false 0001845337 0001845337 2023-08-16 2023-08-16
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): (August 16, 2023)
DAY ONE BIOPHARMACEUTICALS, INC.
(Exact name of registrant as specified in its charter)
|
|
|
|
|
| Delaware |
|
001-40431 |
|
83-2415215 |
(State or other jurisdiction
of incorporation) |
|
(Commission
File Number) |
|
(IRS Employer
Identification No.) |
|
|
|
| 2000 Sierra Point Parkway, Suite 501 |
|
|
| Brisbane, California |
|
94005 |
| (Address of principal executive offices) |
|
(Zip Code) |
Registrant’s telephone number, including area code: (650) 484-0899
N/A
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| ☐ |
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ☐ |
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ☐ |
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ☐ |
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
|
|
|
|
|
| Title of each class |
|
Trading
Symbol(s) |
|
Name of each exchange on which registered |
| Common Stock, par value $0.0001 per share |
|
DAWN |
|
Nasdaq Global Select Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company ☒
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 7.01 Regulation FD Disclosure.
On August 16, 2023, Day One Biopharmaceuticals, Inc. (the “Company”) issued a press release announcing it had entered into a license agreement and research collaboration with Sprint Bioscience (“Sprint”) for Sprint’s VRK1 program. The Company also updated its corporate presentation to reflect the VRK1 program.
A copy of the press release is attached as Exhibit 99.1 to this report. A copy of the updated presentation is attached as Exhibit 99.2 to this report.
The information in this Current Report on Form 8-K, including Exhibits 99.1 and 99.2 to this report, shall not be deemed to be “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section or Sections 11 and 12(a)(2) of the Securities Act of 1933, as amended (the “Securities Act”). The information contained in this Current Report on Form 8-K and in the accompanying Exhibits 99.1 and 99.2 shall not be incorporated by reference into any other filing under the Exchange Act or under the Securities Act, except as shall be expressly set forth by specific reference in such filing.
Item 9.01. Financial Statements and Exhibits.
(d) Exhibits.
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
|
|
|
|
|
|
|
|
|
|
|
|
|
DAY ONE BIOPHARMACEUTICALS, INC. |
|
|
|
|
| Date: August 16, 2023 |
|
|
|
By: |
|
/s/ Charles N. York II, M.B.A. |
|
|
|
|
|
|
Charles N. York II, M.B.A. |
|
|
|
|
|
|
Chief Operating Officer and Chief Financial Officer |
Exhibit 99.1

Day One Announces VRK1 License Agreement and Research Collaboration with Sprint Bioscience
Day One receives an exclusive license to develop and commercialize small molecule drug candidates for pediatric and adult cancers with high
unmet need
Collaboration augments Day One’s portfolio of targeted therapies in oncology
BRISBANE, Calif., Aug. 15, 2023 – Day One Biopharmaceuticals (Nasdaq: DAWN), a clinical-stage biopharmaceutical company dedicated to developing
and commercializing targeted therapies for people of all ages with life-threatening diseases, today announced it has entered into an exclusive, worldwide license agreement and research collaboration with Sprint Bioscience for its VRK1 program.
Vaccinia-related kinase 1 (VRK1) is a novel target involved in the regulation of cell division and DNA damage repair. Over-expression of VRK1 is linked to
poor prognosis in a variety of adult and pediatric cancers, and VRK1 has been identified as a synthetic lethal target in tumors where expression of its paralog, VRK2, is lost. Silencing of VRK2 expression via promoter methylation has been noted in
the majority of high-grade gliomas and high-risk neuroblastomas, providing a concrete approach for selecting patients with tumors sensitive to VRK1 inhibition.
“This collaboration is an important continuation of measured portfolio development at Day One, which focuses on targeted therapies for children and
adults with cancer in need of novel treatment approaches,” said Dr. Samuel Blackman, co-founder and head of research and development, Day One. “We look forward to collaborating with Sprint
Bioscience, who has strong discovery and research expertise, and working to advance the VRK1 program through lead optimization and into the clinic.”
Under the terms of the agreement, Day One will make an upfront payment of $3 million to Sprint Bioscience and reimburse Sprint Bioscience for pre-clinical research and development expenses. Sprint Bioscience will be eligible to receive additional milestone payments of up to approximately $313 million plus single-digit royalties pending achievement of
certain research, development, regulatory and commercial outcomes.
About Day One Biopharmaceuticals
Day One Biopharmaceuticals is a clinical-stage biopharmaceutical company that believes when it comes to pediatric cancer, we can do better. We put kids first
and are developing targeted therapies that deliver to their needs. Day One was founded to address a critical unmet need: the dire lack of therapeutic development in pediatric cancer. The Company’s name was inspired by “The Day One
Talk” that physicians have with patients and their families about an initial cancer diagnosis and treatment plan. Day One aims to re-envision cancer drug development and redefine what’s possible for
all people living with cancer—regardless of age—starting from Day One.
page 1 of 2
Day One partners with leading clinical oncologists, families, and scientists to identify, acquire, and
develop important emerging cancer treatments. The Company’s lead product candidate, tovorafenib, is an investigational, oral, brain-penetrant, highly-selective type II pan-RAF kinase inhibitor. The
Company’s pipeline also includes pimasertib, an investigational, oral, highly-selective small molecule inhibitor of mitogen-activated protein kinases 1 and 2 (MEK-1/-2). Day One is based in Brisbane, California. For more information, please visit www.dayonebio.com or find the company on LinkedIn or Twitter.
Cautionary Note Regarding Forward-Looking Statements
This press release contains “forward-looking” statements within the meaning of the “safe harbor” provisions of the Private Securities
Litigation Reform Act of 1995, including, but not limited to: Day One’s plans to develop cancer therapies, the results of Day One’s VRK1 program and its collaboration with Sprint Bioscience, expectations from current clinical trials, the
execution of the Phase 2 and Phase 3 clinical trials for tovorafenib as designed, any expectations about safety, efficacy, timing and ability to complete clinical trials, release data results and to obtain regulatory approvals for tovorafenib and
other candidates in development, and the ability of tovorafenib to treat pLGG or related indications.
Statements including words such as
“believe,” “plan,” “continue,” “expect,” “will,” “develop,” “signal,” “potential,” or “ongoing” and statements in the future tense are forward-looking
statements. These forward-looking statements involve risks and uncertainties, as well as assumptions, which, if they do not fully materialize or prove incorrect, could cause our results to differ materially from those expressed or implied by such
forward-looking statements.
Forward-looking statements are subject to risks and uncertainties that may cause Day One’s actual activities or results
to differ significantly from those expressed in any forward-looking statement, including risks and uncertainties in this press release and other risks set forth in our filings with the Securities and Exchange Commission, including Day One’s
ability to develop, obtain regulatory approval for or commercialize any product candidate, Day One’s ability to protect intellectual property, the potential impact of global business or macroeconomic conditions, including as a result of
inflation, rising interest rates, instability in the global banking system, and geopolitical conflicts and the sufficiency of Day One’s cash, cash equivalents and investments to fund its operations. These forward-looking statements speak only
as of the date hereof and Day One specifically disclaims any obligation to update these forward-looking statements or reasons why actual results might differ, whether as a result of new information, future events or otherwise, except as required by
law.
DAY ONE MEDIA
Laura Cooper, Head of
Communications
media@dayonebio.com
DAY ONE
INVESTORS
LifeSci Advisors, PJ Kelleher
pkelleher@lifesciadvisors.com
#####
page 2 of 2

Exhibit 99.2 Day One Biopharmaceuticals Targeted Therapies for People of
All Ages August 2023 Day One Biopharmaceuticals

Disclaimer This presentation and the accompanying oral commentary
contain forward-looking statements that are based on our management’s beliefs and assumptions and on information currently available to our management. Forward-looking statements are inherently subject to risks and uncertainties, some of which
cannot be predicted or quantified. In some cases, you can identify forward-looking statements by terminology such as “may,” “will,” “should,” “could,” “expect,” “plan,”
anticipate,” “believe,” “estimate,” “predict,” “intend,” “potential,” “would,” “continue,” “ongoing” or the negative of these terms or other
comparable terminology. Forward-looking statements include all statements other than statements of historical fact contained in this presentation, including information concerning our future financial performance, including the sufficiency of our
cash, cash equivalents and short-term investments to fund our operations, business plans and objectives, timing and success of our planned nonclinical and clinical development activities, the results of any of our strategic collaborations, including
the potential achievement of milestones and provision of royalty payments thereunder, timing and results of nonclinical studies and clinical trials, efficacy and safety profiles of our product candidates, execution of the Phase 2 and Phase 3
clinical trials for tovorafenib and the Phase 1b/2 clinical trial for tovorafenib and pimasertib as designed, any expectations about safety, efficacy, timing and ability to complete clinical trials and to obtain regulatory approvals for tovorafenib
and other candidates in development, the ability of tovorafenib to treat pediatric low-grade glioma (pLGG) or related indications, the potential therapeutic benefits and economic value of our product candidates, potential growth opportunities,
competitive position, industry environment and potential market opportunities, our ability to protect intellectual property and the impact of global business or macroeconomic conditions, including as a result of inflation, rising interest rates,
instability in the global banking system, and geopolitical conflicts, including the war in Ukraine, on our business and operations. Forward-looking statements are subject to known and unknown risks, uncertainties, assumptions and other factors. It
is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in
any forward-looking statements we may make. These factors, together with those that are described under the heading “Risk Factors” contained in our most recent Quarterly Report on Form 10-Q filed with the Securities and Exchange
Commission (“SEC”) and other documents we file from time to time with the SEC, may cause our actual results, performance or achievements to differ materially and adversely from those anticipated or implied by our forward-looking
statements. In addition, statements that “we believe” and similar statements reflect our beliefs and opinions on the relevant subject. These statements are based upon information available to us as of the date of this presentation, and
although we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and our statements should not be read to indicate that we have conducted a thorough inquiry into, or review of, all
potentially available relevant information. These statements are inherently uncertain and investors are cautioned not to unduly rely upon these statements. Furthermore, if our forward-looking statements prove to be inaccurate, the inaccuracy may be
material. In light of the significant uncertainties in these forward-looking statements, you should not regard these statements as a representation or warranty by us or any other person that we will achieve our objectives and plans in any specified
time frame, or at all. We undertake no obligation to publicly update any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law. This presentation also contains estimates and other
statistical data made by independent parties and by us relating to market size and growth and other data about our industry. This data involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such
estimates. In addition, projections, assumptions and estimates of our future performance and the future performance of the markets in which we operate are necessarily subject to a high degree of uncertainty and risk. Day One Biopharmaceuticals
2

Cancer Drug Development For People Of All Ages Mission That Tovorafenib
(DAY101) Growing Portfolio and Runway Lead Program Creates Value Beyond Clinical Milestones • Day One’s mission is to help children • Investigational, oral, CNS-penetrant • Two clinical-stage MEKi assets, in- with cancer,
from day one and every Type II RAF inhibitor licensed for combination trials day after • Being developed as tablets and • Research collaboration and license • Develop medicines for genomically- pediatric-friendly liquid suspension
agreement for preclinical program defined cancers targeting VRK1 • Breakthrough Therapy Designation • Establish first-in-class position • Projected cash runway into 2026 • Rare Pediatric Disease Designation through rapid
pediatric registration • Key FIREFLY-1 milestones • Orphan Drug Designation (US/EU) • Expand to adolescent and adult 1 – Initiated rolling NDA in May 2023 populations in parallel and pursue – Expected completion of
rolling those opportunities with the same NDA by October 2023 following commitment we do for children submission of an amended clinical 2 study report 1 2 NDA data set will include analysis of primary (ORR by RANO-HGG) and secondary (ORR by
RAPNO-LGG, PFS) efficacy endpoints, safety, and exploratory analyses (including ORR by RANO-LGG). Amended clinical study report will include safety and efficacy data from a planned June 2023 data cutoff. Day One Biopharmaceuticals 3

Our Pipeline Recent & Product Candidate Indication Preclinical Phase
1 Phase 2 Phase 3 Anticipated Milestones Initiated rolling NDA: May 2023 Tovorafenib (DAY101) New clinical data presented: 1 Relapsed pLGG FIREFLY-1 (pivotal) June 2023 Type II Pan-RAF Inhibitor Expected rolling NDA complete: October 2023 •
FDA Breakthrough Therapy Designation for relapsed pLGG First patient dosed: Frontline pLGG FIREFLY-2 (pivotal) • FDA Rare Pediatric Disease March 2023 Designation (PRV Eligible) for pLGG • FDA Orphan Drug Designation First patient dosed:
RAF-altered for malignant glioma November 2021 2 FIRELIGHT-1* solid tumors Poster presented: • EC Orphan Designation for glioma (monotherapy) April 2023 MAPK-altered Pimasertib First patient dosed: 3 FIRELIGHT-1* solid tumors May 2022 MEK 1/2
Inhibitor (Combo w/tovorafenib) 4 VRK1 Program Pediatric and In-licensed: August 2023 VRK1 Inhibitor adult cancers 1 2 3 *Includes patients ≥12 years of age. FIREFLY-1 Arm 1 expected to support registration. DAY101 adult monotherapy Phase 1
dose escalation and expansion trial previously completed. Pimasertib Phase 1 dose escalation and expansion trial 4 previously completed. pLGG, pediatric low-grade glioma. Tovorafenib and Pimasertib are investigational products. Safety and efficacy
have not been established by any health authority. Research collaboration and license agreement with Sprint Bioscience for exclusive worldwide rights to a research-stage program targeting VRK1. Day One Biopharmaceuticals 4

Tovorafenib (DAY101) Type II RAF Inhibitor Day One
Biopharmaceuticals

Pediatric Low-Grade Glioma (pLGG): The Most Common Type Of Brain Tumor
In Children PLGGs are chronic and relentless, with patients suffering profound tumor and 6 treatment-associated morbidity that can impact their life trajectory over the long term 7 A Serious and Life-Threatening Disease Disease Symptoms • An
estimated 26,000 children/young adults are living Cerebral gliomas: 1,2 with BRAF-altered pLGGs in the U.S. today Seizures, muscle weakness, Cerebellar gliomas: behavioral changes • Surgery plays a significant role in treatment, but 70% of
Impaired balance, 3,4 coordination or depth patients require systemic therapy perception Hypothalamic gliomas: • For the majority of patients in the relapse setting, there Endocrine dysfunction and is no standard of care and no approved
therapies visual deficits Brain stem gliomas: Difficulty swallowing or with • ~70% of pLGGs have BRAF alterations, of these ~85% speech, abnormal breathing Optic pathway gliomas: 5 are BRAF fusions and ~15% are BRAF V600E mutations Decreased
vision (acuity and/or fields), bulging or misalignment of eyes • Majority of patients have many years of treatment until the tumors typically senesce by their mid-20s 1 2 CBTRUS, Qaddoumi et al 2009, Schreck et al 2019, ClearView Analysis;
SEER US complete prevalence counts of patients aged under 25 with Brain and Other Nervous Systems tumors as of January 1, 2017. Estimated 3 4 5 prevalence are Day One calculations based on publicly available data. Ostrum QT et al., Neuro Oncol.
2015; 16(Suppl 10):x1-x36; De Blank P. et al., Curr Opin Pediatr. 2019 Feb; 31(1):21-27. Jones DTW et al., Cancer Res. 6 7 2008; 68:8673–77. Traunwieser T et al., Neurooncol Adv. 2020; 2:vdaa094. Sievert AJ, Fisher MJ. Pediatric low-grade
gliomas. J Child Neurol. 2009;24(11):1397-1408. doi:10.1177/0883073809342005. Day One Biopharmaceuticals 6

Current Treatments Can Be Disruptive To Childhood and Can Have
Significant Long-Term Consequences Surgery Chemotherapy Radiation Targeted Therapies • Significant recovery times • Requirement for indwelling • Risk of secondary • Rash catheter and weekly malignancy • Risks of
complications • Fever infusions • Risk of malignant • Resection may be limited by • Vomiting transformation • Neutropenia location of tumor • Fatigue • Hypersensitivity reactions • Risk of vascular
proliferation • Potential for functional • Anemia and stroke deficits based on location of • Nausea and vomiting • Nail infections tumor and extend of • Neurocognitive impact, • Peripheral neuropathy resection
depending on location of • Ophthalmologic toxicity tumor and radiation field • Cardiac toxicity High unmet need for an effective therapy for the majority of pLGG relapsed or progressive patients that is minimally disruptive to their
lives. Source: 1. Heitzer AM, Raghubar K, Ris MD, et al. Neuropsychological functioning following surgery for pediatric low-grade glioma: a prospective longitudinal study. J Neurosurg Pediatr. 2019;1-9. doi:10.3171/2019.9.PEDS19357. 2. Bryant R.
Managing side effects of childhood cancer treatment. J Pediatr Nurs. 2003;18(2):113-125. doi:10.1053/jpdn.2003.11. 3. Zahnreich S, Schmidberger H. Childhood cancer: occurrence, treatment and risk of second primary malignancies. Cancers (Basel).
2021;13(11):2607. doi:10.3390/cancers/13112607. 4. National Cancer Institute. Fertility issues in girls and women with cancer. http://www.cancer.gov. Accessed June 13, 2022. 5. Alessi I., Caroleo A.M., de Palma L., Mastronuzzi A., Pro S., Colafati
G.S., Boni A., Della Vecchia N., Velardi M., Evangelisti M., et al. Short and Long-Term Toxicity in Pediatric Cancer Treatment: Central Nervous System Damage. Cancers. 2022;14:1540. doi: 10.3390/cancers14061540. Day One Biopharmaceuticals
7

Tovorafenib (DAY101) Inhibits Both BRAF Fusions And BRAF V600 Mutations
• Tovorafenib (DAY101) is an investigational, MAPK pathway RAS-independent activation of the MAPK pathway oral, selective, CNS-penetrant, type II pan- RAF inhibitor that was designed to inhibit RAS both monomeric and dimeric RAF kinase ‒
Activity in tumors driven by both RAF RAF RAF fusion fusions and BRAF V600E mutations RAF Tovorafenib mutation ‒ Tablet and pediatric-friendly liquid suspension MEK ‒ Once weekly dosing • Currently approved type I BRAFi are
indicated for use in patients with tumors bearing BRAF ERK V600E mutations ‒ Type I BRAF inhibitors cause paradoxical Proliferation and survival Proliferation and survival Proliferation and survival MAPK activation in the setting of wild-type
RAF, increasing the risk of tumor growth in BRAF fusion-driven Source: 1. Sun Y et al., Neuro Oncol. 2017; 19: 774–85; 2. Sievart AJ et al., PNAS. 2013; 110:5957-62; 3. Karajannis MA et al., Neuro Oncol 2014;16(10):1408-16 Day One
Biopharmaceuticals 8

The Current pLGG Treatment Paradigm Reflects The Unrelenting Nature Of
This Chronic Brain Tumor 1L 2L 3L Presentation Surgical Intervention ~35% ~20% Targeted Tx Targeted Tx Targeted Tx GTR No Recurrence (5-10%) (40-50%) (40-50%) 1 Biopsy (>95%) Suspected ~65% ~50% ~50% pLGG ≤ Partial Eventual Additional lines
Chemo Chemo Chemo Resection Recurrence of therapy ~40% ~80% (~90%) (~50%) (~35%) Molecular Testing Biopsy Only Other Other Other ~20% (<5%) (<5%) (<20%) No Biopsy (~5%) Response, Response, ~35% no recurrence ~50% no recurrence Because many
pLGGs undergo senescence when patients reach their 20s, the goal of therapy is to maximize tumor control while minimizing treatment-associated toxicities from surgery, chemotherapy, and radiation. As a result, a large number of pLGG patients will
undergo multiple lines of systemic therapy over the course of their disease. 1 Source: Physician Interviews, Bandopadhayay et al. Pediatric Blood Cancer. 2014; Sievert and Fischer. J Child Neurol. 2009; ClearView Analysis. GTR: Gross Total Resection
Molecular testing of biopsied samples occurs in all patients. Kandels et. al. Retrospective analysis of comprehensive SIOP registry; Hargrave et. al. Phase I/II;Fangusaro et. al. Phase II Day One Biopharmaceuticals 9

Pivotal Phase 2 Trial Of Monotherapy Tovorafenib (DAY101) In Relapsed
Or Progressive pLGG (FIREFLY-1) Trial Design Endpoints (Pivotal Arm 1) 1 • Three arm, open-label, global registrational phase 2 trial • Primary endpoint: ORR based on RANO-HGG , assessed by blinded independent central review •
Pivotal Arm 1 (recurrent/progressive pLGG, n=77): harboring a 2 KIAA1549-BRAF fusion or BRAF V600E mutation • Secondary endpoints: ORR by RAPNO-LGG assessed by blinded independent central review; PFS, DoR; TTR, CBR; safety • Arm 2
(expanded access recurrent/progressive LGG, n=59): harboring 3 an activating RAF alteration • Exploratory analyses: ORR and CBR by RANO-LGG assessed by blinded independent central review • Arm 3 (extracranial solid tumors): harboring an
activating RAF fusion Key Inclusion Criteria Clinical and radiological evaluations at baseline, and every rd nd 3 cycle for pLGG and every 2 cycle for solid tumors • 6 months – 25 years of age • RAF-altered tumor Enrollment/ Day
–28 to 0 • ≥1 prior line of systemic Screening Baseline End of Trial therapy with radiographic (C1D1) Study Drug Administration After Cycle 27: patients may either continue progression C27D1 2 treatment or enter drug holiday period
at any time 420mg/m QW (not to exceed 600mg), • Prior use of MAPK pathway (at discretion of investigator) QW in 28-day cycles targeted therapy was permitted Eligibility evaluation Treatment period: minimum of 2 years or until progression or
toxicity/intolerability 1 2 3 Dec 22, 2022 data cutoff. Wen PY, et al. J Clin Oncol. 2010;28(11):1963-1972. Fangusaro J, et al. Lancet Oncol. 2020;21(6):e305–316. van den Bent MJ, et al. Lancet Oncol. 2011;12(6):583-593. Abbreviations: CBR,
clinical benefit rate; IRC, independent review committee; C, cycle; D, day; LGG, low-grade glioma; ORR, objective response rate; PFS, progression-free survival; DoR, duration of response; QW, once weekly; TTR, time to response; RANO, Response
Assessment in Neuro-Oncology; RAPNO, Response Assessment in Pediatric Neuro-Oncology; MAPK, mitogen-activated protein kinase. For more information, please refer to NCT04775485 Day One Biopharmaceuticals 10

FIREFLY-1 Baseline Patient Characteristics Location (n=77) Deep midline
structures Characteristic Arm 1 (n=77) 12% Optic pathway 51% Median age, years (range) 8 (2-21) Other Sex, n (%) * 16% Male 40 (52) Cerebral hemisphere Female 37 (48) 8% Cerebellum Race, n (%) 6% Brain stem Black or African American 2 (3) 8% Asian 5
(6) White 41 (53) BRAF alteration (n=77) Multiple 3 (4) Other 6 (8) BRAF V600E Not reported 20 (26) 17% Number of lines of prior systemic therapy Median (range) 2 (1-9) # BRAF fusion 1, n (%) 18 (23) 83% 2, n (%) 21 (27) ≥3, n (%) 38 (49)
Prior MAPK pathway targeted therapy, n (%) 46 (60) Dec 22, 2022 data cutoff. *Includes tumors that were extending into multiple regions of the brain, leptomeningeal disease, and/or spinal disease. #Includes 6 patients with BRAF duplication and 2
with BRAF rearrangement per FISH (Fluorescence in situ hybridization) or ISH (in situ hybridization). MAPK, mitogen-activated protein kinase. Day One Biopharmaceuticals 11

Tumor Response To Tovorafenib (DAY101) For All Patients With RANO-HGG
Evaluable Lesions (n=69) 1 RANO-HGG Response (IRC) Evaluable n=69 ORR (cCR + cPR + uPR), n (%) 46 (67%)* Clinical benefit rate, n (%) cCR, cPR/uPR, or SD 64 (93%) cCR, cPR/uPR, or SD for 12 mo+ 49 (71%) Best overall response, n (%) CR 4 (6%) PR
(includes 3 uPR) 42 (61%) SD 18 (26%) PD 4 (6%) Not evaluable 1 (1%) All 3 patients with uPR remain on treatment as of May 23, 2023 Dec 22, 2022 data cutoff. Percents may not add to 100% due to rounding. Two of 69 patients are not shown in the
waterfall plot; one patient passed away due to progressive disease (not related to tovorafenib) before the first imaging assessment and one did not receive T1 Gd+ 2 1 2 follow-up imaging. *P<0.001 from two-sided exact binomial test to test null
hypothesis of ORR=21% based on Bouffet et al. Wen PY, et al. J Clin Oncol. 2010;28(11):1963-1972. Bouffet E, et al. J Clin Oncol. 2012;30(12):1358-1363. CBR, clinical benefit rate; cCR, confirmed completed response; cPR, confirmed partial response;
CR, complete response; HGG, high-grade glioma; IRC, independent radiology review committee; MAPKi, mitogen-activated protein kinase inhibitor; MR, minor response; ORR, overall response rate; PD, progressive disease; PR, partial response; RANO,
Response Assessment in Neuro-Oncology; SD, stable disease; uPR, unconfirmed partial response. There are 17 patients with stable disease of less than 12 months duration and at the time of the data cutoff, 11 remain on treatment. Day One
Biopharmaceuticals 12

Tumor Response To Tovorafenib (DAY101) For All Patients With RAPNO-LGG
Evaluable Lesions (n=69*) 1 RAPNO-LGG Response (IRC) Evaluable n=69 ORR (cCR + cPR/uPR + cMR/uMR), n (%) 35 (51%) Clinical benefit rate, n (%) cCR, cPR/uPR, cMR/uMR, or SD 60 (87%) cCR, cPR/uPR, cMR/uMR, or SD for 12mo+ 36 (52%) Best overall
response, n (%) CR 0 (0%) PR (includes 4 uPR) 17 (25%) MR (includes 4 uMR) 18 (26%) SD 25 (36%) # PD 8 (12%) Not evaluable 1 (1%) All 4 patients with uPR and 3 patients with uMR remain on treatment as of May 23, 2023 Dec 22, 2022 data cutoff.
Percents may not add to 100% due to rounding. Two of 69 patients not shown in waterfall plot; one patient passed away due to progressive disease (not related to tovorafenib) before the first imaging assessment and one patient had visual progressive
disease but no evaluable T2 measurements at 1 # the time of progression. *Pending adjudication. Fangusaro J, et al. Lancet Oncol. 2020;21(6):e305-316. PD for RAPNO-LGG was not used to determine treatment discontinuation; patients could continue
treatment if there was no PD based on RANO-HGG per investigator’s assessment. CBR, clinical benefit rate; cCR, confirmed completed response; cMR, confirmed minor response; cPR, confirmed partial response; CR, complete response; HGG, high-grade
glioma; IRC, independent radiology review committee; LGG, low-grade glioma; MAPKi, mitogen-activated protein kinase inhibitor; MR, minor response; ORR, overall response rate; PD, progressive disease; PR, partial response; RANO, Response Assessment
in Neuro-Oncology; RAPNO, Response Assessment in Pediatric Neuro-Oncology; SD, stable disease; uMR, unconfirmed minor response; uPR, unconfirmed partial response. There are 28 patients with stable disease of less than 12 months duration and at the
time of the data cutoff, 11 remain on treatment. Day One Biopharmaceuticals 13

Tumor Response To Tovorafenib (DAY101) For All Patients With RANO-LGG
Evaluable Lesions (n=76) 1 RANO-LGG Response (IRC) Evaluable n=76 ORR (cCR + cPR/uPR + cMR/uMR), n (%) 37 (49%) Clinical benefit rate, n (%) cCR, cPR/uPR, cMR/uMR, or SD 63 (83%) cCR, cPR/uPR, cMR/uMR, or SD for 12mo+ 39 (51%) Best overall response,
n (%) CR 0 (0%) PR (includes 8 uPR) 20 (26%) MR (includes 2 uMR) 17 (22%) SD 26 (34%) # PD 11 (14%) § Not evaluable 2 (3%) All 8 patients with uPR and 2 patients with uMR remain on treatment as of May 23, 2023 # § Dec 22, 2022 data cutoff.
Percents may not add to 100% due to rounding. PD for RANO-LGG was not used to determine treatment discontinuation; patients could continue treatment if there was no PD based on RANO-HGG per investigator’s assessment. Two of 76 patients are not
shown in the waterfall plot; one 1 patient passed away due to progressive disease (not related to tovorafenib) before the first imaging assessment, and one patient with missing T1 Gd+ imaging at baseline was deemed NE at all timepoints but had a
best SPPD decrease of 65% on T2 imaging. van den Bent MJ, et al. Lancet Oncol. 2011;12(6):583-593. BL, baseline; CBR, clinical benefit rate; cCR, confirmed completed response; cMR, confirmed minor response; cPR, confirmed partial response; CR,
complete response; HGG, high-grade glioma; IRC, independent radiology review committee; LGG, low-grade glioma; MAPKi, mitogen-activated protein kinase inhibitor; MR, minor response; ORR, overall response rate; PD, progressive disease; PR, partial
response; RANO, Response Assessment in Neuro-Oncology; SD, stable disease; SPPD, sum of the products of perpendicular diameters; uMR, unconfirmed minor response; uPR, unconfirmed partial response. There are 27 patients with stable disease of less
than 12 months duration and at the time of the data cutoff, 19 remain on treatment. Day One Biopharmaceuticals 14

Duration Of Tovorafenib (DAY101) Therapy For All Patients With RANO-HGG
Evaluable Lesions (n=69) 2.8 Median time to response* MONTHS 11/17 patients with stable disease <12 months remain on treatment as of data cutoff Median duration of 10.8 MONTHS treatment* Ongoing treatment Partial response Discontinued treatment
Complete response Follow-up after discontinuation of treatment Progressive disease BRAF V600E mutation Discontinuation due to AE Prior MEK inhibitor therapy Death Prior BRAF inhibitor therapy * Analysis for median time to response and median
duration of treatment only included confirmed responses. HGG, high-grade glioma; RANO, Response Assessment in Neuro-Oncology. Day One Biopharmaceuticals 15

Duration Of Tovorafenib (DAY101) Therapy For All Patients With RANO-HGG
& RANO-LGG Evaluable Lesions RANO-HGG (n=69) RANO-LGG (n=76) As of the data cutoff, As of the data cutoff, the median IRC- the median IRC- assessed DOR based on assessed DOR based on RANO-HGG criteria was RANO-LGG criteria was not yet reached
(95% 14.4 months (95% CI: 8.4, not CI: 9.0, not estimable)* estimable)* * * * Analysis for median time to response and median duration of response only included confirmed responses. BOR is shown; circles indicate start of response. PD for the
purpose of treatment was based on RANO-HGG, not RANO-LGG. BOR, best overall response; HGG, high-grade glioma; LGG, low-grade glioma; PD, progressive disease; RANO, Response Assessment in Neuro-Oncology; DOR, duration of response; CI, confidence
interval. Day One Biopharmaceuticals 16

All RANO-LGG Unconfirmed PR Patients Continue On Treatment With
Demonstrable Deepening Of Response (n=8) Responses for Patients with Unconfirmed Partial Response by RANO-LGG SD MR PR CR Months from Baseline All 8 patients with unconfirmed partial response by RANO-LGG remain on treatment as of May 23, 2023 Spider
plot for SPPD – baseline and after treatment (RANO-LGG by IRC) – unconfirmed PR patients EOT status based on May 23, 2023 EDC data. Individual patient response data is current as of the data cutoff of December 22, 2022; treatment status
data is current as of May 23, 2023. Day One Biopharmaceuticals 17 Percent Change from Baseline (%)

Nearly Half Of Patients With Best Response Of PD By RANO-LGG Have Tumor
Stabilization And Response With Continued Treatment (n=11) Ongoing treatment Discontinued treatment 5/11 patients with best response of PD by RANO-LGG remain on treatment as of May 23, 2023 Individual patient response data is current as of the data
cutoff of December 22, 2022; treatment status data is current as of May 23, 2023. Day One Biopharmaceuticals 18

Tovorafenib (DAY101) Safety Data (n=136) • The vast majority of
treatment-emergent AEs Treatment-emergent AEs Treatment-related AEs were Grade 1 or 2 Preferred term, n (%) Any grade Grade ≥3 Any grade Grade ≥3 • 39 patients (29%) required dose modifications Any AE 136 (100) 68 (50) 133 (98) 47
(35) due to treatment-related AEs Hair color changes 96 (71) - 96 (71) - Fatigue 68 (50) 4 (3) 54 (40) 4 (3) • Dose interruptions were brief, with the median time of dose interruption being 2 Vomiting 59 (43) 3 (2) 24 (18) 3 (2) weeks Rash
maculo-papular 56 (41) 10 (7) 51 (38) 10 (7) Headache 53 (39) 1 (1) 27 (20) - • 5 patients (4%)* discontinued due to AE, with Pyrexia 43 (32) 2 (1) 15 (11) 1 (1) 4 patients (3%) discontinuing due to Nausea 40 (29) - 21 (15) - treatment-related
AEs Dry skin 39 (29) - 34 (25) - • The most commonly reported lab Dermatitis acneiform 37 (27) 1 (1) 36 (26) 1 (1) abnormalities were CPK elevation, anemia, Constipation 36 (26) - 28 (21) - hypophosphatemia, and AST elevation Decreased
appetite 35 (26) 4 (3) 25 (18) 3 (2) • Nearly all had no clinical manifestations and Epistaxis 34 (25) - 22 (16) - did not require clinical intervention or change in study treatment Dec 22, 2022 data cutoff. Table shows treatment-emergent AEs
with frequency ≥25% of any grade. Rash erythematous treatment-emergent: any grade, 14 (10%); grade ≥3 1 (1%); treatment-related: any grade, 14 (10%), grade ≥3 1 (1%). *One patient had 2 events (shunt malfunction [not related to
tovorafenib] and tumor hemorrhage [related to tovorafenib]). AEs, adverse events. Day One Biopharmaceuticals 19

Incidence And Prevalence Of BRAF-Altered pLGG In The U.S. 2020 2017
Estimated Incidence Estimated SEER Prevalence Under 25 Under 25 1 US Population ~105,000,000 NA 2 3 Rate of CNS Tumors (0.00521%) ~5,500 ~130,000 2 Gliomas (63%) ~3,500 ~82,000 2 Low Grade (77%) ~2,600 ~63,000 2 Has Received Drug Tx (58%) ~1,500
~36,000 2 BRAF Altered (70%) ~1,100 ~26,000 ~1,100 ~26,000 Estimated Annual Incidence Estimated Prevalence 1 2 3 US Census; CBTRUS, Qaddoumi et al 2009, Schreck et al 2019, ClearView Analysis; SEER US complete prevalence counts of patients aged
under 25 with Brain and Other Nervous Systems tumors as of January 1, 2017. Estimated annual incidence and estimated prevalence are Day One calculations based on publicly available data. Day One Biopharmaceuticals 20

Key Takeaways From FIREFLY-1 Data And Next Steps • Clinically
meaningful data from FIREFLY-1 for pediatric patients with recurrent or progressive LGG harboring BRAF fusions or BRAF V600E mutations • 67% ORR and 93% clinical benefit rate by RANO-HGG Next Steps • 51% ORR and 87% clinical benefit rate
by RAPNO-LGG* • 49% ORR and 83% clinical benefit rate by RANO-LGG • Expected completion of rolling NDA by October 2023 following • Responses were observed in patients with either BRAF fusion or submission of an amended clinical
BRAF V600E mutations study report (CSR) # • Rapid time to response regardless of response assessment criteria • CSR will include safety and efficacy • Responses seen in a heavily-pretreated population where the data from a planned
June 2023 data cutoff majority of patients relapsed or progressed after one or more prior MAPK inhibitors • Encouraging safety and tolerability profile indicating monotherapy tovorafenib to be generally well-tolerated # Dec 22, 2022 data
cutoff. *Pending adjudication. Analysis for median time to response only included confirmed responses. Day One Biopharmaceuticals 21

FIREFLY-2/LOGGIC Pivotal Phase 3 Trial of Tovorafenib (DAY101) in
Frontline pLGG Day One Biopharmaceuticals Confidential Information

FIREFLY-2/LOGGIC Pivotal Phase 3 Trial Of Tovorafenib (DAY101) In
Frontline pLGG Trial Design Endpoints • Randomized, global, registrational Phase 3 trial of monotherapy tovorafenib • Primary endpoint: ORR based on RANO-LGG criteria, assessed by blinded 1 (DAY101) vs SoC chemotherapy independent
central review • Eligibility: Patients aged 6 months to <25 years with LGG harboring a RAF ‒ The ORR primary analysis is expected to occur ~12 months after the last alteration and requiring first-line systemic therapy patient
randomized • Tovorafenib (DAY101) available as tablets and pediatric-friendly liquid • Key secondary endpoints: PFS and DoR by RANO criteria, ORR by RAPNO suspension criteria • Patients who progress after stopping tovorafenib
(DAY101) may be re- • Other secondary endpoints: changes in neurological and visual function, challenged safety, and tolerability • Patients who progress in the SoC arm during or post-treatment may cross- • Key exploratory
objectives: QoL and health utilization measures over to receive tovorafenib 2 Non-resectable or Tovorafenib, 420mg/m QW sub-total resected LGG (not to exceed 600 mg) AND Stratified by Long-term follow-up (48 months) Requiring first-line •
Location of tumor Investigator's choice of systemic therapy • Genomic alteration vincristine/carboplatin* or N ≈ 400 • CDKN2A status vinblastine • Infant CHG diagnosis * COG or SIOPe-LGG regimen. Abbreviations: CHG,
chiasmatic, hypothalamic glioma; DoR, duration of response; LGG, low-grade glioma; ORR, objective response rate; QoL, quality of life; QW, once weekly; SoC, standard 1 of care. Primary endpoint of FIREFLY-2 will be ORR by RANO-LGG (2017) following
full approval by FDA on March 16, 2023 of dabrafenib with trametinib in pediatric patients with low-grade glioma with a BRAF V600E mutation who require systemic therapy based on a study with the same primary endpoint. Day One Biopharmaceuticals 23
1:1 Randomization

FIRELIGHT-1 Phase 1b/2 Trials Evaluating Tovorafenib (DAY101) as a
Monotherapy and as a Combination with Pimasertib Day One Biopharmaceuticals Confidential Information

Phase 2 Study Of Monotherapy Tovorafenib (DAY101) In Solid Tumors
(FIRELIGHT-1) 1 Trial Design Endpoints • Single arm, open-label, global phase 1b/2a trial • Primary endpoint: ORR by RECIST version 1.1 for non-CNS solid tumors and RANO-HGG criteria for any CNS tumors • n = 40 patients
(approximately) • Secondary endpoints: safety and additional • Eligibility: Patients aged 12 years and older with non- efficacy parameters hematologic tumor with an activating BRAF fusion, CRAF/RAF1 fusion, or CRAF/RAF1 amplification
Melanoma cohort Patients with a known BRAF Safety 2 DAY101 QW until disease progression or CRAF/RAF1 fusion, or Follow Up CRAF/RAF1 amplification “Tissue agnostic” cohort Study Drug Administration ≥ 18 years at 600 mg PO QW 2 12 to
<18 years at 420mg/m PO QW 1 Abbreviations: ORR, objective response rate; QW, once weekly ; PO, by mouth; BRAF, B-Raf proto-oncogene. Umbrella master study – DAY101-102 (main protocol) DAY101 and MAPK pathway aberration, Sub-study 1 2
monotherapy (DAY101-102a), Sub-study 2 MEK combo (DAY101-102b). DAY101 QW until disease progression, intolerable toxicity, withdrawal of consent, or death Day One Biopharmaceuticals 25
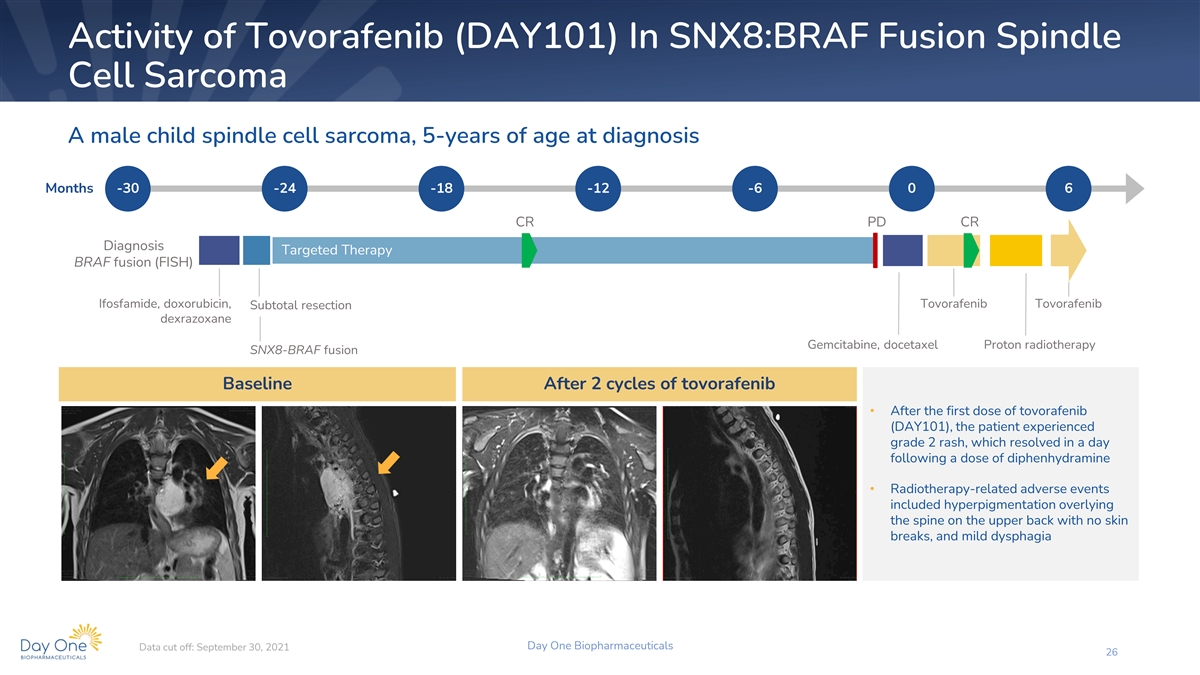
Activity of Tovorafenib (DAY101) In SNX8:BRAF Fusion Spindle Cell
Sarcoma A male child spindle cell sarcoma, 5-years of age at diagnosis Months -30 -24 -18 -12 -6 0 6 CR PD CR Diagnosis Targeted Therapy BRAF fusion (FISH) Ifosfamide, doxorubicin, Tovorafenib Tovorafenib Subtotal resection dexrazoxane Gemcitabine,
docetaxel Proton radiotherapy SNX8-BRAF fusion Baseline After 2 cycles of tovorafenib • After the first dose of tovorafenib (DAY101), the patient experienced grade 2 rash, which resolved in a day following a dose of diphenhydramine •
Radiotherapy-related adverse events included hyperpigmentation overlying the spine on the upper back with no skin breaks, and mild dysphagia Day One Biopharmaceuticals Data cut off: September 30, 2021 26

Pimasertib MEK1/2 Inhibitor Day One Biopharmaceuticals

Pimasertib: Investigational Allosteric MEK1/2 Inhibitor With
Demonstrated Activity In MAPK-Driven Solid Tumors • Pimasertib is an investigational orally-bioavailable, selective, non-competitive MEK1/2 inhibitor in- licensed from Merck KGaA in February 2021 • Extensive non-clinical and clinical
development work through Phase 2, including a solid tumor trial in Japan and combinations with other MOAs • Main AEs typical for all in-class allosteric MEK inhibitors (GI, CPK elevation, skin rash, visual disturbances) • Nearly
three-fold higher CNS penetration than other MEKi inhibitors (trametinib or selumetinib) • Pimasertib showed monotherapy clinical activity, including an improvement in median PFS versus dacarbazine in NRAS mutant melanoma • Combination
with tovorafenib (DAY101) and other targeted therapies may unlock the full value of pimasertib in advanced solid tumors Sources: Pimasertib Investigator Brochure, v12, 2019; de Gooijer et al., Int J Cancer, Day One Biopharmaceuticals 2018; Shaw et
al., AACR LB-456, 2012; Lebbe et al., Cancers, 2020. 28

Vertical MAPK Pathway Inhibition With Tovorafenib (DAY101) And
Pimasertib May Unlock Potential Synergy For Adult Solid Tumors BRAF KRAS or NRAS non-V600 mutant mutant MEK RAF/ PI3K/m RAL MEK/ERK TOR ERK Proliferation, survival Proliferation, survival Type II RAFi + MEKI Type II RAFi + MEKi Pan-RAFi + MEKi is
synergistic in BRAF fusion melanoma PDX model ex vivo (internal data) A Non V600 BRAF dimers are Targeting multiple nodes of effectively inhibited by type II MAPK pathway will drive deeper Sensitivity of KRAS Q61 mutant cells to pimasertib is
enhanced when cells are treated with the B type II BRAF inhibitor BGB-283 (Yuan et al., Mol Onc 2020) RAFi , but not type I BRAFi and more durable response Tovorafenib (DAY101) + MEK inhibitor is synergistic in KRAS G12C and Q61 mutant tumor cells C
(Venetsanakos et al., 2021 AACR poster presentation) Day One Biopharmaceuticals 29

Tovorafenib (DAY101) / Pimasertib Combination To Be Evaluated In Solid
Tumors (FIRELIGHT-1) 1 Trial Design Endpoints 2 • Combination dose escalation, global phase 1b/2 trial • Phase 1b: PK, PD and Safety, MTD/RP2D • Phase 1b, BOIN (adaptive), n = 10/cohort (approximately) • Phase 2: Efficacy
(ORR, DOR) • Phase 2, Simon 2-stage, n = 25/cohort (approximately) • Eligibility: Patients aged 12 years and older, dose escalation will be performed in advanced solid tumor patients with any MAPK alteration. Expansion cohorts will focus
on indications with a potential path to accelerated approval Phase 1b Phase 2* NRASmut Selected tumors Tumors with MAPK BRAF Class 1 (non-E/K) and Safety 3 DAY101 + Pimasertib until disease progression Follow Up Class 2 mutant tumors pathway
alterations BRAF fusion selected tumors Pre-identified patients with advanced *Additional biomarker-selected solid tumors and available clinical cohorts may be pursued based molecular profiling information. on developing data Abbreviations: BOIN,
Bayesian Optimal Interval Design; BRAF, B-Raf proto-oncogene, serine/threonine kinase; MAPK, mitogen-activated protein kinase; NRAS, neuroblastoma rat sarcoma viral oncogene. 1 Umbrella master study – DAY101-102 (main protocol) DAY101 and MAPK
pathway aberration, Sub-study 1 monotherapy (DAY101-102a), Sub-study 2 MEK combo (DAY101-102b). 2 3 Intend to open U.S. and ex-U.S. clinical sties. DAY101 + Pimasertib until disease progression, intolerable toxicity, withdrawal of consent, or death
Day One Biopharmaceuticals 30

Summary Day One Biopharmaceuticals

Financial Summary: DAWN Cash, cash equivalents and short-term
investments ~87.0 million shares of common stock as of June 30, 2023: $442.9 million (no debt) outstanding as of August 1, 2023 Six Months Ended Six Months Ended $ Millions 6/30/23 6/30/22 R&D Expense $60.0 $37.6 G&A Expense $35.1 $26.9 Net
Loss $88.3 $64.3 FIREFLY-1: Pivotal Phase 2 clinical trial of tovorafenib (DAY101) 1 • Initiated rolling NDA in May 2023 Projected • New clinical data presented in June 2023 cash runway 2 • Expected completion of rolling NDA by
October 2023 following submission of an amended clinical study report into 2026 FIREFLY-2/LOGGIC: Pivotal Phase 3 clinical trial of tovorafenib (DAY101) in newly diagnosed pLGG • First patient dosed in March 2023 1 All financial and share
information is unaudited. NDA data set will include analysis of primary (ORR by RANO-HGG) and secondary (ORR by RAPNO, PFS) efficacy endpoints, safety, and exploratory analyses (including ORR by RANO-LGG). 2 Amended clinical study report will
include safety and efficacy data from a planned June 2023 data cutoff. Day One Biopharmaceuticals 32

Next Steps FIREFLY-1 • Initiated rolling NDA in May 2023 •
New clinical data presented in June 2023 • Expected completion of rolling NDA by October 2023 following submission of 1 an amended clinical study report FIREFLY-2 • Advance tovorafenib as a frontline therapy for patients with pLGG
FIRELIGHT-1 • Evaluate tovorafenib in combination and as monotherapy in adolescent and adult populations • Monotherapy abstract presented at EADO in April 2023 Commercial • Continue investment in market and launch preparation
activities Business Development • Further investment in business development activities to expand our multiple asset portfolio 1 Amended clinical study report will include safety and efficacy data from a Day One Biopharmaceuticals 33 planned
June 2023 data cutoff.

Appendix ` 34

Progress Of FIREFLY-1 Program: Monotherapy Tovorafenib In Relapsed pLGG
Updated Clinical Data from FIREFLY-1 Trial Presented at ASCO Pre-NDA Meeting (Jun 2023) Interim Analysis Data (Apr 2023) from FIREFLY-1 Trial (Jun 2022) First Patient Dosed in FIREFLY-1 Trial (May 2021) Initiated Rolling NDA Expected Topline Data
from Submission Completion of FIREFLY-1 Trial Pivotal Cohort (May 2023) Rolling NDA (Jan 2023) Enrollment Complete in Submission FIREFLY-1 Trial (Oct 2023) Initial Discussion (May 2022) with FDA including FIREFLY-1 Trial Design (Apr 2020) Day One
Biopharmaceuticals 35

Case Study: Activity Of Tovorafenib (DAY101) In KIAA1549- BRAF Fusion
Optic Pathway Glioma A 7-years-old female child with an optic pathway glioma, with very poor vision, entropion, folliculitis, eczema, mouth ulceration and xerosis -36 6 Months -30 -24 -18 -12 -6 0 PR Diagnostic biopsy 3L: Tovorafenib 1L:
Vincristine/carboplatin Best response SD 2L: Trametinib Best response PD • PR (-58%) and improvement in vision reported at cycle 3 • AEs included grade 3 erythematous rash requiring dose interruption and dose reduction (400 mg QW to 300
mg QW in cycle 1), and grade 2 eczema and maculopapular rash • Patient continues to receive weekly tovorafenib Baseline After 6 cycles Tumor kinetics (RANO) Tovorafenib Apr 14, 2022 data cutoff. Tovorafenib is an investigational agent. Safety
and efficacy have not been established by any health authority. Day One Biopharmaceuticals 36

Case Study: Activity Of Tovorafenib (DAY101) In KIAA1549-BRAF Fusion
Posterior Fossa Pilocytic Astrocytoma An 8-years-old female child with a posterior fossa pilocytic astrocytoma, eczema, nausea and constipation Months -84 -72 -60 -48 -36 -24 -12 0 12 PR Suboccipital 2L: Carboplatin/vincristine craniotomies 4L:
Trametinib 5L: Tovorafenib Best response PR Best response SD 1L: Carboplatin 3L: Vinblastine Best response PD Best response SD • PR (-69%) at cycle 3 with 500 mg QW tovorafenib, with a deepening of response (80% and 91% in cycles 6 and 9,
respectively) over time • AEs included grade 2 decrease in neutrophil count, pustular rash, and upper respiratory infection • Patient continues to receive weekly tovorafenib Baseline After 3 cycles After 6 cycles After 9 cycles Tumor
kinetics Tovorafenib Apr 14, 2022 data cutoff. Tovorafenib is an investigational agent. Safety and efficacy have not been established by any health authority. Day One Biopharmaceuticals 37

Case Study: Activity Of Tovorafenib (DAY101) In BRAF V600E Mutation
Deep Midline Astrocytoma A 9-year-old female child with deep midline BRAF V600E-mutant astrocytoma with precocious puberty Months -60 -48 -36 -24 -12 0 12 PR Subtotal 1L: Carboplatin/vincristine 2L: Dabrafenib 3L: Tovorafenib resection Best response
PR Best response PR • PR (-74%) at cycle 3, with a deepening of response (-94%) at cycle 6 • AEs included grade 3 maculopapular rash and increased CPK, requiring drug interruption and dose reduction (500 mg QW to 400 mg QW in cycle 1)
• Tovorafenib dose was re-escalated back to 500 mg QW in cycle 4; patient continues on treatment Baseline After 3 cycles After 6 cycles Tumor kinetics Tovorafenib Apr 14, 2022 data cutoff. Tovorafenib is an investigational agent. Safety and
efficacy have not been established by any health authority. Day One Biopharmaceuticals 38

Case Study: Activity Of Tovorafenib (DAY101) In KIAA1549-BRAF Fusion
Optic Pathway Glioma 8-year-old boy with relapsed pilomyxoid astrocytoma of the optic pathway, with visual loss in right eye, visual field loss in left eye, fatigue, intermittent nausea/vomiting, intermittent headaches, anorexia, and temperature
regulation disorder Months -60 0 12 -48 -36 -12 24 -24 Trametinib VC Binimetinib Tovorafenib PR • Initiated treatment with tovorafenib 400 mg/QW following 3 prior therapies, including binimetinib and trametinib, which were discontinued due to
PD • At cycle 3, PR (-88%) per RANO-HGG, and MR (-32% and -40%) per RAPNO-LGG and RANO-LGG, respectively − Sustained improvements in visual acuity reported; logMAR change 0.2 → 0 − PD criteria met (-94% to -91%) with RANO-HGG
at cycle 15; continued treatment as investigator deemed no radiographic progression with subsequent reduction in target lesion (-97%) • AEs were G2 (drug eruption, elevated CPK) and G1 (hair color change, paronychia, growth retardation) T1 + C
(baseline and post-cycle 3) T2 (baseline and post-cycles 3 & 12) Tumor kinetics Dec 22, 2022, data cut-off. AEs, adverse events; C, contrast; CPK, creatine phosphokinase; G, grade; HGG, high-grade glioma; LGG, low-grade glioma; logMAR, Logarithm
of the Minimum Angle of Resolution; MR, minor response; PD, progressive disease; PR, partial response; QW, once weekly; RANO, Response Assessment in Neuro-Oncology; RAPNO, Response Assessment in Pediatric Neuro-Oncology; VC,
vincristine-carboplatin.. Day One Biopharmaceuticals 39

FIREFLY-2/LOGGIC: Pivotal Phase 3 Study Of Tovorafenib (DAY101) In
Newly Diagnosed pLGG • Collaboration between Day One and the Approximately 100 potential sites (~65 from the LOGGIC consortium) LOGGIC consortium, internationally recognized experts in pLGG research ‒ Coupled with the LOGGIC-CORE
molecular diagnostic program ‒ Worked jointly on the study design and ~20 discussions with the U.S. and EU Sites ~65 regulatory authorities Sites ~5 Sites ~10 Sites Day One Biopharmaceuticals 40

Results From Independent Radiology Review Of PNOC014 Best response from
baseline RANO-HGG: Response assessment for neuro- oncology-high grade glioma Volumetric image analysis (exploratory) RAPNO: Response assessment for pediatric neuro-oncology (exploratory) Date of data cutoff: 02 JAN 2020 Wright K et. al. Neuro
Oncology Abstract CTNI-19. 2020 Day One Biopharmaceuticals 41

Multiple Rapid, Deep And Durable Responses Observed Following
Initiation Of Tovorafenib (DAY101) Treatment Of pLGG Patients In PNOC014 Growth kinetics of Target Lesions from Screening Date of data cutoff: 02 JAN 2020 Adapted from Wright K et. al. Neuro Oncology Abstract CTNI-19. 2020 Fangusaro J et al. Lancet
Oncol 2019 Day One Biopharmaceuticals 42

Drug-related Adverse Events Observed for Tovorafenib (DAY101) in
PNOC014 Showed Favorable Safety and Tolerability Profile in pLGG Drug-related AEs for Tovorafenib (DAY101) Drug-related AEs for selumetinib Toxicities Grade 1-2 Grade 3 Grade 4 Toxicities Grade 1-2 Grade 3 Grade 4 Anemia 6 (67%) Increased ALT 20
(40%) 1 (2%) Hypophosphatemia 4 (44%) CPK elevation 34 (68%) 5 (10%) DAY101 AE summary Fatigue 5 (55%) Diarrhea 27 (54%) 2 (4%) Rash 8 (89%) Decreased ejection fraction 19 (38%) 1 (2%) • Most common toxicity: skin Achromotrichia 7 (78%)
Gastric haemorrhage 1 (2%) • AEs reversible and all Pruritis 6 (67%) Headache 14 (28%) 1 (2%) manageable Photosensitivity 1 (11%) Decreased lymphocyte count 19 (38%) 1 (2%) Nevus 7 (78%) Neutropenia 14 (28%) 3 (6%) • Single, reversible
Grade 3 Alopecia 3 (34%) Paronychia 19 (38%) 3 (6%) event Epistaxis 2 (22%) Rash (acneiform) 29 (58%) 2 (4%) • No Grade 4 AEs Dry skin 3 (34%) Rash (maculopapular) 26 (52%) 5 (10%) Myalgias/arthralgias 3 (34%) Skin infection 7 (14%) 1 (2%)
• No dose reductions (vs. Anorexia 2 (22%) Tooth infection 1 (2%) 40% of patients on Cheilitis 3 (34%) Weight gain 5 (10%) 1 (2%) selumetinib montherapy Hypermagnesemia 1 (11%) Vomiting 22 (44%) required dose reductions) Bleeding gums 1 (11%)
Nausea 21 (42%) Increased AST 4 (44%) Increased AST 25 (50%) Nausea/vomiting 3 (33%) Anemia 28 (56%) CPK elevation 1 (11%) Pruritis 10 (20%) Weight loss 2 (22%) Dyspnea 30 (60%) Date of DAY101 data cutoff: 02 JAN 2020; Wright K et. al. Neuro
Oncology Abstract CTNI-19. 2020; Fangusaro J et al. Lancet Oncol 2019 Day One Biopharmaceuticals 43

Preliminary Clinical Activity Of Tovorafenib (DAY101) Monotherapy In
BRAF Fusion Melanoma Patient 1: 53-year-old male with AGK-BRAF fusion non-spitzoid cutaneous melanoma Parameter Description/outcome Stage at diagnosis III EGOC status 0 • Multiple lymphadenectomies and skin lesion excision surgery Prior
therapies • Pembrolizumab (11 weeks): ‒ Best response: SD • 600 mg QW Tovorafenib treatment to date in • 5 cycles with no dose interruption or modifications FL-1 102a (melanoma cohort)* due to AEs † ‡ Antitumor
activity results to date* • CR (11-week scan) ; confirmed at 16 weeks • TRAEs: ‒ Transient rash (G1 and G2) Safety results to date* ‒ Anemia (G2) • TEAE: ‒ Neck pain (G1) *Data cutoff Feb 8, 2023. †Out of
window per protocol. ‡per RECIST v1.1. AE, adverse event; CR, complete response; ECOG, Eastern Cooperative Oncology Group; FL-1, FIRELIGHT-1; G, grade; QW, once weekly; RECIST, response evaluation criteria in solid tumors; SD, stable disease;
TEAE, treatment-emergent adverse event; TRAEs, treatment-related adverse events; y/o, years of age. Day One Biopharmaceuticals 44

Preliminary Clinical Activity Of Tovorafenib (DAY101) Monotherapy In
BRAF Fusion Melanoma Patient 2: 35-year-old male with TRIM33-BRAF fusion malignant melanoma Parameter Description/outcome Stage at diagnosis Unknown EGOC status 1 • Radiation • Nivolumab (12 mo, adjuvant setting): Prior therapies ‒
No best response, disease resected • Nivolumab + ipilimumab (3 cycles): ‒ Best response: PD after 2 mo • 600 mg QW Tovorafenib treatment to date in • 5 cycles with no dose interruption or FL-1 102a (melanoma cohort)*
modifications due to AEs Antitumor activity results to † • PR (8-week scan); confirmed at 16 weeks date* • TRAEs: ‒ Rash - maculopapular (G1) Safety results to date* ‒ Headache (G1) ‒ Fatigue (G1) *Data cutoff Feb
8, 2023. †per RECIST v1.1. AE, adverse event; ECOG, Eastern Cooperative Oncology Group; FL-1, FIRELIGHT-1; G, grade; mo, months; PD, progressive disease; PR, partial response; QW, once weekly; RECIST, response evaluation criteria in solid
tumors; TRAEs, treatment-related adverse events; y/o, years of age. Day One Biopharmaceuticals 45

Preliminary Clinical Activity Of Tovorafenib (DAY101) Monotherapy In
BRAF Fusion Melanoma Patient 3: 71-year-old male with MKRN1-BRAF fusion non-spitzoid cutaneous melanoma Parameter Description/outcome Stage at diagnosis II EGOC status 0 • Radiation Prior therapies • Pembrolizumab (2 mo): ‒ Best
response: SD • 600 mg QW Tovorafenib treatment to date in • 3 cycles with no dose interruption or modifications FL-1 102a (melanoma cohort)* due to AEs Antitumor activity results to • PR (7-week scan)†,‡; is awaiting a
confirmatory scan date* • TRAEs: Safety results to date* ‒ Urticaria (G1) ‒ Hand-foot syndrome (G1) *Data cutoff Feb 8, 2023. †In window per protocol. ‡per RECIST v1.1. AE, adverse event; ECOG, Eastern Cooperative
Oncology Group; FL-1, FIRELIGHT-1; G, grade; mo, months; PR, partial response; QW, once weekly; RECIST, response evaluation criteria in solid tumors; SD, stable disease; TRAEs, treatment-related adverse events; y/o, years of age. Day One
Biopharmaceuticals 46
v3.23.2
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Day One Biopharmaceuticals (NASDAQ:DAWN)
Historical Stock Chart
From Apr 2024 to May 2024
Day One Biopharmaceuticals (NASDAQ:DAWN)
Historical Stock Chart
From May 2023 to May 2024