Amylyx Pharmaceuticals Appoints Bernhardt Zeiher, MD, FCCP, FACP, to its Board of Directors
March 14 2024 - 9:00AM
Business Wire
- Dr. Zeiher brings more than 20 years of drug
development experience in areas with serious unmet treatment needs
at companies including Astellas Pharma, Pfizer, Eli Lilly and
Company, and Merck
Amylyx Pharmaceuticals, Inc. (NASDAQ: AMLX) (“Amylyx” or the
“Company”) today announced the addition of Bernhardt G. Zeiher, MD,
FCCP, FACP, to its Board of Directors (“Board”). Dr. Zeiher brings
more than 20 years of drug development experience where, in his
various roles, he oversaw the approval of 15 new treatments that
addressed people’s unmet needs in serious diseases with few to no
treatment options. Most recently, he served as Chief Medical
Officer (“CMO”) of Astellas Pharma, where he led early- and
late-stage drug development, medical and regulatory affairs,
pharmacovigilance, and quality assurance.
“This is a pivotal time for Amylyx, and we are thrilled to
welcome Dr. Zeiher to our Board to share his perspectives and
expertise as the company continues to build a pipeline supporting
our collective mission to one day end the suffering of
neurodegenerative diseases,” said George M. Milne, Jr., PhD,
Chairman of Amylyx’ Board. “Dr. Zeiher's insights of the
intricacies and complexities of drug development and innovation in
diseases with clear unmet need will be valuable as we continue to
serve neurodegenerative disease communities impacted by these
devastating diseases.”
Dr. Zeiher is a recognized industry leader who spent more than
10 years at Astellas Pharma, holding multiple roles of increasing
responsibility in the drug development division, leading up to his
role as CMO from 2018 to 2022. Prior to his time at Astellas, Dr.
Zeiher held various roles leading drug development at other
pharmaceutical companies including Pfizer, Eli Lilly and Company,
and Merck. Dr. Zeiher also practiced medicine at a tertiary medical
center in Indianapolis and has held a range of board appointments
since 2014, including positions at PhRMA Biomedical Advisory
Committee, TransCelerate Biopharma, Biotechnology Innovation
Organization, Astellas Global Health Foundation, and most recently,
Entrada Therapeutics, which he joined in 2023.
“We are seeing a transformation in neurodegenerative diseases,
both in potential new treatments and what we’re learning through
research, and I believe Amylyx is a catalyst in that change,” said
Dr. Zeiher. “The company’s community-centric values, steadfast
commitment to following the science, and beyond-therapy approaches
and support for the neurodegenerative disease community are a few
examples of why I am excited for the future and what we can
accomplish to support those living with these devastating diseases
and their loved ones.”
“Dr. Zeiher brings to the Board his distinguished drug
development background in overseeing the approval of 15 medicines,
and his first-hand experience as a physician. On behalf of the
entire Amylyx team, we are delighted to welcome him onboard as we
continue to advance our mission to one day end the suffering caused
by neurodegenerative diseases,” said Joshua Cohen and Justin Klee,
Co-CEOs of Amylyx. “We have several important milestones that we’re
working toward this year where Dr. Zeiher’s expertise will be key,
including our goal to bring our antisense oligonucleotide AMX0114,
developed in-house, to the clinic for ALS, continuing to research
the potential of AMX0035 in diseases like Wolfram syndrome and
progressive supranuclear palsy, and sharing additional insights,
learnings, and next steps from our Phase 3 PHOENIX trial in
ALS.”
Dr. Zeiher received a B.S. in biology from the University of
Toledo and an MD from Case Western Reserve University School of
Medicine. He completed his internal medicine residency and chief
residency at University Hospitals of Cleveland and then finished
his physician training as a Pulmonary and Critical Care Fellow at
University of Iowa Hospitals and Clinics.
About Amylyx Pharmaceuticals Amylyx Pharmaceuticals, Inc.
is committed to supporting and creating more moments for the
neurodegenerative disease community through the discovery and
development of innovative new treatments. Amylyx is headquartered
in Cambridge, Massachusetts and has operations in Canada, EMEA, and
Japan. For more information, visit amylyx.com and follow us on
LinkedIn and X, formerly known as Twitter. For investors, please
visit investors.amylyx.com.
Forward-Looking Statements Statements contained in this
press release and related comments in our earnings conference call
regarding matters that are not historical facts are
“forward-looking statements” within the meaning of the Private
Securities Litigation Reform Act of 1995, as amended. Because such
statements are subject to risks and uncertainties, actual results
may differ materially from those expressed or implied by such
forward-looking statements. Such statements include, but are not
limited to, the Company’s expectations of contributions from its
board of directors; the Company’s beliefs regarding the benefits of
AMX0035 in neurodegenerative diseases; expectations regarding the
continued research of AMX0035 in progressive supranuclear palsy and
Wolfram syndrome; the Company’s potential announcement of
additional insights, learnings, and next steps from the Phase 3
PHOENIX trial; the Company’s expectations with respect to its
progress through IND-enabling studies of AMX0114; the Company’s
expectations with respect to its early-stage ALS diagnostic test;
and expectations regarding the Company’s longer-term strategy. Any
forward-looking statements in this press release are based on
management’s current expectations of future events and are subject
to a number of risks and uncertainties that could cause actual
results to differ materially and adversely from those set forth in
or implied by such forward-looking statements. Risks that
contribute to the uncertain nature of the forward-looking
statements include the risks and uncertainties set forth in Amylyx’
filings with the Securities and Exchange Commission (“SEC”),
including its Annual Report on Form 10-K for the year ended
December 31, 2023, and subsequent filings with the SEC. All
forward-looking statements contained in this press release speak
only as of the date on which they were made. Subject to any
obligations under applicable law, Amylyx undertakes no obligation
to update such statements to reflect events that occur or
circumstances that exist after the date on which they were
made.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20240314599408/en/
Media Amylyx Media Team +1 (857) 799-7274
amylyxmediateam@amylyx.com
Investors Lindsey Allen Amylyx Pharmaceuticals, Inc. +1
(857) 320-6244 Investors@amylyx.com
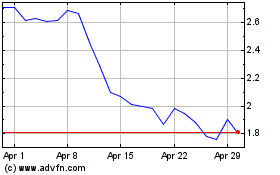
Amylyx Pharmaceuticals (NASDAQ:AMLX)
Historical Stock Chart
From Jul 2024 to Aug 2024
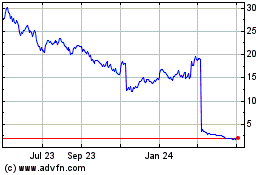
Amylyx Pharmaceuticals (NASDAQ:AMLX)
Historical Stock Chart
From Aug 2023 to Aug 2024