Vigil Neuroscience Launches ALSPAware to Provide Access to No-cost Genetic Testing and Counseling for Adult-Onset Leukoencephalopathy with Axonal Spheroids and Pigmented Glia (ALSP)
May 08 2023 - 7:08AM

Vigil Neuroscience, Inc. (Nasdaq: VIGL), a clinical-stage
biotechnology company committed to harnessing the power of
microglia for the treatment of neurodegenerative diseases, today
announced the launch of ALSPAware, a new program providing no-cost
genetic testing and counseling services for the diagnosis of
adult-onset leukoencephalopathy with axonal spheroids and pigmented
glia (ALSP). The program is coordinated by InformedDNA®, the
largest independent provider of genetic counseling services, and
PreventionGenetics, an Exact Sciences Company running an accredited
laboratory that delivers high-quality genetic testing.
"Limited access to genetic testing and appropriate counseling
has led to diagnostic challenges affecting not only those living
with ALSP, but also the healthcare providers hoping to treat the
disease. Only about one-third of people living with ALSP receive a
correct diagnosis when symptoms first appear, resulting in a delay
that can significantly increase the burden of this devastating
genetic condition,” said Ivana Magovčević-Liebisch, Ph.D.,
J.D., President and Chief Executive Officer of Vigil. “We believe
that everyone deserves access to the services necessary to make the
diagnostic journey as efficient and convenient as possible, and we
are proud to be taking this important step forward in supporting
the ALSP community.”
ALSPAware offers no-cost, confidential genetic testing and
counseling services to U.S. residents age 18 and over. Developed
with both patients and healthcare providers in mind, the program
includes a single gene confirmatory test for individuals with a
family history of ALSP, as well as a custom gene panel available
for physicians to use in diagnosing adult-onset neurological
diseases. Trained genetic counselors are available to facilitate
testing and discuss results, and participants will have access to a
range of specialized information and services created to support
participants and their families. By increasing access to genetic
testing for both individuals and healthcare providers, we believe
ALSPAware has the potential to help reduce initial misdiagnosis of
this disease, while also providing appropriate disease management
services for those living with ALSP.
Increased access to genetic testing has been an important goal
in the ALSP community for many years. ALSPAware was developed with
input from leaders from the patient advocacy community including
Sisters’ Hope Foundation, United Leukodystrophy Foundation and
Hunter’s Hope Foundation, to ensure the program addresses the key
challenges patients, caregivers and healthcare providers face
during the genetic testing and counseling process.
To learn more about ALSPAware or find out how to access genetic
testing services through the program, visit
www.informedDNA.COM/ALSPAware.
About ALSP
ALSP is a rare, inherited, autosomal dominant neurological
disease with high penetrance. It is caused by a mutation to the
CSF1R gene and affects an estimated 10,000 people in the U.S., with
similar prevalence in Europe and Japan. The disease generally
presents itself in adults in their forties, is diagnosed through
genetic testing and established clinical/radiologic criteria and is
characterized by cognitive dysfunction, neuropsychiatric symptoms,
and motor impairment. These symptoms typically exhibit rapid
progression with a life expectancy of approximately six to seven
years on average after diagnosis, causing significant patient and
caregiver burden. There are currently no approved therapies for the
treatment of ALSP, underlining the high unmet need in this rare
indication.
About Vigil Neuroscience
Vigil Neuroscience is a clinical-stage biotechnology company
focused on developing treatments for both rare and common
neurodegenerative diseases by restoring the vigilance of microglia,
the sentinel immune cells of the brain. We are utilizing the tools
of modern neuroscience drug development across multiple therapeutic
modalities in our efforts to develop precision-based therapies to
improve the lives of patients and their families. VGL101, our lead
candidate, is a fully human monoclonal antibody agonist targeting
human triggering receptor expressed on myeloid 2 (TREM2) and is in
a Phase 2 proof-of-concept trial in patients with adult-onset
leukoencephalopathy with axonal spheroids and pigmented glia
(ALSP), a rare and fatal neurodegenerative disease. Vigil is also
conducting Investigational New Drug Application (IND)-enabling
studies with a novel small molecule TREM2 agonist program to treat
common neurodegenerative diseases associated with microglial
dysfunction, with an initial focus on Alzheimer’s disease (AD) in
genetically defined subpopulations. For more information visit
www.vigilneuro.com.
About PreventionGeneticsFounded in 2004 and
located in Marshfield, Wisconsin, PreventionGenetics, a wholly
owned subsidiary of Exact Sciences Corp., is a Clinical Laboratory
Improvement Amendments (CLIA) and International Organization for
Standardization (ISO) 15189:2012 accredited laboratory.
PreventionGenetics delivers clinical genetic testing of the highest
quality at fair prices with exemplary service to people around the
world. PreventionGenetics provides tests for nearly all clinically
relevant genes including the powerful and comprehensive whole
genome sequencing test, PGnome® and whole exome
sequencing test, PGxome®.
About InformedDNA
InformedDNA® is the country’s leading applied genomics solutions
company, helping people harness the full power of the genomics
revolution. With a large staff of board-certified genetics
specialists, InformedDNA ensures that health organizations have
access to the highest quality genomics insights to optimize
clinical decisions and biopharma companies have support to meet the
challenges of precision medicine clinical trials. InformedDNA's
solutions extend beyond successful clinical trial execution to
maximize post-market success in targeted populations.
www.InformedDNA.com.
Vigil Neuroscience Forward-Looking
Statements
This press release includes certain disclosures that contain
“forward-looking statements” of Vigil Neuroscience’s (“Vigil” or
the “Company”) that are made pursuant to the safe harbor provisions
of the federal securities laws, including, without limitation,
express or implied statements regarding: the Company’s strategy,
business plans and focus; the potential design, access, benefits
and success of ALSPAware, including the belief that the program
will ease the diagnostic journey for patients and their families;
the progress and timing of the preclinical and clinical development
of Vigil’s programs, including the availability of data and
expected timing for reporting data from the VGL101 Phase 2
proof-of-concept trial and the filing of an IND application for its
small molecule TREM2 agonist program; and expectations regarding
the development of VGL101 in ALSP and other indications.
Forward-looking statements are based on Vigil’s current
expectations and are subject to inherent uncertainties, risks and
assumptions that are difficult to predict. Factors that could cause
actual results to differ include, but are not limited to, risks and
uncertainties related to uncertainties inherent in the
identification and development of product candidates, including the
conduct of research activities and the initiation and completion of
preclinical studies and clinical trials; uncertainties as to the
availability and timing of results and data from preclinical and
clinical studies; the timing of the Company’s ability to submit and
obtain regulatory clearance for investigational new drug
applications and initiate additional clinical trials; whether
results from preclinical studies will be predictive of the results
of later preclinical studies and clinical trials; the Company’s
ability to initiate and complete its current and expected clinical
trials; whether Vigil’s cash resources will be sufficient to fund
its foreseeable and unforeseeable operating expenses and capital
expenditure requirements; uncertainties associated with the impact
of the COVID-19 pandemic on its business and operations; as well as
the risks and uncertainties identified in the Company’s filings
with the Securities and Exchange Commission (SEC), including
Vigil’s Annual Report on Form 10-K for the year ended December 31,
2022 and in any subsequent filings it may make with the SEC.
Forward-looking statements contained in this announcement are made
as of this date, and Vigil undertakes no duty to update such
information except as required under applicable law. Readers should
not rely upon the information on this page as current or accurate
after its publication date.
Investor Contact:
Sarah Carmody
scarmody@vigilneuro.com
Media Contact:
Megan McGrath
MacDougall Advisors
mmcgrath@macdougall.bio
A photo accompanying this announcement is available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/0217bf0c-f8f0-4cef-aa08-b16c9076faef
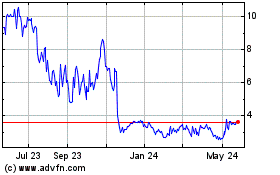
Vigil Neuroscience (NASDAQ:VIGL)
Historical Stock Chart
From Apr 2024 to May 2024
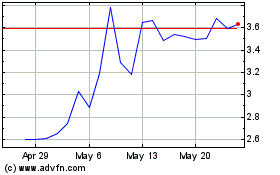
Vigil Neuroscience (NASDAQ:VIGL)
Historical Stock Chart
From May 2023 to May 2024