As filed with the Securities
and Exchange Commission on November 21, 2014
Registration No.
333-_______
UNITED STATES SECURITIES AND
EXCHANGE COMMISSION
WASHINGTON D.C. 20549
____________________________
FORM S-1
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
_____________________________
BIOHEART,
INC.
(Exact name of Registrant as specified in its
charter)
| Florida |
|
8731 |
|
65-0945967 |
| (State or other Jurisdiction |
|
|
|
|
| of
Incorporation or |
|
(Primary Standard Industrial |
|
(I.R.S. Employer |
| Organization) |
|
Classification Code Number) |
|
Identification No.)
|
13794 NW
4th Street,
Suite 212
Sunrise, Florida 33325
(954) 835-1500
(Address, including zip
code, and telephone number including area code, of Registrant’s principal
executive offices)
Mike Tomas, Chief
Executive Officer
Bioheart, Inc.
13794 NW
4th Street,
Suite 212
Sunrise, Florida 33325
(954) 835-1500
(Name, address, including
zip code, and telephone number, including area code, of agent for service)
Copies to:
Jill Arlene Robbins, P.A.
Jill Arlene Robbins
525-93rd Street
Surfside Florida, 33154
Telephone: (305) 531-1174
Facsimile: (305) 531-1274
Email:
jillarlene@jarepa.com
APPROXIMATE DATE OF
COMMENCEMENT OF PROPOSED SALE TO THE PUBLIC:
From time to time after this Registration
Statement becomes effective.
If any of the securities
being registered on this Form are to be offered on a delayed or continuous basis
pursuant to Rule 415 under the Securities Act of 1933, as amended (the
“Securities Act”) check the following box: [X]
1
If this Form is filed to
register additional securities for an offering pursuant to Rule 462(b) under the
Securities Act, please check the following box and list the Securities Act
registration statement number of the earlier effective registration statement
for the same offering. [ ]
If this Form is a
post-effective amendment filed pursuant to Rule 462(c) under the Securities Act,
check the following box and list the Securities Act registration statement
number of the earlier effective registration statement for the same offering.
[ ]
If this Form is a
post-effective amendment filed pursuant to Rule 462(d) under the Securities Act,
check the following box and list the Securities Act registration statement
number of the earlier effective registration statement for the same offering.
[ ]
Indicate by check mark
whether the Registrant is a large accelerated filer, an accelerated filer, a
non-accelerated filer, or a small reporting company. See definitions of “large
accelerated filer,” “accelerated filer,” and “smaller reporting company” in Rule
12b-2 of the Exchange Act:
| Large accelerated filer [ ] |
Accelerated filer [ ] |
| Non-accelerated filer [ ] |
Smaller reporting company [ x ] |
| (Do
not check if a smaller reporting company) |
|
2
CALCULATION OF
REGISTRATION FEE
|
|
Proposed |
Proposed |
|
|
|
Maximum |
Maximum |
|
| Title of each class
of securities |
Amount to be |
Offering Price Per |
Aggregate |
Amount
of |
| to be Registered |
Registered
(1) |
Share |
Offering
Price |
Registration Fee |
| Common Stock, $.001 par
value |
87,812,591(2) |
$0.0161(6) |
$1,413,783 |
$164.28 |
| Common Stock, $.001 par
value |
31,000,000(3) |
$0.0161(6) |
$499,100 |
$58.00 |
| Common Stock, $.001 par
value |
9,109,128 (4) |
$0.0161(6) |
$146,657 |
$17.04 |
| Common Stock, $.001 par
value |
15,890,872(5) |
$0.0161(6) |
$255,843 |
$29.73 |
| Total |
143,812,591 |
$0.0161(6) |
$2,315,383 |
$269.05 |
| |
(1) |
|
Represents shares
offered by the Selling Stockholder. Includes an indeterminable number of
additional shares of Common Stock, pursuant to Rule 416 under the
Securities Act that may be issued to prevent dilution from stock splits,
stock dividends or similar transaction that could affect the shares to be
offered by Selling Stockholder. |
|
(2) |
|
Consists of
87,812,591shares of Common Stock that we may issue to the Selling
Stockholder pursuant to draw downs under the Purchase Agreement with the
Selling Stockholder. |
|
(3) |
|
Consists of
31,000,000 shares of Common Stock issuable on conversion of convertible
promissory note held by the Selling Stockholder. |
|
(4) |
|
Consists of 9,109,128
shares of Common Stock issued to the Selling Stockholder as Initial
Commitment Shares as of October 27, 2014. |
|
(5) |
|
Consists of
15,890,872 shares of Common Stock issuable to the Selling Stockholder as
Additional Commitment Shares. |
|
(6) |
|
Estimated solely for
purposes of calculating the registration fee in accordance with Rule 457
under the Securities Act, using the average of the high and low prices as
reported on the OTC Markets marketplace on November 20th,
2014. |
The Registrant hereby
amends this Registration Statement on such date or dates as may be necessary to
delay its effective date until the Registrant shall file a further amendment
which specifically states that this Registration Statement shall thereafter
become effective in accordance with Section 8(a) of the Securities Act or until
this Registration Statement shall become effective on such date as the
Commission, acting pursuant to said Section 8(a), may determine.
3
The information in this
Prospectus is not complete and may be changed. The Selling Stockholder may not
sell these securities until the Registration Statement filed with the Securities
and Exchange Commission is effective. This Prospectus is not an offer to sell
these securities and it is not soliciting an offer to buy these securities in
any state where the sale is not permitted.
PRELIMINARY PROSPECTUS,
SUBJECT TO COMPLETION, DATED ________, 2014
BIOHEART, INC.
143,813,591 Shares of
Common Stock (includes 9,109,128 issued October 27, 2014)
This prospectus relates to
the resale of up to 143,813,591 shares (includes 9,109,128 issued October 27,
2014) of our common stock, which may be offered by the
selling stockholder, Magna Equities II, LLC, a New York limited liability
company, or Magna. The shares of common stock being offered by the selling
stockholder are issuable (i) upon conversion of a senior convertible note in the
principal amount of $307,500, or the Convertible Note, that we issued to Magna
on October 7, 2014 and (ii) pursuant to a common stock purchase agreement dated
as of October 23, 2014 between us and Magna, or the Purchase
Agreement.
We are not selling any
securities under this prospectus and will not receive any of the proceeds from
the resale of shares of our common stock by the selling stockholder under this
prospectus, however, we have received gross proceeds of $205,000 from the sale
of the Convertible Note to Magna and we may receive gross proceeds of up to
$3,000,000 from sales of our common stock to Magna under the Purchase Agreement.
Magna may offer all or part
of the shares for resale from time to time through public or private
transactions, at either prevailing market prices or at privately negotiated
prices. We provide more information about how Magna may sell its shares of
common stock in the section titled “Plan of Distribution” on page 48. We will
pay the expenses incurred in connection with the offering described in this
prospectus, with the exception of brokerage expenses, fees, discounts and
commissions, which will be paid by the selling stockholder. Of 143,812,591
shares we issued 9,109,128 shares of our common stock to Magna as an initial
commitment fee for entering into the Purchase Agreement and we may issue
additional commitment shares to Magna under certain circumstances described in
this Prospectus. With respect to the shares of common stock that have been and
may be issued pursuant to the Purchase Agreement, Magna is an “underwriter”
within the meaning of Section 2(a) (11) of the Securities Act of 1933, as
amended, or the Securities Act, and with respect to any other shares of common
stock, Magna may be deemed to be an “underwriter” within the meaning of Section
2(a) (11) of the Securities Act.
Our common stock is quoted
on the OTCQB Marketplace operated by OTC Markets Group Inc., or the OTCQB, under
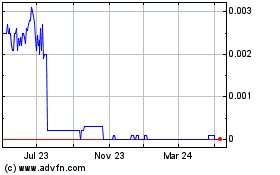
the symbol “BHRT”. The last reported closing price of our common stock on the
OTCQB on October 20, 2014 was $0.0159 per share.
Investing in our common
stock involves a high degree of risk. Please see the sections entitled “Risk
Factors” on page 16 of this prospectus and “Part I—Item 1A Risk Factors” in our
Annual Report on Form 10-K for the year ended December 31, 2013.
Neither the Securities
and Exchange Commission nor any state securities commission has approved or
disapproved of these securities, or determined if this prospectus is truthful or
complete. Any representation to the contrary is a criminal offense.
The date of this
prospectus is [_________], 2014.
You should rely only on the
information contained in this prospectus. We have not authorized any dealer,
salesperson or other person to provide you with information concerning us,
except for the information contained in this prospectus. The information
contained in this prospectus is complete and accurate only as of the date on the
front cover page of this prospectus, regardless of when the time of delivery of
this prospectus or the sale of any Common Stock occurs. The Selling Stockholder
may not sell the securities until the Registration Statement filed with the
Securities and Exchange Commission is effective. This prospectus is not an offer
to sell, nor is it a solicitation of an offer to buy, the Common Stock in any
jurisdiction in which the offer or sale is not permitted.
4
BIOHEART,
INC.
TABLE OF CONTENTS
| PART I: INFORMATION REQUIRED IN PROSPECTUS |
|
| Prospectus Summary |
6 |
| Risk
Factors |
16 |
| Cautionary Note Regarding
Forward-Looking Statements |
39 |
| Use
of Proceeds |
39 |
| Determination of Offering
Price |
40 |
| Selling Stockholder |
48 |
| Plan of Distribution |
48 |
| Description of Securities to be Registered |
73 |
| Interests of Named Experts and
Counsel |
74 |
| Legal Matters |
75 |
| Other Experts |
75 |
| Properties |
75 |
| Legal Proceedings |
76 |
| Market for Common Equity and Related Stockholder
Matters |
77 |
| Management’s Discussion and
Analysis of Financial Condition and Results of Operations |
77 |
| Directors and Executive Officers |
93 |
| Executive Compensation |
97 |
| Security Ownership of Certain Beneficial Owners and
Management |
102 |
| Certain Relationships, Related
Transactions and Director Independence |
105 |
| Disclosure of Commission Position on Indemnification for
Securities Act Liabilities |
107 |
| Where You Can Find More
Information |
107 |
| Index to Financial Statements |
108 |
| |
| PART II – INFORMATION NOT REQUIRED IN
PROSPECTUS |
110 |
| Other Expenses of Issuance and
Distribution |
110 |
| Indemnification of Directors and Officers |
110 |
| Recent Sales of Unregistered
Securities |
111 |
| Exhibits |
112 |
| Undertakings |
116 |
| Signatures |
119 |
You may only rely on the
information contained in this prospectus or that we have referred you to via
this prospectus. We have not authorized anyone to provide you with different or
further information. This prospectus does not constitute an offer to sell or a
solicitation of an offer to buy any securities other than the Common Stock
offered by this prospectus. This prospectus does not constitute an offer to sell
or a solicitation of an offer to buy any Common Stock in any circumstances in
which such offer or solicitation is unlawful. Neither the delivery of this
prospectus nor any sale made in connection with this prospectus shall, under any
circumstances, create any implication that there has been no change in our
affairs since the date of this prospectus or that the information contained
herein by reference thereto in this prospectus is correct as of any time after
its date.
5
PROSPECTUS SUMMARY
The following summary
highlights selected information contained in this prospectus. This summary does
not contain all the information you should consider before investing in the
Common Stock. Before making an investment decision, you should read the entire
prospectus carefully, including the "RISK FACTORS" section, the financial
statements and the notes to the financial statements. As used throughout this
Registration Statement and prospectus, the term “Registrant” refers to Bioheart,
Inc. and the terms "Company", "we," "us," or "our" refer to Bioheart, Inc. and
its consolidated subsidiaries unless the context otherwise requires.
Our Company
Overview
We are a biotechnology
company focused on the discovery, development and, subject to regulatory
approval, commercialization of autologous cell therapies for the treatment of
chronic and acute heart damage. Our lead product candidate is MyoCell, an
innovative clinical therapy designed to populate regions of scar tissue within a
patient’s heart with autologous muscle cells, or cells from a patient’s body,
for the purpose of improving cardiac function in chronic heart failure
patients.
Biotechnology Product
Candidates
Specific to biotechnology,
we are focused on the discovery, development and, subject to regulatory
approval, commercialization of autologous cell therapies for the treatment of
chronic and acute heart damage and peripheral vascular disease. MyoCell is a
clinical muscle-derived cell therapy designed to populate regions of scar tissue
within a patient’s heart with new living cells for the purpose of improving
cardiac function in chronic heart failure patients. Our most recent clinical
trials of MyoCell include the SEISMIC Trial, a completed 40-patient, randomized,
multicenter, controlled, Phase II-a study conducted in Europe and the MYOHEART
Trial, a completed 20-patient, multicenter, Phase I dose-escalation trial
conducted in the United States. We were approved by the U.S. Food and Drug
Administration (the “FDA”) to proceed with a 330-patient, multicenter Phase
II/III trial of MyoCell in North America and Europe (the “MARVEL Trial”). We
completed the MyoCell implantation procedure on the first patient in the MARVEL
Trial on October 24, 2007. Thus far, 20 patients, including 6 control patients,
have been treated. Initial results for the 20 patients were released at the
Heart Failure Society of American meeting in September, 2009, showing a
significant (35%) improvement in the 6 minute walk for those patients who were
treated, and no improvement for those who received a placebo. We are planning,
on the basis of these results, to request the FDA to consider the MARVEL Trial a
pivotal trial (pivotal from Phase II to Phase III) and to reduce the number of
patients in the trial to 150. No assurances can be provided that this request
will be approved. The SEISMIC, MYOHEART and MARVEL Trials have been designed to
test the safety and efficacy of MyoCell in treating patients with severe,
chronic damage to the heart. Upon regulatory approval of MyoCell, we intend to
generate revenue in the United States from the sale of MyoCell cell-culturing
services for treatment of patients by qualified physicians. Abroad, we are
identifying centers where it is already acceptable to use the Myocell treatment
so that greater numbers of patients with this problem can have access to
treatment.
We received approval from
the FDA in July of 2009 to conduct a Phase I safety study on 15 patients of a
combined therapy (Myocell with SDF-1), which we believe is the first approval of
a study combining gene and cell therapies. We initially commenced work on this
study, called the REGEN trial, during the first quarter of 2010. We suspended
activity on the trial in 2010 while seeking additional funding necessary to
conduct the trial. Work on the trial was reinitiated in 2011. Based on the
results of the trial, we intend to either incorporate the combined treatment
into the Marvel Trial, or continue with the Marvel Trial based on the use of
Myocell alone.
We are seeking to secure
sufficient funds to reinitiate enrollment in the MARVEL and REGEN trials. If we
successfully secure such funds, we intend to re-engage a contract research
organization, or CRO, investigators and certain suppliers to advance such
trials. We have initiated and enrolled our first patient in the MIRROR trial in
2013. The trial is very similar to the MARVEL trial but focusses on sites
outside the US. We will continue enrollment in the MIRROR trial once we have
secured sufficient funds.
6
In our pipeline, we have
multiple product candidates for the treatment of heart damage, including
autologous, adipose cell treatment for acute heart damage, chronic ischemia and
critical limb ischemia.
We hope to demonstrate that our
various product candidates are safe and effective complements to existing
therapies for chronic and acute heart damage as well as peripheral arterial
disease.
MyoCell/MyoCell
SDF-1
MyoCell is a clinical
therapy intended to improve cardiac function for those with congestive heart
failure and is designed to be utilized months or even years after a patient has
suffered severe heart damage due to a heart attack or other cause. We believe
that MyoCell has the potential to become a leading treatment for severe, chronic
damage to the heart due to its perceived ability to satisfy, at least in part,
what we believe to be an unmet demand for more effective and/or more affordable
therapies for chronic heart damage. MyoCell uses myoblasts, cells that are
precursors to muscle cells, from the patient’s own body. The myoblasts are
removed from a patient’s thigh muscle, isolated, grown through our proprietary
cell culturing process, and injected directly in the scar tissue of a patient’s
heart. A qualified physician performs this minimally invasive procedure using an
endoventricular catheter. We entered into an agreement with a Johnson &
Johnson company to use its NOGA® Cardiac Navigation System along with its
MyoStar™ injection catheter for the delivery of MyoCell in the MARVEL Trial.
When injected into scar
tissue within the heart wall, myoblasts have been shown to be capable of
engrafting in the damaged tissue and differentiating into mature skeletal muscle
cells. In a number of clinical and animal studies, the engrafted skeletal muscle
cells have been shown to express various proteins that are important components
of contractile function. By using myoblasts obtained from a patient’s own body,
we believe MyoCell is able to avoid certain challenges currently faced by other
types of cell-based clinical therapies including tissue rejection and instances
of the cells differentiating into cells other than muscle. Although a number of
therapies have proven to improve the cardiac function of a damaged heart, no
currently available treatment, to our knowledge, has demonstrated an ability to
generate new muscle tissue within the scarred regions of a heart.
We believe the market for
treating patients in NYHA Class II or NYHA Class III heart failure is
significant. According to the AHA Statistics and the European Society of
Cardiology Task Force for the Treatment of Chronic Heart Failure, in the United
States and Europe there are approximately 5.2 million and 9.6 million,
respectively, patients with heart failure. The AHA Statistics further indicate
that, after heart failure is diagnosed, the one-year mortality rate is high,
with one in five dying and that 80% of men and 70% of women under age 65 who
have heart failure will die within eight years. We believe that approximately
60% of heart failure patients are in either NYHA Class II or NYHA Class III
heart failure based upon a 1999 study entitled “Congestive Heart Failure Due to
Diastolic or Systolic Dysfunction – Frequency and Patient Characteristics in an
Ambulatory Setting” by Diller, PM, et. al.
MyoCell SDF-1 is intended
to be an improvement to MyoCell. MyoCell SDF-1 is similar to MyoCell except that
the myoblast cells to be injected for use in MyoCell SDF-1 will be modified
prior to injection by an adenovirus vector or non-viral vector so that they will
release extra quantities of the SDF-1 protein, which expresses angiogenic
factors. AdipoCell is a patient-derived cell therapy proposed for the treatment
of acute myocardial infarction, chronic heart ischemia, and lower limb ischemia.
We hope to demonstrate that these product candidates are safe and effective
complements to existing therapies for chronic and acute heart damage.
MyoCath
The MyoCath was developed
by Bioheart co-founder Robert Lashinski specifically for delivering new cells to
damaged tissue. It is a deflecting tip needle injection catheter that has a
larger needle which is 25 gauge for better flow rates and less leakage than
systems that are 27 gauge. This larger needle allows for thicker compositions to
be injected which helps with cell retention in the heart. Also, the MyoCath
needle has more fluoroscopic brightness than the normally used nitinol needle,
enabling superior visualization during the procedure. Seeing the needle well
during injections enables the physician who is operating the catheter to
pinpoint targeted areas more precisely, thus improving safety. The MyoCath competes well with other biological
delivery systems on price and efficiency and allows the physician to utilize
standard fluoroscopy and echo equipment found in every cath lab. The MyoCath is
used to inject cells into cardiac tissue in therapeutic procedures to treat
chronic heart ischemic and congestive heart failure. We are currently utilizing
a contract manufacturer to produce catheters as needed.
7
AdipoCell
Bioheart has successfully
completed various trials using adipose stem cells. We have completed the Phase 1 Angel Trial for AdipoCell
(adipose derived stem cells). Five patients were enrolled and treated in the
second quarter of 2013. At the twelve (12) month time point, patients
demonstrated a statistically significant average improvement in ejection
fraction (EF) by echocardiogram.
At the three (3) month time
point, 100% of the patients demonstrated either improvement or stayed the same.
After three (3) months, patients showed an average absolute improvement of 3
percentage points in EF. The patients continued to improve from 3 months to 6
months with a statistically significant average absolute improvement of 10
percentage points (p=0.01) and at the 12 month follow up patients showed this
same level of improvement (p=0.01) .
We have also initiated
several Institutional Review Board studies in 2013 using adipose derived stem
cells for various indications including dry macular degeneration, degenerative
disc disease, erectile dysfunction and chronic obstructive pulmonary disease.
In the second quarter of
2014, we announced the treatment of a patient in Honduras with congestive heart
failure using AdipoCell and MyoCell. This was the first patient treated in the
world using a combination of stem cells.
We have begun two clinical
trials in India. The first cardiac patient has successfully been enrolled and
treated in India using AdipoCell™ or adipose derived stem cells. The second
trial will involve the combination of AdipoCell and MyoCell® or muscle derived
stem cells for congestive heart failure patients. These trials are active and
ongoing
Trading Market
Our common stock, par value
$0.001 per share, commenced trading on February 19, 2008, on the NASDAQ Global
Market under the symbol “BHRT”. Effective June 11, 2008, we transferred the
listing of our common stock to the NASDAQ Capital Market and then, during the
course of 2009 our stock was delisted from NASDAQ and commenced quotation on the
OTC Markets under the symbol “BHRT.QB”.
Corporate Information
We were incorporated in
Florida in 1999 under the name Bioheart, Inc. and continue to be a Florida
corporation.
Our principal executive
offices are located at 13794 NW 4th Street, Suite 212, Sunrise,
Florida 33325. Our telephone number is (954) 835-1500.
Our website address is
www.bioheartinc.com. Our
website and the information contained on our website are not incorporated into
this prospectus or the Registration Statement of which it forms a part. Further,
our references to the URLs for our website are intended to be inactive textual
references only.
8
Recent
Developments
Senior Convertible
Note Financing with Magna Equities II, LLC
Note Purchase Agreement
and Convertible Note
On October 7, 2014, we entered into a securities purchase
agreement with Magna, which we refer to as the Note Purchase Agreement. The Note
Purchase Agreement provides that, upon the terms and subject to the conditions
set forth in the Note Purchase Agreement, Magna will purchase from us the
Convertible Note with an initial principal amount of $307,500 for a purchase
price of $205,000, representing an approximately 33.33% original issue discount.
We issued the Convertible Note to Magna on October 7, 2014.
$40,000 of the outstanding principal amount of the Convertible
Note (together with any accrued and unpaid interest with respect to such portion
of the principal amount) will be automatically extinguished (without any cash
payment by us) upon the filing of the registration statement of which this
prospectus is a part, following the closing of the Note Purchase Agreement . In
addition, $62,500 of the outstanding principal amount of the Convertible Note
(together with any accrued and unpaid interest with respect to such portion of
the principal amount) will be automatically extinguished (without any cash
payment by us) if (i) the registration statement of which this prospectus is a
part is declared effective by the SEC on or prior to the earlier of (A) the
120th calendar day after October 7, 2014 and (B) the fifth business
day after the date we are notified by the Securities and Exchange Commission, or
the Commission, that the registration statement will not be reviewed or will not
be subject to further review, and this prospectus is available for use by Magna
for the resale by Magna of all of the shares of our common stock issued or
issuable upon conversion of the Convertible Note and (ii) no event of default
under the Convertible Note or an event that with the passage of time or giving
of notice would constitute an event of default under the Convertible Note has
occurred on or prior to such date.
The Convertible Note
matures on August 7, 2015 and, in addition to the approximately 33.33% original
issue discount, accrues interest at the rate of 12% per year. The Convertible
Note is convertible at any time, in whole or in part, at Magna’s option into
shares of our common stock at a fixed conversion price of $0.01035 per share,
subject to adjustment pursuant to the “full ratchet” and standard anti-dilution
provisions contained in the Convertible Note. This conversion price represents a
discount of approximately 55% from the lowest trading price our common stock
during the five trading days prior to October 7, 2014, the date we issued the
Convertible Note to Magna. At no time will Magna be entitled to convert any
portion of the Convertible Note to the extent that after such conversion, Magna
(together with its affiliates) would beneficially own more than 4.99% of our
common stock (as calculated pursuant to Section 13(d) of the Securities Exchange
Act of 1934, as amended, or the Exchange Act, and the rules and regulations
thereunder).
The Convertible Note
includes customary event of default provisions, and provides for a default
interest rate of 18%. Upon the occurrence of an event of default, Magna may
require us to pay in cash the “Event of Default Redemption Price” which is
defined in the Convertible Note to mean the greater of (i) the product of (A)
the amount to be redeemed multiplied by (B) 140% (or 100% if an insolvency
related event of default) and (ii) the product of (X) the conversion price in
effect at that time multiplied by (Y) the product of (1) 140% (or 100% if an
insolvency related event of default) multiplied by (2) the greatest closing sale
price of our common stock on any trading day during the period commencing on the
date immediately preceding such event of default and ending on the date we make
the entire payment required to be made under this provision.
We have the right at any
time to redeem all, but not less than all, of the total outstanding amount then
remaining under the Convertible Note in cash at a price equal to 140% of the
total amount of the Convertible Note then outstanding.
The Note Purchase Agreement
contains customary representations, warranties and covenants by, among and for
the benefit of the parties. We also agreed to pay up to $30,000 of reasonable
attorneys' fees and expenses incurred by Magna in connection with the
transaction. The Note Purchase Agreement also provides for indemnification of
Magna and its affiliates in the event that Magna incurs losses, liabilities,
obligations, claims, contingencies, damages, costs and expenses related to a
breach by us of any of our representations, warranties or covenants under the
Note Purchase Agreement.
The issuance of the
Convertible Note to Magna under the Note Purchase Agreement was exempt from the
registration requirements of the Securities Act pursuant to the exemption for
transactions by an issuer not involving any public offering under Section 4(a)
(2) of the Securities Act and Rule 506 of Regulation D promulgated under the Securities Act.
9
Note Registration Rights
Agreement
In connection with the
execution of the Note Purchase Agreement, on October 7, 2014, Magna and we also
entered into a registration rights agreement, which we refer to as the Note
Registration Rights Agreement. Pursuant to the Note Registration Rights
Agreement, we agreed to file the registration statement of which this prospectus
is a part with the Commission to register for resale 31,000,000 shares of our
common stock into which the Convertible Note may be converted, and have it
declared effective at the earlier of (i) the 120th calendar day after
October 7, 2014 and (ii) the fifth business day after the date we are notified
by the Commission that the registration statement will not be reviewed or will
not be subject to further review.
We have agreed to file with
the Commission one or more additional registration statements to cover all of
the securities required to be registered under the Note Registration Rights
Agreement that are not covered by this prospectus, in each case, as soon as
practicable, but in no event later than the applicable filing deadline for such
additional registration statements as provided in the Note Registration Rights
Agreement.
We also agreed, among other
things, to indemnify Magna from certain liabilities and fees and expenses of
Magna incident to our obligations under the Note Registration Rights Agreement,
including certain liabilities under the Securities Act. Magna has agreed to
indemnify and hold harmless us and each of our directors, officers and persons
who control us against certain liabilities that may be based upon written
information furnished by Magna to us for inclusion in the registration statement
of which this prospectus is a part, including certain liabilities under the
Securities Act.
Equity Enhancement
Program with Magna Equities II, LLC
Common Stock Purchase
Agreement
On October 23, 2014, which
we refer to as the Closing Date, we entered into the Purchase Agreement with
Magna. The Purchase Agreement provides that, upon the terms and subject to the
conditions set forth therein, Magna is committed to purchase up to $3,000,000,
which we refer to as the Total Commitment, worth of our common stock, which we
refer to as the Shares, over the 24-month term of the Purchase Agreement.
From time to time over the
term of the Purchase Agreement, commencing on the trading day immediately
following the date on which the registration statement of which this prospectus
is a part is declared effective by the Commission, we may, in our sole
discretion, provide Magna with a draw down notice, each referred to as a Draw
Down Notice, to purchase a specified dollar amount of Shares, which we refer to
as the Draw Down Amount, with each draw down subject to the limitations
discussed below. The maximum dollar amount of Shares requested to be purchased
pursuant to any single Draw Down Notice cannot exceed the lesser of (i) 300% of
the average daily trading volume of our common stock for the 10 trading days
immediately preceding the date of the Draw Down Notice and (ii) $500,000, which
we refer to as the Maximum Draw Down Amount. We may not deliver any Draw Down
Notice to Magna if the Initial Purchase Price (described below) with respect to
the Shares subject to such Draw Down Notice is less than $0.0025 (subject to
adjustment for any stock splits, stock combinations, stock dividends,
recapitalizations and other similar transactions) as of the date the applicable
Draw Down Notice is received by Magna, which we refer to as the Draw Down
Exercise Date.
Once presented with a Draw
Down Notice, Magna is required to purchase the applicable Draw Down Amount at
the applicable “Purchase Price”, which is defined in the Purchase Agreement as
the lesser of:
- the Initial Purchase Price, which is defined as a price equal to 93% of
the lowest of (i) the arithmetic average of the three lowest daily volume
weighted average prices for our common stock, or VWAP, during the 10
consecutive trading days ending on the trading day immediately preceding the
applicable Draw Down Exercise Date, (ii) the arithmetic average of the three
lowest closing sale prices for our common stock during the 10 consecutive
trading days ending on the trading day immediately preceding the applicable Draw Down Exercise Date
and (iii) the closing sale price for our common stock on the trading day
immediately preceding the applicable Draw Down Exercise Date (in each case, to
be appropriately adjusted for any stock splits, stock combinations, stock
dividends, recapitalizations and other similar transactions); and
10
- the True-Up Purchase Price, which is
defined as a price equal to 93% of the arithmetic average of the three lowest
daily VWAPs during the applicable True-Up Pricing Period (described below) (to
be appropriately adjusted for any stock splits, stock combinations, stock
dividends, recapitalizations and other similar transactions), provided that in
no event will the True-Up Purchase Price be less than $0.001.
Accordingly, because the
Purchase Price for Shares subject to any Draw Down Notice is the lesser of the
Initial Purchase Price and the True-Up Purchase Price, and since we cannot
deliver any Draw Down Notice to Magna if the Initial Purchase Price would be
less than $0.0025, the Purchase Price for Shares subject to any Draw Down Notice
will never be less than $0.001 per Share.
The applicable settlement
date, or Settlement Date, with respect to a Draw Down Notice will occur within
one trading day following the Draw Down Exercise Date. On the applicable
Settlement Date for a draw down, we will issue to Magna a number of Shares,
rounded to the nearest whole Share, equal to (i) the Draw Down Amount that we
requested from Magna, divided by (ii) the applicable Initial Purchase Price,
against simultaneous payment by Magna to us in an amount equal to (A) the number
of Shares we issued to Magna on the Settlement Date, multiplied by (B) the
applicable Initial Purchase Price.
With respect to a Draw Down
Notice, on the trading day, which we refer to as the True-Up Date, immediately
following the eight-consecutive trading day period commencing on the trading day
immediately following the applicable Settlement Date for such Draw Down Notice,
which we refer to as the True-Up Pricing Period, a calculation of the True-Up
Purchase Price and the Purchase Price will occur. On the Trading Day immediately
following the True-Up Date, which we refer to as the True-Up Settlement Date, we
will issue to Magna the Additional Shares, if any, in respect of the applicable
Draw Down Notice. “Additional Shares” is defined in the Purchase Agreement as a
number of Shares (to be appropriately adjusted for any stock splits, stock
combinations, stock dividends, recapitalizations and other similar
transactions), rounded to the nearest whole Share, equal to the greater of (I)
zero and (II) the difference of (i) the quotient of (x) the total aggregate
purchase price for Shares we received on the Settlement Date with respect to the
applicable Draw Down Notice divided by (y) the True-Up Purchase Price, less (ii)
the number of Shares we issued to Magna on the applicable Settlement Date with
respect to the applicable Draw Down Notice. Magna is not required to return any
Shares to us in the event the True-Up Purchase Price is greater than the Initial
Purchase Price.
We are prohibited from
issuing a Draw Down Notice if (i) the amount requested in such Draw Down Notice
exceeds the Maximum Draw Down Amount, (ii) the sale of Shares pursuant to such
Draw Down Notice would cause us to issue or sell or Magna to acquire or purchase
an aggregate dollar value of Shares that would exceed the Total Commitment, or
(iii) the sale of Shares pursuant to the Draw Down Notice would cause us to sell
or Magna to purchase an aggregate number of shares of our common stock which
would result in beneficial ownership by Magna of more than 9.99% of our common
stock (as calculated pursuant to Section 13(d) of the Exchange Act, and the
rules and regulations thereunder). We cannot make more than one draw down during
the period commencing on any Draw Down Exercise Date and ending on the
applicable True-Up Date for such draw down, and we must allow at least two
trading days to elapse between the applicable True-Up Date for a draw down and
the delivery of any Draw Down Notice for any other draw down.
Magna has agreed that
during the term of the Purchase Agreement, neither Magna nor any of its
affiliates will, directly or indirectly, engage in any short sales involving our
securities or grant any option to purchase, or acquire any right to dispose of
or otherwise dispose for value of, any of our common stock or any securities
convertible into or exercisable or exchangeable for our common stock, or enter
into any swap, hedge or other similar agreement that transfers, in whole or in
part, the economic risk of ownership of any of our common stock.
11
The Purchase Agreement contains customary representations, warranties and
covenants by, among and for the benefit of the parties. The Purchase Agreement
may be terminated at any time by the mutual written consent of the parties.
Unless earlier terminated, the Purchase Agreement will terminate automatically
on the earliest to occur of (i) the first day of the month next following the
24-month anniversary of the date on which the registration statement of which
this prospectus is a part is declared effective by the Commission, (ii) the date
on which Magna purchases the Total Commitment worth of common stock under the
Purchase Agreement and (iii) the date on which our common stock ceases to be
listed or quoted on an eligible trading market under the Purchase Agreement.
Under certain circumstances set forth in the Purchase Agreement, we and Magna
each may terminate the Purchase Agreement on one trading day’s prior written
notice to the other, without fee, penalty or cost.
We paid to Magna a commitment fee for entering into the Purchase
Agreement equal to $150,000 (or 5.0% of the Total Commitment under the Purchase
Agreement) in the form of 9,109,128 restricted shares of our common stock, which
we refer to as the Initial Commitment Shares, calculated using a per share price
of $0.016467, representing the arithmetic average of the three lowest daily
VWAPs during the 10-consecutive-trading day period immediately preceding the
Closing Date. In addition, promptly following the effective date of the
registration statement of which this prospectus is a part, we are required to
issue to Magna additional shares of our common stock, which we refer to as the
Additional Commitment Shares, equal to the greater of (i) zero and (ii) the
difference of (a) the quotient of (x) $150,000 divided by (y) the greater of (1)
the arithmetic average of the three lowest daily VWAPs during the
10-consecutive-trading day period ending on and including the effective date of
the registration statement of which this prospectus is a part and (2) $0.006,
less (ii) 9,109,128, provided that in no event will we issue more than an
aggregate of 15,890,872 shares of our common stock, subject to adjustment for
any stock splits, stock combinations, stock dividends, recapitalizations and
other similar transactions, as Additional Commitment Shares. The Initial
Commitment Shares, together with 15,890,872 Additional Commitment Shares, are
being registered for resale in the registration statement of which this
prospectus is a part. We sometimes in this prospectus refer to the Initial
Commitment Shares and the Additional Commitment Shares, collectively, as the
Commitment Shares.
We also agreed to pay up to $25,000 of reasonable attorneys' fees and
expenses (exclusive of disbursements and out-of-pocket expenses) incurred by
Magna in connection with the preparation, negotiation, execution and delivery of
the Purchase Agreement and related transaction documentation. Further, if we
issue a Draw Down Notice and fail to deliver the shares to Magna on the
applicable Settlement Date or True-Up Settlement Date, we agreed to pay Magna,
in addition to all other remedies available to Magna under the Purchase
Agreement, an amount in cash equal to 2.0% of the purchase price of such shares
for each 30-day period the shares are not delivered, plus accrued interest.
The Purchase Agreement also provides for indemnification of Magna and its
affiliates in the event that Magna incurs losses, liabilities, obligations,
claims, contingencies, damages, costs and expenses related to a breach by us of
any of our representations and warranties under the Purchase Agreement or the
other related transaction documents or any action instituted against Magna or
its affiliates due to the transactions contemplated by the Purchase Agreement or
other transaction documents, subject to certain limitations.
Registration Rights Agreement
In connection with the execution of the Purchase Agreement, on the
Closing Date, we and Magna also entered into a registration rights agreement
dated as of the Closing Date, which we refer to as the Registration Rights
Agreement. Pursuant to the Registration Rights Agreement, we agreed to file the
registration statement of which this prospectus is a part with the Commission to
register for resale 143,812,591 shares of our common stock, which includes the
9,109,128 issued on October 27, 2014 as Initial Commitment Shares and 15,890,872
Additional Commitment Shares, on or prior to December 8, 2014, which we refer to
as the Filing Deadline, and have it declared effective at the earlier of (A) the
90th calendar day after the earlier of (1) the Filing Deadline and
(2) the date on which the registration statement of which this prospectus is a
part is filed with the Commission and (B) the fifth business day after the date
the Company is notified by the Commission that the registration statement will
not be reviewed or will not be subject to further review, which we refer to as
the Effectiveness Deadline. The effectiveness of the registration statement of
which this prospectus is a part is a condition precedent to our ability to sell
common stock to Magna under the Purchase Agreement.
12
We have agreed to file with the Commission one or more additional
registration statements to cover all of the securities required to be registered
under the Registration Rights Agreement that are not covered by this prospectus,
in each case, as soon as practicable, but in no event later than the applicable
filing deadline for such additional registration statements as provided in the
Registration Rights Agreement.
We also agreed, among other things, to indemnify Magna from certain
liabilities and fees and expenses of Magna incident to our obligations under the
Registration Rights Agreement, including certain liabilities under the
Securities Act. Magna has agreed to indemnify and hold harmless us and each of
our directors, officers and persons who control us against certain liabilities
that may be based upon written information furnished by Magna to us for
inclusion in the registration statement of which this prospectus is a part,
including certain liabilities under the Securities Act.
Transfer
Agent
The Transfer Agent for our Common Stock is Continental Stock Transfer and
Trust Company, 17 Battery Place, New York, New York 10004.
The Offering
As of November 20, 2014, there were 560,564,622 shares of our common
stock outstanding, of which 554,375,697 shares were held by non-affiliates.
Although the Purchase Agreement provides that we may sell up to $3,000,000 of
our common stock to Magna, only 143,812,591 shares of our common stock are being
offered under this prospectus, which represents (i) 31,000,000 shares of common
stock that may be issued to Magna upon conversion of the Convertible Note, (ii)
9,109,128 shares of common stock that we issued to Magna as Initial Commitment
Shares on October 27, 2014, (iii) a maximum of 15,890,872 shares of common stock
that we may be required to issue to Magna as Additional Commitment Shares and
(iv) 87,812,591 shares of common stock that we may issue to Magna as Shares
pursuant to draw downs under the Purchase Agreement. If all of the 143,812,591
shares offered under this prospectus were issued and outstanding as of November
20, 2014, such shares would represent approximately 20.7% of the total number of
shares of our common stock outstanding and 20.9% of the total number of
outstanding shares of our common stock held by non-affiliates, in each case as
of November 20, 2014.
At an assumed purchase price of $0.01460 (equal to 93% of the closing
price of our common stock of $0.01570 on November 10, 2014), and assuming the
sale by us to Magna of all of the 87,812,591 Shares, or approximately 15.7% of
our issued and outstanding common stock, being registered hereunder pursuant to
draw downs under the Purchase Agreement, we would receive only approximately
$1,282,064 in gross proceeds. Furthermore, we may receive substantially less
than $1,282,064 in gross proceeds from the financing due to our share price,
discount to market and other factors relating to our common stock. If we elect
to issue and sell more than the 87,812,591 Shares offered under this prospectus
to Magna, which we have the right, but not the obligation, to do, we must first
register for resale under the Securities Act any such additional Shares, which
could cause additional substantial dilution to our stockholders. Based on the
above assumptions, we would be required to register an additional approximately
117,666,849 shares of our common stock to obtain the balance of $1,717,936 of
the Total Commitment that would be available to us under the Purchase Agreement.
We currently have authorized and available for issuance 2,000,000,000 shares of
our common stock pursuant to our charter. The number of shares of our common
stock ultimately offered for resale by Magna is dependent upon a number of
factors, including the extent to which Magna converts the Convertible Note into
shares of our common stock and the number of Shares we ultimately issue and sell
to Magna under the Purchase Agreement.
The Total Commitment of $3,000,000 was determined based on numerous
factors, including our estimated operating expenses for the next two years.
While it is difficult to estimate the likelihood that we will need the full
Total Commitment, we presently believe that we may need the full Total
Commitment under the Purchase Agreement.
13
Common stock offered by
Selling Stockholder
143,812,591 shares of
common stock, consisting of:
- 31,000,000 shares of common
stock that we may issue to Magna upon conversion of the Convertible Note;
- 9,109,128 shares of common stock that
we issued to Magna as Initial Commitment Shares on October 27, 2014;
- a maximum of 15,890,872 shares of
common stock that we may be required to issue to Magna as Additional
Commitment Shares; and
- 87,812,591 shares of common stock that
we may issue to Magna as Shares pursuant to draw downs under the Purchase
Agreement.
Common stock outstanding
before the offering
560,564,622 shares of
common stock (includes 9,109,128 shares issued to Magna Equities II LLC on
October 27, 2014).
Common stock outstanding
after the offering
695,268,085 shares of
common stock.
We will not receive any
proceeds from the sale of shares by the selling stockholder. However, we have
received gross proceeds of $205,000 from the sale of the Convertible Note to
Magna and we may receive gross proceeds of up to $3,000,000 from the sale of
Shares to Magna pursuant to the Purchase Agreement. The net proceeds received
from the sale of the Convertible Note to Magna and from the sale of Shares
pursuant to the Purchase Agreement will be used for general corporate and
working capital purposes and acquisitions or assets, businesses or operations or
for other purposes that our Board of Directors, in its good faith deem to be in
the best interest of the company and its stockholders.
14
The common stock offered hereby
involves a high degree of risk and should not be purchased by investors who
cannot afford the loss of their entire investment. See “Risk Factors”.
15
RISK FACTORS
You should carefully
consider the risks described below as well as other information provided to you
in this document, including information in the section of this document entitled
“Information Regarding Forward Looking Statements.” If any of the following
risks actually cause the occurrence of adverse circumstances, the Company’s
business, financial condition or results of operations could be materially
adversely affected, the value of the Company’s Common Stock could decline and
you may lose all or part of your investment.
Risks Related to Our
Business
We are a development
stage life sciences company with a limited operating history and a history of
net losses and negative cash flows from operations. We may never be profitable,
and if we incur operating losses and generate negative cash flows from
operations for longer than expected, we may be unable to continue operations.
Risks Related to Our
Financial Position and Need for Additional Financing
We will need to
secure additional financing in order to continue to finance our operations. If
we are unable to secure additional financing on acceptable terms, or at all, we
may be forced to curtail or cease our operations.
As of September 30, 2014, we had cash and cash equivalents of
approximately $46,592 and an accumulated capital deficit of approximately
$119,428,183. As such, our existing cash resources are insufficient to finance
even our immediate operations. Accordingly, we will need to secure additional
sources of capital to develop our business and product candidates as planned. We
are seeking substantial additional financing through public and/or private
financing, which may include equity and/or debt financings, research grants and
through other arrangements, including collaborative arrangements. As part of
such efforts, we may seek loans from certain of our executive officers,
directors and/or current shareholders. We may also seek to satisfy some of our
obligations to the guarantors of our loan with Seaside National Bank &
Trust, or the Guarantors, through the issuance of various forms of securities or
debt on negotiated terms. However, financing and/or alternative arrangements
with the Guarantors may not be available when we need it, or may not be
available on acceptable terms.
If we are unable to secure additional financing in the near term, we may
be forced to:
- curtail or abandon our existing
business plans;
- reduce our headcount;
- default on our debt
obligations;
- file for bankruptcy;
- seek to sell some or all of our
assets; and/or
- cease our operations.
If we are forced to take any of these steps, any investment in our common
stock may be worthless.
Our ability to obtain
additional debt financing and/or alternative arrangements, with the Guarantors
or otherwise, may be limited by the amount of, terms and restrictions of our
then current debt. For instance, we do not anticipate repaying our Northstar
loan (described below) until its scheduled maturity. Accordingly, until such
time, we will generally be restricted from, among other things, incurring
additional indebtedness or liens, with limited exceptions. See “We have a substantial amount of
debt...” Additional debt
financing, if available, may involve restrictive covenants that limit or further
limit our operating and financial flexibility and prohibit us from making
distributions to shareholders.
If we raise additional capital and/or secure alternative arrangements,
with the Guarantors or otherwise, by issuing equity, equity-related or
convertible securities, the economic, voting and other rights of our existing
shareholders may be diluted, and those newly issued securities may be issued at
prices that are a significant discount to current and/or then prevailing market
prices. In addition, any such newly issued securities may have rights superior
to those of our common stock. If we obtain additional capital through
collaborative arrangements, we may be required to relinquish greater rights to
our technologies or product candidates than we might otherwise have or become
subject to restrictive covenants that may affect our business.
16
Our independent
registered public accounting firm has expressed substantial doubt about our
ability to continue as a going concern.
Our independent registered public accounting firm issued its report dated
March 24, 2014 in connection with the audit of our financial statements as of
December 31, 2013, which included an explanatory paragraph describing the
existence of conditions that raise substantial doubt about our ability to
continue as a going concern. In addition, our notes to our unaudited financial
statement for the period ended September 30, 2014 included an explanatory
paragraph describing the existence of conditions that raise substantial doubt
about our ability to continue as a going concern. If we are not able to continue
as a going concern, it is likely that holders of our common stock will lose all
of their investment. Our financial statements do not include any adjustments
that might result from the outcome of this uncertainty.
Current Adverse
Economic Conditions have had a negative impact on our ability to obtain
additional financing. Our inability to obtain additional financing would have a
significant adverse effect on our operations.
In early 2008, as the United States economy began to weaken and there
were increased doubts about the ability of borrowers to pay debts. Housing
values began to fall and marginal loans were first to default, triggering the
sub-prime lending crisis. Financial institutions responded by tightening their
lending policies with respect to counterparties determined to have sub-prime
mortgage risk. This tightening of institutional lending policies led to the
failure of major financial institutions late in the third quarter of 2008.
Continued failures, losses, and write-downs at major financial institutions
through 2013 intensified concerns about credit and liquidity risks and have
resulted in a sharp reduction in overall market liquidity. The global credit
crisis threatens the stability of the global economy and has adversely impacted
consumer confidence and spending. We believe this global credit crisis has also
had a negative impact on our ability to obtain additional financing. As
discussed above, our inability to obtain additional financing would have a
significant adverse effect on our operations, results and financial condition.
We are a development
stage life sciences company with a limited operating history and a history of
net losses and negative cash flows from operations. We may never be profitable,
and if we incur operating losses and generate negative cash flows from
operations for longer than expected, we may be unable to continue
operations.
We are a development stage life sciences company and have a limited
operating history, limited capital, limited sources of revenue and have incurred
losses since inception. Our operations to date have been limited to organizing
our company, developing and engaging in clinical trials of our MyoCell product
candidate, expanding our pipeline of complementary product candidates through
internal development and third party licenses, expanding and strengthening our
intellectual property position through internal programs and third party
licenses and recruiting management, research and clinical personnel.
Consequently, it may be difficult to predict our future success or viability due
to our lack of operating history. Since inception, we have generated substantial
net losses, including net losses of approximately $3.1 million, $4.0 million,
$4.7 million, $5.2 million, and $4.4 million in 2013, 2012, 2011, 2010 and 2009,
respectively, net loss of approximately 2.4 million for the quarter ended
September 30, 2014 and substantial negative cash flows from operations. We
anticipate that we will continue to incur significant and increasing net losses
and negative cash flows from operations for the foreseeable future as we:
- establish a distribution network for
and commence distribution of certain products for which we have acquired
distribution rights;
- resume full scale enrollment of the
MARVEL and REGEN Trials;
- continue research and development and
undertake new clinical trials with respect to our pipeline product candidates,
including clinical trials related to MyoCell SDF-1;
- seek to raise additional
capital;
- apply for regulatory approvals;
17
- make capital expenditures to increase
our research and development and cell culturing capabilities;
- add operational, financial and
management information systems and personnel and develop and protect our
intellectual property;
- make payments pursuant to license
agreements upon achievement of certain milestones; and
- establish sales and marketing
capabilities to commercialize products for which we obtain regulatory
approval, if any.
Our limited experience in conducting and managing preclinical development
activities, clinical trials and the application process necessary to obtain
regulatory approvals might prevent us from successfully designing or
implementing a preclinical study or clinical trial. If we do not succeed in
conducting and managing our preclinical development activities or clinical
trials, or in obtaining regulatory approvals, we might not be able to
commercialize our product candidates, or might be significantly delayed in doing
so, which will materially harm our business.
Our ability to generate revenues from any of our product candidates will
depend on a number of factors, including our ability to successfully complete
clinical trials, obtain necessary regulatory approvals and implement our
commercialization strategy. In addition, even if we are successful in obtaining
necessary regulatory approvals and bringing one or more product candidates to
market, we will be subject to the risk that the marketplace will not accept
those products. We may, and anticipate that we will need to, transition from a
company with a research and development focus to a company capable of supporting
commercial activities and we may not succeed in such a transition.
Because of the numerous risks and uncertainties associated with our
product development and commercialization efforts, we are unable to predict the
extent of our future losses or when or if we will become profitable. Our failure
to successfully commercialize our product candidates or to become and remain
profitable could impair our ability to raise capital, expand our business,
diversify our product offerings and continue our operations.
All of our product
candidates are in an early stage of development and we may never succeed in
developing and/or commercializing them. We depend heavily on the success of our
MyoCell product candidate. If we are unable to commercialize MyoCell or any of
our other product candidates or experience significant delays in doing so, our
business may fail.
- We have invested a significant portion
of our efforts and financial resources in our MyoCell product candidate and
depend heavily on its success. MyoCell is currently in the clinical testing
stage of development, although we have suspended work under our clinical
trials as we seek to raise sufficient funds to complete the trials.
- We need to devote significant
additional research and development, financial resources and personnel to
develop commercially viable products, obtain regulatory approvals and
establish a sales and marketing infrastructure.
- We are likely to encounter hurdles and
unexpected issues as we proceed in the development of MyoCell and our other
product candidates. There are many reasons that we may not succeed in our
efforts to develop our product candidates, including the possibility that:
- our product candidates will be deemed
ineffective, unsafe or will not receive regulatory approvals;
- our product candidates will be too
expensive to manufacture or market or will not achieve broad market
acceptance;
- others will hold proprietary rights
that will prevent us from marketing our product candidates; or
- our competitors will market products
that are perceived as equivalent or superior.
18
Our approach of using
cell-based therapy for the treatment of heart damage is risky and unproven and
no products using this approach have received regulatory approval in the United
States or Europe.
No company has yet been successful in its efforts to obtain regulatory
approval in the United States or Europe of a cell-based therapy product for the
treatment of heart damage. Cell-based therapy products, in general, may be
susceptible to various risks, including undesirable and unintended side effects,
unintended immune system responses, inadequate therapeutic efficacy or other
characteristics that may prevent or limit their approval by regulators or
commercial use. Many companies in the industry have suffered significant
setbacks in advanced clinical trials, despite promising results in earlier
trials. One of our competitors exploring the use of skeletal myoblasts ceased
enrolling new patients in its European Phase II clinical trial based on the
determination of its monitoring committee that there was a low likelihood that
the trial would result in the hypothesized improvement in heart function.
Although our clinical research to date suggests that MyoCell may improve the
contractile function of the heart, we have not yet been able to demonstrate a
mechanism of action and additional research is needed to precisely identify such
mechanism.
Four clinical trials are unsuccessful or
significantly delayed, or if we do not complete our clinical trials, we will not
receive regulatory approval for or be able to commercialize our product
candidates.
We cannot market any product candidate until regulatory agencies grant
approval or licensure. In order to obtain regulatory approval for the sale of
any product candidate, we must, among other requirements, provide the FDA and
similar foreign regulatory authorities with preclinical and clinical data that
demonstrate to the satisfaction of regulatory authorities that our product
candidates are safe and effective for each indication under the applicable
standards relating to such product candidate. The preclinical studies and
clinical trials of any product candidates must comply with the regulations of
the FDA and other governmental authorities in the United States and similar
agencies in other countries.
Even if we achieve positive interim results in clinical trials, these
results do not necessarily predict final results, and positive results in early
trials may not be indicative of success in later trials. For example, MyoCell
has been studied in a limited number of patients to date. Even though our early
data has been promising, we have not yet completed any large-scale pivotal
trials to establish the safety and efficacy of MyoCell. A number of participants
in our clinical trials have experienced serious adverse events adjudicated or
determined by trial investigators to be potentially attributable to MyoCell. See
“ – Our product candidates may
never be commercialized due to unacceptable side effects and increased mortality
that may be associated with such product candidates.” There is a risk that safety concerns relating
to our product candidates or cell-based therapies in general will result in the
suspension or termination of our clinical trials.
We may experience numerous unforeseen events during, or as a result of,
the clinical trial process that could delay or prevent regulatory approval
and/or commercialization of our product candidates, including the following:
- the FDA or similar foreign regulatory
authorities may find that our product candidates are not sufficiently safe or
effective or may find our cell culturing processes or facilities
unsatisfactory;
- officials at the FDA or similar
foreign regulatory authorities may interpret data from preclinical studies and
clinical trials differently than we do;
- our clinical trials may produce
negative or inconclusive results or may not meet the level of statistical
significance required by the FDA or other regulatory authorities, and we may
decide, or regulators may require us, to conduct additional preclinical
studies and/or clinical trials or to abandon one or more of our development
programs;
- the FDA or similar foreign regulatory
authorities may change their approval policies or adopt new regulations;
- there may be delays or failure in
obtaining approval of our clinical trial protocols from the FDA or other
regulatory authorities or obtaining institutional review board approvals or
government approvals to conduct clinical trials at prospective sites
19
- we, or regulators, may suspend or
terminate our clinical trials because the participating patients are being
exposed to unacceptable health risks or undesirable side effects;
- we may experience difficulties in
managing multiple clinical sites;
- enrollment in our clinical trials for
our product candidates may occur more slowly than we anticipate, or we may
experience high drop-out rates of subjects in our clinical trials, resulting
in significant delays;
- we may be unable to manufacture or
obtain from third party manufacturers sufficient quantities of our product
candidates for use in clinical trials; and
- our product candidates may be deemed
unsafe or ineffective, or may be perceived as being unsafe or ineffective, by
healthcare providers for a particular indication.
In the SEISMIC Trial, we experienced delays attributable to slower than
anticipated enrollment of patients. We may continue to experience difficulties
in enrolling patients in our clinical trials, which could increase the costs or
affect the timing or outcome of these trials and could prevent us from
completing these trials.
Failures or perceived failures in our clinical trials would delay and may
prevent our product development and regulatory approval process, make it
difficult for us to establish collaborations, negatively affect our reputation
and competitive position and otherwise have a material adverse effect on our
business.
Our product
candidates may never be commercialized due to unacceptable side effects and
increased mortality that may be associated with such product
candidates.
Possible side effects of our product candidates may be serious and
life-threatening. A number of participants in our clinical trials of MyoCell
have experienced serious adverse events potentially attributable to MyoCell,
including six patient deaths and 18 patients experiencing irregular heartbeats.
A serious adverse event is generally an event that results in significant
medical consequences, such as hospitalization, disability or death, and must be
reported to the FDA. The occurrence of any unacceptable serious adverse events
during or after preclinical and clinical testing of our product candidates could
temporarily delay or negate the possibility of regulatory approval of our
product candidates and adversely affect our business. Both our trials and
independent trials have reported the occurrence of irregular heartbeats in
treated patients, a significant risk to patient safety. We and our competitors
have also, at times, suspended trials studying the effects of myoblasts, at
least temporarily, to assess the risk of irregular heartbeats, and it has been
reported that one of our competitors studying the effect of myoblast
implantation prematurely discontinued a study because of the high incidence of
irregular heartbeats. While we believe irregular heartbeats may be manageable
with the use of certain prophylactic measures including an ICD, and
antiarrhythmic drug therapy, these risk management techniques may not prove to
sufficiently reduce the risk of unacceptable side effects.
Although our early results suggest that patients treated with MyoCell do
not face materially different health risks than heart failure patients with
similar levels of damage to the heart who have not been treated with MyoCell, we
are still in the process of seeking to demonstrate that our product candidates
do not pose unacceptable health risks. We have not yet treated a sufficient
number of patients to allow us to make a determination that serious unintended
consequences will not occur.
20
We depend on third
parties to assist us in the conduct of our preclinical studies and clinical
trials, and any failure of those parties to fulfill their obligations could
result in costs and delays and prevent us from obtaining regulatory approval or
successfully commercializing our product candidates on a timely basis, if at
all.
We engage consultants and CROs to help design, and to assist us in
conducting, our preclinical studies and clinical trials and to collect and
analyze data from those studies and trials. The consultants and contract
research organizations we engage interact with clinical investigators to enroll
patients in our clinical trials. As a result, we depend on these consultants and
CROs to perform the studies and trials in accordance with the investigational
plan and protocol for each product candidate and in compliance with regulations
and standards, commonly referred to as “good clinical practice”, for conducting,
recording and reporting results of clinical trials to assure that the data and results are credible and
accurate and the trial participants are adequately protected, as required by the
FDA and foreign regulatory agencies. We may face delays in our regulatory
approval process if these parties do not perform their obligations in a timely
or competent fashion or if we are forced to change service providers. The risk
of delays is heightened for our clinical trials conducted outside of the United
States, where it may be more difficult for us to ensure that studies are
conducted in compliance with foreign regulatory requirements. Any third parties
that we hire to conduct clinical trials may also provide services to our
competitors, which could compromise the performance of their obligations to us.
If these third parties do not successfully carry out their duties or meet
expected deadlines, or if the quality, completeness or accuracy of the data they
obtain is compromised due to their failure to adhere to our clinical trial
protocols or for other reasons, our clinical trials may be extended, delayed or
terminated or may otherwise prove to be unsuccessful. If there are delays or
failures in clinical trials or regulatory approvals as a result of the failure
to perform by third parties, our development costs will increase, and we may not
be able to obtain regulatory approval for our product candidates. In addition,
we may not be able to establish or maintain relationships with these third
parties on favorable terms, if at all. If we need to enter into replacement
arrangements because a third party is not performing in accordance with our
expectations, we may not be able to do so without undue delays or considerable
expenditures or at all.
Risks Related to
Government Regulation and Regulatory Approvals
Our cell-based
product candidates are based on novel technologies and the FDA and regulatory
agencies in other countries have limited experience reviewing product candidates
using these technologies.
We are subject to the risks
of failure inherent in the development of product candidates based on new
technologies. The novel nature of our product candidates creates significant
challenges in regards to product development and optimization, government
regulation, third party reimbursement and market acceptance. These include:
- the scientific basis of our technology
could be determined to be less sound than we believe;
- the time and effort required to solve
novel technical problems could delay the development of our product
candidates;
- the FDA and regulatory agencies in
other countries have relatively limited experience with therapies based upon
cellular medicine generally and, as a result, the pathway to regulatory
approval for our cell-based product candidates may be more complex and
lengthy; and
- the healthcare community has
relatively little experience with therapies based upon cellular medicine and,
accordingly, following regulatory approval, if any, our product candidates may
not become widely accepted by physicians, patients, third party payers or the
healthcare community.
As a result, the
development and commercialization pathway for our cell-based therapies may be
subject to increased uncertainty, as compared to the pathway for new
conventional drugs.
We are subject to
numerous risks associated with seeking regulatory approval of MyoCell pursuant
to a protocol that permits the use of a catheter system which is still subject
to FDA approval. The catheter system we intend to use in connection with our
MARVEL Trial is owned by an unaffiliated third party. Although we have entered
into a two-year supply agreement for delivery of the catheter system for use in
the MARVEL Trial, we are subject to a number of risks not addressed by the
parties in the supply agreement.
We have been cleared by the
FDA to proceed under the protocol for our MARVEL Trial, which protocol permits
participating trial investigators to use either our MyoCath catheters Biosense
Webster’s (a Johnson & Johnson company) NOGA® Cardiac Navigation System
along with the MyoStar™ injection catheter for the delivery of MyoCell to
patients enrolled in the trial. We anticipate that if MyoCell receives
regulatory approval, such approval will require MyoCell to be injected with a
catheter system that has also secured regulatory approval. Accordingly, the
commercial deployment of MyoCell is dependent upon MyoStar, MyoCath or some
other catheter system securing regulatory approval for use with MyoCell.
Although MyoStar has received CE mark approval in Europe, neither MyoStar,
MyoCath nor any other catheter system is commercially available in the United
States and they may only be used
pursuant to FDA approved investigational protocols. Notwithstanding the devotion
of considerable resources to the development and testing of MyoStar and MyoCath,
they may never receive additional or any, respectively, regulatory approval that
will allow for their commercial use with MyoCell.
21
We are not affiliated with
Biosense Webster, the Cordis Corporation or any other Johnson & Johnson
company. Although we entered into a supply agreement with Biosense Webster on
May 10, 2007 pursuant to which it has agreed to deliver MyoStar to us for a two
year period at an agreed upon price as and when required by the MARVEL Trial, we
currently have no right to control the further development, clinical testing
and/or refinement of MyoStar. Biosense Webster currently has the right to make
the following types of decisions without consulting with or even considering our
views, which decisions could directly or indirectly negatively impact our
efforts and/or ability to secure regulatory approval of MyoCell:
- the terms and conditions under which
MyoStar will be made available for use to trial investigators, if at all,
after the term of the supply agreement;
- the terms and conditions under which
the diagnostic consoles that are part of the NOGA® Cardiac Navigation System
will be made available for use to trial investigators, if at all;
- the modification or not of the MyoStar
System or any of its components and its protocol for use as a result of
information obtained during trials;
- the license or sale of the MyoStar
System related intellectual property to a third party, potentially including
our competitors;
- the use of the MyoStar System or any
of its components in myoblast-based clinical therapies other than ours; and
the suspension or abandonment of other clinical trials involving MyoStar.
The unavailability of the
MyoStar System, for any reason, would have a material adverse effect on our
product development and commercialization efforts as we will be unable to
recover the time and money expended on the MARVEL Trial prior to such
determination of unavailability.
We must comply with
extensive government regulations in order to obtain and maintain marketing
approval for our products in the United States and abroad. If we do not obtain
regulatory approval for our product candidates, we may be forced to cease our
operations.
Our product candidates are
subject to extensive regulation in the United States and in every other country
where they will be tested or used. These regulations are wide-ranging and
govern, among other things:
- product design, development, manufacture and testing;
- product safety and efficacy;
- product labeling;
- product storage and shipping;
- record keeping;
- pre-market clearance or
approval;
- advertising and promotion; and
- product sales and distribution.
We cannot market our
product candidates until we receive regulatory approval. The process of
obtaining regulatory approval is lengthy, expensive and uncertain. Any
difficulties that we encounter in obtaining regulatory approval may have a
substantial adverse impact on our business and cause our stock price to decline
significantly.
In the United States, the
FDA imposes substantial requirements on the introduction of biological products
and many medical devices through lengthy and detailed laboratory and clinical
testing procedures, sampling activities and other costly and time-consuming
procedures. Satisfaction of these requirements typically takes several years and
the time required to do so may vary substantially based upon the type and
complexity of the biological product or medical device.
22
In addition, product
candidates that we believe should be classified as medical devices for purposes
of the FDA regulatory pathway may be determined by the FDA to be biologic
products subject to the satisfaction of significantly more stringent
requirements for FDA approval.
The requirements governing
the conduct of clinical trials and cell culturing and marketing of our product
candidates outside the United States vary widely from country to country.
Foreign approvals may take longer to obtain than FDA approvals and can require,
among other things, additional testing and different clinical trial designs.
Foreign regulatory approval processes generally include all of the risks
associated with the FDA approval processes. Some foreign regulatory agencies
also must approve prices of the products. Regulatory approval in one country
does not ensure regulatory approval in another, but a failure or delay in
obtaining regulatory approval in one country may negatively impact the
regulatory process in others. We may not be able to file for regulatory
approvals and may not receive necessary approvals to market our product
candidates in any foreign country. If we fail to comply with these regulatory
requirements or fail to obtain and maintain required approvals in any foreign
country, we will not be able to sell our product candidates in that country and
our ability to generate revenue will be adversely affected.
We cannot assure you that
we will obtain FDA or foreign regulatory approval to market any of our product
candidates for any indication in a timely manner or at all. If we fail to obtain
regulatory approval of any of our product candidates for at least one
indication, we will not be permitted to market our product candidates and may be
forced to cease our operations.
Even if some of our
product candidates receive regulatory approval, these approvals may be subject
to conditions, and we and our third party manufacturers will in any event be
subject to significant ongoing regulatory obligations and
oversight.
Even if any of our product
candidates receives regulatory approval, the manufacturing, marketing and sale
of our product candidates will be subject to stringent and ongoing government
regulation. Conditions of approval, such as limiting the category of patients
who can use the product, may significantly impact our ability to commercialize
the product and may make it difficult or impossible for us to market a product
profitably. Changes we may desire to make to an approved product, such as cell
culturing changes or revised labeling may require further regulatory review and
approval, which could prevent us from updating or otherwise changing an approved
product. If our product candidates are approved by the FDA or other regulatory
authorities for the treatment of any indications, regulatory labeling may
specify that our product candidates be used in conjunction with other therapies.
For instance, we currently anticipate that prior implantation of an ICD and
treatment with optimal drug therapy will be required at least initially as a
condition to treatment with MyoCell.
Once obtained, regulatory
approvals may be withdrawn for a number of reasons, including the later
discovery of previously unknown problems with the product. Regulatory approval
may also require costly post-marketing follow-up studies, and failure of our
product candidates to demonstrate sufficient efficacy and safety in these
studies may result in either withdrawal of marketing approval or severe
limitations on permitted product usage. In addition, numerous additional
regulatory requirements relating to, among other processes, the labeling,
packaging, adverse event reporting, storage, advertising, promotion and
record-keeping will also apply. Furthermore, regulatory agencies subject a
marketed product, its manufacturer and the manufacturer’s facilities to
continual review and periodic inspections. Compliance with these regulatory
requirements is time consuming and requires the expenditure of substantial
resources.
If any of our product
candidates is approved, we will be required to report certain adverse events
involving our products to the FDA, to provide updated safety and efficacy
information and to comply with requirements concerning the advertisement and
promotional labeling of our products. As a result, even if we obtain necessary
regulatory approvals to market our product candidates for any indication, any
adverse results, circumstances or events that are subsequently discovered could
require that we cease marketing the product for that indication or expend money,
time and effort to ensure full compliance, which could have a material adverse
effect on our business.
23
In response to recent
events regarding questions about the safety of certain approved prescription
products, including the lack of adequate warnings, the FDA and the U.S. Congress
are currently considering new regulatory and legislative approaches to
advertising, monitoring and assessing the safety of marketed drugs, including
legislation authorizing the FDA to mandate labeling changes for approved
products, particularly those related to safety. It is possible that
congressional and FDA initiatives pertaining to ensuring the safety of marketed
biologics and similar initiatives in other countries, or other developments
pertaining to the pharmaceutical industry, could require us to expend additional
resources to comply with such initiatives and could adversely affect our
operations.
In addition, the FDA and
similar foreign governmental authorities have the authority to require the
recall of commercialized products in the event of any failure to comply with
applicable laws and regulations or defects in design or manufacture. In the
event any of our product candidates receives approval and is commercialized, a
government-mandated or voluntary product recall by us could occur as a result of
component failures, device malfunctions, or other negative events such as
serious injuries or deaths, or quality-related issues such as cell culturing
errors or design or labeling defects. Recalls of any of our potential products
could divert managerial and financial resources, harm our reputation and
adversely affect our financial condition, results of operations and stock price.
Any failure by us, or by
any third parties that may manufacture or market our products, to comply with
the law, including statutes and regulations administered by the FDA or other
U.S. or foreign regulatory authorities, could result in, among other things,
warning letters, fines and other civil penalties, suspension of regulatory
approvals and the resulting requirement that we suspend sales of our products,
refusal to approve pending applications or supplements to approved applications,
export or import restrictions, interruption of production, operating
restrictions, closure of the facilities used by us or third parties to
manufacture our product candidates, injunctions or criminal prosecution. Any of
the foregoing actions could have a material adverse effect on our business.
We must comply with
federal, state and foreign laws, regulations and other rules relating to the
healthcare business, and, if we do not fully comply with such laws, regulations
and other rules, we could face substantial penalties.
We are, or will be directly
or indirectly through our customers, subject to extensive regulation by the
federal government, the states and foreign countries in which we may conduct our
business. The laws that directly or indirectly affect our ability to operate our
business include the following:
- the federal Medicare and Medicaid
Anti-Kickback law, which prohibits persons from knowingly and willfully
soliciting, offering, receiving or providing remuneration, directly or
indirectly, in cash or in kind, to induce either the referral of an individual
or furnishing or arranging for a good or service, for which payment may be
made under federal healthcare programs such as Medicare and Medicaid;
- other Medicare laws, regulations,
rules, manual provisions and policies that prescribe the requirements for
coverage and payment for services performed by our customers, including the
amount of such payment;
- the Federal False Claims Act, which
imposes civil and criminal liability on individuals and entities who submit,
or cause to be submitted, false or fraudulent claims for payment to the
government;
- the Federal False Statements Act,
which prohibits knowingly and willfully falsifying, concealing or covering up
a material fact or making any materially false statement in connection with
delivery of or payment for healthcare benefits, items or services; and
- state and foreign law equivalents of
the foregoing.
If our operations are found
to be in violation of any of the laws, regulations, rules or policies described
above or any other law or governmental regulation to which we or our customers
are or will be subject, or if the interpretation of the foregoing changes, we
may be subject to civil and criminal penalties, damages, fines, exclusion from
the Medicare and Medicaid programs and the curtailment or restructuring of our
operations. Similarly, if our customers are found non-compliant with applicable
laws, they may be subject to sanctions, which could also have a negative impact
on us. Any penalties, damages, fines, curtailment or restructuring of our
operations would adversely affect our ability to
operate our business and our financial results. The risk of our being found in
violation of these laws is increased by the fact that many of them have not been
fully interpreted by the regulatory authorities or the courts, and their
provisions are open to a variety of interpretations, and additional legal or
regulatory change.
24
Any action against us for
violation of these laws, even if we successfully defend against it, could cause
us to incur significant legal expenses, divert our management’s attention from
the operation of our business and damage our reputation.
Our business involves
the use of hazardous materials that could expose us to environmental and other
liability.
Our facility in Sunrise,
Florida is subject to various local, state and federal laws and regulations
relating to the use and disposal of hazardous or potentially hazardous
substances, including chemicals and microorganisms used in connection with our
research and development activities. In the United States, these laws include
the Occupational Safety and Health Act, the Toxic Test Substances Control Act
and the Resource Conservation and Recovery Act. Although we believe that our
safety procedures for handling and disposing of these materials comply with the
standards prescribed by these regulations, we cannot assure you that accidental
contamination or injury to employees and third parties from these materials will
not occur.
Risks Related to
Commercialization of our Product Candidates
The healthcare
community has relatively little experience with therapies based on cellular
medicine and, accordingly, if our product candidates do not become widely
accepted by physicians, patients, third party payers or the healthcare
community, we may be unable to generate significant revenue, if
any.
We are developing
cell-based therapy product candidates for the treatment of heart damage that
represent novel and unproven treatments and, if approved, will compete with a
number of more conventional products and therapies manufactured and marketed by
others, including major pharmaceutical companies. We cannot predict or guarantee
that physicians, patients, healthcare insurers, third party payers or health
maintenance organizations, or the healthcare community in general, will accept
or utilize any of our product candidates. We anticipate that, if approved, we
will market MyoCell primarily to interventional cardiologists, who are generally
not the primary care physicians for patients who may be eligible for treatment
with MyoCell. Accordingly, our commercial success may be dependent on third
party physicians referring their patients to interventional cardiologists for
MyoCell treatment.
If we are successful in
obtaining regulatory approval for any of our product candidates, the degree of
market acceptance of those products will depend on many factors, including:
- our ability to provide acceptable
evidence and the perception of patients and the healthcare community,
including third party payers, of the positive characteristics of our product
candidates relative to existing treatment methods, including their safety,
efficacy, cost effectiveness and/or other potential advantages;
- the incidence and severity of any
adverse side effects of our product candidates;
- the availability of alternative
treatments;
- the labeling requirements imposed by
the FDA and foreign regulatory agencies, including the scope of approved
indications and any safety warnings;
- our ability to obtain sufficient third
party insurance coverage or reimbursement for our products candidates;
- the inclusion of our products on
insurance company coverage policies;
- the willingness and ability of
patients and the healthcare community to adopt new technologies;
- the procedure time associated with the
use of our product candidates;
- our ability to manufacture or obtain
from third party manufacturers sufficient quantities of our product candidates
with acceptable quality and at an acceptable cost to meet demand; and
- marketing and distribution support
for our products.
Failure to achieve market
acceptance would limit our ability to generate revenue and would have a material
adverse effect on our business. In addition, if any of our product candidates
achieve market acceptance, we may not be able to maintain that
market acceptance over time if competing products or technologies are introduced
that are received more favorably or are more cost-effective.
25
There is substantial
uncertainty as to the coverage that may be available and the reimbursement rates
that may be established for our product candidates. Any failure to obtain third
party coverage or an adequate level of reimbursement for our product candidates
will likely have a material adverse effect on our business.
If we successfully develop,
and obtain necessary regulatory approvals for, our product candidates we intend
to sell them initially in Europe and the United States. We have not yet
submitted any of our product candidates to CMS or any private or governmental
third party payer in the United States to determine whether or not our product
candidates will be covered under private or public health insurance plans or, if
they are covered, what coverage or reimbursement rates may be available.
Although we believe hospitals may be entitled to some procedure reimbursement
for MyoCell, we cannot assure you that such reimbursement will be adequate or
available at all.
In Europe, the pricing of
prescription pharmaceutical products and services and the level of government
reimbursement generally are subject to governmental control. Reimbursement and
healthcare payment systems in European markets vary significantly by country,
and may include both government-sponsored healthcare and private insurance. In
these countries, pricing negotiations with governmental authorities can take six
to twelve months or longer after the receipt of marketing approval for a
product. To obtain reimbursement or pricing approval in some countries, we may
be required to conduct one or more clinical trials that compare the cost
effectiveness of our product candidates to other available therapies. Conducting
one or more clinical trials for this purpose would be expensive and result in
delays in commercialization of our product candidates. We may not obtain
coverage or reimbursement or pricing approvals from countries in Europe in a
timely manner, or at all. Any failure to receive coverage or reimbursement or
pricing approvals from one or more European countries could effectively prevent
us from selling our product candidates in those countries, which could
materially adversely affect our business.
In the United States, our
revenues will depend upon the coverage and reimbursement rates and policies
established for our product candidates by third party payers, including
governmental authorities, managed-care providers, public health insurers,
private health insurers and other organizations. These third party payers are
increasingly attempting to contain healthcare costs by limiting both coverage
and the level of reimbursement for new healthcare products approved for
marketing by the FDA or regulatory agencies in other countries. As a result,
significant uncertainty exists as to whether newly approved medical products
will be eligible for coverage by third party payers or, if eligible for
coverage, what the reimbursement rates will be for those products. Furthermore,
cell-based therapies like MyoCell may be more expensive than pharmaceuticals due
to, among other things, the higher cost and complexity associated with the
research, development and production of these therapies. This, in turn, may make
it more difficult for us to obtain adequate reimbursement from third party
payers, particularly if we cannot demonstrate a favorable cost-benefit
relationship. Third party payers may also deny coverage or offer inadequate
levels of reimbursement for our potential products if they determine that the
product has not received appropriate clearances from the FDA or other government
regulators or is experimental, unnecessary or inappropriate. Accordingly, we
cannot assure you that adequate third party coverage or reimbursement will be
available for any of our product candidates to allow us to successfully
commercialize these product candidates.
Coverage and reimbursement
rates for our product candidates may be subject to increased restrictions both
in the United States and in other countries in the future. Coverage policies and
reimbursement rates are subject to change and we cannot guarantee that current
coverage policies and reimbursement rates will be applicable to our product
candidates in the future. U.S. federal, state and foreign agencies and
legislatures from time to time may seek to impose restrictions on coverage,
pricing, and reimbursement level of drugs, devices and healthcare services in
order to contain healthcare costs.
If we are unable to
establish sales and marketing capabilities or enter into agreements with third
parties to market and sell our product candidates, we may be unable to generate
product revenues.
26
We do not have a sales and
marketing force and related infrastructure and have limited experience in the
sales, marketing and distribution of our product candidates. To achieve
commercial success for any approved product, we must either develop a sales and
marketing force or outsource these functions to third parties. Currently, we
intend to internally develop a direct sales and marketing force in both Europe
and the United States as we approach commercial approval of our product
candidates. In other regions, we intend to seek to enter into exclusive or
non-exclusive distribution arrangements. The development of our own sales and
marketing force will result in us incurring significant costs before the time
that we may generate revenues. We may not be able to attract, hire, train and
retain qualified sales and marketing personnel to build a significant or
effective marketing and sales force for sales of our product candidates.
Product liability and
other claims against us may reduce demand for our products or result in
substantial damages. We anticipate that we will need to obtain and maintain
additional or increased insurance coverage, and we may not be able to obtain or
maintain such coverage on commercially reasonable terms, if at
all.
A product liability claim,
a clinical trial liability claim or other claim with respect to uninsured
liabilities or for amounts in excess of insured liabilities could have a
material adverse effect on our business. Our business exposes us to potential
liability risks that may arise from the clinical testing of our product
candidates in human clinical trials and the manufacture and sale of any approved
products. Any clinical trial liability or product liability claim or series of
claims or class actions brought against us, with or without merit, could result
in:
- liabilities that substantially exceed
our existing clinical trial liability insurance, or any clinical trial
liability or product liability insurance that we may obtain in the future,
which we would then be required to pay from other sources, if
available;
- an increase in the premiums we pay for
our clinical trial liability insurance and any clinical trial liability or
product liability insurance we may obtain in the future or the inability to
renew or obtain clinical trial liability or product liability insurance
coverage in the future on acceptable terms, or at all;
- withdrawal of clinical trial
volunteers or patients;
- damage to our reputation and the
reputation of our products, including loss of market share;
- regulatory investigations that could
require costly recalls or product modifications;
- litigation costs; and
- diversion of management’s attention
from managing our business.
We do not currently have
product liability insurance because none of our product candidates has yet been
approved for commercialization. While we plan to seek product liability
insurance coverage if any of our product candidates are sold commercially, we
cannot assure you that we will be able to obtain product liability insurance on
commercially acceptable terms, if at all, or that we will be able to maintain
such insurance at a reasonable cost or in sufficient amounts to protect against
potential losses.
Claims may be made by
consumers, healthcare providers, third party strategic collaborators or others
selling our products if one of our products or product candidates causes, or
appears to have caused, an injury. We may be subject to claims against us even
if an alleged injury is due to the actions of others. For example, we rely on
the expertise of physicians, nurses and other associated medical personnel to
perform the medical procedures and processes related to our product candidates.
If these medical personnel are not properly trained or are negligent in using
our product candidates, the therapeutic effect of our product candidates may be
diminished or the patient may suffer injury, which may subject us to liability.
In addition, an injury resulting from the activities of our suppliers may serve
as a basis for a claim against us.
We do not intend to
promote, or to in any way support or encourage the promotion of, our product
candidates for off-label or otherwise unapproved uses. However, if our product
candidates are approved by the FDA or similar foreign regulatory authorities, we
cannot prevent a physician from using them for any off-label applications. If
injury to a patient results from such an inappropriate use, we may become
involved in a product liability suit, which will likely be expensive to defend.
27
These liabilities could
prevent or interfere with our clinical efforts, product development efforts and
any subsequent product commercialization efforts, all of which could have a
material adverse effect on our business.
Our success will
depend in part on establishing and maintaining effective strategic partnerships,
collaborations and licensing agreements.
Our strategy for the
development, testing, culturing and commercialization of our product candidates
relies on establishing and maintaining numerous collaborations with various
corporate partners, consultants, scientists, researchers, licensors, licensees
and others. While we are continually in discussions with a number of companies,
universities, research institutions, consultants, scientists, researchers,
licensors, licensees and others to establish additional relationships and
collaborations, which are typically complex and time consuming to negotiate,
document and implement, we may not reach definitive agreements with any of them.
Even if we enter into these arrangements, we may not be able to maintain these
relationships or establish new ones in the future on acceptable terms.
Furthermore, any
collaboration that we enter into may not be successful. The success of our
collaboration arrangements, if any, will depend heavily on the efforts and
activities of our collaborators. Possible future collaborations have risks,
including the following:
- our collaboration agreements are
likely to be for fixed terms and subject to termination by our collaborators
in the event of a material breach or lack of scientific progress by us;
- our collaborators are likely to have
the first right to maintain or defend our intellectual property rights and,
although we would likely seek to secure the right to assume the maintenance
and defense of our intellectual property rights if our collaborators do not,
our ability to do so may be compromised by our collaborators' acts or
omissions;
- our collaborators may utilize our
intellectual property rights in such a way as to invite litigation that could
jeopardize or invalidate our intellectual property rights or expose us to
potential liability;
- our collaborators may underfund or not
commit sufficient resources to the testing, marketing, distribution or
development of our product candidates; and
- our collaborators may develop
alternative products either on their own or in collaboration with others, or
encounter conflicts of interest or changes in business strategy or other
business issues, which could adversely affect their willingness or ability to
fulfill their obligations to us.
These arrangements also may
require us to grant certain rights to third parties, including exclusive
marketing rights to one or more products, or may have other terms that are
burdensome to us, and may involve the issuance of our securities. If any of our
partners terminates its relationship with us or fails to perform its obligations
in a timely manner, the development or commercialization of our technology and
product candidates may be substantially delayed. Further, disputes may arise
with our collaborators about inventorship and corresponding rights in knowhow
and inventions resulting from the joint creation or use of intellectual property
by us and our collaborators.
Risks Related to Our
Intellectual Property
We hold limited
patent and other intellectual property rights, and our success will be dependent
in large part on safeguarding our existing intellectual property rights and
obtaining patent and other proprietary protection for our product
candidates.
We hold limited patent
rights in our product candidates. Our MyoCath product candidate is protected by
a patent, expiring in September 2017, in which we have an irrevocable
co-exclusive license. Our MyoCell product candidate is no longer protected by
patents, which means that competitors will be free to sell products that
incorporate the same or similar technologies that are used in MyoCell without
infringing our patent rights. As a result, MyoCell, if approved for use, may be
vulnerable to competition in the form of products that use the same or similar
technologies. We have previously licensed certain patents and patent
applications relating to our MyoCell product candidate. These licenses have all
lapsed as of the date of this report, although we have had discussions with the
relevant licensor regarding a potential reinstatement of our rights in such
licenses.
28
Our commercial success will
depend to a significant degree on our ability to:
- compel the owners of the patents
licensed to us to defend and enforce such patents, to the extent such patents
may be applicable to our products and material to their
commercialization;
- obtain new patent and other
proprietary protection for MyoCell and our other product candidates;
- obtain and/or maintain appropriate
licenses to patents, patent applications or other proprietary rights held by
others with respect to our technology, both in the United States and other
countries;
- preserve intellectual property rights
relating to our product candidates; and
- operate without infringing the patents
and proprietary rights of third parties.
Failure to obtain adequate
patent protection for our product candidates, or the failure to protect our
existing patent rights, may impair our ability to be competitive. The
availability of infringing products in markets where we have patent protection,
or the availability of competing products in markets where we do not have
adequate patent protection, could erode the market for our product candidates,
negatively impact the prices we can charge for our product candidates, and harm
our reputation if infringing or competing products are manufactured to inferior
standards.
Our most important
license agreement with respect to MyoCath is co-exclusive and the co-licensor of
the intellectual property, a division of Abbott Laboratories, may also seek to
commercialize MyoCath.
In June 2003, we assigned
our exclusive license to the primary patent protecting MyoCath to ACS,
originally a subsidiary of Guidant Corporation and now d/b/a Abbott Vascular, a
division of Abbott Laboratories. In connection with this agreement, ACS granted
to us a co-exclusive, irrevocable, fully paid-up license to this patent for the
life of the patent. Because our license is co-exclusive with ACS, ACS may,
parallel to our efforts, seek to commercialize MyoCath if MyoCath secures
regulatory approval.
Accordingly, even if ACS
does nothing to assist us to secure regulatory approval of MyoCath, ACS may
become a direct competitor in the MyoCath manufacturing and supply business. In
addition, pursuant to our agreement with ACS, we are prohibited from contracting
with third parties for the distribution of MyoCath.
Our patents may not
be valid or enforceable, and may be challenged by third
parties.
We cannot assure you that
any patents issued or licensed to us would be held valid by a court or
administrative body or that we would be able to successfully enforce our patents
against infringers, including our competitors. The issuance of a patent is not
conclusive as to its validity or enforceability, and the validity and
enforceability of a patent is susceptible to challenge on numerous legal
grounds. Challenges raised in patent infringement litigation brought by or
against us may result in determinations that patents that have been issued or
licensed to us or any patents that may be issued to us or our licensors in the
future are invalid, unenforceable or otherwise subject to limitations. In the
event of any such determinations, third parties may be able to use the
discoveries or technologies claimed in these patents without paying licensing
fees or royalties to us, which could significantly diminish the value of our
intellectual property and our competitive advantage. Even if our patents are
held to be enforceable, others may be able to design around our patents or
develop products similar to our products that are not within the scope of any of
our patents.
In addition, enforcing the
patents that have been licensed to us and any patents that may be issued to us
in the future against third parties may require significant expenditures
regardless of the outcome of such efforts. Our inability to enforce our patents
against infringers and competitors may impair our ability to be competitive and
could have a material adverse effect on our business.
If we are not able to
protect and control unpatented trade secrets, know-how and other technological
innovation, we may suffer competitive harm.
29
We rely to a large extent
on unpatented technology, trade secrets, confidential information and
proprietary know-how to protect our technology and maintain our competitive
position, especially when we do not believe that patent protection is
appropriate or can be obtained. Trade secrets are difficult to protect. In order
to protect proprietary technology and processes, we rely in part on
confidentiality and intellectual property assignment agreements with our
employees, consultants and others. These agreements generally provide that the
individual must keep confidential and not disclose to other parties any
confidential information developed or learned by the individual during the
course of the individual’s relationship with us except in limited circumstances.
These agreements generally also provide that we shall own all inventions
conceived by the individual in the course of rendering services to us. These
agreements may not effectively prevent disclosure of confidential information or
result in the effective assignment to us of intellectual property, and may not
provide an adequate remedy in the event of unauthorized disclosure of
confidential information or other breaches of the agreements. In addition,
others may independently discover trade secrets and proprietary information that
have been licensed to us or that we own, and in such case we could not assert
any trade secret rights against such party.
Enforcing a claim that a
party illegally obtained and is using trade secrets that have been licensed to
us or that we own is difficult, expensive and time-consuming, and the outcome is
unpredictable. In addition, courts outside the United States may be less willing
to protect trade secrets. Costly and time-consuming litigation could be
necessary to seek to enforce and determine the scope of our proprietary rights,
and failure to obtain or maintain trade secret protection could have a material
adverse effect on our business. Moreover, some of our academic institution
licensors, collaborators and scientific advisors have rights to publish data and
information to which we have rights. If we cannot maintain the confidentiality
of our technologies and other confidential information in connection with our
collaborations, our ability to protect our proprietary information or obtain
patent protection in the future may be impaired, which could have a material
adverse effect on our business.
Other Risks Related to
Our Business
We depend on
attracting and retaining key management and scientific personnel and the loss of
these personnel could impair the development of our products
candidates.
Our success depends on our
continued ability to attract, retain and motivate highly qualified management,
clinical and scientific personnel and on our ability to develop and maintain
important relationships with academic institutions, clinicians and scientists.
We are highly dependent upon our senior scientific staff, many of whom have
developed very specialized expertise in their position. The loss of services of
one or more members of our senior scientific staff could significantly delay or
prevent the successful completion of our clinical trials or commercialization of
our product candidates. The employment of each of our employees with us is “at
will,” and each employee can terminate his or her employment with us at any
time. We do not have a succession plan in place for any of our officers and key
employees. We do not carry insurance on any of our other key employees and,
accordingly, their death or disability may have a material adverse effect on our
business.
The competition for
qualified personnel in the life sciences field is intense. We will need to hire
additional personnel, including regulatory and sales personnel, as we continue
to expand our development activities. We may not be able to attract and retain
quality personnel on acceptable terms given our geographic location and the
competition for such personnel among life sciences, biotechnology,
pharmaceutical and other companies. If we are unable to attract new employees
and retain existing employees, we may be unable to continue our development and
commercialization activities and our business may be harmed.
30
We face intense
competition in the biotechnology and healthcare industries.
We face, and will continue
to face, intense competition from pharmaceutical, biopharmaceutical, medical
device and biotechnology companies developing heart failure treatments both in
the United States and abroad, as well as numerous academic and research
institutions, governmental agencies and private organizations engaged in drug
discovery activities or funding both in the United States and abroad. We also
face competition from entities and healthcare providers using more traditional
methods, such as surgery and pharmaceutical regimens, to treat heart failure. We
believe there are a substantial number of heart failure products under
development by numerous pharmaceutical,
biopharmaceutical, medical device and biotechnology companies, and it is likely
that other competitors will emerge. We are also aware of several competitors
developing cell-based therapies for the treatment of heart damage, including
Aastrom Bioscience, Inc., Aldagen, Inc., Angioblast Systems, Inc., Athersys,
Inc., Baxter International, Inc., Cytori Therapeutics, Inc., MG Biotherapeutics,
LLC (a joint venture between Genzyme Corporation and Medtronic, Inc.), Mytogen,
Inc. (a wholly-owned subsidiary of Advanced Cell Technology, Inc.), Osiris
Therapeutics, Inc., ViaCell, Inc. (a wholly-owned subsidiary of PerkinElmer,
Inc.) and potentially others. We also recognize that there may be competitors
and competing technologies, therapies and/or products that we are not aware of.
These third parties also
compete with us in recruiting and retaining qualified personnel, establishing
clinical trial sites and registering patients for clinical trials, as well as
acquiring or licensing intellectual property and technology.
Many competitors have more
experience than we do in research and development, marketing, cell culturing,
manufacturing, preclinical testing, conducting clinical trials, obtaining FDA
and foreign regulatory approvals and marketing approved products. The
competitors, some of which have their own sales and marketing organizations,
have greater financial and technical resources than we do and may be better
equipped than we are to sell competing products, obtain patents that block or
otherwise inhibit our ability to further develop and commercialize our product
candidates, obtain approvals from the FDA or other regulatory agencies for
products more rapidly than we do, or develop treatments or cures that are safer
or more effective than those we propose to develop.
In addition, academic
institutions, governmental agencies, and other public and private organizations
conducting research in the field of heart damage may seek patent protection with
respect to potentially competitive products or technologies and may establish
exclusive collaborative or licensing relationships with our competitors.
MyoCell is a clinical
therapy designed to be utilized at least a few months after a patient has
suffered heart damage. Our competitors may discover technologies and techniques
for the acute treatment of heart failure, which, if successful in treating heart
failure shortly after its occurrence, may reduce the market size for treatments
for chronic heart damage, including MyoCell.
Our industry is
subject to rapid technological change.
Our industry is subject to
rapid technological change and our cellular-based therapies involve new and
rapidly developing technology. Our competitors may discover and develop new
technologies and techniques, or enter into partnerships with collaborators in
order to develop, competing products that are more effective or less costly than
the product candidates we hope to secure regulatory approval for. In light of
the industry’s limited experience with cell-based therapies and the dedication
of significant resources to a better understanding of this field, we expect
these cell-based technologies to undergo significant change in the future. For
example, some of our competitors are exploring whether the use of cells, other
than myoblasts, is safer or more effective than MyoCell. If there is rapid
technological development or new product introductions, our current and future
product candidates or methods may become obsolete or noncompetitive before or
after we commercialize them.
Risks Related to the
Transactions with Magna
Equities II, LLC
Funding from our
Purchase Agreement with Magna Equities II, LLC may be limited or insufficient to
fund our operations or to implement our strategy.
Under our Purchase
Agreement with Magna Equities II, LLC, upon effectiveness of the registration
statement of which this prospectus is a part, and subject to other conditions,
we may direct Magna Equities II, LLC to purchase up to $3,000,000 of our shares
of common stock over a 24-month period. Although the Purchase Agreement provides
that we may sell up to $3,000,000 of our common stock to Magna Equities II, LLC,
only 143,812,591 shares of our common stock are being offered under this
prospectus, which represents (i) 31,000,000 shares of common stock that may be
issued to Magna Equities II, LLC upon conversion of the Convertible Note, (ii)
9,109,128 shares of common stock that we
issued to Magna Equities II, LLC as Initial Commitment Shares on October 27,
2014, (iii) a maximum of 15,890,872 shares of common stock that we may be
required to issue to Magna Equities II, LLC as Additional Commitment Shares and
(iv) 87,812,591 shares of common stock that we may issue to Magna Equities II,
LLC as Shares pursuant to draw downs under the Purchase
Agreement.
31
At an assumed purchase
price of $0.01460 (equal to 93% of the closing price of our common stock of
$0.01570 on November 10, 2014), and assuming the sale by us to Magna Equities
II, LLC of all of the 87,812,591 Shares, or approximately 15.7% of our issued
and outstanding common stock, including the issuance of such shares, being
registered hereunder pursuant to draw downs under the Purchase Agreement, we
would receive only approximately $1,282,064 in gross proceeds. Furthermore, we
may receive substantially less than $1,282,064 in gross proceeds from the
financing due to our share price, discount to market and other factors relating
to our common stock. If we elect to issue and sell more than the 87,812,591
Shares offered under this prospectus to Magna Equities II, LLC, which we have
the right, but not the obligation, to do, we must first register for resale
under the Securities Act any such additional Shares, which could cause
additional substantial dilution to our stockholders. Based on the above
assumptions, we would be required to register an additional approximately
117,666,849 shares of our common stock to obtain the balance of $1,717,936 of
the Total Commitment that would be available to us under the Purchase Agreement.
We currently have authorized and available for issuance 2,000,000,000 shares of
our common stock pursuant to our charter. Depending on the price at which Shares
are ultimately sold, we may have to increase the number of our authorized shares
in order to issue Shares to Magna Equities II, LLC.
There can be no assurance
that we will be able to receive all or any of the Total Commitment from Magna
Equities II, LLC because the Purchase Agreement contains certain limitations,
restrictions, requirements, conditions and other provisions that could limit our
ability to cause Magna Equities II, LLC to buy common stock from us. For
instance, we are prohibited from issuing a Draw Down Notice if the amount
requested in such Draw Down Notice exceeds the Maximum Draw Down Amount, or the
sale of Shares pursuant to the Draw Down Notice would cause us to sell or Magna
Equities II, LLC to purchase an aggregate number of shares of the Company’s
common stock which would result in beneficial ownership by Magna Equities II,
LLC of more than 9.99% of our common stock (as calculated pursuant to Section
13(d) of the Exchange Act and the rules and regulations thereunder). Moreover,
there are limitations with respect to the frequency with which we may provide
Draw Down Notices to Magna Equities II, LLC under the Purchase Agreement. Also,
as discussed above, there must be an effective registration statement covering
the resale of any Shares to be issued pursuant to any draw down under the
Purchase Agreement, and the registration statement of which this prospectus is a
part covers the resale of only 87,812,591 Shares that may be issuable pursuant
to draw downs under the Purchase Agreement. These registration statements may be
subject to review and comment by the staff of the Commission, and will require
the consent of our independent registered public accounting firm. Therefore, the
timing of effectiveness of these registration statements cannot be assured.
The extent to which we rely
on Magna Equities II, LLC as a source of funding will depend on a number of
factors, including the amount of working capital needed, the prevailing market
price of our common stock and the extent to which we are able to secure working
capital from other sources. If obtaining sufficient funding from Magna Equities
II, LLC were to prove unavailable or prohibitively dilutive, we would need to
secure another source of funding. Even if we sell all $3,000,000 of common stock
under the Purchase Agreement with Magna Equities II, LLC, we will still need
additional capital to fully implement our current business, operating plans and
development plans.
The sale or issuance
of our common stock to Magna Equities II, LLC at a discount may cause
substantial dilution and the resale of the shares of common stock by Magna
Equities II, LLC into the public market, or the perception that such sales may
occur, could cause the price of our common stock to fall.
Under the Purchase
Agreement with Magna Equities II, LLC, upon effectiveness of the registration
statement of which this prospectus is a part, and subject to other conditions,
we may direct Magna Equities II, LLC to purchase up to $3,000,000 of our shares
of common stock over a 24-month period. We are registering an aggregate of
143,812,591 (including 9,109,128 shares already issued) shares of common stock
in the registration statement of which this prospectus is a part pursuant to the
Registration Rights Agreement, representing shares which have been and may be
issuable to Magna Equities II, LLC under the Purchase Agreement and shares underlying the Convertible Note we issued to Magna
Equities II, LLC. Notwithstanding Magna Equities II, LLC’s beneficial ownership
limitation set forth in the Purchase Agreement and the Convertible Note, if all
of the 143,812,591 shares offered under this prospectus were issued and
outstanding as of November 20, 2014, such shares would represent approximately
20.7% of the total number of shares of our common stock outstanding and 20.9% of
the total number of outstanding shares of our common stock held by
non-affiliates, in each case as of November 20, 2014. The number of shares
ultimately offered for sale by Magna Equities II, LLC under this prospectus is
dependent upon a number of factors, including the extent to which Magna Equities
II, LLC converts the Convertible Note into shares of our common stock and the
number of Shares we ultimately issue and sell to Magna Equities II, LLC under
the Purchase Agreement. Because the actual purchase price for the Shares that we
may sell to Magna Equities II, LLC will fluctuate based on the market price of
our common stock during the term of the Purchase Agreement, we are not able to
determine at this time the exact number of shares of our common stock that we
will issue under the Purchase Agreement and, therefore, the exact number of
shares we will ultimately register for resale under the Securities Act.
32
Specifically, because the
per share purchase price for the Shares subject to a Draw Down Notice will be
equal to a 7% discount to certain trading prices of our common stock as set
forth in the Purchase Agreement, Magna Equities II, LLC will pay less than the
then-prevailing market price for our common stock, and the actual purchase price
for the Shares that we may sell to Magna Equities II, LLC will fluctuate based
on the VWAPs and closing prices of our common stock during the term of the
Purchase Agreement. As a result of this discount, Magna Equities II, LLC may
have a financial incentive to sell our common stock immediately to realize the
profit equal to the difference between the purchase price and the market price.
If Magna Equities II, LLC sells the common stock, the market price of our common
stock could decrease. If the market price of our common stock decreases, Magna
Equities II, LLC may have a further incentive to sell the common stock that it
holds. These sales may have a further impact on the market price of our common
stock.
Moreover, there is an
inverse relationship between the market price of our common stock and the number
of shares of our common stock that may be sold pursuant to the Purchase
Agreement. That is, the lower the market price, the more shares of our common
stock that may be sold under the Purchase Agreement. Accordingly, if the market
price of our common stock decreases (whether such decrease is due to sales by
Magna Equities II, LLC in the market or otherwise) and, in turn, the purchase
price of our common stock sold to Magna Equities II, LLC under the Purchase
Agreement decreases, this could allow Magna Equities II, LLC to receive greater
numbers of shares of our common stock pursuant to draw downs under the Purchase
Agreement. Although the number of shares of our common stock that our existing
stockholders own will not decrease, the common stock owned by our existing
stockholders will represent a smaller percentage of our total outstanding shares
after any such sales to Magna Equities II, LLC. Depending on market liquidity at
the time, the sale of a substantial number of shares of our common stock to
Magna Equities II, LLC at a discount to the then-prevailing market price for our
common stock under the Purchase Agreement, and the resale of such shares by
Magna Equities II, LLC into the public market, or the perception that such sales
may occur, could cause the trading price of our common stock to decline, result
in substantial dilution to existing stockholders and make it more difficult for
us to sell equity or equity-related securities in the future at a time and at a
price that we might otherwise wish to effect sales.
For a tabular disclosure of
the number of securities and percentage ownership to be issued assuming we sell
all the securities on the registration statement and assuming sales to Magna
Equities II, LLC at various discounts to our current market price, please see
“Equity Enhancement Program With Magna Equities II, LLC” on page 42 of this
prospectus.
You may experience
immediate and substantial dilution in the net tangible book value per share of
the common stock you purchase.
The price per share of our
common stock being offered pursuant to this prospectus may be substantially
higher than the net tangible book value per share of our common stock.
Therefore, if you purchase shares of common stock in this offering at the
current market value, you may suffer immediate and substantial dilution in the
pro forma net tangible book value per share of common stock. See the section
entitled “Dilution” elsewhere in this prospectus for a more detailed discussion
of the dilution you may incur if you purchase shares in this offering.
33
We may use the net
proceeds from sales of our common stock to Magna Equities II, LLC pursuant to
the Purchase Agreement in ways with which you may disagree.
We intend to use the net
proceeds from sales of our common stock to Magna Equities II, LLC pursuant to
the Purchase Agreement for working capital and general corporate purposes. As of
the date of this prospectus, we cannot specify with certainty all of the
particular uses of the proceeds from sales of common stock to Magna Equities II,
LLC pursuant to the Purchase Agreement. Accordingly, we will have significant
discretion in the use of the net proceeds of sales of common stock to Magna
Equities II, LLC pursuant to the Purchase Agreement. It is possible that we may
allocate the proceeds differently than investors in this offering desire or that
we will fail to maximize our return on these proceeds. We may, subsequent to
this offering, modify our intended use of the proceeds from sales of common
stock to Magna Equities II, LLC pursuant to the Purchase Agreement to pursue
strategic opportunities that may arise, such as potential acquisition
opportunities. You will be relying on the judgment of our management with regard
to the use of the net proceeds from the sales of common stock to Magna Equities
II, LLC pursuant to the Purchase Agreement, and you will not have the
opportunity, as part of your investment decision, to assess whether the proceeds
are being used appropriately. Any failure to apply the proceeds from sales of
common stock to Magna Equities II, LLC pursuant to the Purchase Agreement
effectively could have a material adverse effect on our business and cause a
decline in the market price of our common stock.
Risks Related to Our
Common Stock
An active, liquid and
orderly trading market for our common stock may not
continue.
Prior to our initial public
offering, there was no public market for our common stock. Following our initial
public offering, our common stock was traded on the NASDAQ Global Market and,
subsequently, the NASDAQ Capital Market. On February 25, 2009, we received
notification from The NASDAQ Stock Market of its determination to discontinue
our NASDAQ listing effective February 27, 2009. We filed for quotation of our
common stock on the Over-The-Counter Bulletin Board and that application was
approved by FINRA on May 13, 2009. On February 23, 2011 we received notice of
FINRA’s determination of our ineligibility for continued quotation of our shares
on the OTCBB due to quoting inactivity under SEC Rule 15c2-11. On March 9, 2011
we again filed for quotation of our common stock on the Over-The-Counter
Bulletin Board and that application was approved by FINRA on March 29, 2011.
We cannot offer any
assurance that an active trading market for our common stock will continue or
how liquid that market may be. As a result, relatively small trades may have a
disproportionate impact on the price of our common stock, which may contribute
to the price volatility of our common stock and could limit your ability to sell
your shares.
The market price of our
common stock could also be subject to wide fluctuations in response to many risk
factors described in this section and other matters, including:
- publications of clinical trial results
by clinical investigators or others about our products and competitors'
products and/or our industry;
- changes by securities analysts in
financial estimates of our operating results and the operating results of our
competitors;
- publications of research reports by
securities analysts about us, our competitors or our industry;
- fluctuations in the valuation of
companies perceived by investors to be comparable to us;
- actual or anticipated fluctuations in
our quarterly or annual operating results;
- retention and departures of key
personnel;
- our failure or the failure of our
competitors to meet analysts' projections or guidance that we or our
competitors may give to the market;
- strategic decisions by us or our
competitors, such as acquisitions, divestitures, spin-offs, joint ventures,
strategic investments or changes in business strategy;
- the passage of legislation or other
regulatory developments affecting us or our industry;
- speculation in the press or investment community; and
- natural disasters, terrorist acts, act of war or periods of widespread
civil unrest.
34
Furthermore, the stock
markets have experienced extreme price and volume fluctuations that have
affected and continue to affect the market prices of equity securities of many
companies, especially life sciences and pharmaceutical companies. These
fluctuations often have been unrelated or disproportionate to the operating
performance of those companies. These broad market and industry fluctuations, as
well as general economic, political and market conditions, may negatively affect
the market price of our common stock. As a result, the market price of our
common stock is likely to be similarly volatile and investors in our common
stock may experience a decrease, which could be substantial, in the value of
their stock. In the past, many companies that have experienced volatility in the
market price of their stock have been subject to securities class action
litigation. We may be the target of this type of litigation in the future.
Securities litigation against us could result in substantial costs and divert
our management's attention from other business concerns, which could have a
material adverse effect on our business.
Anti-takeover
provisions of Florida law, our articles of incorporation and our bylaws may
prevent or delay an acquisition of us that shareholders may consider favorable
or attempts to replace or remove our management that could be beneficial to our
shareholders.
Our articles of
incorporation and bylaws contain provisions, such as the right of our directors
to issue preferred stock from time to time with voting, economic and other
rights superior to those of our common stock without the consent of our
shareholders, all of which could make it more difficult for a third party to
acquire us without the consent of our board of directors. In addition, our
bylaws impose restrictions on the persons who may call special shareholder
meetings. Furthermore, the Florida Business Corporation Act contains an
“affiliated transaction” provision that prohibits a publicly-held Florida
corporation from engaging in a broad range of business combinations or other
extraordinary corporate transactions with an “interested shareholder” unless,
among others, (i) the transaction is approved by a majority of disinterested
directors before the person becomes an interested shareholder; (ii) the
interested shareholder has owned at least 80% of the corporation's outstanding
voting shares for at least five years; or (iii) the transaction is approved by
the holders of two-thirds of the corporation's voting shares other than those
owned by the interested shareholder. An interested shareholder is defined as a
person who together with affiliates and associates beneficially owns more than
10% of the corporation's outstanding voting shares. The Florida Business
Corporation Act also prohibits the voting of shares in a publicly-held Florida
corporation that are acquired in a “control share acquisition” unless the
holders of a majority of the corporation's voting shares (exclusive of shares
held by officers of the corporation, inside directors or the acquiring party)
approve the granting of voting rights as to the shares acquired in the control
share acquisition or unless the acquisition is approved by the corporation's
Board of Directors. These provisions may have the effect of delaying or
preventing a change of control of our company even if this change of control
would benefit our shareholders.
We do not intend to
pay cash dividends on our common stock in the foreseeable future and,
accordingly, capital appreciation of our common stock, if any, will be a
shareholder's sole source of gain from an investment in our common
stock.
Our policy is to retain
earnings to provide funds for the operation and expansion of our business and,
accordingly, we have never declared or paid any cash dividends on our common
stock or other securities and do not currently anticipate paying any cash
dividends in the foreseeable future. Consequently, shareholders will need to
sell shares of our common stock to realize a return on their investments, if
any, and this capital appreciation, if any, will be a shareholder's sole source
of gain from an investment in the common stock.
The declaration and payment
of dividends by us are subject to the discretion of our Board of Directors and
the restrictions specified in our articles of incorporation and by applicable
law. In addition, under the terms of the Northstar Loan, we are restricted from
paying cash dividends to our shareholders while this loan is outstanding. Any
future determination to pay cash dividends will depend on our results of
operations, financial condition, capital requirements, contractual restrictions
and other factors deemed relevant by our Board of Directors.
35
Our common stock may
be considered a “penny stock,” and thereby be subject to additional sale and
trading regulations that may make it more difficult to sell.
Our common stock is
considered to be a “penny stock.” It does not qualify for one of the exemptions
from the definition of “penny stock” under Section 3a51-1 of the Exchange Act.
Our common stock is a “penny stock” because it meets one or more of the
following conditions (i) the stock trades at a price less than $5.00 per share;
(ii) it is not traded on a “recognized” national exchange or (iii) it is not
quoted on the NASDAQ Global Market, nor has a price less than $5.00 per share.
The principal result or effect of being designated a “penny stock” is that
securities broker-dealers participating in sales of our common stock are subject
to the “penny stock” regulations set forth in Rules 15-2 through 15g-9
promulgated under the Securities Exchange Act. For example, Rule 15g-2 requires
broker-dealers dealing in penny stocks to provide potential investors with a
document disclosing the risks of penny stocks and to obtain a manually signed
and dated written receipt of the document at least two business days before
effecting any transaction in a penny stock for the investor's account. Moreover,
Rule 15g-9 requires broker-dealers in penny stocks to approve the account of any
investor for transactions in such stocks before selling any penny stock to that
investor. This procedure requires the broker-dealer to (i) obtain from the
investor information concerning his or her financial situation, investment
experience and investment objectives; (ii) reasonably determine, based on that
information, that transactions in penny stocks are suitable for the investor and
that the investor has sufficient knowledge and experience as to be reasonably
capable of evaluating the risks of penny stock transactions; (iii) provide the
investor with a written statement setting forth the basis on which the
broker-dealer made the determination in (ii) above; and (iv) receive a signed
and dated copy of such statement from the investor, confirming that it
accurately reflects the investor's financial situation, investment experience
and investment objectives. Compliance with these requirements may make it more
difficult and time consuming for holders of our common stock to resell their
shares to third parties or to otherwise dispose of them in the market or
otherwise.
FINRA sales practice
requirements may limit a shareholder’s ability to buy and sell our common
shares.
In addition to the “penny
stock” rules described above, FINRA has adopted rules that require that in
recommending an investment to a customer, a broker-dealer must have reasonable
grounds for believing that the investment is suitable for that customer. Prior
to recommending speculative low priced securities to their non-institutional
customers, broker-dealers must make reasonable efforts to obtain information
about the customer’s financial status, tax status, investment objectives and
other information. Under interpretations of these rules, FINRA believes that
there is a high probability that speculative low priced securities will not be
suitable for at least some customers. FINRA requirements make it more difficult
for broker-dealers to recommend that their customers buy our common shares,
which may limit your ability to buy and sell our stock and have an adverse
effect on the market for our shares.
Rule 144 sales in the
future may have a depressive effect on the company's stock price as an increase
in supply of shares for sale, with no corresponding increase in demand will
cause prices to fall.
All of the outstanding
shares of common stock held by the present officers, directors, and affiliate
stockholders are "restricted securities" within the meaning of Rule 144 under
the Securities Act of 1933, as amended. As restricted shares, these shares may
be resold only pursuant to an effective registration statement or under the
requirements of Rule 144 or other applicable exemptions from registration under
the Securities Act of 1933 and as required under applicable state securities
laws. Rule 144 provides in essence that a person who is an affiliate or officer
or director who has held restricted securities for six months may, under certain
conditions, sell every three months, in brokerage transactions, a number of
shares that does not exceed the greater of 1.0% of a Company's issued and
outstanding common stock. There is no limit on the amount of restricted
securities that may be sold by a non-affiliate after the owner has held the
restricted securities for a period of six months if the Company is a current
reporting company under the Securities Exchange Act of 1934. A sale under Rule
144 or under any other exemption from the Securities Act of 1933, if available,
or pursuant to subsequent registration of shares of common stock of present
stockholders, may have a depressive effect upon the price of the common stock in
any market that may develop. In addition, if we are deemed a shell company
pursuant to Section 12(b)-2 of the Act, our "restricted securities", whether
held by affiliates or non-affiliates, may not be re-sold for a period of 12
months following the filing of a Form 10 level disclosure or registration
pursuant to the Securities Act of 1933.
36
Future issuances of
shares for various considerations including working capital and operating
expenses will increase the number of shares outstanding which will dilute
existing investors and may have a depressive effect on the company's stock
price.
There may be substantial
dilution to our shareholders purchasing in future offerings as a result of
future decisions of the Board to issue shares without shareholder approval for
cash, services, payment of debt or acquisitions.
There may in all
likelihood be little demand for shares of our common stock and as a result
investors may be unable to sell at or near ask prices or at all if they need to
liquidate their investment.
There may be little demand
for shares of our common stock on the OTC Bulletin Board, or OTC Markets.com,
meaning that the number of persons interested in purchasing our common shares at
or near ask prices at any given time may be relatively small or non-existent.
This situation is attributable to a number of factors, including the fact that
it is a small company which is relatively unknown to stock analysts, stock
brokers, institutional investors and others in the investment community that
generate or influence sales volume, and that even if the Company came to the
attention of such persons, they tend to be risk-averse and would be reluctant to
follow an unproven, early stage company such as ours or purchase or recommend
the purchase of any of our Securities until such time as it became more seasoned
and viable. As a consequence, there may be periods of several days or more when
trading activity in the Company's securities is minimal or non-existent, as
compared to a seasoned issuer which has a large and steady volume of trading
activity that will generally support continuous sales without an adverse effect
on the securities price. We cannot give investors any assurance that a broader
or more active public trading market for the Company's common securities will
develop or be sustained, or that any trading levels will be sustained. Due to
these conditions, we can give investors no assurance that they will be able to
sell their shares at or near ask prices or at all if they need money or
otherwise desire to liquidate their securities of the Company.
Failure to achieve
and maintain effective internal controls in accordance with Section 404 of the
Sarbanes-Oxley Act could have a material adverse effect on our business and
operating results.
It may be time consuming,
difficult and costly for us to develop and implement the additional internal
controls, processes and reporting procedures required by the Sarbanes-Oxley Act.
We may need to hire additional financial reporting, internal auditing and other
finance staff in order to develop and implement appropriate additional internal
controls, processes and reporting procedures.
If we fail to comply in a
timely manner with the requirements of Section 404 of the Sarbanes-Oxley Act
regarding internal control over financial reporting or to remedy any material
weaknesses in our internal controls that we may identify, such failure could
result in material misstatements in our financial statements, cause investors to
lose confidence in our reported financial information and have a negative effect
on the trading price of our common stock.
Pursuant to Section 404 of
the Sarbanes-Oxley Act and current SEC regulations, we are required to prepare
assessments regarding internal controls over financial reporting and, furnish an
annual report by our management on our internal control over financial
reporting. We continue to document and test our internal control procedures in
order to satisfy these requirements, which is likely to result in increased
general and administrative expenses and may shift management time and attention
from revenue-generating activities to compliance activities. While our
management is expending significant resources in an effort to complete this
important project, there can be no assurance that we will be able to achieve our
objective on a timely basis. Failure to achieve and maintain an effective
internal control environment or complete our Section 404 certifications could
have a material adverse effect on our stock price.
In addition, in connection
with our on-going assessment of the effectiveness of our internal control over
financial reporting, we may discover “material weaknesses” in our internal
controls as defined in standards established by the Public Company Accounting
Oversight Board, or the PCAOB. A material weakness is a significant deficiency, or combination of
significant deficiencies, that results in more than a remote likelihood that a
material misstatement of the annual or interim financial statements will not be
prevented or detected. The PCAOB defines “significant deficiency” as a
deficiency that results in more than a remote likelihood that a misstatement of
the financial statements that is more than inconsequential will not be prevented
or detected.
37
In the event that a
material weakness is identified, we will employ qualified personnel and adopt
and implement policies and procedures to address any material weaknesses that we
identify. However, the process of designing and implementing effective internal
controls is a continuous effort that requires us to anticipate and react to
changes in our business and the economic and regulatory environments and to
expend significant resources to maintain a system of internal controls that is
adequate to satisfy our reporting obligations as a public company. We cannot
assure you that the measures we will take will remediate any material weaknesses
that we may identify or that we will implement and maintain adequate controls
over our financial process and reporting in the future.
Currently our CEO and
Principal Financial and Accounting Officer, concluded that our disclosure
controls and procedures were ineffective as of the end of September 30, 2014,
due to the Company’s limited resources and limited number of employees. To
mitigate the current limited resources and limited employees, we rely heavily on
direct management oversight of transactions, along with the use of legal and
outsourced accounting professionals. As we grow, we expect to increase, our
number of employees, which, we believe, will enable us to implement adequate
segregation of duties within the internal control framework.
Any failure to complete our
assessment of our internal control over financial reporting, to remediate any
material weaknesses that we may identify or to implement new or improved
controls, or difficulties encountered in their implementation, could harm our
operating results, cause us to fail to meet our reporting obligations or result
in material misstatements in our financial statements. Any such failure could
also adversely affect the results of the periodic management evaluations of our
internal controls and, in the case of a failure to remediate any material
weaknesses that we may identify, would adversely affect the annual auditor
attestation reports regarding the effectiveness of our internal control over
financial reporting that are required under Section 404 of the Sarbanes-Oxley
Act. Inadequate internal controls could also cause investors to lose confidence
in our reported financial information, which could have a negative effect on the
trading price of our common stock.
Public disclosure
requirements and compliance with changing regulation of corporate governance
pose challenges for our management team and result in additional expenses and
costs which may reduce the focus of management and the profitability of our
company.
Changing laws, regulations
and standards relating to corporate governance and public disclosure, including
the Dodd-Frank Wall Street Reform and Consumer Protection Act and the rules and
regulations promulgated thereunder, the Sarbanes-Oxley Act and SEC regulations,
have created uncertainty for public companies and significantly increased the
costs and risks associated with accessing the U.S. public markets. Our
management team will need to devote significant time and financial resources to
comply with both existing and evolving standards for public companies, which
will lead to increased general and administrative expenses and a diversion of
management time and attention from revenue generating activities to compliance
activities.
SHOULD ONE OR MORE OF
THE FOREGOING RISKS OR UNCERTAINTIES MATERIALIZE, OR SHOULD THE UNDERLYING
ASSUMPTIONS PROVE INCORRECT, ACTUAL RESULTS MAY DIFFER SIGNIFICANTLY FROM THOSE
ANTICIPATED, BELIEVED, ESTIMATED, EXPECTED, INTENDED OR
PLANNED
38
CAUTIONARY NOTE
REGARDING FORWARD-LOOKING STATEMENTS
This prospectus includes
“forward-looking” statements regarding our business development plans, clinical
trials, regulatory reviews, timing, strategies, expectations, anticipated
expenses levels, projected profits, business prospects and positioning
with respect to market, demographic and pricing trends, business outlook,
technology spending and various other matters (including contingent liabilities
and obligations and changes in accounting policies, standards and
interpretations) and express our current intentions, beliefs, expectations,
strategies or predictions, as well as historical information. Such
forward-looking statements involve known and unknown risks, uncertainties and
other factors which may cause our actual results, performance or achievements,
or industry results, to be materially different from anticipated results,
performance or achievements expressed or implied by such forward-looking
statements. When used in this prospectus, statements that are not statements of
current or historical fact may be deemed to be forward-looking statements.
Without limiting the foregoing, the words “plan,” “intend,” “may,” “will,”
“expect,” “believe,” “could,” “anticipate,” “estimate,” or “continue” or similar
expressions or other variations or comparable terminology are intended to
identify such forward-looking statements. Although we believe that the
assumptions on which the forward-looking statements contained herein are based
are reasonable, any of those assumptions could prove to be inaccurate given the
inherent uncertainties as to the occurrence or nonoccurrence of future events.
These statements are not guarantees of future performance and involve risks and
uncertainties that are difficult to predict. Our future operating results are
dependent upon many factors, and our further development is highly dependent on
market acceptance, which is outside its control. Forward-looking statements may
not be realized due to a variety of factors, including, without limitation, (i)
our ability to manage the business despite continuing operating losses and cash
outflows; (ii) our ability to obtain sufficient capital or a strategic business
arrangement to fund its operations and expansion plans, including meeting its
financial obligations under various licensing and other strategic arrangements
and the successful commercialization of the relevant technology; (iii) our
ability to build the management and human resources and infrastructure necessary
to support the growth of the business; (iv) competitive factors and developments
beyond our control; (v) scientific and medical developments beyond our control;
(vi) limitations caused by government regulation of the business; (vii) whether
any of our current or future patent applications result in issued patents and
our ability to obtain and maintain other rights to technology required or
desirable for the conduct of our business; (viii) whether any potential
strategic benefits of various licensing transactions will be realized and
whether any potential benefits from the acquisition of these new licensed
technologies will be realized; (ix) our ability to maintain our eligibility for
quotation on the Over-The-Counter Bulletin Board; and (x) the other factors
discussed in “Risk Factors” and elsewhere in this prospectus.
All forward-looking
statements attributable to us are expressly qualified in their entirety by these
and other factors. We undertake no obligation to update or revise these
forward-looking statements, whether to reflect events or circumstances after the
date initially filed or published, to reflect the occurrence of unanticipated
events or otherwise, except to the extent required by federal securities laws.
USE OF
PROCEEDS
Selling Stockholder may
sell all of the common stock offered by this Prospectus from time-to-time. We
will not receive any proceeds from the sale of those shares of common stock. We
may, however, receive aggregate gross proceeds of $1,717,936 if all shares of
common stock in this offering are sold to Selling Stockholder pursuant to the
Purchase Agreement. Any such proceeds we receive will be used for working
capital and general corporate matters.
The exact purchase price at
which we will sell the Purchase Shares pursuant to the Purchase Agreement is
dependent upon the market price for our common stock; therefore, it is likely
that the number of shares offered in this registration statement will be
insufficient to allow us to receive the full amount of proceeds under the
Purchase Agreement.
At an assumed purchase
price of $0.01570 (equal to the closing price of our common stock of $0.01570 on
November 10, 2014), and assuming the sale by us to Magna Equities II, LLC of all
of the 143,812,591Shares (including 9,109,128 issued shares), or approximately
20.5% of our issued and outstanding common stock, being registered hereunder
pursuant to draw downs under the Purchase Agreement, we would receive only
approximately $1,282,064 in gross proceeds. Furthermore, we may receive
substantially less than $1,282,064 in gross proceeds from the financing due to
our share price, discount to market and other factors relating to our common
stock. If we elect to issue and sell more
than the 143,812,591 Shares (including 9,109,128 issued shares) offered under
this prospectus to Magna, which we have the right, but not the obligation, to
do, we must first register for resale under the Securities Act any such
additional Shares, which could cause additional substantial dilution to our
stockholders. Based on the above assumptions, we would be required to register
an additional approximately 117,666,849 shares of our common stock to obtain the
balance of $1,717,936 of the Total Commitment that would be available to us
under the Purchase Agreement.
39
DETERMINATION OF
OFFERING PRICE
There currently is a
limited public market for our common stock. Selling Stockholder will determine
at what price it may sell the offered shares, and such sales may be made at
prevailing market prices or at privately negotiated prices. See “Plan of
Distribution” below for more information.
SELLING STOCKHOLDER
This prospectus relates to
the possible resale from time to time by the selling stockholder of any or all
of the shares of common stock that have been or may be issued by us to Magna
under the Purchase Agreement and upon conversion of the Convertible Note. For
additional information regarding the issuance of common stock covered by this
prospectus, see “Summary—Recent Developments” and “Equity Enhancement Program
With Magna” above. We are registering the shares of common stock pursuant to the
provisions of the Note Registration Rights Agreement we entered into with Magna
on October 7, 2014 and the Registration Rights Agreement we entered into with
Magna on October 23, 2014, in order to permit the selling stockholder to offer
the shares for resale from time to time. Except for the transactions
contemplated by the Convertible Note, the Note Purchase Agreement, the Note
Registration Rights Agreement, the Purchase Agreement and the Registration
Rights Agreement, Magna has not had any material relationship with us within the
past three years.
The table below presents
information regarding the selling stockholder and the shares of common stock
that it may offer from time to time under this prospectus. This table is
prepared based on information supplied to us by the selling stockholder, and
reflects holdings as of November 20, 2014. As used in this prospectus, the term
“selling stockholder” means Magna. The number of shares in the column “Maximum
Number of Shares of Common Stock to be Offered Pursuant to this Prospectus”
represents all of the shares of common stock that the selling stockholder may
offer under this prospectus. The selling stockholder may sell some, all or none
of its shares in this offering. We do not know how long the selling stockholder
will hold the shares before selling them, and we currently have no agreements,
arrangements or understandings with the selling stockholder regarding the sale
of any of the shares.
Beneficial ownership is
determined in accordance with Rule 13d-3(d) promulgated by the SEC under the
Exchange Act, and includes shares of common stock with respect to which the
selling stockholder has voting and investment power. The percentage of shares of
common stock beneficially owned by the selling stockholder prior to the offering
shown in the table below is based on an aggregate of 560,564,622 shares of our
common stock outstanding on November 20, 2014. Because the purchase price of the
shares of common stock issuable under the Purchase Agreement is determined on
each settlement date, and because the principal amount under the Convertible
Note may be reduced under certain circumstances (thereby resulting in fewer
shares being issued to Magna upon conversion of the Convertible Note), the
number of shares that may actually be sold by the Company under the Purchase
Agreement and the number of shares that may actually be issued to Magna upon
conversion of the Convertible Note may be fewer than the number of shares being
offered by this prospectus. The fourth column assumes the sale of all of the
shares offered by the selling stockholder pursuant to this prospectus.
40
|
|
Maximum Number of |
|
|
|
Shares of Common |
|
|
Number of Shares of |
Stock to be Offered |
Number of
Shares of |
|
Common Stock Owned |
Pursuantto
this |
Common
Stock Owned |
| Name of Selling
Stockholder |
Prior to
Offering |
Prospectus |
After
Offering |
|
Number(1) |
Percent(2) |
|
Number(3) |
Percent(2) |
| Magna Equities II, LLC
(4) |
|
|
143,812,591 (includes |
|
|
|
|
|
9,109,128 shares |
|
|
|
|
|
issued October
27, |
|
|
|
40,109,128 |
4.99% |
2014) |
-0- |
0% |
| (1) |
|
This number
represents the beneficial ownership of up to 31,000,000 shares of common
stock underlying a senior convertible note we issued to MEII on October 7,
2014, which provides that we may not issue shares of our common stock to
Magna to the extent that Magna or any of its affiliates would, at any
time, beneficially own more than 4.99% of our outstanding common stock and
(ii) 9,109,128 shares of common stock we issued to Magna on October 23,
2014 as Initial Commitment Shares in consideration for entering into the
Purchase Agreement with us. In accordance with Rule 13d-3(d) under the
Exchange Act, we have excluded from the number of shares beneficially
owned prior to the offering (i) up to 15,890,872 shares that may be issued
to Magna as Additional Commitment Shares under the terms of the Purchase
Agreement, because the issuance of such shares is dependent on, among
other things, the registration statement of which this prospectus is a
part becoming effective; and (ii) all of the shares that Magna may be
required to purchase pursuant to draw downs under the Purchase Agreement,
because the issuance of such shares is solely at our discretion and is
subject to certain conditions, the satisfaction of all of which are
outside of Magna’s control, including the registration statement of which
this prospectus is a part becoming and remaining effective. Furthermore,
the maximum amount of each put of common stock to Magna under the Purchase
Agreement is subject to certain agreed upon threshold limitations set
forth in the Purchase Agreement. Also, under the terms of the Purchase
Agreement, we may not issue shares of our common stock to Magna to the
extent that Magna or any of its affiliates would, at any time,
beneficially own more than 9.99% of our outstanding common
stock. |
| (2) |
|
Applicable percentage
ownership is based on 560,564,622 shares of our common stock outstanding
as of November 20, 2014. Pursuant to the senior convertible note we issued
to MEII on October 7, 2014, we may not issue shares of our common stock to
Magna to the extent that Magna or any of its affiliates would, at any
time, beneficially own more than 4.99% of our outstanding common stock. If
the contractual limitations were waived and senior convertible note we
issued to MEII on October 7, 2014 was fully converted, MEII would have up
to 6.74% of our outstanding common stock. |
| (3) |
|
Assumes the sale of
all shares being offered pursuant to this prospectus. |
| (4) |
|
The business address
of Magna Equities II, LLC is c/o Magna, 5 Magna Square, New York, New York
10004. Magna’s principal business is that of a private investment firm. We
have been advised that Magna is not a member of the Financial Industry
Regulatory Authority, or FINRA, or an independent broker-dealer, and that
neither Magna nor any of its affiliates is an affiliate or an associated
person of any FINRA member or independent broker-dealer. We have been
further advised that Joshua Season is the Chief Executive Officer and
managing member of Magna and owns all of the membership interests in
Magna, and that Mr. Season has sole power to vote or to direct the vote
and sole power to dispose or to direct the disposition of all securities
owned directly by Magna. |
41
EQUITY ENHANCEMENT
PROGRAM WITH MAGNA EQUITIES II, LLC
Common Stock Purchase
Agreement
On the Closing Date, we
entered into the Purchase Agreement with Magna Equities II, LLC. The Purchase
Agreement provides that, upon the terms and subject to the conditions set forth
therein, Magna Equities II, LLC is committed to purchase up to the Total
Commitment worth of Shares over the 24-month term of the Purchase Agreement.
From time to time over the
term of the Purchase Agreement, commencing on the trading day immediately
following the date on which the registration statement of which this prospectus
is a part is declared effective by the Commission, we may, in our sole
discretion, provide Magna Equities II, LLC with a Draw Down Notice to purchase a
specified Draw Down Amount of Shares, with each draw down subject to the
limitations discussed below. The maximum dollar amount of Shares requested to be
purchased pursuant to any single Draw Down Notice cannot exceed the Maximum Draw
Down Amount. We may not deliver any Draw Down Notice to Magna Equities II, LLC
if the Initial Purchase Price with respect to the Shares subject to such Draw
Down Notice is less than $0.0025 (subject to adjustment for any stock splits,
stock combinations, stock dividends, recapitalizations and other similar
transactions) as of the date the applicable Draw Down Exercise Date.
Once presented with a Draw
Down Notice, Magna Equities II, LLC is required to purchase the applicable Draw
Down Amount at the applicable Purchase Price. The applicable Settlement Date
with respect to a Draw Down Notice will occur within one trading day following
the Draw Down Exercise Date. On the applicable Settlement Date for a draw down,
we will issue to Magna Equities II, LLC a number of Shares, rounded to the
nearest whole Share, equal to (i) the Draw Down Amount that we requested from
Magna Equities II, LLC, divided by (ii) the applicable Initial Purchase Price,
against simultaneous payment by Magna Equities II, LLC to us in an amount equal
to (A) the number of Shares we issued to Magna Equities II, LLC on the
Settlement Date, multiplied by (B) the applicable Initial Purchase
Price.
With respect to a Draw Down
Notice, on the True-Up Date, a calculation of the True-Up Purchase Price and the
Purchase Price will occur. On the True-Up Settlement Date, we will issue to
Magna Equities II, LLC the Additional Shares, if any, in respect of the
applicable Draw Down Notice. Magna Equities II, LLC is not required to return
any Shares to us in the event the True-Up Purchase Price is greater than the
Initial Purchase Price.
We are prohibited from
issuing a Draw Down Notice if (i) the amount requested in such Draw Down Notice
exceeds the Maximum Draw Down Amount, (ii) the sale of Shares pursuant to such
Draw Down Notice would cause us to issue or sell or Magna Equities II, LLC to
acquire or purchase an aggregate dollar value of Shares that would exceed the
Total Commitment, or (iii) the sale of Shares pursuant to the Draw Down Notice
would cause us to sell or Magna Equities II, LLC to purchase an aggregate number
of shares of our common stock which would result in beneficial ownership by
Magna Equities II, LLC of more than 9.99% of our common stock (as calculated
pursuant to Section 13(d) of the Exchange Act, and the rules and regulations
thereunder). We cannot make more than one draw down during the period commencing
on any Draw Down Exercise Date and ending on the applicable True-Up Date for
such draw down, and we must allow at least two trading days to elapse between
the applicable True-Up Date for a draw down and the delivery of any Draw Down
Notice for any other draw down.
As of November 20, 2014,
there were 560,564,622 shares of our common stock outstanding, of which
554,375,697 shares were held by non-affiliates. Although the Purchase Agreement
provides that we may sell up to $3,000,000 of our common stock to Magna Equities
II, LLC, only 143,812,591 shares of our common stock are being offered under
this prospectus, which represents (i) 31,000,000 shares of common stock that may
be issued to Magna Equities II, LLC upon conversion of the Convertible Note,
(ii) 9,109,128 shares of common stock that we issued to Magna Equities II, LLC
as Initial Commitment Shares on October 27, 2014, (iii) a maximum of 15,890,872
shares of common stock that we may be required to issue to Magna Equities II,
LLC as Additional Commitment Shares and (iv) 87,812,591 shares of common stock
that we may issue to Magna Equities II, LLC as Shares pursuant to draw downs
under the Purchase Agreement. If all of the 143,812,591 shares offered under
this prospectus were issued and outstanding as of November 20, 2014, such shares
would represent approximately 20.7% of the total number of shares of our common
stock outstanding and 20.9% of the total number of outstanding shares of our
common stock held by non-affiliates, in each case as of November 20, 2014.
42
At an assumed purchase
price of $0.01460 (equal to 93% of the closing price of our common stock of
$0.01570 on November 10, 2014), and assuming the sale by us to Magna Equities
II, LLC of all of the 87,812,591 Shares, or approximately 15.7% of our issued
and outstanding common stock, being registered hereunder pursuant to draw downs
under the Purchase Agreement, we would receive only approximately $1,282,064 in
gross proceeds. Furthermore, we may receive substantially less than $1,282,064
in gross proceeds from the financing due to our share price, discount to market
and other factors relating to our common stock. If we elect to issue and sell
more than the 87,812,591 Shares offered under this prospectus to Magna Equities
II, LLC, which we have the right, but not the obligation, to do, we must first
register for resale under the Securities Act any such additional Shares, which
could cause additional substantial dilution to our stockholders. Based on the
above assumptions, we would be required to register an additional approximately
117,666,849 shares of our common stock to obtain the balance of $1,717,936 of
the Total Commitment that would be available to us under the Purchase Agreement.
We currently have authorized and available for issuance 2,000,000,000 shares of
our common stock pursuant to our charter.
The number of shares of our
common stock ultimately offered for resale by Magna Equities II, LLC is
dependent upon a number of factors, including the extent to which Magna Equities
II, LLC converts the Convertible Note into shares of our common stock and the
number of Shares we ultimately issue and sell to Magna Equities II, LLC under
the Purchase Agreement. The following table sets forth the total number of
Shares that would be issued at varying purchase prices for us to receive the
entire $3,000,000 in gross proceeds under the Purchase Agreement (without
accounting for certain fees and expenses):
|
|
|
Proceeds from the |
|
Total Number of |
Percentage of |
Sale of Shares to |
|
Shares to be |
Currently |
Magna Equities II, |
| Assumed Average |
Issued if |
Outstanding |
LLC Under the |
| Purchase Price(1) |
Full Purchase |
Shares (2) |
Purchase Agreement |
| $0.00365025 |
(3)
821,861,516 |
59% |
$3,000,000 |
| $0.0073005 |
(4)410,930,758 |
42% |
$3,000,000 |
| $0.01095075 |
(5)273,953,839 |
33% |
$3,000,000 |
| $0.014601 |
(6)205,465,379 |
27% |
$3,000,000 |
| $0.01825125 |
(7)164,372,303 |
23% |
$3,000,000 |
| $0.0219015 |
(8)136,976,919 |
20% |
$3,000,000 |
____________________
| (1) |
|
Because the Purchase
Price for Shares subject to any Draw Down Notice is the lesser of the
Initial Purchase Price and the True-Up Purchase Price (which cannot be
less than $0.001), and since we cannot deliver any Draw Down Notice to
Magna Equities II, LLC if the Initial Purchase Price would be less than
$0.0025, the Purchase Price for Shares subject to any Draw Down Notice
will never be less than $0.001 per Share. |
43
| (2) |
|
The denominator is
based on 560,564,622 shares outstanding as of November 20, 2014, including
9,109,128 Initial Commitment Shares that were issued to Magna Equities II,
LLC on October 27, 2014 as consideration for its commitment to purchase
our common stock pursuant to the Purchase Agreement. The numerator is
based on the number of Shares issuable to Magna Equities II, LLC under the
Purchase Agreement at the corresponding assumed average purchase price set
forth in the adjacent column. |
| (3) |
|
Assumed average
purchase price is equal to 93% of $0.003925, representing 25% of the
closing sale price of our common stock of $0.01570 on November 10,
2014. |
| (4) |
|
Assumed average
purchase price is equal to 93% of $.00785, representing 50% of the closing
sale price of our common stock of $0.01570 on November 10,
2014. |
| (5) |
|
Assumed average
purchase price is equal to 93% of $0.011775, representing 75% of the
closing sale price of our common stock of $0.01570 on November 10,
2014. |
| (6) |
|
Assumed average
purchase price is equal to 93% of $0.01570, representing the closing sale
price of our common stock on November 10, 2014. |
| (7) |
|
Assumed average
purchase price is equal to 93% of $0.019625, representing 125% of the
closing sale price of our common stock of $.01570 on November 10,
2014. |
| (8) |
|
Assumed average
purchase price is equal to 93% of $0.02355, representing 150% of the
closing sale price of our common stock of $0.01570 on November 10,
2014. |
The following table sets
forth the amount of proceeds we would receive from Magna Equities II, LLC from
the sale of Shares under the Purchase Agreement that are registered in this
offering at varying purchase prices (without accounting for certain fees and
expenses):
|
Number of |
Percentage of Currently |
Proceeds from the Sale of Shares to |
| Assumed
Average |
Registered |
Outstanding |
Magna Equities II, LLC Under the |
| Purchase Price(1) |
Shares to be
Issued |
Shares (2) |
Purchase Agreement |
| $0.00365025 |
(3)
143,812,591 |
20.7% |
$491,702 (9) |
| $0.0073005 |
(4)
143,812,591 |
20.7% |
$983,405 (9) |
| $0.01095075 |
(5)
143,812,591 |
20.7% |
$1,475,107 (9) |
| $0.014601 |
(6)
143,812,591 |
20.7% |
$1,966,810 (9) |
| $0.01825125 |
(7)
143,812,591 |
20.7% |
$2,493,703 (9) |
| $0.0219015 |
(8)143,812,591 |
20.7% |
$2,950,214
(9) |
____________________
| (1) |
|
Because the Purchase
Price for Shares subject to any Draw Down Notice is the lesser of the
Initial Purchase Price and the True-Up Purchase Price (which cannot be
less than $0.001), and since we cannot deliver any Draw Down Notice to
Magna Equities II, LLC if the Initial Purchase Price would be less than
$0.0025, the Purchase Price for Shares subject to any Draw Down Notice
will never be less than $0.001 per Share. |
| (2) |
|
The denominator is
based on 560,564,622 shares outstanding as of November 20, 2014,
134,703,463 shares that would be outstanding (143,812,591 less 9,109,128
shares issued to Magna Equities II, LLC on October 27, 2014). The
numerator is based on the number of Shares issuable to Magna Equities II,
LLC under the Purchase Agreement at the corresponding assumed average
purchase price set forth in the adjacent column. |
| (3) |
|
Assumed average
purchase price is equal to 93% of $0.003925, representing 25% of the
closing sale price of our common stock of $0.01570 on November 10,
2014. |
| (4) |
|
Assumed average
purchase price is equal to 93% of $0.00785, representing 50% of the
closing sale price of our common stock of $0.01570 on November 10,
2014. |
44
| (5) |
|
Assumed average
purchase price is equal to 93% of $0.011775, representing 75% of the
closing sale price of our common stock of $0.01570 on November 10,
2014. |
| (6) |
|
Assumed average
purchase price is equal to 93% of $0.01570, representing the closing sale
price of our common stock on November 10, 2014. |
| (7) |
|
Assumed average
purchase price is equal to 93% of $0.019625, representing 125% of the
closing sale price of our common stock of $0.01570 on November 10,
2014. |
| (8) |
|
Assumed average
purchase price is equal to 93% of $0.02355, representing 150% of the
closing sale price of our common stock of $0.01570 on November 10,
2014. |
| (9) |
|
The proceeds have
been reduced by 9,109,128 shares that were issued to Magna Equities II LLC
on October 27, 2014. |
Magna Equities II, LLC has
agreed that during the term of the Purchase Agreement, neither Magna Equities
II, LLC nor any of its affiliates will, directly or indirectly, engage in any
short sales involving our securities or grant any option to purchase, or acquire
any right to dispose of or otherwise dispose for value of, any shares of our
common stock or any securities convertible into or exercisable or exchangeable
for any shares of our common stock, or enter into any swap, hedge or other
similar agreement that transfers, in whole or in part, the economic risk of
ownership of any shares of our common stock.
The Purchase Agreement
contains customary representations, warranties and covenants by, among and for
the benefit of the parties. Before Magna Equities II, LLC is obligated to
purchase any Shares pursuant to a Draw Down Notice, certain conditions specified
in the Purchase Agreement, none of which are in Magna Equities II, LLC's
control, must be satisfied, including the following:
- Each of our representations and
warranties in the Purchase Agreement must be true and correct in all material
respects.
- We must have performed, satisfied and
complied in all material respects with all covenants, agreements and
conditions required to be performed, satisfied or complied with by us.
- The registration statement of which
this prospectus forms a part must be effective under the Securities
Act.
- We must not have knowledge of any
event that could reasonably be expected to have the effect of causing the
suspension of the effectiveness of the registration statement of which this
prospectus forms a part or the prohibition or suspension of the use of this
prospectus.
- We must have filed with the Commission
all required prospectus supplements relating to this prospectus and all
periodic reports and filings required to be filed by us under the Exchange
Act.
- Trading in our common stock must not
have been suspended by the Commission, the OTCBB or the Financial Industry
Regulatory Authority, or FINRA, there must not have been imposed, and we must
not have received any notice of, any suspension of electronic trading or
settlement services by The Depository Trust Company, and trading in securities
generally on the OTCBB must not have been suspended or limited.
- We must have complied with all
applicable federal, state and local governmental laws, rules, regulations and
ordinances in connection with the execution, delivery and performance of the
Purchase Agreement and the Registration Rights Agreement.
- No statute, regulation, order, decree,
writ, ruling or injunction by any court or governmental authority of competent
jurisdiction shall have been enacted, entered, promulgated, threatened or
endorsed which prohibits the consummation of or which would materially modify
or delay any of the transactions contemplated by the Purchase Agreement and
the Registration Rights Agreement.
- No action, suit or proceeding before
any arbitrator or any court or governmental authority shall have been
commenced or threatened, and no inquiry or investigation by any governmental
authority shall have been commenced or threatened seeking to restrain, prevent
or change the transactions contemplated by the Purchase Agreement or the
Registration Rights Agreement, or seeking material damages in connection with
such transaction.
- The absence of any condition,
occurrence, state of facts or event having, or insofar as reasonably can be
foreseen would likely have, any effect on our business, operations, properties
or financial condition that is material and adverse to us.
45
There is no guarantee that
we will be able to meet the foregoing conditions or any of the other conditions
in the Purchase Agreement or that we will be able to draw down any portion of
the Total Commitment available under the Purchase Agreement with Magna Equities
II, LLC.
The obligations of Magna
Equities II, LLC under the Purchase Agreement to purchase shares of our common
stock may not be transferred to any other party, and none of the terms or
conditions contained in the Purchase Agreement may now be amended or waived by
the parties. The registration statement of which this prospectus is a part will
not cover sales by Magna Equities II, LLC’s transferees, notwithstanding Magna
Equities II, LLC’s right to assign its rights under the Registration Rights
Agreement to its affiliates.
The Purchase Agreement may
be terminated at any time by the mutual written consent of the parties. Unless
earlier terminated, the Purchase Agreement will terminate automatically on the
earliest to occur of (i) the first day of the month next following the 24-month
anniversary of the effective date of the Registration Statement of which this
prospectus is a part, (ii) the date on which Magna Equities II, LLC purchases
the Total Commitment worth of common stock under the Purchase Agreement and
(iii) the date on which our common stock ceases to be listed or quoted on an
eligible trading market under the Purchase Agreement. We may terminate the
Purchase Agreement on one trading day’s prior written notice to Magna Equities
II, LLC, subject to certain conditions, without fee, penalty or cost. Magna
Equities II, LLC may terminate the Purchase Agreement effective upon one trading
day’s prior written notice to us under certain circumstances, including the
following:
- The existence of any condition,
occurrence, state of facts or event having, or insofar as reasonably can be
foreseen would likely have, any effect on our business, operations, properties
or financial condition that is material and adverse to us.
- We enter into an agreement providing
for certain types of financing transactions that is similar to the equity
enhancement program with Magna Equities II, LLC.
- Certain transactions involving a
change in control of the company or the sale of all or substantially all of
our assets have occurred.
- We are in breach or default in any
material respect under any of the provisions of the Purchase Agreement or the
Registration Rights Agreement, and, if such breach or default is capable of
being cured, such breach or default are not cured within 10 trading days after
notice of such breach or default is delivered to us.
- While Magna Equities II, LLC holds any
shares issued under the Purchase Agreement, the effectiveness of the
registration statement that includes this prospectus is suspended or the use
of this Prospectus is suspended or prohibited, and such suspension or
prohibition continues for a period of 20 consecutive trading days or for more
than an aggregate of 60 trading days in any 365-day period, subject to certain
exceptions.
- Trading in our common stock is
suspended and such suspension continues for a period of five consecutive
trading days or for more than an aggregate of 20 trading days in any 365-day
period.
- We have filed for and/or are subject
to any bankruptcy, insolvency, reorganization or liquidation proceedings.
The Purchase Agreement
provides that no termination of the Purchase Agreement will limit, alter,
modify, change or otherwise affect any of the parties' rights or obligations
with respect to any pending Draw Down Notice, and that the parties must fully
perform their respective obligations with respect to any such pending Draw Down
Notice under the Purchase Agreement, provided all of the conditions to the
settlement thereof are timely satisfied.
We paid to Magna Equities
II, LLC a commitment fee for entering into the Purchase Agreement equal to
$150,000 (or 5.0% of the Total Commitment under the Purchase Agreement) in the
form of 9,109,128 Initial Commitment Shares, calculated using a per share price
of $0.016467, representing the arithmetic average of the three lowest daily
VWAPs during the 10-consecutive-trading day period immediately preceding the
Closing Date. In addition, promptly following the effective date of the
registration statement of which this prospectus is a part, we are required to
issue to Magna Equities II, LLC Additional Commitment Shares equal to the
greater of (i) zero and (ii) the difference of (a) the quotient of (x) $150,000
divided by (y) the greater of (1) the arithmetic average of the three lowest
daily VWAPs during the 10-consecutive-trading day period ending on and including
the effective date of the registration statement of which this prospectus is a
part and (2) $0.006, less (ii) 9,109,128, provided that in no event will we
issue more than an aggregate of 15,890,872 shares of our common stock, subject
to adjustment for any stock splits, stock combinations, stock dividends,
recapitalizations and other similar transactions, as Additional Commitment
Shares. The Initial Commitment Shares, together with 15,890,872 Additional
Commitment Shares, are being registered for resale in the registration statement
of which this prospectus is a part.
46
We also agreed to pay up to
$25,000 of reasonable attorneys' fees and expenses (exclusive of disbursements
and out-of-pocket expenses) incurred by Magna Equities II, LLC in connection
with the preparation, negotiation, execution and delivery of the Purchase
Agreement and related transaction documentation. Further, if we issue a Draw
Down Notice and fail to deliver the shares to Magna Equities II, LLC on the
applicable Settlement Date or True-Up Settlement Date, we agreed to pay Magna
Equities II, LLC, in addition to all other remedies available to Magna Equities
II, LLC under the Purchase Agreement, an amount in cash equal to 2.0% of the
purchase price of such shares for each 30-day period the shares are not
delivered, plus accrued interest.
The Purchase Agreement also
provides for indemnification of Magna Equities II, LLC and its affiliates in the
event that Magna Equities II, LLC incurs losses, liabilities, obligations,
claims, contingencies, damages, costs and expenses related to a breach by us of
any of our representations and warranties under the Purchase Agreement or the
other related transaction documents or any action instituted against Magna
Equities II, LLC or its affiliates due to the transactions contemplated by the
Purchase Agreement or other transaction documents, subject to certain
limitations.
The issuances of the
Commitment Shares and the sale of the Shares to Magna Equities II, LLC under the
Purchase Agreement are exempt from registration under the Securities Act
pursuant to the exemption for transactions by an issuer not involving any public
offering under Section 4(a)(2) of and Regulation D under the Securities
Act.
Registration Rights
Agreement
In connection with the
execution of the Purchase Agreement, on the Closing Date, we and Magna Equities
II, LLC also entered into the Registration Rights Agreement. Pursuant to the
Registration Rights Agreement, we agreed to file the registration statement of
which this prospectus is a part with the Commission on or prior to the Filing
Deadline to register for resale 143,812,591 shares of our common stock, which
includes the 9,109,128 Initial Commitment Shares issued on October 27, 2014 and
15,890,872 Additional Commitment Shares, and have it declared effective prior to
the Effectiveness Deadline. The effectiveness of the registration statement of
which this prospectus is a part is a condition precedent to our ability to sell
common stock to Magna Equities II, LLC under the Purchase Agreement.
We have agreed to file with
the Commission one or more additional registration statements to cover all of
the securities required to be registered under the Registration Rights Agreement
that are not covered by this prospectus, in each case, as soon as practicable,
but in no event later than the applicable filing deadline for such additional
registration statements as provided in the Registration Rights Agreement.
We also agreed, among other
things, to indemnify Magna Equities II, LLC from certain liabilities and fees
and expenses of Magna Equities II, LLC incident to our obligations under the
Registration Rights Agreement, including certain liabilities under the
Securities Act. Magna Equities II, LLC has agreed to indemnify and hold harmless
us and each of our directors, officers and persons who control us against
certain liabilities that may be based upon written information furnished by
Magna Equities II, LLC to us for inclusion in the registration statement of
which this prospectus is a part, including certain liabilities under the
Securities Act.
As discussed above, the
obligations of Magna Equities II, LLC under the Purchase Agreement to purchase
shares of our common stock may not be transferred to any other party. Magna
Equities II, LLC may not assign its rights under the Registration Rights
Agreement other than to an affiliate of Magna Equities II, LLC. The registration
statement of which this prospectus is a part will not cover sales by Magna
Equities II, LLC’s transferees, notwithstanding Magna Equities II, LLC’s right
to assign its rights under the Registration Rights Agreement to its affiliates.
None of the terms or conditions contained in the Registration Rights Agreement
may now be amended or waived by the parties.
47
The foregoing description
of the Purchase Agreement and the Registration Rights Agreement does not purport
to be complete and is qualified in its entirety by reference to the full text of
the Purchase Agreement and Registration Rights Agreement, copies of which have
been filed or incorporated by reference as exhibits to the registration
statement of which this prospectus is a part.
PLAN OF
DISTRIBUTION
We are registering shares
of common stock that have been or may be issued by us from time to time to Magna
Equities II, LLC under the Purchase Agreement and upon conversion of the
Convertible Note to permit the resale of these shares of common stock after the
issuance thereof by the selling stockholder from time to time after the date of
this prospectus. We will not receive any of the proceeds from the sale by the
selling stockholder of the shares of common stock. We will bear all fees and
expenses incident to our obligation to register the shares of common stock.
The selling stockholder may
decide not to sell any shares of common stock. The selling stockholder may sell
all or a portion of the shares of common stock beneficially owned by it and
offered hereby from time to time directly or through one or more underwriters,
broker-dealers or agents, who may receive compensation in the form of discounts,
concessions or commissions from the selling stockholder and/or the purchasers of
the shares of common stock for whom they may act as agent. In effecting sales,
broker-dealers that are engaged by the selling stockholder may arrange for other
broker-dealers to participate. With respect to the shares of common stock that
have been and may be issued pursuant to the Purchase Agreement, Magna Equities
II, LLC is an “underwriter” within the meaning of Section 2(a) (11) of the
Securities Act, and with respect to any other shares of common stock, Magna
Equities II, LLC may be deemed to be an “underwriter” within the meaning of
Section 2(a) (11) of the Securities Act. Any brokers, dealers or agents who
participate in the distribution of the shares of common stock by the selling
stockholder may also be deemed to be “underwriters,” and any profits on the sale
of the shares of common stock by them and any discounts, commissions or
concessions received by any such brokers, dealers or agents may be deemed to be
underwriting discounts and commissions under the Securities Act. Magna Equities
II, LLC has advised us that it will use an unaffiliated broker-dealer to
effectuate all resales of our common stock. To our knowledge, Magna Equities II,
LLC has not entered into any agreement, arrangement or understanding with any
particular broker-dealer or market maker with respect to the shares of neither
common stock offered hereby, nor do we know the identity of the broker-dealers
or market makers that may participate in the resale of the shares. Because Magna
Equities II, LLC is (with respect to shares of common stock issued under the
Purchase Agreement) and may be deemed to be (with respect to any other shares of
common stock), and any other selling stockholder, broker, dealer or agent may be
deemed to be, an “underwriter” within the meaning of Section 2(a)(11) of the
Securities Act, Magna Equities II, LLC will (and any other selling stockholder,
broker, dealer or agent may) be subject to the prospectus delivery requirements
of the Securities Act and may be subject to certain statutory liabilities of the
Securities Act (including, without limitation, Sections 11, 12 and 17 thereof)
and Rule 10b-5 under the Exchange Act.
The selling stockholder
will act independently of us in making decisions with respect to the timing,
manner and size of each sale. The shares of common stock may be sold in one or
more transactions at fixed prices, at prevailing market prices at the time of
the sale, at varying prices determined at the time of sale, or at negotiated
prices. These sales may be effected in transactions, which may involve crosses
or block transactions, pursuant to one or more of the following methods:
- on any national securities exchange or
quotation service on which the securities may be listed or quoted at the time
of sale;
- in the over-the-counter market in
accordance with the rules of NASDAQ;
- in transactions otherwise than on
these exchanges or systems or in the over-the-counter market;
- through the writing or settlement of
options, whether such options are listed on an options exchange or
otherwise;
- ordinary brokerage transactions and
transactions in which the broker-dealer solicits purchasers;
48
- block trades in which the
broker-dealer will attempt to sell the shares as agent but may position and
resell a portion of the block as principal to facilitate the
transaction;
- purchases by a broker-dealer as
principal and resale by the broker-dealer for its account;
- an exchange distribution in accordance
with the rules of the applicable exchange;
- privately negotiated
transactions;
- broker-dealers may agree with the
selling stockholder to sell a specified number of such shares at a stipulated
price per share;
- a combination of any such methods of
sale; and
- any other method permitted pursuant to
applicable law.
In addition, the selling
stockholder may transfer the shares of common stock by other means not described
in this prospectus.
Any broker-dealer
participating in such transactions as agent may receive commissions from the
selling stockholder (and, if they act as agent for the purchaser of such shares,
from such purchaser). Magna Equities II, LLC has informed us that each such
broker-dealer will receive commissions from Magna Equities II, LLC which will
not exceed customary brokerage commissions. Broker-dealers may agree with the
selling stockholder to sell a specified number of shares at a stipulated price
per share, and, to the extent such a broker-dealer is unable to do so acting as
agent for the selling stockholder, to purchase as principal any unsold shares at
the price required to fulfill the broker-dealer commitment to the selling
stockholder. Broker-dealers who acquire shares as principal may thereafter
resell such shares from time to time in one or more transactions (which may
involve crosses and block transactions and which may involve sales to and
through other broker-dealers, including transactions of the nature described
above and pursuant to the one or more of the methods described above) at fixed
prices, at prevailing market prices at the time of the sale, at varying prices
determined at the time of sale, or at negotiated prices, and in connection with
such resales may pay to or receive from the purchasers of such shares
commissions computed as described above. To the extent required under the
Securities Act, an amendment to this prospectus or a supplemental prospectus
will be filed, disclosing:
- the name of any such
broker-dealers;
- the number of shares involved;
- the price at which such shares are to
be sold;
- the commission paid or discounts or
concessions allowed to such broker-dealers, where applicable;
- that such broker-dealers did not
conduct any investigation to verify the information set out or incorporated by
reference in this prospectus, as supplemented; and
- other facts material to the
transaction.
Magna Equities II, LLC has
informed us that it does not have any written or oral agreement or
understanding, directly or indirectly, with any person to distribute the common
stock.
Under the securities laws
of some states, the shares of common stock may be sold in such states only
through registered or licensed brokers or dealers. In addition, in some states
the shares of common stock may not be sold unless such shares have been
registered or qualified for sale in such state or an exemption from registration
or qualification is available and is complied with.
There can be no assurance
that the selling stockholder will sell any or all of the shares of common stock
registered pursuant to the registration statement, of which this prospectus
forms a part.
49
Underwriters and purchasers
that are deemed underwriters under the Securities Act may engage in transactions
that stabilize maintain or otherwise affect the price of the common stock,
including the entry of stabilizing bids or syndicate covering transactions or
the imposition of penalty bids. The selling stockholder and any other person
participating in the sale or distribution of the shares of common stock will be
subject to applicable provisions of the Exchange Act and the rules and
regulations thereunder (including, without limitation, Regulation M of the
Exchange Act), which may restrict certain activities of, and limit the timing of
purchases and sales of any of the shares of common stock by, the selling
stockholder and any other participating person. To the extent applicable,
Regulation M may also restrict the ability of any person engaged in the
distribution of the shares of common stock to engage in market-making and
certain other activities with respect to the shares of common stock. In
addition, the anti-manipulation rules under the Exchange Act may apply to sales
of the shares of common stock in the market. All of the foregoing may affect the
marketability of the shares of common stock and the ability of any person or
entity to engage in market-making activities with respect to the shares of
common stock.
We have agreed to pay all
expenses of the registration of the shares of common stock pursuant to the
registration rights agreement, estimated to be $22,269.05 in total, including,
without limitation, Securities and Exchange Commission filing fees and expenses
of compliance with state securities or “Blue Sky” laws; provided, however, Magna
Equities II, LLC will pay all selling commissions, concessions and discounts,
and other amounts payable to underwriters, dealers or agents, if any, as well as
transfer taxes and certain other expenses associated with the sale of the shares
of common stock. We have agreed to indemnify Magna Equities II, LLC and certain
other persons against certain liabilities in connection with the offering of
shares of common stock offered hereby, including liabilities arising under the
Securities Act or, if such indemnity is unavailable, to contribute amounts
required to be paid in respect of such liabilities. Magna Equities II, LLC has
agreed to indemnify us against liabilities under the Securities Act that may
arise from any written information furnished to us by Magna Equities II, LLC
specifically for use in this prospectus or, if such indemnity is unavailable, to
contribute amounts required to be paid in respect of such liabilities.
At any time a particular
offer of the shares of common stock is made by the selling stockholder, a
revised prospectus or prospectus supplement, if required, will be distributed.
Such prospectus supplement or post-effective amendment will be filed with the
Commission to reflect the disclosure of any required additional information with
respect to the distribution of the shares of common stock. We may suspend the
sale of shares by the selling stockholder pursuant to this prospectus for
certain periods of time for certain reasons, including if the prospectus is
required to be supplemented or amended to include additional material
information.
DESCRIPTION OF
BUSINESS
Overview
We are a biotechnology
company focused on the discovery, development and, subject to regulatory
approval, commercialization of autologous cell therapies for the treatment of
chronic and acute heart damage and the discovery and development of autologous
cellular therapies to treat a variety of degenerative diseases. Our lead product
candidate is MyoCell, an innovative clinical therapy designed to populate
regions of scar tissue within a patient’s heart with autologous muscle cells, or
cells from a patient’s body, for the purpose of improving cardiac function in
chronic heart failure patients.
We were incorporated in the
State of Florida in August 1999. Our principal executive offices are located at
13794 NW 4th Street, Suite 212, Sunrise, Florida 33325 and our telephone number
is (954) 835-1500. Information about us is available on our corporate web site
at www.bioheartinc.com. Information contained on the web site does not
constitute part of, and is not incorporated by reference in, this report.
Biotechnology Product
Candidates
Specific to biotechnology,
we are focused on the discovery, development and, subject to regulatory
approval, commercialization of autologous cell therapies for the treatment of
chronic and acute heart damage as well as degenerative diseases. In our
pipeline, we have multiple product candidates for the treatment of heart damage,
including MyoCell, Myocell SDF-1, and Adipocell. MyoCell and MyoCell SDF-1 are
clinical muscle-derived cell therapies designed to populate regions of scar
tissue within a patient’s heart with new living cells for the purpose of
improving cardiac function in chronic heart failure patients.
50
MyoCell SDF-1 is intended
to be an improvement to MyoCell. MyoCell SDF-1 is similar to MyoCell except that
the myoblast cells to be injected for use in MyoCell SDF-1 will be modified
prior to injection by an adenovirus vector or non-viral vector so that they will
release extra quantities of the SDF-1 protein, which expresses angiogenic
factors. AdipoCell is a patient-derived cell therapy proposed for the treatment
of acute myocardial infarction, chronic heart ischemia, and lower limb ischemia.
We hope to demonstrate that these product candidates are safe and effective
complements to existing therapies for chronic and acute heart damage.
We have completed various
clinical trials for MyoCell including the SEISMIC Trial, a 40-patient,
randomized, multicenter, controlled, Phase II-a study conducted in Europe and
the MYOHEART Trial, a 20-patient, multicenter, Phase I dose-escalation trial
conducted in the United States. We were approved by the U.S. Food and Drug
Administration, or the “FDA”, to proceed with a 330-patient, multicenter Phase
II/III trial of MyoCell in North America and Europe, or the MARVEL Trial. We
completed the MyoCell implantation procedure on the first patient in the MARVEL
Trial on October 24, 2007. Thus far, 20 patients, including 6 control patients,
have been treated. Initial results for the 20 patients were released at the
Heart Failure Society of American meeting in September, 2009, showing a
significant (35%) improvement in the 6 minute walk for those patients who were
treated, and no improvement for those who received a placebo. On the basis of
these results, we have applied for and received approval from the FDA to reduce
the number of additional patients in the trial to 134, for a total of 154
patients.
We have also initiated the
MIRROR trial, which is a Phase III, double-blind placebo controlled study for
centers outside the US. The SEISMIC, MYOHEART, MARVEL and MIRROR Trials have
been designed to test the safety and efficacy of MyoCell in treating patients
with severe, chronic damage to the heart. Upon regulatory approval of MyoCell,
we intend to generate revenue in the United States from the sale of MyoCell
cell-culturing services for treatment of patients by qualified physicians.
We received approval from
the FDA in July of 2009 to conduct a Phase I safety study on 15 patients of a
combined therapy (Myocell with SDF-1), which we believe was the first approval
of a study combining gene and cell therapies. We initially commenced work on
this study, called the REGEN Trial, during the first quarter of 2010. We
suspended activity on the trial in 2010 while seeking additional funding
necessary to conduct the trial.
We are seeking to secure
sufficient funds to reinitiate enrollment in the MARVEL and REGEN trials. If we
successfully secure such funds, we intend to re-engage a contract research
organization, or CRO, investigators and certain suppliers to advance such
trials.
We have also initiated
several Institutional Review Board studies in 2013 using adipose derived stem
cells for various indications including dry macular degeneration, degenerative
disc disease, erectile dysfunction and chronic obstructive pulmonary
disease.
In the second quarter of
2014, we announced the treatment of a patient in Honduras with congestive heart
failure using AdipoCell and MyoCell. This was the first patient treated in the
world using a combination of stem cells.
We have begun two clinical
trials in India. The first cardiac patient has successfully been enrolled and
treated in India using AdipoCell or adipose derived stem cells. The second trial
will involve the combination of AdipoCellTM and MyoCell® or muscle
derived stem cells for congestive heart failure patients. These trials are
active and ongoing We have also initiated several Institutional Review Board
studies using adipose derived stem cells for various indications including dry
macular degeneration, degenerative disc disease, erectile dysfunction and
chronic obstructive pulmonary disease.
51
MyoCell
MyoCell is a clinical
therapy intended to improve cardiac function for those with congestive heart
failure and is designed to be utilized months or even years after a patient has
suffered severe heart damage due to a heart attack or other cause. We believe
that MyoCell has the potential to become a leading treatment for severe, chronic
damage to the heart due to its perceived ability to satisfy, at least in part,
what we believe to be an unmet demand for more effective and/or more affordable
therapies for chronic heart damage. MyoCell uses myoblasts, cells that are
precursors to muscle cells, from the patient’s own body. The myoblasts are
removed from a patient’s thigh muscle, isolated, grown through our proprietary
cell culturing process, and injected directly in the scar tissue of a patient’s
heart. A qualified physician performs this minimally invasive procedure using an
endoventricular catheter. We entered into an agreement with a Johnson &
Johnson company to use its NOGA® Cardiac Navigation System along with its
MyoStar™ injection catheter for the delivery of MyoCell in the MARVEL Trial.
These cells can also be delivered with our MyoCath catheter.
When injected into scar
tissue within the heart wall, myoblasts have been shown to be capable of
engrafting in the damaged tissue and differentiating into mature skeletal muscle
cells. In a number of clinical and animal studies, the engrafted skeletal muscle
cells have been shown to express various proteins that are important components
of contractile function. By using myoblasts obtained from a patient’s own body,
we believe MyoCell is able to avoid certain challenges currently faced by other
types of cell-based clinical therapies including tissue rejection and instances
of the cells differentiating into cells other than muscle. Although a number of
therapies have proven to improve the cardiac function of a damaged heart, no
currently available treatment, to our knowledge, has demonstrated an ability to
generate new muscle tissue within the scarred regions of a heart.
Our completed clinical
trials of MyoCell to date, l have been primarily targeted to patients with
severe, chronic damage to the heart who are in Class II or Class III heart
failure according to the New York Heart Association, or NYHA, heart failure
classification system. The NYHA system classifies patients in one of four
categories based on how limited they are during physical activity. NYHA Class II
heart failure patients have a mild limitation of activity and are generally
comfortable at rest or with mild exertion while NYHA Class III heart failure
patients suffer from a marked limitation of activity and are generally
comfortable only at rest.
In addition to studies we
have sponsored, we understand that myoblast-based clinical therapies have been
the subject of at least eleven clinical trials involving more than 325
enrollees, including at least 235 treated patients. Although we believe many of
the trials are different from the trials sponsored by us in a number of
important respects, it is our view that the trials have advanced the cell
therapy industry’s understanding of the potential opportunities and limitations
of myoblast-based therapies.
We believe the market for
treating patients in NYHA Class II or NYHA Class III heart failure is
significant. According to the AHA Statistics and the European Society of
Cardiology Task Force for the Treatment of Chronic Heart Failure, in the United
States and Europe there are approximately 5.2 million and 9.6 million,
respectively, patients with heart failure. The AHA Statistics further indicate
that, after heart failure is diagnosed, the one-year mortality rate is high,
with one in five dying and that 80% of men and 70% of women under age 65 who
have heart failure will die within eight years.
We believe that
approximately 60% of heart failure patients are in either NYHA Class II or NYHA
Class III heart failure based upon a 1999 study entitled “Congestive Heart
Failure Due to Diastolic or Systolic Dysfunction – Frequency and Patient
Characteristics in an Ambulatory Setting” by Diller, PM, et. al.
MyoCath Product
Candidate
The MyoCath is a deflecting
tip needle injection catheter that has a larger (25 gauge) needle to allow for
better flow rates and less leakage than systems that are 27 gauge. This larger
needle allows for thicker compositions to be injected, which helps with cell
retention in the heart. Also, the MyoCath needle has more fluoroscopic
brightness than the normally used nitinol needle, enabling superior
visualization during the procedure. Seeing the needle well during injections
enables the physician who is operating the catheter to pinpoint targeted areas
more precisely. The MyoCath is used to inject cells into cardiac tissue in
therapeutic procedures to treat chronic heart ischemia and congestive heart
failure. Investigators in our MARVEL Trial may use either our MyoCath catheters
or Biosense Webster’s (a Johnson & Johnson company) NOGA® Cardiac Navigation
System along with the MyoStarTM injection catheter for the delivery of MyoCell
to patients enrolled in the trial. We are currently producing Myocath catheters
with a contract manufacturer on an as needed basis.
52
AdipoCell
Bioheart has successfully
completed various trials using adipose stem cells. In August 2013, the Company
canceled its license agreement with the Ageless Regenerative Institute for
adipose derived stem cells called LipiCell. Bioheart has entered into a term
sheet agreement with Invitrx to License their adipose derived stem cell
products. Bioheart has changed its adipose derived stem cell product name to
AdipoCell.
Bioheart has funded and
completed enrollment of 5 patients in the Angel Trial in Mexico utilizing
adipose derived cells. At the 3 month time point, patients are demonstrating an
average improvement in exercise capacity or a six minute walk test of 47 meters.
In addition, 60% percent or a majority of the patients are walking greater than
65 meters further at 3 months post stem cell injection. At the twelve (12) month
time point, patients demonstrated a statistically significant average
improvement in ejection fraction. The patients are also reporting an average
improvement of 13 points in their Minnesota Living with Heart Failure
questionnaire. An improvement of 5 points or greater is considered “clinically
meaningful” and 80% of the patients in the trial had a greater than 5 point
improvement.
At the three (3) month time
point, 100% of the patients demonstrated either improvement or stayed the same.
After three (3) months, patients showed an average absolute improvement of 3
percentage points in EF. The patients continued to improve from 3 months to 6
months with a statistically significant average absolute improvement of 10
percentage points (p=0.01) and at the 12 month follow up patients showed this
same level of improvement (p=0.01).
Business
Strategy
Our principal objective is
to become a leading regenerative medicine company that discovers, develops and
commercializes novel, autologous cell therapies, and related devices, for the
treatment and improved care of patients suffering from chronic and acute heart
damage as well as lower limb ischemia. Our secondary objective is the discovery,
development and commercialization of autologous cell therapies for the treatment
of degenerative diseases. The number of heart failure patients is expected to
increase from 25 million worldwide today to over 50 million in five years. Our
focus is on serving these patients. To achieve our primary objective, we plan to
pursue the following strategies:
Obtain initial
regulatory approval of MyoCell and/or MyoCell SDF-1 by targeting patients with
severe heart damage. In July
2007, we treated the final patient in the Phase II SEISMIC Trial, which was
comprised of 40 patients, including 26 treated patients. The SEISMIC study
results demonstrated that 94% of MyoCell treated patients improved or did not
worsen in heart failure class while only 6% worsened, while in the control group
receiving only drugs 42% worsened. 84% of MyoCell treated patients improved or
did not worsen in exercise capacity and only 16% worsened, while 69% of the
control patients worsened. The average improvement in 6 minute walk was 62
meters. This compares very favorably with the current gold standard in advanced
heart failure treatment, Bi-Ventricular pacing, where they achieved 16 to 20
meters improvement over control patients in the Phase II MIRACLE trial that led
to commercial approval of this product. By targeting a class of patients for
whom existing therapies are very expensive, unavailable or not sufficiently
effective, we hope to expedite regulatory approval of MyoCell and/or MyoCell
SDF-1. Obtain regulatory approval
of MyoCell and/or MyoCell SDF-1 to treat patients with less severe heart damage.
If we obtain initial regulatory
approval of MyoCell for the Class III Subgroup, we intend to continue to sponsor
clinical trials in an effort to demonstrate that MyoCell and/or MyoCell SDF-1
should receive regulatory approval to treat all patients in NYHA Class II, NYHA
Class III and NYHA Class IV heart failure and, provided we believe we have a
reasonable basis to support such an indication, we intend to seek regulatory
approval for these patients.
53
Continue existing
studies with adipose derived stem cells and endothelial progenitor cells. We have initiated studies for the
applications of lower limb ischemia, acute myocardial infarction
and chronic heart ischemia. We have treated patients
in the clinical program called the ANGEL trial utilizing adipose derived cells
or AdipoCell for congestive heart failure patients. Using the clinical data from
the trial in Mexico and preclinical studies, Bioheart plans to apply to the FDA
to begin a Phase I study in the US. Bioheart has completed several preclinical
studies demonstrating the safety and efficacy of this product including a study
led by Keith March, MD, PhD, Director of the Vascular and Cardiac Center for
Adult Stem Cell Therapy at the Indiana University. The adipose cells showed a
tendency toward cardiomyocyte regeneration, prominent angiogenesis (growth of
new blood vessels) and reduction in the infarction size. In addition, Bioheart
has several studies for the use of adipose derived stem cells.
Continue to develop our
pipeline of cell-based therapies and related devices for the treatment of
chronic and acute heart damage. In parallel with our efforts to secure regulatory approval of MyoCell, we
intend to continue to develop and test other product candidates for the
treatment of chronic and acute heart damage. These efforts are expected to
initially focus on MyoCell SDF-1, AdipoCell, MyoCath and MyoCath II product
candidates.
Develop our sales and
marketing capabilities. In
advance of U.S. regulatory approval of our MyoCell product candidate, we intend
to internally build a sales force to cover the U.S. market and to utilize
dealers in foreign markets which we anticipate will market MyoCell, MyoCell
SDF-1 and our heart failure focused products primarily to interventional
cardiologists and heart failure specialists. We are currently marketing our
adipose derived stem cell therapy product in the U.S. and foreign
markets.
Continue to refine our
MyoCell and MyoCell SDF-1 cell culturing processes.
License, acquire and/or
develop complementary products and technologies. We intend to strengthen and expand our product
development efforts through the license, acquisition and/or development of
products and technologies that support our business strategy.
Industry Background
Myocardial Infarction
(Heart Attack)
Myocardial infarction, or
MI, commonly known as a heart attack, occurs when a blockage in a coronary
artery severely restricts or completely stops blood flow to a portion of the
heart. When blood supply is greatly reduced or blocked for more than a short
period of time, heart muscle cells die. If the healthy heart muscle cells do not
replace the dead cells within approximately two months, the injured area of the
heart becomes unable to function properly. In the healing phase after a heart
attack, white blood cells migrate into the affected area and remove the dead
heart muscle cells. Then, fibroblasts, the connective tissue cells of the human
body, proliferate and form a collagen scar in the affected region of the heart.
Following a heart attack, the heart's ability to maintain normal function will
depend on the location and amount of damaged tissue. The remaining initially
undamaged heart muscle tissue must perform more work to adequately maintain
cardiac output. Because the uninjured region is then compelled to work harder
than normal, the heart can progressively deteriorate until it is unable to pump
adequate blood to oxygenate the body properly leading to heart failure and
ultimately death.
Congestive Heart
Failure (CHF)
Congestive heart failure,
or “CHF”, is a debilitating condition that occurs as the heart becomes
progressively less able to pump an adequate supply of blood throughout the body
resulting in fluid accumulation in the lungs, kidneys and other body tissues.
Persons suffering from NYHA Class II or worse heart failure experience high
rates of mortality, frequent hospitalization and poor quality of life. CHF has
many causes, generally beginning in patients with a life-long history of high
blood pressure or after a patient has suffered a major heart attack or some
other heart-damaging event. CHF itself may lead to other complicating factors
such as pulmonary hypertension, edema, pulmonary edema, liver dysfunction and
kidney failure. Although medical therapy for CHF is improving, it remains a
major debilitating condition.
54
Classifying Heart
Failure
The NYHA heart failure
classification system provides a simple and widely recognized way of classifying
the extent of heart failure. It places patients in one of four categories based
on how limited they are during physical activity. NYHA Class I heart failure
patients have no limitation of activities and suffer no symptoms from ordinary
activities. NYHA Class II heart failure patients have a mild limitation of
activity and are generally comfortable at rest or with mild exertion. NYHA Class
III heart failure patients suffer from a marked limitation of activity and are
generally comfortable only at rest. NYHA Class IV heart failure patients
generally suffer discomfort and symptoms at rest and should remain confined to a
bed or chair.
The risk of hospitalization
and death increases as patient’s progress through the various stages of heart
failure. The risk of hospitalization due to heart failure for patients in NYHA
Class II, NYHA Class III and NYHA Class IV is approximately 1.2, 2.3 and 3.7
times greater than for patients in NYHA Class I heart failure according to a
2006 American Heart Journal article entitled “Higher New York Heart Association
Classes and Increased Mortality and Hospitalization in Patients with Heart
Failure and Preserved Left Ventricular Function”' by Ahmed, A et al. Similarly,
according to this same article, the risk of death from all causes for patients
in NYHA Class II, NYHA Class III and NYHA Class IV is approximately 1.5, 2.6 and
8.5 times greater than for patients in NYHA Class I heart failure.
The following chart
illustrates the various stages of heart failure, their NYHA classifications and
the associated current standard of treatment.
| NYHA |
|
|
|
| Class |
NYHA Functional
Classification(1) |
Specific Activity
Scale(2)(3) |
Current Standard of
Treatment(4) |
| I |
Symptoms only with above
normal physical activity |
Can perform more than 7
metabolic equivalents |
ACE Inhibitor,
Beta-Blocker |
| II |
Symptoms with normal
physical activity |
Can perform more than 5 metabolic
equivalents |
ACE Inhibitor,
Beta-Blocker, Diuretics |
III |
Symptoms with minimal
physical activity |
Can perform more than 2
metabolic equivalents |
ACE Inhibitor,
Beta-Blocker, Diuretics, Digoxin, Bi-ventricular pacers |
| IV |
Symptoms at rest
|
Cannot perform more than
2 metabolic equivalents |
ACE Inhibitor,
Beta-Blocker, Diuretics, Digoxin, Hemodynamic Support, Mechanical Assist
Devices, Bi-ventricular pacers, Transplant |
____________________
Symptoms include fatigue,
palpitations, shortness of breath and chest pain; normal activity is equivalent
to walking one flight of stairs or several blocks.
Based upon the Goldman
Activity Classification of Heart Failure, which classifies severity of heart
failure based on estimated metabolic cost of various activities; the four
classes of the Goldman Activity Classification system correlate to the NYHA
Classes.
7 metabolic equivalents =
shovel snow, carry 24 lbs. up 8 stairs, recreational sports; 5 metabolic
equivalents = garden, rake, dance, walk 4 mph on level ground, have intercourse;
2 metabolic equivalents = shower without stopping, strip and make bed, dress
without stopping.
Source: American College of
Cardiology/ American Heart Association 2005 Guideline Update for the Diagnosis
and Management of Chronic Heart Failure in the Adult.
55
Diagnosis and
Management of Heart Failure
Heart disease has been the
leading cause of death from 1950 on within the United States, according to the
U.S. Department of Health and Human Services. In addition, heart failure is the
single most frequent reason for hospitalization in the elderly according to a
2007 study entitled “Long-Term Costs and Resource Use in Elderly Participants
with Congestive Heart Failure” by Liao, L., et al. The American College of
Cardiology/ American Heart Association 2005 Guideline Update for the Diagnosis
and Management of Chronic Heart Failure in the Adult, or the ACC/ AHA
Guidelines, provides recommendations for the treatment of chronic heart failure
in adults with normal or low LVEF. The treatment escalates and becomes more
invasive as the heart failure worsens. Current treatment options for severe,
chronic heart damage include, but are not limited to, heart transplantation and
other surgical procedures, bi-ventricular pacers, drug therapies, ICDs, and
ventricular assist devices. Therapies utilizing drugs, ICDs and bi-ventricular
pacers are currently by far the most commonly prescribed treatments for patients
suffering from NYHA Class II or NYHA Class III heart failure. Since the
therapies generally each address a particular feature of heart disease or a
specific subgroup of heart failure patients, the therapies are often
complementary and used in combination.
Drug Therapies
The ACC/AHA Guidelines
recommend that most patients with heart failure should be routinely managed with
a combination of ACE inhibitors, beta-blockers and diuretics. The value of these
drugs has been established by the results of numerous large-scale clinical
trials and the evidence supporting a central role for their use is, according to
the ACC/AHA Guidelines, compelling and persuasive. ACE inhibitors and beta
blockers have been shown to improve a patient’s clinical status and overall
sense of well-being and reduce the risk of death and hospitalization. Side
effects of ACE inhibitors include hypotension, worsening kidney function,
potassium retention, cough and angioedema. Side effects of beta-blockers include
fluid retention, fatigue, bradycardia and heart block and hypotension.
Bi-Ventricular Pacers
The ACC/AHA Guidelines
recommend bi-ventricular pacers for persons who, in addition to suffering from
heart failure, have left and right ventricles that do not contract in sync,
known as ventricular dyssynchrony and who have a LVEF less than or equal to 35%,
sinus rhythm and NYHA Class III or NYHA Class IV symptoms despite recommended
optimal medical therapy. Bi-ventricular pacers are surgically implanted
electrical generators that function primarily by stimulating the un-damaged
portion of the heart to beat more strongly using controlled bursts of electrical
currents in synchrony. Compared with optimal medical therapy alone,
bi-ventricular pacers have been shown in a number of clinical trials to
significantly decrease the risk of all-cause hospitalization and all-cause
mortality as well as to improve LVEF, NYHA Class and Quality of Life. According
to the ACC/AHA Guidelines, there are certain risks associated with the
bi-ventricular pacer including risks associated with implantation and
device-related problems.
Implantable Cardioverter
Defibrillators
ACC/AHA Guidelines
recommend ICDs primarily for patients who have experienced a life-threatening
clinical event associated with a sustained irregular heartbeat and in patients
who have had a prior heart attack and a reduced LVEF. ICDs are surgically
implanted devices that continually monitor patients at high risk of sudden heart
attack. When an irregular rhythm is detected, the device sends an electric shock
to the heart to restore normal rhythm. In 2001, ICDs were implanted in
approximately 62,000 and 18,000 patients in the United States and Europe,
respectively. Although ICDs have not demonstrated an ability to improve cardiac
function, according to the ACC/AHA Guidelines, ICDs are highly effective in
preventing sudden death due to irregular heartbeats. However, according to the
ACC/AHA Guidelines, frequent shocks from an ICD can lead to a reduced quality of
life, whether triggered appropriately or inappropriately. In addition, according
to the ACC/AHA Guidelines, ICDs have the potential to aggravate heart failure
and have been associated with an increase in heart failure
hospitalizations.
56
Heart Transplantation
and Other Surgical Procedures
According to the ACC/AHA
Guidelines, heart transplantation is currently the only established surgical
approach for the treatment of severe heart failure that is not responsive to
other therapies. Heart transplantation is a major surgical procedure in which
the diseased heart is removed from a patient and replaced with a healthy donor
heart. Heart transplantation has proven to dramatically improve cardiac function
in a majority of the patients treated and most heart transplant recipients
return to work, travel and normal activities within three to six months after
the surgery. In addition, the risk of hospitalization and mortality for
transplant recipients is dramatically lower than the risk faced by patients in
NYHA Class III or NYHA Class IV heart failure. Heart transplants are not, for a
variety of reasons, readily available to all patients with severe heart damage.
The availability of heart transplants is limited by, among other things, cost
and donor availability. In addition to the significant cost involved and the
chronic shortage of donor hearts, one of the serious challenges in heart
transplantation is potential rejection of the donor heart. For many heart
transplant recipients, chronic rejection significantly shortens the length of
time the donated heart can function effectively and such recipients are
generally administered costly anti-rejection drug regimens which can have
adverse and potentially severe side effects.
There are a number of
alternate surgical approaches under development for the treatment of severe
heart failure, including cardiomyoplasty, a surgical procedure where the
patient’s own body muscle is wrapped around the heart to provide support for the
failing heart, the Batista procedure, a surgical procedure that reduces the size
of an enlarged heart muscle so that the heart can pump more efficiently and
vigorously, and the Dor procedure. According to the ACC/AHA Guidelines, both
cardiomyoplasty and the Batista procedure have failed to result in clinical
improvement and are associated with a high risk of death. The Dor procedure
involves surgically removing scarred, dead tissue from the heart following a
heart attack and returning the left ventricle to a more normal shape. While the
early published single-center experience with the Dor procedure demonstrated
early and late improvement in NYHA Class and LVEF, according to the ACC/AHA
Guidelines, this procedure’s role in the management of heart failure remains to
be defined.
We believe the heart
failure treatment industry generally has a history of adopting therapies that
have proven to be safe and effective complements to existing therapies and using
them in combination with existing therapies. It is our understanding that there
is no one or two measurement criteria, either quantitative or qualitative, that
define when a therapy for treating heart failure will be deemed safe and
effective by the FDA. We believe that the safety and efficacy of certain
existing FDA approved therapies for heart damage were demonstrated based upon a
variety of endpoints, including certain endpoints (such as LVEF) that
individually did not demonstrate large numerical differences between the treated
patients and untreated patients. For instance, the use of bi-ventricular pacers
with optimal drug therapy has proven to significantly decrease the risk of
all-cause hospitalization and all-cause-mortality as well as to improve LVEF,
NYHA Class and quality of life as compared to the use of optimal drug therapy
alone. In the Multicenter InSync Randomized Clinical Evaluation (MIRACLE) trial,
one of the first large studies to measure the therapeutic benefits of
bi-ventricular pacing, 69% of the patients in the treatment group experienced an
improvement in NYHA Class by one or more classes at six-month follow-up versus a
34% improvement in the control group. However, patients in the treatment group
experienced on average only a 2.1% improvement in LVEF as compared with a 1.7%
improvement for patients in the control group. Although a number of the
therapies described above have proven to improve the cardiac function of a
damaged heart, no currently available heart failure treatment has demonstrated
an ability to generate new muscle tissue within the scarred regions of a
heart.
Our Proposed
Solution
We believe that MyoCell has
the potential to become a leading treatment for severe chronic damage to the
heart due to its perceived ability to satisfy, at least in part, what we believe
to be a presently unmet demand for more effective and/or more affordable
therapies for chronic heart damage.
MyoCell
The human heart does not
have cells that naturally repair or replace damaged heart muscle. Accordingly,
the human body cannot, without medical assistance, repopulate regions of scar
tissue within the heart with functioning muscle. MyoCell is a clinical therapy
designed to improve cardiac function by populating regions of scar tissue within
a patient’s heart with myoblasts derived from a biopsy of a patient’s thigh
muscle. Myoblasts are precursors to muscle cells that have the capacity to fuse
with other myoblasts or with damaged muscle fibers to regenerate skeletal
muscle. When injected into scar tissue within the heart wall, myoblasts have
been shown to be capable of engrafting in the damaged tissue and differentiating
into mature skeletal muscle cells. In a number of clinical and animal studies,
the engrafted skeletal muscle cells have been shown to express various proteins
that are important components of contractile function. By using myoblasts
obtained from a patient’s own body, we believe MyoCell is able to avoid certain
challenges currently faced by other cell-based clinical therapies intended to be
used for the treatment of chronic heart damage including tissue rejection and
instances of the cells differentiating into cells other than muscle.
57
Our clinical research to
date suggests that MyoCell may improve the contractile function of the heart.
However, we have not yet been able to demonstrate a mechanism of action. The
engrafted skeletal muscle tissues are not believed to be coupled with the
surrounding heart muscle by the same chemicals that allow heart muscle cells to
contract simultaneously. The theories regarding why contractile function may
improve include:
- the engrafted muscle tissue can
contract in unison with the other muscles in the heart by stretching or by the
channeling of electric currents;
- the myoblasts acquire certain
characteristics of heart muscle or fuse with them; and/or
- the injected myoblasts release various
proteins that indirectly result in a limit on further scar tissue
formation.
As part of the MyoCell
therapy, a general surgeon removes approximately five to ten grams of thigh
muscle tissue from the patient utilizing local anesthesia, typically on an
outpatient basis. The muscle tissue is then express-shipped to a cell culturing
site. At the cell culturing site, our proprietary techniques are used to isolate
and remove myoblasts from the muscle tissue. We typically produce enough cells
to treat a patient within approximately 21 days of his or her biopsy. Such
production time is expected to continue to decrease as we continue to refine our
cell culturing processes. After the cells are subjected to a variety of tests,
the cultured cells are packaged in injectate media and express shipped to the
interventional cardiologist. Within four days of packaging, the cultured
myoblasts are injected via catheter directly into the scar tissue of the
patient’s heart. The injection process takes on average about one hour and can
be performed with or without general anesthesia. Following treatment, patients
generally remain in the hospital for approximately 48-72 hours for monitoring.
The MyoCell injection
process is a minimally invasive procedure which presents less risk and
considerably less trauma to a patient than conventional (open) heart surgery.
Patients are able to walk immediately following the injection process and
require significantly less time in the hospital compared with surgically treated
patients. In the 69 patients who have received MyoCell injections delivered via
percutaneous catheter, only two minor procedure-related events (2.9%) have been
reported. In both cases, however, no complications resulted from the event, with
the patients in each case remaining asymptomatic at all times during and after
the procedure.
We use a number of
proprietary processes to create therapeutic quantities of myoblasts from a
patient’s thigh muscle biopsy. We have developed and/or licensed what we believe
are proprietary or patented techniques to:
- transport muscle tissue and cultured
cells;
- disassociate muscle tissue with manual
and chemical processes;
- separate myoblasts from other muscle
cells;
- culture and grow myoblasts;
- identify a cell population with the
propensity to engraft, proliferate and adapt to the cardiac environment,
including areas of scar tissue; and
- maintain and test the cell quality and
purity.
58
We have also developed
and/or licensed a number of proprietary and/or patented processes related to the
injection of myoblasts into damaged heart muscle, including the
following:
- package the cultured cells in a manner
that facilitates shipping and use by the physician administering MyoCell;
- methods of using MyoCath;
- the use of an injectate media that
assists in the engraftment of myoblasts;
- cell injection techniques utilizing
contrast media to assist in the cell injection process; and
- cell injection protocols related to
the number and location of injections.
Assuming we secure
regulatory approval of MyoCell for the treatment of all NYHA Class II and NYHA
Class III patients, we believe MyoCell will provide a treatment alternative for
the millions of NYHA Class II and NYHA Class III patients in the United States
and Europe who either do not qualify for or do not have access to heart
transplant therapy. Furthermore, we anticipate that the time incurred and cost
of identifying patients qualified to receive MyoCell as well as the cost of
MyoCell, including any ICD, drug and bi-ventricular pacer therapies that are
simultaneously prescribed, if any, will be less expensive than the current cost
of heart transplant therapy. Moreover, MyoCell is less invasive than a heart
transplant and is not subject to the tissue rejection and immune system
suppression issues associated with heart transplants.
We believe there is still a
large population of patients exhibiting symptoms consistent with NYHA Class II
and NYHA Class III heart failure that is seeking an effective or more effective
therapy for chronic heart damage than ICDs, bi-ventricular pacers and drug
therapies. We hope to demonstrate that MyoCell is complementary to various
therapies using ICDs, bi-ventricular pacers and drugs. In the MYOHEART and
SEISMIC Trials, enrolled patients are required to have an ICD and to be on
optimal drug therapy to be included in the study. While we do not require
patients to have previously received a bi-ventricular pacer to participate in
our clinical trials, we plan to accept patients in our MARVEL Trial who have had
prior placement of a bi-ventricular pacer. We are hopeful that the results of
our future clinical trials will demonstrate that MyoCell is complementary to
existing therapies for treating heart damage.
Metrics Used to
Evaluate Safety and Efficacy of Heart Failure Treatments
The performance of
therapies used to treat damage to the heart is assessed using a number of
metrics, which compare data collected at the time of initial treatment to data
collected when a patient is re-assessed at follow-up. The time periods for
follow-up are usually three, six and twelve months. Statistical data is often
accompanied by a p-value, which is the mathematical probability that the data
are the result of random chance. A result is considered statistically
significant if the p-value is less than or equal to 5%. The common metrics used
to evaluate the efficacy of these therapies include:
| Metric |
Description |
|
NYHA
Class |
The NYHA heart failure
classification system is a functional and therapeutic classification
system based on how much cardiac patients are limited during physical
activity. |
|
Six-Minute
Walk
Distance |
Six-Minute Walk Distance
is an objective evaluation of functional exercise capacity which measures
the distance a patient can walk in six minutes. The distance walked during
this test has been shown to correlate with the severity of heart failure.
|
|
LVEF |
LVEF is a measure of the
heart’s efficiency and can be used to estimate the function of the left
ventricle, which pumps blood to the rest of the body. The LVEF is the
amount of blood pumped divided by the amount of blood the ventricle
contains. A normal LVEF is more than 55% of the blood volume. Damage to
the heart impairs the heart’s ability to efficiently pump and therefore
reduces LVEF. |
|
Quality of Life
|
Quality of Life is
evaluated by patient questionnaire, which measures subjective aspects of
health status in heart failure patients. |
|
Number of
Hospital
Admissions
and Mean
Length of
Stay |
The Number of Hospital
Admissions and Mean Length of Stay measure the aggregate number of
times that a patient is
admitted to the hospital during a defined period and the number of days a
patient remains in the hospital during each such admission.
|
59
|
Total
Days
Hospitalized |
The Total Days
Hospitalized measures the aggregate number of days a patient is admitted
to the hospital during a defined period. |
|
End-Systolic
Volume |
End-Systolic Volume is a
measurement of the adequacy of cardiac emptying, related to the function
of the heart during contraction. |
|
End-Diastolic
Volume |
End-Diastolic Volume is
the amount of blood in the ventricle immediately before a cardiac
contraction begins and is used as a measurement of the function of the
heart at rest. |
|
LV Volume
|
Left Ventricular Volume,
or LV Volume, is measured in terms of left ventricular End-Diastolic
Volume and left ventricular End-Systolic Volume. Both measure the
reduction in volume of blood in the left ventricle of the heart following
expansion and contraction, respectively. Reduction in volume generally is
reflective of positive ventricular remodeling and improvement in the
heart’s ability to circulate oxygenated blood through the
arteries. |
|
Wall Motion
|
Wall Motion is a test
designed to show whether the heart is receiving adequate quantities of
oxygen-rich blood. Wall motion is generally measured by a stress
echocardiography test. |
|
Cardiac
Output
|
Cardiac Output is a
measure of the amount of blood that is pumped by the heart per unit time,
measured in liters per minute. |
|
BNP Level
|
B-Type Natriuretic
Peptide, or BNP, is a substance secreted from the ventricles or lower
chambers of the heart in response to changes in pressure that occur when
heart failure develops and worsens. The level of BNP in the blood
increases when heart failure symptoms worsen and decreases when the heart
failure condition is stable. |
Marvel Phase II/III
Clinical Trial in the United States
The Marvel Trial is
designed to be a double-blind, randomized, placebo-controlled multicenter trial
to evaluate the safety and efficacy of MyoCell. In August 2007, we received
clearance from the FDA to proceed with the trial. We completed the MyoCell
implantation procedure on the first patient in the Marvel Trial on October 24,
2007. Thus far, 20 patients, including 6 control patients, have been treated.
Initial results for the 20 patients were released at the Heart Failure Society
of American meeting in September, 2009, showing a dramatic (35%) improvement in
the 6 minute walk for those patients who were treated, and no improvement for
those who received a placebo. We are planning, on the basis of these results, to
ask the FDA to consider the Marvel Trial a pivotal trial (pivotal from Phase II
to Phase III) and to reduce the number of patients in the trial to 150. The
Seismic, Myoheart and Marvel Trials have been designed to test the safety and
efficacy of MyoCell in treating patients with severe, chronic damage to the
heart. Upon regulatory approval of MyoCell, we intend to generate revenue in the
United States from the sale of MyoCell cell-culturing services for treatment of
patients by qualified physicians. Abroad, we are identifying centers where it is
already acceptable to use the Myocell treatment so that greater numbers of
patients with this problem can have access to treatment.
We are currently in the
process of evaluating our development timeline for MyoCell and the Marvel Trial.
We received approval from the FDA in July of 2009 to conduct a Phase I safety
study on 15 patients of a combined therapy (Myocell with SDF-1), the first
approval of a study combining gene and cell therapies. We initially commenced
work on this study, called the Regen Trial, during the first quarter of 2010.
The Company suspended activity on the trial in 2010 while seeking additional
funding necessary to conduct the trial. Work on the trial was reinitiated in 2011. Based
on the results of the trial, we intend to either incorporate the combined
treatment into the Marvel Trial, or continue with the Marvel Trial based on the
use of Myocell alone.
60
All of the patients selected for enrollment in the Marvel Trial have (i)
symptoms associated with NYHA Class II or NYHA Class III heart failure, (ii)
suffered a previous heart attack at least 90 days prior to the date of
treatment, (iii) a LVEF of less than or equal to 35%, (iv) been on optimal drug
therapy for at least two months prior to enrollment and (v) had prior placement
of an ICD at least 60 days prior to enrollment. Patients were required to use
Amiodarone, an anti-arrhythmic drug therapy, at least 24 hours prior to MyoCell
implantation. This prophylactic treatment successfully ameliorated the problem
with arrhythmias in patients treated with Myocell and the placebo, which,
although never lead to any deterioration in the patients, was considered a
serious adverse event.
The patients were divided into three
groups. Patients in the first group underwent treatment consisting of 16
injections of an aggregate dosage of approximately 800 million myoblast cells.
Patients in the second group underwent treatment consisting of 16 injections of
an aggregate dosage of approximately 400 million myoblast cells. Patients in the
third group received 16 placebo injections.
The Marvel Trial will measure the
following safety and efficacy endpoints of the MyoCell treatment:
| Primary Safety |
Primary Efficacy |
Secondary Efficacy |
Tertiary Efficacy |
| Endpoint |
Endpoints |
Endpoints |
Endpoints |
| Number of serious adverse events in treatment group as compared to
control group |
Change in Six-Minute
Walk Distance from baseline to six months as compared to control group, or
Quality of Life
scores assessed using Minnesota Living with Heart Failure questionnaire
from baseline to six months as compared to control group |
Total Days Hospitalized in treatment group as compared to control
group
Cause-specific
hospitalizations in treatment group as compared to control group
Proportion of
patients with an improved NYHA Class from baseline to six months as
compared to control group
Total days alive out
of hospital over the six-month study period
Change in LVEF from
baseline to six months as compared to control group
Change in LV Volume
and wall motion from baseline to six months as compared to control group
Change in BNP Level
from baseline to six months as compared to control group |
Total cost and
healthcare utilization within six months’ Time to death or CHF
hospitalization
Change in degree of
mitral regurgitation from baseline to six months
Change in Six-Minute
Walk Distance from baseline to three months as compared to control group
Quality of Life
scores assessed using Minnesota Living with Heart Failure questionnaire
from baseline to three months as compared to control group
Proportion of
patients with improved NYHA Class from baseline to three months as
compared to control group |
61
Pipeline
We
are committed to delivering biologics that help treat heart failure and
cardiovascular diseases. In addition to MyoCell, we have multiple cell therapies
and related devices for the treatment of chronic and acute heart damage in
various stages of development. We have also acquired the rights to use certain
devices for the treatment of heart damage. We intend to allocate our capital,
material and personnel resources among MyoCell and the product candidates
described below, a number of which may have complementary therapeutic
applications. For each product candidate, we have developed or are in the
process of developing a regulatory approval plan. Assuming such proposed plans
are able to be followed, we do not anticipate that the regulatory approval of
MyoCell will be necessary for our further development of our other product
candidates.
| Candidate |
Proposed Use or Indication |
Status/Phase |
Comments |
| MyoCell SDF-1 |
Autologous cell therapy |
IND application filed in |
Trial commenced in April 1, |
|
treatment for severe
chronic |
May 2007. Additional |
2010. |
|
damage to the heart;
cells |
animal studies complete. |
|
|
modified to express
angiogenic |
Phase I trial approved
by |
|
|
factors |
FDA
in July of 2009. |
|
| AdipoCell |
Adipose cell therapy treatment |
N/A |
Laboratory kits currently |
|
for the management of |
|
available for purchase |
|
progressive heart failure |
|
|
| MyoCath |
Disposable
endoventricular |
Used in European Phase |
Currently utilizing catheters in |
|
catheter used for the delivery
of |
II clinical
trials of |
trials. |
|
biologic solutions to
the |
MyoCell; used in Phase I |
|
|
myocardium |
clinical trials of |
|
|
|
MyoCell. |
|
| MyoCath II |
Second generation disposable |
Preclinical |
|
|
endoventricular catheter |
|
|
|
modified to provide |
|
|
|
multidirectional cell injection |
|
|
|
and used for the delivery of |
|
|
|
biologic solutions to the |
|
|
|
myocardium |
|
|
MyoCell
SDF-1
Our MyoCell SDF-1 product candidate, which has recently completed
preclinical testing, is intended to be an improvement to MyoCell. In February
2006, we signed a patent licensing agreement with the Cleveland Clinic of
Cleveland, Ohio which gave us exclusive license rights to pending patent
applications in connection with MyoCell SDF-1. Dr. Marc Penn, the Medical
Director of the Cardiac Intensive Care Unit at the Cleveland Clinic and a staff
cardiologist in the Departments of Cardiovascular Medicine and Cell Biology,
joined our Scientific Advisory Board. The license for SDF-1 was passed on to a
Cleveland Clinic affiliate, Juventas, in July of 2009. Bioheart has an
understanding with Juventas pursuant to which the license with Bioheart will be
reinstated upon completion of certain financial milestones.
62
We
anticipate that MyoCell SDF-1 will be similar to MyoCell, except that the
myoblast cells to be injected will be modified prior to injection by an
adenovirus vector or non-viral vector so that they will release extra quantities
of the SDF-1 protein, which expresses angiogenic factors. Following injury which
results in inadequate blood flow to the heart, such as a heart attack, the human
body naturally increases the level of SDF-1 protein in the heart. By modifying
the myoblasts to express additional SDF-1 prior to injection, we are seeking to
increase the SDF-1 protein levels present in the heart. We are seeking to
demonstrate that the presence of additional quantities of SDF-1 protein released
by the myoblasts will stimulate the recruitment of the patient’s existing stem
cells to the cell transplanted area and, thereafter, the recruited stem cells
will assist in the tissue repair and blood vessel formation process. Preclinical
animal studies showed a definite improvement of cardiac function when the
myoblasts were modified to express additional SDF-1 protein prior to injection
as compared to when the myoblasts were injected without modification.
Our Phase I safety study, the REGEN
Trial, was approved by the FDA in July of 2009 and work commenced on the trial
during the first quarter of 2010. The Company suspended activity on the trial in
2010 while seeking additional funding necessary to conduct the trial. Work on
the trial was reinitiated in 2011.
AdipoCell
The AdipoCell product candidate is a
proposed treatment for the management of progressive heart failure in patients
with damaged myocardial tissue resulting from prior myocardial infarction. This
product candidate contains autologous adipose cells that are prepared bedside to
the patient. By injecting AdipoCell into damaged, akinetic areas of myocardium,
these regions may have the ability to recruit stem cells to repair damaged
regions, thereby improving heart function. It is anticipated that AdipoCell will
be delivered directly into the myocardium of the hearts of patients suffering
from Congestive Heart Failure via a needle-injection catheter system.
In addition to the studies in Mexico,
Bioheart is distributing the AdipoCell kits for a variety of indications in many
countries. We are actively involved in a variety of clinical studies including
COPD, macular degeneration, degenerative disc disease, erectile dysfunction and
more. Bioheart therapies are being offered in centers and hospitals in Turkey,
Mexico, Jamaica, Australia, South Africa and others.
MyoCath
The MyoCath was developed by Bioheart
co-founder Robert Lashinski specifically for delivering new cells to damaged
tissue. It is a deflecting tip needle injection catheter that has a larger
needle which is 25 gauge for better flow rates and less leakage than systems
that are 27 gauge. This larger needle allows for thicker compositions to be
injected which helps with cell retention in the heart. Also, the MyoCath needle
has more fluoroscopic brightness than the normally used nitinol needle, enabling
superior visualization during the procedure. Seeing the needle well during
injections enables the physician who is operating the catheter to pinpoint
targeted areas more precisely, thus improving safety. The MyoCath competes well
with other biological delivery systems on price and efficiency and allows the
physician to utilize standard fluoroscopy and echo equipment found in every cath
lab. The MyoCath is used to inject cells into cardiac tissue in therapeutic
procedures to treat chronic heart ischemic and congestive heart
failure.
Research
We supervise and perform experimental
work in the areas of improving cell culturing, cell engraftment, and other
advanced research projects related to our product candidates from our cell
culturing facility in Sunrise, Florida. The primary focus of a substantial
majority of our employees is advancing our clinical trials, preclinical studies,
research and product development.
In addition, we work with a number of
third parties within and outside the United States on various research and
product development projects, including:
- preclinical small and large
animal testing for product candidate enhancements and pipeline product
candidate development; and contract research for clinical
and
- preclinical testing of our pipeline
product candidates.
63
We
have also began studies for an immunotherapy product for cancer. The product is
intended for the vet market and early in vitro studies in dogs have been
initiated. In January 2014, we announced an exclusive license for the patents
associated with these therapies and continue working on the vitro studies.
Cell
Culturing
We have an approximately 2,000 square
foot cell culturing facility at our headquarters in Sunrise, Florida. We began
culturing cells at this facility for preclinical uses in the third quarter of
2006. Upon commencement of the MARVEL Trial in the fourth quarter of 2007, we
began culturing cells at this facility for clinical uses.
Over the last three years, we have
significantly improved our ability to:
- culture in excess of 800 million
myoblast cells per biopsy; and
- produce cell cultures with a
high percentage of viable myoblast cells.
Accordingly, we were able to increase the maximum dosage of myoblast
cells injected as part of the MyoCell therapy to approximately 800 million
myoblast cells. We expect to further refine our MyoCell cell culturing
processes.
We have historically met and, with
respect to the cell culturing of our product candidates in Europe, expect to
meet, our cell culturing needs internally.
In 2013, Bioheart became an FDA
registered tissue bank in order to provide stem cell banking capabilities. We
are currently providing these services to physicians who send tissue samples
from their patients for culture expansion and cryopreservation.
Third Party
Reimbursement
Government and private insurance
programs, such as Medicare, Medicaid, health maintenance organizations and
private insurers, fund the cost of a significant portion of medical care in the
United States. As a result, government imposed limits on reimbursement of
hospitals and other healthcare providers have significantly impacted their
spending budgets and buying decisions. Under certain government insurance
programs, a healthcare provider is reimbursed a fixed sum for services rendered
in treating a patient, regardless of the actual cost of such treatment incurred
by the healthcare provider. Private third party reimbursement plans are also
developing increasingly sophisticated methods of controlling healthcare costs
through redesign of benefits and exploration of more cost-effective methods of
delivering healthcare. In general, we believe that these government and private
measures have caused healthcare providers to be more selective in the purchase
of medical products.
As of the date of this report, CMS has
agreed to reimburse some of the costs at the centers that are participating in
the MARVEL Trial. Specifically, CMS will reimburse costs deemed “routine” in
nature for patients suffering from heart failure. Examples of these reimbursable
costs include, but are not limited to, costs associated with physical
examination of the patients, x-rays, holter monitoring, MUGA scan and
echocardiography. However, at present, CMS reimbursement does not cover the cost
of MyoCell implantation.
Reimbursement for healthcare costs
outside the United States varies from country to country. In European countries,
the pricing of prescription pharmaceutical products and services and the level
of government reimbursement are subject to governmental control. In these
countries, pricing negotiations with governmental authorities can take six to
twelve months or longer after the receipt of marketing approval for a product.
To obtain reimbursement or pricing approval in some countries, we may be
required to conduct one or more clinical trials that compare the cost
effectiveness of our product candidates to other available therapies. Conducting
one or more clinical trials would be expensive and result in delays in
commercialization of our product candidates.
64
Research
Grants
Historically, part of our research and development efforts have been
indirectly funded by research grants to various centers and/or physicians that
have participated in our MyoCell and MyoCath clinical trials. As part of our
development strategy, we intend to continue to seek to develop research
partnerships with centers and/or physicians. On November 1, 2010, Bioheart, Inc.
received written notice of approval of a grant in the approximate amount of
$244,500 under the qualifying therapeutic discovery project under section 48D of
the Internal Revenue code. On November 9, 2010 Bioheart received the $244,500
grant funds.
Patents and Proprietary
Rights
We own or hold licenses or sublicenses to
an intellectual property portfolio consisting of numerous patents and patent
applications in the United States, and in foreign countries, for use in the
field of heart muscle regeneration. References in this report to “our” patents
and patent applications and other similar references include the patents and
patent applications that are owned by us, and references to patents and patent
applications that are “licensed” to us and other similar references refer to
patents, patent applications and other intellectual property that are licensed
or sublicensed to us.
Patent life determination depends on the
date of filing of the application or the date of patent issuance and other
factors as promulgated under the patent laws. Under the U.S. Drug Price
Competition and Patent Term Restoration Act of 1984, as amended, a patent which
claims a product, use or method of manufacture covering drugs and certain other
products, including biologic products, may be extended for up to five years to
compensate the patent holder for a portion of the time required for research and
FDA review of the product. Only one patent applicable to an approved drug or
biologic product is eligible for a patent term extension. This law also
establishes a period of time following approval of a drug or biologic product
during which the FDA may not accept or approve applications for certain similar
or identical drugs or biologic products from other sponsors unless those
sponsors provide their own safety and efficacy data.
MyoCell is no longer protected by patents
outside, which means that competitors will be free to sell products that
incorporate the same or similar technologies that are used in MyoCell without
infringing our patent rights. As a result, MyoCell, if approved for use, may be
vulnerable to competition. In addition, many of the patent and patent
applications that have been licensed to us that pertain to our other product
candidates do not cover certain countries within Europe.
Our commercial success will depend to a
significant degree on our ability to:
- defend and enforce our patents
and/or compel the owners of the patents licensed to us to defend and enforce
such patents, to the extent such patents may be applicable to our products and
material to their commercialization;
- obtain additional patent and
other proprietary protection for MyoCell and our other product
candidates;
- obtain and/or maintain
appropriate licenses to patents, patent applications or other proprietary
rights held by others with respect to our technology, both in the United
States and other countries;
- preserve company trade secrets
and other intellectual property rights relating to our product candidates;
and
- operate without infringing the
patents and proprietary rights of third parties.
In addition to patented intellectual
property, we also rely on our own trade secrets and proprietary know-how to
protect our technology and maintain our competitive position, since patent
protection may not be available or applicable to our technology. Our policy is
to require each of our employees, consultants and advisors to execute a
confidentiality and inventions assignment agreement before beginning their
employment, consulting or advisory relationship with us. The agreements
generally provide that the individual must keep confidential and not disclose to
other parties any confidential information developed or learned by the
individual during the course of the individual’s relationship with us except in
limited circumstances. These agreements generally also provide that we shall own
all inventions conceived by the individual in the course of rendering services
to us. Moreover, some of our academic institution licensors, collaborators and
scientific advisors have rights to publish data and information to which we have
rights, which may impair our ability to protect our proprietary information or
obtain patent protection in the future.
65
We
work with others in our research and development activities and one of our
strategies is to enter into collaborative agreements with third parties to
develop our proposed products. Disputes may arise about inventorship and
corresponding rights in know-how and inventions resulting from the joint
creation or use of intellectual property by us and our licensors, collaborators,
consultants and others. In addition, other parties may circumvent any
proprietary protection we do have. As a result, we may not be able to maintain
our proprietary position.
We are not currently a party to any
litigation or other adverse proceeding related to our patents, patent licenses
or intellectual property rights. However, if we become involved in litigation or
any other adverse intellectual property proceeding, for example, as a result of
an alleged infringement, or a third party alleging an earlier date of invention,
we may have to spend significant amounts of money and time and, in the event of
an adverse ruling, we could be subject to liability for damages, including
treble damages, invalidation of our intellectual property and injunctive relief
that could prevent us from using technologies or developing products, any of
which could have a significant adverse effect on our business, financial
condition and results of operation. In addition, any claims relating to the
infringement of third party proprietary rights, or earlier date of invention,
even if not meritorious, could result in costly litigation, lengthy governmental
proceedings, divert management’s attention and resources and require us to enter
royalty or license agreements which are not advantageous, if available at all.
See Item 1A. “Risk Factors — Risks
Related to Our Intellectual Property” for a discussion of additional risks we
face with respect to our intellectual property rights.
Primary MyoCath
Patent
The Primary MyoCath Patent includes
device claims that we believe covers, among other things, the structure of
MyoCath. The Primary MyoCath Patent expires in the United States in September
2017.
In January 2000, we entered into a
license agreement with Comedicus Incorporated pursuant to which Comedicus
granted us a royalty-free, fully paid-up, non-exclusive and irrevocable license
to the Primary MyoCath Patent in exchange for a payment of $50,000. This
agreement was amended in August 2000 to provide us an exclusive license to the
Primary MyoCath Patent in exchange for a payment of $100,000 and our loan of
$250,000 to Comedicus. Pursuant to this amendment we also received the right,
but not the obligation, with Comedicus’ consent, which consent is not to be
unreasonably withheld, to defend the Primary MyoCath Patent against third party
infringers.
In June 2003, we entered into agreements
with Advanced Cardiovascular Systems, Inc., or ACS, originally a subsidiary of
Guidant Corporation and now d/b/a Abbott Vascular, a division of Abbott
Laboratories, pursuant to which we assigned our rights under the license
agreement with Comedicus, as amended, and committed to deliver 160 units of
MyoCath and sold certain of our other catheter related intellectual property,
or, collectively, with the Primary MyoCath Patent (the Catheter IP), for
aggregate consideration of $900,000. In connection with these agreements, ACS
granted to us a co-exclusive, irrevocable, fully paid-up license to the Catheter
IP for the life of the patents related to the Catheter IP.
ACS has the exclusive right, at its own
expense, to file, prosecute, issue, maintain, license, and defend the Catheter
IP, and the primary right to enforce the Catheter IP against third party
infringers. If ACS fails to enforce the Catheter IP against a third party
infringer within a specified period of time, we have the right to do so at our
expense. The party enforcing the Catheter IP is entitled to retain any
recoveries resulting from such enforcement. The asset purchase agreement only
pertains to the Catheter IP developed or acquired by us prior to June 24, 2003.
Our subsequent catheter related developments and/or acquisitions, such as
MyoCath II, were not sold or licensed to ACS.
MyoCell SDF-1
Patents
To develop our MyoCell SDF-1 product
candidate, we rely primarily on patents. We had an agreement to license patents
from Juventas. These patents relate to methods of repairing damaged heart tissue
by transplanting myoblasts that express SDF-1 and other therapeutic proteins
capable of recruiting other stem cells within a patient’s own body to the cell
transplant area. We believe we will also need to, among other things, license
some additional intellectual property to commercialize MyoCell SDF-1 in the form
we believe may prove to be the most safe and/or effective.
66
In February 2006, we signed a patent
licensing agreement with the Cleveland Clinic which provided us with the
worldwide, exclusive rights to three pending U.S. patent applications and
certain corresponding foreign filings in the following jurisdictions: Australia,
Brazil, Canada, China, Europe and Japan, or, collectively, the Cleveland Clinic
IP, related to methods of repairing damaged heart tissue by transplanting
myoblasts that express SDF-1 and other therapeutic proteins capable of
recruiting other stem cells within a patient’s own body to the cell transplant
area. The term of our agreement with the Cleveland Clinic expired in July of
2009, when the license to the patents was turned over to a Cleveland Clinic
affiliate, Juventas. We have an understanding with Juventas to restore the
license to the patents once certain milestones have been achieved by Bioheart.
In 2007, Bioheart signed a Letter of
Intent with Ono Pharmaceutical which provided rights to conduct clinical
development and testing of SDF-1 to determine the effectiveness of SDF-1 for the
treatment of damaged myocardium and tissues following acute myocardial
infarction, coronary arterial diseases or heart failure. If the results of this
testing is deemed successful then the parties agree to enter into good faith
negotiations in an effort to reach a definitive license agreement that will
allow Bioheart to commercialize its SDF-1 product candidate in all territories
of the world except Japan.
MyoCath II
Patents
In April 2006, we entered into an
agreement with Tricardia, LLC pursuant to which Tricardia granted us a
sublicenseable license to certain patents and patent applications in the United
States, Australia, Canada, Europe and Japan covering the modified injection
needle we intend to use as part of MyoCath II, or the MyoCath II Patents, in
exchange for a one-time payment of $100,000. Our license covers and is exclusive
with respect to products developed under the MyoCath II Patents for the delivery
of therapeutic compositions to the heart. Unless earlier terminated by mutual
consent of the parties, our agreement with Tricardia will terminate upon the
expiration date of the last MyoCath II Patent.
Tricardia has the obligation to take all
actions necessary to file, prosecute and maintain the MyoCath II Patents. We are
required to reimburse Tricardia, on a pro-rata basis with other licensees of
Tricardia of the MyoCath II Patents, for all reasonable out-of-pocket costs and
expenses incurred by Tricardia in prosecuting and maintaining the MyoCath II
Patents. To the extent we do not wish to incur the cost of any undertaking or
defense of any opposition, interference or similar proceeding involving the
MyoCath II Patents with respect to any jurisdiction, the license granted to us
pursuant to agreement will be automatically amended to exclude such
jurisdiction.
Tricardia also has the first right, but
not the obligation, to take any actions necessary to prosecute or prevent any
infringement or threatened infringement of the MyoCath II Patents. To the extent
Tricardia determines not to initiate suit against any infringer, we have the
right, but not the obligation, to commence litigation for such alleged
infringement. Our share of any recovery will equal 50% in the event Tricardia
commences litigation and 90% in the event we commence litigation.
Sales and
Marketing
MyoCell and MyoCell
SDF-1
In advance of any expected commercial
approval of our MyoCell product candidate in the United States, we intend to
internally develop a direct sales and marketing force. We anticipate the team
will comprise salespeople, clinical and reimbursement specialists and product
marketing managers.
We intend to market MyoCell and/or
MyoCell SDF-1 to interventional cardiologists and heart failure specialists. In
the typical healthcare system the interventional cardiologist functions as a
“gatekeeper” for determining the course of appropriate medical care for our
target patient population.
67
We anticipate our marketing efforts will
be focused on informing interventional cardiologists of the availability of our
treatment alternative through the following channels of communication: (i)
articles published in medical journals by widely recognized interventional
cardiologists, including cardiologists that have participated in our clinical
trials; (ii) seminars and speeches featuring widely recognized interventional
cardiologists; and (iii) advertisements in medical journals.
Collaborative
Arrangements for Seeking Regulatory Approvals and Distribution of Products
Outside of the United States and Europe
Adipose-stem cell
therapies
We have partnered with the Global Stem
Cell Group to market and make available our AdipoCell adipose derived stem cell
therapies to all doctors across the U.S. and in foreign markets.
Korea
On February 1, 2005, we entered into a
joint venture agreement with Bioheart Korea, Inc., the predecessor entity of
BHK, Inc., or BHK, pursuant to which we and BHK agreed to create a joint venture
company called Bioheart Manufacturing, Inc., located in Korea to own and operate
a cell culturing facility. The joint venture agreement contemplated that we
would engage Bioheart Manufacturing to provide all cell culturing processes for
our products and processes sold in Asia including Korea for a period of no less
than ten years. Pursuant to the joint venture agreement, we agreed to contribute
approximately $59,000 cash and Myocell manufacturing technology for an 18%
equity interest in Bioheart Manufacturing, and BHK agreed to contribute
approximately $9,592,032 for an 82% equity interest in Bioheart Manufacturing.
On April 1, 2006, we entered into an in-kind investment agreement with Bioheart
Manufacturing pursuant to which we agreed to provide Bioheart Manufacturing with
the technology to manufacture MyoCell and MyoCath and, in exchange, received
25,890 common shares of Bioheart Manufacturing. In February 2009, our ownership
interest in Bioheart Manufacturing, Inc. was reduced to approximately 6% as a
result of additional investments in Bioheart Manufacturing, Inc. by third
parties. Shares in Bioheart Manufacturing, Inc., owned by
Bioheart Inc. became 258,900 shares through a stock split. In May 2009, there
was a corporate reorganization and AnC Bio Inc. was given the business of
Bioheart Manufacturing Inc.
Pursuant to the joint venture agreement,
we provided Bioheart Manufacturing with standard operating procedures, tests and
testing protocols, cell selection methods, cell characterization methods, and
all materials necessary to carry out the activities of the cell culturing
facility in the manner required by us.
In August 2007, we entered into a supply
agreement with BHK pursuant to which we supplied MyoCell and MyoCaths to BHM for
use in clinical studies of MyoCell.
According to the agreement, upon Bioheart
Manufacturing’s inability to continue its operations by reason of law,
governmental order or regulation or Bioheart Manufacturing’s dissolution or
liquidation for any reason, the agreement is null and void.
Government
Regulation
The research and development, preclinical
studies and clinical trials, and ultimately, the culturing, manufacturing,
marketing and labeling of our product candidates are subject to extensive
regulation by the FDA and other regulatory authorities in the United States and
other countries. We believe MyoCell and our medical device products are subject
to regulation in the United States and Europe as a biological product and a
medical device, respectively.
Biological products are subject to
regulation under the Federal Food, Drug, and Cosmetic Act, or the FD&C Act,
the Public Health Service Act, or the PHS Act and their respective regulations
as well as other federal, state, and local statutes and regulations. Medical
devices are subject to regulation under the FD&C Act and the regulations
promulgated hereunder as well as other federal, state, and local statutes and
regulations. The FD&C Act and the PHS Act and the regulations promulgated
hereunder govern, among other things, the testing, cell culturing,
manufacturing, safety, efficacy, labeling, storage, record keeping, approval,
clearance, advertising and promotion of our product candidates. Preclinical
studies, clinical trials and the regulatory approval process typically take
years and require the expenditure of substantial resources. If regulatory
approval or clearance of a product is granted, the approval or clearance may
include significant limitations on the indicated uses for which the product may
be marketed.
68
FDA Regulation —
Approval of Biological Products
The steps ordinarily
required before a biological product may be marketed in the United States
include:
- completion of preclinical
studies according to good laboratory practice regulations;
- the submission of an IND
application to the FDA, which must become effective before human clinical
trials may commence;
- performance of adequate and
well-controlled human clinical trials according to good clinical practices to
establish the safety and efficacy of the proposed biological product for its
intended use;
- satisfactory completion of an
FDA pre-approval inspection of the manufacturing facility or facilities at
which the product is manufactured, processes, packaged or held to assess
compliance cGMP; and
- the submission to, and review
and approval by, the FDA of a biologics license application, or BLA, that
includes satisfactory results of preclinical testing and clinical trials.
Preclinical tests include laboratory evaluation of the product candidate,
its formulation and stability, as well as animal studies to assess the potential
safety and efficacy of the product candidate. The FDA requires that preclinical
tests be conducted in compliance with good laboratory practice regulations. The
results of preclinical testing are submitted as part of an IND application to
the FDA together with manufacturing information for the clinical supply,
analytical data, the protocol for the initial clinical trials and any available
clinical data or literature. A 30-day waiting period after the filing of each
IND application is required by the FDA prior to the commencement of clinical
testing in humans. In addition, the FDA may, at any time during this 30-day
waiting period or any time thereafter, impose a clinical hold on proposed or
ongoing clinical trials. If the FDA imposes a clinical hold, clinical trials
cannot commence or recommence without FDA authorization.
Clinical trials to support BLAs involve
the administration of the investigational product to human subjects under the
supervision of qualified investigators. Clinical trials are conducted under
protocols detailing, among other things, the objectives of the study, the
parameters to be used in monitoring safety and the efficacy criteria to be
evaluated.
Clinical trials are typically conducted
in three sequential phases, but the phases may overlap.
In Phase I clinical trials, the initial
introduction of the biological product candidate into human subjects or
patients, the product candidate is tested to assess safety, dosage tolerance,
absorption, metabolism, distribution and excretion, including any side effects
associated with increasing doses.
Phase II clinical trials usually involve
studies in a limited patient population to identify possible adverse effects and
safety risks, preliminarily assess the efficacy of the product candidate in
specific, targeted indications; and assess dosage tolerance and optimal dosage.
If a product candidate is found to be
potentially effective and to have an acceptable safety profile in Phase II
evaluations, Phase III trials are undertaken within an expanded patient
population at multiple study sites to further demonstrate clinical efficacy and
safety, further evaluate dosage and establish the risk-benefit ratio of the
product and an adequate basis for product labeling.
Phase IV, or post-marketing, trials may
be mandated by regulatory authorities or may be conducted voluntarily. Phase IV
trials are typically initiated to monitor the safety and efficacy of a
biological product in its approved population and indication but over a longer
period of time, so that rare or long-term adverse effects can be detected over a
much larger patient population and time than was possible during prior clinical
trials. Alternatively, Phase IV trials may be used to test a new method of
product administration, or to investigate a product’s use in other indications.
Adverse effects detected by Phase IV trials may result in the withdrawal or
restriction of a drug.
69
If the required Phase I, II and III
clinical testing is completed successfully, the results of the required clinical
trials, the results of product development, preclinical studies and clinical
trials, descriptions of the manufacturing process and other relevant information
concerning the safety and effectiveness of the biological product candidate are
submitted to the FDA in the form of a BLA. In most cases, the BLA must be
accompanied by a substantial user fee. The FDA may deny a BLA if all applicable
regulatory criteria are not satisfied or may require additional data, including
clinical, toxicology, safety or manufacturing data. It can take several years
for the FDA to approve a BLA once it is submitted, and the actual time required
for any product candidate may vary substantially, depending upon the nature,
complexity and novelty of the product candidate.
Before approving an application, the FDA
will inspect the facility or facilities where the product is manufactured. The
FDA will not approve a BLA unless it determines that the manufacturing processes
and facilities are in compliance with cGMP requirements.
If the FDA evaluations of the BLA and the
manufacturing facilities are favorable, the FDA may issue either an approval
letter or an approvable letter. The approvable letter usually contains a number
of conditions that must be met to secure final FDA approval of the BLA. When,
and if, those conditions have been met to the FDA’s satisfaction, the FDA will
issue an approval letter. If the FDA’s evaluation of the BLA or manufacturing
facility is not favorable, the FDA may refuse to approve the BLA or issue a
non-approvable letter that often requires additional testing or information.
FDA Regulation —
Approval of Medical Devices
Medical devices are also subject to
extensive regulation by the FDA. To be commercially distributed in the United
States, medical devices must receive either 510(k) clearance or pre-market
approval, or PMA, from the FDA prior to marketing. Devices deemed to pose
relatively low risk are placed in either Class I or II, which requires the
manufacturer to submit a pre-market notification requesting permission for
commercial distribution, or 510(k) clearance. Devices deemed by the FDA to pose
the greatest risk, such as life-sustaining, life-supporting or implantable
devices, devices deemed not substantially equivalent to a previously 510(k)
cleared device and certain other devices are placed in Class III which requires
PMA. We anticipate that MyoCath will be classified as a Class III device.
To obtain 510(k) clearance, a
manufacturer must submit a pre-market notification demonstrating that the
proposed device is substantially equivalent in intended use and in safety and
efficacy to a previously 510(k) cleared device, a device that has received PMA
or a device that was in commercial distribution before May 28, 1976. The FDA’s
510(k) clearance pathway usually takes from four to twelve months, but it can
last longer.
After a device receives 510(k) clearance,
any modification that could significantly affect its safety or efficacy, or that
would constitute a major change in its intended use, requires a new 510(k)
clearance or could require PMA. The FDA requires each manufacturer to make this
determination, but the FDA can review any such decision. If the FDA disagrees
with a manufacturer’s decision not to seek a new 510(k) clearance, the agency
may retroactively require the manufacturer to seek 510(k) clearance or PMA. The
FDA also can require the manufacturer to cease marketing and/or recall the
modified device until 510(k) clearance or PMA is obtained.
A product not eligible for 510(k)
clearance must follow the PMA pathway, which requires proof of the safety and
efficacy of the device to the FDA’s satisfaction. The PMA pathway is much more
costly, lengthy and uncertain than the 510(k) approval pathway. A PMA
application must provide extensive preclinical and clinical trial data and also
information about the device and its components regarding, among other things,
device design, manufacturing and labeling. As part of the PMA review, the FDA
will typically inspect the manufacturer’s facilities for compliance with quality
system regulation requirements, which impose elaborate testing, control,
documentation and other quality assurance procedures. Upon acceptance by the FDA
of what it considers a completed filing, the FDA commences an in-depth review of
the PMA application, which typically takes from one to two years, but may last longer. The review
time is often significantly extended as a result of the FDA asking for more
information or clarification of information already provided.
70
If the FDA’s evaluation of
the PMA application is favorable, and the applicant satisfies any specific
conditions (e.g., changes in labeling) and provides any specific additional
information (e.g., submission of final labeling), the FDA will issue a PMA for
the approved indications, which can be more limited than those originally sought
by the manufacturer. The PMA can include post-approval conditions that the FDA
believes necessary to ensure the safety and efficacy of the device including,
among other things, restrictions on labeling, promotion, sale and distribution.
Failure to comply with the conditions of approval can result in an enforcement
action, which could have material adverse consequences, including the loss or
withdrawal of the approval.
Even after approval of a
pre-market application, a new PMA or PMA supplement is required in the event of
a modification to the device, its labeling or its manufacturing process.
FDA Regulation —
Post-Approval Requirements
Even if regulatory
clearances or approvals for our product candidates are obtained, our products
and the facilities manufacturing our products will be subject to continued
review and periodic inspections by the FDA. For example, as a condition of
approval of a new drug application, the FDA may require us to engage in
post-marketing testing and surveillance and to monitor the safety and efficacy
of our products. Holders of an approved new BLA, PMA or 510(k) clearance product
are subject to several post-market requirements, including the reporting of
certain adverse events involving their products to the FDA, provision of updated
safety and efficacy information, and compliance with requirements concerning the
advertising and promotion of their products.
In addition, manufacturing
facilities are subject to periodic inspections by the FDA to confirm the
facilities comply with cGMP requirements. In complying with cGMP, manufacturers
must expend money, time and effort in the area of production and quality control
to ensure full compliance. For example, manufacturers of biologic products must
establish validated systems to ensure that products meet high standards of
sterility, safety, purity, potency and identity. Manufacturers must report to
the FDA any deviations from cGMP or any unexpected or unforeseeable event that
may affect the safety, quality, or potency of a product. The regulations also
require investigation and correction of any deviations from cGMP and impose
documentation requirements.
In addition to regulations
enforced by the FDA, we are also subject to regulation under the Occupational
Safety and Health Act, the Environmental Protection Act, the Toxic Substances
Control Act, the Resource Conservation and Recovery Act and other federal, state
and local regulations. Our research and development activities involve the
controlled use of hazardous materials, chemicals, biological materials and
radioactive compounds.
International
Regulation
Our product candidates are
subject to regulation in every country where they will be tested or used.
Whether or not we obtain FDA approval for a product candidate, we must obtain
the necessary approvals from the comparable regulatory authorities of foreign
countries before we can commence testing or marketing of a product candidate in
those countries. The requirements governing the conduct of clinical trials and
the approval processes vary from country to country and the time required may be
longer or shorter than that associated with FDA approval.
The European Economic Area
requires that manufacturers of medical devices obtain the right to affix the CE
mark to their products before selling them in member countries. The CE mark is
an international symbol of adherence to quality assurance standards and
compliance with applicable European medical device directives. In order to
obtain the right to affix the CE mark to a medical device, the medical device in
question must meet the essential requirements defined under the Medical Device
Directive (93/42/ EEC) relating to safety and performance, and the manufacturer
of the device must undergo verification of regulatory compliance by a third
party standards certification provider, known as a notified body.
71
In addition to regulatory
clearance, the conduct of clinical trials in the European Union is governed by
the European Clinical Trials Directive (2001/20/ EC), which was implemented in
May 2004. This directive governs how regulatory bodies in member states may
control clinical trials. No clinical trial may be started without authorization
by the national competent authority and favorable ethics approval.
Manufacturing facilities
are subject to the requirements of the International Standards Organization. In
complying with these requirements, manufacturers must expend money, time and
effort in the area of production and quality control to ensure full compliance.
In some cases, we plan to
submit applications with different endpoints or other elements outside the
United States due to differing practices and requirements in particular
jurisdictions. However, in cases where different endpoints will be used outside
the United States, we expect that such submissions will be discussed with the
FDA to ensure that the FDA is comfortable with the nature of human trials being
conducted in any part of the world. As in the United States, post-approval
regulatory requirements, such as those regarding product manufacture, marketing,
or distribution, would apply to any product that is approved in Europe.
Competition
Our industry is subject to
rapid and intense technological change. We face, and will continue to face,
competition from pharmaceutical, biopharmaceutical, medical device and
biotechnology companies developing heart failure treatments both in the United
States and abroad, as well as numerous academic and research institutions,
governmental agencies and private organizations engaged in drug funding or
discovery activities both in the United States and abroad. We also face
competition from entities and healthcare providers using more traditional
methods, such as surgery and pharmaceutical regimens, to treat heart failure. We
believe there are a substantial number of heart failure products under
development by numerous pharmaceutical, biopharmaceutical, medical device and
biotechnology companies, and it is likely that other competitors will emerge.
Many of our existing and
potential competitors have substantially greater research and product
development capabilities and financial, scientific, marketing and human
resources than we do. As a result, these competitors may succeed in developing
competing therapies earlier than we do; obtain patents that block or otherwise
inhibit our ability to further develop and commercialize our product candidates;
obtain approvals from the FDA or other regulatory agencies for products more
rapidly than we do; or develop treatments or cures that are safer or more
effective than those we propose to develop. These competitors may also devote
greater resources to marketing or selling their products and may be better able
to withstand price competition. In addition, these competitors may introduce or
adapt more quickly to new technologies or scientific advances, which could
render our technologies obsolete, and may introduce products that make the
continued development of our product candidates uneconomical. These competitors
may also be more successful in negotiating third party licensing or
collaborative arrangements and may be able to take advantage of acquisitions or
other strategic opportunities more readily than we can.
Our ability to compete
successfully will depend on our continued ability to attract and retain skilled
and experienced scientific, clinical development and executive personnel, to
identify and develop viable heart failure product candidates and to exploit
these products and compounds commercially before others are able to develop
competitive products.
We believe the principal
competitive factors affecting our markets include, but are not limited
to:
- the safety and efficacy of our product
candidates;
- the freedom to develop and
commercialize cell-based therapies, including appropriate patent and
proprietary rights protection;
- the timing and scope of regulatory
approvals;
- the cost and availability of our
products;
- the availability and scope of third
party reimbursement programs; and
- the availability of alternative
treatments.
72
We are still in the process
of determining, among other things:
- if MyoCell and MyoCell SDF-1 are both
safe and effective;
- the timing and scope of regulatory
approvals; and
- the availability and scope of third
party reimbursement programs.
Accordingly, we have a
limited ability to predict how competitive MyoCell will be relative to existing
treatment alternatives and/or treatment alternatives that are under development.
See “Business — Diagnosis and Management of Heart Failure.”
If approved, MyoCell will
compete with surgical, pharmaceutical and mechanical based therapies. Surgical
options include heart transplantation and left ventricular reconstructive
surgery. Although not readily accessible, heart transplantation has proven to be
an effective treatment for patients with severe damage to the heart who locate a
donor match and are in sufficiently good health to undergo major surgery.
Mechanical therapies such as biventricular pacing, ventricular restraint devices
and mitral valve therapies have been developed by companies such as Medtronic,
Inc., Acorn Cardiovascular, Inc., St. Jude Medical, Inc., World Heart
Corporation, Guidant Corporation, a part of Boston Scientific, and Edwards
Lifesciences Corp. Pharmaceutical therapies include anti-thrombotics, calcium
channel blockers such as Pfizer’s Norvasc® and ACE inhibitors such as Sanofi’s
Delix®.
The field of regenerative
medicine is rapidly progressing, as many organizations are initiating or
expanding their research efforts in this area. We are also aware of several
competitors seeking to develop cell-based therapies for the treatment of
cardiovascular disease, including Aldagen, Inc., Angioblast Systems, Inc.,
Athersys, Inc., Baxter International, Inc., Cytori Therapeutics, Inc., MG
Biotherapeutics, LLC (a joint venture between Genzyme Corporation and Medtronic,
Inc.), Mytogen, Inc. (a wholly-owned subsidiary of Advanced Cell Technology,
Inc.), Osiris Therapeutics, Inc. (products now owned by Mesoblast), ViaCell,
Inc. (a wholly-owned subsidiary of PerkinElmer, Inc.), and potentially
others.
It is our understanding
that some of our large competitors have devoted considerable resources to
developing a myoblast-based cell therapy for treating severe damage to the
heart.
Some organizations are
involved in research using alternative cell sources, including bone marrow,
embryonic and fetal tissue, umbilical cord and peripheral blood, and adipose
tissue.
Many of our existing and
potential competitors have substantially greater research and product
development capabilities and financial, scientific, marketing and human
resources than we do. These competitors may also devote greater resources to
marketing or selling their products and may be better able to withstand price
competition. In addition, these competitors may introduce or adapt more quickly
to new technologies or scientific advances, which could render our technologies
obsolete, and may introduce products that make the continued development of our
product candidates uneconomical. These competitors may also be more successful
in negotiating third party licensing or collaborative arrangements and may be
able to take advantage of acquisitions or other strategic opportunities more
readily than we can.
DESCRIPTION OF SECURITIES
TO BE REGISTERED
General
The following summary
includes a description of material provisions of our capital stock.
Authorized and
Outstanding Securities
The Company is authorized
to issue 2,000,000,000 shares of common stock, par value $0.001 per share. As of
November 20, 2014, there were issued and outstanding 560,564,622 shares of our
common stock.
73
Common
Stock
The holders of our common
stock are entitled to one vote for each share on all matters to be voted on by
the shareholders. Holders of common stock do not have cumulative voting rights.
Holders of common stock are entitled to share ratably in dividends, if any, as
may be declared from time to time by the board of directors in its discretion
from funds legally available therefore. In the event of a liquidation,
dissolution or winding up of the Company, the holders of common stock are
entitled to share pro rata all assets remaining after payment in full of all
liabilities. Holders of common stock have no preemptive rights to purchase the
Company’s common stock. There are no conversion or redemption rights or sinking
fund provisions with respect to the common stock.
Dividends
Dividends, if any, will be
contingent upon our revenues and earnings, if any, capital requirements and
financial conditions. The payment of dividends, if any, will be within the
discretion of our board of directors. We intend to retain earnings, if any, for
use in its business operations and accordingly, the board of directors does not
anticipate declaring any dividends in the foreseeable future.
INTERESTS OF NAMED
EXPERTS AND COUNSEL
No expert or counsel named
in this prospectus as having prepared or certified any part of this prospectus
or having given an opinion upon the validity of the securities being registered
or upon other legal matters in connection with the registration or offering of
the common stock was employed for such purpose on a contingency basis, or had,
or is to receive, in connection with this offering, a substantial interest,
direct or indirect, in us or any of our parents or subsidiaries, nor was any
such person connected with us or any of our parents or subsidiaries as a
promoter, managing or principal underwriter, voting trustee, director, officer,
or employee.
LEGAL
MATTERS
The validity of the
issuance of the common stock hereby will be passed upon for us by Sichenzia Ross
Friedman Ference LLP.
OTHER EXPERTS
Our consolidated financial
statements as of December 31, 2013 and for the year then ended , appearing in
this prospectus and registration statement have been so included in reliance on
the Report of Fiondella, Milone, and LaSaracina LLP, independent registered
public accounting firm, appearing elsewhere in this prospectus, given on the
authority of such firm as experts in accounting and auditing.
Properties
Our headquarters are
located in Sunrise, Florida and consists of 4,860 square feet of space, which we
lease at a current rent of approximately $65,124 per year. The lease expired in
January 2013. In January 2013, the Company amended its facility lease to extend
the term of the lease until April 30, 2013. In April 2013, the Company amended
its facility lease to extend the term of the lease until August 15, 2013. In
September 2013, the Company amended its facility lease to extend the term until
August 2016.
In addition to our
corporate offices, at this location, we maintain:
- our MyoCell cell culturing facility
for supply within the United States; and
- a fully equipped cell culturing
laboratory where we perform experimental work in the areas of cell culturing,
cell engraftment, and other advanced research projects related to our core
business.
We believe the space
available at our headquarters will be sufficient to meet the needs of our
operations for the foreseeable future.
74
Legal Proceedings
November 10, 2014, the
company was served with a lawsuit by an alleged assignee and a guarantor to a
Loan Guarantee, Payment and Security Agreement. These parties claim breach of
that Agreement and damages of approximately $2.3 Million plus interest. The
assignor and assignee also sued the Company’s directors and a past director and
an affiliate shareholder for breach of fiduciary duty, claiming damages as
alleged creditors arising out of these parties' alleged participation in
Northstar Biotech Group, LLC, a secured creditor of the Company.
Other than the above, we
are not presently engaged in any material litigation pursuant to Item 103 of
Regulation S-K. However, the biotechnology and medical device industries have
been characterized by extensive litigation regarding patents and other
intellectual property rights. In addition, from time to time, we may become
involved in litigation relating to claims arising from the ordinary course of
our business.
75
MARKET PRICE AND
DIVIDENDS ON REGISTRANT’S COMMON EQUITY
AND RELATED STOCKHOLDER MATTERS
Our common stock is listed
on the Over-the-Counter Bulletin Board under the symbol “BHRT.QB”. For the
periods indicated, the following table sets forth the high and low bid prices
per share of common stock, as reported by the Over the Counter Bulletin Board.
These prices represent inter-dealer quotations without retail markup, markdown,
or commission and may not necessarily represent actual transactions.
|
High |
Low |
| Period |
|
|
| Quarter ended September 30, 2014 |
$0.0264 |
$0.0229 |
| Quarter ended June 30, 2014 |
$0.03 |
$0.0288 |
| Quarter ended March 31, 2014 |
$0.045 |
$0.0395 |
| Quarter ended December 31, 2013 |
$0.069 |
$0.0135 |
| Quarter ended September 30, 2013 |
$0.270 |
$0.0138 |
| Quarter ended June 30, 2013 |
$0.0447 |
$0.0275 |
| Quarter ended March 31, 2013 |
$0.126 |
$0.045 |
| Quarter ended December 31, 2012 |
$0.029 |
$0.005 |
| Quarter ended September 30, 2012 |
$0.050 |
$0.020 |
| Quarter ended June 30, 2012 |
$0.041 |
$0.0192 |
| Quarter ended March 31, 2012 |
$0.079 |
$0.035 |
Holders
As of November 20, 2014,
there were approximately 445 shareholders of record of our common
stock.
Dividends
We have never declared or
paid any cash dividends on our common stock or other securities and do not
currently anticipate paying any cash dividends in the foreseeable future. The
declaration and payment of dividends by us are subject to the discretion of our
Board of Directors and the restrictions specified in our articles of
incorporation and by applicable law. In addition, under the terms of the
Northstar loan, we are restricted from paying cash dividends to our shareholders
while the loan is outstanding. Any future determination to pay cash dividends
will depend on our results of operations, financial condition, capital
requirements, contractual restrictions and other factors deemed relevant by our
Board of Directors.
The following is
management’s discussion and analysis (“MD&A”) of certain significant factors
that have affected our financial position and operating results during the
periods included in the accompanying financial statements, as well as
information relating to the plans of our current management. This report
includes forward-looking statements. Generally, the words “believes,”
“anticipates,” “may,” “will,” “should,” “expect,” “intend,” “estimate,”
“continue,” and similar expressions or the negative thereof or comparable
terminology are intended to identify forward-looking statements. Such statements
are subject to certain risks and uncertainties, including the matters set forth
in this report or other reports or documents we file with the Securities and
Exchange Commission from time to time, which could cause actual results or
outcomes to differ materially from those projected. Undue reliance should not be
placed on these forward-looking statements which speak only as of the date
hereof. We undertake no obligation to update these forward-looking statements.
76
MANAGEMENT’S DISCUSSION
AND ANALYSIS OF FINANCIAL CONDITION
AND RESULTS OF OPERATIONS
The discussion and analysis
of the Company’s financial condition and results of operations is based upon its
financial statements, which have been prepared in accordance with generally
accepted accounting principles generally accepted in the United States (or
"GAAP"). The preparation of those financial statements requires us to make
estimates and judgments that affect the reported amount of assets and
liabilities at the date of its financial statements. Actual results may differ
from these estimates under different assumptions or conditions.
Our Ability To Continue
as a Going Concern
Our independent registered
public accounting firm has issued its report dated March 20, 2014 in connection
with the audit of our financial statements as of December 31, 2013 that included
an explanatory paragraph describing the existence of conditions that raise
substantial doubt about our ability to continue as a going concern. Our
financial statements as of December 31, 2013 have been prepared under the
assumption that we will continue as a going concern. If we are not able to
continue as a going concern, it is likely that holders of our common stock will
lose all of their investment. Our financial statements do not include any
adjustments that might result from the outcome of this uncertainty.
Overview
We are a biotechnology
company focused on the discovery, development and, subject to regulatory
approval, commercialization of autologous cell therapies for the treatment of
chronic and acute heart damage. Our lead product candidate is MyoCell, an
innovative clinical therapy designed to populate regions of scar tissue within a
patient’s heart with autologous muscle cells, or cells from a patient’s body,
for the purpose of improving cardiac function in chronic heart failure patients.
Biotechnology Product
Candidates
We are focused on the
discovery, development and, subject to regulatory approval, commercialization of
autologous cell therapies for the treatment of chronic and acute heart damage.
In our pipeline, we have multiple product Candidates for the treatment of heart
damage, including MyoCell, Myocell SDF-1 and AdipoCell. MyoCell and MyoCell
SDF-1 are clinical muscle-derived cell therapies designed to populate regions of
scar tissue within a patient’s heart with new living cells for the purpose of
improving cardiac function in chronic heart failure patients. MyoCell SDF-1 is
intended to be an improvement to MyoCell. MyoCell SDF-1 is similar to MyoCell
except that the myoblast cells to be injected for use in MyoCell SDF-1 will be
modified prior to injection by an adenovirus vector or non-viral vector so that
they will release extra quantities of the SDF-1 protein, which expresses
angiogenic factors. AdipoCell is a patient-derived cell therapy proposed for the
treatment of acute myocardial infarction, chronic heart ischemia, and lower limb
ischemia. We hope to demonstrate that these product candidates are safe and
effective complements to existing therapies for chronic and acute heart damage.
Our most recent completed
clinical trials of MyoCell are the SEISMIC Trial, a 40-patient, randomized,
multicenter, controlled, Phase II-a study conducted in Europe and the MYOHEART
Trial, a 20-patient, multicenter, Phase I dose-escalation trial conducted in the
United States.
We were approved by the
FDA, to proceed with a 330-patient, multicenter Phase II/III trial of MyoCell in
North America and Europe, or the MARVEL Trial. We completed the MyoCell
implantation procedure on the first patient in the MARVEL Trial on October 24,
2007. Thus far, 20 patients, including 6 control patients, have been treated.
Initial results for the 20 patients were released at the Heart Failure Society
of American meeting in September, 2009, showing a significant (35%) improvement
in the 6 minute walk for those patients who were treated, and no improvement for
those who received a placebo. On the basis of these results, we have applied for
and received approval from the FDA to reduce the number of additional patients
in the trial to 134, for a total of 154 patients. The SEISMIC, MYOHEART and
MARVEL Trials have been designed to test the safety and efficacy of MyoCell in
treating patients with severe, chronic damage to the heart. Upon regulatory
approval of MyoCell, we intend to generate
revenue in the United States from the sale of MyoCell cell-culturing services
for treatment of patients by qualified physicians.
77
We received approval from
the FDA in July of 2009 to conduct a Phase I safety study on 15 patients of a
combined therapy (Myocell with SDF-1), which we believe was the first approval
of a study combining gene and cell therapies. We initially commenced work on
this study, called the REGEN Trial, during the first quarter of 2010. We
suspended activity on the trial in 2010 while seeking additional funding
necessary to conduct the trial. We are seeking to secure sufficient funds to
reinitiate enrollment in the MARVEL and REGEN trials. If we successfully secure
such funds, we intend to re-engage a contract research organization, or CRO,
investigators and certain suppliers to advance such trials. We have initiated
and enrolled our first patient in the MIRROR trial in 2013. The trial is very
similar to the MARVEL trial but focusses on sites outside the US. We will
continue enrollment in the MIRROR trial once we have secured sufficient
funds.
We have completed the Phase
1 Angel Trial for AdipoCell (adipose derived stem cells). Five patients were
enrolled and treated in the second quarter of 2013. At the twelve (12) month
time point, patients demonstrated a statistically significant average
improvement in ejection fraction (EF) by echocardiogram.
At the three (3) month time
point, 100% of the patients demonstrated either improvement or stayed the same.
After three (3) months, patients showed an average absolute improvement of 3
percentage points in EF. The patients continued to improve from 3 months to 6
months with a statistically significant average absolute improvement of 10
percentage points (p=0.01) and at the 12 month follow up patients showed this
same level of improvement (p=0.01).
We have also initiated
several Institutional Review Board studies in 2013 using adipose derived stem
cells for various indications including dry macular degeneration, degenerative
disc disease, erectile dysfunction and chronic obstructive pulmonary disease. In
the second quarter of 2014, we announced the treatment of a patient in Honduras
with congestive heart failure using AdipoCell and MyoCell. This was the first
patient treated in the world using a combination of stem cells. We have begun
two clinical trials in India. The first cardiac patient has successfully been
enrolled and treated in India using AdipoCell or adipose derived stem cells. The
second trial will involve the combination of AdipoCellTM and MyoCell® or muscle
derived stem cells for congestive heart failure patients. These trials are
active and ongoing.
MyoCath Product
Candidate
The MyoCath is a deflecting
tip needle injection catheter that has a larger (25 gauge) needle to allow for
better flow rates and less leakage than systems that are 27 gauge. This larger
needle allows for thicker compositions to be injected, which helps with cell
retention in the heart.
Also, the MyoCath needle
has more fluoroscopic brightness than the normally used nitinol needle, enabling
superior visualization during the procedure. Seeing the needle well during
injections enables the physician who is operating the catheter to pinpoint
targeted areas more precisely. The MyoCath is used to inject cells into cardiac
tissue in therapeutic procedures to treat chronic heart ischemia and congestive
heart failure. Investigators in our MARVEL Trial may use either our MyoCath
catheters or Biosense Webster’s (a Johnson & Johnson company) NOGA® Cardiac
Navigation System along with the MyoStarTM injection catheter for the delivery
of MyoCell to patients enrolled in the trial. We are currently producing Myocath
catheters with a contract manufacturer on an as needed basis.
78
We conduct operations in
one business segment. We may organize our business into more discrete business
units when and if we generate significant revenue from the sale of our product
candidates. Our revenue since inception has been generated inside and outside
the United States, and the majority of our long-lived assets are located in the
United States.
Securities Purchase
Agreements
On October 7, 2014, we
entered into a securities purchase agreement (the “Purchase Agreement”) with
Magna Equities II, LLC, a New York limited liability company (“Magna”). The
Purchase Agreement provides that, upon the terms and subject to the conditions
set forth therein, Magna shall purchase from us, a senior convertible note with
an initial principal amount of $307,500 (the “Convertible Note”) for a purchase
price of $205,000 (an approximately 33.33% original issue discount). Pursuant to
the Purchase Agreement, we issued the Convertible Note to Magna. The Convertible
Note matures on August 7, 2015 and, in addition to the approximately 33.33%
original issue discount, accrues interest at the rate of 12% per annum. The
Convertible Note is convertible at any time, in whole or in part, at Magna’s
option into shares of the Company’s common stock, par value $0.01 per share (the
“Common Stock”), at a fixed conversion price of $0.01035 per share. $40,000 of
the outstanding principal amount of the Convertible Note (together with any
accrued and unpaid interest with respect to such portion of the principal
amount) shall be automatically extinguished (without any cash payment by us)
under certain conditions described in the Purchase Agreement. In connection with
the execution of the Purchase Agreement, our company and Magna also entered into
a registration rights agreement (the “Registration Rights Agreement”). Pursuant
to the Registration Rights Agreement, we agreed to file an initial registration
statement with the SEC to register the resale of the Common Stock into which the
Convertible Note may be converted,
On October 23, 2014, we
entered into a common stock purchase agreement (the “Purchase Agreement”) with
Magna Equities II, LLC, a New York limited liability company (the “Investor”).
The Purchase Agreement provides that, upon the terms and subject to the
conditions set forth therein, the Investor is committed to purchase up to
$3,000,000 (the “Total Commitment”) worth of our common stock, $0.001 par value
(the “Shares”), over the 24-month term of the Purchase Agreement. In connection
with the execution of the Purchase Agreement, on the Closing Date, our company
and the Investor also entered into a registration rights agreement. We paid to
the Investor a commitment fee for entering into the Purchase Agreement equal to
$150,000 (or 5.0% of the Total Commitment under the Purchase Agreement) in the
form of 9,109,128 restricted shares of our common stock, calculated using a per
share price of $0.016467.
Results of Operations
Overview of Year Ended December 31, 2013
Revenues
We have not generated any
material revenues from our MyoCell product candidate. The revenues we have
recognized to date are related to (i) sales of MyoCath to ACS and other parties,
(ii) fees associated with our assignment to ACS of our rights relating to the
Primary MyoCath Patent, (iii) revenues generated from paid registry trials, (iv)
revenues from the sale of our now discontinued tissue genesis system and related
supplies and (v) revenues generated for providing cell culturing services under
exclusive supply agreements. We did not generate significant revenue in 2013 and
do not expect to do so in 2014. Our revenue may vary substantially from quarter
to quarter and from year to year. We believe that period-to-period comparisons
of our results of operations are not meaningful and should not be relied upon as
indicative of our future performance. We do not expect to generate substantial
revenues until we obtain regulatory approval for and commercialize our product
candidates, which we do not expect to occur before 2015.
We recognized revenues of
$96,085 in 2013 compared to revenues of $61,109 in 2012. Our revenue in 2013 was
generated from the sale of MyoCath catheters, AdipoCell and laboratory services.
Our revenues for 2012 were generated from the sale of MyoCath Catheters and
laboratory services.
79
Cost of
Sales
Cost of sales consists of
the costs associated with the production of MyoCath.
Cost of sales was $30,831
in the twelve-month period ended December 31, 2013 compared to $1,070 in the
twelve-months ended December 31, 2012.
Research and
Development
Our research and
development expenses consist of costs incurred in identifying, developing and
testing our product candidates. These expenses consist primarily of costs
related to our clinical trials, the acquisition of intellectual property
licenses and preclinical studies. We expense research and development costs as
incurred.
Clinical trial expenses
include costs related to the culture and preparation of cells in connection with
our clinical trials, costs of contract research, costs of clinical trial
facilities, costs of delivery systems, salaries and related expenses for
clinical personnel and insurance costs. Preclinical study expenses include costs
of contract research, salaries and related expenses for personnel, costs of
development biopsies, costs of delivery systems and costs of lab supplies.
We are focused on the
development of a number of autologous cell-based therapies, and related devices,
for the treatment of heart damage. Accordingly, many of our costs are not
attributable to a specifically identified product candidate. We use our employee
and infrastructure resources across several projects, and we do not account for
internal research and development costs on a product candidate by product
candidate basis. From inception through December 31, 2013, we incurred aggregate
research and development costs of approximately $65.3 million (unaudited)
related to our product candidates We estimate that at least $48.4 million
(unaudited) of these expenses relate to our preclinical and clinical development
of MyoCell and at least $5.2 million of these expenses relate to our preclinical
and clinical development of MyoCath.
During the third quarter
2009, we received notification that approximately $630,000 in pending projects
(Indiana University, University of Florida, Northwestern University, and other
sites) was completed. As of December 31, 2013, of the $630,000, we still have an
accrual of $219,000 for the completed contracts.
Clinical trials and
preclinical studies are time-consuming and expensive. Our expenditures on
current and future preclinical and clinical development programs are subject to
many uncertainties. We generally test our products in several preclinical
studies and then conduct clinical trials for those product candidates that we
determine to be the most promising. As we obtain results from clinical trials,
we may elect to discontinue or delay trials for some product candidates in order
to focus our resources on more promising product candidates. Completion of
clinical trials may take several years or more, but the length of time generally
varies substantially according to the type, size of trial and intended use of
the product candidate.
Due to the risks inherent
in the clinical trial process, development completion dates and costs vary
significantly for each product candidate, are difficult to estimate and are
likely to change as clinical trials progress.
The cost of clinical trials
may vary significantly over the life of a project as a result of a variety of
factors, including the number of patients who participate in the clinical
trials, the number of sites included in the clinical trials, the length of time
required to enroll trial participants, the efficacy and safety profile of our
product candidates and the costs and timing of and our ability to secure
regulatory approvals.
Marketing, General
and Administrative
Our marketing, general and
administrative expenses primarily consist of the costs associated with our
general management and clinical marketing and trade programs, including, but not
limited to, salaries and related expenses for executive, administrative and
marketing personnel, rent, insurance, legal and accounting fees, consulting
fees, travel and entertainment expenses, conference costs and other clinical
marketing and trade program expenses.
80
Stock-Based
Compensation
Stock-based compensation
reflects our recognition as an expense of the value of stock options and other
equity instruments issued to our employees and non-employees over the vesting
period of the options and other equity instruments. We have granted to our
employees options to purchase shares of common stock at exercise prices equal to
the fair market value of the underlying shares of common stock at the time of
each grant, as determined by our Board of Directors, with input from management.
In valuing our common
stock, our Board of Directors considered a number of factors, including, but not
limited to:
- our financial position and historical financial performance;
- the illiquidity of our capital stock as a private company prior to our
IPO;
- arm's length sales of our common stock;
- the development status of our product candidates;
- the business risks we face;
- vesting restrictions imposed upon the equity awards;
- an evaluation and benchmark of our competitors; and
- the prospects of a liquidity event, such as our initial public offering
in February 2008.
In April 1, 2013, the Board
of Directors approved, subject to shareholder approval, the establishment of the
Bioheart 2013 Omnibus Equity Compensation Plan, or the “2013 Omnibus Plan”. The
2013 Omnibus Plan reserves up to fifty million shares of common stock for
issuance.
Effective April 1, 2013,
the Board of Directors resolved that stock options granted for the past three
years as of February 25, 2013 be repriced for employees, management and board
members, at the exercise price of the average of the last 5 trading days’
closing price as of February 25, 2013. Subsequently, this action was rescinded
and the repricing date was set for August 5, 2013.
A summary of options at
December 31, 2013 and activity during the year then ended is presented below:
|
|
|
Weighted- |
|
|
|
Average |
|
|
Weighted- |
Remaining |
|
|
Average |
Contractual |
|
Shares |
Exercise Price |
Term
(in years) |
| Options
outstanding at January 1, 2012 |
4,636,318 |
$1.20 |
8.1 |
| Granted |
3,300,000 |
$0.04 |
- |
| Exercised |
- |
- |
-- |
| Forfeited/Expired |
(82,942) |
$5.57 |
- |
| Options
outstanding at December 31, 2012 |
7,853,376 |
$0.67 |
8.2 |
| Granted |
17,400,000 |
$0.016 |
9.9 |
| Exercised |
- |
- |
- |
| Forfeited/Expired |
(1,340,433) |
$1.08 |
- |
| Options
outstanding at December 31, 2013 |
23,912,943 |
$0.15 |
9.0 |
| Options
exercisable at December 31, 2013 |
6,600,443 |
$0.48 |
8.0 |
| Available for grant at December 31, 2013 |
32,600,000 |
- |
- |
81
The following information
applies to options outstanding and exercisable at December 31, 2013:
|
Options Outstanding |
Options Exercisable |
|
|
Weighted- |
|
|
|
|
|
Average |
Weighted- |
|
Weighted- |
|
|
Remaining |
Average |
|
Average |
|
|
Contractual |
Exercise |
|
Exercise |
|
Shares |
Term |
Price |
Shares |
Price |
| $0.00 –
$0.70 |
23,290,000 |
9.2 |
$0.03 |
5,977,500 |
$0.04 |
| $0.71 –
$1.28 |
162,286 |
4.4 |
$0.80 |
162,286 |
$0.80 |
| $5.25 –
$5.67 |
435,945 |
2.0 |
$5.57 |
435,945 |
$5.57 |
| $7.69 |
24,712 |
2.6 |
$7.69 |
24,712 |
$7.69 |
|
23,912,943 |
9.0 |
$0.15 |
6,600,443 |
$0.48 |
On January 16, 2012, the
Company granted 500,000 employee stock options in connection services rendered
at the exercise price of $0.10 per share vesting over four years from the date
of issuance.
The fair values of the
employee options issued on January 16, 2012 were determined using the Black
Scholes option pricing model with the following assumptions: Dividend yield: 0%;
Volatility: 164.79% and Risk free rate: 1.89%.
On August 6, 2012, the
Company granted an aggregate 2,800,000 employee stock options in connection
services rendered at the exercise price of $0.03 per share vesting over four
years from the date of issuance.
The fair values of the
employee options issued on August 6, 2012 were determined using the Black
Scholes option pricing model with the following assumptions: Dividend yield: 0%;
Volatility: 164.91% and Risk free rate: 1.59%.
On August 5, 2013, the
Company re-priced options previously issued from 2011 through 2012 for current
employees and officers in aggregate of 4,890,000 options with previous exercise
prices from $0.03 to $0.21 per share to $0.01694 per share, all other terms
remaining unchanged. The gross change in fair value, determined using the Black
Scholes option pricing model, of $1,630 was charged to current period
operations.
On August 1, 2013, the
Company issued an aggregate 15,000,000 options to purchase the Company’s common
stock at $0.01576 per share vesting over four years. The fair value of $245,749,
determined using the Black Scholes option pricing model with the following
assumptions: Dividend yield: 0%; Volatility: 153.27% and Risk free rate: 2.74%,
of which $25,599,240 was charged to current period operations.
On September 1, 2013, the
Company issued an aggregate 2,400,000 options to purchase the Company’s common
stock at $0.01654 per share, respectively; to officers and employees,
exercisable immediately. The fair value of $37,823, determined using the Black
Scholes option pricing model with the following assumptions: Dividend yield: 0%;
Volatility: 150.98% and Risk free rate: 2.78%, was charged to current period
operations.
The fair value of all
options vesting during the year ended December 31, 2013 and 2012 of $125,515 and
$76,674, respectively, was charged to current period operations.
82
Warrants
A summary of common stock
purchase warrants at December 31, 2013 and activity during the year then ended
is presented below:
|
|
|
Weighted- |
|
|
|
Average |
|
|
Weighted- |
Remaining |
|
|
Average |
Contractual |
|
|
Exercise |
Term
(in |
|
Shares |
Price |
years) |
| Outstanding at January 1, 2012 |
32,610,075 |
$0.86 |
3.8 |
| Issued |
42,396,432 |
$0.018 |
5.78 |
| Exercised |
— |
$0.00 |
|
| Forfeited |
(933,185) |
$0.76 |
|
| Outstanding at December 31, 2012 |
74,073,322 |
$0.37 |
4.5 |
| Issued |
50,350,536 |
$0.016 |
9.2 |
| Exercised |
— |
|
|
| Expired |
(6,345,002) |
$0.38 |
|
| Outstanding at December 31, 2013 |
118,078,856 |
$0.22 |
6.3 |
| Exercisable at December 31, 2013 |
101,371,743 |
$0.13 |
5.7 |
In conjunction with the
authorized issuance of common stock, the Company granted approximately 50
million common stock purchase warrants during the year ended December 31, 2013.
The following information
applies to common stock purchase warrants outstanding and exercisable at
December 31, 2013:
|
Warrants Outstanding |
Warrants Exercisable |
|
|
Weighted- |
Weighted |
|
|
|
|
Average |
- |
|
Weighted- |
|
|
Remaining |
Average |
|
Average |
|
|
Contractual |
Exercise |
|
Exercise |
|
Shares |
Term |
Price |
Shares |
Price |
| $0.01 –
$0.50 |
111,829,723 |
6.3 |
$0.03 |
96,667,060 |
$0.03 |
| $0.52 –
$0.68 |
2,699,675 |
5.3 |
$0.58 |
2,699,675 |
$0.58 |
| $0.70 –
$1.62 |
848,176 |
6.0 |
$0.71 |
848,176 |
$0.71 |
| $5.67 –
$7.69 |
2,701,282 |
8.9 |
$7.55 |
1,156,832 |
$7.35 |
| TOTAL |
118,078,856 |
6.3 |
$0.22 |
101,371,743 |
$0.13 |
During the year ended
December 31, 2012, in connection with the sale of common stock, the Company
issued an aggregate of 22,396,432 warrants to purchase the Company’s common
stock at an exercise prices from $0.014 to $0.03 per shares exercisable in six
months and expiring three years from issuance.
On September 21, 2012, the
Company issued 5,000,000 warrants to purchase the Company’s common stock at
$0.02 per share, expiring 10 years from the date of issuance as payment of
interest.
The fair value of $119,023,
determined using the Black Scholes option pricing model with the following
assumptions: Dividend yield: 0%; Volatility: 163.45% and Risk free rate: 1.779%,
was charged to current period operations.
On October 1, 2012, the
Company issued 15,000,000 warrants to purchase the Company’s common stock at
$0.014 per share, expiring 10 years from the date of issuance as payment of
interest with certain reset provisions.
83
The fair value of $311,190,
determined using the Binomial lattice option pricing model with the following
assumptions: Dividend yield: 0%; Volatility: 155.41% and Risk free rate: 1.64%,
was charged to current period operations.
During 2013 and 2012, we
recognized stock-based compensation expense of $125,515 and $76,674,
respectively. The decrease in stock based compensation from 2012 to 2013 was
primarily due a lower valuation for stock options granted to officers,
consultants and key employees. We intend to grant stock options and other
stock-based compensation in the future and we may therefore recognize additional
stock-based compensation in connection with these future grants.
Interest
Expense
Interest expense in 2013
and 2012 primarily consists of interest incurred on the principal amount of the
Northstar loan, our former Bank of America loan, the Seaside National Bank loan,
accrued fees and interest payable to the Guarantors, the amortization of related
deferred loan costs and the amortization of the fair value of warrants issued in
connection with the loans. The deferred loan costs and fair value of warrants
issued in connection with the loans are being amortized to interest expense over
the terms of the respective loans using the effective interest method.
Results of Operations
We are a development stage
company and our MyoCell product candidate has not received regulatory approval
or generated any material revenues and is not expected to until 2015, if ever.
We have generated substantial net losses and negative cash flow from operations
since inception and anticipate incurring significant net losses and negative
cash flows from operations for the foreseeable future as we continue clinical
trials, undertake new clinical trials, apply for regulatory approvals, make
capital expenditures, add information systems and personnel, make payments
pursuant to our license agreements upon our achievement of certain milestones,
continue development of additional product candidates using our technology,
establish sales and marketing capabilities and incur the additional cost of
operating as a public company.
Comparison of Years
Ended December 31, 2013 and December 31, 2012
Revenues
We recognized revenues of
$96,085 in 2013, revenues generated from the sale of MyoCath Catheters,
AdipoCell, and laboratory services. In 2011 we recognized revenues of $61,109,
revenues generated from the sales of MyoCath Catheters and laboratory services.
Cost of
Sales
Cost of sales was $30,831
in 2013 and $1,070 in 2012.
Research and
Development
Research and development
expenses were $626,983 in 2013, an increase of $225,042 from research and
development expenses of $401,941 in 2012. The increase was primarily
attributable to an increase in the amount of available funds.
The timing and amount of
our planned research and development expenditures is dependent on our ability to
obtain additional financing.
84
Marketing, General
and Administrative
Marketing, general and
administrative expenses were $2,267,831 in 2013, an increase of $89,479 from
marketing, general and administrative expenses of $2,178,352 in 2012. The
increase in marketing, general and administrative expenses is attributable, in
part, to an increase in legal fees, salaries and insurance expenses.
Interest
Expense
Interest expense was
$1,437,883 in 2013 compared to interest expense of $1,619,653 in 2012. Non cash
interest comprised of amortization of debt discounts and warrants issued in
connection with debt totaled $597,339 in 2013 as compared to $657,081 in 2012.
In addition, in 2012, we issued warrants in connection with a forbearance
agreement with Northstar at a fair value of $430,213; in 2013 we issued
preferred stock in in connection with a forbearance agreement with Northstar at
a fair value of $274,050.
Gain on settlement of
debt
During the year ended
December 31, 2013, we settled an outstanding note payable and certain accounts
payable by issuances of common stock. As such we realized a net $1,023,439 gain
on settlement of debt during the year ended December 31, 2013, as compared to
nil for same period last year.
Gain on change in fair
value of derivative liabilities.
As of December 31, 2013, we
issued convertible notes and common stock purchase warrants with anti-dilutive
provisions that had the possibility of exceeding our common shares authorized
when considering the number of possible shares that may be issuable to satisfy
settlement provisions of these agreements after consideration of all existing
instruments that could be settled in shares. As such, we are required to
determine the fair value of this derivative and mark to market each reporting
period. For the year ended December 31, 2013, we incurred a $29,179 gain on
change in fair value of our derivative liabilities compared to a gain of
$119,795 the same period last year.
Inflation
Our opinion is that
inflation has not had, and is not expected to have, a material effect on our
operations.
Climate
Change
Our opinion is that neither
climate change, nor governmental regulations related to climate change, have
had, or are expected to have, any material effect on our operations.
Liquidity and Capital
Resources
In 2013, we continued to
finance our considerable operational cash needs with cash generated from
financing activities.
Operating
Activities
Net cash used in operating
activities was $1,913,326 in 2013 as compared to $1,095,276 of cash used in
2012. Our use of cash for operations in 2013 reflected a net loss generated
during the period of $3,143,259, adjusted for non-cash items such as stock-based
compensation of $210,666, amortization of debt discounts associated with our
convertible notes of $368,682, related party notes payable issued for services
rendered of $500,000, preferred stock issued in connection with a forbearance
agreement of $274,050, common stock issued in settlement of accounts payable of
$2,500,non-cash interest payments of $228,657 and depreciation expense of
$2,190, net with gain on settlement of debt of $1,023,439 and gain on change in
fair value of derivative liabilities of $29,179 . A net decrease in operating
assets of $129,633 and a net increase in operating liabilities of $566,173
contributed to our use of operating cash in 2013.
85
Investing
Activities
Net cash used in investing
activities was $9,425 in 2013 compared to $933 in 2012.
Financing
Activities
Net cash provided by
financing activities was $1,968,978 in 2013 as compared to $1,059,381 in 2012.
In 2013 ,we sold, in a private placement and put agreements, shares of common
stock and warrants for aggregate net cash proceeds of approximately $1,426,914.
In addition, we received an aggregate of $215,500 related party loans and
advances and $415,000 from issuance of notes payable, net of repayments of
$88,847.
Existing Capital
Resources and Future Capital Requirements
Our MyoCell product
candidate has not received regulatory approval or generated any material
revenues. We do not expect to generate any material revenues or cash from sales
of our MyoCell product candidate until commercialization of MyoCell, if ever. We
have generated substantial net losses and negative cash flow from operations
since inception and anticipate incurring significant net losses and negative
cash flows from operations for the foreseeable future. Historically, we have
relied on proceeds from the sale of our common stock and our incurrence of debt
to provide the funds necessary to conduct our research and development
activities and to meet our other cash needs.
At December 31, 2013, we
had cash and cash equivalents totaling $46,227; our working capital deficit as
of such date was $13,362,480. Our independent registered public accounting firm
has issued its report dated March 20, 2014 in connection with the audit of our
financial statements as of December 31, 2013 that included an explanatory
paragraph describing the existence of conditions that raise substantial doubt
about our ability to continue as a going concern.
As of December 31, 2013, we
had $5,784,449 in outstanding loans.
On April 30, 2012, we were
informed that Northstar assumed all amounts outstanding under the BlueCrest
loan. On April 30, 2012, Northstar agreed with us to extend until May 1, 2012
the initial payment date for any and all required monthly payments and payable
with respect to the BlueCrest loan and to waive any and all defaults and/or
events of default with respect to such payments. We are currently in discussions
regarding certain additional amendments to the terms of the loan.
On November 2, 2011, we
entered into a Standby Equity Distribution Agreement, or the SEDA, with
Greystone Capital Partners, or GCP. Pursuant to the SEDA’s terms, we may, at our
sole discretion and upon giving written notice to GCP, each an “Advance Notice”,
periodically sell shares of our common stock to GCP. For each share of Common
Stock purchased under the SEDA, GCP will pay us an amount, referred to as the
“Purchase Price”, that is eighty percent (80%) of the lowest daily volume
weighted average price of the Common Stock as quoted by Bloomberg, LP, during
the five (5) consecutive Trading Days (as such term is defined in the SEDA)
immediately subsequent to the date of the relevant Advance Notice. We are not
obligated to sell any shares of common stock to GCP but may, over the term of
the SEDA and in our sole discretion, sell to GCP that number of shares of common
stock valued at the Purchase Price from time to time in effect that equals up to
one million dollars ($1,000,000) in the aggregate. GCP's obligation to purchase
shares of Common Stock under the SEDA is subject to certain conditions,
including (i) periodic sales of shares of our common stock to GCP must be
separated by a time period equal to five Trading Days, and (ii) the amount of
any individual periodic sale designated by us in any Advance Notice shall not
exceed fifty percent (50%) of the average weekly volume of shares of our common
stock traded during the two (2) week period immediately prior to an Advance
Notice, where a “week” is five (5) consecutive Trading Days. GCP’s obligations
under the SEDA are not transferable.
During the year ended
December 31, 2013, the Company issued an aggregate of 31,052,141 shares of its
common stock in exchange for $346,914 draw down on the equity line.
86
Other than the SEDA, we
have no commitments or arrangements from third parties for any additional
financing to fund our research and development and/or other operations. We will
need to seek substantial additional financing through public and/or private
financing, which may include equity and/or debt financings, and through other
arrangements, including collaborative arrangements. We may also seek to satisfy
some of our obligations to the Guarantors through the issuance of various forms
of securities or debt on negotiated terms. However, financing and/or alternative
arrangements with the Guarantors may not be available when we need it, or may
not be available on acceptable terms. In addition, our ability to obtain
additional debt financing and/or alternative arrangements with the Guarantors
may be limited by the amount of, terms and restrictions of our then current
debt. Accordingly, we will generally be restricted from, among other things,
incurring additional indebtedness or liens, with limited exceptions. Additional
debt financing, if available, may involve restrictive covenants that limit or
further limit our operating and financial flexibility and prohibit us from
making distributions to shareholders.
If we raise additional
funds and/or secure alternative arrangements with the Guarantors by issuing
equity, equity-related or convertible securities, the economic, voting and other
rights of our existing shareholders may be diluted, and those securities may
have rights superior to those of our common stock. If we obtain additional
capital through collaborative arrangements, we may be required to relinquish
greater rights to our technologies or product candidates than we might otherwise
have or become subject to restrictive covenants that may affect our business.
Cassel Salpeter &
Co.
On November 20, 2013, we
entered into an Investment Banking Agreement with Cassel Salpeter & Co.
(“CSC”), who will act as exclusive third party financial advisor in connection
with investment banking matters. The term of the Investment Banking Agreement
shall be for a period of twenty four months unless terminated or extended in
accordance with its terms. For these services, CSC will receive
monthly fees, have received 5,207,630 ten year common
stock purchase warrants, exercisable at $.0113 and will receive applicable
consideration in the event the closing of a Mezzanine Financing consisting of
non-convertible subordinated debt and/or sale of equity securities. We will also
reimburse CSC for its reasonable out-of-pocket expenses associated with the
services provided pursuant to the Investment Banking Agreement.
Cassel Salpeter & Co.
is an independent investment banking firm that provides advice to middle-market
and emerging growth companies in the U.S. and worldwide. Together, the firm’s
professionals have more than 50 years of experience providing private and public
companies with a broad spectrum of investment banking and financial advisory
services, including: mergers and acquisitions; equity and debt capital raises;
fairness and solvency opinions; valuations; and restructurings, such as 363
sales and plans of reorganization. Co-founded by James Cassel and Scott
Salpeter, the firm provides objective, unbiased, results-focused services that
clients need to achieve their goals. Personally involved at every stage of all
engagements, the firm’s senior partners have forged relationships and completed
hundreds of transactions and assignments nationwide. The firm’s headquarters are
in Miami.
Off-Balance Sheet
Arrangements
We do not have any
off-balance sheet arrangements that have or are reasonably likely to have a
current or future effect on our financial condition, changes in financial
condition, revenues or expenses, results of operations, liquidity, capital
expenditures or capital resources that are material to investors.
Related
Parties
For the purposes of these financial statements,
parties are considered to be related if one party has the ability, directly or
indirectly, to control the party or exercise significant influence over the
party in making financial and operating decisions, or vice versa, or where the
Company and the party are subject to common control or common significant
influence. Related parties may be individuals or other entities.
87
Three and Nine Months
Ended September 30, 2014 as compared to the Three and Nine Months Ended
September 30, 2014
Revenues
We have not generated any
material revenues from our MyoCell product candidate. The revenues we have
recognized to date are related to (i) sales of MyoCath, (iii) revenues generated
from patient paid studies, (iv) revenues from the sale of our AdipoCell system
and related supplies and (v) revenues generated for providing cell culturing and
banking services. We did not generate significant revenues in 2013. Our revenue
may vary substantially from quarter to quarter and from year to year. We expect
to have steady growth of revenue as the above programs grow and expand.
We recognized revenues of
$579,536 and $1,563,864 for the three and nine months ended September 30, 2014,
respectively, compared to revenues of $31,659 and $55,980 for the three and nine
months ended September 30, 2013, respectively. Our revenue in 2014 was generated
from the sale of MyoCath catheters, AdipoCell, physician training, patient
studies and laboratory services. Our revenues for 2013 were generated from the
sale of MyoCath Catheters and laboratory services
Cost of
Sales
Cost of sales consists of
the costs associated with the production of MyoCath, laboratory supplies
necessary for laboratory services, production of AdipoCell systems and
materials, physician course materials and clinic supplies required for patient
studies.
Cost of sales was $523,222
and $889,509 in the three and nine month periods ended September 30, 2014,
respectively, compared to $-0- in the three and nine months periods ended
September 30, 2013.
Research and
Development
Our research and
development expenses consist of costs incurred in identifying, developing and
testing our product candidates. These expenses consist primarily of costs
related to our clinical trials, the acquisition of intellectual property
licenses and preclinical studies. We expense research and development costs as
incurred.
Clinical trial expenses
include costs related to the culture and preparation of cells in connection with
our clinical trials, costs of contract research, costs of clinical trial
facilities, costs of delivery systems, salaries and related expenses for
clinical personnel and insurance costs. Preclinical study expenses include costs
of contract research, salaries and related expenses for personnel, costs of
development biopsies, costs of delivery systems and costs of lab supplies.
We are focused on the
development of a number of autologous cell-based therapies, and related devices,
for the treatment of heart damage. Accordingly, many of our costs are not
attributable to a specifically identified product candidate. We use our employee
and infrastructure resources across several projects, and we do not account for
internal research and development costs on a product candidate by product
candidate basis. From inception through September 30, 2014, we incurred
aggregate research and development costs of approximately $65.3 million
(unaudited) related to our product candidates. We estimate that at least $48.4
million (unaudited) of these expenses relate to our preclinical and clinical
development of MyoCell and at least $5.2 million (unaudited) of these expenses
relate to our preclinical and clinical development of MyoCath.
During the third quarter
2009, we received notification that approximately $630,000 in pending projects
(Indiana University, University of Florida, Northwestern University, and other
sites) was completed. As of September 30, 2014 and December 31, 2013, of the
$630,000, we still have an accrual of $219,000 for the completed contracts.
Clinical trials and
preclinical studies are time-consuming and expensive. Our expenditures on
current and future preclinical and clinical development programs are subject to
many uncertainties. We generally test our products in several preclinical
studies and then conduct clinical trials for those product candidates that we
determine to be the most promising. As we obtain results from clinical trials,
we may elect to discontinue or delay trials for some product candidates in order
to focus our resources on more promising product candidates. Completion of
clinical trials may take several years or more, but the length of time generally
varies substantially according to the type, size of trial and intended use of
the product candidate.
88
Due to the risks inherent
in the clinical trial process, development completion dates and costs vary
significantly for each product candidate, are difficult to estimate and are
likely to change as clinical trials progress.
The cost of clinical trials
may vary significantly over the life of a project as a result of a variety of
factors, including the number of patients who participate in the clinical
trials, the number of sites included in the clinical trials, the length of time
required to enroll trial participants, the efficacy and safety profile of our
product candidates and the costs and timing of and our ability to secure
regulatory approvals.
Research and development
expenses were $8,581 in the three month period ended in September 30, 2014, a
decrease of $149,800 from the research and development expenses of $158,381 in
the three month period ended in September 30, 2013. The decrease was primarily
attributable to a decrease in the amount of funds allocated to our clinical
trials.
Research and development
expenses were $33,916 in the nine month period ended in September 30, 2014, a
decrease of $337,833 from the research and development expenses of $494,762 in
the nine month period ended in September 30, 2013. The decrease was primarily
attributable to a decrease in the amount of funds allocated to our clinical
trials.
The timing and amount of
our planned research and development expenditures is dependent on our ability to
obtain additional financing.
Marketing, General
and Administrative
Our marketing, general and
administrative expenses primarily consist of the costs associated with our
general management and clinical marketing and trade programs, including, but not
limited to, salaries and related expenses for executive, administrative and
marketing personnel, rent, insurance, legal and accounting fees, consulting
fees, travel and entertainment expenses, conference costs and other clinical
marketing and trade program expenses.
Stock-Based
Compensation
Stock-based compensation
reflects our recognition as an expense of the value of stock options and other
equity instruments issued to our employees and non-employees over the vesting
period of the options and other equity instruments. We have granted to our
employees options to purchase shares of common stock at exercise prices equal to
the fair market value of the underlying shares of common stock at the time of
each grant, as determined by our Board of Directors, with input from management.
The Company follows
Accounting Standards Codification subtopic 718-10. Compensation (“ASC 718-10”)
which requires that all share-based payments to both employee and non-employees
be recognized in the income statement based on their fair values.
In valuing our common
stock, our Board of Directors considered a number of factors, including, but not
limited to:
- our financial position and historical
financial performance;
- arm's length sales of our common
stock;
- the development status of our product
candidates;
- the business risks we face;
- vesting restrictions imposed upon the
equity awards; and
- an evaluation and benchmark of our
competitors; and
- prospects of a liquidity
event.
89
In April 1, 2013, the Board
of Directors approved, subject to shareholder approval, the establishment of the
Bioheart 2013 Omnibus Equity Compensation Plan, or the “2013 Omnibus Plan”. The
2013 Omnibus Plan reserves up to fifty million shares of common stock for
issuance.
Marketing, general and
administrative expenses were approximately $1,512,706 in the three month period
ended September 30, 2014, an increase of $572,352 from marketing, general and
administrative expenses of approximately $940,354 in the three month period
ended in September 30, 2013. The increase in marketing, general and
administrative expenses is attributable, in part, to stock based compensation
paid in the current period of $164,247 for services and increase in employee
compensation and service providers.
Marketing, general and
administrative expenses were approximately $3,182,397 in the nine month period
ended September 30, 2014, an increase of $1,432,121 from marketing, general and
administrative expenses of approximately $1,750,276 in the nine month period
ended in September 30, 2013. The increase in marketing, general and
administrative expenses is attributable, in part, to stock based compensation
paid in the current period of $293,342 for services and increase in employee
compensation and service providers.
Interest
Expense
Interest expense during the
three and nine months ended September 30, 2014 and 2013 primarily consists of
interest incurred on the principal amount of the Northstar loan, our former Bank
of America loan, the Seaside National Bank loan, accrued fees and interest
payable to the Guarantors, the amortization of debt discounts and non-cash
incurred relating to our issued convertible notes payable. The debt discounts
are being amortized to interest expense over the terms of the respective loans
using the effective interest method.
Critical Accounting
Policies
Our discussion and analysis
of our financial condition and results of operations is based upon our financial
statements, which have been prepared in accordance with accounting principles
generally accepted in the United States. The preparation of these financial
statements requires us to make estimates and assumptions that affect the
reported amounts of assets, liabilities, revenues and expenses. We base our
estimates on historical experience and on various other assumptions that we
believe to be reasonable under the circumstances, the results of which form the
basis for making judgments about the carrying values of assets and liabilities
that are not readily apparent from other sources. Actual results may differ from
these estimates under different assumptions or conditions. While our critical
accounting policies are described in Note 1 to our financial statements
appearing elsewhere in this report, we believe the following policies are
important to understanding and evaluating our reported financial results:
Revenue
Recognition
At the time of each
transaction, we assess whether the fee associated with the transaction is fixed
or determinable and whether or not collection is reasonably assured. The
assessment of whether the fee is fixed or determinable is based upon the payment
terms of the transaction. If a significant portion of a fee is due after our
normal payment terms or upon implementation or client acceptance, the fee is
accounted for as not being fixed or determinable and revenue is recognized as
the fees become due or after implementation or client acceptance has occurred.
Collectability is assessed based on a number of factors, including past
transaction history with the client and the creditworthiness of the client.
Revenues for test kits and
equipment sold are not recorded until test kits are delivered. The Company has
revenue sharing arrangements for the sale of goods whereby the Company is the
primary obligor, sets pricing with the customers and bears all associated credit
risks with the customers. Sales under revenue share arrangements are recorded as
gross sales and any portion shared with third parties under such arrangements
are recorded as cost of sales. Revenues from trainings are not recorded until
the completion of the training. Any cash received as a deposit for trainings are
recorded by the company as a liability.
90
Unbilled revenue is revenue
that is recognized but is not currently billable to the customer pursuant to
contractual terms. In general, such amounts become billable in accordance with
predetermined payment schedules, but recognized as revenue as services are
performed. Amounts included in unbilled revenue are expected to be collected
within one year and are included within current assets.
Research and
Development Activities
We account for research and
development costs in accordance with Accounting Standards Codification subtopic
730-10, Research and Development (“ASC 730-10”). Under ASC 730-10, all research
and development costs must be charged to expense as incurred. Accordingly,
internal research and development costs are expensed as incurred. Third-party
research and development costs are expensed when the contracted work has been
performed or as milestone results have been achieved as defined under the
applicable agreement. Company-sponsored research and development costs related
to both present and future products are expensed in the period incurred.
Derivative financial
instruments
Accounting Standards
Codification subtopic 815-40, Derivatives and Hedging, Contracts in Entity’s own
Equity (“ASC 815-40”) became effective for the Company on October 1, 2009. The
Company has identified the embedded derivatives related to the issued Notes and
anti-dilutive warrants. These embedded derivatives included in our debt contain
certain conversion features and reset provision. The accounting treatment of
derivative financial instruments requires that the Company record fair value of
the derivatives as of the inception date of Asher Notes and to fair value as of
each subsequent reporting date.
Inflation
Our opinion is that
inflation has not had, and is not expected to have, a material effect on our
operations.
Climate
Change
Our opinion is that neither
climate change, nor governmental regulations related to climate change, have
had, or are expected to have, any material effect on our operations.
Concentrations of Credit
Risk
As of September 30, 2014,
two customers represented 24% and 18% of the Company’s accounts receivable.
As of December 31, 2013,
three customers represented 20%, 20% and 36% (total 76%) of the Company’s
accounts receivable.
Liquidity and Capital
Resources
In the three months ended
September 30, 2014, we continued to finance our considerable operational cash
needs with cash generated from financing activities.
Operating
Activities
Net cash used in operating
activities was $747,184 in the nine month period ended September 30, 2014 as
compared to $1,568,727 of cash used in the nine month period ended in September
30, 2013.
Our use of cash for
operations in the nine months ended September 30, 2014 reflected a net loss
generated during the period of $1,247,199, adjusted for non-cash items such as
stock-based compensation of $407,616, fair value of warrants issued of $136,837,
depreciation of $3,745, amortization of debt discounts of $343,592, related
party notes payable issued for services of $800,000 and non-cash interest paid
of $385,501, loss on change in fair value of derivative liabilities of $61,339,
net gain on settlement of debt of $2,272,283. In addition we had a net increase
in operating assets of $186,306 and an increase in accrued expenses of $246,486
and in accounts payable of $628,684.
91
Our use of cash for
operations in the nine months ended September 30, 2013 reflected a net loss
generated during the period of approximately $2.4 million, adjusted for non-cash
items such as stock-based compensation of $102,674, depreciation of $1,328,
amortization of debt discounts of $295,750 and non-cash interest paid of
$168,350, net with gain on settlement of debt of $1,004,224 and gain on change
in fair value of derivative liabilities of $39,885. In addition we had a net
decrease in operating assets of $96,933 and an increase in accrued expenses of
$485,518 and a decrease in accounts payable of $76,240.
Investing
Activities
Net cash used in investing
activities was $8,121 for the nine months ended September 30, 2014 were to
acquire office equipment as compared to nil cash used for the same period of
2013.
Financing
Activities
Net cash provided by
financing activities was an aggregate of $755,670 in the nine month period ended
September 30, 2014 as compared to $1,575,411 in the nine month period ended in
September 30, 2013. In the nine month period ended September 30, 2014 we sold,
in private placements, shares of common stock and common stock purchase warrants
for aggregate net cash proceeds of $428,251, received proceeds from issuance of
note payable of $388,000, and proceeds from warrant exercise of $136,000, net
related party advances of $26,759, net with repayments of related party notes
payable of $223,340.
Existing Capital
Resources and Future Capital Requirements
Our MyoCell product
candidate has not received regulatory approval or generated any material
revenues. We do not expect to generate any material revenues or cash from sales
of our MyoCell product candidate until commercialization of MyoCell, if ever. We
have generated substantial net losses and negative cash flow from operations
since inception and anticipate incurring significant net losses and negative
cash flows from operations for the foreseeable future. Historically, we have
relied on proceeds from the sale of our common stock and our incurrence of debt
to provide the funds necessary to conduct our research and development
activities and to meet our other cash needs.
At September 30, 2014, we
had cash and cash equivalents totaling $46,592. However our working capital
deficit as of such date was approximately $10 million. Our independent
registered public accounting firm has issued its report dated March 24, 2014 in
connection with the audit of our financial statements as of December 31, 2013
that included an explanatory paragraph describing the existence of conditions
that raise substantial doubt about our ability to continue as a going concern
and Note 2 of our unaudited financial statement for the quarter ended September
30, 2014 addresses the issue of our ability to continue as a going concern.
Off-Balance Sheet
Arrangements
We do not have any
off-balance sheet arrangements that have or are reasonably likely to have a
current or future effect on our financial condition, changes in financial
condition, revenues or expenses, results of operations, liquidity, capital
expenditures or capital resources that are material to investors.
QUANTITATIVE AND
QUALITATIVE DISCLOSURES ABOUT MARKET RISK
As a smaller reporting
company, the Company is not required to provide this disclosure.
92
DIRECTORS AND EXECUTIVE
OFFICERS
Executive Officers and
Directors
Set forth below is
information regarding our executive officers and directors as of November 21,
2104.
| Mike
Tomas |
48 |
Director, President and Chief Executive Officer, Chief
Financial |
|
|
Officer |
| William
P. Murphy, Jr., M.D. |
90 |
Director, Chairman of the Board |
| Mark P.
Borman |
59 |
Director |
| Charles
A. Hart |
53 |
Director |
| Sam
Ahn |
59 |
Director |
| Kristin
Comella |
37 |
Director, Chief Scientific Officer |
| Sheldon
T. Anderson |
63 |
Director |
Our Bylaws provide that we
shall have that number of directors determined by the majority vote of the board
of directors. Currently we have seven directors. Each director will serve until
our next annual shareholder meeting. Directors are elected for one-year terms.
Our Board of Directors elects our officers at the regular annual meeting of the
Board of Directors following the annual meeting of shareholders. Vacancies may
be filled by a majority vote of the remaining directors then in office. Our
directors and executive officers are as follows:
Executive Officers
and Directors
Mike Tomas.
Mr. Mike Tomas was appointed
President and Chief Executive Officer and a member of our Board of Directors on
June 19, 2010. Mike Tomas was appointed as the Company’s President and Chief
Executive Officer, and as a director on June 19, 2010. Mr. Tomas has been
President for the past nine years of The ASTRI Group, an early stage private
equity investment company in Florida with an investment in Bioheart since 2001.
In 2003, he joined Bioheart’s Board of Directors as the independent
representative of The ASTRI Group. ASTRI provides capital, business development
and strategic marketing support to emerging private companies. Mr. Tomas will
continue to serve as President of The ASTRI Group. Previously from 1983 to 2001,
Mr. Tomas held ascending executive positions including Chief Marketing Officer
at Avantel, a $1 billion dollar joint venture with MCI. Upon retiring from MCI
and WorldCom, Tomas joined other ex-MCI executives and helped raise $40M in
venture capital to form Ineto, an integrated customer communications software
solution that was successfully sold in 2001.
Today Mr. Tomas sits on the
boards of Perimeter Internetworking (SaaS providing secure transfer of
information for medical and financial institutions), Avisena (revenue cycle
management for medical practices) and Total Home Health (Medicare-certified home
care provider). Mr. Tomas is also the current chairman of the Global
Entrepreneurship Center at Florida International University and a founding
coach/mentor at the University of Miami’s Launch Pad at the Toppel Center. Mr.
Tomas holds a Masters of Business Administration from the University of Miami
and a Bachelor’s degree from Florida International University.
William P. Murphy, Jr.,
M.D. Dr. Murphy has served as a
member of our Board of Directors since June 2003. Dr. Murphy founded Small
Parts, Inc., a supplier of high quality mechanical components for design
engineers, in 1964 and served as its Chairman until his retirement in April
2005. Small Parts, Inc. was acquired by Amazon.com, Inc. in March 2005. From
October 1999 until October 2004, Dr. Murphy served as the Chairman and Chief
Executive Officer of Hyperion, Inc., a medical diagnosis company which had an
involuntary bankruptcy filed against it in December 2003. Dr. Murphy is the
founder of Cordis Corporation (now Cordis Johnson & Johnson) which he led as
President, Chairman and Chief Executive Officer at various times during his 28
years at Cordis until his retirement in October 1985. Cordis Johnson &
Johnson is a leading firm in cardiovascular instrumentation. Dr. Murphy received
an M.D. in 1947 from the University of Illinois and a B.S in pre-medicine from
Harvard College in 1946. He also
studied physiologic instrumentation at Massachusetts Institute of Technology, or
MIT. After a two year rotating internship at St. Francis Hospital in Honolulu,
he became a Research Fellow in Medicine at the Peter Bent Brigham Hospital in
Boston where he was the dialysis engineer on the first clinical dialysis team in
the United States. He continued as an Instructor in Medicine and then a research
associate in Medicine at Harvard Medical School. Dr. Murphy is the author of
numerous papers and owns 17 patents. He is the recipient of a number of honors,
including the prestigious Lemelson-MIT Lifetime Achievement Award, the MIT
Corporate Leadership Award, the Distinguished Service Award from North American
Society of Pacing and Electrophysiology, and the Jay Malina Award from the
Beacon Council of Miami, Florida. He is also a member of the Inventors Hall of
Fame
93
Mark P.
Borman. Mr. Borman has served as
a member of the Company’s Board of Directors since May 2009. He is a seasoned
financial officer with more than 30 years of broad-based financial and investor
relations experience. Mr. Borman brings small-company entrepreneurial passion
and larger-company disciplines. In addition to the valuable experience he gained
working with entrepreneurs and their startups from 2009 to present, Mr. Borman
has experience with global, NASDAQ- and NYSE-listed companies in various
executive and financial roles. He most recently served as Corporate Officer,
Treasurer and Vice President of Investor Relations with ADC Telecommunications.
During his career, Mr. Borman has held positions with General Instrument
Corporation, First Chicago Corporation, FMC Corporation, Price Waterhouse, and
KPMG. Mr. Borman received his B.A. in Accounting from Michigan State University
and his M.B.A. from the University of Chicago Graduate School of Business. He is
a Certified Public Accountant and Chartered Financial Analyst and has experience
as an advisor, board member, faculty, speaker, and mentor.
Charles A.
Hart. Mr. Hart has served as a
member of our Board of Directors since May 2009. Mr. Hart has more than 20 years
of entrepreneurial experience. Mr. Hart founded Hart Masonry, Inc. in 1986 and
has served as its President since then. He is also the Founder and President of
Wildridge Enterprises. Mr. Hart is a member of the Board of Directors for Eagle
Street Properties LLP.
Sam Ahn.
Dr. Ahn previously served as a
member of the Company's Board of Directors from January 2001 thru September
2008. Dr Ahn was one of the early pioneers in developing the field of
endovascular surgery by coordinating and leading the first endovascular training
courses in the US and Europe as well as developing some of the endovascular
devices and techniques currently in clinical use today. He is a former Professor
of Surgery in the Division of Vascular Surgery at UCLA, where he was also the
Director of the Endovascular Surgery Program. In 2006 Dr. Ahn founded Vascular
Management Associates, Inc., a consulting and management firm that sets up
outpatient endovascular centers across the US. VMA has set up 8 such sites to
date and is on track to set up two more this year. In 2008, he co-founded
Wright-Ahn Technology, LLC, to develop and commercialize endovascular devices.
In 2009, he co-founded MediBank International, LLC, a global healthcare IT
Company. Dr. Ahn graduated from the University of Texas, Southwestern Medical
School in Dallas, and received his general and vascular surgical residency
training at UCLA. He also earned his MBA from the UCLA Anderson School of
Management in August, 2004. Dr. Ahn sits on five vascular journal editorial
boards, and has published over 120 peer-reviewed manuscripts, 50 book chapters,
and five textbooks, including the first and definitive textbook on Endovascular
Surgery. During the past eighteen years he has consulted for over 50 biomedical
companies, both new and established, and has authored over 15 patents.
Kristin Comella.
Ms. Comella was appointed Chief
Scientific Officer in September 2010. Ms. Comella has served as our Vice
President of R&D and Corporate Development since December 2008 and has
played a major role in managing our product development, manufacturing and
quality systems since joining Bioheart in 2004. Ms. Comella has 15 years of
industry experience with expertise in regenerative medicine, training and
education, research and product development, and currently serves on multiple
advisory boards in the stem cell arena. Ms. Comella has many years of cell
culturing experience including building and managing the stem cell laboratory at
Tulane University's Center for Gene Therapy and developing stem cell therapies
for osteoarthritis at Osiris Therapeutics. Ms. Comella holds an M.S. in Chemical
Engineering from The Ohio State University and a B.S. in Chemical Engineering
from the University of South Florida. On March 12, 2013, Kristin Comella was
appointed to serve as a member of our Board of Directors.
94
Sheldon T. Anderson.
Mr. Anderson is Chairman of the
Florida Advisory Board of Northern Trust Corporation. From 1992 through December
31, 2012, Mr. Anderson served in a variety of executive capacities with Northern
Trust Corporation, including his most recent position as Chairman and Chief
executive Officer Southeast Region of Northern Trust Corporation. Mr. Anderson
is the Chair-elect of the Beacon Council, Miami-Dade County's economic
development agency. He is a Board member of the Miami-Dade College Foundation,
Inc.; Museum of Contemporary Art (MOCA); the New World Symphony; Baptist Health
Systems Governing Board and Carrollton School of the Sacred Heart. He is Past
Chair and a member of the Advisory Council of the United Way of Miami-Dade
County. Anderson is President of the Board of Cleveland Orchestra Miami / Miami
Music Association and also serves on the Advisory Board of the University of
Miami School of Law for Ethics& Public Service. He is a member of the Orange
Bowl Committee and the President's Council of Florida International University.
A Miami native, Sheldon holds a degree in International Studies from Ohio State
University.
Family
Relationships
There are no family
relationships among our executive officers and directors.
Section 16(a) Beneficial Ownership Reporting Compliance
Section 16(a) of the
Exchange Act requires our directors and executive officers, and persons who own
more than ten percent (10%) of our outstanding Common Stock, or the Reporting
Persons, to file with the SEC initial reports of ownership on Form 3 and reports
of changes in ownership of Common Stock on Forms 4 or 5. Such persons are
required by SEC regulation to furnish us with copies of all such reports they
file. Based solely on a review of Forms 3 and 4 furnished to us by the Reporting
Persons or prepared on behalf of the Reporting Persons by the Company and on
written representations from certain Reporting Persons that no Forms 5 was
required, the Company believes that the Reporting Persons have complied with
reporting requirements applicable to them.
Conflicts of Interest
Members of our management
are associated with other firms involved in a range of business activities.
Consequently, there are potential inherent conflicts of interest in their acting
as officers and directors of our company. Although the officers and directors
are engaged in other business activities, we anticipate they will devote an
important amount of time to our affairs. Northstar Biotechnology Group, LLC
(“Northstar”), a limited liability company that is a shareholder and has other
contractual relations with us is owned partly by certain directors and existing
shareholders of the Company, including Dr. William P. Murphy Jr., Dr. Samuel Ahn
and Charles Hart
Our officers and directors
are now and may in the future become shareholders, officers or directors of
other companies, which may be formed for the purpose of engaging in business
activities similar to ours. Accordingly, additional direct conflicts of interest
may arise in the future with respect to such individuals acting on behalf of us
or other entities. Moreover, additional conflicts of interest may arise with
respect to opportunities which come to the attention of such individuals in the
performance of their duties or otherwise. Currently, we do not have a right of
first refusal pertaining to opportunities that come to their attention and may
relate to our business operations.
Our officers and directors
are, so long as they are our officers or directors, subject to the restriction
that all opportunities contemplated by our plan of operation which come to their
attention, either in the performance of their duties or in any other manner,
will be considered opportunities of, and be made available to us and the
companies that they are affiliated with on an equal basis. A breach of this
requirement will be a breach of the fiduciary duties of the officer or director.
If we or the companies with which the officers and directors are affiliated both
desire to take advantage of an opportunity, then said officers and directors
would abstain from negotiating and voting upon the opportunity. However, all
directors may still individually take advantage of opportunities if we should
decline to do so. Except as set forth above, we have not adopted any other
conflict of interest policy with respect to such transactions.
95
Involvement in
Certain Legal Proceedings
None of the following
events have occurred during the past ten years and are material to an evaluation
of the ability or integrity of any director or officer of the Company:
| |
1. |
A petition under the Federal bankruptcy laws or any state
insolvency law was filed by or against, or a receiver, fiscal agent or
similar officer was appointed by a court for the business or property of
such person, or any partnership in which he was a general partner at or
within two years before the time of such filing, or any corporation or
business association of which he was an executive officer at or within two
years before the time of such filing; |
|
2. |
Such person was convicted in a criminal proceeding or is a named
subject of a pending criminal proceeding (excluding traffic violations and
other minor offenses); |
|
3. |
Such person was the subject of any order, judgment, or decree, not
subsequently reversed, suspended or vacated, of any court of competent
jurisdiction, permanently or temporarily enjoining him from, or otherwise
limiting, the following activities: |
|
|
a. |
|
Acting as
a futures commission merchant, introducing broker, commodity trading
advisor, commodity pool operator, floor broker, leverage transaction
merchant, any other person regulated by the Commodity Futures Trading
Commission, or an associated person of any of the foregoing, or as an
investment adviser, underwriter, broker or dealer in securities, or as an
affiliated person, director or employee of any investment company, bank,
savings and loan association or insurance company, or engaging in or
continuing any conduct or practice in connection with such
activity; |
|
|
b. |
|
Engaging
in any type of business practice; or |
|
|
c. |
|
Engaging
in any activity in connection with the purchase or sale of any security or
commodity or in connection with any violation of Federal or State
securities laws or Federal commodities laws; |
|
4. |
Such person was the subject of any order, judgment or decree, not
subsequently reversed, suspended or vacated, of any Federal or State
authority barring, suspending or otherwise limiting for more than 60 days
the right of such person to engage in any activity described in paragraph
(f)(3)(i) of this section, or to be associated with persons engaged in any
such activity; |
|
5. |
Such person was found by a court of competent jurisdiction in a
civil action or by the Commission to have violated any Federal or State
securities law, and the judgment in such civil action or finding by the
Commission has not been subsequently reversed, suspended, or
vacated; |
|
6. |
Such person was found by a court of competent jurisdiction in a
civil action or by the Commodity Futures Trading Commission to have
violated any Federal commodities law, and the judgment in such civil
action or finding by the Commodity Futures Trading Commission has not been
subsequently reversed, suspended or vacated; |
|
7. |
Such person was the subject of, or a party to, any Federal or State
judicial or administrative order, judgment, decree, or finding, not
subsequently reversed, suspended or vacated, relating to an alleged
violation of: |
|
|
a. |
|
Any
Federal or State securities or commodities law or regulation;
or |
|
|
b. |
|
Any law or
regulation respecting financial institutions or insurance companies
including, but not limited to, a temporary or permanent injunction, order
of disgorgement or restitution, civil money penalty or temporary or
permanent cease-and-desist order, or removal or prohibition order;
or |
|
|
c. |
|
Any law or
regulation prohibiting mail or wire fraud or fraud in connection with any
business entity; or |
|
8. |
Such person was the subject of, or a party to, any sanction or
order, not subsequently reversed, suspended or vacated, of any
self-regulatory organization (as defined in Section 3(a)(26) of the
Exchange Act (15 U.S.C. 78c(a)(26))), any registered entity (as defined in
Section 1(a)(29) of the Commodity Exchange Act (7 U.S.C. 1(a)(29)), or any
equivalent exchange, association, entity or organization that has
disciplinary authority over its members or persons associated with a
member. |
96
Code of
Ethics
As part of our system of
corporate governance, our Board of Directors has adopted a code of ethics that
is specifically applicable to our Chief Executive Officer and senior financial
officers. This Code of Ethics for Senior Financial Officers, as well as our Code
of Business Conduct and Ethics, applicable to all directors, officers and
employees, are available on our web site at
http://www.bioheartinc.com/investorrelations.html. If we make substantive
amendments to the Code of Ethics for Senior Financial Officers or the Code of
Business Conduct and Ethics or grant any waiver, including any implicit waiver,
we will disclose the nature of such amendment or waiver on our website or in a
report on Form 8-K within four days of such amendment or waiver.
Shareholder
Recommendations for Board Nominees
There have been no material
changes to the procedures by which security holders may recommend nominees to
the Company’s board of directors from the procedures described in our Annual
Report on Form 10-K for the year ended December 31, 2013.
Audit
Committee
The Board of Directors has
a separately-designated standing Audit Committee established in accordance with
Section 3(a)(58)(A) of the Exchange Act. The members of our Audit Committee are
Mr. Borman, who serves as Chairperson of the Audit Committee, Dr. Murphy and Mr.
Hart. Our Board of Directors has determined that Mr. Borman qualifies as a
“financial expert” as that term is defined in the rules of the SEC implementing
requirements of the Sarbanes-Oxley Act of 2002.
Executive
Compensation.
Summary Compensation
Table
The following table sets
forth, for the fiscal years ended December 31, 2013 and 2012, the aggregate
compensation awarded to, earned by or paid to our Chief Executive Officer and
our two most highly compensated officers (other than the Chief Executive
Officer), who were serving as executive officers as of December 31, 2013, or the
Named Executive Officers.
|
|
|
|
|
|
|
Change in |
|
|
|
|
|
|
|
|
Non- |
Pension
Value |
|
|
|
|
|
|
|
|
Equity |
and Non- |
|
|
|
|
|
|
|
|
Incentive |
Qualified |
All Other |
|
|
|
|
|
Stock |
|
Plan |
Deferred |
Compen- |
|
| Name and |
Fiscal |
Salary |
Bonus |
Awards |
Option |
Compen- |
Compensation |
sation |
Total |
| Principal Position |
Year |
($) |
($) |
($) |
Awards ($) |
sation ($) |
Earnings ($) |
($) |
($) |
| Mike Tomas (5) |
2013 |
$391,667 |
$375,000
(1) |
$ — |
$170,137 (2)(3) |
|
|
|
$936,804 |
| CEO, President, |
|
|
|
|
|
|
|
|
|
| CFO and |
|
|
|
|
|
|
|
|
|
| Director |
2012 |
247,585 |
— |
— |
100,000
(4) |
— |
— |
— |
347,585 |
| |
|
|
|
|
|
|
|
|
|
| Kristin Comella
(6) |
2013 |
159,167 |
125,000
(1) |
— |
81,916
(2) |
|
|
|
366,083 |
| Chief Science |
|
|
|
|
|
|
|
|
|
| Officer
and Director |
2012 |
105,671 |
— |
— |
— |
— |
— |
— |
105,671 |
| (1) |
On August
1, 2013, Mr. Tomas and Ms. Comella received $375,000 and $125,000,
respectively, promissory notes for bonuses awarded. The promissory notes
bear 5% interest per annum, unsecured and are due on demand. |
| (2) |
On August
1, 2013, Mr. Tomas and Ms. Comella were granted 10,000,000 and 5,000,000,
respectively, options to purchase the Company’s common stock at $0.01576
per share for ten years, vesting annually over four years. |
| (3) |
On
September 1, 2013, Mr. Tomas, as a member of the Company’s Board of
Directors, was granted 400,000 options to purchase the Company’s common
stock at $0.01654 for ten years, vesting
immediately. |
97
| (4) |
On January
16, 2012, Mr. Tomas was granted 500,000 options to purchase the Company’s
common stock at $0.10 per share for ten years, vesting annually over four
years. On August 6, 2012, Mr. Tomas was granted 2,000,000 options to
purchase the Company’s common stock at $0.03 per share for ten years
vesting annually over four years. |
| (5) |
Mr. Tomas
was appointed Chief Executive Officer and President on June 18,
2010. |
| (6) |
Ms.
Comella was appointed Chief Scientific Officer on September 24,
2010. |
Our Stock Option
Plans
In December 1999, our Board
of Directors and shareholders adopted our 1999 Officers and Employees Stock
Option Plan, or the Employee Plan, and the 1999 Directors and Consultants Stock
Option Plan, or the Director Plan. The Employee Plan and the Director Plan are
collectively referred to herein as “the Plans”. The Plans are administered by
the Board of Directors and the Compensation Committee. The objectives of the
Plans include attracting and retaining key personnel by encouraging stock
ownership in the Company by such persons. In February 2010 the Directors &
Consultants Plan was amended to extend the termination date of the Plan to
December 1, 2011.
In July 2008, the Board of
Directors approved, subject to shareholder approval, the establishment of the
Bioheart Omnibus Equity Compensation Plan, or the “Omnibus Plan. The
establishment of the Omnibus Plan was approved by the Company’s shareholders at
the Annual Meeting of Shareholders held on July 30, 2008. Pursuant to the
Omnibus Plan, the Company may grant restricted stock, incentive stock options,
non-statutory stock options, stock appreciation rights, deferred stock, stock
awards, performance shares, and other stock-based awards consisting of cash,
restricted stock or unrestricted stock in various combinations to the Company’s
employees, directors and consultants. 5,000,000 shares of common stock have been
reserved for issuance under the Omnibus Plan.
In fiscal, the Board of
Directors approved, subject to shareholder approval 2013 Omnibus Equity
Compensation Plan or the “2013 Omnibus Plan. Pursuant to the 2013 Omnibus Plan,
the Company may grant restricted stock, incentive stock options, non-statutory
stock options, stock appreciation rights, deferred stock, stock awards,
performance shares, and other stock-based awards consisting of cash, restricted
stock or unrestricted stock in various combinations to the Company’s employees,
directors and consultants. 50,000,000 shares of common stock have been reserved
for issuance under the 2013 Omnibus Plan.
Employment
Agreements
As of this time, there are
no employment agreements with any named executive officer.
Outstanding Equity
Awards at Fiscal Year End
The following table sets
forth outstanding equity awards held by our Named Executive Officers as of
December 31, 2013:
|
Number of Securities Underlying |
Option |
|
|
Unexercised Options and Warrants |
Exercise Price |
Option Expiration |
| Name |
Exercisable (#) |
Unexercisable (#) |
($/per share) |
Date |
| Mike
Tomas |
250,000 |
250,000 |
0.0169 |
1/16/2022 |
|
750,000 |
1,250,000 |
0.0169 |
8/6/2022 |
| Mike
Tomas |
125,000 |
375,000 |
0.50 |
06/18/2020 |
|
250,000 |
250,000 |
0.0169 |
08/12/2021 |
|
— |
10,000,000 |
0.0158 |
8/1/2023 |
|
400,000 |
— |
0.0165 |
9/1/2023 |
| Kristin
Comella |
187,500 |
312,500 |
0.0169 |
8/6/2022 |
|
12,356 |
— |
0.71 |
08/31/2014 |
|
24,712 |
— |
0.71 |
02/19/2015 |
|
618 |
— |
0.71 |
12/30/2015 |
|
9,267 |
— |
0.71 |
04/18/2016 |
|
6,178 |
— |
0.71 |
01/01/2017 |
|
6,500 |
— |
0.71 |
10/16/2017 |
|
19,700 |
— |
0.71 |
01/09/2019 |
|
2,955 |
— |
0.74 |
03/13/2019 |
|
30,000 |
— |
0.85 |
05/28/2019 |
|
125,000 |
125,000 |
0.0158 |
08/21/2021 |
|
— |
5,000,000 |
0.0158 |
08/1/2023 |
98
Outstanding Stock Awards
at Year End
The outstanding equity awards as at December 31,
2013 are as follows:
|
Stock awards |
|
|
|
Equity incentive
plan |
Equity incentive plan |
|
Number of |
Market value
of |
awards: |
awards: |
|
shares |
shares or
units |
number of
unearned |
market or payout value of |
|
or units of
stock |
of |
shares, |
unearned shares, units or |
|
that have
not |
stock that
have |
units or other rights
that |
other |
|
vested |
not |
have |
rights that have not vested |
| Name |
(#) |
vested (#) |
not
vested (#) |
($) |
| Mike Tomas, NEO |
1,750,000 |
0.0169 per share |
0 |
0 |
| Kristin Comella, |
312,500 |
0.0169 per share |
0 |
0 |
| NEO |
|
|
|
|
| Kristin Comella, |
125,000 |
0.0158 per share |
0 |
0 |
| NEO |
|
|
|
|
| Mike
Tomas, NEO |
1,750,000 |
0.0169
per share |
0 |
0 |
| Mike
Tomas, NEO |
375,000 |
0.50
per share |
0 |
0 |
| Mike
Tomas, NEO |
10,000,000 |
0.0158
per share |
0 |
0 |
| Kristin Comella, |
5,000,000 |
0.0158 per share |
0 |
0 |
| NEO |
|
|
|
|
Options Exercises and
Stocks Vested
Options exercised and stocks vested as at December
31, 2013 are as follows:
|
Option awards |
Stock awards |
|
Number of
Shares |
|
Number of |
Shares Value |
|
Acquired |
Value Realized
on |
Acquired on Vesting |
Realized
on Investing |
| Name |
on
Exercise (#) |
Exercise ($) |
(#) |
($) |
| Mike
Tomas, NEO |
0 |
0 |
0 |
0 |
| Kristin Comella, |
0 |
0 |
0 |
0 |
| NEO |
|
|
|
|
99
Grants of Plan-Based
Awards
Grants of plan-based awards as at December 31,
2013 are as follows:
|
|
|
|
All
other |
All
other |
|
|
|
|
|
|
stock awards: |
option |
Exercise |
Grant |
|
|
Estimated future
payouts |
Estimated future
payouts |
Number of |
awards: |
or base |
date
fair |
|
|
under non-equity
incentive |
under equity
incentive plan |
shares |
Number of |
price of |
value
of |
|
|
plan awards |
awards |
of stock |
securities |
option |
stock
and |
|
Grant |
Threshold |
Target |
Maximum |
Threshold |
Target |
Maximum |
or units |
underlying |
awards |
option |
| Name |
date |
($) |
($) |
($) |
(#) |
(#) |
(#) |
options
(#) |
(#) |
($/Sh) |
awards |
| Mike |
n/a |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
| Tomas, |
|
|
|
|
|
|
|
|
|
|
|
| NEO |
|
|
|
|
|
|
|
|
|
|
|
| Kristin |
n/a |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
| Comella |
|
|
|
|
|
|
|
|
|
|
|
| ,
NEO |
|
|
|
|
|
|
|
|
|
|
|
Reference – Grant Date -
n/a = not applicable.
Non-Qualified Deferred
Compensation
As at December 31, 2013 the Company had no
formalized deferred compensation plan.
|
Executive |
Registrant |
Aggregate |
Aggregate |
Aggregate |
|
contributions |
contributions |
in earnings in
last |
withdrawals/ |
balance at last |
| Name |
in
last FY ($) |
last
FY ($) |
FY
($) |
distributions ($) |
FYE
($) |
| Mike
Tomas, NEO |
0 |
0 |
0 |
0 |
0 |
| Kristin
Comella, NEO |
0 |
0 |
0 |
0 |
0 |
Golden Parachute
Compensation
As at December 31, 2013, the Company had no
arrangements in place relating to the termination of employees.
|
|
|
|
|
Tax |
|
|
|
Cash |
Equity |
Pension/NQDC
|
Perquisites/benefits |
reimbursement |
Other |
Total |
| Name |
($) |
($) |
($) |
($) |
($) |
($) |
($) |
| Mike
Tomas, NEO |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
| Kristin
Comella, NEO |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
Compensation of
Directors
Directors who provide services to the Company in
other capacities have been previously reported under “Summary Compensation”. The
following table summarizes compensation paid to or earned by our directors who
are not Named Executive Officers for their service as directors of our company
during the fiscal year ended December 31, 2013.
100
|
|
|
|
|
Change in |
|
|
|
|
|
|
|
Pension Value |
|
|
|
Fees |
|
|
|
and |
|
|
|
Earned |
|
|
Non-Equity |
Nonqualified |
All other |
|
|
or Paid |
Stock |
|
Incentive Plan |
Deferred |
Comp- |
|
|
in Cash |
Awards |
Option Awards
(1) |
Compensation |
Compensation |
ensation |
Total(1) |
| Name |
($) |
($) |
($) |
($) |
Earnings ($) |
($) |
($) |
| Mike
Tomas, Director |
0 |
0 |
$6,304 |
0 |
0 |
0 |
$6,304 |
| Charles A. Hart, |
0 |
0 |
$6,304 |
0 |
0 |
0 |
$6,304 |
| Director |
|
|
|
|
|
|
|
| William P. Murphy Jr., |
0 |
0 |
$6,304 |
0 |
0 |
0 |
$6,304 |
| Director |
|
|
|
|
|
|
|
| Samuel S. Ahn, |
0 |
0 |
$6,304 |
0 |
0 |
0 |
$6,304 |
| Director |
|
|
|
|
|
|
|
| Mark
Borman, Director |
0 |
0 |
$6,304 |
0 |
0 |
0 |
$6,304 |
| (1) |
|
The
values in the “Option Awards” and included within the “Total” columns
above do not represent a cash payment of any kind. Rather these values
represent the calculated Black-Scholes theoretical value of granted
options. It is important to note that these granted options may or may not
ever be exercised. Whether granted options are exercised or not will be
based primarily, but not singularly, on the Company’s future stock price
and whether the granted options become “in-the-money”. If these granted
options are unexercised and expire, the cash value or benefit to the above
noted individuals is $nil. |
Pension
Benefits
As of December 31, 2013, the Company had no
pension or retirement plans.
|
|
Number of years |
Present value of |
|
|
|
credited service |
accumulated benefit |
Payments during last |
| Name |
Plan name |
(#) |
($) |
fiscal year ($) |
| Mike
Tomas, NEO |
not
applicable |
0 |
0 |
0 |
| Kristin Comella, NEO |
not
applicable |
0 |
0 |
0 |
Equity Compensation Plan
Information
The following table sets forth information as of
December 31, 2013 for all compensation plans under the Company’s Stock Option
Plan
|
|
|
Additional |
|
|
|
|
No. of |
|
Consideration |
|
|
|
|
Shares of Common |
|
to be Received |
|
|
|
|
Stock Underlying |
|
Upon Exercise |
|
|
|
|
Unexercised |
|
or Material |
|
|
|
|
Common Stock |
|
Conditions |
Option |
Value |
|
|
Purchase Options |
Date of |
Required to |
Exercise |
Realized if |
Option |
| Name |
Exercisable (#) |
Grant |
Exercise |
Price ($) |
Exercised ($) |
Expiration Date |
| Mike
Tomas, NEO |
375,000 |
9/18/2010 |
— |
0.50 |
— |
6/18/2020 |
|
250,000 |
8/12/2011 |
— |
0.0169 |
— |
8/12/2021 |
|
250,000 |
1/16/2012 |
— |
0.0169 |
— |
1/16/2022 |
|
1,250,000 |
8/6/2012 |
— |
0.0169 |
— |
8/6/2022 |
|
400,000 |
9/1/2013 |
— |
0.0165 |
— |
9/1/2023 |
| Kristin Comella, NEO |
24,712 |
2/20/2005 |
— |
0.71 |
|
2/19/2015 |
|
9,267 |
4/19/2006 |
— |
0.71 |
— |
4/19/2016 |
|
6,178 |
1/2/2007 |
— |
0.71 |
— |
1/2/2017 |
|
6,500 |
10/17/2007 |
— |
0.71 |
— |
10/17/2017 |
|
19,700 |
1/9/2009 |
— |
0.71 |
— |
1/9/2019 |
|
2,955 |
3/13/2009 |
— |
0.71 |
— |
3/13/2019 |
|
30,000 |
5/29/2009 |
— |
0.85 |
— |
5/29/2019 |
|
125,000 |
8/12/2011 |
— |
0.0169 |
— |
8/12/2021 |
|
187,500 |
8/06/2012 |
— |
0.0169 |
— |
8/06/2022 |
101
Director
Compensation
As of December 31, 2013, we
had seven directors that qualified for compensation. Our non-employee directors
do not receive cash compensation for their services as directors. However, it is
generally our policy to annually grant each non-employee director options to
purchase shares of our common stock provided that he or she has served as a
member of our Board of Directors for at least six months and one day of the
twelve month period immediately preceding the date of grant. In addition, we
reimburse non-employee directors for actual out-of-pocket expenses
incurred.
Security Ownership of
Certain Beneficial Owners and Management and Related Stockholder
Matters.
The following table sets
forth the beneficial ownership(1) of our common stock as of November
20,2014, based on an aggregate of 560,564,622 common shares issued and
outstanding and 145,377,723 shares issuance upon the conversion of securities
(outstanding options and warrants; specifically 23,376,000 options currently
exercisable and 122,001,723 currently exercisable warrants), for an aggregate
total of 705,942,345 beneficial shares, for each of our greater than 5%
shareholders, directors, named executive officers that continue to serve as
executive officers of Bioheart and by all of our directors and named executive
officers as a group. Unless otherwise indicated, the address of each of the
individuals and entities named below is: c/o Bioheart, Inc., 13794 NW 4th
Street, Suite 212, Sunrise, Florida 33325. Except as noted below, to our
knowledge, each person named in the table has sole voting and investment power
with respect to all shares of our common stock beneficially owned by them.
(1) The number and
percentage of shares beneficially owned is determined in accordance with Rule
13d-3 under the Exchange Act, and the information is not necessarily indicative
of beneficial ownership for any other purpose. The Company believes that each
individual or entity named has sole investment and voting power with respect to
the securities indicated as beneficially owned by them, subject to community
property laws, where applicable, except where otherwise noted. The “Amount of
Beneficial Ownership” in calculated based on total shares held plus warrants
held (plus stock options entitled to exercise). The aggregate of these items,
which totals 704,320,246, will be used as the denominator for the percentage
calculation below.
102
|
Amount of |
|
|
|
Beneficial |
|
|
| Name and
Address of Beneficial Owner |
Ownership |
|
Percent of
Class |
| Mike
Tomas, President, CEO, CFO, and Director |
16,928,947 |
(1) |
2.94 |
| |
|
|
|
| Kristin Comella, Chief Scientific Officer and
Director |
3,198,786 |
(2) |
* |
| |
|
|
|
| William P. Murphy, Director** |
21,837,295 |
(3) |
3.7 |
| |
|
|
|
| Charles A. Hart, Director** |
17,065,852 |
(4) |
2.94 |
| |
|
|
|
| Sam
Ahn, Director** |
39,897,957 |
(5) |
6.6 |
| |
|
|
|
| Mark
P. Borman, Director |
1,833,450 |
(6) |
* |
| |
|
|
|
| Sheldon T. Anderson, Director |
5,081,988 |
(7) |
* |
| |
|
|
|
| All
officers and directors as a group (7 persons) |
105,844,275 |
(8) |
16.72 |
| |
|
|
|
| Northstar Biotechnology Group, LLC |
47,368,582 |
(9) |
8.2 |
| * |
|
less than 1% |
| ** |
|
Excludes Northstar
Biotechnology Group, LLC (“Northstar”), owned partly by certain directors
and existing shareholders of the Company, including Dr. William P. Murphy
Jr., Dr. Samuel Ahn and Charles Hart. |
| (1) |
|
(i) Includes shares
are held by The Astri Group over which Mr. Tomas has shared voting and
investment power and includes (i) includes 6,578,947 shares of common
stock and (ii) 10,350,000 shares of common stock issuable upon exercise of
presently exercisable stock options. |
| (2) |
|
Includes 3,198,786
shares of common stock issuable upon exercise of presently exercisable
stock options. |
| (3) |
|
Includes (i)
19,113,766 shares of common stock and (ii) 2,723,529 shares of common
stock issuable upon exercise of presently exercisable stock options and
warrants. Shares are directly owned by trusts controlled by Dr. Murphy and
his spouse. |
| (4) |
|
Includes (i)
9,194,422 shares of common stock and (ii) 7,871,430 shares of common stock
issuable upon exercise of presently exercisable stock options and
warrants. |
| (5) |
|
Includes (i)
28,240,387 shares of common stock and (ii) 11,657,750 shares of common
stock issuable upon exercise of presently exercisable stock
options. |
| (6) |
|
Includes (i) 23,450
shares of common stock and (ii) 1,810,000 shares of common stock issuable
upon exercise of presently exercisable stock options |
| (7) |
|
Includes (i)
1,940,994 shares of common stock and (ii) 3,140,994 shares of common stock
issuable upon exercise of presently exercisable warrants. |
| (8) |
|
Includes an aggregate
of (i) 46,827,505 shares of common stock and (ii)25,489,251shares of
common stock issuable upon exercise of presently exercisable stock options
and warrants. |
| (9) |
|
Excludes 20,000,000
shares of Series A Preferred Stock (non-convertible) with each share of
Series A Preferred Stock having voting power of twenty-five common
shares. |
103
DESCRIPTION OF
SECURITIES
On February 4, 2013,
effective with the filing of the amendment to the Company's Articles of
Incorporation with the Florida Secretary of State (confirmed as filed on
February 11, 2013), the Company amended its Articles of Incorporation to
increase the authorized shares of capital stock of the Company to nine hundred
and seventy million (970,000,000) shares of capital stock consisting of nine
hundred and fifty million (950,000,000) shares of common stock and twenty
million (20,000,000) shares of preferred stock, both $.001 par value
respectively.
Effective May 19, 2014, the
Company amended its articles of incorporation to increase the authorized shares
of capital stock of the Company from nine hundred and fifty million
(950,000,000) shares of common stock and twenty million (20,000,000) shares of
preferred stock, both $.001 par value respectively, to two billion
(2,000,000,000) shares of shares of common stock and twenty million (20,000,000)
shares of preferred stock, both $.001 par value respectively.
The following statements
relating to the capital stock set forth the material terms of our securities;
however, reference is made to the more detailed provisions of, and such
statements are qualified in their entirety by reference to, the Certificate of
Incorporation, amendment to the Certificate of Incorporation and the By-laws,
copies of which are filed as exhibits to this registration statement.
COMMON
STOCK
The holders of our Common
Stock are entitled to one vote per share on all matters to be voted on by our
stockholders, including the election of directors. Our stockholders are not
entitled to cumulative voting rights, and, accordingly, the holders of a
majority of the shares voting for the election of directors can elect the entire
board of directors if they choose to do so and, in that event, the holders of
the remaining shares will not be able to elect any person to our board of
directors.
On February 4, 2013, the
Company amended its Articles of Incorporation to increase the number of
authorized shares to 970,000,000, consisting of 20,000,000 $0.001 par value
preferred stock and 950,000,000 $0.001 common stock.
The holders of the
Company’s Common Stock are entitled to receive ratably such dividends, if any,
as may be declared from time to time by the board of directors, in its
discretion, from funds legally available there for and subject to prior dividend
rights of holders of any shares of our Preferred Stock which may be outstanding.
Upon the Company’s liquidation, dissolution or winding up, subject to prior
liquidation rights of the holders of our Preferred Stock, if any, the holders of
our Common Stock are entitled to receive on a pro rata basis our remaining
assets available for distribution. Holders of the Company’s Common Stock have no
preemptive or other subscription rights, and there are no conversion rights or
redemption or sinking fund provisions with respect to such shares. All
outstanding shares of the Company’s Common Stock are, and all shares being
offered by this prospectus will be, fully paid and not liable to further calls
or assessment by the Company.
Preferred
Stock
The Company is authorized
to issue 20,000,000 shares of preferred stock, par value $0.001. The
designations, rights, and preferences of such preferred stock are to be
determined by the Board of Directors. Subsequently, 20,000,000 shares were
designated as Series A Preferred Stock.
The Series A Preferred
Stock collectively has voting rights equal to 25 votes on all matters presented
to be voted by the holders of common stock per share of preferred stock and the
right to convert to one share of common stock for each share of preferred stock.
Northstar Biotechnology Group, LLC was issued an aggregate of 20,000,000 shares
of Series A Preferred Stock.
104
DIVIDENDS
Dividends, if any, will be
contingent upon our revenues and earnings, if any, capital requirements and
financial conditions. The payment of dividends, if any, will be within the
discretion of our Board of Directors. We presently intend to retain all
earnings, if any, for use in its business operations and accordingly, the Board
of Directors does not anticipate declaring any dividends prior to a business
combination.
Certain Relationships and
Related Transactions and Director Independence.
Certain Relationships
and Related Party Transactions
Advances
As of September 30, 2014
and December 31, 2013, the Company officers and directors have provided advances
in the aggregate of $141,759 and $416,198 respectively, for working capital
purposes. The advances are unsecured, due on demand and non-interest bearing.
During the nine months
ended September 30, 2014, the Company issued an aggregate of 10,232,286 shares
of common stock in settlement of $298,759 of related party advances and
outstanding accounts payable.
Notes payable-related
party
Northstar
Biotechnology Group, LLC
On February 29, 2012, a
note issued to BlueCrest Master Fund Limited was assigned to Northstar
Biotechnology Group, LLC (“Northstar”), owned partly by certain directors and
existing shareholders of the Company, including Dr. William P. Murphy Jr., Dr.
Samuel Ahn and Charles Hart. At the date of the assignment, the principal amount
of the BlueCrest note was $544,267.
On March 30, 2012, the
Company and Northstar agreed to extend until May 1, 2012 the initial payment
date for any and all required monthly under the Note, such that the first of the
four monthly payments required under the Note will be due and payable on May,
2012 and all subsequent payments will be due on a monthly basis thereafter
commencing on June 1, 2012, and to waive any and all defaults and/or events of
default under the Note with respect to such payments. As of September 30, 2012,
the Company was in default, however, subsequent to September 30, 2012, the
Company renegotiated the terms of the Note, Northstar has agreed to suspend the
requirement of principal payments by the Company and allow payment of
interest-only in common stock.
On September 21, 2012, the
Company issued 5,000,000 common stock purchase warrants to Northstar that was
treated as Additional interest expense upon issuance.
On October 1, 2012, the
Company and Northstar entered into a limited waiver and forbearance agreement
providing a recapitalized new note balance comprised of all sums due Northstar
with a maturity date extended perpetually. The Company agreed to issue 5,000,000
shares of Series A Convertible Preferred Stock and 10,000,000 of common stock in
exchange for $210,000 as payment towards outstanding debt, default interest,
penalties, professional fees outstanding and due Northstar. In addition, the
Company executed a security agreement granting Northstar a lien on all patents,
patent applications, trademarks, service marks, copyrights and intellectual
property rights of any nature, as well as the results of all clinical trials,
know-how for preparing Myoblasts, old and new clinical data, existing approved
trials, all right and title to Myoblasts, clinical trial protocols and other
property rights.
105
In addition, the Company
granted Northstar a perpetual license on products as described for resale,
relicensing and commercialization outside the United States. In connection with
the granted license, Northstar shall pay the Company a royalty of up to 8% on
revenues generated.
Effective October 1, 2012,
the effective interest rate was 12.85% per annum. The parties agreed, as of
February 28, 2013, to reduce the interest rate to 7% per annum.
In connection with the
consideration paid, Northstar waived, from the effective date through the
earlier of termination or expiration of the agreement, satisfaction of the
obligations as described in the forbearance agreement. In 2012, 5,000,000 shares
of Series A Convertible Preferred Stock were approved to be issued, which was
subsequently increased to 20,000,000 shares of preferred stock as Series A
Convertible Preferred Stock. In addition, the Company is obligated to issue
additional preferred stock equal in lieu of payment of cash of accrued and
unpaid interest on each six month anniversary of the effective date (October 1,
2012). In lieu of the initial two payments in preferred stock, the parties have
determined to modify the voting rights of the Series A Convertible Preferred
Stock from 20 votes per share on matters to be voted on by the common stock
holders to 25 votes per share on matters to be voted on by the common stock
holders and all prior and subsequent payments of interest will be in common
stock. The Company is required to issue additional shares of its common stock
(as amended), in lieu of cash, each six month anniversary of the effective date
for any accrued and unpaid interest.
On April 2, 2014, the
Company issued 274,681 shares of its common stock in lieu of payment in cash of
accrued and unpaid interest of $12,635 due April 1, 2014 per the forbearance
agreement.
On September 17, 2014,
limited waiver and forbearance agreement was amended to provide that the
perpetual license on products as described for resale, relicensing and
commercialization outside the United States was conditioned upon NorthStar
providing certain financing, which financing the Company, in its sole
discretion, could decline and retain the license.
As of September 30, 2014
and December 31, 2013, the principle of this note was $362,000.
Officer and Director
Notes
At September 30, 2014 and
December 31, 2013, the Company has outstanding notes payable to officers and
directors with interest at 8% per annum due at maturity in aggregate $240,000
and $365,000, respectively. The remaining subordinated notes $100,000 and
$140,000 were previously due on November 30, 2012 and June 4, 2011 respectively,
and are unsecured. The Company is not obligated to make payment until Northstar
loan is paid off.
On September 30, 2013, the
Company issued an aggregate of 15,350,876 shares of its common stock in
settlement of $175,000 of these related party notes payable.
On June 20, 2014, the
Company issued 4,045,796 shares of its common stock in settlement of $125,000 of
related party note payable and accrued interest of $36,832.
On August 1, 2013, the
Company issued an aggregate of $500,000 promissory notes due on demand to
officers and employee in settlement of accrued compensation. The promissory
notes bear interest of 5% per annum and due at various maturity dates. During
the nine months ended September 30, 2014, the Company paid off $240,821 of the
outstanding promissory notes. The principle outstanding balance of these notes
as of September 30, 2014 is $189,813.
On July 1, 2014, the
Company issued an aggregate of $800,000 promissory notes to officers and
employee in settlement of accrued compensation. The promissory notes bear
interest of 5% per annum and due on January 1, 2015.
106
As of September 30, 2014
and December 31, 2013, the Company officers and directors have outstanding notes
in aggregate of $785,458 and $1,014,477, respectively. The notes are at from
4.75% to 8% per annum and are due upon payoff of the Northstar note payable
described above.
On June 20, 2014, the
Company issued 10,333,475 shares of its common stock in settlement of $300,385
of related party note payable and accrued interest of $112,954.
DISCLOSURE OF COMMISSION
POSITION OF
INDEMNIFICATION FOR SECURITIES ACT LIABILITIES
Sections 78.7502 and 78.751
of the Nevada Revised Statutes authorizes a court to award, or a corporation’s
board of directors to grant indemnity to directors and officers in terms
sufficiently broad to permit indemnification, including reimbursement of
expenses incurred, under certain circumstances for liabilities arising under the
Securities Act of 1933, as amended. In addition, the registrant’s Bylaws provide
that the registrant has the authority to indemnify the registrant’s directors
and officers and may indemnify the registrant’s employees and agents (other than
officers and directors) against liabilities to the fullest extent permitted by
Nevada law. The registrant is also empowered under the registrant’s Bylaws to
purchase insurance on behalf of any person whom the registrant is required or
permitted to indemnify.
Insofar as indemnification
for liabilities arising under the Securities Act may be permitted to directors,
officers or persons controlling us pursuant to the foregoing provisions, or
otherwise, we have been advised that in the opinion of the SEC, such
indemnification is against public policy as expressed in the Securities Act and
is therefore unenforceable.
WHERE YOU CAN FIND MORE
INFORMATION
The Securities and Exchange
Commission has declared effective our registration statement on Form S-1,
together with any amendments and related exhibits, under the Securities Act of
1933, with respect to our shares of common stock offered by this prospectus. The
registration statement contains additional information about us and our shares
of common stock being offered by this prospectus.
For further information
with respect to us and the securities covered by this prospectus, please see the
Registration Statement and the exhibits filed with the Registration Statement. A
copy of the Registration Statement and the exhibits filed with the Registration
Statement may be inspected without charge at the Public Reference Room
maintained by the SEC, located at 100 F Street, N.E., Washington, D.C. 20549.
Please call the SEC at 1-800-732-0330 for more information about the operation
of the Public Reference Room. The SEC also maintains an Internet website that
contains reports, proxy and information statements and other information
regarding registrants that file electronically with the SEC. The address of the
website is www.sec.gov.
We are subject to the
information and periodic reporting requirements of the Exchange Act, and, in
accordance therewith, we file periodic reports, proxy statements and other
information with the SEC. Such periodic reports, proxy statements and other
information are available for inspection and copying at the Public Reference
Room and website of the SEC referred to above.
107
INDEX TO FINANCIAL
STATEMENTS
BIOHEART, INC.
Table of Contents
|
|
Page |
|
| Financial Statements for the Year
Ended September 30, 2014 |
|
|
|
| |
|
|
|
| Condensed Balance Sheets as of September 30, 2014 (unaudited) and December 31, 2013 |
|
F-1 |
|
| |
|
|
|
Unaudited Condensed Statements of Operations (unaudited) – Three and Nine Months
Ended September 30, 2014 and 2013 |
|
F-2 |
|
| |
|
|
|
| Unaudited Condensed Statement of Stockholders’ Deficit (unaudited) – Nine Months |
|
|
|
| Ended September 30, 2014 |
|
F-3 |
|
| |
|
|
|
| Unaudited Condensed Statements of Cash Flows (unaudited) – Nine Months Ended |
|
|
|
| September 30, 2014 and 2013 |
|
F-4 |
|
| |
|
|
|
| Notes to Unaudited Condensed Financial Statements |
|
F-5 |
|
| |
|
| Financial Statements for the Year Ended December 31, 2013 |
|
|
|
| |
|
|
|
| Report of Independent Registered Accounting Firm |
|
F-23 |
|
| |
|
|
|
| Balance Sheets as of December 31, 2013 and 2012 |
|
F-24 |
|
| |
|
|
|
| Statements of Operations for the Years Ended December 31, 2013, 2012 and the |
|
|
|
| Cumulative Period from August 12, 1999 (date of inception) to December 31, 2013 |
|
F-25 |
|
| |
|
|
|
| Statement of Stockholders’ Deficit from August 12, 1999 (date of inception) through |
|
|
|
| December 31, 2013 |
|
F-26 |
|
| |
|
|
|
| Statements of Cash Flows for the Years Ended December 31, 2013, 2012 and the |
|
|
|
| Cumulative Period from August 12, 1999 (date of inception) to December 31, 2013 |
|
F-36 |
|
| |
|
|
|
| Notes to Financial Statements |
|
F-38 |
|
108
BIOHEART, INC.
CONDENSED BALANCE SHEETS
|
|
September 30, |
|
December 31, |
|
|
2014 |
|
2013 |
|
|
(unaudited) |
|
|
|
|
| ASSETS |
|
|
|
|
|
|
|
|
| Current assets: |
|
|
|
|
|
|
|
|
| Cash
and cash equivalents |
|
$ |
46,592 |
|
|
$ |
46,227 |
|
| Accounts receivable, net |
|
|
153,863 |
|
|
|
19,913 |
|
| Prepaid and other |
|
|
26,164 |
|
|
|
784 |
|
| Total
current assets |
|
|
226,619 |
|
|
|
66,924 |
|
| |
| Property and equipment, net |
|
|
13,431 |
|
|
|
9,055 |
|
| |
| Other assets |
|
|
8,404 |
|
|
|
10,160 |
|
| |
| Total
assets |
|
$ |
248,454 |
|
|
$ |
86,139 |
|
| |
| LIABILITIES AND STOCKHOLDERS' DEFICIT |
|
|
|
|
|
|
|
|
| Current liabilities: |
|
|
|
|
|
|
|
|
| Accounts payable |
|
$ |
2,068,256 |
|
|
$ |
2,382,267 |
|
| Accrued expenses |
|
|
2,291,809 |
|
|
|
4,480,335 |
|
| Advances, related party |
|
|
141,759 |
|
|
|
416,198 |
|
| Deposits |
|
|
478,286 |
|
|
|
478,286 |
|
| Subordinated debt, related party |
|
|
1,500,000 |
|
|
|
1,500,000 |
|
| Notes payable, related party |
|
|
2,377,271 |
|
|
|
2,241,477 |
|
| Notes payable, net of debt discount |
|
|
1,478,934 |
|
|
|
1,930,841 |
|
| Total
current liabilities |
|
|
10,336,315 |
|
|
|
13,429,404 |
|
| |
| Long
term debt: |
|
|
|
|
|
|
|
|
| Derivative liability |
|
|
725,122 |
|
|
|
403,811 |
|
| |
| Stockholders' deficit: |
|
|
|
|
|
|
|
|
| Preferred stock, par value $0.001; 20,000,000
shares authorized, 20,000,000 issued |
|
|
|
|
|
|
|
|
| and
outstanding |
|
|
20,000 |
|
|
|
20,000 |
|
| Common stock, par value $0.001;2,000,000 and
950,000,000 shares authorized, |
|
|
|
|
|
|
|
|
| respectively, 533,694,420 and 379,787,745
shares issued and outstanding as of |
|
|
|
|
|
|
|
|
| September 30, 2014 and December 31, 2013,
respectively |
|
|
533,694 |
|
|
|
379,788 |
|
| Additional paid in capital |
|
|
107,851,505 |
|
|
|
103,819,119 |
|
| Common stock subscription |
|
|
210,000 |
|
|
|
215,000 |
|
| Accumulated deficit |
|
|
(119,428,182 |
) |
|
|
(118,180,983 |
) |
| Total
stockholders' deficit |
|
|
(10,812,983 |
) |
|
|
(13,747,076 |
) |
| |
| Total liabilities and stockholders'
deficit |
|
$ |
248,454 |
|
|
$ |
86,139 |
|
See the accompanying notes
to these financial statements
F-1
BIOHEART,
INC.
CONDENSED STATEMENTS
OF OPERATIONS
(unaudited)
|
|
Three months ended September 30, |
|
Nine
months ended September 30, |
|
|
2014 |
|
2013 |
|
2014 |
|
2013 |
| Revenue: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Products |
|
$ |
281,752 |
|
|
$ |
25,350 |
|
|
$ |
869,012 |
|
|
$ |
25,350 |
|
| Services |
|
|
297,784 |
|
|
|
6,309 |
|
|
|
694,852 |
|
|
|
30,630 |
|
| Total
revenue |
|
|
579,536 |
|
|
|
31,659 |
|
|
|
1,563,864 |
|
|
|
55,980 |
|
| |
| Cost
and operating expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Cost
of sales |
|
|
523,222 |
|
|
|
- |
|
|
|
889,509 |
|
|
|
- |
|
| Research and development |
|
|
8,581 |
|
|
|
158,381 |
|
|
|
33,916 |
|
|
|
494,762 |
|
| Marketing, general and administrative |
|
|
1,512,706 |
|
|
|
940,354 |
|
|
|
3,182,397 |
|
|
|
1,750,276 |
|
| Depreciation and amortization |
|
|
1,759 |
|
|
|
77 |
|
|
|
3,745 |
|
|
|
1,327 |
|
| Total
operating expenses |
|
|
2,046,268 |
|
|
|
1,098,812 |
|
|
|
4,109,567 |
|
|
|
2,246,365 |
|
| |
| Net
loss from operations |
|
|
(1,466,732 |
) |
|
|
(1,067,153 |
) |
|
|
(2,545,703 |
) |
|
|
(2,190,385 |
) |
| |
| Other income (expenses): |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Gain
on Settlement of debt |
|
|
85,2229 |
|
|
|
- |
|
|
|
2,272,283 |
|
|
|
1,004,224 |
|
| Gain
on change of fair value of derivative |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| liability |
|
|
239,296 |
|
|
|
129,298 |
|
|
|
61,339 |
|
|
|
39,885 |
|
| Other income |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
939 |
|
| Interest expense |
|
|
(331,026 |
) |
|
|
(208,914 |
) |
|
|
(1,035,118 |
) |
|
|
(1,270,663 |
) |
| Total
other income (expenses) |
|
|
(6,501 |
) |
|
|
(79,616 |
) |
|
|
1,298,504 |
|
|
|
(225,615 |
) |
| |
| Net
loss before income taxes |
|
|
(1,473,233 |
) |
|
|
(1,146,769 |
) |
|
|
(1,247,199 |
) |
|
|
(2,416,000 |
) |
| |
| Income taxes (benefit) |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
| |
| NET
LOSS |
|
$ |
(1,473,233 |
) |
|
$ |
(1,146,769 |
) |
|
$ |
(1,247,199 |
) |
|
$ |
(2,416,000 |
) |
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Net
loss per common share, basic and diluted |
|
$ |
(0.003 |
) |
|
$ |
(0.005 |
) |
|
$ |
(0.003 |
) |
|
$ |
(0.011 |
) |
| |
| Weighted average number of common
shares |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| outstanding, basic and diluted |
|
|
519,135,426 |
|
|
|
246,229,899 |
|
|
|
464,607,869 |
|
|
|
217,392,664 |
|
See the accompanying notes
to these financial statements
F-2
BIOHEART,
INC.
CONDENSED STATEMENT OF STOCKHOLDERS' DEFICIT
NINE MONTHS ENDED
SEPTEMBER 30, 2014
(unaudited)
|
|
|
|
|
|
|
|
|
|
|
|
Additional |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Preferred stock |
|
Common stock |
|
Paid
in |
|
Subscription |
|
Accumulated |
|
|
|
|
|
|
Shares |
|
Amount |
|
Shares |
|
Amount |
|
Capital |
|
Receivable |
|
Deficit |
|
Total |
| Balance, December 31, 2013 |
|
20,000,000 |
|
$ |
20,000 |
|
379,787,745 |
|
$ |
379,788 |
|
$ |
103,819,119 |
|
$ |
215,000 |
|
|
$ |
(118,180,983 |
) |
|
$ |
(13,747,076 |
) |
| Common stock issued in settlement of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| accounts payable |
|
- |
|
|
- |
|
45,232,118 |
|
|
45,232 |
|
|
946,285 |
|
|
- |
|
|
|
- |
|
|
|
991,517 |
|
| Common stock issued in connection |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| with
settlement of other debt |
|
- |
|
|
- |
|
64,434,256 |
|
|
64,434 |
|
|
1,832,646 |
|
|
- |
|
|
|
- |
|
|
|
1,897,080 |
|
| Common stock issued for services |
|
- |
|
|
- |
|
3,839,832 |
|
|
3,840 |
|
|
180,151 |
|
|
- |
|
|
|
- |
|
|
|
183,991 |
|
| Common stock issued upon exercise of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| warrants |
|
- |
|
|
- |
|
11,918,181 |
|
|
11,918 |
|
|
124,082 |
|
|
- |
|
|
|
- |
|
|
|
136,000 |
|
| Proceeds from issuance of common |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| stock |
|
- |
|
|
- |
|
28,482,288 |
|
|
28,482 |
|
|
404,769 |
|
|
(5,000 |
) |
|
|
- |
|
|
|
428,251 |
|
| Fair
value of vesting warrants issued in |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| connection with joint venture and |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| intellectual property |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
136,837 |
|
|
- |
|
|
|
- |
|
|
|
136,837 |
|
| Stock based compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
407,616 |
|
|
- |
|
|
|
- |
|
|
|
407,616 |
|
| Net
income |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
|
(1,247,199 |
) |
|
|
(1,247,199 |
) |
| Balance,
September 30, 2014 |
|
20,000,000 |
|
$ |
20,000 |
|
533,694,420 |
|
$ |
533,694 |
|
$ |
107,851,505 |
|
$ |
210,000 |
|
|
$ |
(119,428,182 |
) |
|
$ |
(10,812,983 |
) |
See the accompanying notes
to these financial statements
F-3
BIOHEART,
INC.
CONDENSED STATEMENTS
OF CASH FLOWS
(unaudited)
|
|
Nine
months ended September 30, |
|
|
2014 |
|
2013 |
| CASH
FLOWS FROM OPERATING ACTIVITIES: |
|
|
|
|
|
|
|
|
| Net
loss |
|
$ |
(1,247,199 |
) |
|
$ |
(2,416,000 |
) |
| Adjustments to reconcile net loss to net cash used in
operating activities: |
|
|
|
|
|
|
|
|
| Depreciation and amortization |
|
|
3,745 |
|
|
|
1,328 |
|
| Bad
debt expense |
|
|
28,732 |
|
|
|
- |
|
| Discount on convertible debt |
|
|
343,592 |
|
|
|
295,750 |
|
| Gain
on change in fair value of derivative liability |
|
|
(61,339 |
) |
|
|
(39,885 |
) |
| Gain
on settlement of debt |
|
|
(2,272,283 |
) |
|
|
(1,004,224 |
) |
| Non-cash payment of interest |
|
|
385,501 |
|
|
|
168,350 |
|
| Warrants issued in connection with joint venture agreement
and intellectual |
|
|
|
|
|
|
|
|
| property |
|
|
136,837 |
|
|
|
- |
|
| Related party notes payable issued for services
rendered |
|
|
800,000 |
|
|
|
500,000 |
|
| Common stock issued in connection with accounts
payable |
|
|
- |
|
|
|
2,500 |
|
| Common stock issued in exchange for services |
|
|
38,750 |
|
|
|
40,519 |
|
| Preferred stock issued in settlement of debt and forbearance
agreement |
|
|
- |
|
|
|
274,050 |
|
| Stock based compensation |
|
|
407,616 |
|
|
|
102,674 |
|
| (Increase) decrease in: |
|
|
|
|
|
|
|
|
| Receivables |
|
|
(162,682 |
) |
|
|
(7,553 |
) |
| Inventory |
|
|
- |
|
|
|
62,953 |
|
| Prepaid and other current assets |
|
|
(23,624 |
) |
|
|
41,533 |
|
| Increase (decrease) in: |
|
|
|
|
|
|
|
|
| Accounts payable |
|
|
628,684 |
|
|
|
(76,240 |
) |
| Accrued expenses |
|
|
246,486 |
|
|
|
485,518 |
|
| Net cash used in
operating activities |
|
|
(747,184 |
) |
|
|
(1,568,727 |
) |
| |
| CASH
FLOWS FROM INVESTING ACTIVITIES: |
|
|
|
|
|
|
|
|
| Acquisitions of property and equipment |
|
|
(8,121 |
) |
|
|
- |
|
| Net cash used by
investing activities |
|
|
(8,121 |
) |
|
|
- |
|
| |
| CASH
FLOWS FROM FINANCING ACTIVITIES: |
|
|
|
|
|
|
|
|
| Bank
overdraft |
|
|
- |
|
|
|
(89 |
) |
| Proceeds from issuance of common stock, net |
|
|
428,251 |
|
|
|
1,100,000 |
|
| (Repayments (proceeds) from related party advances |
|
|
26,759 |
|
|
|
180,500 |
|
| Proceeds from exercise of stock options and
warrants |
|
|
136,000 |
|
|
|
- |
|
| Proceeds from notes payable |
|
|
388,000 |
|
|
|
295,000 |
|
| Repayments of notes payable |
|
|
(223,340 |
) |
|
|
- |
|
| Net cash provided in
financing activities |
|
|
755,670 |
|
|
|
1,575,411 |
|
| |
| Net increase in cash and
cash equivalents |
|
|
365 |
|
|
|
6,684 |
|
| |
| Cash
and cash equivalents, beginning of period |
|
|
46,227 |
|
|
|
- |
|
| Cash
and cash equivalents, end of period |
|
$ |
46,592 |
|
|
$ |
6,684 |
|
| |
| SUPPLEMENTAL DISCLOSURES OF CASH FLOW INFORMATION |
|
|
|
|
|
|
|
|
| Interest paid |
|
$ |
161,532 |
|
|
$ |
513,986 |
|
| Income taxes paid |
|
$ |
- |
|
|
$ |
- |
|
| |
| Non-cash financing activities: |
|
|
|
|
|
|
|
|
| Common stock issued in settlement of notes payable |
|
$ |
938,075 |
|
|
$ |
345,000 |
|
| Common stock issued in settlement of accounts
payable |
|
$ |
857,445 |
|
|
$ |
149,239 |
|
| Common stock issued in settlement of related party notes and
advances payable |
|
$ |
862,880 |
|
|
$ |
- |
|
| Preferred stock issued in settlement of notes
payable |
|
$ |
- |
|
|
$ |
70,000 |
|
See the accompanying notes
to these financial statements
F-4
NOTE 1 – SIGNIFICANT
ACCOUNTING POLICIES
A summary of the
significant accounting policies applied in the presentation of the accompanying
unaudited condensed financial statements follows:
General
The accompanying unaudited
condensed financial statements of Bioheart, Inc., (the “Company”), have been
prepared in accordance with the rules and regulations (Regulation S-X) of the
Securities and Exchange Commission (the “SEC”) and with the instructions to Form
10-Q. Accordingly, they do not include all of the information and footnotes
required by generally accepted accounting principles for complete financial
statements.
In the opinion of
management, all adjustments (consisting of normal recurring accruals) considered
necessary for a fair presentation have been included. The results from
operations for the nine month period ended September 30, 2014 are not
necessarily indicative of the results that may be expected for the year ended
December 31, 2014. The unaudited condensed financial statements should be read
in conjunction with the December 31, 2013 financial statements and footnotes
thereto included in the Company’s Annual Report on Form 10-K.
Basis and business
presentation
Bioheart, Inc. (the
“Company”) was incorporated under the laws of the State of Florida in August,
1999. The Company is in the cardiovascular sector of the cell technology
industry delivering cell therapies and biologics that help address congestive
heart failure, lower limb ischemia, chronic heart ischemia, acute myocardial
infarctions and other issues. To date, the Company has not generated significant
sales revenues, has incurred expenses, and has sustained losses. Consequently,
its operations are subject to all the risks inherent in the establishment of a
research and development business enterprise.
Revenue
Recognition
The Company recognizes
revenue in accordance with Accounting Standards Codification subtopic 605-10,
Revenue Recognition (“ASC 605-10”) which requires that four basic criteria must
be met before revenue can be recognized: (1) persuasive evidence of an
arrangement exists; (2) delivery has occurred; (3) the selling price is fixed
and determinable; and (4) collectability is reasonably assured. Determination of
criteria (3) and (4) are based on management’s judgments regarding the fixed
nature of the selling prices of the products delivered and the collectability of
those amounts. Provisions for discounts and rebates to customers, estimated
returns and allowances, and other adjustments are provided for in the same
period the related sales are recorded.
At the time of each
transaction, management assesses whether the fee associated with the transaction
is fixed or determinable and whether or not collection is reasonably assured.
The assessment of whether the fee is fixed or determinable is based upon the
payment terms of the transaction. If a significant portion of a fee is due after
our normal payment terms or upon implementation or client acceptance, the fee is
accounted for as not being fixed or determinable and revenue is recognized as
the fees become due or after implementation or client acceptance has occurred.
Collectability is assessed based on a number of factors, including past
transaction history with the client and the creditworthiness of the client.
Revenues for test kits and
equipment sold are not recorded until test kits are delivered. The Company has
revenue sharing arrangements for the sale of goods whereby the Company is the
primary obligor, sets pricing with the customers and bears all associated credit
risks with the customers. Sales under revenue share arrangements are recorded as
gross sales and any portion shared with third parties under such arrangements
are recorded as cost of sales. Revenues from trainings are not recorded until
the completion of the training. Any cash received as a deposit for trainings are
recorded by the company as a liability.
Unbilled revenue is revenue
that is recognized but is not currently billable to the customer pursuant to
contractual terms. In general, such amounts become billable in accordance with
predetermined payment schedules, but recognized as revenue as services are
performed. Amounts included in unbilled revenue are expected to be collected
within one year and are included within current assets. As of December 31, 2013
and September 30, 2014 there were no unbilled revenues.
F-5
Comprehensive
Income
The Company does not have
any items of comprehensive income in any of the periods presented.
Net loss per Common
Share, basic and diluted
The Company has adopted
Accounting Standards Codification subtopic 260-10, Earnings Per Share (“ASC
260-10”) specifying the computation, presentation and disclosure requirements of
earnings per share information. Basic loss per share has been calculated based
upon the weighted average number of common shares outstanding. Stock options,
warrants and shares issuable upon conversion of notes payable have been excluded
as common stock equivalents in the diluted loss per share because their effect
is anti-dilutive on the computation for the three and nine months ended
September 30, 2014 and 2013, respectively. Fully diluted shares outstanding were
659,543,477 and 323,296,916 for the three months ended September 30, 2014 and
2013, respectively and 605,015,919 and 336,682,241 for the nine months ended
September 30, 2014 and 2013, respectively.
Stock Based
Compensation
The Company follows
Accounting Standards Codification subtopic 718-10, Compensation (“ASC 718-10”)
which requires that all share-based payments to both employees and non-employees
be recognized in the income statement based on their fair values. (See note 10).
As of September 30, 2014,
there were outstanding stock options to purchase 63,486,987 shares of common
stock, 24,576,000 shares of which were vested.
Concentrations of Credit
Risk
The Company’s financial
instruments that are exposed to a concentration of credit risk are cash and
accounts receivable. The financial stability of these institutions is
periodically reviewed by senior management.
As of September 30, 2014,
two customers represented 24% and 18% of the Company’s accounts receivable.
As of December 31, 2013,
three customers represented 20%, 20% and 36% (total 76%) of the Company’s
accounts receivable.
For the three months ended
September 30, 2014, the Company’s revenues earned from the sale of products and
services were $579,536, of which no customer represented 10% or more of the
Company’s revenues. For the three months ended September 30, 2013, the Company’s
revenues earned from the sale of products and services were $31,659, of which
two customers represented 18% and 80% of the Company’s revenues.
For the nine months ended
September 30, 2014, the Company’s revenues earned from the sale of products and
services were $1,563,864, of which one customer represented 11% of the Company’s
revenues. For the nine months ended September 30, 2013, the Company’s revenues
earned from the sale of products and services were $55,980, of which three
customers represented 18%, 23% and 44% of the Company’s revenues.
Research and
Development
The Company accounts for
research and development costs in accordance with Accounting Standards
Codification subtopic 730-10, Research and Development (“ASC 730-10”). Under ASC
730-10, all research and development costs must be charged to expense as
incurred. Accordingly, internal research and development costs are expensed as
incurred. Third-party research and development costs are expensed when the
contracted work has been performed or as milestone results have been achieved as
defined under the applicable agreement. Company-sponsored research and
development costs related to both present and future products are expensed in
the period incurred. The Company incurred research and development expenses of
$8,581 and $33,916 for the three and nine months ended September 30, 2014,
respectively; and $158,381 and $494,762 for the three and nine months ended
September 30, 2013, respectively.
F-6
Fair
Value
Accounting Standards
Codification subtopic 825-10, Financial Instruments (“ASC 825-10”) requires
disclosure of the fair value of certain financial instruments. The carrying
value of cash and cash equivalents, accounts payable and accrued liabilities,
and short-term borrowings, as reflected in the balance sheets, approximate fair
value because of the short-term maturity of these instruments. All other
significant financial assets, financial liabilities and equity instruments of
the Company are either recognized or disclosed in the financial statements
together with other information relevant for making a reasonable assessment of
future cash flows, interest rate risk and credit risk. Where practicable the
fair values of financial assets and financial liabilities have been determined
and disclosed; otherwise only available information pertinent to fair value has
been disclosed.
The Company follows
Accounting Standards Codification subtopic 820-10, Fair Value Measurements and
Disclosures (“ASC 820-10”) and Accounting Standards Codification subtopic
825-10, Financial Instruments (“ASC 825-10”), which permits entities to choose
to measure many financial instruments and certain other items at fair value.
Neither of these statements had an impact on the Company’s financial position,
results of operations nor cash flows.
Reliance on Key
Personnel and Consultants
The Company has 4 full-time
employees and 1 part-time employee. The Company is heavily dependent on the
continued active participation of these current executive officers, employees
and key consultants. The loss of any of the senior management or key consultants
could significantly and negatively impact the business until adequate
replacements can be identified and put in place.
Derivative Instrument
Liability
The Company accounts for
derivative instruments in accordance with ASC 815, which establishes accounting
and reporting standards for derivative instruments and hedging activities,
including certain derivative instruments embedded in other financial instruments
or contracts and requires recognition of all derivatives on the balance sheet at
fair value, regardless of hedging relationship designation. Accounting for
changes in fair value of the derivative instruments depends on whether the
derivatives qualify as hedge relationships and the types of relationships
designated are based on the exposures hedged. At September 30, 2014 and December
31, 2013, the Company did not have any derivative instruments that were
designated as hedges.
Recent Accounting
Pronouncements
The Company has adopted
Accounting Standards Update (ASU) No. 2014-10, Development Stage Entities (Topic 915):
Elimination of Certain Financial Reporting Requirements, Including an Amendment
to Variable Interest Entities Guidance in Topic 810,
Consolidation. The amendments in
this ASU remove all incremental financial reporting requirements from U.S. GAAP
for development stage entities, including the removal of Topic 915,
Development Stage
Entities, from the FASB
Accounting Standards
Codification™. A development
stage entity is one that devotes substantially all of its efforts to
establishing a new business and for which: (a)
planned principal operations have not commenced; or (b)
planned principal operations have commenced, but have produced no significant
revenue. For example, many start-ups or even long-lived organizations that have
not yet begun their principal operations or do not have significant revenue
would be identified as development stage entities. For public business entities,
the presentation and disclosure requirements in Topic 915 will no longer be
required for the first annual period beginning after December 15, 2014. The
revised consolidation standards are effective one year later, in annual periods
beginning after December 15, 2015. Early adoption is permitted. The Company has
early adopted the standards under ASN No. 2014-10.
F-7
The FASB has issued ASU No.
2014-12, Compensation – Stock
Compensation (Topic 718): Accounting for Share-Based Payments When the Terms of
an Award Provide That a Performance Target Could Be Achieved after the Requisite
Service Period. This ASU requires
that a performance target that affects vesting, and that could be achieved after
the requisite service period, be treated as a performance condition. As such,
the performance target should not be reflected in estimating the grant date fair
value of the award. This update further clarifies that compensation cost should
be recognized in the period in which it becomes probable that the performance
target will be achieved and should represent the compensation cost attributable
to the period(s) for which the requisite service has already been rendered.. The
amendments in this ASU are effective for annual periods and interim periods
within those annual periods beginning after December 15, 2015. Earlier adoption
is permitted. The Company has not yet determined the effect of the adoption of
this standard and it is expected to have a material impact on the Company’s
condensed financial position and results of operations.
The FASB has issued ASU No.
2014-09, Revenue from Contracts
with Customers. This ASU
supersedes the revenue recognition requirements in Accounting Standards
Codification 605 - Revenue Recognition and most industry-specific guidance
throughout the Codification. The standard requires that an entity recognizes
revenue to depict the transfer of promised goods or services to customers in an
amount that reflects the consideration to which the company expects to be
entitled in exchange for those goods or services. This ASU is effective on
January 1, 2017 and should be applied retrospectively to each prior reporting
period presented or retrospectively with the cumulative effect of initially
applying the ASU recognized at the date of initial application. The Company has
not yet determined the effect of the adoption of this standard and it is
expected to have a material impact on the Company’s consolidated financial
position and results of operations.
There are various other
updates recently issued, most of which represented technical corrections to the
accounting literature or application to specific industries and are not expected
to a have a material impact on the Company's financial position, results of
operations or cash flows
Reclassification
Certain reclassifications
have been made to prior periods’ data to conform to the current year’s
presentation. More specifically, product and service revenue has been separately
disclosed on the face of the statement of operations. These reclassifications
had no effect on reported income or losses.
NOTE 2 – GOING CONCERN
MATTERS
The accompanying unaudited
condensed financial statements have been prepared on a going concern basis,
which contemplates the realization of assets and the satisfaction of liabilities
in the normal course of business. As shown in the accompanying unaudited
condensed financial statements, during nine months ended September 30, 2014, the
Company incurred an operating loss of $1,247,199 and used $747,184 in cash for
operating activities. As of September 30, 2014, the Company had a working
capital deficit (current liabilities in excess of current assets) of
approximately $10.0 million. These factors among others may indicate that the
Company will be unable to continue as a going concern for a reasonable period of
time.
The Company’s existence is
dependent upon management’s ability to develop profitable operations and to
obtain additional funding sources. There can be no assurance that the Company’s
financing efforts will result in profitable operations or the resolution of the
Company’s liquidity problems. The accompanying statements do not include any
adjustments that might result should the Company be unable to continue as a
going concern.
F-8
NOTE 3 — PROPERTY AND
EQUIPMENT
Property and equipment as
of September 30, 2014 and December 31, 2013 is summarized as follows:
|
|
|
September
30, |
|
December
31, |
|
|
|
|
2014 |
|
2013 |
|
| |
Laboratory and medical equipment |
|
$ |
352,358 |
|
|
$ |
352,358 |
|
|
|
Furniture, fixtures and equipment |
|
|
125,634 |
|
|
|
125,634 |
|
|
|
Computer equipment |
|
|
47,646 |
|
|
|
39,525 |
|
|
|
Leasehold improvements |
|
|
362,046 |
|
|
|
362,046 |
|
|
|
|
|
|
887,684 |
|
|
|
879,563 |
|
|
|
Less accumulated depreciation and
amortization |
|
|
(874,253 |
) |
|
|
(870,508 |
) |
|
|
|
|
$ |
13,431 |
|
|
$ |
9,055 |
|
|
Property and equipment are
recorded on the basis of cost. For financial statement purposes, property, plant
and equipment are depreciated using the straight-line method over their
estimated useful lives.
Expenditures for repair and
maintenance which do not materially extend the useful lives of property and
equipment are charged to operations. When property or equipment is sold or
otherwise disposed of, the cost and related accumulated depreciation are removed
from the respective accounts with the resulting gain or loss reflected in
operations. Management periodically reviews the carrying value of its property
and equipment for impairment in accordance with the guidance for impairment of
long lived assets.
NOTE 4 — ACCRUED
EXPENSES
Accrued expenses consisted
of the following as of September 30, 2014 and December 31, 2013:
|
|
|
September
30, |
|
December
31, |
|
| |
|
|
2014 |
|
2013 |
|
|
License and royalty fees |
|
$ |
— |
|
$ |
2,122,130 |
|
|
Amounts payable to the Guarantors of the
Company’s loan agreement |
|
|
|
|
|
|
|
| |
with
Bank of America and Seaside Bank, including fees and interest |
|
|
1,493,961 |
|
|
1,373,775 |
|
|
Interest payable on notes payable |
|
|
666,370 |
|
|
714,180 |
|
|
Vendor accruals and other |
|
|
114,259 |
|
|
120,692 |
|
|
Employee commissions,
compensation, etc. |
|
|
17,219 |
|
|
149,558 |
|
|
|
|
$ |
2,291,809 |
|
$ |
4,480,335 |
|
In June 2000, the Company
had entered into an agreement with William Beaumont Hospital, or WBH, pursuant
to which WBH granted the Company a worldwide, exclusive, non-sublicenseable
license to two U.S. method patents covering the inducement of human adult
myocardial cell proliferation in vitro, or the WBH IP. The term of the agreement
was for the life of the patents, which expire in 2015. The Company did not use
this license in any of their technologies or made any payments to WBH other than
the initial payment to acquire the license. On April 2, 2014, the Company
received confirmation from WBH that it has no obligation under the patent
license agreement and WBH agreed to terminate the patent license agreement
pursuant to a termination letter dated March 3, 2014.
Accordingly, the Company
recognized approximately $2,122,130 in settlement of debt which represents the
accumulative accrual and related interest from past years under the 2000 patent
license agreement.
During the nine months
ended September 30, 2014, the Company issued an aggregate of 45,232,118 shares
of its common stock in settlement of outstanding accounts payable and accrued
expenses. In connection with the issuance, the Company incurred $105,737 loss in
settlement of debt.
F-9
NOTE 5 – STANDBY EQUITY
DISTRIBUTION AGREEMENT
On November 2, 2011, the
Company and Greystone Capital Partners (“Greystone”) had entered into a Standby
Equity Distribution Agreement (the “Agreement”). Pursuant to the Agreement,
Greystone had agreed to provide the Company with up to $1.0 million of funding
for the 24-month period following the date a registration statement of the
Company’s common stock is declared effective by the SEC (the “Equity Line”). The
registration statement went effective on February 10, 2012. The Agreement
automatically terminated on the first of April, 2014 (the first day of the month
next following the second (2nd) anniversary of the Effective Date).
NOTE 6 – NOTES
PAYABLE
Notes payable were
comprised of the following as of September 30, 2014 and December 31, 2013:
|
|
|
September
30, |
|
December
31, |
|
| |
|
|
2014 |
|
2013 |
|
|
Seaside Bank note payable. |
|
$ |
980,000 |
|
|
$ |
980,000 |
|
|
|
August 2008 Unsecured Promissory
Note |
|
|
- |
|
|
|
500,000 |
|
|
| |
Hunton & Williams notes payable |
|
|
384,972 |
|
|
|
384,972 |
|
|
|
Asher notes payable |
|
|
175,500 |
|
|
|
143,000 |
|
|
|
Daniel James Management note payable |
|
|
50,000 |
|
|
|
- |
|
|
|
Fourth Man, LLC note payable |
|
|
75,000 |
|
|
|
35,000 |
|
|
|
Total notes payable |
|
|
1,665,472 |
|
|
|
2,042,972 |
|
|
|
Less
unamortized debt discount |
|
|
(186,538 |
) |
|
|
(112,131 |
) |
|
|
Total notes payable net of
unamortized debt discount |
|
$ |
1,478,934 |
|
|
$ |
1,930,841 |
|
|
Seaside
Bank
On October 25, 2010, the
Company entered into a Loan Agreement with Seaside National Bank and Trust for a
$980,000 loan at 4.25% per annum interest that was used to refinance the
Company’s loan with Bank of America. The obligation is guaranteed by certain
shareholders of the Company. The Company renewed the loan with Seaside National
Bank and Trust during the first quarter of 2014 to extend the maturity date to
December 23, 2015.
August 2008 Unsecured
Promissory Note
On August 20, 2008, the
Company borrowed $1.0 million from a third party pursuant to the terms of an
unsecured Promissory Note and Agreement. Outstanding principal and interest on
the loan, which accrues at the rate of 13.5% per annum, would be payable in one
balloon payment upon the Company’s repayment of the BlueCrest Loan, which was
scheduled to mature in May 2010, however the Company was not obligated to make
payments until BlueCrest Loan is paid off. In the event the Company completes a
private placement of its common stock and/or securities exercisable for or
convertible into its common stock which generates at least $19.0 million of
gross proceeds, the Company may prepay, without penalty, all outstanding
principal and interest due under the loan using the same type of securities
issued in the subject private placement. Because repayment of the loan could
occur within 12 months from the date of the balance sheet, the Company has
classified this loan as short term.
Subject to certain
conditions, at the end of each calendar quarter during the time the loan is
outstanding, the Company may, but is not required to, pay all or any portion of
the interest accrued but unpaid as of such date with shares of its common stock.
In April 2009, as consideration for the authorization to amend certain documents
related to the Note, the Company issued to the Note holder a warrant to purchase
451,053 shares of common stock at an exercise price of $0.5321 per share.
The warrant, which became
exercisable immediately upon issuance, has a ten year term. This warrant had a
fair value of $195,694, which was accounted for as additional paid in capital
and reflected as a component of debt discount and has been fully amortized as
interest expense ratably over the term of the loan.
F-10
In 2013, 1,000,000 shares of common stock were issued to the debt holder, in exchange for $500,000 in principal and $598,125 of
accrued interest relating to a previously issued note resulting in a gain of
$1,078,625. A gain of $1,078,625 was included in the net gain on settlement of
debt and trade payables on the statement of operations for the year ended
December 31, 2013.
On June 16, 2014,
14,539,191 shares of common stock were issued to the debt holder, in exchange
for the remaining $500,000 principal and $81,568 of accrued interest resulting
in a gain of $255,890. A gain of $255,890 was included in the net gain on
settlement of debt and trade payable on the statement of operations for the nine
months ended September 30, 2014.
Hunton & Williams
Notes
At September 30, 2014 and
December 31, 2013, the Company has two outstanding notes payable with interest
at 8% per annum due at maturity. The two notes, $61,150 and $323,822 , are
payable in one balloon payment upon the date the Noteholder provides written
demand, however the Company is not obligated to make payments until the
Northstar (or successor) Loan is paid off.
Asher Notes (During
this year)
During the nine months
ended September 30, 2014, the Company entered into a Securities Purchase
Agreements with Asher Enterprises, Inc. (“Asher”) or affiliates, for the sale of
8% convertible notes in aggregate principal amount of $258,000 (the “Asher
Notes”).
The Asher Notes bear
interest at the rate of 8% per annum. As of the quarter ended September 30,
2014, all interest and principal must be repaid nine months from the issuance
date, with the last note being due May 12, 2015. The Notes are convertible into
common stock, at Asher’s option, at a 45% discount to the average of the three
lowest closing bid prices of the common stock during the 10 trading day period
prior to conversion. The Company has identified the embedded derivatives related
to the Asher Notes.
These embedded derivatives
included certain conversion features and reset provision. The accounting
treatment of derivative financial instruments requires that the Company record
fair value of the derivatives as of the inception date of Asher Notes and to
fair value as of each subsequent reporting date, which at September 30, 2014 was
$201,607. At the inception of the Asher Notes, the Company determined the
aggregate fair value of $480,669 of the embedded derivatives.
The remaining unconverted
principle balance as of September 30, 2014 was 175,500.
Daniel James
Management
During the nine months
ended September 30, 2014, the Company entered into Securities Purchase
Agreements with Daniel James Management (“Daniel”) for the sale of 8% to 9.5%
convertible notes in aggregate principal amount of $85,000 (the “Daniel Notes”).
The Daniel Notes bear
interest at the rate of 8% to 9.5% per annum. As of the quarter ended September
30, 2014, all interest and principal must be repaid one year from the issuance
dated, with the last note being due July 29, 2015. The Daniel Notes are
convertible into common stock, at holder’s option, at a 47% discount to the
average of the three lowest closing bid prices of the common stock during the 10
trading day period prior to conversion. The Company has identified the embedded
derivatives related to the Daniel Note. These embedded derivatives included
certain conversion features and reset provision.
The accounting treatment of
derivative financial instruments requires that the Company record fair value of
the derivatives as of the inception date of Daniel Notes and to fair value as of
each subsequent reporting date which at September 30, 2014 was $66,630. At the
inception of the Daniel Note, the Company determined the aggregate fair value of
$170,270 of the embedded derivatives.
F-11
The remaining unconverted
principle balance as of September 30, 2014 was 50,000.
Fourth Man,
LLC
During the nine months
ended September 30, 2014, the Company entered into Securities Purchase
Agreements with Fourth Man, LLC. (“Fourth Man”), for the sale of an 8% to 9.5%
convertible note in the aggregate principal amount of $75,000 (the “Note”).
The Notes bears interest at
the rate of 8% to 9.5% per annum. As of the quarter ended September 30, 2014,
all interest and principal must be repaid one year from the issuance dated, with
the last note being due August 28, 2015. The Notes are convertible into common
stock, at Fourth Man’s option, at a 47% discount to the average of the three
lowest closing bid prices of the common stock during the 10 trading day period
prior to conversion. The Company has identified the embedded derivatives related
to the Fourth Man Notes. These embedded derivatives included certain conversion
features and reset provision.
The accounting treatment of
derivative financial instruments requires that the Company record fair value of
the derivatives as of the inception date of Fourth Man Notes and to fair value
as of each subsequent reporting date which at September 30, 2014 was $91,319. At
the inception of the Fourth Man Notes, the Company determined the aggregate fair
value of $122,561 of the embedded derivatives.
The fair value of the
embedded derivatives of the Asher, Daniel and Fourth Man Notes, was determined
using the Binomial Option Pricing Model based on the following assumptions: (1)
dividend yield of 0%; (2) expected volatility of 166.07% to 171.91%, (3)
weighted average risk-free interest rate of 0.10% to 0.13%, (4) expected lives
of 0.76 to 1.00 years, and (5) estimated fair value of the Company’s common
stock from $0.0231 to $0.0486 per share. The initial fair value of the embedded
debt derivative of $773,501 was allocated as a debt discount up to the proceeds
of the note ($418,000) with the remainder ($355,501) charged to current period
operations as interest expense. For the three and nine months ended September
30, 2014, the Company amortized an aggregate of $141,977 and $343,592 of debt
discounts to current period operations as interest expense, respectively. For
the three and nine months ended September 30, 2013, the Company amortized
$53,115 and $295,750 of debt discount to operations as interest expense,
respectively.
NOTE 7 — RELATED PARTY
TRANSACTIONS
Advances
As of September 30, 2014
and December 31, 2013, the Company officers and directors have provided advances
in the aggregate of $141,759 and $416,198 respectively, for working capital
purposes. The advances are unsecured, due on demand and non-interest bearing.
During the nine months
ended September 30, 2014, the Company issued an aggregate of 10,232,286 shares
of common stock in settlement of $298,759 of related party advances and
outstanding accounts payable.
Notes payable-related
party
Northstar
Biotechnology Group, LLC
On February 29, 2012, a
note issued to BlueCrest Master Fund Limited was assigned to Northstar
Biotechnology Group, LLC (“Northstar”), owned partly by certain directors and
existing shareholders of the Company, including Dr. William P. Murphy Jr., Dr.
Samuel Ahn and Charles Hart. At the date of the assignment, the principal amount
of the BlueCrest note was $544,267.
F-12
On March 30, 2012, the
Company and Northstar agreed to extend until May 1, 2012 the initial payment
date for any and all required monthly under the Note, such that the first of the
four monthly payments required under the Note will be due and payable on May,
2012 and all subsequent payments will be due on a monthly basis thereafter
commencing on June 1, 2012, and to waive any and all defaults and/or events of
default under the Note with respect to such payments. As of September 30, 2012,
the Company was in default, however, subsequent to September 30, 2012, the
Company renegotiated the terms of the Note, Northstar has agreed to suspend the
requirement of principal payments by the Company and allow payment of
interest-only in common stock.
On September 21, 2012, the
Company issued 5,000,000 common stock purchase warrants to Northstar that was
treated as Additional interest expense upon issuance.
On October 1, 2012, the
Company and Northstar entered into a limited waiver and forbearance agreement
providing a recapitalized new note balance comprised of all sums due Northstar
with a maturity date extended perpetually. The Company agreed to issue 5,000,000
shares of Series A Convertible Preferred Stock and 10,000,000 of common stock in
exchange for $210,000 as payment towards outstanding debt, default interest,
penalties, professional fees outstanding and due Northstar. In addition, the
Company executed a security agreement granting Northstar a lien on all patents,
patent applications, trademarks, service marks, copyrights and intellectual
property rights of any nature, as well as the results of all clinical trials,
know-how for preparing Myoblasts, old and new clinical data, existing approved
trials, all right and title to Myoblasts, clinical trial protocols and other
property rights.
In addition, the Company
granted Northstar a perpetual license on products as described for resale,
relicensing and commercialization outside the United States. In connection with
the granted license, Northstar shall pay the Company a royalty of up to 8% on
revenues generated.
Effective October 1, 2012,
the effective interest rate was 12.85% per annum. The parties agreed, as of
February 28, 2013, to reduce the interest rate to 7% per annum.
In connection with the
consideration paid, Northstar waived, from the effective date through the
earlier of termination or expiration of the agreement, satisfaction of the
obligations as described in the forbearance agreement. In 2012, 5,000,000 shares
of Series A Convertible Preferred Stock were approved to be issued, which was
subsequently increased to 20,000,000 shares of preferred stock as Series A
Convertible Preferred Stock. In addition, the Company is obligated to issue
additional preferred stock equal in lieu of payment of cash of accrued and
unpaid interest on each six month anniversary of the effective date (October 1,
2012). In lieu of the initial two payments in preferred stock, the parties have
determined to modify the voting rights of the Series A Convertible Preferred
Stock from 20 votes per share on matters to be voted on by the common stock
holders to 25 votes per share on matters to be voted on by the common stock
holders and all prior and subsequent payments of interest will be in common
stock. The Company is required to issue additional shares of its common stock
(as amended), in lieu of cash, each six month anniversary of the effective date
for any accrued and unpaid interest.
On April 2, 2014, the
Company issued 274,681 shares of its common stock in lieu of payment in cash of
accrued and unpaid interest of $12,635 due April 1, 2014 per the forbearance
agreement.
On September 17, 2014,
limited waiver and forbearance agreement was amended to provide that the
perpetual license on products as described for resale, relicensing and
commercialization outside the United States was conditioned upon NorthStar
providing certain financing, which financing the Company, in its sole
discretion, could decline and retain the license.
As of September 30, 2014
and December 31, 2013, the principle of this note was $362,000.
Officer and Director
Notes
At September 30, 2014 and
December 31, 2013, the Company has outstanding notes payable to officers and
directors with interest at 8% per annum due at maturity in aggregate $240,000
and $365,000, respectively. The remaining subordinated notes $100,000 and
$140,000 were previously due on November 30, 2012 and June 4, 2011 respectively,
and are unsecured. The Company is not obligated to make payment until Northstar
loan is paid off.
F-13
On September 30, 2013, the
Company issued an aggregate of 15,350,876 shares of its common stock in
settlement of $175,000 of these related party notes payable.
On June 20, 2014, the
Company issued 4,045,796 shares of its common stock in settlement of $125,000 of
related party note payable and accrued interest of $36,832.
On August 1, 2013, the
Company issued an aggregate of $500,000 promissory notes due on demand to
officers and employee in settlement of accrued compensation. The promissory
notes bear interest of 5% per annum and due at various maturity dates. During
the nine months ended September 30, 2014, the Company paid off $240,821 of the
outstanding promissory notes. The principle outstanding balance of these notes
as of September 30, 2014 is $189,813.
On July 1, 2014, the
Company issued an aggregate of $800,000 promissory notes to officers and
employee in settlement of accrued compensation. The promissory notes bear
interest of 5% per annum and due on January 1, 2015.
As of September 30, 2014
and December 31, 2013, the Company officers and directors have outstanding notes
in aggregate of $785,458 and $1,014,477, respectively. The notes are at from
4.75% to 8% per annum and are due upon payoff of the Northstar note payable
described above.
On June 20, 2014, the
Company issued 10,333,475 shares of its common stock in settlement of $300,385
of related party note payable and accrued interest of $112,954.
NOTE 8 — DERIVATIVE
LIABILITIES
Reset
warrants
On October 1, 2012, in
connection with the forbearance agreement with Northstar as discussed in Note 7
above, the Company issued an aggregate of 15,000,000 common stock purchase
warrants to purchase the Company’s common stock with an exercise price of $0.014
per share for ten years with anti-dilutive (reset) provisions.
The Company has identified
embedded derivatives related to the issued warrants. These embedded derivatives
included certain and anti-dilutive (reset) provisions. The accounting treatment
of derivative financial instruments requires that the Company record fair value
of the derivatives as of the inception date and to fair value as of each
subsequent reporting date.
At September 30, 2014, the
fair value of the reset provision of $365,566 was determined using the Binomial
Option Pricing model with the following assumptions: dividend yield: 0%;
volatility: 153.88%; risk free rate: 2.22%; and expected life: 8.00 years. The
Company recorded a gain (loss) on change in derivative liabilities of $35,263
and $(218,711) during the three and nine months ended September 30, 2014,
respectively.
Convertible
notes
In 2013 and the nine months
ended September 30, 2014, the Company issued convertible notes (see Note 6
above).
These notes are convertible
into common stock, at holders’ option, at a discount to the market price of the
Company’s common stock. The Company has identified the embedded derivatives
related to these notes relating to certain anti-dilutive (reset) provisions.
These embedded derivatives included certain conversion features. The accounting
treatment of derivative financial instruments requires that the Company record
fair value of the derivatives as of the inception date of Asher Note and to fair
value as of each subsequent reporting date.
F-14
The fair value of the
embedded derivatives at September 30, 2014, in the amount of $359,556, was
determined using the Binomial Option Pricing Model based on the following
assumptions: (1) dividend yield of 0%;
(2) expected volatility of
153.88%, (3) weighted average risk-free interest rate of 0.02 to 0.13%, (4)
expected lives of 0.24 to 0.91 years, and (5) estimated fair value of the
Company’s common stock of $0.0249 per share. The Company recorded a gain on
change in derivative liabilities of $204,033 and $280,050 during the three and
nine months ended September 30, 2014, respectively.
Based upon ASC 840-15-25 (EITF Issue 00-19, paragraph 11) the Company has
adopted a sequencing approach regarding the application of ASC 815-40 to its
outstanding convertible notes. Pursuant to the sequencing approach, the Company
evaluates its contracts based upon earliest issuance date.
At September 30, 2014, the aggregate derivative liabilities were valued
at $725,122. The Company believes an event under the contract that would create
an obligation to settle in cash or other current assets is remote and has
classified the obligation as a long term liability.
NOTE 9 – STOCKHOLDERS’
EQUITY
Preferred
stock
On August 17, 2012, the board of directors designated 5,000,000 shares of
preferred stock as Series A Convertible Preferred Stock which was increased to
20,000,000 shares of preferred stock as Series A Convertible Preferred Stock
(currently held by Northstar Biotechnology Group, LLC). Each share of preferred
stock is convertible into equal number of common shares at the option of the
holder; entitled to 20 votes on all matters presented to be voted by the holders
of common stock; upon event of liquidation, entitled to amount equal to stated
value plus any accrued and unpaid dividends or other fees before distribution to
junior securities. In lieu of the initial two payments due to Northstar on April
1, 2013 and October 1, 2013, the parties have determined to modify the voting
rights of the Series A Convertible Preferred Stock from 20 votes per share on
matters to be voted on by the common stock holders to 25 votes per share on
matters to be voted on by the common stock holders (see Note 7 above).
Common
stock
Effective May 19, 2014, the Company amended its articles of incorporation
to increase the authorized shares of capital stock of the Company from nine
hundred and fifty million (950,000,000) shares of common stock and twenty
million (20,000,000) shares of preferred stock, both $.001 par value
respectively, to two billion (2,000,000,000) shares of shares of common stock
and twenty million (20,000,000) shares of preferred stock, both $.001 par value
respectively.
During the nine months ended September 30, 2014, the Company issued an
aggregate of 45,232,118 shares of its common stock in settlement of outstanding
accounts payable and accrued expenses. In connection with the issuance, the
Company incurred $105,737 loss in settlement of debt.
During the nine months ended September 30, 2014, the Company issued an
aggregate of 64,434,256 shares of its common stock for the conversion of
$1,897,080 of notes payable and related accrued interest. In connection with the
issuance, the Company incurred $255,890 gain in settlement of debt.
During the nine months ended September 30, 2014, the Company issued an
aggregate of 3,839,832 shares of its common stock for services provided to the
Company.
NOTE 10 — STOCK OPTIONS
AND WARRANTS
Stock
Options
In December 1999, the Board of Directors and shareholders adopted the
1999 Officers and Employees Stock Option Plan, or the Employee Plan, and the
1999 Directors and Consultants Stock Option Plan, or the Director Plan. The
Employee Plan and the Director Plan are collectively referred to herein as the
Plans. The Plans are administered by the Board of Directors and the Compensation
Committee. The objectives of the Plans include attracting and retaining key
personnel by encouraging stock ownership in the Company by such persons. In
February 2010, the Directors & Consultants Plan was amended to extend the
termination date of the Plan to December 1, 2011.
F-15
In April 1, 2013, the Board of Directors approved, subject to shareholder
approval, the establishment of the Bioheart 2013 Omnibus Equity Compensation
Plan, or the “2013 Omnibus Plan”. The 2013 Omnibus Plan reserves up to fifty
million shares of common stock for issuance.
A summary of options at
September 30, 2014 and activity during the nine months then ended is presented
below:
|
|
|
|
|
|
|
|
|
Weighted- |
|
|
|
|
|
|
|
|
|
|
Average |
|
|
|
|
|
|
|
Weighted- |
|
Remaining |
|
|
|
|
|
|
|
Average |
|
Contractual |
|
|
|
|
|
Shares |
|
Exercise Price |
|
Term (in years) |
|
|
Options outstanding at January 1, 2013 |
|
7,853,376 |
|
|
$ |
0.67 |
|
8.2 |
|
|
Granted |
|
17,400,000 |
|
|
$ |
0.016 |
|
9.9 |
|
|
Exercised |
|
— |
|
|
$ |
|
|
|
|
| |
Forfeited/Expired |
|
(1,340,433 |
) |
|
$ |
1.08 |
|
|
|
|
Options outstanding at December 31, 2013 |
|
23,912,943 |
|
|
$ |
0.15 |
|
9.0 |
|
|
Granted |
|
39,648,487 |
|
|
$ |
0.023 |
|
10.0 |
|
|
Exercised |
|
— |
|
|
|
|
|
|
|
|
Forfeited/Expired |
|
(74,443 |
) |
|
$ |
4.8 |
|
|
|
|
Options outstanding at September 30, 2014 |
|
63,486,987 |
|
|
$ |
0.063 |
|
9.1 |
|
|
Options exercisable at September 30, 2014 |
|
24,576,000 |
|
|
$ |
0.13 |
|
8.7 |
|
|
Available for grant at September
30, 2014 |
|
42,951,513 |
|
|
|
|
|
|
|
The following information
applies to options outstanding and exercisable at September 30, 2014:
|
|
|
Options Outstanding |
|
Options Exercisable |
|
|
|
|
|
|
Weighted- |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Average |
|
Weighted- |
|
|
|
Weighted- |
|
|
|
|
|
|
Remaining |
|
Average |
|
|
|
Average |
|
|
|
|
|
|
Contractual |
|
Exercise |
|
|
|
Exercise |
|
| |
|
|
Shares |
|
Term |
|
Price |
|
Shares |
|
Price |
|
|
$0.00 – $0.70 |
|
62,938,487 |
|
9.2 |
|
$ |
0.03 |
|
24,027,500 |
|
$ |
0.03 |
|
|
$0.71 – $1.28 |
|
149,930 |
|
4.0 |
|
$ |
0.81 |
|
149,930 |
|
$ |
0.81 |
|
|
$5.25 – $5.67 |
|
373,858 |
|
1.6 |
|
$ |
5.55 |
|
373,858 |
|
$ |
5.55 |
|
|
$7.69 |
|
24,712 |
|
1.8 |
|
$ |
7.69 |
|
24,712 |
|
$ |
7.69 |
|
|
|
|
63,486,987 |
|
9.1 |
|
$ |
0.063 |
|
24,576,000 |
|
$ |
0.13 |
|
On February 24, 2014, the Company issued an aggregate 15,000,000 options
to purchase the Company’s common stock at $0.019 per share to officers, vesting
at 25% immediately and the remainder over approximately 42 months, exercisable
over 10 years. The aggregate fair value of $282,597, determined using the Black
Scholes option pricing model with the following assumptions: Dividend yield: 0%;
Volatility: 163.63% and Risk free rate: 2.75% .
On February 24, 2014, the Company issued an aggregate 4,800,000 options
to purchase the Company’s common stock at $0.019 per share to officers, vesting
immediately and exercisable over 10 years. The aggregate fair value of $90,431,
determined using the Black Scholes option pricing model with the following
assumptions: Dividend yield: 0%; Volatility: 163.63% and Risk free rate: 2.75% .
F-16
On May 12, 2014, the Company issued an aggregate 4,848,487 options to
purchase the Company’s common stock at $0.0272 per share to officers and
employees, vesting over four years at anniversary and exercisable over 10 years.
The aggregate fair value of $130,135, determined using the Black Scholes option
pricing model with the following assumptions: Dividend yield: 0%; Volatility:
161.36% and Risk free rate: 2.66% .
On August 1, 2014, the Company issued an aggregate 15,000,000 options to
purchase the Company’s common stock at $0.02694 per share to officers and
employees, vesting at 25% immediately and 75% over three years at anniversary
and exercisable over 10 years. The aggregate fair value of $391,798, determined
using the Black Scholes option pricing model with the following assumptions:
Dividend yield: 0%; Volatility: 168.62% and Risk free rate: 2.52% .
The fair value of all options vesting during the three and nine months
ended September 30, 2014 of $164,247 and $407,616, respectively, was charged to
current period operations.
Warrants
A
summary of common stock purchase warrants at September 30, 2014 and activity
during the three months then ended is presented below:
|
|
|
|
|
|
|
|
|
Weighted- |
|
|
|
|
|
|
|
|
|
|
Average |
|
|
|
|
|
|
|
Weighted- |
|
Remaining |
|
|
|
|
|
|
|
Average |
|
Contractual |
|
|
|
|
|
|
|
Exercise |
|
Term
(in |
|
|
|
|
|
Shares |
|
Price |
|
years) |
|
|
Outstanding at January 1, 2013 |
|
74,073,322 |
|
|
$ |
0.37 |
|
4.5 |
|
| |
Issued |
|
50,350,536 |
|
|
$ |
0.16 |
|
9.2 |
|
|
Exercised |
|
— |
|
|
$ |
0.00 |
|
|
|
| |
Forfeited |
|
(6,345,002 |
) |
|
$ |
0.38 |
|
|
|
|
Outstanding at December 31, 2013 |
|
118,078,856 |
|
|
$ |
0.22 |
|
6.3 |
|
|
Issued |
|
36,556,846 |
|
|
$ |
0.02 |
|
7.4 |
|
|
Exercised |
|
(11,918,181 |
) |
|
$ |
0.01 |
|
|
|
|
Expired |
|
(6,765,430 |
) |
|
$ |
0.11 |
|
|
|
|
Outstanding at September 30, 2014 |
|
135,952,091 |
|
|
$ |
0.19 |
|
6.1 |
|
|
Exercisable at September 30,
2014 |
|
126,143,578 |
|
|
$ |
0.11 |
|
6.2 |
|
The following information
applies to common stock purchase warrants outstanding and exercisable at
September 30, 2014:
|
|
|
Warrants Outstanding |
|
Warrants Exercisable |
|
| |
|
|
|
|
Weighted- |
|
|
|
|
|
|
|
|
|
|
|
Average |
|
Weighted- |
|
|
|
Weighted- |
|
| |
|
|
|
|
Remaining |
|
Average |
|
|
|
Average |
|
|
|
|
|
|
Contractual |
|
Exercise |
|
|
|
Exercise |
|
|
|
|
Shares |
|
Term |
|
Price |
|
Shares |
|
Price |
|
|
$0.01 – $0.50 |
|
129,702,958 |
|
6.1 |
|
$ |
0.02 |
|
121,438,895 |
|
$ |
0.02 |
|
|
$0.52 – $0.68 |
|
2,699,675 |
|
4.6 |
|
$ |
0.58 |
|
2,699,675 |
|
$ |
0.58 |
|
|
$0.70 – $1.62 |
|
848,176 |
|
5.3 |
|
$ |
0.71 |
|
848,176 |
|
$ |
0.71 |
|
|
$5.67 – $7.69 |
|
2,701,282 |
|
8.1 |
|
$ |
7.55 |
|
1,156,832 |
|
$ |
7.35 |
|
|
|
|
135,952,091 |
|
6.1 |
|
$ |
0.19 |
|
126,143,578 |
|
$ |
0.11 |
|
F-17
In conjunction with the authorized issuance of common stock, the Company
granted 24,556,846 common stock purchase warrants during the nine months ended
September 30, 2014.
During the nine months ended September 30, 2014, the Company issued an
aggregate of 8,000,000 warrants in connection with a joint venture agreement
dated March 10, 2014. The warrants are exercisable at $0.0217 for four years
vesting from June 8, 2014 through March 10, 2016. During the three and nine
months ended September 30, 2014, the Company charged $15,762 and $95,018 to
current period operations for the vesting portion.
During the nine months ended September 30, 2014, the Company issued an
aggregate of 4,000,000 warrants in connection with use of certain intellectual
property. The warrants are exercisable at $0.0481 for four years vesting from
July 6, 2014 through April 6, 2017. During the three and nine months ended
September 30, 2014, the Company charged $9,005 and $41,819 to current period
operations for the vesting portion.
NOTE 11 — COMMITMENTS
AND CONTINGENCIES
Revenue Share
Agreement
On March 10, 2014, the Company entered into a profits sharing agreement
(the Agreement) with Global Stem Cells, Group, LLC and its subsidiaries whereby
both parties will participate in marketing for obtaining patients and provide
physician training for stem cell treatments under the names of “Regenestem” and
“Stem Cell Training”, respectively. In addition, each party will be responsible
for selling equipment and kit to existing and previous customers. A fee will be
paid to Global Stem Cells Group based on revenue loss with costs of accounting
for patients and physician training and 30 days with sales of equipment and
kits.
In consideration of Global Stem Cell Group, LLC’s participation, the
Company issued an aggregate of 8,000,000 warrants to purchase the Company’s
common stock for four years at $0.0217 per share with 2,000,000 warrants vesting
90 days from the effective date, 2,000,000 vesting on each anniversary date for
three years. During the three and nine months ended September 30, 2014, the
Company charged $15,762 and $95,018 to current period operations for the vesting
portion.
Joint
Venture
We announced a joint venture in South Africa and the facilities called
“South African Stem Cell Institute” were successfully opened in September, 2014
with the intention to retain a 49% ownership of the new entity. As of September
30, 2014, however, there was no formal legal entity established and no formal
operating agreements for this joint venture. In addition, the Company has not
yet incurred any material expense associated with this venture. Management has
concluded that as of September 30, 2014 this announcement is not material to the
Company’s financial statements.
William Beaumont
Hospital
In June 2000, the Company entered into an agreement with William Beaumont
Hospital, or WBH, pursuant to which WBH granted to the Company worldwide,
exclusive, non-sublicenseable license to two U.S. method patents covering the
inducement of human adult myocardial cell proliferation in vitro, or the WBH IP.
The term of the agreement is for the life of the patents, which expire in 2015.
The Company did not use this license in any of our technologies. The Company had
not made any payments to WBH other than the initial payment to acquire the
license. The Company has received confirmation from WBH that it has no
obligation under the patent license agreement and WBH agreed to terminate the
patent license agreement. (See Note 4)
Accordingly, the Company has recognized approximately $2,122,130 in
settlement of debt which represents the accumulative accrual and related
interest from past years under the 2000 patent license agreement.
F-18
Employment
agreements
On July 28, 2014, the Company’s Board of Directors approved the 2014/2015
salary for Mike Tomas, Chief Executive Officer, at $525,000 per year, beginning
July 1, 2014 with an incentive bonus ranging from $150,000 to $500,000. In
addition, the Board of Directors will grant Mr. Tomas options to be determined
on or before June 30, 2015. The Company’s Board of Directors approved a bonus of
$500,000 and options to acquire 10,000,000 shares of the Company’s common stock
for ten years with four year vesting and a cashless exercise provision at an
exercise price equal to the five day average closing price of the Company’s
common stock as of August 1, 2014. The cash bonus may be paid in the form a six
month promissory note.
On July 28, 2014, the Company’s Board of Directors approved the 2014/2015
salary for Kristin Comella, Chief Scientific Officer, at $250,000 per year,
beginning July 1, 2014 with an incentive bonus ranging from $100,000 to
$300,000. In addition, the Board of Directors will grant Ms. Comella options to
be determined on or before June 30, 2015. The Company’s Board of Directors
approved a bonus of $300,000 and options to acquire 5,000,000 shares of the
Company’s common stock for ten years with four year vesting and a cashless
exercise provision at an exercise price equal to the five day average closing
price of the Company’s common stock as of August 1, 2014. The cash bonus may be
paid in the form a six month promissory note.
Litigation
The Company is subject to other legal proceedings that arise in the
ordinary course of business. In the opinion of management, as of September 30,
2014, the amount of ultimate liability with respect to such matters, if any, in
excess of applicable insurance coverage, is not likely to have a material impact
on the Company’s business, financial position, results of operations or
liquidity. However, as the outcome of litigation and other claims is difficult
to predict significant changes in the estimated exposures could exist.
Consulting
agreements
On November 20, 2013, the Company entered into an investment banking
agreement with Cassel Salpeter &
Co. (“CSC”), who will act as exclusive third party financial advisor in
connection with investment banking matters. The term of the Investment Banking
Agreement shall be for a period of twenty four months unless terminated or
extended in accordance with its terms. For these services, CSC will receive a
one-time $25,000 fee, $5,000 monthly
fees and 5,207,630 ten year common
stock purchase warrants, exercisable at $.0113 and applicable consideration in
the event the closing of a Mezzanine Financing consisting of non-convertible
subordinated debt and/or sale of equity securities. The Company will also
reimburse CSC for its reasonable out-of-pocket expenses associated with the
services provided pursuant to the Investment Banking Agreement. As of September
30, 2014 and December 31, 2013, the Company accrued $49,234 and $32,424 under
the agreement, respectively.
NOTE 12 — FAIR VALUE
MEASUREMENT
The Company adopted the provisions of Accounting Standards Codification
subtopic 825-10, Financial Instruments (“ASC 825-10”) on January 1, 2008. ASC
825-10 defines fair value as the price that would be received from selling an
asset or paid to transfer a liability in an orderly transaction between market
participants at the measurement date. When determining the fair value
measurements for assets and liabilities required or permitted to be recorded at
fair value, the Company considers the principal or most advantageous market in
which it would transact and considers assumptions that market participants would
use when pricing the asset or liability, such as inherent risk, transfer
restrictions, and risk of nonperformance. ASC 825-10 establishes a fair value
hierarchy that requires an entity to maximize the use of observable inputs and
minimize the use of unobservable inputs when measuring fair value. ASC 825-10
establishes three levels of inputs that may be used to measure fair value:
Level 1 – Quoted prices in active markets for identical assets or
liabilities.
Level 2 – Observable inputs other than Level 1 prices such as quoted
prices for similar assets or liabilities; quoted prices in markets with
insufficient volume or infrequent transactions (less active markets); or
model-derived valuations in which all significant inputs are observable or can
be derived principally from or corroborated by observable market data for
substantially the full term of the assets or liabilities.
F-19
Level 3 – Unobservable inputs to the valuation methodology that are
significant to the measurement of fair value of assets or liabilities.
All items required to be recorded or measured on a recurring basis are
based upon level 3 inputs.
To the extent that valuation is based on models or inputs that are less
observable or unobservable in the market, the determination of fair value
requires more judgment. In certain cases, the inputs used to measure fair value
may fall into different levels of the fair value hierarchy. In such cases, for
disclosure purposes, the level in the fair value hierarchy within which the fair
value measurement is disclosed and is determined based on the lowest level input
that is significant to the fair value measurement.
Upon adoption of ASC 825-10, there was no cumulative effect adjustment to
beginning retained earnings and no impact on the financial statements.
The carrying value of the Company’s cash and cash equivalents, accounts
receivable, accounts payable, short-term borrowings (including convertible notes
payable), and other current assets and liabilities approximate fair value
because of their short-term maturity.
As of September 30, 2014 or December 31, 2013, the Company did not have
any items that would be classified as level 1 or 2 disclosures.
The Company recognizes its derivative liabilities as level 3 and values
its derivatives using the methods discussed in notes 7 and 9. While the Company
believes that its valuation methods are appropriate and consistent with other
market participants, it recognizes that the use of different methodologies or
assumptions to determine the fair value of certain financial instruments could
result in a different estimate of fair value at the reporting date. The primary
assumptions that would significantly affect the fair values using the methods
discussed in Notes 7 and 9 are that of volatility and market price of the
underlying common stock of the Company.
As of September 30, 2013 and December 31, 2013, the Company did not have
any derivative instruments that were designated as hedges.
The derivative liability as of September 30, 2014, in the amount of
$725,122 has a level 3 classification.
The following table provides a summary of changes in fair value of the
Company’s Level 3 financial liabilities as of September 30, 2014:
|
|
|
Excess |
|
|
|
|
|
|
|
|
|
|
|
|
Share |
|
Warrant |
|
Debt |
|
| |
|
|
Derivative |
|
Liability |
|
Derivative |
|
|
Balance, December 31, 2012 |
|
$ |
390,048 |
|
|
|
221,179 |
|
|
$ |
— |
|
|
|
Total (gains) losses |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Initial fair value of debt derivative at note
issuance |
|
|
|
|
|
|
— |
|
|
|
673,219 |
|
|
|
Initial fair value of derivative relating to reset
warrants |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
Mark-to-market at December 31, 2013: |
|
|
84.906 |
|
|
|
(74,324 |
) |
|
|
(39,761 |
) |
|
| |
Transfers out of Level 3 upon increase in authorized
shares |
|
|
(474,954 |
) |
|
|
— |
|
|
|
— |
|
|
|
Transfers out of Level 3 upon conversion and settlement of
notes |
|
|
|
|
|
|
|
|
|
|
(376,502 |
) |
|
|
Balance, December 31, 2013 |
|
$ |
— |
|
|
$ |
146,855 |
|
|
$ |
256,956 |
|
|
|
Total (gains) losses |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Initial fair value of debt derivative at note
issuance |
|
|
— |
|
|
|
— |
|
|
|
773,500 |
|
|
|
Mark-to-market at September 30, 2014: |
|
|
— |
|
|
|
218,711 |
|
|
|
(280,050 |
) |
|
|
Transfers out of Level 3 upon conversion of notes
payable |
|
|
— |
|
|
|
— |
|
|
|
(390,850 |
) |
|
|
Balance, September 30, 2014 |
|
$ |
— |
|
|
$ |
365,566 |
|
|
$ |
359,556 |
|
|
|
Net
(Loss) Gain for the period included in earnings relating to the |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
liabilities held at September 30,
2014 |
|
$ |
— |
|
|
$ |
(218,711 |
) |
|
$ |
280,050 |
|
|
F-20
Fluctuations in the Company’s stock price are a primary driver for the
changes in the derivative valuations during each reporting period. The Company’s
stock price increased approximately 149% from December 31, 2013 to September 30,
2014. As the stock price increases for each of the related derivative
instruments, the value to the holder of the instrument generally increases,
therefore increasing the liability on the Company’s balance sheet. Additionally,
stock price volatility is one of the significant unobservable inputs used in the
fair value measurement of each of the Company’s derivative
instruments.
The simulated fair value of these liabilities is sensitive to changes in
the Company’s expected volatility. Increases in expected volatility would
generally result in a higher fair value measurement.
NOTE 13 — SUBSEQUENT
EVENTS
Subsequent
financing
KBM
Worldwide
On October 6, 2014, the Company entered into a Securities Purchase
Agreement with KBM Worldwide, Inc., for the sale of an 8% convertible note in
the principal amount of $38,000 (the “Note”).
The Note bears interest at the rate of 8% per annum. All interest and
principal must be repaid on July 8, 2015; The Note is convertible into common
stock, at holder’s option, at a 45% discount to the lowest daily trading price
of the common stock during the 10 trading day period prior to conversion. In the
event the Company prepays the Note in full, the Company is required to pay off
all principal and accrued interest at 150%, and any other amounts.
Daniel James
Management
On October 3, 2014, the Company entered into a Securities Purchase
Agreement with Daniel James Management, Inc., for the sale of a 9.5% convertible
note in the principal amount of $25,000 (the “Note”).
The Note bears interest at the rate of 9.5% per annum. All interest and
principal must be repaid on October 2, 2015, 2015. The Note is convertible into
common stock, at holder’s option, at a 47% discount to the lowest daily trading
price of the common stock during the 10 trading day period prior to conversion.
In the event the Company prepays the Note in full, the Company is required to
pay off all principal and accrued interest at 150%, and any other amounts.
Magna Equities,
LLC
On October 7, 2014, the Company entered into a
securities purchase agreement (the “Purchase Agreement”) with Magna Equities II, LLC, a New York limited liability company
(“Magna”). The Purchase Agreement provides that, upon the
terms and subject to the conditions set forth therein, Magna shall purchase from
the Company, a senior convertible note with an initial principal amount of
$307,500 (the “Convertible
Note”) for a purchase price of
$205,000 (an approximately 33.33% original issue discount). Pursuant to the
Purchase Agreement, the Company issued the Convertible Note to Magna. The
Convertible Note matures on August 7, 2015 and, in addition to the approximately
33.33% original issue discount, accrues interest at the rate of 12% per
annum.
The Convertible Note is convertible at any time, in whole or in part, at
Magna’s option into shares of the Company’s common stock, par value $0.01 per
share (the “Common
Stock”), at a fixed conversion
price of $0.01035 per share. $40,000 of the outstanding principal amount of the
Convertible Note (together with any accrued and unpaid interest with respect to
such portion of the principal amount) shall be automatically extinguished
(without any cash payment by the Company) under certain conditions described in
the Purchase Agreement. In connection with the execution of the Purchase
Agreement, the Company and Magna also entered into a registration rights
agreement (the “Registration
Rights Agreement”). Pursuant to
the Registration Rights Agreement, the Company has agreed to file an initial
registration statement with the SEC to register the resale of the Common Stock
into which the Convertible Note may be converted,
F-21
On October 23, 2014, the Company entered into a
common stock purchase agreement (the “Purchase Agreement”) with Magna Equities II, LLC, a New York limited liability company (the
“Investor”). The Purchase Agreement provides that, upon the
terms and subject to the conditions set forth therein, the Investor is committed
to purchase up to $3,000,000 (the “Total Commitment”) worth
of the Company’s common stock, $0.001 par value (the “Shares”), over the 24-month term of the Purchase Agreement. In connection with
the execution of the Purchase Agreement, on the Closing Date, the Company and
the Investor also entered into a registration rights agreement.
The Company paid to the Investor a commitment fee for entering into the
Purchase Agreement equal to $150,000 (or 5.0% of the Total Commitment under the
Purchase Agreement) in the form of 9,109,128 restricted shares of the Company’s
common stock, calculated using a per share price of $0.016467.
Subsequent
issuances
On October 3, 2014, the Company issued 514,886 shares of its common stock
as payment of $70,521 interest on its Northstar (related party) debt.
In October 2014, the Company issued 1,818,182 shares of its common stock
in settlement of $20,000 of convertible debt.
In October 2014, the Company issued 1,293,103 shares of its common stock
in settlement of $15,000 of convertible debt.
In October 2014, the Company issued 2,260,764 shares of its common stock
in settlement of $18,000 of convertible debt and accrued interest of $2,120.
In October 2014, the Company issued 552,846 shares of its common stock in
settlement of $5,500 of convertible debt and accrued interest of
$1,300.
In October 2014, the Company issued an aggregate 2,773,549 shares of
common stock for consulting services.
In October 2014, the Company issued 538,875 shares of commons stock in
settlement of accounts payable.
F-22
REPORT OF INDEPENDENT
REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors
and
Stockholders of Bioheart, Inc.
13794 NW 4th Street, Suite
212,
Sunrise, Florida 33325
We have audited the
accompanying balance sheets of Bioheart, Inc. (the “Company”) (a development
stage company) as of December 31, 2013 and 2012, and the related statements of
operations, stockholders’ deficit, and cash flows for each of the years then
ended. The Company’s management is responsible for these financial statements.
Our responsibility is to express an opinion on these financial statements based
on our audits.
We conducted our audits in
accordance with the standards of the Public Company Accounting Oversight Board
(United States). Those standards require that we plan and perform the audit to
obtain reasonable assurance about whether the financial statements are free of
material misstatement. The company is not required to have, nor were we engaged
to perform, an audit of its internal control over financial reporting. Our audit
included consideration of internal control over financial reporting as a basis
for designing audit procedures that are appropriate in the circumstances, but
not for the purpose of expressing an opinion on the effectiveness of the
company’s internal control over financial reporting. Accordingly, we express no
such opinion. An audit also includes examining, on a test basis, evidence
supporting the amounts and disclosures in the financial statements, assessing
the accounting principles used and significant estimates made by management, as
well as evaluating the overall financial statement presentation. We believe that
our audits provide a reasonable basis for our opinion.
In our opinion, the
financial statements referred to above present fairly, in all material respects,
the financial position of Bioheart, Inc. (a development stage enterprise) as of
December 31, 2013 and 2012, and the results of its operations and its cash flows
for each of the two years ended December 31, 2013.
The accompanying financial
statements have been prepared assuming the Company will continue as a going
concern. As discussed in Note 2 to the financial statements, the Company is in
the development stage, and has incurred net losses of $118,180,983 since
inception. In addition, as of December 31, 2013 the Company’s current
liabilities exceed its current assets by $13,362,480. These conditions raise
substantial doubt about the Company’s ability to continue as a going concern.
The financial statements do not include any adjustments that might result from
the outcome of this uncertainty.
/s/ Fiondella, Milone and LaSaracina LLP
Glastonbury, Connecticut
March 24, 2014
F-23
BIOHEART, INC.
(a
development stage company)
BALANCE SHEETS
DECEMBER 31, 2013 AND 2012
|
2013 |
|
2012 |
| ASSETS |
|
|
|
|
|
|
|
| Current assets: |
|
|
|
|
|
|
|
| Cash
and cash equivalents |
$ |
46,227 |
|
|
$ |
- |
|
| Accounts receivable, net |
|
19,913 |
|
|
|
1,342 |
|
| Inventory |
|
- |
|
|
|
62,953 |
|
| Prepaid and other |
|
784 |
|
|
|
41,533 |
|
| Total
current assets |
|
66,924 |
|
|
|
105,828 |
|
| |
| Property and equipment, net |
|
9,055 |
|
|
|
1,820 |
|
| |
| Other assets |
|
10,160 |
|
|
|
54,662 |
|
| |
| Total assets |
$ |
86,139 |
|
|
$ |
162,310 |
|
| |
| LIABILITIES AND STOCKHOLDERS' DEFICIT |
|
|
|
|
|
|
|
| Current liabilities: |
|
|
|
|
|
|
|
| Bank
overdraft |
$ |
- |
|
|
$ |
89 |
|
| Accounts payable |
|
2,382,267 |
|
|
|
2,556,043 |
|
| Accrued expenses |
|
4,480,335 |
|
|
|
4,731,488 |
|
| Advances, related party |
|
416,198 |
|
|
|
313,448 |
|
| Deposits |
|
478,286 |
|
|
|
465,286 |
|
| Subordinated debt, related party |
|
1,500,000 |
|
|
|
1,500,000 |
|
| Notes payable, related party |
|
2,241,477 |
|
|
|
2,215,324 |
|
| Notes payable, net of debt discount |
|
1,930,841 |
|
|
|
2,364,972 |
|
| Total
current liabilities |
|
13,429,404 |
|
|
|
14,146,650 |
|
| |
| Long
term debt: |
|
|
|
|
|
|
|
| Derivative liability |
|
403,811 |
|
|
|
611,227 |
|
| |
| Stockholders' deficit: |
|
|
|
|
|
|
|
| Preferred stock, par value $0.001; 20,000,000
and 5,000,000 shares |
|
|
|
|
|
|
|
| authorized as of December 31, 2013 and 2012,
respectively, 20,000,000 |
|
|
|
|
|
|
|
| and
-0- issued and outstanding as of December 31, 2013 and 2012, |
|
|
|
|
|
|
|
| respectively |
|
20,000 |
|
|
|
- |
|
| Common stock, par value $0.001; 950,000,000 and
195,000,000 shares |
|
|
|
|
|
|
|
| authorized as of December 31, 2013 and 2012,
respectively, 379,787,745 |
|
|
|
|
|
|
|
| and
182,062,802 shares issued and outstanding as of December 31,
2013 |
|
|
|
|
|
|
|
| and
2012, respectively |
|
379,788 |
|
|
|
182,063 |
|
| Additional paid in capital |
|
103,819,119 |
|
|
|
100,260,094 |
|
| Common stock subscription |
|
215,000 |
|
|
|
- |
|
| Deficit accumulated during development
stage |
|
(118,180,983 |
) |
|
|
(115,037,724 |
) |
| Total
stockholders' deficit |
|
(13,747,076 |
) |
|
|
(14,595,567 |
) |
| |
| Total liabilities and stockholders'
deficit |
$ |
86,139 |
|
|
$ |
162,310 |
|
See the accompanying notes
to these financial statements
F-24
BIOHEART, INC.
(a
development stage company)
STATEMENTS OF OPERATIONS
|
|
|
|
|
|
|
|
|
|
From August
12, |
|
|
|
|
|
|
|
|
|
|
1999 (date
of |
|
|
Year ended
December 31, |
|
Inception)
to |
|
|
2013 |
|
2012 |
|
|
December 31,
2013 |
|
|
|
|
|
|
|
|
|
|
(unaudited) |
| Revenue |
|
$ |
96,085 |
|
|
$ |
61,109 |
|
|
$ |
1,365,725 |
|
| |
| Cost of
sales |
|
|
30,831 |
|
|
|
1,070 |
|
|
|
582,735 |
|
| Gross profit |
|
|
65,254 |
|
|
|
60,039 |
|
|
|
782,990 |
|
| |
| Operating
expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
| Research and
development |
|
|
626,983 |
|
|
|
401,941 |
|
|
|
65,320,035 |
|
| Marketing,
general and administrative |
|
|
2,267,831 |
|
|
|
2,178,352 |
|
|
|
39,105,667 |
|
| Impairment
of investment |
|
|
- |
|
|
|
- |
|
|
|
58,695 |
|
| Depreciation
and amortization |
|
|
2,190 |
|
|
|
14,589 |
|
|
|
900,104 |
|
| Total operating
expenses |
|
|
2,897,004 |
|
|
|
2,594,882 |
|
|
|
105,384,501 |
|
| |
| Net loss
from operations |
|
|
(2,831,750 |
) |
|
|
(2,534,843 |
) |
|
|
(104,601,511 |
) |
| |
| Other income
(expenses): |
|
|
|
|
|
|
|
|
|
|
|
|
| Development
revenues |
|
|
- |
|
|
|
- |
|
|
|
117,500 |
|
| |
| Gain on
settlement of debt |
|
|
1,023,439 |
|
|
|
- |
|
|
|
1,023,439 |
|
| Gain on
change of fair value of derivative |
|
|
|
|
|
|
|
|
|
|
|
|
| liability |
|
|
29,179 |
|
|
|
119,795 |
|
|
|
123,948 |
|
| Interest
income |
|
|
- |
|
|
|
- |
|
|
|
762,277 |
|
| Other
income |
|
|
73,756 |
|
|
|
18,234 |
|
|
|
344,821 |
|
| |
| Interest
expense |
|
|
(1,437,883 |
) |
|
|
(1,619,653 |
) |
|
|
(15,951,457 |
) |
| |
| Total other income
(expenses) |
|
|
(311,509 |
) |
|
|
(1,481,624 |
) |
|
|
(13,579,472 |
) |
| |
| Net loss
before income taxes |
|
|
(3,143,259 |
) |
|
|
(4,016,467 |
) |
|
|
(118,180,983 |
) |
| |
| Income taxes
(benefit) |
|
|
- |
|
|
|
- |
|
|
|
- |
|
| |
| NET
LOSS |
|
$ |
(3,143,259 |
) |
|
$ |
(4,016,467 |
) |
|
$ |
(118,180,983 |
) |
| |
| Net loss per
common share, basic and diluted |
|
$ |
(0.01 |
) |
|
$ |
(0.03 |
) |
|
|
|
|
| |
| Weighted
average number of common shares |
|
|
|
|
|
|
|
|
|
|
|
|
| outstanding,
basic and diluted |
|
|
251,828,970 |
|
|
|
146,463,828 |
|
|
|
|
|
See the accompanying notes
to these financial statements
F-25
BIOHEART, INC.
(a
development stage company)
STATEMENT OF
STOCKHOLDERS'
DEFICIT
FROM AUGUST 12, 1999
(DATE OF INCEPTION)
THROUGH DECEMBER 31, 2013
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deficit |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated |
|
|
|
|
|
|
|
|
Preferred |
|
|
|
Additional |
|
|
|
|
|
|
|
|
During |
|
|
|
|
|
|
|
|
stock |
|
Common
stock |
|
Paid
in |
|
Deferred |
|
Subscription |
|
Development |
|
|
|
|
|
|
Shares |
|
Amount |
|
Shares |
|
Amount |
|
Capital |
|
Compensation |
|
Receivable |
|
Stage |
|
Total |
| Balance,
August 12, 1999 (date of inception) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| (unaudited) |
|
- |
|
$ |
- |
|
- |
|
$ |
- |
|
$ |
- |
|
|
$ |
- |
|
|
$ |
- |
|
$ |
- |
|
|
$ |
|
|
| Issuance of common stock |
|
- |
|
|
- |
|
4,324,458 |
|
|
4,324 |
|
|
395,676 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
400,000 |
|
| Stock based
compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
98,000 |
|
|
|
(98,000 |
) |
|
|
- |
|
|
- |
|
|
|
- |
|
| Amortization of stock based
compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
49,000 |
|
|
|
- |
|
|
- |
|
|
|
49,000 |
|
| Net
loss |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
(903,290 |
) |
|
|
(903,290 |
) |
| Balance, December 31, 1999
(unaudited) |
|
- |
|
|
- |
|
4,324,458 |
|
|
4,324 |
|
|
493,676 |
|
|
|
(49,000 |
) |
|
|
- |
|
|
(903,290 |
) |
|
|
(454,290 |
) |
| Issuance of
common stock, net of issuance costs of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $61,905 |
|
- |
|
|
- |
|
1,493,575 |
|
|
1,494 |
|
|
9,607,201 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
9,608,695 |
|
| Stock based compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
2,559,000 |
|
|
|
(2,559,000 |
) |
|
|
- |
|
|
- |
|
|
|
- |
|
| Fair value
of warrants issued in exchange for |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| licenses and
intellectual property |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
5,220,000 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
5,220,000 |
|
| Amortization of stock based
compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
1,080,692 |
|
|
|
- |
|
|
- |
|
|
|
1,080,692 |
|
| Contributed
capital |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
1,050,000 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
1,050,000 |
|
| Common stock issued in exchange for
services |
|
- |
|
|
- |
|
7,964 |
|
|
8 |
|
|
51,993 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
52,001 |
|
| Net
loss |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
(14,113,93 |
) |
|
|
(14,113,933 |
) |
| Balance, December 31, 2000
(unaudited) |
|
- |
|
|
- |
|
5,825,997 |
|
|
5,826 |
|
|
18,981,870 |
|
|
|
(1,527,308 |
) |
|
|
- |
|
|
(15,017,22 |
) |
|
|
2,443,165 |
|
| Issuance of
common stock, net of issuance costs of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| $98,996 |
|
- |
|
|
- |
|
985,667 |
|
|
986 |
|
|
6,282,018 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
6,283,004 |
|
| Stock based compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
779,000 |
|
|
|
(779,000 |
) |
|
|
- |
|
|
- |
|
|
|
- |
|
| Amortization
of stock based compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
1,523,000 |
|
|
|
- |
|
|
- |
|
|
|
1,523,000 |
|
| Conversion of contributed capital to common
stock |
|
- |
|
|
- |
|
81,084 |
|
|
81 |
|
|
(81 |
) |
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
- |
|
| Common stock
issued in exchange for services |
|
- |
|
|
- |
|
8,291 |
|
|
8 |
|
|
53,993 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
54,001 |
|
| Net loss |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
(8,173,464 |
) |
|
|
(8,173,464 |
) |
| |
| Balance,
December 31, 2001 (unaudited) |
|
- |
|
$ |
- |
|
6,901,039 |
|
$ |
6,901 |
|
$ |
26,096,800 |
|
|
$ |
(783,308 |
) |
|
$ |
- |
|
$ |
(23,190,6 |
) |
|
$ |
2,129,706 |
|
F-26
BIOHEART, INC.
(a
development stage company)
STATEMENT OF
STOCKHOLDERS'
DEFICIT
FROM AUGUST 12, 1999
(DATE OF INCEPTION)
THROUGH DECEMBER 31, 2013
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deficit |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Additional |
|
|
|
|
|
|
|
|
During |
|
|
|
|
|
|
Preferred
stock |
|
Common
stock |
|
Paid
in |
|
Deferred |
|
Subscription |
|
Development |
|
|
|
|
|
|
Shares |
|
Amount |
|
Shares |
|
Amount |
|
Capital |
|
Compensation |
|
Receivable |
|
Stage |
|
Total |
| Balance,
December 31, 2001 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| (unaudited) |
|
- |
|
$ |
- |
|
6,901,039 |
|
$ |
6,901 |
|
$ |
26,096,800 |
|
|
$ |
(783,308 |
) |
|
$ |
- |
|
$ |
(23,190,687 |
) |
|
$ |
2,129,706 |
|
| |
| Issuance of
common stock |
|
- |
|
|
- |
|
1,092,883 |
|
|
1,093 |
|
|
7,075,105 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
7,076,198 |
|
| |
| Stock based
compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
143,521 |
|
|
|
(143,521 |
) |
|
|
- |
|
|
- |
|
|
|
- |
|
| Amortization
of stock based |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| compensation
|
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
613,083 |
|
|
|
- |
|
|
- |
|
|
|
613,083 |
|
| Common stock
issued in exchange for |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| services |
|
- |
|
|
- |
|
35,137 |
|
|
35 |
|
|
227,468 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
227,503 |
|
| |
| Net
loss |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
(9,257,954 |
) |
|
|
(9,257,954 |
) |
| Balance,
December 31, 2002 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| (unaudited) |
|
- |
|
|
- |
|
8,029,059 |
|
|
8,029 |
|
|
33,542,894 |
|
|
|
(313,746 |
) |
|
|
- |
|
|
(32,448,641 |
) |
|
|
788,536 |
|
| |
| Issuance of
common stock |
|
- |
|
|
- |
|
561,701 |
|
|
562 |
|
|
3,181,712 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
3,182,274 |
|
| |
| Stock based
compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
(155,893 |
) |
|
|
155,893 |
|
|
|
- |
|
|
- |
|
|
|
- |
|
| Amortization
of stock based |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
79,371 |
|
|
|
- |
|
|
- |
|
|
|
79,371 |
|
| Common stock
issued in exchange for |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| services |
|
- |
|
|
- |
|
144,300 |
|
|
144 |
|
|
823,743 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
823,887 |
|
| |
| Net
loss |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
(6,037,528 |
) |
|
|
(6,037,528 |
) |
| Balance,
December 31, 2003 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| (unaudited) |
|
- |
|
|
- |
|
8,735,060 |
|
|
8,735 |
|
|
37,392,456 |
|
|
|
(78,482 |
) |
|
|
- |
|
|
(38,486,169 |
) |
|
|
(1,163,460 |
) |
| |
| Issuance of
common stock |
|
- |
|
|
- |
|
808,570 |
|
|
809 |
|
|
4,580,104 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
4,580,913 |
|
| |
| Stock based
compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
637,858 |
|
|
|
(637,858 |
) |
|
|
- |
|
|
- |
|
|
|
- |
|
| Amortization
of stock based |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
148,812 |
|
|
|
- |
|
|
- |
|
|
|
148,812 |
|
| Common stock
issued in exchange for |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| services |
|
- |
|
|
- |
|
17,004 |
|
|
17 |
|
|
96,314 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
96,331 |
|
| Net
loss |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
(5,519,151 |
) |
|
|
(5,519,151 |
) |
F-27
| Balance,
December 31, 2004 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| (unaudited) |
|
- |
|
|
- |
|
9,560,634 |
|
|
9,561 |
|
|
42,706,732 |
|
|
|
(567,528 |
) |
|
|
- |
|
|
(44,005,320 |
) |
|
|
(1,856,555 |
) |
| Issuance of
common stock, net of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| issuance
costs of $32,507 |
|
- |
|
|
- |
|
1,994,556 |
|
|
1,994 |
|
|
11,265,560 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
11,267,554 |
|
| Issuance of
common stock in lieu of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| cash
compensation |
|
- |
|
|
- |
|
1,210 |
|
|
1 |
|
|
6,852 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
6,853 |
|
| |
| Stock based
compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
1,566,147 |
|
|
|
(1,566,147 |
) |
|
|
- |
|
|
- |
|
|
|
- |
|
| Amortization
of stock based |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
1,952,350 |
|
|
|
- |
|
|
- |
|
|
|
1,952,350 |
|
| Issuance of
common stock in |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| exchange for
release of accrued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| liabilities |
|
- |
|
|
|
|
-
95,807 |
|
|
96 |
|
|
542,691 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
542,787 |
|
| |
| Net
loss |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
(7,326,557 |
) |
|
|
(7,326,557 |
) |
| Balance,
December 31, 2005 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| (unaudited) |
|
- |
|
$ |
- |
|
11,652,207 |
|
$ |
11,652 |
|
$ |
56,087,982 |
|
|
$ |
(181,325 |
) |
|
$ |
- |
|
$ |
(51,331,877 |
) |
|
$ |
4,586,432 |
|
F-28
BIOHEART, INC.
(a
development stage company)
STATEMENT OF
STOCKHOLDERS'
DEFICIT
FROM AUGUST 12, 1999
(DATE OF INCEPTION)
THROUGH DECEMBER 31, 2013
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deficit |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Additional |
|
|
|
|
|
|
|
|
During |
|
|
|
|
|
|
Preferred
stock |
|
Common
stock |
|
Paid
in |
|
Deferred |
|
Subscription |
|
Development |
|
|
|
|
|
|
Shares |
|
Amount |
|
Shares |
|
Amount |
|
Capital |
|
Compensation |
|
Receivable |
|
Stage |
|
Total |
| Balance,
December 31, 2005 (unaudited) |
|
- |
|
$ |
- |
|
11,652,207 |
|
$ |
11,652 |
|
$ |
56,087,982 |
|
|
$ |
(181,325 |
) |
|
$ |
- |
|
$ |
(51,331,877 |
) |
|
$ |
4,586,432 |
|
| Reclassification of deferred compensation
due |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| to adoption
of SFAS No. 123 ( R ) |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
(181,325 |
) |
|
|
181,325 |
|
|
|
- |
|
|
- |
|
|
|
- |
|
| Issuance of
common stock, net of issuance |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| costs of
$100,038 |
|
- |
|
|
- |
|
1,069,699 |
|
|
1,069 |
|
|
8,123,623 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
8,124,692 |
|
| Equity
instruments issued in connection with |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| settlement
agreement |
|
- |
|
|
- |
|
47,657 |
|
|
48 |
|
|
3,294,381 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
3,294,429 |
|
| Common stock
issued in exchange for |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| services |
|
- |
|
|
- |
|
2,903 |
|
|
3 |
|
|
16,440 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
16,443 |
|
| Common stock
issued in exchange for |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| distribution
rights and intellectual property |
|
- |
|
|
- |
|
13,006 |
|
|
13 |
|
|
99,984 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
99,997 |
|
| Warrants
issued in exchange for licenses and |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| intellectual
property |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
144,867 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
144,867 |
|
| |
| Stock based
compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
1,224,430 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
1,224,430 |
|
| |
| Net
loss |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
(13,180,646 |
) |
|
|
(13,180,646 |
) |
| |
| Balance,
December 31, 2006 (unaudited) |
|
- |
|
|
- |
|
12,785,472 |
|
|
12,785 |
|
|
68,810,382 |
|
|
|
- |
|
|
|
- |
|
|
(64,512,523 |
) |
|
|
4,310,644 |
|
| Issuance of
common stock, net of issuance |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| costs of
$150,000 |
|
- |
|
|
- |
|
529,432 |
|
|
530 |
|
|
3,920,186 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
3,920,716 |
|
| |
| Exercise of
stock options |
|
- |
|
|
- |
|
31,955 |
|
|
32 |
|
|
181,008 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
181,040 |
|
| Warrants
issued in connection with notes |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| payable |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
3,162,488 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
3,162,488 |
|
| |
| Warrants
issued in exchange for services |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
30,559 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
30,559 |
|
| Warrants
issued in exchange for licenses and |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| intellectual
property |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
48,289 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
48,289 |
|
| Shares
issued in connection with reverse stock |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| split |
|
- |
|
|
- |
|
279 |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
- |
|
| |
| Stock based
compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
931,233 |
|
|
|
- |
|
|
|
- |
|
|
- |
|
|
|
931,233 |
|
| |
| Net
loss |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
(18,067,084 |
) |
|
|
(18,067,084 |
) |
| |
| Balance,
December 31, 2007 (unaudited) |
|
- |
|
$ |
- |
|
13,347,138 |
|
$ |
13,347 |
|
$ |
77,084,145 |
|
|
$ |
- |
|
|
$ |
- |
|
$ |
(82,579,607 |
) |
|
$ |
(5,482,115 |
) |
F-29
BIOHEART, INC.
(a
development stage company)
STATEMENT OF
STOCKHOLDERS'
DEFICIT
FROM AUGUST 12, 1999
(DATE OF INCEPTION)
THROUGH DECEMBER 31, 2013
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deficit |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Additional |
|
|
|
|
|
|
|
|
During |
|
|
|
|
|
|
Preferred
stock |
|
Common
stock |
|
Paid
in |
|
Deferred |
|
Subscription |
|
Development |
|
|
|
|
|
|
Shares |
|
Amount |
|
Shares |
|
Amount |
|
Capital |
|
Compensation |
|
Receivable |
|
Stage |
|
Total |
| Balance,
December 31, 2007 (unaudited) |
|
- |
|
$ |
- |
|
$ |
13,347,138 |
|
$ |
13,347 |
|
$ |
77,084,145 |
|
$ |
- |
|
$ |
- |
|
|
$ |
(82,579,607 |
) |
|
$ |
(5,482,115 |
) |
| Initial
public offering of common stock, |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| net of
offering costs of $4,327,171 |
|
- |
|
|
- |
|
|
1,100,000 |
|
|
1,100 |
|
|
1,446,729 |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
1,447,829 |
|
| Issuance of
common stock, net of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| issuance
costs of $24,325 |
|
- |
|
|
- |
|
|
1,230,280 |
|
|
1,230 |
|
|
2,117,275 |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
2,118,505 |
|
| |
| Stock based
compensation |
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
1,320,995 |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
1,320,995 |
|
| |
| Warrants
issued in exchange for services |
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
251,850 |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
251,850 |
|
| Warrants
issued in exchange for notes |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| payable |
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
168,387 |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
168,387 |
|
| Warrants
issued in connection with |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| settlement
agreement |
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
87,200 |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
87,200 |
|
| |
| Exercise of
stock options |
|
- |
|
|
- |
|
|
61,778 |
|
|
62 |
|
|
79,014 |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
79,076 |
|
| |
| Net
loss |
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
|
(14,149,401 |
) |
|
|
(14,149,401 |
) |
| Balance,
December 31, 2008 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| (unaudited) |
|
- |
|
|
- |
|
|
15,739,196 |
|
|
15,739 |
|
|
82,555,595 |
|
|
- |
|
|
- |
|
|
|
(96,729,008 |
) |
|
|
(14,157,674 |
) |
| Issuance of
common stock, net of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| issuance
costs of $13,664 |
|
- |
|
|
- |
|
|
2,680,230 |
|
|
2,682 |
|
|
1,857,140 |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
1,859,822 |
|
| |
| Subscription
receivable |
|
- |
|
|
- |
|
|
85,090 |
|
|
85 |
|
|
59,451 |
|
|
- |
|
|
(59,536 |
) |
|
|
- |
|
|
|
- |
|
| |
| Stock based
compensation |
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
296,838 |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
296,838 |
|
| |
| Common stock
in exchange for services |
|
- |
|
|
- |
|
|
45,000 |
|
|
45 |
|
|
45,855 |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
45,900 |
|
| Common stock
issued in connection with |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| the
settlement of accounts payable |
|
- |
|
|
- |
|
|
519,460 |
|
|
519 |
|
|
456,275 |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
456,794 |
|
| Common stock
issued in connection with |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| issuance of
note payable |
|
- |
|
|
- |
|
|
320,000 |
|
|
320 |
|
|
297,681 |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
298,001 |
|
| Common stock
issued upon conversion |
|
|
|
|
- |
|
|
|
|
|
|
|
|
|
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
|
|
F-30
| of notes
payable |
|
- |
|
|
|
|
|
606,708 |
|
|
606 |
|
|
261,618 |
|
|
|
|
|
|
|
|
|
|
|
|
|
262,224 |
|
| Warrants
issued in connection with notes |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| payable |
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
1,913,487 |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
1,913,487 |
|
| |
| Exercise of
stock options |
|
- |
|
|
- |
|
|
40,000 |
|
|
40 |
|
|
31,960 |
|
|
- |
|
|
- |
|
|
|
- |
|
|
|
32,000 |
|
| |
| Net
loss |
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
|
(4,435,756 |
) |
|
|
(4,435,756 |
) |
| Balance,
December 31, 2009 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| (unaudited) |
|
- |
|
$ |
- |
|
|
20,035,684 |
|
$ |
20,036 |
|
$ |
87,775,900 |
|
$ |
- |
|
$ |
(59,536 |
) |
|
$ |
(101,164,764 |
) |
|
$ |
(13,428,364 |
) |
F-31
BIOHEART, INC.
(a
development stage company)
STATEMENT OF
STOCKHOLDERS'
DEFICIT
FROM AUGUST 12, 1999
(DATE OF INCEPTION)
THROUGH DECEMBER 31, 2013
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deficit |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated |
|
|
|
|
|
|
|
|
|
|
|
|
Additional |
|
|
|
|
|
|
|
During |
|
|
|
|
|
|
Preferred stock |
|
|
Common stock |
|
Paid in |
|
Deferred |
|
Subscription |
|
Development |
|
|
|
|
|
|
Shares |
|
Amount |
|
Shares |
|
Amount |
|
Capital |
|
Compensation |
|
Receivable |
|
Stage |
|
Total |
| Balance,
December 31, 2009 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| (unaudited) |
|
- |
|
$ |
- |
|
$ |
20,035,684 |
|
$ |
20,036 |
|
$ |
87,775,900 |
|
- |
|
$ |
(59,536 |
) |
|
|
(101,164,764 |
) |
|
$ |
(13,428,364 |
) |
| Issuance of
common stock, net of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| issuance
costs of $2,950 |
|
- |
|
|
- |
|
|
11,334,705 |
|
|
11,335 |
|
|
2,328,929 |
|
|
|
|
59,536 |
|
|
|
- |
|
|
|
2,399,800 |
|
| |
| Subscription
receivable |
|
- |
|
|
- |
|
|
20,000 |
|
|
20 |
|
|
3,780 |
|
- |
|
|
(3,800 |
) |
|
|
- |
|
|
|
- |
|
| |
| Stock based
compensation |
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
248,457 |
|
- |
|
|
- |
|
|
|
- |
|
|
|
248,457 |
|
| Common stock
in exchange for |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| services |
|
- |
|
|
- |
|
|
529,520 |
|
|
529 |
|
|
359,503 |
|
- |
|
|
- |
|
|
|
- |
|
|
|
360,032 |
|
| Common stock
issued in connection |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| with
settlement of accounts payable |
|
- |
|
|
- |
|
|
831,526 |
|
|
831 |
|
|
411,829 |
|
- |
|
|
- |
|
|
|
- |
|
|
|
412,660 |
|
| Common stock
issued in connection |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| with bank
guarantor liabilities |
|
- |
|
|
- |
|
|
4,794,430 |
|
|
4,794 |
|
|
2,960,576 |
|
- |
|
|
- |
|
|
|
- |
|
|
|
2,965,370 |
|
| Warrants
issued in connection with |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| notes
payable |
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
185,307 |
|
- |
|
|
- |
|
|
|
- |
|
|
|
185,307 |
|
| |
| Net
loss |
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
- |
|
- |
|
|
- |
|
|
|
(5,159,456 |
) |
|
|
(5,159,456 |
) |
| Balance, December 31,
2010 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| (unaudited) |
|
- |
|
$ |
- |
|
|
37,545,865 |
|
$ |
37,545 |
|
$ |
94,274,281 |
|
- |
|
$ |
(3,800 |
) |
|
$ |
(106,324,220 |
) |
|
$ |
(12,016,194 |
) |
F-32
BIOHEART, INC.
(a
development stage company)
STATEMENT OF
STOCKHOLDERS'
DEFICIT
FROM AUGUST 12, 1999
(DATE OF INCEPTION)
THROUGH DECEMBER 31, 2013
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deficit |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Additional |
|
|
|
|
|
|
|
|
|
During |
|
|
|
|
|
|
Preferred
stock |
|
Common
stock |
|
Paid
in |
|
Deferred |
|
Subscription |
|
Development |
|
|
|
|
|
|
Shares |
|
Amount |
|
Shares |
|
Amount |
|
Capital |
|
Compensation |
|
Receivable |
|
Stage |
|
Total |
| Balance,
December 31, 2010 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| (unaudited) |
|
- |
|
$ |
- |
|
37,545,865 |
|
|
$ |
37,545 |
|
|
$ |
94,274,281 |
|
|
$ |
- |
|
|
$ |
(3,800 |
) |
|
$ |
(106,324,220 |
) |
|
$ |
(12,016,194 |
) |
| Cancellation
of previously issued |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| shares |
|
- |
|
|
- |
|
(3,450 |
) |
|
|
(3 |
) |
|
|
3 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
| Proceeds
from common stock |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| subscription |
|
- |
|
|
- |
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
3,800 |
|
|
|
- |
|
|
|
3,800 |
|
| Common stock
issued in |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| exchanged of
options exercised |
|
- |
|
|
- |
|
1,982,995 |
|
|
|
1,983 |
|
|
|
(351 |
) |
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
1,632 |
|
| Common stock
in exchange for |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| services |
|
- |
|
|
- |
|
1,000,000 |
|
|
|
1,000 |
|
|
|
114,035 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
115,035 |
|
| Common stock
issued upon |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| conversion
of notes payable |
|
- |
|
|
- |
|
27,120,856 |
|
|
|
27,121 |
|
|
|
1,514,988 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
1,542,109 |
|
| Common stock
in connection |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| with
settlement agreement |
|
- |
|
|
- |
|
4,521,700 |
|
|
|
4,522 |
|
|
|
312,997 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
317,519 |
|
| Issuance of
common stock, net of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| issuance
costs of $40,000 |
|
- |
|
|
- |
|
22,184,540 |
|
|
|
22,184 |
|
|
|
1,402,616 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
1,424,800 |
|
| Common stock
issued in |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| settlement
of related party |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| advance |
|
- |
|
|
- |
|
1,272,730 |
|
|
|
1,273 |
|
|
|
138,727 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
140,000 |
|
| |
| Stock based
compensation |
|
- |
|
|
- |
|
- |
|
|
|
- |
|
|
|
409,314 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
409,314 |
|
| Beneficial
conversion feature |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| connection
with issuance of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| convertible
note |
|
- |
|
|
- |
|
- |
|
|
|
- |
|
|
|
748,545 |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
748,545 |
|
| |
| Net
loss |
|
- |
|
|
- |
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
- |
|
|
|
(4,697,037 |
) |
|
|
(4,697,037 |
) |
| |
| Balance, December 31,
2011 |
|
- |
|
$ |
- |
|
95,625,236 |
|
|
$ |
95,625 |
|
|
$ |
98,915,155 |
|
|
$ |
- |
$ |
|
|
- |
|
|
$ |
(111,021,257 |
) |
|
$ |
(12,010,477 |
) |
F-33
BIOHEART, INC.
(a
development stage company)
STATEMENT OF
STOCKHOLDERS'
DEFICIT
FROM
AUGUST 12, 1999 (DATE OF INCEPTION)
THROUGH DECEMBER 31, 2013
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deficit |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated |
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
Additional |
|
|
|
|
|
|
|
During |
|
|
|
|
|
|
Preferred
stock |
|
Common
stock |
|
Paid
in |
|
Deferred |
|
Subscription |
|
Development |
|
|
|
|
|
|
Shares |
|
Amount |
|
Shares |
|
Amount |
|
Capital |
|
Compensation |
|
Receivable |
|
Stage |
|
Total |
| Balance,
December 31, 2011 |
|
- |
|
$ |
- |
|
95,625,236 |
|
$ |
95,625 |
|
$ |
98,915,155 |
|
|
$ |
- |
|
$ |
- |
|
$ |
(111,021,257 |
) |
|
$ |
(12,010,477 |
) |
| |
| Issuance of
common stock |
|
- |
|
|
- |
|
24,085,718 |
|
|
24,085 |
|
|
485,715 |
|
|
|
- |
|
|
- |
|
|
- |
|
|
|
509,800 |
|
| |
| Common stock
issued for services |
|
- |
|
|
- |
|
952,851 |
|
|
953 |
|
|
33,647 |
|
|
|
- |
|
|
- |
|
|
- |
|
|
|
34,600 |
|
| |
| Common stock
issued under put agreement |
|
- |
|
|
- |
|
8,947,859 |
|
|
8,948 |
|
|
141,052 |
|
|
|
- |
|
|
- |
|
|
- |
|
|
|
150,000 |
|
| Common stock
issued upon conversion of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| notes
payable |
|
- |
|
|
- |
|
51,751,138 |
|
|
51,752 |
|
|
668,462 |
|
|
|
|
|
|
|
|
|
|
|
|
|
720,214 |
|
| |
| Common stock
issued for accrued liabilities |
|
|
|
|
|
|
700,000 |
|
|
700 |
|
|
13,300 |
|
|
|
- |
|
|
- |
|
|
- |
|
|
|
14,000 |
|
| |
| Stock based
compensation |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
76,674 |
|
|
|
- |
|
|
- |
|
|
- |
|
|
|
76,674 |
|
| Fair value
of warrants issued in connection |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| with
forbearance agreement |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
119,023 |
|
|
|
- |
|
|
- |
|
|
- |
|
|
|
119,023 |
|
| Beneficial
conversion feature connection with |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| issuance of
convertible note |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
234,729 |
|
|
|
- |
|
|
- |
|
|
- |
|
|
|
234,729 |
|
| Reclassify
committed common shares in |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| excess of
authorized amount to liability |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
(427,663 |
) |
|
|
- |
|
|
- |
|
|
- |
|
|
|
(427,663 |
) |
| |
| Net
loss |
|
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
|
- |
|
|
- |
|
|
(4,016,467 |
) |
|
|
(4,016,467 |
) |
| Balance, December 31,
2012 |
|
- |
|
$ |
- |
|
182,062,802 |
|
$ |
182,063 |
|
$ |
100,260,094 |
|
|
$ |
- |
|
$ |
- |
|
$ |
(115,037,724 |
) |
|
$ |
(14,595,567 |
) |
F-34
BIOHEART, INC.
(a
development stage company)
STATEMENT OF
STOCKHOLDERS'
DEFICIT
FROM
AUGUST 12, 1999 (DATE OF INCEPTION)
THROUGH DECEMBER 31, 2013
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deficit |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Additional |
|
|
|
|
|
|
|
During |
|
|
|
|
|
Preferred stock |
|
Common stock |
|
Paid
in |
|
Deferred |
|
Subscription |
|
Development |
|
|
|
|
|
Shares |
|
Amount |
|
Shares |
|
Amount |
|
Capital |
|
Compensation |
|
Receivable |
|
Stage |
|
|
Total |
| Balance, December 31, 2012 |
- |
|
$ |
- |
|
182,062,802 |
|
|
182,063 |
|
|
100,260,094 |
|
|
- |
|
|
- |
|
|
(115,037,724 |
) |
|
|
(14,595,567 |
) |
| Reclassify the fair value of excess |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| committed shares liability to equity
upon |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| common share authorization increase |
- |
|
|
- |
|
- |
|
|
- |
|
|
474,954 |
|
|
- |
|
|
- |
|
|
- |
|
|
|
474,954 |
|
| |
| Issuance of common stock |
- |
|
|
- |
|
50,029,227 |
|
|
50,029 |
|
|
814,971 |
|
|
- |
|
|
- |
|
|
- |
|
|
|
865,000 |
|
| |
| Common stock issued under put
agreement |
- |
|
|
- |
|
31,052,141 |
|
|
31,053 |
|
|
315,861 |
|
|
- |
|
|
- |
|
|
- |
|
|
|
346,914 |
|
| Common stock issued in connection
with |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| issuance of convertible debt |
- |
|
|
- |
|
2,500,000 |
|
|
2,500 |
|
|
33,750 |
|
|
- |
|
|
- |
|
|
- |
|
|
|
36,250 |
|
| Common stock issued in connection
with |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| settlement of debt |
- |
|
|
- |
|
57,967,906 |
|
|
57,968 |
|
|
735,167 |
|
|
- |
|
|
- |
|
|
- |
|
|
|
793,135 |
|
| Common stock issued in settlement of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| interest and penalty in connection
with |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| convertible debt |
- |
|
|
- |
|
9,408,718 |
|
|
9,409 |
|
|
141,406 |
|
|
- |
|
|
- |
|
|
- |
|
|
|
150,815 |
|
| |
| Common stock issued for services |
- |
|
|
- |
|
6,220,263 |
|
|
6,220 |
|
|
78,931 |
|
|
- |
|
|
- |
|
|
- |
|
|
|
85,151 |
|
| Common stock issued in settlement of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| related party notes payable |
- |
|
|
- |
|
34,890,348 |
|
|
34,890 |
|
|
362,860 |
|
|
- |
|
|
- |
|
|
- |
|
|
|
397,750 |
|
| Common stock issued in settlement of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| accounts payable |
- |
|
|
- |
|
5,656,340 |
|
|
5,656 |
|
|
151,560 |
|
|
- |
|
|
- |
|
|
- |
|
|
|
157,216 |
|
| |
| Preferred stock issued in settlement of
debt |
5,000,000 |
|
|
5,000 |
|
- |
|
|
- |
|
|
65,000 |
|
|
- |
|
|
- |
|
|
- |
|
|
|
70,000 |
|
| Preferred stock issued in settlement
of |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| forbearance agreement |
15,000,000 |
|
15,000 |
|
- |
|
|
- |
|
|
259,050 |
|
|
|
|
|
|
|
|
|
|
|
|
274,050 |
|
| |
| Proceeds from common stock
subscription |
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
215,000 |
|
|
- |
|
|
|
215,000 |
|
| |
| Stock based compensation |
- |
|
|
- |
|
- |
|
|
- |
|
|
125,515 |
|
|
- |
|
|
- |
|
|
- |
|
|
|
125,515 |
|
| |
| Net
loss |
- |
|
|
- |
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
- |
|
|
(3,143,259 |
) |
|
|
(3,143,259 |
) |
| |
| Balance, December 31, 2013 |
20,000,000 |
|
$ |
20,000 |
|
379,787,745 |
|
$ |
379,788 |
|
$ |
103,819,119 |
|
$ |
- |
|
$ |
215,000 |
|
$ |
(118,180,983 |
) |
|
$ |
(13,747,076 |
) |
See the accompanying notes to these financial statements
F-35
BIOHEART, INC.
(a
development stage company)
STATEMENTS OF CASH FLOWS
|
|
|
|
|
|
|
|
|
From
August 12, |
|
|
|
|
|
|
|
|
|
1999
(date of |
|
Year
ended December 31, |
|
Inception) to |
|
2013 |
|
2012 |
|
December 31,
2013 |
|
|
|
|
|
|
|
|
|
(unaudited) |
| CASH
FLOWS FROM OPERATING ACTIVITIES: |
|
|
|
|
|
|
|
|
|
|
|
| Net
loss |
$ |
(3,143,259 |
) |
|
$ |
(4,016,467 |
) |
|
$ |
(118,180,983 |
) |
| Adjustments to reconcile net loss to net cash used in
operating |
|
|
|
|
|
|
|
|
|
|
|
| activities: |
|
|
|
|
|
|
|
|
|
|
|
| Depreciation and amortization |
|
2,190 |
|
|
|
14,589 |
|
|
|
900,104 |
|
| Bad
debt expense |
|
- |
|
|
|
- |
|
|
|
166,266 |
|
| Discount on convertible debt |
|
368,682 |
|
|
|
468,581 |
|
|
|
1,980,250 |
|
| Loss
(gain) on change in fair value of derivative liability |
|
(29,179 |
) |
|
|
(119,795 |
) |
|
|
(123,948 |
) |
| Gain
on settlement of debt |
|
(1,023,439 |
) |
|
|
- |
|
|
|
(1,023,439 |
) |
| Non
cash payment of interest |
|
228,657 |
|
|
|
93,209 |
|
|
|
499,767 |
|
| Amortization of warrants issued in exchange for licenses
and |
|
|
|
|
|
|
|
|
|
|
|
| intellectual property |
|
- |
|
|
|
- |
|
|
|
5,413,156 |
|
| Amortization of warrants issued in connection with notes
payable |
|
- |
|
|
|
95,291 |
|
|
|
5,437,604 |
|
| Amortization of loan costs |
|
- |
|
|
|
927 |
|
|
|
1,228,717 |
|
| Related party notes payable issued for services
rendered |
|
500,000 |
|
|
|
- |
|
|
|
500,000 |
|
| Warrants issued in exchange for services |
|
- |
|
|
|
- |
|
|
|
285,659 |
|
| Warrants issued in exchange for forbearance
agreement |
|
- |
|
|
|
430,213 |
|
|
|
430,213 |
|
| Equity instruments issued in connection with R&D
agreement |
|
- |
|
|
|
- |
|
|
|
360,032 |
|
| Equity instruments issued in connection with settlement
agreement |
|
- |
|
|
|
- |
|
|
|
3,381,629 |
|
| Common stock issued in connection with accounts
payable |
|
2,500 |
|
|
|
- |
|
|
|
759,316 |
|
| Common stock issued in exchange for services |
|
85,151 |
|
|
|
34,600 |
|
|
|
1,567,673 |
|
| Common stock issued in connection with amounts due to
guarantors |
|
|
|
|
|
|
|
|
|
|
|
| of
Bank of America loan |
|
- |
|
|
|
- |
|
|
|
69,159 |
|
| Common stock issued in exchange for distribution rights
and |
|
|
|
|
|
|
|
|
|
|
|
| intellectual property |
|
- |
|
|
|
- |
|
|
|
99,997 |
|
| Preferred stock issued in settlement of forbearance
agreement |
|
274,050 |
|
|
|
- |
|
|
|
274,050 |
|
| Warrants issued in connection with accounts payable |
|
|
|
|
|
- |
|
|
|
7,758 |
|
| Stock based compensation |
|
125,515 |
|
|
|
76,674 |
|
|
|
10,076,517 |
|
| (Increase) decrease in: |
|
|
|
|
|
|
|
|
|
|
|
| Receivables |
|
(18,571 |
) |
|
|
2,153 |
|
|
|
(21,178 |
) |
| Inventory |
|
62,953 |
|
|
|
749 |
|
|
|
- |
|
| Prepaid and other current assets |
|
85,251 |
|
|
|
8,295 |
|
|
|
33,748 |
|
| Other assets |
|
- |
|
|
|
- |
|
|
|
(28,854 |
) |
| Increase (decrease) in: |
|
|
|
|
|
|
|
|
|
|
|
| Accounts payable |
|
(59,059 |
) |
|
|
324,264 |
|
|
|
3,200,620 |
|
| Accrued expenses |
|
625,232 |
|
|
|
1,491,441 |
|
|
|
7,363,476 |
|
| Deferred revenue |
|
- |
|
|
|
- |
|
|
|
465,287 |
|
| Net
cash used in operating activities |
|
(1,913,326 |
) |
|
|
(1,095,276 |
) |
|
|
(74,877,404 |
) |
F-36
BIOHEART, INC.
(a
development stage company)
STATEMENTS OF CASH FLOWS
|
|
|
|
|
|
|
|
|
|
From
August 12, |
|
|
|
|
|
|
|
|
|
|
1999
(date of |
|
|
Year
ended December 31, |
|
Inception) to |
|
|
|
|
|
|
|
|
|
|
December 31, |
|
|
2013 |
|
2012 |
|
2013 |
|
|
|
|
|
|
|
|
|
|
(unaudited) |
| CASH
FLOWS FROM INVESTING ACTIVITIES: |
|
|
|
|
|
|
|
|
|
|
|
|
| Acquisitions of property and equipment |
|
$ |
(9,425 |
) |
|
$ |
(933 |
) |
|
$ |
(909,158 |
) |
| Net
cash used by investing activities |
|
|
(9,425 |
) |
|
|
(933 |
) |
|
|
(909,158 |
) |
| CASH
FLOWS FROM FINANCING ACTIVITIES: |
|
|
|
|
|
|
|
|
|
|
|
|
| Bank
overdraft |
|
|
(89 |
) |
|
|
89 |
|
|
|
- |
|
| Proceeds from issuance of common stock, net |
|
|
1,426,914 |
|
|
|
584,800 |
|
|
|
65,308,689 |
|
| Proceeds from (payments for) initial public offering of
common stock, net |
|
|
- |
|
|
|
- |
|
|
|
1,447,829 |
|
| Proceeds from subordinated related party note |
|
|
- |
|
|
|
- |
|
|
|
3,000,000 |
|
| Payment of note payable |
|
|
- |
|
|
|
- |
|
|
|
(3,000,000 |
) |
| Proceeds from notes payable, related party |
|
|
- |
|
|
|
- |
|
|
|
505,000 |
|
| Proceeds from related party advances |
|
|
215,500 |
|
|
|
411,492 |
|
|
|
1,082,992 |
|
| Proceeds from exercise of stock options |
|
|
- |
|
|
|
- |
|
|
|
293,749 |
|
| Proceeds from notes payable |
|
|
415,500 |
|
|
|
63,000 |
|
|
|
12,036,250 |
|
| Repayments of notes payable |
|
|
(88,847 |
) |
|
|
- |
|
|
|
(3,622,452 |
) |
| Payment of loan costs |
|
|
- |
|
|
|
- |
|
|
|
(1,219,268 |
) |
| Net
cash provided in financing activities |
|
|
1,968,978 |
|
|
|
1,059,381 |
|
|
|
75,832,789 |
|
| Net
(decrease) increase in cash and cash equivalents |
|
|
46,227 |
|
|
|
(36,828 |
) |
|
|
46,227 |
|
| Cash
and cash equivalents, beginning of period |
|
|
- |
|
|
|
36,828 |
|
|
|
- |
|
| Cash
and cash equivalents, end of period |
|
$ |
46,227 |
|
|
|
- |
|
|
$ |
46,227 |
|
| SUPPLEMENTAL DISCLOSURES OF CASH FLOW INFORMATION |
|
|
|
|
|
|
|
|
|
|
|
|
| Interest paid |
|
$ |
521,742 |
|
|
$ |
403,618 |
|
|
$ |
2,705,824 |
|
| Income taxes paid |
|
|
- |
|
|
|
- |
|
|
|
- |
|
| Non
cash financing activities: |
|
|
|
|
|
|
|
|
|
|
|
|
| Common stock issued in settlement of notes payable |
|
$ |
793,135 |
|
|
$ |
720,214 |
|
|
$ |
5,046,094 |
|
| Common stock issued in settlement of accounts
payable |
|
$ |
157,216 |
|
|
$ |
14,000 |
|
|
$ |
171,216 |
|
| Common stock issued in settlement of related party notes
payable |
|
$ |
397,750 |
|
|
|
- |
|
|
$ |
397,750 |
|
| Common stock issued in settlement of interest and penalties
in connection with convertible debt |
|
$ |
150,815 |
|
|
|
- |
|
|
$ |
150,815 |
|
| Preferred stock issued in settlement of notes
payable |
|
$ |
70,000 |
|
|
|
- |
|
|
$ |
70,000 |
|
| Reclassification of fair value of excess committed shares
liability to equity upon common share |
|
|
|
|
|
|
|
|
|
|
|
|
| authorization increase |
|
$ |
474,954 |
|
|
|
- |
|
|
$ |
474,945 |
|
| Reclassification from notes payable to related party
advances |
|
|
- |
|
|
$ |
544,267 |
|
|
$ |
544,267 |
|
See the accompanying notes
to these consolidated financial statements
F-37
NOTE 1 — SIGNIFICANT
ACCOUNTING POLICIES
A summary of the
significant a ccounting policies applied in the presentation of the accompanying
financial statements follows:
Basis and business
presentation
Bioheart, Inc. (the “Company”) was incorporated
under the laws of the State of Florida in August 1999. The Company is in the
development stage, as defined by Accounting Standards Codification subtopic
915-10, Development Stage Entities (“ASC 915-10”) and is the cardiovascular
sector of the cell technology industry delivering cell therapies and biologics
that help address congestive heart failure, lower limb ischemia, chronic heart
ischemia, acute myocardial infarctions and other issues. To date, the Company
has not generated significant sales revenues, has incurred expenses and has
sustained losses. Consequently, its operations are subject to all the risks
inherent in the establishment of a new business enterprise. For the period from
inception through December 31, 2013, the Company has accumulated a deficit
through its development stage of $118,180,983.
Estimates
The preparation of the
financial statements in conformity with generally accepted accounting principles
requires management to make estimates and assumptions that affect certain
reported amounts and disclosures. The most significant estimates are those used
in determination of derivative liabilities and stock compensation. Accordingly,
actual results could differ from those estimates.
Revenue
Recognition
The Company recognizes
revenue in accordance with Accounting Standards Codification subtopic 605-10,
Revenue Recognition (“ASC 605-10”) which requires that four basic criteria must
be met before revenue can be recognized: (1) persuasive evidence of an
arrangement exists; (2) delivery has occurred; (3) the selling price is fixed
and determinable; and (4) collectability is reasonably assured. Determination of
criteria (3) and (4) are based on management’s judgments regarding the fixed
nature of the selling prices of the products delivered and the collectability of
those amounts. Provisions for discounts and rebates to customers, estimated
returns and allowances, and other adjustments are provided for in the same
period the related sales are recorded.
At the time of each
transaction, management assesses whether the fee associated with the transaction
is fixed or determinable and whether or not collection is reasonably assured.
The assessment of whether the fee is fixed or determinable is based upon the
payment terms of the transaction. If a significant portion of a fee is due after
our normal payment terms or upon implementation or client acceptance, the fee is
accounted for as not being fixed or determinable and revenue is recognized as
the fees become due or after implementation or client acceptance has occurred.
Collectability is assessed based on a number of factors, including past
transaction history with the client and the creditworthiness of the
client.
Unbilled revenue is revenue
that is recognized but is not currently billable to the customer pursuant to
contractual terms. In general, such amounts become billable in accordance with
predetermined payment schedules, but recognized as revenue as services are
performed. Amounts included in unbilled revenue are expected to be collected
within one year and are included within current assets.
Cash
The Company considers cash
to consist of cash on hand and temporary investments having an original maturity
of 90 days or less that are readily convertible into cash.
Accounts
Receivable
Trade receivables are
carried at their estimated collectible amounts. Trade credit is generally
extended on a short-term basis; thus trade receivables do not bear interest.
Trade accounts receivable are periodically evaluated for collectability based on
past credit history with customers and their current financial
condition.
Allowance for Doubtful
Accounts
Any changes to the
allowance for doubtful accounts on accounts receivable are charged to operations
in amounts sufficient to maintain the allowance for uncollectible accounts at a
level management believes is adequate to cover any probable losses. Management
determines the adequacy of the allowance based on historical write-off
percentages and the current status of accounts receivable. Accounts receivable
are charged off against the allowance when collectability is determined to be
permanently impaired. As of December 31, 2013 and 2012, allowance for doubtful
accounts was $-0-.
F-38
Long-Lived
Assets
The Company follows FASB
ASC 360-10-15-3, “Impairment or Disposal of Long-lived Assets,” which
established a “primary asset” approach to determine the cash flow estimation
period for a group of assets and liabilities that represents the unit of
accounting for a long-lived asset to be held and used. Long-lived assets to be
held and used are reviewed for impairment whenever events or changes in
circumstances indicate that the carrying amount of an asset may not be
recoverable. The carrying amount of a long-lived asset is not recoverable if it
exceeds the sum of the undiscounted cash flows expected to result from the use
and eventual disposition of the asset. Long-lived assets to be disposed of are
reported at the lower of carrying amount or fair value less cost to sell. The
Company determined that there was no impairment on its long-lived assets during
2013 and 2012.
Other
assets
During the year ended
December 2011, the Company’s management performed an evaluation of its other
assets (cost basis investment) for purposes of determining the implied fair
value of the asset at December 31, 2011, respectively. In 2011, the test
indicated that the recorded remaining book value of its investment exceeded its
fair value for the year ended December 31, 2011, as determined by discounted
future cash flows. As a result, upon completion of the assessment, management
recorded a non-cash impairment charge of $58,695 (unaudited), net of tax during
the year ended December 31, 2011 to reduce the carrying value of the investment
to $-0- (unaudited). Considerable management judgment is necessary to estimate
the fair value. Accordingly, actual results could vary significantly from
management’s estimates.
Property and
Equipment
Property and equipment are
stated at cost. When retired or otherwise disposed, the related carrying value
and accumulated depreciation are removed from the respective accounts and the
net difference less any amount realized from disposition, is reflected in
earnings. For financial statement purposes, property and equipment are recorded
at cost and depreciated using the straight-line method over their estimated
useful lives of 3 to 15 years.
Income
Taxes
The Company has adopted
Accounting Standards Codification subtopic 740-10, Income Taxes (“ASC 740-10”)
which requires the recognition of deferred tax liabilities and assets for the
expected future tax consequences of events that have been included in the
financial statement or tax returns. Under this method, deferred tax liabilities
and assets are determined based on the difference between financial statements
and tax basis of assets and liabilities using enacted tax rates in effect for
the year in which the differences are expected to reverse. Temporary differences
between taxable income reported for financial reporting purposes and income tax
purposes consist primarily of derivative liability and stock compensation
accounting versus tax differences.
Comprehensive
Income
The Company does not have
any items of comprehensive income in any of the periods presented.
Net Loss per Common
Share, basic and diluted
The Company has adopted
Accounting Standards Codification subtopic 260-10, Earnings per Share (“ASC
260-10”) specifying the computation, presentation and disclosure requirements of
earnings per share information. Basic loss per share has been calculated based
upon the weighted average number of common shares outstanding. Stock options and
warrants have been excluded as common stock equivalents in the diluted loss per
share because their effect is anti-dilutive on the computation. Fully diluted
shares outstanding were 344,241,761 and 195,204,110 for the years ended December
31, 2013 and 2012, respectively.
Stock based
compensation
The Company follows
Accounting Standards Codification subtopic 718-10, Compensation (“ASC 718-10”)
which requires that all share-based payments to both employees and non-employees
be recognized in the income statement based on their fair values. (See note
10)
As of December 31, 2013, there were outstanding
stock options to purchase 23,912,943 shares of common stock, 6,600,443 shares of
which were vested.
F-39
Concentrations of Credit
Risk
The Company’s financial
instruments that are exposed to a concentration of credit risk are cash and
accounts receivable. Effective December 31, 2010 and extending through December
31, 2012, all non-interest-bearing transaction accounts are fully insured by the
Federal Deposit Insurance Corporation (FDIC), regardless of the balance of the
account. Generally, the Company’s cash and cash equivalents in interest-bearing
accounts does not exceed FDIC insurance limits. The financial stability of these
institutions is periodically reviewed by senior management.
As of December 31, 2013,
three customers represented 36%, 20% and 20%, aggregate of 76% of the Company’s
accounts receivable. As of December 31, 2012, three customers represented 96% of
the Company’s accounts receivable
The Company’s revenues
earned from sale of products and services for the year ended December 31, 2013
included 20% of the Company’s total revenues from one customer. For the year
ended December 31, 2012, Company’s revenues earned from sale of products and
services included 80% of the Company’s total revenues from two
customers.
Reliance on Key
Personnel and Consultants
The Company has three
full-time employees and no part-time employees. The Company is heavily dependent
on the continued active participation of its two current executive officers, one
employee and key consultants. The loss of any of the senior management or key
consultants could significantly and negatively impact the business until
adequate replacements can be identified and put in place.
Research and
Development
The Company accounts for
research and development costs in accordance with Accounting Standards
Codification subtopic 730-10, Research and Development (“ASC 730-10”). Under ASC
730-10, all research and development costs must be charged to expense as
incurred. Accordingly, internal research and development costs are expensed as
incurred. Third-party research and development costs are expensed when the
contracted work has been performed or as milestone results have been achieved as
defined under the applicable agreement. Company-sponsored research and
development costs related to both present and future products are expensed in
the period incurred. The Company incurred research and development expenses of
$626,983, $401,941 and $65,320,035 (unaudited) for the years ended December 31,
2013 and 2012, and from August 12, 1999 (date of inception) to December 31,
2013, respectively.
Derivative Instrument
Liability
The Company accounts for
derivative instruments in accordance with ASC 815, which establishes accounting
and reporting standards for derivative instruments and hedging activities,
including certain derivative instruments embedded in other financial instruments
or contracts and requires recognition of all derivatives on the balance sheet at
fair value, regardless of hedging relationship designation. Accounting for
changes in fair value of the derivative instruments depends on whether the
derivatives qualify as hedge relationships and the types of relationships
designated are based on the exposures hedged. At December 31, 2013 and 2012, the
Company did not have any derivative instruments that were designated as
hedges.
Research
Grants
On November 9, 2010, we
received a grant in the amount of $244,500 (unaudited) under the qualifying
therapeutic discovery project under section 48D of the Internal Revenue code. We
have not received any research grant income in recent years.
Fair
Value
Accounting Standards
Codification subtopic 825-10, Financial Instruments (“ASC 825-10”) requires
disclosure of the fair value of certain financial instruments. The carrying
value of cash and cash equivalents, accounts payable and accrued liabilities,
and short-term borrowings, as reflected in the balance sheets, approximate fair
value because of the short-term maturity of these instruments. All other
significant financial assets, financial liabilities and equity instruments of
the Company are either recognized or disclosed in the financial statements
together with other information relevant for making a reasonable assessment of
future cash flows, interest rate risk and credit risk. Where practicable the
fair values of financial assets and financial liabilities have been determined
and disclosed; otherwise only available information pertinent to fair value has
been disclosed.
The company follows
Accounting Standards Codification subtopic 820-10, Fair Value Measurements and
Disclosures (“ASC 820-10”) and Accounting Standards Codification subtopic
825-10, Financial Instruments (“ASC 825-10”), which permits entities to choose
to measure many financial instruments and certain other items at fair value.
Neither of these statements had an impact on the Company’s financial position,
results of operations nor cash flows.
F-40
Reclassification
Certain reclassifications
have been made to prior periods’ data to conform to the current year’s
presentation. These reclassifications had no effect on reported income or
losses.
Recent Accounting
Pronouncements
There were various updates
recently issued, most of which represented technical corrections to the
accounting literature or application to specific industries and are not expected
to a have a material impact on the Company’s consolidated financial position,
results of operations or cash flows.
NOTE 2 — GOING CONCERN
MATTERS
The accompanying
consolidated financial statements have been prepared on a going concern basis,
which contemplates the realization of assets and the satisfaction of liabilities
in the normal course of business. As shown in the accompanying consolidated
financial statements during year ended December 31, 2013, the Company incurred
net losses of $3,143,259 and used $1,913,326 in cash for operating activities.
These factors among others may indicate that the Company will be unable to
continue as a going concern for a reasonable period of time.
The Company’s existence is
dependent upon management’s ability to develop profitable operations and to
obtain additional funding sources. There can be no assurance that the Company’s
financing efforts will result in profitable operations or the resolution of the
Company’s liquidity problems. The accompanying statements do not include any
adjustments that might result should the Company be unable to continue as a
going concern.
The Company’s ability to
obtain additional debt financing and/or alternative arrangements, with the
Guarantors or otherwise, may be limited by the amount of, terms and restrictions
of our then current debt. Accordingly, until such time, we will generally be
restricted from, among other things, incurring additional indebtedness or liens,
with limited exceptions. Additional debt financing, if available, may involve
restrictive covenants that limit or further limit our operating and financial
flexibility and prohibit us from making distributions to
shareholders.
NOTE 3 —
INVENTORY
Inventory consists of raw
materials. Costs of raw materials are determined using the FIFO method.
Inventory is stated at the lower of costs or market (estimated net realizable
value).
NOTE 4 — PROPERTY AND
EQUIPMENT
Property and equipment as
of December 31, 2013 and 2012 is summarized as follows:
|
2013 |
|
2012 |
| Laboratory and medical
equipment |
$ |
352,358 |
|
|
$ |
352,358 |
|
| Furniture, fixtures and
equipment |
|
125,634 |
|
|
|
130,916 |
|
| Computer
equipment |
|
39,525 |
|
|
|
54,414 |
|
| Leasehold improvements |
|
362,046 |
|
|
|
362,046 |
|
|
|
879,563 |
|
|
|
899,734 |
|
| Less accumulated depreciation and
amortization |
|
(870,508 |
) |
|
|
(897,914 |
) |
|
$ |
9,055 |
|
|
$ |
1,820 |
|
Property and equipment are
recorded on the basis of cost. For financial statement purposes, property, plant
and equipment are depreciated using the straight-line method over their
estimated useful lives.
Expenditures for repair and
maintenance which do not materially extend the useful lives of property and
equipment are charged to operations. When property or equipment is sold or
otherwise disposed of, the cost and related accumulated depreciation are removed
from the respective accounts with the resulting gain or loss reflected in
operations. Management periodically reviews the carrying value of its property
and equipment for impairment in accordance with the guidance for impairment of
long lived assets.
NOTE 5 — ACCRUED
EXPENSES
Accrued expenses consisted
of the following as of December 31, 2013 and 2012:
F-41
|
|
2013 |
|
2012 |
|
| |
License and royalty fees |
$ |
2,122,130 |
|
$ |
1,825,675 |
|
|
Amounts payable to the Guarantors of the
Company’s loan agreement with Bank of |
|
|
|
|
|
|
|
America and Seaside Bank, including fees and
interest |
|
1,373,775 |
|
|
1,284,705 |
|
|
Interest payable on notes payable |
|
714,180 |
|
|
1,100,174 |
|
|
Vendor accruals and other |
|
120,692 |
|
|
120,133 |
|
|
Employee commissions,
compensation, etc. |
|
149,558 |
|
|
400,801 |
|
|
|
$ |
4,480,335 |
|
$ |
4,731,488 |
|
During the year ended
December 31, 2013, 1,000,000 shares of common stock at a common market price of
$0.0195 were issued to one debt holder in exchange for $500,000 in principal and
$598,125 of accrued interest relating to a previously issued note. As a result
of the settlement, the Company recognized a gain of $1,078,625 during the year
ended December 31, 2013.
NOTE 6 – STANDBY EQUITY
DISTRIBUTION AGREEMENT
On November 2, 2011, the
Company and Greystone Capital Partners (“Greystone”) entered into a Standby
Equity Distribution Agreement (the “Agreement”). Pursuant to the Agreement,
Greystone has agreed to provide the Company with up to $1,000,000 of funding for
the 24-month period following the date a registration statement of the Company’s
common stock is declared effective by the SEC (the “Equity Line”).
During this 24-month
period, commencing on the date on which the SEC first declared the registration
statement effective, the Company may request a draw down under the Equity Line
by which the Company would sell shares of its common stock to Greystone, which
is obligated to purchase the shares under the Agreement.
For each share of the
Company common stock purchased under the Agreement, Greystone will pay eighty
percent (80%) of the average of the lowest daily volume weighted average price
for five consecutive trading days immediately preceding Advance Notice (the
"Valuation Period") commencing the date an Advance Notice (the "Advance Notice")
is delivered to Greystone in a manner provided by the Agreement. Subject to
certain limitations and floor price reductions, the Company may, at its sole
discretion, issue a Put Notice to Greystone and Greystone will then be
irrevocably bound to acquire such shares. The registration statement of the
Company's common stock pursuant to the Agreement was declared effective on
February 10, 2012 and a Post-Effective Amendment was declared effective on May
7, 2013. On December 1, 2012, the parties to the Equity Line agreed that the
Purchase Price be adjusted to seventy-five percent (75%) of the lowest daily
volume weighted average price of the Common Stock as quoted by Bloomberg, LP,
during the five (5) consecutive Trading Days (as such term is defined in the
Equity Line) immediately subsequent to the date of the relevant Advance Notice.
During the year ended
December 31, 2013, the Company issued an aggregate of 31,052,141 shares of its
common stock in exchange for $346,914 draw down on the equity line. During the
year ended December 31, 2012, the Company “put” 8,797,859 shares of common stock
for a total of $150,000.
NOTE 7 – NOTES PAYABLE
Notes payable were
comprised of the following as of December 31, 2013 and 2012:
|
|
2013 |
|
2012 |
|
|
Seaside Bank note payable. |
$ |
980,000 |
|
|
$ |
980,000 |
|
|
August 2008 Unsecured Promissory
Note |
|
500,000 |
|
|
|
1,000,000 |
|
|
Hunton & Williams notes payable |
|
384,972 |
|
|
|
384,972 |
|
|
Asher notes payable |
|
143,000 |
|
|
|
— |
|
| |
Fourth Man, LLC note payable |
|
35,000 |
|
|
|
— |
|
|
Total notes payable |
|
2,042,972 |
|
|
|
2,364,972 |
|
|
Less unamortized debt discount |
|
(112,131 |
) |
|
|
— |
|
|
Total notes payable net of unamortized debt
discount |
$ |
1,930,841 |
|
|
$ |
2,364,972 |
|
F-42
Seaside
Bank
On October 25, 2010, the
Company entered into a Loan Agreement with Seaside National Bank and Trust for a
$980,000 loan at 4.25% per annum interest that was used to refinance the
Company’s loan with Bank of America. The obligation is guaranteed by certain
shareholders of the Company. The Loan Agreement was scheduled to mature on
December 23, 2013, however the Company is renewing the loan with Seaside
National Bank and Trust during the first quarter of 2014 to extend the maturity
date. The loan is not in default as of December 31, 2013.
August 2008 Unsecured
Promissory Note
On August 20, 2008, the
Company borrowed $1.0 million from a third party pursuant to the terms of an
unsecured Promissory Note and Agreement. Outstanding principal and interest on
the loan, which accrues at the rate of 13.5% per annum, is payable in one
balloon payment upon the Company’s repayment of the BlueCrest Loan, which is
scheduled to mature in May 2010, however the Company is not obligated to make
payments until BlueCrest Loan is paid off. In the event the Company completes a
private placement of its common stock and/or securities exercisable for or
convertible into its common stock which generates at least $19.0 million of
gross proceeds, the Company may prepay, without penalty, all outstanding
principal and interest due under the loan using the same type of securities
issued in the subject private placement. Because repayment of the loan could
occur within 12 months from the date of the balance sheet, the Company has
classified this loan as short term.
Subject to certain
conditions, at the end of each calendar quarter during the time the loan is
outstanding, the Company may, but is not required to, pay all or any portion of
the interest accrued but unpaid as of such date with shares of its common
stock.
In April 2009, as
consideration for the authorization to amend certain documents related to the
Note, the Company issued to the Note holder a warrant to purchase 451,053 shares
of common stock at an exercise price of $0.5321 per share. The warrant, which
became exercisable immediately upon issuance, has a ten year term. This warrant
had a fair value of $195,694, which was accounted for as additional paid in
capital and reflected as a component of debt discount and has been fully
amortized as interest expense ratably over the term of the loan.
During the year ended
December 31, 2013, 1,000,000 shares of common stock were issued to the debt
holder valued at $19,500, in exchange for $500,000 in principal and $598,125 of
accrued interest relating to a previously issued note resulting in a gain of
$1,078,625. A gain of $1,078,625 was included in the net gain on settlement of
debt and trade payables on the statement of operations. As of December 31, 2013
the remaining principle of this note was $500,000.
Hunton & Williams
Notes
At December 31, 2013 and
2012, the Company has two unsecured outstanding notes payable with interest due
at maturity. The two notes, $61,150 (11.5% interest per annum) and $323,822 (8%
interest per annum) are payable in one balloon payment upon the date the
Noteholder provides written demand, however the Company is not obligated to make
payments until the Northstar (or successor) Loan is paid off.
Asher
Notes
2012
On April 2, 2012, the
Company entered into a Securities Purchase Agreement with Asher Enterprises,
Inc. (“Asher”), for the sale of an 8% convertible note in the principal amount
of $63,000 (the “Note”).
The Note bears interest at
the rate of 8% per annum. All interest and principal must be repaid on January
3, 2013. The Note is convertible into common stock, at Asher’s option, at a 42%
discount to the average of the three lowest closing bid prices of the common
stock during the 10 trading day period prior to conversion. The Company has
identified the embedded derivatives related to the Asher Note. These embedded
derivatives included certain conversion features and reset provision. The
accounting treatment of derivative financial instruments requires that the
Company record fair value of the derivatives as of the inception date of Asher
Note and to fair value as of each subsequent reporting date. At the inception of
the Asher Note, the Company determined the aggregate fair value of $76,682 of
the embedded derivatives.
The fair value of the
embedded derivatives was determined using the Binomial Option Pricing Model
based on the following assumptions: (1) dividend yield of 0%; (2) expected
volatility of 222.81%, (3) weighted average risk-free interest rate of 0.18%,
(4) expected life of 0.76 year, and (5) estimated fair value of the Company’s
common stock of $0.0373 per share. The initial fair value of the embedded debt
derivative of $76,682 was allocated as a debt discount up to the proceeds of the
note ($63,000) with the remainder ($13,682) charged to current period operations
as interest expense. For the year ended December 31, 2012, the Company amortized
or wrote off $63,000 of debt discount to current period operations as interest
expense. During the year ended December 31, 2012, the Company issued 6,297,578
shares of its common stock in settlement of the April 2, 2012 Unsecured
Convertible Note and related accrued interest.
F-43
2013
During the year ended
December 31, 2013, the Company entered into a Securities Purchase Agreements
with Asher Enterprises, Inc. (“Asher”), for the sale of 8% convertible notes in
aggregate principal amount of $255,500 (the “Asher Notes”).
The Asher Notes bear
interest at the rate of 8% per annum. As of the December 31, 2013 all interest
and principal must be repaid nine months from the issuance date, the last note
due August 7, 2014. The Notes are convertible into common stock, at Asher’s
option, at a 42% to 45% discount to the average of the three lowest closing bid
prices of the common stock during the 10 trading day period prior to conversion.
The Company has identified the embedded derivatives related to the Asher Notes.
These embedded derivatives included certain conversion features and reset
provision.
The accounting treatment of
derivative financial instruments requires that the Company record fair value of
the derivatives as of the inception date of Asher Notes and to fair value as of
each subsequent reporting date which at December 31, 2013 was $207,310. At the
inception of the Asher Notes, the Company determined the aggregate fair value of
$335,089 of the embedded derivatives.
The fair value of the
embedded derivatives was determined using the Binomial Option Pricing Model
based on the following assumptions: (1) dividend yield of 0%; (2) expected
volatility of 154.80% to 164.29%, (3) weighted average risk-free interest rate
of 0.09% to 0.17%, (4) expected lives of 0.67 to .77 years, and (5) estimated
fair value of the Company’s common stock from $0.01 to $0.0373 per share. The
initial fair value of the embedded debt derivative of $335,089 was allocated as
a debt discount up to the proceeds of the note ($255,500) with the remainder
($79,589) charged to current period operations as interest expense. For the
years ended December 31, 2013 and 2012, the Company amortized $162,706 and
$63,000 of debt discount to current period operations as interest expense,
respectively. As of December 31, 2013 the gross balance of the Asher Notes was
$143,000.
Fourth Man,
LLC
On October 11, 2013, the
Company entered into a Securities Purchase Agreement with Fourth Man, LLC.
(“Fourth Man”), for the sale of an 8% convertible note in the principal amount
of $35,000 (the “Note”).
The Note bears interest at
the rate of 8% per annum. All interest and principal must be repaid on April 10,
2014. The Note is convertible into common stock, at Fourth Man’s option, at a
47% discount to the average of the three lowest closing bid prices of the common
stock during the 10 trading day period prior to conversion. The Company has
identified the embedded derivatives related to the Fourth Man Note. These
embedded derivatives included certain conversion features and reset provision.
The accounting treatment of derivative financial instruments requires that the
Company record fair value of the derivatives as of the inception date of Fourth
Man Note and to fair value as of each subsequent reporting date. At the
inception of the Fourth Man Note, the Company determined the aggregate fair
value of $52,894 of the embedded derivatives.
The fair value of the
embedded derivatives was determined using the Binomial Option Pricing Model
based on the following assumptions: (1) dividend yield of 0%; (2) expected
volatility of 164.29%, (3) weighted average risk-free interest rate of 0.07%,
(4) expected life of 0.50 year, and (5) estimated fair value of the Company’s
common stock of $0.0121 per share. The initial fair value of the embedded debt
derivative of $52,894 was allocated as a debt discount up to the proceeds of the
note ($35,000) with the remainder ($17,894) charged to current period operations
as interest expense. For the year ended December 31, 2013, the Company amortized
$15,663 of debt discount to current period operations as interest expense.
NOTE 8 — RELATED PARTY
TRANSACTIONS
Advances
As of December 31, 2013 and
2012, the Company officers and directors have provided advances in the aggregate
of $416,198 and $313,448 respectively, for working capital purposes. The
advances are unsecured, due on demand and non-interest bearing.
F-44
On August 1, 2013, advances
in aggregate of $22,750 were converted into a demand promissory note with 5%
interest per annum. On September 30, 2013, the Company issued 1,995,614 shares
of common stock in settlement of the $22,750 promissory note.
On September 30, 2013, the
Company issued 8,771,929 shares of its common stock as payment of $100,000
towards cash advances.
Notes payable-related
party
Northstar
Biotechnology Group, LLC
On
February 29, 2012, a note issued to BlueCrest Master Fund Limited was assigned
to Northstar Biotechnology Group, LLC (“Northstar”), owned partly by certain
directors and existing shareholders of the Company, including Dr. William P.
Murphy Jr., Dr. Samuel Ahn and Charles Hart. At the date of the assignment, the
principal amount of the BlueCrest note was $544,267.
On March 30, 2012, the Company and
Northstar agreed to extend until May 1, 2012 the initial payment date for any
and all required monthly under the Note, such that the first of the four monthly
payments required under the Note will be due and payable on May, 2012 and all
subsequent payments will be due on a monthly basis thereafter commencing on June
1, 2012, and to waive any and all defaults and/or events of default under the
Note with respect to such payments. As of September 30, 2012, the Company was in
default, however, subsequent to September 30, 2012, the Company renegotiated the
terms of the Note, Northstar has agreed to suspend the requirement of principal
payments by the Company and allow payment of interest-only in common stock.
On September 21, 2012, the Company issued
5,000,000 common stock purchase warrants to Northstar that was treated as
Additional interest expense upon issuance.
On October 1, 2012, the Company and
Northstar entered into a limited waiver and forbearance agreement providing a
recapitalized new note balance comprised of all sums due Northstar with a
maturity date extended perpetually. The Company agreed to issue 5,000,000 shares
of Series A Convertible Preferred Stock and 10,000,000 of common stock in
exchange for $210,000 as payment towards outstanding debt, default interest,
penalties, professional fees outstanding and due Northstar. In addition, the
Company executed a security agreement granting Northstar a lien on all patents,
patent applications, trademarks, service marks, copyrights and intellectual
property rights of any nature, as well as the results of all clinical trials,
know-how for preparing Myoblasts, old and new clinical data, existing approved
trials, all right and title to Myoblasts, clinical trial protocols and other
property rights.
In addition, the Company granted
Northstar a perpetual license on products as described for resale, relicensing
and commercialization outside the United States. In connection with the granted
license, Northstar shall pay the Company a royalty of up to 8% on revenues
generated.
Effective October 1, 2012, the effective
interest rate was 12.85% per annum. The parties agreed, as of February 28, 2013,
to reduce the interest rate to 7% per annum (see Note 14 below).
In connection with the consideration
paid, Northstar waived, from the effective date through the earlier of
termination or expiration of the agreement, satisfaction of the obligations as
described in the forbearance agreement. In 2012, 5,000,000 shares of Series A
Convertible Preferred Stock were approved to be issued. In addition, the Company
is obligated to issue additional preferred stock equal in lieu of payment of
cash of accrued and unpaid interest on each six month anniversary of the
effective date (October 1, 2012). In lieu of the initial two payments in
preferred stock, the parties have determined to modify the voting rights of the
Series A Convertible Preferred Stock from 20 votes per share on matters to be
voted on by the common stock holders to 25 votes per share on matters to be
voted on by the common stock holders and all prior and subsequent payments of
interest will be in common stock; payments of common stock for April 1, 2013 and
October 1, 2013 were made the fourth quarter of 2013 based on the closing price
of the common stock on April 1, 2013 and October 1, 2013 respectively (see Note
14 below).
As described above, during the year ended
December 31, 2013, the Company issued the 5,000,000 shares of Series A
Convertible Preferred Stock and the 10,000,000 of common stock described above
in exchange for the $210,000 as payment towards outstanding principle of the
debt. In addition, the Company issued 15,000,000 shares of Series A Convertible
Preferred Stock as a penalty in settlement of the terms of the forbearance
agreement. The fair value of the Preferred Stock of $274,050 was included in
interest expense for the year ended December 31, 2013.
The Company is required to issue
additional shares of its common stock (as amended), in lieu of cash, each six
month anniversary of the effective date for any accrued and unpaid interest.
F-45
On
September 30, 2013, the Company issued 8,771,929 shares of its common stock as
payment of $100,000 towards cash advances.
On December 24, 2013, the Company issued
3,915,662 shares of its common stock as payment of accrued interest through June
30, 2013 of $85,447.
As of December 31, 2013, the principle of
this note was $362,000.
Officer and Director
Notes
At December 31, 2013 and 2012, the
Company has outstanding notes payable to officers and directors with interest at
8% per annum due at maturity. The three subordinated notes, $125,000, $100,000
and $140,000 were previously due on October 22, 2012, November 30, 2012 and June
4, 2011 respectively, and are unsecured. The Company is not obligated to make
payment until Northstar loan is paid off.
On October 9, 2012, the Company issued an
aggregate of $1,278,324 of promissory notes due October 9, 2013 to officers and
directors in settlement of outstanding advances and accrued compensation
(currently in default). The promissory notes bear interest of 5% per annum and
due at maturity. On September 30, 2013, the Company issued an aggregate of
15,350,876 shares of its common stock in settlement of $175,000 of related party
notes payable. During the year ended December 31, 2013, the Company paid $2,000
of the outstanding promissory notes and the principle balance as of December 31,
2013 is $1,101,324.
On August 1, 2013, the Company issued an
aggregate of $500,000 promissory notes due on demand to officers and employee in
settlement of accrued compensation. The promissory notes bear interest of 5% per
annum and due at various maturity dates. During the year ended December 31,
2013, the Company paid off $86,847 of the outstanding promissory notes. The
principle outstanding balance of these notes as of December 31, 2013 is
$413,153.
Subordinated debt,
related party
As of December 31, 2013 and 2012, the
Company officers and directors have provided notes in aggregate of $1,500,000.
The notes are at from 4.75% to 8% per annum and are due upon payoff of the
Northstar note payable described above.
Sales
Transactions
During the year ended December 31, 2013,
the Company entered into related party sales transactions with vendors that the
Company’s Chief Scientific Officer is a part owner.
NOTE 9 — DERIVATIVE
LIABILITIES
Excessive committed
shares
On December 31, 2012, in connection with
the previously issued stock options and warrants, the Company had the
possibility of exceeding their common shares authorized when considering the
number of possible shares that may be issuable to satisfy settlement provisions
of these agreements after consideration of all existing instruments that could
be settled in shares. The accounting treatment of derivative financial
instruments required that the Company reclassify the derivative from equity to a
liability at their fair values as of the date possible issuable shares exceeded
the authorized level and at fair value as of each subsequent balance sheet date.
Any change in fair value was recorded as non-operating, non-cash income or
expense at each reporting date. If the fair value of the derivatives was higher
at the subsequent balance sheet date, the Company recorded a non-operating,
non-cash charge. If the fair value of the derivatives was lower at the
subsequent balance sheet date, the Company recorded non-operating, non-cash
income.
On February 4, 2013, in conjunction with
the increase in authorized number of shares to 970,000,000, the Company
determined it had adequate authorized shares to settle all of these agreements.
As such, the Company adjusted the derivative liability to fair value on February
4, 2013 and reclassified the derivative liability to equity. The fair value of
the derivative liability of $474,954 (a non-cash item) as of February 4, 2013
was determined using the Binomial Option Pricing model with the following
assumptions: dividend yield: 0%; volatility: 156.52%; risk free rate: 0.38%; and
expected life: 3.5 years. The Company recorded a loss on change in derivative
liabilities of $84,907 during the year ended December 31, 2013.
F-46
Reset warrants
On October 1, 2012, in connection with
the forbearance agreement with Northstar as discussed in Note 8 above, the
Company issued an aggregate of 15,000,000 common stock purchase warrants to
purchase the Company’s common stock with an exercise price of $0.014 per share
for ten years with anti-dilutive (reset) provisions.
The Company has identified embedded
derivatives related to the issued warrants. These embedded derivatives included
certain and anti-dilutive (reset) provisions. The accounting treatment of
derivative financial instruments requires that the Company record fair value of
the derivatives as of the inception date and to fair value as of each subsequent
reporting date.
At December 31, 2013, the fair value of
the reset provision of $146,855 was determined using the Binomial Option Pricing
model with the following assumptions: dividend yield: 0%; volatility: 160.41%;
risk free rate: 3.04%; and expected life: 8.75 years. The Company recorded a
gain on change in derivative liabilities of $74,324 during the year ended
December 31, 2013.
Convertible
notes
During the year ended December 31, 2013,
the Company issued convertible notes (see Note 7 above).
These notes are convertible into common
stock, at holders’ option, at a discount to the market price of the Company’s
common stock. The Company has identified the embedded derivatives related to
these notes relating to certain anti-dilutive (reset) provisions. These embedded
derivatives included certain conversion features. The accounting treatment of
derivative financial instruments requires that the Company record fair value of
the derivatives as of the inception date of Asher Note and to fair value as of
each subsequent reporting date.
The fair value of the embedded
derivatives at December 31, 2013, in the amount of $256,956, was determined
using the Binomial Option Pricing Model based on the following assumptions: (1)
dividend yield of 0%; (2) expected volatility of 160.41%, (3) weighted average
risk-free interest rate of 0.07 to 0.10%, (4) expected lives of 0.27 to 0.60
years, and (5) estimated fair value of the Company’s common stock of $0.01 per
share. The Company recorded a gain on change in derivative liabilities of
$39,762 during the year ended December 31, 2013.
Based upon ASC 840-15-25 (EITF Issue
00-19, paragraph 11) the Company has adopted a sequencing approach regarding the
application of ASC 815-40 to its outstanding convertible notes. Pursuant to the
sequencing approach, the Company evaluates its contracts based upon earliest
issuance date.
At December 31, 2013, the aggregate
derivative liabilities was valued at $403,811, the Company believes an event
under the contract that would create an obligation to settle in cash or other
current assets is remote and has classified the obligation as a long term
liability.
NOTE 10 – STOCKHOLDERS’
EQUITY
Preferred
stock
On August 17, 2012, the board of
directors designated 5,000,000 shares of preferred stock as Series A Convertible
Preferred Stock which was increased to 20,000,000 shares of preferred stock as
Series A Convertible Preferred Stock. Each share of preferred stock is
convertible into equal number of common shares at the option of the holder;
entitled to 20 votes on all matters presented to be voted by the holders of
common stock; upon event of liquidation, entitled to amount equal to stated
value plus any accrued and unpaid dividends or other fees before distribution to
junior securities. In lieu of the initial two payments due to Northstar, the
parties have determined to modify the voting rights of the Series A Convertible
Preferred Stock from 20 votes per share on matters to be voted on by the common
stock holders to 25 votes per share on matters to be voted on by the common
stock holders (see Note 8 above).
During the year ended December 31, 2013,
the Company issued an aggregate of 20,000,000 shares of Series A Convertible
Preferred Stock for principle payment and settlement of forbearance (see note 8
above).
Common
stock
On September 19, 2011, the Company
amended its Articles of Incorporation to increase the number of authorized
shares to 200,000,000, consisting of 5,000,000 $0.001 par value preferred stock
and 195,000,000 $0.001 common stock.
F-47
On
August 6, 2008, the Company amended its Articles of Incorporation to increase
the number of authorized shares of its common stock from 50 million to 75
million shares. This amendment was approved by the Company’s shareholders at the
Annual Meeting of Shareholders held on July 30, 2008.
In September 2007, by way of a written
consent, the Company’s shareholders holding a majority of its outstanding shares
of common stock, the Company’s shareholders approved an amendment to Bioheart’s
Articles of Incorporation, increasing the number of authorized shares of capital
stock so that, following the reverse stock split that was effectuated on
September 27, 2007, the Company had 50 million shares of common stock authorized
with a par value of $0.001 per share and five million shares of preferred stock
authorized with a par value of $0.001 per share.
On February 4, 2013, the Company amended
its Articles of Incorporation to increase the number of authorized shares to
970,000,000, consisting of 20,000,000 $0.001 par value preferred stock and
950,000,000 $0.001 common stock.
On February 22, 2008 the Company
completed its initial public offering (“IPO”) pursuant to which it sold
1,100,000 shares of common stock at a price per share of $5.25 for net proceeds
of approximately $1.45 million after deducting underwriter discounts of
approximately $400,000 and offering costs of approximately $3.92 million. The
Consolidated Statement of Cash Flows for the year ended December 31, 2008
reflects the Company’s receipt of approximately $4.24 million of “Proceeds from
(payments for) initial public offering of common stock, net”. The $4.24 million
cash proceeds figure is approximately $2.79 million higher than the $1.45
million net proceeds figure identified above due to payment of $2.79 million of
various offering expenses prior to January 1, 2008. During the year ended
December, 31, 2013, the Company issued 50,029,227 shares of common stock for
proceeds of $855,000.
During the year ended December 31, 2013,
the Company issued 31,052,141 shares of common stock issued under its standby
equity distribution agreement with Greystone Capital Partners.
During the year ended December 31, 2013,
the Company issued an aggregate of 5,656,340 shares of its common stock for
settlement of $82,339 of accounts payable. In connection with the settlement,
the Company recorded a loss on settlement of debt of $74,877.
During the year ended December 31, 2013,
the Company issued 2,500,000 shares of its common stock in connection with the
issuance of a note payable.
During the year ended December 31, 2013,
the Company issued 57,967,906 shares of its common stock in connection with the
settlement and/or conversion of various notes payable.
During the year ended December 31, 2013,
the Company issued 34,890,348 shares of its common stock in connection with the
settlement of related party notes payable and advances.
During the year ended December 31, 2013,
the Company issued 9,408,718 shares of its common stock in settlement of
interest and penalty in connection with convertible debt.
During the year ended December 31, 2013,
the Company issued 6,220,263 shares of its common stock services rendered valued
at $85,151.
During the year ended December 31, 2012,
the Company issued an aggregate of 952,851 shares of its common stock, valued at
$34,600, in exchange for services rendered.
During the year ended December 31, 2012,
the Company issued an aggregate of 51,751,138 shares of its common stock in
exchange for $720,214 of outstanding notes payable and related accrued interest.
During the year ended December 31, 2012,
the Company issued an aggregate of 700,000 shares of its common stock in
exchange for $14,000 of accrued liabilities.
During the year ended December 31, 2011,
the Company issued an aggregate of 1,982,995 shares of its common stock on
exercise of options.
During the year ended December 31, 2011,
the Company issued an aggregate of 27,120,856 shares of its common stock in
exchange for $1,542,109 of outstanding notes payable and related accrued
interest.
F-48
During the year ended December 31, 2011,
the Company issued an aggregate of 1,000,000 shares of its common stock, valued
at $115,035, in exchange for services rendered.
During the year ended December 31, 2011,
the Company issued an aggregate of 1,272,730 shares of its common stock in
settlement of outstanding related party advance in the amount of $140,000.
In 2010, the Company also sold, in a
private placement initiated in 2009, an aggregate of 1,512,890 shares of its
common stock and warrants (the “Warrants”) to purchase 453,867 shares of its
common stock for aggregate gross cash proceeds of approximately $852,964. The
Warrants are (i) exercisable solely for cash at a weighted average exercise
price of $0.68 per share, (ii) non-transferable for six months following
issuance and (iii) exercisable, in whole or in part, at any time and from time
to time during the period commencing on the date that is six months and one day
following the date of issuance and ending on the third year anniversary of the
date of issuance. (Unaudited)
In
2010, the Company issued, in connection with the conversion of $1,121,195 of
debt an aggregate of 7,459,720 shares of its common stock and warrants (the
“Warrants”) to purchase 3,729,860 shares of its common stock. The Warrants are
(i) exercisable solely for cash at an exercise price of $0.15 per share, (ii)
non-transferable for six months following issuance and (iii) exercisable, in
whole or in part, at any time and from time to time during the period commencing
on the date that is six months and one day following the date of issuance and
ending on the third year anniversary of the date of issuance. The shares were
issued under the same terms as the above mentioned private placement and
therefore are included in issuance of common stock in the consolidated statement
of shareholders deficit. (Unaudited)
In
2010, the Company also sold, in a private placement, an aggregate of 1,553,885
shares of its common stock for aggregate gross cash proceeds of approximately
$234,020. (Unaudited)
In
2010, the Company also sold, in a private placement, an aggregate of 808,210
shares of its common stock and warrants (the “Warrants”) to purchase 404,105
shares of its common stock for aggregate gross cash proceeds of approximately
$135,885. The Warrants are (i) exercisable solely for cash at a weighted average
exercise price of $0.20 per share, (ii) non-transferable for six months
following issuance and (iii) exercisable, in whole or in part, at any time and
from time to time during the period commencing on the date that is six months
and one day following the date of issuance and ending on the third year
anniversary of the date of issuance. (Unaudited)
As
of December 31, 2010 the Company recorded a Subscription Receivable, under the
above mentioned private placement, of 20,000 shares of its common stock and
warrants (the “Warrants”) to purchase 10,000 shares of its common stock for
aggregate gross cash proceeds of approximately $3,800. The Warrants are (i)
exercisable solely for cash at a weighted average exercise price of $0.23 per
share, (ii) non-transferable for six months following issuance and (iii)
exercisable, in whole or in part, at any time and from time to time during the
period commencing on the date that is six months and one day following the date
of issuance and ending on the third year anniversary of the date of issuance.
(Unaudited)
In 2010, the Company issued, in
connection with services, an aggregate of 529,520 shares of its common stock and
warrants (the “Warrants”) to purchase 158,856 shares of its common stock. The
Warrants are (i) exercisable solely for cash at a weighted average exercise
price of $0.78 per share, (ii) non-transferable for six months following
issuance and (iii) exercisable, in whole or in part, at any time and from time
to time during the period commencing on the date that is six months and one day
following the date of issuance and ending on the third year anniversary of the
date of issuance. (Unaudited)
In
2010, the Company issued an aggregate of 831,526 shares of its common stock, in
connection with the exercise of stock options, issued for Accounts Payables.
(Unaudited)
In
2010, the Company issued, in connection with the Bank of America guarantor’s
liability of $2,172,000 principal along with accrued expenses, an aggregate of
4,794,430 shares of its common stock and warrants (the “Warrants”) to purchase
1,438,329 shares of its common stock. The Warrants are (i) exercisable solely
for cash at an weighted average exercise price of $0.74 per share, (ii)
non-transferable for six months following issuance and (iii) exercisable, in
whole or in part, at any time and from time to time during the period commencing
on the date that is six months and one day following the date of issuance and
ending on the third year anniversary of the date of issuance.
(Unaudited)
In
December 2009, the Company also sold, in a private placement an aggregate of
255,830 shares of its common stock and warrants (the “Warrants”) to purchase
76,749 shares of its common stock for aggregate gross cash proceeds of
approximately $188,996. The Warrants are (i) exercisable solely for cash at a
weighted average exercise price of $0.89 per share, (ii) non-transferable for
six months following issuance and (iii) exercisable, in whole or in part, at any
time and from time to time during the period commencing on the date that is six
months and one day following the date of issuance and ending on the third year
anniversary of the date of issuance. (Unaudited)
F-49
In 2009, the Company also sold, in a
private placement initiated in 2008, an aggregate of 2,509,480 shares of its
common stock and warrants (the “Warrants”) to purchase 752,844 shares of its
common stock for aggregate gross cash proceeds of approximately $1.74 million.
The Warrants are (i) exercisable solely for cash at a weighted average exercise
price of $0.83 per share, (ii) non-transferable for six months following
issuance and (iii) exercisable, in whole or in part, at any time and from time
to time during the period commencing on the date that is six months and one day
following the date of issuance and ending on the third year anniversary of the
date of issuance. In connection with the private placement, the Company paid to
a finder who introduced certain investors to the Company aggregate cash fees of
$17,856 and warrants to purchase 26,592 shares of common stock at a weighted
average exercise price of $0.89 per share. The warrants issued to the finder
have the same terms and conditions as the Warrants issued in the private
placement. The 2008 private placement was closed on October 31, 2009, with total
capital raised of $3,887,032. (Unaudited)
In
2008, the Company also sold, in a private placement, an aggregate of 1,230,280
shares of its common stock and warrants (the “Warrants”) to purchase 369,084
shares of its common stock for aggregate gross cash proceeds of approximately
$2.14 million. The Warrants are (i) exercisable solely for cash at a weighted
average exercise price of $2.09 per share, (ii) non-transferable for six months
following issuance and (iii) exercisable, in whole or in part, at any time and
from time to time during the period commencing on the date that is six months
and one day following the date of issuance and ending on the third year
anniversary of the date of issuance. In connection with the private placement,
the Company paid to a finder who introduced certain investors to the Company
aggregate cash fees of $24,325 and warrants to purchase 24,325 shares of common
stock at a weighted average exercise price of $1.95 per share. The warrants
issued to the finder have the same terms and conditions as the Warrants issued
in the private placement. (Unaudited)
In
2007, the Company sold 529,432 shares of common stock at a price of $7.69 per
share to various investors for net proceeds of approximately $3.9 million.
(Unaudited)
In
2006, the Company sold 1,069,699 shares of common stock at a price of $7.69 per
share to various investors. The Company also issued 63,566 shares in exchange
for services at a price ranging from $5.67 to $7.69 per share.
(Unaudited)
In
2005, the Company sold 1,994,556 shares of common stock at a price of $5.67 per
share to various investors. The Company also issued 1,210 shares in exchange for
services and issued 95,807 shares in exchange for debt at a price of $5.67 per
share. (Unaudited)
In
2004, the Company sold 808,570 shares of common stock at a price of $5.67 per
share to various investors. The Company also issued 1,854 shares to various
vendors in exchange for services valued at $10,500. The Company also issued
15,150 shares to the Company’s Chairman of the Board as compensation for
services valued at $85,830. (Unaudited)
In
March 2003, the Company effected a recapitalization. The recapitalization
provided two shares of common stock for every one share issued as of that date.
The Company’s former Chairman of the Board and founding shareholder, who owned
4,405,541 shares of common stock, did not participate in the recapitalization.
The number of shares and prices per share in the accompanying financial
statements has been retroactively adjusted to reflect the effect of the
recapitalization. (Unaudited)
After the 2003 recapitalization, the Company sold 561,701 shares of
common stock at a price of $5.67 per share to various investors. The Company
issued 72,980 shares valued at $416,383 to employees as compensation for
services related to the closing of various locations. The Company also issued
4,248 shares to various vendors in exchange for services valued at $24,066 and
issued 67,073 shares to the Company’s former Chairman of the Board as
compensation for services provided to the Company during 2003 and 2002. The
shares were valued based on the underlying market price of the common stock and
did not differ materially from the fair value of the common stock issued.
(Unaudited)
In
2002, the Company sold 1,092,883 shares of common stock at a price of $6.47 per
share to various investors. The Company also issued 35,137 shares to various
vendors in exchange for services valued at $227,503. (Unaudited)
In
2001, the Company sold 985,668 shares of common stock at a price of $6.47 per
share to various investors. The Company also issued 8,291 shares to various
vendors in exchange for services valued at $54,001 and issued 81,084 shares to
the Company’s Chairman of the Board as compensation for services provided to the
Company during 2001. (Unaudited)
F-50
In 2000, the Company sold 1,493,575
shares of common stock at a price of $6.47 per share to various investors. Of
the 1,493,575 shares sold in 2000, payment on 77,222 of these shares was not
received until January 2001. The Company also issued 7,964 shares to various
vendors in exchange for services valued at $52,001. (Unaudited)
In
1999, the Company’s former Chairman of the Board and founding shareholder
contributed $400,000 to the Company in exchange for 4,324,458 shares of common
stock. (Unaudited)
Former Chairman of the
Board Paid in and Contributed Capital
In 2006, the Company’s former Chairman of
the Board was issued 2,903 shares of the Company’s common stock at a price of
$5.67 per share in exchange for $16,443 of services provided during the year.
(Unaudited)
In
2005, the Company’s former Chairman of the Board was issued 95,807 shares of the
Company’s common stock at a price of $5.67 per share in exchange for $542,787 of
debt due to travel and other related expenses advanced by the Company’s Chairman
of the Board during the previous three years. (Unaudited)
The Company’s former Chairman of the Board elected not to receive salary
payments of $85,830, $130,000 and $250,000 for services provided to the Company
during 2004, 2003 and 2002, respectively. Such amounts were converted into
15,150, 22,946 and 44,127 shares of the Company’s common stock at a price of
$5.67 per share on December 31, 2004 and 2003, respectively, where the 2003 and
2002 shares were both issued in 2003. The shares were valued based on the
underlying market price of the common stock and did not differ materially from
the fair value of the common stock issued.
In 2001, the Company’s former Chairman of
the Board also elected not to receive a salary payment or a stock conversion of
$250,000 for services provided during 2001.
In 2000, the Company’s former Chairman of
the Board contributed $800,000 to the Company and elected not to receive payment
for $250,000 of salary related to services provided to the Company during 2000.
Such amounts were recorded as contributed capital during 2000. On June 28, 2001,
the Company’s Board of Directors approved the conversion of this contributed
capital and salary deferral into 81,084 shares of the Company’s common stock at
a price of $12.94 per share.
NOTE 11 — STOCK OPTIONS
AND WARRANTS
Stock
Options
In December 1999, the Board of Directors
and shareholders adopted the 1999 Officers and Employees Stock Option Plan, or
the Employee Plan, and the 1999 Directors and Consultants Stock Option Plan, or
the Director Plan. The Employee Plan and the Director Plan are collectively
referred to herein as the Plans. The Plans are administered by the Board of
Directors and the Compensation Committee. The objectives of the Plans include
attracting and retaining key personnel by encouraging stock ownership in the
Company by such persons. In February 2010, the Directors & Consultants Plan
was amended to extend the termination date of the Plan to December 1, 2011.
In April 1, 2013, the Board of Directors
approved, subject to shareholder approval, the establishment of the Bioheart
2013 Omnibus Equity Compensation Plan, or the “2013 Omnibus Plan”. The 2013
Omnibus Plan reserves up to fifty million shares of common stock for issuance.
Effective April 1, 2013, the Board of
Directors resolved that stock options granted for the past three years as of
February 25, 2013 be repriced for employees, management and board members, at
the exercise price of the average of the last 5 trading days’ closing price as
of February 25, 2013. Subsequently, this action was rescinded and the repricing
date was set for August 5, 2013.
F-51
A
summary of options at December 31, 2013 and activity during the year then ended
is presented below:
|
|
|
Weighted- |
|
|
|
Average |
|
|
Weighted- |
Remaining |
|
|
Average |
Contractual |
|
Shares |
Exercise Price |
Term (in years) |
| Options outstanding at January 1, 2012 |
4,636,318 |
$1.20 |
8.1 |
| Granted |
3,300,000 |
$0.04 |
|
| Exercised |
- |
|
|
| Forfeited/Expired |
(82,942) |
$5.57 |
|
| Options outstanding at December 31, 2012 |
7,853,376 |
$0.67 |
8.2 |
| Granted |
17,400,000 |
$0.016 |
9.9 |
| Exercised |
- |
|
|
| Forfeited/Expired |
(1,340,433) |
$1.08 |
|
| Options outstanding at December 31, 2013 |
23,912,943 |
$0.15 |
9.0 |
| Options exercisable at December 31, 2013 |
6,600,443 |
$0.48 |
8.0 |
| Available for grant at December 31, 2013 |
32,600,000 |
|
|
The following information applies to
options outstanding and exercisable at December 31, 2013:
|
|
Options Outstanding |
|
Options Exercisable |
|
|
|
|
|
Weighted- |
|
|
|
|
|
|
|
|
|
|
|
Average |
|
Weighted- |
|
|
|
Weighted- |
|
|
|
|
|
Remaining |
|
Average |
|
|
|
Average |
|
|
|
|
|
Contractual |
|
Exercise |
|
|
|
Exercise |
|
|
|
Shares |
|
Term |
|
Price |
|
Shares |
|
Price |
|
| $0.00 – $0.70 |
|
23,290,000 |
|
9.2 |
|
$ |
0.03 |
|
5,977,500 |
|
$ |
0.07 |
|
| $0.71 – $1.28 |
|
162,286 |
|
4.4 |
|
$ |
0.80 |
|
162,286 |
|
$ |
0.80 |
|
| $5.25 – $5.67 |
|
435,945 |
|
2.0 |
|
$ |
5.57 |
|
435,945 |
|
$ |
5.57 |
|
| $7.69 |
|
24,712 |
|
2.6 |
|
$ |
7.69 |
|
24,712 |
|
$ |
7.69 |
|
|
|
23,912,943 |
|
9.0 |
|
$ |
0.15 |
|
6,600,443 |
|
$ |
0.48 |
|
On January 16, 2012, the
Company granted 500,000 employee stock options in connection services rendered
at the exercise price of $0.10 per share vesting over four years from the date
of issuance.
The fair values of the
employee options issued on January 16, 2012 were determined using the Black
Scholes option pricing model with the following assumptions: Dividend yield: 0%;
Volatility: 164.79% and Risk free rate: 1.89%.
On August 6, 2012, the
Company granted an aggregate 2,800,000 employee stock options in connection
services rendered at the exercise price of $0.03 per share vesting over four
years from the date of issuance.
The fair values of the
employee options issued on August 6, 2012 were determined using the Black
Scholes option pricing model with the following assumptions: Dividend yield: 0%;
Volatility: 164.91% and Risk free rate: 1.59%.
On August 5, 2013, the
Company re-priced options previously issued from 2011 through 2012 for current
employees and officers in aggregate of 4,890,000 options with previous exercise
prices from $0.03 to $0.21 per share to $0.01694 per share, all other terms
remaining unchanged. The gross change in fair value, determined using the Black
Scholes option pricing model, of $1,630 was charged to current period
operations.
On August 1, 2013, the
Company issued an aggregate 15,000,000 options to purchase the Company’s common
stock at $0.01576 per share to employees, exercisable over 4 years. The fair
value of $245,749, determined using the Black Scholes option pricing model with
the following assumptions: Dividend yield: 0%; Volatility: 153.27% and Risk free
rate: 2.74%, of which $25,599 was charged to current period operations.
F-52
On
September 1, 2013, the Company issued an aggregate 2,400,000 options to purchase
the Company’s common stock at $0.01654 per share, respectively; to officers and
employees, exercisable immediately. The fair value of $37,823, determined using
the Black Scholes option pricing model with the following assumptions: Dividend
yield: 0%; Volatility: 150.98% and Risk free rate: 2.78%, was charged to current
period operations.
The fair value of all options vesting
during the year ended December 31, 2013 and 2012 of $125,515 and $76,674,
respectively, was charged to current period operations.
Warrants
A summary of common stock
purchase warrants at December 31, 2013 and activity during the year then ended
is presented below:
|
|
|
|
|
|
|
Weighted- |
|
|
|
|
|
|
|
Average |
|
|
|
|
|
Weighted- |
|
Remaining |
|
|
|
|
|
Average |
|
Contractual |
|
|
|
|
|
Exercise |
|
Term (in |
|
|
Shares |
|
|
Price |
|
years) |
| Outstanding at January 1, 2012 |
|
32,610,075 |
|
|
$
|
0.86 |
|
3.8 |
| Issued |
|
42,396,432 |
|
|
$ |
0.018 |
|
5.78 |
| Exercised |
|
— |
|
|
$ |
0.00 |
|
|
| Forfeited |
|
(933,185) |
|
|
$ |
0.76 |
|
|
| Outstanding at December 31, 2012 |
|
74,073,322 |
|
|
$ |
0.37 |
|
4.5 |
| Issued |
|
50,350,536 |
|
|
$ |
0.016 |
|
9.2 |
| Exercised |
|
— |
|
|
$ |
|
|
|
| Expired |
|
(6,345,002) |
|
|
$ |
0.38 |
|
|
| Outstanding at December 31, 2013 |
|
118,078,856 |
|
|
$ |
0.22 |
|
6.3 |
| Exercisable at December 31, 2013 |
|
101,371,743 |
|
|
$ |
0.13 |
|
5.7 |
In conjunction with the authorized
issuance of common stock, the Company granted approximately 50 million common
stock purchase warrants during the year ended December 31, 2013.
The following information applies to
common stock purchase warrants outstanding and exercisable at December 31, 2013:
|
|
Warrants Outstanding |
|
Warrants Exercisable |
|
|
|
|
Weighted- |
|
|
|
|
|
|
|
|
|
|
Average |
|
Weighted- |
|
|
|
Weighted- |
|
|
|
|
Remaining |
|
Average |
|
|
|
Average |
|
|
|
|
Contractual |
|
Exercise |
|
|
|
Exercise |
|
|
|
Shares |
Term |
|
Price |
|
Shares |
|
Price |
|
| $0.01 – $0.50 |
|
111,829,723 |
6.3 |
|
$ |
0.03 |
|
96,667,060 |
|
$ |
0.03 |
|
| $0.52 – $0.68 |
|
2,699,675 |
5.3 |
|
$ |
0.58 |
|
2,699,675 |
|
$ |
0.58 |
|
| $0.70 – $1.62 |
|
848,176 |
6.0 |
|
$ |
0.71 |
|
848,176 |
|
$ |
0.71 |
|
| $5.67 – $7.69 |
|
2,701,282 |
8.9 |
|
$ |
7.55 |
|
1,156,832 |
|
$ |
7.35 |
|
|
|
118,078,856 |
6.3 |
|
$ |
0.22 |
|
101,371,743 |
|
$ |
0.13 |
|
During the year ended December 31, 2012, in connection with the sale of
common stock, the Company issued an aggregate of 22,396,432 warrants to purchase
the Company’s common stock at an exercise prices from $0.014 to $0.03 per shares
exercisable in six months and expiring three years from issuance.
On September 21, 2012, the Company issued
5,000,000 warrants to purchase the Company’s common stock at $0.02 per share,
expiring 10 years from the date of issuance as payment of interest.
F-53
The fair value of $119,023, determined using the Black Scholes option
pricing model with the following assumptions: Dividend yield: 0%; Volatility:
163.45% and Risk free rate: 1.779%, was charged to current period
operations.
On October 1, 2012, the Company issued
15,000,000 warrants to purchase the Company’s common stock at $0.014 per share,
expiring 10 years from the date of issuance as payment of interest with certain
reset provisions.
The fair value of $311,190, determined
using the Binomial lattice option pricing model with the following assumptions:
Dividend yield: 0%; Volatility: 155.41% and Risk free rate: 1.64%, was charged
to current period operations.
NOTE 12 — COMMITMENTS
AND CONTINGENCIES
Leases
The Company entered into several
operating lease agreements for facilities and equipment. Terms of certain lease
arrangements include renewal options, escalation clauses, payment of executory
costs such as real estate taxes, insurance and common area
maintenance.
In February 2010, the Company amended its
facility lease to extend the term of the lease until January 2013.
In August 2011, the Company amended its
facility lease to eliminate excess space. The amendment contains terms similar
to the terms of the existing facility lease, including escalation
clauses.
In July 2013, the Company amended its
facility lease to extend the term of the lease until July 31, 2014. Approximate
annual future minimum lease obligations under non-cancelable operating lease
agreements as of December 31, 2013 are as follows:
| Year ending December 31, |
|
|
| 2014 |
$ |
48,195 |
| Total |
$ |
48,195 |
Rent expense was $123,216 and $89,507 for
the years ended December 31, 2013 and 2012, respectively and $2,102,062 for the
cumulative period from August 12, 1999 (date of inception) to December 31,
2013.
Royalty
Payments
The Company is obligated to pay royalties
on commercial sales of certain products that may be developed and sold under
various licenses and agreements that have been obtained by the
Company.
The Company has entered into a licensing
agreement, which include the potential for royalty payments, as
follows: William Beaumont Hospital
In June 2000, the Company entered into an
exclusive license agreement to use certain patents for the life of the patents
in future projects. The patents expire in 2015. In addition to a payment of
$55,000, the Company made to acquire the license, the Company is required to pay
an annual license fee of $10,000 and royalties ranging from 2% to 4% of net
sales of products that are covered by the patents. In order to maintain the
exclusive license rights, the agreement also calls for a minimum annual royalty
threshold. The minimum royalty threshold was $200,000 for 2011 and $200,000 for
2010. This minimum royalty threshold will remain $200,000 for 2012 and
thereafter. As of December 31, 2013, the Company has not made any payments other
than the initial payment to acquire the license. At December 31, 2013 and
December 31, 2012, the Company’s liability under this agreement was $2,122,130
and $1,825,675, respectively, which is reflected as a component of accrued
expenses on the balance sheets (see Note 5).
During the year ended December 31, 2013
and 2012, the Company incurred expenses of $210,000, $210,000 respectively, and
$2,122,130 from August 12, 1999 (date of inception) to December 31, 2013. The
Company has accrued interest for the past due commitment at 2% over the prime
rate per the terms of the agreement. The Company has included $2,122,130 in
accrued expenses as of December 31, 2013.
Approximate annual future minimum
obligations under this agreement as of December 31, 2013 are as follows:
F-54
| Year Ending December 31, |
|
| 2014 |
210,000 |
| 2015 |
210,000 |
| Total |
$420,000 |
Consulting
agreements
On November 20, 2013, the
Company entered into an investment banking agreement with Cassel Salpeter &
Co. (“CSC”), who will act as exclusive third party financial advisor in
connection with investment banking matters. The term of the Investment Banking
Agreement shall be for a period of twenty four months unless terminated or
extended in accordance with its terms. For these services, CSC will receive a
one-time $25,000 fee, $5,000 monthly
fees and 5,207,630 ten year common
stock purchase warrants, exercisable at $.0113 and applicable consideration in
the event the closing of a Mezzanine Financing consisting of non-convertible
subordinated debt and/or sale of equity securities. The Company will also
reimburse CSC for its reasonable out-of-pocket expenses associated with the
services provided pursuant to the Investment Banking Agreement. As of December
31, 2013, the Company accrued $32,424 under the agreement.
Contingency for
Registration of the Company’s common stock
The Company believes that
it may have issued options to purchase common stock to certain of its employees,
directors and consultants in California in violation of the registration or
qualification provisions of applicable California securities laws. As a result,
the Company intends to make a rescission offer to these persons. The Company
will make this offer to all persons who have a continuing right to rescission,
which it believes to include two persons. In the rescission offer, in accordance
with California law, the Company will offer to repurchase all unexercised
options issued to these persons at 77% of the option exercise price multiplied
by the number of option shares, plus interest at the rate of 7% from the date
the options were granted. Based upon the number of options that were subject to
rescission as of December 31, 2009, assuming that all such options are tendered
in the rescission offer, the Company estimated that its total rescission
liability would be up to approximately $371,000. However, as the Company
believes there is only a remote likelihood the rescission offer will be accepted
by any of these persons in an amount that would result in a material expenditure
by the Company, no liability was recorded as of December 31, 2013 or 2012.
Litigation
The Company is subject to
other legal proceedings that arise in the ordinary course of business. In the
opinion of management, as of December 31, 2013, the amount of ultimate liability
with respect to such matters, if any, in excess of applicable insurance
coverage, is not likely to have a material impact on the Company’s business,
financial position, results of operations or liquidity. However, as the outcome
of litigation and other claims is difficult to predict significant changes in
the estimated exposures could exist.
NOTE 13 -INCOME TAXES
The Company follows
Accounting Standards Codification subtopic 740, Income Taxes (“ASC 740”) which
requires the recognition of deferred tax liabilities and assets for the expected
future tax consequences of events that have been included in the financial
statements or tax returns. Under such method, deferred tax assets and
liabilities are recognized for the future tax consequences attributable to
differences between the financial statement carrying amounts of existing assets
and liabilities and their respective tax bases using enacted tax rates in effect
for the year in which the differences are expected to reverse.
F-55
The difference between
income tax expense computed by applying the federal statutory corporate tax rate
and actual income tax expense is as follows:
| |
|
|
2013 |
|
|
|
2012 |
|
|
|
Income taxes using U.S. federal
statutory rate |
$ |
(1,068,708 |
) |
|
|
(1,365,599 |
) |
|
|
State income taxes, net of federal
benefit |
|
(108,788 |
) |
|
|
(80,708 |
) |
|
|
Stock Option Expirations |
|
78,864 |
|
|
|
326,995 |
|
|
|
Net Operating Loss
adjustments |
|
(20,008 |
) |
|
|
196,416 |
|
|
|
Nontaxable Gain on Derivative
Instrument |
|
(9,921 |
) |
|
|
(40,730 |
) |
|
| |
Change in Valuation
Allowance |
|
1,127,875 |
|
|
|
974,536 |
|
|
|
Other |
|
686 |
|
|
|
(10,910 |
) |
|
|
|
$ |
— |
|
|
$ |
— |
|
|
At December 31, 2013, the significant components
of the deferred tax assets (liabilities) are summarized below:
| |
|
|
2013 |
|
|
2012 |
|
|
Deferred tax
assets: |
|
|
|
|
|
|
|
Stock Based Compensation |
$ |
4,465,854 |
|
$ |
4,505,907 |
|
|
Net Operating Losses |
|
35,429,429 |
|
|
34,266,157 |
|
|
Other |
|
125,317 |
|
|
120,661 |
|
|
Total deferred tax
assets |
|
40,020,600 |
|
|
38,892,725 |
|
|
|
|
|
Deferred tax
liabilities: |
|
— |
|
|
— |
|
| |
Total deferred tax liabilities |
|
— |
|
|
— |
|
| |
Valuation allowance |
|
40,020,600 |
|
|
38,892,725 |
|
|
Net deferred tax assets |
$ |
— |
|
$ |
— |
|
As of December 31, 2013 and
December 31, 2012, the Company had U.S. federal net operating loss carry
forwards of approximately $94.2 million and $91.1 million, respectively, which
expire at various dates from 2019 through 2033. These net operating loss carry
forwards may be used to offset future taxable income and thereby reduce the
Company’s U.S. federal income taxes. Section 382 of the Internal Revenue Code of
1986 (the “Code”) imposes an annual limit on the ability of a corporation that
undergoes a greater than 50% ownership change to use its net operating loss
carry forwards to reduce its tax liability. If in the future the Company issues
common stock or additional equity instruments convertible in common shares which
result in an ownership change exceeding the 50% limitation threshold imposed by
section 382 of the Code, the Company’s net operating loss carry-forwards may be
significantly limited as to the amount of use in a particular years. In
addition, all or a portion of the Company’s net operating loss carry forwards
may expire unutilized.
As of December 31, 2013 and
December 31, 2012, the Company had net operating loss carry forwards for state
income tax purposes of approximately $94.2 million and $91.1 million,
respectively, which expire at various dates from 2019 through 2033.
The Company
has provided a full valuation allowance against its net deferred tax assets,
since in the opinion of management based upon the earnings history of the
Company; it is more likely than not that the benefits of these assets will not
be realized.
The Company complies with
the provisions of FASB ASC 740-10 in accounting for its uncertain tax positions.
ASC 740-10 addresses the determination of whether tax benefits claimed or
expected to be claimed on a tax return should be recorded in the financial
statements. Under ASC 740-10, the Company may recognize the tax benefit from an
uncertain tax position only if it is more likely that not that the tax position
will be sustained on examination by the taxing authorities, based on the
technical merits of the position. Management has determined that the Company has
no significant uncertain tax positions requiring recognition under ASC
740-10.
The Company is subject to
income tax in the U.S., and certain state jurisdictions. The Company has not
been audited by the U.S. Internal Revenue Service, or any states in connection
with income taxes. The periods from December 31, 2006 to December 31, 2013
remain open to examination by the U.S. Internal Revenue Service, and state tax
authorities. In addition, federal and state tax authorities can generally reduce
a net operating loss (but not create taxable income) for a period outside the
statute of limitations in order to determine the correct amount of net operating
loss which may be allowed as a deduction against income for a period within the
statute of limitations.
F-56
The Company recognizes
interest and penalties related to unrecognized tax benefits, if incurred, as a
component of income tax expense.
NOTE 14 — FAIR VALUE
MEASUREMENT
The Company adopted the
provisions of Accounting Standards Codification subtopic 825-10, Financial
Instruments (“ASC 825-10”) on January 1, 2008. ASC 825-10 defines fair value as
the price that would be received from selling an asset or paid to transfer a
liability in an orderly transaction between market participants at the
measurement date. When determining the fair value measurements for assets and
liabilities required or permitted to be recorded at fair value, the Company
considers the principal or most advantageous market in which it would transact
and considers assumptions that market participants would use when pricing the
asset or liability, such as inherent risk, transfer restrictions, and risk of
nonperformance. ASC 825-10 establishes a fair value hierarchy that requires an
entity to maximize the use of observable inputs and minimize the use of
unobservable inputs when measuring fair value. ASC 825-10 establishes three
levels of inputs that may be used to measure fair value:
Level 1 – Quoted prices in
active markets for identical assets or liabilities.
Level 2 – Observable inputs
other than Level 1 prices such as quoted prices for similar assets or
liabilities; quoted prices in markets with insufficient volume or infrequent
transactions (less active markets); or model-derived valuations in which all
significant inputs are observable or can be derived principally from or
corroborated by observable market data for substantially the full term of the
assets or liabilities.
Level 3 – Unobservable
inputs to the valuation methodology that are significant to the measurement of
fair value of assets or liabilities.
All items required to be
recorded or measured on a recurring basis consist of derivative liabilities and
are based upon level 3 inputs.
To the extent that
valuation is based on models or inputs that are less observable or unobservable
in the market, the determination of fair value requires more judgment. In
certain cases, the inputs used to measure fair value may fall into different
levels of the fair value hierarchy. In such cases, for disclosure purposes, the
level in the fair value hierarchy within which the fair value measurement is
disclosed and is determined based on the lowest level input that is significant
to the fair value measurement.
Upon adoption of ASC
825-10, there was no cumulative effect adjustment to beginning retained earnings
and no impact on the financial statements.
The carrying value of the
Company’s cash and cash equivalents, accounts receivable, accounts payable,
short-term borrowings (including convertible notes payable), and other current
assets and liabilities approximate fair value because of their short-term
maturity.
As of December 31, 2013 and
2012, the Company did not have any items that would be classified as level 1 or
2 disclosures.
The Company recognizes its
derivative liabilities as level 3 and values its derivatives using the methods
discussed in notes 7 and 9. While the Company believes that its valuation
methods are appropriate and consistent with other market participants, it
recognizes that the use of different methodologies or assumptions to determine
the fair value of certain financial instruments could result in a different
estimate of fair value at the reporting date. The primary assumptions that would
significantly affect the fair values using the methods discussed in Notes 7 and
9 are that of volatility and market price of the underlying common stock of the
Company.
As of December 31, 2013 and
2012, the Company did not have any derivative instruments that were designated
as hedges.
The derivative liability as
of December 31, 2013, in the amount of $403,811 has a level 3 classification.
F-57
The following table
provides a summary of changes in fair value of the Company’s Level 3 financial
liabilities as of December 31, 2013:
| |
|
|
Excess
Share |
|
|
Warrant |
|
Debt |
|
|
|
|
Derivative |
|
|
Liability |
|
Derivative |
|
|
Balance, December 31, 2011 |
|
$ |
- |
|
|
|
|
- |
|
|
|
$ |
- |
|
|
|
Total (gains) losses |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Initial fair value of debt derivative at note
issuance |
|
|
|
|
|
|
|
- |
|
|
|
|
76,862 |
|
|
|
Initial fair value of derivative relating to reset
warrants |
|
|
- |
|
|
|
|
311,190 |
|
|
|
|
- |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Initial fair value of derivative relating to exceeding
authorized common shares |
|
|
427,663 |
|
|
|
|
- |
|
|
|
|
- |
|
|
|
Mark-to-market at December 31, 2012 |
|
|
(37,615 |
) |
|
|
|
(90,011 |
) |
|
|
|
7,651 |
|
|
|
Transfers out of Level 3 upon
conversion and settlement of notes |
|
|
|
|
|
|
|
|
|
|
|
|
(84,513 |
) |
|
|
Balance, December 31, 2012 |
|
$ |
390,048 |
|
|
|
$ |
221,179 |
|
|
|
$ |
- |
|
|
|
Total (gains) losses |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Initial fair value of debt derivative at note
issuance |
|
|
- |
|
|
|
|
- |
|
|
|
|
673,219 |
|
|
| |
Mark-to-market at December 31, 2013: |
|
|
84,906 |
|
|
|
|
(74,324 |
) |
|
|
|
(39,761 |
) |
|
|
Transfers out of Level 3 upon increase in authorized
shares |
|
|
(474,954 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Transfers out of Level 3 upon
conversion of notes payable |
|
|
- |
|
|
|
|
- |
|
|
|
|
(376,502 |
|
|
|
Balance, December 31, 2013 |
|
$ |
- |
|
|
|
$ |
146,855 |
|
|
|
$ |
256,956 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net Gain for the period included
in earnings relating to the liabilities held at
December 31, 2013
$ |
|
|
(84,906 |
) |
|
|
$ |
74,324 |
|
|
|
$ |
39,761 |
|
|
Fluctuations in the
Company’s stock price are a primary driver for the changes in the derivative
valuations during each reporting period. The Company’s stock price decreased
approximately 50% from December 31, 2012 to December 31, 2013. As the stock
price decreases for each of the related derivative instruments, the value to the
holder of the instrument generally decreases, therefore decreasing the liability
on the Company’s balance sheet. Additionally, stock price volatility is one of
the significant unobservable inputs used in the fair value measurement of each
of the Company’s derivative instruments. The simulated fair value of these
liabilities is sensitive to changes in the Company’s expected volatility.
Decreases in expected volatility would generally result in a lower fair value
measurement. A 10 percent change in pricing inputs and changes in volatilities
and correlation factors would result in less than a $60,318 change in our Level
3 fair value.
NOTE 15 — SUBSEQUENT
EVENTS
Subsequent stock
issuances
In January 2014, the
Company issued 1,977,155 shares of its common stock in settlement of 2013
services provided for $12,000 and 2,941,176 shares of its common stock in
settlement of $15,000 of outstanding convertible notes payable.
In February 2014, the
Company sold an aggregate of 25,114,500 shares of its common stock for net
proceeds of $331,500. In connection with the stock sale, the Company issued an
aggregate of 25,114,500 warrants to purchase the Company’s common stock for five
years at $0.011 to $0.0214 per share. In February 2014, the Company issued
1,746,032 shares of its common stock in settlement of $10,000 of outstanding
convertible notes payable. In February 2014, the Company issued an aggregate of
3,925,442 shares of its common stock in settlement of $55,000 of common stock
subscriptions received in 2013.
Options granted
On February 23, 2014, the
Company granted an aggregate of 15,000,000 options to purchase the Company’s
common stock to an officer and key employee at an exercise price of $0.019 for
ten years, vesting annually over four years. In addition, the Company granted
400,000 options to purchase the Company’s common stock to each current Director
of the Company who was also a board member in fiscal 2011 at an exercise price
of $0.019 per share for ten years, vesting immediately with a cashless exercise
provision. Additionally, the Company granted 400,000 options to purchase the
Company’s common stock to each current Director of the Company at an exercise
price of $0.019 per share for ten years, vesting immediately.
Option expiry terms
On February 23, 2013, the
Company affirmed that options issued to previous board members of the Company
will not cease and will terminate at the earlier of the date of exercise or
expiration of the option in accordance to its term.
F-58
Related debt conversions
On February 23, 2014, the
Board of Directors approved the conversion of an aggregate of $300,000 related
party notes payable at a conversion price of $0.019 per share
Subsequent
financing
On January 14, 2014, the
Company entered into a Securities Purchase Agreement with Asher Enterprises,
Inc. ("Asher"), for the sale of an 8% convertible note in the principal amount
of $32,500 (the "Note").
The Note bears interest at
the rate of 8% per annum. All interest and principal must be repaid on October
16, 2014. The Note is convertible into common stock, at Asher’s option, at a 45%
discount to the average of the three lowest closing bid prices of the common
stock during the 10 trading day period prior to conversion. In the event the
Company prepays the Note in full, the Company is required to pay off all
principal, interest and any other amounts owing multiplied by (i) 140% if
prepaid during the period commencing on the closing date through 179 days
thereafter. After the expiration of 180 days following the date of the Note, the
Company has no right of prepayment.
On February 10, 2014, the
Company entered into a Securities Purchase Agreement with Asher Enterprises,
Inc. ("Asher"), for the sale of an 8% convertible note in the principal amount
of $32,500 (the "Note").
The Note bears interest at
the rate of 8% per annum. All interest and principal must be repaid on November
12, 2014. The Note is convertible into common stock, at Asher’s option, at a 45%
discount to the average of the three lowest closing bid prices of the common
stock during the 10 trading day period prior to conversion. In the event the
Company prepays the Note in full, the Company is required to pay off all
principal, interest and any other amounts owing multiplied by (i) 140% if
prepaid during the period commencing on the closing date through 179 days
thereafter. After the expiration of 180 days following the date of the Note, the
Company has no right of prepayment.
On February 19, 2014, the
Company entered into a Securities Purchase Agreement with Daniel James
Management, Inc., for the sale of an 8% convertible note in the principal amount
of $35,000 (the "Note").
The Note bears interest at
the rate of 8% per annum. All interest and principal must be repaid on February
18, 2015. The Note is convertible into common stock, at Asher’s option, at a 47%
discount to the lowest daily closing bid price of the common stock during the 10
trading day period prior to conversion. In the event the Company prepays the
Note in full, the Company is required to pay off all principal at 150%, interest
and any other amounts.
F-59
PART II
INFORMATION NOT REQUIRED
IN PROSPECTUS
Item 13. Other Expenses of Issuance and Distribution
The following table sets
forth an itemization of all estimated expenses, all of which we will pay, in
connection with the issuance and distribution of the securities being
registered:
|
|
Nature of Expense: |
|
Amount |
|
|
|
SEC Registration Fee |
|
$ |
269.05 |
|
|
|
Accounting fees and expenses |
|
$ |
9,500 |
|
|
| |
Legal fees and expenses |
|
$ |
11,500 |
|
|
|
Miscellaneous |
|
$ |
1,000 |
|
|
|
Total |
|
$ |
22,269.05 |
* |
|
*Estimated
Item 14. Indemnification of Directors and Officers
We are incorporated under the laws of the State of Florida. Our articles
of incorporation require us to indemnify and limit the liability of directors to
the fullest extent permitted by the Florida Business Corporation Act, or the
“FBCA”, as it currently exists or as it may be amended in the future.
Pursuant to the FBCA, a Florida corporation may indemnify any person who
may be a party to any third party proceeding by reason of the fact that such
person is or was a director, officer, employee or agent of the corporation, or
is or was serving at the request of the corporation as a director, officer,
employee, or agent of another entity, against liability incurred in connection
with such proceeding (including any appeal thereof) if he or she acted in good
faith and in a manner he or she reasonably believed to be in, or not opposed to,
the best interests of the corporation, and, with respect to any criminal action
or proceeding, had no reasonable cause to believe his or her conduct was
unlawful.
In addition, in accordance with the FBCA, a Florida corporation is
permitted to indemnify any person who may be a party to a derivative action if
such person acted in any of the capacities set forth in the preceding paragraph,
against expenses and amounts paid in settlement not exceeding, in the judgment
of the board of directors, the estimated expenses of litigating the proceeding
to conclusion, actually and reasonably incurred in connection with the defense
or settlement of such proceeding (including appeals), provided that the person
acted under the standards set forth in the preceding paragraph. However, no
indemnification shall be made for any claim, issue, or matter for which such
person is found to be liable unless, and only to the extent that, the court
determines that, despite the adjudication of liability, but in view of all the
circumstances of the case, such person is fairly and reasonably entitled to
indemnification for such expenses which the court deems proper.
Any indemnification made under the above provisions, unless pursuant to a
court’s determination, may be made only after a determination that the person to
be indemnified has met the standard of conduct described above. This
determination is to be made by a majority vote of a quorum consisting of the
disinterested directors of the board of directors, by duly selected independent
legal counsel, or by a majority vote of the disinterested shareholders. The
board of directors also may designate a special committee of disinterested
directors to make this determination. Notwithstanding the foregoing, a Florida
corporation must indemnify any director, officer, employee or agent of a
corporation who has been successful in the defense of any proceeding referred to
above.
Generally, pursuant to the FBCA, a director of a Florida corporation is
not personally liable for monetary damages to our company or any other person
for any statement, vote, decision, or failure to act, regarding corporate
management or policy, unless: (a) the director breached or failed to perform his
duties as a director; and (b) the director’s breach of, or failure to perform,
those duties constitutes (i) a violation of criminal law, unless the director
had reasonable cause to believe his conduct was lawful or had no reasonable
cause to believe his conduct was unlawful, (ii) a transaction from which the
director derived an improper personal benefit, either directly or indirectly,
(iii) an approval of an unlawful distribution, (iv) with respect to a proceeding
by or in the right of the company to procure a judgment in its favor or by or in
the right of a shareholder, conscious disregard for the best interest of the
company, or willful misconduct, or (v) with respect to a proceeding by or in the
right of someone other than the company or a shareholder, recklessness or an act
or omission which was committed in bad faith or with malicious purpose or in a
manner exhibiting wanton and willful disregard of human rights, safety, or
property. The term “recklessness,” as used above, means the action, or omission
to act, in conscious disregard of a risk: (a) known, or so obvious that it
should have been known, to the directors; and (b) known to the director, or so
obvious that it should have been known, to be so great as to make it highly
probable that harm would follow from such action or omission.
110
Furthermore, under the FBCA, a Florida corporation is authorized to make
any other further indemnification or advancement of expenses of any of its
directors, officers, employees or agents under any bylaw, agreement, vote of
shareholders or disinterested directors, or otherwise, both for actions taken in
an official capacity and for actions taken in other capacities while holding
such office. However, a corporation cannot indemnify or advance expenses if a
judgment or other final adjudication establishes that the actions of the
director, officer, employee, or agent were material to the adjudicated cause of
action and the director, officer, employee, or agent (a) violated criminal law,
unless the director, officer, employee, or agent had reasonable cause to believe
his or her conduct was unlawful, (b) derived an improper personal benefit from a
transaction, (c) was or is a director in a circumstance where the liability for
unlawful distributions applies, or (d) engaged in willful misconduct or
conscious disregard for the best interests of the corporation in a proceeding by
or in right of the corporation to procure a judgment in its favor.
At present, there is no pending litigation or proceeding involving any of
our directors or executive officers as to which indemnification is required or
permitted and we are not aware of any threatened litigation or proceeding that
may result in a claim for indemnification.
We maintain a liability insurance policy, pursuant to which our directors
and officers may be insured against liability they incur for serving in their
capacities as directors and officers of our company, including liabilities
arising under the Securities Act or otherwise.
Insofar as indemnification for liabilities arising under the Securities
Act may be permitted to directors, officers or persons controlling us pursuant
to the foregoing provisions, we have been informed that, in the opinion of the
Securities and Exchange Commission, such indemnification is against public
policy as expressed in the Securities Act and is therefore unenforceable. In the
event that a claim for indemnification against such liabilities (other than the
payment by the registrant of expenses incurred or paid by a director, officer or
controlling person of the registrant in the successful defense of any action,
suit or proceeding) is asserted by such director, officer or controlling person
in connection with the securities being registered, we will, unless in the
opinion of our counsel the matter has been settled by controlling precedent,
submit to a court of appropriate jurisdiction the question whether such
indemnification by us is against public policy as expressed hereby in the
Securities Act and we will be governed by the final adjudication of such issue.
Item 15. Recent Sales of
Unregistered Securities
During the nine months ended September 30, 2014, the Company sold an
aggregate of 24,556,846 shares of the Company’s common stock and common stock
purchase warrants to purchase 24,556,846 shares of the Company’s common stock
for aggregate gross cash proceeds of $371,000. The warrants are (i) exercisable
solely for cash at an exercise price of $0.011 to $0.0275 per share, (ii)
non-transferable for six months following issuance and (iii) exercisable, in
whole or in part, at any time during the period commencing on the date that is
six months and one day following the date of issuance and ending on the tenth
year anniversary of the date of issuance.
111
The issuance of such shares of our common stock was effected in reliance
on the exemptions for sales of securities not involving a public offering, as
set forth in Rule 506 promulgated under the Securities Act of 1933, as mended
(the “Securities Act”) and in Section 4(2) of the Securities Act, based on the
following: the investors confirmed to us that they were “accredited investors,”
as defined in Rule 501 of Regulation D promulgated under the Securities Act and
had such background, education and experience in financial and business matters
as to be able to evaluate the merits and risks of an investment in the
securities; (b) there was no public offering or general solicitation with
respect to the offering; (c) the investors were provided with certain disclosure
materials and all other information requested with respect to our company; (d)
the investors acknowledged that all securities being purchased were “restricted
securities” for purposes of the Securities Act, and agreed to transfer such
securities only in a transaction registered under the Securities Act or exempt
from registration under the Securities Act; and (e) a legend was placed on the
certificates representing each such security stating that it was restricted and
could only be transferred if subsequent registered under the Securities Act or
transferred in a transaction exempt from registration under the Securities Act.
Item 16. Exhibits
The following exhibits are included as part of this Form S-1. References
to “the Company” in this Exhibit List mean Bioheart, Inc., a Florida
corporation.
| Exhibit No. |
|
Exhibit Description |
| 3.1(6) |
|
Amended and Restated
Articles of Incorporation of the registrant, as amended |
| 3.2(9) |
|
Articles of Amendment to the
Articles of Incorporation of the registrant |
| 3.3(8) |
|
Amended and Restated
Bylaws |
| 4.1(5) |
|
Loan and Security Agreement, dated
as of May 31, 2007 by and between BlueCrest Capital Finance, L.P. and the
registrant |
| 4.2(12) |
|
Notice of Event of Default,
from BlueCrest Venture Finance Master Fund Limited to the Company, dated
January 28, 2009 |
| 4.3(12) |
|
Notice of Acceleration, from
BlueCrest Venture Finance Master Fund Limited to the Company, dated
February 2, 2009 |
| 4.4(12) |
|
Amendment to Loan and
Security Agreement, between the Company and BlueCrest Venture Finance
Master Fund Limited, dated as of April 2, 2009 |
| 4.5(13) |
|
Grant of Security Interest
(Patents), between the Company and BlueCrest Venture Finance Master Fund
Limited, dated as of April 2, 2009 |
| 4.6(13) |
|
Security Agreement
(Intellectual Property), between the Company and BlueCrest Venture Finance
Master Fund Limited, dated as of April 2, 2009 |
| 4.7(13) |
|
Subordination Agreement, by Hunton
& Williams, LLP in favor of BlueCrest Venture Finance Master Fund
Limited, entered into and effective April 2, 2009 |
| 4.8(13) |
|
Amended and Restated
Promissory Note, dated April 2, 2009, by the Company to BlueCrest Venture
Finance Master Fund Limited |
| 4.9(13) |
|
Warrant to purchase 1,315,542
shares of the registrant’s common stock, dated April 2, 2009, issued to
BlueCrest Venture Finance Master Fund Limited |
| 4.10(14) |
|
Warrant to purchase 451,043
shares of the registrant’s common stock, dated April 2, 2009, issued to
Rogers Telecommunications Limited |
| 4.11(14) |
|
Warrant to purchase 173,638 shares
of the registrant’s common stock, dated April 2, 2009, issued to Hunton
& Williams, LLP |
| 4.12(4) |
|
Warrant to purchase shares
of the registrant's common stock issued to Howard J. Leonhardt and Brenda
Leonhardt |
| 4.12(19) |
|
10% Convertible Promissory Note
Due July 23, 2010, in the amount of $20,000, payable to Dana
Smith |
112
| 4.13(19) |
|
10% Convertible Promissory Note Due July 23,
2010, in the amount of $100,000, payable to Bruce Meyers |
| 4.14(19) |
|
Registration Rights Agreement, dated July 23,
2009 |
| 4.15(4) |
|
Warrant to purchase shares of the registrant's
common stock issued to the R&A Spencer Family Limited
Partnership |
| 4.15(19) |
|
Subordination Agreement, dated July 23,
2009 |
| 4.16(19) |
|
Note Purchase Agreement, dated July 23,
2009 |
| 4.17(19) |
|
Closing Confirmation of Conversion Election,
dated July 23, 2009 |
| 4.20(6) |
|
Warrant to purchase shares of the registrant's
common stock issued to Samuel S. Ahn, M.D. |
| 4.23(7) |
|
Warrant to purchase shares of the registrant's
common stock issued to Howard and Brenda Leonhardt |
| 4.27(11) |
|
Form of Warrant Agreement for October 2008
Private Placement |
| 4.30(19) |
|
10%
Convertible Promissory Note Due July 23, 2010, in the amount of $100,000,
payable to Bruce Meyers |
| 4.31(34) |
|
Series A Convertible Preferred
Stock |
| 4.32(35) |
|
Amendment to the Series A Convertible Preferred
Stock |
| 5.1 |
|
Consent of Sichenzia Ross Friedman Ference
LLP |
| 10.1**(1) |
|
1999
Officers and Employees Stock Option Plan |
| 10.2**(1) |
|
1999 Directors and Consultants Stock Option
Plan |
| 10.3(1) |
|
Form
of Option Agreement under 1999 Officers and Employees Stock Option
Plan |
| 10.4(3) |
|
Form of Option Agreement under 1999 Directors
and Consultants Stock Option Plan |
| 10.5**(4) |
|
Employment Letter Agreement between the
registrant and Scott Bromley, dated August 24, 2006. |
| 10.6(1) |
|
Lease Agreement between the registrant and
Sawgrass Business Plaza, LLC, as amended, dated November 14,
2006. |
| 10.7(1) |
|
Asset Purchase Agreement between the registrant
and Advanced Cardiovascular Systems, Inc., dated June 24,
2003. |
| 10.8(4) |
|
Conditionally Exclusive License Agreement
between the registrant, Dr. Peter Law and Cell Transplants International,
LLC, dated February 7, 2000, as amended. |
| 10.9(4) |
|
Loan
Guarantee, Payment and Security Agreement, dated as of June 1, 2007, by
and between the registrant, Howard J. Leonhardt and Brenda
Leonhardt |
| 10.10(4) |
|
Loan Guarantee, Payment and Security Agreement,
dated as of June 1, 2007, by and between the registrant and William P.
Murphy Jr., M.D. |
| 10.11(4) |
|
Loan
Agreement, dated as of June 1, 2007, by and between the registrant and
Bank of America, N.A. |
| 10.13(4) |
|
Warrant to purchase shares of the registrant's
common stock issued to Howard J. Leonhardt and Brenda
Leonhardt |
| 10.14(4) |
|
Warrant to purchase shares of the registrant's
common stock issued to William P. Murphy, Jr., M.D. |
| 10.16(4) |
|
Material Supply Agreement, dated May 10, 2007,
by and between the registrant and Biosense Webster |
| 10.17(5) |
|
Warrant to purchase shares of the registrant's
common stock issued to BlueCrest Capital Finance, L.P. |
| 10.18(6) |
|
Loan Guarantee, Payment and Security Agreement,
dated as of September 12, 2007, by and between the registrant and Samuel
S. Ahn, M.D. |
| 10.19(6) |
|
Loan
Guarantee, Payment and Security Agreement, dated as of September 12, 2007,
by and between the registrant and Dan Marino |
| 10.21(6) |
|
Loan Guarantee, Payment and Security Agreement,
dated as of September 19, 2007, by and between the registrant and Jason
Taylor |
| 10.22(7) |
|
Loan
Guarantee, Payment and Security Agreement, dated as of October 10, 2007,
by and between the registrant and Howard and Brenda
Leonhardt |
113
| 10.24(7) |
|
Second Amendment to Loan
Guarantee, Payment and Security Agreement, dated as of October 10, 2007,
by and between the registrant and Howard and Brenda Leonhardt |
| 10.25(7) |
|
Second Amendment to Loan Guarantee, Payment and
Security Agreement, dated as of October 10, 2007, by and between the
registrant and William P. Murphy, Jr., M.D. |
| 10.26**(10) |
|
Bioheart, Inc. Omnibus Equity
Compensation Plan |
| 10.28(11) |
|
Form of Registration Rights Agreement for
October 2008 Private Placement |
| 10.29(19) |
|
10% Convertible Promissory Note
Due July 23, 2010, in the amount of $20,000, payable to Dana
Smith |
| 10.31(19) |
|
Registration Rights Agreement, dated July 23,
2009 |
| 10.32(19) |
|
Subordination Agreement, dated
July 23, 2009 |
| 10.33(19) |
|
Note Purchase Agreement, dated July 23,
2009 |
| 10.34(19) |
|
Closing Confirmation of Conversion
Election, dated July 23, 2009 |
| 10.35**(20) |
|
Amended and Restated 1999 Directors and
Consultants Stock Option Plan |
| 10.36(21) |
|
Preliminary Commitment Letter with
Seaside National Bank and Trust, dated September 30, 2010. |
| 10.37(22) |
|
Loan Agreement with Seaside National Bank and
Trust, dated October 25, 2010. |
| 10.38(22) |
|
Promissory Note with Seaside
National Bank and Trust, dated October 25, 2010. |
| 10.39(22) |
|
Amended and Restated Loan and Security
Agreement with BlueCrest Venture Finance Master Fund Limited, dated
October 25, 2010. |
| 10.40(23) |
|
Form of Subscription Agreement,
executed November 30, 2010. |
| 10.41(23) |
|
Form of Common Stock Purchase Warrant, issued
November 30, 2010. |
| 10.42(23) |
|
Form of Registration Rights
Agreement, dated November 30, 2010. |
| 10.43(24) |
|
Unsecured Convertible Promissory Note for
$25,000, with Magna Group, LLC, dated January 3, 2011. |
| 10.44(24) |
|
Promissory Note for $139,728.82
with Magna Group, LLC, dated January 3, 2011. |
| 10.45(24) |
|
Securities Purchase Agreement with Magna Group,
LLC, dated January 3, 2011. |
| 10.46(24) |
|
Subordination Agreement, dated
January 3, 2011. |
| 10.47(24) |
|
Notice of Conversion Election, dated January 3,
2011. |
| 10.48(25) |
|
Unsecured Convertible Promissory
Note for $34,750, with Magna Group, LLC, dated May 16, 2011. |
| 10.49(25) |
|
Promissory Note for $139,728.82 with Magna
Group, LLC, dated May 16, 2011. |
| 10.50(25) |
|
Securities Purchase Agreement with
Magna Group, LLC, dated May 16, 2011. |
| 10.51(25) |
|
Subordination Agreement, dated May 16,
2011. |
| 10.52(26) |
|
Promissory Note for $139,728.82
with Lotus Funding Group, LLC, dated June 15, 2011. |
| 10.53(26) |
|
Partial Assignment and Modification Agreement,
dated June 15, 2011. |
| 10.54(26) |
|
Subordination Agreement, dated
June 15, 2011. |
| 10.55(27) |
|
Promissory Note for $140,380.21 with Greystone
Capital Partners, dated July 8, 2011. |
| 10.56(27) |
|
Partial Assignment and
Modification Agreement, dated July 8, 2011. |
| 10.57(28) |
|
Subordination Agreement, dated July 8,
2011. |
| 10.58(29) |
|
Promissory Note for $139,728.82
with Greystone Capital Partners, dated August 1, 2011. |
| 10.59(29) |
|
Partial Assignment and Modification Agreement,
dated August 1, 2011. |
| 10.60(29) |
|
Subordination Agreement, dated
August 1, 2011. |
| 10.61(30) |
|
Promissory Note for $139,728.82 with Greystone
Capital Partners, dated September 1, 2011. |
| 10.62(30) |
|
Partial Assignment and
Modification Agreement, dated September 1, 2011. |
| 10.63(30) |
|
Subordination Agreement, dated September 1,
2011. |
| 10.64(31) |
|
Standby Equity Distribution
Agreement dated as of November 2, 2011. |
| 10.65(31) |
|
Registration Rights Agreement dated as of
November 2, 2011. |
| 10.66(32) |
|
Promissory Note for $139,728.82
with Greystone Capital Partners, dated January 3, 2012 |
| 10.67(32) |
|
Term Note B Promissory Note for $139,728.82
with Greystone Capital Partners, dated January 3,
2012 |
114
| 10.68(32) |
|
Unsecured
Convertible Promissory Note for $63,000, with Asher Enterprises, Inc.
dated April 2, 2012 |
| 10.69(32) |
|
Unsecured Convertible
Promissory Note for $125,000, with IBC Funds LLC., dated January 9,
2013 |
| 10.70(32) |
|
Unsecured
Convertible Promissory Note for $37,500, with Asher Enterprises, Inc.
dated February 20, 2013 |
| 10.71(32) |
|
Unsecured Convertible
Promissory Note for $42,500, with Asher Enterprises, Inc. dated January 9,
2013 |
| 10.80(33) |
|
2013
Bioheart, Inc. Omnibus Equity Compensation Plan |
| 14.1(2) |
|
Code of Ethics for Chief
Executive Officer, Chief Financial Officer, Chief Accounting Officer and
persons performing similar functions |
| 14.2(2) |
|
Code of
Business Conduct and Ethics (FOOTNOTE] |
| 23.1 |
|
Consent of Fiondella,
Milone and LaSaracina LLP * |
| 23.2 |
|
Consent of
Sichenzia Ross Friedman Ference LLP (filed as part of Exhibit
5.1)* |
| |
|
|
| * |
|
Filed herewith |
| |
| (1) |
|
Incorporated by reference to the Company’s
Form S-1 filed with the Securities and Exchange Commission (the “SEC”) on
February 13, 2007. |
| (2) |
|
Incorporated by reference
to Amendment No. 1 to the Company’s Form S-1 filed with the SEC on June 5,
2007. |
| (3) |
|
Incorporated by reference to Amendment No. 2 to the Company’s Form
S-1 filed with the SEC on July 12, 2007. |
| (4) |
|
Incorporated by reference
to Amendment No. 3 to the Company’s Form S-1 filed with the SEC on August
9, 2007. |
| (5) |
|
Incorporated by reference to Amendment No. 4 to the Company’s Form
S-1 filed with the SEC on September 6, 2007. |
| (6) |
|
Incorporated by reference
to Amendment No. 5 to the Company’s Form S-1 filed with the SEC on October
1, 2007. |
| (7) |
|
Incorporated by reference to Post-effective Amendment No. 1 to the
Company’s Form S-1 filed with the SEC on October 11, 2007. |
| (8) |
|
Incorporated by reference
to the Company’s Current Report on Form 8-K filed with the SEC on July 3,
2008. |
| (9) |
|
Incorporated by reference to the Company’s Current Report on Form
8-K filed with the SEC on August 8, 2008. |
| (10) |
|
Incorporated by reference
to the Company’s Quarterly Report on Form 10-Q filed with the SEC on
August 14, 2008. |
| (11) |
|
Incorporated by reference to the Company’s Quarterly Report on Form
10-Q filed with the SEC on November 14, 2008. |
| (12) |
|
Incorporated by reference
to the Company’s Current Report on Form 8-K filed with the SEC on February
3, 2009. |
| (13) |
|
Incorporated by reference to the Company’s Current Report on Form
8-K filed with the SEC on April 8, 2009. |
| (14) |
|
Incorporated by reference
to the Company’s Annual Report on Form 10-K filed with the SEC on April
15, 2009. |
| (15) |
|
Incorporated by reference to the Company’s Annual Report on Form
10-K/A filed with the SEC on April 30, 2009. |
| (16) |
|
Incorporated by reference
to the Company’s Current Report on Form 8-K filed with the SEC on May 18,
2009. |
| (17) |
|
Incorporated by reference to the Company’s
Quarterly Report on Form 10-Q filed with the SEC on May 20,
2009. |
115
| (18) |
|
Incorporated by reference to the
Company’s Current Report on Form 8-K filed with the SEC on July 9,
2009. |
| (19) |
|
Incorporated by reference to the Company’s
Current Report on Form 8-K filed with the SEC on August 3,
2009. |
| (20) |
|
Incorporated by reference to Exhibit 4.6 to the
Company’s Post-Effective Amendment to Registration Statement on Form
S-8/A, filed with the SEC on June 2, 2010. |
| (21) |
|
Incorporated by reference to Exhibit 10.1 to
the Company’s Current Report on Form 8-K filed with the SEC on October 6,
2010. |
| (22) |
|
Incorporated by reference to the Company’s
Current Report on Form 8-K filed with the SEC on October 29,
2010. |
| (23) |
|
Incorporated by reference to the Company’s
Current Report on Form 8-K filed with the SEC on December 6,
2010. |
| (24) |
|
Incorporated by reference to the Company’s
Current Report on Form 8-K filed with the SEC on January 12,
2011. |
| (25) |
|
Incorporated by reference to the Company
Current Report on Form 8-K filed with the SEC on May 25, 2011 |
| (26) |
|
Incorporated by reference to the Company
Current Report on Form 8-K filed with the SEC on June 21,2011 |
| (27) |
|
Incorporated by reference to the Company’s
Quarterly Report on Form 10-Q filed with the Securities and Exchange
Commission on August 15. 2011 |
| (28) |
|
Incorporated by reference to the Company’s
Quarterly Report on Form 10-Q filed with the Securities and Exchange
Commission on November 14, 2011 |
| (29) |
|
Incorporated by reference to the Company
Current Report on Form 8-K filed with the SEC on January 13,
2012 |
| (30) |
|
Incorporated by reference to the Company
Current Report on Form 8-K filed with the SEC on January 30,
2012 |
| (31) |
|
Incorporated by reference to the Company
Registration Statement on Form S-1/A filed with the SEC on February 8,
2012 |
| (32) |
|
Incorporated by reference to the Company Annual
Report on Form 10-K filed with the SEC on March 29, 2013 |
| (33) |
|
Incorporated by reference to the Company
Quarterly Report on Form 10-Q filed with the SEC on May 9,
2013 |
| (34) |
|
Incorporated by reference to the Company
Current Report on Form Pre-14C filed with the SEC on December 18,
2012 |
| (35) |
|
Incorporated by reference to the Company
Current Report on Form 8-K filed with the SEC on December 31,
2013 |
| (36) |
|
Incorporated by reference to the Company
Information Statement on Form 14C filed with the SEC on October 10,
2014. |
| (37) |
|
Incorporated by reference to the Company
Current Report on Form 8-K filed with the SEC on October 24,
2014 |
Item 17.
Undertakings
The undersigned Registrant
hereby undertakes to:
| (1) |
|
File, during any period in which offers or sales are being made, a
post-effective amendment to this Registration Statement to: |
| |
|
|
(i) |
|
Include
any prospectus required by Section 10(a)(3) of the Securities
Act; |
| |
|
|
(ii) |
|
Reflect in
the prospectus any facts or events which, individually or together,
represent a fundamental change in the information in the Registration
Statement. Notwithstanding the foregoing, any increase or decrease in
volume of securities offered (if the total dollar value of the securities
offered would not exceed that which was registered) and any deviation from
the low or high end of the estimated maximum offering range may be
reflected in the form of prospectus filed with the Commission pursuant to
Rule 424(b) under the Securities Act if, in the aggregate, the changes in
volume and price represent no more than a 20% change in the maximum
aggregate offering price set forth in the “Calculation of Registration
Fee” table in the effective Registration Statement;
and |
116
|
|
(iii) |
|
Include
any additional or changed material information on the plan of
distribution. |
| |
| (2) |
|
For determining liability under the Securities Act, treat each
post-effective amendment as a new registration statement of the securities
offered, and the offering of the securities at that time to be the initial
bona fide offering. |
| |
| (3) |
|
File a post-effective amendment to remove from registration any of
the securities that remain unsold at the end of the offering. |
| |
| (4) |
|
For purposes of determining any liability under the Securities Act,
treat the information omitted from the form of prospectus filed as part of
this Registration Statement in reliance upon Rule 430A and contained in a
form of prospectus filed by the Registrant pursuant to Rule 424(b)(1) or
(4) or 497(h) under the Securities Act as part of this Registration
Statement as of the time it was declared effective. |
| |
| (5) |
|
For the purpose of determining liability of the Registrant under
the Securities Act to any purchaser in the initial distribution of the
securities: The undersigned Registrant undertakes that in a primary
offering of securities of the undersigned Registrant pursuant to this
Registration Statement, regardless of the underwriting method used to sell
the securities to the purchaser, if the securities are offered or sold to
such purchaser by means of any of the following communications, the
undersigned Registrant will be a seller to the purchaser and will be
considered to offer or sell such securities to such
purchaser: |
| |
|
|
(i) |
|
Any
preliminary prospectus or prospectus of the undersigned Registrant
relating to the offering required to be filed pursuant to the Rule
424; |
| |
|
|
(ii) |
|
Any free
writing prospectus relating to the offering prepared by or on behalf of
the undersigned Registrant or used or referred to by the undersigned
Registrant; |
| |
|
|
(iii) |
|
The
portion of any other free writing prospectus relating to the offering
containing material information about the undersigned Registrant or its
securities provided by or on behalf of the undersigned Registrant;
and |
| |
|
|
(iv) |
|
Any other
communication that is an offer in the offering made by the undersigned
Registrant to the purchaser. |
| |
| (6) |
|
For determining any liability under the Securities Act, treat each
post-effective amendment that contains a form of prospectus as a new
registration statement for the securities offered in the Registration
Statement, and that offering of the securities at that time as the initial
bona fide offering of those securities. |
| |
| (7) |
|
Insofar as indemnification for liabilities arising under the
Securities Act may be permitted to directors, officers and controlling
persons of the Registrant pursuant to the foregoing provisions, or
otherwise, the Registrant has been advised that in the opinion of the
Securities and Exchange Commission such indemnification is against public
policy as expressed in the Securities Act and is, therefore,
unenforceable. In the event that a claim for indemnification against such
liabilities (other than the payment by the Registrant of expenses incurred
or paid by a director, officer or controlling person of the Registrant in
the successful defense of any action, suit or proceeding) is asserted by
such director, officer or controlling person in connection with the
securities being registered, the Registrant will, unless in the opinion of
its counsel the matter has been settled by controlling precedent, submit
to a court of appropriate jurisdiction the question whether such
indemnification by it is against public policy as expressed in the
Securities Act and will be governed by the final adjudication of such
issue. |
| |
| (8) |
|
Each prospectus filed pursuant to Rule 424(b) as part of a
registration statement relating to an offering shall be deemed to be part
of and included in the Registration Statement as of the date it is first
used after effectiveness. |
| |
|
|
Provided, however, that no statement made in a registration
statement or prospectus that is part of the Registration Statement or made
in a document incorporated or deemed incorporated by reference into the
Registration Statement or prospectus that is part of the Registration
Statement will, as to a purchaser with a time of contract of sale prior to
such first use, supersede or modify any statement that was made in the
Registration Statement or prospectus that was part of the registration
statement or made in any document immediately prior to such date of first
use. |
117
SIGNATURES
Pursuant to the
requirements of the Securities Act, the Registrant has duly caused this
Registration Statement to be signed on its behalf by the undersigned, thereunto
duly authorized on November 21, 2014.
|
BIOHEART, INC. |
| Date: November 21, 2014 |
|
|
By: |
/s/ Mike Tomas |
|
|
Mike
Tomas |
|
Chief Executive Officer & President: |
| |
| |
| |
| |
| Date: November 21, 2014 |
By:
|
/s/ Mike Tomas |
|
|
Mike
Tomas |
|
Chief Financial Officer (Principal Accounting |
|
Officer) |
POWER OF
ATTORNEY
Each person whose signature
appears below constitutes and appoints Mike Tomas his true and lawful
attorney-in-fact and agent, acting alone, with full powers of substitution and
re-substitution, for him and in his name, place and stead, in any and all
capacities, to sign any and all amendments (including post-effective amendments)
to this Registration Statement, any Amendments thereto and any Registration
Statement of the same offering which is effective upon filing pursuant to Rule
462(b) under the Securities Act, and to file the same, with all exhibits
thereto, and other documents in connection therewith, with the Commission,
granting unto said attorney-in-fact and agent, acting alone, full powers and
authority to do and perform each and every act and thing requisite and necessary
to be done in and about the premises, as fully to all intents and purposes as he
might or could do in person, hereby ratifying and confirming said
attorney-in-fact and agent, acting alone, or his substitute or substitutes, may
lawfully do or cause to be done by virtue hereof.
In accordance with the
requirements of the Securities Act, this Registration Statement has been signed
below by the following persons on behalf of the Company in the capacities and on
the dates indicated.
| Signature |
|
Title |
|
Date |
| /s/ William P.
Murphy, Jr., M.D. |
|
Chairman of the Board |
|
November 21, 2014 |
| William P. Murphy, Jr., M.D. |
|
|
|
|
| |
|
|
|
|
| /s/ Mike Tomas |
|
Chief Executive Officer & Director |
|
November 21, 2014 |
| Mike
Tomas |
|
|
|
|
| |
|
|
|
|
| /s/ Mark P. Borman |
|
Director |
|
November 21, 2014 |
| Mark
Borman |
|
|
|
|
|
|
|
|
|
| /s/ Kristin Comella |
|
Director |
|
November 21, 2014 |
| Kristin Comella |
|
|
|
|
|
|
|
|
|
|
| /s/ Sheldon Anderson |
|
Director |
|
November 21, 2014 |
| Sheldon Anderson |
|
|
|
|
| |
|
|
|
|
| /s/ Charles A. Hart |
|
Director |
|
November 21, 2014 |
| Charles A. Hart |
|
|
|
|
| |
|
|
|
|
| /s/ Samuel S. Ahn, MD, MBA |
|
Director |
|
November 21, 2014 |
| Samuel S. Ahn, MD, MBA |
|
|
|
|
118
EXHIBIT 5.1
SICHENZIA ROSS FRIEDMAN
FERENCE LLP
61 Broadway,
32nd Floor
New York, NY 10006
Telephone: (212)
930-9700
Facsimile: (212) 930-9725
November 21, 2014
VIA ELECTRONIC
TRANSMISSION
Securities and Exchange
Commission
100 F Street, N.E.
Washington, DC 20549
| RE: |
Bioheart,
Inc. |
|
Registration Statement on Form
S-1 |
Ladies and
Gentlemen:
We refer to the
above-captioned registration statement on Form S-1 (the “Registration Statement”) under the Securities Act of 1933, as amended
(the “Act”), filed by Bioheart, Inc., a Florida corporation
(the “Company”), with the Securities and Exchange Commission on
this November 21, 2014.
We have examined the
originals, photocopies, certified copies or other evidence of such records of
the Company, certificates of officers of the Company and public officials, and
other documents as we have deemed relevant and necessary as a basis for the
opinion hereinafter expressed. In such examination, we have assumed the
genuineness of all signatures, the authenticity of all documents submitted to us
as certified copies or photocopies and the authenticity of the originals of such
latter documents.
Based on our examination
mentioned above, we are of the opinion that the 9,109,128 shares of common stock
being offered pursuant to the Registration Statement are duly authorized,
legally and validly issued, fully paid and non-assessable and the 134,703,463
shares of common stock, issuable upon the occurrence of certain events as set
forth in the Registration Statements and being offered pursuant to the
Registration Statement will, when issued in the manner described in the
Registration Statement, be duly authorized, legally and validly issued, fully
paid and non-assessable.
We hereby consent to the
filing of this opinion as Exhibit 5.1 to the Registration Statement and to the
reference to our firm under “Legal Matters” in the related Prospectus. In giving
the foregoing consent, we do not hereby admit that we are in the category of
persons whose consent is required under Section 7 of the Act, or the rules and
regulations of the Securities and Exchange Commission.
| Very
truly yours, |
| |
| /s/ Sichenzia Ross Friedman Ference LLP |
| |
| Sichenzia Ross Friedman Ference
LLP |
EXHIBIT
23.1
CONSENT OF INDEPENDENT
REGISTERED PUBLIC ACCOUNTING FIRM
We hereby consent to the
use in this Registration Statement of Bioheart, Inc. on Form S-1 of our report
dated March 24, 2014 appearing in the Prospectus which is part of this
Registration Statement. We also consent to the reference to our firm under the
heading "Experts" in such Prospectus.
/s/ Fiondella Milone and LaSaracina LLP
Glastonbury, Connecticut
November 21, 2014
US Stem Cell (CE) (USOTC:USRM)
Historical Stock Chart
From Mar 2024 to Apr 2024
US Stem Cell (CE) (USOTC:USRM)
Historical Stock Chart
From Apr 2023 to Apr 2024