UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
_________________
FORM 8-K
_________________
CURRENT REPORT
Pursuant to Section 13 or 15(d) of
the Securities Exchange Act of 1934
Date
of Report (Date of Earliest Event Reported): October 3, 2014
Ekso Bionics Holdings, Inc.
(Exact
Name of Registrant as specified in its charter)
| Nevada |
333-181229 |
99-0367049 |
(State or Other Jurisdiction of
Incorporation) |
(Commission File Number) |
(IRS Employer
Identification No.) |
1414 Harbour Way South, Suite 1201
Richmond, California 94804
(Address of principal executive offices,
including zip code)
(510) 984-1761
(Registrant’s telephone number, including
area code)
Not Applicable
(Registrant’s name or former address,
if change since last report)
Check the appropriate box below if the
Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions
(see General Instruction A.2. below):
| o | Written communications pursuant to Rule 425 under the
Securities Act (17 CFR 230.425) |
| o | Soliciting material pursuant to Rule 14a-12 under the
Exchange Act (17 CFR 240.14a-12) |
| o | Pre-commencement communications pursuant to Rule 14d-2(b)
under the Exchange Act (17 CFR 240.14d-2(b)) |
| o | Pre-commencement communications pursuant to Rule 13e-4(c)
under the Exchange Act (17 CFR 240.13e-4(c)) |
Item 5.02 Departure of Directors
or Certain Officers; Election of Directors; Appointment of Certain Officers; Compensatory Arrangements of Certain Officers.
Appointment of President and Chief Commercial Officer
On October 8, 2014, the Board of Directors of Ekso
Bionics Holdings, Inc. (the “Company”) appointed Thomas Looby to serve as President and Chief Commercial Officer
of the Company, effective immediately. As President and Chief Commercial Officer, Mr. Looby will be responsible for the daily
operations of the Sales, Marketing, Clinical, Customer Service, and Quality departments of the Company. Mr. Looby will report
directly to Nathan Harding, the Company’s Chief Executive Officer.
Mr. Looby, age 42, joined the Company in April 2014,
serving since then as the Company’s Chief Marketing Officer leading the development and execution of the
Company’s global hospital and rehabilitation marketing strategy. Prior to joining the Company, from September 2006 to
March 2014, Mr. Looby served as Senior Vice President and Chief Marketing Officer at Given
Imaging, where he was responsible for worldwide market development for PillCam® capsule endoscopy and other novel
diagnostic technologies to gastrointestinal diseases. Prior to joining Given Imaging, Mr. Looby also served as Corporate
Director of Marketing and Business Development at Eastman Kodak.
Departure of Directors or Certain Officers
Frank Moreman’s employment as Chief Operating Officer
of the Company terminated effective October 3, 2014.
Item 7.01 Regulation FD Disclosure.
On October 9, 2014, the Company issued a press release announcing
the appointment of Mr. Looby as described above. A copy of the press release is furnished as Exhibit 99.1 to this Current Report
on Form 8-K and is incorporated by reference herein.
The information in this Item 7.01, including the exhibit attached
hereto, is furnished pursuant to Item 7.01 and shall not be deemed “filed” for any other purpose, including for the
purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject
to the liabilities of that Section. The information in this Item 7.01 of this Current Report on Form 8-K shall not be deemed incorporated
by reference into any filing under the Securities Act of 1933 or the Exchange Act regardless of any general incorporation language
in such filing unless specifically provided otherwise.
Item 9.01 Financial Statements and Exhibits
| 99.1 | Press Release dated October 9, 2014. |
SIGNATURE
Pursuant to the requirements
of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the
undersigned thereunto duly authorized.
| |
EKSO BIONICS HOLDINGS, INC. |
|
| |
|
|
| |
By: |
/s/
Nathan Harding |
|
| |
Name: |
Nathan Harding |
|
| |
Title: |
Chief Executive Officer |
|
Dated: October 9, 2014
Exhibit 99.1

FOR IMMEDIATE RELEASE
EKSO BIONICS™ ELEVATES THOMAS
LOOBY TO PRESIDENT AND CHIEF COMMERCIAL OFFICER
Expands Team to Build on Recent
Sales Growth in Medical Division
RICHMOND, Calif., October 9, 2014
-- Ekso Bionics Holdings, Inc. (OTCQB: EKSO), a leading robotic exoskeleton company, announced today the promotion of Thomas
Looby to President and Chief Commercial Officer, effective immediately. Tom will be responsible for expanding global sales
and marketing operations, including overseeing Ekso Bionics' clinical, customer service and regulatory divisions.
"Tom has made great progress
for us towards 'cracking the code', as we call it. That is, to begin laying down a foundation for significant growth over time
in the medical space,” said Nate Harding, Chief Executive Officer and co-founder. "While we are pleased with our recent
quarter on quarter growth, we now wish to make a more focused effort to drive adoption by leading further clinical studies and
systematically educating the market on the economic benefits of using exoskeletons in the rehabilitation setting. Tom's
medical device experience and seasoned leadership skills are poised to make that happen."
Mr. Looby, age 42, joined Ekso in
April 2014, serving since then as the Company’s Chief Marketing Officer leading the development and execution of the Company’s
global hospital and rehabilitation marketing strategy. Prior to joining the Company, from September 2006 to March 2014,
Tom served as Senior Vice President and Chief Marketing Officer at Given Imaging, where he was responsible for worldwide
market development for PillCam® capsule endoscopy and other novel diagnostic technologies for gastrointestinal diseases. Prior
to joining Given Imaging, Tom also served as Corporate Director of Marketing and Business Development at Eastman Kodak.
“Hospital facilities around
the world are demonstrating the benefits of rehabilitation using patented Ekso Bionics robotic exoskeleton technology for people
living with spinal cord injuries, stroke, traumatic brain injuries, MS, and other neurological injuries. Our early successes
in the rehabilitation clinic are very encouraging and we plan to expand our leadership into the home setting where mobility and
associated wellness are of utmost importance. Additionally, we believe the unique, broad-based technology that Ekso Bionics has
developed will satisfy unmet needs in other large markets, including heavy industry and construction – where we are in the
early stages of developing preliminary use cases for able-bodied applications. I am honored to be helping to lead the growth of
this important technology.”
Ekso Bionics also announced the
hiring of Niel McCaffrey as VP of Engineering, Medical. Niel is an expert in complex electronic imaging, diagnostic and communication
systems. At Becton Dickinson Medical, Niel built a team of over 40 highly skilled engineers and scientists to field complex systems;
having earned a reputation for developing both foundation technology and effective strategic intellectual property pipelines,
Niel will be focused on advancing Ekso Bionics’ medical engineering team and intellectual property library.


About Ekso Bionics
Since 2005, Ekso Bionics has been
pioneering the field of robotic exoskeletons, or wearable robots, to augment human strength, endurance and mobility. The company’s
first commercially available product called Ekso has helped thousands of people living with paralysis take millions of steps not
otherwise possible. By designing and creating some of the most forward-thinking and innovative solutions for people looking to
augment human capabilities, Ekso Bionics is helping people rethink current physical limitations and achieve the remarkable.
Ekso Bionics is headquartered in
Richmond, CA and is listed on the OTC QB under the symbol EKSO. To learn more about Ekso Bionics please visit us at www.eksobionics.com
Facebook: www.facebook.com/eksobionics
Twitter: @eksobionics
YouTube: http://www.youtube.com/user/EksoBionics/
###
Media Contact:
Heidi Darling, Marketing Manager
Phone: 415.302.4777
hdarling@eksobionics.com
Investor Contact:
Lauren Glaser, Vice President
Phone: 646.378.2972
lglaser@troutgroup.com
Any statements contained in this
press release that do not describe historical facts may constitute forward-looking statements. Forward-looking statements
may include, without limitation, statements regarding (i) the plans and objectives of management for future operations, including
strategic and other plans or objectives relating to the design, development and commercialization of human exoskeletons, (ii)
a projection of income (including income/loss), earnings (including earnings/loss) per share, capital expenditures, dividends,
capital structure or other financial items, (iii) the Company's future financial performance and (iv) the assumptions underlying
or relating to any statement described in points (i), (ii) or (iii) above. Such forward-looking statements are not meant
to predict or guarantee actual results, performance, events or circumstances and may not be realized because they are based upon
the Company's current projections, plans, objectives, beliefs, expectations, estimates and assumptions and are subject to a number
of risks and uncertainties and other influences, many of which the Company has no control over. Actual results and the timing
of certain events and circumstances may differ materially from those described by the forward-looking statements as a result of
these risks and uncertainties. Factors that may influence or contribute to the inaccuracy of the forward-looking statements
or cause actual results to differ materially from expected or desired results may include, without limitation, the Company's inability
to obtain adequate financing to fund the Company’s operations and necessary to develop or enhance our technology, the significant
length of time and resources associated with the development of the Company’s products, the Company’s failure to achieve
broad market acceptance of the Company’s products, , the failure of our sales and marketing organization or partners to
market our products effectively, adverse results in future clinical studies of the Company’s medical device products,
the failure to obtain or maintain patent protection for the Company’s technology, failure to obtain or maintain regulatory
approval to market the Company’s medical devices, lack of product diversification, volatility in the price of the Company's
raw materials, existing or increased competition, , and the Company's failure to implement the Company's business plans or strategies.
These and other factors are identified and described in more detail in the Company's filings with the SEC at http://www.sec.gov.
The Company does not undertake to update these forward-looking statements.

Ekso Bionics (NASDAQ:EKSO)
Historical Stock Chart
From Mar 2024 to Apr 2024
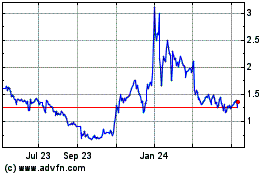
Ekso Bionics (NASDAQ:EKSO)
Historical Stock Chart
From Apr 2023 to Apr 2024