0001602078
false
0001602078
2022-04-01
2022-12-31
0001602078
dei:BusinessContactMember
2022-04-01
2022-12-31
0001602078
2022-03-31
0001602078
2021-03-31
0001602078
2022-12-31
0001602078
2021-04-01
2022-03-31
0001602078
2020-04-01
2021-03-31
0001602078
2022-10-01
2022-12-31
0001602078
2021-10-01
2021-12-31
0001602078
2021-04-01
2021-12-31
0001602078
us-gaap:CommonStockMember
2020-03-31
0001602078
us-gaap:AdditionalPaidInCapitalMember
2020-03-31
0001602078
us-gaap:RetainedEarningsMember
2020-03-31
0001602078
us-gaap:AccumulatedOtherComprehensiveIncomeMember
2020-03-31
0001602078
2020-03-31
0001602078
us-gaap:CommonStockMember
2021-03-31
0001602078
us-gaap:AdditionalPaidInCapitalMember
2021-03-31
0001602078
us-gaap:RetainedEarningsMember
2021-03-31
0001602078
us-gaap:AccumulatedOtherComprehensiveIncomeMember
2021-03-31
0001602078
us-gaap:CommonStockMember
2022-09-30
0001602078
us-gaap:AdditionalPaidInCapitalMember
2022-09-30
0001602078
us-gaap:RetainedEarningsMember
2022-09-30
0001602078
us-gaap:AccumulatedOtherComprehensiveIncomeMember
2022-09-30
0001602078
2022-09-30
0001602078
us-gaap:CommonStockMember
2021-09-30
0001602078
us-gaap:AdditionalPaidInCapitalMember
2021-09-30
0001602078
us-gaap:RetainedEarningsMember
2021-09-30
0001602078
us-gaap:AccumulatedOtherComprehensiveIncomeMember
2021-09-30
0001602078
2021-09-30
0001602078
us-gaap:CommonStockMember
2022-03-31
0001602078
us-gaap:AdditionalPaidInCapitalMember
2022-03-31
0001602078
us-gaap:RetainedEarningsMember
2022-03-31
0001602078
us-gaap:AccumulatedOtherComprehensiveIncomeMember
2022-03-31
0001602078
us-gaap:CommonStockMember
2020-04-01
2021-03-31
0001602078
us-gaap:AdditionalPaidInCapitalMember
2020-04-01
2021-03-31
0001602078
us-gaap:RetainedEarningsMember
2020-04-01
2021-03-31
0001602078
us-gaap:AccumulatedOtherComprehensiveIncomeMember
2020-04-01
2021-03-31
0001602078
us-gaap:CommonStockMember
2021-04-01
2022-03-31
0001602078
us-gaap:AdditionalPaidInCapitalMember
2021-04-01
2022-03-31
0001602078
us-gaap:RetainedEarningsMember
2021-04-01
2022-03-31
0001602078
us-gaap:AccumulatedOtherComprehensiveIncomeMember
2021-04-01
2022-03-31
0001602078
us-gaap:CommonStockMember
2022-10-01
2022-12-31
0001602078
us-gaap:AdditionalPaidInCapitalMember
2022-10-01
2022-12-31
0001602078
us-gaap:RetainedEarningsMember
2022-10-01
2022-12-31
0001602078
us-gaap:AccumulatedOtherComprehensiveIncomeMember
2022-10-01
2022-12-31
0001602078
us-gaap:CommonStockMember
2021-10-01
2021-12-31
0001602078
us-gaap:AdditionalPaidInCapitalMember
2021-10-01
2021-12-31
0001602078
us-gaap:RetainedEarningsMember
2021-10-01
2021-12-31
0001602078
us-gaap:AccumulatedOtherComprehensiveIncomeMember
2021-10-01
2021-12-31
0001602078
us-gaap:CommonStockMember
2022-04-01
2022-12-31
0001602078
us-gaap:AdditionalPaidInCapitalMember
2022-04-01
2022-12-31
0001602078
us-gaap:RetainedEarningsMember
2022-04-01
2022-12-31
0001602078
us-gaap:AccumulatedOtherComprehensiveIncomeMember
2022-04-01
2022-12-31
0001602078
us-gaap:CommonStockMember
2021-04-01
2021-12-31
0001602078
us-gaap:AdditionalPaidInCapitalMember
2021-04-01
2021-12-31
0001602078
us-gaap:RetainedEarningsMember
2021-04-01
2021-12-31
0001602078
us-gaap:AccumulatedOtherComprehensiveIncomeMember
2021-04-01
2021-12-31
0001602078
us-gaap:CommonStockMember
2022-12-31
0001602078
us-gaap:AdditionalPaidInCapitalMember
2022-12-31
0001602078
us-gaap:RetainedEarningsMember
2022-12-31
0001602078
us-gaap:AccumulatedOtherComprehensiveIncomeMember
2022-12-31
0001602078
us-gaap:CommonStockMember
2021-12-31
0001602078
us-gaap:AdditionalPaidInCapitalMember
2021-12-31
0001602078
us-gaap:RetainedEarningsMember
2021-12-31
0001602078
us-gaap:AccumulatedOtherComprehensiveIncomeMember
2021-12-31
0001602078
2021-12-31
0001602078
nmrd:WarrantsMember
2021-04-01
2022-03-31
0001602078
us-gaap:OptionMember
2021-04-01
2022-03-31
0001602078
nmrd:OptionOneMember
2021-04-01
2022-03-31
0001602078
nmrd:WarrantsOneMember
2021-04-01
2022-03-31
0001602078
nmrd:WarrantsMember
2020-04-01
2021-03-31
0001602078
us-gaap:OptionMember
2020-04-01
2021-03-31
0001602078
nmrd:WarrantsOneMember
2020-04-01
2021-03-31
0001602078
2020-05-31
0001602078
2021-06-30
0001602078
us-gaap:ForwardContractsMember
2021-04-01
2022-03-31
0001602078
2014-03-31
0001602078
nmrd:MarketingRightsAgreementMember
2014-03-31
0001602078
nmrd:MarketingRightsAgreementMember
2022-12-31
0001602078
nmrd:MarketingRightsAgreementMember
2022-03-31
0001602078
nmrd:NotePurchaseAgreementMember
us-gaap:InvestorMember
nmrd:SecuredNoteMember
2020-04-15
0001602078
nmrd:NotePurchaseAgreementMember
us-gaap:InvestorMember
nmrd:SecuredNoteMember
2020-04-01
2020-04-15
0001602078
nmrd:NotePurchaseAgreementMember
us-gaap:InvestorMember
nmrd:InvestorNote1Member
2020-04-15
0001602078
nmrd:NotePurchaseAgreementMember
us-gaap:InvestorMember
nmrd:InvestorNote2Member
2020-04-15
0001602078
nmrd:NotePurchaseAgreement2Member
us-gaap:InvestorMember
nmrd:SecuredNoteMember
2021-02-08
0001602078
nmrd:NotePurchaseAgreement2Member
us-gaap:InvestorMember
nmrd:SecuredNoteMember
2021-02-01
2021-02-08
0001602078
nmrd:NotePurchaseAgreement2Member
us-gaap:InvestorMember
nmrd:SecuredNoteMember
2021-02-01
2021-02-09
0001602078
nmrd:NotePurchaseAgreement1Member
us-gaap:InvestorMember
nmrd:SecuredNoteMember
2020-04-15
0001602078
2021-02-01
2021-02-08
0001602078
nmrd:AscendiantCapitalPartnersLLCMember
2021-02-01
2021-02-08
0001602078
nmrd:NotePurchaseAgreement2Member
2022-10-01
2022-10-21
0001602078
us-gaap:InvestorMember
2022-10-21
0001602078
2022-06-01
2022-06-30
0001602078
us-gaap:SubsequentEventMember
2023-02-01
2023-02-23
0001602078
us-gaap:SubsequentEventMember
2023-03-01
2023-03-31
0001602078
us-gaap:SubsequentEventMember
nmrd:NotePurchaseAgreement2Member
2023-03-01
2023-03-31
0001602078
nmrd:NotePurchaseAgreement2Member
2022-06-01
2022-06-30
0001602078
nmrd:SecuredNoteMember
us-gaap:InvestorMember
nmrd:NotePurchaseAgreement3Member
2022-05-20
0001602078
nmrd:SecuredNoteMember
us-gaap:InvestorMember
nmrd:NotePurchaseAgreement3Member
2022-05-01
2022-05-20
0001602078
country:US
2021-04-01
2022-03-31
0001602078
country:US
2020-04-01
2021-03-31
0001602078
country:GB
2021-04-01
2022-03-31
0001602078
country:GB
2020-04-01
2021-03-31
0001602078
country:US
2022-03-31
0001602078
country:GB
2022-03-31
0001602078
2022-03-24
0001602078
nmrd:AtTheMarketofferingAgreementMember
2021-04-01
2022-03-31
0001602078
nmrd:AtTheMarketofferingAgreementMember
2022-03-31
0001602078
nmrd:TigerManagementLLCMember
2021-04-01
2022-03-31
0001602078
nmrd:TigerManagementLLCMember
2022-03-31
0001602078
2018-10-01
2018-10-19
0001602078
nmrd:DistributionAgreementMember
2020-04-01
2021-03-31
0001602078
nmrd:PlacementAgencyAgreementMember
us-gaap:OptionMember
2018-12-02
2018-12-18
0001602078
nmrd:PlacementAgencyAgreementMember
nmrd:WarrantsOneMember
2018-12-02
2018-12-18
0001602078
nmrd:PlacementAgencyAgreementMember
2018-12-18
0001602078
nmrd:PlacementAgencyAgreementMember
2018-12-02
2018-12-20
0001602078
nmrd:PlacementAgencyAgreementMember
2018-12-20
0001602078
nmrd:PlacementAgencyAgreementMember
nmrd:KingswoodMember
2020-07-01
2020-07-30
0001602078
nmrd:PlacementAgencyAgreementMember
2020-07-01
2020-07-30
0001602078
nmrd:PlacementAgencyAgreementMember
nmrd:KingswoodMember
2020-07-30
0001602078
nmrd:PlacementAgencyAgreementMember
nmrd:KingswoodMember
2021-04-01
2022-03-31
0001602078
nmrd:PlacementAgencyAgreementMember
nmrd:KingswoodMember
2022-03-31
0001602078
srt:DirectorMember
us-gaap:StockOptionMember
2022-01-28
0001602078
us-gaap:StockOptionMember
2021-03-31
0001602078
us-gaap:StockOptionMember
2021-04-01
2022-03-31
0001602078
us-gaap:StockOptionMember
2022-03-31
0001602078
nmrd:InvestorRelationsAgreementsMember
2021-04-01
2022-03-31
0001602078
nmrd:InvestorRelationsAgreementsMember
2020-04-01
2021-03-31
0001602078
us-gaap:SubsequentEventMember
2022-04-02
0001602078
us-gaap:SubsequentEventMember
2022-05-20
0001602078
us-gaap:SubsequentEventMember
2022-05-01
2022-05-20
0001602078
us-gaap:SubsequentEventMember
nmrd:SecuredNoteMember
2022-05-01
2022-05-20
0001602078
nmrd:ConcurrentPrivatePlacementMember
us-gaap:SubsequentEventMember
2023-01-01
2023-01-27
0001602078
nmrd:ConcurrentPrivatePlacementMember
us-gaap:SubsequentEventMember
2023-01-27
0001602078
us-gaap:PrivatePlacementMember
us-gaap:SubsequentEventMember
2023-01-31
0001602078
us-gaap:WarrantMember
2022-10-01
2022-12-31
0001602078
us-gaap:WarrantMember
2022-04-01
2022-12-31
0001602078
us-gaap:WarrantMember
2021-10-01
2021-12-31
0001602078
us-gaap:WarrantMember
2021-04-01
2021-12-31
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
xbrli:pure
iso4217:GBP
As filed with the Securities and Exchange Commission
on March 13, 2023
Registration No. 333-
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, DC 20549
FORM S-1
REGISTRATION STATEMENT
UNDER THE SECURITIES ACT OF 1933
Nemaura Medical, Inc.
(Exact name of registrant as specified in its charter)
| Nevada |
|
3841 |
|
46-5027260 |
| (State or Other Jurisdiction of |
|
(Primary Standard Industrial |
|
(I.R.S. Employer |
| Incorporation or Organization) |
|
Classification Code Number) |
|
Identification Number) |
57 West 57th Street
New York, NY 10019
(646) 416-8000
(Address, including zip code, and telephone number,
including area code, of registrant’s principal executive offices)
Corporate Creations Network Inc.
8275 South Eastern Avenue #200
Las Vegas, Nevada 89123
(702) 951-9324
(Name, address, including zip code, and telephone number,
including area code, of agent for service)
Copies to:
Laura Anthony, Esq.
Craig D. Linder, Esq.
Anthony L.G., PLLC
625 N. Flagler Drive, Suite 600
West Palm Beach, FL 33401
(561) 514-0936
Approximate date of commencement
of proposed sale to the public: As soon as practicable after the effective date of this registration statement.
If any of the securities
being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933,
check the following box: ☒
If this Form is filed to
register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list
the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective
amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement
number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective
amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement
number of the earlier effective registration statement for the same offering. ☐
Indicate by check mark whether
the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging
growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting
company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer ☐ |
|
Accelerated filer ☐ |
| Non-Accelerated filer ☒ |
|
Smaller reporting company ☒ |
| |
|
Emerging growth company ☐ |
If an emerging growth company,
indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial
accounting standards provided pursuant to Section 7(a)(2)(B) of the Securities Act. ☐
The registrant hereby
amends this registration statement on such date or dates as may be necessary to delay its effective date until the registrant shall file
a further amendment which specifically states that this registration statement shall thereafter become effective in accordance with section
8(a) of the Securities Act of 1933 or until the registration statement shall become effective on such date as the commission, acting pursuant
to section 8(a) may determine.
The information
in this preliminary prospectus is not complete and may be changed. The selling stockholders may not sell these securities until the registration
statement filed with the Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell these securities,
and it is not soliciting an offer to buy these securities, in any jurisdiction where the offer or sale is not permitted.
| PRELIMINARY PROSPECTUS |
|
SUBJECT TO COMPLETION |
|
DATED MARCH 13, 2023 |
Nemaura Medical, Inc.

4,796,206
Shares of Common Stock
__________________
This prospectus covers the sale of an aggregate of
4,796,206 shares (the “shares”) of our common stock, $0.001 par value per share (the “common stock”), by two selling
stockholders identified in this prospectus (collectively with any of the holder’s transferees, pledgees, donees or successors, the
“selling stockholders”). The shares are issuable upon the exercise of warrants (the “warrants”) purchased by the
selling stockholders in a private placement transaction exempt from registration under Section 4(a)(2) of the Securities Act of 1933,
as amended (the “Securities Act”), pursuant to a Securities Purchase Agreement dated January 27, 2023 (the “Purchase
Agreement”). Each warrant will entitle the holder to purchase our common stock at an exercise price of $2.00 per share. We are registering
the resale of the shares of common stock covered by this prospectus as required by the Purchase Agreement.
The Company will not receive any proceeds from the
sale by the selling stockholders of the shares, however, we will receive proceeds from the exercise of the warrants if the warrants are
exercised for cash. We intend to use those proceeds, if any, for general corporate purposes. We are paying the cost of registering the
shares covered by this prospectus as well as various related expenses, including with regard to compliance with state securities or “blue
sky” laws. The selling stockholders are responsible for all selling commissions, transfer taxes and other costs related to the offer
and sale of the shares.
Sales of the shares by the selling stockholders may
occur at fixed prices, at market prices prevailing at the time of sale, at prices related to prevailing market prices, or at negotiated
prices. The selling stockholders may sell shares to or through underwriters, broker-dealers or agents, who may receive compensation in
the form of discounts, concessions or commissions from the selling stockholders, the purchasers of the shares, or both. If required, the
number of shares to be sold, the public offering price of those shares, the names of any underwriters, broker-dealers or agents and any
applicable commission or discount will be included in a supplement to this prospectus, called a prospectus supplement. Because all of
the shares offered under this prospectus are being offered by the selling stockholders, we cannot currently determine the price or prices
at which the shares may be sold under this prospectus.
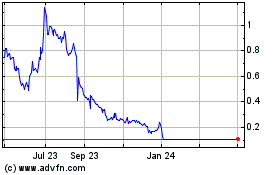
Our common stock is currently quoted on the Nasdaq
Capital Market under the symbol “NMRD”. On March 9, 2023 the last reported sale price per share of our common stock on the
Nasdaq Capital Market was $0.9068 You are urged to obtain current market quotations for our common stock.
Our principal executive offices are located at 57
West 57th Street, New York, NY 10019.
Investing in our securities involves risks.
You should carefully consider the Risk Factors beginning on page 22 of this prospectus before you make an investment
in our securities.
Neither the Securities and Exchange Commission
nor any state securities commission has approved or disapproved of these securities or determined if this prospectus is truthful or complete.
Any representation to the contrary is a criminal offense.
The date of this prospectus
is ____________, 2023.
TABLE OF CONTENTS
No dealer, salesperson or other individual has
been authorized to give any information or to make any representation other than those contained in this prospectus in connection with
the offer made by this prospectus and, if given or made, such information or representations must not be relied upon as having been authorized
by us or the selling stockholders. This prospectus does not constitute an offer to sell or a solicitation of an offer to buy any securities
in any jurisdiction in which such an offer or solicitation is not authorized or in which the person making such offer or solicitation
is not qualified to do so, or to any person to whom it is unlawful to make such offer or solicitation. Neither the delivery of this prospectus
nor any sale made hereunder shall, under any circumstances, create any implication that there has been no change in our affairs or that
information contained herein is correct as of any time subsequent to the date hereof.
For investors outside the United States: We have
not and the selling stockholders have not done anything that would permit this offering or possession or distribution of this prospectus
in any jurisdiction where action for that purpose is required, other than in the United States. Persons outside the United States who
come into possession of this prospectus must inform themselves, and observe any restrictions relating to, the offering of the shares of
our common stock and the distribution of this prospectus outside the United States.
PROSPECTUS SUMMARY
This summary highlights certain information
about us, this offering, and selected information contained in this prospectus. This summary is not complete and does not contain all
of the information that you should consider before deciding whether to invest in our common stock. For a more complete understanding of
the Company and this offering, we encourage you to read and consider the more detailed information in this prospectus, including “Risk
Factors” and the financial statements and related notes. Unless the context requires otherwise, references to “Company,”
“we,” “us” or “our” refer to Nemaura Medical, Inc., a Nevada corporation and its subsidiaries.
Business
Overview
We
are a medical technology company developing sugarBEAT®, a non-invasive glucose monitoring
system for adjunctive use by persons with diabetes, as well as for consumers in the non-diabetes space for monitoring metabolic health
and general health and wellness. sugarBEAT® consists of a disposable adhesive skin-patch
connected to a rechargeable wireless transmitter that displays glucose readings at regular five-minute intervals via a mobile app. sugarBEAT®
works by extracting glucose from the skin into a chamber in the patch that is in direct contact with an electrode-based sensor. The transmitter
sends the raw data to a mobile app where it is processed by an algorithm and displayed as a glucose reading after calibration, with the
ability to track and trend the data over 14 hours for each sensor wear period, including an initial warm-up period of 60-90 minutes on
average. Sensors can be worn as frequently as a user chooses. While sugarBEAT® requires a
minimum once per day calibration by the patient using a blood sample obtained by a finger stick, we believe sugarBEAT®
may be adopted by non-insulin dependent persons with diabetes alongside insulin-injecting persons with diabetes, who all perform multiple
daily finger sticks to manage their disease. In the consumer application, or non-medical applications, the relative glucose fluctuations
are used and therefore finger-prick calibrations are not required.
CE approval was granted
by the European Notified Body BSI in May 2019, allowing the product to be made available for commercial sale. This approval is subject
to an annual review of the underlying ISO 13485 accredited Quality Management System. The accreditation was successfully renewed in November
2021 and 2022. In conjunction with the UK Licensee, the Company commenced a phase 1 launch whereby devices were made available to limited
cohorts of users to gauge their feedback so that any fine-tuning could be completed prior to a mass market launch. The UK Licensee has
also confirmed that it plans to undertake Key Opinion Leader (“KOL”) studies in the UK for its white-labelled service offering
that is supported by sugarBEAT®. The KOL studies are intended to provide additional support
for the UK Licensee’s broader ongoing marketing plans and more specifically to support potential reimbursement for the device and
the licensees overall diabetes management offering(s)
The UK Licensee placed
an initial order for sugarBEAT® in April 2021 and provided a forecast for its post-launch
volume expectations, which the Company has used to establish both a short and medium term view to inform the Company’s commercial
operational requirements. In line with this view, the Company has taken the following actions since the initial order was received:
| |
· |
Entered into a new leased facility to provide the additional space requirements for commercial product assembly. |
| |
· |
Increased headcount of production operatives; this will be phased in line with the volume forecasts currently available, however the Company has also factored in an ability to scale further and faster should this be required. |
| |
· |
Moved forward with placing phased orders for raw materials to ensure future product availability to support both our UK Licensee while also providing for capacity to flex up further as other routes to market materialize in line with management’s commercialization program. |
| |
· |
Commenced phased deliveries in December 2021 to the UK Licensee of its continuous glucose monitor. |
In July 2020,
Nemaura filed a PMA application with the FDA to use sugarBEAT® as an adjunct to
finger prick testing for blood glucose trending. We, along with other applicants, were then informed by the FDA that the approval process
was currently subject to delays as a result of the FDA’s Center for Devices and Radiological Health (“CDRH”) being actively
engaged in responding to the current pandemic caused by COVID-19 which resulted in staff being reallocated to other approval requests
associated with COVID-19. During April 2021 the FDA confirmed that they would recommence their review of the PMA application and this
is now ongoing and in-progress. In December 2021 the FDA’s Bio-monitoring research division conducted an audit of the clinical program
submitted in support of the PMA application. A single 483 observation was raised, and the Company submitted a full response in January
2022. The FDA subsequently scheduled a pre-market inspection for the second calendar quarter of 2022, intended to cover the FDA’s
Quality System/Current Good Manufacturing Practice regulations for Medical Devices (21 CFR Part 820). This audit was conducted in
the first quarter of the fiscal year ending March 31, 2023. The Company reported that a single 483 observation was raised to
which the Company responded in a timely manner, and Dialogue with the FDA continues with respect to the PMA application.
In addition to this, Nemaura
established that proBEAT™, which is based on the sugarBEAT® platform, can be classified
under the Wellness guidance when it is used according to the FDA Wellness guidance notes, to provide prompts and educate users on factors
affecting their blood sugar profiles. Nemaura launched proBEAT™ in the U.S. in December 2020, as part of a diabetes prevention and
reversal program branded BEATdiabetes.life, in the form of pilot studies. During the quarter ended December 31, 2020, Nemaura licensed
a clinically validated weight loss program for the management of diabetes from Healthimation, LLC, which was originally developed at the
Joslin Diabetes Center, an affiliate of Harvard Medical School. This program, together with proBEAT™, originally formed the basis
of the BEATdiabetes.life program that is currently being developed for commercialization in the U.S. The program is under further refinement
based on feedback gathered to date. Further KOL studies are planned to provide additional marketing support of the program in preparation
for a broader U.S.-wide roll-out. While still in the relatively early stages, we are pleased with initial results and feedback received
from these user-groups and one key outcome has been that the company is now focusing on providing proBEAT as a standalone plug-in for
existing diabetes management programs, therefore potentially allowing the company to accelerate commercialization as well as reduce the
cost burden associated with running its own program.
We believe there are
additional applications for sugarBEAT® and the underlying BEAT technology platform,
which may include:
| |
· |
a web-server accessible by physicians and diabetes professionals to track the condition remotely, thereby reducing healthcare costs and managing the condition more effectively; |
| |
· |
a complete virtual doctor that monitors a person's vital signs and transmits results via the web; |
| |
· |
other patches using the BEAT technology platform to measure alternative analytes, including lactate, uric acid, lithium and drugs. This would be a step-change in the monitoring of conditions, particularly in the hospital setting. Lactate monitoring is currently used to determine the relative fitness of professional athletes and we completed preliminary studies demonstrating the application of the BEAT technology for continuous lactate monitoring; and |
| |
· |
a continuous temperature monitoring system which could have various applications, including use for individuals to monitor their temperature in connection with diagnosis and monitoring of symptoms of viral infections, and in females to monitor the ovulation cycle. |
During this period of product development, the Company
has experienced recurring losses and negative cash flows from operations. For the fiscal years ended March 31, 2022 and 2021, we generated
$503,906 and $0 revenues from product sales and reported net losses of $13,886,805 and $6,258,596, respectively, and negative cash flow
from operating activities of $6,504,041 and $5,998,097, respectively. For the nine months ended December 31, 2022 and 2021, we generated
$77,044 and $183,628 revenues from product sales and reported net losses of $9,460,888 and $10,269,557, respectively, and negative cash flow from operating activities of
$6,090,181 and $4,353,494, respectively. As noted in our
financial statements, as of December 31, 2022, we had a cash balance of $7,340,840, working capital deficit of $2,309,666, an accumulated
deficit of $47,192,364 and a deficiency in total stockholders' equity of $9,858,709. There is substantial doubt regarding our ability
to continue as a going concern as a result of our historical recurring losses, negative cash flows from operations, existing debt of
$20,070,259 due as of December 31, 2022, which will require the restructure of the debt or obtain additional, new, funding. See “Risk
Factors—We have a history of operating losses, our management has concluded that factors raise substantial doubt about our ability
to continue as a going concern and our auditor has included an explanatory paragraph relating to our ability to continue as a going concern
in its audit report for the fiscal years ended March 31, 2022 and 2021.”
While the Company expects
to continue to incur losses from operations for the near-term and these losses could be significant as product development, regulatory
activities, clinical trials, and other commercial and product development related expenses are incurred, the Company reached a significant
milestone during the three month period ended December 31, 2021, as the Company commenced commercial delivery of its sugarBEAT®
device to its UK Licensee, allowing them to continue studies dedicated to developing user based feedback and evidence that could
potentially support reimbursement in the UK.
Management's strategic
assessment continues to include the following potential options:
| |
· |
obtaining further regulatory approval for the sugarBEAT® device in other global territories, including the U.S., and the Middle East; |
| |
· |
signing new/additional licensing and collaboration opportunities beyond our existing licensee partners; |
| |
· |
pursuing further capital raising opportunities to support and accelerate the commercialization strategy; and |
| |
· |
developing the sugarBEAT® device platform for commercialization for other applications. |
Our Business
Strategy
We intend to
lead in the discovery, development and commercialization of innovative and targeted diagnostic medical devices, and data-driven digital
platforms, that improve disease monitoring, management and overall patient care. Specifically, we intend to focus on the monitoring of
molecules that can be drawn out through the skin non-invasively using our technology platform. In addition to glucose, such molecules
may include lactic acid monitoring and the monitoring of prescription drugs and blood biomarkers that may help in the diagnosis, prevention,
or management of diseases, such as diabetes. We plan to take the following steps to implement our broad business strategy. Our key commercial
strategies post-approval will first be implemented in Europe and then in parts of the Middle East and Asia, and then the U.S., as follows:
| |
- |
Commercialize sugarBEAT® in the United Kingdom and Republic of Ireland. We intend to commercialize sugarBEAT® in the United Kingdom, and Republic of Ireland with MySugarWatch Limited (previously known as Dallas Burston Ethitronix Limited) (“MSW”), with whom we have an exclusive marketing rights agreement for these two countries. We have also signed a full commercial agreement with MySugarWatch (Europe) Limited (previously known as Dallas Burston Ethitronix (Europe) Limited) in May 2018 for all other European territories as part of an equal joint venture agreement. The joint venture intends to seek sub-license rights opportunities to one or more leading companies in the diabetes monitoring space, to leverage their network, infrastructure and resources. |
| |
- |
Establish licensing or joint venture agreements with other parties to market sugarBEAT® in other geographies. We are in detailed discussions and negotiations with several other parties worldwide for licensing or joint venture agreements for the sale of the sugarBEAT® device and have signed a commercial agreement with TP MENA for the Gulf Cooperation Council, excluding Qatar. |
| |
- |
Seek FDA PMA approval of sugarBEAT®. The PMA application is currently in review by the FDA. |
| |
- |
Expand the indications for which the sugarBEAT® device may be used. We believe that the sugarBEAT® device may offer significant benefits as compared to those found in the non-acute setting for the monitoring of other diseases. This includes monitoring of lactic acid for performance athletics, and the monitoring of drugs. We have completed initial proof of concept for lactate monitoring and now plan to explore the routes to commercialization.. |
| |
- |
Expand our product pipeline through our proprietary platform technologies, acquisitions and strategic licensing arrangements. We intend to leverage our proprietary platform technologies to grow our portfolio of product candidates for the diagnosis of diabetes and other diseases. This includes digital platforms driven by data gathered by our sensors within the medical and wellbeing markets, such as for metabolic health monitoring. In addition, we intend to license our product and acquire products and technologies that are consistent with our research and development and business focus and strategies. This may include drug delivery products for the improved management of diabetes, for example improved insulin injector systems, and/or combination drug products for diabetes related drugs. |
Product
Development
Management
has extensive experience in regulatory and clinical development of diagnostic medical devices. We intend to take advantage of this experience
in the field of diagnostic medical devices in an attempt to increase the probability of product approval. The overall regulatory
process for diagnostic medical devices for diabetes is currently similar to those governing other diagnostic devices. The timelines are
shorter than, for example, when new drugs or completely invasive diagnostic devices are trialed in clinics. We have successfully tested
and evaluated the device for its clinical output, in this case the accuracy and safety with which it can trend blood glucose levels,
based on which CE approval was granted by the Notified Body BSI. A PMA (pre-market approval) application was also submitted to the FDA
and is currently under review. We continue to seek collaborations with future licensees and marketing partners to achieve our commercial
growth milestones.
The table below provides our current estimate of our timeline:
Product Development and Commercialization
Timelines
| |
|
|
| Milestone |
Target Start Date |
Current Status |
| Completion of clinical studies in Type 1 and Type 2 diabetic subjects to define final device claims and for submission for CE Mark approval with final device claims. |
July 2017 |
Completed |
| CE Mark for body worn transmitter device |
August 2018 |
Completed |
| U.S. FDA PMA Submission |
June 2020 |
Submission Completed, FDA review ongoings |
| Commercial launch in the UK, followed by major territories in Europe |
July - September 2020 |
Staggered launch in progress |
| Commercial launch of proBEATÔ in the U.S. |
October - December 2020 |
Commenced December 2020 |
|
Scale up of commercial sensor / patch manufacturing.
Scale up means we have started looking at larger scales sufficient
for product launch in the UK and relates to the manufacturing process for sensors. |
December 2020 |
In progress |
| Scale up of device (transmitter) manufacturing |
December 2020 |
In progress |
| Commencement of product delivery to UK Licensee |
December 2021 |
On-going |
Market
Opportunity for the Company's Products
According
to the International Diabetes Federation Atlas 10th Edition 2021 (the "IDF"), there are approximately 537 million
adults living with diabetes, representing 10.5% of the world’s population in this age group. This number is predicted to rise to
643 million (11.3%) by 2030 and to 783 million (12.2%) by 2045. Additionally an estimated 240 million people are living with undiagnosed
diabetes worldwide, meaning almost one-in-two adults with diabetes are unaware they have the condition. The IDF identifies that almost
90% of people with undiagnosed diabetes live in low-and-middle income countries.
Statistics
published by the IDF evidence the fact that diabetes is a huge and growing problem, and that whilst the costs to society are already high,
they continue to escalate. In addition, the IDF also notes that Europe has the highest prevalence of children and adolescents with Type
1 diabetes, as well as the highest incidence annually. Europe is also reported as having the second highest average cost per person with
diabetes ($3,086), with only North America and the Caribbean being higher ($8,208).
Statistical
Data for Diabetes Globally
| |
|
|
|
| |
2021 |
2030 |
2045 |
| Total world population |
7.9 billion |
8.6 billion |
9.5 billion |
| Adult population (20-79 years) |
5.1 billion |
5.7 billion |
6.4 billion |
| |
Diabetes (20 – 79 years) |
| Prevalence (%) |
10.5% |
11.3% |
12.2% |
| Number of people with diabetes |
536.6 million |
642.7 million |
783.2 million |
| Total health expenditure due to diabetes (2021 $) |
$966 billion |
$1,028 billion |
$1,054 billion |
| |
Impaired Glucose Tolerance “IGT” (20 – 79 years) |
| Prevalence (%) |
10.6% |
11.0% |
11.4% |
| Number of people with IGT |
541.0 million |
622.7 million |
730.3 million |
| |
Type 1 diabetes (0 – 19 years) |
| Number of children / adolescents with Type 1 diabetes |
1.2 million |
- |
- |
| Number of newly diagnosed cases per year |
184,100 |
- |
- |
Type
1 diabetes, once known as juvenile diabetes or insulin-dependent diabetes, is a chronic condition in which the pancreas produces little
or no insulin, a hormone needed to allow sugar (glucose) to enter cells to produce energy. The far more common Type 2 diabetes occurs
when the body becomes resistant to the effects of insulin or doesn't make enough insulin.
Various
factors may contribute to Type 1 diabetes including genetics and exposure to certain viruses. Although Type 1 diabetes typically appears
during childhood or adolescence, it also can develop in adults.
Despite
active research, Type 1 diabetes has no cure, although it can be managed. With proper treatment, people who have Type 1 diabetes can expect
to live longer, healthier lives than they did in the past. Type 1 diabetes includes autoimmune Type 1 diabetes (Type 1a) which is characterized
by having positive autoantibodies, as well as idiopathic Type 1 diabetes (Type 1b) where autoantibodies are negative, and c-peptide is
low. Patients with Type 1 diabetes (insulin dependent) require long term treatment with exogenous insulin and these patients perform self-monitoring
of blood glucose (SMBG) to calculate the appropriate dose of insulin. SMBG is done by using blood samples obtained by finger sticks but
frequent SMBG does not detect all the significant deviations in blood glucose, specifically in patients who have rapidly fluctuating glucose
levels.
Type
2 diabetes, once known as adult-onset or non-insulin-dependent diabetes, is a chronic condition that affects the way your body metabolizes
sugar (glucose), your body's main source of fuel. With Type 2 diabetes, your body either resists the effects of insulin, a hormone that
regulates the movement of sugar into your cells or doesn't produce enough insulin to maintain a normal glucose level. Untreated, Type
2 diabetes can be life-threatening.
More
common in adults, Type 2 diabetes increasingly affects children as childhood obesity increases. Whilst there is currently no acknowledged
cure for Type 2 diabetes, there is increasing evidence to suggest that it can be effectively managed by eating well, exercising and maintaining
a healthy weight. If diet and exercise don't control the blood sugar, diabetes medications or insulin therapy may be required.
Each
year, millions of patients undergo diabetes testing in the European Union and in the U.S. The main reason for this testing is to detect
and evaluate diabetes in patients with symptoms of diabetes. These studies provide clinical benefit in the initial evaluation of patients
with suspected but unproven diabetes, and in those patients in whom a diagnosis of diabetes has been established and information on prognosis
or risk is required.
We
believe that our market opportunity is a direct function of the number of persons tested, diagnosed and treated for Type 2 diabetes. The
IDF indicates that the total world market opportunity for a continuous glucose monitoring device is in the billions of dollars and is
projected to grow annually as incidences of diabetes continue to grow.
We
do not believe it is possible to estimate the number of diabetes patients that undergo finger pricks or other types of invasive glucose
monitoring. However, we are unaware of any product currently on the market that may allow for non-invasive continuous glucose monitoring.
We believe the sugarBEAT® device may be readily adopted by the medical community for the assessment of a patient continuously.
We
believe our non-invasive sugarBEAT® device possesses many significant advantages and may represent an ideal device for the detection
of discordances in an individual's blood sugar levels. We believe the CE approved sugarBEAT® device may represent a best-in-class
non-invasive continuous glucose monitoring device to reach those afflicted with diabetes. While we cannot estimate the market share that
our sugarBEAT® device may capture, we believe that the sugarBEAT® device will capture a significant share of the non-invasive
continuous glucose monitoring market, in-particular the market that has been established by the Abbott Freestyle Libre device for glucose
trending, as well as be adopted by non-insulin dependent diabetics who have not historically used continuous glucose monitoring devices
due to their invasiveness.
Commercialization
Plan
Throughout
the fiscal year ended March 31, 2022, we continued to work with our UK Licensee, MSW, to provide support in the development of their go-to-market
strategy which incorporates the utilization of our sugarBEAT® device into their own branded product offering. While COVID-19 did result
in some short delays to MSW’s user assessment program, the overall feedback was positive, albeit the anticipated timetable for purchase
orders to be placed by MSW was extended out, with the first order for 5,000 sugarBEAT® transmitters and 200,000 sugarBEAT® sensors
not being placed until April 2021. Our focus continues to be to support and optimize MSW’s launch program and activities designed
to gather evidence that could potentially support reimbursement, in line with which, we took the following actions during the fiscal year
ended March 31, 2022:
| |
· |
Entered into a new leased facility to provide additional capacity for commercial assembly to commence. |
| |
· |
Increased headcount of production operatives to facilitate product manufacture. |
| |
· |
Placed forward orders for raw materials to support scale-up and secure inventory of those items that are currently in short supply globally i.e. semi-conductors etc. |
| |
· |
Appointed Benchmark Electronics Inc.as our CMO partner to facilitate future volume scale up of transmitter production via its FDA approved facility in Thailand, in relation to which technology transfer is in-progress. |
| |
· |
Signed a new global agreement for the provision of our sugarBEAT® device with MySugarWatch DuoPack Limited (“MSW-DP”). Under the terms of the agreement, our CGM and sensors will be provided as Duo-Packs with prescription only medicines that are widely prescribed for people with Type 2 diabetes. The first of these medicines lost its patent protection in the fourth calendar quarter of 2022 in the UK, and the initial Duo-Pack presentation is expected to be launched in 2023 by the licensee. |
| |
· |
Commenced phased delivery of transmitters against the purchase order received from MSW in December 2021. |
We also advanced our plans to develop our
go-to-market capabilities in the U.S., which included:
| |
· |
In July 2020, we submitted a PMA application to the FDA for the sugarBEAT® device for glucose profiling as an adjunct to a finger-stick measurement. We, along with other applicants, were then informed by the FDA that the approval process was subject to delays as a result of the CDRH being actively engaged in responding to the pandemic caused by COVID-19 which resulted in staff being reallocated to other approval requests associated with COVID-19. In April 2021, the FDA confirmed that it would recommence its review of the PMA application. |
| |
· |
In December 2021, the FDA’s Bio-monitoring research division conducted an audit of the clinical program submitted in support of the PMA application. A single 483 observation was raised, and the Company submitted a full response in January 2022. |
| |
· |
The FDA subsequently conducted a pre-market inspection for during the second calendar quarter of 2022, covering the FDA’s Quality System / Current Good Manufacturing Practice regulations for Medical Devices (21 CFR Part 820). Once again a single 483 observation was made, and this was responded to within the mandated time frame. The company continues its dialogue with the FDA with respect to the PMA submission and plans to provide further material updates as they arise in due course. |
In
addition to this, we continue to explore commercialization opportunities in other key geographic markets, which includes engaging with
the German regulatory authority (GBA) to establish how best to proceed with achieving reimbursement for sugarBEAT® in Germany, as
well as continuing to engage in dialogue with additional potential licensees / distributors in other geographical territories.
Competitive
Landscape
To
the best of our knowledge, there are currently no other competing devices on the market that offer continuous glucose monitoring and profiling,
non-invasively, with a single day sensor wear. We believe this positions us uniquely in a market where we can target persons with diabetes
as well as those that are pre-diabetic. Additionally, we believe that this can also be used to improve outcomes in weight management and
wellbeing markets. There are companies, such as Dexcom and Abbott, that currently offer Continuous Glucose Monitoring (CGM) sensors with
10 and 14 continuous day wear, respectively. These companies could be deemed future competitors were they to:
| |
– |
develop and market products that are less expensive or more effective than our current and/or future products; |
| |
– |
operate larger research and development programs or have substantially greater financial resources than we do; |
| |
– |
initiate or withstand substantial price competition more successfully than we can; |
| |
– |
have greater success in recruiting skilled technical and scientific workers from the limited pool of available talent; |
| |
– |
more effectively negotiate third-party licenses and strategic relationships; and |
| |
– |
take advantage of acquisition or other opportunities more readily than we can. |
We
may compete for market share against these companies and potential newcomers in this general field. These potential competitors, either
alone or together with their partners, may develop new products that will compete with ours, and these competitors may, and in certain
cases do, operate larger research and development programs, or have substantially greater financial resources than we do.
As
noted, while it is difficult to analyze our major competitors since currently there are no non-invasive diagnostic medical devices to
continuously monitor blood glucose levels, we anticipate that specific companies may compete with us in the future.
Regulatory
Requirements
Our
device has undergone the applicable electrical safety testing and biocompatibility has been demonstrated against the relevant European
Directives, Regulations and Standards. If and when new materials are introduced, they will undergo a biocompatibility risk assessment,
and further testing where necessary. Batches of the device and patches were manufactured for human clinical studies that took place between
November 2014 and December 2015. This was a functional watch device with a wire connection to a skin adhered sensor and electrode. Subsequent
to studies conducted in India the device received a CE mark approval in February 2016. The device has since been upgraded to reduce it
in size, include an enhanced sensor system and allow wireless communication from a body worn transmitter. This miniaturized wireless device
achieved CE approval in May 2019, and a PMA was submitted to the U.S. FDA in July 2020 and is currently in review. An application for
CE mark approval requires the Company to have an ISO13485 Quality Management System, covering the design, development and manufacture
of a medical device. Nemaura Medical does not have this accreditation, and instead under the terms of a service contract dated April 4,
2018, with Nemaura Pharma Limited (“Pharma”), Nemaura Medical has outsourced the CE approval registration process to Pharma.
Pharma, a related company, is controlled by our Chief Executive Officer, President, Chairman of the Board and majority shareholder, Dr
D.F.H. Chowdhury. Under the terms of the service contract Pharma has undertaken all required activities to register the product for CE
approval under a fee for service arrangement, while Nemaura Medical will retain full title and beneficial ownership of the CE mark, and
all related intellectual property without any further payments or royalties becoming due other than the fee for service.
Intellectual Property
We believe that clear and extensive intellectual property
relating to our technologies is central to long-term success and we intend to invest accordingly. This applies to both domestic and international
patent coverage, and trade secrets, and trademarks.
The sugarBEAT® technology is protected by filed
patents and substantial trade secrets covering methods and apparatus for measuring glucose extracted from human skin in a non-invasive
manner, devices for extracting glucose from the skin is a stable manner, and the formulation and process for preparation of the enzyme
solution used in the sensor.
On May 8, 2014, NDM Technologies Limited, a related
company, assigned the UK patent application 1208950.4 and International (PCT) patent application PCT/GB2013/051322 entitled "Cumulative
Measurement of an Analyte" to Dermal Diagnostics Limited (“DDL”) for a nominal consideration.
Additional patents are intended to be filed in the
future relating to the device and sensor, providing new intellectual property protection. Some of the recently filed patents and future
patents may supersede previous intellectual property.
The company retains substantial trade secrets relating
to aspects of the sensor manufacture process and the sensor formulation, which has taken several years to develop, and we believe will
prove challenging to reverse engineer as it consists of formulation components in addition to processing methods in complex combinations
that are unique to the final functional sensor. Patents will not be filed on this aspect of the technology to avoid any public dissemination
of the know-how.
These patents and know-how cover aspects of the technology
platform. Furthermore, the trademarks BEAT and sugarBEAT® have been registered in multiple key global territories. Accordingly, all
intellectual property essential to the sugarBEAT® product is owned by us, and not subject to royalty payments. We intend to take the
lead in the preservation and/or prosecution of these patents and patent applications going forward as required. We intend to file additional
patents as the development progresses, where deemed to be of value to protecting the technology platform and future modifications and
improvements. New patents and patent filings could supersede existing patents as the technology is further developed and optimized. Where
patents cannot be secured, the intellectual property will be limited to know-how and trade secrets, and these will be diligently guarded.
Trade Secrets, Trademarks, and Patents Filed, Granted
and Pending
| IP: Patent (Core Claim), Know-how, Trademark |
|
Expiration Date |
|
Jurisdictions in which Granted / Issued |
|
Jurisdictions in which Pending |
|
Ongoing Royalty or Milestone Payments |
| |
|
|
|
|
|
|
|
|
| Patent: Cumulative Measurement of an Analyte (1) |
|
May 20, 2033 |
|
Australia, France, Germany, Italy, Poland, Spain, Netherlands, UK, China, Japan, USA, Canada, UAE |
|
Brazil, Qatar |
|
None. Internal development |
| Skin Prep Patch (2) |
|
December 2, 2039 |
|
N/A |
|
UK, Europe, USA |
|
None. Internal development |
| Know-how: Sensor Formulation and manufacture processes |
|
N/A |
|
Trade Secret |
|
N/A |
|
None. Internal development |
|
Trademark:
BEAT |
|
Renewal due in 2026 |
|
UK, Canada, China, EU, India, Japan, Norway, Russia, Singapore |
|
Malaysia, Brazil, Mexico, Switzerland, Turkey |
|
None. Internal development |
| Trademark: sugarBEAT |
|
Renewal due in 2025 |
|
UK, Canada, Australia, Switzerland, China, Egypt, EU, Israel, India, Iran, Japan, North Korea, Morocco, Mexico, Norway, New Zealand, Russia, Singapore, Tunisia, Turkey, USA |
|
N/A |
|
None. Internal development |
(1) This patent provides a formula for calculating
the amount of glucose extracted over a defined period of time by deducting the difference between two readings to allow rapid sensing
without needing to deplete the analyte being measured.
(2) This patent describes a device and method for
preparing the skin for the measurement of glucose on the skin surface.
Clinical
Trials
Our
clinical testing is conducted by contract clinical research organizations in various centers around the world to cover a wide demographic
– including Asia and Europe – and is managed by our in-house management team.
We
had 2 pre-submission meetings with the FDA in June 2016, to define the clinical roadmap. As a result, a detailed clinical plan was developed
and approved internally and a clinical site in Europe was selected and audited and approved for commencement of clinical studies using
the body worn transmitter device version of the sugarBEAT®. The study was completed, and a PMA application submitted to the FDA in
July 2020.
The
data from these studies was also submitted as part of the CE approval in Europe was received in May 2019.
Research
and development
We
spent $1,556,988 and $1,554,603 during the fiscal years ended March 31, 2022 and 2021, respectively, on research and development; management
currently anticipated that spend in this area will remain reasonably consistent in the coming fiscal year.
Manufacturing
The
manufacture and sale of CE certified medical devices are controlled and governed by guidelines stipulated in the International Organization
for Standardization (ISO), more specifically ISO13485; sugarBEAT® will be manufactured and marketed according to ISO13485 quality
standards.
In
support of commercial sales of sugarBEAT® in the UK and EU we have worked with our manufacturing partner Nemaura Pharma, to scale-up
manufacturing of the various sugarBEAT® components alongside facilities for final assembly and packaging. As part of this process,
we have expanded our manufacturing and assembly capabilities by occupying additional space within our existing headquarters site at Loughborough
University Science and Enterprise Park (LUSEP) in the UK.
We
have entered into the following types of agreements with various manufacturing partners:
| |
– |
Manufacturing agreements for the sensor manufacture |
| |
– |
Manufacturing agreements for the patch manufacture |
| |
– |
Manufacturing agreements for the CGM transmitter device and re-charging station manufacture |
Sales
and Marketing
An
Exclusive Marketing Rights agreement for the UK and Republic of Ireland was signed on March 31, 2014 with Dallas Burston Pharma, a Jersey
(Channel Island) based company (“DB Pharma”) (subsequently updated in 2018 and again in 2021 to include a change in the company
name to MySugarWatch Limited “MSW”), who has pharmaceutical product marketing operations in the UK and has demonstrated a
very successful model for the marketing of prescription medical products directly to general practitioners. We received a non-refundable
upfront payment of £1 million ($1.67 million at the then exchange rate) in return for providing MSW with the exclusive right to
sell the sugarBEAT® device in the UK and Republic of Ireland, both direct to consumer and through prescriptions by general practitioners.
The key terms of the Exclusive Marketing Rights Agreement were concluded in a Commercial Agreement signed in August 2015. This agreement
was updated and re-issued in October 2019 to cover new IP / improvements to the technology.
In
addition, a joint venture agreement was entered into with MySugar Watch (Europe) Limited (previously known as Dallas Burston Ethitronix
(Europe) Limited) in May 2018, whereby we will share equally the costs and net profits of the sales of our sugarBEAT® system in all
territories in Europe, with the exception of the United Kingdom, which is the subject of a separate agreement with MSW. This agreement
was updated and re-issued in October 2019 to cover new IP/ improvements to the technology. Commercial agreements were signed in 2018 with
TPMENA and Al-Danah Medical, for the Gulf Region (GCC) and Qatar respectively.
Regulatory
matters
Government
Regulation
Our
business is subject to extensive federal, state, local and foreign laws and regulations, including those relating to the protection of
the environment, and health and safety. Some of the pertinent laws have not been definitively interpreted by the regulatory authorities
or the courts, and their provisions are open to a variety of subjective interpretations. In addition, these laws and their interpretations
are subject to change, or new laws may be enacted.
Both
federal and state governmental agencies continue to subject the healthcare industry to intense regulatory scrutiny, including heightened
civil and criminal enforcement efforts. We believe that we have structured our business operations to comply with all applicable legal
requirements. However, it is possible that governmental entities or other third parties could interpret these laws differently and assert
otherwise. We discuss below the statutes and regulations that are most relevant to our business.
United
Kingdom and Wales and the European Union regulations
Government
authorities in the United Kingdom and Wales and the European Union as well as other foreign countries extensively regulate, among other
things, the research, development, testing, manufacture, labelling, promotion, advertising, distribution, sampling, marketing and import
and export of medical devices, including patches and other pharmaceutical products. Our body worn transmitter devices in the United Kingdom
and Wales will be subject to strict regulation and require regulatory approval prior to commercial distribution. The process of obtaining
governmental approvals and complying with ongoing regulatory requirements requires the expenditure of substantial time and financial resources.
In addition, statutes, rules, regulations and policies may change and new legislation or regulations may be issued that could delay such
approvals. If we fail to comply with applicable regulatory requirements at any time during the product development process, approval process,
or after approval, we may become subject to administrative or judicial sanctions. These sanctions could include the authority's refusal
to approve pending applications, withdrawals of approvals, clinical holds, warning letters, product recalls, product seizures, total or
partial suspension of our operations, injunctions, fines, civil penalties or criminal prosecution. Any agency enforcement action could
have a material adverse effect on us.
The
European Commission on Public Health (the "ECPH") provides the regulation for the development and commercialization of new medical
diagnostic devices. Any medical device placed on the European market must comply with the relevant legislation, notably with Directive
93/42/EEC for medical devices, with the active implantable devices Directive (90/385/EEC) or with the in vitro devices
Directive (98/79/EC). From 26th May 2021, all newly approved medical devices must comply with the Medical Device Regulation
(2017/745). Before manufacture / import, it must be determined whether the device in question falls under any of these Directives. All
medical devices must fulfil the essential requirements set out in the above-mentioned directives. Where available, relevant standards
may be used to demonstrate compliance with the essential requirements defined in the devices Directives.
Manufacturers
also need to determine the appropriate conformity assessment route. For devices falling under Directive 93/42/EEC / Regulation 2017/745,
other than custom-made devices and devices intended for clinical investigation, the conformity assessment route depends on the class of
the device, to be determined in accordance with certain rules set forth in the directives / regulations. Once the applicable class or
list has been determined, manufacturers need to follow the appropriate conformity assessment procedure. Subject to the type of the device,
this may require manufacturers to have their quality systems and technical documentation reviewed by a Notified Body before they can place
their products on the market. A Notified Body is a third-party body that can carry out a conformity assessment recognized by the European
Union. The Notified Body will need to assure itself that relevant requirements have been met before issuing relevant certification. Manufacturers
can then place the CE marking on their products to demonstrate compliance with the requirements.
The
CE approval is the process of achieving a mandatory conformity marking for the sugarBEAT® device to allow it to be legally sold in
the European Union. It is a manufacturers' declaration that the product meets the requirements of the applicable European laws. The process
for the sugarBEAT® device CE submission and approval involved the following:
1. The device
is classified depending on certain categories described by the European Directive with Class I products being low risk (e.g., band aid
plasters), with Class III devices being the highest risk. The classes are Class I, IIa, IIb and III. Risk is based upon the potential
harm to the patient should a problem arise with a product or its use. The sugarBEAT® device is classified as a IIb device.
2. A 'technical
file' containing all of the information required to demonstrate that the product meets the essential requirements of the European directive
will be prepared. This includes information relating to performance and safety of the device such as product specifications, labelling,
instructions for use, risk analysis and specific test information/clinical evidence relating to the product that support the claims being
made for the product.
3. Clinical evidence
included in the technical file is expected to demonstrate that the device is safe and meets defined performance requirements. This clinical
evidence can be in the form of literature data where substantial published data exists that utilizes the same technique for glucose extraction
and measurement (albeit in a different device format), or data from actual clinical studies performed using the sugarBEAT® device.
The first CE mark submission was based on literature evaluation of 3rd party published clinical data available in the
public domain. The final CE mark submission has claims based on the clinical performance of the device, based on clinical studies described
earlier herein. The clinical data showed that the sugarBEAT® device can trend blood glucose levels in a human subject by taking measurements
every 5 minutes. The clinical trial data demonstrates the sugarBEAT® device blood glucose trend can be used to supplement normal finger
prick measurements.
4. The technical
file has been assessed by an independent inspector (the Notified Body), regulated by the competent authority, (Medicines and Healthcare
products Regulatory Agency, MHRA in the United Kingdom). The Notified Body (an organization in the European Union that has been accredited
by a member state to determine whether a medical device complies with the European medical device directives), will then notify The European
Commission on Public Health (the "ECPH") of the approval and a certificate will be issued to the Company by the notified body
and we will then be able to apply the CE mark to the device, and legally offer the product for sale in the European Economic Area (EEA).
The CE mark has been issued as of May 2019 and the company is now able to offer the device for commercial sale in the EU.
5. The review
of the technical file commenced in August 2018, and the final review and sign off was received in May 2019. Since the CE mark was approved,
we have undergone routine inspections of our ISO 13485 Quality Management System in order to maintain our CE mark accreditation. An addendum
was also submitted to the notified body and approval obtained, to include within the approved CE marked device, the iOS version of the
smart device app that the transmitter connects to.
U.S.
Food and Drug Administration regulation of medical devices
The
US Food, Drug, and Cosmetic Act (the “FDCA”) and FDA regulations establish a comprehensive system for the regulation of medical
devices intended for human use. sugarBEAT® is a medical device that is subject to these, as well as other federal, state, local and
foreign, laws and regulations. The FDA is responsible for enforcing the laws and regulations governing medical devices in the United States.
The
FDA classifies medical devices into one of three classes (Class I, Class II, or Class III) depending on their level of risk and the types
of controls that are necessary to ensure device safety and effectiveness. The class assignment is a factor in determining the type of
premarketing submission or application, if any, that will be required before marketing in the United States. SugarBEAT® falls under
Class III.
| |
– |
Class I devices present a low risk and are not life-sustaining or life-supporting. The majority of Class I devices are subject only to "general controls" (e.g., prohibition against adulteration and misbranding, registration and listing, good manufacturing practices, labelling, and adverse event reporting. General controls are baseline requirements that apply to all classes of medical devices.) |
| |
– |
Class II devices present a moderate risk and are devices for which general controls alone are not sufficient to provide a reasonable assurance of safety and effectiveness. Devices in Class II are subject to both general controls and "special controls" (e.g., special labelling, compliance with performance standards, and post market surveillance. Unless exempted, Class II devices typically require FDA clearance before marketing, through the premarket notification (510(k)) process.) |
| |
– |
Class III devices present the highest risk. These devices generally are life-sustaining, life-supporting, or for a use that is of substantial importance in preventing impairment of human health or present a potential unreasonable risk of illness or injury. Class III devices are devices for which general controls, by themselves, are insufficient and for which there is insufficient information to determine that application of special controls would provide a reasonable assurance of safety and effectiveness. Class III devices are subject to general controls and typically require FDA approval of a PMA application before marketing. |
Unless
it is exempt from premarket review requirements, a medical device must receive marketing authorization from the FDA prior to being commercially
marketed, distributed or sold in the United States. The most common pathways for obtaining marketing authorization are 510(k) clearance
and PMA. After preliminary discussions with the FDA in June 2016 as part of a pre-submission meeting it was determined that the pathway
for sugarBEAT® would be a PMA approval.
Premarket
approval pathway
The
PMA approval process requires an independent demonstration of the safety and effectiveness of a device. PMA is the most stringent type
of device marketing application required by the FDA. PMA approval is based on a determination by the FDA that the PMA contains sufficient
valid scientific evidence to ensure that the device is safe and effective for its intended use(s). A PMA application generally includes
extensive information about the device including the results of clinical testing conducted on the device and a detailed description of
the manufacturing process.
After
a PMA application is accepted for review, the FDA begins an in-depth review of the submitted information. FDA regulations provide 180
days to review the PMA and make a determination; however, in reality, the review time is normally longer (e.g., 1-3 years). During this
review period, the FDA may request additional information or clarification of information already provided. Also, during the review period,
an advisory panel of experts from outside the FDA may be convened to review and evaluate the data supporting the application and provide
recommendations to the FDA as to whether the data provides a reasonable assurance that the device is safe and effective for its intended
use. In addition, the FDA generally will conduct a preapproval inspection of the manufacturing facility to ensure compliance with Quality
System Regulation, which imposes comprehensive development, testing, control, documentation and other quality assurance requirements for
the design and manufacturing of a medical device.
Based
on its review, the FDA may (i) issue an order approving the PMA, (ii) issue a letter stating the PMA is "approvable" (e.g.,
minor additional information is needed), (iii) issue a letter stating the PMA is "not approvable," or (iv) issue an order denying
PMA. A company may not market a device subject to PMA review until the FDA issues an order approving the PMA. As part of a PMA approval,
the FDA may impose post-approval conditions intended to ensure the continued safety and effectiveness of the device including, among other
things, restrictions on labelling, promotion, sale and distribution, and requiring the collection of additional clinical data. Failure
to comply with the conditions of approval can result in materially adverse enforcement action, including withdrawal of the approval.
Most
modifications to a PMA approved device, including changes to the design, labelling, or manufacturing process, require prior approval before
being implemented. Prior approval is obtained through submission of a PMA supplement. The type of information required to support a PMA
supplement and the FDA's time for review of a PMA supplement vary depending on the nature of the modification.
In
February 2020 Nemaura announced that following discussions with the FDA, Nemaura established that Nemaura may sell its CGM product with
a digital service offering in the U.S. without FDA approval as a non-medical wellbeing application. Nemaura further announced that it
intended to launch this product under the brand proBEATÔ in the U.S.. The product enables
users to wear the CGM device from which data will be sent to Nemaura’s servers in the cloud, from where data will be processed to
provide users with educational material and insights into factors that can affect their sugar levels and tips for healthy lifestyle and
diet, with a view to helping pre-diabetics and diabetics alike live healthier lives. A limited product launch commenced by way of pilot
studies in the U.S. in December 2020 to enabled potential customers to register their interest utilizing proBEATÔ in
conjunction with a digital program for weight loss targeted at persons with diabetes, under the brand BEATdiabetes.life.
Clinical
trials
Clinical
trials of medical devices in the U.S. are governed by the FDA's Investigational Device Exemption ("IDE") regulation. This regulation
places significant responsibility on the sponsor of the clinical study including, but not limited to, choosing qualified investigators,
monitoring the trial, submitting required reports, maintaining required records, and assuring investigators obtain informed consent, comply
with the study protocol, control the disposition of the investigational device, submit required reports, etc.
Clinical
trials of significant risk devices (e.g., implants, devices used in supporting or sustaining human life, devices of substantial importance
in diagnosing, curing, mitigating or treating disease or otherwise preventing impairment of human health) require FDA and Institutional
Review Board ("IRB") approval prior to starting the trial. FDA approval is obtained through submission of an IDE application.
Clinical trials of non-significant risk ("NSR") devices (i.e., devices that do not meet the regulatory definition of a significant
risk device) only require IRB approval before starting. The clinical trial sponsor is responsible for making the initial determination
of whether a clinical study is significant risk or NSR; however, a reviewing IRB and/or FDA may review this decision and disagree with
the determination.
An
IDE application must be supported by appropriate data, such as performance data, animal and laboratory testing results, showing that it
is safe to evaluate the device in humans and that the clinical study protocol is scientifically sound. There is no assurance that submission
of an IDE will result in the ability to commence clinical trials. Additionally, after a trial begins, the FDA may place it on hold or
terminate it if, among other reasons, it concludes that the clinical subjects are exposed to an unacceptable health risk.
As
noted above, the FDA may require a company to collect clinical data on a device in the post-market setting.
The
collection of such data may be required as a condition of PMA approval. The FDA also has the authority to order, via a letter, a post-market
surveillance study for certain devices at any time after they have been cleared or approved.
Pervasive
and continuing FDA regulation
After
a device is placed on the market, regardless of its classification or premarket pathway, numerous additional FDA requirements generally
apply. These include, but are not limited to:
| |
– |
Establishment registration and device listing requirements; |
| |
– |
Quality System Regulation ("QSR"), which governs the methods used in, and the facilities and controls used for, the design, manufacture, packaging, labelling, storage, installation, and servicing of finished devices; |
| |
– |
Labelling requirements, which mandate the inclusion of certain content in device labels and labelling, and generally require the label and package of medical devices to include a unique device identifier ("UDI"), and which also prohibit the promotion of products for uncleared or unapproved, i.e., "off-label," uses; |
| |
– |
Medical Device Reporting ("MDR") regulation, which requires that manufacturers and importers report to the FDA if their device may have caused or contributed to a death or serious injury or malfunctioned in a way that would likely cause or contribute to a death or serious injury if it were to recur; and |
| |
– |
Reports of Corrections and Removals regulation, which requires that manufacturers and importers report to the FDA recalls (i.e., corrections or removals) if undertaken to reduce a risk to health posed by the device or to remedy a violation of the Federal Food, Drug and Cosmetic Act that may present a risk to health; manufacturers and importers must keep records of recalls that they determine to be non-reportable. |
The
FDA enforces these requirements by inspection and market surveillance. Failure to comply with applicable regulatory requirements can result
in enforcement action by the FDA, which may include, but is not limited to, the following sanctions:
| |
– |
Untitled letters or warning letters; |
| |
– |
Fines, injunctions and civil penalties; |
| |
– |
Recall or seizure of our products; |
| |
– |
Operating restrictions, partial suspension or total shutdown of production; |
| |
– |
Refusing a request for 510(k) clearance or premarket approval of new products; |
| |
– |
Withdrawing 510(k) clearance or premarket approvals that are already granted; and |
We
would be subject to unannounced device inspections by the FDA, as well as other regulatory agencies overseeing the implementation of and
compliance with applicable state public health regulations. These inspections may include our suppliers' facilities.
Other
Regulation in the United Kingdom and Wales and the EU
Healthcare
Reimbursement
Government
and private sector initiatives to limit the growth of healthcare costs, including price regulation, competitive pricing, coverage and
payment policies, and managed-care arrangements, are continuing in many countries where we do business, including the United Kingdom and
Wales. These changes are causing the marketplace to put increased emphasis on the delivery of more cost-effective medical products. Government
programs, private healthcare insurance and managed-care plans have attempted to control costs by limiting the amount of reimbursement
they will pay for particular procedures or treatments. This has created an increasing level of price sensitivity among customers for products.
Some third-party payers must also approve coverage for new or innovative devices or therapies before they will reimburse healthcare providers
who use the medical devices or therapies. Even though a new medical product may have been cleared for commercial distribution, we may
find limited demand for the product until reimbursement approval has been obtained from governmental and private third-party payers.
Environmental
Regulation
We
are also subject to various environmental laws and regulations both within and outside the United Kingdom and Wales. Like many other medical
device companies, our operations involve the use of substances, including hazardous wastes, which are regulated under environmental laws,
primarily manufacturing and sterilization processes. We do not expect that compliance with environmental protection laws will have a material
impact on our consolidated results of operations, financial position or cash flow. These laws and regulations are all subject to change,
however, and we cannot predict what impact, if any, such changes might have on our business, financial condition or results of operations.
Foreign
Regulation
Whether
or not we obtain regulatory approval for a product, we must obtain approval from the comparable regulatory authorities of foreign countries
before we can commence clinical trials or marketing of the product in those countries. The approval process varies from country to country,
and the time may be longer or shorter than that required for EC approval. The requirements governing the conduct of clinical trials, product
licensing, pricing and reimbursement also vary greatly from country to country.
In
addition, regulatory approval of prices is required in most countries other than the United States. We face the risk that the prices which
result from the regulatory approval process would be insufficient to generate an acceptable return to us or our collaborators.
EU
General Data Protection Regulation
The
EU General Data Protection Regulation (the “GDPR”) came into force in all EU Member States from May 25, 2018 and replaced
previous EU data privacy laws. Although a number of basic existing principles will remain the same, the GDPR introduces new obligations
on data controllers and rights for data subjects, including, among others:
| |
– |
accountability and transparency requirements, which will require data controllers to demonstrate and record compliance with the GDPR and to provide more detailed information to data subjects regarding processing; |
| |
– |
enhanced data consent requirements, which includes “explicit” consent in relation to the processing of sensitive data; |
| |
– |
obligations to consider data privacy as any new products or services are developed and limit the amount of information collected, processed, stored and its accessibility; |
| |
– |
constraints on using data to profile data subjects; |
| |
– |
providing data subjects with personal data in a useable format on request and erasing personal data in certain circumstances; and |
| |
– |
reporting of breaches without undue delay (72 hours where feasible). |
The
GDPR also introduced new fines and penalties for a breach of requirements, including fines for serious breaches of up to the higher of
4% of annual worldwide revenue or €20m and fines of up to the higher of 2% of annual worldwide revenue or €10m (whichever is
highest) for other specified infringements. The GDPR identifies a list of points to consider when imposing fines (including the nature,
gravity and duration of the infringement).
The
Company has assessed the implications of the GDPR on all personal data it holds and has implemented measures to ensure that personal data
shall be:
| |
- |
Processed lawfully, fairly and in a transparent manner in relation to the data subject. |
| |
- |
Collected for a specified, explicit and legitimate purpose and not further processed in a manner that is incompatible with those purposes. |
| |
- |
Adequate, relevant and limited to what is necessary in relation to the purposes for which they are processed. |
| |
- |
Kept in a form which permits identification of data subjects for no longer than is necessary for the purposes for which the personal data is processed. |
| |
- |
Processed in a manner that ensures appropriate security of the personal data, including protection against unauthorised or unlawful processing and against accidental loss, destruction or damage, using appropriate technical or organisational measures. |
| |
- |
Maintained accurately and up to date and that every reasonable step is taken to ensure that personal data that is inaccurate, having regard to the purposes for which they are processed, are erased or rectified without delay. |
At
the current stage of the Company’s development and, with being pre-revenue at this stage, the scope of data held, and consequently
the impact of GDPR, is limited. Increased application of GDPR will be assessed and implemented prior to further Company developments that
warrant additional GDPR measures. As the Company progresses with product commercialization, the extent to which GDPR will affect the Company
will increase, which will require additional changes to the Company’s procedures and policies which could adversely impact operational
and compliance costs. Further, there is a risk that the measures will not be implemented correctly or that individuals within the business
will not be fully compliant with the new procedures. If there are breaches of these measures, the Company could face significant administrative
and monetary sanctions as well as reputational damage which may have a material adverse effect on its operations, financial condition,
and prospects.
Human
Capital Management
We
believe that a diverse workforce is important to our success. We will continue to focus on the hiring, retention and advancement of women
and underrepresented populations, and to cultivate an inclusive and diverse corporate culture. In the future, we intend to continue to
evaluate our use of human capital measures or objectives in managing our business such as the factors we employ or seek to employ in the
development, attraction and retention of personnel and maintenance of diversity in our workforce.
The
success of our business is fundamentally connected to the well-being of our people. Accordingly, we are committed to the health, safety,
and wellness of our employees. We provide our employees with access to a variety of flexible and convenient health and wellness programs,
including benefits that provide protection and security so they can have peace of mind concerning events that may require time away from
work or that impact their financial well-being; that support their physical and mental health by providing tools and resources to help
them improve or maintain their health status and encourage engagement in healthy behaviors; and that offer choice where possible so they
can customize their benefits to meet their needs and the needs of their families.
We
also provide robust compensation and benefits programs to help meet the needs of our employees. We believe that we maintain a satisfactory
working relationship with our employees and have not experienced any labor disputes. As of March 31, 2022, we had 37 personnel employed
on our payroll, which equates to approximately 32 full-time equivalents.
Organizational
Structure
We
are a holding corporation that owns 100% of a diagnostic medical device company specializing in discovering, developing, and commercializing
specialty medical devices. We were organized on December 24, 2013, under the laws of the State of Nevada. We own 100% of Dermal Diagnostic
(Holdings) Limited, an England and Wales corporation formed on December 11, 2013. Dermal Diagnostics (Holdings) Limited owns 100% of the
stock in Dermal Diagnostics Limited (“DDL”), an England and Wales corporation formed on January 20, 2009, and 100% of the
stock in Trial Clinic Limited (“TCL”), an England and Wales corporation formed on January 12, 2011.
The
following diagram illustrates Nemaura’s corporate structure as of February 28, 2023:

During
the fiscal year ended March 31, 2021, the Board of Directors assessed the adequacy of the group’s organizational structure and concluded
that an intermediary holding company, Region Green Limited, was no longer required as the entity had been effectively dormant since inception
and no longer represented a requirement to be maintained. It was therefore determined that Region Green Limited should be unwound, with
the assets held by Region Green Limited being transferred up to Nemaura Medical Inc. following which Region Green Limited would be dissolved.
The
transfer of assets took place on March 5, 2021 and Region Green Limited was formally dissolved as of April 23, 2021.
In
December 2013, we restructured the Company and re-domiciled as a domestic corporation in the United States. The corporate re-organization
was accomplished to preserve the tax advantages under the laws of the England and Wales tax laws for the benefit of the shareholders of
both Dermal Diagnostics Limited and Trial Clinic Limited.
DDL
is a diagnostic medical device company headquartered in Loughborough, Leicestershire, England. DDL was founded on January 20, 2009, to
engage in the discovery, development and commercialization of diagnostic medical devices. The Company’s initial focus has been on
the development of a novel CGM device.
Recent Developments
Termination
of Chief Financial Officer
Effective July 1,
2022, the Company terminated its Company’s Chief Financial Officer and has commenced a search for a U.S. based replacement. Until
a replacement has been selected, the Company’s President and Chief Executive Officer will act as principal financial and accounting
officer of the Company, and the Company’s finance team will continue to support the Company with respect to its accounting and financial
reporting compliance requirements.
Commercial Deliveries
December 2021 marked a significant
milestone in the Company’s evolutionary journey with the first deliveries of the sugarBEAT® non-invasive glucose monitor (“CGM”)
being made to the UK licensee, MySugarWatch Limited (“MSW”). MSW has developed a subscription-based diabetes coaching and
management service that will be provided alongside the CGM, primarily targeting those with type 2 diabetes and continues to undertake
studies to support full reimbursement for the sensors and the diabetes management program in the UK.
The deliveries reflect the
phased delivery schedule agreed upon with MSW in relation to MSW’s initial order that was placed earlier in 2021, as a result of
which the Company started to recognize revenues.
Furthermore, on September
24, 2021 the Company entered into a License, Supply and Distribution Agreement with MySugarWatch DuoPack Limited (“MSW-DP”),
a sister company of MSW, whereby MSW-DP will provide CGM sensors free of charge with certain medications that are widely prescribed to
persons with Type 2 diabetes. These medications came off patent in the fourth calendar quarter of 2022 in Europe and the UK, and due to
come off patent in 2023 in the U.S. The agreed sale price of sensors to MSW-DP under the terms of the agreement is $20 per box of five
sensors for the U.S. market, and in Europe and the UK 12.50 Euros in the first 12 months from product launch and 10 Euros thereafter per
box of four sensors. Nemaura’s anticipated cost of goods per sensor on large-scale production is $1 per sensor. As of January 2022,
there were over 2 million prescriptions written for these medications each month in the combined key EU and UK territories. The Company
believes this will provide an opportunity for rapid market penetration in the use of its CGM sensors, at a scale that can enable the targeted
lower cost of goods to be achieved and thereby support both revenue and margin growth into the future.
Management is now focused
on fulfilling the remainder of the UK licensees’ initial orders and supporting MSW’s UK launch, while also developing the
capabilities of the Company to develop and service new channels of business across other geographic markets via the use of our BEAT platform.
This includes expansion of the consumer metabolic health offering Miboko, launched in late 2021, to employers and insurers across the
U.S.
On January 26, 2023, we announced
receipt of a first purchase order for 5,000 proBEAT™ subscriptions from HealthFleet Inc. (“HealthFleet”), a leading
telehealth provider focusing on care, coaching, and health recommendations. The purchase order consists of 75,000 proBEAT glucose sensors
over an initial five-month period and is valued at $500,000 in revenue. HealthFleet has an option to increase volumes based on customer
response.
ATM Offering
In July 2021, the Company
entered into an At The Market Offering Agreement (the “ATM Agreement”) with H.C. Wainwright & Co., LLC (the “Agent”)
pursuant to which the Company may offer and sell from time to time to or through the Agent shares of the Company’s common stock.
On April 1, 2022, the Company and Agent entered into an amendment (the “Amendment”) to the ATM Agreement, pursuant to which
the parties agreed to expand the meaning of the defined term “Registration Statement” in the ATM Agreement to include, for
the period from April 1, 2022 and thereafter, a new shelf registration statement (File Number 333-263618) on Form S-3 (“New Registration
Statement”) that was filed on March 16, 2022 with the SEC and declared effective by the SEC on March 28, 2022. No other changes
to the ATM Agreement were made by the Amendment.
The offer and sale of shares
of Common Stock through the Agent will be made pursuant to the New Registration Statement, and a related prospectus supplement filed with
the SEC pursuant to which the Company is offering shares of its common stock having an aggregate offering price of up to $3,000,000.
Preliminary
agreement with EVERSANA
On September 27,
2022, the Company entered into a preliminary agreement with EVERSANA to collaborate on the launch strategy of the Company’s BEATdiabetes
program.
Amendment of
Uptown Capital Secured Promissory Note
On February 8, 2021, the
Company, Dermal Diagnostics Limited, a wholly owned subsidiary of the Company (“Dermal Diagnostics”), and Trial Clinic Limited,
a wholly owned subsidiary of the Company (“Trial Clinic” and collectively with the Company and Dermal Diagnostics) issued
to Uptown Capital, LLC (“Uptown”) a secured promissory note (the “Uptown Note”) in the original principal amount
of $24,015,000. The Uptown Note carried an original issue discount of $4,000,000. In addition, the Company agreed to pay $15,000 to Uptown
to cover Uptown’s legal fees, accounting costs, due diligence, monitoring and other transaction costs incurred in connection with
the purchase and sale of the Uptown Note, all of which amount was included in the initial principal balance of the Uptown Note. The purchase
price of the Uptown Note, therefore, was $20,000,000. The original maturity date of the Uptown Note was 24 months after the date the purchase
price for the Uptown Note was delivered.
On October 21, 2022, the
Company entered into an amendment to Secured Promissory Note, dated as of October 21, 2022, by and among the Company, Dermal Diagnostics,
Trial Clinic and Uptown. Pursuant to the terms of the amendment, the Company and Uptown agreed to extend the maturity date of the Uptown
Note to July 1, 2024. In consideration thereof, the Company agreed to pay to Uptown an extension fee in the amount of 5% of the outstanding
balance of the Uptown Note which results in $813,834 being added onto the liability due to Uptown.
The Company and Uptown previously
agreed to reduce the maximum monthly redemption amount from $2,000,000 to $500,000 from June 2022 to February 2023, which reduction remains
in force. Pursuant to the terms of the Amendment, the Company and Uptown agreed to reduce the maximum monthly redemption amount during
the period beginning March 2023 until the Uptown Note is paid in full from $2,000,000 to $1,000,000; provided, however, that upon the
occurrence of an event of default under the Uptown Note, the maximum monthly redemption amount will automatically be increased back to
$2,000,000.
Completion of $8.4
Million Registered Direct Offering and Concurrent Private Placement for Warrants
On January 31, 2023, the
Company completed the closing pursuant to a securities purchase agreement with two accredited institutional investors to purchase 4,796,206
shares of its common stock in a registered direct offering under the Form S-3 and warrants to purchase 4,796,206 shares of its common
stock at an exercise price of $2.00 per share in a concurrent private placement. The combined purchase price for one share of common stock
and one warrant is $1.75 for gross proceeds of $8,393,360.
Impact of COVID-19
A regional or global health
pandemic, including COVID-19, could severely affect our business, results of operations and financial condition. A regional or global
health pandemic, depending upon its duration and severity, could have a material adverse effect on our business. For example, the COVID-19
pandemic has had numerous effects on the global economy and governmental authorities around the world have implemented measures to reduce
the spread of COVID-19. These measures, including shutdowns and “shelter-in-place” orders suggested or mandated by governmental
authorities or otherwise elected by companies as a preventive measure, have adversely affected workforces, customers, consumer sentiment,
economies and financial markets, and, along with decreased consumer spending, have led to an economic downturn in many of our markets.
As a result of the COVID-19
pandemic, we evaluated and executed the steps available to us to ensure we were able to provide protection of our employees and instigated
remote working where possible combined with following all government advice and guidance regarding any engagement within the workplace
that could not be completed remotely. To date this transition to remote working has had little impact on our employee productivity and
has not caused any interruption to our business. Due to the uncertainty of COVID-19, we will continue to assess the situation, including
abiding by any government-imposed restrictions, as and where relevant.
We are unable to accurately predict the impact that
COVID-19 will have on our operations going forward due to uncertainties that will be dictated by the length of time that the pandemic
and related disruptions continue, the impact of governmental regulations that might be imposed in response to the pandemic and overall
changes in consumer behavior.
During this period, we, along with other companies,
were notified by the FDA in the U.S., that our PMA application for sugarBEAT® would be delayed due to the prioritization being given
to COVID-19 related applications and resource activity. While the PMA review resumed as of April 15, 2021, however due to the FDA’s
current workload in light of the pandemic, the review has taken longer than a review normally has taken before the pandemic.
While key suppliers have not been accessible throughout
the whole period of the outbreak, we have been able to be flexible in our priorities and respond favorably to the challenges faced during
the outbreak. We have seen an increase in the adoption of technologies for remote and patient self-monitoring, which therefore potentially
enhances the prospects for Nemaura Medical and its CGM product and planned digital healthcare offering.
At this point in time, there
remains significant uncertainty relating to the potential effect of COVID-19 on our business. As infections may continue to become more
widespread, we could experience a severe negative impact on our business, financial condition, and results of operations. To the extent
the COVID-19 pandemic adversely affects our business and financial results, it may also have the effect of heightening many of the other
risks described in this “Risk factors” section.
Risk Factors
Our business is subject to
numerous risks and uncertainties, including those described in “Risk Factors” immediately following this prospectus summary.
These risks represent challenges to the successful implementation of our strategy and to the growth and future profitability of our business.
These risks include, but are not limited to, the following:
| · | We have a history of operating
losses. |
| · | We will need to raise additional
funds in order to finance the anticipated commercialization of our product by incurring indebtedness, through collaboration and licensing
arrangements, or by issuing securities which may cause dilution to existing stockholders, or require us to relinquish rights to our technologies
and our product. |
| · | Revenue generation from product
sales has only commenced in the prior fiscal year ended March 31, 2021 and may never become profitable. |
| · | Our substantial amount of indebtedness
may adversely affect our cash flow and our ability to operate our business, remain in compliance with debt covenants and make payments
on our indebtedness. |
| · | We are largely dependent on
the success of our sole product candidate, the sugarBEAT® device, and we may not be able to successfully commercialize this potential
product. |
| · | If we fail to obtain regulatory
approval of the sugarBEAT® device or any of our other future products, we will be unable to commercialize these potential products. |
| · | Failure to enroll patients in
our clinical trials may cause delays in developing the sugarBEAT® device or any of our future products. |
| · | Delays in clinical testing could
result in increased costs to us and delay our ability to generate revenue. |
| · | Our clinical trials for any
of our current or future products may produce negative or inconclusive results and we may decide, or regulators may require us, to conduct
additional clinical and/or preclinical testing for these products or cease our trials. |
| · | If approved, the commercialization
of our product, the sugarBEAT® device, may not be profitable due to the need to develop sales, marketing and distribution capabilities,
or make arrangements with a third party to perform these functions. |
| · | Our proprietary rights may not
adequately protect our intellectual property and product and if we cannot obtain adequate protection of our intellectual property and
product, we may not be able to successfully market our product. |
| · | Our ability to commercialize
our product will depend on our ability to sell such products without infringing the patent or proprietary rights of third parties. If
we are sued for infringing intellectual property rights of third parties, such litigation will be costly and time consuming and an unfavorable
outcome would have a significant adverse effect on our business. |
| · | If our product, the sugarBEAT® device,
does not gain market acceptance among physicians, patients and the medical community, we will be unable to generate significant revenue,
if any. |
| · | We have outsourced the bulk
of the commercial manufacturing operations for the various components of the sugarBEAT®, with the exception of the Sensor chemistry
which is being conducted in-house. The failure to find manufacturing partners or expand our internal manufacturing facility could have
an adverse impact on our ability to grow our business. |
| · | If we fail to attract and retain
senior management, consultants, advisors and scientific and technical personnel, our product development and commercialization efforts
could be impaired. |
| · | We expect to expand our marketing
capabilities and, as a result of which we may encounter difficulties in managing our growth, which could disrupt our operations. |
| · | Fluctuations in foreign exchange
rates may adversely affect our financial condition and results of operations. |
| · | Our business, financial condition
and results of operations may be materially adversely affected by global health epidemics, including the COVID-19 pandemic. |
| · | Our competitors may develop
products that are less expensive, safer or more effective, which may diminish or eliminate the commercial success of any potential products
that we may commercialize. |
| · | The use of hazardous materials
in our operations may subject us to environmental claims or liabilities. |
| · | If we fail to comply with extensive
regulations enforced by regulatory agencies with respect to diagnostic medical device products, the commercialization of our product could
be prevented, delayed or halted. |
| · | In the future, we hope to distribute
and sell our product outside of the United Kingdom and the European Union, which will subject us to further regulatory risk. |
| · | Market acceptance of our product
will be limited if users are unable to obtain adequate reimbursement from third-party payers. |
| · | Product liability claims may
damage our reputation and, if insurance proves inadequate, the product liability claims may harm our business. |
| · | We could be negatively impacted
by the application or enforcement of fraud and abuse laws, including anti-kickback laws and other anti-referral laws. |
| · | Our common stock may be delisted
from The Nasdaq Capital Market if we cannot maintain compliance with Nasdaq’s continued listing requirements. |
| · | Our common stock may be affected
by limited trading volume and may fluctuate significantly. |
| · | Our stock price may be volatile. |
Our management has concluded that our historical
recurring losses, negative cash flows from operations, existing debt of $20,070,259 due as of February 28, 2023, which will require the
restructure of the debt or obtain additional, new, funding raise substantial doubt about our ability to continue as a going concern and
our auditor has included an explanatory paragraph relating to our ability to continue as a going concern in its audit reports for the
fiscal years ended March 31, 2022 and 2021.
Implications of Being a Smaller Reporting Company
We are a “smaller reporting
company,” as defined in Rule 12b-2 under the Securities Exchange Act of 1934, as amended (the “Exchange Act”). As a
smaller reporting company, we may take advantage of certain reduced reporting requirements and are relieved of certain other significant
requirements that are otherwise generally applicable to public companies. For instance, smaller reporting companies are not required to
obtain an auditor attestation and report regarding management’s assessment of internal control over financial reporting; are not
required to provide a compensation discussion and analysis; are not required to provide a pay-for-performance graph or Chief Executive
Officer pay ratio disclosure; and may present only two years of audited financial statements and related MD&A disclosure. We will
remain a “smaller reporting company” until the last day of the fiscal year in which we have at least $250.0 million in outstanding
voting and non-voting common equity held by our non-affiliates on the last day of the fiscal year in which we have at least $100 million
in revenue and at least $700 million in outstanding voting and non-voting common equity held by our non-affiliates (in each case, with
respect to common equity value, as measured as of the last business day of the second quarter of such fiscal year).
Corporate Information
Our principal executive offices are located at 57
West 57th Street New York, NY 10019. Our website is located at www.nemauramedical.com and our telephone number is + 1 646-416-8000. Information
found on, or accessible through, our website is not a part of, and is not incorporated into, this prospectus, and you should not consider
it part of the prospectus.
THE OFFERING
We are registering for resale by the selling stockholders named herein
the 4,796,206 shares as described below:
| Common stock to be issued upon exercise of warrants |
4,796,206 shares of our common stock issuable upon exercise of warrants acquired by the selling stockholders in a private placement transaction on January 27, 2023. |
| |
|
| Common stock outstanding prior to exercise of warrants |
28,899,402 shares of common stock (1) |
| |
|
| Common stock to be outstanding assuming exercise of all warrants |
33,695,608 shares of common stock. |
| |
|
| Terms of the warrants |
Each warrant will be exercisable for one share of common stock at an exercise price of $2.00 per share. The warrants are exercisable at the later of the effective date of shareholder approval or six months following the issue date and will expire five and one-half years from the issue date. In the event that there is no effective registration statement registering the shares underlying the warrants, then the warrants may be exercised by means of a “cashless exercise” at the holder’s option, such that the holder may use the appreciated value of the warrants (the difference between the market price of the underlying shares of common stock and the exercise price of the underlying warrants) to exercise the warrants without the payment of any cash. |
| |
|
| Use of proceeds |
We will not receive any of the proceeds from the sale by the selling stockholders of 4,796,206 shares of common stock being registered hereby. However, we expect to receive approximately $9,592,412 in gross proceeds assuming the cash exercise of all of the warrants by the selling stockholders to purchase the 4,796,206 shares of common stock being registered hereby at an exercise price of $2.00 per share of common stock. However, the warrants may be exercised on a cashless basis, in which case we would not expect to receive any gross proceeds from the cash exercise of the warrants. We intend to use any net proceeds from the cash exercise of the warrants for working capital and general corporate purposes. |
| |
|
| Risk factors |
Investing in our securities involves a high degree of risk. See the information contained in or incorporated by reference under the heading “Risk Factors” in this prospectus and in the documents incorporated by reference into this prospectus and any free writing prospectus that we authorize for use. |
| |
|
| Market symbol and trading |
Our common stock is listed on the Nasdaq Capital Market under the symbol “NMRD.” |
| |
|
| (1) |
The number of shares of common stock expected to be outstanding after this offering is based on 28,899,402 shares of common stock outstanding as of February 28, 2023 and excludes: |
| |
• |
573,098 shares of common stock issuable upon the exercise of warrants outstanding as of February 28, 2023, with a weighted average exercise price of $8.57 per share; and |
| |
|
|
| |
• |
49,709 shares of common stock issuable upon the exercise of options outstanding as of February 28, 2023, with a weighted average exercise price of $5.74 per share. |
SELECTED HISTORICAL CONSOLIDATED FINANCIAL DATA
The following table presents our selected historical
consolidated financial data for the periods indicated. The selected historical consolidated financial data for the years ended March 31,
2022 and 2021 and the balance sheet data as of March 31, 2022 and 2021 are derived from the audited financial statements. The summary
historical financial data for the nine months ended December 31, 2022 and 2021 and the balance sheet data as of December 31, 2022 and
2021 are derived from our unaudited financial statements.
Historical results are included for illustrative and
informational purposes only and are not necessarily indicative of results we expect in future periods, and results of interim periods
are not necessarily indicative of results for the entire year. The data presented below should be
read in conjunction with, and are qualified in their entirety by reference to, “Management’s Discussion and Analysis of Financial
Condition and Results of Operations” and our consolidated financial statements and the notes thereto included elsewhere in this
prospectus.
| | |
Year Ended | |
Nine Months Ended |
| | |
March 31, 2022 | |
March 31, 2021 | |
December 31, 2022 | |
December 31, 2021 |
| | |
| |
| |
(unaudited) |
| Statement of Operations Data | |
| |
| |
| |
|
| Sales | |
$ | 503,906 | | |
$ | — | | |
$ | 77,044 | | |
$ | 183,628 | |
| Cost of sales | |
| (344,300 | ) | |
| — | | |
| (75,327 | ) | |
| (172,393 | ) |
| Gross profit | |
| 159,606 | | |
| — | | |
| 1,717 | | |
| 11,235 | |
| Total operating expenses | |
| 7,730,037 | | |
| 4,586,741 | | |
| 5,310,168 | | |
| 5,139,091 | |
| Loss from operations | |
| (7,570,431 | ) | |
| (4,586,741 | ) | |
| (5,308,451 | ) | |
| (5,127,856 | ) |
| Interest expense | |
| (6,666,630 | ) | |
| (2,007,687 | ) | |
| (4,152,437 | ) | |
| (5,141,701 | ) |
| Loss before income tax benefit | |
$ | (14,237,061 | ) | |
$ | (6,594,428 | ) | |
$ | (9,460,888 | ) | |
$ | (10,269,557 | ) |
| Provision for income tax benefit | |
| 350,256 | | |
| 335,832 | | |
| — | | |
| — | |
| Net loss | |
| (13,886,805 | ) | |
| (6,258,596 | ) | |
| (9,460,888 | ) | |
$ | (10,269,557 | ) |
| Foreign currency translation adjustment | |
| (257,885 | ) | |
| (472,559 | ) | |
| (864,328 | ) | |
| (142,922 | ) |
| Comprehensive loss | |
| (14,144,690 | ) | |
| (5,786,037 | ) | |
| (10,325,216 | ) | |
| (10,412,479 | ) |
| Basic and diluted net loss per share | |
$ | (0.59 | ) | |
$ | (0.28 | ) | |
$ | (0.39 | ) | |
$ | (0.44 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Balance Sheet Data (at period end) | |
| | | |
| | | |
| | | |
| | |
| Cash | |
$ | 17,749,233 | | |
$ | 31,865,371 | | |
$ | 7,340,840 | | |
$ | 23,046,278 | |
| Working capital (deficit) (1) | |
| (494,444 | ) | |
| 27,565,625 | | |
| (2,309,666 | ) | |
| 8,920,045 | |
| Total assets | |
| 22,101,956 | | |
| 35,242,907 | | |
| 12,961,698 | | |
| 27,073,899 | |
| Total liabilities | |
| 21,635,872 | | |
| 26,884,735 | | |
| 22,820,407 | | |
| 26,050,139 | |
| Stockholders’ (deficit) equity | |
| 466,084 | | |
| 8,358,172 | | |
| (9,858,709 | ) | |
| 1,023,760 | |
(1) Working capital (deficit) represents total current
assets less total current liabilities
RISK FACTORS
An
investment in our securities carries a significant degree of risk. You should carefully consider the following risks, as well as the other
information contained in this prospectus, including our historical financial statements and related notes included elsewhere in this prospectus,
before you decide to purchase our securities. Any one of these risks and uncertainties has the potential to cause material adverse effects
on our business, prospects, financial condition and operating results which could cause actual results to differ materially from any forward-looking
statements expressed by us and a significant decrease in the value of our common shares. Refer to “Cautionary Statement Regarding
Forward-Looking Statements.”
We
may not be successful in preventing the material adverse effects that any of the following risks and uncertainties may cause. These potential
risks and uncertainties may not be a complete list of the risks and uncertainties facing us. There may be additional risks and uncertainties
that we are presently unaware of, or presently consider immaterial, that may become material in the future and have a material adverse
effect on us. You could lose all or a significant portion of your investment due to any of these risks and uncertainties.
Below
is a summary of material risks, uncertainties and other factors that could have a material effect on the Company and its operations:
| · | We have a history of operating
losses, our management has concluded that factors raise substantial doubt about our ability to continue as a going concern and our auditor
has included an explanatory paragraph relating to our ability to continue as a going concern in its audit report for the fiscal years
ended March 31, 2022 and 2021. |
| · | We will need to raise additional
funds in order to finance the anticipated commercialization of our product by incurring indebtedness, through collaboration and licensing
arrangements, or by issuing securities which may cause dilution to existing stockholders, or require us to relinquish rights to our technologies
and our product. |
| · | Revenue generation from product
sales has only commenced in the prior fiscal year ended March 31, 2021 and may never become profitable. |
| · | Our substantial amount of indebtedness
may adversely affect our cash flow and our ability to operate our business, remain in compliance with debt covenants and make payments
on our indebtedness. |
| · | We are largely dependent on
the success of our sole product candidate, the sugarBEAT® device, and we may not be able to successfully commercialize this potential
product. |
| · | If we fail to obtain regulatory
approval of the sugarBEAT® device or any of our other future products, we will be unable to commercialize these potential products. |
| · | Failure to enroll patients in
our clinical trials may cause delays in developing the sugarBEAT® device or any of our future products. |
| · | Delays in clinical testing could
result in increased costs to us and delay our ability to generate revenue. |
| · | Our clinical trials for any
of our current or future products may produce negative or inconclusive results and we may decide, or regulators may require us, to conduct
additional clinical and/or preclinical testing for these products or cease our trials. |
| · | If approved, the commercialization
of our product, the sugarBEAT® device, may not be profitable due to the need to develop sales, marketing and distribution capabilities,
or make arrangements with a third party to perform these functions. |
| · | Our proprietary rights may not
adequately protect our intellectual property and product and if we cannot obtain adequate protection of our intellectual property and
product, we may not be able to successfully market our product. |
| · | Our ability to commercialize
our product will depend on our ability to sell such products without infringing the patent or proprietary rights of third parties. If
we are sued for infringing intellectual property rights of third parties, such litigation will be costly and time consuming and an unfavorable
outcome would have a significant adverse effect on our business. |
| · | If our product, the sugarBEAT® device,
does not gain market acceptance among physicians, patients and the medical community, we will be unable to generate significant revenue,
if any. |
| · | We have outsourced the bulk
of the commercial manufacturing operations for the various components of the sugarBEAT®, with the exception of the Sensor chemistry
which is being conducted in-house. The failure to find manufacturing partners or expand our internal manufacturing facility could have
an adverse impact on our ability to grow our business. |
| · | If we fail to attract and retain
senior management, consultants, advisors and scientific and technical personnel, our product development and commercialization efforts
could be impaired. |
| · | We expect to expand our marketing
capabilities and, as a result of which we may encounter difficulties in managing our growth, which could disrupt our operations. |
| · | Fluctuations in foreign exchange
rates may adversely affect our financial condition and results of operations. |
| · | Our business, financial condition
and results of operations may be materially adversely affected by global health epidemics, including the COVID-19 pandemic. |
| · | Our competitors may develop
products that are less expensive, safer or more effective, which may diminish or eliminate the commercial success of any potential products
that we may commercialize. |
| · | The use of hazardous materials
in our operations may subject us to environmental claims or liabilities. |
| · | If we fail to comply with extensive
regulations enforced by regulatory agencies with respect to diagnostic medical device products, the commercialization of our product could
be prevented, delayed or halted. |
| · | In the future, we hope to distribute
and sell our product outside of the United Kingdom and the European Union, which will subject us to further regulatory risk. |
| · | Market acceptance of our product
will be limited if users are unable to obtain adequate reimbursement from third-party payers. |
| · | Product liability claims may
damage our reputation and, if insurance proves inadequate, the product liability claims may harm our business. |
| · | We could be negatively impacted
by the application or enforcement of fraud and abuse laws, including anti-kickback laws and other anti-referral laws. |
| · | Our common stock may be delisted
from The Nasdaq Capital Market if we cannot maintain compliance with Nasdaq’s continued listing requirements. |
| · | Our common stock may be affected
by limited trading volume and may fluctuate significantly. |
| · | Our stock price may be volatile. |
Risks Related to Our Business
We
have a history of operating losses, our management has concluded that factors raise substantial doubt about our ability to continue as
a going concern and our auditor has included an explanatory paragraph relating to our ability to continue as a going concern in its audit
report for the fiscal years ended March 31, 2022 and 2021.
We
have incurred net losses every year since our inception in 2009. For the fiscal years ended March 31, 2022 and 2021, we generated $503,906
and $0 revenues from product sales and reported net losses of $13,886,805 and $6,258,596, respectively, and negative cash flow from operating
activities of $6,504,041 and $5,998,097, respectively. For the nine months ended December 31, 2022 and 2021, we generated $77,044 and
$183,628 revenues from product sales and reported net losses of $9,460,888 and $10,269,557, respectively, and negative cash flow from operating activities of $6,090,181 and $4,353,494, respectively. As noted in our financial statements, as of December 31, 2022,
we had a cash balance of $7,340,840, working capital deficit of $2,309,666, an accumulated deficit of $47,192,364 and a deficiency in
total stockholders' equity of $9,858,709. We expect to incur losses until our product is successfully launched and cannot be certain that
we will ever achieve profitability. As a result, our business is subject to all of the risks inherent in the development of a new business
enterprise, such as the risk that we may not obtain substantial additional capital needed to support the expenses of developing our technology
and commercializing our potential products; develop a market for our potential products; successfully transition from a company with a
research focus to a company capable of either manufacturing and selling potential products or profitably licensing our potential products
to others; and/or attract and retain qualified management, technical and scientific staff. Our management has concluded that our historical
recurring losses, negative cash flows from operations, existing debt of $20,070,259 due as of December 31, 2022, which will require the
restructure of the debt or obtain additional, new, funding raise substantial doubt about our ability to continue as a going concern and
our auditor has included an explanatory paragraph relating to our ability to continue as a going concern in its audit report for the fiscal
year ended March 31, 2022 and 2021.
Our financial statements do not include any adjustments
that might result from the outcome of this uncertainty. These adjustments would likely include substantial impairment of the carrying
amount of our assets and potential contingent liabilities that may arise if we are unable to fulfill various operational commitments.
In addition, the value of our securities would be greatly impaired. Our ability to continue as a going concern is dependent upon generating
sufficient cash flow from operations and obtaining additional capital and financing. If our ability to generate cash flow from operations
is delayed or reduced and we are unable to raise additional funding from other sources, we may be unable to continue in business even
if this offering is successful. For further discussion about our ability to continue as a going concern and our plan for future liquidity,
see “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Ability to Continue as a
Going Concern.”
We
will need to raise additional funds in order to finance the anticipated commercialization of our product by incurring indebtedness, through
collaboration and licensing arrangements, or by issuing securities which may cause dilution to existing stockholders, or require us to
relinquish rights to our technologies and our product.
Developing
our product, conducting clinical trials, establishing manufacturing facilities and developing marketing and distribution capabilities
is expensive. We will need to finance future cash needs through additional public or private equity offerings, debt financings or corporate
collaboration and licensing arrangements. We cannot be certain that additional funding will be available to us on acceptable terms, or
at all. If adequate funds are not available, we may be required to delay, reduce the scope of, or eliminate one or more of our research
or development programs or our commercialization efforts. To the extent that we raise additional funds by issuing equity securities, our
stockholders may experience dilution. To the extent that we raise additional funds through collaboration and licensing arrangements, it
may be necessary to relinquish some rights to our technologies or our product or grant licenses on terms that are not favorable to us.
Revenue
generation from product sales has only commenced in the prior fiscal year ended March 31, 2021 and may never become profitable.
To
date, we have generated revenue for the first time in the prior fiscal year ended March 31, 2021 for product sales. Our ability to generate
and grow revenue depends on several factors, including our ability to support the market launch of our UK Licensee, successfully obtain
regulatory approval in all key markets identified to commercialize our product pipeline. Even then, we will need to establish and maintain
sales, marketing, distribution and to the extent we do not outsource manufacturing, manufacturing capabilities. We plan to rely on one
or more strategic collaborators to help generate revenues in markets outside of Great Britain however, we cannot be sure that our collaborators,
if any, will be successful. Our ability to generate revenue will also be impacted by certain challenges, risks and uncertainties frequently
encountered in the establishment of new technologies and products in emerging markets and evolving industries. These challenges include
our ability to:
| |
– |
execute our business model; |
| |
– |
create brand recognition; |
| |
– |
manage growth in our operations; |
| |
– |
create a customer base cost-effectively; |
| |
– |
access additional capital when required; and |
| |
– |
attract and retain key personnel. |
We
cannot be certain that our business model will be successful or that it will successfully address these and other challenges, risks, and
uncertainties. If we are unable to generate significant revenue, we may not become profitable, and we may be unable to continue our operations.
Even if we are able to commercialize the sugarBEAT® device, we may not achieve profitability for at least several years, if at all,
after generating material revenue.
Our
substantial amount of indebtedness may adversely affect our cash flow and our ability to operate our business, remain in compliance with
debt covenants and make payments on our indebtedness.
Our
substantial level of indebtedness increases the possibility that we may be unable to generate cash sufficient to pay, when due, the principal
of, interest on or other amounts due with respect to our indebtedness. Our indebtedness could have other important consequences to you
as a stockholder. For example, it could:
| |
– |
make it more difficult for us to satisfy our obligations with respect to our indebtedness and any failure to comply with the obligations of any of our debt instruments, including financial and other restrictive covenants, could result in an event of default under the senior secured credit facility and the senior subordinated note; |
| |
– |
make is more vulnerable to adverse changes in general economic, industry and competitive conditions and adverse change in government regulation; |
| |
– |
require us to dedicate a substantial portion of our cashflow from operations to payments on our indebtedness, thereby reducing the availability of our cashflows to fund working capital, capital expenditures, acquisitions and other general corporate purposes; |
| |
– |
limit our flexibility in planning for, or reacting to, changes in our business and the industry in which we operate; |
| |
– |
place us at a competitive disadvantage compared to our competitors that have less debt; and |
| |
– |
limit our ability to borrow amounts for working capital, capital expenditures, acquisitions, debt service requirements, execution of our business strategy or other purposes. |
Risks
Related to Our Product Candidate and Operations
We
are largely dependent on the success of our sole product candidate, the sugarBEAT® device, and we may not be able
to successfully commercialize this potential product.
We
have incurred and will continue to incur significant costs relating to the development and marketing of our sole product candidate, the
sugarBEAT® device. We have obtained approval to market this product in the EU, but it is not guaranteed that we will achieve this
in any jurisdiction and we may never be able to obtain approval or, if approvals are obtained, to commercialize this product successfully
in other territories.
If
we fail to successfully commercialize our product(s) in multiple territories, we may be unable to generate sufficient revenue to sustain
and grow our business, and our business, financial condition and results of operations will be adversely affected.
If
we fail to obtain regulatory approval of the sugarBEAT® device or any of our other future products, we will be
unable to commercialize these potential products.
The
development, testing, manufacturing and marketing of our product is subject to extensive regulation by governmental authorities in Great
Britain and the European Union. In particular, the process of obtaining CE approval by a Notified Body, a third party that can carry out
a conformity assessment recognized by the European Union, is costly and time consuming, and the time required for such approval is uncertain.
Our product must undergo rigorous preclinical and clinical testing and an extensive regulatory approval process mandated for the CE. Such
regulatory review includes the determination of manufacturing capability and product performance. CE approval was granted by the European
Notified Body BSI in May 2019, allowing the product to be made available for commercial sale. This approval is subject to an annual review
of the underlying ISO 13485 accredited Quality Management System. The accreditation was successfully renewed in November 2021.
There
can be no assurance that all necessary approvals will be granted for future products or that CE review or actions will not involve delays
caused by requests for additional information or testing that could adversely affect the time to market for and sale of our product. Further
failure to comply with applicable regulatory requirements can, among other things, result in the suspension of regulatory approval as
well as possible civil and criminal sanctions.
Failure
to enroll patients in our clinical trials may cause delays in developing the sugarBEAT® device or any of our future
products.
We
may encounter delays in the development and commercialization, or fail to obtain marketing approval, of the sugarBEAT® device or any
other future products if we are unable to enroll enough patients to complete clinical trials. Our ability to enroll sufficient numbers
of patients in our clinical trials depends on many factors, including the severity of illness of the population, the size of the patient
population, the nature of the clinical protocol, the proximity of patients to clinical sites, and the eligibility criteria for the trial
and competing clinical trials. Delays in any possible future patient enrollment, based on request by local regulatory agencies to conduct
studies in their territory, may result in increased costs and harm our ability to complete our clinical trials and obtain regulatory approval.
Delays
in clinical testing could result in increased costs to us and delay our ability to generate revenue.
Significant
delays in clinical testing could materially, adversely impact our product development costs. We do not know whether planned clinical trials
will begin on time, will need to be restructured or will be completed on schedule, if at all. Clinical trials can be delayed for a variety
of reasons, including delays in obtaining regulatory approval to commence and continue a study, delays in reaching agreement on acceptable
clinical study terms with prospective sites, delays in obtaining institutional review board approval to conduct a study at a prospective
site and delays in recruiting patients to participate in a study.
Significant
delays in testing or regulatory approvals for any of our current or future products, including the sugarBEAT® device, could prevent
or cause delays in the commercialization of such product candidates, reduce potential revenues from the sale of such product candidates
and cause our costs to increase.
Our
clinical trials for any of our current or future products may produce negative or inconclusive results and we may decide, or regulators
may require us, to conduct additional clinical and/or preclinical testing for these products or cease our trials.
We
will only receive regulatory approval to commercialize a product candidate if we can demonstrate to the satisfaction of the applicable
regulatory agency that the product is safe and effective. We do not know whether our future clinical trials will demonstrate safety and
efficacy sufficiently to result in marketable products. Because our clinical trials for the sugarBEAT® device may produce negative
or inconclusive results, we may decide, or regulators may require us, to conduct additional clinical and/or preclinical testing for this
product or cease our clinical trials. If this occurs, we may not be able to obtain approval for this product or our anticipated time to
market for this product may be substantially delayed and we may also experience significant additional development costs. We may also
be required to undertake additional clinical testing if we change or expand the indications for our product.
If
approved, the commercialization of our product, the sugarBEAT® device, may not be profitable due to the need to
develop sales, marketing and distribution capabilities, or make arrangements with a third party to perform these functions.
In
order for the commercialization of our potential product to be profitable, our product must be cost-effective and economical to manufacture
on a commercial scale. Subject to regulatory approval, we expect to incur significant sales, marketing, distribution, and to the extent
we do not outsource manufacturing, manufacturing expenses in connection with the commercialization of the sugarBEAT® device and our
other potential products. We do not currently have a dedicated sales force and our current manufacturing capability has limited capacity,
we also have limited experience in the sales, marketing and distribution of medical diagnostic device products. In order to commercialize
the sugarBEAT® device or any of our other potential products that we may develop, we must develop sales, marketing and distribution
capabilities or make arrangements with a third party to perform these functions. Developing a sales force is expensive and time-consuming,
and we may not be able to develop this capacity. If we are unable to establish adequate sales, marketing and distribution capabilities,
independently or with others, we may not be able to generate significant revenue and may not become profitable. Our future profitability
will depend on many factors, including, but not limited to:
| |
– |
the costs and timing of developing a commercial scale manufacturing facility or the costs of outsourcing the manufacturing of the sugarBEAT® device; |
| |
– |
receipt of regulatory approval of the sugarBEAT® device; |
| |
– |
the terms of any marketing restrictions or post-marketing commitments imposed as a condition of approval by regulatory authorities; |
| |
– |
the costs of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights; |
| |
– |
costs of establishing sales, marketing and distribution capabilities; |
| |
– |
the effect of competing technological and market developments; and |
| |
– |
the terms and timing of any collaborative, licensing and other arrangements that we may establish. |
Even
if we receive regulatory approval for the sugarBEAT® device or any other product candidates, we may never receive significant revenues
from any of them. To the extent that we are not successful in commercializing our potential products, we will incur significant additional
losses.
Our
proprietary rights may not adequately protect our intellectual property and product and if we cannot obtain adequate protection of our
intellectual property and product, we may not be able to successfully market our product.
Our
commercial success will depend in part on obtaining and maintaining intellectual property protection for our technologies and product.
We will only be able to protect our technologies and product from unauthorized use by third parties to the extent that valid and enforceable
patents cover them, or that other market exclusionary rights apply. While we have issued enforceable patents covering the sugarBEAT®
device, the patent positions of companies like ours can be highly uncertain and involve complex legal and factual questions for which
important legal principles remain unresolved. No consistent policy regarding the breadth of claims allowed in such companies’ patents
has emerged to date in Great Britain and the European Union. The general patent environment outside the United States involves significant
uncertainty. Accordingly, we cannot predict the breadth of claims that may be allowed or that the scope of these patent rights would provide
a sufficient degree of future protection that would permit us to gain or keep our competitive advantage with respect to this product and
technology. Additionally, companies like ours are dependent on creating a pipeline of products. We may not be able to develop additional
proprietary technologies or products that produce commercially viable products or that are themselves patentable.
Our
issued patents may be subject to challenge and possibly invalidated by third parties. Changes in either the patent laws or in the interpretations
of patent laws in Great Britain or the European Union or other countries may diminish the market exclusionary ability of our intellectual
property.
In
addition, others may independently develop similar or alternative technologies that may be outside the scope of our intellectual property.
Should third parties obtain patent rights to similar technology, this may have an adverse effect on our business.
To
the extent that consultants or key employees apply technological information independently developed by them or by others to our product,
disputes may arise as to the proprietary rights of the information, which may not be resolved in our favor. Consultants and key employees
that work with our confidential and proprietary technologies are required to assign all intellectual property rights in their discoveries
to us. However, these consultants or key employees may terminate their relationship with us, and we cannot preclude them indefinitely
from dealing with our competitors. If our trade secrets become known to competitors with greater experience and financial resources, the
competitors may copy or use our trade secrets and other proprietary information in the advancement of their products, methods or technologies.
If we were to prosecute a claim that a third party had illegally obtained and was using our trade secrets, it would be expensive and time
consuming and the outcome would be unpredictable. In addition, courts in Great Britain and the European Union are sometimes less willing
to protect trade secrets than courts in the United States. Moreover, if our competitors independently develop equivalent knowledge, we
would lack any contractual claim to this information, and our business could be harmed.
Our
ability to commercialize our product will depend on our ability to sell such products without infringing the patent or proprietary rights
of third parties. If we are sued for infringing intellectual property rights of third parties, such litigation will be costly and time
consuming and an unfavorable outcome would have a significant adverse effect on our business.
Our
ability to commercialize our product will depend on our ability to sell such products without infringing the patents or other proprietary
rights of third parties. Third-party intellectual property in the field of diagnostic medical devices is complicated, and third-party
intellectual property rights in this field are continuously evolving. We have not performed searches for third-party intellectual property
rights that may raise freedom-to-operate issues, and we have not obtained legal opinions regarding commercialization of our product other
than patent research prior to the filing of our patent applications, and search and examination reports from the respective patent examination
offices.
In
addition, because patent applications are published months after their filing, and because applications can take several years to issue,
there may be currently pending third-party patent applications that are unknown to us, which may later result in issued patents. If a
third-party claim that we infringe on its patents or other proprietary rights, we could face a number of issues that could seriously harm
our competitive position, including:
| |
– |
infringement claims that, with or without merit, can be costly and time consuming to litigate, can delay the regulatory approval process and can divert management’s attention from our core business strategy; |
| |
– |
substantial damages for past infringement which we may have to pay if a court determines that our products or technologies infringe upon a competitor’s patent or other proprietary rights; |
| |
– |
if a license is available from a holder, we may have to pay substantial royalties or grant cross licenses to our patents or other proprietary rights; and |
| |
– |
Re-designing our process so that it does not infringe the third-party intellectual property, which may not be possible, or which may require substantial time and expense including delays in bringing our own products to market. |
Such
actions could harm our competitive position and our ability to generate revenue and could result in increased costs.
If
our product, the sugarBEAT® device, does not gain market acceptance among physicians, patients and the medical
community, we will be unable to generate significant revenue, if any.
The
sugarBEAT® device that we developed may not achieve market acceptance among physicians, patients, third-party payers and others in
the medical community. If we receive the regulatory approvals necessary for commercialization, the degree of market acceptance will depend
upon a number of factors, including:
| |
– |
limited indications of regulatory approvals; |
| |
– |
the establishment and demonstration in the medical community of the clinical efficacy and safety of our product and its potential advantages over existing diagnostic medical devices; |
| |
– |
the prevalence and severity of any side effects; |
| |
– |
our ability to offer our product at an acceptable price; |
| |
– |
the relative convenience and ease of use of our product; |
| |
– |
the strength of marketing and distribution support; and |
| |
– |
sufficient third-party coverage or reimbursement. |
The
market may not accept the sugarBEAT® device based on any number of the above factors. If the sugarBEAT® device is approved, there
may be other therapies available which directly compete for the same target market. The market may choose to continue utilizing the existing
products for any number of reasons, including familiarity with or pricing of these existing products. The failure of any of our products
to gain market acceptance could impair our ability to generate revenue, which could have a material adverse effect on our future business.
We
have outsourced the bulk of the commercial manufacturing operations for the various components of the sugarBEAT®, with
the exception of the Sensor chemistry which is being conducted in-house. The failure to find manufacturing partners or expand our internal
manufacturing facility could have an adverse impact on our ability to grow our business.
We
are largely dependent on third parties to supply our product according to our specifications, in sufficient quantities, on time, in compliance
with appropriate regulatory standards and at competitive prices. We cannot be sure that we will be able to obtain an adequate supply of
our product candidates on acceptable terms, or at all.
Manufacturers
supplying diagnostic medical devices must comply with regulations which require, among other things, compliance with evolving regulations
under Medical Device Directives stipulated under ISO13485. The manufacturing of products at any facility will be subject to strict quality
control, testing and record keeping requirements, and continuing obligations regarding the submission of safety reports and other post-market
information. Both the sensor and patch manufacturing facilities for the sugarBEAT® device are currently ISO13485 certified. We cannot
guarantee that the facilities will continue to pass regulatory inspection, or that future changes to ISO13485 standards will not also
affect the manufacture of the sensors and patches.
If
we fail to attract and retain senior management, consultants, advisors and scientific and technical personnel, our product development
and commercialization efforts could be impaired.
Our
performance is substantially dependent on the performance of our senior management and key scientific and technical personnel, particularly
Dr. Dewan Fazlul Hoque Chowdhury, President, Chairman and Chief Executive Officer. The loss of the services of any member of our senior
management or our scientific or technical staff may significantly delay or prevent the development of our product and other business objectives
by diverting management’s attention to transition matters and identification of suitable replacements, if any, and could have a
material adverse effect on our business, operating results and financial condition.
We
also rely on consultants and advisors to assist us in formulating our research and development strategy. All of our consultants and advisors
are either self-employed or employed by other organizations, and they may have conflicts of interest or other commitments, such as consulting
or advisory contracts with other organizations, that may affect their ability to contribute to us.
In
addition, we believe that we will need to recruit additional executive management and scientific and technical personnel. There is currently
intense competition for skilled executives and employees with relevant scientific and technical expertise, and this competition is likely
to continue. The inability to attract and retain sufficient scientific, technical and managerial personnel could limit or delay our product
development efforts, which would adversely affect the development of our product and commercialization of our potential product and growth
of our business.
We
expect to expand our marketing capabilities and, as a result of which we may encounter difficulties in managing our growth, which could
disrupt our operations.
We
expect to have growth in expenditures, the number of our employees and the scope of our operations, in particular with respect to those
potential products that we elect to commercialize independently or together with others. To manage our anticipated future growth, we must
continue to implement and improve our managerial, operational and financial systems, expand our facilities and continue to train qualified
personnel. Due to our limited resources, we may not be able to effectively manage the expansion of our operations or train additional
qualified personnel. The physical expansion of our operations may lead to significant costs and may divert our management and business
development resources. Any inability to manage growth could delay the execution of our business plan or disrupt our operations.
Fluctuations
in foreign exchange rates may adversely affect our financial condition and results of operations.
Our
functional currency is the Great Britain Pound Sterling (“GBP”). The reporting currency is the United States dollar (U.S.$).
Income and expenditures are translated at the appropriate weighted average exchange rates prevailing during the reporting period. Assets
and liabilities are translated at the exchange rates as of balance sheet date. Stockholders’ equity is translated into United States
dollars from GBP at historical exchange rates. Currency fluctuations and restrictions on currency exchange may adversely affect our business,
including limiting our ability to convert GBP into foreign currencies and, if the GBP were to decline in value, reducing our revenue in
U.S. dollar terms. To the extent the U.S. dollar strengthens against foreign currencies, the translation of these foreign currencies denominated
transactions results in reduced revenue, operating expenses and net income for our international operations. Similarly, to the extent
the U.S. dollar weakens against foreign currencies, the translation of these foreign currency denominated transactions results in increased
revenue, operating expenses and net income for our international operations. We are also exposed to foreign exchange rate fluctuations
as we convert the financial statements of our foreign subsidiaries into U.S. dollars in consolidation. If there is a change in foreign
currency exchange rates, the conversion of the foreign subsidiaries’ financial statements into U.S. dollars will lead to a translation
gain or loss which is recorded as a component of other comprehensive income (loss). We have not entered into agreements or purchased instruments
to hedge our exchange rate risks. The availability and effectiveness of any hedging transaction may be limited, and we may not be able
to successfully hedge our exchange rate risks.
In
addition, a number of events have occurred in recent years, including the UK’s Brexit vote to leave the EU, the impact of Covid-19,
and the invasion of Ukraine by Russia, that have had significant and potentially lasting effect of both the global economic outlook as
well as a weakening of GBP against many currencies. We expect to have to pay some of our service providers and vendors in U.S.$ which
given the exchange rate impact and knock on inflationary pressure, will represent a significant increase in costs to the business compared
to prior years. The currency exchange rate continues to be very unstable and therefore the future impact or further weakening of GBP is
not known at this time.
Our business, financial condition and results
of operations may be materially adversely affected by global health epidemics, including the recent COVID-19 outbreak.
The outbreak of COVID-19 in December 2019 has since
rapidly increased its exposure globally. On March 11, 2020, the World Health Organization declared the outbreak a pandemic. A regional
or global health pandemic, including COVID-19, could severely affect our business, results of operations and financial condition. A regional
or global health pandemic, depending upon its duration and severity, could have a material adverse effect on our business. For example,
the COVID-19 pandemic has had numerous effects on the global economy and governmental authorities around the world have implemented measures
to reduce the spread of COVID-19. These measures, including shutdowns and “shelter-in-place” orders suggested or mandated
by governmental authorities or otherwise elected by companies as a preventive measure, have adversely affected workforces, customers,
consumer sentiment, economies and financial markets, and, along with decreased consumer spending, have led to an economic downturn in
many of our markets.
As a result of the COVID-19 pandemic, we evaluated
and executed the steps available to us to ensure we were able to provide protection of our employees and instigated remote working where
possible combined with following all government advice and guidance regarding any engagement within the workplace that could not be completed
remotely. To date this transition to remote working has had little impact on our employee productivity and has not caused any interruption
to our business. Due to the uncertainty of COVID-19, we will continue to assess the situation, including abiding by any government-imposed
restrictions, as and where relevant.
We are unable to accurately predict the impact
that COVID-19 will have on our operations going forward due to uncertainties that will be dictated by the length of time that the pandemic
and related disruptions continue, the impact of governmental regulations that might be imposed in response to the pandemic and overall
changes in consumer behavior. During this period, we, along with other companies, were notified by the FDA in the U.S., that our PMA application
for sugarBEAT® would be delayed due to the prioritization being given to COVID-19 related applications and resource activity. The
PMA review resumed as of April 15, 2021, and this is now ongoing and in-progress. In December 2021 the FDA’s Bio-monitoring research
division conducted an audit of the clinical program submitted in support of the PMA application. A single 483 observation was raised and
the Company submitted a full response in January 2022, intended to cover the FDA’s Quality System/Current Good Manufacturing Practice
regulations for Medical Devices (21 CFR Part 820).
We continue to monitor the impact of COVID-19
on our own operations and are working with our employees, suppliers, and other stakeholders to mitigate the risks posed by its spread,
but COVID-19 is not expected to have any long-term detrimental effect on the Company’s success. While key suppliers have not always
been accessible throughout the whole period of the outbreak, we have been able to be flexible in our priorities and respond favorably
to the challenges faced during this period. We also recognize that one of the consequences of this pandemic has been a surge in the uptake
of technologies for remote monitoring of patients and patient self-monitoring, which potentially enhances the prospects for the Company,
its CGM product and its planned digital healthcare offering.
To the extent the COVID-19
pandemic adversely affects our business and financial results, it may also have the effect of heightening many of the other risks described
in this “Risk factors” section.
Risks
Related to Our Industry
Our
competitors may develop products that are less expensive, safer or more effective, which may diminish or eliminate the commercial success
of any potential products that we may commercialize.
If
our competitors market products that are less expensive, safer or more effective than our future products developed from our product candidates,
or that reach the market before our products, we may not achieve commercial success. For example, if approved, the sugarBEAT® device’s
primary competition in the glucose monitoring device setting will be companies such as Dexcom, Abbott, and Senseonics who produce glucose
monitoring devices. The market may choose to continue utilizing the existing products for any number of reasons, including familiarity
with or pricing of these existing products. The failure of our product to compete with products marketed by our competitors would impair
our ability to generate revenue, which would have a material adverse effect on our future business, financial condition and results of
operations.
We
expect to compete with several companies including Dexcom, Abbott, and Senseonics, and our competitors may:
| |
– |
develop and market products that are less expensive or more effective than our future product; |
| |
– |
commercialize competing products before we can launch any products developed from our product candidate; |
| |
– |
operate larger research and development programs or have substantially greater financial resources than we do; |
| |
– |
initiate or withstand substantial price competition more successfully than we can; |
| |
– |
have greater success in recruiting skilled technical and scientific workers from the limited pool of available talent; |
| |
– |
more effectively negotiate third-party licenses and strategic relationships; and |
| |
– |
take advantage of acquisition or other opportunities more readily than we can. |
We
expect to compete for market share against large medical diagnostic device manufacturing companies, smaller companies that are collaborating
with larger companies, new companies, and other public and private research organizations.
In
addition, our industry is characterized by rapid technological change. Because our research approach integrates many technologies, it
may be difficult for us to stay abreast of the rapid changes in each technology. If we fail to stay at the forefront of technological
change, we may be unable to compete effectively. Our competitors may render our technologies obsolete by advances in existing technological
approaches or the development of new or different approaches, potentially eliminating the advantages in our product discovery process
that we believe we derive from our research approach and proprietary technologies.
The
use of hazardous materials in our operations may subject us to environmental claims or liabilities.
Our
research and development activities involve the use of hazardous chemical materials. Injury or contamination from these materials may
occur and we could be held liable for any damages, which could exceed our available financial resources. This liability could materially
adversely affect our business, financial condition and results of operations.
We
are subject to laws and regulations governing the use, manufacture, storage, handling and disposal of hazardous materials and waste products.
We may be required to incur significant costs to comply with environmental laws and regulations in the future that could materially adversely
affect our business, financial condition and results of operations.
If
we fail to comply with extensive regulations enforced by regulatory agencies with respect to diagnostic medical device products, the commercialization
of our product could be prevented, delayed or halted.
Research,
preclinical development, clinical trials, manufacturing and marketing of our product is subject to extensive regulation by various government
authorities. We have not received marketing approval for the sugarBEAT® device in all of our target markets. The process of obtaining
the required regulatory approvals is lengthy and expensive, and the time required for such approvals is uncertain. The approval process
is affected by such factors as:
| |
– |
the indication and claims of the diagnostic device; |
| |
– |
the quality of submission relating to the product; |
| |
– |
the product’s clinical efficacy and safety; |
| |
– |
the manufacturing facility compliance; |
| |
– |
the availability of alternative devices; |
| |
– |
the risks and benefits demonstrated in clinical trials; and |
| |
– |
the patent status and marketing exclusivity rights of certain innovative products. |
Any
regulatory approvals that we or our partners receive for our product may also be subject to limitations on the indicated uses for which
the product may be marketed or contain requirements for potentially costly post-marketing follow-up studies. The subsequent discovery
of previously unknown problems with the product, including adverse events of unanticipated severity or frequency, may result in restrictions
on the marketing of the product and withdrawal of the product from the market.
Manufacturing,
labelling, storage and distribution activities also are subject to strict regulation and licensing by government authorities. The manufacturing
facilities for our product will be subject to periodic inspection by the regulatory authorities and from time to time, these agencies
may send notice of deficiencies as a result of such inspections. Our failure, or the failure of our manufacturing facilities, to continue
to meet regulatory standards or to remedy any deficiencies could result in corrective action by the authorities, including the interruption
or prevention of marketing, closure of our manufacturing facilities, and fines or penalties.
Regulatory
authorities also will require post-marketing surveillance to monitor and report potential adverse effects of our product. If approved,
any of our products’ subsequent failure to comply with applicable regulatory requirements could, among other things, result in warning
letters, fines, suspension or revocation of regulatory approvals, product recalls or seizures, operating restrictions, injunctions and
criminal prosecutions.
Government
policies may change, and additional government regulations may be enacted that could prevent or delay regulatory approval of our product.
We cannot predict the likelihood, nature or extent of adverse government regulation that may arise from future legislation or administrative
action. If we are not able to maintain regulatory compliance, we might not be permitted to market our product and our business could suffer.
In
the future, we hope to distribute and sell our product outside of the United Kingdom and the European Union, which will subject us to
further regulatory risk.
In
addition to maintaining approval from the United Kingdom and the European Union for the sugarBEAT® device, we may seek regulatory
approval from Saudi Arabia and the United Arab Emirates, Hong Kong, Australia, and the U.S., to market the sugarBEAT® device, however,
there is no guarantee we will do so. We may in the future also seek approvals for additional countries. The regulatory review process
varies from country to country, and approval by foreign government authorities is unpredictable, uncertain and generally expensive. The
ability to market our product could be substantially limited due to delays in receipt of, or failure to receive, the necessary approvals
or clearances. Marketing of our product in these countries, and in most other countries, is not permitted until we have obtained required
approvals or exemptions in each individual country. Failure to obtain necessary regulatory approvals could impair our ability to generate
revenue from international sources.
Market
acceptance of our product will be limited if users are unable to obtain adequate reimbursement from third-party payers.
Government
health administration authorities, private health insurers and other organizations generally provide reimbursement for products like our
product and our commercial success will depend in part on these third-party payers agreeing to reimburse patients for the costs of our
product. Even if we succeed in bringing our product to market, we cannot assure you that third-party payers will consider our product
cost effective or provide reimbursement in whole or in part for its use.
Significant
uncertainty exists as to the reimbursement status of newly approved health care products. Our product is intended to replace or alter
existing therapies or procedures. These third-party payers may conclude that our product is less safe, effective or cost-effective than
existing therapies or procedures. Therefore, third-party payers may not approve our product for reimbursement.
If
third-party payers do not approve our product for reimbursement or fail to reimburse for them adequately, sales will suffer as some physicians,
or their patients will opt for a competing product that is approved for reimbursement or is adequately reimbursed. Even if third-party
payers make reimbursement available, these payers’ reimbursement policies may adversely affect our ability and the ability of our
potential collaborators to sell our product on a profitable basis.
The
trend toward managed healthcare, the growth of organizations such as health maintenance organizations and legislative proposals to reform
healthcare and government insurance programs could significantly influence the purchase of healthcare services and products, resulting
in lower prices and reduced demand for our product which could adversely affect our business, financial condition and results of operations.
In
addition, legislation and regulations affecting the pricing of our product may change in ways adverse to us before or after the regulatory
agencies approve our product for marketing. While we cannot predict the likelihood of any of these legislative or regulatory proposals,
if any government or regulatory agencies adopt these proposals, they could materially adversely affect our business, financial condition
and results of operations.
Product
liability claims may damage our reputation and, if insurance proves inadequate, the product liability claims may harm our business.
As
with other companies in our field, we may be exposed to the risk of product liability claims that is inherent in the diagnostic medical
device sector. A product liability claim may damage our reputation by raising questions about our product’s safety and efficacy
and could limit our ability to sell our product by preventing or interfering with commercialization of our product.
In
addition, product liability insurance for our industry is generally expensive to the extent it is available at all. There can be no assurance
that we will be able to maintain such insurance on acceptable terms or that we will be able to secure increased coverage as the commercialization
of our product progresses, or that future claims against us will be covered by our product liability insurance. Moreover, there can be
no assurance that any product liability coverage from any insurance policy and/or any rights of indemnification and contribution that
we have in place currently will offset any / all future claims. A successful claim against us with respect to uninsured liabilities and
not subject to any indemnification or contribution could have a material adverse effect on our business, financial condition, and results
of operations.
We
could be negatively impacted by the application or enforcement of fraud and abuse laws, including anti-kickback laws and other anti-referral
laws.
We
are not aware of any current business practice which is in violation of any fraud and abuse law. However, continued vigilance to assure
compliance with all potentially applicable laws will be a necessary expense associated with product development. For example, all product
marketing efforts must be strictly scrutinized to assure that they are not associated with improper remunerations to referral sources
in violation of any anti-kickback statutes. Remunerations may include potential future activities for our product, including discounts,
rebates and bundled sales, which must be appropriately structured to take advantage of statutory and regulatory “safe harbors”.
From time to time we may engage physicians in consulting activities. In addition, we may decide to sponsor continuing medical education
activities for physicians or other medical personnel. We may also award or sponsor study grants to physicians from time to time. All relationships
with physicians, including consulting arrangements, continuing medical education and study grants, must be similarly reviewed for compliance
with any anti-kickback statute to assure that remuneration is not provided in return for referrals. Patient inducements may also be unlawful.
Inaccurate reports of product pricing, or a failure to provide a product at an appropriate price to various governmental entities, could
also serve as a basis for an enforcement action under various theories.
Claims
which are “tainted” by virtue of kickbacks or a violation of self-referral rules may be alleged as false claims if other elements
of a violation are established. Because our potential customers may seek payments from healthcare programs for our product, even during
the clinical trial stages, we must assure that we take no actions which could result in the submission of false claims. For example, free
product samples which are knowingly or with reckless disregard billed to healthcare programs could constitute false claims. If the practice
was facilitated or fostered by us, we could be liable. Moreover, inadequate accounting for or a misuse of grant funds used for product
research and development could be alleged as a violation of relevant statutes.
The
risk of our being found in violation of these laws is increased by the fact that many of them have not been fully interpreted by the regulatory
authorities or the courts, and their provisions are open to a variety of interpretations, and additional legal or regulatory change.
Risks
Related to Our Common Stock, the Offering and Lack of Liquidity
Our common stock
may be delisted from The Nasdaq Capital Market if we cannot maintain compliance with Nasdaq’s continued listing requirements.
Our common stock is listed on the Nasdaq Capital Market.
There are a number of continued listing requirements that we must satisfy in order to maintain our listing on the Nasdaq Capital Market.
On April 2, 2020, the Company received a written notice
(the “Notice”) from staff of Listing Qualifications of The Nasdaq Stock Market LLC (“Nasdaq”) indicating that
the Company has not complied with the requirements of Nasdaq Listing Rule 5620(a) of Nasdaq’s listing rules due to its failure to
hold an annual meeting of stockholders within 12 months of the end of the Company’s fiscal year ended December 31, 2018. The Company
had 45 calendar days to submit a plan to regain compliance and, if Nasdaq accepted the plan, Nasdaq could grant an exception of up to
180 calendar days from the fiscal year end, or until September 28, 2020, to regain compliance. As discussed with Nasdaq, the Company filed
and mailed its proxy materials relating to its annual meeting on April 10, 2020 and held such annual meeting on May 15, 2020. As a result,
the Company regained compliance with Nasdaq Listing Rule 5620(a), and that matter is now closed.
We cannot assure you our securities will meet the
continued listing requirements to be listed on Nasdaq in the future. If Nasdaq delists our common stock from trading on its exchange,
we could face significant material adverse consequences including:
| |
• |
a limited availability of market quotations for our securities; |
| |
• |
a determination that our common stock is a “penny stock” which will require brokers trading in our common stock to adhere to more stringent rules and possibly resulting in a reduced level of trading activity in the secondary trading market for our common stock; |
| |
• |
a limited amount of news and analyst coverage for our company; and |
| |
• |
a decreased ability to issue additional securities or obtain additional financing in the future. |
If we fail to maintain compliance with all applicable
continued listing requirements for the Nasdaq Capital Market and Nasdaq determines to delist our common stock, the delisting could adversely
affect the market liquidity of our common stock, our ability to obtain financing to repay debt and fund our operations.
Our common stock may be affected by limited
trading volume and may fluctuate significantly.
Our common stock is traded on the Nasdaq Capital Market.
Although an active trading market has developed for our common stock, there can be no assurance that an active trading market for our
common stock will be sustained. Failure to maintain an active trading market for our common stock may adversely affect our shareholders’
ability to sell our common stock in short time periods, or at all. Our common stock has experienced, and may experience in the future,
significant price and volume fluctuations, which could adversely affect the market price of our common stock.
Our
stock price may be volatile.
The
stock market, particularly in recent years, has experienced significant volatility with respect to pharmaceutical, biotechnology and other
diagnostic medical device company stocks. The volatility of pharmaceutical, biotechnology and other diagnostic medical device company
stocks often does not relate to the operating performance of the companies represented by the stock. Factors that could cause this volatility
in the market price of our common stock include:
| |
– |
results from and any delays in our clinical trials; |
| |
– |
failure or delays in entering our product into clinical trials; |
| |
– |
failure or discontinuation of any of our research programs; |
| |
– |
delays in establishing new strategic relationships; |
| |
– |
delays in the development or commercialization of our product; |
| |
– |
market conditions in the diagnostic medical device sectors and issuance of new or changed securities analysts’ reports or recommendations; |
| |
– |
actual and anticipated fluctuations in our financial and operating results; |
| |
– |
developments or disputes concerning our intellectual property or other proprietary rights; |
| |
– |
introduction of technological innovations or new commercial products by us or our competitors; |
| |
– |
issues in manufacturing our product; |
| |
– |
market acceptance of our product; |
| |
– |
third-party healthcare reimbursement policies; |
| |
– |
regulatory actions affecting us or our industry; |
| |
– |
litigation or public concern about the safety of our product; and |
| |
– |
additions or departures of key personnel. |
These
and other external factors may cause the market price and demand for our common stock to fluctuate substantially, which may limit or prevent
investors from readily selling their shares of common stock and may otherwise negatively affect the liquidity of our common stock. In
the past, when the market price of a stock has been volatile, holders of that stock have instituted securities class action litigation
against the company that issued the stock. If any of our stockholders brought a lawsuit against us, we could incur substantial costs defending
the lawsuit. Such a lawsuit could also divert the time and attention of our management.
We
have not paid and may not pay any dividends on our common stock.
We
have paid no dividends on our common stock to date and may not pay dividends to holders of our common stock in the foreseeable future.
While our future dividend policy will be based on the operating results and capital needs of the business, it is currently anticipated
that any earnings will be retained to finance our future expansion and for the implementation of our business plan. As an investor, you
should take note of the fact that a lack of a dividend can further affect the market value of our stock and could significantly affect
the value of any investment in our Company.
We
are subject to the reporting requirements of federal securities laws. This can be expensive and may divert resources from other projects,
and thus impairing our ability to grow.
We
are subject to the information and reporting requirements of the Securities Exchange Act of 1934, as amended (the “Exchange Act”),
and other federal securities laws, including compliance with the Sarbanes-Oxley Act of 2002 (the “Sarbanes-Oxley Act”). The
costs of preparing and filing annual and quarterly reports, proxy statements and other information with the Securities and Exchange Commission
(“SEC”) (including reporting of any Merger that may occur in the future) and furnishing audited reports to stockholders will
cause our expenses to be higher than they would have been if we had remained privately held.
If
we fail to establish and maintain an effective system of internal control, we may not be able to report our financial results accurately
or to prevent fraud. Any inability to report and file our financial results accurately and timely could harm our reputation and adversely
impact the trading price of our common stock.
We
are subject to reporting obligations under the U.S. securities laws. The SEC, as required by Section 404 of the Sarbanes- Oxley Act (“SOX”),
adopted rules requiring every public company to include a management report on such company’s internal control over financial reporting
in its annual report, which contains management’s assessment of the effectiveness of the company’s internal control over financial
reporting.
Public
company compliance may make it more difficult to attract and retain officers and directors.
The
Sarbanes-Oxley Act and rules subsequently implemented by the SEC have required changes in corporate governance practices of public companies.
As a public company, we expect these rules and regulations to increase our compliance costs in 2021 and beyond and to make certain activities
more time consuming and costly. As a public company, we also expect that these rules and regulations may make it more difficult and expensive
for us to obtain director and officer liability insurance in the future and we may be required to accept reduced policy limits and coverage
or incur substantially higher costs to obtain the same or similar coverage. As a result, it may be more difficult for us to attract and
retain qualified persons to serve on our board of directors or as executive officers.
If our common stock
is delisted from the Nasdaq Capital Market and the price of our common stock remains below $5.00 per share, our common stock would come
within the definition of “penny stock”.
Our
common stock will be subject to the “penny stock” rules adopted under Section 15(g) of the Exchange Act. The penny stock rules
adopted under Section 15(g) of the Exchange Act generally apply to companies whose common stock is not listed on The Nasdaq Stock Market
or other national securities exchange and trades at less than $5.00 per share, other than companies that have had average revenue of at
least $6,000,000 for the last three years or that have tangible net worth of at least $5,000,000 ($2,000,000 if the company has been operating
for three or more years). These rules require, among other things, that brokers who trade penny stock to persons other than “established
customers” complete certain documentation, make suitability inquiries of investors and provide investors with certain information
concerning trading in the security, including a risk disclosure document and quote information under certain circumstances. Many brokers
have decided not to trade penny stocks because of the requirements of the penny stock rules and, as a result, the number of broker-dealers
willing to act as market makers in such securities is limited. If our common stock is delisted from the Nasdaq Capital Market and the
price of our common stock remains below $5.00 per share and our net tangible assets remain less than $2,000,000, our common stock would
come within the definition of “penny stock”. If we remain subject to the penny stock rules for any significant period, it
could have an adverse effect on the market, if any, for our securities. If our securities are subject to the penny stock rules, investors
will find it more difficult to dispose of our securities.
Offers
or availability for sale of a substantial number of shares of our common stock may cause the price of our common stock to decline.
If
our stockholders sell substantial amounts of our common stock in the public market upon the expiration of any statutory holding period,
under Rule 144, or issued upon the exercise of outstanding options or warrants, it could create a circumstance commonly referred to as
an “overhang” and in anticipation of which the market price of our common stock could fall. The existence of an overhang,
whether or not sales have occurred or are occurring, also could make more difficult our ability to raise additional financing through
the sale of equity or equity-related securities in the future at a time and price that we deem reasonable or appropriate.
The
interests of Dr. D.F.H. Chowdhury, or the controlling shareholders, may not always coincide with the interests of us and our other shareholders,
and the controlling shareholders may exert significant control or substantial influence over us and may take actions that are not in,
or may conflict with, public shareholders’ best interests.
The
controlling shareholders control the exercise of voting rights of over 50% of the shares eligible to vote in any of our annual or special
meetings. Therefore, these controlling shareholders will be able to exercise significant influence over all matters that require us to
obtain shareholder approval, including the election of directors to our board and approval of significant corporate transactions that
we may consider, such as a merger or other sale of our company or its assets. The controlling shareholders may cause us to take actions
that are not in, or may conflict with, the interests of us or the public shareholders. In the case where the interests of the controlling
shareholders conflict with those of our other shareholders, or if the controlling shareholders choose to cause us to pursue objectives
that would conflict with the interests of our other shareholders, such other shareholders could be left in a disadvantageous position
by such actions caused by the controlling shareholders and the price of our common stock could be adversely affected.
We
are subject to the anti-takeover provisions of the Nevada Revised Statutes governing business combinations and control share acquisitions.
Applicability
of the Nevada business combination statute would discourage parties interested in taking control of our company if they cannot obtain
the approval of our board of directors. These provisions could prohibit or delay a merger or other takeover or change in control attempt
and, accordingly, may discourage attempts to acquire our company even though such a transaction may offer our stockholders the opportunity
to sell their stock at a price above the prevailing market price.
The
effect of the Nevada control share statute is that the acquiring person, and those acting in association with the acquiring person, will
obtain only such voting rights in the control shares as are conferred by a resolution of the stockholders at an annual or special meeting
of the stockholders. The Nevada control share law, if applicable, could have the effect of discouraging takeovers of our company based
on our organizational structure.
We
are subject to compliance with multiple tax jurisdictions.
As
we transact out of both the UK and United States, we must comply with tax filing requirements in both jurisdictions.
USE OF PROCEEDS
We are registering 4,796,206 shares of common stock
for resale by the selling stockholders. We will not receive any proceeds from the sale of the shares offered by this prospectus.
However, we expect to receive approximately $9,592,412
in gross proceeds assuming the cash exercise of all of the warrants to purchase the 4,796,206 shares of common stock being registered
hereby at an exercise price of $2.00 per share of common stock. However, the warrants may be exercised on a cashless basis, in which case
we would not expect to receive any gross proceeds from the cash exercise of the warrants. We intend to use any net proceeds from the cash
exercise of the warrants for working capital and general corporate purposes.
DIVIDEND POLICY
We have never declared or paid any cash dividends
on our capital stock, and we do not currently intend to pay any cash dividends on our common stock for the foreseeable future. We expect
to retain future earnings, if any, to fund the development and growth of our business. Any future determination to pay dividends on our
common stock will be at the discretion of our board of directors and will depend upon, among other factors, our results of operations,
financial condition, capital requirements and any contractual restrictions.
CAPITALIZATION
The following table sets forth our capitalization
as of December 31, 2022 on an actual basis.
This table should be read in conjunction with the
information contained in this prospectus, including “Management’s Discussion and Analysis of Financial Condition and Results
of Operations,” and our financial statements and the related notes thereto which are incorporated by reference into this prospectus.
| | |
As
of December
31, 2022 | |
| | |
Actual | |
| | |
| (Unaudited) | |
| Cash, cash equivalents and short-term investments | |
$ | 7,340,840 | |
| | |
| | |
| Notes Payable | |
| 20,070,259 | |
| | |
| | |
| Stockholders’ equity: | |
| | |
| Common stock, $0.001 par value, 42,000,000 shares authorized and 24,103,196 shares issued and outstanding on an actual basis | |
| 24,103 | |
| Additional paid-in capital | |
| 38,296,198 | |
| Accumulated deficit | |
| (47,192,364 | ) |
| Accumulated other comprehensive (deficit) income | |
| (986,646 | ) |
| Total stockholders’ equity | |
| (9,858,709 | ) |
| Total capitalization | |
$ | 17,552,390 | |
MARKET PRICE FOR COMMON EQUITY AND RELATED
STOCKHOLDER MATTERS
Market Information
Our
common stock began quotation on the OTCBB under the symbol “NMRD” on November 4, 2014. On June 30, 2017, our common stock
began quotation on the OTCQB.
On January 25, 2018, the Company’s common stock
commenced trading on the NASDAQ Capital Market under its existing trading symbol, “NMRD”. For the periods indicated, the following
table sets forth the high and low bid prices per share of common stock. The following quotations reflect the high and low bids for our
shares of common stock based on inter-dealer prices, without retail mark-up, mark-down or commission and may not represent actual transactions.
| |
|
Bid Prices |
|
| |
|
Low |
|
|
High |
|
| FISCAL 2020 |
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
| First Quarter (April 1, 2019 through June 30, 2019) |
|
$ |
8.00 |
|
|
$ |
14.800 |
|
| Second Quarter (July 1, 2019 through September 30, 2019) |
|
$ |
7.06 |
|
|
$ |
11.000 |
|
| Third Quarter (October 1, 2019 through December 31, 2019) |
|
$ |
3.22 |
|
|
$ |
8.600 |
|
| Fourth Quarter (January 1, 2020 through March 31, 2020) |
|
$ |
2.50 |
|
|
$ |
10.400 |
|
| |
|
|
|
|
|
|
|
|
| FISCAL 2021 |
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
| First Quarter (April 1, 2020 through June 30, 2020) |
|
$ |
4.620 |
|
|
$ |
16.500 |
|
| Second Quarter (July 1, 2020 through September 30, 2020) |
|
$ |
3.117 |
|
|
$ |
10.660 |
|
| Third Quarter (October 1, 2020 through December 31, 2020) |
|
$ |
3.301 |
|
|
$ |
4.75 |
|
| Fourth Quarter (January 1, 2021 through March 31, 2021) |
|
$ |
3.71 |
|
|
$ |
12.59 |
|
| |
|
|
|
|
|
|
|
|
| FISCAL 2022 |
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
| First Quarter (April 1, 2021 through June 30, 2021) |
|
$ |
4.58 |
|
|
$ |
17.40 |
|
| Second Quarter (July 1, 2021 through September 30, 2021) |
|
$ |
5.72 |
|
|
$ |
10.726 |
|
| Third Quarter (October 1, 2021 through December 31, 2021) |
|
$ |
4.03 |
|
|
$ |
6.55 |
|
| Fourth Quarter (January 1, 2022 through March 31, 2022) |
|
$ |
3.51 |
|
|
$ |
4.85 |
|
| |
|
|
|
|
|
|
|
|
| FISCAL 2023 |
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
| First Quarter (April 1, 2022 through June 30, 2022) |
|
$ |
2.15 |
|
|
$ |
4.500 |
|
| Second Quarter (July 1, 2022 through September 30, 2022) |
|
$ |
1.500 |
|
|
$ |
2.605 |
|
| Third Quarter (October 1, 2022 through December 31, 2022) |
|
$ |
1.690 |
|
|
$ |
3.399 |
|
| Fourth Quarter (January 1, 2023 through March 9, 2023) |
|
$ |
0.870 |
|
|
$ |
3.430 |
|
On March 9, 2023, the closing price of our common
stock as reported on the Nasdaq Capital Market was $0.9068 per share.
As of February 28, 2022, there were approximately
28,899,402 shares of common stock issued and outstanding and 81 active record holders of our common stock. The number of record holders
does not include beneficial owners of common stock whose shares are held in the names of banks, brokers, nominees or other fiduciaries.
Dividends
We have never declared or paid any cash dividends
on our capital stock, and we do not currently intend to pay any cash dividends on our common stock for the foreseeable future. We expect
to retain future earnings, if any, to fund the development and growth of our business. Any future determination to pay dividends on our
common stock will be at the discretion of our board of directors and will depend upon, among other factors, our results of operations,
financial condition, capital requirements and any contractual restrictions.
Securities Authorized for Issuance Under Equity
Compensation Plans
We have not adopted an equity compensation plan.
Unregistered Sales of Securities
On January 31, 2023, the
Company completed the closing pursuant to a securities purchase agreement with two accredited institutional investors to purchase 4,796,206
shares of its common stock in a registered direct offering under the Form S-3 and warrants to purchase 4,796,206 shares of its common
stock at an exercise price of $2.00 per share in a concurrent private placement. The combined purchase price for one share of common stock
and one warrant is $1.75 for gross proceeds of $8,393,360.
Purchases of Equity Securities by the Registrant and Affiliated Purchasers
We have not repurchased any shares of our common stock.
PLAN OF DISTRIBUTION
The selling stockholders and any of their pledgees,
donees, transferees, assignees or other successors-in-interest may, from time to time, sell, transfer or otherwise dispose of any or all
of their shares of common stock or interests in shares of common stock on any stock exchange, market or trading facility on which the
shares are traded or in private transactions. These dispositions may be at fixed prices, at prevailing market prices at the time of sale,
at prices related to the prevailing market price, at varying prices determined at the time of sale, or at negotiated prices. These sales
may be affected in transactions, which may involve crosses or block transactions. The selling stockholders may use one or more of the
following methods when disposing of the shares or interests therein:
| |
● |
ordinary brokerage transactions and transactions in which the broker-dealer solicits purchasers; |
| |
|
|
| |
● |
block trades in which the broker-dealer will attempt to sell the shares as agent but may position and resell a portion of the block as principal to facilitate the transaction; |
| |
|
|
| |
● |
through brokers, dealers or underwriters that may act solely as agents; |
| |
|
|
| |
● |
purchases by a broker-dealer as principal and resale by the broker-dealer for its account; |
| |
|
|
| |
● |
an exchange distribution in accordance with the rules of the applicable exchange; |
| |
|
|
| |
● |
privately negotiated transactions; |
| |
|
|
| |
● |
through the writing or settlement of options or other hedging transactions entered into after the effective date of the registration statement of which this prospectus is a part, whether through an options exchange or otherwise; |
| |
|
|
| |
● |
settlement of short sales entered into after the effective date of the registration statement of which this prospectus is a part; |
| |
|
|
| |
● |
broker-dealers may agree with the selling stockholders to sell a specified number of such shares at a stipulated price per share; |
| |
|
|
| |
● |
a combination of any such methods of disposition; and |
| |
|
|
| |
● |
any other method permitted pursuant to applicable law. |
The selling stockholders may also sell shares under
Rule 144 under the Securities Act, if available, or Section 4(a)(1) under the Securities Act, rather than under this prospectus, provided
that they meet the criteria and conform to the requirements of those provisions.
If the selling stockholders effect such transactions
by selling shares of common stock to or through underwriters, broker-dealers or agents, such underwriters, broker-dealers or agents engaged
by the selling stockholders may arrange for other broker-dealers to participate in sales. Broker-dealers may receive commissions or discounts
from the selling stockholders (or, if any broker-dealer acts as agent for the purchaser of shares, from the purchaser) in amounts to be
negotiated. The selling stockholders do not expect these commissions and discounts to exceed what is customary in the types of transactions
involved.
The selling stockholders may, from time to time, pledge
or grant a security interest in some or all of the shares of common stock owned by them and, if they default in the performance of their
secured obligations, the pledgees or secured parties may offer and sell the shares of common stock from time to time under this prospectus,
or under a supplement or amendment to this prospectus under Rule 424(b)(3) or other applicable provision of the Securities Act amending,
if necessary, the list of selling stockholders to include the pledgee, transferee or other successors in interest as selling stockholders
under this prospectus.
Each selling stockholder has informed the Company
that it is not a registered broker-dealer and does not have any written or oral agreement or understanding, directly or indirectly, with
any person to distribute the common stock. If the Company is notified in writing by a selling stockholder that any material arrangement
has been entered into with a broker-dealer for the sale of common stock through a block trade, special offering, exchange distribution
or secondary distribution or a purchase by a broker or dealer, we will file a supplement to this prospectus, if required, pursuant to
Rule 424(b) under the Securities Act, disclosing (i) the name of each such selling stockholder and of the participating broker-dealer(s),
(ii) the number of shares involved, (iii) the price at which such shares of common stock were sold, (iv) the commissions paid or discounts
or concessions allowed to such broker-dealer(s), where applicable, (v) that such broker-dealer(s) did not conduct any investigation to
verify the information set out or incorporated by reference in this prospectus, and (vi) other facts material to the transaction. In addition,
upon being notified in writing by a selling stockholder that a donee or pledge intends to sell more than 500 shares of common stock, the
Company will file a supplement to this prospectus if then required in accordance with applicable securities law.
The selling stockholders also may transfer the shares
of common stock in other circumstances, in which case the transferees, pledgees or other successors in interest will be the selling beneficial
owners for purposes of this prospectus.
In connection with the sale of the shares of common
stock or interests in shares of common stock, the selling stockholders may enter into hedging transactions after the effective date of
the registration statement of which this prospectus is a part with broker-dealers or other financial institutions, which may in turn engage
in short sales of the common stock in the course of hedging the positions they assume. The selling stockholders may also sell shares of
common stock short after the effective date of the registration statement of which this prospectus is a part and deliver these securities
to close out their short positions, or loan or pledge the common stock to broker-dealers that in turn may sell these securities. The selling
stockholders may also enter into option or other transactions after the effective date of the registration statement of which this prospectus
is a part with broker-dealers or other financial institutions or the creation of one or more derivative securities which require the delivery
to such broker-dealer or other financial institution of shares offered by this prospectus, which shares such broker-dealer or other financial
institution may resell pursuant to this prospectus (as supplemented or amended to reflect such transaction).
The selling stockholders and any broker-dealers or
agents that are involved in selling the shares will be deemed to be “underwriters” within the meaning of the Securities Act
in connection with such sales. Broker-dealers may receive commissions or discounts from the Selling Stockholders (or, if any broker-dealer
acts as agent for the purchaser of securities, from the purchaser) in amounts to be negotiated, but, except as set forth in a supplement
to this Prospectus, in the case of an agency transaction not in excess of a customary brokerage commission in compliance with Financial
Industry Regulatory Authority (“FINRA”) Rule 2121; and in the case of a principal transaction a markup or markdown in compliance
with FINRA Rule 2121. In such event, any commissions received by such broker-dealers or agents and any profit on the resale of the shares
purchased by them may be deemed to be underwriting commissions or discounts under the Securities Act.
The Company has advised the selling stockholders that
they are required to comply with Regulation M promulgated under the Securities Exchange Act of 1934, as amended, during such time as they
may be engaged in a distribution of the shares. The foregoing may affect the marketability of the common stock.
The aggregate proceeds to the selling stockholders
from the sale of the common stock offered by them will be the purchase price of the common stock less discounts or commissions, if any.
Each of the selling stockholders reserves the right to accept and, together with their agents from time to time, to reject, in whole or
in part, any proposed purchase of common stock to be made directly or through agents. The Company will not receive any of the proceeds
from this offering.
The Company is required to pay all fees and expenses
incident to the registration of the shares. The Company has agreed to indemnify the selling stockholders against certain losses, claims,
damages and liabilities, including liabilities under the Securities Act or otherwise.
The Company has agreed with the selling stockholders
to keep the registration statement of which this prospectus constitutes a part effective until the earlier of (a) such time as all of
the shares covered by this prospectus have been disposed of pursuant to and in accordance with the registration statement, or (b) the
date on which the shares of common stock covered by this prospectus may be sold or transferred by non-affiliates without any volume limitations
or pursuant to Rule 144 of the Securities Act.
SELLING STOCKHOLDERS
We have prepared this prospectus to allow Armistice
Capital Master Fund Ltd. and Alyeska Master Fund, L.P., as selling stockholders, to offer for resale, from time to time, up to 4,796,206
shares of our common stock issuable to the selling stockholders upon exercise of certain warrants currently held by the respective selling
stockholders.
On January 27, 2023, the Company entered into a securities
purchase agreement with the selling stockholders, which are institutional accredited investors (the “Purchase Agreements”),
whereby, among other things, the Company issued and sold warrants (the “warrants”) exercisable for an aggregate of up to 4,796,206
shares of common stock (the “shares”) and an exercise price of $2.00 per share. The
warrants will be exercisable at the later of the effective date of shareholder approval or six months following the issue date and will
expire five years and six months following the issuance date.
The warrants and the shares of common stock issuable
thereunder were sold and issued without registration under the Securities Act of 1933, in reliance on the exemptions provided by Section
4(a)(2) of the Securities Act as transactions not involving a public offering and Rule 506 promulgated under the Securities Act as sales
to accredited investors.
The selling stockholders listed in the table below
may from time to time offer and sell any or all shares of our common stock set forth below pursuant to this prospectus. When we refer
to “selling stockholders” in this prospectus, we mean the persons listed in the table below, and the pledgees, donees, permitted
transferees, assignees, successors and others who later come to hold any of the selling stockholders’ interests in shares of our
common stock other than through a public sale.
The following table sets forth, as of the date of
this prospectus, the name of the selling stockholders for whom we are registering shares for resale to the public, and the number of such
shares that each such selling stockholder may offer pursuant to this prospectus. Applicable percentages are based on 28,899,402 shares
of common stock outstanding on February 28, 2023.
We cannot advise as to whether the selling stockholders
will in fact sell any or all of such shares. In addition, the selling stockholders may have sold, transferred or otherwise disposed of,
or may sell, transfer or otherwise dispose of, at any time and from time to time, the shares in transactions exempt from the registration
requirements of the Securities Act after the date on which they provided the information set forth on the table below.
| |
|
|
|
|
|
|
|
Shares
beneficially owned
after this
Offering(2) |
|
| Selling Stockholders(1) |
|
Number of Shares beneficially
owned through
February 28, 2023 |
|
|
Number of Shares Underlying
Warrants that may be sold in this Offering |
|
|
Number of
Shares |
|
|
Percentage of total outstanding
common stock |
|
| Armistice Capital Master Fund Ltd. (3) |
|
|
2,398,103 |
|
|
|
2,398,103 |
|
|
|
0 |
|
|
|
0 |
% |
| Alyeska Master Fund, L.P. (4) |
|
|
4,881,374 |
|
|
|
2,398,103 |
|
|
|
2,483,271 |
|
|
|
7.4 |
% |
| (1) |
If required, information about other selling stockholders, except for any future transferees, pledgees, donees or successors of the Selling Stockholder named in the table above, will be set forth in a prospectus supplement or amendment to the registration statement of which this prospectus is a part. Additionally, post-effective amendments to the registration statement will be filed to disclose any material changes to the plan of distribution from the description contained in the final prospectus. |
| (2) |
Assumes all shares offered by the selling stockholders hereby are sold and that the selling stockholders buys or sells no additional shares of common stock prior to the completion of this offering. |
| (3) |
Includes 2,398,103 shares of our common stock issuable upon exercise of the warrants held by Armistice Capital Master Fund Ltd. (“Master Fund”), which warrants are subject to, as applicable, certain beneficial ownership limitations, which provide that the holder of such warrants will not have the right to exercise any portion thereof if such holder, together with its affiliates, would beneficially own in excess of 4.99% of the number of shares of common stock outstanding immediately after giving effect to such exercise, provided that upon at least 61 days’ prior notice to us, such holder may increase or decrease such limitation up to a maximum of 9.99% of the number of shares of common stock outstanding. Steven Boyd, CIO of Armistice Capital, LLC (“Armistice”), has voting control and investment discretion over the securities held by Master Fund. Armistice and Steven Mr. Boyd disclaim beneficial ownership of the reported securities except to the extent of their respective pecuniary interest therein. Accordingly, notwithstanding the number of shares of common stock listed above as being beneficially owned by the Master Fund, Armistice and Mr. Boyd, each of the Master Fund, Armistice and Mr. Boyd further disclaim beneficial ownership of the shares of common stock issuable upon exercise of all of the warrants to the extent the number of shares of common stock beneficially owned by each of the Master Fund, Armistice and Mr. Boyd and any other person or entities with which their respective beneficial ownership would be aggregated for purposes of Section 13(d) of the Exchange Act would exceed 9.99% of the total number of shares of common stock outstanding. The address of Armistice Capital Master Fund Ltd. is 510 Madison Avenue, 7th Floor, New York, NY 10022. |
| (4) |
Includes (a) 2,483,271 shares of common stock directly held by Alyeska Master Fund, L.P. and (b) 2,398,103 shares of our common stock issuable upon exercise of the warrants held by Alyeska Master Fund, L.P., which warrants are subject to, as applicable, certain beneficial ownership limitations, which provide that the holder of such warrants will not have the right to exercise any portion thereof if such holder, together with its affiliates, would beneficially own in excess of 4.99% (or 9.99%, as applicable), of the number of shares of common stock outstanding immediately after giving effect to such exercise, provided that upon at least 61 days’ prior notice to us, such holder may increase or decrease such limitation up to a maximum of 9.99% of the number of shares of common stock outstanding. Alyeska Investment Group, L.P., the investment manager of Alyeska Master Fund, L.P., has voting and investment control of the shares held by Alyeska Master Fund, L.P. Anand Parekh is the Chief Executive Officer of Alyeska Investment Group, L.P. and may be deemed to be the beneficial owner of such shares. Mr. Parekh, however, disclaims any beneficial ownership of the shares held by Alyeska Master Fund, L.P. The registered address of Alyeska Master Fund, L.P. is at c/o Maples Corporate Services Limited, P.O. Box 309, Ugland House, South Church Street George Town, Grand Cayman, KY1-1104, Cayman Islands. Alyeska Investment Group, L.P. is located at 77 W. Wacker, Suite 700, Chicago IL 60601. |
MANAGEMENT’S DISCUSSION
AND ANALYSIS OF FINANCIAL CONDITION
AND RESULTS OF OPERATIONS
The following discussion and analysis summarizes
the significant factors affecting our operating results, financial condition, liquidity and cash flows of the Company as of and for the
periods presented below. The following discussion and analysis should be read in conjunction with our consolidated financial statements
and the related notes thereto included elsewhere in this prospectus. The discussion contains forward-looking statements that are based
on the beliefs of management, as well as assumptions made by, and information currently available to, our management. Actual results could
differ materially from those discussed in or implied by forward-looking statements as a result of various factors, including those discussed
below and elsewhere in this prospectus, particularly in the sections titled “Risk Factors” and “Cautionary Statement
Regarding Forward-Looking Statements”. When used herein, the terms the “Company,” “we,” “us”
and “our” refer to Nemaura Medical, Inc. and its consolidated subsidiaries.
Overview of the Company
We are a medical technology
company developing sugarBEAT®, a non-invasive glucose monitoring system for adjunctive
use by persons with diabetes, as well as for consumers in the non-diabetes space for monitoring metabolic health and general health and
wellness. sugarBEAT® consists of a disposable adhesive skin-patch connected to a rechargeable
wireless transmitter that displays glucose readings at regular five-minute intervals via a mobile app. sugarBEAT®
works by extracting glucose from the skin into a chamber in the patch that is in direct contact with an electrode-based sensor. The transmitter
sends the raw data to a mobile app where it is processed by an algorithm and displayed as a glucose reading after calibration, with the
ability to track and trend the data over 14 hours for each sensor wear period, including an initial warm-up period of 60-90 minutes on
average. Sensors can be worn as frequently as a user chooses. While sugarBEAT® requires a
minimum once per day calibration by the patient using a blood sample obtained by a finger stick, we believe sugarBEAT®
may be adopted by non-insulin dependent persons with diabetes alongside insulin-injecting persons with diabetes, who all perform multiple
daily finger sticks to manage their disease. In the consumer application, or non-medical applications, the relative glucose fluctuations
are used and therefore finger-prick calibrations are not required.
CE approval was granted
by the European Notified Body BSI in May 2019, allowing the product to be made available for commercial sale. This approval is subject
to an annual review of the underlying ISO 13485 accredited Quality Management System. The accreditation was successfully renewed in November
2021 and 2022. In conjunction with the UK Licensee, the Company commenced a phase 1 launch whereby devices were made available to limited
cohorts of users to gauge their feedback so that any fine-tuning could be completed prior to a mass market launch. The UK Licensee has
also confirmed that it plans to undertake Key Opinion Leader (“KOL”) studies in the UK for its white-labelled service offering
that is supported by sugarBEAT®. The KOL studies are intended to provide additional support
for the UK Licensee’s broader ongoing marketing plans and more specifically to support potential reimbursement for the device and
the licensees overall diabetes management offering(s)
The UK Licensee placed
an initial order for sugarBEAT® in April 2021 and provided a forecast for its post-launch
volume expectations, which the Company has used to establish both a short and medium term view to inform the Company’s commercial
operational requirements. In line with this view, the Company has taken the following actions since the initial order was received:
| |
· |
Entered into a new leased facility to provide the additional space requirements for commercial product assembly. |
| |
· |
Increased headcount of production operatives; this will be phased in line with the volume forecasts currently available, however the Company has also factored in an ability to scale further and faster should this be required. |
| |
· |
Moved forward with placing phased orders for raw materials to ensure future product availability to support both our UK Licensee while also providing for capacity to flex up further as other routes to market materialize in line with management’s commercialization program. |
| |
· |
Commenced phased deliveries in December 2021 to the UK Licensee of its continuous glucose monitor. |
In July 2020,
Nemaura filed a PMA application with the FDA to use sugarBEAT® as an adjunct to
finger prick testing for blood glucose trending. We, along with other applicants, were then informed by the FDA that the approval process
was currently subject to delays as a result of the FDA’s Center for Devices and Radiological Health (“CDRH”) being actively
engaged in responding to the current pandemic caused by COVID-19 which resulted in staff being reallocated to other approval requests
associated with COVID-19. During April 2021 the FDA confirmed that they would recommence their review of the PMA application and this
is now ongoing and in-progress. In December 2021 the FDA’s Bio-monitoring research division conducted an audit of the clinical program
submitted in support of the PMA application. A single 483 observation was raised, and the Company submitted a full response in January
2022. The FDA subsequently scheduled a pre-market inspection for the second calendar quarter of 2022, intended to cover the FDA’s
Quality System/Current Good Manufacturing Practice regulations for Medical Devices (21 CFR Part 820). This audit was conducted in
the first quarter of the fiscal year ending March 31, 2023. The Company reported that a single 483 observation was raised to
which the Company responded in a timely manner, and Dialogue with the FDA continues with respect to the PMA application.
In addition to this, Nemaura
established that proBEAT™, which is based on the sugarBEAT® platform, can be classified
under the Wellness guidance when it is used according to the FDA Wellness guidance notes, to provide prompts and educate users on factors
affecting their blood sugar profiles. Nemaura launched proBEAT™ in the U.S. in December 2020, as part of a diabetes prevention and
reversal program branded BEATdiabetes.life, in the form of pilot studies. During the quarter ended December 31, 2020, Nemaura licensed
a clinically validated weight loss program for the management of diabetes from Healthimation, LLC, which was originally developed at the
Joslin Diabetes Center, an affiliate of Harvard Medical School. This program, together with proBEAT™, originally formed the basis
of the BEATdiabetes.life program that is currently being developed for commercialization in the U.S. The program is under further refinement
based on feedback gathered to date. Further KOL studies are planned to provide additional marketing support of the program in preparation
for a broader U.S.-wide roll-out. While still in the relatively early stages, we are pleased with initial results and feedback received
from these user-groups and one key outcome has been that the company is now focusing on providing proBEAT as a standalone plug-in for
existing diabetes management programs, therefore potentially allowing the company to accelerate commercialization as well as reduce the
cost burden associated with running its own program.
We believe there are
additional applications for sugarBEAT® and the underlying BEAT technology platform,
which may include:
| |
· |
a web-server accessible by physicians and diabetes professionals to track the condition remotely, thereby reducing healthcare costs and managing the condition more effectively; |
| |
· |
a complete virtual doctor that monitors a person's vital signs and transmits results via the web; |
| |
· |
other patches using the BEAT technology platform to measure alternative analytes, including lactate, uric acid, lithium and drugs. This would be a step-change in the monitoring of conditions, particularly in the hospital setting. Lactate monitoring is currently used to determine the relative fitness of professional athletes and we completed preliminary studies demonstrating the application of the BEAT technology for continuous lactate monitoring; and |
| |
· |
a continuous temperature monitoring system which could have various applications, including use for individuals to monitor their temperature in connection with diagnosis and monitoring of symptoms of viral infections, and in females to monitor the ovulation cycle. |
During this period of product development, the
Company has experienced recurring losses and negative cash flows from operations. For the fiscal years ended March 31, 2022 and
2021, we generated $503,906 and $0 revenues from product sales and reported net losses of $13,886,805 and $6,258,596, respectively,
and negative cash flow from operating activities of $6,504,041 and $5,998,097, respectively. For the nine months ended December 31,
2022 and 2021, we generated $77,044 and $183,628 revenues from product sales and reported net losses of $9,460,888 and $10,269,557,
respectively, and negative cash flow from operating activities of $6,090,181 and $4,353,494, respectively. As noted in our financial
statements, as of December 31, 2022, we had a cash balance of $7,340,840, working capital deficit of $2,309,666, an accumulated
deficit of $47,192,364 and a deficiency in total stockholders' equity of $9,858,709. There is substantial doubt regarding our
ability to continue as a going concern as a result of our historical recurring losses, negative cash flows from operations, existing
debt of $20,070,259 due as of December 31, 2022, which will require the restructure of the debt or obtain additional, new, funding.
See “Risk Factors—We have a history of operating losses, our management has concluded that factors raise substantial
doubt about our ability to continue as a going concern and our auditor has included an explanatory paragraph relating to our ability
to continue as a going concern in its audit report for the fiscal years ended March 31, 2022 and 2021.”
While the Company expects
to continue to incur losses from operations for the near-term and these losses could be significant as product development, regulatory
activities, clinical trials, and other commercial and product development related expenses are incurred, the Company reached a significant
milestone during the three month period ended December 31, 2021, as the Company commenced commercial delivery of its sugarBEAT®
device to its UK Licensee, allowing them to continue studies dedicated to developing user based feedback and evidence that could
potentially support reimbursement in the UK.
Management's strategic
assessment continues to include the following potential options:
| |
· |
obtaining further regulatory approval for the sugarBEAT® device in other global territories, including the U.S., and the Middle East; |
| |
· |
signing new/additional licensing and collaboration opportunities beyond our existing licensee partners; |
| |
· |
pursuing further capital raising opportunities to support and accelerate the commercialization strategy; and |
| |
· |
developing the sugarBEAT® device platform for commercialization for other applications. |
RESULTS
OF OPERATIONS
Management’s
plans and basis of presentation
The
Company has experienced recurring losses and negative cash flows from operations. On December 31, 2022, the Company had cash balances
of $7,340,840, total stockholders’ equity of $9,858,709 and an accumulated deficit of $47,192,364. To date, the Company has in large
part relied on equity and debt financing to fund its operations. Initially additional funding also came from related party contributions.
The Company expects to continue to incur losses from operations for the near-term and these losses could be significant as the Company
implements and scales its product commercialization strategy.
Management’s
strategic assessment includes the following potential options:
| |
– |
support the UK and EU launch of sugarBEAT®; |
| |
– |
obtaining further regulatory approval for the sugarBEAT® device in other countries such as the U.S.; |
| |
– |
exploring licensing and partnership opportunities in other territories; |
| |
– |
developing the sugarBEAT® device for commercialization for other applications; and |
| |
– |
considering whether additional future capital raises can further enhance and accelerate the delivery of the Company’s strategic growth objectives. |
Results
of Operations
Comparative Results for
the Nine Months Ended December 31, 2022 and 2021
Revenue
The
Company recognized revenue of $77,044 in the nine month period ended December 31, 2022, relating to deliveries of sugarBEAT®
to MSW pursuant to the initial order placed in April 2021. A portion also related to the recognition of the GBP 1 million (approximately
$1.20 million), that was previously received and held within deferred revenue relating to the exclusive Marketing Rights Agreement that
was signed with MSW.
Revenue
recognized in the comparative nine month period ended December 30, 2021 was $183,628 relating to the delivery of goods and including a
small portion of the recognition of the deferred revenue relating to the exclusive Marketing Rights Agreement that was signed with MSW.
Research and Development
Expenses
Research
and development (“R&D”) expenses were $980,862 and $987,711 for the nine months ended December 31, 2022 and 2021, respectively.
This amount consisted primarily of expenditures on wages and sub-contractor activities incurred for improvements made to the sugarBEAT® device.
General
and Administrative Expenses
General
and administrative expenses were $4,329,306 and $4,151,380 for the nine months ended December 31, 2022 and 2021, respectively. These expenses
consisted of fees for legal, professional, consultancy, audit services, investor relations, insurance, advertising and general and operational
wages. Non-cash charges of $635,494 and $199,522 were recorded in the nine-month period ended December 31, 2022 and 2021 as a result of
the mark-to-market adjustments on the Company’s outstanding foreign currency forward contracts.
As
the Company continues to scale up to service its existing order book, it is expected that general and administrative expenses will continue
to increase in a similar way moving forward, as the business transitions to a more operational focused base that will encompass an increase
in functional expenses relating to production, sales, marketing, customer service, as well as enhancements to other existing functions.
Other Comprehensive
Loss
For the nine months ended
December 31, 2022 and 2021, other comprehensive loss was $864,328 and $142,922, respectively. Currently all transactions recorded through
other comprehensive loss arise from fluctuations in the USD: GBP exchange rate and the impact that this has on consolidation of the Company’s
non-USD denominated assets and liabilities.
Comparative Results for
the Three Months Ended December 31, 2022 and 2021
Revenue
Revenue
of $3,017 was recognized in the three month period ended December 31, 2022, relating to deliveries of sugarBEAT®
to MSW pursuant to the initial order placed in April 2021. A portion also related to the recognition of the deferred revenue relating
to the exclusive Marketing Rights Agreement that was signed with MSW.
The
comparative revenue recognized in the three month period ended December 31, 2021 was $183,628.
Research and Development
Expenses
R&D
expenses were $393,747 and $412,341 for the three month periods ended December 31, 2022 and 2021, respectively. This continues to be largely
composed of expenditures on wages and sub-contractor activities incurred in finalizing the product design for the sugarBEAT® device
in order to enable scaling of the production ability combined with costs associated with new pipeline products as they move through their
respective development phases.
General
and Administrative Expenses
General
and administrative expenses were $239,628 and $1,391,278 for the three month periods ended December 31, 2022 and 2021, respectively. Given
the nature of the Company’s activities has remained unchanged, the cost drivers in this area have also remained consistent and are
largely representative of fees for legal, professional, consultancy, audit services, investor relations, insurance and wages. Non-cash
credits of $990,531 and $70,878 were recorded for the mark-to-market adjustments on the Company’s outstanding foreign currency forward
contracts.
We
anticipate that general and administrative expenses will continue to increase moving forward, as the business transitions to a more operational
focused base that will encompass an increase in functions expenses associated with sales, marketing, customer service, as well as enhancements
to other existing functions.
Other Comprehensive
Loss
For
the three months ended December 31, 2022 and 2021, other comprehensive income was $556,080 and ($25,065) loss, respectively. Currently
all transactions recorded through other comprehensive loss arise from fluctuations in the USD:GBP exchange rate and the impact that this
has on consolidation of the Company’s non-USD denominated assets and liabilities.
Fiscal
Year Ended March 31, 2022 Compared to Fiscal Year Ended March 31, 2021
Revenue
December
2021 marked a pivotal milestone for the Company as the Company commenced deliveries of sugarBEAT® to MSW pursuant to the initial order
placed in April 2021. These deliveries continued in line with the schedule agreed with MSW during the remainder of the current fiscal
year.
While
the majority of the $503,906 revenue recognized related to the delivery of goods, a proportion also related to the recognition of the
GBP 1 million (approximately $1.32 million), that was previously received and held within deferred revenue, relating to the exclusive
Marketing Rights Agreement that was signed with MSW. We expect to record the remainder of the revenue over an approximately 10-year term
from the date sales to MSW commenced.
There
was no revenue recognized in the year ended March 31, 2021.
Research
and Development Expenses
Research
and development expenses were $1,556,988 and $1,554,603 for the fiscal years ended March 31, 2022 and 2021, respectively. The stabilization
in costs here being established as the Company’s historically more significant research and development expenditure relating to
clinical trials and improvements made to the sugarBEAT® device started to flatten out over the year. We expect that the sugarBEAT®
related research and development expenses will reduce in future periods as the product is launched, however the Company expects to continue
to incur research and development costs to both enhance, refine and extend the platform capabilities for alternative applications.
General
and Administrative Expenses
General
and administrative expenses were $6,173,049 and $3,032,138 for the fiscal years ended March 31, 2022 and 2021, respectively. These consisted
of fees for legal, professional, consultancy, audit services, investor relations, insurance, advertising and general and operational wages.
The increase in expenses being driven predominantly by increased wages, as additional headcount has been added to support the operational
scale up process across both our UK and U.S. teams. Increases have also been seen in insurance, rent and depreciation and amortization,
which are considered to be directly related to the commercialization steps undertaken during the period. In addition to this, a non-cash
item charge of $440,196 was booked as a result of the mark-to-market impact from the revaluation of the foreign currency forward contracts
in place as of the fiscal period end, along with a further non-cash charge of $133,529 in relation to the fair value of share options
issued to directors.
As
the Company continues to scale up to service its existing order book, it is expected that general and administrative expenses will continue
to exhibit a similar higher cost profile moving forward, as the business continues to transition to an operationally focused base that
is expected to result in increased functional expenses relating to production, sales, marketing, customer service, as well as enhancements
to other existing functions.
Other
Comprehensive Income
For
the fiscal years ended March 31, 2022 and 2021 other comprehensive income saw a charge of $257,885 versus a credit of $472,559, respectively,
arising from foreign currency translation adjustments.
Liquidity
and Capital Resources
We have experienced
net losses and negative cash flows from operations since our inception. We have sustained cumulative losses of $47,192,364 through December
31, 2022. We have historically financed our operations through a combination of debt and equity funding.
As of December 31,
2022, the Company had a deficiency in net working capital of $2,309,666, which included cash balances of $7,340,840. The Company reported
a net loss for the three and nine-month periods ended December 31, 2022 of $1,716,278 and $9,460,888, respectively. This loss is after
taking account of interest and debt discount charges arising from the note purchase agreements for the three- and nine- month periods
ended December 31, 2022 of $1,082,949 and $4,152,437, respectively.
Having reviewed
the Company’s forward looking cashflow requirements in relation to the cash balance held at December 31, 2022, management is aware
of the need to raise additional funds in order to finance the ongoing commercialization of sugarBEAT®. The Company had $7,340,840
of cash at December 31, 2022. The Company has debt on its balance sheet which will reach maturity in July 2024, following a restructure
in October 2022.
Ability
to Continue as a Going Concern
In evaluating
the going concern position of the Company, management has considered the ability of the Company to raise additional funding in combination
with one or more of the different funding options available to it at this time. Based on current and ongoing engagement with potential
funding providers, management believes that there is a reasonable expectation that funding could be provided by one, or more, of the following
options:
| |
· |
Equity funding – the Company has immediate access to funds through the ATM facility that is currently in place; in addition to this, the Company believes there are various alternative mechanisms available to the Company similar to those used previously e.g. direct sale of shares to interested third parties, similar to the stake sold to Tiger Trading Partners L.L.C. in February 2022, as well as other mechanisms to sell common stock via an underwritten agreement or the further exercise of warrants by the current warrant holders etc |
| |
· |
Debt funding – the Company continues to be in ongoing discussions with third party debt providers, including the incumbent, to enable the existing debt facility to be restructured or renewed, should management feel that this route offers a more attractive option compared to the sale of equity that is dependent on the current market conditions. |
| |
· |
Alternative funding as used in the past such as the sale of licenses. As product development is now at a significantly more advanced stage then it was, it is management’s belief that the sufficient funding could be provided through the sale of licenses or a large-scale partnership that could bring in additional funds and infrastructure to support the commercial growth ambitions of the Company. |
There can be
no assurance that any such equity, debt or alternative funding will be available on terms acceptable to the Company, or at all. As a consequence
of this funding requirement being triggered without the funding bridge having been put in place by the filing date of these unaudited
condensed consolidated financial statements, ASC 205-40 requires that management recognize and disclose this point as an event which creates
a substantial doubt as to the Company’s ability to continue as a going concern for at least one year from the date of filing of
these unaudited condensed consolidated financial statements.
Cash Flows
For the Nine-Months
Ended December 31, 2022 and 2021
Negative cash flow from operating
activities for the nine months ended December 31, 2022 was $6,090,181, reflecting a net loss of $9,460,888, adjusted for the add back
of the accretion of debt discount expense of $4,152,437, the mark-to-market charge booked in relation to the revaluation of the foreign
currency forward contracts of $635,494 and the depreciation and amortization charge of $268,595. Cash was also impacted by increases in
inventory of $864,636, which was directly driven as a result of commercial scale up.
Prepayments increased by
$467,070, which was a result of an increase in amount paid to Hamilton Court (approximately
$876,000), our forward contract provider, partially offset by savings on other prepayments, including a reduction in prepaid insurance.
There was a $34,897 increase
in accounts payable during the nine months ended December 31, 2022 with decreases seen in other liabilities and accrued expenses of $167,568
and deferred revenue of $297,419. The related party payable balance increased by $75,977.
Negative cash flow from operating activities for the nine months ended December 31, 2021 was $4,353,494, reflecting a net loss of $10,269,557, adjusted
for the add back of the non-cash amortization of debt discount expense of $5,141,701, the mark-to-market charge recorded in relation to
the foreign currency forward contracts of $199,522 and the depreciation and amortization charge of $139,751. Cash was also impacted by
increases in inventory of $533,656, which was directly driven as a result of the commercial scale up.
Prepayments
increased by $797,155, which was also a direct result of commercial scale up with prepayments for raw materials being pre-purchased to
ensure that inventory would be on-hand to support the Company’s shift to commercial operations. The decrease was partly offset by
an increase in accounts receivable – related party, of $301,387, which relates to additional advance purchasing of inventory that
is completed through a related party company.
There
was also a reduction in accounts payable of $77,075 with increases seen in both other liabilities and accrued expenses and deferred revenue.
Net
cash used in investing activities for the nine months ended December 31, 2022, was $438,805, which included patent filing costs of $135,168
and the purchase of property and equipment of $275,758 driven by the procurement to support the transition to operational production.
Net
cash used in investing activities was $880,008 for the nine months ended December 31, 2021, which reflected patent filing costs of $60,241,
the purchase of property and equipment of $359,301 driven by the procurement of cleanroom facilities and injection molding tooling to
support the operational production steps taken in advance of product delivery to the UK licensee. In addition to this, $460,466 was invested
in software development costs relating to the digital health program in the US and recent Beta launch of our consumer health program,
Miboko, in the UK.
Net
cash used in financing activities for the nine months ended December 31, 2022 was $3,273,859, comprised of $4,700,000 from proceeds from
the issuance of notes payable, offset by $7,974,282 for the repayments of notes payable.
Net
cash used in financing activities for the nine months ended December 31, 2021 was $3,421,933, comprised of $2,963,658 of proceeds from
the exercise of warrants, $114,409 of proceeds (net of costs) from the sale of common stock through the ATM facility, offset by $6,500,000
for the scheduled repayments of notes payable.
For
the Fiscal Years Ended March 31, 2022 and 2021
Negative cash flow from operating activities for the fiscal year ended March 31, 2022, was $6,504,041 which reflected the following key cashflow
movements: a net loss of $13,886,805 which included non-cash items booked as an expense relating to the accretion of the debt discount
($6,666,630), mark-to-market valuation of the foreign currency forward contracts that were held at fiscal year-end ($440,196), stock-based
compensation paid to an employee combined the fair value of options issued to directors ($220,917), and depreciation and amortization
($229,810).
Cashflows
were also impacted by increases in inventory of $637,149, and accounts receivable – related parties of $250,092 relating to the
acquisition of raw materials to support manufacture and delivery of product to our UK Licensee; the increase in inventory being largely
offset by the decrease in prepaid expenses of $519,346 vs the prior year end which reflected upfront payments for inventory at that time.
Working
capital cashflow was also impacted by a decrease in accounts payable of $117,384, due to timing of purchases and an increase in other
liabilities and accrued expenses of $310,490.
Negative cash flow from operating activities for the fiscal year ended March 31, 2021, was $5,998,097 which were driven by cashflow movements
relating to the following: A net loss of $6,258,596 which was partially offset by non-cash items booked as an expense relating to the
accretion of the debt discount ($2,007,687), stock-based compensation paid to third party suppliers ($113,171), and depreciation and amortization
($98,075). Cashflows were also impacted by an increase in inventory of $564,313, combined with and increase in prepaid expenses of $767,050,
and an increase in Accounts receivable – related party, as the Company geared up towards commercialization.
Net
cash used in investing activities was $956,482 for the fiscal year ended March 31, 2022, which reflected expenditure on property and equipment
to support the commencement of manufacture of product for sale during the year of $481,718, combined with ongoing spend on software development
($391,073) and patent costs ($83,691), to enhance the businesses digital offering and protect the intellectual property developed.
Net
cash used in investing activities was $836,440 for the fiscal year ended March 31, 2021, which reflected expenditure for software development
costs of $663,758, combined with costs related to patent filings of $81,952, and the purchase of property and equipment of $90,730.
Net
cash utilized in financing activities for the fiscal year ended March 31, 2022, was $6,368,315. This includes repayments made in relation
to debt funding of $12,400,000, which was partially offset by the proceeds from the issuance of common stock in relation to equity funding
was $3,118,792, with associated cash costs of $50,765, combined with the sale of warrants which provided a further $2,963,658 of cash
funding.
Net
cash provided by financing activities for the fiscal year ended March 31, 2021, was $37,986,392. Proceeds from the issuance of common
stock in relation to equity funding was $15,750,672 with associated cash costs of $957,193; the sale of warrants providing a further $400,503.
$25,000,000 was provided via the issuance of two notes payable during the year, with associated cash costs incurred of $1,525,035 while
repayments made were $600,000. $82,555 of cash expense was incurred in relation to concluding the full repayment of the insurance financing
arrangement.
Off-Balance
Sheet Arrangements
We
have no off-balance sheet arrangements, including unrecorded derivative instruments that have or are reasonably likely to have a current
or future material effect on our financial condition, changes in financial condition, revenues or expenses, results of operations, liquidity,
capital expenditures or capital resources.
Contractual
Obligations
None.
Critical
Accounting Policies and Significant Judgments and Estimates
Our
management’s discussion and analysis of our financial condition and results of operations is based on our audited consolidated financial
statements, which have been prepared in accordance with United States generally accepted accounting principles (“U.S. GAAP”).
The preparation of these consolidated financial statements requires us to make estimates and assumptions that affect the reported amounts
of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the consolidated financial statements,
as well as the reported results of operations during the reporting periods. Our estimates are based on our historical experience and on
various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments
about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results could differ from
these estimates under different assumptions or conditions. Our significant accounting policies are described in more detail in the notes
to our consolidated financial statements included elsewhere in this prospectus.
DESCRIPTION OF THE BUSINESS
Business
Overview
We are a medical technology
company developing sugarBEAT®, a non-invasive glucose monitoring system for adjunctive
use by persons with diabetes, as well as for consumers in the non-diabetes space for monitoring metabolic health and general health and
wellness. sugarBEAT® consists of a disposable adhesive skin-patch connected to a rechargeable
wireless transmitter that displays glucose readings at regular five-minute intervals via a mobile app. sugarBEAT®
works by extracting glucose from the skin into a chamber in the patch that is in direct contact with an electrode-based sensor. The transmitter
sends the raw data to a mobile app where it is processed by an algorithm and displayed as a glucose reading after calibration, with the
ability to track and trend the data over 14 hours for each sensor wear period, including an initial warm-up period of 60-90 minutes on
average. Sensors can be worn as frequently as a user chooses. While sugarBEAT® requires a
minimum once per day calibration by the patient using a blood sample obtained by a finger stick, we believe sugarBEAT®
may be adopted by non-insulin dependent persons with diabetes alongside insulin-injecting persons with diabetes, who all perform multiple
daily finger sticks to manage their disease. In the consumer application, or non-medical applications, the relative glucose fluctuations
are used and therefore finger-prick calibrations are not required.
CE approval was granted
by the European Notified Body BSI in May 2019, allowing the product to be made available for commercial sale. This approval is subject
to an annual review of the underlying ISO 13485 accredited Quality Management System. The accreditation was successfully renewed in November
2021 and 2022. In conjunction with the UK Licensee, the Company commenced a phase 1 launch whereby devices were made available to limited
cohorts of users to gauge their feedback so that any fine-tuning could be completed prior to a mass market launch. The UK Licensee has
also confirmed that it plans to undertake Key Opinion Leader (“KOL”) studies in the UK for its white-labelled service offering
that is supported by sugarBEAT®. The KOL studies are intended to provide additional support
for the UK Licensee’s broader ongoing marketing plans and more specifically to support potential reimbursement for the device and
the licensees overall diabetes management offering(s)
The UK Licensee placed
an initial order for sugarBEAT® in April 2021 and provided a forecast for its post-launch
volume expectations, which the Company has used to establish both a short and medium term view to inform the Company’s commercial
operational requirements. In line with this view, the Company has taken the following actions since the initial order was received:
| |
· |
Entered into a new leased facility to provide the additional space requirements for commercial product assembly. |
| |
· |
Increased headcount of production operatives; this will be phased in line with the volume forecasts currently available, however the Company has also factored in an ability to scale further and faster should this be required. |
| |
· |
Moved forward with placing phased orders for raw materials to ensure future product availability to support both our UK Licensee while also providing for capacity to flex up further as other routes to market materialize in line with management’s commercialization program. |
| |
· |
Commenced phased deliveries in December 2021 to the UK Licensee of its continuous glucose monitor. |
In July 2020,
Nemaura filed a PMA application with the FDA to use sugarBEAT® as an adjunct to
finger prick testing for blood glucose trending. We, along with other applicants, were then informed by the FDA that the approval process
was currently subject to delays as a result of the FDA’s Center for Devices and Radiological Health (“CDRH”) being actively
engaged in responding to the current pandemic caused by COVID-19 which resulted in staff being reallocated to other approval requests
associated with COVID-19. During April 2021 the FDA confirmed that they would recommence their review of the PMA application and this
is now ongoing and in-progress. In December 2021 the FDA’s Bio-monitoring research division conducted an audit of the clinical program
submitted in support of the PMA application. A single 483 observation was raised, and the Company submitted a full response in January
2022. The FDA subsequently scheduled a pre-market inspection for the second calendar quarter of 2022, intended to cover the FDA’s
Quality System/Current Good Manufacturing Practice regulations for Medical Devices (21 CFR Part 820). This audit was conducted in
the first quarter of the fiscal year ending March 31, 2023. The Company reported that a single 483 observation was raised to
which the Company responded in a timely manner, and Dialogue with the FDA continues with respect to the PMA application.
In addition to this, Nemaura
established that proBEAT™, which is based on the sugarBEAT® platform, can be classified
under the Wellness guidance when it is used according to the FDA Wellness guidance notes, to provide prompts and educate users on factors
affecting their blood sugar profiles. Nemaura launched proBEAT™ in the U.S. in December 2020, as part of a diabetes prevention and
reversal program branded BEATdiabetes.life, in the form of pilot studies. During the quarter ended December 31, 2020, Nemaura licensed
a clinically validated weight loss program for the management of diabetes from Healthimation, LLC, which was originally developed at the
Joslin Diabetes Center, an affiliate of Harvard Medical School. This program, together with proBEAT™, originally formed the basis
of the BEATdiabetes.life program that is currently being developed for commercialization in the U.S. The program is under further refinement
based on feedback gathered to date. Further KOL studies are planned to provide additional marketing support of the program in preparation
for a broader U.S.-wide roll-out. While still in the relatively early stages, we are pleased with initial results and feedback received
from these user-groups and one key outcome has been that the company is now focusing on providing proBEAT as a standalone plug-in for
existing diabetes management programs, therefore potentially allowing the company to accelerate commercialization as well as reduce the
cost burden associated with running its own program.
We believe there are
additional applications for sugarBEAT® and the underlying BEAT technology platform,
which may include:
| |
· |
a web-server accessible by physicians and diabetes professionals to track the condition remotely, thereby reducing healthcare costs and managing the condition more effectively; |
| |
· |
a complete virtual doctor that monitors a person's vital signs and transmits results via the web; |
| |
· |
other patches using the BEAT technology platform to measure alternative analytes, including lactate, uric acid, lithium and drugs. This would be a step-change in the monitoring of conditions, particularly in the hospital setting. Lactate monitoring is currently used to determine the relative fitness of professional athletes and we completed preliminary studies demonstrating the application of the BEAT technology for continuous lactate monitoring; and |
| |
· |
a continuous temperature monitoring system which could have various applications, including use for individuals to monitor their temperature in connection with diagnosis and monitoring of symptoms of viral infections, and in females to monitor the ovulation cycle. |
During this period of product development, the Company
has experienced recurring losses and negative cash flows from operations. For the fiscal years ended March 31, 2022 and 2021, we generated
$503,906 and $0 revenues from product sales and reported net losses of $13,886,805 and $6,258,596, respectively, and negative cash flow
from operating activities of $6,504,041 and $5,998,097, respectively. For the nine months ended December 31, 2022 and 2021, we generated
$77,044 and $183,628 revenues from product sales and reported net losses of $9,460,888 and $10,269,557, respectively, and negative cash
flow from operating activities of $6,090,181 and $4,353,494, respectively. As noted in our financial statements, as of December 31, 2022,
we had a cash balance of $7,340,840, working capital deficit of $2,309,666, an accumulated deficit of $47,192,364 and a deficiency in
total stockholders' equity of $9,858,709. There is substantial doubt regarding our ability to continue as a going concern as a result
of our historical recurring losses, negative cash flows from operations, existing debt of $20,070,259 due as of December 31, 2022, which
will require the restructure of the debt or obtain additional, new, funding. See “Risk Factors—We have a history of operating
losses, our management has concluded that factors raise substantial doubt about our ability to continue as a going concern and our auditor
has included an explanatory paragraph relating to our ability to continue as a going concern in its audit report for the fiscal years
ended March 31, 2022 and 2021.”
While the Company expects
to continue to incur losses from operations for the near-term and these losses could be significant as product development, regulatory
activities, clinical trials, and other commercial and product development related expenses are incurred, the Company reached a significant
milestone during the three month period ended December 31, 2021, as the Company commenced commercial delivery of its sugarBEAT®
device to its UK Licensee, allowing them to continue studies dedicated to developing user based feedback and evidence that could
potentially support reimbursement in the UK.
Management's strategic
assessment continues to include the following potential options:
| |
· |
obtaining further regulatory approval for the sugarBEAT® device in other global territories, including the U.S., and the Middle East; |
| |
· |
signing new/additional licensing and collaboration opportunities beyond our existing licensee partners; |
| |
· |
pursuing further capital raising opportunities to support and accelerate the commercialization strategy; and |
| |
· |
developing the sugarBEAT® device platform for commercialization for other applications. |
Our Business
Strategy
We intend to
lead in the discovery, development and commercialization of innovative and targeted diagnostic medical devices, and data-driven digital
platforms, that improve disease monitoring, management and overall patient care. Specifically, we intend to focus on the monitoring of
molecules that can be drawn out through the skin non-invasively using our technology platform. In addition to glucose, such molecules
may include lactic acid monitoring and the monitoring of prescription drugs and blood biomarkers that may help in the diagnosis, prevention,
or management of diseases, such as diabetes. We plan to take the following steps to implement our broad business strategy. Our key commercial
strategies post-approval will first be implemented in Europe and then in parts of the Middle East and Asia, and then the U.S., as follows:
| |
- |
Commercialize sugarBEAT® in the United Kingdom and Republic of Ireland. We intend to commercialize sugarBEAT® in the United Kingdom, and Republic of Ireland with MySugarWatch Limited (previously known as Dallas Burston Ethitronix Limited) (“MSW”), with whom we have an exclusive marketing rights agreement for these two countries. We have also signed a full commercial agreement with MySugarWatch (Europe) Limited (previously known as Dallas Burston Ethitronix (Europe) Limited) in May 2018 for all other European territories as part of an equal joint venture agreement. The joint venture intends to seek sub-license rights opportunities to one or more leading companies in the diabetes monitoring space, to leverage their network, infrastructure and resources. |
| |
- |
Establish licensing or joint venture agreements with other parties to market sugarBEAT® in other geographies. We are in detailed discussions and negotiations with several other parties worldwide for licensing or joint venture agreements for the sale of the sugarBEAT® device and have signed a commercial agreement with TP MENA for the Gulf Cooperation Council, excluding Qatar. |
| |
- |
Seek FDA PMA approval of sugarBEAT®. The PMA application is currently in review by the FDA. |
| |
- |
Expand the indications for which the sugarBEAT® device may be used. We believe that the sugarBEAT® device may offer significant benefits as compared to those found in the non-acute setting for the monitoring of other diseases. This includes monitoring of lactic acid for performance athletics, and the monitoring of drugs. We have completed initial proof of concept for lactate monitoring and now plan to explore the routes to commercialization.. |
| |
- |
Expand our product pipeline through our proprietary platform technologies, acquisitions and strategic licensing arrangements. We intend to leverage our proprietary platform technologies to grow our portfolio of product candidates for the diagnosis of diabetes and other diseases. This includes digital platforms driven by data gathered by our sensors within the medical and wellbeing markets, such as for metabolic health monitoring. In addition, we intend to license our product and acquire products and technologies that are consistent with our research and development and business focus and strategies. This may include drug delivery products for the improved management of diabetes, for example improved insulin injector systems, and/or combination drug products for diabetes related drugs. |
Product
Development
Management
has extensive experience in regulatory and clinical development of diagnostic medical devices. We intend to take advantage of this experience
in the field of diagnostic medical devices in an attempt to increase the probability of product approval. The overall regulatory
process for diagnostic medical devices for diabetes is currently similar to those governing other diagnostic devices. The timelines are
shorter than, for example, when new drugs or completely invasive diagnostic devices are trialed in clinics. We have successfully tested
and evaluated the device for its clinical output, in this case the accuracy and safety with which it can trend blood glucose levels,
based on which CE approval was granted by the Notified Body BSI. A PMA (pre-market approval) application was also submitted to the FDA
and is currently under review. We continue to seek collaborations with future licensees and marketing partners to achieve our commercial
growth milestones.
The table below provides our current estimate of our timeline:
Product Development and Commercialization
Timelines
| |
|
|
| Milestone |
Target Start Date |
Current Status |
| Completion of clinical studies in Type 1 and Type 2 diabetic subjects to define final device claims and for submission for CE Mark approval with final device claims. |
July 2017 |
Completed |
| CE Mark for body worn transmitter device |
August 2018 |
Completed |
| U.S. FDA PMA Submission |
June 2020 |
Submission Completed, FDA review ongoing |
| Commercial launch in the UK, followed by major territories in Europe |
July - September 2020 |
Staggered launch in progress |
| Commercial launch of proBEATÔ in the U.S. |
October - December 2020 |
Commenced December 2020 |
|
Scale up of commercial sensor / patch manufacturing.
Scale up means we have started looking at larger scales sufficient
for product launch in the UK and relates to the manufacturing process for sensors. |
December 2020 |
In progress |
| Scale up of device (transmitter) manufacturing |
December 2020 |
In progress |
| Commencement of product delivery to UK Licensee |
December 2021 |
On-going |
Market
Opportunity for the Company's Products
According
to the International Diabetes Federation Atlas 10th Edition 2021 (the "IDF"), there are approximately 537 million
adults living with diabetes, representing 10.5% of the world’s population in this age group. This number is predicted to rise to
643 million (11.3%) by 2030 and to 783 million (12.2%) by 2045. Additionally an estimated 240 million people are living with undiagnosed
diabetes worldwide, meaning almost one-in-two adults with diabetes are unaware they have the condition. The IDF identifies that almost
90% of people with undiagnosed diabetes live in low-and-middle income countries.
Statistics
published by the IDF evidence the fact that diabetes is a huge and growing problem, and that whilst the costs to society are already high,
they continue to escalate. In addition, the IDF also notes that Europe has the highest prevalence of children and adolescents with Type
1 diabetes, as well as the highest incidence annually. Europe is also reported as having the second highest average cost per person with
diabetes ($3,086), with only North America and the Caribbean being higher ($8,208).
Statistical
Data for Diabetes Globally
| |
|
|
|
| |
2021 |
2030 |
2045 |
| Total world population |
7.9 billion |
8.6 billion |
9.5 billion |
| Adult population (20-79 years) |
5.1 billion |
5.7 billion |
6.4 billion |
| |
Diabetes (20 – 79 years) |
| Prevalence (%) |
10.5% |
11.3% |
12.2% |
| Number of people with diabetes |
536.6 million |
642.7 million |
783.2 million |
| Total health expenditure due to diabetes (2021 $) |
$966 billion |
$1,028 billion |
$1,054 billion |
| |
Impaired Glucose Tolerance “IGT” (20 – 79 years) |
| Prevalence (%) |
10.6% |
11.0% |
11.4% |
| Number of people with IGT |
541.0 million |
622.7 million |
730.3 million |
| |
Type 1 diabetes (0 – 19 years) |
| Number of children / adolescents with Type 1 diabetes |
1.2 million |
- |
- |
| Number of newly diagnosed cases per year |
184,100 |
- |
- |
Type
1 diabetes, once known as juvenile diabetes or insulin-dependent diabetes, is a chronic condition in which the pancreas produces little
or no insulin, a hormone needed to allow sugar (glucose) to enter cells to produce energy. The far more common Type 2 diabetes occurs
when the body becomes resistant to the effects of insulin or doesn't make enough insulin.
Various
factors may contribute to Type 1 diabetes including genetics and exposure to certain viruses. Although Type 1 diabetes typically appears
during childhood or adolescence, it also can develop in adults.
Despite
active research, Type 1 diabetes has no cure, although it can be managed. With proper treatment, people who have Type 1 diabetes can expect
to live longer, healthier lives than they did in the past. Type 1 diabetes includes autoimmune Type 1 diabetes (Type 1a) which is characterized
by having positive autoantibodies, as well as idiopathic Type 1 diabetes (Type 1b) where autoantibodies are negative, and c-peptide is
low. Patients with Type 1 diabetes (insulin dependent) require long term treatment with exogenous insulin and these patients perform self-monitoring
of blood glucose (SMBG) to calculate the appropriate dose of insulin. SMBG is done by using blood samples obtained by finger sticks but
frequent SMBG does not detect all the significant deviations in blood glucose, specifically in patients who have rapidly fluctuating glucose
levels.
Type
2 diabetes, once known as adult-onset or non-insulin-dependent diabetes, is a chronic condition that affects the way your body metabolizes
sugar (glucose), your body's main source of fuel. With Type 2 diabetes, your body either resists the effects of insulin, a hormone that
regulates the movement of sugar into your cells or doesn't produce enough insulin to maintain a normal glucose level. Untreated, Type
2 diabetes can be life-threatening.
More
common in adults, Type 2 diabetes increasingly affects children as childhood obesity increases. Whilst there is currently no acknowledged
cure for Type 2 diabetes, there is increasing evidence to suggest that it can be effectively managed by eating well, exercising and maintaining
a healthy weight. If diet and exercise don't control the blood sugar, diabetes medications or insulin therapy may be required.
Each
year, millions of patients undergo diabetes testing in the European Union and in the U.S. The main reason for this testing is to detect
and evaluate diabetes in patients with symptoms of diabetes. These studies provide clinical benefit in the initial evaluation of patients
with suspected but unproven diabetes, and in those patients in whom a diagnosis of diabetes has been established and information on prognosis
or risk is required.
We
believe that our market opportunity is a direct function of the number of persons tested, diagnosed and treated for Type 2 diabetes. The
IDF indicates that the total world market opportunity for a continuous glucose monitoring device is in the billions of dollars and is
projected to grow annually as incidences of diabetes continue to grow.
We
do not believe it is possible to estimate the number of diabetes patients that undergo finger pricks or other types of invasive glucose
monitoring. However, we are unaware of any product currently on the market that may allow for non-invasive continuous glucose monitoring.
We believe the sugarBEAT® device may be readily adopted by the medical community for the assessment of a patient continuously.
We
believe our non-invasive sugarBEAT® device possesses many significant advantages and may represent an ideal device for the detection
of discordances in an individual's blood sugar levels. We believe the CE approved sugarBEAT® device may represent a best-in-class
non-invasive continuous glucose monitoring device to reach those afflicted with diabetes. While we cannot estimate the market share that
our sugarBEAT® device may capture, we believe that the sugarBEAT® device will capture a significant share of the non-invasive
continuous glucose monitoring market, in-particular the market that has been established by the Abbott Freestyle Libre device for glucose
trending, as well as be adopted by non-insulin dependent diabetics who have not historically used continuous glucose monitoring devices
due to their invasiveness.
Commercialization
Plan
Throughout
the fiscal year ended March 31, 2022, we continued to work with our UK Licensee, MSW, to provide support in the development of their go-to-market
strategy which incorporates the utilization of our sugarBEAT® device into their own branded product offering. While COVID-19 did result
in some short delays to MSW’s user assessment program, the overall feedback was positive, albeit the anticipated timetable for purchase
orders to be placed by MSW was extended out, with the first order for 5,000 sugarBEAT® transmitters and 200,000 sugarBEAT® sensors
not being placed until April 2021. Our focus continues to be to support and optimize MSW’s launch program and activities designed
to gather evidence that could potentially support reimbursement, in line with which, we took the following actions during the fiscal year
ended March 31, 2022:
| |
· |
Entered into a new leased facility to provide additional capacity for commercial assembly to commence. |
| |
· |
Increased headcount of production operatives to facilitate product manufacture. |
| |
· |
Placed forward orders for raw materials to support scale-up and secure inventory of those items that are currently in short supply globally i.e. semi-conductors etc. |
| |
· |
Appointed Benchmark Electronics Inc.as our CMO partner to facilitate future volume scale up of transmitter production via its FDA approved facility in Thailand, in relation to which technology transfer is in-progress. |
| |
· |
Signed a new global agreement for the provision of our sugarBEAT® device with MySugarWatch DuoPack Limited (“MSW-DP”). Under the terms of the agreement, our CGM and sensors will be provided as Duo-Packs with prescription only medicines that are widely prescribed for people with Type 2 diabetes. The first of these medicines lost its patent protection in the fourth calendar quarter of 2022 in the UK, and the initial Duo-Pack presentation is expected to be launched in 2023 by the licensee. |
| |
· |
Commenced phased delivery of transmitters against the purchase order received from MSW in December 2021. |
We also advanced our plans to develop our
go-to-market capabilities in the U.S., which included:
| |
· |
In July 2020, we submitted a PMA application to the FDA for the sugarBEAT® device for glucose profiling as an adjunct to a finger-stick measurement. We, along with other applicants, were then informed by the FDA that the approval process was subject to delays as a result of the CDRH being actively engaged in responding to the pandemic caused by COVID-19 which resulted in staff being reallocated to other approval requests associated with COVID-19. In April 2021, the FDA confirmed that it would recommence its review of the PMA application. |
| |
· |
In December 2021, the FDA’s Bio-monitoring research division conducted an audit of the clinical program submitted in support of the PMA application. A single 483 observation was raised, and the Company submitted a full response in January 2022. |
| |
· |
The FDA subsequently conducted a pre-market inspection for during the second calendar quarter of 2022, covering the FDA’s Quality System / Current Good Manufacturing Practice regulations for Medical Devices (21 CFR Part 820). Once again a single 483 observation was made, and this was responded to within the mandated time frame. The company continues its dialogue with the FDA with respect to the PMA submission and plans to provide further material updates as they arise in due course. |
In
addition to this, we continue to explore commercialization opportunities in other key geographic markets, which includes engaging with
the German regulatory authority (GBA) to establish how best to proceed with achieving reimbursement for sugarBEAT® in Germany, as
well as continuing to engage in dialogue with additional potential licensees / distributors in other geographical territories.
Competitive
Landscape
To
the best of our knowledge, there are currently no other competing devices on the market that offer continuous glucose monitoring and profiling,
non-invasively, with a single day sensor wear. We believe this positions us uniquely in a market where we can target persons with diabetes
as well as those that are pre-diabetic. Additionally, we believe that this can also be used to improve outcomes in weight management and
wellbeing markets. There are companies, such as Dexcom and Abbott, that currently offer Continuous Glucose Monitoring (CGM) sensors with
10 and 14 continuous day wear, respectively. These companies could be deemed future competitors were they to:
| |
– |
develop and market products that are less expensive or more effective than our current and/or future products; |
| |
– |
operate larger research and development programs or have substantially greater financial resources than we do; |
| |
– |
initiate or withstand substantial price competition more successfully than we can; |
| |
– |
have greater success in recruiting skilled technical and scientific workers from the limited pool of available talent; |
| |
– |
more effectively negotiate third-party licenses and strategic relationships; and |
| |
– |
take advantage of acquisition or other opportunities more readily than we can. |
We
may compete for market share against these companies and potential newcomers in this general field. These potential competitors, either
alone or together with their partners, may develop new products that will compete with ours, and these competitors may, and in certain
cases do, operate larger research and development programs, or have substantially greater financial resources than we do.
As
noted, while it is difficult to analyze our major competitors since currently there are no non-invasive diagnostic medical devices to
continuously monitor blood glucose levels, we anticipate that specific companies may compete with us in the future.
Regulatory
Requirements
Our
device has undergone the applicable electrical safety testing and biocompatibility has been demonstrated against the relevant European
Directives, Regulations and Standards. If and when new materials are introduced, they will undergo a biocompatibility risk assessment,
and further testing where necessary. Batches of the device and patches were manufactured for human clinical studies that took place between
November 2014 and December 2015. This was a functional watch device with a wire connection to a skin adhered sensor and electrode. Subsequent
to studies conducted in India the device received a CE mark approval in February 2016. The device has since been upgraded to reduce it
in size, include an enhanced sensor system and allow wireless communication from a body worn transmitter. This miniaturized wireless device
achieved CE approval in May 2019, and a PMA was submitted to the U.S. FDA in July 2020 and is currently in review. An application for
CE mark approval requires the Company to have an ISO13485 Quality Management System, covering the design, development and manufacture
of a medical device. Nemaura Medical does not have this accreditation, and instead under the terms of a service contract dated April 4,
2018, with Nemaura Pharma Limited (“Pharma”), Nemaura Medical has outsourced the CE approval registration process to Pharma.
Pharma, a related company, is controlled by our Chief Executive Officer, President, Chairman of the Board and majority shareholder, Dr
D.F.H. Chowdhury. Under the terms of the service contract Pharma has undertaken all required activities to register the product for CE
approval under a fee for service arrangement, while Nemaura Medical will retain full title and beneficial ownership of the CE mark, and
all related intellectual property without any further payments or royalties becoming due other than the fee for service.
Intellectual Property
We believe that clear and extensive intellectual property
relating to our technologies is central to long-term success and we intend to invest accordingly. This applies to both domestic and international
patent coverage, and trade secrets, and trademarks.
The sugarBEAT® technology is protected by filed
patents and substantial trade secrets covering methods and apparatus for measuring glucose extracted from human skin in a non-invasive
manner, devices for extracting glucose from the skin is a stable manner, and the formulation and process for preparation of the enzyme
solution used in the sensor.
On May 8, 2014, NDM Technologies Limited, a related
company, assigned the UK patent application 1208950.4 and International (PCT) patent application PCT/GB2013/051322 entitled "Cumulative
Measurement of an Analyte" to Dermal Diagnostics Limited (“DDL”) for a nominal consideration.
Additional patents are intended to be filed in the
future relating to the device and sensor, providing new intellectual property protection. Some of the recently filed patents and future
patents may supersede previous intellectual property.
The company retains substantial trade secrets relating
to aspects of the sensor manufacture process and the sensor formulation, which has taken several years to develop, and we believe will
prove challenging to reverse engineer as it consists of formulation components in addition to processing methods in complex combinations
that are unique to the final functional sensor. Patents will not be filed on this aspect of the technology to avoid any public dissemination
of the know-how.
These patents and know-how cover aspects of the technology
platform. Furthermore, the trademarks BEAT and sugarBEAT® have been registered in multiple key global territories. Accordingly, all
intellectual property essential to the sugarBEAT® product is owned by us, and not subject to royalty payments. We intend to take the
lead in the preservation and/or prosecution of these patents and patent applications going forward as required. We intend to file additional
patents as the development progresses, where deemed to be of value to protecting the technology platform and future modifications and
improvements. New patents and patent filings could supersede existing patents as the technology is further developed and optimized. Where
patents cannot be secured, the intellectual property will be limited to know-how and trade secrets, and these will be diligently guarded.
Trade Secrets, Trademarks, and Patents Filed, Granted
and Pending
| IP: Patent (Core Claim), Know-how, Trademark |
|
Expiration Date |
|
Jurisdictions in which Granted / Issued |
|
Jurisdictions in which Pending |
|
Ongoing Royalty or Milestone Payments |
| |
|
|
|
|
|
|
|
|
| Patent: Cumulative Measurement of an Analyte (1) |
|
May 20, 2033 |
|
Australia, France, Germany, Italy, Poland, Spain, Netherlands, UK, China, Japan, USA, Canada, UAE |
|
Brazil, Qatar |
|
None. Internal development |
| Skin Prep Patch (2) |
|
December 2, 2039 |
|
N/A |
|
UK, Europe, USA |
|
None. Internal development |
| Know-how: Sensor Formulation and manufacture processes |
|
N/A |
|
Trade Secret |
|
N/A |
|
None. Internal development |
|
Trademark:
BEAT |
|
Renewal due in 2026 |
|
UK, Canada, China, EU, India, Japan, Norway, Russia, Singapore |
|
Malaysia, Brazil, Mexico, Switzerland, Turkey |
|
None. Internal development |
| Trademark: sugarBEAT |
|
Renewal due in 2025 |
|
UK, Canada, Australia, Switzerland, China, Egypt, EU, Israel, India, Iran, Japan, North Korea, Morocco, Mexico, Norway, New Zealand, Russia, Singapore, Tunisia, Turkey, USA |
|
N/A |
|
None. Internal development |
(1) This patent provides a formula for calculating
the amount of glucose extracted over a defined period of time by deducting the difference between two readings to allow rapid sensing
without needing to deplete the analyte being measured.
(2) This patent describes a device and method for
preparing the skin for the measurement of glucose on the skin surface.
Clinical
Trials
Our
clinical testing is conducted by contract clinical research organizations in various centers around the world to cover a wide demographic
– including Asia and Europe – and is managed by our in-house management team.
We
had 2 pre-submission meetings with the FDA in June 2016, to define the clinical roadmap. As a result, a detailed clinical plan was developed
and approved internally and a clinical site in Europe was selected and audited and approved for commencement of clinical studies using
the body worn transmitter device version of the sugarBEAT®. The study was completed, and a PMA application submitted to the FDA in
July 2020.
The
data from these studies was also submitted as part of the CE approval in Europe was received in May 2019.
Research
and development
We
spent $1,556,988 and $1,554,603 during the fiscal years ended March 31, 2022 and 2021, respectively, on research and development; management
currently anticipated that spend in this area will remain reasonably consistent in the coming fiscal year.
Manufacturing
The
manufacture and sale of CE certified medical devices are controlled and governed by guidelines stipulated in the International Organization
for Standardization (ISO), more specifically ISO13485; sugarBEAT® will be manufactured and marketed according to ISO13485 quality
standards.
In
support of commercial sales of sugarBEAT® in the UK and EU we have worked with our manufacturing partner Nemaura Pharma, to scale-up
manufacturing of the various sugarBEAT® components alongside facilities for final assembly and packaging. As part of this process,
we have expanded our manufacturing and assembly capabilities by occupying additional space within our existing headquarters site at Loughborough
University Science and Enterprise Park (LUSEP) in the UK.
We
have entered into the following types of agreements with various manufacturing partners:
| |
– |
Manufacturing agreements for the sensor manufacture |
| |
– |
Manufacturing agreements for the patch manufacture |
| |
– |
Manufacturing agreements for the CGM transmitter device and re-charging station manufacture |
Sales
and Marketing
An
Exclusive Marketing Rights agreement for the UK and Republic of Ireland was signed on March 31, 2014 with Dallas Burston Pharma, a Jersey
(Channel Island) based company (“DB Pharma”) (subsequently updated in 2018 and again in 2021 to include a change in the company
name to MySugarWatch Limited “MSW”), who has pharmaceutical product marketing operations in the UK and has demonstrated a
very successful model for the marketing of prescription medical products directly to general practitioners. We received a non-refundable
upfront payment of £1 million ($1.67 million at the then exchange rate) in return for providing MSW with the exclusive right to
sell the sugarBEAT® device in the UK and Republic of Ireland, both direct to consumer and through prescriptions by general practitioners.
The key terms of the Exclusive Marketing Rights Agreement were concluded in a Commercial Agreement signed in August 2015. This agreement
was updated and re-issued in October 2019 to cover new IP / improvements to the technology.
In
addition, a joint venture agreement was entered into with MySugar Watch (Europe) Limited (previously known as Dallas Burston Ethitronix
(Europe) Limited) in May 2018, whereby we will share equally the costs and net profits of the sales of our sugarBEAT® system in all
territories in Europe, with the exception of the United Kingdom, which is the subject of a separate agreement with MSW. This agreement
was updated and re-issued in October 2019 to cover new IP/ improvements to the technology. Commercial agreements were signed in 2018 with
TPMENA and Al-Danah Medical, for the Gulf Region (GCC) and Qatar respectively.
Regulatory
matters
Government
Regulation
Our
business is subject to extensive federal, state, local and foreign laws and regulations, including those relating to the protection of
the environment, and health and safety. Some of the pertinent laws have not been definitively interpreted by the regulatory authorities
or the courts, and their provisions are open to a variety of subjective interpretations. In addition, these laws and their interpretations
are subject to change, or new laws may be enacted.
Both
federal and state governmental agencies continue to subject the healthcare industry to intense regulatory scrutiny, including heightened
civil and criminal enforcement efforts. We believe that we have structured our business operations to comply with all applicable legal
requirements. However, it is possible that governmental entities or other third parties could interpret these laws differently and assert
otherwise. We discuss below the statutes and regulations that are most relevant to our business.
United
Kingdom and Wales and the European Union regulations
Government
authorities in the United Kingdom and Wales and the European Union as well as other foreign countries extensively regulate, among other
things, the research, development, testing, manufacture, labelling, promotion, advertising, distribution, sampling, marketing and import
and export of medical devices, including patches and other pharmaceutical products. Our body worn transmitter devices in the United Kingdom
and Wales will be subject to strict regulation and require regulatory approval prior to commercial distribution. The process of obtaining
governmental approvals and complying with ongoing regulatory requirements requires the expenditure of substantial time and financial resources.
In addition, statutes, rules, regulations and policies may change and new legislation or regulations may be issued that could delay such
approvals. If we fail to comply with applicable regulatory requirements at any time during the product development process, approval process,
or after approval, we may become subject to administrative or judicial sanctions. These sanctions could include the authority's refusal
to approve pending applications, withdrawals of approvals, clinical holds, warning letters, product recalls, product seizures, total or
partial suspension of our operations, injunctions, fines, civil penalties or criminal prosecution. Any agency enforcement action could
have a material adverse effect on us.
The
European Commission on Public Health (the "ECPH") provides the regulation for the development and commercialization of new medical
diagnostic devices. Any medical device placed on the European market must comply with the relevant legislation, notably with Directive
93/42/EEC for medical devices, with the active implantable devices Directive (90/385/EEC) or with the in vitro devices
Directive (98/79/EC). From 26th May 2021, all newly approved medical devices must comply with the Medical Device Regulation
(2017/745). Before manufacture / import, it must be determined whether the device in question falls under any of these Directives. All
medical devices must fulfil the essential requirements set out in the above-mentioned directives. Where available, relevant standards
may be used to demonstrate compliance with the essential requirements defined in the devices Directives.
Manufacturers
also need to determine the appropriate conformity assessment route. For devices falling under Directive 93/42/EEC / Regulation 2017/745,
other than custom-made devices and devices intended for clinical investigation, the conformity assessment route depends on the class of
the device, to be determined in accordance with certain rules set forth in the directives / regulations. Once the applicable class or
list has been determined, manufacturers need to follow the appropriate conformity assessment procedure. Subject to the type of the device,
this may require manufacturers to have their quality systems and technical documentation reviewed by a Notified Body before they can place
their products on the market. A Notified Body is a third-party body that can carry out a conformity assessment recognized by the European
Union. The Notified Body will need to assure itself that relevant requirements have been met before issuing relevant certification. Manufacturers
can then place the CE marking on their products to demonstrate compliance with the requirements.
The
CE approval is the process of achieving a mandatory conformity marking for the sugarBEAT® device to allow it to be legally sold in
the European Union. It is a manufacturers' declaration that the product meets the requirements of the applicable European laws. The process
for the sugarBEAT® device CE submission and approval involved the following:
1. The device
is classified depending on certain categories described by the European Directive with Class I products being low risk (e.g., band aid
plasters), with Class III devices being the highest risk. The classes are Class I, IIa, IIb and III. Risk is based upon the potential
harm to the patient should a problem arise with a product or its use. The sugarBEAT® device is classified as a IIb device.
2. A 'technical
file' containing all of the information required to demonstrate that the product meets the essential requirements of the European directive
will be prepared. This includes information relating to performance and safety of the device such as product specifications, labelling,
instructions for use, risk analysis and specific test information/clinical evidence relating to the product that support the claims being
made for the product.
3. Clinical evidence
included in the technical file is expected to demonstrate that the device is safe and meets defined performance requirements. This clinical
evidence can be in the form of literature data where substantial published data exists that utilizes the same technique for glucose extraction
and measurement (albeit in a different device format), or data from actual clinical studies performed using the sugarBEAT® device.
The first CE mark submission was based on literature evaluation of 3rd party published clinical data available in the
public domain. The final CE mark submission has claims based on the clinical performance of the device, based on clinical studies described
earlier herein. The clinical data showed that the sugarBEAT® device can trend blood glucose levels in a human subject by taking measurements
every 5 minutes. The clinical trial data demonstrates the sugarBEAT® device blood glucose trend can be used to supplement normal finger
prick measurements.
4. The technical
file has been assessed by an independent inspector (the Notified Body), regulated by the competent authority, (Medicines and Healthcare
products Regulatory Agency, MHRA in the United Kingdom). The Notified Body (an organization in the European Union that has been accredited
by a member state to determine whether a medical device complies with the European medical device directives), will then notify The European
Commission on Public Health (the "ECPH") of the approval and a certificate will be issued to the Company by the notified body
and we will then be able to apply the CE mark to the device, and legally offer the product for sale in the European Economic Area (EEA).
The CE mark has been issued as of May 2019 and the company is now able to offer the device for commercial sale in the EU.
5. The review
of the technical file commenced in August 2018, and the final review and sign off was received in May 2019. Since the CE mark was approved,
we have undergone routine inspections of our ISO 13485 Quality Management System in order to maintain our CE mark accreditation. An addendum
was also submitted to the notified body and approval obtained, to include within the approved CE marked device, the iOS version of the
smart device app that the transmitter connects to.
U.S.
Food and Drug Administration regulation of medical devices
The
US Food, Drug, and Cosmetic Act (the “FDCA”) and FDA regulations establish a comprehensive system for the regulation of medical
devices intended for human use. sugarBEAT® is a medical device that is subject to these, as well as other federal, state, local and
foreign, laws and regulations. The FDA is responsible for enforcing the laws and regulations governing medical devices in the United States.
The
FDA classifies medical devices into one of three classes (Class I, Class II, or Class III) depending on their level of risk and the types
of controls that are necessary to ensure device safety and effectiveness. The class assignment is a factor in determining the type of
premarketing submission or application, if any, that will be required before marketing in the United States. SugarBEAT® falls under
Class III.
| |
– |
Class I devices present a low risk and are not life-sustaining or life-supporting. The majority of Class I devices are subject only to "general controls" (e.g., prohibition against adulteration and misbranding, registration and listing, good manufacturing practices, labelling, and adverse event reporting. General controls are baseline requirements that apply to all classes of medical devices.) |
| |
– |
Class II devices present a moderate risk and are devices for which general controls alone are not sufficient to provide a reasonable assurance of safety and effectiveness. Devices in Class II are subject to both general controls and "special controls" (e.g., special labelling, compliance with performance standards, and post market surveillance. Unless exempted, Class II devices typically require FDA clearance before marketing, through the premarket notification (510(k)) process.) |
| |
– |
Class III devices present the highest risk. These devices generally are life-sustaining, life-supporting, or for a use that is of substantial importance in preventing impairment of human health or present a potential unreasonable risk of illness or injury. Class III devices are devices for which general controls, by themselves, are insufficient and for which there is insufficient information to determine that application of special controls would provide a reasonable assurance of safety and effectiveness. Class III devices are subject to general controls and typically require FDA approval of a PMA application before marketing. |
Unless
it is exempt from premarket review requirements, a medical device must receive marketing authorization from the FDA prior to being commercially
marketed, distributed or sold in the United States. The most common pathways for obtaining marketing authorization are 510(k) clearance
and PMA. After preliminary discussions with the FDA in June 2016 as part of a pre-submission meeting it was determined that the pathway
for sugarBEAT® would be a PMA approval.
Premarket
approval pathway
The
PMA approval process requires an independent demonstration of the safety and effectiveness of a device. PMA is the most stringent type
of device marketing application required by the FDA. PMA approval is based on a determination by the FDA that the PMA contains sufficient
valid scientific evidence to ensure that the device is safe and effective for its intended use(s). A PMA application generally includes
extensive information about the device including the results of clinical testing conducted on the device and a detailed description of
the manufacturing process.
After
a PMA application is accepted for review, the FDA begins an in-depth review of the submitted information. FDA regulations provide 180
days to review the PMA and make a determination; however, in reality, the review time is normally longer (e.g., 1-3 years). During this
review period, the FDA may request additional information or clarification of information already provided. Also, during the review period,
an advisory panel of experts from outside the FDA may be convened to review and evaluate the data supporting the application and provide
recommendations to the FDA as to whether the data provides a reasonable assurance that the device is safe and effective for its intended
use. In addition, the FDA generally will conduct a preapproval inspection of the manufacturing facility to ensure compliance with Quality
System Regulation, which imposes comprehensive development, testing, control, documentation and other quality assurance requirements for
the design and manufacturing of a medical device.
Based
on its review, the FDA may (i) issue an order approving the PMA, (ii) issue a letter stating the PMA is "approvable" (e.g.,
minor additional information is needed), (iii) issue a letter stating the PMA is "not approvable," or (iv) issue an order denying
PMA. A company may not market a device subject to PMA review until the FDA issues an order approving the PMA. As part of a PMA approval,
the FDA may impose post-approval conditions intended to ensure the continued safety and effectiveness of the device including, among other
things, restrictions on labelling, promotion, sale and distribution, and requiring the collection of additional clinical data. Failure
to comply with the conditions of approval can result in materially adverse enforcement action, including withdrawal of the approval.
Most
modifications to a PMA approved device, including changes to the design, labelling, or manufacturing process, require prior approval before
being implemented. Prior approval is obtained through submission of a PMA supplement. The type of information required to support a PMA
supplement and the FDA's time for review of a PMA supplement vary depending on the nature of the modification.
In
February 2020 Nemaura announced that following discussions with the FDA, Nemaura established that Nemaura may sell its CGM product with
a digital service offering in the U.S. without FDA approval as a non-medical wellbeing application. Nemaura further announced that it
intended to launch this product under the brand proBEATÔ in the U.S.. The product enables
users to wear the CGM device from which data will be sent to Nemaura’s servers in the cloud, from where data will be processed to
provide users with educational material and insights into factors that can affect their sugar levels and tips for healthy lifestyle and
diet, with a view to helping pre-diabetics and diabetics alike live healthier lives. A limited product launch commenced by way of pilot
studies in the U.S. in December 2020 to enabled potential customers to register their interest utilizing proBEATÔ in
conjunction with a digital program for weight loss targeted at persons with diabetes, under the brand BEATdiabetes.life.
Clinical
trials
Clinical
trials of medical devices in the U.S. are governed by the FDA's Investigational Device Exemption ("IDE") regulation. This regulation
places significant responsibility on the sponsor of the clinical study including, but not limited to, choosing qualified investigators,
monitoring the trial, submitting required reports, maintaining required records, and assuring investigators obtain informed consent, comply
with the study protocol, control the disposition of the investigational device, submit required reports, etc.
Clinical
trials of significant risk devices (e.g., implants, devices used in supporting or sustaining human life, devices of substantial importance
in diagnosing, curing, mitigating or treating disease or otherwise preventing impairment of human health) require FDA and Institutional
Review Board ("IRB") approval prior to starting the trial. FDA approval is obtained through submission of an IDE application.
Clinical trials of non-significant risk ("NSR") devices (i.e., devices that do not meet the regulatory definition of a significant
risk device) only require IRB approval before starting. The clinical trial sponsor is responsible for making the initial determination
of whether a clinical study is significant risk or NSR; however, a reviewing IRB and/or FDA may review this decision and disagree with
the determination.
An
IDE application must be supported by appropriate data, such as performance data, animal and laboratory testing results, showing that it
is safe to evaluate the device in humans and that the clinical study protocol is scientifically sound. There is no assurance that submission
of an IDE will result in the ability to commence clinical trials. Additionally, after a trial begins, the FDA may place it on hold or
terminate it if, among other reasons, it concludes that the clinical subjects are exposed to an unacceptable health risk.
As
noted above, the FDA may require a company to collect clinical data on a device in the post-market setting.
The
collection of such data may be required as a condition of PMA approval. The FDA also has the authority to order, via a letter, a post-market
surveillance study for certain devices at any time after they have been cleared or approved.
Pervasive
and continuing FDA regulation
After
a device is placed on the market, regardless of its classification or premarket pathway, numerous additional FDA requirements generally
apply. These include, but are not limited to:
| |
– |
Establishment registration and device listing requirements; |
| |
– |
Quality System Regulation ("QSR"), which governs the methods used in, and the facilities and controls used for, the design, manufacture, packaging, labelling, storage, installation, and servicing of finished devices; |
| |
– |
Labelling requirements, which mandate the inclusion of certain content in device labels and labelling, and generally require the label and package of medical devices to include a unique device identifier ("UDI"), and which also prohibit the promotion of products for uncleared or unapproved, i.e., "off-label," uses; |
| |
– |
Medical Device Reporting ("MDR") regulation, which requires that manufacturers and importers report to the FDA if their device may have caused or contributed to a death or serious injury or malfunctioned in a way that would likely cause or contribute to a death or serious injury if it were to recur; and |
| |
– |
Reports of Corrections and Removals regulation, which requires that manufacturers and importers report to the FDA recalls (i.e., corrections or removals) if undertaken to reduce a risk to health posed by the device or to remedy a violation of the Federal Food, Drug and Cosmetic Act that may present a risk to health; manufacturers and importers must keep records of recalls that they determine to be non-reportable. |
The
FDA enforces these requirements by inspection and market surveillance. Failure to comply with applicable regulatory requirements can result
in enforcement action by the FDA, which may include, but is not limited to, the following sanctions:
| |
– |
Untitled letters or warning letters; |
| |
– |
Fines, injunctions and civil penalties; |
| |
– |
Recall or seizure of our products; |
| |
– |
Operating restrictions, partial suspension or total shutdown of production; |
| |
– |
Refusing a request for 510(k) clearance or premarket approval of new products; |
| |
– |
Withdrawing 510(k) clearance or premarket approvals that are already granted; and |
We
would be subject to unannounced device inspections by the FDA, as well as other regulatory agencies overseeing the implementation of and
compliance with applicable state public health regulations. These inspections may include our suppliers' facilities.
Other
Regulation in the United Kingdom and Wales and the EU
Healthcare
Reimbursement
Government
and private sector initiatives to limit the growth of healthcare costs, including price regulation, competitive pricing, coverage and
payment policies, and managed-care arrangements, are continuing in many countries where we do business, including the United Kingdom and
Wales. These changes are causing the marketplace to put increased emphasis on the delivery of more cost-effective medical products. Government
programs, private healthcare insurance and managed-care plans have attempted to control costs by limiting the amount of reimbursement
they will pay for particular procedures or treatments. This has created an increasing level of price sensitivity among customers for products.
Some third-party payers must also approve coverage for new or innovative devices or therapies before they will reimburse healthcare providers
who use the medical devices or therapies. Even though a new medical product may have been cleared for commercial distribution, we may
find limited demand for the product until reimbursement approval has been obtained from governmental and private third-party payers.
Environmental
Regulation
We
are also subject to various environmental laws and regulations both within and outside the United Kingdom and Wales. Like many other medical
device companies, our operations involve the use of substances, including hazardous wastes, which are regulated under environmental laws,
primarily manufacturing and sterilization processes. We do not expect that compliance with environmental protection laws will have a material
impact on our consolidated results of operations, financial position or cash flow. These laws and regulations are all subject to change,
however, and we cannot predict what impact, if any, such changes might have on our business, financial condition or results of operations.
Foreign
Regulation
Whether
or not we obtain regulatory approval for a product, we must obtain approval from the comparable regulatory authorities of foreign countries
before we can commence clinical trials or marketing of the product in those countries. The approval process varies from country to country,
and the time may be longer or shorter than that required for EC approval. The requirements governing the conduct of clinical trials, product
licensing, pricing and reimbursement also vary greatly from country to country.
In
addition, regulatory approval of prices is required in most countries other than the United States. We face the risk that the prices which
result from the regulatory approval process would be insufficient to generate an acceptable return to us or our collaborators.
EU
General Data Protection Regulation
The
EU General Data Protection Regulation (the “GDPR”) came into force in all EU Member States from May 25, 2018 and replaced
previous EU data privacy laws. Although a number of basic existing principles will remain the same, the GDPR introduces new obligations
on data controllers and rights for data subjects, including, among others:
| |
– |
accountability and transparency requirements, which will require data controllers to demonstrate and record compliance with the GDPR and to provide more detailed information to data subjects regarding processing; |
| |
– |
enhanced data consent requirements, which includes “explicit” consent in relation to the processing of sensitive data; |
| |
– |
obligations to consider data privacy as any new products or services are developed and limit the amount of information collected, processed, stored and its accessibility; |
| |
– |
constraints on using data to profile data subjects; |
| |
– |
providing data subjects with personal data in a useable format on request and erasing personal data in certain circumstances; and |
| |
– |
reporting of breaches without undue delay (72 hours where feasible). |
The
GDPR also introduced new fines and penalties for a breach of requirements, including fines for serious breaches of up to the higher of
4% of annual worldwide revenue or €20m and fines of up to the higher of 2% of annual worldwide revenue or €10m (whichever is
highest) for other specified infringements. The GDPR identifies a list of points to consider when imposing fines (including the nature,
gravity and duration of the infringement).
The
Company has assessed the implications of the GDPR on all personal data it holds and has implemented measures to ensure that personal data
shall be:
| |
- |
Processed lawfully, fairly and in a transparent manner in relation to the data subject. |
| |
- |
Collected for a specified, explicit and legitimate purpose and not further processed in a manner that is incompatible with those purposes. |
| |
- |
Adequate, relevant and limited to what is necessary in relation to the purposes for which they are processed. |
| |
- |
Kept in a form which permits identification of data subjects for no longer than is necessary for the purposes for which the personal data is processed. |
| |
- |
Processed in a manner that ensures appropriate security of the personal data, including protection against unauthorised or unlawful processing and against accidental loss, destruction or damage, using appropriate technical or organisational measures. |
| |
- |
Maintained accurately and up to date and that every reasonable step is taken to ensure that personal data that is inaccurate, having regard to the purposes for which they are processed, are erased or rectified without delay. |
At
the current stage of the Company’s development and, with being pre-revenue at this stage, the scope of data held, and consequently
the impact of GDPR, is limited. Increased application of GDPR will be assessed and implemented prior to further Company developments that
warrant additional GDPR measures. As the Company progresses with product commercialization, the extent to which GDPR will affect the Company
will increase, which will require additional changes to the Company’s procedures and policies which could adversely impact operational
and compliance costs. Further, there is a risk that the measures will not be implemented correctly or that individuals within the business
will not be fully compliant with the new procedures. If there are breaches of these measures, the Company could face significant administrative
and monetary sanctions as well as reputational damage which may have a material adverse effect on its operations, financial condition,
and prospects.
Human
Capital Management
We
believe that a diverse workforce is important to our success. We will continue to focus on the hiring, retention and advancement of women
and underrepresented populations, and to cultivate an inclusive and diverse corporate culture. In the future, we intend to continue to
evaluate our use of human capital measures or objectives in managing our business such as the factors we employ or seek to employ in the
development, attraction and retention of personnel and maintenance of diversity in our workforce.
The
success of our business is fundamentally connected to the well-being of our people. Accordingly, we are committed to the health, safety,
and wellness of our employees. We provide our employees with access to a variety of flexible and convenient health and wellness programs,
including benefits that provide protection and security so they can have peace of mind concerning events that may require time away from
work or that impact their financial well-being; that support their physical and mental health by providing tools and resources to help
them improve or maintain their health status and encourage engagement in healthy behaviors; and that offer choice where possible so they
can customize their benefits to meet their needs and the needs of their families.
We
also provide robust compensation and benefits programs to help meet the needs of our employees. We believe that we maintain a satisfactory
working relationship with our employees and have not experienced any labor disputes. As of March 31, 2022, we had 37 personnel employed
on our payroll, which equates to approximately 32 full-time equivalents.
Organizational
Structure
We
are a holding corporation that owns 100% of a diagnostic medical device company specializing in discovering, developing, and commercializing
specialty medical devices. We were organized on December 24, 2013, under the laws of the State of Nevada. We own 100% of Dermal Diagnostic
(Holdings) Limited, an England and Wales corporation formed on December 11, 2013. Dermal Diagnostics (Holdings) Limited owns 100% of the
stock in Dermal Diagnostics Limited (“DDL”), an England and Wales corporation formed on January 20, 2009, and 100% of the
stock in Trial Clinic Limited (“TCL”), an England and Wales corporation formed on January 12, 2011.
The
following diagram illustrates Nemaura’s corporate structure as of February 28, 2023:

During
the fiscal year ended March 31, 2021, the Board of Directors assessed the adequacy of the group’s organizational structure and concluded
that an intermediary holding company, Region Green Limited, was no longer required as the entity had been effectively dormant since inception
and no longer represented a requirement to be maintained. It was therefore determined that Region Green Limited should be unwound, with
the assets held by Region Green Limited being transferred up to Nemaura Medical Inc. following which Region Green Limited would be dissolved.
The
transfer of assets took place on March 5, 2021 and Region Green Limited was formally dissolved as of April 23, 2021.
In
December 2013, we restructured the Company and re-domiciled as a domestic corporation in the United States. The corporate re-organization
was accomplished to preserve the tax advantages under the laws of the England and Wales tax laws for the benefit of the shareholders of
both Dermal Diagnostics Limited and Trial Clinic Limited.
DDL
is a diagnostic medical device company headquartered in Loughborough, Leicestershire, England. DDL was founded on January 20, 2009, to
engage in the discovery, development and commercialization of diagnostic medical devices. The Company’s initial focus has been on
the development of a novel CGM device.
Recent Developments
Termination
of Chief Financial Officer
Effective July 1,
2022, the Company terminated its Company’s Chief Financial Officer and has commenced a search for a U.S. based replacement. Until
a replacement has been selected, the Company’s President and Chief Executive Officer will act as principal financial and accounting
officer of the Company, and the Company’s finance team will continue to support the Company with respect to its accounting and financial
reporting compliance requirements.
Commercial Deliveries
December 2021 marked a significant
milestone in the Company’s evolutionary journey with the first deliveries of the sugarBEAT® non-invasive glucose monitor (“CGM”)
being made to the UK licensee, MySugarWatch Limited (“MSW”). MSW has developed a subscription-based diabetes coaching and
management service that will be provided alongside the CGM, primarily targeting those with type 2 diabetes and continues to undertake
studies to support full reimbursement for the sensors and the diabetes management program in the UK.
The deliveries reflect the
phased delivery schedule agreed upon with MSW in relation to MSW’s initial order that was placed earlier in 2021, as a result of
which the Company started to recognize revenues.
Furthermore, on September
24, 2021 the Company entered into a License, Supply and Distribution Agreement with MySugarWatch DuoPack Limited (“MSW-DP”),
a sister company of MSW, whereby MSW-DP will provide CGM sensors free of charge with certain medications that are widely prescribed to
persons with Type 2 diabetes. These medications came off patent in the fourth calendar quarter of 2022 in Europe and the UK, and due to
come off patent in 2023 in the U.S. The agreed sale price of sensors to MSW-DP under the terms of the agreement is $20 per box of five
sensors for the U.S. market, and in Europe and the UK 12.50 Euros in the first 12 months from product launch and 10 Euros thereafter per
box of four sensors. Nemaura’s anticipated cost of goods per sensor on large-scale production is $1 per sensor. As of January 2022,
there were over 2 million prescriptions written for these medications each month in the combined key EU and UK territories. The Company
believes this will provide an opportunity for rapid market penetration in the use of its CGM sensors, at a scale that can enable the targeted
lower cost of goods to be achieved and thereby support both revenue and margin growth into the future.
Management is now focused
on fulfilling the remainder of the UK licensees’ initial orders and supporting MSW’s UK launch, while also developing the
capabilities of the Company to develop and service new channels of business across other geographic markets via the use of our BEAT platform.
This includes expansion of the consumer metabolic health offering Miboko, launched in late 2021, to employers and insurers across the
U.S.
On January 26, 2023, we announced
receipt of a first purchase order for 5,000 proBEAT™ subscriptions from HealthFleet Inc. (“HealthFleet”), a leading
telehealth provider focusing on care, coaching, and health recommendations. The purchase order consists of 75,000 proBEAT glucose sensors
over an initial five-month period and is valued at $500,000 in revenue. HealthFleet has an option to increase volumes based on customer
response.
ATM Offering
In July 2021, the Company
entered into an At The Market Offering Agreement (the “ATM Agreement”) with H.C. Wainwright & Co., LLC (the “Agent”)
pursuant to which the Company may offer and sell from time to time to or through the Agent shares of the Company’s common stock.
On April 1, 2022, the Company and Agent entered into an amendment (the “Amendment”) to the ATM Agreement, pursuant to which
the parties agreed to expand the meaning of the defined term “Registration Statement” in the ATM Agreement to include, for
the period from April 1, 2022 and thereafter, a new shelf registration statement (File Number 333-263618) on Form S-3 (“New Registration
Statement”) that was filed on March 16, 2022 with the SEC and declared effective by the SEC on March 28, 2022. No other changes
to the ATM Agreement were made by the Amendment.
The offer and sale of shares
of Common Stock through the Agent will be made pursuant to the New Registration Statement, and a related prospectus supplement filed with
the SEC pursuant to which the Company is offering shares of its common stock having an aggregate offering price of up to $3,000,000.
Preliminary
agreement with EVERSANA
On September 27,
2022, the Company entered into a preliminary agreement with EVERSANA to collaborate on the launch strategy of the Company’s BEATdiabetes
program.
Amendment of
Uptown Capital Secured Promissory Note
On February 8, 2021, the
Company, Dermal Diagnostics Limited, a wholly owned subsidiary of the Company (“Dermal Diagnostics”), and Trial Clinic Limited,
a wholly owned subsidiary of the Company (“Trial Clinic” and collectively with the Company and Dermal Diagnostics) issued
to Uptown Capital, LLC (“Uptown”) a secured promissory note (the “Uptown Note”) in the original principal amount
of $24,015,000. The Uptown Note carried an original issue discount of $4,000,000. In addition, the Company agreed to pay $15,000 to Uptown
to cover Uptown’s legal fees, accounting costs, due diligence, monitoring and other transaction costs incurred in connection with
the purchase and sale of the Uptown Note, all of which amount was included in the initial principal balance of the Uptown Note. The purchase
price of the Uptown Note, therefore, was $20,000,000. The original maturity date of the Uptown Note was 24 months after the date the purchase
price for the Uptown Note was delivered.
On October 21, 2022, the
Company entered into an amendment to Secured Promissory Note, dated as of October 21, 2022, by and among the Company, Dermal Diagnostics,
Trial Clinic and Uptown. Pursuant to the terms of the amendment, the Company and Uptown agreed to extend the maturity date of the Uptown
Note to July 1, 2024. In consideration thereof, the Company agreed to pay to Uptown an extension fee in the amount of 5% of the outstanding
balance of the Uptown Note which results in $813,834 being added onto the liability due to Uptown.
The Company and Uptown previously
agreed to reduce the maximum monthly redemption amount from $2,000,000 to $500,000 from June 2022 to February 2023, which reduction remains
in force. Pursuant to the terms of the Amendment, the Company and Uptown agreed to reduce the maximum monthly redemption amount during
the period beginning March 2023 until the Uptown Note is paid in full from $2,000,000 to $1,000,000; provided, however, that upon the
occurrence of an event of default under the Uptown Note, the maximum monthly redemption amount will automatically be increased back to
$2,000,000.
Completion of $8.4
Million Registered Direct Offering and Concurrent Private Placement for Warrants
On January 31, 2023, the
Company completed the closing pursuant to a securities purchase agreement with two accredited institutional investors to purchase 4,796,206
shares of its common stock in a registered direct offering under the Form S-3 and warrants to purchase 4,796,206 shares of its common
stock at an exercise price of $2.00 per share in a concurrent private placement. The combined purchase price for one share of common stock
and one warrant is $1.75 for gross proceeds of $8,393,360.
Impact of COVID-19
A regional or global health
pandemic, including COVID-19, could severely affect our business, results of operations and financial condition. A regional or global
health pandemic, depending upon its duration and severity, could have a material adverse effect on our business. For example, the COVID-19
pandemic has had numerous effects on the global economy and governmental authorities around the world have implemented measures to reduce
the spread of COVID-19. These measures, including shutdowns and “shelter-in-place” orders suggested or mandated by governmental
authorities or otherwise elected by companies as a preventive measure, have adversely affected workforces, customers, consumer sentiment,
economies and financial markets, and, along with decreased consumer spending, have led to an economic downturn in many of our markets.
As a result of the COVID-19
pandemic, we evaluated and executed the steps available to us to ensure we were able to provide protection of our employees and instigated
remote working where possible combined with following all government advice and guidance regarding any engagement within the workplace
that could not be completed remotely. To date this transition to remote working has had little impact on our employee productivity and
has not caused any interruption to our business. Due to the uncertainty of COVID-19, we will continue to assess the situation, including
abiding by any government-imposed restrictions, as and where relevant.
We are unable to accurately predict the impact that
COVID-19 will have on our operations going forward due to uncertainties that will be dictated by the length of time that the pandemic
and related disruptions continue, the impact of governmental regulations that might be imposed in response to the pandemic and overall
changes in consumer behavior.
During this period, we, along with other companies,
were notified by the FDA in the U.S., that our PMA application for sugarBEAT® would be delayed due to the prioritization being given
to COVID-19 related applications and resource activity. While the PMA review resumed as of April 15, 2021, however due to the FDA’s
current workload in light of the pandemic, the review has taken longer than a review normally has taken before the pandemic.
While key suppliers have not been accessible throughout
the whole period of the outbreak, we have been able to be flexible in our priorities and respond favorably to the challenges faced during
the outbreak. We have seen an increase in the adoption of technologies for remote and patient self-monitoring, which therefore potentially
enhances the prospects for Nemaura Medical and its CGM product and planned digital healthcare offering.
At this point in time, there
remains significant uncertainty relating to the potential effect of COVID-19 on our business. As infections may continue to become more
widespread, we could experience a severe negative impact on our business, financial condition, and results of operations. To the extent
the COVID-19 pandemic adversely affects our business and financial results, it may also have the effect of heightening many of the other
risks described in this “Risk factors” section.
Properties
We
have registered corporate offices in the U.S. at 57 West 57th Street, Manhattan, NY 10019. We have offices and laboratories
located across two locations on the Loughborough University Science and Enterprise Park (LUSEP), Loughborough, Leicestershire, United
Kingdom. The aggregate monthly rent is approximately $34,000. All leases are currently operated on rolling 12-month terms. The terms of
the different leases provide break options allowing both landlord and tenant to terminate on provision of not less than one month’s
prior written notice.
Legal
Proceedings
We
do not know of any material, active, pending or threatened proceeding against us or our subsidiaries, nor are we, or any subsidiary, involved
as a plaintiff or defendant in any material proceeding or pending litigation.
MANAGEMENT
The
following persons are our executive officers and directors, and hold the positions set forth opposite their respective names as of the
date hereof.
| Name |
|
Age |
|
|
Position |
|
Date
of Appointment |
|
| Dewan Fazlul Hoque Chowdhury |
|
|
49 |
|
|
Chief Executive Officer, President, Interim Chief Financial Officer and Director |
|
|
December 24, 2013 |
|
| Arash Ghadar |
|
|
45 |
|
|
Chief Operating Officer |
|
|
January 5, 2022 |
|
| Bashir Timol |
|
|
47 |
|
|
Director,
Chief Business Officer |
|
|
December 24, 2013
April 9, 2018 |
|
| Thomas Moore |
|
|
58 |
|
|
Independent Director |
|
|
August 3, 2017 |
|
| Dr. Salim Natha |
|
|
55 |
|
|
Independent Director |
|
|
July 26, 2017 |
|
| Timothy Johnson |
|
|
38 |
|
|
Independent Director |
|
|
July 17, 2017 |
|
Our
directors hold office until the earlier of their death, resignation, or removal or until their successors have been qualified.
Dewan
Fazlul Hoque Chowdhury. Dr. D.F.H. Chowdhury has been our President, Chief Executive Officer and
a member of our board of directors since the incorporation of DDL on January 20, 2009 and has served as our Interim Chief Financial Officer
since July 1, 2022. He is in charge of research and development of our core technologies, product development, innovation and commercialization.
He also coordinates and oversees legal compliance; development of the company mission; policy and planning. Prior to establishing the
Company, Dr. D.F.H. Chowdhury was the founder and CEO of Microneedle Technologies and Nemaura Pharma Limited. Dr. D.F.H. Chowdhury has
been responsible for negotiating licensing deals for a transdermal patch to treat Alzheimer’s disease. Additionally, he is involved
in commercial negotiations and global strategy development.
Dr.
D.F.H. Chowdhury originally trained as a pharmaceutical scientist and has an MSc in Microsystems and Nanotechnology from Cranfield University,
and a Doctorate from the University of Oxford on nano-drug delivery. His experience in the Pharmaceutical Industry includes product development;
manufacturing; and technical and corporate management.
Arash
Ghadar. Dr Ghadar joined the business as Chief Operating Officer on January 5, 2022, prior to joining Nemaura, Dr. Ghadar spent
a decade as the Technical Director of Datalink Electronics (Datalink) in Loughborough, England where he managed the design team as an
autonomous entity within Datalink. He was responsible for management of the day-to-day operations, business planning, legal affairs, finance,
sales, and business development of the design team. In this role, he also oversaw numerous technical projects for healthcare and industrial
customers that included product development lifecycle, feasibility studies, design, development, prototyping, validation, certification,
quality management, and volume manufacturing.
Dr.
Ghadar is also currently a non-executive director at Medilink Midlands, the Midlands (England) Life Sciences industry association with
a vision to stimulate the growth of the Midlands life science sector. He has a BSc Degree and Masters in Electronics and Control Systems
Engineering, where he achieved a First Class degree and Distinction respectively, and he also has a Ph.D. in Biosensors from the University
of Warwick (U.K.).
Bashir
Timol. Mr. Timol has served as member of the board of Nemaura Medical since formation in
December 2013. He has co-founded, managed, and funded several biotech and life science companies, and led the investment consortium
that provided capital for the initial two funding rounds for Nemaura Medical. Mr. Timol obtained his Bachelor of Arts degree in Economics
from the University of Central Lancashire, UK.
Timothy
Johnson. Mr. Johnson was elected as a director in July 2017. He is currently serving in executive positions in several tax consultancy
and accountancy businesses in the UK. He is a practicing Chartered Tax Adviser and holds a first-class Master of Science in Mathematics
and Physics from the University of Manchester, UK. Mr Johnson’s work involves in depth review and analysis of financial statements
on a daily basis, and he has significant experience in matters relating to financial accounts, tax, financial management, financial regulatory
requirements and anti-money laundering requirements.
Thomas
Moore. Mr. Moore was elected as a director in August 2017. He is currently working as a director, tax consultant and co-owner
of a tax consultancy and pensions administration business (WestBridge), having built up three decades of experience in accounting and
consulting fields at leading accounting firms including Grant Thornton, KPMG and PricewaterhouseCoopers. Throughout the last five years,
Mr Moore has held his current role with WestBridge since May 2017 and before that was a Director with Grant Thornton UK PLC. He is a practicing
Chartered Tax Adviser and earned his first-class Bachelor of Arts in French and Russian from the University of Northumbria, UK. The qualifications
Mr Moore brings to the role include a wealth of experience in matters relating to accounts, financial management and financial regulatory
requirements including his current experience as an MLRO in two companies.
Dr.
Salim Natha. Dr. Natha was elected as a director in July 2017. He is currently practicing as an Eye Surgeon in the UK National
Health Service (NHS), and is the clinical lead for a retinopathy screening program for over 20,000 diabetics in the Ashton, Wigan and
Leigh region. He has published several articles in the medical literature and is a peer reviewer for the English National Diabetic Retinopathy
Screening Program. Dr. Natha graduated with honours from the University of Liverpool Medical School.
Family
Relationships
There
are no family relationships between any of our directors or executive officers.
Involvement
in Certain Legal Proceedings.
None.
Board
of Directors
All
directors hold office until the next Annual Meeting of shareholders and until their successors have been duly elected and qualified. Directors
are elected at the annual meetings to serve for one-year terms. Officers are elected by, and serve at the discretion of, the Board of
Directors. Our Board of Directors shall hold meetings on at least a quarterly basis.
The
Board of Directors complies with the NASDAQ Listing Rules with respect to corporate governance matters. Under the NASDAQ rules we are
required to maintain a board of directors comprised of at least 50% independent directors, and an audit committee of at least two members,
comprised solely of independent directors who also meet the requirements of Rule 10A-3 under the Securities Exchange Act of 1934.
Director
Independence
The
board of directors has reviewed the independence of our directors, applying the NASDAQ independence standards. Based on this review, the
board of directors determined that each of Thomas Moore, Dr. Salim Natha and Timothy Johnson are independent within the meaning of the
NASDAQ rules. In making this determination, our board of directors considered the relationships that each of these non-employee directors
has with us and all other facts and circumstances our board of directors deemed relevant in determining their independence. As required
under applicable NASDAQ rules, we anticipate that our independent directors will meet on a regular basis as often as necessary to fulfil
their responsibilities, including at least annually in executive session without the presence of non-independent directors and management.
Board
Committees
Our
board of directors has established standing committees in connection with the discharge of its responsibilities. These committees include
an Audit Committee, a Compensation Committee and a Nominating and Corporate Governance Committee. Our board of directors has adopted written
charters for each of these committees. Copies of the charters are available on our website. Our board of directors may establish other
committees as it deems necessary or appropriate from time to time.
Audit
Committee
Our
Audit Committee is comprised of our independent directors: Thomas Moore, Dr. Salim Natha and Timothy Johnson. Mr. Johnson qualifies as
the Audit Committee financial expert as defined in Item 407(d)(5) of Regulation S-K promulgated under the Securities Act.
According
to its charter, the Audit Committee consists of at least three members, each of whom shall be a non-employee director who has been determined
by the Board to meet the independence requirements of NASDAQ, and also Rule 10A-3(b)(1) of the SEC, subject to the exemptions provided
in Rule 10A-3(c). The Audit Committee Charter describes the primary functions of the Audit Committee, including the following:
| |
– |
Oversee the Company’s accounting and financial reporting processes; |
| |
– |
Oversee audits of the Company’s consolidated financial statements; |
| |
– |
Discuss policies with respect to risk assessment and risk management, and discuss the Company’s major financial risk exposures and the steps management has taken to monitor and control such exposures; |
|
– |
Review and discuss with management the Company’s audited consolidated financial statements and review with management and the Company’s independent registered public accounting firm the Company’s consolidated financial statements prior to the filing with the SEC of any report containing such consolidated financial statements. |
| |
– |
Recommend to the board that the Company’s audited consolidated financial statements be included in its annual report on Form 10-K for the last fiscal year; |
| |
– |
Meet separately, periodically, with management, with the Company’s internal auditors (or other personnel responsible for the internal audit function) and with the Company’s independent registered public accounting firm; |
| |
– |
Be directly responsible for the appointment, compensation, retention and oversight of the work of any independent registered public accounting firm engaged to prepare or issue an audit report for the Company; |
| |
– |
Take, or recommend that the board take, appropriate action to oversee and ensure the independence of the Company’s independent registered public accounting firm; and |
| |
– |
Review major changes to the Company’s auditing and accounting principles and practices as suggested by the Company’s independent registered public accounting firm, internal auditors or management. |
Compensation
Committee
The
Compensation Committee is responsible for, among other matters:
| |
– |
reviewing and approving, or recommending to the board of directors to approve the compensation of our CEO and other executive officers and directors reviewing key employee compensation goals, policies, plans and programs; |
| |
– |
administering incentive and equity-based compensation; |
| |
– |
reviewing and approving employment agreements and other similar arrangements between us and our executive officers; and |
| |
– |
appointing and overseeing any compensation consultants or advisors. |
Our
Compensation Committee consists of Thomas Moore, Dr. Salim Natha and Timothy Johnson. Dr. Salim Natha serves as chair of the Compensation
Committee.
Corporate
Governance and Nominating Committee
The
Corporate Governance and Nominating Committee is responsible for, among other matters:
| |
– |
selecting or recommending for selection candidates for directorships; |
| |
– |
evaluating the independence of directors and director nominees; |
| |
– |
reviewing and making recommendations regarding the structure and composition of our board and the board committees; |
| |
– |
developing and recommending to the board corporate governance principles and practices; |
| |
– |
reviewing and monitoring the Company’s Code of Ethics; and |
| |
– |
overseeing the evaluation of the Company’s management. |
Our
Corporate Governance and Nominating Committee consists of Thomas Moore, Dr. Salim Natha and Timothy Johnson. Mr. Johnson serves as chair
of the Corporate Governance and Nominating Committee.
Material
Changes to Procedures by which Security Holders May Recommend Board Nominees
We
do not currently have a procedure by which security holders may recommend nominees to the Board. Prior to the listing of our common stock
on NASDAQ, as a private company with a limited shareholder base, we did not believe that it was important to provide such a procedure.
However, as a publicly traded NASDAQ company with the requirement to hold annual shareholder meetings, we will consider implementing such
a policy in the future.
The
Board does not have a formal policy on Board candidate qualifications. The Board may consider those factors it deems appropriate in evaluating
director nominees made either by the Board or stockholders, including judgment, skill, strength of character, experience with businesses
and organizations comparable in size or scope to the Company, experience and skill relative to other Board members, and specialized knowledge
or experience. Depending upon the current needs of the Board, certain factors may be weighed more or less heavily. In considering candidates
for the Board, the directors evaluate the entirety of each candidate’s credentials and do not have any specific minimum qualifications
that must be met. The directors will consider candidates from any reasonable source, including current Board members, stockholders, professional
search firms or other persons. The directors will not evaluate candidates differently based on who has made the recommendation.
Board
Leadership Structure and Role in Risk Oversight
Dr.
Chowdhury holds the positions of chief executive officer, and chairman of the board of the Company. Prior to the appointment of Mr. Justin
Mclarney to the role of chief financial officer as of September 15, 2020, Dr. Chowdhury also acted as interim chief financial officer.
The board believes that Dr. Chowdhury’s services as both chief executive officer and chairman of the board is in the best interest
of the Company and its shareholders. Dr. Chowdhury possesses detailed and in-depth knowledge of the issues, opportunities and challenges
facing the Company in its business and is thus best positioned to develop agendas that ensure that the Board’s time and attention
are focused on the most critical matters relating to the business of the Company. His combined role enables decisive leadership, ensures
clear accountability, and enhances the Company’s ability to communicate its message and strategy clearly and consistently to the
Company’s shareholders, employees and customers.
The
board has not designated a lead director. Given the limited number of directors comprising the Board, the independent directors call and
plan their executive sessions collaboratively and, between meetings of the Board, communicate with management and one another directly.
Under these circumstances, the directors believe designating a lead director to take on responsibility for functions in which they all
currently participate might detract from rather than enhance performance of their responsibilities as directors.
Management
is responsible for assessing and managing risk, subject to oversight by the board of directors. The board oversees our risk management
policies and risk appetite, including operational risks and risks relating to our business strategy and transactions. Various committees
of the board assist the board in this oversight responsibility in their respective areas of expertise.
| |
– |
The Audit Committee assists the board with the oversight of our financial reporting, independent auditors, and internal controls. It is charged with identifying any flaws in business management and recommending remedies, detecting fraud risks and implementing anti-fraud measures. The audit committee further discusses Nemaura’s policies with respect to risk assessment and management with respect to financial reporting. |
| |
– |
The Compensation Committee oversees compensation, retention, succession and other human resources-related issues and risks. |
| |
– |
The Corporate Governance and Nominating Committee overviews risks relating to our governance policies and initiatives. |
Code
of Ethics
We
have adopted a Code of Ethics that applies to our principal executive officer, principal financial officer and other persons performing
similar functions. A copy of our Code of Ethics is available on our website. We intend to post amendments to, or waivers from a provision
of, our Code of Ethics that apply to our principal executive officer, principal financial officer or persons performing similar functions
on our website.
EXECUTIVE
COMPENSATION
2022
Summary Compensation Table
This
table provides disclosure, for fiscal years 2022 and 2021, of the compensation paid to our named executive officers.
Named Executive Officer
and Principal Position |
|
Year |
|
Salary |
|
Bonus |
|
Stock Awards |
|
Option Awards |
|
All Other Compensation |
|
Total |
| |
|
|
|
$ |
|
$ |
|
$ |
|
$ |
|
$ |
|
$ |
| Dr. D.F.H. Chowdhury, Chief Executive Officer, Interim Chief Financial Officer and President |
|
2022 |
|
|
|
109,416 |
|
|
|
— |
|
|
|
— |
|
|
|
26,706 |
(1) |
|
|
3,849 |
(4) |
|
|
139,971 |
|
| |
|
2021 |
|
|
|
104,840 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
3,368 |
(4) |
|
|
108.208 |
|
Justin Mclarney
former Chief Financial Officer (2) |
|
2022 |
|
|
|
169,055 |
|
|
|
— |
|
|
|
87,389 |
(3) |
|
|
— |
|
|
|
2,498 |
(4) |
|
|
258,942 |
|
| |
|
2021 |
|
|
|
67,107 |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
1,162 |
(4) |
|
|
68,269 |
|
| |
| (1) |
On January 28, 2022, in compensation for Dr. Chowdhury’s service as a director, the Company’s Board of Directors granted to Dr. Chowdhury an option to purchase 8,000 shares of common stock at an exercise price of $3.98 per share, the closing price of the Company’s common stock on the date of grant. The option was fully vested at grant and is exercisable for a period of five years from the date of grant. The fair value attributed to the options has been calculated using a Black-Scholes Option Pricing Model. |
| (2) |
Effective July 1, 2022, the Company terminated Justin Mclarney as its Chief Financial
Officer. Mr. Mclarney served as the Company’s Chief Financial Officer from September 15, 2020 until June 30, 2022.
Prior to Mr. Mclarney’s appointment, Dr. Chowdhury acted as Interim Chief Financial Officer. Following the termination of Mr. Mclarney as Chief Financial Officer, Dr.
Chowdhury has acted as Interim Chief Financial Officer. |
| (3) |
On January 31, 2022, Mr. Mclarney received 22,293 shares at the market price of $3.92. |
| (4) |
Represents (i) premiums paid for health insurance in fiscal year 2022 for $1,999 and $702 for Dr. Chowdhury and Mr. Mclarney, respectively, and in fiscal year 2021 for $1,889 and $202 for Dr. Chowdhury and Mr. Mclarney, respectively, and (ii) payments by the Company towards pension in fiscal year 2022 for $1,851 and $1,797 for Dr. Chowdhury and Mr. Mclarney, respectively, and in fiscal year 2021 for $1,480 and $961 for Dr. Chowdhury and Mr. Mclarney, respectively. |
Dr.
D.F.H. Chowdhury
We
entered into an employment agreement with Dr. D.F.H. Chowdhury on November 2, 2013. Dr. D.F.H. Chowdhury’s contract is for an unspecified
period. He may leave the Company with notice, or the Company may terminate his contract with notice. Termination may be with or without
cause. Dr. D.F.H. Chowdhury receives an annual salary of £80,000 pounds sterling (approximately $109,000). Our contract with Dr.
D.F.H. Chowdhury does not include any provision for stock options or equity incentives.
Under
the executive employment agreement Dr. D.F.H. Chowdhury’s annual salary was adjusted on a pro rata basis to reflect only work that
was performed for Nemaura Medical Inc. The disclosure set forth in the table reflects his pro rata compensation for the periods ending
March 31, 2022 and March 31, 2021, respectively.
On
July 11, 2022, the Compensation Committee and the Board of Directors has approved an increase in the annual salary of Dr. Chowdhury, the
Chief Executive Officer, to $400,000 per annum, and has approved a discretionary bonus to Mr. Chowdhury in an amount up to $1,000,000,
to be determined and issued at the discretion of the Board of Directors.
Outstanding
Equity Awards for fiscal year ended March 31, 2022.
The
table below sets forth the outstanding option awards for the named executive officers, as of March 31, 2022; there were no outstanding
stock awards as of this date:
| |
|
Number of
Securities
Underlying
Unexercised
Options
(#)
Exercisable |
|
|
Number of
Securities
Underlying
Unexercised
Options
(#)
Unexercisable |
|
|
Option
Exercise
Price
($) |
|
|
Option
Expiration
Date |
| Dr. D.F.H. Chowdhury |
|
|
8,000 |
|
|
|
— |
|
|
|
3.98 |
|
|
January 28, 2027 |
Potential
payments upon termination or change-in-control.
None.
Director
Compensation
Each
of our independent directors received annual fees of £5,000 pounds sterling (approximately $6,838) for the year ended March 31,
2022, for their service on our board of directors and committees. In addition, on January 28, 2022, the Board of Directors granted to
each of the directors, including Dr. Chowdhury, an option to purchase 8,000 shares of common stock at an exercise price of $3.98 per share,
the closing price of the Company’s common stock on the date of grant. The option was fully vested at grant and is exercisable for
a period of five years from the date of grant.
| Name |
|
Fees Earned or
Paid in Cash
($) |
|
|
Option Awards
($) |
|
All Other Compensation
($) |
|
|
Total
($) |
|
| Timothy Johnson |
|
|
6,838 |
|
|
26,706 (1) |
|
|
— |
|
|
|
33,544 |
|
| Dr. Salim Natha |
|
|
6,838 |
|
|
26,706 (1) |
|
|
— |
|
|
|
33,544 |
|
| Thomas Moore |
|
|
6,838 |
|
|
26,706 (1) |
|
|
— |
|
|
|
33,544 |
|
| |
(1) |
On January 28, 2022, the Board of Directors granted to each of the directors, including Dr. Chowdhury, an option to purchase 8,000 shares of common stock at an exercise price of $3.98 per share, the closing price of the Company’s common stock on the date of grant. The option was fully vested at grant and is exercisable for a period of five years from the date of grant. The fair value attributed to the options has been calculated using a Black-Scholes Option Pricing Model. |
SECURITY
OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The
following tables set forth certain information as of February 28, 2023, regarding the beneficial ownership of our common stock, by (i)
each person or entity who, to our knowledge, owns more than 5% of our common stock; (ii) our named executive officers; (iii) each director;
and (iv) all of our executive officers and directors as a group.
Unless
otherwise indicated in the footnotes to the following table, each person named in the table has sole voting and investment power and that
person’s address is c/o NEMAURA MEDICAL INC., Advanced Technology Innovation Centre, 5 Oakwood Drive, Loughborough, Leicestershire,
United Kingdom LE11 3QF.
| Name of Beneficial Owner |
|
Amount and Nature of Beneficial
Ownership |
|
|
Percentage (1) |
|
| Dr. D.F.H. Chowdhury |
|
|
8,761,700 |
(2) |
|
|
30.03% |
|
| Bashir Timol |
|
|
2,798,310 |
(3) |
|
|
9.7% |
|
| Timothy Johnson |
|
|
8,000 |
(4) |
|
|
* |
|
| Dr. Salim Natha |
|
|
427,390 |
(2) |
|
|
1.5% |
|
| Thomas Moore |
|
|
8,000 |
(4) |
|
|
* |
|
| All Executive Officers and Directors as a Group (5 persons) |
|
|
12,003,400 |
(5) |
|
|
41.5% |
|
| Holders of 5% or more of our common stock |
|
|
|
|
|
|
|
|
| Sufyan Ismail (6) |
|
|
2,216,395 |
|
|
|
7.9% |
|
| Armistice Capital Master Fund Ltd. (7) |
|
|
2,398,103 |
|
|
|
4.99% |
|
| Alyeska Master Fund, L.P. (8) |
|
|
4,881,374 |
|
|
|
9.99% |
|
*
Less than 1%. 31,297,505
| (1) | | Based upon 28,899,402 shares of our common stock outstanding at February 28, 2023. |
| (2) | | Includes 8,000 shares the reporting person has the right to acquire within 60 days of February
28, 2023 upon exercise of a vested option to purchase 8,000 shares of common stock. |
| (3) | | Represents (i) 2,708,210 shares held directly by the reporting person, (ii) 82,100 shares
held by the reporting person’s spouse, and (iii) 8,000 shares the reporting person has the right to acquire within 60 days of February
28, 2023 upon exercise of a vested option to purchase 8,000 shares of common stock. |
| (4) | | Represents 8,000 shares the reporting person has the right to acquire within 60 days of February
28, 2023 upon exercise of a vested option to purchase 8,000 shares of common stock. |
| (5) | | Includes 40,000 shares the Company’s executive officers and directors have the right
to acquire within 60 days of February 28, 2023 upon exercise of vested options to purchase 40,000 shares of common stock. |
| (6) | | Mr. Ismail’s address is Hollybank High Bank Lane, Lostock, Bolton, Lancashire BL6 HDT
United Kingdom. Represents (i) 2,134,295 shares held directly by the reporting person and (ii) 82,100 shares held by the reporting person’s
spouse. |
| (7) | | Includes 2,398,103 shares of our common stock issuable upon exercise of the warrants held
by Armistice Capital Master Fund Ltd. (“Master Fund”), which warrants are subject to, as applicable, certain beneficial ownership
limitations, which provide that the holder of such warrants will not have the right to exercise any portion thereof if such holder, together
with its affiliates, would beneficially own in excess of 4.99% of the number of shares of common stock outstanding immediately after
giving effect to such exercise, provided that upon at least 61 days’ prior notice to us, such holder may increase or decrease such
limitation up to a maximum of 9.99% of the number of shares of common stock outstanding. Steven Boyd, CIO of Armistice Capital, LLC (“Armistice”),
has voting control and investment discretion over the securities held by Master Fund. Armistice
and Steven Mr. Boyd disclaim beneficial ownership of the reported securities except to the extent of their respective pecuniary interest
therein. Accordingly, notwithstanding the number of shares of common stock listed above as being beneficially owned by the Master Fund,
Armistice and Mr. Boyd, each of the Master Fund, Armistice and Mr. Boyd further disclaim beneficial ownership of the shares of common
stock issuable upon exercise of all of the warrants to the extent the number of shares of common stock beneficially owned by each of
the Master Fund, Armistice and Mr. Boyd and any other person or entities with which their respective beneficial ownership would be aggregated
for purposes of Section 13(d) of the Exchange Act would exceed 9.99% of the total number of shares of common stock outstanding. The
address of Armistice Capital Master Fund Ltd. is 510 Madison Avenue, 7th Floor, New York, NY 10022. |
| (8) | | Includes (a) 2,483,271 shares of common stock directly held by Alyeska Master Fund, L.P.
and (b) 2,398,103 shares of our common stock issuable upon exercise of the Warrants held by Alyeska Master Fund, L.P., which Warrants
are subject to, as applicable, certain beneficial ownership limitations, which provide that the holder of such warrants will not have
the right to exercise any portion thereof if such holder, together with its affiliates, would beneficially own in excess of 4.99% (or
9.99%, as applicable), of the number of shares of common stock outstanding immediately after giving effect to such exercise, provided
that upon at least 61 days’ prior notice to us, such holder may increase or decrease such limitation up to a maximum of 9.99% of
the number of shares of common stock outstanding. Alyeska Investment Group, L.P., the investment manager of Alyeska Master Fund, L.P.,
has voting and investment control of the shares held by Alyeska Master Fund, L.P. Anand Parekh is the Chief Executive Officer of Alyeska
Investment Group, L.P. and may be deemed to be the beneficial owner of such shares. Mr. Parekh, however, disclaims any beneficial ownership
of the shares held by Alyeska Master Fund, L.P. The registered address of Alyeska Master Fund, L.P. is at c/o Maples Corporate Services
Limited, P.O. Box 309, Ugland House, South Church Street George Town, Grand Cayman, KY1-1104, Cayman Islands. Alyeska Investment Group,
L.P. is located at 77 W. Wacker, Suite 700, Chicago IL 60601. |
CERTAIN
RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
Pharma
and NDM are entities controlled by our Chief Executive Officer, President, Chairman of the Board and majority shareholder, Dr. D.F.H.
Chowdhury.
Pharma
has invoiced our subsidiaries, DDL and TCL for research and development services. In addition, certain operating expenses of DDL and TCL
were incurred and paid by Pharma and NDM which have been invoiced to us. Certain costs incurred by Pharma and NDM are directly attributable
to DDL and TCL and such costs were billed to us.
Total
costs charged to us by Pharma and NDM were $3,245,985 for the year ended March 31, 2022.
DDL has a service agreement with Nemaura Pharma
Limited (“Pharma”), an entity controlled by the Company’s President and Chief Executive officer, to provide development,
manufacture, and regulatory approval process under Pharma’s ISO13485 accreditation. Pharma invoices DDL for these services on a
cost-plus basis.
The table below provides a summary of activity between
the Company and Pharma for the nine months ended December 31, 2022 and 2021, and the year ended March 31, 2022.
| |
|
Nine Months Ended
December 31, 2022
(unaudited) |
|
|
Nine Months Ended
December 31, 2021
(unaudited) |
|
|
Year Ended
March 31, 2022 |
|
| Due to (from) related parties at beginning of period |
|
$ |
(101,297 |
) |
|
$ |
148,795 |
|
|
$ |
148,795 |
|
| Amounts invoiced by Pharma to DDL |
|
|
2,833,546 |
|
|
|
2,114,801 |
|
|
|
3,245,985 |
|
| Amounts invoiced by DDL to Pharma |
|
|
(3,159 |
) |
|
|
(2,495 |
) |
|
|
(2,495 |
) |
| Amounts paid by DDL to Pharma |
|
|
(2,785,487 |
) |
|
|
(2,316,544 |
) |
|
|
(3,492,962 |
) |
| Foreign exchange differences |
|
|
31,077 |
|
|
|
(97,149 |
) |
|
|
(620 |
) |
| Due to (from) related parties at end of period |
|
$ |
(25,320 |
) |
|
$ |
(152,592 |
) |
|
$ |
(101,297 |
) |
DESCRIPTION OF COMMON STOCK
General
The following description of our common stock is a
summary and does not purport to be complete. It is subject to and qualified in its entirety by reference to our Articles of Incorporation,
as amended (the “Articles of Incorporation”), and our Amended and Restated Bylaws (the “Bylaws”) which are filed
as exhibits to the registration statement of which this prospectus is a part. We encourage you to read our Articles of Incorporation,
our Bylaws and the applicable provisions of the Nevada Revised Statutes (“NRS”) for additional information.
As of February 28, 2023, our authorized capital
stock consists of 42,000,000 shares of common stock, par value $0.001 per share, of which 28,899,402 shares were issued and outstanding,
and 200,000 shares of preferred stock, par value $0.001, of which no shares were issued and outstanding. The authorized and unissued shares
of both common and preferred stock are available for issuance without further action by the Company’s stockholders, unless such
action is required by applicable law, the Nasdaq Capital Market, or the rules of any other stock exchange on which our securities may
be listed. Unless approval of the Company’s stockholders is so required, the Company’s board of directors will not seek stockholder
approval for the issuance and sale of either our common stock or preferred stock.
Common Stock
Holders of the Company’s common stock are entitled
to one vote for each share on all matters submitted to a stockholder vote. Holders of common stock do not have cumulative voting rights.
Therefore, holders of a majority of the shares of common stock voting for the election of directors can elect all of the directors. Holders
of the Company’s common stock representing a majority of the voting power of the Company’s capital stock issued, outstanding
and entitled to vote, represented in person or by proxy, are necessary to constitute a quorum at any meeting of stockholders.
Holders of the Company’s common stock are entitled
to share in all dividends that our Board of Directors, in its discretion, declares from legally available funds. In the event of a liquidation,
dissolution or winding up, each outstanding share entitles its holder to participate pro rata in all assets that remain after payment
of liabilities and after providing for each class of stock, if any, having preference over the common stock. The Company’s common
stock has no pre-emptive rights, no conversion rights and there are no redemption provisions applicable to the Company’s common
stock.
Anti-Takeover Effects of Various Provisions of
Nevada Law and our Articles of Incorporation
Provisions of the NRS and our
Articles of Incorporation and Bylaws could make it more difficult to acquire us by means of a tender offer, a proxy contest or otherwise,
or to remove incumbent officers and directors. These provisions, summarized below, would be expected to discourage certain types of coercive
takeover practices and takeover bids our board of directors may consider inadequate and to encourage persons seeking to acquire control
of us to first negotiate with us. We believe that the benefits of increased protection of our ability to negotiate with the proponent
of an unfriendly or unsolicited proposal to acquire or restructure us will outweigh the disadvantages of discouraging takeover or acquisition
proposals because, among other things, negotiation of these proposals could result in an improvement of their terms.
Effects of authorized but
unissued common stock and blank check preferred stock. One of the effects of the existence of authorized but unissued
common stock and undesignated preferred stock may be to enable our board of directors to make more difficult or to discourage an attempt
to obtain control of our company by means of a merger, tender offer, proxy contest or otherwise, and thereby to protect the continuity
of management. If, in the due exercise of its fiduciary obligations, the board of directors were to determine that a takeover proposal
was not in our best interest, such shares could be issued by the board of directors without stockholder approval in one or more transactions
that might prevent or render more difficult or costly the completion of the takeover transaction by diluting the voting or other rights
of the proposed acquirer or insurgent stockholder group, by putting a substantial voting block in institutional or other hands that might
undertake to support the position of the incumbent board of directors, by effecting an acquisition that might complicate or preclude the
takeover, or otherwise.
In addition, our Articles of Incorporation
grants our board of directors broad power to establish the rights and preferences of authorized and unissued shares of preferred stock.
The issuance of shares of preferred stock could decrease the amount of earnings and assets available for distribution to holders of shares
of common stock. The issuance also may adversely affect the rights and powers, including voting rights, of those holders and may have
the effect of delaying, deterring or preventing a change in control of our company.
Interested Stockholder Statute. We
are subject to Nevada’s Combination with Interested Stockholders Statute (Nevada Revised Statutes (“NRS”) Sections 78.411
through 78.444) which prohibits an “interested stockholder” from entering into a “combination” with us, unless
certain conditions are met. An “interested stockholder” is a person who, together with affiliates and associates, beneficially
owns (or within the prior two years, did beneficially own) 10% or more of our capital stock entitled to vote. We have, however, elected
in our Amended and Restated Articles of Incorporation to not be governed by the provisions of NRS Sections 78.411 through 78.444.
Control Share Statute.
We are subject to Nevada’s Control Share Statute (NRS Sections 78.378 to 78.3793) which restricts the ability of individuals and
groups from acquiring one-fifth or more of the voting shares of a Nevada corporation that has 200 or more stockholders of record, at least
100 of whom have addresses in Nevada, from exercising the voting rights of the acquired shares, absent required stockholder approval of
the share acquisition transaction or an opt out election by the corporation. The prohibition on the voting of the acquired shares is limited
to three years after acquisition. To avoid the voting restriction, the acquisition of a controlling interest must be approved by both
(a) the holders of a majority of the voting power of the corporation, and (b) if the acquisition would adversely alter or change any preference
or any relative or other right given to any other class or series of outstanding shares, the holders of the majority of each class or
series affected, excluding those shares as to which any interested stockholder exercises voting rights, and the approval must specifically
include the conferral of such voting rights. Although we have not opted out of this statute, a corporation alternatively may expressly
elect not to be governed by the provisions in either its articles of incorporation or its bylaws. Additionally, in the face of potential
control share transaction, a corporation, if it has not opted out of the statutory provisions, may opt out of the control share statute
by amending its articles of incorporation or its bylaws prior to the 10th day following the acquisition of a controlling interest by an
acquiring person.
Limitations on Liability
and Indemnification of Officers and Directors. NRS limits or eliminates the personal liability of directors to corporations
and their stockholders for monetary damages for breaches of directors’ fiduciary duties as directors. Our Amended and Restated Articles
of Incorporation include provisions that require us to indemnify, to the fullest extent allowable under the NRS, our directors or officers
against monetary damages for actions taken as a director or officer of our company, or for serving at our request as a director or officer
or another position at another corporation or enterprise, as the case may be. Our Amended and Restated Articles of Incorporation also
provide that we must indemnify and advance reasonable expenses to our directors and officers, subject to our receipt of an undertaking
from the indemnified party as may be required under the NRS. We are also expressly authorized to carry directors’ and officers’
insurance to protect our company, our directors, officers and certain employees for some liabilities.
The limitation of liability and
indemnification provisions under the NRS and in our Amended and Restated Articles of Incorporation and Bylaws may discourage stockholders
from bringing a lawsuit against directors for breach of their fiduciary duties. These provisions may also have the effect of reducing
the likelihood of derivative litigation against directors and officers, even though such an action, if successful, might otherwise benefit
us and our stockholders. However, these provisions do not limit or eliminate our rights, or those of any stockholder, to seek non-monetary
relief such as injunction or rescission in the event of a breach of a director’s fiduciary duties. Moreover, the provisions do not
alter the liability of directors under the federal securities laws.
Transfer Agent
Nevada Agency and Transfer Company
(“Transfer Agent”) is our transfer agent and registrar. The Transfer Agent’s address is at 50 West Liberty Street, Suite
880, Reno Nevada 89501 and its telephone number is (775) 322 0626.
SHARES ELIGIBLE FOR FUTURE
SALE
We cannot predict the effect,
if any, that market sales of shares of our common stock or the availability of shares of our common stock for sale will have on the market
price of our common stock prevailing from time to time. Future sales of our common stock in the public market, or the availability of
such shares for sale in the public market, could adversely affect market prices prevailing from time to time. The availability for sale
of a substantial number of shares of our common stock acquired through the exercise of outstanding warrants could materially adversely
affect the market price of our common stock. In addition, sales of our common stock in the public market after the restrictions lapse
as described below, or the perception that those sales may occur, could cause the prevailing market price to decrease or to be lower than
it might be in the absence of those sales or perceptions.
Sale of Restricted Shares
As of February 28, 2023,
there were 28,899,402 shares of common stock outstanding. The 4,796,206 shares of common stock being offered by this Prospectus will be
freely tradable, other than by any of our “affiliates,” as defined in Rule 144(a) under the Securities Act, without restriction
or registration under the Securities Act.
Rule 144
In general, under Rule 144,
as currently in effect, a person (or persons whose shares are required to be aggregated), including a person who may be deemed an “affiliate”
of a company, who has beneficially owned restricted securities for at least six months may sell, within any three-month period, a number
of shares that does not exceed the greater of: (1) 1% of the then-outstanding shares of common stock, or (2) if and when the common stock
is listed on a national securities exchange, the average weekly trading volume of the common stock during the four calendar weeks preceding
the date on which notice of such sale was filed under Rule 144. Sales under Rule 144 are also subject to certain requirements as to the
manner of sale, notice, and availability of current public information about our company. A person who is not deemed to have been an affiliate
of us at any time during the 90 days preceding a sale by such person, and who has beneficially owned the restricted shares for at least
one year, is entitled to sell such shares under Rule 144 without regard to any of the restrictions described above.
We cannot estimate the number
of shares of our common stock that our existing stockholders will elect to sell under Rule 144.
LEGAL MATTERS
The validity of the securities
offered by this prospectus will be passed upon for us by Anthony L.G., PLLC, 625 N. Flagler Drive, Suite 600, West Palm Beach, Florida
33401.
EXPERTS
The consolidated financial statements of Nemaura Medical, Inc. as
of and for the years ended March 31, 2022 and 2021 included in this registration statement, of which this prospectus forms a part, have
been audited by Mayer Hoffman McCann P.C., independent registered public accounting firm, as set forth in their report (which includes
an explanatory paragraph related to the existence of substantial doubt about the Company’s ability to continue as a going concern)
appearing elsewhere herein, and are included in reliance upon such report given on the authority of such firm as experts in auditing and
accounting in giving said report.
DISCLOSURE OF COMMISSION’S
POSITION ON INDEMNIFICATION FOR SECURITIES ACT LIABILITIES
Our directors and officers
are indemnified as provided by Nevada law, our articles of incorporation, as amended, and our bylaws. We have agreed to indemnify each
of our directors and certain officers against certain liabilities, including liabilities under the Securities Act. Insofar as indemnification
for liabilities arising under the Securities Act may be permitted to our directors, officers and controlling persons pursuant to the provisions
described above, or otherwise, we have been advised that in the opinion of the SEC, such indemnification is against public policy as expressed
in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other
than our payment of expenses incurred or paid by our director, officer or controlling person in the successful defense of any action,
suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, we
will, unless in the opinion of our counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction
the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the
final adjudication of such issue.
WHERE YOU CAN FIND ADDITIONAL
INFORMATION
We have filed with the SEC
the registration statement on Form S-1 under the Securities Act for the securities offered by this prospectus. This prospectus, which
is a part of the registration statement, does not contain all of the information in the registration statement and the exhibits filed
with it, portions of which have been omitted as permitted by SEC rules and regulations. For further information concerning us and the
securities offered by this prospectus, we refer to the registration statement and to the exhibits filed with it. Statements contained
in this prospectus as to the content of any contract or other document referred to are not necessarily complete. In each instance, we
refer you to the copy of the contracts and/or other documents filed as exhibits to the registration statement.
The registration statement
on Form S-1, of which this prospectus forms a part, including exhibits, is available at the SEC’s website at http://www.sec.gov.
You may also read and copy any document we file with, or furnish to, the SEC at its public reference facilities:
| |
Public Reference Room Office |
| |
100 F Street, N.E. |
| |
Room 1580 |
| |
Washington, D.C. 20549 |
You may also obtain copies
of the documents at prescribed rates by writing to the Public Reference Section of the SEC at 100 F Street, N.E., Room 1580, Washington,
D.C. 20549. Callers in the United States can also call (202) 551-8090 for further information on the operations of the public reference
facilities.
NEMAURA MEDICAL, INC.
Index to Financial Statements
REPORT OF INDEPENDENT REGISTERED PUBLIC
ACCOUNTING FIRM
To the Board of Directors and Stockholders of Nemaura Medical
Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated
balance sheets of Nemaura Medical Inc. (the Company) as of March 31, 2022 and 2021, and the related consolidated statements of operations
and comprehensive loss, stockholders’ equity, and cash flows for each of the years in the two-year period ended March 31, 2022,
and the related notes (collectively referred to as the financial statements). In our opinion, the financial statements present fairly,
in all material respects, the financial position of the Company as of March 31, 2022 and 2021, and the results of its operations and its
cash flows for each of the years in the two-year period ended March 31, 2022, in conformity with accounting principles generally accepted
in the United States of America.
Substantial Doubt about Going Concern
The accompanying financial statements have
been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the financial statements, the Company
has suffered recurring losses from operations, current debt due over current cash balances, and has accumulated deficits that raised substantial
doubt about its ability to continue as a going concern. Management's plans in regard to these matters are also described in Note 1. The
financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These financial statements are the responsibility
of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our
audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are
required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and
regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with
the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the
financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we
engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding
of internal control over financial reporting, but not for the purpose of expressing an opinion on the effectiveness of the Company’s
internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures
to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that
respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial
statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as
evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matters
Critical audit matters are matters arising
from the current period audit of the financial statements that were communicated or required to be communicated to the audit committee
and that (1) relate to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging,
subjective, or complex judgments. We determined that there were no critical audit matters.
/s/ Mayer Hoffman McCann P.C.
We have served as the Company’s auditor since 2018.
Denver, Colorado
June 29, 2022
NEMAURA MEDICAL INC.
Consolidated Balance Sheets
| | |
| | | |
| | |
| | |
As of March 31, | | |
As of March 31, | |
| | |
2022 ($) | | |
2021 ($) | |
| | |
| | |
| |
| ASSETS | |
| | | |
| | |
| Current assets: | |
| | | |
| | |
| Cash | |
| 17,749,233 | | |
| 31,865,371 | |
| Prepaid expenses and other receivables | |
| 750,167 | | |
| 1,269,513 | |
| Accounts receivable – related party | |
| 101,297 | | |
| — | |
| Inventory | |
| 1,487,771 | | |
| 850,622 | |
| Total current assets | |
| 20,088,468 | | |
| 33,985,506 | |
| | |
| | | |
| | |
| Other assets: | |
| | | |
| | |
| Property and equipment, net of accumulated depreciation | |
| 532,508 | | |
| 202,145 | |
| Intangible assets, net of accumulated amortization | |
| 1,480,980 | | |
| 1,055,256 | |
| Total other assets | |
| 2,013,488 | | |
| 1,257,401 | |
| Total assets | |
| 22,101,956 | | |
| 35,242,907 | |
| | |
| | | |
| | |
| LIABILITIES AND STOCKHOLDERS’ EQUITY | |
| | | |
| | |
| Current liabilities: | |
| | | |
| | |
| Accounts payable | |
| 136,310 | | |
| 253,694 | |
| Liability due to related parties | |
| — | | |
| 148,795 | |
| Other liabilities and accrued expenses | |
| 998,622 | | |
| 180,522 | |
| Notes payable, current portion | |
| 19,188,724 | | |
| 5,733,370 | |
| Deferred revenue | |
| 259,256 | | |
| 103,470 | |
| Total current liabilities | |
| 20,582,912 | | |
| 6,419,881 | |
| | |
| | | |
| | |
| Non-current portion of notes payable | |
| — | | |
| 19,188,724 | |
| Non-current portion of deferred revenue | |
| 1,052,960 | | |
| 1,276,130 | |
| Total liabilities | |
| 21,635,872 | | |
| 26,884,735 | |
| | |
| | | |
| | |
| Commitments and contingencies | |
| | | |
| | |
| | |
| | | |
| | |
| Stockholders’ equity: | |
| | | |
| | |
| Common stock, par value $0.001 - authorized: 42,000,000 shares; issued and outstanding: 24,102,866 and 22,941,157 as of March 31, 2022 and 2021, respectively | |
| 24,103 | | |
| 22,941 | |
| Additional paid-in capital | |
| 38,295,775 | | |
| 32,044,335 | |
| Accumulated deficit | |
| (37,731,476 | ) | |
| (23,844,671 | ) |
| Accumulated other comprehensive (loss) income | |
| (122,318 | ) | |
| 135,567 | |
| Total stockholders’ equity | |
| 466,084 | | |
| 8,358,172 | |
| Total liabilities and stockholders’ equity | |
| 22,101,956 | | |
| 35,242,907 | |
See notes to consolidated financial
statements.
NEMAURA MEDICAL INC.
Consolidated Statements of Operations
and Comprehensive Loss
| | |
| | | |
| | |
| | |
Years Ended March 31, | |
| | |
2022
($) | | |
2021
($) | |
| Sales | |
| 503,906 | | |
| — | |
| Cost of Sales | |
| (344,300 | ) | |
| — | |
| Gross Profit | |
| 159,606 | | |
| — | |
| | |
| | | |
| | |
| Operating expenses: | |
| | | |
| | |
| Research and development | |
| 1,556,988 | | |
| 1,554,603 | |
| General and administrative | |
| 6,173,049 | | |
| 3,032,138 | |
| Total operating expenses | |
| 7,730,037 | | |
| 4,586,741 | |
| | |
| | | |
| | |
| Loss from operations | |
| (7,570,431 | ) | |
| (4,586,741 | ) |
| | |
| | | |
| | |
| Interest expense | |
| (6,666,630 | ) | |
| (2,007,687 | ) |
| | |
| | | |
| | |
| Provision for income tax benefit | |
| 350,256 | | |
| 335,832 | |
| Net loss | |
| (13,886,805 | ) | |
| (6,258,596 | ) |
| | |
| | | |
| | |
| Other comprehensive income: | |
| | | |
| | |
| Foreign currency translation adjustment | |
| (257,885 | ) | |
| 472,559 | |
| Comprehensive loss | |
| (14,144,690 | ) | |
| (5,786,037 | ) |
| | |
| | | |
| | |
| Net loss per share, basic and diluted | |
$ | (0.59 | ) | |
$ | (0.28 | ) |
| Weighted average number of shares outstanding | |
| 23,383,758 | | |
| 22,283,377 | |
See notes to consolidated financial
statements.
NEMAURA MEDICAL INC.
Consolidated Statements of Changes in
Stockholders’ Equity
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| | |
| Common
Stock | | |
| | | |
| | | |
| | | |
| | |
| | |
| Shares | | |
| Amount
($) | | |
| Additional
Paid-in Capital ($) | | |
| Accumulated
Deficit ($) | | |
| Accumulated
Other Comprehensive Loss ($) | | |
| Total
Stockholders’ Equity (Deficit) ($) | |
| Balance
at March 31, 2020 | |
| 20,850,848 | | |
| 20,851 | | |
| 16,589,272 | | |
| (17,586,075 | ) | |
| (336,992 | ) | |
| (1,312,944 | ) |
| Issuance of common shares, net of costs of $957,193 | |
| 1,994,924 | | |
| 1,995 | | |
| 14,791,484 | | |
| — | | |
| — | | |
| 14,793,479 | |
| Exercise of warrants | |
| 38,683 | | |
| 38 | | |
| 400,465 | | |
| — | | |
| — | | |
| 400,503 | |
| Restricted shares issued as stock-based
compensation | |
| 56,702 | | |
| 57 | | |
| 263,114 | | |
| — | | |
| — | | |
| 263,171 | |
| Foreign currency translation adjustment | |
| — | | |
| — | | |
| — | | |
| — | | |
| 472,559 | | |
| 472,559 | |
| Net loss | |
| — | | |
| — | | |
| — | | |
| (6,258,596 | ) | |
| — | | |
| (6,258,596 | ) |
| Balance at
March 31, 2021 | |
| 22,941,157 | | |
| 22,941 | | |
| 32,044,335 | | |
| (23,844,671 | ) | |
| 135,567 | | |
| 8,358,172 | |
| Issuance of common shares, net of costs of $50,765 | |
| 772,524 | | |
| 773 | | |
| 3,067,254 | | |
| — | | |
| — | | |
| 3,068,027 | |
| Exercise of warrants | |
| 366,892 | | |
| 367 | | |
| 2,963,291 | | |
| — | | |
| — | | |
| 2,963,658 | |
| Restricted shares issued as stock-based
compensation | |
| 22,293 | | |
| 22 | | |
| 87,366 | | |
| — | | |
| — | | |
| 87,388 | |
| Options issued to directors | |
| — | | |
| — | | |
| 133,529 | | |
| — | | |
| — | | |
| 133,529 | |
| Foreign currency translation adjustment | |
| — | | |
| — | | |
| — | | |
| — | | |
| (257,885 | ) | |
| (257,885 | ) |
| Net loss | |
| — | | |
| — | | |
| — | | |
| (13,886,805 | ) | |
| — | | |
| (13,886,805 | ) |
| Balance at
March 31, 2022 | |
| 24,102,866 | | |
| 24,103 | | |
| 38,295,775 | | |
| (37,731,476 | ) | |
| (122,318 | ) | |
| 466,084 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
See notes to consolidated financial statements.
NEMAURA MEDICAL INC.
Consolidated Statements of Cash Flows
| | |
| | | |
| | |
| | |
Year Ended March 31, | |
| | |
2022
($) | | |
2021
($) | |
| Cash Flows from Operating Activities: | |
| | | |
| | |
| Net loss | |
| (13,886,805 | ) | |
| (6,258,596 | ) |
| | |
| | | |
| | |
| Adjustments to reconcile net loss to net cash used in operating activities: | |
| | | |
| | |
| Depreciation and amortization | |
| 229,810 | | |
| 98,075 | |
| Accretion of debt discount | |
| 6,666,630 | | |
| 2,007,687 | |
| Mark-to-market foreign exchange revaluation | |
| 440,196 | | |
| — | |
| Stock-based compensation | |
| 220,917 | | |
| 113,171 | |
| | |
| | | |
| | |
| Changes in assets and liabilities: | |
| | | |
| | |
| Prepaid expenses and other receivables | |
| 519,346 | | |
| (767,050 | ) |
| Inventory | |
| (637,149 | ) | |
| (564,313 | ) |
| Accounts payable | |
| (117,384 | ) | |
| (39,914 | ) |
| Accounts receivable – related party | |
| (250,092 | ) | |
| (681,298 | ) |
| Other liabilities and accrued expenses | |
| 310,490 | | |
| 94,141 | |
| Net cash used in operating activities | |
| (6,504,041 | ) | |
| (5,998,097 | ) |
| | |
| | | |
| | |
| Cash Flows from Investing Activities: | |
| | | |
| | |
| Capitalized patent costs | |
| (83,691 | ) | |
| (81,952 | ) |
| Purchase of property and equipment | |
| (481,718 | ) | |
| (90,730 | ) |
| Capitalized software development costs | |
| (391,073 | ) | |
| (663,758 | ) |
| Net cash used in investing activities | |
| (956,482 | ) | |
| (836,440 | ) |
| | |
| | | |
| | |
| Cash Flows from Financing Activities: | |
| | | |
| | |
| Proceeds from issuance of common stock | |
| 3,118,792 | | |
| 15,750,672 | |
| Costs incurred in relation to equity financing | |
| (50,765 | ) | |
| (957,193 | ) |
| Proceeds from warrant exercise | |
| 2,963,658 | | |
| 400,503 | |
| Proceeds from issuance of notes payable | |
| — | | |
| 25,000,000 | |
| Debt issuance costs paid | |
| — | | |
| (1,525,035 | ) |
| Repayments of notes payable | |
| (12,400,000 | ) | |
| (600,000 | ) |
| Repayments of insurance financing | |
| — | | |
| (82,555 | ) |
| Net cash (used in) provided by financing activities | |
| (6,368,315 | ) | |
| 37,986,392 | |
| | |
| | | |
| | |
| Net (decrease) increase in cash | |
| (13,828,838 | ) | |
| 31,151,855 | |
| Effect of exchange rate changes on cash | |
| (287,300 | ) | |
| 607,409 | |
| Cash at beginning of year | |
| 31,865,371 | | |
| 106,107 | |
| Cash at end of year | |
| 17,749,233 | | |
| 31,865,371 | |
Supplemental disclosure of non-cash financing activities: | |
| | | |
| | |
| Prepayment of equity compensation | |
| — | | |
$ | 50,000 | |
| Licenses acquired through stock issuance | |
| — | | |
$ | 100,000 | |
| Monitoring fees added to notes payable | |
$ | 2,764,775 | | |
$ | 718,661 | |
See notes to consolidated financial
statements.
NEMAURA MEDICAL INC.
Notes to Consolidated Financial Statements
NOTE 1 – ORGANIZATION, PRINCIPAL ACTIVITIES AND
MANAGEMENT’S PLANS
Nemaura Medical Inc. (“Nemaura” or the “Company”),
through its operating subsidiaries, performs medical device research and manufacturing of a continuous glucose monitoring system (“CGM”),
named sugarBEAT®. The sugarBEAT® device is a non-invasive, wireless device for use by persons with Type I and Type II diabetes
and may also be used to screen pre-diabetic patients. The sugarBEAT® device extracts analytes, such as glucose, to the surface of
the skin in a non-invasive manner where it is measured using unique sensors and interpreted using a unique algorithm.
Nemaura is a Nevada holding company organized in 2013. Nemaura
owns one hundred percent (100%) of Dermal Diagnostic (Holdings) Limited, an England and Wales corporation (“DDHL”) formed
on December 11, 2013, which in turn owns one hundred percent (100%) of Dermal Diagnostics Limited, an England and Wales corporation formed
on January 20, 2009 (“DDL”), and one hundred percent (100%) of Trial Clinic Limited, an England and Wales corporation formed
on January 12, 2011 (“TCL”).
DDL is a diagnostic medical device company headquartered in
Loughborough, Leicestershire, England, and is engaged in the discovery, development and commercialization of diagnostic medical devices.
The Company’s initial focus has been on the development of the sugarBEAT® device, which consists of a disposable patch containing
a sensor, and a non-disposable miniature transmitter device with a re-chargeable power source, which is designed to enable trending or
tracking of blood glucose levels. All of the Company’s operations and assets are located in England.
The following diagram illustrates Nemaura’s
corporate structure as of March 31, 2022:

During the fiscal year ended March 31, 2021, the Board of Directors assessed the adequacy of the group’s organizational structure
and concluded that an intermediary holding company, Region Green Limited, was no longer required, as the entity had been effectively dormant
since inception, and no longer represented a requirement to be maintained. It was therefore determined that Region Green Limited should
be unwound, with the assets held by Region Green Limited being transferred up to Nemaura Medical Inc. following which Region Green Limited
would be dissolved.
The transfer of assets took place on March 5, 2021 and Region
Green Limited was formally dissolved as of April 23, 2021.
NEMAURA MEDICAL INC.
Notes to Consolidated Financial Statements
The Company was incorporated in 2013 and has reported recurring
losses from operations to date and an accumulated deficit of $37,731,476 as of March 31, 2022. These operations have resulted in the
successful completion of clinical programs to support a CE mark (European Union approval of the product) approval, as well as a De Novo
510(k) medical device application to the U.S. Food and Drug Administration (“FDA”) submission. The Company expects to continue
to incur losses from operations until revenues are generated through licensing fees or product sales. However, given the completion of
the requisite clinical programs, these losses are expected to decrease over time. Management has entered into licensing, supply, or collaboration
agreements with unrelated third parties relating to the United Kingdom (“UK”), Europe, Qatar, and all countries in the Gulf
Cooperation Council.
Going Concern
As identified under Item 1A, management is
aware of the need to raise additional funds in order to finance the ongoing commercialization of sugarBEAT®. The Company had $17,749,233
of cash at March 31, 2022, which management consider to be more than sufficient to fund the ongoing operational expenses of the business,
however the terms of the existing debt held on balance sheet will fall due for repayment as of February 2023, which will trigger a requirement
to either restructure the debt or obtain additional, new, funding.
In evaluating the going concern position
of the company, management have considered the ability of the company to raise additional funding in combination with one or more of the
different funding options available to it at this time. Based on current and ongoing engagement with potential funding providers,
Management believe that there is a reasonable expectation that funding could be provided by one, or more, of the following options:
| · | Equity funding – the company
has immediate access to funds through the ATM facility that is currently in place; in addition to this, there are various alternative
mechanisms available to the company similar to those used previously e.g. direct sale of shares to interested third parties, similar to
the stake sold to Tiger Trading Partners L.L.C. in February 2022, as well as other mechanisms to sell common stock via an underwritten
agreement or the further exercise of warrants by the current warrant holders etc. |
| · | Debt funding – the company
continues to be in ongoing discussions with third party debt providers, including the incumbent, to enable the existing debt facility
to be restructured or renewed, should management feel that this route offers a more attractive option compared to the sale of equity that
is dependent on the current market conditions. |
| · | Alternative funding as used
in the past such as the sale of licenses. As product development is now at a significant more advanced stage then it was, it is
management’s belief that the sufficient funding could be provided through the sale of licenses in a similar way to the UK license
agreement sale that help provided early-stage development funding. |
However, as a consequence of this funding
requirement being triggered without the funding bridge having been put in place by the filing date of these consolidated financial statements,
ASC 205-40 requires that management recognise and disclose this point as an event which creates a substantial doubt as to the Company’s
ability to continue as a going concern for at least one year from the date of filing of these consolidate financial statements.
Following the receipt of the CE mark approval in the EU, and
in support of our plans for similar certification with the FDA in the U.S., our plan is to utilize the cash on hand to continue establishing
commercial manufacturing operations for the commercial supply of the sugarBEAT® device and sensor patches in our target markets.
Management's strategic plans include the following:
| – | support the UK and EU launch of sugarBEAT®; |
| – | obtaining further regulatory approval for the sugarBEAT® device in other
countries such as the U.S.; |
| – | exploring licensing and partnership opportunities in other territories;
|
| – | developing the sugarBEAT® device platform for commercialization across
other applications; and |
| – | pursue additional capital raising opportunities as and when required to
further enhance our growth plans. |
NOTE 2 – BASIS OF PRESENTATION
The accompanying consolidated financial statements include
the accounts of the Company and the Company’s subsidiaries, DDL, TCL, DDHL and RGL. The consolidated financial statements are prepared
in accordance with accounting principles generally accepted in the United States of America (“U.S. GAAP”), and all significant
intercompany balances and transactions have been eliminated in consolidation.
The functional currency for the majority
of the Company’s operations is the Great Britain Pound Sterling (“GBP”), and the reporting currency is the U.S. Dollar
(“U.S.$”).
NEMAURA MEDICAL INC.
Notes to Consolidated Financial Statements
NOTE 3 – SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
AND RECENT ACCOUNTING PRONOUNCEMENTS
Revenue recognition
The Company has considered the guidelines within the Financial
Accounting Standards Board’s (the “FASB”) Accounting Standards Codification (“ASC”) Topic 606, Revenue
from Contracts with Customers as a requirement of the revenue recognition that it commenced during the current fiscal year. This
standard applies to all contracts with customers, except for contracts that are within the scope of other standards, such as leases,
insurance, collaboration arrangements and financial instruments. Under ASC Topic 606, an entity recognizes revenue when its customer
obtains control of promised goods or services, in an amount that reflects the consideration that the entity expects to receive in exchange
for those goods or services.
To determine revenue recognition for arrangements that an entity
determines are within the scope of ASC Topic 606, the entity performs the following five steps: (i) identify the contract(s) with a customer;
(ii) identify the performance obligations in the contract; (iii) determine the transaction price; (iv) allocate the transaction price
to the performance obligations in the contract; and (v) recognize revenue when (or as) the entity satisfies a performance obligation.
The Company only applies the five-step model to contracts when
it is probable that the entity will collect the consideration it is entitled to in exchange for the goods or services it transfers to
the customer. At contract inception, once the contract is determined to be within the scope of ASC Topic 606, the Company assesses the
goods or services promised within each contract and determines those that are performance obligations and assesses whether each promised
good or service is distinct. The Company then recognizes as revenue the amount of the transaction price that is allocated to the respective
performance obligation when (or as) the performance obligation is satisfied.
The Company may enter into product development and other agreements
with collaborative partners. The terms of the agreements may include non-refundable signing and licensing fees, milestone payments and
royalties on any product sales derived from collaborations.
Deferred revenue
The Company has entered into license agreements and
recognizes up front license payments as revenue upon delivery of the license only if the license has stand-alone value to the
customer. However, where further performance criteria must be met, revenue is deferred and recognized on a basis that is considered
to be appropriate to the conditions associated with the license and over the period the Company is expected to complete these
performance obligations.
Royalty revenue will be recognized upon the sale of the related
products provided the Company has no remaining performance obligations under the agreement.
Research and development expenses
The Company charges research and development expenses to operations
as incurred. Research and development expenses primarily consist of salaries and related expenses for personnel and outside contractor
and consulting services. Other research and development expenses include the costs of materials and supplies used in research and development,
prototype manufacturing, clinical studies, related information technology and an allocation of facilities costs.
Cash
Cash consists primarily of cash deposits maintained in the
UK.
Fair value of financial instruments
In accordance with ASC 820, “Fair
Value Measurements and Disclosures,” the Company determines the fair value of financial instruments with the use of observable inputs
and minimize the use of unobservable inputs when measuring fair value. ASC 820 establishes a fair value hierarchy based on the level of
independent, objective evidence surrounding the inputs used to measure fair value. A financial instrument’s categorization within
the fair value hierarchy is based upon the lowest level of input that is significant to the fair value measurement. ASC 820 prioritizes
the inputs into three levels that may be used to measure fair value:
Level 1: Applies to assets
or liabilities for which there are quoted prices in active markets for identical assets or liabilities.
Level 2: Applies to assets or liabilities
for which there are inputs other than quoted prices that are observable for the asset or liability such as quoted prices for similar assets
or liabilities in active markets; quoted prices for identical assets or liabilities in markets with insufficient volume or infrequent
transactions (less active markets); or model-derived valuations in which significant inputs are observable or can be derived principally
from, or corroborated by, observable market data.
Level 3: Applies to assets or liabilities
for which there are unobservable inputs to the valuation methodology that are significant to the measurement of the fair value of the
assets or liabilities.
NEMAURA MEDICAL INC.
Notes to Consolidated Financial Statements
Property and equipment
Property and equipment is stated at cost and depreciated using
the straight-line method over the estimated useful lives of the assets, generally four to five years. This is charged to operating expenses.
Intangible assets
Intangible assets consist of licenses and
patents associated primarily with the sugarBEAT® device and are amortized on a straight-line basis, generally over their legal lives
of up to 20 years and are reviewed for impairment. Costs capitalized relate to invoices received from third parties and not any internal
costs. The Company evaluates its intangible assets (all have finite lives) and other long-lived assets for impairment whenever events
or circumstances indicate that they may not be recoverable, or at least annually. Recoverability of finite and other long-lived assets
is measured by comparing the carrying amount of an asset group to the future undiscounted net cash flows expected to be generated by that
asset group. The Company groups assets for purposes of such review at the lowest level for which identifiable cash flows of the asset
group are largely independent of the cash flows of the other groups of assets and liabilities. The amount of impairment to be recognized
for finite and other long-lived assets is calculated as the difference between the carrying value and the fair value of the asset group,
generally measured by discounting estimated future cash flows. There were no impairment indicators present during the fiscal years ended
March 31, 2022 or 2021.
Software development costs
Capitalization of software development costs incurred in the
research and development of new software products and enhancements to existing software products for external use begins when a product’s
technological feasibility has been established and ends when the resulting product is available for general market release. Amortization
of the capitalized software is classified within product cost of goods sold in the consolidated statements of operations and comprehensive
loss.
For each capitalized software
product, the annual amortization is equal to the greater of:
1. The amount computed using the ratio of software product’s
current fiscal year gross revenue bears to the total current fiscal year and anticipated future gross revenues for the product, or
2. The amount computed based on a straight-line
method over the remaining estimated economic life of the product, which can be a range between 3 – 8 years.
Annually, or more frequently if required by triggering events,
an analysis of the net realizable value of the capitalized software is completed and the amount by which unamortized software costs exceeds
the net realisable value, if any, is recognized as a charge to income in the period it is determined.
Inventory
Inventory is stated at the lower of cost or net realizable
value, with cost determined on a first-in first-out basis. At present all inventory relates to raw materials purchased from third parties
and to be used in the Company’s product.
Income taxes
Income taxes are accounted for under the asset and liability
method. Deferred income tax assets and liabilities are recognized for the future tax consequences attributable to differences between
the financial statement carrying amounts of existing assets and liabilities and their respective tax bases, and operating loss carry
forwards. Deferred income tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the
years in which those temporary differences are expected to be recovered or settled. The effect on deferred income tax assets and liabilities
of a change in tax rates is recognized in income in the period that includes the enactment date. A valuation allowance is provided to
reduce the carrying amount of deferred income tax assets if it is considered more likely than not that some portion, or all, of the deferred
income tax assets will not be realized.
The Company recognizes the effect of income tax positions only
if those positions are more likely than not of being sustained. Recognized income tax positions are measured at the largest amount that
is greater than 50% likely of being realized. Changes in recognition or measurement are reflected in the period in which the change in
judgment occurs. The Company has elected to classify interest and penalties related to unrecognized tax benefits as part of income tax
expense in the consolidated statements of comprehensive loss. The Company does not have any accrued interest or penalties associated
with any unrecognized tax benefits, nor was any interest expense related to unrecognized tax benefits recognized for the years ended
March 31, 2022 and 2021.
In December 2017, the U.S. Tax Cuts and
Jobs Act was signed into law. Generally, this Act reduces corporate rates from a top rate of 35% to a top rate of 21%, effective January
1, 2018. As the Company’s U.S. operations are minimal, and all deferred tax assets maintain a full valuation allowance, there is
no significant impact to the Company as of and for the years ended March 31, 2022 and 2021.
NEMAURA MEDICAL INC.
Notes to Consolidated Financial Statements
Earnings (loss) per share
Basic earnings (loss) per share is computed by dividing income
(loss) available to common stockholders by the weighted-average number of common shares outstanding during the period. For the fiscal
year ended March 31, 2022, warrants to purchase 1,573,098 shares of common stock, options to purchase 40,000 shares of common stock and
a unit purchase option to purchase 9,710 shares of common stock as well as 9,710 warrants were considered anti-dilutive and were excluded
from the calculation of diluted loss per share. For the fiscal year ended March 31, 2021, warrants to purchase 1,939,990 shares of common
stock and a unit purchase option to purchase 9,710 shares of common stock as well as 9,710 warrants were considered anti-dilutive and
were excluded from the calculation of diluted loss per share.
Use of estimates
The preparation of consolidated financial statements in conformity
with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure
of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts of revenues and expenses
during the periods presented. Actual results may differ from those estimates.
Foreign currency translation
The functional currency of the Company is the GBP, while the
reporting currency is the U.S.$. Assets and liabilities are translated at the exchange rates as of the balance sheet date with income
and expenses being translated at the weighted-average exchange rates prevailing during the reporting period. Stockholders' equity is
translated into U.S.$ from GBP at historical exchange rates.
Adjustments resulting from translating the consolidated financial
statements into U.S.$ are recorded as a separate component of accumulated other comprehensive loss in stockholders’ equity.
Derivative Financial Instruments
Derivative financial instruments are used as part of the overall
strategy to manage exposure to foreign currency primarily associated with fluctuations in foreign currency exchange rates. Derivative
financial instruments are included in the consolidated balance sheets and are measured at fair value on a recurring basis.
The Company is exposed to the impact of foreign currency exchange
fluctuations as a significant proportion of our expenses are incurred within our UK subsidiary which is denominated GBP, with the remaining
portion denominated in USD and a small amount in Euros (“EUR”). In addition to this, we hold the majority of our cash in
USD, with amounts also held in GBP and, to a much smaller amount, in EURs. The Company’s objective is to reduce the volatility
associated with these foreign exchange rate changes to allow management to focus our attention on our core business strategy and objectives.
Accordingly, the Company entered into a target accrual redemption forward (“TARF”) agreement to sell USD and buy GBP across
25 defined monthly fixings in order to fix the costs associated with the foreign currency exchange fluctuations associated with our GBP
denominated expenses. These fixings allow for $250,000 to be converted into GBP at a rate of $1.359 subject to the spot rate on the fixing
date being above the fixed rate. Should the spot rate fall below $1.359 on the scheduled fixing date but above a rate of $1.319, the
Company can exchange the fixing amount at the spot rate on the day; should the spot rate fall below $1.319 the Company is obligated to
convert $500,000 to GBP at the fixed rate of $1.359. The exchange rate range experienced by the Company over the last two years for USD:
GBP has seen a high of approximately $1.216 in May 2020 and a low of approximately $1.423 in June 2021. Cumulative profit on the sale
of USD is capped at an aggregate of approximately $55,000 over the shorter of the life of the contract fixings or the utilization of
the cap.
At March 31, 2022, the Company held a forward contract to sell
up to $12.5 million, which when remeasured at fair value generated a non-cash item loss of $440,196 and has been accounted for within
the foreign exchange translation adjustments line within general and administrative expenses and is held on the Company’s balance
sheet within other liabilities and accrued expenses. No such similar derivative financial instruments were in place at the fiscal year
ended March 31, 2021.
The Company’s foreign currency forward contracts are
measured at fair value on a recurring basis and are classified as Level 2 under our fair value of financial instruments policy.
Retirement benefit plan
The Company operates a retirement plan which
covers most of our regular employees in the UK and allows them to make contributions. The Company also provides a matching contribution
on a portion of the employee contributions. Total expenses incurred under this plan for the fiscal years ended March 31, 2022 and 2021,
were approximately $24,300 and $12,100, respectively. The increase in the fiscal year ended March 31, 2022 was driven by the increase
in our employee numbers.
NEMAURA MEDICAL INC.
Notes to Consolidated Financial Statements
Stock-based
compensation
The
Company accounts for stock-based payments in accordance with stock-based payment accounting guidance which requires all stock-based payments
to be recognized based upon their fair values. The fair value of stock-based awards is estimated at the grant date using the Black-Scholes
Option Pricing Model and the portion that is ultimately expected to vest is recognized as compensation cost over the requisite service
period. The determination of fair value using the Black-Scholes Option Pricing Model is affected by the Company’s stock price as
well as assumptions regarding a number of complex and subjective variables, including expected stock price volatility, risk-free interest
rate, expected dividends and projected employee stock option exercise behaviors. The Company accounts for forfeitures of unvested awards
as they occur.
The Company accounts for share-based payments to non-employees
by aligning it with the accounting for share-based payments to employees, with certain exceptions.
Direct costs incurred for equity
financing
The Company includes all direct costs incurred in connection
with successful equity financings as a component of additional paid-in capital. Direct costs incurred for equity financings that are
unsuccessful are expensed.
Risks and Uncertainties
The Company is in the commercialization stage for its primary
product, sugarBEAT®, following the receipt of the CE mark covering the EU, with the intention of entering into sales and marketing
agreements for the product with prioritization having been initially set for the UK and Germany.
Aside from the UK and Germany, the Company considers the U.S.A.
to be a primary market for its product offerings, and while uncertainties exist with regards to regulatory acceptance of the Company’s
primary product, an FDA PMA application has been submitted and is currently being reviewed; some delays have been experienced as a direct
consequence of COVID-19, whereby the application remained dormant with the FDA for a period of 6 months. In the interim, and further
to discussions with the FDA, the Company has determined that it may sell an adapted version of the CGM device as a wellbeing device,
whereby the Company will gather the data and provide feedback in the form of educational reports providing insights into factors that
may be causing glucose fluctuations and therefore how lifestyle interventions may help improve control of the fluctuations.
The Company has taken steps to support the commercialization
process by increasing raw material inventory over the year in order to support the transition to product manufacturing in relation to
sale to the UK Licensee.
The Company is also in the process of establishing options
to broaden the existing internal manufacturing capabilities with the expectation that it will leverage the manufacturing capacity and
capabilities of one or more contract manufacturers as volume increases.
Recent accounting pronouncements
The Company continually assesses any new accounting pronouncements
to determine their applicability. When it is determined that a new accounting pronouncement affects the Company's financial reporting,
the Company undertakes a study to determine the consequences of the change to its consolidated financial statements and assures that
there are proper controls in place to ascertain that the Company's consolidated financial statements properly reflect the change.
NOTE 4 – LICENSING AGREEMENTS
United Kingdom and the Republic of Ireland, the Channel
Islands and the Isle of Man
In March 2014, the Company entered into an Exclusive Marketing
Rights Agreement with an unrelated third party that granted to the third party the exclusive right to market and promote the sugarBEAT®
device and related patches under its own brand in the United Kingdom and the Republic of Ireland, the Channel Islands and the Isle of
Man. The Company received a non-refundable, up-front cash payment of GBP 1,000,000 (approximately $1.32 million and $1.38 million as
of March 31, 2022 and 2021, respectively), which was wholly non-refundable, upon signing the agreement.
As the Company has continuing performance
obligations under the agreement, the up-front fees received from this agreement have been deferred with the expectation that this deferred
revenue would be treated income over the term of the commercial licensing agreement beginning from the date of clinical evaluation approval.
The Company received this confirmation during the current fiscal year, along with an initial order against which deliveries commenced
in December 2021. At March 31, 2022, approximately $107,000 of this deferred revenue has been treated as a current liability within the
$259,256 deferred revenue balance held; the remainder being shown as a non-current liability balance sheet item.
NEMAURA MEDICAL INC.
Notes to Consolidated Financial Statements
NOTE 5 – PROPERTY AND EQUIPMENT
As of March 31, 2022 and 2021, property and equipment is summarized
as follows:
| PROPERTY AND EQUIPMENT | |
| | | |
| | |
| | |
March 31, | |
| | |
2022
($) | | |
2021
($) | |
| Property and equipment | |
| 806,117 | | |
| 346,500 | |
| Less accumulated depreciation | |
| (273,609 | ) | |
| (144,335 | ) |
| | |
| 532,508 | | |
| 202,145 | |
Depreciation expensed within the consolidated statements of
operations and comprehensive loss relating to property and equipment for the years ended March 31, 2022 and 2021 was approximately $138,000
and $69,000, respectively.
NOTE 6 - INTANGIBLE ASSETS
The following table summarises our intangible assets and capitalized
software development costs at March 31, 2022 and 2021:
| Schedule of Intangible Assets | |
| | | |
| | |
| | |
March 31, | |
| | |
2022
($) | | |
2021 ($) | |
| Patents and licenses | |
| 1,084,081 | | |
| 516,935 | |
| Less accumulated amortization | |
| (186,927 | ) | |
| (125,437 | ) |
| | |
| 897,154 | | |
| 391,498 | |
| | |
| | | |
| | |
| Software development costs | |
| 583,826 | | |
| 663,758 | |
| | |
| | | |
| | |
| | |
| 1,480,980 | | |
| 1,055,256 | |
Amortization expensed within the consolidated statements of operations
and comprehensive loss relating to intangible assets for the years ended March 31, 2022 and 2021 was approximately $92,000 and $29,000,
respectively.
Assuming a constant currency, the following table represents
the estimated amortization for intangible assets relating to patents and licenses for the years ending March 31; no amortization has been
estimated for software development as this is considered to be work-in-progress and the final costs are yet to be determined:
| Schedule of amortization expenses | | |
| |
| | | |
($) | |
| | 2023 | | |
| 174,964 | |
| | 2024 | | |
| 173,910 | |
| | 2025 | | |
| 173,857 | |
| | 2026 | | |
| 149,783 | |
| | 2027 | | |
| 98,307 | |
| | Thereafter | | |
| 126,333 | |
| | Total future net intangible amortization expense | | |
| 897,154 | |
NOTE 7 – PREPAID EXPENSES
| Schedule of prepaid expenses and other receivables | |
| | | |
| | |
| | |
March 31, | |
| | |
2022
($) | | |
2021
($) | |
| Prepaid expenses | |
| 473,799 | | |
| 592,695 | |
| Prepaid inventory | |
| — | | |
| 587,493 | |
| Other taxes | |
| 276,368 | | |
| 89,325 | |
| |
| 750,167 | | |
| 1,269,513 | |
NEMAURA MEDICAL INC.
Notes to Consolidated Financial Statements
NOTE 8 – NOTES PAYABLE
NOTE PURCHASE AGREEMENT 1
On April 15, 2020, the Company entered into a note purchase
agreement (the “Note Purchase Agreement 1”) by and among the Company, DDL, TCL and a third-party investor (the “Investor”).
Pursuant to the terms of the Note Purchase Agreement, the Company
agreed to issue and sell to the Investor and the Investor agreed to purchase from the Company a secured promissory note (the “Secured
Note”) in the original principal amount of $6,015,000. In consideration thereof, on April 15, 2020 (the closing date), (i) the
Investor (a) paid $1,000,000 in cash, (b) issued to the Company (1) Investor Note #1 in the principal amount of $2,000,000 (“Investor
Note #1”), and (2) Investor Note #2 in the principal amount of $2,000,000 (“Investor Note #2” and together with Investor
Note #1, the “Investor Notes”), and (ii) the Company delivered the Secured Note on behalf of the Company, to the Investor,
against delivery of the Purchase Price. For these purposes, the “Purchase Price” means the Investor’s initial cash
purchase price, together with the sum of the initial principal amounts of the Investor Notes.
The Secured Note is secured by the Collateral (as hereinafter
defined). The Secured Note carries an original issue discount (“OID”) of $1,000,000 (16.7%). In addition, the Company agreed
to pay $15,000 to the Investor to cover the Investor’s legal fees, accounting costs, due diligence, monitoring and other transaction
costs incurred in connection with the purchase and sale of the Secured Note (the “Transaction Expense Amount”). In addition
to this, a payment of $325,000 was made to Ascendiant Capital Markets, LLC, (the “Commission”) for structuring the agreement
between both parties. The Purchase Price for the Secured Note is $4,675,000, computed as follows: $6,015,000 original principal balance,
less: OID, Transaction Expense Amount, and commission paid.
The borrowing period is 24 months, and the Company shall pay
the outstanding balance and all fees on maturity. A monitoring fee equal to 0.833% of the outstanding balance will automatically be added
to the outstanding balance on the first day of each month. The debt less the discount and transaction expenses will be accreted over
the term of the Note using the effective interest method.
Security Agreement
On April 15, 2020, the Company entered into the Security Agreement
by the Company, DDL and TCL, in favor of the Investor (the “Security Agreement”). Pursuant to the terms of the Security Agreement,
the Company granted the Investor a first-priority security interest in all rights, title, interest, claims and demands of the Company
in and to all of the Company’s patents and all other proprietary rights, and all rights corresponding to the Company’s patents
throughout the world, now owned and existing, and all replacements, proceeds, products, and accessions thereof.
NOTE PURCHASE AGREEMENT 2
On February 8, 2021, the Company
entered into an additional note purchase agreement (“Note Purchase Agreement 2”) with the Investor. Pursuant to the
terms of Note Purchase Agreement 2, the Company agreed to issue and sell to the Investor and the Investor agreed to purchase from the
Company, a secured promissory note (“Secured Note 2”) in the original principal amount of $24,015,000. The Secured Note carries
an OID of $4,000,000 (16.7%), and the Company agreed to pay $15,000 to the Investor to cover the Investor’s transaction expenses.
In addition to this, a Commission of $1,200,000 was also payable to Ascendiant Capital Partners, LLC.
In consideration thereof, on
February 9, 2021 (the “closing date”), (i) the Investor paid $20,000,000 in cash to the Company, and (ii) the Company delivered
Secured Note 2 on behalf of the Company, to the Investor, against the delivery of the Purchase Price. For these purposes, the “Purchase
Price” means the Investor’s initial cash purchase price. After adjusting for transaction expenses of $1,200,000, cash
proceeds received were $18,800,000.
The borrowing terms for Note
Purchase Agreement 2 are consistent with those of Note Purchase Agreement 1, with the borrowing period being 24 months from the date of
the agreement, the Company being required to pay the outstanding balance and all fees on maturity, and a monitoring fee equal to 0.833%
of the outstanding balance being automatically added to the outstanding balance on the first day of each month. The debt less discount
and transaction expenses will be accreted over the term of the Note using the effective interest rate method.
NEMAURA MEDICAL INC.
Notes to Consolidated Financial Statements
Security Agreement
On February 8, 2021, the Security Agreement established in
respect to Note Purchase Agreement 1 was extended to include Note Purchase Agreement 2, which is also secured against all of the Company’s
assets owned as of the closing date and extends to any assets acquired at any time that the Company’s obligations under Secured
Note 2 are outstanding.
As of March 31, 2022, all outstanding debt in relation to the
Note Purchase Agreements is due for repayment within the next 12 months.
NOTE 9 – RELATED PARTY TRANSACTIONS
Nemaura Pharma Limited (“Pharma”), Black and White
Health Care Limited (“B&W”) and NDM Technologies Limited (“NDM”) are entities controlled by the Company’s
chief executive officer and majority shareholder, D.F.H. Chowdhury.
Pharma has a service agreement with DDL, to undertake development,
manufacture, and regulatory approvals under Pharma’s ISO13485 Accreditation. In lieu of these services, Pharma invoices DDL on
a periodic basis for said services. Services are provided at cost plus a service surcharge amounting to less than 10% of the total costs
incurred.
The following is a summary of activity between the Company
and Pharma, B&W and NDM for the years ended March 31, 2022 and 2021:
| Schedule of Related Party Transactions | |
| | | |
| | |
| | |
March 31, | |
| | |
2022
($) | | |
2021
($) | |
| Liability due to related parties at beginning of year | |
| 148,795 | | |
| 830,093 | |
| Amounts invoiced by Pharma to DDL, NM and TCL (1) | |
| 3,245,985 | | |
| 2,441,108 | |
| Amounts invoiced by DDL to Pharma | |
| (2,495 | ) | |
| (17,213 | ) |
| Amounts repaid by DDL to Pharma | |
| (3,492,962 | ) | |
| (3,209,084 | ) |
| Foreign exchange differences | |
| (620 | ) | |
| 103,891 | |
| (Receivable)/Liability due (from) to related parties at end
of year | |
| (101,297 | ) | |
| 148,795 | |
| (1) |
These invoiced amounts primarily relate to research and development expenses. |
All related party transactions relate to operating activities
in the years ended March 31, 2022 and 2021.
NOTE 10 – INCOME TAXES
The Company and its subsidiaries file separate income tax returns.
United States of America
The Company is incorporated in the U.S. and is subject to a
U.S. federal corporate income tax rate of 21% for the years ended March 31, 2022 and 2021.
British Virgin Islands
RGL was incorporated in the British Virgin Islands (“BVI”).
Under the current laws of the BVI, RGL was not subject to tax on income or capital gains. In addition, upon payments of dividends by
RGL, no BVI withholding tax was imposed. During the years ended March 31, 2022 and 2021, there were no income or expenses in the BVI;
RGL was formally dissolved as of April 23, 2021.
UK
DDL, TCL and DDHL are all incorporated in
the UK and the applicable UK statutory income tax rate for these companies is 19%.
NEMAURA MEDICAL INC.
Notes to Consolidated Financial Statements
For the fiscal years ended March 31, 2022 and 2021 loss before
income tax benefit arose in the UK and U.S. as follows:
| Schedule of loss before Income Tax, Domestic and Foreign | |
| | | |
| | |
| | |
March 31, | |
| | |
2022 | | |
2021 | |
| | |
| $ | | |
| $ | |
| Loss before income taxes arising in UK | |
| (11,716,916 | ) | |
| (5,030,204 | ) |
| Loss before income taxes arising in U.S. | |
| (2,520,145 | ) | |
| (1,564,224 | ) |
| Total loss before income tax benefit | |
| (14,237,061 | ) | |
| (6,594,428 | ) |
Reconciliation of our effective tax rate to the loss calculated
at the statutory U.S. federal tax rate is as follows:
| Reconciliation of effective tax rate | |
| | | |
| | | |
| | | |
| | |
| | |
March 31, | |
| | |
2022 | | |
2021 | |
| | |
| $ | | |
| | | |
| $ | | |
| | |
| Loss before income taxes | |
| ) | |
| | | |
| ) | |
| | |
| Expected tax benefit | |
| (2,989,783 | ) | |
| (21%) | | |
| (1,384,830 | ) | |
| (21%) | |
| Foreign tax differential | |
| 234,338 | | |
| 2% | | |
| 100,604 | | |
| 2% | |
| Enhanced research and development | |
| (463,591 | ) | |
| (3%) | | |
| (259,861 | ) | |
| (4%) | |
| Prior year true-up of NOL’s | |
| 2,401,930 | | |
| 17% | | |
| — | | |
| — | |
| Other | |
| 74,579 | | |
| 1% | | |
| 20,226 | | |
| — | |
| Change in valuation allowance | |
| 742,527 | | |
| 5% | | |
| 1,523,861 | | |
| 23% | |
| R&D credit received | |
| 350,256 | | |
| 2% | | |
| 335,832 | | |
| 5% | |
| Actual income tax benefit | |
| 350,256 | | |
| 2% | | |
| 335,832 | | |
| 5% | |
| | |
| | | |
| | | |
| | | |
| | |
The tax effects of the temporary differences
that give rise to significant portions of deferred income tax assets are presented below:
| Schedule of deferred income tax assets | |
| | | |
| | |
| | |
March 31, | |
| | |
2022 | | |
2021 | |
| | |
| $ | | |
| $ | |
| Net operating tax loss carried forward | |
| 6,671,000 | | |
| 5,204,000 | |
| Research and development enhancement | |
| 335,000 | | |
| 1,057,000 | |
| Other items | |
| (335,000 | ) | |
| (333,000 | ) |
| Valuation allowance | |
| (6,671,000 | ) | |
| (5,928,000 | ) |
| Net deferred tax assets | |
| — | | |
| — | |
In the fiscal year ended March 31, 2022, the Company received
$350,256 from HMRC (Her Majesty’s Revenue and Customs) in tax credits relating to the reimbursement of research and development
expenses incurred during the fiscal year ended March 31, 2021. For the fiscal year ended March 31, 2021, the research and development
tax credit received was $335,832, relating to expenses incurred for the fiscal year ended March 31, 2020. These amounts are reflected
as a credit provision for income taxes in the Company’s consolidated statements of operations and comprehensive loss in the respective
years received.
For each of the fiscal years ended March 31, 2022 and 2021, the Company did not have unrecognized tax benefits, and therefore no interest
or penalties related to unrecognized tax benefits were accrued. Management does not expect that the amount of unrecognized tax benefits
will change significantly within the next twelve months.
The Company mainly files income
tax returns in the U.S. and the UK. The Company is subject to U.S. federal income tax examination by tax authorities for tax years beginning
in 2017. The UK tax returns for the Company’s UK subsidiaries are open to examination by the UK tax authorities for the tax
years beginning April 1, 2016.
As of March 31, 2022, the Company
has net operating losses (“NOLs”) of approximately $8,351,000 in the U.S. and $25,879,000 in the UK. NOLs may be carried forward
indefinitely. Additionally, the Company has a research and development enhancement deduction carry forward of approximately $1,762,000
for purposes of UK income tax filings.
NEMAURA MEDICAL INC.
Notes to Consolidated Financial Statements
NOTE 11 – STOCKHOLDERS’
EQUITY
Shelf Registration Statement
The Company filed a shelf registration
statement on Form S-3 with the SEC, which was declared effective by the SEC on March 24, 2022 (the “2022 Shelf Registration Statement”).
The 2022 Shelf Registration Statement provides the Company with the ability to issue common stock and other securities as described in
the registration statement from time to time up to an aggregate amount of $224,634,031, dependent upon available shares.
Other equity transactions
On July 23, 2021, the Company entered into an At The Market
Offering Agreement (the “2021 ATM”) with H.C. Wainwright & Co., LLC (the “Agent”) pursuant to which
the Company may offer and sell from time to time to, at its option, up to an aggregate of $100 million of shares of its common stock.
Shares sold under the 2021 ATM are issued pursuant to the Company’s 2019 Shelf Registration Statement and a prospectus supplement
dated July 23, 2021.
The Company is required to pay the Agent a commission of 3%
of the gross proceeds from the sale of shares and has also agreed to provide the Agent with customary indemnification rights. During
the year ended March 31, 2022, the Company issued and sold 397,524 shares
of its common stock at an average price of $4.07 per
share under the 2021 ATM for aggregate net proceeds of $1.6 million
after deducting commissions and offering expenses payable by the Company.
During the fourth quarter of the fiscal year ended March 31,
2022, the Company was approached by Tiger Management L.L.C. (a vehicle for the family office of Julian H. Robertson) with a view to acquiring
a direct stake in the Company. The Company agreed to sell 750,000 shares to Tiger Trading Partners L.L.C. (an affiliate undertaking)
at a price of $4 per share and gross proceeds of $3 million; 50% of the shares being sold within the 2021 ATM facility noted above, and
the remainder as a direct issuance which completed on February 10, 2022.
The Company commenced an offering of up to $20,000,000 worth
of shares of its common stock on October 19, 2018. For the fiscal year ended March 31, 2021, a total of 408,718 shares were issued pursuant
to the offering, generating gross proceeds of $4,250,676 and costs of $127,520. For the fiscal year ended March 31, 2022, no shares were
issued pursuant to the offering as the offering agreement had been terminated as of August 18, 2020.
On December 18, 2018, the Company issued a unit purchase option,
to purchase 9,710 shares and 9,710 warrants, to Dawson James Securities, Inc. The Company has classified this option as equity. The unit
purchase option has an exercise price of $13.00 per unit and can be exercised for a period of three years from 180 days following the
date that the registration became effective.
On December 20, 2018, the Company sold 194,206 units, with
each unit consisting of one share of common stock and one 5-year warrant to purchase one share of common stock at an exercise price of
$10.40 per share, at a purchase price of $10.40 per unit, for gross proceeds of $2,019,743 and net proceeds to the Company of $1,691,541,
after deducting $328,302 of placement agent commissions and other offering expenses. As of March 31, 2022, 58,569 of the warrants had
been exercised, generating $609,118 of additional funds. At March 31, 2022, there were 135,753 warrants outstanding.
On July 30, 2020, the Company sold 1,586,206 shares of the
Company’s common stock and warrants to purchase up to 793,103 shares of common stock. Each share of common stock and accompanying
one-half of a warrant were sold for a combined purchase price of $7.25, for gross proceeds of $11.5 million and net proceeds to the Company
of $10.7 million, after deducting placement agent fees and offering expenses. Each whole warrant is immediately exercisable at a price
of $8.00 per share, subject to adjustment in certain circumstances, and will expire five years from the date of issuance. As of March
31, 2022, 58,569 of the warrants had been exercised, generating $2,846,064 of additional funds. At March 31, 2022, there were 437,345
warrants outstanding.
NEMAURA MEDICAL INC.
Notes to Consolidated Financial Statements
Stock options
On January 28, 2022, the Board of Directors
granted to each of the directors, an option to purchase 8,000 shares of common stock at an exercise price of $3.98 per share, being the
closing price of the Company’s common stock on the date of grant. The option was fully vested at grant and is exercisable for a
period of five years from the date of grant.
The following table provides a summary
of the Options Award activity is presented below:
| Schedule of stock options | |
| | | |
| | | |
| | |
| | |
Number of Options | |
Weighted Average Exercise Price
$ | |
Weight Average remaining Contractual Term (years) |
| Balance at April 1, 2021 | |
| — | | |
| — | | |
| | |
| Granted | |
| 40,000 | | |
| 3.98 | | |
| | |
| Exercised | |
| — | | |
| — | | |
| | |
| Forfeited | |
| — | | |
| — | | |
| | |
| Expired | |
| — | | |
| — | | |
| | |
| Balance at March 31, 2022 | |
| 40,000 | | |
| 3.98 | | |
| 4.83 | |
| Vested and exercisable at March 31, 2022 | |
| 40,000 | | |
| 3.98 | | |
| 4.83 | |
The fair value of stock options granted
during the fiscal year ended March 31, 2022 was determined using a Black-Scholes Option Pricing Model (there were no options granted
as at April 1, 2021). The key assumptions for which have been set-out below:
| Schedule of assumptions for stock options | |
| | |
| Stock Price | |
$ | 3.98 | |
| Exercise Price | |
$ | 3.98 | |
| Term | |
| 5 years | |
| Volatility | |
| 122.52 | % |
| Expected dividend yield (%) | |
| — | |
| Discount Rate (Bond Equivalent Yield) | |
| 2.28 | % |
NOTE 12 – OTHER ITEMS
(a)
COVID-19 Pandemic
The outbreak of COVID-19 originating
in Wuhan, China, in December 2019 has since rapidly increased its exposure globally. On March 11, 2020, the World Health Organization
declared the outbreak a pandemic. We continue to monitor the global outbreak of COVID-19 and are working with our employees, suppliers
and other stakeholders to mitigate the risks posed by its spread, COVID-19 is not expected to have any long-term detrimental effect on
the Company’s success. While key suppliers have not been accessible throughout the whole period of the outbreak, we have, to date,
been able to be flexible in our priorities and respond favorably to the challenges faced during
the outbreak. We have also seen a surge in the uptake of technologies for remote and patient self-monitoring, which therefore potentially
enhances the prospects for the likes of the Company and its CGM product and planned digital healthcare offering.
NEMAURA MEDICAL INC.
Notes to Consolidated Financial Statements
Whilst restrictions associated with
COVID-19 have largely been removed in our operational locations, we will continue to assess the situation, including abiding by any government-imposed
restrictions, as and where relevant.
(b)
Investor relations agreements
The Company has entered into contracts with several investor
relations specialists to help support the ongoing financing activities of the business.
During the fiscal year ended March 31, 2022, the Company extended
the contractual agreement that it had entered into in the year ended March 31, 2021, into a rolling monthly agreement, compensation for
which was settled in cash. Stock-based compensation of $50,000 was expensed during the current year end, all of which related to the
previous agreement terms.
During the fiscal year ended March 31, 2021, the Company entered
into a contractual agreement with a new investor relations company, the term of which was set at 12 months with the related compensation
being paid via a mixture of cash and common stock. Total stock-based compensation expense for the year ended March 31, 2021, in relation
to this was $50,000. In addition to this, $59,000 was paid by way of stock-based compensation to two additional investor relations companies,
whose services were terminated during the year.
NOTE 13 – SUBSEQUENT EVENTS
At The Market Offering
The At The Market Offering Agreement, or the sales agreement,
that was entered into with H.C. Wainwright & Co., LLC, or the sales agent or Wainwright, dated as of July 23, 2021 was amended as
of April 1, 2022, relating to the offer and sale of shares of our common stock. In accordance with the terms of the sales agreement,
we may offer and sell up to a maximum aggregate amount of $3,000,000 (as opposed to $100,000,000) shares of our common stock from time
to time through the sales agent.
Note Purchase Agreement
On May 20, 2022, the Company entered into a new note purchase
agreement (“Note Purchase Agreement 3”) by and among the Company, DDL, TCL and a third-party investor.
Pursuant to the terms of the Note Purchase Agreement 3, the
Company agreed to issue and sell to the Investor and the Investor agreed to purchase from the Company a secured promissory note (the
“Secured Note”) in the original principal amount of $6,015,000. In consideration thereof, on May 20, 2022 (the closing date),
(i) the Investor paid $5,000,000 in cash, and (ii) the Company delivered the Secured Note on behalf of the Company, to the Investor,
against delivery of the Purchase Price. For these purposes, the “Purchase Price” means the Investor’s initial cash
purchase price.
The Secured Note is secured by the Collateral (as hereinafter
defined). The Secured Note carries an original issue discount (“OID”) of $1,000,000 (16.7%). In addition, the Company agreed
to pay $15,000 to the Investor to cover the Investor’s legal fees, accounting costs, due diligence, monitoring and other transaction
costs incurred in connection with the purchase and sale of the Secured Note (the “Transaction Expense Amount”). In addition
to this, a payment of $300,000 was made to Ascendiant Capital Markets, LLC, (the “Commission”) for structuring the agreement
between both parties. The Purchase Price for the Secured Note is $4,700,000, computed as follows: $6,015,000 original principal balance,
less: OID, Transaction Expense Amount, and commission paid.
The borrowing period is 24 months, and the Company shall pay
the outstanding balance and all fees on maturity. A monitoring fee equal to 0.833% of the outstanding balance will automatically be added
to the outstanding balance on the first day of each month. The debt less the discount and transaction expenses will be accreted over
the term of the Note using the effective interest method.
Security Agreement
On May 20, 2022, the Company entered into
the Security Agreement by the Company, DDL and TCL, in favor of the Investor (the “Security Agreement”). Pursuant to the terms
of the Security Agreement, the Company granted the Investor a first-priority security interest in all rights, title, interest, claims
and demands of the Company in and to all of the Company’s patents and all other proprietary rights, and all rights corresponding
to the Company’s patents throughout the world, now owned and existing, and all replacements, proceeds, products, and accessions
thereof.
| NEMAURA MEDICAL INC. |
| Condensed Consolidated Balance Sheets |
| | |
| | | |
| | |
| | |
| | |
| |
| | |
December 31, 2022 (Unaudited) | | |
March 31, 2022 | |
| | |
| | |
| |
| ASSETS | |
| | | |
| | |
| Current assets: | |
| | | |
| | |
| Cash | |
$ | 7,340,840 | | |
$ | 17,749,233 | |
| Prepaid expenses and other receivables | |
| 1,217,237 | | |
| 750,167 | |
| Accounts receivable - related party | |
| 25,320 | | |
| 101,297 | |
| Inventory | |
| 2,352,407 | | |
| 1,487,771 | |
| Total current assets | |
| 10,935,804 | | |
| 20,088,468 | |
| | |
| | | |
| | |
| | |
| | | |
| | |
| Property and equipment, net of accumulated depreciation | |
| 581,903 | | |
| 532,508 | |
| Intangible assets, net of accumulated amortization | |
| 1,443,991 | | |
| 1,480,980 | |
| Total other assets | |
| 2,025,894 | | |
| 2,013,488 | |
| Total assets | |
$ | 12,961,698 | | |
$ | 22,101,956 | |
| | |
| | | |
| | |
| LIABILITIES AND STOCKHOLDERS’ (DEFICIT) EQUITY | |
| | | |
| | |
| Current liabilities: | |
| | | |
| | |
| Accounts payable | |
$ | 171,207 | | |
$ | 136,310 | |
| Other liabilities and accrued expenses | |
| 390,858 | | |
| 558,426 | |
| Foreign currency contract | |
| 1,075,692 | | |
| 440,196 | |
| Notes payable, current portion | |
| 11,512,711 | | |
| 19,188,724 | |
| Deferred revenue | |
| 69,681 | | |
| 259,256 | |
| Total current liabilities | |
| 13,220,149 | | |
| 20,582,912 | |
| | |
| | | |
| | |
| Notes payable, net of current portion | |
| 8,557,548 | | |
| — | |
| Deferred revenue, net of current portion | |
| 1,042,710 | | |
| 1,052,960 | |
| Total liabilities | |
| 22,820,407 | | |
| 21,635,872 | |
| | |
| | | |
| | |
| Commitments and contingencies | |
| | | |
| | |
| | |
| | | |
| | |
| Stockholders’ (deficit) equity: | |
| | | |
| | |
| |
| | | |
| | |
| Common stock, $0.001 par value, 42,000,000 shares authorized and 24,103,196 and 24,102,866 shares
issued and outstanding at December 31, 2022 and March 31, 2022 | |
| 24,103 | | |
| 24,103 | |
| Additional paid-in capital | |
| 38,296,198 | | |
| 38,295,775 | |
| Accumulated deficit | |
| (47,192,364 | ) | |
| (37,731,476 | ) |
| Accumulated other comprehensive loss | |
| (986,646 | ) | |
| (122,318 | ) |
| Total stockholders’ (deficit) equity | |
| (9,858,709 | ) | |
| 466,084 | |
| Total liabilities and stockholders’ (deficit) equity | |
$ | 12,961,698 | | |
$ | 22,101,956 | |
See notes to the unaudited condensed consolidated
financial statements.
NEMAURA MEDICAL INC. |
| Condensed Consolidated Statements of Operations and Comprehensive Loss |
|
(Unaudited)
(in Dollars, except Share Amounts) |
| | |
| |
| |
| |
|
| | |
Three Months Ended December 31, | |
Nine Months Ended December 31, |
| | |
2022 | |
2021 | |
2022 | |
2021 |
| | |
| |
| |
| |
|
| Sales | |
$ | 3,017 | | |
$ | 183,628 | | |
$ | 77,044 | | |
$ | 183,628 | |
| Cost of Sales | |
| (2,971 | ) | |
| (172,393 | ) | |
| (75,327 | ) | |
| (172,393 | ) |
| Gross Profit | |
| 46 | | |
| 11,235 | | |
| 1,717 | | |
| 11,235 | |
| | |
| | | |
| | | |
| | | |
| | |
| Operating expenses: | |
| | | |
| | | |
| | | |
| | |
| Research and development | |
| 393,747 | | |
| 412,341 | | |
| 980,862 | | |
| 987,711 | |
| General and administrative | |
| 239,628 | | |
| 1,391,278 | | |
| 4,329,306 | | |
| 4,151,380 | |
| Total operating expenses | |
| 633,375 | | |
| 1,803,619 | | |
| 5,310,168 | | |
| 5,139,091 | |
| | |
| | | |
| | | |
| | | |
| | |
| Loss from operations | |
| (633,329 | ) | |
| (1,792,384 | ) | |
| (5,308,451 | ) | |
| (5,127,856 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Interest expense | |
| (1,082,949 | ) | |
| (1,639,184 | ) | |
| (4,152,437 | ) | |
| (5,141,701 | ) |
| Net loss | |
| (1,716,278 | ) | |
| (3,431,568 | ) | |
| (9,460,888 | ) | |
| (10,269,557 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Other comprehensive loss: | |
| | | |
| | | |
| | | |
| | |
| Foreign currency translation adjustment | |
| 556,080 | | |
| (25,065 | ) | |
| (864,328 | ) | |
| (142,922 | ) |
| Comprehensive loss | |
$ | (1,160,198 | ) | |
$ | (3,456,633 | ) | |
$ | (10,325,216 | ) | |
$ | (10,412,479 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Net loss per share, basic and diluted | |
$ | (0.07 | ) | |
$ | (0.15 | ) | |
$ | (0.39 | ) | |
$ | (0.44 | ) |
| Weighted average number of shares outstanding, basic and diluted | |
| 24,103,196 | | |
| 23,313,629 | | |
| 24,102,976 | | |
| 23,244,345 | |
See notes to the unaudited condensed consolidated
financial statements.
NEMAURA MEDICAL INC.
Condensed Consolidated Statements of Changes
in Stockholders’ (Deficit) Equity
Three Months Ended December 31, 2022 and 2021
(Unaudited)
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| | |
| Common
Stock | | |
| | | |
| | | |
| | | |
| | |
| | |
| Shares | | |
| Amount ($) | | |
| Additional
Paid-in Capital ($) | | |
| Accumulated
Deficit ($) | | |
| Accumulated
Other Comprehensive (Loss) Income ($) | | |
| Total
Stockholders’ Equity (Deficit) ($) | |
| Balance at September 30, 2022 | |
| 24,102,866 | | |
| 24,103 | | |
| 38,295,775 | | |
| (45,476,086 | ) | |
| (1,542,726 | ) | |
| (8,698,934 | ) |
| Shares issued under ATM facility | |
| 330 | | |
| — | | |
| 423 | | |
| — | | |
| — | | |
| 423 | |
| Foreign currency translation adjustment | |
| — | | |
| — | | |
| — | | |
| — | | |
| 556,080 | | |
| 556,080 | |
| Net loss | |
| — | | |
| — | | |
| — | | |
| (1,716,278 | ) | |
| — | | |
| (1,716,278 | ) |
| Balance at December 31, 2022 | |
| 24,103,196 | | |
| 24,103 | | |
| 38,296,198 | | |
| (47,192,364 | ) | |
| (986,646 | ) | |
| (9,858,709 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Balance at September 30, 2021 | |
| 23,308,049 | | |
| 23,308 | | |
| 35,007,626 | | |
| (30,682,660 | ) | |
| 17,710 | | |
| 4,365,984 | |
| Shares issued under ATM facility | |
| 22,524 | | |
| 23 | | |
| 114,386 | | |
| — | | |
| — | | |
| 114,409 | |
| Foreign currency translation adjustment | |
| — | | |
| — | | |
| — | | |
| — | | |
| (25,065 | ) | |
| (25,065 | ) |
| Net loss | |
| — | | |
| — | | |
| — | | |
| (3,431,568 | ) | |
| — | | |
| (3,431,568 | ) |
| Balance at December 31, 2021 | |
| 23,330,573 | | |
| 23,331 | | |
| 35,122,012 | | |
| (34,114,228 | ) | |
| (7,355 | ) | |
| 1,023,760 | |
See notes to the unaudited condensed consolidated
financial statements.
NEMAURA MEDICAL INC.
Condensed Consolidated Statements of Changes
in Stockholders’ Equity (deficit)
Nine Months Ended December 31, 2022 and 2021
(Unaudited)
| |
| | |
| Common
Stock | |
| | | |
| | | |
| | | |
| | |
| | |
| Shares | | |
| Amount ($) | | |
| Additional
Paid-in Capital ($) | | |
| Accumulated
Deficit ($) | | |
| Accumulated
Other Comprehensive (Loss) Income ($) | | |
| Total
Stockholders’ Equity (Deficit) ($) | |
| Balance at March 31, 2022 | |
| 24,102,866 | | |
| 24,103 | | |
| 38,295,775 | | |
| (37,731,476 | ) | |
| (122,318 | ) | |
| 466,084 | |
| Shares issued under ATM facility | |
| 330 | | |
| — | | |
| 423 | | |
| — | | |
| — | | |
| 423 | |
| Foreign currency translation adjustment | |
| — | | |
| — | | |
| — | | |
| — | | |
| (864,328 | ) | |
| (864,328 | ) |
| Net loss | |
| — | | |
| — | | |
| — | | |
| (9,460,888 | ) | |
| — | | |
| (9,460,888 | ) |
| Balance at December 31, 2022 | |
| 24,103,196 | | |
| 24,103 | | |
| 38,296,198 | | |
| (47,192,364 | ) | |
| (986,646 | ) | |
| (9,858,709 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Balance at March 31, 2021 | |
| 22,941,157 | | |
| 22,941 | | |
| 32,044,335 | | |
| (23,844,671 | ) | |
| 135,567 | | |
| 8,358,172 | |
| Shares issued under ATM facility | |
| 22,524 | | |
| 23 | | |
| 114,386 | | |
| — | | |
| — | | |
| 114,409 | |
| Exercise of warrants | |
| 366,892 | | |
| 367 | | |
| 2,963,291 | | |
| — | | |
| — | | |
| 2,963,658 | |
| Foreign currency translation adjustment | |
| — | | |
| — | | |
| — | | |
| — | | |
| (142,922 | ) | |
| (142,922 | ) |
| Net loss | |
| — | | |
| — | | |
| — | | |
| (10,269,557 | ) | |
| — | | |
| (10,269,557 | ) |
| Balance at December 31, 2021 | |
| 23,330,573 | | |
| 23,331 | | |
| 35,122,012 | | |
| (34,114,228 | ) | |
| (7,355 | ) | |
| 1,023,760 | |
|
See notes to the unaudited condensed consolidated financial
statements
|
| |
| NEMAURA MEDICAL INC. |
| Condensed Consolidated Statements of Cash Flows |
| (Unaudited) |
| | |
| | |
| |
| | |
Nine Months Ended
December 31, | |
| | |
2022 | | |
2021 | |
| | |
| | |
| |
| Cash Flows From Operating Activities: | |
| | | |
| | |
| Net loss | |
$ | (9,460,888 | ) | |
$ | (10,269,557 | ) |
| | |
| | | |
| | |
| Adjustments to reconcile net loss to net cash used in operating activities: | |
| | | |
| | |
| Depreciation and amortization | |
| 268,595 | | |
| 139,751 | |
| Amortization of debt discount | |
| 4,152,437 | | |
| 5,141,701 | |
| Change in fair value of foreign currency contract | |
| 635,494 | | |
| 199,522 | |
| Changes in assets and liabilities: | |
| | | |
| | |
| Prepaid expenses and other receivables | |
| (467,070 | ) | |
| 797,155 | |
| Inventory | |
| (864,636 | ) | |
| (533,656 | ) |
| Accounts payable | |
| 34,897 | | |
| (77,075 | ) |
| Due to (from) related parties | |
| 75,977 | | |
| (301,387 | ) |
| Other liabilities and accrued expenses | |
| (167,568 | ) | |
| 264,786 | |
| Deferred revenue | |
| (297,419 | ) | |
| 285,266 | |
| Net cash used in operating activities | |
| (6,090,181 | ) | |
| (4,353,494 | ) |
| | |
| | | |
| | |
| Cash Flows From Investing Activities: | |
| | | |
| | |
| Capitalized patent costs | |
| (135,168 | ) | |
| (60,241 | ) |
| Capitalized software development costs | |
| (27,879 | ) | |
| (460,466 | ) |
| Purchase of property and equipment | |
| (275,758 | ) | |
| (359,301 | ) |
| Net cash used in investing activities | |
| (438,805 | ) | |
| (880,008 | ) |
| | |
| | | |
| | |
| Cash Flows From Financing Activities: | |
| | | |
| | |
| Proceeds from issuance of common stock | |
| 696 | | |
| 118,791 | |
| Equity issuance cost paid | |
| (273 | ) | |
| (4,382 | ) |
| Proceeds from issuance of notes payable | |
| 4,700,000 | | |
| — | |
| Proceeds from warrant exercise | |
| — | | |
| 2,963,658 | |
| Repayments of note payable | |
| (7,974,282 | ) | |
| (6,500,000 | ) |
| Net cash used in financing activities | |
| (3,273,859 | ) | |
| (3,421,933 | ) |
| | |
| | | |
| | |
| Effect of exchange rate changes on cash | |
| (605,548 | ) | |
| (163,658 | ) |
| | |
| | | |
| | |
| Net decrease in cash | |
| (10,408,393 | ) | |
| (8,819,093 | ) |
| | |
| | | |
| | |
| Cash at beginning of period | |
| 17,749,233 | | |
| 31,865,371 | |
| Cash at end of period | |
| 7,340,840 | | |
| 23,046,278 | |
| | |
| | | |
| | |
| Supplemental disclosure of non-cash financing activities: | |
| | | |
| | |
| Release of prepayment from equity compensation | |
| — | | |
| 50,000 | |
| Monitoring fees related to notes payable | |
| 1,522,372 | | |
| — | |
See notes to the unaudited condensed consolidated
financial statements.
NEMAURA MEDICAL INC.
Notes to Condensed Consolidated Financial Statements
(Unaudited)
NOTE 1 – ORGANIZATION AND PRINCIPAL ACTIVITIES
Nemaura Medical Inc. (“Nemaura”
or the “Company”), through its operating subsidiaries, performs medical device research and manufacturing of a continuous
glucose monitoring system (“CGM”), named sugarBEAT®. The sugarBEAT®
device is a non-invasive, wireless device for use by persons with Type I and Type II diabetes and may also be used to screen pre-diabetic
patients. The sugarBEAT® device extracts analytes, such as glucose, to the surface of
the skin in a non-invasive manner where it is measured using unique sensors and interpreted using a unique algorithm.
Nemaura is a Nevada holding company organized
in 2013. Nemaura owns 100% of the stock in Dermal Diagnostic (Holdings) Limited, an England and Wales corporation (“DDHL”)
formed on December 11, 2013, which in turn owns 100% of Dermal Diagnostics Limited, an England and Wales corporation formed on January
20, 2009 (“DDL”), and 100% of Trial Clinic Limited, an England and Wales corporation formed on January 12, 2011 (“TCL”).
DDL is a diagnostic medical device company
headquartered in Loughborough, Leicestershire, England, and is engaged in the discovery, development, and commercialization of diagnostic
medical devices. The Company’s initial focus has been on the development of the sugarBEAT®
device, which consists of a disposable patch containing a sensor, and a non-disposable miniature wireless transmitter with a re-chargeable
power source, which is designed to enable trending or tracking of blood glucose levels. All of the Company’s operations and assets
are located in England.
During the fiscal year ended March 31,
2021, the Board of Directors assessed the adequacy of the group’s organizational structure and concluded that the intermediate holding
company that sat below Nemaura Medical Inc., Region Green Limited (a British Virgin Islands corporation), was no longer required as the
entity had been effectively dormant since inception and no longer represented a requirement to be maintained. It was therefore determined
that Region Green Limited should be unwound, with the intention that the assets held by Region Green Limited be transferred up to Nemaura
Medical Inc. following which Region Green Limited would be dissolved.
The transfer of assets took place on March 5, 2021 and Region Green Limited
was formally dissolved as of April 23, 2021.
The following diagram illustrates Nemaura’s
corporate structure as of December 31, 2022:

The Company was incorporated in 2013 and has reported
recurring losses from operations to date and an accumulated deficit of $47,192,364 as of December 31, 2022. These
operations have resulted in the successful completion of clinical programs to support a CE mark (European Union (“EU”) approval
of the product) approval, as well as a De Novo 510(k) medical device application to the U.S. Food and Drug Administration (“FDA”)
submission.
The Company
expects to continue to incur losses from operations until revenues are generated through licensing fees or product sales. However, given
the completion of the requisite clinical programs, these losses are expected to decrease over time. Management has entered into licensing,
supply, or collaboration agreements with unrelated third parties relating to the United Kingdom (“UK”), Europe, Qatar, and
all countries in the Gulf Cooperation Council.
Going Concern
As identified under Item 1A, included in the
Company’s Annual Report on Form 10-K for the year ended March 31, 2022, as filed with the SEC, management is aware of the need to
raise additional funds in order to finance the ongoing commercialization of sugarBEAT®. The Company had $7,340,840 of cash at
December 31, 2022. The Company has debt on its balance sheet which will reach maturity in July 2024.
In evaluating the going concern position of the
Company, management has considered the ability of the Company to raise additional funding in combination with one or more of the different
funding options available to it at this time. Based on current and ongoing engagement with potential funding providers, management
believes that there is a reasonable expectation that funding could be provided by one, or more, of the following options:
Equity funding – the Company
has immediate access to funds through the ATM facility that is currently in place; in addition to this, there are various alternative
mechanisms available to the Company similar to those used previously e.g. direct sale of shares to interested third parties, as well as
other mechanisms to sell common stock via an underwritten agreement or the further exercise of warrants by the current warrant holders
etc. The Company completed a Registered Direct Offering and concurrent Private Placement in January 2023 which has increased cash by $7,655,974.
Debt funding – the Company continues
to be in ongoing discussions with third party debt providers, including the incumbent, to enable the existing debt facility to be restructured
or renewed, should management feel that this route offers a more attractive option compared to the sale of equity that is dependent on
the current market conditions.
Alternative funding as used in the
past such as the sale of licenses. As product development is now at a significant more advanced stage then it was, it is management’s
belief that the sufficient funding could be provided through the sale of licenses or a large-scale partnership that could bring in additional
funds and infrastructure to support the commercial growth ambitions of the company.
However, as a consequence of this funding
requirement being triggered without the funding bridge having been put in place by the filing date of these unaudited condensed consolidated
financial statements, Financial Accounting Standards Board’s (the “FASB”) Accounting Standards Codification (“ASC”)
205-40: “Going Concern”, requires that management recognize and disclose this point as an event which creates a substantial doubt as to
the Company’s ability to continue as a going concern for at least one year from the date of filing of these unaudited condensed
consolidated financial statements.
Following the receipt of the CE mark approval
in the EU, and in support of our plans for similar certification with the FDA in the U.S., our plan is to utilize the cash on hand to
continue establishing commercial manufacturing operations for the commercial supply of the sugarBEAT® device and sensor patches in
our target markets.
Management's strategic plans include the
following:
| |
– |
support the UK and EU launch of sugarBEAT®; |
| |
– |
obtaining further regulatory approval for the sugarBEAT® device in other countries such as the U.S.; |
| |
– |
exploring licensing and partnership opportunities in other territories; |
| |
– |
developing the sugarBEAT® device platform for commercialization across other applications; and |
| |
– |
pursue additional capital raising opportunities as and when required to further enhance our growth plans. |
NOTE 2 – BASIS OF PRESENTATION
(a)
Basis of presentation
The accompanying unaudited condensed consolidated
financial statements have been prepared pursuant to the rules and regulations of the SEC, and do not include all of the information and
footnotes required by U.S. generally accepted accounting principles (“U.S. GAAP”) for complete financial statements. However,
such information reflects all adjustments consisting of normal recurring accruals which are, in the opinion of management, necessary for
a fair statement of the financial condition and results of operations for the interim periods. The results for the three- and nine- months
ended December 31, 2022 are not indicative of annual results. The accompanying unaudited condensed consolidated financial statements have
been prepared in accordance with U.S. GAAP for interim financial information and with the instructions to Form 10-Q and Article 8 of Regulation
S-X. These unaudited condensed consolidated financial statements should be read in conjunction with the consolidated financial statements
and the notes thereto included in the Company’s Annual Report on Form 10-K for the year ended March 31, 2022, as filed with the
SEC.
The accompanying unaudited condensed consolidated
financial statements include the accounts of the Company and the Company’s subsidiaries. References to “we”, “us”,
“our”, or the “Company” refer to Nemaura Medical Inc. and its consolidated subsidiaries. The unaudited condensed
consolidated financial statements are prepared in accordance with U.S. GAAP, and all significant intercompany balances and transactions
have been eliminated in consolidation.
The functional currency for the majority of
the Company’s operations is the Great Britain Pound Sterling (“GBP”), and the reporting currency is the U.S. Dollar
(“USD”). Financial statements for foreign subsidiaries are translated into USD using period end exchange rates for assets
and liabilities and average exchange rates for each period for revenue, costs and expenses.
Reclassification - We have reclassified certain amounts as previously
disclosed within the March 31, 2022 consolidated balance sheets to conform to our current period presentation. The reclassification of
$440,196 from Other liabilities and accrued expenses to Foreign currency contract at March 31, 2022 has no impact to prior year net loss,
current quarter net loss or year-to-date net loss.
(b) – Summary of Significant Accounting Policies
Use of Estimates
The preparation of the financial statements in conformity
with U.S. GAAP requires management to make estimates and assumptions that affect the amounts reported in the financial statements and
accompanying notes. Actual results could differ materially from those estimates. The Company’s most significant estimates include
the useful life of intangible assets, valuation of foreign currency contract and valuation allowance on deferred tax assets.
Our estimates are often based on complex judgments,
probabilities and assumptions that management believes to be reasonable, but that are inherently uncertain and unpredictable. It is also
possible that other professionals, applying reasonable judgment to the same facts and circumstances, could develop and support a range
of alternative estimated amounts. For a complete discussion of our critical accounting policies, see the “Critical Accounting Policies”
section of the Management’s Discussion & Analysis in our March 31, 2022 Form 10-K.
Cash and Cash Equivalents
Cash includes cash deposited in major financial institutions
in the United Kingdom. The Company’s cash balances exceed amounts covered by the Financial Services Compensation scheme. The Company
has never suffered a loss due to such excess balances.
The Company considers highly liquid investments with
maturities of three months or less from the date of purchase to be cash equivalents. These investments are carried at cost, which approximates
fair value. As of December 31, 2022 and March 31, 2022, the Company had no cash equivalents.
Revenue Recognition
The Company recognizes revenue when obligations under
the terms of a contract with a customer are satisfied; generally this occurs with the transfer of control or access of the Company’s
licenses or performance of services. Revenue is measured as the amount of consideration the company expects to receive in exchange for
transferring goods or providing services. A performance obligation is a promise in a contract to transfer a distinct good or service to
the customer, and is the unit of account in the contract. A contract’s transaction price is allocated to each distinct performance
obligation and recognized as revenue when, or as, the performance obligation is satisfied.
Contracts with customers consist of licensing arrangements
and, to a lesser extent, research and development related services. Revenues from licensing and royalty fees are received from the granting
of exclusive sales, marketing, manufacturing and distribution rights associated with the Company’s functional intellectual property
(IP). The Company’s performance obligation is satisfied at a point in time (upon delivery to the customer), where the Company has
no remaining obligation to support or maintain the intellectual property licensed to the customer. The Company typically requires a non-refundable
license fee, paid upfront.
Revenue from license fees are recognized at a point
in time when the Company transfers the functional IP to the customer as long as management believes the total consideration owed by the
customer for the license fee is probable of being received.
The Company’s contracts do not include multiple performance obligations
or variable consideration. Since the Company’s revenue is generated from a small number of customer contracts, the Company does
not have material contract assets or liabilities.
Fair
value of financial instruments
In accordance with the FASB ASC 820, “Fair
Value Measurements and Disclosures,” the Company determines the fair value of financial instruments with the use of observable inputs
and minimize the use of unobservable inputs when measuring fair value. ASC 820 establishes a fair value hierarchy based on the level of
independent, objective evidence surrounding the inputs used to measure fair value. A financial instrument’s categorization within
the fair value hierarchy is based upon the lowest level of input that is significant to the fair value measurement. ASC 820 prioritizes
the inputs into three levels that may be used to measure fair value:
Level 1: Applies to assets or liabilities
for which there are quoted prices in active markets for identical assets or liabilities.
Level 2: Applies to assets or liabilities
for which there are inputs other than quoted prices that are observable for the asset or liability such as quoted prices for similar assets
or liabilities in active markets; quoted prices for identical assets or liabilities in markets with insufficient volume or infrequent
transactions (less active markets); or model-derived valuations in which significant inputs are observable or can be derived principally
from, or corroborated by, observable market data.
Level 3: Applies to assets or liabilities
for which there are unobservable inputs to the valuation methodology that are significant to the measurement of the fair value of the
assets or liabilities.
Intangible Assets
The Company’s intangible assets consist of patents
relating to the sensor and algorithm that are granted in some territories, and pending still in others. The Company also plans to file
further patents as the opportunity arises. The cost of issued patents is capitalized and amortized over the life of the patents which
is 20 years. The costs of patents in development are expensed as incurred. Any unamortized costs previously capitalized associated with
patents that have expired or have been abandoned are written off as an impairment loss. The company has also capitalized certain software
development costs which are regularly reviewed to ensure that if development has been abandoned, costs are written off as an impairment
loss.
Share-Based Payments
The Company measures the cost of services received
in exchange for an award of equity instruments to employees and nonemployees based on the grant date fair value of the award, which is
recognized as compensation expense over the vesting term.
Income Taxes
The Company accounts for income taxes under the asset
and liability method, which requires the recognition of deferred tax assets and liabilities for the expected future tax consequences of
events that have been included in the financial statements. Under this method, deferred tax assets and liabilities are determined on the
basis of the differences between the financial statement and tax bases of assets and liabilities using enacted tax rates in effect for
the year in which the differences are expected to reverse. The effect of a change in tax rates on deferred tax assets and liabilities
is recognized in income in the period that includes the enactment date.
The Company recognizes deferred tax assets to the
extent that management believes these assets are more likely than not to be realized. In making such a determination, management considers
all available positive and negative evidence, including future reversals of existing taxable temporary differences, projected future taxable
income, tax-planning strategies, and results of recent operations. If we determine that the Company would be able to realize deferred
tax assets in the future in excess of their net recorded amount, the Company would make an adjustment to the deferred tax asset valuation
allowance, which would reduce the provision for income taxes.
(c) Recently adopted accounting pronouncements
Accounting standard
updates issued but not yet added were assessed and determined to be either not applicable or not expected to have a material impact on
our unaudited condensed consolidated financial statements.
NOTE 3 – LICENSING AGREEMENTS
United Kingdom and the Republic of Ireland,
the Channel Islands, and the Isle of Man
In March 2014, the Company entered into an Exclusive
Marketing Rights Agreement (the “Marketing Rights Agreement”) with an unrelated third party (the “Licensee”),
that granted to the Licensee the exclusive right to market and promote the sugarBEAT®
device and related patches under its own brand in the UK and the Republic of Ireland, the Channel Islands, and the Isle of Man. The Company
received a non-refundable, up-front cash payment of GBP 1,000,000 (approximately $1.20 million and $1.31 million as of December 31, 2022
and March 31, 2022, respectively), upon signing the Marketing Rights Agreement. The upfront payment received from the Marketing Rights
Agreement has been deferred and will be recorded as income over the term of the Marketing Rights Agreement, which commenced upon the first
delivery of the sugarBEAT® device to the Licensee in December 2021. Consequently, approximately
$70,000, and $259,000 is included in deferred revenue classified as a current liability as of December 31, 2022 and March 31, 2022, respectively,
with the remainder being shown in the non-current portion of deferred revenue.
NOTE 4 – RELATED PARTY TRANSACTIONS
DDL has a service agreement with Nemaura Pharma
Limited (“Pharma”), an entity controlled by the Company’s President and Chief Executive officer, to provide development,
manufacture, and regulatory approval process under Pharma’s ISO13485 accreditation. Pharma invoices DDL for these services on a
cost-plus basis.
The table below provides a summary of activity
between the Company and Pharma for the nine months ended December 31, 2022 and 2021, and the year ended March 31, 2022.
| Schedule of related party transactions | |
| | | |
| | | |
| | |
| | |
Nine Months Ended December 31, 2022 (unaudited) | | |
Nine Months Ended December 31, 2021 (unaudited) | | |
Year Ended March 31, 2022 | |
| Due to (from) related parties at beginning of period | |
$ | (101,297 | ) | |
$ | 148,795 | | |
$ | 148,795 | |
| Amounts invoiced by Pharma to DDL | |
| 2,833,546 | | |
| 2,114,801 | | |
| 3,245,985 | |
| Amounts invoiced by DDL to Pharma | |
| (3,159 | ) | |
| (2,495 | ) | |
| (2,495 | ) |
| Amounts paid by DDL to Pharma | |
| (2,785,487 | ) | |
| (2,316,544 | ) | |
| (3,492,962 | ) |
| Foreign exchange differences | |
| 31,077 | | |
| (97,149 | ) | |
| (620 | ) |
| Due to (from) related parties at end of period | |
$ | (25,320 | ) | |
$ | (152,592 | ) | |
$ | (101,297 | ) |
NOTE 5 – NOTES PAYABLE
NOTE PURCHASE AGREEMENT 1
On April 15, 2020, the Company entered into a note
purchase agreement (the “Note Purchase Agreement 1”) by and among the Company, DDL, TCL and a third-party investor (the “Investor”).
Pursuant to the terms of Note Purchase Agreement 1,
the Company agreed to issue and sell to the Investor, and the Investor agreed to purchase from the Company, a secured promissory note
(the “2020 Secured Note”) in the original principal amount of $6,015,000. In consideration thereof, on April 15, 2020, (i)
the Investor (a) paid $1,000,000 in cash, (b) issued to the Company (1) Investor Note #1 in the principal amount of $2,000,000 (“Investor
Note #1”), and (2) Investor Note #2 in the principal amount of $2,000,000 (“Investor Note #2” and together with Investor
Note #1, the “2020 Investor Notes”), and (ii) the Company delivered the 2020 Secured Note on behalf of the Company, to the
Investor, against delivery of the 2020 Purchase Price. For these purposes, the “2020 Purchase Price” means the Investor’s
initial cash purchase price, together with the sum of the initial principal amounts of the Investor Notes.
The 2020 Secured Note is secured by the Collateral
(as hereinafter defined). The 2020 Secured Note carries an original issue discount (“OID”) of $1,000,000 (16.7%). In addition,
the Company agreed to pay $15,000 to the Investor to cover the Investor’s legal fees, accounting costs, due diligence, monitoring
and other transaction costs incurred in connection with the purchase and sale of the 2020 Secured Note (the “Transaction Expense
Amount”). In addition to this, a payment of $325,000 was made to Ascendiant Capital Markets, LLC (“Ascendiant”) for
structuring the agreement between both parties. The 2020 Purchase Price for the 2020 Secured Note is $4,675,000, computed as follows:
$6,015,000 original principal balance, less: OID, Transaction Expense Amount, and commission paid.
The borrowing period is 24 months, and the Company
shall pay the outstanding balance and all fees on maturity. A monitoring fee equal to 0.833% of the outstanding balance will automatically
be added to the outstanding balance on the first day of each month. The debt less the discount and transaction expenses will be accreted
over the term of the 2020 Secured Note using the effective interest method.
Security Agreement
On April 15, 2020, the Company entered into the Security
Agreement by the Company, DDL and TCL, in favor of the Investor (the “2020 Security Agreement”). Pursuant to the terms of
the 2020 Security Agreement, the Company granted the Investor a first-priority security interest in all rights, title, interest, claims
and demands of the Company in and to all of the Company’s patents and all other proprietary rights, and all rights corresponding
to the Company’s patents throughout the world, now owned and existing, and all replacements, proceeds, products, and accessions
thereof (the “Collateral”).
Note Purchase Agreement 1 was settled in full on April
22, 2022.
NOTE PURCHASE AGREEMENT 2
On February 8, 2021, the Company entered
into an additional note purchase agreement (“Note Purchase Agreement 2”) with the Investor. Pursuant to the terms of
Note Purchase Agreement 2, the Company agreed to issue and sell to the Investor, and the Investor agreed to purchase from the Company,
a secured promissory note (the “Secured Note 2”) in the original principal amount of $24,015,000. The Secured Note 2 carries
an OID of $4,000,000 (16.7%), and the Company agreed to pay $15,000 to the Investor to cover the Investor’s transaction expenses.
In addition to this, a commission of $1,200,000 was also payable to Ascendiant.
In consideration thereof, on February
9, 2021, (i) the Investor paid $20,000,000 in cash to the Company, and (ii) the Company delivered Secured Note 2 on behalf of the Company,
to the Investor, against the delivery of the 2021 Purchase Price. For these purposes, the “2021 Purchase Price” means
the Investor’s initial cash purchase price. After adjusting for transaction expenses of $1,200,000, cash proceeds received
were $18,800,000.
The borrowing terms for Note Purchase
Agreement 2 were originally consistent with those of Note Purchase Agreement 1, with the borrowing period being 24 months from the date
of the agreement, the Company being required to pay the outstanding balance and all fees on maturity, and a monitoring fee equal to 0.833%
of the outstanding balance being automatically added to the outstanding balance on the first day of each month. The debt less discount
and transaction expenses will be accreted over the term of the Secured Note 2 using the effective interest rate method.
On October 21, 2022, the Company entered into
an amendment to Note Purchase Agreement 2. Pursuant to the terms of the amendment, the Company and Investor agreed to extend the maturity
date of Note Purchase Agreement 2 to July 1, 2024. In consideration thereof, the Company agreed to pay to the Investor an extension fee
in the amount of 5% of the outstanding balance of Note Purchase Agreement 2, which resulted in $813,834 being added onto the liability
due to the Investor.
The Company and the Investor previously agreed
to reduce the maximum monthly redemption amount from $2,000,000 to $500,000 from June 2022 to February 2023, which reduction remains in
force. Pursuant to the terms of the amendment, the Company and Investor agreed to reduce the maximum monthly redemption amount during
the period beginning March 2023 until Note Purchase Agreement 2 is paid in full from $2,000,000 to $1,000,000; provided, however, that
upon the occurrence of an event of default under the Note Purchase Agreement 2, the maximum monthly redemption amount will automatically
be increased back to $2,000,000.
Security Agreement
On February 8, 2021, the 2020 Security Agreement was
extended to include Note Purchase Agreement 2, which is also secured against all of the Company’s assets owned as of February 9,
2021 and extends to any assets acquired at any time that the Company’s obligations under Secured Note 2 are outstanding.
NOTE PURCHASE AGREEMENT 3
On
May 20, 2022, the Company entered into a new note purchase agreement (“Note Purchase Agreement 3”) by and among the Company,
DDL, TCL and a third-party investor.
Pursuant
to the terms of the Note Purchase Agreement 3, the Company agreed to issue and sell to the Investor and the Investor agreed to purchase
from the Company a secured promissory note (the “Secured Note”) in the original principal amount of $6,015,000. In consideration
thereof, on May 20, 2022 (the closing date), (i) the Investor paid $5,000,000 in cash, and (ii) the Company delivered the Secured
Note on behalf of the Company, to the Investor, against delivery of the Purchase Price. For these purposes, the “Purchase Price”
means the Investor’s initial cash purchase price.
The
Secured Note is secured by the Collateral (as hereinafter defined). The Secured Note carries an original issue discount (“OID”)
of $1,000,000 (16.7%). In addition, the Company agreed to pay $15,000 to the Investor to cover the Investor’s legal fees,
accounting costs, due diligence, monitoring and other transaction costs incurred in connection with the purchase and sale of the Secured
Note (the “Transaction Expense Amount”). In addition to this, a payment of $300,000 was made to Ascendiant Capital Markets,
LLC, (the “Commission”) for structuring the agreement between both parties. The Purchase Price for the Secured Note is $4,700,000,
computed as follows: $6,015,000 original principal balance, less: OID, Transaction Expense Amount, and commission paid.
The
borrowing period is 24 months, and the Company shall pay the outstanding balance and all fees on maturity. A monitoring fee
equal to 0.833% of the outstanding balance will automatically be added to the outstanding balance on the first day of each month.
The debt less the discount and transaction expenses will be accreted over the term of the Note using the effective interest method.
Security
Agreement
On
May 20, 2022, the Company entered into the Security Agreement by the Company, DDL and TCL, in favor of the Investor (the “Security
Agreement”). Pursuant to the terms of the Security Agreement, the Company granted the Investor a first-priority security interest
in all rights, title, interest, claims and demands of the Company in and to all of the Company’s patents and all other proprietary
rights, and all rights corresponding to the Company’s patents throughout the world, now owned and existing, and all replacements,
proceeds, products, and accessions thereof. As of December 31, 2022, long-term debt matures as follows:
| Schedule of long term debt |
|
|
|
|
| |
|
|
Notes Payable
($) |
|
| |
Within 12 months |
|
|
|
11,512,711 |
|
| |
Within 24 months |
|
|
|
8,557,548 |
|
| |
|
|
|
|
20,070,259 |
|
NOTE 6 – STOCKHOLDERS’
(DEFICIT) EQUITY
During the three month period ended December 31, 2022,
shares were sold under the ATM Equity Distribution Agreement in place with H.C. Wainwright & Co., for total gross proceeds of $696,
with associated costs of $273. No other shares were issued during the nine month period ended December 31, 2022.
During the nine month period ended December 31, 2021,
366,892 warrants were exercised generating gross proceeds of $2,963,658. There were a total of 1,573,098 warrants outstanding at this
date. During the three month period ended December 31, 2021, 22,524 shares were sold under the ATM Equity Distribution Agreement in place
with H.C. Wainwright & Co., for total gross proceeds of $118,791, with associated costs of $4,382. No other shares were issued during
the three and nine month periods ended December 31, 2021.
Loss per share
The following table sets forth the computation
of basic and diluted loss per share for the periods indicated.
| Schedule of earnings (loss) per share | |
| | | |
| | | |
| | | |
| | |
| | |
Three Months Ended
December 31, | | |
Nine Months Ended
December 31, | |
| | |
2022 | | |
2021 | | |
2022 | | |
2021 | |
| | |
| (in Dollars, except Share Amounts) | | |
| (in Dollars, except Share Amounts) | |
| Net loss attributable to common stockholders | |
| (1,716,278 | ) | |
| (3,431,568 | ) | |
| (9,460,888 | ) | |
| (10,269,557 | ) |
| Weighted average basic and diluted shares outstanding | |
| 24,103,196 | | |
| 23,313,629 | | |
| 24,102,976 | | |
| 23,244,345 | |
| Basic and diluted loss per share: | |
| (0.07 | ) | |
| (0.15 | ) | |
| (0.39 | ) | |
| (0.44 | ) |
The Company excludes warrants outstanding,
which are anti-dilutive given the Company is in a loss position, from the basic and diluted loss per share calculation.
Basic loss per share is computed by dividing
loss available to common stockholders by the weighted-average number of common shares outstanding during the period. For the three and
nine month periods ended December 31, 2022, warrants to purchase 1,573,098 shares of common stock and a unit purchase option to purchase
9,710 shares of common stock, as well as warrants to purchase 9,710 shares of common stock, were considered anti-dilutive and were excluded
from the calculation of diluted loss per share. For the three and nine month periods ended December 31, 2021, warrants to purchase 1,573,098
shares of common stock and a unit purchase option to purchase 9,710 shares of common stock, as well as warrants to purchase 9,710 shares
of common stock, were considered anti-dilutive and were also excluded from the calculation of diluted loss per share.
NOTE 7 – OTHER ITEMS
The outbreak of COVID-19 in December 2019 has
since rapidly increased its exposure globally. On March 11, 2020, the World Health Organization declared the outbreak a pandemic. We continue
to monitor the impact of COVID-19 on our own operations and are working with our employees, suppliers and other stakeholders to mitigate
the risks posed by its spread, but COVID-19 is not expected to have any long-term detrimental effect on the Company’s success. While
key suppliers have not been accessible throughout the whole period of the outbreak, we have been able to be flexible in our priorities
and respond favorably to the challenges faced during the outbreak. We have also seen a surge in the uptake of technologies for remote
monitoring of patients and patient self-monitoring, which potentially enhances the prospects for the Company, its CGM product and its
planned digital healthcare offering.
NOTE 8 – SUBSEQUENT EVENTS
Management has evaluated subsequent events and transactions
for potential recognition or disclosure in the financial statements through February 23, 2023, the date these financial statements were
available to be issued.
The Company commenced a Registered Direct
Offering and concurrent Private Placement on January 27, 2023 with two healthcare-focused U.S. institutional investors to sell 4,796,206
shares of its common stock, pursuant to a registered direct offering and warrants to purchase up to 4,796,206
shares in a concurrent private placement. The combined purchase price for one share and one warrant was $1.75.
The warrants have an exercise price of $2.00
per share and are initially exercisable at the later of shareholder approval or six months following the date of issuance and will
expire 5.5 five and a half years from January 31, 2023, the closing date. The aggregate gross proceeds from the Registered Direct
Offering and the concurrent Private Placement were $8.4
million before deducting placement agent fees and other estimated offering expenses. The Company received net proceeds of $7,655,974
after costs.

NEMAURA MEDICAL, INC.
Up to 4,796,206
Shares of Common Stock
PROSPECTUS
__________, 2023
Through and including ,
2023 (the 40th day after the date of this offering), all dealers effecting transactions in these securities, whether or
not participating in this offering, may be required to deliver a prospectus. This is in addition to a dealer’s obligation to deliver
a prospectus when acting as an underwriter and with respect to an unsold allotment or subscription.
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
Item 13. Other Expenses of Issuance and Distribution.
Set forth below is an estimate (except in the case
of the SEC registration fee) of the amount of fees and expenses to be incurred in connection with the issuance and distribution of the
offered securities registered hereby, other than underwriting discounts and commission, if any, incurred in connection with the sale of
the offered securities. All such amounts will be borne by Nemaura Medical, Inc., a Nevada corporation.
| Type | |
Amount |
| SEC registration fee | |
$ | 1,057.08 | |
| Accounting fees and expenses* | |
| 20,000.00 | |
| Legal fees and expenses* | |
| 45,000.00 | |
| Miscellaneous fees and expenses* | |
| 1,000.00 | |
| Total expenses* | |
$ | 67,057.08 | |
* Estimated
Item 14. Indemnification of Directors and Officers.
We are a Nevada corporation and generally governed
by the Nevada Private Corporations Code, Title 78 of the Nevada Revised Statutes, or NRS.
Section 78.138 of the NRS provides that, unless
the corporation’s Articles of Incorporation provide otherwise, a director or officer will not be individually liable unless it is
proven that (i) the director’s or officer’s acts or omissions constituted a breach of his or her fiduciary duties, and (ii)
such breach involved intentional misconduct, fraud, or a knowing violation of the law. Our Articles of Incorporation provide that no director
or officer shall be personally liable to the corporation or any of its stockholders for damages for any breach of fiduciary duty as a
director or officer except (i) for acts or omissions that involve intentional misconduct or a knowing violation of law by the director,
(ii) for conduct violating the NRS, or (iii) for any transaction from which the director will personally receive a benefit in money, property,
or services to which the director is not legally entitled.
Section 78.7502 of the NRS permits a company
to indemnify its directors and officers against expenses, judgments, fines, and amounts paid in settlement actually and reasonably incurred
in connection with a threatened, pending, or completed action, suit, or proceeding, if the officer or director (i) is not liable pursuant
to NRS 78.138, or (ii) acted in good faith and in a manner the officer or director reasonably believed to be in or not opposed to the
best interests of the corporation and, if a criminal action or proceeding, had no reasonable cause to believe the conduct of the officer
or director was unlawful. Section 78.7502 of the NRS also precludes indemnification by the corporation if the officer or director has
been adjudged by a court of competent jurisdiction, after exhaustion of all appeals, to be liable to the corporation or for amounts paid
in settlement to the corporation, unless and only to the extent that the court determines that in view of all the circumstances, the person
is fairly and reasonably entitled to indemnity for such expenses and requires a corporation to indemnify its officers and directors if
they have been successful on the merits or otherwise in defense of any claim, issue, or matter resulting from their service as a director
or officer.
Section 78.751 of the NRS permits a Nevada
company to indemnify its officers and directors against expenses incurred by them in defending a civil or criminal action, suit, or proceeding
as they are incurred and in advance of final disposition thereof, upon determination by the stockholders, the disinterested board members,
or by independent legal counsel. Section 78.751 of NRS requires a corporation to advance expenses as incurred upon receipt of an undertaking
by or on behalf of the officer or director to repay the amount if it is ultimately determined by a court of competent jurisdiction that
such officer or director is not entitled to be indemnified by the company if so provided in the corporations articles of incorporation,
bylaws, or other agreement. Section 78.751 of the NRS further permits the company to grant its directors and officer’s additional
rights of indemnification under its articles of incorporation, bylaws, or other agreement.
Section 78.752 of the NRS provides that a Nevada company may purchase
and maintain insurance or make other financial arrangements on behalf of any person who is or was a director, officer, employee, or agent
of the company, or is or was serving at the request of the company as a director, officer, employee, or agent of another company, partnership,
joint venture, trust, or other enterprise, for any liability asserted against him and liability and expenses incurred by him in his capacity
as a director, officer, employee, or agent, or arising out of his status as such, whether or not the company has the authority to indemnify
him against such liability and expenses.
The foregoing discussion of indemnification
merely summarizes certain aspects of indemnification provisions and is limited by reference to the above discussed sections of the NRS.
Our By-Laws implement the indemnification provisions
permitted by Chapter 78 of the NRS by providing that we shall indemnify our directors and officers to the fullest extent and under all
circumstances permitted by the NRS against expense, liability, and loss reasonably incurred or suffered by them in connection with their
service as an officer or director. Our By-Laws also provide that we will indemnify our directors and officers to the fullest extent permitted
by the NRS and shall advance reasonable costs and expenses incurred with respect to any proceeding to which a person is made a party as
a result of being a director or officer in advance of final disposition of such proceeding without regard to any limitations set forth
in the NRS 78.7502. Insofar as indemnification by our company for liabilities arising under the Securities Act may be permitted to officers
and directors of our company pursuant to the foregoing provisions or otherwise, we are aware that in the opinion of the SEC, such indemnification
is against public policy as expressed in the Securities Act and is, therefore, unenforceable.
We may purchase and maintain liability insurance,
or make other arrangements for such obligations or otherwise, to the extent permitted by the NRS.
At the present time, there is no pending litigation
or proceeding involving a director, officer, employee, or other agent of ours in which indemnification would be required or permitted.
We are not aware of any threatened litigation or proceeding that may result in a claim for such indemnification.
Item 15. Recent Sales of Unregistered Securities
In the three
years preceding the filing of this registration statement, we have issued the following securities that were not registered under the
Securities Act. No underwriters were involved in the sales and the certificates representing the securities sold and issued contain legends
restricting transfer of the securities without registration under the Securities Act or an applicable exemption from registration.
On January 27, 2023, we issued
to two accredited investors warrants to purchase an aggregate of 4,796,206 shares of Common Stock at an exercise price per share of $2.00
(the “Warrants”).
The Warrants and the shares
issuable upon exercise of the Warrants have not been registered under the Securities Act of 1933, as amended (the “Securities Act”),
and are instead were offered and sold pursuant to the exemption provided in Section 4(a)(2) under the Securities Act and Rule 506(b) under
Regulation D promulgated thereunder. The recipients of the securities in the above transaction represented their intentions to acquire
the securities for investment only and not with a view to or for sale in connection with any distribution thereof, and appropriate legends
were placed upon the stock certificates issued in these transactions.
Item 16. Exhibits.
The following documents are filed as exhibits to this registration
statement:
| Exhibit No. |
|
Description |
|
Location |
| 3.1 |
|
Articles of Incorporation filed December 24, 2013 |
|
Incorporated by reference from the registrant's registration statement on Form S-1 (File No. 333-194857), filed March 28, 2014 |
| 3.2 |
|
Certificate of Amendment to the Articles of Incorporation |
|
Incorporated by reference from the registrant’s Annual Report on Form 10-K for the fiscal year ended March 31, 2018, filed June 12, 2018 |
| 3.3 |
|
Certificate
of Designation for Series A Convertible Preferred Stock
|
|
Incorporated by reference from the registrant’s Annual Report on Form 10-K for the fiscal year ended March 31, 2018, filed with the SEC on June 12, 2018 |
| 3.4 |
|
Certificate of Change |
|
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on December 4, 2019 |
| 3.5 |
|
Amended and Restated Bylaws |
|
Incorporated by reference from the registrant’s Annual Report on Form 10-K for the fiscal year ended March 31, 2018, filed June 12, 2018 |
| 4.1 |
|
Form of Subscription Agreement |
|
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on December 2, 2015 |
| Exhibit No. |
|
Description |
|
Location |
| 4.2 | |
Common Stock Purchase Warrant by and between Nemaura Medical, Inc. and Dr. Dallas John Burston, dated November 26, 2015 | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on December 2, 2015 |
| 4.3 | |
Form of warrant | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on December 26, 2018 |
| 4.4 | |
Form of Warrant | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on July 30, 2020 |
| 4.5 | |
Form of Warrant | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on January 31, 2023. |
| 5.1 | |
Opinion of Anthony L.G., PLLC | |
Filed herewith |
| 10.1 | |
Employment Agreement between the Company and Dewan F.H. Chowdhury, dated November 1, 2013
| |
Incorporated by reference from the registrant's registration statement on Form S-1 (File No. 333-194857), filed with SEC on March 28, 2014 |
| 10.2 | |
Exclusive Rights License Agreement between Dallas Burston Pharma (DBP) Jersey Limited and Dermal Diagnostics Limited, dated March 31, 2014 | |
Incorporated by reference from the registrant's registration statement on Form S-1/A (File No. 333-194857), filed with SEC on July 11, 2014 |
| 10.4 | |
Assignment Agreement between Nemaura Pharma Limited and Dermal Diagnostics Limited | |
Incorporated by reference from the registrant's Registration Statement on Form S-1/A (File No. 333-194857), filed with SEC on July 11, 2014 |
| 10.5+ | |
License, Supply and Distribution Agreement between Nemaura Medical, Inc. and Dallas Burston Pharma (Jersey) Limited dated November 26, 2015 | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on December 2, 2015 |
| 10.6 | |
Form of Common Stock Exchange Agreement | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed on November 7, 2017 |
| 10.7+ | |
Joint Collaboration Agreement, between Dallas Burton Ethitroniox (Europe) Limited and Nemaura Medical, Inc., dated May 21, 2018 | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on May 25, 2018 |
| 10.8 | |
Placement Agency Agreement between Nemaura Medical, Inc. and Dawson James Securities, Inc., dated December 18, 2018 | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on December 26, 2018 |
| 10.9 | |
Warrant Agency Agreement between Nemaura Medical, Inc. and Island Stock Transfer, dated December 20, 2018 | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on December 26, 2018 |
| 10.10 | |
Note Purchase Agreement among Nemaura Medical, Inc., Dermal Diagnostics Limited, Trial Clinic Limited, and Chicago Venture Partners, L.P. dated April 15, 2020 | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on April 21, 2020 |
| 10.11 | |
Secured Promissory Note issued by Nemaura Medical, Inc. in favor of Dermal Diagnostics Limited, Trial Clinic Limited and Chicago Venture Partners, L.P. dated April 15, 2020 | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on April 21, 2020 |
| 10.12 | |
Investor Note #1 issued by Chicago Venture Partners, L.P. in favor of Nemaura Medical, Inc., Dermal Diagnostics Limited and Trial Clinic Limited dated April 15, 2020 | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on April 21, 2020 |
| 10.13 | |
Investor Note #2 issued by Chicago Venture Partners, L.P. in favor of Nemaura Medical, Inc., Dermal Diagnostics Limited and Trial Clinic Limited dated April 15, 2020 | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on April 21, 2020 |
| 10.14 | |
Security Agreement by Nemaura Medical, Inc., Dermal Diagnostics Limited and Trial Clinic Limited, in favor of Chicago Venture Partners, L.P. dated April 15, 2020 | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on April 21, 2020 |
| 10.15 | |
Warrant Agency Agreement, dated July 30, 2020, by and between Nemaura Medical, Inc. and Nevada Agency & Trust Company | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on July 30, 2020 |
| 10.16 | |
Employment Agreement between Nemaura Medical, Inc. and Justin Mclarney dated September 15, 2020 | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on September 21, 2020 |
| Exhibit No. |
|
Description |
|
Location |
| 10.17 | |
Healthimation License Agreement dated as of September 16, 2020 by and between Healthimation, LLC and Nemaura Medical, Inc. | |
Incorporated by reference from the restaurant’s Current Report on Form 8-K filed with the SEC on November 2, 2020 |
| 10.18 | |
Amendment, dated as of October 23, 2020, to Healthimation License Agreement by and between Healthimation, LLC and Nemaura Medical, Inc. | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on November 2, 2020 |
| 10.19 | |
Note Purchase Agreement between Nemaura Medical, Inc. and Uptown Capital, LLC dated February 8, 2021 | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on February 11, 2021 |
| 10.20 | |
Secured Promissory Note of Nemaura Medical, Inc. issued to Uptown Capital, LLC dated February 8, 2021. | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on February 11, 2021 |
| 10.21 | |
Security Agreement between Nemaura Medical, Inc. and Uptown Capital, LLC dated February 8, 2021. | |
Incorporated by reference from the registrant’s Current Report on Form 8-K filed with the SEC on February 11, 2021 |
| 10.22 | |
At The Market Offering Agreement, dated July 23, 2021, by and between Nemaura Medical, Inc. and H.C. Wainwright & Co., LLC. | |
Incorporated by reference from the registrant’s Current Report on Form 8-K Filed with the SEC on July 23, 2021. |
| 10.23 | |
License, Supply and Distribution Agreement, entered into on September 24, 2021 and dated as of September 17, 2021, by and between Nemaura Medical, Inc. and MySugarWatch Duopack Limited. | |
Incorporated by reference from the registrant’s Current Report on Form 8-K Filed with the SEC on September 30, 2021. |
| 10.24 | |
Statement of Main Terms of Employment dated January 5, 2022 with Arash Ghadar. | |
Incorporated by reference from the registrant’s Current Report on Form 8-K Filed with the SEC on January 10, 2022. |
| 10.25 | |
Form of Securities Purchase Agreement between Nemaura Medical, Inc. and certain purchasers dated January 27, 2023. | |
Incorporated by reference from the registrant’s Current Report on Form 8-K Filed with the SEC on January 31, 2023. |
| 10.26 | |
Placement Agent Agreement between Nemaura Medical, Inc. and EF Hutton, division of Benchmark Investments, LLC dated January 27, 2023. | |
Incorporated by reference from the registrant’s Current Report on Form 8-K Filed with the SEC on January 31, 2023. |
| 21.1 | |
Subsidiaries of the Registrant. | |
Incorporated by reference from the registrant’s Annual Report on Form 10-K for the year ended March 31, 2022. |
| 23.1 | |
Consent of Mayer Hoffman McCann P.C., an independent registered public accounting firm | |
Filed herewith. |
| 23.2 | |
Consent of Anthony L.G., PLLC | |
Included in Exhibit 5.1. |
| 24.1 | |
Power of Attorney | |
Included on the signature page. |
| 107 | |
Filing Fee Table | |
Filed herewith. |
† Includes management contracts and compensation plans and arrangements.
+ Confidential treatment of certain portions of this exhibit has been granted
by the Securities and Exchange Commission.
Item 17. Undertakings.
(b) The undersigned registrant
hereby undertakes that, for purposes of determining any liability under the Securities Act, each filing of the registrant’s annual
report pursuant to Section 13(a) or Section 15(d) of the Exchange Act (and, where applicable, each filing of an employee benefit plan’s
annual report pursuant to Section 15(d) of the Exchange Act) that is incorporated by reference in the registration statement shall be
deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time
shall be deemed to be the initial bona fide offering thereof.
(h) Insofar as indemnification
for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the registrant pursuant
to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the SEC such indemnification is against
public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against
such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of
the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person
in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled
by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public
policy as expressed in the Securities Act and will be governed by the final adjudication of such issue.
(i) The undersigned registrant
hereby undertakes that:
(1) For purposes of determining
any liability under the Securities Act of 1933, the information omitted from the form of prospectus filed as part of this registration
statement in reliance upon Rule 430A and contained in a form of prospectus filed by the registrant pursuant to Rule 424(b)(1) or (4) or
497(h) under the Securities Act shall be deemed to be part of this registration statement as of the time it was declared effective.
(2)
For the purpose of determining any liability under the Securities Act of 1933, each post-effective amendment that contains a form of prospectus
shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at
that time shall be deemed to be the initial bona fide offering thereof.
SIGNATURES
Pursuant to the requirements of the Securities Act
of 1933, as amended, the registrant certifies that it has reasonable grounds to believe that it meets all of the requirements for filing
on Form S-1 and has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized in
the city of New York, New York on March 13, 2023.
| |
NEMAURA MEDICAL, INC.,
a Nevada corporation |
| |
|
|
| |
By: |
/s/ Dewan F. H. Chowdhury |
| |
|
Dewan F. H. Chowdhury, |
| |
|
Chief Executive Officer |
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that each
person whose signature appears below constitutes and appoints Mr. Dewan F. H. Chowdhury, his true and lawful attorney-in-fact and agent,
with full power of substitution and resubstitution, for him and in his name, place and stead, in any and all capacities, to sign any and
all amendments (including post-effective amendments) to this Registration Statement and to sign any and all additional registration statements
relating to the Registration Statement and filed pursuant to Rule 462(b) of the Securities Act of 1933, as amended, and to file the same,
with all exhibits thereto, and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said
attorney-in-fact and agent or his substitute or substitutes, full power and authority to do and perform each and every act and thing requisite
and necessary to be done in connection therewith, as fully to all intents and purposes as he might or could do in person, hereby ratifying
and confirming all that said attorney-in-fact and agent or his substitute or substitutes, may lawfully do or cause to be done by virtue
hereof.
Pursuant to the requirements of the Securities
Act of 1933, as amended, this registration statement has been signed by the following persons in the capacities and on the dates indicated.
| Name |
|
Position |
|
Date |
| |
|
|
|
|
| /s/ Dewan F.H. Chowdhury |
|
President, Chief Executive Officer, Interim Chief Financial Officer and Director |
|
March 13, 2023 |
|
Dewan F.H. Chowdhury
|
|
(Principal Executive Officer and Principal Financial and Accounting Officer)
|
|
|
| |
|
|
|
|
| /s/ Bashir Timol |
|
Director |
|
March 13, 2023 |
|
Bashir Timol
|
|
|
|
|
| /s/ Timothy Johnson |
|
Director |
|
March 13, 2023 |
|
Timothy Johnson
|
|
|
|
|
| /s/ Salim Natha |
|
Director |
|
March 13, 2023 |
|
Salim Natha
|
|
|
|
|
| /s/ Thomas Moore |
|
Director |
|
March 13, 2023 |
| Thomas Moore |
|
|
|
|
Nemaura Medical (NASDAQ:NMRD)
Historical Stock Chart
From Jun 2024 to Jul 2024
Nemaura Medical (NASDAQ:NMRD)
Historical Stock Chart
From Jul 2023 to Jul 2024