Emergent BioSolutions Announces Topline Data from NIAID Phase 3 ITAC Trial (INSIGHT-013) Evaluating Immunoglobulins as a Trea...
April 02 2021 - 8:00AM

Emergent BioSolutions Inc. (NYSE:EBS) today provided an update on
the evaluation of its investigational SARS-CoV-2 Immune Globulin
Intravenous (Human) (COVID-HIG) for the treatment of hospitalized
patients with COVID-19. The Phase 3 Inpatient Treatment with
Anti-Coronavirus Immunoglobulin (ITAC) trial, also known as
INSIGHT-013, sponsored and supported by the National Institute of
Allergy and Infectious Diseases (NIAID), part of the National
Institutes of Health (NIH), assessed the safety and efficacy of
four immunoglobulin candidates plus standard of care versus placebo
plus standard of care in hospitalized patients with COVID-19.
Topline data from the ITAC trial demonstrated that the addition of
anti-SARS-CoV-2 hyperimmunoglobulin to standard of care, inclusive
of remdesivir, for hospitalized adult COVID-19 patients with
symptoms for less than 12 days did not provide clinical benefit
when compared to standard of care plus placebo. There were no
serious safety concerns identified.
“While we are disappointed by these data in hospitalized
patients where there remains a high unmet need, we recognize that,
similar to other antibody-based therapies, intervention with
COVID-HIG earlier in the disease course may be necessary to impact
COVID-19 in patients,” said Dr. Laura Saward, SVP and therapeutics
business unit head at Emergent BioSolutions. “Emergent will
continue to explore COVID-HIG as a treatment in ongoing clinical
trials. The public-private partnership with NIAID and the
Biomedical Advanced Research and Development Authority (BARDA) has
been important to generate data on the COVID-HIG investigational
product. We also would like to thank all study participants, sites,
and investigators across the globe who contributed to this
effort.”
COVID-HIG is also being developed as a potential treatment for
outpatients at high risk of progression to severe disease with
funding from BARDA. It is also being supported with funding from
the U.S. Department of Defense.
This project has been funded in whole or in part with Federal
funds from the Department of Health and Human Services, Office of
the Assistant Secretary for Preparedness and Response, Biomedical
Advanced Research and Development Authority under contract
HHSO100201200004I, task order 75A50120F33006.
About the ITAC Trial The Phase 3
Inpatient Treatment with Anti-Coronavirus Immunoglobulin (ITAC)
clinical trial was a global, multi-center, double-blind,
placebo-controlled, randomized trial sponsored and funded by the
National Institute of Allergy and Infectious Diseases (NIAID), part
of the National Institutes of Health (NIH).
It was designed to test the safety, tolerability and
efficacy of a combination treatment regimen for coronavirus disease
2019 (COVID-19) consisting of the
antiviral remdesivir along with an anti-coronavirus
hyperimmune intravenous immunoglobulin (H-Ig), which contains a
highly concentrated solution of antibodies that neutralize
SARS-CoV-2. The antibodies in the H-Ig come from the liquid portion
of blood, or plasma, donated by healthy people who have recovered
from COVID-19.
Through the NIAID-funded INSIGHT network, the study team
enrolled nearly 600 adult patients at 63 sites in the
United States and 10 other countries on five continents. Volunteers
were eligible for the trial if they had been hospitalized for
COVID-19 and had symptoms for 12 days or fewer without
life-threatening organ dysfunction or end-organ failure. Four
companies provided investigational H-Ig materials for the trial,
including CSL Behring and Takeda on behalf of the CoVIg-19 Plasma
Alliance, as well as Emergent BioSolutions and
Grifols. Further information about the ITAC trial is available
at ClinicalTrials.gov under study
identifier NCT04546581.
About Emergent BioSolutionsEmergent
BioSolutions is a global life sciences company whose mission is to
protect and enhance life. Through our specialty products and
contract development and manufacturing services, we are dedicated
to providing solutions that address public health threats. Through
social responsibility, we aim to build healthier and safer
communities. We aspire to deliver peace of mind to our patients and
customers so they can focus on what’s most important in their
lives. In working together, we envision protecting or enhancing 1
billion lives by 2030. For more information visit
www.emergentbiosolutions.com. For additional information, visit
Emergent’s website and follow Emergent
on LinkedIn, Twitter and Instagram.
Emergent’s Response to COVID-19In addition to
leveraging its established hyperimmune platform to develop
COVID-HIG, Emergent is deploying its molecule-to-market contract
development and manufacturing (CDMO) capabilities, capacities, and
expertise to help governments/non-government organizations and
pharmaceutical/biotechnology partners advance their COVID-19
programs. The company has engaged in nine collaborations to develop
and manufacture COVID-19 vaccine and therapeutic candidates. For
the COVID-19 response, Emergent’s integrated CDMO network provides
development services from its Gaithersburg facility, drug substance
manufacturing at its Baltimore Bayview facility, and drug product
manufacturing at its Baltimore Camden, Rockville, and Winnipeg
facilities.
For more than 22 years Emergent has focused on advancing public
health, and its multi-pronged approach to tackling COVID-19
demonstrates its commitment to its mission – to protect and enhance
life.
Safe Harbor StatementThis press release
includes forward-looking statements within the meaning of the
Private Securities Litigation Reform Act of 1995. Any statements,
other than statements of historical fact, including statements
regarding the ability of COVID-HIG to effectively treat
hospitalized patients with COVID-19, as well as high-risk, acute
symptomatic patients and to become an effective PEP therapeutic for
groups at high risk of developing COVID-19, as well as statements
regarding planned clinical trials, are forward-looking statements.
These forward-looking statements are based on our current
intentions, beliefs and expectations regarding future events. We
cannot guarantee that any forward-looking statement will be
accurate. Investors should realize that if underlying assumptions
prove inaccurate or unknown risks or uncertainties materialize,
actual results could differ materially from our expectations.
Investors are, therefore, cautioned not to place undue reliance on
any forward-looking statement. Any forward-looking statement speaks
only as of the date of this press release, and, except as required
by law, we do not undertake to update any forward-looking statement
to reflect new information, events or circumstances.
There are a number of important factors that could cause the
company's actual results to differ materially from those indicated
by such forward-looking statements, including the overall success
of the collaboration and planned development programs; our ability
to maintain a sufficient level of convalescent plasma; the results
of planned clinical trials and the timing of and our ability to
obtain and maintain regulatory authorizations for emergency or
broader patient use or approvals; and our commercialization,
marketing and manufacturing capabilities. The foregoing sets forth
many, but not all, of the factors that could cause actual results
to differ from our expectations in any forward-looking statement.
Investors should consider this cautionary statement, as well as the
risk factors identified in our periodic reports filed with the SEC,
when evaluating our forward-looking statements.
Media Contact:Matt HartwigDirector, Media
Relationsmediarelations@ebsi.com
Investor Contact:Robert G. BurrowsVice
President, Investor Relations240-631-3280burrowsr@ebsi.com
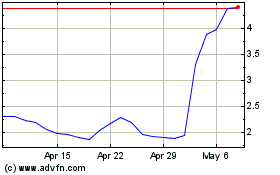
Emergent Biosolutions (NYSE:EBS)
Historical Stock Chart
From Aug 2024 to Sep 2024
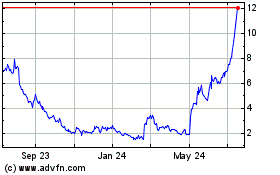
Emergent Biosolutions (NYSE:EBS)
Historical Stock Chart
From Sep 2023 to Sep 2024