Outlook Therapeutics® Requests Type A Meeting With FDA
September 29 2023 - 8:05AM

Outlook Therapeutics, Inc. (Nasdaq: OTLK), a biopharmaceutical
company working to achieve FDA approval for the first ophthalmic
formulation of bevacizumab for the treatment of retinal diseases,
today announced that a Type A Meeting request has been submitted to
the U.S. Food and Drug Administration (FDA) to discuss the Complete
Response Letter (CRL) dated August 29, 2023 regarding the Biologics
License Application (BLA) for ONS-5010, an investigational
ophthalmic formulation of bevacizumab under development to treat
wet AMD.
In the FDA’s recently issued CRL, although the
Agency acknowledged the NORSE TWO pivotal trial met its safety and
efficacy endpoints, it concluded it could not approve the BLA
during this review cycle due to several CMC issues, open
observations from pre-approval manufacturing inspections, and a
lack of substantial evidence. The Company is committed to working
with the FDA to address its concerns.
“Our belief remains unwavering that the retina
community needs an FDA-approved ophthalmic bevacizumab to deliver
an alternative on-label bevacizumab option for patients with wet
AMD. We look forward to a productive meeting with FDA to discuss
the CRL with the goal of obtaining a clear understanding of the
items that need to be addressed to resubmit our BLA for ONS-5010 at
the earliest opportunity,” said Russell Trenary, President and CEO
of Outlook Therapeutics.
About Outlook Therapeutics,
Inc.
Outlook Therapeutics is a biopharmaceutical
company working to achieve FDA approval for the launch of ONS-5010/
LYTENAVA™ (bevacizumab-vikg) as the first FDA-approved ophthalmic
formulation of bevacizumab for use in retinal indications,
including wet AMD, DME and BRVO. The FDA accepted Outlook
Therapeutics’ BLA submission for ONS-5010 to treat wet AMD with an
initial PDUFA goal date of August 29, 2023; FDA did not approve the
BLA during this review cycle and the Company is working with the
FDA to address the issues that have been raised so that the BLA may
be re-submitted. The submission is supported by Outlook
Therapeutics’ wet AMD clinical program, which consists of three
clinical trials: NORSE ONE, NORSE TWO, and NORSE THREE. If ONS-5010
ophthalmic bevacizumab is approved, Outlook Therapeutics expects to
commercialize it as the first and only FDA-approved ophthalmic
formulation of bevacizumab for use in treating retinal diseases in
the United States, United Kingdom, Europe, Japan, and other
markets. As part of the Company’s multi-year commercial planning
process, Outlook Therapeutics and Cencora, formerly
AmerisourceBergen, entered into a strategic commercialization
agreement to expand the Company’s reach for connecting to retina
specialists and their patients. Cencora will provide third-party
logistics (3PL) services and distribution, as well as
pharmacovigilance services and other services in the United States.
For more information, please visit www.outlooktherapeutics.com.
Forward-Looking Statements
This press release contains forward-looking
statements. All statements other than statements of historical
facts are “forward-looking statements,” including those relating to
future events. In some cases, you can identify forward-looking
statements by terminology such as “anticipate,” “believe,”
“continue,” “could,” “estimate,” “expect,” “may,” “might,”
“intend,” “potential,” “predict,” “should,” or “will,” the negative
of terms like these or other comparable terminology, and other
words or terms of similar meaning. These include, among others,
statements about ONS-5010’s potential as the first FDA-approved
ophthalmic formulation of bevacizumab-vikg, expectations concerning
discussions at a Type A Meeting with the FDA and the results
thereof, expectations concerning the ability to remediate or
otherwise resolve deficiencies identified in the CRL, expectations
concerning decisions of regulatory bodies, including the FDA, and
the timing thereof, expectations concerning the relationship with
Cencora and the benefits and potential expansion thereof and other
statements that are not historical fact. Although Outlook
Therapeutics believes that it has a reasonable basis for the
forward-looking statements contained herein, they are based on
current expectations about future events affecting Outlook
Therapeutics and are subject to risks, uncertainties and factors
relating to its operations and business environment, all of which
are difficult to predict and many of which are beyond its control.
These risk factors include those risks associated with developing
pharmaceutical product candidates, risks of conducting clinical
trials and risks in obtaining necessary regulatory approvals, the
content and timing of the expected Type A Meeting with the FDA, the
content and timing of decisions by the FDA, as well as those risks
detailed in Outlook Therapeutics’ filings with the Securities and
Exchange Commission (the “SEC”), including the Annual Report on
Form 10-K for the fiscal year ended September 30, 2022 as
supplemented by the Quarterly Report on Form 10-Q for the quarter
ended June 30, 2023, in each case as filed with the SEC and future
quarterly reports to be filed with the SEC, which include the
uncertainty of future impacts related to macroeconomic factors,
including as a result of the ongoing conflict between Russia and
Ukraine, high interest rates, inflation and potential future bank
failures on the global business environment. These risks may cause
actual results to differ materially from those expressed or implied
by forward-looking statements in this press release. All
forward-looking statements included in this press release are
expressly qualified in their entirety by the foregoing cautionary
statements. You are cautioned not to place undue reliance on these
forward-looking statements, which speak only as of the date hereof.
Outlook Therap
eutics does not undertake any obligation to
update, amend or clarify these forward-looking statements whether
as a result of new information, future events or otherwise, except
as may be required under applicable securities law.
CONTACTS:
Media Inquiries:Harriet UllmanVice
PresidentLaVoieHealthScienceT:
617.429.5475hullman@lavoiehealthscience.com
Investor Inquiries: Jenene Thomas Chief
Executive Officer JTC Team, LLC T: 833.475.8247
OTLK@jtcir.com
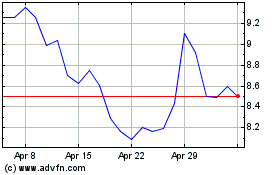
Outlook Therapeutics (NASDAQ:OTLK)
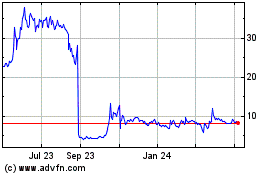
Historical Stock Chart
From Apr 2024 to May 2024
Outlook Therapeutics (NASDAQ:OTLK)
Historical Stock Chart
From May 2023 to May 2024