- Starlight Therapeutics Inc., a Lantern subsidiary, will focus
exclusively on the clinical development of therapies for CNS
(central nervous system) and brain cancers with limited or no
effective therapeutic options.
- Lantern's AI platform, RADR®, aided in the identification and
accelerated development of Starlight's portfolio of therapeutic
indications.
- The leading drug candidate, STAR-001, has demonstrated
blood-brain barrier permeability, has favorable brain tumor
bioavailability, and has shown nanomolar potency across an
extensive number of in-vitro and in-vivo CNS and brain cancer
models.
- Clinical trials in adult and pediatric CNS cancer indications
are anticipated for late 2023 and early 2024.
- Globally, STAR-001’s targeted treatment indications represent
an anticipated 500,000+ new cases each year and have a combined
estimated annual market potential of over $6 billion (USD).
Lantern Pharma Inc. (NASDAQ: LTRN), a clinical-stage
biopharmaceutical company using its proprietary RADR® artificial
intelligence (AI) and machine learning (ML) platform to transform
the cost, pace, and timeline of oncology drug discovery and
development, today announced that it has formed a wholly-owned
subsidiary, Starlight Therapeutics Inc. (“Starlight”), to develop
drug candidate LP-184’s central nervous system (CNS) and brain
cancer indications – including glioblastoma (GBM), brain metastases
(brain mets.), and several rare pediatric CNS cancers. Starlight
will refer to the molecule LP-184, as it is developed in CNS
indications, as “STAR-001”. Combined, STAR-001’s targeted treatment
indications are estimated to represent an annual global market
potential of approximately $6.0 billion (USD) and over 500,000
global cases each year.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20230306005304/en/
Starlight Therapeutics Pipeline of Adult
and Pediatric CNS Cancer Indications (Graphic: Business Wire)
Starlight plans on establishing a leading CNS oncology franchise
to develop the unique drug candidate STAR-001 for some of the most
malignant and unaddressed primary and secondary CNS cancers. The
programs being developed by Starlight were born from the analysis
of billions of oncology-focused data points and by using Lantern’s
AI platform, RADR®. STAR-001’s powerful anti-tumor mechanism of
action, synthetic lethality, and collaborations with
internationally recognized institutions, including the Kennedy
Krieger Institute at Johns Hopkins and the Greehey Children’s
Cancer Research Institute at UT Health – San Antonio, make it well
positioned to advance in targeted and efficient clinical
development programs. Starlight intends to pursue human clinical
trials for multiple CNS indications starting in late 2023, building
on prior IND-enabling studies and the Phase 1 clinical testing that
will be conducted by Lantern.
“The formation of Starlight allows Lantern to put extreme focus
on advancing STAR-001 through targeted clinical trials and dedicate
increased time, resources, and personnel to progress one of the
most promising drug candidates for CNS cancer patients in decades,”
stated Panna Sharma, Lantern’s CEO and President. “Additionally,
establishing Starlight as a wholly-owned subsidiary will increase
the potential to partner with other biopharma companies who are
looking to develop a franchise in CNS cancers and to further
accelerate the progression of STAR-001 towards changing patient
outcomes in this devastating set of diseases,” continued Sharma.
“Since our initial discovery, in which we used large-scale,
multi-omic network analysis from our RADR® AI platform, to validate
that GBM was sensitive to LP-184 and that LP-184 had excellent
blood-brain barrier permeability, our team has found, validated,
and published on several additional pediatric and adult brain
cancers that have shown early promise to LP-184, now STAR-001, in
CNS cancers,” stated Sharma. “Our portfolio of opportunities and
pipeline in CNS cancers has grown five-fold and includes multiple
indications lacking any accepted standard-of-care. We believe that
by focusing our efforts via Starlight Therapeutics we can
accelerate and deepen our commitment to the CNS cancer patient
community, while also creating the potential for meaningful
additional upside for our investors,” concluded Sharma.
STAR-001 is a unique blood-brain barrier permeable small
molecule that utilizes its powerful mechanism of action, synthetic
lethality, to exploit common vulnerabilities in CNS cancers with
DNA damage repair (DDR) deficiencies. The anti-tumor potential of
STAR-001 has been demonstrated across an extensive number of
in-vitro and in-vivo CNS cancer models, including GBM, brain mets.,
and atypical teratoid rhabdoid tumors (ATRT), and has been
presented at leading conferences and publications including, the
Society for Neuro-Oncology annual meeting, the American Association
for Cancer Research annual meeting, and the Frontiers in Drug
Discovery Journal. Highlights of STAR-001’s promising preclinical
results from these presentations and publications are included
below:
- Pharmacokinetic studies have shown STAR-001 to have 2X the
bioavailability in brain tumors, compared to normal brain tissue,
and to have 2X the bioavailability in brain tumors compared to the
bioavailability of temozolomide (TMZ), the GBM standard-of-care
(SOC) agent. Additional details from these experiments can be found
here.
- In mice implanted with subcutaneous GBM cell-derived xenograft
(CDX) tumors from models of the two major GBM subtypes, known as
MGMT methylated and MGMT unmethylated, STAR-001 treatment resulted
in 107% tumor growth inhibition in both tumor types. In these
experiments, 75% of mice with tumors from the MGMT unmethylated GBM
model and 30% of mice with tumors from the MGMT methylated GBM
model were entirely tumor-free after STAR-001 treatment. Additional
details from these experiments can be found here.
- STAR-001 has the potential for combination with the
FDA-approved agent spironolactone to enhance STAR-001’s anti-tumor
potency. In in-vitro GBM models with different MGMT methylation
states, STAR-001 treatment with spironolactone significantly
decreased STAR-001’s IC50 by 3-6X, resulting in IC50s in the low
nanomolar range of 34-94nM. Additional details from these
experiments can be found here.
- STAR-001 has been demonstrated to have nanomolar potency in
brain mets. cell lines that originated from non-small cell lung
cancer, melanoma, and breast cancer (HER2- and Triple Negative).
Additional details from these experiments can be found here.
- STAR-001 treatment of mice implanted with ATRT CDX tumors, at
either 2 mg/kg or 4 mg/kg doses (I.V.), showed near complete tumor
growth inhibition between 82-91%, respectively. Additional details
from these experiments can be found here.
Based on STAR-001’s demonstrated anti-tumor potential in CNS
cancers, the FDA has granted STAR-001 Orphan Drug Designations
(ODD) for malignant gliomas (including GBM) and ATRT. Additionally,
STAR-001 was granted a Rare Pediatric Disease Designation for ATRT,
which occurs in 60-70 pediatric patients a year in the US.
Starlight’s clinical development strategy will initially focus
on progressing STAR-001 through early-stage clinical trials for
adult recurrent high-grade gliomas (HGGs), including GBM. There
have been no effective single-agent treatment options approved for
GBM in nearly two decades. The current GBM standard-of-care agent,
TMZ, is ineffective in MGMT unmethylated patients, who represent
over 65% of all GBM patients. Starlight is planning several
additional clinical programs for STAR-001 including adult and
pediatric CNS cancers and combination regimens.
Commencing in mid-2023, Lantern is anticipating a Phase 1A
basket trial for LP-184 (STAR-001), in a range of solid tumors
including: recurrent brain cancers (including GBM and HGGs),
metastatic CNS cancers (brain mets.), pancreatic cancer, and solid
tumors with DDR deficiencies. The clinical development of STAR-001
in CNS cancers beyond the Phase 1A trial will be conducted
exclusively by Starlight. Following the launch of Starlight,
Lantern will continue to advance LP-184’s preclinical and clinical
development for non-CNS indications (including pancreatic cancer
and other solid tumors) and will also provide AI-driven
bioinformatic and computational biology support to Starlight.
About Lantern Pharma:
Lantern Pharma (NASDAQ: LTRN) is a clinical-stage
oncology-focused biopharmaceutical company leveraging its
proprietary RADR® AI and machine learning platform to discover
biomarker signatures that identify patients most likely to respond
to its pipeline of genomically-targeted therapeutics. By targeting
drugs to patients whose genomic profile identifies them as having
the highest probability of benefiting from the drug, Lantern's
approach represents the potential to deliver best-in-class
outcomes.
Forward-looking Statements:
This press release contains forward-looking statements within
the meaning of Section 27A of the Securities Act of 1933, as
amended, and Section 21E of the Securities Exchange Act of 1934, as
amended. These forward-looking statements include, among other
things, statements relating to: the potential advantages of
STAR-001; the potential advantages of our RADR® platform in
identifying drug candidates and patient populations that are likely
to respond to a drug candidate; our strategic plans to advance the
clinical development of STAR-001; and expectations and estimates
regarding clinical trial timing. Any statements that are not
statements of historical fact (including, without limitation,
statements that use words such as "anticipate," "believe,"
"contemplate," "could," "estimate," "expect," "intend," "seek,"
"may," "might," "plan," "potential," "predict," "project,"
"target," "model," "objective," "aim," "upcoming," "should,"
"will," "would," or the negative of these words or other similar
expressions) should be considered forward-looking statements. There
are a number of important factors that could cause our actual
results to differ materially from those indicated by the
forward-looking statements, such as (i) we have not commenced human
clinical trials for any of the indications to be addressed by
STAR-001, (ii) the risk that the results of our clinical trials for
STAR-001 may not be successful or warrant further development,
(iii) the risk that success in early phases of pre-clinical and
clinicals trials does not ensure later clinical trials will be
successful, (iv) our ability to fund the clinical trials and
further development of STAR-001 and other product candidates and
the availability of capital if and when needed, (v) the risk that
none of our product candidates has received FDA marketing approval,
and we may not be able to successfully initiate, conduct, or
conclude clinical testing for or obtain marketing approval for our
product candidates, (vi) the risk that no drug product based on our
proprietary RADR® AI platform has received FDA marketing approval
or otherwise been incorporated into a commercial product, and (vii)
those other factors set forth in the Risk Factors section in our
Annual Report on Form 10-K for the year ended December 31, 2021,
filed with the Securities and Exchange Commission on March 10,
2022. You may access our Annual Report on Form 10-K for the year
ended December 31, 2021 under the investor SEC filings tab of our
website at www.lanternpharma.com or on the SEC's website at
www.sec.gov. Given these risks and uncertainties, we can give no
assurances that our forward-looking statements will prove to be
accurate, or that any other results or events projected or
contemplated by our forward-looking statements will in fact occur,
and we caution investors not to place undue reliance on these
statements. All forward-looking statements in this press release
represent our judgment as of the date hereof, and, except as
otherwise required by law, we disclaim any obligation to update any
forward-looking statements to conform the statement to actual
results or changes in our expectations.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20230306005304/en/
Drew Sturtevant, Ph.D. Sr. Associate, Scientific Affairs and
Communications contact@starlightthera.com
Please find more information at: Lantern Pharma’s Website:
www.lanternpharma.com LinkedIn:
https://www.linkedin.com/company/lanternpharma/ Lantern Pharma’s
Monthly Newsletter – SPARK: Sign-up here Twitter:
@lanternpharma
Lantern Pharma (NASDAQ:LTRN)
Historical Stock Chart
From Jun 2024 to Jul 2024
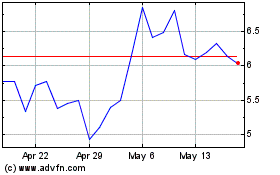
Lantern Pharma (NASDAQ:LTRN)
Historical Stock Chart
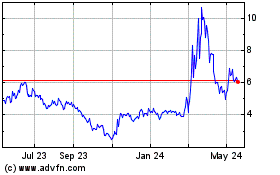
From Jul 2023 to Jul 2024