
|
|
|
|
|
100,000
|
| Common
Shares issued for cash january 4 2011 |
17,600
|
|
17
|
|
|
|
|
|
|
|
|
|
|
|
|
17,583
|
|
|
|
|
|
17,600
|
| Common
Shares issued for Debt January 3, 2011 |
38,712
|
|
37
|
|
|
|
|
|
|
|
|
|
|
|
|
39,952
|
|
-
|
|
-
|
|
39,989
|
| Common
Shares issued for Debt April 4, 2011 |
600,000
|
|
600
|
|
|
|
|
|
|
|
|
|
|
|
|
250,578
|
|
|
|
|
|
251,178
|
| Common
Shares issued for Debt August 29 2011 |
143,000
|
|
143
|
|
|
|
|
|
|
|
|
|
|
|
|
49,857
|
|
|
|
|
|
50,000
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Common
Shares issued to Consultants 11/ 16/ 2010 |
45,000
|
|
45
|
|
|
|
|
|
|
|
|
|
|
|
|
53,055
|
|
-
|
|
-
|
|
53,100
|
| Common
Shares issued to Consultants 11/ 30/ 2010 |
10,000
|
|
10
|
|
|
|
|
|
|
|
|
|
|
|
|
9,290
|
|
|
|
|
|
9,300
|
| Common
Shares issued to Consultants 2/4/2011 |
10,000
|
|
10
|
|
|
|
|
|
|
|
|
|
|
|
|
41,990
|
|
|
|
|
|
42,000
|
| Common
Shares issued to Consultants 1/4/2011 |
9,383
|
|
9
|
|
|
|
|
|
|
|
|
|
|
|
|
17,632
|
|
|
|
|
|
17,641
|
| Common
Stock issued to employees January 3, 2011 |
38,000
|
|
38
|
|
|
|
|
|
|
|
|
|
|
|
|
71,402
|
|
-
|
|
-
|
|
71,440
|
| Common
Stock issued to employees January 14, 2011 |
102,766
|
|
103
|
|
|
|
|
|
|
|
|
|
|
|
|
193,097
|
|
|
|
|
|
193,200
|
| Common
Stock issued to employees February4, 2011 |
5,000
|
|
5
|
|
|
|
|
|
|
|
|
|
|
|
|
20,595
|
|
|
|
|
|
20,600
|
| Common
Stock issued to employees march 25, 2011 |
7,000
|
|
7
|
|
|
|
|
|
|
|
|
|
|
|
|
14,923
|
|
|
|
|
|
14,930
|
| Common
Stock issued to employees August 11, 2011 |
11,000
|
|
11
|
|
|
|
|
|
|
|
|
|
|
|
|
3,218
|
|
|
|
|
|
3,229
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Restricted
Stock Awards issued to employee 4/ 1 /2011 |
50,000
|
|
50
|
|
|
|
|
|
|
|
|
|
|
|
|
(50) |
|
-
|
|
-
|
|
-
|
| Restricted
Stock Award Compensation |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| expense
recognized |
-
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
10,000
|
|
-
|
|
-
|
|
10,000
|
| Discount
on Convertible Debt recognized |
-
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
88,998
|
|
-
|
|
-
|
|
88,998
|
| Increases
in Contributed capital |
-
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
-
|
|
259,058
|
|
-
|
|
259,058
|
| Preferred
Stock issued for Accrued compensation June 9, 2011 |
|
|
|
|
5,000
|
|
5
|
|
|
|
|
|
|
|
|
1,995
|
|
|
|
|
|
2,000
|
| Net
Loss Year ended August 31, 2011 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(954,398) |
|
(954,398) |
| Discontinued
operations |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2,615
|
|
2,615
|
| Balance
August 31 2011 |
20,840,501
|
|
20,838
|
|
5000 |
|
5 |
|
|
|
|
|
|
|
|
1,619,330
|
|
274162 |
|
(1,742,597) |
|
171,738
|
| Common
Shares issued for services 10/07/2011 |
50,474
|
|
50
|
|
|
|
|
|
|
|
|
|
|
|
|
17,950
|
|
|
|
|
|
18,000
|
| Common
Shares issued for services 11/22/2011 |
18,499
|
|
18
|
|
|
|
|
|
|
|
|
|
|
|
|
4,565
|
|
|
|
|
|
4,583
|
| Common
Shares issued for services 1/10/2012 |
27,499
|
|
27
|
|
|
|
|
|
|
|
|
|
|
|
|
8,241
|
|
|
|
|
|
8,268
|
| Common
Shares issued for services 2/26/2012 |
175,528
|
|
173
|
|
|
|
|
|
|
|
|
|
|
|
|
52,225
|
|
|
|
|
|
52,398
|
| Common
Shares issued for services 3/14/2012 |
3,000,000
|
|
3,000
|
|
|
|
|
|
|
|
|
|
|
|
|
85,500
|
|
|
|
|
|
88,500
|
| Common
Shares issued for intangible assets 11/11/2011 |
2,500,000
|
|
2,500
|
|
|
|
|
|
|
|
|
|
|
|
|
697,500
|
|
|
|
|
|
700,000
|
| Discount
on Convertible Note Recognized |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
350,915
|
|
|
|
|
|
350,915
|
| Common
Shares issued for debt 11/28/2011 |
134,983
|
|
134
|
|
|
|
|
|
|
|
|
|
|
|
|
11,866
|
|
|
|
|
|
12,000
|
| Common
Shares issued for debt 12/12/2011 |
258,824
|
|
259
|
|
|
|
|
|
|
|
|
|
|
|
|
10,741
|
|
|
|
|
|
11,000
|
| Common
Shares issued for debt 12/19/2011 |
338,983
|
|
339
|
|
|
|
|
|
|
|
|
|
|
|
|
11,661
|
|
|
|
|
|
12,000
|
| Common
Shares issued for debt 1/27/2012 |
227,963
|
|
228
|
|
|
|
|
|
|
|
|
|
|
|
|
7,272
|
|
|
|
|
|
7,500
|
| Common
Shares issued for debt 2/3/2012 |
528,821
|
|
529
|
|
|
|
|
|
|
|
|
|
|
|
|
14,471
|
|
|
|
|
|
15,000
|
| Common
Shares issued for debt 2/16/2012 |
721,154
|
|
721
|
|
|
|
|
|
|
|
|
|
|
|
|
14,279
|
|
|
|
|
|
15,000
|
| Common
Shares issued for debt 3/07/2012 |
2,322,695
|
|
2,323
|
|
|
|
|
|
|
|
|
|
|
|
|
30,427
|
|
|
|
|
|
32,750
|
| Common
Shares issued for debt 3/21/2012 |
1,162,791
|
|
1,163
|
|
|
|
|
|
|
|
|
|
|
|
|
13,837
|
|
|
|
|
|
15,000
|
| Common
Shares issued for debt 3/26/2012 |
1,411,765
|
|
1,412
|
|
|
|
|
|
|
|
|
|
|
|
|
10,588
|
|
|
|
|
|
12,000
|
| Common
Shares issued for debt 3/21/2012 |
723,404
|
|
723
|
|
|
|
|
|
|
|
|
|
|
|
|
9,477
|
|
|
|
|
|
10,200
|
| Common
Shares issued for debt 3/26/2012 |
1,605,737
|
|
1,606
|
|
|
|
|
|
|
|
|
|
|
|
|
11,044
|
|
|
|
|
|
12,650
|
| Common
Shares issued for debt 4/02/2012 |
886,222
|
|
886
|
|
|
|
|
|
|
|
|
|
|
|
|
3,102
|
|
|
|
|
|
3,988
|
| Common
Shares issued for debt 4/03/2012 |
1,590,909
|
|
1,591
|
|
|
|
|
|
|
|
|
|
|
|
|
5,409
|
|
|
|
|
|
7,000
|
| Common
Shares issued for debt 4/02/2012 |
788,889
|
|
789
|
|
|
|
|
|
|
|
|
|
|
|
|
2,761
|
|
|
|
|
|
3,550
|
| Common
Shares issued for debt 4/10/2012 |
1,944,444
|
|
1,944
|
|
|
|
|
|
|
|
|
|
|
|
|
1,556
|
|
|
|
|
|
3,500
|
| Common
Shares issued for debt 4/10/2012 |
3,611,111
|
|
3,611
|
|
|
|
|
|
|
|
|
|
|
|
|
2,889
|
|
|
|
|
|
6,500
|
| Common
Shares issued for debt 4/12/2012 |
4,722,222
|
|
4,722
|
|
|
|
|
|
|
|
|
|
|
|
|
3,778
|
|
|
|
|
|
8,500
|
| Common
Shares issued for debt 4/13/2012 |
8,250,166
|
|
8,250
|
|
|
|
|
|
|
|
|
|
|
|
|
6,600
|
|
|
|
|
|
14,850
|
| Common
Shares issued for debt 4/17/2012 |
5,000,000
|
|
5,000
|
|
|
|
|
|
|
|
|
|
|
|
|
3,000
|
|
|
|
|
|
8,000
|
| Common
Shares issued for debt 4/17/2012 |
6,250,000
|
|
6,250
|
|
|
|
|
|
|
|
|
|
|
|
|
3,750
|
|
|
|
|
|
10,000
|
| Common
Shares issued for debt 4/19/2012 |
2,724,968
|
|
2,725
|
|
|
|
|
|
|
|
|
|
|
|
|
1,575
|
|
|
|
|
|
4,300
|
| Common
Shares issued for debt 4/18/2012 |
1,250,000
|
|
1,250
|
|
|
|
|
|
|
|
|
|
|
|
|
750
|
|
|
|
|
|
2,000
|
| Common
Shares issued for debt 4/24/2012 |
4,005,787
|
|
4,006
|
|
|
|
|
|
|
|
|
|
|
|
|
2,916
|
|
|
|
|
|
6,922
|
| Common
Shares issued for debt 5/01/2012 |
8,000,000
|
|
8,000
|
|
|
|
|
|
|
|
|
|
|
|
|
4,000
|
|
|
|
|
|
12,000
|
| Common
Shares issued for debt 5/03/2012 |
7,142,857
|
|
7,143
|
|
|
|
|
|
|
|
|
|
|
|
|
2,857
|
|
|
|
|
|
10,000
|
| Common
Shares issued for debt 5/07/2012 |
7,692,308
|
|
7,692
|
|
|
|
|
|
|
|
|
|
|
|
|
2,308
|
|
|
|
|
|
10,000
|
| Common
Shares issued for debt 5/09/2012 |
500,000
|
|
500
|
|
|
|
|
|
|
|
|
|
|
|
|
0
|
|
|
|
|
|
500
|
| Common
Shares issued for debt 6/20/2012 |
1,590,562
|
|
1,591
|
|
|
|
|
|
|
|
|
|
|
|
|
6,330
|
|
|
|
|
|
7,921
|
| Common
Shares issued for debt 6/20/2012 |
15,873,016
|
|
15,873
|
|
|
|
|
|
|
|
|
|
|
|
|
44,127
|
|
|
|
|
|
60,000
|
| Common
Shares issued for debt 6/20/2012 |
1,590,562
|
|
1,591
|
|
|
|
|
|
|
|
|
|
|
|
|
6,330
|
|
|
|
|
|
7,921
|
| Common
Shares issued for debt 6/25/2012 |
1,808,172
|
|
1,808
|
|
|
|
|
|
|
|
|
|
|
|
|
4,819
|
|
|
|
|
|
6,627
|
| Common
Shares issued for debt 8/09/2012 |
10,650,777
|
|
10,651
|
|
|
|
|
|
|
|
|
|
|
|
|
2,602
|
|
|
|
|
|
13,253
|
| Common
Shares issued for debt 8/09/2012 |
5,325,603
|
|
5,326
|
|
|
|
|
|
|
|
|
|
|
|
|
1,301
|
|
|
|
|
|
6,627
|
| Common
Shares issued for interest 1/27/2012 |
51,672
|
|
52
|
|
|
|
|
|
|
|
|
|
|
|
|
1,648
|
|
|
|
|
|
1,700
|
| Common
Shares issued for interest 4/10/2012 |
833,333
|
|
833
|
|
|
|
|
|
|
|
|
|
|
|
|
667
|
|
|
|
|
|
1,500
|
| Common
Shares issued for interest 4/18/2012 |
875,000
|
|
875
|
|
|
|
|
|
|
|
|
|
|
|
|
525
|
|
|
|
|
|
1,400
|
| Common
Shares issued for interest 5/09/2012 |
1,300,000
|
|
1,300
|
|
|
|
|
|
|
|
|
|
|
|
|
0
|
|
|
|
|
|
1,300
|
| Cancellation
of Common Shares Issued 1/10/2012 |
(90,000) |
|
(83) |
|
|
|
|
|
|
|
|
|
|
|
|
(86,687) |
|
|
|
|
|
(86,687) |
| Restricted
Stock Award to employees 4/3/2012 |
77,000,000
|
|
77,000
|
|
|
|
|
|
|
|
|
|
|
|
|
(77,000) |
|
|
|
|
|
0
|
| Restricted
Stock Award to consultant 4/3/2012 |
15,000,000
|
|
15,000
|
|
|
|
|
|
|
|
|
|
|
|
|
(15,000) |
|
|
|
|
|
0
|
| Restricted
Stock Award compensation |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Expense
Recognized |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
58,491
|
|
|
|
|
|
58,491
|
| Series
B Preferred Dividend paid 3/06/2012 |
|
|
|
|
|
|
|
|
|
|
3,201,397
|
3,201
|
|
|
|
(3,201) |
|
|
|
|
|
0
|
| Issuance
of Non Voting Convertible |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Preferred
Shares 3/27/2012 |
|
|
|
|
|
|
|
|
|
|
|
|
75,000
|
75,000
|
|
|
|
|
|
|
|
75,000
|
| Recognition
of Beneficial Conversion Feature |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
32,142
|
|
|
|
|
|
32,142
|
| Non
Voting Convertible Preferred Shares |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Shares
issued pursuant to Contractual Obligations 4/11/2012 |
1,029,679
|
|
1,030
|
|
|
|
|
|
|
|
|
|
|
|
|
3,192
|
|
|
|
|
|
4,222
|
| Shares
issued pursuant to Contractual Obligations 4/11/2012 |
1,276,996
|
|
1,277
|
|
|
|
|
|
|
|
|
|
|
|
|
3,957
|
|
|
|
|
|
5,234
|
| Shares
issued pursuant to Contractual Obligations 4/13/2012 |
1,101,420
|
|
1,101
|
|
|
|
|
|
|
|
|
|
|
|
|
2,643
|
|
|
|
|
|
3,745
|
| Shares
issued pursuant to Contractual Obligations 4/13/2012 |
1,329,633
|
|
1,329
|
|
|
|
|
|
|
|
|
|
|
|
|
3,190
|
|
|
|
|
|
4,520
|
| Shares
issued pursuant to Contractual Obligations 8/02/2012 |
1,536,998
|
|
1,537
|
|
|
|
|
|
|
|
|
|
|
|
|
4,611
|
|
|
|
|
|
6,148
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Cancellation
of Restricted Stock Award to Employee |
(15,000,000) |
|
(15,000) |
|
|
|
|
|
|
|
|
|
|
|
|
6,813
|
|
|
|
|
|
(8,187) |
| 8/6/2012 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Net
Loss Year ended August 31, 2012 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1,885,824) |
|
(1,885,824) |
| Beneficial
Conversion Feauture Deemed Dividend |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(32,142) |
|
(32,142) |
| Discontinued
Operations |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(3,739) |
|
(3,739) |
| Balance
August 31 2012 |
223,492,927
|
|
223,493
|
|
5,000
|
|
5
|
|
|
|
3,201,397
|
3,201
|
75,000
|
75,000
|
|
3,030,642
|
|
274,162
|
|
(3,664,302) |
|
(57,799) |
| Common
Shares issued for debt 10/22/2012 |
10,810,811 |
|
10,811 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10,811
|
| Common
Shares issued for debt 10/23/2012 |
10,897,436 |
|
10,897 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10,897
|
| Common
Shares issued for debt 10/24/2012 |
10,833,333 |
|
10,833 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10,833
|
| Common
Shares issued for debt 10/25/2012 |
10,833,333 |
|
10,833 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10,833
|
| Common
Shares issued for debt 10/26/2012 |
10,833,333 |
|
10,833 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10,833
|
| Common
Shares issued for debt 11/06/2012 |
4,642,587 |
|
4,643 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
4,643
|
| Common
Shares issued for debt 10/09/2012 |
23,000,000
|
|
23,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
23,000
|
| Common
Shares issued for debt 10/25/2012 |
31,346,154
|
|
31,346
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
31,346
|
| Common
Shares issued for debt 11/16/2012 |
36,261,905
|
|
36,262
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
36,262
|
| Common
Shares issued for debt 07/08/2012 |
22,727,273
|
|
22,727
|
|
|
|
|
|
|
|
|
|
|
|
|
2,273 |
|
|
|
|
|
25,000
|
| Common
Shares issued for Interest 11/06/2012 |
3,035,894 |
|
3,036 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
3,036
|
| Common
Shares issued for cash 9/4/2012 |
10,000,000 |
|
10,000 |
|
|
|
|
|
|
|
|
|
|
|
|
8,655 |
|
|
|
|
|
18,655 |
| Common
Shares issued for cash 9/10/2012 |
10,000,000
|
|
10,000
|
|
|
|
|
|
|
|
|
|
|
|
|
8,655
|
|
|
|
|
|
18,655
|
| Common
Shares issued for cash 9/17/2012 |
6,802,465
|
|
6,802
|
|
|
|
|
|
|
|
|
|
|
|
|
5,888
|
|
|
|
|
|
12,690
|
| Common
Shares issued for cash 10/02/2012 |
10,837,849 |
|
10,838 |
|
|
|
|
|
|
|
|
|
|
|
|
3,462 |
|
|
|
|
|
14,300 |
| Common
Shares issued for Nonvoting Converible Preferred |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Stock
3/13/2013 |
3,011,583
|
|
3,012
|
|
|
|
|
|
|
|
|
|
(7,800) |
(7,800) |
|
4,788
|
|
|
|
|
|
0
|
| Common
Shares issued for Nonvoting Converible Preferred |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Stock
5/22/2013 |
17,928,571
|
|
17,929
|
|
|
|
|
|
|
|
|
|
(18,825) |
(18,825) |
|
896
|
|
|
|
|
|
0
|
| Common
Shares issued for Nonvoting Converible Preferred |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Stock
2/15/2013 |
30,649,531
|
|
30,649
|
|
|
|
|
|
|
|
|
|
(23,600) |
(23,600) |
|
|
|
|
|
|
|
7,049
|
| Common
Shhares issued as legal settlement 3/12/2013 |
41,000,000
|
|
41,000
|
|
|
|
|
|
|
|
|
|
|
|
|
155,800
|
|
|
|
|
|
196,800
|
| Discount
on Convertible Note recognized |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
63,000
|
|
|
|
|
|
63,000
|
| Discount
on penalty on Convertible Note recognized |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
34,303 |
|
|
|
|
|
34,303
|
| Restricted
Stock Award compensation |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Expense
Recognized |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
123,200 |
|
|
|
|
|
123,200
|
| Common
Shares issued pursuant to |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Contractual
Obligations 11/14/2012 |
26,868,132 |
|
26,868 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
26,868
|
| Common
Shares issued pursuant to |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Contractual
Obligations 12/05/2012 |
6,043,651 |
|
6,043 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
6,043
|
| Net
Loss Year ended August 31, 2013 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1,293,583) |
|
(1,293,583) |
| Discontinued
Operations |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(34,554) |
|
(34,554) |
| Balance
August 31 2013 |
561,856,768
|
|
561,855
|
|
5,000
|
|
5
|
|
|
|
3,201,397
|
3,201
|
24,775
|
24,775
|
|
3,441,563
|
|
274,162
|
|
(4,992,439) |
|
(686,878) |
| Common
Shares Issued for debt 9/4/2013 |
27,887,324 |
|
27,887 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
27,887
|
| Common
Shares issued for Debt 9/16/2013 |
28,000,000
|
|
28,000 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
28,000
|
| Common
Shares issued for Debt 9/20/2013 |
20,820,513 |
|
20821 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
20,821
|
| Common
Shares issued for Nonvoting Convertible Preferred 9/23/2013 |
39,325,397 |
|
39,325 |
|
|
|
|
|
|
|
|
|
(24,775) |
(24,775) |
|
|
|
|
|
|
|
14,550
|
| Common
Shares issued for Debt 9/24/2013 |
27,948,718 |
|
27,949 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
27,949 |
| Common
Shares issued for Debt 9/27/2014 |
27,948,718 |
|
27,949 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
27,949 |
| Common
Shares issued for Debt 9/30/2013 |
60,000,000 |
|
60,000 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
60,000 |
| Common
Shares issued for Debt 10/02/2013 |
24,848,485 |
|
24,848 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
24,848 |
| Common
Shares issued for Debt 10/08/2013 |
27,727,273 |
|
27,727 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
27,727 |
| Common
Shares issued for Debt 10/09/2013 |
28,000,000 |
|
28,000 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
28,000 |
| Common
Shares issued for Debt 10/10/2013 |
27,777,778 |
|
27,778 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
27,778 |
| Common
Shares issued for Debt 10/14/2013 |
60,000,000 |
|
60,000 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
60,000 |
| Common
Shares issued for Debt 10/15/2013 |
27,777,778 |
|
27,778 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
27,778 |
| Common
Shares issued for Debt 10/18/2013 |
20,652,000 |
|
20,652 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
20,652 |
| Common
Shares issued for Debt 10/22/2013 |
27,500,000 |
|
27,500 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
27,500 |
| Common
Shares issued for Debt 10/22/2013 |
27,500,000 |
|
27,500 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
27,500 |
| Common
Shares issued for Debt 10/24/2013 |
27,500,000 |
|
27,500 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
27,500 |
| Common
Shares issued for Debt 11/ 13/2013 |
60,000,000 |
|
60,000 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
60,000 |
| Preferred
Shares issued for Accrued Salaries 1/20/2014 |
|
|
|
|
95,000 |
|
95 |
|
|
|
|
|
|
|
|
9,905 |
|
|
|
|
|
10,000 |
| Reduction
in par value 1/24/2014 |
|
|
(1,038,662) |
|
|
|
(90) |
|
|
|
|
(2,880) |
|
|
|
1,041,632
|
|
|
|
|
|
0
|
| Preferred
Shares issued for Accrued Salaries 1/20/2014 |
|
|
|
|
|
|
|
|
|
|
1,000,000 |
100 |
|
|
|
900 |
|
|
|
|
|
1,000 |
| Common
Shares issued for Debt 1/24/2014 |
15,000,000 |
|
1,500
|
|
|
|
|
|
|
|
|
|
|
|
|
7,500
|
|
|
|
|
|
9,000
|
| Common
Shares issued for debt 1/28/2014 |
115,000,000 |
|
11,500 |
|
|
|
|
|
|
|
|
|
|
|
|
57500 |
|
|
|
|
|
69,000
|
| Common
Shares issued for Debt 3/03/2014 |
125,000,000
|
|
12,500 |
|
|
|
|
|
|
|
|
|
|
|
|
75,000 |
|
|
|
|
|
87,500 |
| Common
Shares issued for Debt 3/13/2014 |
140,000,000
|
|
14,000 |
|
|
|
|
|
|
|
|
|
|
|
|
84,000 |
|
|
|
|
|
98,000 |
| Common
Shares issued for Debt 3/28/2014 |
155,000,000
|
|
15,500 |
|
|
|
|
|
|
|
|
|
|
|
|
124,000 |
|
|
|
|
|
139,500 |
| Common
Shares issued for Debt 4/22/2014 |
170,000,000
|
|
17,000 |
|
|
|
|
|
|
|
|
|
|
|
|
119,000 |
|
|
|
|
|
136,000 |
| Common
Shares issued for Debt 5/16/2014 |
125,000,000
|
|
12,500 |
|
|
|
|
|
|
|
|
|
|
|
|
62,500 |
|
|
|
|
|
75,000 |
| Preferred
Shares issued for Accrued Salaries 5/22/2014 |
|
|
|
|
|
|
|
80,000 |
|
8 |
|
|
|
|
|
9,992
|
|
|
|
|
|
10,000
|
| Common
Shares issued for Debt 8/07/2014 |
195,000,000
|
|
19,500 |
|
|
|
|
|
|
|
|
|
|
|
|
78,000
|
|
|
|
|
|
97,500
|
| Common
shares issued for compensation 8/11/2014 |
12,500,000
|
|
1,250 |
|
|
|
|
|
|
|
|
|
|
|
|
5,000
|
|
|
|
|
|
6,250
|
| Restricted
Stock Expense Recognized |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
123,200 |
|
|
|
|
|
123,200
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Net
Loss Year Ended August 31, 2014 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(1,416,943) |
|
(1,416,943) |
| Discontinued
Operations |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(3,753) |
|
(3,753) |
| Balance
August 31, 2014 |
2,205,570,752
|
|
219,657 |
|
100,000
|
|
10
|
80,000
|
|
8
|
4,201,397
|
421
|
0
|
0
|
|
5,239,692
|
|
274,162 |
|
(6,413,135) |
|
(679,185) |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The Accompanying
Notes are an Integral Part of these Financial Statements.
| ENTEST
BIOMEDICAL, INC. |
|
|
|
| (
A Development Stage Company) |
|
|
|
| Consolidated
Statement of Cash Flows |
|
|
|
| |
|
|
|
|
|
| |
|
|
Year
Ended |
Year
Ended |
Inception
until |
| |
|
|
August
31, 2014 |
August
31, 2013 |
August
31, 2014 |
| |
|
|
|
|
|
| |
OPERATING
ACTIVITIES |
|
|
|
| |
Net
(loss) |
(1,416,943) |
(1,293,583) |
(6,341,562) |
| |
Adjustments |
to
reconcile net loss to net cash used |
|
|
|
| |
by operating |
activities: |
|
|
|
| |
|
Depreciation
Expense |
|
2,073 |
16,125 |
| |
|
Amortization
Expense |
270 |
770 |
2,287 |
| |
|
Stock
issued as Compensation to Employees |
6,250 |
99,200 |
667,530 |
| |
|
Stock
issued as Compensation to Consultants |
|
24,000 |
532,142 |
| |
|
Preferred
Stock issued for accrued compensation |
21,000 |
|
23,000 |
| |
Change
in |
operating
assets and liabilities: |
|
|
|
| |
|
(Increase)
Decrease in Trade Accounts Receivable |
|
2,268 |
|
| |
|
(Increase)
Decrease in Inventory |
|
10,298 |
|
| |
|
(Increase)
Decrease in Employee Receivable |
|
4,349 |
|
| |
|
Increase
(Decrease) in Accounts Payable |
14,234 |
27,040 |
115,848 |
| |
|
(Increase)Decrease
in Due to Other |
|
|
|
| |
|
(Increase)
Decrease in Prepaid Expenses |
|
5,000 |
(8,000) |
| |
|
(Increase)
Decrease in Due from Affiliate |
34,895 |
4,245 |
0 |
| |
|
(Increase)
Decrease in Deposits |
|
1,151 |
|
| |
|
Increase(Decrease)
in amortization of intangibles |
|
|
16,667 |
| |
|
Increase
(Decrease) in Accrued Expenses |
(75,764) |
138,586 |
182,547 |
| |
|
(Increase)Decrease
on gain realized on cancellation |
|
|
|
| |
|
of
stock |
|
|
(94,937) |
| |
|
Increase(Decrease)
in Loss on Impairment |
|
|
|
| |
|
of
Intangible Assets |
|
|
683,333 |
| |
|
|
|
|
|
| |
|
Net
Cash Provided Used in Operating Activities |
(1,416,058) |
(974,603) |
(4,205,020) |
| |
|
|
|
|
|
| |
INVESTING
ACTIVITIES |
|
|
|
| |
|
Purchase/Sale
of Equipment |
|
|
|
| |
|
|
0 |
0 |
(22,884) |
| |
|
Net
cash Provided by (Used in) Investing Activities |
0 |
0 |
(22,884) |
| |
|
|
|
|
|
| |
FINANCING
ACTIVITIES |
|
|
|
| |
|
(Increase)
Decrease in Goodwill from acquisition |
|
405000 |
|
| |
|
Noncash
Loss on derecognition of assets |
|
4,840 |
4,840 |
| |
|
Expenses
incurred resulting from issuance of stock for |
|
|
|
| |
|
less
than par |
420,369 |
56,343 |
476,712 |
| |
|
(increase)
Decrease in gain on issuance of stock for more than fair value |
(6,000) |
|
(6,000) |
| |
|
Increase
(Decrease) in loss on issuance of stock for less than fair value |
600,994 |
|
600,994 |
| |
|
Increase
(Decrease) in Common stock issued for cash |
|
64,300 |
431,900 |
| |
|
(Increase)
Decrease in Intangible Assets (net) |
|
|
(2,299) |
| |
|
Increase
(Decrease) in Common stock issued |
|
|
|
| |
|
for
expenses |
4,420 |
231,411 |
340,654 |
| |
|
Increase
(Decrease) in Due to Affiliate |
|
|
8,000 |
| |
|
Increase
(Decrease) in Due to Shareholder |
|
|
|
| |
|
Increase
(Decrease) in Notes Payable |
267,952 |
116,832 |
1,478,337 |
| |
|
Increase
(Decrease) in Contributed Capital |
|
|
274,161 |
| |
|
Increase(Decrease)
in penalties attributable to Convertible Debt |
|
|
|
| |
|
Increase
(Decrease) in Additional paid in Capital |
123,200 |
97,304 |
660,770 |
| |
|
Net
Cash Provided by Financing Activities |
1,410,935 |
976,030 |
4,268,069 |
| |
DISCONTINUED
OPERATION |
|
|
|
| |
|
Profit(Loss)
from discontinued operations |
(3,753) |
(34,554) |
(39,431) |
| |
|
Net
Increase in Cash |
(8,876) |
(33,127) |
734 |
| |
|
|
|
|
|
| |
|
Cash
at Beginning of Period |
9610 |
42,737 |
0 |
| |
|
|
|
|
|
| |
|
|
|
|
|
| |
|
Cash
at End of Period |
734 |
9,610 |
734 |
| |
|
|
|
|
|
| Supplemental
Disclosure of Noncash investing and financing activities: |
|
|
|
| |
|
|
|
|
|
| |
|
|
|
|
|
| Stock
issued in payment of Debt |
$258,156 |
$126,500 |
$1,144,882 |
| Stock
issued in Acquisition of McDonald Animal Hospital |
|
|
$210,000 |
| Stock
issued pursuant to Titterington Agreement |
|
|
$700,000 |
The Accompanying
Notes are an Integral Part of these Financial Statements.
Entest
BioMedical, Inc.
(A
Development Stage Company)
Notes
to Consolidated Financial Statements
As
of August 31, 2014
NOTE
1. ORGANIZATION AND DESCRIPTION OF BUSINESS
The
Company was incorporated in the State of Nevada on September 24, 2008 as JB Clothing Corporation. Until July 10, 2009, the
Company’s principal business objective was the offering of active/leisure fashion design clothing.
On
July 10, 2009 the Company abandoned its efforts in the field of active/leisure fashion design clothing when it acquired 100% of
the share capital of Entest BioMedical, Inc., a California corporation, (“Entest CA”).
The
Company’s current business consists of the development and commercialization of immunotherapeutic therapies for the veterinary
market as well as the acquisition and operation of veterinary hospitals.
NOTE
2. SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
A.
BASIS OF ACCOUNTING
The
financial statements have been prepared using the basis of accounting generally accepted in the United States of America. Under
this basis of accounting, revenues are recorded as earned and expenses are recorded at the time liabilities are incurred. The
Company has adopted an August 31 fiscal year-end. The Company recognizes revenue from services and product sales when the
following four revenue recognition criteria are met: persuasive evidence of an arrangement exists, delivery has occurred or services
have been rendered, the selling price is fixed or determinable, and collectability is reasonably assured. Product sales and service
revenues are recorded when the products are delivered and title passes to customers. The customer’s credit card is authorized
and charged, or checks/cash are received at the time the services are rendered, thereby providing reasonable assurance of collectability.
B.
PRINCIPLES OF CONSOLIDATION
The
consolidated financial statements include the accounts of Entest CA, the Company’s wholly owned subsidiary. Significant
inter-company transactions have been eliminated.
C.
USE OF ESTIMATES
The
preparation of financial statements in conformity with generally accepted accounting principles requires management to make estimates
and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities
at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual
results could differ from those estimates.
D.
CASH EQUIVALENTS
The
Company considers all highly liquid investments with a maturity of three months or less when purchased to be cash equivalents.
E.
PROPERTY AND EQUIPMENT
As
of August 31, 2014 Property and Equipment consists of $1,919 of Computer equipment. No depreciation expense has been recorded
with regards to this equipment as it has yet to be put into service
F.
FAIR VALUE OF FINANCIAL INSTRUMENTS
Fair
value is the price that would be received for an asset or the exit price that would be paid to transfer a liability in the principal
or most advantageous market in an orderly transaction between market participants on the measurement date. A fair value
hierarchy requires an entity to maximize the use of observable inputs, where available. The following summarizes the three levels
of inputs required by the standard that the Company uses to measure fair value:
Level
1: Quoted prices in active markets for identical assets or liabilities
Level
2: Observable inputs other than Level 1 prices such as quoted prices for similar assets or liabilities; quoted prices in
markets that are not active or other inputs that are observable or can be corroborated by observable market data for substantially
the full term of the related assets or liabilities.
Level
3: Unobservable inputs that are supported by little or no market activity and that are significant to the fair value of
the assets or liabilities.
The
Company’s financial instruments as of August 31, 2014 consisted of $ 383,440 of Notes Payable and $8,000 due to TheraCyte,
Inc.. The fair value of all of the Company’s financial instruments as of August 31, 2014 were valued according to the Level
3 input. The carrying amount of the financial instruments is equal to the fair value as determined by the Company.
The
Company has determined that there are no Level 1 or Level 2 inputs for determining the fair value of the Company’s financial
instruments. Fair value was determined by the Company utilizing its own assumptions and estimation. There were no transfers between
levels for the period presented.
G.
INCOME TAXES
The
Company accounts for income taxes using the liability method prescribed by ASC 740, “ Income Taxes. ”
Under this method, deferred tax assets and liabilities are determined based on the difference between the financial reporting
and tax bases of assets and liabilities using enacted tax rates that will be in effect in the year in which the differences are
expected to reverse. The Company records a valuation allowance to offset deferred tax assets if based on the weight of available
evidence, it is more-likely-than-not that some portion, or all, of the deferred tax assets will not be realized. The effect on
deferred taxes of a change in tax rates is recognized as income or loss in the period that includes the enactment date.
The
Company applied the provisions of ASC 740-10-50, “Accounting for Uncertainty in Income Taxes”, which provides clarification
related to the process associated with accounting for uncertain tax positions recognized in our financial statements. Audit periods
remain open for review until the statute of limitations has passed. The completion of review or the expiration of the statute
of limitations for a given audit period could result in an adjustment to the Company’s liability for income taxes. Any such
adjustment could be material to the Company’s results of operations for any given quarterly or annual period based, in part,
upon the results of operations for the given period. As of August 31, 2014 the Company had no uncertain tax positions, and will
continue to evaluate for uncertain positions in the future.
The
Company generated a deferred tax credit through net operating loss carry forward. However, a valuation allowance of 100%
has been established.
Interest
and penalties on tax deficiencies recognized in accordance with ACS accounting standards are classified as income taxes in accordance
with ASC Topic 740-10-50-19.
H.
BASIC EARNINGS (LOSS) PER SHARE
The
Financial Accounting Standards Board (FASB) issued Accounting Standards Codification (ASC) 260, "Earnings Per Share",
which specifies the computation, presentation and disclosure requirements for earnings (loss) per share for entities with publicly
held common stock. ASC 260 requires the presentation of basic earnings (loss) per share and diluted earnings (loss) per share.
The Company has adopted the provisions of ASC 260 effective from inception. Basic net loss per share amounts is computed by dividing
the net income by the weighted average number of common shares outstanding. All convertible debt has an anti-dilutive effect on
the EPS, therefore Diluted earnings per share are the same as basic earnings per share.
NOTE
3. RECENT ACCOUNTING PRONOUNCEMENTS
The
following accounting standards updates were recently issued and have not yet been adopted by us. These standards are currently
under review to determine their impact on our consolidated financial position, results of operations, or cash flows.
On
January 31, 2013, the FASB issued Accounting Standards Update [ASU] 2013-01, entitled Clarifying the Scope of Disclosures about
Offsetting Assets and Liabilities. The guidance in ASU 2013-01 amends the requirements in the FASB Accounting Standards Codification
[FASB ASC] Topic 210, entitled Balance Sheet. The ASU 2013-01 amendments to FASB ASC 210 clarify that ordinary trade receivables
and receivables in general are not within the scope of ASU 2011-11, entitled Disclosure about Offsetting Assets and Liabilities,
where that ASU amended the guidance in FASB ASC 210. As those disclosures now are modified with the ASU 2013-01 amendments, the
FASB ASC 210 balance sheet offsetting disclosures now clearly are applicable only where reporting entities are involved with bifurcated
embedded derivatives, repurchase agreements, reverse repurchase agreements, and securities borrowing and lending transactions
that either are offset using the FASB ASC 210 or 815 requirements, or that are subject to enforceable master netting arrangements
or similar agreements. ASU 2013-01 is effective for annual reporting periods beginning on or after January 1, 2013, and interim
periods within those annual periods. The adoption of this ASU is not expected to have a material impact on our financial statements.
On
February 28, 2013, the FASB issued Accounting Standards Update [ASU] 2013-04, entitled Obligations Resulting from Joint and Several
Liability Arrangements for Which the Total Amount of the Obligation Is Fixed at the Reporting Date. The ASU 2013-04 amendments
add to the guidance in FASB Accounting Standards Codification [FASB ASC] Topic 405, entitled Liabilities and require reporting
entities to measure obligations resulting from certain joint and several liability arrangements where the total amount of the
obligation is fixed as of the reporting date, as the sum of the following:
The
amount the reporting entity agreed to pay on the basis of its arrangement among co-obligors.
Any
additional amounts the reporting entity expects to pay on behalf of its co-obligors.
While
early adoption of the amended guidance is permitted, for public companies, the guidance is required to be implemented in fiscal
years, and interim periods within those years, beginning after December 15, 2013. The amendments need to be implemented retrospectively
to all prior periods presented for obligations resulting from joint and several liability arrangements that exist at the beginning
of the year of adoption. The adoption of ASU 2013-04 is not expected to have a material effect on the Company’s operating
results or financial position.
On
April 22, 2013, the FASB issued Accounting Standards Update [ASU] 2013-07, entitled Liquidation Basis of Accounting. With ASU
2013-07, the FASB amends the guidance in the FASB Accounting Standards Codification [FASB ASC] Topic 205, entitled Presentation
of Financial Statements. The amendments serve to clarify when and how reporting entities should apply the liquidation basis of
accounting. The guidance is applicable to all reporting entities, whether they are public or private companies or not-for-profit
entities. The guidance also provides principles for the recognition of assets and liabilities and disclosures, as well as related
financial statement presentation requirements. The requirements in ASU 2013-07 are effective for annual reporting periods beginning
after December 15, 2013, and interim reporting periods within those annual periods. Reporting entities are required to apply the
requirements in ASU 2013-07 prospectively from the day that liquidation becomes imminent. Early adoption is permitted. The adoption
of ASU 2013-07 is not expected to have a material effect on the Company’s operating results or financial position.
Variety
of proposed or otherwise potential accounting standards are currently under study by standard setting organizations and various
regulatory agencies. Due to the tentative and preliminary nature of those proposed standards, the Company’s management
has not determined whether implementation of such standards would be material to its financial statements.
NOTE
4. GOING CONCERN
The
accompanying financial statements have been prepared assuming that the Company will continue as a going concern. The Company generated
net losses of $6,413,135 during the period from August 22, 2008 (inception) through August 31, 2014. This condition raises substantial
doubt about the Company's ability to continue as a going concern. The Company's continuation as a going concern is dependent on
its ability to meet its obligations, to obtain additional financing as may be required and ultimately to attain profitability.
The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Management
plans to raise additional funds primarily by offering securities for cash. Management has yet to decide what type of offering
the Company will use or how much capital the Company will raise. There is no guarantee that the Company will be able to raise
any capital through any type of offerings.
NOTE
5. NOTES PAYABLE
As
of August 31, 2014
Notes
Payable:
| Bio Technology
Partners Business Trust | |
$ | 58,000 | |
| The Sherman Family
Trust (10% Interest) | |
$ | 26,700 | |
| The Sherman Family
Trust (0% Interest) | |
$ | 180,500 | |
| Regen BioPharma Inc.
( See Note 6) | |
$ | 10,422 | |
| David Koos ( See
Note 6) | |
$ | 107,818 | |
| Total | |
$ | 383,440 | |
Convertible
Notes Payable
No
Convertible Notes payable outstanding as of August 31, 2014
As
of August 31, 2013
Notes
Payable:
| Bio
Technology Partners Business Trust |
|
$ |
13,550 |
| The Sherman Family
Trust (10% Interest) |
|
$ |
98,200 |
| Officer Loans (Note
6) |
|
$ |
117,588 |
| Venture Bridge Advisors |
|
$ |
37,606 |
| Southridge Partners
II LLP |
|
$ |
5,700 |
| Total |
|
$ |
272,644 |
Convertible
Notes Payable
8%
Convertible Notes Payable, $101,000
Bio
Technology Partners Business Trust has provided a line of credit to the Company in the amount of $200,000 or so much thereof as
may be disbursed to, or for the benefit of the Company by Lender in Lender's sole and absolute discretion. The unpaid principal
of this line of credit bears simple interest at the rate of ten percent per annum. Interest is calculated based on the principal
balance as may be adjusted from time to time to reflect additional advances or payments made hereunder. Principal balance and
accrued interest shall become due and payable in whole or in part at the demand of the Lender. The Sherman Family Trust (10% Interest)
has provided a line of credit to the Company in the amount of $700,000 or so much thereof as may be disbursed to, or for the benefit
of the Company by Lender in Lender's sole and absolute discretion. The unpaid principal of this line of credit bears simple interest
at the rate of ten percent per annum. Interest is calculated based on the principal balance as may be adjusted from time to time
to reflect additional advances or payments made hereunder. Principal balance and accrued interest shall become due and payable
in whole or in part at the demand of the Lender. $180,500 due to The Sherman Family Trust (0% Interest) is due and payable in
whole or in part at the option of the Holder and bears no interest. Amounts due to Regen Biopharma Inc. are due and payable at
the demand of the holder and bear simple interest at a rate of 10% per annum.
NOTE
6. RELATED PARTY TRANSACTIONS
As
of August 31, 2014 the Company remains indebted to David R. Koos in the principal amount of $107,818 due and payable in
whole or in part at the demand of David Koos and bearing simple interest at a rate of 15% per annum.
As
of August 31, 2014 the Company remains indebted to Regen Biopharma, Inc. in the principal amount of $10,422 due and payable
in whole or in part at the demand of Regen Biopharma, Inc and bearing simple interest at a rate of 10% per annum. David Koos,
the Company’s Chairman and CEO, is also the Chairman and CEO of Regen of Regen Biopharma, Inc.
The
Company allows Bio Matrix Scientific Group, Inc, (“BMSN”) and its affiliate, Regen Biopharma, Inc. ( entities under
common control with the Company) to share office space with the Company at 4700 Spring Street, Suite 304, La Mesa California,
91941 free of charge.
NOTE
7. INCOME TAXES
| As
of August 31, 2014 |
|
|
| Deferred
tax assets: |
|
|
|
|
| Net operating tax
carry forwards |
|
$ |
2,185,106 |
|
| Other |
|
|
-0- |
|
| |
|
|
|
|
| Gross
deferred tax assets |
|
|
2,185,106 |
|
| Valuation allowance |
|
|
(2,185,106) |
|
| Net deferred tax
assets |
|
$ |
-0- |
|
As
of August 31, 2014 the Company has a Deferred Tax Asset of $ 2,185,106 completely attributable to net operating loss
carry forwards of approximately $ 6,426,782 (which expire 20 years from the date the loss was incurred) consisting of:
(a)
$ 13,647 of Net Operating Loss carry forwards acquired in the reverse acquisition of Entest BioMedical, Inc., a California corporation,
and
(b)
$ 6,413,135 of Net Operating Loss carry forwards attributable to Entest BioMedical, Inc.
Realization
of deferred tax assets is dependent upon sufficient future taxable income during the period that deductible temporary differences
and carry forwards are expected to be available to reduce taxable income. A valuation allowance is recorded when it is “more
likely-than-not” that a deferred tax asset will not be realized. In addition, the reverse acquisition in which Entest BioMedical,
Inc. was involved in 2009 has resulted in a change of control. Internal Revenue Code Sec 382 limits the amount of income
that may be offset by net operating loss (NOL) carryovers after an ownership change. As a result, the Company has recorded a valuation
allowance reducing all deferred tax assets to $ -0-.
Income
tax is calculated at the 34% Federal Corporate Rate.
NOTE
8. ACQUISITION OF ENTEST CA
On
July 10, 2009 the Company acquired 100% of Entest CA, a California corporation and wholly owned subsidiary of the Company, from
BMSN for consideration consisting of (a) the issuance to BMSN of 10,000,000 newly issued common shares of Entest and (b) the return
by Mr. Rick Plote of 10,000,000 shares of Entest’s common stock previously issued to him by Entest for cancellation.
NOTE
9. ACQUISITION OF THE ASSETS OF PET POINTERS, INC.
On
January 4, 2011 Entest CA acquired from Pet Pointers, Inc., a California corporation doing business as McDonald Animal
Hospital (“Seller”), and Dr. Gregory McDonald DVM (“McDonald”) all the goodwill from McDonald and
assets of Seller except cash and accounts receivables used in connection with the operation of a veterinary medical clinic
located at 225 S. Milpas Street, Santa Barbara, CA 93103 (the "Business").
Consideration
for the acquisition consisted of:
I.
$70,000 in cash
II.
$210,000 of the Company’s common shares valued at the closing price per share as of January 4, 2011
III.
Payment of no more than $78,000 to a creditor of the Seller to be paid in monthly installments of $1,500 per month
IV.
Payment of no more than $25,000 to additional creditors of the Seller to be paid in monthly installments of $825 per month
V.
Payment of $50,000 to McDonald on the first business day of the fourth month following the closing of the acquisition (“Closing”).
NOTE
10. DISPOSITION OF THE ASSETS OF PET POINTERS, INC.
On
November 28, 2012 the “Company executed an agreement (“Agreement”) with Gregory McDonald ("McDonald"),
Pet Pointers, Inc. ("Pet Pointer") whereby Mc Donald and Pet Pointer would acquire from the Company all assets (with
the exception of cash and accounts receivable) utilized by the Company in the operation of the McDonald Animal Hospital, a full
service veterinary clinic owned and operated by the Company and located in Santa Barbara, California (“McDonald Asset Sale”).
On
October 10, 2012 a Complaint (“Complaint”) was filed in the Superior Court of the State of California against the
Company and David Koos by McDonald, a former employee of the Company, alleging breach of contract and breach of the covenant of
good faith and dealing in connection with the assumption of lease obligations by the Company in connection with the acquisition
of the assets of Pet Pointers, Inc breach of contract and breach of the covenant of good faith and dealing in connection with
an employment agreement enters into with McDonald inc connection with the Acquisition, breach of contract in connection with the
Acquisition purchase agreement, breach of the covenant of good faith and dealing in connection with the Acquisition purchase agreement,
implied indemnity in connection to amounts owed by McDonald to Anthony and Judi Marinelli, the Internal Revenue Service, and the
California Franchise Tax Board, intentional misrepresentation, negligent misrepresentation , failure to pay wages and violations
of Sections 2802, 203, and 2806 of the California Labor Code. The Complaint sought judgment for nominal damages, actual damages,
compensatory damages, lost wages, compensation, expenses wage benefits and penalties pursuant to California Labor Code Sections
203 et al, 2802 and 2806, indemnification, accrued interest, punitive damages, costs of suit and attorney’s fees.
As
consideration to the Company for the assets acquired, McDonald and Pet Pointers provided to the Company a General release whereby
McDonald and Pet Pointer waive, release and discharge the Company and their respective assignees, officers, directors, shareholders,
boards, owners, employees, attorneys, agents, trustors, trustees, beneficiaries, heirs, successors, and representatives from all
known and unknown claims, demands, causes of action, attorney's fees, costs, or expenses including:
(1)
All claims relating to the Complaint.
(2)
Those owed by McDonald to Anthony and Judi Marinelli which the Company became obligated to pay on McDonald’s behalf pursuant
to the asset purchase agreement entered into between the Company and Gregory McDonald and Pet Pointers, Inc on January 4, 2011.
(3)
Those amounts owed by McDonald to the Internal Revenue Service which the Company became obligated to pay on McDonald’s behalf
pursuant to the asset purchase agreement entered into between the Company and Gregory McDonald and Pet Pointers, Inc on January
4, 2011.
(4)
Those amounts owed by McDonald to the California Franchise Tax Board which the Company became obligated to pay on McDonald’s
behalf pursuant to the asset purchase agreement entered into between the Company and Gregory McDonald and Pet Pointers, Inc on
January 4, 2011.
Assets
disposed of pursuant to the Agreement include approximately $4,840 of Property Plant and Equipment net of accumulated depreciation
as well as all inventory held at the McDonald Animal Hospital.
Assets
disposed of pursuant to the Agreement also include
(i)
Essentially all intellectual property, including computer software, utilized in connection with the operation of the McDonald
Animal Hospital
(ii)
All telephone numbers, fax numbers, service marks, trademarks, trade names, fictitious business names, websites, business email
addresses, vendor lists, promotional materials, vendor records and any and all business records including, but not limited to,
such items stored in computer memories, microfiche, paper record or by any other means relevant to the operation of the McDonald
Animal Hospital.
(iii)
All customer lists, customer contacts, and any and all customer records that are related to the McDonald Animal Hospital.
As
a result of the agreement, the Company recorded a non-cash pre-tax charge for the impairment of goodwill recorded in connection
with the acquisition of the McDonald Animal Hospital of approximately $405,000 for the quarter ended November 30, 2012.
Pursuant
to the Agreement, the Company is obligated to make payment of $13,000 within five days of the Closing of the Agreement as such
term is defined in the Agreement.
Pursuant
to the Agreement, the Company agrees to waive, release and discharge McDonald and Pet Pointer from all known and unknown claims,
demands, causes of action, attorney's fees, costs, or expenses.
NOTE
11. COMMITMENTS AND CONTINGENCIES
On
November 1, 2011, the Company entered into an agreement to lease approximately 2,320 square feet of office space beginning December
1, 2011 for a period of five years.
Rent
to be charged to the Company pursuant to the lease is as follows:
$2,996
per month for the period beginning December 1, 2011 and ending November 30, 2012
$3,116
per month for the period beginning December 1, 2012 and ending November 30, 2013
$3,241
per month for the period beginning December 1, 2013 and ending November 30, 2014
$3,371
per month for the period beginning December 1, 2014 and ending November 30, 2015
$3,506
per month for the period beginning December 1, 2015 and ending November 30, 2016
This
property is utilized as office space. The Company believes that the foregoing property is adequate to meet its current needs.
While it is anticipated that the Company will require access to laboratory facilities in the future, the Company believes that
access to such facilities are available from a variety of sources.
On
May 24, 2012, a Complaint (“Complaint”) was filed in the U.S. Bankruptcy Court for the District of Oregon against
the Company by Titterington Veterinary Services Inc. (“TVS”). The Complaint is an adversary proceeding filed by TVS
arising from TVS’s bankruptcy case currently pending in U.S. Bankruptcy Court for the District of Oregon. The Complaint
alleges Breach of Contract resulting from the Company’s alleged failure to pay certain expenses the Company was required
to pay pursuant to an agreement with TVS, Dr. Ronald Titterington, DVM and Dr. Kathy Snell, DVM (“TVS Agreement”).
TVS is seeking a judgment and money award against the Company in an amount to be proven at trial which TVS estimates in the Complaint
to be up to $50,000. TVS is also seeking a judgment and order against the Company to provide an accounting of all revenues received
by the Company pursuant to the TVS Agreement, all expenses paid, unpaid, and due and owing pursuant to the TVS Agreement
as well as a revenue share which TVS claims is due them pursuant to the TVS Agreement. TVS is also seeking a judgment requiring
the Company to turn over a sum of money equal to expenses the Company was obligated to pay pursuant to the TVS Agreement. TVS
is also seeking attorney’s fees and expenses. The Company believes that the allegations in the complaint are without
merit and intends to vigorously defend its interests in this matter. At this time, it is not possible to predict the ultimate
outcome of these matters and an outcome unfavorable to the Company may have a material adverse effect on the Company. On September
19, 2012 the Plaintiff’s Claim for Relief for turnover and an accounting under 11 U.S.C. § 542 and the Plaintiff's
Claim for Relief for attorney fees were dismissed with prejudice and , as per the claim of breach of contract, the proceeding
was transferred to the United States Bankruptcy Court for the District of Southern California for all further proceedings. This
Complaint was dismissed in its entirety on June 30, 2014.
There
were no other legal proceedings against the Company with respect to matters arising in the ordinary course of business. The Company
is not involved in any other litigation either as plaintiffs or defendants, and has no knowledge of any threatened or pending
litigation against the Company.
NOTE
12. STOCKHOLDERS EQUITY
The
stockholders' equity section of the Company contains the following classes of capital stock as of August 31, 2014:
Common
Stock:
$0.0001
par value, 6,000,000,000 shares authorized and 2,205,570,752 shares issued and outstanding as of August 31, 2014.
Preferred
Stock:
$0.0001
par value 5,000,000 shares authorized of which
| |
(a) |
100,000
are authorized as Series AA Preferred Stock of which 100,000 shares are issued and outstanding as of August 31,
2014 and |
| |
(b) |
4,400,000
are authorized as Series B Preferred Stock of which 4,201,397 shares are issued and outstanding
as of August31, 2014.
|
(c)
300,000 are authorized as Series AAA Preferred Stock of which 80,000 shares are issued and outstanding as of August
31, 2014
Upon
any liquidation, dissolution, or winding up of the Company, whether voluntary or involuntary (collectively, a “Liquidation”),
before any distribution or payment shall be made to any of the holders of Common Stock or any other series of preferred stock,
the holders of Series B Preferred Stock shall be entitled to receive out of the assets of the Company, whether such assets are
capital, surplus or earnings, an amount equal to $0.10 per share of Series B Preferred Stock (the “Liquidation Amount”)
plus all declared and unpaid dividends thereon, for each share of Series B Preferred Stock held by them.
If,
upon any Liquidation, the assets of the Company shall be insufficient to pay the Liquidation Amount, together with declared and
unpaid dividends thereon, in full to all holders of Series B Preferred Stock, then the entire net assets of the Company shall
be distributed among the holders of the Series B Preferred Stock, ratably in proportion to the full amounts to which they would
otherwise be respectively entitled and such distributions may be made in cash or in property taken at its fair value (as determined
in good faith by the Board), or both, at the election of the Board..
Non
Voting Convertible Preferred Stock having a $1.00 par value:
200,000
shares authorized of which 0 shares are issued and outstanding as of August 31, 2014 .
Non
Voting Convertible Preferred Stock shall convert at the option of the holder into shares of the corporation’s common stock
at a conversion price equal to seventy percent (70%) of the lowest Closing Price for the five (5) trading days immediately preceding
written receipt by the corporation of the holder’s intent to convert.
“CLOSING
PRICE" shall mean the closing bid price for the corporation’s common stock on the Principal Market on a Trading Day
as reported by Bloomberg Finance L.P.
“PRINCIPAL
MARKET" shall mean the principal trading exchange or market for the corporation’s common stock.
“TRADING
DAY” shall mean a day on which the Principal Market shall be open for business.
NOTE
13. STOCK TRANSACTIONS
During
the quarter ended November 30, 2013:
On
September 4, 2013 The Company issued 27,887,324 of its common shares in satisfaction of $19,800 of convertible notes payable.
On
September 16, 2013 The Company issued 28,000,000 of its common shares in satisfaction of $12,600 of convertible notes
payable.
On
September 20, 2013 The Company issued 20,820,513 of its common shares in satisfaction of $5,600 of convertible notes payable and
$2,520 of accrued interest on convertible notes payable.
On
September 23, 2013 The Company issued 39,325,397 of its common shares in conversion of 24,775 of the Company’s Non Voting
Convertible Preferred Stock
On
September 24, 2013 The Company issued 27,948,718 of its common shares in satisfaction of $10,900 of convertible notes payable.
On
September 27, 2013 The Company issued 27,948,718 of its common shares in satisfaction of $10,900 of convertible notes payable.
On
September 30, 2013 The Company issued 60,000,000 of its common shares in satisfaction of $13,550 of notes payable.
On
October 2, 2013 The Company issued 24,848,485 of its common shares in satisfaction of $8,200 of convertible notes payable.
On
October 8, 2013 The Company issued 27,727,273 of its common shares in satisfaction of $6,100 of convertible notes payable.
On
October 9, 2013 The Company issued 28,000,000 of its common shares in satisfaction of $5,600 of convertible notes payable.
On
October 10, 2013 The Company issued 27,777,778 of its common shares in satisfaction of $5,000 of convertible notes payable.
On
October 14, 2013 The Company issued 60,000,000 of its common shares in satisfaction of $13,550 of notes payable.
On
October 15, 2013 The Company issued 27,777,778 of its common shares in satisfaction of $5,000 of convertible notes payable.
On
October 18, 2013 The Company issued 20,652,000 of its common shares in satisfaction of $3,300 of convertible notes payable.
On
October 22, 2013 The Company issued 55,000,000 of its common shares in satisfaction of $6,600 of convertible notes payable.
On
October 24, 2013 The Company issued 27,500,000 of its common shares in satisfaction of $1,400 of convertible notes payable and
$1,900 of accrued interest on convertible notes payable.
On
November 23, 2013 The Company issued 60,000,000 of its common shares in satisfaction of $13,550 of notes payable.
During
the quarter ended February 28, 2014:
On
January 20, 2014 the Company issued to David Koos, the Company’s Chairman and CEO, 95,000 shares of the Company’s
Series AA Preferred Stock in satisfaction of $10,000 of salary accrued but unpaid owed to David Koos.
On
January 24, 2014 the Company issued to David Koos, the Company’s Chairman and CEO, 1,000,000 shares of the Company’s
Series B Preferred Stock in satisfaction of $1,000 of salary accrued but unpaid owed to David Koos.
On
January 24, 2014 the Company issued to David Koos, the Company’s Chairman and CEO, 15,000,000 shares of the Company’s
Common Stock in satisfaction of $15,000 of principal indebtedness owed to David Koos.
On
January 28, 2014 the Company issued 115,000,000 shares of the Company’s Common Stock in satisfaction of $10,506 of principal
indebtedness.
During
the quarter ended May 31, 2014:
On
March 3, 2014 the Company issued 125,000,000 shares of the Company’s Common Stock in satisfaction of $12,500 of principal
indebtedness.
On
March 13, 2014 the Company issued 140,000,000 shares of the Company’s Common Stock in satisfaction of $14,000 of principal
indebtedness.
On
March 28, 2014 the Company issued 155,000,000 shares of the Company’s Common Stock in satisfaction of $15,500 of principal
indebtedness
On
April 22, 2014 the Company issued 170,000,000 shares of the Company’s Common Stock in satisfaction of $17,000 of principal
indebtedness
On
May 16, 2014 the Company issued 125,000,000 shares of the Company’s Common Stock in satisfaction of $12,500 of principal
indebtedness.
On
May 22, 2014 the Company issued 80,000 shares of Series AAA Preferred Stock to David R. Koos, the Company’s Chairman, President
and CEO as consideration for $10,000 of salary accrued and unpaid owed to David R. Koos by the Company.
During
the quarter ended August 31, 2014:
On
August 7, 2014 the Company issued 195,000,000 shares of the Company’s Common Stock in satisfaction of $19,500 of principal
indebtedness.
On
August 11, 2014 the Company issued 12,500,000 shares of the Company’s Common Stock as compensation to an employee.
NOTE
14. SUBSEQUENT EVENTS
On
October 22, 2014 the Company issued 200,000,000 shares of the Company’s Common Stock in satisfaction of $20,000 of principal
indebtedness.
Item
9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
During the Registrant's most two most recent fiscal years there were no disagreements with Seale and Beers, Certified Public Accountants
LLC (“S&B”) , the Company’s independent registered public accounting firm, whether or not resolved, on any
matter of accounting principles or practices, financial statement disclosure, or auditing scope or procedure, which, if not
resolved to S&B’s satisfaction, would have caused it to make reference to the subject matter of the disagreement in
connection with its report on the Registrant's financial statements.
Item
9A. Controls and Procedure
a)
Evaluation of disclosure controls and procedures.
The
principal executive officer and principal financial officer have evaluated the Company’s disclosure controls and procedures
as of August 31, 2014. Based on this evaluation, they have concluded that the disclosure controls and procedures were effective
to ensure that the information required to be disclosed by the Company in the reports that it files or submits under the Securities
Exchange Act of 1934 is recorded, processed, summarized and reported, within the time periods specified in the Commission’s
rules and forms and to ensure that information required to be disclosed by the Company in the reports that it files or submits
under the Securities Exchange Act of 1934 is accumulated and communicated to the Company’s management, including its principal
executive and principal financial officers, or persons performing similar functions, as appropriate to allow timely decisions
regarding required disclosure. David Koos is the Company’s CEO and acting CFO. He functions as the Company’s principal
executive officer and principal financial officer.
b)
Management’s annual report on internal control over financial reporting.
Management
of the Company is responsible for establishing and maintaining adequate internal control over financial reporting as defined in
Rule 13a-15(f) promulgated under the Securities and Exchange Act of 1934. Rule 13a-15(f) defines internal control over financial
reporting as follows:
“The
term internal control over financial reporting is defined as a process designed by, or under the supervision of, the issuer's
principal executive and principal financial officers, or persons performing similar functions, and effected by the issuer's board
of directors, management and other personnel, to provide reasonable assurance regarding the reliability of financial reporting
and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles
and includes those policies and procedures that:
Pertain
to the maintenance of records that in reasonable detail accurately and fairly reflect the transactions and dispositions of the
assets of the issuer;
Provide
reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with
generally accepted accounting principles, and that receipts and expenditures of the issuer are being made only in accordance with
authorizations of management and directors of the issuer; and
Provide
reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of the issuer's
assets that could have a material effect on the financial statements.”
The
Company’s internal control over financial reporting is a process designed under the supervision of the Company’s management
to provide reasonable assurance regarding the reliability of financial reporting and the preparation of the Company’s financial
statements for external purposes in accordance with U.S. generally accepted accounting principles.
In
designing and evaluating our disclosure controls and procedures, our management recognized that disclosure controls and procedures,
no matter how well conceived and operated, can provide only a reasonable, not absolute, assurance that the objectives of the disclosure
controls and procedures are met.
The
Company’s management assessed the effectiveness of its internal control over financial reporting as of August 31, 2014 based
on the framework in “Internal Control over Financial Reporting – Guidance for Smaller Public Companies (2006) issued
by the Committee of Sponsoring Organizations of the Treadway Commission.” Based on its assessment, management believes that,
as of August 31, 2014, the Company’s internal control over financial reporting is effective.
Management's
report was not subject to attestation by the Company's registered public accounting firm pursuant to temporary rules of the Securities
and Exchange Commission that permit the company to provide only management's report in this annual report. This exemption for
smaller reporting companies provided under the temporary rules referenced above has been made permanent under Section 989G of
the Dodd-Frank Wall Street Reform and Consumer Protection Act.
(c)
There have been no changes during the quarter ended August 31, 2014 in the Company’s internal controls over financial reporting
that have materially affected, or are reasonably likely to materially affect, internal control over financial reporting.
Item
10. Directors, Executive Officers and Corporate Governance
On
June 19, 2009 the Board of Directors of the Company elected David R. Koos, 52, a director of the Company and appointed
Dr. Koos President, Chief Executive Officer, Secretary, Chief Financial Officer, Principal Accounting Officer of the Company.
Dr. Koos resigned as Chief Financial Officer on March 31, 2010 and assumed the position of Acting Chief Financial Officer and
Principal Accounting Officer on August 8, 2011 upon the resignation of Tammy L. Reynolds who served as Chief Financial Officer
from the period from March 31, 2010 to August 8, 2011.
Dr.
Koos has served as Chairman, CEO, President, Secretary, and Acting CFO of the BMSN since June 19, 2006, and as Chairman CEO, President,
Secretary, and Acting CFO of Entest CA since August 22, 2008
education:
DBA
- Finance (December 2003)
Atlantic
International University
Ph.D.
- Sociology (September 2003)
Atlantic
International University
MA
- Sociology (June 1983)
University
of California - Riverside, California
Five
Year Employment History:
| |
|
|
| Position: |
Company
Name: |
Employment
Dates: |
| |
|
|
| Chairman,
President, CEO and Acting CFO |
Bio-Matrix
Scientific Group, Inc.*
|
June
14, 2006 (Chairman) to Present
June
19, 2006 (President, CEO and Acting CFO)
June
19, 2006 (Secretary) to Present |
| Chairman
, CEO and Acting CFO |
Regen
Biopharma, Inc. |
April
24, 2012 to Present |
| President |
Regen
Biopharma, Inc |
May
29, 2013 to Present |
| Chairman,
CEO, Secretary & Acting CFO |
Frezer
Inc. |
May
2, 2005 to February 2007 |
| Chairman,
CEO & Acting CFO |
BMXP
Holdings, Inc.
|
December
6, 2004 to June 2008 |
| Managing
Director & President |
Cell
Source Research Inc. |
December
5, 2001 to Present |
| Managing
Director & President |
Venture
Bridge Inc. |
November
21, 2001 to Present |
| Chairman
of the Board of Directors, CFO & Secretary |
Cell
Bio-Systems Inc.
(New
York) |
July
17, 2003 to December 1, 2003 |
| Registered
Representative |
Amerivet
Securities Inc.** |
March
31, 2004 to February 2008 |
*
As of November 25,2014 Bio-Matrix Scientific Group Inc. owned 10,000,000 common shares of the company
**
Amerivet Securities Inc. has not been active during the period as the Chief Executive Officer was on deployment in Iraq through
the U.S. Army Reserves.
Code
of Ethics
On
November 11, 2009 we adopted a Code of Ethics pursuant to Section 406 of the Sarbanes-Oxley Act of 2002
Director
Independence
Audit
Committee and Audit Committee Financial Expert
The
sole member of the Company’s board of Directors may not be considered independent as he is also its sole officer. The
Company is not a "listed company" under Securities and Exchange Commission (“SEC”) rules and is therefore
not required to have an audit committee comprised of independent directors. The Company does not currently have an audit committee,
however, for certain purposes of the rules and regulations of the SEC and in accordance with the Sarbanes-Oxley Act of 2002, the
Company’s Board of Directors is deemed to be its audit committee and as such functions as an audit
committee and performs some of the same functions as an audit committee including: (1) selection and oversight of our independent
accountant; (2) establishing procedures for the receipt, retention and treatment of complaints regarding accounting, internal
controls and auditing matters; and (3) engaging outside advisors. The Board of Directors has determined that its member is able
to read and understand fundamental financial statements and has substantial business experience that results in that member's
financial sophistication. Accordingly, the Board of Directors believes that its member has the sufficient knowledge
and experience necessary to fulfill the duties and obligations that an audit committee would have.
Nominating
and Compensation Committees
The
Company does not have standing nominating or compensation committees, or committees performing similar functions. The board
of directors believes that it is not necessary to have a compensation committee at this time because the functions of such committee
are adequately performed by the board of directors. The board of directors also is of the view that it is appropriate for the
Company not to have a standing nominating committee because the board of directors has performed and will perform adequately the
functions of a nominating committee. The Company is not a "listed company" under SEC rules and is therefore
not required to have a compensation committee or a nominating committee.
Shareholder
Communications
There
has not been any defined policy or procedure requirements for stockholders to submit recommendations or nomination for directors.
There are no specific, minimum qualifications that the board of directors believes must be met by a candidate recommended by the
board of directors. Currently, the entire board of directors decides on nominees, on the recommendation of any member of the board
of directors followed by the board’s review of the candidates’ resumes and interview of candidates. Based on the information
gathered, the board of directors then makes a decision on whether to recommend the candidates as nominees for director. The Company
does not pay any fee to any third party or parties to identify or evaluate or assist in identifying or evaluating potential nominee.
Because management
and the Board of Directors of the Company are the same people, the Board of Directors has determined not to adopt
a formal methodology for communications from shareholders on the belief that any communication would be brought to the Board of
Directors’ attention by virtue of the co-extensive capacities of the Board of Directors.
Executive
Compensation
SUMMARY
COMPENSATION TABLE
| Name
and Principal Position |
Year |
Salary
($) |
Bonus
($) |
Stock
Awards
($) |
Restricted
Stock
Awards
($) |
Option
Awards
($) |
Non
Equity
Incentive
Plan
Compensation
($) |
Nonqualified
Deferred
Compensation
Earnings
($) |
All
Other
Compensation
($) |
Total
($) |
| |
|
|
|
|
|
|
|
|
|
|
David
Koos
Chairman,
President and CEO |
From
September 1, 2012 to
August
31, 2013 |
$120,000* |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
$120,000 |
| |
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
| Name
and Principal Position |
Year |
Salary
($) |
Bonus
($) |
Stock
Awards
($) |
Restricted
Stock
Awards
($)(a) |
Option
Awards
($) |
Non
Equity
Incentive
Plan
Compensation
($) |
Nonqualified
Deferred
Compensation
Earnings
($) |
All
Other
Compensation
($) |
Total
($) |
| |
|
|
|
|
|
|
|
|
|
|
David
Koos
Chairman,
President and CEO |
From
September 1, 2013 to August 31, 2014 |
$120,000** |
0 |
11,000*** |
0 |
0 |
0 |
0 |
0 |
$131,000 |
*
Includes $105,500 in salary earned by David Koos which was accrued but unpaid for the fiscal year ended August 31, 2013.
**
Includes 120,000 in salary earned by David Koos which was accrued but unpaid for the fiscal year ended August 31, 2014.
***
On January 20, 2014 the Company issued to David Koos 95,000 Series AA Preferred shares in satisfaction of $10,000 of salary accrued
but unpaid and 1,000,000 Series B Preferred shares in satisfaction of $1,000 of salary accrued but unpaid.
Item
12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The
following table sets forth information known to the Company with respect to the beneficial ownership of each class of the Company’s
capital stock as of November 21, 2014 for (1) each person known by the Company to beneficially own more than 5% of each class
of the Company’s voting securities, (2) each executive officer, (3) each of the Company’s directors and
(4) all of the Company’s executive officers and directors as a group. As of November 21, 2014 the Company had 2,405,570,752 common
shares outstanding, 4, 201,397 Series B Preferred Shares outstanding , Preferred Shares outstanding 100,000 Series AA Preferred
Shares outstanding, and 80,000 Series AAA Preferred Shares outstanding.
Based
on 2,405,570, 642 shares issued and outstanding as of November 21, 2014
| |
|
|
|
| Title
of Class |
Name
and Address of Beneficial Owner |
Amount
and Nature
of
Beneficial Owner |
Percent
of Class |
| Common |
David
R. Koos*
C/o
Entest Bio Medical Inc., Inc
4700
SPRING STREET, SUITE 203, LA MESA, CALIFORNIA, 91942 |
75,000,000 |
3.1% |
| |
|
|
|
| Common |
All
Officers and Directors As a Group |
75,000,000 |
3.1% |
*Includes
10,000,000 common shares owned by Bio-Matrix Scientific Group, Inc. (David Koos is CEO, President and Chairman of Bio-Matrix Scientific
Group, Inc.).
.
Based
on 4,201,397 shares issued and outstanding as of November 21, 2014
| |
|
|
|
| Title
of Class |
Name
and Address of Beneficial Owner |
Amount
and Nature
of
Beneficial Owner |
Percent
of Class |
| Series
B Preferred |
David
R. Koos*
c/o Entest Bio Medical Inc.
4700 Spring Street, Suite 203, La Mesa, California, 91942 |
2,250,000 |
53% |
| Series
B Preferred |
Sleezer
Family Trust
c/o Charles Sleezer
12550 Carson Street, Hawaiin Gardens, CA |
687,500 |
16.4% |
| Series
B Preferred |
Ronald
J. Titterington and Kathy Snell
3880 W 11th Street, Eugene, Oregon |
312,500 |
7.4% |
| Series
B Preferred |
All
Officers and Directors as a Group* |
2,250,000 |
53% |
| |
*Includes
1,250,000 Series B Preferred shares owned by Bio-Matrix Scientific Group, Inc. (David Koos is CEO, President and Chairman
of Bio-Matrix Scientific Group, Inc.).
Based
on 100,000 shares issued and outstanding as of November 21, 2014
| |
|
|
|
| Title
of Class |
Name
and Address of Beneficial Owner |
Amount
and Nature
of
Beneficial Owner |
Percent
of Class |
| Series
AA Preferred Shares |
David
R. Koos
C/o
Entest Bio Medical Inc., Inc
4700
SPRING STREET, SUITE 203, LA MESA, CALIFORNIA, 91942 |
100,000 |
100% |
| Series
AA Preferred Shares |
All
Officers and Directors
As
a Group |
100,000 |
100% |
Based
on 80,000 shares issued and outstanding as of November 21, 2014
| |
|
|
|
| Title
of Class |
Name
and Address of Beneficial Owner |
Amount
and Nature
of
Beneficial Owner |
Percent
of Class |
| Series
AAA Preferred Shares |
David
R. Koos
C/o
Entest Bio Medical Inc., Inc
4700
SPRING STREET, SUITE 203, LA MESA, CALIFORNIA, 91942 |
80,000 |
100% |
| Series
AAA Preferred Shares |
All
Officers and Directors
As
a Group |
80,000 |
100% |
Item
13. Certain Relationships and Related Transactions, and Director Independence
During
the year ended August 31, 2014 , David Koos made net loans to the Company totaling $5,230. These loans are due and payable at
the demand of David Koos and bear simple interest at a rate of 15% per annum.
As
of August 31, 2014 the Company remains indebted to David R. Koos in the principal amount of $107,818 due and payable at
the demand of David Koos and bearing simple interest at a rate of 15% per annum.
During
the year ended August 31, 2014 Regen Biopharma Inc., a company under common control, lent the Company $10,422 due and payable
in whole or in part at the demand of Regen Biopharma, Inc and bearing simple interest at a rate of 10% per annum.
The
Company allows Bio Matrix Scientific Group, Inc, and its affiliate, Regen Biopharma, Inc. ( entities under common control with
the Company) to share office space with the Company at 4700 Spring Street, Suite 304, La Mesa California, 91941free of charge.
Director
Independence
Audit
Committee and Audit Committee Financial Expert
The
Company’s sole director may not be considered independent as he is also an officer. The
Company is not a "listed company" under Securities and Exchange Commission (“SEC”) rules and is therefore
not required to have an audit committee comprised of independent directors. The Company does not currently have an audit committee,
however, for certain purposes of the rules and regulations of the SEC and in accordance with the Sarbanes-Oxley Act of 2002, the
Company’s Board of Directors is deemed to be its audit committee and as such functions as an audit
committee and performs some of the same functions as an audit committee including: (1) selection and oversight of our independent
accountant; (2) establishing procedures for the receipt, retention and treatment of complaints regarding accounting, internal
controls and auditing matters; and (3) engaging outside advisors. The Board of Directors has determined that its sole member is
able to read and understand fundamental financial statements and has substantial business experience that results in the member's
financial sophistication. Accordingly, the Board of Directors believes that its member has the sufficient knowledge and experience
necessary to fulfill the duties and obligations that an audit committee would have.
Nominating
and Compensation Committees
The
Company does not have standing nominating or compensation committees, or committees performing similar functions. The Board
of Directors believes that it is not necessary to have a compensation committee at this time because the functions of such committee
are adequately performed by the board of directors. The Board of Directors also is of the view that it is appropriate for the
Company not to have a standing nominating committee because the Board of Directors has performed and will perform adequately the
functions of a nominating committee. The Company is not a "listed company" under SEC rules and is therefore
not required to have a compensation committee or a nominating committee.
Shareholder
Communications
There
has not been any defined policy or procedure requirements for stockholders to submit recommendations or nomination for directors.
There are no specific, minimum qualifications that the board of directors believes must be met by a candidate recommended by the
board of directors. Currently, the entire board of directors decides on nominees, on the recommendation of any member of the board
of directors followed by the board’s review of the candidates’ resumes and interview of candidates. Based on the information
gathered, the board of directors then makes a decision on whether to recommend the candidates as nominees for director. The Company
does not pay any fee to any third party or parties to identify or evaluate or assist in identifying or evaluating potential nominee.
The
Board of Directors has determined not to adopt a formal methodology for communications from shareholders on the belief that any
communication would be brought to the board of directors’ attention by virtue of communication with management
Item
14. Principal Accounting Fees and Services
The
following table sets forth the aggregate fees billed to us by Seale and Beers , CPAs during the period beginning September 1,
2013 and ending August 31, 2014:
| Audit
Fees |
|
$ |
15,000 |
|
| Audit
Related Fees |
|
|
13,592 |
|
| Tax
Fees |
|
|
0 |
|
| All
Other Fees |
|
|
0 |
|
| |
|
$ |
28,592 |
|
Audit
Fees: Aggregate fees billed for professional services rendered for the audit of the Company's annual financial statements.
Audit
Related Fees: Aggregate fees billed for professional services rendered for assurance and related services that
were reasonably related to the performance of the audit or review of our financial statements and are not reported under “Audit
Fees” above. During the year ended August 31, 2014 these fees were primarily derived from review of financial statements
in the Company's Form 10Q Reports.
All
services listed were pre-approved by the Board of Directors, functioning as the Audit Committee in accordance with Section 2(a)
3 of the Sarbanes-Oxley Act of 2002.
The
Board has considered whether the services described above are compatible with maintaining the independent accountant's independence
and has determined that such services have not adversely affected the independence of Seale and Beers , CPAs.
Item
15. Exhibit Index
EXHIBIT
INDEX
| Exhibit
No. |
Description |
|
|
| |
3(i)(a) |
|
Articles
of Incorporation |
| |
3(ii)(b) |
|
Bylaws |
| |
10.1
( c) |
|
Agreement
by and Between David Koos and the Company |
| |
10.2
(Incorporated by Reference to Exhibit 10.2 of the Company’s Form S-1 filed June 12, 2012) |
|
Terms
of Employment Agreement by and between the Company and Tammy Reynolds |
| |
10.3
(d) |
|
August
2010 Agreement by and between the Company and TheraCyte, Inc. |
| |
10.3
(e) |
|
Asset
Purchase Agreement by and between the Company, Dr. Gregory McDonald and Pet Pointers |
| |
10.4
(f) |
|
Exhibit
A to Asset Purchase Agreement by and between the Company, Dr. Gregory McDonald and Pet Pointers |
| |
10.5
(g) |
|
Exhibit
B to Asset Purchase Agreement by and between the Company, Dr. Gregory McDonald and Pet Pointers |
| |
10.6
(h) |
|
CRO
Agreement by and between the Company and RenovoCyte LLC |
| |
3(i)(2)(i) |
|
Certificate
of Designations Series AA Preferred Stock |
| |
10.7(j) |
|
8%
ASHER NOTE $42,500 |
| |
10.8(k) |
|
8%
ASHER NOTE $37,500 |
| |
10.9(l) |
|
8%
ASHER NOTE $35,000 |
| |
10.10(m) |
|
8%
ASHER NOTE $32,500 |
| |
3(i)(3)
(n) |
|
Amendment
to Certificate of Incorporation dated June 4, 2009 |
| |
3(ii)(2)
(o) |
|
Bylaws
of Entest Biomedical, inc., a California corporation and wholly owned subsidiary of the Registrant |
| |
3(i)(4)
(p) |
|
Certificate
of Incorporation of Entest Biomedical, inc., a California corporation and wholly owned subsidiary of the Registrant |
| |
10.11(q) |
|
8%
ASHER NOTE $37,500 |
| |
3(i)(5)
( r) |
|
Certificate
of Designations Series B Preferred Stock |
| |
3(i)
(6)(s) |
|
Amendment
to Certificate of Incorporation |
| |
10.12
(t) |
|
Equity
purchase Agreement, Southridge Feb 27, 2012 |
| |
10.13(u) |
|
Registration
Rights Agreement, Southridge |
| |
10.14(Incorporated
by Reference to Exhibit 10.14 of the Company’s Form S-1 filed June 12, 2012) |
|
Line
of Credit Promissory Note December 1, 2010 Bio Technology Partners Business Trust |
| |
10.15(Incorporated
by Reference to Exhibit 10.15 of the Company’s Form S-1 filed June 12, 2012) |
|
Line
of Credit Promissory Note December 1, 2010 Venture Bridge Advisors, Inc. |
| |
10.16(Incorporated
by Reference to Exhibit 10.16 of the Company’s Form S-1 filed June 12, 2012) |
|
Line
of Credit Promissory Note December 1, 2010 Bombardier Pacific ventures |
| |
10.17(Incorporated
by Reference to Exhibit 10.17 of the Company’s Form S-1 filed June 12, 2012) |
|
Line
of Credit Promissory Note December 1, 2010 David Koos |
| |
3(i)(7)
(v) |
|
Amendment
to Certificate of Incorporation |
| |
10.18(Incorporated
by Reference to Exhibit 10.18 of the Company’s Form S-1 filed June 12, 2012) |
|
Agreement
amending terms of outstanding Debt by the Company |
| |
10.19(Incorporated
by Reference to Exhibit 10.19 of the Company’s Form S-1 filed June 12, 2012) |
|
Agreement
amending terms of outstanding Debt by the Company |
| |
10.20(Incorporated
by Reference to Exhibit 10.20 of the Company’s Form S-1 filed June 12, 2012) |
|
Agreement
amending terms of outstanding Debt by the Company |
| |
10.21(w) |
|
Form
of Bonus Share Agreement |
| |
5.1 |
|
Opinion
Regarding Legality |
| |
14
(y) |
|
Code
of Ethics |
| |
10.24
(aaaa) |
|
Assignment
dated June 15, 2009 by and between Entest Biomedical, Inc. and Bio-Matrix Scientific Group, Inc. |
| |
10.22
(z) |
|
Lease |
| |
3(i)(8)
(aa) |
|
Text
of Amendment to Certificate of Incorporation |
| |
10.23
(aaa) |
|
8%
ASHER NOTE $42,500 |
| |
10.25(Incorporated
by Reference to Exhibit 10.25 of the Company’s Form S-1 filed June 12, 2012) |
|
Advisory
Board Letter Agreement |
| |
10.26
(aaaaa) |
|
Equity
Purchase Agreement Southridge June 1 2012 |
| |
10.27(Incorporated
by Reference to Exhibit 10.27 of the Company’s Form S-1 filed June 12, 2012) |
|
Amendment
to Registration Rights Agreement |
| |
10.28(Incorporated
by Reference to Exhibit 10.28 of the Company’s Form S-1 filed June 12, 2012) |
|
Termination
Letter February 27 Equity Purchase Agreement |
| |
10.29(zzzzzzzz1) |
|
AGREEMENT
AMENDING TERMS OF OUTSTANDING DEBT BY THE COMPANY |
| |
10.30(zzzzzzzz2) |
|
AGREEMENT
AMENDING TERMS OF OUTSTANDING DEBT BY THE COMPANY |
| |
10.31(zzzzzzzz3) |
|
AGREEMENT
AMENDING TERMS OF OUTSTANDING DEBT BY THE COMPANY |
| |
10.32(zzzzzzzz4) |
|
AGREEMENT
AMENDING TERMS OF OUTSTANDING DEBT BY THE COMPANY |
| |
10.33(zzzzzzzz5) |
|
AGREEMENT
AMENDING TERMS OF OUTSTANDING DEBT BY THE COMPANY |
| |
10.34(zzzzzzzz6) |
|
AGREEMENT
AMENDING TERMS OF OUTSTANDING DEBT BY THE COMPANY |
| |
10.35
( incorporated by Reference to Exhibit 10.35 of the company’s amended Form S-1 Filed August 17, 2012) |
|
ASHER
NOTE $63,000 |
| |
10.36(
incorporated by Reference to Exhibit 10.36 of the company’s amended Form S-1 Filed August 17, 2012) |
|
TERMS
OF COMPENSATION DAVID KOOS |
| |
10.37
(zzzzzzzz7) |
|
ASHER
NOTE $63,000 |
| |
10.38
(incorporated by reference to Exhibit 10.1 of the Company’s 8-K filed December 12, 2012) 3.i (8) (Incorporated by Reference
to Exhibit 3(i) of the Company’s 8-K filed October 19, 2012) |
|
Settlement
Agreement with G. McDonald |
| |
10.39
(incorporated by reference to Exhibit 10.39 of the Company’s 10-K for the year ended August 31, 2012) |
|
Line
of Credit Promissory Note Sherman family Trust |
| |
Exhibit
10.40 SETTLEMENT AGREEMENT AND MUTUAL GENERAL RELEASE (incorporated by reference to exhibit 10.1 of the Company’s 8-K
filed March 12, 2013) |
|
|
| |
3(1)(8)(zzzzzzzz8) |
|
Text
of Amendment to Certificate of Incorporation |
| |
3(i)(9)
(zzzzzzzz9) |
|
Certificate
of Designations |
| |
3(i)
(10) (zzzzzzzz10) |
|
Text
of Amendment to Certificate of Incorporation |
| |
10.41((zzzzzzzz11) |
|
Employment
Offer |
| |
31.1 |
|
CERTIFICATION
OF CHIEF EXECUTIVE OFFICER PURSUANT TO SECTION 302 OF THE SARBANESE-OXLEY ACT OF 2002 |
| |
31.2 |
|
CERTIFICATION
OF CHIEF FINANCIAL OFFICER PURSUANT TO SECTION 302 OF THE SARBANESE-OXLEY ACT OF 2002 |
| |
32.1 |
|
CERTIFICATION
OF CHIEF EXECUTIVE OFFICER PURSUANT TO 18 U.S.C SECTION 1350 AS ADOPTED PURSUANT TO SECTION 906 OF THE SARBANES-OXLEY ACT
OF 2002 |
| |
32.2 |
|
CERTIFICATION
OF CHIEF FINANCIAL OFFICER PURSUANT TO 18 U.S.C SECTION 1350 AS ADOPTED PURSUANT TO SECTION 906 OF THE SARBANES-OXLEY ACT
OF 2002 |
| |
|
|
|
| (a) |
Incorporated
by Reference to Exhibit 3(1) of the Company's Form S-1 Filed November 4, 2008 |
| (b) |
Incorporated
by Reference to Exhibit 3(2) of the Company's Form S-1 Filed November 4, 2008 |
| (
c) |
Incorporated
by Reference to Exhibit 10(1) of the Company's Form10-K Filed November 17, 2009 |
| (d) |
Incorporated
by Reference to Exhibit 10(1) of the Company's Form10-K Filed November 16, 2010 |
| (e) |
Incorporated
by Reference to Exhibit 10(1) of the Company's Form 8-K Filed December 17, 2010 |
| (f) |
Incorporated
by Reference to Exhibit 10(2) of the Company's Form 8-K Filed December 17, 2010 |
| (g) |
Incorporated
by Reference to Exhibit 10(3) of the Company's Form 8-K Filed December 17, 2010 |
| (h) |
Incorporated
by Reference to Exhibit 10(3) of the Company's Form 8-K Filed October 24, 2011 |
| (i) |
Incorporated
by Reference to Exhibit 3(i) of the Company's Form 8-K Filed June 10, 2011 |
| (j) |
Incorporated
by Reference to Exhibit 10(9) of the Company's Form 10-K filed November 3 2011 |
| (k) |
Incorporated
by Reference to Exhibit 10(10) of the Company's Form 10-K filed November 3 2011 |
| (l) |
Incorporated
by Reference to Exhibit 10(11) of the Company's Form 10-K filed November 3 2011 |
| (m) |
Incorporated
by Reference to Exhibit 10(12) of the Company's Form 10-K filed November 3 2011 |
| (n) |
Incorporated
by Reference to Exhibit 3(i) (2)of the Company's Form 8-K Filed June 10, 2011 |
| (o) |
Incorporated
by Reference to Exhibit 3(ii) of the Company's Form 8-K Filed June 10, 2011 |
| (p) |
Incorporated
by Reference to Exhibit 3(i) of the Company's Form 8-K Filed June 10, 2011 |
| (q) |
Incorporated
by Reference to Exhibit 10.1 of the Company's Form 10-Q Filed January 11, 2012 |
| (
r) |
Incorporated
by Reference to Exhibit 3(i) of the Company's Form 8-K Filed February 9, 2012 |
| (s) |
Incorporated
by Reference to Exhibit (3)(i) of the Company's Form 10-Q Filed February 9, 2012 |
| (t) |
Incorporated
by Reference to Exhibit 10.1 of the Company's Form 10-Q Filed February 9, 2012 |
| (u) |
Incorporated
by Reference to Exhibit 10.2 of the Company's Form 10-Q Filed February 9, 2012 |
| (v) |
Incorporated
by Reference to Exhibit 3(i) of the Company's Form 8-K Filed March 22, 2012 |
| (w) |
Incorporated
by Reference to Exhibit 10.1 of the Company's Form 8-K Filed April 5, 2012 |
| (x) |
Filed
as Exhibit 5.1 |
| (y) |
Incorporated
by Reference to Exhibit 14 of the Company's Form10-K Filed November 17, 2009 |
| (z) |
Incorporated
by Reference to Exhibit 10.1 of the Company's Form 10-Q Filed March 28, 2012 |
| (aa) |
Incorporated
by Reference to Exhibit 3(i) of the Company's Form 8-K Filed April 25, 2012 |
| (aaa) |
Incorporated
by Reference to Exhibit 10 of the Company's Form 8-K Filed April 25, 2012 |
| (aaaa) |
Incorporated
by Reference to Exhibit 10.2 of the Company's Form 8-K Filed July 10, 2009 |
| (aaaaa) |
Incorporated
by Reference to Exhibit 10.1 of the Company's Form 8-K Filed June 4, 2012 |
| (zzzzzzzz1) |
Incorporated
by Reference to Exhibit 10.1 of the Company's Form 10-Q Filed July 23, 2012 |
| (zzzzzzzz2) |
Incorporated
by Reference to Exhibit 10.2 of the Company's Form 10-Q Filed July 23, 2012 |
| (zzzzzzzz3) |
Incorporated
by Reference to Exhibit 10.3 of the Company's Form 10-Q Filed July 23, 2012 |
| (zzzzzzzz4) |
Incorporated
by Reference to Exhibit 10.4 of the Company's Form 10-Q Filed July 23, 2012 |
| (zzzzzzzz5) |
Incorporated
by Reference to Exhibit 10.5 of the Company's Form 10-Q Filed July 23, 2012 |
| (zzzzzzzz6) |
Incorporated
by Reference to Exhibit 10.6 of the Company's Form 10-Q Filed July 23, 2012 |
| (zzzzzzzz7) |
Incorporated
by Reference to Exhibit 10.37 of the Company's Form 10-K for the year ended August 31, 2012) |
| (zzzzzzzz8) |
Incorporated
by Reference to Exhibit 3(i) of the company’s Form 8-K dated January 24, 2014 |
| (zzzzzzzz9) |
Incorporated
by Reference to Exhibit 3(i) of the company’s Form 8-K dated May 22, 2014 |
| (zzzzzzzz10) |
Incorporated
by Reference to Exhibit 3(i) of the company’s Form 8-K dated July 14, 2014 |
| (zzzzzzzz11) |
Incorporated
by Reference to Exhibit 10.1 of the company’s Form 8-K dated November 7, 2014 |
SIGNATURES
Pursuant
to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report
to be signed on its behalf by the undersigned, thereunto duly authorized.
| |
|
|
Entest
BioMedical, Inc. |
| |
|
|
|
| |
|
By: |
/s/David
R. Koos |
| |
|
|
Name:
David R. Koos |
| |
|
|
Title:
President, Chairman, Chief
Executive
Officer |
| |
|
|
Date:
November 25, 2014. |
Pursuant
to the requirements of the Securities Exchange Act of 1934, this Report has been signed below by the following persons on behalf
of the Registrant and in the capacities indicated on November 25, 2014.
| |
|
By: |
/s/ David
R. Koos |
| |
|
|
Name:
David R. Koos |
| |
|
|
Title:
President, Chairman of the Board of Directors, Chief Executive Officer, Acting Chief Financial Officer |
| |
|
|
Date:
November 25, 2014. |
EXHIBIT
31.1
CERTIFICATION
OF CHIEF EXECUTIVE OFFICER PURSUANT TO
SECTION
302 OF THE SARBANES-OXLEY ACT OF 2002
I,
David R. Koos, certify that:
1.
I have reviewed this annual report on Form 10-K for the YEAR ended August 31, 2014 of Entest BioMedical, Inc.;
2.
Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary
to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect
to the period covered by this report;
3.
Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all
material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods
presented in this report;
4.
The registrant's other certifying officer(s) and I are responsible for establishing and maintaining disclosure controls and procedures
(as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal controls over financial reporting (as defined in Exchange
Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have:
(a)
Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision,
to ensure that material information relating to the registrant’s, including its consolidated subsidiaries, is made known
to us by others within those entities, particularly during the period in which this report is being prepared;
(b)
Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed
under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of
financial statements for external purposes in accordance with generally accepted accounting principles:
(c)
Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions
about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on
such evaluation; and
(d)
Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the
registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that
has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial
reporting; and
5.
The registrant’s other certifying officer(s) and I have disclosed, based on our most recent evaluation of internal control
over financial reporting, to the registrant’s auditors and the audit committee of registrant’s board of directors
(or persons performing the equivalent function):
(a)
All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which
are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information;
and
(b)
Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s
internal control over financial reporting.
| Dated:
November 25, 2014 |
|
By: |
/s/ David
R. Koos |
| |
|
|
David
R. Koos |
| |
|
|
Chief
Executive Officer |
| |
|
|
|
EXHIBIT
31.2
CERTIFICATION
OF CHIEF FINANCIAL OFFICER PURSUANT TO
SECTION
302 OF THE SARBANES-OXLEY ACT OF 2002
I,
David R. Koos, certify that:
1.
I have reviewed this annual report on Form 10-K for the YEAR ended August 31, 2014 of Entest BioMedical, Inc.;
2.
Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary
to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect
to the period covered by this report;
3.
Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all
material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods
presented in this report;
4.
The registrant's other certifying officer(s) and I are responsible for establishing and maintaining disclosure controls and procedures
(as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal controls over financial reporting (as defined in Exchange
Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have:
(a)
Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision,
to ensure that material information relating to the registrant’s, including its consolidated subsidiaries, is made known
to us by others within those entities, particularly during the period in which this report is being prepared;
(b)
Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed
under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of
financial statements for external purposes in accordance with generally accepted accounting principles:
(c)
Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions
about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on
such evaluation; and
(d)
Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the
registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that
has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial
reporting; and
5.
The registrant’s other certifying officer(s) and I have disclosed, based on our most recent evaluation of internal control
over financial reporting, to the registrant’s auditors and the audit committee of registrant’s board of directors
(or persons performing the equivalent function):
(a)
All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which
are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information;
and
(b)
Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s
internal control over financial reporting.
| Dated:
November 25, 2014 |
|
By: |
/s/ David
R. Koos |
| |
|
|
David
R. Koos |
| |
|
|
Chief
Financial Officer |
| |
|
|
|
EXHIBIT
32.1
CERTIFICATION
OF CHIEF EXECUTIVE OFFICER
PURSUANT
TO 18 U.S.C. SECTION 1350
AS
ADOPTED PURSUANT TO SECTION 906
OF
THE SARBANES-OXLEY ACT OF 2002
In
connection with the Annual Report of Entest BioMedical, Inc. on Form 10-K for the year ended August 31, 2014 as filed with the
Securities and Exchange Commission on the date hereof (the “Report”), I, David R. Koos, Chief Executive Officer certify,
pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, that, to the best of
my knowledge and belief:
(1)
The Report fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934 and
(2)
The information contained in the Report fairly presents, in all material respects, the financial condition and results of operations
of the Company.
| Dated:
November 25, 2014 |
|
By: |
/s/ David
R. Koos |
| |
|
|
David
R. Koos |
| |
|
|
Chief
Executive Officer |
| |
|
|
|
EXHIBIT
32.2
CERTIFICATION
OF CHIEF FINANCIAL OFFICER
PURSUANT
TO 18 U.S.C. SECTION 1350
AS
ADOPTED PURSUANT TO SECTION 906
OF
THE SARBANES-OXLEY ACT OF 2002
In
connection with the Annual Report of Entest BioMedical, Inc. on Form 10-K for the year ended August 31, 2014 as filed with the
Securities and Exchange Commission on the date hereof (the “Report”), I, David R. Koos, Chief Financial Officer certify,
pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, that, to the best of
my knowledge and belief:
(1)
The Report fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934 and
(2)
The information contained in the Report fairly presents, in all material respects, the financial condition and results of operations
of the Company.
| Dated:
November 25, 2014 |
|
By: |
/s/ David
R. Koos |
| |
|
|
David
R. Koos |
| |
|
|
Chief
Financial Officer |
| |
|
|
|
Entest (CE) (USOTC:ETNI)
Historical Stock Chart
From Mar 2024 to Apr 2024
Entest (CE) (USOTC:ETNI)
Historical Stock Chart
From Apr 2023 to Apr 2024