Caribou Biosciences Announces Appointment of Sundar Jagannath, MD, to its Scientific Advisory Board
November 06 2023 - 8:00AM

Caribou Biosciences, Inc. (Nasdaq: CRBU), a leading
clinical-stage CRISPR genome-editing biopharmaceutical company,
today announced the appointment of Sundar Jagannath, MD, to its
scientific advisory board. With four decades of experience, Dr.
Jagannath is a renowned expert in bone marrow transplantation and
treating patients with multiple myeloma, and he is the director of
the Center of Excellence for Multiple Myeloma at The Tisch Cancer
Institute and professor of medicine, hematology and medical
oncology, at Mount Sinai School of Medicine, New York.
“We are honored to have Dr. Jagannath join our scientific
advisory board. Dr. Jagannath is a distinguished oncologist who is
renowned for his groundbreaking contributions to the field of
multiple myeloma,” said Syed Rizvi, MD, Caribou’s chief medical
officer. “Dr. Jagannath has a stellar reputation as a compassionate
physician and researcher, whose guidance will be invaluable to
Caribou as we advance the CaMMouflage Phase 1 trial for CB-011, our
allogeneic anti-BCMA CAR-T cell therapy, for patients with relapsed
or refractory multiple myeloma.”
With a medical career spanning over 40 years, Dr. Jagannath is a
distinguished leader in multiple myeloma and bone marrow
transplantation. His clinical and translational research has
resulted in more than 180 peer reviewed articles, 150 abstracts,
and 30 book chapters. After earning his medical degree and
training, Dr. Jagannath completed a medical oncology fellowship at
The University of Texas MD Anderson Cancer Center in Houston,
Texas. Prior to Mount Sinai, New York, he also served at University
of Arkansas and Saint Vincent’s Comprehensive Cancer Center, New
York, in leadership roles in the myeloma programs.
“Caribou’s differentiated approach in using chRDNA
genome-editing technology to armor CAR-T cell therapies for
improved antitumor activity is very promising,” said Dr. Jagannath.
“I look forward to advising on the development of CB-011 as
off-the-shelf and readily available treatment in multiple myeloma.
We urgently need such treatment options for patients living with
relapsed or refractory multiple myeloma.”
About CB-011 CB-011 is a product candidate from
Caribou’s allogeneic CAR-T cell therapy platform and is being
evaluated in patients with relapsed or refractory multiple myeloma
(r/r MM) in the CaMMouflage Phase 1 trial. CB-011 is an allogeneic
anti-BCMA CAR-T cell therapy engineered using Cas12a chRDNA
genome-editing technology. To Caribou’s knowledge, CB-011 is the
first allogeneic CAR-T cell therapy in the clinic that is
engineered to improve antitumor activity through an immune cloaking
strategy with a B2M knockout and insertion of a B2M–HLA-E fusion
protein to blunt immune-mediated rejection. CB-011 has been granted
Fast Track designation by the FDA. Additional information on the
CaMMouflage trial (NCT05722418) can be found at
clinicaltrials.gov.
About Caribou’s novel next-generation CRISPR
platformCRISPR genome editing uses easily designed,
modular biological tools to make DNA changes in living cells. There
are two basic components of Class 2 CRISPR systems: the nuclease
protein that cuts DNA and the RNA molecule(s) that guide the
nuclease to generate a site-specific, double-stranded break,
leading to an edit at the targeted genomic site. CRISPR systems are
capable of editing unintended genomic sites, known as off-target
editing, which may lead to harmful effects on cellular function and
phenotype. In response to this challenge, Caribou has developed
CRISPR hybrid RNA-DNA guides (chRDNAs; pronounced “chardonnays”)
that direct substantially more precise genome editing compared to
all-RNA guides. Caribou is deploying the power of its Cas12a chRDNA
technology to carry out high efficiency multiple edits, including
multiplex gene insertions, to develop CRISPR-edited therapies.
About Caribou Biosciences, Inc.Caribou
Biosciences is a clinical-stage CRISPR genome-editing
biopharmaceutical company dedicated to developing transformative
therapies for patients with devastating diseases. The company’s
genome-editing platform, including its Cas12a chRDNA technology,
enables superior precision to develop cell therapies that are
armored to potentially improve antitumor activity. Caribou is
advancing a pipeline of off-the-shelf cell therapies from its CAR-T
and CAR-NK platforms as readily available treatments for patients
with hematologic malignancies and solid tumors. Follow us
@CaribouBio and visit www.cariboubio.com.
Forward-looking statements This press release
contains forward-looking statements within the meaning of the
Private Securities Litigation Reform Act of 1995. These
forward-looking statements include, without limitation, statements
related to Caribou’s strategy, plans, and objectives, and
expectations regarding its clinical and preclinical development
programs. Management believes that these forward-looking statements
are reasonable as and when made. However, such forward-looking
statements are subject to risks and uncertainties, and actual
results may differ materially from any future results expressed or
implied by the forward-looking statements. Risks and uncertainties
include, without limitation, risks inherent in the development of
cell therapy products; uncertainties related to the initiation,
cost, timing, progress, and results of Caribou’s current and future
research and development programs, preclinical studies, and
clinical trials; and the risk that initial, preliminary, or interim
clinical trial data will not ultimately be predictive of the safety
and efficacy of Caribou’s product candidates or that clinical
outcomes may differ as patient enrollment continues and as more
patient data becomes available; the risk that preclinical study
results observed will not be borne out in human patients or
different conclusions or considerations are reached once additional
data have been received and fully evaluated; as well as other risk
factors described from time to time in Caribou’s filings with the
Securities and Exchange Commission, including its Annual Report on
Form 10-K for the year ended December 31, 2022 and subsequent
filings. In light of the significant uncertainties in these
forward-looking statements, you should not rely upon
forward-looking statements as predictions of future events. Except
as required by law, Caribou undertakes no obligation to update
publicly any forward-looking statements for any reason.
Caribou Biosciences, Inc.
contacts:Investors:Amy Figueroa,
CFAinvestor.relations@cariboubio.com
Media:Peggy Vorwald,
PhDmedia@cariboubio.com
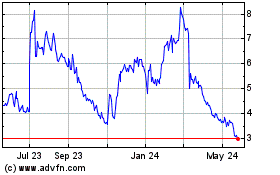
Caribou Biosciences (NASDAQ:CRBU)
Historical Stock Chart
From Apr 2024 to May 2024
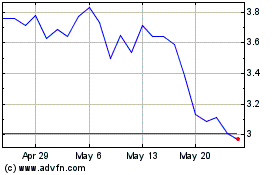
Caribou Biosciences (NASDAQ:CRBU)
Historical Stock Chart
From May 2023 to May 2024