Avidity Biosciences: FDA Places Partial Hold on AOC 1001 Study
September 27 2022 - 7:54AM
Dow Jones News
By Colin Kellaher
Avidity Biosciences Inc. on Tuesday said the U.S. Food and Drug
Administration placed a partial clinical hold on new enrollment in
a Phase 1/2 study of its lead product candidate after one
participant suffered a serious adverse event.
The San Diego biopharmaceutical company said participants
already enrolled in the study of AOC 1001 in adults with the
genetic disorder myotonic dystrophy type 1 can continue in their
current dosing cohort, but that it can't add any more participants
until the partial hold is resolved.
Avidity said it is working closely with the FDA and the trial's
investigator to assess the cause of the adverse event, adding that
it is taking all necessary steps to resolve the partial hold on new
study enrollment as quickly as possible.
The company said it is on track to conduct a preliminary
assessment of safety, tolerability and key biomarkers in roughly
half of the study's participants in the fourth quarter.
There are currently no approved treatment options for myotonic
dystrophy type 1, a progressive and often fatal neuromuscular
disease.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
September 27, 2022 07:39 ET (11:39 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
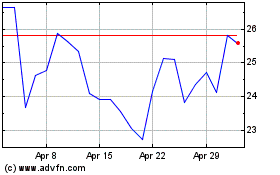
Avidity Biosciences (NASDAQ:RNA)
Historical Stock Chart
From Aug 2024 to Sep 2024
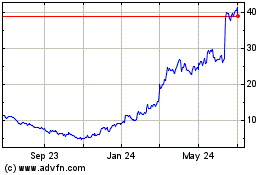
Avidity Biosciences (NASDAQ:RNA)
Historical Stock Chart
From Sep 2023 to Sep 2024